Supplementary Appendix
|
|
|
- Bertina Walters
- 6 years ago
- Views:
Transcription
1 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Park S-J, Ahn J-M, Kim Y-H, et al. Trial of everolimus-eluting stents or bypass surgery for coronary disease. N Engl J Med 2015;372: DOI: /NEJMoa
2 Table of Contents Supplementary Appendix Item Page I. Investigators and Trial Personnel List 3 II. Inclusion/Exclusion Criteria 6 III. Definitions of Trial End Points 9 IV. Monitoring of Data Quality 13 V. Premature Termination of the Trial 14 VI. Figures Figure S1. Enrollment, Randomization and Follow-up 15 Figure S2. Kaplan-Meier Curves of the composite primary outcome of 16 death, myocardial infarction, or Stroke. Figure S3. Kaplan-Meier Curves for the Landmark Analyses 17 Figure S4. Kaplan-Meier Curves for Repeat Revascularization 18 Figure S5. Subgroup Analyses of the Composite of Death, Myocardial 19 Infarction, Stroke, or Any Repeat Revascularization. Figure S6. Clinical Outcomes According to the Type of Revascularization 20 and Diabetes VII. Tables Table S1. Reasons for Screening Failure for Study Enrollment 21 Table S2. Medications at discharge and follow up 22 Table S3. Clinical End Points in the CABG and PCI Groups In As- 25 Treated Analysis 2
3 I. Investigators and Trial Personnel List The following persons participated in the enrollment of patients, data collection, or study coordination and number of patients enrolled per site. Investigators: Seung-Jung Park, Jung-Min Ahn, Young-Hak Kim, Duk-Woo Park, Sung- Cheol Yun, Jong-Young Lee, Soo-Jin Kang, Seung-Whan Lee, Cheol Whan Lee, Seong-Wook Park, Suk Jung Choo, Cheol-Hyun Chung, and Jae-Won Lee, Asan Medical Center, Seoul, Korea (487); David J. Cohen, Saint Luke s Mid America Heart Institute, University of Missouri-Kansas City, Kansas City, MO, USA; Alan C. Yeung, Stanford University School of Medicine, Palo Alto, California, USA; Seung Ho Hur, Keimyung University Dongsan Medical Center, Daegu, Korea (54); Ki Bae Seung, Catholic University of Korea, St. Mary s Hospital, Seoul, Korea (52); Damras Tresukosol, Siriraj Hospital, Thailand (50); Tae Hoon Ahn, Gachon University Gil Hospital, Incheon, Korea (49); Hyuck Moon Kwon, Gangnam Severance Hospital, Seoul, Korea (39); Do-Sun Lim, and Seung-Woon Rha, Korea University Anam (23) and Guro Hospital (28), Seoul, Korea; Myung-Ho Jeong, Chonnam National University Hospital, Gwangju, Korea (18); Bong-Ki Lee, Kangwon National University Hospital, Chuncheon, Korea (17); Guo Sheng Fu, Sir Run Run Shaw Hospital, China (10); Kyoung Soo Kim, Hanyang University Medical Center, Seoul, Korea (9); Jang Ho Bae, Konyang University Hospital, Daejeon, Korea (9); Byung Ok Kim, Inje University Sanggye Paik Hospital, Seoul, Korea (6); Tiong Kiam Ong, Sarawak General Hospital, Malaysia (6); Junghan Yoon, Wonju Christian Hospital, Wonju, Korea (5); Tae-Hyun Yang, Inje University Pusan Paik Hospital, Pusan, Korea (3); Yang-Soo Jang, Yonsei University Severance Hospital, Seoul, Korea (3); Joo-Young Yang, National Health Insurance Corporation Center, Goyang, Korea (2); Jong-Seon Park, Yeungnam University Medical Center, Daegu, Korea (2); JunBo Ge, Zhongshan Hospital, China (1); Sung Yun Lee, Inje University Ilsan Paik Hospital, Goyang, Korea (1); Jun Hong Kim, Pusan National University Yangsan Hospital, Yangsan, 3
4 Korea (1); Jang-Hyun Cho, St. Carollo Hospital, Suncheon, Korea (1); Yun Seok Choi, Yeouido St. Mary s Hospital, Seoul, Korea (1); Sang-Gon Lee, Ulsan University Hospital, Ulsan, Korea (1); and Robaaya Zambahari, National Heart Institute, Malaysia (1). Author Contributions: conception and design Seung-Jung Park, Jung-Min Ahn, Young- Hak Kim, Sung-Cheol Yun and Jae-Won Lee; analysis and interpretation of data Seung- Jung Park, Jung-Min Ahn and Sung-Cheol Yun; drafting of the manuscript Seung-Jung Park, Jung-Min Ahn, Young-Hak Kim and Sung-Cheol Yun; critical revision of the manuscript for important intellectual content Seung-Jung Park, Jung-Min Ahn, Young-Hak Kim, Duk-Woo Park, Sung-Cheol Yun, Jong-Young Lee, Soo-Jin Kang, Seung-Whan Lee, Cheol Whan Lee, Seong-Wook Park, Suk Jung Choo, Cheol-Hyun Chung, Jae-Won Lee, David J. Cohen, Alan C. Yeung, Seung Ho Hur, Ki Bae Seung, Damras Tresukosol, Tae Hoon Ahn, Hyuck Moon Kwon, Do-Sun Lim, Seung-Woon Rha, Myung-Ho Jeong, Bong-Ki Lee, Guo Sheng Fu and Tiong Kiam Ong; final approval of the manuscript Seung-Jung Park, Jung-Min Ahn, Young-Hak Kim, Duk-Woo Park, Sung-Cheol Yun, Jong-Young Lee, Soo-Jin Kang, Seung-Whan Lee, Cheol Whan Lee, Seong-Wook Park, Suk Jung Choo, Cheol-Hyun Chung, Jae-Won Lee, David J. Cohen, Alan C. Yeung, Seung Ho Hur, Ki Bae Seung, Damras Tresukosol, Tae Hoon Ahn, Hyuck Moon Kwon, Do-Sun Lim, Seung-Woon Rha, Myung-Ho Jeong, Bong-Ki Lee, Guo Sheng Fu and Tiong Kiam Ong; statistical expertise Sung-Cheol Yun, Seung-Jung Park, and Jung-Min Ahn; obtaining of public funding Seung-Jung Park; administrative, technical, or logistic support Seung-Jung Park, Jung-Min Ahn, Young-Hak Kim, Duk-Woo Park, Sung-Cheol Yun, Jong-Young Lee, Soo-Jin Kang and Seung-Whan Lee; acquisition of data Seung-Jung Park, Jung-Min Ahn, Young-Hak Kim, Duk-Woo Park, Sung-Cheol Yun, Jong-Young Lee, Soo-Jin Kang, Seung-Whan Lee, Cheol Whan Lee, Seong- Wook Park, Suk Jung Choo, Cheol-Hyun Chung, Jae-Won Lee, Seung Ho Hur, Ki Bae Seung, 4
5 Damras Tresukosol, Tae Hoon Ahn, Hyuck Moon Kwon, Do-Sun Lim, Seung-Woon Rha, Myung-Ho Jeong, Bong-Ki Lee, Guo Sheng Fu and Tiong Kiam Ong. Executive Committee: Seung-Jung Park, Jung-Min Ahn, Young-Hak Kim, Duk-Woo Park, Sung-Cheol Yun, Jong-Young Lee, Soo-Jin Kang, Seung-Whan Lee, Cheol Whan Lee, Seong- Wook Park and Jae-Won Lee (University of Ulsan College of Medicine). Data and Safety Monitoring Board: Moo Song Lee (University of Ulsan College of Medicine), Jae-Joong Kim (University of Ulsan College of Medicine) and Goo-Yeong Cho (Seoul National University Bundang Hospital). Event-Adjudication Committee: Gyung-Min Park (University of Ulsan College of Medicine) and Young-Rak Cho (University of Ulsan College of Medicine). 5
6 II. Inclusion and Exclusion Criteria Inclusion criteria 1. Age 18 years. 2. Angiographically confirmed multivessel CAD [critical (> 70%) lesions in at least two major epicardial vessels ( 2.0 mm in diameter) in at least two separate coronary artery territories (LAD, LCX, RCA)] expected to be equally treatable with percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG) by clinicians at the investigating site. 3. Indication for revascularization based on symptoms of angina and/or objective evidence of myocardial ischemia. 4. Geographically accessible and willing to come in for required study visits. 5. Agreement by the patient or guardian to the study protocol and the clinical follow-up schedule, including the provision of written informed consent, as approved by the appropriate Institutional Review Board/Ethical Committee at the respective clinical site. Exclusion criteria 1. Known hypersensitivity or contraindication to any of the following: Heparin Aspirin Clopidogrel Everolimus Stainless steel and/or Contrast media (patients with documented sensitivity to contrast who can be effectively pre-medicated with steroids and diphenhydramine [e.g., for rash] may be enrolled. Patients with true anaphylaxis to prior contrast media, 6
7 however, should not be enrolled). 2. Severe congestive heart failure (class III or IV according to NYHA, or pulmonary edema) at the time of enrollment. The left ventricular ejection fraction was not considered a criterion for exclusion. 3. Planned simultaneous surgical procedure unrelated to coronary revascularization (e.g., valve repair/replacement, aneurysmectomy, carotid endarterectomy or carotid stent). 4. Previous bypass surgery. 5. Prior percutaneous coronary intervention (PCI) with DES implantation within 1 year. 6. Two or more chronic total occlusions in major coronary territories. 7. Acute ST-elevation myocardial infarction (Q-wave) within 72 hours prior to enrollment requiring revascularization. 8. Abnormal creatine kinase (CK > 2 normal) and/or abnormal CK-MB levels and/or elevated troponin levels at the time of randomization. Enrollment was allowed when these cardiac enzymes returned to normal. 9. Previous stroke within 6 months or stroke at more than 6 months with significant residual neurologic involvement. 10. Extra-cardiac illness expected to limit survival to less than 2 years, e.g., oxygendependent chronic obstructive pulmonary disease, active hepatitis, significant hepatic failure, or severe renal disease. 11. Prior history of significant bleeding (within the previous 6 months) that might be expected to recur during coronary artery bypass grafting (CABG) or PCI/DES-related anticoagulation. 12. Contraindication to either CABG or PCI/DES due to a coexisting clinical condition. 13. Intolerance or contraindication to aspirin or clopidogrel. 7
8 14. Suspected pregnancy. 15. Concurrent enrollment in another clinical trial. 16. Left main coronary artery stenosis ( 50% diameter). 8
9 III. Definitions of Trial End Points Complete Revascularization No residual stenosis > 50% in any major coronary artery or their large branches with reference diameters of the arteries adjacent to the site of stenosis >2 mm, or a bypass graft feeding the territory distal to a diseased segment. Death The primary end point includes death from any cause. In addition, the cause of death (cardiac vs. non-cardiac) will be adjudicated. If the cause of death cannot be adjudicated the most severe cause will be considered. Death is divided into 2 categories: Cardiac death is defined as death due to any of the following: 1. Acute myocardial infarction. 2. Cardiac perforation/pericardial tamponade. 3. Arrhythmia or conduction abnormality. 4. Cerebrovascular accident within 30 days of the procedure or cerebrovascular accident suspected of being related to the procedure. 5. Death due to complication of the procedure 6. Any death in which a cardiac cause cannot be excluded. Non-cardiac death is defined as a death not due to cardiac causes (as defined above), including bleeding-related death. Major Bleeding (protocol) Major Bleeding is defined as the occurrence of any of the following: intracranial bleeding, intraocular bleeding, retroperitoneal bleeding, access site hemorrhage requiring surgery or a radiological or interventional procedure, hematoma 5cm in 9
10 diameter at the puncture site, reduction in hemoglobin concentration of 4g/dL without an overt source of bleeding, reduction in hemoglobin concentration of 3g/dL with an overt source of bleeding, re-operation for bleeding, or use of any blood product transfusion. Bleeding complications will be adjudicated as either related to or unrelated to surgery. Myocardial Infarction 1. AMI prior to enrollment (if occurred within the past 1 week, it is an exclusion criterion and subject should not be enrolled) Typical rise and gradual fall of troponin or more rapid rise and fall of CK for detecting myocardial necrosis with at least one of the following: a. Ischemic symptoms or atypical symptoms of ischemia; b. Development of pathologic Q waves on the ECG, or new LBBB; OR c. ECG changes indicative of ischemia (ST segment elevation or depression); Troponin rise is defined as at least one value exceeding the 99th percentile of the value for reference control group. CK rise is defined as at least 2 values exceeding the 99th percentile for the reference group or at least one value exceeding twice the upper limit of normal for the specific institution AND an elevated CK-MB. 2. AMI within first 48 hours after the procedure The presence of new Q waves in at least 2 or more contiguous leads, or new LBBB. AND- One plasma level of CK-MB 5X upper normal limit. Periprocedural cardiac enzyme elevations in the absence of Q waves will not be considered an event. 3. AMI more than 48 hours after the procedure 10
11 Typical rise and gradual fall of troponin or more rapid rise and fall of CK-MB for detecting myocardial necrosis with at least one of the following: a. Ischemic symptoms or atypical symptoms of ischemia; b. Development of pathologic Q waves on the ECG, or new LBBB; AND c. Enzyme changes defined as one or more than one plasma level of CK- MB above upper normal level. Stent Thrombosis 1. Definite ST Angiographic confirmation of ST and at least 1 of the following signs present within 48 h: new onset of ischemic symptoms at rest, new electrocardiographic changes suggestive of acute ischemia, or typical rise and fall in cardiac biomarkers. 2. Probable ST Any unexplained death within the first 30 days after intracoronary stenting. 3. Possible ST Any unexplained death occurring from 30 days after intracoronary stenting until the end of the follow-up period. Stent thrombosis was further characterized as acute (0 h to <24 h), subacute ( 1 day to <30 days), late ( 30 days to <1 year), and very late ( 1 year to 15 months). Stroke Stroke is defined as a focal neurological deficit of central origin lasting more than 72 hours. The definitive evaluation for stroke will be conducted in both treatment arms, using the Neurological Assessment (defined in the trial protocol) at baseline, at 72 hours or hospital discharge, whichever comes first, 30 days and 24 months after the assigned 11
12 treatment is performed. Target Lesion Revascularization Any repeat PCI of the target lesion due to recurrent target lesion stenosis. A target lesion revascularization will be considered ischemia-driven if the target lesion diameter stenosis is 50% by QCA (analysis segment measurement, involving the lesion itself and/or 5 mm of proximal and/or distal margin) and any of the following occur: The subject has a positive functional study corresponding to the area served by the target lesion; ischemic ECG changes at rest in a distribution consistent with the target vessel; or ischemic symptoms referable to the target lesion. A target lesion revascularization for a diameter stenosis less than 50% might also be considered ischemia-driven by the CEC if there was a markedly positive functional study or ECG changes corresponding to the area served by the target lesion. A target lesion revascularization will also be considered as ischemia-driven if the lesion diameter stenosis is 70% in the absence of documented clinical or functional ischemia. A target lesion revascularization will be considered clinically-driven if the patient has one of the following: The subject had a positive functional study corresponding to the area served by the target lesion; ischemic ECG changes at rest in a distribution consistent with the target vessel; or ischemic symptoms referable to the target lesion. Target Vessel Revascularization Any repeat PCI or bypass surgery to target vessel. 12
13 IV. Monitoring of Data Quality For the informed consent form, enrollment criteria, and major outcomes, the research team in the Data Coordinating Center in the CardioVascular Research Foundation visited all participating centers and performed monitoring through Source Document Verification. For other variables, the central monitoring method was used. Based on the electronic case reporting form, the research team issued a Data Clarification Form identifying any logical errors, which was sent to each institution. Each investigator corrected the issues on the electronic case reporting form. Additionally, during visits to participating centers, the research team did Source Document Verification of roughly 10% of all cases. 13
14 V. Premature Termination of the Trial This study was prematurely terminated due to slow enrollment. The reason for slow enrollment was presumed to be due to the rapid spread of the clinical use of fractional flow reserve measurement (FFR). Because this study had angiographic inclusion criteria, the clinical use of FFR reduced the enrollment rate of study participants. In addition, the evolution of FFR measurement led the investigators to conclude that this trial no longer represents state-of-the-art PCI and therefore would be less relevant to future therapy. Finally, competing enrollment in another trial (FAME 3 trial [NCT ]) also limited patient enrollment. The executive committee, which also served as the steering committee, asked the data and safety monitoring board (DSMB) to consider premature termination of the trial, blinded to the clinical outcomes. The DSMB recommended stopping enrollment, as it was acknowledged that the past slow rate of enrollment would not be likely to improve in the future, and thus continuing enrollment would be futile. The executive committee accepted the DSMB recommendation. 14
15 VI. Figures Figure S1. Enrollment, Randomization and Follow-up 15
16 Figure S2. Kaplan-Meier Curves of the composite primary outcome of death, myocardial infarction, or Stroke. Event rates shown are Kaplan-Meier estimates. The P value was calculated by means of the log-rank test on the basis of all available follow-up data. 16
17 Figure S3. Kaplan-Meier Curves for the Landmark Analyses. Shown are the cumulative incidences of death (A), the composite of death or myocardial infarction (B), and myocardial infarction (C) in the two study groups, stratified on the basis of a landmark point at 30 days after randomization (vertical dashed line). 17
18 Figure S4. Kaplan-Meier Curves for Repeat Revascularization. Shown are the cumulative incidences of any repeat revascularization (A), target lesion revascularization (B), and new lesion revascularization (C) after randomization. 18
19 Figure S5. Subgroup Analyses of the Composite of Death, Myocardial Infarction, Stroke, or Any Repeat Revascularization. 19
20 Figure S6. Clinical Outcomes According to the Type of Revascularization and Diabetes. Blue bar is PCI and red bar is CABG 20
21 VII. Tables Table S1. Reasons for Screening Failure for Study Enrollment Reasons Number of Patients Left main stenosis (at least 50% diameter stenosis) 766 Concurrent enrollment in another clinical trial 639 Two or more chronic total occlusions in major coronary territories 248 Planned simultaneous surgical procedure unrelated to coronary revascularization 235 Prior CABG surgery 209 Acute ST-elevation MI (Q-wave) within 72 hours prior to enrollment requiring revascularization 253 Prior PCI with DES implantation within 1 year 192 Elevated cardiac enzymes at time of randomization 145 Heart failure (NYHA class III or IV) 83 Extra-cardiac illness that is expected to limit survival to less than 2 years 58 Contraindication to either CABG or PCI/DES because of a coexisting clinical condition 42 Previous stroke within 6 months or patients with stroke at more than 6 months with significant residual neurologic involvement 30 Prior history of significant bleeding (< 6 months) 10 Not possible to access the research center 7 Hypersensitivity or contraindication to medication 6 Intolerance or contraindication to aspirin or both clopidogrel and ticlopidine 6 21
22 Table S2. Medications at Discharge and Follow-up Medication PCI CABG P-value (N=438) (N=442) Aspirin At discharge 425/438 (97.0) 427/442 (96.6) year after randomization 397/426 (93.2) 401/433 (92.6) year after randomization 369/394 (93.7) 375/401 (93.5) year after randomization 288/342 (84.2) 286/354 (80.8) year after randomization 232/276 (84.1) 235/288 (81.6) year after randomization 136/174 (78.2) 140/184 (76.1) 0.64 Thienopyridine At discharge 423/438 (96.6) 395/442 (89.4) < year after randomization 376/426 (88.3) 299/433 (69.1) < year after randomization 277/394 (70.3) 205/401 (51.1) < year after randomization 199/342 (58.2) 174/354 (49.2) year after randomization 135/276 (48.9) 118/288 (41.0) year after randomization 93/173 (53.8) 88/182 (48.4) 0.31 Any antiplatelet drug At discharge 434/438 (99.1) 430/442 (97.3) year after randomization 420/426 (98.6) 420/433 (97.0) year after randomization 389/394 (98.7) 389/401 (97.0) year after randomization 325/342 (95.0) 323/354 (91.2) year after randomization 261/276 (94.6) 266/288 (92.4) year after randomization 160/174 (92.0) 167/184 (90.8) 0.69 Beta-blocker 22
23 At discharge 300/438 (68.5) 189/442 (42.8) < year after randomization 379/426 (89.0) 358/433 (82.7) year after randomization 257/394 (65.2) 216/401 (53.9) year after randomization 189/342 (55.3) 161/354 (45.5) year after randomization 129/276 (46.7) 123/288 (42.7) year after randomization 87/174 (50.0) 68/184 (37.0) Calcium-channel blocker At discharge 254/438 (58.0) 205/442 (46.4) year after randomization 246/426 (57.7) 225/433 (52.0) year after randomization 255/394 (57.1) 203/401 (50.6) year after randomization 160/342 (46.8) 150/354 (42.4) year after randomization 125/276 (45.3) 103/288 (35.8) year after randomization 96/174 (55.2) 67/184 (36.4) <0.001 ACE inhibitor or ARB At discharge 195/438 (44.5) 112/442 (25.3) < year after randomization 176/426 (41.3) 155/433 (35.8) year after randomization 176/394 (44.7) 143/401 (35.7) year after randomization 124/342 (36.3) 116/354 (32.8) year after randomization 89/276 (32.2) 76/288 (26.4) year after randomization 60/174 (34.5) 40/184 (21.7) Statin At discharge 364/438 (83.1) 369/442 (83.5) year after randomization 383/426 (89.9) 370/433 (85.8) year after randomization 343/394 (87.1) 333/401 (83.0) year after randomization 280/342 (81.9) 267/354 (75.4)
24 4 year after randomization 223/276 (80.8) 217/288 (75.3) year after randomization 138/174 (79.3) 138/184 (75.0) 0.33 * NE, not evaluable ACE denotes angiotensin-converting enzyme, ARB angiotensin II receptor blocker. 24
25 Table S3. Long-Term Clinical End Points in the CABG and PCI Groups In As-Treated Analysis End points PCI CABG Hazard ratio* P-value (N=464) (N=401) (95% CI) Number (percentage) Primary End Point Death, Myocardial Infarction, or Target 72 (15.5) 40 (10.0) 1.57 ( ) Vessel Revascularization Secondary End Points Death 28 ( (5.5) 1.08 ( ) 0.78 Cardiac Death 18 ( (3.7) 1.03 ( ) 0.94 Non Cardiac Death 10 (2.2) 7 (1.7) 1.21 ( ) 0.71 Myocardial Infarction 22 (4.7) 10 (2.5) 1.88 ( ) Fatal Myocardial Infarction 3 (0.6) 7 (0.2) 2.57 ( ) 0.40 Spontaneous Myocardial Infarction 20 (4.3) 5 (1.2) 3.43 ( ) Q wave Myocardial Infarction 4 (0.9) 2 (0.5) 1.69 ( )
26 Death or Myocardial Infarction 44 (9.5) 31 (7.7) 1.21 ( ) 0.42 Stroke 12 (2.6) 10 (2.5) 1.03 ( ) 0.94 Ischemic Stroke 9 (2.1) 10 (2.5) 0.77 ( ) 0.57 Hemorrhagic Stroke 3 (0.6) 0 (0) NA NA Death, Myocardial Infarction, or stroke 53 (11.4) 39 (9.7) 1.17 ( ) 0.46 Cardiac Death, Myocardial Infarction, or Stroke 44 (9.5) 33 (8.2) 1.15 ( ) 0.55 Any Repeat Revascularization 54 (11.6) 17 (4.2) 2.82 ( ) <0.001 Target Vessel Revascularization 36 (7.8) 12 (3.0) 2.63 ( ) Target Lesion Revascularization 30 (6.5) 12 (3.0) 2.18 ( ) 0.19 New Lesion Revascularization 27 (5.8) 6 (1.5) 3.93 ( ) Death, Myocardial Infarction, Stroke, or Any Repeat Revascularization Cardiac Death, Myocardial Infarction, Stroke, or Any Repeat Revascularization 92 (19.8) 52 (13.0) 1.57 ( ) (18.1) 46 (11.5) 1.62 ( ) Bleeding 26
27 TIMI Major Bleeding 23 (5.0) 139 (34.7) 0.12 ( ) <0.001 Fatal Bleeding 5 (1.1) 5 (1.2) 0.85 ( ) 0.80 *Hazard ratio and 95% confidence interval (CI) were assessed for events on the basis of all available follow-up data. P-values were calculated using the log-rank test on the basis of all available follow-up data. Thrombolysis in Myocardial Infarction (TIMI) major bleeding refers to events that were adjudicated on the basis of previously used TIMI criteria. CABG denotes coronary artery bypass graft, NA not applicable, PCI percutaneous coronary intervention. 27
Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease (BEST Trial)
 Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease (BEST Trial) Seung-Jung Park, MD, PhD On behalf of the BEST investigators Professor of Medicine, University of Ulsan College of
Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease (BEST Trial) Seung-Jung Park, MD, PhD On behalf of the BEST investigators Professor of Medicine, University of Ulsan College of
Optimal Duration of Clopidogrel Therapy with DES to Reduce Late Coronary Arterial Thrombotic Event. The DES LATE Trial
 Optimal Duration of Clopidogrel Therapy with DES to Reduce Late Coronary Arterial Thrombotic Event The DES LATE Trial Cheol Whan Lee, MD, Seung-Jung Park, MD, PhD, On Behalf of the DES LATE Investigators
Optimal Duration of Clopidogrel Therapy with DES to Reduce Late Coronary Arterial Thrombotic Event The DES LATE Trial Cheol Whan Lee, MD, Seung-Jung Park, MD, PhD, On Behalf of the DES LATE Investigators
DECISION-CTO. Optimal Medical Therapy With or Without Stenting For Coronary Chronic Total Occlusion. Seung-Jung Park, MD., PhD.
 DECISION-CTO Optimal Medical Therapy With or Without Stenting For Coronary Chronic Total Occlusion Seung-Jung Park, MD., PhD. Heart Institute, University of Ulsan College of Medicine Asan Medical Center,
DECISION-CTO Optimal Medical Therapy With or Without Stenting For Coronary Chronic Total Occlusion Seung-Jung Park, MD., PhD. Heart Institute, University of Ulsan College of Medicine Asan Medical Center,
Hyeon-Cheol Gwon, On the behalf of SMART-DATE trial investigators ACC LBCT 2018
 Six-month versus 12-month or longer dual antiplatelet therapy after percutaneous coronary intervention in patients with acute coronary syndromes (SMART-DATE): a randomized, openlabel, multicenter trial
Six-month versus 12-month or longer dual antiplatelet therapy after percutaneous coronary intervention in patients with acute coronary syndromes (SMART-DATE): a randomized, openlabel, multicenter trial
Revascularization after Drug-Eluting Stent Implantation or Coronary Artery Bypass Surgery for Multivessel Coronary Disease
 Impact of Angiographic Complete Revascularization after Drug-Eluting Stent Implantation or Coronary Artery Bypass Surgery for Multivessel Coronary Disease Young-Hak Kim, Duk-Woo Park, Jong-Young Lee, Won-Jang
Impact of Angiographic Complete Revascularization after Drug-Eluting Stent Implantation or Coronary Artery Bypass Surgery for Multivessel Coronary Disease Young-Hak Kim, Duk-Woo Park, Jong-Young Lee, Won-Jang
Incidence and Predictors of Stent Thrombosis after Percutaneous Coronary Intervention in Acute Myocardial Infarction
 Incidence and Predictors of Stent Thrombosis after Percutaneous Coronary Intervention in Acute Myocardial Infarction Sungmin Lim, Yoon Seok Koh, Hee Yeol Kim, Ik Jun Choi, Eun Ho Choo, Jin Jin Kim, Mineok
Incidence and Predictors of Stent Thrombosis after Percutaneous Coronary Intervention in Acute Myocardial Infarction Sungmin Lim, Yoon Seok Koh, Hee Yeol Kim, Ik Jun Choi, Eun Ho Choo, Jin Jin Kim, Mineok
TRATAMIENTO INVASIVO ENFERMEDAD ISQUEMICA ESTABLE. Jonathan Poveda CLINICA BIBLICA 2015
 TRATAMIENTO INVASIVO ENFERMEDAD ISQUEMICA ESTABLE Jonathan Poveda CLINICA BIBLICA 2015 COURAGE First coronary angioplasty lesion (circles) two days before (A), immediately after (B), and one month after
TRATAMIENTO INVASIVO ENFERMEDAD ISQUEMICA ESTABLE Jonathan Poveda CLINICA BIBLICA 2015 COURAGE First coronary angioplasty lesion (circles) two days before (A), immediately after (B), and one month after
The MAIN-COMPARE Study
 Long-Term Outcomes of Coronary Stent Implantation versus Bypass Surgery for the Treatment of Unprotected Left Main Coronary Artery Disease Revascularization for Unprotected Left MAIN Coronary Artery Stenosis:
Long-Term Outcomes of Coronary Stent Implantation versus Bypass Surgery for the Treatment of Unprotected Left Main Coronary Artery Disease Revascularization for Unprotected Left MAIN Coronary Artery Stenosis:
The MAIN-COMPARE Registry
 Long-Term Outcomes of Coronary Stent Implantation versus Bypass Surgery for the Treatment of Unprotected Left Main Coronary Artery Disease Revascularization for Unprotected Left MAIN Coronary Artery Stenosis:
Long-Term Outcomes of Coronary Stent Implantation versus Bypass Surgery for the Treatment of Unprotected Left Main Coronary Artery Disease Revascularization for Unprotected Left MAIN Coronary Artery Stenosis:
Seung-Woon Rha, MD, PhD FACC, FAHA, FESC, FSCAI, FAPSIC. Cardiovascular Center, Korea University Guro Hospital, Seoul, Korea.
 Controversies in Acute Myocardial Infarction - Is Early Statin Therapy is Beneficial in all Patients?- : Insights from Korea Acute Myocardial Infarction Registry (KAMIR) Seung-Woon Rha, MD, PhD FACC, FAHA,
Controversies in Acute Myocardial Infarction - Is Early Statin Therapy is Beneficial in all Patients?- : Insights from Korea Acute Myocardial Infarction Registry (KAMIR) Seung-Woon Rha, MD, PhD FACC, FAHA,
What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation?
 What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation? Keun-Ho Park, Myung Ho Jeong, Min Goo Lee, Jum Suk Ko,
What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation? Keun-Ho Park, Myung Ho Jeong, Min Goo Lee, Jum Suk Ko,
DECISION - CTO. optimal Medical Treatment in patients with. Seung-Jung Park, MD, PhD, FACC for the DECISION-CTO Study investigators
 DECISION - CTO Drug-Eluting stent Implantation versus optimal Medical Treatment in patients with ChronIc Total OccluSION Seung-Jung Park, MD, PhD, FACC for the DECISION-CTO Study investigators Asan Medical
DECISION - CTO Drug-Eluting stent Implantation versus optimal Medical Treatment in patients with ChronIc Total OccluSION Seung-Jung Park, MD, PhD, FACC for the DECISION-CTO Study investigators Asan Medical
DIAMOND Study Investigators
 Seung-Ho Hur, Wooyeong Jang, Jang-Whan Bae, Dong-Ju Choi, Young-Keun Ahn, Jong-Sun Park, Rak-Kyeong Choi, Dong-Hoon Choi, Joon-Hong Kim, Kyoo-Rok Han, Hun-Sik Park, So-Yeon Choi, Jung-Han Yoon, Hyeon-Cheol
Seung-Ho Hur, Wooyeong Jang, Jang-Whan Bae, Dong-Ju Choi, Young-Keun Ahn, Jong-Sun Park, Rak-Kyeong Choi, Dong-Hoon Choi, Joon-Hong Kim, Kyoo-Rok Han, Hun-Sik Park, So-Yeon Choi, Jung-Han Yoon, Hyeon-Cheol
Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease
 The new england journal of medicine original article Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease Seung-Jung Park, M.D., Ph.D., Jung-Min Ahn, M.D., Young-Hak Kim, M.D., Duk-Woo
The new england journal of medicine original article Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease Seung-Jung Park, M.D., Ph.D., Jung-Min Ahn, M.D., Young-Hak Kim, M.D., Duk-Woo
PROMUS Element Experience In AMC
 Promus Element Luncheon Symposium: PROMUS Element Experience In AMC Jung-Min Ahn, MD. University of Ulsan College of Medicine, Heart Institute, Asan Medical Center, Seoul, Korea PROMUS Element Clinical
Promus Element Luncheon Symposium: PROMUS Element Experience In AMC Jung-Min Ahn, MD. University of Ulsan College of Medicine, Heart Institute, Asan Medical Center, Seoul, Korea PROMUS Element Clinical
Differences in Clinical Outcomes Between Patients With ST-Elevation Versus Non-ST-Elevation Acute Myocardial Infarction in Korea
 REVIEW DOI 10.4070 / kcj.2009.39.8.297 Print ISSN 1738-5520 / On-line ISSN 1738-5555 Copyright c 2009 The Korean Society of Cardiology Differences in Clinical Outcomes Between Patients With ST-Elevation
REVIEW DOI 10.4070 / kcj.2009.39.8.297 Print ISSN 1738-5520 / On-line ISSN 1738-5555 Copyright c 2009 The Korean Society of Cardiology Differences in Clinical Outcomes Between Patients With ST-Elevation
Coronary Artery Stenosis. Insight from MAIN-COMPARE Study
 PCI for Unprotected Left Main Coronary Artery Stenosis Insight from MAIN-COMPARE Study Young-Hak Kim, MD, PhD Cardiac Center, University of Ulsan College of Medicine, Asan Medical Center Current Practice
PCI for Unprotected Left Main Coronary Artery Stenosis Insight from MAIN-COMPARE Study Young-Hak Kim, MD, PhD Cardiac Center, University of Ulsan College of Medicine, Asan Medical Center Current Practice
PCI for Long Coronary Lesion
 PCI for Long Coronary Lesion Shift of a General Idea with the Introduction of DES In the Bare Metal Stent Era Higher Restenosis Rate With Increasing Stent Length and Decreasing Stent Area Restenosis.6.4.2
PCI for Long Coronary Lesion Shift of a General Idea with the Introduction of DES In the Bare Metal Stent Era Higher Restenosis Rate With Increasing Stent Length and Decreasing Stent Area Restenosis.6.4.2
Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial
 Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial Myeong-Ki Hong, MD. PhD on behalf of the IVUS-XPL trial investigators
Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial Myeong-Ki Hong, MD. PhD on behalf of the IVUS-XPL trial investigators
Korea University Guro Hospital, Seoul, Korea * Chonnam National University Hospital, Gwangju, Korea
 Left Main Disease versus Non Left Main Disease in Acute Myocardial Infarction Patients in Real world Clinical Practice : Lessons from Korea Acute Myocardial Infarction Registry (KAMIR) Seung-Woon Rha*,
Left Main Disease versus Non Left Main Disease in Acute Myocardial Infarction Patients in Real world Clinical Practice : Lessons from Korea Acute Myocardial Infarction Registry (KAMIR) Seung-Woon Rha*,
Abstract Background: Methods: Results: Conclusions:
 Two-Year Clinical and Angiographic Outcomes of Overlapping Sirolimusversus Paclitaxel- Eluting Stents in the Treatment of Diffuse Long Coronary Lesions Kang-Yin Chen 1,2, Seung-Woon Rha 1, Yong-Jian Li
Two-Year Clinical and Angiographic Outcomes of Overlapping Sirolimusversus Paclitaxel- Eluting Stents in the Treatment of Diffuse Long Coronary Lesions Kang-Yin Chen 1,2, Seung-Woon Rha 1, Yong-Jian Li
SERUM CYSTATIN C CONCENTRATION IS A POWERFUL PROGNOSTIC INDICATOR IN PATIENTS WITH CIRRHOTIC ASCITES
 SERUM CYSTATIN C CONCENTRATION IS A POWERFUL PROGNOSTIC INDICATOR IN PATIENTS WITH CIRRHOTIC ASCITES YEON SEOK SEO, 1 SOO YOUNG PARK, 2 MOON YOUNG KIM, 3 SANG GYUNE KIM, 4 JUN YONG PARK, 5 HYUNG JOON YIM,
SERUM CYSTATIN C CONCENTRATION IS A POWERFUL PROGNOSTIC INDICATOR IN PATIENTS WITH CIRRHOTIC ASCITES YEON SEOK SEO, 1 SOO YOUNG PARK, 2 MOON YOUNG KIM, 3 SANG GYUNE KIM, 4 JUN YONG PARK, 5 HYUNG JOON YIM,
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Kim BJ, Lee E-J, Kwon SU, et al on behalf of
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Kim BJ, Lee E-J, Kwon SU, et al on behalf of
Sirolimus- Versus Paclitaxel-Eluting Stents for the Treatment of Coronary Bifurcations
 Journal of the American College of Cardiology Vol. 55, No. 16, 2010 2010 by the American College of Cardiology Foundation ISSN 0735-1097/10/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2010.02.008
Journal of the American College of Cardiology Vol. 55, No. 16, 2010 2010 by the American College of Cardiology Foundation ISSN 0735-1097/10/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2010.02.008
Count Down to COMBAT
 Count Down to COMBAT Randomized COMparison of Bypass Surgery versus AngioplasTy using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease Roxana Mehran, MD Associate Professor of
Count Down to COMBAT Randomized COMparison of Bypass Surgery versus AngioplasTy using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease Roxana Mehran, MD Associate Professor of
Duration of Dual Antiplatelet Therapy after Implantation of Drug-Eluting Stents
 The new england journal of medicine original article Duration of Dual Antiplatelet Therapy after Implantation of Drug-Eluting Stents Seung-Jung Park, M.D., Duk-Woo Park, M.D., Young-Hak Kim, M.D., Soo-Jin
The new england journal of medicine original article Duration of Dual Antiplatelet Therapy after Implantation of Drug-Eluting Stents Seung-Jung Park, M.D., Duk-Woo Park, M.D., Young-Hak Kim, M.D., Soo-Jin
AOSpine Principles Symposium Daegu
 KOREA Course program AOSpine Principles Symposium Daegu March 5, 2016 Daegu, South Korea AOSpine the leading global academic community for innovative education and research in spine care, inspiring lifelong
KOREA Course program AOSpine Principles Symposium Daegu March 5, 2016 Daegu, South Korea AOSpine the leading global academic community for innovative education and research in spine care, inspiring lifelong
Jun-Won Lee, Sang Wook Park, Jung-Woo Son, Young Jin Youn, Min-Soo Ahn, Sung Gyun Ahn, Jang-Young Kim, Byung-Soo Yoo, Junghan Yoon, Seung-Hwan Lee
 The procedural success and complication rate of the left distal radial approach for coronary angiography and percutaneous coronary intervention. Prospective observational study (LeDRA) Jun-Won Lee, Sang
The procedural success and complication rate of the left distal radial approach for coronary angiography and percutaneous coronary intervention. Prospective observational study (LeDRA) Jun-Won Lee, Sang
Long-term outcomes of simple crossover stenting from the left main to the left anterior descending coronary artery
 ORIGINAL ARTICLE Korean J Intern Med 2014;29:597-602 Long-term outcomes of simple crossover stenting from the left main to the left anterior descending coronary artery Ho-Myung Lee 1,*, Chang-Wook Nam
ORIGINAL ARTICLE Korean J Intern Med 2014;29:597-602 Long-term outcomes of simple crossover stenting from the left main to the left anterior descending coronary artery Ho-Myung Lee 1,*, Chang-Wook Nam
Ticagrelor compared with clopidogrel in patients with acute coronary syndromes the PLATO trial
 compared with clopidogrel in patients with acute coronary syndromes the PLATO trial August 30, 2009 at 08.00 CET PLATO background In NSTE-ACS and STEMI, current guidelines recommend 12 months aspirin and
compared with clopidogrel in patients with acute coronary syndromes the PLATO trial August 30, 2009 at 08.00 CET PLATO background In NSTE-ACS and STEMI, current guidelines recommend 12 months aspirin and
ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions
 Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
New Generation Drug- Eluting Stent in Korea
 New Generation Drug- Eluting Stent in Korea Young-Hak Kim, MD, PhD Department of Cardiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea Purpose To briefly introduce the
New Generation Drug- Eluting Stent in Korea Young-Hak Kim, MD, PhD Department of Cardiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea Purpose To briefly introduce the
Declaration of conflict of interest. Nothing to disclose
 Declaration of conflict of interest Nothing to disclose Hong-Seok Lim, Seung-Jea Tahk, Hyoung-Mo Yang, Jin-Woo Kim, Kyoung- Woo Seo, Byoung-Joo Choi, So-Yeon Choi, Myeong-Ho Yoon, Gyo-Seung Hwang, Joon-Han
Declaration of conflict of interest Nothing to disclose Hong-Seok Lim, Seung-Jea Tahk, Hyoung-Mo Yang, Jin-Woo Kim, Kyoung- Woo Seo, Byoung-Joo Choi, So-Yeon Choi, Myeong-Ho Yoon, Gyo-Seung Hwang, Joon-Han
Original Article. Introduction. Korean Circulation Journal
 Original Article Print ISSN 1738-552 On-line ISSN 1738-5555 Korean Circulation Journal The Practice Pattern of Percutaneous Coronary Intervention in Korea Based on Year 214 Cohort of Korean Percutaneous
Original Article Print ISSN 1738-552 On-line ISSN 1738-5555 Korean Circulation Journal The Practice Pattern of Percutaneous Coronary Intervention in Korea Based on Year 214 Cohort of Korean Percutaneous
Unprotected LM intervention
 Unprotected LM intervention Guideline for COMBAT Seung-Jung Park, MD, PhD Professor of Internal Medicine, Seoul, Korea Current Recommendation for unprotected LMCA Stenosis Class IIb C in ESC guideline
Unprotected LM intervention Guideline for COMBAT Seung-Jung Park, MD, PhD Professor of Internal Medicine, Seoul, Korea Current Recommendation for unprotected LMCA Stenosis Class IIb C in ESC guideline
COMBAT- Revised Protocol
 COMBAT- Revised Protocol Randomized COMparison of Bypass Surgery versus AngioplasTy using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease Roxana Mehran, MD Associate Professor
COMBAT- Revised Protocol Randomized COMparison of Bypass Surgery versus AngioplasTy using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease Roxana Mehran, MD Associate Professor
Impact of Lesion Length on Chronic Total Occlusion Intervention Outcomes
 Impact of Lesion Length on Chronic Total Occlusion Intervention Outcomes Seung-Woon Rha, Amro Elnagar, Byoung Geol Choi, Sung Il Im, Sun Won Kim, Jin Oh Na, Seong Woo Han, Cheol Ung Choi, Hong Euy Lim,
Impact of Lesion Length on Chronic Total Occlusion Intervention Outcomes Seung-Woon Rha, Amro Elnagar, Byoung Geol Choi, Sung Il Im, Sun Won Kim, Jin Oh Na, Seong Woo Han, Cheol Ung Choi, Hong Euy Lim,
LAMIS (Livalo in AMI Study)
 JCR 2018. 12. 8 LAMIS (Livalo in AMI Study) Young Joon Hong Division of Cardiology, Chonnam National University Hospital Gwangju, Korea Trend of hypercholesterolemia in Korea < Prevalence of hypercholesterolemia
JCR 2018. 12. 8 LAMIS (Livalo in AMI Study) Young Joon Hong Division of Cardiology, Chonnam National University Hospital Gwangju, Korea Trend of hypercholesterolemia in Korea < Prevalence of hypercholesterolemia
1. Whether the risks of stent thrombosis (ST) and major adverse cardiovascular and cerebrovascular events (MACCE) differ from BMS and DES
 1 Comparison of Ischemic and Bleeding Events After Drug- Eluting Stents or Bare Metal Stents in Subjects Receiving Dual Antiplatelet Therapy: Results from the Randomized Dual Antiplatelet Therapy (DAPT)
1 Comparison of Ischemic and Bleeding Events After Drug- Eluting Stents or Bare Metal Stents in Subjects Receiving Dual Antiplatelet Therapy: Results from the Randomized Dual Antiplatelet Therapy (DAPT)
Safety of Single- Versus Multi-vessel Angioplasty for Patients with AMI and Multi-vessel CAD
 Safety of Single- Versus Multi-vessel Angioplasty for Patients with AMI and Multi-vessel CAD Mun K. Hong, MD Associate Professor of Medicine Director, Cardiovascular Intervention and Research Weill Cornell
Safety of Single- Versus Multi-vessel Angioplasty for Patients with AMI and Multi-vessel CAD Mun K. Hong, MD Associate Professor of Medicine Director, Cardiovascular Intervention and Research Weill Cornell
Journal of the American College of Cardiology Vol. 46, No. 5, by the American College of Cardiology Foundation ISSN /05/$30.
 Journal of the American College of Cardiology Vol. 46, No. 5, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.06.009
Journal of the American College of Cardiology Vol. 46, No. 5, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.06.009
WHI Form Report of Cardiovascular Outcome Ver (For items 1-11, each question specifies mark one or mark all that apply.
 WHI Form - Report of Cardiovascular Outcome Ver. 6. COMMENTS To be completed by Physician Adjudicator Date Completed: - - (M/D/Y) Adjudicator Code: OMB# 095-044 Exp: 4/06 -Affix label here- Clinical Center/ID:
WHI Form - Report of Cardiovascular Outcome Ver. 6. COMMENTS To be completed by Physician Adjudicator Date Completed: - - (M/D/Y) Adjudicator Code: OMB# 095-044 Exp: 4/06 -Affix label here- Clinical Center/ID:
Successful Percutaneous Coronary Intervention in a Centenarian Patient With Acute Myocardial Infarction
 Case Report Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Successful Percutaneous Coronary Intervention in a Centenarian Patient With Acute Myocardial Infarction Ki Hong Lee, MD,
Case Report Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Successful Percutaneous Coronary Intervention in a Centenarian Patient With Acute Myocardial Infarction Ki Hong Lee, MD,
1. Diabetes mellitus (DM) is associated with worse clinical and angiographic outcomes even in acute myocardial Infarction (AMI) patients.
 Midterm Clinical Outcomes of Insulin Requiring Diabetes Mellitus versus Non-insulin Dependent Diabetes Mellitus in Acute Myocardial Infarction Patients in Drug Eluting Stent Era : Insight from Korea Acute
Midterm Clinical Outcomes of Insulin Requiring Diabetes Mellitus versus Non-insulin Dependent Diabetes Mellitus in Acute Myocardial Infarction Patients in Drug Eluting Stent Era : Insight from Korea Acute
Supplementary material 1. Definitions of study endpoints (extracted from the Endpoint Validation Committee Charter) 1.
 Rationale, design, and baseline characteristics of the SIGNIFY trial: a randomized, double-blind, placebo-controlled trial of ivabradine in patients with stable coronary artery disease without clinical
Rationale, design, and baseline characteristics of the SIGNIFY trial: a randomized, double-blind, placebo-controlled trial of ivabradine in patients with stable coronary artery disease without clinical
Role of Clopidogrel in Acute Coronary Syndromes. Hossam Kandil,, MD. Professor of Cardiology Cairo University
 Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
APPENDIX F: CASE REPORT FORM
 APPENDIX F: CASE REPORT FORM Instruction: Complete this form to notify all ACS admissions at your centre to National Cardiovascular Disease Registry. Where check boxes are provided, check ( ) one or more
APPENDIX F: CASE REPORT FORM Instruction: Complete this form to notify all ACS admissions at your centre to National Cardiovascular Disease Registry. Where check boxes are provided, check ( ) one or more
NCDR CathPCI Registry v4.4 Diagnostic Catheterization and Percutaneous Coronary Intervention Registry
 A. DEMOGRAPHICS Last Name 2000 : First Name 2010 : Middle Name 2020 : SSN 2030 : - - - SSN N/A 2031 Patient ID 2040 : (auto) Other ID 2045 : Birth Date 2050 : *** @ Sex 2060 : *** @ O Male O Female Race:
A. DEMOGRAPHICS Last Name 2000 : First Name 2010 : Middle Name 2020 : SSN 2030 : - - - SSN N/A 2031 Patient ID 2040 : (auto) Other ID 2045 : Birth Date 2050 : *** @ Sex 2060 : *** @ O Male O Female Race:
Prevention of Coronary Stent Thrombosis and Restenosis
 Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017
 EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017 Igor F. Palacios, MD Director of Interventional Cardiology Professor of Medicine Massachusetts
EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017 Igor F. Palacios, MD Director of Interventional Cardiology Professor of Medicine Massachusetts
What oral antiplatelet therapy would you choose? a) ASA alone b) ASA + Clopidogrel c) ASA + Prasugrel d) ASA + Ticagrelor
 76 year old female Prior Hypertension, Hyperlipidemia, Smoking On Hydrochlorothiazide, Atorvastatin New onset chest discomfort; 2 episodes in past 24 hours Heart rate 122/min; BP 170/92 mm Hg, Killip Class
76 year old female Prior Hypertension, Hyperlipidemia, Smoking On Hydrochlorothiazide, Atorvastatin New onset chest discomfort; 2 episodes in past 24 hours Heart rate 122/min; BP 170/92 mm Hg, Killip Class
Which drug do you prefer for stable CAD? - P2Y12 inhibitor
 Which drug do you prefer for stable CAD? - P2Y12 inhibitor Jung Rae Cho, MD, PhD Cardiovascular Division, Department of Internal Medicine Kangnam Sacred Heart Hospital, Hallym University Medical Center,
Which drug do you prefer for stable CAD? - P2Y12 inhibitor Jung Rae Cho, MD, PhD Cardiovascular Division, Department of Internal Medicine Kangnam Sacred Heart Hospital, Hallym University Medical Center,
Periprocedural Myocardial Infarction and Clinical Outcome In Bifurcation Lesion
 Periprocedural Myocardial Infarction and Clinical Outcome In Bifurcation Lesion Hyeon-Cheol Gwon Cardiac and Vascular Center Samsung Medical Center Sungkyunkwan University School of Medicine Dr. Hyeon-Cheol
Periprocedural Myocardial Infarction and Clinical Outcome In Bifurcation Lesion Hyeon-Cheol Gwon Cardiac and Vascular Center Samsung Medical Center Sungkyunkwan University School of Medicine Dr. Hyeon-Cheol
PCI for In-Stent Restenosis. CardioVascular Research Foundation
 PCI for In-Stent Restenosis ISR of BMS Patterns of In-Stent Restenosis Pattern I : Focal Type IA: Articulation / Gap Type IB: Marginal Type IC: Focal body Type ID: Multifocal Pattern II,III,IV : Diffuse
PCI for In-Stent Restenosis ISR of BMS Patterns of In-Stent Restenosis Pattern I : Focal Type IA: Articulation / Gap Type IB: Marginal Type IC: Focal body Type ID: Multifocal Pattern II,III,IV : Diffuse
Coronary Artery Bypass Grafting vs. Drug-Eluting Stent Implantation for Multivessel Disease in Patients with Chronic Kidney Disease
 Original Article https://doi.org/1.47/kcj.216.439 Print ISSN 1738-552 On-line ISSN 1738-5555 Korean Circulation Journal Coronary Artery Bypass Grafting vs. Drug-Eluting Stent Implantation for Multivessel
Original Article https://doi.org/1.47/kcj.216.439 Print ISSN 1738-552 On-line ISSN 1738-5555 Korean Circulation Journal Coronary Artery Bypass Grafting vs. Drug-Eluting Stent Implantation for Multivessel
Medicine OBSERVATIONAL STUDY
 Medicine OBSERVATIONAL STUDY It Is Not Mandatory to Use Triple Rather Than Dual Anti-Platelet Therapy After a Percutaneous Coronary Intervention With a Second-Generation Drug-Eluting Stent Ju-Youn Kim,
Medicine OBSERVATIONAL STUDY It Is Not Mandatory to Use Triple Rather Than Dual Anti-Platelet Therapy After a Percutaneous Coronary Intervention With a Second-Generation Drug-Eluting Stent Ju-Youn Kim,
HEART AND SOUL STUDY OUTCOME EVENT - MORBIDITY REVIEW FORM
 REVIEW DATE REVIEWER'S ID HEART AND SOUL STUDY OUTCOME EVENT - MORBIDITY REVIEW FORM : DISCHARGE DATE: RECORDS FROM: Hospitalization ER Please check all that may apply: Myocardial Infarction Pages 2, 3,
REVIEW DATE REVIEWER'S ID HEART AND SOUL STUDY OUTCOME EVENT - MORBIDITY REVIEW FORM : DISCHARGE DATE: RECORDS FROM: Hospitalization ER Please check all that may apply: Myocardial Infarction Pages 2, 3,
Everolimus-Eluting Stent Implantation for Unprotected Left Main Coronary Artery Stenosis
 JACC: CARDIOVASCULAR INTERVENTIONS VOL. 5, NO. 7, 2012 2012 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. http://dx.doi.org/10.1016/j.jcin.2012.05.002
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 5, NO. 7, 2012 2012 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. http://dx.doi.org/10.1016/j.jcin.2012.05.002
Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction
 Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction The Bavarian Reperfusion Alternatives Evaluation (BRAVE) 4
Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction The Bavarian Reperfusion Alternatives Evaluation (BRAVE) 4
Research Article. Open Access
 Journal of Geriatric Cardiology (2015) 12: 208 217 2015 JGC All rights reserved; www.jgc301.com Research Article Open Access Comparison of clinical outcomes between culprit vessel only and multivessel
Journal of Geriatric Cardiology (2015) 12: 208 217 2015 JGC All rights reserved; www.jgc301.com Research Article Open Access Comparison of clinical outcomes between culprit vessel only and multivessel
FastTest. You ve read the book now test yourself
 FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
PCI for Left Anterior Descending Artery Ostial Stenosis
 PCI for Left Anterior Descending Artery Ostial Stenosis Why do you hesitate PCI for LAD ostial stenosis? LAD Ostial Lesion Limitations of PCI High elastic recoil Involvement of the distal left main coronary
PCI for Left Anterior Descending Artery Ostial Stenosis Why do you hesitate PCI for LAD ostial stenosis? LAD Ostial Lesion Limitations of PCI High elastic recoil Involvement of the distal left main coronary
TCTAP Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI
 Indian TUXEDO Trial In Medically Treated Diabetics Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI Executive Director and Dean Escorts Heart Institute & Medical Research Center and Fortis Hospitals, New Delhi
Indian TUXEDO Trial In Medically Treated Diabetics Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI Executive Director and Dean Escorts Heart Institute & Medical Research Center and Fortis Hospitals, New Delhi
PCI vs. CABG From BARI to Syntax, Is The Game Over?
 PCI vs. CABG From BARI to Syntax, Is The Game Over? Seung-Jung Park, MD, PhD Professor of Medicine, University of Ulsan College of Medicine Asan Medical Center, Seoul, Korea PCI vs CABG Multi-Vessel Disease
PCI vs. CABG From BARI to Syntax, Is The Game Over? Seung-Jung Park, MD, PhD Professor of Medicine, University of Ulsan College of Medicine Asan Medical Center, Seoul, Korea PCI vs CABG Multi-Vessel Disease
A Randomized Trial Evaluating Clinically Significant Bleeding with Low-Dose Rivaroxaban vs Aspirin, in Addition to P2Y12 inhibition, in ACS
 A Randomized Trial Evaluating Clinically Significant Bleeding with Low-Dose Rivaroxaban vs Aspirin, in Addition to P2Y12 inhibition, in ACS Magnus Ohman MB, on behalf of the GEMINI-ACS-1 Investigators
A Randomized Trial Evaluating Clinically Significant Bleeding with Low-Dose Rivaroxaban vs Aspirin, in Addition to P2Y12 inhibition, in ACS Magnus Ohman MB, on behalf of the GEMINI-ACS-1 Investigators
Remodeling of the Remnant Aorta after Acute Type A Aortic Dissection Surgery
 Remodeling of the Remnant Aorta after Acute Type A Aortic Dissection Surgery Are Young Patients More Likely to Develop Adverse Aortic Remodeling of the Remnant Aorta Over Time? Suk Jung Choo¹, Jihoon Kim¹,
Remodeling of the Remnant Aorta after Acute Type A Aortic Dissection Surgery Are Young Patients More Likely to Develop Adverse Aortic Remodeling of the Remnant Aorta Over Time? Suk Jung Choo¹, Jihoon Kim¹,
Supplementary Online Content
 Supplementary Online Content Berwanger O, Santucci EV, de Barros e Silva PGM, et al; SECURE-PCI Investigators. Effect of loading dose of atorvastatin prior to planned percutaneous coronary intervention
Supplementary Online Content Berwanger O, Santucci EV, de Barros e Silva PGM, et al; SECURE-PCI Investigators. Effect of loading dose of atorvastatin prior to planned percutaneous coronary intervention
Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G.
 Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G. Ellis MD Dean J. Kereiakes MD and Gregg W. Stone MD for the ABSORB
Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G. Ellis MD Dean J. Kereiakes MD and Gregg W. Stone MD for the ABSORB
Beta-blockers in Patients with Mid-range Left Ventricular Ejection Fraction after AMI Improved Clinical Outcomes
 Beta-blockers in Patients with Mid-range Left Ventricular Ejection Fraction after AMI Improved Clinical Outcomes Seung-Jae Joo and other KAMIR-NIH investigators Department of Cardiology, Jeju National
Beta-blockers in Patients with Mid-range Left Ventricular Ejection Fraction after AMI Improved Clinical Outcomes Seung-Jae Joo and other KAMIR-NIH investigators Department of Cardiology, Jeju National
Triple Therapy: A review of the evidence in acute coronary syndrome. Stephanie Kling, PharmD, BCPS Sanford Health
 Triple Therapy: A review of the evidence in acute coronary syndrome Stephanie Kling, PharmD, BCPS Sanford Health Objectives 1. Describe how the presented topic impacts patient outcomes. 2. Review evidence
Triple Therapy: A review of the evidence in acute coronary syndrome Stephanie Kling, PharmD, BCPS Sanford Health Objectives 1. Describe how the presented topic impacts patient outcomes. 2. Review evidence
Myocardial Infarction In Dr.Yahya Kiwan
 Myocardial Infarction In 2007 Dr.Yahya Kiwan New Definition Of Acute Myocardial Infarction The term of myocardial infarction should be used when there is evidence of myocardial necrosis in a clinical setting
Myocardial Infarction In 2007 Dr.Yahya Kiwan New Definition Of Acute Myocardial Infarction The term of myocardial infarction should be used when there is evidence of myocardial necrosis in a clinical setting
FFR-guided Jailed Side Branch Intervention
 FFR-guided Jailed Side Branch Intervention - Pressure wire in Bifurcation lesions - Bon-Kwon Koo, MD, PhD Seoul National University Hospital, Seoul, Korea Bifurcation Lesions Bifurcation Lesions Still
FFR-guided Jailed Side Branch Intervention - Pressure wire in Bifurcation lesions - Bon-Kwon Koo, MD, PhD Seoul National University Hospital, Seoul, Korea Bifurcation Lesions Bifurcation Lesions Still
Angiotensin II type 1 receptor blockers as a first choice in patients with acute myocardial infarction
 ORIGINAL ARTICLE Korean J Intern Med 2016;31:267-276 Angiotensin II type 1 receptor blockers as a first choice in patients with acute myocardial infarction Jang Hoon Lee 1, Myung Hwan Bae 1, Dong Heon
ORIGINAL ARTICLE Korean J Intern Med 2016;31:267-276 Angiotensin II type 1 receptor blockers as a first choice in patients with acute myocardial infarction Jang Hoon Lee 1, Myung Hwan Bae 1, Dong Heon
Fractional Flow Reserve: Review of the latest data
 Fractional Flow Reserve: Review of the latest data Michalis Hamilos, MD, PhD, FESC University Hospital of Heraklion Fractional Flow Reserve (FFR) Coronary angiography does not always tell the truth Most
Fractional Flow Reserve: Review of the latest data Michalis Hamilos, MD, PhD, FESC University Hospital of Heraklion Fractional Flow Reserve (FFR) Coronary angiography does not always tell the truth Most
Clinical outcomes between different stent designs with the same polymer and drug: comparison between the Taxus Express and Taxus Liberte stents
 ORIGINAL ARTICLE Korean J Intern Med 2013;28:72-80 Clinical outcomes between different stent designs with the same polymer and drug: comparison between the Taxus Express and Taxus Liberte stents Jang-Won
ORIGINAL ARTICLE Korean J Intern Med 2013;28:72-80 Clinical outcomes between different stent designs with the same polymer and drug: comparison between the Taxus Express and Taxus Liberte stents Jang-Won
Journal of the American College of Cardiology Vol. 57, No. 21, by the American College of Cardiology Foundation ISSN /$36.
 Journal of the American College of Cardiology Vol. 57, No. 21, 2011 2011 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2011.01.033
Journal of the American College of Cardiology Vol. 57, No. 21, 2011 2011 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2011.01.033
Effect of Early Statin Treatment in Patients with Cardiogenic Shock Complicating Acute Myocardial Infarction
 Original Article Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Effect of Early Statin Treatment in Patients with Cardiogenic Shock Complicating Acute Myocardial Infarction Doo
Original Article Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Effect of Early Statin Treatment in Patients with Cardiogenic Shock Complicating Acute Myocardial Infarction Doo
Optimal Duration and Dose of Antiplatelet Therapy after PCI
 Optimal Duration and Dose of Antiplatelet Therapy after PCI Donghoon Choi, MD, PhD Severance Cardiovascular Center Yonsei University College of Medicine Optimal Duration of Antiplatelet Therapy after PCI
Optimal Duration and Dose of Antiplatelet Therapy after PCI Donghoon Choi, MD, PhD Severance Cardiovascular Center Yonsei University College of Medicine Optimal Duration of Antiplatelet Therapy after PCI
Stent Fracture and Longitudinal Compression on CT Angiography between the
 2014 ASCI Stent Fracture and Longitudinal Compression on CT Angiography between the First- and New-Generation Drug-Eluting Stent Mi Sun Chung, Dong Hyun Yang,Young-Hak Kim, Jae-Hyung Roh, Joon-Won Kang,
2014 ASCI Stent Fracture and Longitudinal Compression on CT Angiography between the First- and New-Generation Drug-Eluting Stent Mi Sun Chung, Dong Hyun Yang,Young-Hak Kim, Jae-Hyung Roh, Joon-Won Kang,
Efficacy of eculizumab in paroxysmal nocturnal hemoglobinuria patients with or without aplastic anemia: prospective study of a Korean PNH cohort
 BLOOD RESEARCH VOLUME 52 ㆍ NUMBER 3 September 2017 ORIGINAL ARTICLE Efficacy of eculizumab in paroxysmal nocturnal hemoglobinuria patients with or without aplastic anemia: prospective study of a Korean
BLOOD RESEARCH VOLUME 52 ㆍ NUMBER 3 September 2017 ORIGINAL ARTICLE Efficacy of eculizumab in paroxysmal nocturnal hemoglobinuria patients with or without aplastic anemia: prospective study of a Korean
A New Strategy for Discontinuation of Dual Antiplatelet Therapy
 Journal of the American College of Cardiology Vol. 60, No. 15, 2012 2012 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. http://dx.doi.org/10.1016/j.jacc.2012.06.043
Journal of the American College of Cardiology Vol. 60, No. 15, 2012 2012 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. http://dx.doi.org/10.1016/j.jacc.2012.06.043
Acute coronary syndromes
 Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Michael Mack, M.D. Baylor Healthcare System Heart Hospital Baylor Plano Dallas, TX
 Michael Mack, M.D. Baylor Healthcare System Heart Hospital Baylor Plano Dallas, TX Maquet, Inc.,- unpaid consultant Cordis, Inc.,- unpaid consultant Boston Scientific, Inc.,- travel expenses paid for Syntax
Michael Mack, M.D. Baylor Healthcare System Heart Hospital Baylor Plano Dallas, TX Maquet, Inc.,- unpaid consultant Cordis, Inc.,- unpaid consultant Boston Scientific, Inc.,- travel expenses paid for Syntax
(For items 1-12, each question specifies mark one or mark all that apply.)
 Form 121 - Report of Cardiovascular Outcome Ver. 9.2 COMMENTS -Affix label here- Member ID: - - To be completed by Physician Adjudicator Date Completed: - - (M/D/Y) Adjudicator Code: - Central Case No.:
Form 121 - Report of Cardiovascular Outcome Ver. 9.2 COMMENTS -Affix label here- Member ID: - - To be completed by Physician Adjudicator Date Completed: - - (M/D/Y) Adjudicator Code: - Central Case No.:
Diagnosis and Management of Acute Myocardial Infarction
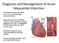 Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Case Report Left Main Stenosis. Percutaneous Coronary Intervention (PCI) or Coronary Artery Bypass Graft Surgery (CABG)?
 Cronicon OPEN ACCESS CARDIOLOGY Case Report Left Main Stenosis. Percutaneous Coronary Intervention (PCI) or Coronary Artery Bypass Graft Surgery (CABG)? Valentin Hristov* Department of Cardiology, Specialized
Cronicon OPEN ACCESS CARDIOLOGY Case Report Left Main Stenosis. Percutaneous Coronary Intervention (PCI) or Coronary Artery Bypass Graft Surgery (CABG)? Valentin Hristov* Department of Cardiology, Specialized
FFR-guided Complete vs. Culprit Only Revascularization in AMI Patients Ki Hong Choi, MD On Behalf of FRAME-AMI Investigators
 FFR-guided Complete vs. Culprit Only Revascularization in AMI Patients Ki Hong Choi, MD On Behalf of FRAME-AMI Investigators Heart Vascular Stroke Institute, Samsung Medical Center, Seoul, Republic of
FFR-guided Complete vs. Culprit Only Revascularization in AMI Patients Ki Hong Choi, MD On Behalf of FRAME-AMI Investigators Heart Vascular Stroke Institute, Samsung Medical Center, Seoul, Republic of
Bivalirudin Clinical Trials Update Evidence and Future Perspectives
 Bivalirudin Clinical Trials Update Evidence and Future Perspectives Andreas Baumbach Consultant Cardiologist/ hon. Reader in Cardiology Bristol Heart Institute University Hospitals Bristol MY CONFLICTS
Bivalirudin Clinical Trials Update Evidence and Future Perspectives Andreas Baumbach Consultant Cardiologist/ hon. Reader in Cardiology Bristol Heart Institute University Hospitals Bristol MY CONFLICTS
2017 Obesity Fact Sheet
 2017 Fact Sheet Welcome message 2017 Fact Sheet Dear Colleagues, It is my great honor and pleasure to publish the 2017 Fact Sheet of the Korean Society for the Study of (KSSO). The 2017 Fact Sheet is the
2017 Fact Sheet Welcome message 2017 Fact Sheet Dear Colleagues, It is my great honor and pleasure to publish the 2017 Fact Sheet of the Korean Society for the Study of (KSSO). The 2017 Fact Sheet is the
Ticagrelor compared with clopidogrel in patients with acute coronary syndromes the PLATelet Inhibition and patient Outcomes trial
 compared with clopidogrel in patients with acute coronary syndromes the PLATelet Inhibition and patient Outcomes trial Outcomes in patients with and planned PCI Ph.Gabriel Steg*, Stefan James, Robert A
compared with clopidogrel in patients with acute coronary syndromes the PLATelet Inhibition and patient Outcomes trial Outcomes in patients with and planned PCI Ph.Gabriel Steg*, Stefan James, Robert A
Clinical Trial Synopsis TL-OPI-516, NCT#
 Clinical Trial Synopsis, NCT#00225277 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl Versus
Clinical Trial Synopsis, NCT#00225277 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl Versus
Benefit of Early Statin Therapy in Patients With Acute Myocardial Infarction Who Have Extremely Low Low-Density Lipoprotein Cholesterol
 Journal of the American College of Cardiology Vol. 58, No. 16, 2011 2011 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2011.05.057
Journal of the American College of Cardiology Vol. 58, No. 16, 2011 2011 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2011.05.057
Supplementary Appendix
 Supplementary Appendix Increased Risk of Atrial Fibrillation and Thromboembolism in Patients with Severe Psoriasis: a Nationwide Population-based Study Tae-Min Rhee, MD 1, Ji Hyun Lee, MD 2, Eue-Keun Choi,
Supplementary Appendix Increased Risk of Atrial Fibrillation and Thromboembolism in Patients with Severe Psoriasis: a Nationwide Population-based Study Tae-Min Rhee, MD 1, Ji Hyun Lee, MD 2, Eue-Keun Choi,
A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in Patients with Non-ST Elevation ACS
 Angioplasty to Blunt the rise Of troponin in Acute coronary syndromes Randomized for an immediate or Delayed intervention A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in
Angioplasty to Blunt the rise Of troponin in Acute coronary syndromes Randomized for an immediate or Delayed intervention A Multicenter Randomized Trial of Immediate Versus Delayed Invasive Strategy in
Coronary vasospastic angina (VA) is relatively common in
 Clinical Outcomes of Vasospastic Angina Patients Presenting With Acute Coronary Syndrome Sung Woo Cho, MD, PhD;* Taek Kyu Park, MD;* Hye Bin Gwag, MD; A. Young Lim, MD; Min Seok Oh, MD; Da Hyon Lee, MD;
Clinical Outcomes of Vasospastic Angina Patients Presenting With Acute Coronary Syndrome Sung Woo Cho, MD, PhD;* Taek Kyu Park, MD;* Hye Bin Gwag, MD; A. Young Lim, MD; Min Seok Oh, MD; Da Hyon Lee, MD;
The Site of Plaque Rupture in Native Coronary Arteries
 Journal of the American College of Cardiology Vol. 46, No. 2, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.03.067
Journal of the American College of Cardiology Vol. 46, No. 2, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.03.067
Percutaneous Intervention of Unprotected Left Main Disease
 Percutaneous Intervention of Unprotected Left Main Disease Technical feasibility and Clinical outcomes Seung-Jung Park, MD, PhD, FACC Professor of Internal Medicine Asan Medical Center, Seoul, Korea Unprotected
Percutaneous Intervention of Unprotected Left Main Disease Technical feasibility and Clinical outcomes Seung-Jung Park, MD, PhD, FACC Professor of Internal Medicine Asan Medical Center, Seoul, Korea Unprotected
Original Article. Korean Circulation Journal
 Original Article Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Clinical Outcomes in Patients with Intermediate Coronary Stenoses: MINIATURE Investigators (Korea MultIceNter TrIal
Original Article Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Clinical Outcomes in Patients with Intermediate Coronary Stenoses: MINIATURE Investigators (Korea MultIceNter TrIal
Randomized Trial of Stents versus Bypass Surgery for Left Main Coronary Artery Disease
 T h e n e w e ngl a nd j o u r na l o f m e dic i n e original article Randomized Trial of Stents versus Bypass Surgery for Left Main Coronary Artery Disease Seung-Jung Park, M.D., Young-Hak Kim, M.D.,
T h e n e w e ngl a nd j o u r na l o f m e dic i n e original article Randomized Trial of Stents versus Bypass Surgery for Left Main Coronary Artery Disease Seung-Jung Park, M.D., Young-Hak Kim, M.D.,
