1. Whether the risks of stent thrombosis (ST) and major adverse cardiovascular and cerebrovascular events (MACCE) differ from BMS and DES
|
|
|
- Ethelbert Stone
- 5 years ago
- Views:
Transcription
1 1 Comparison of Ischemic and Bleeding Events After Drug- Eluting Stents or Bare Metal Stents in Subjects Receiving Dual Antiplatelet Therapy: Results from the Randomized Dual Antiplatelet Therapy (DAPT) Study Dean J. Kereiakes, Robert Yeh, Joseph M. Massaro, Priscilla-Driscoll-Shempp, Donald E. Cutlip, Sharon-Lise T. Normand, P. Gabriel Steg, Anthony Gershlick, Jean Francois Tanguay, Stephan Windecker, Kirk Garratt, David Kandzari, David Lee, Daniel Simon, Adrian Corneliu Iancu, Jaroslaw Trebacz, Laura Mauri, on behalf of he Dual Antiplatelet Therapy (DAPT) Study Investigators
2 2 DAPT Trial Secondary Analysis: Bare metal stents are a commonly used alternative to drug eluting stents (DES) particularly for patients presenting with acute coronary syndromes or in whom dual antiplatelet therapy (DAPT) has perceived increased bleeding risk. We aimed to determine: 1. Whether the risks of stent thrombosis (ST) and major adverse cardiovascular and cerebrovascular events (MACCE) differ from BMS and DES 2. Whether the optimal duration of DAPT therapy differs for BMS and DES
3 DAPT: Study Design Eligible for Enrollment after PCI Any PCI with FDA approved DES or BMS >18 years of age No contraindications to dual antiplatelet therapy Able and willing to provide written informed consent Eligible for Randomization at 12 m Stratified by DES v BMS, drug type, study site, complexity (ACS or lesion-based) Not Eligible for Randomization at 12 m Death MI or repeat PCI at > 6 weeks CABG Stroke Major Bleed Poor Compliance Total 33 month follow-up 18 mos 12 m DAPT Arm Aspirin + blinded placebo 30 m DAPT Arm Aspirin + blinded thienopyridine Randomized treatment period m Observation period 30-33m off treatment Total 33 month follow-up Primary analysis Randomized DES treated subjects, 12-30m Secondary analysis 1.propensity matched BMS to DES 0-33m 2. BMS randomized 12 vs 30m ITT 2 co-primary endpoints: stent thrombosis (ST) and MACCE (death, myocardial infarction or stroke) Powered safety endpoint: moderate/severe bleeding (GUSTO) Mauri, Kereiakes et al AHJ 2010;160:
4 Subject Flow: 452 Sites / 11 Countries 4 Index Stent Procedure 0-12 Months: All Subjects on Open-Label DAPT At Month 12: 1:1 Randomization Occurs Months: Blinded Treatment Occurs Follow-Up 30 Months: Primary End Point 33 Months DES Treated Subjects 22,866 9,961 5,020 Receive Thienopyridine + Aspirin 4,941 Receive Placebo + Aspirin 9,499 (95.4%) 9,390 (94.3%) BMS Treated Subjects 2,816 1, Receive Thienopyridine + Aspirin 845 Receive Placebo + Aspirin 1,580 (93.7%) 1,565 (92.8%)
5 Prespecified Key Secondary Analysis Propensity matched BMS to DES 0-33 months comparison Hypothesis: DES are non-inferior to BMS for ST and/or MACCE using risk difference (RD) approach Analysis population: Enrolled subjects with 29 months follow-up or ST / MACCE event. BMS to DES match (1: 1-8 ratio) exactly on STEMI and remaining (total 55) variables via propensity score using caliper of 0.10.
6 Prespecified Key Secondary Analysis Sample size assumptions: a. True ST and MACCE rates in both DES + BMS over 0-33 months 1.67%, 9.5% respectively b. NI margins 0.97% (ST); 2.28% (MACCE) c. Minimum matched DES:BMS ratio 2: evaluable DES matched with 1965 evaluable BMS subjects provides >80% power to reject null hypothesis using 1-sided test of non-inferiority at significance for each endpoint BMS randomized 30 vs 12 months DAPT therapy (ITT)
7 7 Propensity Analysis Cohort Baseline Characteristics Measure DES N=13257 Before Match BMS N=2056 After Match (weighted for match ratio) DES N=8308 BMS N=1718 Standardized Difference Clinical Characteristic Age (years) Female 27.0% 25.6% 26.4% 26.4% Race Non-White 10.0% 8.1% 9.0% 8.6% Hispanic or Latino 3.7% 5.1% 4.8% 4.7% Weight (kg) BMI Diabetes mellitus 32.0% 23.8% 25.8% 25.6% Hypertension 76.3% 67.0% 69.7% 69.6% Cigarette smoker (within past year) 25.0% 42.2% 36.5% 38.7% Stroke / TIA 3.4% 4.9% 4.4% 5.0% Congestive heart failure 5.6% 4.8% 5.2% 5.0% Peripheral arterial disease 6.6% 6.0% 6.5% 6.4% Prior PCI 33.4% 20.2% 23.2% 22.7% Prior CABG 13.1% 7.5% 8.3% 8.3% Previous MI 22.6% 20.5% 21.0% 21.6% Positive Stress Test 40.3% 20.7% 28.6% 24.2% Indication for PCI STEMI 10.5% 36.1% 27.6% 27.6% NSTEMI 15.1% 20.8% 20.6% 21.9% Stable angina 37.5% 23.5% 28.1% 27.8% Unstable Angina 18.0% 9.8% 11.3% 11.4% Other 18.9% 9.8% 12.4% 11.3% 0.034
8 8 Propensity Analysis Cohort Procedure Characteristics Measure DES N=13257 Before Match BMS N=2056 After Match (weighted for match ratio) DES N=8308 BMS N=1718 Standardized Difference Procedure Characteristics Number of treated lesions (per subject) Number of treated vessels (per subject) Treated vessels Native coronary 96.4% 96.5% 96.4% 96.3% Left main 1.0% 0.3% 0.8% 0.3% LAD 40.6% 30.4% 33.2% 31.8% RCA 31.7% 45.3% 39.2% 42.9% Circumflex 23.1% 20.6% 23.3% 21.3% Venous graft 2.9% 3.5% 3.1% 3.7% Arterial graft 0.7% 0.0% 0.6% 0.1% Modified ACC/AHA lesion class B2 or C 47.0% 50.6% 48.7% 48.7% Number of stents (per subject) Minimum stent diameter (per subject) <3 48.9% 25.5% 30.3% 28.9% =3 30.1% 32.2% 32.0% 32.7% >3 21.0% 42.3% 37.7% 38.5% Total stent length (mm, per subject) Prasugrel at discharge 38.7% 12.8% 16.6% 15.0% Clopidogrel at discharge 61.3% 87.2% 83.4% 85.0%
9 Propensity Analysis Cohort Baseline Stent Thrombosis Risk Factors 9 Before Match After Match (weighted for match ratio) Measure DES N=13257 BMS N=2056 DES N=8308 BMS N=1718 Standardized Difference Any Clinical 32.1% 62.4% 54.8% 55.7% Enzyme positive ACS (STEMI or NSTEMI) 25.6% 56.9% 48.2% 49.5% Renal insufficiency / failure 4.8% 4.0% 4.0% 3.9% LVEF < 30% 1.9% 4.2% 3.5% 3.5% Any Lesion-Related 33.3% 37.9% 33.7% 33.7% >2 vessels stented 0.5% 0.0% 0.1% 0.1% >2 lesions per vessel 2.3% 1.2% 1.4% 1.3% Lesion length 30 mm* 11.1% 6.6% 7.0% 6.7% Bifurcation lesion with sidebranch 2.5mm 6.0% 4.4% 4.9% 4.7% In-stent restenosis of a DES 4.5% 0.8% 1.1% 0.9% Vein bypass graft stented 3.4% 3.6% 3.4% 3.8% Unprotected left main stented 0.6% 0.1% 0.2% 0.2% Thrombus-containing lesion 10.5% 25.6% 20.1% 20.1% Prior brachytherapy 0.3% 0.1% 0.1% 0.1% Any Risk Factor 51.8% 69.3% 64.0% 63.9% 0.002
10 Drug-Eluting Stent Types Implanted at Index Procedure 10
11 Study Drug and Aspirin Compliance Post-Randomization Propensity-Matched DES + BMS Subjects 11
12 12 Primary Outcomes: Stent Thrombosis and MACCE, 0-33 Months Propensity-Matched DES + BMS Subjects Outcome DES N=8308 BMS N=1718 Weighted risk difference 1-sided 97.5% upper CL 1-sided P value non-inferiority P value for difference ST ARC definite / probable 138 (1.70%) 42 (2.61%) -1.05% -0.27% < ARC definite 115 (1.44%) 40 (2.48%) -1.08% 0.01 ARC probable 25 (0.28%) 2 (0.13%) 0.05% 0.56 MACCE (Death, MI, Stroke) 945 (11.37%) 227 (13.24%) -1.83% 0.03% < Death 349 (4.21%) 84 (5.05%) -0.83% 0.16 Cardiac 198 (2.43%) 47 (2.87%) -0.59% 0.19 Vascular 27 (0.32%) 4 (0.25%) 0.07% 0.64 Non-cardiovascular 124 (1.55%) 33 (2.04%) -0.30% 0.39 MI 570 (7.23%) 134 (8.11%) -0.80% 0.27 Stroke (total) 160 (1.83%) 34 (2.12%) -0.24% 0.49 Ischemic 125 (1.45%) 25 (1.56%) -0.13% 0.67 Hemorrhagic 35 (0.39%) 6 (0.38%) 0.05% 0.75 Uncertain type 6 (0.05%) 4 (0.25%) -0.17% 0.12
13 Cumulative Incidence of Stent Thrombosis* Stent Thrombosis Propensity-Matched DES + BMS Subjects 20% DES BMS 15% 10% 0-12 Months: RD -1.00% 0.70% vs. 1.68% P=0.002 (Difference) 0-33 Months: RD -1.05% 1.70% vs. 2.61% P<0.001 (Non-inferiority) P=0.01 (Difference) 5% 0% Months After Enrollment No. At Risk DES BMS *Weighted Kaplan-Meier and risk differences (RD) are presented. 13
14 Cumulative Incidence of Death, Myocardial Infarction or Stroke* MACCE Propensity-Matched DES + BMS Subjects 20% 15% DES BMS 0-12 Months: RD -1.76% 5.07% vs. 6.83% P=0.01 (Difference) 0-33 Months: RD -1.83% 11.37% vs % P<0.001 (Non-inferiority) P=0.053 (Difference) 10% 5% 0% Months After Enrollment No. At Risk DES BMS *Weighted Kaplan-Meier and risk differences (RD) are presented. 14
15 15 Primary Outcomes by DES Type (Propensity Matched Weighted RD vs. BMS), 0-33 Months Abbreviations: EES, everolimus-eluting stent; SES, sirolimus-eluting stent; PES, paclitaxel-eluting stent; ZES, zotarolimus-eluting stent (Endeavor).
16 Cumulative Incidence of Moderate or Severe Bleeding (GUSTO) Moderate or Severe Bleeding Propensity-Matched DES + BMS Subjects 5% 4% DES BMS Weighted RD 0.49% 4.04% vs. 3.67% P= % 2% 1% 0% Months After Procedure No. At Risk DES BMS *Weighted Kaplan-Meier and risk differences (RD) are presented. 16
17 Randomization and Follow-up BMS-Treated Subjects 17 1,687 BMS-Treated Randomized at 12 Months 13 Withdrew Consent 30 Lost to Follow-Up 3 Not Available for Follow-Up* 2 Withdrew Consent 10 Lost to Follow-Up 842 Randomized to Aspirin + Blinded Thienopyridine 796 (94.5%) Clinical Follow-up Available at 30 Months 784 (93.1%) Clinical Follow-up Available at 33 Months 845 Randomized to Aspirin + Blinded Placebo 20 Withdrew Consent 37 Lost to Follow-Up 4 Not Available for Follow-Up* 784 (92.8%) Clinical Follow-up Available at 30 Months 781 (92.4%) Clinical Follow-up Available at 33 Months 0 Withdrew Consent 3 Lost to Follow-Up *Subjects were prematurely exited from the study or incarcerated.
18 Baseline Characteristics Randomized BMS-Treated Subjects 18 Measure Thienopyridine N=842 Placebo N=845 Clinical Characteristics Age (years) Female 25.5% 21.8% 0.08 Race Non-White 7.5% 7.3% 0.93 Hispanic or Latino 4.8% 5.8% 0.44 Weight (kg) BMI Diabetes mellitus 21.7% 20.7% 0.63 Hypertension 64.0% 64.6% 0.80 Cigarette smoker (current or within past year) 44.3% 43.3% 0.69 Stroke / TIA 5.1% 4.0% 0.30 Congestive heart failure 4.2% 3.3% 0.37 Peripheral arterial disease 4.2% 5.5% 0.26 Prior PCI 17.9% 20.3% 0.22 Prior CABG 6.0% 5.9% 1.00 Prior MI 19.4% 21.5% 0.33 Positive Stress Test 19.0% 19.4% 0.85 Indication for PCI STEMI 36.9% 38.3% 0.58 NSTEMI 21.9% 20.0% 0.37 Unstable angina 9.1% 9.6% 0.80 Stable angina 23.6% 23.4% 0.95 Other 8.4% 8.6% 0.93 P value
19 Procedure Characteristics Randomized BMS-Treated Subjects 19 Measure Thienopyridine N=842 Placebo N=845 P value Procedure Characteristics Number of treated lesions (per subject) Number of treated vessels (per subject) Treated vessel(s) Native coronary 97.5% 97.5% 1.00 Left main 0.00% 0.1% 1.00 LAD 31.6% 30.9% 0.77 RCA 44.8% 45.6% 0.75 Circumflex 21.1% 20.9% 0.91 Venous graft 2.5% 2.5% 1.00 Arterial graft 0.0% 0.0% - Modified ACC/AHA lesion class B2 or C 47.6% 47.8% 0.93 Number of stents (per subject) Minimum stent diameter (per subject) 0.50 <3 23.9% 24.4% =3 31.5% 32.9% >3 44.7% 42.7% Total stent length (mm, per subject) Clopidogrel at discharge 87.2% 87.7% 0.77 Prasugrel at discharge 12.8% 12.3% 0.77
20 ST and MACCE, Months Randomized BMS-Treated Subjects 20 Outcome Stent Thrombosis ARC Definite/Probable Thienopyridine N=842 Placebo N=845 Stratified HR, 95% CI Stratified Log-rank P-Value 4 (0.5%) 9 (1.1%) 0.49 ( ) 0.24 ARC Definite 4 (0.5%) 9 (1.1%) 0.49 ( ) 0.24 ARC Probable 0 (0.0%) 0 (0.0%) -- (--, --) MACCE (Death, MI, Stroke) 33 (4.0%) 38 (4.7%) 0.92 ( ) 0.72 Death 8 (1.0%) 10 (1.2%) 0.90 ( ) 0.83 Cardiac 4 (0.5%) 5 (0.6%) 1.03 ( ) 0.97 Vascular 0 (0.0%) 0 (0.0%) -- (--, --) Non-Cardiovascular 4 (0.5%) 5 (0.6%) 0.79 ( ) 0.73 MI 22 (2.7%) 25 (3.1%) 0.91 ( ) 0.74 Stroke (total) 6 (0.7%) 5 (0.6%) 1.22 ( ) 0.74 Ischemic stroke 4 (0.5%) 5 (0.6%) 0.82 ( ) 0.77 Hemorrhagic stroke 1 (0.1%) 0 (0.0%) -- (--, --) 0.32 Type Uncertain 1 (0.1%) 0 (0.0%) -- (--, --) 0.32
21 Bleeding Outcomes, Months Randomized BMS-Treated Subjects 21 Bleeding Complications Thienopyridine N=790 Placebo N=776 Risk Difference 2-Sided P Value Difference GUSTO Severe/Moderate 16 (2.03%) 7 (0.90%) 1.12% 0.07 GUSTO Severe 6 (0.76%) 3 (0.39%) 0.37% 0.33 GUSTO Moderate 10 (1.27%) 4 (0.52%) 0.75% 0.12 BARC Types 2, 3, or 5 36 (4.56%) 14 (1.80%) 2.75% BARC Type 2 22 (2.78%) 7 (0.90%) 1.88% 0.01 BARC Type 3 16 (2.03%) 6 (0.77%) 1.25% 0.04 BARC Type 5 0 (0.00%) 1 (0.13%) -0.13% 0.31
22 Treatment Duration by Stent Type Interaction on Stent Thrombosis/MACCE 22 ARC Definite/Probable ST Stent Type 30 Month DAPT N (%) 12 Month DAPT N (%) HR (95% CI) DES (N=9961) 19 (0.4%) 65 (1.4%) 0.29 ( ) BMS (N=1687) 4 (0.5%) 9 (1.1%) 0.49 ( ) P Value Interaction 0.42 MACCE Stent Type 30 Month DAPT N (%) 12 Month DAPT N (%) HR (95% CI) DES (N=9961) 211 (4.3%) 285 (5.9%) 0.71 ( ) BMS (N=1687) 33 (4.0%) 38 (4.7%) 0.92 ( ) P Value Interaction 0.32
23 23 Conclusions DES vs BMS Propensity Matched Comparison DES not inferior to BMS for ST and MACCE over the 0-33 month follow-up period DES superior to BMS for ST over the 0-33 month followup period (for MACCE 0-12 month) Greatest portion of risk difference between stent platforms accrues within the first 12 months for both ST and MACCE
24 24 Conclusions (2) BMS 12 vs 30 months Randomized ITT Magnitude of reduction in ST risk with longer (30 months) duration thienopyridine therapy appears consistent for both BMS and DES, based on lack of interaction (P int=0.42) and hazard ratios < 1.0 (DES HR 0.29, BMS HR 0.49) In BMS treated patients, prolonged thienopyridine therapy (30 months or longer) may provide durable ischemic benefit in addition to increased bleeding risk, and requires further study.
25 ACS (STEMI / NSTEMI) subgroup analysis ST and MACCE BMS ITT; Months F/U Month DAPT N (%) 12 Month DAPT N (%) HR (95% CI) P Value for Interaction No ACS Within 72 Hours (N=699) ACS Within 72 Hours (N=988) ARC Definite/Probable ST 2 (0.6%) 1 (0.3%) 2.04 ( ) 2 (0.4%) 8 (1.7%) 0.24 ( ) 0.14 No ACS Within 72 Hours (N=699) ACS Within 72 Hours (N=988) MACCE 17 (5.0%) 17 (5.0%) 1.02 ( ) 16 (3.3%) 21 (4.5%) 0.74 ( ) 0.50
26 Dot Plot 26
Dual Antiplatelet Therapy Beyond One Year After Drug-eluting Coronary Stent Procedures
 Dual Antiplatelet Therapy Beyond One Year After Drug-eluting Coronary Stent Procedures Laura Mauri, Dean J. Kereiakes, Robert W. Yeh, Priscilla Driscoll-Shempp, Donald E. Cutlip, P. Gabriel Steg, Sharon-Lise
Dual Antiplatelet Therapy Beyond One Year After Drug-eluting Coronary Stent Procedures Laura Mauri, Dean J. Kereiakes, Robert W. Yeh, Priscilla Driscoll-Shempp, Donald E. Cutlip, P. Gabriel Steg, Sharon-Lise
Protocol. This trial protocol has been provided by the authors to give readers additional information about their work.
 Protocol This trial protocol has been provided by the authors to give readers additional information about their work. Protocol for: Mauri L, Kereiakes DJ, Yeh RW, et al. Twelve or 30 months of dual antiplatelet
Protocol This trial protocol has been provided by the authors to give readers additional information about their work. Protocol for: Mauri L, Kereiakes DJ, Yeh RW, et al. Twelve or 30 months of dual antiplatelet
Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G.
 Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G. Ellis MD Dean J. Kereiakes MD and Gregg W. Stone MD for the ABSORB
Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G. Ellis MD Dean J. Kereiakes MD and Gregg W. Stone MD for the ABSORB
Bern-Rotterdam Cohort Study
 Bern-Rotterdam Cohort Study Newer generation everolimus-eluting stents eliminate the risk of very late stent thrombosis compared with early generation sirolimus-eluting and paclitaxel-eluting stents Lorenz
Bern-Rotterdam Cohort Study Newer generation everolimus-eluting stents eliminate the risk of very late stent thrombosis compared with early generation sirolimus-eluting and paclitaxel-eluting stents Lorenz
Hyeon-Cheol Gwon, On the behalf of SMART-DATE trial investigators ACC LBCT 2018
 Six-month versus 12-month or longer dual antiplatelet therapy after percutaneous coronary intervention in patients with acute coronary syndromes (SMART-DATE): a randomized, openlabel, multicenter trial
Six-month versus 12-month or longer dual antiplatelet therapy after percutaneous coronary intervention in patients with acute coronary syndromes (SMART-DATE): a randomized, openlabel, multicenter trial
TRIAL UPDATE 1. ISAR TRIPLE SECURITY Trial. Dr Deven Patel Royal Free Hospital
 TRIAL UPDATE 1 ISAR TRIPLE SECURITY Trial Dr Deven Patel Royal Free Hospital NO CONFLICT OF INTEREST TO DECLARE ISAR TRIPLE Comparison of 6 weeks vs 6 months Triple Therapy in patients on oral anticoagulation
TRIAL UPDATE 1 ISAR TRIPLE SECURITY Trial Dr Deven Patel Royal Free Hospital NO CONFLICT OF INTEREST TO DECLARE ISAR TRIPLE Comparison of 6 weeks vs 6 months Triple Therapy in patients on oral anticoagulation
Supplementary Online Content
 Supplementary Online Content Valle JA, Tamez H, Abbott JD, et al. Contemporary use and trends in unprotected left main coronary artery percutaneous coronary intervention in the United States: an analysis
Supplementary Online Content Valle JA, Tamez H, Abbott JD, et al. Contemporary use and trends in unprotected left main coronary artery percutaneous coronary intervention in the United States: an analysis
Διάρκεια διπλής αντιαιμοπεταλιακής αγωγής. Νικόλαος Γ.Πατσουράκος Καρδιολόγος, Επιμελητής Α ΕΣΥ Τζάνειο Γενικό Νοσοκομείο Πειραιά
 Διάρκεια διπλής αντιαιμοπεταλιακής αγωγής Νικόλαος Γ.Πατσουράκος Καρδιολόγος, Επιμελητής Α ΕΣΥ Τζάνειο Γενικό Νοσοκομείο Πειραιά International ACS guidelines: Recommendations on duration of dual
Διάρκεια διπλής αντιαιμοπεταλιακής αγωγής Νικόλαος Γ.Πατσουράκος Καρδιολόγος, Επιμελητής Α ΕΣΥ Τζάνειο Γενικό Νοσοκομείο Πειραιά International ACS guidelines: Recommendations on duration of dual
Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis Alaide Chieffo
 Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis 11.45-12.07 Alaide Chieffo San Raffaele Scientific Institute, Milan, Italy Historical Perspective 25 20 15 10 5 0 Serruys 1991
Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis 11.45-12.07 Alaide Chieffo San Raffaele Scientific Institute, Milan, Italy Historical Perspective 25 20 15 10 5 0 Serruys 1991
EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017
 EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017 Igor F. Palacios, MD Director of Interventional Cardiology Professor of Medicine Massachusetts
EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017 Igor F. Palacios, MD Director of Interventional Cardiology Professor of Medicine Massachusetts
Which drug do you prefer for stable CAD? - P2Y12 inhibitor
 Which drug do you prefer for stable CAD? - P2Y12 inhibitor Jung Rae Cho, MD, PhD Cardiovascular Division, Department of Internal Medicine Kangnam Sacred Heart Hospital, Hallym University Medical Center,
Which drug do you prefer for stable CAD? - P2Y12 inhibitor Jung Rae Cho, MD, PhD Cardiovascular Division, Department of Internal Medicine Kangnam Sacred Heart Hospital, Hallym University Medical Center,
New Generation Drug- Eluting Stent in Korea
 New Generation Drug- Eluting Stent in Korea Young-Hak Kim, MD, PhD Department of Cardiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea Purpose To briefly introduce the
New Generation Drug- Eluting Stent in Korea Young-Hak Kim, MD, PhD Department of Cardiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea Purpose To briefly introduce the
Balancing Efficacy and Safety of P2Y12 Inhibitors for ACS Patients
 SYP.CLO-A.16.07.01 Balancing Efficacy and Safety of P2Y12 Inhibitors for ACS Patients dr. Hariadi Hariawan, Sp.PD, Sp.JP (K) TOPICS Efficacy Safety Consideration from Currently Available Antiplatelet Agents
SYP.CLO-A.16.07.01 Balancing Efficacy and Safety of P2Y12 Inhibitors for ACS Patients dr. Hariadi Hariawan, Sp.PD, Sp.JP (K) TOPICS Efficacy Safety Consideration from Currently Available Antiplatelet Agents
Dual Antiplatelet Therapy Made Practical
 Dual Antiplatelet Therapy Made Practical David Parra, Pharm.D., FCCP, BCPS Clinical Pharmacy Program Manager in Cardiology/Anticoagulation VISN 8 Pharmacy Benefits Management Clinical Associate Professor
Dual Antiplatelet Therapy Made Practical David Parra, Pharm.D., FCCP, BCPS Clinical Pharmacy Program Manager in Cardiology/Anticoagulation VISN 8 Pharmacy Benefits Management Clinical Associate Professor
PROlonging Dual antiplatelet treatment after Grading stent-induced Intimal hyperplasia study
 ESC, Hotline III, Paris, August, 30, 2011 PROlonging Dual antiplatelet treatment after Grading stent-induced Intimal hyperplasia study M. Valgimigli, MD, PhD University of Ferrara, ITALY On behalf of the
ESC, Hotline III, Paris, August, 30, 2011 PROlonging Dual antiplatelet treatment after Grading stent-induced Intimal hyperplasia study M. Valgimigli, MD, PhD University of Ferrara, ITALY On behalf of the
Optimal Duration and Dose of Antiplatelet Therapy after PCI
 Optimal Duration and Dose of Antiplatelet Therapy after PCI Donghoon Choi, MD, PhD Severance Cardiovascular Center Yonsei University College of Medicine Optimal Duration of Antiplatelet Therapy after PCI
Optimal Duration and Dose of Antiplatelet Therapy after PCI Donghoon Choi, MD, PhD Severance Cardiovascular Center Yonsei University College of Medicine Optimal Duration of Antiplatelet Therapy after PCI
Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial
 Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial Myeong-Ki Hong, MD. PhD on behalf of the IVUS-XPL trial investigators
Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial Myeong-Ki Hong, MD. PhD on behalf of the IVUS-XPL trial investigators
Supplementary Material to Mayer et al. A comparative cohort study on personalised
 Suppl. Table : Baseline characteristics of the patients. Characteristic Modified cohort Non-modified cohort P value (n=00) Age years 68. ±. 69.5 ±. 0. Female sex no. (%) 60 (0.0) 88 (.7) 0.0 Body Mass
Suppl. Table : Baseline characteristics of the patients. Characteristic Modified cohort Non-modified cohort P value (n=00) Age years 68. ±. 69.5 ±. 0. Female sex no. (%) 60 (0.0) 88 (.7) 0.0 Body Mass
Clinical Study Age Differences in Long Term Outcomes of Coronary Patients Treated with Drug Eluting Stents at a Tertiary Medical Center
 Aging Research Volume 2013, Article ID 471026, 4 pages http://dx.doi.org/10.1155/2013/471026 Clinical Study Age Differences in Long Term Outcomes of Coronary Patients Treated with Drug Eluting Stents at
Aging Research Volume 2013, Article ID 471026, 4 pages http://dx.doi.org/10.1155/2013/471026 Clinical Study Age Differences in Long Term Outcomes of Coronary Patients Treated with Drug Eluting Stents at
The MAIN-COMPARE Study
 Long-Term Outcomes of Coronary Stent Implantation versus Bypass Surgery for the Treatment of Unprotected Left Main Coronary Artery Disease Revascularization for Unprotected Left MAIN Coronary Artery Stenosis:
Long-Term Outcomes of Coronary Stent Implantation versus Bypass Surgery for the Treatment of Unprotected Left Main Coronary Artery Disease Revascularization for Unprotected Left MAIN Coronary Artery Stenosis:
What oral antiplatelet therapy would you choose? a) ASA alone b) ASA + Clopidogrel c) ASA + Prasugrel d) ASA + Ticagrelor
 76 year old female Prior Hypertension, Hyperlipidemia, Smoking On Hydrochlorothiazide, Atorvastatin New onset chest discomfort; 2 episodes in past 24 hours Heart rate 122/min; BP 170/92 mm Hg, Killip Class
76 year old female Prior Hypertension, Hyperlipidemia, Smoking On Hydrochlorothiazide, Atorvastatin New onset chest discomfort; 2 episodes in past 24 hours Heart rate 122/min; BP 170/92 mm Hg, Killip Class
Are Asian Patients Different? - Updates Of Biomatrix Experience In Regional Settings: BEACON II (3 Yr F up) &
 Are Asian Patients Different? - Updates Of Biomatrix Experience In Regional Settings: BEACON II (3 Yr F up) & Biomatrix TM Single Center Experience (Indonesia)(Final 5 Yr F up) T. Santoso University of
Are Asian Patients Different? - Updates Of Biomatrix Experience In Regional Settings: BEACON II (3 Yr F up) & Biomatrix TM Single Center Experience (Indonesia)(Final 5 Yr F up) T. Santoso University of
PROMUS Element Experience In AMC
 Promus Element Luncheon Symposium: PROMUS Element Experience In AMC Jung-Min Ahn, MD. University of Ulsan College of Medicine, Heart Institute, Asan Medical Center, Seoul, Korea PROMUS Element Clinical
Promus Element Luncheon Symposium: PROMUS Element Experience In AMC Jung-Min Ahn, MD. University of Ulsan College of Medicine, Heart Institute, Asan Medical Center, Seoul, Korea PROMUS Element Clinical
Komplexe Koronarintervention heute: Von Syntax zu bioresorbierbaren Stents
 Komplexe Koronarintervention heute: Von Syntax zu bioresorbierbaren Stents Prof. Dr. med. Julinda Mehilli Medizinische Klinik und Poliklinik I Klinikum der Universität München Campus Großhadern Key Factors
Komplexe Koronarintervention heute: Von Syntax zu bioresorbierbaren Stents Prof. Dr. med. Julinda Mehilli Medizinische Klinik und Poliklinik I Klinikum der Universität München Campus Großhadern Key Factors
SKG Congress, 2015 EVOLVE II. Stephan Windecker
 SKG Congress, 2015 EVOLVE II Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland BIODEGRADABLE POLYMER DES Stefanini,
SKG Congress, 2015 EVOLVE II Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland BIODEGRADABLE POLYMER DES Stefanini,
BIOFLOW V Comparison of UltraThin Sirolimus-Eluting Bioresorbable Polymer with Thin Everolimus- Eluting Durable Polymer Stents
 Comparison of UltraThin Sirolimus-Eluting Bioresorbable Polymer with Thin Everolimus- Eluting Durable Polymer Stents David E Kandzari, MD; Laura Mauri, MD, MSc; Jacques Koolen, MD, PhD; Joseph M Massaro,
Comparison of UltraThin Sirolimus-Eluting Bioresorbable Polymer with Thin Everolimus- Eluting Durable Polymer Stents David E Kandzari, MD; Laura Mauri, MD, MSc; Jacques Koolen, MD, PhD; Joseph M Massaro,
GRAND ROUNDS - DILEMMAS IN ANTICOAGULATION AND ANTIPLATELET THERAPY. Nick Collins February 2017
 GRAND ROUNDS - DILEMMAS IN ANTICOAGULATION AND ANTIPLATELET THERAPY Nick Collins February 2017 DISCLOSURES Before I commence Acknowledge.. Interventional Cardiologist Perception evolved. Interventional
GRAND ROUNDS - DILEMMAS IN ANTICOAGULATION AND ANTIPLATELET THERAPY Nick Collins February 2017 DISCLOSURES Before I commence Acknowledge.. Interventional Cardiologist Perception evolved. Interventional
Optimal lenght of DAPT in different clinical scenarios
 Optimal lenght of DAPT in different clinical scenarios After PCI with DES in the light of recent and ongoing studies Dr Grégoire Rangé / CH Chartres / France DAPT duration depend on the evolution of risk
Optimal lenght of DAPT in different clinical scenarios After PCI with DES in the light of recent and ongoing studies Dr Grégoire Rangé / CH Chartres / France DAPT duration depend on the evolution of risk
Learning Objectives. Epidemiology of Acute Coronary Syndrome
 Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions
 Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
3/23/2017. Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate Europace Oct;14(10): Epub 2012 Aug 24.
 Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Controversies in Cardiac Pharmacology
 Controversies in Cardiac Pharmacology Thomas D. Conley, MD FACC FSCAI Disclosures I have no relevant relationships with commercial interests to disclose. 1 Doc, do I really need to take all these medicines?
Controversies in Cardiac Pharmacology Thomas D. Conley, MD FACC FSCAI Disclosures I have no relevant relationships with commercial interests to disclose. 1 Doc, do I really need to take all these medicines?
Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017
 Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Nine-year clinical outcomes of drug-eluting stents vs. bare metal stents for large coronary vessel lesions
 Journal of Geriatric Cardiology (2017) 14: 35 41 2017 JGC All rights reserved; www.jgc301.com Research Article Open Access Nine-year clinical outcomes of drug-eluting stents vs. bare metal stents for large
Journal of Geriatric Cardiology (2017) 14: 35 41 2017 JGC All rights reserved; www.jgc301.com Research Article Open Access Nine-year clinical outcomes of drug-eluting stents vs. bare metal stents for large
Triple Therapy: A review of the evidence in acute coronary syndrome. Stephanie Kling, PharmD, BCPS Sanford Health
 Triple Therapy: A review of the evidence in acute coronary syndrome Stephanie Kling, PharmD, BCPS Sanford Health Objectives 1. Describe how the presented topic impacts patient outcomes. 2. Review evidence
Triple Therapy: A review of the evidence in acute coronary syndrome Stephanie Kling, PharmD, BCPS Sanford Health Objectives 1. Describe how the presented topic impacts patient outcomes. 2. Review evidence
TCT mdbuyline.com Clinical Trial Results Summary
 TCT 2012 Clinical Trial Results Summary FAME2 Trial: FFR (fractional flow reserve) guided PCI in all target lesions Patients with significant ischemia, randomized 1:1 Control arm: not hemodynamically significant
TCT 2012 Clinical Trial Results Summary FAME2 Trial: FFR (fractional flow reserve) guided PCI in all target lesions Patients with significant ischemia, randomized 1:1 Control arm: not hemodynamically significant
Ian T. Meredith AM. MBBS, PhD, FRACP, FCSANZ, FACC, FSCAI, FAPSIC MonashHeart, Monash Medical Centre & Monash University Melbourne, Australia
 Final five-year clinical outcomes in the EVOLVE trial: A randomised evaluation of a novel bioabsorbable polymer-coated, everolimus-eluting stent Ian T. Meredith AM MBBS, PhD, FRACP, FCSANZ, FACC, FSCAI,
Final five-year clinical outcomes in the EVOLVE trial: A randomised evaluation of a novel bioabsorbable polymer-coated, everolimus-eluting stent Ian T. Meredith AM MBBS, PhD, FRACP, FCSANZ, FACC, FSCAI,
Mid-term results from real-world REPARA registry. Felipe Hernandez, on behalf of the REPARA investigators
 Mid-term results from real-world REPARA registry Felipe Hernandez, on behalf of the REPARA investigators Potential conflicts of interest Speaker's name: Felipe Hernandez I have the following potential
Mid-term results from real-world REPARA registry Felipe Hernandez, on behalf of the REPARA investigators Potential conflicts of interest Speaker's name: Felipe Hernandez I have the following potential
A Randomized Trial Evaluating Clinically Significant Bleeding with Low-Dose Rivaroxaban vs Aspirin, in Addition to P2Y12 inhibition, in ACS
 A Randomized Trial Evaluating Clinically Significant Bleeding with Low-Dose Rivaroxaban vs Aspirin, in Addition to P2Y12 inhibition, in ACS Magnus Ohman MB, on behalf of the GEMINI-ACS-1 Investigators
A Randomized Trial Evaluating Clinically Significant Bleeding with Low-Dose Rivaroxaban vs Aspirin, in Addition to P2Y12 inhibition, in ACS Magnus Ohman MB, on behalf of the GEMINI-ACS-1 Investigators
Optimal Duration of Clopidogrel Therapy with DES to Reduce Late Coronary Arterial Thrombotic Event. The DES LATE Trial
 Optimal Duration of Clopidogrel Therapy with DES to Reduce Late Coronary Arterial Thrombotic Event The DES LATE Trial Cheol Whan Lee, MD, Seung-Jung Park, MD, PhD, On Behalf of the DES LATE Investigators
Optimal Duration of Clopidogrel Therapy with DES to Reduce Late Coronary Arterial Thrombotic Event The DES LATE Trial Cheol Whan Lee, MD, Seung-Jung Park, MD, PhD, On Behalf of the DES LATE Investigators
What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation?
 What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation? Keun-Ho Park, Myung Ho Jeong, Min Goo Lee, Jum Suk Ko,
What is the Optimal Triple Anti-platelet Therapy Duration in Patients with Acute Myocardial Infarction Undergoing Drug-eluting Stents Implantation? Keun-Ho Park, Myung Ho Jeong, Min Goo Lee, Jum Suk Ko,
DAPT in CAD, Acute & Chronic CAD, antiplatelet therapy non-responders
 DAPT in CAD, Acute & Chronic CAD, antiplatelet therapy non-responders Annual Ohio ACC Conference October 14, 2017 Ernest L. Mazzaferri Jr, MD, FACC, FSCAI Disclosures No financial disclosures related to
DAPT in CAD, Acute & Chronic CAD, antiplatelet therapy non-responders Annual Ohio ACC Conference October 14, 2017 Ernest L. Mazzaferri Jr, MD, FACC, FSCAI Disclosures No financial disclosures related to
Diabetic Patients: Current Evidence of Revascularization
 Diabetic Patients: Current Evidence of Revascularization Alexandra J. Lansky, MD Yale University School of Medicine University College of London The Problem with Diabetic Patients Endothelial dysfunction
Diabetic Patients: Current Evidence of Revascularization Alexandra J. Lansky, MD Yale University School of Medicine University College of London The Problem with Diabetic Patients Endothelial dysfunction
Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease (BEST Trial)
 Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease (BEST Trial) Seung-Jung Park, MD, PhD On behalf of the BEST investigators Professor of Medicine, University of Ulsan College of
Trial of Everolimus-Eluting Stents or Bypass Surgery for Coronary Disease (BEST Trial) Seung-Jung Park, MD, PhD On behalf of the BEST investigators Professor of Medicine, University of Ulsan College of
Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland
 Advances in Antiplatelet Therapy in PCI and ACS Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland Targets for Platelet
Advances in Antiplatelet Therapy in PCI and ACS Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland Targets for Platelet
A patient with an acute coronary syndrome one year ago. Options for antiplatelet treatment
 A patient with an acute coronary syndrome one year ago. Options for antiplatelet treatment Ronen Durst, MD Cardiology Department Hadassah, Hebrew University Medical Center. Chairman, Israeli society for
A patient with an acute coronary syndrome one year ago. Options for antiplatelet treatment Ronen Durst, MD Cardiology Department Hadassah, Hebrew University Medical Center. Chairman, Israeli society for
Biolimus-Eluting Stent With Biodegradable Polymer Versus Sirolimus-Eluting Stent With Durable Polymer: A Randomised, Non-Inferiority Trial
 Limus Eluted From A Durable vs ERodable Stent Coating Biolimus-Eluting Stent With Biodegradable Polymer Versus Sirolimus-Eluting Stent With Durable Polymer: A Randomised, Non-Inferiority Trial Stephan
Limus Eluted From A Durable vs ERodable Stent Coating Biolimus-Eluting Stent With Biodegradable Polymer Versus Sirolimus-Eluting Stent With Durable Polymer: A Randomised, Non-Inferiority Trial Stephan
DECLARATION OF CONFLICT OF INTEREST. Lecture fees: AstraZeneca, Ely Lilly, Merck.
 DECLARATION OF CONFLICT OF INTEREST Lecture fees: AstraZeneca, Ely Lilly, Merck. Risk of stopping dual therapy. S D Kristensen, FESC Aarhus Denmark Acute coronary syndrome: coronary thrombus Platelets
DECLARATION OF CONFLICT OF INTEREST Lecture fees: AstraZeneca, Ely Lilly, Merck. Risk of stopping dual therapy. S D Kristensen, FESC Aarhus Denmark Acute coronary syndrome: coronary thrombus Platelets
Antiplatelet Therapy After PCI: How Much and How Long?
 Antiplatelet Therapy After PCI: How Much and How Long? Adnan Kastrati Deutsches Herzzentrum, Munich, GERMANY Antiplatelet Therapy after PCI: How much and how long? The simplest and extreme response: Give
Antiplatelet Therapy After PCI: How Much and How Long? Adnan Kastrati Deutsches Herzzentrum, Munich, GERMANY Antiplatelet Therapy after PCI: How much and how long? The simplest and extreme response: Give
TCTAP Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI
 Indian TUXEDO Trial In Medically Treated Diabetics Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI Executive Director and Dean Escorts Heart Institute & Medical Research Center and Fortis Hospitals, New Delhi
Indian TUXEDO Trial In Medically Treated Diabetics Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI Executive Director and Dean Escorts Heart Institute & Medical Research Center and Fortis Hospitals, New Delhi
Prasugrel vs. Ticagrelor in ACS/PCI Which one to choose? V. Voudris MD FESC FACC 2 nd Cardiology Division Onassis Cardiac Surgery Center
 Prasugrel vs. Ticagrelor in ACS/PCI Which one to choose? V. Voudris MD FESC FACC 2 nd Cardiology Division Onassis Cardiac Surgery Center Hospitalizations in the U.S. Due to ACS Acute Coronary Syndromes
Prasugrel vs. Ticagrelor in ACS/PCI Which one to choose? V. Voudris MD FESC FACC 2 nd Cardiology Division Onassis Cardiac Surgery Center Hospitalizations in the U.S. Due to ACS Acute Coronary Syndromes
Lessons learned From The National PCI Registry
 Lessons learned From The National PCI Registry w a v e On Behalf of The Publication Committee of the National PCI Registry Objectives & Anticipated Achievements To determine the epidemiology of patients
Lessons learned From The National PCI Registry w a v e On Behalf of The Publication Committee of the National PCI Registry Objectives & Anticipated Achievements To determine the epidemiology of patients
David E. Kandzari, MD Director, Interventional Cardiology Research Scripps Clinic La Jolla, California
 38 RCTs 18,000 pts Identifying the Optimal Duration of DAPT Less is More, More or Less David E. Kandzari, MD Director, Interventional Cardiology Research Scripps Clinic La Jolla, California kandzari.david@scrippshealth.org
38 RCTs 18,000 pts Identifying the Optimal Duration of DAPT Less is More, More or Less David E. Kandzari, MD Director, Interventional Cardiology Research Scripps Clinic La Jolla, California kandzari.david@scrippshealth.org
Coronary Stent Choice in Patients With Diabetes Mellitus
 Rome Cardiology Forum 2014 Coronary Stent Choice in Patients With Diabetes Mellitus Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University
Rome Cardiology Forum 2014 Coronary Stent Choice in Patients With Diabetes Mellitus Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University
ANGIOPLASY SUMMIT 2007 TCT ASIA PACIFIC. Seoul, Korea: April The problem is exaggerated: Data from Real World Registries
 ANGIOPLASY SUMMIT 007 TCT ASIA PACIFIC Seoul, Korea: 5-7 7 April 007 15 min Plenary Session: State-of of-the-art Lectures The problem is exaggerated: Data from Real World Registries Antonio Colombo Centro
ANGIOPLASY SUMMIT 007 TCT ASIA PACIFIC Seoul, Korea: 5-7 7 April 007 15 min Plenary Session: State-of of-the-art Lectures The problem is exaggerated: Data from Real World Registries Antonio Colombo Centro
Supplementary Table S1: Proportion of missing values presents in the original dataset
 Supplementary Table S1: Proportion of missing values presents in the original dataset Variable Included (%) Missing (%) Age 89067 (100.0) 0 (0.0) Gender 89067 (100.0) 0 (0.0) Smoking status 80706 (90.6)
Supplementary Table S1: Proportion of missing values presents in the original dataset Variable Included (%) Missing (%) Age 89067 (100.0) 0 (0.0) Gender 89067 (100.0) 0 (0.0) Smoking status 80706 (90.6)
Run-Lin Gao, MD, FACC. On Behalf of I-LOVE-IT Trial Investigators
 I-LOVE-IT A Prospective, Multicenter Clinical Trial of TIVOLI Bioabsorbable Polymer Based Sirolimus-Eluting vs. ENDEAVOR Zotarolimus- Eluting Stent in Patients with Coronary Artery Disease: 8-Month Angiographic
I-LOVE-IT A Prospective, Multicenter Clinical Trial of TIVOLI Bioabsorbable Polymer Based Sirolimus-Eluting vs. ENDEAVOR Zotarolimus- Eluting Stent in Patients with Coronary Artery Disease: 8-Month Angiographic
eluting Stents The SPIRIT Trials
 Everolimus-eluting eluting Stents The SPIRIT Trials Gregg W. Stone, MD Columbia University Medical Center Cardiovascular Research Foundation Abbott XIENCE V Everolimus-eluting eluting Stent Everolimus
Everolimus-eluting eluting Stents The SPIRIT Trials Gregg W. Stone, MD Columbia University Medical Center Cardiovascular Research Foundation Abbott XIENCE V Everolimus-eluting eluting Stent Everolimus
OUTPATIENT ANTITHROMBOTIC MANAGEMENT POST NON-ST ELEVATION ACUTE CORONARY SYNDROME. TARGET AUDIENCE: All Canadian health care professionals.
 OUTPATIENT ANTITHROMBOTIC MANAGEMENT POST NON-ST ELEVATION ACUTE CORONARY SYNDROME TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To review the use of antiplatelet agents and oral
OUTPATIENT ANTITHROMBOTIC MANAGEMENT POST NON-ST ELEVATION ACUTE CORONARY SYNDROME TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To review the use of antiplatelet agents and oral
A Polymer-Free Dual Drug-Eluting Stent in Patients with Coronary Artery Disease: Randomized Trial Versus Polymer-Based DES.
 A Polymer-Free Dual Drug-Eluting Stent in Patients with Coronary Artery Disease: Randomized Trial Versus Polymer-Based DES ISAR-TEST 2 Trial Robert A. Byrne, MB MRCPI Deutsches Herzzentrum and 1. Med.
A Polymer-Free Dual Drug-Eluting Stent in Patients with Coronary Artery Disease: Randomized Trial Versus Polymer-Based DES ISAR-TEST 2 Trial Robert A. Byrne, MB MRCPI Deutsches Herzzentrum and 1. Med.
The MAIN-COMPARE Registry
 Long-Term Outcomes of Coronary Stent Implantation versus Bypass Surgery for the Treatment of Unprotected Left Main Coronary Artery Disease Revascularization for Unprotected Left MAIN Coronary Artery Stenosis:
Long-Term Outcomes of Coronary Stent Implantation versus Bypass Surgery for the Treatment of Unprotected Left Main Coronary Artery Disease Revascularization for Unprotected Left MAIN Coronary Artery Stenosis:
MINERVA MEDICA COPYRIGHT. Aspirin is accepted as standard antiplatelet. Dual antiplatelet therapy for primary and secondary prevention
 This document is protected by international copyright laws. No additional reproduction is authorized. It is permitted for personal use to download and save only one file and print only one copy of this
This document is protected by international copyright laws. No additional reproduction is authorized. It is permitted for personal use to download and save only one file and print only one copy of this
Antiplatelet and Anti-Thrombotic Therapy. Ivan Anderson, MD RIHVH Cardiology
 Antiplatelet and Anti-Thrombotic Therapy Ivan Anderson, MD RIHVH Cardiology Outline Anti-thrombotic therapy Risk stratification of stroke with atrial fibrillation DVT and PE treatment Pharmacology Anti-platelet
Antiplatelet and Anti-Thrombotic Therapy Ivan Anderson, MD RIHVH Cardiology Outline Anti-thrombotic therapy Risk stratification of stroke with atrial fibrillation DVT and PE treatment Pharmacology Anti-platelet
The SORT OUT VI Trial
 A Prospective, Randomized, "All-Comers" Trial of Biodegradable Polymer-Coated Biolimus-Eluting Stents vs. Biocompatible Polymer-Coated Zotarolimus-Eluting Stents The SORT OUT VI Trial Bent Raungaard, Lisette
A Prospective, Randomized, "All-Comers" Trial of Biodegradable Polymer-Coated Biolimus-Eluting Stents vs. Biocompatible Polymer-Coated Zotarolimus-Eluting Stents The SORT OUT VI Trial Bent Raungaard, Lisette
Asian AMI Registry Session The 17 th Joint Meeting of Coronary Revascularization (JCR 2017) Busan, Korea Dec 8 th 2017
 Asian AMI Registry Session The 17 th Joint Meeting of Coronary Revascularization (JCR 2017) Busan, Korea Dec 8 th 2017 Trends of acute myocardial infarction in Korea from the experience of Korea Acute
Asian AMI Registry Session The 17 th Joint Meeting of Coronary Revascularization (JCR 2017) Busan, Korea Dec 8 th 2017 Trends of acute myocardial infarction in Korea from the experience of Korea Acute
Ticagrelor compared with clopidogrel in patients with acute coronary syndromes the PLATO trial
 compared with clopidogrel in patients with acute coronary syndromes the PLATO trial August 30, 2009 at 08.00 CET PLATO background In NSTE-ACS and STEMI, current guidelines recommend 12 months aspirin and
compared with clopidogrel in patients with acute coronary syndromes the PLATO trial August 30, 2009 at 08.00 CET PLATO background In NSTE-ACS and STEMI, current guidelines recommend 12 months aspirin and
Two-Year Outcomes of High Bleeding Risk Patients after Polymer-Free Drug-Coated Stents
 Two-Year Outcomes of High Bleeding Risk Patients after Polymer-Free Drug-Coated Stents Philip Urban, Philippe Garot, Damras Tresukosol, Stuart J. Pocock, Ian Meredith, Alex Abizaid, Didier Carrié, Christoph
Two-Year Outcomes of High Bleeding Risk Patients after Polymer-Free Drug-Coated Stents Philip Urban, Philippe Garot, Damras Tresukosol, Stuart J. Pocock, Ian Meredith, Alex Abizaid, Didier Carrié, Christoph
Timing of Surgery After Percutaneous Coronary Intervention
 Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Conflict of interest :None. Meta-analysis. Zhangwei Chen, MD
 Meta-analysis Addition of Cilostazol to Conventional Dual Antiplatelet Therapy Reduces the Risk of Cardiac Events and Restenosis after Drug-Eluting Stent Implantation Zhangwei Chen, MD Department of Cardiology,
Meta-analysis Addition of Cilostazol to Conventional Dual Antiplatelet Therapy Reduces the Risk of Cardiac Events and Restenosis after Drug-Eluting Stent Implantation Zhangwei Chen, MD Department of Cardiology,
Polymer-Free Stent CX - ISAR
 Polymer-Free Stent CX - ISAR Moo Hyun Kim, MD, FACC on behalf of Dr. Florian Krackhardt, Germany CX ISAR Stent : Features Intracoronary Stenting and Angiographic Results Strut Thickness of only 50/60 μm
Polymer-Free Stent CX - ISAR Moo Hyun Kim, MD, FACC on behalf of Dr. Florian Krackhardt, Germany CX ISAR Stent : Features Intracoronary Stenting and Angiographic Results Strut Thickness of only 50/60 μm
Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction
 Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction The Bavarian Reperfusion Alternatives Evaluation (BRAVE) 4
Randomized Comparison of Prasugrel and Bivalirudin versus Clopidogrel and Heparin in Patients with ST-Segment Elevation Myocardial Infarction The Bavarian Reperfusion Alternatives Evaluation (BRAVE) 4
Unprotected LM intervention
 Unprotected LM intervention Guideline for COMBAT Seung-Jung Park, MD, PhD Professor of Internal Medicine, Seoul, Korea Current Recommendation for unprotected LMCA Stenosis Class IIb C in ESC guideline
Unprotected LM intervention Guideline for COMBAT Seung-Jung Park, MD, PhD Professor of Internal Medicine, Seoul, Korea Current Recommendation for unprotected LMCA Stenosis Class IIb C in ESC guideline
Alex versus Xience Registry Preliminary report
 Interventional Cardiology Network Alex versus Xience Preliminary report Mariusz Gąsior 1,2, Marek Gierlotka 1, Lech Poloński 1,2 1 3rd Department of Cardiology, Medical University of Silesia Centre tor
Interventional Cardiology Network Alex versus Xience Preliminary report Mariusz Gąsior 1,2, Marek Gierlotka 1, Lech Poloński 1,2 1 3rd Department of Cardiology, Medical University of Silesia Centre tor
Clinical and Economic Value of Rivaroxaban in Coronary Artery Disease
 CHRISTOPHER B. GRANGER, MD Professor of Medicine Division of Cardiology, Department of Medicine; Director, Cardiac Care Unit Duke University Medical Center, Durham, NC Clinical and Economic Value of Rivaroxaban
CHRISTOPHER B. GRANGER, MD Professor of Medicine Division of Cardiology, Department of Medicine; Director, Cardiac Care Unit Duke University Medical Center, Durham, NC Clinical and Economic Value of Rivaroxaban
Osler Journal Club Outcomes Research
 Osler Journal Club Outcomes Research Malenka DJ, et al. Outcomes Following Coronary Stenting in the Era of Bare-Metal vs. the Era of Drug- Eluting Stents. JAMA 2008; 299(24):2868-2876 Mentor: Dr. Boulware
Osler Journal Club Outcomes Research Malenka DJ, et al. Outcomes Following Coronary Stenting in the Era of Bare-Metal vs. the Era of Drug- Eluting Stents. JAMA 2008; 299(24):2868-2876 Mentor: Dr. Boulware
Transient Atrial Fibrillation and Risk of Stroke after Acute Myocardial Infarction
 Transient Atrial Fibrillation and Risk of Stroke after Acute Myocardial Infarction Doron Aronson MD, Gregory Telman MD, Fadel BahouthMD, Jonathan Lessick MD, DSc and Rema Bishara MD Department of Cardiology
Transient Atrial Fibrillation and Risk of Stroke after Acute Myocardial Infarction Doron Aronson MD, Gregory Telman MD, Fadel BahouthMD, Jonathan Lessick MD, DSc and Rema Bishara MD Department of Cardiology
7-Year Follow-up From a Randomized Trial of Sirolimus-Eluting vs. Everolimus-Eluting Stents in Patients With Coronary Artery Disease (RESET)
 7-Year Follow-up From a Randomized Trial of Sirolimus-Eluting vs. Everolimus-Eluting Stents in Patients With Coronary Artery Disease (RESET) Hiroki Shiomi, MD Kyoto University Graduate School of Medicine
7-Year Follow-up From a Randomized Trial of Sirolimus-Eluting vs. Everolimus-Eluting Stents in Patients With Coronary Artery Disease (RESET) Hiroki Shiomi, MD Kyoto University Graduate School of Medicine
From STEMIs to Stents: Updates in PCI practice
 From STEMIs to Stents: Updates in PCI practice Arnold Seto, MD, MPA Assistant Clinical Professor, UC-Irvine and Long Beach VA Director of Interventional Cardiology Research Hospitalizations in the U.S.
From STEMIs to Stents: Updates in PCI practice Arnold Seto, MD, MPA Assistant Clinical Professor, UC-Irvine and Long Beach VA Director of Interventional Cardiology Research Hospitalizations in the U.S.
Antiplatelet therapy in myocardial infarction and coronary stent thrombosis Heestermans, Antonius Adrianus Cornelius Maria
 University of Groningen Antiplatelet therapy in myocardial infarction and coronary stent thrombosis Heestermans, Antonius Adrianus Cornelius Maria IMPORTANT NOTE: You are advised to consult the publisher's
University of Groningen Antiplatelet therapy in myocardial infarction and coronary stent thrombosis Heestermans, Antonius Adrianus Cornelius Maria IMPORTANT NOTE: You are advised to consult the publisher's
Low Dose Rivaroxaban Versus Aspirin, in Addition to P2Y12 Inhibition, in Acute Coronary Syndromes (GEMINI-ACS-1)
 Low Dose Rivaroxaban Versus Aspirin, in Addition to P2Y12 Inhibition, in Acute Coronary Syndromes (GEMINI-ACS-1) Caitlin C. Akerman, PharmD PGY2 Cardiology Resident WakeMed Health & Hospitals Raleigh,
Low Dose Rivaroxaban Versus Aspirin, in Addition to P2Y12 Inhibition, in Acute Coronary Syndromes (GEMINI-ACS-1) Caitlin C. Akerman, PharmD PGY2 Cardiology Resident WakeMed Health & Hospitals Raleigh,
Surgical vs. Percutaneous Revascularization in Patients with Diabetes and Acute Coronary Syndrome
 Surgical vs. Percutaneous Revascularization in Patients with Diabetes and Acute Coronary Syndrome Chris C. Cook, MD Associate Professor of Surgery Director, CT Residency Program, WVU ACOI 10/17/18 No Disclosures
Surgical vs. Percutaneous Revascularization in Patients with Diabetes and Acute Coronary Syndrome Chris C. Cook, MD Associate Professor of Surgery Director, CT Residency Program, WVU ACOI 10/17/18 No Disclosures
Resolute in Bifurcation Lesions: Data from the RESOLUTE Clinical Program
 Resolute in Bifurcation Lesions: Data from the RESOLUTE Clinical Program Prof. Ran Kornowski, MD, FESC, FACC Director - Division of Interventional Cardiology Rabin Medical Center and Tel Aviv University,
Resolute in Bifurcation Lesions: Data from the RESOLUTE Clinical Program Prof. Ran Kornowski, MD, FESC, FACC Director - Division of Interventional Cardiology Rabin Medical Center and Tel Aviv University,
TRIAS HR Pilot Study
 Late Breaking Clinical Trials TCT, October 22 nd 27, Washington, USA TRIAS HR Pilot Study RCT comparing Genous EPC capturing stent with Taxus Paclitaxel eluting stent Robbert J de Winter MD PhD FESC Academic
Late Breaking Clinical Trials TCT, October 22 nd 27, Washington, USA TRIAS HR Pilot Study RCT comparing Genous EPC capturing stent with Taxus Paclitaxel eluting stent Robbert J de Winter MD PhD FESC Academic
The Dual Antiplatelet Therapy (DAPT) Trial: An FDA Perspective
 The Dual Antiplatelet Therapy (DAPT) Trial: An FDA Perspective Bram Zuckerman, MD, FACC Director, FDA Division of Cardiovascular Devices Office of Device Evaluation Center for Devices and Radiological
The Dual Antiplatelet Therapy (DAPT) Trial: An FDA Perspective Bram Zuckerman, MD, FACC Director, FDA Division of Cardiovascular Devices Office of Device Evaluation Center for Devices and Radiological
Unprotected Left Main Stenting: Patient Selection and Recent Experience. Alaide Chieffo. S. Raffaele Hospital, Milan, Italy
 Unprotected Left Main Stenting: Patient Selection and Recent Experience Alaide Chieffo S. Raffaele Hospital, Milan, Italy Class IIa (Level B) AHA/ACC 2005 Guidelines Left Main CAD The use of PCI for pts
Unprotected Left Main Stenting: Patient Selection and Recent Experience Alaide Chieffo S. Raffaele Hospital, Milan, Italy Class IIa (Level B) AHA/ACC 2005 Guidelines Left Main CAD The use of PCI for pts
A Large Prospective Randomized Trial of DES vs BMS in Patients with STEMI
 HORIZONS-AMI: A Large Prospective Randomized Trial of DES vs BMS in Patients with STEMI Gregg W. Stone MD Columbia University Medical Center Cardiovascular Research Foundation Disclosures Gregg W. Stone
HORIZONS-AMI: A Large Prospective Randomized Trial of DES vs BMS in Patients with STEMI Gregg W. Stone MD Columbia University Medical Center Cardiovascular Research Foundation Disclosures Gregg W. Stone
COMPARE Trial Elvin Kedhi Maasstad Ziekenhuis Rotterdam The Netherlands
 COMPARE Trial Elvin Kedhi Maasstad Ziekenhuis Rotterdam The Netherlands TCTAP 2010 Seoul, Korea Disclosures Research Foundation of the Cardiology Department has received unrestricted research grants from:
COMPARE Trial Elvin Kedhi Maasstad Ziekenhuis Rotterdam The Netherlands TCTAP 2010 Seoul, Korea Disclosures Research Foundation of the Cardiology Department has received unrestricted research grants from:
Perspective of LM stenting with Current registry and Randomized Clinical Data
 Asian Pacific TCT Perspective of LM stenting with Current registry and Randomized Clinical Data Patrick W. Serruys MD PhD Yoshinobu Onuma MD Seung-Jung Park MD, PhD 14:48-15:00, 2009 Symposium Arena, Level
Asian Pacific TCT Perspective of LM stenting with Current registry and Randomized Clinical Data Patrick W. Serruys MD PhD Yoshinobu Onuma MD Seung-Jung Park MD, PhD 14:48-15:00, 2009 Symposium Arena, Level
PRASUGREL HYDROCHLORIDE (Effient Eli Lilly Canada Inc.) Indication: Acute Coronary Syndrome
 CEDAC FINAL RECOMMENDATION PRASUGREL HYDROCHLORIDE (Effient Eli Lilly Canada Inc.) Indication: Acute Coronary Syndrome Recommendation: The Canadian Expert Drug Advisory Committee (CEDAC) recommends that
CEDAC FINAL RECOMMENDATION PRASUGREL HYDROCHLORIDE (Effient Eli Lilly Canada Inc.) Indication: Acute Coronary Syndrome Recommendation: The Canadian Expert Drug Advisory Committee (CEDAC) recommends that
Absorbable Scaffolds the Future of Coronary Interventions?
 Absorbable Scaffolds the Future of Coronary Interventions? Brock Cookman, DO, MSA Interventional Cardiology Mercy Heart and Vascular Institute April 8, 2017 None Disclosures Objectives History of cardiac
Absorbable Scaffolds the Future of Coronary Interventions? Brock Cookman, DO, MSA Interventional Cardiology Mercy Heart and Vascular Institute April 8, 2017 None Disclosures Objectives History of cardiac
What s New in Antiplatelet Therapy and DES in 2016
 What s New in Antiplatelet Therapy and DES in 2016 Peter Pollak MD Director of Structural Heart Disease & Intervention Mayo Clinic Florida 2016 slide-1 Disclosures Financial None Off-label - None 2016
What s New in Antiplatelet Therapy and DES in 2016 Peter Pollak MD Director of Structural Heart Disease & Intervention Mayo Clinic Florida 2016 slide-1 Disclosures Financial None Off-label - None 2016
Disclosures. Theodore A. Bass MD, FSCAI. The following relationships exist related to this presentation. None
 SCAI Fellows Course December 10, 2013 Disclosures Theodore A. Bass MD, FSCAI The following relationships exist related to this presentation None Current Controversies on DAPT in PCI Which drug? When to
SCAI Fellows Course December 10, 2013 Disclosures Theodore A. Bass MD, FSCAI The following relationships exist related to this presentation None Current Controversies on DAPT in PCI Which drug? When to
Clinical Study Everolimus-Eluting versus Paclitaxel-Eluting Stents in Percutaneous Coronary Intervention: Meta-Analysis of Randomized Trials
 Thrombosis Volume 2012, Article ID 126369, 8 pages doi:10.1155/2012/126369 Clinical Study Everolimus-Eluting versus Paclitaxel-Eluting Stents in Percutaneous Coronary Intervention: Meta-Analysis of Randomized
Thrombosis Volume 2012, Article ID 126369, 8 pages doi:10.1155/2012/126369 Clinical Study Everolimus-Eluting versus Paclitaxel-Eluting Stents in Percutaneous Coronary Intervention: Meta-Analysis of Randomized
Randomized Trials and Registries: all smoke and mirrors or clear enlightenment?
 Randomized Trials and Registries: all smoke and mirrors or clear enlightenment? Laura Mauri, MD, MSc Brigham and Women s Hospital Associate Professor of Medicine, Harvard Medical School ADVANCED CARDIOVASCULAR
Randomized Trials and Registries: all smoke and mirrors or clear enlightenment? Laura Mauri, MD, MSc Brigham and Women s Hospital Associate Professor of Medicine, Harvard Medical School ADVANCED CARDIOVASCULAR
Is There A LIfe for DES after discontinuation of Clopidogrel
 EMBARGOED UNTIL 3:45pm CT, Sunday, 11/16/14 - FOR MEDIA BACKGROUND ONLY - Do Not Publish Chicago 2014 Is There A LIfe for DES after discontinuation of Clopidogrel Six-month versus 24-month dual antiplatelet
EMBARGOED UNTIL 3:45pm CT, Sunday, 11/16/14 - FOR MEDIA BACKGROUND ONLY - Do Not Publish Chicago 2014 Is There A LIfe for DES after discontinuation of Clopidogrel Six-month versus 24-month dual antiplatelet
Moins de 6 mois d antiagrégants après DES?
 Moins de 6 mois d antiagrégants après DES? High Tech - Marseille 25 au 27 janvier 2011 Marie-Claude MORICE, MD, FESC, FACC Massy, France Pas de conflit d interet Was optimal duration of DAPT already established
Moins de 6 mois d antiagrégants après DES? High Tech - Marseille 25 au 27 janvier 2011 Marie-Claude MORICE, MD, FESC, FACC Massy, France Pas de conflit d interet Was optimal duration of DAPT already established
Safety-outcomes of bioresorbable Everolimus eluting scaffold in (German-Austrian-ABSORB RegIsteR)
 Safety-outcomes of bioresorbable Everolimus eluting scaffold in 2168 patients with CAD: 30 days follow-up from the GABI-R (German-Austrian-ABSORB RegIsteR) C. Hamm, University of Giessen, Germany on behalf
Safety-outcomes of bioresorbable Everolimus eluting scaffold in 2168 patients with CAD: 30 days follow-up from the GABI-R (German-Austrian-ABSORB RegIsteR) C. Hamm, University of Giessen, Germany on behalf
Adults With Diagnosed Diabetes
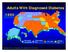 Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Surveying the Landscape of Oral Antiplatelet Therapy in Acute Coronary Syndrome Management
 Surveying the Landscape of Oral Antiplatelet Therapy in Acute Coronary Syndrome Management Jeffrey S Berger, MD, MS Assistant Professor of Medicine and Surgery Director of Cardiovascular Thrombosis Disclosures
Surveying the Landscape of Oral Antiplatelet Therapy in Acute Coronary Syndrome Management Jeffrey S Berger, MD, MS Assistant Professor of Medicine and Surgery Director of Cardiovascular Thrombosis Disclosures
PTCA 1979: : I
 The SYNTAX Message is Clear: CABG is Preferred in Complex MVD Angioplasty Summit TCTAP 2012 Seoul, Korea April 2012 David R. Holmes, MD Mayo Clinic Rochester, MN Presenter Disclosure Information David
The SYNTAX Message is Clear: CABG is Preferred in Complex MVD Angioplasty Summit TCTAP 2012 Seoul, Korea April 2012 David R. Holmes, MD Mayo Clinic Rochester, MN Presenter Disclosure Information David
