African Journal of Pharmaceutical Research & Development
|
|
|
- David Walton
- 5 years ago
- Views:
Transcription
1 African Journal of Pharmaceutical Research & Development Vol. 5 No.1 pp.44-51(213) Studies on Microbiological Quality and Stability of Folic Acid Tablets Formulated with Tapioca Starch Kubili Mshelbwala 1, Kenneth Chibuzor Ofokansi 2, Franklin Chimaobi Kenechukwu 2 1 Department of Pharmaceutics and Pharmaceutical Microbiology, Faculty of Pharmaceutical Sciences, Almadu Bello University, Zaria, Kaduna State, Nigeria 2 Drug Delivery Research Unit, Department of Pharmaceutics, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka 411, Enugu State, Nigeria. ABSTRACT The purpose of this study was to assess the microbiological quality and stability of folic acid tablets formulated with tapioca starch as a binder in a comparative study with corn starch BP as the standard binder. This investigation was carried out by evaluating the microbiological quality of the starches, the granules and the folic acid tablets prepared by wet granulation, at specified periods of time under various temperature and humidity conditions. Results indicate that the imported maize starch B.P. had a level of microbial contamination which is comparable with the locally extracted cassava starch. The level of microbial contamination, which was higher when non-sterile distilled water was used as the diluting medium than when sterile distilled water was employed, was shown to be dependent on the both the temperature and relative humidity, with high humidity supporting microbial growth in the products. Folic acid tablets formulated with the two different starches exhibited lower level of microbial contaminants than the raw starches, indicating possible denaturation of the mainly vegetative organisms during tableting. Further bioevaluation is ongoing in our laboratory on the effect of compression during tableting on the level of microbial contamination. KEY WORDS: Tapioca starch, tablets, stability, folic acid, microbiological quality. INTRODUCTION In recent times, focus on plant research has increased all over the world and a lot of evidence has been collected to show immense potential of medicinal plants used as phyto-medicines for the treatment of disease [1] as well as traditional excipients for the manufacture of various dosage forms [2]. Cassava starch is obtained from the rasped tubers of Manihot utillisima, a tropical root tuber commonly used as food in Nigeria and other tropical countries. The plant is cultivated and grows to a height of about 2.4 m. The roots or tubers that contain the starch radiate from the stem just below the surface of the ground. The starch has been widely studied with respect to its suitability as an excipient in pharmaceutical dosage forms particularly in tableting technology [3 7]. The International Starch Institute has demonstrated for the first time on an industrial scale, that tapioca (cassava) starch can be produced with the same purity and yield as that of the potato starch industry [8]. Folic acid (also known as vitamin B9 or folacin) are forms of the water-soluble vitamins. Folic acid is itself not biologically active, but its biological importance is due to tetrahydrofolate and other derivatives after its conversion to dihydrofolic acid in the liver. Absorption of folic acid by the body is facilitated by enzymes associated with the mucosal cell membrane. More specifically, absorption primarily occurs in the mucosa of the upper intestine, known as the jejunum and duodenum. Insufficient folic acid in the diet and the inability to absorb folic acid can cause anemia or birth defects, namely, anecephaly and spina bifida, the latter *Author for correspondence; chimafrankduff@yahoo.com;frankline.kenechukwu@unn.edu.ng;tel:
2 Vol. 5, N 1pp , 213 resulting in brain development abnormalities [9]. Tablets are solid dosage forms containing medicinal substances with or without suitable diluents. Tablets are simple and convenient to use. They provide an accurately measured dosage of the active ingredient in a convenient portable package, and can be designed to achieve sustained release, protect unstable medications or disguise unpalatable ingredients [1, 11]. They may be classified according to the method of manufacture as either compressed tablets or molded tablets. The compressed tablets are the most widely used dosage form in the world and the vast majority of all tablets manufactured are made by compression. The compressed tablet is the most popular dosage form in use nowadays. About two-thirds of all prescriptions are dispensed as solid dosage forms, and half of these are compressed tablets [12]. Tablet dosage forms are the most popular and preferred drug delivery systems in terms of precision of unit dose, low cost, patient compliance, and good physical and chemical stability and account for 7-8 % of all pharmaceutical dosage forms [13]. Tablets have remained the most common dosage form by which medicaments are usually administered to patients because of their advantages over the other dosage forms [14]. It is always very important to assess the stability (short-term or long-term) of pharmaceutical formulations. Stability could be viewed from the degradation of the active ingredients or a change in the physicochemical properties of the formulations [15, 16]. Expectedly, stability test which involves examining for quality and potency at suitable time intervals, is ought to be conducted for a period corresponding to the normal time that the prodcut would remain in stock or in use. Since this test is time-consuming and expensive, accelerated stability tests are employed during the product development stage to enable rapid prediction of long-term stability of the product. It is then possible to identify the most stable formulation without resorting to the lenghty conventional stablity tests. No published work is available in the literature concerning the stability of folic acid tablets formulated with a native starch. Consequently, the objective of this study was to investigate the microbiological quality and stability of folic acid tablets formulated with tapioca starch as a binder in a comparative study with corn starch BP as the standard binder. This investigation was carried out by evaluating the microbiological quality of the starches, the granules and folic acid tablets prepared by wet granulation, at specified periods of time under various tempearture and humidity conditions. MATERIALS AND METHODS Materials The materials used include folic acid (Hoffman La Roche Ltd, Basel, Switzerland), Maize starch BP (BDH Chemicals Ltd, Pool, UK), Lactose BP (AB Knight and Co, London, UK). Tapioca starch was processed in the formulation laboratory of the Department of Pharmaceutics and Pharmaceutical Microbiology of Ahmadu Bello University, Zaria. Preparation and physicochemical properties of Cassava starch The tubers of cassava (Mannihot utilissima) were procured from the Institute for Agricultural Research, Ahmadu Bello University, Zaria.The Tapioca starch was prepared according to an established procedure [4] and as reported [7]. Preliminary physicochemical tests were then performed on the cassava and maize starches following standard procedures [3-6]. Preparation of starch mucilage Starch mucilage was prepared following a method already reported [5] by weighing amounts of starch powder that would produce 5 %w/w of the starch binders in the formulation. Each weighed quantity was then suspended in the required amount of distilled water in a beaker and heated with continuous stirring until the mucilage was formed. The mucilage was used while still hot for a more effective binding. Preparation of granules The formulation compositions of different batches of the granules prepared by wet granulation method [4] and tablets are presented in Tables 1 and 2. Briefly, accurately weighed quantities of folic acid and the starches were mixed together in an automatic mixer followed by addition of 5 % binder mucilage and further mixing.the resulting wet mass was transferred to a granulator fitted with an appropriate sieve. The granules formed were dried and sieved followed by addition of magnesium stearate and Talc powder, and mixing in a hand mixer. Granules size analysis The sieve method of analysis was employed [4-6]. Briefly, four sieves (5 µm, 355 µm, 25 µm, and 18 µm pore sizes) were arranged in order of decreasing size in a stack such that the sieve with the largest pore size was at the top. A 1 g of the 45
3 Vol. 5, N 1pp , 213 Batch code granules was poured into the topmost sieve and the Endecott sieve shaker was then switched on for 15 min. The amount of the granules left on each of sieve was weighed and the percentage of the granules on that sieve was calculated. Table 1: Formulation compositions of the tablets Folic acid Ingredient (mg) Starch Lactose Binder Talc Magnesium stearate I II III IV V Table 2: Composition of starch and lactose in the tablets (total weight = 6 mg) Batch Starch (%) Lactose (%) Folic acid (%) code I II III V V Tablet compression The granules were compressed into tablets on a single punch Manesty tabletting machine (Manesty, England) fitted with a circular 5.5 mm set of punches and die, according to an established method [3]. The compression pressure was maintained at 4.5 kg/sq.cm and the speed of compression was 7 tab/min. Bioevaluation Microbiological tests were carried out on the raw starches and the formulations (granules and tablets) using standard procedures [15, 16]. The pour plate method was employed and the suspending media were sterile distilled water and non-sterile distilled water. A 5 % starch suspension was thoroughly mixed on a Gallenkamp whirl mixer and 2 ml volume was withdrawn and serially diluted in sterile nutrient agar (N/A) for the bacteria and in Sabouraud dextrose agar (SDA) for the fungi. The plates were incubated at 37 C for 48 h for bacteria and at C for 96 h (for fungi). The plates were observed daily for presence or absence of growth. Viable counts were taken at the end of the incubation period using the Gallenkamp colony counter. Each experiment was replicated and the mean colony/spores taken and the colony forming units per ml (CFU/ml) or spore forming units per ml (SFU/ml) as the case may be were calculated. The entire procedure was repeated using the granules and the tablets. Identification and confirmatory tests for microorganisms Identification and confirmatory tests were carried out, using standard procedures [17], on the starches to detect the presence or absence of the following micro-organisms in the samples: Escherichia Coli, Salmonella spp. Staphylococcus aureus,and Pseudomonas aeruginosa for bacteria and Candida albicans, Aspergillus niger, Rhizopus mucor and Penicillium spp for fungi. Stability studies Time and relative humidity-dependent microbiological studies were also carried out on the raw starches and the formulations, following the ICH guidelines [18] and in accordance with previous study [12]. RESULTS AND DISCUSSION The tapioca starch passed all the preliminary tests (iodine and acidity tests) carried out to confirm the presence of starch [3]. There was no cyanide detected in the cassava starch sample. Cassava contains cyanide in different quantities [8]. The species of cassava cultivated in the northern parts of Nigeria could be eaten raw or cooked [19]. Most pharmaceutical industries have objected to the use of cassava starch due to the presence of cyanide [8]. There was no evidence of the presence of cyanide in the starch employed in the present study. The cyanide present in the species employed in this study could have been destroyed during the process of starch extraction, consistent with previous report [19]. The Tapioca starches were spherical with a particle density and size range of g/cm 3 and μm respectively. Their moisture content varied between 8.9 and 2. % and with poor flow properties. The steeping period is known to influence the physical characteristics of the starches [7]. Table 3 shows the moisture absorption and desorption properties of the local cassava starch and imported maize starch B.P. It is evident from the Table that the two starches absorbed moisture gradually and lost all the moisture almost at the same rate under the experimental conditions employed. Cassava starch absorbed larger amounts of moisture than maize starch B.P. at all levels of relative humidities employed. At the highest RH investigated i.e. 85 %, cassava starch 46
4 S.F.U x 1 5 /ml S.F.U x 1 5 /ml Vol. 5, N 1pp , 213 absorbed 12. % while the maize starch absorbed 11.1 % of moisture. The moisture absorbed is reversible as shown by the amounts of moisture desorbed from the starches (Table 3). While Figs. 1a-b depict the bacterial count which was carried out first in the months of August and September and second in the months of November and December in both sterile and non-sterile distilled water, respectively; Figs. 2a-d show the level of fungi in the starches. Generally, there were higher bacterial and fungal counts in the months of August and September than in the months of November and December. For bacterial count, in the first case in the two starches, there were more bacteria in the starches formulated in non-sterile water than in sterile distilled water. Comparing the bacterial count in the two starches prepared with sterile distilled water, the imported maize starch B.P. had higher bacterial count than the local cassava starach. In the second case using sterile distilled water, the bacterial count in the cassava starch was higher than that in the imported maize starch in both sterile and non-sterile distilled water. More so, the level of fungal contaminants was lower in the imported maize starch B.P. than in the local cassava starch. Table 3: Moisture absorption and desorption properties of the starches Relative Starch Parameter humidity (%) Maize Cassava Moisture absorption Moisture desorption Figure 1a: Level of bacteria count in the starches prepared using sterile distilled water. Figure 1b: Level of bacteria count in the starches Fungal isolates prepared using non-sterile distilled water. August and September November and December August and September November and December Fungal isolates 47
5 S.F.U x 1 5 /ml Vol. 5, N 1pp , 213 Figure 2a: Level of fungal SFU recorded in cassava starch prepared using sterile distilled water Figure 2b: Level of fungal SFU recorded in cassava starch prepared using non-sterile distilled water August and September November and December Fungal isolates Figure 2c: Level of fungal SFU recorded in maize starch prepared using sterile distilled water Comparing the bacteria count in the granules and tablets prepared with the starches, there was marked reduction in the bacterial count in the tablets prepared with maize starch B.P. The reverse was the case with granules and tablets prepared with cassava starch where the tablets recorded higher bacteria count than the granules. As illustrated in Fig. 4, at % R.H., the bacteria count in the two starches was between 1 x 1 4 to 2 x 1 4 CFU/ml. However, at 85 % R.H., the two bacteria count increased considerably to between 3 x 1 4 to 5 x 1 4 CFU/ml with tablets formulated with maize starch B.P. recording a higher number than tablets prepared with cassava starch. Fig. 5a-b show the level of fungal contaminants in the folic acid tablets stored under % and 85 % relative humidities for five weeks. The figure indicates that, at % R.H., the level of fungi was less in all the tablets formulated with the two different starches as compared to that at 85 % R.H. Tablets formulated with cassava starch, however, recorded a lower level of fungal contaminat than tablets prepared with maize starch B.P. at both % and 85 % R.H. Identification/confirmatory tests revealed the presence of bacteria (Staph aureus and E. coli) and fungi (Candida albicans, penicillium notatum, Asp. flavus, Asp. niger and Rhizopus mucor) in the cassava and maize starches. However, the maize starch B.P. had less level of contaminants than cassava starch in sterile media. In fact, in the months of August and September in sterile media, there were only two types of contaminants identified in the maize starch B.P., namely Asp. flavus and Candida albicans. Figure 2d: Level of fungal SFU recorded in maize starch prepared using non-sterile distilled water Fig. 3 shows the level of bacterial contaminants in the folic acid granules and tablets prepared with the starches. The test was carried out in sterile distilled water only. The granules prepared with the imported maize starch B. P. recorded higher number of bacterial count than the granules prepared with the local cassava starch. The effect of relative humidity on the level of bacterial contaminants is shown in Fig. 4. The level of bacterial contaminants in the tablets prepared with cassava starch was higher than in the tablets prepared with maize starch B. P. Fig. 3: Level of bacterial count recorded in cassava starch based folic acid granules and tablets. 48
6 Vol. 5, N 1pp ,
7 S.F.U x 1 3 /ml Candida albicans Aspergillius Rhizopus mucor Aspergillius Penicillium S.F.U x 1 7 /ml C.F.U. x 1 4 /ml Vol. 5, N 1pp , Cassava starch Maize starch Low relative humidity ( % RH) High relative humidity (85 % RH) Fig. 4: Viable count of bacteria recorded in cassava starch based folic acid tablets upon storage Fungal isolates Fungal isolates Low relative humidity ( % RH) High relative humidity (85 % RH) Fig. 5a: Level of fungal contaminants recorded in cassava starch based folic acid tablets upon storage Low relative humidity ( % RH) High relative humidity (85 % RH) Fig. 5b: Level of fungal contaminants recorded in maize starch based folic acid tablets upon storage. It is discernible from the results of the bioevaluation that the levels of both bacteria and fungi isolated were higher when non-sterile distilled water was used than when sterile distilled water was used. This was due to the killing of a lot of the bacteria during the sterilization process [17]. It was also observed that the level of contaminant was higher during the months of August and September than during the months of November and December. This was due to the high humidity plus the high temperature which provided a good breeding ground for the bacteria during the months of August and September as against the dry weather condition, low humidity and low temperature during the months of November and December; environmental conditions that are not conducive for bacterial growth [17]. Results equally indicate that, during the months of August and September, the imported maize starch B.P. recorded higher level of bacterial contaminant than the cassava starch. The reason for this is not certain but may be related to chanced contamination during tablet production [17]. Ideally, the imported maize starch ought to have recorded a lower amount of bacterial contaminants than the cassava starch. However, during the months of November and December, there was higher bacterial contamination in the cassava starch than in the imported maize starch B.P. In the case of fungi, maize starch B.P. recorded less number of fungi than the cassava starch. In the months of November and December, the two different starches recorded less number of SFU/ml of fungi possibly as a result of lower level of relative humidity and lower temperature which were not conducive for fungal growth. Therefore, the weather affected the growth of the fungi since they are all vegetative in nature. When the level of contaminants from the raw material, the granules and the tablets were compared, the starting raw material contained higher level of contaminats than the granules and the tablets contained the least level of contaminants in the case of bacteria. The only exception was the cassava starch-based tablet which contained a higher level of contaminants in the tablets than the granules, which may be attributed to chanced contamination during tableting. There might have been gradual destruction of the bacterial cells during the process of granulation and tableting [17]. The process of drying and tableting involved heat [3, 5, 9-12, 19] and since most of these microorganisms were vegetative they might have been destroyed in the process. The higher bacterial growth recorded under 85 % R.H. than the % RH is further proof that high humidity supports bacterial growth whereas low RH retards bacterial growth. 5
8 Vol. 5, N 1pp , 213 The contaminants isolated from the starch samples, granules and tablets call for the observation of current Good Manufacturing Practices (cgmp). If cgmp is not followed, it could lead to microbiological spoilage of pharmaceuticals [17, 2]. This condition is caused by the presence of specific types of pathogens in the pharmaceutical products. Bacterial contaminants of raw materials and the finished products have been known to lead to microbial spoilage, which is manifested when the presence of active pathogenic organism is high or when a higher level of opportunists is observed (when the level of opportunists are high, they can become pathogenic); when toxic microbial metabolites are present; when there is detectable physical and chemical changes which have occurred in the product [2]. cgmp requires that correct raw materials should be used and appropriate procedures adopted by adequately trained personnel working with suitable and necessary facilities. Moreover, pharmaceutical raw materials should be free from potential pathogens namely Salmonella, Escherichia coli and Pseudomonas aeruginosa [2]. Therefore, the detection and isolation of these pathogenic microorganisms from samples calls for extra care in the manufacturing process. The level of opportunistic organisms present under given suitable conditions could become pathogenic. On the whole, the results of this study indicate that cassava and maize starches contained both high level of opportunists and some active pathogens. Furthermore, the final products (tablets) had lower level of contaminants than the starting raw materials (starches), an implication that a lot of the microorganisms were destroyed during the processes of granulation and tableting. CONCLUSIONS This study has shown that the imported maize starch B.P. had a level of microbial contaminants which is comparable with the locally extracted cassava starch. The level of microbial contamination, which was higher when non-sterile distilled water was used as the diluting medium than when sterile distilled water was employed, was shown to be dependent on both the temperature and relative humidity, with high humidity supporting microbial growth in the products. Folic acid tablets formulated with the two different starches exhibited lower level of microbial contaminants than the raw starches, indicating possible denaturation of the mainly vegetative organisms during tableting. Further bioevaluation is ongoing in our laboratory on the effect of compression during tableting on the level of microbial contamination. REFERENCES 1. Dahanuka SA, Kulkarni RA, Rege NN. Pharmacology of medicinal plants and natural products, Indian J. Pharmacol., 22; 32: Kottke MK, Chueh HR, Rhodes CT. Comparison of disintegrant and binder activity of three corn starch products, Drug Dev. Ind. Pharm., 1992; 18: Okor RS, Obarisiagbon J. Evaluation of tapioca powder as a tablet base, Nig. J. Pharm., 1981; 12: Saringat B, Eseldin IK, Ching HS, The study of some mechanical properties of Tapioca starch, Acta Scientia, 1997; 7: Saringat B, Eseldin IK, Ching HS. Compaction behaviour of modified tapioca starch. Acta Scientia, 1997; 7: Eichie FE, Okor RS. Effect of acid treatment on the consolidation and plastoelasticity of tapioca powder, Trop. J. Pharm. Res., 22; 1: Alebiowu G. Steeping period influence on physical, compressional and mechanical properties of tapioca starch, J. Pharm. Res., 27; 6: International starch institute, (29). Tapioca (cassava) starch. Accessed on the 21/2/29 through 9. Gowtham M, Vasanti S, Rohan RD, Ashwath N, Paridav M. Formulation and evaluation of immediate release folic acid tablets, Der Pharmacia Lettre, 211; 3(6): Gohel MC, Bansal G, Bhatt N. Formulation and evaluation of orodispersible taste masked tablets of famotidine, Pharm. Bio. World, 25; 3: Patel R, Baria A. Formulation development and process optimization of theophylline sustained release matrix tablet, Int. J. Pharm., 29; 1: Vikramjit S, Nawazish A, Monika S, Alam MS, Ali MS, Md. Alam MI. Development, evaluation and stability studies of zidovudine and lamivudine tablet dosage form, J. App. Pharm Sci., 212; 2(9): Yüksel N, Türkmen B, Kurdoğlu AH, Başaran B, Erkin J, Baykara T. Lubricant efficiency of magnesium stearate in direct compressible powder mixtures comprising cellactose 8 and pyridoxine hydrochloride, FABAD J. Pharm. Sci., 27; 32:
9 Vol. 5, N 1pp , Okoye EI, Onyekweli1 AO, Olobayo OK, Arhewoh MI. Brittle fracture index (BFI) as a tool in the classification, grouping and ranking of some binders used in tablet formulation: Lactose tablets., Sci. Res. Essays, 21; 5 (5): Attama AA, Okafor CE, Builders PF, Ogbonna O. Formulation and in vitro evaluation of a PEGylated microscopic lipospheres delivery system of ceftriazole, Drug Deliv., 29; 16: Umeyor CE, Kenechukwu FC, Ogbonna JDN, Builders PF, Attama AA. Preliminary studies on the functional properties of gentamicin in SRMS-based solid lipid microparticles, Int. J. Novel Drug Deliv. Tech., 211; 1: Okore VC. Evaluation of chemical antimicrobial agents. In: Pharmaceutical Microbiology: Principles of the pharmaceutical applications of antimicrobial agents. Vincent C. Okore (ed.), 1 st edn., Enugu: EL DEMAK Publishers, Singh J, Tripathi KT, Sakia TR. Effect of penetration enhancers on the in vitro transport of ephedrine through rat skin and human epidermis from matrix based transdermal formulations, Drug Dev. Ind. Pharm., 23; 19: Isah AB, Abdulsamad A, Gwarzo MS and Abbah HM. Evaluation of the disintegrant properties of microcrystalline starch obtained from cassava in metronidazole tablet formulations, Nig. J. Pharm. Sci., 29; 8(2): Sharp J. Contamination and contamination control. In: Good pharmaceutical manufacturing practice: rationale and compliance. Sharp J (ed.), CRS Press: Boca Raton, Florida, USA; 25, pp
10 Vol. 3, pp , 211 Apeji, et al 48
THE PRESENCE OF MICROORGANISMS IN SOME COMMON EXCIPIENTS USED IN TABLET FORMULATION
 Acta Poloniae Pharmaceutica ñ Drug Research, Vol. 63 No. 2 pp. 121ñ125, 2006 ISSN 0001-6837 Polish Pharmaceutical Society PHARMACEUTICAL TECHNOLOGY THE PRESENCE OF MICROORGANISMS IN SOME COMMON EXCIPIENTS
Acta Poloniae Pharmaceutica ñ Drug Research, Vol. 63 No. 2 pp. 121ñ125, 2006 ISSN 0001-6837 Polish Pharmaceutical Society PHARMACEUTICAL TECHNOLOGY THE PRESENCE OF MICROORGANISMS IN SOME COMMON EXCIPIENTS
Distribution of low dose drugs in granules: Influence of method of incorporation
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Archives of Applied Science Research, 2011, 3 (3):241-245 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-508X
Available online at www.scholarsresearchlibrary.com Scholars Research Library Archives of Applied Science Research, 2011, 3 (3):241-245 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-508X
Formulation and Evaluation
 Chapter-5 Formulation and Evaluation 5.1 OBJECTIVE After successful taste masking and solubility enhancement of drugs in preliminary studies, by using Mannitol Solid Dispersion, next step includes the
Chapter-5 Formulation and Evaluation 5.1 OBJECTIVE After successful taste masking and solubility enhancement of drugs in preliminary studies, by using Mannitol Solid Dispersion, next step includes the
STABILITY STUDIES OF FORMULATED CONTROLLED RELEASE ACECLOFENAC TABLETS
 Int. J. Chem. Sci.: 8(1), 2010, 405-414 STABILITY STUDIES OF FORMULATED CONTROLLED RELEASE ACECLOFENAC TABLETS V. L. NARASAIAH, T. KARTHIK KUMAR, D. SRINIVAS, K. SOWMYA, P. L. PRAVALLIKA and Sk. Md. MOBEEN
Int. J. Chem. Sci.: 8(1), 2010, 405-414 STABILITY STUDIES OF FORMULATED CONTROLLED RELEASE ACECLOFENAC TABLETS V. L. NARASAIAH, T. KARTHIK KUMAR, D. SRINIVAS, K. SOWMYA, P. L. PRAVALLIKA and Sk. Md. MOBEEN
Compression and Mechanical Properties of Tablet Formulations
 Compression and Mechanical Properties of Tablet Formulations Containing Corn, Sweet, and Starches as Binders O.A. Odeku*, O.O. Awe, B. Popoola, M.A. Odeniyi, and O.A. Itiola The authors study the effects
Compression and Mechanical Properties of Tablet Formulations Containing Corn, Sweet, and Starches as Binders O.A. Odeku*, O.O. Awe, B. Popoola, M.A. Odeniyi, and O.A. Itiola The authors study the effects
Asian Journal of Pharmacy and Life Science ISSN Vol. 2 (2), July-Sept,2012
 STUDIES ON EFFECT OF SUPERDISINTEGRANTS ON ETORICOXIB TABLET FORMULATIONS Chowdary K. P. R 1, Venugopal. K *2 1 College of Pharmaceutical Sciences, Andhra University, Vishakapattanam. 2 * Nirmala college
STUDIES ON EFFECT OF SUPERDISINTEGRANTS ON ETORICOXIB TABLET FORMULATIONS Chowdary K. P. R 1, Venugopal. K *2 1 College of Pharmaceutical Sciences, Andhra University, Vishakapattanam. 2 * Nirmala college
Technical brochure StarLac
 T R TABLETING AC DIRECT COMPRESSION CO-PROCESSED LACTOSE Technical brochure MEGGLE co-processed lactose grades for direct compression: General information Direct compression (DC) tablet manufacture is
T R TABLETING AC DIRECT COMPRESSION CO-PROCESSED LACTOSE Technical brochure MEGGLE co-processed lactose grades for direct compression: General information Direct compression (DC) tablet manufacture is
African Journal of Pharmaceutical Research & Development
 [Type text] African Journal of Pharmaceutical Research & Development Vol. 7 No.2; pp.101-108 (2015) The Use of Multifunctional Starch Based Coprocessed Excipients (Starac) in the Formulation of Metronidazole
[Type text] African Journal of Pharmaceutical Research & Development Vol. 7 No.2; pp.101-108 (2015) The Use of Multifunctional Starch Based Coprocessed Excipients (Starac) in the Formulation of Metronidazole
Laboratorios CONDA, S.A. Distributed by Separations
 Culture Media as on Pharmacopoeia 7.3, Harmonized Method for Microbiological Examination of non sterile products -FORMULATIONS Buffered sodium chloride-peptone solution ph 7.0 Cat. Nº 1401 Potassium dihydrogen
Culture Media as on Pharmacopoeia 7.3, Harmonized Method for Microbiological Examination of non sterile products -FORMULATIONS Buffered sodium chloride-peptone solution ph 7.0 Cat. Nº 1401 Potassium dihydrogen
Packing and cohesive properties of some locally extracted starches
 Research Article Itiola and Odeku Tropical Journal of Pharmaceutical Research, June 2005; 4 (1): 363-368 Pharmacotherapy Group, Faculty of Pharmacy, University of Benin, Benin City, Nigeria. All rights
Research Article Itiola and Odeku Tropical Journal of Pharmaceutical Research, June 2005; 4 (1): 363-368 Pharmacotherapy Group, Faculty of Pharmacy, University of Benin, Benin City, Nigeria. All rights
STUDIES ON EFFECT OF BINDERS ON ETORICOXIB TABLET FORMULATIONS
 Int. J. Chem. Sci.: 10(4), 2012, 1934-1942 ISSN 0972-768X www.sadgurupublications.com STUDIES ON EFFECT OF BINDERS ON ETORICOXIB TABLET FORMULATIONS K. VENUGOPAL * and K. P. R. CHOWDARY a Nirmala College
Int. J. Chem. Sci.: 10(4), 2012, 1934-1942 ISSN 0972-768X www.sadgurupublications.com STUDIES ON EFFECT OF BINDERS ON ETORICOXIB TABLET FORMULATIONS K. VENUGOPAL * and K. P. R. CHOWDARY a Nirmala College
Available online through
 Research Article Available online through www.ijrap.net DESIGN AND EVALUATION OF LOW COST DIRECTLY COMPRESSIBLE EXCIPIENTS Swamy P. V. *, Patil A. N., Shirsand S. B., Amitkumar T., Laeeq Farhana H.K.E
Research Article Available online through www.ijrap.net DESIGN AND EVALUATION OF LOW COST DIRECTLY COMPRESSIBLE EXCIPIENTS Swamy P. V. *, Patil A. N., Shirsand S. B., Amitkumar T., Laeeq Farhana H.K.E
REVISION OF MONOGRAPH ON TABLETS. Tablets
 March 2011 REVISION OF MONOGRAPH ON TABLETS Final text for addition to The International Pharmacopoeia This monograph was adopted by the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
March 2011 REVISION OF MONOGRAPH ON TABLETS Final text for addition to The International Pharmacopoeia This monograph was adopted by the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
Int. Res J Pharm. App Sci., 2014; 4(1):47-51 ISSN:
 International Research Journal of Pharmaceutical and Applied Sciences (IRJPAS) Available online at www.irjpas.com Int. Res J Pharm. App Sci., 2014; 4(1):47-51 Research Article FORMULATION AND EVALUATION
International Research Journal of Pharmaceutical and Applied Sciences (IRJPAS) Available online at www.irjpas.com Int. Res J Pharm. App Sci., 2014; 4(1):47-51 Research Article FORMULATION AND EVALUATION
A Comparative Evaluation of Cross Linked Starch Urea-A New Polymer and Other Known Polymers for Controlled Release of Diclofenac
 Asian Journal of Chemistry Vol. 22, No. 6 (2010), 4239-4244 A Comparative Evaluation of Cross Linked Starch Urea-A New Polymer and Other Known Polymers for Controlled Release of Diclofenac K.P.R. CHOWDARY*
Asian Journal of Chemistry Vol. 22, No. 6 (2010), 4239-4244 A Comparative Evaluation of Cross Linked Starch Urea-A New Polymer and Other Known Polymers for Controlled Release of Diclofenac K.P.R. CHOWDARY*
Guidance on Pharmaceutical Excipient Suitability Studies (PESS) with Chinese Pharmacopeia (Volume 4): Basics and Examples
 Guidance on Pharmaceutical Excipient Suitability Studies (PESS) with Chinese Pharmacopeia (Volume 4): Basics and Examples Professor Jiasheng Tu, Ph. D. France 2018/09 content 1 Introduction 2 PESS guidance
Guidance on Pharmaceutical Excipient Suitability Studies (PESS) with Chinese Pharmacopeia (Volume 4): Basics and Examples Professor Jiasheng Tu, Ph. D. France 2018/09 content 1 Introduction 2 PESS guidance
ASEAN GUIDELINES ON LIMITS OF CONTAMINANTS FOR TRADITIONAL MEDICINES (TM) AND HEALTH SUPPLEMENTS (HS)
 Association of South East Asian Nations (ASEAN) ASEAN GUIDELINES ON LIMITS OF CONTAMINANTS FOR TRADITIONAL MEDICINES (TM) AND HEALTH SUPPLEMENTS (HS) Version 3.0 CONTENTS 1. Introduction...2 2. Objective...
Association of South East Asian Nations (ASEAN) ASEAN GUIDELINES ON LIMITS OF CONTAMINANTS FOR TRADITIONAL MEDICINES (TM) AND HEALTH SUPPLEMENTS (HS) Version 3.0 CONTENTS 1. Introduction...2 2. Objective...
Evaluation of Millet (Pennisetum Glaucum And Pennisetum Americanum) Starches as Tablet Binders
 Evaluation of Millet (Pennisetum Glaucum And Pennisetum Americanum) Starches as Tablet Binders *H. Musa,Y. Amodu,R.Oyi Department of Pharmaceutics and Pharmaceutical Microbiology, Faculty of Pharmaceutical
Evaluation of Millet (Pennisetum Glaucum And Pennisetum Americanum) Starches as Tablet Binders *H. Musa,Y. Amodu,R.Oyi Department of Pharmaceutics and Pharmaceutical Microbiology, Faculty of Pharmaceutical
National Exams May hours duration
 National Exams May 2012 04-Agric-A 7, Chemistry and Microbiology of Foods 3 hours duration NOTES: 1. If doubt exists as to the interpretation of any question, the candidate is urged to submit with the
National Exams May 2012 04-Agric-A 7, Chemistry and Microbiology of Foods 3 hours duration NOTES: 1. If doubt exists as to the interpretation of any question, the candidate is urged to submit with the
346 Full Text Available On
 International Journal of Universal Pharmacy and Bio Sciences 2(5): September-October 2013 INTERNATIONAL JOURNAL OF UNIVERSAL PHARMACY AND BIO SCIENCES IMPACT FACTOR 1.89*** Pharmaceutical Sciences ICV
International Journal of Universal Pharmacy and Bio Sciences 2(5): September-October 2013 INTERNATIONAL JOURNAL OF UNIVERSAL PHARMACY AND BIO SCIENCES IMPACT FACTOR 1.89*** Pharmaceutical Sciences ICV
FORMULATION AND EVALUATION OF PIROXICAM AND CELECOXIB TABLETS EMPLOYING PROSOLVE BY DIRECT COMPRESSION METHOD
 Int. J. Chem. Sci.: 6(3), 2008, 1270-1275 FORMULATION AND EVALUATION OF PIROXICAM AND CELECOXIB TABLETS EMPLOYING PROSOLVE BY DIRECT COMPRESSION METHOD K. P. R. CHOWDARY, P. TRIPURA SUNDARI and K. SURYA
Int. J. Chem. Sci.: 6(3), 2008, 1270-1275 FORMULATION AND EVALUATION OF PIROXICAM AND CELECOXIB TABLETS EMPLOYING PROSOLVE BY DIRECT COMPRESSION METHOD K. P. R. CHOWDARY, P. TRIPURA SUNDARI and K. SURYA
Microbiological Quality of Non-sterile Products Culture Media for Compendial Methods
 Microbiological Quality of Non-sterile Products Culture Media for Compendial Methods The life science business of Merck operates as MilliporeSigma in the U.S. and Canada. Culture Media for Compendial Methods
Microbiological Quality of Non-sterile Products Culture Media for Compendial Methods The life science business of Merck operates as MilliporeSigma in the U.S. and Canada. Culture Media for Compendial Methods
ANNEX III ASEAN GUIDELINES ON LIMITS OF CONTAMINANTS FOR TRADITIONAL MEDICINES
 Association of South East Asian Nations (ASEAN) ANNEX III ASEAN GUIDELINES ON LIMITS OF CONTAMINANTS FOR TRADITIONAL MEDICINES Disclaimer: This document is provided for information purpose only and subject
Association of South East Asian Nations (ASEAN) ANNEX III ASEAN GUIDELINES ON LIMITS OF CONTAMINANTS FOR TRADITIONAL MEDICINES Disclaimer: This document is provided for information purpose only and subject
Tablet is a major category of solid dosage forms which are widely used worldwide. Extensive information is required to prepare tablets with good
 TABLET PRODUCTİON Tablet is a major category of solid dosage forms which are widely used worldwide. Extensive information is required to prepare tablets with good quality at high standards. Based on preformulation
TABLET PRODUCTİON Tablet is a major category of solid dosage forms which are widely used worldwide. Extensive information is required to prepare tablets with good quality at high standards. Based on preformulation
Formulation and evaluation of sublingual tablets of lisinopril
 Journal of GROVER Scientific & Industrial AGARWAL: Research FORMULATION AND EVALUATION OF SUBLINGUAL TABLETS OF LISINOPRIL Vol. 71, June 2012, pp. 413-417 413 Formulation and evaluation of sublingual tablets
Journal of GROVER Scientific & Industrial AGARWAL: Research FORMULATION AND EVALUATION OF SUBLINGUAL TABLETS OF LISINOPRIL Vol. 71, June 2012, pp. 413-417 413 Formulation and evaluation of sublingual tablets
DESIGN AND EVALUATION OF CONTROLLED RELEASE MATRIX TABLETS OF FLURBIPROFEN
 Int. J. Chem. Sci.: 10(4), 2012, 2199-2208 ISSN 0972-768X www.sadgurupublications.com DESIGN AND EVALUATION OF CONTROLLED RELEASE MATRIX TABLETS OF FLURBIPROFEN K. V. R. N. S. RAMESH *, B. HEMA KIRNAMAYI
Int. J. Chem. Sci.: 10(4), 2012, 2199-2208 ISSN 0972-768X www.sadgurupublications.com DESIGN AND EVALUATION OF CONTROLLED RELEASE MATRIX TABLETS OF FLURBIPROFEN K. V. R. N. S. RAMESH *, B. HEMA KIRNAMAYI
Available online Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 26, 8(2):7-7 Research Article ISSN : 975-7384 CODEN(USA) : JCPRC5 Optimization of directly compressible mixtures of microcrystalline
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 26, 8(2):7-7 Research Article ISSN : 975-7384 CODEN(USA) : JCPRC5 Optimization of directly compressible mixtures of microcrystalline
Performance of starch obtained from Dioscorea dumetorium as disintegrant in sodium salicylate tablets
 African Journal of Pharmacy and Pharmacology Vol. 2(3). pp. 52-58, May, 8 Available online http://www.academicjournals.org/ajpp ISSN 1996-816 8 Academic Journals Full Length Research Paper Performance
African Journal of Pharmacy and Pharmacology Vol. 2(3). pp. 52-58, May, 8 Available online http://www.academicjournals.org/ajpp ISSN 1996-816 8 Academic Journals Full Length Research Paper Performance
STUDY OF THE DETERIORATION OF ASPIRIN IN THE PRESENCE OF VARIOUS EXCIPIENTS
 STUDY OF THE DETERIORATION OF ASPIRIN IN THE PRESENCE OF VARIOUS EXCIPIENTS D.L.D.A.N DAHANAYAKA, A. MUNISINGHE, D.T.U. ABEYTUNGA DEPARTMENT OF CHEMISTRY, UNIVERSITY OF COLOMBO Abstract Aspirin is a non
STUDY OF THE DETERIORATION OF ASPIRIN IN THE PRESENCE OF VARIOUS EXCIPIENTS D.L.D.A.N DAHANAYAKA, A. MUNISINGHE, D.T.U. ABEYTUNGA DEPARTMENT OF CHEMISTRY, UNIVERSITY OF COLOMBO Abstract Aspirin is a non
EVALUATION OF TABLETS BINDING PROPERTIES OF DIGITARIA IBURUA STARCH IN PARACETAMOL TABLETS FORMULATION
 International Journal of Current Pharmaceutical Research ISSN- 0975-7066 Vol 3, Issue 2, 2011 Research Article EVALUATION OF TABLETS BINDING PROPERTIES OF DIGITARIA IBURUA STARCH IN PARACETAMOL TABLETS
International Journal of Current Pharmaceutical Research ISSN- 0975-7066 Vol 3, Issue 2, 2011 Research Article EVALUATION OF TABLETS BINDING PROPERTIES OF DIGITARIA IBURUA STARCH IN PARACETAMOL TABLETS
Good pharmacopoeial practices: Chapter on monographs on herbal medicines
 Annex 7 Good pharmacopoeial practices: Chapter on monographs on herbal medicines Background Following the fiftieth meeting of the WHO Expert Committee on Specifications for Pharmaceutical Preparations,
Annex 7 Good pharmacopoeial practices: Chapter on monographs on herbal medicines Background Following the fiftieth meeting of the WHO Expert Committee on Specifications for Pharmaceutical Preparations,
Journal of Chemical and Pharmaceutical Research, 2012, 4(6): Research Article. Studies on Carica Papaya Starch as a Pharmaceutical Excipient
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2012, 4(6):3134-3138 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Studies on Carica Papaya Starch as a Pharmaceutical
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2012, 4(6):3134-3138 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Studies on Carica Papaya Starch as a Pharmaceutical
Studies of the Effect of Storage Conditions on Some Pharmaceutical Parameters of Powders and Tablets
 Studies of the Effect of Storage Conditions on Some Pharmaceutical Parameters of Powders and Tablets Bakre Lateef Gbenga and Yusuf Taiwo Department of Pharmaceutics and Pharmaceutical Technology, Faculty
Studies of the Effect of Storage Conditions on Some Pharmaceutical Parameters of Powders and Tablets Bakre Lateef Gbenga and Yusuf Taiwo Department of Pharmaceutics and Pharmaceutical Technology, Faculty
Formulation and evaluation of immediate release salbutamol sulphate
 5 Formulation, optimization and evaluation of immediate release layer of salbutamol sulphate Salbutamol is moderately selective beta (2)-receptor agonist similar in structure to terbutaline and widely
5 Formulation, optimization and evaluation of immediate release layer of salbutamol sulphate Salbutamol is moderately selective beta (2)-receptor agonist similar in structure to terbutaline and widely
International Journal of Scientific & Engineering Research, Volume 7, Issue 8, August ISSN
 International Journal of Scientific & Engineering Research, Volume 7, Issue 8, August-2016 105 Antimicrobial activity of Andrographis paniculata stem extracts. S.Gurupriya 1 and Dr.L.Cathrine 2 1 M.phil
International Journal of Scientific & Engineering Research, Volume 7, Issue 8, August-2016 105 Antimicrobial activity of Andrographis paniculata stem extracts. S.Gurupriya 1 and Dr.L.Cathrine 2 1 M.phil
FORMULATION AND DEVELOPMENT OF ER METOPROLAOL SUCCINATE TABLETS
 International Journal of PharmTech Research CODEN( USA): IJPRIF ISSN : 0974-4304 Vol.1, No.3, pp 634-638, July-Sept 2009 FORMULATION AND DEVELOPMENT OF ER METOPROLAOL SUCCINATE TABLETS K. Reeta Vijaya
International Journal of PharmTech Research CODEN( USA): IJPRIF ISSN : 0974-4304 Vol.1, No.3, pp 634-638, July-Sept 2009 FORMULATION AND DEVELOPMENT OF ER METOPROLAOL SUCCINATE TABLETS K. Reeta Vijaya
Formulation of alternative culture media for bacterial and fungal growth
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2016, 8 (1):431-436 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2016, 8 (1):431-436 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
HARMONISED PHARMACOPOEIA DEHYDRATED CULTURE MEDIA FOR SUPPORTING REGULATORY COMPLIANCE AVAILABLE NOW P O RTF O LIO.
 DEHYDRATED CULTURE MEDIA FOR ENHANCED P O RTF O LIO AVAILABLE NOW HARMONISED PHARMACOPOEIA SUPPORTING REGULATORY COMPLIANCE A Neogen Company THE GATEWAY TO MICROBIOLOGY INTRODUCTION Harmonised Pharmacopoeia;
DEHYDRATED CULTURE MEDIA FOR ENHANCED P O RTF O LIO AVAILABLE NOW HARMONISED PHARMACOPOEIA SUPPORTING REGULATORY COMPLIANCE A Neogen Company THE GATEWAY TO MICROBIOLOGY INTRODUCTION Harmonised Pharmacopoeia;
Stability of Captopril in an Extemporaneously Compounded Humco s Oral Products
 Stability of an Extemporaneously Compounded Humco s Oral Products Pradeep Gautam, MS* 1 1 Address correspondence to Pradeep Gautam, HUMCO Research and Development Laboratory, 201 W. 5 th St. Suite 1250,
Stability of an Extemporaneously Compounded Humco s Oral Products Pradeep Gautam, MS* 1 1 Address correspondence to Pradeep Gautam, HUMCO Research and Development Laboratory, 201 W. 5 th St. Suite 1250,
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(3):481-488 Some Pharmacopoeial and Diluent-Binder Properties
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(3):481-488 Some Pharmacopoeial and Diluent-Binder Properties
Changes in the Microflora of Bovine Colostrum During Natural Fermentation
 27 f. Milk Food Techno/. Vol. 39. No. I, Pages 27-31!January, 1976) Copyright 1976, International Association of Milk, Food, and Environmental Sanitarians Changes in the Microflora of Bovine Colostrum
27 f. Milk Food Techno/. Vol. 39. No. I, Pages 27-31!January, 1976) Copyright 1976, International Association of Milk, Food, and Environmental Sanitarians Changes in the Microflora of Bovine Colostrum
FLORITER. New Technology for Innovative Formulation Design.
 FLORITER New Technology for Innovative Formulation Design www.tomitaph.co.jp FLORITE Dramatically Change Your Formulation FLORITE is synthetic Calcium Silicate with exceptional liquid absorbency and excellent
FLORITER New Technology for Innovative Formulation Design www.tomitaph.co.jp FLORITE Dramatically Change Your Formulation FLORITE is synthetic Calcium Silicate with exceptional liquid absorbency and excellent
Direct Compression. With the right ingredients it s a simple, cost-effective manufacturing process
 Direct With the right ingredients it s a simple, cost-effective manufacturing process TM Trademark of The Dow Chemical Company ( Dow ) or an affiliated company of Dow Speed and savings sounds good to us
Direct With the right ingredients it s a simple, cost-effective manufacturing process TM Trademark of The Dow Chemical Company ( Dow ) or an affiliated company of Dow Speed and savings sounds good to us
OCE TABLETING DIRECT COMPRESSION CO-PROCESSED LACTOSE. Technical brochure MicroceLac 100
 IC OCE TABLETING DIRECT COMPRESSION CO-PROCESSED LACTOSE AC Technical brochure MEGGLE co-processed lactose grades for direct compression: General information Direct compression (DC) tablet manufacture
IC OCE TABLETING DIRECT COMPRESSION CO-PROCESSED LACTOSE AC Technical brochure MEGGLE co-processed lactose grades for direct compression: General information Direct compression (DC) tablet manufacture
Puducherry. Antimicrobial activity, Crude drug extraction, Zone of Inhibition, Culture Media, RVSPHF567.
 ANTI-MICROBIAL ACTIVITY OF THE CRUDE DRUGS AND THE POLYHERBAL FORMULATION (RVSPHF567) BY STANDARDIZED CUP AND PLATE METHOD C.S. Kandasamy 1,2*, Suman Nath 2, P. Arulraj 1,2, V. Gopal 3, P. Muthusamy 4,
ANTI-MICROBIAL ACTIVITY OF THE CRUDE DRUGS AND THE POLYHERBAL FORMULATION (RVSPHF567) BY STANDARDIZED CUP AND PLATE METHOD C.S. Kandasamy 1,2*, Suman Nath 2, P. Arulraj 1,2, V. Gopal 3, P. Muthusamy 4,
SENTRY TM POLYOX Water Soluble Resins
 SENTRY TM POLYOX Water Soluble Resins Technical Information on Stability Introduction Key Points Antioxidants SENTRY POLYOX WSR resins consist of a family of high molecular weight polyethers with nominal
SENTRY TM POLYOX Water Soluble Resins Technical Information on Stability Introduction Key Points Antioxidants SENTRY POLYOX WSR resins consist of a family of high molecular weight polyethers with nominal
FABRICATION AND EVALUATION OF GLIMEPIRIDE CORDIA DICHOTOMA G.FORST FRUIT MUCILAGE SUSTAINED RELEASE MATRIX TABLETS
 Int. J. Chem. Sci.: 7(4), 2009, 2555-2560 FABRICATION AND EVALUATION OF GLIMEPIRIDE CORDIA DICHOTOMA G.FORST FRUIT MUCILAGE SUSTAINED RELEASE MATRIX TABLETS HINDUSTAN ABDUL AHAD *, B. PRADEEP KUMAR, C.
Int. J. Chem. Sci.: 7(4), 2009, 2555-2560 FABRICATION AND EVALUATION OF GLIMEPIRIDE CORDIA DICHOTOMA G.FORST FRUIT MUCILAGE SUSTAINED RELEASE MATRIX TABLETS HINDUSTAN ABDUL AHAD *, B. PRADEEP KUMAR, C.
Re-compaction properties of lactose and microcrystalline cellulose
 Re-compaction properties of lactose and microcrystalline cellulose Individual excipients MCC Starch Lactose Inhalation Superdisintegrants SuperTab 21AN (anhydrous lactose) is the preferred form of lactose
Re-compaction properties of lactose and microcrystalline cellulose Individual excipients MCC Starch Lactose Inhalation Superdisintegrants SuperTab 21AN (anhydrous lactose) is the preferred form of lactose
Bioprospecting of Neem for Antimicrobial Activity against Soil Microbes
 ISSN: 2454-132X Impact factor: 4.295 (Volume3, Issue1) Available online at: www.ijariit.com Bioprospecting of Neem for Antimicrobial Activity against Soil Microbes R. Prasanna PRIST University, Tamilnadu
ISSN: 2454-132X Impact factor: 4.295 (Volume3, Issue1) Available online at: www.ijariit.com Bioprospecting of Neem for Antimicrobial Activity against Soil Microbes R. Prasanna PRIST University, Tamilnadu
Staphylococci. Gram stain: gram positive cocci arranged in clusters.
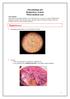 Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
STARCH Proven and Trusted Excipient for Performance and Versatility EXCIPIENTS. Effective and economical disintegrant
 EXCIPIENTS STARCH 1500 Proven and Trusted Excipient for Performance and Versatility Effective and economical disintegrant Excellent stability for moisture sensitive drugs Manufactured exclusively for the
EXCIPIENTS STARCH 1500 Proven and Trusted Excipient for Performance and Versatility Effective and economical disintegrant Excellent stability for moisture sensitive drugs Manufactured exclusively for the
Formulation and evaluation of oro-dispersible tablets of lafutidine
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2015, 7 (5):226-235 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Available online at www.scholarsresearchlibrary.com Scholars Research Library Der Pharmacia Lettre, 2015, 7 (5):226-235 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-5071 USA CODEN: DPLEB4
Formulation and Evaluation of Gastroretentive Dosage form of Ciprofloxacin Hydrochloride.
 Available online on www.ijcpr.com International Journal of Current Pharmaceutical Review and Research, 3(4), 105-109 Research Article ISSN: 0976-822X Formulation and Evaluation of Gastroretentive Dosage
Available online on www.ijcpr.com International Journal of Current Pharmaceutical Review and Research, 3(4), 105-109 Research Article ISSN: 0976-822X Formulation and Evaluation of Gastroretentive Dosage
Volume: 2: Issue-3: July-Sept ISSN FORMULATION AND EVALUATION OF SUSTAINED RELEASE MATRIX TABLETS OF NICORANDIL
 Volume: 2: Issue-3: July-Sept -2011 ISSN 0976-4550 FORMULATION AND EVALUATION OF SUSTAINED RELEASE MATRIX TABLETS OF NICORANDIL Ajaykumar Patil*, Ashish Pohane, Ramya Darbar, Sharanya Koutika, Alekhya
Volume: 2: Issue-3: July-Sept -2011 ISSN 0976-4550 FORMULATION AND EVALUATION OF SUSTAINED RELEASE MATRIX TABLETS OF NICORANDIL Ajaykumar Patil*, Ashish Pohane, Ramya Darbar, Sharanya Koutika, Alekhya
Evaluation of Terminalia randii Baker F. gum as a disintegrant in paracetamol tablet formulation
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2014, 6(10):155-159 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Evaluation of Terminalia randii Baker F. gum as
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2014, 6(10):155-159 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Evaluation of Terminalia randii Baker F. gum as
EVALUATON OF BARLEY HORDEUM VULGARE STARCH AS TABLET DISINTEGRANT
 International Journal of Pharmacy and Pharmaceutical Sciences ISSN- 975-1491 Vol 3 Suppl 3, 211 Research Article EVALUATON OF BARLEY HORDEUM VULGARE STARCH AS TABLET DISINTEGRANT *H.MUSA, S.N. OCHU, P.G.BHATIA
International Journal of Pharmacy and Pharmaceutical Sciences ISSN- 975-1491 Vol 3 Suppl 3, 211 Research Article EVALUATON OF BARLEY HORDEUM VULGARE STARCH AS TABLET DISINTEGRANT *H.MUSA, S.N. OCHU, P.G.BHATIA
Summary and Conclusion
 Summary and Conclusion Milk and milk products are consumed daily by Indian citizens. This type of food is easily perishable and support growth of foodborne pathogens keeping this view in mind the investigation
Summary and Conclusion Milk and milk products are consumed daily by Indian citizens. This type of food is easily perishable and support growth of foodborne pathogens keeping this view in mind the investigation
Preservative A2 Plus. 1. INCI Composition. 2. Overview. 3. Specification data. Diazolidinyl Urea 99.0% Iodopropynyl Butylcarbamate 1.
 Preservative A2 Plus Diazolidinyl Urea 99.0% Iodopropynyl Butylcarbamate 1.0% 1. ICI Composition 2. verview Preservative A2 Plus is a broad spectrum cosmetic preservative in powder. It is a synergistic
Preservative A2 Plus Diazolidinyl Urea 99.0% Iodopropynyl Butylcarbamate 1.0% 1. ICI Composition 2. verview Preservative A2 Plus is a broad spectrum cosmetic preservative in powder. It is a synergistic
Application of Starches, Modified Starches and Starch Derivatives in Pharmaceutical Products
 57. Starch Convention, Detmold, April 26-28, 2006 K.-J. Steffens Application of Starches, Modified Starches and Starch Derivatives in Pharmaceutical Products Starches, Pharmaceutical Applications _ Starches
57. Starch Convention, Detmold, April 26-28, 2006 K.-J. Steffens Application of Starches, Modified Starches and Starch Derivatives in Pharmaceutical Products Starches, Pharmaceutical Applications _ Starches
LubriTose Mannitol Michael Crowley, Director of R&D, Excipients
 LubriTose Mannitol Michael Crowley, Director of R&D, Excipients Introduction Michael Crowley Director of R&D Excipients 158 St. Highway 320 Norwich, NY 13815 PH 315-802-5970 Michael.Crowley@Kerry.com 2
LubriTose Mannitol Michael Crowley, Director of R&D, Excipients Introduction Michael Crowley Director of R&D Excipients 158 St. Highway 320 Norwich, NY 13815 PH 315-802-5970 Michael.Crowley@Kerry.com 2
Biopharmaceutics Dosage form factors influencing bioavailability Lec:5
 Biopharmaceutics Dosage form factors influencing bioavailability Lec:5 Ali Y Ali BSc Pharmacy MSc Industrial Pharmaceutical Sciences Dept. of Pharmaceutics School of Pharmacy University of Sulaimani 09/01/2019
Biopharmaceutics Dosage form factors influencing bioavailability Lec:5 Ali Y Ali BSc Pharmacy MSc Industrial Pharmaceutical Sciences Dept. of Pharmaceutics School of Pharmacy University of Sulaimani 09/01/2019
Microbial quality of some herbal solid dosage forms
 African Journal of Biotechnology Vol. 9 (11), pp. 1701-1705, 15 March, 2010 Available online at http://www.academicjournals.org/ajb ISSN 1684 5315 2010 Academic Journals Full Length Research Paper Microbial
African Journal of Biotechnology Vol. 9 (11), pp. 1701-1705, 15 March, 2010 Available online at http://www.academicjournals.org/ajb ISSN 1684 5315 2010 Academic Journals Full Length Research Paper Microbial
Higher plants produced hundreds to thousands of diverse chemical compounds with different biological activities (Hamburger and Hostettmann, 1991).
 4. ANTIMICROBIAL ACTIVITY OF PHYSALIS MINIMA L. 4.1. Introduction Use of herbal medicines in Asia represents a long history of human interactions with the environment. Plants used for traditional medicine
4. ANTIMICROBIAL ACTIVITY OF PHYSALIS MINIMA L. 4.1. Introduction Use of herbal medicines in Asia represents a long history of human interactions with the environment. Plants used for traditional medicine
J. Muazu et al / Journal of Pharmaceutical Science and Technology Vol. 4 (3), 2012,
 Preliminary studies on Hausa potato starch I: The disintegrant properties J. Muazu*, A Girbo, A Usman and G. T. Mohammed Department of Pharmaceutics and Pharmaceutical Microbiology, University of Maiduguri,
Preliminary studies on Hausa potato starch I: The disintegrant properties J. Muazu*, A Girbo, A Usman and G. T. Mohammed Department of Pharmaceutics and Pharmaceutical Microbiology, University of Maiduguri,
7. SUMMARY, CONCLUSION AND RECOMMENDATIONS
 211 7. SUMMARY, CONCLUSION AND RECOMMENDATIONS Drug absorption from the gastro intestinal tract can be limited by various factors with the most common one being poor aqueous solubility and poor permeability
211 7. SUMMARY, CONCLUSION AND RECOMMENDATIONS Drug absorption from the gastro intestinal tract can be limited by various factors with the most common one being poor aqueous solubility and poor permeability
Evaluation of native and modified starch dispersions in mucilage obtained from sweet potato in ascorbic acid chewable tablet formulations
 World Journal of Pharmaceutical Sciences ISSN (Print): 2321-3310; ISSN (Online): 2321-3086 Published by Atom and Cell Publishers All Rights Reserved Available online at: http://www.wjpsonline.org/ Original
World Journal of Pharmaceutical Sciences ISSN (Print): 2321-3310; ISSN (Online): 2321-3086 Published by Atom and Cell Publishers All Rights Reserved Available online at: http://www.wjpsonline.org/ Original
Pharmaceutical Preparation For Internal Use
 Pharmaceutical Preparation For Internal Use 1. Solid Preparations (Tablet, Capsule, Pill) 2. Liquid Preparations (Aqua, Syrup, Elixir, Extract, Liquor, Emulsion, Mixture, Infusion, Decoction). 3. Powder
Pharmaceutical Preparation For Internal Use 1. Solid Preparations (Tablet, Capsule, Pill) 2. Liquid Preparations (Aqua, Syrup, Elixir, Extract, Liquor, Emulsion, Mixture, Infusion, Decoction). 3. Powder
Studies on Curcuma angustifolia Starch as a Pharmaceutical Excipient
 International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.2, No.4, pp 2456-2460, Oct-Dec 2010 Studies on Curcuma angustifolia Starch as a Pharmaceutical Excipient P.RAJEEVKUMAR
International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.2, No.4, pp 2456-2460, Oct-Dec 2010 Studies on Curcuma angustifolia Starch as a Pharmaceutical Excipient P.RAJEEVKUMAR
DETERMINATION OF MICROBIAL CONTAMINANTS IN SOME MARKETED HERBAL FORMULATION
 DETERMINATION OF MICROBIAL CONTAMINANTS IN SOME MARKETED HERBAL FORMULATION INTRODUCTION: Herbal medicines are in great demand in the developed as well as developing countries for primary health care because
DETERMINATION OF MICROBIAL CONTAMINANTS IN SOME MARKETED HERBAL FORMULATION INTRODUCTION: Herbal medicines are in great demand in the developed as well as developing countries for primary health care because
PRESENTER: DENNIS NYACHAE MOSE KENYATTA UNIVERSITY
 18/8/2016 SOURCES OF MICROBIAL CONTAMINANTS IN BIOSAFETY LABORATORIES IN KENYA PRESENTER: DENNIS NYACHAE MOSE KENYATTA UNIVERSITY 1 INTRODUCTION Contamination occurs through avoidable procedural errors
18/8/2016 SOURCES OF MICROBIAL CONTAMINANTS IN BIOSAFETY LABORATORIES IN KENYA PRESENTER: DENNIS NYACHAE MOSE KENYATTA UNIVERSITY 1 INTRODUCTION Contamination occurs through avoidable procedural errors
MEDICNES CONTROL AGENCY GUIDELINE FOR REGISTRATION OF HERBAL MEDICINAL PRODUCTS IN THE GAMBIA
 MEDICNES CONTROL AGENCY GUIDELINE FOR REGISTRATION OF HERBAL MEDICINAL PRODUCTS IN THE GAMBIA Document No.: MCA/HMPG/17/12/01 Date of Adoption: 20 th January 2017 Date of Issue: 25 th July 2017 Version
MEDICNES CONTROL AGENCY GUIDELINE FOR REGISTRATION OF HERBAL MEDICINAL PRODUCTS IN THE GAMBIA Document No.: MCA/HMPG/17/12/01 Date of Adoption: 20 th January 2017 Date of Issue: 25 th July 2017 Version
Formulation Development of Aceclofenac Tablets Employing Starch Phosphate -A New Modified Starch
 Abstract K.P.R. Chowdary et al. / International Journal of Pharma Sciences and Research (IJPSR) Formulation Development of Aceclofenac Tablets Employing Starch Phosphate -A New Modified Starch K.P.R. Chowdary*,
Abstract K.P.R. Chowdary et al. / International Journal of Pharma Sciences and Research (IJPSR) Formulation Development of Aceclofenac Tablets Employing Starch Phosphate -A New Modified Starch K.P.R. Chowdary*,
International Journal of Pharma and Bio Sciences. Preparation and Evaluation of Aegle marmelos Gum as Tablet Binder
 Patil D.N 1*, Kulkarni A.R 2, Hatapakki B.C 1 and Patil B.S 1 1 Department of Pharmacognosy & Phytochemistry, KLES s College of Pharmacy, Gadag-582101, Karnataka (India). 2 Department of Pharmacology,
Patil D.N 1*, Kulkarni A.R 2, Hatapakki B.C 1 and Patil B.S 1 1 Department of Pharmacognosy & Phytochemistry, KLES s College of Pharmacy, Gadag-582101, Karnataka (India). 2 Department of Pharmacology,
Effect of Compaction Forces on Powder Bed Permeability of Magnesium Silicate "Common Excipient Mixture"
 Effect of Compaction Forces on Powder Bed Permeability of Magnesium Silicate "Common Excipient Mixture" SAMEER AL-ASHEH a, FAWZI BANAT a, ALA A SALEM a, IAD RASHID b, ADNAN BADWAN b a Department of Chemical
Effect of Compaction Forces on Powder Bed Permeability of Magnesium Silicate "Common Excipient Mixture" SAMEER AL-ASHEH a, FAWZI BANAT a, ALA A SALEM a, IAD RASHID b, ADNAN BADWAN b a Department of Chemical
Use of different Additives in Retting Cassava tubers for Fufu production
 International Journal of Pharmaceutical Science Invention ISSN (Online): 2319 6718, ISSN (Print): 2319 670X Volume 3 Issue 4 April 2014 PP.11-17 Use of different Additives in Retting Cassava tubers for
International Journal of Pharmaceutical Science Invention ISSN (Online): 2319 6718, ISSN (Print): 2319 670X Volume 3 Issue 4 April 2014 PP.11-17 Use of different Additives in Retting Cassava tubers for
DEVELOPMENT OF NON SODIUM EFFERVESCENT TABLET OF PARACETAMOL USING ARGININE CARBONATE
 IJPSR (2013), Vol. 4, Issue 5 (Research Article) Received on 17 July, 2012; received in revised form, 23 February, 2013; accepted, 14 April, 2013 DEVELOPMENT OF NON SODIUM EFFERVESCENT TABLET OF PARACETAMOL
IJPSR (2013), Vol. 4, Issue 5 (Research Article) Received on 17 July, 2012; received in revised form, 23 February, 2013; accepted, 14 April, 2013 DEVELOPMENT OF NON SODIUM EFFERVESCENT TABLET OF PARACETAMOL
USP Chewable Gels Monographs
 USP Dietary Supplements Stakeholder Forum Tuesday, May 15, 2018 USP Chewable Gels Monographs Natalia Davydova, Ph.D. Scientific Liaison DS Gummies Market Value of the gummy vitamins market in the United
USP Dietary Supplements Stakeholder Forum Tuesday, May 15, 2018 USP Chewable Gels Monographs Natalia Davydova, Ph.D. Scientific Liaison DS Gummies Market Value of the gummy vitamins market in the United
Vimta Labs Ltd., Life Sciences Facility, Plot No. 5, Alexandria Knowledge Park, Genome Valley, Shameerpet, Hyderabad, Telangana
 Last Amended on - Page 1 of 14 I. DRUGS & PHARMACEUTICALS 1. Biological Assays Antibiotics And Other Drugs Bulk Drugs & Their Formulations: Erythromycin, Gentamicin, Nystatin IP Appendix 9.1 2.2.10 BP
Last Amended on - Page 1 of 14 I. DRUGS & PHARMACEUTICALS 1. Biological Assays Antibiotics And Other Drugs Bulk Drugs & Their Formulations: Erythromycin, Gentamicin, Nystatin IP Appendix 9.1 2.2.10 BP
DRY SYRUPS SWAPNA.M. Ist semester DEPARTMENT OF PHARMACEUTICS UNIVERSITY COLLEGE OF PHARMACEUTICAL SCIENCES KAKATIYA UNIVERSITY, WARANGAL SEMINAR BY
 DRY SYRUPS SEMINAR BY SWAPNA.M M.PHARMACY Ist semester DEPARTMENT OF PHARMACEUTICS UNIVERSITY COLLEGE OF PHARMACEUTICAL SCIENCES KAKATIYA UNIVERSITY, WARANGAL CONTENTS DEFINITION CHARACTERISTICS OF SUSPENSIONS
DRY SYRUPS SEMINAR BY SWAPNA.M M.PHARMACY Ist semester DEPARTMENT OF PHARMACEUTICS UNIVERSITY COLLEGE OF PHARMACEUTICAL SCIENCES KAKATIYA UNIVERSITY, WARANGAL CONTENTS DEFINITION CHARACTERISTICS OF SUSPENSIONS
THE EFFECT OF SOME MICROORGANISMS IN GASTRO-INTESTINAL TRACTS ON THE NUTRITIVE VALUE OF BROILER DIETS
 THE EFFECT OF SOME MICROORGANISMS IN GASTRO-INTESTINAL TRACTS ON THE NUTRITIVE VALUE OF BROILER DIETS Settasit SANGSOPONJIT 1, Wichai SUPHALUCKSANA 1 1 Faculty of Agricultural Technology, King Mongkut
THE EFFECT OF SOME MICROORGANISMS IN GASTRO-INTESTINAL TRACTS ON THE NUTRITIVE VALUE OF BROILER DIETS Settasit SANGSOPONJIT 1, Wichai SUPHALUCKSANA 1 1 Faculty of Agricultural Technology, King Mongkut
NOVASTREAK. Microbial Contamination Monitoring Device TYPICAL CULTURAL MORPHOLOGY Baird Parker Agar. S. aureus growth on Baird Parker Agar
 NOVASTREAK Microbial Contamination Monitoring Device TYPICAL CULTURAL MORPHOLOGY Baird Parker Agar S. aureus growth on Baird Parker Agar Baird Parker Agar is used for the selective isolation and enumeration
NOVASTREAK Microbial Contamination Monitoring Device TYPICAL CULTURAL MORPHOLOGY Baird Parker Agar S. aureus growth on Baird Parker Agar Baird Parker Agar is used for the selective isolation and enumeration
Formulation and Development of Sustained Release Tablets of Valsartan Sodium
 INTERNATIONAL JOURNAL OF ADVANCES IN PHARMACY, BIOLOGY AND CHEMISTRY Research Article Formulation and Development of Sustained Release Tablets of Valsartan Sodium G. Sandeep * and A. Navya Department of
INTERNATIONAL JOURNAL OF ADVANCES IN PHARMACY, BIOLOGY AND CHEMISTRY Research Article Formulation and Development of Sustained Release Tablets of Valsartan Sodium G. Sandeep * and A. Navya Department of
Technical brochure CombiLac
 OM I AC TABLETING DIRECT COMPRESSION CO-PROCESSED LACTOSE Technical brochure MEGGLE co-processed lactose grades for direct compression: General information Direct compression (DC) tablet manufacture is
OM I AC TABLETING DIRECT COMPRESSION CO-PROCESSED LACTOSE Technical brochure MEGGLE co-processed lactose grades for direct compression: General information Direct compression (DC) tablet manufacture is
Available Online through Research Article
 ISSN: 0975-766X Available Online through Research Article www.ijptonline.com DESIGN AND EVALUATION OF GASTRORETENTIVE TABLETS FOR CONTROLLED DELIVERY OF NORFLOXOCIN Ganesh Kumar Gudas*, Subal Debnath,
ISSN: 0975-766X Available Online through Research Article www.ijptonline.com DESIGN AND EVALUATION OF GASTRORETENTIVE TABLETS FOR CONTROLLED DELIVERY OF NORFLOXOCIN Ganesh Kumar Gudas*, Subal Debnath,
This PDF is available for free download from a site hosted by Medknow Publications
 26 CMYK Research Paper Possible Use of Psyllium Husk as a Release Retardant ANGIRA DESAI, SUPRIYA SHIDHAYE*, V. J. KADAM Department of Pharmaceutics, Bharati Vidhyapeeth s College of Pharmacy, Sector-8,
26 CMYK Research Paper Possible Use of Psyllium Husk as a Release Retardant ANGIRA DESAI, SUPRIYA SHIDHAYE*, V. J. KADAM Department of Pharmaceutics, Bharati Vidhyapeeth s College of Pharmacy, Sector-8,
J.Ayyappan et al, /J. Pharm. Sci. & Res. Vol.2 (7), 2010,
 Development and Evaluation of a Directly Compressible Co-processed Multifunction Sustained Release Agent for Gliclazide Sustained Release Tablets J. Ayyappan* 1, P.Umapathi 1, Darlin Quine 2 1 Department
Development and Evaluation of a Directly Compressible Co-processed Multifunction Sustained Release Agent for Gliclazide Sustained Release Tablets J. Ayyappan* 1, P.Umapathi 1, Darlin Quine 2 1 Department
Survival of Aerobic and Anaerobic Bacteria in
 APPLIED MICROBIOLOGY, Mar. 1968, p. 445-449 Copyright 1968 American Society for Microbiology Vol. 16, No. 3 Printed in U.S.A. Survival of Aerobic and Anaerobic Bacteria in Chicken Meat During Freeze-Dehydration,
APPLIED MICROBIOLOGY, Mar. 1968, p. 445-449 Copyright 1968 American Society for Microbiology Vol. 16, No. 3 Printed in U.S.A. Survival of Aerobic and Anaerobic Bacteria in Chicken Meat During Freeze-Dehydration,
FACTORS AFFECTING THE GROWTH OF MICRO-ORGANISMS IN FOODS
 FACTORS AFFECTING THE GROWTH OF MICRO-ORGANISMS IN FOODS FACTORS AFFECTING THE GROWH OF MICRO-ORGANISMS IN FOODS Several factors related to the environment and the conditions in which food is stored influence
FACTORS AFFECTING THE GROWTH OF MICRO-ORGANISMS IN FOODS FACTORS AFFECTING THE GROWH OF MICRO-ORGANISMS IN FOODS Several factors related to the environment and the conditions in which food is stored influence
Maisammaguda, Dulapally, Secundrabad.
 121 P a g e International Standard Serial Number (ISSN): 2319-8141 International Journal of Universal Pharmacy and Bio Sciences 3(6): November-December 2014 INTERNATIONAL JOURNAL OF UNIVERSAL PHARMACY
121 P a g e International Standard Serial Number (ISSN): 2319-8141 International Journal of Universal Pharmacy and Bio Sciences 3(6): November-December 2014 INTERNATIONAL JOURNAL OF UNIVERSAL PHARMACY
A FACTORIAL STUDY ON THE ENHANCEMENT OF DISSOLUTION RATE OF KETOPROFEN BY SOLID DISPERSION IN COMBINED CARRIERS
 Research Article A FACTORIAL STUDY ON THE ENHANCEMENT OF DISSOLUTION RATE OF KETOPROFEN BY SOLID DISPERSION IN COMBINED CARRIERS K. P. R. Chowdary *, Tanniru Adinarayana, T. Vijay, Mercy. R. Prabhakhar
Research Article A FACTORIAL STUDY ON THE ENHANCEMENT OF DISSOLUTION RATE OF KETOPROFEN BY SOLID DISPERSION IN COMBINED CARRIERS K. P. R. Chowdary *, Tanniru Adinarayana, T. Vijay, Mercy. R. Prabhakhar
Pelagia Research Library
 Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2011, 2 (1): 201-207 ISSN: 0976-8688 CODEN (USA): PSHIBD Isolation of Cordia mucilage and its comparative evaluation as a binding
Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2011, 2 (1): 201-207 ISSN: 0976-8688 CODEN (USA): PSHIBD Isolation of Cordia mucilage and its comparative evaluation as a binding
Antimicrobial activity of Karuveppilai vadagam against Enteric pathogens
 International Journal of Current Research in Medical Sciences ISSN: 2454-5716 P-ISJN: A4372-3064, E -ISJN: A4372-3061 www.ijcrims.com Original Research Article Volume 4, Issue 6-2018 Antimicrobial activity
International Journal of Current Research in Medical Sciences ISSN: 2454-5716 P-ISJN: A4372-3064, E -ISJN: A4372-3061 www.ijcrims.com Original Research Article Volume 4, Issue 6-2018 Antimicrobial activity
Int. Res J Pharm. App Sci., 2012; 2(6): ISSN:
 International Research Journal of Pharmaceutical and Applied Sciences (IRJPAS) Available online at www.irjpas.com Int. Res J Pharm. App Sci., 2013; 3(1): 269-273 Research Article FORMULATION DEVELOPMENT
International Research Journal of Pharmaceutical and Applied Sciences (IRJPAS) Available online at www.irjpas.com Int. Res J Pharm. App Sci., 2013; 3(1): 269-273 Research Article FORMULATION DEVELOPMENT
LAB.2. Tablet Production Methods
 LAB.2 Tablet Production Methods Dry methods Direct compression Dry granulation Wet methods Wet granulation Regardless whether tablets are made by direct compression or granulation, the first step, milling
LAB.2 Tablet Production Methods Dry methods Direct compression Dry granulation Wet methods Wet granulation Regardless whether tablets are made by direct compression or granulation, the first step, milling
In vitro study of antibacterial activity of Carissa carandas leaf extracts
 Available online at www.pelagiaresearchlibrary.com Asian Journal of Plant Science and Research, 2012, 2 (1):36-40 ISSN : 2249-7412 CODEN (USA): AJPSKY In vitro study of antibacterial activity of Carissa
Available online at www.pelagiaresearchlibrary.com Asian Journal of Plant Science and Research, 2012, 2 (1):36-40 ISSN : 2249-7412 CODEN (USA): AJPSKY In vitro study of antibacterial activity of Carissa
Formulation and In-vitro Evaluation of Chewable Tablets of Montelukast Sodium
 Available online on www.ijddt.com International Journal of Drug Delivery Technology 214; (3); 98-13 Research Article ISSN: 97 441 Formulation and In-vitro Evaluation of Chewable Tablets of Montelukast
Available online on www.ijddt.com International Journal of Drug Delivery Technology 214; (3); 98-13 Research Article ISSN: 97 441 Formulation and In-vitro Evaluation of Chewable Tablets of Montelukast
Designed and manufactured specifically for pharmaceutical capsule filling
 EXCIPIENTS Designed and manufactured specifically for pharmaceutical capsule filling Simple formulation Superior flow and weight uniformity Clean and efficient processing This document is valid at the
EXCIPIENTS Designed and manufactured specifically for pharmaceutical capsule filling Simple formulation Superior flow and weight uniformity Clean and efficient processing This document is valid at the
Microbial load and prevalence of pathogens on surface of fresh vegetables in local market yards across Junagadh district of Gujarat
 International Journal of Plant Protection Volume 5 Issue 1 April, 2012 84-88 Research Article IJPP Microbial load and prevalence of pathogens on surface of fresh vegetables in local market yards across
International Journal of Plant Protection Volume 5 Issue 1 April, 2012 84-88 Research Article IJPP Microbial load and prevalence of pathogens on surface of fresh vegetables in local market yards across
Evaluation of Rubber Seed Oil as Foundry Sand-Core Binder in Castings
 The International Journal Of Engineering And Science (IJES) Volume 3 Issue 8 Pages 20-28 2014 ISSN (e): 2319 1813 ISSN (p): 2319 1805 Evaluation of Rubber Seed Oil as Foundry Sand-Core Binder in Castings
The International Journal Of Engineering And Science (IJES) Volume 3 Issue 8 Pages 20-28 2014 ISSN (e): 2319 1813 ISSN (p): 2319 1805 Evaluation of Rubber Seed Oil as Foundry Sand-Core Binder in Castings
FORMULATION, SENSORY EVALUATION AND NUTRIENT ANALYSIS OF PRODUCTS WITH ALOE VERA
 WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Joy K et al. Volume 2, Issue 6, 5321-5328. Research Article ISSN 2278 4357 FORMULATION, SENSORY EVALUATION AND NUTRIENT ANALYSIS OF PRODUCTS WITH ALOE
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Joy K et al. Volume 2, Issue 6, 5321-5328. Research Article ISSN 2278 4357 FORMULATION, SENSORY EVALUATION AND NUTRIENT ANALYSIS OF PRODUCTS WITH ALOE
