The Influence of Light on Temperature Preference in Drosophila
|
|
|
- Shauna Bryan
- 6 years ago
- Views:
Transcription
1 Report The Influence of Light on Temperature Preference in Drosophila Highlights d Light positively influences temperature preference behavior in flies Authors Lauren M. Head, Xin Tang,..., Paul A. Garrity, Fumika N. Hamada d d d LDTP is controlled separately from the circadian clock PDFR acts in DN1 p s to control LDTP Response to acute light on temperature regulation may be conserved evolutionarily Correspondence fumika.hamada@cchmc.org In Brief Head et al. show the direct effect of light on temperature preference behavior (LDTP) in flies. LDTP is controlled by dorsal posterior clock neurons but separately from the circadian clock. Human body temperature increases during the night in response to light. Thus, the effect of light on temperature regulation may be conserved from flies to humans. Head et al., 201, Current Biology 2, April 20, 201 ª201 Elsevier Ltd All rights reserved
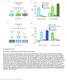
2 Current Biology Report The Influence of Light on Temperature Preference in Drosophila Lauren M. Head, 1, Xin Tang, 1, Sean E. Hayley, 1 Tadahiro Goda, 1 Yujiro Umezaki, 1 Elaine C. Chang, 3 Jennifer R. Leslie, 1 Mana Fujiwara, 1 Paul A. Garrity, 3 and Fumika N. Hamada 1,2,4,, 1 Visual Systems Group, Abrahamson Pediatric Eye Institute, Division of Pediatric Ophthalmology, Cincinnati Children s Hospital Medical Center, 3333 Burnet Avenue, Cincinnati, OH 4, USA 2 Japan Science and Technology Agency, PRESTO, 41 Honcho Kawaguchi, Saitama , Japan 3 Department of Biology, National Center for Behavioral Genomics and Volen Center for Complex Systems, Brandeis University, 41 South Street, Waltham, MA 0, USA 4 Division of Developmental Biology, Cincinnati Children s Hospital Medical Center, Cincinnati, OH 4, USA Department of Ophthalmology, College of Medicine, University of Cincinnati, Cincinnati, OH 4, USA Cofirst author Correspondence: fumika.hamada@cchmc.org SUMMARY Ambient light affects multiple physiological functions and behaviors, such as circadian rhythms, sleepwake activities, and development, from flies to mammals [1 ]. Mammals exhibit a higher body temperature when exposed to acute light compared to when they are exposed to the dark, but the underlying mechanisms are largely unknown [ 10]. The body temperature of small ectotherms, such as Drosophila, relies on the temperature of their surrounding environment, and these animals exhibit a robust temperature preference behavior [11 13]. Here, we demonstrate that Drosophila prefer a 1 higher temperature when exposed to acute light rather than the dark. This acute light response, lightdependent temperature preference (LDTP), was observed regardless of the time of day, suggesting that LDTP is regulated separately from the circadian clock. However, screening of eye and circadian clock mutants suggests that the circadian clock neurons posterior dorsal neurons 1 (DN1 p s) and PigmentDispersing Factor Receptor (PDFR) play a role in LDTP. To further investigate the role of DN1 p s in LDTP, PDFR in DN1 p s was knocked down, resulting in an abnormal LDTP. The phenotype of the pdfr mutant was rescued sufficiently by expressing PDFR in DN1 p s, indicating that PDFR in DN1 p s is responsible for LDTP. These results suggest that light positively influences temperature preference via the circadian clock neurons, DN1 p s, which may result from the integration of light and temperature information. Given that both Drosophila and mammals respond to acute light by increasing their body temperature, the effect of acute light on temperature regulation may be conserved evolutionarily between flies and humans. RESULTS Acute Light Positively Influences Temperature Preference in Drosophila Drosophila exhibit robust temperature preference behavior. Flies not only avoid noxious temperatures [11, 12, 14 1], but they also exhibit a temperature preference rhythm (TPR) in which preferred temperature is lower in the morning and higher at dusk [1]. We previously observed that flies entrained with light and dark (LD) cycles prefer a higher temperature than flies in free run (constant darkness [DD]) during the daytime [1], suggesting that acute light may affect the temperature preference in flies. To determine whether acute light influences the selection of preferred temperature in Drosophila, we performed behavioral experiments to compare their preferred temperatures when ambient light was on verses when ambient light was off. We found that wildtype (w 111 : WT) flies preferred 1 C higher temperature in the light compared to their temperature preference in the dark (Figure 1A), suggesting that acute light positively influences the selection of preferred temperature. We refer to this behavior as lightdependent temperature preference (LDTP) and investigated the neural circuits that regulate this behavior. LDTP Is Controlled Separately from the Circadian Clock To determine whether LDTP is observed regardless of the time of day, we tested the temperature preference behavior at different time points throughout the day (Figures 1A and 1B). We found that the flies consistently preferred a higher temperature throughout the day when the behavioral assays were performed in the light (Figures 1A and 1B). In the same way, the flies consistently preferred a lower temperature throughout the day when the behavioral assays were performed in the dark, although preferred temperatures at zeitgeber time (ZT) 1 were similar (Figure 1B). These data suggest that LDTP occurs irrespective of the circadian clock. To confirm that LDTP is independent of circadian clock function, we examined LDTP in mutants for period (per) and timeless (tim), which disrupt the circadian clock. If LDTP is independent of the circadian clock, per 01 and tim 01 mutants should still exhibit LDTP. We found that per 01 and tim 01 mutants exhibited a normal Current Biology 2, , April 20, 201 ª201 Elsevier Ltd All rights reserved 103
3 A C w 111 light w 111 dark 1012 tim 01 light tim 01 dark B w 111 light w 111 dark LDTP and preferred a higher temperature in the light than in the dark at all time points throughout the daytime (Figures 1C and 1D), with the exception of the tim 01 mutants at ZT4. At this time point, the tim 01 mutants preferred a slightly higher temperature in the light, but the difference was not statistically significant. Thus, we concluded that LDTP is regulated separately from the circadian clock and is dependent solely on light. glass Is Required for LDTP To investigate the neural circuits that regulate LDTP, we first examined the effect of eye components on LDTP. Flies have seven eye components: two compound eyes, three ocelli, and two HofbauerBuchner (HB) eyelets [1]. Subsets of eye components are abnormal in the mutant fly strains eyes absent (eya 1 ), sine oculis (so 1 ), histidine decarboxylase (hdc JK10 ), and glass (gl 0j ), and in flies in which the proapoptosis gene hid is expressed under the control of a glass multimer response element (GMRhid)[20, ](Figure 2G). We found that eya 1, so 1, hdc JK10, and GMRhid mutants all showed normal LDTP, preferring a higher temperature in the light compared to the dark (Figure 2A). These data suggest that abnormalities in the compound eyes, ocelli, and HB eyelets do not disrupt LDTP and thus, these eye components are not essential for LDTP per 01 light per 01 dark Figure 1. Acute Light Positively Influences Temperature Preference in Drosophila (A and B) Comparison of preferred temperature between light (gray line) and dark (black line) conditions for w 111 flies during the daytime (A) or nighttime (B). w 111 flies were raised in LD (light [12 hr]dark [12 hr]) cycles. Ambient light was either on or off when the behavioral experiments were performed for 30 min. (C and D) Comparison of preferred temperature between light (gray line) and dark (black line) conditions for tim 01 (C) and per 01 (D) during the daytime. tim 01 (C) and per 01 (D) flies were raised in LD. Italicized numbers represent the number of assays. ZT, zeitgeber time (ZT0 is lights on, ZT12 is lights off). The same behavioral data in light (A), dark (B), light (C), and light (D) from [1] are used. t test compared preferred temperature between light and dark conditions: p < 0.001, p < 0.01, or p < 0.0. Error bars indicate the SEM. D However, we found that a null allele of glass, gl 0j, had abnormal LDTP, preferring a higher temperature in the dark than in the light. Even the weak lossoffunction alleles of glass, gl 1, gl 2, and gl 3 [], had abnormal LDTP, in which the flies preferred a similar temperature in the light and dark (Figure 2A). To determine whether glass is responsible for LDTP, we used the 10kb genomic glass minigene to rescue the glass mutants []. Both of the gl(10kb); gl 3 and gl(10kb); gl 0j flies preferred significantly higher temperatures in the light than in the dark, indicating that the normal LDTP was restored and that glass function is required for LDTP. Interestingly, the gl 0j mutants not only have abnormal eye components but also lack a subset of circadian clock cells, the posterior dorsal neurons 1 (DN1 p s) (Figure 2G). Previous studies show that glass is expressed in DN1 p s, but not in the anterior DN1s, DN1 a s[, ]. To confirm that Glass is expressed in DN1 p s, we used the DN1 p s driver, Clk4.FGal4 [, 2], to label DN1 p s in the brain (Figure 2B). We performed immunostaining on the UASmCD::GFP;Clk4.FGal4 (Clk4.FGal4>UASmCD::GFP) flies using the Glass antibody and confirmed that Glass is expressed in the DN1 p s(figure 2B). Conversely, we found that Clk4.FGal4>UASmCD::GFP signals were not detected in gl 0j /gl 0j mutants (Figure 2D) but were still present in the gl 0j heterozygous control (Figure 2C), indicating that DN1 p s were ablated in the gl 0j mutants. If DN1 p s are key neurons for LDTP, DN1 p s should be restored in the gl(10kb); gl 0j flies given that gl(10kb); gl 0j flies exhibit a normal LDTP (Figure 2A). To determine this, we performed immunostaining using the Timeless (TIM) antibody and found that DN1 p s were restored in gl(10kb); gl 0j flies (Figure 2E). In addition, as a control, we confirmed that DN1 p s were present in GMRhid flies (Figure 2F). These data suggested that the DN1 p s may be critical for LDTP. TRPA1 and Rhodopsin 1 Are Not Necessary for LDTP Transient receptor potential A 1 (TrpA1) is important for temperature preference behavior as flies use TRPA1 to detect and avoid warm temperatures. TRPA1 not only is a warm sensor in both larvae and adult flies [12, ], but also is involved in lightsensing behavior in the body wall of larvae []. Furthermore, Rhodopsin 1 (Rh1), encoded by the neither inactivation nor afterpotential E (ninae) gene, is a molecular light sensor and has been suggested to regulate temperaturesensing behavior in larvae [2]. Therefore, we sought to determine whether TRPA1 and Rh1 were involved in LDTP by using strong lossoffunction mutants for TrpA1 (TrpA1 ins )[12] and null mutants for ninae (ninae 1 ) [2] (Figure 3A). However, both mutants showed normal LDTP, indicating that TRPA1 and Rh1 are not necessary for LDTP. LN v s Are Dispensable for LDTP The clock neurons, small ventrolateral neurons (sln v s), project to DN1s [2, 30]. sln v s not only contact DN1 p s but also receive information from the light sensors, large ventrolateral neurons (lln v s) [31 33], and receive light inputs from the optic lobe [34]. To determine whether LN v s are involved in LDTP, we used a mammalian inward rectifier K + channel (UASKir) to genetically inhibit sln v s with RGal4 [34] as well as sln v s and lln v s with Mz20Gal4 [3, 3]. Because RGal4/UASKir flies did not survive to adulthood, we used a temperaturedependent 104 Current Biology 2, , April 20, 201 ª201 Elsevier Ltd All rights reserved
4 A light dark w 111 eya DN1p Glass Clk4.FG4::GFP gl 0j DN1p so 1 GMRhid gl 1 hdc JK10 DN1p B1 B2 B3 C gl(10kb);gl 0j DN3 E DN1 L<D gl 2 gl 3 conditional repressor of Gal4, tubgal0 ts, to transiently inhibit the LN v s depending on the permissive temperature (1 C) and the restrictive temperature (2 C). However, at both permissive and restrictive temperatures, the RGal4 and Mz20Gal4 with UASKir; tubgal0 ts flies exhibited normal LDTP (Figure 3B), suggesting that LNvs are not important for LDTP. As a positive control using locomotor activity, we showed that Mz20Gal4 with UASKir; tubgal0 ts flies exhibited abnormal rhythmicity at 2 C but normal rhythmicity at 1 C(Figure S1 and Table S1). PDFR Acts in DN1 p s to Control LDTP Our data suggest that DN1 p s are critical for LDTP. Although the persistence of LDTP in per and tim mutants indicates that this behavior does not require a functional circadian clock (Figures 1C and 1D), the DN1 p s participate in circadian clock function and thus express many clock genes. To determine which molecules might act within the DN1 p cells to control LDTP, we examined the involvement of additional clock genes, including cryptochrome (cry), Clock (Clk), and pigmentdispersing factor receptor (pdfr), and tested LDTP of mutations in the following genes: cry b, cry 01, cry 02, Clk Jrk, pdfr 304, and pdfr 33 (Figure 4A). Like per 01 and tim 01 mutants, cry b, cry 01, cry 02, and Clk Jrk mutants all preferred a higher temperature in the light than in the dark, although cry 01 preferred a much lower temperature in the dark. Interestingly, we found that pdfr 304 and pdfr 33 mutants Clk4.FG4::GFP TIM gl 0j /gl 0j D Clk4.FG4::GFP GMRhid DN3 F gl 0j DN1p Glass Clk4.FG4::GFP DN1 TIM G compound eyes ocelli HB eyelets DN1 p s gl(10kb) ; gl 3 eya 1 so 1 hdc JK10 gl(10kb) ; gl 0j gl 0j GMRhid Figure 2. glass Is Required for LDTP (A) Comparison of preferred temperature of each genotype in light (white bar) and dark (gray bar) conditions. The behavioral experiments were performed at ZT1 3. t test compared preferred temperature between light and dark conditions: p < 0.001, p < 0.01, or p < 0.0. Italicized numbers represent the number of assays. Error bars indicate the SEM. (B1 B3) The Clk4.FGal4>UASmCD::GFP brains were stained with antigfp (green) and antiglass (red). The GFP signals, labeled by Clk4.FGal4>UASmCD::GFP, overlap with the signals labeled by antiglass (B1). Arrowheads show the cells for which antigfp and antiglass overlapped (B1 B3). (C and D) The DN1 p s labeled by Clk4.FGal4>UASmCD::GFP with antigfp (green) were still present in the gl 0j heterozygous control (C) but were not detected in gl 0j /gl 0j mutants (D). (E and F) The gl(10kb);gl 0j (E) and GMRhid (F) brains were stained with antitim (red). Arrowheads show the DN1s. (G) A summary of eye and DN1 p s phenotypes for each eye mutant fly line. Plus sign (+) indicates normal; minus sign ( ) indicates abnormal. displayed abnormal LDTP, in which they preferred similar temperatures in the light and the dark, suggesting that pdfr is required for LDTP (Figure 4A). PDFR is a G proteincoupled receptor and is critical for locomotor activity and synchronization of the circadian clock [3 40]. To determine whether PDFR expression in DN1 p s is necessary for LDTP, we knocked down PDFR in DN1 p s by using UASpdfrRNAi with Clk4.FGal4, which is selectively expressed in subsets of DN1 p s(figure 4B). Clk4.FGal4/UASpdfrRNAi flies exhibited an abnormal LDTP, showing similar preferred temperatures in the light and the dark. However, each Gal4 and UAS control fly line exhibited a normal LDTP, indicating that PDFR expression in DN1 p s is necessary for LDTP (Figure 4B). To determine whether PDFR expression in DN1 p s is sufficient to rescue the pdfr 304 mutants phenotype, we expressed UASpdfr using Clk4.FGal4 in the pdfr 304 mutants. The pdfr 304 flies that expressed PDFR in DN1 p s preferred a higher temperature in the light than in the dark, while the control flies did not, indicating that PDFR expression in DN1 p s restored LDTP of pdfr 304 mutants. Thus, PDFR expression in DN1 p s is necessary and sufficient to support the role of PDFR in LDTP (Figure 4C). Because PigmentDispersing Factor (PDF) and Diuretic Hormone 31 (DH31) activate PDFR in vitro [3], we examined whether PDF and DH31 are involved in LDTP. We used the pdf null mutant, pdf 01, and the Dh31 mutant, Dh31 #1, which was generated by P element excision. Dh31 #1 is a strong lossoffunction mutation, as it contains a deletion of the entire active peptide of DH31 (Figures S2 and S3). Nonetheless, both pdf 01 and Dh31 #1 and even the double mutant of Dh31 #1 ; pdf 01 Current Biology 2, , April 20, 201 ª201 Elsevier Ltd All rights reserved 10
5 A B exhibited a normal LDTP, indicating that PDF and DH31 are not required for LDTP. These results suggest that LDTP mediated by PDFR in DN1 p s is not due to the pathway activated by these known neuropeptides. DISCUSSION TrpA1 ins 10 1 C 2 C 4 ninae ninae 1 RGal4/ UASKir;tubG0 ts light dark 1 C 2 C Mz20Gal4/ UASKir;tubG0 ts Figure 3. TrpA1, Rhodopsin 1, and slnvs Are Not Critical for LDTP (A) Comparison of preferred temperature of each genotype in light (white bar) and dark (gray bar) conditions. The behavioral experiments were performed at ZT1 3. t test compared preferred temperature between light and dark conditions: p < 0.001, p < 0.01, or p < 0.0. Italicized numbers represent the number of assays. Error bars indicate the SEM. (B) Comparison of preferred temperature of each genotype in light (white bar) and dark (gray bar) conditions. RGal4 is expressed in slnvs, and Mz20 Gal4 is expressed in both slnvs and llnvs. A temperaturedependent conditional repressor of Gal4, tubgal0 ts, and a mammalian inward rectifier K + channel, Kir, were used to transiently inhibit the slnvs depending on permissive temperature (1 C) and restrictive temperature (2 C) as adults. The behavioral experiments were performed at ZT1 3. t test compared preferred temperature between light and dark conditions: p < 0.001, p < 0.01, or p < 0.0. Italicized numbers represent the number of assays. Error bars indicate the SEM. Here, we show that acute light positively affects temperature preference in Drosophila. LDTP is controlled by PDFRexpressing DN1 p s, independently from the circadian clock, suggesting DN1 p s play an important role in integrating light and temperature information. Although we tested several eye component mutants, abnormal light or temperaturesensing mutants, and cry mutants, these A per 01 tim 01 cry b B C 14 pdfr cry 01 cry02 Clk4.FGal4 UASpdfrRNAi Clk4.FGal4 /UASpdfrRNAi w 111 pdfr 304 pdfr 33 pdfr 304 Clk4.FGal4 /UASpdfr UASpdfr Clk4.FGal Clk Jrk pdfr 304 pdfr mutants still exhibited LDTP behavior. Because light sensors can be redundant in the eye and body wall, partial disruption of these light sensors may not be sufficient for abnormal LDTP (Figure 2). In fact, the double mutants of GMRhid; cry 01, which lack the functions of the compound eye, ocelli, HB eyelet, and CRY, exhibited an abnormal LDTP (Figure 4A). This result suggests that 1 11 pdf 01 Dh31 #1 Dh31 #1 ;pdf light dark GMRhid ;cry 01 Figure 4. PDFR in DN1 p s Is Necessary and Sufficient for LDTP (A) Comparison of preferred temperature of circadian clock mutants in light (white bar) and dark (gray bar) conditions is shown. The behavioral experiments were performed at ZT1 3. t test compared preferred temperature between light and dark conditions: p < 0.001, p < 0.01, or p < 0.0. Italicized numbers represent the number of assays. Error bars indicate the SEM. (B) PDFR expression in DN1 p s is necessary for LDTP. Comparison of preferred temperature for flies with UASpdfrRNAi in a subset of clock cells using a DN1 p s driver, Clk4.FGal4, and its controls is shown. The behavioral experiments were performed at ZT1 3. t test compared preferred temperature between light and dark conditions: p < 0.001, p < 0.01, or p < 0.0. Italicized numbers represent the number of assays. Error bars indicate the SEM. (C) PDFR expression in DN1 p s is sufficient for LDTP. Comparison of preferred temperature of pdfr 304 mutants with UASpdfr in a subset of clock cells using a DN1 p s driver, Clk4.FGal4, and its controls is shown. The behavioral experiments were performed at ZT1 3. t test compared preferred temperature between light and dark conditions: p < 0.001, p < 0.01, or p < 0.0. Italicized numbers represent the number of assays. Error bars indicate the SEM. 10 Current Biology 2, , April 20, 201 ª201 Elsevier Ltd All rights reserved
6 at least two pathways, such as the visual system and cry, act together to mediate light detection and play an important role in LDTP. Notably, in humans, 40nm light is important for an increase in body temperature during the night [10]. Therefore, it would be interesting to examine which light pathway and wavelengths are critical for LDTP. Although we show that LDTP is circadian clock independent, PDFR expression in DN1 p s is critical for LDTP (Figure 4). However, it is unclear how PDFR is activated because neither PDF nor DH31, the ligands of PDFR, are important for LDTP (Figure 4). Therefore, our data suggest that PDFR in DN1 p s is activated by other unknown mechanisms responsible for LDTP. One possible mechanism is through CRY because CRY is expressed in the clock cells, including DN1 p s, and has convergent roles with PDFR for the circadian rhythm of locomotor activity [41]. CRY also antagonizes the temperature synchronization in the dorsal neurons, suggesting that CRY may be involved in the integration of light and temperature [42]. Therefore, it is possible that CRY and PDFR work to regulate LDTP. For example, DN1 p s may directly receive light input via CRY, which regulates the signal cascade of PDFR. Light is critical not only for entraining the circadian clock, but also for a behavior termed masking, in which the flies exhibit a robust increase of locomotor activity after light is turned on or off [43]. The masking effect is controlled separately from the circadian clock [20, ], and DN1 p s are involved in a masking effect for locomotor activity when light is on []. Given that light positively affects preferred temperature separately from the circadian clock, LDTP could be part of the masking effect. However, the light input pathways for the masking effect in locomotor activity and LDTP are not the same. This is demonstrated through evidence that shows that disruption of the compound eye is sufficient for the masking effect of locomotor activity [1], but not for LDTP (Figure 2). Furthermore, the molecular mechanisms controlling the masking effect in locomotor activity and LDTP are different, as pdfr mutants exhibit a normal masking effect for locomotor activity [20] but an abnormal LDTP (Figure 4). Therefore, our data indicate that the masking effects of locomotor activity and LDTP are controlled differently. Here, we show the positive effect of acute light on the preferred temperature in flies. Given that flies adapt their body temperature to ambient temperature [13], the flies body temperature increases in light as a result of their temperature preference behavior. In humans, light exposure increases body temperature during the nighttime [ ] and is dependent on light intensity [10]. Whereas humans control body temperature through the generation of heat, ectotherms use behavioral strategies to regulate body temperature [13]. Although the mechanism of heat generation is different between humans and flies, the body temperature of both humans and flies increases when exposed to light. Thus, we propose that the effect of light on temperature regulation may be evolutionarily conserved from flies to humans. SUPPLEMENTAL INFORMATION Supplemental Information includes Supplemental Experimental Procedures, three figures, and one table and can be found with this article online at AUTHOR CONTRIBUTIONS F.N.H., L.M.H., and X.T. designed the research. F.N.H., L.M.H., X.T., Y.U., J.R.L., M.F., T.G., and S.E.H. performed the behavioral experiments. X.T. performed immunostaining. F.N.H., E.C.C., and P.A.G. created Dh31 mutants. F.N.H., L.M.H., and X.T. wrote the manuscript. ACKNOWLEDGMENTS We are grateful to Dr. Patrick Emery for the Clk4.FGal4, cry 02, and cry b flies; Dr. Paul Taghert for the Pdf 01, pdfr 304, and pdfr 304 ; UASpdfr flies; Dr. Orie Shafer for RGal4; Dr. Charlotte HelfrichFörster for Mz20Gal4; Dr. Michael Rosbash for the TIM antibody; Dr. Hugo Bellen for hdc JK10 ; and the Bloomington Drosophila Stock Center, Vienna Drosophila RNAi Center, and Developmental Studies Hybridoma Bank for the fly lines and antibodies. We thank the F.N.H. laboratory members for their comments and advice on the manuscript. This research was supported by a Trustee Grant and RIP funding from Cincinnati Children s Hospital, the Precursory Research for Embryonic Science and Technology (PRESTO) from Japan Science and Technology (JST), the March of Dimes, NIH R01 GM102 to F.N.H., and NIH P01 GM1030 to P.A.G. Received: June 30, 2014 Revised: January 12, 201 Accepted: February 13, 201 Published: April, 201 REFERENCES 1. Sehgal, A., and Mignot, E. (2011). Genetics of sleep and sleep disorders. Cell 14, Rao, S., Chun, C., Fan, J., Kofron, J.M., Yang, M.B., Hegde, R.S., Ferrara, N., Copenhagen, D.R., and Lang, R.A. (2013). A direct and melanopsindependent fetal light response regulates mouse eye development. Nature 44, LeGates, T.A., Altimus, C.M., Wang, H., Lee, H.K., Yang, S., Zhao, H., Kirkwood, A., Weber, E.T., and Hattar, S. (2012). Aberrant light directly impairs mood and learning through melanopsinexpressing neurons. Nature 41, Yuan, Q., Xiang, Y., Yan, Z., Han, C., Jan, L.Y., and Jan, Y.N. (2011). Lightinduced structural and functional plasticity in Drosophila larval visual system. Science 333, Xiang, Y., Yuan, Q., Vogt, N., Looger, L.L., Jan, L.Y., and Jan, Y.N. (2010). Lightavoidancemediating photoreceptors tile the Drosophila larval body wall. Nature 4,.. Mazzoni, E.O., Desplan, C., and Blau, J. (200). Circadian pacemaker neurons transmit and modulate visual information to control a rapid behavioral response. Neuron 4, Myers, B.L., and Badia, P. (13). Immediate effects of different light intensities on body temperature and alertness. Physiol. Behav. 4, Cajochen, C., Zeitzer, J.M., Czeisler, C.A., and Dijk, D.J. (2000). Doseresponse relationship for light intensity and ocular and electroencephalographic correlates of human alertness. Behav. Brain Res. 11, 3.. Dijk, D.J., Cajochen, C., and Borbély, A.A. (11). Effect of a single 3hour exposure to bright light on core body temperature and sleep in humans. Neurosci. Lett. 1, Cajochen, C., Münch, M., Kobialka, S., Kräuchi, K., Steiner, R., Oelhafen, P., Orgül, S., and WirzJustice, A. (200). High sensitivity of human melatonin, alertness, thermoregulation, and heart rate to short wavelength light. J. Clin. Endocrinol. Metab. 0, Hong, S.T., Bang, S., Hyun, S., Kang, J., Jeong, K., Paik, D., Chung, J., and Kim, J. (200). camp signalling in mushroom bodies modulates temperature preference behaviour in Drosophila. Nature 44, Hamada, F.N., Rosenzweig, M., Kang, K., Pulver, S.R., Ghezzi, A., Jegla, T.J., and Garrity, P.A. (200). An internal thermal sensor controlling temperature preference in Drosophila. Nature 44, 0. Current Biology 2, , April 20, 201 ª201 Elsevier Ltd All rights reserved 10
7 13. Stevenson, R.D. (1). The relative importance of behavioral and physiological adjustments controlling body temperature in terrestrial ectotherms. Am. Nat. 1, Garrity, P.A., Goodman, M.B., Samuel, A.D., and Sengupta, P. (2010). Running hot and cold: behavioral strategies, neural circuits, and the molecular machinery for thermotaxis in C. elegans and Drosophila. Genes Dev., Tomchik, S.M. (2013). Dopaminergic neurons encode a distributed, asymmetric representation of temperature in Drosophila. J. Neurosci. 33, a. 1. Ueno, T., Tomita, J., Kume, S., and Kume, K. (2012). Dopamine modulates metabolic rate and temperature sensitivity in Drosophila melanogaster. PLoS ONE, e Bang, S., Hyun, S., Hong, S.T., Kang, J., Jeong, K., Park, J.J., Choe, J., and Chung, J. (2011). Dopamine signalling in mushroom bodies regulates temperaturepreference behaviour in Drosophila. PLoS Genet., e Kaneko, H., Head, L.M., Ling, J., Tang, X., Liu, Y., Hardin, P.E., Emery, P., and Hamada, F.N. (2012). Circadian rhythm of temperature preference and its neural control in Drosophila. Curr. Biol., Buschbeck, E.K., and Friedrich, M. (200). Evolution of Insect Eyes: Tales of Ancient Heritage, Deconstruction, Reconstruction, Remodeling, and Recycling. Evo. Edu. Outreach 1, Rieger, D., Stanewsky, R., and HelfrichFörster, C. (2003). Cryptochrome, compound eyes, HofbauerBuchner eyelets, and ocelli play different roles in the entrainment and masking pathway of the locomotor activity rhythm in the fruit fly Drosophila melanogaster. J. Biol. Rhythms 1, Klarsfeld, A., Malpel, S., MichardVanhée, C., Picot, M., Chélot, E., and Rouyer, F. (2004). Novel features of cryptochromemediated photoreception in the brain circadian clock of Drosophila. J. Neurosci., Moses, K., Ellis, M.C., and Rubin, G.M. (1). The glass gene encodes a zincfinger protein required by Drosophila photoreceptor cells. Nature 340, Shafer, O.T., HelfrichFörster, C., Renn, S.C., and Taghert, P.H. (200). Reevaluation of Drosophila melanogaster s neuronal circadian pacemakers reveals new neuronal classes. J. Comp. Neurol. 4, Zhang, L., Chung, B.Y., Lear, B.C., Kilman, V.L., Liu, Y., Mahesh, G., Meissner, R.A., Hardin, P.E., and Allada, R. (2010). DN1(p) circadian neurons coordinate acute light and PDF inputs to produce robust daily behavior in Drosophila. Curr. Biol. 20, Zhang, Y., Liu, Y., BilodeauWentworth, D., Hardin, P.E., and Emery, P. (2010). Light and temperature control the contribution of specific DN1 neurons to Drosophila circadian behavior. Curr. Biol. 20, Rosenzweig, M., Brennan, K.M., Tayler, T.D., Phelps, P.O., Patapoutian, A., and Garrity, P.A. (200). The Drosophila ortholog of vertebrate TRPA1 regulates thermotaxis. Genes Dev. 1, Shen, W.L., Kwon, Y., Adegbola, A.A., Luo, J., Chess, A., and Montell, C. (2011). Function of rhodopsin in temperature discrimination in Drosophila. Science 331, O Tousa, J.E., Baehr, W., Martin, R.L., Hirsh, J., Pak, W.L., and Applebury, M.L. (1). The Drosophila ninae gene encodes an opsin. Cell 40, HelfrichFörster, C. (2003). The neuroarchitecture of the circadian clock in the brain of Drosophila melanogaster. Microsc. Res. Tech. 2, Cavanaugh, D.J., Geratowski, J.D., Wooltorton, J.R., Spaethling, J.M., Hector, C.E., Zheng, X., Johnson, E.C., Eberwine, J.H., and Sehgal, A. (2014). Identification of a circadian output circuit for rest:activity rhythms in Drosophila. Cell 1, Sheeba, V., Gu, H., Sharma, V.K., O Dowd, D.K., and Holmes, T.C. (200). Circadian and lightdependent regulation of resting membrane potential and spontaneous action potential firing of Drosophila circadian pacemaker neurons. J. Neurophysiol.,. 32. Fogle, K.J., Parson, K.G., Dahm, N.A., and Holmes, T.C. (2011). CRYPTOCHROME is a bluelight sensor that regulates neuronal firing rate. Science 331, Parisky, K.M., Agosto, J., Pulver, S.R., Shang, Y., Kuklin, E., Hodge, J.J., Kang, K., Liu, X., Garrity, P.A., Rosbash, M., and Griffith, L.C. (200). PDF cells are a GABAresponsive wakepromoting component of the Drosophila sleep circuit. Neuron 0, HelfrichFörster, C., Shafer, O.T., Wülbeck, C., Grieshaber, E., Rieger, D., and Taghert, P. (200). Development and morphology of the clockgeneexpressing lateral neurons of Drosophila melanogaster. J. Comp. Neurol. 00, Grima, B., Chélot, E., Xia, R., and Rouyer, F. (2004). Morning and evening peaks of activity rely on different clock neurons of the Drosophila brain. Nature 431, Fujii, S., and Amrein, H. (2010). Ventral lateral and DN1 clock neurons mediate distinct properties of male sex drive rhythm in Drosophila. Proc. Natl. Acad. Sci. USA 10, Mertens, I., Vandingenen, A., Johnson, E.C., Shafer, O.T., Li, W., Trigg, J.S., De Loof, A., Schoofs, L., and Taghert, P.H. (200). PDF receptor signaling in Drosophila contributes to both circadian and geotactic behaviors. Neuron 4, Lear, B.C., Merrill, C.E., Lin, J.M., Schroeder, A., Zhang, L., and Allada, R. (200). A G proteincoupled receptor, groomofpdf, is required for PDF neuron action in circadian behavior. Neuron 4, Hyun, S., Lee, Y., Hong, S.T., Bang, S., Paik, D., Kang, J., Shin, J., Lee, J., Jeon, K., Hwang, S., et al. (200). Drosophila GPCR Han is a receptor for the circadian clock neuropeptide PDF. Neuron 4, Taghert, P.H., and Nitabach, M.N. (2012). Peptide neuromodulation in invertebrate model systems. Neuron, Im, S.H., Li, W., and Taghert, P.H. (2011). PDFR and CRY signaling converge in a subset of clock neurons to modulate the amplitude and phase of circadian behavior in Drosophila. PLoS ONE, e Gentile, C., Sehadova, H., Simoni, A., Chen, C., and Stanewsky, R. (2013). Cryptochrome antagonizes synchronization of Drosophila s circadian clock to temperature cycles. Curr. Biol., Page, T.L. (1). Masking in invertebrates. Chronobiol. Int., Current Biology 2, , April 20, 201 ª201 Elsevier Ltd All rights reserved
Report. Circadian Rhythm of Temperature Preference and Its Neural Control in Drosophila
 Current Biology, 1, October, 2 ª2 Elsevier td All rights reserved http://dx.doi.org/1./j.cub.2.. Circadian Rhythm of Temperature Preference and Its Neural Control in rosophila Report Haruna Kaneko, 1 auren
Current Biology, 1, October, 2 ª2 Elsevier td All rights reserved http://dx.doi.org/1./j.cub.2.. Circadian Rhythm of Temperature Preference and Its Neural Control in rosophila Report Haruna Kaneko, 1 auren
Interactions between circadian neurons control temperature synchronization of Drosophila behavior
 University of Massachusetts Medical School escholarship@umms GSBS Student Publications Graduate School of Biomedical Sciences 2007-10-05 Interactions between circadian neurons control temperature synchronization
University of Massachusetts Medical School escholarship@umms GSBS Student Publications Graduate School of Biomedical Sciences 2007-10-05 Interactions between circadian neurons control temperature synchronization
The Drosophila melanogaster life cycle
 The Drosophila melanogaster life cycle Eclosion The first phenotype described in Drosophila as an endogenous rhythm (1954): Number of eclosed flies Hamblen et al., Gene3cs 1998 rhythm with a periodicity
The Drosophila melanogaster life cycle Eclosion The first phenotype described in Drosophila as an endogenous rhythm (1954): Number of eclosed flies Hamblen et al., Gene3cs 1998 rhythm with a periodicity
Article. Cryptochrome Antagonizes Synchronization of Drosophila s Circadian Clock to Temperature Cycles
 Current Biology 23, 185 195, February 4, 2013 ª2013 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2012.12.023 Cryptochrome Antagonizes Synchronization of Drosophila s Circadian Clock
Current Biology 23, 185 195, February 4, 2013 ª2013 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2012.12.023 Cryptochrome Antagonizes Synchronization of Drosophila s Circadian Clock
A Role for Blind DN2 Clock Neurons in Temperature Entrainment of the Drosophila Larval Brain
 8312 The Journal of Neuroscience, July 1, 2009 29(26):8312 8320 Cellular/Molecular A Role for Blind DN2 Clock Neurons in Temperature Entrainment of the Drosophila Larval Brain Marie Picot,* André Klarsfeld,*
8312 The Journal of Neuroscience, July 1, 2009 29(26):8312 8320 Cellular/Molecular A Role for Blind DN2 Clock Neurons in Temperature Entrainment of the Drosophila Larval Brain Marie Picot,* André Klarsfeld,*
Endogenous circadian clocks prepare organisms according to
 Moonlight shifts the endogenous clock of Drosophila melanogaster Wolfgang Bachleitner, Lena Kempinger, Corinna Wülbeck, Dirk Rieger, and Charlotte Helfrich-Förster* University of Regensburg, Institute
Moonlight shifts the endogenous clock of Drosophila melanogaster Wolfgang Bachleitner, Lena Kempinger, Corinna Wülbeck, Dirk Rieger, and Charlotte Helfrich-Förster* University of Regensburg, Institute
Organization of the Drosophila Circadian Control Circuit
 Current Biology 18, R84 R93, January 22, 2008 ª2008 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2007.11.061 Organization of the Drosophila Circadian Control Circuit Review Michael N. Nitabach 1
Current Biology 18, R84 R93, January 22, 2008 ª2008 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2007.11.061 Organization of the Drosophila Circadian Control Circuit Review Michael N. Nitabach 1
The period (per) gene in Drosophila melanogaster plays a central
 Seasonal behavior in Drosophila melanogaster requires the photoreceptors, the circadian clock, and phospholipase C B. H. Collins*, E. Rosato, and C. P. Kyriacou* Departments of *Genetics and Biology, University
Seasonal behavior in Drosophila melanogaster requires the photoreceptors, the circadian clock, and phospholipase C B. H. Collins*, E. Rosato, and C. P. Kyriacou* Departments of *Genetics and Biology, University
RNA-interference knockdown of Drosophila pigment dispersing factor in neuronal subsets: The anatomical basis of a neuropeptide s circadian functions
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 2009 RNA-interference knockdown of Drosophila pigment dispersing factor in neuronal subsets: The anatomical basis
Washington University School of Medicine Digital Commons@Becker Open Access Publications 2009 RNA-interference knockdown of Drosophila pigment dispersing factor in neuronal subsets: The anatomical basis
The CREB-binding protein affects the circadian regulation of behaviour
 The CREB-binding protein affects the circadian regulation of behaviour Christian Maurer, Tobias Winter, Siwei Chen, Hsiu-Cheng Hung and Frank Weber Biochemistry Center Heidelberg, University of Heidelberg,
The CREB-binding protein affects the circadian regulation of behaviour Christian Maurer, Tobias Winter, Siwei Chen, Hsiu-Cheng Hung and Frank Weber Biochemistry Center Heidelberg, University of Heidelberg,
Neural circuits underlying circadian behavior in Drosophila melanogaster
 Behavioural Processes 71 (2006) 211 225 Review Neural circuits underlying circadian behavior in Drosophila melanogaster Dennis C. Chang Department of Biology, Brandeis University, 415 South Street, MS-008,
Behavioural Processes 71 (2006) 211 225 Review Neural circuits underlying circadian behavior in Drosophila melanogaster Dennis C. Chang Department of Biology, Brandeis University, 415 South Street, MS-008,
Light-Mediated TIM Degradation within Drosophila Pacemaker Neurons (s-lnvs) Is Neither Necessary nor Sufficient for Delay Zone Phase Shifts
 Report Light-Mediated TIM egradation within rosophila Pacemaker Neurons (s) Is Neither Necessary nor Sufficient for elay Zone Phase Shifts hih-hang nthony Tang, Erica Hinteregger,, Yuhua Shang, and Michael
Report Light-Mediated TIM egradation within rosophila Pacemaker Neurons (s) Is Neither Necessary nor Sufficient for elay Zone Phase Shifts hih-hang nthony Tang, Erica Hinteregger,, Yuhua Shang, and Michael
What Is There Left to Learn about the Drosophila Clock?
 What Is There Left to Learn about the Drosophila Clock? J. BLAU, F. BLANCHARD, B. COLLINS, D. DAHDAL, A. KNOWLES, D. MIZRAK, AND M. RUBEN Department of Biology, New York University, New York, New York
What Is There Left to Learn about the Drosophila Clock? J. BLAU, F. BLANCHARD, B. COLLINS, D. DAHDAL, A. KNOWLES, D. MIZRAK, AND M. RUBEN Department of Biology, New York University, New York, New York
PDF Cells Are a GABA-Responsive Wake-Promoting Component of the Drosophila Sleep Circuit
 Article PDF Cells Are a GABA-Responsive Wake-Promoting Component of the Drosophila Sleep Circuit Katherine M. Parisky, 1,3 Jose Agosto, 1,2,3 Stefan R. Pulver, 1,3 Yuhua Shang, 1,2,3 Elena Kuklin, 1,3
Article PDF Cells Are a GABA-Responsive Wake-Promoting Component of the Drosophila Sleep Circuit Katherine M. Parisky, 1,3 Jose Agosto, 1,2,3 Stefan R. Pulver, 1,3 Yuhua Shang, 1,2,3 Elena Kuklin, 1,3
Article. Calcitonin Gene-Related Peptide Neurons Mediate Sleep-Specific Circadian Output in Drosophila
 Current Biology 24, 2652 2664, November 17, 2014 ª2014 Elsevier Ltd All rights reserved Calcitonin Gene-Related Peptide Neurons Mediate Sleep-Specific Circadian Output in Drosophila http://dx.doi.org/10.1016/j.cub.2014.09.077
Current Biology 24, 2652 2664, November 17, 2014 ª2014 Elsevier Ltd All rights reserved Calcitonin Gene-Related Peptide Neurons Mediate Sleep-Specific Circadian Output in Drosophila http://dx.doi.org/10.1016/j.cub.2014.09.077
Regulation of sleep plasticity by a thermo-sensitive circuit in Drosophila
 Regulation of sleep plasticity by a thermo-sensitive circuit in Drosophila Angelique Lamaze 1, Arzu Öztürk-Çolak 2, Robin Fischer 3, Nicolai Peschel 3, Kyunghee Koh 2 and James E.C. Jepson 1* 1 UCL Institute
Regulation of sleep plasticity by a thermo-sensitive circuit in Drosophila Angelique Lamaze 1, Arzu Öztürk-Çolak 2, Robin Fischer 3, Nicolai Peschel 3, Kyunghee Koh 2 and James E.C. Jepson 1* 1 UCL Institute
EXCMA. SRA. DRA. LESLIE C. GRIFFITH
 EXCMA. SRA. DRA. LESLIE C. GRIFFITH Answering speech Pedro Clarós, M.D., Ph.D. Numerary Academic of The Royal European Academy of Doctors Mr. Chairman of the Royal European Academy of Doctors. Distinguished
EXCMA. SRA. DRA. LESLIE C. GRIFFITH Answering speech Pedro Clarós, M.D., Ph.D. Numerary Academic of The Royal European Academy of Doctors Mr. Chairman of the Royal European Academy of Doctors. Distinguished
Clicker Question. The Need to Decompose. Mechanism and Reduction: Decomposing Circadian Clocks
 Mechanism and Reduction: Decomposing Circadian Clocks Clicker Question On the Deductive-Nomological (DN) model of reduction, which of the following does not figure in providing the explanation (i.e., is
Mechanism and Reduction: Decomposing Circadian Clocks Clicker Question On the Deductive-Nomological (DN) model of reduction, which of the following does not figure in providing the explanation (i.e., is
Report. The GABA A Receptor RDL Acts in Peptidergic PDF Neurons to Promote Sleep in Drosophila
 Current Biology 19, 386 390, March 10, 2009 ª2009 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2009.01.040 The GABA A Receptor RDL Acts in Peptidergic PDF Neurons to Promote Sleep in Drosophila Report
Current Biology 19, 386 390, March 10, 2009 ª2009 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2009.01.040 The GABA A Receptor RDL Acts in Peptidergic PDF Neurons to Promote Sleep in Drosophila Report
NIH Public Access Author Manuscript Cell Rep. Author manuscript; available in PMC 2012 September 04.
 NIH Public Access Author Manuscript Published in final edited form as: Cell Rep. 2012 August 30; 2(2): 332 344. doi:10.1016/j.celrep.2012.06.021. Autoreceptor Modulation of Peptide/Neurotransmitter Co-release
NIH Public Access Author Manuscript Published in final edited form as: Cell Rep. 2012 August 30; 2(2): 332 344. doi:10.1016/j.celrep.2012.06.021. Autoreceptor Modulation of Peptide/Neurotransmitter Co-release
Neural clocks and Neuropeptide F/Y regulate circadian gene expression in a peripheral metabolic tissue
 RESEARCH ARTICLE Neural clocks and Neuropeptide F/Y regulate circadian gene expression in a peripheral metabolic tissue Renske Erion 1, Anna N King 1, Gang Wu 2, John B Hogenesch 3, Amita Sehgal 1 * 1
RESEARCH ARTICLE Neural clocks and Neuropeptide F/Y regulate circadian gene expression in a peripheral metabolic tissue Renske Erion 1, Anna N King 1, Gang Wu 2, John B Hogenesch 3, Amita Sehgal 1 * 1
Why am I working in a Drosophila lab? Dr. Stefan Pulver, Brandeis University
 Why am I working in a Drosophila lab? Dr. Stefan Pulver, Brandeis University !Short life span The advantages of Drosophila as a model organism in neurocience!genetic manipulations are relatively easy!forward
Why am I working in a Drosophila lab? Dr. Stefan Pulver, Brandeis University !Short life span The advantages of Drosophila as a model organism in neurocience!genetic manipulations are relatively easy!forward
Drosophila Free-Running Rhythms Require Intercellular Communication
 Drosophila Free-Running Rhythms Require Intercellular Communication Ying Peng 1,2, Dan Stoleru 1,2, Joel D. Levine 1, Jeffrey C. Hall 1, Michael Rosbash 1,2* PLoS BIOLOGY 1 Department of Biology, Brandeis
Drosophila Free-Running Rhythms Require Intercellular Communication Ying Peng 1,2, Dan Stoleru 1,2, Joel D. Levine 1, Jeffrey C. Hall 1, Michael Rosbash 1,2* PLoS BIOLOGY 1 Department of Biology, Brandeis
Sexual Interactions Influence the Molecular Oscillations in DN1 Pacemaker Neurons in Drosophila melanogaster
 Sexual Interactions Influence the Molecular Oscillations in DN1 Pacemaker Neurons in Drosophila melanogaster Shiho Hanafusa, Tomoaki Kawaguchi, Yujiro Umezaki, Kenji Tomioka, Taishi Yoshii * Graduate School
Sexual Interactions Influence the Molecular Oscillations in DN1 Pacemaker Neurons in Drosophila melanogaster Shiho Hanafusa, Tomoaki Kawaguchi, Yujiro Umezaki, Kenji Tomioka, Taishi Yoshii * Graduate School
Setting the clock by nature: Circadian rhythm in the fruitfly Drosophila melanogaster
 FEBS Letters 585 (2011) 1435 1442 journal homepage: www.febsletters.org Review Setting the clock by nature: Circadian rhythm in the fruitfly Drosophila melanogaster Nicolai Peschel, Charlotte Helfrich-Förster
FEBS Letters 585 (2011) 1435 1442 journal homepage: www.febsletters.org Review Setting the clock by nature: Circadian rhythm in the fruitfly Drosophila melanogaster Nicolai Peschel, Charlotte Helfrich-Förster
8. Have the authors tested if the effects of expressing mir-92a are developmental or adult-specific?
 Reviewers' comments: Reviewer #1 (Remarks to the Author): The manuscript from Chen and Rosbash identifies a specific microrna (mir -92a) that is rhythmically expressed in the master pacemaker clock neurons
Reviewers' comments: Reviewer #1 (Remarks to the Author): The manuscript from Chen and Rosbash identifies a specific microrna (mir -92a) that is rhythmically expressed in the master pacemaker clock neurons
The effect of light exposure on Drosophila melanogaster survival
 The effect of light exposure on Drosophila melanogaster survival Grace M. Chen, Caroline M. Kim, Steve Lee, Elsie Ng ABSTRACT The purpose of our study was to observe how exposure to light affects the survival
The effect of light exposure on Drosophila melanogaster survival Grace M. Chen, Caroline M. Kim, Steve Lee, Elsie Ng ABSTRACT The purpose of our study was to observe how exposure to light affects the survival
R1 Neural Circuitry on Ellipsoid Body Promotes Sleep in Drosophila Melanogaster. Senior Thesis. Presented to
 R1 Neural Circuitry on Ellipsoid Body Promotes Sleep in Drosophila Melanogaster Senior Thesis Presented to The Faculty of the School of Arts and Sciences Brandeis University Undergraduate Program in Neuroscience
R1 Neural Circuitry on Ellipsoid Body Promotes Sleep in Drosophila Melanogaster Senior Thesis Presented to The Faculty of the School of Arts and Sciences Brandeis University Undergraduate Program in Neuroscience
Pigment-Dispersing Factor Modulates Pheromone Production in Clock Cells that Influence Mating in Drosophila
 rticle Pigment-Dispersing Factor Modulates Pheromone Production in Clock Cells that Influence Mating in Drosophila Joshua J. Krupp, Jean-Christophe illeter,,2 my Wong, Charles Choi, 3 Michael N. Nitabach,
rticle Pigment-Dispersing Factor Modulates Pheromone Production in Clock Cells that Influence Mating in Drosophila Joshua J. Krupp, Jean-Christophe illeter,,2 my Wong, Charles Choi, 3 Michael N. Nitabach,
The Nobel Assembly at Karolinska Institutet has today decided to award. the 2017 Nobel Prize in Physiology or Medicine. jointly to
 The Nobel Assembly at Karolinska Institutet has today decided to award the 2017 Nobel Prize in Physiology or Medicine jointly to Jeffrey C. Hall, Michael Rosbash and Michael W. Young for their discoveries
The Nobel Assembly at Karolinska Institutet has today decided to award the 2017 Nobel Prize in Physiology or Medicine jointly to Jeffrey C. Hall, Michael Rosbash and Michael W. Young for their discoveries
Seeing the light: photobehavior in fruit fly larvae
 Review Seeing the light: photobehavior in fruit fly larvae Alex C. Keene 1 and Simon G. Sprecher 2 1 Department of Biology, MS 314, University of Nevada, Reno, NV 89557, USA 2 Institute of Cell and Developmental
Review Seeing the light: photobehavior in fruit fly larvae Alex C. Keene 1 and Simon G. Sprecher 2 1 Department of Biology, MS 314, University of Nevada, Reno, NV 89557, USA 2 Institute of Cell and Developmental
Significance. 1 To whom correspondence should be addressed.
 Pacemaker-neuron dependent disturbance of the molecular clockwork by a Drosophila CLOCK mutant homologous to the mouse Clock mutation Euna Lee a,b, Eunjoo Cho c, Doo Hyun Kang a,b, Eun Hee Jeong b, Zheng
Pacemaker-neuron dependent disturbance of the molecular clockwork by a Drosophila CLOCK mutant homologous to the mouse Clock mutation Euna Lee a,b, Eunjoo Cho c, Doo Hyun Kang a,b, Eun Hee Jeong b, Zheng
Welcome to Bi !
 Welcome to Bi156 2012! Professors: Paul Patterson (php@caltech.edu) Kai Zinn (zinnk@caltech.edu) TAs: Janna Nawroth Yanan Sui ysui@caltech.edu Student presentations Select a topic
Welcome to Bi156 2012! Professors: Paul Patterson (php@caltech.edu) Kai Zinn (zinnk@caltech.edu) TAs: Janna Nawroth Yanan Sui ysui@caltech.edu Student presentations Select a topic
Molecular and Neuronal Analysis of Circadian Photoresponses in Drosophila: A Dissertation
 University of Massachusetts Medical School escholarship@umms GSBS Dissertations and Theses Graduate School of Biomedical Sciences 2007-10-25 Molecular and Neuronal Analysis of Circadian Photoresponses
University of Massachusetts Medical School escholarship@umms GSBS Dissertations and Theses Graduate School of Biomedical Sciences 2007-10-25 Molecular and Neuronal Analysis of Circadian Photoresponses
The Neuropeptide Pigment-Dispersing Factor Adjusts Period and Phase of Drosophila s Clock
 The Journal of Neuroscience, February 25, 2009 29(8):2597 2610 2597 Cellular/Molecular The Neuropeptide Pigment-Dispersing Factor Adjusts Period and Phase of Drosophila s Clock Taishi Yoshii, 1 Corinna
The Journal of Neuroscience, February 25, 2009 29(8):2597 2610 2597 Cellular/Molecular The Neuropeptide Pigment-Dispersing Factor Adjusts Period and Phase of Drosophila s Clock Taishi Yoshii, 1 Corinna
The Role of the Clock Gene in Protection Against Neural and Retinal Degeneration
 The Role of the Clock Gene in Protection Against Neural and Retinal Degeneration Jenny Yu Senior Honors Thesis Program in Biological Sciences Northwestern University Spring 2012 Principal Investigator:
The Role of the Clock Gene in Protection Against Neural and Retinal Degeneration Jenny Yu Senior Honors Thesis Program in Biological Sciences Northwestern University Spring 2012 Principal Investigator:
Article. Glial Cells Physiologically Modulate Clock Neurons and Circadian Behavior in a Calcium-Dependent Manner
 Current Biology 21, 625 634, April 26, 2011 ª2011 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2011.03.027 Glial Cells Physiologically Modulate Clock Neurons and Circadian Behavior in a Calcium-Dependent
Current Biology 21, 625 634, April 26, 2011 ª2011 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2011.03.027 Glial Cells Physiologically Modulate Clock Neurons and Circadian Behavior in a Calcium-Dependent
A constant light-genetic screen identifies KISMET as a regulator of circadian photoresponses
 University of Massachusetts Medical School escholarship@umms Open Access Articles Open Access Publications by UMMS Authors 12-31-2009 A constant light-genetic screen identifies KISMET as a regulator of
University of Massachusetts Medical School escholarship@umms Open Access Articles Open Access Publications by UMMS Authors 12-31-2009 A constant light-genetic screen identifies KISMET as a regulator of
Report. NEMO Kinase Contributes to Core Period Determination by Slowing the Pace of the Drosophila Circadian Oscillator
 Current Biology 21, 756 761, May 10, 2011 ª2011 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2011.02.037 NEMO Kinase Contributes to Core Period Determination by Slowing the Pace of the Drosophila
Current Biology 21, 756 761, May 10, 2011 ª2011 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2011.02.037 NEMO Kinase Contributes to Core Period Determination by Slowing the Pace of the Drosophila
Neurons and Hormones 1. How do animals perform the right behaviors at the right time? In the right context?
 Neurons and Hormones 1 How do animals perform the right behaviors at the right time? In the right context? Active at night only What if conflicting signals? Magnetic cues are always present But migrate
Neurons and Hormones 1 How do animals perform the right behaviors at the right time? In the right context? Active at night only What if conflicting signals? Magnetic cues are always present But migrate
The Transcription Factor Mef2 Is Required for Normal Circadian Behavior in Drosophila
 The Journal of Neuroscience, April 28, 2010 30(17):5855 5865 5855 Cellular/Molecular The Transcription Factor Mef2 Is Required for Normal Circadian Behavior in Drosophila Florence J. Blanchard, 1 Ben Collins,
The Journal of Neuroscience, April 28, 2010 30(17):5855 5865 5855 Cellular/Molecular The Transcription Factor Mef2 Is Required for Normal Circadian Behavior in Drosophila Florence J. Blanchard, 1 Ben Collins,
Biological Clocks. Lu Chen, Ph.D. MCB, UC Berkeley. What is biological clock?
 Biological Clocks Lu Chen, Ph.D. MCB, UC Berkeley 1 What is biological clock? All eukaryotes and some prokaryotes display changes in gene activity, biochemistry, physiology, and behavior that wax and wane
Biological Clocks Lu Chen, Ph.D. MCB, UC Berkeley 1 What is biological clock? All eukaryotes and some prokaryotes display changes in gene activity, biochemistry, physiology, and behavior that wax and wane
Biological Clocks. Lu Chen, Ph.D. MCB, UC Berkeley. Why Does Melatonin Now Outsell Vitamin C??
 Biological Clocks Lu Chen, Ph.D. MCB, UC Berkeley 1 Why Does Melatonin Now Outsell Vitamin C?? Wake / sleep complaints are extremely prevalent. Much melatonin is consumed in an attempt to overcome the
Biological Clocks Lu Chen, Ph.D. MCB, UC Berkeley 1 Why Does Melatonin Now Outsell Vitamin C?? Wake / sleep complaints are extremely prevalent. Much melatonin is consumed in an attempt to overcome the
Supplemental Information. Proprioceptive Opsin Functions. in Drosophila Larval Locomotion
 Neuron, Volume 98 Supplemental Information Proprioceptive Opsin Functions in Drosophila Larval Locomotion Damiano Zanini, Diego Giraldo, Ben Warren, Radoslaw Katana, Marta Andrés, Suneel Reddy, Stephanie
Neuron, Volume 98 Supplemental Information Proprioceptive Opsin Functions in Drosophila Larval Locomotion Damiano Zanini, Diego Giraldo, Ben Warren, Radoslaw Katana, Marta Andrés, Suneel Reddy, Stephanie
Lmo Mutants Reveal a Novel Role for Circadian Pacemaker Neurons in Cocaine-Induced Behaviors
 Lmo Mutants Reveal a Novel Role for Circadian Pacemaker Neurons in Cocaine-Induced Behaviors Linus T.-Y. Tsai 1, Roland J. Bainton 2, Justin Blau 3, Ulrike Heberlein 4 * Open access, freely available online
Lmo Mutants Reveal a Novel Role for Circadian Pacemaker Neurons in Cocaine-Induced Behaviors Linus T.-Y. Tsai 1, Roland J. Bainton 2, Justin Blau 3, Ulrike Heberlein 4 * Open access, freely available online
Regulation of Sleep and Circadian Rhythms by Metabolic Neuropeptides
 University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 1-1-2014 Regulation of Sleep and Circadian Rhythms by Metabolic Neuropeptides Renske Erion University of Pennsylvania,
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 1-1-2014 Regulation of Sleep and Circadian Rhythms by Metabolic Neuropeptides Renske Erion University of Pennsylvania,
Transcriptional Feedback Loop Regulation, Function, and Ontogeny in Drosophila
 Transcriptional Feedback Loop Regulation, Function, and Ontogeny in Drosophila J. BENITO, H. ZHENG, F.S. NG, AND P.E. HARDIN Center for Research on Biological Clocks, Department of Biology, Texas A&M University,
Transcriptional Feedback Loop Regulation, Function, and Ontogeny in Drosophila J. BENITO, H. ZHENG, F.S. NG, AND P.E. HARDIN Center for Research on Biological Clocks, Department of Biology, Texas A&M University,
Uppers and Downers: Understanding Sleep Regulation Using Small Molecules in Drosophila
 University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 1-1-2014 Uppers and Downers: Understanding Sleep Regulation Using Small Molecules in Drosophila Aleksandra Nall University
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 1-1-2014 Uppers and Downers: Understanding Sleep Regulation Using Small Molecules in Drosophila Aleksandra Nall University
Short Neuropeptide F Is a Sleep-Promoting Inhibitory Modulator
 Article Short Neuropeptide F Is a Sleep-Promoting Inhibitory Modulator Yuhua Shang, 1,3 Nathan C. Donelson, 1,3 Christopher G. Vecsey, 1 Fang Guo, 1,2 Michael Rosbash, 1,2, * and Leslie C. Griffith 1,
Article Short Neuropeptide F Is a Sleep-Promoting Inhibitory Modulator Yuhua Shang, 1,3 Nathan C. Donelson, 1,3 Christopher G. Vecsey, 1 Fang Guo, 1,2 Michael Rosbash, 1,2, * and Leslie C. Griffith 1,
Transcriptional and Translational Mechanisms Controlling Circadian Rhythms in Drosophila: A Dissertation
 University of Massachusetts Medical School escholarship@umms GSBS Dissertations and Theses Graduate School of Biomedical Sciences 6-14-2013 Transcriptional and Translational Mechanisms Controlling Circadian
University of Massachusetts Medical School escholarship@umms GSBS Dissertations and Theses Graduate School of Biomedical Sciences 6-14-2013 Transcriptional and Translational Mechanisms Controlling Circadian
Even a stopped clock tells the right time twice a day: circadian timekeeping in Drosophila
 Pflugers Arch - Eur J Physiol (2007) 454:857 867 DOI 10.1007/s00424-006-0188-9 INVITED REVIEW Even a stopped clock tells the right time twice a day: circadian timekeeping in Drosophila Ben Collins & Justin
Pflugers Arch - Eur J Physiol (2007) 454:857 867 DOI 10.1007/s00424-006-0188-9 INVITED REVIEW Even a stopped clock tells the right time twice a day: circadian timekeeping in Drosophila Ben Collins & Justin
CIRCADIAN SIGNALING NETWORKS
 Transcription Regulation And Gene Expression in Eukaryotes Cycle G2 (lecture 13709) FS 2014 P. Matthias and RG Clerc Roger G. Clerc 07.05.2014 CIRCADIAN SIGNALING NETWORKS Master pacemaker SCN «Slave clocks»
Transcription Regulation And Gene Expression in Eukaryotes Cycle G2 (lecture 13709) FS 2014 P. Matthias and RG Clerc Roger G. Clerc 07.05.2014 CIRCADIAN SIGNALING NETWORKS Master pacemaker SCN «Slave clocks»
Simulation of Drosophila Circadian Oscillations, Mutations, and Light Responses by a Model with VRI, PDP-1, and CLK
 Title: Simulation of Drosophila Circadian Oscillations, Mutations, and Light Responses by a Model with VRI, PDP-1, and CLK Authors: Paul Smolen, Paul E. Hardin *, Brian S. Lo, Douglas A. Baxter, and John
Title: Simulation of Drosophila Circadian Oscillations, Mutations, and Light Responses by a Model with VRI, PDP-1, and CLK Authors: Paul Smolen, Paul E. Hardin *, Brian S. Lo, Douglas A. Baxter, and John
PER-TIM Interactions with the Photoreceptor Cryptochrome Mediate Circadian Temperature Responses in Drosophila
 PLoS BIOLOGY PER-TIM Interactions with the Photoreceptor Cryptochrome Mediate Circadian Temperature Responses in Drosophila Rachna Kaushik 1,2,3, Pipat Nawathean 1,2,3, Ania Busza 4,5,6, Alejandro Murad
PLoS BIOLOGY PER-TIM Interactions with the Photoreceptor Cryptochrome Mediate Circadian Temperature Responses in Drosophila Rachna Kaushik 1,2,3, Pipat Nawathean 1,2,3, Ania Busza 4,5,6, Alejandro Murad
PEER REVIEW FILE. Reviewers' Comments: Reviewer #1 (Remarks to the Author):
 PEER REVIEW FILE Reviewers' Comments: Reviewer #1 (Remarks to the Author): In this manuscript entitled "Neural circuits for long-term water-reward memory processing in thirsty Drosophila", the authors
PEER REVIEW FILE Reviewers' Comments: Reviewer #1 (Remarks to the Author): In this manuscript entitled "Neural circuits for long-term water-reward memory processing in thirsty Drosophila", the authors
Index. sleep.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Accidents, at work, effect of shift work disorder on, 263 264 Acetylcholine, in circadian rhythms, 100 105 Acrophase, definition of, 301 Actigraphy,
Note: Page numbers of article titles are in boldface type. A Accidents, at work, effect of shift work disorder on, 263 264 Acetylcholine, in circadian rhythms, 100 105 Acrophase, definition of, 301 Actigraphy,
Biochemical and Biophysical Research Communications
 Biochemical and Biophysical Research Communications 372 (2008) 798 803 Contents lists available at ScienceDirect Biochemical and Biophysical Research Communications journal homepage: www.elsevier.com/locate/ybbrc
Biochemical and Biophysical Research Communications 372 (2008) 798 803 Contents lists available at ScienceDirect Biochemical and Biophysical Research Communications journal homepage: www.elsevier.com/locate/ybbrc
Advance in circadian rhythm genetics in mammals
 16 2 2004 4 Chinese Bulletin of Life Sciences Vol. 16, No. 2 Apr., 2004 1004-0374 (2004) 02-0104-05 1 100101 2 434025 9 24, Q41 A Advance in circadian rhythm genetics in mammals XU Zu-Yuan 1,2 (1 Beijing
16 2 2004 4 Chinese Bulletin of Life Sciences Vol. 16, No. 2 Apr., 2004 1004-0374 (2004) 02-0104-05 1 100101 2 434025 9 24, Q41 A Advance in circadian rhythm genetics in mammals XU Zu-Yuan 1,2 (1 Beijing
PHYSIOLOGY AND MAINTENANCE Vol. V - Biological Rhythms - Tarja Porkka-Heiskanen, Jarmo T. Laitinen
 BIOLOGICAL RHYTHMS Tarja Porkka-Heiskanen, Institute of Biomedicine, University of Helsinki, Finland Jarmo T. Laitinen Department of Physiology, University of Kuopio, Finland Keywords: Light, melatonin,
BIOLOGICAL RHYTHMS Tarja Porkka-Heiskanen, Institute of Biomedicine, University of Helsinki, Finland Jarmo T. Laitinen Department of Physiology, University of Kuopio, Finland Keywords: Light, melatonin,
Transcription Regulation And Gene Expression in Eukaryotes (Cycle G2 # )
 Transcription Regulation And Gene Expression in Eukaryotes (Cycle G2 #13709-01) CIRCADIAN SIGNALING NETWORKS RG. Clerc May 19. 2010 www.fmi.ch/training/teaching Circadian rythms : most physiological processes
Transcription Regulation And Gene Expression in Eukaryotes (Cycle G2 #13709-01) CIRCADIAN SIGNALING NETWORKS RG. Clerc May 19. 2010 www.fmi.ch/training/teaching Circadian rythms : most physiological processes
Supplementary Figure 1. Flies form water-reward memory only in the thirsty state
 1 2 3 4 5 6 7 Supplementary Figure 1. Flies form water-reward memory only in the thirsty state Thirsty but not sated wild-type flies form robust 3 min memory. For the thirsty group, the flies were water-deprived
1 2 3 4 5 6 7 Supplementary Figure 1. Flies form water-reward memory only in the thirsty state Thirsty but not sated wild-type flies form robust 3 min memory. For the thirsty group, the flies were water-deprived
The Use of Bright Light in the Treatment of Insomnia
 Chapter e39 The Use of Bright Light in the Treatment of Insomnia Leon Lack and Helen Wright Department of Psychology, Flinders University, Adelaide, South Australia PROTOCOL NAME The use of bright light
Chapter e39 The Use of Bright Light in the Treatment of Insomnia Leon Lack and Helen Wright Department of Psychology, Flinders University, Adelaide, South Australia PROTOCOL NAME The use of bright light
Behavioral and molecular analyses suggest that circadian output is disrupted by disconnected mutants
 The EMBO Journal vol. 1 1 no. 1 pp. 1-6, 1992 Behavioral and molecular analyses suggest that circadian output is disrupted by disconnected mutants in D.melanogaster Paul E.Hardin1,2,3, Jeffrey C.Hall2
The EMBO Journal vol. 1 1 no. 1 pp. 1-6, 1992 Behavioral and molecular analyses suggest that circadian output is disrupted by disconnected mutants in D.melanogaster Paul E.Hardin1,2,3, Jeffrey C.Hall2
Circadian rhythm and Sleep. Radwan Banimustafa MD
 Circadian rhythm and Sleep Radwan Banimustafa MD Homeostasis Maintenance of equilibrium by active regulation of internal states: Cardiovascular function (blood pressure, heart rate) Body temperature Food
Circadian rhythm and Sleep Radwan Banimustafa MD Homeostasis Maintenance of equilibrium by active regulation of internal states: Cardiovascular function (blood pressure, heart rate) Body temperature Food
Circadian modulation of short-term memory in Drosophila
 Research Circadian modulation of short-term memory in Drosophila Lisa C. Lyons 1 3 and Gregg Roman 1 1 Department of Biology and Biochemistry, University of Houston, Houston, Texas 77204, USA; 2 Department
Research Circadian modulation of short-term memory in Drosophila Lisa C. Lyons 1 3 and Gregg Roman 1 1 Department of Biology and Biochemistry, University of Houston, Houston, Texas 77204, USA; 2 Department
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10643 Supplementary Table 1. Identification of hecw-1 coding polymorphisms at amino acid positions 322 and 325 in 162 strains of C. elegans. WWW.NATURE.COM/NATURE 1 Supplementary Figure
doi:10.1038/nature10643 Supplementary Table 1. Identification of hecw-1 coding polymorphisms at amino acid positions 322 and 325 in 162 strains of C. elegans. WWW.NATURE.COM/NATURE 1 Supplementary Figure
Supplementary Figure 1. Procedures to independently control fly hunger and thirst states.
 Supplementary Figure 1 Procedures to independently control fly hunger and thirst states. (a) Protocol to produce exclusively hungry or thirsty flies for 6 h water memory retrieval. (b) Consumption assays
Supplementary Figure 1 Procedures to independently control fly hunger and thirst states. (a) Protocol to produce exclusively hungry or thirsty flies for 6 h water memory retrieval. (b) Consumption assays
The Possible Role of Extra-ocular Photoreceptors in the Entrainment of Lobster Circadian Rhythms to Light:Dark Cycles
 University of New Hampshire University of New Hampshire Scholars' Repository Honors Theses and Capstones Student Scholarship Spring 2013 The Possible Role of Extra-ocular Photoreceptors in the Entrainment
University of New Hampshire University of New Hampshire Scholars' Repository Honors Theses and Capstones Student Scholarship Spring 2013 The Possible Role of Extra-ocular Photoreceptors in the Entrainment
Cell Signaling Components of the Drosophila Circadian Pacemaker
 Washington University in St. Louis Washington University Open Scholarship All Theses and Dissertations (ETDs) Winter 1-1-2012 Cell Signaling Components of the Drosophila Circadian Pacemaker Laura B. Duvall
Washington University in St. Louis Washington University Open Scholarship All Theses and Dissertations (ETDs) Winter 1-1-2012 Cell Signaling Components of the Drosophila Circadian Pacemaker Laura B. Duvall
Characterization of Circadian Feeding Rhythms in Drosophila Using the Fly Liquid-Food Interaction Counter (FLIC) Assay
 Characterization of Circadian Feeding Rhythms in Drosophila Using the Fly Liquid-Food Interaction Counter (FLIC) Assay by Qi Zhang A dissertation submitted in partial fulfillment of the requirements for
Characterization of Circadian Feeding Rhythms in Drosophila Using the Fly Liquid-Food Interaction Counter (FLIC) Assay by Qi Zhang A dissertation submitted in partial fulfillment of the requirements for
Stochastic simulations
 Circadian rhythms Stochastic simulations Circadian rhythms allow living organisms to live in phase with the alternance of day and night... Application to circadian clocks Didier Gonze Circadian rhythms
Circadian rhythms Stochastic simulations Circadian rhythms allow living organisms to live in phase with the alternance of day and night... Application to circadian clocks Didier Gonze Circadian rhythms
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
www.sciencemag.org/cgi/content/full/1171320/dc1 Supporting Online Material for A Frazzled/DCC-Dependent Transcriptional Switch Regulates Midline Axon Guidance Long Yang, David S. Garbe, Greg J. Bashaw*
Drosophila Photoreceptors Contain an Autonomous Circadian Oscillator That Can Function without period mrna Cycling
 The Journal of Neuroscience, January 15, 1998, 18(2):741 750 Drosophila Photoreceptors Contain an Autonomous Circadian Oscillator That Can Function without period mrna Cycling Yuzhong Cheng 1 and Paul
The Journal of Neuroscience, January 15, 1998, 18(2):741 750 Drosophila Photoreceptors Contain an Autonomous Circadian Oscillator That Can Function without period mrna Cycling Yuzhong Cheng 1 and Paul
Circadian rhythms of locomotor activity in Drosophila
 Behavioural Processes 64 (2003) 161 175 Circadian rhythms of locomotor activity in Drosophila André Klarsfeld, Jean-Christophe Leloup, François Rouyer Institut de Neurobiologie Alfred Fessard, CNRS UPR
Behavioural Processes 64 (2003) 161 175 Circadian rhythms of locomotor activity in Drosophila André Klarsfeld, Jean-Christophe Leloup, François Rouyer Institut de Neurobiologie Alfred Fessard, CNRS UPR
The Success of Decomposition
 11/21/11 Mechanism and Levels of Organization: Recomposing and Situating Circadian Clocks The Success of Decomposition Moving beyond per, researchers in the 1990s and early 2000s identified many clock
11/21/11 Mechanism and Levels of Organization: Recomposing and Situating Circadian Clocks The Success of Decomposition Moving beyond per, researchers in the 1990s and early 2000s identified many clock
Sleep-Wake Cycle I Brain Rhythms. Reading: BCP Chapter 19
 Sleep-Wake Cycle I Brain Rhythms Reading: BCP Chapter 19 Brain Rhythms and Sleep Earth has a rhythmic environment. For example, day and night cycle back and forth, tides ebb and flow and temperature varies
Sleep-Wake Cycle I Brain Rhythms Reading: BCP Chapter 19 Brain Rhythms and Sleep Earth has a rhythmic environment. For example, day and night cycle back and forth, tides ebb and flow and temperature varies
Circadian rhythms are genetically determined biological oscillations
 Circadian modulation of dopamine receptor responsiveness in Drosophila melanogaster Rozi Andretic and Jay Hirsh* Department of Biology, University of Virginia, Charlottesville, VA 22903 Edited by Edward
Circadian modulation of dopamine receptor responsiveness in Drosophila melanogaster Rozi Andretic and Jay Hirsh* Department of Biology, University of Virginia, Charlottesville, VA 22903 Edited by Edward
Supplementary Materials for
 Supplementary Materials for Simoni, A; Wolfgang, W; Topping, MP; Kavlie, RG; Stanewsky, R; Albert, JT; (2014) A mechanosensory pathway to the Drosophila circadian clock. Science, 343 (6170) 525-528. 10.1126/science.1245710.
Supplementary Materials for Simoni, A; Wolfgang, W; Topping, MP; Kavlie, RG; Stanewsky, R; Albert, JT; (2014) A mechanosensory pathway to the Drosophila circadian clock. Science, 343 (6170) 525-528. 10.1126/science.1245710.
Drosophila ATF-2 Regulates Sleep and Locomotor Activity in Pacemaker Neurons
 MOLECULAR AND CELLULAR BIOLOGY, Oct. 2008, p. 6278 6289 Vol. 28, No. 20 0270-7306/08/$08.00 0 doi:10.1128/mcb.02242-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Drosophila
MOLECULAR AND CELLULAR BIOLOGY, Oct. 2008, p. 6278 6289 Vol. 28, No. 20 0270-7306/08/$08.00 0 doi:10.1128/mcb.02242-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Drosophila
Ectopic Expression of the Neuropeptide Pigment-Dispersing Factor Alters Behavioral Rhythms in Drosophila melanogaster
 The Journal of Neuroscience, May 1, 2000, 20(9):3339 3353 Ectopic Expression of the Neuropeptide Pigment-Dispersing Factor Alters Behavioral Rhythms in Drosophila melanogaster Charlotte Helfrich-Förster,
The Journal of Neuroscience, May 1, 2000, 20(9):3339 3353 Ectopic Expression of the Neuropeptide Pigment-Dispersing Factor Alters Behavioral Rhythms in Drosophila melanogaster Charlotte Helfrich-Förster,
Circadian Rhythm Disturbances: What Happens When Your Biological Clock Is In The Wrong Time Zone
 Circadian Rhythm Disturbances: What Happens When Your Biological Clock Is In The Wrong Time Zone Steven A. Thau MD Chief, Pulmonary, Sleep Department. Phelps Hospital, Northwell Health Internal Clock Examples
Circadian Rhythm Disturbances: What Happens When Your Biological Clock Is In The Wrong Time Zone Steven A. Thau MD Chief, Pulmonary, Sleep Department. Phelps Hospital, Northwell Health Internal Clock Examples
AWARD NUMBER: W81XWH TITLE: Sleep Homeostasis and Synaptic Plasticity. PRINCIPAL INVESTIGATOR: Ravi Allada
 AWARD NUMBER: W81XWH-16-1-0169 TITLE: Sleep Homeostasis and Synaptic Plasticity PRINCIPAL INVESTIGATOR: Ravi Allada CONTRACTING ORGANIZATION: Northwestern University Evanston, IL 60201 REPORT DATE: June
AWARD NUMBER: W81XWH-16-1-0169 TITLE: Sleep Homeostasis and Synaptic Plasticity PRINCIPAL INVESTIGATOR: Ravi Allada CONTRACTING ORGANIZATION: Northwestern University Evanston, IL 60201 REPORT DATE: June
THE CIRCADIAN REGULATION OF FEEDING IN ADULT DROSOPHILA MELANOGASTER
 THE CIRCADIAN REGULATION OF FEEDING IN ADULT DROSOPHILA MELANOGASTER by Shreya Shekhar A thesis submitted in conformity with the requirements for the degree of Master of Science Graduate Department of
THE CIRCADIAN REGULATION OF FEEDING IN ADULT DROSOPHILA MELANOGASTER by Shreya Shekhar A thesis submitted in conformity with the requirements for the degree of Master of Science Graduate Department of
Cephalization. Nervous Systems Chapter 49 11/10/2013. Nervous systems consist of circuits of neurons and supporting cells
 Nervous Systems Chapter 49 Cephalization Nervous systems consist of circuits of neurons and supporting cells Nervous system organization usually correlates with lifestyle Organization of the vertebrate
Nervous Systems Chapter 49 Cephalization Nervous systems consist of circuits of neurons and supporting cells Nervous system organization usually correlates with lifestyle Organization of the vertebrate
Mechanisms of Behavioral Modulation
 Feb 19: Rhythms Mechanisms of Behavioral Modulation "Global" modulating mechanisms: act on diverse neural subsystems, changing threshold, selectivity, or strength of many responses EXAMPLES: hormones and
Feb 19: Rhythms Mechanisms of Behavioral Modulation "Global" modulating mechanisms: act on diverse neural subsystems, changing threshold, selectivity, or strength of many responses EXAMPLES: hormones and
Neuronal Machinery of Sleep Homeostasis in Drosophila
 Article Neuronal Machinery of Sleep Homeostasis in Drosophila Jeffrey M. Donlea, 1,2 Diogo Pimentel, 1,2 and Gero Miesenböck 1, * 1 Centre for Neural Circuits and Behaviour, University of Oxford, Tinsley
Article Neuronal Machinery of Sleep Homeostasis in Drosophila Jeffrey M. Donlea, 1,2 Diogo Pimentel, 1,2 and Gero Miesenböck 1, * 1 Centre for Neural Circuits and Behaviour, University of Oxford, Tinsley
Genetic and anatomical basis of the barrier separating wakefulness and anesthetic-induced unresponsiveness.
 Thomas Jefferson University Jefferson Digital Commons Department of Neuroscience Sidney Kimmel Medical College 9-1-2013 Genetic and anatomical basis of the barrier separating wakefulness and anesthetic-induced
Thomas Jefferson University Jefferson Digital Commons Department of Neuroscience Sidney Kimmel Medical College 9-1-2013 Genetic and anatomical basis of the barrier separating wakefulness and anesthetic-induced
Manipulations of Amyloid Precursor Protein Cleavage Disrupt the Circadian Clock in Aging Drosophila
 Manipulations of Amyloid Precursor Protein Cleavage Disrupt the Circadian Clock in Aging Drosophila Blake, M. R., Holbrook, S. D., Kotwica-Rolinska, J., Chow, E. S., Kretzschmar, D., & Giebultowicz, J.
Manipulations of Amyloid Precursor Protein Cleavage Disrupt the Circadian Clock in Aging Drosophila Blake, M. R., Holbrook, S. D., Kotwica-Rolinska, J., Chow, E. S., Kretzschmar, D., & Giebultowicz, J.
A Drosophila model for alcohol reward. Supplemental Material
 Kaun et al. Supplemental Material 1 A Drosophila model for alcohol reward Supplemental Material Kaun, K.R. *, Azanchi, R. *, Maung, Z. *, Hirsh, J. and Heberlein, U. * * Department of Anatomy, University
Kaun et al. Supplemental Material 1 A Drosophila model for alcohol reward Supplemental Material Kaun, K.R. *, Azanchi, R. *, Maung, Z. *, Hirsh, J. and Heberlein, U. * * Department of Anatomy, University
STUDYING GENETIC ORIGINS OF BEHAVIOUR: FRUIT FLIES, KNOCKOUT MICE, HUMANS AND SLEEP
 STUDYING GENETIC ORIGINS OF BEHAVIOUR: FRUIT FLIES, KNOCKOUT MICE, HUMANS AND SLEEP The twenty-first century seems to be the age of studying the genetic origins of behaviour. The everincreasing knowledge
STUDYING GENETIC ORIGINS OF BEHAVIOUR: FRUIT FLIES, KNOCKOUT MICE, HUMANS AND SLEEP The twenty-first century seems to be the age of studying the genetic origins of behaviour. The everincreasing knowledge
Neuropeptides are one of the principal physiological regulators
 Sex- and clock-controlled expression of the neuropeptide F gene in Drosophila Gyunghee Lee, Jae Hoon Bahn, and Jae H. Park* Department of Biochemistry and Cellular and Molecular Biology, University of
Sex- and clock-controlled expression of the neuropeptide F gene in Drosophila Gyunghee Lee, Jae Hoon Bahn, and Jae H. Park* Department of Biochemistry and Cellular and Molecular Biology, University of
This is a repository copy of Circadian Rhythms in Visual Responsiveness in the Behaviorally Arrhythmic Drosophila Clock Mutant ClkJrk.
 This is a repository copy of Circadian Rhythms in Visual Responsiveness in the Behaviorally Arrhythmic Drosophila Clock Mutant ClkJrk. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/124703/
This is a repository copy of Circadian Rhythms in Visual Responsiveness in the Behaviorally Arrhythmic Drosophila Clock Mutant ClkJrk. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/124703/
Drosophila CRY Is a Deep Brain Circadian Photoreceptor
 Neuron, Vol. 26, 493 504, May, 2000, Copyright 2000 by Cell Press Drosophila CRY Is a Deep Brain Circadian Photoreceptor Patrick Emery,* Ralf Stanewsky, Charlotte Helfrich-Förster, Myai Emery-Le,* Jeffrey
Neuron, Vol. 26, 493 504, May, 2000, Copyright 2000 by Cell Press Drosophila CRY Is a Deep Brain Circadian Photoreceptor Patrick Emery,* Ralf Stanewsky, Charlotte Helfrich-Förster, Myai Emery-Le,* Jeffrey
Stochastic simulations
 Stochastic simulations Application to circadian clocks Didier Gonze Circadian rhythms Circadian rhythms allow living organisms to live in phase with the alternance of day and night... Circadian rhythms
Stochastic simulations Application to circadian clocks Didier Gonze Circadian rhythms Circadian rhythms allow living organisms to live in phase with the alternance of day and night... Circadian rhythms
Beyond our eyes: the non-visual impact of light
 Beyond our eyes: the non-visual impact of light Christian Cajochen, PhD Centre for Chronobiology Psychiatric Hospital of the University of Basel, Switzerland Workshop Intelligent Efficient Human Centric
Beyond our eyes: the non-visual impact of light Christian Cajochen, PhD Centre for Chronobiology Psychiatric Hospital of the University of Basel, Switzerland Workshop Intelligent Efficient Human Centric
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Rhythmic behavior is controlled by the SRm160 splicing factor in
 Genetics: Early Online, published on August 11, 2017 as 10.1534/genetics.117.300139 1 2 Rhythmic behavior is controlled by the SRm160 splicing factor in Drosophila melanogaster 3 4 5 6 Esteban J. Beckwith
Genetics: Early Online, published on August 11, 2017 as 10.1534/genetics.117.300139 1 2 Rhythmic behavior is controlled by the SRm160 splicing factor in Drosophila melanogaster 3 4 5 6 Esteban J. Beckwith
Biological rhythms. Types of biological rhythms
 Biological rhythms Types of biological rhythms 2/33 what do we call rhythm in a living organism? physiological events occurring at approximately regular times internally controlled rhythms: breathing,
Biological rhythms Types of biological rhythms 2/33 what do we call rhythm in a living organism? physiological events occurring at approximately regular times internally controlled rhythms: breathing,
Selection on the timing of adult emergence results in altered circadian clocks in fruit flies Drosophila melanogaster
 96 The Journal of Experimental Biology 21, 96-9 Published by The Company of Biologists 27 doi:1.1242/jeb.1354 Selection on the timing of adult emergence results in altered circadian clocks in fruit flies
96 The Journal of Experimental Biology 21, 96-9 Published by The Company of Biologists 27 doi:1.1242/jeb.1354 Selection on the timing of adult emergence results in altered circadian clocks in fruit flies
Altered Entrainment and Feedback Loop Function Effected by a Mutant Period Protein
 The Journal of Neuroscience, February 1, 2000, 20(3):958 968 Altered Entrainment and Feedback Loop Function Effected by a Mutant Period Protein Peter Schotland, Melissa Hunter-Ensor, Todd Lawrence, and
The Journal of Neuroscience, February 1, 2000, 20(3):958 968 Altered Entrainment and Feedback Loop Function Effected by a Mutant Period Protein Peter Schotland, Melissa Hunter-Ensor, Todd Lawrence, and
