Hans Hockey & Kristian Brock Biometrics Matters Ltd, Hamilton, NZ & Cancer Research UK Clinical Trials Unit, Birmingham, UK
|
|
|
- Marvin Austin
- 5 years ago
- Views:
Transcription
1 HOCKEY STICKS AND BROKEN STICKS A DESIGN FOR A SINGLE-TREATMENT, PLACEBO-CONTROLLED, DOUBLE-BLIND, RANDOMIZED CLINICAL TRIAL SUITABLE FOR CHRONIC DISEASES Hans Hockey & Kristian Brock Biometrics Matters Ltd, Hamilton, NZ & Cancer Research UK Clinical Trials Unit, Birmingham, UK hans@biometricsmatters.com AASC, Rotorua, NZ, 5 December
2 HOCKEY STICKS AND BROKEN STICKS A DESIGN FOR A SINGLE-TREATMENT, PLACEBO-CONTROLLED, DOUBLE-BLIND, RANDOMIZED CLINICAL TRIAL SUITABLE FOR CHRONIC DISEASES Hans Hockey & Kristian Brock Biometrics Matters Ltd, Hamilton, NZ & Cancer Research UK Clinical Trials Unit, Birmingham, UK hans@biometricsmatters.com AASC, Rotorua, NZ, 5 December
3 Abstract This work is motivated and exemplified by a genetic disorder causing early onset diabetes, blindness and deafness, which is extremely rare, inevitably fatal and has no current direct treatment. While the standard placebo-controlled RCT is the gold standard required by the regulatory agency for a new proposed drug study, it is conjectured that potential study participants will prefer a design which guarantees that they are always assigned to the drug under study. A design is proposed which meets this patient need and hence probably increases recruitment and compliance. At the same time, it meets the requirement for full randomization. Analyses which follow naturally from this design are also described. If time, comparison with other possible designs will be made, still from the patient perspective. Which would YOU choose? 3
4 Talk outline Motivating example clinical trial Hockey sticks and broken sticks A proposed design Summary 4
5 Talk outline Motivating example clinical trial 5
6 Wolfram Syndrome A 31-year-old woman was diagnosed with type 1 diabetes at age 5, with hypothyroidism at age 16. She developed progressive visual loss at age 19 and progressive hearing loss at age 28. Life expectancy with this disease is about 30 years.
7 The TreatWolfram study Treatment with sodium valproate, an epilepsy drug Double-blind, randomised, placebo-controlled trial International (4 countries) Children and adults Endpoint: Visual acuity (VA) logmar N=70 (2:1) gives 80% power to detect 50% lower rate of progression in VA with mixed model analysis VA will be assessed at baseline and every 6 months t = (0, 0.5, 1.0, 1.5, 2.0, 2.5, 3.0) years 7
8 Visual acuity (VA) log Minimum Angle of Resolution 8
9 Talk outline Motivating example clinical trial Hockey sticks and broken sticks 10
10 What are hockey sticks and broken sticks? 11
11 What are hockey sticks and broken sticks? 12
12 This is not a hockey stick with apologies to René Magritte 13
13 This is the most famous hockey stick? 14
14 This is the most famous hockey stick 15
15 Patient report outcome (PRO) A real hockey stick example Screening Placebo-controlled All on active IV Follow-up EOS Partially controlled design 16
16 Patient report outcome (PRO) A real hockey stick example Screening Placebo-controlled All on active IV Follow-up EOS Partially controlled design 17
17 Talk outline Motivating example clinical trial Hockey sticks and broken sticks A proposed design 18
18 Simulated VA in six patients with Wolfram syndrome Worse Source: Simulation based on parameters from Hershey data. Within patient slope = 8 units/year 20
19 Figure 1: What is the design and what is the model? Based on historical data Shifted VA Year in study
20 Figure 2: What is the current design and what is the model? Shifted VA Year in study
21 Figure 3: What is the proposed design and what is the model? Shifted VA Year in study
22 Figure 4: What is the proposed design and what is the model? Shifted VA Year in study
23 Figure 5: Simulated data shifted to study year over three years Shifted VA Year in study
24 Figure 6: Simulated data shifted to year of treatment with underlying model Shifted VA Year relative to treatment start
25 Figure 7: Simple hockey/broken stick model fit Effect Estimate SE DF t Value Pr > t SlopeChangeYear Shifted VA Year relative to treatment start
26 Figure 8: RC broken stick model with per patient predicted lines (random effect) Effect Estimate SE DF t Value Pr > t SlopeChangeYear Shifted VA Year relative to treatment start
27 Figure 9: RC broken stick model standardized to same initial slope Effect Estimate SE DF t Value Pr > t SlopeChangeYear Shifted VA Year relative to treatment start
28 Figure 10: RC broken stick model standardized to same initial slope, with jitter Effect Estimate SE DF t Value Pr > t SlopeChangeYear Shifted VA Year relative to treatment start
29 Talk outline Motivating example clinical trial Hockey sticks and broken sticks A proposed design Summary 21
30 Consider a hockey stick design when: 22
31 Consider a hockey stick design when: No current treatment exists, but Placebo is unethical, or having no placebo encourages recruitment Randomization is mandatory Meets patient preference Study is longitudinal, because Disease is chronic, long-term Response is continuous, not greatly variable Drug effect is more rapid than size of gap (Available and valid data history helps) 23
32 Simulations? Why? 24
33 Simulations? Why? Within-patient studies should be more powerful than between-patient (and more informative of mechanism) Realistic differential dropout simulation must favour hockey stick design Equal replication of two treatments being compared is more powerful than unequal replication... 25
34 Three patients on either design (9 years total) 1:2 (3:6) 1:1 (4.5:4.5) 26
35 Thank you for your attention! 27
36 QUESTION: Is adherence to placebo control sometimes doing a disservice to both current and future patients? Hans Hockey Biometrics Matters Ltd, Hamilton, New Zealand hans@biometricsmatters.com 3 rd EFSPI Workshop on Regulatory Statistics Basel, 25 September 2018
37 VA Current RCT design Time in study
38 VA Hockey stick design Time in study
39 VA Partially controlled design Time in study
40 VA Single arm study Time in study
41
42 Historical Placebo Drug
43
44 1:2 N = 72 Placebo effect lowers control treatment Historical Placebo Drug Active treatment effect as expected Trial non-significant Lower total N = 60 Result exactly as was powered for. Significant
45 Single arm study VA Time in study
46 Single arm study VA Time in study
47 Thank you for your continued attention! 41
48 Thank you for your continued attention! This presentation has been brought to you by: 42
Some Thoughts on Estimands in a Chronic Pain Indication
 Some Thoughts on Estimands in a Chronic Pain Indication 1 st EFSPI-Workshop on Regulatory Statistics 12 SEP 2016 / PD Dr. Christoph Gerlinger; Dr. Michael Kunz / Basel Agenda Medical Background Problem
Some Thoughts on Estimands in a Chronic Pain Indication 1 st EFSPI-Workshop on Regulatory Statistics 12 SEP 2016 / PD Dr. Christoph Gerlinger; Dr. Michael Kunz / Basel Agenda Medical Background Problem
Conducting and managing randomised controlled trials (RCTs)
 Conducting and managing randomised controlled trials (RCTs) Introduction Study Design We often wish to investigate the efficacy of new treatments and interventions on patient outcomes In this session,
Conducting and managing randomised controlled trials (RCTs) Introduction Study Design We often wish to investigate the efficacy of new treatments and interventions on patient outcomes In this session,
Evidence Based Practice
 Evidence Based Practice RCS 6740 7/26/04 Evidence Based Practice: Definitions Evidence based practice is the integration of the best available external clinical evidence from systematic research with individual
Evidence Based Practice RCS 6740 7/26/04 Evidence Based Practice: Definitions Evidence based practice is the integration of the best available external clinical evidence from systematic research with individual
Treating an ultra-rare condition: clinician-led repurposing for Wolfram syndrome
 Treating an ultra-rare condition: clinician-led repurposing for Wolfram syndrome Timothy Barrett University of Birmingham Birmingham, UK Findacure scientific conference London 2017 t.g.barrett@bham.ac.uk
Treating an ultra-rare condition: clinician-led repurposing for Wolfram syndrome Timothy Barrett University of Birmingham Birmingham, UK Findacure scientific conference London 2017 t.g.barrett@bham.ac.uk
Statistics for Clinical Trials: Basics of Phase III Trial Design
 Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
CHAMP STUDY SIMULATIONS: PICK THE WINNER. Elizabeth Zahn 19 May 2014
 CHAMP STUDY SIMULATIONS: PICK THE WINNER Elizabeth Zahn 19 May 2014 2 Outline Introduction Design Endpoints Pick the Winner Simulation Results 3 CHAMP Study: Introduction CHAMP = Childhood and Adolescent
CHAMP STUDY SIMULATIONS: PICK THE WINNER Elizabeth Zahn 19 May 2014 2 Outline Introduction Design Endpoints Pick the Winner Simulation Results 3 CHAMP Study: Introduction CHAMP = Childhood and Adolescent
Cost-utility of routine cataract surgery Rasanen P, Krootila K, Sintonen H, Leivo T, Koivisto A M, Ryynanen O P, Blom M, Roine R P
 Cost-utility of routine cataract surgery Rasanen P, Krootila K, Sintonen H, Leivo T, Koivisto A M, Ryynanen O P, Blom M, Roine R P Record Status This is a critical abstract of an economic evaluation that
Cost-utility of routine cataract surgery Rasanen P, Krootila K, Sintonen H, Leivo T, Koivisto A M, Ryynanen O P, Blom M, Roine R P Record Status This is a critical abstract of an economic evaluation that
Efficient analysis of ordinal functional outcome scales
 Efficient analysis of ordinal functional outcome scales Gordon D Murray University of Edinburgh Outline of presentation Functional outcome scales Ordinal analysis Case study: SCAST Results Points to consider
Efficient analysis of ordinal functional outcome scales Gordon D Murray University of Edinburgh Outline of presentation Functional outcome scales Ordinal analysis Case study: SCAST Results Points to consider
Quantifying the clinical measure of interest in the presence of missing data:
 Quantifying the clinical measure of interest in the presence of missing data: choosing primary and sensitivity analyses in neuroscience clinical trials Sept 26, 2016 Elena Polverejan, Ph.D. Statistical
Quantifying the clinical measure of interest in the presence of missing data: choosing primary and sensitivity analyses in neuroscience clinical trials Sept 26, 2016 Elena Polverejan, Ph.D. Statistical
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
Practical and ethical advantages of Bayesian approaches in adaptive clinical trial designs. Kristian Thorlund
 Practical and ethical advantages of Bayesian approaches in adaptive clinical trial designs Kristian Thorlund Background This talk was previously given as an invited talk at a DSEN sponsored meeting on
Practical and ethical advantages of Bayesian approaches in adaptive clinical trial designs Kristian Thorlund Background This talk was previously given as an invited talk at a DSEN sponsored meeting on
AS 3.12, Externally assessed, 4 credits Experiments and Observational Studies
 AS 3.12, Externally assessed, 4 credits Experiments and Observational Studies Do drugs have an adverse effect on teenage brains? Does listening to music help students study? What information would you
AS 3.12, Externally assessed, 4 credits Experiments and Observational Studies Do drugs have an adverse effect on teenage brains? Does listening to music help students study? What information would you
The Non-inferiority Margin in Diabetes Trials
 The non-inferioirty margin in diabetes trials 20NOV2008 The Non-inferiority Margin in Diabetes Trials Lars Endahl Biostatistics, Novo Nordisk The non-inferioirty margin in diabetes trials 20NOV2008 Slide
The non-inferioirty margin in diabetes trials 20NOV2008 The Non-inferiority Margin in Diabetes Trials Lars Endahl Biostatistics, Novo Nordisk The non-inferioirty margin in diabetes trials 20NOV2008 Slide
When a Threshold Crossing approach may and may not be appropriate: A Case Study in SMA
 When a Threshold Crossing approach may and may not be appropriate: A Case Study in SMA EFSPI Regulatory Statistics Workshop, 24-25 th September 2018 Carol Reid and Uli Burger picture placeholder Outline
When a Threshold Crossing approach may and may not be appropriate: A Case Study in SMA EFSPI Regulatory Statistics Workshop, 24-25 th September 2018 Carol Reid and Uli Burger picture placeholder Outline
Randomization: Too Important to Gamble with
 Randomization: Too Important to Gamble with A Presentation for the Delaware Chapter of the ASA Oct 18, 2012 Dennis Sweitzer, Ph.D., Principal Biostatistician Medidata Randomization Center of Excellence
Randomization: Too Important to Gamble with A Presentation for the Delaware Chapter of the ASA Oct 18, 2012 Dennis Sweitzer, Ph.D., Principal Biostatistician Medidata Randomization Center of Excellence
Implementation of estimands in Novo Nordisk
 Implementation of estimands in Novo Nordisk Søren Andersen Helle Lynggaard Biostatistics, Novo Nordisk A/S DSBS meeting 26 October 2017 2 Agenda Overview of implementation process Cross-functional working
Implementation of estimands in Novo Nordisk Søren Andersen Helle Lynggaard Biostatistics, Novo Nordisk A/S DSBS meeting 26 October 2017 2 Agenda Overview of implementation process Cross-functional working
EU regulatory concerns about missing data: Issues of interpretation and considerations for addressing the problem. Prof. Dr.
 EU regulatory concerns about missing data: Issues of interpretation and considerations for addressing the problem Prof. Dr. Karl Broich Disclaimer No conflicts of interest Views expressed in this presentation
EU regulatory concerns about missing data: Issues of interpretation and considerations for addressing the problem Prof. Dr. Karl Broich Disclaimer No conflicts of interest Views expressed in this presentation
Study Designs in Epidemiology
 Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan Epidemiologic Study
Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan Epidemiologic Study
Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials
 Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials Dominik Heinzmann, PhD Global Development Team Leader HER2 Associate Director
Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials Dominik Heinzmann, PhD Global Development Team Leader HER2 Associate Director
Suicidal Ideation & Behavior Discussion. Roger E. Meyer, MD Professor of Psychiatry Penn State Hershey Medical Center
 Suicidal Ideation & Behavior Discussion Roger E. Meyer, MD Professor of Psychiatry Penn State Hershey Medical Center FDA Guidance Documents 2010 & 2012 2010 Suicidal ideation 2012 Passive Suicidal ideation
Suicidal Ideation & Behavior Discussion Roger E. Meyer, MD Professor of Psychiatry Penn State Hershey Medical Center FDA Guidance Documents 2010 & 2012 2010 Suicidal ideation 2012 Passive Suicidal ideation
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON MISSING DATA
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 15 November 2001 CPMP/EWP/1776/99 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 15 November 2001 CPMP/EWP/1776/99 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO
Issues regarding non-inferiority within the anti-bacterials area. Jon Armstrong, AstraZeneca Pharmaceuticals
 Issues regarding non-inferiority within the anti-bacterials area Jon Armstrong, AstraZeneca Pharmaceuticals Topics Covered Disease area background The need for non-inferiority studies for anti-bacterials
Issues regarding non-inferiority within the anti-bacterials area Jon Armstrong, AstraZeneca Pharmaceuticals Topics Covered Disease area background The need for non-inferiority studies for anti-bacterials
Choosing the right study design
 Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Study design continued: intervention studies. Outline. Repetition principal approaches. Gustaf Edgren, PhD Karolinska Institutet.
 Study design continued: intervention studies Gustaf Edgren, PhD Karolinska Institutet Background Outline Uncontrolled trials Non randomized, controlled trials Randomized controlled trials (with an overview
Study design continued: intervention studies Gustaf Edgren, PhD Karolinska Institutet Background Outline Uncontrolled trials Non randomized, controlled trials Randomized controlled trials (with an overview
GATE: Graphic Appraisal Tool for Epidemiology picture, 2 formulas & 3 acronyms
 GATE: Graphic Appraisal Tool for Epidemiology 1991-2015 1 picture, 2 formulas & 3 acronyms 1 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach To
GATE: Graphic Appraisal Tool for Epidemiology 1991-2015 1 picture, 2 formulas & 3 acronyms 1 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach To
Prof. Armando Torres Nephrology Section Hospital Universitario de Canarias University of La Laguna Tenerife, Canary Islands, Spain.
 Does RAS blockade improve outcomes after kidney transplantation? Armando Torres, La Laguna, Spain Chairs: Hans De Fijter, Leiden, The Netherlands Armando Torres, La Laguna, Spain Prof. Armando Torres Nephrology
Does RAS blockade improve outcomes after kidney transplantation? Armando Torres, La Laguna, Spain Chairs: Hans De Fijter, Leiden, The Netherlands Armando Torres, La Laguna, Spain Prof. Armando Torres Nephrology
Research Design & Protocol Development
 Research Design & Protocol Development Paul J Kim, DPM, MS, FACFAS Associate Professor Director of Research Definitions Randomization Sampling Allocation Variables Independent Variable Treatment, intervention,
Research Design & Protocol Development Paul J Kim, DPM, MS, FACFAS Associate Professor Director of Research Definitions Randomization Sampling Allocation Variables Independent Variable Treatment, intervention,
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials
 In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
Getting ready for propensity score methods: Designing non-experimental studies and selecting comparison groups
 Getting ready for propensity score methods: Designing non-experimental studies and selecting comparison groups Elizabeth A. Stuart Johns Hopkins Bloomberg School of Public Health Departments of Mental
Getting ready for propensity score methods: Designing non-experimental studies and selecting comparison groups Elizabeth A. Stuart Johns Hopkins Bloomberg School of Public Health Departments of Mental
The comparison or control group may be allocated a placebo intervention, an alternative real intervention or no intervention at all.
 1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs
 Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs David Manner, Brenda Crowe and Linda Shurzinske BASS XVI November 9-13, 2009 Acknowledgements
Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs David Manner, Brenda Crowe and Linda Shurzinske BASS XVI November 9-13, 2009 Acknowledgements
Design and analysis of clinical trials
 Design and analysis of clinical trials Lecture 3 1.Basic Design Considerations 2.Sample Size determinationa 3.Randomization 2018-01-31 Previous Lectures Definition of a clinical trial The drug development
Design and analysis of clinical trials Lecture 3 1.Basic Design Considerations 2.Sample Size determinationa 3.Randomization 2018-01-31 Previous Lectures Definition of a clinical trial The drug development
The AB of Random Effects Models
 The AB of Random Effects Models Rick Chappell, Ph.D. Professor, Department of Biostatistics and Medical Informatics University of Wisconsin Madison ADC Meetings, Baltimore, 10/2016 Outline A. Linear Models
The AB of Random Effects Models Rick Chappell, Ph.D. Professor, Department of Biostatistics and Medical Informatics University of Wisconsin Madison ADC Meetings, Baltimore, 10/2016 Outline A. Linear Models
Statistical challenges of meta-analyses of randomised clinical trials in a regulatory setting
 Statistical challenges of meta-analyses of randomised clinical trials in a regulatory setting Frank Pétavy ISCTM 14th Annual Scientific Meeting, Washington D.C. Presented by Frank Pétavy on 21 February
Statistical challenges of meta-analyses of randomised clinical trials in a regulatory setting Frank Pétavy ISCTM 14th Annual Scientific Meeting, Washington D.C. Presented by Frank Pétavy on 21 February
Possibilities and risks associated with directly moving from Phase I to Phase III: Adaptive designs and Co
 Possibilities and risks associated with directly moving from Phase I to Phase III: Adaptive designs and Co Dominik Heinzmann, PhD Global Development Team Leader Breast/Gyn Cancer Associate Director Biostatistics,
Possibilities and risks associated with directly moving from Phase I to Phase III: Adaptive designs and Co Dominik Heinzmann, PhD Global Development Team Leader Breast/Gyn Cancer Associate Director Biostatistics,
Lecture 5 Conducting Interviews and Focus Groups
 Lecture 5 Conducting Interviews and Focus Groups Talking to participants enables in-depth information about the experiences of health and illness; and of factors that influence health and illness behaviour
Lecture 5 Conducting Interviews and Focus Groups Talking to participants enables in-depth information about the experiences of health and illness; and of factors that influence health and illness behaviour
Levels of Evaluation. Question. Example of Method Does the program. Focus groups with make sense? target population. implemented as intended?
 Program Evaluation Jonathan Howland, PhD, MPH 1 Formative Process Level Outcome Levels of Evaluation Question Example of Method Does the program Focus groups with make sense? target population. Was the
Program Evaluation Jonathan Howland, PhD, MPH 1 Formative Process Level Outcome Levels of Evaluation Question Example of Method Does the program Focus groups with make sense? target population. Was the
Authors Introduction to Pragmatic Trials. and Stepped Wedge Designs. Sandra Eldridge
 Title 2 nd KCE TRIALS SYMPOSIUM 28 th November 2017 Authors Introduction to Pragmatic Trials Including Centre for Cluster Care & Randomised Public Health Trials Blizard Institute and Stepped Wedge Designs
Title 2 nd KCE TRIALS SYMPOSIUM 28 th November 2017 Authors Introduction to Pragmatic Trials Including Centre for Cluster Care & Randomised Public Health Trials Blizard Institute and Stepped Wedge Designs
Improving trial methodology: Examples from epilepsy. Tony Marson University of Liverpool
 Improving trial methodology: Examples from epilepsy Tony Marson University of Liverpool This talk Examples of trial methodology research Focus on epilepsy but Examples are relevant to any field Epilepsy
Improving trial methodology: Examples from epilepsy Tony Marson University of Liverpool This talk Examples of trial methodology research Focus on epilepsy but Examples are relevant to any field Epilepsy
Statistical Challenges for Novel Technologies. Roseann M. White, MA Director of Pragmatic Clinical Trial Statistics Duke Clinical Research Institute
 Statistical Challenges for Novel Technologies Roseann M. White, MA Director of Pragmatic Clinical Trial Statistics Duke Clinical Research Institute ISCTM 2016 Autumn Conference, Philadelphia, PA 27 September
Statistical Challenges for Novel Technologies Roseann M. White, MA Director of Pragmatic Clinical Trial Statistics Duke Clinical Research Institute ISCTM 2016 Autumn Conference, Philadelphia, PA 27 September
Adjusting overall survival for treatment switch
 Adjusting overall survival for treatment switch Recommendations of a crossinstitutional statistical working group Claire Watkins Director and Consultant Statistician, Clarostat Consulting Ltd Cytel East
Adjusting overall survival for treatment switch Recommendations of a crossinstitutional statistical working group Claire Watkins Director and Consultant Statistician, Clarostat Consulting Ltd Cytel East
What You Need to Know About Clinical Trials A Guide for People With Cancer
 Developed by Pfizer in partnership with the Colon Cancer Alliance. What You Need to Know About Clinical Trials A Guide for People With Cancer THE VOICE OF SURVIVORS Why You Need to Know About Clinical
Developed by Pfizer in partnership with the Colon Cancer Alliance. What You Need to Know About Clinical Trials A Guide for People With Cancer THE VOICE OF SURVIVORS Why You Need to Know About Clinical
CRITICAL APPRAISAL OF MEDICAL LITERATURE. Samuel Iff ISPM Bern
 CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
Sequence balance minimisation: minimising with unequal treatment allocations
 Madurasinghe Trials (2017) 18:207 DOI 10.1186/s13063-017-1942-3 METHODOLOGY Open Access Sequence balance minimisation: minimising with unequal treatment allocations Vichithranie W. Madurasinghe Abstract
Madurasinghe Trials (2017) 18:207 DOI 10.1186/s13063-017-1942-3 METHODOLOGY Open Access Sequence balance minimisation: minimising with unequal treatment allocations Vichithranie W. Madurasinghe Abstract
BAYESIAN JOINT LONGITUDINAL-DISCRETE TIME SURVIVAL MODELS: EVALUATING BIOPSY PROTOCOLS IN ACTIVE-SURVEILLANCE STUDIES
 BAYESIAN JOINT LONGITUDINAL-DISCRETE TIME SURVIVAL MODELS: EVALUATING BIOPSY PROTOCOLS IN ACTIVE-SURVEILLANCE STUDIES Lurdes Y. T. Inoue, PhD Professor Department of Biostatistics University of Washington
BAYESIAN JOINT LONGITUDINAL-DISCRETE TIME SURVIVAL MODELS: EVALUATING BIOPSY PROTOCOLS IN ACTIVE-SURVEILLANCE STUDIES Lurdes Y. T. Inoue, PhD Professor Department of Biostatistics University of Washington
Addressing multiple treatments II: multiple-treatments meta-analysis basic methods
 Deborah Caldwell and Georgia Salanti d.m.caldwell@bristol.ac.uk Addressing multiple treatments II: multiple-treatments meta-analysis basic methods Keystone, 2010 18 th Cochrane Colloquium, 18 th to 22
Deborah Caldwell and Georgia Salanti d.m.caldwell@bristol.ac.uk Addressing multiple treatments II: multiple-treatments meta-analysis basic methods Keystone, 2010 18 th Cochrane Colloquium, 18 th to 22
Dynamic borrowing of historical data: Performance and comparison of existing methods based on a case study
 Introduction Methods Simulations Discussion Dynamic borrowing of historical data: Performance and comparison of existing methods based on a case study D. Dejardin 1, P. Delmar 1, K. Patel 1, C. Warne 1,
Introduction Methods Simulations Discussion Dynamic borrowing of historical data: Performance and comparison of existing methods based on a case study D. Dejardin 1, P. Delmar 1, K. Patel 1, C. Warne 1,
Placebo and Belief Effects: Optimal Design for Randomized Trials
 Placebo and Belief Effects: Optimal Design for Randomized Trials Scott Ogawa & Ken Onishi 2 Department of Economics Northwestern University Abstract The mere possibility of receiving a placebo during a
Placebo and Belief Effects: Optimal Design for Randomized Trials Scott Ogawa & Ken Onishi 2 Department of Economics Northwestern University Abstract The mere possibility of receiving a placebo during a
Dichotomizing partial compliance and increased participant burden in factorial designs: the performance of four noncompliance methods
 Merrill and McClure Trials (2015) 16:523 DOI 1186/s13063-015-1044-z TRIALS RESEARCH Open Access Dichotomizing partial compliance and increased participant burden in factorial designs: the performance of
Merrill and McClure Trials (2015) 16:523 DOI 1186/s13063-015-1044-z TRIALS RESEARCH Open Access Dichotomizing partial compliance and increased participant burden in factorial designs: the performance of
Challenges in the Design and Analysis of Non-Inferiority Trials: A Case Study. Key Points from FDA Guidance
 Challenges in the Design and Analysis of Non-Inferiority Trials: A Case Study Valerie Durkalski NETT Statistical and Data Management Center (NINDS U01 NS059041) Division of Biostatistics & Epidemiology
Challenges in the Design and Analysis of Non-Inferiority Trials: A Case Study Valerie Durkalski NETT Statistical and Data Management Center (NINDS U01 NS059041) Division of Biostatistics & Epidemiology
The Cardiac Arrythmia Suppression Trial
 The Cardiac Arrythmia Suppression Trial 1 The Cardiac Arrythmia Suppression Trial Background and significance Methods Statistical Analysis Results Discussion Summary on Therapy 2 Background and significance
The Cardiac Arrythmia Suppression Trial 1 The Cardiac Arrythmia Suppression Trial Background and significance Methods Statistical Analysis Results Discussion Summary on Therapy 2 Background and significance
Matching: Observational research
 Matching: Observational research 1 When is it deemed acceptable to observe without consent? In a public place where participants would normally expect to be observed by strangers These factors should always
Matching: Observational research 1 When is it deemed acceptable to observe without consent? In a public place where participants would normally expect to be observed by strangers These factors should always
The table below shows the density and velocity of waves in two different substances. Density / kg m 3 Velocity / m s 1
 Q1.(a) When ultrasound is incident at an interface between two different media some energy is transmitted and some is reflected. The ratio of the reflected energy intensity I r to the incident energy intensity
Q1.(a) When ultrasound is incident at an interface between two different media some energy is transmitted and some is reflected. The ratio of the reflected energy intensity I r to the incident energy intensity
Real World Evidence in the Treatment of Ovarian Cancer. Elizabeth Eisenhauer MD FRCPC Queen s University
 Real World Evidence in the Treatment of Ovarian Cancer Elizabeth Eisenhauer MD FRCPC Queen s University Outline What is real world evidence (RWE) and how can it shape policy and practice? In ovarian cancer,
Real World Evidence in the Treatment of Ovarian Cancer Elizabeth Eisenhauer MD FRCPC Queen s University Outline What is real world evidence (RWE) and how can it shape policy and practice? In ovarian cancer,
Estimands and Their Role in Clinical Trials
 Estimands and Their Role in Clinical Trials Frank Bretz (Novartis) EFSPI/PSI UK September 28, 2015 Acknowledgements ICH E9(R1) Expert Working Group M Akacha, D Ohlssen, H Schmidli, G Rosenkranz (Novartis)
Estimands and Their Role in Clinical Trials Frank Bretz (Novartis) EFSPI/PSI UK September 28, 2015 Acknowledgements ICH E9(R1) Expert Working Group M Akacha, D Ohlssen, H Schmidli, G Rosenkranz (Novartis)
Guidance Document for Claims Based on Non-Inferiority Trials
 Guidance Document for Claims Based on Non-Inferiority Trials February 2013 1 Non-Inferiority Trials Checklist Item No Checklist Item (clients can use this tool to help make decisions regarding use of non-inferiority
Guidance Document for Claims Based on Non-Inferiority Trials February 2013 1 Non-Inferiority Trials Checklist Item No Checklist Item (clients can use this tool to help make decisions regarding use of non-inferiority
RANDOMIZED CLINICAL TRIALS. Study Designs. Randomized Clinical Trials (RCT) ASSIGN EXPOSURE Follow up Check for OUTCOME. Experimental studies
 RANDOMIZED CLINICAL TRIALS Study Designs Observational studies Analytic studies Descriptive studies Randomized Controlled trials Case control Cohort Cross sectional Randomized Clinical Trials (RCT) Assign
RANDOMIZED CLINICAL TRIALS Study Designs Observational studies Analytic studies Descriptive studies Randomized Controlled trials Case control Cohort Cross sectional Randomized Clinical Trials (RCT) Assign
Methodological aspects of non-inferiority and equivalence trials
 Methodological aspects of non-inferiority and equivalence trials Department of Clinical Epidemiology Leiden University Medical Center Capita Selecta 09-12-2014 1 Why RCTs Early 1900: Evidence based on
Methodological aspects of non-inferiority and equivalence trials Department of Clinical Epidemiology Leiden University Medical Center Capita Selecta 09-12-2014 1 Why RCTs Early 1900: Evidence based on
SHORTENING TIMELINES AND IMPROVING EFFICIENCY IN DRUG DEVELOPMENT -Seamless drug development paradigms
 SHORTENING TIMELINES AND IMPROVING EFFICIENCY IN DRUG DEVELOPMENT -Seamless drug development paradigms Claudia Filozof, MD PhD Executive Medical Director Covance Challenges in Clinical Development in NASH
SHORTENING TIMELINES AND IMPROVING EFFICIENCY IN DRUG DEVELOPMENT -Seamless drug development paradigms Claudia Filozof, MD PhD Executive Medical Director Covance Challenges in Clinical Development in NASH
GETTING CLOSE UP & FRIENDLY WITH YOUR QUANTITATIVE DATA. Love your data. Research holds the answers but research often seems to hide the answers!
 T H E I N S T I T U T E O F O P T O M E T RY GETTING CLOSE UP & FRIENDLY WITH YOUR QUANTITATIVE DATA Love your data Prof Bruce Evans BSc (Hons) PhD FCOptom DipCLP DipOrth FAAO FBCLA The Institute of Optometry
T H E I N S T I T U T E O F O P T O M E T RY GETTING CLOSE UP & FRIENDLY WITH YOUR QUANTITATIVE DATA Love your data Prof Bruce Evans BSc (Hons) PhD FCOptom DipCLP DipOrth FAAO FBCLA The Institute of Optometry
How to use prior knowledge and still give new data a chance?
 How to use prior knowledge and still give new data a chance? Kristina Weber1, Rob Hemmings2, Armin Koch 19.12.18 1 now with Roche, 1 2 MHRA, London, UK Part I Background Extrapolation and Bayesian methods:
How to use prior knowledge and still give new data a chance? Kristina Weber1, Rob Hemmings2, Armin Koch 19.12.18 1 now with Roche, 1 2 MHRA, London, UK Part I Background Extrapolation and Bayesian methods:
Disclosure. No relevant financial relationships. Placebo-Controlled Statin Trials
 PREVENTING CARDIOVASCULAR DISEASE IN WOMEN: Current Guidelines for Hypertension, Lipids and Aspirin Disclosure Robert B. Baron, MD MS Professor and Associate Dean UCSF School of Medicine No relevant financial
PREVENTING CARDIOVASCULAR DISEASE IN WOMEN: Current Guidelines for Hypertension, Lipids and Aspirin Disclosure Robert B. Baron, MD MS Professor and Associate Dean UCSF School of Medicine No relevant financial
Interpreting Adult Human Thorough QT Studies: Are They Relevant to Pediatric Safety?
 Interpreting Adult Human Thorough QT Studies: Are They Relevant to Pediatric Safety? Hao Zhu, Ph.D. Scientific Lead, QT-IRT, FDA (Dec 10, 2010) Disclaimer: The views expressed in this talk are those of
Interpreting Adult Human Thorough QT Studies: Are They Relevant to Pediatric Safety? Hao Zhu, Ph.D. Scientific Lead, QT-IRT, FDA (Dec 10, 2010) Disclaimer: The views expressed in this talk are those of
Department of OUTCOMES RESEARCH
 Department of OUTCOMES RESEARCH Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland
Department of OUTCOMES RESEARCH Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland
HIERARCHICAL MODELS A framework for evidence synthesis
 HIERARCHICAL MODELS A framework for evidence synthesis Innovative Methodology for Small Populations Research (InSPiRe), WP4 Evidence synthesis Tim Friede Dept. Medical Statistics University Medical Center
HIERARCHICAL MODELS A framework for evidence synthesis Innovative Methodology for Small Populations Research (InSPiRe), WP4 Evidence synthesis Tim Friede Dept. Medical Statistics University Medical Center
Bayesian Joint Modelling of Benefit and Risk in Drug Development
 Bayesian Joint Modelling of Benefit and Risk in Drug Development EFSPI/PSDM Safety Statistics Meeting Leiden 2017 Disclosure is an employee and shareholder of GSK Data presented is based on human research
Bayesian Joint Modelling of Benefit and Risk in Drug Development EFSPI/PSDM Safety Statistics Meeting Leiden 2017 Disclosure is an employee and shareholder of GSK Data presented is based on human research
Sample-size re-estimation in Multiple Sclerosis trials
 Novartis Basel, Switzerland Sample-size re-estimation in Multiple Sclerosis trials Heinz Schmidli PSI Meeting on Sample Size Re-Estimation London, November 2, 2016 Outline Multiple Sclerosis Sample size
Novartis Basel, Switzerland Sample-size re-estimation in Multiple Sclerosis trials Heinz Schmidli PSI Meeting on Sample Size Re-Estimation London, November 2, 2016 Outline Multiple Sclerosis Sample size
A FRAMEWORK FOR SIMULATIONS IN CLINICAL RESEARCH
 A FRAMEWORK FOR SIMULATIONS IN CLINICAL RESEARCH with applications in small populations and rare diseases Tim Friede 1, Norbert Benda 2 1 University Medical Center Göttingen, Göttingen, Germany 2 Federal
A FRAMEWORK FOR SIMULATIONS IN CLINICAL RESEARCH with applications in small populations and rare diseases Tim Friede 1, Norbert Benda 2 1 University Medical Center Göttingen, Göttingen, Germany 2 Federal
Ben Cipollini & Garrison Cottrell
 COGSCI 2014 Ben Cipollini & Garrison Cottrell UC San1 Diego July 25, 2014. A Developmental Model of Hemispheric Asymmetry of Spatial Frequencies COGSCI 2014 Ben Cipollini & Garrison Cottrell UC San2 Diego
COGSCI 2014 Ben Cipollini & Garrison Cottrell UC San1 Diego July 25, 2014. A Developmental Model of Hemispheric Asymmetry of Spatial Frequencies COGSCI 2014 Ben Cipollini & Garrison Cottrell UC San2 Diego
Heart rate lowering treatment in chronic heart failure
 Heart rate lowering treatment in chronic heart failure Jaromir Hradec 3rd Dept. Med., Charles Univ., Prague Czech Republic Things we knew, things we did Things we have learnt, things we should do What
Heart rate lowering treatment in chronic heart failure Jaromir Hradec 3rd Dept. Med., Charles Univ., Prague Czech Republic Things we knew, things we did Things we have learnt, things we should do What
1 Executive summary. Background
 1 Executive summary Background Rheumatoid Arthritis (RA) is the most common inflammatory polyarthropathy in the UK affecting between.5% and 1% of the population. The mainstay of RA treatment interventions
1 Executive summary Background Rheumatoid Arthritis (RA) is the most common inflammatory polyarthropathy in the UK affecting between.5% and 1% of the population. The mainstay of RA treatment interventions
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use plondon, 20 February 2003 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) APPENDIX TO THE NOTE FOR
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use plondon, 20 February 2003 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) APPENDIX TO THE NOTE FOR
CLINICIAN INTERVIEW AS i M: When you examine a clinical trial in new- onset epilepsy, how relevant are the results to your daily clinical practice?
 FROM CLINICAL TRIALS TO CLINICAL PRACTICE: TRANSLATING EPILEPSY RESEARCH INTO PATIENT CARE Interview with Jacqueline A. French, MD Dr Jacqueline A. French is a Professor in the Department of Neurology
FROM CLINICAL TRIALS TO CLINICAL PRACTICE: TRANSLATING EPILEPSY RESEARCH INTO PATIENT CARE Interview with Jacqueline A. French, MD Dr Jacqueline A. French is a Professor in the Department of Neurology
Critical appraisal Randomised controlled trial questions. Tipold et al J Vet Pharm Therap Introduction
 Critical appraisal Randomised controlled trial questions Tipold et al J Vet Pharm Therap 2014 Introduction Are the aims clearly stated? to evaluate safety and efficacy of a new oral preparation of imepitoin
Critical appraisal Randomised controlled trial questions Tipold et al J Vet Pharm Therap 2014 Introduction Are the aims clearly stated? to evaluate safety and efficacy of a new oral preparation of imepitoin
Touch Behavior Analysis for Large Screen Smartphones
 Proceedings of the Human Factors and Ergonomics Society 59th Annual Meeting - 2015 1433 Touch Behavior Analysis for Large Screen Smartphones Yu Zhang 1, Bo Ou 1, Qicheng Ding 1, Yiying Yang 2 1 Emerging
Proceedings of the Human Factors and Ergonomics Society 59th Annual Meeting - 2015 1433 Touch Behavior Analysis for Large Screen Smartphones Yu Zhang 1, Bo Ou 1, Qicheng Ding 1, Yiying Yang 2 1 Emerging
Problem Gambling Foundation of New Zealand
 Problem Gambling Foundation of New Zealand Outcome Measures When Results Matter Partners for Change Outcome Management System Laurie Siegel-Woodward and Ifo Fa anana Problem Gambling Foundation of New
Problem Gambling Foundation of New Zealand Outcome Measures When Results Matter Partners for Change Outcome Management System Laurie Siegel-Woodward and Ifo Fa anana Problem Gambling Foundation of New
The Practice of Statistics 1 Week 2: Relationships and Data Collection
 The Practice of Statistics 1 Week 2: Relationships and Data Collection Video 12: Data Collection - Experiments Experiments are the gold standard since they allow us to make causal conclusions. example,
The Practice of Statistics 1 Week 2: Relationships and Data Collection Video 12: Data Collection - Experiments Experiments are the gold standard since they allow us to make causal conclusions. example,
depression in England: CLAHRC findings on current performance &
 Management of severe and chronic depression in England: CLAHRC findings on current performance & NICE service models for the future Richard Morriss Professor of Psychiatry and Community Mental Health,
Management of severe and chronic depression in England: CLAHRC findings on current performance & NICE service models for the future Richard Morriss Professor of Psychiatry and Community Mental Health,
GATE: Graphic Appraisal Tool for Epidemiology picture, 2 formulas & 3 acronyms
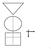 1 GATE: Graphic Appraisal Tool for Epidemiology 1991-2016 1 picture, 2 formulas & 3 acronyms 2 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach
1 GATE: Graphic Appraisal Tool for Epidemiology 1991-2016 1 picture, 2 formulas & 3 acronyms 2 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach
WHAT DOES EVIDENCE-BASED TREATMENT MEAN FOR REALITY-BASED PRACTICE?
 WHAT DOES EVIDENCE-BASED TREATMENT MEAN FOR REALITY-BASED PRACTICE? Miss Information Miss Behaviour Guess what i think of evidencebased treatment! Miss Behaviour Psychologists in clinical practice are
WHAT DOES EVIDENCE-BASED TREATMENT MEAN FOR REALITY-BASED PRACTICE? Miss Information Miss Behaviour Guess what i think of evidencebased treatment! Miss Behaviour Psychologists in clinical practice are
Table of Contents. Preface to the third edition xiii. Preface to the second edition xv. Preface to the fi rst edition xvii. List of abbreviations xix
 Table of Contents Preface to the third edition xiii Preface to the second edition xv Preface to the fi rst edition xvii List of abbreviations xix PART 1 Developing and Validating Instruments for Assessing
Table of Contents Preface to the third edition xiii Preface to the second edition xv Preface to the fi rst edition xvii List of abbreviations xix PART 1 Developing and Validating Instruments for Assessing
Meta-analysis to compare combination therapies: A case study in kidney transplantation
 Meta-analysis to compare combination therapies: A case study in kidney transplantation Steffen Witte, Amy Racine, Heinz Schmidli Novartis Pharma AG, Switzerland, Basel, June 2009 Outline Introduction Methods
Meta-analysis to compare combination therapies: A case study in kidney transplantation Steffen Witte, Amy Racine, Heinz Schmidli Novartis Pharma AG, Switzerland, Basel, June 2009 Outline Introduction Methods
Treatment changes in cancer clinical trials: design and analysis
 Treatment changes in cancer clinical trials: design and analysis Ian White Statistical methods and designs in clinical oncology Paris, 9 th November 2017 Plan 1. Treatment changes
Treatment changes in cancer clinical trials: design and analysis Ian White Statistical methods and designs in clinical oncology Paris, 9 th November 2017 Plan 1. Treatment changes
How to improve the reliability of Single Arm Trials
 Workshop on Single Arm Trials in Oncology Products EMA, London, June 30, 2016 How to improve the reliability of Single Arm Trials Paolo Bruzzi Clinical Epidemiology Unit IRCCS San Martino-IST - National
Workshop on Single Arm Trials in Oncology Products EMA, London, June 30, 2016 How to improve the reliability of Single Arm Trials Paolo Bruzzi Clinical Epidemiology Unit IRCCS San Martino-IST - National
Accessibility for People With Disabilities
 Accessibility for People With Family Day Care Services 2010 A G E N D A Why are we having this training? Accessibility Standards for Customer Service / Ontario Regulation 429/07 Meeting the requirements
Accessibility for People With Family Day Care Services 2010 A G E N D A Why are we having this training? Accessibility Standards for Customer Service / Ontario Regulation 429/07 Meeting the requirements
Case Study in Placebo Modeling and Its Effect on Drug Development
 Case Study in Placebo Modeling and Its Effect on Drug Development Modeling and Simulation Strategy for Phase 3 Dose Selection of Dasotraline in Patients With Attention-Deficit/Hyperactivity Disorder Julie
Case Study in Placebo Modeling and Its Effect on Drug Development Modeling and Simulation Strategy for Phase 3 Dose Selection of Dasotraline in Patients With Attention-Deficit/Hyperactivity Disorder Julie
New Medicines Profile
 New Medicines Profile February 2010 Issue No. 10/02 Eslicarbazepine Concise evaluated information to support the managed entry of new medicines in the NHS Brand Name, (Manufacturer): Zebinix (Eisai Limited)
New Medicines Profile February 2010 Issue No. 10/02 Eslicarbazepine Concise evaluated information to support the managed entry of new medicines in the NHS Brand Name, (Manufacturer): Zebinix (Eisai Limited)
Landmarking, immortal time bias and. Dynamic prediction
 Landmarking and immortal time bias Landmarking and dynamic prediction Discussion Landmarking, immortal time bias and dynamic prediction Department of Medical Statistics and Bioinformatics Leiden University
Landmarking and immortal time bias Landmarking and dynamic prediction Discussion Landmarking, immortal time bias and dynamic prediction Department of Medical Statistics and Bioinformatics Leiden University
Crohn's disease. Appendix I. Clinical Guideline < > 10 October Research recommendations
 Crohn's disease Clinical Guideline < > 10 October 2012 Commissioned by the National Institute for Health and Clinical Excellence Contents Published by the National Clinical Guideline Centre at The Royal
Crohn's disease Clinical Guideline < > 10 October 2012 Commissioned by the National Institute for Health and Clinical Excellence Contents Published by the National Clinical Guideline Centre at The Royal
Challenges in Meeting International Requirements for Clinical Bioequivalence of Inhaled Drug Products
 Challenges in Meeting International Requirements for Clinical Bioequivalence of Inhaled Drug Products Tushar Shah, M.D. Sr. VP, Global Respiratory Research and Development TEVA Pharmaceuticals 1 Presentation
Challenges in Meeting International Requirements for Clinical Bioequivalence of Inhaled Drug Products Tushar Shah, M.D. Sr. VP, Global Respiratory Research and Development TEVA Pharmaceuticals 1 Presentation
A Case Study: Ipilimumab in Pre-treated Metastatic Melanoma
 A Case Study: Ipilimumab in Pre-treated Metastatic Melanoma Tai-Tsang Chen, PhD Global Biometric Sciences, Bristol-Myers Squibb EFSPI Statistical Meeting on Evidence Synthesis Brussels, Belgium November
A Case Study: Ipilimumab in Pre-treated Metastatic Melanoma Tai-Tsang Chen, PhD Global Biometric Sciences, Bristol-Myers Squibb EFSPI Statistical Meeting on Evidence Synthesis Brussels, Belgium November
Treating alcoholrelated. depression. James Foulds Senior Lecturer National Addiction Centre Christchurch, NZ
 Treating alcoholrelated depression James Foulds Senior Lecturer National Addiction Centre Christchurch, NZ Overview of talk Epidemiology and etiology of alcoholrelated comorbidity Alcohol-related depression
Treating alcoholrelated depression James Foulds Senior Lecturer National Addiction Centre Christchurch, NZ Overview of talk Epidemiology and etiology of alcoholrelated comorbidity Alcohol-related depression
Psychology - MR. CALLAWAY Mundy s Mill High School Unit RESEARCH METHODS
 Psychology - MR. CALLAWAY Mundy s Mill High School Unit 2.1 - RESEARCH METHODS Intro to Research How do psychologists ask & answer questions? Differentiate types of research with regard to purpose, strengths,
Psychology - MR. CALLAWAY Mundy s Mill High School Unit 2.1 - RESEARCH METHODS Intro to Research How do psychologists ask & answer questions? Differentiate types of research with regard to purpose, strengths,
Simpson s Paradox and the implications for medical trials
 Simpson s Paradox and the implications for medical trials Norman Fenton, Martin Neil and Anthony Constantinou 31 August 2015 Abstract This paper describes Simpson s paradox, and explains its serious implications
Simpson s Paradox and the implications for medical trials Norman Fenton, Martin Neil and Anthony Constantinou 31 August 2015 Abstract This paper describes Simpson s paradox, and explains its serious implications
What are the regulatory issues. prevention trials?
 What are the regulatory issues that impact endpoints for prevention trials? Ann T. Farrell, M.D. Deputy Division Director Division of Drug Oncology Products Office of New Drugs Center for Drug Evaluation
What are the regulatory issues that impact endpoints for prevention trials? Ann T. Farrell, M.D. Deputy Division Director Division of Drug Oncology Products Office of New Drugs Center for Drug Evaluation
Ureteroscopic treatment of renal stones: Dusting vs Extraction
 Ureteroscopic treatment of renal stones: Dusting vs Extraction Khurshid Ghani Dept of Urology Will Meurer Depts of Emergency Medicine and Neurology University of Michigan Background Use of flexible ureteroscopy
Ureteroscopic treatment of renal stones: Dusting vs Extraction Khurshid Ghani Dept of Urology Will Meurer Depts of Emergency Medicine and Neurology University of Michigan Background Use of flexible ureteroscopy
CHAPTER 6. Experiments in the Real World
 CHAPTER 6 Experiments in the Real World EQUAL TREATMENT FOR ALL SUBJECTS The underlying assumption of randomized comparative experiments is that all subjects are handled equally in every respect except
CHAPTER 6 Experiments in the Real World EQUAL TREATMENT FOR ALL SUBJECTS The underlying assumption of randomized comparative experiments is that all subjects are handled equally in every respect except
Bias in randomised factorial trials
 Research Article Received 17 September 2012, Accepted 9 May 2013 Published online 4 June 2013 in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/sim.5869 Bias in randomised factorial trials
Research Article Received 17 September 2012, Accepted 9 May 2013 Published online 4 June 2013 in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/sim.5869 Bias in randomised factorial trials
Supplementary Online Content
 Supplementary Online Content Gao TY, Guo CX, Babu RJ, et al; the BRAVO Study Team. Effectiveness of a binocular video game vs placebo video game for improving visual functions in older children, teenagers,
Supplementary Online Content Gao TY, Guo CX, Babu RJ, et al; the BRAVO Study Team. Effectiveness of a binocular video game vs placebo video game for improving visual functions in older children, teenagers,
