CLINICAL GUIDELINE FOR THE ADMINISTRATION OF MESNA WITH IFOSFAMIDE AND CYCLOPHOSPHAMIDE Summary.
|
|
|
- Bruce Knight
- 6 years ago
- Views:
Transcription
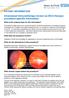
1 CLINICAL GUIDELINE FOR THE ADMINISTRATION OF MESNA WITH IFOSFAMIDE AND CYCLOPHOSPHAMIDE Summary. Yes Is patient prescribed ifosfamide or cyclophosphamide >1g/m 2? Chemotherapy prescription on Aria should include prophylactic mesna. action required Test urine using dipstick prior to commencing treatment and with each urine sample during infusion Trace Retest Retest If + on more than 1 consecutive test Administer 1g IV mesna bolus over 5 mins Double dose of mesna infusion Trace or no further haematuria + ++/+++ Continue with double Continue with double Stop ifosfamide / dose infusion. dose infusion. cyclophosphamide Clinical Guideline Amend for the Aria Administration for of Mesna with Amend Ifosfamide Aria and for Cyclophosphamide infusion subsequent cycles subsequent cycles Page 1 of 9
2 1. Aim/Purpose of this Guideline 1.1. The objectives of these guidelines are to promote consistent clinical practice in relation to the use of mesna in the prevention and treatment of ifosfamide and cyclophosphamide-induced urinary tract toxicity This guideline is relevant to: Oncology and haematology doctors Chemotherapy nurses working on Lowen ward and the Headland Unit Pharmacists 1.3. It is not intended to be used for non-cancer use of cyclophosphamide and ifosfamide (although the general principles may apply to these patients) or to paediatric patients. 2. The Guidance 2.1. Background The overall reported incidence of oxazaphosphorine-induced haemorrhagic cystitis varies considerably due to a lack of agreement for diagnostic criteria, variability in definition of the relevant time frame, and uncertainty regarding the relative contributing factors such as thrombocytopenia, concurrent or previous chemotherapy or radiotherapy, and viral infections. It is suggested that in patients treated with ifosfamide without urothelial protection the overall incidence of haemorrhagic cystitis ranges from 18% to 40% and is considered dose-limiting. Among patients treated with high-dose cyclophosphamide with aggressive hydration in the bone marrow transplantation setting, the reported incidence of severe haemorrhagic cystitis ranges from 0.5% to 40%. The incidence of haemorrhagic cystitis is considerably lower with cyclophosphamide as compared with ifosfamide and so mesna use is unnecessary with standard doses of cyclophosphamide i.e.<1g/m Rationale for the use of mesna Mesna is a thiol compound, which functions as a regional detoxicant of the oxazaphosphorine metabolites. After oral or intravenous administration, mesna undergoes rapid oxidation in the plasma to dimesna. Only a small portion of the dose remains in the circulation as the physiologically active compound. Both mesna and dimesna are very hydrophilic and therefore remain in the intravascular compartment, where they are rapidly cleared by the kidneys. The free sulfhydryl (thiol) groups of mesna combine directly with a double bond of acrolein and with other urotoxic 4- hydroxy-oxazaphosphorine metabolites to form stable nontoxic compounds. Because urinary mesna concentration greatly exceeds plasma mesna concentrations regional detoxification of urotoxic oxazaphosphorine metabolites occurs in the urinary system. This restriction of mesna to the urinary system implies that mesna neither protects against non-urologic toxicities of oxazaphosphorines nor interferes with their cytotoxic activity. Mesna in doses of up to mg/kg IV was shown to produce no toxic effect on bone marrow, hepatic, renal or CNS functions. Vomiting and diarrhoea only occurred after doses greater than 80mg/kg Prescribing information for mesna These are general principles followed for the majority of regimens used at RCHT but they may not represent all regimens currently in use Mesna should be used prophylactically for all patients receiving ifosfamide and high dose cyclophosphamide (>1g/m2). Page 2 of 9
3 2.3.2 Mesna dosing with ifosfamide - example regimen Pre-ifosfamide Mesna equivalent of 20% total daily ifosfamide dose given as IV bolus preifosfamide During ifosfamide Mesna equivalent of % total daily ifosfamide dose given as IV infusion over 24 hours (mixed in bags with ifosfamide) Post-ifosfamide Mesna equivalent of 60% total daily ifosfamide dose given as IV infusion over 12 hours Mesna dosing with high dose cyclophosphamide (>1g/m2) e.g. HSCT mobilisation (cyclophosphamide 1.5g/m2) Mesna equivalent of 40% total daily cyclophosphamide dose given as IV bolus pre-cyclophosphamide Mesna equivalent of 40% total daily cyclophosphamide dose given as IV bolus 3, 6 and 9 hours after then start of the cyclophosphamide infusion Bioavailability After oral administration mesna has a bioavailability of 50-75% and the urinary mesna concentrations are approximately one half of those observed after IV infusion. N.B. Mesna tablet strengths: 400 mg or 600mg. Round up dose if necessary Haematuria Haemorrhagic cystitis is generally graded as mild, moderate or severe according to the degree of pain and haematuria. Severe haemorrhagic cystitis typically includes the presence of gross haematuria with clots and occurrence of clinical complications; it can be extremely painful and debilitating, requiring prolonged and expensive hospitalisation Monitoring for haematuria Patients should always have their urine tested by dipstick for blood prior to starting ifosfamide or cyclophosphamide. This will provide a baseline level from which to assess any haematuria that may occur. Thereafter urine should be tested with each sample Presentation of haematuria Prior to the start of the ifosfamide or cyclophosphamide infusion An unexplained positive test for blood prior to treatment should be investigated. Once other causes of haematuria have been excluded, for example urinary tract infection or menstruation, take a spot urine sample for microscopy for red cells in order to exclude false positive results. Results should be awaited before commencing ifosfamide or cyclophosphamide. If cause identified further testing of urine may be unnecessary due to false positive results Whilst receiving intravenous mesna hydration. Exclude other causes of haematuria, for example urinary tract infection Page 3 of 9
4 or menstruation. Take a spot urine sample for microscopy for red cells in order to exclude false positive results and follow the guidelines below Test result Action Trace Re-test + Re-test If + on more than one consecutive test give additional bolus mesna ++ Double intravenous mesna +++ Double intravenous mesna Doubling mesna doses If the infusion is running containing ifosfamide mixed with mesna, leave this bag running and piggyback an additional bag containing the same amount of mesna. If the bag running contains mesna only, stop this infusion and replace with a new bag containing double the amount of mesna and run for the time remaining i.e. until a minimum of 12 hours after final ifosfamide infusion. NB ensure that future bags are prescribed with the increased dose Whilst receiving oral mesna Patients who are switched to oral mesna for discharge should be educated to test their urine for up to 12 hours after the end of the ifosfamide/cyclophosphamide infusion. For patients experiencing haematuria after discharge on oral mesna the following guide should be followed: Test result Action Trace Re-test + Re-test If + on more than one consecutive test give intravenous mesna ++ Admit for intravenous mesna +++ Admit for intravenous mesna These test results are all as above baseline results. An extra dose of oral mesna will be supplied to patients to be taken if an episode of vomiting is experienced while taking their regular oral mesna dose. Consider advising taking this dose (if not already taken) in addition to being admitted to hospital. An intravenous mesna bolus followed by a continuous infusion as per previous days should be given and continued until haematuria resolved. Consideration should also be given to the suitability of the patient for oral mesna with further cycles of ifosfamide Subsequent courses If higher doses of IV mesna or a switch from oral to IV mesna were required during a treatment cycle and were effective at preventing further haematuria ensure future prescriptions on Aria are amended as appropriate and document on treatment notes. Page 4 of 9
5 3. Monitoring compliance and effectiveness Element to be monitored Lead Tool Frequency Reporting arrangements Acting on recommendations and Lead(s) Change in practice and lessons to be shared Appropriate prescribing of mesna All incidences of haematuria following ifosfamide/cyclophosphamide to be reported via Datix. to review Datix as standing item, and review whether appropriate management occurred. Monthly via chemotherapy MDT Recorded in meeting minutes Required changes to practice will be identified and actioned within one month. A lead member of the team will be identified to take each change forward where appropriate. Lessons will be shared with all the relevant stakeholders 4. Equality and Diversity 4.1. This document complies with the Royal Cornwall Hospitals NHS Trust service Equality and Diversity statement which can be found in the 'Equality, Diversity & Human Rights Policy' or the Equality and Diversity website Equality Impact Assessment The Initial Equality Impact Assessment Screening Form is at Appendix 2. Page 5 of 9
6 Appendix 1. Governance Information Document Title Date Issued/Approved: January 2016 Clinical Guideline for the Administration of Mesna with Ifosfamide and Cyclophosphamide Date Valid From: January 2016 Date Valid To: January 2019 Directorate / Department responsible (author/owner): Emma Nicholls, Lead Cancer Pharmacist Contact details: Brief summary of contents Suggested Keywords: Target Audience Executive Director responsible for Policy: Date revised: This document replaces (exact title of previous version): Approval route (names of committees)/consultation: Divisional Manager confirming approval processes Name and Post Title of additional signatories Name and Signature of Divisional/Directorate Governance Lead confirming approval by specialty and divisional management meetings Signature of Executive Director giving approval Publication Location (refer to Policy on Policies Approvals and Ratification): Recommendations for the prescribing and administration of mesna with ifosfamide and cyclophosphamide for adult cancer patients Mesna, cyclophosphamide, ifosfamide, cystitis RCHT PCH CFT KCCG Medical Director N/A New Document Medicines Practice Committee ( ) CSSC Governance DMB Sally Kennedy, Divisional Director CSSC t Required {Original Copy Signed} Name: Janet Gardner, CSSC Governance Lead {Original Copy Signed} Internet & Intranet Intranet Only Page 6 of 9
7 Document Library Folder/Sub Folder Links to key external standards Related Documents: Training Need Identified? Clinical / Cancer Services Adapted from policy of London Cancer rth & East Version Control Table Date Version 2/11/15 V1.0 Initial Issue Summary of Changes Changes Made by (Name and Job Title) Emma Nicholls Lead Cancer Pharmacist All or part of this document can be released under the Freedom of Information Act 2000 This document is to be retained for 10 years from the date of expiry. This document is only valid on the day of printing Controlled Document This document has been created following the Royal Cornwall Hospitals NHS Trust Policy on Document Production. It should not be altered in any way without the express permission of the author or their Line Manager. Page 7 of 9
8 Appendix 2. Initial Equality Impact Assessment Form Name of Name of the strategy / policy /proposal / service function to be assessed (hereafter referred to as policy) (Provide brief description): Directorate and service area: Is this a new or existing Policy? CSSC New Name of individual completing Telephone: assessment: Emma Nicholls 1. Policy Aim* Who is the strategy / policy / proposal / service function aimed at? Safe and effective prescribing of cyclophosphamide, ifosfamide and mesna 2. Policy Objectives* Safe and effective prescribing of cyclophosphamide, ifosfamide and mesna 3. Policy intended Ensure that all patients are prescribed required mesna and that staff Outcomes* are aware of action to be taken in the event of haematuria 4. *How will you measure the outcome? 5. Who is intended to benefit from the policy? 6a) Is consultation required with the workforce, equality groups, local interest groups etc. around this policy? Annual review Adult cancer patients at the Royal Cornwall Hospital Yes b) If yes, have these *groups been consulted? C). Please list any groups who have been consulted about this procedure. Yes 7. The Impact Please complete the following table. Are there concerns that the policy could have differential impact on: Equality Strands: Yes Rationale for Assessment / Existing Evidence Age Sex (male, female, transgender / gender reassignment) Race / Ethnic communities /groups Page 8 of 9
9 Disability - Learning disability, physical disability, sensory impairment and mental health problems Religion / other beliefs Marriage and civil partnership Pregnancy and maternity Sexual Orientation, Bisexual, Gay, heterosexual, Lesbian You will need to continue to a full Equality Impact Assessment if the following have been highlighted: You have ticked Yes in any column above and consultation or evidence of there being consultation- this excludes any policies which have been identified as not requiring consultation. or Major service redesign or development 8. Please indicate if a full equality analysis is recommended. 9. If you are not recommending a Full Impact assessment please explain why. Signature of policy developer / lead manager / director Date of completion and submission Names and signatures of members carrying out the Screening Assessment 1. Emma Nicholls 2. Keep one copy and send a copy to the Human Rights, Equality and Inclusion Lead, c/o Royal Cornwall Hospitals NHS Trust, Human Resources Department, Knowledge Spa, Truro, Cornwall, TR1 3HD A summary of the results will be published on the Trust s web site. Signed Date Page 9 of 9
Blood Glucose and Hyperglycaemia Management in Hospital for Adults with Diabetes Clinical Guideline V2.0. March 2018
 Blood Glucose and Hyperglycaemia Management in Hospital for Adults with Diabetes Clinical Guideline V2.0 March 2018 Page 1 of 8 Summary flow chart for monitoring of blood glucose if >11mmol/L For Adults
Blood Glucose and Hyperglycaemia Management in Hospital for Adults with Diabetes Clinical Guideline V2.0 March 2018 Page 1 of 8 Summary flow chart for monitoring of blood glucose if >11mmol/L For Adults
CLINICAL GUIDELINE FOR THE ADMINISTRATION OF NEBULISED PENTAMIDINE Summary. 1.
 CLINICAL GUIDELINE FOR THE ADMINISTRATION OF NEBULISED PENTAMIDINE Summary. 1. Patient requires nebulised Pentamidine Ensure equipment listed is available Ensure HEPA filtered room in Haematology Clinic
CLINICAL GUIDELINE FOR THE ADMINISTRATION OF NEBULISED PENTAMIDINE Summary. 1. Patient requires nebulised Pentamidine Ensure equipment listed is available Ensure HEPA filtered room in Haematology Clinic
Clinical Guideline for Intravenous Opioids for Adults in Recovery Areas The Recovery Protocol
 Clinical Guideline for Intravenous Opioids for Adults in Recovery Areas The Recovery Protocol 1. Aim/Purpose of this Guideline 1.1. To Provide safe and efficient administration of Opioids in Recovery.
Clinical Guideline for Intravenous Opioids for Adults in Recovery Areas The Recovery Protocol 1. Aim/Purpose of this Guideline 1.1. To Provide safe and efficient administration of Opioids in Recovery.
SALBUTAMOL INHALER PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline
 SALBUTAMOL INHALER PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline 1.1. This Patient Group Direction (PGD) applies to all nursing and clinical staff in the Child Health Department
SALBUTAMOL INHALER PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline 1.1. This Patient Group Direction (PGD) applies to all nursing and clinical staff in the Child Health Department
28 th September Author Jeremy Gilbert Bariatric Nurse Specialist
 POLICY FOR SELF ADMINISTRATION OF CONTINUOUS POSITIVE AIRWAY PRESSURE BY COMPETENT PATIENTS COMING IN FOR METABOLIC AND OBESITY SURGERY (BARIATRIC SURGERY) TO PENDENNIS WARD 28 th September 2014 Author
POLICY FOR SELF ADMINISTRATION OF CONTINUOUS POSITIVE AIRWAY PRESSURE BY COMPETENT PATIENTS COMING IN FOR METABOLIC AND OBESITY SURGERY (BARIATRIC SURGERY) TO PENDENNIS WARD 28 th September 2014 Author
1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of acamprosate.
 SHARED CARE GUIDELINE FOR ACAMPROSATE 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of
SHARED CARE GUIDELINE FOR ACAMPROSATE 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of
CLINICAL GUIDELINE FOR THE MANAGEMENT OF BARRETT S OESOPHAGUS Summary.
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF BARRETT S OESOPHAGUS Summary. Page 1 of 12 Recurrent LGD at any point- discuss at MDT And consider RFA Patients with LGD should have a repeat endoscopy in 6 months.
CLINICAL GUIDELINE FOR THE MANAGEMENT OF BARRETT S OESOPHAGUS Summary. Page 1 of 12 Recurrent LGD at any point- discuss at MDT And consider RFA Patients with LGD should have a repeat endoscopy in 6 months.
DIAGNOSIS AND MANAGEMENT OF PYLORIC STENOSIS IN CHILDREN CLINICAL GUIDELINE V3.0
 DIAGNOSIS AND MANAGEMENT OF PYLORIC STENOSIS IN CHILDREN CLINICAL GUIDELINE V3.0 1. Aim/Purpose of this Guideline This guideline is relevant to all medical and nursing staff caring for children with Pyloric
DIAGNOSIS AND MANAGEMENT OF PYLORIC STENOSIS IN CHILDREN CLINICAL GUIDELINE V3.0 1. Aim/Purpose of this Guideline This guideline is relevant to all medical and nursing staff caring for children with Pyloric
CLINICAL GUIDELINE FOR MANAGEMENT OF NEUTROPENIC SEPSIS IN ADULT CANCER PATIENTS (this guideline excludes haematology patients)
 CLINICAL GUIDELINE FOR MANAGEMENT OF NEUTROPENIC SEPSIS IN ADULT CANCER PATIENTS (this guideline excludes haematology patients) 1. Aim/Purpose of this Guideline 1.1. Systemic cancer treatments and immunological
CLINICAL GUIDELINE FOR MANAGEMENT OF NEUTROPENIC SEPSIS IN ADULT CANCER PATIENTS (this guideline excludes haematology patients) 1. Aim/Purpose of this Guideline 1.1. Systemic cancer treatments and immunological
Start. What is the serum phosphate concentration? Moderate Hypophosphataemia mmol/l. Replace using oral. phosphate. (See section 3.
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF HYPOPHOSPHATAEMIA IN ADULTS Summary. Key: General Notes GP/SWASFT ED/MAU/SRU/Acute GP/Amb-Care In-patient wards Start What is the serum concentration? Mild Hypophosphataemia
CLINICAL GUIDELINE FOR THE MANAGEMENT OF HYPOPHOSPHATAEMIA IN ADULTS Summary. Key: General Notes GP/SWASFT ED/MAU/SRU/Acute GP/Amb-Care In-patient wards Start What is the serum concentration? Mild Hypophosphataemia
School Hearing Screening Policy
 School Hearing Screening Policy V2.1 1st August 2017 Page 1 of 13 Table of Contents 1. Introduction... 3 2. Purpose of this Policy... 3 3. Scope... 3 4. Definitions / Glossary... 3 5. Ownership and Responsibilities...
School Hearing Screening Policy V2.1 1st August 2017 Page 1 of 13 Table of Contents 1. Introduction... 3 2. Purpose of this Policy... 3 3. Scope... 3 4. Definitions / Glossary... 3 5. Ownership and Responsibilities...
HYPOSPADIAS NEONATAL CLINICAL GUIDELINE. 1. Aim/Purpose of this Guideline. 2. The Guidance
 HYPOSPADIAS NEONATAL CLINICAL GUIDELINE 1. Aim/Purpose of this Guideline 1.1. This guideline applies to all staff managing the initial care of infants born with hypospadias. It includes assessment and
HYPOSPADIAS NEONATAL CLINICAL GUIDELINE 1. Aim/Purpose of this Guideline 1.1. This guideline applies to all staff managing the initial care of infants born with hypospadias. It includes assessment and
SHARED CARE GUIDELINE FOR BUCCAL MIDAZOLAM FOR THE TREATMENT OF PROLONGED SEIZURES IN CHILDREN
 SHARED CARE GUIDELINE FOR BUCCAL MIDAZOLAM FOR THE TREATMENT OF PROLONGED SEIZURES IN CHILDREN 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the
SHARED CARE GUIDELINE FOR BUCCAL MIDAZOLAM FOR THE TREATMENT OF PROLONGED SEIZURES IN CHILDREN 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the
METABOLIC BONE DISEASE OF PREMATURITY NEONATAL CLINICAL GUIDELINE V3.0
 METABOLIC BONE DISEASE OF PREMATURITY NEONATAL CLINICAL GUIDELINE V3.0 Page 1 of 10 1. Aim/Purpose of this Guideline To provide guidance on the prevention of metabolic bone disease in the neonate. All
METABOLIC BONE DISEASE OF PREMATURITY NEONATAL CLINICAL GUIDELINE V3.0 Page 1 of 10 1. Aim/Purpose of this Guideline To provide guidance on the prevention of metabolic bone disease in the neonate. All
MANAGEMENT OF THE BLADDER IN THE POSTOPERATIVE PERIOD FOLLOWING UNCOMPLICATED GYNAECOLOGICAL SURGERY CLINICAL GUIDELINES
 MANAGEMENT OF THE BLADDER IN THE POSTOPERATIVE PERIOD FOLLOWING UNCOMPLICATED GYNAECOLOGICAL SURGERY CLINICAL GUIDELINES 1. Aim/Purpose of this Guideline All clinical staff working in the Division of women,
MANAGEMENT OF THE BLADDER IN THE POSTOPERATIVE PERIOD FOLLOWING UNCOMPLICATED GYNAECOLOGICAL SURGERY CLINICAL GUIDELINES 1. Aim/Purpose of this Guideline All clinical staff working in the Division of women,
CLINICAL GUIDELINE FOR THE MANAGEMENT OF HYPOKALAEMIA
 POLICY UNDER REVIEW Please note that this policy is under review. It does, however, remain current Trust policy subject to any recent legislative changes, national policy instruction (NHS or Department
POLICY UNDER REVIEW Please note that this policy is under review. It does, however, remain current Trust policy subject to any recent legislative changes, national policy instruction (NHS or Department
Fasting for Adults (including Young Adults Age 16+ years) who require Anaesthesia or Intravenous Sedation Clinical Guideline V5.0
 Fasting for Adults (including Young Adults Age 16+ years) who require Anaesthesia or Intravenous Sedation November 2018 Summary. Fasting for Adults (including Young Adults Age 16+ years) who require Anaesthesia
Fasting for Adults (including Young Adults Age 16+ years) who require Anaesthesia or Intravenous Sedation November 2018 Summary. Fasting for Adults (including Young Adults Age 16+ years) who require Anaesthesia
SHARED CARE GUIDELINE FOR RIFAXIMIN FOR PREVENTING EPISODES OF OVERT HEPATIC ENCEPHALOPATHY IN ADULT PATIENTS 1. Aim/Purpose of this Guideline
 SHARED CARE GUIDELINE FOR RIFAXIMIN FOR PREVENTING EPISODES OF OVERT HEPATIC ENCEPHALOPATHY IN ADULT PATIENTS 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy
SHARED CARE GUIDELINE FOR RIFAXIMIN FOR PREVENTING EPISODES OF OVERT HEPATIC ENCEPHALOPATHY IN ADULT PATIENTS 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy
CLINICAL GUIDELINE FOR INTRAVENOUS FLUID THERAPY FOR ADULTS IN HOSPITAL 1. Aim/Purpose of this Guideline
 CLINICAL GUIDELINE FOR INTRAVENOUS FLUID THERAPY FOR ADULTS IN HOSPITAL 1. Aim/Purpose of this Guideline 1.1. This guideline contains recommendations about general principles for managing intravenous (IV)
CLINICAL GUIDELINE FOR INTRAVENOUS FLUID THERAPY FOR ADULTS IN HOSPITAL 1. Aim/Purpose of this Guideline 1.1. This guideline contains recommendations about general principles for managing intravenous (IV)
PRESEPTAL AND ORBITAL CELLULITIS IN CHILDREN- CLINICAL GUIDELINE V3.0
 PRESEPTAL AND ORBITAL CELLULITIS IN CHILDREN- CLINICAL GUIDELINE V3.0 Page 1 of 8 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical and nursing staff caring for a child with Preseptal
PRESEPTAL AND ORBITAL CELLULITIS IN CHILDREN- CLINICAL GUIDELINE V3.0 Page 1 of 8 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical and nursing staff caring for a child with Preseptal
CLINICAL GUIDELINE FOR MANAGEMENT OF GALLSTONES PATHOLOGY IN ADULTS
 CLINICAL GUIDELINE FOR MANAGEMENT OF GALLSTONES PATHOLOGY IN ADULTS 1. Aim/Purpose of this Guideline This guideline is for the management of gallstones pathology in adults. It has been benchmarked against
CLINICAL GUIDELINE FOR MANAGEMENT OF GALLSTONES PATHOLOGY IN ADULTS 1. Aim/Purpose of this Guideline This guideline is for the management of gallstones pathology in adults. It has been benchmarked against
Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall
 Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall Page 1 of 15 Summary Patient clinically assessed and reviewed by a Consultant Cardiologist, Cardiology
Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall Page 1 of 15 Summary Patient clinically assessed and reviewed by a Consultant Cardiologist, Cardiology
SHARED CARE GUIDELINE FOR MODAFINIL 1. Aim/Purpose of this Guideline. 2. The Guidance
 SHARED CARE GUIDELINE FOR MODAFINIL 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of modafinil.
SHARED CARE GUIDELINE FOR MODAFINIL 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of modafinil.
GESTATIONAL DIABETES MELLITUS AND SUBSEQUENT MANAGEMENT OF CONFIRMED GESTATIONAL DIABETES MELLITUS (GDM) AND SELECTIVE SCREENING - CLINICAL GUIDELINE
 GESTATIONAL DIABETES MELLITUS AND SUBSEQUENT MANAGEMENT OF CONFIRMED GESTATIONAL DIABETES MELLITUS (GDM) AND SELECTIVE SCREENING - CLINICAL GUIDELINE V 1.5 2017 Screening - Clinical Guideline Page 1 of
GESTATIONAL DIABETES MELLITUS AND SUBSEQUENT MANAGEMENT OF CONFIRMED GESTATIONAL DIABETES MELLITUS (GDM) AND SELECTIVE SCREENING - CLINICAL GUIDELINE V 1.5 2017 Screening - Clinical Guideline Page 1 of
CLINICAL GUIDELINE FOR THE MANAGEMENT OF HIGH BLOOD GLUCOSE LEVELS AND SICK DAYS FOR INSULIN PUMP USERS UNDER THE PAEDIATRIC DIABETES SERVICE. V4.
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF HIGH BLOOD GLUCOSE LEVELS AND SICK DAYS FOR INSULIN PUMP USERS UNDER THE PAEDIATRIC DIABETES SERVICE. V4.1 Page 1 of 11 1. Aim/Purpose of this Guideline 1.1. The
CLINICAL GUIDELINE FOR THE MANAGEMENT OF HIGH BLOOD GLUCOSE LEVELS AND SICK DAYS FOR INSULIN PUMP USERS UNDER THE PAEDIATRIC DIABETES SERVICE. V4.1 Page 1 of 11 1. Aim/Purpose of this Guideline 1.1. The
Captopril and Enalapril (Ace Inhibitor) Therapy Clinical Guideline V1.0
 Captopril and Enalapril (Ace Inhibitor) Therapy Clinical Guideline V1.0 November 2018 Summary Prescribing, monitoring and administration of Captopril and Enalapril FOR STAFF PATIENTS Medical and Nursing
Captopril and Enalapril (Ace Inhibitor) Therapy Clinical Guideline V1.0 November 2018 Summary Prescribing, monitoring and administration of Captopril and Enalapril FOR STAFF PATIENTS Medical and Nursing
MANAGEMENT OF NEONATAL HYPOTENSION CLINICAL GUIDELINE
 MANAGEMENT OF NEONATAL HYPOTENSION CLINICAL GUIDELINE 1. Aim/Purpose of this Guideline 1.1. To provide guidance on the assessment and management of infants with hypotension. All involved will benefit from
MANAGEMENT OF NEONATAL HYPOTENSION CLINICAL GUIDELINE 1. Aim/Purpose of this Guideline 1.1. To provide guidance on the assessment and management of infants with hypotension. All involved will benefit from
Osteoblasts (cells which form new bone) Osteoclasts (cells which break down old bone)
 Clinical guideline for the administration of Bisphosphonates and other drugs affecting bone metabolism in Haematology and Oncology patients 1. Aim/Purpose of this Guideline 1.1. To provide education to
Clinical guideline for the administration of Bisphosphonates and other drugs affecting bone metabolism in Haematology and Oncology patients 1. Aim/Purpose of this Guideline 1.1. To provide education to
CLINICAL GUIDELINE FOR USE OF A PATIENT CONTROLLED ANALGESIA OR INTRAVENOUS OPIATE INFUSION IN CHILD HEALTH. 1. Aim/Purpose of this Guideline
 CLINICAL GUIDELINE FOR USE OF A PATIENT CONTROLLED ANALGESIA OR INTRAVENOUS OPIATE INFUSION IN CHILD HEALTH. 1. Aim/Purpose of this Guideline Guideline for children with a Patient Controlled Analgesia
CLINICAL GUIDELINE FOR USE OF A PATIENT CONTROLLED ANALGESIA OR INTRAVENOUS OPIATE INFUSION IN CHILD HEALTH. 1. Aim/Purpose of this Guideline Guideline for children with a Patient Controlled Analgesia
CLINICAL GUIDELINE FOR THE MANAGEMENT OF ANAPHYLAXIS IN INFANTS AND CHILDREN UNDER SIXTEEN YEARS OF AGE V3.0
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF ANAPHYLAIS IN INFANTS AND CHILDREN UNDER SITEEN YEARS OF AGE V3.0 Page 1 of 16 1. Aim/Purpose of this Guideline 1.1. The purpose of this guideline is to provide
CLINICAL GUIDELINE FOR THE MANAGEMENT OF ANAPHYLAIS IN INFANTS AND CHILDREN UNDER SITEEN YEARS OF AGE V3.0 Page 1 of 16 1. Aim/Purpose of this Guideline 1.1. The purpose of this guideline is to provide
Patient Controlled Analgesia/Intravenous Opiate Infusion in Child Health Clinical Guideline V4.0 October 2018
 Patient Controlled Analgesia/Intravenous Opiate Infusion in Child Health Clinical Guideline V4.0 October 2018 Page 1 of 12 1. Aim/Purpose of this Guideline Guideline for children with a Patient Controlled
Patient Controlled Analgesia/Intravenous Opiate Infusion in Child Health Clinical Guideline V4.0 October 2018 Page 1 of 12 1. Aim/Purpose of this Guideline Guideline for children with a Patient Controlled
Hypoglycaemia in Adults with Diabetes Clinical Guideline V5.0. March 2018
 March 2018 Page 1 of 11 Adult with Diabetes and blood glucose < 4 mmol/l Treatment as per Guideline: Nursing / Clinical Staff Patient reviewed as per guideline guidance notes: Nursing / Clinical Staff
March 2018 Page 1 of 11 Adult with Diabetes and blood glucose < 4 mmol/l Treatment as per Guideline: Nursing / Clinical Staff Patient reviewed as per guideline guidance notes: Nursing / Clinical Staff
CLINICAL GUIDELINE FOR THE USE OF PHENYTOIN IN EPILEPSY
 This applies to adult patients only CLINICAL GUIDELINE FOR THE USE OF PHENYTOIN IN EPILEPSY Key: General Notes ED/MAU/SRU/Acute GP/Amb-Care GP/SWASFT In-patient wards Start Yes Patient already taking phenytoin?
This applies to adult patients only CLINICAL GUIDELINE FOR THE USE OF PHENYTOIN IN EPILEPSY Key: General Notes ED/MAU/SRU/Acute GP/Amb-Care GP/SWASFT In-patient wards Start Yes Patient already taking phenytoin?
CLINICAL GUIDELINE FOR AN EPIDURAL INFUSION IN CHILD HEALTH 1. Aim/Purpose of this Guideline
 CLINICAL GUIDELINE FOR AN EPIDURAL INFUSION IN CHILD HEALTH 1. Aim/Purpose of this Guideline 1.1. The purpose of this guideline is to provide guidance on caring for children who are receiving epidural
CLINICAL GUIDELINE FOR AN EPIDURAL INFUSION IN CHILD HEALTH 1. Aim/Purpose of this Guideline 1.1. The purpose of this guideline is to provide guidance on caring for children who are receiving epidural
CLINICAL GUIDELINE FOR THE MANAGEMENT OF HYPONATRAEMIA Summary. Start. End. Key: Na + below normal range ( mmol/L) Symptomatic?
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF HYPONATRAEMIA Summary Key: General tes ED/MAU/SRU/Acute GP/Amb-Care GP/SWASFT In-patient wards Start Na + below normal range (135 145mmol/L) Refer to endocrinology
CLINICAL GUIDELINE FOR THE MANAGEMENT OF HYPONATRAEMIA Summary Key: General tes ED/MAU/SRU/Acute GP/Amb-Care GP/SWASFT In-patient wards Start Na + below normal range (135 145mmol/L) Refer to endocrinology
Suspected Pulmonary embolus Ambulatory Pathway. Document Title. Date Issued/Approved: Date Valid From: 11/11/17. Date Valid To: 11/05/18
 POLICY UNDER REVIEW Please note that this policy is under review. It does, however, remain current Trust policy subject to any recent legislative changes, national policy instruction (NHS or Department
POLICY UNDER REVIEW Please note that this policy is under review. It does, however, remain current Trust policy subject to any recent legislative changes, national policy instruction (NHS or Department
CLINICAL PROCEDURE FOR THE SAFE REMOVAL OF FEMORAL ARTERIAL SHEATHS USING A DIGITAL APPROACH 1. Aim/Purpose of this Guideline
 POLICY UNDER REVIEW Please note that this policy is under review. It does, however, remain current Trust policy subject to any recent legislative changes, national policy instruction (NHS or Department
POLICY UNDER REVIEW Please note that this policy is under review. It does, however, remain current Trust policy subject to any recent legislative changes, national policy instruction (NHS or Department
Prevention and Treatment of Mucositis in Children and Young People with Cancer Clinical Guideline V3.1 December 2018
 Prevention and Treatment of Mucositis in Children and Young People with Cancer Clinical Guideline V3.1 December 2018 1. Aim/Purpose of this Guideline This guideline applies to all medical and nursing staff
Prevention and Treatment of Mucositis in Children and Young People with Cancer Clinical Guideline V3.1 December 2018 1. Aim/Purpose of this Guideline This guideline applies to all medical and nursing staff
MUCOSITIS IN CHILDREN AND YOUNG PEOPLE WITH CANCER- CLINICAL GUIDELINE FOR PREVENTION AND TREATMENT V3.0
 MUCOSITIS IN CHILDREN AND YOUNG PEOPLE WITH CANCER- CLINICAL GUIDELINE FOR PREVENTION AND TREATMENT V3.0 Page 1 of 10 1. Aim/Purpose of this Guideline 1.1. This guideline applies to all medical and nursing
MUCOSITIS IN CHILDREN AND YOUNG PEOPLE WITH CANCER- CLINICAL GUIDELINE FOR PREVENTION AND TREATMENT V3.0 Page 1 of 10 1. Aim/Purpose of this Guideline 1.1. This guideline applies to all medical and nursing
SHARED CARE GUIDELINE FOR TREATMENT OF DEMENTIA 1. Aim/Purpose of this Guideline
 POLICY UNDER REVIEW Please note that this policy is under review. It does, however, remain current Trust policy subject to any recent legislative changes, national policy instruction (NHS or Department
POLICY UNDER REVIEW Please note that this policy is under review. It does, however, remain current Trust policy subject to any recent legislative changes, national policy instruction (NHS or Department
CLINICAL GUIDELINE FOR NEONATAL BCG VACCINATION V3.0
 CLINICAL GUIDELINE FOR NEONATAL BCG VACCINATION V3.0 Page 1 of 11 1. Aim/Purpose of this Guideline 1.1. Tuberculosis is a notifiable disease in the UK and although incidence is low it remains a Public
CLINICAL GUIDELINE FOR NEONATAL BCG VACCINATION V3.0 Page 1 of 11 1. Aim/Purpose of this Guideline 1.1. Tuberculosis is a notifiable disease in the UK and although incidence is low it remains a Public
SHARED CARE GUIDELINE FOR MYCOPHENOLATE MOFETIL FOR RHEUMATOLOGY INDICATIONS 1. Aim/Purpose of this Guideline
 SHARED CARE GUIDELINE FOR MYCOPHENOLATE MOFETIL FOR RHEUMATOLOGY INDICATIONS 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate
SHARED CARE GUIDELINE FOR MYCOPHENOLATE MOFETIL FOR RHEUMATOLOGY INDICATIONS 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate
Procedure for Subcutaneous Injection of Insulin or GLP1 Analogue in Adults Using a Pen Device V2.0
 Procedure for Subcutaneous Injection of Insulin or GLP1 Analogue in Adults Using a Pen Device V2.0 11 October 2017 Table of Contents 1. Introduction.. 3 2. Purpose of this Policy/Procedure. 3 3. Scope
Procedure for Subcutaneous Injection of Insulin or GLP1 Analogue in Adults Using a Pen Device V2.0 11 October 2017 Table of Contents 1. Introduction.. 3 2. Purpose of this Policy/Procedure. 3 3. Scope
CLINICAL GUIDELINE FOR THE MANAGEMENT OF CONVULSIVE STATUS EPILEPTICUS IN CHILDHOOD V3.0
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF CONVULSIVE STATUS EPILEPTICUS IN CHILDHOOD V3.0 Clinical Guideline Template Page 1 of 14 1. Aim/Purpose of this Guideline 1.1. This guideline applies to all nursing
CLINICAL GUIDELINE FOR THE MANAGEMENT OF CONVULSIVE STATUS EPILEPTICUS IN CHILDHOOD V3.0 Clinical Guideline Template Page 1 of 14 1. Aim/Purpose of this Guideline 1.1. This guideline applies to all nursing
CLINICAL GUIDELINE FOR MANAGEMENT OF ACUTE CHOLECYSTITIS IN ADULTS
 CLINICAL GUIDELINE FOR MANAGEMENT OF ACUTE CHOLECYSTITIS IN ADULTS 1. Aim/Purpose of this Guideline This guideline is for the management of acute cholecystitis in adults. It has been benchmarked against
CLINICAL GUIDELINE FOR MANAGEMENT OF ACUTE CHOLECYSTITIS IN ADULTS 1. Aim/Purpose of this Guideline This guideline is for the management of acute cholecystitis in adults. It has been benchmarked against
DIABETES IN PREGNANCY, TYPE 1 AND TYPE 2 DIABETES MELLITUS (DM), CLINICAL GUIDELINE FOR MIDWIVES V1.4
 DIABETES IN PREGNANCY, TYPE 1 AND TYPE 2 DIABETES MELLITUS (DM), CLINICAL GUIDELINE FOR MIDWIVES V1.4 1 1. Aim/Purpose of this Guideline To provide guidance to Midwives, Obstetricians and the Joint Obstetric/Diabetes
DIABETES IN PREGNANCY, TYPE 1 AND TYPE 2 DIABETES MELLITUS (DM), CLINICAL GUIDELINE FOR MIDWIVES V1.4 1 1. Aim/Purpose of this Guideline To provide guidance to Midwives, Obstetricians and the Joint Obstetric/Diabetes
Clinical Guideline for the Management of Pot Operative Atrial Fibrillation
 Clinical Guideline for the Management of Pot Operative Atrial Fibrillation 1. Aim/Purpose of this Guideline 1.1. Atrial Fibrillation is the most common cardiac arrhythmia with a prevalence of around 0.5%
Clinical Guideline for the Management of Pot Operative Atrial Fibrillation 1. Aim/Purpose of this Guideline 1.1. Atrial Fibrillation is the most common cardiac arrhythmia with a prevalence of around 0.5%
CLINICAL GUIDELINE FOR THE MANAGEMENT OF VIRAL LARYNGO-TRACHEOBRONCHITIS (CROUP) V3.0
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF VIRAL LARYNGO-TRACHEOBRONCHITIS (CROUP) V3.0 Clinical Guideline Template Page 1 of 13 1. Aim/Purpose of this Guideline 1.1. This guideline applies to all nursing
CLINICAL GUIDELINE FOR THE MANAGEMENT OF VIRAL LARYNGO-TRACHEOBRONCHITIS (CROUP) V3.0 Clinical Guideline Template Page 1 of 13 1. Aim/Purpose of this Guideline 1.1. This guideline applies to all nursing
CLINICAL GUIDELINE FOR ANTIEMETIC USE IN PAEDIATRIC ONCOLOGY 1. Aim/Purpose of this Guideline
 CLINICAL GUIDELINE FOR ANTIEMETIC USE IN PAEDIATRIC ONCOLOGY 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical and nursing staff working with paediatric oncology patients. 2. The
CLINICAL GUIDELINE FOR ANTIEMETIC USE IN PAEDIATRIC ONCOLOGY 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical and nursing staff working with paediatric oncology patients. 2. The
SHARED CARE GUIDELINE FOR LITHIUM. 1. Aim/Purpose of this Guideline. 2. The Guidance
 SHARED CARE GUIDELINE FOR LITHIUM 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of lithium.
SHARED CARE GUIDELINE FOR LITHIUM 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration of lithium.
Title Use of Pre-Operative Tests for Elective Surgery Guideline. Department. Anaesthetics. Comment / Changes / Approval
 Document Control Title Use of Pre-Operative Tests for Elective Surgery Guideline Author Consultant Anaesthetist Author s job title Consultant Anaesthetist Directorate Clinical Support Services Department
Document Control Title Use of Pre-Operative Tests for Elective Surgery Guideline Author Consultant Anaesthetist Author s job title Consultant Anaesthetist Directorate Clinical Support Services Department
The Management of Children and Young People with Newly Presenting Diabetes Clinical Guideline V5.0 December 2018
 The Management of Children and Young People with Newly Presenting Diabetes Clinical Guideline December 2018 Summary GP suspects diabetes Under 16 years or still in School Year 11 NO Refer Adults YES Section
The Management of Children and Young People with Newly Presenting Diabetes Clinical Guideline December 2018 Summary GP suspects diabetes Under 16 years or still in School Year 11 NO Refer Adults YES Section
CLINICAL GUIDELINE FOR THE EVALUATION OF A CHILD PRESENTING WITH FEVER AND SEIZURE V3.0
 CLINICAL GUIDELINE FOR THE EVALUATION OF A CHILD PRESENTING WITH FEVER AND SEIZURE V3.0 Clinical Guideline Template Page 1 of 18 Page 1 of 13 1. Aim/Purpose of this Guideline 1.1. This guideline applies
CLINICAL GUIDELINE FOR THE EVALUATION OF A CHILD PRESENTING WITH FEVER AND SEIZURE V3.0 Clinical Guideline Template Page 1 of 18 Page 1 of 13 1. Aim/Purpose of this Guideline 1.1. This guideline applies
SHARED CARE GUIDELINE FOR CICLOSPORIN IN DERMATOLOGY. 1. Aim/Purpose of this Guideline. 2. The Guidance
 SHARED CARE GUIDELINE FOR CICLOSPORIN IN DERMATOLOGY 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration
SHARED CARE GUIDELINE FOR CICLOSPORIN IN DERMATOLOGY 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical, nursing and pharmacy staff in the safe and appropriate prescription and administration
PALIPERIDONE LONG ACTING INJECTION PRESCRIBING GUIDELINE. Chief Pharmacist. Chief Pharmacist
 REFERENCE NUMBER: PALIPERIDONE LONG ACTING INJECTION PRESCRIBING GUIDELINE AREA: NAME OF RESPONSIBLE COMMITTEE / INDIVIDUAL NAME OF ORIGINATOR / AUTHOR Trust-wide Chief Pharmacist Chief Pharmacist DATE
REFERENCE NUMBER: PALIPERIDONE LONG ACTING INJECTION PRESCRIBING GUIDELINE AREA: NAME OF RESPONSIBLE COMMITTEE / INDIVIDUAL NAME OF ORIGINATOR / AUTHOR Trust-wide Chief Pharmacist Chief Pharmacist DATE
CLINICAL GUIDELINE FOR THE MANAGEMENT OF INPATIENTS WITH PARKINSON S DISEASE
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF INPATIENTS WITH PARKINSON S DISEASE 1. Aim/Purpose of this Guideline To assist all doctors and nurses in the care of inpatients with Parkinson s disease. This guideline
CLINICAL GUIDELINE FOR THE MANAGEMENT OF INPATIENTS WITH PARKINSON S DISEASE 1. Aim/Purpose of this Guideline To assist all doctors and nurses in the care of inpatients with Parkinson s disease. This guideline
Melatonin shared care guideline. Document Title. Corporate: Clinical. Type of document. Brief summary of contents
 Document Title Melatonin shared care guideline Type of document Corporate: Clinical Brief summary of contents Executive Director responsible for Policy: Directorate / Department responsible (author/owner):
Document Title Melatonin shared care guideline Type of document Corporate: Clinical Brief summary of contents Executive Director responsible for Policy: Directorate / Department responsible (author/owner):
Pelvic Inflammatory Disease Clinical Guideline V1.0 February 2019
 Pelvic Inflammatory Disease Clinical Guideline V1.0 February 2019 Summary This guideline will provide evidence based guidance on the management of Pelvic Inflammatory Disease A low threshold for empirical
Pelvic Inflammatory Disease Clinical Guideline V1.0 February 2019 Summary This guideline will provide evidence based guidance on the management of Pelvic Inflammatory Disease A low threshold for empirical
Blood Transfusion Policy for Children and Neonates. November 2017
 Blood Transfusion Policy for Children and Neonates V5 November 2017 1. Introduction 3 2. Purpose of this Policy/Procedure 3 3. Scope 3 4. Ownership and Responsibilities 3 4.1. Role of the Managers 3 4.2.
Blood Transfusion Policy for Children and Neonates V5 November 2017 1. Introduction 3 2. Purpose of this Policy/Procedure 3 3. Scope 3 4. Ownership and Responsibilities 3 4.1. Role of the Managers 3 4.2.
The Newcastle upon Tyne Hospitals NHS Foundation Trust. Pre-filled Patient Controlled Analgesia (PCA) syringes
 The Newcastle upon Tyne Hospitals NHS Foundation Trust Pre-filled Patient Controlled Analgesia (PCA) syringes Version.: 2.2 Effective From: 1 June 2016 Expiry Date: 1 June 2019 Date Ratified: 20 April
The Newcastle upon Tyne Hospitals NHS Foundation Trust Pre-filled Patient Controlled Analgesia (PCA) syringes Version.: 2.2 Effective From: 1 June 2016 Expiry Date: 1 June 2019 Date Ratified: 20 April
Admission of a Child/Young Person with Cystic Fibrosis Clinical Guideline V3.0 May 2018
 Admission of a Child/Young Person with Cystic Fibrosis Clinical Guideline V3.0 May 2018 Page 1 of 16 1. Aim/Purpose of this Guideline This guideline applies to all staff caring for children/young people
Admission of a Child/Young Person with Cystic Fibrosis Clinical Guideline V3.0 May 2018 Page 1 of 16 1. Aim/Purpose of this Guideline This guideline applies to all staff caring for children/young people
Chemotherapy Training and Assessment Policy. For Medical Prescribers and Pharmacy Verifiers
 Chemotherapy Training and Assessment Policy For Medical Prescribers and Pharmacy Verifiers For approvals and version control see Document Management Record on page 6 Doc Ref: AngCN-CCG-C36 Approved and
Chemotherapy Training and Assessment Policy For Medical Prescribers and Pharmacy Verifiers For approvals and version control see Document Management Record on page 6 Doc Ref: AngCN-CCG-C36 Approved and
Spirometry Clinical Guideline V2.0. May 2018
 Spirometry Clinical Guideline V2.0 May 2018 Spirometry Clinical Guideline V2.0 1 Summary REFERRAL FOR SPIROMETRY QUALITY ASSURED SPIROMETRY PERFORMED BY TECHNICIAN TRAINED AND ASSESSED AS COMPETENT SPIROMETRY
Spirometry Clinical Guideline V2.0 May 2018 Spirometry Clinical Guideline V2.0 1 Summary REFERRAL FOR SPIROMETRY QUALITY ASSURED SPIROMETRY PERFORMED BY TECHNICIAN TRAINED AND ASSESSED AS COMPETENT SPIROMETRY
Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital
 Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
OBSESSIVE COMPULSIVE DISORDER
 SHARED CARE GUIDELINE FOR SSRIs for the treatment of DEPRESSION, OBSESSIVE COMPULSIVE DISORDER and ANXIETY in CHILDREN and YOUNG PEOPLE 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical,
SHARED CARE GUIDELINE FOR SSRIs for the treatment of DEPRESSION, OBSESSIVE COMPULSIVE DISORDER and ANXIETY in CHILDREN and YOUNG PEOPLE 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical,
VIP (Etoposide, Ifosfamide and Cisplatin)
 VIP (Etoposide, Ifosfamide and Cisplatin) Indication First line treatment for metastatic seminoma, non seminoma or combined tumours where bleomycin is contra-indicated. Usually used for patients with intermediate
VIP (Etoposide, Ifosfamide and Cisplatin) Indication First line treatment for metastatic seminoma, non seminoma or combined tumours where bleomycin is contra-indicated. Usually used for patients with intermediate
Acute Alcohol Withdrawal Protocol
 Acute Alcohol Withdrawal Protocol Controlled document This document is uncontrolled when downloaded or printed Reference number Version 1 Author WHHT: C268 Dr Mohamed Shariff Date ratified August 2014
Acute Alcohol Withdrawal Protocol Controlled document This document is uncontrolled when downloaded or printed Reference number Version 1 Author WHHT: C268 Dr Mohamed Shariff Date ratified August 2014
Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc)
 Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author) Guideline on the management of excessive coumarin anticoagulation in adults
Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author) Guideline on the management of excessive coumarin anticoagulation in adults
DERBY-BURTON LOCAL CANCER NETWORK FILENAME R-IVE.DOC CONTROLLED DOC NO: HCCPG B53 CSIS Regimen Name: R-IVE. R-IVE Regimen
 R-IVE Regimen Available for Routine Use in Burton in-patient Derby in-patient Burton day-case Derby day-case Burton community Derby community Burton out-patient Derby out-patient Indication Relapsed/ refractory
R-IVE Regimen Available for Routine Use in Burton in-patient Derby in-patient Burton day-case Derby day-case Burton community Derby community Burton out-patient Derby out-patient Indication Relapsed/ refractory
The Newcastle upon Tyne Hospitals NHS Foundation Trust. Essential Out-of Hours Cancer Chemotherapy
 The Newcastle upon Tyne Hospitals NHS Foundation Trust Essential Out-of Hours Cancer Chemotherapy Version No.: 4.0 Effective From: 27 December 2017 Expiry Date: 27 December 2020 Date Ratified: 19 July
The Newcastle upon Tyne Hospitals NHS Foundation Trust Essential Out-of Hours Cancer Chemotherapy Version No.: 4.0 Effective From: 27 December 2017 Expiry Date: 27 December 2020 Date Ratified: 19 July
Acutely Painful testes
 2.0 FINAL Guideline adopted from the Bedside Clinical Guideline Partnership EQUALITY IMPACT The Trust strives to ensure equality of opportunity for all both as a major employer and as a provider of health
2.0 FINAL Guideline adopted from the Bedside Clinical Guideline Partnership EQUALITY IMPACT The Trust strives to ensure equality of opportunity for all both as a major employer and as a provider of health
MANAGEMENT OF DIABETES MELLITUS (DM) IN PREGNANCY TYPE 1, TYPE 2 AND GESTATIONAL DIABETES (GDM),CLINICAL GUIDELINE. V1.0
 MANAGEMENT OF DIABETES MELLITUS (DM) IN PREGNANCY TYPE 1, TYPE 2 AND GESTATIONAL DIABETES (GDM),CLINICAL GUIDELINE. V1.0 1 1. Aim/Purpose of this Guideline To provide guidance to Midwives, Obstetricians
MANAGEMENT OF DIABETES MELLITUS (DM) IN PREGNANCY TYPE 1, TYPE 2 AND GESTATIONAL DIABETES (GDM),CLINICAL GUIDELINE. V1.0 1 1. Aim/Purpose of this Guideline To provide guidance to Midwives, Obstetricians
Policy for the use of Cytomegalovirus (CMV) negative blood products
 Policy for the use of Cytomegalovirus (CMV) negative blood products SharePoint Location Clinical Policies and Guidelines SharePoint Index Directory Cancer and Specialist Care Sub Area Haematology and Blood
Policy for the use of Cytomegalovirus (CMV) negative blood products SharePoint Location Clinical Policies and Guidelines SharePoint Index Directory Cancer and Specialist Care Sub Area Haematology and Blood
NO SMOKING POLICY. Organisational
 NO SMOKING POLICY Policy Title State previous title where relevant. State if Policy New or Revised Policy Strand Org, HR, Clinical, H&S, Infection Control, Finance For clinical policies only - state index
NO SMOKING POLICY Policy Title State previous title where relevant. State if Policy New or Revised Policy Strand Org, HR, Clinical, H&S, Infection Control, Finance For clinical policies only - state index
Title Protocol for the Management of Urinary Tract Infections for Adult Females and Children in MIUs and WICs
 Document Control Title Protocol for the Management of Urinary Tract Infections for Adult Females and Children in MIUs and WICs Author Author s job title Professional Lead, Minor Injuries Unit Directorate,
Document Control Title Protocol for the Management of Urinary Tract Infections for Adult Females and Children in MIUs and WICs Author Author s job title Professional Lead, Minor Injuries Unit Directorate,
1. Intrathecal must never be abbreviated to IT on a prescription form True / False
 Appendix 1: Intrathecal SACT Assessment Questions Core General Questions must be completed by all staff. 1. Intrathecal must never be abbreviated to IT on a prescription form 2. Intrathecal SACT must be
Appendix 1: Intrathecal SACT Assessment Questions Core General Questions must be completed by all staff. 1. Intrathecal must never be abbreviated to IT on a prescription form 2. Intrathecal SACT must be
31 December a programme of dates of meetings for Full Council, Policy Page: 241 Committees, and Area Committees for the twelve months commencing 1 May
 Page: 240 Business Services REPORT TO ABERDEENSHIRE COUNCIL 28 SEPTEMBER, 2017 TIMETABLE OF MEETINGS 2018/19 1 Recommendation 1.1 The Council is recommended to approve the timetable of Council, Policy
Page: 240 Business Services REPORT TO ABERDEENSHIRE COUNCIL 28 SEPTEMBER, 2017 TIMETABLE OF MEETINGS 2018/19 1 Recommendation 1.1 The Council is recommended to approve the timetable of Council, Policy
Specialised Services Commissioning Policy. CP29: Bariatric Surgery
 Specialised Services Commissioning Policy CP29: Bariatric Surgery Document Author: Specialist Planner, Cardiothoracic Executive Lead: Director of Planning Approved by: Management Group Issue Date: 12 June
Specialised Services Commissioning Policy CP29: Bariatric Surgery Document Author: Specialist Planner, Cardiothoracic Executive Lead: Director of Planning Approved by: Management Group Issue Date: 12 June
Guideline for the Use of Granulocyte Colony Stimulating Factor (G-CSF) for Adults in Oncology and Haematology
 (G-CSF) for Adults in Oncology and Haematology For Use in: By: Oncology and Haematology Inpatients and Outpatients Oncologists and Haematologists For: Division responsible for document: Key words: Name
(G-CSF) for Adults in Oncology and Haematology For Use in: By: Oncology and Haematology Inpatients and Outpatients Oncologists and Haematologists For: Division responsible for document: Key words: Name
N-Acetylcysteine Dosing Guidelines for Paracetamol Overdose in Adults
 N-Acetylcysteine Dosing Guidelines for Paracetamol Overdose in Adults This guidance does not override the individual responsibility of health professionals to make appropriate decision according to the
N-Acetylcysteine Dosing Guidelines for Paracetamol Overdose in Adults This guidance does not override the individual responsibility of health professionals to make appropriate decision according to the
AROMATHERAPY - CLINICAL GUIDELINE FOR MIDWIVES. January 2018
 AROMATHERAPY - CLINICAL GUIDELINE FOR MIDWIVES V2 January 2018 1. Aim/Purpose of this Guideline 1.1. Aromatherapy is the use of essential oils, which can be administered by topical application via massage,
AROMATHERAPY - CLINICAL GUIDELINE FOR MIDWIVES V2 January 2018 1. Aim/Purpose of this Guideline 1.1. Aromatherapy is the use of essential oils, which can be administered by topical application via massage,
Clinical Guideline for the management of paediatric patients with Diabetes Type 1 & 2 requiring Surgery or General Anaesthetic. V4
 Clinical Guideline for the management of paediatric patients with Diabetes Type 1 & 2 requiring Surgery or General Anaesthetic. V4 Page 1 of 27 Abbreviation Meaning PDSN DKA BG SC IV RCHT HDU PEWS Paediatric
Clinical Guideline for the management of paediatric patients with Diabetes Type 1 & 2 requiring Surgery or General Anaesthetic. V4 Page 1 of 27 Abbreviation Meaning PDSN DKA BG SC IV RCHT HDU PEWS Paediatric
In Hospital Management of Hypoglycaemia in Adults with Diabetes Mellitus
 In Hospital Management of Hypoglycaemia in Adults with Diabetes Mellitus This procedural document supersedes any previous guidelines in relation to this subject: PAT/T 49 v.2 In Hospital Management of
In Hospital Management of Hypoglycaemia in Adults with Diabetes Mellitus This procedural document supersedes any previous guidelines in relation to this subject: PAT/T 49 v.2 In Hospital Management of
Index No: MMG11/1. Version: 1. Date ratified: 12 th November 2013
 Index No: Intravenous fluid prescription in children For previously well children aged one month to 16 years (excluding renal, cardiac, diabetic ketoacidosis and acute burns patients) Version: 1 Date ratified:
Index No: Intravenous fluid prescription in children For previously well children aged one month to 16 years (excluding renal, cardiac, diabetic ketoacidosis and acute burns patients) Version: 1 Date ratified:
Clinical Commissioning Policy Statement: Siklos In Sickle Cell Anaemia. December Reference : NHSCB/B8/2
 Clinical Commissioning Policy Statement: Siklos In Sickle Cell Anaemia December 2012 Reference : NHSCB/B8/2 NHS Commissioning Board Clinical Commissioning Policy Statement: Siklos In Sickle Cell Anaemia
Clinical Commissioning Policy Statement: Siklos In Sickle Cell Anaemia December 2012 Reference : NHSCB/B8/2 NHS Commissioning Board Clinical Commissioning Policy Statement: Siklos In Sickle Cell Anaemia
Policy for the Prevention & Management of Occupational Dermatitis and Latex Allergy in a Healthcare Setting V2.0
 Policy for the Prevention & Management of Occupational Dermatitis and Latex Allergy in a Healthcare Setting V2.0 March 2016 Summary Document Title: Policy for the Prevention and Management of Occupational
Policy for the Prevention & Management of Occupational Dermatitis and Latex Allergy in a Healthcare Setting V2.0 March 2016 Summary Document Title: Policy for the Prevention and Management of Occupational
Pathology Service User Guide Haematology
 Pathology Service User Guide Haematology St Richard s This section of the Pathology Service User Guide includes: Anticoagulant Therapy Information about the Anticoagulant Clinic Low Molecular Weight Heparin
Pathology Service User Guide Haematology St Richard s This section of the Pathology Service User Guide includes: Anticoagulant Therapy Information about the Anticoagulant Clinic Low Molecular Weight Heparin
Olanzapine Long-Acting Injection (Zypadhera ) - Guidelines for Prescribing and Administration (Version 3 May 2015)
 1. Key Points Olanzapine Long-Acting Injection (Zypadhera ) - Guidelines for Prescribing and Administration (Version 3 May 2015) 1.1 Olanzapine long acting injection (LAI) is indicated for the maintenance
1. Key Points Olanzapine Long-Acting Injection (Zypadhera ) - Guidelines for Prescribing and Administration (Version 3 May 2015) 1.1 Olanzapine long acting injection (LAI) is indicated for the maintenance
ANTI NEUTROPHIL CYTOPLASMIC ANTIBODY (ANCA) TESTING GUIDELINES
 ANTI NEUTROPHIL CYTOPLASMIC ANTIBODY (ANCA) TESTING GUIDELINES Version: 1.0 Ratified by: Clinical Biochemistry Senior Staff Group Date ratified: Name of originator/author: Director responsible for implementation:
ANTI NEUTROPHIL CYTOPLASMIC ANTIBODY (ANCA) TESTING GUIDELINES Version: 1.0 Ratified by: Clinical Biochemistry Senior Staff Group Date ratified: Name of originator/author: Director responsible for implementation:
Insert heading depending line on length; please delete delete. length; please delete other cover options once
 Insert heading depending Insert on Interim Insert heading line length; Clinical please Commissioning depending on line on length; please delete delete other Policy: on line line cover Circumcision length;
Insert heading depending Insert on Interim Insert heading line length; Clinical please Commissioning depending on line on length; please delete delete other Policy: on line line cover Circumcision length;
StRs and CT doctors in haematology. September Folinic acid dose modified.
 High dose Methotrexate and folinic acid rescue Full Title of Guideline: Author (include email and role): Division & Speciality: Clinical Guideline Review Date September 2018 GUIDELINE FOR THE USE OF HIGH
High dose Methotrexate and folinic acid rescue Full Title of Guideline: Author (include email and role): Division & Speciality: Clinical Guideline Review Date September 2018 GUIDELINE FOR THE USE OF HIGH
Intravesical immunotherapy (known as BCG therapy): procedure-specific information
 PATIENT INFORMATION Intravesical immunotherapy (known as BCG therapy): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels,
PATIENT INFORMATION Intravesical immunotherapy (known as BCG therapy): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels,
Vincristine Ifosfamide Doxorubicin Etoposide (VIDE) Sarcoma
 Systemic Anti Cancer Treatment Protocol Vincristine Ifosfamide Doxorubicin Etoposide (VIDE) Sarcoma PROTOCOL REF: MPHAVIDE (Version No: 1.0) Approved for use in: Ewings sarcoma Desmoplastic small round
Systemic Anti Cancer Treatment Protocol Vincristine Ifosfamide Doxorubicin Etoposide (VIDE) Sarcoma PROTOCOL REF: MPHAVIDE (Version No: 1.0) Approved for use in: Ewings sarcoma Desmoplastic small round
CLINICAL GUIDELINE FOR THE MANAGEMENT OF IMMUNE THROMBOCYTOPENIA (ITP) IN CHILDREN V3.0
 CLINICAL GUIDELINE FOR THE MANAGEMENT OF IMMUNE THROMBOCYTOPENIA (ITP) IN CHILDREN V3.0 Clinical Guideline Template Page 1 of 16 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical
CLINICAL GUIDELINE FOR THE MANAGEMENT OF IMMUNE THROMBOCYTOPENIA (ITP) IN CHILDREN V3.0 Clinical Guideline Template Page 1 of 16 1. Aim/Purpose of this Guideline 1.1. This guideline applies to medical
LOCAL EQUALITY ADVISORY FORUM (LEAF) A Staffordshire CCGs Equality & Inclusion Group. Terms of Reference
 LOCAL EQUALITY ADVISORY FORUM (LEAF) A Staffordshire CCGs Equality & Inclusion Group Terms of Reference (1) Introduction and Purpose Promoting equality and inclusion is at the heart of our values. We will
LOCAL EQUALITY ADVISORY FORUM (LEAF) A Staffordshire CCGs Equality & Inclusion Group Terms of Reference (1) Introduction and Purpose Promoting equality and inclusion is at the heart of our values. We will
Adult Chest Wall Injury Pathway V2.0 May 2018
 ` Adult Chest Wall Injury Pathway V2.0 May 2018 Page 1 of 22 Summary Adult Chest Wall Injury Pathway Blunt chest wall injury requiring admission. Trauma Call if appropriate. Strongly consider Trauma CT.
` Adult Chest Wall Injury Pathway V2.0 May 2018 Page 1 of 22 Summary Adult Chest Wall Injury Pathway Blunt chest wall injury requiring admission. Trauma Call if appropriate. Strongly consider Trauma CT.
PROCEDURE FOR BLOOD GLUCOSE MONITORING
 PROCEDURE FOR BLOOD GLUCOSE MONITORING First Issued Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote safe and effective blood glucose monitoring using Trust equipment
PROCEDURE FOR BLOOD GLUCOSE MONITORING First Issued Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote safe and effective blood glucose monitoring using Trust equipment
Patient Group Directions Policy
 Patient Group Directions Policy Category: Summary: Equality Analysis undertaken: Valid From: Date of Next Review: Approval Date/ Via: Distribution: Related Documents: Author(s): Further Information: This
Patient Group Directions Policy Category: Summary: Equality Analysis undertaken: Valid From: Date of Next Review: Approval Date/ Via: Distribution: Related Documents: Author(s): Further Information: This
CLINICAL PROTOCOL THE PREVENTION OF FATALITIES FROM MEDICATION LOADING DOSES
 National Patient Safety Alert RRR018 Preventing Fatalities From Medication Loading Doses (November 2010) CP11 CLINICAL PROTOCOL THE PREVENTION OF FATALITIES FROM MEDICATION LOADING DOSES INTRODUCTION The
National Patient Safety Alert RRR018 Preventing Fatalities From Medication Loading Doses (November 2010) CP11 CLINICAL PROTOCOL THE PREVENTION OF FATALITIES FROM MEDICATION LOADING DOSES INTRODUCTION The
