Theron J. Hamilton 1 *, Harry Z. Qui 1, Katherine V. R. Dozier 1 and Zachary J. Fuller 2. Testing Laboratory, Fort Meade, MD, USA
|
|
|
- Sybil Park
- 5 years ago
- Views:
Transcription
1 Journal of Analytical Toxicology 2014;38: doi: /jat/bku069 Advance Access publication June 20, 2014 Technical Note Phentermine Interference and High L-Methamphetamine Concentration Problems in GC EI-MS SIM Analyses of R-(2)-a-Methoxy-a-(Trifluoromethyl)Phenylacetyl Chloride-Derivatized Amphetamines and Methamphetamines Theron J. Hamilton 1 *, Harry Z. Qui 1, Katherine V. R. Dozier 1 and Zachary J. Fuller 2 1 Department of Chemistry, United States Naval Academy, Annapolis, MD, USA, and 2 United States Army Forensic Toxicology Drug Testing Laboratory, Fort Meade, MD, USA *Author to whom correspondence should be addressed. theron@usna.edu In order to achieve chromatographic separation, urine samples shown to be initially positive for amphetamines and methamphetamines in US Department of Defense immunoassays are derivatized with R-(2)-a-methoxy-a-(trifluoromethyl)phenylacetyl chloride (R-(2)-MTPA) prior to gas chromatography electron impact-mass spectrometry (GC EI-MS) analysis. Phentermine, a member of the phenethylamine class of drugs and a common appetite suppressant, interferes with GC EI-MS assays of R-(2)-MTPA-derivatized D-amphetamine, degrading the chromatography of the internal standard and analyte ions and skewing concentration calculations. Additionally, when specimens with high concentrations of L-methamphetamine are derivatized with R-(2)-MTPA, signal peaks have the potential to be misidentified by integration software as D-methamphetamine. We have found that replacing R-(2) MTPA with (S)- (1)-a-methoxy-a-(trifluoromethyl)phenylacetyl chloride reduces phentermine interference problems related to internal standard chromatography, reduces the possibility of concentrated L-methamphetamine peaks being misidentified by integration software, improves resolution of D-methamphetamine in the presence of high L-methamphetamine concentrations, and is a cost-neutral change that can be applied to current amphetamines GC EI-MS methods without the need for method modification. Introduction Amphetamine and methamphetamine are routinely included in Department of Defense (DoD) drug screening panels. In the DoD regimen, urine specimens are screened with three amphetamine-sensitive commercial immunological assays before confirmation of drug presence is ultimately made through gas chromatography electron impact-mass spectrometry (GC EI-MS) analysis using selected ion monitoring (SIM). Since 2004, the standard confirmational GC EI-MS SIM and sample preparation methods for amphetamine and methamphetamine used by the DoD have closely followed the recommendations published by Paul et al. (1), which suggest the use of R-(2)- a-methoxy-a-(trifluoromethyl)phenylacetyl chloride (R-(2)- MTPA) as a chiral derivatizing agent for distinguishing enantiomeric pairs of amphetamines and methamphetamines. The use of MTPA allows for the complete chromatographic separation of the S-(þ) or D andther-(2) or L molecular stereoisomers The views expressed in this article are those of the author and do not necessarily reflect the official policy or position of the Department of the Navy, Department of the Army and Department of Defense nor the US Government. of amphetamine and methamphetamine. However, the US Army Forensic Toxicology Drug Testing Laboratory (FTDTL), at Fort Meade, MD, USA, has recently noticed several issues related to the interference of GC EI-MS assays by phentermine (C 10 H 15 N) an FDA approved appetite suppressant for the treatment of obesity and a member of the phenethylamine class of drugs. Phentermine is not eliminated by specimen pretreatment with iodate in a way similar to phenylpropanolamine and ephedrine. Attempts to modify the current DoD GC EI-MS method in order to achieve chromatographic separation between the interferent and analyte ions were unproductive; moreover, a prevalence study showed that thousands of tested military specimens per year could be contaminated with phentermine at the Ft. Meade FTDTL alone. This interference issue translates into a relatively significant operational and economic burden that is likely to also affect other drug testing laboratories. In particular, we have observed that the effect of phentermine interference on the established DoD GC EI-MS amphetamine method (no phentermine interference was observed with methamphetamines) can be divided into two cases. Case 1: GC EI-MS analyses showing the complete absence of amphetamines in samples that initially screened positive in three separate amphetamines-sensitive immunological assays. The problem in this case is troublesome for two reasons. First, it highlights a limitation of this application of the immunoassay screening reactions which cross-react with phentermine a well-known problem that has proven difficult to circumvent (2, 3). Secondly, although there is an absence of target analyte, phentermine complicates GC EI-MS analysis by interfering with the internal standard quantification and qualifier ions. The effect is a shift of the internal standard ion peaks to retention times outside of the defined retention time integration windows and possibly unacceptable chromatography. In response, standard operating procedure calls for manual integration of the internal standard ion peaks and subsequent re-extractions of the original specimen. According to the US Army FTDTL drug testing protocol, even negative results with internal standard failures will not be released without the lab first attempting to obtain acceptable internal standard results through follow-up assays. In other DoD drug testing laboratories, acceptable internal standard results with analyte-negative samples are defined as those exhibiting retention times and mass ion ratios that, minimally, allow for the identification of the relevant internal standard ions. However, even with this less strict criterion, rework is still likely since most phentermine-contaminated samples will have skewed retention times. For drug testing laboratories experiencing a high prevalence of phentermine-contaminated specimens, the Published by Oxford University Press This work is written by (a) US Government employee(s) and is in the public domain in the US.
2 economic and man-hour costs of additional extractions, new GC EI-MS confirmation assays and multiple data reviews as a result of internal standard failures can become significant. Case 2: GC EI-MS analyses showing the presence of D- and L-amphetamine in the sample, but failing acceptability criteria because of poor chromatographic resolution of the 162 m/z and 118 m/z analyte qualifier ions due to interference from co-eluting phentermine fragments. Instances of this problem in practice are less impactful than those in Case 1 since the occurrence frequency is low, but nonetheless, we noticed some interesting consequences. For example, the effect of phentermine fragments co-eluting with the internal standard target and qualifier ions is quantifiable. Consider the following experiment with control samples. In general, final analyte concentrations are given by C ¼ C 0 ða dt=a dis Þ ða ct =A cis Þ where C 0 is the concentration cutoff, A dt is the drug target ion area, A dis is the drug internal standard area, A ct is the calibrator target ion area and A cis is the calibrator internal standard ion area. Regardless of whether or not the qualifier ions resolve, in these control experiments, the internal standard quantification (264 m/z) and the drug quantification (262 m/z) ion peaks are consistently within defined retention time integration windows with acceptable chromatographic resolution. Our data show that, when compared with the known control concentration, experimental samples containing phentermine will have calculated analyte concentrations up to 15% below the known value, implying that latent co-eluting phentermine fragments are artificially increasing internal standard peak area values (data not shown). This result is consistent with the equation above, which predicts that larger internal standard areas will concurrently reduce final analyte concentrations, C. Considering this fact, we can envision at least some scenarios where the potential to release artificially low quantification results exists when phentermine concentrations are not high enough to degrade chromatography enough to fail acceptability standards. A more serious problem unrelated to phentermine interference is revealed when analyzing urine with high L- methamphetamine concentrations. It is possible that GC EI-MS assays of specimens overloaded with L-methamphetamine isomers will show L-methamphetamine peak migration to later retention times. Achieving urinary L-methamphetamine concentrations, high enough to induce this effect is possible through the use of over-the-counter products. For example, published studies have reported urinary concentrations of L-methamphetamine as high as 6,000 ng/ml following the use of a Vicks w inhaler (4). A chromatographically well resolved, but retention time-shifted L-methamphetamine peak could cause integration software to misidentify it as D-methamphetamine when derivatized with R-(2)-MTPA. If also coupled with proper mass ion ratios and chromatographically acceptable internal standard ion peaks, recognizing the error could be particularly difficult for data review officials. This fact and the potential for the artificially lowconcentration values discussed above are particularly disconcerting in an environment where accurate quantitation is critical to both the success of the DoD s drug-use deterrence efforts and to those individuals whose careers hinge on the fairness and accuracy of the results. Here, we report a simple and cost-neutral improvement to reduce the problems discussed above based on the use of a derivatizing agent with chirality opposite to the one currently used in DoD amphetamine protocols, S-(þ)-a-methoxy-a-trifluoromethylphenylacetyl (S-(þ)-MTPA). Substitution of R-(2)-MTPA with S-(þ)-MTPA reduces phentermine interference by changing the elution order of the D- andl-enantiomers. This change has the effect of shifting target D-amphetamine ion away from the retention times of the phentermine fragments, removing the internal standard interference issues and the potential for calculating artificially low-concentration values in the process. In this application, the use of S-(þ)-MTPA could significantly reduce the amount of rework triggered by phentermine-related GC EI-MS amphetamine confirmation assay failures in DoD and other drug testing laboratories. Most importantly, the possibility of erroneous peak integration with R-(2)-MTPAderivatized specimens containing high concentrations of L-methamphetamine isomers is avoided as a result of shifted retention times. Finally, it should be noted that the substitution of R-(2)-MTPA for S-(þ)-MTPA can be made without any modification to the current DoD amphetamine GC EI-MS analysis method. Experimental The extraction and derivatization procedures as well as the GC EI-MS SIM parameters used in all experiments were identical to those described in Paul et al., with the following exceptions: (i) the sample preparation procedures employed in the study of phentermine prevalence did not include a derivatization step and (ii) a calibrator cutoff concentration of 100 ng/ml for DoD amphetamines assays was used to quantitate isomer concentrations rather than a 500 ng/ml calibrator in all cases. S-(þ)-MTPA was purchased from Sigma Aldrich Corporation (St. Louis, MO, USA). Service member urine specimens analyzed in the effort to estimate the prevalence of phentermine had been held for further investigation after screening positive in amphetamine immunoassays, but confirming negative with GC EI-MS. In order to verify the presence of phentermine, aliquots of the original specimens were extracted, purified without derivatization and subjected to a total (raw signal) mass scan on an Agilent w 6890 GC EI-MS. Mass spectra were extracted from all recorded chromatographic peaks and compared against reference mass spectra from a commercially available National Institutes of Standards and Technology library. Validation of the use of S-(þ)-MTPA as a replacement derivatizer for amphetamine quantitation was conducted with a series of side-by-side control experiments. Each contained two groups of a racemic amphetamine mixture at concentrations of 20, 50, 75, 100 (reporting cutoff concentration), 150, 500 and 1,000 ng/ml in a certified drug-free urine matrix. Each amphetamine concentration contained 50,000, 25,000, 12,500 or 0ng/mL of phentermine. One group was derivatized with R-(2)-MTPA and the other with S-(þ)-MTPA. In order to demonstrate how the use of R-(2)-MTPA could increase the risk of peak misidentification in L-methamphetamine-overloaded specimens, a 98.7% chirally pure Cerilliant w L-amphetamine/methamphetamine isomer control set was made with analyte concentrations increasing in Interference and Concentration Problems in Amphetamines GC MS Assays 457
3 1,000 ng/ml increments from 1,000 to 10,000 ng/ml in certified urine. Following extraction, the samples were derivatized with R-(2)-MTPA and subjected to GC EI-MS analysis. In a separate experiment designed to demonstrate improved D-methamphetamine ion resolution in samples with high L-methamphetamine ion concentrations, duplicate 10-sample control sets were prepared. Each sample had a constant 100 ng/ml D-amphetamine concentration with L-methamphetamine concentrations increasing in 1,000 ng/ml increments from 1,000 to 10,000 ng/ml in certified urine. Following extraction, one set was derivatized with R-(2)-MTPA and the other with S-(þ)- MTPA followed by analysis with GC EI-MS. Results The number of phentermine-contaminated specimens triggering rework at FTDTL, Ft. Meade, was found to be significant. Based on the acceptability criterion that only spectra with a minimum match of 70% to the reference library spectrum were considered reliable, 68% of the 279 service member specimens that had screened initially positive for amphetamines, but confirmed amphetamine negative during a 2-week period in July of 2013 were shown to contain phentermine (data not shown). Assuming that the submission frequency of phentermine-contaminated specimens remains constant and that the number can be extrapolated to.4,000 specimens per year with the potential to interfere with the testing process. Substitution of S-(þ)-MTPA for R-(2)-MTPA can help reduce the need for confirmation rework in some of those cases and for routine amphetamines GC EI-MS assays the two derivatizers were shown to be equivalent. Concentration calculations of phentermine-free amphetamine controls used in the experiments showed no significant difference between those derivatized with R-(2)-MTPA and those derivatized with S-(þ)-MTPA. Additionally, with both derivatizers, chromatography results for target and qualifier analyte ions were similar with or without phentermine and at all phentermine to amphetamine concentration ratios. Unfortunately, these similarities also include phentermine-induced resolution problems with the m/z analyte qualifier ion that were present when using the R-(2)-MTPA derivatizer and were not improved with the use of S-(þ)-MTPA. Efforts to correct the issue by replacing the m/z with the m/z ion yielded similarly poor results even after various reductions in the He flow rate and a series of temperature ramp changes between 15 and 458C/s. However, derivatization with S-(þ)-MTPA eliminated all phentermine-induced problems affecting internal standard ions. Figure 1 illustrates this effect with the results of the control experiment using a 1:2,500 amphetamine to phentermine concentration (ng/ml) ratio the lowest studied. Figure 1a and b Figure 1. The results of a control experiment with a 20 50,000 amphetamines to phentermine concentration ratio (ng/ml) showing internal standard ion resolution when derivatized with either R-(2)-MTPA or S-(þ)-MTPA. (a) The D-methamphetamine/L-methamphetamine m/z internal standard quantification ions with R(2)-MTPA. The retention time of D-methamphetamine internal standard is 5.21 s. The peaks are unresolved due to phentermine interference. (b) The D-methamphetamine/L-methamphetamine m/z 98.0 internal standard qualification ions with R-(2)-MTPA. The peaks are unresolved due to phentermine interference. (c) and (d) The result of an identical control experiment with S-(þ)-MTPA used in place of R-(2)-MTPA. The retention times of the m/z and m/z 98.0 D-methamphetamine internal standard ions are now 4.99 and 4.98 s, respectively. The effect of phentermine interference on the D-methamphetamine internal standard ions has been removed. 458 Hamilton et al.
4 Figure 2. Data from a GC EI-MS assay of a control sample with 98.7% pure L-methamphetamine at 8,000 ng/ml. The data show how specimens with high concentrations of L-methamphetamine may be misidentified by integration software when L-methamphetamine peaks migrate into retention time windows defined for D-methamphetamine following derivatization with R-(2)-MTPA. All chromatographic and mass ion ratio criteria are met. (a) m/z target analyte ion. (b) m/z qualifier analyte ion. (c) m/z qualifier ion. (d) m/z D-methamphetamine internal standard quantification ion (the integrated peak). (e) m/z 98.0 D-methamphetamine internal standard qualifier ion (integrated peak). shows the chromatographic failure of the D-methamphetamine internal standard quantification and qualifier ions as result of phentermine interference. Figure 1C and D shows the result of the same experiment, but with S-(þ)-MTPA used as the derivatizing agent in place of R-(2)-MTPA. In this case, although chromatography for the analyte ions failed acceptability criteria, the effect of phentermine interference on the D-methamphetamine internal standard ions has been completely removed and the results of such an assay in Army labs could be released as negative without the need for rework. As Figure 2 shows, specimens with high concentrations of L-methamphetamine could lead to peak misidentification by integration software when L-methamphetamine peaks migrate into retention time windows defined for D-methamphetamine when derivatized with R-(2)-MTPA. Figure 2d and e shows the D-methamphetamine/L-methamphetamine internal standard quantification and qualifier ion windows the D- methamphetamine peak is properly integrated and meets all US Army FTDTL chromatographic and mass ion ratio acceptability criteria. Figure 2a c shows the drug target and two qualifier ion windows, respectively, with properly integrated and chromatographically acceptable peaks. However, as described above, the only analyte present in this control sample is 98.7% pure L-methamphetamine at 8,000 ng/ml. In fact, in this experiment with R-(2)-MTPA-derivatized analyte, all L-methamphetamine concentrations between 8,000 and 10,000 ng/ml produced results that were misidentified as D-methamphetamine by the integration software due to retention time shifts concentrations achievable with the use of a typical Vicks w inhaler (4). The data plotted in Figure 3 tracks the shift in retention times of the L-methamphetamine (O) signal peak as a function of L-methamphetamine concentrations. Clearly, when such samples are instead derivatized with S-(þ)-MTPA, L-methamphetamine peaks will elute at later retention times, sufficiently far from the defined D-methamphetamine integration window that any potential for peak misidentification will be effectively avoided. The second dataset ( ) in Figure 3 shows that there is also a concurrent increase in the retention time of the D-methamphetamine internal standard ion peak; however, in these experiments the change was not enough for the peak to fall outside of the defined integration window (see Figure 2). Furthermore, the ratio of the rates of retention time shift for L-methamphetamine and the D-methamphetamine internal standard with respect to L-methamphetamine concentration, dt L drug =dt D IS ; shows that the L-methamphetamine peak shifts 1.33 times faster than the D-methamphetamine internal standard peak. Finally, Figure 4 shows the advantage S-(þ)-MTPA has over R-(2)-MTPA as a derivatizing agent in terms of D-methamphetamine resolution when concentrations of L-methamphetamine Interference and Concentration Problems in Amphetamines GC MS Assays 459
5 Figure 3. L-Methamphetamine (O) and D-methamphetamine ( ) internal standard retention times as a function of L-methamphetamine drug concentration from a control experiment using 98.7% pure L-methamphetamine. Concentrations at and.8,000 ng/ml caused L-methamphetamine (O) signal peaks to migrate into the retention time integration windows of D-methamphetamine analyte and qualifier ions. D-Methamphetamine ( ) internal standard peaks also migrate as a function of L-methamphetamine drug concentration, but at rate that is 33% slower. Figure 4. Choosing S-(þ)-MTPA over R-(2)-MTPA as a derivatizing agent for methamphetamines improves D-methamphetamine peak resolution when concentrations of L-methamphetamine are high relative to the D isomer. (a) Target and qualifier ions of R-(2)-MTPA-derivatized D-methamphetamine at a concentration of 100 ng/ml and L-methamphetamine at a concentration 10,000 ng/ml. None of the three ions meet DoD resolution acceptability criteria. (b) The same experiment, but with S-(þ)-MTPA used as the derivatizing agent. In this case, all three ions are now resolved and all mass ion ratios are in acceptable ranges. 460 Hamilton et al.
6 are high. In this experiment, D-amphetamine concentrations were held constant at 100 ng/ml while L-amphetamine concentrations were increased in increments of 1,000 ng/ml. Analysis showed samples derivatized with R-(2)-MTPA failed resolution criteria with L-methamphetamine concentrations at and.2,000 ng/ml, but analysis of all samples derivatized with S-(þ)-MTPA displayed well-resolved D-methamphetamine ion peaks even at the highest L-methamphetamine concentrations. Figure 4a shows the R-(2)-MTPA-derivatized target and qualifier ions with 100 ng/ml D-methamphetamine and 10,000 ng/ml L-methamphetamine. Figure 4b shows the same experiment, but with S-(þ)-MTPA used as the derivatizing agent all DoD-defined chromatographic and mass ion ratio criteria are met for analyte and internal standard ions. resolution of D-methamphetamine peaks in the presence of high L-methamphetamine concentrations. Acknowledgments The authors thank William Bronner, PhD for suggesting the use of (S)-(þ)-MTPA in place of (R)-(2)-MTPA as a means of improving this method; Ms Paula Underwood for preparing all experimental samples and controls; and Ms. Michelle Serafin and Mr James Callies, FTCB for their thoughtful critiques of this report. The authors are especially grateful to Major Matthew Moser, PhD, and Captain Robert Nadeau, PhD of the United States Army Forensic Toxicology Drug Testing Laboratory, Fort Meade, for their generous support. Conclusion Substitution of R-(2)-MTPA with S-(þ)-MTPA as an amphetamines derivatizing agent should aid drug testing laboratories which are experiencing GC EI-MS assay failures as a result of phentermine interference with internal standards. In particular, the use of S-(þ)-MTPA should allow data review officials to release negative D-amphetamine results without subjecting the original specimen to re-extractions and GC EI-MS analyses. Most importantly, S-(þ)-MTPA would ensure the avoidance of peak misidentification in cases of high L-methamphetamine concentrations. Both problems have the potential to undermine the accuracy of reported results. Reducing these uncertainties with a simple, cost-neutral change in derivatizing agent is easy, requiring no modification to existing amphetamines GC EI-MS methods and has the added benefit of enhancing the References 1. Paul, B.D., Jemionek, J., Lesser, D., Jacobs, A., Searles, D.A. (2004) Enantiomeric separation and quantitation of (þ/-)-amphetamine, (þ/-)-methamphetamine, (þ/-)-mda, (þ/-)-mdma, and (þ/-)-mdea in urine specimens by GC-EI-MS after derivatization with (R)-(-)- or (S)-(þ)-alpha-methoxy-alpha-(trifluoromethy)phenylacetyl chloride (MTPA). Journal of Analytical Toxicology, 28, Stout, P.R., Klette, K.L., Horn, C.K. (2004) Evaluation of ephedrine, pseudoephedrine and phenylpropanolamine concentrations in human urine samples and a comparison of the specificity of DRI amphetamines and Abuscreen online (KIMS) amphetamines screening immunoassays. Journal of Forensic Sciences, 49, Herring, C., Muzyk, A.J., Johnston, C. (2011) Interferences with urine drug screens. Journal of Pharmacy Practice, 24, Fitzgerald, R.L., Ramos, J.M., Jr, Bogema, S.C., Poklis, A. (1988) Resolution of methamphetamine stereoisomers in urine drug testing: urinary excretion of R(2)-methamphetamine following use of nasal inhalers. Journal of Analytical Toxicology, 12, Interference and Concentration Problems in Amphetamines GC MS Assays 461
Abstract. Introduction
 Quantitative and Isomeric Determination of Amphetamine and Methamphetamine from Urine using a Nonprotic Elution Solvent and R(-)-@-Methoxy-@-Trifluoromethylphenylacetic Acid Chloride Derivatization Justin
Quantitative and Isomeric Determination of Amphetamine and Methamphetamine from Urine using a Nonprotic Elution Solvent and R(-)-@-Methoxy-@-Trifluoromethylphenylacetic Acid Chloride Derivatization Justin
Quantitative Method for Amphetamines, Phentermine, and Designer Stimulants Using an Agilent 6430 LC/MS/MS
 Quantitative Method for Amphetamines, Phentermine, and Designer Stimulants Using an Agilent 6430 LC/MS/MS Application Note Forensic Toxicology Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca
Quantitative Method for Amphetamines, Phentermine, and Designer Stimulants Using an Agilent 6430 LC/MS/MS Application Note Forensic Toxicology Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca
Toxicology Screening of Whole Blood Extracts Using GC/Triple Quadrupole/MS
 Toxicology Screening of Whole Blood Extracts Using GC/Triple Quadrupole/MS Application Note Forensic Toxicology Authors Bruce Quimby and Mike Szelewski Agilent Technologies, Inc. 2850 Centerville Road
Toxicology Screening of Whole Blood Extracts Using GC/Triple Quadrupole/MS Application Note Forensic Toxicology Authors Bruce Quimby and Mike Szelewski Agilent Technologies, Inc. 2850 Centerville Road
Determination of Amphetamine and Derivatives in Urine
 Determination of Amphetamine and Derivatives in Urine Using a Modified QuEChERS and Capillary Electrophoresis Tandem Mass Spectrometry Analysis Application Note Authors Vagner B. dos Santos and Claudimir
Determination of Amphetamine and Derivatives in Urine Using a Modified QuEChERS and Capillary Electrophoresis Tandem Mass Spectrometry Analysis Application Note Authors Vagner B. dos Santos and Claudimir
Determination of Gamma-Hydroxy-Butyrate (GHB) in Biological Samples
 Determination of Gamma-Hydroxy-Butyrate (GHB) in Biological Samples Application Note Forensic Toxicology Authors Joe Crifasi Saint Louis University Forensic Toxicology Laboratory Saint Louis, MO, USA Ron
Determination of Gamma-Hydroxy-Butyrate (GHB) in Biological Samples Application Note Forensic Toxicology Authors Joe Crifasi Saint Louis University Forensic Toxicology Laboratory Saint Louis, MO, USA Ron
Determination of Bath Salts (Pyrovalerone Analogs) in Biological Samples
 Determination of Bath Salts (Pyrovalerone Analogs) in Biological Samples Application Note Forensic Toxicology Authors Joe Crifasi Saint Louis University Forensic Toxicology Laboratory Saint Louis, Mo.
Determination of Bath Salts (Pyrovalerone Analogs) in Biological Samples Application Note Forensic Toxicology Authors Joe Crifasi Saint Louis University Forensic Toxicology Laboratory Saint Louis, Mo.
Quantitative Analysis of Amphetamine-Type Drugs by Extractive Benzoylation and LC/MS/MS. Application. Introduction. Authors. Abstract.
 Quantitative Analysis of Amphetamine-Type Drugs by Extractive Benzoylation and LC/MS/MS Application Forensics Authors Neil Campbell, B. Sc. Forensic Science Laboratory Chemistry Centre (WA) 5 Hay Street
Quantitative Analysis of Amphetamine-Type Drugs by Extractive Benzoylation and LC/MS/MS Application Forensics Authors Neil Campbell, B. Sc. Forensic Science Laboratory Chemistry Centre (WA) 5 Hay Street
Workflow for Screening and Quantification of the SAMHSA (NIDA) Panel in Urine Using UHPLC-TOF
 APPLICATIO OTE Liquid Chromatography/ Mass Spectrometry Authors: Avinash Dalmia Joanne Mather PerkinElmer, Inc. Shelton, CT Workflow for Screening and Quantification of the SAMHSA (IDA) Panel in Urine
APPLICATIO OTE Liquid Chromatography/ Mass Spectrometry Authors: Avinash Dalmia Joanne Mather PerkinElmer, Inc. Shelton, CT Workflow for Screening and Quantification of the SAMHSA (IDA) Panel in Urine
Elevated Urine Zinc Concentration Reduces the Detection of Methamphetamine, Cocaine, THC and Opiates in Urine by EMIT
 Journal of Analytical Toxicology 2013;37:665 669 doi:10.1093/jat/bkt056 Advance Access publication July 10, 2013 Article Elevated Urine Zinc Concentration Reduces the Detection of Methamphetamine, Cocaine,
Journal of Analytical Toxicology 2013;37:665 669 doi:10.1093/jat/bkt056 Advance Access publication July 10, 2013 Article Elevated Urine Zinc Concentration Reduces the Detection of Methamphetamine, Cocaine,
Determination of red blood cell fatty acid profiles in clinical research
 Application Note Clinical Research Determination of red blood cell fatty acid profiles in clinical research Chemical ionization gas chromatography tandem mass spectrometry Authors Yvonne Schober 1, Hans
Application Note Clinical Research Determination of red blood cell fatty acid profiles in clinical research Chemical ionization gas chromatography tandem mass spectrometry Authors Yvonne Schober 1, Hans
A NOVEL METHOD OF M/Z DRIFT CORRECTION FOR OA-TOF MASS SPECTROMETERS BASED ON CONSTRUCTION OF LIBRARIES OF MATRIX COMPONENTS.
 A NOVEL METHOD OF M/Z DRIFT CORRECTION FOR OA-TOF MASS SPECTROMETERS BASED ON CONSTRUCTION OF LIBRARIES OF MATRIX COMPONENTS. Martin R Green*, Keith Richardson, John Chipperfield, Nick Tomczyk, Martin
A NOVEL METHOD OF M/Z DRIFT CORRECTION FOR OA-TOF MASS SPECTROMETERS BASED ON CONSTRUCTION OF LIBRARIES OF MATRIX COMPONENTS. Martin R Green*, Keith Richardson, John Chipperfield, Nick Tomczyk, Martin
A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography
 Application Note LCMS-109 A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography Time of Flight (LC-TOF) Mass Spectrometry Introduction Many clinical
Application Note LCMS-109 A Simple and Accurate Method for the Rapid Quantitation of Drugs of Abuse in Urine Using Liquid Chromatography Time of Flight (LC-TOF) Mass Spectrometry Introduction Many clinical
MEDICAL POLICY Drug Testing
 POLICY: PG0069 ORIGINAL EFFECTIVE: 01/01/11 LAST REVIEW: 11/13/18 MEDICAL POLICY Drug Testing GUIDELINES This policy does not certify benefits or authorization of benefits, which is designated by each
POLICY: PG0069 ORIGINAL EFFECTIVE: 01/01/11 LAST REVIEW: 11/13/18 MEDICAL POLICY Drug Testing GUIDELINES This policy does not certify benefits or authorization of benefits, which is designated by each
NEW YORK STATE DEPARTMENT OF HEALTH CLINICAL LABORATORY EVALUATION PROGRAM
 Any Forensic Toxicology standards not addressed here remain in effect. (changes are underlined) Forensic Toxicology Sustaining Standard of Practice 27 (FT S27): Urine SVT Calibration and QC Requirements
Any Forensic Toxicology standards not addressed here remain in effect. (changes are underlined) Forensic Toxicology Sustaining Standard of Practice 27 (FT S27): Urine SVT Calibration and QC Requirements
Introduction. Goals. Peter Stout
 Introduction Peter Stout Dr. Peter Stout, a senior research forensic scientist in the Center for Forensic Sciences (CFS) at RTI International, has more than 15 years of experience in forensic urine drug
Introduction Peter Stout Dr. Peter Stout, a senior research forensic scientist in the Center for Forensic Sciences (CFS) at RTI International, has more than 15 years of experience in forensic urine drug
Douglas A. Searles Navy Drug Screening Laboratory, San Diego, California Abstract[ Introduction
 Enantiomeric Separation and Quantitation of (_+)-Amphetamine, (_+)-Methamphetamine, (_+)-MDA, (_+)-MDMA, and (_+)-MDEA in Urine Specimens by GC-EI-MS after Derivatization with (R)-(-)- or (S)-(+)- Methoxy-(x-(trifluoromethy)phenylacetyl
Enantiomeric Separation and Quantitation of (_+)-Amphetamine, (_+)-Methamphetamine, (_+)-MDA, (_+)-MDMA, and (_+)-MDEA in Urine Specimens by GC-EI-MS after Derivatization with (R)-(-)- or (S)-(+)- Methoxy-(x-(trifluoromethy)phenylacetyl
Amphetamines, Phentermine, and Designer Stimulant Quantitation Using an Agilent 6430 LC/MS/MS
 Amphetamines, Phentermine, and Designer Stimulant Quantitation Using an Agilent 643 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia
Amphetamines, Phentermine, and Designer Stimulant Quantitation Using an Agilent 643 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia
Public Understanding of Chemistry
 Public Understanding of Chemistry Public Understanding of Chemistry: Chemistry and its social-political-economic contexts continue to change. Chemistry and chemistry-based technology that impact our lives
Public Understanding of Chemistry Public Understanding of Chemistry: Chemistry and its social-political-economic contexts continue to change. Chemistry and chemistry-based technology that impact our lives
Rapid and Robust Detection of THC and Its Metabolites in Blood
 Rapid and Robust Detection of THC and Its Metabolites in Blood Application Note Forensics/Doping Control Author Stephan Baumann Agilent Technologies, Inc. Santa Clara CA 95051 USA Abstract A robust method
Rapid and Robust Detection of THC and Its Metabolites in Blood Application Note Forensics/Doping Control Author Stephan Baumann Agilent Technologies, Inc. Santa Clara CA 95051 USA Abstract A robust method
Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate Mass AxION 2 TOF Mass Spectrometer
 application Note Liquid Chromatography/ Mass Spectrometry Authors Sharanya Reddy Blas Cerda PerkinElmer, Inc. Shelton, CT USA Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate
application Note Liquid Chromatography/ Mass Spectrometry Authors Sharanya Reddy Blas Cerda PerkinElmer, Inc. Shelton, CT USA Quantitative Analysis of Drugs of Abuse in Urine using UHPLC Coupled to Accurate
Shawn P. Vorce Technical Director Chesapeake Toxicology Resources
 Shawn P. Vorce Technical Director Chesapeake Toxicology Resources "The opinions or assertions presented hereafter are the private views of Shawn P. Vorce and should not be construed as official or as reflecting
Shawn P. Vorce Technical Director Chesapeake Toxicology Resources "The opinions or assertions presented hereafter are the private views of Shawn P. Vorce and should not be construed as official or as reflecting
Chromatographic and Mass Spectral Studies on Methoxymethcathinones Related to 3,4-Methylenedioxymethamphetamine
 Chromatographic and Mass Spectral Studies on Methoxymethcathinones Related to 3,4-Methylenedioxymethamphetamine Tamer Awad, C. Randall Clark*, and Jack DeRuiter Department of Pharmacal Sciences, School
Chromatographic and Mass Spectral Studies on Methoxymethcathinones Related to 3,4-Methylenedioxymethamphetamine Tamer Awad, C. Randall Clark*, and Jack DeRuiter Department of Pharmacal Sciences, School
Determination of Benzodiazepines in Urine by CE-MS/MS
 Determination of Benzodiazepines in Urine by CE-MS/MS Application ote Forensic Toxicology Authors audimir Lucio do Lago Department of Fundamental Chemistry, Institute of Chemistry University of São Paulo,
Determination of Benzodiazepines in Urine by CE-MS/MS Application ote Forensic Toxicology Authors audimir Lucio do Lago Department of Fundamental Chemistry, Institute of Chemistry University of São Paulo,
Independent Column Temperature Control Using an LTM Oven Module for Improved Multidimensional Separation of Chiral Compounds
 Independent Column Temperature Control Using an LTM Oven Module for Improved Multidimensional Separation of Chiral Compounds Application Note Food and Flavors Author Frank David Research Institute for
Independent Column Temperature Control Using an LTM Oven Module for Improved Multidimensional Separation of Chiral Compounds Application Note Food and Flavors Author Frank David Research Institute for
Payment Policy: Urine Specimen Validity Testing Reference Number: CC.PP.056 Product Types: ALL Effective Date: 11/01/2017 Last Review Date:
 Payment Policy: Reference Number: CC.PP.056 Product Types: ALL Effective Date: 11/01/2017 Last Review Date: Coding Implications Revision Log See Important Reminder at the end of this policy for important
Payment Policy: Reference Number: CC.PP.056 Product Types: ALL Effective Date: 11/01/2017 Last Review Date: Coding Implications Revision Log See Important Reminder at the end of this policy for important
Pain Management Drug Testing: A Laboratory Perspective
 Ernest Jimenez III, M.T.(ASCP) Nothing to declare. Laboratory manager at Pharos Diagnostics, LLC., located in Tucson, AZ. Pain Management Drug Testing: A Laboratory Perspective 1 2 Pain Management Drug
Ernest Jimenez III, M.T.(ASCP) Nothing to declare. Laboratory manager at Pharos Diagnostics, LLC., located in Tucson, AZ. Pain Management Drug Testing: A Laboratory Perspective 1 2 Pain Management Drug
Metabolite identification in metabolomics: Metlin Database and interpretation of MSMS spectra
 Metabolite identification in metabolomics: Metlin Database and interpretation of MSMS spectra Jeevan K. Prasain, PhD Department of Pharmacology and Toxicology, UAB jprasain@uab.edu Outline Introduction
Metabolite identification in metabolomics: Metlin Database and interpretation of MSMS spectra Jeevan K. Prasain, PhD Department of Pharmacology and Toxicology, UAB jprasain@uab.edu Outline Introduction
Quantitative Analysis of -Hydroxybutyrate in Hair Using Target Analyte Finding Processing of Comprehensive GC-HRT Data
 Quantitative Analysis of -Hydroxybutyrate in Hair Using Target Analyte Finding Processing of Comprehensive GC-HRT Data LECO Corporation; Saint Joseph, Michigan USA Key Words: Pegasus GC-HRT, GHB, Hair,
Quantitative Analysis of -Hydroxybutyrate in Hair Using Target Analyte Finding Processing of Comprehensive GC-HRT Data LECO Corporation; Saint Joseph, Michigan USA Key Words: Pegasus GC-HRT, GHB, Hair,
Liquid Liquid Extraction of Gamma Hydroxybutyric Acid (GHB) in Blood and Urine for GC-MS analysis
 Liquid Liquid Extraction of Gamma Hydroxybutyric Acid (GHB) in Blood and Urine for GC-MS analysis 1.0 Purpose - This procedure specifies the required elements for the extraction of GHB from blood, urine
Liquid Liquid Extraction of Gamma Hydroxybutyric Acid (GHB) in Blood and Urine for GC-MS analysis 1.0 Purpose - This procedure specifies the required elements for the extraction of GHB from blood, urine
MEDICAL POLICY Drug Testing
 POLICY: PG0069 ORIGINAL EFFECTIVE: 01/01/11 LAST REVIEW: 04/10/18 MEDICAL POLICY Drug Testing GUIDELINES This policy does not certify benefits or authorization of benefits, which is designated by each
POLICY: PG0069 ORIGINAL EFFECTIVE: 01/01/11 LAST REVIEW: 04/10/18 MEDICAL POLICY Drug Testing GUIDELINES This policy does not certify benefits or authorization of benefits, which is designated by each
Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies
 Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies Frederick G. Strathmann, PhD, DABCC, (CC,TC) Medical Director, Toxicology Associate Scientific Director of MS ARUP Laboratories
Laboratory Testing to Support Pain Management: Methods, Concepts and Case Studies Frederick G. Strathmann, PhD, DABCC, (CC,TC) Medical Director, Toxicology Associate Scientific Director of MS ARUP Laboratories
Drug Testing Policy. Reimbursement Policy CMS Approved By. Policy Number. Annual Approval Date. Reimbursement Policy Oversight Committee
 Reimbursement Policy CMS 1500 Drug Testing Policy Policy Number 2017R6005A Annual Approval Date 05/10/2017 Approved By Reimbursement Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY
Reimbursement Policy CMS 1500 Drug Testing Policy Policy Number 2017R6005A Annual Approval Date 05/10/2017 Approved By Reimbursement Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY
Two chiral centres (diastereoisomers)
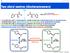 1 Two chiral centres (diastereoisomers) Mirror N S N Two enantiomers differ by absolute configuration N N 2 N 2 N A molecule with 1 stereogenic centre exists as 2 stereoisomers or enantiomers Enantiomers
1 Two chiral centres (diastereoisomers) Mirror N S N Two enantiomers differ by absolute configuration N N 2 N 2 N A molecule with 1 stereogenic centre exists as 2 stereoisomers or enantiomers Enantiomers
Comprehensive Forensic Toxicology Screening in Serum using On-Line SPE LC-MS/MS
 Comprehensive Forensic Toxicology Screening in Serum using On-Line SPE LC-MS/MS SCIEX QTRAP 4500 LC-MS/MS System and Spark Holland PICO Adrian M. Taylor 1, Peter Ringeling 2, Martin Sibum 2, Stefan Sturm
Comprehensive Forensic Toxicology Screening in Serum using On-Line SPE LC-MS/MS SCIEX QTRAP 4500 LC-MS/MS System and Spark Holland PICO Adrian M. Taylor 1, Peter Ringeling 2, Martin Sibum 2, Stefan Sturm
Application Note. Abstract. Authors. Pharmaceutical
 Analysis of xycodone and Its Metabolites-oroxycodone, xymorphone, and oroxymorphone in Plasma by LC/MS with an Agilent ZRBAX StableBond SB-C18 LC Column Application ote Pharmaceutical Authors Linda L.
Analysis of xycodone and Its Metabolites-oroxycodone, xymorphone, and oroxymorphone in Plasma by LC/MS with an Agilent ZRBAX StableBond SB-C18 LC Column Application ote Pharmaceutical Authors Linda L.
How to Use TOF and Q-TOF Mass Spectrometers
 How to Use TOF and Q-TOF Mass Spectrometers October 2011 What do TOF and Q-TOF offer? TOF Fast scanning of full spectrum High resolution full scan spectra Accurate mass measurements Q-TOF Fast scanning
How to Use TOF and Q-TOF Mass Spectrometers October 2011 What do TOF and Q-TOF offer? TOF Fast scanning of full spectrum High resolution full scan spectra Accurate mass measurements Q-TOF Fast scanning
Drug Testing Basics. by Erowid
 Drug Testing Basics by Erowid INDEX 1. What do they Test for? 2. Test Types 3. Test Sensitivity 4. Detection Periods WHAT DO THEY TEST FOR? The first thing to know about drug testing is what the standard
Drug Testing Basics by Erowid INDEX 1. What do they Test for? 2. Test Types 3. Test Sensitivity 4. Detection Periods WHAT DO THEY TEST FOR? The first thing to know about drug testing is what the standard
Metabolite identification in metabolomics: Database and interpretation of MSMS spectra
 Metabolite identification in metabolomics: Database and interpretation of MSMS spectra Jeevan K. Prasain, PhD Department of Pharmacology and Toxicology, UAB jprasain@uab.edu utline Introduction Putative
Metabolite identification in metabolomics: Database and interpretation of MSMS spectra Jeevan K. Prasain, PhD Department of Pharmacology and Toxicology, UAB jprasain@uab.edu utline Introduction Putative
ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS
 ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS Barry K Logan PhD, DABFT National Director of Forensic Services, NMS Labs Willow Grove PA Disclaimer
ORAL FLUID AS A CHEMICAL TEST FOR THE DRE PROGRAM : HISTORY, THE FUTURE, AND PRACTICAL CONSIDERATIONS Barry K Logan PhD, DABFT National Director of Forensic Services, NMS Labs Willow Grove PA Disclaimer
DRUG TESTING POLICY. Policy Number: ADMINISTRATIVE T0 Effective Date: October 1, Related Policies None
 DRUG TESTING POLICY UnitedHealthcare Oxford Reimbursement Policy Policy Number: ADMINISTRATIVE 259.1 T0 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 APPLICABLE LINES
DRUG TESTING POLICY UnitedHealthcare Oxford Reimbursement Policy Policy Number: ADMINISTRATIVE 259.1 T0 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 APPLICABLE LINES
DRUG TESTING POLICY. Policy Number: ADMINISTRATIVE T0 Effective Date: March 1, Related Policies None
 DRUG TESTING POLICY UnitedHealthcare Oxford Reimbursement Policy Policy Number: ADMINISTRATIVE 259.3 T0 Effective Date: March 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 APPLICABLE LINES OF
DRUG TESTING POLICY UnitedHealthcare Oxford Reimbursement Policy Policy Number: ADMINISTRATIVE 259.3 T0 Effective Date: March 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 APPLICABLE LINES OF
Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research
 Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research Dominic Foley, Michelle Wills, and Lisa Calton Waters Corporation, Wilmslow, UK APPLICATION
Analysis of Testosterone, Androstenedione, and Dehydroepiandrosterone Sulfate in Serum for Clinical Research Dominic Foley, Michelle Wills, and Lisa Calton Waters Corporation, Wilmslow, UK APPLICATION
[ APPLICATION NOTE ] The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns INTRODUCTION APPLICATION BENEFITS
![[ APPLICATION NOTE ] The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns INTRODUCTION APPLICATION BENEFITS [ APPLICATION NOTE ] The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns INTRODUCTION APPLICATION BENEFITS](/thumbs/78/77330968.jpg) The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns Jacquelyn Runco, Andrew Aubin, and Catharine Layton Waters Corporation, Milford, MA, USA APPLICATION BENEFITS
The Separation of 8 -THC, 9 -THC, and Their Enantiomers by UPC 2 Using Trefoil Chiral Columns Jacquelyn Runco, Andrew Aubin, and Catharine Layton Waters Corporation, Milford, MA, USA APPLICATION BENEFITS
Ultrafast Analysis of Benzodiazepines in Urine by the Agilent RapidFire High-Throughput Triple Quadrupole Mass Spectrometry System
 Ultrafast Analysis of Benzodiazepines in Urine by the Agilent RapidFire High-Throughput Triple Quadrupole Mass Spectrometry System Application Note Forensic Toxicology Authors Nikunj R. Parikh, Michelle
Ultrafast Analysis of Benzodiazepines in Urine by the Agilent RapidFire High-Throughput Triple Quadrupole Mass Spectrometry System Application Note Forensic Toxicology Authors Nikunj R. Parikh, Michelle
C. Randall Clark* and Jack DeRuiter. F. Taylor Noggle. Abstract. Introduction
 Chromatographic and Mass Spectrometry Methods for the Differentiation of N-Methyl-1-(3,4-methylenedioxyphenyO^-butanamine from Regioisomeric Derivatives C. Randall Clark* and Jack DeRuiter Department of
Chromatographic and Mass Spectrometry Methods for the Differentiation of N-Methyl-1-(3,4-methylenedioxyphenyO^-butanamine from Regioisomeric Derivatives C. Randall Clark* and Jack DeRuiter Department of
Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS
 Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia Department
Validation of a Benzodiazepine and Z-Drug Method Using an Agilent 6430 LC/MS/MS Application Note Forensics Authors Jason Hudson, Ph.D., James Hutchings, Ph.D., and Rebecca Wagner, Ph.D. Virginia Department
Procedure for Toxicology Analysis Version 4 Toxicology Unit Effective Date: 05/10/2013. Toxicology Analysis
 Toxicology Analysis 1.0 Purpose - This procedure specifies the required elements for analyzing toxicology submissions and reporting the results of the analysis. 2.0 Scope This procedure applies to all
Toxicology Analysis 1.0 Purpose - This procedure specifies the required elements for analyzing toxicology submissions and reporting the results of the analysis. 2.0 Scope This procedure applies to all
DRUG TESTING POLICY. Policy Number: ADMINISTRATIVE T0 Effective Date: January 1, Related Policies None
 DRUG TESTING POLICY UnitedHealthcare Oxford Reimbursement Policy Policy Number: ADMINISTRATIVE 259.2 T0 Effective Date: January 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 APPLICABLE LINES
DRUG TESTING POLICY UnitedHealthcare Oxford Reimbursement Policy Policy Number: ADMINISTRATIVE 259.2 T0 Effective Date: January 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 APPLICABLE LINES
Procedure for Toxicology Analysis Version 7 Toxicology Unit Effective Date: 03/14/2014 Issued by Drug Chemistry Forensic Scientist Manager
 Toxicology Analysis 1.0 Purpose - This procedure specifies the required elements for analyzing toxicology submissions and reporting drug testing results. 2.0 Scope This procedure applies to all submissions
Toxicology Analysis 1.0 Purpose - This procedure specifies the required elements for analyzing toxicology submissions and reporting drug testing results. 2.0 Scope This procedure applies to all submissions
Drug Testing Policy. Approved By 05/10/2017. Application This reimbursement policy applies to UnitedHealthcare Community Plan Medicaid products.
 Policy Number 2017R6005B Annual Approval Date Drug Testing Policy 05/10/2017 Approved By Reimbursement Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible for
Policy Number 2017R6005B Annual Approval Date Drug Testing Policy 05/10/2017 Approved By Reimbursement Policy Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY You are responsible for
Analysis of drugs of abuse in biological matrix using Time of Flight technology
 Analysis of drugs of abuse in biological matrix using Time of Flight technology Niclas Stephanson, PhD Department of Clinical Pharmacology Karolinska University Hospital Karolinska Institutet Stockholm,
Analysis of drugs of abuse in biological matrix using Time of Flight technology Niclas Stephanson, PhD Department of Clinical Pharmacology Karolinska University Hospital Karolinska Institutet Stockholm,
Student Handout. This experiment allows you to explore the properties of chiral molecules. You have
 Student Handout This experiment allows you to explore the properties of chiral molecules. You have learned that some compounds exist as enantiomers non-identical mirror images, such as your left and right
Student Handout This experiment allows you to explore the properties of chiral molecules. You have learned that some compounds exist as enantiomers non-identical mirror images, such as your left and right
Drug Testing Technologies: Sweat Patch
 Drug Testing Technologies: Sweat Patch A recent innovation in the science of drug testing, the PharmChek Drugs of Abuse Patch - or "sweat patch" - is used to test for various illegal drugs. The sweat patch
Drug Testing Technologies: Sweat Patch A recent innovation in the science of drug testing, the PharmChek Drugs of Abuse Patch - or "sweat patch" - is used to test for various illegal drugs. The sweat patch
4-Fluoroethamphetamine
 NMS Labs 2300 Stratford Ave Willow Grove, PA 19090 4-Fluoroethamphetamine Sample Type: Seized Material Latest Revision: January 3, 2019 Date Received: July 31, 2018 Date of Report: January 3, 2019 1. GENERAL
NMS Labs 2300 Stratford Ave Willow Grove, PA 19090 4-Fluoroethamphetamine Sample Type: Seized Material Latest Revision: January 3, 2019 Date Received: July 31, 2018 Date of Report: January 3, 2019 1. GENERAL
What You Can t See Can Hurt You. How MS/MS Specificity Can Bite Your Backside
 What You Can t See Can Hurt You How MS/MS Specificity Can Bite Your Backside Johan van den Heever, Tom Thompson, and Don Noot Agri-Food Laboratories Branch Advances in Trace rganic Residue Analysis early
What You Can t See Can Hurt You How MS/MS Specificity Can Bite Your Backside Johan van den Heever, Tom Thompson, and Don Noot Agri-Food Laboratories Branch Advances in Trace rganic Residue Analysis early
METABOLISM OF SELEGILINE IN HUMANS Identification, Excretion, and Stereochemistry of Urine Metabolites
 0090-9556/97/2506-0657 662$02.00/0 DRUG METABOLISM AND DISPOSITION Vol. 25, No. 6 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in U.S.A. METABOLISM OF SELEGILINE
0090-9556/97/2506-0657 662$02.00/0 DRUG METABOLISM AND DISPOSITION Vol. 25, No. 6 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in U.S.A. METABOLISM OF SELEGILINE
An Introduction and Overview on Comprehensive Two-Dimensional Gas Chromatography (GCxGC): New Opportunities for Unresolved Complex Mixtures
 An Introduction and Overview on Comprehensive Two-Dimensional Gas Chromatography (GCxGC): New Opportunities for Unresolved Complex Mixtures Daniela Cavagnino GC Product Manager Thermo Fisher Scientific,
An Introduction and Overview on Comprehensive Two-Dimensional Gas Chromatography (GCxGC): New Opportunities for Unresolved Complex Mixtures Daniela Cavagnino GC Product Manager Thermo Fisher Scientific,
Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System
 Application Note Forensics, Workplace Drug Testing Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System Authors Fred Feyerherm and Anthony Macherone Agilent Technologies, Inc.
Application Note Forensics, Workplace Drug Testing Detection of Cannabinoids in Oral Fluid with the Agilent 7010 GC-MS/MS System Authors Fred Feyerherm and Anthony Macherone Agilent Technologies, Inc.
PosterReprint OVERVIEW
 OVERVIEW A rapid multi-residue GC/MS method was developed for pesticides in foodstuffs. Plum extract was used as an example matrix. Analysis time was reduced from minutes to minutes. pesticides were analysed
OVERVIEW A rapid multi-residue GC/MS method was developed for pesticides in foodstuffs. Plum extract was used as an example matrix. Analysis time was reduced from minutes to minutes. pesticides were analysed
Glycerolipid Analysis. LC/MS/MS Analytical Services
 Glycerolipid Analysis LC/MS/MS Analytical Services Molecular Characterization and Quantitation of Glycerophospholipids in Commercial Lecithins by High Performance Liquid Chromatography with Mass Spectrometric
Glycerolipid Analysis LC/MS/MS Analytical Services Molecular Characterization and Quantitation of Glycerophospholipids in Commercial Lecithins by High Performance Liquid Chromatography with Mass Spectrometric
Workflow for Screening and Quantification of the SAMHSA (NIDA) Panel in Serum Using UHPLC-TOF
 APPLICATIO OTE Liquid Chromatography/ Mass Spectrometry Authors: Avinash Dalmia Bonnie Marmor PerkinElmer, Inc. Shelton, CT Workflow for Screening and Quantification of the SAMHSA (IDA) Panel in Serum
APPLICATIO OTE Liquid Chromatography/ Mass Spectrometry Authors: Avinash Dalmia Bonnie Marmor PerkinElmer, Inc. Shelton, CT Workflow for Screening and Quantification of the SAMHSA (IDA) Panel in Serum
The Development of LC/MS Methods for Determination of Polar Drugs of Abuse in Biological Samples
 WA20259 The Development of LC/MS Methods for Determination of Polar Drugs of Abuse in Biological Samples Michael S. Young and Kevin M. Jenkins Waters Corporation, 34 Maple Street, Milford, MA 01757 Introduction
WA20259 The Development of LC/MS Methods for Determination of Polar Drugs of Abuse in Biological Samples Michael S. Young and Kevin M. Jenkins Waters Corporation, 34 Maple Street, Milford, MA 01757 Introduction
EDUCATIONAL COMMENTARY rd TEST EVENT Chemistry Urine Drug Testing
 EDUCATIONAL COMMENTARY 2003 3 rd TEST EVENT Chemistry Urine Drug Testing Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE
EDUCATIONAL COMMENTARY 2003 3 rd TEST EVENT Chemistry Urine Drug Testing Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE
Metabolomics: quantifying the phenotype
 Metabolomics: quantifying the phenotype Metabolomics Promises Quantitative Phenotyping What can happen GENOME What appears to be happening Bioinformatics TRANSCRIPTOME What makes it happen PROTEOME Systems
Metabolomics: quantifying the phenotype Metabolomics Promises Quantitative Phenotyping What can happen GENOME What appears to be happening Bioinformatics TRANSCRIPTOME What makes it happen PROTEOME Systems
Library identifications for Lemnaceae
 Library identifications for Lemnaceae NP-001984 ESI+ NP-001984 present in, enriched in (15.47min) NP-001984 -MS^E CH 3 Moupinamide H NH NP-005512 + NP-016675 ESI- s NP-005512 NP-016675 NP-005512 present
Library identifications for Lemnaceae NP-001984 ESI+ NP-001984 present in, enriched in (15.47min) NP-001984 -MS^E CH 3 Moupinamide H NH NP-005512 + NP-016675 ESI- s NP-005512 NP-016675 NP-005512 present
DETERMINATION OF CANNABINOIDS, THC AND THC-COOH, IN ORAL FLUID USING AN AGILENT 6490 TRIPLE QUADRUPOLE LC/MS
 FORENSICS AND TOXICOLOGY ANALYSIS DETERMINATION OF CANNABINOIDS, THC AND THC-COOH, IN ORAL FLUID USING AN AGILENT 6490 TRIPLE QUADRUPOLE LC/MS Solutions for Your Analytical Business Markets and Applications
FORENSICS AND TOXICOLOGY ANALYSIS DETERMINATION OF CANNABINOIDS, THC AND THC-COOH, IN ORAL FLUID USING AN AGILENT 6490 TRIPLE QUADRUPOLE LC/MS Solutions for Your Analytical Business Markets and Applications
[Application Note] TOXICOLOGY SCREENING BY UPLC/PDA IN COMBINATION WITH AN EXTENSIVE COMPOUND LIBRARY
![[Application Note] TOXICOLOGY SCREENING BY UPLC/PDA IN COMBINATION WITH AN EXTENSIVE COMPOUND LIBRARY [Application Note] TOXICOLOGY SCREENING BY UPLC/PDA IN COMBINATION WITH AN EXTENSIVE COMPOUND LIBRARY](/thumbs/74/70091865.jpg) TOXICOLOGY SCREENING BY UPLC/PDA IN COMBINATION WITH AN EXTENSIVE COMPOUND LIBRARY Camille Chatenay, 1,2 Fabien Bévalot, 1,2 Cyril Mounier, 1 Jean Michel Prévosto 1 1 Hôpital d Instruction des Armées Desgenettes,
TOXICOLOGY SCREENING BY UPLC/PDA IN COMBINATION WITH AN EXTENSIVE COMPOUND LIBRARY Camille Chatenay, 1,2 Fabien Bévalot, 1,2 Cyril Mounier, 1 Jean Michel Prévosto 1 1 Hôpital d Instruction des Armées Desgenettes,
Confirmation by Triple Quadrupole LC/MS/MS for HHS-compliant Workplace Urine Drug Testing
 Confirmation by Triple Quadrupole LC/MS/MS for HHS-compliant Workplace Urine Drug Testing A complete applications solution for forensic toxicology laboratories John Hughes Agilent Technologies Pleasanton,
Confirmation by Triple Quadrupole LC/MS/MS for HHS-compliant Workplace Urine Drug Testing A complete applications solution for forensic toxicology laboratories John Hughes Agilent Technologies Pleasanton,
False-Positive Carbamazepine Results by Gas Chromatography Mass Spectrometry and VITROS 5600 Following a Massive Oxcarbazepine Ingestion
 False-Positive Results by Gas Chromatography Mass Spectrometry and VITRS 5600 Following a Massive xcarbazepine Ingestion Uttam Garg, 1 * Leo Johnson, 1 Amy Wiebold, 1 Angela Ferguson, 1 Clint Frazee, 1
False-Positive Results by Gas Chromatography Mass Spectrometry and VITRS 5600 Following a Massive xcarbazepine Ingestion Uttam Garg, 1 * Leo Johnson, 1 Amy Wiebold, 1 Angela Ferguson, 1 Clint Frazee, 1
Improving Benzodiazepine Immunoassay Sensitivity by Rapid Glucuronide Hydrolysis Technology
 Improving Benzodiazepine Immunoassay Sensitivity by Rapid Glucuronide Hydrolysis Technology Pongkwan (Nikki) Sitasuwan, Margarita Marinova, and L. Andrew Lee Integrated Micro-Chromatography Systems, LLC
Improving Benzodiazepine Immunoassay Sensitivity by Rapid Glucuronide Hydrolysis Technology Pongkwan (Nikki) Sitasuwan, Margarita Marinova, and L. Andrew Lee Integrated Micro-Chromatography Systems, LLC
Analysis of anti-epileptic drugs in human serum using an Agilent Ultivo LC/TQ
 Application Note Clinical Research Analysis of anti-epileptic drugs in human serum using an Agilent Ultivo LC/TQ Authors Jennifer Hitchcock 1, Lauren Frick 2, Peter Stone 1, and Vaughn Miller 2 1 Agilent
Application Note Clinical Research Analysis of anti-epileptic drugs in human serum using an Agilent Ultivo LC/TQ Authors Jennifer Hitchcock 1, Lauren Frick 2, Peter Stone 1, and Vaughn Miller 2 1 Agilent
High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring Buprenorphine and Norbuprenorphine in Urine
 High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring and in Urine Xiaolei Xie, Joe DiBussolo, Marta Kozak; Thermo Fisher Scientific, San Jose, CA Application Note 627 Key Words, norbuprenorphine,
High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring and in Urine Xiaolei Xie, Joe DiBussolo, Marta Kozak; Thermo Fisher Scientific, San Jose, CA Application Note 627 Key Words, norbuprenorphine,
Headspace Gas Chromatography to Quantitate and Identify Volatiles in Liquids
 Headspace Gas Chromatography to Quantitate and 1.0 Purpose - This procedure specifies the required elements for calibration and use of the headspace gas chromatograph to determine alcohol (ethanol, methanol,
Headspace Gas Chromatography to Quantitate and 1.0 Purpose - This procedure specifies the required elements for calibration and use of the headspace gas chromatograph to determine alcohol (ethanol, methanol,
Targeted and untargeted metabolic profiling by incorporating scanning FAIMS into LC-MS. Kayleigh Arthur
 Targeted and untargeted metabolic profiling by incorporating scanning FAIMS into LC-MS Kayleigh Arthur K.Arthur@lboro.ac.uk Introduction LC-MS is a highly used technique for untargeted profiling analyses
Targeted and untargeted metabolic profiling by incorporating scanning FAIMS into LC-MS Kayleigh Arthur K.Arthur@lboro.ac.uk Introduction LC-MS is a highly used technique for untargeted profiling analyses
Payment Policy Drug Testing EFFECTIVE DATE: POLICY LAST UPDATED:
 Payment Policy Drug Testing EFFECTIVE DATE: 05 23 2013 POLICY LAST UPDATED: 06 05 2018 OVERVIEW This policy documents the criteria and documentation requirements for immunoassay (IA) testing (also called
Payment Policy Drug Testing EFFECTIVE DATE: 05 23 2013 POLICY LAST UPDATED: 06 05 2018 OVERVIEW This policy documents the criteria and documentation requirements for immunoassay (IA) testing (also called
Analyze Drugs of Abuse with Agilent J&W Ultimate Plus Tubing in an Inert Flow Path
 Analyze Drugs of Abuse with J&W Ultimate Plus Tubing in an Inert Flow Path Application Note Forensic Toxicology Author Ngoc A Dang Technologies, Inc. Abstract J&W Ultimate Plus deactivated fused silica
Analyze Drugs of Abuse with J&W Ultimate Plus Tubing in an Inert Flow Path Application Note Forensic Toxicology Author Ngoc A Dang Technologies, Inc. Abstract J&W Ultimate Plus deactivated fused silica
Analysis and Quantitation of Cocaine on Currency Using GC-MS/MS. No. GCMS No. SSI-GCMS-1501
 Gas Chromatograph Mass Spectrometer No. GCMS-1501 Analysis and Quantitation of Cocaine on Currency Using GC-MS/MS Shilpi Chopra, Ph.D., Laura Chambers Introduction Cocaine (CAS # 50-36-2), a white crystalline
Gas Chromatograph Mass Spectrometer No. GCMS-1501 Analysis and Quantitation of Cocaine on Currency Using GC-MS/MS Shilpi Chopra, Ph.D., Laura Chambers Introduction Cocaine (CAS # 50-36-2), a white crystalline
Improving Selectivity in Quantitative Analysis Using MS 3 on a Hybrid Quadrupole-Linear Ion Trap Mass Spectrometer
 Improving Selectivity in Quantitative Analysis Using MS 3 on a Hybrid Quadrupole-Linear Ion Trap Mass Spectrometer Overview Increased selectivity is achieved in quantitative analysis by using MS 3. The
Improving Selectivity in Quantitative Analysis Using MS 3 on a Hybrid Quadrupole-Linear Ion Trap Mass Spectrometer Overview Increased selectivity is achieved in quantitative analysis by using MS 3. The
Clinical Mass Spectrometry Case Studies: How we can Read Between the Lines
 Clinical Mass Spectrometry Case Studies: How we can Read Between the Lines Alec Saitman Ph.D., DABCC (CC, TC) Director Special Chemistry and Toxicology Providence Regional Laboratories Disclosures None
Clinical Mass Spectrometry Case Studies: How we can Read Between the Lines Alec Saitman Ph.D., DABCC (CC, TC) Director Special Chemistry and Toxicology Providence Regional Laboratories Disclosures None
Advancing your Forensic Toxicology Analyses; Adopting the Latest in Mass Spectrometry Innovations
 Advancing your Forensic Toxicology Analyses; Adopting the Latest in Mass Spectrometry Innovations For Research Use Only. Not for use in diagnostic procedures. 1 2015 AB Sciex RUO-MKT-11-1018-A For research
Advancing your Forensic Toxicology Analyses; Adopting the Latest in Mass Spectrometry Innovations For Research Use Only. Not for use in diagnostic procedures. 1 2015 AB Sciex RUO-MKT-11-1018-A For research
Headspace Gas Chromatography to Quantitate and Identify Volatiles in Liquids
 Headspace Gas Chromatography to Quantitate and 1.0 Purpose - This procedure specifies the required elements for calibration and use of the headspace gas chromatograph to determine alcohol (ethanol, methanol,
Headspace Gas Chromatography to Quantitate and 1.0 Purpose - This procedure specifies the required elements for calibration and use of the headspace gas chromatograph to determine alcohol (ethanol, methanol,
Drug Testing Policy. Approved By 06/14/2017
 Drug Testing Policy Policy Number Annual Approval Date 06/14/2017 Approved By Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare Medicare Advantage
Drug Testing Policy Policy Number Annual Approval Date 06/14/2017 Approved By Oversight Committee IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare Medicare Advantage
September HCMC Toxicology Transition: Additional information and Frequently Asked Questions
 September 2016 HCMC Toxicology Transition: Additional information and Frequently Asked Questions Many clinicians have asked for more information about the Urine Drug Compliance Analysis (LAB8742) switch
September 2016 HCMC Toxicology Transition: Additional information and Frequently Asked Questions Many clinicians have asked for more information about the Urine Drug Compliance Analysis (LAB8742) switch
Ultra Performance Liquid Chromatography Coupled to Orthogonal Quadrupole TOF MS(MS) for Metabolite Identification
 22 SEPARATION SCIENCE REDEFINED MAY 2005 Ultra Performance Liquid Chromatography Coupled to Orthogonal Quadrupole TOF MS(MS) for Metabolite Identification In the drug discovery process the detection and
22 SEPARATION SCIENCE REDEFINED MAY 2005 Ultra Performance Liquid Chromatography Coupled to Orthogonal Quadrupole TOF MS(MS) for Metabolite Identification In the drug discovery process the detection and
A Comprehensive Screening of Illicit and Pain Management Drugs from Whole Blood Using SPE and LC/MS/MS
 A Comprehensive Screening of Illicit and Pain Management Drugs from Whole Blood Using SPE and LC/MS/MS Introduction Drug analysis from whole blood is gaining popularity due to a more complete measurement
A Comprehensive Screening of Illicit and Pain Management Drugs from Whole Blood Using SPE and LC/MS/MS Introduction Drug analysis from whole blood is gaining popularity due to a more complete measurement
Identifying New Cannabis Use with Urine Creatinine- Normalized THCCOOH Concentrations and Time Intervals Between Specimen Collections *
 Identifying New Cannabis Use with Urine Creatinine- Normalized THCCOOH Concentrations and Time Intervals Between Specimen Collections * Michael L. Smith 1, Allan J. Barnes 2, and Marilyn A. Huestis 2,
Identifying New Cannabis Use with Urine Creatinine- Normalized THCCOOH Concentrations and Time Intervals Between Specimen Collections * Michael L. Smith 1, Allan J. Barnes 2, and Marilyn A. Huestis 2,
Organic Chemistry Laboratory Fall Lecture 3 Gas Chromatography and Mass Spectrometry June
 344 Organic Chemistry Laboratory Fall 2013 Lecture 3 Gas Chromatography and Mass Spectrometry June 19 2013 Chromatography Chromatography separation of a mixture into individual components Paper, Column,
344 Organic Chemistry Laboratory Fall 2013 Lecture 3 Gas Chromatography and Mass Spectrometry June 19 2013 Chromatography Chromatography separation of a mixture into individual components Paper, Column,
Possibility of Use a Saliva for Determination Ethanol and Opiates
 Possibility of Use a Saliva for Determination Ethanol and Opiates Wojciech Piekoszewski 1,2, Wojciech Gubała 1, Ewa Janowska 1, Janusz Pach 3, Dariusz Zuba 1 1 Institute of Forensic Research, Westerplatte
Possibility of Use a Saliva for Determination Ethanol and Opiates Wojciech Piekoszewski 1,2, Wojciech Gubała 1, Ewa Janowska 1, Janusz Pach 3, Dariusz Zuba 1 1 Institute of Forensic Research, Westerplatte
Porphyrin testing and heavy metal toxicity: unresolved questions and concerns
 Porphyrin testing and heavy metal toxicity: unresolved questions and concerns by William Shaw, Ph.D. The assertion by Nataf et al (1) that the Laboratoire Philippe Auguste could detect a specific porphyrin
Porphyrin testing and heavy metal toxicity: unresolved questions and concerns by William Shaw, Ph.D. The assertion by Nataf et al (1) that the Laboratoire Philippe Auguste could detect a specific porphyrin
Learning Objectives. Drug Testing 10/17/2012. Utilization of the urine drug screen: The good, the bad, and the ugly
 Utilization of the urine drug screen: The good, the bad, and the ugly Jennifer A. Lowry, MD Chief, Section of Medical Toxicology Children s Mercy Hospital Kansas City, MO Learning Objectives Describe the
Utilization of the urine drug screen: The good, the bad, and the ugly Jennifer A. Lowry, MD Chief, Section of Medical Toxicology Children s Mercy Hospital Kansas City, MO Learning Objectives Describe the
Rapid, Simple Impurity Characterization with the Xevo TQ Mass Spectrometer
 Robert Plumb, Michael D. Jones, and Marian Twohig Waters Corporation, Milford, MA, USA INTRODUCTION The detection and characterization of impurities and degradation products of an active pharmaceutical
Robert Plumb, Michael D. Jones, and Marian Twohig Waters Corporation, Milford, MA, USA INTRODUCTION The detection and characterization of impurities and degradation products of an active pharmaceutical
A Novel Platform of On-line Sample Pre-treatment and LC/MS/MS Analysis for Screening and Quantitation of Illicit Drugs in Urine
 PO-CON737E A Novel Platform of On-line Sample Pre-treatment and LC/MS/MS Analysis for Screening and Quantitation of Illicit Drugs in Urine ASMS 7 WP 353 Shao Hua Chia ; Zhi Wei Edwin Ting ; Daisuke Kawakami
PO-CON737E A Novel Platform of On-line Sample Pre-treatment and LC/MS/MS Analysis for Screening and Quantitation of Illicit Drugs in Urine ASMS 7 WP 353 Shao Hua Chia ; Zhi Wei Edwin Ting ; Daisuke Kawakami
Supplementary Figure S1. Appearacne of new acetyl groups in acetylated lysines using 2,3-13 C 6 pyruvate as a tracer instead of labeled glucose.
 Supplementary Figure S1. Appearacne of new acetyl groups in acetylated lysines using 2,3-13 C 6 pyruvate as a tracer instead of labeled glucose. (a) Relative levels of both new acetylation and all acetylation
Supplementary Figure S1. Appearacne of new acetyl groups in acetylated lysines using 2,3-13 C 6 pyruvate as a tracer instead of labeled glucose. (a) Relative levels of both new acetylation and all acetylation
The Challenges of Analytical Method Validation for Hallucinogens and Designer Stimulants in Biological Samples Using LC-TOF
 The Challenges of Analytical Method Validation for Hallucinogens and Designer Stimulants in Biological Samples Using LC-TOF Barry K Logan, Ph.D., NMS Labs National Director, Forensic Services Goals The
The Challenges of Analytical Method Validation for Hallucinogens and Designer Stimulants in Biological Samples Using LC-TOF Barry K Logan, Ph.D., NMS Labs National Director, Forensic Services Goals The
SUPPLEMENTARY DATA. Materials and Methods
 SUPPLEMENTARY DATA Materials and Methods HPLC-UV of phospholipid classes and HETE isomer determination. Fractionation of platelet lipid classes was undertaken on a Spherisorb S5W 150 x 4.6 mm column (Waters
SUPPLEMENTARY DATA Materials and Methods HPLC-UV of phospholipid classes and HETE isomer determination. Fractionation of platelet lipid classes was undertaken on a Spherisorb S5W 150 x 4.6 mm column (Waters
What Can Dead People Tell Us? Postmortem Toxicology for Attorneys
 What Can Dead People Tell Us? Postmortem Toxicology for Attorneys Dr. Allison A. Muller Diplomate, American Board of Applied Toxicology Principal/Owner: Acri Muller Consulting, LLC Learning objectives
What Can Dead People Tell Us? Postmortem Toxicology for Attorneys Dr. Allison A. Muller Diplomate, American Board of Applied Toxicology Principal/Owner: Acri Muller Consulting, LLC Learning objectives
Probing for Packaging Migrants in a Pharmaceutical Impurities Assay Using UHPLC with UV and Mass Detection INTRODUCTION
 Probing for Packaging Migrants in a Pharmaceutical Impurities Assay Using UHPLC with UV and Mass Detection Michael Jones Waters Corporation, Wilmslow, UK APPLICATION BENEFITS The ACQUITY Arc System is
Probing for Packaging Migrants in a Pharmaceutical Impurities Assay Using UHPLC with UV and Mass Detection Michael Jones Waters Corporation, Wilmslow, UK APPLICATION BENEFITS The ACQUITY Arc System is
In-stream attenuation of neuro-active pharmaceuticals and their
 In-stream attenuation of neuro-active pharmaceuticals and their metabolites Supporting Information Analytical Methods and Figures (10 pages) Jeffrey H. Writer, Ronald C. Antweiler, Imma Ferrer, Joseph
In-stream attenuation of neuro-active pharmaceuticals and their metabolites Supporting Information Analytical Methods and Figures (10 pages) Jeffrey H. Writer, Ronald C. Antweiler, Imma Ferrer, Joseph
Rapid Hydrolysis of Benzodiazepines in Urine. Alicia Zook 1 and Crystal Xander B.S. 2. Cedar Crest College, Allentown, PA 1
 Rapid Hydrolysis of Benzodiazepines in Urine Alicia Zook 1 and Crystal Xander B.S. 2 Cedar Crest College, Allentown, PA 1 Health Network Laboratories, Allentown, PA 2 Abstract: Benzodiazepines are sedative/hypnotic
Rapid Hydrolysis of Benzodiazepines in Urine Alicia Zook 1 and Crystal Xander B.S. 2 Cedar Crest College, Allentown, PA 1 Health Network Laboratories, Allentown, PA 2 Abstract: Benzodiazepines are sedative/hypnotic
[ APPLICATION NOTE ] High Sensitivity Intact Monoclonal Antibody (mab) HRMS Quantification APPLICATION BENEFITS INTRODUCTION WATERS SOLUTIONS KEYWORDS
![[ APPLICATION NOTE ] High Sensitivity Intact Monoclonal Antibody (mab) HRMS Quantification APPLICATION BENEFITS INTRODUCTION WATERS SOLUTIONS KEYWORDS [ APPLICATION NOTE ] High Sensitivity Intact Monoclonal Antibody (mab) HRMS Quantification APPLICATION BENEFITS INTRODUCTION WATERS SOLUTIONS KEYWORDS](/thumbs/79/80328542.jpg) Yun Wang Alelyunas, Henry Shion, Mark Wrona Waters Corporation, Milford, MA, USA APPLICATION BENEFITS mab LC-MS method which enables users to achieve highly sensitive bioanalysis of intact trastuzumab
Yun Wang Alelyunas, Henry Shion, Mark Wrona Waters Corporation, Milford, MA, USA APPLICATION BENEFITS mab LC-MS method which enables users to achieve highly sensitive bioanalysis of intact trastuzumab
