Blood Drug Analysis Test No Summary Report
|
|
|
- Margaret Wilson
- 5 years ago
- Views:
Transcription
1 Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Blood Drug Analysis Test No Summary Report This test was sent to 135 participants. The sample sets contained blood samples from three cases, each with an individual case scenario. Each case sample consisted of two grey-topped vials containing human blood with various drugs/metabolites. Participants were requested to examine these items and report their findings. Data were returned from 98 participants (72.6% response rate) and are compiled into the following tables: Page Manufacturer's Information Summary Comments Table 1: Item 1 Results Table 2: Item 2 Results Table 3: Item 3 Results Table 4: Additional Comments Appendix: Data Sheet This report contains the data received from the participants in this test. Since these participants are located in many countries around the world, and it is their option how the samples are to be used (e.g., training exercise, known or blind proficiency testing, research and development of new techniques, etc.), the results compiled in the Summary Report are not intended to be an overview of the quality of work performed in the profession and cannot be interpreted as such. The Summary Comments are included for the benefit of participants to assist with maintaining or enhancing the quality of their results. These comments are not intended to reflect the general state of the art within the profession. Participant results are reported using a randomly assigned "WebCode". This code maintains participant's anonymity, provides linking of the various report sections, and will change with every report.
2 Manufacturer's Information The sample sets contained blood samples from three cases, each with an individual case scenario. Each case sample consisted of two grey-topped vials containing 10mL of human blood. Participants were asked to analyze the blood samples and report the presence of any drugs/metabolites, any quantitative data obtained (including uncertainty), methods used, and any additional comments. SAMPLE PREPARATION- The human blood used in this test was from the same lot, which tested negative for a variety of common controlled substances prior to being obtained from a commercial supplier. A stock solution of 1.0mg/mL of each drug in methanol or acetonitrile was used to spike each item. These solutions were obtained in sealed ampoules and were not opened until needed for production. Items were prepared at separate times using the following procedure, and different glassware was used for each item. ITEMS 1, 2, and 3 (PREPARATION): Item preparation consisted of adding a predetermined amount of drug stock solution to a beaker containing human whole blood. It was stirred using a magnetic stirrer for 10 minutes before pipetting 10mL of the mixture into each of the pre-labeled vials, which contained 20mg Potassium Oxalate and 100mg Sodium Fluoride. The vials were sealed and inverted 8-10 times to mix the chemicals in the vials with the blood solution. All vials were placed in a refrigerator immediately after production until the sample sets were prepared. SAMPLE SET ASSEMBLY: Each sample set contained two vials of each of Items 1, 2, and 3 and was placed into a Department of Transportation regulated shipping container. Each sample pack was labeled and returned to the refrigerator until shipment. VERIFICATION- Laboratories that conducted predistribution analysis of the samples reported consistent results that were comparable to the preparation drug concentrations. Item 1 Drug ( Concentration ) Item 2 Drug ( Concentration ) Item 3 Drug ( Concentration ) Zolpidem (250 ng/ml) Heroin (120 ng/ml) Diazepam (400 ng/ml) Morphine (1000 ng/ml) Nordiazepam (450 ng/ml) 6-monoacetylmorphine (175 ng/ml) Please note that the Preparation Value is the value used for calculations during the test preparation phase and may not necessarily represent the final concentration of the samples. It is advised to wait for the Grand Mean statistics available in the Summary and Individual Reports before evaluating performance. ( 2 )
3 Summary Comments This test was designed to allow participants to assess their proficiency in the examination for the presence and concentration of drugs and/or metabolites in blood. Each participant was supplied with two vials containing 10mL of human blood spiked with differing drugs and/or metabolites for each of three case scenarios. Participants were asked to report the presence of any drugs/metabolites, any quantitative data obtained (including uncertainty), methods used, and any additional comments. (Refer to the Manufacturer's Information for preparation details.) Of the 97 participants who reported results for Item 1, 92 (94.8%) reported the presence of zolpidem. The remaining 5 participants reported no controlled drugs/metabolites detected. Of the 97 participants who reported results for Item 2, 87 (89.7%) reported the presence of morphine and/or monoacetylmorphine. Six participants reported no drugs/metabolites detected. Of the 98 participants who reported results for Item 3, 95 (96.9%) reported the presence of diazepam and/or nordiazepam. Additional drugs/metabolites were reported in all samples by many participants, including diphenhydramine, valproic acid, and phenytoin. If a laboratory indicated that the "reported" quantitative result was a single determination, the conclusive quantitative result was included in the raw data table. The raw data was used to calculate the grand mean and standard deviation for each item. One participant was determined to have "extreme" data (±5 STD from grand mean) for Zolpidem in Item 1. The grand mean and standard deviation are supplied to assist the participants and accrediting bodies in determining the acceptability of results. CTS acknowledges that drugs/metabolites other than those used to spike the samples were present. The blood used in producing the samples was tested for a variety of common controlled substances. CTS is examining methods to obtain whole blood with fewer contaminating drugs/metabolites from prescription and over the counter medications. In order to narrow the focus of testing in future Blood-Drug proficiency tests, CTS will also be providing a possible list of drugs/metabolites. ( 3 )
4 Reported Results - Item 1 What drugs/metabolites were detected in Item 1? TABLE 1A Item 1 Results Item 1 Scenario: A 20-year-old female was found partially undressed in a bedroom by friends at a party. She was taken to the hospital after friends observed her exhibiting memory loss, confusion, agitation, and dizziness. Item Contents and Preparation Concentration: Zolpidem (250 ng/ml) Analyte Reported Qualitative Only Reported Concentration Uncertainty Units 23C9Q4 Zolpidem 0.27 ± 0.08 mg/l 2H8PF2 Zolpidem mg/l 2J47RY Zolpidem 2UJVTP Zolpidem 0.32 ± 0.10 mg/l 2W63QQ Zolpidem % mg/l Phenytoin % mg/l Trazodone % mg/l 34K3J9 Zolpidem 340 +/- 100 or 30% ng/ml 38LT7N Zolpidem 220 ng/ml 3B2MJZ ZOLPIDEM 3BJC72 ZOLPIDEN[sic] 3BJC9L Zolpidem 3GUF3U zolpidem 3XE7Z9 Zolpidem 0.29 ± 0.09 mg/l 3YVBLD Zolpidem ( 4 )
5 TABLE 1A Item 1 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units 46RLYW Zolpidem % 4FDLZY Zolpidem 0.31 ± 0.09 mg/l 4KPHRF No drugs/metabolites detected 4PW6AX Zolpidem 6G487V Zolpidem 6JQWEF Zolpidem 0.35 ± 0.11 mg/l 6R4WMW Zolpidem mg/l 76M28Y Zolpidem Trazodone Diphenhydramine 8MHUTW Zolpidem ng/ml 8YVRZL Zolpidem 94NFUX Zolpidem 9CBJTK Zolpidem mg/l B96VBZ Zolpidem mg/l BJP7FL BX9K4E No drugs/metabolites detected Zolpidem 0.33 ± 0.10 mg/l C49BZP zolpidem CAW9CJ Zolpidem ( 5 )
6 TABLE 1A Item 1 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units CGEL6Q Zolpidem CTBMUD Zolpidem mcg/ml D8LN2N Zolpidem DERB36 Zolpidem / ug/ml ng/ml Ibuprofen EB6ZFK zolpidem valproic acid acetaminophen caffeine phenytoin ibuprofen EE4D2N Zolpidem ng/ml EGUGUV Zolpidem mg/l F38PKE Zolpidem ng/ml FCW7L3 Zolpidem mg/l FMVXYA Zolpidem ng/ml GHDBLF Zolpidem GN2VQG Zolpidem GPEP8H zolpidem ( 6 )
7 TABLE 1A Item 1 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units GX8MAK zolpidem Greater than 200 ng/ml phenytoin diphenhdramine[sic] H4T3EG Zolpidem 240 +/- 43 % HA2G7E Zolpidem HVY836 Zolpidem 0.32 ±0.10 mg/l HWBQZG Zolpidem HZDK6D Zolpidem 197 +/- 18 ng/ml Phenytoin J27LU2 Zolpidem 0.33 ±0.10 mg/l JB4WRB JGTD7L No drugs/metabolites detected Zolpidem ng/ml JRWDX9 Zolpidem 253 +/-20% ng/ml KGNDLC Zolpidem KLKZDE Zolpidem KRBCE3 Zolpidem 0.37 ± 0.11 mg/l KY96KB Zolpidem L6C99N Zolpidem mg/l L77U42 Zolpidem mg/l ( 7 )
8 TABLE 1A Item 1 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units M3Y32D Zolpidem MC6ZUN Zolpidem 0.31 ± 0.09 mg/l MF9PLW Zolpidem % mg/l MHHJFB Zolpidem N3WWZA NCLGNG No drugs/metabolites detected No drugs/metabolites detected NCPYPZ Zolpidem 0.34 ± 30% mg/l NGHZTZ Zolpidem 0.34 ± 0.10 mg/l NHQD9G Zolpidem ng/ml P27WTQ Zolpidem % mg/l P793BA Zolpidem Phenytoin PT4W84 Zolpidem ng/ml PU33NX Zolpidem 0.33 ±0.10 mg/l PUFJW6 QXF699 Zolpidem Valproic Acid - do not report < 10 ng/ml Zolpidem RAMGU2 Zolpidem RBZ2MB Zolpidem ng/ml ( 8 )
9 TABLE 1A Item 1 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units RMVYQB zolpidem TM8CRC Zolpidem 290 ± 90 ng/ml TY4C72 Zolpidem Ibuprofen Valproic acid UDWDC8 Zolpidem UGKUYM zolpidem UZQDMA Zolpidem 0.26 ± 0.08 mg/l VDCYTN Zolpidem Cotinine Caffeine VEVMEP Zolpidem mg/l WA9CT6 Zolpidem WB494K Zolpidem 260 N/A ng/ml WL3ZFR Zolpidem ng/ml X9CFJ2 Zolpidem XUUQHE Zolpidem /- 30% mg/l XXT8U9 Zolpidem Trazodone Diphenhydramine ( 9 )
10 TABLE 1A Item 1 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units XYLVFP Zolpidem 0.33 ± 0.10 mg/l Y9Q6H9 Zolpidem YA4WBP Zolpidem 0.31 ± 0.09 mg/l YANN8F zolpidem 240 ng/ml YXAH98 Zolpidem ZU6AJM Zolpidem 0.31 ± 0.09 (30%) mg/l ZYKVEF Zolpidem Response Summary for Item 1 Participants: 97 Zolpidem: No drugs/metabolites detected: 5 92 Other: 21 Totals may add up to more than the total number of participants because some participants reported multiple drugs/metabolites. ( 10 )
11 Raw Data - Item 1 List of raw data determinations in ng/ml. TABLE 1B Item 1 Raw Data - Zolpidem 23C9Q4 2H8PF2 2UJVTP 2W63QQ 34K3J9 38LT7N 3XE7Z9 3YVBLD 46RLYW 4FDLZY 6JQWEF 6R4WMW 8MHUTW 9CBJTK B96VBZ BX9K4E CTBMUD DERB36 EE4D2N EGUGUV F38PKE FCW7L3 FMVXYA H4T3EG HVY836 HZDK6D J27LU2 JGTD7L JRWDX9 Raw Data (ng/ml) Participant Mean , , , X ( 11 )
12 Item 1 Raw Data - Zolpidem KRBCE3 L6C99N L77U42 MC6ZUN MF9PLW NCPYPZ NGHZTZ NHQD9G P27WTQ PT4W84 PU33NX RBZ2MB TM8CRC UZQDMA VEVMEP WB494K WL3ZFR XUUQHE XYLVFP YA4WBP YANN8F ZU6AJM Raw Data (ng/ml) Participant Mean Statistical Analysis for Item 1- Zolpidem Grand Mean Standard Deviation Number of Participants Included Number of Participants Excluded 50 1 Number of Participants without Raw Data or Data that was not reported in ng/ml 41 ( 12 )
13 Item 1 Raw Data - 2W63QQ Analyte Raw Data (ng/ml) Phenytoin Trazodone Participant Mean Statistical Analysis for Item 1- Other Please note statistical analysis is not provided for other drug responses. ( 13 )
14 Reporting Procedures - Item 1 If quantitative analysis was performed, the reported concentrations are: TABLE 1C - Item 1 23C9Q4 2H8PF2 2UJVTP 2W63QQ 34K3J9 38LT7N 3XE7Z9 3YVBLD 46RLYW 4FDLZY 6JQWEF 6R4WMW 8MHUTW 9CBJTK B96VBZ BX9K4E CTBMUD DERB36 EE4D2N EGUGUV F38PKE FCW7L3 FMVXYA GHDBLF GX8MAK H4T3EG HVY836 HZDK6D J27LU2 JGTD7L JRWDX9 Quantitative Reporting Procedures Single points were used for Zolpidem and Phenytoin. The average of two values was used for Trazodone. Undiluted Results Reported; Other Results Confirmed Approximate Level The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. ( 14 )
15 TABLE 1C - Item 1 KRBCE3 L6C99N L77U42 MC6ZUN MF9PLW NCPYPZ NGHZTZ NHQD9G P27WTQ PT4W84 PU33NX RBZ2MB TM8CRC UZQDMA VEVMEP WB494K WL3ZFR XUUQHE XYLVFP YA4WBP YANN8F ZU6AJM Quantitative Reporting Procedures The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. Response Summary for Item 1 Participants: 53 A single determination: The mean of duplicate/several determinations: Other: 40 (75.5%) 11 (20.8%) 2 (3.8%) ( 15 )
16 Method of Analysis - Item 1 TABLE 1D Method of Analysis 23C9Q4 2H8PF2 2J47RY 2UJVTP 2W63QQ 34K3J9 38LT7N 3B2MJZ 3BJC72 3BJC9L 3GUF3U 3XE7Z9 3YVBLD 46RLYW 4FDLZY 4KPHRF 4PW6AX 6G487V Method Screening Confirmatory Quantitation LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS LC/MS/MS LC/MS/MS LC QTOF MS GC/NPD LC/MS/MS LC/MS/MS ( 16 )
17 TABLE 1D Method of Analysis 6JQWEF 6R4WMW 76M28Y 8MHUTW 8YVRZL 94NFUX 9CBJTK B96VBZ BJP7FL BX9K4E C49BZP CAW9CJ CGEL6Q CTBMUD D8LN2N DERB36 EB6ZFK EE4D2N EGUGUV Method Screening Confirmatory Quantitation LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS LC/MS/MS LC/MS/MS LC/MS/MS GC/NPD LC/MS/MS LC/MS/MS LC/MS/MS ( 17 )
18 TABLE 1D Method of Analysis F38PKE FCW7L3 FMVXYA GHDBLF GN2VQG GPEP8H GX8MAK H4T3EG HA2G7E HVY836 HWBQZG HZDK6D J27LU2 JB4WRB JGTD7L JRWDX9 KGNDLC Method Screening Confirmatory Quantitation LC/MS/MS LC/MS/MS LC/MS GC/NPD LC/MS/MS GC/NPD LC/MS/MS LC/MS/MS LC/MS/MS LC/TOF LC/MS/MS ( 18 )
19 TABLE 1D Method of Analysis KLKZDE KRBCE3 KY96KB L6C99N L77U42 M3Y32D MC6ZUN MF9PLW MHHJFB N3WWZA NCLGNG NCPYPZ NGHZTZ NHQD9G P27WTQ P793BA PT4W84 PU33NX PUFJW6 QXF699 Method Screening Confirmatory Quantitation LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC-QTOF LC-DAD (UV) LC/MS/MS LC/MS/MS ( 19 )
20 TABLE 1D Method of Analysis RAMGU2 RBZ2MB RMVYQB TM8CRC TY4C72 UDWDC8 UGKUYM UZQDMA VDCYTN VEVMEP WA9CT6 WB494K WL3ZFR X9CFJ2 XUUQHE XXT8U9 XYLVFP Y9Q6H9 Method Screening Confirmatory Quantitation LC/MS/MS LC/MS/MS LC/MS/MS GC\NPD LC/MS/MS LC/MS LC/MS/MS LC/MS/MS LC/MS/MS LC/MS LC/MS/MS LC/MS/MS ( 20 )
21 TABLE 1D Method of Analysis YA4WBP YANN8F YXAH98 ZU6AJM ZYKVEF Method Screening Confirmatory Quantitation LC/MS/MS LC/MS/MS LC/MS/MS LC/MS/MS LC-TOFMS Response Summary for Item 1 Participants: 97 Screening Confirmatory Quantitation : : LC/MS: LC/MS/MS: ( 21 )
22 Additional Comments for Item 1 TABLE 1E 23C9Q4 2H8PF2 2J47RY 2UJVTP 2W63QQ 38LT7N 3GUF3U 3XE7Z9 3YVBLD 46RLYW 4FDLZY 6JQWEF 6R4WMW 79PP4N 8MHUTW 94NFUX 9CBJTK B96VBZ C49BZP CGEL6Q D8LN2N DERB36 EE4D2N EGUGUV FMVXYA GN2VQG mepivacaine Mepivacaine Item 1 - Comments Acid/Base Internal Standard (lot# ) containing Prazepam, Nalorphine, Methanol, and Methohexital. cut-off concentration (ng/ml) for Zolpidem: 20. Base extract 100 (ng/ml) for Zolpidem: 10. Internal standard used in testing for the reported analyte was mepivacaine. Zolpidem: Internal standard - D6-Zolpidem. Administrative LOQ set at 0.01 mg/l. Trazodone: Internal standard- D6-Trazodone. Administrative LOQ set at 0.01 mg/l. Phenytoin: Internal standard - D10-Phenytoin. Phenytoin and Trazodone detected at similar levels in all three items. Confirmation limit of detection - 20ng/mL. Internal standard - Zolpidem-D6. The following drugs were found but are not being reported: phenytoin, caffeine, valproic acid, ibuprofen, acetaminophen, naproxen. Internal standard: mepivacaine Salting-out assisted extraction; ISTD = D3-Methadone; analysis by UPLC - QTOF MS (Waters) Promazine used as internal standard. Uncertainty calculated using 6 injections of UTAK control. CTS #1 sample quanted in duplicate but diluted samples (x2) not used for sample calculation. Mepivacaine - I. S. Mepivacaine Internal standard - mepivacaine No Analysis - scenario does not meet case acceptance criteria. IS: mepivacaine, LOD: 25 ng/ml. The [State] Police only report zolpidem qualitatively. Internal Standard: Zolpidem-d6. Low cut-off level (ng/ml): 5 ng/ml. Limit of Quantitation: 20 ng/ml. Quantitative Range: 20 to 600 ng/ml. Mepivacaine (int. std.) Screened negative by ELISA. Used barbital and SKF-525a for internal standards. drug screen internal standard- promazine Zolpidem: LOD is 12.5 ng/ml. Diphenhydramine: LOD is 12.5 ng/ml - not reported, weak peak. Internal Standard: Prazepam. Internal Standards: methaqualone, barbital, Zolpidem D7. Limit of detection for Zolpidem 50 ng/ml. Internal standard - mepivacaine Mepivicaine[sic] Zolpidem-D6 was used as an internal standard. Internal standard: Prazepam, zolpidem LOD: 12.5 ng/ml. Weak diphenhydramine detected. ( 22 )
23 TABLE 1E GPEP8H GX8MAK HVY836 HZDK6D J27LU2 JGTD7L JRWDX9 KLKZDE KRBCE3 L77U42 MC6ZUN MF9PLW NGHZTZ NHQD9G P27WTQ P793BA PU33NX QXF699 RAMGU2 TM8CRC TY4C72 UDWDC8 UZQDMA Item 1 - Comments Phenytoin, valproic acid, naproxen, ibuprofen, and caffeine were also confirmed in the blood sample. The drugs were not reported independently in section 1-2 because they were confirmed in all three PT samples. Phenytoin and diphenhydramine were identified in all three items and are suspected to be artifacts of sample preparation. Zolpidem was quantitated using mepivacaine on the internal standard. At this time we do not quantitate Phenytoin. However, the GCMS peak appeared to be a sufficient strength so as to possibly surpass the 10 ng/ml threshold. Therefore, we are reporting Phenytoin. We are reporting Phenytoin as a presumptive positive since it was analyzed by only one type of test. Internal standard: mepivacaine. Calibration range: ug/l. Limit of reporting: 25 ug/l. Internal standard - mepivacaine, Limit of report - 25 ug/l CTS items 1, 2 and 3 all contained small amounts of several additional drugs/medications, including valproic acid, phenytoin and naproxen. Prazepam - Internal standard. Zolpidem LOD ng/ml. ISTD = mepivacaine. Calibration range = ng/ml for zolpidem. LOR= 25 ng/ml. Zolpidem not detected by immunoassay procedure. mepivacaine Mepivacaine is the internal standard. Zolpidem quanted off of the internal standard mepivacaine. The limit of report is mg/l. Zolpidem is reported with 30% variance making the result 0.30 mg/l +/ mg/l. Additional tests for other drugs indicative during screening were less than the limit of report. Internal Standard was mepivacaine. Limit of report for zolpidem is 25 ng/ml. Internal standard: mepivacaine. Limit of report: 25 ng/ml. Internal standard used for quantification: prazepam Hexobarbital (weak acid neutral fraction) and Methapyrilene (basic fraction) were used as internal standards Internal standard: Mepivacaine is used: Prazepam. ELISA cutoff for zolpidem: 20 ng/ml. GCMS LOD for Zolpidem: 12.5 ng/ml. Diphenhydramine detected: not reported Internal standard used: cocaine-d3, methaqualone, 6-MAM-d3, morphine-d3, methamphetamine-d9, ketamine-d4. Relatively low level of caffeine, phenytoin, trazodone, carbamazepine and paroxetine detected in sample. Due to level, caffeine, phenytoin, trazodone, carbamazepine and paroxetine were not reported. Internal std = mepivacaine. Also detected, but not reported due to poor quality of mass spec and/or quantitation below limit of report: acetaminophen, gabapentin, caffeine, trazodone, diphenhydramine, phenytoin, carbamazepine, paroxetine, aripiprazole. Valproic acid and Ibuprofen were identified by library match only from the extraction to rule out the presence of GHB. Promazine was used as the Internal Standard for the drug screen analysis for GC\NPD & GC\MS. internal standard: mepivacaine ( 23 )
24 TABLE 1E VEVMEP WA9CT6 WB494K X9CFJ2 XUUQHE XXT8U9 XYLVFP YA4WBP YANN8F YXAH98 ZU6AJM Internal standard: mepivacaine Item 1 - Comments The internal standard used was Prazepam. The cut-off concentration for Zolpidem is 20 ng/ml. The LOD for Zolpidem is 12.5 ng/ml when a base extraction is performed and a is used. Diphenhydramine was identified but not reported due to a poor mass spectrum. The LOD for Diphenhydramine is 12/5 ng/ml when a base extraction is performed and a is used. Valproic acid and phenytoin were detected in this sample. Valproic acid present, phenytoin less than 100 ng/ml. Our limit of detection is 2000 ng/ml and 1.0 mg/ml, respectively for Valproic acid and phenytoin. Internal standard (IS): Prazepam. Zolpidem LOD: 12.5 ng/ml. Internal standard used - mepivacaine Preliminary testing indicates the presence of zolpidem, trazodone, and diphenhydramine; however, no confirmation test was conducted. ISTD - Mepivacaine Internal std used was mepiv. nordiazepam-d5 used as internal standard in method, not used for drug quantitation. Contemporaneous spiked blood samples at three known drug concentrations analysed simultaneously with samples for drug quantitation. Limits of reporting () - zolpidem 46 ng/ml. Drug concentrations are usually reported in mg/l units (except for THC which is ug/l), and to one significant figure. For example drug concentrations mg/l are reported to one decimal place, concentrations > 5.0 mg/l are reported as whole numbers only. Concentrations less than 0.1 mg/l are reported to two decimal places. We currently do not quant Zolpidem due to updating extraction methods. Internal standard - mepivacaine. LOD/LOR - 25 ng/ml ( 24 )
25 Reported Results - Item 2 What drugs/metabolites were detected in Item 2? Item 2 Scenario: TABLE 2A Item 2 Results A 42-year-old male with a history of opiate abuse was found dead in his cell in a detention center. The previous night his cell had been searched for suspicion of drug trafficking, but no drugs were found. Item Contents and Preparation Concentration: Heroin (120 ng/ml) Morphine (1000 ng/ml) 6-monoacetylmorphine (175 ng/ml) Analyte Reported Qualitative Only Reported Concentration Uncertainty Units 23C9Q4 Morphine mg/l 6-MAM 2H8PF2 Morphine mg/l 6-monoacetyl-morphine 2J47RY Morphine 6-monoacetylmorphine (6-MAM) 2UJVTP Morphine 1.3 ± 0.3 mg/l 6-monoacetyl-morphine (6-MAM) 2W63QQ Morphine % mg/l 6-Monoacetylmorphine % mg/l Trazodone % mg/l Phenytoin % mg/l 34K3J9 Morphine 1200 ± 300 or 25% ng/ml 6-MAM 38LT7N Morphine 1000 ng/ml 6-monoacetylmorphine Positive 3B2MJZ MORPHINE 3BJC72 MORFINE[sic] ( 25 )
26 TABLE 2A Item 2 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units 3BJC9L Morphine 6-Monoacetylmorphine 3GUF3U No drugs/metabolites detected 3XE7Z9 Morphine 1.3 ± 0.3 mg/l 6-monoacetylmorphine 3YVBLD Morphine Monoacetylmorphine RLYW Morphine 6-Monoacetylmorphine 4FDLZY Morphine 1.3 ± 0.3 mg/l 6-monoacetylmorphine 4KPHRF Opiates panel 4PW6AX 6G487V 6-Acetylmorphine Morphine 6-monoacetylmorphine 6JQWEF Morphine 1.3 +/- 0.3 mg/l 6-MAM 6R4WMW Morphine mg/l 6-monoacetylmorphine 76M28Y Morphine 6-Monoacetylmorphine Trazodone Diphenhydramine 8MHUTW Morphine ng/ml 6-MAM ( 26 )
27 TABLE 2A Item 2 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units 8YVRZL Morphine ng/ml 6-acetylmorphine ng/ml 94NFUX Morphine 6-Monoacetylmorphine 24 1 ng/ml 9CBJTK Morphine ng/ml 6-monoacetylmorphine B96VBZ Morphine mg/l 6-monoacetylmorphine BJP7FL No drugs/metabolites detected BX9K4E Morphine 1.3 ± 0.3 mg/l 6-MAM C49BZP No drugs/metabolites detected CAW9CJ CGEL6Q 6-MAM morphine 6-monoacetylmorphine CTBMUD Morphine ng/ml 6-monoacetylmorphine D8LN2N Morphine 6-monoacetylmorphine DERB36 Morphine /- 0.1ug/mL ng/ml 6-acetylmorphine <200 N/A ng/ml Phenytoin Naproxen Ibuprofen Diphenhydramine ( 27 )
28 TABLE 2A Item 2 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units EB6ZFK valproic acid phenytoin ibuprofen caffeine acetaminophen EE4D2N Morphine ng/ml 6-monoacetylmorphine EGUGUV Morphine mg/l 6-monoacetylmorphine F38PKE Morphine > ng/ml 6-monoacetylmorphine (MAM) 38.0 ng/ml FCW7L3 Morphine ng/ml 6-monoacetylmorphine Paroxetine <50 ng/ml FMVXYA Morphine > ng/ml 6-Acetylmorphine (6-MAM) 57 6 ng/ml GHDBLF Morphine 6-Monoacetylmorphine 17 1 ng/ml GN2VQG Morphine 6-monoacetylmorphine GPEP8H 6-acetylmorphine GX8MAK morphine greater than 200 ng/ml phenytoin diphenhdramine[sic] H4T3EG Morphine % 6-Monoacetylmorphine ( 28 )
29 TABLE 2A Item 2 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units HA2G7E Morphine 6-MAM HVY836 Morphine 1.2 ± 0.3 mg/l 6-monoacetyl-morphine HWBQZG Morphine 6-Monoacetylmorphine HZDK6D Morphine >1000 +/- 130 ng/ml 6-acetylmorphine 33 +/- 4 ng/ml Phenytoin J27LU2 Morphine 1.3 ±0.3 mg/l 6-monoacetylmorphine JB4WRB Morphine 6-monoacetylmorphine JGTD7L Morphine ng/ml 6-MAM JRWDX9 Morphine /-20% ng/ml 6-Monoacetylmorphine KGNDLC Morphine 1390 ng/ml 6-acetylmorphine 66 ng/ml KLKZDE Morphine 6-monoacetylmorphine KRBCE3 Morphine 1.3 ±0.3 mg/l 6-monoacetylmorphine KY96KB Morphine 6-MAM ( 29 )
30 TABLE 2A Item 2 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units L6C99N Morphine mg/l 6-monoacetylmorphine L77U42 Morphine mg/l 6-monoacetylmorphine M3Y32D Morfina[sic] 6-MAM MC6ZUN Morphine 1.3 ± 0.3 mg/l 6-monoacetylmorphine MF9PLW Morphine % mg/l 6-monoacetylmorphine MHHJFB N3WWZA NCLGNG 6-monoacetylmorphine 12 1 ng/ml Morphine 1, ng/ml No drugs/metabolites detected NCPYPZ Morphine 1.3 ± 25% mg/l 6-MAM NGHZTZ Morphine 1.3 ± 0.3 mg/l 6-monoacetyl morphine (6MAM) NHQD9G Morphine ng/ml 6-monoacetylmorphine P27WTQ Morphine % mg/l 6-monoacetylmorphine P793BA Morphine Phenytoin PT4W84 No drugs/metabolites detected ( 30 )
31 TABLE 2A Item 2 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units PU33NX Morphine 1.3 ±0.3 mg/l 6-MAM PUFJW6 QXF699 Morphine 6-Acetylmorphine Valproic Acid - do not report < 10 ng/ml Morphine 6-MAM(monoacetylmorphine) RAMGU2 Morphine 6-monoacetylmorphine RBZ2MB Morphine ng/ml 6-MAM RMVYQB No drugs/metabolites detected TM8CRC Morphine 1300 ± 300 ng/ml 6-monoacetylmorphine TY4C72 UDWDC8 unknown Opiate(s) Morphine 6-Monoacetyl-Morpine[sic] UGKUYM opiates UZQDMA Morphine 1.2 ± 0.3 mg/l 6-monoacetylmorphine VDCYTN Morphine 1136 ±204 ng/ml 6-monoacetylmorphine Cotinine Caffeine ( 31 )
32 TABLE 2A Item 2 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units VEVMEP Morphine mg/l 6-monoacetylmorphine WA9CT6 Morphine 6-monoacetylmorphine WB494K Morphine 1600 ng/ml 6-Monoacetylmorphine WL3ZFR Morphine ng/ml 6-Monoacetylmorphine 49 7 ng/ml X9CFJ2 Morphine 6-monoacetylmorphine XUUQHE Morphine 1.3 ± 25% mg/l XXT8U9 Morphine 6-O-Monoacetylmorphine Zolpidem Trazodone Diphenhydramine XYLVFP Morphine 1.4 ± 0.4 mg/l 6-monoacetylmorphine Y9Q6H9 Morphine 6-monoacetylmorphine 47 2 ng/ml YA4WBP Morphine 1.3 ± 0.3 mg/l 6-MAM YANN8F morphine 1200 ng/ml 6-MAM 80 ng/ml YXAH98 Morphine 6-monoacetylmorphine 43 2 ng/ml ( 32 )
33 TABLE 2A Item 2 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units ZU6AJM Morphine 1.3 ± 0.3 (25%) mg/l 6-MAM ZYKVEF morphine 1200 ng/ml acetylmorphine Response Summary for Item 2 Participants: 97 Morphine: 6-monoacetylmorphine: No drugs/metabolites detected: Other: 27 Totals may add up to more than the total number of participants because some participants reported multiple drugs/metabolites. ( 33 )
34 Raw Data - Item 2 List of raw data determinations in ng/ml. TABLE 2B Item 2 Raw Data - Morphine 23C9Q4 2H8PF2 2UJVTP 2W63QQ 34K3J9 38LT7N 3XE7Z9 Raw Data (ng/ml) 1, , , , , , , Participant Mean 1, , , , , , , YVBLD 4FDLZY 6JQWEF 6R4WMW 8MHUTW 8YVRZL 94NFUX 9CBJTK B96VBZ BX9K4E CTBMUD DERB36 EE4D2N EGUGUV F38PKE FCW7L3 FMVXYA H4T3EG HVY836 HZDK6D J27LU2 JGTD7L JRWDX9 1, , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , ( 34 )
35 TABLE 2B Item 2 Raw Data - Morphine KGNDLC KRBCE3 L6C99N L77U42 MC6ZUN MF9PLW N3WWZA Raw Data (ng/ml) 1, , , , , , , Participant Mean 1, , , , , , , NCPYPZ NGHZTZ NHQD9G P27WTQ PU33NX RBZ2MB TM8CRC UZQDMA VDCYTN VEVMEP WB494K WL3ZFR XUUQHE XYLVFP Y9Q6H9 YA4WBP YANN8F YXAH98 ZU6AJM ZYKVEF 1, , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , Statistical Analysis for Item 2 - Morphine Grand Mean 1, Standard Deviation Number of Participants Included Number of Participants Excluded 57 0 Number of Participants without Raw Data or Data that was not reported in ng/ml 26 ( 35 )
36 TABLE 2B Item 2 Raw Data - 6-monoacetylmorphine 2W63QQ Raw Data (ng/ml) Participant Mean YVBLD 8YVRZL 94NFUX F38PKE FMVXYA GHDBLF HZDK6D KGNDLC MHHJFB WL3ZFR Y9Q6H9 YANN8F YXAH Statistical Analysis for Item 2-6-monoacetylmorphine Grand Mean Standard Deviation Number of Participants Included Number of Participants Excluded 14 0 Number of Participants without Raw Data or Data that was not reported in ng/ml 67 ( 36 )
37 TABLE 2B Item 2 Raw Data - Analyte Raw Data (ng/ml) 2W63QQ Trazodone Phenytoin Participant Mean Statistical Analysis for Item 2- Other Please note statistical analysis is not provided for other drug responses. ( 37 )
38 Reporting Procedures - Item 2 If quantitative analysis was performed, the reported concentrations are: TABLE 2C - Item 2 23C9Q4 2H8PF2 2UJVTP 2W63QQ 34K3J9 38LT7N 3XE7Z9 3YVBLD 4FDLZY 6JQWEF 6R4WMW 8MHUTW 8YVRZL 94NFUX 9CBJTK B96VBZ BX9K4E CTBMUD DERB36 EE4D2N EGUGUV F38PKE FCW7L3 FMVXYA GHDBLF GX8MAK H4T3EG HVY836 HZDK6D J27LU2 JGTD7L Quantitative Reporting Procedures The mean of duplicate/several determinations. least dilute, truncated Diluted Results Reported (Morphine); Other Results Confirmed Appoximate Level/Range The data from was higher than the highest calibrator. So data from was used only (for morphine). The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. ( 38 )
39 TABLE 2C - Item 2 JRWDX9 KGNDLC KRBCE3 L6C99N L77U42 MC6ZUN MF9PLW MHHJFB N3WWZA NCPYPZ NGHZTZ NHQD9G P27WTQ PU33NX RBZ2MB TM8CRC UZQDMA VDCYTN VEVMEP WB494K WL3ZFR XUUQHE XYLVFP Y9Q6H9 YA4WBP YANN8F YXAH98 ZU6AJM ZYKVEF Quantitative Reporting Procedures The mean of duplicate/several determinations. a reduced volume testing with calculations to reflect volume adjustment The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. The mean of duplicate/several determinations. Response Summary for Item 2 Participants: 60 A single determination: The mean of duplicate/several determinations: Other: 46 (76.7%) 10 (16.7%) 4 (6.7%) ( 39 )
40 Method of Analysis - Item 2 TABLE 2D Method of Analysis Method Screening Confirmatory Quantitation 23C9Q4 2H8PF2 LC/MS/MS LC/MS/MS 2J47RY 2UJVTP 2W63QQ 34K3J9 38LT7N 3B2MJZ LC/MS/MS LC/MS LC/MS/MS 3BJC72 LC/MS 3BJC9L 3GUF3U 3XE7Z9 LC/MS/MS LC/MS/MS ( 40 )
41 TABLE 2D Method of Analysis Method Screening Confirmatory Quantitation 3YVBLD LC/MS/MS LC QTOF MS 46RLYW GC/NPD 4FDLZY LC/MS/MS 4KPHRF 4PW6AX LC/MS/MS 6G487V 6JQWEF 6R4WMW LC/MS/MS 76M28Y LC/MS/MS 8MHUTW LC/MS/MS 8YVRZL 94NFUX 9CBJTK LC/MS LC/MS/MS ( 41 )
42 TABLE 2D Method of Analysis Method Screening Confirmatory Quantitation B96VBZ BJP7FL BX9K4E LC/MS/MS C49BZP CAW9CJ LC/MS/MS CGEL6Q CTBMUD GC/NPD LC/MS/MS D8LN2N DERB36 EB6ZFK EE4D2N EGUGUV F38PKE FCW7L3 FMVXYA LC/MS/MS LC/MS/MS ( 42 )
43 TABLE 2D Method of Analysis Method Screening Confirmatory Quantitation GHDBLF LC/MS GN2VQG GPEP8H GX8MAK H4T3EG LC/MS HA2G7E LC/MS/MS HVY836 HWBQZG HZDK6D J27LU2 LC/MS/MS GC/NPD JB4WRB LC/MS/MS JGTD7L JRWDX9 LC/TOF LC/MS/MS ( 43 )
44 TABLE 2D Method of Analysis Method Screening Confirmatory Quantitation KGNDLC LC/MS/MS KLKZDE KRBCE3 LC/MS/MS KY96KB LC/MS/MS L6C99N L77U42 M3Y32D LC/MS/MS MC6ZUN MF9PLW LC/MS/MS LC/MS/MS MHHJFB LC/MS N3WWZA NCLGNG NCPYPZ NGHZTZ LC/MS/MS LC/MS/MS ( 44 )
45 TABLE 2D Method of Analysis Method Screening Confirmatory Quantitation NHQD9G P27WTQ LC-QTOF LC-DAD (UV) P793BA PT4W84 PU33NX PUFJW6 LC/MS/MS QXF699 RAMGU2 RBZ2MB RMVYQB TM8CRC LC/MS/MS LC/MS/MS LC/MS/MS TY4C72 UDWDC8 GC-NPD UGKUYM ( 45 )
46 TABLE 2D Method of Analysis Method Screening Confirmatory Quantitation UZQDMA VDCYTN VEVMEP LC/MS/MS WA9CT6 WB494K WL3ZFR LC/MS/MS X9CFJ2 XUUQHE XXT8U9 LC/MS XYLVFP Y9Q6H9 YA4WBP YANN8F YXAH98 LC/MS/MS LC/MS/MS LC/MS/MS ( 46 )
47 TABLE 2D Method of Analysis Method Screening Confirmatory Quantitation ZU6AJM ZYKVEF LC/MS/MS LC-TOFMS Response Summary for Item 2 Participants: 97 Screening Confirmatory Quantitation : : LC/MS: LC/MS/MS: ( 47 )
48 Additional Comments for Item 2 TABLE 2E Item 2 - Comments 23C9Q4 2H8PF2 2J47RY 2UJVTP 2W63QQ 38LT7N 3GUF3U 3XE7Z9 3YVBLD 46RLYW 4FDLZY 6JQWEF 6R4WMW 79PP4N 8MHUTW 94NFUX 9CBJTK B96VBZ BX9K4E C49BZP mepivacaine, nalorphine nalorphine Acid/Base Internal Standard (lot# ) containing Prazepam, Nalorphine, Methanol and Methohexital. Mepivacaine Internal Standard (lot# ) containing Mepivacaine and Methanol. cut-off concentration (ng/ml) for 6-MAM and Morphine, respectively: Not available, 50. Base extract LOD (ng/ml) for 6-MAM and Morphine, respectively: 150, not available. Base extract, TMS derivative LOD (ng/ml) for 6-MAM: 25. Base extract, di-tms derivative LOD (ng/ml) for Morphine: 25. The internal standard used was mepivacaine. Morphine: Internal standard - D3-Morphine. 6-Monoacetylmorphine: Internal standard - D3-6-Monoacetylmorphine. Trazodone: Internal standard- D6-Trazodone. Administrative LOQ set at 0.01 mg/l. Phenytoin: Internal standard - D10-Phenytoin. Phenytoin and Trazodone detected at similar levels in all three items. Confirmation limit of detection (Morphine) - 10ng/mL. Confirmation limit of detection (6-MAM) - 15ng/mL. Internal standard (Morphine and 6-MAM) - Morphine-D3. The following were detected but are not being reported: phenytoin, caffeine, valproic acid, ibuprofen, acetaminophen, naproxen. Internal standard: Nalorphine Quantitation by UPLC TQD (Waters); using deuterated internal standards; LOD for both analytes is 2 ng/ml Promazine used for internal standard for butyl acetate extraction ran on GC/NPD and. Nalorphine used for internal standard for opiate confirmation ran on. Nalorphine - I.S. Nalorphine Internal standard - nalorphine No analysis - scenario does not meet case acceptance criteria. IS: Mepivacaine, LOD: 3.1 ng/ml Morpine[sic] reported qualitatively based on [Laboratory] protocol. The sample concentration was greater than the established calibration range of ng/ml. For 6-Monoacetylmorphine - Internal Standard: 6-Monoacetylmorphine-d6. Low cut-off level: 6.25 ng/ml. For Morphine - Internal Standard: Morphide-d6, Low cut-off level: 12.5 ng/ml, Limit of Quantitation: 25 ng/ml, Quantitative Range: 25 to 800 ng/ml. [Raw data value for Morphine: ">800"] nalorphine (int. std) The data from was higher than the highest calibrator for morphine. The testing was reported, with a reduced volume on Calculations were done to account for the reduced volume and only this data was used for the results. Screened positive by ELISA for opiates. Used SKF-525a and barbital as internal standards. ( 48 )
49 TABLE 2E CAW9CJ CGEL6Q D8LN2N DERB36 EB6ZFK EE4D2N EGUGUV FCW7L3 FMVXYA GHDBLF GN2VQG GPEP8H GX8MAK HVY836 HZDK6D J27LU2 JGTD7L JRWDX9 KLKZDE KRBCE3 L77U42 MC6ZUN Item 2 - Comments Morphine appeared present but analyst was unable to complete test because the LC/MS/MS had a collision cell error and had to be taken out of service. Contacted Service Engineer and they are on vacation and will not be able to repair the instrument before the results are due. drug screen internal standard - promazine. opiate confirmation internal standard- nalorphine. Morphine: LOD 50 ng/ml. 6-MAM: LOD 150 ng/ml, TMS - 25 ng/ml. Diphenhydramine - not reported, weak peak. IS - Prazepam, Nalorphine - ditms, Mepivacaine. [Raw data value for 6-Acetylmorphine: <200, <200.] Internal Standards: methaqualone, barbital, nalorphine. Limits of detection: morphine 25 ng/ml; 6-Acetylmorphine 25 ng/ml Item 2 blood sample screened positive for opiates but currently our confirmation method is not optimized for codeine, morphine or 6- acetylmorphine. Internal standard - nalorphine Nalorphine 1. Morphine concentration determined using dilution factor 4; 2. For paroxetine, limit of quantitation raised due to 1:4 dilution (i.e., LOQ = 50 ng/ml, upon dilution 4xLOQ = 200 ng/ml) [Raw data values for Paroxetine: <50, <200, <200] 6-MAM-D6 and Morphine-D3 were used as internal standards. The laboratories upper limit of quantitation is 1,000 ng/ml for morphine. Morphine was greater than Upper Limit of Quantitation. Presence Only. Internal standards: prazepam & nalorphine. Morphine LOD: 50 ng/ml (di-tms derivative). 6-monoacetylmorphine LOD: 150 ng/ml. 6-monoactylmorphine[sic] - TMS derivative: 25 ng/ml. Weak diphenhydramine detected. Phenytoin, valproic acid, naproxen, ibuprofen, and caffeine were also confirmed in the blood sample. The drugs were not reported independently in section 2-2 [Table 2A-Item 2 Results] because they were confirmed in all three PT samples. Phenytoin and diphenhydramine were identified in all three items and are suspected to be artifacts of sample preparation. Nalorphine was used as the internal standard to quantitate morphine. Mepivacaine was used as the internal standard to report 6-monoacetylmorphine. See page 3 of 9 [Table 1E-Item 1-Comments] Internal standard: Nalorphine. Calibration range: ug/l. Limit of reporting: 3.1ug/L. Note: First quantitative run item #2 was not on the calibration curve. Second quantitative run item #2 was diluted to a 1:10 dilution in order for level to fall on calibration curve. Internal standard - nalorphine. Limit of report μg/l. As for items 1 [Table 1E-Item 1-Comments]. Prazepam, nalorphine - Internal standards. 6-monoacetylmorphine LOD - 25 ng/ml (derivatized), 150 ng/ml (underivatized). Morphine LOD: 50 ng/ml (derivatized). Internal std = nalorphine. LOR morphine = 3.1 ng/ml (mg/l). *Calibration range = ng/ml*. Reduced volume (0.5 ml rather than 3 ml) used to obtain reported concentration ( ng/ml x 6 = ). Nalorphine Nalorphine, Mepivacaine was used to positively identify 6 MAM ( 49 )
50 TABLE 2E MF9PLW MHHJFB N3WWZA NCPYPZ NGHZTZ NHQD9G P27WTQ P793BA PT4W84 PU33NX QXF699 RAMGU2 TM8CRC TY4C72 UDWDC8 UZQDMA VDCYTN VEVMEP Item 2 - Comments Morphine quantitated off of internal standard, nalorphine. Both morphine + 6- monoacetyl-morphine butylated like nalorphine during testing. Morphine limit of report is 3.1 ug/l. It's reported with 25% variance making the result 1.2 mg/l +/- 0.3 mg/l. 6-monoacetyl-morphine qualitatively identified based of mepivacaine internal standard. Additional tests for other drugs indicative during screening were less than the limit of report. Morphine was above the ULOQ, report as presence only. Internal Standards: Cocaine-D3, Benzoylecgonine-D3, Codeine-D6, Morphine-D6. Limits of Detection: Cocaine = 10 ng/ml, Benzoylecgonine = 20 ng/ml, Codeine = 20 ng/ml, Hydrocodone = 20 ng/ml, Hydromorphone = 20 ng/ml, Morphine = 20 ng/ml, Oxycodone = 10 ng/ml. 6-MAM = 6 - monoacetylmorphine Initial quantitation of morphine was above/outside calibration curve - repeated w/ lesser volume. Internal standard used was nalorphine, w/ both drugs being butylated during extraction procedure. Limit of report for morphine is 3.1 ng/ml. Internal standard: nalorphine. Limit of report 3.1 ng/ml. Internal standard used for quantification: D3-morphine Hexobarbital and Methapyrilene were used for internal standards. Confirmation of morphine was done by TMS Deriviatization[sic]. Opiate positive on the EMIT screen, No Basic Drugs Detected on the basic drug screen performed by. Internal standard: Mepivacaine GCMS is Prazepam, nalorphine (for derivatized samples), mepivacaine. ELISA opiate cutoff: 50 ng/ml (morphine). GCMS LODs morphine: 50 ng/ml (derivatized), 6-MAM: 25ng/mL (derivatized). Diphenhydramine detected: not reported (poor MS). Internal standard used: cocaine-d3, methaqualone, 6-MAM-d3, morphine-d3, methamphetamine-d9, ketamine-d4. Relatively low level of caffeine, phenytoin, trazodone, carbamazepine and paroxetine detected in sample. Due to level, caffeine, phenytoin, trazodone, carbamazepine and paroxetine were not reported. Int. std used for morphine = nalorphine. Initial quantitation was higher than highest calibrator of 200 ng/ml. Reported concentration comes from a reduced volume extraction as well as a calibration curve up to 400 ng/ml. Also detected, but not reported due to poor quality of mass spec and/or quantitation below limit of report: acetaminophen, gabapentin, caffeine, trazodone, diphenhydramine, phenytoin, carbamazepine, paroxetine, aripiprazole. [From Table 2B-Item 2 Raw Data-Morphine: First value "Not used".] Item #2 screened positive for opiates but [Laboratory] was unable to confirm the presence of an opiate. Promazine was used as the Internal Standard for the drug screen analysis for GC\NPD & GC\MS. Nalorphine was used as the Internal Standard for the opiate confirmation on GC\MS. internal standards: mepivacaine, nalorphine Morphine: Internal Std = Morphine D3, LOD = 25 ng/ml Internal standard: Nalorphine ( 50 )
51 TABLE 2E WA9CT6 WB494K WL3ZFR X9CFJ2 XUUQHE XXT8U9 XYLVFP Y9Q6H9 YA4WBP YANN8F YXAH98 ZU6AJM ZYKVEF Item 2 - Comments The internal standard used for the Base extraction was Prazepam. The internal standards used for the derivatized extraction was Prazepam, Mepivacaine, and Nalorphine. The cut-off concentration for Morphine is 50 ng/ml. The LOD for Morphine is 50 ng/ml when a derivatized extraction is performed and a is used. The LOD for 6-Monoacetyl-morphine is 150 ng/ml when a base extraction is performed and 25 ng/ml when a derivatized extraction is performed and a is used. Diphenhydramine was identified but not reported due to a poor mass spectrum. The LOD for Diphenhydramine is 12.5 ng/ml when a base extraction is performed and a is used. Phenytoin was detected at less than 1.0 ug/ml (LOD = 0.50 ug/ml). Valproic was detected qualitatively only (LOD= 2.0 ug/ml) The intiial[sic] analysis for morphine gave us a result that was above our calibration range (10-500ng/ml). The sample was then re-extracted and this time there was a 1/2 dilution performed to bring the value within the calibration range (450.87). The result was then multiplied by two to get the final concentration of 901 ng/ml IS: Prazepam, Nalorphine (TMS). 6-MAM LOD: 150 ng/ml non-derivatized, 25 ng/ml derivatized. Morphine LOD: 50 ng/ml derivatized. Internal standard used - nalorphine Preliminary testing indicates the presence of morphine, 6-o-monoacetylmorphine, zolpidem, trazodone, and diphenhydramine; however, no confirmation test was conducted. ISTD - nalorphine. 3mL test, sampled at reduced volume of 0.5mL Morphine was reported as presence only because the concentration was above the calibrated range for this compound in this extraction. Internal std used is nalorphine. nordiazepam-d5 used as internal standard in method, not used for drug quantitation. Contemporaneous spiked blood samples at three known drug concentrations analysed simultaneously with samples for drug quantitation. MDMA-d5 used as internal standard to quantitate 6-MAM in LC/MS/MS method. morphine-d3 used as an internal standard to quantitate morphine in LC/MS/MS method. Limits of reporting () - 6-MAM 82 ng/ml, morphine 21 ng/ml; (LC/MS/MS) - 6-MAM 10 ng/ml, morphine 10 ng/ml. Drug concentrations are usually reported in mg/l units (except for THC which is ug/l), and to one significant figure. For example drug concentrations mg/l are reported to one decimal place, concentrations > 5.0 mg/l are reported as whole numbers only. Concentrations less than 0.1 mg/l are reported to two decimal places. Morphine value fell above ULOQ (400ng/mL), reported as presence only per our policy. Internal standard - nalorphine. LOD/LOR ng/ml. The morphine level would be reported as approximate as the sample required dilution prior to analysis. Our measurement of uncertainty does not cover this situation. The morphine level was determined using solid-phase extraction and a deuterated internal standard. The limit of quantitation is 20 ng/ml for this assay. ( 51 )
52 Reported Results - Item 3 What drugs/metabolites were detected in Item 3? Item 3 Scenario: TABLE 3A Item 3 Results A 50-year-old female was pulled over for drifting in and out of traffic lanes. The officer observed horizontal gaze nystagmus, slurred speech, and poor balance. A breath alcohol test resulted in 0.00 percent. Blood was drawn approximately 90 minutes later. Item Contents and Preparation Concentration: Diazepam (400 ng/ml) Nordiazepam (450 ng/ml) Analyte Reported Qualitative Only Reported Concentration Uncertainty Units 23C9Q4 Diazepam 0.42 ± 0.13 mg/l Nordiazepam 0.50 ± 0.15 mg/l 2H8PF2 Diazepam mg/l Nordiazepam mg/l 2J47RY Diazepam Nordiazepam 2UJVTP Diazepam 0.49 ± 0.15 mg/l Nordiazepam 0.56 ± 0.17 mg/l 2W63QQ Diazepam % mg/l Nordiazepam mg/l Trazodone % mg/l Phenytoin mg/l 34K3J9 Diazepam 460 ± 140 or 30% ng/ml Nordiazepam 460 ± 140 or 30% ng/ml 38LT7N Diazepam 330 ng/ml Nordiazepam 410 ng/ml 3B2MJZ DIAZEPAM NORDIAZEPAM ( 52 )
53 TABLE 3A Item 3 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units 3BJC72 DIAZEPAM NORDIAZEPAM 3BJC9L Diazepam Nordiazepam 4-Acetoxt DMT 3GUF3U diazepam nordiazepam 3XE7Z9 Diazepam 0.42 ± 0.13 mg/l Nordiazepam 0.42 ± 0.13 mg/l 3YVBLD Diazepam Nordiazepam RLYW Diazepam % Nordiazepam % Phenytoin 4FDLZY Diazepam 0.51 ± 0.15 mg/l Nordiazepam 0.58 ± 0.17 mg/l 4KPHRF Benzodiazepines panel 4PW6AX Diazepam Nordiazepam 6G487V Diazepam Nordiazepam 6JQWEF Diazepam 0.47 ± 0.14 mg/l Nordiazepam 0.51 ± 0.15 mg/l ( 53 )
54 TABLE 3A Item 3 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units 6R4WMW Diazepam mg/l Nordiazepam mg/l 76M28Y Diazepam Nordiazepam Trazodone Diphenhydramine 79PP4N Diazepam Nordiazepam 8MHUTW Diazepam ng/ml Nordiazepam ng/ml 8YVRZL Diazepam ng/ml Nordiazepam ng/ml 94NFUX Diazepam ng/ml Nordiazepam ng/ml 9CBJTK Diazepam mg/l Nordiazepam mg/l B96VBZ Diazepam mg/l Nordiazepam mg/l BJP7FL diazepam BX9K4E Diazepam 0.56 ± 0.17 mg/l Nordiazepam 0.50 ± 0.15 mg/l C49BZP diazepam nordiazepam ( 54 )
55 TABLE 3A Item 3 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units CAW9CJ Diazepam Nordiazepam CGEL6Q diazepam nordiazepam CTBMUD Diazepam ng/ml Nordiazepam ng/ml D8LN2N Diazepam Nordiazepam DERB36 Diazepam 370 +/ ug/ml ng/ml Nordiazepam / ug/ml ng/ml Ibuprofen Phenytoin EB6ZFK diazepam nordiazepam phenytoin valproic acid caffeine acetaminophen ibuprofen EE4D2N Diazepam ng/ml Nordiazepam ng/ml EGUGUV Diazepam mg/l Nordiazepam mg/l ( 55 )
56 TABLE 3A Item 3 Results Analyte Reported Qualitative Only Reported Concentration Uncertainty Units F38PKE Diazepam ng/ml Nordiazepam ng/ml FCW7L3 Diazepam mg/l Nordiazepam mg/l FMVXYA Diazepam ng/ml Nordiazepam ng/ml GHDBLF Diazepam ng/ml Nordiazepam ng/ml GN2VQG Diazepam Nordiazepam GPEP8H diazepam nordiazepam GX8MAK diazepam 394 ng/ml N-desmethyldiazepam 438 ng/ml diphenhydramine phenytoin H4T3EG Diazepam Nordiazepam HA2G7E Diazepam Nordiazepam HVY836 Diazepam 0.45 ± 0.14 mg/l Nordiazepam 0.52 ± 0.16 mg/l ( 56 )
Urine Drug Analysis Test No Summary Report
 Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Urine Drug Analysis Test No. 15-5671 Summary Report This test was sent to 72 participants. Each sample set contained three cases with individual
Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Urine Drug Analysis Test No. 15-5671 Summary Report This test was sent to 72 participants. Each sample set contained three cases with individual
Urine Drug Analysis Test No Summary Report
 Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Urine Drug Analysis Test No. 18-5671 Summary Report A sample set contained one specimen bottle of human urine for each of the three case scenarios.
Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Urine Drug Analysis Test No. 18-5671 Summary Report A sample set contained one specimen bottle of human urine for each of the three case scenarios.
Blood Drug Analysis Test No Summary Report
 Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Blood Drug Analysis Test No. 18-5661 Summary Report The sample sets contained blood samples from three cases, each with an individual case scenario.
Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Blood Drug Analysis Test No. 18-5661 Summary Report The sample sets contained blood samples from three cases, each with an individual case scenario.
Blood Drug Analysis Test No Summary Report
 Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Blood Drug Analysis Test No. 17-5661 Summary Report This test was sent to 130 participants. The sample sets contained blood samples from three
Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Blood Drug Analysis Test No. 17-5661 Summary Report This test was sent to 130 participants. The sample sets contained blood samples from three
Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS
 Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia Department
Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia Department
Forensic Toxicology Scope of Testing and Detection Limits
 Forensic Toxicology Scope of Testing and Detection Limits Table of Contents QUALITATIVE ANALYSES... 2 Volatile Screen by GC/FID... 2 Carbon Monoxide by Microdiffusion... 2 Ethylene Glycol by GC/MS... 2
Forensic Toxicology Scope of Testing and Detection Limits Table of Contents QUALITATIVE ANALYSES... 2 Volatile Screen by GC/FID... 2 Carbon Monoxide by Microdiffusion... 2 Ethylene Glycol by GC/MS... 2
Validation Report for the Neogen Fentanyl Kit for ELISA Screening of Whole Blood and Urine Specimens
 Validation Report for the Neogen Fentanyl Kit for ELISA Screening of Whole Blood and Urine Specimens This document describes the validation of a Neogen Fentanyl kit for the semi-quantitative analysis of
Validation Report for the Neogen Fentanyl Kit for ELISA Screening of Whole Blood and Urine Specimens This document describes the validation of a Neogen Fentanyl kit for the semi-quantitative analysis of
Evaluation of the Impact of Expanding ELISA Screening in DUID Investigations. Aileen Lu*, Karen S. Scott, Aya Chan-Hosokawa, and Barry K.
 Evaluation of the Impact of Expanding ELISA Screening in DUID Investigations Aileen Lu*, Karen S. Scott, Aya Chan-Hosokawa, and Barry K. Logan FSF Emerging Forensic Scientist Award Oral Presentation Disclosure
Evaluation of the Impact of Expanding ELISA Screening in DUID Investigations Aileen Lu*, Karen S. Scott, Aya Chan-Hosokawa, and Barry K. Logan FSF Emerging Forensic Scientist Award Oral Presentation Disclosure
NeoSal Oral Fluid Collection System Solutions for Forensic Drug Detection
 PERFORMANCE OF THE NEOSAL ORAL FLUID COLLECTION SYSTEM Neogen develops and manufactures a comprehensive range of ELISA test kits and accessories for forensic drug detection. Neogen offers solutions to
PERFORMANCE OF THE NEOSAL ORAL FLUID COLLECTION SYSTEM Neogen develops and manufactures a comprehensive range of ELISA test kits and accessories for forensic drug detection. Neogen offers solutions to
Lyndsey Knoy, D-ABFT-FT Forensic Scientist Washington State Toxicology Laboratory.
 Lyndsey Knoy, D-ABFT-FT Forensic Scientist Washington State Toxicology Laboratory Lyndsey.Knoy@wsp.wa.gov Forensic Toxicology is an interdisciplinary science that analyzes blood, fluid and/or tissues for
Lyndsey Knoy, D-ABFT-FT Forensic Scientist Washington State Toxicology Laboratory Lyndsey.Knoy@wsp.wa.gov Forensic Toxicology is an interdisciplinary science that analyzes blood, fluid and/or tissues for
Toxicological Investigation of Drug Impaired Driving Toxicology Laboratory Survey
 Toxicological Investigation of Drug Impaired Driving Toxicology Laboratory Survey Kayla J. Lowrie, MS, Jennifer Turri, BS, Jill Yeakel, MSFS, Barry K. Logan, PhD, DABFT, Arcadia University, 450 S. Easton
Toxicological Investigation of Drug Impaired Driving Toxicology Laboratory Survey Kayla J. Lowrie, MS, Jennifer Turri, BS, Jill Yeakel, MSFS, Barry K. Logan, PhD, DABFT, Arcadia University, 450 S. Easton
A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography
 Application Note LCMS-109 A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography Time of Flight (LC-TOF) Mass Spectrometry Introduction Many clinical
Application Note LCMS-109 A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography Time of Flight (LC-TOF) Mass Spectrometry Introduction Many clinical
Marihuana Identification and THC Quantification Test No Summary Report
 Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Marihuana Identification and THC Quantification Test. 17-503 Summary Report Each sample pack consisted of three questioned samples containing
Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Marihuana Identification and THC Quantification Test. 17-503 Summary Report Each sample pack consisted of three questioned samples containing
Applications of High Resolution Mass Spectrometry in Forensic Toxicology. Patrick Kyle, PhD.
 Applications of High Resolution Mass Spectrometry in Forensic Toxicology Patrick Kyle, PhD pkyle@umc.edu Financial Disclosures Grant/Research Support: None Salary/Consultant Fees: None Board/Committee/Advisory
Applications of High Resolution Mass Spectrometry in Forensic Toxicology Patrick Kyle, PhD pkyle@umc.edu Financial Disclosures Grant/Research Support: None Salary/Consultant Fees: None Board/Committee/Advisory
Tests that have had changes to the method/ CPT code, units of measurement, scope of analysis, reference comments, or specimen requirements.
 In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed below are the
In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed below are the
Blood Alcohol Test No Summary Report
 Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Blood Alcohol Summary Report This test was sent to 84 participants. Each sample pack consisted of four vials of blood with differing amounts
Collaborative Testing Services, Inc FORENSIC TESTING PROGRAM Blood Alcohol Summary Report This test was sent to 84 participants. Each sample pack consisted of four vials of blood with differing amounts
Quantitative Analysis of Opiates in Urine Using RRHT LC/MS/MS. Application. Authors. Introduction. Abstract. Forensics
 Quantitative Analysis of piates in Urine Using RRHT LC/MS/MS Application Forensics Authors Sheher Mohsin Agilent Technologies, Inc. 10 N. Martingale Rd., Suite 550 Schaumburg, IL 60173 USA Yanan Yang Agilent
Quantitative Analysis of piates in Urine Using RRHT LC/MS/MS Application Forensics Authors Sheher Mohsin Agilent Technologies, Inc. 10 N. Martingale Rd., Suite 550 Schaumburg, IL 60173 USA Yanan Yang Agilent
Oxymorphone Distribution in Biological Matrices: A Collection of Case Studies
 Oxymorphone Distribution in Biological Matrices: A Collection of Case Studies Brian Simons, Forensic Toxicologist Montgomery County Coroner s Office / Miami Valley Regional Crime Laboratory May 1, 2008
Oxymorphone Distribution in Biological Matrices: A Collection of Case Studies Brian Simons, Forensic Toxicologist Montgomery County Coroner s Office / Miami Valley Regional Crime Laboratory May 1, 2008
September HCMC Toxicology Transition: Additional information and Frequently Asked Questions
 September 2016 HCMC Toxicology Transition: Additional information and Frequently Asked Questions Many clinicians have asked for more information about the Urine Drug Compliance Analysis (LAB8742) switch
September 2016 HCMC Toxicology Transition: Additional information and Frequently Asked Questions Many clinicians have asked for more information about the Urine Drug Compliance Analysis (LAB8742) switch
E XCEL LENCE JUST GOT BETTER UCT FORENSICS CLEAN SCREEN XCEL SPE COLUMNS
 FORENSICS UCT E XCEL LENCE JUST GOT BETTER CLEAN SCREEN XCEL SPE COLUMNS CLEAN SCREEN XCEL COLUMNS EXTRACTION OF BASIC DRUGS AND METABOLITES FROM URINE/ BLOOD USING SPE CARTRIDGES 130mg Clean Screen Xcel
FORENSICS UCT E XCEL LENCE JUST GOT BETTER CLEAN SCREEN XCEL SPE COLUMNS CLEAN SCREEN XCEL COLUMNS EXTRACTION OF BASIC DRUGS AND METABOLITES FROM URINE/ BLOOD USING SPE CARTRIDGES 130mg Clean Screen Xcel
The Drug Testing Process. Employer or Practice
 Disclosures Clinical Professor, Jefferson Medical College BOD MROCC [Medical Review Officer Certification Council] BOD National Sleep Foundation BOD POEMS [Pennsylvania Occupational & Environmental Medicine
Disclosures Clinical Professor, Jefferson Medical College BOD MROCC [Medical Review Officer Certification Council] BOD National Sleep Foundation BOD POEMS [Pennsylvania Occupational & Environmental Medicine
Test Definition: PDSOX Pain Clinic Drug Screen, Chain of Custody, Urine
 Reporting Title: Pain Clinic Drug Screen, CoC, U Performing Location: Rochester Specimen Requirements: Container/Tube: Chain-of-Custody Kit (Supply T282) containing the specimen containers, seals, and
Reporting Title: Pain Clinic Drug Screen, CoC, U Performing Location: Rochester Specimen Requirements: Container/Tube: Chain-of-Custody Kit (Supply T282) containing the specimen containers, seals, and
Schedule of Accreditation
 Schedule of Accreditation Organisation Name Trading As INAB Reg No Contact Name Address Medical Bureau Of Road Safety 30T Helen Kearns Contact Phone No 01-7165555 Email Website Accreditation Standard Health
Schedule of Accreditation Organisation Name Trading As INAB Reg No Contact Name Address Medical Bureau Of Road Safety 30T Helen Kearns Contact Phone No 01-7165555 Email Website Accreditation Standard Health
Urine Drug Testing to Monitor Opioid Use In Managing Chronic Pain
 Faculty Disclosure Henry C. Nipper, PhD, DABCC Dr. Nipper has listed no financial interest/arrangement that would be considered a conflict of interest. Urine Drug Testing to Monitor Opioid Use In Managing
Faculty Disclosure Henry C. Nipper, PhD, DABCC Dr. Nipper has listed no financial interest/arrangement that would be considered a conflict of interest. Urine Drug Testing to Monitor Opioid Use In Managing
A Comprehensive Screening of Illicit and Pain Management Drugs from Whole Blood Using SPE and LC/MS/MS
 A Comprehensive Screening of Illicit and Pain Management Drugs from Whole Blood Using SPE and LC/MS/MS Introduction Drug analysis from whole blood is gaining popularity due to a more complete measurement
A Comprehensive Screening of Illicit and Pain Management Drugs from Whole Blood Using SPE and LC/MS/MS Introduction Drug analysis from whole blood is gaining popularity due to a more complete measurement
Cross-reactivity reactivity in EMIT
 Blood Drug Analysis at the State Bureau of Investigation Crime Laboratory Richard W. Waggoner, Jr. Sample Requirements 2-10 ml gray top vacutainers At least 10 ml of urine sample for drug facilitated sexual
Blood Drug Analysis at the State Bureau of Investigation Crime Laboratory Richard W. Waggoner, Jr. Sample Requirements 2-10 ml gray top vacutainers At least 10 ml of urine sample for drug facilitated sexual
Frequently Asked Questions: Opiate Dependency and Methadone Maintenance Treatment program follow-up
 Frequently Asked Questions: Opiate Dependency and Methadone Maintenance Treatment program follow-up Dr. Bhushan M. Kapur Associate Professor Department of Laboratory Medicine and Pathobiology, Faculty
Frequently Asked Questions: Opiate Dependency and Methadone Maintenance Treatment program follow-up Dr. Bhushan M. Kapur Associate Professor Department of Laboratory Medicine and Pathobiology, Faculty
Rapid LC/TOF MS for Analytical Screening of Drugs in the Clinical Research Lab
 Application Note Clinical Research Rapid LC/TOF MS for Analytical Screening of Drugs in the Clinical Research Lab Authors Adam Barker 1,2, Frederick G. Strathmann 3, Natalie N. Rasmussen 4, and Carrie
Application Note Clinical Research Rapid LC/TOF MS for Analytical Screening of Drugs in the Clinical Research Lab Authors Adam Barker 1,2, Frederick G. Strathmann 3, Natalie N. Rasmussen 4, and Carrie
Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate Mass AxION 2 TOF Mass Spectrometer
 application Note Liquid Chromatography/ Mass Spectrometry Authors Sharanya Reddy Blas Cerda PerkinElmer, Inc. Shelton, CT USA Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate
application Note Liquid Chromatography/ Mass Spectrometry Authors Sharanya Reddy Blas Cerda PerkinElmer, Inc. Shelton, CT USA Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate
Urine drug testing it s not always crystal clear
 Urine drug testing it s not always crystal clear Kirk Moberg, MD, PhD, FASAM Executive Medical Director, UnityPoint Health Illinois Institute for Addiction Recovery Clinical Professor of Internal Medicine
Urine drug testing it s not always crystal clear Kirk Moberg, MD, PhD, FASAM Executive Medical Director, UnityPoint Health Illinois Institute for Addiction Recovery Clinical Professor of Internal Medicine
DrugSmartCup & DrugSmartDip Accuracy Report as produced by Ameditech, Inc.
 Accuracy The accuracy of the DrugSmart Drugs of Abuse Tests was evaluated in comparison to commercially available drug screen tests. Sixty (60) negative urine samples collected from presumed non-user volunteers
Accuracy The accuracy of the DrugSmart Drugs of Abuse Tests was evaluated in comparison to commercially available drug screen tests. Sixty (60) negative urine samples collected from presumed non-user volunteers
3703 Camino del Rio South 100-A San Diego, CA, Phone Fax CLIA# 05D years
 Drug Adherence Assessment Report CleanAssure TM (DRIED BLOOD SPOT): Detection Range see NOTES. Prescribed Medications: NO MEDICATION LIST PROVIDED CONSISTENT RESULTS - MEDICATION DETECTED (PARENT DRUG
Drug Adherence Assessment Report CleanAssure TM (DRIED BLOOD SPOT): Detection Range see NOTES. Prescribed Medications: NO MEDICATION LIST PROVIDED CONSISTENT RESULTS - MEDICATION DETECTED (PARENT DRUG
Urine Drug Testing Methods 3-5
 Urine Drug Testing Methods 3-5 Type of Test Logistics Pearls Initial Screening Test: Immunoassay Confirmatory Test: Gas chromatography-mass spectrometry (GCMS) + or Liquid chromatography-mass spectrometry
Urine Drug Testing Methods 3-5 Type of Test Logistics Pearls Initial Screening Test: Immunoassay Confirmatory Test: Gas chromatography-mass spectrometry (GCMS) + or Liquid chromatography-mass spectrometry
Development of a Screening Analysis by LC Time-Of-Flight MS for Drugs of Abuse Application
 Development of a Screening Analysis by LC Time-Of-Flight MS for Drugs of Abuse Application Forensic Toxicology Authors Courtney Milner and Russell Kinghorn Baseline Separation Technologies Pty Ltd. Abstract
Development of a Screening Analysis by LC Time-Of-Flight MS for Drugs of Abuse Application Forensic Toxicology Authors Courtney Milner and Russell Kinghorn Baseline Separation Technologies Pty Ltd. Abstract
CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva)
 CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva) Cat. No.: DTSJZ023 Pkg. Size: Intended Use The CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva) is an immunochromatography based one step
CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva) Cat. No.: DTSJZ023 Pkg. Size: Intended Use The CDIA TM Marijuana Colloidal Gold Test Cassette (Saliva) is an immunochromatography based one step
DAU. Scheme Description. Drugs of Abuse in Urine Proficiency Testing Scheme
 DAU Drugs of Abuse in Urine Proficiency Testing Scheme Scheme Description LGC Standards Proficiency Testing 1 Chamberhall Business Park Chamberhall Green Bury Lancashire BL9 0AP United Kingdom Telephone:
DAU Drugs of Abuse in Urine Proficiency Testing Scheme Scheme Description LGC Standards Proficiency Testing 1 Chamberhall Business Park Chamberhall Green Bury Lancashire BL9 0AP United Kingdom Telephone:
Workflow for Screening and Quantification of the SAMHSA (NIDA) Panel in Urine Using UHPLC-TOF
 APPLICATIO OTE Liquid Chromatography/ Mass Spectrometry Authors: Avinash Dalmia Joanne Mather PerkinElmer, Inc. Shelton, CT Workflow for Screening and Quantification of the SAMHSA (IDA) Panel in Urine
APPLICATIO OTE Liquid Chromatography/ Mass Spectrometry Authors: Avinash Dalmia Joanne Mather PerkinElmer, Inc. Shelton, CT Workflow for Screening and Quantification of the SAMHSA (IDA) Panel in Urine
Intercept i2he Opiate Oral Fluid Assay
 FOR FORENSIC USE ONLY For Use with Oral Fluid Collection Device 1001-0387 (65 ml Kit) INTENDED USE The is intended for use in the qualitative determination of Opiate in human oral fluid at a cutoff concentration
FOR FORENSIC USE ONLY For Use with Oral Fluid Collection Device 1001-0387 (65 ml Kit) INTENDED USE The is intended for use in the qualitative determination of Opiate in human oral fluid at a cutoff concentration
Procedure for Toxicology Analysis Version 4 Toxicology Unit Effective Date: 05/10/2013. Toxicology Analysis
 Toxicology Analysis 1.0 Purpose - This procedure specifies the required elements for analyzing toxicology submissions and reporting the results of the analysis. 2.0 Scope This procedure applies to all
Toxicology Analysis 1.0 Purpose - This procedure specifies the required elements for analyzing toxicology submissions and reporting the results of the analysis. 2.0 Scope This procedure applies to all
DOF. Scheme Description. Drugs in Oral Fluid Scheme
 DOF Drugs in Oral Fluid Scheme Scheme Description LGC Standards Proficiency Testing 1 Chamberhall Business Park Chamberhall Green Bury Lancashire BL9 0AP United Kingdom Telephone: +44 (0) 161 762 2500
DOF Drugs in Oral Fluid Scheme Scheme Description LGC Standards Proficiency Testing 1 Chamberhall Business Park Chamberhall Green Bury Lancashire BL9 0AP United Kingdom Telephone: +44 (0) 161 762 2500
«Tit» «Name» «Fam_Nam» «Organization» «Address1» «Address_2» «PO_Box» «City». «Country» Sevilla, February, 2016
 «Tit» «Name» «Fam_Nam» «Organization» «Address» «Address_» «PO_Box» «City». «Country» Proficiency Test 5- Laboratory Code: «Lab_code» Sevilla, February, 6 Dear participants, Enclosed you will find the
«Tit» «Name» «Fam_Nam» «Organization» «Address» «Address_» «PO_Box» «City». «Country» Proficiency Test 5- Laboratory Code: «Lab_code» Sevilla, February, 6 Dear participants, Enclosed you will find the
Morphine ELISA Kit. Catalog Number KA assays Version: 05. Intended for research use only.
 Morphine ELISA Kit Catalog Number KA0935 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3 General Information...
Morphine ELISA Kit Catalog Number KA0935 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3 General Information...
Fully Automated Online Sample Preparation and LC-MS/MS Analysis of Drugs of Abuse in Oral Fluids
 PO-CON1753E Fully Automated Online Sample Preparation and LC-MS/MS Analysis of Drugs of Abuse in Oral Fluids ASMS 2017 TP-442 Joshua F. Emory 1, Nathan DeFreitas 2, Michael Roberts 1, Manoj Tyagi 2, M.
PO-CON1753E Fully Automated Online Sample Preparation and LC-MS/MS Analysis of Drugs of Abuse in Oral Fluids ASMS 2017 TP-442 Joshua F. Emory 1, Nathan DeFreitas 2, Michael Roberts 1, Manoj Tyagi 2, M.
T R A I N I N G G U I D E
 TRAINING GUIDE InstaCube Oral Fluid Drug Test For Forensic Use Only Powered By: Contents 3 The information in this presentation is a general overview on performing and interpreting the InstaCube Oral Fluid
TRAINING GUIDE InstaCube Oral Fluid Drug Test For Forensic Use Only Powered By: Contents 3 The information in this presentation is a general overview on performing and interpreting the InstaCube Oral Fluid
Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies
 Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies Frederick G. Strathmann, PhD, DABCC, (CC,TC) Medical Director, Toxicology Associate Scientific Director of MS ARUP Laboratories
Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies Frederick G. Strathmann, PhD, DABCC, (CC,TC) Medical Director, Toxicology Associate Scientific Director of MS ARUP Laboratories
ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS
 ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS Barry K Logan PhD, DABFT National Director of Forensic Services, NMS Labs Willow Grove PA Disclaimer
ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS Barry K Logan PhD, DABFT National Director of Forensic Services, NMS Labs Willow Grove PA Disclaimer
Drug Profiles of Apprehended Drivers in Victoria
 Drug Profiles of Apprehended Drivers in Victoria J Gerostamoulos, P McCaffrey, O H. Drummer and M Odell*. Victorian Institute of Forensic Medicine, Department of Forensic Medicine, Monash University, 57-83
Drug Profiles of Apprehended Drivers in Victoria J Gerostamoulos, P McCaffrey, O H. Drummer and M Odell*. Victorian Institute of Forensic Medicine, Department of Forensic Medicine, Monash University, 57-83
Laboratory Service Report
 4 05/25/19 Client C702884-DLP ROCHESTER Amphetamines, Confirmation Positive Confirmed POSITIVE by LC-S/S for the following: Amphetamine = 52 ethamphetamine = 124 ethamphetamine exists in the d- and l-isomeric
4 05/25/19 Client C702884-DLP ROCHESTER Amphetamines, Confirmation Positive Confirmed POSITIVE by LC-S/S for the following: Amphetamine = 52 ethamphetamine = 124 ethamphetamine exists in the d- and l-isomeric
LC Application Note. Dangerous driver?
 LC Application Note Dangerous driver? www.palsystem.com Dangerous driver? Robert M. Sears 1 ; Kenneth C. Lewis 2 ; and Kim Gamble 3 1 SC Law Enforcement Division, Columbia, SC 29221; 2 OpAns LLC, Durham
LC Application Note Dangerous driver? www.palsystem.com Dangerous driver? Robert M. Sears 1 ; Kenneth C. Lewis 2 ; and Kim Gamble 3 1 SC Law Enforcement Division, Columbia, SC 29221; 2 OpAns LLC, Durham
Opiates, Opioids and Benzodiazepines, Amphetamines & Illicit Drug Forensic Analysis by LC/MS
 Opiates, Opioids and Benzodiazepines, Amphetamines & Illicit Drug Forensic Analysis by LC/MS Julie Cichelli, PhD Agilent Technologies Application Engineer April 29, 2014 Agenda A method for the rapid analysis
Opiates, Opioids and Benzodiazepines, Amphetamines & Illicit Drug Forensic Analysis by LC/MS Julie Cichelli, PhD Agilent Technologies Application Engineer April 29, 2014 Agenda A method for the rapid analysis
Procedure for Toxicology Analysis Version 7 Toxicology Unit Effective Date: 03/14/2014 Issued by Drug Chemistry Forensic Scientist Manager
 Toxicology Analysis 1.0 Purpose - This procedure specifies the required elements for analyzing toxicology submissions and reporting drug testing results. 2.0 Scope This procedure applies to all submissions
Toxicology Analysis 1.0 Purpose - This procedure specifies the required elements for analyzing toxicology submissions and reporting drug testing results. 2.0 Scope This procedure applies to all submissions
Modernizing the Forensic Lab with LC-MS/MS Technology
 Modernizing the Lab with LC-MS/MS Technology Innovative SCIEX Analytical Tools for the Rapid Identification of Drugs of Abuse in Samples Oscar G. Cabrices 1, Holly McCall 1, Xiang He 1, Alexandre Wang
Modernizing the Lab with LC-MS/MS Technology Innovative SCIEX Analytical Tools for the Rapid Identification of Drugs of Abuse in Samples Oscar G. Cabrices 1, Holly McCall 1, Xiang He 1, Alexandre Wang
Testing for Controlled Substances
 Testing for illicit drugs Testing for Controlled Substances 1 Purposes: Employment Sports Screening medical eval. Legal Monitoring Treatment Probation Prescribing controlled substances Forensics 2 Drug
Testing for illicit drugs Testing for Controlled Substances 1 Purposes: Employment Sports Screening medical eval. Legal Monitoring Treatment Probation Prescribing controlled substances Forensics 2 Drug
Driving Under The Influence Of Drugs In Ireland: A Growing And Significant Danger
 Driving Under The Influence Of Drugs In Ireland: A Growing And Significant Danger D.A.Cusack, G.Harrington, P.Furney, K.Flynn and C.P.Leavy Medical Bureau of Road Safety, Department of Forensic Medicine,
Driving Under The Influence Of Drugs In Ireland: A Growing And Significant Danger D.A.Cusack, G.Harrington, P.Furney, K.Flynn and C.P.Leavy Medical Bureau of Road Safety, Department of Forensic Medicine,
EDUCATIONAL COMMENTARY rd TEST EVENT Chemistry Urine Drug Testing
 EDUCATIONAL COMMENTARY 2003 3 rd TEST EVENT Chemistry Urine Drug Testing Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE
EDUCATIONAL COMMENTARY 2003 3 rd TEST EVENT Chemistry Urine Drug Testing Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE
INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE, MARYLAND QUARTERLY REPORT: FOURTH QUARTER, 2008 AND 2008 SUMMARY
 INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE, MARYLAND QUARTERLY REPORT: FOURTH QUARTER, 2008 AND 2008 SUMMARY A report from the Office of Epidemiology and Planning Baltimore
INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE, MARYLAND QUARTERLY REPORT: FOURTH QUARTER, 2008 AND 2008 SUMMARY A report from the Office of Epidemiology and Planning Baltimore
Determination of Opiates and Metabolites in Blood Using Electrospray LC/MS. Application Note
 Deteration of piates and Metabolites in Blood Using Electrospray LC/MS Application Note Scott A. Schlueter and James D. Hutchison, Jr. Montana Department of Justice, Division of Forensic Science John M.
Deteration of piates and Metabolites in Blood Using Electrospray LC/MS Application Note Scott A. Schlueter and James D. Hutchison, Jr. Montana Department of Justice, Division of Forensic Science John M.
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 Schedule of ccreditation United Kingdom ccreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK ccredited to Laboratory locations: Gavenny Court Brecon Road bergavenny Monmouthshire
Schedule of ccreditation United Kingdom ccreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK ccredited to Laboratory locations: Gavenny Court Brecon Road bergavenny Monmouthshire
Validation of an Automated Method to Remove
 Shahana Wahab Huq 1, Richard Thomas 2, Agnes Cua 2, Seyed Sadjadi 1 1 Phenomenex, 411 Madrid Avenue, Torrance, CA 951 2 Precision Toxicology, 33 Bunker Hill Street, San Diego, CA 9219 Validation of an
Shahana Wahab Huq 1, Richard Thomas 2, Agnes Cua 2, Seyed Sadjadi 1 1 Phenomenex, 411 Madrid Avenue, Torrance, CA 951 2 Precision Toxicology, 33 Bunker Hill Street, San Diego, CA 9219 Validation of an
Drug Adherence Assessment Report
 Prescribed Medications: Drug Adherence Assessment Report FENTANYL, OXYCODONE CONSISTENT RESULTS - REPORTED MEDICATION DETECTED (PARENT DRUG AND/OR METABOLITE) REPORTED ANTICIPATED TEST PRESCRIPTION (S)
Prescribed Medications: Drug Adherence Assessment Report FENTANYL, OXYCODONE CONSISTENT RESULTS - REPORTED MEDICATION DETECTED (PARENT DRUG AND/OR METABOLITE) REPORTED ANTICIPATED TEST PRESCRIPTION (S)
OraSure Technologies, Inc. 150 Webster Street, Bethlehem, PA Phone: (USA) Fax:
 OraSure Technologies, Inc. 150 Webster Street, Bethlehem, PA 18015-1389 Phone: 610-882-1820 1-800-869-3538 (USA) Fax: 610-882-1830 www.orasure.com OPIATES INTERCEPT MICRO-PLATE EIA for use with Intercept
OraSure Technologies, Inc. 150 Webster Street, Bethlehem, PA 18015-1389 Phone: 610-882-1820 1-800-869-3538 (USA) Fax: 610-882-1830 www.orasure.com OPIATES INTERCEPT MICRO-PLATE EIA for use with Intercept
Test Definition: PCDSO Pain Clinic Drug Screen, Urine
 Reporting Title: Pain Clinic Drug Screen, U Performing Location: Rochester Specimen Requirements: Collection Container/Tube: Plastic urine container Submission Container/Tube: Plastic, 60-mL urine bottle
Reporting Title: Pain Clinic Drug Screen, U Performing Location: Rochester Specimen Requirements: Collection Container/Tube: Plastic urine container Submission Container/Tube: Plastic, 60-mL urine bottle
3703 Camino del Rio South 100-A San Diego, CA, Phone Fax CLIA# 05D Director: David J.
 Drug Adherence Assessment Report Prescribed Medications: NO MEDICATION LIST PROVIDED CONSISTENT RESULTS - REPORTED MEDICATION DETECTED (PARENT DRUG AND/OR METABOLITE) REPORTED PRESCRIPTION FLAG ANTICIPATED
Drug Adherence Assessment Report Prescribed Medications: NO MEDICATION LIST PROVIDED CONSISTENT RESULTS - REPORTED MEDICATION DETECTED (PARENT DRUG AND/OR METABOLITE) REPORTED PRESCRIPTION FLAG ANTICIPATED
LCMS-8050 Drugs of Abuse: 113 Analytes with Polarity Switching
 Liquid Chromatography Mass Spectrometry SSI-LCMS-8 LCMS-8 Drugs of Abuse: Analytes with Polarity Switching LCMS-8 Summary Seventy six analytes and their internal standards are described below. Multiple
Liquid Chromatography Mass Spectrometry SSI-LCMS-8 LCMS-8 Drugs of Abuse: Analytes with Polarity Switching LCMS-8 Summary Seventy six analytes and their internal standards are described below. Multiple
2013 Clinical drug and alcohol testing solutions. Product Catalog. CLIA-waived point of care test devices
 2013 Clinical drug and alcohol testing solutions Product Catalog CLIA-waived point of care test devices HELPING YOU MAKE INFORMED DECISIONS ABOUT ABUSE. Substance abuse testing with more substance. 2 of
2013 Clinical drug and alcohol testing solutions Product Catalog CLIA-waived point of care test devices HELPING YOU MAKE INFORMED DECISIONS ABOUT ABUSE. Substance abuse testing with more substance. 2 of
Workflow for Screening and Quantification of the SAMHSA (NIDA) Panel in Serum Using UHPLC-TOF
 APPLICATIO OTE Liquid Chromatography/ Mass Spectrometry Authors: Avinash Dalmia Bonnie Marmor PerkinElmer, Inc. Shelton, CT Workflow for Screening and Quantification of the SAMHSA (IDA) Panel in Serum
APPLICATIO OTE Liquid Chromatography/ Mass Spectrometry Authors: Avinash Dalmia Bonnie Marmor PerkinElmer, Inc. Shelton, CT Workflow for Screening and Quantification of the SAMHSA (IDA) Panel in Serum
INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE CITY
 2009 FINAL REPORT INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE CITY This report was prepared by: Dr. Jose Arbelaez, M.D. of Baltimore Substance Abuse Systems, and Ryan J. Petteway,
2009 FINAL REPORT INTOXICATION DEATHS ASSOCIATED WITH DRUGS OF ABUSE OR ALCOHOL BALTIMORE CITY This report was prepared by: Dr. Jose Arbelaez, M.D. of Baltimore Substance Abuse Systems, and Ryan J. Petteway,
European Guidelines for Workplace Drug Testing in UrineandOral fluid
 EWDTS European Workplace Drug Testing Society European Guidelines for Workplace Drug Testing in UrineandOral fluid 9th EWDTS Symposium in Lisbon Sanna Taskinen Michaela Neuhofer Updating project # 2 Current
EWDTS European Workplace Drug Testing Society European Guidelines for Workplace Drug Testing in UrineandOral fluid 9th EWDTS Symposium in Lisbon Sanna Taskinen Michaela Neuhofer Updating project # 2 Current
1/27/ New Release, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
Using Liquid Chromatography Tandem Mass Spectrometry Urine Drug Testing to Identify Licit and Illicit Drug-Use in a Community-based Patient Population
 Using Liquid Chromatography Tandem Mass Spectrometry Urine Drug Testing to Identify Licit and Illicit Drug-Use in a Community-based Patient Population Adam S. Ptolemy 1, Colleen Murray 2, Edward Dunn 3,
Using Liquid Chromatography Tandem Mass Spectrometry Urine Drug Testing to Identify Licit and Illicit Drug-Use in a Community-based Patient Population Adam S. Ptolemy 1, Colleen Murray 2, Edward Dunn 3,
Oral Fluid Drugs of Abuse Testing
 Solutions at Work Oral Fluid Drugs of Abuse Testing Volume indicator turns blue when collection is complete (average 3 minutes) Non-invasive collection test anytime, anywhere Directly observed, no gender
Solutions at Work Oral Fluid Drugs of Abuse Testing Volume indicator turns blue when collection is complete (average 3 minutes) Non-invasive collection test anytime, anywhere Directly observed, no gender
A FORENSIC TOXICOLOGY METHOD FOR THE DETERMINATION OF DESOMORPHINE, HEROIN, METHADONE, BUPRENORPHINE AND METABOLITES IN URINE USING LC/MS QQQ
 FORENSICS MARKET A FORENSIC TOXICOLOGY METHOD FOR THE DETERMINATION OF DESOMORPHINE, HEROIN, METHADONE, BUPRENORPHINE AND METABOLITES IN URINE USING LC/MS QQQ Desomorphine, also known by its street name
FORENSICS MARKET A FORENSIC TOXICOLOGY METHOD FOR THE DETERMINATION OF DESOMORPHINE, HEROIN, METHADONE, BUPRENORPHINE AND METABOLITES IN URINE USING LC/MS QQQ Desomorphine, also known by its street name
Drugs of Abuse I Serum
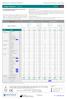 Drugs of Abuse I Serum 2017-11 1.10 For quality monitoring of quantitative measurements by chromatographic methods These reference materials are produced on the basis of human matrices. The highly accurate
Drugs of Abuse I Serum 2017-11 1.10 For quality monitoring of quantitative measurements by chromatographic methods These reference materials are produced on the basis of human matrices. The highly accurate
METHAMPHETAMINE & AMPHETAMINE
 METHAMPHETAMINE & AMPHETAMINE Enantiomers from Urine Rapid and Accurate Chiral Separation Polysaccharide Chiral Chiral LC LC Columns and SFC PHEN-RUO-49 Revision: Lux 3 µm AMP Column High Efficiency LC/MS/MS
METHAMPHETAMINE & AMPHETAMINE Enantiomers from Urine Rapid and Accurate Chiral Separation Polysaccharide Chiral Chiral LC LC Columns and SFC PHEN-RUO-49 Revision: Lux 3 µm AMP Column High Efficiency LC/MS/MS
Welcome! Supreme Court of Ohio Specialized Dockets Conference. October 23-24, 2017
 Welcome! Supreme Court of Ohio Specialized Dockets Conference October 23-24, 2017 Drug Testing: Do you know enough to be dangerous? Presented by William L. Parker President & CEO American Court & Drug
Welcome! Supreme Court of Ohio Specialized Dockets Conference October 23-24, 2017 Drug Testing: Do you know enough to be dangerous? Presented by William L. Parker President & CEO American Court & Drug
Fastect II Drug Screen Dipstick Test Training and Certification Program
 Fastect II Drug Screen Dipstick Test Training and Certification Program Fastect II Training Page 1 of 7 Rev. B Fastect II Drug Screen Dipstick Test Training and Certification for Test Administrators The
Fastect II Drug Screen Dipstick Test Training and Certification Program Fastect II Training Page 1 of 7 Rev. B Fastect II Drug Screen Dipstick Test Training and Certification for Test Administrators The
Analysis of drugs of abuse in biological matrix using Time of Flight technology
 Analysis of drugs of abuse in biological matrix using Time of Flight technology Niclas Stephanson, PhD Department of Clinical Pharmacology Karolinska University Hospital Karolinska Institutet Stockholm,
Analysis of drugs of abuse in biological matrix using Time of Flight technology Niclas Stephanson, PhD Department of Clinical Pharmacology Karolinska University Hospital Karolinska Institutet Stockholm,
Patient-Centered Urine Drug Testing. Douglas Gourlay, MD, MSc, FRCPC, FASAM
 Patient-Centered Urine Drug Testing Douglas Gourlay, MD, MSc, FRCPC, FASAM Declaration of Potential Conflict of Interest The content of this presentation is non- commercial and does not represent any conflict
Patient-Centered Urine Drug Testing Douglas Gourlay, MD, MSc, FRCPC, FASAM Declaration of Potential Conflict of Interest The content of this presentation is non- commercial and does not represent any conflict
DRUG-CONJUGATES The drug buprenorphine is conjugated to protein and immobilized as a line on a membrane at the BUP location labeled on the device.
 MEDTOX BUPRENORPHINE MEDTOX Buprenorphine is a rapid qualitative screening test for buprenorphine and its metabolites in human urine. 1. INTENDED USE The MEDTOX Buprenorphine Test uses immunochromatographic
MEDTOX BUPRENORPHINE MEDTOX Buprenorphine is a rapid qualitative screening test for buprenorphine and its metabolites in human urine. 1. INTENDED USE The MEDTOX Buprenorphine Test uses immunochromatographic
Clearing Up The Confusion About Substance Abuse Testing
 Clearing Up The Confusion About Substance Abuse Testing 2018 Indiana Safety and Health Conference and Expo Presented by Tiffany Ellefson, DISA/Midwest Toxicology Services Items to cover Latest Trends in
Clearing Up The Confusion About Substance Abuse Testing 2018 Indiana Safety and Health Conference and Expo Presented by Tiffany Ellefson, DISA/Midwest Toxicology Services Items to cover Latest Trends in
Drug Screening: Things You Need to Know
 Drug Screening: Things You Need to Know (a view inside the clinical laboratory) Gary L. Horowitz, MD Director, Clinical Chemistry, Beth Israel Deaconess Medical Center Associate Professor of Pathology,
Drug Screening: Things You Need to Know (a view inside the clinical laboratory) Gary L. Horowitz, MD Director, Clinical Chemistry, Beth Israel Deaconess Medical Center Associate Professor of Pathology,
DRUGS OF ABUSE
 DRUGS OF ABUSE www.btnx.com 1-888-339-9964 Rapid Response Drugs of Abuse and Alcohol Screening Devices BTNX Inc. prides itself in offering quality drug detection tests for accurate, safe and rapid testing.
DRUGS OF ABUSE www.btnx.com 1-888-339-9964 Rapid Response Drugs of Abuse and Alcohol Screening Devices BTNX Inc. prides itself in offering quality drug detection tests for accurate, safe and rapid testing.
Rapid Alcohol Screening Devices. p19
 CLIA-WAIVED Product Catalog RAPID DRUG SCREENING DEVICES Rapid Urine Drug Screening Devices. p5 Rapid Alcohol Screening Devices. p19 Complementary Products. p23 HELPING YOU MAKE INFORMED DECISIONS ABOUT
CLIA-WAIVED Product Catalog RAPID DRUG SCREENING DEVICES Rapid Urine Drug Screening Devices. p5 Rapid Alcohol Screening Devices. p19 Complementary Products. p23 HELPING YOU MAKE INFORMED DECISIONS ABOUT
LABORATORY TECHNICAL SPECIFICATIONS MANUAL
 LABORATORY TECHNICAL SPECIFICATIONS MANUAL Produced by Synergy Health Laboratory Services Controlled Document ID 103.00250 January 2015 - Version 9 Purpose of Procedure This document outlines the technical
LABORATORY TECHNICAL SPECIFICATIONS MANUAL Produced by Synergy Health Laboratory Services Controlled Document ID 103.00250 January 2015 - Version 9 Purpose of Procedure This document outlines the technical
The Development of LC/MS Methods for Determination of MDMA (Ecstasy) and Metabolites in Biological Samples
 WA20714 The Development of LC/MS Methods for Determination of MDMA (Ecstasy) and Metabolites in Biological Samples Michael S. Young and Kevin M. Jenkins Waters Corporation, 34 Maple Street, Milford, MA
WA20714 The Development of LC/MS Methods for Determination of MDMA (Ecstasy) and Metabolites in Biological Samples Michael S. Young and Kevin M. Jenkins Waters Corporation, 34 Maple Street, Milford, MA
LABORATORY TECHNICAL SPECIFICATIONS MANUAL
 LABORATORY TECHNICAL SPECIFICATIONS MANUAL Produced by Synergy Health Laboratory Services Controlled Document ID 103.00250 August 2015 - Version 12 Purpose of Procedure This document outlines the technical
LABORATORY TECHNICAL SPECIFICATIONS MANUAL Produced by Synergy Health Laboratory Services Controlled Document ID 103.00250 August 2015 - Version 12 Purpose of Procedure This document outlines the technical
Confirmation by Triple Quadrupole LC/MS/MS for HHS-compliant Workplace Urine Drug Testing
 Confirmation by Triple Quadrupole LC/MS/MS for HHS-compliant Workplace Urine Drug Testing A complete applications solution for forensic toxicology laboratories John Hughes Agilent Technologies Pleasanton,
Confirmation by Triple Quadrupole LC/MS/MS for HHS-compliant Workplace Urine Drug Testing A complete applications solution for forensic toxicology laboratories John Hughes Agilent Technologies Pleasanton,
Effective Date: Approved by: Laboratory Executive Director, Ed Hughes (electronic signature)
 1 Policy #: 803 (PLH-803-02) Effective Date: NA Reviewed Date: 4/11/2008 Subject: URINE DRUG SCREENS Approved by: Laboratory Executive Director, Ed Hughes (electronic signature) Approved by: Laboratory
1 Policy #: 803 (PLH-803-02) Effective Date: NA Reviewed Date: 4/11/2008 Subject: URINE DRUG SCREENS Approved by: Laboratory Executive Director, Ed Hughes (electronic signature) Approved by: Laboratory
CLIA Regulations and Toxicology
 Wednesday April 5, 2017 8:45am Regulations and Toxicology Sandy Pearson, MT(ASCP), MBA Laboratory Consultant, CMS Dallas Regional Office Survey Branch DESCRIPTION: Sandy Pearson will begin the workshop
Wednesday April 5, 2017 8:45am Regulations and Toxicology Sandy Pearson, MT(ASCP), MBA Laboratory Consultant, CMS Dallas Regional Office Survey Branch DESCRIPTION: Sandy Pearson will begin the workshop
Quantitative Analysis of Amphetamine-Type Drugs by Extractive Benzoylation and LC/MS/MS. Application. Introduction. Authors. Abstract.
 Quantitative Analysis of Amphetamine-Type Drugs by Extractive Benzoylation and LC/MS/MS Application Forensics Authors Neil Campbell, B. Sc. Forensic Science Laboratory Chemistry Centre (WA) 5 Hay Street
Quantitative Analysis of Amphetamine-Type Drugs by Extractive Benzoylation and LC/MS/MS Application Forensics Authors Neil Campbell, B. Sc. Forensic Science Laboratory Chemistry Centre (WA) 5 Hay Street
3/8/2018. Reasons for Doing UDT. UDT: A Tool in Risk Assessment. Faculty/Presenter Disclosure. Urine Drug Testing in Chronic Pain Management
 Urine Drug Testing in Chronic Pain Management March 8, 2018 Faculty/Presenter Disclosure Faculty: Andrew J Smith, MDCM Relationships with commercial interests: None to report Andrew J Smith, MDCM Staff
Urine Drug Testing in Chronic Pain Management March 8, 2018 Faculty/Presenter Disclosure Faculty: Andrew J Smith, MDCM Relationships with commercial interests: None to report Andrew J Smith, MDCM Staff
Proof of Concept for Automated SPE/HPLC/MS/MS Methods to Replace Traditional Immunoassay with MS Confirmation of Driving Under the Influence Samples
 Proof of Concept for Automated SPE/HPLC/MS/MS Methods to Replace Traditional Immunoassay with MS Confirmation of Driving Under the Influence Samples Robert M. Sears, Toxicology Technical Leader 1, Kenneth
Proof of Concept for Automated SPE/HPLC/MS/MS Methods to Replace Traditional Immunoassay with MS Confirmation of Driving Under the Influence Samples Robert M. Sears, Toxicology Technical Leader 1, Kenneth
Analysis of Benzodiazepines in Alternate Matrices by HPLC. Overview
 Analysis of Benzodiazepines in Alternate Matrices by HPLC. Overview Benzodiazepines/Class/Structure Benzodiazepine Metabolism/Elimination Biological Specimens Instrumentation Analytical Method Controls
Analysis of Benzodiazepines in Alternate Matrices by HPLC. Overview Benzodiazepines/Class/Structure Benzodiazepine Metabolism/Elimination Biological Specimens Instrumentation Analytical Method Controls
One Source Toxicology Laboratory, 1213 Genoa Red Bluff, Pasadena, Texas 77504
 Validation of Analysis of Amphetamines, Opiates, Phencyclidine, Cocaine, and Benzoylecgonine in Oral Fluids by Liquid Chromatography Tandem Mass Spectrometry Subbarao V. Kala*, Steve E. Harris, Tom D.
Validation of Analysis of Amphetamines, Opiates, Phencyclidine, Cocaine, and Benzoylecgonine in Oral Fluids by Liquid Chromatography Tandem Mass Spectrometry Subbarao V. Kala*, Steve E. Harris, Tom D.
A Novel Platform of On-line Sample Pre-treatment and LC/MS/MS Analysis for Screening and Quantitation of Illicit Drugs in Urine
 PO-CON737E A Novel Platform of On-line Sample Pre-treatment and LC/MS/MS Analysis for Screening and Quantitation of Illicit Drugs in Urine ASMS 7 WP 353 Shao Hua Chia ; Zhi Wei Edwin Ting ; Daisuke Kawakami
PO-CON737E A Novel Platform of On-line Sample Pre-treatment and LC/MS/MS Analysis for Screening and Quantitation of Illicit Drugs in Urine ASMS 7 WP 353 Shao Hua Chia ; Zhi Wei Edwin Ting ; Daisuke Kawakami
The Utility of Urine Drug Screening
 The Utility of Urine Drug Screening Treating Addiction, Saving Lives Sea Cruises Bye Tazmania, still far from New Zealand February 8 th, 2018 Mandy Manak, MD FASAM, ISAM, CSAM, MRO Medical Director, ICDO
The Utility of Urine Drug Screening Treating Addiction, Saving Lives Sea Cruises Bye Tazmania, still far from New Zealand February 8 th, 2018 Mandy Manak, MD FASAM, ISAM, CSAM, MRO Medical Director, ICDO
QuickTox Drug Screen Dipcard (with and without Adulteration Tests)
 QuickTox Drug Screen Dipcard (with and without Adulteration Tests) Training and Certification Program Presented by Branan Medical Corporation Branan Medical Corporation 10015 Muirlands Road Irvine, California
QuickTox Drug Screen Dipcard (with and without Adulteration Tests) Training and Certification Program Presented by Branan Medical Corporation Branan Medical Corporation 10015 Muirlands Road Irvine, California
Intercept i2he Methamphetamine Oral Fluid Assay
 Intercept i2he Methamphetamine FOR FORENSIC USE ONLY For Use with Intercept i2he Oral Fluid Collection Device 1001-0386 (65 ml Kit) INTENDED USE The Intercept i2he Methamphetamine is intended for use in
Intercept i2he Methamphetamine FOR FORENSIC USE ONLY For Use with Intercept i2he Oral Fluid Collection Device 1001-0386 (65 ml Kit) INTENDED USE The Intercept i2he Methamphetamine is intended for use in
Urine Opioid Dependency Panel (UODP) 1
 Drug Testing Panels Urine Opioid Dependency Panel (UODP) 1 Amphetamines Amphetamine Methamphetamine Methylenedioxyamphetamine Adderall, Dexedrine, Vyvanse, Lisdexamfetamine (Speed, Bennies, Crystal Meth,
Drug Testing Panels Urine Opioid Dependency Panel (UODP) 1 Amphetamines Amphetamine Methamphetamine Methylenedioxyamphetamine Adderall, Dexedrine, Vyvanse, Lisdexamfetamine (Speed, Bennies, Crystal Meth,
LCMS APPLICATIONS TCFS LAB. Applications of Triple Quadrupole LC/MS/MS in a Forensic Toxicology Laboratory CURRENT LCMS ANALYTES
 Applications of Triple Quadrupole LC/MS/MS in a Forensic Toxicology Laboratory Jinee D. Rizzo, Ph.D. Travis County Forensic Center Austin, Texas jinee.rizzo@co.travis.tx.us LCMS APPLICATIONS TCFS LAB Simplify
Applications of Triple Quadrupole LC/MS/MS in a Forensic Toxicology Laboratory Jinee D. Rizzo, Ph.D. Travis County Forensic Center Austin, Texas jinee.rizzo@co.travis.tx.us LCMS APPLICATIONS TCFS LAB Simplify
