NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice
|
|
|
- Derek Garrison
- 6 years ago
- Views:
Transcription
1 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice Review of Clinical Guideline (CG85) Glaucoma : Diagnosis and management of chronic open angle glaucoma and ocular hypertension Background information Guideline issue date: year review: 2012 National Collaborating Centre: National Clinical Guidelines Centre for Acute and Chronic Conditions Review recommendation The guideline should be updated at a time which will facilitate a scheduled updating of the quality standard. Factors influencing the decision Literature search 1. Through an assessment of abstracts from a high-level randomised control trial (RCT) search, new evidence was identified relating to the following clinical areas within the guideline: Diagnosis and monitoring of patients with ocular hypertension (OHT), chronic open angled glaucoma (COAG), and suspected COAG Treatment of OHT and suspected COAG and COAG 1 of 54
2 o Pharmacological treatment o Non-pharmacological treatment Complementary and alternative therapies 2. No new evidence was identified in these areas which would invalidate the current guideline recommendations. 3. From initial intelligence gathering, qualitative feedback from other NICE departments, the views expressed by the Guideline Development Group, as well as the high-level RCT indicated that there were no additional clinical areas that required further focused literature searches. 4. Numerous clinical trials (publication dates unknown) were identified focusing on non-pharmacological devices or procedures including: lasers, trabeculoplasty, trabeculotomy, dosing aids, istents, shunts, adherence monitors and diagnosis tools the safety and/or efficacy of pharmacological agents; either in fixed combination, as adjuncts, switching therapies or new agents the GATE study funded by NIHR HTA, which was scheduled to run to April 2012 to compare the diagnostic performance of three automated imaging technologies (the Heidelberg retina tomograph, scanning laser polarimetry and optical coherence tomography) optimal surveillance regimes for individuals with ocular hypertension: modelling and economic evaluation funded by NIHR HTA which is due to publish in However, at this time it is unclear whether the ongoing clinical trials will have any impact on the guideline recommendations in the future. 5. New evidence was identified which directly answered research recommendations in the original guideline relating to: 2 of 54
3 The clinical and cost effectiveness of initial argon, diode or selective laser trabeculoplasty compared with prostaglandin analogues alone or laser trabeculoplasty plus prostaglandin analogues in combination, in people with COAG. Guideline Development Group and National Collaborating Centre perspective 6. A questionnaire was distributed to GDG members and the National Collaborating Centre to consult them on the need for an update of the guideline. Five responses were received with respondents highlighting that there is a growing body of evidence on the relative safety and effectiveness of tube drainage devices compared to trabeculectomy and that trabectome surgery (a minimally invasive surgery) is becoming established in clinical practice. 7. With regard to practices recommended in the current guideline, referral was highlighted as having been an issue at the time of publication which had resulted in volume problems for services. However, respondents reported that there had been activity in design and implementation of referral refinement and shared care programmes with community based optometrists and that waiting times had come down and appropriate monitoring intervals were now being adhered to. In was noted that there had been slight modifications to referral criteria for optometrists published by the College of Optometrists and Ophthalmologists Joint Working Party since the publication of CG85. Respondents indicated that the guideline may require further work relating to the clarity of referral recommendations. 8. GDG members indicated that Xalatan (latanoprost) has come off patent and several other agents (not specified) including combined preparations are available as generics. For CG85 economic modelling was conducted to identify the most cost-effective treatment options for people with OHT and suspected COAG. The cost-utility analyses compared no treatment, medical treatment with prostaglandin 3 of 54
4 analogues (PGA), medical treatment with beta blockers and trabeculectomy (for COAG patients only). The original model constructed for the guideline was sensitive to changes in costs. As a PGA available on the NHS (Latanoprost) is now generically available, the expected cost of medical treatment of PGA is likely to change. Furthermore, other costs associated with referral, diagnosis and treatment have changed from the 2006/07 sources used to populate the model. Feedback from the NCC and NICE health economist team indicate that the unit cost changes are highly likely to alter the recommendations for the treatment of OHT and suspected COAG. In addition, a respondent highlighted that the cost of referral of patients for central corneal thickness in case finding for OHT could be factored into the model. 9. The majority of respondents felt that there is sufficient variation in current practice and new evidence to warrant an update of the current guideline. They indicated that the guideline should be updated due to potential changes that PGA pricing may have on the current economic model and recommendations within CG85. In addition, suggested areas that should be revised included: clearer guidance for referral thresholds, the incorporation of emerging evidence of effectiveness of organisation of care and new health technologies. Implementation and post publication feedback 10. In total 70 enquiries were received from post-publication feedback, most of which were routine. In addition, the following themes were identified: clarification of intraocular pressure level for referral due to concern over increasing numbers of patients, and clarification on prevalence of glaucoma stated in the clinical guideline. 11. It was recognised before publication of the guideline that implementation would result in an increased number of referrals and monitoring visits to the hospital eye service. In order to address this, it 4 of 54
5 was also recognised that local commissioners would need to explore different models of services and to support them a NICE commissioning guide was produced. Post publication, there was a widespread increase in referrals to hospital eye services that the services could not cope with. This resulted in correspondence to NICE from Association of Optometrists) and FODO (Federation of Dispensing Opticians and the Association of British Dispensing Opticians and a number of articles and letters in optometry publications. The unmanageable increase in referrals to hospital eye services may have been related to: Clarity of the criteria in the NICE guideline on when to refer patients to the hospital eye service Expectations about when to refer patients at the lower threshold to the hospital eye service. Relationship to other NICE guidance 12. NICE guidance related to CG85 can be viewed in Appendix 1. Summary of Feedback Review proposal put to consultees: The guideline should be updated at this time. 13. In total 9 stakeholders commented on the review proposal recommendation during the two week consultation period. The table of stakeholder comments can be viewed in Appendix Eight stakeholders agreed with the review proposal and 1 stakeholder offered no opinion on the proposal. 5 of 54
6 15. Literature submitted through stakeholder consultation related to: Predictors of visual field progression Prevalence of ocular surface disease in COAG patients Medicine adherence and dosing styles The efficacy and tolerability of pharmacological treatments for OHT and COAG Terminology 16. During stakeholder consultation, areas to consider for review in an update of the guideline were highlighted The clinical and economical consequences of ocular surface disease The role of preservative-free formulations in the treatment of COAG Screening and case finding for COAG particularly in high risk groups Laser treatment Patient information with regards to dropper style and medicine adherence Anti-discrimination and equalities considerations 17. No evidence was identified to indicate that the guideline does not comply with anti-discrimination and equalities legislation. The original is inclusive of all patients over 18 years and covers adults with a diagnosis of chronic open angle glaucoma or ocular hypertension and those with chronic open angle glaucoma or ocular hypertension associated with pseudoexfoliation or pigment dispersion. The guideline covers populations who have a higher prevalence of glaucoma and may have worse clinical outcomes including people with 6 of 54
7 a family history of glaucoma, younger people (<50 years) and people who are of black African or black Caribbean descent. The guideline does not cover patients with secondary glaucoma or at risk of, primary or secondary angle closure glaucoma and adults with primary congenital, infantile or childhood glaucoma (excluding those detailed above). Relationship to quality standards 18. This guideline relates to a published quality standard on Glaucoma (2011). 19. The update of recommendations within CG85: Glaucoma is unlikely to invalidate the published quality standard: Glaucoma (2011) (see Appendix 3). 20. This topic is part of the library of NICE Quality Standard NHS healthcare topics. Conclusion 21. The evidence and intelligence identified through the process suggests that some areas of the guideline may need updating, particularly in relation to: The economic model for cost effective treatment options for people with OHT and suspected COAG Clarification of the referral threshold for patients to the hospital eye service The clinical and economical consequences of ocular surface disease and the role of preservative-free formulations in the treatment of COAG and OHT Laser treatment Patient information with regards to dropper style and medicine adherence 7 of 54
8 22. s suggested that the should be extended to cover screening and case finding for COAG particularly in high risk groups. 23. Guidance Executive confirmed that Centre capacity will be prioritised to accommodate new topic referrals. Where there is currently a published quality standard or where a quality standard is not required, guideline updates will be subject to NICE scheduling processes at a later date. 24. An update of CG85 Glaucoma would inform a review of the glaucoma quality standard (2011). 25. The guideline should not be updated at this time. The guideline update should be timed to support a future review of the glaucoma quality standard. Mark Baker Centre Director Louise Millward Associate Director Katy Harrison Technical Analyst Centre for Clinical Practice 7 th August of 54
9 Appendix 1 The following NICE guidance is related to CG85: Guidance IPG260: Canaloplasty for primary open-angle glaucoma 28 May 2008 IPG397: Trabeculotomy ab interno for open angle glaucoma Issued: May IPG 396:Trabecular stent bypass micro-surgery for open angle glaucoma May 2011 Related NICE guidance in progress CMG44: Services for people at risk of developing glaucoma (update) Review date N/A N/A N/A 2015 March 2012 Related NICE quality standard Glaucoma Quality Standard. Issued March 2011 TBC 9 of 54
10 Appendix 2 National Institute for Health and Clinical Excellence Glaucoma Diagnosis and management of chronic open angle glaucoma and ocular hypertension Guideline Review Consultation Table 25 th June 9 th July 2012 Agree SH SH University of Winchester, RCGP University of Winchester, RCGP Yes Yes There are several abbreviations that will slow the lay reader. OHT is still less commonly used in practice and ocular hypertension might read better at this stage (Clinical area 2) Has sufficient attention been paid to the use of new ocular imaging techniques used in high street opticians These allow more accurate estimation of cupping, and retinal disease Not excluded No problems Possibly an excluded area Balance community and hospital services comment. It will be passed to developers for consideration during scoping when this 10 of 54
11 SH University of Winchester, RCGP Yes Level of evidence is unclear and I found myself looking for a reference on classification of level of evidence to help me decide how useful the studies were. No problems guideline is updated. comment. Please note that for the process of preparing a guideline review consultation document we do not conduct a full systematic review of the literature. We undertake an assessment of abstracts. SH University of Winchester, RCGP Yes Increased referrals are noted but there is no reference to alternative models of service being commissioned. This includes more integrated services or services based in primary care with ophthalmic optician links (page 30) Possibly an excluded area Balance community and hospital services comment. Please note NICE has published a commissioning guideline: CGM44 Services for people at 11 of 54
12 risk of developing glaucoma (March 2012) SH RNIB Yes Diagnosis: comment one We are aware of problems with inappropriate referrals and its impact on hospital capacity. We also know that due to the ageing population, the prevalence of glaucoma is set to rise. Managing demand will be critical for Glaucoma care in the future and this is something that an update of guideline 85 could emphasise. These will be passed to developers for consideration during scoping when this guideline is updated. Perhaps the use of an intermediary screening process could be considered. Where optometrists or GPs refer suspected cases of COAG/OHT to a screening unit who could then decide whether the patient needs to be referred to hospital for treatment. This might help prevent patients with false positive tests taking up space in eye clinics. SH RNIB Diagnosis: comment two comment. This will be 12 of 54
13 Clinicians also tell us that there is an overemphasis on OHT which could mean normal pressure Glaucoma is being missed. An updated guideline could provide advice on this issue. SH RNIB Diagnosis: comment three Some optometrists do not provide the right tests to identify Glaucoma which does not facilitate early diagnosis. Perhaps an updated guideline could stress the need for all the right machinery to be in place in order to properly diagnose COAG/OHT. (We acknowledge that the tests required to diagnose these conditions are listed in the current guideline). SH RNIB Diagnosis: comment four passed to developers for consideration during scoping when this guideline is updated. However, please note that the diagnosis algorithm within CG85 does indicate that optic nerve head and visual field assessment changes should suspect COAG. This will be passed to developers for consideration during scoping when this guideline is updated. 13 of 54
14 We know that certain groups are more at risk of developing Glaucoma, such as those from BME/low socio-economic groups. These will be passed to developers for consideration during scoping when this guideline is updated. An updated guideline could consider ways of identifying undiagnosed Glaucoma patients, particularly those in the hard to reach groups. For example - is there any value in a screening programme for at risk groups SH RNIB Treatment: comment five We are encouraged that the "person centred" section of guideline 85 notes that: treatment and CG85 already states that the healthcare 14 of 54
15 care should take into account people s needs and preferences. We are aware that since Xalatan came off patent, different generic versions are being offered to patients - with different types of droppers. We have been told by patients that some droppers are easier to use than others. We would like an updated guideline to take this into consideration and note that clinicians should discuss the different types of droppers and help patients select the one that is right for them - or at least spend time showing the patient how to use the new dropper. professional should offer people the opportunity to discuss their treatment and provide them with relevant information. This was stated to include how to apply eye drops, and technique(s) (punctal occlusion and devices). SH RNIB Monitoring: comment six Recent RNIB research shows that many hospitals do not keep a record of how many Glaucoma follow-up appointments are rescheduled. In the ones that do, a high proportion are cancelled/re-schedules (e.g. 50% in Heart of England). These will be passed to developers for consideration during scoping when this guideline is updated. We would like an updated guideline to provide 15 of 54
16 advice on the importance of keeping records about Glaucoma patients. This could ensure patients are properly monitored and do not suffer unnecessary sight loss due to cancelled appointments. SH RNIB Monitoring: comment seven There is a case for using different professionals to manage the low, medium and high risk patients. This could be addressed in an updated guideline. SH RNIB Provision of information: comment eight Although the current guideline covers the provision of information (point 1.6), we believe an updated guideline could put more emphasis on this section and highlight the importance of good communication skills. Many patients tell us that they are not: comment. This will be passed to developers for consideration during scoping when this guideline is updated. These will be passed to developers for consideration during scoping when this guideline is updated and also to the NICE implementation team. told why they are being given eye drops instead of surgery 16 of 54
17 informed that the drops need to be taken for life - some patients think they only need to take one course and that is enough shown how to use the eye drops nor given a point of contact should they need further support/training told about follow-up appointments and why they are given a specific interval such as one month, three months or six months informed about possible side effects and what to do if they experience them We believe more needs to be done to address the issue of information provision to ensure patients really understand their eye condition and the importance of treatment/follow-up. Patients also tell us that it is hard to get hold of preservative free drops. Although this issue is probably outside the remit of this guideline, it is a problem that needs addressing. SH The Optical Confederation Yes The Optical Confederation supports a review of guideline G85 for the reasons identified in the second bullet point conclusion of the Centre for We believe the identification and triaging of patients These will be passed 17 of 54
18 SH The Optical Confederation Yes Clinical Practice - Review Consultation document. The review group should include representatives of community optometric practice who are able to contribute having a national perspective, who have an understanding of relevant medico-legal issues and who can help ensure that the publication of a revised and updated guideline that does not cause unintended difficulties. in community optometric practice who have, or are at risk of developing, chronic open angle glaucoma should be included in the of the review and form part of a revised guideline. The whole patient pathway, beginning with case finding in primary care, should be considered. The criteria for the referral of patients to secondary care should be considered and form part of a revised guideline as recommended by the Centre for People from certain ethnic backgrounds are at increased risk of developing chronic open angle glaucoma; in particular those with an African origin are at increased risk of developing a more to developers for consideration during scoping when this guideline is updated. When a guideline is updated positions for the Guidance Development Group are advertised on the NICE website. NICE strives to include 18 of 54
19 SH The Optical Confederation Yes There is currently much inefficiency, unnecessary pressure in the system and care waste in transaction costs by slight variants of the same pathways being commissioned and implemented in piece-meal and staccato fashion Clinical Practice. aggressive form of glaucoma at an earlier age. Many patients continue to present as late stage with significant visual impairment as a consequence. Although it may be beyond the of the guideline review we feel that it would be beneficial to highlight these issues and identify steps that might be taken to minimise barriers and improve access to services. representation from across the range of stakeholders. The criteria for referral to secondary care have been highlighted as a potential area for update by the current process. Screening and active case finding were beyond the of CG85; however your comments will be passed to developers for consideration during t scoping when this guideline is updated. Please note that NICE has published a 19 of 54
20 SH SH Department of Health Merck Sharp & Dohme Ltd Agree across the country. It would be helpful if NICE were able to commend a pathway (such the LOCSU Pathway for Glaucoma Referral Refinement ( which NICE officials have commended in the past) for national adoption. This would enable optometrists and ophthalmologists across the country to harmonise standards, audit, prejudicial delays and outcome measurement for to achieve best outcomes and greatest efficiency within available resources. No substantive comments to make on this consultation. MSD agree with the proposal that CG85 (Glaucoma: Diagnosis and management of chronic open angle glaucoma and ocular hypertension) requires updating. commissioning guideline: CGM44 Services for people at risk of developing glaucoma (March 2012). Your comments will be passed on to the implementation team at NICE. comment. comment. SH Merck Sharp & Dohme Ltd With regards to Clinical Area 2: Overview of treatment of OHT and suspected COAG (pages 5 & 6 and the RCD), the summary states that the evidence identified supports the conclusions and economic model within CG85 (i.e. that the treatment of all individuals with OHT is not cost The current proposal is that the guideline should not be updated 20 of 54
21 effective. However treatment is cost-effective in preventing eventual visual loss from COAG in patients in higher risk OHT subgroups). Given the increased availability of generic medicines since the initial review, the economic considerations in the wider treatment of OHT are now altered. In addition, there is an earlier diagnosis and treatment of OHT combined with an uncertainty in the relationship and timecourse between the diagnosis of OHT and the development of COAG. It is suggested that the relationship between OHT and the development of COAG, treatment options in OHT patients and the economic considerations of these treatments merit further review. at this time. The guideline update should be timed to support a future review of the glaucoma quality standard. At that stage, the economic model for cost effective treatment options for people with OHT and suspected COAG would also be updated. Your comments will be passed to developers for consideration during scoping when this guideline is updated. SH Merck Sharp & Dohme Ltd Regarding Clinical Area 3: Treatment of COAG (pages 6-24), the RCD states that 14 studies 21 of 54
22 were identified that met the inclusion criteria (RCTs with a duration of > 6 months). However no mention has been made of tafluprost (Saflutan ) data, despite results being available from a 2010 study which assessed the efficacy and safety of tafluprost % (preserved formulation) versus latanoprost 0.005% eye drops in open-angle glaucoma and ocular hypertension 1. We suggest that these data be considered during the update of CG85. All published references will be passed to developers for consideration during scoping when this guideline is updated and subject to NICE appraisal techniques. Tafluprost (Saflutan ) is licensed in the UK for the reduction of elevated intraocular pressure in open angle glaucoma and ocular hypertension, and has received a positive recommendation (restricted) 2 from the SMC for its use in Scotland. Ref 1: Uusitalo H., Pillunat L.E., Ropo A. (2010) Efficacy and safety of tafluprost % versus latanoprost 0.005% eye drops in open-angle glaucoma and ocular hypertension: 24-month results of a randomized, double-masked phase III study. Acta Ophthalmol. 88(1): of 54
23 Ref 2: SMC Advice No. 581/09: tafluprost 15 micrograms/ml preservative-free eye drops single-dose container (Saflutan ). Available at: st Salflutan FINAL_November_2009_for_we bsite.pdf (accessed 27 June 2012) SH Merck Sharp & Dohme Ltd The related clinical question from the guideline which appears on page 7 of the RCD is Which are the most clinically and cost effective and least harmful pharmacological treatments from the following classes of drugs...". The summary of evidence presented refers to a network meta-analysis, which combined direct and indirect estimates of the effect of eight drugs and placebo from 28 RCTs, and describes the rank order of these drugs from high to low in their treatment effect on a mean reduction in IOP. comment. Treatment options that have supporting evidence will be passed to developers for consideration during scoping when this guideline is updated and subject to NICE appraisal. Tafluprost (Saflutan ) does not currently appear in the rank order presented on page 8 as it was not available at the time the meta-analysis was conducted. Since 2009, tafluprost (Saflutan ) has been available on the UK market and it is 23 of 54
24 suggested this product be included as a treatment option. SH Merck Sharp & Dohme Ltd Within Clinical area 3: Treatment of COAG, a crucial data set has not been included in the evidence identified. Martinez et al 3 conducted a 5-year RCT, the results of which indicated a beneficial effect of dorzolamide+timolol over brinzolamide+timolol. Though no clear difference in IOP lowering effect was shown, the dorzolamide+timolol combination achieved a significantly enhanced protective effect against visual field loss. Ref 3: Martínez A., Sanchez-Salorio M. (2010) Predictors for visual field progression and the effects of treatment with dorzolamide 2% or brinzolamide 1% each added to timolol 0.5% in primary open-angle glaucoma. Acta Ophthalmol. 88(5): The RCT by Martinez et al was not included in the consultation document as the abstract discussed risk factors for progression in primary open-angle glaucoma. This area is outside the current of the guideline. SH Merck Sharp & Dohme Ltd Page 11/ 12 (Clinical area 3: Treatment of COAG ) has a sub-heading Adverse events associated with pharmacological treatments. This section refers to 3 studies (a meta-analysis Any studies that investigated 24 of 54
25 of the safety and tolerability from two RCTs for tds brimonidine purite 0.1% and brimonidine purite 0.15%, in reducing IOP in patients with OAG or OHT; a meta-analysis investigating latanoprost versus travoprost and bimatoprost in patients with OHT or OAG; and a pooled analysis of six RCTs comparing once daily to twice daily bimatoprost 0.03%). However no reference is made to the role of preservativefree formulations in relieving the clinical sequelae of ocular surface disease. We believe the role of preservative-free formulations as a treatment option in COAG/OHT merits consideration. preservative free formulations and did not specifically report adverse events as their primary outcome will have being included in the section on treatment effectiveness. SH Merck Sharp & Dohme Ltd Page 28 of the RCD describes the cost-utility analysis that was conducted for the purpose of economic modelling for CG85. We suggest that any update to the economic model for CG85 should take into account the feasibility of cost comparisons based on the medium to long-term benefits of preservative-free formulations. The cost consequences of ocular surface disease relating to the use of preservative-containing products should also be considered when These references will be passed to developers for consideration during scoping when this guideline is updated and subject to NICE 25 of 54
26 updating the economic model. The emerging body of evidence demonstrates that approximately 50% of glaucoma patients suffer from concurrent ocular surface disease 4-6, and that preservatives contribute to, and exacerbate this. appraisal techniques. Ref 4: Fechtner R.D., Godfrey D.G., Budenz D., et al. (2010) Prevalence of ocular surface complaints in patients with glaucoma using topical intraocular pressure-lowering medications. Cornea. 29(6): Ref 5: Leung E.W., Medeiros F.A., Weinreb R.N. (2008) Prevalence of ocular surface disease in glaucoma patients. J Glaucoma. 17(5): Ref 6: Baudouin C., Renard J.P., Nordmann J.P., et al. (2012) Prevalence and risk factors for ocular surface disease among patients treated over the long term for glaucoma or ocular hypertension. Eur J Ophthalmol. Jun; 11:0. doi: /ejo [Epub ahead of print] SH Merck Sharp & With respect to the conclusions listed on page 26 of 54
27 Dohme Ltd 32, it is suggested that the following key issues form part of the updated Guideline's review / discussion:- The clinical consequences of ocular surface disease and the effects of preservatives The economic burden produced by ocular surface disease. The role of preservative-free formulations in the treatment of COAG (it is proposed that the role of preservative-free formulations be clearly defined within the updated Guideline). Tafluprost (Saflutan ) be included in the Guideline following its licence in 2009 and subsequent receipt of a positive recommendation (restricted) from the SMC. 27 of 54 Details of these suggested areas will be passed to developers for consideration during scoping when this guideline is updated and subject to NICE appraisal techniques. The effectiveness and adverse events of relevant interventions that meet NICE s criteria of a published evidence base and UK licence will be considered by the developers when they update specific areas of the guideline. SH Merck Sharp & We suggest that consideration be given in the
28 Dohme Ltd Guideline to the current guidance from the European Glaucoma Society (EGS).The 3 rd edition of the Terminology and Guidelines for Glaucoma 7 states that the use of preservativefree preparations/delivery systems may be considered to avoid problems such as inflammatory conjunctival side effects and toxicity of the ocular surface. It goes on to state that preservative-free formulations may be relevant for certain conditions such as dry eyes or eyes with other ocular surface disorders. We also suggest that consideration be given in the Guideline to the public statement on antimicrobial preservatives in ophthalmic preparations for human use, issued by the European Medicines Agency (EMEA) in The effectiveness and adverse events of relevant interventions that meet NICE s criteria of a published evidence base and UK licence will be considered by the developers when they update specific areas of the guideline. This statement included the following: "Ophthalmic preparations without preservatives are needed for those patients who do not tolerate eye drops with preservatives. In addition, for long term treatment, formulations without preservatives are considered to be 28 of 54
29 valuable alternatives." The statement is available online at: s/ en.pdf (accessed 27 June 2012). Ref 7: Terminology and Guidelines for Glaucoma 3 rd Edition (2008) European Glaucoma Society SH Merck Sharp & Dohme Ltd As a general comment, we suggest that variations in drug delivery systems should be considered in the Guideline. Differences in the delivery mechanism (i.e. bottles, single-dose containers etc.) of generic versions of a number of the formulations referred to in the RCD, can have a direct impact on patient compliance and safety. comment. This will be passed to developers for consideration during scoping when this guideline is updated. SH Royal College of Nursing Yes We agree that the guideline should be considered for an update at this time. Not that we are aware of. None comment. SH Royal College The criteria for referral should be IOP done by 29 of 54
30 SH of Nursing Peterborough & Stamford NHS FT No Goldman Tonometer (as that is the way they specify that the hospitals do it) and that more than one episode of raised IOP should be split by at least 24 hours (rather than the 10 minutes between, which currently happens). Clinical area 1: recommends that automated perimetry should be offered for diagnosis and monitoring. comment. This will be passed to developers for consideration during scoping when this guideline is updated. comment. Agree should continue SH Peterborough & Stamford NHS FT Yes Clinical area 2: Overview of treatment of OHT and suspected COAG: treatment was cost effective in preventing eventual vision loss from COAG in patients in higher risk OHT subgroups. Agree with the statement. Is nice going to review the economic model based on current practice -The current guidelines algorithm has been seriously criticized for over threating at low OHT in patients The current proposal is that the guideline should not be updated at this time. The guideline update should be timed to support a future review of the glaucoma quality standard. 30 of 54
31 with thin corneas. No comments in the review. At that stage, the economic model for cost effective treatment options for people with OHT and suspected COAG would be updated. Your comment will be passed to developers for consideration during scoping when this guideline is updated. SH Peterborough & Stamford NHS FT Yes Clinical area 3: Treatment of COAG -More clarity is needed about laser recommendation. -Cataract operation as treatment for glaucoma should be consider, as is a very common practice. These will be passed to developers for consideration during scoping when this guideline is updated. 31 of 54
32 SH SH SH Peterborough & Stamford NHS FT Peterborough & Stamford NHS FT Peterborough & Stamford NHS FT no yes Yes Clinical area 4: Complementary and alternative therapies Laser questionnaire was distributed to GDG members and the National Collaborating Centre to consult them on the need for an update of the guideline: I didn t receive any about it. Was this for a new GDG group Or was this the original GDG group which I was part of -Referral was highlighted as having been an issue at the time of publication which had -I think will be a good idea to endorse the referral comment. comment. The GDG questionnaire was distributed to the GDG members via the contact list provided by the orginal GDG that developed the guideline. We are sorry to learn that in this instance you did not receive the questionnaire. We appreciate your submission during the consultation phase of this guideline review. 32 of 54
33 resulted in volume problems for services. question. -latanoprost coming off patent needing the OHT/GS algorithm to be reviewed. These will be passed to developers for consideration during scoping when this guideline is updated.. -Agree screening/case detection should be reviewed. -l Agree clearer guidance for referral thresholds would be desirable College of Optometrists Yes Page 1: The evidence reviewed has resulted in the Panel indicating that current evidence is satisfactory with the possible exception of the treatment recommendations where factoring in NICE methodology for 33 of 54
34 College of Optometrists other data into the health economics model may influence guidelines. We would suggest that it is worth reviewing what constitutes appropriate evidence. RCTs might not always be the correct study design for some questions and the Panel might not always have the right expertise to have opinions on all aspects. Page 1 typo: Chronic Open Angled Glaucoma should read Chronic Open Angle Glaucoma. guideline development allows for the utilisation of the highest level and most appropriate study type. The guideline review process considers a high level search and an assessment of abstracts. The guideline review process also includes public consultation in order to ascertain the opinions of a wide range of experts and stakeholders. comment. College of Optometrists Pages 3 5: Clinical Area 1: Diagnosis and Monitoring of patients with OHT, COAG and suspected COAG. A section explicitly dealing with diagnosis and referral in primary care would comment. This will be passed to developers for 34 of 54
35 College of Optometrists be useful to avoid the large, unintended impact that the current guideline had on referrals to the hospital eye service (warned of in response to first draft by Moorfields) also see comments below. Page 28: We welcome the mention of the Guidance on the referral of glaucoma suspects by community optometrists which was issued by the College of Optometrists and Royal College of Ophthalmologists. We should like to draw your attention to the Joint Supplementary College Guidance on Supervision in relation to Glaucoma-related Care by Optometrists, which the two Colleges also issued. These documents are attached. consideration during scoping when this guideline is updated.. These documents will be passed to developers for consideration during scoping when this guideline is updated. College of Optometrists Pages 27, 28, 29 Referral: The phrase with regard to practices recommended in the current guideline is misleading because the current guideline excludes screening/case finding and is about diagnosis and management. Although this has implications for referral it is not referral. The place for any recommendation about referral may need to be elsewhere unless the guidelines 35 of 54 Referral refers to the transfer of care for a patient from one clinician to another and is not specific to
36 College of Optometrists emphasis changes completely to referral, diagnosis and management. The issue of referral needs to be addressed with clarity because of the amount of feedback received on this point. General Comment: Originally we understood that you could not mention specific qualifications in your guidelines. However, we note that you now do mention them in your related commissioning guidance so the view may have changed. We should like to draw your attention, therefore, to the screening/ case finding. Hence referral within the guideline may occur during diagnosis and treatment of the patient. These will be passed on to the NICE implementation team. 36 of 54
37 fact that, to support optometrists in glaucoma care, the College is replacing its current higher qualifications (Glaucoma A and Glaucoma B) which are mentioned in your commissioning guidance. We are now introducing a suite of three related qualifications run by universities and other bodies that we accredit: a certificate level qualification, which is already in place, and which would fit the top-up qualification that you mention in your 37 of 54
38 Allergan Yes In the full guideline, there are tables regarding: - Monitoring intervals for people with COAG (page 54, 105,108 ) -Currently the section on Monitoring intervals for people with OHT or suspected COAG who are recommended to receive Medication (page 53, 107,108) recommends the review of target IOP of glaucoma treatment and change in treatment plan at 1-4 months in order to delay progression. Therefore, we believe that this guidance is inconsistent in its recommendations as it 38 of 54 commissioning guidance for monitoring OHT and suspect COAG, a higher certificate, which will shortly replace Glaucoma A and a diploma, which will shortly replace Glaucoma B. Details of each are attached. These will be passed to developers for consideration during scoping when this guideline is updated.
39 proposes clinical practice therapeutic reviews between 1-4 months reflecting real-life practice, yet the evidence base that this is based on includes only studies of >6 months. Further, this cut-off is different and inconsistent with the requirements of assessment of efficacy from the licensing authorities. Interestingly, we also note the decision of the Agency for Healthcare Research and Quality (AHRQ), a US Government Agency with the stated aim of providing patients and physicians with state-of- the-science information on which medical treatments work best for a given condition. On April , the AHRQ published a review of the effectiveness of glaucoma treatments based on the assessment of medical treatment outcomes at a minimum of 1 month post intervention (page ES-8) As such we would recommend inclusion of a more complete dataset to include robust clinical 39 of 54
40 studies of <6 months duration Allergan Yes Regarding the pharmacological treatment for OHT, suspected COAG and COAG, the following prostaglandin analogue/prostamide (PGA/PM) vs. PGA/PM clinical studies show significant differences in treatment effects on IOP. We believe these studies should be included to represent a full picture and may have an impact on the guideline recommendations. Supporting data Noecker RS, Dirks MS, Choplin NT, et al. A six-month randomized clinical trial comparing the intraocular pressure-lowering efficacy of bimatoprost and latanoprost in patients with ocular hypertension or glaucoma. Am J Ophthalmol 2003;135: Cantor LB, Hoop J, et al. Intraocular pressurelowering efficacy of bimatoprost 0.03% and travoprost 0.004% in patients with glaucoma or ocular hypertension. Br J Ophthalmol 2006;90: Van der Valk et al. J Clin Epidemiol References pre 2009 would have been included for assessment within the development of CG85 if they met the relevant criteria. The Van der valk et al (2009) reference is included within the guideline review evidence. However, the study by Kontstas et al (2010) was of less than 6 months duration and as such does not meet the guideline inclusion criteria. The reference provided by Kymes et al is a methodology paper on how to 40 of 54
41 2009;62: A network meta-analysis combined direct and indirect comparisons between glaucoma drugs to rank effectiveness in lowering intraocular pressure Aptel F, Cucherat M, Denis P. Efficacy and tolerability of prostaglandin analogs: a meta-analysis of randomized controlled clinical trials. J Glaucoma 2008;17: conduct an online systematic review and was not considered to be within the of guideline review process. Dirks Monte S. Advances in therapy 2006;23: A 3-Month Clinical Trial Comparing the IOP- Lowering Efficacy of Bimatoprost and Latanoprost in Patients With Normal-Tension Glaucoma Konstas G P et al Br J Ophthalmol : Twenty-four-hour intraocular pressure control with bimatoprost and the bimatoprost/timolol fixed combination administered in the morning, or evening in exfoliative glaucoma 41 of 54
42 Denis Ph et al Current Medical Research Opinion 2007;23 (3) A meta-analysis of topical prostaglandin analogues intra-ocular pressure lowering in glaucoma therapy Kymes SM et al, Demonstration of an online tool to assist managed care formulary evidence-based decision making: meta-analysis of topical prostaglandin analog efficacy Allergan Yes Regarding the pharmacological treatment for OHT, suspected COAG and COAG, the following combination therapy clinical studies show significant differences in treatment effects on IOP. We believe these studies should be included to represent a full picture and may have an impact on the guideline recommendations. Supporting data Aptel F, Cucherat M, Denis P. Efficacy and tolerability of prostaglandin-timolol fixed combinations: a meta-analysis of randomized clinical trials. Eur J Ophthalmol 2012;22:5-18 The reference by Aptel et al 2012 was published after the literature search was conducted. It will be to developers for consideration during scoping when this guideline is updated. However, the 2 42 of 54
43 Centofanti M, Oddone F et al, Eur J Ophthalmol 2009;19: Efficacy of the fixed combinations of bimatoprost or latanoprost plus timolol in patients uncontrolled with prostaglandin monotherapy: a multicenter, randomized, investigator-masked, clinical study. references by Centofanti et al (2009, 2010) did not meet the inclusion criteria for the guideline review of studies of greater than 6 months duration. Centofanti M, Oddone F, Gandolfi S, et al. Am J Ophthalmol 2010;150: Comparison of travoprost and bimatoprost plus timolol fixed combinations in open-angle glaucoma patients previously treated with latanoprost plus timolol fixed combination. Allergan Yes Regarding the pharmacological treatment of OHT, suspect COAG and COAG, we believe that the guideline is incomplete without a review of the switch data (inter- and intra- class monotherapy to monotherapy) for the recommended first line agents. Provision of switch recommendations will guide and support clinical judgments and reflect clinical practice. Additionally it will reflect the European Glaucoma Society Guidelines. These will be passed to developers for consideration during scoping when this guideline is updated and subject to NICE appraisal techniques 43 of 54
44 In addition, non-response to first line therapy has been reported in literature. A clear statement should be included in the guideline regarding the switch of monotherapy first line agents. It is proposed to specify within sections 7.8 and 8.8 Offer alternative pharmacological treatment to people requiring monotherapy if they achieve less than the expected response to the first line treatment Supporting reference European Glaucoma Society. Terminology and Guidelines for Glaucoma. 3rd ed. Savona, Italy, Editrice Dogma, 2008; pgs: 33 (flowcharts VII, IX) Camras CB, Hedman K. Rate of response to latanoprost or timolol in patients with ocular hypertension or glaucoma. J Glaucoma. 2003; 12: Choplin N, Bernstein P, Batoosingh AL, et al. A 44 of 54
45 randomized, investigator-masked comparison of diurnal responder rates with bimatoprost and latanoprost in the lowering of intraocular pressure. Surv Ophthalmol. 2004;49(Suppl 1):S19 S25 Allergan Yes On P211 Regarding treatment of COAG the guidelines states Patients may not be fit for surgery or may not wish to proceed to surgery because of anxiety or other issues. Where this situation arises alternative attempts at IOP lowering may be necessary. Options which may need to be considered include laser treatments, or multiple topical pharmacological treatments. There is a similar comment on p214 about those not wanting surgery more than one agent may be needed concurrently to achieve target IOP These will be passed to developers for consideration during scoping when this guideline is updated and subject to NICE appraisal As many patients have poor compliance, adherence and persistence with their medication (Reardon G, Kotak S, Schwartz GF. Objective assessment of compliance and persistence among patients treated for glaucoma and ocular hypertension: a systematic review. Patient Preference and Adherence 2011: ) it would be appropriate to recommend fixed 45 of 54
46 combination therapy, if suitable, for the patient to reduce the frequency of dosing for those patients. Combination therapies have further advantages in terms of reducing preservative exposure for those on concurrent treatment. Additionally they remove the risk of a second eye drop instillation occurring immediately after a prior instillation and washing away the first eye drop, thereby reducing drug concentration of the first drop (Chrai SS, Makoid MC, Eriksen SP, Robinson JR. Drop size and initial dosing frequency problems of topically applied ophthalmic drugs. J Pharm Sci. 1974; 63: ) Further, analysis of actual real-life prescribing patterns would imply that glaucoma prescribers recognise these benefits. Since the launch of the first guidelines in April 2009, the number of bottles dispensed for glaucoma has increased by 12.39%. Of these, monotherapy bottle dispensing has increased by 6.86%, whilst all fixed combinations have increased by 36.47%, 46 of 54
47 and prostaglandin fixed combinations by 60.25% (Source: IMS Retail Sales Analysis S1E; Moving Annual Total [MAT] April 2009 MAT April 2012; Data on File). Although we appreciate the concern about the inclusion of timolol 0.5% in all fixed combinations and the resultant risk of systemic adverse events, we recommend that the new guidelines advise that fixed combinations be recommended for those patients on multiple glaucoma medications (with appropriate screening for systemic risk) in order to minimise the risk of poor compliance, adherence and persistence which may result in a loss of IOP control and risk of progression to sight loss. This would also further align the Guidelines with emerging prescribing practise. Allergan Yes There is a range of bottle designs which have been selected by generic manufacturers of glaucoma These will be passed to developers for consideration during 47 of 54
48 treatments (Ref: Royal College of Ophthalmology Advice on Generic Latanoprost Prescribing, available at ac.uk/page.aspse ction=442§iont itle=clinical+statem ents%2fadvice ). scoping when this guideline is updated and subject to NICE appraisal This could result in a change in bottle design, aperture, colour, labeling, force required to dispense just one drop, storage requirements and extent of license. This could be further 48 of 54
49 exacerbated with a monthly change of product manufacturer. Allergan Yes We note that the full guideline classifies bimatoprost as a prostaglandin analogue. 49 of 54 With regards to Chapter 11, Provision of Information for Patients, we recommend the inclusion of a comment regarding healthcare professionals enquiring as to whether the patient is experiencing any confusion or difficulty with the use of a generic glaucoma treatment. comment.
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE. Glaucoma: diagnosis and management of chronic open angle glaucoma and ocular hypertension
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Glaucoma: diagnosis and management of chronic open angle glaucoma and ocular hypertension 1.1 Short title Glaucoma 2 Background
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Glaucoma: diagnosis and management of chronic open angle glaucoma and ocular hypertension 1.1 Short title Glaucoma 2 Background
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC)
 Guidelines for the medical treatment of chronic open angle glaucoma and ocular hypertension Summary: DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) Diagnosis and management of ocular hypertension (OHT)
Guidelines for the medical treatment of chronic open angle glaucoma and ocular hypertension Summary: DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) Diagnosis and management of ocular hypertension (OHT)
LOCSU Community Services. Glaucoma Repeat Readings & OHT Monitoring Community Service Pathway. Issued by Local Optical Committee Support Unit May 2009
 LOCSU Community Services Glaucoma Repeat Readings & OHT Monitoring Community Service Pathway Issued by Local Optical Committee Support Unit May 2009 [Revised November 2013] Contents Page Executive Summary...
LOCSU Community Services Glaucoma Repeat Readings & OHT Monitoring Community Service Pathway Issued by Local Optical Committee Support Unit May 2009 [Revised November 2013] Contents Page Executive Summary...
East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults
 East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults Introduction Glaucoma is a group of eye diseases causing optic nerve damage. In most cases
East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults Introduction Glaucoma is a group of eye diseases causing optic nerve damage. In most cases
LOCSU Community Services. Glaucoma Repeat Readings & OHT Monitoring Community Service Pathway. Issued by Local Optical Committee Support Unit May 2009
 LOCSU Community Services Glaucoma Repeat Readings & OHT Monitoring Community Service Pathway Issued by Local Optical Committee Support Unit May 2009 [Revised May 2016, Version 3.3] Contents Page Executive
LOCSU Community Services Glaucoma Repeat Readings & OHT Monitoring Community Service Pathway Issued by Local Optical Committee Support Unit May 2009 [Revised May 2016, Version 3.3] Contents Page Executive
South East London Area Prescribing Committee Chronic Open Angle Glaucoma and Ocular Hypertension Treatment Pathway
 Proceed to 2 nd line treatment if further reduction in IOP required and there is good response to PGAs or (& no South East London Area Prescribing Committee Chronic Open Angle Glaucoma and Ocular Hypertension
Proceed to 2 nd line treatment if further reduction in IOP required and there is good response to PGAs or (& no South East London Area Prescribing Committee Chronic Open Angle Glaucoma and Ocular Hypertension
GLAUCOMA REPEAT READINGS PATHWAY
 GLAUCOMA REPEAT READINGS PATHWAY Level 1a: Goldmann style applanation tonometry repeat readings A first level community service for IOP refinement where other signs of glaucoma are not present will reduce
GLAUCOMA REPEAT READINGS PATHWAY Level 1a: Goldmann style applanation tonometry repeat readings A first level community service for IOP refinement where other signs of glaucoma are not present will reduce
Glaucoma: diagnosis and management (large print version)
 Glaucoma: diagnosis and management (large print version) 1 November 2017 Recommendations People have the right to be involved in discussions and make informed decisions about their care, as described in
Glaucoma: diagnosis and management (large print version) 1 November 2017 Recommendations People have the right to be involved in discussions and make informed decisions about their care, as described in
Glaucoma Care Pathway
 PRIMARY CARE OPTOMETRY UPDATE Glaucoma Care Pathway March 2018 Colleagues, In June 2017 the Health and Social Care Board provided you with the second Glaucoma Care Pathway Primary Care Optometry Update,
PRIMARY CARE OPTOMETRY UPDATE Glaucoma Care Pathway March 2018 Colleagues, In June 2017 the Health and Social Care Board provided you with the second Glaucoma Care Pathway Primary Care Optometry Update,
Glaucoma (update) Consultation on draft scope Stakeholder comments table. 19 May June 2016
 1 SH Roy college of Practitioners (RCGP) The RCGP advises that GPs and practice staff have an important role in glaucoma care in the community. As well as providing records of significant past medic history,
1 SH Roy college of Practitioners (RCGP) The RCGP advises that GPs and practice staff have an important role in glaucoma care in the community. As well as providing records of significant past medic history,
PRESCRIBING IN GLAUCOMA: GUIDELINES FOR NZ OPTOMETRISTS
 PRESCRIBING IN GLAUCOMA: GUIDELINES FOR NZ OPTOMETRISTS Introduction Independent prescribing relates to the capacity to use clinical judgement in respect of diagnosis and treatment. It does not mean working
PRESCRIBING IN GLAUCOMA: GUIDELINES FOR NZ OPTOMETRISTS Introduction Independent prescribing relates to the capacity to use clinical judgement in respect of diagnosis and treatment. It does not mean working
Costing report. Glaucoma: diagnosis and management of chronic open angle glaucoma and ocular hypertension. Implementing NICE guidance.
 Glaucoma: diagnosis and management of chronic open angle glaucoma and ocular hypertension Costing report Implementing NICE guidance April 2009 NICE clinical guideline 85 National costing report: glaucoma
Glaucoma: diagnosis and management of chronic open angle glaucoma and ocular hypertension Costing report Implementing NICE guidance April 2009 NICE clinical guideline 85 National costing report: glaucoma
NICE guideline Published: 1 November 2017 nice.org.uk/guidance/ng81
 Glaucoma: diagnosis and management NICE guideline Published: 1 November 2017 nice.org.uk/guidance/ng81 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Glaucoma: diagnosis and management NICE guideline Published: 1 November 2017 nice.org.uk/guidance/ng81 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care
 The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Glaucoma November 2016 Association of Health Professions in Ophthalmology General basic competences
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Glaucoma November 2016 Association of Health Professions in Ophthalmology General basic competences
Glaucoma is the most frequent
 Refined glaucoma referral practice offers prospect of improved capacity and expanded role for primary eye care professionals BY ROD MCNEIL Glaucoma is the most frequent cause of irreversible blindness
Refined glaucoma referral practice offers prospect of improved capacity and expanded role for primary eye care professionals BY ROD MCNEIL Glaucoma is the most frequent cause of irreversible blindness
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care
 The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Acute & Emergency Care November 2016 Association of Health Professions in Ophthalmology General
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Acute & Emergency Care November 2016 Association of Health Professions in Ophthalmology General
THE CHRONIC GLAUCOMAS
 THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA? People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA? People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
England. Suspect OHT/Glaucoma. Repeat Readings Service Common Pathway
 England Suspect OHT/Glaucoma Repeat Readings Service Common Pathway Greater Manchester Local Eye Health Network April 2015 Contents Page Executive Summary... 3 Background... 5 NICE Guidance and Quality
England Suspect OHT/Glaucoma Repeat Readings Service Common Pathway Greater Manchester Local Eye Health Network April 2015 Contents Page Executive Summary... 3 Background... 5 NICE Guidance and Quality
THE CHRONIC GLAUCOMAS
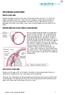 THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
Commissioning Guide: Glaucoma (Long Version)
 Draft for Consultation Commissioning Guide: Glaucoma (Long Version) March 2015 Contents Section page 1. ABBREVIATIONS 4 2. Introduction: Glaucoma 5 3. Commissioning Guidance for Glaucoma 7 4. HIGH VALUE
Draft for Consultation Commissioning Guide: Glaucoma (Long Version) March 2015 Contents Section page 1. ABBREVIATIONS 4 2. Introduction: Glaucoma 5 3. Commissioning Guidance for Glaucoma 7 4. HIGH VALUE
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice Review of Clinical Guideline (CG54) - Urinary tract infection in children Background information Guideline issue date:
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice Review of Clinical Guideline (CG54) - Urinary tract infection in children Background information Guideline issue date:
Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181)
 Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
VI.2.2 Summary of treatment benefits
 EU-Risk Management Plan for Bimatoprost V01 aetiology), both OAG and ACG can be secondary conditions. Secondary glaucoma refers to any case in which another disorder (e.g. injury, inflammation, vascular
EU-Risk Management Plan for Bimatoprost V01 aetiology), both OAG and ACG can be secondary conditions. Secondary glaucoma refers to any case in which another disorder (e.g. injury, inflammation, vascular
LOCSU/WOPEC Courses to support the development of local enhanced eye care services. Excellence in Education for Eye Care Professionals
 LOCSU/WOPEC Courses to support the development of local enhanced eye care services Excellence in Education for Eye Care Professionals LOCSU/WOPEC training courses are designed to support the delivery of
LOCSU/WOPEC Courses to support the development of local enhanced eye care services Excellence in Education for Eye Care Professionals LOCSU/WOPEC training courses are designed to support the delivery of
Clinical Guidance and Monitoring for Change. Cecilia Fenerty MD FRCOphth Manchester Royal Eye Hospital
 Clinical Guidance and Monitoring for Change Cecilia Fenerty MD FRCOphth Manchester Royal Eye Hospital Glaucoma Referral Criteria 2000 Original referral scheme Simple criteria based on IOP/Disc/Field Solitary
Clinical Guidance and Monitoring for Change Cecilia Fenerty MD FRCOphth Manchester Royal Eye Hospital Glaucoma Referral Criteria 2000 Original referral scheme Simple criteria based on IOP/Disc/Field Solitary
Volume 9; Number 6 May 2015 PRESCRIBING FOR CHRONIC OPEN ANGLE GLAUCOMA (COAG) AND OCULAR HYPERTENSION (OHT)
 Greater East Midlands Commissioning Support Unit in association with Lincolnshire Clinical Commissioning Groups, Lincolnshire Community Health Services, United Lincolnshire Hospitals Trust and Lincolnshire
Greater East Midlands Commissioning Support Unit in association with Lincolnshire Clinical Commissioning Groups, Lincolnshire Community Health Services, United Lincolnshire Hospitals Trust and Lincolnshire
Multidisciplinary Glaucoma Service Design. Paul GD Spry PhD MCOptom DipGlau DipTp(IP) Head of Optometry, Bristol Eye Hospital
 Multidisciplinary Glaucoma Service Design Paul GD Spry PhD MCOptom DipGlau DipTp(IP) Head of Optometry, Bristol Eye Hospital 1. HES Optometrist Consultant 2. Text book co-author Spry and Harper, Essential
Multidisciplinary Glaucoma Service Design Paul GD Spry PhD MCOptom DipGlau DipTp(IP) Head of Optometry, Bristol Eye Hospital 1. HES Optometrist Consultant 2. Text book co-author Spry and Harper, Essential
GENERAL INFORMATION GLAUCOMA GLAUCOMA
 GENERAL INFORMATION GLAUCOMA GLAUCOMA WHAT IS GLAUCOMA? Glaucoma is commonly known as the sneak thief of sight because it can cause irreversible vision loss without any obvious symptoms. The term glaucoma
GENERAL INFORMATION GLAUCOMA GLAUCOMA WHAT IS GLAUCOMA? Glaucoma is commonly known as the sneak thief of sight because it can cause irreversible vision loss without any obvious symptoms. The term glaucoma
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Centre for Clinical Practice
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice Review of Clinical Guideline (CG3) Preoperative tests: the use of routine preoperative testes for elective surgery Background
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice Review of Clinical Guideline (CG3) Preoperative tests: the use of routine preoperative testes for elective surgery Background
Chronic eye diseases. Title of section divider. What s new, and how GPs can help. Eg. Case Study 1. Dr Jesse Gale, ophthalmologist
 Chronic eye diseases Title of section divider What s new, and how GPs can help Eg. Case Study 1. Dr Jesse Gale, ophthalmologist 1 Title of section divider Cataract Eg. Case Study 1. 2 Cataract - what s
Chronic eye diseases Title of section divider What s new, and how GPs can help Eg. Case Study 1. Dr Jesse Gale, ophthalmologist 1 Title of section divider Cataract Eg. Case Study 1. 2 Cataract - what s
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care
 The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Cataract November 2016 Association of Health Professions in Ophthalmology General basic competences
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Cataract November 2016 Association of Health Professions in Ophthalmology General basic competences
Micro-Invasive Glaucoma Surgery (Aqueous Stents)
 Micro-Invasive Glaucoma Surgery (Aqueous Stents) Policy Number: Original Effective Date: MM.06.029 02/01/2019 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 02/01/2019 Section:
Micro-Invasive Glaucoma Surgery (Aqueous Stents) Policy Number: Original Effective Date: MM.06.029 02/01/2019 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 02/01/2019 Section:
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE QUALITY AND OUTCOMES FRAMEWORK (QOF) INDICATOR DEVELOPMENT PROGRAMME Briefing paper QOF indicator area: Peripheral arterial disease Potential output:
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE QUALITY AND OUTCOMES FRAMEWORK (QOF) INDICATOR DEVELOPMENT PROGRAMME Briefing paper QOF indicator area: Peripheral arterial disease Potential output:
Humber. Cataract Surgery Commissioning Policy
 Intervention Elective Eye Surgery for the treatment of Cataracts in adults OPCS codes C62 Incision of iris C621 Iridosclerotomy C622 Surgical iridotomy C623 Laser iridotomy C624 Correction iridodialysis
Intervention Elective Eye Surgery for the treatment of Cataracts in adults OPCS codes C62 Incision of iris C621 Iridosclerotomy C622 Surgical iridotomy C623 Laser iridotomy C624 Correction iridodialysis
Glaucoma. College of Optometrists
 Glaucoma Overview Glaucoma is a group of eye diseases in which the optic nerve, which connects your eye to your brain, is damaged by the pressure of the fluid inside your eye. This may be because the pressure
Glaucoma Overview Glaucoma is a group of eye diseases in which the optic nerve, which connects your eye to your brain, is damaged by the pressure of the fluid inside your eye. This may be because the pressure
National Institute for Health and Clinical Excellence. Glaucoma. Guideline Consultation Comments Table. 29 September November 2008
 National Institute for Health and Clinical Excellence Glaucoma Guideline Consultation Comments Table 29 September 2008 24 November 2008 Stat us Organisa tion Order no. Version Page no Line no/se ction
National Institute for Health and Clinical Excellence Glaucoma Guideline Consultation Comments Table 29 September 2008 24 November 2008 Stat us Organisa tion Order no. Version Page no Line no/se ction
10 year surveillance (2017) Chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy) (2007) NICE guideline CG53
 10 year surveillance (2017) Chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy) (2007) NICE guideline CG53 Stakeholder consultation comments form - proposal for no update Consultation
10 year surveillance (2017) Chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy) (2007) NICE guideline CG53 Stakeholder consultation comments form - proposal for no update Consultation
Comparative study of new imaging technologies for the diagnosis of glaucoma: Protocol Approved by the Ethics Committee
 Comparative study of new imaging technologies for the diagnosis of glaucoma: Protocol Approved by the Ethics Committee HTA 09/22/111. Applicants: Augusto Azuara-Blanco (CI), Jennifer Burr,, Rodolfo Hernández,
Comparative study of new imaging technologies for the diagnosis of glaucoma: Protocol Approved by the Ethics Committee HTA 09/22/111. Applicants: Augusto Azuara-Blanco (CI), Jennifer Burr,, Rodolfo Hernández,
Collaboration in the care of glaucoma patients and glaucoma suspects. Barry Emara MD FRCS(C) Nico Ristorante November 29, 2012
 Collaboration in the care of glaucoma patients and glaucoma suspects Barry Emara MD FRCS(C) Nico Ristorante November 29, 2012 Goals of Collaboration Patient-centred and evidence based approach Timely access
Collaboration in the care of glaucoma patients and glaucoma suspects Barry Emara MD FRCS(C) Nico Ristorante November 29, 2012 Goals of Collaboration Patient-centred and evidence based approach Timely access
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care
 The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Medical Retina November 2016 Association of Health Professions in Ophthalmology General basic
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Medical Retina November 2016 Association of Health Professions in Ophthalmology General basic
Hearing aid dispenser approval process review Introduction Hearing aid dispenser data transfer... 6
 Hearing aid dispenser approval process review 2010 11 Content 1.0 Introduction... 4 1.1 About this document... 4 1.2 Overview of the approval process... 4 2.0 Hearing aid dispenser data transfer... 6 2.1
Hearing aid dispenser approval process review 2010 11 Content 1.0 Introduction... 4 1.1 About this document... 4 1.2 Overview of the approval process... 4 2.0 Hearing aid dispenser data transfer... 6 2.1
Closed Angle Glaucoma Or Narrow Angle Glaucoma. What s is a closed angle type of glaucoma,
 Closed Angle Glaucoma Or Narrow Angle Glaucoma What s is a closed angle type of glaucoma, This is where the iris is found to be blocking the drainage of the eye through the trabecular meshwork. The eye
Closed Angle Glaucoma Or Narrow Angle Glaucoma What s is a closed angle type of glaucoma, This is where the iris is found to be blocking the drainage of the eye through the trabecular meshwork. The eye
[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v
![[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v [TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v](/thumbs/94/119684929.jpg) [TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN TRAVOPR-v2-270214 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people
[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN TRAVOPR-v2-270214 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people
WHAT IS ESEP? Contact / for more information: Michael Bowen, Director of Research, College of Optometrists:
 FIVE YEARS OF ESEP College-funded research to evaluate community-based eye care service models in order to better understand the impact of the organisation of services on clinical effectiveness; cost effectiveness;
FIVE YEARS OF ESEP College-funded research to evaluate community-based eye care service models in order to better understand the impact of the organisation of services on clinical effectiveness; cost effectiveness;
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 28 May 2008
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 28 May 2008 COSOPT 20 mg/5 mg/ml, eye drops, solution in single dose container Box of 60 0.2 ml single dose containers
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 28 May 2008 COSOPT 20 mg/5 mg/ml, eye drops, solution in single dose container Box of 60 0.2 ml single dose containers
Single Technology Appraisal (STA) Erenumab for preventing migraine ID1188
 Single Technology Appraisal (STA) Erenumab for preventing migraine ID1188 Comment 1: the draft remit Response to consultee and commentator comments on the draft remit and draft scope (pre-referral) Appropriateness
Single Technology Appraisal (STA) Erenumab for preventing migraine ID1188 Comment 1: the draft remit Response to consultee and commentator comments on the draft remit and draft scope (pre-referral) Appropriateness
CABINET PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND
 CABINET Report No: 105/2017 PUBLIC REPORT 16 May 2017 PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND Report of the Director of Public Health Strategic Aim: Safeguarding Key Decision:
CABINET Report No: 105/2017 PUBLIC REPORT 16 May 2017 PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND Report of the Director of Public Health Strategic Aim: Safeguarding Key Decision:
The Future of Optometric Services in Primary Care in Wales
 IMPROVING HEALTH IN WALES The Future of Optometric Services in Primary Care in Wales a consultation document ISBN 0 7504 2968 2 Designed by CartoGraphic Services G/254/02-03 September 2002 INA - 15-02
IMPROVING HEALTH IN WALES The Future of Optometric Services in Primary Care in Wales a consultation document ISBN 0 7504 2968 2 Designed by CartoGraphic Services G/254/02-03 September 2002 INA - 15-02
ADVISORY OPINION OF THE CODE OF ETHICS
 ADVISORY OPINION OF THE CODE OF ETHICS Subject: Issues Raised: Applicable Rules: Appropriate Examination and Treatment Procedures How is it determined which examination and treatment procedures are considered
ADVISORY OPINION OF THE CODE OF ETHICS Subject: Issues Raised: Applicable Rules: Appropriate Examination and Treatment Procedures How is it determined which examination and treatment procedures are considered
Vision and eye healthcare study in residential aged care facilities
 Vision and eye healthcare study in residential aged care facilities Study report Report prepared by: Rob Cummins, Director, Research & Policy Julie Heraghty, Former Chief Executive Officer Macular Disease
Vision and eye healthcare study in residential aged care facilities Study report Report prepared by: Rob Cummins, Director, Research & Policy Julie Heraghty, Former Chief Executive Officer Macular Disease
Quickfire Audit Session. Quickfire Audits. Sourced and facilitated by. Trevor Warburton Barbara Ryan
 Quickfire Audits Sourced and facilitated by Trevor Warburton Barbara Ryan Referral Refinement Service in Shipley Stewart Mitchell SOAP Shipley Ophthalmic Assessment Programme Dr Stewart Mitchell, Dr Mark
Quickfire Audits Sourced and facilitated by Trevor Warburton Barbara Ryan Referral Refinement Service in Shipley Stewart Mitchell SOAP Shipley Ophthalmic Assessment Programme Dr Stewart Mitchell, Dr Mark
Micro-Invasive Glaucoma Surgery (Aqueous Stents)
 Micro-Invasive Glaucoma Surgery (Aqueous Stents) Policy Number: Original Effective Date: MM.06.029 02/01/2019 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 02/01/2019 Section:
Micro-Invasive Glaucoma Surgery (Aqueous Stents) Policy Number: Original Effective Date: MM.06.029 02/01/2019 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 02/01/2019 Section:
PATIENT GUIDE TO LUMIGAN 0.01%
 Treatment Tracker enclosed PATIENT GUIDE TO LUMIGAN 0.01% LUMIGAN (bimatoprost ophthalmic solution) 0.01% is used for the treatment of high eye pressure, also called intraocular pressure (IOP), in people
Treatment Tracker enclosed PATIENT GUIDE TO LUMIGAN 0.01% LUMIGAN (bimatoprost ophthalmic solution) 0.01% is used for the treatment of high eye pressure, also called intraocular pressure (IOP), in people
THE RESPONSIBLE PHARMACIST REGULATIONS
 THE RESPONSIBLE PHARMACIST REGULATIONS A SUMMARY OF THE RESPONSES TO PUBLIC CONSULTATION ON PROPOSALS FOR THE CONTENT OF THE REGULATIONS DH INFORMATION READER BOX Policy HR / Workforce Management Planning
THE RESPONSIBLE PHARMACIST REGULATIONS A SUMMARY OF THE RESPONSES TO PUBLIC CONSULTATION ON PROPOSALS FOR THE CONTENT OF THE REGULATIONS DH INFORMATION READER BOX Policy HR / Workforce Management Planning
Ocular Hypotensive Efficacy of Netarsudil Ophthalmic Solution 0.02% Over a 24-Hour Period: A Pilot Study
 Ocular Hypotensive Efficacy of Netarsudil Ophthalmic Solution 0.02% Over a 24-Hour Period: A Pilot Study James H. Peace, M.D. 1, Casey K. Kopczynski, Ph.D. 2, and Theresa Heah, M.D. 2 1 Inglewood, CA 2
Ocular Hypotensive Efficacy of Netarsudil Ophthalmic Solution 0.02% Over a 24-Hour Period: A Pilot Study James H. Peace, M.D. 1, Casey K. Kopczynski, Ph.D. 2, and Theresa Heah, M.D. 2 1 Inglewood, CA 2
Elements for a public summary. Overview of disease epidemiology
 VI.2 VI.2.1 Elements for a public summary Overview of disease epidemiology Glaucoma is an eye disease that can result in damage to the optic nerve and loss of vision (blindness). It is the major cause
VI.2 VI.2.1 Elements for a public summary Overview of disease epidemiology Glaucoma is an eye disease that can result in damage to the optic nerve and loss of vision (blindness). It is the major cause
Optometric Cataract Refined Referral
 Optometric Cataract Refined Referral Guidance Notes for Optometrists Version Control: v1: April 2013 v2: August 2015 REFINED CATARACT REFERRAL PATHWAY GUIDANCE FOR OPTOMETRISTS Background Approximately
Optometric Cataract Refined Referral Guidance Notes for Optometrists Version Control: v1: April 2013 v2: August 2015 REFINED CATARACT REFERRAL PATHWAY GUIDANCE FOR OPTOMETRISTS Background Approximately
UKAS Guidance on the Application of ISO/IEC Dealing with Expressions of Opinions and Interpretations
 LAB 13 Expression of opinions & interpretations UKAS Guidance on the Application of ISO/IEC 17025 Dealing with Expressions of Opinions and Interpretations [UKAS Publication ref: LAB 13] Contents Section
LAB 13 Expression of opinions & interpretations UKAS Guidance on the Application of ISO/IEC 17025 Dealing with Expressions of Opinions and Interpretations [UKAS Publication ref: LAB 13] Contents Section
Managing the Patient with POAG
 Managing the Patient with POAG Vision Institute Annual Fall Conference Mitchell W. Dul, OD, MS, FAAO mdul@sunyopt.edu Richard J. Madonna, MA, OD, FAAO rmadonna@sunyopt.edu Ocular Hypertension (OHT) Most
Managing the Patient with POAG Vision Institute Annual Fall Conference Mitchell W. Dul, OD, MS, FAAO mdul@sunyopt.edu Richard J. Madonna, MA, OD, FAAO rmadonna@sunyopt.edu Ocular Hypertension (OHT) Most
Enhanced Scheme Evaluation Project (ESEP) Robert Harper On behalf of the ESEP team
 Enhanced Scheme Evaluation Project (ESEP) Robert Harper On behalf of the ESEP team ESEP Team Project Leads: Robert Harper and John Lawrenson Co-applicants: David Edgar, Cecilia Fenerty, David Henson, Ian
Enhanced Scheme Evaluation Project (ESEP) Robert Harper On behalf of the ESEP team ESEP Team Project Leads: Robert Harper and John Lawrenson Co-applicants: David Edgar, Cecilia Fenerty, David Henson, Ian
PRACTICAL APPROACH TO MEDICAL MANAGEMENT OF GLAUCOMA
 PRACTICAL APPROACH TO MEDICAL MANAGEMENT OF GLAUCOMA DR. RAVI THOMAS, DR. RAJUL PARIKH, DR. SHEFALI PARIKH IJO MAY 2008 PRESENTER AT JDOS : DR. RAHUL SHUKLA T.N. SHUKLA EYE HOSPITAL TERMINOLOGY POAG: PRIMARY
PRACTICAL APPROACH TO MEDICAL MANAGEMENT OF GLAUCOMA DR. RAVI THOMAS, DR. RAJUL PARIKH, DR. SHEFALI PARIKH IJO MAY 2008 PRESENTER AT JDOS : DR. RAHUL SHUKLA T.N. SHUKLA EYE HOSPITAL TERMINOLOGY POAG: PRIMARY
Antibiotic Prophylaxis Against Infective Endocarditis Implementation Advice Consultation Feedback Form
 Antibiotic Prophylaxis Against Infective Endocarditis Implementation Advice Consultation Feedback Form Consultation is the opportunity for anyone with an interest in this implementation advice to provide
Antibiotic Prophylaxis Against Infective Endocarditis Implementation Advice Consultation Feedback Form Consultation is the opportunity for anyone with an interest in this implementation advice to provide
Treatments for Open-Angle Glaucoma. A Review of the Research for Adults
 Treatments for Open-Angle Glaucoma A Review of the Research for Adults Is This Information Right for Me? Yes, this information is for you if: Your eye doctor has said that you have open-angle glaucoma,
Treatments for Open-Angle Glaucoma A Review of the Research for Adults Is This Information Right for Me? Yes, this information is for you if: Your eye doctor has said that you have open-angle glaucoma,
Cataract Surgery Criteria Based Access Protocol Supporting people in Dorset to lead healthier lives
 NHS Dorset Clinical Commissioning Group Cataract Surgery Criteria Based Access Protocol Supporting people in Dorset to lead healthier lives 1. INTRODUCTION AND SCOPE NHS DORSET CLINICAL COMMISSIONING GROUP
NHS Dorset Clinical Commissioning Group Cataract Surgery Criteria Based Access Protocol Supporting people in Dorset to lead healthier lives 1. INTRODUCTION AND SCOPE NHS DORSET CLINICAL COMMISSIONING GROUP
Glaucoma Care Pathway
 PRIMARY CARE OPTOMETRY UPDATE Glaucoma Care Pathway June 2017 Colleagues, As the work of Developing Eyecare Partnerships progresses and the Health and Social Care Board (HSCB) continue to work with primary
PRIMARY CARE OPTOMETRY UPDATE Glaucoma Care Pathway June 2017 Colleagues, As the work of Developing Eyecare Partnerships progresses and the Health and Social Care Board (HSCB) continue to work with primary
Evidence-Based Clinical Practice Guideline. Treatment & Management of Ocular Disease
 Evidence-Based Clinical Practice Guideline Treatment & Management of Ocular Disease Effective November 29, 2017 Treatment & Management Of Ocular Disease Clinical Practice Guideline The objective of this
Evidence-Based Clinical Practice Guideline Treatment & Management of Ocular Disease Effective November 29, 2017 Treatment & Management Of Ocular Disease Clinical Practice Guideline The objective of this
Alcohol interventions in secondary and further education
 National Institute for Health and Care Excellence Guideline version (Draft for Consultation) Alcohol interventions in secondary and further education NICE guideline: methods NICE guideline Methods
National Institute for Health and Care Excellence Guideline version (Draft for Consultation) Alcohol interventions in secondary and further education NICE guideline: methods NICE guideline Methods
Financial Disclosure. Prostaglandin Analogs (PGs) The Glaucoma Grab Bag: Practical Guidelines for Effective Glaucoma Therapy
 The Glaucoma Grab Bag: Practical Guidelines for Effective Glaucoma Therapy Danica J. Marrelli, OD, FAAO University of Houston College of Optometry Financial Disclosure I have received I have received speaking
The Glaucoma Grab Bag: Practical Guidelines for Effective Glaucoma Therapy Danica J. Marrelli, OD, FAAO University of Houston College of Optometry Financial Disclosure I have received I have received speaking
A proposal for collaboration between the Psychometrics Committee and the Association of Test Publishers of South Africa
 A proposal for collaboration between the Psychometrics Committee and the Association of Test Publishers of South Africa 27 October 2015 Table of contents Introduction... 3 Overview of the Association of
A proposal for collaboration between the Psychometrics Committee and the Association of Test Publishers of South Africa 27 October 2015 Table of contents Introduction... 3 Overview of the Association of
nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd
 nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Low back pain and sciatica in over 16s NICE quality standard
 March 2017 Low back pain and sciatica in over 16s NICE quality standard Draft for consultation This quality standard covers the assessment and management of non-specific low back pain and sciatica in young
March 2017 Low back pain and sciatica in over 16s NICE quality standard Draft for consultation This quality standard covers the assessment and management of non-specific low back pain and sciatica in young
Class Update with New Drug Evaluation: Glaucoma Drugs
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Abbreviated Update: Ophthalmic Glaucoma Agents
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Abbreviated Update: Ophthalmic Glaucoma Agents Month/Year
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Abbreviated Update: Ophthalmic Glaucoma Agents Month/Year
Glaucoma. National Guideline Centre. Glaucoma: diagnosis and management. NICE guideline 81. October Final. Methods, evidence and recommendations
 National Guideline Centre Final Glaucoma Glaucoma: diagnosis and management NICE guideline 81 Methods, evidence and recommendations October 2017 Final Commissioned by the National Institute for Health
National Guideline Centre Final Glaucoma Glaucoma: diagnosis and management NICE guideline 81 Methods, evidence and recommendations October 2017 Final Commissioned by the National Institute for Health
Case scenarios: Patient Group Directions
 Putting NICE guidance into practice Case scenarios: Patient Group Directions Implementing the NICE guidance on Patient Group Directions (MPG2) Published: March 2014 [updated March 2017] These case scenarios
Putting NICE guidance into practice Case scenarios: Patient Group Directions Implementing the NICE guidance on Patient Group Directions (MPG2) Published: March 2014 [updated March 2017] These case scenarios
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Centre for Clinical Practice Surveillance Programme
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Centre for Clinical Practice Surveillance Programme Clinical guideline CG28: Depression in children and young people: identification and management in
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Centre for Clinical Practice Surveillance Programme Clinical guideline CG28: Depression in children and young people: identification and management in
National Institute for Health and Clinical Excellence
 National Institute for Health and Clinical Excellence NICE pilot quality standards for social care: Consultation on the draft quality standard for Dementia: Supporting people to live well with dementia
National Institute for Health and Clinical Excellence NICE pilot quality standards for social care: Consultation on the draft quality standard for Dementia: Supporting people to live well with dementia
Integrated Diabetes Care in Oxfordshire -patient's perspective. Avril Surridge
 Integrated Diabetes Care in Oxfordshire -patient's perspective Avril Surridge Today How does diabetes care in Oxfordshire look like from a patient s perspective? Good things What could be improved? National
Integrated Diabetes Care in Oxfordshire -patient's perspective Avril Surridge Today How does diabetes care in Oxfordshire look like from a patient s perspective? Good things What could be improved? National
Surveillance report Published: 8 June 2017 nice.org.uk. NICE All rights reserved.
 Surveillance report 2017 Antenatal and postnatal mental health: clinical management and service guidance (2014) NICE guideline CG192 Surveillance report Published: 8 June 2017 nice.org.uk NICE 2017. All
Surveillance report 2017 Antenatal and postnatal mental health: clinical management and service guidance (2014) NICE guideline CG192 Surveillance report Published: 8 June 2017 nice.org.uk NICE 2017. All
Glaucoma Disease Progression Role of Intra Ocular Pressure. Is Good Enough, Low Enough?
 Glaucoma Disease Progression Role of Intra Ocular Pressure Is Good Enough, Low Enough? Glaucoma Diseases Progression Key Considerations Good number of patients may be diagnosed only after some damage the
Glaucoma Disease Progression Role of Intra Ocular Pressure Is Good Enough, Low Enough? Glaucoma Diseases Progression Key Considerations Good number of patients may be diagnosed only after some damage the
This paper contains analysis of the results of these processes and sets out the programme of future development.
 Fitness to Practise Committee, 14 February 2013 HCPC witness support programme Executive summary and recommendations Introduction This paper outlines the approach taken by HCPC in relation to witness management
Fitness to Practise Committee, 14 February 2013 HCPC witness support programme Executive summary and recommendations Introduction This paper outlines the approach taken by HCPC in relation to witness management
Summary of the risk management plan (RMP) for Izba (travoprost)
 EMA/14138/2014 Summary of the risk management plan (RMP) for Izba (travoprost) This is a summary of the risk management plan (RMP) for Izba, which details the measures to be taken in order to ensure that
EMA/14138/2014 Summary of the risk management plan (RMP) for Izba (travoprost) This is a summary of the risk management plan (RMP) for Izba, which details the measures to be taken in order to ensure that
Guideline scope Hypertension in adults (update)
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline scope Hypertension in adults (update) This guideline will update the NICE guideline on hypertension in adults (CG127). The guideline will be
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline scope Hypertension in adults (update) This guideline will update the NICE guideline on hypertension in adults (CG127). The guideline will be
Aneurin Bevan University Health Board. Directorate of Ophthalmology. Action Plan Ophthalmology Thematic Review Final Version 2015/16 WET AMD
 Aneurin Bevan University Health Board Directorate of Ophthalmology Action Plan Ophthalmology Thematic Review Final Version 2015/16 WET AMD No. Item Action Responsible Person(s) Timeframe 1 Patient Referrals
Aneurin Bevan University Health Board Directorate of Ophthalmology Action Plan Ophthalmology Thematic Review Final Version 2015/16 WET AMD No. Item Action Responsible Person(s) Timeframe 1 Patient Referrals
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Scope for Partial Update
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Scope for Partial Update 1 Guideline title Anxiety: management of generalised anxiety disorder in adults in primary, secondary and community care (update)
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Scope for Partial Update 1 Guideline title Anxiety: management of generalised anxiety disorder in adults in primary, secondary and community care (update)
DISTANCE LEARNING COURSE. Scope of the Eye Care Practice , BSM Consulting All rights reserved.
 DISTANCE LEARNING COURSE Scope of the Eye Care Practice 2008 2012, BSM Consulting All rights reserved. Table of Contents OVERVIEW... 1 THREE O'S IN EYE CARE... 1 ROUTINE VS. MEDICAL EXAMS... 2 CONTACT
DISTANCE LEARNING COURSE Scope of the Eye Care Practice 2008 2012, BSM Consulting All rights reserved. Table of Contents OVERVIEW... 1 THREE O'S IN EYE CARE... 1 ROUTINE VS. MEDICAL EXAMS... 2 CONTACT
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE QUALITY AND OUTCOMES FRAMEWORK (QOF) INDICATOR DEVELOPMENT PROGRAMME Briefing paper QOF indicator area: Diabetes dietary review Potential output: Recommendations
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE QUALITY AND OUTCOMES FRAMEWORK (QOF) INDICATOR DEVELOPMENT PROGRAMME Briefing paper QOF indicator area: Diabetes dietary review Potential output: Recommendations
Get a grip on your glaucoma Booklet 1
 Information for Patients Glaucoma services Welcome! Get a grip on your glaucoma Booklet 1 Group-based Glaucoma information Course Session 1 What happens in session 1? Finding out what questions you have
Information for Patients Glaucoma services Welcome! Get a grip on your glaucoma Booklet 1 Group-based Glaucoma information Course Session 1 What happens in session 1? Finding out what questions you have
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE SCOPE. Dementia: the management of dementia, including the use of antipsychotic medication in older people
 NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE 1 Guideline title SCOPE Dementia: the management of dementia, including the use of antipsychotic medication in older people 1.1 Short title Dementia 2 Background
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE 1 Guideline title SCOPE Dementia: the management of dementia, including the use of antipsychotic medication in older people 1.1 Short title Dementia 2 Background
Local Healthwatch Quality Statements. February 2016
 Local Healthwatch Quality Statements February 2016 Local Healthwatch Quality Statements Contents 1 About the Quality Statements... 3 1.1 Strategic context and relationships... 5 1.2 Community voice and
Local Healthwatch Quality Statements February 2016 Local Healthwatch Quality Statements Contents 1 About the Quality Statements... 3 1.1 Strategic context and relationships... 5 1.2 Community voice and
MAINTAINING COMPLIANCE IN GLAUCOMA PATIENTS. by : Abdalla El-Sawy, M.D. Professor of Ophthalmology, Benha Faculty of Medicine.
 MAINTAINING COMPLIANCE IN GLAUCOMA PATIENTS by : Abdalla El-Sawy, M.D. Professor of Ophthalmology, Benha Faculty of Medicine. The problem is especially critical for eye doctors who manage patients with
MAINTAINING COMPLIANCE IN GLAUCOMA PATIENTS by : Abdalla El-Sawy, M.D. Professor of Ophthalmology, Benha Faculty of Medicine. The problem is especially critical for eye doctors who manage patients with
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE. Type 2 diabetes: the management of type 2 diabetes (update)
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Type 2 diabetes: the management of type 2 diabetes (update) 1.1 Short title Type 2 diabetes (update) 2 Background a) The National
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Type 2 diabetes: the management of type 2 diabetes (update) 1.1 Short title Type 2 diabetes (update) 2 Background a) The National
Surveillance report Published: 13 April 2017 nice.org.uk. NICE All rights reserved.
 Surveillance report 2017 Antisocial behaviour and conduct disorders in children and young people: recognition and management (2013) NICE guideline CG158 Surveillance report Published: 13 April 2017 nice.org.uk
Surveillance report 2017 Antisocial behaviour and conduct disorders in children and young people: recognition and management (2013) NICE guideline CG158 Surveillance report Published: 13 April 2017 nice.org.uk
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Methadone and buprenorphine for the management of opioid dependence
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Appraisal Methadone and buprenorphine for the management of opioid dependence Comments received from Consultees and Commentators on the draft scope
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Appraisal Methadone and buprenorphine for the management of opioid dependence Comments received from Consultees and Commentators on the draft scope
UK National Screening Committee. Vision Screening in Children aged 4-5 years - an evidence review. Consultation comments pro-forma
 UK National Screening Committee Vision Screening in Children aged 4-5 years - an evidence review Consultation comments pro-forma Organisation: College of Optometrists (jointly with the Optical Confederation,
UK National Screening Committee Vision Screening in Children aged 4-5 years - an evidence review Consultation comments pro-forma Organisation: College of Optometrists (jointly with the Optical Confederation,
Re: Inhaled insulin for the treatment of type 1 and type 2 diabetes comments on the Assessment Report for the above appraisal
 Dear Alana, Re: Inhaled insulin for the treatment of type 1 and type 2 diabetes comments on the Assessment Report for the above appraisal Thank you for allowing the Association of British Clinical Diabetologists
Dear Alana, Re: Inhaled insulin for the treatment of type 1 and type 2 diabetes comments on the Assessment Report for the above appraisal Thank you for allowing the Association of British Clinical Diabetologists
National Institute for Health and Clinical Excellence
 National Institute for Health and Clinical Excellence CG54: Urinary Tract Infections in children Guideline Review Consultation Comments Table 18 vember 1 December 2010 Stakeholder GDG member NO The algorithm
National Institute for Health and Clinical Excellence CG54: Urinary Tract Infections in children Guideline Review Consultation Comments Table 18 vember 1 December 2010 Stakeholder GDG member NO The algorithm
Elements for a Public Summary. Overview of disease epidemiology
 VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Epidemiology of the disease Incidence and prevalence Increased pressure in the eye occurs in more than 100 million people, and
VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Epidemiology of the disease Incidence and prevalence Increased pressure in the eye occurs in more than 100 million people, and
PRESCRIBING BY RADIOGRAPHERS: A VISION PAPER
 PRESCRIBING BY RADIOGRAPHERS: A VISION PAPER 1 INTRODUCTION 1.1 The Review of Prescribing, Supply & Administration of Medicines (Crown II) Final Report was submitted to The Secretary of State for Health
PRESCRIBING BY RADIOGRAPHERS: A VISION PAPER 1 INTRODUCTION 1.1 The Review of Prescribing, Supply & Administration of Medicines (Crown II) Final Report was submitted to The Secretary of State for Health
ROBERT L. SHIELDS, M.D. GLAUCOMA SURGERY AND TREATMENT
 1 CURRICULUM VITAE ROBERT L. SHIELDS, M.D. GLAUCOMA SURGERY AND TREATMENT University of Colorado Health Eye Center Cherry Creek 2000 South Colorado Boulevard Annex Building, Suite 100 Denver, Colorado
1 CURRICULUM VITAE ROBERT L. SHIELDS, M.D. GLAUCOMA SURGERY AND TREATMENT University of Colorado Health Eye Center Cherry Creek 2000 South Colorado Boulevard Annex Building, Suite 100 Denver, Colorado
