Myometrial Insertion of Essure Mirco-Insert
|
|
|
- Angelica Dean
- 5 years ago
- Views:
Transcription
1 Case Report Myometrial Insertion of Essure Mirco-Insert Jeffrey J Woo 1*, Barbara E Simpson 1, Dale W Stovall 2 1 Department of Obstetrics and Gynecology, Riverside Regional Medical Center, Virginia, USA 2 Department of Obstetrics and Gynecology, Methodist Dallas Medical Center, Dallas, USA Abstract Complications arising from hysteroscopically-placed intratubal micro-insert devices are uncommon. Partial and complete perforations occur in approximately 0.9% to 2.6% while movement of the micro-insert device within the abdomen is even more rare. In this case report, the initial placement of the micro-insert device was uncomplicated, but concerns of cornual or fallopian tube perforation were apparent on the confirmation hysterosalpingogram (HSG) when free tubal spill was appreciated. After a succession of hysteroscopic and laparoscopic evaluations, and a final specimen dissection, the micro-insert device was found penetrating superior to the tubal ostia and embedded in the myometrium. Keywords: Myometrium, Hysteroscopic sterilization, Hysteroscopic Essure insertion Accepted on October 13, 2017 Introduction Permanent sterilization by Essure (Conceptus Inc, San Carlos, CA) is currently the only Food and Drug Administration (FDA) approved in-office hysteroscopic sterilization procedure in the United States (US). Over 600,000 procedures have been performed worldwide [1-4]. An analysis of sterilization procedures performed between 2005 and 2012 found that 38% of all procedures in the US were performed hysteroscopically [5]. Hysteroscopic sterilization has the advantage of sterilization without an abdominal entry or the associated risks of an abdominal procedure. Additionally, hysteroscopic sterilization decreases procedure costs, operating times, and offers the option of placement in the ambulatory setting. Although the Essure device is a highly effective form of sterilization with a 5-year effectiveness rate of 99.83%, it has not been without controversy [4]. From the device s introduction in November 2002 until May 2015, the FDA received a total of 5093 reports related to the device which ranged from complaints such as pelvic pain (3353) and irregular menses (1408), to fatigue (966) and weight fluctuations (936) [6,7]. In September of 2015, the FDA assembled a meeting of the Obstetrics and Gynecology Devices Panel of the Medical Devices Advisory Committee to hear expert opinions as well as patients experiences regarding the benefits and risks of the device [6,7]. In February 2016, the FDA published a sample black box warning regarding post placement adverse events and risks [6,7]. Additionally, in cooperation with the manufacturer, the FDA called for and has now implemented both improved patient information and counseling materials and the accrual of additional post marketing safety-related data. The FDA has never sought to remove the device from the marketplace and has held a consistent stance that the benefits of the device outweigh its risks [6,7]. Our case report is in regards to a patient who complained of one month s duration of menorrhagia after an uncomplicated inoffice hysteroscopic Essure insertion. Free spill was apparent unilaterally on the confirmation HSG which was concerning for fallopian tube or cornual perforation. After a succession of hysteroscopic and laparoscopic evaluations, and a final specimen dissection, we found that the device had penetrated and embedded into more than 50% of the uterine myometrium without perforation through the serosa of the uterus. Case Presentation A healthy 44-year-old G3P2012 with satisfied fertility desired permanent sterilization after the delivery of her 2 nd child. Her medical history was pertinent for 2 vaginal deliveries, 1 early first trimester miscarriage, and 3 umbilical hernia repairs that included mesh placement and removal. She was otherwise healthy without any significant past medical history or current medications. Due to the patient s prior 3 umbilical hernia repairs, the decision was made to do an in-office Essure sterilization procedure. The patient received a deep muscular injection of Depo-Provera (medroxyprogesterone 150 mg/ml) (Pfizer Inc., New York, NY) three weeks prior to the procedure. The procedure was performed 8 weeks postpartum by a resident physician under the direct supervision of an attending physician. In addition, the device deployment was observed by a Bayer product representative to ensure proper use and placement of the device. One hour prior to the procedure, the patient was given a low-dose benzodiazpene (diazepam 10mg) for anxiety and a low-dose oral opiate (oxycodone hydrochloride/ acetaminophen 5 mg/325 mg). Twenty minutes prior to the procedure, the patient received an intramuscular injection of a low-dose nonsteroidal anti-inflammatory drug (ketorolac 30 mg) for analgesia. Two and a half milliliters of 1% lidocaine was injected in the peri-cervical area at the 12, 4, 7 and 10 o'clock positions to create a cervical block. On hysteroscopic evaluation, no gross abnormalities of the uterine cavity or bilateral tubal ostia were appreciated. The micro-insert device placement was uncomplicated and the insertion was performed without resistance. At the conclusion of the procedure, 3 to 4 coils were noted to be trailing from the ostia bilaterally. The 1
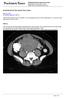
2 Citation: Woo JJ, Simpson BE, Stovall DW. Myometrial Insertion of Essure Mirco-Insert. Res Rep Gynaecol Obstet. 2017;1(4):1-5 patient tolerated the procedure well and was discharged home in good condition. Four months after the procedure, the patient had a routine confirmation HSG. Assessment of the HSG revealed that the left micro-insert appeared to be properly placed in the left fallopian tube (Figure 1). In contrast, the right micro-insert had an acute curvilinear appearance (Figure 1). As dye was injected, free spill was evident from the right side but the origin was unknown (Figure 2). The radiologist report stated the right fallopian tube was not occluded and the right micro-insert was not contained within the right fallopian tube. The patient was scheduled for counseling in regards to further assessment and management with presumption of a cornual or fallopian tube perforation. In an outpatient follow-up visit, the patient was advised to proceed with surgical management due to concerns for bowel injury due to the device s assumed peritoneal location. The patient reported that since her initial micro-insert placement she had vaginal bleeding for one month but no pelvic pain. The patient was consented for hysteroscopy, diagnostic laparoscopy, right salpingectomy, and possible cornual resection. Due to the patient s strong desire to remove the entire foreign body, she requested a total hysterectomy if needed. This was thoroughly discussed with her and she consented to a total laparoscopic hysterectomy. On the day of her procedure, initial hysteroscopic evaluation revealed trailing coils extending from the right tubal ostia (Figure 3). This was in contrast to the 3 to 4 trailing coils appreciated after the initial insertion. The left tubal ostia was clearly visualized with no micro-insert coils identified in the intrauterine cavity. Due to the length of the right-sided microinsert present within the uterine cavity, attempts were made to release and retrieve the micro-insert using hysteroscopic operative forceps. Resistance was appreciated and several attempts to retrieve the micro-insert only expanded the coils without release of the device. At this point a diagnostic laparoscopy was deemed appropriate to assess the pelvis for uterine or tubal injury and to identify both micro-inserts. A complete pelvic survey was performed and no uterine or tubal perforations were identified. A right salpingectomy was completed due to the patient s request of permanent sterilization and the fallopian tube was examined with no luminal position of the micro-insert. The right cornua was thoroughly evaluated and there was no sign of the microinsert. Due to uncertainty of the micro-insert site, a second hysteroscopic evaluation was performed. Several attempts to remove the micro-insert from the right side by operative forceps were again unsuccessful. Due to the patient s request Figure 1. HSG image displaying an acute curvilinear appearance of right micro-insert. Figure 2. HSG image displaying free-spill from right side with injection of dye. 2
3 Woo/Simpson/Stovall of removal of the entire foreign body, the previously discussed and consented total laparoscopic hysterectomy was performed without complication. The uterine specimen was removed and thoroughly examined. Grossly, as noted by diagnostic laparoscopy, there was no evidence of perforation of the uterine serosa by either microinsert. The uterine specimen was dissected in a midline coronal manner superiorly from the cervix to the fundus and the device was found inserted superior to the right tubal ostia (Figures 4 and 5). On further dissection, it was discovered that the device was firmly embedded through more than half of the myometrium (Figure 6). Discussion The mechanism of occlusion of the Essure mico-insert device includes an inflammatory response of the fallopian tube to the micro-insert. The inner coil is composed of stainless steel and polyethylene terephthalate (PET) fibers and the outer coil is composed of nickel-titanium (nitinol). The fallopian tubes inflammatory response to these materials causes fibroblastic growth within and around the double-coiled micro-insert to occlude the fallopian tube lumen [8]. Bayer states that an Essure Confirmation Test (ECT) should be completed 3 months after micro-insert device placement to verify correct bilateral placement and tubal occlusion [9,10]. This confirmation test may be completed by either a HSG or transvaginal ultrasound (TVU) [9,10]. A patient is eligible for TVU confirmation if the placement is uncomplicated without visualization difficulties, there are no concerns of perforation, the procedure time is 15 minutes or less, 1-8 trailing coils are apparent bilaterally, the patient is not on any active immunosuppressive therapy, and the post-operative course is uncomplicated [10]. In all other cases, an HSG is indicated. According to the HSG protocol, correct placement of the microinsert is based upon both tubal occlusion and location of both the inner and outer coil radiographic markers. Otherwise, placement is considered unsatisfactory for purposes of reliable sterilization [9]. A too proximal location occurs when 50% or more of the inner coil is trailing into the uterine cavity while a too distal insert location occurs when the insert is in the tube with the proximal end of the inner coil >30 mm from the cornua [9]. If the insert location is satisfactory but there is patency beyond the distal end of the outer coil or free spill is appreciated, a repeat ECT should be completed after an additional 3 months with alternative contraception during the interim [10]. Figure 3. Thirty to forty trailing micro-insert coils visualized in uterine cavity during hysteroscopic evaluation. Figure 4. Midline coronal dissection displaying the micro-insert penetrating into the myometrium. 3
4 Citation: Woo JJ, Simpson BE, Stovall DW. Myometrial Insertion of Essure Mirco-Insert. Res Rep Gynaecol Obstet. 2017;1(4):1-5 Figure 5. Micro-insert penetrating superior to right tubal ostia. Figure 6. Micro-insert embedded into uterine myometrium without penetration through the uterine serosa. In this case report, although a TVU confirmation test was an option, an HSG interpreted by a competent radiologist was our confirmation test of choice. The HSG revealed no clear concerns for a too proximal or distal insertion; however, because of free spill of contrast media from the right fallopian tube and an acute curvilinear appearance of the right-sided device, it was clear that the mico-insert was not placed in the fallopian tube nor was the tube occluded. Four months after the initial insertion, partial expulsion of the right microinsert had also occurred with trailing coils noted in the intrauterine cavity on hysteroscopy. After dissection of the uterus, it was apparent that the right micro-insert device was not properly inserted into the fallopian tube, but was in fact inserted into the myometrium. There are two critical clinical questions that arise from this case report. First, how was the right micro-insert inserted into the myometrium without any suspicion of malposition? Second, what mechanisms explain the retrograde movement of the micro-insert from the right tubal ostia 4 months after insertion? Adenomyosis theory It is unclear exactly how the micro-insert was placed into the myometrium. One theory is that the device was not catheterized through the tubal ostia, but perhaps into a glandular-like opening or dimpling of adenomyosis. This might explain the lack of resistance at the time of insertion. Catheterization of a possible adenomyosis gland opening has been discussed in one case report that resulted in a likely cornual uterine perforation [11-13]. Common hysteroscopic adenomyosis findings include the appearance of several gland-like openings with and without blood as well as hemosiderin deposits within the endometrium [14]. In this case, the endometrial gland-like opening or dimpling from adenomyosis may have been misidentified as a tubal ostium. Although the appearance of the endometrium in this patient suggests adenomyosis may be possible, hysteroscopy only detects adenomyosis in 10% to 20% of cases and cannot determine focal versus extensive disease [14]. The final pathology diagnosis made no comment regarding adenomyosis. 4
5 Woo/Simpson/Stovall The expulsion of coils visualized in the uterine cavity 4 months after insertion on the right side may be explained by recoil from a dense, non-compliant myometrial tissue. Unlike the compliant fallopian tube lumen, the myometrium s dense non-complaint tissue likely rejected the device causing an initial post-procedure partial expulsion and then a later fibroblastic inflammatory response resulting in myometrial embedding (Figure 6). There are several take-home points that one can learn from this unusual case. First, due to the inflammatory properties of the micro-insert device, the more time the device is in place, the more difficult it will be to remove hysteroscopically. Second, removal of the Essure device requires the removal of both the inner coil of stainless steel and polyethylene terephthalate (PET) fibers and an outer coil of nickel-titanium (nitinol). Third, when placing the device, it is imperative to have a clear visualization and identification of both tubal ostia prior to placement. This is especially important if there is a high suspicion of adenomyosis. Hormonal medications may aid in hysteroscopic visualization during the procedure. Fourth, while most patients malpositioned micro-insert devices can present with pelvic pain, in this particular case, the only symptom was menorrhagia. Lastly, if a malpositioned micro-insert is suspected, the patient s desires regarding removal should be discussed thoroughly. If necessary, a possible hysterectomy should be considered and consented. While a hysterectomy is the last resort for the removal of a malpositioned micro-insert, it may be required for definitive management. Conclusion In conclusion, to our knowledge, this is the first case to be reported of an Essure micro-insert that has been identified embedded into myometrium without perforation through the serosa of the uterus. Management of an embedded myometrial micro-insert without perforation may be treated in a less invasive surgical approach, but it is difficult to determine the optimal treatment based on one case report. While our patient had only one month of menorrhagia without pelvic pain, it is uncertain whether pain may have developed with time and eventually require further definitive surgical management. However, a total laparoscopic hysterectomy was necessary in this particular case because of the patient s request for removal of the entire device. References 1. Al-Safi Z, Shavell V, Hobson D, et al. Analysis of adverse events with Essure hysteroscopic sterilization reported to the manufacturer and user facility device experience database. J Minim Invasive Gynecol. 2013;20(6): Braginsky L, George S, Locher S. Management of perforated Essure with migration into small and large bowel mesentery. J Minim Invasive Gynecol. 2015;22(3): Ricci G, Restaino S, DiLorenzo G, et al. Risk of Essure microinsert abdominal migration: case report and review of literature. Ther Clin Risk Manag. 2014;10: Garcia AL, Lewis RM, Sloan AL. Essure insert expulsion after 3-month hysterosalpingogram confirmation of bilateral tubal occlusion and bilateral correct placement: Case report. J Minim Invasive Gynecol. 2013;20(1): Conover M, Howell J, Wu J, et al. Incidence of opioidmanaged pelvic pain after hysteroscopic sterilization versus laparoscopic sterilization, U.S Pharmacoepidemiol Drug Saf. 2015;24(8): U.S. Food And Drug Administration. Meeting Materials of the Obstetrics and Gynecology Devices Panel U.S. Food And Drug Administration. FDA Activities: Essure Veersema S, VIeugels M, Koks C, et al. Confirmation of Essure placement using transvaginal ultrasound. J Minim Invasive Gynecol. 2011;18(2): Thoma V, Chua I, Garbin O, et al. Tubal perforation by ESSURE microinsert. J Minim Invasive Gynecol. 200;13(2): Glasser MH. Tubal perforation by ESSURE microinsert: A tubal perforation? J Minim Invasive Gynecol. 2006;13(5): Valle RF. Tubal peroration by ESSURE microinsert: Clearly not a tubal peroration but cornual-uterine perforation. J Minim Invasive Gynecol. 2008;13(5): Bradley, Linda D, Tommaso F. Indications and contraindication for office hysteroscopy. Hysteroscopy: Office Evaluation and Management of the Uterine Cavity. Mosby/Elsevier, Philadelphia, *Correspondence to: Jeffrey J Woo Department of Obstetrics and Gynecology Riverside Regional Medical Center Virginia United States of America Jeffrey.Woo@rivhs.com 5
ESSURE A RESOURCE FOR CODING
 ESSURE REIMBURSEMENT GUIDE A RESOURCE FOR CODING INDICATION Essure is indicated for women who desire permanent birth control (female sterilization) by bilateral occlusion of fallopian tubes. IMPORTANT
ESSURE REIMBURSEMENT GUIDE A RESOURCE FOR CODING INDICATION Essure is indicated for women who desire permanent birth control (female sterilization) by bilateral occlusion of fallopian tubes. IMPORTANT
Update on the Essure System for Permanent Birth Control
 Update on the Essure System for Permanent Birth Control Heidi Collins Fantasia IIn 2002, the U.S. Food and Drug Administration (FDA) approved Essure (Bayer, Whippany, NJ), a minimally invasive procedure
Update on the Essure System for Permanent Birth Control Heidi Collins Fantasia IIn 2002, the U.S. Food and Drug Administration (FDA) approved Essure (Bayer, Whippany, NJ), a minimally invasive procedure
Unintended pregnancies after Essure sterilization in the Netherlands
 Unintended pregnancies after Essure sterilization in the Netherlands S. Veersema M.P.H. Vleugels L.M. Moolenaar C.A.H. Janssen H.A.M. Brölmann Fertil Steril. 2010;93:35-8. 13 Hysteroscopic Sterilization
Unintended pregnancies after Essure sterilization in the Netherlands S. Veersema M.P.H. Vleugels L.M. Moolenaar C.A.H. Janssen H.A.M. Brölmann Fertil Steril. 2010;93:35-8. 13 Hysteroscopic Sterilization
Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization Draft Guidance for Industry and Food and Drug
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization Draft Guidance for Industry and Food and Drug
Clinical Experience With Contrast Infusion Sonography as an Essure Confirmation Test
 ORIGINAL RESEARCH Clinical Experience With Contrast Infusion Sonography as an Essure Confirmation Test Viviane F. Connor, MD Received November 3, 2010, from the Department of Gynecology, Section of Minimally
ORIGINAL RESEARCH Clinical Experience With Contrast Infusion Sonography as an Essure Confirmation Test Viviane F. Connor, MD Received November 3, 2010, from the Department of Gynecology, Section of Minimally
Case Report. Introduction. E. Scott Sills 1-3, Gianpiero D. Palermo 4
 Case Report Obstet Gynecol Sci 2016;59(4):337-341 http://dx.doi.org/10.5468/ogs.2016.59.4.337 pissn 2287-8572 eissn 2287-8580 Combined hysteroscopy-laparoscopy approach for excision of pelvic nitinol fragment
Case Report Obstet Gynecol Sci 2016;59(4):337-341 http://dx.doi.org/10.5468/ogs.2016.59.4.337 pissn 2287-8572 eissn 2287-8580 Combined hysteroscopy-laparoscopy approach for excision of pelvic nitinol fragment
MEDICAL POLICY SUBJECT: FEMALE STERILIZATION. POLICY NUMBER: CATEGORY: Contract Clarification
 MEDICAL POLICY SUBJECT: FEMALE STERILIZATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: FEMALE STERILIZATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Essure Permanent Birth Control Device: Radiological followup results at our center
 Essure Permanent Birth Control Device: Radiological followup results at our center Poster No.: C-0212 Congress: ECR 2013 Type: Scientific Exhibit Authors: R. Díaz Aguilera, A. M. Higuera Higuera, V. Palomo
Essure Permanent Birth Control Device: Radiological followup results at our center Poster No.: C-0212 Congress: ECR 2013 Type: Scientific Exhibit Authors: R. Díaz Aguilera, A. M. Higuera Higuera, V. Palomo
Case Report Essure Surgical Removal and Subsequent Resolution of Chronic Pelvic Pain: A Case Report and Review of the Literature
 Case Reports in Obstetrics and Gynecology Volume 2016, Article ID 6961202, 5 pages http://dx.doi.org/10.1155/2016/6961202 Case Report Essure Surgical Removal and Subsequent Resolution of Chronic Pelvic
Case Reports in Obstetrics and Gynecology Volume 2016, Article ID 6961202, 5 pages http://dx.doi.org/10.1155/2016/6961202 Case Report Essure Surgical Removal and Subsequent Resolution of Chronic Pelvic
National Institute for Health and Clinical Excellence
 National Institute for Health and Clinical Excellence 218_2 Hysteroscopic sterilisation by tubal cannulation and placement of intrafallopian implants Consultation Comments table IPAC date: Thursday 16
National Institute for Health and Clinical Excellence 218_2 Hysteroscopic sterilisation by tubal cannulation and placement of intrafallopian implants Consultation Comments table IPAC date: Thursday 16
Clinical Policy: Essure Removal Reference Number: CP.MP.131
 Clinical Policy: Reference Number: CP.MP.131 Effective Date: 11/16 Last Review Date: 11/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.131 Effective Date: 11/16 Last Review Date: 11/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
ESSURE CLINICAL RESOURCE
 ESSURE CLINICAL RESOURCE Physician Training Manual IMPORTANT SAFETY INFORMATION WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or reported adverse
ESSURE CLINICAL RESOURCE Physician Training Manual IMPORTANT SAFETY INFORMATION WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or reported adverse
Complications of hysteroscopic Essure â sterilisation: report on 4306 procedures performed in a single centre
 DOI: 10.1111/j.1471-0528.2012.03292.x www.bjog.org Fertility control Complications of hysteroscopic Essure â sterilisation: report on 4306 procedures performed in a single centre B Povedano, a JE Arjona,
DOI: 10.1111/j.1471-0528.2012.03292.x www.bjog.org Fertility control Complications of hysteroscopic Essure â sterilisation: report on 4306 procedures performed in a single centre B Povedano, a JE Arjona,
Figure 1b: Essure Insert (NOT TO SCALE)
 Figure 1b: Essure Insert Expanded configuration, detached from the delivery system (NOT TO SCALE) WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or
Figure 1b: Essure Insert Expanded configuration, detached from the delivery system (NOT TO SCALE) WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or
Transcervical Sterilization
 Q UESTIONS & ANSWERS A BOUT Transcervical Sterilization A New Choice in Permanent Birth Control Choosing a Birth Control Method Women and their partners now have more birth control choices than ever. How
Q UESTIONS & ANSWERS A BOUT Transcervical Sterilization A New Choice in Permanent Birth Control Choosing a Birth Control Method Women and their partners now have more birth control choices than ever. How
V. Mijatovic S. Veersema M.H. Emanuel R. Schats P.G. Hompes. Fertil Steril. 2010;93:
 Essure hysteroscopic tubal occlusion device for the treatment of hydrosalpinx prior to in vitro fertilization-embryo transfer in patients with a contraindication for laparoscopy. V. Mijatovic S. Veersema
Essure hysteroscopic tubal occlusion device for the treatment of hydrosalpinx prior to in vitro fertilization-embryo transfer in patients with a contraindication for laparoscopy. V. Mijatovic S. Veersema
Imaging evaluation of gynaecological devices
 Imaging evaluation of gynaecological devices Poster No.: C-0850 Congress: ECR 2013 Type: Educational Exhibit Authors: R. M. Lorente Ramos, J. Azpeitia Arman, P. Aparicio Rodríguez- 1 1 1 1 1 Miñón, F.
Imaging evaluation of gynaecological devices Poster No.: C-0850 Congress: ECR 2013 Type: Educational Exhibit Authors: R. M. Lorente Ramos, J. Azpeitia Arman, P. Aparicio Rodríguez- 1 1 1 1 1 Miñón, F.
Permanent Sterilization: When you are really sure!
 Permanent Sterilization: When you are really sure! Tony Ogburn MD Department of Ob/Gyn 2006-8 National Survey of Family Growth 6.1% of women had a sterilized male partner The History of Female Sterilization
Permanent Sterilization: When you are really sure! Tony Ogburn MD Department of Ob/Gyn 2006-8 National Survey of Family Growth 6.1% of women had a sterilized male partner The History of Female Sterilization
Hysteroscopic Tubal Sterilization: An Evidence-Based Analysis
 Hysteroscopic Tubal Sterilization: An Evidence-Based Analysis K McMartin October 2013 Ontario Health Technology Assessment Series; Vol.13: No. 21, pp. 1 35, October 2013 Suggested Citation This report
Hysteroscopic Tubal Sterilization: An Evidence-Based Analysis K McMartin October 2013 Ontario Health Technology Assessment Series; Vol.13: No. 21, pp. 1 35, October 2013 Suggested Citation This report
Excessive menstrual blood loss
 Ian Chilcott Excessive menstrual blood loss >80mls - That interferes with physical, emotional, social and material quality of life 1 in 20 women aged 30 to 49 years consult their GP each year with menorrhagia
Ian Chilcott Excessive menstrual blood loss >80mls - That interferes with physical, emotional, social and material quality of life 1 in 20 women aged 30 to 49 years consult their GP each year with menorrhagia
Essure By Mayo Clinic staff
 Page 1 of 5 Reprints A single copy of this article may be reprinted for personal, noncommercial use only. Essure By Mayo Clinic staff Original Article: http://www.mayoclinic.com/health/essure/my00999 Definition
Page 1 of 5 Reprints A single copy of this article may be reprinted for personal, noncommercial use only. Essure By Mayo Clinic staff Original Article: http://www.mayoclinic.com/health/essure/my00999 Definition
Figure 1b: Essure Insert (NOT TO SCALE) (NOT TO SCALE)
 Figure 1b: Essure Insert Expanded configuration, detached from the delivery system (NOT TO SCALE) WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or
Figure 1b: Essure Insert Expanded configuration, detached from the delivery system (NOT TO SCALE) WARNING: Some patients implanted with the Essure System for Permanent Birth Control have experienced and/or
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of sterilisation by insertion of intrafallopian implants Sterilisation is sometimes
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of sterilisation by insertion of intrafallopian implants Sterilisation is sometimes
Female sterilisation: a cohort controlled comparative study of ESSURE versus laparoscopic sterilisation
 BJOG: an International Journal of Obstetrics and Gynaecology November 2005, Vol. 112, pp. 1522 1528 DOI: 10.1111/j.1471-0528.2005.00726.x Female sterilisation: a cohort controlled comparative study of
BJOG: an International Journal of Obstetrics and Gynaecology November 2005, Vol. 112, pp. 1522 1528 DOI: 10.1111/j.1471-0528.2005.00726.x Female sterilisation: a cohort controlled comparative study of
Female sterilization by the natural pathways
 info-canada@conceptus.com www.essure.com Female sterilization by the natural pathways magazine 1.Hatcher R et al. Contraceptive Technology, 17 th Edition. New York : Ardent Media, 1998. 2. Bhiwandiwala
info-canada@conceptus.com www.essure.com Female sterilization by the natural pathways magazine 1.Hatcher R et al. Contraceptive Technology, 17 th Edition. New York : Ardent Media, 1998. 2. Bhiwandiwala
Evidence Based Guideline Intrauterine Ablation or Resection of the Endometrium
 Evidence Based Guideline Intrauterine Ablation or Resection of the Endometrium File Name: intrauterine_ablation_or_resection_of_the_endometrium Guideline Number: EBG.OBGYN3030 Origination: 4/1993 Last
Evidence Based Guideline Intrauterine Ablation or Resection of the Endometrium File Name: intrauterine_ablation_or_resection_of_the_endometrium Guideline Number: EBG.OBGYN3030 Origination: 4/1993 Last
Satisfaction and tolerance with office hysteroscopic tubal sterilization
 TECHNIQUES AND INSTRUMENTATION Satisfaction and tolerance with office hysteroscopic tubal sterilization Jose E. Arjona, M.D., Ph.D., a Monica Mi~no, M.D., Ph.D., a Javier Cordon, M.D., Ph.D., a Balbino
TECHNIQUES AND INSTRUMENTATION Satisfaction and tolerance with office hysteroscopic tubal sterilization Jose E. Arjona, M.D., Ph.D., a Monica Mi~no, M.D., Ph.D., a Javier Cordon, M.D., Ph.D., a Balbino
Endometrial Cancer Biopsy of the endometrium Evaluation of women of all ages
 Endometrial Cancer Biopsy of the endometrium Evaluation of women of all ages Barbara S. Apgar, MD, MS Professor of Family Medicine University of Michigan Health System Ann Arbor, Michigan Cancer of the
Endometrial Cancer Biopsy of the endometrium Evaluation of women of all ages Barbara S. Apgar, MD, MS Professor of Family Medicine University of Michigan Health System Ann Arbor, Michigan Cancer of the
Myometrial scoring: a new technique for the management of severe Asherman s syndrome
 FERTILITY AND STERILITY VOL. 69, NO. 5, MAY 1998 Copyright 1998 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Myometrial scoring: a
FERTILITY AND STERILITY VOL. 69, NO. 5, MAY 1998 Copyright 1998 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Myometrial scoring: a
TUBAL PLASTIC SURGERY is an accepted form of therapy in the treatment
 Tubal Plastic Surgery ADNAN MROUEH, M.D., ROBERT H. GLASS, M.D., and C. LEE BUXTON, M.D. TUBAL PLASTIC SURGERY is an accepted form of therapy in the treatment of infertility. However, reports have differed
Tubal Plastic Surgery ADNAN MROUEH, M.D., ROBERT H. GLASS, M.D., and C. LEE BUXTON, M.D. TUBAL PLASTIC SURGERY is an accepted form of therapy in the treatment of infertility. However, reports have differed
Laparoscopy-Hysteroscopy
 Laparoscopy-Hysteroscopy Patient Information Laparoscopy The laparoscope, a surgical instrument similar to a telescope, is inserted through a small incision (cut) in the belly button during laparoscopy.
Laparoscopy-Hysteroscopy Patient Information Laparoscopy The laparoscope, a surgical instrument similar to a telescope, is inserted through a small incision (cut) in the belly button during laparoscopy.
What s New in Adolescent Contraception?
 What s New in Adolescent Contraception? Abby Furukawa, MD Legacy Medical Group Portland Obstetrics and Gynecology April 29, 2017 Objectives Provide an update on contraception options for the adolescent
What s New in Adolescent Contraception? Abby Furukawa, MD Legacy Medical Group Portland Obstetrics and Gynecology April 29, 2017 Objectives Provide an update on contraception options for the adolescent
Family Planning Eligibility Program
 INDIANA HEALTH COVERAGE PROGRAMS PROVIDER REFERENCE M ODULE Family Planning Eligibility Program L I B R A R Y R E F E R E N C E N U M B E R : P R O M O D 0 0 0 5 3 P U B L I S H E D : N O V E M B E R 2
INDIANA HEALTH COVERAGE PROGRAMS PROVIDER REFERENCE M ODULE Family Planning Eligibility Program L I B R A R Y R E F E R E N C E N U M B E R : P R O M O D 0 0 0 5 3 P U B L I S H E D : N O V E M B E R 2
The use of long-acting reversible contraceptive
 Overcoming LARC complications: 7 case challenges The strings to your patient s intrauterine device (IUD) are missing. Clinical experience and ACOG direction guide the management plans for this and more
Overcoming LARC complications: 7 case challenges The strings to your patient s intrauterine device (IUD) are missing. Clinical experience and ACOG direction guide the management plans for this and more
SURGICAL PROBLEMS IN FERTILITY- FIBROIDS. Dr.Māris Arājs gyn-ob specialist Cell phone:
 SURGICAL PROBLEMS IN FERTILITY- FIBROIDS Dr.Māris Arājs gyn-ob specialist maris@myclinicriga.lv Cell phone: +371 26556466 There is NO Industry Sponsorship and Financial Conflict of Interest for this presentation
SURGICAL PROBLEMS IN FERTILITY- FIBROIDS Dr.Māris Arājs gyn-ob specialist maris@myclinicriga.lv Cell phone: +371 26556466 There is NO Industry Sponsorship and Financial Conflict of Interest for this presentation
Dr John Short. Obstetrician and Gynaecologist Christchurch Women s Hospital Oxford Women's Health Christchurch
 Dr John Short Obstetrician and Gynaecologist Christchurch Women s Hospital Oxford Women's Health Christchurch 16:30-17:30 WS #125: Everything GPs Should Know About Gynaecologists 17:35-18:30 WS #135: Everything
Dr John Short Obstetrician and Gynaecologist Christchurch Women s Hospital Oxford Women's Health Christchurch 16:30-17:30 WS #125: Everything GPs Should Know About Gynaecologists 17:35-18:30 WS #135: Everything
Female Sterilization. Kavita Nanda, MD, MHS FHI 360 Expanding Contraceptive Choice December 6, 2018
 Female Sterilization Kavita Nanda, MD, MHS FHI 360 Expanding Contraceptive Choice December 6, 2018 What is female sterilization? Family planning method that provides permanent contraception to women and
Female Sterilization Kavita Nanda, MD, MHS FHI 360 Expanding Contraceptive Choice December 6, 2018 What is female sterilization? Family planning method that provides permanent contraception to women and
Tissue Morcellation: Managing Risks to Drive Best Patient Outcomes
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
ASHERMAN S SYNDROME FOLLOWING THERMAL ABLATION OF THE ENDOMETRIUM Sheila K. Pillai 1, Bhuvana S 2, Jaya Vijayaraghavan 3
 ASHERMAN S SYNDROME FOLLOWING THERMAL ABLATION OF THE ENDOMETRIUM Sheila K. Pillai 1, Bhuvana S 2, Jaya Vijayaraghavan 3 HOW TO CITE THIS ARTICLE: Sheila K. Pillai, Bhuvana S, Jaya Vijayaraghavan. Asherman
ASHERMAN S SYNDROME FOLLOWING THERMAL ABLATION OF THE ENDOMETRIUM Sheila K. Pillai 1, Bhuvana S 2, Jaya Vijayaraghavan 3 HOW TO CITE THIS ARTICLE: Sheila K. Pillai, Bhuvana S, Jaya Vijayaraghavan. Asherman
Laparoscopy and Hysteroscopy
 AMERICAN SOCIETY FOR REPRODUCTIVE MEDICINE Laparoscopy and Hysteroscopy A Guide for Patients PATIENT INFORMATION SERIES Published by the American Society for Reproductive Medicine under the direction of
AMERICAN SOCIETY FOR REPRODUCTIVE MEDICINE Laparoscopy and Hysteroscopy A Guide for Patients PATIENT INFORMATION SERIES Published by the American Society for Reproductive Medicine under the direction of
A COMPARISON OF HYSTEROSALPINGOGRAPHY AND LAPAROSCOPY IN THE INVESTIGATION OF INFERTILITY
 Basrah Journal of Surgery A COMPARISON OF HYSTEROSALPINGOGRAPHY AND LAPAROSCOPY IN THE INVESTIGATION OF INFERTILITY Fouad Hamad Al-Dahhan * & Zainab Baker @ *FRCOG, Assistant Professor, @ M.B.Ch.B. Department
Basrah Journal of Surgery A COMPARISON OF HYSTEROSALPINGOGRAPHY AND LAPAROSCOPY IN THE INVESTIGATION OF INFERTILITY Fouad Hamad Al-Dahhan * & Zainab Baker @ *FRCOG, Assistant Professor, @ M.B.Ch.B. Department
What is endometrial cancer?
 Uterine cancer What is endometrial cancer? Endometrial cancer is the growth of abnormal cells in the lining of the uterus. The lining is called the endometrium. Endometrial cancer usually occurs in women
Uterine cancer What is endometrial cancer? Endometrial cancer is the growth of abnormal cells in the lining of the uterus. The lining is called the endometrium. Endometrial cancer usually occurs in women
1. Ortiz, M. E et al. Mechanisms of action of intrauterine devices. Obstet & Gynl Survey 1996; 51(12), 42S-51S.
 1 2 1. Ortiz, M. E et al. Mechanisms of action of intrauterine devices. Obstet & Gynl Survey 1996; 51(12), 42S-51S. The contraceptive action of all IUDs is mainly in the uterine cavity. The major effect
1 2 1. Ortiz, M. E et al. Mechanisms of action of intrauterine devices. Obstet & Gynl Survey 1996; 51(12), 42S-51S. The contraceptive action of all IUDs is mainly in the uterine cavity. The major effect
MPH Quiz. 1. How many primaries are present based on this pathology report? 2. What rule is this based on?
 MPH Quiz Case 1 Surgical Pathology from hysterectomy performed July 11, 2007 Final Diagnosis: Uterus, resection: Endometrioid adenocarcinoma, Grade 1 involving most of endometrium, myometrial invasion
MPH Quiz Case 1 Surgical Pathology from hysterectomy performed July 11, 2007 Final Diagnosis: Uterus, resection: Endometrioid adenocarcinoma, Grade 1 involving most of endometrium, myometrial invasion
Cortisone in the Treatment of Tubal Occlusion Caused by Healed Genital Tuberculosis
 Cortisone in the Treatment of Tubal Occlusion Caused by Healed Genital Tuberculosis ISAC HALBRECHT, M.D. THERE IS a general agreement on the importance of the tubal factor in sterility. In certain geographic
Cortisone in the Treatment of Tubal Occlusion Caused by Healed Genital Tuberculosis ISAC HALBRECHT, M.D. THERE IS a general agreement on the importance of the tubal factor in sterility. In certain geographic
Index. B Bladder, injury of, Bowel, injury of, , Brachytherapy, for cervical cancer, 357 Burns, electrosurgical,
 Perioperative Nursing Clinics 1 (2006) 375 379 Index Note: Page numbers of article titles are in boldface type. A Abdominal hysterectomy Acidosis, from insufflation, 323 Active electrode monitoring, in
Perioperative Nursing Clinics 1 (2006) 375 379 Index Note: Page numbers of article titles are in boldface type. A Abdominal hysterectomy Acidosis, from insufflation, 323 Active electrode monitoring, in
Corporate Medical Policy
 Corporate Medical Policy Intrauterine Ablation or Resection of the Endometrium File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intrauterine_ablation_or_resection_of_the_endometrium
Corporate Medical Policy Intrauterine Ablation or Resection of the Endometrium File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intrauterine_ablation_or_resection_of_the_endometrium
Endometrial Stromal Sarcoma
 May 26, 2011 By Sushila Ladumor, MD [1] Endometrial stromal sarcoma (ESS) is a rare malignant tumor of the endometrium, occurring in the age group of 40-50 years. History The 50-year-old, female patient
May 26, 2011 By Sushila Ladumor, MD [1] Endometrial stromal sarcoma (ESS) is a rare malignant tumor of the endometrium, occurring in the age group of 40-50 years. History The 50-year-old, female patient
ENDOMETRIAL ABLATION: TRENDS AND CHALLENGES IN 2017
 ENDOMETRIAL ABLATION: TRENDS AND CHALLENGES IN 2017 Philippe Laberge MD FRCSC ACGE Professor Obstetrics and Gynecology Laval University Quebec, Canada Disclosures I have used products or done clinical
ENDOMETRIAL ABLATION: TRENDS AND CHALLENGES IN 2017 Philippe Laberge MD FRCSC ACGE Professor Obstetrics and Gynecology Laval University Quebec, Canada Disclosures I have used products or done clinical
Example CLINICAL GUIDELINES for Postpartum IUD insertion
 Example CLINICAL GUIDELINES for Postpartum IUD insertion Postpartum Intrauterine Device Insertion 1.0 Indications: 1.1 Insertion of an intrauterine device (IUD) for long-acting reversible contraception
Example CLINICAL GUIDELINES for Postpartum IUD insertion Postpartum Intrauterine Device Insertion 1.0 Indications: 1.1 Insertion of an intrauterine device (IUD) for long-acting reversible contraception
Please complete prior to the webinar. HOSPITAL REGISTRY WEBINAR FEMALE REPRODUCTIVE SYSTEM EXERCISES CASE 1: FEMALE REPRODUCTIVE
 Please complete prior to the webinar. HOSPITAL REGISTRY WEBINAR FEMALE REPRODUCTIVE SYSTEM EXERCISES PHYSICAL EXAMINATION CASE 1: FEMALE REPRODUCTIVE 3/5 Patient presents through the emergency room with
Please complete prior to the webinar. HOSPITAL REGISTRY WEBINAR FEMALE REPRODUCTIVE SYSTEM EXERCISES PHYSICAL EXAMINATION CASE 1: FEMALE REPRODUCTIVE 3/5 Patient presents through the emergency room with
INTERVENTIONAL PROCEDURES PROGRAMME
 NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of microwave endometrial ablation Introduction This overview has been prepared to assist
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of microwave endometrial ablation Introduction This overview has been prepared to assist
M-AFRAKHTEH. MD OCT.2017 SHOHADA HOSPITAL TAJRISH
 Unrestricted M-AFRAKHTEH. MD OCT.2017 SHOHADA HOSPITAL TAJRISH Patients at imminent risk of exsanguination Manual aortic compression Resuscitative endovascular balloon occlusion of the aorta Uterine tourniquet
Unrestricted M-AFRAKHTEH. MD OCT.2017 SHOHADA HOSPITAL TAJRISH Patients at imminent risk of exsanguination Manual aortic compression Resuscitative endovascular balloon occlusion of the aorta Uterine tourniquet
FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system)
 FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system) Mirena does not protect against HIV infection (AIDS) and other sexually transmitted infections
FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system) Mirena does not protect against HIV infection (AIDS) and other sexually transmitted infections
38 OBG Management August 2012 Vol. 24 No. 8 obgmanagement.com
 FIGURE 1 Copper intrauterine device displaced in the lower uterine segment with the left arm embedded in the myometrium. illustrations: craig zuckerman for obg management 38 OBG Management August 2012
FIGURE 1 Copper intrauterine device displaced in the lower uterine segment with the left arm embedded in the myometrium. illustrations: craig zuckerman for obg management 38 OBG Management August 2012
Role of diagnostic hysteroscopy in evaluation of abnormal uterine bleeding and its histopathological correlation
 International Journal of Reproduction, Contraception, Obstetrics and Gynecology Chaudhari KR et al. Int J Reprod Contracept Obstet Gynecol. 2014 Sep;3(3):666-670 www.ijrcog.org pissn 2320-1770 eissn 2320-1789
International Journal of Reproduction, Contraception, Obstetrics and Gynecology Chaudhari KR et al. Int J Reprod Contracept Obstet Gynecol. 2014 Sep;3(3):666-670 www.ijrcog.org pissn 2320-1770 eissn 2320-1789
Medicaid Family Planning Waiver Services CPT Codes and ICD-10 Diagnosis Codes
 CPT Code Description of Covered Codes Evaluation and Management 99384FP 99385FP Family planning new visit 99386FP 99394FP 99395FP Family planning established visit 99396FP 99401FP HIV counseling (pre-test)
CPT Code Description of Covered Codes Evaluation and Management 99384FP 99385FP Family planning new visit 99386FP 99394FP 99395FP Family planning established visit 99396FP 99401FP HIV counseling (pre-test)
bleeding Studies naar de diagnostiek van endom triumcarcinoom bij vrouwen met postm nopauzaal bloedverlies. Studies on the
 Studies on the diagnosis of endometria cancer in women with postmenopausal bleeding. Studies naar de diagnostiek va endometriumcarcinoom bij vrouwen m postmenopauzaal bloedverlies. Studies on the diagnosis
Studies on the diagnosis of endometria cancer in women with postmenopausal bleeding. Studies naar de diagnostiek va endometriumcarcinoom bij vrouwen m postmenopauzaal bloedverlies. Studies on the diagnosis
A Rare Case of Invasive Squamous Cell Carcinoma of Cervix Extending to Endometrium and Right Fallopian Tube
 A Rare Case of Invasive Squamous Cell Carcinoma of Cervix Extending to Endometrium and Right Fallopian Tube Kate Madhuri S 1, Gulhane Sushma R 2, Mane Sheetal V 3 1 Professor and Head, 2 Specialist cum
A Rare Case of Invasive Squamous Cell Carcinoma of Cervix Extending to Endometrium and Right Fallopian Tube Kate Madhuri S 1, Gulhane Sushma R 2, Mane Sheetal V 3 1 Professor and Head, 2 Specialist cum
ADENOMYOSIS CHRONIC PELVIC PAIN IN WOMEN IMAGING CHRONIC PELVIC PAIN IN WOMEN CHRONIC PELVIC PAIN IN WOMEN ADENOMYOSIS: PATHOLOGY ADENOMYOSIS
 CHRONIC PELVIC PAIN IN WOMEN IMAGING CHRONIC PELVIC PAIN IN WOMEN MOSTAFA ATRI, MD Dipl. Epid. UNIVERSITY OF TORONTO Non-menstrual pain of 6 months Prevalence 15%: 18-50 years of age 10-40% of gynecology
CHRONIC PELVIC PAIN IN WOMEN IMAGING CHRONIC PELVIC PAIN IN WOMEN MOSTAFA ATRI, MD Dipl. Epid. UNIVERSITY OF TORONTO Non-menstrual pain of 6 months Prevalence 15%: 18-50 years of age 10-40% of gynecology
5/5/2010 FINANCIAL DISCLOSURE. Abnormal Uterine Bleeding. Is This A Problem? About me % of visits to gynecologist
 Abnormal Uterine FINANCIAL DISCLOSURE I HAVE NO FINANCIAL INTEREST IN ANY OF THE PRODUCTS MENTIONED IN MY PRESENTATION Bryan K. Rone, M.D. University of Kentucky Obstetrics and Gynecology May 5, 2010 About
Abnormal Uterine FINANCIAL DISCLOSURE I HAVE NO FINANCIAL INTEREST IN ANY OF THE PRODUCTS MENTIONED IN MY PRESENTATION Bryan K. Rone, M.D. University of Kentucky Obstetrics and Gynecology May 5, 2010 About
Microinsert Nonincisional Hysteroscopic Sterilization
 Microinsert Nonincisional Hysteroscopic Sterilization Jay M. Cooper, MD, Charles S. Carignan, MD, Daniel Cher, MD, and John F. Kerin, MD, for the Selective Tubal Occlusion Procedure 2000 Investigators
Microinsert Nonincisional Hysteroscopic Sterilization Jay M. Cooper, MD, Charles S. Carignan, MD, Daniel Cher, MD, and John F. Kerin, MD, for the Selective Tubal Occlusion Procedure 2000 Investigators
THE WOMAN-FRIENDLY STERILIZATION METHOD
 THE WOMAN-FRIENDLY STERILIZATION METHOD Urogyn BV Transistorweg 5a 6534 AT Nijmegen The Netherlands t +31(0) 24 711 41 30 info@urogynbv.com www.urogynbv.com THE MOST WOMAN-FRIENDLY STERILIZATION METHOD
THE WOMAN-FRIENDLY STERILIZATION METHOD Urogyn BV Transistorweg 5a 6534 AT Nijmegen The Netherlands t +31(0) 24 711 41 30 info@urogynbv.com www.urogynbv.com THE MOST WOMAN-FRIENDLY STERILIZATION METHOD
CNGOF Guidelines for the Management of Endometriosis
 CNGOF Guidelines for the Management of Endometriosis Anatomoclinical forms of endometriosis Definitions Endometriosis is defined as the presence of endometrial tissue containing both glands and stroma
CNGOF Guidelines for the Management of Endometriosis Anatomoclinical forms of endometriosis Definitions Endometriosis is defined as the presence of endometrial tissue containing both glands and stroma
Program Schedule. Update in Gynecology and Minimally Invasive Surgery 2018
 Program Schedule Update in Gynecology and Minimally Invasive Surgery 2018 Wednesday, February 7, 2018 6:00 a.m. Registration & Breakfast with Exhibitors 6:55 a.m. Welcome Announcements SESSION: Practical
Program Schedule Update in Gynecology and Minimally Invasive Surgery 2018 Wednesday, February 7, 2018 6:00 a.m. Registration & Breakfast with Exhibitors 6:55 a.m. Welcome Announcements SESSION: Practical
CODING GUIDELINES FOR CONTRACEPTIVES. Effective June 1, 2017 Version 1.40
 CODING GUIDELINES FOR CONTRACEPTIVES Effective June 1, 2017 Version 1.40 TABLE OF CONTENTS ICD-10 CM Diagnosis Codes: Encounter for Contraception page 2 Coding for IUD Insertion and Removal Procedures
CODING GUIDELINES FOR CONTRACEPTIVES Effective June 1, 2017 Version 1.40 TABLE OF CONTENTS ICD-10 CM Diagnosis Codes: Encounter for Contraception page 2 Coding for IUD Insertion and Removal Procedures
Fibroid mapping. Haitham Hamoda MD FRCOG Consultant Gynaecologist, Subspecialist in Reproductive Medicine & Surgery King s College Hospital
 Fibroid mapping Haitham Hamoda MD FRCOG Consultant Gynaecologist, Subspecialist in Reproductive Medicine & Surgery King s College Hospital Fibroids Common condition >70% of women by onset of menopause.
Fibroid mapping Haitham Hamoda MD FRCOG Consultant Gynaecologist, Subspecialist in Reproductive Medicine & Surgery King s College Hospital Fibroids Common condition >70% of women by onset of menopause.
Radiological assessment of infertility: A pictorial review
 Radiological assessment of infertility: A pictorial review Poster No.: C-1681 Congress: ECR 2015 Type: Educational Exhibit Authors: J. P. Walsh, N. Healy, M. O'sullivan, S. Harte, M. T. Knox; Dublin/ IE
Radiological assessment of infertility: A pictorial review Poster No.: C-1681 Congress: ECR 2015 Type: Educational Exhibit Authors: J. P. Walsh, N. Healy, M. O'sullivan, S. Harte, M. T. Knox; Dublin/ IE
Advanced 3D Ultrasound Incorporating Fly Thru Virtual Imaging Promotes the Concept of Ultrasound Hysteroscopy
 Advanced 3D Ultrasound Incorporating Fly Thru Virtual Imaging Promotes the Concept of Ultrasound Hysteroscopy Bill Smith Clinical Diagnostics Services, London, UK Introduction Conventional hysteroscopy
Advanced 3D Ultrasound Incorporating Fly Thru Virtual Imaging Promotes the Concept of Ultrasound Hysteroscopy Bill Smith Clinical Diagnostics Services, London, UK Introduction Conventional hysteroscopy
PALM-COEIN: Your AUB Counseling Guide
 PALM-COEIN: Your AUB Counseling Guide 10 million+ Treat the cause, not the symptom In the U.S, more than 10 million women between the ages of 35 and 49 are affected by AUB 1 Diagnosis Cause Structural
PALM-COEIN: Your AUB Counseling Guide 10 million+ Treat the cause, not the symptom In the U.S, more than 10 million women between the ages of 35 and 49 are affected by AUB 1 Diagnosis Cause Structural
INFERTILITY CAUSES. Basic evaluation of the female
 INFERTILITY Infertility is the inability to conceive after 12 months of unprotected intercourse. There are multiple causes of infertility and a systematic way to evaluate the condition. Let s look at some
INFERTILITY Infertility is the inability to conceive after 12 months of unprotected intercourse. There are multiple causes of infertility and a systematic way to evaluate the condition. Let s look at some
CHAPTER 13 Gynaecological Procedures
 CHAPTER 13 Propunere noua clasificare proceduri folosind codificarea ICD-10-AM versiunea 3, 30 martie 2004 Gynaecological Procedures BLOCK 1240 Application, insertion or removal procedures on ovary 35518-00
CHAPTER 13 Propunere noua clasificare proceduri folosind codificarea ICD-10-AM versiunea 3, 30 martie 2004 Gynaecological Procedures BLOCK 1240 Application, insertion or removal procedures on ovary 35518-00
Accreditation Council for Graduate Medical Education
 Obstetrics And Gynecology Case Logs National Data Report Accreditation Council for Graduate Medical Education Prepared by: Department of Applications and Data Analysis I. National Resident Statistics Main
Obstetrics And Gynecology Case Logs National Data Report Accreditation Council for Graduate Medical Education Prepared by: Department of Applications and Data Analysis I. National Resident Statistics Main
Evaluation of the Infertile Couple
 Overview and Definition Infertility is defined as the inability of a couple to fall pregnant after one year of unprotected intercourse. Infertility is a very common condition as in any given year about
Overview and Definition Infertility is defined as the inability of a couple to fall pregnant after one year of unprotected intercourse. Infertility is a very common condition as in any given year about
Case Scenario 1. History
 History Case Scenario 1 A 53 year old white female presented to her primary care physician with post-menopausal vaginal bleeding. The patient is not a smoker and does not use alcohol. She has no family
History Case Scenario 1 A 53 year old white female presented to her primary care physician with post-menopausal vaginal bleeding. The patient is not a smoker and does not use alcohol. She has no family
Product Information. Confidence that lasts
 Confidence that lasts What is Mirena? Inhibition of sperm motility and function inside the uterus and the fallopian tubes, preventing fertilization (Videla-Rivero et al. 1987). Section of system Levonorgestrel
Confidence that lasts What is Mirena? Inhibition of sperm motility and function inside the uterus and the fallopian tubes, preventing fertilization (Videla-Rivero et al. 1987). Section of system Levonorgestrel
Examining Long-Acting Reversible Contraceptive Methods
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/examining-long-acting-reversible-contraceptivemethods/7078/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/examining-long-acting-reversible-contraceptivemethods/7078/
Investigating HMB- an evidence based approach
 BSGE Meeting: Contemporary management of heavy menstrual bleeding (HMB) in primary and secondary care: (7 th December 2018, RCOG) Investigating HMB- an evidence based approach T. Justin Clark MB ChB, MD(Hons),
BSGE Meeting: Contemporary management of heavy menstrual bleeding (HMB) in primary and secondary care: (7 th December 2018, RCOG) Investigating HMB- an evidence based approach T. Justin Clark MB ChB, MD(Hons),
Endometriosis. *Chocolate cyst in the ovary
 Endometriosis What is endometriosis? Endometriosis is a common condition in young women. It's chronic, painful, and it often progressively gets worse over the time. *Chocolate cyst in the ovary Normally,
Endometriosis What is endometriosis? Endometriosis is a common condition in young women. It's chronic, painful, and it often progressively gets worse over the time. *Chocolate cyst in the ovary Normally,
Intrauterine device penetrating the jejunum
 ISSN:-077219200- ISSN-(print): 2593-1431- Intrauterine device penetrating the jejunum Jacek Leszek Borowski 1, Maximilian Paprotny 1, Julia Borowska 1! Abstract Intrauterine devices are nowadays one of
ISSN:-077219200- ISSN-(print): 2593-1431- Intrauterine device penetrating the jejunum Jacek Leszek Borowski 1, Maximilian Paprotny 1, Julia Borowska 1! Abstract Intrauterine devices are nowadays one of
Prevention, Diagnosis and Treatment of Gynecologic Cancers
 Prevention, Diagnosis and Treatment of Gynecologic Cancers Jubilee Brown MD and Pamela T. Soliman MD, MPH Department of Gynecologic Oncology and Reproductive Medicine University of Texas MD Anderson Cancer
Prevention, Diagnosis and Treatment of Gynecologic Cancers Jubilee Brown MD and Pamela T. Soliman MD, MPH Department of Gynecologic Oncology and Reproductive Medicine University of Texas MD Anderson Cancer
The value of laparoscopy alone or combined with hysteroscopy in the treatment of interstitial pregnancy: analysis of 22 cases
 Arch Gynecol Obstet (2012) 285:727 732 DOI 10.1007/s00404-011-2060-1 GENERAL GYNECOLOGY The value of laparoscopy alone or combined with hysteroscopy in the treatment of interstitial pregnancy: analysis
Arch Gynecol Obstet (2012) 285:727 732 DOI 10.1007/s00404-011-2060-1 GENERAL GYNECOLOGY The value of laparoscopy alone or combined with hysteroscopy in the treatment of interstitial pregnancy: analysis
levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer
 levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer 6 March 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer 6 March 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
X-Plain Ovarian Cancer Reference Summary
 X-Plain Ovarian Cancer Reference Summary Introduction Ovarian cancer is fairly rare. Ovarian cancer usually occurs in women who are over 50 years old and it may sometimes be hereditary. This reference
X-Plain Ovarian Cancer Reference Summary Introduction Ovarian cancer is fairly rare. Ovarian cancer usually occurs in women who are over 50 years old and it may sometimes be hereditary. This reference
An Overview of Uterine Factors That Influence Implantation
 An Overview of Uterine Factors That Influence Implantation Bulent Urman, M.D. Dept. of Obstetrics and Gynecology Koc University School of Medicine Assisted Reproduction Unit, American Hospital, ISTANBUL
An Overview of Uterine Factors That Influence Implantation Bulent Urman, M.D. Dept. of Obstetrics and Gynecology Koc University School of Medicine Assisted Reproduction Unit, American Hospital, ISTANBUL
Sterilisation for women at the RD&E: what you need to know Reference Number: CW
 Sterilisation for women at the RD&E: what you need to know Royal Devon and Exeter NHS Foundation Trust Patient Information Sterilisation for Women at The Royal Devon and Exeter Hospital What you need to
Sterilisation for women at the RD&E: what you need to know Royal Devon and Exeter NHS Foundation Trust Patient Information Sterilisation for Women at The Royal Devon and Exeter Hospital What you need to
Hysterectomy. What is a hysterectomy? Why is hysterectomy done? Are there alternatives to hysterectomy?
 301.681.3400 OBGYNCWC.COM What is a hysterectomy? Hysterectomy Hysterectomy is surgery to remove the uterus. It is a very common type of surgery for women in the United States. Removing your uterus means
301.681.3400 OBGYNCWC.COM What is a hysterectomy? Hysterectomy Hysterectomy is surgery to remove the uterus. It is a very common type of surgery for women in the United States. Removing your uterus means
Application for inclusion of levonorgestrel - releasing IUD for contraception in the WHO Model List of Essential Medicines
 Application for inclusion of levonorgestrel - releasing IUD for contraception in the WHO Model List of Essential Medicines 1. Summary statement of the proposal for inclusion LNG-IUS is an effective contraceptive;
Application for inclusion of levonorgestrel - releasing IUD for contraception in the WHO Model List of Essential Medicines 1. Summary statement of the proposal for inclusion LNG-IUS is an effective contraceptive;
Surgery and Infertility
 Surgery and Infertility Dr Phill McChesney BHB MBChB FRANZCOG MRMed CREI Laparoscopy Prior to Considering IVF Diagnostic Tubal Surgery Treatment of peritubal adhesions Reconstructive surgery Sterilization
Surgery and Infertility Dr Phill McChesney BHB MBChB FRANZCOG MRMed CREI Laparoscopy Prior to Considering IVF Diagnostic Tubal Surgery Treatment of peritubal adhesions Reconstructive surgery Sterilization
Endometrial line thickness in different conditions.
 Endometrial line thickness in different conditions 1 Endometrial thickens in response to Rising estrogen levels during the menstrual cycle and then shedding endometrial at the times of menses 2 The thickens
Endometrial line thickness in different conditions 1 Endometrial thickens in response to Rising estrogen levels during the menstrual cycle and then shedding endometrial at the times of menses 2 The thickens
Fertility Assessment and Treatment Pathway
 Rejected referrals sent back to GP Fertility Assessment and Treatment Pathway Patients with fertility problems go to the GP GP Advice and Assessment GP to inform patient of access criteria for NHS-funded
Rejected referrals sent back to GP Fertility Assessment and Treatment Pathway Patients with fertility problems go to the GP GP Advice and Assessment GP to inform patient of access criteria for NHS-funded
One Thousand Cases of Infertility
 One Thousand Cases of Infertility Clinical Review of a Five-Year Series Robert B. Wilson, M.D. THE RECORDS of 1032 women who complained of infertility have been reviewed. These patients were seen by various
One Thousand Cases of Infertility Clinical Review of a Five-Year Series Robert B. Wilson, M.D. THE RECORDS of 1032 women who complained of infertility have been reviewed. These patients were seen by various
Heavy Menstrual Bleeding. Mr Nick Nicholas MD FRCOG Grad Dip Law. Consultant Gynaecologist
 Heavy Menstrual Bleeding Mr Nick Nicholas MD FRCOG Grad Dip Law. Consultant Gynaecologist Why is HMB so important? 1:20 women aged 30-49 consult their GP with HMB Once referred to gynaecologist, surgical
Heavy Menstrual Bleeding Mr Nick Nicholas MD FRCOG Grad Dip Law. Consultant Gynaecologist Why is HMB so important? 1:20 women aged 30-49 consult their GP with HMB Once referred to gynaecologist, surgical
VirtaMed GynoS hysteroscopy Module descriptions
 VirtaMed GynoS hysteroscopy Module descriptions VirtaMed AG Rütistr. 12, 8952 Zurich Switzerland info@virtamed.com www.virtamed.com Phone: +41 44 500 9690 Table of contents Table of contents... 1 Essential
VirtaMed GynoS hysteroscopy Module descriptions VirtaMed AG Rütistr. 12, 8952 Zurich Switzerland info@virtamed.com www.virtamed.com Phone: +41 44 500 9690 Table of contents Table of contents... 1 Essential
Hysteroscopic sterilization using a micro-insert device: results of a multicentre Phase II study*
 Human Reproduction Vol.18, No.6 pp. 1223±1230, 2003 DOI: 10.1093/humrep/deg256 Hysteroscopic sterilization using a micro-insert device: results of a multicentre Phase II study* John F.Kerin 1,8, Jay M.Cooper
Human Reproduction Vol.18, No.6 pp. 1223±1230, 2003 DOI: 10.1093/humrep/deg256 Hysteroscopic sterilization using a micro-insert device: results of a multicentre Phase II study* John F.Kerin 1,8, Jay M.Cooper
HYSTERECTOMY FOR BENIGN CONDITIONS
 UnitedHealthcare Commercial Medical Policy HYSTERECTOMY FOR BENIGN CONDITIONS Policy Number: 2018T0572G Effective Date: September 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
UnitedHealthcare Commercial Medical Policy HYSTERECTOMY FOR BENIGN CONDITIONS Policy Number: 2018T0572G Effective Date: September 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
PRETREATMENT ASSESSMENT & MANAGEMENT (MODULE 1 B) March, 2018
 PRETREATMENT ASSESSMENT & MANAGEMENT (MODULE 1 B) March, 2018 Clinical Assessment A thorough clinical evaluation is a prerequisite for ART A thorough clinical evaluation as detailed in the female and male
PRETREATMENT ASSESSMENT & MANAGEMENT (MODULE 1 B) March, 2018 Clinical Assessment A thorough clinical evaluation is a prerequisite for ART A thorough clinical evaluation as detailed in the female and male
16 East 40 th St, 2 nd Fl, New York, NY Ph fax
 Page 1 of 9 16 East 40 th St, 2 nd Fl, New York, NY 10016 Ph 212-679-2289 fax 212-679-2288 Please complete the following: Fertility Evaluation Name: Date of birth: Age: Partner s Name: Date of birth: Age:
Page 1 of 9 16 East 40 th St, 2 nd Fl, New York, NY 10016 Ph 212-679-2289 fax 212-679-2288 Please complete the following: Fertility Evaluation Name: Date of birth: Age: Partner s Name: Date of birth: Age:
An Evaluation of the PSP (Speck) Test for Tubal Patency. M. Edward Davis, M.D., Mildred E. Ward, M.D., and Albert G. King, M.D.
 An Evaluation of the PSP (Speck) Test for Tubal Patency M. Edward Davis, M.D., Mildred E. Ward, M.D., and Albert G. King, M.D. IN 1948 Speck described an ingenious procedure for the demonstration of tubal
An Evaluation of the PSP (Speck) Test for Tubal Patency M. Edward Davis, M.D., Mildred E. Ward, M.D., and Albert G. King, M.D. IN 1948 Speck described an ingenious procedure for the demonstration of tubal
Levosert levonorgestrel 20mcg/24hour intrauterine device
 Levosert levonorgestrel 20mcg/24hour intrauterine device Verdict: Formulary inclusion: Formulary category: Restrictions: Reason for inclusion: Link to formulary: Link to medicine review summary: Levosert
Levosert levonorgestrel 20mcg/24hour intrauterine device Verdict: Formulary inclusion: Formulary category: Restrictions: Reason for inclusion: Link to formulary: Link to medicine review summary: Levosert
