The Role of Plasmacytoid Dendritic Cells in Psoriasis
|
|
|
- Christina Cannon
- 6 years ago
- Views:
Transcription
1 6 ème Journée du Groupe de Recherche sur le Psoriasis Paris, 9 novembre 2012 The Role of Plasmacytoid Dendritic Cells in Psoriasis Dr Curdin Conrad, PD & MER Department of Dermatology
2 Psoriasis: Clinical Spectrum
3 Histology Keratinocyte or Immune Defect? Angiogenesis Epidermal Hyperproliferation Inflammation
4 Psoriasis Uniquely human disease
5 AGR Mouse Model New mouse model with spontaneous development of psoriasis Boyman, Hefti, Conrad, Nickoloff, Suter, Nestle. J Exp Med. 2004
6 AGR Mouse Model Uninvolved skin from a psoriasis patienten A = Type I IFN receptor deficiency G = Type II IFN receptor deficiency R = Rag -/-
7 Day 56 Day 0 Spontaneous Conversion Uninvolved skin on AGR a Macroscopic b CD3 c d Boyman et al. J Exp Med. 2004
8 AGR Mouse Model - Pathogenesis PN day 0 Conrad et al. Nat Med 2007
9 AGR Mouse Model - Pathogenesis PN day 7 Conrad et al. Nat Med 2007
10 AGR Mouse Model - Pathogenesis PN day 21 Conrad et al. Nat Med 2007
11 AGR Mouse Model - Pathogenesis PN day 35 Conrad et al. Nat Med 2007
12 AGR Mouse Model - Pathogenesis Conrad et al. Nat Med 2007
13 AGR Mouse Model - Pathogenesis Conrad et al. Nat Med 2007
14 AGR Mouse Model - Pathogenesis Conrad et al. Nat Med 2007
15 Chronic Plaque Psoriasis psoriasis IFN- IL-17 IL-22 Th1/Tc1 cells Th17/Tc17 cells IL-23 TNF Chronic phase
16 Immunopathogenesis Koebner Phenomenon (skin wounding) psoriasis?? Th1/Tc1 cells IFN- IL-17 IL-22 Th17/Tc17 cells IL-23 Triggers of acute phase? TNF Chronic phase
17 Aldara (Imiquimod) Induces Psoriasis M. Bowen? Psoriasis! Week 0 Week 6 Week 10
18 pdcs in Antiviral Immunity DNA and ssrna Viruses Cellular resistance to viral infection TLR7 TLR9 IFN-α/β Immature cdc Activated cdc plasmaytoid DC (pdc) B NK Antibodies Cytotoxicity
19 pdcs in Psoriasis BDCA-2 Psoriasis Skin Lesion pre-psoriatic skin Nestle et al. J Exp Med. 2005
20 IFNα Induces Psoriasis Nestle et al. J Exp Med. 2005
21 pdc-derived IFNα Induces Psoriasis Nestle et al. J Exp Med. 2005
22 Pathophysiology of Psoriasis Koebner phenomenon (wounding, mechanical stress) psoriasis? IFN- IL-17 IL-22 plasmacytoid DC Acute phase IFN- Th1/Tc1 cells TNF Th17/Tc17 cells IL-23 Chronic phase
23 AMP LL37 - Key Trigger of pdc Activation HPLC of psoriatic skin 2 LL Elution time (min) Lande et al. Nature. 2007
24 Cationic Antimicrobial Peptides (AMP) - evolutionary ancient antimicrobial defense mechanism - highly cationic, amphipathic peptides - 2 main classes in humans: DEFENSINS (beta-sheet peptides) -defensins -defensins CATHELICIDINS (alpha-helical peptides) LL37 is the only human member
25 Discrimination between viral and self-dna VIRAL INFECTION HOST CELL DEATH DNA virus human DNA TLR9 plasmacytoid DC IFN-α/β
26 LL37 Converts Otherwise Inert self-dna into a Trigger of IFNα Lande et al. Nature. 2007
27 LL37 controls ability of pdcs to sense self-nucleic acids HOST CELL DEATH LL37 Self-DNA/RNA Lande et al. Nature Ganguly et al. J Exp Med Self-DNA/RNA-LL37complexes 1- PROTECTION 2- INTERNALIZATION 3- ENDOSOMAL RETENTION TLR9/7 pdc IFN-α/β
28 Psoriasis: Pathogenesis AMP gene copy number polymorphism HLA-C ERAP1/ZAP70 Tyk2 IL-23 TNF TNFAIP IL23R IL23A IFIH1 Conrad et al. Semin Immunol. 2009
29 Psoriasis: Pathogenesis psoriasis IFN- IL-17 IL-22 Th1/Tc1 cells Th17/Tc17 cells IL-23 TNF
30 Biologics for Psoriasis Therapy RP = Rheumatoid polyarthritis Pso = Psoriasis PsoA = Psoriatic arthritis SA = Ankylosing spondylarthritis CD = Crohn s disease UC = Ulcerative colitis * For patients >100 kg
31 Case Report Patient S.C., Crohn s disease, first diagnosed 2007 steroid-refractory started with azathioprine (Imurek ) reactivation of Crohn s disease Adalimumab (Humira )
32 Cutaneous Eruptions Under anti-tnf Therapy Crohn s disease, under adalimumab
33 Cutaneous Eruptions Under anti-tnf Therapy Crohn s disease, under adalimumab
34 Case Report Patient C.M., 1953 Spondylarthritis 2 months after starting anti-tnf therapy (infliximab, Remicade )
35 Cutaneous Eruptions Under anti-tnf Therapy Spondylarthritis, under infiliximab
36 Cutaneous Eruptions Under anti-tnf Therapy Spondylarthritis, under infiliximab
37 Cutaneous Side Effects of anti-tnf Therapy Immediate adverse events Acute infusion reaction (frequent) Immune-mediated hypersensitivity reaction (rare) Delayed cutaneous eruptions Psoriasis Worsening of pre-existing psoriasis Onset of a psoriasiform eruption Pustular psoriasis Lupus erythematosus Discoid lupus erythematosus Systemic-lupus-erythematosus-like syndromes Interface dermatitis Erythema multiforme Lichenoid eruption Bullous eruption Vascular lesions a Leucocytoclastic vasculitis b Serum sickness c Eczematid-like purpura d Perniosis Eczematous eruption: atopic-dermatitis-like eruption Granuloma annulare Infections a Acute bacterial folliculitis b Dermatophytosis c Molluscum contagiosum d Herpes Cutaneous lymphomas: CD30 T-cell lymphoma Non-melanoma skin cancer a Kerato-acanthomas b Squamous cell carcinoma
38 Cutaneous Side Effects of anti-tnf Therapy Psoriatic skin lesions induced by anti-tnf therapy «paradoxic psoriasis» - Histologically confirmed
39 Paradoxic Psoriasis Induced by TNF-Inhibitors Well know side effect of anti-tnf therapy Numerous cases reported in the literature 2% of patients receiving anti-tnf treatment
40 Paradoxic Psoriasis Induced by TNF-Inhibitors Indication for anti-tnf Type of anti-tnf PA psoriasis FA psoriasis Delay after therapy start 1. C.M SA Infliximab months 2. C.I Crohn Infliximab months 3. M.V Crohn Infliximab months 4. R.G SA Adalimumab Etanercept months 2 months 5. C.C.1952 SAPHO/PsoA Adalimumab weeks 6. J.R RP Adalimumab Etanercept months immediatly 7. R.C Crohn Infliximab months 8. R.Y PsoA Adalimumab months
41 TNF-Inhibitors for Chronic Psoriasis week 0 week 2 week 10 Remicade (infliximab 5mg/kg)
42 Paradoxic Psoriasis Induced by TNF-Inhibitors Pathogenesis unknown
43 Yin-yang of TNF and IFN-α Immunity as a dynamic system driven by sets of opposite vectors (TNF/IFN-α) adapted from: Banchereau et al. Immunity 2004
44 Dysregulation by TNF-Inhibitors Anti-TNF- Acute/paradoxic psoriasis
45 Clinical Relevance Class effect of TNF-inhibitors no switch to another TNF-inhibitor - topical steroids - MTX, cyclosporine, short-term systemic steroids, retinoids - switch to another class: anti-il12/23, anti-il17 (psoriasis) anti-ctla4, anti-cd20, anti-il6 (arthritis) - anti-ifn-?
46 Clinical Relevance Arthritis Immunity as a dynamic system driven by sets of opposite vectors (TNF/IFN-α) TNF prevailing: TNF-mediated autoimmunity (arthritis) Lupus IFN-α: IFN autoimmunity (SLE, thyroiditis, diabetes) adapted from: Banchereau et al. Immunity 2004
47 Dysregulation by TNF-Inhibitors Anti-TNF- Lupus erythematosus Acute/paradoxic psoriasis
48 Acknowledgements Cyrine Belkhouja Dr Olivier Demaria Dr Maxime Vernez Anne-Karine Lapointe Prof. Dr Michel Gilliet
IMO-8400, a novel TLR7, TLR8 and TLR9 antagonist, psoriasis
 IMO-8400, a novel TLR7, TLR8 and TLR9 antagonist, inhibits disease development in mouse models of psoriasis Weiwen e Ja Jiang, Fu-Gang Zhu, Dong Yu, Ekambar R. Kandimalla, a a, Nicola La Monica, and Sudhir
IMO-8400, a novel TLR7, TLR8 and TLR9 antagonist, inhibits disease development in mouse models of psoriasis Weiwen e Ja Jiang, Fu-Gang Zhu, Dong Yu, Ekambar R. Kandimalla, a a, Nicola La Monica, and Sudhir
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients
 IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
Rashes Not To Be Missed In Children
 May 2016 Rashes Not To Be Missed In Children Dr Chan Yuin Chew Dermatologist Dermatology Associates Gleneagles Medical Centre Scope of presentation Focus on rashes May lead to significant morbidity if
May 2016 Rashes Not To Be Missed In Children Dr Chan Yuin Chew Dermatologist Dermatology Associates Gleneagles Medical Centre Scope of presentation Focus on rashes May lead to significant morbidity if
Antimicrobial Peptides in Defense and Inflammatory Skin Diseases
 Antimicrobial Peptides in Defense and Inflammatory Skin Diseases Richard L. Gallo, M.D.,Ph.D. Professor of Medicine and Pediatrics, Chief, Division of Dermatology!The Problem Mycobacterium TB Pseudomonas
Antimicrobial Peptides in Defense and Inflammatory Skin Diseases Richard L. Gallo, M.D.,Ph.D. Professor of Medicine and Pediatrics, Chief, Division of Dermatology!The Problem Mycobacterium TB Pseudomonas
Drugs and Applicable Coding: J-code: Enbrel-J1438; Humira-J0135; Remicade-J1745; Inflectra-Q5102; Cimzia-J0718; Simponi-J1602 Renflexis - pending
 Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Index. derm.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Abatacept for DLE, 493 for SLE, 497 Ablative therapies, localized, for cutaneous T-cell lymphoma, 502 506. See also Cutaneous T-cell lymphoma,
Note: Page numbers of article titles are in boldface type. A Abatacept for DLE, 493 for SLE, 497 Ablative therapies, localized, for cutaneous T-cell lymphoma, 502 506. See also Cutaneous T-cell lymphoma,
DESCRIPTIONS FOR MED 3 ROTATIONS Dermatology A3S
 Regardless of your future field of practice, you will be exposed to a considerable amount of dermatology and this rotation provides you the chance to see a range of skin diseases. You will have the opportunity
Regardless of your future field of practice, you will be exposed to a considerable amount of dermatology and this rotation provides you the chance to see a range of skin diseases. You will have the opportunity
Središnja medicinska knjižnica
 Središnja medicinska knjižnica Kaštelan, D., Kaštelan, M., Prpić Massari, L., Koršić, M. (2006) Possible association of psoriasis and reduced bone mineral density due to increased TNF-alpha and IL-6 concentrations.
Središnja medicinska knjižnica Kaštelan, D., Kaštelan, M., Prpić Massari, L., Koršić, M. (2006) Possible association of psoriasis and reduced bone mineral density due to increased TNF-alpha and IL-6 concentrations.
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Cutaneous Immunology: Innate Immune Responses. Skin Biology Lecture Series
 Cutaneous Immunology: Innate Immune Responses Skin Biology Lecture Series The Immune Response: Innate and Adaptive Components Source: Wolff, Goldsmith, Katz, Gilchrest, Paller, Leffell. Fitzpatrick s Dermatology
Cutaneous Immunology: Innate Immune Responses Skin Biology Lecture Series The Immune Response: Innate and Adaptive Components Source: Wolff, Goldsmith, Katz, Gilchrest, Paller, Leffell. Fitzpatrick s Dermatology
What prescribers need to know
 HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65
 Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Remicade (infliximab) DRUG.00002
 Applicability/Effective Date *- Florida Healthy Kids Remicade (infliximab) DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Approval Duration 1 year Comment Intravenous
Applicability/Effective Date *- Florida Healthy Kids Remicade (infliximab) DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Approval Duration 1 year Comment Intravenous
LINKING PATHOPHYSIOLOGY TO CLINICAL OPTIONS IN PSORIASIS: NEW INSIGHTS INTO INTERLEUKIN-23
 LINKING PATHOPHYSIOLOGY TO CLINICAL OPTIONS IN PSORIASIS: NEW INSIGHTS INTO INTERLEUKIN-23 This symposium took place on 29 th September 2017, as part of the 47th Annual European Society for Dermatological
LINKING PATHOPHYSIOLOGY TO CLINICAL OPTIONS IN PSORIASIS: NEW INSIGHTS INTO INTERLEUKIN-23 This symposium took place on 29 th September 2017, as part of the 47th Annual European Society for Dermatological
Index. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Adhesive tape impression 1 Diff Quik in EPD diagnosis, 583 Allergy(ies). See also Food allergy; specific types, e.g., Culicoides hypersensitivity
Note: Page numbers of article titles are in boldface type. A Adhesive tape impression 1 Diff Quik in EPD diagnosis, 583 Allergy(ies). See also Food allergy; specific types, e.g., Culicoides hypersensitivity
Diseases of Immunity 2017 CL Davis General Pathology. Paul W. Snyder, DVM, PhD Experimental Pathology Laboratories, Inc.
 Diseases of Immunity 2017 CL Davis General Pathology Paul W. Snyder, DVM, PhD Experimental Pathology Laboratories, Inc. Autoimmunity Reflects a loss of immunologic tolerance Mechanisms Auto-antibodies
Diseases of Immunity 2017 CL Davis General Pathology Paul W. Snyder, DVM, PhD Experimental Pathology Laboratories, Inc. Autoimmunity Reflects a loss of immunologic tolerance Mechanisms Auto-antibodies
Undergraduate Dermatology Curriculum July 2016
 Undergraduate Dermatology Curriculum July 2016 British Association of Dermatologists Introduction This document is the 2016 revised dermatology undergraduate curriculum (UK) from the British Association
Undergraduate Dermatology Curriculum July 2016 British Association of Dermatologists Introduction This document is the 2016 revised dermatology undergraduate curriculum (UK) from the British Association
Table of Contents: Part 1 Medical Dermatology. Chapter 1 Acneiform Disorders. Acne. Acne Vulgaris. Pomade Acne. Steroid Acne
 Table of Contents: Part 1 Medical Dermatology Chapter 1 Acneiform Disorders Acne Acne Vulgaris Pomade Acne Steroid Acne Infantile Acne Pediatric Perspectives Neonatal Acne (Acne Neonatorum) Pediatric Perspectives
Table of Contents: Part 1 Medical Dermatology Chapter 1 Acneiform Disorders Acne Acne Vulgaris Pomade Acne Steroid Acne Infantile Acne Pediatric Perspectives Neonatal Acne (Acne Neonatorum) Pediatric Perspectives
Skin cancers in patients treated with immunomodulating drugs. Manuelle Viguier, MD, PhD Dermatology department Saint-Louis Hospital Paris, France
 Skin cancers in patients treated with immunomodulating drugs Manuelle Viguier, MD, PhD Dermatology department Saint-Louis Hospital Paris, France Immunomodulating drugs used in Methotrexate Mycophenolate
Skin cancers in patients treated with immunomodulating drugs Manuelle Viguier, MD, PhD Dermatology department Saint-Louis Hospital Paris, France Immunomodulating drugs used in Methotrexate Mycophenolate
Recent insights into atopic dermatitis and implications for management of infectious complications
 Mark Boguniewicz, MD Professor, Division of Allergy-Immunology Department of Pediatrics National Jewish Health and University of Colorado School of Medicine Denver, Colorado USA Recent insights into atopic
Mark Boguniewicz, MD Professor, Division of Allergy-Immunology Department of Pediatrics National Jewish Health and University of Colorado School of Medicine Denver, Colorado USA Recent insights into atopic
Innate immunity. Abul K. Abbas University of California San Francisco. FOCiS
 1 Innate immunity Abul K. Abbas University of California San Francisco FOCiS 2 Lecture outline Components of innate immunity Recognition of microbes and dead cells Toll Like Receptors NOD Like Receptors/Inflammasome
1 Innate immunity Abul K. Abbas University of California San Francisco FOCiS 2 Lecture outline Components of innate immunity Recognition of microbes and dead cells Toll Like Receptors NOD Like Receptors/Inflammasome
Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds
 Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds Scott Stienecker MD FACP FSHEA Medical Director for Epidemiology and Infection Prevention Parkview
Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds Scott Stienecker MD FACP FSHEA Medical Director for Epidemiology and Infection Prevention Parkview
Infliximab/Infliximab-dyyb DRUG.00002
 Infliximab/Infliximab-dyyb DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Inflectra (inflectra-dyyb) Approval Duration 1 year Comment Intravenous administration
Infliximab/Infliximab-dyyb DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Inflectra (inflectra-dyyb) Approval Duration 1 year Comment Intravenous administration
This questionnaire was used both during the face-to-face interviews with the
 Additional file 1: Primary research questionnaire This questionnaire was used both during the face-to-face interviews with the dermatologists and during the expert panel 1. During the last month, how many
Additional file 1: Primary research questionnaire This questionnaire was used both during the face-to-face interviews with the dermatologists and during the expert panel 1. During the last month, how many
Cutaneous side effects of antietumor necrosis factor biologic therapy: A clinical review
 REVIEW Cutaneous side effects of antietumor necrosis factor biologic therapy: A clinical review Aikaterini-Evaggelia Moustou, MD, a Athina Matekovits, MD, a Clio Dessinioti, MD, a Christina Antoniou, MD,
REVIEW Cutaneous side effects of antietumor necrosis factor biologic therapy: A clinical review Aikaterini-Evaggelia Moustou, MD, a Athina Matekovits, MD, a Clio Dessinioti, MD, a Christina Antoniou, MD,
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Medication Policy Manual. Topic: Otezla, apremilast Date of Origin: May 9, 2014
 Medication Policy Manual Policy No: dru342 Topic: Otezla, apremilast Date of Origin: May 9, 2014 Committee Approval Date: January 19, 2015 Next Review Date: January 2016 Effective Date: April 1, 2015 IMPORTANT
Medication Policy Manual Policy No: dru342 Topic: Otezla, apremilast Date of Origin: May 9, 2014 Committee Approval Date: January 19, 2015 Next Review Date: January 2016 Effective Date: April 1, 2015 IMPORTANT
Hot topics in Pediatric Dermatology. Yvonne Chiu, MD
 Hot topics in Pediatric Dermatology Yvonne Chiu, MD Hot Topics for Pediatric Dermatology Yvonne Chiu, MD WDS Summer Meeting July 21, 2012 Disclosure Statement I, Yvonne Chiu, MD, do not have any relevant
Hot topics in Pediatric Dermatology Yvonne Chiu, MD Hot Topics for Pediatric Dermatology Yvonne Chiu, MD WDS Summer Meeting July 21, 2012 Disclosure Statement I, Yvonne Chiu, MD, do not have any relevant
Psoriasiform Dermatitis in Children: Calling in the Troops
 Psoriasiform Dermatitis in Children: Calling in the Troops Markus Boos, MD PhD Attending Physician, Dermatology Seattle Children s Hospital Assistant Professor of Pediatrics, University of Washington School
Psoriasiform Dermatitis in Children: Calling in the Troops Markus Boos, MD PhD Attending Physician, Dermatology Seattle Children s Hospital Assistant Professor of Pediatrics, University of Washington School
CPC. Chutika Srisuttiyakorn, M.D. Kobkul Aunhachoke, M.D. Phramongkutklao Hospital Bangkok, Thailand
 CPC Chutika Srisuttiyakorn, M.D. Kobkul Aunhachoke, M.D. Phramongkutklao Hospital Bangkok, Thailand A 53 year-old woman with fever, facial swelling and rashes on face, trunk and upper extremities for 3
CPC Chutika Srisuttiyakorn, M.D. Kobkul Aunhachoke, M.D. Phramongkutklao Hospital Bangkok, Thailand A 53 year-old woman with fever, facial swelling and rashes on face, trunk and upper extremities for 3
Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG CG-DRUG-64
 Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG.00002 CG-DRUG-64 Override(s) Prior Authorization *Washington Medicaid See State Specific Mandates Medications Inflectra
Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG.00002 CG-DRUG-64 Override(s) Prior Authorization *Washington Medicaid See State Specific Mandates Medications Inflectra
Systemic Medications for the Dermatology Toolbox: Azathioprine
 Systemic Medications for the Dermatology Toolbox: Azathioprine Wynnis Tom, M.D. Associate Clinical Professor University of California, San Diego and Rady Children s Hospital Faculty Disclosure Information
Systemic Medications for the Dermatology Toolbox: Azathioprine Wynnis Tom, M.D. Associate Clinical Professor University of California, San Diego and Rady Children s Hospital Faculty Disclosure Information
Use of extrapolation in small clinical trials:
 Use of extrapolation in small clinical trials: Infliximab for pediatric ulcerative colitis Jessica J. Lee, MD, MMSc Medical Officer Division of Gastroenterology and Inborn Errors Products CDER/ FDA 1 Learning
Use of extrapolation in small clinical trials: Infliximab for pediatric ulcerative colitis Jessica J. Lee, MD, MMSc Medical Officer Division of Gastroenterology and Inborn Errors Products CDER/ FDA 1 Learning
Humira. (adalimumab) Drug Update Slideshow NEW INDICATION
 Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
HELPING YOU AND YOUR PATIENTS TALK OPENLY ABOUT MODERATELY TO SEVERELY ACTIVE RA
 SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 6 Last Review Date: December 8, 2017 Stelara Description Stelara (ustekinumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 6 Last Review Date: December 8, 2017 Stelara Description Stelara (ustekinumab)
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Immunity to Viruses. Patricia Fitzgerald-Bocarsly September 25, 2008
 Immunity to Viruses Patricia Fitzgerald-Bocarsly September 25, 2008 The Immune System Deals with a Huge Range of Pathogens Roitt, 2003 Immune Responses to Viruses Viruses are dependent on the host cell
Immunity to Viruses Patricia Fitzgerald-Bocarsly September 25, 2008 The Immune System Deals with a Huge Range of Pathogens Roitt, 2003 Immune Responses to Viruses Viruses are dependent on the host cell
Regulatory Status FDA-approved indication: Humira and Amjevita are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
REGISTRY OF SEVERE CUTANEOUS ADVERSE REACTIONS TO DRUGS AND COLLECTION OF BIOLOGICAL SAMPLES. R e g i S C A R PATIENT'S DATA. Age country of birth
 REGISTRY OF SEVERE CUTANEOUS ADVERSE REACTIONS TO DRUGS AND COLLECTION OF BIOLOGICAL SAMPLES R e g i S C A R PATIENT'S DATA Initials of the patient date of birth Age country of birth Gender male female
REGISTRY OF SEVERE CUTANEOUS ADVERSE REACTIONS TO DRUGS AND COLLECTION OF BIOLOGICAL SAMPLES R e g i S C A R PATIENT'S DATA Initials of the patient date of birth Age country of birth Gender male female
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Humira (adalimumab) DRUG.00002
 Humira (adalimumab) DRUG.00002 Override(s) Prior Authorization Quantity Limit Approval Duration 1 year Medications Humira 10 mg/0.2 ml syringe Humira pediatric Crohn s Disease starter pack 40 mg/0.8 ml
Humira (adalimumab) DRUG.00002 Override(s) Prior Authorization Quantity Limit Approval Duration 1 year Medications Humira 10 mg/0.2 ml syringe Humira pediatric Crohn s Disease starter pack 40 mg/0.8 ml
Immune Modulating Drugs Prior Authorization Request Form
 Patient: HPHC member ID #: Requesting provider: Phone: Servicing provider: Diagnosis: Contact for questions (name and phone #): Projected start and end date for requested Requesting provider NPI: Fax:
Patient: HPHC member ID #: Requesting provider: Phone: Servicing provider: Diagnosis: Contact for questions (name and phone #): Projected start and end date for requested Requesting provider NPI: Fax:
C. Assess clinical response after the first three months of treatment.
 Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Autoimmune diseases, their pathogenic mechanisms and treatment of unwanted immune responses (Janeway s Immunobiology)
 Autoimmune diseases, their pathogenic mechanisms and treatment of unwanted immune responses (Janeway s Immunobiology) Picture source: https://www.klini kum.uniheidelberg.de/fil eadmin/pressest elle/pm_neu/20
Autoimmune diseases, their pathogenic mechanisms and treatment of unwanted immune responses (Janeway s Immunobiology) Picture source: https://www.klini kum.uniheidelberg.de/fil eadmin/pressest elle/pm_neu/20
The Natural History of Psoriasis and Treatment Goals
 The Natural History of Psoriasis and Treatment Goals Psoriasis Epidemiology Prevalence Affects 2 3% of adult population (>7 million in US) Caucasians: 25% 2.5% African Americans: 1.3% (more likely to have
The Natural History of Psoriasis and Treatment Goals Psoriasis Epidemiology Prevalence Affects 2 3% of adult population (>7 million in US) Caucasians: 25% 2.5% African Americans: 1.3% (more likely to have
An otherwise healthy 12-year-old
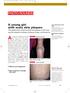 1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
Paradoxial Safety Signals from Biologics
 Paradoxial Safety Signals from Biologics Christopher Ritchlin, MD, MPH Professor of Medicine Director, Translational Immunology Research Center University of Rochester Medical Center Rochester, NY Disclosures
Paradoxial Safety Signals from Biologics Christopher Ritchlin, MD, MPH Professor of Medicine Director, Translational Immunology Research Center University of Rochester Medical Center Rochester, NY Disclosures
Measles virus vaccine induces oncolysis of tumor cells and activates immune responses.
 Measles virus vaccine induces oncolysis of tumor cells and activates immune responses. Marc Grégoire*, J.-F. Fonteneau, N. Boisgerault, J.-B. Guillerme and F. Tangy. * INSERM, Nantes, F-44, France. Pasteur
Measles virus vaccine induces oncolysis of tumor cells and activates immune responses. Marc Grégoire*, J.-F. Fonteneau, N. Boisgerault, J.-B. Guillerme and F. Tangy. * INSERM, Nantes, F-44, France. Pasteur
SINGLE CHOICE. 5. The gamma invariant chain binds to this molecule during its intracytoplasmic transport. A TCR B BCR C MHC II D MHC I E FcγR
 A Name: Group: SINGLE CHOICE 1. Which is the most important ligand of TLR5? A endospore B flagellin C polysaccharide capsule D DNA E pilus 2. The antibody-binding site is formed primarily by... A the constant
A Name: Group: SINGLE CHOICE 1. Which is the most important ligand of TLR5? A endospore B flagellin C polysaccharide capsule D DNA E pilus 2. The antibody-binding site is formed primarily by... A the constant
Evolving therapies for posterior uveitis. Infliximab (Remicade) Infliximab: pharmacology. FDA-approved monoclonal antibody therapy Target
 Evolving therapies for posterior uveitis Sam Dahr, M.D. September 17, 2005 Midwest Ophthalmology Conference Infliximab (Remicade) FDA approved for Crohn s disease, rheumatoid arthritis, and psoriatic arthritis
Evolving therapies for posterior uveitis Sam Dahr, M.D. September 17, 2005 Midwest Ophthalmology Conference Infliximab (Remicade) FDA approved for Crohn s disease, rheumatoid arthritis, and psoriatic arthritis
10/25/2018. Autoimmunity and how to treat it. Disclosure. Why do we get autoimmunity? James Verbsky MD/PhD Pediatric Rheumatology/Immunology
 Autoimmunity and how to treat it James Verbsky MD/PhD Pediatric Rheumatology/Immunology Disclosure None I will mention drug names and some brand names but I have no financial interest or any other ties
Autoimmunity and how to treat it James Verbsky MD/PhD Pediatric Rheumatology/Immunology Disclosure None I will mention drug names and some brand names but I have no financial interest or any other ties
DNA vaccine, peripheral T-cell tolerance modulation 185
 Subject Index Airway hyperresponsiveness (AHR) animal models 41 43 asthma inhibition 45 overview 41 mast cell modulation of T-cells 62 64 respiratory tolerance 40, 41 Tregs inhibition role 44 respiratory
Subject Index Airway hyperresponsiveness (AHR) animal models 41 43 asthma inhibition 45 overview 41 mast cell modulation of T-cells 62 64 respiratory tolerance 40, 41 Tregs inhibition role 44 respiratory
Autoimmunity. Autoimmunity arises because of defects in central or peripheral tolerance of lymphocytes to selfantigens
 Autoimmunity Autoimmunity arises because of defects in central or peripheral tolerance of lymphocytes to selfantigens Autoimmune disease can be caused to primary defects in B cells, T cells and possibly
Autoimmunity Autoimmunity arises because of defects in central or peripheral tolerance of lymphocytes to selfantigens Autoimmune disease can be caused to primary defects in B cells, T cells and possibly
Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y.
 UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
Simponi / Simponi ARIA (golimumab)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 6 Last Review Date: September 15, 2016 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 6 Last Review Date: September 15, 2016 Simponi / Simponi
Priority Health Medicare prior authorization form Fax completed form to: toll free, or
 Priority Health Medicare prior authorization form Fax completed form to: 877.974.4411 toll free, or 616.942.8206 This form applies to: Medicare Part B Medicare Part D This request is: Expedited request
Priority Health Medicare prior authorization form Fax completed form to: 877.974.4411 toll free, or 616.942.8206 This form applies to: Medicare Part B Medicare Part D This request is: Expedited request
ESCMID Online Lecture Library. by author
 Therapies or diseases predisposing to infection Congenital immunodeficiences Acquired conditions Common variable immunedeficiency particular diseases (e.g. HIV, cancer,leukemia, lymphoma, diabetes, cystic
Therapies or diseases predisposing to infection Congenital immunodeficiences Acquired conditions Common variable immunedeficiency particular diseases (e.g. HIV, cancer,leukemia, lymphoma, diabetes, cystic
INFLIXIMAB Remicade (infliximab), Inflectra (infliximab-dyyb), Ixifi* (infliximabqbtx), Renflexis (infliximab-abda)
 Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
TUBERCULOSIS AND THE TNF-α INHIBITORS. Lloyd Friedman, M.D. Yale University Milford Hospital
 TUBERCULOSIS AND THE TNF-α INHIBITORS Lloyd Friedman, M.D. Yale University Milford Hospital Outline TNF-α Anti-TNF-α medications Rates of tuberculosis Lower rates with etanercept Screening for latent tuberculosis
TUBERCULOSIS AND THE TNF-α INHIBITORS Lloyd Friedman, M.D. Yale University Milford Hospital Outline TNF-α Anti-TNF-α medications Rates of tuberculosis Lower rates with etanercept Screening for latent tuberculosis
Case Example: Exposure Response to Support Extrapolation of Efficacy in Pediatric Ulcerative Colitis
 Case Example: Exposure Response to Support Extrapolation of Efficacy in Pediatric Ulcerative Colitis Kerry Jo Lee, MD Medical Officer Division of Gastroenterology and Inborn Error Products Office of New
Case Example: Exposure Response to Support Extrapolation of Efficacy in Pediatric Ulcerative Colitis Kerry Jo Lee, MD Medical Officer Division of Gastroenterology and Inborn Error Products Office of New
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
Rayos Prior Authorization Program Summary
 Rayos Prior Authorization Program Summary FDA APPROVED INDICATIONS AND DOSAGE FDA-Approved Indications: 1 Agent Indication Dosage Rayos (prednisone delayedrelease tablet) as an anti-inflammatory or immunosuppressive
Rayos Prior Authorization Program Summary FDA APPROVED INDICATIONS AND DOSAGE FDA-Approved Indications: 1 Agent Indication Dosage Rayos (prednisone delayedrelease tablet) as an anti-inflammatory or immunosuppressive
What's New in Oncodermatopathology: Immunotherapy Reactions
 What's New in Oncodermatopathology: Immunotherapy Reactions Emily Y. Chu, M.D., Ph.D. Assistant Professor of Dermatology & Pathology and Laboratory Medicine Hospital of the University of Pennsylvania March
What's New in Oncodermatopathology: Immunotherapy Reactions Emily Y. Chu, M.D., Ph.D. Assistant Professor of Dermatology & Pathology and Laboratory Medicine Hospital of the University of Pennsylvania March
Drug Name (specify drug) Quantity Frequency Strength
 Prior Authorization Form GEHA FEDERAL - STANDARD OPTION Autoimmune Conditions (FA-PA) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Prior Authorization Form GEHA FEDERAL - STANDARD OPTION Autoimmune Conditions (FA-PA) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Outline. Biologic Drugs in Inflammatory Bowel Disease Dr. Jason Etzel MD The Vancouver Clinic. Biologic Drugs. Biologic Drugs. Biologic Drugs Anti-TNF
 Outline Biologic Drugs in Inflammatory Bowel Disease Dr. Jason Etzel MD The Vancouver Clinic Types of biologic drugs How do they work? How effective are they? Safety/Toxicity concerns with biologics Biologic
Outline Biologic Drugs in Inflammatory Bowel Disease Dr. Jason Etzel MD The Vancouver Clinic Types of biologic drugs How do they work? How effective are they? Safety/Toxicity concerns with biologics Biologic
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 3 October 2012
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 3 October 2012 REMICADE 100 mg, powder for concentrate for solution for infusion B/1 vial (CIP code: 562 070-1) Applicant:
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 3 October 2012 REMICADE 100 mg, powder for concentrate for solution for infusion B/1 vial (CIP code: 562 070-1) Applicant:
Supplemental Figure 1. IL-3 blockade with Fab CSL362 depletes plasmacytoid dendritic cells (pdcs), but not basophils, at higher doses.
 Supplemental Figure 1. IL-3 blockade with Fab CSL362 depletes plasmacytoid dendritic cells (pdcs), but not basophils, at higher doses. Percentage of viable (A) pdcs (Sytox Blue-, Lin1-, HLADR+, BDCA2++)
Supplemental Figure 1. IL-3 blockade with Fab CSL362 depletes plasmacytoid dendritic cells (pdcs), but not basophils, at higher doses. Percentage of viable (A) pdcs (Sytox Blue-, Lin1-, HLADR+, BDCA2++)
Michael P. Heffernan, M.D San Luis Dermatology & Laser Clinic Director, US Probity Medical Research
 Michael P. Heffernan, M.D San Luis Dermatology & Laser Clinic Director, US Probity Medical Research mpheffernanmd@gmail.com DISCLOSURES Consultant, Speaker, Investigator: Abbvie, Amgen, Brickell Biotech,
Michael P. Heffernan, M.D San Luis Dermatology & Laser Clinic Director, US Probity Medical Research mpheffernanmd@gmail.com DISCLOSURES Consultant, Speaker, Investigator: Abbvie, Amgen, Brickell Biotech,
Immunological Tolerance
 Immunological Tolerance Introduction Definition: Unresponsiveness to an antigen that is induced by exposure to that antigen Tolerogen = tolerogenic antigen = antigen that induces tolerance Important for
Immunological Tolerance Introduction Definition: Unresponsiveness to an antigen that is induced by exposure to that antigen Tolerogen = tolerogenic antigen = antigen that induces tolerance Important for
Subject: Remicade (Page 1 of 5)
 Subject: Remicade (Page 1 of 5) Objective: I. To ensure that Health Share/Tuality Health Alliance (THA) has a process by which the appropriate utilization of Remicade (Infliximab) for members whose diagnosis
Subject: Remicade (Page 1 of 5) Objective: I. To ensure that Health Share/Tuality Health Alliance (THA) has a process by which the appropriate utilization of Remicade (Infliximab) for members whose diagnosis
Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics
 Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics Dr Sarah Sasson SydPATH Registrar 23 rd June 2014 TLRs: Introduction Discovered in 1990s Recognise conserved structures in pathogens Rely
Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics Dr Sarah Sasson SydPATH Registrar 23 rd June 2014 TLRs: Introduction Discovered in 1990s Recognise conserved structures in pathogens Rely
It is estimated that about 26,000 new cases of
 Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
DISCLOSURES. Online A. Infectious Complications of Monoclonal Antibody Therapies 6/22/2012. Cytokine blocking. Lymphocyte depleting.
 Online A Infectious Complications of Monoclonal Antibody Therapies Steven M. Holland, M.D. Off-Label Usage None DISCLOSURES Financial Relationships with Relevant Commercial Interests None Resolution: N/A
Online A Infectious Complications of Monoclonal Antibody Therapies Steven M. Holland, M.D. Off-Label Usage None DISCLOSURES Financial Relationships with Relevant Commercial Interests None Resolution: N/A
Announcing HUMIRA. Psoriasis Starter Package
 Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Oncology and surgery Yolanda Gilaberte. Hospital Universitario Miguel Servet, Zaragoza
 Oncology and surgery Yolanda Gilaberte. Hospital Universitario Miguel Servet, Zaragoza Dermatologic adverse events of oncologic drugs Grading of Dermatologic Adverse Events (ver 5.0) 5 grades: mild, moderate,
Oncology and surgery Yolanda Gilaberte. Hospital Universitario Miguel Servet, Zaragoza Dermatologic adverse events of oncologic drugs Grading of Dermatologic Adverse Events (ver 5.0) 5 grades: mild, moderate,
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Emergent and Urgent Dermatology, Eruptions, and Wound Care
 Emergent and Urgent Dermatology, Eruptions, and Wound Care G. Scott Drew, DO, FAAD, FAOCD Smith Clinic Department of Dermatology Tucson Osteopathic Medical Foundation April 27, 2018 Acute Cutaneous Lupus
Emergent and Urgent Dermatology, Eruptions, and Wound Care G. Scott Drew, DO, FAAD, FAOCD Smith Clinic Department of Dermatology Tucson Osteopathic Medical Foundation April 27, 2018 Acute Cutaneous Lupus
Moderately to severely active ulcerative colitis
 Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
1. TLR. TLR Toll-like receptors. Toll Toll-like receptor, TLR TLR TLR TLR. type I TLR TLR. Toll
 54pp.145 152 2004 1. TLR T B TLR Toll-like receptors TLR TLR I IFN TLR T B B T Toll NF- B 1996 565-0871 3-1 TEL 06-6879-8303 FAX 06-6879-8305 E-mail uemattsu@biken.osaka-u.ac.jp Toll Toll-like receptor,
54pp.145 152 2004 1. TLR T B TLR Toll-like receptors TLR TLR I IFN TLR T B B T Toll NF- B 1996 565-0871 3-1 TEL 06-6879-8303 FAX 06-6879-8305 E-mail uemattsu@biken.osaka-u.ac.jp Toll Toll-like receptor,
PSORIASIS PSORIASIS. Anatomic sites FACTS ABOUT PROSIASIS PSORIASIS 11/16/2017 STIGMATA OF PSORIASIS
 PSORIASIS Dr. Sami Alsuwaidan President, Saudi Society of Dermatology & Dermatologic Surgery Associate Professor and Consultant Department of Dermatology, College of Medicine King Saud University PSORIASIS
PSORIASIS Dr. Sami Alsuwaidan President, Saudi Society of Dermatology & Dermatologic Surgery Associate Professor and Consultant Department of Dermatology, College of Medicine King Saud University PSORIASIS
Medical Management of Rheumatoid Arthritis (RA)
 Medical Management of Rheumatoid Arthritis (RA) Dr Lee-Suan Teh Rheumatologist Royal Blackburn Hospital Educational objectives ABC Appreciate the epidemiology of RA Be able to diagnosis of RA Competent
Medical Management of Rheumatoid Arthritis (RA) Dr Lee-Suan Teh Rheumatologist Royal Blackburn Hospital Educational objectives ABC Appreciate the epidemiology of RA Be able to diagnosis of RA Competent
The implication of an immunologic
 Evidence-Based Review of Biologic Therapy for Psoriasis: Infliximab, Etanercept, Adalimumab, Efalizumab, and Alefacept Jeffrey M. Weinberg, MD; Robin Buchholz, MD; Noah Scheinfeld, MD DERMATOLOGY ABSTRACT
Evidence-Based Review of Biologic Therapy for Psoriasis: Infliximab, Etanercept, Adalimumab, Efalizumab, and Alefacept Jeffrey M. Weinberg, MD; Robin Buchholz, MD; Noah Scheinfeld, MD DERMATOLOGY ABSTRACT
Biologic agents in Internal Medicine-2018: Targeted therapies for.
 Biologic agents in Internal Medicine-2018: Targeted therapies for. Chronic inflammatory diseases affecting the skin Chronic inflammatory diseases affecting the gut Chronic inflammatory diseases affecting
Biologic agents in Internal Medicine-2018: Targeted therapies for. Chronic inflammatory diseases affecting the skin Chronic inflammatory diseases affecting the gut Chronic inflammatory diseases affecting
1 P a g e. Systemic Juvenile Idiopathic Arthritis (SJIA) (1.3) Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
 LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of:
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Cell-Mediated Immunity and T Lymphocytes
 Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release)
 Market DC Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release) Override(s) Prior Authorization Quantity Limit Medications Xeljanz (tofacitinib) Approval Duration 1 year Quantity Limit May be
Market DC Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release) Override(s) Prior Authorization Quantity Limit Medications Xeljanz (tofacitinib) Approval Duration 1 year Quantity Limit May be
Sarcoidosis: is there a role for anti-tnf-α?
 Sarcoidosis: is there a role for anti-tnf-α? Abstract In severe cases of sarcoidosis treatment can be very difficult. The common treatment strategies might be failing. Tumour necrosis factor (TNF) α therapy
Sarcoidosis: is there a role for anti-tnf-α? Abstract In severe cases of sarcoidosis treatment can be very difficult. The common treatment strategies might be failing. Tumour necrosis factor (TNF) α therapy
Psoriasis: Therapeutic goals
 Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Clinical Policy: Abatacept (Orencia) Reference Number: ERX.SPA.123 Effective Date:
 Clinical Policy: (Orencia) Reference Number: ERX.SPA.123 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Orencia) Reference Number: ERX.SPA.123 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs)
 Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Skin Immune Mechanisms in Health and Disease
 Skin Immune Mechanisms in Health and Disease by Barbara S. Baker Published by Garner Press (http://www.garnerpress.com) Copyright 2006 Garner Press All rights reserved. This book, or parts thereof, may
Skin Immune Mechanisms in Health and Disease by Barbara S. Baker Published by Garner Press (http://www.garnerpress.com) Copyright 2006 Garner Press All rights reserved. This book, or parts thereof, may
Environmental factors including drugs. Jyoti Ranjan Parida
 Environmental factors including drugs Jyoti Ranjan Parida Scheme of presentation Pathogenesis of autoimmune disease Evidence for environmental influence Environmental factors Proposed -Role of Sunlight
Environmental factors including drugs Jyoti Ranjan Parida Scheme of presentation Pathogenesis of autoimmune disease Evidence for environmental influence Environmental factors Proposed -Role of Sunlight
