Adaptive Trial Design and Incorporation of Biomarkers to Maximize Achievable Objectives. In Early Phase Clinical Studies
|
|
|
- Emory Harvey
- 5 years ago
- Views:
Transcription
1 Adaptive Trial Design and Incorporation of Biomarkers to Maximize Achievable Objectives In Early Phase Clinical Studies
2 Exclusive Offer for Attendees! Stay tuned until after the webinar to receive details on our exclusive offer for webinar attendees!
3 Polling Question #1
4 Key Objectives Over the course of this webinar, we will aim to: Discuss adaptive designs and strategies for incorporation in early phase studies. Discuss the utility of biomarker evaluation and its influence on drug development in early stages. Present relevant examples from previous WCCT programs in which the aforementioned strategies were implemented. Address the relevant statistical issues that arise in this setting, and discuss strategies to ensure that valid statistical inferences can be drawn for each of the objectives.
5 Adaptive Clinical Trial Design
6 Adaptive Clinical Trial Design FDA (2010): An adaptive design clinical study is defined as a study that includes a prospectively planned opportunity for modification of one or more specified aspects of the study design and hypotheses based on analysis of data (usually interim data) from subjects in the study. Are our assumptions correct? Is the study worth continuing, or in need of modifications?
7 Adaptive Clinical Trial Design Structure of an Adaptive Design Scope of Adaptations Stop trial early Resize the trial Modify endpoints, etc. Event rate Effect size Variability, etc. Assumptions to Check Adaptive Design Decision Rules Frequentist/Bayesian Blinded/Unblinded Probability-based Control type 1 error Combine before/after info Valid Inference Trial Integrity Study governance Objective, blinded assessment
8 Bayesian Decision Rule Historical Data Publications Expert Knowledge Average of Past & Present info Contextual Evidence + Observed Data = Updated Evidence Prior Likelihood Posterior
9 Adaptive Designs in Early Phase Trials Scopes Futility Dose Finding Adaptive randomization Sample-size Re-estimation Enrichment Seamless Phase 2/3
10 Futility via Conditional Power Are we going anywhere? At interim, calculate the probability of success, given the data so far. If the probability is low then stop the study. Can be accomplished by a frequentist or a Bayesian calculation. No type-1 error penalty Need to consider: Expected sample size vs. Max sample size
11 Adaptive Dose Finding A host of designs: Goal is to identify the maximum tolerable dose (MTD) Design choices: Escalating Cohort Design: Assign 6 subjects to dose 1 If toxicity < 0.2 (<= 1 DLT in 6 subjects) then assign 6 new subjects to dose 2 Otherwise stop, and declare MTD at lower dose 3+3 Design Assign 3 subjects to dose 1 If 0 DLT in 3 subjects then assign 6 new subjects to dose 2 If 1 DLT in 3 subjects then add 3 new subjects to dose 1 1/6 go to dose 2 2 or more stop, and declare MTD at lowerdose If 2-3 DLT in first 3, then stop, and declare MTD at lower dose converges on MTD defined with Pr [DLT] = 20% That is changeable (e.g. for a target Pr [DLT] = 10%, one can use a 5+5 Design
12 Adaptive Dose Finding Additional Design Choices: Up and Down Design Search can go in both directions Continual Re-assessment Method (CRM) MTD -= a dose associated with Pr [DLT] = x% Model-based
13 Adaptive Dose Escalation 1. Select a mathematical model to describe the relationship between dose and PR [DLT] 2. After each patient, update the model,and estimate the probability of toxicity for each dose level 3. Treat the next patient at the dose who estimate is closes to some pre-specified target, for example, 20% 4. Stop when a maximum sample size is reached Probability of DLT 20% MTD Dose
14 Adaptive Randomization Updating Treatment Assignments Baseline adaptive randomization A large number of stratification variables Balancing treatment arms for all stratification variables is impossible Balance marginally Adaptive minimization Response adaptive randomization Based urn models Biomarker-adaptive randomization
15 Polling Question #2
16 Incorporation of Biomarkers
17 Utility of Biomarkers In Early Phase Clinical Trials Increase likelihood for success Evaluate the population who can benefit Exclude population with off-target effects Multiple barometers of PD Better define mechanism of action More clearly understand disease Identify targets for future development Strongly encouraged by regulators Personalized medicine FDA Biomarker Qualification Program
18 Biomarker Categories Each biomarker category can have a variety of Context of Uses (e.g., a prognostic biomarker can be used for patient stratification of enrichment in clinical trials). Diagnostic Patient Selection Detect a change in the degree or extent of a disease Biomarker Categories* Monitoring Predictive Prognostic Pharmacodynamic /Response Safety Indicate toxicity or assess safety Provide evidence of exposure Identify individuals on basis of effect from a specific intervention or exposure Stratify Patients Enrichment: inclusion/exclusion data Efficacy biomarker/surrogate endpoint Show biological response related to an intervention/exposure Indicate the presence or extent of toxicity related to an intervention or exposure Context of Use Examples Susceptibility/ Risk Indicate the potential for developing a disease or sensitivity to an exposure *Source: FDA Biomarker Qualification Program
19 Biomarker Categories Diagnostic Monitoring Biomarker Categories Predictive Prognostic Pharmacodynamic /Response Safety Susceptibility/ Risk
20 Biomarker Categories Biomarker Categories Diagnostic Monitoring Predictive Prognostic Pharmacodynamic /Response Safety Susceptibility/ Risk Use in Trial Design If the evidence suggests that the benefit of a treatment is limited to the biomarker-positive sub-population, an enrichment design strategy with only biomarker-positive patients may be appropriate If there is sufficient reason to suggest that a biomarker can predict that therapy will be more effective in biomarkerpositive patients, but the evidence is not compelling enough to rule out clinical efficacy in biomarker-negative patients, a biomarker-stratified trial design or an adaptive enrichment trial design may be more appropriate In the biomarker-stratified trial design, biomarkers are used to guide analysis but not treatment assignment In the adaptive enrichment trial design, biomarkers are used to guide the enrollment and not treatment assignment
21 Enrichment Design Assess Biomarker Biomarker Positive Treatment A Treatment B Biomarker Negative Off study
22 Biomarker-Stratified Design Biomarker Positive Treatment A Treatment B Assess Biomarker Biomarker Negative Treatment A Treatment B
23 Case Studies
24 Case Study ALS Pilot Trial In this Amytrophic Lateral Sclerosis pilot study, over 10 biomarkers were measured across five different biomarker categories: Biomarker Categories* Diagnostic Monitoring Predictive Prognostic Pharmacodynamic /Response Safety Susceptibility/ Risk 4 ALS Target biomarkers across 2 modalities (CSF & Plasma) SOD1, phosphorylated neurofilament heavy chain (pnfh), total tau, and phosphorylated tau 4 efficacy biomarkers/surrogate endpoints ALS Functional Rating Scale (ALSFRS-R), Force Vital Capacity (FVC), Time Up and Go (TUG), and Hand-Held Dynamometry (HHD) Several safety biomarkers including QT measurement and hematology parameters
25 Multiparameter Technologies Maximizing Potential for Biomarker Discover in Early Phase Trials Proteomics Single-cell proteomics (FACS, CyTOF) Metabolomics Metabolism-related small molecules Multiplexed immunoassays Cytokines Chemokines Growth factors Microbiome 18S rrna sequencing Genetics Whole genome sequencing mrna expression Single-cell sequencing
26 Case Study: Biomarker Discovery Influenza Challenge Study A Phase I, Open-label, Ascending Dose Study to Determine the Safety and Reactogenicity of a Wild Type Seasonal A/California/H1N Influenza Challenge Virus in Healthy Volunteers, Following a Single Intranasal Administration. Study Population: Normal Healthy Volunteers Main Inclusion Criteria: Absent or low levels of detectable pre-existing antibodies to influenza virus subtypes, the minimum being subjects who have undetectable or low levels of antibody to the potential challenge strain, as determined by a hemagglutination-inhibition (HAI) titer of 10 prior to challenge. Subjects not to have received any influenza vaccine for the previous 2 years. Subjects Enrolled: 36 subjects Study Objective: Determine the dose with the optimal safety profile and infectivity rate of the viral challenge strain for use in subsequent challenge intervention studies to test potential influenza vaccines and/or therapeutics. Additionally, the study aimed to determine immunological responses over the study period, including humoral and cellular immune responses to challenge virus.
27 Influenza Challenge Model At WCCT Virus Strain Clinical isolate from 3-year-old during the 2009 flu season cgmp manufactured in SPF eggs Extensive adventitious virus testing Pre-clinical safety established in ferrets For more information, please refer to handout entitled WCCT Global Influenza Challenge Model Symptoms and Virus Shedding Symptom score (arbitrary units) * * Virus Titer (Log 10 copies/ml) Virus shedding Symptoms (virus shedders) Symptoms (non-shedders) Atomizer used for nasal virus dosing Virus inoculation
28 Influenza Challenge Model Quantifying Cellular Immune Responses N=36 Volunteers CyTOF: 40+ parameter single-cell proteomic analysis Peripheral Blood Samples Day: Inoculation with A/California/H1N influenza virus
29 CyTOF 40+ Parameter Single-Cell Proteomic Analysis
30 CyTOF: Simultaneous Quantification of Multiple Immune Cell Subsets a Study day: MONOCYTES cmc intmc ncmc pdc mdc VIRUS NONE VIRUS NONE Study day: LYMPHOCYTES B cells B cells Naïve B cells NCSM B cells CSM B cells plasma T cells CD4+ T cells CD4+ Naïve T cells CD4+ CM T cells CD4+ Effector T cells CD4+ EM T cells CD 8+ T cells CD 8+ Naïve T cells CD 8+ CM T cells CD 8+ Effector T cells CD 8+ EM NK cells NK cells CD Row max (ABS)
31 CyTOF: Simultaneous Quantification of Multiple Immune Cell Subsets a Study day: MONOCYTES cmc intmc ncmc pdc mdc VIRUS NONE VIRUS NONE Study day: LYMPHOCYTES B cells B cells Naïve B cells NCSM B cells CSM B cells plasma T cells CD4+ T cells CD4+ Naïve T cells CD4+ CM T cells CD4+ Effector T cells CD4+ EM T cells CD 8+ T cells CD 8+ Naïve T cells CD 8+ CM T cells CD 8+ Effector T cells CD 8+ EM NK cells NK cells CD Row max (ABS)
32 CyTOF Computationally-driven Cell Clustering with SCAFFoLD CD8+ Tcells CD4+ Tcells NK cells Basophils Granulocytes ncmcs pdcs cmcs B cells mdcs
33 CyTOF Computationally-driven Cell Clustering with SCAFFoLD CD8+ Tcells CD4+ Tcells CD19 NK cells Basophils Granulocytes ncmcs pdcs cmcs B cells mdcs Median expression MIN 0 MAX
34 CyTOF Computationally-driven Cell Clustering with SCAFFoLD CD8+ Tcells CD4+ Tcells CD8 NK cells Basophils Granulocytes ncmcs pdcs cmcs B cells mdcs Median expression MIN 0 MAX
35 CyTOF Computationally-driven Cell Clustering with SCAFFoLD CD4+ Tcells NK cells CD8+ Tcells Basophils Fold vs. Baseline (log 2 ) SCAFFOLD cluster * Day ncmcs Granulocytes CD14+CD16+ Monocyte like cells pdcs cmcs B cells mdcs
36 Monocyte Responses During Influenza Challenge
37 Monocyte Responses During Influenza Challenge CD14+CD16+ Monocytes are an early biomarker for total symptoms, peak virus titer, and development of T cell responses during influenza Symptoms Virus titer Activated T cells Total symptoms (arbitrary units) 4 r = 0.37 p = Peak virus titer (log10) r = 0.72 p= < Day 8 CD8+CD38+ T cells r = 0.79 p =< CD14+CD16+ Mono (Day 6) /baseline (log 2 ) CD16+CD14+ Mono (Day 5)/baseline CD14+CD16+ Mono (Day 5)/baseline
38 Mapping Disease Course Applying Multivariate Analysis of Biomarkers 1.1 Bcells 1.1 Tcells 6.5 intmcs Fold vs. Baseline * Day * Day * Day Fold vs. Baseline PlasmaBcells * Day CD38+Ki67+CD8+Tcells * Day Basophils * Day
39 Mapping Disease Course Applying Multivariate Analysis of Biomarkers 1.1 Bcells 1.1 Tcells 6.5 intmcs Fold vs. Baseline * Day * Day * Day Fold vs. Baseline PlasmaBcells * Day CD38+Ki67+CD8+Tcells * Day Basophils * Day
40 Mapping Disease Course Applying Multivariate Analysis of Biomarkers Bcells Tcells intmcs PlasmaBcells CD38+ki67+CD8+Tcells Basophils
41 Mapping Disease Course Applying Multivariate Analysis of Biomarkers Bcells Tcells intmcs PlasmaBcells CD38+ki67+CD8+Tcells Basophils
42 Multivariate Predictors of Disease Recent Literature A Four-Biomarker Blood Signature Discriminates Systemic Inflammation Due to Viral Infection Versus Other Etiologies -Scientific Reports 2017 Transcriptomic signatures differentiate survival from fatal outcomes in humans infected with Ebola virus Genome Biology 2017 Integrated, Multi-cohort Analysis Identifies Conserved Transcriptional Signatures across Multiple Respiratory Viruses Immunity 2015 A comprehensive time-course based multicohort analysis of sepsis and sterile inflammation reveals a robust diagnostic gene set Science Translational Medicine 2015 An Integrated Clinico-Metabolomic Model Improves Prediction of Death in Sepsis -Science Translational Medicine 2013 An immune clock of human pregnancy Science Immunology 2017 Lipidomic Profiling of Influenza Infection Identifies Mediators that Induce and Resolve Inflammation Cell 2013
43 Statistical Considerations
44 Challenges with Statistical Analysis Controlling false positive results More advanced in hypothesis testing area Less understood in estimation and selection type problems Validation of biomarkers Easier for prognostic markers Difficult for predictive markers in small trials Need multiple studies to test surrogacy Pattern recognition and dimensionality Computer-aided methods Multivariate analysis
45 Polling Question #3
46 Exclusive Offer for Attendees! WCCT is offering up to 4 hours of consulting time with our experts on your study design or clinical development plan. This will include: 1hr. Discovery Call PROTOCOL 2hrs. For research, planning, and design 1hr. Review and presentation
47 Kalyan Ghosh, VP Biostatistics Dave McIlwain, Scientific & Medical Affairs SME (657)
System Biology analysis of innate and adaptive immune responses during HIV infection
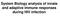 System Biology analysis of innate and adaptive immune responses during HIV infection Model of T cell memory persistence and exhaustion Naive Ag+APC Effector TEM (Pfp, Gr.B, FasL, TNF) Ag stim. IL-2, IL-7,
System Biology analysis of innate and adaptive immune responses during HIV infection Model of T cell memory persistence and exhaustion Naive Ag+APC Effector TEM (Pfp, Gr.B, FasL, TNF) Ag stim. IL-2, IL-7,
Clinical Trial Design to Expedite Drug Development Mary W. Redman, Ph.D.
 Clinical Trial Design to Expedite Drug Development Mary W. Redman, Ph.D. What do we mean by expediting drug development? Phase I Single Arm Phase II (expansion cohort) Randomized Phase II Phase III Necessary?
Clinical Trial Design to Expedite Drug Development Mary W. Redman, Ph.D. What do we mean by expediting drug development? Phase I Single Arm Phase II (expansion cohort) Randomized Phase II Phase III Necessary?
AbSeq on the BD Rhapsody system: Exploration of single-cell gene regulation by simultaneous digital mrna and protein quantification
 BD AbSeq on the BD Rhapsody system: Exploration of single-cell gene regulation by simultaneous digital mrna and protein quantification Overview of BD AbSeq antibody-oligonucleotide conjugates. High-throughput
BD AbSeq on the BD Rhapsody system: Exploration of single-cell gene regulation by simultaneous digital mrna and protein quantification Overview of BD AbSeq antibody-oligonucleotide conjugates. High-throughput
Fluzone High-Dose Vaccine and FIM12 Efficacy Trial Results
 Fluzone High-Dose Vaccine and FIM12 Efficacy Trial Results Corey A. Robertson, MD, MPH Director, Scientific and Medical Affairs, Sanofi Pasteur 1 Older Adults Suffer Disproportionately from Influenza-related
Fluzone High-Dose Vaccine and FIM12 Efficacy Trial Results Corey A. Robertson, MD, MPH Director, Scientific and Medical Affairs, Sanofi Pasteur 1 Older Adults Suffer Disproportionately from Influenza-related
The questions are changing in early phase cancer clinical trials: Opportunities for novel statistical designs
 The questions are changing in early phase cancer clinical trials: Opportunities for novel statistical designs Elizabeth Garrett-Mayer, PhD Associate Professor Director of Biostatistics, Hollings Cancer
The questions are changing in early phase cancer clinical trials: Opportunities for novel statistical designs Elizabeth Garrett-Mayer, PhD Associate Professor Director of Biostatistics, Hollings Cancer
Understanding Poor Vaccine Responses: Transcriptomics of Vaccine Failure
 Final Report - Summer Research Project 1 Introduction Understanding Poor Vaccine Responses: Transcriptomics of Vaccine Failure Katherine Miller, Year 3, Dalhousie University (Supervisor: Dr. Lisa Barrett,
Final Report - Summer Research Project 1 Introduction Understanding Poor Vaccine Responses: Transcriptomics of Vaccine Failure Katherine Miller, Year 3, Dalhousie University (Supervisor: Dr. Lisa Barrett,
SYSTEMS BIOLOGY APPROACHES TO IDENTIFY MECHANISMS OF IMMUNE MEDIATED PROTECTION TRANSLATING RESEARCH INTO HEALTH
 SYSTEMS BIOLOGY APPROACHES TO IDENTIFY MECHANISMS OF IMMUNE MEDIATED PROTECTION TRANSLATING RESEARCH INTO HEALTH Novel assays to decipher protective immune responses Decoding the immune response to infectious
SYSTEMS BIOLOGY APPROACHES TO IDENTIFY MECHANISMS OF IMMUNE MEDIATED PROTECTION TRANSLATING RESEARCH INTO HEALTH Novel assays to decipher protective immune responses Decoding the immune response to infectious
Clinical Study Report AI Final 28 Feb Volume: Page:
 Study Design, Continued Electrocardiogram (ECG) and vital sign assessments were done at select times during the study. Blood and urine samples for clinical laboratory evaluations were collected at specified
Study Design, Continued Electrocardiogram (ECG) and vital sign assessments were done at select times during the study. Blood and urine samples for clinical laboratory evaluations were collected at specified
Accelerating Innovation in Statistical Design
 Implementing a National Cancer Clinical Trials System for the 21 st Century, Workshop #2 Session #5: Accelerating Innovation Through Effective Partnerships Accelerating Innovation in Statistical Design
Implementing a National Cancer Clinical Trials System for the 21 st Century, Workshop #2 Session #5: Accelerating Innovation Through Effective Partnerships Accelerating Innovation in Statistical Design
PKPD modelling to optimize dose-escalation trials in Oncology
 PKPD modelling to optimize dose-escalation trials in Oncology Marina Savelieva Design of Experiments in Healthcare, Issac Newton Institute for Mathematical Sciences Aug 19th, 2011 Outline Motivation Phase
PKPD modelling to optimize dose-escalation trials in Oncology Marina Savelieva Design of Experiments in Healthcare, Issac Newton Institute for Mathematical Sciences Aug 19th, 2011 Outline Motivation Phase
Protocol Development: The Guiding Light of Any Clinical Study
 Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Bayesian Dose Escalation Study Design with Consideration of Late Onset Toxicity. Li Liu, Glen Laird, Lei Gao Biostatistics Sanofi
 Bayesian Dose Escalation Study Design with Consideration of Late Onset Toxicity Li Liu, Glen Laird, Lei Gao Biostatistics Sanofi 1 Outline Introduction Methods EWOC EWOC-PH Modifications to account for
Bayesian Dose Escalation Study Design with Consideration of Late Onset Toxicity Li Liu, Glen Laird, Lei Gao Biostatistics Sanofi 1 Outline Introduction Methods EWOC EWOC-PH Modifications to account for
DARPA-BAA Frequently Asked Questions
 DARPA-BAA-16-42 Frequently Asked Questions Last Updated: 7/19/2016 (Version 3) New Questions highlighted GENERAL INFORMATION Q: If my research is relevant in this field, but is not geared specifically
DARPA-BAA-16-42 Frequently Asked Questions Last Updated: 7/19/2016 (Version 3) New Questions highlighted GENERAL INFORMATION Q: If my research is relevant in this field, but is not geared specifically
Mapping the Antigenic and Genetic Evolution of Influenza Virus
 Mapping the Antigenic and Genetic Evolution of Influenza Virus Derek J. Smith, Alan S. Lapedes, Jan C. de Jong, Theo M. Bestebroer, Guus F. Rimmelzwaan, Albert D. M. E. Osterhaus, Ron A. M. Fouchier Science
Mapping the Antigenic and Genetic Evolution of Influenza Virus Derek J. Smith, Alan S. Lapedes, Jan C. de Jong, Theo M. Bestebroer, Guus F. Rimmelzwaan, Albert D. M. E. Osterhaus, Ron A. M. Fouchier Science
Lecture Outline Biost 517 Applied Biostatistics I
 Lecture Outline Biost 517 Applied Biostatistics I Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics University of Washington Lecture 2: Statistical Classification of Scientific Questions Types of
Lecture Outline Biost 517 Applied Biostatistics I Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics University of Washington Lecture 2: Statistical Classification of Scientific Questions Types of
Malik Sallam. Ola AL-juneidi. Ammar Ramadan. 0 P a g e
 1 Malik Sallam Ola AL-juneidi Ammar Ramadan 0 P a g e Today's lecture will be about viral upper respiratory tract infections. Those include: common cold, sinusitis, otitis, etc. Infections in the upper
1 Malik Sallam Ola AL-juneidi Ammar Ramadan 0 P a g e Today's lecture will be about viral upper respiratory tract infections. Those include: common cold, sinusitis, otitis, etc. Infections in the upper
Fundamental Clinical Trial Design
 Design, Monitoring, and Analysis of Clinical Trials Session 1 Overview and Introduction Overview Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics, University of Washington February 17-19, 2003
Design, Monitoring, and Analysis of Clinical Trials Session 1 Overview and Introduction Overview Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics, University of Washington February 17-19, 2003
Basket and Umbrella Trial Designs in Oncology
 Basket and Umbrella Trial Designs in Oncology Eric Polley Biomedical Statistics and Informatics Mayo Clinic Polley.Eric@mayo.edu Dose Selection for Cancer Treatment Drugs Stanford Medicine May 2017 1 /
Basket and Umbrella Trial Designs in Oncology Eric Polley Biomedical Statistics and Informatics Mayo Clinic Polley.Eric@mayo.edu Dose Selection for Cancer Treatment Drugs Stanford Medicine May 2017 1 /
pplementary Figur Supplementary Figure 1. a.
 pplementary Figur Supplementary Figure 1. a. Quantification by RT-qPCR of YFV-17D and YFV-17D pol- (+) RNA in the supernatant of cultured Huh7.5 cells following viral RNA electroporation of respective
pplementary Figur Supplementary Figure 1. a. Quantification by RT-qPCR of YFV-17D and YFV-17D pol- (+) RNA in the supernatant of cultured Huh7.5 cells following viral RNA electroporation of respective
Bayesian and Frequentist Approaches
 Bayesian and Frequentist Approaches G. Jogesh Babu Penn State University http://sites.stat.psu.edu/ babu http://astrostatistics.psu.edu All models are wrong But some are useful George E. P. Box (son-in-law
Bayesian and Frequentist Approaches G. Jogesh Babu Penn State University http://sites.stat.psu.edu/ babu http://astrostatistics.psu.edu All models are wrong But some are useful George E. P. Box (son-in-law
Bayesian Inference for Single-cell ClUstering and ImpuTing (BISCUIT) Elham Azizi
 Bayesian Inference for Single-cell ClUstering and ImpuTing (BISCUIT) Elham Azizi BioC 2017: Where Software and Biology Connect Profiling Tumor-Immune Ecosystem in Breast Cancer Immunotherapy treatments
Bayesian Inference for Single-cell ClUstering and ImpuTing (BISCUIT) Elham Azizi BioC 2017: Where Software and Biology Connect Profiling Tumor-Immune Ecosystem in Breast Cancer Immunotherapy treatments
Hierarchy of Statistical Goals
 Hierarchy of Statistical Goals Ideal goal of scientific study: Deterministic results Determine the exact value of a ment or population parameter Prediction: What will the value of a future observation
Hierarchy of Statistical Goals Ideal goal of scientific study: Deterministic results Determine the exact value of a ment or population parameter Prediction: What will the value of a future observation
Experimental Design for Immunologists
 Experimental Design for Immunologists Hulin Wu, Ph.D., Dean s Professor Department of Biostatistics & Computational Biology Co-Director: Center for Biodefense Immune Modeling School of Medicine and Dentistry
Experimental Design for Immunologists Hulin Wu, Ph.D., Dean s Professor Department of Biostatistics & Computational Biology Co-Director: Center for Biodefense Immune Modeling School of Medicine and Dentistry
Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era
 Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era Shivaani Kummar, MD, FACP Professor of Medicine (Oncology) Director, Phase I Clinical Research Program Co-Director, Translational
Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era Shivaani Kummar, MD, FACP Professor of Medicine (Oncology) Director, Phase I Clinical Research Program Co-Director, Translational
Discussion Meeting for MCP-Mod Qualification Opinion Request. Novartis 10 July 2013 EMA, London, UK
 Discussion Meeting for MCP-Mod Qualification Opinion Request Novartis 10 July 2013 EMA, London, UK Attendees Face to face: Dr. Frank Bretz Global Statistical Methodology Head, Novartis Dr. Björn Bornkamp
Discussion Meeting for MCP-Mod Qualification Opinion Request Novartis 10 July 2013 EMA, London, UK Attendees Face to face: Dr. Frank Bretz Global Statistical Methodology Head, Novartis Dr. Björn Bornkamp
BIOMARKER DRIVEN EARLY PHASE ONCOLOGY CLINICAL TRIALS; CHALLENGES IN THE REAL WORLD SID KATUGAMPOLA CENTRE FOR DRUG DEVELOPMENT CANCER RESEARCH UK
 BIOMARKER DRIVEN EARLY PHASE ONCOLOGY CLINICAL TRIALS; CHALLENGES IN THE REAL WORLD SID KATUGAMPOLA CENTRE FOR DRUG DEVELOPMENT CANCER RESEARCH UK Cancer; Some Sobering Thoughts Getting cancer is one of
BIOMARKER DRIVEN EARLY PHASE ONCOLOGY CLINICAL TRIALS; CHALLENGES IN THE REAL WORLD SID KATUGAMPOLA CENTRE FOR DRUG DEVELOPMENT CANCER RESEARCH UK Cancer; Some Sobering Thoughts Getting cancer is one of
Chapter 1. Introduction
 Chapter 1 Introduction 1.1 Motivation and Goals The increasing availability and decreasing cost of high-throughput (HT) technologies coupled with the availability of computational tools and data form a
Chapter 1 Introduction 1.1 Motivation and Goals The increasing availability and decreasing cost of high-throughput (HT) technologies coupled with the availability of computational tools and data form a
Risk-prediction modelling in cancer with multiple genomic data sets: a Bayesian variable selection approach
 Risk-prediction modelling in cancer with multiple genomic data sets: a Bayesian variable selection approach Manuela Zucknick Division of Biostatistics, German Cancer Research Center Biometry Workshop,
Risk-prediction modelling in cancer with multiple genomic data sets: a Bayesian variable selection approach Manuela Zucknick Division of Biostatistics, German Cancer Research Center Biometry Workshop,
Heterosubtypic immunity. Professor Ajit Lalvani FMedSci Chair of Infectious Diseases 14/07/2014
 Protective cellular immune correlates against pandemic influenza: implications for universal vaccines 2 nd WHO Meeting on development and clinical trials of broadly protective influenza vaccines 5th 7th
Protective cellular immune correlates against pandemic influenza: implications for universal vaccines 2 nd WHO Meeting on development and clinical trials of broadly protective influenza vaccines 5th 7th
Synopsis (C1034T02) CNTO 95 Module 5.3 Clinical Study Report C1034T02
 Module 5.3 Protocol: EudraCT No.: 2004-002130-18 Title of the study: A Phase 1/2, Multi-Center, Blinded, Randomized, Controlled Study of the Safety and Efficacy of the Human Monoclonal Antibody to Human
Module 5.3 Protocol: EudraCT No.: 2004-002130-18 Title of the study: A Phase 1/2, Multi-Center, Blinded, Randomized, Controlled Study of the Safety and Efficacy of the Human Monoclonal Antibody to Human
Information collected from influenza surveillance allows public health authorities to:
 OVERVIEW OF INFLUENZA SURVEILLANCE IN NEW JERSEY Influenza Surveillance Overview Surveillance for influenza requires monitoring for both influenza viruses and disease activity at the local, state, national,
OVERVIEW OF INFLUENZA SURVEILLANCE IN NEW JERSEY Influenza Surveillance Overview Surveillance for influenza requires monitoring for both influenza viruses and disease activity at the local, state, national,
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Statistics for Clinical Trials: Basics of Phase III Trial Design
 Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
Statistics for Clinical Trials: Basics of Phase III Trial Design Gary M. Clark, Ph.D. Vice President Biostatistics & Data Management Array BioPharma Inc. Boulder, Colorado USA NCIC Clinical Trials Group
Safety, Tolerability and Target Engagement Demonstrated in Phase 1 Study of LRRK2 Inhibitor DNL201 in Healthy Young and Elderly Adults
 Safety, Tolerability and Target Engagement Demonstrated in Phase 1 Study of LRRK2 Inhibitor DNL201 in Healthy Young and Elderly Adults MJFF Parkinson s Disease Therapeutics Conference October 25, 2018
Safety, Tolerability and Target Engagement Demonstrated in Phase 1 Study of LRRK2 Inhibitor DNL201 in Healthy Young and Elderly Adults MJFF Parkinson s Disease Therapeutics Conference October 25, 2018
Therapeutic Immunization with Autologous DC Pulsed with Autologous Inactivated HIV-1 Infected Apoptotic Cells
 Therapeutic Immunization with Autologous DC Pulsed with Autologous Inactivated HIV-1 Infected Apoptotic Cells Sharon A. Riddler, MD, MPH University of Pittsburgh May 2008 Slide 1 HIV and DC Vaccines During
Therapeutic Immunization with Autologous DC Pulsed with Autologous Inactivated HIV-1 Infected Apoptotic Cells Sharon A. Riddler, MD, MPH University of Pittsburgh May 2008 Slide 1 HIV and DC Vaccines During
Technology Overview. Summary
 Live Attenuated Influenza Vaccines with Altered NS1 Technology Overview Summary Transformative Technology: Live attenuated influenza vaccines (LAIVs) with precise, genetically stable truncations of the
Live Attenuated Influenza Vaccines with Altered NS1 Technology Overview Summary Transformative Technology: Live attenuated influenza vaccines (LAIVs) with precise, genetically stable truncations of the
Clinical Development Challenges: Trial Designs and Endpoints
 Clinical Development Challenges: Trial Designs and Endpoints Menno de Jong Department of Medical Microbiology Academic Medical Center, University of Amsterdam ISIRV - Options IX for the Control of Influenza
Clinical Development Challenges: Trial Designs and Endpoints Menno de Jong Department of Medical Microbiology Academic Medical Center, University of Amsterdam ISIRV - Options IX for the Control of Influenza
Supplementary Table 1: Summary of Reactogenicity Events by Group and Time of Onset in Entebbe
 Supplementary Table 1: Summary of Reactogenicity Events by Group and Time of Onset in Group A (n=39) B (n=11) A (n=39) B (n=11) Onset Post-Vaccination Event 3 Minutes 3-14 days Pain 5 (13%) 1 (1%) 6 (15%)
Supplementary Table 1: Summary of Reactogenicity Events by Group and Time of Onset in Group A (n=39) B (n=11) A (n=39) B (n=11) Onset Post-Vaccination Event 3 Minutes 3-14 days Pain 5 (13%) 1 (1%) 6 (15%)
ArQule Jefferies Global Healthcare Conference June 2015
 ArQule Jefferies Global Healthcare Conference June 2015 1 ArQule Corporate Update Safe Harbor This presentation and other statements by ArQule may contain forward-looking statements within the meaning
ArQule Jefferies Global Healthcare Conference June 2015 1 ArQule Corporate Update Safe Harbor This presentation and other statements by ArQule may contain forward-looking statements within the meaning
Combination dose finding studies in oncology: an industry perspective
 Combination dose finding studies in oncology: an industry perspective Jian Zhu, Ling Wang Symposium on Dose Selection for Cancer Treatment Drugs Stanford, May 12 th 2017 Outline Overview Practical considerations
Combination dose finding studies in oncology: an industry perspective Jian Zhu, Ling Wang Symposium on Dose Selection for Cancer Treatment Drugs Stanford, May 12 th 2017 Outline Overview Practical considerations
Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs
 Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs David Manner, Brenda Crowe and Linda Shurzinske BASS XVI November 9-13, 2009 Acknowledgements
Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs David Manner, Brenda Crowe and Linda Shurzinske BASS XVI November 9-13, 2009 Acknowledgements
Case Studies in Bayesian Augmented Control Design. Nathan Enas Ji Lin Eli Lilly and Company
 Case Studies in Bayesian Augmented Control Design Nathan Enas Ji Lin Eli Lilly and Company Outline Drivers for innovation in Phase II designs Case Study #1 Pancreatic cancer Study design Analysis Learning
Case Studies in Bayesian Augmented Control Design Nathan Enas Ji Lin Eli Lilly and Company Outline Drivers for innovation in Phase II designs Case Study #1 Pancreatic cancer Study design Analysis Learning
HAI and NAI as Correlates of Protection After Influenza Vaccination
 HAI and NAI as Correlates of Protection After Influenza Vaccination Arnold S. Monto Thomas Francis Jr. Professor University of Michigan School of Public Health Ann Arbor, Michigan Having Correlates is
HAI and NAI as Correlates of Protection After Influenza Vaccination Arnold S. Monto Thomas Francis Jr. Professor University of Michigan School of Public Health Ann Arbor, Michigan Having Correlates is
Developing Understanding of CMI. Dr Tom Wilkinson Associate Professor of Respiratory Medicine Faculty of Medicine University of Southampton UK
 Pre-existing influenza-specific CD4+ T cells correlate with homologous and heterotypic response and disease protection against influenza challenge in humans Do At Risk Groups Rely more on CMI to Viruses?
Pre-existing influenza-specific CD4+ T cells correlate with homologous and heterotypic response and disease protection against influenza challenge in humans Do At Risk Groups Rely more on CMI to Viruses?
Surveillance strategies for Foot and Mouth Disease to prove absence from disease and absence of viral circulation
 Surveillance strategies for Foot and Mouth Disease to prove absence from disease and absence of viral circulation Vincenzo Capora Asuncion, 25 June 20 Introduction In FMD control the need to prove absence
Surveillance strategies for Foot and Mouth Disease to prove absence from disease and absence of viral circulation Vincenzo Capora Asuncion, 25 June 20 Introduction In FMD control the need to prove absence
Experiences with Live Attenuated Avian Influenza Vaccine Trials in Thailand
 Experiences with Live Attenuated Avian Influenza Vaccine Trials in Thailand Punnee Pitisuttithum, MBBS,DTM&H,FRCPT Vaccine Trial Center,Faculty of Tropical Medicine, Mahidol University For MOPH,CDC,MU,Geneva
Experiences with Live Attenuated Avian Influenza Vaccine Trials in Thailand Punnee Pitisuttithum, MBBS,DTM&H,FRCPT Vaccine Trial Center,Faculty of Tropical Medicine, Mahidol University For MOPH,CDC,MU,Geneva
SUPPLEMENTARY APPENDIX
 SUPPLEMENTARY APPENDIX 1) Supplemental Figure 1. Histopathologic Characteristics of the Tumors in the Discovery Cohort 2) Supplemental Figure 2. Incorporation of Normal Epidermal Melanocytic Signature
SUPPLEMENTARY APPENDIX 1) Supplemental Figure 1. Histopathologic Characteristics of the Tumors in the Discovery Cohort 2) Supplemental Figure 2. Incorporation of Normal Epidermal Melanocytic Signature
Bayesian Prediction Tree Models
 Bayesian Prediction Tree Models Statistical Prediction Tree Modelling for Clinico-Genomics Clinical gene expression data - expression signatures, profiling Tree models for predictive sub-typing Combining
Bayesian Prediction Tree Models Statistical Prediction Tree Modelling for Clinico-Genomics Clinical gene expression data - expression signatures, profiling Tree models for predictive sub-typing Combining
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON THE DEVELOPMENT OF LIVE ATTENUATED INFLUENZA VACCINES
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 20 February 2003 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON THE
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 20 February 2003 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON THE
Lecture Outline Biost 517 Applied Biostatistics I. Statistical Goals of Studies Role of Statistical Inference
 Lecture Outline Biost 517 Applied Biostatistics I Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics University of Washington Statistical Inference Role of Statistical Inference Hierarchy of Experimental
Lecture Outline Biost 517 Applied Biostatistics I Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics University of Washington Statistical Inference Role of Statistical Inference Hierarchy of Experimental
Basket Trials: Features, Examples, and Challenges
 : Features, s, and Challenges Lindsay A. Renfro, Ph.D. Associate Professor of Research Division of Biostatistics University of Southern California ASA Biopharm / Regulatory / Industry Statistics Workshop
: Features, s, and Challenges Lindsay A. Renfro, Ph.D. Associate Professor of Research Division of Biostatistics University of Southern California ASA Biopharm / Regulatory / Industry Statistics Workshop
Min Levine, Ph. D. Influenza Division US Centers for Disease Control and Prevention. June 18, 2015 NIBSC
 Workshop on Immunoassay Standardization for Universal Flu Vaccines Min Levine, Ph. D. Influenza Division US Centers for Disease Control and Prevention June 18, 2015 NIBSC 1 Multiple Immune Mechanisms Contribute
Workshop on Immunoassay Standardization for Universal Flu Vaccines Min Levine, Ph. D. Influenza Division US Centers for Disease Control and Prevention June 18, 2015 NIBSC 1 Multiple Immune Mechanisms Contribute
Chapter 24 The Immune System
 Chapter 24 The Immune System The Immune System Layered defense system The skin and chemical barriers The innate and adaptive immune systems Immunity The body s ability to recognize and destroy specific
Chapter 24 The Immune System The Immune System Layered defense system The skin and chemical barriers The innate and adaptive immune systems Immunity The body s ability to recognize and destroy specific
Induction of Innate Immune Responses in HVTN 071: a Trial using the MRKAd5 gag/pol/nef Vaccine from the Step Study
 Induction of Innate Immune Responses in HVTN 71: a Trial using the MRKAd5 gag/pol/nef Vaccine from the Step Study Erica Andersen-Nissen Vaccine and Infectious Disease Institute Fred Hutchinson Cancer Research
Induction of Innate Immune Responses in HVTN 71: a Trial using the MRKAd5 gag/pol/nef Vaccine from the Step Study Erica Andersen-Nissen Vaccine and Infectious Disease Institute Fred Hutchinson Cancer Research
MERS H7N9. coronavirus. influenza virus. MERS: Since September 2012: 160 cases, including 68 deaths (42%)
 MERS coronavirus H7N9 influenza virus Marc Van Ranst, KU Leuven MERS: Since September 2012: 160 cases, including 68 deaths (42%) MERS coronavirus patient, 83 years, Saudi-Arabia H7N9: Since February 2013:
MERS coronavirus H7N9 influenza virus Marc Van Ranst, KU Leuven MERS: Since September 2012: 160 cases, including 68 deaths (42%) MERS coronavirus patient, 83 years, Saudi-Arabia H7N9: Since February 2013:
Design for Targeted Therapies: Statistical Considerations
 Design for Targeted Therapies: Statistical Considerations J. Jack Lee, Ph.D. Department of Biostatistics University of Texas M. D. Anderson Cancer Center Outline Premise General Review of Statistical Designs
Design for Targeted Therapies: Statistical Considerations J. Jack Lee, Ph.D. Department of Biostatistics University of Texas M. D. Anderson Cancer Center Outline Premise General Review of Statistical Designs
General Overview of Immunology. Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center
 General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
REGULATORY REQUIREMENTS FOR VIRAL-CHALLENGE STUDIES: INFLUENZA CASE STUDY
 LIFE SCIENCE I TECHNICAL BULLETIN JUNE 2014 REGULATORY REQUIREMENTS FOR VIRAL-CHALLENGE STUDIES: INFLUENZA CASE STUDY AUTHOR:BRUNO SPEDER, HEAD CLINICAL REGULATORY AFFAIRS & DOCUMENT MANAGEMENT - CLINICAL
LIFE SCIENCE I TECHNICAL BULLETIN JUNE 2014 REGULATORY REQUIREMENTS FOR VIRAL-CHALLENGE STUDIES: INFLUENZA CASE STUDY AUTHOR:BRUNO SPEDER, HEAD CLINICAL REGULATORY AFFAIRS & DOCUMENT MANAGEMENT - CLINICAL
PATH Influenza Vaccine Projects
 PATH Influenza Vaccine Projects Overview John W. Boslego, MD John Boslego Director, Vaccine Development Global Program March 25 th, 2014 Influenza Vaccine Project (IVP) at PATH IVP Goal: Advance the development
PATH Influenza Vaccine Projects Overview John W. Boslego, MD John Boslego Director, Vaccine Development Global Program March 25 th, 2014 Influenza Vaccine Project (IVP) at PATH IVP Goal: Advance the development
Predictive Assays in Radiation Therapy
 Outline Predictive Assays in Radiation Therapy Radiation Biology Introduction Early predictive assays Recent trends in predictive assays Examples for specific tumors Summary Lecture 4-23-2014 Introduction
Outline Predictive Assays in Radiation Therapy Radiation Biology Introduction Early predictive assays Recent trends in predictive assays Examples for specific tumors Summary Lecture 4-23-2014 Introduction
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
Lecture Outline. Biost 590: Statistical Consulting. Stages of Scientific Studies. Scientific Method
 Biost 590: Statistical Consulting Statistical Classification of Scientific Studies; Approach to Consulting Lecture Outline Statistical Classification of Scientific Studies Statistical Tasks Approach to
Biost 590: Statistical Consulting Statistical Classification of Scientific Studies; Approach to Consulting Lecture Outline Statistical Classification of Scientific Studies Statistical Tasks Approach to
Janet E. Dancey NCIC CTG NEW INVESTIGATOR CLINICAL TRIALS COURSE. August 9-12, 2011 Donald Gordon Centre, Queen s University, Kingston, Ontario
 Janet E. Dancey NCIC CTG NEW INVESTIGATOR CLINICAL TRIALS COURSE August 9-12, 2011 Donald Gordon Centre, Queen s University, Kingston, Ontario Session: Correlative Studies in Phase III Trials Title: Design
Janet E. Dancey NCIC CTG NEW INVESTIGATOR CLINICAL TRIALS COURSE August 9-12, 2011 Donald Gordon Centre, Queen s University, Kingston, Ontario Session: Correlative Studies in Phase III Trials Title: Design
Studying Repeated Immunization in an Animal Model. Kanta Subbarao Laboratory of Infectious Diseases, NIAID
 Studying Repeated Immunization in an Animal Model Kanta Subbarao Laboratory of Infectious Diseases, NIAID Animal models in Influenza Research Commonly used Mice Ferrets Guinea pigs Non human primates Less
Studying Repeated Immunization in an Animal Model Kanta Subbarao Laboratory of Infectious Diseases, NIAID Animal models in Influenza Research Commonly used Mice Ferrets Guinea pigs Non human primates Less
Chapter 14 Part One Biotechnology and Industry: Microbes at Work
 Chapter 14 Part One Biotechnology and Industry: Microbes at Work Objectives: After reading Chapter 14, you should understand How biotechnology has resulted in numerous pharmaceutical products to help lessen
Chapter 14 Part One Biotechnology and Industry: Microbes at Work Objectives: After reading Chapter 14, you should understand How biotechnology has resulted in numerous pharmaceutical products to help lessen
The Innate Immune Response
 The Innate Immune Response FUNCTIONS OF THE IMMUNE SYSTEM: Recognize, destroy and clear a diversity of pathogens. Initiate tissue and wound healing processes. Recognize and clear damaged self components.
The Innate Immune Response FUNCTIONS OF THE IMMUNE SYSTEM: Recognize, destroy and clear a diversity of pathogens. Initiate tissue and wound healing processes. Recognize and clear damaged self components.
New Challenges: Model-based Dose Finding in the Era of Targeted Agents
 New Challenges: Model-based Dose Finding in the Era of Targeted Agents Elizabeth Garrett-Mayer, PhD 1 Cody Chiuzan, PhD 2 1 Hollings Cancer Center Department of Public Health Sciences Medical University
New Challenges: Model-based Dose Finding in the Era of Targeted Agents Elizabeth Garrett-Mayer, PhD 1 Cody Chiuzan, PhD 2 1 Hollings Cancer Center Department of Public Health Sciences Medical University
2.0 Synopsis. ABT-333 M Clinical Study Report R&D/09/956
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: ABT-333 Volume: Name of Active Ingredient: Page: Sodium N-{6-[3-tert-butyl-5-(2,4-
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: ABT-333 Volume: Name of Active Ingredient: Page: Sodium N-{6-[3-tert-butyl-5-(2,4-
Mass Cytometry Applications for Immunology Research. Pub Note PN 13 01_150505
 Mass Cytometry Applications for Immunology Research Pub Note PN 13 01_150505 Contents Continuum of CD8 + T cell phenotypes revealed by deep profiling with mass cytometry 3 The transcriptional landscape
Mass Cytometry Applications for Immunology Research Pub Note PN 13 01_150505 Contents Continuum of CD8 + T cell phenotypes revealed by deep profiling with mass cytometry 3 The transcriptional landscape
Therapeutic Efficacy of a TLR7 Agonist for HBV Chronic Infection in Chimpanzees
 Therapeutic Efficacy of a TLR7 Agonist for HBV Chronic Infection in Chimpanzees Robert Lanford 1, Bernadette Guerra 1, Deborah Chavez 1, Vida L. Hodara 1, Xubin Zheng 2, Grushenka Wolfgang 3, Daniel B.
Therapeutic Efficacy of a TLR7 Agonist for HBV Chronic Infection in Chimpanzees Robert Lanford 1, Bernadette Guerra 1, Deborah Chavez 1, Vida L. Hodara 1, Xubin Zheng 2, Grushenka Wolfgang 3, Daniel B.
Third line of Defense
 Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Randomized Clinical Trials
 Randomized Clinical Trials p. 1/42 Randomized Clinical Trials Hematology/Oncology Lecture Series Elizabeth G. Hill, PhD Associate Professor of Biostatistics 17 November 2011 Randomized Clinical Trials
Randomized Clinical Trials p. 1/42 Randomized Clinical Trials Hematology/Oncology Lecture Series Elizabeth G. Hill, PhD Associate Professor of Biostatistics 17 November 2011 Randomized Clinical Trials
Update of WHO biosafety risk assessment and guidelines for the production and quality control of human influenza pandemic vaccines
 Update of WHO biosafety risk assessment and guidelines for the production and quality control of human influenza pandemic vaccines 28 May 2009 Introduction This document updates WHO guidance 1 to national
Update of WHO biosafety risk assessment and guidelines for the production and quality control of human influenza pandemic vaccines 28 May 2009 Introduction This document updates WHO guidance 1 to national
National Surgical Adjuvant Breast and Bowel Project (NSABP) Foundation Annual Progress Report: 2009 Formula Grant
 National Surgical Adjuvant Breast and Bowel Project (NSABP) Foundation Annual Progress Report: 2009 Formula Grant Reporting Period July 1, 2011 June 30, 2012 Formula Grant Overview The National Surgical
National Surgical Adjuvant Breast and Bowel Project (NSABP) Foundation Annual Progress Report: 2009 Formula Grant Reporting Period July 1, 2011 June 30, 2012 Formula Grant Overview The National Surgical
GSK s Adjuvanted Influenza Vaccines The Taming of the Flu
 GSK s Adjuvanted Influenza Vaccines The Taming of the Flu JITMM, Bangkok, October 2008 Bruce L. Innis, MD Global Clinical Research and Development GlaxoSmithKline Biologicals 1 Annual Burden of Influenza
GSK s Adjuvanted Influenza Vaccines The Taming of the Flu JITMM, Bangkok, October 2008 Bruce L. Innis, MD Global Clinical Research and Development GlaxoSmithKline Biologicals 1 Annual Burden of Influenza
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS. Choompone Sakonwasun, MD (Hons), FRCPT
 ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
Age-Related Changes in Immune System Function. Professor Justin Hall October 12, 2010
 Age-Related Changes in Immune System Function Professor Justin Hall October 12, 2010 Immune Function: Effect of Aging and Exercise Why study this? INCREASED RISK for infectious diseases, tumorigenesis,
Age-Related Changes in Immune System Function Professor Justin Hall October 12, 2010 Immune Function: Effect of Aging and Exercise Why study this? INCREASED RISK for infectious diseases, tumorigenesis,
Week 11: March 11 to March 17, 2018
 2017/18 Season Week in Review: Week 11: March 11 to March 17, 2018 Influenza Activity Indicator Assessment of Indicator* Interpretation Reported cases Percent Positive of Flu Specimens Local Institutional
2017/18 Season Week in Review: Week 11: March 11 to March 17, 2018 Influenza Activity Indicator Assessment of Indicator* Interpretation Reported cases Percent Positive of Flu Specimens Local Institutional
Week 15: April 8 to April 14, 2018
 2017/18 Season Week in Review: Week 15: April 8 to April 14, 2018 Influenza Activity Indicator Assessment of Indicator* Interpretation Reported cases Percent Positive of Flu Specimens Local Institutional
2017/18 Season Week in Review: Week 15: April 8 to April 14, 2018 Influenza Activity Indicator Assessment of Indicator* Interpretation Reported cases Percent Positive of Flu Specimens Local Institutional
The 2010 Gastrointestinal Cancers Symposium Oral Abstract Session: Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract
 The 2010 Gastrointestinal Cancers Symposium : Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract Abstract #131: Phase I study of MK 0646 (dalotuzumab), a humanized monoclonal antibody against
The 2010 Gastrointestinal Cancers Symposium : Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract Abstract #131: Phase I study of MK 0646 (dalotuzumab), a humanized monoclonal antibody against
Highlights from AACR 2015: The Emerging Potential of Immunotherapeutic Approaches in Non-Small Cell Lung Cancer
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
US Regulatory Considerations for Therapeutic Cancer Vaccines
 US Regulatory Considerations for Therapeutic Cancer Vaccines Peter Bross, M.D., Team Leader, Clinical Oncology, FDA Center for Biologics Evaluation and Research 1 Immune Therapies: The Future Is Now Tuesday,
US Regulatory Considerations for Therapeutic Cancer Vaccines Peter Bross, M.D., Team Leader, Clinical Oncology, FDA Center for Biologics Evaluation and Research 1 Immune Therapies: The Future Is Now Tuesday,
24 26 January 2013, Hong Kong SAR, CHINA. TITLE from VIEW and SLIDE MASTER February 27, 2013
 The first WHO integrated meeting on development and clinical trials of influenza vaccines that induce broadly protective and long-lasting immune responses 24 26 January 2013, Hong Kong SAR, CHINA 1 TITLE
The first WHO integrated meeting on development and clinical trials of influenza vaccines that induce broadly protective and long-lasting immune responses 24 26 January 2013, Hong Kong SAR, CHINA 1 TITLE
Mon, Wed, Fri 11:00 AM-12:00 PM. Owen, Judy, Jenni Punt, and Sharon Stranford Kuby-Immunology, 7th. Edition. W.H. Freeman and Co., New York.
 Course Title: Course Number: Immunology Biol-341/541 Semester: Fall 2013 Location: HS 268 Time: Instructor: 8:00-9:30 AM Tue/Thur Dr. Colleen M. McDermott Office: Nursing Ed 101 (424-1217) E-mail*: mcdermot@uwosh.edu
Course Title: Course Number: Immunology Biol-341/541 Semester: Fall 2013 Location: HS 268 Time: Instructor: 8:00-9:30 AM Tue/Thur Dr. Colleen M. McDermott Office: Nursing Ed 101 (424-1217) E-mail*: mcdermot@uwosh.edu
Development of a Novel Recombinant Influenza Vaccine in Insect Cells
 Development of a Novel Recombinant Influenza Vaccine in Insect Cells Clifton McPherson New Cells for New Vaccines II September 18, 2007 cmcpherson@proteinsciences.com New Cell for New Vaccines II Topics:
Development of a Novel Recombinant Influenza Vaccine in Insect Cells Clifton McPherson New Cells for New Vaccines II September 18, 2007 cmcpherson@proteinsciences.com New Cell for New Vaccines II Topics:
Basis and Clinical Applications of Interferon
 Interferon Therapy Basis and Clinical Applications of Interferon JMAJ 47(1): 7 12, 2004 Jiro IMANISHI Professor, Kyoto Prefectural University of Medicine Abstract: Interferon (IFN) is an antiviral substance
Interferon Therapy Basis and Clinical Applications of Interferon JMAJ 47(1): 7 12, 2004 Jiro IMANISHI Professor, Kyoto Prefectural University of Medicine Abstract: Interferon (IFN) is an antiviral substance
Autoimmune Diseases. Betsy Kirchner CNP The Cleveland Clinic
 Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
NASDAQ:NVAX Novavax, Inc. All rights reserved.
 Novavax vaccine induced improved immune responses against homologous and drifted A(H3N2) viruses in older adults compared to egg-based, high-dose, influenza vaccine World Vaccine Congress April 4, 2018
Novavax vaccine induced improved immune responses against homologous and drifted A(H3N2) viruses in older adults compared to egg-based, high-dose, influenza vaccine World Vaccine Congress April 4, 2018
A Just in Time Primer on H1N1 Influenza A and Pandemic Influenza developed by the National Association of State EMS Officials and Revised by the
 A Just in Time Primer on H1N1 Influenza A and Pandemic Influenza developed by the National Association of State EMS Officials and Revised by the Michigan Department of Community Health EMS and Trauma Systems
A Just in Time Primer on H1N1 Influenza A and Pandemic Influenza developed by the National Association of State EMS Officials and Revised by the Michigan Department of Community Health EMS and Trauma Systems
Using Biomarkers to Increase the Efficiency of Early Stage Drug Development
 Using Biomarkers to Increase the Efficiency of Early Stage Drug Development Personal consulting compensation received from Cytokinetics, Biogen, MT Pharma, Lilly, Karyopharm, Denali Goals of Talk Describe
Using Biomarkers to Increase the Efficiency of Early Stage Drug Development Personal consulting compensation received from Cytokinetics, Biogen, MT Pharma, Lilly, Karyopharm, Denali Goals of Talk Describe
Full title: A likelihood-based approach to early stopping in single arm phase II cancer clinical trials
 Full title: A likelihood-based approach to early stopping in single arm phase II cancer clinical trials Short title: Likelihood-based early stopping design in single arm phase II studies Elizabeth Garrett-Mayer,
Full title: A likelihood-based approach to early stopping in single arm phase II cancer clinical trials Short title: Likelihood-based early stopping design in single arm phase II studies Elizabeth Garrett-Mayer,
Discrimination and Reclassification in Statistics and Study Design AACC/ASN 30 th Beckman Conference
 Discrimination and Reclassification in Statistics and Study Design AACC/ASN 30 th Beckman Conference Michael J. Pencina, PhD Duke Clinical Research Institute Duke University Department of Biostatistics
Discrimination and Reclassification in Statistics and Study Design AACC/ASN 30 th Beckman Conference Michael J. Pencina, PhD Duke Clinical Research Institute Duke University Department of Biostatistics
Protocol Synopsis. Administrative information
 Protocol Synopsis Item (SPIRIT item no.) Administrative information Title (1) Introduction Description of research question (6a) Description An optimal schedule for the post-polio eradication era: multicentre
Protocol Synopsis Item (SPIRIT item no.) Administrative information Title (1) Introduction Description of research question (6a) Description An optimal schedule for the post-polio eradication era: multicentre
RESEARCH WITH HIGHLY PATHOGENIC AVIAN INFLUENZA H5N1
 Page 1 of 8 RESEARCH WITH HIGHLY PATHOGENIC AVIAN INFLUENZA H5N1 The University of Pittsburgh has developed guidelines to establish a system of education and safeguards to ensure compliance with both the
Page 1 of 8 RESEARCH WITH HIGHLY PATHOGENIC AVIAN INFLUENZA H5N1 The University of Pittsburgh has developed guidelines to establish a system of education and safeguards to ensure compliance with both the
Feline Panleukopenia Vaccine
 Feline Panleukopenia Vaccine Modified Live Virus For use in cats only P FPO 2D PRODUCT DESCRIPTION: Felocell P is for vaccination of healthy cats as an aid in preventing feline panleukopenia caused by
Feline Panleukopenia Vaccine Modified Live Virus For use in cats only P FPO 2D PRODUCT DESCRIPTION: Felocell P is for vaccination of healthy cats as an aid in preventing feline panleukopenia caused by
Patterns of hemagglutinin evolution and the epidemiology of influenza
 2 8 US Annual Mortality Rate All causes Infectious Disease Patterns of hemagglutinin evolution and the epidemiology of influenza DIMACS Working Group on Genetics and Evolution of Pathogens, 25 Nov 3 Deaths
2 8 US Annual Mortality Rate All causes Infectious Disease Patterns of hemagglutinin evolution and the epidemiology of influenza DIMACS Working Group on Genetics and Evolution of Pathogens, 25 Nov 3 Deaths
14/07/2014. Disclosures and acknowledgements. Study Design (NCT ) Rationale for a QIV efficacy study in children
 /7/ Disclosures and acknowledgements I am employed by the GlaxoSmithKline group of companies and I own stocks/options of the GlaxoSmithKline group of companies; my travel to this meeting was funded by
/7/ Disclosures and acknowledgements I am employed by the GlaxoSmithKline group of companies and I own stocks/options of the GlaxoSmithKline group of companies; my travel to this meeting was funded by
BIOSTATISTICAL METHODS
 BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Designs on Micro Scale: DESIGNING CLINICAL RESEARCH THE ANATOMY & PHYSIOLOGY OF CLINICAL RESEARCH We form or evaluate a research or research
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Designs on Micro Scale: DESIGNING CLINICAL RESEARCH THE ANATOMY & PHYSIOLOGY OF CLINICAL RESEARCH We form or evaluate a research or research
Durable Response Rate in High Grade Glioma: an Emerging Endpoint for Immunotherapeutics. Timothy Cloughesy, MD University of California, Los Angeles
 Durable Response Rate in High Grade Glioma: an Emerging Endpoint for Immunotherapeutics Timothy Cloughesy, MD University of California, Los Angeles Disclosure 2 FDA Endpoints for the Approval of Cancer
Durable Response Rate in High Grade Glioma: an Emerging Endpoint for Immunotherapeutics Timothy Cloughesy, MD University of California, Los Angeles Disclosure 2 FDA Endpoints for the Approval of Cancer
Cross-Reactivity to Field Isolates of Canine Influenza Virus by a Killed Canine Influenza Virus (H3N8, Iowa05) Vaccine
 Cross-Reactivity to Field Isolates of Canine Influenza Virus by a Killed Canine Influenza Virus (H3N8, Iowa05) Vaccine Nancee Oien, B.S., M.S. a Sally Mattern, B.S a Jaime Brozowski, B.S., M.S. b Janet
Cross-Reactivity to Field Isolates of Canine Influenza Virus by a Killed Canine Influenza Virus (H3N8, Iowa05) Vaccine Nancee Oien, B.S., M.S. a Sally Mattern, B.S a Jaime Brozowski, B.S., M.S. b Janet
