Notification of a Body in the framework of a technical harmonization directive
|
|
|
- Lorena Harmon
- 5 years ago
- Views:
Transcription
1 Notification of a Body in the framework of a technical harmonization directive From : ANSM : Agence Nationale de Sécurité du Médicament et des produits de santé - Direction de l'inspection 143, 147, Boulevard Anatole France SAINT-DENIS CEDEX France To : European Commission GROWTH Directorate-General 200 Rue de la Loi, B-1049 Brussels. Other Member States Reference : Legislation : 93/42/EEC Medical devices Body name, address, telephone, fax, , website : GMED 1, rue Gaston Boissier PARIS France Phone : Fax : info@lne-gmed.com Website : Body : NB 0459 The body is formally accredited against : EN EN ISO/IEC Name of National Accreditation Body (NAB) : COFRAC - Comité Français d'accréditation The accreditation covers the product categories and conformity assessment procedures concerned by this notification : Yes 1 / 6
2 Tasks performed by the Body : Last approval date : 03/08/2018 Product family, product /Intended use/product *MD General non-active, non-implantable medical devices - *MD Non-active devices for anaesthesia, emergency and intensive care Full quality assurance system - *MD Non-active devices for injection, infusion, Full quality assurance system transfusion and dialysis - *MD Non-active orthopaedic and rehabilitationfull quality assurance system devices - *MD Non-active medical devices with measuring function Full quality assurance system - *MD Non-active ophthalmologic devices Full quality assurance system - *MD Non-active instruments Full quality assurance system - *MD Contraceptive medical devices Full quality assurance system - *MD Non-active medical devices for disinfecting, cleaning, rinsing Full quality assurance system - *MD Non-active devices for in vitro fertilisationfull quality assurance system (IVF) and assisted reproductive technologies (ART) - *MD Non-active medical devices for ingestion *MD Non-active implants - *MD Non-active cardiovascular implants I I I I I I I I I I I 2 / 6
3 - *MD Non-active orthopaedic implants - *MD Non-active functional implants - *MD Non-active soft tissue implants *MD Devices for wound care - *MD Bandages and wound dressings - *MD Suture material and clamps - *MD Other medical devices for wound care *MD Non-active dental devices and accessories - *MD Non-active dental equipment and instruments - *MD Dental materials I I I I I I I I 3 / 6
4 - *MD Dental implants *MD General active medical devices - *MD Devices for extra-corporal circulation, infusion and haemopheresis - *MD Respiratory devices, devices including hyperbaric chambers for oxygen therapy, inhalation anaesthesia - *MD Devices for stimulation or inhibition - *MD Active surgical devices - *MD Active ophthalmologic devices - *MD Active dental devices - *MD Active devices for disinfection and sterilisation I I I I I I I I 4 / 6
5 - *MD Active rehabilitation devices and active prostheses - *MD Active devices for patient positioning and transport - *MD Active devices for in vitro fertilisation (IVF) and assisted reproductive therapy (ART) - *MD Software - *MD Medical gas supply systems and parts thereof *MD Devices for imaging - *MD Imaging devices utilising ionizing radiation - *MD Imaging devices utilising non-ionizing radiation *MD Monitoring devices - *MD Monitoring devices of non-vital physiological parameters I I I I I I I I 5 / 6
6 - *MD Monitoring devices of vital physiological parameters *MD Devices for radiation therapy and thermo therapy - *MD Devices utilising ionizing radiation - *MD Devices utilising non-ionizing radiation - *MD Devices for hyperthermia / hypothermia - *MD Devices for (extracorporal) shock-wave therapy (lithotripsy) I I I I I Horizontal technical competence *MDS Medical devices incorporating medicinal substances, according to Directive 2001/83/EC *MDS Medical devices utilising tissues of animal origin, including Regulation 722/2012 (Directive 2003/32/EC up to ) *MDS Medical devices referencing the Directive 2006/42/EC on machinery *MDS Medical devices in sterile condition Chemical sterilization/dry heat sterilization/hydrogen peroxid with or without plasma process sterilization/ultra High Temperature Infusion sterilization process *MDS Medical devices utilising micromechanics *MDS Medical devices utilising nanomaterials *MDS Medical devices utilising biological active coatings and/or materials or being wholly or mainly absorbed *MDS Medical devices incorporating software /utilising software /controlled by software 6 / 6
Notification of a Body in the framework of a technical harmonization directive
 Notification of a Body in the framework of a technical harmonization directive From : National Organization for Medicines (EOF) Mesogeion 284, 155 62 Holargos Greece To : European Commission GROWTH Directorate-General
Notification of a Body in the framework of a technical harmonization directive From : National Organization for Medicines (EOF) Mesogeion 284, 155 62 Holargos Greece To : European Commission GROWTH Directorate-General
Notification of a Body in the framework of a technical harmonization directive
 Notification of a Body in the framework of a technical harmonization directive From : Ministry of Health ul. Miodowa 15 00-952 Warszawa Poland To : European Commission GROWTH Directorate-General 200 Rue
Notification of a Body in the framework of a technical harmonization directive From : Ministry of Health ul. Miodowa 15 00-952 Warszawa Poland To : European Commission GROWTH Directorate-General 200 Rue
Notification of a Body in the framework of a technical harmonization directive
 Notification of a Body in the framework of a technical harmonization directive From : Zentralstelle der Länder für Gesundheitsschutz bei Arzneimitteln und Medizinprodukten (ZLG) Heinrich-Böll-Ring 10 53119
Notification of a Body in the framework of a technical harmonization directive From : Zentralstelle der Länder für Gesundheitsschutz bei Arzneimitteln und Medizinprodukten (ZLG) Heinrich-Böll-Ring 10 53119
Notification of a Body in the framework of a technical harmonization directive
 Notification of a Body in the framework of a technical harmonization directive From : Zentralstelle der Länder für Gesundheitsschutz bei Arzneimitteln und Medizinprodukten (ZLG) Heinrich-Böll-Ring 10 53119
Notification of a Body in the framework of a technical harmonization directive From : Zentralstelle der Länder für Gesundheitsschutz bei Arzneimitteln und Medizinprodukten (ZLG) Heinrich-Böll-Ring 10 53119
Notification of a Body in the framework of a technical harmonization directive
 Notification of a Body in the framework of a technical harmonization directive From : Ministero dello Sviluppo Economico - Direzione Generale per il Mercato, la Concorrenza, il Consumatore, la Vigilanza
Notification of a Body in the framework of a technical harmonization directive From : Ministero dello Sviluppo Economico - Direzione Generale per il Mercato, la Concorrenza, il Consumatore, la Vigilanza
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK ISO/IEC 17021-1:2015 to provide quality management systems Davy Avenue Knowhill Milton Keynes MK5 8NL Contact: Dr Eugene Kotlirov Tel: +44
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK ISO/IEC 17021-1:2015 to provide quality management systems Davy Avenue Knowhill Milton Keynes MK5 8NL Contact: Dr Eugene Kotlirov Tel: +44
Office No. E E, Amber Gem Tower, SHeikh Khalifa Bin Zayed Road 2, Ajman, U.A.E
 IAS Accreditation Number MSCB-119 Company Name AQC MIDDLE EAST F.Z.E Address Office No. E1-1401 E, Amber Gem Tower, SHeikh Khalifa Bin Zayed Road 2, Ajman, U.A.E Contact Name Nadeem Pasha Telephone +971
IAS Accreditation Number MSCB-119 Company Name AQC MIDDLE EAST F.Z.E Address Office No. E1-1401 E, Amber Gem Tower, SHeikh Khalifa Bin Zayed Road 2, Ajman, U.A.E Contact Name Nadeem Pasha Telephone +971
Notice Information: - Advisory 01 November 2010
 Notice Information: - Advisory 01 November 2010 Part 1. Product Information a) Title: Single Use & Single Patient Use Medical Devices b) Product Name/Type: Single Use & Single Patient Use Medical Devices
Notice Information: - Advisory 01 November 2010 Part 1. Product Information a) Title: Single Use & Single Patient Use Medical Devices b) Product Name/Type: Single Use & Single Patient Use Medical Devices
EUROPEAN COMMISSION DIRECTORATE-GENERAL III INDUSTRY Industrial affairs III: Consumer goods industries Pharmaceuticals and Cosmetics
 EUROPEAN COMMISSION DIRECTORATE-GENERAL III INDUSTRY Industrial affairs III: Consumer goods industries Pharmaceuticals and Cosmetics Community Basic Format for Manufacturing Authorisation Explanatory Notes
EUROPEAN COMMISSION DIRECTORATE-GENERAL III INDUSTRY Industrial affairs III: Consumer goods industries Pharmaceuticals and Cosmetics Community Basic Format for Manufacturing Authorisation Explanatory Notes
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 14708-7 First edition 2013-01-15 Implants for surgery Active implantable medical devices Part 7: Particular requirements for cochlear implant systems Implants chirurgicaux Dispositifs
INTERNATIONAL STANDARD ISO 14708-7 First edition 2013-01-15 Implants for surgery Active implantable medical devices Part 7: Particular requirements for cochlear implant systems Implants chirurgicaux Dispositifs
PRAC non-interventional imposed PASS final study report assessment report
 1 December 2016 EMA/101726/2017 Pharmacovigilance Risk Assessment Committee (PRAC) PRAC non-interventional imposed PASS final study report assessment report Cyproterone/ethinylestradiol Active substance:
1 December 2016 EMA/101726/2017 Pharmacovigilance Risk Assessment Committee (PRAC) PRAC non-interventional imposed PASS final study report assessment report Cyproterone/ethinylestradiol Active substance:
Updates on take-home naloxone in France Anne-Claire BRISACIER OFDT
 Updates on take-home naloxone in France Anne-Claire BRISACIER OFDT French Monitoring Center for Drugs and Drug addiction EMCDDA DRD national expert Meeting Lisbon 18-20 September 2017 Background 1977 MA
Updates on take-home naloxone in France Anne-Claire BRISACIER OFDT French Monitoring Center for Drugs and Drug addiction EMCDDA DRD national expert Meeting Lisbon 18-20 September 2017 Background 1977 MA
DECLARATION OF CONFORMITY To Council Directive 93/42/EEC of 14 June 1993
 DECLARATION OF CONFORMITY To Council Directive 93/42/EEC of 14 June 1993 Manufacturer: EC Authorized Representative: T.A.G. Medical Products Corporation LTD. Kibbutz Gaaton 2513000 Israel Tel: (972) 4
DECLARATION OF CONFORMITY To Council Directive 93/42/EEC of 14 June 1993 Manufacturer: EC Authorized Representative: T.A.G. Medical Products Corporation LTD. Kibbutz Gaaton 2513000 Israel Tel: (972) 4
Pulpdent Corporation Revision Date: January 1, 2014
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Kool-Dam Heatless Liquid Dam & Block Out Resin 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Kool-Dam Heatless Liquid Dam & Block Out Resin 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for
O(K) SUMMARY. Mega'Gen Co., Ltd , Eupchun-Ri, Jain-Myun. Gyeongsan, Gyeongbuk South Korea Phone: , Fax:
 EZ PLUS IMPLANT SYSTEM 13. 510O(K) SUMMARY Mega'Gen Co., Ltd. 114-8, Eupchun-Ri, Jain-Myun. Gyeongsan, Gyeongbuk South Korea Phone: 82-53-857-5770, Fax: 82-53-857-5432 510(K) Summary 510O(K) SUMMARY AND
EZ PLUS IMPLANT SYSTEM 13. 510O(K) SUMMARY Mega'Gen Co., Ltd. 114-8, Eupchun-Ri, Jain-Myun. Gyeongsan, Gyeongbuk South Korea Phone: 82-53-857-5770, Fax: 82-53-857-5432 510(K) Summary 510O(K) SUMMARY AND
Pulpdent Corporation Revision Date: May 7, 2014
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation 1.2 1.2.2 Application / Use SIC 1.2.3 Use Category 1.3 Manufacturer Pulpdent Corporation 80 Oakland Street, P.O. Box 780
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation 1.2 1.2.2 Application / Use SIC 1.2.3 Use Category 1.3 Manufacturer Pulpdent Corporation 80 Oakland Street, P.O. Box 780
BS EN ISO 14971: Risk Analysis and Risk Management Report
 BLACKHORSE ROAD, LETCHWORTH, HERTS, SG6 1HB, ENGLAND Tel: + 44 (0) 1462 686221 www.stoddard.co.uk Fax: + 44 (0) 1462 480711 BS EN ISO 14971: Risk Analysis and Risk Management Report Product: Gates and
BLACKHORSE ROAD, LETCHWORTH, HERTS, SG6 1HB, ENGLAND Tel: + 44 (0) 1462 686221 www.stoddard.co.uk Fax: + 44 (0) 1462 480711 BS EN ISO 14971: Risk Analysis and Risk Management Report Product: Gates and
Pulpdent Corporation Revision Date: May 1, 2017
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Tuff-Temp Plus Provisional Veneer, Crown & Bridge Resin 1.2 1.2.2 1.2.3 Application / Use SIC Use Category 1.3 Manufacturer
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Tuff-Temp Plus Provisional Veneer, Crown & Bridge Resin 1.2 1.2.2 1.2.3 Application / Use SIC Use Category 1.3 Manufacturer
Pulpdent Corporation Revision Date: May 1, 2017
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Ortho-Choice Ortho-Coat 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for use by dental professional
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Ortho-Choice Ortho-Coat 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for use by dental professional
Draft Guidance for Industry and FDA Staff
 Draft Guidance for Industry and FDA Staff Submission and Review of Sterility Information in Premarket Notification (510(k)) Submissions for Devices Labeled as Sterile DRAFT GUIDANCE This guidance document
Draft Guidance for Industry and FDA Staff Submission and Review of Sterility Information in Premarket Notification (510(k)) Submissions for Devices Labeled as Sterile DRAFT GUIDANCE This guidance document
BS EN ISO 14971: Risk Analysis and Risk Management Report
 BLACKHORSE ROAD, LETCHWORTH, HERTS, SG6 1HB, ENGLAND Tel: + 44 (0) 1462 686221 www.stoddard.co.uk Fax: + 44 (0) 1462 480711 BS EN ISO 14971: Risk Analysis and Risk Management Report Product: Maxflex Pop
BLACKHORSE ROAD, LETCHWORTH, HERTS, SG6 1HB, ENGLAND Tel: + 44 (0) 1462 686221 www.stoddard.co.uk Fax: + 44 (0) 1462 480711 BS EN ISO 14971: Risk Analysis and Risk Management Report Product: Maxflex Pop
BS EN ISO 14971: Risk Analysis and Risk Management Report
 BLACKHORSE ROAD, LETCHWORTH, HERTS, SG6 1HB, ENGLAND Tel: + 44 (0) 1462 686221 www.stoddard.co.uk Fax: + 44 (0) 1462 480711 BS EN ISO 14971: Risk Analysis and Risk Management Report Product: Tungsten Carbide
BLACKHORSE ROAD, LETCHWORTH, HERTS, SG6 1HB, ENGLAND Tel: + 44 (0) 1462 686221 www.stoddard.co.uk Fax: + 44 (0) 1462 480711 BS EN ISO 14971: Risk Analysis and Risk Management Report Product: Tungsten Carbide
ko~ Tk (K) SUMMARY
 ko~3367-3 13. 510(K) SUMMARY 7-Tk 1 92006 Mega'Gen Co., Ltd. 114-8, Eupchun-Ri, Jain-Myun, Gyeongsan, Gyeongbuk South Korea Phone: 82-53-857-5770, Fax: 82-53-857-5432 510(K) Summary 510O(K) SUMMARY AND
ko~3367-3 13. 510(K) SUMMARY 7-Tk 1 92006 Mega'Gen Co., Ltd. 114-8, Eupchun-Ri, Jain-Myun, Gyeongsan, Gyeongbuk South Korea Phone: 82-53-857-5770, Fax: 82-53-857-5432 510(K) Summary 510O(K) SUMMARY AND
ต วอย าง AF เคร องม อทางการแพทย ท จะศ กษาท ม ความเส ยงน อยและความเส ยงมาก
 EXAMPLES: ต วอย าง เคร องม อทางการแพทย ท จะศ กษาท ม ความเส ยงน อยและความเส ยงมาก ต วอย างเคร องม อทางการแพทย ท จะศ กษาท ม ความเส ยงน อย NON-SIGNIFICANT RISK DEVICE STUDIES Bio-stimulation Lasers for treatment
EXAMPLES: ต วอย าง เคร องม อทางการแพทย ท จะศ กษาท ม ความเส ยงน อยและความเส ยงมาก ต วอย างเคร องม อทางการแพทย ท จะศ กษาท ม ความเส ยงน อย NON-SIGNIFICANT RISK DEVICE STUDIES Bio-stimulation Lasers for treatment
Pulpdent Corporation Revision Date: January 1, 2014
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Lime-Lite Light-cured Cavity Liner 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for use by dental
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Lime-Lite Light-cured Cavity Liner 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for use by dental
Pulpdent Corporation Revision Date: May 1, 2017 Safety Data Sheet
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Etch-All 10% Phosphoric Acid Etching Gel 1.2 1.2.2 1.2.3 Application / Use SIC Use Category 1.3 Manufacturer Pulpdent
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Etch-All 10% Phosphoric Acid Etching Gel 1.2 1.2.2 1.2.3 Application / Use SIC Use Category 1.3 Manufacturer Pulpdent
Transparency, Equity, Ethics: access to care. quality of life. Improving. and patients. An Agency of the French Ministry of Health
 An Agency of the French Ministry of Health Transparency, Equity, Ethics: Improving access to care and patients quality of life AgenceBiomedecine_Plaquette_GB.indd 1 25/08/14 12:15 Contents P.1 Agence de
An Agency of the French Ministry of Health Transparency, Equity, Ethics: Improving access to care and patients quality of life AgenceBiomedecine_Plaquette_GB.indd 1 25/08/14 12:15 Contents P.1 Agence de
Sterilization of health care products Radiation. Part 3: Guidance on dosimetric aspects of development, validation and routine control
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 11137-3 Second edition 2017-06 Sterilization of health care products Radiation Part 3: Guidance on dosimetric aspects of development, validation
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 11137-3 Second edition 2017-06 Sterilization of health care products Radiation Part 3: Guidance on dosimetric aspects of development, validation
Archives - Implementation of the GLP Directives in the European states
 Ref. Ares(2015)5384581-26/11/2015 EUROPEAN COMMISSION Directorate-General for Internal Market, Industry, Entrepreneurship and SMEs Consumer, Environmental and Health Technologies Chemicals Archives - Implementation
Ref. Ares(2015)5384581-26/11/2015 EUROPEAN COMMISSION Directorate-General for Internal Market, Industry, Entrepreneurship and SMEs Consumer, Environmental and Health Technologies Chemicals Archives - Implementation
Workshop on the reprocessing of medical devices. 5. December Brussels. Nikou Ghassemieh Managing Director
 Workshop on the reprocessing of medical devices 5. December 2008 - Brussels Nikou Ghassemieh Managing Director Established: 2003 Members from science, industry and medical practice Tasks and Goals: - Increasing
Workshop on the reprocessing of medical devices 5. December 2008 - Brussels Nikou Ghassemieh Managing Director Established: 2003 Members from science, industry and medical practice Tasks and Goals: - Increasing
EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL VOLUME 2C. Guidelines. Medicinal products for human use
 EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Brussels, Draft Revision 2 NOTICE TO APPLICANTS VOLUME 2C Guidelines Medicinal products for human use Safety, environment and information
EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Brussels, Draft Revision 2 NOTICE TO APPLICANTS VOLUME 2C Guidelines Medicinal products for human use Safety, environment and information
Single market, regulatory environment, industries under vertical legislation Pharmaceuticals : regulatory framework and market authorisations
 EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single market, regulatory environment, industries under vertical legislation Pharmaceuticals : regulatory framework and market authorisations Brussels,
EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single market, regulatory environment, industries under vertical legislation Pharmaceuticals : regulatory framework and market authorisations Brussels,
Pulpdent Corporation Revision Date: May 6, 2014 Safety Data Sheet
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Ortho-Choice OBA Bonding Resin 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for use by dental professional
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Ortho-Choice OBA Bonding Resin 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for use by dental professional
INDEPENDANT CONSULTING FOR QUALITY AND REGULATORY AFFAIRS EXPERTS FOR MEDICAL DEVICES REFERENCES. DATE POSITION ACTIVITIES MEDICAL DEVICES 2009 to NOW
 INDEPENDANT CONSULTING FOR QUALITY AND EXPERTS FOR MEDICAL DEVICES REFERENCES OUTSOURCED SERVICES DATE POSITION ACTIVITIES MEDICAL DEVICES 2009 to NOW QUALITY AND MANAGER company which designs and manufactures
INDEPENDANT CONSULTING FOR QUALITY AND EXPERTS FOR MEDICAL DEVICES REFERENCES OUTSOURCED SERVICES DATE POSITION ACTIVITIES MEDICAL DEVICES 2009 to NOW QUALITY AND MANAGER company which designs and manufactures
PRAC non-interventional imposed PASS final study report assessment report
 1 December 2016 EMA/101714/2017 Pharmacovigilance Risk Assessment Committee (PRAC) PRAC non-interventional imposed PASS final study report assessment report Active substance: cyproterone/ethinylestradiol
1 December 2016 EMA/101714/2017 Pharmacovigilance Risk Assessment Committee (PRAC) PRAC non-interventional imposed PASS final study report assessment report Active substance: cyproterone/ethinylestradiol
Preparation Instructions for the CAMLOG /CONELOG Implant System
 J8000.0032 Rev.6 EN 06/2015 Preparation Instructions for the CAMLOG /CONELOG Implant System ENGLISH The following descriptions contain detailed instructions on cleaning, disinfection and sterilization
J8000.0032 Rev.6 EN 06/2015 Preparation Instructions for the CAMLOG /CONELOG Implant System ENGLISH The following descriptions contain detailed instructions on cleaning, disinfection and sterilization
: GLYCEROL FORMAL CRS
 in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 27/06/2013 Revision date: 27/06/2013 Supersedes: 14/12/2011 Version: 1.0 SECTION 1: Identification of the substance/mixture
in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 27/06/2013 Revision date: 27/06/2013 Supersedes: 14/12/2011 Version: 1.0 SECTION 1: Identification of the substance/mixture
ISO INTERNATIONAL STANDARD. Implants for surgery Active implantable medical devices Part 2: Cardiac pacemakers
 INTERNATIONAL STANDARD ISO 14708-2 Second edition 2012-08-15 Implants for surgery Active implantable medical devices Part 2: Cardiac pacemakers Implants chirurgicaux Dispositifs médicaux implantables actifs
INTERNATIONAL STANDARD ISO 14708-2 Second edition 2012-08-15 Implants for surgery Active implantable medical devices Part 2: Cardiac pacemakers Implants chirurgicaux Dispositifs médicaux implantables actifs
Facility Information for Initial Assessment
 DIAGNOSTIC ACCREDITATION PROGRAM 300 669 Howe Street Telephone: 604-733-7758 Vancouver BC V6C 0B4 Toll Free: 1-800-461-3008 (in BC) www.cpsbc.ca Fax: 604-733-3503 Facility Information for Initial Assessment
DIAGNOSTIC ACCREDITATION PROGRAM 300 669 Howe Street Telephone: 604-733-7758 Vancouver BC V6C 0B4 Toll Free: 1-800-461-3008 (in BC) www.cpsbc.ca Fax: 604-733-3503 Facility Information for Initial Assessment
GUIDELINE ON THE CATEGORISATION OF EXTENSION APPLICATIONS (EA) versus VARIATIONS APPLICATIONS (V) OCTOBER 2003
 EUROPN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single market, implementation and legislation for consumer goods Pharmaceuticals : regulary framework and market authorisations Brussels, F2/AW D(2002)
EUROPN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single market, implementation and legislation for consumer goods Pharmaceuticals : regulary framework and market authorisations Brussels, F2/AW D(2002)
Université de Moncton, Edmundston Campus, full-time students. Maliseet First Nations Health Centre, Madawaska
 3.1.4 Health Zone 4 - SERVICE Cervical Screening LOCATION Université de Moncton, Campus, full-time students Maliseet First Nations Health Centre, Madawaska CONTACT COORDINATES Phone: 506-737-5295 Phone:
3.1.4 Health Zone 4 - SERVICE Cervical Screening LOCATION Université de Moncton, Campus, full-time students Maliseet First Nations Health Centre, Madawaska CONTACT COORDINATES Phone: 506-737-5295 Phone:
Provläsningsexemplar / Preview. First edition
 Provläsningsexemplar / Preview TECHNICAL SPECIFICATION ISO/TS 19930 First edition 2017-12 Guidance on aspects of a risk-based approach to assuring sterility of terminally sterilized, single-use health
Provläsningsexemplar / Preview TECHNICAL SPECIFICATION ISO/TS 19930 First edition 2017-12 Guidance on aspects of a risk-based approach to assuring sterility of terminally sterilized, single-use health
EC Declaration of Conformity
 EC Declaration of Conformity MegaGen Implant Co., Ltd. 377-2, Gyochon-ri, Jain-myeon, Gyeongsan-si, Gyeongbuk, Korea declare that the medical devices described hereafter The Dental Implant Superstructure
EC Declaration of Conformity MegaGen Implant Co., Ltd. 377-2, Gyochon-ri, Jain-myeon, Gyeongsan-si, Gyeongbuk, Korea declare that the medical devices described hereafter The Dental Implant Superstructure
Electrodes (for TransAeris System)
 Surgical Procedures for Implantation of TransLoc Electrodes (for TransAeris System) 1 R L 1 TransAeris System TransLoc Electrode Kit 3 3 TransAeris Patient Kit (FrictionLoc) TransAeris Patient Kit (TransAeris
Surgical Procedures for Implantation of TransLoc Electrodes (for TransAeris System) 1 R L 1 TransAeris System TransLoc Electrode Kit 3 3 TransAeris Patient Kit (FrictionLoc) TransAeris Patient Kit (TransAeris
Delayed hemolytic transfusion reaction in the French hemovigilance system
 Delayed hemolytic transfusion reaction in the French hemovigilance system C. Rieux, G. Brittenham, D. Bachir, E. De Meyer, K. Boudjedir for the French hemovigilance network First Seminar on delayed hemolytic
Delayed hemolytic transfusion reaction in the French hemovigilance system C. Rieux, G. Brittenham, D. Bachir, E. De Meyer, K. Boudjedir for the French hemovigilance network First Seminar on delayed hemolytic
This document is a preview generated by EVS
 EESTI STANDARD EVS-EN 1641:2004 Stomatoloogia. Meditsiinivahendid stomatoloogias. Materjalid Dentistry - Medical devices for dentistry - Materials EESTI STANDARDIKESKUS EESTI STANDARDI EESSÕNA NATIONAL
EESTI STANDARD EVS-EN 1641:2004 Stomatoloogia. Meditsiinivahendid stomatoloogias. Materjalid Dentistry - Medical devices for dentistry - Materials EESTI STANDARDIKESKUS EESTI STANDARDI EESSÕNA NATIONAL
not Registered with the Ministry of Justice of the Russian Federation dated July 9, 2012, No MINISTRY OF HEALTH OF THE RUSSIAN FEDERATION ORDER
 not Registered with the Ministry of Justice of the Russian Federation dated July 9, 2012, No. 24852 MINISTRY OF HEALTH OF THE RUSSIAN FEDERATION ORDER dated June 6, 2012, No. 4n ON APPROVAL OF THE MEDICAL
not Registered with the Ministry of Justice of the Russian Federation dated July 9, 2012, No. 24852 MINISTRY OF HEALTH OF THE RUSSIAN FEDERATION ORDER dated June 6, 2012, No. 4n ON APPROVAL OF THE MEDICAL
Pulpdent Corporation Revision Date: May 1, 2017 Safety Data Sheet
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Ortho-Choice Orthodontic Bracket Adhesive (OBA) 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Ortho-Choice Orthodontic Bracket Adhesive (OBA) 1.2 1.2.2 1.2.3 Application / Use SIC Use Category Dental material for
Compressor Nebulizer. Model UN-014
 Compressor Nebulizer Model UN-014 Instruction Manual Original Manuel d instructions Traduction Manual de Instrucciones Traducción Manuale di Istruzioni Traduzione ITALIANO ESPAÑOL FRANÇAIS ENGLISH 1WMPD4003398
Compressor Nebulizer Model UN-014 Instruction Manual Original Manuel d instructions Traduction Manual de Instrucciones Traducción Manuale di Istruzioni Traduzione ITALIANO ESPAÑOL FRANÇAIS ENGLISH 1WMPD4003398
Sterilization of health care products Radiation. Part 2: Establishing the sterilization dose
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 11137-2 Third edition 2013-06-01 Sterilization of health care products Radiation Part 2: Establishing the sterilization dose Stérilisation des
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 11137-2 Third edition 2013-06-01 Sterilization of health care products Radiation Part 2: Establishing the sterilization dose Stérilisation des
: Exposure may produce an allergic reaction.
 Date of issue: 27/06/2013 Revision date: 03/12/2014 Supersedes: 16/12/2009 Version: 6.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product
Date of issue: 27/06/2013 Revision date: 03/12/2014 Supersedes: 16/12/2009 Version: 6.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product
Daily use of an artificial intelligence by caregivers Prediction and personalization of the expected information to impact patients care
 Daily use of an artificial intelligence by caregivers Prediction and personalization of the expected information to impact patients care Agenda 1. Drugs don t kill, bad usage does 2. Peut-on demander le
Daily use of an artificial intelligence by caregivers Prediction and personalization of the expected information to impact patients care Agenda 1. Drugs don t kill, bad usage does 2. Peut-on demander le
QA Regulatory forum: Classification and Risk assessment according to MDR Date : T h e r e s e. A l b i n s s o m e d q t e c h.
 QA Regulatory forum: Classification and Risk assessment according to MDR Date : 05.04.2018 THERESE ALBINSSON T h e r e s e. A l b i n s s o n @ m e d q t e c h. s e MedQtech AB MedQtech AB is a consultant
QA Regulatory forum: Classification and Risk assessment according to MDR Date : 05.04.2018 THERESE ALBINSSON T h e r e s e. A l b i n s s o n @ m e d q t e c h. s e MedQtech AB MedQtech AB is a consultant
Knee Arthroscopy. Anatomy
 Knee Arthroscopy Knee arthroscopy is a surgical procedure that allows doctors to view the knee joint without making a large incision (cut) through the skin and other soft tissues. Arthroscopy is used to
Knee Arthroscopy Knee arthroscopy is a surgical procedure that allows doctors to view the knee joint without making a large incision (cut) through the skin and other soft tissues. Arthroscopy is used to
Safety Data Sheet Safety Data Sheet in accordance with regulation (EC) No 1907/2006, as amended. Date of issue: 31/05/2017 Version: 1.
 in accordance with regulation (EC) No 1907/2006, as amended. Date of issue: 31/05/2017 Version: 1.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier
in accordance with regulation (EC) No 1907/2006, as amended. Date of issue: 31/05/2017 Version: 1.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier
5101: of 5 APPENDIX B. Revenue Center Codes Requiring CPT or HCPCS Coding
 ACTION: Final ENACTED Appendix 5101:3-2-21 DATE: 03/17/2011 1:51 PM 5101:3-2-21 1 of 5 IV Therapy 0260 General Classification 0261 Infusion Pump 0269 Other IV Therapy Oncology 0280 General Classification
ACTION: Final ENACTED Appendix 5101:3-2-21 DATE: 03/17/2011 1:51 PM 5101:3-2-21 1 of 5 IV Therapy 0260 General Classification 0261 Infusion Pump 0269 Other IV Therapy Oncology 0280 General Classification
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Derby Manor Derby Road Bournemouth Dorset BH1 3QB Contact: Mr D Riggs Tel: +44 (0)1202 552 153 Fax: +44 (0)1202 290 716 E-Mail: info@urs-certifcation.co.uk
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Derby Manor Derby Road Bournemouth Dorset BH1 3QB Contact: Mr D Riggs Tel: +44 (0)1202 552 153 Fax: +44 (0)1202 290 716 E-Mail: info@urs-certifcation.co.uk
Kidner procedure. For the treatment of accessory navicular. Information for patients Department of Podiatric Surgery
 Kidner procedure For the treatment of accessory navicular Information for patients Department of Podiatric Surgery What is an accessory? An accessory is an additional /bony segment which is not usually
Kidner procedure For the treatment of accessory navicular Information for patients Department of Podiatric Surgery What is an accessory? An accessory is an additional /bony segment which is not usually
Safety Data Sheet Safety Data Sheet in accordance with regulation (EC) No 1907/2006, as amended. Date of issue: 31/05/2017 Version: 1.
 Date of issue: 31/05/2017 Version: 1.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product form Substance name : Substance : Salicylic acid
Date of issue: 31/05/2017 Version: 1.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product form Substance name : Substance : Salicylic acid
Please find enclosed Dosimetry Service Licence No which replaces Dosimetry Service Licence No
 Directorate of Nuclear Substance Regulation Telephone: 1 (888) 229-2672 April 12, 2017 Licence Number Christopher Passmore Landauer Inc. 2 Science Road Glenwood, IL 60425-1586 Subject: Dosimetry Service
Directorate of Nuclear Substance Regulation Telephone: 1 (888) 229-2672 April 12, 2017 Licence Number Christopher Passmore Landauer Inc. 2 Science Road Glenwood, IL 60425-1586 Subject: Dosimetry Service
SAFETY DATA SHEET. according to 1907/2006/EC, Article 31. Water Based Wet And Reveal Spray Ink
 SAFETY DATA SHEET according to 1907/2006/EC, Article 31 Page 1/5 Revision 3 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product name 1.2. Relevant
SAFETY DATA SHEET according to 1907/2006/EC, Article 31 Page 1/5 Revision 3 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product name 1.2. Relevant
Safety Data Sheet Safety Data Sheet in accordance with regulation (EC) No 1907/2006, as amended. Date of issue: 31/05/2017 Version: 1.
 in accordance with regulation (EC) No 1907/2006, as amended. Date of issue: 31/05/2017 Version: 1.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier
in accordance with regulation (EC) No 1907/2006, as amended. Date of issue: 31/05/2017 Version: 1.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier
REVENUE CODE LIST REQUIRING CPT/HCPCS CODES FOR OUTPATIENT FACILITY CLAIMS
 REVENUE CODE LIST REQUIRING CPT/HCPCS CODES FOR OUTPATIENT FACILITY CLAIMS For Providers Effective July 15, 2018 Revenue Code Description 240 All inclusive ancillary, general 250 Pharmacy 251 Drugs, generic
REVENUE CODE LIST REQUIRING CPT/HCPCS CODES FOR OUTPATIENT FACILITY CLAIMS For Providers Effective July 15, 2018 Revenue Code Description 240 All inclusive ancillary, general 250 Pharmacy 251 Drugs, generic
Martin Bullemer Key Account Manager Medical 2008 April 16th
 Medical Device e-manufacturing Targets Martin Bullemer Key Account Manager Medical 2008 April 16th The Medical Market for Laser Sintering Every human is different, which results in a wide variety of different
Medical Device e-manufacturing Targets Martin Bullemer Key Account Manager Medical 2008 April 16th The Medical Market for Laser Sintering Every human is different, which results in a wide variety of different
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 17510 First edition 2015-08-01 Medical devices Sleep apnoea breathing therapy Masks and application accessories Dispositifs médicaux Thérapie respiratoire de l apnée du sommeil
INTERNATIONAL STANDARD ISO 17510 First edition 2015-08-01 Medical devices Sleep apnoea breathing therapy Masks and application accessories Dispositifs médicaux Thérapie respiratoire de l apnée du sommeil
GUIDANCE ON SUBMISSIONS FOR SAFETY EVALUATION OF SOURCES OF NUTRIENTS OR OF OTHER INGREDIENTS PROPOSED FOR USE IN THE MANUFACTURE OF FOODS
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C2 - Management of scientific committee; scientific co-operation and networks Scientific Committee
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C2 - Management of scientific committee; scientific co-operation and networks Scientific Committee
KEY DIFFERENCES BETWEEN THE NEW MEDICAL DEVICE AND THE OTC REGULATIONS IN EUROPE
 RNI Conseil 2017 Tous droits réservés Toute reproduction interdite sans l'autorisation de l'auteur. KEY DIFFERENCES BETWEEN THE NEW MEDICAL DEVICE AND THE OTC REGULATIONS IN EUROPE Anne Laure TARDY, PhD
RNI Conseil 2017 Tous droits réservés Toute reproduction interdite sans l'autorisation de l'auteur. KEY DIFFERENCES BETWEEN THE NEW MEDICAL DEVICE AND THE OTC REGULATIONS IN EUROPE Anne Laure TARDY, PhD
: BENZYL ALCOHOL CRS
 in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 27/06/2013 Revision date: 27/06/2013 Supersedes: 04/02/2010 Version: 5.0 SECTION 1: Identification of the substance/mixture
in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 27/06/2013 Revision date: 27/06/2013 Supersedes: 04/02/2010 Version: 5.0 SECTION 1: Identification of the substance/mixture
Please find below the Commission Communication and the Commission Recommendation for the substance zinc sulphate
 Please find below the Commission Communication and the Commission Recommendation for the substance zinc sulphate CAS No: 7733-02-0 EINECS No: 231-793-3 20.6.2008 EN C 155/1 II (Information) INFORMATION
Please find below the Commission Communication and the Commission Recommendation for the substance zinc sulphate CAS No: 7733-02-0 EINECS No: 231-793-3 20.6.2008 EN C 155/1 II (Information) INFORMATION
Trust Policy 218 Ionising Radiation Safety Policy
 Trust Policy 218 Ionising Radiation Safety Policy Purpose Date Version August 2016 7 To ensure that Plymouth Hospitals NHS Trust complies with all relevant legislation with regard to the use of ionising
Trust Policy 218 Ionising Radiation Safety Policy Purpose Date Version August 2016 7 To ensure that Plymouth Hospitals NHS Trust complies with all relevant legislation with regard to the use of ionising
Guidance for Industry
 MEDICAL DEVICES SECTOR Guidance for Industry Format and Content of Proposed Bundling/Grouping Criteria for Medical Devices DRAFT GUIDANCE This guidance document is being distributed for comment purposes
MEDICAL DEVICES SECTOR Guidance for Industry Format and Content of Proposed Bundling/Grouping Criteria for Medical Devices DRAFT GUIDANCE This guidance document is being distributed for comment purposes
Implants for surgery Active implantable medical devices. Part 7: Particular requirements for cochlear implant systems
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 14708-7 First edition 2013-01-15 Implants for surgery Active implantable medical devices Part 7: Particular requirements for cochlear implant systems
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 14708-7 First edition 2013-01-15 Implants for surgery Active implantable medical devices Part 7: Particular requirements for cochlear implant systems
Peninsula Dental Social Enterprise (PDSE)
 Peninsula Dental Social Enterprise (PDSE) Decontamination - Cleaning and Disinfection Version 3.0 Date approved: November 2016 Approved by: The Board Review due: November 2019 Policy will be updated as
Peninsula Dental Social Enterprise (PDSE) Decontamination - Cleaning and Disinfection Version 3.0 Date approved: November 2016 Approved by: The Board Review due: November 2019 Policy will be updated as
Emergency Care Progress Log
 Emergency Care Progress Log For further details on the National Occupational Competencies for EMRs, please visit www.paramedic.ca. Check off each skill once successfully demonstrated the Instructor. All
Emergency Care Progress Log For further details on the National Occupational Competencies for EMRs, please visit www.paramedic.ca. Check off each skill once successfully demonstrated the Instructor. All
Chemical CAS No. EINECS Registration % Classification Classification No. Number Conc 1999/45/EC 1272/2008
 AUTO DISHWASHER TABLETS 1 SAFETY DATA SHEET Safety data sheet in accordance with Commission Regulation (EU) No. 453/2010 of 20th May 2010 of the European parliament and of the Council on the Registration,
AUTO DISHWASHER TABLETS 1 SAFETY DATA SHEET Safety data sheet in accordance with Commission Regulation (EU) No. 453/2010 of 20th May 2010 of the European parliament and of the Council on the Registration,
BSD-500 Hyperthermia System
 BSD-500 Hyperthermia System What are you doing for your recurrent cancer patients? BSD-500 No shielding room needed Portable FDA Approved for pallative treatment of certain tumors Delivers both superficial
BSD-500 Hyperthermia System What are you doing for your recurrent cancer patients? BSD-500 No shielding room needed Portable FDA Approved for pallative treatment of certain tumors Delivers both superficial
: SALBUTAMOL IMPURITY B CRS
 in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 30/08/2013 Revision date: 30/08/2013 Supersedes: 27/06/2013 Version: 4.1 SECTION 1: Identification of the substance/mixture
in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 30/08/2013 Revision date: 30/08/2013 Supersedes: 27/06/2013 Version: 4.1 SECTION 1: Identification of the substance/mixture
DIRECT ACCESS Guidance to BSDHT Members
 DIRECT CCESS Guidance to BSDHT Members Direct ccess comes into effect from 1 May 2013. But what does it mean for dental hygienists and dental therapists? The GDC have published guidance notes on the subject
DIRECT CCESS Guidance to BSDHT Members Direct ccess comes into effect from 1 May 2013. But what does it mean for dental hygienists and dental therapists? The GDC have published guidance notes on the subject
ESEMeD/MHEDEA The European Study of the Epidemiology of Mental Disorders/Mental Health Disability : a
 ESEMeD/MHEDEA 2000 - The European Study of the Epidemiology of Mental Disorders/Mental Health Disability : a European Assessment in the year 2000 (ESEMeD/MHEDEA 2000) Head : Kovess-Masfety Viviane, INSERM
ESEMeD/MHEDEA 2000 - The European Study of the Epidemiology of Mental Disorders/Mental Health Disability : a European Assessment in the year 2000 (ESEMeD/MHEDEA 2000) Head : Kovess-Masfety Viviane, INSERM
SAFETY DATA SHEET according to Regulation (EC) No. 453/2010 Dental Use
 Page 1 of 8 SECTION 1 : Identification of the substance/preparation and of the company/undertaking 1.1. Product identifier Product code Product name Product description : PK/63400 : : Dental bonding resin
Page 1 of 8 SECTION 1 : Identification of the substance/preparation and of the company/undertaking 1.1. Product identifier Product code Product name Product description : PK/63400 : : Dental bonding resin
On the use of Amalgam for dental fillings in Sweden
 On the use of Amalgam for dental fillings in Sweden Director Medical Devices Medical Products Agency Uppsala, Sweden Content of this presentation Short regulatory introduction Current use of Amalgams in
On the use of Amalgam for dental fillings in Sweden Director Medical Devices Medical Products Agency Uppsala, Sweden Content of this presentation Short regulatory introduction Current use of Amalgams in
Catalogue. Medication Safety Labels CONTENTS 1. For the Safe Management of Medication Version 2. Anaesthetic Syringe Labels
 Medication Safety Labels Catalogue For the Safe Management of Medication Version 2 CONTENTS 1 4 8 11 12 Sterile Labels Injectable Medicines, Fluids & Line Labels Pre-printed Medicine Line Labels Anaesthetic
Medication Safety Labels Catalogue For the Safe Management of Medication Version 2 CONTENTS 1 4 8 11 12 Sterile Labels Injectable Medicines, Fluids & Line Labels Pre-printed Medicine Line Labels Anaesthetic
KNEE LIGAMENT RECONSTRUCTION
 PATIENT INFORMATION SHEET YOU ARE GOING TO UNDERGO KNEE LIGAMENT RECONSTRUCTION and sports traumatology ORTHOPAEDIC SURGERY Doctor Philippe Paillard Office YOU HAVE A RUPTURED ANTERIOR CRUCIATE LIGAMENT
PATIENT INFORMATION SHEET YOU ARE GOING TO UNDERGO KNEE LIGAMENT RECONSTRUCTION and sports traumatology ORTHOPAEDIC SURGERY Doctor Philippe Paillard Office YOU HAVE A RUPTURED ANTERIOR CRUCIATE LIGAMENT
Pulpdent Corporation Revision Date: May 21, 2014 Safety Data Sheet. 1.1 Commercial product name / designation Root Canal Sealer Powder and Liquid
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Root Canal Sealer Powder and Liquid 1. 1.. 1..3 Application / Use SIC Use Category Dental material for use by dental professional
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Root Canal Sealer Powder and Liquid 1. 1.. 1..3 Application / Use SIC Use Category Dental material for use by dental professional
Schedule of Benefits PPO MASSACHUSETTS
 Schedule of s PPO MASSACHUSETTS ID: MD0000017711_A5 X This Schedule of s states any Limits and the amounts you must pay for Covered s. However, it is only a summary of your benefits. Please see your Handbook
Schedule of s PPO MASSACHUSETTS ID: MD0000017711_A5 X This Schedule of s states any Limits and the amounts you must pay for Covered s. However, it is only a summary of your benefits. Please see your Handbook
PRODUCT INFORMATION SHEET
 PRODUCT INFORMATION SHEET FEATURES & APPLICATIONS FOOD SAFE SANITISER Unico Limited, North Main Street, Carronshore, Falkirk. FK2 8HT Tel: 0845 27 86426 Fax: 01324 573401 Unico Food Safe Sanitiser is a
PRODUCT INFORMATION SHEET FEATURES & APPLICATIONS FOOD SAFE SANITISER Unico Limited, North Main Street, Carronshore, Falkirk. FK2 8HT Tel: 0845 27 86426 Fax: 01324 573401 Unico Food Safe Sanitiser is a
Medical Information Form
 Fill in form with a computer Medical Information Form Child s Name: first name, last name Date of Birth: month, day, year Health Insurance information o Insurance group number, policy number, contact information:
Fill in form with a computer Medical Information Form Child s Name: first name, last name Date of Birth: month, day, year Health Insurance information o Insurance group number, policy number, contact information:
INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust)
 A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC17 SINGLE USE AND SINGLE PATIENT USE MEDICAL
A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC17 SINGLE USE AND SINGLE PATIENT USE MEDICAL
MATERIAL SAFETY DATA SHEET In accordance with CE Regulation n 1907/2006. Annex II.
 Page: 1 of 7 RRR1. Identification of preparation and of the company/undertaking 1.1 Product name: Product code: 62AM4PEB camp Gs Dynamic kit 1,000 tests 1.2 Use With the exception of products specifically
Page: 1 of 7 RRR1. Identification of preparation and of the company/undertaking 1.1 Product name: Product code: 62AM4PEB camp Gs Dynamic kit 1,000 tests 1.2 Use With the exception of products specifically
MATERIAL SAFETY DATA SHEET In accordance with CE Regulation n 1907/2006. Annex II.
 Ref.: BA/909 HGHRIACT Page: 1 of 5 RRR1. Identification of preparation and of the company/undertaking 1.1 Product name: HGHRIACT 1.2 Use With the exception of products specifically labeled for In Vitro
Ref.: BA/909 HGHRIACT Page: 1 of 5 RRR1. Identification of preparation and of the company/undertaking 1.1 Product name: HGHRIACT 1.2 Use With the exception of products specifically labeled for In Vitro
Supporting positive clinical outcomes. Hospital and patient care product offering
 healthcare Supporting positive clinical outcomes Hospital and patient care product offering Positively impacting the health of your patients and your organisation Thermo Fisher Scientifi c s extensive
healthcare Supporting positive clinical outcomes Hospital and patient care product offering Positively impacting the health of your patients and your organisation Thermo Fisher Scientifi c s extensive
: POVIDONE IODINATED CRS
 in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 27/06/2013 Revision date: 27/06/2013 Supersedes: 04/02/2010 Version: 6.0 SECTION 1: Identification of the substance/mixture
in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 27/06/2013 Revision date: 27/06/2013 Supersedes: 04/02/2010 Version: 6.0 SECTION 1: Identification of the substance/mixture
Use of Standards in Substantial Equivalence Determinations
 Guidance for Industry and for FDA Staff Use of Standards in Substantial Equivalence Determinations Document issued on: March 12, 2000 U.S. Department Of Health And Human Services Food and Drug Administration
Guidance for Industry and for FDA Staff Use of Standards in Substantial Equivalence Determinations Document issued on: March 12, 2000 U.S. Department Of Health And Human Services Food and Drug Administration
Extra-corporeal Shock Wave Therapy (ESWT) for non-insertional Achilles tendinopathy Inflamed Achilles tendon
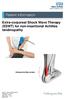 Extra-corporeal Shock Wave Therapy (ESWT) for non-insertional Achilles tendinopathy Inflamed Achilles tendon Source: Trauma & Orthopaedics Reference No: 6459-1 Issue date: 1/2/19 Review date: 1/2/22 Page
Extra-corporeal Shock Wave Therapy (ESWT) for non-insertional Achilles tendinopathy Inflamed Achilles tendon Source: Trauma & Orthopaedics Reference No: 6459-1 Issue date: 1/2/19 Review date: 1/2/22 Page
SAFETY DATA SHEET Afinion Lipid Panel Control
 Afinion Lipid Panel Control Page 1 of 6 SAFETY DATA SHEET Afinion Lipid Panel Control SECTION 1: Identification of the substance/mixture and of the company/undertaking Date issued 22.06.2012 1.1. Product
Afinion Lipid Panel Control Page 1 of 6 SAFETY DATA SHEET Afinion Lipid Panel Control SECTION 1: Identification of the substance/mixture and of the company/undertaking Date issued 22.06.2012 1.1. Product
SAFETY DATA SHEET. according to 1907/2006/EC, Article 31. Reinforced ZOE Cement Liquid
 SAFETY DATA SHEET according to 907/2006/EC, Article 3 Page /5 date SECTION : Identification of the substance/mixture and of the company/undertaking.. Product identifier Product name.2. Relevant identified
SAFETY DATA SHEET according to 907/2006/EC, Article 3 Page /5 date SECTION : Identification of the substance/mixture and of the company/undertaking.. Product identifier Product name.2. Relevant identified
: VALSARTAN FOR SYSTEM SUITABILITY CRS
 in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 27/06/2013 Revision date: 27/06/2013 Supersedes: 27/11/2009 Version: 3.0 SECTION 1: Identification of the substance/mixture
in accordance with Regulation (EC) No. 1907/2006, as amended. Date of issue: 27/06/2013 Revision date: 27/06/2013 Supersedes: 27/11/2009 Version: 3.0 SECTION 1: Identification of the substance/mixture
