Contrasting Roles of Basolateral Amygdala and Orbitofrontal Cortex in Impulsive Choice
|
|
|
- Bethanie Woods
- 5 years ago
- Views:
Transcription
1 4718 The Journal of Neuroscience, May 19, (20): Brief Communication Contrasting Roles of Basolateral Amygdala and Orbitofrontal Cortex in Impulsive Choice Catharine A. Winstanley, David E. H. Theobald, Rudolf N. Cardinal, and Trevor W. Robbins Department of Experimental Psychology, University of Cambridge, Downing Site, Cambridge CB2 3EB, United Kingdom The orbitofrontal cortex (OFC) and basolateral nucleus of the amygdala (BLA) share many reciprocal connections, and a functional interaction between these regions is important in controlling goal-directed behavior. However, their relative roles have proved hard to dissociate. Although injury to these brain regions can cause similar effects, it has been suggested that the resulting impairments arise through damage to different, yet converging, cognitive processes. Patients with OFC or amygdala lesions exhibit maladaptive decision making and aberrant social behavior often described as impulsive. Impulsive choice may be measured in both humans and rodents by evaluating intolerance to delay of reinforcement. Rats with excitotoxic lesions of the BLA and OFC were tested on such a delay-discounting procedure. Although lesions of the BLA increased choice of the small immediate reward, indicating greater impulsivity, OFC lesions had the opposite effect, increasing preference for the larger but delayed reward. The fact that the delay did not devalue the large reward to such an extent in OFC-lesioned animals supports the suggestion that the OFC is involved in updating the incentive value of outcomes in response to devaluation. In contrast, the BLA-lesioned animals markedly decreased their preference for the large reward when it was delayed, potentially because of an inability to maintain a representation of the reward in its absence. This is the first time that lesions to these two structures have produced opposite behavioral effects, indicating their distinct contributions to cognition. Key words: amygdala; cognitive; frontal; impulsivity; goal directed; delay discounting Introduction The orbitofrontal cortex (OFC), encompassing ventrolateral and ventromedial frontal cortex, is integral to various corticocortical networks implicated in higher-order cognition, receiving polymodal input from sensory cortices as well as interacting with subcortical areas including the mediodorsal thalamus (Nauta, 1971; Öngür and Price, 2000). In particular, the OFC shares many reciprocal connections with the basolateral nucleus of the amygdala (BLA) (Krettek and Price, 1977; McDonald, 1991), one of the larger amygdaloid nuclei, which interconnects with temporal lobe structures such as the hippocampus as well as cortical and striatal regions (Pitkänen, 2000). OFC and BLA lesions often cause similar behavioral effects (Schoenbaum et al., 2003a), a pattern attributed to the extensive connections linking these regions. Dissociating the roles these structures play in goal-directed behavior has therefore proved difficult. The OFC and BLA are important nodes in the limbic corticostriatal loop, linking them to other structures involved in reward learning, including the nucleus accumbens (NAC) (Cardinal et Received Dec. 19, 2003; revised April 7, 2004; accepted April 11, This work was supported by a Wellcome Trust Programme grant and completed within the Medical Research Council (MRC) Centre for Behavioral and Clinical Neuroscience. C.A.W. was supported by an MRC studentship. We are grateful to Yogita Chudasama and Jonathan Lee regarding histological verification of the lesions. Correspondence should be addressed to Catharine Winstanley, Department of Psychiatry, University of Texas Southwestern Medical Center, Dallas, TX cath.winstanley@utsouthwestern.edu. DOI: /JNEUROSCI Copyright 2004 Society for Neuroscience /04/ $15.00/0 al., 2002). NAC lesions increase impulsivity in rats on a delay-togratification task, in which impulsive choice is defined as the selection of a small immediate over a larger delayed reward (Ainslie, 1975; Logue, 1988; Evenden and Ryan, 1996; Cardinal et al., 2001). Impulsivity is a core deficit in neuropsychiatric illnesses, such as attention deficit hyperactivity disorder, and rodent delayto-gratification tasks have been used to investigate the pharmacology of impulsive behavior (Evenden and Ryan, 1996). However, structures conveying information to the NAC necessary for appropriate response selection are unknown. Lesions of prelimbic (PrL) or anterior cingulate (ACC) cortex cause little or no change in impulsive choice (Cardinal et al., 2001) despite strong interconnections with the NAC. Unlike patients with damage to dorsolateral prefrontal cortex (PFC), patients with OFC and amygdala lesions exhibit patterns of behavior often described as impulsive as exemplified by their performance on gambling tasks (Damasio, 1994; Bechara et al., 1998, 1999; Rogers et al., 1999). Although healthy volunteers select options leading to long-term gain, patients with OFC or amygdala lesions make risky choices, preferring options associated with large short-term rewards but long-term loss. However, the behavior of OFC patients can suggest paradoxically reduced impulsivity. Their risky decision making can be very slow, contradicting the impulsive trait of making over-hasty judgements (Damasio, 1994; Rogers et al., 1999). Although patients with OFC damage generate appropriate autonomic responses to affective stimuli and to losing money in the gambling paradigm,
2 Winstanley et al. Roles of BLA and OFC in Impulsive Choice J. Neurosci., May 19, (20): patients with amygdala lesions do not (Bechara et al., 1999). The amygdala plays an important role in processing emotionally significant stimuli (LeDoux, 2000), which can guide goal-directed behavior (Gallagher, 2000). For example, BLA lesions profoundly impair fear conditioning to a discrete conditioned stimulus (CS), yet less is known about the role of the OFC in such processes. However, lesions to both structures disrupt the utilization of CSs in goal seeking (Gallagher, 2000). We therefore postulated that BLA and OFC lesions would alter behavior in a rodent model of impulsive choice, and that this paradigm could reveal dissociable deficits after damage to these areas. Materials and Methods Subjects. Subjects were 32 male Lister Hooded rats (Charles River, Kent, UK) weighing gm at the start of each experiment and maintained at 85% of their free-feeding weight. Water was available ad libitum. Animals were housed in groups of four under a reverse light cycle (lights on from 7:00 P.M. to 7:00 A.M.). Testing took place between 8:00 A.M. and 7:00 P.M. 5 6 d per week. Experiments were undertaken in accordance with the UK Animals (Scientific Procedures) Act Behavioral testing. The behavioral apparatus and testing procedure have been described previously in detail (Cardinal et al., 2000). Testing occurred in eight operant conditioning chambers (Med Associates, Georgia, VT) fitted with two retractable levers located on either side of a food magazine, into which 45 mg of food pellets (Noyes dustless pellets; Sandown Scientific, Middlesex, UK) could be delivered, a magazine light, and a houselight. Magazine entry was detected by an infrared photocell beam located across the entrance. Each chamber was encased in a soundattenuating box and fan ventilated. The apparatus was controlled by software written in Arachnid running on Acorn Archimedes Series computers (Cambridge, UK). Subjects first learned to lever-press for food reward and to nose-poke in the magazine to trigger presentation of the levers. Animals were then trained to perform a delay-discounting task until stable baseline behavior was achieved (Fig. 1). Each session lasted 100 min and consisted of 5 blocks of 12 trials. Each trial lasted 100 sec regardless of the choice made by the subject, and each block began with two forced-choice trials. The onset of the houselight signaled the beginning of each trial, in which the rat had to nose-poke in the magazine to trigger lever presentation. An omission was scored if the rat failed to respond at the food magazine or subsequently on the levers within 10 sec, and the box returned to the inter-trial interval (ITI) state with the houselight extinguished until the next trial was scheduled to begin. The houselight was extinguished and the levers retracted once a response was made. Responding on one lever (lever A) always provided a small immediate reward of one pellet, the other (lever B) a large reward of four pellets. The location of levers A and B were counterbalanced between subjects. As the session progressed, the delay to the large reward increased in each block of trials from 0 to 10, 20, 40, and then 60 sec. Onset of the traylight signaled food delivery, after which the box returned to the ITI state. Surgery. Subjects were divided into three groups, matched for baseline performance, and received either bilateral lesions of the OFC (n 12), BLA (n 10), or vehicle infusions (n 10) into one of these areas. Subjects were anesthetized using 100 ml/kg Avertin [10 gm of 2,2,2- tribromoethanol (Sigma, Poole, UK) in 5 gm of tertiary amyl alcohol, diluted in a solution of 40 ml of ethanol and 450 ml of PBS] and secured in a stereotaxic frame. Excitotoxic lesions were made using 0.09 M quinolinic acid dissolved in 0.1 M phosphate buffer, and the ph was adjusted with 0.1 M NaOH to between 6 and 7. Infusions of toxin vehicle were made using a 31 gauge stainless steel injector (Coopers Needleworks, Cambridge UK) attached to a Hamilton (Reno, NV) microinfusion pump by polyethylene tubing (Portex, Hythe, UK) according to the following coordinates (Paxinos and Watson, 1998): BLA: site 1 anteroposterior (AP), 3.0, lateral (L), 4.6, dorsoventral (DV), 7.8, 0.1 l infused over 2 min, site 2 AP 2.8, L, 4.6, DV, 7.8, 0.2 l per 2 min; OFC: site 1 AP, 4.0, L, 0.8, DV, 3.4, 0.2 l per 3 min, site 2 AP, 3.7, L, 2.0, DV, 3.6, 0.3 l per 3 min, site 3 AP 3.2, L, 2.6, DV, 4.4, Figure 1. A, B, Basic outline of the delay-discounting task in terms of the sequence of events in a trial ( A) and the timeline of the experiment ( B). 0.3 l per 3 min. The AP coordinate was taken from bregma, the L coordinate from the midline, and the DV coordinate from dura. Data analysis. Analyses were conducted using SPSS for Windows (version 9.0; SPSS, Chicago, IL). The total number of choices of the large reward per delay per session (maximum 10 responses) was used to analyze choice behavior. These data were arcsine transformed to limit the effect of an artificially imposed ceiling. The number of trials omitted did not affect calculation of this measure. To determine whether animals had successfully acquired the task and reached stable baseline performance, data from seven sessions were analyzed by repeated-measures ANOVA with two within-subjects factors (DAY and DELAY). If the effect of DE- LAY was significant at the p 0.05 level but there was no main effect of DAY, animals were judged to have reliably acquired the task. The behavioral effects of the two lesions were assessed by comparing the performance of animals in each lesion group to data from sham-operated controls. Data from seven stable postoperative sessions were analyzed using repeated-measures ANOVA with LESION as a between-subjects factor and DAY and DELAY as within-subjects factors. The number of trials omitted, the latency to respond on the levers, and the latency to collect reward were also analyzed. Histology. After the completion of behavioral testing, animals were terminally anesthetized with sodium pentobarbitone (3 ml/kg, i.p.; Genus Express, Dunnington, UK) and perfused transcardially with 0.01 M PBS followed by 4% paraformaldehyde. The brains were removed, postfixed in 4% paraformaldehyde, and transferred into 20% sucrose in 0.01 M PBS 24 hr before being cut on a freezing microtome. Coronal sections (60 m) were stained with cresyl violet, and the location of the lesions was mapped onto standardized sections of the rat brain (Paxinos and Watson, 1998).
3 4720 J. Neurosci., May 19, (20): Winstanley et al. Roles of BLA and OFC in Impulsive Choice Results Lesion analysis Histological verification of the size and location of lesions depicted in Figure 2 was conducted by observers who were blind to the experimental conditions and results. Five animals were excluded from the OFC lesion group because the lesions were incomplete, unilateral, or extended into the prelimbic cortex. Three animals were excluded from the BLA lesion group because lesions incorporated the central nucleus of the amygdala, extended too far rostrally, or were incomplete. In general, the behavioral profile of animals with lesions encompassing the targeted area behaved in a similar manner to those with lesions restricted to that area, but the former were omitted from the analysis. Behavioral analysis Preoperatively, a typical pattern of delaydependent choice behavior was observed, with animals preferring the large reward at the beginning of the session when its delivery was immediate and shifting their preference to the small reward as the delay to the large reward increased (DELAY: F (4,116) 66.4; p ) (Fig. 3). Postoperatively, animals with BLA lesions (n 7) preferred the small immediate reward to a greater extent than shamoperated controls (DELAY LESION: F (4,240) 3.346; p 0.017), whereas OFClesioned rats (n 7) showed the opposite pattern of behavior, increasing their choice of the larger delayed reward (DE- LAY LESION: F (4,240) 3.156; p 0.024) (Fig. 3). When the delays were removed, all animals consistently chose the large reward, indicating that subjects in each group were capable of discriminating relative reward magnitude. Animals with BLA or OFC lesions did not omit more trials compared with sham-operated controls (LESION: F (2,20) 0.231; NS), and the latencies to respond and to collect reward were similar in all three groups (response latency, LESION: F (2,20) 1.51, NS; collection latency, LESION: F (2,20) 0.233, NS) (Table 1). Discussion Although selective damage to the BLA increased impulsivity in a rodent delay-discounting task, OFC lesions decreased impulsivity. The contrasting nature of these changes provides perhaps the first behavioral evidence of a dissociation between the functional role of these two areas in modulating cognition and also delineates the neural circuitry involved in mediating this form of impulsivity. BLA lesions increase impulsive choice: impairments in maintaining representations of reward Theoretically, BLA lesions may have increased impulsive choice by preventing the animal from remembering that responding on lever Figure 2. A D, Photomicrographs depicting a typical OFC lesion ( B) and BLA lesion ( D) compared with appropriate sham control tissue (A and C, respectively). MO, Medial orbital cortex; VO, ventral orbital cortex; LO, lateral orbital cortex; AI, agranular insular cortex; CeN, central amygdaloid nucleus. Figure 3. Effect of lesions to the OFC and BLA on performance of a delay-to-gratification task compared with sham-operated controls. Data shown are mean number of responses and SEM taken from seven consecutive postoperative sessions. B leads to delivery of the large reward when it is delayed. Few studies have directly investigated whether BLA lesions impair conditioning with long CS unconditioned stimulus (US) intervals, although blockade of GABA A receptors in the BLA facilitates, rather than impairs, aversive learning with CS US delays (Ferry and Di Scala, 1997). However, memories for reward and reward-predicting stimuli can be formed and used after BLA lesions, as indicated by the lack of impairment produced by these lesions on autoshaping paradigms (Parkinson et al., 2000). Because BLA-lesioned rats strongly preferred the large reward in the absence of delays, it is unlikely that impulsive choice occurred simply because the large reward lacked value under all conditions. The increase in impulsivity may also have reflected an increased sensitivity to satiation in BLA-lesioned rats (and vice versa in OFC-lesioned animals). Although possible, previous work has indicated that changes in satiation do not significantly alter delay-discounting in trained animals (Cardinal et al., 2000). Alternatively, increased impulsive choice after BLA lesions
4 Winstanley et al. Roles of BLA and OFC in Impulsive Choice J. Neurosci., May 19, (20): Table 1. Levels of omissions, latency to respond, and latency to collect reward per session Variable Preoperative Postoperative Sham BLA OFC Sham BLA OFC Omissions 0.34 (0.30) 0.28 (0.30) 0.37 (0.28) 0.69 (0.56) 0.50 (0.33) 0.35 (0.17) Latency to respond (sec) 1.01 (0.07) 0.96 (0.07) 0.92 (0.04) 1.19 (0.16) 0.99 (0.05) 1.22 (0.09) Latency to collect reward (sec) 0.29 (0.01) 0.29 (0.03) 0.32 (0.04) 0.29 (0.14) 0.23 (0.07) 0.32 (0.03) Data shown are mean (SEM). may reflect an impairment in using representations of the incentive value of reward to guide behavior. To maintain responding for the large reward when it is delayed, subjects may hold a representation of the reward on-line during the delay period. Responding on the lever leading to delivery of the large reward, or the postresponse delay itself, can function as conditioned reinforcers, thus helping to maintain responding despite the delay to gratification (Garrud et al., 1981). The BLA is critically involved in such conditioned reinforcement processes through maintaining a representation of the appetitive value of conditioned stimuli (Cador et al., 1989; Burns et al., 1993; Gallagher, 2000), and there is considerable behavioral evidence suggesting that interactions between the BLA and NAC are important in enabling such stimuli to guide behavior (Cardinal et al., 2002; Setlow et al., 2002). In this study, BLA lesions may have prevented the utilization of such representations. Therefore, the value of the large reward was not maintained across the delay, leading the subject to choose impulsively. The NAC has been described as the limbicmotor interface, providing an important route whereby the limbic system influences actions through allowing CSs to guide behavior (Mogenson et al., 1980). The fact that similar increases in impulsive choice are observed after NAC lesions indicates that the NAC and BLA may be part of a common circuit promoting choice of the large delayed reward, potentially through maintaining a representation of such a reward in its absence. OFC lesions decrease impulsive choice: impairments in updating representations of incentive value on the basis of consequences of a response The decrease in impulsive choice caused by OFC lesions contrasts with the comparative lack of effect of PrL or ACC lesions on this paradigm. Dissociable effects after damage to different frontal regions have been observed in numerous behavioral tests, leading to the suggestion that dorsomedial prefrontal cortex is integral to attentional and mnemonic processes, whereas ventral prefrontal regions such as OFC enable cognitive and behavioral flexibility (Robbins, 1996; Bechara et al., 1998; Chudasama et al., 2003). In particular, subjects with OFC lesions perseverate in responding to the previously rewarded stimulus during reversal learning (Jones and Mishkin, 1972; Chudasama and Robbins, 2003). Decreased impulsive choice after OFC lesions may therefore arise through perseveration in the response associated with the largest reward, regardless of the consequences of that response. Although neurons encoding reinforcement are found throughout the PFC, dissociations have been observed between the firing pattern of neurons within the primate dorsolateral and orbitofrontal PFC in response to CSs indicative of a more primary role for the OFC in representing reward-related information (Wallis and Miller, 2003). The fact that OFC lesions increase responding for the large delayed reward and increase perseverative behavior supports the view that the OFC is involved in revising the representation of the incentive value of a response as the outcome associated with that response change (Schoenbaum et al., 2003a). As the delay to the large reward increases, responding for that reward becomes less attractive as the delay decreases the subjective value of the large reward (Ainslie, 1975). OFC-lesioned rats did not show such a marked shift toward preferring the smaller immediate reward, indicating that the delay failed to sufficiently devalue the large reward, and that the change in reinforcement contingency had minimal impact on decision-making processes. The finding that OFC lesions reduced impulsive choice seemingly contradicts the observation that patients with OFC damage choose more impulsively in gambling paradigms (Bechara et al., 1999; Rogers et al., 1999). However, increased choice of risky options in gambling tasks and increased choice of the large delayed reward in the present study may both result from inadequate integration of the consequences of responding for the large reward with the incentive value of that reward, so that the anticipated punishment (loss of money in the former task, delay to reward delivery in the latter) ultimately fails to sufficiently devalue the response outcome. Related yet dissociable roles for the BLA and OFC in regulating goal-directed behavior These data add to a growing body of evidence suggesting that, although the BLA and OFC cooperate to regulate goal-directed behavior (Baxter et al., 2000), the BLA primarily encodes the incentive value of a stimulus, whereas the OFC is also implicated in monitoring and updating representations of expected outcomes associated with making responses on the basis of such stimuli. This distinction is supported by electrophysiological recordings in the BLA and OFC obtained during performance of an odor-mediated go/no-go discrimination task (Schoenbaum et al., 1999). Although neuronal firing in the BLA primarily encodes the current motivational significance of these stimuli, the firing pattern of OFC neurons signals the specific sensory properties of the stimulus in addition to its predictive value. Furthermore, the firing of OFC neurons is more indicative of the current behavioral strategy, particularly when the outcome predicted by a stimulus is altered or reversed (Schoenbaum et al., 2000). A previous study has shown that lesions to the OFC performed before the acquisition of a delay-discounting task have the opposite effect, increasing choice of the small immediate reward (Mobini et al., 2002). This accords with observations that OFC neurons encoding the expected outcome of a response are only observed late in training once accurate performance has been acquired, unlike neuronal activity in the BLA (Schoenbaum et al., 1999). Removing the modulatory influence of the OFC before and after the acquisition of reward contingencies may thus have very different effects on choice behavior. Because the BLA and OFC are heavily interconnected, the behavioral changes caused by damage to one region could arise in part from disrupted functioning in the other. Recently, it has been demonstrated that lesions to the BLA alter the response properties of neurons in the OFC during go/no-go performance,
5 4722 J. Neurosci., May 19, (20): Winstanley et al. Roles of BLA and OFC in Impulsive Choice so that cues predicting reward delivery fail to activate neurons encoding the expected outcome in this region (Schoenbaum et al., 2003b). Hence, the fact that BLA lesions decreased choice of the large delayed reward could reflect alterations in the capability of the OFC to use information maintained in the BLA regarding cues predictive of reward in the task (i.e., responding on the lever associated with reward delivery) to strengthen the representation of the expected outcome (delivery of the large reward). In summary, current findings implicate pathways involving the OFC, BLA, and NAC core in mediating impulsive choice, whereas input from the PrL and ACC does not play a critical role. Clearly, different frontal regions make relatively specific contributions to the regulation of behavior despite gross similarities in their efferent and afferent projections (Öngür and Price, 2000; Pitkänen, 2000). Such functional dissociations may reflect differences in the precise topographical organization of frontostriatal projections, in which a ventral-to-dorsal gradient in the PFC corresponds to a ventromedial-to-dorsolateral gradient in the striatum (Berendse et al., 1992). The finding that lesions to the OFC and BLA can shift the delay-discounting function in opposite directions also delineates the relative contributions of these two areas in the regulation of goal-directed behavior and impulsive decision making. References Ainslie G (1975) A behavioral theory of impulsiveness and impulse control. Psychol Bull 82: Baxter MG, Parker A, Lindner CCC, Izquierdo AD, Murray EA (2000) Control of response selection by reinforcer value requires interaction of amygdala and orbital prefrontal cortex. J Neurosci 20: Bechara A, Damasio H, Tranel D, Anderson SW (1998) Dissociation of working memory from decision making within the human prefrontal cortex. J Neurosci 18: Bechara A, Damasio H, Damasio AR, Lee GP (1999) Different contributions of the human amygdala and ventromedial prefrontal cortex to decisionmaking. J Neurosci 19: Berendse HW, Galis-de Graaf Y, Groenewegen HJ (1992) Topographical organization and relationship with ventral striatal compartments of prefrontal corticostriatal projections in the rat. J Comp Neurol 316: Burns LH, Robbins TW, Everitt BJ (1993) Differential effects of excitotoxic lesions of the basolateral amygdala, ventral subiculum and prefrontal cortex on responding with conditioned reinforcement and locomotor activity potentiated by intra-accumbens infusions of amphetamine. Behav Brain Res 55: Cador M, Robbins TW, Everitt BJ (1989) Involvement of the amygdala in stimulus-reward associations: interaction with the ventral striatum. Neuroscience 30: Cardinal RN, Robbins TW, Everitt BJ (2000) The effects of d-amphetamine, chlordiazepoxide, alpha-flupenthixol and behavioral manipulations on choice of signalled and unsignalled delayed reinforcement in rats. Psychopharmacology 152: Cardinal RN, Pennicott DR, Sugathapala CL, Robbins TW, Everitt BJ (2001) Impulsive choice induced in rats by lesions of the nucleus accumbens core. Science 292: Cardinal RN, Parkinson JA, Hall J, Everitt BJ (2002) Emotion and motivation: the role of the amygdala, ventral striatum, and prefrontal cortex. Neurosci Biobehav Rev 26: Chudasama Y, Robbins TW (2003) Dissociable contributions of the orbitofrontal and infralimbic cortex to pavlovian autoshaping and discrimination reversal learning: further evidence for the functional heterogeneity of the rodent frontal cortex. J Neurosci 23: Chudasama Y, Passetti F, Rhodes SEV, Lopian D, Desai A, Robbins TW (2003) Dissociable aspects of performance on the 5-choice serial reaction time task following lesions of the dorsal anterior cingulae, infralimbic and orbitofrontal cortex in the rat: differential effects on selectivity, impulsivity and compulsivity. Behav Brain Res 146: Damasio AR (1994) Descartes error. New York: Putman. Evenden JL, Ryan CN (1996) The pharmacology of impulsive behavior in rats: the effects of drugs on response choice with varying delays of reinforcement. Psychopharmacology 128: Ferry B, Di Scala G (1997) Bicuculline administration into basolateral amygdala facilitates trace conditioning of odor aversion in the rat. Neurobiol Learn Mem 67: Gallagher M (2000) The amygdala and associative learning. In: The amygdala: a functional analysis, Ed 2 (Aggleton JP, ed), pp New York: Oxford UP. Garrud P, Goodall G, Mackintosh NJ (1981) Overshadowing of a stimulusreinforcer association by an instrumental response. J Exp Psychol 33B: Jones B, Mishkin M (1972) Limbic lesions and the problem of stimulusreinforcement associations. Exp Neurol 36: Krettek JE, Price JL (1977) Projections from the amygdaloid complex to the cerebral cortex and thalamus in the rat and cat. J Comp Neurol 172: LeDoux JE (2000) The amygdala and emotion: a view through fear. In: The amygdala: a functional analysis, Ed 2 (Aggleton JP, ed), pp New York: Oxford UP. Logue AW (1988) Res on self-control an integrating framework. Behav Brain Sci 11: McDonald AJ (1991) Organization of amygdaloid projections to the prefrontal cortex and associated striatum in the rat. Neuroscience 44:1 14. Mobini S, Body S, Ho M-Y, Bradshaw CM, Szabadi E, Deakin JFW, Anderson IM (2002) Effects of lesions of the orbitofrontal cortex on sensitivity to delayed and probabilistic reinforcement. Psychopharmacology 160: Mogenson GJ, Jones DL, Yim CY (1980) From motivation to action: functional interface between the limbic system and the motor system. Prog Neurobiol 14: Nauta WJH (1971) The problem of the frontal lobe: a reinterpretation. J Psychiat Res 8: Öngür D, Price JL (2000) The organization of networks within the orbital and medial prefrontal cortex of rats, monkeys and humans. Cereb Cortex 10: Parkinson JA, Robbins TW, Everitt BJ (2000) Dissociable roles of the central and basolateral amygdala in appetitive emotional learning. Eur J Neurosci 12: Paxinos G, Watson C (1998) The rat brain in stereotaxic co-ordinates, Ed 4. Sydney: Academic. Pitkänen A (2000) Connectivity of the rat amygdaloid complex. In: The amygdala: a functional analysis, Ed 2 (Aggleton JP, ed), pp New York: Oxford UP. Robbins TW (1996) Dissociating executive functions of the prefrontal cortex. Phil Trans R Soc Lond B Biol Sci 351: Rogers RD, Everitt BJ, Baldacchino A, Blackshaw AJ, Swainson R, Wynne K, Baker NB, Hunter J, Carthy T, Booker E, London M, Deakin JFW, Sahakian BJ, Robbins TW (1999) Dissociable deficits in the decision-making cognition of chronic amphetamine abusers, opiate abusers, patients with focal damage to prefrontal cortex, and tryptophan-depleted normal volunteers: evidence for monoaminergic mechanisms. Neuropsychopharmacol 20: Schoenbaum G, Chiba AA, Gallagher M (1999) Neural encoding in orbitofrontal cortex and basolateral amygdala during olfactory discrimination learning. J Neurosci 19: Schoenbaum G, Chiba AA, Gallagher M (2000) Changes in functional connectivity in orbitofrontal cortex and basolateral amygdala during learning and reversal learning. J Neurosci 20: Schoenbaum G, Setlow B, Ramus SJ (2003a) A systems approach to orbitofrontal cortex function: recordings in rat orbitofrontal cortex reveal interactions with different learning systems. Behav Brain Res 146: Schoenbaum G, Setlow B, Saddoris MP, Gallagher M (2003b) Encoding predicted outcome and acquired value in orbitofrontal cortex during cue sampling depends upon input from basolateral amygdala. Neuron 39: Setlow B, Holland PC, Gallagher M (2002) Disconnection of the basolateral amygdala complex and nucleus accumbens impairs Pavlovian secondorder conditioned responses. Behav Neurosci 116: Wallis JD, Miller EK (2003) Neuronal activity in primate dorsolateral and orbital prefrontal cortex during performance of a reward preference task Eur J Neurosci 18:
Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer
 Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer RN Cardinal, JA Parkinson *, TW Robbins, A Dickinson, BJ Everitt Departments of Experimental
Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer RN Cardinal, JA Parkinson *, TW Robbins, A Dickinson, BJ Everitt Departments of Experimental
Role of the anterior cingulate cortex in the control over behaviour by Pavlovian conditioned stimuli
 Role of the anterior cingulate cortex in the control over behaviour by Pavlovian conditioned stimuli in rats RN Cardinal, JA Parkinson, H Djafari Marbini, AJ Toner, TW Robbins, BJ Everitt Departments of
Role of the anterior cingulate cortex in the control over behaviour by Pavlovian conditioned stimuli in rats RN Cardinal, JA Parkinson, H Djafari Marbini, AJ Toner, TW Robbins, BJ Everitt Departments of
Effects of limbic corticostriatal lesions on a u t o shaping performance in rats
 Effects of limbic corticostriatal lesions on a u t o shaping performance in rats BJ Everitt, JA Parkinson, G Lachenal, KM Halkerston, N Rudarakanchana, RN Cardinal, J Hall, CH Morrison, JW Dalley, SR Howes,
Effects of limbic corticostriatal lesions on a u t o shaping performance in rats BJ Everitt, JA Parkinson, G Lachenal, KM Halkerston, N Rudarakanchana, RN Cardinal, J Hall, CH Morrison, JW Dalley, SR Howes,
Fronto-executive functions in rodents: neural and neurochemical substrates
 Fronto-executive functions in rodents: neural and neurochemical substrates Rudolf Cardinal, Jeff Dalley, Filippo Passetti, David Theobald, Catharine Winstanley, Trevor Robbins MRC Centre for Behavioural
Fronto-executive functions in rodents: neural and neurochemical substrates Rudolf Cardinal, Jeff Dalley, Filippo Passetti, David Theobald, Catharine Winstanley, Trevor Robbins MRC Centre for Behavioural
The orbitofrontal cortex, predicted value, and choice
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Critical Contributions of the Orbitofrontal Cortex to Behavior The orbitofrontal cortex, predicted value, and choice
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Critical Contributions of the Orbitofrontal Cortex to Behavior The orbitofrontal cortex, predicted value, and choice
The Contribution of the Amygdala to Reward-Related Learning and Extinction
 Chapter 14 The Contribution of the Amygdala to Reward-Related Learning and Extinction Rose Chesworth and Laura Corbit Additional information is available at the end of the chapter Abstract There has been
Chapter 14 The Contribution of the Amygdala to Reward-Related Learning and Extinction Rose Chesworth and Laura Corbit Additional information is available at the end of the chapter Abstract There has been
The Contribution of the Medial Prefrontal Cortex, Orbitofrontal Cortex, and Dorsomedial Striatum to Behavioral Flexibility
 The Contribution of the Medial Prefrontal Cortex, Orbitofrontal Cortex, and Dorsomedial Striatum to Behavioral Flexibility MICHAEL E. RAGOZZINO Department of Psychology, University of Illinois at Chicago,
The Contribution of the Medial Prefrontal Cortex, Orbitofrontal Cortex, and Dorsomedial Striatum to Behavioral Flexibility MICHAEL E. RAGOZZINO Department of Psychology, University of Illinois at Chicago,
Succumbing to instant gratification without the nucleus accumbens
 Succumbing to instant gratification without the nucleus accumbens Rudolf N. Cardinal Department of Experimental Psychology, University of Cambridge Downing Street, Cambridge CB2 3EB, United Kingdom. Telephone:
Succumbing to instant gratification without the nucleus accumbens Rudolf N. Cardinal Department of Experimental Psychology, University of Cambridge Downing Street, Cambridge CB2 3EB, United Kingdom. Telephone:
Amygdala and Orbitofrontal Cortex Lesions Differentially Influence Choices during Object Reversal Learning
 8338 The Journal of Neuroscience, August 13, 2008 28(33):8338 8343 Behavioral/Systems/Cognitive Amygdala and Orbitofrontal Cortex Lesions Differentially Influence Choices during Object Reversal Learning
8338 The Journal of Neuroscience, August 13, 2008 28(33):8338 8343 Behavioral/Systems/Cognitive Amygdala and Orbitofrontal Cortex Lesions Differentially Influence Choices during Object Reversal Learning
Lesions of the Basolateral Amygdala Disrupt Selective Aspects of Reinforcer Representation in Rats
 The Journal of Neuroscience, November 15, 2001, 21(22):9018 9026 Lesions of the Basolateral Amygdala Disrupt Selective Aspects of Reinforcer Representation in Rats Pam Blundell, 1 Geoffrey Hall, 1 and
The Journal of Neuroscience, November 15, 2001, 21(22):9018 9026 Lesions of the Basolateral Amygdala Disrupt Selective Aspects of Reinforcer Representation in Rats Pam Blundell, 1 Geoffrey Hall, 1 and
Instrumental Conditioning VI: There is more than one kind of learning
 Instrumental Conditioning VI: There is more than one kind of learning PSY/NEU338: Animal learning and decision making: Psychological, computational and neural perspectives outline what goes into instrumental
Instrumental Conditioning VI: There is more than one kind of learning PSY/NEU338: Animal learning and decision making: Psychological, computational and neural perspectives outline what goes into instrumental
Prefrontal dysfunction in drug addiction: Cause or consequence? Christa Nijnens
 Prefrontal dysfunction in drug addiction: Cause or consequence? Master Thesis Christa Nijnens September 16, 2009 University of Utrecht Rudolf Magnus Institute of Neuroscience Department of Neuroscience
Prefrontal dysfunction in drug addiction: Cause or consequence? Master Thesis Christa Nijnens September 16, 2009 University of Utrecht Rudolf Magnus Institute of Neuroscience Department of Neuroscience
Orbital prefrontal cortex and guidance of instrumental behaviour in rats under reversal conditions
 Behavioural Brain Research 143 (2003) 49 56 Research report Orbital prefrontal cortex and guidance of instrumental behaviour in rats under reversal conditions Ines Bohn, Christian Giertler, Wolfgang Hauber
Behavioural Brain Research 143 (2003) 49 56 Research report Orbital prefrontal cortex and guidance of instrumental behaviour in rats under reversal conditions Ines Bohn, Christian Giertler, Wolfgang Hauber
Separate neural pathways process different decision costs
 Separate neural pathways process different decision costs Peter H Rudebeck, Mark E Walton, Angharad N Smyth, David M Bannerman & Matthew F S Rushworth Behavioral ecologists and economists emphasize that
Separate neural pathways process different decision costs Peter H Rudebeck, Mark E Walton, Angharad N Smyth, David M Bannerman & Matthew F S Rushworth Behavioral ecologists and economists emphasize that
Neural systems of reinforcement for drug addiction: from actions to habits to compulsion
 NEUROBIOLOGY OF ADDICTION REVIEW Neural systems of reinforcement for drug addiction: from actions to habits to compulsion Barry J Everitt & Trevor W Robbins Drug addiction is increasingly viewed as the
NEUROBIOLOGY OF ADDICTION REVIEW Neural systems of reinforcement for drug addiction: from actions to habits to compulsion Barry J Everitt & Trevor W Robbins Drug addiction is increasingly viewed as the
THE AMYGDALA AND REWARD
 THE AMYGDALA AND REWARD Mark G. Baxter* and Elisabeth A. Murray The amygdala an almond-shaped group of nuclei at the heart of the telencephalon has been associated with a range of cognitive functions,
THE AMYGDALA AND REWARD Mark G. Baxter* and Elisabeth A. Murray The amygdala an almond-shaped group of nuclei at the heart of the telencephalon has been associated with a range of cognitive functions,
Connect with amygdala (emotional center) Compares expected with actual Compare expected reward/punishment with actual reward/punishment Intuitive
 Orbitofronal Notes Frontal lobe prefrontal cortex 1. Dorsolateral Last to myelinate Sleep deprivation 2. Orbitofrontal Like dorsolateral, involved in: Executive functions Working memory Cognitive flexibility
Orbitofronal Notes Frontal lobe prefrontal cortex 1. Dorsolateral Last to myelinate Sleep deprivation 2. Orbitofrontal Like dorsolateral, involved in: Executive functions Working memory Cognitive flexibility
BIOMED 509. Executive Control UNM SOM. Primate Research Inst. Kyoto,Japan. Cambridge University. JL Brigman
 BIOMED 509 Executive Control Cambridge University Primate Research Inst. Kyoto,Japan UNM SOM JL Brigman 4-7-17 Symptoms and Assays of Cognitive Disorders Symptoms of Cognitive Disorders Learning & Memory
BIOMED 509 Executive Control Cambridge University Primate Research Inst. Kyoto,Japan UNM SOM JL Brigman 4-7-17 Symptoms and Assays of Cognitive Disorders Symptoms of Cognitive Disorders Learning & Memory
Cortico-limbic-striatal circuits subserving different forms of cost benefit decision making
 Cognitive, Affective, & Behavioral Neuroscience 2008, 8 (4), 375-389 doi:10.3758/cabn.8.4.375 Cortico-limbic-striatal circuits subserving different forms of cost benefit decision making Sta n B. Floresco,
Cognitive, Affective, & Behavioral Neuroscience 2008, 8 (4), 375-389 doi:10.3758/cabn.8.4.375 Cortico-limbic-striatal circuits subserving different forms of cost benefit decision making Sta n B. Floresco,
Lesions of the Orbitofrontal but not Medial Prefrontal Cortex Disrupt Conditioned Reinforcement in Primates
 The Journal of Neuroscience, December 3, 2003 23(35):11189 11201 11189 Behavioral/Systems/Cognitive Lesions of the Orbitofrontal but not Medial Prefrontal Cortex Disrupt Conditioned Reinforcement in Primates
The Journal of Neuroscience, December 3, 2003 23(35):11189 11201 11189 Behavioral/Systems/Cognitive Lesions of the Orbitofrontal but not Medial Prefrontal Cortex Disrupt Conditioned Reinforcement in Primates
LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT
 ACTA NEUROBIOL: EXP. 1988, 48: 117-121 Short communication LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT W. Jeffrey WILSON and Jennifer C. HALL Department of Psychological
ACTA NEUROBIOL: EXP. 1988, 48: 117-121 Short communication LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT W. Jeffrey WILSON and Jennifer C. HALL Department of Psychological
Nucleus accumbens lesions impair context, but not cue, conditioning in rats
 Learning and Memory PREVIOUS work has provided evidence of a role for the hippocampal formation in contextual as opposed to cue conditioning. Similar deficits have been observed after transection of the
Learning and Memory PREVIOUS work has provided evidence of a role for the hippocampal formation in contextual as opposed to cue conditioning. Similar deficits have been observed after transection of the
Enhancement of Latent Inhibition in Rats With Electrolytic Lesions of the Hippocampus
 Behavioral Neuroscience 1995. Vol. 109, No. 2, 366-370 Copyright 1995 by the American Psychological Association, Inc. 0735-7044/95/$3.00 BRIEF COMMUNICATIONS Enhancement of Latent Inhibition in Rats With
Behavioral Neuroscience 1995. Vol. 109, No. 2, 366-370 Copyright 1995 by the American Psychological Association, Inc. 0735-7044/95/$3.00 BRIEF COMMUNICATIONS Enhancement of Latent Inhibition in Rats With
Orbitofrontal cortex. From Wikipedia, the free encyclopedia. Approximate location of the OFC shown on a sagittal MRI
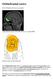 Orbitofrontal cortex From Wikipedia, the free encyclopedia Approximate location of the OFC shown on a sagittal MRI Orbital surface of left frontal lobe. The orbitofrontal cortex (OFC) is a prefrontal cortex
Orbitofrontal cortex From Wikipedia, the free encyclopedia Approximate location of the OFC shown on a sagittal MRI Orbital surface of left frontal lobe. The orbitofrontal cortex (OFC) is a prefrontal cortex
The Role of Orbitofrontal Cortex in Decision Making
 The Role of Orbitofrontal Cortex in Decision Making A Component Process Account LESLEY K. FELLOWS Montreal Neurological Institute, McGill University, Montréal, Québec, Canada ABSTRACT: Clinical accounts
The Role of Orbitofrontal Cortex in Decision Making A Component Process Account LESLEY K. FELLOWS Montreal Neurological Institute, McGill University, Montréal, Québec, Canada ABSTRACT: Clinical accounts
Thalamic--Prefrontal Cortical--Ventral Striatal Circuitry Mediates Dissociable Components of Strategy Set Shifting
 Cerebral Cortex Advance Access published September 8, 2006 Cerebral Cortex doi:10.1093/cercor/bhl073 Thalamic--Prefrontal Cortical--Ventral Striatal Circuitry Mediates Dissociable Components of Strategy
Cerebral Cortex Advance Access published September 8, 2006 Cerebral Cortex doi:10.1093/cercor/bhl073 Thalamic--Prefrontal Cortical--Ventral Striatal Circuitry Mediates Dissociable Components of Strategy
THE PREFRONTAL CORTEX. Connections. Dorsolateral FrontalCortex (DFPC) Inputs
 THE PREFRONTAL CORTEX Connections Dorsolateral FrontalCortex (DFPC) Inputs The DPFC receives inputs predominantly from somatosensory, visual and auditory cortical association areas in the parietal, occipital
THE PREFRONTAL CORTEX Connections Dorsolateral FrontalCortex (DFPC) Inputs The DPFC receives inputs predominantly from somatosensory, visual and auditory cortical association areas in the parietal, occipital
The Adolescent Developmental Stage
 The Adolescent Developmental Stage o Physical maturation o Drive for independence o Increased salience of social and peer interactions o Brain development o Inflection in risky behaviors including experimentation
The Adolescent Developmental Stage o Physical maturation o Drive for independence o Increased salience of social and peer interactions o Brain development o Inflection in risky behaviors including experimentation
Distinct contributions of frontal areas to emotion and social behaviour in the rat
 European Journal of Neuroscience, Vol. 26, pp. 2315 2326, 2007 doi:10.1111/j.1460-9568.2007.05844.x Distinct contributions of frontal areas to emotion and social behaviour in the rat Peter H. Rudebeck,
European Journal of Neuroscience, Vol. 26, pp. 2315 2326, 2007 doi:10.1111/j.1460-9568.2007.05844.x Distinct contributions of frontal areas to emotion and social behaviour in the rat Peter H. Rudebeck,
Reward Systems: Human
 Reward Systems: Human 345 Reward Systems: Human M R Delgado, Rutgers University, Newark, NJ, USA ã 2009 Elsevier Ltd. All rights reserved. Introduction Rewards can be broadly defined as stimuli of positive
Reward Systems: Human 345 Reward Systems: Human M R Delgado, Rutgers University, Newark, NJ, USA ã 2009 Elsevier Ltd. All rights reserved. Introduction Rewards can be broadly defined as stimuli of positive
BRAIN MECHANISMS OF REWARD AND ADDICTION
 BRAIN MECHANISMS OF REWARD AND ADDICTION TREVOR.W. ROBBINS Department of Experimental Psychology, University of Cambridge Many drugs of abuse, including stimulants such as amphetamine and cocaine, opiates
BRAIN MECHANISMS OF REWARD AND ADDICTION TREVOR.W. ROBBINS Department of Experimental Psychology, University of Cambridge Many drugs of abuse, including stimulants such as amphetamine and cocaine, opiates
Subjects. Thirty-five adult male Lister Hooded rats (Charles River, Kent, UK),
 Supplemental Material: Subjects. Thirty-five adult male Lister Hooded rats (Charles River, Kent, UK), weighing between 280 300 g at the beginning of the experiment, were housed singly in holding rooms
Supplemental Material: Subjects. Thirty-five adult male Lister Hooded rats (Charles River, Kent, UK), weighing between 280 300 g at the beginning of the experiment, were housed singly in holding rooms
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/330/6009/1408/dc1 Supporting Online Material for Paradoxical False Memory for Objects After Brain Damage Stephanie M. McTighe, Rosemary A. Cowell, Boyer D. Winters,
www.sciencemag.org/cgi/content/full/330/6009/1408/dc1 Supporting Online Material for Paradoxical False Memory for Objects After Brain Damage Stephanie M. McTighe, Rosemary A. Cowell, Boyer D. Winters,
Parallel incentive processing: an integrated view of amygdala function
 ARTICLE IN PRESS Parallel incentive processing: an integrated view of amygdala function Bernard W. Balleine 1 and Simon Killcross 2 1 Department of Psychology and the Brain Research Institute, University
ARTICLE IN PRESS Parallel incentive processing: an integrated view of amygdala function Bernard W. Balleine 1 and Simon Killcross 2 1 Department of Psychology and the Brain Research Institute, University
The Effects of Temporary Inactivation of the Orbital Cortex in the Signal Attenuation Rat Model of Obsessive Compulsive Disorder
 Behavioral Neuroscience Copyright 2006 by the American Psychological Association 2006, Vol. 120, No. 4, 976 983 0735-7044/06/$12.00 DOI: 10.1037/0735-7044.120.4.976 The Effects of Temporary Inactivation
Behavioral Neuroscience Copyright 2006 by the American Psychological Association 2006, Vol. 120, No. 4, 976 983 0735-7044/06/$12.00 DOI: 10.1037/0735-7044.120.4.976 The Effects of Temporary Inactivation
Stop-Signal Reaction-Time Task Performance: Role of Prefrontal Cortex and Subthalamic Nucleus
 Cerebral Cortex January 2008;18:178-188 doi:10.1093/cercor/bhm044 Advance Access publication May 20, 2007 Stop-Signal Reaction-Time Task Performance: Role of Prefrontal Cortex and Subthalamic Nucleus Dawn
Cerebral Cortex January 2008;18:178-188 doi:10.1093/cercor/bhm044 Advance Access publication May 20, 2007 Stop-Signal Reaction-Time Task Performance: Role of Prefrontal Cortex and Subthalamic Nucleus Dawn
The effects of amygdala lesions on conditioned stimulus-potentiated eating in rats
 Physiology & Behavior 76 (2002) 117 129 The effects of amygdala lesions on conditioned stimulus-potentiated eating in rats Peter C. Holland a, *, Gorica D. Petrovich b, Michela Gallagher b a Department
Physiology & Behavior 76 (2002) 117 129 The effects of amygdala lesions on conditioned stimulus-potentiated eating in rats Peter C. Holland a, *, Gorica D. Petrovich b, Michela Gallagher b a Department
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
Distinct valuation subsystems in the human brain for effort and delay
 Supplemental material for Distinct valuation subsystems in the human brain for effort and delay Charlotte Prévost, Mathias Pessiglione, Elise Météreau, Marie-Laure Cléry-Melin and Jean-Claude Dreher This
Supplemental material for Distinct valuation subsystems in the human brain for effort and delay Charlotte Prévost, Mathias Pessiglione, Elise Météreau, Marie-Laure Cléry-Melin and Jean-Claude Dreher This
Psych3BN3 Topic 4 Emotion. Bilateral amygdala pathology: Case of S.M. (fig 9.1) S.M. s ratings of emotional intensity of faces (fig 9.
 Psych3BN3 Topic 4 Emotion Readings: Gazzaniga Chapter 9 Bilateral amygdala pathology: Case of S.M. (fig 9.1) SM began experiencing seizures at age 20 CT, MRI revealed amygdala atrophy, result of genetic
Psych3BN3 Topic 4 Emotion Readings: Gazzaniga Chapter 9 Bilateral amygdala pathology: Case of S.M. (fig 9.1) SM began experiencing seizures at age 20 CT, MRI revealed amygdala atrophy, result of genetic
Brain Imaging studies in substance abuse. Jody Tanabe, MD University of Colorado Denver
 Brain Imaging studies in substance abuse Jody Tanabe, MD University of Colorado Denver NRSC January 28, 2010 Costs: Health, Crime, Productivity Costs in billions of dollars (2002) $400 $350 $400B legal
Brain Imaging studies in substance abuse Jody Tanabe, MD University of Colorado Denver NRSC January 28, 2010 Costs: Health, Crime, Productivity Costs in billions of dollars (2002) $400 $350 $400B legal
nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
 Nucleus accumbens From Wikipedia, the free encyclopedia Brain: Nucleus accumbens Nucleus accumbens visible in red. Latin NeuroNames MeSH NeuroLex ID nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
Nucleus accumbens From Wikipedia, the free encyclopedia Brain: Nucleus accumbens Nucleus accumbens visible in red. Latin NeuroNames MeSH NeuroLex ID nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
Limbic system outline
 Limbic system outline 1 Introduction 4 The amygdala and emotion -history - theories of emotion - definition - fear and fear conditioning 2 Review of anatomy 5 The hippocampus - amygdaloid complex - septal
Limbic system outline 1 Introduction 4 The amygdala and emotion -history - theories of emotion - definition - fear and fear conditioning 2 Review of anatomy 5 The hippocampus - amygdaloid complex - septal
John A. Parkinson, 1 Mary C. Olmstead, 2 Lindsay H. Burns, 3 Trevor W. Robbins, 1 and Barry J. Everitt 1
 The Journal of Neuroscience, March 15, 1999, 19(6):2401 2411 Dissociation in Effects of Lesions of the Nucleus Accumbens Core and Shell on Appetitive Pavlovian Approach Behavior and the Potentiation of
The Journal of Neuroscience, March 15, 1999, 19(6):2401 2411 Dissociation in Effects of Lesions of the Nucleus Accumbens Core and Shell on Appetitive Pavlovian Approach Behavior and the Potentiation of
Abstract. Introduction. Keywords: appetitive conditioning, amygdala, context, cue, hippocampus
 European Journal of Neuroscience, Vol. 23, pp. 3071 3080, 2006 doi:10.1111/j.1460-9568.2006.04883.x Selective excitotoxic lesions of the hippocampus and basolateral amygdala have dissociable effects on
European Journal of Neuroscience, Vol. 23, pp. 3071 3080, 2006 doi:10.1111/j.1460-9568.2006.04883.x Selective excitotoxic lesions of the hippocampus and basolateral amygdala have dissociable effects on
25 Things To Know. Pre- frontal
 25 Things To Know Pre- frontal Frontal lobes Cognition Touch Sound Phineas Gage First indication can survive major brain trauma Lost 1+ frontal lobe Gage Working on a railroad Gage (then 25) Foreman on
25 Things To Know Pre- frontal Frontal lobes Cognition Touch Sound Phineas Gage First indication can survive major brain trauma Lost 1+ frontal lobe Gage Working on a railroad Gage (then 25) Foreman on
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival Threats are much better reinforcers Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival Threats are much better reinforcers Fear is a prime
The Frontal Lobes. Anatomy of the Frontal Lobes. Anatomy of the Frontal Lobes 3/2/2011. Portrait: Losing Frontal-Lobe Functions. Readings: KW Ch.
 The Frontal Lobes Readings: KW Ch. 16 Portrait: Losing Frontal-Lobe Functions E.L. Highly organized college professor Became disorganized, showed little emotion, and began to miss deadlines Scores on intelligence
The Frontal Lobes Readings: KW Ch. 16 Portrait: Losing Frontal-Lobe Functions E.L. Highly organized college professor Became disorganized, showed little emotion, and began to miss deadlines Scores on intelligence
Chapter 4. Role of the nucleus accumbens core and shell in Pavlovian instrumental transfer
 Chapter 4: Pavlovian instrumental transfer 128 Chapter 4. Role of the nucleus accumbens core and shell in Pavlovian instrumental transfer Abstract. When an initially neutral stimulus has been paired in
Chapter 4: Pavlovian instrumental transfer 128 Chapter 4. Role of the nucleus accumbens core and shell in Pavlovian instrumental transfer Abstract. When an initially neutral stimulus has been paired in
TO BE MOTIVATED IS TO HAVE AN INCREASE IN DOPAMINE. The statement to be motivated is to have an increase in dopamine implies that an increase in
 1 NAME COURSE TITLE 2 TO BE MOTIVATED IS TO HAVE AN INCREASE IN DOPAMINE The statement to be motivated is to have an increase in dopamine implies that an increase in dopamine neurotransmitter, up-regulation
1 NAME COURSE TITLE 2 TO BE MOTIVATED IS TO HAVE AN INCREASE IN DOPAMINE The statement to be motivated is to have an increase in dopamine implies that an increase in dopamine neurotransmitter, up-regulation
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/328/5983/1288/dc1 Supporting Online Material for Induction of Fear Extinction with Hippocampal-Infralimbic BDNF Jamie Peters, Laura M. Dieppa-Perea, Loyda M. Melendez,
www.sciencemag.org/cgi/content/full/328/5983/1288/dc1 Supporting Online Material for Induction of Fear Extinction with Hippocampal-Infralimbic BDNF Jamie Peters, Laura M. Dieppa-Perea, Loyda M. Melendez,
Basic definition and Classification of Anhedonia. Preclinical and Clinical assessment of anhedonia.
 Basic definition and Classification of Anhedonia. Preclinical and Clinical assessment of anhedonia. Neurobiological basis and pathways involved in anhedonia. Objective characterization and computational
Basic definition and Classification of Anhedonia. Preclinical and Clinical assessment of anhedonia. Neurobiological basis and pathways involved in anhedonia. Objective characterization and computational
Basolateral amygdala lesions in the rat produce an abnormally persistent latent inhibition with weak preexposure but not with context shift
 Behavioural Brain Research 163 (2005) 115 121 Research report Basolateral amygdala lesions in the rat produce an abnormally persistent latent inhibition with weak preexposure but not with context shift
Behavioural Brain Research 163 (2005) 115 121 Research report Basolateral amygdala lesions in the rat produce an abnormally persistent latent inhibition with weak preexposure but not with context shift
Impaired Outcome-Specific Devaluation of Instrumental Responding in Mice with a Targeted Deletion of the AMPA Receptor Glutamate Receptor 1 Subunit
 The Journal of Neuroscience, March 2, 2005 25(9):2359 2365 2359 Behavioral/Systems/Cognitive Impaired Outcome-Specific Devaluation of Instrumental Responding in Mice with a Targeted Deletion of the AMPA
The Journal of Neuroscience, March 2, 2005 25(9):2359 2365 2359 Behavioral/Systems/Cognitive Impaired Outcome-Specific Devaluation of Instrumental Responding in Mice with a Targeted Deletion of the AMPA
Thalamic Cortical Striatal Circuitry Subserves Working Memory during Delayed Responding on a Radial Arm Maze
 The Journal of Neuroscience, December 15, 1999, 19(24):11061 11071 Thalamic Cortical Striatal Circuitry Subserves Working Memory during Delayed Responding on a Radial Arm Maze Stan B. Floresco, Deanna
The Journal of Neuroscience, December 15, 1999, 19(24):11061 11071 Thalamic Cortical Striatal Circuitry Subserves Working Memory during Delayed Responding on a Radial Arm Maze Stan B. Floresco, Deanna
Still at the Choice-Point
 Still at the Choice-Point Action Selection and Initiation in Instrumental Conditioning BERNARD W. BALLEINE AND SEAN B. OSTLUND Department of Psychology and the Brain Research Institute, University of California,
Still at the Choice-Point Action Selection and Initiation in Instrumental Conditioning BERNARD W. BALLEINE AND SEAN B. OSTLUND Department of Psychology and the Brain Research Institute, University of California,
Behavioural Brain Research
 Behavioural Brain Research 223 (2011) 255 261 Contents lists available at ScienceDirect Behavioural Brain Research j ourna l ho mepage: www.elsevier.com/locate/bbr Research report Rats prone to attribute
Behavioural Brain Research 223 (2011) 255 261 Contents lists available at ScienceDirect Behavioural Brain Research j ourna l ho mepage: www.elsevier.com/locate/bbr Research report Rats prone to attribute
Differential involvement of serotonin and dopamine systems in cost-benefit decisions about delay or effort
 Psychopharmacology (2005) 179: 587 596 DOI 10.1007/s00213-004-2059-4 ORIGINAL INVESTIGATION F. Denk. M. E. Walton. K. A. Jennings. T. Sharp. M. F. S. Rushworth. D. M. Bannerman Differential involvement
Psychopharmacology (2005) 179: 587 596 DOI 10.1007/s00213-004-2059-4 ORIGINAL INVESTIGATION F. Denk. M. E. Walton. K. A. Jennings. T. Sharp. M. F. S. Rushworth. D. M. Bannerman Differential involvement
How cocaine impairs flexible behavior: A neuroscience perspective. By Heather Ortega
 Running head: HOW COCAINE IMPAIRS FLEXIBLE BEHAVIOR How cocaine impairs flexible behavior: A neuroscience perspective By Heather Ortega Senior Honors Thesis Psychology and Neuroscience University of North
Running head: HOW COCAINE IMPAIRS FLEXIBLE BEHAVIOR How cocaine impairs flexible behavior: A neuroscience perspective By Heather Ortega Senior Honors Thesis Psychology and Neuroscience University of North
Selective Ablations Reveal That Orbital and Lateral Prefrontal Cortex Play Different Roles in Estimating Predicted Reward Value
 5878 The Journal of Neuroscience, November 24, 200 30(47):5878 5887 Behavioral/Systems/Cognitive Selective Ablations Reveal That Orbital and Lateral Prefrontal Cortex Play Different Roles in Estimating
5878 The Journal of Neuroscience, November 24, 200 30(47):5878 5887 Behavioral/Systems/Cognitive Selective Ablations Reveal That Orbital and Lateral Prefrontal Cortex Play Different Roles in Estimating
brain valuation & behavior
 brain valuation & behavior 9 Rangel, A, et al. (2008) Nature Neuroscience Reviews Vol 9 Stages in decision making process Problem is represented in the brain Brain evaluates the options Action is selected
brain valuation & behavior 9 Rangel, A, et al. (2008) Nature Neuroscience Reviews Vol 9 Stages in decision making process Problem is represented in the brain Brain evaluates the options Action is selected
Double dissociation of value computations in orbitofrontal and anterior cingulate neurons
 Supplementary Information for: Double dissociation of value computations in orbitofrontal and anterior cingulate neurons Steven W. Kennerley, Timothy E. J. Behrens & Jonathan D. Wallis Content list: Supplementary
Supplementary Information for: Double dissociation of value computations in orbitofrontal and anterior cingulate neurons Steven W. Kennerley, Timothy E. J. Behrens & Jonathan D. Wallis Content list: Supplementary
Introduction to Systems Neuroscience. Nov. 28, The limbic system. Daniel C. Kiper
 Introduction to Systems Neuroscience Nov. 28, 2017 The limbic system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html LIMBIC SYSTEM The term limbic system mean
Introduction to Systems Neuroscience Nov. 28, 2017 The limbic system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html LIMBIC SYSTEM The term limbic system mean
<student name> Undergraduate Research Grant Proposal
 Undergraduate Research Grant Proposal A. Project Description Objective of research: The objective of this study is to determine if hippocampal dopamine D1 receptors facilitate novel object
Undergraduate Research Grant Proposal A. Project Description Objective of research: The objective of this study is to determine if hippocampal dopamine D1 receptors facilitate novel object
Appetitive overshadowing is disrupted by systemic amphetamine but not by electrolytic lesions to the nucleus accumbens shell
 Original Papers Appetitive overshadowing is disrupted by systemic amphetamine but not by electrolytic lesions to the nucleus accumbens shell Journal of Psychopharmacology XX(X) (200X) 1 10 2007 British
Original Papers Appetitive overshadowing is disrupted by systemic amphetamine but not by electrolytic lesions to the nucleus accumbens shell Journal of Psychopharmacology XX(X) (200X) 1 10 2007 British
Supplementary Methods
 1 Supplementary Methods Social Preference Test Subjects Seventy-four Long-Evans, male rats served as subjects (S-rats) in the foodpreference test, with 40 assigned to the CXT-Same (CXT-S) Condition and
1 Supplementary Methods Social Preference Test Subjects Seventy-four Long-Evans, male rats served as subjects (S-rats) in the foodpreference test, with 40 assigned to the CXT-Same (CXT-S) Condition and
590,000 deaths can be attributed to an addictive substance in some way
 Mortality and morbidity attributable to use of addictive substances in the United States. The Association of American Physicians from 1999 60 million tobacco smokers in the U.S. 14 million dependent on
Mortality and morbidity attributable to use of addictive substances in the United States. The Association of American Physicians from 1999 60 million tobacco smokers in the U.S. 14 million dependent on
GUIDANCE OF INSTRUMENTAL BEHAVIOR UNDER REVERSAL CONDITIONS REQUIRES DOPAMINE D1 AND D2 RECEPTOR ACTIVATION IN THE ORBITOFRONTAL CORTEX
 Neuroscience 154 (2008) 1195 1204 GUIDANCE OF INSTRUMENTAL BEHAVIOR UNDER REVERSAL CONDITIONS REQUIRES DOPAMINE D1 AND D2 RECEPTOR ACTIVATION IN THE ORBITOFRONTAL CORTEX C. CALAMINUS AND W. HAUBER* Abteilung
Neuroscience 154 (2008) 1195 1204 GUIDANCE OF INSTRUMENTAL BEHAVIOR UNDER REVERSAL CONDITIONS REQUIRES DOPAMINE D1 AND D2 RECEPTOR ACTIVATION IN THE ORBITOFRONTAL CORTEX C. CALAMINUS AND W. HAUBER* Abteilung
The Neuroscience of Addiction: A mini-review
 The Neuroscience of Addiction: A mini-review Jim Morrill, MD, PhD MGH Charlestown HealthCare Center Massachusetts General Hospital Disclosures Neither I nor my spouse/partner has a relevant financial relationship
The Neuroscience of Addiction: A mini-review Jim Morrill, MD, PhD MGH Charlestown HealthCare Center Massachusetts General Hospital Disclosures Neither I nor my spouse/partner has a relevant financial relationship
Pattern Memory Involves Both Elemental and Configural Processes: Evidence From the Effects of Hippocampal Lesions
 Behavioral Neuroscience 2011 American Psychological Association 2011, Vol. 125, No. 4, 567 577 0735-7044/11/$12.00 DOI: 10.1037/a0023762 Pattern Memory Involves Both Elemental and Configural Processes:
Behavioral Neuroscience 2011 American Psychological Association 2011, Vol. 125, No. 4, 567 577 0735-7044/11/$12.00 DOI: 10.1037/a0023762 Pattern Memory Involves Both Elemental and Configural Processes:
Neural Basis of Decision Making. Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD
 Neural Basis of Decision Making Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD Phineas Gage: Sept. 13, 1848 Working on the rail road Rod impaled his head. 3.5 x 1.25 13 pounds What happened
Neural Basis of Decision Making Mary ET Boyle, Ph.D. Department of Cognitive Science UCSD Phineas Gage: Sept. 13, 1848 Working on the rail road Rod impaled his head. 3.5 x 1.25 13 pounds What happened
Amount of training effects in representationmediated food aversion learning: No evidence of a role for associability changes
 Journal Learning & Behavior 2005,?? 33 (?), (4),???-??? 464-478 Amount of training effects in representationmediated food aversion learning: No evidence of a role for associability changes PETER C. HOLLAND
Journal Learning & Behavior 2005,?? 33 (?), (4),???-??? 464-478 Amount of training effects in representationmediated food aversion learning: No evidence of a role for associability changes PETER C. HOLLAND
BINGES, BLUNTS AND BRAIN DEVELOPMENT
 BINGES, BLUNTS AND BRAIN DEVELOPMENT Why delaying the onset of alcohol and other drug use during adolescence is so important Aaron White, PhD Division of Epidemiology and Prevention Research National Institute
BINGES, BLUNTS AND BRAIN DEVELOPMENT Why delaying the onset of alcohol and other drug use during adolescence is so important Aaron White, PhD Division of Epidemiology and Prevention Research National Institute
Role of Cholinergic Projections to the Orbitofrontal Cortex in Probability Discounting in a Rat Model
 College of William and Mary W&M ScholarWorks Undergraduate Honors Theses Theses, Dissertations, & Master Projects 5-2015 Role of Cholinergic Projections to the Orbitofrontal Cortex in Probability Discounting
College of William and Mary W&M ScholarWorks Undergraduate Honors Theses Theses, Dissertations, & Master Projects 5-2015 Role of Cholinergic Projections to the Orbitofrontal Cortex in Probability Discounting
Encoding Specific Associative Memory: Evidence From Behavioral and Neural Manipulations
 Journal of Experimental Psychology: Animal Behavior Processes 2011, Vol. 37, No. 3, 317 329 2011 American Psychological Association 0097-7403/11/$12.00 DOI: 10.1037/a0022497 Encoding Specific Associative
Journal of Experimental Psychology: Animal Behavior Processes 2011, Vol. 37, No. 3, 317 329 2011 American Psychological Association 0097-7403/11/$12.00 DOI: 10.1037/a0022497 Encoding Specific Associative
Contributions of the prefrontal cortex to the neural basis of human decision making
 Neuroscience and Biobehavioral Reviews 26 (2002) 631 664 Review Contributions of the prefrontal cortex to the neural basis of human decision making Daniel C. Krawczyk* Department of Psychology, University
Neuroscience and Biobehavioral Reviews 26 (2002) 631 664 Review Contributions of the prefrontal cortex to the neural basis of human decision making Daniel C. Krawczyk* Department of Psychology, University
ZNZ Advanced Course in Neuroscience Mon Limbic System II. David P. Wolfer MD
 ZNZ Advanced Course in Neuroscience Mon 05.05.2014 Limbic System II David P. Wolfer MD Institute of Anatomy, University of Zurich Institute for Human Movement Sciences and Sport, ETH Zurich http://www.dpwolfer.ch
ZNZ Advanced Course in Neuroscience Mon 05.05.2014 Limbic System II David P. Wolfer MD Institute of Anatomy, University of Zurich Institute for Human Movement Sciences and Sport, ETH Zurich http://www.dpwolfer.ch
Animal models of gambling. Catharine A. Winstanley University of British Columbia, Canada
 Animal models of gambling Catharine A. Winstanley University of British Columbia, Canada Disclosures Advisory Board: Shire What questions can preclinical research help to answer? Which brain areas are
Animal models of gambling Catharine A. Winstanley University of British Columbia, Canada Disclosures Advisory Board: Shire What questions can preclinical research help to answer? Which brain areas are
Ludis e Málková, 1 David Gaffan, 2 and Elisabeth A. Murray 1
 The Journal of Neuroscience, August 1, 1997, 17(15):6011 6020 Excitotoxic Lesions of the Amygdala Fail to Produce Impairment in Visual Learning for Auditory Secondary Reinforcement But Interfere with Reinforcer
The Journal of Neuroscience, August 1, 1997, 17(15):6011 6020 Excitotoxic Lesions of the Amygdala Fail to Produce Impairment in Visual Learning for Auditory Secondary Reinforcement But Interfere with Reinforcer
Overlapping neural systems mediating extinction, reversal and regulation of fear
 Review Overlapping neural systems mediating extinction, reversal and regulation of fear Daniela Schiller 1,2 and Mauricio R. Delgado 3 1 Center for Neural Science, New York University, New York, NY 10003,
Review Overlapping neural systems mediating extinction, reversal and regulation of fear Daniela Schiller 1,2 and Mauricio R. Delgado 3 1 Center for Neural Science, New York University, New York, NY 10003,
Psychology & Neuroscience ISSN: Pontifícia Universidade Católica do Rio de Janeiro. Brasil
 Psychology & Neuroscience ISSN: 1984-3054 landeira@puc-rio.br Pontifícia Universidade Católica do Rio de Janeiro Brasil de Oliveira Bueno, José Lino; Judice-Daher, Danielle Marcilio; Ferreira Tavares,
Psychology & Neuroscience ISSN: 1984-3054 landeira@puc-rio.br Pontifícia Universidade Católica do Rio de Janeiro Brasil de Oliveira Bueno, José Lino; Judice-Daher, Danielle Marcilio; Ferreira Tavares,
Decision neuroscience seeks neural models for how we identify, evaluate and choose
 VmPFC function: The value proposition Lesley K Fellows and Scott A Huettel Decision neuroscience seeks neural models for how we identify, evaluate and choose options, goals, and actions. These processes
VmPFC function: The value proposition Lesley K Fellows and Scott A Huettel Decision neuroscience seeks neural models for how we identify, evaluate and choose options, goals, and actions. These processes
Emotion I: General concepts, fear and anxiety
 C82NAB Neuroscience and Behaviour Emotion I: General concepts, fear and anxiety Tobias Bast, School of Psychology, University of Nottingham 1 Outline Emotion I (first part) Studying brain substrates of
C82NAB Neuroscience and Behaviour Emotion I: General concepts, fear and anxiety Tobias Bast, School of Psychology, University of Nottingham 1 Outline Emotion I (first part) Studying brain substrates of
Ventral Striatum and Orbitofrontal Cortex Are Both Required for Model-Based, But Not Model-Free, Reinforcement Learning
 27 The Journal of Neuroscience, February 16, 11 31(7):27 275 Behavioral/Systems/Cognitive Ventral Striatum and Orbitofrontal Cortex Are Both Required for Model-Based, But Not Model-Free, Reinforcement
27 The Journal of Neuroscience, February 16, 11 31(7):27 275 Behavioral/Systems/Cognitive Ventral Striatum and Orbitofrontal Cortex Are Both Required for Model-Based, But Not Model-Free, Reinforcement
The primate amygdala combines information about space and value
 The primate amygdala combines information about space and Christopher J Peck 1,8, Brian Lau 1,7,8 & C Daniel Salzman 1 6 npg 213 Nature America, Inc. All rights reserved. A stimulus predicting reinforcement
The primate amygdala combines information about space and Christopher J Peck 1,8, Brian Lau 1,7,8 & C Daniel Salzman 1 6 npg 213 Nature America, Inc. All rights reserved. A stimulus predicting reinforcement
Phasic Accumbal Firing May Contribute to the Regulation of Drug Taking during Intravenous Cocaine Self-administration Sessions
 Phasic Accumbal Firing May Contribute to the Regulation of Drug Taking during Intravenous Cocaine Self-administration Sessions LAURA L. PEOPLES, a ANTHONY J. UZWIAK, FRED GEE, ANTHONY T. FABBRICATORE,
Phasic Accumbal Firing May Contribute to the Regulation of Drug Taking during Intravenous Cocaine Self-administration Sessions LAURA L. PEOPLES, a ANTHONY J. UZWIAK, FRED GEE, ANTHONY T. FABBRICATORE,
Reinforcement learning and the brain: the problems we face all day. Reinforcement Learning in the brain
 Reinforcement learning and the brain: the problems we face all day Reinforcement Learning in the brain Reading: Y Niv, Reinforcement learning in the brain, 2009. Decision making at all levels Reinforcement
Reinforcement learning and the brain: the problems we face all day Reinforcement Learning in the brain Reading: Y Niv, Reinforcement learning in the brain, 2009. Decision making at all levels Reinforcement
NIH Public Access Author Manuscript Nat Neurosci. Author manuscript; available in PMC 2006 September 5.
 NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. 2006 August ; 9(8): 1004 1006. Maternal presence serves as a switch between learning fear and attraction in infancy
NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. 2006 August ; 9(8): 1004 1006. Maternal presence serves as a switch between learning fear and attraction in infancy
THE PERSISTENCE OF MALADAPTIVE MEMORY: ADDICTION, DRUG
 THE PERSISTENCE OF MALADAPTIVE MEMORY: ADDICTION, DRUG MEMORIES AND ANTI-RELAPSE TREATMENTS A.L. Milton 1 & B.J. Everitt 1 1 Behavioural and Clinical Neuroscience Institute, Department of Experimental
THE PERSISTENCE OF MALADAPTIVE MEMORY: ADDICTION, DRUG MEMORIES AND ANTI-RELAPSE TREATMENTS A.L. Milton 1 & B.J. Everitt 1 1 Behavioural and Clinical Neuroscience Institute, Department of Experimental
Hierarchical Control over Effortful Behavior by Dorsal Anterior Cingulate Cortex. Clay Holroyd. Department of Psychology University of Victoria
 Hierarchical Control over Effortful Behavior by Dorsal Anterior Cingulate Cortex Clay Holroyd Department of Psychology University of Victoria Proposal Problem(s) with existing theories of ACC function.
Hierarchical Control over Effortful Behavior by Dorsal Anterior Cingulate Cortex Clay Holroyd Department of Psychology University of Victoria Proposal Problem(s) with existing theories of ACC function.
Systems Neuroscience November 29, Memory
 Systems Neuroscience November 29, 2016 Memory Gabriela Michel http: www.ini.unizh.ch/~kiper/system_neurosci.html Forms of memory Different types of learning & memory rely on different brain structures
Systems Neuroscience November 29, 2016 Memory Gabriela Michel http: www.ini.unizh.ch/~kiper/system_neurosci.html Forms of memory Different types of learning & memory rely on different brain structures
Class 16 Emotions (10/19/17) Chapter 10
 Class 16 Emotions (10/19/17) Chapter 10 Notes By: Rashea Psych 302 10/19/17 Emotions The issues o Innate or learned? o Voluntary or involuntary? (conscious/unconscious) o Adaptive behavior or communication?
Class 16 Emotions (10/19/17) Chapter 10 Notes By: Rashea Psych 302 10/19/17 Emotions The issues o Innate or learned? o Voluntary or involuntary? (conscious/unconscious) o Adaptive behavior or communication?
Double Dissociation between Serotonergic and Dopaminergic Modulation of Medial Prefrontal and Orbitofrontal Cortex during a Test of Impulsive Choice
 Cerebral Cortex Advance Access published April 13, 2005 Cerebral Cortex doi:10.1093/cercor/bhi088 Double Dissociation between Serotonergic and Dopaminergic Modulation of Medial Prefrontal and Orbitofrontal
Cerebral Cortex Advance Access published April 13, 2005 Cerebral Cortex doi:10.1093/cercor/bhi088 Double Dissociation between Serotonergic and Dopaminergic Modulation of Medial Prefrontal and Orbitofrontal
Stimulus control of foodcup approach following fixed ratio reinforcement*
 Animal Learning & Behavior 1974, Vol. 2,No. 2, 148-152 Stimulus control of foodcup approach following fixed ratio reinforcement* RICHARD B. DAY and JOHN R. PLATT McMaster University, Hamilton, Ontario,
Animal Learning & Behavior 1974, Vol. 2,No. 2, 148-152 Stimulus control of foodcup approach following fixed ratio reinforcement* RICHARD B. DAY and JOHN R. PLATT McMaster University, Hamilton, Ontario,
The role of the orbitofrontal cortex in reward related behavior in addiction and alcoholism in particular
 The role of the orbitofrontal cortex in reward related behavior in addiction and alcoholism in particular Thesis Neuroscience and Cognition Rebecca Steketee 0309427 Supervisor: Dr. E.J. van Honk September
The role of the orbitofrontal cortex in reward related behavior in addiction and alcoholism in particular Thesis Neuroscience and Cognition Rebecca Steketee 0309427 Supervisor: Dr. E.J. van Honk September
Associative learning
 Introduction to Learning Associative learning Event-event learning (Pavlovian/classical conditioning) Behavior-event learning (instrumental/ operant conditioning) Both are well-developed experimentally
Introduction to Learning Associative learning Event-event learning (Pavlovian/classical conditioning) Behavior-event learning (instrumental/ operant conditioning) Both are well-developed experimentally
REVIEW Neural and psychological mechanisms underlying compulsive drug seeking habits and drug memories indications for novel treatments of addiction*
 European Journal of Neuroscience, Vol. 40, pp. 2163 2182, 2014 doi:10.1111/ejn.12644 REVIEW Neural and psychological mechanisms underlying compulsive drug seeking habits and drug memories indications for
European Journal of Neuroscience, Vol. 40, pp. 2163 2182, 2014 doi:10.1111/ejn.12644 REVIEW Neural and psychological mechanisms underlying compulsive drug seeking habits and drug memories indications for
Appetitive Pavlovian goal-tracking memories reconsolidate only under specific conditions
 Research Appetitive Pavlovian goal-tracking memories reconsolidate only under specific conditions Amy C. Reichelt 1 and Jonathan L.C. Lee School of Psychology, University of Birmingham, Edgbaston, Birmingham
Research Appetitive Pavlovian goal-tracking memories reconsolidate only under specific conditions Amy C. Reichelt 1 and Jonathan L.C. Lee School of Psychology, University of Birmingham, Edgbaston, Birmingham
JLC Lee: Memory reconsolidation mediates the strengthening of memories by additional learning
 JLC Lee: Memory reconsolidation mediates the strengthening of memories by additional learning 8 6 4 Cond1 Cond2 Test VEH ANI Session Supplementary Fig. 1. Protein synthesis inhibition after a second contextual
JLC Lee: Memory reconsolidation mediates the strengthening of memories by additional learning 8 6 4 Cond1 Cond2 Test VEH ANI Session Supplementary Fig. 1. Protein synthesis inhibition after a second contextual
Memory for Temporal Order of New and Familiar Spatial Location Sequences: Role of the Medial Prefrontal Cortex
 Memory for Temporal Order of New and Familiar Spatial Location Sequences: Role of the Medial Prefrontal Cortex Andrea A. Chiba, Raymond P. Kesner, 1 and Chris J. Gibson Department of Psychology University
Memory for Temporal Order of New and Familiar Spatial Location Sequences: Role of the Medial Prefrontal Cortex Andrea A. Chiba, Raymond P. Kesner, 1 and Chris J. Gibson Department of Psychology University
