Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement
|
|
|
- Jacob Houston
- 6 years ago
- Views:
Transcription
1 Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation Final Draft Karen Costiloe, RPh. Paula Mialon, Pharm.D., Facilitator WPPD Program-University of Florida
2 Outcomes Literature Evaluation Treatment Strategies for the Prevention of Ischemic Complications in Patients undergoing Percutaneous Coronary Intervention Karen Costiloe, RPh Introduction Coronary stenting has evolved into the most frequent revascularization procedure performed in cardiac catheterization labs across the world, and is used in over 80% of all procedures. 1 The primary complication associated with stent placement is thrombosis in the target vessel, which can result in death, myocardial infarction (MI), or the need for emergency revascularization by means of coronary artery bypass graft (CABG) or a repeat percutaneous coronary intervention (PCI). Variables associated with increased mortality in patients undergoing PCI include advanced age, female gender, diabetes, prior MI, multivessel disease, a large area of myocardium at risk, pre-existing impairment of left ventricular (LV) function, impaired renal function, and collateral vessels supplying significant areas of myocardium that originate distal to the segment to be dilated. 2-3 Stents have been shown to reduce both the risk of major complications and late-term restenosis. 4 Several antiplatelet strategies have been studied in the prevention of ischemic complications in patients undergoing PCI with stent placement. Glycoprotein IIb/IIIa(GP IIb/IIIa) inhibitors, including abciximab, eptifibatide, and tirofiban, inhibit the final common pathway for platelet aggregation, and have been widely studied in this setting. Each agent differs in regard to duration of action at the platelet target receptor, receptor affinity, and binding sites on the GP IIb/IIIa receptor. Abciximab binds to at least two other integrins expressed in endothelial cells and monocytes. They also differ in regard to cost. 5 The first of the GP IIb/IIIa inhibitors, abciximab was studied in the EPIC trial, and found significantly improved outcomes with regard to the composite endpoint of death, MI, and urgent target vessel revascularization (UTVR), including long term survival benefits. 6 That study also found a significantly higher incidence of bleeding complications which led to the now reduced dose of heparin used with GP IIb/IIIa inhibitors. 7 Two other trials, IMPACT II (eptifibatide) and RESTORE (tirofiban), did not find statistically significant differences in the composite endpoint in patients undergoing balloon angioplasty or atherectomy, though there were trends toward a decrease in events. 8-9 Since these earlier trials were conducted, thienopyridines have been added to the arsenal of antiplatelet therapy. Thienopyridines such as clopidogrel and ticlopidine have been shown to have a synergistic anti-platelet effect when given with aspirin, and the combination has been shown to be superior to aspirin alone or aspirin plus oral anticoagulation in patients receiving stents
3 Lastly, a direct thrombin inhibitor, bivalirudin, has been studied as an option that may be as effective as heparin plus GP IIb/IIIa inhibitors with less incidence of major and minor bleeding as well as decreased cost. Each of these strategies is not without adverse effects, the foremost of which is major and minor bleeding complications. Several questions remain to be answered. Is one agent more effective than another? Is there a survival benefit related to the use of these agents with stenting procedures? Are there other agents available that would be equally as effective? The purpose of this paper is to review five published trials involving strategies of antiplatelet therapy to prevent ischemic complications in patients undergoing PCI. Evaluation of Studies ESPRIT, EPISTENT, and TARGET Since the EPIC trial, abciximab has been the preferred agent for many interventional cardiologists because it was the only agent to show survival benefit in the earlier trials. With the onset of stenting, several new trials were published including ESPRIT (eptifibatide), EPISTENT (abciximab) and TARGET (tirofiban and abciximab) There are many similarities between these three trials. Each included the composite primary endpoint of Death, MI, and UTVR at 30 days. In addition, ESPRIT also included GP IIb/IIIa bailout within 48hrs of procedure. All patients received aspirin as well as thienopyridine prior to the procedure. ESPRIT and EPISTENT were randomized, double-blind, placebo controlled trials of similar size. Each trial showed a significant decrease in the occurrence of the composite primary outcome measures as compared to placebo, with a rate of 5.3% in the stent/abciximab group (p<0.001) and 6.6% in the eptifibatide group (p=0.0015). There are also several distinct differences in the two trials. ESPRIT enrolled patients who were to undergo elective PCI, and in the opinion of the treating physician, would not routinely be treated with glycoprotein IIb/IIIa inhibitors. It was therefore a low risk patient population as compared to EPISTENT, which included a higher risk group of patients undergoing both elective and urgent PCI. ESPRIT utilized high inflation deployment stents, the standard of practice based on clinical trials to date, whereas EPISTENT utilized an older low-inflation deployment stent. 4 Although the event rate for abciximab/stent appears lower than that of eptifibatide, there are several reasons why primary outcome measures may have been under-reported in the EPISTENT trial. First, creatinine kinase MB isoenzymes (CK-MB) were not routinely monitored for the first 24hrs after PCI, therefore events may have been missed, particularly non-q MI s. Second, the investigators defined MI as CK-MB of 5X the upper limit of normal (ULN). ACC/AHA guidelines recommend 3X the ULN for identifying an MI after PCI. 4 Lastly, 30.3% of patients in the placebo group had >1 stent placed vs 26% in the abciximab group. The ESPRIT trial may
4 also have missed primary outcome measures. While their criteria for MI included CK-MB 3X ULN as their threshold for MI, they also chose not to routinely monitor CK-MB. Also the use of GP IIb/IIIa bailout may have decreased the event rate in the placebo arm. Interestingly, the greatest decrease in the primary event rate in the EPISTENT trial occurred in men (10.5% placebo vs 4.2% stent plus abciximab (SA) p<0.001), diabetics (12.1% placebo vs 5.6% SA p=0.04) and patients <65 years of age (10.2% placebo vs 3.6% SA). TARGET is the only head-to-head trial of two GP IIb/IIIa inhibitors, abciximab and tirofiban, which enrolled more than twice the number of patients as the previous two trials. 14 The study utilized the higher dose of tirofiban from the RESTORE trial rather than the current Food and Drug Administration (FDA) approved dose, and enrolled a higher risk patient population, those undergoing both elective and urgent PCI, similar to EPISTENT. Of the trials evaluated, TARGET is the only trial to measure CK-MB routinely during the first 24hrs following PCI utilizing 3X the ULN as the threshold for MI. This may have accounted for a slightly higher event rate for abciximab, 6%, vs. that of 5.3% found in the EPISTENT trial. The investigators attempted to prove the noninferiority of tirofiban to abciximab, however the difference in the occurrence of the primary composite endpoint was statistically significant, 7.6% for tirofiban and 6% for abciximab (p=0.038) showing abciximab to be superior. Most of the benefit was derived from a decrease in non-fatal MI (tirofiban 6.9% vs abciximab 5.4% p=0.04). None of the GP IIb/IIIa trials showed a statistically significant difference in the incidence of death. All three trials demonstrated that a large portion of the benefit was derived from a decrease in the occurrence of MI. REPLACE II REPLACE II was a large randomized double blind double dummy (active control) trial which enrolled 6,010 patients and evaluated bivalirudin plus provisional GP IIb/IIIa inhibitor vs heparin plus GP IIb/IIIa inhibitor. 15 The study purpose was to prove the non-inferiority of bivalirudin with provisional GP IIb/IIIa inhibitor compared to heparin plus GP IIb/IIIa inhibitors. Abciximab or eptifibatide were approved for GP IIb/IIIa inhibitor use in this trial. The primary outcome measure included death, MI, UTVR, and in-hospital major bleeding. There was no statistically significant difference in the occurrence of the primary endpoint (9.2% bivalirudin versus 10% heparin plus GP IIb/IIIa, p=0.32). The percentage of patients receiving provisional GP IIb/IIIa inhibitor therapy was 7.2%. The investigators measured CK-MB levels every 8 hrs for 24 hrs after PCI. Subgroup analysis of the triple composite endpoint of death, MI, or UTVR, showed that heparin plus GP IIb/IIIa inhibitor therapy was superior in all groups except women and patients >75 yrs of age. There was not a statistically significant difference in the rate of death or MI between the two groups. Although there was a trend toward a higher rate of MI in the bivalirudin group, it is unclear whether provisional GP IIb/IIIa or bivalirudin alone is primarily responsible for this trend. There was a significant decrease in in-hospital major bleeding rates for bivalirudin (2.4% for bivalirudin vs 4.1% in the heparin/gp IIb/IIIa group). This
5 represents the highest rate of major bleeding among the studies evaluated thus far. ESPRIT reported a major bleeding rate of 1%, EPISTENT 1.5%, and TARGET 0.9% for tirofiban and 0.7% for abciximab. ISAR-REACT This most recent study was designed to show the superiority of abciximab plus clopidogrel vs clopidogrel alone. 16 It enrolled patients scheduled to undergo elective PCI in a native coronary artery, a relatively low risk patient population similar to ESPRIT. Although the authors concluded that there was no statistically significant difference in the occurrence of the primary composite endpoint of death (0.3% abciximab and 0.3% clopidogrel), MI (4% abciximab and 4% clopidogrel) or UTVR (1% abciximab and 1% clopidogrel), the primary endpoints were measured by patient interviews conducted at 30 days after randomization. This potentially introduced bias into the results based on patient perceptions of their symptoms and patient understanding of the questions posed, and may again have led to an under-reporting of events. As in TARGET and REPLACE II, CK-MB levels were drawn every 8 hrs for 24hrs following PCI. Furthermore the investigators excluded patients with insulin dependent diabetes mellitus, a large subset of patients with coronary artery disease that have been shown to benefit from GP IIb/IIIa inhibition. Another difference in this study is that patients enrolled in Europe who were in the placebo group received 140 units of heparin/kg with no activated clotting time (ACT) monitoring. This accounts for about 95% of all patients enrolled in the study. Placebo patients in the United States received 100 units heparin/kg and maintained an ACT of sec. It is unknown whether these differences affected the study results. SUMMARY There were problems in each of the studies related to reporting of outcome measurements. There were also differences in the types of stents used. Despite these issues, eptifibatide and abciximab have been shown to significantly reduce death, MI, and UTVR in patients undergoing PCI with stent placement. While earlier data for patients undergoing PCI with balloon angioplasty or atherectomy indicated abciximab to be a superior choice for GP IIb/IIIa inhibition, there is no evidence to date which would indicate abciximab to be superior to eptifibatide in the setting of stent placement, particularly with doses adequate to induce sufficient platelet inhibition, and the newer, less thrombogenic stents now being utilized. High-risk patients seem to derive the greatest benefit from either abciximab or eptifibatide. Subgroup analysis in the EPISTENT trial might indicate that abciximab is a more appropriate choice for patients with diabetes; however, the study was not powered to detect such a difference. Therefore it would be premature to assume superiority of abciximab over eptifibatide in this population. The TARGET trial failed to show equivalence
6 between abciximab and tirofiban; the trial showed the superiority of abciximab in the primary composite endpoint. However, there was not a statistically significant difference in the rates of death or UTVR. It remains to be seen whether the decreased rate of MI translates into a long-term survival benefit. Tirofiban, with its differing pharmacokinetic and dynamic properties, needs further evaluation to determine the optimal dose to achieve >80% inhibition of the GP IIb/IIIa receptor, at which time it may prove to be an appropriate choice. REPLACE II concluded that bivalirudin was not inferior to heparin plus GP IIb/IIIa inhibitor in the prevention of death, MI, or UTVR, and that it produced a decrease in major in-hospital bleeding rates. One of the reasons for the hope that bivalirudin would be effective is its decreased cost vs. that of GP IIb/IIIa inhibitors since it was infused only for the duration of the procedure. As discussed previously, the use of provisional GP IIb/IIIa inhibition at a rate of 7.2% in the bivalirudin group may have decreased the occurrence of the primary outcome in that group and therefore we cannot conclude that the two are equivalent without further study. Once more, while the study did show a significant decrease in the rate of major bleeding events in the bivalirudin group, the question remains as to why the rate for heparin/gp IIb/IIIa inhibitor was so different from the previously conducted trials, especially since both trials utilized the Thrombolysis in Myocardial Infarction criteria for major and minor bleeding. 17 While the ISAR-REACT investigators concluded that clopidogrel 600mg may be appropriate without the addition of GP IIb/IIIa inhibitor in certain low risk patients, the exclusion of insulin dependent diabetes mellitus patients and the method by which the investigators measured endpoints as well as the method and dose of heparin administration during the procedure would indicate that further study is needed. RECOMMENDATIONS Based on evaluation of these five trials, patients undergoing PCI with stent placement should receive aspirin, thienopyridine as well as GP IIb/IIIa inhibitor. Abciximab and eptifibatide are both appropriate therapy for high-risk patients, including those with multiple target lesions, diabetes and renal failure. There is much supposition about the possible additional benefits of abciximab, which binds to at least two other integrins expressed in endothelial cells and monocytes, which may further influence the adhesion of platelets and endothelial cells as well as platelets and white cells. 5 These potential differences are yet to be elucidated. Eptifibatide also showed significantly lower rates of bleeding and thrombocytopenia as compared to abciximab. Eptifibatide is therefore a cost effective alternative ($400-$500/patient) compared to abciximab ($ /patient). Long- term studies are still lacking for the use of bivalirudin in the PCI setting, therefore, it cannot yet be recommended as monotherapy for patients undergoing stent placement but is an alternative for patients who are unable to
7 receive heparin or are at a higher risk for bleeding complications. Bivalirudin may actually be added to GP IIb/IIIa inhibitor since 7.2% of patients in REPLACE II received both agents without a higher incidence of bleeding complications. Although a high loading dose of clopidogrel yields additional platelet inhibition, it cannot be recommended as monotherapy in patients undergoing PCI with stent placement until long-term studies are conducted. These recommendations are consistent with the recently published consensus guidelines. 18 The guidelines are more specific with regard to medication choice in different types of MI. The five trials reviewed in this evaluation were not powered to detect differences to this detail. References: 1. Belli G, Ellis SG, Topol EJ. Stenting for ischemic heart disease. Prog Cardiovasc Dis 1997;40: Ellis SG, Roubin GS, King SB, III, et.al. Angiographic and clinical predictors of acute closure after native vessel coronary angioplasty. Circulation 1988;284: Ellis SG, Roubin GS, King SB, III, et.al. In-hospital cardiac mortality after acute closure after coronary angioplasty: analysis of risk factors from 8207 procedures. J Am Coll Cardiol 1988;12: Smith SC Jr, Dove JT, Jacobs AK, et.al. ACC/AHA guidelines for percutaneous coronary intervention (revision of the 1993 PTCA guidelines)- executive summary: A report of the American College of Cardiology/American Heart Association task force on practice guidelines (Committee to revise the 1993 guidelines for percutaneous transluminal coronary angioplasty) endorsed by the Society for Cardiac Angiography and Interventions. Circulation 2001;103: Kerieakes DJ, Runyon JP, Broderick TM, et.al. IIb s Are not IIb s. Am J Cardiol 2000;85:24c-31c 6. Topol EJ, Ferguson JJ, Weisman HF, et.al. Long-term protection from myocardial ischemic events in a randomized trial of brief integrin beta 3 blockade with percutaneous coronary intervention. EPIC Investigator Group. Evaluation of Platelet IIb/IIIa inhibition for prevention of ischemic complication. JAMA 1997;278: The EPILOG Investigators. Platelet glycoprotein IIb/IIIa receptor blockage and low-dose heparin during percutaneous coronary revascularization. N Engl J Med 1997;336: The IMPACT II Investigators. Randomized placebo-controlled trial of effect of eptifibatide on complications of percutaneous coronary intervention. IMPACT II. Lancet 1997;349: RESTORE investigators. Effects of platelet glycoprotein IIb/IIIa blockade with Tirofiban on adverse cardiac events in patients with unstable angina or acute myocardial infarction undergoing coronary angioplasty. Circulation 1997;96:
8 10. Lembo NJ, Black AJ, Roubin GS, et.al. Effect of pre-treatment with aspirin vs aspiring plus dipyridamole on frequency and type of acute complications of percutaneous transluminal coronary angioplasty. Am J Cardiol 1990;65: Urban P, Macaya C, Rupprecht HJ, et.al. Randomized evaluation of anticoagulation vs antplatelet therapy after coronary stent implantation in high-risk patients: The multicenter aspirin and ticlopidine after intracoronary stenting (MATTIS) trial. Circulation 1998;98: Tcheng JE, O Shea JC, Cohen EA, et.al. Enhanced Suppression of the Platelet IIb/IIIa Receptor with Integrilin Therapy (ESPRIT). Lancet 2000;356: Topol EJ, Lincoff AM, Califf RM, et.al. Randomized Placebo Controlled and Balloon Angioplasty Controlled Trial to Assess Safety of Coronary Stenting with use of Platelet Glycoprotein IIb/IIIa Blockade. Lancet 1998;352: Topol EJ, Moliterno DJ, Herrmann HD, et.al. Comparison of Two Platelet Glycoprotein IIb/IIIa Inhibitors, Tirofiban and Abciximab, for the Prevention of Ischemic Events with Percutaneous Coronary Revascularization. N Engl J Med 2001;344: Lincoff A, Bittle J, Harrington R, et.al. Bivalirudin and Provisional Glycoprotein IIb/IIIa Blockade Compared with Heparin and Planned Glycoprotein IIb/IIIa Blockade During Percutaneous Coronary Intervention. JAMA 2003;289: Kastrati A, Mehilli J, Schuhlen H, et.al. A clinical trial of abciximab in elective percutaneous coronary intervention after pretreatment with clopidogrel. N Engl J Med 2004;350: Rao AK, Pratt C, Berke A, et.al. Thrombolysis in myocardial infarction (TIMI) trial, Phase I: Hemorrhagic manifestations and changes in plasma fibrinogen and the fibrinolytic system in patients treated with recombinant tissue plasminogen activator and streptokinase. J Am Coll Cardiol. 1988;11: Popma JJ, Berger P, Ohman EM, et.al. Antithrombotic Therapy During Percutaneous Coronary Intervention CHEST 2004;126:576S-599S
9 Table 1 Summary of evaluated clinical trials Study Trial Design # of subjects EPISTENT R DB Pl Ct Multicenter trial in the U.S. and Canada. Duration: 30 days Inclusion criteria 2399 Elective or urgent revascularization (UR) with at least 76% stenosis in target lesion that is not an unprotected left main. Exclusion Criteria Bleeding diathesis, intracranial neoplasm, hx of stroke in the past 2 yrs, uncontrolled HTN (SBP >180 or DBP >100), recent surgery or PCI within the past 3 mos, concurrent warfarin therapy or an INR >1.5 ESPRIT TARGET R DB Pl Ct crossover permitted Multicenter trial in the U.S. and Canada Duration: 30days R DB active control (double dummy) Multicenter trial in 18 countries Duration: 30 days REPLACE II R DB active control (double dummy) ISAR- REACT Multicenter trial in 9 countries Duration: 30days R DB Pl Ct Multicenter trial in Europe and the U.S. (5% of patients enrolled) Duration: 30 days 2064 Patients scheduled to undergo PCI with stent implantation in a native coronary artery and who would not routinely be treated with GP IIb/IIIa inhibitors 4809 Patients to undergo coronary stenting procedure (elective or urgent) of a newly stenotic or restenotic atherosclerotic lesion in a native vessel or a bypass graft. All lesions that were judged to have stenosis of more than 70% on angiography had to be amenable to stenting >21 years of age and scheduled to undergo PCI with an approved device. MI in the last 24hrs, continuing CP, PCI in the last 3 mos, previous stent in target lesion, anticipated staged PCI in 30 days after randomization, GP IIb/IIIa inhibitor or thienopyridine 30 days before randomization, stroke or TIA in the last 30 days, any hx of hemorrhagic stroke, hx of bleeding diathesis or abnormal bleeding within the last 30 days, major surgery in the last 6 wks, uncontrolled HTN (SBP >200 or DBP >100), documented thrombocytopenia (<100,000) or Scr >350mc mol/l Patients with cardiogenic shock, acute MI with ECG evidence of STsegment elevation, SCr >2.5, ongoing bleeding or bleeding diathesis including a plt count of <120,000. PCI performed as reperfusion therapy for acute MI, poorly controlled HTN (SBP >180 or DBP >100), unprotected LM stenosis of >50%, pregnancy, PCI in the last 30 days or planned staged PCI within the next 30 days, active internal bleeding or bleeding diathesis, surgery, trauma or GI or genitourinary tract bleeding in the last 6wks, prior intracranial bleeding or structural abnormality. PLT <100,000, SCr >4 or dialysis, bivalirudin within the last 24hrs, warfarin therapy, unfractionated heparin in the last 6hrs, LMWH in the last 8hrs, abciximab in the last 7days, eptifibatide or tirofiban in the last 12 hrs Patients to undergo elective PCI in a native coronary artery MI in the last 14 days, US with ST changes in 2 leads at rest, Troponin T of >0.03ng/ml, target lesion in a graft, chronic occlusion present for >3 mos, target lesion with angiographically visible thrombus, EF <30%, hemodynamic instability, IDDM, pericarditis, cancer, stroke in prior 3mos, active bleeding or bleeding diathesis, trauma or major surgery in the last month, suspected aortic dissection, oral anticoagulation, GP IIb/IIIa inhibitor within the last 14 days, severe uncontrolled HTN (SBP>180), Hgb <10, HCT <34%, PLT <100,000 or >600,000, known allergic reaction to the study medication, pregnancy
10 Table 1 (cont) Study Treatment Regimens Evaluated Outcome variables and methods of measurement EPISTENT ESPRIT Stent + placebo + heparin (hep) 100 units/kg and maintain ACT 300sec Stent + abciximab(0.25mcg/kg bolus and 0.125mcg/kg/min infusion x 12hrs) + hep 70 units/kg and maintain ACT 200 sec Balloon angioplasty + abciximab + heparin as described in the stent + abciximab group Placebo bolus + placebo infusion Eptifibatide 180mcg/kg bolus + 2mcg/kg/min infusion with an additional bolus of 180mcg/kg 10 min after the initial bolus. (pt with SCr >1.7 received 1mcg/kg/min) Death from any cause, MI, Urgent Target-vessel revascularization(utvr) within 30 days of intervention (no measurements are given for primary endpoints) Death, MI, UTVR, and thrombotic bailout with GP IIb/IIIa inhibitor within 48hrs after randomization MI defined as CK-MB 3x ULN in 2 samples for the first 24hrs after PCI. OR Clinical syndrome consistent with MI that included Q waves (at least 0.04 s) in 2 or more contiguous leads, or new left bundle branch block. Major Findings Primary outcome: Stent + placebo 10.8% Stent + abciximab 5.3% (p<0.001) Angioplasty + abciximab 6.9% (p=.007) Major bleeding complications Stent + placebo 2.2% Stent + abciximab 1.5% Angioplasty + abciximab 1.4% (p=0.38) 8% RR reduction of death or MI at 48hrs study was terminated early for efficacy TARGET Tirofiban 10mcg/kg bolus mcg/kg/mins x 18-24hr + hep 70 units/kg to maintain ACT 250 sec Abciximab 0.25mg/kg mcg/kg/min (max 10mcg/min) x 12hrs + hep as above Death, MI, UTVR within 30days of procedure MI defined as MB 3 x ULN in 2 blood samples or by the finding of abnormal Q waves in 2 or more contiguous leads (measured at baseline and q6h x 24hrs after procedure). Composite endpoints Tirofiban 7.6% Abciximab 6% (p=0.038) Death Tirofiban 0.5% Abciximab 0.4% (p=0.66) Nonfatal MI Tirofiban 6.9% Abciximab 5.4% (p=0.04) UTVR Tirofiban 0.8% Abciximab 0.7% (p=0.49) REPLACE II Bivalirudin 0.75mg/kg prior to start of intervention + infusion of 1.75mg/kg/hr for the duration of the procedure (provisional GP II/IIIa inhibitor) Heparin 65 units/kg (max of 7000 units) prior to PCI plus either abciximab 0.25mg/kg bolus and 0.125mcg/kg/min infusion (max of 10mcg/min) x 12hrs OR Death from any cause, MI, UTVR, or in-hospital major bleeding MI defined by new Q waves in 2 or more contiguous leads or elevation in CK or CK-MB to 3x ULN within 2 days of revasculariztion or to 2 x ULN thereafter. (levels obtained in all pts Q8h x 24hrs after procedure) Primary endpoint Bivalirudin 9.2% Heparin 10% (p=0.32) In hospital major bleeding rates Bivalirudin 2.4% Heparin 4.1% (p<0.001) ISAR-REACT Heparin as described above plus eptifibatide 180mcg/kg x 2 boluses 10min apart and infusion at 2mcg/kg/min Clopidogrel 600mg + abciximab 0.25mg/kg and 0.125mcg/kg/min x 12hrs Clopidogrel 600mg + placebo bolus and infusion Major bleeding was defined as intracranial, intraocular, or retroperitoneal hemorrhage, clinically overt blood loss resulting in a decrease in Hgb of >3g/dl, any decrease in Hgb of >4g/dl or transfusion of 2 or more units of PRBC s or whole blood. Death from any cause, MI, UTVR owing to ischemia within 30 days after randomization. Diagnosis of MI was based on the development of pathologic Q waves in 2 or more contiguous leads or an elevation of CK or its MB isoenzyme to at least 3 x ULN in at least 2 blood samples. Primary endpoint Abciximab 4% Clopidogrel 4% Large MI was based on the presence of pathologic Q waves in 2 or more contiguous leads or an elevation of CK or its MB isoenzyme to at least 5x ULN in at least 2 samples.
Guideline for STEMI. Reperfusion at a PCI-Capable Hospital
 MANSOURA. 2015 Guideline for STEMI Reperfusion at a PCI-Capable Hospital Mahmoud Yossof MANSOURA 2015 Reperfusion Therapy for Patients with STEMI *Patients with cardiogenic shock or severe heart failure
MANSOURA. 2015 Guideline for STEMI Reperfusion at a PCI-Capable Hospital Mahmoud Yossof MANSOURA 2015 Reperfusion Therapy for Patients with STEMI *Patients with cardiogenic shock or severe heart failure
Learning Objectives. Epidemiology of Acute Coronary Syndrome
 Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Percutaneous coronary intervention (PCI), in
 ECONOMIC IMPLICATIONS OF THE USE OF DIRECT THROMBIN INHIBITORS IN PERCUTANEOUS CORONARY INTERVENTION David J. Cohen, MD, MSc * ABSTRACT More than 1.2 million percutaneous coronary intervention (PCI) procedures
ECONOMIC IMPLICATIONS OF THE USE OF DIRECT THROMBIN INHIBITORS IN PERCUTANEOUS CORONARY INTERVENTION David J. Cohen, MD, MSc * ABSTRACT More than 1.2 million percutaneous coronary intervention (PCI) procedures
Abciximab plus Heparin versus Bivalirudin in Patients with NSTEMI Undergoing PCI. ISAR-REACT 4 Trial
 Abciximab plus Heparin versus Bivalirudin in Patients with NSTEMI Undergoing PCI ISAR-REACT 4 Trial Adnan Kastrati, MD Deutsches Herzzentrum, Technische Universität, Munich, Germany On behalf of F.-J.
Abciximab plus Heparin versus Bivalirudin in Patients with NSTEMI Undergoing PCI ISAR-REACT 4 Trial Adnan Kastrati, MD Deutsches Herzzentrum, Technische Universität, Munich, Germany On behalf of F.-J.
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015
 Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Interventional Cardiology
 Clinical Investigations Interventional Cardiology Benefit of bolus-only platelet glycoprotein IIb/IIIa inhibition during percutaneous coronary intervention: Insights from the very early outcomes in the
Clinical Investigations Interventional Cardiology Benefit of bolus-only platelet glycoprotein IIb/IIIa inhibition during percutaneous coronary intervention: Insights from the very early outcomes in the
Update on Antithrombotic Therapy in Acute Coronary Syndrome
 Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
Journal of the American College of Cardiology Vol. 44, No. 9, by the American College of Cardiology Foundation ISSN /04/$30.
 Journal of the American College of Cardiology Vol. 44, No. 9, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.05.085
Journal of the American College of Cardiology Vol. 44, No. 9, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.05.085
The use of percutaneous coronary intervention (PCI) as
 Antithrombotic Therapy During Percutaneous Coronary Intervention The Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy Jeffrey J. Popma, MD; Peter Berger, MD; E. Magnus Ohman, MD, FCCP;
Antithrombotic Therapy During Percutaneous Coronary Intervention The Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy Jeffrey J. Popma, MD; Peter Berger, MD; E. Magnus Ohman, MD, FCCP;
Timing of Surgery After Percutaneous Coronary Intervention
 Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
QUT Digital Repository:
 QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
Role of Clopidogrel in Acute Coronary Syndromes. Hossam Kandil,, MD. Professor of Cardiology Cairo University
 Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
 Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy Franz-Josef Neumann Herz-Zentrum Bad Krozingen Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy Franz-Josef Neumann Herz-Zentrum Bad Krozingen Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention
 Original Contribution Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Pompeiu Vladutiu, MD,
Original Contribution Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Pompeiu Vladutiu, MD,
An update on the management of UA / NSTEMI. Michael H. Crawford, MD
 An update on the management of UA / NSTEMI Michael H. Crawford, MD New ACC/AHA Guidelines 2007 What s s new in the last 5 years CT imaging advances Ascendancy of troponin and BNP Clarification of ACEI/ARB
An update on the management of UA / NSTEMI Michael H. Crawford, MD New ACC/AHA Guidelines 2007 What s s new in the last 5 years CT imaging advances Ascendancy of troponin and BNP Clarification of ACEI/ARB
Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction. Is it beneficial to patients?
 Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction Is it beneficial to patients? Seung-Jea Tahk, MD. PhD. Suwon, Korea Facilitated PCI.. background Degree of coronary flow at
Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction Is it beneficial to patients? Seung-Jea Tahk, MD. PhD. Suwon, Korea Facilitated PCI.. background Degree of coronary flow at
THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH?
 THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH? Paul I. Oh, 1 Eric A. Cohen, 2 Nicole Mittmann, 3, 4 Soo Jin Seung 4 1 Division of Clinical Pharmacology, Sunnybrook
THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH? Paul I. Oh, 1 Eric A. Cohen, 2 Nicole Mittmann, 3, 4 Soo Jin Seung 4 1 Division of Clinical Pharmacology, Sunnybrook
High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention
 Original Contribution High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Nagarathna Manjappa MD, Erdal
Original Contribution High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Nagarathna Manjappa MD, Erdal
NOVEL ANTI-THROMBOTIC THERAPIES FOR ACUTE CORONARY SYNDROME: DIRECT THROMBIN INHIBITORS
 Judd E. Hollander, MD Professor, Clinical Research Director, Department of Emergency Medicine University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania OBJECTIVES: 1. Discuss the concept
Judd E. Hollander, MD Professor, Clinical Research Director, Department of Emergency Medicine University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania OBJECTIVES: 1. Discuss the concept
FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS
 New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
Acute Coronary Syndromes
 Overview Acute Coronary Syndromes Rabeea Aboufakher, MD, FACC, FSCAI Section Chief of Cardiology Altru Health System Grand Forks, ND Epidemiology Pathophysiology Clinical features and diagnosis STEMI management
Overview Acute Coronary Syndromes Rabeea Aboufakher, MD, FACC, FSCAI Section Chief of Cardiology Altru Health System Grand Forks, ND Epidemiology Pathophysiology Clinical features and diagnosis STEMI management
Otamixaban for non-st-segment elevation acute coronary syndrome
 Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
Journal of the American College of Cardiology Vol. 38, No. 3, by the American College of Cardiology ISSN /01/$20.
 Journal of the American College of Cardiology Vol. 38, No. 3, 2001 2001 by the American College of Cardiology ISSN 0735-1097/01/$20.00 Published by Elsevier Science Inc. PII S0735-1097(01)01423-1 Effect
Journal of the American College of Cardiology Vol. 38, No. 3, 2001 2001 by the American College of Cardiology ISSN 0735-1097/01/$20.00 Published by Elsevier Science Inc. PII S0735-1097(01)01423-1 Effect
Novel Anticoagulation Therapy in Acute Coronary Syndrome
 Novel Anticoagulation Therapy in Acute Coronary Syndrome Soon Jun Hong Korea University Anam Hospital 1 Thrombus Formation Cascade Coagulation Cascade Platelet Cascade TXA2 Aspirin R Inhibitor Fondaparinux
Novel Anticoagulation Therapy in Acute Coronary Syndrome Soon Jun Hong Korea University Anam Hospital 1 Thrombus Formation Cascade Coagulation Cascade Platelet Cascade TXA2 Aspirin R Inhibitor Fondaparinux
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company
 bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
Anti-platelet Therapies in Cardiovascular Disease: From Stable CAD to ACS and Afib!
 Anti-platelet Therapies in Cardiovascular Disease: From Stable CAD to ACS and Afib! Roxana Mehran, MD Columbia University Medical Center Cardiovascular Research Foundation Disclosures Research support
Anti-platelet Therapies in Cardiovascular Disease: From Stable CAD to ACS and Afib! Roxana Mehran, MD Columbia University Medical Center Cardiovascular Research Foundation Disclosures Research support
Oral Antiplatelet Therapy in PCI/ACS. Dominick J. Angiolillo, MD, PhD, FACC, FESC Director of Cardiovascular Research Assistant Professor of Medicine
 Oral Antiplatelet Therapy in PCI/ACS Dominick J. Angiolillo, MD, PhD, FACC, FESC Director of Cardiovascular Research Assistant Professor of Medicine Basic Concepts Thrombus Formation Two key elements:
Oral Antiplatelet Therapy in PCI/ACS Dominick J. Angiolillo, MD, PhD, FACC, FESC Director of Cardiovascular Research Assistant Professor of Medicine Basic Concepts Thrombus Formation Two key elements:
Prevention of Coronary Stent Thrombosis and Restenosis
 Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
Chapter 15 Glycoprotein IIb/IIIa Antagonists
 Chapter 15 Glycoprotein IIb/IIIa Antagonists Introduction - GP IIb/IIIa receptors - Pharmacologic approaches - Preparations and dosages Mechanism of Action Clinical Efficacy - UA / NSTEMI - STEMI - PCI
Chapter 15 Glycoprotein IIb/IIIa Antagonists Introduction - GP IIb/IIIa receptors - Pharmacologic approaches - Preparations and dosages Mechanism of Action Clinical Efficacy - UA / NSTEMI - STEMI - PCI
Platelet-fibrin clot. 50Kd STEMI. Abciximab. Video of a IIb/IIIa inhibitor in action. Unstable Angina and non-stsegment
 Objectives IIb/IIIa, Vitamin K, and Direct Thrombin Inhibition in Cardiology Michael Gulseth, Pharm. D., BCPS Assistant Professor, Duluth Pharmacy 6122 February 14, 2005 Describe the pharmacology, kinetics,
Objectives IIb/IIIa, Vitamin K, and Direct Thrombin Inhibition in Cardiology Michael Gulseth, Pharm. D., BCPS Assistant Professor, Duluth Pharmacy 6122 February 14, 2005 Describe the pharmacology, kinetics,
Myocardial Infarction In Dr.Yahya Kiwan
 Myocardial Infarction In 2007 Dr.Yahya Kiwan New Definition Of Acute Myocardial Infarction The term of myocardial infarction should be used when there is evidence of myocardial necrosis in a clinical setting
Myocardial Infarction In 2007 Dr.Yahya Kiwan New Definition Of Acute Myocardial Infarction The term of myocardial infarction should be used when there is evidence of myocardial necrosis in a clinical setting
Tailoring adjunctive antithrombotic therapy to reperfusion strategy in STEMI
 Tailoring adjunctive antithrombotic therapy to reperfusion strategy in STEMI Adel El-Etriby; MD Professor of Cardiology Ain Shams University President of the Egyptian Working Group of Interventional Cardiology
Tailoring adjunctive antithrombotic therapy to reperfusion strategy in STEMI Adel El-Etriby; MD Professor of Cardiology Ain Shams University President of the Egyptian Working Group of Interventional Cardiology
Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI
 Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI Charles A. Simonton MD, FACC, FSCAI Sanger Clinic Medical Director Clinical Innovation and Research Carolinas Heart
Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI Charles A. Simonton MD, FACC, FSCAI Sanger Clinic Medical Director Clinical Innovation and Research Carolinas Heart
Impact of Gender on In-Hospital Percutaneous Coronary Interventional Outcomes in African-Americans
 Original Contribution Impact of Gender on In-Hospital Percutaneous Coronary Interventional Outcomes in African-Americans Shyam Poludasu, MD, Erdal Cavusoglu, MD, Luther T. Clark, MD, Jonathan D. Marmur,
Original Contribution Impact of Gender on In-Hospital Percutaneous Coronary Interventional Outcomes in African-Americans Shyam Poludasu, MD, Erdal Cavusoglu, MD, Luther T. Clark, MD, Jonathan D. Marmur,
C.R.E.D.O. Multicenter Multinational (USA, Canada) Prospective Randomized Double Blind Placebo Controlled Trial
 Clopidogrel for the Reduction of Events During Observation Multicenter Multinational (USA, Canada) Prospective Randomized Double Blind Placebo Controlled Trial From Steinhubl et al, JAMA 2002;228:2411-20
Clopidogrel for the Reduction of Events During Observation Multicenter Multinational (USA, Canada) Prospective Randomized Double Blind Placebo Controlled Trial From Steinhubl et al, JAMA 2002;228:2411-20
Report on New Patented Drug - Angiomax
 Report on New Patented Drug - Angiomax March 2006 Under its transparency initiative, the PMPRB publishes the results of the reviews of new patented drugs by Board Staff, for purposes of applying the PMPRB
Report on New Patented Drug - Angiomax March 2006 Under its transparency initiative, the PMPRB publishes the results of the reviews of new patented drugs by Board Staff, for purposes of applying the PMPRB
Index. Note: Page numbers of article titles are in boldface type.
 Cardiol Clin 24 (2006) 299 304 Index Note: Page numbers of article titles are in boldface type. A Abciximab in PCI, 180 182 ACE trial, 182 Actinomycin D-eluting stent, 224 ACUITY trial, 190 Acute myocardial
Cardiol Clin 24 (2006) 299 304 Index Note: Page numbers of article titles are in boldface type. A Abciximab in PCI, 180 182 ACE trial, 182 Actinomycin D-eluting stent, 224 ACUITY trial, 190 Acute myocardial
Anti-thrombotic management in interventional cardiology
 Chapter 8 Anti-thrombotic management in interventional cardiology James Tcheng, Steve Kindsvater Introduction Since the dawn of the interventional age, the need for anti-thrombotic therapy during percutaneous
Chapter 8 Anti-thrombotic management in interventional cardiology James Tcheng, Steve Kindsvater Introduction Since the dawn of the interventional age, the need for anti-thrombotic therapy during percutaneous
Adjunctive Antithrombotic for PCI. SCAI Fellows Course December 9, 2013
 Adjunctive Antithrombotic for PCI SCAI Fellows Course December 9, 2013 Theodore A Bass, MD FSCAI President SCAI Professor of Medicine, University of Florida Medical Director UF Shands CV Center,Jacksonville
Adjunctive Antithrombotic for PCI SCAI Fellows Course December 9, 2013 Theodore A Bass, MD FSCAI President SCAI Professor of Medicine, University of Florida Medical Director UF Shands CV Center,Jacksonville
Continuing Medical Education Post-Test
 Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction
 Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction The Bavarian Reperfusion Alternatives Evaluation (BRAVE) 4
Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction The Bavarian Reperfusion Alternatives Evaluation (BRAVE) 4
Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention
 Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention Aggregate Results From 6 Randomized, Controlled Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; A. Michael Lincoff,
Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention Aggregate Results From 6 Randomized, Controlled Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; A. Michael Lincoff,
Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network
 Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Adults With Diagnosed Diabetes
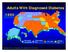 Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Impact of Clinical Syndrome Acuity on the Differential Response to 2 Glycoprotein IIb/IIIa Inhibitors in Patients Undergoing Coronary Stenting
 Impact of Clinical Syndrome Acuity on the Differential Response to 2 Glycoprotein IIb/IIIa Inhibitors in Patients Undergoing Coronary Stenting The TARGET Trial* Gregg W. Stone, MD; David J. Moliterno,
Impact of Clinical Syndrome Acuity on the Differential Response to 2 Glycoprotein IIb/IIIa Inhibitors in Patients Undergoing Coronary Stenting The TARGET Trial* Gregg W. Stone, MD; David J. Moliterno,
Acute Coronary Syndrome (ACS) Initial Evaluation and Management
 Acute Coronary Syndrome (ACS) Initial Evaluation and Management Symptoms of Possible ACS Chest discomfort with or without radiation to the arm(s), jaw, or epigastrium Short of breath Weakness Diaphoresis
Acute Coronary Syndrome (ACS) Initial Evaluation and Management Symptoms of Possible ACS Chest discomfort with or without radiation to the arm(s), jaw, or epigastrium Short of breath Weakness Diaphoresis
ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions
 Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
International Journal of Collaborative Research on Internal Medicine & Public Health (IJCRIMPH)
 Same-day discharge after percutaneous coronary intervention in light of the society for cardiovascular angiography and intervention's proposed guidelines: A single-center experience Yazan Khouri, Sachin
Same-day discharge after percutaneous coronary intervention in light of the society for cardiovascular angiography and intervention's proposed guidelines: A single-center experience Yazan Khouri, Sachin
Contemporary Percutaneous Coronary Intervention Versus Balloon Angioplasty for Multivessel Coronary Artery Disease
 Contemporary Percutaneous Coronary Intervention Versus Balloon Angioplasty for Multivessel Coronary Artery Disease A Comparison of the National Heart, Lung and Blood Institute Dynamic Registry and the
Contemporary Percutaneous Coronary Intervention Versus Balloon Angioplasty for Multivessel Coronary Artery Disease A Comparison of the National Heart, Lung and Blood Institute Dynamic Registry and the
Triple Therapy: A review of the evidence in acute coronary syndrome. Stephanie Kling, PharmD, BCPS Sanford Health
 Triple Therapy: A review of the evidence in acute coronary syndrome Stephanie Kling, PharmD, BCPS Sanford Health Objectives 1. Describe how the presented topic impacts patient outcomes. 2. Review evidence
Triple Therapy: A review of the evidence in acute coronary syndrome Stephanie Kling, PharmD, BCPS Sanford Health Objectives 1. Describe how the presented topic impacts patient outcomes. 2. Review evidence
Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC
 Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC Professor, Medicine/Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical
Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC Professor, Medicine/Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical
Adjunctive Antithrombotic for PCI. SCAI Fellows Course December 8, 2014
 Adjunctive Antithrombotic for PCI SCAI Fellows Course December 8, 2014 Theodore A Bass, MD FSCAI Immediate Past-President SCAI Professor of Medicine, University of Florida Medical Director UF Health CV
Adjunctive Antithrombotic for PCI SCAI Fellows Course December 8, 2014 Theodore A Bass, MD FSCAI Immediate Past-President SCAI Professor of Medicine, University of Florida Medical Director UF Health CV
Health technology Abciximab use in high-risk patients undergoing percutaneous transluminal coronary angioplasty.
 Costs and effects in therapy for acute coronary syndromes: the case of abciximab in highrisk patients undergoing percutaneous transluminal coronary angioplasty in the EPIC study van Hout B A, Bowman L,
Costs and effects in therapy for acute coronary syndromes: the case of abciximab in highrisk patients undergoing percutaneous transluminal coronary angioplasty in the EPIC study van Hout B A, Bowman L,
Pathophysiology of ACS
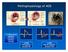 Pathophysiology of ACS ~ 2.0 MM patients admitted to CCU or telemetry annually 0.6 MM ST-segment elevation MI 1.4 MM Non-ST-segment elevation ACS NSTEMI vs STEMI VANQWISH Boden et al N Engl J Med 1998;338:1785-1792
Pathophysiology of ACS ~ 2.0 MM patients admitted to CCU or telemetry annually 0.6 MM ST-segment elevation MI 1.4 MM Non-ST-segment elevation ACS NSTEMI vs STEMI VANQWISH Boden et al N Engl J Med 1998;338:1785-1792
Clinical Lessons from BMC2-PCI
 Clinical Lessons from BMC2-PCI The Blue Cross Blue Shield of Michigan Cardiovascular Consortium Hitinder Gurm, M.D. University of Michigan Overview 32 papers since inception 10 papers published this year
Clinical Lessons from BMC2-PCI The Blue Cross Blue Shield of Michigan Cardiovascular Consortium Hitinder Gurm, M.D. University of Michigan Overview 32 papers since inception 10 papers published this year
ST-segment Elevation Myocardial Infarction (STEMI): Optimal Antiplatelet and Anti-thrombotic Therapy in the Emergency Department
 ST-segment Elevation Myocardial Infarction (STEMI): Optimal Antiplatelet and Anti-thrombotic Therapy in the Emergency Department decision-making. They have become the cornerstone of many ED protocols for
ST-segment Elevation Myocardial Infarction (STEMI): Optimal Antiplatelet and Anti-thrombotic Therapy in the Emergency Department decision-making. They have become the cornerstone of many ED protocols for
Acute Coronary Syndrome. Cindy Baker, MD FACC Director Peripheral Vascular Interventions Division of Cardiovascular Medicine
 Acute Coronary Syndrome Cindy Baker, MD FACC Director Peripheral Vascular Interventions Division of Cardiovascular Medicine Topics Timing is everything So many drugs to choose from What s a MINOCA? 2 Acute
Acute Coronary Syndrome Cindy Baker, MD FACC Director Peripheral Vascular Interventions Division of Cardiovascular Medicine Topics Timing is everything So many drugs to choose from What s a MINOCA? 2 Acute
ST Elevated Myocardial Infarction- Latest AHA recommendations
 ST Elevated Myocardial Infarction- Latest AHA recommendations Sherry Turner, DO, MPH, FACOEP Medical Director Emergency Services Wesley Medical Center The Problem 250,000 Americans each year 30% fail to
ST Elevated Myocardial Infarction- Latest AHA recommendations Sherry Turner, DO, MPH, FACOEP Medical Director Emergency Services Wesley Medical Center The Problem 250,000 Americans each year 30% fail to
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin, 250mg powder for concentrate for solution for injection or infusion (Angiox ) No. (516/08) The Medicines Company UK Ltd 07 November 2008 The Scottish Medicines
Scottish Medicines Consortium bivalirudin, 250mg powder for concentrate for solution for injection or infusion (Angiox ) No. (516/08) The Medicines Company UK Ltd 07 November 2008 The Scottish Medicines
Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland
 Advances in Antiplatelet Therapy in PCI and ACS Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland Targets for Platelet
Advances in Antiplatelet Therapy in PCI and ACS Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland Targets for Platelet
Thrombolysis in Acute Myocardial Infarction
 CHAPTER 70 Thrombolysis in Acute Myocardial Infarction J. S. Hiremath Introduction Reperfusion of the occluded coronary artery at the earliest is the most important aim of management of STEMI. Once a flow
CHAPTER 70 Thrombolysis in Acute Myocardial Infarction J. S. Hiremath Introduction Reperfusion of the occluded coronary artery at the earliest is the most important aim of management of STEMI. Once a flow
Columbia University Medical Center Cardiovascular Research Foundation
 STEMI and NSTEMI Pharmacology Confusion: How to Choose and Use Antithrombins (Unfractionated and Low Molecular Heparins, Bivalirudin, Fondaparinux) and Antiplatelet Agents (Aspirin, Clopidogrel and Prasugrel)
STEMI and NSTEMI Pharmacology Confusion: How to Choose and Use Antithrombins (Unfractionated and Low Molecular Heparins, Bivalirudin, Fondaparinux) and Antiplatelet Agents (Aspirin, Clopidogrel and Prasugrel)
Acute coronary syndromes
 Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Horizon Scanning Centre November 2012
 Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
STEMI 2014 YAHYA KIWAN. Consultant Cardiologist Head Of Cardiology Belhoul Specialty Hospital
 STEMI 2014 YAHYA KIWAN Consultant Cardiologist Head Of Cardiology Belhoul Specialty Hospital Aspiration Thrombectomy Manual aspiration thrombectomy is reasonable for patients undergoing primary PCI. I
STEMI 2014 YAHYA KIWAN Consultant Cardiologist Head Of Cardiology Belhoul Specialty Hospital Aspiration Thrombectomy Manual aspiration thrombectomy is reasonable for patients undergoing primary PCI. I
Bivalirudin Clinical Trials Update Evidence and Future Perspectives
 Bivalirudin Clinical Trials Update Evidence and Future Perspectives Andreas Baumbach Consultant Cardiologist/ hon. Reader in Cardiology Bristol Heart Institute University Hospitals Bristol MY CONFLICTS
Bivalirudin Clinical Trials Update Evidence and Future Perspectives Andreas Baumbach Consultant Cardiologist/ hon. Reader in Cardiology Bristol Heart Institute University Hospitals Bristol MY CONFLICTS
Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes
 European Heart Journal (00) 3, 1441 1448 doi:10.1053/euhj.00.3160, available online at http://www.idealibrary.com on Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes Gradient of benefit
European Heart Journal (00) 3, 1441 1448 doi:10.1053/euhj.00.3160, available online at http://www.idealibrary.com on Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes Gradient of benefit
Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions
 Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions Anna Sonia Petronio, Marco De Carlo, Roberta Rossini, Giovanni Amoroso, Ugo Limbruno, Nicola Ciabatti, Caterina
Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions Anna Sonia Petronio, Marco De Carlo, Roberta Rossini, Giovanni Amoroso, Ugo Limbruno, Nicola Ciabatti, Caterina
NCDR CathPCI Registry v4.4 Diagnostic Catheterization and Percutaneous Coronary Intervention Registry
 A. DEMOGRAPHICS Last Name 2000 : First Name 2010 : Middle Name 2020 : SSN 2030 : - - - SSN N/A 2031 Patient ID 2040 : (auto) Other ID 2045 : Birth Date 2050 : *** @ Sex 2060 : *** @ O Male O Female Race:
A. DEMOGRAPHICS Last Name 2000 : First Name 2010 : Middle Name 2020 : SSN 2030 : - - - SSN N/A 2031 Patient ID 2040 : (auto) Other ID 2045 : Birth Date 2050 : *** @ Sex 2060 : *** @ O Male O Female Race:
The Evolving Role of Glycoprotein IIb/IIIa Inhibitors in the Setting of Percutaneous Coronary Intervention
 JACC: CARDIOVASCULAR INTERVENTIONS VOL. 3, NO. 12, 2010 2010 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2010.09.015 STATE-OF-THE-ART
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 3, NO. 12, 2010 2010 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2010.09.015 STATE-OF-THE-ART
Safety and Efficacy of Angioplasty with Intracoronary Stenting in Patients with Unstable Coronary Syndromes. Comparison with Stable Coronary Syndromes
 Original Article Safety and Efficacy of Angioplasty with Intracoronary Stenting in Patients with Unstable Coronary Syndromes. Comparison with Stable Coronary Syndromes Luís C. L. Correia, José Carlos Brito,
Original Article Safety and Efficacy of Angioplasty with Intracoronary Stenting in Patients with Unstable Coronary Syndromes. Comparison with Stable Coronary Syndromes Luís C. L. Correia, José Carlos Brito,
ARMYDA-RECAPTURE (Atorvastatin for Reduction of MYocardial Damage during Angioplasty) trial
 ARMYDA-RECAPTURE ( for Reduction of MYocardial Damage during Angioplasty) trial Prospective, multicenter, randomized, double blind trial investigating efficacy of atorvastatin reload in patients on chronic
ARMYDA-RECAPTURE ( for Reduction of MYocardial Damage during Angioplasty) trial Prospective, multicenter, randomized, double blind trial investigating efficacy of atorvastatin reload in patients on chronic
Anticoagulation therapy in acute coronary syndromes according to current guidelines
 Acute management of ACS Anticoagulation therapy in acute coronary syndromes according to current guidelines Marcin Grabowski, Marcin Leszczyk, Andrzej Cacko, Krzysztof J. Filipiak, Grzegorz Opolski 1 st
Acute management of ACS Anticoagulation therapy in acute coronary syndromes according to current guidelines Marcin Grabowski, Marcin Leszczyk, Andrzej Cacko, Krzysztof J. Filipiak, Grzegorz Opolski 1 st
Updated and Guideline Based Treatment of Patients with STEMI
 Updated and Guideline Based Treatment of Patients with STEMI Eli I. Lev, MD Director, Cardiac Catheterization Laboratory Hasharon Hospital, Rabin Medical Center Associate Professor of Cardiology Tel-Aviv
Updated and Guideline Based Treatment of Patients with STEMI Eli I. Lev, MD Director, Cardiac Catheterization Laboratory Hasharon Hospital, Rabin Medical Center Associate Professor of Cardiology Tel-Aviv
TCT mdbuyline.com Clinical Trial Results Summary
 TCT 2012 Clinical Trial Results Summary FAME2 Trial: FFR (fractional flow reserve) guided PCI in all target lesions Patients with significant ischemia, randomized 1:1 Control arm: not hemodynamically significant
TCT 2012 Clinical Trial Results Summary FAME2 Trial: FFR (fractional flow reserve) guided PCI in all target lesions Patients with significant ischemia, randomized 1:1 Control arm: not hemodynamically significant
Clinical Case. Management of ACS Based on ACC/AHA & ESC Guidelines. Clinical Case 4/22/12. UA/NSTEMI: Definition
 Clinical Case Management of ACS Based on ACC/AHA & ESC Guidelines Dr Badri Paudel Mr M 75M Poorly controlled diabetic Smoker Presented on Sat 7pm Intense burning in the retrosternal area Clinical Case
Clinical Case Management of ACS Based on ACC/AHA & ESC Guidelines Dr Badri Paudel Mr M 75M Poorly controlled diabetic Smoker Presented on Sat 7pm Intense burning in the retrosternal area Clinical Case
Management of Acute Myocardial Infarction
 Management of Acute Myocardial Infarction Prof. Hossam Kandil Professor of Cardiology Cairo University ST Elevation Acute Myocardial Infarction Aims Of Management Emergency care (Pre-hospital) Early care
Management of Acute Myocardial Infarction Prof. Hossam Kandil Professor of Cardiology Cairo University ST Elevation Acute Myocardial Infarction Aims Of Management Emergency care (Pre-hospital) Early care
Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach
 Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach Frans Van de Werf, MD, PhD University Hospitals, Leuven, Belgium Frans Van de Werf: Disclosures Research grants
Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach Frans Van de Werf, MD, PhD University Hospitals, Leuven, Belgium Frans Van de Werf: Disclosures Research grants
Keywords Non ST-segment elevation ACS Antithrombotic therapy Glycoprotein IIb/IIIa inhibitor Tirofiban. Introduction
 J Thromb Thrombolysis (2007) 24:241 246 DOI 10.1007/s19-007-0015-y Routine upstream versus selective down stream use of tirofiban in non-st elevation myocardial infarction patients scheduled for early
J Thromb Thrombolysis (2007) 24:241 246 DOI 10.1007/s19-007-0015-y Routine upstream versus selective down stream use of tirofiban in non-st elevation myocardial infarction patients scheduled for early
Manual of Interventional Cardiology
 782 PLATELET GLYCOPROTEIN IIb/IIIa RECEPTOR ANTAGONISTS (Tables 34.15, 34.16) Activation of the platelet glycoprotein (GP) IIb/IIIa receptor complex constitutes the final common pathway for platelet aggregation,
782 PLATELET GLYCOPROTEIN IIb/IIIa RECEPTOR ANTAGONISTS (Tables 34.15, 34.16) Activation of the platelet glycoprotein (GP) IIb/IIIa receptor complex constitutes the final common pathway for platelet aggregation,
Antiplatelet therapy and Coronary Interventions. Georgios I. Papaioannou, MD Hartford Hospital Grand Rounds 4/22/2003
 Antiplatelet therapy and Coronary Interventions Georgios I. Papaioannou, MD Hartford Hospital Grand Rounds 4/22/2003 Objectives Pharmacology of GP IIb/IIIa inhibitors and Monitoring of Platelet Inhibition
Antiplatelet therapy and Coronary Interventions Georgios I. Papaioannou, MD Hartford Hospital Grand Rounds 4/22/2003 Objectives Pharmacology of GP IIb/IIIa inhibitors and Monitoring of Platelet Inhibition
Objectives. Treatment of ACS. Early Invasive Strategy. UA/NSTEMI General Concepts. UA/NSTEMI Initial Therapy/Antithrombotic
 Objectives Treatment of ACS Michael P. Gulseth, Pharm. D., BCPS Pharmacotherapy II Spring 2006 Define early invasive strategy and what patients typically receive this approach Compare/contrast the medications
Objectives Treatment of ACS Michael P. Gulseth, Pharm. D., BCPS Pharmacotherapy II Spring 2006 Define early invasive strategy and what patients typically receive this approach Compare/contrast the medications
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS EPTIFIBATIDE (INTEGRILIN) PROTOCOL
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
How to approach non-infarct related artery disease in patients with STEMI in a limited resource setting
 How to approach non-infarct related artery disease in patients with STEMI in a limited resource setting Ahmed A A Suliman, MBBS, FACP, FESC Associate Professor, University of Khartoum Interventional Cardiologist,
How to approach non-infarct related artery disease in patients with STEMI in a limited resource setting Ahmed A A Suliman, MBBS, FACP, FESC Associate Professor, University of Khartoum Interventional Cardiologist,
Clinical Outcomes After Detection of Elevated Cardiac Enzymes in Patients Undergoing Percutaneous Intervention
 88 JACC Vol. 33, No. 1 Clinical Outcomes After Detection of Elevated Cardiac Enzymes in Patients Undergoing Percutaneous Intervention BARBARA E. TARDIFF, MD, ROBERT M. CALIFF, MD, FACC, JAMES E. TCHENG,
88 JACC Vol. 33, No. 1 Clinical Outcomes After Detection of Elevated Cardiac Enzymes in Patients Undergoing Percutaneous Intervention BARBARA E. TARDIFF, MD, ROBERT M. CALIFF, MD, FACC, JAMES E. TCHENG,
Clopidogrel Date: 15 July 2008
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
Clopidogrel and ASA after CABG for NSTEMI
 Clopidogrel and ASA after CABG for NSTEMI May 17, 2007 Justin Lee Pharmacy Resident University Health Network Objectives At the end of this session, you should be able to: Explain the rationale for antiplatelet
Clopidogrel and ASA after CABG for NSTEMI May 17, 2007 Justin Lee Pharmacy Resident University Health Network Objectives At the end of this session, you should be able to: Explain the rationale for antiplatelet
Disclosures. Inpatient Management of Non-ST Elevation Acute Coronary Syndromes. Edward McNulty MD, FACC. None
 Inpatient Management of Non-ST Elevation Acute Coronary Syndromes Edward McNulty MD, FACC Assistant Clinical Professor UCSF Director, SF VAMC Cardiac Catheterization Laboratory Disclosures None New Guidelines
Inpatient Management of Non-ST Elevation Acute Coronary Syndromes Edward McNulty MD, FACC Assistant Clinical Professor UCSF Director, SF VAMC Cardiac Catheterization Laboratory Disclosures None New Guidelines
CLINICIAN INTERVIEW RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE. An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA
 RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA Dr Lincoff is an interventional cardiologist and the Vice Chairman for Research
RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA Dr Lincoff is an interventional cardiologist and the Vice Chairman for Research
A randomized trial of a fixed high dose vs a weight-adjusted low dose of intravenous heparin during coronary angioplasty
 European Heart Journal (1997) 18, 631-635 A randomized trial of a fixed high dose vs a weight-adjusted low dose of intravenous heparin during coronary angioplasty A. Boccara, H. Benamer, J.-M. Juliard,
European Heart Journal (1997) 18, 631-635 A randomized trial of a fixed high dose vs a weight-adjusted low dose of intravenous heparin during coronary angioplasty A. Boccara, H. Benamer, J.-M. Juliard,
EPTIFIBATIDE INDUCED SEVERE THROMBOCYTOPENIA IN AN ASYMPTOMATIC PATIENT
 EPTIFIBATIDE INDUCED SEVERE THROMBOCYTOPENIA IN AN ASYMPTOMATIC PATIENT 1 Dr. M. Adnan Raufi MD FACC FSCAI, 2 Ayesha S. BaigMDRama Siddiqui, 3 Nouman Akbar 4 Shakaib Jaffery International Journal of Drug
EPTIFIBATIDE INDUCED SEVERE THROMBOCYTOPENIA IN AN ASYMPTOMATIC PATIENT 1 Dr. M. Adnan Raufi MD FACC FSCAI, 2 Ayesha S. BaigMDRama Siddiqui, 3 Nouman Akbar 4 Shakaib Jaffery International Journal of Drug
A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines
 A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction 1
A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction 1
COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY
 COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY VALTER CASATI, M.D. DIVISION OF CARDIOVASCULAR ANESTHESIA AND INTENSIVE CARE CLINICA S. GAUDENZIO NOVARA (ITALY) ANTIPLATELET
COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY VALTER CASATI, M.D. DIVISION OF CARDIOVASCULAR ANESTHESIA AND INTENSIVE CARE CLINICA S. GAUDENZIO NOVARA (ITALY) ANTIPLATELET
What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation?
 What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation? Keun-Ho Park, Myung Ho Jeong, Min Goo Lee, Jum Suk Ko,
What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation? Keun-Ho Park, Myung Ho Jeong, Min Goo Lee, Jum Suk Ko,
A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in Patients with Non-ST Elevation ACS
 Angioplasty to Blunt the rise Of troponin in Acute coronary syndromes Randomized for an immediate or Delayed intervention A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in
Angioplasty to Blunt the rise Of troponin in Acute coronary syndromes Randomized for an immediate or Delayed intervention A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in
Percutaneous Coronary Interventions Without On-site Cardiac Surgery
 Percutaneous Coronary Interventions Without On-site Cardiac Surgery Hassan Al Zammar, MD,FESC Consultant & Interventional Cardiologist Head of Cardiology Department European Gaza Hospital Palestine European
Percutaneous Coronary Interventions Without On-site Cardiac Surgery Hassan Al Zammar, MD,FESC Consultant & Interventional Cardiologist Head of Cardiology Department European Gaza Hospital Palestine European
Cardiac evaluation for the noncardiac. Nathaen Weitzel MD University of Colorado Denver Dept of Anesthesiology
 Cardiac evaluation for the noncardiac patient Nathaen Weitzel MD University of Colorado Denver Dept of Anesthesiology Objectives! Review ACC / AHA guidelines as updated for 2009! Discuss new recommendations
Cardiac evaluation for the noncardiac patient Nathaen Weitzel MD University of Colorado Denver Dept of Anesthesiology Objectives! Review ACC / AHA guidelines as updated for 2009! Discuss new recommendations
Critical Review Form Therapy Objectives: Methods:
 Critical Review Form Therapy Clinical Trial Comparing Primary Coronary Angioplasty with Tissue-Plasminogen Activator for Acute Myocardial Infarction (GUSTO-IIb), NEJM 1997; 336: 1621-1628 Objectives: To
Critical Review Form Therapy Clinical Trial Comparing Primary Coronary Angioplasty with Tissue-Plasminogen Activator for Acute Myocardial Infarction (GUSTO-IIb), NEJM 1997; 336: 1621-1628 Objectives: To
3/23/2017. Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate Europace Oct;14(10): Epub 2012 Aug 24.
 Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
