Good Feasibility of the New German Blood Donor Questionnaire
|
|
|
- Ethan Shepherd
- 6 years ago
- Views:
Transcription
1 Original Article Transfus Med Hemother 2017;44: DOI: / Received: November 9, 2016 Acepted: May 30, 2017 Published online: July 14, 2017 Good Feasibility of the New German Blood Donor Questionnaire Claudia Houareau a Robert Deitenbeck b Ariane Sümnig c Anette Moeller d e Christiane Saadé Frank Stötzer f Margarethe Heiden g Hinnak Northoff h Ruth Offergeld a a Department of Infectious Disease Epidemiology, Robert Koch Institute, Berlin, Germany; b German Red Cross Blood Donation Service West, Hagen, Germany; c Institute for Immunology and Transfusion Medicine, University Greifswald, Greifswald, Germany; d Institute for Immunology and Transfusion Medicine, University Gießen, Gießen, Germany; e Institute for Laboratory Medicine, Transfusion Medicine and Microbiology, Helios Clinic, Pforzheim, Germany; f German Red Cross Blood Donation Service Baden-Württemberg-Hessen, Mannheim, Germany; g Department for Transfusion Medicine, Paul Ehrlich Institute, Langen, Germany; h Center for Clinical Transfusion Medicine, ZKT, University of Tübingen, Tübingen, Germany Keywords Blood donors Donor selection Blood donor questionnaire Summary Background: We assessed the effect of the uniform donor questionnaire (UDQ) on deferral rates in first-time and repeat donors. We focused on the introduced question about unprotected sexual contact with a new partner. Another goal was a stratified comparison of the deferral rates of the donor questionnaire (DQ) and UDQ. Methods: Data on donors and deferrals using the DQ and UDQ were collected at four blood establishments. The comparison included a 2-year period by questionnaire version. For the comparison of the questionnaires, an adjusted multinomial logistic regression was performed. Results: The analysis included 260,848 donations. First-time (FTD) and repeat donations (RD) showed higher deferral rates with the UDQ (FTD +5.4%, RD +1.4%). Deferral due to a new partner was 3.0% in firsttime and 0.4% in repeat donors. The majority of these occurred in the youngest age groups. The most frequent deferral criterion was disease (5.1%). Conclusion: The regression revealed stronger predictors for deferral than the questionnaire version. Especially younger age carried a higher and independent risk for deferral. The additional deferrals of mainly young first-time donors due to a new sexual partner may identify those donors with potential heterosexual risk behavior who would otherwise not be identified S. Karger GmbH, Freiburg Introduction The selection of healthy donors is crucial to maintain the existing high level of blood safety. Thus, the donor questionnaire (DQ) becomes an important part of the donor selection process. In Germany, at present, different questionnaires are in use due to the de-centralized structure of the blood supply [1]. All DQs fulfil the criteria required by the German Hemotherapy Guidelines and regulations of the competent authority [2]. To optimize the quality of the DQ, a group on behalf of the National Advisory Committee Blood developed a uniform DQ (UDQ) which aimed to be as simple as possible, comprehensible, accepted by donors, and effective in selecting low-risk healthy donors [1, 3]. Special attention was paid to (hetero-)sexual risk behavior. A new question on unprotected sexual contact with a new partner within the 4 months preceding the donation was introduced. Results of a first evaluation of the newly developed UDQ showed that especially young male firsttime donors were deferred due to a new sexual partner [3]. This raised the concern that many donors, including repeat donors, could be lost if the UDQ become mandatory, especially within the younger donor population. This concern was addressed in a second multicenter trial with a larger sample size including first-time and repeat donors. The goal of this study was to determine the effect of the UDQ on deferral rates in first-time and repeat donors. We focused on the effects of the newly introduced question about unprotected sexual contact with a new partner. In a second step, we made a stratified comparison of deferral rates between the UDQ and the DQ. Fax Information@Karger.com S. Karger GmbH, Freiburg Accessible online at: Claudia Houareau Department of Infectious Diseases Robert Koch Institute Seestraße 10, Berlin, Germany HouareauC@rki.de
2 Material and Methods Call for Participants All blood establishments (BE) in Germany were invited to participate in the study via the homepage of the National Competent Authority (Paul Ehrlich Institute). Different levels of data entry for deferrals were offered to the participants of the study. All data were stratified for sex, age group (18 24 years; years; years; years; >54 years), and donor status (first-time or repeat donor). The following levels of data entry were used by the BE which participated in the study: 1) Deferrals were reported on an individual level differentiating between temporary and permanent deferrals. Reasons for deferral were summed in ten categories: disease (which included surgery, medication, vaccination, and all types of medical treatment or underlying conditions); risk for Creutzfeldt- Jacob disease (CJD) and variant CJD; pregnancy and breast feeding; physical examination; travel-associated risks; sexual risk behavior (except for new partner); unprotected sexual contact with a new partner within the 4 months preceding the donation; injecting drug use; tattoo, piercing, needle stick injury; other reasons. 2) Deferrals were reported as aggregated data. Within this level of data entry, five categories for deferral (new partner, disease, travel-associated, tattoos, and sexual risk behavior) were used. These data were also stratified for sex, age group, and donor status. Both levels included the number of successful donations and the number of confidential unit exclusions (CUE) stratified for sex, age group, and donor status. Study Participants Our multicenter study was conducted between 2009 and 2013 at the following four BE: German Red Cross Blood Service West, Hagen (BSD West); Department for Transfusion Medicine, University of Greifswald; Institute for Immunology and Transfusion Medicine, Medical University Gießen and Marburg; Institute for Laboratory Medicine and Transfusion Medicine, Helios Hospital Pforzheim. All BE submitted their data generated using the UDQ. In addition, two BE (BSD West, Gießen/Marburg) added comparative data using the DQ (table 1). Retrospectively, BSD West submitted data sets based on the 2 years preceding the study when the established DQ was used. Statistical Analyses The analysis was based on donations. The aggregated data on successful donations were imputed to individual data. Differences on descriptive level were analyzed with the chi-square test according to Pearson. Throughout statistical analyses, we aggregated temporary and permanent deferral into one deferral variable. We excluded CUE cases from any regression analyses. To further investigate the contribution of the newly introduced category new partner, we performed multinomial logistic regression. We employed multinomial logistic regression analysis to estimate deferral within the UDQ in the following categories: new partner, disease, tattoos, and other reasons depending on sex and age group. The base outcome was successful donation. Interactions between sex and age group were included to characterize possible risks for deferral. We adjusted regression analysis for sex, age group, and donor status. For sex, age group, and donor status, we used donations from female donors, age group years, and donations from repeat donors as reference categories. Since we found an overall effect of first-time donation, we limited the model to repeat donations and adjusted it for sex and age group as described above. For the comparison of DQ and UDQ, one BE (BSD West) gathered information fit for the adjusted multinomial logistic regression. The comparison of DQ and UDQ was adjusted with regard to sex, age group, and questionnaire version (DQ as reference category). The comparison did not include the deferral category new partner since this category did not exist in the established DQ. All regression results are reported in relative risks and their significant contribution to the model. We compared 2 years with the DQ versus 2 years with the UDQ. P values < 0.05 were considered significant. All analyses were performed using STATA 14.1 (StataCorp LP, College Station, TX, USA). Results Of all participating BE, data of 261,016 donations entered the study. Missing data summed up to 0.1% of the total sample size. All remaining 260,848 donations were included into the analysis (99.9%). These included 145,965 (55.9%) donations collected with the UDQ. Two sites applied both questionnaires (BSD West and Gießen/Marburg). At the BSD West the UDQ rate was 49.1%, and at Giessen/Marburg it was 39.0% of all donations. Descriptive Analysis The deferral rate in the four study sites ranged from 9.4% in repeat donations to 34.7% in first-time donations. A CUE was made in 0.3% of all donations, irrespective of donor status. Details are presented in table 2. Reasons for Deferral Of all deferral categories applied in this study, disease showed the highest deferral rate in first-time (13.8%) and repeat donations (3.7%). Deferral rates for all categories are listed in table 3. Table 1. Overview of the period and questionnaires applied by study participants Study site Questionnaire Time period BSD West DQ UDQ Gießen/Marburg DQ , UDQ , , Greifswald UDQ Pforzheim UDQ Good Feasibility of the New German Blood Donor Questionnaire Transfus Med Hemother 2017;44:
3 Table 2. Deferrals with the UDQ by study site and donor status (n = 145,965) Study site Donor status Total Deferral (%) CUE (%) Successful donations (%) BSD West FTD 17,423 6,619 (38.0) 38 (0.2) 10,766 (61.8) RD 90,907 10,107 (11.1) 302 (0.3) 80,498 (88.6) Marburg FTD (10.5) 2 (0.9) 206 (89.6) RD 1, (2.1) 4 (0.3) 1,464 (97.6) Greifswald FTD 1, (19.8) 15 (1.0) 1,207 (79.3) RD 17,216 1,329 (7.7) 48 (0.3) 15,839 (92.0) Pforzheim FTD 1, (16.4) 0 (0.0) 981 (83.6) RD 15, (2.0) 1 (0.0) 15,675 (98.0) All FTD 20,350 7,135 (34.7) 55 (0.3) 13,160 (64.7) RD 125,615 11,784 (9.4) 355 (0.3) 113,476 (90.3) all 145,965 18,919 (13.0) 410 (0.3) 126,636 (86.8) FTD = First-time donation; RD = repeat donation; CUE = confidential unit exclusion. Table 3. Descriptive of deferral categories of the UDQ (n = 145,965) Deferral category Donor status FTD (%) RD (%) All (%) Successful donation 13,160 (64.7) 113,476 (90.3) 126,636 (86.8) Disease 2,809 (13.8) 4,588 (3.7) 7,397 (5.1) Examination 1,522 (7.5) 3,973 (3.2) 5,495 (3.8) Tattoos 1,057 (5.2) 737 (0.6) 1,794 (1.2) New partner 616 (3.0) 532 (0.4) 1,148 (0.8) Travel associated 149 (0.7) 302 (0.2) 451 (0.3) Injecting drug use # 167 (0.8) 59 (0.1) 226 (0.2) Sexual risk behavior 79 (0.4) 41 (0.0) 120 (0.1) Pregnancy # 55 (0.3) 52 (0.0) 107 (0.1) CJD/ vcjd # 28 (0.1) 13 (0.0) 41 (0.0) Other # 653 (3.2) 1,487 (1.2) 2,140 (1.5) Total 20,355 (100.0) 125,615 (100.0) 145,965 (100.0) FTD = First-time donation; RD = repeat donation; # Not reported by Gießen/Marburg. The two youngest age groups revealed the highest deferral due to a new partner: 2.1% (531/25,403) of all donations of 18- to 24-year-old donors and 1.4% (452/32,050) of all donations of 25- to 34-year-old donors were deferred. Stratified for donor status, deferrals of male first-time donors were significantly higher than deferrals of female first-time donors within the same age groups (18- to 24-year-old donors: chi-square (1) = 35.5; p < 0.01; 25- to 34-year-old donors: chi-square (1) = 34.0; p < 0.01) Details are shown in figure 1. Multinomial Logistic Regression Model The adjusted multinomial logistic regression model to identify risk factors for deferral showed an overall effect of first-time donations (chi-square (5) = , p < 0.01). Therefore, we limited the model to repeat donations, which constitute 86.1% of all donations applying the UDQ (125,615/145,965). Relative Risks of Deferral due to Newly Introduced Category New Partner Results of the final model revealed: Donations of the two youngest age groups had a three (18 24 years) and two times (25 34 years) higher expected risk of exclusion due to new partner (RR years = 3.06, p < 0.01; RR years = 2.45, p < 0.01). The two oldest age groups had significantly lower expected risks of being deferred by indicating unprotected intercourse with new sexual partner within the preceding 4 months (RR years = 0.38, p < 0.01; RR >54 years = 0.08, p < 0.01). Neither sex nor the interaction of sex and age group significantly contributed to the estimation of deferral in the category new partner regarding repeat donations. Influence of Sex Within the categories disease, tattoos, and other, female donors had an expected higher risk of deferral (disease: RR male = 0.67, p < 0.01; tattoos: RR male = 0.45, p < 0.01; other: RR male = 0.48, p < 0.01). 234 Transfus Med Hemother 2017;44: Houareau/Deitenbeck/Sümnig/Moeller/Saadé/ Stötzer/Heiden/Northoff/Offergeld
4 Fig. 1. a Proportion of deferral due to new partner in first-time donors (n = 20,355 donations). b Proportion of deferral due to new partner in repeat donors (n = 125,615). Good Feasibility of the New German Blood Donor Questionnaire Transfus Med Hemother 2017;44:
5 Table 4. Sociodemographic characteristics (n = 219,559) DQ RD (n = 93,141) all (n = 112,176) UDQ RD (n = 90,510) all (n = 107,383) Sex (%) Female Age groups (%) years ** 13.8** years ** 21.2** years years >54 years ** 31.0** DQ = Donor questionnaire; UDQ = uniform DQ; RD = repeat donation; **p < Table 5. Deferrals according to the UDQ versus DQ by donor status (excluding category new partner ) (n = 219,559) Deferral category DQ (n=112,176) UDQ (n=107,383) FTD RD all FTD RD all Disease 2,112 (11.10) 2,911 (3.13) 5,023 (4.48) 2,594 (15.37) 4,138 (4.57) 6,732 (6.27) Examination 1,555 (8.15) 3,210 (3.45) 4,765 (4.25) 1,412 (8.37) 3,210 (3.55) 4,622 (4.30) Other 629 (3.30) 1,4321 (1.54) 2,060 (1.84) 576 (3.41) 1,283 (1.42) 1,859 (1.73) Tattoos 1,115 (5.86) 709 (0.76) 1,824 (1.63) 1,037 (6.15) 683 (0.75) 1,720 (1.60) Travel 91 (0.48) 239 (0.26) 330 (0.29) 131 (0.78) 251 (0.28) 382 (0.36) IDU 162 (0.85) 38 (0.44) 200 (0.18) 166 (0.98) 59 (0.07) 225 (0.21) Sexual risk 57 (0.30) 18 (0.21) 75 (0.07) 72 (0.43) 29 (0.03) 101 (0.09) Pregnancy 48 (0.25) 68 (0.07) 116 (0.10) 54 (0.32) 46 (0.05) 100 (0.09) CJD/vCJD 51 (0.27) 22 (0.02) 73 (0.07) 27 (0.16) 11 (0.01) 38 (0.04) Total deferral 5,820 (30.6) 8,646 (9.3) 14,466(12.9) 6,069 (36.0)** 9,710 (10.7)** 15,779 (14.7)** CUE 43 (0.2) 340 (0.4) 383 (0.3) 38 (0.2) n.s. 302 (0.3) n.s. 340 (0.3) n.s. Total donations 19,035 (100) 93,141(100) 112,176 (100) 16,873 (100) 90,510 (100) 107,383 (100) DQ = Donor questionnaire; UDQ = uniform DQ; FTD = first-time donation; RD = repeat donation; CUE = confidential unit exclusion; IDU = injecting drug use; ** p < 0.01; n.s. = not significant. Influence of Age Group As in the category new partner, we found in tattoos and other a highly significant expected risk of the two youngest age groups to be deferred (tattoos: RR years = 2.06, p < 0.01; RR years = 1.47, p < 0.01; other: RR years = 1.79, p < 0.01; RR years = 1.57; p < 0.01) while the two oldest age groups showed a highly significant lower expected risk to be deferred within these categories (tattoos: RR years = 0.51, p < 0.01; RR >54 years = 0.22, p < 0.01; other: RR years = 0.76, p < 0.01; RR >54 years = 0.63, p < 0.01). In contrast to these results, within the category disease only donations of 25- to 34-year-old donors have a higher expected risk for deferral due to disease (disease: RR years = 1.33, p < 0.01). For all categories, we found interactions between sex and age group in the category other only; indicating lower risks for deferral due to other in donations of men within the two youngest age groups (other: RR male*18 24 years = 0.72, p < 0.01; RR male*25 34 years = 0.70; p < 0.01). Donations of men within the eldest age group show higher expected risk for deferral within the category other (RR male*>54 years = 1.80, p < 0.01). Comparison of the DQ and the UDQ Of 260,848 data sets, 219,559 (84.2%) were from one BE (BSD- West) using both questionnaire versions and all deferral categories each over a 2-year period: 112,176 (51.1%) with the DQ and 107,383 (48.9%) with the UDQ (table 4). Female donors were more often deferred than male donors in both questionnaires (UDQ: female 19.7% vs. male: 12.2%; chisquare (1) = 1,300; p < 0.01; DQ: female 16.8% vs. male 9.9%; chisquare (1) = 1,200; p < 0.01). However, there were no sex differences in deferrals between questionnaire versions (chi-square (1) = 0.00; p > 0.05). The overall deferral rate increased with the UDQ by 1.8%. Especially, first-time donors had a 5.4% higher deferral rate (table 5). This is mainly attributed to the deferral category disease. Deferrals due to pregnancy, travel, and sexual risk behavior, increased by less than 0.3% with the UDQ. CUE did not differ between questionnaire versions (chi-square (1) = 0.02; p > 0.05); irrespective of donor status. However, in both questionnaires male first-time donors used CUE more frequently than female first-time donors (UDQ: chisquare (1) = 16.13; p < 0.01; DQ: chi-square (1) = 4.03; p < 0.05). 236 Transfus Med Hemother 2017;44: Houareau/Deitenbeck/Sümnig/Moeller/Saadé/ Stötzer/Heiden/Northoff/Offergeld
6 Adjusted Multinomial Logistic Regression Adjusted multinomial logistic regression was confined to the group of repeat donations due to an overall effect of first-time donors (chi-square (4) = , p < 0.01; 80.4% (176,589/219,559). Influence of Questionnaire Version Donors using the UDQ had a higher expected risk of deferral in the category disease (RR UDQ = 1.5; p < 0.01) than donors using the DQ regardless of age and sex. On the other hand, donors using the DQ had a higher expected risk of deferral in the categories tattoos (RR UDQ = 0.87, p < 0.01) and other (RR UDQ = 0.92, p < 0.01) independent of age and sex. Influence of Sex Women showed significantly higher expected risks for deferral in the categories disease and tattoos independent of questionnaire version and age group (disease: RR male = 0.72, p < 0.01; tattoos: RR male = 0.59, p < 0.01). Influence of Age Group In almost all categories the two youngest age groups were at a higher expected risk of exclusion independent of sex and questionnaire version (disease: RR years = 1.2, p > 0.05; RR years = 1.2, p < 0.01; tattoos: RR years = 3.6, p < 0.01; RR years = 2.0, p < 0.01; other: RR years = 1.5, p < 0.01; RR years = 1.2, p < 0.05), whereas donations of the two oldest age groups were less likely to be excluded (disease: RR years = 0.9, p < 0.05; RR >54 years = 0.8, p < 0.01; tattoos: RR years = 0.6, p < 0.01; RR >54 years = 0.2, p < 0.01; other: RR years = 0.7, p < 0.01; RR >54 years = 0.7, p < 01). Influence of Sex and Age Group Within the category disease men aged years were at risk for deferral (RR male*18 24 years = 1.3, p < 0.05). Discussion Donor selection is an important step in maintaining the high level of safety for donor and recipient. The presented study assessed deferral rates in first-time and repeat donors when a recently developed uniform UDQ was used. We focused on the effect of the introduced question about a new sexual partner. Additionally, we compared the UDQ with the established DQ. We found an overall deferral rate of 13.0% with the new UDQ. This rate was similar to comparable studies: In the literature, about 10 15% of applicant donors are deferred due to the DQ [4 6]. Still, in comparison to the established DQ, we found an increase in deferrals by 5.4% and 1.4% in first-time and repeat donations respectively. These additional deferrals were mainly due to higher deferral rates in the category disease. This corresponds well to the results of the previous DQ study in which the UDQ was evaluated in a multicenter trial with 6,500 first-time donors [3]. The category disease included medical treatment of any kind or underlying medical conditions. Donor deferral due to this category is intended to reduce the risk of undetected transfusion-transmissible infections (TTI) or to assure donor health [7, 8]. Therefore, we consider the moderate increase in deferrals due to medical treatment or conditions to be of benefit with respect to blood and donor safety. An essential innovation in the UDQ was the introduction of a question regarding unprotected intercourse with a new sexual partner in the 4 months preceding the donation. This question addressed heterosexual risk behavior as sex between men lead to a permanent deferral according to the German Hemotherapy Guidelines [2]. In recent analyses, undisclosed heterosexual contacts revealed to be a risk factor for TTI (Preußel, personal communication; [9 12]). In Germany, an analysis demonstrated that unreported heterosexual risks were more prevalent in HIV- or HCVinfected blood donors than in cases from the general population (Preußel, personal communication). According to this study, the identification of all heterosexual risk contacts might prevent acceptance of 53% of HIV-infected donors. A further study of undisclosed risk behavior in HIV-positive donors in Italy revealed heterosexual risk contacts in 68.1% [9]. Another retrospective analysis of pre-donation screening revealed acute HBV infections in donors reporting new heterosexual partners [11]. Also in the UK, there were more reported heterosexual exposures among infected blood donors (both with and without a partner with an identifiable risk) in 2014 than in the previous year, while other reported exposure like sex between men decreased [12]. The UK blood donor surveillance report showed that 18 of 24 donors with a recent infection were infected by heterosexual contacts. Of these, 4 male and 3 female donors reported one or more new partners. Also, like in Germany, a rise in recent syphilis infections among new donors, especially heterosexual men, was observed in the UK [12]. The rise was most prominent in young male donors but also seen to a lesser extent in young female donors, which could imply a rise in (hetero-)sexual risk behavior. Heterosexual risk behavior was addressed in former DQs using leading questions about, e.g., one-night stands, casual contacts, or frequent change of partner. These questions partly left the risk assessment to the donor. However, recent studies confirmed that in post-donation interviews, the majority of HIV-positive donors did not realize they engaged in risk behavior [9, 10, 13]. Therefore, a precise definition of heterosexual risk behavior would be desirable. Attempts to make this definition failed with exception of highrisk behavior like sex work [14]. Therefore, in the conception of the UDQ, we decided to address heterosexual exposure (unprotected sexual contact with a new partner prior to the donation) rather than making an arbitrary definition of heterosexual risk behavior. This procedure is well established in other countries like Spain or Italy [14]. Further advantages of this approach are that a question about sexual contact with a new partner is clear and non-judgmental and leaves little room for interpretation by the donor [3]. The question about a new sexual partner chiefly lead to deferrals of younger first-time donors: On the descriptive level, we found significantly higher deferral rates in the new partner category among male first-time and to a lesser extent among repeat donors. If we are controlling for the large sample size by using effect Good Feasibility of the New German Blood Donor Questionnaire Transfus Med Hemother 2017;44:
7 sizes, i.e. relative risks, provided by a multinomial logistic regression analysis [15], the indications of higher deferral rates of young male donors did not achieve significance in repeat donations. The multinomial logistic regression showed higher risks for exclusion due to a new sexual partner in the two youngest age groups, independent of sex. The new partner question identified the group which empirically carries the highest risk for TTIs [16]. Thus the new partner question is capable of preventing window period donations of donors with unspecified heterosexual risks. Still, demographic changes in Germany will most likely lead to fewer younger donors in the future on the one hand and to an increasing need of blood and blood products due to an ageing population on the other [17, 18]. Therefore, the reasons for deferral need to be clearly communicated to potential donors, and low-risk donors should be recruited continuously in order to provide enough appropriate blood products for patients in need. The recently published first results of the donor of the future project also addressed demographic changes of the donor population and showed the need for new service strategies for future donor recruitment in order to meet the high expectations of donors [19]. Ultimately, the safety gained by rigid and straightforward donor selection for sexual risks must not lead to a disproportional loss of recruited and retained young repeat donors. In the first UDQ study [3], some donors felt that the character of direct questions about their sexual contacts were too private. Nonetheless, the UDQ was acceptable to the majority of donors [17] and demonstrated its superiority compared to the established DQ regarding comprehensibility as well as donor adherence [3]. Furthermore, results of a cognitive interview evaluation of the new AABB DQ found no evidence that blood donors modified their answers due to a perceived inappropriate sensitive character of questions [10]. The comparison of questionnaire versions did not include the new partner question as it was not asked in the DQ. In the comparative analysis of the remaining categories, the only significant influence of the UDQ on deferral rates was found in the category disease. This was demonstrated on the descriptive level in a previous study [20]. Our regression analysis revealed that the observed higher deferral rates in the category disease were independent of age and sex. The data available for this study did not allow for analysis of underlying reasons for deferral in any category. Still, a comparison of the questionnaires showed that in the DQ almost all medical reasons for deferral were asked in one grouped question. In contrast with the UDQ, it was initially asked whether the donor is feeling sick or is on sick leave. This was followed by a set of grouped questions regarding medical conditions. Furthermore, with the UDQ it was explicitly asked about certain commonly used drugs, i.e. acetylsalicylic acid. Similar reasons for higher deferral rates with the DQ in the category tattoos can be assumed. With the DQ, tattoo, piercing, and needle stick injury were asked in one grouped question with acupuncture, colonoscopy as well as contact to a patient with hepatitis. In contrast to the DQ, the UDQ included separate questions to tattoo, piercing, and needle stick injury. These differences in layout and questions may have led to the above mentioned differences in deferral rates between the two questionnaire versions and warrant further investigations. Overall, female donors were more often deferred than male with both questionnaires. This has also been observed in other investigations [20, 21]. However, the regression analysis showed that women were not significantly more often deferred with the UDQ. Also, in almost all categories for deferral the two youngest age groups were at a higher risk of deferral independent of sex and questionnaire version. These results from the regression analyses differed from the results of a previous DQ study [20]. There are limitations with regard to our study: Due to the categorization of reasons for deferral, we were unable to specify precise differences in deferrals. This applied especially to the category disease which includes all types of medical treatment or underlying conditions. The UDQ was offered for nationwide use via the homepage of the competent authority in 2011 together with an invitation to participate in the study. Still, the evaluation was limited to four BE of which only one was able to provide a complete set of comparative data for the UDQ and the DQ, allowing for regression analysis. Only two university BE participated, and thus it is difficult to predict if the UDQ has specific effects on a predominantly student donor population. In conclusion, we were able to demonstrate the feasibility of the UDQ in this multicenter study. Most deferrals are due to medical treatment or conditions and ensure blood product safety and donor health. The subsequent higher deferral rates need to be monitored, and appropriate measures need to be taken to ensure the blood supply also in the future. The temporary deferral of individuals with a new heterosexual partner is warranted to deal with non-specified but increasing heterosexual risk behavior among donors. Further studies, including donor perception of questionnaires and correlations with screening results of those who are deferred, will be beneficial. We hope that this work motivates and guides future research in this area. Acknowledgment The authors would like to thank Dr. Schwenz for her support in finishing this manuscript and Mr. Rolf Kessel for his invaluable support in data extraction. Disclosure Statement All authors declare that they have no conflict of interests due to sponsorship or funding arrangements related to this study. 238 Transfus Med Hemother 2017;44: Houareau/Deitenbeck/Sümnig/Moeller/Saadé/ Stötzer/Heiden/Northoff/Offergeld
8 References 1 Stellungnahme des AK Blut: Verwendung eines einheitlichen Fragebogens für Blut- und Plasmaspender. Bundesgesundheitsbl Gesundheitsforsch Gesundheitsschutz 2010; 53: Bundesärztekammer: Richtlinien zur Gewinnung von Blut und Blutbestandteilen und zur Anwendung von Blutprodukten (Hämotherapie). Cologne, Deutscher Ärzte-Verlag, Offergeld R, Heiden M, Stötzer F, Northoff H: Der einheitliche Spenderfragebogen. Hämotherapie 2011; 16: Zou S, Fujii K, Johnson S, Spencer B, Washington N, Iv EN, Musavi F, Newman B, Cable R, Rios J: Prevalence of selected viral infections among blood donors deferred for potential risk to blood safety. Transfusion 2006; 46: Zou S, Musavi F, Notari EP, Fujii KE, Dodd RY: Prevalence of selected viral infections among temporarily deferred donors who returned to donate blood: American Red Cross blood donor study. Transfusion 2005; 45: Custer B, Schlumpf K, Simon TL, Spencer BR, Wright DJ, Wilkinson SL: Demographics of successful, unsuccessful and deferral visits at six blood centers over a 4 year period. Transfusion 2012; 52: Lin C, Leung J, So B, Lee C: Donor selection for blood safety: is it still necessary? ISBT Sci Ser 2014; 9: Yang H, Anderson SA, Forshee R, Williams A, Epstein JS, Marks PW. Modeling complete removal of risk assessment questions in the USA predicts the risk of HIV exposure in blood recipients could increase despite the use of nucleic acid testing. Vox Sang 2016; 110: Raimondo M, Facco G, Regine V, Pupella S, Grazzini G, Suligoi B: HIV positive blood donors unaware of their sexual at risk behaviours before donation in Italy. Vox Sang 2016; 110: Willson S, Miller K, Seem D, Kuehnert MJ: Cognitive evaluation of the AABB uniform donor history questionnaire. Transfusion 2016; 56: Lieshout Krikke RW, Zaaijer HL, Laar TJ: Predonation screening of candidate donors and prevention of window period donations. Transfusion 2015; 55: Public Health England: Safe Supplies: Annual Review. 2013, last updated November 18, government/publications/safe-supplies-annual-review (last accessed June 8, 2017). 13 Lucky TT, Seed CR, Waller D, Lee FJ, McDonald A, Wand H, Wroth S, Shuttleworth G, Keller AJ, Pink J, Wilson DP: Understanding noncompliance with selective donor deferral criteria for high-risk behaviours in Australian Blood donors. Transfusion 2014; 54: Offergeld R, Kamp C, Heiden M, Norda R, Behr Gross ME: Sexual risk behaviour and donor deferral in Europe. Vox Sang 2014; 107: Lin M, Lucas HC Jr, Shmueli G: Research commentary too big to fail: large samples and the p-value problem. Information Systems Research 2013; 24: Offergeld R, Ritter S, Hamouda O: HIV-, HCV-, HBVund Syphilissurveillance unter Blutspendern in Deutschland Bundesgesundheitsblatt 2012; 55: Sümnig A, Lembcke H, Weber H, Deitenbeck R, Greffin K, Bux J, Greinacher A: Evaluation of a new German blood donor questionnaire. Vox Sang 2014; 106: Ritter S, Hamouda O, Offergeld R: Demografie und Spendeaktivität von Blut- und Plasmaspendern in Deutschland. Bundesgesundheitsbl Gesundheitsforsch Gesundheitsschutz 2012; 55: Sundermann L, Kort W, Boenigk S: The donor of the future project first results and further research domains. Vox Sang 2017; DOI: /vox Müller-Steinhardt M, Weidmann C, Wiesneth M, Weck E, Seifried E, Brade J, Klüter H: Donor deferral rates after the implementation of a new german blood donor questionnaire. Transfus Med Hemother 2012; 39: Misje AH, Bosnes V, Heier H: Gender differences in presentation rates, deferrals and return behaviour among Norwegian blood donors. Vox Sang 2010; 98:e241 e248.
Donor Satisfaction with a New German Blood Donor Questionnaire and Intention of the Donor to Return for Further Donations
 Original Article DOI: 10.1159/000354844 Received: May 31, 2012 Accepted: December 13, 2012 Published online: August 26, 2013 Donor Satisfaction with a New German Blood Donor Questionnaire and Intention
Original Article DOI: 10.1159/000354844 Received: May 31, 2012 Accepted: December 13, 2012 Published online: August 26, 2013 Donor Satisfaction with a New German Blood Donor Questionnaire and Intention
Blood Donor Counselling
 Blood Donor Counselling A presentation for the Nepal Red Cross Society Blood Transfusion Service Dr Che Kit Lin On behalf of GAP and Hong Kong Red Cross 26 th August 2014 Purpose and Outcomes of Workshop
Blood Donor Counselling A presentation for the Nepal Red Cross Society Blood Transfusion Service Dr Che Kit Lin On behalf of GAP and Hong Kong Red Cross 26 th August 2014 Purpose and Outcomes of Workshop
Change in deferrals for men who have sex with men: Assessing the risk of HIV transmission by transfusion
 Change in deferrals for men who have sex with men: Assessing the risk of HIV transmission by transfusion Josiane Pillonel, Rachid Djoudi, Florence Lot, Chistophe Martinaud, Claire Sauvage, Marie Jauffret-Roustide,
Change in deferrals for men who have sex with men: Assessing the risk of HIV transmission by transfusion Josiane Pillonel, Rachid Djoudi, Florence Lot, Chistophe Martinaud, Claire Sauvage, Marie Jauffret-Roustide,
R Offergeld, D Faensen, S Ritter, O Hamouda Department for Infectious Disease Epidemiology, Robert Koch Institute, Berlin, Germany
 Eurosurveillance, Volume 10, Issue 2, 01 February 2005 Surveillance report HUMAN IMMUNODEFICIENCY VIRUS, HEPATITIS C AND HEPATITIS B INFECTIONS AMONG BLOOD DONORS IN GERMANY 2000-2002: RISK OF VIRUS TRANSMISSION
Eurosurveillance, Volume 10, Issue 2, 01 February 2005 Surveillance report HUMAN IMMUNODEFICIENCY VIRUS, HEPATITIS C AND HEPATITIS B INFECTIONS AMONG BLOOD DONORS IN GERMANY 2000-2002: RISK OF VIRUS TRANSMISSION
The safety of blood donation by elderly blood donors
 ORIGINAL PAPER Vox Sanguinis (2011) 101, 313 319 ª 2011 The Author(s) Vox Sanguinis ª 2011 International Society of Blood Transfusion DOI: 10.1111/j.1423-0410.2011.01492.x The safety of blood donation
ORIGINAL PAPER Vox Sanguinis (2011) 101, 313 319 ª 2011 The Author(s) Vox Sanguinis ª 2011 International Society of Blood Transfusion DOI: 10.1111/j.1423-0410.2011.01492.x The safety of blood donation
Versorgungsforschung in Deutschland Der Beitrag der Transfusionsmedizin. Health Services Research in Germany The Input of Transfusion Medicine
 Versorgungsforschung in Deutschland Der Beitrag der Transfusionsmedizin Health Services Research in Germany The Input of Transfusion Medicine Prof. Dr. Harald Klüter Institute of Transfusion Medicine and
Versorgungsforschung in Deutschland Der Beitrag der Transfusionsmedizin Health Services Research in Germany The Input of Transfusion Medicine Prof. Dr. Harald Klüter Institute of Transfusion Medicine and
MSM DONOR POLICY: STUDIES FROM THE USA AND OTHER COUNTRIES. Brian Custer IPFA PEI Workshop Rome 22 May 2014
 MSM DONOR POLICY: STUDIES FROM THE USA AND OTHER COUNTRIES Brian Custer IPFA PEI Workshop Rome 22 May 2014 Financial Disclosure Nothing to disclose International Context for Blood Donation by MSM Country
MSM DONOR POLICY: STUDIES FROM THE USA AND OTHER COUNTRIES Brian Custer IPFA PEI Workshop Rome 22 May 2014 Financial Disclosure Nothing to disclose International Context for Blood Donation by MSM Country
Policy Document. Blood Donation Deferral. Background
 Policy Document Blood Donation Deferral Background The Australian Medical Students Association (AMSA) is the peak representative body for medical students in Australia. Accordingly, AMSA advocates on issues
Policy Document Blood Donation Deferral Background The Australian Medical Students Association (AMSA) is the peak representative body for medical students in Australia. Accordingly, AMSA advocates on issues
transfusion-transmissible HCV and HIV in Italy Haemovigilance and the residual risk of
 Haemovigilance and the residual risk of transfusion-transmissible Giuliano Grazzini, MD National Blood Centre, National Institute of Health, Rome, Italy & International Federation of Blood Donor Organizations
Haemovigilance and the residual risk of transfusion-transmissible Giuliano Grazzini, MD National Blood Centre, National Institute of Health, Rome, Italy & International Federation of Blood Donor Organizations
BLOOD DONATION OR BLOOD DONOR DEFERRAL BASED ON DATA ON NONCONFORMITIES DETECTED IN POST-SELECTION PERIOD (DURING AND AFTER BLOOD COLLECTION)
 BLOOD DONATION OR BLOOD DONOR DEFERRAL BASED ON DATA ON NONCONFORMITIES DETECTED IN POST-SELECTION PERIOD (DURING AND AFTER BLOOD COLLECTION) Tomislav Vuk Croatian Institute of Transfusion Medicine Zagreb
BLOOD DONATION OR BLOOD DONOR DEFERRAL BASED ON DATA ON NONCONFORMITIES DETECTED IN POST-SELECTION PERIOD (DURING AND AFTER BLOOD COLLECTION) Tomislav Vuk Croatian Institute of Transfusion Medicine Zagreb
New Brunswick Report on Sexually Transmitted and Blood Borne Infections, 2016
 New Brunswick Report on Sexually Transmitted and Blood Borne Infections, 6 Table of Contents. Introduction.... Methodology... 3. Data Limitations.... Definitions used... 3 5. Overview of STBBI epidemiology
New Brunswick Report on Sexually Transmitted and Blood Borne Infections, 6 Table of Contents. Introduction.... Methodology... 3. Data Limitations.... Definitions used... 3 5. Overview of STBBI epidemiology
Evaluation of causes of deferral of blood donors in blood bank - A study of 2 years at tertiary care hospital blood bank
 Original Research Article Evaluation of causes of deferral of blood donors in blood bank - A study of 2 years at tertiary care hospital blood bank K. Padma Malini *, Tamil Arasi, K. Sudha, O. Shravan Kumar
Original Research Article Evaluation of causes of deferral of blood donors in blood bank - A study of 2 years at tertiary care hospital blood bank K. Padma Malini *, Tamil Arasi, K. Sudha, O. Shravan Kumar
EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS
 EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits
EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits
PUBLICATION OF THE SUPERIOR HEALTH COUNCIL No. 9291
 PUBLICATION OF THE SUPERIOR HEALTH COUNCIL No. 9291 Sexual risk behaviour and blood donation Part I: Blood donations by MSM This version was validated by the Board on October 2016 1 This advisory report
PUBLICATION OF THE SUPERIOR HEALTH COUNCIL No. 9291 Sexual risk behaviour and blood donation Part I: Blood donations by MSM This version was validated by the Board on October 2016 1 This advisory report
[Submitted Electronically]
![[Submitted Electronically] [Submitted Electronically]](/thumbs/82/85246007.jpg) [Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
[Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING October2010. Issue Summary
 TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING 28-29October2010 Issue Summary Informational Topic: FDA s Geographic Donor Deferral Policy to Reduce the Possible Risk of Transmission
TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING 28-29October2010 Issue Summary Informational Topic: FDA s Geographic Donor Deferral Policy to Reduce the Possible Risk of Transmission
HEPATITIS C, ACUTE CRUDE DATA. Number of Cases 5 Annual Incidence a LA County 0.05 California b 0.10 United States b 0.68 Age at Diagnosis Mean 38
 2016 Annual Morbidity Report HEPATITIS C, ACUTE a Rates calculated based on less than 19 cases or events are considered unreliable b Calculated from: CDC. Notice to Readers: Final 2016 Reports of Nationally
2016 Annual Morbidity Report HEPATITIS C, ACUTE a Rates calculated based on less than 19 cases or events are considered unreliable b Calculated from: CDC. Notice to Readers: Final 2016 Reports of Nationally
DHQ Flowcharts v2.0 eff. February 2016
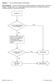 Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Lookback studies to assess viral risks The French experience
 Lookback studies to assess viral risks The French experience 2000-2012 S Laperche 1, MF Leconte des Floris 2, J Pillonel 3, L Hauser 2, C Lefort 2, JY Py 2, I Hervé 2, R Djoudi 2 For the French haemovigilance
Lookback studies to assess viral risks The French experience 2000-2012 S Laperche 1, MF Leconte des Floris 2, J Pillonel 3, L Hauser 2, C Lefort 2, JY Py 2, I Hervé 2, R Djoudi 2 For the French haemovigilance
Persons with Early Syphilis Identified through Blood or Plasma Donation Screening in the United States
 545 CONCISE COMMUNICATION Persons with Early Syphilis Identified through Blood or Plasma Donation Screening in the United States Carolyn Gardella, 1,2 Anthony A. Marfin, 1,a Richard H. Kahn, 1 Emmett Swint,
545 CONCISE COMMUNICATION Persons with Early Syphilis Identified through Blood or Plasma Donation Screening in the United States Carolyn Gardella, 1,2 Anthony A. Marfin, 1,a Richard H. Kahn, 1 Emmett Swint,
Hot Topics in Tissue Safety
 Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Instruction to implement measures for risk minimisation in using HIV-1 NAT test systems
 Paul-Ehrlich-Institut P.O. Box 63207 Langen, Germany To: The marketing authorisation holders of cellular blood products and fresh frozen plasma and the holders of authorisations for stem cell preparations
Paul-Ehrlich-Institut P.O. Box 63207 Langen, Germany To: The marketing authorisation holders of cellular blood products and fresh frozen plasma and the holders of authorisations for stem cell preparations
THE PORTUGUESE COMMUNITY SCREENING NETWORK
 THE PORTUGUESE COMMUNITY SCREENING NETWORK COBATEST Annual Network Meeting 14 May 2018 Presented by: Name goes here Paula Meireles, on behalf of the Community Screening Network EPIUnit ISPUP FUNDING (from
THE PORTUGUESE COMMUNITY SCREENING NETWORK COBATEST Annual Network Meeting 14 May 2018 Presented by: Name goes here Paula Meireles, on behalf of the Community Screening Network EPIUnit ISPUP FUNDING (from
23 November Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD 20852
 23 November 2016 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA-2016-N-1502,
23 November 2016 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA-2016-N-1502,
Blood supply and blood products: Regulatory issues in the pandemic context
 Blood supply and blood products: Regulatory issues in the pandemic context M. Heiden R. Seitz Paul-Ehrlich-Straße 51-59 63225 Langen GERMANY +49 (0) 6103 77 0 +49 (0) 6103 77-1234 Email: pei@pei.de Homepage:
Blood supply and blood products: Regulatory issues in the pandemic context M. Heiden R. Seitz Paul-Ehrlich-Straße 51-59 63225 Langen GERMANY +49 (0) 6103 77 0 +49 (0) 6103 77-1234 Email: pei@pei.de Homepage:
Changing eligibility criteria for MSM: The Canadian Perspective
 Changing eligibility criteria for MSM: The Canadian Perspective Mindy Goldman MD Canadian Blood Services ASFA & WAA Joint Meeting, San Francisco April 4, 2014 Outline - Historical background - International
Changing eligibility criteria for MSM: The Canadian Perspective Mindy Goldman MD Canadian Blood Services ASFA & WAA Joint Meeting, San Francisco April 4, 2014 Outline - Historical background - International
MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE BACKGROUND
 MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE Countries around the world are reviewing the policy of deferring from blood donation men who have had sex with men (MSM). Both Canadian Blood Services
MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE Countries around the world are reviewing the policy of deferring from blood donation men who have had sex with men (MSM). Both Canadian Blood Services
Relationship between first-year blood donation, return rate for subsequent donation and demographic characteristics
 ORIGINAL ARTICLE Relationship between first-year blood donation, return rate for subsequent donation and demographic characteristics Leila Kasraian, Alireza Tavassoli Blood Transfusion Research Centre,
ORIGINAL ARTICLE Relationship between first-year blood donation, return rate for subsequent donation and demographic characteristics Leila Kasraian, Alireza Tavassoli Blood Transfusion Research Centre,
Viral Hepatitis B and C in North African Countries
 Viral Hepatitis B and C in North African Countries Prevalence, Risk factors and How to prevent Prof. Ossama Rasslan President, ESIC ICAN, Vice-Chair ICAN 2014, Harare, Zimbabwe, Nov 3rd 5th Overview Viral
Viral Hepatitis B and C in North African Countries Prevalence, Risk factors and How to prevent Prof. Ossama Rasslan President, ESIC ICAN, Vice-Chair ICAN 2014, Harare, Zimbabwe, Nov 3rd 5th Overview Viral
Transfusion-transmitted Cytomegalovirus
 Transfusion-transmitted Cytomegalovirus Can you confidently abandon CMV seronegative products in the modern era of pre-storage leukoreduction? Jeannie Callum, BA, MD, FRCPC Really? Are we still talking
Transfusion-transmitted Cytomegalovirus Can you confidently abandon CMV seronegative products in the modern era of pre-storage leukoreduction? Jeannie Callum, BA, MD, FRCPC Really? Are we still talking
SEROPREVALENCE OF ANTI-HCV ANTIBODIES AMONG VOLUNTARY BLOOD DONORS IN FATEHABAD DISTRICT OF HARYANA
 SEROPREVALENCE OF ANTI-HCV ANTIBODIES AMONG VOLUNTARY BLOOD DONORS IN FATEHABAD DISTRICT OF HARYANA *Sangwan L., Arun P. 2 and Munjal A. 3 Haryana Civil Medical Service, Fatehabad, Haryana *Author for
SEROPREVALENCE OF ANTI-HCV ANTIBODIES AMONG VOLUNTARY BLOOD DONORS IN FATEHABAD DISTRICT OF HARYANA *Sangwan L., Arun P. 2 and Munjal A. 3 Haryana Civil Medical Service, Fatehabad, Haryana *Author for
Pathogen Safety and BSE / variant CJD
 Pathogen Safety and BSE / variant CJD Thomas R. Kreil, Global Pathogen Safety Parenteral Drug Industry Plasma Protein Industry Summit September 7, 2017; Beijing Pathogen Safety Those who cannot remember
Pathogen Safety and BSE / variant CJD Thomas R. Kreil, Global Pathogen Safety Parenteral Drug Industry Plasma Protein Industry Summit September 7, 2017; Beijing Pathogen Safety Those who cannot remember
patients with blood borne viruses Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled Document Lead:
 CONTROLLED DOCUMENT Procedure for the management of patients with blood borne viruses CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled
CONTROLLED DOCUMENT Procedure for the management of patients with blood borne viruses CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled
Maximizing Cornea and Tissue Donation through Specimen Quality
 Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
REPORT OF THE COUNCIL ON SCIENCE AND PUBLIC HEALTH
 REPORT OF THE COUNCIL ON SCIENCE AND PUBLIC HEALTH (A-0) Revision of the Lifetime Deferral for Blood Donation of the Men Who Have Sex with Men (MSM) Population (Resolution, A-0) (Reference Committee E)
REPORT OF THE COUNCIL ON SCIENCE AND PUBLIC HEALTH (A-0) Revision of the Lifetime Deferral for Blood Donation of the Men Who Have Sex with Men (MSM) Population (Resolution, A-0) (Reference Committee E)
HIV Epidemiology March 7, Stefanie Rhodes Inova Juniper Program
 HIV Epidemiology March 7, 2019 Stefanie Rhodes Inova Juniper Program Stefanie.Rhodes@inova.org Human Immunodeficiency Virus Virus that attacks and weakens immune system Can be treated, but not cured Transmitted
HIV Epidemiology March 7, 2019 Stefanie Rhodes Inova Juniper Program Stefanie.Rhodes@inova.org Human Immunodeficiency Virus Virus that attacks and weakens immune system Can be treated, but not cured Transmitted
Directed Semen Donor Eligibility Requirements
 Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Breastfeeding Center Milk Connection Questionnaire and Release for Donor
 Breastfeeding Center Milk Connection Questionnaire and Release for Donor All of us at The Breastfeeding Center are deeply grateful to you for your important gift to infants in our community. Your donated
Breastfeeding Center Milk Connection Questionnaire and Release for Donor All of us at The Breastfeeding Center are deeply grateful to you for your important gift to infants in our community. Your donated
BLOOD DONATION AFTER ACUPUNCTURE: A JUSTIFIED DEFERRAL?
 BLOOD DONATION AFTER ACUPUNCTURE: A JUSTIFIED DEFERRAL? What if your patients want to donate blood? Are you aware that patients undergoing Acupuncture treatment, according to international guidelines,
BLOOD DONATION AFTER ACUPUNCTURE: A JUSTIFIED DEFERRAL? What if your patients want to donate blood? Are you aware that patients undergoing Acupuncture treatment, according to international guidelines,
Surveillance Report 2014
 Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Routine NAT-screening for West Nile Virus Infections in Germany: Being prepared
 Routine NAT-screening for West Nile Virus Infections in Germany: Being prepared R. Thermann, W. K. Roth IPFA / PEI 18 th international workshop on Surveillance and screening of blood borne pathogens Budapest,
Routine NAT-screening for West Nile Virus Infections in Germany: Being prepared R. Thermann, W. K. Roth IPFA / PEI 18 th international workshop on Surveillance and screening of blood borne pathogens Budapest,
Current Infectious Disease Screening for the Live Organ Donor
 2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director
2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director
risk of Transfusion-Transmitted Transmitted Viral Infections (HIV, HCV and HBV)
 Prevention and ResidualR risk of Transfusion-Transmitted Transmitted Viral Infections (HIV, HCV and HBV) Syria Laperche L (Centre National de référence r rence pour les hépatites h B et C en Transfusion)
Prevention and ResidualR risk of Transfusion-Transmitted Transmitted Viral Infections (HIV, HCV and HBV) Syria Laperche L (Centre National de référence r rence pour les hépatites h B et C en Transfusion)
A cross-sectional study among community on knowledge, attitude and practice about blood donation in Udaipur city of Rajasthan, India
 International Journal of Community Medicine and ublic Health Jain J et al. Int J Community Med ublic Health. 2016 Apr;3(4):952-956 http://www.ijcmph.com pissn 2394-6032 eissn 2394-6040 Research Article
International Journal of Community Medicine and ublic Health Jain J et al. Int J Community Med ublic Health. 2016 Apr;3(4):952-956 http://www.ijcmph.com pissn 2394-6032 eissn 2394-6040 Research Article
DHQ v2.0, February 2016 Table Detailing Changes from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you
 DHQ v2.0, February 2016 Table Detailing s from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you Q 1, No Feeling healthy and well today? Question 2: Are you Q 2, No change Currently taking
DHQ v2.0, February 2016 Table Detailing s from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you Q 1, No Feeling healthy and well today? Question 2: Are you Q 2, No change Currently taking
Question: 1. Are you currently taking an antibiotic?
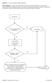 Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Research Article Decreasing Prevalence of Transfusion Transmitted Infection in Indian Scenario
 e Scientific World Journal, Article ID 173939, 4 pages http://dx.doi.org/10.1155/2014/173939 Research Article Decreasing Prevalence of Transfusion Transmitted Infection in Indian Scenario Tulika Chandra,
e Scientific World Journal, Article ID 173939, 4 pages http://dx.doi.org/10.1155/2014/173939 Research Article Decreasing Prevalence of Transfusion Transmitted Infection in Indian Scenario Tulika Chandra,
Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services
 Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services Outline 1 Current criteria Proposed criteria Stakeholder outreach Outline 2 Safety Assessment Results since implementation
Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services Outline 1 Current criteria Proposed criteria Stakeholder outreach Outline 2 Safety Assessment Results since implementation
HIV/AIDS Surveillance Technical Notes
 HIV/AIDS Surveillance Technical Notes Surveillance of HIV/AIDS The Minnesota Department of Health (MDH) collects case reports of HIV infection and AIDS diagnoses through a passive and active HIV/AIDS surveillance
HIV/AIDS Surveillance Technical Notes Surveillance of HIV/AIDS The Minnesota Department of Health (MDH) collects case reports of HIV infection and AIDS diagnoses through a passive and active HIV/AIDS surveillance
Characteristics of Lapsed German Whole Blood Donors and Barriers to Return Four Years after the Initial Donation
 Original Article Originalarbeit Transfus Med Hemother 2012;39:9 15 DOI: 10.1159/000335602 Received: August 17, 2011 Accepted: September 20, 2011 Published online: December 23, 2011 Characteristics of Lapsed
Original Article Originalarbeit Transfus Med Hemother 2012;39:9 15 DOI: 10.1159/000335602 Received: August 17, 2011 Accepted: September 20, 2011 Published online: December 23, 2011 Characteristics of Lapsed
HEALTH SCREENING QUESTIONNAIRE. Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
Ask at Least Annually. Ask Older Adults. Have you been sexually active in the last year? Have you ever been sexually active?
 Essential Sexual Health Questions to Ask Adults Ask all of your adult patients the sexual health questions on this card. They will help you assess the patient s level of sexual risk and determine whether
Essential Sexual Health Questions to Ask Adults Ask all of your adult patients the sexual health questions on this card. They will help you assess the patient s level of sexual risk and determine whether
Cord Blood Medical History/ Health Assessment Questionnaire
 OTT BRM EDM VAN Date: YYYY / MM / DD Phone Interview (if applicable) Informed Consent on file * Mandatory fields GENERAL INFORMATION *Last Name As per government ID *First Name As per government ID *DOB
OTT BRM EDM VAN Date: YYYY / MM / DD Phone Interview (if applicable) Informed Consent on file * Mandatory fields GENERAL INFORMATION *Last Name As per government ID *First Name As per government ID *DOB
Linkage of hepatitis and HIV surveillance systems to improve completeness of injection drug use risk data for co-infected Floridians
 Linkage of hepatitis and HIV surveillance systems to improve completeness of injection drug use risk data for co-infected Floridians Shana Geary, MPH, CPH United States Conference on AIDS September 7,
Linkage of hepatitis and HIV surveillance systems to improve completeness of injection drug use risk data for co-infected Floridians Shana Geary, MPH, CPH United States Conference on AIDS September 7,
ANNALS OF HEALTH LAW Advance Directive VOLUME 23 SPRING 2014 PAGES
 ANNALS OF HEALTH LAW Advance Directive VOLUME 23 SPRING 2014 PAGES 101-112 United States Blood Donor Policy on Gay Men: Adopting an Italian Individual Risk Assessment Policy Melissa Kong* I. INTRODUCTION
ANNALS OF HEALTH LAW Advance Directive VOLUME 23 SPRING 2014 PAGES 101-112 United States Blood Donor Policy on Gay Men: Adopting an Italian Individual Risk Assessment Policy Melissa Kong* I. INTRODUCTION
Exploring the risks of liver cancer after successful treatment for hepatitis C virus
 CATIE-News CATIE s bite-sized HIV and hepatitis C news bulletins. Exploring the risks of liver cancer after successful treatment for hepatitis C virus 11 June 2013 In Canada and other high-income countries,
CATIE-News CATIE s bite-sized HIV and hepatitis C news bulletins. Exploring the risks of liver cancer after successful treatment for hepatitis C virus 11 June 2013 In Canada and other high-income countries,
Version for the Silent Procedure 29 April Agenda item January Hepatitis
 Version for the Silent Procedure 29 April 2014 134th session EB134.R18 Agenda item 10.5 25 January 2014 Hepatitis The Executive Board, Having considered the report on hepatitis, 1 RECOMMENDS to the Sixty-seventh
Version for the Silent Procedure 29 April 2014 134th session EB134.R18 Agenda item 10.5 25 January 2014 Hepatitis The Executive Board, Having considered the report on hepatitis, 1 RECOMMENDS to the Sixty-seventh
ANALYSIS OF BLOOD DONOR DEFERRAL CHARACTERISTICS IN DELHI, INDIA
 ANALYSIS OF BLOOD DONOR DEFERRAL CHARACTERISTICS IN DELHI, INDIA Shalini Bahadur 1, Sonal Jain 1, Ruchika K Goel 1, Sangeeta Pahuja 2 and Manjula Jain 1 1 Department of Pathology, 2 Blood Bank, Lady Hardinge
ANALYSIS OF BLOOD DONOR DEFERRAL CHARACTERISTICS IN DELHI, INDIA Shalini Bahadur 1, Sonal Jain 1, Ruchika K Goel 1, Sangeeta Pahuja 2 and Manjula Jain 1 1 Department of Pathology, 2 Blood Bank, Lady Hardinge
The current status of PrEP in Europe. Pep Coll AIDS Research Institute-IrsiCaixa Fight AIDS Foundation BCN Checkpoint
 The current status of PrEP in Europe Pep Coll AIDS Research Institute-IrsiCaixa Fight AIDS Foundation BCN Checkpoint DISCLOSURES I have received a research grant from Gilead Sciences awarded to my institution
The current status of PrEP in Europe Pep Coll AIDS Research Institute-IrsiCaixa Fight AIDS Foundation BCN Checkpoint DISCLOSURES I have received a research grant from Gilead Sciences awarded to my institution
EDMA HIV-AIDS TEAM Fact Sheet November 2007
 EDMA HIV-AIDS TEAM Fact Sheet November 2007 1. HIV Facts AIDS epidemic update UNAIDS Epidemic Update, November 2007 (1) 760,000 people to be living with HIV in Western and Central Europe in 2007. 31,000
EDMA HIV-AIDS TEAM Fact Sheet November 2007 1. HIV Facts AIDS epidemic update UNAIDS Epidemic Update, November 2007 (1) 760,000 people to be living with HIV in Western and Central Europe in 2007. 31,000
The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P
 The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P Record Status This is a critical abstract of an economic evaluation that meets
The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P Record Status This is a critical abstract of an economic evaluation that meets
Transfusion-transmitted HIV from seroconverted blood donors has not been identified in England since the introduction of HIV NAT screening
 Transfusion-transmitted HIV from seroconverted blood donors has not been identified in England since the introduction of HIV NAT screening Bhavita Vishram Timeline of introduction of microbiological tests
Transfusion-transmitted HIV from seroconverted blood donors has not been identified in England since the introduction of HIV NAT screening Bhavita Vishram Timeline of introduction of microbiological tests
Government of Canada Federal AIDS Initiative Milestones
 HIV in Canada: Trends and Issues for Advancing Prevention, Care, Treatment and Support Through Knowledge Exchange Michael R Smith, Senior Policy Advisor, Programs and Coordination Division, Centre for
HIV in Canada: Trends and Issues for Advancing Prevention, Care, Treatment and Support Through Knowledge Exchange Michael R Smith, Senior Policy Advisor, Programs and Coordination Division, Centre for
Egg and Sperm Bank Edinburgh Fertility & Reproductive Endocrine Centre (EFREC) DONOR INFORMATION Screening Tests
 Egg and Sperm Bank Edinburgh Fertility & Reproductive Endocrine Centre (EFREC) DONOR INFORMATION Screening Tests Introduction As a potential donor, it is important you are aware of the screening tests
Egg and Sperm Bank Edinburgh Fertility & Reproductive Endocrine Centre (EFREC) DONOR INFORMATION Screening Tests Introduction As a potential donor, it is important you are aware of the screening tests
Transfusion Transmissible Infections among Voluntary Blood Donors at the University Teaching Hospital, Lusaka, Zambia
 ORIGINAL ARTICLE Transfusion Transmissible Infections among Voluntary Blood Donors at the University Teaching Hospital, Lusaka, Zambia,2,3 2 3 3 D Chama, Y Ahmed, KS Baboo, H Halwindi, J Mulenga, 2 Zambia
ORIGINAL ARTICLE Transfusion Transmissible Infections among Voluntary Blood Donors at the University Teaching Hospital, Lusaka, Zambia,2,3 2 3 3 D Chama, Y Ahmed, KS Baboo, H Halwindi, J Mulenga, 2 Zambia
CALM LEARNER OUTCOMES 1 :
 CALM STI AND HIV LESSON 2 STI AND HIV Lesson 2 GRADE CALM LEARNER OUTCOMES 1 : Examine aspects of healthy sexuality, sexual wellness and responsible sexual behaviour. Describe sexually healthy choices
CALM STI AND HIV LESSON 2 STI AND HIV Lesson 2 GRADE CALM LEARNER OUTCOMES 1 : Examine aspects of healthy sexuality, sexual wellness and responsible sexual behaviour. Describe sexually healthy choices
Overview of Blood Transfusion System of Iran:
 Report Article Overview of Blood Transfusion System of Iran: 2002-2011 AM Cheraghali High Institute for Research and Education on Transfusion Medicine, Iran Blood Transfusion Organization and University
Report Article Overview of Blood Transfusion System of Iran: 2002-2011 AM Cheraghali High Institute for Research and Education on Transfusion Medicine, Iran Blood Transfusion Organization and University
2014 Surveillance Report. Transfusion-transmissible infections in Australia
 Transfusion-transmissible infections in Australia 2014 Surveillance Report This publication is available online at: http://www.kirby.unsw.edu.au and http://www.transfusion.com.au Recommended citation:
Transfusion-transmissible infections in Australia 2014 Surveillance Report This publication is available online at: http://www.kirby.unsw.edu.au and http://www.transfusion.com.au Recommended citation:
Hepatitis Case Investigation
 * indicates required fields Does patient also have: Hepatitis Case Investigation West Virginia Electronic Disease Surveillance System Division of Surveillance and Disease Control Infectious Disease Epidemiology
* indicates required fields Does patient also have: Hepatitis Case Investigation West Virginia Electronic Disease Surveillance System Division of Surveillance and Disease Control Infectious Disease Epidemiology
2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies
 2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies Submitted by: Malaysia Policy Dialogue and Workshop on Attaining a Safe and Sustainable Blood Supply Chain Manila, Philippines 30 September 1
2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies Submitted by: Malaysia Policy Dialogue and Workshop on Attaining a Safe and Sustainable Blood Supply Chain Manila, Philippines 30 September 1
Epidemiology of hepatitis B in The Netherlands
 Viral Hepatitis Prevention Board Meeting Rotterdam, The Netherlands, November 13-14, 2008 National Institute for Public Health and the Environment Epidemiology of hepatitis B in The Netherlands Mirjam
Viral Hepatitis Prevention Board Meeting Rotterdam, The Netherlands, November 13-14, 2008 National Institute for Public Health and the Environment Epidemiology of hepatitis B in The Netherlands Mirjam
BC Enhanced Hepatitis Strain Surveillance Project Hepatitis B. Acute HBV Questionnaire
 BC Enhanced Hepatitis Strain Surveillance Project Hepatitis B Informed Consent: Please read to individual and answer any questions There are about 60,000 people with hepatitis B in BC. We want to find
BC Enhanced Hepatitis Strain Surveillance Project Hepatitis B Informed Consent: Please read to individual and answer any questions There are about 60,000 people with hepatitis B in BC. We want to find
RAPID BACTERIAL DETECTION METHODS FOR PLATELETS. M. Schmidt
 RAPID BACTERIAL DETECTION METHODS FOR PLATELETS M. Schmidt Deutsches Rotes Kreuz Blutspendedienst Baden-Württemberg-Hessen Goethe Universität Frankfurt am Main Institut für Transfusionsmedizin und Immunhämatologie
RAPID BACTERIAL DETECTION METHODS FOR PLATELETS M. Schmidt Deutsches Rotes Kreuz Blutspendedienst Baden-Württemberg-Hessen Goethe Universität Frankfurt am Main Institut für Transfusionsmedizin und Immunhämatologie
HIV/AIDS. Kuna High School Mr. Stanley
 HIV/AIDS Kuna High School Mr. Stanley Questions 1. Write an example of how your immune system helps prevent you from getting diseases. Terms to know Epidemic - a widespread occurrence of an infectious
HIV/AIDS Kuna High School Mr. Stanley Questions 1. Write an example of how your immune system helps prevent you from getting diseases. Terms to know Epidemic - a widespread occurrence of an infectious
Epidemiology of Hepatitis C Iran
 Seyed Moayed Alavian M.D. Professor of Gastro & Hepatology Editor-in-chief of Glob Hepat Comm Editor-in-chief of Hepatitis Monthly E mail: editor@hepatmon.com Epidemiology of Hepatitis C Iran Epidemiology
Seyed Moayed Alavian M.D. Professor of Gastro & Hepatology Editor-in-chief of Glob Hepat Comm Editor-in-chief of Hepatitis Monthly E mail: editor@hepatmon.com Epidemiology of Hepatitis C Iran Epidemiology
HEALTH SCREENING QUESTIONNAIRE CT Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE CT Work-up Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca
HEALTH SCREENING QUESTIONNAIRE CT Work-up Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca
Question: 1. Are you currently taking an antibiotic?
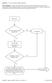 Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey
 Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Understanding the spread of HIV among men who have sex with men in Belgium: results from online and on site special surveys
 Understanding the spread of HIV among men who have sex with men in Belgium: results from online and on site special surveys Wim Vanden Berghe ITM HIV/AIDS center Department of Public Health Institute of
Understanding the spread of HIV among men who have sex with men in Belgium: results from online and on site special surveys Wim Vanden Berghe ITM HIV/AIDS center Department of Public Health Institute of
Formative Research results
 FRR Annex 5: Presentation of the FR results in the project meeting, Berlin, 11. 13.02.2013 Formative Research results Sandra Dudareva Vizule, Ulrich Marcus Robert Koch Institute 11 February 2013 Content
FRR Annex 5: Presentation of the FR results in the project meeting, Berlin, 11. 13.02.2013 Formative Research results Sandra Dudareva Vizule, Ulrich Marcus Robert Koch Institute 11 February 2013 Content
Canada s Blood Donation Eligibility Criteria for Men Who Have Sex with Men
 Criteria for Men Who Have Sex with Men Dr. Graham D. Sher Chief Executive Officer, Canadian Blood Services Dr. Dana Devine Chief Medical and Scientific Officer, Canadian Blood Services Executive summary
Criteria for Men Who Have Sex with Men Dr. Graham D. Sher Chief Executive Officer, Canadian Blood Services Dr. Dana Devine Chief Medical and Scientific Officer, Canadian Blood Services Executive summary
Preparedness for candidate HCV vaccine trials in people who inject. C Vaccine Initiative*
 Preparedness for candidate HCV vaccine trials in people who inject drugs: Challenges and opportunities Li M h b h lf f th UNSW H titi Lisa Maher, on behalf of the UNSW Hepatitis C Vaccine Initiative* Background
Preparedness for candidate HCV vaccine trials in people who inject drugs: Challenges and opportunities Li M h b h lf f th UNSW H titi Lisa Maher, on behalf of the UNSW Hepatitis C Vaccine Initiative* Background
The new German strategy on HIV, Hepatitis B, C and STI, an integrated approach. Ines Perea Ministry of Health, Germany
 The new German strategy on HIV, Hepatitis B, C and STI, an integrated approach Ines Perea Ministry of Health, Germany Reasons for a new strategy in 2016 New international agreements (SDG s) Renewed political
The new German strategy on HIV, Hepatitis B, C and STI, an integrated approach Ines Perea Ministry of Health, Germany Reasons for a new strategy in 2016 New international agreements (SDG s) Renewed political
Original article J Bas Res Med Sci 2017; 4(2):4-8.
 Prevalence of HIV, hepatitis B and C infections among volunteer blood donors at the blood transfusion center of Ilam city, Iran Hasan Boustani 1, Enayat Anvari 2, Sedighe Saiadi Sartang 2, Mehdi Omidi
Prevalence of HIV, hepatitis B and C infections among volunteer blood donors at the blood transfusion center of Ilam city, Iran Hasan Boustani 1, Enayat Anvari 2, Sedighe Saiadi Sartang 2, Mehdi Omidi
NATIONAL BLOOD TRANSFUSION SERVICES STRATEGY
 FEDERAL DEMOCRATIC REPUBLIC OF ETHIOPIA MINISTRY OF HEALTH NATIONAL BLOOD TRANSFUSION SERVICES STRATEGY February 2005 Addis Ababa Ethiopia Acknowledgement The Ministry of Health of the Federal Democratic
FEDERAL DEMOCRATIC REPUBLIC OF ETHIOPIA MINISTRY OF HEALTH NATIONAL BLOOD TRANSFUSION SERVICES STRATEGY February 2005 Addis Ababa Ethiopia Acknowledgement The Ministry of Health of the Federal Democratic
WHO Blood Regulators Network (BRN)
 Distribution: General English only WHO Blood Regulators Network (BRN) Position Paper on Collection and Use of Convalescent Plasma or Serum as an Element in Middle East Respiratory Syndrome Coronavirus
Distribution: General English only WHO Blood Regulators Network (BRN) Position Paper on Collection and Use of Convalescent Plasma or Serum as an Element in Middle East Respiratory Syndrome Coronavirus
Source of platelet concentrates FACHBEREICH MEDIZIN
 FACHBEREICH MEDIZIN Efficacy and safety of platelet concentrates from platelet apheresis donations or whole blood donations Gregor Bein Institute for Clinical Immunology and Transfusion Medicine Justus-Liebig-University
FACHBEREICH MEDIZIN Efficacy and safety of platelet concentrates from platelet apheresis donations or whole blood donations Gregor Bein Institute for Clinical Immunology and Transfusion Medicine Justus-Liebig-University
Epidemiology of Acute Hepatitis C Infection in Canada Results from the Enhanced Hepatitis Strain Surveillance System (EHSSS)
 Epidemiology of Acute Hepatitis C Infection in Canada Results from the Enhanced Hepatitis Strain Surveillance System (EHSSS) At a Glance Reported rates of acute HCV declined from.5 per, population in to.
Epidemiology of Acute Hepatitis C Infection in Canada Results from the Enhanced Hepatitis Strain Surveillance System (EHSSS) At a Glance Reported rates of acute HCV declined from.5 per, population in to.
June 8, Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD 20852
 June 8, 2018 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA 2016 D 0545,
June 8, 2018 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA 2016 D 0545,
Updat Dent. Coll.j 2016;6(2):21-26
 Updat Dent. Coll.j 2016;6(2):21-26 Original article: Awareness about blood among donors at a specialized tertiary level public hospital. Dr. Begum Sharifa Akhtar 1, *Dr. Nihar Sultana 2, Dr. Saieda Farzana
Updat Dent. Coll.j 2016;6(2):21-26 Original article: Awareness about blood among donors at a specialized tertiary level public hospital. Dr. Begum Sharifa Akhtar 1, *Dr. Nihar Sultana 2, Dr. Saieda Farzana
TGA regulation of local manufacturers of plasma derivatives CSL Behring Australia s overview
 IPFA Workshop Session 7: REGULATORY CONSIDERATIONS TGA regulation of local manufacturers of plasma derivatives CSL Behring Australia s overview Mary Lavithis, CSL Behring Australia Presented at: IPFA Asia
IPFA Workshop Session 7: REGULATORY CONSIDERATIONS TGA regulation of local manufacturers of plasma derivatives CSL Behring Australia s overview Mary Lavithis, CSL Behring Australia Presented at: IPFA Asia
Transfusion Risk for Hepatitis B, Hepatitis C and HIV in the State of Santa Catarina, Brazil,
 236 BJID 2004; 8 (June) Transfusion Risk for Hepatitis B, Hepatitis C and HIV in the State of Santa Catarina, Brazil, 1991-2001 Emil Kupek Department of Public Health, Federal University of Santa Catarina,
236 BJID 2004; 8 (June) Transfusion Risk for Hepatitis B, Hepatitis C and HIV in the State of Santa Catarina, Brazil, 1991-2001 Emil Kupek Department of Public Health, Federal University of Santa Catarina,
Transmission/Prevention
 Transmission/Prevention Section Three Transmission/Prevention Hepatitis C is transmitted by blood-to-blood contact. Any break in the skin may allow HCV to enter the body, even if no blood is visible. The
Transmission/Prevention Section Three Transmission/Prevention Hepatitis C is transmitted by blood-to-blood contact. Any break in the skin may allow HCV to enter the body, even if no blood is visible. The
Jackie Williams BBV/Sexual Health Trainer
 Jackie Williams BBV/Sexual Health Trainer HEPATITIS The Basics What does your liver do? FUNCTIONS n storage of substances - glycogen, iron and vitamins n disposal of metabolic wastes - urea and bile n
Jackie Williams BBV/Sexual Health Trainer HEPATITIS The Basics What does your liver do? FUNCTIONS n storage of substances - glycogen, iron and vitamins n disposal of metabolic wastes - urea and bile n
Overall Blood Supply Strategy with Regard to Variant Creutzfeldt-Jakob Disease (vcjd)
 Vol. 33, Supplement 2, December 2006 Overall Blood Supply Strategy with Regard to Variant Creutzfeldt-Jakob Disease (vcjd) Report of the Working Group Commissioned by the German Federal Ministry of Health
Vol. 33, Supplement 2, December 2006 Overall Blood Supply Strategy with Regard to Variant Creutzfeldt-Jakob Disease (vcjd) Report of the Working Group Commissioned by the German Federal Ministry of Health
Modernization of North Carolina s HIV control measures
 Modernization of North Carolina s HIV control measures North Carolina Division of Public Health Victoria Mobley, MD MPH Evelyn Foust, MPH CPM Agenda Introduction Summary of scientific evidence used to
Modernization of North Carolina s HIV control measures North Carolina Division of Public Health Victoria Mobley, MD MPH Evelyn Foust, MPH CPM Agenda Introduction Summary of scientific evidence used to
IN VITRO FERTILIZATION
 FERTILITY AND STERILITY VOL. 76, NO. 1, JULY 2001 Copyright 2001 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. IN VITRO FERTILIZATION
FERTILITY AND STERILITY VOL. 76, NO. 1, JULY 2001 Copyright 2001 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. IN VITRO FERTILIZATION
The Blood Donor: Demographics, Donor Selection and Tests on Donor Blood. Liz Caffrey, Patricia Hewitt and John Barbara
 CHAPTER 1 The Blood Donor: Demographics, Donor Selection and Tests on Donor Blood Liz Caffrey, Patricia Hewitt and John Barbara 0.3 OVERVIEW Total 0.254 0.247 A safe and sufficient blood supply depends
CHAPTER 1 The Blood Donor: Demographics, Donor Selection and Tests on Donor Blood Liz Caffrey, Patricia Hewitt and John Barbara 0.3 OVERVIEW Total 0.254 0.247 A safe and sufficient blood supply depends
INTEGRATED HCV SCREENING AND TESTING IN SUBSTANCE ABUSE TREATMENT
 INTEGRATED HCV SCREENING AND TESTING IN SUBSTANCE ABUSE TREATMENT HEPATITIS Hepatitis is the name for a family of viral infections that affect the liver. The most common types are Hepatitis A, B, and C.
INTEGRATED HCV SCREENING AND TESTING IN SUBSTANCE ABUSE TREATMENT HEPATITIS Hepatitis is the name for a family of viral infections that affect the liver. The most common types are Hepatitis A, B, and C.
