Bricanyl Turbuhaler in the treatment of asthma: a six week multi-centre study carried out in Sweden, the United Kingdom, Denmark, Norway and Finland
|
|
|
- Adam Flowers
- 5 years ago
- Views:
Transcription
1 Eur Resplr J 1991, 4, Bricanyl Turbuhaler in the treatment of asthma: a six week multi-centre study carried out in Sweden, the United Kingdom, Denmark, Norway and Finland K. Osterman*, E. StAhl**, A. Kallent Bricanyl Turbuhaler* in the treatment of asthma: a six week multi-centre study carried out in Sweden, the United Kingdom, Denmark, Norway and Finland. K. Osterman, E. Stdhl, A. Kiillen. ABSTRACT: Two hundred and fifty eight adult patients with bronchial asthma participated In an open, parallel group study of 8 wks duration. During a 2 wk run-in period the 8 3 -agonlst terbutaline (.5 mg q.i.d) was delivered via a pressurized freon aerosol, Bricanyl metered dose inhaler (MDI). During the following 6 wks, one third of the patients continued with MDI and two thirds with the same dose of terbutallne in the form of dry powder, Bricanyl Turbuhaler. There were no statistically significant differences in the increases in peak expiratory flow rate (PEFR) after inhalation between the two treatment groups. The asthma symptom scores decreased in the Turbuhaler group during the study but remained the same in the MDI group. No difference in use of extra trial medication during the day was found. During the night, the difference was statistically significant in favour of Turbuhaler (p<o.os). Turbuhaler was well accepted and easy to use. As compared with the MDI used during run-in, SO% of the patients in the Turbubaler group preferred to use Turbubaler and 26% MDI, whilst 24% had no preference. In conclusion, Turbuhaler was at least as effective as MDI during the whole Turbuhaler life-span (6 wks). Eur Respir J., 1991, 4, Dept of Allergology, Karolinska sjukhuset, Stockholm, Sweden. Medical Dept, AB Draco, Lund, Sweden. t Dept of Biostatistics, AB Draco, Lund, Sweden. Correspondence: K. Osterman, Dept of Allergy, Karolinska sjukhuset, S-14 1 Stockholm, Sweden. Keywords: Acceptance; asthma; powder inhaler; preference; terbutaline; Turbuhaler. Received: November 3, 1989; accepted after revision August 27, 199. Participating investigators: R. Dahl, J. Fredriksen, E.R. Weeke (Denamrk); K. Kiviranta, K. Lehtonen, (Finland); C.H. Hagelund, N. Ringdal, (Norway); G. Malmer, H. Matsols, A-M. Norborg, J. Perk, G. Svensson, (Sweden); M. Bone, M.G. Britton, A.J. Johnson, R.L. Joshi, F. Moran, P.R. Studdy, (UK). Several anti-asthmatic drugs, such as agonists, corticosteroids and ipratropium bromide, are available as inhalation therapy. The most commonly used inhalation device is the pressurized metered dose inhaler (MDI). However, many patients have problems with MDis due to difficulties in co-ordinating actuation of the inhaler with inhalation [1]. MDis also contain freons and lubricants which may further irritate the hyperreactive airways of asthmatic patients [2]. In order to reduce these problems alternative devices, such as Turbuhaler [3], have been developed. Bricanyl Turbuhaler is a breath-actuated dry powder inhaler which delivers pure terbutaline sulphate. It is preloaded with 2 doses of.5 mg terbutaline sulphate and contains no additives. Since there is a steady increase in pollution, the absence of freons will be an advantage for the environment. Previous studies have shown that Bricanyl Turbuhaler treatment is equipotent to Bricanyl MDI [4-6]. The aim of this study was to evaluate the acceptability and efficacy of Bricanyl Turbuhaler in the course of its life-span in regular treatment of asthma. Patients and methods Two hundred and fifty eight patients (145 females) with stable asthma and forced expiratory volume in one second (FEV ) 1 of 1 l or more entered the study. All patients required inhaled agonists daily. Twenty seven patients did not complete the study according to the protocol: six patients did not return to the clinic and 21 patients dropped out because of deterioration of asthma or concurrent illness (17 patients in the Turbuhaler group and four in the MDI group). The mean age of the patients was 48 yrs (range yrs). Demographic data are shown in table t and maintenance therapy in table 2. The study was of an open, parallel group design, with a total duration of 8 wks, including a two week run-in period in which Bricanyl MDI was used (2 x.25 mg terbutaline sulphate q. i.d. ). The patients measured peak expiratory flow rate (PEFR) every morning and evening before and after the inhalation. During week 2, the patients had to show an increase in PEFR after inhalation of.5 mg terbutaline sulphate of at least 15%
2 176 K. OSTERMAN, E. STAHL, A. KALIEN Table 1. - Demographic data for each treatment group Turbuhaler MDI n=177 n=81 Age yrs 48 (17-74) 47 (19-77) Duration of 16 (1-6) 15 (1-45) disease yrs Reversibility 27 (15-79) 26 (15-78) Data are mean and range in parenthesis. MDI: pressurized metered dose inhaler. Table 2. - Maintenance therapy for each treatment group Concomitant medication Turbuhaler MDI n=177 n=81 Oral j3 -agonists 27% 22% Inhaled steroids 82% 74% Oral steroids 16% 11% Anticholinergics 4% % Theophylline 56% 53% Number of patients in %. MDI: pressurized metered dose inhaler. on at least one occasion at home using a mini Wright peak flow meter in order to be included in the study. After the run-in period, the patients were randomly divided into two groups. One third of the patients continued with MDI treatment and two thirds with the same dose of Bricanyl Turbuhaler, i.e..5 mg of terbutaline sulphate q.i.d. (on waking, at midday, late afternoon and before going to bed). The patients received the instruction leaflet provided by the manufacturers and were also instructed verbally. Extra inhalations of terbutaline were allowed and were recorded every morning and evening in diary cards, together with PEFR and asthma symptoms according to the following scoring system: = none; 1 = mild; 2 = moderate; and 3 = severe. The patients in the Turbuhaler group were also given a standard questionnaire about inhaler use. The question about preference was: "Which inhaler do you prefer to use?" The patients were informed verbally and in writing and their informed consent was obtained. The study was approved by the local Ethics Committees. Efficacy variables (PEFR, asthma symptom score (--3) and number of extra inhalations) were defined as the mean during the treatment period minus the mean during the run-in period. Table 3. - PEFR morning values Weekly means Turbuhaler group MDI group Week Before After Increase Before After Increase Run-in: : Period mean Run-in Turbuhaler group Before After Increase MDI group Before After Increase Within-group analysis Between-group analysis Turbuhaler MDI Turb.-MDI p-value MDI: pressurized metered dose inhaler.
3 A SIX WEEK TREATMENT WITII BRICANYL TURBUHALER 177 PEFR Day a Fig Mean PEFR values in the morning (before inhalation). - : Turbuhaler group (n=177); - - : MDI group (n=sl). Note that both groups used MDI during the run-in period. MDI: pressurized metered dose inhaler; PEFR: peak expiratory flow rate. Statistical methods PEFR were analysed using multivariate techniques (Hotelling's T2) on the two variables: measurement before inhalation and measurement after inhalation. In addition to the between-group comparison, the temporal behaviour of PEFR was analysed using a non-parametric trend test (Page's test). The distribution of asthma scores, 1, 2, 3 on an arbitrary day was estimated for descriptive purposes. These analyses were conducted for the run-in period and for the treatment period using a stationary Markov chain model [7]. Results Out of 258 patients, 177 were allocated to the Turbuhaler group and 81 to the MDI group. Peak flow Mean PEFR values in the morning in the Turbuhaler group before inhalation were 321 lmin 1 during run-in (MDI) and 328 l min 1 with Turbuhaler. The corresponding values in the MDI group were 339 lmin 1 and 338 Z.min 1 The increase in the morning values in the Turbuhaler group was statistically significant (p=.26). The increases in PEFR after inhalation were 56 Z.min 1 during run-in and 53 lmin 1 during the treatment in the Turbuhaler group. In the MDI group the mean increases in PEFR were 56 lmin 1 and 55 lmin 1 after run-in and treatment, respectively. No statistically significant differences were seen between the groups (table 3). The daily means of PEFR before inhalation were analysed for the detection of a trend with time. The daily means differed only little between days, and the mean value curves were very similar for the two groups (fig. 1). Asthma symptoms The two treatment groups were compared using the mean score during the treatment period minus mean score during the run-in period. There were no statistically significant differences in the between-group analysis (figs 2 and 3). Additional medication, patient preference Mean consumption of extra trial medication during the daytime was analysed. There was a decrease of.3 inhalations from run-in to treatment period in the Turbuhaler group (p<.5). In the MDI group there was no statistically significant difference from run-in to treatment period. The inter-group analysis did not show a significant difference between the two treatments. During the night, there was an inter-group difference of.21 inhalations (p<.5) in favour of Turbuhaler. Fifty six percent of the patients in the Turbuhaler group found both inhalers practical to carry around. Thirty three percent of the patients found Turbuhaler
4 178 K. OSTERMAN, E. STAin., A. KALI.EN Turbuhaler roup (n=177 % of patients % of patients 1 1 MOl group (n=81) Fig Mean asthma symptom score during the day (O=none; l=mild; 2=moderate; 3=severe). CJ : run-in (MDI); ~ : treatment (Turbuhaler); [=:J : treatment (MDI); MDI: pressurized metered dose inhaler. % of patients Turbuhaler group (n=177) % of patients MOl group (n=81) Fig Mean asthma symptom score during the night (O=none; l=mild; 2=moderate; 3=severe). c::::j: run-in (MDI); ~: treatment (Turbuhaler); r.::::;j : treatment (MDI); MDI: pressurized metered dose inhaler. more practical, whilst 1% preferred MDI in that respect. Only 2% found it difficult to inhale from any of the inhalers. Fifty percent of the patients in the Turbuhaler group preferred to use Turbuhaler and 24% of the patients were uncertain. Discussion The results of this study clearly show that Bricanyl Turbuhaler is as effective as Bricanyl MDI, during the whole Turbuhaler life-span i.e. the efficacy was not changed with time. Morning PEFR values before inhalation increased slightly during Turbuhaler treatment as compared with run-in. This was not seen in the MDI group. In earlier studies the corresponding PEFR values have been shown to be higher during Bricanyl Turbuhaler treatment as compared with Ventolin Rotahaler treatment [8, 9]. This could be due to a difference in duration of action of the two drugs [1, 11]. In this study, the higher morning PEFR values during Turbuhaler treatment could, at least in part, be attributable to a long duration of action of Bricanyl Turbuhaler. A possible explanation may be that
5 A SIX WEEK TREATMENT WITH BRICANYL TURBUHALER 179 Turbuhaler is easier to use [12] and therefore more drug can reach the airways. The reduction in asthma symptoms score and need for extra inhalations in the present study also indicate a favourable duration of action of Bricanyl Turbuhaler. However, since this was an open study it can not be excluded that the improvement in lung function as well as in asthma symptoms during Turbuhaler treatment in the present study, could be due to patient expectation of the new inhaler. Turbuhaler was well accepted by the patients, which is in agreement with a previous study [12]. Turbuhaler was preferred by twice as many patients as MDI in the present study. This is in accordance with the results from previous studies [13, 14]. In conclusion, this study showed that Bricanyl Turbuhaler is an effective and well accepted treatment in the long-term therapy of asthma. The preferences, asthma symptom scores and reduced need for extra inhalations all show a tendency in favour of Bricanyl Turbuhaler compared with Bricanyl MDI. References 1. Crompton GK. - Problems patients have using pressurized aerosol inhalers. Eur J Respir Dis, 1982, 63 (Suppl. 119), Yarbrough J, Mansfield LE, Ting S. - Metered dose inhaler induced bronchospasm in asthmatic patients. Ann Allergy, 1985, 55, Wetterlin KIL. - Turbuhaler: a new powder inhaler for administration of drugs to the airways. Pharm Res, 1988, 5, (8), Johnsen CR, Weeke ER. - Turbuhaler: a new device for dry powder terbutaline inhalation. Allergy, 1988, 43, Persson G, Gruvstad E, StAhl E.- A new multiple dose powder inhaler, (Turbuhaler ), compared with a pressurized inhaler in a study of Terbutaline in asthmatics. Eur Respir J, 1988, 1, Osterman K, Norborg A M, StAhl E.- A multiple dose powder inhaler (Turbuhaler ), compared with a conventional aerosol. An acceptance study in asthmatics. Allergy, 1989, 44, Andersen EB. - Discrete statistical models with social science application. North-Holland, Anani A, Higgins AJ, Crompton GK.- Breath-actuated inhalers: comparison of terbutaline Turbuhaler with salbutamol Rotahaler. Eur Respir J, 1989, 2, Stallaert RALM. - The Bricanyl Turbuhaler, efficacy and acceptability compared to the Ventolin Rotahaler. Abstract. ICACI XIII Congress, Montreux, October Schwarz F, Engler R, Villiger B. Turbuhaler. -A new concept in inhalation therapy. Abstract, Davos, April Simonsson BG et al. - Double-blind trial with increasing doses of salbutamol and terbutaline aerosols in patients with reversible airway obstruction. Acta Med Scand, 1972, 192, Crompton GK. - New inhalation devices. (Editorial). Eur Respir J, 1988, 1, Warner JO, Chetcuti P. - Efficacy and acceptability of terbutaline sulphate Turbuhaler in children. In: A New Concept in Inhalation Therapy. S.P. Newman et al. eds, Ribeiro LB. -A comparison of Bricanyl Turbuhaler and Berotec dry powder inhaler in asthmatic children. Eur Respir J, 1989, 2 (Suppl. 8), Abstract No. 99. Le Turbuhaler~ de Bricany~ dans le traitement de l'asthme. Une etude multicentrique de six semaines conduite en Suede, en Grande-Bretagne, au Danemark, en Norvege et en Finlande. K. Osterman, E. Stdhl, A. Kiillen. RESUME: Deux cent-cinquante-huit adultes atteints d'asthme bronchique ont participe tt une etude ouverte, en groupe parallele, d'une duree de huit semaines. L'etude a demarre par une periode de mise en route de deux semaines pendant laquelle la terbutaline (betal agoniste) a ete administree tt raison de 5mg quatre fois par jour par un aerosol pressurise au freon, le Bricanyl MDI. Pendant les six semaines suivantes, un tiers des patients a continue a employer le MDI, et les deux tiers ont re~u la meme dose de terbutaline sous forme de poudre seche par le Turbuhaler. L'on n'a pas note de difference statistiquement significative dans les augmentations du DEP avant et apres inhalation entre les deux groupes de traitement. Les scores des symptomes d'asthme ont diminue dans le groupe Turbuhaler pendant I' etude, m a is sont res test inchanges dans le groupe MDI. L'on n'a pas observe de difference d'utilisation des medications hors essai pendant le jour entre Ies deux groupes. Pendant la nuit la difference etait statistiquement significative en faveur du Turbuhaler (p<.5). Le Turbuhaler a ete bien supporte et s'est avere facile a utiliser. Compare au MD! utilise pendant la periode de mise en route, 5% des patients du groupe Turbuhaler lui ont donne la preference, 26% ont prefere le MD!, tandis que 24% n'avaient pas de preference. En conclusion, cette etude montre que le Turbuhaler est au moins aussi effectif que MD! pendant toute la periode de vie du Turbuhaler (6 semaines). Eur Respir J., 1991, 4,
Correspondence: C-G. Löfdahl Dept of Respiratory Medicine and Allergology University Hospital S Lund Sweden.
 Eur Respir J 1997; 10: 2474 2478 DOI: 10.1183/09031936.97.10112474 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 Differences in bronchodilating
Eur Respir J 1997; 10: 2474 2478 DOI: 10.1183/09031936.97.10112474 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 Differences in bronchodilating
Public Assessment Report for paediatric studies submitted in accordance with Article 45 of Regulation (EC) No1901/2006, as amended.
 Public Assessment Report for paediatric studies submitted in accordance with Article 45 of Regulation (EC) No1901/2006, as amended Bricanyl () DK/W/0017/pdWS/001 Rapporteur: Denmark Finalisation procedure
Public Assessment Report for paediatric studies submitted in accordance with Article 45 of Regulation (EC) No1901/2006, as amended Bricanyl () DK/W/0017/pdWS/001 Rapporteur: Denmark Finalisation procedure
G. Boyd on behalf of a UK Study group
 Eur Respir J, 1995, 8, 1494 1498 DOI: 10.1183/09031936.95.08091494 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1995 European Respiratory Journal ISSN 0903-1936 Salmeterol xinafoate in
Eur Respir J, 1995, 8, 1494 1498 DOI: 10.1183/09031936.95.08091494 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1995 European Respiratory Journal ISSN 0903-1936 Salmeterol xinafoate in
Is the correct use of a dry powder inhaler (Turbohaler) age dependent?
 Is the correct use of a dry powder inhaler (Turbohaler) age dependent? Kris De Boeck, MD, PhD, Marek Alifier, MD, and Gerd Warnier, RN Leuven, Belgium Background: The metered-dose inhalers are the most
Is the correct use of a dry powder inhaler (Turbohaler) age dependent? Kris De Boeck, MD, PhD, Marek Alifier, MD, and Gerd Warnier, RN Leuven, Belgium Background: The metered-dose inhalers are the most
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
L. Borgström*, E. Bondesson*, F. Morén**, E. Trofast*, S.P. Newman +
 Eur Respir J, 1994, 7, 69 73 DOI: 10.1183/09031936.94.07010069 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1994 European Respiratory Journal ISSN 0903-1936 Lung deposition of budesonide
Eur Respir J, 1994, 7, 69 73 DOI: 10.1183/09031936.94.07010069 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1994 European Respiratory Journal ISSN 0903-1936 Lung deposition of budesonide
Budesonide treatment of moderate and severe asthma in children: A doseresponse
 Budesonide treatment of moderate and severe asthma in children: A doseresponse study Soren Pedersen, MD, PhD, and Ove Ramsgaard Hansen, MD Kolding, Denmark Objective: The purpose of the study was to evaluate
Budesonide treatment of moderate and severe asthma in children: A doseresponse study Soren Pedersen, MD, PhD, and Ove Ramsgaard Hansen, MD Kolding, Denmark Objective: The purpose of the study was to evaluate
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
On completion of this chapter you should be able to: discuss the stepwise approach to the pharmacological management of asthma in children
 7 Asthma Asthma is a common disease in children and its incidence has been increasing in recent years. Between 10-15% of children have been diagnosed with asthma. It is therefore a condition that pharmacists
7 Asthma Asthma is a common disease in children and its incidence has been increasing in recent years. Between 10-15% of children have been diagnosed with asthma. It is therefore a condition that pharmacists
Terbutaline as powder inhalation from Bricanyl Turbuhaler compared to terbutaline as nebulizer solution in severe chronic airways obstruction
 Eur Respir J 1989, 2, 71&-720 Terbutaline as powder inhalation from Bricanyl Turbuhaler compared to terbutaline as nebulizer solution in severe chronic airways obstruction N.C.G. Hansen 1'erbutaline as
Eur Respir J 1989, 2, 71&-720 Terbutaline as powder inhalation from Bricanyl Turbuhaler compared to terbutaline as nebulizer solution in severe chronic airways obstruction N.C.G. Hansen 1'erbutaline as
International co-ordinating investigator Dr Dencho Osmanliev, St Sofia, Pulmonary Dept, 19 D Nestorov Str, Sofia, 1431, Bulgaria.
 Drug product: SYNOPSIS Drug substance(s): Budesonide/formoterol Document No.: SD-039-CR-0681 Referring to part Edition No.: 1 of the dossier Study code: SD-039-0681 Date: 24 Oktober, 2003 (For national
Drug product: SYNOPSIS Drug substance(s): Budesonide/formoterol Document No.: SD-039-CR-0681 Referring to part Edition No.: 1 of the dossier Study code: SD-039-0681 Date: 24 Oktober, 2003 (For national
Cost effectiveness of fluticasone and budesonide in patients with moderate asthma Steinmetz K O, Volmer T, Trautmann M, Kielhorn A
 Cost effectiveness of fluticasone and budesonide in patients with moderate asthma Steinmetz K O, Volmer T, Trautmann M, Kielhorn A Record Status This is a critical abstract of an economic evaluation that
Cost effectiveness of fluticasone and budesonide in patients with moderate asthma Steinmetz K O, Volmer T, Trautmann M, Kielhorn A Record Status This is a critical abstract of an economic evaluation that
Improved asthma control with budesonide/formoterol in a single inhaler, compared with budesonide alone
 Eur Respir J 2001; 18: 262 268 Printed in UK all rights reserved Copyright #ERS Journals Ltd 2001 European Respiratory Journal ISSN 0903-1936 Improved asthma control with budesonide/formoterol in a single
Eur Respir J 2001; 18: 262 268 Printed in UK all rights reserved Copyright #ERS Journals Ltd 2001 European Respiratory Journal ISSN 0903-1936 Improved asthma control with budesonide/formoterol in a single
Starting with a Higher Dose of Inhaled Corticosteroids in Primary Care Asthma Treatment
 Starting with a Higher Dose of Inhaled Corticosteroids in Primary Care Asthma Treatment THYS van der MOLEN, BETTY MEYBOOM-DE JONG, HELMA H. MULDER, and DIRKJE S. POSTMA Department of General Practice,
Starting with a Higher Dose of Inhaled Corticosteroids in Primary Care Asthma Treatment THYS van der MOLEN, BETTY MEYBOOM-DE JONG, HELMA H. MULDER, and DIRKJE S. POSTMA Department of General Practice,
Dual-Controller Asthma Therapy: Rationale and Clinical Benefits
 B/1 Dual-Controller Asthma Therapy: Rationale and Clinical Benefits MODULE B The 1997 National Heart, Lung, and Blood Institute (NHLBI) Expert Panel guidelines on asthma management recommend a 4-step approach
B/1 Dual-Controller Asthma Therapy: Rationale and Clinical Benefits MODULE B The 1997 National Heart, Lung, and Blood Institute (NHLBI) Expert Panel guidelines on asthma management recommend a 4-step approach
Meenu Singh, Joseph L. Mathew, Prabhjot Malhi, B.R. Srinivas and Lata Kumar
 Comparison of Improvement in Quality of Life Score with Objective Parameters of Pulmonary Function in Indian Asthmatic Children Receiving Inhaled Corticosteroid Therapy Meenu Singh, Joseph L. Mathew, Prabhjot
Comparison of Improvement in Quality of Life Score with Objective Parameters of Pulmonary Function in Indian Asthmatic Children Receiving Inhaled Corticosteroid Therapy Meenu Singh, Joseph L. Mathew, Prabhjot
Salmeterol, a new long acting inhaled,f2 adrenoceptor agonist: comparison with salbutamol in adult asthmatic patients
 Thorax 1988;43:674-678 Salmeterol, a new long acting inhaled,f2 adrenoceptor agonist: comparison with salbutamol in adult asthmatic patients ANDERS ULLMAN, NILS SVEDMYR From the Department of Clinical
Thorax 1988;43:674-678 Salmeterol, a new long acting inhaled,f2 adrenoceptor agonist: comparison with salbutamol in adult asthmatic patients ANDERS ULLMAN, NILS SVEDMYR From the Department of Clinical
Asthma training. Mike Levin Division of Asthma and Allergy Red Cross Hospital
 Asthma training Mike Levin Division of Asthma and Allergy Red Cross Hospital Introduction Physiology Diagnosis Severity Treatment Control Stage 3 of guidelines Acute asthma Drug delivery Conclusion Overview
Asthma training Mike Levin Division of Asthma and Allergy Red Cross Hospital Introduction Physiology Diagnosis Severity Treatment Control Stage 3 of guidelines Acute asthma Drug delivery Conclusion Overview
Inhalers containing CFCs. CFC-free inhalers
 Propellants used in medical metered dose inhalers and aerosol-based breath activated devices in New Zealand August to October 2002 (most recent period for which data is available) (February 2003) Inhalers
Propellants used in medical metered dose inhalers and aerosol-based breath activated devices in New Zealand August to October 2002 (most recent period for which data is available) (February 2003) Inhalers
Step-down approach in chronic stable asthma: A comparison of reducing dose Inhaled Formoterol/ Budesonide with maintaining Inhaled Budesonide.
 Step-down approach in chronic stable asthma: A comparison of reducing dose Inhaled Formoterol/ Budesonide with maintaining Inhaled Budesonide. By: DR MOHD SHAMSUL AMRI Supervisor: Associate Professor Dr
Step-down approach in chronic stable asthma: A comparison of reducing dose Inhaled Formoterol/ Budesonide with maintaining Inhaled Budesonide. By: DR MOHD SHAMSUL AMRI Supervisor: Associate Professor Dr
International Co-ordinating investigator None appointed.
 Drug product: Budesonide/formoterol SYNOPSIS Drug substance(s): Budesonide/formoterol Document No.: SD-039-CR-0673 Referring to part Edition No.: 1 of the dossier Study code: SD-039-0673 Date: 13 June,
Drug product: Budesonide/formoterol SYNOPSIS Drug substance(s): Budesonide/formoterol Document No.: SD-039-CR-0673 Referring to part Edition No.: 1 of the dossier Study code: SD-039-0673 Date: 13 June,
Full Length Original Research Paper
 Copyright 2013 By IYPF All rights reserved Open Access Contents Int. J. Drug Dev. & Res. October - December 2013 Vol. 5 Issue 4 ISSN 0975-9344 www.ijddr.in Effect of Budesonide by metered dose inhaler
Copyright 2013 By IYPF All rights reserved Open Access Contents Int. J. Drug Dev. & Res. October - December 2013 Vol. 5 Issue 4 ISSN 0975-9344 www.ijddr.in Effect of Budesonide by metered dose inhaler
Onset of bronchodilation and finger tremor induced by salmeterol and salbutamol in asthmatic patients
 ORIGINAL ARTICLE Onset of bronchodilation and finger tremor induced by salmeterol and salbutamol in asthmatic patients Jan Lötvall MD PhD, Helen Lunde MD, Nils Svedmyr MD PhD Department of Clinical Pharmacology,
ORIGINAL ARTICLE Onset of bronchodilation and finger tremor induced by salmeterol and salbutamol in asthmatic patients Jan Lötvall MD PhD, Helen Lunde MD, Nils Svedmyr MD PhD Department of Clinical Pharmacology,
beclometasone 100 MDI 2 puffs twice a day (recently changed to non CFC (Clenil Modulite))
 Case 1 Mr Thomson, a 32 year old asthmatic who is well known to you comes into your pharmacy. He is known to have a best peak flow of 640 L/min. He tells you that over the last few weeks he has been wakening
Case 1 Mr Thomson, a 32 year old asthmatic who is well known to you comes into your pharmacy. He is known to have a best peak flow of 640 L/min. He tells you that over the last few weeks he has been wakening
THE NEW ZEALAND MEDICAL JOURNAL
 THE NEW ZEALAND MEDICAL JOURNAL Vol 120 No 1267 ISSN 1175 8716 Is Salamol less effective than Ventolin? A randomised, blinded, crossover study in New Zealand Catherina L Chang, Manisha Cooray, Graham Mills,
THE NEW ZEALAND MEDICAL JOURNAL Vol 120 No 1267 ISSN 1175 8716 Is Salamol less effective than Ventolin? A randomised, blinded, crossover study in New Zealand Catherina L Chang, Manisha Cooray, Graham Mills,
BUDESONIDE AND FORMOTEROL (SYMBICORT ): Α A REVIEW
 Volume 23, Issue 3 December 2007 BUDESONIDE AND FORMOTEROL (SYMBICORT ): A REVIEW Donna L. Smith, Pharm. D. Candidate More than 22 million people in the United States have asthma according to the Centers
Volume 23, Issue 3 December 2007 BUDESONIDE AND FORMOTEROL (SYMBICORT ): A REVIEW Donna L. Smith, Pharm. D. Candidate More than 22 million people in the United States have asthma according to the Centers
Clinical effect of Diskus dry-powder inhaler at low and high inspiratory flow-rates in asthmatic children
 Eur Respir J 1998; 11: 35 354 DOI: 1.1183/931936.98.11235 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1998 European Respiratory Journal ISSN 93-1936 Clinical effect of Diskus dry-powder
Eur Respir J 1998; 11: 35 354 DOI: 1.1183/931936.98.11235 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1998 European Respiratory Journal ISSN 93-1936 Clinical effect of Diskus dry-powder
Asthma in Pregnancy. Asthma. Chronic Airway Inflammation. Objective Measures of Airflow. Peak exp. flow rate (PEFR)
 Chronic Airway Inflammation Asthma in Pregnancy Robin Field, MD Maternal Fetal Medicine Kaiser Permanente San Francisco Asthma Chronic airway inflammation increased airway responsiveness to a variety of
Chronic Airway Inflammation Asthma in Pregnancy Robin Field, MD Maternal Fetal Medicine Kaiser Permanente San Francisco Asthma Chronic airway inflammation increased airway responsiveness to a variety of
SYNOPSIS. Co-ordinating investigator Not applicable. Study centre(s) This study was conducted in Japan (57 centres).
 Drug product: Symbicort Turbuhaler Drug substance(s): ST (Symbicort Turbuhaler ) Edition No.: 1.0 Study code: D5890C00010 Date: 15 March 2007 SYNOPSIS An 8-week, randomised, double blind, parallel-group,
Drug product: Symbicort Turbuhaler Drug substance(s): ST (Symbicort Turbuhaler ) Edition No.: 1.0 Study code: D5890C00010 Date: 15 March 2007 SYNOPSIS An 8-week, randomised, double blind, parallel-group,
Asthma Medications: Information for Children and Families. What You Need to Know about Medicines for Asthma
 Page 1 of 8 PED-ALL-005-1992 Asthma Medications: Information for Children and Families What You Need to Know about Medicines for Asthma What Medicines Are used to Treat Asthma? There are two kinds of medicines:
Page 1 of 8 PED-ALL-005-1992 Asthma Medications: Information for Children and Families What You Need to Know about Medicines for Asthma What Medicines Are used to Treat Asthma? There are two kinds of medicines:
University of Groningen. Transdermal delivery of anticholinergic bronchodilators Bosman, Ingrid Jolanda
 University of Groningen Transdermal delivery of anticholinergic bronchodilators Bosman, Ingrid Jolanda IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
University of Groningen Transdermal delivery of anticholinergic bronchodilators Bosman, Ingrid Jolanda IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
Effect of 1 year daily treatment with 400 µg budesonide (Pulmicort Turbuhaler ) in newly diagnosed asthmatics
 Eur Respir J 1997; 10: 2210 2215 DOI: 10.1183/09031936.97.10102210 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 Effect of 1 year daily
Eur Respir J 1997; 10: 2210 2215 DOI: 10.1183/09031936.97.10102210 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 Effect of 1 year daily
SYNOPSIS THIS IS A PRINTED COPY OF AN ELECTRONIC DOCUMENT. PLEASE CHECK ITS VALIDITY BEFORE USE.
 Drug product: Drug substance(s): Document No.: Edition No.: 1 Study code: Accolate Zafirlukast (ZD9188) 9188IL/0138 Date: 02 May 2007 SYNOPSIS A Multicenter, Randomized, Double-blind, -controlled, Parallel
Drug product: Drug substance(s): Document No.: Edition No.: 1 Study code: Accolate Zafirlukast (ZD9188) 9188IL/0138 Date: 02 May 2007 SYNOPSIS A Multicenter, Randomized, Double-blind, -controlled, Parallel
Patient. Device Clinician. Safety & efficacy
 Patient Device Clinician Formulation Safety & efficacy 1. Modified from Daley-Yates et al., Expert Opin. Drug Deliv. 2011: 8(10):1297-1308 2. Modified from Laube et al., Eur Respir J 2011; 37: 1308 1331
Patient Device Clinician Formulation Safety & efficacy 1. Modified from Daley-Yates et al., Expert Opin. Drug Deliv. 2011: 8(10):1297-1308 2. Modified from Laube et al., Eur Respir J 2011; 37: 1308 1331
CLINICAL RELEVANCE OF IN VITRO PARTICLE SIZING DATA. Steve Newman, PhD Nottingham, UK November 2006
 IPAC-RS Conference 6-8 November 2006 CLINICAL RELEVANCE OF IN VITRO PARTICLE SIZING DATA Steve Newman, PhD Nottingham, UK November 2006 IPAC-RS Conference 6-8 November 2006 1 EFFECT OF PARTICLE SIZE ON
IPAC-RS Conference 6-8 November 2006 CLINICAL RELEVANCE OF IN VITRO PARTICLE SIZING DATA Steve Newman, PhD Nottingham, UK November 2006 IPAC-RS Conference 6-8 November 2006 1 EFFECT OF PARTICLE SIZE ON
measured Improvement in am PEFR FP vs BUD gave +11l/min FP vs BDP gave nonsignificant improvement in PEFR +3l/min FP: BUD ratio 1.
 1 of 8 09/05/2018, 11:29 Guideline topic: Pharmacological management of asthma Evidence table 4.25: Budesonide vs Beclomethasone Different inhaled corticosteroids (ICS) flixotide propionate (FP) vs budesonide
1 of 8 09/05/2018, 11:29 Guideline topic: Pharmacological management of asthma Evidence table 4.25: Budesonide vs Beclomethasone Different inhaled corticosteroids (ICS) flixotide propionate (FP) vs budesonide
A multitude of devices
 A multitude of devices Dr Andrew Scroop Respiratory Consultants 15 th September 2018 STEPWISE PHARMACOLOGICAL MANAGEMENT OF STABLE COPD COPD Inhalers MILD FEV 1 60 80% predicted few symptoms breathless
A multitude of devices Dr Andrew Scroop Respiratory Consultants 15 th September 2018 STEPWISE PHARMACOLOGICAL MANAGEMENT OF STABLE COPD COPD Inhalers MILD FEV 1 60 80% predicted few symptoms breathless
Respiratory Health. Asthma and COPD
 Respiratory Health Asthma and COPD Definition of asthma Working definition by AAH 2014: Chronic lung disease Can be controlled not cured Large variation in lung function Large variation in respiratory
Respiratory Health Asthma and COPD Definition of asthma Working definition by AAH 2014: Chronic lung disease Can be controlled not cured Large variation in lung function Large variation in respiratory
Ivax Pharmaceuticals UK Sponsor Submission to the National Institute for Health and Clinical Excellence
 Ivax Pharmaceuticals UK Sponsor Submission to the National Institute for Health and Clinical Excellence Clinical and cost-effectiveness of QVAR for the treatment of chronic asthma in adults and children
Ivax Pharmaceuticals UK Sponsor Submission to the National Institute for Health and Clinical Excellence Clinical and cost-effectiveness of QVAR for the treatment of chronic asthma in adults and children
Patient preference for a particular inhaler
 Eur Respir Rev 2005; 14: 96, 109 116 DOI: 10.1183/09059180.05.00009606 CopyrightßERSJ Ltd 2005 Patient preference for and satisfaction with inhaler devices P. Anderson ABSTRACT: Preference for and satisfaction
Eur Respir Rev 2005; 14: 96, 109 116 DOI: 10.1183/09059180.05.00009606 CopyrightßERSJ Ltd 2005 Patient preference for and satisfaction with inhaler devices P. Anderson ABSTRACT: Preference for and satisfaction
Tolerance to beta-agonists during acute bronchoconstriction
 Eur Respir J 1999; 14: 283±287 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 1999 European Respiratory Journal ISSN 0903-1936 Tolerance to beta-agonists during acute bronchoconstriction
Eur Respir J 1999; 14: 283±287 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 1999 European Respiratory Journal ISSN 0903-1936 Tolerance to beta-agonists during acute bronchoconstriction
C.M. Bosley*, D.T. Parry**, G.M. Cochrane***
 Eur Respir J, 1994, 7, 504 509 DOI: 10.1183/09031936.94.07030504 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1994 European Respiratory Journal ISSN 0903-1936 Patient compliance with
Eur Respir J, 1994, 7, 504 509 DOI: 10.1183/09031936.94.07030504 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1994 European Respiratory Journal ISSN 0903-1936 Patient compliance with
Potency ratio fluticasone propionate (Flixotide Diskus)/budesonide (Pulmicort Turbuhaler)
 Respiratory Medicine (2007) 101, 610 615 Potency ratio fluticasone propionate (Flixotide Diskus)/budesonide (Pulmicort Turbuhaler) Björn Ställberg a, Eva Pilman b, Bengt-Eric Skoogh c,, Bengt Arne Hermansson
Respiratory Medicine (2007) 101, 610 615 Potency ratio fluticasone propionate (Flixotide Diskus)/budesonide (Pulmicort Turbuhaler) Björn Ställberg a, Eva Pilman b, Bengt-Eric Skoogh c,, Bengt Arne Hermansson
SYNOPSIS. Study centres This was a multicentre study conducted in 53 centres in Sweden.
 Drug product: Symbicort Turbuhaler Drug substance(s): Budesonide/Formoterol Edition No.: 1 Study code: LD-039-0003 Date: 20 May 2005 SYNOPSIS An open, randomized, parallel group, multicentre, phase IIIB
Drug product: Symbicort Turbuhaler Drug substance(s): Budesonide/Formoterol Edition No.: 1 Study code: LD-039-0003 Date: 20 May 2005 SYNOPSIS An open, randomized, parallel group, multicentre, phase IIIB
Clinical efficacy of low-dose inhaled budesonide once or twice daily in children with mild asthma not previously treated with steroids
 Eur Respir J 1998; 12: 1099 1104 DOI: 10.1183/09031936.98.12051099 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1998 European Respiratory Journal ISSN 0903-1936 Clinical efficacy of low-dose
Eur Respir J 1998; 12: 1099 1104 DOI: 10.1183/09031936.98.12051099 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1998 European Respiratory Journal ISSN 0903-1936 Clinical efficacy of low-dose
Better Living with Obstructive Pulmonary Disease A Patient Guide
 Better Living with Obstructive Pulmonary Disease A Patient Guide Second Edition November 2012 Queensland Health a Better Living with Chronic Obstructive Pulmonary Disease A Patient Guide is a joint project
Better Living with Obstructive Pulmonary Disease A Patient Guide Second Edition November 2012 Queensland Health a Better Living with Chronic Obstructive Pulmonary Disease A Patient Guide is a joint project
SUMMARY THIS IS A PRINTED COPY OF AN ELECTRONIC DOCUMENT. PLEASE CHECK ITS VALIDITY BEFORE USE.
 i SUMMARY ZENECA PHARMACEUTICALS FINISHED PRODUCT: ACTIVE INGREDIENT: ACCOLATE zafirlukast (ZD9188) Trial title (number): A Dose-ranging, Safety and Efficacy Trial with Zafirlukast (ACCOLATE ) in the Treatment
i SUMMARY ZENECA PHARMACEUTICALS FINISHED PRODUCT: ACTIVE INGREDIENT: ACCOLATE zafirlukast (ZD9188) Trial title (number): A Dose-ranging, Safety and Efficacy Trial with Zafirlukast (ACCOLATE ) in the Treatment
Dose-response protective effect of salbutamol on methacholine airway responsiveness using pressurized metered dose inhalers and Turbuhalers
 ORIGINAL ARTICLE Dose-response protective effect of salbutamol on methacholine airway responsiveness using pressurized metered dose inhalers and Turbuhalers Albert G Wong MD, Paul M O Byrne MB, Christer
ORIGINAL ARTICLE Dose-response protective effect of salbutamol on methacholine airway responsiveness using pressurized metered dose inhalers and Turbuhalers Albert G Wong MD, Paul M O Byrne MB, Christer
Concurrent Use Of Metered Dose Inhalers Without Spacer And Dry Powder Inhalers By Asthmatic Children Adversely Affect Proper Inhalation Technique
 ISPUB.COM The Internet Journal of Pediatrics and Neonatology Volume 13 Number 1 Concurrent Use Of Metered Dose Inhalers Without Spacer And Dry Powder Inhalers By Asthmatic Children Adversely Affect Proper
ISPUB.COM The Internet Journal of Pediatrics and Neonatology Volume 13 Number 1 Concurrent Use Of Metered Dose Inhalers Without Spacer And Dry Powder Inhalers By Asthmatic Children Adversely Affect Proper
Inhalation Therapy. Inhalation Therapy
 Matching Device and Patient Matching Patient to the Device Søren Pedersen University of Southern Denmark Kolding Hospital Anatomical factors (age) Training and education Delivered dose Psycomotor skills
Matching Device and Patient Matching Patient to the Device Søren Pedersen University of Southern Denmark Kolding Hospital Anatomical factors (age) Training and education Delivered dose Psycomotor skills
Abstract Background Theophylline is widely used in the treatment of asthma, and there is evidence that theophylline has antiinflammatory
 Thorax 2000;55:837 841 837 National Heart and Lung Institute, Imperial College School of Medicine and Royal Brompton Hospital, London SW3 6LY, UK S Lim A Jatakanon K F Chung P J Barnes Napp Laboratories
Thorax 2000;55:837 841 837 National Heart and Lung Institute, Imperial College School of Medicine and Royal Brompton Hospital, London SW3 6LY, UK S Lim A Jatakanon K F Chung P J Barnes Napp Laboratories
Asthma. chapter 7. Overview
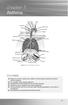 chapter 7 Asthma Sinus Sinus Sinus Right lung Adenoids Tonsils Pharynx Epiglottis Oesophagus Right bronchus Nasal cavity Oral cavity Tongue Larynx Trachea Ribs Left bronchus Diaphragm Bronchiole Pleura
chapter 7 Asthma Sinus Sinus Sinus Right lung Adenoids Tonsils Pharynx Epiglottis Oesophagus Right bronchus Nasal cavity Oral cavity Tongue Larynx Trachea Ribs Left bronchus Diaphragm Bronchiole Pleura
The clinical effectiveness and costeffectiveness. treatment of chronic asthma in children under the age of 12 years
 The clinical effectiveness and costeffectiveness of corticosteroids for the treatment of chronic asthma in children under the age of 12 years Submission of evidence from AstraZeneca UK Ltd regarding the
The clinical effectiveness and costeffectiveness of corticosteroids for the treatment of chronic asthma in children under the age of 12 years Submission of evidence from AstraZeneca UK Ltd regarding the
It is recommended that a mask and protective eyewear be worn when providing care to a patient with a cough
 UNIVERSITY HEALTH NETWORK POLICY #: PAGE 1 OF 7 POLICY AND PROCEDURE MANUAL: RESPIRATORY THERAPY DEPT PATIENT CARE SECTION ORIGINAL DATE: 04/03 ISSUED BY: SITE LEADER APPROVED BY: Infection Prevention
UNIVERSITY HEALTH NETWORK POLICY #: PAGE 1 OF 7 POLICY AND PROCEDURE MANUAL: RESPIRATORY THERAPY DEPT PATIENT CARE SECTION ORIGINAL DATE: 04/03 ISSUED BY: SITE LEADER APPROVED BY: Infection Prevention
ASTHMA POLICY KYABRAM P-12 COLLEGE
 Asthma Policy ASTHMA POLICY KYABRAM P-12 COLLEGE THE VICTORIAN SCHOOLS ASTHMA POLICY 1. ASTHMA AWARENESS As asthma can affect up to one in five children and one in ten adults, it is important for teachers
Asthma Policy ASTHMA POLICY KYABRAM P-12 COLLEGE THE VICTORIAN SCHOOLS ASTHMA POLICY 1. ASTHMA AWARENESS As asthma can affect up to one in five children and one in ten adults, it is important for teachers
Misuse of corticosteroid metered-dose inhaler is associated with decreased asthma stability
 Eur Respir J 2002; 19: 246 251 DOI: 10.1183/09031936.02.00218402 Printed in UK all rights reserved Copyright #ERS Journals Ltd 2002 European Respiratory Journal ISSN 0903-1936 Misuse of corticosteroid
Eur Respir J 2002; 19: 246 251 DOI: 10.1183/09031936.02.00218402 Printed in UK all rights reserved Copyright #ERS Journals Ltd 2002 European Respiratory Journal ISSN 0903-1936 Misuse of corticosteroid
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
A comparison of global questions versus health status questionnaires as measures of the severity and impact of asthma
 Eur Respir J 1999; 1: 591±596 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 1999 European Respiratory Journal ISSN 93-1936 A comparison of global questions versus health status questionnaires
Eur Respir J 1999; 1: 591±596 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 1999 European Respiratory Journal ISSN 93-1936 A comparison of global questions versus health status questionnaires
L. Thorsson, S. Edsbäcker, T-B. Conradson
 Eur Respir J, 1994, 7, 1839 1844 DOI: 10.1183/09031936.94.07101839 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1994 European Respiratory Journal ISSN 0903-1936 Lung deposition of budesonide
Eur Respir J, 1994, 7, 1839 1844 DOI: 10.1183/09031936.94.07101839 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1994 European Respiratory Journal ISSN 0903-1936 Lung deposition of budesonide
James P. Kemp, MD; Margaret C. Minkwitz, PhD; Catherine M. Bonuccelli, MD; and Marshelle S. Warren, MD
 Therapeutic Effect of Zafirlukast as Monotherapy in Steroid-Naive Patients With Severe Persistent Asthma* James P. Kemp, MD; Margaret C. Minkwitz, PhD; Catherine M. Bonuccelli, MD; and Marshelle S. Warren,
Therapeutic Effect of Zafirlukast as Monotherapy in Steroid-Naive Patients With Severe Persistent Asthma* James P. Kemp, MD; Margaret C. Minkwitz, PhD; Catherine M. Bonuccelli, MD; and Marshelle S. Warren,
T he conventional first line treatment of acute asthma for
 319 ORIGINAL ARTICLE Comparative efficacy of terbutaline sulphate delivered by Turbuhaler dry powder inhaler or pressurised metered dose inhaler with Nebuhaler spacer in children during an acute asthmatic
319 ORIGINAL ARTICLE Comparative efficacy of terbutaline sulphate delivered by Turbuhaler dry powder inhaler or pressurised metered dose inhaler with Nebuhaler spacer in children during an acute asthmatic
The Novolizer s : overcoming inherent problems of dry powder inhalers
 Respiratory Medicine (2004) Supplement A, S17 S21 ARTICLE IN PRESS The Novolizer s : overcoming inherent problems of dry powder inhalers Dieter Kohler* Chefarzt Innere Medizin und Pneumologie, Fachkrankenhaus
Respiratory Medicine (2004) Supplement A, S17 S21 ARTICLE IN PRESS The Novolizer s : overcoming inherent problems of dry powder inhalers Dieter Kohler* Chefarzt Innere Medizin und Pneumologie, Fachkrankenhaus
Users of antiasthma drugs in Iceland: a drug utilization study
 Eur Respir J 1997; 10: 1230 1234 DOI: 10.1183/09031936.97.10061230 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 Users of antiasthma drugs
Eur Respir J 1997; 10: 1230 1234 DOI: 10.1183/09031936.97.10061230 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 Users of antiasthma drugs
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Time course of action of two inhaled corticosteroids, fluticasone propionate and budesonide
 ASTHMA Time course of action of two inhaled corticosteroids, fluticasone propionate and budesonide K Phillips, J Oborne, S Lewis, T W Harrison, A E Tattersfield... See end of article for authors affiliations...
ASTHMA Time course of action of two inhaled corticosteroids, fluticasone propionate and budesonide K Phillips, J Oborne, S Lewis, T W Harrison, A E Tattersfield... See end of article for authors affiliations...
DATE: 09 December 2009 CONTEXT AND POLICY ISSUES:
 TITLE: Tiotropium Compared with Ipratropium for Patients with Moderate to Severe Chronic Obstructive Pulmonary Disease: A Review of the Clinical Effectiveness DATE: 09 December 2009 CONTEXT AND POLICY
TITLE: Tiotropium Compared with Ipratropium for Patients with Moderate to Severe Chronic Obstructive Pulmonary Disease: A Review of the Clinical Effectiveness DATE: 09 December 2009 CONTEXT AND POLICY
Diagnosis, Treatment and Management of Asthma
 Diagnosis, Treatment and Management of Asthma Asthma is a complex disorder characterized by variable and recurring symptoms, airflow obstruction, bronchial hyperresponsiveness, and an underlying inflammation.
Diagnosis, Treatment and Management of Asthma Asthma is a complex disorder characterized by variable and recurring symptoms, airflow obstruction, bronchial hyperresponsiveness, and an underlying inflammation.
Decramer 2014 a &b [21]
![Decramer 2014 a &b [21] Decramer 2014 a &b [21]](/thumbs/90/101390504.jpg) Buhl 2015 [19] Celli 2014 [20] Decramer 2014 a &b [21] D Urzo 2014 [22] Maleki-Yazdi 2014 [23] Inclusion criteria: Diagnosis of chronic obstructive pulmonary disease; 40 years of age or older; Relatively
Buhl 2015 [19] Celli 2014 [20] Decramer 2014 a &b [21] D Urzo 2014 [22] Maleki-Yazdi 2014 [23] Inclusion criteria: Diagnosis of chronic obstructive pulmonary disease; 40 years of age or older; Relatively
My Asthma Log. Tommy Traffic Light. NHS Number: Name:
 My Asthma Log Tommy Traffic Light NHS Number: Name: My Asthma Log Book This book is designed to help you understand and learn about managing your asthma. You should take your book with you to all your
My Asthma Log Tommy Traffic Light NHS Number: Name: My Asthma Log Book This book is designed to help you understand and learn about managing your asthma. You should take your book with you to all your
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A.
 aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
10/18/2012. Penn State University Children s Hospital JODIE STABINSKI CRNP MSN AE-C
 Penn State University Children s Hospital JODIE STABINSKI CRNP MSN AE-C Daily: Long-Term Control Corticosteroids (inhaled and systemic) Long-acting beta 2 -agonists (Serevent, Foradil) Methylxanthines
Penn State University Children s Hospital JODIE STABINSKI CRNP MSN AE-C Daily: Long-Term Control Corticosteroids (inhaled and systemic) Long-acting beta 2 -agonists (Serevent, Foradil) Methylxanthines
Treatment of acute asthmatic exacerbations with an increased dose of inhaled steroid
 12 Paediatrics and Child Health, Dunedin School of Medicine, PO Box 913, University of Otago Medical School, Dunedin, New Zealand J Garrett Preventive and Social Medicine, Dunedin School of Medicine S
12 Paediatrics and Child Health, Dunedin School of Medicine, PO Box 913, University of Otago Medical School, Dunedin, New Zealand J Garrett Preventive and Social Medicine, Dunedin School of Medicine S
DR REBECCA THOMAS CONSULTANT RESPIRATORY PHYSICIAN YORK DISTRICT HOSPITAL
 DR REBECCA THOMAS CONSULTANT RESPIRATORY PHYSICIAN YORK DISTRICT HOSPITAL Definition Guidelines contact complicated definitions Central to this is Presence of symptoms Variable airflow obstruction Diagnosis
DR REBECCA THOMAS CONSULTANT RESPIRATORY PHYSICIAN YORK DISTRICT HOSPITAL Definition Guidelines contact complicated definitions Central to this is Presence of symptoms Variable airflow obstruction Diagnosis
Clinical efficacy of montelukast in anti-inflammatory treatment of asthma and allergic rhinitis
 Clinical efficacy of montelukast in anti-inflammatory treatment of asthma and allergic rhinitis Kim Hyun Hee, MD, PhD. Dept. of Pediatrics The Catholic University of Korea College of Medicine Achieving
Clinical efficacy of montelukast in anti-inflammatory treatment of asthma and allergic rhinitis Kim Hyun Hee, MD, PhD. Dept. of Pediatrics The Catholic University of Korea College of Medicine Achieving
Efficacy and cost benefit of inhaled corticosteroids in patients considered to have mild asthma in primary care practice
 ORIGINAL ARTICLE Efficacy and cost benefit of inhaled corticosteroids in patients considered to have mild asthma in primary care practice PAUL O BYRNE MB, LAUREN CUDDY MA*,D WAYNE TAYLOR MA, STEPHEN BIRCH
ORIGINAL ARTICLE Efficacy and cost benefit of inhaled corticosteroids in patients considered to have mild asthma in primary care practice PAUL O BYRNE MB, LAUREN CUDDY MA*,D WAYNE TAYLOR MA, STEPHEN BIRCH
Your Inhaler Devices & You
 1 Your Inhaler Devices & You COUNSEL ON THE APPROPRIATE USE OF A: METERED DOSE INHALER (MDI) DRY POWDER INHALER (DPI) DISCUSS THE APPROPRIATE USAGE OF A PEAK FLOW METER AND SPACER/HOLDING CHAMBER DEVICE
1 Your Inhaler Devices & You COUNSEL ON THE APPROPRIATE USE OF A: METERED DOSE INHALER (MDI) DRY POWDER INHALER (DPI) DISCUSS THE APPROPRIATE USAGE OF A PEAK FLOW METER AND SPACER/HOLDING CHAMBER DEVICE
Inhalation devices, proper technique and cleaning
 Preventing Your Symptoms and Taking Your Medications Inhalation devices, proper technique and cleaning Knowing how to use your medications properly is important because inhaled drugs are meant to get directly
Preventing Your Symptoms and Taking Your Medications Inhalation devices, proper technique and cleaning Knowing how to use your medications properly is important because inhaled drugs are meant to get directly
Zopiclone Versus Nitrazepam: A Double-Blind Comparative Study of Efficacy and Tolerance in Elderly Patients with Chronic Insomnia
 Sleep lo(suppl. 1):73-78, Raven Press, Ltd., New York 1987 Association of Professional Sleep Societies Zopiclone Versus Nitrazepam: A Double-Blind Comparative Study of Efficacy and Tolerance in Elderly
Sleep lo(suppl. 1):73-78, Raven Press, Ltd., New York 1987 Association of Professional Sleep Societies Zopiclone Versus Nitrazepam: A Double-Blind Comparative Study of Efficacy and Tolerance in Elderly
Asthma Management for the Athlete
 Asthma Management for the Athlete Khanh Lai, MD Assistant Professor Division of Pediatric Pulmonary and Sleep Medicine University of Utah School of Medicine 2 nd Annual Sports Medicine Symposium: The Pediatric
Asthma Management for the Athlete Khanh Lai, MD Assistant Professor Division of Pediatric Pulmonary and Sleep Medicine University of Utah School of Medicine 2 nd Annual Sports Medicine Symposium: The Pediatric
Efficacy of Nebulised Ipratropium in Acute Bronchial Asthma
 ORIGINAL ARTICLE JIACM 2002; 3(4): 353-59 Efficacy of Nebulised Ipratropium in Acute Bronchial Asthma Praveen Aggarwal*, Onkar Singh**, Jyoti P Wali***, Rohini Handa*, Sada N Dwivedi****, Ashutosh Biswas*****,
ORIGINAL ARTICLE JIACM 2002; 3(4): 353-59 Efficacy of Nebulised Ipratropium in Acute Bronchial Asthma Praveen Aggarwal*, Onkar Singh**, Jyoti P Wali***, Rohini Handa*, Sada N Dwivedi****, Ashutosh Biswas*****,
Acceptance of the Accuhaler, a Multidose Powder Inhaler, among Asthmatic
 ASAN PACFC JOURNAL OF ALLERGY AND MMUNOLOGY (2000) 18: 135-140 i Acceptance of the Accuhaler, a Multidose Powder nhaler, among Asthmatic t 1 ~ Patients: A Comparison with the Pres surized Metered-Dose
ASAN PACFC JOURNAL OF ALLERGY AND MMUNOLOGY (2000) 18: 135-140 i Acceptance of the Accuhaler, a Multidose Powder nhaler, among Asthmatic t 1 ~ Patients: A Comparison with the Pres surized Metered-Dose
Asthma quality of life during 1 year of treatment with budesonide with or without formoterol
 Eur Respir J 1999; 14: 1038±1043 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 1999 European Respiratory Journal ISSN 0903-1936 Asthma quality of life during 1 year of treatment with
Eur Respir J 1999; 14: 1038±1043 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 1999 European Respiratory Journal ISSN 0903-1936 Asthma quality of life during 1 year of treatment with
Supplementary Medications during asthma attack. Prof. Dr Finn Rasmussen PhD. DrMedSc. Near East University Hospital North Cyprus
 Supplementary Medications during asthma attack Prof. Dr Finn Rasmussen PhD. DrMedSc. Near East University Hospital North Cyprus Conflicts of Interest None Definition of Asthma Airway narrowing that is
Supplementary Medications during asthma attack Prof. Dr Finn Rasmussen PhD. DrMedSc. Near East University Hospital North Cyprus Conflicts of Interest None Definition of Asthma Airway narrowing that is
AIRNERGY REPORT. May Dr Nyjon Eccles BSc MBBS PhD MRCP The Chiron Clinic, Harley St, London, England W1G 6AX Tel:
 AIRNERGY REPORT May 2004 Dr Nyjon Eccles BSc MBBS PhD MRCP The Chiron Clinic, Harley St, London, England W1G 6AX Tel: 0207 2244622 Abstract Six volunteers were recruited for a study of the effect of 4
AIRNERGY REPORT May 2004 Dr Nyjon Eccles BSc MBBS PhD MRCP The Chiron Clinic, Harley St, London, England W1G 6AX Tel: 0207 2244622 Abstract Six volunteers were recruited for a study of the effect of 4
Airway deposition and airway effects of antiasthma drugs delivered from metered-dose inhalers
 Eur Respir J 1997; 10: 2127 2138 DOI: 10.1183/09031936.97.10092127 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 REVIEW Airway deposition
Eur Respir J 1997; 10: 2127 2138 DOI: 10.1183/09031936.97.10092127 Printed in UK - all rights reserved Copyright ERS Journals Ltd 1997 European Respiratory Journal ISSN 0903-1936 REVIEW Airway deposition
Package leaflet: Information for the user. Bricanyl Turbohaler 0.5 mg/dose terbutaline sulfate
 Package leaflet: Information for the user Bricanyl Turbohaler 0.5 mg/dose terbutaline sulfate Read all of this leaflet carefully before you start using this medicine because it contains important information
Package leaflet: Information for the user Bricanyl Turbohaler 0.5 mg/dose terbutaline sulfate Read all of this leaflet carefully before you start using this medicine because it contains important information
ADULT ASTHMA GUIDE SUMMARY. This summary provides busy health professionals with key guidance for assessing and treating adult asthma.
 ADULT ASTHMA GUIDE SUMMARY This summary provides busy health professionals with key guidance for assessing and treating adult asthma. Its source document Asthma and Respiratory Foundation NZ Adult Asthma
ADULT ASTHMA GUIDE SUMMARY This summary provides busy health professionals with key guidance for assessing and treating adult asthma. Its source document Asthma and Respiratory Foundation NZ Adult Asthma
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Papi A, Canonica GW, Maestrelli P, et al. Rescue use of beclomethasone
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Papi A, Canonica GW, Maestrelli P, et al. Rescue use of beclomethasone
History & Development
 RSPT 2317 Anticholinergic Bronchodilators () History & Development Prototypical parasympatholytic agent is atropine an alkaloid found naturally in the plants Atropa belladona (nightshade) and Datura species
RSPT 2317 Anticholinergic Bronchodilators () History & Development Prototypical parasympatholytic agent is atropine an alkaloid found naturally in the plants Atropa belladona (nightshade) and Datura species
Formoterol (OXIS s ) Turbuhaler s as a rescue therapy compared with salbutamol pmdi plus spacer in patients with acute severe asthma
 Respiratory Medicine (2003) 97, 1067 1074 Formoterol (OXIS s ) Turbuhaler s as a rescue therapy compared with salbutamol pmdi plus spacer in patients with acute severe asthma W. Boonsawat*, S. Charoenratanakul,
Respiratory Medicine (2003) 97, 1067 1074 Formoterol (OXIS s ) Turbuhaler s as a rescue therapy compared with salbutamol pmdi plus spacer in patients with acute severe asthma W. Boonsawat*, S. Charoenratanakul,
Chronic obstructive pulmonary disease (COPD) is characterized
 DANIEL E. HILLEMAN, PharmD ABSTRACT OBJECTIVE: To review the role of long-acting bronchodilators in the treatment of chronic obstructive pulmonary disease (COPD), including the importance of treatment
DANIEL E. HILLEMAN, PharmD ABSTRACT OBJECTIVE: To review the role of long-acting bronchodilators in the treatment of chronic obstructive pulmonary disease (COPD), including the importance of treatment
Learning the Asthma Guidelines by Case Studies
 Learning the Asthma Guidelines by Case Studies Timothy Craig, DO Professor of Medicine and Pediatrics Distinguished Educator Penn State University Hershey Medical Center Objectives 1. Learn the Asthma
Learning the Asthma Guidelines by Case Studies Timothy Craig, DO Professor of Medicine and Pediatrics Distinguished Educator Penn State University Hershey Medical Center Objectives 1. Learn the Asthma
LONGITUDINAL STUDY OF LUNG FUNCTION DEVELOPMENT IN A COHORT OF INDIAN MEDICAL STUDENTS: INTERACTION OF RESPIRATORY ALLERGY AND SMOKING
 Indian J Physiol Pharmacol 1991; 35(1): 44-48 LONTUDINAL STUDY OF LUNG FUNCTION DEVELOPMENT IN A COHORT OF INDIAN MEDICAL STUDENTS: INTERACTION OF RESPIRATORY ALLERGY AND SMOKING S. WALTER* AND J. RICHARD**
Indian J Physiol Pharmacol 1991; 35(1): 44-48 LONTUDINAL STUDY OF LUNG FUNCTION DEVELOPMENT IN A COHORT OF INDIAN MEDICAL STUDENTS: INTERACTION OF RESPIRATORY ALLERGY AND SMOKING S. WALTER* AND J. RICHARD**
T he use of inhaled corticosteroids to control the inflammatory
 791 ORIGINAL ARTICLE Early asthma control and maintenance with eformoterol following reduction of inhaled corticosteroid dose D Price, D Dutchman, A Mawson, B Bodalia, S Duggan, P Todd on behalf of the
791 ORIGINAL ARTICLE Early asthma control and maintenance with eformoterol following reduction of inhaled corticosteroid dose D Price, D Dutchman, A Mawson, B Bodalia, S Duggan, P Todd on behalf of the
Individual Study Table Referring to Part of the Dossier. Volume:
 Final Report M/100977/21Final Version () 2. SYNOPSIS A Title of Study: A PHASE IIa, RANDOMISED, DOUBLE-BLIND, MULTIPLE DOSE, PLACEBO CONTROLLED, 3 PERIOD CROSS-OVER, ASCENDING DOSE CLINICAL TRIAL TO ASSESS
Final Report M/100977/21Final Version () 2. SYNOPSIS A Title of Study: A PHASE IIa, RANDOMISED, DOUBLE-BLIND, MULTIPLE DOSE, PLACEBO CONTROLLED, 3 PERIOD CROSS-OVER, ASCENDING DOSE CLINICAL TRIAL TO ASSESS
Long-term treatment of chronic obstructive pulmonary disease with salmeterol and the additive effect of ipratropium
 Eur Respir J 2; 15: 878±885 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 2 European Respiratory Journal ISSN 93-1936 Long-term treatment of chronic obstructive pulmonary disease with
Eur Respir J 2; 15: 878±885 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 2 European Respiratory Journal ISSN 93-1936 Long-term treatment of chronic obstructive pulmonary disease with
Guide to Inhaled Treatment Choices
 Guide to Inhaled Treatment Choices Note: this is guidance only, it is important to consider which device is best suited to the patient. This may NOT be the first line choice (but should be on the joint
Guide to Inhaled Treatment Choices Note: this is guidance only, it is important to consider which device is best suited to the patient. This may NOT be the first line choice (but should be on the joint
Asthma: Chronic Management. Yung-Yang Liu, MD Attending physician, Chest Department Taipei Veterans General Hospital April 26, 2015
 Asthma: Chronic Management Yung-Yang Liu, MD Attending physician, Chest Department Taipei Veterans General Hospital April 26, 2015 Global Strategy for Asthma Management and Prevention Evidence-based Implementation
Asthma: Chronic Management Yung-Yang Liu, MD Attending physician, Chest Department Taipei Veterans General Hospital April 26, 2015 Global Strategy for Asthma Management and Prevention Evidence-based Implementation
Scottish Medicines Consortium
 Scottish Medicines Consortium budesonide/formoterol 100/6, 200/6 turbohaler (Symbicort SMART ) No. (362/07) Astra Zeneca UK Limited 9 March 2007 (Issued May 2007) The Scottish Medicines Consortium (SMC)
Scottish Medicines Consortium budesonide/formoterol 100/6, 200/6 turbohaler (Symbicort SMART ) No. (362/07) Astra Zeneca UK Limited 9 March 2007 (Issued May 2007) The Scottish Medicines Consortium (SMC)
Guide to Inhaled Treatment Choices
 Guide to Inhaled Treatment Choices Note: this is guidance only, it is important to consider which device is best suited to the patient. This may NOT be the first line choice (but should be on the joint
Guide to Inhaled Treatment Choices Note: this is guidance only, it is important to consider which device is best suited to the patient. This may NOT be the first line choice (but should be on the joint
