STUDIES IN HYPERTHERMIA II. THE ACID-BASE EQUILIBRIUM IN HYPERTHERMIA INDUCED BY SHORT RADIO WAVES
|
|
|
- Amos White
- 5 years ago
- Views:
Transcription
1 STUDIES IN HYPERTHERMIA II. THE ACIDBASE EQUILIBRIUM IN HYPERTHERMIA INDUCED BY SHORT RADIO WAVES BY FRITZ BISCHOFF, M. LOUISA LONG, AND ELSIE HILL (From the Chemical Laboratory of the Potter Metabolic Clinic, Santa Barbara Cottage Hospital, Santa Barbara) (Received for publication, October 30, 1930) INTRODUCTION In an earlier paper (1) the effect of a hyperthermia induced in human subjects by the high frequency electric current was studied. The effects of hyperthermia produced by short radio waves are reported in the present paper. The machine utilizing the high frequency electric current was in principle the same as the ordinary diathermy machine in general use, with the exception that it was designed to give an especially smooth high frequency wave to prevent the subject from suffering any disagreeable sensations due to passage of current. In the radio wave machine, the subject rests between the plates without any part of his body s being in contact with the plates so that the waves oscillate through the body from one side to the other. A detailed description of the machine used in our experiments has been given by Carpenter and Page (2). The present study was made to determine whether there was any difference in effect between the hyperthermia induced by radio waves and that induced by the diathermy principle or by the other well known methods of producing artificial fevers such as hot baths or vaccine injections. Carpenter and Page (2) believe... that the development of heat [by the short radio waves] is due to the resistance of the body to the conduction of current between the surfaces adjacent to the opposedplates. At each alternation of polarity of the plates the corresponding polarities are induced upon the adjacent boundaries of the interposed body and current is conducted through the material for a brief interval. The heating of solutions similar to the blood serum is dependent directly upon their electrical resistance. 321
2 322 Hyperthermia by Radio Waves If this explanation be accepted (we do not feel qualified to pass a critical judgment on the various theories) there would be little reason for supposing that the effect of the high frequency current (diathermy) would be different from the effect of the short radio wave. The explanation of Carpenter and Boak (3), however, led us to believe that there were theoretical grounds for a possible difference in physiological effect. These authors state, That such a rise in temperature accompanies the passage of the short radio waves through the organism in such a field may be explained in a number of ways, such as that it is conducting induced alternating currents and the heat developed is proportional to the current squared times the resistance of the body. Hosmer s conception is that the heating is due to the increased rate of vibration of the molecules of the cells produced by their alternate attraction to each of the plates in turn. Others consider it analogous to dielectric hysteresis, i.e., a resistance to the changing of the molecules. Plan of Experiment The plan of experimentation was essentially the same as that followed in our studies with the high frequency electric current. In addition, a study of the perspiration was made. Collection of PerspirationAn attempt was made to collect the constituents of the perspiration quantitatively for the entire heating period. The subject was washed with soap and water prior to heating and rinsed with distilled water. He was then wrapped in a sheet and placed upon a cotton blanket, under which was a rubber sheet. He was covered with a cotton blanket. The blankets, sheet, and rubber had previously been rinsed three times in distilled water. The blankets and sheet were dried in a clothes vacuum centrifuge. After the heating the sheets and blankets were washed four times in distilled water. The subject, as well as his hair, was washed with distilled water, gauze sponges being used. All the washings were combined, 1 cc. of concentrated sulfuric acid was added, and the solution was concentrated below 500 cc. Insoluble material was filtered off and washed with dilute acid. The total concentrate was made to a volume of 500 cc. The washings from the rubber were concentrated separately. The amount of perspiration collected from the rubber was not significant. The concentrate gave no precipitate with tungstic acid. Standard methods of blood analysis were used for the determination of non
3 F. Bischoff, M. L. Long, and E. Hill 323 protein, urea, ammonia, and creatinine nitrogen and uric acid. Ammonia was separated with permutit. AnalysesThe analytical methods were the same as described in a former paper. The ph, as previously described, was determined by the quinhydrone electrode upon the separated plasma of blood as drawn. The ph was determined at room temperatures. The ph values are reported corrected to the oral temperature of the patient, with the temperature coefficient Laug (4) has recently determined the temperature coefficient by the quinhydrone electrode as ph per degree for dog plasma. Our value (5) was obtained for two human plasma samples at 25 and 37. For rabbit plasma we obtained a coefficient of The value obtained depends somewhat upon the method of extrapolation of the drifting potential to zero time. For the present we are using our value of until more is known regarding the behavior of the electrode the first 15 seconds of the determination. EXPERIMENTAL SubjectsWe have not felt justified in raising the body temperature of normal individuals above 39. In the present series, one normal individual was heated on three occasions. The quantitative collection of perspiration was made for this individual. In addition two paretics were subjected to the hyperthermia. One of these showed an initial high temperature of Her temperature was raised to The changes in oral temperature, pulse rate, and respiration rate are given in the protocols. Blood Volume ChangesWith the increase in the oxygen capacity of the blood as a measure of volume change, an appreciable loss in blood volume during the radio wave hyperthermia is noted. Reductions in blood volume amounting to 3.5, 15.0, 6.5, and 7.0 per cent were noted. In hyperthermia induced by the high frequency electric current, the changes in the oxygen capacity approached the error of the analytical determination, increases of 1 and 2 per cent in blood volume being noted in three cases and decreases in 1 and 4 per cent in two others. The data for total hemoglobin are given in Table I. Blood ph and Alkali ReserveThere was a decided increase in the plasma ph in three of the experiments during the hyperthermia. Shifts of ph from 7.47 to 7.59, from 7.44 to 7.70, and
4 324 Hyperthermia by Radio Waves from 7.46 to 7.55 were noted. As the normal range for the plasma ph by the quinhydrone electrode is 7.40 to 7.50, the increases are above the upper limits of normal. In a fourth experiment, the ph remained within normal limits, shifting from 7.40 to TABLE Blood Changes Following Rise in Body Temperature The values for total Hb, HbOs, and total COz are measured in volumes per cent; those for urea N, nonprotein N, cholesterol, creatinine, uric acid, and amino acid N are measured in mg. per 100 cc. Subject A. I( L. D. Date 1950 Apr. 15 Sept. 23 July 12 June 7 Oral temperature C I. _ lasma PH $ TABLE I Whole blood findings II Changes in Distribution of Base Bound by Bu$ers of Blood During Heatin.g Subject A. L. D. A. ABHCOs AB(Hb) ABP,?nM pm 1. rnm per 2. mdb per ZAB per $0.9 4 correction was applied for the change in blood volume during heating. This subject had a fever at the time the experimental hyperthermia was induced. The significance of her condition will be discussed later. An appreciable fall in the whole blood total CO2 was noted in
5 F. Bischoff, M. L. Long, and E. Hill 325 the three experiments in which the plasma ph was markedly increased. A 3 volume per cent rise was noted for the subject whose plasma ph did not change during the hyperthermia. The hemoglobin became highly oxygenated in the three experiments in which the ph rose, and remained unchanged in the experiment in which the plasma ph was unchanged. In order to determine whether the fall in the total CO2 could be accounted for by a shift of base to the blood proteins in accordance with the increased ph, the base bound by the blood buffers has been calculated. The results are given in Table II. The method of calculation is given in an earlier paper (1). These calculations can be considered only semiquantitatively. In the present instance, they have been complicated by the changes in blood volume. Since the interest is TABLE Quantitative Estimation of Perspiratory Elimination of Subject A Nitrogen as nonprotein N Urea, NHIN... N... NHaN.... Creatinine N.... Amino acid N... P III 2.25 hr. period, hr. period, gm Less than 0.5 mg Less than 0.5 mg. primarily whether or not the alkali reserve as a whole is increased, the data obtained during the heating period have been corrected for changes in blood volume. The results indicate a slight increase in the alkali reserve. These results are in harmony with data obtained by us in hyperthermia induced by the high frequency electric current and by Cajori et al. (6), who used the electric bake and determined the change in alkali reserve by absorption curves. Cajori obtained increases ranging from 0 to 6.0 millimols per liter. Nitrogenous Blood ConstituentsA slight increase in the blood nonprotein nitrogen and urea nitrogen during hyperthermia was noted. The increases were comparable to the change in the oxygen capacity. Increases of 1.8 and 1.3 mg. per 100 cc. for urea nitrogen
6 326 Hyperthermia by Radio Waves and 3.3 mg. per 100 cc. for nonprotein nitrogen were observed. There was no change and a slight decrease in the amino acid nitrogen for two cases studied. Changes noted in blood uric acid and creatinine were within the error of the analytical determination. There was likewise no change in the whole blood cholesterol. The data are given in Table I. Nitrogenous Constituents of PerspirationThe high percentage of NH3 nitrogen as compared with the total nitrogen is perhaps the most interesting observation of the perspiration analyses. The sum of the urea, ammonia, creatinine, and amino acid nitrogen values does not account for the nonprotein nitrogen. The data are given in Table III. Urine phin the present study the urine ph was followed at intervals for the preheating, heating, cooling, and recovery periods for three individuals. There was no indication of the urine s becoming alkaline during the period of increased alkalinity of the blood. There was a tendency of the ph to become more acid during the recovery period. The results are given in Table II of the companion paper (7). DISCUSSION Concerning the effect upon the acidbase equilibrium, there is apparently no difference whether the rise in temperature is induced by the external application of heat (hot water or air baths) or by the generation of heat within the body (high frequency current or radio wave). Of fundamental importance is the loss of COS with rise in the blood ph. The fall in the CO2 content of the blood is readily accounted for by a shift of base to the blood proteins due to the increase in ph. Cajori et al. (6) found no significant change in the blood volume as measured by the oxygen capacity in their studies with the electric bake. We found none in our studies with the high frequency electric current, though the concentration in blood volume following the radio wave hyperthermia was significant. When the high frequency current was used it was necessary to insulate the subject from loss of heat by wrapping him in blankets. In the radio wave series, the effect of the machine was so powerful that the subject was covered with only a thin cotton blanket. It is probable that in the radio wave experiments there was a greater evaporation of
7 F. Bischoff, M. L. Long, and E. Hill 327 perspiration, while with the high frequency current the subject became bathed in his own perspiration. Since the radio wave hyperthermia is now under investigation for possible clinical use, a warning is sounded as to the change in blood volume it may bring about. In our experiments the subjects drank over a liter of water during the heating and still showed a fall in blood volume. Donath and Heilig (8) have divided hyperthermias into two classes: those which bring about an increase in the blood amino acid nitrogen with increased nitrogen excretion in the urine and those which do not affect these constituents. According to this classification the radio wave hyperthermia is analogous to the manipulation of the heat centers, which brings about no changes, in contrast to the injection of nucleic acids or vaccines. The present series of experiments have confirmed our impression that there is little reason for believing that the body is attempting to compensate for the lowered CO, tension by lowering the alkali reserve. In experiments with forced breathing and those in which the body temperature is raised very rapidly by immersion in hot water, very alkaline urines and falls in urine ammonia are observed. In our series with the high frequency electric current, some alkaline urines were observed, but they were in no case as alkaline as the blood. In the present series the urine ph at the peak of the heating was 5.0, 6.2, and 5.8. Moreover, the ratio of ammonia to urea in the perspiration increased with rise of temperature and the urine ph became more acid during the cooling period. The latter observation overrules the possibility that the kidneys may have been unable to secrete alkali during the hyperthermia because of the decrease in urine volume. The findings of the experiment in which the subject had a fever (37.8 ) at the time the artificial hyperthermia was induced were unexpected. Her initial plasma ph was within the lower limits of normal. The induced 2 rise in temperature brought about no significant blood changes with the exception of reduction of blood volume. This subject was given a small dose of morphine before the experiment. It is possible that either the morphine or the paretic condition inhibited the normal response of the heat centers. Since another paretic responded normally the condition is apparently not characteristic of paresis. It is not in the scope of the present study to investigate pathological conditions. The
8 328 Hyperthermia by Radio Waves results suggest that studies of hyperthermia induced in various pathological conditions would help materially in elucidating the mechanism of heat regulation. A study of the nature reported in this paper is possible only with the cooperation of a large number of people. We are indebted to Dr. H. J. Ullmann for the clinical supervision of the problem, to Dr. R.. F. Atsatt, who was the normal subject studied, to Dr. N. H. Brush for supplying us with suitable human subjects for study, and to Miss Ella M. Ottery for the care of the subjects. Mr. Carl Darnell of the General Electric Company was responsible for the radio wave machine placed at our disposal. SUMMARY With the exception of a fall in blood volume, which is probably not a direct effect, no difference in effect was noted between raising the body temperat.ure by placing the subject in the field of condenser plates in circuit with a short wave radio transmitter and in raising the body temperature by means of diathermy, warmed air, or hot water baths. Of fundamental importance was the loss of coz. The ph of the blood became more alkaline, there was a shift of bases to the blood proteins and an increased oxygenation of t.he hemoglobin of venous blood. No significant change in the nonprotein nitrogen constituents of the blood was noted. No evidence was obtained that the body was attempting to compensate for the condition of alkalosis through the urinary or perspiratory excretions. BIBLIOGRAPHY 1. Bischoff, F., Ullmann, H. J., Hill, E., and Long, M. L., J. Biol. Chem., 86,675 (1930). 2. Carpenter, C. M., and Page, A. B., Science, 71,450 (1930). 3. Carpenter, C. M., and Boak, R. A., Am. J. Syphilis, 14,346 (1930). 4. Laug, E. P., J. Biol. Chem., 88,551 (1930). 5. Bischoff, F., Long, M. L., and Hill, E., J. Pharmacol. and Ezp. Therap., 39,425 (1930). 6. Cajori, F. A., Crouter, C. Y., and Pemberton, R., J. BioZ. Chem., 67, 217 (1923). 7. Bischoff, F., Maxwell, L. C., and Hill, E., J. BioZ. Chem., SO, 331 (1931). 8. Donath, J. D., and Heilig, R., KZin. Woch., 3,834 (1924).
9 F. Bischoff, M. L. Long, and E. Hill 329 Protocols T = oral temperature in C., P = pulse rate, R = respiration rate. Time 1 T I P I R Time I T 1 P I R p.m. 7.15* % A., male, Apr. 15, 1930 t A., male, July, *? A., male, Sept. 23, 1930 p.m. 7.50*t *t L., male, July 12, *t *t * Taking of blood. t Beginning and termination of radio wave heating. D., female. June 7, 1930 p.m. 2.30*f * t 38.c ! 39.f 38.f 37.f 37.t 38.:
10 STUDIES IN HYPERTHERMIA: II. THE ACIDBASE EQUILIBRIUM IN HYPERTHERMIA INDUCED BY SHORT RADIO WAVES Fritz Bischoff, M. Louisa Long and Elsie Hill J. Biol. Chem. 1931, 90: Access the most updated version of this article at Alerts: When this article is cited When a correction for this article is posted Click here to choose from all of JBC's alerts This article cites 0 references, 0 of which can be accessed free at ml#reflist1
THE EFFECT OF ROENTGEN RADIATION UPON THE REACTION OF THE FLUID OF RAT SARCOMA 10
 THE EFFECT OF ROENTGEN RADIATION UPON THE REACTION OF THE FLUID OF RAT SARCOMA 10 L. C. MAXWELL AND H. J. ULLMANN WITH THE TECHNICAL ASSISTANCE ELLA MAY OTTERY OF (From the Department oj Cancer Research,
THE EFFECT OF ROENTGEN RADIATION UPON THE REACTION OF THE FLUID OF RAT SARCOMA 10 L. C. MAXWELL AND H. J. ULLMANN WITH THE TECHNICAL ASSISTANCE ELLA MAY OTTERY OF (From the Department oj Cancer Research,
A STUDY OF THE METABOLISM OF THEOBROMINE, THEOPHYLLINE, AND CAFFEINE IN MAN* Previous studies (1, 2) have shown that after the ingestion of caffeine
 A STUDY OF THE METABOLISM OF THEOBROMINE, THEOPHYLLINE, AND CAFFEINE IN MAN* BY HERBERT H. CORNISH AND A. A. CHRISTMAN (From the Department of Biological Chemistry, Medical School, University of Michigan,
A STUDY OF THE METABOLISM OF THEOBROMINE, THEOPHYLLINE, AND CAFFEINE IN MAN* BY HERBERT H. CORNISH AND A. A. CHRISTMAN (From the Department of Biological Chemistry, Medical School, University of Michigan,
THE EXCRETION OF BORIC ACID FROM THE HUMAN BODY.
 THE EXCRETION OF BORIC ACID FROM THE HUMAN BODY. BY HARVEY W. WILEY, M.D. (From the Bureau of Chemistry, Washington, D. C.) (Received for publication, December 15, 1906.) In the studies which I have inaugurated
THE EXCRETION OF BORIC ACID FROM THE HUMAN BODY. BY HARVEY W. WILEY, M.D. (From the Bureau of Chemistry, Washington, D. C.) (Received for publication, December 15, 1906.) In the studies which I have inaugurated
METABOLISM OF d-mannohepttjlose. EXCRETION OF THE SUGAR AFTER EATING AVOCADO
 METABOLISM OF d-mannohepttjlose. EXCRETION OF THE SUGAR AFTER EATING AVOCADO BY N. R. BLATHERWICK, HARDY W. LARSON, AND SUSAN D. SAWYER (From the Biochemical Laboratory of the Metropolitan Life Insurance
METABOLISM OF d-mannohepttjlose. EXCRETION OF THE SUGAR AFTER EATING AVOCADO BY N. R. BLATHERWICK, HARDY W. LARSON, AND SUSAN D. SAWYER (From the Biochemical Laboratory of the Metropolitan Life Insurance
CURVE OF SUGAR EXCRETION IN SEVERE DIABETES.
 CURVE OF SUGAR EXCRETION IN SEVERE DIABETES. BY HANNAH FELSHER. (From the Otho S. A. Sprague Memorial Institute Laboratory oj Clinical Research, Rush Medical College, Chicago.) (Received for publication,
CURVE OF SUGAR EXCRETION IN SEVERE DIABETES. BY HANNAH FELSHER. (From the Otho S. A. Sprague Memorial Institute Laboratory oj Clinical Research, Rush Medical College, Chicago.) (Received for publication,
PYRROLE AS A CATALYST FOR CERTAIN BIOLOGICAL OXIDATIONS
 PYRROLE AS A CATALYST FOR CERTAIN BIOLOGICAL OXIDATIONS BY FREDERICK BERNHEIM AND MARY L. C. BERNHEIM* (From the Departments of Physiology and Biochemistry, Duke University School of Medicine, Durham)
PYRROLE AS A CATALYST FOR CERTAIN BIOLOGICAL OXIDATIONS BY FREDERICK BERNHEIM AND MARY L. C. BERNHEIM* (From the Departments of Physiology and Biochemistry, Duke University School of Medicine, Durham)
BY W. W. SWANSON. (Prom the Biochemical Laboratory, Department of Physiology, University of Minnesota, Minneapolis.)
 THE EFFECT OF SODIUM BENZOATE INGESTION UPON THE COMPOSITION OF THE BLOOD AND URINE WITH ESPECIAL REFERENCE TO THE POSSIBLE SYNTHESIS OF GLYCINE IN THE BODY. PRELIMINARY PAPER. BY W. W. SWANSO (Prom the
THE EFFECT OF SODIUM BENZOATE INGESTION UPON THE COMPOSITION OF THE BLOOD AND URINE WITH ESPECIAL REFERENCE TO THE POSSIBLE SYNTHESIS OF GLYCINE IN THE BODY. PRELIMINARY PAPER. BY W. W. SWANSO (Prom the
THE EFFECT OF DENATURATION ON THE VISCOSITY OF PROTEIN SYSTEMS BY M. L. ANSON A~D A. E. MIRSKY. (Accepted for publication, December 2, 1931)
 THE EFFECT OF DENATURATION ON THE VISCOSITY OF PROTEIN SYSTEMS BY M. L. ANSON A~D A. E. MIRSKY (From tke Laboratories of The Rockefeller Institute for Medical Research, Princeton, N. Y., and the ttospital
THE EFFECT OF DENATURATION ON THE VISCOSITY OF PROTEIN SYSTEMS BY M. L. ANSON A~D A. E. MIRSKY (From tke Laboratories of The Rockefeller Institute for Medical Research, Princeton, N. Y., and the ttospital
THE EFFECT OF ANTICOAGULANTS ON DETERMINA- TIONS OF INORGANIC PHOSPHATE AND PROTEIN IN PLASMA BY OLIVER HENRY GAEBLER
 THE EFFECT OF ANTICOAGULANTS ON DETERMINA TIONS OF INORGANIC PHOSPHATE AND PROTEIN IN PLASMA BY OLIVER HENRY GAEBLER (From the Department of Laboratories, Henry Ford Hospital, Detroit) (Received for publication,
THE EFFECT OF ANTICOAGULANTS ON DETERMINA TIONS OF INORGANIC PHOSPHATE AND PROTEIN IN PLASMA BY OLIVER HENRY GAEBLER (From the Department of Laboratories, Henry Ford Hospital, Detroit) (Received for publication,
Cushny(4) has shown, however, that the amount of urea in the kidney. by some vital process, retain those diflusible substances which are of
 THE FUNCTION OF THE TUBULES IN KIDNEY EXCRETION. BY E. B. MAYRS. (From the Department of Pharmacology, Edinburgh.) IT is becoming generally recognised that filtration through the glomeruli and some degree
THE FUNCTION OF THE TUBULES IN KIDNEY EXCRETION. BY E. B. MAYRS. (From the Department of Pharmacology, Edinburgh.) IT is becoming generally recognised that filtration through the glomeruli and some degree
THE CARBOHYDRATE METABOLISM OF TUMORS.
 THE CARBOHYDRATE METABOLISM OF TUMORS. II. CHANGES IN THE SUGAR, LACTIC ACID, AND CO COMBINING POWER OF BLOOD PASSING THROUGH A TUMOR. BY CARL F. CORI AND GERTY T. CORI. (From the State Institute for ihe
THE CARBOHYDRATE METABOLISM OF TUMORS. II. CHANGES IN THE SUGAR, LACTIC ACID, AND CO COMBINING POWER OF BLOOD PASSING THROUGH A TUMOR. BY CARL F. CORI AND GERTY T. CORI. (From the State Institute for ihe
(Received for publication, February 26, 1945)
 TRYPTOPHANE METABOLISM XI. CHANGES IN THE CONTENT OF TRYPTQPHANE, UREA, AND NON PROTEIN NITROGEN IN THE BLOOD AND OF TRYPTOPHANE IN THE TISSUES AFTER THE ORAL ADMINISTRATION OF TRYPTOPHANE OR TRYPTOPHANE
TRYPTOPHANE METABOLISM XI. CHANGES IN THE CONTENT OF TRYPTQPHANE, UREA, AND NON PROTEIN NITROGEN IN THE BLOOD AND OF TRYPTOPHANE IN THE TISSUES AFTER THE ORAL ADMINISTRATION OF TRYPTOPHANE OR TRYPTOPHANE
A MICRO TIME METHOD FOR DETERMINATION OF REDUCING SUGARS, AND ITS APPLICATION TO ANALYSIS OF BLOOD AND URINE.
 A MICRO TIME METHOD FOR DETERMINATION OF REDUCING SUGARS, AND ITS APPLICATION TO ANALYSIS OF BLOOD AND URINE. BY JAMES A. HAWKINS. (From Ike Hospital of The Rockefeller Institute for Medical Research,
A MICRO TIME METHOD FOR DETERMINATION OF REDUCING SUGARS, AND ITS APPLICATION TO ANALYSIS OF BLOOD AND URINE. BY JAMES A. HAWKINS. (From Ike Hospital of The Rockefeller Institute for Medical Research,
DETERMINATION OF ACETONE IN EXPIRED AIR.*
 DETERMINATION OF ACETONE IN EXPIRED AIR.* BY ROGER S. HUBBARD. (From the Laboratory of the Clifton Springs SanitarizLm, Clifton Springs.) (Received for publication, June 1, 1920.) The fact that acetone
DETERMINATION OF ACETONE IN EXPIRED AIR.* BY ROGER S. HUBBARD. (From the Laboratory of the Clifton Springs SanitarizLm, Clifton Springs.) (Received for publication, June 1, 1920.) The fact that acetone
II. THE EFFECT OF THE INGESTION OF GLYCINE ON THE EXCRETION OF ENDOGENOUS URIC ACID.
 PURINE METABOLISM. II. THE EFFECT OF THE INGESTION OF GLYCINE ON THE EXCRETION OF ENDOGENOUS URIC ACID. BY A. A. CHRISTMAN AND E. C. MOSIER. (From the Laboratory of Physiological Chemistry, Medical School,
PURINE METABOLISM. II. THE EFFECT OF THE INGESTION OF GLYCINE ON THE EXCRETION OF ENDOGENOUS URIC ACID. BY A. A. CHRISTMAN AND E. C. MOSIER. (From the Laboratory of Physiological Chemistry, Medical School,
for Medical Research. (Received May 10th, 1922.)
 XLV. NOTE ON URINARY TIDES AND EXCRETORY RHYTHM. BY JAMES ARGYLL CAMPBELL AND THOMAS ARTHUR WEBSTER. From the Department of Applied Physiology, National Institute for Medical Research. (Received May 10th,
XLV. NOTE ON URINARY TIDES AND EXCRETORY RHYTHM. BY JAMES ARGYLL CAMPBELL AND THOMAS ARTHUR WEBSTER. From the Department of Applied Physiology, National Institute for Medical Research. (Received May 10th,
JEROME, M.B. (Lond.), Lecturer in Medical Pharmacology and Materia Medica. (From the Pharmacological Department, Oxford.)
 THE RELATION OF THE DEGREE OF ACIDITY OF THE URINE AND THE PERCENTAGE OF URIC ACID THEREIN CONTAINED TO THE PRECIPI- TATION OF THE LATTER IN THE FORM OF URIC ACID GRAVEL. BY WILLIAM J. SMITH JEROME, M.B.
THE RELATION OF THE DEGREE OF ACIDITY OF THE URINE AND THE PERCENTAGE OF URIC ACID THEREIN CONTAINED TO THE PRECIPI- TATION OF THE LATTER IN THE FORM OF URIC ACID GRAVEL. BY WILLIAM J. SMITH JEROME, M.B.
THE DETERMINATION OF ACETONE BODIES IN BLOOD AND URINE.
 THE DETERMINATION OF ACETONE BODIES IN BLOOD AND URINE. REPLY TO CRITICISMS BY E. C. SMITH. BY DONALD D. VAN SLYKE. (From the Hospital of The Rockefeller Institute jar Medical Research, New York.) (Received
THE DETERMINATION OF ACETONE BODIES IN BLOOD AND URINE. REPLY TO CRITICISMS BY E. C. SMITH. BY DONALD D. VAN SLYKE. (From the Hospital of The Rockefeller Institute jar Medical Research, New York.) (Received
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
PREPARATION OF LIPIDE EXTRACTS FROM BRAIN TISSUE*
 PREPARATION OF LIPIDE EXTRACTS FROM BRAIN TISSUE* JORDI FOLCH, I. ASCOLI, M. LEES,? J. A. MEATH,$ AND F. N. LEBARON (From the McLean Hospital Research Laboratories, Waverley, Massachusetts, and the Department
PREPARATION OF LIPIDE EXTRACTS FROM BRAIN TISSUE* JORDI FOLCH, I. ASCOLI, M. LEES,? J. A. MEATH,$ AND F. N. LEBARON (From the McLean Hospital Research Laboratories, Waverley, Massachusetts, and the Department
GLUCOSE is the most important diffusible substance in the blood which
 ON THE ACTION OF PHLORHIZIN ON THE KIDNEY. By E. B. MAYRS. (From the Department of Pharmacology, Edinburgh.) GLUCOSE is the most important diffusible substance in the blood which is completely held back
ON THE ACTION OF PHLORHIZIN ON THE KIDNEY. By E. B. MAYRS. (From the Department of Pharmacology, Edinburgh.) GLUCOSE is the most important diffusible substance in the blood which is completely held back
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Observing Respiration
 Chapter 9 Cellular Respiration Design an Experiment Observing Respiration Introduction Cellular respiration occurs in all living things. During this process, animals take in oxygen and release carbon dioxide
Chapter 9 Cellular Respiration Design an Experiment Observing Respiration Introduction Cellular respiration occurs in all living things. During this process, animals take in oxygen and release carbon dioxide
University College, London.)
 6I2.I2I:547.472*3 LACTIC ACID FORMATION AND REMOVAL WITH CHANGE OF BLOOD REACTION. BY M. GRACE EGGLETON1 AND C. LOVATT EVANS. (From the Department of Physiology and Biochemistry, University College, London.)
6I2.I2I:547.472*3 LACTIC ACID FORMATION AND REMOVAL WITH CHANGE OF BLOOD REACTION. BY M. GRACE EGGLETON1 AND C. LOVATT EVANS. (From the Department of Physiology and Biochemistry, University College, London.)
XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES.
 XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES. II. THE PREPARATION OF GLUCOSE UREIDE. BY ALEXANDER HYND. From the Department of Physiology, University of St Andrews. (Received
XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES. II. THE PREPARATION OF GLUCOSE UREIDE. BY ALEXANDER HYND. From the Department of Physiology, University of St Andrews. (Received
skin in individuals whose activity was minimal Two normal adult male medical students were the and maintenance calories. The intake of tap water was
 MINIMAL SODIUM LOSSES THROUGH THE SKIN1 By KENNETH D. ARN AND ANN REIMER (From the Department of Internal Medicine, The Medical School, University of Michigan, Ann Arbor, Mich.) (Submitted for publication
MINIMAL SODIUM LOSSES THROUGH THE SKIN1 By KENNETH D. ARN AND ANN REIMER (From the Department of Internal Medicine, The Medical School, University of Michigan, Ann Arbor, Mich.) (Submitted for publication
Objectives. Blood Buffers. Definitions. Strong/Weak Acids. Fixed (Non-Volatile) Acids. Module H Malley pages
 Blood Buffers Module H Malley pages 120-126 Objectives Define a buffer system and differentiate between the buffering systems present in the body. Given an arterial blood-gas result, determine the degree
Blood Buffers Module H Malley pages 120-126 Objectives Define a buffer system and differentiate between the buffering systems present in the body. Given an arterial blood-gas result, determine the degree
M6ller, McIntosh and Van Slyke (5) has been employed. The cases. changes in functional activity. Indications suggesting that such changes
 STUDIES OF UREA EXCRETION. VIII. THE EFFECTS ON THE UREA CLEARANCE OF CHANGES IN PROTEIN AND SALT CONTENTS OF THE DIET BY CUTHBERT L. COPE I (From the Hospital of the Rockefeller Institute for Medical
STUDIES OF UREA EXCRETION. VIII. THE EFFECTS ON THE UREA CLEARANCE OF CHANGES IN PROTEIN AND SALT CONTENTS OF THE DIET BY CUTHBERT L. COPE I (From the Hospital of the Rockefeller Institute for Medical
STUDIES ON THE MECHANISM OF NITROGEN STORAGE
 STUDIES ON THE MECHANISM OF NITROGEN STORAGE VI. RATE OF PROTEIN SYNTHESIS AND SIZE OF THE NITROGEN POOL* BY PAUL D. BARTLETT AND OLIVER H. GAEBLER WITH THE TECHNICAL ASSISTANCE OF BEVERLY CADY (From the
STUDIES ON THE MECHANISM OF NITROGEN STORAGE VI. RATE OF PROTEIN SYNTHESIS AND SIZE OF THE NITROGEN POOL* BY PAUL D. BARTLETT AND OLIVER H. GAEBLER WITH THE TECHNICAL ASSISTANCE OF BEVERLY CADY (From the
THE FATE OF SUGAR IN THE ANIMAL
 THE FATE OF SUGAR IN THE ANIMAL BODY. III. THE RATE OF GLYCOGEN FORMATION IN THE LIVER OF NORMAL AND INSULINIZED RATS DURING THE ABSORP- TION OF GLUCOSE, FRUCTOSE, AND GALACTOSE. BY CARL F. CORI. (From
THE FATE OF SUGAR IN THE ANIMAL BODY. III. THE RATE OF GLYCOGEN FORMATION IN THE LIVER OF NORMAL AND INSULINIZED RATS DURING THE ABSORP- TION OF GLUCOSE, FRUCTOSE, AND GALACTOSE. BY CARL F. CORI. (From
THE ASSIMILATION OF AMMONIA NITROGEN BY THE TOBACCO PLANT: A PRELIMINARY STUDY WITH ISOTOPIC NITROGEN. (Received for publication, July 3, 1940)
 THE ASSIMILATION OF AMMONIA NITROGEN BY THE TOBACCO PLANT: A PRELIMINARY STUDY WITH ISOTOPIC NITROGEN BY HUBERT BRADFORD VICKERY AND GEORGE W. PUCHER (Prom the Biochemical Laboratory of the Connecticut
THE ASSIMILATION OF AMMONIA NITROGEN BY THE TOBACCO PLANT: A PRELIMINARY STUDY WITH ISOTOPIC NITROGEN BY HUBERT BRADFORD VICKERY AND GEORGE W. PUCHER (Prom the Biochemical Laboratory of the Connecticut
CHEMICAL AND METABOLIC STUDIES ON PHENYLALANINE
 CHEMICAL AND METABOLIC STUDIES ON PHENYLALANINE II. THE PHENYLALANINE CONTENT OF THE BLOOD AND SPINAL FLUID IN PHENYLPYRUVIC OLIGOPHRENIA* BY GEORGE A. JERVIS, RfCHARD J. BLOCK, DIANA BOLLING, AND EDNA
CHEMICAL AND METABOLIC STUDIES ON PHENYLALANINE II. THE PHENYLALANINE CONTENT OF THE BLOOD AND SPINAL FLUID IN PHENYLPYRUVIC OLIGOPHRENIA* BY GEORGE A. JERVIS, RfCHARD J. BLOCK, DIANA BOLLING, AND EDNA
BCH 447. Estimation of Serum Urea
 BCH 447 Estimation of Serum Urea 1 Objective: Estimation of Blood urea nitrogen (BUN) in serum sample. 2 -Urea: Urea is the highest non-protein nitrogen compound in the blood. Urea is the major excretory
BCH 447 Estimation of Serum Urea 1 Objective: Estimation of Blood urea nitrogen (BUN) in serum sample. 2 -Urea: Urea is the highest non-protein nitrogen compound in the blood. Urea is the major excretory
The respiratory quotient is the relation by volume of the carbon dioxide DEXTROSE AND LE VULOSE IN HUMANS
 415 THE METABOLIC EFFECT OF ENEMA TA OF ALCOHOL, DEXTROSE AND LE VULOSE IN HUMANS By THORNZ, M. CARPZNTBR NUTRITION LABORATORY OF THU CARNEGIS INSTITUTION OF WASHINGTON, BOSTON, MASS. Communicated April
415 THE METABOLIC EFFECT OF ENEMA TA OF ALCOHOL, DEXTROSE AND LE VULOSE IN HUMANS By THORNZ, M. CARPZNTBR NUTRITION LABORATORY OF THU CARNEGIS INSTITUTION OF WASHINGTON, BOSTON, MASS. Communicated April
HISTAMINE AND PROTEOLYTIC ENZYMES. (Received for publication, March 31, 1943)
 HISTAMINE AND PROTEOLYTIC ENZYMES LIBERATION OF HISTAMINE BY PAPAIN BY M. ROCHA E SILVA AND SYLVIA 0. ANDRADE (From the Department of Biochemistry and Pharmacodynamics, Instituto Biologico, &io Paulo,
HISTAMINE AND PROTEOLYTIC ENZYMES LIBERATION OF HISTAMINE BY PAPAIN BY M. ROCHA E SILVA AND SYLVIA 0. ANDRADE (From the Department of Biochemistry and Pharmacodynamics, Instituto Biologico, &io Paulo,
April 08, biology 2201 ch 11.3 excretion.notebook. Biology The Excretory System. Apr 13 9:14 PM EXCRETORY SYSTEM.
 Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
Organic Molecule Composition of Milk: Lab Investigation
 Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
CRYSTALLINE PEPSIN V. ISOLATION OF CRYSTALLINE PEPSIN FROM BOVINE GASTRIC JUICE BY JOHN H. NORTHROP
 CRYSTALLINE PEPSIN V. ISOLATION OF CRYSTALLINE PEPSIN FROM BOVINE GASTRIC JUICE BY JOHN H. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton, N. J.) (Accepted
CRYSTALLINE PEPSIN V. ISOLATION OF CRYSTALLINE PEPSIN FROM BOVINE GASTRIC JUICE BY JOHN H. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton, N. J.) (Accepted
PURIFICATION OF PROTHROMBIN AND THROMBIN : CHEMICAL PROPERTIES OF PURIFIED PREPARATIONS*
 PURIFICATION OF PROTHROMBIN AND THROMBIN : CHEMICAL PROPERTIES OF PURIFIED PREPARATIONS* BY WALTER H. SEEGERS (Prom the Department of Pathology, State University of Zowa, Iowa City) (Received for publication,
PURIFICATION OF PROTHROMBIN AND THROMBIN : CHEMICAL PROPERTIES OF PURIFIED PREPARATIONS* BY WALTER H. SEEGERS (Prom the Department of Pathology, State University of Zowa, Iowa City) (Received for publication,
Excretion (IGCSE Biology Syllabus )
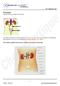 Excretion (IGCSE Biology Syllabus 2016-2018) Structure of the Kidney Excretion is the removal from organisms of toxic materials, the waste products of metabolism and substances in excess of requirements
Excretion (IGCSE Biology Syllabus 2016-2018) Structure of the Kidney Excretion is the removal from organisms of toxic materials, the waste products of metabolism and substances in excess of requirements
THE SPARING ACTION OF FAT ON VITAMIN B
 THE SPARING ACTION OF FAT ON VITAMIN B VI. THE INFLUENCE OF THE LEVELS OF PROTEIN AND VITAMIN G BY HERBERT M. EVANS, SAMUEL LEPKOVSKY, AND ELIZABETH A. MURPHY (From the Institute of Experimental Biology,
THE SPARING ACTION OF FAT ON VITAMIN B VI. THE INFLUENCE OF THE LEVELS OF PROTEIN AND VITAMIN G BY HERBERT M. EVANS, SAMUEL LEPKOVSKY, AND ELIZABETH A. MURPHY (From the Institute of Experimental Biology,
ON THE COMPOSITION OF URINARY ALBUMIN.* BY FLORENTIN MEDIGRECEANU.
 ON THE COMPOSITION OF URINARY ALBUMIN.* BY FLORENTIN MEDIGRECEANU. (From the Hospital of the Rockefeller Institute for Medical Research, New York.) Since the studies of Brown-Sequard, Teissier, L. Brunton,
ON THE COMPOSITION OF URINARY ALBUMIN.* BY FLORENTIN MEDIGRECEANU. (From the Hospital of the Rockefeller Institute for Medical Research, New York.) Since the studies of Brown-Sequard, Teissier, L. Brunton,
THE ISOLATION OF A MUCOPOLYSACCHARIDE FROM SYNOVIAL FLUID*
 THE ISOLATION OF A MUCOPOLYSACCHARIDE FROM SYNOVIAL FLUID* BY KARL MEYER, ELIZABETH M. SMYTH, AND MARTIN H. DAWSON (From the Department of Ophthalmology, College of Physicians and Surgeons, Columbia University,
THE ISOLATION OF A MUCOPOLYSACCHARIDE FROM SYNOVIAL FLUID* BY KARL MEYER, ELIZABETH M. SMYTH, AND MARTIN H. DAWSON (From the Department of Ophthalmology, College of Physicians and Surgeons, Columbia University,
Chapter 16. Urinary System and Thermoregulation THERMOREGULATION. Homeostasis
 Homeostasis Chapter 16 Urinary System and Thermoregulation! Homeostasis Maintenance of steady internal conditions despite fluctuations in the external environment! Examples of homeostasis Thermoregulation
Homeostasis Chapter 16 Urinary System and Thermoregulation! Homeostasis Maintenance of steady internal conditions despite fluctuations in the external environment! Examples of homeostasis Thermoregulation
FREEZING POINTS OF ANTI-COAGULANT SALT SOLUTIONS
 Published Online: 20 March, 1935 Supp Info: http://doi.org/10.1085/jgp.18.4.485 Downloaded from jgp.rupress.org on October 21, 2018 FREEZING POINTS OF ANTI-COAGULANT SALT SOLUTIONS B~ DAVID I. HITCI~OCK
Published Online: 20 March, 1935 Supp Info: http://doi.org/10.1085/jgp.18.4.485 Downloaded from jgp.rupress.org on October 21, 2018 FREEZING POINTS OF ANTI-COAGULANT SALT SOLUTIONS B~ DAVID I. HITCI~OCK
I. Metabolic Wastes Metabolic Waste:
 I. Metabolic Wastes Metabolic Waste: a) Carbon Dioxide: by-product of cellular respiration. b) Water: by-product of cellular respiration & dehydration synthesis reactions. c) Inorganic Salts: by-product
I. Metabolic Wastes Metabolic Waste: a) Carbon Dioxide: by-product of cellular respiration. b) Water: by-product of cellular respiration & dehydration synthesis reactions. c) Inorganic Salts: by-product
necessity for an investigation into possible different types of urine acidity. In
 456 J. Physiol. (I947) io6, 456-465 6I2.46i SOME FACTORS AFFECTING THE ACIDITY OF URINE IN MAN BY M. GRACE EGGLETON From the Department of Physiology, University College, London (Received 22 February 1947)
456 J. Physiol. (I947) io6, 456-465 6I2.46i SOME FACTORS AFFECTING THE ACIDITY OF URINE IN MAN BY M. GRACE EGGLETON From the Department of Physiology, University College, London (Received 22 February 1947)
IN a previous paper (Wood & BarHen, 1939) an experiment was described wherein
 5 STUDIES ON THE SULPHUR METABOLISM OF PLANTS II. THE EFFECT OF NITROGEN SUPPLY ON THE AMOUNTS OF PROTEIN SULPHUR, SULPHATE SULPHUR AND ON THE VALUE OF THE RATIO OF PROTEIN NITROGEN TO PROTEIN SULPHUR
5 STUDIES ON THE SULPHUR METABOLISM OF PLANTS II. THE EFFECT OF NITROGEN SUPPLY ON THE AMOUNTS OF PROTEIN SULPHUR, SULPHATE SULPHUR AND ON THE VALUE OF THE RATIO OF PROTEIN NITROGEN TO PROTEIN SULPHUR
THE CALORIGENIC ACTION OF EPINEPHRINE IN FROGS BEFORE AND AFTER HEPATECTOMY. (From the State Institute for the Study of Malignant Disease, Buflalo)
 THE CALORIGENIC ACTION OF EPINEPHRINE IN FROGS BEFORE AND AFTER HEPATECTOMY BY CARL F. CORI AND K. W. BUCHWALD (From the State Institute for the Study of Malignant Disease, Buflalo) (Received for publication,
THE CALORIGENIC ACTION OF EPINEPHRINE IN FROGS BEFORE AND AFTER HEPATECTOMY BY CARL F. CORI AND K. W. BUCHWALD (From the State Institute for the Study of Malignant Disease, Buflalo) (Received for publication,
simultaneously excreted. They also brought forward some evidence to
 THE EXCRETION OF CHLORIDES AND BICARBON- ATES BY THE HUMAN KIDNEY. BY H. W. DAVIES, M.B., B.S., J. B. S. HALDANE, M.A. AND G. L. PESKETT, B.A. (From the Laboratory, Cherwell, Oxford.) AM BARD and PAPI
THE EXCRETION OF CHLORIDES AND BICARBON- ATES BY THE HUMAN KIDNEY. BY H. W. DAVIES, M.B., B.S., J. B. S. HALDANE, M.A. AND G. L. PESKETT, B.A. (From the Laboratory, Cherwell, Oxford.) AM BARD and PAPI
PHYSICAL PROPERTIES AND DETECTION OF NORMAL CONSTITUENTS OF URINE
 PHYSICAL PROPERTIES AND DETECTION OF NORMAL CONSTITUENTS OF URINE - OBJECTIVES: 1- The simple examination of urine. 2- To detect some of the normal organic constituents of urine. 3- To detect some of the
PHYSICAL PROPERTIES AND DETECTION OF NORMAL CONSTITUENTS OF URINE - OBJECTIVES: 1- The simple examination of urine. 2- To detect some of the normal organic constituents of urine. 3- To detect some of the
THE EQUILIBRIUM BETWEEN ACTIVE NATIVE TRYPSIN AND INACTIVE DENATURED TRYPSIN
 Published Online: 20 January, 1934 Supp Info: http://doi.org/10.1085/jgp.17.3.393 Downloaded from jgp.rupress.org on November 8, 2018 THE EQUILIBRIUM BETWEEN ACTIVE NATIVE TRYPSIN AND INACTIVE DENATURED
Published Online: 20 January, 1934 Supp Info: http://doi.org/10.1085/jgp.17.3.393 Downloaded from jgp.rupress.org on November 8, 2018 THE EQUILIBRIUM BETWEEN ACTIVE NATIVE TRYPSIN AND INACTIVE DENATURED
Topic 1: Chemistry of Living Things
 1. Some processes that occur in a cell are listed below.1 utilize energy 2 detect changes in the environment 3 rearrange and synthesize chemical compounds 4. The diagram below represents a sequence of
1. Some processes that occur in a cell are listed below.1 utilize energy 2 detect changes in the environment 3 rearrange and synthesize chemical compounds 4. The diagram below represents a sequence of
THE EFFECT OF TITANIUM ON THE OXIDATION OF SULFHYDRYL GROUPS BY VARIOUS TISSUES
 THE EFFECT OF TITANIUM ON THE OXIDATION OF SULFHYDRYL GROUPS BY VARIOUS TISSUES BY FREDERICK BERNHEIM AND MARY L. C. BERNHEIM (From the Departments oj Physiology and Pharmacology and Biochemistry, Duke
THE EFFECT OF TITANIUM ON THE OXIDATION OF SULFHYDRYL GROUPS BY VARIOUS TISSUES BY FREDERICK BERNHEIM AND MARY L. C. BERNHEIM (From the Departments oj Physiology and Pharmacology and Biochemistry, Duke
dynamic action of ingested amino acids effected
 THE.EFFECT OF GLYCINE ON THE PRODUCTION AND EXCRETION OF URIC ACID1 BY MEYER FRIEDMAN (Fromn the Harold Brunn Institute for Cardiovascular Research, San Francisco, California) Mt. Zion Hospital, (Received
THE.EFFECT OF GLYCINE ON THE PRODUCTION AND EXCRETION OF URIC ACID1 BY MEYER FRIEDMAN (Fromn the Harold Brunn Institute for Cardiovascular Research, San Francisco, California) Mt. Zion Hospital, (Received
THE SOLUBILITY CURVE AND THE PURITY OF INSULIN
 THE SOLUBILITY CURVE AND THE PURITY OF INSULIN BY J. LENS (From the Organon Laboratories, Oss, Holland) (Received for publication, December 29, 1945) A method suitable for determining the degree of purity
THE SOLUBILITY CURVE AND THE PURITY OF INSULIN BY J. LENS (From the Organon Laboratories, Oss, Holland) (Received for publication, December 29, 1945) A method suitable for determining the degree of purity
A STUDY OF THE CONCENTRATION AND PROPERTIES OF TWO AMYLASES OF BARLEY MALT
 A STUDY OF THE CONCENTRATION AND PROPERTIES OF TWO AMYLASES OF BARLEY MALT BY M. L. CALDWELL AND S. E. DOEBBELING (From the Department of Chemistry, Columbia University, New York) (Received for publication,
A STUDY OF THE CONCENTRATION AND PROPERTIES OF TWO AMYLASES OF BARLEY MALT BY M. L. CALDWELL AND S. E. DOEBBELING (From the Department of Chemistry, Columbia University, New York) (Received for publication,
ON THE DETERMINATION OF UROBILIN IN URINE.
 ON THE DETERMINATION OF UROBILIN IN URINE. PRELIMINARY REPORT. RY S. MARCUSSEN AND SVEND HANSEN. (From the Rigshospitalet, University of Copenhagen, Copenhagen,.) (Received for publication, September 20,
ON THE DETERMINATION OF UROBILIN IN URINE. PRELIMINARY REPORT. RY S. MARCUSSEN AND SVEND HANSEN. (From the Rigshospitalet, University of Copenhagen, Copenhagen,.) (Received for publication, September 20,
Aim: To study the effect of ph on the action of salivary amylase. NCERT
 Exercise 28 Aim: To study the effect of ph on the action of salivary amylase. Principle: Optimal activity for most of the enzymes is generally observed between ph 5.0 and 9.0. However, a few enzymes, e.g.,
Exercise 28 Aim: To study the effect of ph on the action of salivary amylase. Principle: Optimal activity for most of the enzymes is generally observed between ph 5.0 and 9.0. However, a few enzymes, e.g.,
FURTHER STUDIES UPON THE PURIFICATION AND PROPERTIES OF MALT AMYLASE
 FURTHER STUDIES UPON THE PURIFICATION AND PROPERTIES OF MALT AMYLASE BY H. C. SHERMAN, M. L. CALDWELL, AND S. E. DOEBBELING (From the Department of Chemistry, Columbia University, New York) (Received for
FURTHER STUDIES UPON THE PURIFICATION AND PROPERTIES OF MALT AMYLASE BY H. C. SHERMAN, M. L. CALDWELL, AND S. E. DOEBBELING (From the Department of Chemistry, Columbia University, New York) (Received for
Chapter 10 EXCRETION
 Chapter 10 EXCRETION Control of Body Temperature and Water Balance Control of Body Temperature and Water Balance as a part of homeostasis Homeostasis means Maintenance of steady internal conditions despite
Chapter 10 EXCRETION Control of Body Temperature and Water Balance Control of Body Temperature and Water Balance as a part of homeostasis Homeostasis means Maintenance of steady internal conditions despite
THE DETERMINATION OF SUGAR IN BLOOD AND SPINAL FLUID WITH ANTHRONE REAGENT*
 THE DETERMINATION OF SUGAR IN BLOOD AND SPINAL FLUID WITH ANTHRONE REAGENT* BY JOSEPH H. ROE (From the Department of Biochemistry, School of Medicine, George Washington University, Washington, D. C.) (Received
THE DETERMINATION OF SUGAR IN BLOOD AND SPINAL FLUID WITH ANTHRONE REAGENT* BY JOSEPH H. ROE (From the Department of Biochemistry, School of Medicine, George Washington University, Washington, D. C.) (Received
COLORIMETRIC DETERMINATION OF URIC ACID.
 COLORIMETRIC DETERMINATION OF URIC ACID. ESTIMATION OF 0.03 TO 0.5 MG. QUANTITIES BY A NEW METHOD. BY J. LUCIEN MORRIS AND A. GARRARD MACLEOD. (From the Biochemistry Laboratory of the School of Medicine,
COLORIMETRIC DETERMINATION OF URIC ACID. ESTIMATION OF 0.03 TO 0.5 MG. QUANTITIES BY A NEW METHOD. BY J. LUCIEN MORRIS AND A. GARRARD MACLEOD. (From the Biochemistry Laboratory of the School of Medicine,
ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017
 ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017 WHAT INFORMATION DOES AN ABG GIVE US? ph = measure of hydrogen ion concentration (acidity or alkalinity) PaCO2 = partial pressure
ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017 WHAT INFORMATION DOES AN ABG GIVE US? ph = measure of hydrogen ion concentration (acidity or alkalinity) PaCO2 = partial pressure
Normal cooling mechanisms Heat-related illnesses. Evaluating the risk of heat illness Controlling heat stress First aid
 Normal cooling mechanisms Heat-related illnesses Evaluating the risk of heat illness Controlling heat stress First aid When body core temperature rises Blood flow to skin increases Sweating increases Heart
Normal cooling mechanisms Heat-related illnesses Evaluating the risk of heat illness Controlling heat stress First aid When body core temperature rises Blood flow to skin increases Sweating increases Heart
THE OHIO JOURNAL OF SCIENCE
 THE OHIO JOURNAL OF SCIENCE VOL. XLI SEPTEMBER, 1941 No. 5 THE HEART AND THE RED BLOOD CELLS AS GENERATOR AND DISTRIBUTORS OP STATIC ELECTRICITY PART I by GEORGE CRILE, M. D., PART II by OTTO GLASSER,
THE OHIO JOURNAL OF SCIENCE VOL. XLI SEPTEMBER, 1941 No. 5 THE HEART AND THE RED BLOOD CELLS AS GENERATOR AND DISTRIBUTORS OP STATIC ELECTRICITY PART I by GEORGE CRILE, M. D., PART II by OTTO GLASSER,
- process by which waste products are eliminated from an organism. 1. The maintenance of internal temperature within narrow limits is called
 Ch. 10: Excretion 1. Excretion means the - disposal of nitrogen-containing wastes - process by which waste products are eliminated from an organism 1. The disposal of nitrogen-containing wastes is called
Ch. 10: Excretion 1. Excretion means the - disposal of nitrogen-containing wastes - process by which waste products are eliminated from an organism 1. The disposal of nitrogen-containing wastes is called
Acid - base equilibrium
 Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid Base Balance. Chapter 26 Balance. ph Imbalances. Acid Base Balance. CO 2 and ph. Carbonic Acid. Part 2. Acid/Base Balance
 Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
THE METABOLISM OF SULFUR.
 THE METABOLISM OF SULFUR. XVI. DIETARY FACTORS IN RELATION TO THE CHEMICAL COMPOSITION OF THE HAIR OF THE YOUNG WHITE RAT. BY HOWARD D. LIGHTBODY AND HOWARD B. LEWIS. (From the Laboratory of Physiological
THE METABOLISM OF SULFUR. XVI. DIETARY FACTORS IN RELATION TO THE CHEMICAL COMPOSITION OF THE HAIR OF THE YOUNG WHITE RAT. BY HOWARD D. LIGHTBODY AND HOWARD B. LEWIS. (From the Laboratory of Physiological
CONTEXT POINT 2: Plants and animals transport dissolved nutrients and gases in a fluid medium.
 CONTEXT POINT 2: Plants and animals transport dissolved nutrients and gases in a fluid medium. Identify the form(s) in which each of the following is carried in mammalian blood: Carbon dioxide 70% as hydrogen
CONTEXT POINT 2: Plants and animals transport dissolved nutrients and gases in a fluid medium. Identify the form(s) in which each of the following is carried in mammalian blood: Carbon dioxide 70% as hydrogen
THE EFFECT OF HIGH TEMPERATURES ON THE NUTRITIVE VALUE OF FOODS.
 THE EFFECT OF HIGH TEMPERATURES ON THE NUTRITIVE VALUE OF FOODS. BY ALBERT G. HOGAN. (From the Department of Chemistry, Kansas Agricultural Experiment Station, Manhattan.) (Received for publication, March
THE EFFECT OF HIGH TEMPERATURES ON THE NUTRITIVE VALUE OF FOODS. BY ALBERT G. HOGAN. (From the Department of Chemistry, Kansas Agricultural Experiment Station, Manhattan.) (Received for publication, March
A. Correct! Flushing acids from the system will assist in re-establishing the acid-base equilibrium in the blood.
 OAT Biology - Problem Drill 16: The Urinary System Question No. 1 of 10 1. Which of the following would solve a drop in blood ph? Question #01 (A) Decreased retention of acids. (B) Increased excretion
OAT Biology - Problem Drill 16: The Urinary System Question No. 1 of 10 1. Which of the following would solve a drop in blood ph? Question #01 (A) Decreased retention of acids. (B) Increased excretion
3.4.6 The Excretory System in the Human
 3.4.6 The Excretory System in the Human Objectives What you will need to know from this section Explain the role of the excretory system in homeostasis -- the ability and necessity to maintain constancy
3.4.6 The Excretory System in the Human Objectives What you will need to know from this section Explain the role of the excretory system in homeostasis -- the ability and necessity to maintain constancy
CRYSTALLINE PEPSIN BY JOHN H. NORTHROP. (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton, iv. J.
 CRYSTALLINE PEPSIN III. PREPARATION OF ACTIVE CRYSTALLINE PEPSIN FROM INACTIVE DENATURED PEPSIN BY JOHN H. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton,
CRYSTALLINE PEPSIN III. PREPARATION OF ACTIVE CRYSTALLINE PEPSIN FROM INACTIVE DENATURED PEPSIN BY JOHN H. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton,
I. Decrease in Activity of Protein Nitrogen of Pepsin Solutions Exposed to Radium Bromide at ptt 5.0 and O C.
 CRYSTALLINE PEPSIN VI. INACTIVATION BY BETA AND GAMM~ RAYS FROM RADIUM AND BY ULTRA-VIOLET LIGHT BY JOHN:I-I. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Primeton,
CRYSTALLINE PEPSIN VI. INACTIVATION BY BETA AND GAMM~ RAYS FROM RADIUM AND BY ULTRA-VIOLET LIGHT BY JOHN:I-I. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Primeton,
ION ANTAGONISMS AFFECTING GLYCOLYSIS BY BACTERIAL SUSPENSIONS*
 ION ANTAGONISMS AFFECTING GLYCOLYSIS BY BACTERIAL SUSPENSIONS* BY HIROSHI TSUYUKIt AND ROBERT A. MAcLEOD (From the Department of Biochemistry, Queen s University, Kingston, Ontario, Canada) (Received for
ION ANTAGONISMS AFFECTING GLYCOLYSIS BY BACTERIAL SUSPENSIONS* BY HIROSHI TSUYUKIt AND ROBERT A. MAcLEOD (From the Department of Biochemistry, Queen s University, Kingston, Ontario, Canada) (Received for
excreted, in spite of its constant presence in the blood. Similarly, a salt-free diet will rapidly cause the practical disappearance of chlorides
 THE REGULATION OF EXCRETION OF WATER BY THE KIDNEYS. I. By J. S. HALDANE, M.D., F.R.S. AND J. G. PRIESTLEY, B.M., Captain R.A.M.C., Beit Memorial Research Fellow. NUMEROUS observations tend to show that
THE REGULATION OF EXCRETION OF WATER BY THE KIDNEYS. I. By J. S. HALDANE, M.D., F.R.S. AND J. G. PRIESTLEY, B.M., Captain R.A.M.C., Beit Memorial Research Fellow. NUMEROUS observations tend to show that
EFFECTS OF ANTICOAGULANTS ON THE ph. (Studies on the blood ph estimated by the glass electrode method. II)
 The Journal of Biochemistry, vol. 22, No. 2. EFFECTS OF ANTICOAGULANTS ON THE ph OF THE BLOOD. (Studies on the blood ph estimated by the glass electrode method. II) BY HISATO YOSHIMURA (From the First
The Journal of Biochemistry, vol. 22, No. 2. EFFECTS OF ANTICOAGULANTS ON THE ph OF THE BLOOD. (Studies on the blood ph estimated by the glass electrode method. II) BY HISATO YOSHIMURA (From the First
hold for the human kidney.2 Shannon and Smith (4) have rightfully stressed
 THE RENAL EXCRETION OF INULIN AT LOW PLASMA CONCEN- TRATIONS OF THIS COMPOUND, AND ITS RELATIONSHIP TO THE GLOMERULAR FILTRATION RATE IN NORMAL, NEPHRITIC AND HYPERTENSIVE INDIVIDUALS' By BENJAMIN F. MILLER,
THE RENAL EXCRETION OF INULIN AT LOW PLASMA CONCEN- TRATIONS OF THIS COMPOUND, AND ITS RELATIONSHIP TO THE GLOMERULAR FILTRATION RATE IN NORMAL, NEPHRITIC AND HYPERTENSIVE INDIVIDUALS' By BENJAMIN F. MILLER,
CASE 27. What is the response of the kidney to metabolic acidosis? What is the response of the kidney to a respiratory alkalosis?
 CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
Human Creatinine Urinary Detection Kit
 Human Creatinine Urinary CATALOG NO: IRAAKT2509 Detection Kit LOT NO: SAMPLE INTENDED USE The Urinary Creatinine kit is designed to quantitatively measure creatinine present in urine samples. BACKGROUND
Human Creatinine Urinary CATALOG NO: IRAAKT2509 Detection Kit LOT NO: SAMPLE INTENDED USE The Urinary Creatinine kit is designed to quantitatively measure creatinine present in urine samples. BACKGROUND
Acid/Base Balance. the concentrations of these two ions affect the acidity or alkalinity of body fluids
 Acid/Base Balance some of most critical ions in body fluids are H + (hydrogen) and OH - (hydroxyl) ions the concentrations of these two ions affect the acidity or alkalinity of body fluids acidity/alkalinity
Acid/Base Balance some of most critical ions in body fluids are H + (hydrogen) and OH - (hydroxyl) ions the concentrations of these two ions affect the acidity or alkalinity of body fluids acidity/alkalinity
Other dogs were given -ether anesthesia and the variations in heart
 THE EFFECT OF REGULAR AND IRREGULAR TACHY- CARDIAS ON THE SIZE OF THE HEART By. HAROLD J. STEWART AND J. HAMILTON CRAWFORD (From the Hospital of the Rockefeller Institute for Medical Research, New York,
THE EFFECT OF REGULAR AND IRREGULAR TACHY- CARDIAS ON THE SIZE OF THE HEART By. HAROLD J. STEWART AND J. HAMILTON CRAWFORD (From the Hospital of the Rockefeller Institute for Medical Research, New York,
Conditions inside the body must be kept constant. Urea must be removed from the body. Name the organ which makes urea.
 Conditions inside the body must be kept constant. (a) Urea must be removed from the body. (i) Name the organ which makes urea. () Which organ removes urea from the body? () (iii) What is urea made from?
Conditions inside the body must be kept constant. (a) Urea must be removed from the body. (i) Name the organ which makes urea. () Which organ removes urea from the body? () (iii) What is urea made from?
The Urinary S. (Chp. 10) & Excretion. What are the functions of the urinary system? Maintenance of water-salt and acidbase
 10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
Biology Notes. Homeostasis
 Biology Notes Homeostasis Homeostasis is defined as the maintenance of constant internal conditions within organisms. The principle can be developed in a wide range of contexts, including the maintenance
Biology Notes Homeostasis Homeostasis is defined as the maintenance of constant internal conditions within organisms. The principle can be developed in a wide range of contexts, including the maintenance
THE ESTIMATION OF TRYPSIN WITH HEMOGLOBIN
 THE ESTIMATION OF TRYPSIN WITH HEMOGLOBIN BY M. L. ANSON Am) A. E. MIRSKY (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton, N. J., and the Hospital of The Rockefeller
THE ESTIMATION OF TRYPSIN WITH HEMOGLOBIN BY M. L. ANSON Am) A. E. MIRSKY (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton, N. J., and the Hospital of The Rockefeller
THE INHIBITION OF URICASE BY XANTHINE
 THE INHIBITION OF URICASE BY XANTHINE BY JOHN F. VAN PILSUM [From the Deparfment of Biological Chemistry, University of Utah College of Medicine, Salt Lake City, Utah, and the Department of Biochemistry,
THE INHIBITION OF URICASE BY XANTHINE BY JOHN F. VAN PILSUM [From the Deparfment of Biological Chemistry, University of Utah College of Medicine, Salt Lake City, Utah, and the Department of Biochemistry,
Hydrolysis of Irradiated Ovalbumin by Pepsin
 Hydrolysis of Irradiated Ovalbumin by Pepsin HECTOR A. DIEU and V. DESREUX From the Department of Physical Chemistry, University of Liege, Liege, Belgium ABSTRACT Solid ovalbumin has been irradiated at
Hydrolysis of Irradiated Ovalbumin by Pepsin HECTOR A. DIEU and V. DESREUX From the Department of Physical Chemistry, University of Liege, Liege, Belgium ABSTRACT Solid ovalbumin has been irradiated at
THE ENZYMATIC HYDROLYSIS OF GLUTATHIONE BY RAT KIDNEY
 THE ENZYMATIC HYDROLYSIS OF GLUTATHIONE BY RAT KIDNEY BY E. F. SCHROEDER AND GLADYS E. WOODWARD (From The Biochemical Research Foundation of the Franklin Institute, Philadelphia) (Received for publication,
THE ENZYMATIC HYDROLYSIS OF GLUTATHIONE BY RAT KIDNEY BY E. F. SCHROEDER AND GLADYS E. WOODWARD (From The Biochemical Research Foundation of the Franklin Institute, Philadelphia) (Received for publication,
RICINOLEATE UPON BACTERIA
 A COMPARATIVE STUDY OF THE ACTION OF SODIUM RICINOLEATE UPON BACTERIA From the Division of Laboratories and Research, New York State Department of Health, Albany Received for publication, May 14, 1928
A COMPARATIVE STUDY OF THE ACTION OF SODIUM RICINOLEATE UPON BACTERIA From the Division of Laboratories and Research, New York State Department of Health, Albany Received for publication, May 14, 1928
THE INHIBITION OF CHOLINESTERASE BY PHYSOSTIGMINE AND PROSTIGMINE
 THE INHIBITION OF CHOLINESTERASE BY PHYSOSTIGMINE AND PROSTIGMINE BY G. S. EADIE (From the Department of Physiology and Pharmacology, Duke University School of Medicine, Durham, North Carolina) (Received
THE INHIBITION OF CHOLINESTERASE BY PHYSOSTIGMINE AND PROSTIGMINE BY G. S. EADIE (From the Department of Physiology and Pharmacology, Duke University School of Medicine, Durham, North Carolina) (Received
ITEC Unit 13 Facial Electrical Treatments Recommended guided learning hours 250
 ITEC Unit 13 Facial Electrical Treatments Recommended guided learning hours 250 Pre-requisite Students must hold the ITEC Level 2 Diploma for Beauty Specialists or equivalent Unit 13 Facial Electrical
ITEC Unit 13 Facial Electrical Treatments Recommended guided learning hours 250 Pre-requisite Students must hold the ITEC Level 2 Diploma for Beauty Specialists or equivalent Unit 13 Facial Electrical
12/7/10. Excretory System. The basic function of the excretory system is to regulate the volume and composition of body fluids by:
 Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
CXII. THE EXCRETION OF CALCIUM BY THE LARGE INTESTINE OF THE RABBIT
 CXII. THE EXCRETION OF CALCIUM BY THE LARGE INTESTINE OF THE RABBIT BY STUART JASPER COWELL From St Thomas's Hospital Medical School and the Medical Unit, University College Hospital, London (Received
CXII. THE EXCRETION OF CALCIUM BY THE LARGE INTESTINE OF THE RABBIT BY STUART JASPER COWELL From St Thomas's Hospital Medical School and the Medical Unit, University College Hospital, London (Received
Prepared using Wordle.net for fiendishlyclever.com by R Butler
 Prepared using Wordle.net for fiendishlyclever.com by R Butler Scientific enquiry Level 1 Level 2 Level 3 Pupils describe or respond appropriately to simple features of objects, living things and events
Prepared using Wordle.net for fiendishlyclever.com by R Butler Scientific enquiry Level 1 Level 2 Level 3 Pupils describe or respond appropriately to simple features of objects, living things and events
Excretory System 1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Urea Nitrogen (BUN) detection Kit
 K-ASSAY KAMIYA BIOMEDICAL COMPANY KAMIYA BIOMEDICAL COMPANY Urea Nitrogen (BUN) detection Kit For the quantitative determination of urea nitrogen in saliva and TCM Cat. No. KT-747 For Research Use Only.
K-ASSAY KAMIYA BIOMEDICAL COMPANY KAMIYA BIOMEDICAL COMPANY Urea Nitrogen (BUN) detection Kit For the quantitative determination of urea nitrogen in saliva and TCM Cat. No. KT-747 For Research Use Only.
CAROTENASE. THE TRANSFORMATION OF CAROTENE TO VITAMIN A IN VITRO *
 CAROTENASE. THE TRANSFORMATION OF CAROTENE TO VITAMIN A IN VITRO * BY H. S. OLCOTT Ai id D. C. MCCANN (From the Laboratories of Biochemistry and Analytical Chemistry, State University of Iowa, Iowa City)
CAROTENASE. THE TRANSFORMATION OF CAROTENE TO VITAMIN A IN VITRO * BY H. S. OLCOTT Ai id D. C. MCCANN (From the Laboratories of Biochemistry and Analytical Chemistry, State University of Iowa, Iowa City)
