Susceptible Populations Workshop January 20-21, 2010 Greenbelt, MD
|
|
|
- Todd Gilmore
- 6 years ago
- Views:
Transcription
1 Susceptible Populations Workshop January 20-21, 2010 Greenbelt, MD CHEMICAL HAZARDS SUSCEPTIBLE POPULATIONS DISCUSSION Participants: Frederick Angulo, Kerry Dearfield, Michael DiNovi, Diana Doell, Michael Firestone, Daniel Folmer, Brenda Foos, Gary Ginsberg, Sara Henry, Susan Laessig, Susan Makris, Jacqueline Moya, James Pestka, Kathleen Raffaele, Helen Rogers, Ryan Simon, Deborah Smegal, Kathy Sykes, Steve Taylor, Michelle Twaroski, Jane Van Doren DEFINITION: A Susceptible Population is a group of individuals that have an identifiable common characteristic for which the rate/magnitude of response or type of health outcome differs (or could reasonably be expected to differ) from that of a reference population. This definition includes: (1) Qualitative or quantitative differences in response arising from a given exposure. (2) It may or may not include differences arising from different exposures. (All agreed differing exposures must be included in a risk assessment but did not agree on whether groups with high exposure should be classified as susceptible populations or not.) What questions should a risk assessor ask to identify relevant susceptible populations for a given chemical hazard? Does the chemical have a known harmful effect? Is there a known Mode of Action (MOA) that implicates particular sensitive populations? Have there been reports of illness in exposed populations/individuals? Is there an activation/deactivation step that may involve a susceptible population? Are there likely to be interactions with other common exposures (e.g., Tylenol/alcohol)? Are there known groups with expected high exposure or unique exposure patterns? What are the chemical s characteristics? (Will it bioaccumulate? Persist? Sequester in animal tissue or the environment? Lipophilicity?) Are there indicators in available hazard data pointing to possible susceptible populations/life-stages? Is the MOA unique or will it interact with background disease process (and perhaps augment them)? How will the chemical be used? What data are needed to define/characterize a susceptible population? Susceptibility? Need to start from context of risk assessment Context = new product See list of questions for Risk Assessor above Characteristics of the potentially exposed population o Demographics o Lifestyle
2 o Culture o Genetics o Metabolism differences o Dietary habits o Nutritional status o Health status or Disease characteristics e.g., diabetes/sugar substitutes, allergies/proteins, liver disease/vibrio, immune compromised/listeria, pregnancy (developmental, maternal), drug interactions o Drug exposure o Other co-exposures o Life-stage o Geography o Occupation o Socioeconomic status Context = known susceptible population Characterize the population see list above o Health status o Diseases status o Diet o Geologic location o demographics What are the challenges to doing a risk assessment (RA) that incorporates susceptible populations? Lack of resources Uncertainty as to how the data will be used Level of effort Data Animal models don t address question of concern Human subjects protection for rare/unusual conditions Exposure data o Biomonitoring o Ultimate uses o Data for population subgroups, especially early ages and higher age group o Can t monitor for new substances or unknown metabolites o Behavior/lifestyle information Lack of up-to-date information (e.g., consumption) Epidemiological studies o Longitudinal o Specific targeted exposure data vs. identifying associations o Resource: hospital discharge data Much data is not of sufficient quality to inform risk assessments MOA information that can feed into analyses o Toxicity pathways o How they change with life-stages
3 Ways to use toxicological/genomic data to inform susceptibility How representative are genomic/screening studies of susceptible populations? Can gene arrays or other screening techniques be developed in such a way that they can be fed into risk assessments and or identification or characterization of susceptible populations? How do we define the baseline for comparison? Epidemiological studies potential for confounding co-exposures Burden of proof required re: effects/associations Basic lack of knowledge o Windows of vulnerability o Critical toxicity pathways o Paradoxical dose-response curves o Defining adversity Connection between molecular or genotoxic changes to effects of concern Clear connection between assay results and disease outcomes Delayed sequelae o How do we identify? o Delayed effects of early life exposure What do we need to overcome the challenges? Exposure models vs. toxicokinetic models vs. toxicodynamic models Dose models Increased accessibility and better standardization of models o More transparency of model parameters and assumptions o More consistent terminology Model Parameterization o Exposure OK o Kinetics Fair amount of data o Polymorphisms some data o Dynamics- Black box Toxicology testing methods that better address susceptible populations o Life-stages o Genetics o Disease states o Can we make better use of available disease models in understanding disease/chemical interactions? Knockout mice (for example) Humanized mice Better data regarding how consumer products are used. o What chemicals were used in what products Better matching of cross-sectional survey data with long-term outcomes How do we improve considerations of susceptibility in RA? Check box method unlikely to work
4 Incorporate all available information Identify tests/pathways most relevant to issue of concern Protect known sensitive groups Descriptive data versus numerical data Risk Management options for susceptible populations o Warn group via labels? Resource Center for Disease Control & Prevention (CDC): National Environmental Public Health Tracking Program Top Risk Assessment Issues/Improvements Value of animal models in identifying susceptible factors Increased collection of biomonitoring data for early life stages Identifying interactions between chemicals and other factors as they impact susceptibility (including non-chemical stressors) What toxicity pathways are most predictive/correlated with susceptibility? What are pharmacokinetic and pharmacodynamic differences between susceptible population and reference population? Identifying surveillance systems we can use to track disease in populations How are susceptible populations addressed in each of our agencies? Food & Drug Administration (FDA): case-by-case basis, often include assessments for identifiable populations associated with use of that food (e.g., children for BPA, adults taking Coumadin for Olestra, diabetics for artificial sweeteners) Start with exposure assessment in pre-market assessments o Office of Food Additive Safety (OFAS) reviews new food additive petitions for direct and indirect food additives pre-market. (Indirect additives include industrial compounds that are not supposed to be in foods, 85% of these will result in <50 ppb exposure). OFAS has 120 days respond to petition. Exposure to the additive is calculated based on the proposed use of the additive and the intended use level. Exposure is calculated for the general population as well as any other population that should be considered (e.g., children) Don t always know ultimate use of food additive, so simply use-type and foods involved. No standard way to address. CDC: gather information, ask about identifiable populations and look for susceptible populations from these data, ID populations, and develop target educational programs. o Effort for chemicals and toxins, priority to be proactive is increasing o Info from outbreaks then ask about characteristics of the people who involved o Routine surveys through National Center for Health Statistics (NCHS) o National surveillance for lead o Blood & urine samples from National Health & Nutrition Examination Survey (NHANES) 200 chemicals, trying to measure health outcomes, food exposures, trying to make correlations with diet, susceptible populations
5 United States Department of Agriculture (USDA): lot of work on the microbial side. Chemical work building back up. Right now focus is on testing for residues rather than risk assessment because we do not set regulatory limits. o Lot of work on microbial side, Food Safety & Inspection Service (FSIS) has big testing program, Agricultural Research Service (ARS) has huge sampling and research question. ARS is moving to genomics. o USDA National residue program that addresses chemicals: annual sampling of meats for vet drugs, pesticide residues. Have not tied that to outbreaks yet. USDA testing for adulterated foods based on regulations from FDA and others. o Problem is finding residues for which there are no levels set (by FDA/Center for Food Safety & Applied Nutrition). USDA is exploring whether they can establish levels for which there is not regulatory level (e.g., Cd, dioxin). Have trigger levels and alert FDA to ID sources of chemicals to address through compliance. Susceptible populations pop up but just starting with chemicals. Farm bill, catfish now on USDA, and environmental chemicals will be most important rather than microbial contamination. May be able to set levels in catfish now that it is their responsibility. States (Connecticut and regional work): Body burden chemicals looking at susceptible populations, e.g., females of child-bearing age o When looking at fish looking at earlier life-stages as well as pregnancy (for PCBs). o Thought about trans-generational susceptibility o It also starts with question: who is exposed? Looking at identifiable populations (children, pregnant women). o Generally, we don t have access to data. o States working to come up with watch lists for chemicals (looking at Toxic Substances Control Act (TSCA)). Additional Questions Raised How do you communicate susceptibility? What do you do with information on susceptibility once you have it? Can we make a common framework for addressing susceptible populations in risk assessments? Where do we go from here? (1) Continue the conversation (through additional meetings organized by IRAC membership.) Can we develop a common framework? (2) Create databases for the following (& possible post on FoodRisk.org) case studies that have addressed risk assessment for susceptible populations characterization of susceptible populations o disease-food connections o risk assessments that have included consideration of susceptible populations (Good starting point: Rebecca s talk, identified 9 such risk assessments) o ILSI (International Life Sciences Institute) has workgroup on thresholds, 4 working groups,--steve Baldwin will have copy Steve Taylor
6 Groups that have very high intakes, e.g., Vit. A
Introduction. Dietary Exposure Assessment Tools for Prioritizing Food Safety Concerns. Workshop on. Stephen S. Olin
 Introduction Workshop on Dietary Exposure Assessment Tools for Prioritizing Food Safety Concerns Stephen S. Olin International Life Sciences Institute Research Foundation Issues Ensuring a safe and wholesome
Introduction Workshop on Dietary Exposure Assessment Tools for Prioritizing Food Safety Concerns Stephen S. Olin International Life Sciences Institute Research Foundation Issues Ensuring a safe and wholesome
Building Capacity for the Identification of Emerging Food Safety Risks
 Building Capacity for the Identification of Emerging Food Safety Risks David P. Goldman, MD, MPH Assistant Administrator Office of Public Health Science APEC FSCF PTIN Food Safety Incident Management Workshop
Building Capacity for the Identification of Emerging Food Safety Risks David P. Goldman, MD, MPH Assistant Administrator Office of Public Health Science APEC FSCF PTIN Food Safety Incident Management Workshop
Assessing and Managing Health Risks from Chemical Constituents and Contaminants of Food
 16 17 September 2013 Assessing and Managing Health Risks from Chemical Constituents and Contaminants of Food Workshop on A Framework for Assessing the Health, Environmental and Social Effects of the Food
16 17 September 2013 Assessing and Managing Health Risks from Chemical Constituents and Contaminants of Food Workshop on A Framework for Assessing the Health, Environmental and Social Effects of the Food
Chemical & Microbial Risk Assessment: Case studies to demonstrate similarities and differences
 Chemical & Microbial Risk Assessment: Case studies to demonstrate similarities and differences Jane M. Van Doren Branch Chief, Risk Analysis Branch Deborah Smegal Branch Chief, Contaminants Assessment
Chemical & Microbial Risk Assessment: Case studies to demonstrate similarities and differences Jane M. Van Doren Branch Chief, Risk Analysis Branch Deborah Smegal Branch Chief, Contaminants Assessment
- draft scientific opinion -
 The Re-evaluation of faspartame - draft scientific opinion - Dr. Alicja Mortensen Chair of EFSA s ANS Panel Follow-up meeting on the web-based public consultation on aspartame 9 April 2013, Bruxelles Draft
The Re-evaluation of faspartame - draft scientific opinion - Dr. Alicja Mortensen Chair of EFSA s ANS Panel Follow-up meeting on the web-based public consultation on aspartame 9 April 2013, Bruxelles Draft
Case Study Application of the WHO Framework for Combined Exposures. Presented by: M.E. (Bette) Meek University of Ottawa
 Case Study Application of the WHO Framework for Combined Exposures Presented by: M.E. (Bette) Meek University of Ottawa bmeek@uottawa.ca WHO IPCS Framework for Combined Exposures Objectives Building on
Case Study Application of the WHO Framework for Combined Exposures Presented by: M.E. (Bette) Meek University of Ottawa bmeek@uottawa.ca WHO IPCS Framework for Combined Exposures Objectives Building on
Overview of Regulatory Science of Food Contact Substances
 Overview of Regulatory Science of Food Contact Substances Michael A. Adams, PhD Deputy Director, Office of Food Additive Safety FDA, Center for Food Safety and Applied Nutrition 5001 Campus Drive, College
Overview of Regulatory Science of Food Contact Substances Michael A. Adams, PhD Deputy Director, Office of Food Additive Safety FDA, Center for Food Safety and Applied Nutrition 5001 Campus Drive, College
Food Safety and Inspection Service:
 1 Identification and Prioritization of Chemical Hazards in Animal Products Monitored by the U. S. National Residue Program Randolph Duverna, PhD. Chemist, Science Staff Office of Public Health Science
1 Identification and Prioritization of Chemical Hazards in Animal Products Monitored by the U. S. National Residue Program Randolph Duverna, PhD. Chemist, Science Staff Office of Public Health Science
Meeting Report: Seminar on Uses and Safety of Sweeteners, May 30, 2013, Jakarta, Indonesia
 Meeting Report: Seminar on Uses and Safety of Sweeteners, May 30, 2013, Jakarta, Indonesia On May 30, 2013, ILSI Southeast Asia Region, together with the ILSI Indonesia Country Committee and Southeast
Meeting Report: Seminar on Uses and Safety of Sweeteners, May 30, 2013, Jakarta, Indonesia On May 30, 2013, ILSI Southeast Asia Region, together with the ILSI Indonesia Country Committee and Southeast
Food Consumption Data in Microbiological Risk Assessment
 1972 Journal of Food Protection, Vol. 67, No. 9, 2004, Pages 1972 1976 Copyright, International Association for Food Protection Food Consumption Data in Microbiological Risk Assessment LEILA M. BARRAJ*
1972 Journal of Food Protection, Vol. 67, No. 9, 2004, Pages 1972 1976 Copyright, International Association for Food Protection Food Consumption Data in Microbiological Risk Assessment LEILA M. BARRAJ*
ADVANCES ON RISK ASSESSMENT FOR ALLERGENS IN FOOD: AN OVERVIEW AND SUPPORTING TOOLS
 ADVANCES ON RISK ASSESSMENT FOR ALLERGENS IN FOOD: AN OVERVIEW AND SUPPORTING TOOLS Steve L. Taylor, Ph.D. Food Allergy Research & Resource Program University of Nebraska VIII Updates on Food Safety Symposium
ADVANCES ON RISK ASSESSMENT FOR ALLERGENS IN FOOD: AN OVERVIEW AND SUPPORTING TOOLS Steve L. Taylor, Ph.D. Food Allergy Research & Resource Program University of Nebraska VIII Updates on Food Safety Symposium
FDA s Food Additives Program
 FDA s Food Additives Program LaShonda T. Cureton, PhD Office of Food Additive Safety US Food and Drug Administration Food Additives: A Global Perspective on Safety Evaluation and Use Procedures for Approval
FDA s Food Additives Program LaShonda T. Cureton, PhD Office of Food Additive Safety US Food and Drug Administration Food Additives: A Global Perspective on Safety Evaluation and Use Procedures for Approval
Inter-Agency Overlap and Jurisdictional Boundaries
 Inter-Agency Overlap and Jurisdictional Boundaries FDLI Food Advertising, Labeling, and Litigation Conference September 14, 2017 Jessica P. O Connell Covington & Burling LLP jpoconnell@cov.com 1 FDA Approach/Perspective
Inter-Agency Overlap and Jurisdictional Boundaries FDLI Food Advertising, Labeling, and Litigation Conference September 14, 2017 Jessica P. O Connell Covington & Burling LLP jpoconnell@cov.com 1 FDA Approach/Perspective
Food Safety and Inspection Service Research Priorities
 Food Safety and Inspection Service Research Priorities David Goldman, MD, MPH Assistant Administrator Science Food Safety and Inspection Service 1 Technology Workshop on Food Safety and National Defense
Food Safety and Inspection Service Research Priorities David Goldman, MD, MPH Assistant Administrator Science Food Safety and Inspection Service 1 Technology Workshop on Food Safety and National Defense
Chemical food safety in the U.S. analysis of FDA s scientific basis for assessing chemical risk. Tom Neltner October 9, 2014
 Chemical food safety in the U.S. analysis of FDA s scientific basis for assessing chemical risk Tom Neltner October 9, 2014 Topics 1. Current focus of U.S. public interest community 2. Comparison of U.S.
Chemical food safety in the U.S. analysis of FDA s scientific basis for assessing chemical risk Tom Neltner October 9, 2014 Topics 1. Current focus of U.S. public interest community 2. Comparison of U.S.
Listeria monocytogenes Risk Assessment: Executive Summary
 Listeria monocytogenes Assessment: Executive Summary FDA/Center for Food Safety and Applied Nutrition USDA/Food Safety and Inspection Service September 2003 Background The U.S. Department of Health and
Listeria monocytogenes Assessment: Executive Summary FDA/Center for Food Safety and Applied Nutrition USDA/Food Safety and Inspection Service September 2003 Background The U.S. Department of Health and
Safety Evaluation for Substances Directly Added to Food
 Safety Evaluation for Substances Directly Added to Food Teresa Croce, PhD Office of Food Additive Safety Center for Food Safety and Applied Nutrition US Food and Drug Administration How Does FDA Regulate
Safety Evaluation for Substances Directly Added to Food Teresa Croce, PhD Office of Food Additive Safety Center for Food Safety and Applied Nutrition US Food and Drug Administration How Does FDA Regulate
Food Additives Program
 U.S. FDA s Food Additive Program: An Update on Resources and Challenges 18 th Food Packaging Law Seminar October 11, 2017 Arlington, VA 1 Food Additives Program Dennis Keefe, PhD Director, Office of Food
U.S. FDA s Food Additive Program: An Update on Resources and Challenges 18 th Food Packaging Law Seminar October 11, 2017 Arlington, VA 1 Food Additives Program Dennis Keefe, PhD Director, Office of Food
Science Policy Notice
 Science Policy Notice SPN2002-01 Children s Health Priorities within the Pest Management Regulatory Agency (publié aussi en français) January 3, 2002 This document is published by the Submission Coordination
Science Policy Notice SPN2002-01 Children s Health Priorities within the Pest Management Regulatory Agency (publié aussi en français) January 3, 2002 This document is published by the Submission Coordination
Risk Characterization
 Risk Characterization 1 Learning Objectives By the end of this module, participants should have an understanding of: The purpose of risk characterization Integrating the results of hazard identification,
Risk Characterization 1 Learning Objectives By the end of this module, participants should have an understanding of: The purpose of risk characterization Integrating the results of hazard identification,
Simplified Modeling Framework for Microbial Food-Safety Risk Assessments
 Food Safety and Inspection Service Simplified Modeling Framework for Microbial Food-Safety Risk Assessments Michael Williams Risk Assessment and Analytics Staff Food Safety and Inspection Service, USDA
Food Safety and Inspection Service Simplified Modeling Framework for Microbial Food-Safety Risk Assessments Michael Williams Risk Assessment and Analytics Staff Food Safety and Inspection Service, USDA
FDA Regulation of Claims on Dietary Supplement and Food Products
 FDA Regulation of Claims on Dietary Supplement and Food Products Patricia Kaeding August 2004 Topics FDA Overview What are claims Why claims matter Categories of claims Regulatory requirements for types
FDA Regulation of Claims on Dietary Supplement and Food Products Patricia Kaeding August 2004 Topics FDA Overview What are claims Why claims matter Categories of claims Regulatory requirements for types
11/29/2010 FOOD SAFETY CAPACITY BUILDING ON RESIDUE CONTROL
 Risk Analysis and MRLs OIE Training Workshop Pretoria 23 26 th November 2010 Antoine Kabwit Nguz Content Food Safety Risk Analysis Process of setting MRLs Good practices and MRLs Conclusion 1 Food Safety
Risk Analysis and MRLs OIE Training Workshop Pretoria 23 26 th November 2010 Antoine Kabwit Nguz Content Food Safety Risk Analysis Process of setting MRLs Good practices and MRLs Conclusion 1 Food Safety
STUDIES TO EVALUATE THE SAFETY OF RESIDUES OF VETERINARY DRUGS IN HUMAN FOOD: GENERAL APPROACH TO ESTABLISH AN ACUTE REFERENCE DOSE
 VICH GL54 (SAFETY) ARfD November 2016 For Implementation at Step 7 STUDIES TO EVALUATE THE SAFETY OF RESIDUES OF VETERINARY DRUGS IN HUMAN FOOD: GENERAL APPROACH TO ESTABLISH AN ACUTE REFERENCE DOSE (ARfD)
VICH GL54 (SAFETY) ARfD November 2016 For Implementation at Step 7 STUDIES TO EVALUATE THE SAFETY OF RESIDUES OF VETERINARY DRUGS IN HUMAN FOOD: GENERAL APPROACH TO ESTABLISH AN ACUTE REFERENCE DOSE (ARfD)
Quantitative Risk Assessment: An Overview and Discussion of Emerging Issues. Anne-Marie Nicol, PhD
 Quantitative Risk Assessment: An Overview and Discussion of Emerging Issues Anne-Marie Nicol, PhD Today s talk Broader overview of Risk Assessment process (at the US EPA) Explanation of the 4 step formal
Quantitative Risk Assessment: An Overview and Discussion of Emerging Issues Anne-Marie Nicol, PhD Today s talk Broader overview of Risk Assessment process (at the US EPA) Explanation of the 4 step formal
From Farm to Fork: Practical Applications for Food Composition Data. Catherine E. Woteki, Ph.D., R.D. Dean of Agriculture Iowa State University
 From Farm to Fork: Practical Applications for Food Composition Data Catherine E. Woteki, Ph.D., R.D. Dean of Agriculture Iowa State University Nutrition Monitoring and Surveillance Systems in the United
From Farm to Fork: Practical Applications for Food Composition Data Catherine E. Woteki, Ph.D., R.D. Dean of Agriculture Iowa State University Nutrition Monitoring and Surveillance Systems in the United
EU policy on acrylamide in food reducing human exposure to ensure a high level of human health protection
 Directorate-General for Health & Food Safety EU policy on acrylamide in food reducing human exposure to ensure a high level of human health protection Frans Verstraete Principles for regulating contaminants
Directorate-General for Health & Food Safety EU policy on acrylamide in food reducing human exposure to ensure a high level of human health protection Frans Verstraete Principles for regulating contaminants
Module 34: Legal aspects, ADI and GRAS status of food additives
 Paper No.: 13 Paper Title: FOOD ADDITIVES Module 34: Legal aspects, ADI and GRAS status of food additives 34.1 Legal Aspects of Food Additives The data provided by Joint Expert Committee on Food Additives
Paper No.: 13 Paper Title: FOOD ADDITIVES Module 34: Legal aspects, ADI and GRAS status of food additives 34.1 Legal Aspects of Food Additives The data provided by Joint Expert Committee on Food Additives
D. Health and Environmental Sciences D1. Health Sciences. (1) Public Health. (2) Disease Prevention
 D. Health and Environmental Sciences D1. Health Sciences GIO: To acquire the basic knowledge, skills, and behavior about prevention of diseases and nutrition in contemporary society and to contribute to
D. Health and Environmental Sciences D1. Health Sciences GIO: To acquire the basic knowledge, skills, and behavior about prevention of diseases and nutrition in contemporary society and to contribute to
An Introduction CORE Network
 An Introduction CORE Network FDA s New Approach to Managing Foodborne Outbreak Surveillance, Response and Post-Response Efforts Presented by Carla Tuite, LCDR, USPHS Response Team Leader CORE August 30,
An Introduction CORE Network FDA s New Approach to Managing Foodborne Outbreak Surveillance, Response and Post-Response Efforts Presented by Carla Tuite, LCDR, USPHS Response Team Leader CORE August 30,
Reproductive Health and the Environment: The Clinicians Role in Protecting Early Development
 Reproductive Health and the Environment: The Clinicians Role in Protecting Early Development Jeanne A. Conry, MD, PhD Immediate Past President The American Congress of Obstetricians and Gynecologists 2014-2015
Reproductive Health and the Environment: The Clinicians Role in Protecting Early Development Jeanne A. Conry, MD, PhD Immediate Past President The American Congress of Obstetricians and Gynecologists 2014-2015
The regulatory landscape. The now and the not yet
 The regulatory landscape The now and the not yet Perspectives Aims Promote common understanding Anticipate the coming changes Prepare for afternoon sessions Who governs pesticides? All EU legislation comes
The regulatory landscape The now and the not yet Perspectives Aims Promote common understanding Anticipate the coming changes Prepare for afternoon sessions Who governs pesticides? All EU legislation comes
Dietary exposure assessment in the Slovak Republic
 Šalgovičová Danka, Pavlovičová Daniela Dietary exposure assessment in the Slovak Republic Food Research Institute - Bratislava Safefoodnet - Chemical Food Safety Network for the enlarging Europe, Bratislava,
Šalgovičová Danka, Pavlovičová Daniela Dietary exposure assessment in the Slovak Republic Food Research Institute - Bratislava Safefoodnet - Chemical Food Safety Network for the enlarging Europe, Bratislava,
Problems with the 1906 Act
 Problems with the 1906 Act failed to provide clear-cut meanings and specific means for enforcement insufficient funding for enforcement USDA was responsible for testing, but no standards for foods were
Problems with the 1906 Act failed to provide clear-cut meanings and specific means for enforcement insufficient funding for enforcement USDA was responsible for testing, but no standards for foods were
Thought Starter Combined Exposures to Multiple Chemicals Second International Conference on Risk Assessment
 Thought Starter Combined Exposures to Multiple Chemicals Second International Conference on Risk Assessment M.E. (Bette) Meek & A. Kortenkamp 1 Outline State of the Art Assessment of Mixtures (aka Combined
Thought Starter Combined Exposures to Multiple Chemicals Second International Conference on Risk Assessment M.E. (Bette) Meek & A. Kortenkamp 1 Outline State of the Art Assessment of Mixtures (aka Combined
CHAPTER 2: RISK ANALYSIS
 Update Project Chapter : Risk Analysis Draft May 00 0 0 0 0 0 PRINCIPLES AND METHODS FOR THE RISK ASSESSMENT OF CHEMICALS IN FOOD CHAPTER : RISK ANALYSIS Contents CHAPTER : RISK ANALYSIS.... INTRODUCTION....
Update Project Chapter : Risk Analysis Draft May 00 0 0 0 0 0 PRINCIPLES AND METHODS FOR THE RISK ASSESSMENT OF CHEMICALS IN FOOD CHAPTER : RISK ANALYSIS Contents CHAPTER : RISK ANALYSIS.... INTRODUCTION....
Applying Risk Assessment Outcomes to Establish Food Standards
 ILSI SEA Region 6th Asian Conference on Food and Nutrition Safety (Nov 2012) http://www.ilsi.org/sea_region/pages/vieweventdetails.aspx?webid=4d540914-eeb6-40e4-89eb-0b73ba3d76c1&listid=478be3cb-581b-4ba2-a280-8e00ccb26f9c&itemid=66
ILSI SEA Region 6th Asian Conference on Food and Nutrition Safety (Nov 2012) http://www.ilsi.org/sea_region/pages/vieweventdetails.aspx?webid=4d540914-eeb6-40e4-89eb-0b73ba3d76c1&listid=478be3cb-581b-4ba2-a280-8e00ccb26f9c&itemid=66
area of toxicology: The following are examples of some LD50 Values LD50 Dose Response Curve Sample
 Chemical hazards are associated with a significant number of high profile food safety alerts, recalls and withdrawals. The principles of HACCP require chemical hazards to be considered during the hazard
Chemical hazards are associated with a significant number of high profile food safety alerts, recalls and withdrawals. The principles of HACCP require chemical hazards to be considered during the hazard
Risk Assessment for Food Safety in Japan
 2009/SOM2/SCSC/WKSP2/010 Risk Assessment for Food Safety in Japan Submitted by: Japan Examination of Hot Issues in Risk Analysis Workshop Singapore 1-2 August 2009 Risk Assessment for Food Safety in Japan
2009/SOM2/SCSC/WKSP2/010 Risk Assessment for Food Safety in Japan Submitted by: Japan Examination of Hot Issues in Risk Analysis Workshop Singapore 1-2 August 2009 Risk Assessment for Food Safety in Japan
From risk assessment to risk management focus on contaminants. Frans Verstraete European Commission DG Health and Consumer Protection
 From risk assessment to risk management focus on contaminants Frans Verstraete European Commission DG Health and Consumer Protection 1 GENERAL FOOD LAW Regulation (EC) 178/2002 of the European Parliament
From risk assessment to risk management focus on contaminants Frans Verstraete European Commission DG Health and Consumer Protection 1 GENERAL FOOD LAW Regulation (EC) 178/2002 of the European Parliament
Using new scientific knowledge to update regulations in the U.S.
 Using new scientific knowledge to update regulations in the U.S. Maricel Maffini, Ph.D. Food Packaging Forum 5 October, 2017 US Food Additives Regulatory Program Administered by the Food and Drug Administration
Using new scientific knowledge to update regulations in the U.S. Maricel Maffini, Ph.D. Food Packaging Forum 5 October, 2017 US Food Additives Regulatory Program Administered by the Food and Drug Administration
Risk Assessment of Chemicals in Foods- WHO Principles and Methods
 Risk Assessment of Chemicals in Foods- WHO Principles and Methods Presented by Dr Debabrata Kanungo DK 31-07-2018 Seminar on Food Additives: A Global Perspect on Safety Evaluation and Use July 19-20, 2018
Risk Assessment of Chemicals in Foods- WHO Principles and Methods Presented by Dr Debabrata Kanungo DK 31-07-2018 Seminar on Food Additives: A Global Perspect on Safety Evaluation and Use July 19-20, 2018
CPH601 Chapter 3 Risk Assessment
 University of Kentucky From the SelectedWorks of David M. Mannino 2013 CPH601 Chapter 3 Risk Assessment David M. Mannino Available at: https://works.bepress.com/david_mannino/64/ + Risk Assessment David
University of Kentucky From the SelectedWorks of David M. Mannino 2013 CPH601 Chapter 3 Risk Assessment David M. Mannino Available at: https://works.bepress.com/david_mannino/64/ + Risk Assessment David
Consultation Response
 Consultation Response CHEM Trust response to EFSA consultation on a draft scientific opinion on: Recent developments in the risk assessment of chemicals in food and their potential impact on the safety
Consultation Response CHEM Trust response to EFSA consultation on a draft scientific opinion on: Recent developments in the risk assessment of chemicals in food and their potential impact on the safety
Introduction. Future U.S. initiatives regarding the food safety for fresh produce. FoodNet Partners. FoodNet Partners
 Introduction Future U.S. initiatives regarding the food safety for fresh produce This presentation is based upon FDA s testimony about the E. coli outbreaks to the U.S. Congress delivered on November 15,
Introduction Future U.S. initiatives regarding the food safety for fresh produce This presentation is based upon FDA s testimony about the E. coli outbreaks to the U.S. Congress delivered on November 15,
Health Canada s Safety Assessment Process for Sugar Substitutes
 Health Canada s Safety Assessment Process for Sugar Substitutes Janice Weightman Bureau of Chemical Safety Food Directorate, Health Canada April 24, 2014 Overview Canadians generally have a taste for sweet
Health Canada s Safety Assessment Process for Sugar Substitutes Janice Weightman Bureau of Chemical Safety Food Directorate, Health Canada April 24, 2014 Overview Canadians generally have a taste for sweet
Food Safety Performance Standards: an Epidemiologic Perspective
 Food Safety Performance Standards: an Epidemiologic Perspective Institute t of Medicine i Food dforum Meeting Rajal Mody, MD MPH LCDR US Public Health Service Enteric Diseases Epidemiology Branch Centers
Food Safety Performance Standards: an Epidemiologic Perspective Institute t of Medicine i Food dforum Meeting Rajal Mody, MD MPH LCDR US Public Health Service Enteric Diseases Epidemiology Branch Centers
Texas Commission on Environmental Quality
 Roberta L. Grant, Susan L. Santos, Mike L. Dourson, Stephanie Shirley, Neeraja K. Erraguntla, R. Jeffrey Lewis, and Nancy B. Beck Society of Toxicology, March 22-26, 2015 San Diego, CA Texas Commission
Roberta L. Grant, Susan L. Santos, Mike L. Dourson, Stephanie Shirley, Neeraja K. Erraguntla, R. Jeffrey Lewis, and Nancy B. Beck Society of Toxicology, March 22-26, 2015 San Diego, CA Texas Commission
Incorporating Computational Approaches into Safety Assessment
 Incorporating Computational Approaches into Safety Assessment Kristi Muldoon Jacobs, Ph.D. Supervisory Toxicologist, DFCN, Office of Food Additive Safety Director (Acting), RIS, Office of Dietary Supplement
Incorporating Computational Approaches into Safety Assessment Kristi Muldoon Jacobs, Ph.D. Supervisory Toxicologist, DFCN, Office of Food Additive Safety Director (Acting), RIS, Office of Dietary Supplement
Understanding the Public Health Significance of Shiga Toxin-Producing E. coli. Betsy Booren, Ph.D. Director, Scientific Affairs
 Understanding the Public Health Significance of Shiga Toxin-Producing E. coli Betsy Booren, Ph.D. Director, Scientific Affairs June 21, 2011 A New Concern for the Meat Industry? The meat industry has long
Understanding the Public Health Significance of Shiga Toxin-Producing E. coli Betsy Booren, Ph.D. Director, Scientific Affairs June 21, 2011 A New Concern for the Meat Industry? The meat industry has long
Risk assessment of mycotoxins: the EFSA approach. Katleen Baert Scientific officer, EFSA
 Risk assessment of mycotoxins: the EFSA approach Katleen Baert Scientific officer, EFSA International Conference The burden of Mycotoxins on animal and human health Rome, 15 December 2017 RECEIPT OF A
Risk assessment of mycotoxins: the EFSA approach Katleen Baert Scientific officer, EFSA International Conference The burden of Mycotoxins on animal and human health Rome, 15 December 2017 RECEIPT OF A
FSMA, FSVP, and FCS. Deborah Attwood May 12, 2016 Global Food Contact 2016
 FSMA, FSVP, and FCS Deborah Attwood May 12, 2016 Global Food Contact 2016 Food Safety Modernization Act (FSMA) Became law in January 2011 Improves safety and security of food supply through prevention,
FSMA, FSVP, and FCS Deborah Attwood May 12, 2016 Global Food Contact 2016 Food Safety Modernization Act (FSMA) Became law in January 2011 Improves safety and security of food supply through prevention,
Distinguishing between Mode and Mechanism of Action
 Distinguishing between Mode and Mechanism of Action Michael Dourson Toxicology Excellence for Risk Assessment Center University of Cincinnati College of Medicine Conflict of Interest Statement The research
Distinguishing between Mode and Mechanism of Action Michael Dourson Toxicology Excellence for Risk Assessment Center University of Cincinnati College of Medicine Conflict of Interest Statement The research
Impact of Melamine Pet Food Contamination on the Meat Industry
 Impact of Melamine Pet Food Contamination on the Meat Industry American Meat Science Association 60 th Reciprocal Meat Conference South Dakota State University June 20, 2007 James H. Hodges President American
Impact of Melamine Pet Food Contamination on the Meat Industry American Meat Science Association 60 th Reciprocal Meat Conference South Dakota State University June 20, 2007 James H. Hodges President American
Hormones. Hormones. When you hear the word hormones, what do you think of? When you hear the word hormones, what do you think of?
 When you hear the word hormones, what When you hear the word hormones, what When you hear the word hormones, what When you hear the word hormones, what Hormones As your body s chemical messengers, hormones
When you hear the word hormones, what When you hear the word hormones, what When you hear the word hormones, what When you hear the word hormones, what Hormones As your body s chemical messengers, hormones
USDA s New Shiga Toxin- Producing Escherichia coli Policy. James Hodges Executive Vice President American Meat Institute
 USDA s New Shiga Toxin- Producing Escherichia coli Policy James Hodges Executive Vice President American Meat Institute June 20, 2012 Final Determination and Request for Comments (FDRC) September 20, 2011
USDA s New Shiga Toxin- Producing Escherichia coli Policy James Hodges Executive Vice President American Meat Institute June 20, 2012 Final Determination and Request for Comments (FDRC) September 20, 2011
Application of human epidemiological studies to pesticide risk assessment
 Workshop What does the future hold for harmonised human health risk assessment of plant protection products? Application of human epidemiological studies to pesticide risk assessment Antonio F. Hernández,
Workshop What does the future hold for harmonised human health risk assessment of plant protection products? Application of human epidemiological studies to pesticide risk assessment Antonio F. Hernández,
The Role of USDA s Food Safety and Inspection Service to Ensure Foodborne Disease Control and Prevention
 The Role of USDA s Food Safety and Inspection Service to Ensure Foodborne Disease Control and Prevention David Goldman, MD, MPH Assistant Administrator Office of Public Health Science Agricultural Outlook
The Role of USDA s Food Safety and Inspection Service to Ensure Foodborne Disease Control and Prevention David Goldman, MD, MPH Assistant Administrator Office of Public Health Science Agricultural Outlook
Pesticide Product Labels What the label says.and Why. Dr. Jeff Birk BASF Corporation Regulatory Manager
 Pesticide Product Labels What the label says.and Why Dr. Jeff Birk BASF Corporation Regulatory Manager Regulation of Pesticides The pesticide industry is regulated by the USEPA. The pesticide industry
Pesticide Product Labels What the label says.and Why Dr. Jeff Birk BASF Corporation Regulatory Manager Regulation of Pesticides The pesticide industry is regulated by the USEPA. The pesticide industry
THE EU LEGISLATION FOR HONEY RESIDUE CONTROL
 APIACTA 38 (2003) 15-20 15 THE EU LEGISLATION FOR HONEY RESIDUE CONTROL Roberto Piro, Franco Mutinelli Istituto Zooprofilattico Sperimentale delle Venezie, 35020 Legnaro (PD) Italy, Tel ++39 049 8084340,
APIACTA 38 (2003) 15-20 15 THE EU LEGISLATION FOR HONEY RESIDUE CONTROL Roberto Piro, Franco Mutinelli Istituto Zooprofilattico Sperimentale delle Venezie, 35020 Legnaro (PD) Italy, Tel ++39 049 8084340,
Risk management of mycotoxin presence in food products:major unsolved points
 Ministero della Salute Risk management of mycotoxin presence in food products:major unsolved points Directorate General for food hygiene and safety and for nutrition Dr Monica CAPASSO Risk management Regulation
Ministero della Salute Risk management of mycotoxin presence in food products:major unsolved points Directorate General for food hygiene and safety and for nutrition Dr Monica CAPASSO Risk management Regulation
RISK21: Q-KEDRF. ILSI Health and Environmental Sciences Institute
 RISK21: Q-KEDRF ILSI Health and Environmental Sciences Institute Mode of Action 3 Risk / Safety In vivo In vitro 4 Toxicity QSAR/ TTC Matrix Biomonitoring 2 Probabilistic Deterministic Minimal Info Exposure
RISK21: Q-KEDRF ILSI Health and Environmental Sciences Institute Mode of Action 3 Risk / Safety In vivo In vitro 4 Toxicity QSAR/ TTC Matrix Biomonitoring 2 Probabilistic Deterministic Minimal Info Exposure
International Federation of Gynecology and Obstetrics
 International Federation of Gynecology and Obstetrics WORKING GROUP ON REPRODUCTIVE AND DEVELOPMENTAL ENVIRONMENTAL HEALTH Health Practitioners Warning on the Impacts of Chemical Contamination on Human
International Federation of Gynecology and Obstetrics WORKING GROUP ON REPRODUCTIVE AND DEVELOPMENTAL ENVIRONMENTAL HEALTH Health Practitioners Warning on the Impacts of Chemical Contamination on Human
Message Testing Results
 Message Testing Results From April to June 2014, HealthPartners Institute for Education and Research conducted a survey of women of child-bearing age in which they assessed responses to a series of messages
Message Testing Results From April to June 2014, HealthPartners Institute for Education and Research conducted a survey of women of child-bearing age in which they assessed responses to a series of messages
Update FDA/Center for Food Safety and Applied Nutrition
 Update FDA/Center for Food Safety and Applied Nutrition Comments by Susan Mayne, Ph.D. Director Center for Food Safety and Applied Nutrition FDLI Annual Conference May 3, 2018 Food Safety, Nutrition and
Update FDA/Center for Food Safety and Applied Nutrition Comments by Susan Mayne, Ph.D. Director Center for Food Safety and Applied Nutrition FDLI Annual Conference May 3, 2018 Food Safety, Nutrition and
CDR Kimberly Elenberg, Office of Food Defense and Emergency Response, Food Safety Inspection Service, United States Department of Agriculture
 The Consumer Complaint Monitoring System Enhancing Discovery and Mitigation of Foodborne Threats to Health through Pattern Surveillance, and Multiple-Attribute Algorithms CDR Kimberly Elenberg, Office
The Consumer Complaint Monitoring System Enhancing Discovery and Mitigation of Foodborne Threats to Health through Pattern Surveillance, and Multiple-Attribute Algorithms CDR Kimberly Elenberg, Office
Risk Assessment Toolbox. Risk Analysis Training
 Risk Assessment Toolbox Risk Analysis Training Risk Profiles in Codex PURPOSE: The risk profile is an abbreviated discussion paper that lays out the key elements of a MRM concern in order to facilitate
Risk Assessment Toolbox Risk Analysis Training Risk Profiles in Codex PURPOSE: The risk profile is an abbreviated discussion paper that lays out the key elements of a MRM concern in order to facilitate
b Public Consultation on Aspartame: ISA COMMENTS
 EFSA follow-up meeting on the web-based b Public Consultation on Aspartame: ISA COMMENTS Emeritus Prof. Andrew G. Renwick, OBE, University of Southampton (UK), Scientific consultant to the ISA Dr. Hervé
EFSA follow-up meeting on the web-based b Public Consultation on Aspartame: ISA COMMENTS Emeritus Prof. Andrew G. Renwick, OBE, University of Southampton (UK), Scientific consultant to the ISA Dr. Hervé
FDA Food Contact Fundamentals
 FDA Food Contact Fundamentals Deborah Attwood 2017 SPE International Polyolefins Conference March 1, 2017 Legal Highlights for Food 1906: Pure Food and Drug Act 1938: Federal Food, Drug and Cosmetic Act
FDA Food Contact Fundamentals Deborah Attwood 2017 SPE International Polyolefins Conference March 1, 2017 Legal Highlights for Food 1906: Pure Food and Drug Act 1938: Federal Food, Drug and Cosmetic Act
International Safety Assessment of Sweeteners
 ILSI SEA Region Seminar on Uses and Safety of Sweeteners (May 2013) http://www.ilsi.org/sea_region/pages/vieweventdetails.aspx?webid=4d540914-eeb6-40e4-89eb- 0B73BA3D76C1&ListId=478BE3CB-581B-4BA2-A280-8E00CCB26F9C&ItemID=73
ILSI SEA Region Seminar on Uses and Safety of Sweeteners (May 2013) http://www.ilsi.org/sea_region/pages/vieweventdetails.aspx?webid=4d540914-eeb6-40e4-89eb- 0B73BA3D76C1&ListId=478BE3CB-581B-4BA2-A280-8E00CCB26F9C&ItemID=73
Assessing Risks of Exposure to Allergens from Foods
 Assessing Risks of Exposure to Allergens from Foods Joe Baumert, Ph.D. Co-Director Food Allergy Research & Resource Program University of Nebraska The National Academies of Science Institute of Medicine
Assessing Risks of Exposure to Allergens from Foods Joe Baumert, Ph.D. Co-Director Food Allergy Research & Resource Program University of Nebraska The National Academies of Science Institute of Medicine
CONTRASTS AND INTERACTIONS
 (Italian ) Monsanto. Portugal! CONTRASTS AND INTERACTIONS BETWEEN EPIDEMIOLOGY AND TOXICOLOGY BRIEF COMMENTS Daniel A. Goldstein, M.D. Senior Science Fellow Lead, Medical Sciences and Outreach Monsanto
(Italian ) Monsanto. Portugal! CONTRASTS AND INTERACTIONS BETWEEN EPIDEMIOLOGY AND TOXICOLOGY BRIEF COMMENTS Daniel A. Goldstein, M.D. Senior Science Fellow Lead, Medical Sciences and Outreach Monsanto
Consumer Concerns About Hormones in Food
 Consumer Concerns About Hormones in Food Fact Sheet #37, June 2000 This fact sheet addresses some of the consumer concerns that have been brought to BCERF regarding health effects of hormones used by the
Consumer Concerns About Hormones in Food Fact Sheet #37, June 2000 This fact sheet addresses some of the consumer concerns that have been brought to BCERF regarding health effects of hormones used by the
FDA Center for Tobacco Products: Tobacco Research and the Population Assessment of Tobacco and Health (PATH) Study
 FDA Center for Tobacco Products: Tobacco Research and the Population Assessment of Tobacco and Health (PATH) Study Regulatory Public Laws Compliance & Education Policies Science & Enforcement & Communications
FDA Center for Tobacco Products: Tobacco Research and the Population Assessment of Tobacco and Health (PATH) Study Regulatory Public Laws Compliance & Education Policies Science & Enforcement & Communications
Infant TDS. Results of the ANSES study on dietary exposure of children under 3 years of age to chemical substances
 Infant TDS Results of the ANSES study on dietary exposure of children under 3 years of age to chemical substances 2010-2016 29 th Focal point meeting Uppsala, Sweden Why an infant Total Diet Study TDS
Infant TDS Results of the ANSES study on dietary exposure of children under 3 years of age to chemical substances 2010-2016 29 th Focal point meeting Uppsala, Sweden Why an infant Total Diet Study TDS
OVERVIEW OF FOOD SAFETY REGULATION IN THE UNITED STATES. Presented By William C. Balek International Sanitary Supply Association March 30, 2001
 OVERVIEW OF FOOD SAFETY REGULATION IN THE UNITED STATES Presented By William C. Balek International Sanitary Supply Association March 30, 2001 I INTRODUCTION The regulation of food safety in the United
OVERVIEW OF FOOD SAFETY REGULATION IN THE UNITED STATES Presented By William C. Balek International Sanitary Supply Association March 30, 2001 I INTRODUCTION The regulation of food safety in the United
The role of biokinetics in in vitro tests and the interpretation of results. Emanuela Testai
 International Symposium on Alternative in vitro methods to characterize the role of EAS in hormone-targeted tissues Rome 17.12.2012 The role of biokinetics in in vitro tests and the interpretation of results
International Symposium on Alternative in vitro methods to characterize the role of EAS in hormone-targeted tissues Rome 17.12.2012 The role of biokinetics in in vitro tests and the interpretation of results
ACTIVITIES DEVELOPED AND PROMOTED BY WHO
 IFCS FORUM V SIDE EVENT ON HEAVY METALS Hosted by the Swiss Confederation 23 September 2006 Budapest, Hungary Health and environmental concerns associated with heavy metals: Global needs for further action?
IFCS FORUM V SIDE EVENT ON HEAVY METALS Hosted by the Swiss Confederation 23 September 2006 Budapest, Hungary Health and environmental concerns associated with heavy metals: Global needs for further action?
RECALL INDEX 2016 Q2
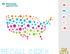 RECALL INDEX Recall Index, Consumer Products FURNISHINGS IN THE HOT SEAT Consumer product recalls increased by just 8%, but that rise represents the second highest number of recalls since 2011. Recalled
RECALL INDEX Recall Index, Consumer Products FURNISHINGS IN THE HOT SEAT Consumer product recalls increased by just 8%, but that rise represents the second highest number of recalls since 2011. Recalled
Canada-U.S. Food Safety Risk Assessment Organization: Case Study
 Canada-U.S. Food Safety Risk Assessment Organization: Case Study Wilson Center, Washington DC, Monday, October 23, 2017 Yadira Tejeda, Postdoctoral Research Fellow Lawrence Centre, Ivey Business School,
Canada-U.S. Food Safety Risk Assessment Organization: Case Study Wilson Center, Washington DC, Monday, October 23, 2017 Yadira Tejeda, Postdoctoral Research Fellow Lawrence Centre, Ivey Business School,
CONCEPTUAL FRAMEWORK FOR A TIERED APPROACH TO RISK RANKING AND PRIORITIZATION
 Consultants in Human Health, Toxicology & Regulatory Affairs CONCEPTUAL FRAMEWORK FOR A TIERED APPROACH TO RISK RANKING AND PRIORITIZATION Ian C. Munro, Ph.D., F.A.T.S., FRCPath Workshop: Tools for Prioritizing
Consultants in Human Health, Toxicology & Regulatory Affairs CONCEPTUAL FRAMEWORK FOR A TIERED APPROACH TO RISK RANKING AND PRIORITIZATION Ian C. Munro, Ph.D., F.A.T.S., FRCPath Workshop: Tools for Prioritizing
Discussion of Changes in the Draft Preamble
 Discussion of Changes in the Draft Preamble Prepared by the staff of the IARC Monographs programme 31 August 2005 This paper describes the major changes that appear in the draft Preamble that will be reviewed
Discussion of Changes in the Draft Preamble Prepared by the staff of the IARC Monographs programme 31 August 2005 This paper describes the major changes that appear in the draft Preamble that will be reviewed
Managing Risk in a Zero Tolerance World: International Impact of Risk Assessment
 Managing Risk in a Zero Tolerance World: International Impact of Risk Assessment Robert L. Buchanan Department of Nutrition and Food Science Presentation Historical Perspective Consideration of Dose-Response
Managing Risk in a Zero Tolerance World: International Impact of Risk Assessment Robert L. Buchanan Department of Nutrition and Food Science Presentation Historical Perspective Consideration of Dose-Response
Mercury Contaminated Seafood: State Advisories and Other Protective Steps
 Mercury Contaminated Seafood: State Advisories and Other Protective Steps Michael T. Bender, Director 1420 North Street Montpelier, VT 05602 802-223-9000 (ph) 802-223-7914 (f) www.mercurypolicy.org Mercury
Mercury Contaminated Seafood: State Advisories and Other Protective Steps Michael T. Bender, Director 1420 North Street Montpelier, VT 05602 802-223-9000 (ph) 802-223-7914 (f) www.mercurypolicy.org Mercury
RECALL INDEX Q3 2017
 RECALL INDEX Recall Index Consumer product recalls dipped 8% to 59 the lowest number since. Recalled units increased 35% to 4.6 million, which is still the second lowest since 2012. 80 CONSUMER RECALLS
RECALL INDEX Recall Index Consumer product recalls dipped 8% to 59 the lowest number since. Recalled units increased 35% to 4.6 million, which is still the second lowest since 2012. 80 CONSUMER RECALLS
DANIEL R. DOERGE U.S. Food and Drug Administration National Center for Toxicological Research Jefferson, AR
 Research support by Interagency Agreement between NTP/NIEHS and NCTR/FDA The opinions presented are not necessarily those of the U.S. FDA or NTP NCTR/FDA Research on BPA: Integrating pharmacokinetics in
Research support by Interagency Agreement between NTP/NIEHS and NCTR/FDA The opinions presented are not necessarily those of the U.S. FDA or NTP NCTR/FDA Research on BPA: Integrating pharmacokinetics in
AGRICULTURAL CHEMICAL SAFETY ASSESSMENT (ACSA)
 HESI ILSI Health and Environmental Sciences Institute AGRICULTURAL CHEMICAL SAFETY ASSESSMENT (ACSA) ADME Task Force Hugh A. Barton, PhD National Center for Computational Toxicology Office of Research
HESI ILSI Health and Environmental Sciences Institute AGRICULTURAL CHEMICAL SAFETY ASSESSMENT (ACSA) ADME Task Force Hugh A. Barton, PhD National Center for Computational Toxicology Office of Research
Ongoing review of legislation on cadmium in food in the EU: Background and current state of play
 Directorate-General for Health & Ongoing review of legislation on cadmium in food in the EU: Background and current state of play - International ICCO workshop, London, 3-4 May 2012 Michael Flüh bind the
Directorate-General for Health & Ongoing review of legislation on cadmium in food in the EU: Background and current state of play - International ICCO workshop, London, 3-4 May 2012 Michael Flüh bind the
Cydia pomonella Granulovirus Strain M
 Registration Decision RD2014-25 Cydia pomonella Granulovirus Strain M (publié aussi en français) 21 August 2014 This document is published by the Health Canada Pest Management Regulatory Agency. For further
Registration Decision RD2014-25 Cydia pomonella Granulovirus Strain M (publié aussi en français) 21 August 2014 This document is published by the Health Canada Pest Management Regulatory Agency. For further
Food. Safety Risk Analysis. Training Program. University of Maryland. Food and Drug Administration Patapsco Bldg.
 Food JIFSAN Joint Institute for Food Safety and Applied Nutrition University of Maryland Food and Drug Administration Safety Risk Analysis 2134 Patapsco Bldg. University of Maryland College Park, Maryland
Food JIFSAN Joint Institute for Food Safety and Applied Nutrition University of Maryland Food and Drug Administration Safety Risk Analysis 2134 Patapsco Bldg. University of Maryland College Park, Maryland
Taylor C. Wallace, PhD, CFS, FACN, March 22, 2018
 Food Ingredients Taylor C. Wallace, PhD, CFS, FACN, March 22, 2018 Disclosures Think Healthy Group, Inc. George Mason University, Department of Nutrition and Food Studies Journal of the American College
Food Ingredients Taylor C. Wallace, PhD, CFS, FACN, March 22, 2018 Disclosures Think Healthy Group, Inc. George Mason University, Department of Nutrition and Food Studies Journal of the American College
Persistent Organic Pollutants (POPs) in Human Breast Milk Collected from Asian Developing Countries: Risk Assessment for Infants
 Persistent Organic Pollutants (POPs) in Human Breast Milk Collected from Asian Developing Countries: Risk Assessment for Infants Shinsuke Tanabe Ehime University, Japan SL 1: Title Thank you Mr. Chairman
Persistent Organic Pollutants (POPs) in Human Breast Milk Collected from Asian Developing Countries: Risk Assessment for Infants Shinsuke Tanabe Ehime University, Japan SL 1: Title Thank you Mr. Chairman
Risk Assessment for Food Safety an introduction
 Risk Assessment for Food Safety an introduction Chrystalleni Hadjicharalambous EFSA Fellow, EU-FORA Programme 2018 UNIVERSITY OF CRETE Outline Burden of foodborne disease EU Regulations and the establishment
Risk Assessment for Food Safety an introduction Chrystalleni Hadjicharalambous EFSA Fellow, EU-FORA Programme 2018 UNIVERSITY OF CRETE Outline Burden of foodborne disease EU Regulations and the establishment
Overview of USDA Inspection
 Overview of USDA Inspection Dr. Douglas Powell Professor Department of Diagnostic Medicine/Pathobiology Kansas State University dpowell@ksu.edu bites.ksu.edu barfblog.com http://barfblog.com/2013/05/food-safety-apology-i-texas-health-type-says-sorry-to-family/
Overview of USDA Inspection Dr. Douglas Powell Professor Department of Diagnostic Medicine/Pathobiology Kansas State University dpowell@ksu.edu bites.ksu.edu barfblog.com http://barfblog.com/2013/05/food-safety-apology-i-texas-health-type-says-sorry-to-family/
Human Health Risk Assessment Overview [For the APS/OPP Roundtable]
![Human Health Risk Assessment Overview [For the APS/OPP Roundtable] Human Health Risk Assessment Overview [For the APS/OPP Roundtable]](/thumbs/74/69622095.jpg) Human Health Risk Assessment Overview [For the APS/OPP Roundtable] Christina Swartz, USEPA Nov. 12, 2008 [Slides Courtesy of Mike Metzger, USEPA] The Risk Assessment Paradigm: The Red Book Hazard Identification
Human Health Risk Assessment Overview [For the APS/OPP Roundtable] Christina Swartz, USEPA Nov. 12, 2008 [Slides Courtesy of Mike Metzger, USEPA] The Risk Assessment Paradigm: The Red Book Hazard Identification
What are the challenges in addressing adjustments for data uncertainty?
 What are the challenges in addressing adjustments for data uncertainty? Hildegard Przyrembel, Berlin Federal Institute for Risk Assessment (BfR), Berlin (retired) Scientific Panel for Dietetic Foods, Nutrition
What are the challenges in addressing adjustments for data uncertainty? Hildegard Przyrembel, Berlin Federal Institute for Risk Assessment (BfR), Berlin (retired) Scientific Panel for Dietetic Foods, Nutrition
Risk analysis of foodborne antimicrobial resistance (AMR) Suphachai Nuanualsuwan DVM, MPVM, PhD1
 Risk analysis of foodborne antimicrobial resistance (AMR) Suphachai Nuanualsuwan 1 Introduction Antimicrobial resistance (AMR) is major global public health concern & food safety issue Risk Analysis :-
Risk analysis of foodborne antimicrobial resistance (AMR) Suphachai Nuanualsuwan 1 Introduction Antimicrobial resistance (AMR) is major global public health concern & food safety issue Risk Analysis :-
Submitted via Consumers Union National Consumers League Center for Foodborne Illness Research & Prevention
 U.S. Department of Agriculture Food Safety and Inspection Service Patriots Plaza 3 1400 Independence Avenue SW Mailstop 3782, Room 8-163B Washington, DC 20250-3700 Submitted via www.regulations.gov Consumers
U.S. Department of Agriculture Food Safety and Inspection Service Patriots Plaza 3 1400 Independence Avenue SW Mailstop 3782, Room 8-163B Washington, DC 20250-3700 Submitted via www.regulations.gov Consumers
FAO/WHO Training Manual. Training Programme for Developing Food Standards within a Risk Analysis Framework
 FAO/WHO Training Manual Training Programme for Developing Food Standards within a Risk Analysis Framework Food and Agriculture Organization of the United Nations World Health Organization July 2006 Finalized
FAO/WHO Training Manual Training Programme for Developing Food Standards within a Risk Analysis Framework Food and Agriculture Organization of the United Nations World Health Organization July 2006 Finalized
Proposition 65 and Supplements
 Proposition 65 and Supplements Nutrition Industry Association John Venardos Senior Vice President Worldwide Regulatory & Government Affairs Herbalife International of America, Inc. 5/23/2011 SYNOPSIS Now
Proposition 65 and Supplements Nutrition Industry Association John Venardos Senior Vice President Worldwide Regulatory & Government Affairs Herbalife International of America, Inc. 5/23/2011 SYNOPSIS Now
