2017 URAC PHARMACY BENEFIT MANAGEMENT PERFORMANCE MEASUREMENT: AGGREGATE SUMMARY PERFORMANCE REPORT
|
|
|
- Justina Butler
- 5 years ago
- Views:
Transcription
1 2017 URAC PHARMACY BENEFIT MANAGEMENT PERFORMANCE MEASUREMENT: AGGREGATE SUMMARY PERFORMANCE REPORT December 2017
2 Table of Contents Executive Summary... 1 Pharmacy Benefit Management Organization Characteristics... 1 Data Validation Overview... 2 Results: Pharmacy Benefit Management Measures... 3 Measure 1 Proportion of Days Covered (DM )... 3 Measure 2 Adherence to Non-Warfarin Oral Anticoagulants (DTM )...15 Measure 3 Generic Dispensing Rates (MP )...16 Measure 4 Drug-Drug Interactions (DM )...17 Measure 5 Use of High-Risk Medications in the Elderly (HRM) (HIM )...18 Measure 6 Call Center Performance (DTM )...19 Measure 7 Turnaround Time for Prescriptions (MP )...20 Measure 8 Antipsychotic Use in Children Under 5 Years Old (PH )...21 Measure 9 Use of Benzodiazepine Sedative Hypnotic Medications in the Elderly (PH )...22 Measure 10 Diabetes Medication Dosing (PH )...23 Measure 11 Statin Use in Persons with Diabetes (PH )...24 Measure 12 Consumer Experience with Pharmacy Services (PH )...25 Concluding Remarks...26 Table of Exhibits Exhibit 1: Total Number of Prescription for Pharmacy Benefit Management Program by Organization... 2 Exhibit 2: Proportion of Days Covered (80% Threshold) Commercial... 3 Exhibit 3: Proportion of Days Covered (80% Threshold) Medicaid... 4 Exhibit 4: Proportion of Days Covered (80% Threshold) Medicare... 4 Exhibit 5: Proportion of Days Covered (80% Threshold) Rate 1: Beta Blocker Medications... 5 Exhibit 6: Proportion of Days Covered (80% Threshold) Rate 1: Beta Blocker Medications (Summary Data)... 5 Exhibit 7: Proportion of Days Covered (80% Threshold) Rate 2: Renin Angiotensin System Antagonists... 6 Exhibit 8: Proportion of Days Covered (80% Threshold) Rate 2: Renin Angiotensin System Antagonists (Summary Data)... 6 Exhibit 9: Proportion of Days Covered (80% Threshold) Rate 3: Calcium Channel Blocker Medications... 7 Exhibit 10: Proportion of Days Covered (80% Threshold) Rate 3: Calcium Channel Blocker
3 Medications (Summary Data)... 7 Exhibit 11: Proportion of Days Covered (80% Threshold) Rate 4: Biguanides... 8 Exhibit 12: Proportion of Days Covered (80% Threshold) Rate 4: Biguanides... 8 Exhibit 13: Proportion of Days Covered (80% Threshold) Rate 5: Sulfonylureas... 9 Exhibit 14: Proportion of Days Covered (80% Threshold) Rate 5: Sulfonylureas (Summary Data)... 9 Exhibit 15: Proportion of Days Covered (80% Threshold) Rate 6: Thiazolidinediones...10 Exhibit 16: Proportion of Days Covered (80% Threshold) Rate 6: Thiazolidinediones (Summary Data)...10 Exhibit 17: Proportion of Days Covered (80% Threshold) Rate 7: DPP-IV Inhibitors...11 Exhibit 18: Proportion of Days Covered (80% Threshold) Rate 7: DPP-IV Inhibitors (Summary Data)...11 Exhibit 19: Proportion of Days Covered (80% Threshold) Rate 8: Diabetes All Class Rate...12 Exhibit 20: Proportion of Days Covered (80% Threshold) Rate 8: Diabetes All Class Rate (Summary Data)...12 Exhibit 21: Proportion of Days Covered (80% Threshold) Rate 9: Statins...13 Exhibit 22: Proportion of Days Covered (80% Threshold) Rate 9: Statins (Summary Data)...13 Exhibit 23: Proportion of Days Covered (80% Threshold) Rate 10: Anti-Retroviral Medications...14 Exhibit 24: Proportion of Days Covered (80% Threshold) Rate 10: Anti-Retroviral Medications (Summary Data)...14 Exhibit 25: Adherence to Non-Warfarin Oral Anticoagulants...15 Exhibit 26: Adherence to Non-Warfarin Oral Anticoagulants (Summary Data)...15 Exhibit 27: Generic Dispensing Rates...16 Exhibit 28: Generic Dispensing Rates (Summary Data)...16 Exhibit 29: Drug-Drug Interactions...17 Exhibit 30: Drug-Drug Interactions (Summary Data)...17 Exhibit 31: Use of High-Risk Medications in the Elderly...18 Exhibit 32: Use of High-Risk Medications in the Elderly (Summary Data)...18 Exhibit 33: Call Center Performance...19 Exhibit 34: Call Center Performance (Summary Data)...19
4 Executive Summary Presented in this report are the 2016 measurement year (2017 reporting year) results based on URAC s Pharmacy Benefit Management (PBM) Accreditation program performance measures. The report includes only aggregate summary rates; there are no individual performance results included. Organizations were required to report data for six mandatory measures, and they had the option to report data for six exploratory measures. Below is the list of mandatory [M] and exploratory [E] measures for 2017 reporting: 1. Proportion of Days Covered (DM ) [M] 2. Adherence to Non-Warfarin Oral Anticoagulants (DTM ) [M] 3. Generic Dispensing Rates (MP ) [M] 4. Drug-Drug Interactions (DM ) [M] 5. Use of High-Risk Medications in the Elderly (HRM) (HIM ) [M] 6. Call Center Performance (DTM ) [M] 7. Turnaround Time for Prescriptions (MP ) [E] 8. Antipsychotic Use in Children Under 5 Years Old (PH ) [E] 9. Use of Benzodiazepine Sedative Hypnotic Medications in the Elderly (PH [E] 10. Diabetes Medication Dosing (PH ) [E] 11. Statin Use in Persons with Diabetes (PH ) [E] 12. Consumer Experience with Pharmacy Services (PH ) [E] For PBM, performance measurement for the 2017 reporting year aligns with Phase 2 of URAC s measurement process. With Phase 2, mandatory performance measures are subject to an external auditing and verification process. Additionally, the audited performance measure results become publicly available via aggregated, de-identified reports. With Phase 3, organization-specific measure results that have undergone an external auditing and verification process will be publicly available on the URAC website. Data Analysis Procedures and Future Considerations In 2018, Kiser Healthcare Solutions implemented a relational database management system, Microsoft SQL Server (MSSQL), to capture and normalize all accreditation submission data into a consistent format across programs. This improvement allows for a consistent model to be used year over year and allows for trends to build. In addition, MSSQL aids in consolidating all data objects used for aggregations, guaranteeing consistent logic across programs and ease of updates. Finally, Kiser Healthcare Solutions implemented Microsoft Power BI as the business intelligence tool to develop the data visuals and tables in the report. Through manual data review and cleaning, data entry errors were corrected by Kiser Healthcare Solutions and noted in the data files and at the end of this report. Respondent organizations will be notified in the individual reports where data entry corrections were made and where the data validation vendors indicated materially inaccurate results. Pharmacy Benefit Management Organization Characteristics Five URAC-accredited Pharmacy Benefit Management (PBM) organizations reported 2016 measurement year data for the 2017 reporting year. There were initially six organizations survey submissions, but one was determined to be a duplicate entry. The five organizations reported 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 1
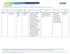
5 commercial data for at least one measure; two organizations also reported Medicaid data; and one organization reported Medicare data in addition to the commercial and Medicaid data. The PBMs represented national geographical coverage across all four URAC-specified regions (Midwest, Northeast, South, and West). The total number of prescriptions covered by the PBMs ranged from 5,853,937 to 23,325,461, with a median of 11,824,390, a mean of 12,117,045, and grand total of 60,585,227 prescriptions across all organizations reporting (Exhibit 1). Exhibit 1: Total Number of Prescription for Pharmacy Benefit Management Program by Organization Data Validation Overview For 2017 reporting, URAC required that organizations have their measure results reviewed by a URACapproved data validation vendor (DVV). There were four vendors that participated: Advent Advisory Group, Attest Health Care Advisors, Healthcare Data Company, and Metastar. This represents an increase in vendors compared to 2016 where only Attest Health Care participated for URAC s first year requirement of data validation URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 2
6 Results: Pharmacy Benefit Management Measures All five PBM organizations reported data for at least one of the performance measures. The organizations reported primarily commercial results. Only one organization reported separate results for all three service lines and one reported commercial and Medicaid results. The analysis results are included for each measure, but due to the limited number of data submissions for Medicaid and Medicare, aggregate data summaries are limited, and benchmarks are not provided (e.g., percentiles and identification of outliers). Measure 1 Proportion of Days Covered (DM ) Measure Description This mandatory measure assesses the percentage of participants 18 years and older who met the proportion of days covered (PDC) threshold of 80% during the measurement period. A performance rate is calculated separately for the following medication categories: Beta-blockers(BB); Renin Angiotensin System (RAS) Antagonists; Calcium Channel Blockers (CCB); Statins; Biguanides; Sulfonyluraes; Thiazolidinediones; Dipeptidyl Peptidase (DPP)-IV Inhibitors; Diabetes All Class; Antiretrovirals (this measure has a threshold of 90% for at least two measures). The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc Exhibit 2: Proportion of Days Covered (80% Threshold) Commercial 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 3
7 Exhibit 3: Proportion of Days Covered (80% Threshold) Medicaid Exhibit 4: Proportion of Days Covered (80% Threshold) Medicare 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 4
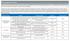
8 Summary of Findings Given only five organizations submitted data for commercial, with two of those also reporting for Medicaid, and one that also reported for Medicare, the measure summaries are limited to basic aggregate statistics. The minimum, maximum, median, and mean were used for benchmarking individual performance with commercial, given the small sample size for commercial. Medicaid and Medicare benchmarks are calculated, but do not provide useful comparative benchmarks. In some cases, commercial analytics were restricted to a limited set of comparisons given the small number of data submissions. Rate 1: Beta Blocker (BB) Medications All five organizations submitted data for commercial, two organizations submitted for Medicaid, and one submitted for Medicare. The aggregate summary rate for commercial is 67.71% with the mean of 65.10% and median of 67.59% across the range of 50.12% to 71.74%. The aggregate summary rate for Medicaid is 55.22% and the single reported Medicare rate is 67.84%. Exhibit 5: Proportion of Days Covered (80% Threshold) Rate 1: Beta Blocker Medications Exhibit 6: Proportion of Days Covered (80% Threshold) Rate 1: Beta Blocker Medications (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 5
9 Rate 2: Renin Angiotensin System (RAS) Antagonists Four organizations submitted data for commercial, one organization submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 68.69% with the mean of 65.92% and median of 68.96% across the range of 52.79% to 72.97%. The single reported Medicaid rate is 56.08% and the single reported Medicare rate is 73.15%. Exhibit 7: Proportion of Days Covered (80% Threshold) Rate 2: Renin Angiotensin System Antagonists Exhibit 8: Proportion of Days Covered (80% Threshold) Rate 2: Renin Angiotensin System Antagonists (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 6
10 Rate 3: Calcium Channel Blocker (CCB) Medications All five organizations submitted data for commercial, two organizations submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 66.73% with the mean of 64.61% and median of 69.32% across the range of 51.04% to 72.51%. The aggregate summary rate for Medicaid is 52.70% and the single reported Medicare rate is 68.88%. Exhibit 9: Proportion of Days Covered (80% Threshold) Rate 3: Calcium Channel Blocker Medications Exhibit 10: Proportion of Days Covered (80% Threshold) Rate 3: Calcium Channel Blocker Medications (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 7
11 Rate 4: Biguanides Four organizations submitted data for commercial, one organization submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 60.34% with the mean of 56.19% and median of 59.80% across the range of 42.24% to 62.91%. The single reported Medicaid rate is 55.43% and the single reported Medicare rate is 64.14%. Exhibit 11: Proportion of Days Covered (80% Threshold) Rate 4: Biguanides Exhibit 12: Proportion of Days Covered (80% Threshold) Rate 4: Biguanides 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 8
12 Rate 5: Sulfonylureas Four organizations submitted data for commercial, one organization submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 60.26% with the mean of 58.23% and median of 60.94% across the range of 46.42% to 64.65%. The single reported Medicaid rate is 51.54% and the single reported Medicare rate is 66.74%. Exhibit 13: Proportion of Days Covered (80% Threshold) Rate 5: Sulfonylureas Exhibit 14: Proportion of Days Covered (80% Threshold) Rate 5: Sulfonylureas (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 9
13 Rate 6: Thiazolidinediones Four organizations submitted data for commercial, one organization submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 65.07% with the mean of 62.65% and median of 64.07% across the range of 55.36% to 67.11%. The single reported Medicaid rate is 56.53% and the single reported Medicare rate is 71.97%. Exhibit 15: Proportion of Days Covered (80% Threshold) Rate 6: Thiazolidinediones Exhibit 16: Proportion of Days Covered (80% Threshold) Rate 6: Thiazolidinediones (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 10
14 Rate 7: Dipeptidyl Peptidase-4 (DPP-IV) Inhibitors Four organizations submitted data for commercial, one organization submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 67.21% with the mean of 62.02% and median of 67.59% across the range of 44.01% to 68.89%. The single reported Medicaid rate is 61.32% and the single reported Medicare rate is 68.91%. Exhibit 17: Proportion of Days Covered (80% Threshold) Rate 7: DPP-IV Inhibitors Exhibit 18: Proportion of Days Covered (80% Threshold) Rate 7: DPP-IV Inhibitors (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 11
15 Rate 8: Diabetes All Class Rate Four organizations submitted data for commercial, one organization submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 68.65% with the mean of 63.38% and median of 68.19% across the range of 47.12% to 70.01%. The single reported Medicaid rate is 53.96% and the single reported Medicare rate is 70.29%. Exhibit 19: Proportion of Days Covered (80% Threshold) Rate 8: Diabetes All Class Rate Exhibit 20: Proportion of Days Covered (80% Threshold) Rate 8: Diabetes All Class Rate (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 12
16 Rate 9: Statins All five organizations submitted data for commercial, two organizations submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 62.21% with the mean of 58.77% and median of 61.50% across the range of 42.50% to 64.51%. The aggregate summary rate for Medicaid is 54.90% and the single reported Medicare rate is 72.37%. Exhibit 21: Proportion of Days Covered (80% Threshold) Rate 9: Statins Exhibit 22: Proportion of Days Covered (80% Threshold) Rate 9: Statins (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 13
17 Rate 10: Anti-Retroviral Medications All five organizations submitted data for commercial, two organizations submitted for Medicaid, and one organization submitted for Medicare. One of commercial submissions had a small denominator of less than 30. The aggregate summary rate for the four valid commercial submissions is 52.92% with the mean of 43.50% and median of 48.74% across the range of 18.50% to 58.01%. The aggregate summary rate for Medicaid is 46.30% and the single reported Medicare rate is 41.73%. Exhibit 23: Proportion of Days Covered (80% Threshold) Rate 10: Anti-Retroviral Medications Exhibit 24: Proportion of Days Covered (80% Threshold) Rate 10: Anti-Retroviral Medications (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 14
18 Measure 2 Adherence to Non-Warfarin Oral Anticoagulants (DTM ) Measure Description This mandatory measure assesses the percentage of patients 18 years and older who met the Proportion of Days Covered (PDC) threshold of 80% during the measurement period for non-warfarin oral anticoagulants. The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc Exhibit 25: Adherence to Non-Warfarin Oral Anticoagulants Summary of Findings All five organizations submitted data for commercial, one organization submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 77.09% with the mean of 78.45% and median of 78.46% across the range of 75.62% to 81.51%. The single reported Medicaid rate is 62.12% and the single reported Medicare rate is 68.39%. Exhibit 26: Adherence to Non-Warfarin Oral Anticoagulants (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 15
19 Measure 3 Generic Dispensing Rates (MP ) Measure Description This mandatory measure assesses the percentage of all prescriptions that were dispensed as generics, branded generics, or brands for which members paid the generic co-pay. Exhibit 27: Generic Dispensing Rates Summary of Findings All five organizations submitted data for commercial, two organizations submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 97.84% with the mean of 97.27% and median of 97.80% across the range of 94.53% to 98.34%. The aggregate summary rate for Medicaid is 98.59% and the single reported Medicare rate is 98.30%. Exhibit 28: Generic Dispensing Rates (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 16
20 Measure 4 Drug-Drug Interactions (DM ) Measure Description This mandatory measure assesses the percentage of patients who received a prescription for a target medication during the measurement period and who were dispensed a concurrent prescription for a precipitant medication. The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc A lower rate represents better performance. Exhibit 29: Drug-Drug Interactions Note: Lower rate represents better performance Summary of Findings All five organizations submitted data for commercial, two organizations submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 2.14% with the mean of 2.23% and median of 2.13% across the range of 1.53% to 3.23%. The aggregate summary rate for Medicaid is 3.15% and the single reported Medicare rate is 6.15%. Exhibit 30: Drug-Drug Interactions (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 17
21 Measure 5 Use of High-Risk Medications in the Elderly (HRM) (HIM ) Measure Description This mandatory measure assesses the percentage of patients 65 years of age and older who received two or more prescription fills for a high-risk medication during the measurement period. The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc A lower rate represents better performance. Exhibit 31: Use of High-Risk Medications in the Elderly Note: Lower rate represents better performance Summary of Findings All five organizations submitted data for commercial, two organizations submitted for Medicaid, and one organization submitted for Medicare. The aggregate summary rate for commercial is 8.64% with the mean of 8.17% and median of 7.80% across the range of 0.57% to 19.76%. The aggregate summary rate for Medicaid is 8.53% and the single reported Medicare rate is 2.58%. Exhibit 32: Use of High-Risk Medications in the Elderly (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 18
22 Measure 6 Call Center Performance (DTM ) Measure Description This mandatory measure has two parts: Part A evaluates the percentage of calls during normal business hours to the organization s call service center(s) during the measurement period that were answered by a live voice within 30 seconds; Part B evaluates the percentage of calls made during normal business hours to the organization s call service center(s) during the reporting year that were abandoned by callers before being answered by a live customer service representative. For Part B, a lower rate represents better performance. Exhibit 33: Call Center Performance Note: Lower rate represents better performance for Part B: Call Abandonment. Summary of Findings All five organizations submitted data combined for all books of business. The aggregate summary rate for Part A: 30-Second Response Rate is 86.22% with the mean of 80.51% and median of 86.87% across the range of 46.23% to 94.36%. The aggregate summary rate for Part B: Call Abandonment Rate is 1.82% with the mean of 4.12% and median of 1.62% across the range of 1.17% to 14.29%. Exhibit 34: Call Center Performance (Summary Data) 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 19
23 Measure 7 Turnaround Time for Prescriptions (MP ) Measure Description This exploratory 3-part measure assesses the average speed with which the organization fills prescriptions, once the prescription is clean. Part A measures prescription turnaround time for clean prescriptions; Part B measures prescription turnaround time for prescriptions that required intervention; and Part C measures prescription turnaround time for all prescriptions. Summary of Findings No organizations submitted data for this exploratory measure URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 20
24 Measure 8 Antipsychotic Use in Children Under 5 Years Old (PH ) Measure Description This exploratory measure assesses percentage of children under age 5 using antipsychotic medications during the measurement period. The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc Summary of Findings Only one organization submitted data for this measure. Analysis and benchmarks were not produced given there were less than five valid data submissions URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 21
25 Measure 9 Use of Benzodiazepine Sedative Hypnotic Medications in the Elderly (PH ) Measure Description This exploratory measure assesses percentage of individuals 65 years of age and older that received two or more prescription fills for any benzodiazepine sedative hypnotic for a cumulative period of more than 90 days. The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc Summary of Findings Only one organization submitted data for this measure. Analysis and benchmarks were not produced given there were less than five valid data submissions URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 22
26 Measure 10 Diabetes Medication Dosing (PH ) Measure Description This exploratory measure assesses percentage patients who were dispensed a dose higher than the daily recommended dose for the following therapeutic categories of oral hypoglycemics: biguanides, sulfonlyureas, thiazolidinediones, and DPP-IV inhibitors. Dosing rates for each category are to be reported separately. The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc Summary of Findings Only one organization submitted data for this measure. Analysis and benchmarks were not produced given there were less than five valid data submissions URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 23
27 Measure 11 Statin Use in Persons with Diabetes (PH ) Measure Description This exploratory measure assesses the percentage of patients ages 40-75years who were dispensed a medication for diabetes that receive a statin medication. The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc Summary of Findings Only one organization submitted data for this measure. Analysis and benchmarks were not produced given there were less than five valid data submissions URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 24
28 Measure 12 Consumer Experience with Pharmacy Services (PH ) Measure Description This exploratory measure assesses the consumer experience. The Pharmacy Quality Alliance (PQA) is the measure steward and all rights are retained by PQA Inc Summary of Findings No organizations submitted data for this exploratory measure URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 25
29 Concluding Remarks This performance report has been prepared by the URAC Quality, Research and Measurement Department. If you have any questions about the results contained herein, please contact us at: 2017 URAC. All rights reserved. Reproduction by any means in whole or part without written permission is prohibited. 26
MEASURE STEWARD Pharmacy Quality Alliance (PQA) D ATA SOURCE Enrollment; U R A C DOMAIN Engagement & Experience of Care
 MANDATORY S Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. DM2012-12 DTM2015-01 Portion of Days Covered (PDC) Adherence
MANDATORY S Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. DM2012-12 DTM2015-01 Portion of Days Covered (PDC) Adherence
Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis.
 COMMUNITY PHARMACY V1.1 MANDATORY S Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. # NAME DESCRIPTION NUMERATOR DENOMINATOR
COMMUNITY PHARMACY V1.1 MANDATORY S Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. # NAME DESCRIPTION NUMERATOR DENOMINATOR
Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis.
 Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. DESCRIPTION NUMERATOR DENOMINATOR DM2012-12 Portion of Days Covered
Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. DESCRIPTION NUMERATOR DENOMINATOR DM2012-12 Portion of Days Covered
Pharmacy Benefit Management
 Benefit Management Measures At A Glance Updated: 10/8/2015 Page 1 of 7 Benefit Management Mandatory Measures (6) Note: Mandatory measures are those measures that are a requirement of accreditation and
Benefit Management Measures At A Glance Updated: 10/8/2015 Page 1 of 7 Benefit Management Mandatory Measures (6) Note: Mandatory measures are those measures that are a requirement of accreditation and
Drug Therapy Management
 Measures At A Glance Updated: 11/01/2018 2018 Page 1 of 9 Mandatory Measures (10) Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to on an annual
Measures At A Glance Updated: 11/01/2018 2018 Page 1 of 9 Mandatory Measures (10) Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to on an annual
Mail Service Pharmacy
 Mail Service s At A Glance Disclaimer: reserves the right to update its measures and measure sets to maintain measure relevancy and to remedy any unintended consequences that may arise during implementation.
Mail Service s At A Glance Disclaimer: reserves the right to update its measures and measure sets to maintain measure relevancy and to remedy any unintended consequences that may arise during implementation.
Disease Management. Measures At A Glance
 s At A Glance Updated: 11/2/2017 Page 1 of 7 Cross Cutting Mandatory s (4) Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual
s At A Glance Updated: 11/2/2017 Page 1 of 7 Cross Cutting Mandatory s (4) Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual
Disease Management. Measures At A Glance
 s At A Glance Updated: 11/01/2018 URAC 2018 Page 1 of 7 Cross-Cutting Mandatory s (3) Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on
s At A Glance Updated: 11/01/2018 URAC 2018 Page 1 of 7 Cross-Cutting Mandatory s (3) Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on
PATIENT-IMPACT SCORECARD
 UNDERSTANDING THE GSC PATIENT-IMPACT SCORECARD What is the GSC Patient-Impact Scorecard? The GSC Patient-Impact Scorecard shows pharmacy performance scores for GSC patients during the period indicated.
UNDERSTANDING THE GSC PATIENT-IMPACT SCORECARD What is the GSC Patient-Impact Scorecard? The GSC Patient-Impact Scorecard shows pharmacy performance scores for GSC patients during the period indicated.
Condition/Procedure Measure Compliance Criteria Reference Attribution Method
 Premium Specialty: Cardiology Credentialed Specialties include: Cardiac Diagnostic, Cardiology, Cardiovascular Disease, Clinical Cardiac Electrophysiology, and Interventional Cardiology This document is
Premium Specialty: Cardiology Credentialed Specialties include: Cardiac Diagnostic, Cardiology, Cardiovascular Disease, Clinical Cardiac Electrophysiology, and Interventional Cardiology This document is
6/18/2015. Disclosure. Objectives. Star Ratings. Understand the current climate of healthcare reform
 Star Ratings John A. Galdo, Pharm.D., BCPS, CGP (Jake) Assistant Professor of Pharmacy Practice Community Practice Residency Director jgaldo@samford.edu Disclosure I do not have (nor does any immediate
Star Ratings John A. Galdo, Pharm.D., BCPS, CGP (Jake) Assistant Professor of Pharmacy Practice Community Practice Residency Director jgaldo@samford.edu Disclosure I do not have (nor does any immediate
9/25/15. Pharmacy Quality Measures: Financial Support. Learning Objectives. Speaker Disclosure. Access to Preferred Networks and Clinical Performance
 Pharmacy Quality Measures: Action Steps for Improvement Financial Support Financial support was provided for this activity through an unrestricted grant from Health Mart Systems, Inc. Christine Jacobson
Pharmacy Quality Measures: Action Steps for Improvement Financial Support Financial support was provided for this activity through an unrestricted grant from Health Mart Systems, Inc. Christine Jacobson
Alabama Medicaid Pharmacy Override
 Alabama Medicaid Pharmacy Override Therapeutic Duplication, Early Refill, Maximum Unit, Prescription Limit Switchover, Dispense as Written, Accumulation Edit, Maintenance Supply Opt Out, and Maximum Cost
Alabama Medicaid Pharmacy Override Therapeutic Duplication, Early Refill, Maximum Unit, Prescription Limit Switchover, Dispense as Written, Accumulation Edit, Maintenance Supply Opt Out, and Maximum Cost
ANALYSIS OF KENTUCKY MEDICAID MANAGED CARE VERSUS FEE-FOR- SERVICE SYSTEMS: MEDICATION ADHERENCE IN PATIENTS WITH PREVALENT CHRONIC DISEASES
 University of Kentucky UKnowledge Theses and Dissertations--Pharmacy College of Pharmacy 2016 ANALYSIS OF KENTUCKY MEDICAID MANAGED CARE VERSUS FEE-FOR- SERVICE SYSTEMS: MEDICATION ADHERENCE IN PATIENTS
University of Kentucky UKnowledge Theses and Dissertations--Pharmacy College of Pharmacy 2016 ANALYSIS OF KENTUCKY MEDICAID MANAGED CARE VERSUS FEE-FOR- SERVICE SYSTEMS: MEDICATION ADHERENCE IN PATIENTS
Medicare Star Ratings and the Shift to Quality- Based Payment Models. David Nau, RPh, PhD, FAPhA PQS President
 Medicare Star Ratings and the Shift to Quality- Based Payment Models David Nau, RPh, PhD, FAPhA PQS President The shift to Value-Driven Healthcare The U.S. health care system is rapidly moving to value-based
Medicare Star Ratings and the Shift to Quality- Based Payment Models David Nau, RPh, PhD, FAPhA PQS President The shift to Value-Driven Healthcare The U.S. health care system is rapidly moving to value-based
Understanding Your Patient Care Opportunity Report (PCOR)
 Understanding Your Patient Care Opportunity Report (PCOR) Use your January/February 208 PCOR to help improve performance on Medicare Part D Clinical Star Ratings measures. Your January/February 208 Patient
Understanding Your Patient Care Opportunity Report (PCOR) Use your January/February 208 PCOR to help improve performance on Medicare Part D Clinical Star Ratings measures. Your January/February 208 Patient
116th Annual Convention
 116th Annual Convention Date: Tuesday, October 21, 2014 Time: 8:30 am 10:00 am Location: Austin Convention Center, Room 17AB, Level 4 Title: Activity Type: Speaker: Medicare Part D Star Ratings What They
116th Annual Convention Date: Tuesday, October 21, 2014 Time: 8:30 am 10:00 am Location: Austin Convention Center, Room 17AB, Level 4 Title: Activity Type: Speaker: Medicare Part D Star Ratings What They
STARS SYSTEM 5 CATEGORIES
 TMG STARS 2018 1 2 STARS Program Implemented in 2008 by CMS. Tool to inform beneficiaries of quality of various health plans 5-star rating system Used to adjust payments to health plans (bonus to plans
TMG STARS 2018 1 2 STARS Program Implemented in 2008 by CMS. Tool to inform beneficiaries of quality of various health plans 5-star rating system Used to adjust payments to health plans (bonus to plans
Pharmacy Technician Course
 Pharmacy Technician Course VERSION HISTORY SECTION 1: History and Scope of the Pharmacy Technician Scope of the Pharmacy Technician Section 1 Quiz Check Your Knowledge Section 1 Quiz Answers SECTION 2:
Pharmacy Technician Course VERSION HISTORY SECTION 1: History and Scope of the Pharmacy Technician Scope of the Pharmacy Technician Section 1 Quiz Check Your Knowledge Section 1 Quiz Answers SECTION 2:
PQS Summary of Pharmacy/ Medication-Related Updates in the CY 2020 Final Call Letter
 Managing Performance Information in a Quality Driven World PQS Summary of Pharmacy/ Medication-Related Updates in the CY 2020 Final Call Letter REGULATORY UPDATE PQS Summary of Pharmacy/ Medication-Related
Managing Performance Information in a Quality Driven World PQS Summary of Pharmacy/ Medication-Related Updates in the CY 2020 Final Call Letter REGULATORY UPDATE PQS Summary of Pharmacy/ Medication-Related
USING MTM TO IMPROVE STAR RATINGS : CASE STUDIES
 USING MTM TO IMPROVE STAR RATINGS : CASE STUDIES Sept 13, 2015 Amanda Applegate, PharmD, BCACP Disclosures Nothing to disclose Learning Objectives Describe how MTM can improve star ratings Discuss strategies
USING MTM TO IMPROVE STAR RATINGS : CASE STUDIES Sept 13, 2015 Amanda Applegate, PharmD, BCACP Disclosures Nothing to disclose Learning Objectives Describe how MTM can improve star ratings Discuss strategies
PHARMACY BENEFITS MANAGER SELECTION FAQ FOR PRODUCERS
 PHARMACY BENEFITS MANAGER SELECTION FAQ FOR PRODUCERS For Producer Audience Only - Please Do Not Distribute Regence has selected Prime Therapeutics as the Pharmacy benefits manager for its health plans.
PHARMACY BENEFITS MANAGER SELECTION FAQ FOR PRODUCERS For Producer Audience Only - Please Do Not Distribute Regence has selected Prime Therapeutics as the Pharmacy benefits manager for its health plans.
2016 Medicaid Adult CAHPS 5.0H. At-A-Glance Report
 2016 Medicaid Adult CAHPS 5.0H At-A-Glance Report Health Partners Plans Project Number(s): 4109174 Current data as of: 06/16/2016 1965 Evergreen Boulevard Suite 100, Duluth, Georgia 30096 2016 At-A-Glance
2016 Medicaid Adult CAHPS 5.0H At-A-Glance Report Health Partners Plans Project Number(s): 4109174 Current data as of: 06/16/2016 1965 Evergreen Boulevard Suite 100, Duluth, Georgia 30096 2016 At-A-Glance
Drug Use Evaluation: Low Dose Quetiapine
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Electronic Health Records (EHR) HP Provider Relations October 2012
 Electronic Health Records (EHR) HP Provider Relations October 2012 Agenda Session Objectives Electronic Health Record (EHR) EHR Incentive Program Certified Technology EHR Meaningful Use EHR Incentive Program
Electronic Health Records (EHR) HP Provider Relations October 2012 Agenda Session Objectives Electronic Health Record (EHR) EHR Incentive Program Certified Technology EHR Meaningful Use EHR Incentive Program
Introducing ValueOptions Clinical Care Alerts
 Introducing ValueOptions Clinical Care Alerts January 2013 Agenda What are Clinical Care Alerts? How does it work? What are the benefits? Questions and Answers 2 The Issue of Poor Medication Adherence
Introducing ValueOptions Clinical Care Alerts January 2013 Agenda What are Clinical Care Alerts? How does it work? What are the benefits? Questions and Answers 2 The Issue of Poor Medication Adherence
Capital Health Plan CMS Star Ratings Strategies for Improvement
 Capital Health Plan CMS Star Ratings Strategies for Improvement ESTRELLITA REDMON, MD, MBA MEDICAL DIRECTOR The Ultimate Goal Outline Current 5 Star Plans CHP History Importance of Ratings Part C and Part
Capital Health Plan CMS Star Ratings Strategies for Improvement ESTRELLITA REDMON, MD, MBA MEDICAL DIRECTOR The Ultimate Goal Outline Current 5 Star Plans CHP History Importance of Ratings Part C and Part
Understanding and Interpreting Adverse Drug Event (ADE) Readmission Reports
 Understanding and Interpreting Adverse Drug Event (ADE) Readmission Reports Lindsay Holland Director, Care Transitions Health Services Advisory Group (HSAG) October 24, 2017 Today s Webinar Objectives
Understanding and Interpreting Adverse Drug Event (ADE) Readmission Reports Lindsay Holland Director, Care Transitions Health Services Advisory Group (HSAG) October 24, 2017 Today s Webinar Objectives
New Measure Recommended for Endorsement by PQA
 New Measure Recommended for Endorsement by PQA Measure: Statin Use in Persons with Diabetes Description: The percentage of patients ages 40 75 years who were dispensed a medication for diabetes that receive
New Measure Recommended for Endorsement by PQA Measure: Statin Use in Persons with Diabetes Description: The percentage of patients ages 40 75 years who were dispensed a medication for diabetes that receive
Modernized Reference Drug Program
 Modernized Reference Drug Program Monitoring Report For the period ending May 31, 2017 Issued: November 2017 Published by: Pharmaceutical Services Division and Health Sector Information, Analysis and Reporting
Modernized Reference Drug Program Monitoring Report For the period ending May 31, 2017 Issued: November 2017 Published by: Pharmaceutical Services Division and Health Sector Information, Analysis and Reporting
Tips for Evolving Medicaid Pharmacy Benefits Management (PBM) Programs. June 5, 2015
 Tips for Evolving Medicaid Pharmacy Benefits Management (PBM) Programs 1 June 5, 2015 Introductions Mark Steck Pharm.D Independent Consultant, MAXIMUS John J.P. Crouse Vice President, MAXIMUS Market Lead
Tips for Evolving Medicaid Pharmacy Benefits Management (PBM) Programs 1 June 5, 2015 Introductions Mark Steck Pharm.D Independent Consultant, MAXIMUS John J.P. Crouse Vice President, MAXIMUS Market Lead
Legislative & Regulatory Update Brad Young, RxPlus Government Affairs
 Legislative & Regulatory Update 2018 Brad Young, RxPlus Government Affairs Disclosures Brad Young reports no actual or potential conflicts of interest associated with this presentation. 2 Learning Objectives
Legislative & Regulatory Update 2018 Brad Young, RxPlus Government Affairs Disclosures Brad Young reports no actual or potential conflicts of interest associated with this presentation. 2 Learning Objectives
2017 Medicare CAHPS At-A-Glance Report
 2017 Medicare CAHPS At-A-Glance Report CMS MA PD Contract: H9207 Project Number(s): 30120117 and 10123759 Current data as of: 07/10/2017 1965 Evergreen Boulevard Suite 100, Duluth, Georgia 30096 2017 At-A-Glance
2017 Medicare CAHPS At-A-Glance Report CMS MA PD Contract: H9207 Project Number(s): 30120117 and 10123759 Current data as of: 07/10/2017 1965 Evergreen Boulevard Suite 100, Duluth, Georgia 30096 2017 At-A-Glance
General Providers. Tamper-Resistant Prescription Requirements
 September 2008 Provider Bulletin Number 8138 General Providers Tamper-Resistant Prescription Requirements Effective with processing dates on and after October 1, 2008, written prescriptions for Kansas
September 2008 Provider Bulletin Number 8138 General Providers Tamper-Resistant Prescription Requirements Effective with processing dates on and after October 1, 2008, written prescriptions for Kansas
This guide alerts you to important preventive care and services that you can provide to patients to help boost Star Ratings.
 2019 HEDIS AT-A-GLANCE GUIDE STAR MEASURES This guide alerts you to important preventive care and services that you can provide to patients to help boost Star Ratings. At WellCare, we value everything
2019 HEDIS AT-A-GLANCE GUIDE STAR MEASURES This guide alerts you to important preventive care and services that you can provide to patients to help boost Star Ratings. At WellCare, we value everything
Value Based Pay for Performance Results & Highlights Measurement Year September 2017
 Value Based Pay for Performance Results & Highlights Measurement Year 2016 September 2017 IHA s Value Based Pay for Performance (VBP4P) ~200 Medical Groups & IPAs 9 Health Plans Common Measurement Public
Value Based Pay for Performance Results & Highlights Measurement Year 2016 September 2017 IHA s Value Based Pay for Performance (VBP4P) ~200 Medical Groups & IPAs 9 Health Plans Common Measurement Public
Commercial HMO/POS Effectiveness of Care Measure
 Commercial HMO/POS Effectiveness of Care Measure HEDIS 2017 NCQA Quality Compass National Average Adult BMI Assessment 91.85% 76.17% Weight Assessment and Counseling for Nutrition and Physical Activity
Commercial HMO/POS Effectiveness of Care Measure HEDIS 2017 NCQA Quality Compass National Average Adult BMI Assessment 91.85% 76.17% Weight Assessment and Counseling for Nutrition and Physical Activity
Effective Strategies to Help Customers Use Their EHR to Improve Quality of Care
 Effective Strategies to Help Customers Use Their EHR to Improve Quality of Care Brian Bamberger Life Sciences Practice Leader Technology is transforming healthcare Health technology will continue to diffuse
Effective Strategies to Help Customers Use Their EHR to Improve Quality of Care Brian Bamberger Life Sciences Practice Leader Technology is transforming healthcare Health technology will continue to diffuse
Challenges for U.S. Attorneys Offices (USAO) in Opioid Cases
 Challenges for U.S. Attorneys Offices (USAO) in Opioid Cases Overview On August 2, 2017, U.S. Attorney General Jeff Sessions announced a pilot program whereby a new federal data analysis program is being
Challenges for U.S. Attorneys Offices (USAO) in Opioid Cases Overview On August 2, 2017, U.S. Attorney General Jeff Sessions announced a pilot program whereby a new federal data analysis program is being
Supplementary Online Content
 Supplementary Online Content Lin Y-S, Chen Y-L, Chen T-H, et al. Comparison of Clinical Outcomes Among Patients With Atrial Fibrillation or Atrial Flutter Stratified by CHA 2 DS 2 -VASc Score. JAMA Netw
Supplementary Online Content Lin Y-S, Chen Y-L, Chen T-H, et al. Comparison of Clinical Outcomes Among Patients With Atrial Fibrillation or Atrial Flutter Stratified by CHA 2 DS 2 -VASc Score. JAMA Netw
CMS EHR Incentive Program: Proposed Rules for Stage 2 and Stage 3 Meaningful Use Focus on Eligible Hospitals
 CMS EHR Incentive Program: Proposed Rules for Stage 2 and Stage 3 Meaningful Use Focus on Eligible Hospitals April 28, 2015 Hosted by: Sponsored by: Today s Event Sponsored By One of the nation s largest
CMS EHR Incentive Program: Proposed Rules for Stage 2 and Stage 3 Meaningful Use Focus on Eligible Hospitals April 28, 2015 Hosted by: Sponsored by: Today s Event Sponsored By One of the nation s largest
Hypoglycemia and Quality Measurement
 Hypoglycemia and Quality Measurement Sam Stolpe Senior Director The Triple Aim Affordable Care Better Care Healthy People/ Communities 1 Comprehensive Overview of CMS Quality Programs Hospital Quality
Hypoglycemia and Quality Measurement Sam Stolpe Senior Director The Triple Aim Affordable Care Better Care Healthy People/ Communities 1 Comprehensive Overview of CMS Quality Programs Hospital Quality
2018 Commercial HMO/POS HEDIS 1 Results
 08 Commercial HMO/POS HEDIS Results Weight Assessment & Counseling for Nutrition & Physical Activity for Children/Adolescents HEDIS 06 CY 05 HEDIS 07 CY 06 HEDIS 08 CY 07 Compass BMI Percentile 70.47%
08 Commercial HMO/POS HEDIS Results Weight Assessment & Counseling for Nutrition & Physical Activity for Children/Adolescents HEDIS 06 CY 05 HEDIS 07 CY 06 HEDIS 08 CY 07 Compass BMI Percentile 70.47%
The Advantage. A Newsletter for Providers. Welcome. What is an MI Health Link Medicare-Medicaid plan?
 A Newsletter for Providers Welcome AmeriHealth Caritas VIP Care Plus is pleased to welcome you to the AmeriHealth Caritas VIP Care Plus network of providers. AmeriHealth Caritas VIP Care Plus is an MI
A Newsletter for Providers Welcome AmeriHealth Caritas VIP Care Plus is pleased to welcome you to the AmeriHealth Caritas VIP Care Plus network of providers. AmeriHealth Caritas VIP Care Plus is an MI
PHARMACY Section 9. Overview. Preferred Drug List. Additions and Exceptions to the Preferred Drug List
 Overview The management of outpatient prescription drugs is an integral part of the medical management program to improve the health and well-being of our members. Prescriber and member involvement is
Overview The management of outpatient prescription drugs is an integral part of the medical management program to improve the health and well-being of our members. Prescriber and member involvement is
B&T Format. New Measures. Better health care. Better choices. Better health.
 1100 13th Street NW, Third Floor Washington, DC 20005 phone 202.955.3500 fax 202.955.3599 www.ncqa.org TO: Interested Organizations FROM: Patrick Dahill, Assistant Vice President, Policy DATE: February
1100 13th Street NW, Third Floor Washington, DC 20005 phone 202.955.3500 fax 202.955.3599 www.ncqa.org TO: Interested Organizations FROM: Patrick Dahill, Assistant Vice President, Policy DATE: February
Trending Determinations by Measure
 TO: Interested Parties FROM: Cindy Ottone, Director, Policy DATE: February 2018 RE: HEDIS 1 2018 Measure Trending Determinations This memo communicates trending determinations for measures in the HEDIS
TO: Interested Parties FROM: Cindy Ottone, Director, Policy DATE: February 2018 RE: HEDIS 1 2018 Measure Trending Determinations This memo communicates trending determinations for measures in the HEDIS
Prescription Drug Abuse: Developing Evidence-Based State Level Interventions
 Prescription Drug Abuse: Developing Evidence-Based State Level Interventions Signe Shackelford, MPH Policy Analyst APHA Annual Meeting November 5, 2013 Presenter Disclosure The following personal financial
Prescription Drug Abuse: Developing Evidence-Based State Level Interventions Signe Shackelford, MPH Policy Analyst APHA Annual Meeting November 5, 2013 Presenter Disclosure The following personal financial
Pharmacogenomic Testing for Warfarin Response (NCD 90.1)
 Policy Number 90.1 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 01/08/2014 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare
Policy Number 90.1 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 01/08/2014 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare
The University of Mississippi School of Pharmacy
 LONG TERM PERSISTENCE WITH ACEI/ARB THERAPY AFTER ACUTE MYOCARDIAL INFARCTION: AN ANALYSIS OF THE 2006-2007 MEDICARE 5% NATIONAL SAMPLE DATA Lokhandwala T. MS, Yang Y. PhD, Thumula V. MS, Bentley J.P.
LONG TERM PERSISTENCE WITH ACEI/ARB THERAPY AFTER ACUTE MYOCARDIAL INFARCTION: AN ANALYSIS OF THE 2006-2007 MEDICARE 5% NATIONAL SAMPLE DATA Lokhandwala T. MS, Yang Y. PhD, Thumula V. MS, Bentley J.P.
SUMMARY TABLE OF MEASURES, PRODUCT LINES AND CHANGES
 Summary Table of Measures, Product Lines and Changes 1 SUMMARY TABLE OF MEASURES, PRODUCT LINES AND CHANGES General Guidelines for Data Collection and Reporting Guidelines for Calculations and Sampling
Summary Table of Measures, Product Lines and Changes 1 SUMMARY TABLE OF MEASURES, PRODUCT LINES AND CHANGES General Guidelines for Data Collection and Reporting Guidelines for Calculations and Sampling
Leveraging Information Technology for Evidence-Based Design. December 15, 2005
 Leveraging Information Technology for Evidence-Based Design December 15, 2005 Introduction ActiveHealth Management Technology Evidence Based Formulary Program Lessons Learned 2 ActiveHealth Management
Leveraging Information Technology for Evidence-Based Design December 15, 2005 Introduction ActiveHealth Management Technology Evidence Based Formulary Program Lessons Learned 2 ActiveHealth Management
NEW JERSEY 2011 HOSPITAL PERFORMANCE REPORT TECHNICAL REPORT: METHODOLOGY RECOMMENDED CARE (PROCESS OF CARE) MEASURES
 NEW JERSEY 2011 HOSPITAL PERFORMANCE REPORT TECHNICAL REPORT: METHODOLOGY RECOMMENDED CARE (PROCESS OF CARE) MEASURES New Jersey Department of Health and Senior Services Health Care Quality Assessment
NEW JERSEY 2011 HOSPITAL PERFORMANCE REPORT TECHNICAL REPORT: METHODOLOGY RECOMMENDED CARE (PROCESS OF CARE) MEASURES New Jersey Department of Health and Senior Services Health Care Quality Assessment
a guide to Reimbursement of Intermittent Catheters Know your options M2116N 04.08
 a guide to Reimbursement of Intermittent Catheters 1 Know your options Coloplast Corp. Minneapolis, MN 55411 1.800.533.0464 usmedweb@coloplast.com www.us.coloplast.com is a registered trademark of Coloplast
a guide to Reimbursement of Intermittent Catheters 1 Know your options Coloplast Corp. Minneapolis, MN 55411 1.800.533.0464 usmedweb@coloplast.com www.us.coloplast.com is a registered trademark of Coloplast
Adverse Drug Events Impact on Hospital Readmissions
 Adverse Drug Events Impact on Hospital Readmissions Co hosted by FHA HIIN and HSAG Facilitators Edna Clifton HSAG, Florida QIN QIO Phyllis Byles FHA Scott King, PharmD Orlando Health Dr. P. Phillips Hospital
Adverse Drug Events Impact on Hospital Readmissions Co hosted by FHA HIIN and HSAG Facilitators Edna Clifton HSAG, Florida QIN QIO Phyllis Byles FHA Scott King, PharmD Orlando Health Dr. P. Phillips Hospital
Overview. Ontario Public Drug Programs, Ministry of Health and Long-Term Care
 Ontario Public Drug Programs, Ministry of Health and Long-Term Care Frequently Asked Questions for Pharmacists October 2015 Pharmacist Administration of the Publicly Funded Influenza Vaccine and Claims
Ontario Public Drug Programs, Ministry of Health and Long-Term Care Frequently Asked Questions for Pharmacists October 2015 Pharmacist Administration of the Publicly Funded Influenza Vaccine and Claims
Drug Utilization Review & Cost Reduction Strategies. Esther Liu, PharmD, MSIA, CGP Clinical Pharmacist Outcome Resources
 Drug Utilization Review & Cost Reduction Strategies Esther Liu, PharmD, MSIA, CGP Clinical Pharmacist Outcome Resources Goals and Objectives Define Drug Utilization Reviews Interpret various performance
Drug Utilization Review & Cost Reduction Strategies Esther Liu, PharmD, MSIA, CGP Clinical Pharmacist Outcome Resources Goals and Objectives Define Drug Utilization Reviews Interpret various performance
Medicare Advantage Measurement Period Handbook for Enhanced Personal Health Care Measurement Period beginning January 1, 2015
 Medicare Advantage Measurement Period Handbook for Enhanced Personal Health Care Measurement Period beginning January 1, 2015 Amerivantage is an HMO plan with a contract with the New Mexico Medicare program.
Medicare Advantage Measurement Period Handbook for Enhanced Personal Health Care Measurement Period beginning January 1, 2015 Amerivantage is an HMO plan with a contract with the New Mexico Medicare program.
Lindsay Erickson, Director, Value Based P4P, Integrated Healthcare Association. Measurement Years 2016 & 2017 Proposed Changes to the VBP4P Program
 To: From: Subject: Value Based Pay for Performance (VBP4P) Stakeholders Lindsay Erickson, Director, Value Based P4P, Integrated Healthcare Association Measurement Years 2016 & 2017 Proposed Changes to
To: From: Subject: Value Based Pay for Performance (VBP4P) Stakeholders Lindsay Erickson, Director, Value Based P4P, Integrated Healthcare Association Measurement Years 2016 & 2017 Proposed Changes to
Star Measures At-A-Glance Guide
 Star Measures At-A-Glance Guide This guide alerts you to important preventive care and services that you can provide to patients to help boost Star Ratings. ASSESSMENT AND SCREENING At Easy Choice, we
Star Measures At-A-Glance Guide This guide alerts you to important preventive care and services that you can provide to patients to help boost Star Ratings. ASSESSMENT AND SCREENING At Easy Choice, we
Exploring Temporal Patterns in Hypertensive Drug Therapy
 Exploring Temporal Patterns in Hypertensive Drug Therapy Sophia Wu 1, Margret Bjarnadottir 2, Eberechukwu Onukwugha 3, Catherine Plaisant 4, Sana Malik 5! 1. MSIS, Smith School of Business 2. Assistant
Exploring Temporal Patterns in Hypertensive Drug Therapy Sophia Wu 1, Margret Bjarnadottir 2, Eberechukwu Onukwugha 3, Catherine Plaisant 4, Sana Malik 5! 1. MSIS, Smith School of Business 2. Assistant
Frequently Asked Questions (FAQ) IHA Align. Measure. Perform. (AMP) Programs January Audit Audit Roadmap Posted 1/23/19
 Frequently Asked Questions (FAQ) IHA Align. Measure. Perform. (AMP) Programs January 2019 Audit Audit Roadmap Question: For the AMP MY 2018 Roadmap, is the last part of question 5.2W in Section 5, Describe
Frequently Asked Questions (FAQ) IHA Align. Measure. Perform. (AMP) Programs January 2019 Audit Audit Roadmap Question: For the AMP MY 2018 Roadmap, is the last part of question 5.2W in Section 5, Describe
Integrated Healthcare Association California Value Based Pay for Performance Program. DRAFT Measurement Year 2017 VBP4P Manual
 Integrated Healthcare Association California Value Based Pay for Performance Program DRAFT Measurement Year 2017 VBP4P Manual Updated No part of this publication may be reproduced or transmitted in any
Integrated Healthcare Association California Value Based Pay for Performance Program DRAFT Measurement Year 2017 VBP4P Manual Updated No part of this publication may be reproduced or transmitted in any
Texas Vendor Drug Program. Formulary Drug Index File Layout. Layout effective: Jul. 2, 2018 Document update: Oct. 1, 2018
 Texas Vendor Drug Program Formulary Drug Index File Layout Layout effective: Jul. 2, 2018 Document update: Oct. 1, 2018 The Vendor Drug Program provides a weekly update of resource data available for download
Texas Vendor Drug Program Formulary Drug Index File Layout Layout effective: Jul. 2, 2018 Document update: Oct. 1, 2018 The Vendor Drug Program provides a weekly update of resource data available for download
NWCDC 2018 Pharmacy Analytics in Workers Comp
 NWCDC 2018 Pharmacy Analytics in Workers Comp Cliff Belliveau VP Business Intelligence What is Visual Analytics? Transforming data into insights that tell a story 2 A little history Jacquard loom Punch
NWCDC 2018 Pharmacy Analytics in Workers Comp Cliff Belliveau VP Business Intelligence What is Visual Analytics? Transforming data into insights that tell a story 2 A little history Jacquard loom Punch
Thank you for your continued engagement and member support of the Pharmacy Quality Alliance.
 Optimizing Health by Advancing the Quality of Medication Use November 14, 2017 FROM: Jacqueline Green, Chief Operating Officer, Pharmacy Quality Alliance SUBJECT: PQA Headlines & Highlights as we head
Optimizing Health by Advancing the Quality of Medication Use November 14, 2017 FROM: Jacqueline Green, Chief Operating Officer, Pharmacy Quality Alliance SUBJECT: PQA Headlines & Highlights as we head
B&T Format. New Measures. 2 CAHPS is a registered trademark of the Agency for Healthcare Research and Quality (AHRQ).
 TO: Interested Organizations FROM: Patrick Dahill, Assistant Vice President, Policy DATE: February 4, 2018 RE: 2018 Accreditation Benchmarks and Thresholds This document reports national benchmarks and
TO: Interested Organizations FROM: Patrick Dahill, Assistant Vice President, Policy DATE: February 4, 2018 RE: 2018 Accreditation Benchmarks and Thresholds This document reports national benchmarks and
B&T Format. New Measures. Better health care. Better choices. Better health.
 1100 13th Street NW, Third Floor Washington, DC 20005 phone 202.955.3500 fax 202.955.3599 www.ncqa.org TO: Interested Organizations FROM: Patrick Dahill, Assistant Vice President, Policy DATE: August 13,
1100 13th Street NW, Third Floor Washington, DC 20005 phone 202.955.3500 fax 202.955.3599 www.ncqa.org TO: Interested Organizations FROM: Patrick Dahill, Assistant Vice President, Policy DATE: August 13,
ADMINISTRATIVE POLICY AND PROCEDURES MedStar Family Choice Medicare Advantage Plans
 ADMINISTRATIVE POLICY AND PROCEDURES MedStar Family Choice Medicare Advantage Plans DEPARTMENT: Medicare Compliance POLICY TITLE: RELATED DEPARTMENTS: All POLICY #: 706 C VERSION #: 4 REVISION DATE: Identifying
ADMINISTRATIVE POLICY AND PROCEDURES MedStar Family Choice Medicare Advantage Plans DEPARTMENT: Medicare Compliance POLICY TITLE: RELATED DEPARTMENTS: All POLICY #: 706 C VERSION #: 4 REVISION DATE: Identifying
Quick Guide MEDICATIONS 7th Edition Evan Sisson, Pharm.D., MHA, CDE
 Quick Guide to MEDICATIONS 7th Edition Evan Sisson, Pharm.D., MHA, CDE Adapted from The Art and Science of Diabetes Self-Management Education Desk Reference 2017, American Association of Diabetes Educators,
Quick Guide to MEDICATIONS 7th Edition Evan Sisson, Pharm.D., MHA, CDE Adapted from The Art and Science of Diabetes Self-Management Education Desk Reference 2017, American Association of Diabetes Educators,
Trending Determinations by Measure
 1100 13th Street NW, Third Floor Washington, DC 20005 phone 202.955.3500 fax 202.955.3599 www.ncqa.org TO: Interested Parties FROM: Cindy Ottone, Director, Policy DATE: March 2019 RE: HEDIS 1 2019 Measure
1100 13th Street NW, Third Floor Washington, DC 20005 phone 202.955.3500 fax 202.955.3599 www.ncqa.org TO: Interested Parties FROM: Cindy Ottone, Director, Policy DATE: March 2019 RE: HEDIS 1 2019 Measure
Star Measures At-A-Glance Guide
 Star Measures At-A-Glance Guide This guide alerts you to important preventive care and services that you can provide to patients to help boost Star Ratings. ASSESSMENT AND SCREENING At WellCare, we value
Star Measures At-A-Glance Guide This guide alerts you to important preventive care and services that you can provide to patients to help boost Star Ratings. ASSESSMENT AND SCREENING At WellCare, we value
ProviderNews FEBRUARY
 ProviderNews FEBRUARY 2017 Reminder: decimal billing required on time-based therapy codes for BadgerCare Plus members In accordance with Forward Health guidelines, Security Health Plan requires decimal
ProviderNews FEBRUARY 2017 Reminder: decimal billing required on time-based therapy codes for BadgerCare Plus members In accordance with Forward Health guidelines, Security Health Plan requires decimal
Title: Antipsychotic Use in Persons with Dementia CMS ID: ARCO3 NQF #: N/A
 Source(s) Pharmacy Quality Alliance (PQA). Technical specifications for PQA approved measures. Springfield (VA): Pharmacy Quality Alliance (PQA); 2015 Jul. 66 p. Measure Domain Clinical Quality Measures:
Source(s) Pharmacy Quality Alliance (PQA). Technical specifications for PQA approved measures. Springfield (VA): Pharmacy Quality Alliance (PQA); 2015 Jul. 66 p. Measure Domain Clinical Quality Measures:
How Independent Pharmacies Can Use Medicare Star Ratings to Improve Their Businesses
 How Independent Pharmacies Can Use Medicare Star Ratings to Improve Their Businesses We are in a new, changing healthcare environment that is affecting every stakeholder in the market hospitals, doctors,
How Independent Pharmacies Can Use Medicare Star Ratings to Improve Their Businesses We are in a new, changing healthcare environment that is affecting every stakeholder in the market hospitals, doctors,
STATE & FEDERAL EFFORTS TO COMBAT THE OPIOID EPIDEMIC & IMPACT ON COMPLIANCE PROGRAMS GRACE E. REBLING OSBORN MALEDON P.A.
 STATE & FEDERAL EFFORTS TO COMBAT THE OPIOID EPIDEMIC & IMPACT ON COMPLIANCE PROGRAMS GRACE E. REBLING OSBORN MALEDON P.A. Arizona Corporate Counsel Presentation May 8, 2018 Agenda 1. Overview of the Opioid
STATE & FEDERAL EFFORTS TO COMBAT THE OPIOID EPIDEMIC & IMPACT ON COMPLIANCE PROGRAMS GRACE E. REBLING OSBORN MALEDON P.A. Arizona Corporate Counsel Presentation May 8, 2018 Agenda 1. Overview of the Opioid
Amy Larrick Chavez-Valdez, Director, Medicare Drug Benefit and C & D Data Group
 DEPARTMENT OF HEALTH & HUMAN SERVICES Centers for Medicare & Medicaid Services 7500 Security Boulevard Baltimore, Maryland 21244-1850 CENTER FOR MEDICARE TO: FROM: SUBJECT: All Part D Sponsors Amy Larrick
DEPARTMENT OF HEALTH & HUMAN SERVICES Centers for Medicare & Medicaid Services 7500 Security Boulevard Baltimore, Maryland 21244-1850 CENTER FOR MEDICARE TO: FROM: SUBJECT: All Part D Sponsors Amy Larrick
Notice from the Executive Officer: Promoting Compliance with the Existing Limited Use Criteria for Fentanyl Transdermal Patch
 Notice from the Executive Officer: Promoting Compliance with the Existing Limited Use Criteria for Fentanyl Transdermal Patch Frequently Asked Questions 1. What is the new Health Network System feature
Notice from the Executive Officer: Promoting Compliance with the Existing Limited Use Criteria for Fentanyl Transdermal Patch Frequently Asked Questions 1. What is the new Health Network System feature
Quality Corp Measures Description and Methodologies
 Quality Corp Measures Description and Methodologies Overview: The Oregon Health Care Quality Corporation (Q Corp) is dedicated to improving the quality and affordability of health care in Oregon by leading
Quality Corp Measures Description and Methodologies Overview: The Oregon Health Care Quality Corporation (Q Corp) is dedicated to improving the quality and affordability of health care in Oregon by leading
Kentucky Department for Medicaid Services. Drug Review Options
 Kentucky Department for Medicaid Services Drug Review Options The following chart lists the agenda items scheduled and the options submitted for review at the March 18, 2010 meeting of the Pharmacy and
Kentucky Department for Medicaid Services Drug Review Options The following chart lists the agenda items scheduled and the options submitted for review at the March 18, 2010 meeting of the Pharmacy and
Jukti Kumar Kalita, PhD Business Analytics and Insights Pfizer Presented at:
 Hospital to Retail Spillover Analysis and Its Impact on Commercial Decision Making Jukti Kumar Kalita, PhD Business Analytics and Insights Pfizer Presented at: 1 Disclaimer The information provided and
Hospital to Retail Spillover Analysis and Its Impact on Commercial Decision Making Jukti Kumar Kalita, PhD Business Analytics and Insights Pfizer Presented at: 1 Disclaimer The information provided and
Plan Change Alert. New Market Priced Drug (MPD) Program Effective 11/1/2016. Alaska United Food and Commercial Workers Trust
 Plan Change Alert New Market Priced Drug (MPD) Program Effective 11/1/2016 Plan Sponsor Alaska United Food and Commercial Workers Trust Geographic Area Alaska Number of Participants Serviced 2,000 Announcement
Plan Change Alert New Market Priced Drug (MPD) Program Effective 11/1/2016 Plan Sponsor Alaska United Food and Commercial Workers Trust Geographic Area Alaska Number of Participants Serviced 2,000 Announcement
Study Exposures, Outcomes:
 GSK Medicine: Coreg IR, Coreg CR, and InnoPran Study No.: WWE111944/WEUSRTP3149 Title: A nested case-control study of the association between Coreg IR and Coreg CR and hypersensitivity reactions: anaphylactic
GSK Medicine: Coreg IR, Coreg CR, and InnoPran Study No.: WWE111944/WEUSRTP3149 Title: A nested case-control study of the association between Coreg IR and Coreg CR and hypersensitivity reactions: anaphylactic
Clinical Policy: Naltrexone (Vivitrol) Reference Number: CP.PHAR.96 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Vivitrol) Reference Number: CP.PHAR.96 Effective Date: 03.01.12 Last Review Date: 02.19 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Clinical Policy: (Vivitrol) Reference Number: CP.PHAR.96 Effective Date: 03.01.12 Last Review Date: 02.19 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Clinical Policy: Naltrexone (Vivitrol) Reference Number: CP.PHAR.96 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Vivitrol) Reference Number: CP.PHAR.96 Effective Date: 03.01.12 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Clinical Policy: (Vivitrol) Reference Number: CP.PHAR.96 Effective Date: 03.01.12 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
This Frequently Asked Questions document contains various aspects of UIIP in the following sections. Overview Eligibility...
 Ontario Public Drug Programs, Ministry of Health and Long-Term Care Frequently Asked Questions for Pharmacists October 2017: Pharmacist Administration of the Publicly Funded Influenza Vaccine and Claims
Ontario Public Drug Programs, Ministry of Health and Long-Term Care Frequently Asked Questions for Pharmacists October 2017: Pharmacist Administration of the Publicly Funded Influenza Vaccine and Claims
PDE File Medication Utilization; Medication Possession Ratio. Kyoungrae Jung, Ph.D. Assistant Professor Penn State University
 PDE File Medication Utilization; Medication Possession Ratio Kyoungrae Jung, Ph.D. Assistant Professor Penn State University Outline PDE variables related to medication utilization Measures of medication
PDE File Medication Utilization; Medication Possession Ratio Kyoungrae Jung, Ph.D. Assistant Professor Penn State University Outline PDE variables related to medication utilization Measures of medication
US Tardive Dyskinesia Market: Size, Trends & Forecasts ( ) December 2016
 US Tardive Dyskinesia Market: Size, Trends & Forecasts (2016-2025) December 2016 US Tardive Dyskinesia Market Report Scope of the Report The report entitled US Tardive Dyskinesia Market: Size, Trends &
US Tardive Dyskinesia Market: Size, Trends & Forecasts (2016-2025) December 2016 US Tardive Dyskinesia Market Report Scope of the Report The report entitled US Tardive Dyskinesia Market: Size, Trends &
Proposed Changes to Existing Measure for HEDIS : Use of Opioids at High Dosage (UOD)
 Proposed Changes to Existing Measure for HEDIS 1 2020: Use of Opioids at High Dosage (UOD) NCQA seeks comments on proposed revisions to the Use of Opioids at High Dosage HEDIS measure. The current measure
Proposed Changes to Existing Measure for HEDIS 1 2020: Use of Opioids at High Dosage (UOD) NCQA seeks comments on proposed revisions to the Use of Opioids at High Dosage HEDIS measure. The current measure
Secretary for Health and Family Services Selections for Preferred Products
 Secretary for Health and Family Services Selections for Preferred Products This is a summary of the final Preferred Drug List (PDL) selections made by the Secretary for Health and Family Services based
Secretary for Health and Family Services Selections for Preferred Products This is a summary of the final Preferred Drug List (PDL) selections made by the Secretary for Health and Family Services based
Taking Part B Therapy Beyond the $3,700 Threshold New Manual Medical Review Process Effective date October 1, 2012
 Taking Part B Therapy Beyond the $3,700 Threshold New Manual Medical Review Process Effective date October 1, 2012 Presented by: Leigh Ann Frick, PT, MBA Vice President of Clinical Services Heritage Healthcare
Taking Part B Therapy Beyond the $3,700 Threshold New Manual Medical Review Process Effective date October 1, 2012 Presented by: Leigh Ann Frick, PT, MBA Vice President of Clinical Services Heritage Healthcare
A COMPREHENSIVE APPROACH TO MANAGING DIABETES
 A COMPREHENSIVE APPROACH TO MANAGING DIABETES Programs and services to help individuals avoid costly complications and improve outcomes 2 0 1 8 ENTER Supporting each member with targeted guidance and services.
A COMPREHENSIVE APPROACH TO MANAGING DIABETES Programs and services to help individuals avoid costly complications and improve outcomes 2 0 1 8 ENTER Supporting each member with targeted guidance and services.
Cancer Care Ontario / Ontario Public Drug Programs
 Cancer Care Ontario / Ontario Public Drug Programs Policy: Public funding of cancer drugs and their administration within the context of clinical trials Version: 3.0 Effective Date: December 12, 2016 Replaces
Cancer Care Ontario / Ontario Public Drug Programs Policy: Public funding of cancer drugs and their administration within the context of clinical trials Version: 3.0 Effective Date: December 12, 2016 Replaces
Methodologic. Flaw. Metric Discussion Recommendations. ACO Metrics for Recommended Changes. Amb Sensitive Conditions for Admission
 ACO Metrics for Recommended Changes Metric Discussion Recommendations Amb Sensitive Conditions for Admission (9,10) Claims based measures are problematic if diagnosis codes are based on hospital claims
ACO Metrics for Recommended Changes Metric Discussion Recommendations Amb Sensitive Conditions for Admission (9,10) Claims based measures are problematic if diagnosis codes are based on hospital claims
Supplementary Online Content
 Supplementary Online Content Choudhry NK, Krumme AA, Ercole PM, et al. Effect of reminder devices on medication adherence: the REMIND randomized clinical trial. JAMA Int Med. Published online February
Supplementary Online Content Choudhry NK, Krumme AA, Ercole PM, et al. Effect of reminder devices on medication adherence: the REMIND randomized clinical trial. JAMA Int Med. Published online February
Ontario s Narcotics Strategy
 Ontario s Narcotics Strategy Ontario Public Drug Programs Ministry of Health and Long-Term Care January 31, 2012 Ontario Harm Reduction Distribution Program Conference 2012 1 Background The Need for Action
Ontario s Narcotics Strategy Ontario Public Drug Programs Ministry of Health and Long-Term Care January 31, 2012 Ontario Harm Reduction Distribution Program Conference 2012 1 Background The Need for Action
Texas Vendor Drug Program. Formulary Delimited File Layout. April 26, 2017
 Texas Vendor Drug Program Formulary Delimited File Layout April 26, 2017 The Vendor Drug Program provides a weekly update of resource data available for download from txvendordrug.com/resources/downloads.
Texas Vendor Drug Program Formulary Delimited File Layout April 26, 2017 The Vendor Drug Program provides a weekly update of resource data available for download from txvendordrug.com/resources/downloads.
Proposed Changes to Existing Measure for HEDIS : Adherence to Antipsychotic Medications for Individuals With Schizophrenia (SAA)
 Proposed Changes to Existing Measure for HEDIS 1 2020: Adherence to Antipsychotic Medications for Individuals With Schizophrenia (SAA) NCQA seeks comments on proposed modifications to the HEDIS Health
Proposed Changes to Existing Measure for HEDIS 1 2020: Adherence to Antipsychotic Medications for Individuals With Schizophrenia (SAA) NCQA seeks comments on proposed modifications to the HEDIS Health
Patient-Centered Measurement: Innovation Challenge Series
 Patient-Centered Measurement: Innovation Challenge Series Learning Collaborative 2018 Webinar Thursday, March 1, 2018 1 Hala Durrah, MTA Patient Family Engagement Consultant, Speaker & Advocate 3 2017-18
Patient-Centered Measurement: Innovation Challenge Series Learning Collaborative 2018 Webinar Thursday, March 1, 2018 1 Hala Durrah, MTA Patient Family Engagement Consultant, Speaker & Advocate 3 2017-18
Low Back Pain Report October 2013: Cost and Utilization of Health Care in Oregon
 Low Back Pain Report October 2013: Cost and Utilization of Health Care in Oregon INTRODUCTION Most people in the United States will experience low back pain at least once during their lives. According
Low Back Pain Report October 2013: Cost and Utilization of Health Care in Oregon INTRODUCTION Most people in the United States will experience low back pain at least once during their lives. According
