Policy for the use of Irradiated blood products
|
|
|
- Lorraine Dorsey
- 5 years ago
- Views:
Transcription
1 Policy for the use of Irradiated blood products SharePoint Location General Policies and Guidelines SharePoint Index Directory Haematology and Blood Transfusion Sub Area - Key words (for search purposes) Irradiated blood products, Transfusion Central Index No 0662 v1 Endorsing Body Clinical Governance Committee Endorsement Date September 2010 Review Date September Lead author and designation Dr Kanchan Rege (if under review) Review led by N/A Policy for the use of Irradiated blood products September
2 KEY POINTS : This policy - Applies to all staff with responsibility for prescribing and administering blood and blood components. Gives guidance on when and how to request Irradiated blood products This policy has been assessed using an equality impact assessment initial screening template and is deemed to meet current equality requirements. The equality impact assessment does take into account the refusal by some religions to have blood products. The screening template can be found in Appendix 3. Policy for the use of Irradiated blood products September
3 Background In certain at risk patient groups, transfused donor lymphocytes that are compatible with the recipient, but which recognise the recipient as foreign, can engraft and initiate transfusion associated graft versus host disease (Ta-GvHD) Affected patients can develop skin rashes, diarrhoea, abnormal liver function and bone marrow failure. Death from infection usually occurs within 2-3 weeks of the transfusion. The use of gamma or X ray irradiation (25 Gy) inactivates the donor T-Lymphocytes whilst preserving the function of the other cells therefore preventing Ta-GvHD. For at risk patients, all red cell, (except cryopreserved) platelet and granulocyte transfusions should be irradiated. It is not necessary to irradiate fresh frozen plasma, cryoprecipitate or fractionated plasma. Since universal leucocyte depletion of blood products, there have been fewer reports of Ta-GVHD. These guidelines, however, give an indication of best transfusion practice. Include the reason for the document, which may include the publication of new evidence guiding practice, critical incidents or complaints. Purpose and scope of policy To ensure that irradiated blood products are requested for and transfused to patients who require them. 1 Definition of Terms Blood products- Any therapeutic product derived from human whole blood or plasma donations Irradiated blood component- Cellular blood component treated with 25 gray (Gy) gamma irradiation or Xray irradiation to inactivate lymphocytes that could cause graft-versus-host disease in a recipient. 2.1 Process The issue of irradiated blood components to those patients who require them. 2.2 Content Indications All transfusions from first or second-degree relatives, regardless of the patients immune status. HLA-selected platelets regardless of recipients immune status Intra-uterine transfusion blood should be transfused within 24hours of irradiation Exchange transfusions where there has been a previous IUT or if the donation is from a 1 st or 2 nd degree relative Policy for the use of Irradiated blood products September
4 Top-up transfusion in neonates only if previously a recipient of an intra-uterine or exchange transfusion. Bone marrow and stem cell transplant recipients from the time of conditioning treatment up to 3 months after transplant (autologous transplants) or 6 months (allogeneic transplants). It may be necessary to irradiate blood products for SCID patients for up to 2 years, and for patient with chronic GvHD if there is evidence of immuno-suppression NB It is not necessary to irradiate red cells or platelets for children or adults with acute leukaemia, except for HLA matched platelets or donations from first or second-degree relatives Transfusion to bone marrow donors prior to or during harvest Autologous marrow or stem-cell donations in the 7 days prior to harvest Hodgkin s disease Treatment with purine analogues (Fludarabine, Cladrabine and Deoxycoformycin). Patients with the following congenital immunity deficiencies o SCID o 3 rd and 4 th arch/pouch syndrome (Di George s) o Wiskott-Aldrich syndrome o Purine nucleoside phosphorylase deficiency o Cell-mediated immunodeficiency, not otherwise classified o Reticular dysgenesis o Adenosine deaminase deficiency o MHC Class I and II deficiency o Immunodeficiency with eosinophilia (Omenn s syndrome) o Ataxia telangiectasia NB - There is currently no indication for the irradiation of blood for infants or children with HIV or AIDS, however this remains under review How to request irradiated blood products: Using the Anglia requesting system, the requirement for irradiated blood products should be indicated by clicking the yes button at the special requirement option screen. The transfusion laboratory must also be informed by telephone of the requirement for irradiated blood products Once the requirement for irradiated blood products has been communicated to the Transfusion Laboratory, all further blood products issued will be irradiated until the Transfusion Laboratory is informed otherwise. The requirement for irradiated blood products must be noted in the Special Requirement box on the dedicated Blood and Blood Products prescription and Transfusion Record (see Appendix 1). Once the indication for irradiated blood products has been established, the patient should be provided with an appropriate card and encouraged to carry it at all times (see Appendix 2) Policy for the use of Irradiated blood products September
5 2.3 Endorsement The policy will be approved by the Hospital Transfusion Committee Final endorsement will be by the Clinical Governance Committee. 2.4 Distribution The policy will be recorded on SharePoint. Staff will be made aware of the policy during induction and clinical update sessions, and via e Brief. 3. Implementation 3.1 The policy will be reviewed every three years or sooner if required in light of new evidence or statutory requirements. 3.2 The transfusion laboratory will maintain responsibility for ensuring Irradiated products are issued when requested appropriately, referring to laboratory SOP TRA-CC-TRA 001/09. This SOP is reviewed annually. 3.3 An audit of the requirement for Irradiated blood products to be noted in the Special Requirement box on the dedicated Blood and Blood Products prescription and Transfusion Record will be performed to monitor compliance. 3.4 An audit to monitor the issue of Irradiated units to the patients which require them will be performed 6 monthly. 3.5 If there is any deviation from this policy, an incident form will be raised via DATIX, and the incident investigated by the transfusion operational management team and corrective and preventative actions implemented. If appropriate, a report will be made to SHOT and SABRE Glossary of Terms Irradiated Blood Component-Cellular blood component treated with 25 Gray (Gy) Gamma or Xray irradiation to inactivate lymphocytes that could couse graft-versushost disease SOP- Laboratory Standard Operational Procedure DATIX- Electronic Adverse event and near miss reporting form SHOT -Serious Hazards of Transfusion UK wide reporting system for adverse transfusion events and near misses SABRE- Serious Adverse Blood Reactions and Events This system allows reporters to electronically submit reports of serious adverse events or serious adverse reactions directly to the Medicines and Healthcare Products Regulatory Agency (MHRA) References British Committee for Standards in Haematology Blood Transfusion Taskforce Guidelines on gamma irradiation of blood components for the prevention of transfusionassociated graft versus host disease (Ta GvHD) Transfusion Medicine McClelland, DBL (ed) (2007) Handbook of Transfusion Medicine (4th edition). The Stationery Office, London: Available Policy for the use of Irradiated blood products September
6 (Accessed 24 th Feb 2010) National Blood Service (2007) Portfolio of blood components and guidance for their clinical use. Available ortfolio.pdf (Accessed 24 th February 2010) Policy for the use of Irradiated blood products September
7 Appendix 1 Prescription chart Policy for the use of Irradiated blood products September
8 Appendix 2 Irradiated Blood information leaflet and card Policy for the use of Irradiated blood products September
9 Appendix 3 Equality Impact assessment Form Peterborough and Stamford Hospitals NHS Foundation Trust STAGE ONE : Equality Impact Assessment screening form Assessing Functions/Policies for Relevance Blue boxes are to be filled in Yellow boxes - Click the box to select from the drop down list Free text Select from drop down box Name of function / service / strategy / policy / project (activity) to be assessed: Policy for the use of Irradiated blood products Name(s) of those completing this EqIA Screening form: Kaye Bowen Transfusion Coordinator CBU / Department Clinical Services Date 24 Feb 2010 Function/service/strategy/policy/project (activity) aim or purpose: To ensure that irradiated blood products are requested for and transfused to patients who require them. Is this a new or existing activity: Existing activity Policy for the use of Irradiated blood products September
10 What are the intended results of this activity: Reduce risk by ensuring that all patients who require irradiated blood products receive them. How will you measure the outcome of the activity: Audit of patient and laboratory records. Who is intended to benefit from the activity Patients and staff Please identify any internal/external groups who have been consulted regarding this activity: Hospital Transfusion Team. Hospital Transfusion Committee. National Blood and Transplant Service Use the table below to identify whether the activity could/does have a positive impact, a negative impact or no impact at all on either any or all of the equality groups specified. Age *Disability Ethnicity / Race Gender Religion / Belief Sexual Orientation Eliminating unlawful/unjustifiable discrimination Promoting equality of opportunity Promoting positive attitudes and good community relations between and towards differing equality groups Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Policy for the use of Irradiated blood products September
11 Eliminating harassment or victimization Encourage involvement and participation Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Neutral Eliminating health inequalities Neutral Neutral Neutral Neutral Neutral Neutral If the answer to any of the above is Positive or Negative then please complete the Stage Two Full Equality Impact Assessment form to avoid or address the potential adverse impact. Decision to proceed (please select): No, we have decided that it is not necessary to carryout a full EqIA Reason for decision There are no aspects of the policy which could discriminate against any group Executive Director/General Manager - I confirm that I have been briefed on the results of this impact assessment. Name Rob Heywood Date Signature Please note the following: It is an essential that this EqIA screening form is discussed by your management team and remains readily available for inspection. A copy should also be forwarded to the Communications team for publication on the Trusts internet site. Policy for the use of Irradiated blood products September
12 Appendix 4 Compliance Monitoring Process to be monitored How will compliance with the outlined process be monitored? Frequency By who? If compliance gaps have been identified, who is responsible for creating an action plan, and ensuring implementation of required changes? Completion of trust prescription chart indicating need for irradiated components Audit of prescription charts Monthly Clinical Audit department Transfusion Operational Management Team Requesting of irradiated negative products Audit of patient and Laboratory records 6 monthly Transfusion Laboratory staff and Transfusion Coordinator Transfusion Operational Management Team Issue of irradiated negative products Audit of patient and Laboratory records 6 monthly Transfusion Laboratory staff and Transfusion Coordinator Transfusion Operational Management Team Deviation from policy resulting in adverse event or near miss Review of DATIX incident forms Within 1 week of incident report Transfusion Operational Management Team Transfusion Operational Management Team Policy for the use of Irradiated blood products September
13 Appendix 5 Summary and Audit Trail Development process Title: Policy for the use of Irradiated blood products Trustwide & Healthcare Economy Multidisciplin ary Trustwide CBU Dept Medical Staff X Allied Health Professionals Nursing/ Midwifery X Reason for Development: (e.g. planned review of existing document, patient complaint, critical incident, publication of new evidence, inconsistent practice, NICE Guidance) To ensure awareness of importance of requesting blood components of correct specification Development Lead: Dr Kanchan Rege Consultant Haematologist Tel. Number: 4167 Development Team Members: Martin Drury - Transfusion Laboratory Manager Kaye Bowen- Transfusion Coordinator Address: Kanchan.Rege@pbhtr.nhs.uk Key sources of evidence used in the development of the document or the method of achieving consensus where evidence is not available: National Blood Service Portfolio of Components and Guidance for their Clinical Use British Committee For Standards in Haematology guidelines on gamma irradiation of blood components for the prevention of transfusion associated graft-versus-host disease. Consultation Process Please list key Staff Members and Groups/Committees involved in the Consultation Process: Hospital transfusion Team Please identify committee(s) which will approve the policy (see flow chart for development) : Hospital Transfusion committee Once this form has been completed it should be sent to the Compliance Officer with the final copy of the policy Policy for the use of Irradiated blood products September
Policy for the use of Cytomegalovirus (CMV) negative blood products
 Policy for the use of Cytomegalovirus (CMV) negative blood products SharePoint Location Clinical Policies and Guidelines SharePoint Index Directory Cancer and Specialist Care Sub Area Haematology and Blood
Policy for the use of Cytomegalovirus (CMV) negative blood products SharePoint Location Clinical Policies and Guidelines SharePoint Index Directory Cancer and Specialist Care Sub Area Haematology and Blood
CAUTION: Refer to the Document Library for the most recent version of this policy. Policy for the use of Cytomegalovirus (CMV) negative blood products
 Directorate Department Year Version Number Central Index Number Endorsing Committee Date Endorsed Approval Committee Date Approved Author Name and Job Title Key Words (for search purposes) Date Published
Directorate Department Year Version Number Central Index Number Endorsing Committee Date Endorsed Approval Committee Date Approved Author Name and Job Title Key Words (for search purposes) Date Published
Guidelines for Gamma Irradiation of Blood Components
 AUSTRALIAN & NEW ZEALAND SOCIETY OF BLOOD TRANSFUSION INC. AUSTRALIAN RED CROSS BLOOD SERVICE NEW ZEALAND BLOOD SERVICE Guidelines for Gamma Irradiation of Blood Components Revised 2003 AN ZS B T Australian
AUSTRALIAN & NEW ZEALAND SOCIETY OF BLOOD TRANSFUSION INC. AUSTRALIAN RED CROSS BLOOD SERVICE NEW ZEALAND BLOOD SERVICE Guidelines for Gamma Irradiation of Blood Components Revised 2003 AN ZS B T Australian
Human Albumin Solution Infusion (HAS) - Guideline for practice
 Human Albumin (HAS) - Guideline for practice SharePoint Location Clinical Policies and Guidelines SharePoint Index Directory General Policies and Guidelines Sub Area Haematology and Blood Transfusion Key
Human Albumin (HAS) - Guideline for practice SharePoint Location Clinical Policies and Guidelines SharePoint Index Directory General Policies and Guidelines Sub Area Haematology and Blood Transfusion Key
GUIDELINES FOR IRRADIATED BLOOD COMPONENTS
 GUIDELINES FOR IRRADIATED BLOOD COMPONENTS 1. Policy Statement 2. Definitions 1.1 Blood transfusion services shall have a policy which indicates recipients who are to receive irradiated blood components.
GUIDELINES FOR IRRADIATED BLOOD COMPONENTS 1. Policy Statement 2. Definitions 1.1 Blood transfusion services shall have a policy which indicates recipients who are to receive irradiated blood components.
Specific Requirements
 Specific Requirements AIMS Specific requirements your patients have for transfusion and how this is managed Classify which patients require: Irradiated components CMV negative components Washed components
Specific Requirements AIMS Specific requirements your patients have for transfusion and how this is managed Classify which patients require: Irradiated components CMV negative components Washed components
Transfusion-Associated Graft-versus-Host Disease TRANSFUSION-ASSOCIATED GRAFT-VERSUS-HOST DISEASE
 TRANSFUSION-ASSOCIATED GRAFT-VERSUS-HOST DISEASE D H Pamphilon for the NBS Transfusion Medicine Clinical Policies Group. Membership: M F Murphy (Chair), A Copplestone, M Gesinde, S MacLennan, C Morgan,
TRANSFUSION-ASSOCIATED GRAFT-VERSUS-HOST DISEASE D H Pamphilon for the NBS Transfusion Medicine Clinical Policies Group. Membership: M F Murphy (Chair), A Copplestone, M Gesinde, S MacLennan, C Morgan,
CAUTION: Refer to the Document Library for the most recent version of this document. Cryoprecipitate Transfusion Guideline for Practice.
 Directorate Department Year Version Number Central Index Number Endorsing Committee Date Endorsed Approval Committee Date Approved Author Name and Job Title Key Words (for search purposes) Date Published
Directorate Department Year Version Number Central Index Number Endorsing Committee Date Endorsed Approval Committee Date Approved Author Name and Job Title Key Words (for search purposes) Date Published
with special requirements Contributes to: CQC Regulation 12 Consulted With Individual/Body Date Lead
 Special Requirements for Blood Transfusion Policy Register No: 16015 Status: Public Developed in response to: NHSBT & SaBTO guidance for patients with special requirements Contributes to: CQC Regulation
Special Requirements for Blood Transfusion Policy Register No: 16015 Status: Public Developed in response to: NHSBT & SaBTO guidance for patients with special requirements Contributes to: CQC Regulation
CAUTION: Refer to the Document Library for the most recent version of this document. Platelet Transfusion Guideline for practice
 Platelet Transfusion Guideline for practice Directorate SharePoint Index Directory Year Version Number Central Index Number Endorsing Committee Date Endorsed Approval Committee Date Approved Author Name
Platelet Transfusion Guideline for practice Directorate SharePoint Index Directory Year Version Number Central Index Number Endorsing Committee Date Endorsed Approval Committee Date Approved Author Name
Carol Cantwell Blood Transfusion Laboratory Manager St Mary s Hospital, ICHNT
 Carol Cantwell Blood Transfusion Laboratory Manager St Mary s Hospital, ICHNT History Why is blood transfusion involved? What tests are performed in blood transfusion and why? What does a protocol look
Carol Cantwell Blood Transfusion Laboratory Manager St Mary s Hospital, ICHNT History Why is blood transfusion involved? What tests are performed in blood transfusion and why? What does a protocol look
Developed By Name Signature Date
 Patient Group Direction 2155 version 2.0 Administration / Supply of Inhaled Salbutamol in Asthma by Registered Practitioners employed by Torbay and South Devon NHS Foundation Trust Date of Introduction:
Patient Group Direction 2155 version 2.0 Administration / Supply of Inhaled Salbutamol in Asthma by Registered Practitioners employed by Torbay and South Devon NHS Foundation Trust Date of Introduction:
Specialised Services Policy: CP91 Extracorporeal Photophoresis (ECP) for the Treatment of Chronic Graft versus Host Disease in Adults
 Specialised Services Policy: Extracorporeal Photophoresis (ECP) for the Treatment of Chronic Graft versus Host Disease in Adults Document Author: Specialised Planner Executive Lead: Director of Planning
Specialised Services Policy: Extracorporeal Photophoresis (ECP) for the Treatment of Chronic Graft versus Host Disease in Adults Document Author: Specialised Planner Executive Lead: Director of Planning
All institutions that transfuse blood components and products should implement national and local policies and written procedures for:
 5.0 GENERAL GUIDE TO GOOD TRANSFUSION PRACTICE Blood and the various components prepared or manufactured from it are biologic (in the case of blood cells, living human tissues) products intended for use
5.0 GENERAL GUIDE TO GOOD TRANSFUSION PRACTICE Blood and the various components prepared or manufactured from it are biologic (in the case of blood cells, living human tissues) products intended for use
Blood Product Modifications: Leukofiltration, Irradiation and Washing
 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs
1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs
SALBUTAMOL INHALER PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline
 SALBUTAMOL INHALER PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline 1.1. This Patient Group Direction (PGD) applies to all nursing and clinical staff in the Child Health Department
SALBUTAMOL INHALER PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline 1.1. This Patient Group Direction (PGD) applies to all nursing and clinical staff in the Child Health Department
28 th September Author Jeremy Gilbert Bariatric Nurse Specialist
 POLICY FOR SELF ADMINISTRATION OF CONTINUOUS POSITIVE AIRWAY PRESSURE BY COMPETENT PATIENTS COMING IN FOR METABOLIC AND OBESITY SURGERY (BARIATRIC SURGERY) TO PENDENNIS WARD 28 th September 2014 Author
POLICY FOR SELF ADMINISTRATION OF CONTINUOUS POSITIVE AIRWAY PRESSURE BY COMPETENT PATIENTS COMING IN FOR METABOLIC AND OBESITY SURGERY (BARIATRIC SURGERY) TO PENDENNIS WARD 28 th September 2014 Author
Developed By Name Signature Date
 Patient Group Direction 2156 version 2.0 Administration of Ipratropium 250mcg/ml Nebuliser Solution in Acute Asthma by Registered Practitioners employed by Torbay and South Devon NHS Foundation Date of
Patient Group Direction 2156 version 2.0 Administration of Ipratropium 250mcg/ml Nebuliser Solution in Acute Asthma by Registered Practitioners employed by Torbay and South Devon NHS Foundation Date of
Special Requirements Lab Matters. 18 th May Katy Cowan PBM Practitioner NHSBT
 Special Requirements Lab Matters. 18 th May 2016 Katy Cowan PBM Practitioner NHSBT Special requirements Frozen components and platelets HEV negative Irradiated CMV negative Frozen components and platelets
Special Requirements Lab Matters. 18 th May 2016 Katy Cowan PBM Practitioner NHSBT Special requirements Frozen components and platelets HEV negative Irradiated CMV negative Frozen components and platelets
Guide to the preparation, use and quality assurance of blood components
 Contents Foreword...3 Members of the European Committee (Partial Agreement) on Blood Transfusion... 8 Members of the GTS working group... 22 Members of the TS066 working group... 30 Recommendation No.
Contents Foreword...3 Members of the European Committee (Partial Agreement) on Blood Transfusion... 8 Members of the GTS working group... 22 Members of the TS066 working group... 30 Recommendation No.
Guidelines for the management of warfarin reversal in adults
 SharePoint Location Clinical Policies and Guidelines SharePoint Index Directory General Policies and Guidelines Sub Area Haematology and Transfusion Key words (for search purposes) Warfarin, Bleeding Central
SharePoint Location Clinical Policies and Guidelines SharePoint Index Directory General Policies and Guidelines Sub Area Haematology and Transfusion Key words (for search purposes) Warfarin, Bleeding Central
BLOOD TRANSFUSION. Dr Lumka Ntabeni
 BLOOD TRANSFUSION Dr Lumka Ntabeni Blood transfusion definition SAFE transfer of BLOOD COMPONENTS from a DONOR to a RECEPIENT CONTENT Brief history of blood transfusion How is safety guaranteed? How do
BLOOD TRANSFUSION Dr Lumka Ntabeni Blood transfusion definition SAFE transfer of BLOOD COMPONENTS from a DONOR to a RECEPIENT CONTENT Brief history of blood transfusion How is safety guaranteed? How do
PALIPERIDONE LONG ACTING INJECTION PRESCRIBING GUIDELINE. Chief Pharmacist. Chief Pharmacist
 REFERENCE NUMBER: PALIPERIDONE LONG ACTING INJECTION PRESCRIBING GUIDELINE AREA: NAME OF RESPONSIBLE COMMITTEE / INDIVIDUAL NAME OF ORIGINATOR / AUTHOR Trust-wide Chief Pharmacist Chief Pharmacist DATE
REFERENCE NUMBER: PALIPERIDONE LONG ACTING INJECTION PRESCRIBING GUIDELINE AREA: NAME OF RESPONSIBLE COMMITTEE / INDIVIDUAL NAME OF ORIGINATOR / AUTHOR Trust-wide Chief Pharmacist Chief Pharmacist DATE
A Review of Guidelines and Evidence for the Use of Irradiated Blood Products in Solid Tumor, Chemotherapy Patients. Chris Kim 11/29/12
 A Review of Guidelines and Evidence for the Use of Irradiated Blood Products in Solid Tumor, Chemotherapy Patients Chris Kim 11/29/12 Background Prevention of TAGVHD Irradiation: induces DNA crosslinks,
A Review of Guidelines and Evidence for the Use of Irradiated Blood Products in Solid Tumor, Chemotherapy Patients Chris Kim 11/29/12 Background Prevention of TAGVHD Irradiation: induces DNA crosslinks,
Special Requirements Lab Matters. 21 st June Barrie Ferguson
 Special Requirements Lab Matters. 21 st June 2017 Barrie Ferguson Special requirements Patients requiring Methylene Blue or Solvent Detergent treated Fresh Frozen Plasma Patients requiring apheresis platelets
Special Requirements Lab Matters. 21 st June 2017 Barrie Ferguson Special requirements Patients requiring Methylene Blue or Solvent Detergent treated Fresh Frozen Plasma Patients requiring apheresis platelets
25/10/2017. Clinical Relevance of the HLA System in Blood Transfusion. Outline of talk. Major Histocompatibility Complex
 Clinical Relevance of the HLA System in Blood Transfusion Dr Colin J Brown PhD FRCPath. October 2017 Outline of talk HLA genes, structure and function HLA and immune complications of transfusion TA-GVHD
Clinical Relevance of the HLA System in Blood Transfusion Dr Colin J Brown PhD FRCPath. October 2017 Outline of talk HLA genes, structure and function HLA and immune complications of transfusion TA-GVHD
Non-Medical Authorisation Course
 Non-Medical Authorisation Course November 2017 Authorising, Prescribing & Sampling Janice Smith Matron Transfusion Specialist Aims of this session To discuss and understand: Authorising blood / blood components
Non-Medical Authorisation Course November 2017 Authorising, Prescribing & Sampling Janice Smith Matron Transfusion Specialist Aims of this session To discuss and understand: Authorising blood / blood components
Therapeutic Apheresis Services Annual Review 2016/17
 Therapeutic Apheresis Services Annual Review 2016/17 NHS Blood and Transplant (NHSBT) is a major provider of Therapeutic Apheresis Services (TAS) in the NHS. Services are provided across England. Collection
Therapeutic Apheresis Services Annual Review 2016/17 NHS Blood and Transplant (NHSBT) is a major provider of Therapeutic Apheresis Services (TAS) in the NHS. Services are provided across England. Collection
CTYOMEGALOVIRUS (CMV) - BACKGROUND
 CTYOMEGALOVIRUS (CMV) - BACKGROUND PURPOSE The flowing information provides guidance on the use of CMV negative blood components provided by the blood bank at the Royal Children s Hospital (RCH) including
CTYOMEGALOVIRUS (CMV) - BACKGROUND PURPOSE The flowing information provides guidance on the use of CMV negative blood components provided by the blood bank at the Royal Children s Hospital (RCH) including
Blood Components & Indications for Transfusion. Neda Kalhor
 Blood Components & Indications for Transfusion Neda Kalhor Blood products Cellular Components: Red blood cells - Leukocyte-reduced RBCs - Washed RBCs - Irradiated RBCs Platelets - Random-donor platelets
Blood Components & Indications for Transfusion Neda Kalhor Blood products Cellular Components: Red blood cells - Leukocyte-reduced RBCs - Washed RBCs - Irradiated RBCs Platelets - Random-donor platelets
Blood is serious business
 Transfusion at RCH BLOOD TRANSFUSION Anthea Greenway Dept of Clinical Haematology >10000 fresh blood products per year Supports craniofacial and cardiac surgery Support bone marrow, liver transplant and
Transfusion at RCH BLOOD TRANSFUSION Anthea Greenway Dept of Clinical Haematology >10000 fresh blood products per year Supports craniofacial and cardiac surgery Support bone marrow, liver transplant and
CLINICAL GUIDELINE FOR THE ADMINISTRATION OF MESNA WITH IFOSFAMIDE AND CYCLOPHOSPHAMIDE Summary.
 CLINICAL GUIDELINE FOR THE ADMINISTRATION OF MESNA WITH IFOSFAMIDE AND CYCLOPHOSPHAMIDE Summary. Yes Is patient prescribed ifosfamide or cyclophosphamide >1g/m 2? Chemotherapy prescription on Aria should
CLINICAL GUIDELINE FOR THE ADMINISTRATION OF MESNA WITH IFOSFAMIDE AND CYCLOPHOSPHAMIDE Summary. Yes Is patient prescribed ifosfamide or cyclophosphamide >1g/m 2? Chemotherapy prescription on Aria should
Disclosures. Committee on Blood and Blood Products) Co-investigator CBS Small Project. in Solid Organ Transplant Recipients
 Neonatal Transfusion: Irradiation and CMV 6 th Annual Blood Matters Conference November 6 th, 2015 Disclosures Member of NAC (National Advisory Committee on Blood and Blood Products) Co-investigator CBS
Neonatal Transfusion: Irradiation and CMV 6 th Annual Blood Matters Conference November 6 th, 2015 Disclosures Member of NAC (National Advisory Committee on Blood and Blood Products) Co-investigator CBS
Transfusion-associated graft-versus-host disease
 CHAPTER 6 Transfusion-associated graft-versus-host disease Eric A. Gehrie, 1 Edward L. Snyder, 1 & Alex B. Ryder 2 1 Department of Laboratory Medicine, Yale School of Medicine, Yale University; and Blood
CHAPTER 6 Transfusion-associated graft-versus-host disease Eric A. Gehrie, 1 Edward L. Snyder, 1 & Alex B. Ryder 2 1 Department of Laboratory Medicine, Yale School of Medicine, Yale University; and Blood
Trust Policy 218 Ionising Radiation Safety Policy
 Trust Policy 218 Ionising Radiation Safety Policy Purpose Date Version August 2016 7 To ensure that Plymouth Hospitals NHS Trust complies with all relevant legislation with regard to the use of ionising
Trust Policy 218 Ionising Radiation Safety Policy Purpose Date Version August 2016 7 To ensure that Plymouth Hospitals NHS Trust complies with all relevant legislation with regard to the use of ionising
Blood Transfusion Guidelines in Clinical Practice
 Blood Transfusion Guidelines in Clinical Practice Salwa Hindawi Director of Blood Transfusion Services Associate Professor in Haematology and Transfusion Medicine King Abdalaziz University, Jeddah Saudi
Blood Transfusion Guidelines in Clinical Practice Salwa Hindawi Director of Blood Transfusion Services Associate Professor in Haematology and Transfusion Medicine King Abdalaziz University, Jeddah Saudi
Trust Policy. Control of Smoking Policy (Patients and Visitors)
 Trust Policy Control of Smoking Policy (Patients and Visitors) Purpose Date May 2016 Version V3 Final To discharge the Trust s responsibilities under the Health Act 2006, and associated NICE Guidelines
Trust Policy Control of Smoking Policy (Patients and Visitors) Purpose Date May 2016 Version V3 Final To discharge the Trust s responsibilities under the Health Act 2006, and associated NICE Guidelines
RECOMMENDATIONS FOR USE OF IRRADIATED BLOOD COMPONENTS IN CANADA
 RECOMMENDATIONS FOR USE OF IRRADIATED BLOOD COMPONENTS IN CANADA 2016-09-26 Page 1 RECOMMENDATIONS FOR USE OF IRRADIATED BLOOD COMPONENTS IN CANADA BACKGROUND: The National Advisory Committee on Blood
RECOMMENDATIONS FOR USE OF IRRADIATED BLOOD COMPONENTS IN CANADA 2016-09-26 Page 1 RECOMMENDATIONS FOR USE OF IRRADIATED BLOOD COMPONENTS IN CANADA BACKGROUND: The National Advisory Committee on Blood
Patient Group Directions Policy
 Patient Group Directions Policy Category: Summary: Equality Analysis undertaken: Valid From: Date of Next Review: Approval Date/ Via: Distribution: Related Documents: Author(s): Further Information: This
Patient Group Directions Policy Category: Summary: Equality Analysis undertaken: Valid From: Date of Next Review: Approval Date/ Via: Distribution: Related Documents: Author(s): Further Information: This
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey
 Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
FOR CHILDREN ATTENDING FOR EXODONTIA UNDER GENERAL ANAESTHETIC
 CLINICAL PROTOCOL FOR CHILDREN ATTENDING FOR EXODONTIA UNDER GENERAL ANAESTHETIC CLINICAL RECORDS AND PATIENT INFORMATION RATIONALE The purpose of this clinical protocol is to ensure that the clinical
CLINICAL PROTOCOL FOR CHILDREN ATTENDING FOR EXODONTIA UNDER GENERAL ANAESTHETIC CLINICAL RECORDS AND PATIENT INFORMATION RATIONALE The purpose of this clinical protocol is to ensure that the clinical
Blood Glucose and Hyperglycaemia Management in Hospital for Adults with Diabetes Clinical Guideline V2.0. March 2018
 Blood Glucose and Hyperglycaemia Management in Hospital for Adults with Diabetes Clinical Guideline V2.0 March 2018 Page 1 of 8 Summary flow chart for monitoring of blood glucose if >11mmol/L For Adults
Blood Glucose and Hyperglycaemia Management in Hospital for Adults with Diabetes Clinical Guideline V2.0 March 2018 Page 1 of 8 Summary flow chart for monitoring of blood glucose if >11mmol/L For Adults
Chemotherapy Training and Assessment Policy. For Medical Prescribers and Pharmacy Verifiers
 Chemotherapy Training and Assessment Policy For Medical Prescribers and Pharmacy Verifiers For approvals and version control see Document Management Record on page 6 Doc Ref: AngCN-CCG-C36 Approved and
Chemotherapy Training and Assessment Policy For Medical Prescribers and Pharmacy Verifiers For approvals and version control see Document Management Record on page 6 Doc Ref: AngCN-CCG-C36 Approved and
GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS
 CHILDREN S HOSPITALS AND CLINICS OF MINNESOTA Introduction: GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS These guidelines have been developed in conjunction with the hospital Transfusion Committee.
CHILDREN S HOSPITALS AND CLINICS OF MINNESOTA Introduction: GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS These guidelines have been developed in conjunction with the hospital Transfusion Committee.
Acutely Painful testes
 2.0 FINAL Guideline adopted from the Bedside Clinical Guideline Partnership EQUALITY IMPACT The Trust strives to ensure equality of opportunity for all both as a major employer and as a provider of health
2.0 FINAL Guideline adopted from the Bedside Clinical Guideline Partnership EQUALITY IMPACT The Trust strives to ensure equality of opportunity for all both as a major employer and as a provider of health
COMPETENT AUTHORITY (UK) MEDICAL DEVICES DIRECTIVES GUIDANCE NOTES FOR MANUFACTURERS OF DENTAL APPLIANCES
 COMPETENT AUTHORITY (UK) 10 EC MEDICAL DEVICES DIRECTIVES GUIDANCE NOTES FOR MANUFACTURERS OF DENTAL APPLIANCES (CUSTOM MADE DEVICES) Updated March 2008 CONTENTS PAGE Introduction 3 Definition of dental
COMPETENT AUTHORITY (UK) 10 EC MEDICAL DEVICES DIRECTIVES GUIDANCE NOTES FOR MANUFACTURERS OF DENTAL APPLIANCES (CUSTOM MADE DEVICES) Updated March 2008 CONTENTS PAGE Introduction 3 Definition of dental
LifeBridge Health Transfusion Service Sinai Hospital of Baltimore Northwest Hospital Center BQA Transfusion Criteria Version#2 POLICY NO.
 LifeBridge Health Transfusion Service Sinai Hospital of Baltimore Northwest Hospital Center BQA 1011.02 Transfusion Criteria Version#2 Department POLICY NO. PAGE NO. Blood Bank Quality Assurance Manual
LifeBridge Health Transfusion Service Sinai Hospital of Baltimore Northwest Hospital Center BQA 1011.02 Transfusion Criteria Version#2 Department POLICY NO. PAGE NO. Blood Bank Quality Assurance Manual
Clinical Relevance of the HLA System in Blood Transfusion. Dr Colin J Brown PhD FRCPath. October 2017
 Clinical Relevance of the HLA System in Blood Transfusion Dr Colin J Brown PhD FRCPath. October 2017 Outline of talk HLA genes, structure and function HLA and immune complications of transfusion TA-GVHD
Clinical Relevance of the HLA System in Blood Transfusion Dr Colin J Brown PhD FRCPath. October 2017 Outline of talk HLA genes, structure and function HLA and immune complications of transfusion TA-GVHD
Autologous plasma eye drops. Patient information
 Autologous plasma eye drops Patient information Autologous plasma eye drops are made from the patient s own blood and are used to help treat their corneal surface defects and diseases. This leaflet gives
Autologous plasma eye drops Patient information Autologous plasma eye drops are made from the patient s own blood and are used to help treat their corneal surface defects and diseases. This leaflet gives
GAMETE STORAGE. Reviewing body Review date Version no
 GAMETE STORAGE Document History Date of publication July 2016 Version Number: V1 Review date July 2018 Consultation Consultee Which group Public health INPUT Derbyshire Affiliated Commissioning Committee
GAMETE STORAGE Document History Date of publication July 2016 Version Number: V1 Review date July 2018 Consultation Consultee Which group Public health INPUT Derbyshire Affiliated Commissioning Committee
Guidelines for the Management of Chronic Lymphocytic Leukaemia (CLL)
 Guidelines for the Management of Chronic Lymphocytic Leukaemia (CLL) Version History Version Date Summary of Change/Process 2.0 08.05.08 Endorsed by the Governance Committee 2.1 16.02.11 Circulated at
Guidelines for the Management of Chronic Lymphocytic Leukaemia (CLL) Version History Version Date Summary of Change/Process 2.0 08.05.08 Endorsed by the Governance Committee 2.1 16.02.11 Circulated at
Transplants. Mickey B. C. Koh
 Transfusion in Stem Cell Transplants Mickey B. C. Koh Director: Stem Cell Transplant Programme Department of Haematology, St. George s Hospital and Medical School, London, UK Medical Director: Cell Therapy
Transfusion in Stem Cell Transplants Mickey B. C. Koh Director: Stem Cell Transplant Programme Department of Haematology, St. George s Hospital and Medical School, London, UK Medical Director: Cell Therapy
No Smoking Policy. No Smoking Policy
 No Smoking Policy Document Status Version: V4.0 Approved DOCUMENT CHANGE HISTORY Initiated by Date Author HR Version Date Comments (i.e. viewed, or reviewed, amended approved by person or committee) V1.1
No Smoking Policy Document Status Version: V4.0 Approved DOCUMENT CHANGE HISTORY Initiated by Date Author HR Version Date Comments (i.e. viewed, or reviewed, amended approved by person or committee) V1.1
Clinical Guideline for Intravenous Opioids for Adults in Recovery Areas The Recovery Protocol
 Clinical Guideline for Intravenous Opioids for Adults in Recovery Areas The Recovery Protocol 1. Aim/Purpose of this Guideline 1.1. To Provide safe and efficient administration of Opioids in Recovery.
Clinical Guideline for Intravenous Opioids for Adults in Recovery Areas The Recovery Protocol 1. Aim/Purpose of this Guideline 1.1. To Provide safe and efficient administration of Opioids in Recovery.
Policy and Procedure for the Development, Approval and Implementation of Patient Group Directions in NHS Haringey Clinical Commissioning Group
 BEFORE USING THIS POLICY ALWAYS ENSURE YOU ARE USING THE MOST UP TO DATE VERSION Policy and Procedure for the Development, Approval and Implementation of Patient Group Directions in NHS Haringey Clinical
BEFORE USING THIS POLICY ALWAYS ENSURE YOU ARE USING THE MOST UP TO DATE VERSION Policy and Procedure for the Development, Approval and Implementation of Patient Group Directions in NHS Haringey Clinical
Apheresis: Transfusion Indications. Sasha Wilson: Transfusion Senior Nurse
 Apheresis: Transfusion Indications Sasha Wilson: Transfusion Senior Nurse Apheresis: Transfusion Indications Focus of talk will be blood component transfusion in the context of apheresis procedures: Special
Apheresis: Transfusion Indications Sasha Wilson: Transfusion Senior Nurse Apheresis: Transfusion Indications Focus of talk will be blood component transfusion in the context of apheresis procedures: Special
Corporate Medical Policy
 Corporate Medical Policy Hematopoietic Stem-Cell Transplantation for Waldenstrom Macroglobulinemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: hematopoietic_stem_cell_transplantation_for_waldenstrom_macroglobulinemia
Corporate Medical Policy Hematopoietic Stem-Cell Transplantation for Waldenstrom Macroglobulinemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: hematopoietic_stem_cell_transplantation_for_waldenstrom_macroglobulinemia
Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital
 Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
Will I need a blood transfusion? Patient information
 Will I need a blood transfusion? Patient information Will I need a blood transfusion? Important information for all patients who may need a blood transfusion Like all medical treatments, a blood transfusion
Will I need a blood transfusion? Patient information Will I need a blood transfusion? Important information for all patients who may need a blood transfusion Like all medical treatments, a blood transfusion
PROCEDURE REFERENCE NUMBER SABP/EXECUTIVE BOARD/0017/PROCEDURE15 PROCEDURE NAME MEDICINES PROCEDURE 15: METHOTREXATE
 PROCEDURE REFERENCE NUMBER SABP/EXECUTIVE BOARD/0017/PROCEDURE15 PROCEDURE NAME MEDICINES PROCEDURE 15: METHOTREXATE BRIEF OUTLINE OF THIS PROCEDURE This procedure sets out the requirements for prescribing,
PROCEDURE REFERENCE NUMBER SABP/EXECUTIVE BOARD/0017/PROCEDURE15 PROCEDURE NAME MEDICINES PROCEDURE 15: METHOTREXATE BRIEF OUTLINE OF THIS PROCEDURE This procedure sets out the requirements for prescribing,
GOVERNING BODY. Kingston Assisted Conception Guidelines
 GOVERNING BODY LEAD: Dr Naeem Iqbal REPORT AUTHOR: Niran Rehill & Livia Royle, Public Health Royal Borough of Kingston / Kingston CCG ATTACHMENT: AGENDA ITEM: 8 H RECOMMENDATION: The Governing Body is
GOVERNING BODY LEAD: Dr Naeem Iqbal REPORT AUTHOR: Niran Rehill & Livia Royle, Public Health Royal Borough of Kingston / Kingston CCG ATTACHMENT: AGENDA ITEM: 8 H RECOMMENDATION: The Governing Body is
Standard Operating Procedure CPFT/SOP003 Serious Breach of Protocol or GCP in CTIMPs
 Standard Operating Procedure CPFT/SOP003 Serious Breach of Protocol or GCP in CTIMPs Author: Sponsor/Executive: Responsible committee: Ratified by: Consultation & Approval: (Committee/Groups which signed
Standard Operating Procedure CPFT/SOP003 Serious Breach of Protocol or GCP in CTIMPs Author: Sponsor/Executive: Responsible committee: Ratified by: Consultation & Approval: (Committee/Groups which signed
HAEMATOPOIETIC STEM CELL TRANSPLANTATION
 PRIMARY IMMUNODEFICIENCIES HAEMATOPOIETIC STEM CELL TRANSPLANTATION HAEMATOPOIETIC STEM CELL TRANSPLANTATION 1 PRIMARY IMMUNODEFICIENCIES KEY ABBREVIATIONS CID GvHD HSCT IPOPI PID SCID BMT HSC Combined
PRIMARY IMMUNODEFICIENCIES HAEMATOPOIETIC STEM CELL TRANSPLANTATION HAEMATOPOIETIC STEM CELL TRANSPLANTATION 1 PRIMARY IMMUNODEFICIENCIES KEY ABBREVIATIONS CID GvHD HSCT IPOPI PID SCID BMT HSC Combined
Better Blood Transfusion Level 2 BLOOD COMPONENT USE. Self-Directed Learning Pack
 Better Blood Transfusion Level 2 BLOOD COMPONENT USE Self-Directed Learning Pack Effective Use of Blood Group Scottish National Blood Transfusion Service 2005 Disclaimer Whilst the information and advice
Better Blood Transfusion Level 2 BLOOD COMPONENT USE Self-Directed Learning Pack Effective Use of Blood Group Scottish National Blood Transfusion Service 2005 Disclaimer Whilst the information and advice
North of England Cancer Network. Policies and Procedures. Standards for the Safe Use of Oral Anticancer Medicines
 \ North of England Cancer Network Policies and Procedures Standards for the Safe Use of Oral Anticancer Medicines NECN Oral Anticancer medicine Policy version 1.6 Page 1 of 17 Issue Date: Feb 2017 Contents
\ North of England Cancer Network Policies and Procedures Standards for the Safe Use of Oral Anticancer Medicines NECN Oral Anticancer medicine Policy version 1.6 Page 1 of 17 Issue Date: Feb 2017 Contents
Fresh Frozen Plasma (FFP) and Cryoprecipitate Patient information
 Fresh Frozen Plasma (FFP) and Cryoprecipitate Patient information What are Fresh Frozen Plasma (FFP) and Cryoprecipitate? FFP and Cryoprecipitate (often just called cryo ) are both blood components made
Fresh Frozen Plasma (FFP) and Cryoprecipitate Patient information What are Fresh Frozen Plasma (FFP) and Cryoprecipitate? FFP and Cryoprecipitate (often just called cryo ) are both blood components made
West Midlands Sarcoma Advisory Group
 West Midlands Sarcoma Advisory Group Guideline for the Initial Investigation and Referral to Specialist Sarcoma Multi Disciplinary Team for Suspected Bone Sarcoma Version History Version Date Brief Summary
West Midlands Sarcoma Advisory Group Guideline for the Initial Investigation and Referral to Specialist Sarcoma Multi Disciplinary Team for Suspected Bone Sarcoma Version History Version Date Brief Summary
Yorkshire and Humber Children and Young People s Cancer Network
 Humber and Yorkshire Coast Cancer Network Yorkshire Cancer Network Yorkshire and Humber Children and Young People s Cancer Network Guideline for Blood Component Support in Paediatric and Adolescent Oncology
Humber and Yorkshire Coast Cancer Network Yorkshire Cancer Network Yorkshire and Humber Children and Young People s Cancer Network Guideline for Blood Component Support in Paediatric and Adolescent Oncology
Blood transfusion. Dr. J. Potgieter Dept. of Haematology NHLS - TAD
 Blood transfusion Dr. J. Potgieter Dept. of Haematology NHLS - TAD General Blood is collected from volunteer donors >90% is separated into individual components and plasma Donors should be: healthy, have
Blood transfusion Dr. J. Potgieter Dept. of Haematology NHLS - TAD General Blood is collected from volunteer donors >90% is separated into individual components and plasma Donors should be: healthy, have
IT and Information Acceptable Use Policy
 BMI IMpol04 Information Management IT and Information Acceptable Use Policy This is a controlled document and whilst this document may be printed, the electronic version posted on the intranet/shared drive
BMI IMpol04 Information Management IT and Information Acceptable Use Policy This is a controlled document and whilst this document may be printed, the electronic version posted on the intranet/shared drive
Clinical Commissioning Policy: Haematopoietic Stem Cell Transplantation. December Reference : NHSCB/B4/a/1
 Clinical Commissioning Policy: Haematopoietic Stem Cell Transplantation December 2012 Reference : NHSCB/B4/a/1 NHS Commissioning Board Clinical Commissioning Policy: Haematopoietic Stem Cell Transplantation
Clinical Commissioning Policy: Haematopoietic Stem Cell Transplantation December 2012 Reference : NHSCB/B4/a/1 NHS Commissioning Board Clinical Commissioning Policy: Haematopoietic Stem Cell Transplantation
Annual Progress and Safety Reporting SOP for HEY-sponsored CTIMPs
 R&D Department Annual Progress and Safety Reporting SOP for HEY-sponsored CTIMPs Hull and East Yorkshire Hospitals NHS Trust 2010 All Rights Reserved No part of this document may be reproduced, stored
R&D Department Annual Progress and Safety Reporting SOP for HEY-sponsored CTIMPs Hull and East Yorkshire Hospitals NHS Trust 2010 All Rights Reserved No part of this document may be reproduced, stored
Clinical Commissioning Policy Proposition: Treatments for Graft versus Host Disease (GvHD) following Haematopoietic Stem Cell Transplantation
 Clinical Commissioning Policy Proposition: Treatments for Graft versus Host Disease (GvHD) following Haematopoietic Stem Cell Transplantation Reference: NHS England F01X08/01 1 Information Reader Box (IRB)
Clinical Commissioning Policy Proposition: Treatments for Graft versus Host Disease (GvHD) following Haematopoietic Stem Cell Transplantation Reference: NHS England F01X08/01 1 Information Reader Box (IRB)
Guidelines for the Management of Platelet Transfusion Refractoriness GUIDELINES FOR THE MANAGEMENT OF PLATELET TRANSFUSION REFRACTORINESS
 GUIDELINES FOR THE MANAGEMENT OF PLATELET TRANSFUSION REFRACTORINESS Reviewed by Dr Colin Brown (26/03/2008) Author(s): Colin Brown Page 1 of 7 Purposes To define and recommend policies and procedures
GUIDELINES FOR THE MANAGEMENT OF PLATELET TRANSFUSION REFRACTORINESS Reviewed by Dr Colin Brown (26/03/2008) Author(s): Colin Brown Page 1 of 7 Purposes To define and recommend policies and procedures
Policy Statement. Title/Topic: Hyaluronic Acid Injections for Osteoarthritis Date: April 2014 Reference: GM037
 Policy Statement Title/Topic: Hyaluronic Acid Injections for Osteoarthritis Date: April 2014 Reference: GM037 VERSION CONTROL Version Date Details Page number 0.1 09/09/2013 Initial draft N/A 0.2 19/09/2013
Policy Statement Title/Topic: Hyaluronic Acid Injections for Osteoarthritis Date: April 2014 Reference: GM037 VERSION CONTROL Version Date Details Page number 0.1 09/09/2013 Initial draft N/A 0.2 19/09/2013
Contributes to CQC Outcome number: 9&12 Consulted With Post/Committee/Group Date. Pharmacist. Alison Felton Head of Pharmacy/Deputy Chief.
 Prescribing paracetamol in adult inpatients at MEHT Type: Clinical Guideline Register No: 18005 Status: Public on ratification Developed in response to: In line with Basildon Contributes to CQC Outcome
Prescribing paracetamol in adult inpatients at MEHT Type: Clinical Guideline Register No: 18005 Status: Public on ratification Developed in response to: In line with Basildon Contributes to CQC Outcome
1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of acamprosate.
 SHARED CARE GUIDELINE FOR ACAMPROSATE 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of
SHARED CARE GUIDELINE FOR ACAMPROSATE 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of
DHQ Flowcharts v2.0 eff. February 2016
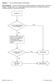 Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
School Hearing Screening Policy
 School Hearing Screening Policy V2.1 1st August 2017 Page 1 of 13 Table of Contents 1. Introduction... 3 2. Purpose of this Policy... 3 3. Scope... 3 4. Definitions / Glossary... 3 5. Ownership and Responsibilities...
School Hearing Screening Policy V2.1 1st August 2017 Page 1 of 13 Table of Contents 1. Introduction... 3 2. Purpose of this Policy... 3 3. Scope... 3 4. Definitions / Glossary... 3 5. Ownership and Responsibilities...
Changing practice to support service delivery
 Nurse and Midwife Medicinal Product Prescribing Toolkit Authorised Medicinal Products, Off-label Prescription and Exempt Medicinal Products Toolkit Changing practice to support service delivery Introduction
Nurse and Midwife Medicinal Product Prescribing Toolkit Authorised Medicinal Products, Off-label Prescription and Exempt Medicinal Products Toolkit Changing practice to support service delivery Introduction
The Practical Application of Statistics: Reading papers and interpreting clinical studies...
 The Practical Application of Statistics: Reading papers and interpreting clinical studies... Richard Szydlo Imperial College / Hammersmith Hospital, London Karlskirche Evaluating new treatments
The Practical Application of Statistics: Reading papers and interpreting clinical studies... Richard Szydlo Imperial College / Hammersmith Hospital, London Karlskirche Evaluating new treatments
CLINICAL GUIDELINE FOR THE ADMINISTRATION OF NEBULISED PENTAMIDINE Summary. 1.
 CLINICAL GUIDELINE FOR THE ADMINISTRATION OF NEBULISED PENTAMIDINE Summary. 1. Patient requires nebulised Pentamidine Ensure equipment listed is available Ensure HEPA filtered room in Haematology Clinic
CLINICAL GUIDELINE FOR THE ADMINISTRATION OF NEBULISED PENTAMIDINE Summary. 1. Patient requires nebulised Pentamidine Ensure equipment listed is available Ensure HEPA filtered room in Haematology Clinic
Clinical Protocol Documentation for Patients Attending for Exodontia under General Anaesthetic Clinical Records and Patient Information
 Clinical Protocol Documentation for Patients Attending for Exodontia under General Anaesthetic Clinical Records and Patient Information CP:92 [Version:1] Applies to:- Specialist Dental Services Sub Committee
Clinical Protocol Documentation for Patients Attending for Exodontia under General Anaesthetic Clinical Records and Patient Information CP:92 [Version:1] Applies to:- Specialist Dental Services Sub Committee
General Terms: Appendix B. National Marrow Donor Program and The Medical College of Wisconsin
 Glossary of Terms This appendix is divided into two sections. The first section, General Terms, defines terms used throughout the CIBMTR data collection forms. The second section, FormsNet TM 2 Terms,
Glossary of Terms This appendix is divided into two sections. The first section, General Terms, defines terms used throughout the CIBMTR data collection forms. The second section, FormsNet TM 2 Terms,
Wessex Paediatric Oncology Supportive Care Guidelines: Transfusion & Coagulation.
 Wessex Paediatric Oncology Supportive Care Guidelines: Transfusion & Coagulation. Scope This guideline applies to all Paediatric Oncology patients in the region. It does not apply to neonates on neonatal
Wessex Paediatric Oncology Supportive Care Guidelines: Transfusion & Coagulation. Scope This guideline applies to all Paediatric Oncology patients in the region. It does not apply to neonates on neonatal
Rob Wynn RMCH & University of Manchester, UK. HCT in Children
 Rob Wynn RMCH & University of Manchester, UK HCT in Children Summary Indications for HCT in children Donor selection for Paediatric HCT Using cords Achieving engraftment in HCT Conditioning Immune action
Rob Wynn RMCH & University of Manchester, UK HCT in Children Summary Indications for HCT in children Donor selection for Paediatric HCT Using cords Achieving engraftment in HCT Conditioning Immune action
CAUTION: You must refer to the intranet for the most recent version of this procedural document.
 Procedure for the use of Intravenous Iron Dextran (CosmoFer ) Sharepoint Location Sharepoint Index Directory Clinical Policies and Guidelines General Policies and Guidelines/ Haematology And blood transfusion
Procedure for the use of Intravenous Iron Dextran (CosmoFer ) Sharepoint Location Sharepoint Index Directory Clinical Policies and Guidelines General Policies and Guidelines/ Haematology And blood transfusion
Preventing CMV Transmission through Leukodepletion
 Preventing CMV Transmission through Leukodepletion Possibility & Facts Prof.S.B.Rajadhyaksha, MD,DTM,PGDMLS Head, Dept. of Transfusion Medicine Tata Memorial Hospital, Mumbai 1 Donor Leukocytes Linked
Preventing CMV Transmission through Leukodepletion Possibility & Facts Prof.S.B.Rajadhyaksha, MD,DTM,PGDMLS Head, Dept. of Transfusion Medicine Tata Memorial Hospital, Mumbai 1 Donor Leukocytes Linked
West Midlands Sarcoma Advisory Group
 West Midlands Sarcoma Advisory Group Guideline for the Initial Investigation and Referral to Sarcoma Specialist Multi Disciplinary Team for Suspected Sarcoma of Soft Tissue Extremities (limbs and trunk
West Midlands Sarcoma Advisory Group Guideline for the Initial Investigation and Referral to Sarcoma Specialist Multi Disciplinary Team for Suspected Sarcoma of Soft Tissue Extremities (limbs and trunk
Smoke Free Policy. Printed copies must not be considered the definitive version. Policy Group. Author Version no 3.0
 Smoke Free Policy Printed copies must not be considered the definitive version Policy Group DOCUMENT CONTROL POLICY NO Smoke Free Grounds Author Version no 3.0 Reviewer Smoke Free Working Group Implementation
Smoke Free Policy Printed copies must not be considered the definitive version Policy Group DOCUMENT CONTROL POLICY NO Smoke Free Grounds Author Version no 3.0 Reviewer Smoke Free Working Group Implementation
making a referral for breast imaging Standard Operating Procedure
 Document Control Title Reporting Radiographer Author Directorate Surgery Date Version Issued 0.1 May 2016 Status Draft Author s job title Reporting Radiographer Department Breast Imaging Comment / Changes
Document Control Title Reporting Radiographer Author Directorate Surgery Date Version Issued 0.1 May 2016 Status Draft Author s job title Reporting Radiographer Department Breast Imaging Comment / Changes
SKIN CARE AND PREVENTION AND MANAGEMENT OF WORK RELATED DERMATITIS
 SKIN CARE AND PREVENTION AND MANAGEMENT OF WORK RELATED DERMATITIS Version No. Date Comments Date Author/s Approved by Amended Approved 1. 10/10/2011 LCP/GC LCP Skin care & prevention of work related dermatitis
SKIN CARE AND PREVENTION AND MANAGEMENT OF WORK RELATED DERMATITIS Version No. Date Comments Date Author/s Approved by Amended Approved 1. 10/10/2011 LCP/GC LCP Skin care & prevention of work related dermatitis
Document Details. Patient Group Direction
 Document Details Title Patient Group Direction (PGD) Hepatitis B Vaccine (Engerix ) Trust Ref No 1505-41182 Local Ref (optional) Main points the Immunisation against Hepatitis B document covers Who is
Document Details Title Patient Group Direction (PGD) Hepatitis B Vaccine (Engerix ) Trust Ref No 1505-41182 Local Ref (optional) Main points the Immunisation against Hepatitis B document covers Who is
Great Ormond Street Hospital for Children. Patient Transfer. Penny Eyton-Jones TLM, Great Ormond Street NHS Foundation Trust
 Patient Transfer TLM, NHS Foundation Trust Patient Transfer - intro Hospital was founded in 1852 by Charles West as the first hospital dedicated to the treatment of children. The original 10 bed hospital
Patient Transfer TLM, NHS Foundation Trust Patient Transfer - intro Hospital was founded in 1852 by Charles West as the first hospital dedicated to the treatment of children. The original 10 bed hospital
Assessment of Mental Capacity and Best Interest Decisions
 Standard Operating Procedure 1 (SOP 1) Assessment of Mental Capacity and Best Interest Decisions Why we have a procedure? This Standard Operating Procedure (SOP) is required to set out how a person s capacity
Standard Operating Procedure 1 (SOP 1) Assessment of Mental Capacity and Best Interest Decisions Why we have a procedure? This Standard Operating Procedure (SOP) is required to set out how a person s capacity
Specialised Services Commissioning Policy. CP29: Bariatric Surgery
 Specialised Services Commissioning Policy CP29: Bariatric Surgery Document Author: Specialist Planner, Cardiothoracic Executive Lead: Director of Planning Approved by: Management Group Issue Date: 12 June
Specialised Services Commissioning Policy CP29: Bariatric Surgery Document Author: Specialist Planner, Cardiothoracic Executive Lead: Director of Planning Approved by: Management Group Issue Date: 12 June
South East Coast Operational Delivery Network. Critical Care Rehabilitation
 South East Coast Operational Delivery Networks Hosted by Medway Foundation Trust South East Coast Operational Delivery Network Background Critical Care Rehabilitation The optimisation of recovery from
South East Coast Operational Delivery Networks Hosted by Medway Foundation Trust South East Coast Operational Delivery Network Background Critical Care Rehabilitation The optimisation of recovery from
Specialised Services Policy:
 Specialised Services Policy: CP35 Cochlear Implants Document Author: Specialised Planner for Women & Children s Services Executive Lead: Director of Planning Approved by: Executive Board Issue Date: 05
Specialised Services Policy: CP35 Cochlear Implants Document Author: Specialised Planner for Women & Children s Services Executive Lead: Director of Planning Approved by: Executive Board Issue Date: 05
Initiation of Clozapine Treatment Community Patients
 Initiation of Clozapine Treatment Community Patients Who Should Read This Policy Target Audience All clinical staff working in the community N/A N/A Initiation of Clozapine Treatment for Patients in the
Initiation of Clozapine Treatment Community Patients Who Should Read This Policy Target Audience All clinical staff working in the community N/A N/A Initiation of Clozapine Treatment for Patients in the
