SPECIAL COMMUNICATION
|
|
|
- Angelica Lane
- 6 years ago
- Views:
Transcription
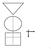
1 Evidence-Based Medicine, Part 5. An Introduction to Critical Appraisal of Articles on Prognosis Roberto Cardarelli, DO, MPH Joseph R. Oberdorfer, OMS IV This article provides an introductory step-by-step process to appraise an article on prognosis. The authors introduce these principles using a systematic approach and case-based format. The process of assessing the validity of an article on prognosis, determining its importance, and applying it to an individual patient is reviewed. The concepts of study population homogeneity, sufficient follow-up periods, and completeness are discussed to help physicians determine an article s validity. Instruction on how to evaluate confidence intervals is provided. Finally, information that is learned from the previously mentioned steps is applied to patient care. Study generalizability and the role of patient values, expectations, and concerns are also addressed. The skills learned from appraising an article on prognosis in the manner outlined provides a solid basis for life-long learning and improved patient care. [Editor s note: This article is part 5 of a six-article series intended to introduce the principles of evidence-based medicine (EBM) to busy clinicians, physician residents, and medical students. Because the application of EBM is a career-long process, further training is needed beyond the information provided within this article and series. A foundation of knowledge about research methods is critical in understanding EBM; however, such details, though introduced, are beyond the scope of this series.] J Am Osteopath Assoc. 2007;107: From the Department of Family Medicine at the University of North Texas Health Science Center Texas College of Osteopathic Medicine in Fort Worth. Address correspondence to Roberto Cardarelli, DO, MPH, Department of Family Medicine, Texas College of Osteopathic Medicine, University of North Texas Health Science Center, 855 Montgomery St, Patient Care Center, 2nd Fl, Fort Worth, TX rcardare@hsc.unt.edu Submitted March 13, 2007; revision received July 5, 2007; accepted July 9, After patients receive a diagnosis, a common question they ask their physicians is, What is my prognosis? Clear prognoses are important for patient care and counseling, but they also guide the physician and patient in selecting the most effective treatment. This complex process requires physicians to connect certain clinical predictors of disease and associated comorbidities to the temporal outcome of that disease. 1 However, not every physician can confidently provide appropriate prognoses in terms of the evidence available in the most current medical literature. For most physicians, prognostic skills develop with practice and experience and alongside clinical research. A framework exists in which any physician can master the art of making a prognosis and helping patients make informed decisions on the basis of available evidence provided by the current literature, the use of one s clinical expertise, and the unique circumstances relevant to a patient. This approach to medicine, called evidence-based medicine (EBM), is a way for physicians to accelerate their effectiveness in practicing various clinical skills, such as prognosis. 2,3 Applying EBM to prognoses requires that physicians thoroughly and critically review the current pertinent literature to verify the accuracy and validity of the evidence and its applicability to a specific clinical case. In this article, we introduce a strategy for busy physicians, physician residents, and medical students to critically assess the medical literature on prognosis. In-depth details of research methods are beyond the scope of this introductory series on EBM. Readers are encouraged to seek further training on these topics with supplemental learning opportunities and continuing medical education. Finally, the clinical scenario described has been simplified to provide readers with an illustrative example for the general concepts introduced. Searching the Evidence Choosing a high quality article to evaluate is a critical first step in practicing EBM. The preferred articles to support prognoses are systematic evidence-based reviews of studies of the specific disease or disorder in peer-reviewed journals. Unfortunately, systematic reviews on prognosis are sometimes hard to find. In such instances, individual research articles can be used with caution. Often the best study designs for prognostic articles are cohort studies (Figure 1). In such studies, investigators identify a group of individuals with a previously specified characteristic of interest and follow up with them over a predetermined period of time. The results of the group with the disease of interest are then compared with normal subjects who do not have the disease. 2-4 Other study designs, such as randomized controlled trials (RCTs) and case-control studies, JAOA Vol 107 No 8 August
2 Clinical Scenario A 65-year-old woman visits your primary care clinic for her annual physical. She tells you that she has noticed shortness of breath after walking up the stairs to work, after walking from her car to her home, and even while sitting at her desk. She presents to you with dyspnea and restlessness. The patient s medical history is negative for major illnesses or surgeries. The family history reveals that her father died of a disorder related to his heart and her mother died while she was very young. She reports that she drinks a glass of wine every night and denies smoking. On physical examination, her blood pressure is 183/95 mm Hg and her heart rate is 102 beats per minute. There is bilateral pitting edema of both ankles. During the pulmonary examination, you hear crackles in the right and left lower lobes of her lungs, and her cardiac examination is significant with an S 3 gallop on auscultation. You suspect class III heart failure based on New York Heart Association classification 5 and begin a full workup. After the blood work and echocardiograph you mention to the patient that she is suffering from congestive heart failure (CHF) and that you used a test that measured B- type natriuretic peptides (BNP) to support your diagnosis. Your patient is visibly worried about having something called heart failure and wishes to know what will happen to her and what to expect from this disease. You decide to use EBM techniques to evaluate the usefulness of BNP in determining the likelihood that she will have a cardiac event in the future. You are interested in studies that address the benefit of BNP to predict future cardiac events in patients with CHF. A PubMed search of the terms future cardiac events, dyspnea, and B-type natriuretic peptide yields a cohort study titled B-type natriuretic peptide predicts future cardiac events in patients presenting to the emergency department with dyspnea. You decide to review this study. Figure 1. Clinical scenario. have important roles in research and are introduced elsewhere in this series. 5-8 Validity of Articles on Prognosis The process of critically appraising an article on prognosis begins with evaluating the quality of information provided in the selected article. To ascertain the validity of an article, physicians need to determine not only if the methods used to arrive at the conclusions were free of error and bias but also if the study s results and conclusions were accurately deduced. Physicians may use the following questions 2 to help them determine the validity of an article on prognosis: Are the study subjects truly representative of the population of interest? A study of an entire population with a specific disease or disorder would be impractical. Instead, studies analyze a representative sample of a patient population and generalize the results to a larger population. The representative sample used in a study can have a direct impact on the usefulness of the results. For example, if the study s population has substantially different characteristics (eg, age, race or ethnicity, comorbidity) compared with a clinician s patient, the applicability of the results in this particular instance becomes questionable. Therefore, a study population may not be com- pletely representative of the general population or a particular clinician s patients. Were the study subjects chosen at a common point in the disease course? It is important to determine the stage of the disease that researchers began to evaluate the study s participants because the stage of the disease can impact whether the results are valid and applicable to a specific patient. For example, if the subjects in a study are all in an advanced stage of the disease (eg, New York Heart Association [NYHA] class III or IV for congestive heart failure [CHF] 9 ), a physician whose patient is in an earlier stage of the disease (eg, NYHA class I or II for CHF) might incorrectly deduce that his or her patient has a small amount of time before the patient will experience an expected endpoint. To assess the applicability of the article regarding the disease stage of recruited study participants, physicians should look at the article s abstract and its Methods section (Figure 2). Was the follow-up period sufficiently long and complete? Follow-up in a valid study should be sufficiently long and complete. Short follow-up periods may allow too little time for an outcome of interest to occur. The appropriate length of the study is dependent on the study question, the intervention used, the outcomes of interest, and any special circumstances (eg, funding). 316 JAOA Vol 107 No 8 Agust 2007
3 Clinical Scenario Clinical Scenario In the Methods section of the study, the authors report that patients presenting to an emergency department were eligible to participate in the study if they presented with shortness of breath at rest, on exertion, or lying down as their chief complaint. The demographic characteristics include a history of chronic heart failure (CHF), chronic obstructive pulmonary disease, and coronary artery disease, and a mean age of 65 years. This study has limitations in its applicability to your patient because it is not specific to CHF. In addition, the outcome of this study looked at the common chief complaint of shortness of breath, not diagnosis of CHF. Also, patients with different presentations of dyspnea were included. You decide to continue reviewing the article but will make sure to inform the patient of these limitations and how they affect any conclusions that can be made. The study shows that none of the 325 patients who entered the study were lost to follow-up. Although this result is initially reassuring, you soon realize that no patients were lost most likely because the follow-up period was only 6 months. This relatively short follow-up time period limits the applicability of the study s results to your patient to only those months where patients were observed. In addition, the authors define endpoint as repeat emergency department visits for chronic heart failure, hospital admission (cardiac), or death (cardiac and noncardiac). The high percentage of patients who reached an endpoint during a short follow-up leads you to read further and test the design against more standards. Figure 3. Clinical scenario. Figure 2. Clinical scenario. A clinical investigation is considered complete when all study participants are accounted for at the study s completion. Subjects who dropped out of the study early because of adverse effects or death should be included and analyzed with their original group assignment. This is called an intention-to-treat analysis. Failure to perform an intention-to-treat analysis may direct researchers to provide readers with misleading results. The 5-and-20 rule can be used by a critical reader to evaluate a study s completeness. If less than 5% of the study population is lost to follow-up, one can be assured that the loss minimally impacted the results. If, however, more than 20% of the study population is lost to follow-up, caution is advised when making clinical decisions based on study findings. An attrition rate of 5% to 20% and its impact on the researchers results must be determined by the reader based on other specifics of the study (Figure 3). For example, a study design that is complex with high participant burden or a long follow-up period can contribute to a large number of patients lost to follow-up. Were the study subjects and investigators blinded to the measures of interest? Assessment should also be blinded to prevent observer bias, which occurs when the researcher unconsciously (or consciously) looks harder for outcomes in the study group than in the control group. Blinding means that the researcher is unaware of the participants group assignment. Double-blinding means that study participants are also masked to group allocation. In prognosis articles, which are often presented as cohort studies, clinicians are usually single-blinded. In other words, clinicians are blinded to the method by which the outcome is measured, but not to the characteristics of the patients involved in the study (Figure 4). Were adjustments made for important prognostic factors? Studies on prognosis often stratify the study group into cohorts based on comorbid conditions that influence prognostic outcomes. 10 These factors can include the stage of disease or other diseases, such as coronary artery disease, that may affect the prognosis. In addition, factors that may confound the prognostic outcomes, such as age, sex, and environmental risk factors (eg, living conditions, pollution) must be accounted for. It is important to make sure that the authors of any study have identified factors applicable to their study s participants and determined if any of these factors influenced the predictive value of their conclusions (Figure 5). Study Results After the quality of a study on prognosis has been ascertained, physicians must determine what the results are. Evaluating the endpoints and projected outcomes can help physicians analyze the results and determine their importance. How likely are the projected outcomes over time? JAOA Vol 107 No 8 August
4 Clinical Scenario Clinical Scenario In reading the study, you learn that the physicians coded the patients into a high or low probability of chronic heart failure as the primary cause of their dyspnea. The investigators were blinded to the initial B-type natriuretic peptides reading; therefore, the reading did not bias the researchers and subjects. Figure 4. Clinical scenario. The Results section of the study reports that patients with B-type natriuretic peptides levels higher than 480 pg/ml had a 51% chance of experiencing one of the study s three primary endpoints (ie, a visit to the clinic, admission to the hospital, or death). However, patients with B-type natriuretic peptides less than 230 pg/ml only had a 2.5% probability of reaching one of the study s measured endpoints. Clinical Scenario You read through the Methods section of the study to determine if subgroups of patients with different prognoses are identified. The authors have identified four subgroups based on history of chronic disease, including: chronic heart failuire (CHF), chronic obstructive pulmonary disease (COPD), coronary artery disease, and CHF with COPD. You also review the demographic characteristics. In the Results section, the investigators used these subgroups to determine the best predictors of death or future cardiac events using the B-type natriuretic peptides lab evaluation. Figure 5. Clinical scenario. This question deals with the common question most physicians receive from their patients: What are my chances of having another event? Valid and accurate studies allow physicians to respond to this question quickly and within a reasonable amount of confidence. Often these findings can be located in the Results section of an article. One caveat that must be considered is the study endpoint. As presented in Figure 6, the endpoint can be defined as (1) an emergency department visit, (2) hospitalization as a result of CHF, or (3) death. Many times, studies provide a more stratified endpoint, such as only death caused by CHF, which may make the prognosis results more clinically applicable by allowing the clinician to provide information that is specific to the patient s concern. Moreover, composite endpoints vary in the severity of the outcome, making the results less clinically applicable. For Figure 6. Clinical scenario. example, an emergency department visit is substantially different from death. What is the precision of the relationship between the endpoint and the outcome? Odds ratios and relative risks are estimates of some unknown true value. If one were to repeat the study with different subjects, it would yield similar, but not identical, results. 2 The results of repeat studies are dispersed above and below the true value. This sampling variation is referred to as precision. The principle measure of precision is the 95% confidence interval (CI). The 95% CI quantifies the uncertainty of a measurement by reporting a range of values within which we can be 95% certain that the true value lies for the entire population. 2,11 Results are said to be significant if the 95% CI does not include 1.0. If the 95% CI includes 1.0, there is a 95% chance that there is no difference in the outcome between the comparison groups (Figure 7). Practical Use Having verified the validity and importance of a study on prognosis, a physician s ultimate consideration of the research would then become how the study s results and the authors conclusions can be applied in practice. 2 Making an evidence-based decision requires skills that integrate the patient s unique values and circumstances with current and valid evidence. One of the goals of EBM is to assist physicians in developing a system for backing up clinical decisions with sound evidence. Those who criticize EBM presumably focus on the first goal of treating patients according to the evidence. However, the goal of EBM is twofold. The second, and most important, goal of EBM is to shift physicians focus from the disease to the patient, restoring the patient to the center of the clinical decision-making process and better aligning the healthcare professions with longstanding osteopathic principles and practice. 318 JAOA Vol 107 No 8 August 2007
5 Clinical Scenario Looking at the study, which lists the relative risk of reaching a chronic heart failure (CHF) endpoint, you see that participants with B-type natriuretic peptides (BNP) levels over 230 pg/ml have a 15.5% greater risk of a CHF event than subjects with a BNP below 230 pg/ml. The confidence interval (CI) for this value is This wide range of the CI brings into question how sure you can be about the results of this study. Figure 7. Clinical scenario. Clinical Scenario The result of your patient s B-type natriuretic peptides is 360 pg/ml, which places her at increased risk for a future chronic heart failure event. After evaluating the study, you question the applicability of the results to your patient. The subjects of the study were predominantly men, and many of them were on some form of treatment or had a history of chronic obstructive pulmonary disease or coronary artery disease. These characteristics do not apply to your patient. You explain to the patient that the study s duration was only 6 months and there is no evidence beyond that point. You also explain that you searched the literature and that this study was fairly limited in its impact on treatment. Figure 8. Clinical scenario. Are the study subjects substantially different from your specific patient? The findings of a valid, important study are considered applicable if the patient to be treated is similar to those described in the study. As mentioned earlier, such information can be accessed by reviewing the study population characteristics. How important is the evidence for your specific patient? If the evidence from the study shows that the prognosis for the patient is disastrous without treatment, then this information should be presented to and discussed with the patient (Figure 8). The risks and benefits must be weighed while taking into account the patient s special circumstances (eg, financial concerns, health insurance coverage, and personal wishes). Any evidence becomes moot if the patient is not informed or willing to undergo further diagnostic tests or treatment. Conclusion Although most clinicians are already incorporating EBM principles in their practices, often instinctively, some physicians may require a more organized approach to integrating this relatively new model of self-education. Improved comfort levels and true expertise in the practice of EBM are the result of additional education, repetition, and self-assessment. The principles of EBM allow physicians to stay informed while also improving the quality of the information communicated to patients during patient encounters. The systematic approach that is used to appraise an article on prognosis is but one step in practicing EBM. Remember, the goal is always to provide the best care possible to patients using one s clinical expertise to address patient values and expectations for treatment. References 1. Hagland M. Medicare & Medicaid. A case against cookie-cutter care. Hosp Health Netw. 1997;71:78, Straus SE, Richardson WS, Glasziou P, Haynes RB. Evidence-Based Medicine: How to Practice and Teach EBM. 3rd ed. London, England: Churchill Livingstone; Laupacis A, Wells G, Richardson WS, Tugwell P. Users guides to the medical literature. V. How to use an article about prognosis. Evidence-Based Medicine Working Group. JAMA. 1994;272: Rothman KJ, Greenland S. Causation and causal inference. In: Rothman KJ, Greenland S, eds. Modern Epidemiology. 2nd ed. Boston, Mass: Lippincott Williams & Willkins; 1998: Cardarelli R, Virgilio RF, Taylor L. Evidence-based medicine, part 2. An introduction to critical appraisal of articles on therapy. J Am Osteopath Assoc. 2007;107: Available at: 6. Schranz DA, Dunn MA. Evidence-based medicine, part 3. An introduction to critical appraisal of articles on diagnosis. J Am Osteopath Assoc. 2007; 107: Available at: 7. Cardarelli R, Seater MM. Evidence-based medicine, part 4. An introduction to critical appraisal of articles on harm. J Am Osteopath Assoc. 2007;107: Available at: 8. Sanderlin BW, AbdulRahim N. Evidence-based medicine, part 6. An introduction to critical appraisal of clinical practice guidelines. J Am Osteopath Assoc. 2007;107: Available at: 9. The Stages of Heart Failure NYHA Classification. Heart Failure Society of America Web site Available at: _stages.htm. Accessed August 8, Hemingway H. Prognosis research: why is Dr Lydgate still waiting? J Clin Epidemiol. 2006;59: Rosner B. Fundamentals of Biostatistics. 6th ed. Belmont, Calif: Duxbury Press; JAOA Vol 107 No 8 August
SPECIAL COMMUNICATION
 Evidence-Based Medicine, Part 2. An Introduction to Critical Appraisal of Articles on Therapy Roberto Cardarelli, DO, MPH Richard F. Virgilio, DO Lockwood Taylor, MPH This article provides an introductory
Evidence-Based Medicine, Part 2. An Introduction to Critical Appraisal of Articles on Therapy Roberto Cardarelli, DO, MPH Richard F. Virgilio, DO Lockwood Taylor, MPH This article provides an introductory
CRITICAL APPRAISAL WORKSHEET 1
 For Residents CRITICAL APPRAISAL WORKSHEET 1 AN ARTICLE ON THERAPY This worksheet should be submitted along with the completed Submission Form to your supervisor. It is based on the articles on therapy
For Residents CRITICAL APPRAISAL WORKSHEET 1 AN ARTICLE ON THERAPY This worksheet should be submitted along with the completed Submission Form to your supervisor. It is based on the articles on therapy
SPECIAL COMMUNICATION. Evidence-Based Medicine, Part 3. An Introduction to Critical Appraisal of Articles on Diagnosis
 Evidence-Based Medicine, Part 3. An Introduction to Critical Appraisal of Articles on Diagnosis Damon A. Schranz, DO Michael A. Dunn, OMS III, MBA This article provides an introductory step-by-step process
Evidence-Based Medicine, Part 3. An Introduction to Critical Appraisal of Articles on Diagnosis Damon A. Schranz, DO Michael A. Dunn, OMS III, MBA This article provides an introductory step-by-step process
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
BIOSTATISTICAL METHODS
 BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH PROPENSITY SCORE Confounding Definition: A situation in which the effect or association between an exposure (a predictor or risk factor) and
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH PROPENSITY SCORE Confounding Definition: A situation in which the effect or association between an exposure (a predictor or risk factor) and
Appraising the Literature Overview of Study Designs
 Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
School of Population and Public Health SPPH 503 Epidemiologic methods II January to April 2019
 School of Population and Public Health SPPH 503 Epidemiologic methods II January to April 2019 Time: Tuesday, 1330 1630 Location: School of Population and Public Health, UBC Course description Students
School of Population and Public Health SPPH 503 Epidemiologic methods II January to April 2019 Time: Tuesday, 1330 1630 Location: School of Population and Public Health, UBC Course description Students
RACE611 CLINICAL EPIDEMIOLOGY AND EVIDENCE-BASED MEDICINE Prognostic study
 RACE611 CLINICAL EPIDEMIOLOGY AND EVIDENCE-BASED MEDICINE Prognostic study Assoc.Prof.Dr.Atiporn Ingsathit Master of Science Program in Medical Epidemiology and Doctor of Philosophy Program in Clinical
RACE611 CLINICAL EPIDEMIOLOGY AND EVIDENCE-BASED MEDICINE Prognostic study Assoc.Prof.Dr.Atiporn Ingsathit Master of Science Program in Medical Epidemiology and Doctor of Philosophy Program in Clinical
Essential Skills for Evidence-based Practice Understanding and Using Systematic Reviews
 J Nurs Sci Vol.28 No.4 Oct - Dec 2010 Essential Skills for Evidence-based Practice Understanding and Using Systematic Reviews Jeanne Grace Corresponding author: J Grace E-mail: Jeanne_Grace@urmc.rochester.edu
J Nurs Sci Vol.28 No.4 Oct - Dec 2010 Essential Skills for Evidence-based Practice Understanding and Using Systematic Reviews Jeanne Grace Corresponding author: J Grace E-mail: Jeanne_Grace@urmc.rochester.edu
The comparison or control group may be allocated a placebo intervention, an alternative real intervention or no intervention at all.
 1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
Outline. What is Evidence-Based Practice? EVIDENCE-BASED PRACTICE. What EBP is Not:
 Evidence Based Practice Primer Outline Evidence Based Practice (EBP) EBP overview and process Formulating clinical questions (PICO) Searching for EB answers Trial design Critical appraisal Assessing the
Evidence Based Practice Primer Outline Evidence Based Practice (EBP) EBP overview and process Formulating clinical questions (PICO) Searching for EB answers Trial design Critical appraisal Assessing the
Evidence Informed Practice Online Learning Module Glossary
 Term Abstract Associations Attrition Bias Background and Significance Baseline Basic Science Bias Blinding Definition An abstract is a summary of a research article. It usually includes the purpose, methods,
Term Abstract Associations Attrition Bias Background and Significance Baseline Basic Science Bias Blinding Definition An abstract is a summary of a research article. It usually includes the purpose, methods,
INTERPRETATION OF STUDY FINDINGS: PART I. Julie E. Buring, ScD Harvard School of Public Health Boston, MA
 INTERPRETATION OF STUDY FINDINGS: PART I Julie E. Buring, ScD Harvard School of Public Health Boston, MA Drawing Conclusions TRUTH IN THE UNIVERSE Infer TRUTH IN THE STUDY Infer FINDINGS IN THE STUDY Designing
INTERPRETATION OF STUDY FINDINGS: PART I Julie E. Buring, ScD Harvard School of Public Health Boston, MA Drawing Conclusions TRUTH IN THE UNIVERSE Infer TRUTH IN THE STUDY Infer FINDINGS IN THE STUDY Designing
How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study:
 CASP Checklist: 11 questions to help you make sense of a Case Control Study How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study: Are the results
CASP Checklist: 11 questions to help you make sense of a Case Control Study How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study: Are the results
Consider the following hypothetical
 Nurse Educator Nurse Educator Vol. 32, No. 1, pp. 16-20 Copyright! 2007 Wolters Kluwer Health Lippincott Williams & Wilkins How to Read, Interpret, and Understand Evidence-Based Literature Statistics Dorette
Nurse Educator Nurse Educator Vol. 32, No. 1, pp. 16-20 Copyright! 2007 Wolters Kluwer Health Lippincott Williams & Wilkins How to Read, Interpret, and Understand Evidence-Based Literature Statistics Dorette
11 questions to help you make sense of a case control study
 Critical Appraisal Skills Programme (CASP) making sense of evidence 11 questions to help you make sense of a case control study How to use this appraisal tool Three broad issues need to be considered when
Critical Appraisal Skills Programme (CASP) making sense of evidence 11 questions to help you make sense of a case control study How to use this appraisal tool Three broad issues need to be considered when
Critical Appraisal. Dave Abbott Senior Medicines Information Pharmacist
 Critical Appraisal Dave Abbott Senior Medicines Information Pharmacist Aims Identify key components of clinical trial design and apply these to a critical appraisal of the literature Be able to work out
Critical Appraisal Dave Abbott Senior Medicines Information Pharmacist Aims Identify key components of clinical trial design and apply these to a critical appraisal of the literature Be able to work out
APPLYING EVIDENCE-BASED METHODS IN PSYCHIATRY JOURNAL CLUB: HOW TO READ & CRITIQUE ARTICLES
 University of Miami Scholarly Repository Faculty Research, Publications, and Presentations Department of Health Informatics 7-26-2017 APPLYING EVIDENCE-BASED METHODS IN PSYCHIATRY JOURNAL CLUB: HOW TO
University of Miami Scholarly Repository Faculty Research, Publications, and Presentations Department of Health Informatics 7-26-2017 APPLYING EVIDENCE-BASED METHODS IN PSYCHIATRY JOURNAL CLUB: HOW TO
CONSORT 2010 checklist of information to include when reporting a randomised trial*
 CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Title and abstract Introduction Background and objectives Item No Checklist item 1a Identification as a
CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Title and abstract Introduction Background and objectives Item No Checklist item 1a Identification as a
CareFirst Hospice. Health care for the end of life. CareFirst
 Hospice Health care for the end of life 1 What is Hospice? Hospice is a philosophy- When a person in end stages of an illness can no longer receive, or wants to receive, life sustaining treatment, he or
Hospice Health care for the end of life 1 What is Hospice? Hospice is a philosophy- When a person in end stages of an illness can no longer receive, or wants to receive, life sustaining treatment, he or
Critical Appraisal for Research Papers. Appraisal Checklist & Guide Questions
 Critical Appraisal for Research Papers Appraisal Checklist & Guide Questions EBP@NUHS Critically Appraised Topic (CAT) Checklist and Guide Questions Sullivan 2008 2 Critical Appraisal for Research Papers
Critical Appraisal for Research Papers Appraisal Checklist & Guide Questions EBP@NUHS Critically Appraised Topic (CAT) Checklist and Guide Questions Sullivan 2008 2 Critical Appraisal for Research Papers
The role of Randomized Controlled Trials
 The role of Randomized Controlled Trials Dr. Georgia Salanti Lecturer in Epidemiology University of Ioannina School of Medicine Outline Understanding study designs and the role of confounding Observational
The role of Randomized Controlled Trials Dr. Georgia Salanti Lecturer in Epidemiology University of Ioannina School of Medicine Outline Understanding study designs and the role of confounding Observational
Critical Review Form Therapy Objectives: Methods:
 Critical Review Form Therapy Clinical Trial Comparing Primary Coronary Angioplasty with Tissue-Plasminogen Activator for Acute Myocardial Infarction (GUSTO-IIb), NEJM 1997; 336: 1621-1628 Objectives: To
Critical Review Form Therapy Clinical Trial Comparing Primary Coronary Angioplasty with Tissue-Plasminogen Activator for Acute Myocardial Infarction (GUSTO-IIb), NEJM 1997; 336: 1621-1628 Objectives: To
Evidence Based Practice (EBP) Five Step Process EBM. A Definition of EBP 10/13/2009. Fall
 What is EBP? Classic Definition of Evidence Based Medicine (EBM) By Aaron Eakman PTOT 413/513 OT Profession Fall 2009 the explicit, judicious and conscientious use of current best evidence from health
What is EBP? Classic Definition of Evidence Based Medicine (EBM) By Aaron Eakman PTOT 413/513 OT Profession Fall 2009 the explicit, judicious and conscientious use of current best evidence from health
Welcome to the Louis Calder Memorial Library NW 10 Ave., Miami, FL 33136
 Welcome to the Louis Calder Memorial Library 1601 NW 10 Ave., Miami, FL 33136 Objectives: Participants will be able to: 0 Use usearch to find electronic and print resources from all UM libraries 0 Navigate
Welcome to the Louis Calder Memorial Library 1601 NW 10 Ave., Miami, FL 33136 Objectives: Participants will be able to: 0 Use usearch to find electronic and print resources from all UM libraries 0 Navigate
Types of Biomedical Research
 INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
12/26/2013. Types of Biomedical Research. Clinical Research. 7Steps to do research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH S T A T I S T I C
 Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
Evidence- and Value-based Solutions for Health Care Clinical Improvement Consults, Content Development, Training & Seminars, Tools
 Definition Key Points Key Problems Bias Choice Lack of Control Chance Observational Study Defined Epidemiological study in which observations are made, but investigators do not control the exposure or
Definition Key Points Key Problems Bias Choice Lack of Control Chance Observational Study Defined Epidemiological study in which observations are made, but investigators do not control the exposure or
CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S
 CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S What is critical appraisal? Critical appraisal is the assessment of evidence
CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S What is critical appraisal? Critical appraisal is the assessment of evidence
GATE: Graphic Appraisal Tool for Epidemiology picture, 2 formulas & 3 acronyms
 GATE: Graphic Appraisal Tool for Epidemiology 1991-2015 1 picture, 2 formulas & 3 acronyms 1 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach To
GATE: Graphic Appraisal Tool for Epidemiology 1991-2015 1 picture, 2 formulas & 3 acronyms 1 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach To
Chapter Three: Sampling Methods
 Chapter Three: Sampling Methods The idea of this chapter is to make sure that you address sampling issues - even though you may be conducting an action research project and your sample is "defined" by
Chapter Three: Sampling Methods The idea of this chapter is to make sure that you address sampling issues - even though you may be conducting an action research project and your sample is "defined" by
1. Draft checklist for judging on quality of animal studies (Van der Worp et al., 2010)
 Appendix C Quality appraisal tools (QATs) 1. Draft checklist for judging on quality of animal studies (Van der Worp et al., 2010) 2. NICE Checklist for qualitative studies (2012) 3. The Critical Appraisal
Appendix C Quality appraisal tools (QATs) 1. Draft checklist for judging on quality of animal studies (Van der Worp et al., 2010) 2. NICE Checklist for qualitative studies (2012) 3. The Critical Appraisal
Glossary of Practical Epidemiology Concepts
 Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Professional Development: proposals for assuring the continuing fitness to practise of osteopaths. draft Peer Discussion Review Guidelines
 5 Continuing Professional Development: proposals for assuring the continuing fitness to practise of osteopaths draft Peer Discussion Review Guidelines February January 2015 2 draft Peer Discussion Review
5 Continuing Professional Development: proposals for assuring the continuing fitness to practise of osteopaths draft Peer Discussion Review Guidelines February January 2015 2 draft Peer Discussion Review
OCW Epidemiology and Biostatistics, 2010 Michael D. Kneeland, MD November 18, 2010 SCREENING. Learning Objectives for this session:
 OCW Epidemiology and Biostatistics, 2010 Michael D. Kneeland, MD November 18, 2010 SCREENING Learning Objectives for this session: 1) Know the objectives of a screening program 2) Define and calculate
OCW Epidemiology and Biostatistics, 2010 Michael D. Kneeland, MD November 18, 2010 SCREENING Learning Objectives for this session: 1) Know the objectives of a screening program 2) Define and calculate
Why do Psychologists Perform Research?
 PSY 102 1 PSY 102 Understanding and Thinking Critically About Psychological Research Thinking critically about research means knowing the right questions to ask to assess the validity or accuracy of a
PSY 102 1 PSY 102 Understanding and Thinking Critically About Psychological Research Thinking critically about research means knowing the right questions to ask to assess the validity or accuracy of a
Mapping the Informed Health Choices (IHC) Key Concepts (KC) to core concepts for the main steps of Evidence-Based Health Care (EBHC).
 KC No KC Short Title KC Statement EBHC concept EBHC sub concept EBHC statement EBHC Step Reported 1.01 Treatments can harm Treatments may be harmful Explain the use of harm/aetiologies for (rare) adverse
KC No KC Short Title KC Statement EBHC concept EBHC sub concept EBHC statement EBHC Step Reported 1.01 Treatments can harm Treatments may be harmful Explain the use of harm/aetiologies for (rare) adverse
W e have previously described the disease impact
 606 THEORY AND METHODS Impact numbers: measures of risk factor impact on the whole population from case-control and cohort studies R F Heller, A J Dobson, J Attia, J Page... See end of article for authors
606 THEORY AND METHODS Impact numbers: measures of risk factor impact on the whole population from case-control and cohort studies R F Heller, A J Dobson, J Attia, J Page... See end of article for authors
Patient Decision Aid. Summary Guide for Clinicians. Clinician s Checklist
 U.S. Department of Health & Human Services About Us Careers Contact Us Español FAQ Email Updates Effective Health Care Home / Decision Aids / Lung Cancer Screening Tools Patient Decision Aid Summary Guide
U.S. Department of Health & Human Services About Us Careers Contact Us Español FAQ Email Updates Effective Health Care Home / Decision Aids / Lung Cancer Screening Tools Patient Decision Aid Summary Guide
Evidence-based practice
 CHAPTER 1 Evidence-based practice 1.1 Why evidence-based practice? We all like to think we are practicing medicine based on the best evidence available. However, we sometimes do things in medicine for
CHAPTER 1 Evidence-based practice 1.1 Why evidence-based practice? We all like to think we are practicing medicine based on the best evidence available. However, we sometimes do things in medicine for
observational studies Descriptive studies
 form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
Study Design STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL STUDY DESIGN
 STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL Daniel E. Ford, MD, MPH Vice Dean for Clinical Investigation Johns Hopkins School of Medicine Introduction to Clinical Research July 15, 2014 STUDY DESIGN Provides
STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL Daniel E. Ford, MD, MPH Vice Dean for Clinical Investigation Johns Hopkins School of Medicine Introduction to Clinical Research July 15, 2014 STUDY DESIGN Provides
Journal Club: Fairfax Internal Medicine. Stephanie Shin PGY-2 Bianca Ummat PGY-2 July 27, 2012
 Journal Club: Fairfax Internal Medicine Stephanie Shin PGY-2 Bianca Ummat PGY-2 July 27, 2012 S Objective Review EBM basic principles Learn how to apply the principles in critically appraising the article.
Journal Club: Fairfax Internal Medicine Stephanie Shin PGY-2 Bianca Ummat PGY-2 July 27, 2012 S Objective Review EBM basic principles Learn how to apply the principles in critically appraising the article.
PubH 7405: REGRESSION ANALYSIS. Propensity Score
 PubH 7405: REGRESSION ANALYSIS Propensity Score INTRODUCTION: There is a growing interest in using observational (or nonrandomized) studies to estimate the effects of treatments on outcomes. In observational
PubH 7405: REGRESSION ANALYSIS Propensity Score INTRODUCTION: There is a growing interest in using observational (or nonrandomized) studies to estimate the effects of treatments on outcomes. In observational
MCQ Course in Pediatrics Al Yamamah Hospital June Dr M A Maleque Molla, FRCP, FRCPCH
 MCQ Course in Pediatrics Al Yamamah Hospital 10-11 June Dr M A Maleque Molla, FRCP, FRCPCH Q1. Following statements are true in the steps of evidence based medicine except ; a) Convert the need for information
MCQ Course in Pediatrics Al Yamamah Hospital 10-11 June Dr M A Maleque Molla, FRCP, FRCPCH Q1. Following statements are true in the steps of evidence based medicine except ; a) Convert the need for information
Guidelines for Writing and Reviewing an Informed Consent Manuscript From the Editors of Clinical Research in Practice: The Journal of Team Hippocrates
 Guidelines for Writing and Reviewing an Informed Consent Manuscript From the Editors of Clinical Research in Practice: The Journal of Team Hippocrates 1. Title a. Emphasize the clinical utility of the
Guidelines for Writing and Reviewing an Informed Consent Manuscript From the Editors of Clinical Research in Practice: The Journal of Team Hippocrates 1. Title a. Emphasize the clinical utility of the
CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness. 11 questions to help you make sense of case control study
 CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness 11 questions to help you make sense of case control study General comments Three broad issues need to be considered
CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness 11 questions to help you make sense of case control study General comments Three broad issues need to be considered
There For You. Your Compassionate Guide. World-Class Hospice Care Since 1979
 There For You Your Compassionate Guide World-Class Hospice Care Since 1979 What Is Hospice? Hospice is a type of care designed to provide support during an advanced illness. Hospice care focuses on comfort
There For You Your Compassionate Guide World-Class Hospice Care Since 1979 What Is Hospice? Hospice is a type of care designed to provide support during an advanced illness. Hospice care focuses on comfort
Evidence Based Medicine
 Course Goals Goals 1. Understand basic concepts of evidence based medicine (EBM) and how EBM facilitates optimal patient care. 2. Develop a basic understanding of how clinical research studies are designed
Course Goals Goals 1. Understand basic concepts of evidence based medicine (EBM) and how EBM facilitates optimal patient care. 2. Develop a basic understanding of how clinical research studies are designed
Washington, DC, November 9, 2009 Institute of Medicine
 Holger Schünemann, MD, PhD Chair, Department of Clinical Epidemiology & Biostatistics Michael Gent Chair in Healthcare Research McMaster University, Hamilton, Canada Washington, DC, November 9, 2009 Institute
Holger Schünemann, MD, PhD Chair, Department of Clinical Epidemiology & Biostatistics Michael Gent Chair in Healthcare Research McMaster University, Hamilton, Canada Washington, DC, November 9, 2009 Institute
Critical Appraisal Series
 Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
GATE: Graphic Appraisal Tool for Epidemiology picture, 2 formulas & 3 acronyms
 1 GATE: Graphic Appraisal Tool for Epidemiology 1991-2016 1 picture, 2 formulas & 3 acronyms 2 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach
1 GATE: Graphic Appraisal Tool for Epidemiology 1991-2016 1 picture, 2 formulas & 3 acronyms 2 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach
TEACHERS TOPICS. The Role of Matching in Epidemiologic Studies. American Journal of Pharmaceutical Education 2004; 68 (3) Article 83.
 TEACHERS TOPICS American Journal of Pharmaceutical Education 2004; 68 (3) Article 83. The Role of Matching in Epidemiologic Studies Kevin W. Garey, PharmD College of Pharmacy, University of Houston Submitted
TEACHERS TOPICS American Journal of Pharmaceutical Education 2004; 68 (3) Article 83. The Role of Matching in Epidemiologic Studies Kevin W. Garey, PharmD College of Pharmacy, University of Houston Submitted
RATING OF A RESEARCH PAPER. By: Neti Juniarti, S.Kp., M.Kes., MNurs
 RATING OF A RESEARCH PAPER RANDOMISED CONTROLLED TRIAL TO COMPARE SURGICAL STABILISATION OF THE LUMBAR SPINE WITH AN INTENSIVE REHABILITATION PROGRAMME FOR PATIENTS WITH CHRONIC LOW BACK PAIN: THE MRC
RATING OF A RESEARCH PAPER RANDOMISED CONTROLLED TRIAL TO COMPARE SURGICAL STABILISATION OF THE LUMBAR SPINE WITH AN INTENSIVE REHABILITATION PROGRAMME FOR PATIENTS WITH CHRONIC LOW BACK PAIN: THE MRC
Evidence-Based Medicine: Diagnostic study
 Evidence-Based Medicine: Diagnostic study What is Evidence-Based Medicine (EBM)? Expertise in integrating 1. Best research evidence 2. Clinical Circumstance 3. Patient values in clinical decisions Haynes,
Evidence-Based Medicine: Diagnostic study What is Evidence-Based Medicine (EBM)? Expertise in integrating 1. Best research evidence 2. Clinical Circumstance 3. Patient values in clinical decisions Haynes,
Abstract: Learning Objectives: Posttest:
 Title of Course: Evidence Based Practice in Communication Disorders CE Credit: 6 Hours (0.6 CEUs) Learning Level: Advanced Author: Christine A. Dollaghan, PhD, CCC SLP Abstract: With this landmark textbook,
Title of Course: Evidence Based Practice in Communication Disorders CE Credit: 6 Hours (0.6 CEUs) Learning Level: Advanced Author: Christine A. Dollaghan, PhD, CCC SLP Abstract: With this landmark textbook,
To evaluate a single epidemiological article we need to know and discuss the methods used in the underlying study.
 Critical reading 45 6 Critical reading As already mentioned in previous chapters, there are always effects that occur by chance, as well as systematic biases that can falsify the results in population
Critical reading 45 6 Critical reading As already mentioned in previous chapters, there are always effects that occur by chance, as well as systematic biases that can falsify the results in population
Critical Appraisal of RCT
 Critical Appraisal of RCT What is critical appraisal? Definition Process of systematically examining research evidence to assess its reliability (validity/internal validity), results and relevance (external
Critical Appraisal of RCT What is critical appraisal? Definition Process of systematically examining research evidence to assess its reliability (validity/internal validity), results and relevance (external
QUESTIONNAIRE USER'S GUIDE
 QUESTIONNAIRE USER'S GUIDE OUTBREAK INVESTIGATION Arthur L. Reingold's article "Outbreak Investigations--A Perspective" 1 provides an excellent synopsis of the procedures, challenges, and unique attributes
QUESTIONNAIRE USER'S GUIDE OUTBREAK INVESTIGATION Arthur L. Reingold's article "Outbreak Investigations--A Perspective" 1 provides an excellent synopsis of the procedures, challenges, and unique attributes
Hayden Smith, PhD, MPH /\ v._
 Hayden Smith, PhD, MPH.. + /\ v._ Information and clinical examples provided in presentation are strictly for educational purposes, and should not be substituted for clinical guidelines or up-to-date medical
Hayden Smith, PhD, MPH.. + /\ v._ Information and clinical examples provided in presentation are strictly for educational purposes, and should not be substituted for clinical guidelines or up-to-date medical
PRESS RELEASE. New NICE guidance will improve diagnosis and treatment of chronic heart failure
 Tel: 0845 003 7782 www.nice.org.uk Ref: 2010/118 ISSUED: WEDNESDAY, 25 AUGUST 2010 PRESS RELEASE New NICE guidance will improve diagnosis and treatment of chronic heart failure The National Institute for
Tel: 0845 003 7782 www.nice.org.uk Ref: 2010/118 ISSUED: WEDNESDAY, 25 AUGUST 2010 PRESS RELEASE New NICE guidance will improve diagnosis and treatment of chronic heart failure The National Institute for
PROMOTION OF PHYSICAL ACTIVITY
 General objective Resources «Living Well with COPD» Reference guide for individual patient education «Integrating physical activity and/or an exercise program into your life», Section Promotion of physical
General objective Resources «Living Well with COPD» Reference guide for individual patient education «Integrating physical activity and/or an exercise program into your life», Section Promotion of physical
CRITICAL APPRAISAL OF MEDICAL LITERATURE. Samuel Iff ISPM Bern
 CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
Mixed Methods Study Design
 1 Mixed Methods Study Design Kurt C. Stange, MD, PhD Professor of Family Medicine, Epidemiology & Biostatistics, Oncology and Sociology Case Western Reserve University 1. Approaches 1, 2 a. Qualitative
1 Mixed Methods Study Design Kurt C. Stange, MD, PhD Professor of Family Medicine, Epidemiology & Biostatistics, Oncology and Sociology Case Western Reserve University 1. Approaches 1, 2 a. Qualitative
EBM: Therapy. Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University
 EBM: Therapy Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University How to appraise therapy literature Are the result valid? What are the result?
EBM: Therapy Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University How to appraise therapy literature Are the result valid? What are the result?
Overview of Study Designs in Clinical Research
 Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
EBP ASKING. Constructing a Good Clinical Question Using the PICO Format
 EBP ASKING Constructing a Good Clinical Question Using the PICO Format Objectives: To demonstrate understanding of a good clinical question. To distinguish between a background question, usually answerable
EBP ASKING Constructing a Good Clinical Question Using the PICO Format Objectives: To demonstrate understanding of a good clinical question. To distinguish between a background question, usually answerable
Evidence-Based Medicine Journal Club. A Primer in Statistics, Study Design, and Epidemiology. August, 2013
 Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Evidence Based Medicine Prof P Rheeder Clinical Epidemiology. Module 2: Applying EBM to Diagnosis
 Evidence Based Medicine Prof P Rheeder Clinical Epidemiology Module 2: Applying EBM to Diagnosis Content 1. Phases of diagnostic research 2. Developing a new test for lung cancer 3. Thresholds 4. Critical
Evidence Based Medicine Prof P Rheeder Clinical Epidemiology Module 2: Applying EBM to Diagnosis Content 1. Phases of diagnostic research 2. Developing a new test for lung cancer 3. Thresholds 4. Critical
Clinical Epidemiology for the uninitiated
 Clinical epidemiologist have one foot in clinical care and the other in clinical practice research. As clinical epidemiologists we apply a wide array of scientific principles, strategies and tactics to
Clinical epidemiologist have one foot in clinical care and the other in clinical practice research. As clinical epidemiologists we apply a wide array of scientific principles, strategies and tactics to
Learning Objectives. Guidance Hierarchy. AHA Coding Clinic Update
 1 AHA Coding Clinic Update Nelly Leon Chisen, RHIA Director, Coding and Classification Executive Editor, Coding Clinic American Hospital Association Chicago, IL Learning Objectives At the completion of
1 AHA Coding Clinic Update Nelly Leon Chisen, RHIA Director, Coding and Classification Executive Editor, Coding Clinic American Hospital Association Chicago, IL Learning Objectives At the completion of
Journal Club Critical Appraisal Worksheets. Dr David Walbridge
 Journal Club Critical Appraisal Worksheets Dr David Walbridge The Four Part Question The purpose of a structured EBM question is: To be directly relevant to the clinical situation or problem To express
Journal Club Critical Appraisal Worksheets Dr David Walbridge The Four Part Question The purpose of a structured EBM question is: To be directly relevant to the clinical situation or problem To express
Best Practice Model Communication/Relational Skills in Soliciting the Patient/Family Story Stuart Farber
 Best Practice Model Communication/Relational Skills in Soliciting the Patient/Family Story Stuart Farber Once you have set a safe context for the palliative care discussion soliciting the patient's and
Best Practice Model Communication/Relational Skills in Soliciting the Patient/Family Story Stuart Farber Once you have set a safe context for the palliative care discussion soliciting the patient's and
An Update & Suggestions on How to Use It
 Information as of November 2014 NT-proBNP NT-proBNP may be used to help detect, diagnose and evaluate the severity of heart failure. An Update & Suggestions on How to Use It ExamOne s NT-proBNP Experience
Information as of November 2014 NT-proBNP NT-proBNP may be used to help detect, diagnose and evaluate the severity of heart failure. An Update & Suggestions on How to Use It ExamOne s NT-proBNP Experience
Lnformation Coverage Guidance
 Lnformation Coverage Guidance Coverage Indications, Limitations, and/or Medical Necessity Abstract: B-type natriuretic peptide (BNP) is a cardiac neurohormone produced mainly in the left ventricle. It
Lnformation Coverage Guidance Coverage Indications, Limitations, and/or Medical Necessity Abstract: B-type natriuretic peptide (BNP) is a cardiac neurohormone produced mainly in the left ventricle. It
Chapter 5: Field experimental designs in agriculture
 Chapter 5: Field experimental designs in agriculture Jose Crossa Biometrics and Statistics Unit Crop Research Informatics Lab (CRIL) CIMMYT. Int. Apdo. Postal 6-641, 06600 Mexico, DF, Mexico Introduction
Chapter 5: Field experimental designs in agriculture Jose Crossa Biometrics and Statistics Unit Crop Research Informatics Lab (CRIL) CIMMYT. Int. Apdo. Postal 6-641, 06600 Mexico, DF, Mexico Introduction
Evidence-based Imaging: Critically Appraising Studies of Diagnostic Tests
 Evidence-based Imaging: Critically Appraising Studies of Diagnostic Tests Aine Marie Kelly, MD Critically Appraising Studies of Diagnostic Tests Aine Marie Kelly B.A., M.B. B.Ch. B.A.O., M.S. M.R.C.P.I.,
Evidence-based Imaging: Critically Appraising Studies of Diagnostic Tests Aine Marie Kelly, MD Critically Appraising Studies of Diagnostic Tests Aine Marie Kelly B.A., M.B. B.Ch. B.A.O., M.S. M.R.C.P.I.,
Rapid appraisal of the literature: Identifying study biases
 Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Clinical Epidemiology I: Deciding on Appropriate Therapy
 Clinical Epidemiology I: Deciding on Appropriate Therapy 1 Clinical Scenario [UG2B] 65 yo man controlled HTN 6-mo Hx cardioversion-resistant afib Benefit vs risk of long-term anticoagulation:? prevent
Clinical Epidemiology I: Deciding on Appropriate Therapy 1 Clinical Scenario [UG2B] 65 yo man controlled HTN 6-mo Hx cardioversion-resistant afib Benefit vs risk of long-term anticoagulation:? prevent
STUDIES OF THE ACCURACY OF DIAGNOSTIC TESTS: (Relevant JAMA Users Guide Numbers IIIA & B: references (5,6))
 STUDIES OF THE ACCURACY OF DIAGNOSTIC TESTS: (Relevant JAMA Users Guide Numbers IIIA & B: references (5,6)) Introduction: The most valid study design for assessing the accuracy of diagnostic tests is a
STUDIES OF THE ACCURACY OF DIAGNOSTIC TESTS: (Relevant JAMA Users Guide Numbers IIIA & B: references (5,6)) Introduction: The most valid study design for assessing the accuracy of diagnostic tests is a
Setting The setting was secondary care. The economic study was carried out in Switzerland.
 Cost-effectiveness of B-type natriuretic peptide testing in patients with acute dyspnea Mueller C, Laule-Kilian K, Schindler C, Klima T, Frana B, Rodriguez D, Scholer A, Christ M, Perruchoud A P Record
Cost-effectiveness of B-type natriuretic peptide testing in patients with acute dyspnea Mueller C, Laule-Kilian K, Schindler C, Klima T, Frana B, Rodriguez D, Scholer A, Christ M, Perruchoud A P Record
Protocol Development: The Guiding Light of Any Clinical Study
 Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Delfini Evidence Tool Kit
 General 1. Who is sponsoring and funding the study? What are the affiliations of the authors? Study Design Assessment Internal Validity Assessment Considerations: This can be helpful information and is
General 1. Who is sponsoring and funding the study? What are the affiliations of the authors? Study Design Assessment Internal Validity Assessment Considerations: This can be helpful information and is
Presentation Preparation
 November 2015 TABLE OF CONTENTS page 1 CHIROPRACTORS PRESENTING CHIROPRACTIC TO OTHER HEALTH PRACTITIONERS Presentation Tips Getting Ready Presentation Day Following Up page 3 COMMON QUESTIONS AND HOW
November 2015 TABLE OF CONTENTS page 1 CHIROPRACTORS PRESENTING CHIROPRACTIC TO OTHER HEALTH PRACTITIONERS Presentation Tips Getting Ready Presentation Day Following Up page 3 COMMON QUESTIONS AND HOW
Evidence-Based Integrative Therapies for Common Problems in Family Medicine. Pamela Wiseman MD Associate Professor of Family Medicine
 Evidence-Based Integrative Therapies for Common Problems in Family Medicine Pamela Wiseman MD Associate Professor of Family Medicine Learning Objectives As a result of this session, the participant will
Evidence-Based Integrative Therapies for Common Problems in Family Medicine Pamela Wiseman MD Associate Professor of Family Medicine Learning Objectives As a result of this session, the participant will
BIOSTATISTICAL METHODS
 BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Designs on Micro Scale: DESIGNING CLINICAL RESEARCH THE ANATOMY & PHYSIOLOGY OF CLINICAL RESEARCH We form or evaluate a research or research
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Designs on Micro Scale: DESIGNING CLINICAL RESEARCH THE ANATOMY & PHYSIOLOGY OF CLINICAL RESEARCH We form or evaluate a research or research
Introduzione al metodo GRADE
 Introduzione al metodo GRADE Atto Billio MD, MSc EBHC Ematologia e TMO-Bolzano Gruppo linee guida SIE Critique of EBM De-emphasizes patient values Doesn t account for individual variation Devalues clinical
Introduzione al metodo GRADE Atto Billio MD, MSc EBHC Ematologia e TMO-Bolzano Gruppo linee guida SIE Critique of EBM De-emphasizes patient values Doesn t account for individual variation Devalues clinical
Bias and confounding special issues. Outline for evaluation of bias
 EPIDEMIOLOGI BIAS special issues and discussion of paper April 2009 Søren Friis Institut for Epidemiologisk Kræftforskning Kræftens Bekæmpelse AGENDA Bias and confounding special issues Confounding by
EPIDEMIOLOGI BIAS special issues and discussion of paper April 2009 Søren Friis Institut for Epidemiologisk Kræftforskning Kræftens Bekæmpelse AGENDA Bias and confounding special issues Confounding by
Critical Appraisal Istanbul 2011
 Critical Appraisal Istanbul 2011 The Conviction with which many Nephrologists hold an opinion varies inversely with the Evidence Ed Lewis The Good Old Times. The Google Generation. ASN Kidney Daily LEADING
Critical Appraisal Istanbul 2011 The Conviction with which many Nephrologists hold an opinion varies inversely with the Evidence Ed Lewis The Good Old Times. The Google Generation. ASN Kidney Daily LEADING
Bias. Zuber D. Mulla
 Bias Zuber D. Mulla Explanations when you Observe or Don t Observe an Association Truth Chance Bias Confounding From Epidemiology in Medicine (Hennekens & Buring) Bias When you detect an association or
Bias Zuber D. Mulla Explanations when you Observe or Don t Observe an Association Truth Chance Bias Confounding From Epidemiology in Medicine (Hennekens & Buring) Bias When you detect an association or
PHO MetaQAT Guide. Critical appraisal in public health. PHO Meta-tool for quality appraisal
 PHO MetaQAT Guide Critical appraisal in public health Critical appraisal is a necessary part of evidence-based practice and decision-making, allowing us to understand the strengths and weaknesses of evidence,
PHO MetaQAT Guide Critical appraisal in public health Critical appraisal is a necessary part of evidence-based practice and decision-making, allowing us to understand the strengths and weaknesses of evidence,
Epidemiologic Methods and Counting Infections: The Basics of Surveillance
 Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Assignment 4: True or Quasi-Experiment
 Assignment 4: True or Quasi-Experiment Objectives: After completing this assignment, you will be able to Evaluate when you must use an experiment to answer a research question Develop statistical hypotheses
Assignment 4: True or Quasi-Experiment Objectives: After completing this assignment, you will be able to Evaluate when you must use an experiment to answer a research question Develop statistical hypotheses
The Cochrane Collaboration
 The Cochrane Collaboration Version and date: V1, 29 October 2012 Guideline notes for consumer referees You have been invited to provide consumer comments on a Cochrane Review or Cochrane Protocol. This
The Cochrane Collaboration Version and date: V1, 29 October 2012 Guideline notes for consumer referees You have been invited to provide consumer comments on a Cochrane Review or Cochrane Protocol. This
Randomized Controlled Trial
 Randomized Controlled Trial Training Course in Sexual and Reproductive Health Research Geneva 2016 Dr Khalifa Elmusharaf MBBS, PgDip, FRSPH, PHD Senior Lecturer in Public Health Graduate Entry Medical
Randomized Controlled Trial Training Course in Sexual and Reproductive Health Research Geneva 2016 Dr Khalifa Elmusharaf MBBS, PgDip, FRSPH, PHD Senior Lecturer in Public Health Graduate Entry Medical
ARE THE RESULTS VALID?
 ARE THE RESULTS VALID? At the beginning, did intervention and control groups start with the same prognosis? Was randomization process adequate? Was randomization concealed? Were patients in the study groups
ARE THE RESULTS VALID? At the beginning, did intervention and control groups start with the same prognosis? Was randomization process adequate? Was randomization concealed? Were patients in the study groups
Structural Approach to Bias in Meta-analyses
 Original Article Received 26 July 2011, Revised 22 November 2011, Accepted 12 December 2011 Published online 2 February 2012 in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/jrsm.52 Structural
Original Article Received 26 July 2011, Revised 22 November 2011, Accepted 12 December 2011 Published online 2 February 2012 in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/jrsm.52 Structural
Purpose. Study Designs. Objectives. Observational Studies. Analytic Studies
 Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
The Research Enterprise in Psychology Chapter 2
 The Research Enterprise in Psychology Chapter 2 This multimedia product and its contents are protected under copyright law. The following are prohibited by law: any public performance or display, including
The Research Enterprise in Psychology Chapter 2 This multimedia product and its contents are protected under copyright law. The following are prohibited by law: any public performance or display, including
Evidence based practice. Dr. Rehab Gwada
 Evidence based practice Dr. Rehab Gwada Objectives Define the ABP Identify the steps of the EBP process Recognize hierarchy of evidence and types of study design Explain how to build focused clinical questions
Evidence based practice Dr. Rehab Gwada Objectives Define the ABP Identify the steps of the EBP process Recognize hierarchy of evidence and types of study design Explain how to build focused clinical questions
