Dual-Path Platform (DPP ) technologies, has overcome. de Oliveira et al. Parasites & Vectors (2015) 8:45 DOI /s
|
|
|
- Rose McDowell
- 5 years ago
- Views:
Transcription
1 de Oliveira et al. Parasites & Vectors (2015) 8:45 DOI /s SHORT REPORT Open Access Multi-antigen print immunoassay (MAPIA)-based evaluation of novel recombinant Leishmania infantum antigens for the serodiagnosis of canine visceral leishmaniasis Isaac Queiroz de Oliveira 1, Rodrigo Araujo Silva 1, Michel Vergne Sucupira 2, Edmilson Domingos da Silva 2, Alexandre Barbosa Reis 3,4, Gabriel Grimaldi Jr 1, Deborah Bittencourt Mothé Fraga 1,3,5 and Patrícia Sampaio Tavares Veras 1,3* Abstract Background: Domestic dogs are the principal reservoir hosts of Leishmania infantum in regions where visceral leishmaniasis (VL) is endemic. Although serologic methods are frequently used for the screening of infected dogs, antibody-based tests require further assessment, due to lack of sensitivity and specificity. In this study, we employed a multi-antigen printing immunoassay (MAPIA) to compare the antibody responses to novel recombinant proteins of L. infantum with the potential for the detection of canine VL. Findings: MAPIA strips were prepared employing 12 recombinant proteins. Antibody reactivity to these antigens was compared using a panel of sera collected from clinically asymptomatic (n = 16) and symptomatic (n = 41) culture-positive animals. Our findings showed that the canine immune response to antigen differs between dogs and depends on infection status. Using this screening assay, when five out of the 12 antigens were combined, an overall 81% detection rate of L. infantum-infected dogs was achieved. Conclusions: We conclude that MAPIA is an effective screening tool to rapidly select multiple antigens of diagnostic utility to be used in a more sensitive point of care diagnostic test such as the Dual-Path Platform (DPP) multiplex test for the rapid detection of infected dogs. Keywords: MAPIA, Recombinant antigens, Leishmaniasis Background Zoonotic visceral leishmaniasis (VL) caused by L. infantum is an important emerging parasitic disease in many regions [1]. In the neotropics, L. infantum transmission to humans occurs as a result of Lutzomyia longipalpis bites [2]. Accordingly, dogs are the major source of L. infantum for humans; thus, early and accurate detection of infected dogs is critical to successfully controling the spread of leishmaniasis [3,4]. Additionally, it is also important to highlight the value of a reliable test to screen * Correspondence: pveras@bahia.fiocruz.br 1 Laboratório de Patologia e Biointervenção, Centro de Pesquisas Gonçalo Moniz, FIOCRUZ, Rua Waldemar Falcão, 121 (Candeal), Salvador, BA, Brazil 3 Instituto de Ciência e Tecnologia de Doenças Tropicais, INCT-DT, Salvador, BA, Brazil Full list of author information is available at the end of the article seronegative dogs before vaccination and to confirm infection before culling of seropositive dogs. Current parasitological diagnostic tests, including microscopic examination and in vitro culturing, offer limited sensitivity with respect to the direct detection of Leishmania. In addition, parasite-specific antibody tests, such as the immunofluorescent-antibody test [IFAT], direct agglutination test [DAT], enzyme-linked immunosorbent assay [ELISA], although widely used to diagnose infection, employ crude antigens derived from wholeparasite extracts and lack the appropriate sensitivity and specificity required for accurate serodiagnosis [5-11]. Recently, the use of recombinant protein-based immunochromatographic testing, such as lateral-flow and Dual-Path Platform (DPP ) technologies, has overcome 2015 de Oliveira et al.; licensee BioMed Central. This is an Open Access article distributed under the terms of the Creative Commons Attribution License ( which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly credited. The Creative Commons Public Domain Dedication waiver ( applies to the data made available in this article, unless otherwise stated.
2 de Oliveira et al. Parasites & Vectors (2015) 8:45 Page 2 of 5 the practical limitations of other serological-based methods in the field [6,11-13]. Previous studies have indicated the promising potential of antigen-based serodiagnostic assays for VL, employing a cocktail of antigens [14] or chimeric proteins [15] that cover a broad spectrum of immunoreactivities [16-19]. Although these tests perform well, they do have limitations; for example, DPP shows high sensitivity (98%) and specificity (96%) towards sera from symptomatic dogs, but shows a low sensitivity of only 47% towards sera from asymptomatic dogs [13]. With this in mind, we used the screening test, multiantigen print immunoassay (MAPIA), to further characterize antibody responses in order to select those L. infantum recombinant proteins with a greater capacity to be utilized for the serodiagnosis of canine visceral leishmaniasis (CVL). MAPIA is more efficient, cost-effective, and reproducible than other screening techniques. In addition, as MAPIA is a membrane-based assay, it can easily be developed into a rapid test that utilizes thin-layer immunochromatography, similar to rapid diagnostic tests for other infectious diseases [20]. This advantage is important because our future goal is to generate a more reliable DPP assay [13], using MAPIA to carefully select multiple antigens for the effective serodiagnosis of L. infantum-infected dogs. Methods Leishmania infantum antigens A set of 12 recombinant L. infantum antigens (rlci1a, rlci2b, rlci3, rlci4, rlci5, rlci6, rlci7, rlci8, rlci10, rlci11, rlci12, rlci13) was previously selected from DNA libraries based on antibody reactivity using sera from culture-positive dogs [21,22]. Histidine-tagged recombinant proteins were produced after sub-cloning DNA fragments as described previously [21]. The antigens were then purified by affinity chromatography using PD-10 Desalting Workmate nickel-sepharose columns (Amersham Pharmacia Biotech AB, Sweden), in accordance with the manufacturer s instructions. Dog sera and infection status A panel of 138 canine sera was used. Negative control sera were obtained from 40 kennel dogs from Pelotas, Rio Grande do Sul (a VL-free area of Brazil). These dogs tested negative for L. infantum via serology, culturing, and qpcr of splenic aspirate [23]. To test for crossreactivity of the 12 recombinant antigens with other pathogens, we also screened sera from dogs infected with Leishmania braziliensis (n = 10), Trypanosoma cruzi (n = 10), Babesia spp. (n = 10), and Ehrlichia canis (n = 11). To determine sensitivity, the antibody reactivity was assessed using a panel of 57 sera from symptomatic (n = 41) and asymptomatic (n = 16) culture-positive dogs. All infected dogs enrolled in the study were selected during epidemiological surveys of CVL carried out in four endemic areas in Brazil: Camaçari, Bahia; Dias D Àvila, Bahia; Jequié, Bahia; and Pancas, Espírito Santo. At the time of sampling, dogs were clinically examined for seven typical signs of CVL and were scored clinically as asymptomatic if they had total scores of 0 to 4 and as symptomatic if they had scores greater than 4 [8]. MAPIA strip preparation Antigens were sprayed onto a 0.45-μm, pore-size nitrocellulose membrane (HiFlow Plus HFB24004, Millipore, MA) in parallel bands via use of a semi-automatic air-brush printing device (CAMAG automatic TLC sample 4, CAMAG, Muttenz, Switzerland) with a volume of 5 μl/ mm. As described by Lyashchenko and collaborators [20], each antigen solution was printed in 15 cm length lines using the concentration of antigen according to solubility in phosphate-buffered saline (PBS): Lci1 = mg/ml, Lci2 = mg/ml, Lci3 = mg/ml, Lci4 = mg/ml, Lci5 = mg/ml, Lci6 = mg/ml, Lci7 = mg/ml, Lci8 = mg/ml, Lci10 = mg/ml, Lci11 = mg/ml, Lci12 = mg/ml, Lci13 = mg/ml. Three additional lines were saturated with L. major lysate = mg/ml, recombinant CRA&FRA T. cruzi proteins = mg/ml, and a protein A solution = mg/ml. The printed nitrocellulose membranes were dried in ambient air and cut into 5-mm strips. Serum incubation and antibody detection Before incubation with test sera, strips were blocked for 1 h in 800 μl of PBS with 0.3% Tween 20 (Calbiochem, La Jolla, CA) and 5% instant nonfat dry milk at room temperature while rocking. Then, the strips were incubated with 1:100 dilution of each serum for 30 min at room temperature while rocking. After being triplewashed with PBS-Tween under agitation for 5 min at 37 C, the strips were incubated with 1 ml of goat antidog IgG antibodies conjugated with peroxidase (1:150 dilution), at room temperature for 1 h, and then washed twice. Enzyme activity was visualized by incubating the strips for 5 min with 1 ml of substrate-chromogenic solution (5 mg of DAB in 19,995 μl of PBS, with 5 μl of hydrogen peroxidase). To stop the reaction, the strips were rinsed extensively in distilled water at room temperature. The strips were then dried and immediately stored in dark conditions until the results were visually read and digitalized. Data analysis Test positivity was evaluated by interpretation of serum antibody reactivity by visual detection of a brown band on the antigen-impregnated nitrocellulose strips. Epi Info 7 (CDC, Atlanta, Georgia - USA) was used to perform combinatorial analysis.
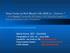
3 de Oliveira et al. Parasites & Vectors (2015) 8:45 Page 3 of 5 Ethical approval for animal use All experiments involving animals were performed in compliance with Brazilian federal law for animal experimentation (Law 11794). In conformity with the Oswaldo Cruz Foundation (FIOCRUZ) animal experimentation guidelines, and according to instructions outlined in the Brazilian Ministry of Health s manual for the surveillance and control of VL. The present study was approved by the Institutional Review Board (CEUA protocol no. 015/ 2009) of the Gonçalo Moniz Research Center in Bahia, Brazil (CPqGM-FIOCRUZ/BA). Finding All MAPIA procedures were optimized with regard to antigen concentrations and serum dilution (data not shown). A total of 138 sera from clinically symptomatic (n = 41) and asymptomatic (n = 16) L. infantum-infected dogs, healthy controls (n = 40) and animals harboring other infections (n = 40) were tested against the selected panel of 12 antigens. The results showed variable antigen recognition patterns among the evaluated serum samples, as indicated in Figure 1. As shown in Table 1, the individual sensitivities of the 12 recombinant proteins coated onto nitrocellulose membranes ranged from 4 to 58% for identifying parasite-positive dogs. Nonetheless, each of the antigens detected some positive sera that others missed. When the individual recombinant proteins were combined, the total sensitivity increased to 81% (Figure 2), revealing that the antigens complemented each other. The well-known heterogeneous humoral immune response that develops in L. infantum-infected dogs [24] likely involves multiple antigens that are differentially recognized by the serum of each animal depending on the state of disease [8]. Therefore, further research into the development of a more reliable rapid test based on the combination of multiple antigens in a DPP format should be pursued. Efforts to develop a diagnostic test based on antibodydetection for VL or CVL have been underway for decades although few of such tests are chromatographic immunoassays currently used in endemic countries [12,13,25]. Prior studies have demonstrated that antibodies against these antigens are mainly detectable in cases with advanced stages of disease, while they are much less detectable in sera from asymptomatic cases [12,13]. Variable multi-antigen recognition by canine serum provides an explanation for the variable performance of commercial tests for detecting infected dogs. Here, we found the recombinant antigens evaluated were more sensitive in symptomatic cases (89%) in comparison to dogs without signs of VL (56%), thus confirming that these antigens are differentially recognized at different stages of infection. Using MAPIA, the Lci2B and Lci1A antigens gave the best results in terms of sensitivity (76% and 71%, respectively) for the detection of dogs with active disease. Although individually, none of these Figure 1 MAPIA with recombinant Leishmania infantum antigens. Images of strips containing printed individual antigens. Strips were incubated with serum samples from L. infantum-positive dogs (lanes 1, 3 13), serum from a normal control (uninfected) dog (lane 2), and the positive standard control serum pool (lane 14). The optimum concentration of each antigen werelci1 = mg/ml, Lci2 = mg/ml, Lci3 = mg/ml, Lci4 = mg/ml, Lci5 = mg/ml, Lci6 = mg/ml, Lci7 = mg/ml, Lci8 = mg/ml, Lci10 = mg/ml, Lci11 = mg/ml, Lci12 = mg/ml, Lci13 = mg/ml, L. major lysate = mg/ml, recombinant CRA&FRA T. cruzi proteins = mg/ml and protein A solution = mg/ml. Antibody reactivity was detected as described in Methods. The test bands were visually read by two independent operators. Any visible band in the test area (in addition to the control line) was considered a positive reaction.
4 de Oliveira et al. Parasites & Vectors (2015) 8:45 Page 4 of 5 Table 1 Sensitivity and specificity of MAPIA with recombinant antigens of Leishmania infantum for the serodiagnosis of canine visceral leishmaniasis Study groups (n) Reactivity with individual L. infantum antigens (number of positive sera) Lci1A Lci2B Lci3 Lci4 Lci5 Lci6 Lci7 Lci8 Lci10 Lci11 Lci12 Lci13 Any antigen Dogs without signs of VL (16) Dogs with signs of VL (41) Total (57) % Sensitivity Normal control (40) Dermal leishmaniasis* (10) Trypanosomiasis* (10) Babesiosis* (10) Ehrlichiosis* (11) Total (81) % Specificity *Sera of dogs affected with other infections. antigens performed well in detecting asymptomatic dogs (sensitivity ranging from 6 to 38%), 46 out of 57 serum samples recognized two groups of combined antigens (namely, rlci1a, rlci2b, rlci8, rlci12 and rlci4 or rlci1a, rlci2b, rlci8, rlci12 and rlci5), thus improving the overall sensitivity of the test to 81%. This result is in agreement with previously published results suggesting that more than one recombinant antigen may be useful to maximize the sensitivity of diagnostic tests for CVL [22,26]. As shown in Table 1, the individual antigens displayed variable specificities (ranging from 86% to 99%). The rlci1a antigen had a specificity of 90% and the rlci2b antigen had a specificity of 91%. These results are in accordance with previous study, which reported specificity values of 92% for rlci1a and 95% for rlci2b [27]. In addition, rlci8, which composes the groups of combined antigens, also showed a very high specificity value (99%). By contrast, the other three antigens, rlci4, rlci12 and rlci5, included in at least one of the two groups of combined antigens, showed lower specificity values of, respectively, 86, 88 and 89%, reducing overall specificity value to 74% (Table 1). MAPIA has previously proven to facilitate rapid screening of antigens, since in comparison to ELISA [20], it is an easy and rapid test that simultaneously detects the response of sera to multiple antigens coated onto a single nitrocellulose strip. Indeed, the serological performance of antigens in MAPIA shown to be a good predictor of their performance in several point-of-care assays [14,15,28]. Here, we reveal MAPIA to be a useful tool for L. infantum antigen selection for the future development of an immunodiagnostic test for CVL. This screening test allowed the selection of two sets of antigens (rlci1a, rlci2b, rlci8, rlci12 and rlci4 or rlci1a, rlci2b, rlci8, rlci12 and rlci5) offering both high sensitivity and specificity; our results illustrate the benefit of utilizing a more effective multi-antigen point-of-care test in DPP format for application in mass assessment surveys of L. infantum-exposed dogs. Competing interests The authors declare that they have no competing interests. Figure 2 Increased diagnostic sensitivity by combining the individual recombinant antigens for detecting clinically symptomatic and asymptomatic Leishmania infantum infected dogs. Authors contributions Conceived and designed the experiments: EDS, GG Jr, DBMF, PSTV. Performed the experiments: IQO, RAS, MVS. Analyzed the data: GG Jr, DBMF, PSTV. Contributed reagents/materials/analysis tools: ABR. All authors read and approved the final version of the manuscript.
5 de Oliveira et al. Parasites & Vectors (2015) 8:45 Page 5 of 5 Acknowledgments This work was supported by INCT-DT, FAPESB, and FIOCRUZ. The authors would like to thank Dr. Flávia W. Cruz McBride for support to obtain negative control samples in Pelotas. We are also grateful to Andris K. Walter for providing English revision and consulting services. In addition, this manuscript has been edited by native English-speaking experts from BioMed Proofreading LLC. Author details 1 Laboratório de Patologia e Biointervenção, Centro de Pesquisas Gonçalo Moniz, FIOCRUZ, Rua Waldemar Falcão, 121 (Candeal), Salvador, BA, Brazil. 2 Laboratório de Tecnologia Diagnóstica, Instituto de Tecnologia em Imunobiológicos, Bio-Manguinhos, FIOCRUZ, Rio de Janeiro, RJ, Brazil. 3 Instituto de Ciência e Tecnologia de Doenças Tropicais, INCT-DT, Salvador, BA, Brazil. 4 Laboratório de Imunopatologia, Núcleo de Pesquisas em Ciências Biológicas, Universidade Federal de Ouro Preto, Ouro Preto, MG, Brazil. 5 Departamento de Medicina Veterinária Preventiva e Produção Animal, Escola de Medicina Veterinária e Zootecnia, Universidade Federal da Bahia, Salvador, BA, Brazil. Received: 8 September 2014 Accepted: 11 January 2015 References 1. Alvar J, Velez ID, Bern C, Herrero M, Desjeux P, Cano J, et al. Leishmaniasis worldwide and global estimates of its incidence. PLoS One. 2012;7(5):e Quinnell RJ, Courtenay O. Transmission, reservoir hosts and control of zoonotic visceral leishmaniasis. Parasitology. 2009;136(14): Ashford DA, David JR, Freire M, David R, Sherlock I, Eulalio MC, et al. Studies on control of visceral leishmaniasis: impact of dog control on canine and human visceral leishmaniasis in Jacobina, Bahia. Brazil Am J Trop Med Hyg. 1998;59(1): Moreira Jr ED, Mendes de Souza VM, Sreenivasan M, Nascimento EG, Pontes de Carvalho L. Assessment of an optimized dog-culling program in the dynamics of canine Leishmania transmission. Vet Parasitol. 2004;122(4): Aisa MJ, Castillejo S, Gallego M, Fisa R, Riera MC, de Colmenares M, et al. Diagnostic potential of Western blot analysis of sera from dogs with leishmaniasis in endemic areas and significance of the pattern. Am J Trop Med Hyg. 1998;58(2): Alvar J, Molina R, San Andres M, Tesouro M, Nieto J, Vitutia M, et al. Canine leishmaniasis: clinical, parasitological and entomological follow-up after chemotherapy. Ann Trop Med Parasitol. 1994;88(4): Falqueto A, Ferreira AL, dos Santos CB, Porrozzi R, da Costa MV, Teva A, et al. Cross-sectional and longitudinal epidemiologic surveys of human and canine Leishmania infantum visceral infections in an endemic rural area of southeast Brazil (Pancas, Espirito Santo). Am J Trop Med Hyg. 2009;80(4): Quinnell RJ, Courtenay O, Davidson S, Garcez L, Lambson B, Ramos P, et al. Detection of Leishmania infantum by PCR, serology and cellular immune response in a cohort study of Brazilian dogs. Parasitology. 2001;122(Pt 3): Reithinger R, Quinnell RJ, Alexander B, Davies CR. Rapid detection of Leishmania infantum infection in dogs: comparative study using an immunochromatographic dipstick test, enzyme-linked immunosorbent assay, and PCR. J Clin Microbiol. 2002;40(7): Dye C, Vidor E, Dereure J. Serological diagnosis of leishmaniasis: on detecting infection as well as disease. Epidemiol Infect. 1993;110(3): Lombardo G, Pennisi MG, Lupo T, Chicharro C, Solano-Gallego L. Papular dermatitis due to Leishmania infantum infection in seventeen dogs: diagnostic features, extent of the infection and treatment outcome. Parasit Vectors. 2014;7: da Costa RT, Franca JC, Mayrink W, Nascimento E, Genaro O, Campos-Neto A. Standardization of a rapid immunochromatographic test with the recombinant antigens K39 and K26 for the diagnosis of canine visceral leishmaniasis. Trans R Soc Trop Med Hyg. 2003;97(6): Grimaldi Jr G, Teva A, Ferreira AL, dos Santos CB, Pinto I, De-Azevedo CT, et al. Evaluation of a novel chromatographic immunoassay based on Dual- Path Platform technology (DPP(R) CVL rapid test) for the serodiagnosis of canine visceral leishmaniasis. Trans R Soc Trop Med Hyg. 2012;106(1): Lyashchenko KP, Greenwald R, Esfandiari J, Chambers MA, Vicente J, Gortazar C, et al. Animal-side serologic assay for rapid detection of Mycobacterium bovis infection in multiple species of free-ranging wildlife. Vet Microbiol. 2008;132(3 4): Lyashchenko KP, Greenwald R, Esfandiari J, Meylan M, Burri IH, Zanolari P. Antibody responses in New World camelids with tuberculosis caused by Mycobacterium microti. Vet Microbiol. 2007;125(3 4): Boarino A, Scalone A, Gradoni L, Ferroglio E, Vitale F, Zanatta R, et al. Development of recombinant chimeric antigen expressing immunodominant B epitopes of Leishmania infantum for serodiagnosis of visceral leishmaniasis. Clin Diagn Lab Immunol. 2005;12(5): Costa MM, Penido M, dos Santos MS, Doro D, de Freitas E, Michalick MS, et al. Improved canine and human visceral leishmaniasis immunodiagnosis using combinations of synthetic peptides in enzyme-linked immunosorbent assay. PLoS Negl Trop Dis. 2012;6(5):e Passos S, Carvalho LP, Orge G, Jeronimo SM, Bezerra G, Soto M, et al. Recombinant Leishmania antigens for serodiagnosis of visceral leishmaniasis. Clin Diagn Lab Immunol. 2005;12(10): Rosati S, Ortoffi M, Profiti M, Mannelli A, Mignone W, Bollo E, et al. Prokaryotic expression and antigenic characterization of three recombinant Leishmania antigens for serological diagnosis of canine leishmaniasis. Clin Diagn Lab Immunol. 2003;10(6): Lyashchenko KP, Singh M, Colangeli R, Gennaro ML. A multi-antigen print immunoassay for the development of serological diagnosis of infectious diseases. J Immunol Methods. 2000;242(1 2): Oliveira GG, Magalhaes FB, Teixeira MC, Pereira AM, Pinheiro CG, Santos LR, et al. Characterization of novel Leishmania infantum recombinant proteins encoded by genes from five families with distinct capacities for serodiagnosis of canine and human visceral leishmaniasis. Am J Trop Med Hyg. 2011;85(6): Teixeira MC, Oliveira GG, Silvany MA, Alcantara-Neves NM, Soares MB, Ribeiro-Dos-Santos R, et al. A strategy for identifying serodiagnostically relevant antigens of Leishmania or other pathogens in genetic libraries. Biologicals. 2007;35(1): Solca Mda S, Guedes CE, Nascimento EG, Oliveira GG, dos Santos WL, Fraga DB, et al. Qualitative and quantitative polymerase chain reaction (PCR) for detection of Leishmania in spleen samples from naturally infected dogs. Vet Parasitol. 2012;184(2 4): Solano-Gallego L, Riera C, Roura X, Iniesta L, Gallego M, Valladares JE, et al. Leishmania infantum-specific IgG, IgG1 and IgG2 antibody responses in healthy and ill dogs from endemic areas. Evolution in the course of infection and after treatment. Vet Parasitol. 2001;96(4): Mettler M, Grimm F, Capelli G, Camp H, Deplazes P. Evaluation of enzymelinked immunosorbent assays, an immunofluorescent-antibody test, and two rapid tests (immunochromatographic-dipstick and gel tests) for serological diagnosis of symptomatic and asymptomatic Leishmania infections in dogs. J Clin Microbiol. 2005;43(11): Lyashchenko KP, Greenwald R, Esfandiari J, Greenwald D, Nacy CA, Gibson S, et al. PrimaTB STAT-PAK assay, a novel, rapid lateral-flow test for tuberculosis in nonhuman primates. Clin Vaccine Immunol. 2007;14(9): de Souza CM, Silva ED, Ano Bom AP, Bastos RC, Nascimento HJ, da Silva Junior JG. Evaluation of an ELISA for canine leishmaniasis immunodiagnostic using recombinant proteins. Parasite Immunol. 2012;34(1): Buddle BM, Wilson T, Denis M, Greenwald R, Esfandiari J, Lyashchenko KP, et al. Sensitivity, specificity, and confounding factors of novel serological tests used for the rapid diagnosis of bovine tuberculosis in farmed red deer (Cervus elaphus). Clin Vaccine Immunol. 2010;17(4): Submit your next manuscript to BioMed Central and take full advantage of: Convenient online submission Thorough peer review No space constraints or color figure charges Immediate publication on acceptance Inclusion in PubMed, CAS, Scopus and Google Scholar Research which is freely available for redistribution Submit your manuscript at
Evaluation of Change in Canine Diagnosis Protocol Adopted by the Visceral Leishmaniasis Control Program in Brazil and a New Proposal for Diagnosis
 Evaluation of Change in Canine Diagnosis Protocol Adopted by the Visceral Leishmaniasis Control Program in Brazil and a New Proposal for Diagnosis Wendel Coura-Vital 1,2,7, Henrique Gama Ker 1,3, Bruno
Evaluation of Change in Canine Diagnosis Protocol Adopted by the Visceral Leishmaniasis Control Program in Brazil and a New Proposal for Diagnosis Wendel Coura-Vital 1,2,7, Henrique Gama Ker 1,3, Bruno
Received 1 November 2006/Returned for modification 4 January 2007/Accepted 8 February 2007
 CLINICAL AND VACCINE IMMUNOLOGY, May 2007, p. 544 548 Vol. 14, No. 5 1556-6811/07/$08.00 0 doi:10.1128/cvi.00420-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Comparative Evaluation
CLINICAL AND VACCINE IMMUNOLOGY, May 2007, p. 544 548 Vol. 14, No. 5 1556-6811/07/$08.00 0 doi:10.1128/cvi.00420-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Comparative Evaluation
Evaluation of rk28 antigen for serodiagnosis of visceral Leishmaniasis in India
 ORIGINAL ARTICLE TROPICAL AND PARASITIC DISEASES Evaluation of rk8 antigen for serodiagnosis of visceral Leishmaniasis in India M. Vaish, A. Bhatia, S. G. Reed, J. Chakravarty and S. Sundar ) Department
ORIGINAL ARTICLE TROPICAL AND PARASITIC DISEASES Evaluation of rk8 antigen for serodiagnosis of visceral Leishmaniasis in India M. Vaish, A. Bhatia, S. G. Reed, J. Chakravarty and S. Sundar ) Department
Serodiagnosis of Canine Babesiosis
 Serodiagnosis of Canine Babesiosis Xuenan XUAN, DVM, PhD National Research Center for Protozoan Diseases Obihiro University of Agriculture and Veterinary Medicine Obihiro, Hokkaido 080-8555, Japan Tel.:+81-155-495648;
Serodiagnosis of Canine Babesiosis Xuenan XUAN, DVM, PhD National Research Center for Protozoan Diseases Obihiro University of Agriculture and Veterinary Medicine Obihiro, Hokkaido 080-8555, Japan Tel.:+81-155-495648;
PUC Minas and Minasfungi do Brasil Ltda, Belo Horizonte, Minas Gerais, Brazil.
 CVI Accepts, published online ahead of print on 3 April 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00023-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 4 5 6 7 8 9 10
CVI Accepts, published online ahead of print on 3 April 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00023-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 4 5 6 7 8 9 10
Development of a Dipstick Assay for Detection of Leishmania-Specific Canine Antibodies
 JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2004, p. 193 197 Vol. 42, No. 1 0095-1137/04/$08.00 0 DOI: 10.1128/JCM.42.1.193 197.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Development
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2004, p. 193 197 Vol. 42, No. 1 0095-1137/04/$08.00 0 DOI: 10.1128/JCM.42.1.193 197.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Development
Evaluating the Accuracy of Molecular Diagnostic Testing for Canine Visceral Leishmaniasis Using Latent Class Analysis
 Evaluating the Accuracy of Molecular Diagnostic Testing for Canine Visceral Leishmaniasis Using Latent Class Analysis Manuela da Silva Solcà 1, Leila Andrade Bastos 1, Carlos Eduardo Sampaio Guedes 1,
Evaluating the Accuracy of Molecular Diagnostic Testing for Canine Visceral Leishmaniasis Using Latent Class Analysis Manuela da Silva Solcà 1, Leila Andrade Bastos 1, Carlos Eduardo Sampaio Guedes 1,
Evaluation of a prototype flow cytometry test for serodiagnosis of canine visceral. leishmaniasis
 CVI Accepts, published online ahead of print on 9 October 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00575-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 Evaluation of
CVI Accepts, published online ahead of print on 9 October 2013 Clin. Vaccine Immunol. doi:10.1128/cvi.00575-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 Evaluation of
Montenegro s skin reactions and antibodies against different Leishmania species in dogs from a visceral leishmaniosis endemic area
 Veterinary Parasitology 139 (2006) 21 28 www.elsevier.com/locate/vetpar Montenegro s skin reactions and antibodies against different Leishmania species in dogs from a visceral leishmaniosis endemic area
Veterinary Parasitology 139 (2006) 21 28 www.elsevier.com/locate/vetpar Montenegro s skin reactions and antibodies against different Leishmania species in dogs from a visceral leishmaniosis endemic area
A Novel Noninvasive Method for Diagnosis of Visceral Leishmaniasis by. rk39 Test in Sputum Samples
 JCM Accepts, published online ahead of print on 0 June 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
JCM Accepts, published online ahead of print on 0 June 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
ORIGINAL ARTICLE. The performance of serological tests for Leishmania infantum infection screening in dogs depends on the prevalence of the disease
 ORIGINAL ARTICLE http://dx.doi.org/10.1590/s1678-9946201759039 The performance of serological tests for Leishmania infantum infection screening in dogs depends on the prevalence of the disease Ivete Lopes
ORIGINAL ARTICLE http://dx.doi.org/10.1590/s1678-9946201759039 The performance of serological tests for Leishmania infantum infection screening in dogs depends on the prevalence of the disease Ivete Lopes
Recommendations for Coping with Leishmaniasis: A Review of Control Strategies. Centro de Convenções de Reboças Red Room 17: 00h
 Recommendations for Coping with Leishmaniasis: A Review of Control Strategies Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000
Recommendations for Coping with Leishmaniasis: A Review of Control Strategies Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000
Follow-up of Leishmania infantum naturally infected dogs treated with allopurinol: immunofluorescence antibody test, ELISA and Western blot
 Acta Tropica 84 (2002) 175/181 www.parasitology-online.com Follow-up of Leishmania infantum naturally infected dogs treated with allopurinol: immunofluorescence antibody test, ELISA and Western blot Francis
Acta Tropica 84 (2002) 175/181 www.parasitology-online.com Follow-up of Leishmania infantum naturally infected dogs treated with allopurinol: immunofluorescence antibody test, ELISA and Western blot Francis
New Insights into Diagnosing Leishmaniasis
 New Insights into Diagnosing Leishmaniasis Eric Zini Snow meeting, 13 March 2009 Climate Variability and Visceral Leishmaniasis in Europe WHO/TDR, Jan. 2008 Late Eighties Maroli et al., Trop Med Int Health
New Insights into Diagnosing Leishmaniasis Eric Zini Snow meeting, 13 March 2009 Climate Variability and Visceral Leishmaniasis in Europe WHO/TDR, Jan. 2008 Late Eighties Maroli et al., Trop Med Int Health
Comparison of serological assays for the diagnosis of canine visceral leishmaniasis in animals presenting different clinical manifestations
 Veterinary Parasitology 146 (2007) 235 241 www.elsevier.com/locate/vetpar Comparison of serological assays for the diagnosis of canine visceral leishmaniasis in animals presenting different clinical manifestations
Veterinary Parasitology 146 (2007) 235 241 www.elsevier.com/locate/vetpar Comparison of serological assays for the diagnosis of canine visceral leishmaniasis in animals presenting different clinical manifestations
INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED BY NATURAL EXPOSURE TO LUTZOMYIA LONGIPALPIS BITES
 Am. J. Trop. Med. Hyg., 72(1), 2005, pp. 94 98 Copyright 2005 by The American Society of Tropical Medicine and Hygiene INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED
Am. J. Trop. Med. Hyg., 72(1), 2005, pp. 94 98 Copyright 2005 by The American Society of Tropical Medicine and Hygiene INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED
The Application of Latent Class Analysis for Diagnostic Test Validation of Chronic Trypanosoma cruzi Infection in Blood Donors
 BJID 2002; 6 (August) 181 The Application of Latent Class Analysis for Diagnostic Test Validation of Chronic Trypanosoma cruzi Infection in Blood Donors Dante M. Langhi Junior, José O. Bordin, Adauto Castelo,
BJID 2002; 6 (August) 181 The Application of Latent Class Analysis for Diagnostic Test Validation of Chronic Trypanosoma cruzi Infection in Blood Donors Dante M. Langhi Junior, José O. Bordin, Adauto Castelo,
CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças Red Room 17: 00h
 CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000 deaths (6.7%) 310 million at
CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000 deaths (6.7%) 310 million at
Canine leishmaniosis new concepts and insights on an expanding zoonosis: part two
 Review Canine leishmaniosis new concepts and insights on an expanding zoonosis: part two Guadalupe Miró 1*, Luis Cardoso 2*, Maria Grazia Pennisi 3*, Gaetano Oliva 4* and Gad Baneth 5* 1 Departmento de
Review Canine leishmaniosis new concepts and insights on an expanding zoonosis: part two Guadalupe Miró 1*, Luis Cardoso 2*, Maria Grazia Pennisi 3*, Gaetano Oliva 4* and Gad Baneth 5* 1 Departmento de
Comparative evaluation of several methods for Canine Visceral Leishmaniasis diagnosis
 Braz. J. Vet. Parasitol., Jaboticabal, v. 23, n. 2, p. 179-186, abr.-jun. 2014 ISSN 0103-846X (Print) / ISSN 1984-2961 (Electronic) Doi: http://dx.doi.org/10.1590/s1984-29612014033 Original Article Comparative
Braz. J. Vet. Parasitol., Jaboticabal, v. 23, n. 2, p. 179-186, abr.-jun. 2014 ISSN 0103-846X (Print) / ISSN 1984-2961 (Electronic) Doi: http://dx.doi.org/10.1590/s1984-29612014033 Original Article Comparative
Serological tests fail to discriminate dogs with visceral leishmaniasis that transmit Leishmania infantum to the vector Lutzomyia longipalpis
 Rev Soc Bras Med Trop 50(4):483-488, July-August, 2017 doi: 10.1590/0037-8682-0014-2017 Major Article Serological tests fail to discriminate dogs with visceral leishmaniasis that transmit Leishmania infantum
Rev Soc Bras Med Trop 50(4):483-488, July-August, 2017 doi: 10.1590/0037-8682-0014-2017 Major Article Serological tests fail to discriminate dogs with visceral leishmaniasis that transmit Leishmania infantum
Use of PCR RFLP to identify Leishmania species in naturally-infected dogs
 Veterinary Parasitology 140 (2006) 231 238 www.elsevier.com/locate/vetpar Use of PCR RFLP to identify Leishmania species in naturally-infected dogs Hélida Monteiro de Andrade a, *, Alexandre Barbosa Reis
Veterinary Parasitology 140 (2006) 231 238 www.elsevier.com/locate/vetpar Use of PCR RFLP to identify Leishmania species in naturally-infected dogs Hélida Monteiro de Andrade a, *, Alexandre Barbosa Reis
Utility of Western Blot Analysis for the Diagnosis of Cutaneous Leishmaniasis
 Iran J Parasitol: Vol. 1, No. 4, Oct -Dec 215, pp.599-64 Iran J Parasitol Tehran University of Medical Sciences Publication http:// tums.ac.ir Open access Journal at http:// ijpa.tums.ac.ir Iranian Society
Iran J Parasitol: Vol. 1, No. 4, Oct -Dec 215, pp.599-64 Iran J Parasitol Tehran University of Medical Sciences Publication http:// tums.ac.ir Open access Journal at http:// ijpa.tums.ac.ir Iranian Society
Received 24 March 2003/Returned for modification 1 May 2003/Accepted 8 July 2003
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2003, p. 826 830 Vol. 10, No. 5 1071-412X/03/$08.00 0 DOI: 10.1128/CDLI.10.5.826 830.2003 Copyright 2003, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2003, p. 826 830 Vol. 10, No. 5 1071-412X/03/$08.00 0 DOI: 10.1128/CDLI.10.5.826 830.2003 Copyright 2003, American Society for Microbiology. All Rights
SCHISTOSOMIASIS MANSONI: IMMUNOBLOT ANALYSIS TO DIAGNOSE AND DIFFERENTIATE RECENT AND CHRONIC INFECTION
 Am. J. Trop. Med. Hyg., 61(2), 1999, pp. 302307 Copyright 1999 by The American Society of Tropical Medicine and Hygiene SCHISTOSOMIASIS MANSONI: IMMUNOBLOT ANALYSIS TO DIAGNOSE AND DIFFERENTIATE RECENT
Am. J. Trop. Med. Hyg., 61(2), 1999, pp. 302307 Copyright 1999 by The American Society of Tropical Medicine and Hygiene SCHISTOSOMIASIS MANSONI: IMMUNOBLOT ANALYSIS TO DIAGNOSE AND DIFFERENTIATE RECENT
SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World
 MIT OpenCourseWare http://ocw.mit.edu SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World Spring 2009 For information about citing these materials or our
MIT OpenCourseWare http://ocw.mit.edu SP.718 Special Topics at Edgerton Center: D-Lab Health: Medical Technologies for the Developing World Spring 2009 For information about citing these materials or our
Diagnostic Techniques To Detect Cryptic Leishmaniasis in Dogs
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1137 1141 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1137 1141.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1137 1141 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1137 1141.2002 Copyright 2002, American Society for Microbiology. All Rights
Eliza Yoshie do Rosário, Odair Genaro, João C França-Silva, Roberto T da Costa, Wilson Mayrink, Alexandre Barbosa Reis*, Mariângela Carneiro/ +
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 100(2): 197-203, April 2005 197 Evaluation of enzyme-linked immunosorbent assay using crude Leishmania and recombinant antigens as a diagnostic marker for canine
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 100(2): 197-203, April 2005 197 Evaluation of enzyme-linked immunosorbent assay using crude Leishmania and recombinant antigens as a diagnostic marker for canine
Introduction. In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology.
 Introduction In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology. - Genomics: fasten antigen discovery for complex pathogens
Introduction In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology. - Genomics: fasten antigen discovery for complex pathogens
Belo Horizonte, Minas Gerais, Brazil b Laboratório de Parasitologia e Histopatologia, Núcleo de Pesquisas em Ciências Biológicas/NUPEB;
 Veterinary Immunology and Immunopathology 112 (2006) 102 116 www.elsevier.com/locate/vetimm Isotype patterns of immunoglobulins: Hallmarks for clinical status and tissue parasite density in brazilian dogs
Veterinary Immunology and Immunopathology 112 (2006) 102 116 www.elsevier.com/locate/vetimm Isotype patterns of immunoglobulins: Hallmarks for clinical status and tissue parasite density in brazilian dogs
IVD. DRG Toxocara canis ELISA (EIA-3518) Revised 12 Feb rm (Vers. 6.1)
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
E. Histolytica IgG ELISA Kit
 E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Introduction Leishmaniasis is a group of zoonotic infections caused by protozoan parasites of the genus Leishmania; an estimated persons are
 Sero-epidemological Survey on Canine Visceral Leishmaniasis and the Distribution of Sandfly Vectors in Northwestern Turkey: Prevention Strategies for Childhood Visceral Leishmaniasis by Nihal Dogan, a
Sero-epidemological Survey on Canine Visceral Leishmaniasis and the Distribution of Sandfly Vectors in Northwestern Turkey: Prevention Strategies for Childhood Visceral Leishmaniasis by Nihal Dogan, a
Major Article. CD8+ T cells in situ in different clinical forms of human cutaneous leishmaniasis
 Major Article Revista da Sociedade Brasileira de Medicina Tropical 46(6):728-734, Nov-Dec, 213 http://dx.doi.org/1.159/37-8682-174-213 CD8+ T cells in situ in different clinical forms of human cutaneous
Major Article Revista da Sociedade Brasileira de Medicina Tropical 46(6):728-734, Nov-Dec, 213 http://dx.doi.org/1.159/37-8682-174-213 CD8+ T cells in situ in different clinical forms of human cutaneous
LEISHMANIA IFA SLIDE STORAGE REQUIREMENTS:
 1 STORAGE REQUIREMTS: SLEDO: Slides for the diagnosis of Leishmania infantum antibodies in human serum by indirect immunofluorescent assay (IFA). INTRODUCTION: Leishmania infantum is a causal agent of
1 STORAGE REQUIREMTS: SLEDO: Slides for the diagnosis of Leishmania infantum antibodies in human serum by indirect immunofluorescent assay (IFA). INTRODUCTION: Leishmania infantum is a causal agent of
Leishmania Canine IgG -ELISA
 Leishmania Canine IgG -ELISA GWB-521229 www.genwaybio.com FOR RESEARCH USE ONLY Enzyme immunoassay for the qualitative detection of IgG-class antibodies against Leishmania in canine serum 1. INTRODUCTION
Leishmania Canine IgG -ELISA GWB-521229 www.genwaybio.com FOR RESEARCH USE ONLY Enzyme immunoassay for the qualitative detection of IgG-class antibodies against Leishmania in canine serum 1. INTRODUCTION
The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters.
 Effectiveness of Insecticide Spraying and Culling of Dogs on the Incidence of Leishmania infantum Infection in Humans: A Cluster Randomized Trial in Teresina, Brazil The Harvard community has made this
Effectiveness of Insecticide Spraying and Culling of Dogs on the Incidence of Leishmania infantum Infection in Humans: A Cluster Randomized Trial in Teresina, Brazil The Harvard community has made this
EVALUATION OF AN ENZYME LINKED IMMUNOSORBENT ASSAY-KIT FOR THE DETECTION OF BABESIA BOVIS-ANTIBODIES IN CATTLE IN ARGENTINA
 EVALUATION OF AN ENZYME LINKED IMMUNOSORBENT ASSAY-KIT FOR THE DETECTION OF BABESIA BOVIS-ANTIBODIES IN CATTLE IN ARGENTINA S.T. DE ECHAIDE*, I.E. ECHAIDE*, A.B. GAIDO**, A.J. MANGOLD*, C.I. LUGARESI*,
EVALUATION OF AN ENZYME LINKED IMMUNOSORBENT ASSAY-KIT FOR THE DETECTION OF BABESIA BOVIS-ANTIBODIES IN CATTLE IN ARGENTINA S.T. DE ECHAIDE*, I.E. ECHAIDE*, A.B. GAIDO**, A.J. MANGOLD*, C.I. LUGARESI*,
Toxocara. Cat # Toxocara ELISA. Enzyme Linked Immunosorbent Assay. ELISA-Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Protein MultiColor Stable, Low Range
 Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Leishmania major-like Antigen for Specific and Sensitive Serodiagnosis of Human and Canine Visceral Leishmaniasis
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 2002, p. 1361 1366 Vol. 9, No. 6 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.6.1361 1366.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 2002, p. 1361 1366 Vol. 9, No. 6 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.6.1361 1366.2002 Copyright 2002, American Society for Microbiology. All Rights
Chagas Disease in the Americas: Impacts and Opportunities for Control
 Chagas Disease in the Americas: Impacts and Opportunities for Control Rick Tarleton Center for Tropical and Emerging Global Diseases University of Georgia The Chagas Disease Foundation Trypanosoma cruzi
Chagas Disease in the Americas: Impacts and Opportunities for Control Rick Tarleton Center for Tropical and Emerging Global Diseases University of Georgia The Chagas Disease Foundation Trypanosoma cruzi
Mouse C-peptide EIA. Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY
 Mouse C-peptide EIA Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY TOYO 2CHOME, KOTO-KU, TOKYO, 135-0016, JAPAN http://www.cosmobio.co.jp e-mail : export@cosmobio.co.jp Phone : +81-3-5632-9617 FAX : +81-3-5632-9618
Mouse C-peptide EIA Cat. No. YII-YK013-EX FOR LABORATORY USE ONLY TOYO 2CHOME, KOTO-KU, TOKYO, 135-0016, JAPAN http://www.cosmobio.co.jp e-mail : export@cosmobio.co.jp Phone : +81-3-5632-9617 FAX : +81-3-5632-9618
Canine C-Reactive Protein ELISA
 Canine C-Reactive Protein ELISA Product Data Sheet Cat. No.: RH931CRP01DCR For Research Use Only Page 1 of 12 VERSION 51 020611 46 CONTENTS 1. INTENDED USE 3 2. PRINCIPLE OF ASSAY 3 3. REAGENTS AND MATERIALS
Canine C-Reactive Protein ELISA Product Data Sheet Cat. No.: RH931CRP01DCR For Research Use Only Page 1 of 12 VERSION 51 020611 46 CONTENTS 1. INTENDED USE 3 2. PRINCIPLE OF ASSAY 3 3. REAGENTS AND MATERIALS
Iranian J Parasitol: Vol.2, No.2, 2007, pp
 Original Article Preparation of a K39sub Recombinant Antigen for the Detection of Leishmania infantum Antibodies in Human: a Comparative Study with an Immunochromatographic Test and Direct Agglutination
Original Article Preparation of a K39sub Recombinant Antigen for the Detection of Leishmania infantum Antibodies in Human: a Comparative Study with an Immunochromatographic Test and Direct Agglutination
Mycoplasma pneumoniae IgG ELISA Kit
 Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Documentation, Codebook, and Frequencies
 2 Documentation, Codebook, and Frequencies Laboratory Component: Hepatitis B: core antibody, surface antibody and surface antigen; Hepatitis C: confirmed antibody; Hepatitis D antibody Survey Years: 2003
2 Documentation, Codebook, and Frequencies Laboratory Component: Hepatitis B: core antibody, surface antibody and surface antigen; Hepatitis C: confirmed antibody; Hepatitis D antibody Survey Years: 2003
Min Levine, Ph. D. Influenza Division US Centers for Disease Control and Prevention. June 18, 2015 NIBSC
 Workshop on Immunoassay Standardization for Universal Flu Vaccines Min Levine, Ph. D. Influenza Division US Centers for Disease Control and Prevention June 18, 2015 NIBSC 1 Multiple Immune Mechanisms Contribute
Workshop on Immunoassay Standardization for Universal Flu Vaccines Min Levine, Ph. D. Influenza Division US Centers for Disease Control and Prevention June 18, 2015 NIBSC 1 Multiple Immune Mechanisms Contribute
Division of Global Epidemiology, Research Center for Zoonosis Control. Conduct of ELISA test sample collection for the research study in Japan
 (Abroad)Official trip report form(student) 13/07/29 Name Laboratory Year (Grade) Destination Marvin Ardeza Villanueva Division of Global Epidemiology, Research Center for Zoonosis Control First Year PhD
(Abroad)Official trip report form(student) 13/07/29 Name Laboratory Year (Grade) Destination Marvin Ardeza Villanueva Division of Global Epidemiology, Research Center for Zoonosis Control First Year PhD
HIV Diagnostic Testing
 In The name of God HIV Diagnostic Testing By : Dr. Shahzamani PhD of Medical virology Purpose of HIV Testing To identify asymptomatic individuals To diagnose HIV infection in those who practice high risk
In The name of God HIV Diagnostic Testing By : Dr. Shahzamani PhD of Medical virology Purpose of HIV Testing To identify asymptomatic individuals To diagnose HIV infection in those who practice high risk
Herpes Simplex Virus 2 IgG HSV 2 IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Module 3: Overview of HIV Testing Technologies
 Module 3: Overview of HIV Testing Technologies Purpose Pre-requisite Modules Module Time Learning Objectives To provide the participants with a basic knowledge of HIV testing and how HIV rapid test results
Module 3: Overview of HIV Testing Technologies Purpose Pre-requisite Modules Module Time Learning Objectives To provide the participants with a basic knowledge of HIV testing and how HIV rapid test results
MALARIA P. FALCIPARUM / P. VIVAX
 MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
Leishmania Antigen ELISA (VL ELISA)
 Leishmania Antigen ELISA (VL ELISA) A double antibody sandwich enzyme immunoassay for the detection of the Visceral Leishmaniasis antigen in human urine. For Research Use Only Catalogue Number E12-060
Leishmania Antigen ELISA (VL ELISA) A double antibody sandwich enzyme immunoassay for the detection of the Visceral Leishmaniasis antigen in human urine. For Research Use Only Catalogue Number E12-060
Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test
 Case Reports in Medicine Volume 2011, Article ID 631079, 4 pages doi:10.1155/2011/631079 Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test Paulo R. Machado, 1, 2 Augusto M.
Case Reports in Medicine Volume 2011, Article ID 631079, 4 pages doi:10.1155/2011/631079 Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test Paulo R. Machado, 1, 2 Augusto M.
Title:Evaluating HBsAg rapid tests performance in different biological samples from low- and high infection rates settings & populations
 Author's response to reviews Title:Evaluating HBsAg rapid tests performance in different biological samples from low- and high infection rates settings & populations Authors: Helena M Cruz (h.medina@ioc.fiocruz.br)
Author's response to reviews Title:Evaluating HBsAg rapid tests performance in different biological samples from low- and high infection rates settings & populations Authors: Helena M Cruz (h.medina@ioc.fiocruz.br)
This is a repository copy of Detection of Leishmania infantum by PCR, serology and cellular immune response in a cohort study of Brazilian dogs.
 This is a repository copy of Detection of Leishmania infantum by PCR, serology and cellular immune response in a cohort study of Brazilian dogs. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/1262/
This is a repository copy of Detection of Leishmania infantum by PCR, serology and cellular immune response in a cohort study of Brazilian dogs. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/1262/
Tropical medicine rounds
 Oxford, IJD International 0011-9059 Blackwell 41 UK Science, Journal Ltd 2002 of Dermatology Tropical medicine rounds Cutaneous André Báfica, Leishmaniasis Fabiano Oliveira, Luiz A. R. Freitas, Eliane
Oxford, IJD International 0011-9059 Blackwell 41 UK Science, Journal Ltd 2002 of Dermatology Tropical medicine rounds Cutaneous André Báfica, Leishmaniasis Fabiano Oliveira, Luiz A. R. Freitas, Eliane
Leishmania (Viannia) braziliensis in dogs in Brazil: epidemiology, co-infection, and clinical aspects
 in Brazil: epidemiology, co-infection, and clinical aspects F.S. Carvalho 1, A.A. Wenceslau 2, G.R. Albuquerque 2, A.D. Munhoz 2, E. Gross 2, P.L.S. Carneiro 3, H.C. Oliveira 4, J.M. Rocha 4, I.A. Santos
in Brazil: epidemiology, co-infection, and clinical aspects F.S. Carvalho 1, A.A. Wenceslau 2, G.R. Albuquerque 2, A.D. Munhoz 2, E. Gross 2, P.L.S. Carneiro 3, H.C. Oliveira 4, J.M. Rocha 4, I.A. Santos
Please use only the valid version of the package insert provided with the kit.
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
CONTENTS. STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1
 CONTENTS STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1 ELISA protocol for mite (Dermatophagoides spp.) Group 2 ALLERGENS RESULTS (SUMMARY) TABLE
CONTENTS STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1 ELISA protocol for mite (Dermatophagoides spp.) Group 2 ALLERGENS RESULTS (SUMMARY) TABLE
SOMAPLEX REVERSE PHASE PROTEIN MICROARRAY HUMAN KIDNEY TUMOR & NORMAL TISSUE
 SOMAPLEX REVERSE PHASE PROTEIN MICROARRAY HUMAN KIDNEY TUMOR & NORMAL TISSUE 45 CLINICAL CASES SERIAL DILUTION MULTIPLE PROTEIN CONCENTRATION QUANTITATIVE ASSAY PRODUCT NUMBER: PM1-001-N SOMAPLEX REVERSE
SOMAPLEX REVERSE PHASE PROTEIN MICROARRAY HUMAN KIDNEY TUMOR & NORMAL TISSUE 45 CLINICAL CASES SERIAL DILUTION MULTIPLE PROTEIN CONCENTRATION QUANTITATIVE ASSAY PRODUCT NUMBER: PM1-001-N SOMAPLEX REVERSE
Leishmaniasis Direct Agglutination Test: Using Pictorials as Training Materials to Reduce Inter-Reader Variability and Improve Accuracy
 Leishmaniasis Direct Agglutination Test: Using Pictorials as Training Materials to Reduce Inter-Reader Variability and Improve Accuracy Emily R. Adams 1 *, Diane Jacquet 2, Gerard Schoone 1, Kamlesh Gidwani
Leishmaniasis Direct Agglutination Test: Using Pictorials as Training Materials to Reduce Inter-Reader Variability and Improve Accuracy Emily R. Adams 1 *, Diane Jacquet 2, Gerard Schoone 1, Kamlesh Gidwani
Anti-Lamin B1/LMNB1 Picoband Antibody
 Anti-Lamin B1/LMNB1 Picoband Antibody Catalog Number:PB9611 About LMNB1 Lamin-B1 is a protein that in humans is encoded by the LMNB1 gene. The nuclear lamina consists of a two-dimensional matrix of proteins
Anti-Lamin B1/LMNB1 Picoband Antibody Catalog Number:PB9611 About LMNB1 Lamin-B1 is a protein that in humans is encoded by the LMNB1 gene. The nuclear lamina consists of a two-dimensional matrix of proteins
Human HBcAb IgM ELISA kit
 Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
HIV-1 p24 Antigen ELISA Catalog Number:
 INTENDED USE The RETRO-TEK HIV-1 p24 Antigen ELISA is supplied for research purposes only. It is not intended for use in the diagnosis or prognosis of disease, or for screening and may not be used as a
INTENDED USE The RETRO-TEK HIV-1 p24 Antigen ELISA is supplied for research purposes only. It is not intended for use in the diagnosis or prognosis of disease, or for screening and may not be used as a
Identification of Microbes Lecture: 12
 Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet
 Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Key words: malaria - Plasmodium vivax - merozoite antigens - IgG antibody response MATERIALS AND METHODS
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 102(3): 335-339, June 2007 335 Comparative recognition by human IgG antibodies of recombinant proteins representing three asexual erythrocytic stage vaccine
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 102(3): 335-339, June 2007 335 Comparative recognition by human IgG antibodies of recombinant proteins representing three asexual erythrocytic stage vaccine
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007 Hosted by: Australian Small Animal Veterinary Association (ASAVA) Australian Small Animal Veterinary Association (ASAVA)
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007 Hosted by: Australian Small Animal Veterinary Association (ASAVA) Australian Small Animal Veterinary Association (ASAVA)
Health Systems Research at Imperial College London
 Health Systems Research at Imperial College London CONFAP/MRC Workshop Brasília Dr Thomas Hone Research Fellow Department of Primary Care and Public Health About us Department of Primary Care and Public
Health Systems Research at Imperial College London CONFAP/MRC Workshop Brasília Dr Thomas Hone Research Fellow Department of Primary Care and Public Health About us Department of Primary Care and Public
Trichinella Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
SIV p27 Antigen ELISA Catalog Number:
 INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
Parasite density and impaired biochemical/hematological status are associated with severe clinical aspects of canine visceral leishmaniasis
 Research in Veterinary Science 81 (2006) 68 75 www.elsevier.com/locate/rvsc Parasite density and impaired biochemical/hematological status are associated with severe clinical aspects of canine visceral
Research in Veterinary Science 81 (2006) 68 75 www.elsevier.com/locate/rvsc Parasite density and impaired biochemical/hematological status are associated with severe clinical aspects of canine visceral
Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis!
 Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis! Marie Kroun, MD Denmark Presentation in York, UK June
Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis! Marie Kroun, MD Denmark Presentation in York, UK June
Visceral leishmaniasis: an endemic disease with global impact
 Visceral leishmaniasis: an endemic disease with global impact Professor Olivier Lortholary, MD, PhD Department of Infectious and Tropical diseases Hôpital Necker-Enfants Malades Université Paris Descartes
Visceral leishmaniasis: an endemic disease with global impact Professor Olivier Lortholary, MD, PhD Department of Infectious and Tropical diseases Hôpital Necker-Enfants Malades Université Paris Descartes
DIAGNOSTIC POTENTIAL OF WESTERN BLOT ANALYSIS OF SERA FROM DOGS WITH LEISHMANIASIS IN ENDEMIC AREAS AND SIGNIFICANCE OF THE PATTERN
 Am. J. Trop. Med. Hyg., 58(2), 1998, pp. 154 159 Copyright 1998 by The American Society of Tropical Medicine and Hygiene DIAGNOSTIC POTENTIAL OF WESTERN BLOT ANALYSIS OF SERA FROM DOGS WITH LEISHMANIASIS
Am. J. Trop. Med. Hyg., 58(2), 1998, pp. 154 159 Copyright 1998 by The American Society of Tropical Medicine and Hygiene DIAGNOSTIC POTENTIAL OF WESTERN BLOT ANALYSIS OF SERA FROM DOGS WITH LEISHMANIASIS
IgG antibody responses in early experimental sparganosis and IgG subclass responses in human sparganosis
 145 The Korean Journal of Parasitology Vol. 38, No. 3, 145-150, September 2000 IgG antibody responses in early experimental sparganosis and IgG subclass responses in human sparganosis Young Bae CHUNG 2),
145 The Korean Journal of Parasitology Vol. 38, No. 3, 145-150, September 2000 IgG antibody responses in early experimental sparganosis and IgG subclass responses in human sparganosis Young Bae CHUNG 2),
ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS
 Molecular Diagnostics Symposium 2014 Zurich, 27 February 2014 ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS Stijn Deborggraeve Parasite Diagnostics Unit, Institute of Tropical Medicine
Molecular Diagnostics Symposium 2014 Zurich, 27 February 2014 ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS Stijn Deborggraeve Parasite Diagnostics Unit, Institute of Tropical Medicine
Subclinical Form of the American Visceral Leishmaniasis
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 99(8): 889-893, December 2004 889 Subclinical Form of the American Visceral Leishmaniasis Mônica Elinor Alves Gama/ +, Jackson Maurício Lopes Costa*, Cláudia
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 99(8): 889-893, December 2004 889 Subclinical Form of the American Visceral Leishmaniasis Mônica Elinor Alves Gama/ +, Jackson Maurício Lopes Costa*, Cláudia
FOR LABORATORY USE ONLY
 YK060 Insulin ELISA FOR LABORATORY USE ONLY Kasumigaseki Place, 3-6-7, Kasumigaseki, Chiyoda-ku Tokyo 100-0013 Japan URL: http://www.sceti.co.jp/export/ e-mail: medical@sceti.co.jp Contents
YK060 Insulin ELISA FOR LABORATORY USE ONLY Kasumigaseki Place, 3-6-7, Kasumigaseki, Chiyoda-ku Tokyo 100-0013 Japan URL: http://www.sceti.co.jp/export/ e-mail: medical@sceti.co.jp Contents
A Leishmania Infantum FML-ELISA for the Detection of Symptomatic and Asymptomatic Canine Visceral Leishmaniasis in an Endemic Area of Iran
 ISSN 1735-1383 Iran. J. Immunol. December 2011, 8 (4), 244-250 Behnam Mohammadi-Ghalehbin, Gholam Reza Hatam, Bahador Sarkari, Mehdi Mohebali, Zabih Zarei, Mansoureh Jaberipour, Shahab Bohlouli A Leishmania
ISSN 1735-1383 Iran. J. Immunol. December 2011, 8 (4), 244-250 Behnam Mohammadi-Ghalehbin, Gholam Reza Hatam, Bahador Sarkari, Mehdi Mohebali, Zabih Zarei, Mansoureh Jaberipour, Shahab Bohlouli A Leishmania
Relationship between incident types and impact on patients in drug name errors: a correlational study
 Tsuji et al. Journal of Pharmaceutical Health Care and Sciences (2015) 1:11 DOI 10.1186/s40780-015-0011-x RESEARCH ARTICLE Open Access Relationship between incident types and impact on patients in drug
Tsuji et al. Journal of Pharmaceutical Health Care and Sciences (2015) 1:11 DOI 10.1186/s40780-015-0011-x RESEARCH ARTICLE Open Access Relationship between incident types and impact on patients in drug
Comparison of different diagnostic tests in dogs uninfected and naturally infected with visceral leishmaniasis
 The Journal of Venomous Animals and Toxins including Tropical Diseases ISSN 1678-9199 2011 volume 17 issue 3 pages 348-352 Short Communication Comparison of different diagnostic tests in dogs uninfected
The Journal of Venomous Animals and Toxins including Tropical Diseases ISSN 1678-9199 2011 volume 17 issue 3 pages 348-352 Short Communication Comparison of different diagnostic tests in dogs uninfected
Human Apolipoprotein A1 EIA Kit
 A helping hand for your research Product Manual Human Apolipoprotein A1 EIA Kit Catalog Number: 83901 96 assays 1 Table of Content Product Description 3 Assay Principle 3 Kit Components 3 Storage 4 Reagent
A helping hand for your research Product Manual Human Apolipoprotein A1 EIA Kit Catalog Number: 83901 96 assays 1 Table of Content Product Description 3 Assay Principle 3 Kit Components 3 Storage 4 Reagent
WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT. Product: Murex HIV Ag/Ab Combination Number: PQDx
 WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT Product: Murex HIV Ag/Ab Combination Number: PQDx 0144-043-00 Abstract Murex HIV Ag/Ab Combination with product codes 7G79-09 (GE41,
WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT Product: Murex HIV Ag/Ab Combination Number: PQDx 0144-043-00 Abstract Murex HIV Ag/Ab Combination with product codes 7G79-09 (GE41,
Detection of Multiple Reactive Protein Species by Immunoblotting after Recombinant Outer Surface Protein A Lyme Disease Vaccination
 42 Detection of Multiple Reactive Protein Species by Immunoblotting after Recombinant Outer Surface Protein A Lyme Disease Vaccination Philip J. Molloy, 1,2 Victor P. Berardi, 2 David H. Persing, 2,3,a
42 Detection of Multiple Reactive Protein Species by Immunoblotting after Recombinant Outer Surface Protein A Lyme Disease Vaccination Philip J. Molloy, 1,2 Victor P. Berardi, 2 David H. Persing, 2,3,a
CDIA TM Rubella IgG/IgM Rapid Test Kit
 CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
The Challenges in Developing and Commercializing HIV Tests that are Useful in Differentiating VISP/R VISP/R Workshop Bethesda, MD March 2013
 The Challenges in Developing and Commercializing HIV Tests that are Useful in Differentiating VISP/R VISP/R Workshop Bethesda, MD March 2013 Christopher Bentsen, M.S.,RAC, FRAPS Bio-Rad Laboratories, Redmond,
The Challenges in Developing and Commercializing HIV Tests that are Useful in Differentiating VISP/R VISP/R Workshop Bethesda, MD March 2013 Christopher Bentsen, M.S.,RAC, FRAPS Bio-Rad Laboratories, Redmond,
Toward Diagnosing Leishmania infantum Infection in Asymptomatic Dogs in an Area Where Leishmaniasis Is Endemic
 CLINICAL AND VACCINE IMMUNOLOGY, Mar. 2009, p. 337 343 Vol. 16, No. 3 1556-6811/09/$08.00 0 doi:10.1128/cvi.00268-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Toward Diagnosing
CLINICAL AND VACCINE IMMUNOLOGY, Mar. 2009, p. 337 343 Vol. 16, No. 3 1556-6811/09/$08.00 0 doi:10.1128/cvi.00268-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Toward Diagnosing
Chagas disease: Challenges in Diagnostics
 Chagas disease: Challenges in Diagnostics Emily Adams Royal Tropical Institute (KIT), KIT Biomedical Research The Netherlands e-mail: e.adams@kit.nl Aims of KIT Biomedical Research To contribute to the
Chagas disease: Challenges in Diagnostics Emily Adams Royal Tropical Institute (KIT), KIT Biomedical Research The Netherlands e-mail: e.adams@kit.nl Aims of KIT Biomedical Research To contribute to the
GLP-2 ELISA. For the quantitative determination of GLP-2 in human serum and plasma samples.
 GLP-2 ELISA For the quantitative determination of GLP-2 in human serum and plasma samples. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2HU-E01.1 Size: 96 wells Version:
GLP-2 ELISA For the quantitative determination of GLP-2 in human serum and plasma samples. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2HU-E01.1 Size: 96 wells Version:
Hepatitis A virus IgM ELISA Kit
 Hepatitis A virus IgM ELISA Kit Catalog Number KA0285 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Hepatitis A virus IgM ELISA Kit Catalog Number KA0285 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Antibodies against Lutzomyia longipalpis saliva in the fox Cerdocyon thous and the sylvatic cycle of Leishmania chagasi
 Transactions of the Royal Society of Tropical Medicine and Hygiene (2007) 101, 127 133 available at www.sciencedirect.com journal homepage: www.elsevierhealth.com/journals/trst Antibodies against Lutzomyia
Transactions of the Royal Society of Tropical Medicine and Hygiene (2007) 101, 127 133 available at www.sciencedirect.com journal homepage: www.elsevierhealth.com/journals/trst Antibodies against Lutzomyia
CHAPTER 4 RESULTS 42
 CHAPTER 4 RESULTS 42 4.1 Introduction This chapter is divided in two main sections. The first section deals with results obtained for the point-of-care tests, which encompass results of optimization final
CHAPTER 4 RESULTS 42 4.1 Introduction This chapter is divided in two main sections. The first section deals with results obtained for the point-of-care tests, which encompass results of optimization final
Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit
 PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
Seroprevalence of canine visceral leishmaniasis in southeast of Iran
 J Parasit Dis (Apr-June 2014) 38(2):218 222 DOI 10.1007/s12639-012-0226-9 ORIGINAL ARTICLE Seroprevalence of canine visceral leishmaniasis in southeast of Iran Mostafavi Mahshid Akhtardanesh Baharak Sharifi
J Parasit Dis (Apr-June 2014) 38(2):218 222 DOI 10.1007/s12639-012-0226-9 ORIGINAL ARTICLE Seroprevalence of canine visceral leishmaniasis in southeast of Iran Mostafavi Mahshid Akhtardanesh Baharak Sharifi
2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit
 2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as
2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as
STAT3 (py705) (Human/Mouse/Rat) ELISA Kit
 STAT3 (py705) (Human/Mouse/Rat) ELISA Kit Catalog Number KA2175 96 assays Version: 01 Intended for research use only www.abnova.com I. INTRODUCTION STAT3 (py705) (Human/Mouse/Rat) ELISA (Enzyme-Linked
STAT3 (py705) (Human/Mouse/Rat) ELISA Kit Catalog Number KA2175 96 assays Version: 01 Intended for research use only www.abnova.com I. INTRODUCTION STAT3 (py705) (Human/Mouse/Rat) ELISA (Enzyme-Linked
Toxoplasma gondii IgM (Toxo IgM)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
