Wood Extractives II Agenda. Wood Chemistry. Hydrolyzable Tannins Structure. Hydrolyzable Tannins. PSE 406/Chem E 470. Polymer Structure Example
|
|
|
- Derek Simon
- 6 years ago
- Views:
Transcription
1 Wood II Agenda PSE 406/Chem E 470 Lecture 16 Wood, Components and Analysis Hydrolyzable Tannins» Chemistry, biological significance, commercial uses, pulp and paper problems. Miscellaneous» Tetraterpenes, Phenolics, Alkaloids, Etc. Extractive Procedures Extractive Contents Wood Analysis Procedures General Wood Compositions PSE 406: Lecture 16 1 PSE 406: Lecture 16 2 Hydrolyzable Tannins Structure Hydrolyzable Tannins Polymer Structure Example Polymers (esters) of a sugar (usually glucose) with one or more polyphenolic carboxylic acids. Gallotannins: Gallic acid polymer Ellagitannins: Ellagic Acid polymer H H H H H H H H H H H H H H H Sugar H H H CH Gallic Acid C H H C H Digallic Acid C C H H Ellagic Acid H H H H H H n PSE 406: Lecture 16 3 PSE 406: Lecture
2 Hydrolyzable Tannins General Information Tetraterpenes Rare to nonexistent in softwoods. Hardwoods which contain large amounts:» ak (gallic and ellagic tannins)» Eucalyptus (Ellagitannins)» Chestnut (gallic tannins) Hydrolyzable tannins located in heartwood. Pulp and paper problems:» Increased consumption of bleaching chemicals.» Coloration problems. Polymers of isoprene containing 40 carbons (8 isoprene units) Most common are the carotenoids» Generally derivatives of lycopene This is a carontenoid which is in high concentration in red fruits (gives color): tomatoes, watermelon, guava, etc. PSE 406: Lecture 16 5 PSE 406: Lecture 16 6 β Carotene Phenolics Tetraterpene carotenoid found in dark green and orange yellow vegetables. In the human body, it is converted to vitamin A Has been shown to be have many health related benefits. PSE 406: Lecture 16 7 Aromatic odor» Balsam odor» Foliage, bark, fruits Sometimes in heartwood» Variety of aromatic compounds» Typically in small amounts (1%<) CH2 CH CH2 H CH3 Eugenol (il of Clove) C H 3 CH 2 CH CH 2 C H2 Myristicin (Extracted fron utmeg) CH2 CH CH2 CH2 Safrole (75% of Sassafras) CH3 CH2 CH CH2 H Elemicn CH3 PSE 406: Lecture
3 Phenolics Highly Colored Compounds CH H Vanillin CH3 H CH Salicylic Acid (asprin - close) CH CH3 CH3 Veratraldehyde (Smells like vanilla) H CH2H Salicyl Alcohol C H CH CH Cinnamic Acid (Cinnamon - Bark CH CH3 H Vanillic Acid CH 2 CH CH2 CH3 Methyl Chavicol (Pine turpentine) CH 3 C CH3 H Acetovanillone PSE 406: Lecture 16 9 Anthraquinone» Found in Teak» Used as pulping catalyst* aphthaquinones» Used as UV absorbers» Used in polymer products* Benzoquinones * Produced from other sources Anthraquinone H PSE 406: Lecture Juglone (Pigment in walnuts) H Lawsone (apthaquinone) Coumarin ther Coumarin Facts Chemical which has the aroma of fresh mowed hay (or vanilla).» Produced by white clover.» When clover is cut, a glycosylated cinnamic acid is enzymatically cleaved releasing glucose and a hydroxy cinnamic acid which esterifies. Coumarin produced in the fruit and nuts of certain trees (tonka trees for example) Coumarin is used as a rat poison. Coumarin Phytoalexins» Anti-microbial agents ften exist as glycosides Can cause health problems» Coumarin substituted for vanillin by shady individuals.» FDA banned coumarin for human consumption in Some coumarins are commercially used as UV absorbers. (These are typically synthetic products from petroleum) H H Esculetin CH 3 H Scopoletin CH3 H H Fraxetin PSE 406: Lecture PSE 406: Lecture
4 Alkaloids Alkaloids These nitrogen containing compounds are found in a variety of different plants.» Located in the leaves, fruits, and bark. Quinine» Used to treat malaria» Isolated from bark of cinchonas (South American Hardwood) some eucalyptus species» Causes pulping problems Strychnine» Found in seeds of tree Strychnos nux - vomica (Tropical hardwood) H 3C PSE 406: Lecture H Quinine H - + CH 3 Strychnine CH 2 You are all aware of the alkaloids shown on this page you probably have never seen their structures. These are typically found in small amounts in plants but are worth large sums of cash. icotine PSE 406: Lecture Cocaine Caffeine Miscellaneous Chlorophylls Proteins & Amino acids» Enzymes used by living organisms are proteins made of amino acids.» Plant contain small amounts (~0.5%) of proteins.» Chlorophyll (need the structure) Sugars» Glycosides or free sugars. Many extractives in plants have sugars attached which are often freed in enzymatic processes. There are typically low levels of free sugars in plants with som e notable exceptions: sugar cane and sugar beets. These plants have high levels of free sucrose.» Some of the water soluble hemicelluloses Pectins are often called extractives. PSE 406: Lecture Probably the most well known chemicals in plants are the chlorophylls. These are tetrapyrroles that are located in the chloroplasts of most all photosynthetic plant tissues. As we all know, chlorophylls give leaves their green color. PSE 406: Lecture M e 2 C M g Chlorophyll a 4 4
5 Inorganic Materials Inorganics in Grasses Inorganic materials» Plants contains small amounts of most metals. Some are used by living cells Some present as contaminants Ash content gives a rough idea of the amounts of metals present.» Sugar cane: % ash» Corn Cobs: 2% ash Temperate woods contain 0.1-1% ash while tropical and subtropical species contain up to 5% ash.» Main components Ca (50%), potassium and magnesium. Grasses contain high levels of inorganics particularly silica. Wheat straw: Reported values 4-12+% ash» Leaves: 14-19% ash, 10-14% silica» odes: 8-14% ash, 5-7% silica» Internodes: 3+% ash, 2+% silica» Potasium: 1% PSE 406: Lecture PSE 406: Lecture Extraction Procedures Western Hemlock Extraction Technique Steam Distillation Products Terpenes Phenols Hydrocarbons Lignans Ether Extraction Fats/Waxes Resin Acids Sterols Alcohol Extraction Flavonoids Phlobaphenes Tannins Stilbenes Water Extraction Carbohydrates Protein/Alkaloids Inorganics PSE 406: Lecture Extractive % on Wood Wax 0.3% Flavonoids/Lignans 1.7% Condensed Tannin 1.7% Water sol. Carbohydrates 0.8% Total 4.5% * Western Hemlock contains only minimal tall oil type compounds (resin acids, fatty acids, sterols, terpenes, etc) PSE 406: Lecture
6 on-volatile Resin Composition (1960 data for reference) in Douglas Fir Wood Ether Spruce Pine Aspen Birch % % 1-2.7% % - Resin Acids 31% 30-41% 0% 31% - Free Fatty Acids 0% 3-8% 35% 6% - Fatty Acid Esters 42% 40-60% 50% 25% - Unsaponifiables 21% 7-11% 14% 30% Use this information as a relative comparison only Sapwood Heartwood Information taken from a variety of different sources Condensed Tannins Flavonoids Stilbenes Lignans Fatty Acids Resin Acids Waxes Fats eutrals (sterols, etc) Monoterpenes PSE 406: Lecture PSE 406: Lecture Quantification of Major Wood Components Quantification of Major Wood Components 1. Wood Meal Preparation Wiley Wood Chips, etc. Mill Wood Meal (40-60 mesh) 2. Extraction Using Sohxlet Apparatus (to remove extractives) a. Alcohol:Benzene (2:1) b. Acetone c. Water 3. Lignin a. Swell with 72% H 2 S 4 for several hours b. Reflux in 3% H 2 S 4 c. Weigh residue = Klason Lignin d. Hardwood Lignin Soluble Lignin by UV 4. Holocellulose (Total Polysaccharides) Cl Extractive Free Wood Meal 2 Holocellulose Procedure dissolves lignin 5. Alpha Cellulose 17.5% ah Holocellulose Macerate 6. Hemicellulose Cellulose (Insoluble Fraction) Hemicellulose = Holocellulose - Alpha Cellulose PSE 406: Lecture PSE 406: Lecture
7 Quantification of Major Wood Components Chemical Composition of Wood: Hardwoods Extraneous Soluble in eutral Solvents Compounds Insoluble Acetyl Groups Wood Meal Solvent Extraction Lignin H 2 S 4 Cl % ah Insoluble Cellulose 2 H 2 S 4 D- Glucose Uronic Acids Pentoses Holocellulose Hemicellulose (soluble) 2 H 2 S 4 Hexoses PSE 406: Lecture % Red Maple English ak Iroko Balsa Cellulose* Hemicellulose* Lignin* * Data for Cellulose, Hemicellulose & Lignin on extractive free wood basis PSE 406: Lecture Chemical Composition of Wood: Softwoods % Cellulose* Hemicellulose* Lignin* 0 Douglas Fir Redwood Yellow Pine Balsam Fir * Data for Cellulose, Hemicellulose & Lignin on extractive free wood basis PSE 406: Lecture
Wood Chemistry. What are Fats and Oils? Agenda. PSE 406/Chem E 470. Chemical Composition of Fat/Oils. Lecture 14 Fats/Waxes/Lignans
 Agenda PSE 406/Chem E 470 Lecture 14 Fats/Waxes/ Fats and Fatty Acids» Chemistry» Role in the tree Tall oil Waxes» Chemistry» Role in the tree» Commercialization PSE 406 Lecture 14 1 PSE 406 Lecture 14
Agenda PSE 406/Chem E 470 Lecture 14 Fats/Waxes/ Fats and Fatty Acids» Chemistry» Role in the tree Tall oil Waxes» Chemistry» Role in the tree» Commercialization PSE 406 Lecture 14 1 PSE 406 Lecture 14
The Chemistry of Wood and Kraft Pulping. 1
 The Chemistry of Wood and Kraft Pulping ragauskas@hotmail.com 1 Typical Composition of Wood Cellulose (41-53%) Hemicellulose (25-41%) Lignin (16-33%) Extractives (2-5%) Inorganics (
The Chemistry of Wood and Kraft Pulping ragauskas@hotmail.com 1 Typical Composition of Wood Cellulose (41-53%) Hemicellulose (25-41%) Lignin (16-33%) Extractives (2-5%) Inorganics (
Terpènes, colophane et TOFA extraits du bois : une valeur ajoutée pour les usines de pâtes à papier
 Terpènes, colophane et TOFA extraits du bois : une valeur ajoutée pour les usines de pâtes à papier F.BERTAUD, M. PETIT-CONIL Centre Technique du Papier - Grenoble Mercredi 10 Mai 2017, CTP, Grenoble Journée
Terpènes, colophane et TOFA extraits du bois : une valeur ajoutée pour les usines de pâtes à papier F.BERTAUD, M. PETIT-CONIL Centre Technique du Papier - Grenoble Mercredi 10 Mai 2017, CTP, Grenoble Journée
Agenda. Wood Chemistry. Stilbenes Biological Significance. Stilbenes. PSE 406/Chem E 470. Stilbenes. Flavonoids
 Agenda PSE 06/Chem E 70 Lecture 1,, and Condensed Tannins PSE 06: Lecture 1 1 PSE 06: Lecture 1 Biological Significance Phenolic extractive found in the heartwood of softwoods» Particularly prevalent in
Agenda PSE 06/Chem E 70 Lecture 1,, and Condensed Tannins PSE 06: Lecture 1 1 PSE 06: Lecture 1 Biological Significance Phenolic extractive found in the heartwood of softwoods» Particularly prevalent in
NATURAL PRODUCTS A Laboratory Guide
 NATURAL PRODUCTS A Laboratory Guide Second Edition Raphael Ikan Department of Organic Chemistry The Hebrew University of Jerusalem Jerusalem, Israel Academic Press San Diego New York Boston London Sydney
NATURAL PRODUCTS A Laboratory Guide Second Edition Raphael Ikan Department of Organic Chemistry The Hebrew University of Jerusalem Jerusalem, Israel Academic Press San Diego New York Boston London Sydney
Formulating for Tannin Stain Inhibition
 Formulating for Tannin Stain Inhibition April 23, 2013 Nathan Kofira Tannin staining is the discoloration of a painted surface due to the migration of water soluble Tannins (tannic acids) through the film
Formulating for Tannin Stain Inhibition April 23, 2013 Nathan Kofira Tannin staining is the discoloration of a painted surface due to the migration of water soluble Tannins (tannic acids) through the film
Woody Biomass Conversion: Process Improvements at ESF
 Woody Biomass Conversion: Process Improvements at ESF Shijie Liu Biorefinery Research Institute Department of Paper and Bioprocess Engineering SUNY College of Environmental Science and Forestry Outline
Woody Biomass Conversion: Process Improvements at ESF Shijie Liu Biorefinery Research Institute Department of Paper and Bioprocess Engineering SUNY College of Environmental Science and Forestry Outline
Terpenes and Phenylpropanoids
 Terpenes and Phenylpropanoids Microapproach to Organic Chemistry VY NGUYEN 26 March 2016 ABSTRACT Essential oil components are often found in the glands or intercellular spaces in plant tissue. They may
Terpenes and Phenylpropanoids Microapproach to Organic Chemistry VY NGUYEN 26 March 2016 ABSTRACT Essential oil components are often found in the glands or intercellular spaces in plant tissue. They may
Ethnobotany. Lecture 25
 Ethnobotany. Lecture 25 Alexey Shipunov Minot State University March 28th, 2011 Shipunov (MSU) BIOL 310.25 March 28th, 2011 1 / 42 Outline 1 Shipunov (MSU) BIOL 310.25 March 28th, 2011 2 / 42 Polyketides
Ethnobotany. Lecture 25 Alexey Shipunov Minot State University March 28th, 2011 Shipunov (MSU) BIOL 310.25 March 28th, 2011 1 / 42 Outline 1 Shipunov (MSU) BIOL 310.25 March 28th, 2011 2 / 42 Polyketides
Lecture 19: Soil Organic Matter
 Lecture 19: Soil Organic Matter Effects of OM Properties on Soil Property Dark color Excellent water retention Binds to clay minerals Metal chelation Low water solubility ph buffering High CEC Nutrient
Lecture 19: Soil Organic Matter Effects of OM Properties on Soil Property Dark color Excellent water retention Binds to clay minerals Metal chelation Low water solubility ph buffering High CEC Nutrient
6 CHAPTER-6 TOTAL PHENOLIC AND FLAVONOID CONTENT DETERMINATION
 6 CHAPTER-6 TOTAL PHENOLIC AND FLAVONOID CONTENT DETERMINATION 6.1 PHENOLIC COMPOUNDS Phenolic compounds are a group of chemical compounds that are widely distributed in nature. They are simple compounds
6 CHAPTER-6 TOTAL PHENOLIC AND FLAVONOID CONTENT DETERMINATION 6.1 PHENOLIC COMPOUNDS Phenolic compounds are a group of chemical compounds that are widely distributed in nature. They are simple compounds
Salicylic acid. Hi I m Molly Cool. Extracted from the bark of the willow tree and used to ease aches and pains and reduce fever -
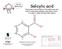 Salicylic acid Extracted from the bark of the willow tree and used to ease aches and pains and reduce fever - it is used to make the pain-killer aspirin and makeitmolecular designed by 7 C Carbon (black)
Salicylic acid Extracted from the bark of the willow tree and used to ease aches and pains and reduce fever - it is used to make the pain-killer aspirin and makeitmolecular designed by 7 C Carbon (black)
9/21/2016. Composition and Compositional Changes During Development: Part II. V. Major Components of Fruits and Vegetables.
 Composition and Compositional Changes During Development: Part II Dr. Jeffrey K. Brecht Horticultural Sciences Department, Gainesville Dr. Mark A. Ritenour Indian River Research and Education Center, Fort
Composition and Compositional Changes During Development: Part II Dr. Jeffrey K. Brecht Horticultural Sciences Department, Gainesville Dr. Mark A. Ritenour Indian River Research and Education Center, Fort
CHEMISTRY OF LIFE 05 FEBRUARY 2014
 CHEMISTRY OF LIFE 05 FEBRUARY 2014 In this lesson we will: Lesson Description Discuss inorganic compounds and their importance Discuss organic compounds and their biological importance. Summary Inorganic
CHEMISTRY OF LIFE 05 FEBRUARY 2014 In this lesson we will: Lesson Description Discuss inorganic compounds and their importance Discuss organic compounds and their biological importance. Summary Inorganic
Fiber. Total Digestible Fiber. Carbohydrate Fractions of Forages Fiber Fractions. 4/18/2014. Week 3 Lecture 9. Clair Thunes, PhD
 Carbohydrate Fractions of Forages Fiber Fractions. Week 3 Lecture 9 Clair Thunes, PhD Fiber Chemically fiber is a mix of cellulose, hemicellulose, pectins, lignin, indigestable protein and fats. Most measures
Carbohydrate Fractions of Forages Fiber Fractions. Week 3 Lecture 9 Clair Thunes, PhD Fiber Chemically fiber is a mix of cellulose, hemicellulose, pectins, lignin, indigestable protein and fats. Most measures
nanogreens 10 : Vegetable & Fruit SuperFood
 nanogreens 10 : Vegetable & Fruit SuperFood BioPharma Scientific Better taste ~ Better Science ~ nanogreens 10 New Great Tasting, Organic Super Food for the whole family! Importance of Diet Longevity is
nanogreens 10 : Vegetable & Fruit SuperFood BioPharma Scientific Better taste ~ Better Science ~ nanogreens 10 New Great Tasting, Organic Super Food for the whole family! Importance of Diet Longevity is
1 Preparation and Characterization of Lignin-Carbohydrate Complexes
 1 Preparation and Characterization of Lignin-Carbohydrate Complexes To explain the difficulty in separating lignin from carbohydrates in wood, Erdman (1866) hypothesized that the two combined chemically
1 Preparation and Characterization of Lignin-Carbohydrate Complexes To explain the difficulty in separating lignin from carbohydrates in wood, Erdman (1866) hypothesized that the two combined chemically
Biosynthesis of Secondary Metabolites
 Biosynthesis of Secondary Metabolites Secondary Metabolism Secondary metabolism, metabolic pathways that are not essential for growth, development or reproduction. Secondary metabolites are those chemical
Biosynthesis of Secondary Metabolites Secondary Metabolism Secondary metabolism, metabolic pathways that are not essential for growth, development or reproduction. Secondary metabolites are those chemical
Browning Reactions. Maillard browning. Caramelization high temps. Enzymatic browning. + flavors. brown pigments. + flavors.
 Browning Reactions Maillard browning reducing sugar + amine Caramelization sugar high temps Enzymatic browning phenolics polyphenoloxidase brown pigments + flavors brown pigments + flavors brown pigments
Browning Reactions Maillard browning reducing sugar + amine Caramelization sugar high temps Enzymatic browning phenolics polyphenoloxidase brown pigments + flavors brown pigments + flavors brown pigments
Class Agenda. Wood Chemistry. How are the hemicelluloses separated from cellulose? PSE 406/Chem E 470
 Class Agenda PSE 406/Chem E 470 Lecture 18 Chemical Isolation and Analysis II Hemicelluloses How are hemicelluloses separated from cellulose and lignin? How are individual hemicelluloses separated? How
Class Agenda PSE 406/Chem E 470 Lecture 18 Chemical Isolation and Analysis II Hemicelluloses How are hemicelluloses separated from cellulose and lignin? How are individual hemicelluloses separated? How
Soil organic matter composition, decomposition, mineralization and immobilization
 Soil organic matter composition, decomposition, mineralization and immobilization SOIL ORGANIC MATTER Substances containing carbon are organic matter. Soil organic matter consists of decomposing plant
Soil organic matter composition, decomposition, mineralization and immobilization SOIL ORGANIC MATTER Substances containing carbon are organic matter. Soil organic matter consists of decomposing plant
Determination of Tanninoids. Analytical Pharmacognosy
 QUIZ If the manager of a phytopharmaceutical industry wish to buy one chromatographic equipment, which one you recommend, HPLC or TLC densitometer. What are the reasons to support your recommendation.
QUIZ If the manager of a phytopharmaceutical industry wish to buy one chromatographic equipment, which one you recommend, HPLC or TLC densitometer. What are the reasons to support your recommendation.
ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES Carbohydrate Chemistry
 I. General structures A. D-Aldoses ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES 1. All monosaccharides are aldehydes or ketones with multiple hydroxyl groups (i.e., alcohol groups). 2. Smallest
I. General structures A. D-Aldoses ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES 1. All monosaccharides are aldehydes or ketones with multiple hydroxyl groups (i.e., alcohol groups). 2. Smallest
ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES
 I. General structures ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES A. D-Aldoses 1. All monosaccharides are aldehydes or ketones with multiple hydroxyl groups (i.e., alcohol groups). 2. Smallest
I. General structures ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES A. D-Aldoses 1. All monosaccharides are aldehydes or ketones with multiple hydroxyl groups (i.e., alcohol groups). 2. Smallest
Wood Saccharification: A Modified Rheinau Process
 http://douglasdrenkow.com/write2a.html Wood Saccharification: A Modified Rheinau Process After Chemistry of Wood by Erik Hägglund 1976 INTRODUCTION (UPDATED) The Rheinau (or Bergius) Process was one of
http://douglasdrenkow.com/write2a.html Wood Saccharification: A Modified Rheinau Process After Chemistry of Wood by Erik Hägglund 1976 INTRODUCTION (UPDATED) The Rheinau (or Bergius) Process was one of
Structural Polysaccharides
 Carbohydrates & ATP Carbohydrates include both sugars and polymers of sugars. The simplest carbohydrates are the monosaccharides, or simple sugars; these are the monomers from which more complex carbohydrates
Carbohydrates & ATP Carbohydrates include both sugars and polymers of sugars. The simplest carbohydrates are the monosaccharides, or simple sugars; these are the monomers from which more complex carbohydrates
Chromophores in wood and kraft pulp
 hromophores hromophores in wood and kraft pulp Greek chroma; colour and phoros; bearing hromophore is an organic structure which gives rise to colour Some chromophores: Lecture 9 Auxochrome (Greek auxanein,
hromophores hromophores in wood and kraft pulp Greek chroma; colour and phoros; bearing hromophore is an organic structure which gives rise to colour Some chromophores: Lecture 9 Auxochrome (Greek auxanein,
What are the most common elements in living organisms? What is the difference between monomers, dimers and polymers?
 What do each of these terms mean? Atom Molecule Element Compound Organic Inorganic What are the most common elements in living organisms? What are the roles of magnesium, iron, phosphate and calcium in
What do each of these terms mean? Atom Molecule Element Compound Organic Inorganic What are the most common elements in living organisms? What are the roles of magnesium, iron, phosphate and calcium in
Unit 3: Chemistry of Life Mr. Nagel Meade High School
 Unit 3: Chemistry of Life Mr. Nagel Meade High School IB Syllabus Statements 3.2.1 Distinguish between organic and inorganic compounds. 3.2.2 Identify amino acids, glucose, ribose and fatty acids from
Unit 3: Chemistry of Life Mr. Nagel Meade High School IB Syllabus Statements 3.2.1 Distinguish between organic and inorganic compounds. 3.2.2 Identify amino acids, glucose, ribose and fatty acids from
High value chemicals from spruce (Picea abies) extracts losses in the supply chain
 High value chemicals from spruce (Picea abies) extracts losses in the supply chain Eelis Halmemies, Ms.Sc Hanna Brännström, Ph.D. Kokkola Material Week, 1 22.11.2017 Routes to valuable bioactive extractives
High value chemicals from spruce (Picea abies) extracts losses in the supply chain Eelis Halmemies, Ms.Sc Hanna Brännström, Ph.D. Kokkola Material Week, 1 22.11.2017 Routes to valuable bioactive extractives
PRODUCTION OF VALUE ADDED PRODUCTS FROM SOME CEREAL MILLING BY-PRODUCTS SAYED SAAD ABOZAIED SMUDA THESIS DOCTOR OF PHILOSOPHY
 PRODUCTION OF VALUE ADDED PRODUCTS FROM SOME CEREAL MILLING BY-PRODUCTS By SAYED SAAD ABOZAIED SMUDA B.Sc. Agric. Sci. (Food Science), Fac. Agric., Cairo Univ., 2004 M.Sc. Agric. Sci. (Food Science), Fac.
PRODUCTION OF VALUE ADDED PRODUCTS FROM SOME CEREAL MILLING BY-PRODUCTS By SAYED SAAD ABOZAIED SMUDA B.Sc. Agric. Sci. (Food Science), Fac. Agric., Cairo Univ., 2004 M.Sc. Agric. Sci. (Food Science), Fac.
Carbohydrates. Organic compounds which comprise of only C, H and O. C x (H 2 O) y
 Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
Carbohydrates Organic compounds which comprise of only C, H and O C x (H 2 O) y Carbohydrates Monosaccharides Simple sugar Soluble in water Precursors in synthesis triose sugars of other (C3) molecules
The building blocks for this molecule are A) amino acids B) simple sugars C) fats D) molecular bases
 1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
Hot water extraction of inner and outer bark of Norway Spruce (Picea abies)
 Hot water extraction of inner and outer bark of Norway Spruce (Picea abies) Jens Krogell COST FP0901 meeting 21.08.2010 1 Spruce bark Inner bark (~90 vol-%) and outer bark (~10 vol-%) Huge valuable side
Hot water extraction of inner and outer bark of Norway Spruce (Picea abies) Jens Krogell COST FP0901 meeting 21.08.2010 1 Spruce bark Inner bark (~90 vol-%) and outer bark (~10 vol-%) Huge valuable side
1.3.1 Function of Food. Why do we need food?
 1.3.1 Function of Food Why do we need food? Need to know The Function of Food Three reasons for requiring food 2 Food is needed for: 1.Energy 2.Growth of new cells and Repair of existing cells, tissues,
1.3.1 Function of Food Why do we need food? Need to know The Function of Food Three reasons for requiring food 2 Food is needed for: 1.Energy 2.Growth of new cells and Repair of existing cells, tissues,
Effective Fractionation of Lignocellulose Using a Mild Acetone-based Organosolv Process
 Effective Fractionation of Lignocellulose Using a Mild Acetone-based Organosolv Process A.T. Smit W.J.J. Huijgen J.W. van Hal L. Lanting K.J. Damen June 2017 ECN-L--17-016 Presented @25th European Biomass
Effective Fractionation of Lignocellulose Using a Mild Acetone-based Organosolv Process A.T. Smit W.J.J. Huijgen J.W. van Hal L. Lanting K.J. Damen June 2017 ECN-L--17-016 Presented @25th European Biomass
Chapter 11: Lipids. Voet & Voet: Pages
 Chapter 11: Lipids Voet & Voet: Pages 380-394 Slide 1 Lipids Lipids are distinguished by their high solubility in non polar solvents and low solubility in H2O Diverse group of compounds including Fats,
Chapter 11: Lipids Voet & Voet: Pages 380-394 Slide 1 Lipids Lipids are distinguished by their high solubility in non polar solvents and low solubility in H2O Diverse group of compounds including Fats,
Carbohydrates, Lipids, Proteins, and Nucleic Acids
 Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
The Chemistry of Bleaching and Post-Color Formation in Kraft Pulps. Göran Gellerstedt
 The Chemistry of Bleaching and Post-Color Formation in Kraft Pulps Göran Gellerstedt Content The structure of residual lignin The meaning of kappa number General aspects on bleaching The oxygen stage The
The Chemistry of Bleaching and Post-Color Formation in Kraft Pulps Göran Gellerstedt Content The structure of residual lignin The meaning of kappa number General aspects on bleaching The oxygen stage The
Hetero polysaccharides
 Hetero polysaccharides Up to 1/3 rd of biomass is composed of hemicelluloses What is hemicellulose? riginally believed to be a precursor to cellulose, denoted by hemi Better referred to as hetero polysaccharide
Hetero polysaccharides Up to 1/3 rd of biomass is composed of hemicelluloses What is hemicellulose? riginally believed to be a precursor to cellulose, denoted by hemi Better referred to as hetero polysaccharide
CARBOHYDRATES. Created for BCLM Pony Club Nutrition #14
 CARBOHYDRATES Created for BCLM Pony Club Nutrition #14 Carbohydrates Collective term for starches, sugars and dietary fibres The dietary fibres are cellulose and hemicellulose Glycogen is how energy is
CARBOHYDRATES Created for BCLM Pony Club Nutrition #14 Carbohydrates Collective term for starches, sugars and dietary fibres The dietary fibres are cellulose and hemicellulose Glycogen is how energy is
THE RELATIONSHIP BETWEEN TWO METHODS FOR EVALUATING FIVE-CARBON SUGARS IN EUCALYPTUS EXTRACTION LIQUOR
 THE RELATIONSHIP BETWEEN TWO METHODS FOR EVALUATING FIVE-CARBON SUGARS IN EUCALYPTUS EXTRACTION LIQUOR Congcong Chi, a,b* Zeng Zhang, a Weiwei Ge, a and Hasan Jameel b Alkaline pre-extraction and hydrothermal
THE RELATIONSHIP BETWEEN TWO METHODS FOR EVALUATING FIVE-CARBON SUGARS IN EUCALYPTUS EXTRACTION LIQUOR Congcong Chi, a,b* Zeng Zhang, a Weiwei Ge, a and Hasan Jameel b Alkaline pre-extraction and hydrothermal
BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud
 BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud 1 Carbohydrates Carbohydrates: Compounds composed of carbon, oxygen, and hydrogen arranged as monosaccharides or multiples of monosaccharides. Most,
BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud 1 Carbohydrates Carbohydrates: Compounds composed of carbon, oxygen, and hydrogen arranged as monosaccharides or multiples of monosaccharides. Most,
Macro molecule = is all the reactions that take place in cells, the sum of all chemical reactions that occur within a living organism Anabolism:
 Macromolecule Macro molecule = molecule that is built up from smaller units The smaller single subunits that make up macromolecules are known as Joining two or more single units together form a M is all
Macromolecule Macro molecule = molecule that is built up from smaller units The smaller single subunits that make up macromolecules are known as Joining two or more single units together form a M is all
Topic 3.1 Nutrients. - Lipids are an essential part of the and are a part of cell in the body.
 Name: Topic 3.1 Nutrients Date: IB SEHS 3.1.1. List the macronutrients and micronutrients Macronutrients: - lipid (fat) - carbohydrate - protein - water (says the book) Micronutrients: - vitamins - minerals
Name: Topic 3.1 Nutrients Date: IB SEHS 3.1.1. List the macronutrients and micronutrients Macronutrients: - lipid (fat) - carbohydrate - protein - water (says the book) Micronutrients: - vitamins - minerals
Topic 3: Molecular Biology
 Topic 3: Molecular Biology 3.2 Carbohydrates and Lipids Essen=al Understanding: Carbon, hydrogen and oxygen are used to supply and store energy. Carbohydrates CARBOHYDRATES CHO sugars Primarily consist
Topic 3: Molecular Biology 3.2 Carbohydrates and Lipids Essen=al Understanding: Carbon, hydrogen and oxygen are used to supply and store energy. Carbohydrates CARBOHYDRATES CHO sugars Primarily consist
Chapter 1. Chemistry of Life - Advanced TABLE 1.2: title
 Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
Condensation and Hydrolysis Condensation reactions are the chemical processes by which large organic compounds are synthesized from their monomeric units. Hydrolysis reactions are the reverse process.
CHEMISTRY OF LIFE 30 JANUARY 2013
 CHEMISTRY OF LIFE 30 JANUARY 2013 Lesson Description In this lesson, we will: Investigate the structure and function of molecules that are essential for life. Key Concepts Terminology A molecule is any
CHEMISTRY OF LIFE 30 JANUARY 2013 Lesson Description In this lesson, we will: Investigate the structure and function of molecules that are essential for life. Key Concepts Terminology A molecule is any
Chapter Organic Chemistry. Functional Groups. Chapter The study of the compounds of carbon, not classified as inorganic.
 Chapter 22 rganic Compounds, Polymers & Biochemicals 1 22.1 rganic Chemistry The study of the compounds of carbon, not classified as inorganic Plastics, fibers, dues, drugs, insecticides, perfumes, petroleum
Chapter 22 rganic Compounds, Polymers & Biochemicals 1 22.1 rganic Chemistry The study of the compounds of carbon, not classified as inorganic Plastics, fibers, dues, drugs, insecticides, perfumes, petroleum
5.2 Lipids 5.21 Triglycerides 5.22 Phospholipids 5.23 Wax 5.24 Steroids. 5.3 Proteins 5.4 Nucleic Acids
 BIOCHEMISTRY Class Notes Summary Table of Contents 1.0 Inorganic and Organic Compounds 2.0 Monomers and Polymers 3.0 Dehydration (Condensation) Synthesis 4.0 Hydrolysis Reaction 5.0 Organic Compounds 5.1
BIOCHEMISTRY Class Notes Summary Table of Contents 1.0 Inorganic and Organic Compounds 2.0 Monomers and Polymers 3.0 Dehydration (Condensation) Synthesis 4.0 Hydrolysis Reaction 5.0 Organic Compounds 5.1
Chapter 11 Nutrition: Food for Thought
 Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
from Miscanthus Cellulose - Lignin
 Characterizing Lignocellulosics from Miscanthus Cellulose - Lignin Poulomi Sannigrahi A.J. Ragauskas School of Chemistry and Biochemistry Georgia Institute of Technology Agro-energy gy feedstock: Miscanthus
Characterizing Lignocellulosics from Miscanthus Cellulose - Lignin Poulomi Sannigrahi A.J. Ragauskas School of Chemistry and Biochemistry Georgia Institute of Technology Agro-energy gy feedstock: Miscanthus
REVISION: CHEMISTRY OF LIFE 19 MARCH 2014
 REVISION: CHEMISTRY OF LIFE 19 MARCH 2014 Lesson Description In this lesson we revise: The Chemistry of Life Food tests Summary Inorganic Nutrients Water Solvent Medium in which chemical reactions occur
REVISION: CHEMISTRY OF LIFE 19 MARCH 2014 Lesson Description In this lesson we revise: The Chemistry of Life Food tests Summary Inorganic Nutrients Water Solvent Medium in which chemical reactions occur
Chemistry 1050 Exam 3 Study Guide
 Chapter 12 Chemistry 1050 Exam 3 Study Guide 12.1 a) Identify alkenes, alkynes and aromatics as unsaturated hydrocarbons. Determine the number of hydrogen atoms needed to complete an alkene structure.
Chapter 12 Chemistry 1050 Exam 3 Study Guide 12.1 a) Identify alkenes, alkynes and aromatics as unsaturated hydrocarbons. Determine the number of hydrogen atoms needed to complete an alkene structure.
Biology. Slide 1 of 37. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 37 2 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. 3 of 37 Macromolecules Macromolecules Macromolecules
Biology 1 of 37 2 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. 3 of 37 Macromolecules Macromolecules Macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
A. Lipids: Water-Insoluble Molecules
 Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Biomolecule: Carbohydrate
 Biomolecule: Carbohydrate This biomolecule is composed of three basic elements (carbon, hydrogen, and oxygen) in a 1:2:1 ratio. The most basic carbohydrates are simple sugars, or monosaccharides. Simple
Biomolecule: Carbohydrate This biomolecule is composed of three basic elements (carbon, hydrogen, and oxygen) in a 1:2:1 ratio. The most basic carbohydrates are simple sugars, or monosaccharides. Simple
Cellulose - Hemicellulose
 Lecture in line with the course Biotechnology of trees and fungi Cellulose - Hemicellulose Dr. Christian Schöpper Phone: 0551-39 9745 mailto: cschoep@gwdg.de Major cell wall polysaccharides Celluloses
Lecture in line with the course Biotechnology of trees and fungi Cellulose - Hemicellulose Dr. Christian Schöpper Phone: 0551-39 9745 mailto: cschoep@gwdg.de Major cell wall polysaccharides Celluloses
A. There are about 100 elements; 25 of them are necessary for life. B. Carbon atoms can form long chains, leading to a huge number of possible
 Ch. 2 How Cells Function 2.1 Chemical reactions take place inside cells. 1. All cells are made of the same elements. A. There are about 100 elements; 25 of them are necessary for life. B. The smallest
Ch. 2 How Cells Function 2.1 Chemical reactions take place inside cells. 1. All cells are made of the same elements. A. There are about 100 elements; 25 of them are necessary for life. B. The smallest
Chemistry of Carbon. All living things rely on one particular type of molecule: carbon
 Ach Chemistry of Carbon All living things rely on one particular type of molecule: carbon Carbon atom with an outer shell of four electrons can form covalent bonds with four atoms. In organic molecules,
Ach Chemistry of Carbon All living things rely on one particular type of molecule: carbon Carbon atom with an outer shell of four electrons can form covalent bonds with four atoms. In organic molecules,
BY: RASAQ NURUDEEN OLAJIDE
 BY: RASAQ NURUDEEN OLAJIDE LECTURE CONTENT INTRODUCTION CLASSIFICATION OF LIPIDS PROPERTIES OF LIPIDS REACTIONS OF LIPIDS (CHEMICAL PROPERTIES) SOME QUANTITATIVE TESTS FOR LIPIDS CHEMISTRY AND PROPERTIES
BY: RASAQ NURUDEEN OLAJIDE LECTURE CONTENT INTRODUCTION CLASSIFICATION OF LIPIDS PROPERTIES OF LIPIDS REACTIONS OF LIPIDS (CHEMICAL PROPERTIES) SOME QUANTITATIVE TESTS FOR LIPIDS CHEMISTRY AND PROPERTIES
Products Detailed Tests/Clause No Standards/Tests Method Sample Preparation LABORATORY: CONSUMER PRODUCTS LABORATORY Beehoon
 Beehoon MS 955 : 2011 (Rice Vermicelli) 2. Total Ash 3. Total Protein 4. Crude Fibre 5. Cooking Test : Total Solid in Gruel Butter/Recombined Butter 1. Milk Fat MS 242 : 1988 8 X 250 g 3. Salt 4. Non-Fat
Beehoon MS 955 : 2011 (Rice Vermicelli) 2. Total Ash 3. Total Protein 4. Crude Fibre 5. Cooking Test : Total Solid in Gruel Butter/Recombined Butter 1. Milk Fat MS 242 : 1988 8 X 250 g 3. Salt 4. Non-Fat
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
The four stomachs of a dairy cow
 The four stomachs of a dairy cow Left side view 1) Rumen 2) Reticulum 3) Omasum 4) Abomasum Reticulo-omasal orifice (reticulo-rumen exit) (on the right side of the cow) (on the right side of the cow) Esophagus
The four stomachs of a dairy cow Left side view 1) Rumen 2) Reticulum 3) Omasum 4) Abomasum Reticulo-omasal orifice (reticulo-rumen exit) (on the right side of the cow) (on the right side of the cow) Esophagus
Outline. Sources, characteristics and common properties. Xylans. Mannans. Xyloglucans. Mixed-linkage β D-glucans
 FS630 Dr Nicolas Bordenave Room 3151 nbordena@purdue.edu Outline Sources, characteristics and common properties Xylans Mannans Xyloglucans Mixed-linkage β D-glucans Hemicellulose derivatives and their
FS630 Dr Nicolas Bordenave Room 3151 nbordena@purdue.edu Outline Sources, characteristics and common properties Xylans Mannans Xyloglucans Mixed-linkage β D-glucans Hemicellulose derivatives and their
Nature s Chemistry. Fragrances and Skin Care
 St Andrew s and St Bride s High School CfE Higher Chemistry Nature s Chemistry Page 1 of 12 Essential Oils Essential oils are concentrated extracts of the volatile, non-water soluble (hydrophobic) aroma
St Andrew s and St Bride s High School CfE Higher Chemistry Nature s Chemistry Page 1 of 12 Essential Oils Essential oils are concentrated extracts of the volatile, non-water soluble (hydrophobic) aroma
Dr. Nafith Abu Tarboush
 5 Dr. Nafith Abu Tarboush June 25 th 2013 Mohammad Abu Dosh Sheet 5.. Lipids ( Dr. Nafith ) : Classification of fatty acids : - they are classified depending on the existence of double bonds to : 1) Saturated
5 Dr. Nafith Abu Tarboush June 25 th 2013 Mohammad Abu Dosh Sheet 5.. Lipids ( Dr. Nafith ) : Classification of fatty acids : - they are classified depending on the existence of double bonds to : 1) Saturated
For example, monosaccharides such as glucose are polar and soluble in water, whereas lipids are nonpolar and insoluble in water.
 Biology 4A Laboratory Biologically Important Molecules Objectives Perform tests to detect the presence of carbohydrates, lipids, proteins, and nucleic acids Recognize the importance of a control in a biochemical
Biology 4A Laboratory Biologically Important Molecules Objectives Perform tests to detect the presence of carbohydrates, lipids, proteins, and nucleic acids Recognize the importance of a control in a biochemical
Lignin Isolation from Pulp
 Lignin Isolation from Pulp Several different enzymatic, chemical and mechanical methods have been developed for the isolation of lignin from wood and pulp. However, due to the heterogeneous nature of wood
Lignin Isolation from Pulp Several different enzymatic, chemical and mechanical methods have been developed for the isolation of lignin from wood and pulp. However, due to the heterogeneous nature of wood
What are the 6 Nutrients. Carbohydrates Proteins Fats/Oils (Lipids) Vitamins Minerals Water
 Nutrients AG 240 What are the 6 Nutrients Carbohydrates Proteins Fats/Oils (Lipids) Vitamins Minerals Water Carbohydrates (CHO) Primary component of livestock feed Referred to as energy CHO Characteristics
Nutrients AG 240 What are the 6 Nutrients Carbohydrates Proteins Fats/Oils (Lipids) Vitamins Minerals Water Carbohydrates (CHO) Primary component of livestock feed Referred to as energy CHO Characteristics
2. Compared to normal corn syrup, high fructose corn syrup is A) sweeter. B) less sweet. C) the same sweetness. D) higher in calories.
 CHEM 100 MODULE 10 PRETEST 1. Blood sugar is A) fructose. B) glucose. C) lactose. D) sucrose. 2. Compared to normal corn syrup, high fructose corn syrup is A) sweeter. B) less sweet. C) the same sweetness.
CHEM 100 MODULE 10 PRETEST 1. Blood sugar is A) fructose. B) glucose. C) lactose. D) sucrose. 2. Compared to normal corn syrup, high fructose corn syrup is A) sweeter. B) less sweet. C) the same sweetness.
Organic/Biochem Test #2 Takehome Name: Spring 2012 Page 1 of 6. Multiple choice: Circle the best answer for each of the following questions.
 Spring 2012 Page 1 of 6 Multiple choice: Circle the best answer for each of the following questions. 1. The general formula for a carboxylic acid is: a) RCHO b) RCOR c) R-OH d) R-COOH 2. In which of the
Spring 2012 Page 1 of 6 Multiple choice: Circle the best answer for each of the following questions. 1. The general formula for a carboxylic acid is: a) RCHO b) RCOR c) R-OH d) R-COOH 2. In which of the
Hydrolysis and Fractionation of Hot-Water Wood Extracts
 C Hydrolysis and Fractionation of Hot-Water Wood Extracts Thomas E. Amidon Christopher D. Wood, Jian Xu, Yang Wang, Mitchell Graves and Shijie Liu Biorefinery Research Institute Department of Paper and
C Hydrolysis and Fractionation of Hot-Water Wood Extracts Thomas E. Amidon Christopher D. Wood, Jian Xu, Yang Wang, Mitchell Graves and Shijie Liu Biorefinery Research Institute Department of Paper and
Photosynthesis Digestion Respiration. ., proteins. ... Glucose,.., fatty acids and glycerol, respectively.
 BIOMOLECULES Dear Reader In the previous chapter you have read about DNA present in the chromosomes. It is one of the many organic chemical compounds present in all living organisms. The organic compounds
BIOMOLECULES Dear Reader In the previous chapter you have read about DNA present in the chromosomes. It is one of the many organic chemical compounds present in all living organisms. The organic compounds
Elements & Macromolecules in Organisms
 Name: Period: Date: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
Name: Period: Date: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
In any solution, a scientist can talk about the concentration of the atoms that are dissolved in the solvent.
 Acids and Bases Acids and Bases In any solution, a scientist can talk about the concentration of the atoms that are dissolved in the solvent. i.e. Salt water is an example of Na + and Cl - in a solution
Acids and Bases Acids and Bases In any solution, a scientist can talk about the concentration of the atoms that are dissolved in the solvent. i.e. Salt water is an example of Na + and Cl - in a solution
Chapter 3 Guided Reading Notes Carbon and the Molecular Diversity of Life
 AP Biology Name: Block Chapter 3 Guided Reading Notes Carbon and the Molecular Diversity of Life Most of this chapter is new material. We will discuss it all in detail. Section 1 1. Make an electron distribution
AP Biology Name: Block Chapter 3 Guided Reading Notes Carbon and the Molecular Diversity of Life Most of this chapter is new material. We will discuss it all in detail. Section 1 1. Make an electron distribution
Carbon. Has four valence electrons Can bond with many elements. Can bond to other carbon atoms. Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
 Organic Compounds Carbon Has four valence electrons Can bond with many elements Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen Can bond to other carbon atoms Gives carbon the ability to form chains
Organic Compounds Carbon Has four valence electrons Can bond with many elements Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen Can bond to other carbon atoms Gives carbon the ability to form chains
Organic Chemistry. Organic chemistry is the chemistry of carbon compounds. Biochemistry is the study of carbon compounds that crawl.
 Organic Chemistry Organic chemistry is the chemistry of carbon compounds. Biochemistry is the study of carbon compounds that crawl. Organic Compounds - have carbon bonded to other atoms and determine structure/function
Organic Chemistry Organic chemistry is the chemistry of carbon compounds. Biochemistry is the study of carbon compounds that crawl. Organic Compounds - have carbon bonded to other atoms and determine structure/function
Macromolecules. The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1.
 Macromolecules The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1. CARBOHYDRATES 1. LIPIDS 1. NUCLEIC ACIDS Carbon Compounds All compounds
Macromolecules The four groups of biomolecules or macromolecules found in living things which are essential to life are: 1. PROTEINS 1. CARBOHYDRATES 1. LIPIDS 1. NUCLEIC ACIDS Carbon Compounds All compounds
Tannic Acid Production. Ensymm abstract for solvent-free production of Tannic Acid from fruit peels and organic waste
 Tannic Acid Production Ensymm abstract for solvent-free production of Tannic Acid from fruit peels and organic waste 1 INTRODUCTION- WHAT IS TANNIC ACID? INVERT Tannic SUGAR acid (also ABSTRACT known as
Tannic Acid Production Ensymm abstract for solvent-free production of Tannic Acid from fruit peels and organic waste 1 INTRODUCTION- WHAT IS TANNIC ACID? INVERT Tannic SUGAR acid (also ABSTRACT known as
B. Two basic types of chemical compounds: 1. Organic Compounds a. Based on a chain of 2 or more carbon atoms. b. Mainly the elements: C,H,N,O.
 NOTES: Chemistry of Life I. Chemical Compounds A. Substances formed from elements joined together. B. Two basic types of chemical compounds: 1. Organic Compounds a. Based on a chain of 2 or more carbon
NOTES: Chemistry of Life I. Chemical Compounds A. Substances formed from elements joined together. B. Two basic types of chemical compounds: 1. Organic Compounds a. Based on a chain of 2 or more carbon
Example. Biomentor Foundation. Advice Example
 Example Advice Example Biomentor Foundation URL WERK The normal values that are given, are our interpretation of all the results that we have seen in relation to (subject) judgements. So there is no absolute
Example Advice Example Biomentor Foundation URL WERK The normal values that are given, are our interpretation of all the results that we have seen in relation to (subject) judgements. So there is no absolute
Ch13. Sugars. What biology does with monosaccharides disaccharides and polysaccharides. version 1.0
 Ch13 Sugars What biology does with monosaccharides disaccharides and polysaccharides. version 1.0 Nick DeMello, PhD. 2007-2015 Ch13 Sugars Haworth Structures Saccharides can form rings. That creates a
Ch13 Sugars What biology does with monosaccharides disaccharides and polysaccharides. version 1.0 Nick DeMello, PhD. 2007-2015 Ch13 Sugars Haworth Structures Saccharides can form rings. That creates a
Carbon. p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms
 Organic Compounds Carbon p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms n Gives carbon the ability to form chains that are almost unlimited in length. p Organic
Organic Compounds Carbon p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms n Gives carbon the ability to form chains that are almost unlimited in length. p Organic
Do Now: Sort the following into the order of life from smallest to largest:
 Do Now: Sort the following into the order of life from smallest to largest: organ, molecule, atom, organelle, cell, organ system, tissue, organism Correct Order: atom, molecule, organelle, cell, tissue,
Do Now: Sort the following into the order of life from smallest to largest: organ, molecule, atom, organelle, cell, organ system, tissue, organism Correct Order: atom, molecule, organelle, cell, tissue,
INTEGRATED POSSIBILITIES OF PRODUCING CHEMICALS AND BIOFUELS IN CHEMICAL PULPING
 INTEGRATED POSSIBILITIES OF PRODUCING CHEMICALS AND BIOFUELS IN CHEMICAL PULPING International Forest Biorefining Conference (IFBC) May 9-11, 2017, Thunder Bay, ON, Canada Raimo Alén University of Jyväskylä,
INTEGRATED POSSIBILITIES OF PRODUCING CHEMICALS AND BIOFUELS IN CHEMICAL PULPING International Forest Biorefining Conference (IFBC) May 9-11, 2017, Thunder Bay, ON, Canada Raimo Alén University of Jyväskylä,
Elements & Macromolecules in Organisms
 Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be
Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds can be
Chapter. Phytochemicals: The Other Food Components. Images shutterstock.com
 14 Chapter Phytochemicals: The Other Food Components Images shutterstock.com Objectives List at least eight groups of phytochemicals and give a food source for each group. Identify possible links between
14 Chapter Phytochemicals: The Other Food Components Images shutterstock.com Objectives List at least eight groups of phytochemicals and give a food source for each group. Identify possible links between
UGRC 145: FOOD AND NUTRITION IN EVERYDAY LIFE
 UGRC 145: FOOD AND NUTRITION IN EVERYDAY LIFE Session 2 MACRONUTRIENTS Lecturer: PROF. MATILDA STEINER-ASIEDU, SBS, CBAS; University of Ghana, Email: tillysteiner@gmail.com College of Education School
UGRC 145: FOOD AND NUTRITION IN EVERYDAY LIFE Session 2 MACRONUTRIENTS Lecturer: PROF. MATILDA STEINER-ASIEDU, SBS, CBAS; University of Ghana, Email: tillysteiner@gmail.com College of Education School
2.3 Carbon Compounds 12/19/2011 BIOLOGY MRS. MICHAELSEN. Lesson Overview. Carbon Compounds The Chemistry of Carbon. Lesson Overview.
 2.3 The Chemistry of Carbon A. Carbon atoms have four valence electrons 1. Form strong covalent bonds with many other elements: H, O, P, S, N. 2. Living organisms are made up of carbon and these other
2.3 The Chemistry of Carbon A. Carbon atoms have four valence electrons 1. Form strong covalent bonds with many other elements: H, O, P, S, N. 2. Living organisms are made up of carbon and these other
Unit B: Seed Germination, Growth, and Development. Lesson 4: Determining Nutrient Functions and Utilization
 Unit B: Seed Germination, Growth, and Development Lesson 4: Determining Nutrient Functions and Utilization 1 Terms Denitrification Leach Macronutrient Micronutrient Nitrification Nitrogen cycle Nitrogen
Unit B: Seed Germination, Growth, and Development Lesson 4: Determining Nutrient Functions and Utilization 1 Terms Denitrification Leach Macronutrient Micronutrient Nitrification Nitrogen cycle Nitrogen
Name: Class: Honors Biology Period: Question: What is the molecular formula of this molecule?
 Chapter 3: The Chemistry of Organic Molecules Exercise 1 Diversity of Carbon-Based Molecules (3.1) The great variety of organic compounds results from the ability of carbon atoms to bond with four other
Chapter 3: The Chemistry of Organic Molecules Exercise 1 Diversity of Carbon-Based Molecules (3.1) The great variety of organic compounds results from the ability of carbon atoms to bond with four other
New Approaches for Improving Edible Oil Quality
 New Approaches for Improving Edible Oil Quality Selma Turkay and Hale Gurbuz Istanbul Technical University, Chemical Engineering Department OFI TURKEY 2014 13 May Istanbul Introduction In recent years,
New Approaches for Improving Edible Oil Quality Selma Turkay and Hale Gurbuz Istanbul Technical University, Chemical Engineering Department OFI TURKEY 2014 13 May Istanbul Introduction In recent years,
Biomolecules. Unit 3
 Biomolecules Unit 3 Atoms Elements Compounds Periodic Table What are biomolecules? Monomers vs Polymers Carbohydrates Lipids Proteins Nucleic Acids Minerals Vitamins Enzymes Triglycerides Chemical Reactions
Biomolecules Unit 3 Atoms Elements Compounds Periodic Table What are biomolecules? Monomers vs Polymers Carbohydrates Lipids Proteins Nucleic Acids Minerals Vitamins Enzymes Triglycerides Chemical Reactions
Lesson 3 Understanding Nutrients and Their Importance
 Unit B Understanding Animal Body Systems Lesson 3 Understanding Nutrients and Their Importance 1 Terms Balanced ration Carbohydrates Complex carbohydrates Disaccharides Essential nutrients Ether Fat Fat-soluble
Unit B Understanding Animal Body Systems Lesson 3 Understanding Nutrients and Their Importance 1 Terms Balanced ration Carbohydrates Complex carbohydrates Disaccharides Essential nutrients Ether Fat Fat-soluble
Soil Organic Matter. Unit 2. Forms of Soil Organic Matter: OM OC x (assumes 30% C) (1.72 typically used as a conversion factor)
 Unit 2 Soil Organic Matter OM OC x 1.7-2.0 (assumes 30% C) (1.72 typically used as a conversion factor) Histosol Alfisol Spodosol Forms of Soil Organic Matter: - dissolved (soil solution): DOM Nonliving
Unit 2 Soil Organic Matter OM OC x 1.7-2.0 (assumes 30% C) (1.72 typically used as a conversion factor) Histosol Alfisol Spodosol Forms of Soil Organic Matter: - dissolved (soil solution): DOM Nonliving
Chapter 11 Nutrition: Food for Thought
 Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
