MEDICAL POLICY SUBJECT: GENETIC TESTING FOR SUSCEPTIBILITY TO HEREDITARY CANCERS EFFECTIVE DATE: 06/19/14, 09/15/15.
|
|
|
- Jewel Garrison
- 6 years ago
- Views:
Transcription
1 MEDICAL POLICY SUBJECT: GENETIC TESTING FOR SUSCEPTIBILITY PAGE: 1 OF: 7 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied. Medical policies apply to commercial and Medicaid products only when a contract benefit for the specific service exists. Medical policies only apply to Medicare products when a contract benefit exists and where there are no National or Local Medicare coverage decisions for the specific service. POLICY STATEMENT: I. Based upon our criteria and assessment of the peer-reviewed literature, including National Comprehensive Cancer Network (NCCN) clinical guidelines genetic testing for hereditary breast and/or ovarian cancer has been medically proven to be effective and therefore medically appropriate in the following circumstances, when performed by a qualified laboratory and offered in a setting with adequately trained health care providers to provide appropriate pre-and post-test genetic counseling that will guide decisions regarding cancer prevention, surveillance, and treatment options. Testing should be performed in the affected family member first unless an unaffected individual has a family history of a known mutation; an affected individual has the highest likelihood for a positive test result. A negative result for an unaffected individual with only a family history of cancer is considered indeterminate (or uninformative) and does not provide the same level of information as when there is a known deleterious mutation in the family. Testing of unaffected family members in the absence of having tested affected family members significantly limits the interpretation of the test results. A. Cowden Syndrome (CS) (germline mutations of PTEN) (CPT: 81321, 81323): 1. An individual from a family with a known PTEN mutation (CPT: 81322); or 2. An individual with a personal history of Bannayan-Riley-Ruvalcaba syndrome (BRRS); or 3. An individual with adult Lhermitte-Duclos disease (cerebellar tumors); or 4. An individual with autism spectrum disorder and macrocephaly; or 5. An individual with two or more biopsy-proven trichilemmomas; or 6. An individual meeting the clinical diagnostic criteria for CS; or 7. An individual with a personal history of: a. Two or more major criteria (one must be macrocephaly); or b. Three major criteria, without macrocephaly; or c. One major and three or more minor criteria*; or d. Four or more minor criteria. *If an individual has two or more major criteria, but does not have macrocephaly, one of the major criteria may be included as one of the three minor criteria to meet testing criteria. 8. An individual with a first-degree relative with a clinical diagnosis of CS or BRRS for whom testing has not been performed. The individual must meet the following diagnostic criteria: a. Any one major criterion; or b. Two minor criteria. Major Criteria: a. Breast cancer; or b. Endometrial cancer; or Minor Criteria: a. Autism spectrum disorder; or b. Colon cancer; or
2 c. Follicular thyroid cancer; or d. Multiple GI hamartomas or ganglioneuromas e. Macrocephaly (megalocephaly (i.e., greater than or equal to 97%, 58 cm in adult women, 60 cm in adult men); or f. Macular pigmentation of glans penis; or g. Mucocutaneous lesions: i. One biopsy proven trichilemmoma ii. Multiple palmoplantar keratosis iii. Multifocal or extensive oral mucosal papillomatosis iv. Multiple cutaneous facial papules (often verrucous) PAGE: 2 OF: 7 c. Esophageal glycogenic acanthosis (greater than or equal to 3); or d. Lipomas; or e. Mental retardation (i.e., IQ less than or equal to 75); or f. Papillary or follicular variant of papillary thyroid cancer; or g. Thyroid structural lesions (e.g., adenoma, nodeules(s), goiter); or h. Renal cell carcinoma; or i. Single GI hamartoma or glanglioneuroma; or j. Testicular lipomatosis; or k. Vascular anomalies (including multiple intracranial developmental venous anomalies) II. B. Li Fraumeni Syndrome (LFS) (germline mutations of TP53): 1. An individual from a family with a known TP53 mutation; or 2. Personal history of breast cancer diagnosed at age 35 years or younger; may be performed simultaneously with BRCA mutation testing or when BRCA1/BRCA2 is negative; or 3. Personal history of adrenocortical carcinoma or choroid plexus carcinoma diagnosed at any age; or 4. Personal history of sarcoma diagnosed at age 45 years or younger; AND a. Has one 1 st degree relative diagnosed with cancer at age less than 45 years; AND b. Has one 1 st or 2 nd degree relative diagnosed with cancer at age less than 45 years, or a sarcoma at any age 5. Personal history of a tumor from the *LFS tumor spectrum diagnosed before age 46 years; AND a. Has one or more 1 st or 2 nd degree relative with a tumor from the LFS tumor spectrum (other than breast cancer if the proband has breast cancer) diagnosed before age 56 years; or b. Has one or more 1 st or 2 nd degree relative with multiple primaries diagnosed at any age *Tumors associated with LFS include but are not limited to: premenopausal breast cancer, bone and soft tissue sarcoma, acute leukemia, brain tumor, adrenocortical carcinoma, choroid plexus carcinoma, colon cancer, early onset of other adenocarcinomas or other childhood cancers) 6. Personal history of multiple tumors (except multiple breast tumors); AND a. The initial tumor was diagnosed before age 46 years; AND b. 2 of the tumors are from the *LFS tumor spectrum *Tumors associated with LFS include but are not limited to: premenopausal breast cancer, bone and soft tissue sarcoma, acute leukemia, brain tumor, adrenocortical carcinoma, choroid plexus carcinoma, colon cancer, early onset of other adenocarcinomas or other childhood cancers) Based upon our criteria and assessment of the peer-reviewed literature, genetic testing for mutations in other high to moderate and low penetrance genes associated with breast and ovarian cancer (e.g., ATM, BARD1, BRIP1, CDH1, CHEK2, NBN, PALB2, RAD50, RAD51C, and STK11) which are part of next generation sequencing panels (e.g., BRCAPlus, BreastNext, CancerNext, OvaNext, Panexia, Melaris, Prolaris, and myrisk Hereditary Cancer ) is considered investigational. Refer to Corporate Medical Policy # regarding Genetic Testing for Specific Diseases. Refer to Corporate Medical Policy Genetic Testing for Hereditary BRCA Mutations.
3 PAGE: 3 OF: 7 Refer to Corporate Medical Policy # regarding Genetic Testing for Inherited Susceptibility to Colorectal Cancer. POLICY GUIDELINES: I. A first-degree relative is a blood relative with whom an individual shares approximately 50% of her/his genes (parents, full siblings, and children). A second-degree relative is a blood relative with whom an individual shares approximately 25% of her/his genes (grandparents, grandchildren, aunts, uncles, nephews, nieces and half-siblings). A third-degree relative is a blood relative with whom an individual shares approximately 12.5% of her/his genes (great-grandparents, great-grandchildren, great-aunts, great-uncles, first cousin, and grand-niece or nephew.) II. For the majority of families in whom mutation status is unknown, it is best to consider testing an affected family member first, especially a family member with early-onset disease, bilateral disease, or multiple primaries, because that individual is most likely to test positive. The testing of unaffected family members may be considered when no affected member is available. III. It is recommended that unaffected individuals with a strong family history who do not meet criteria for BRCA testing or who test negative be referred to an adequately trained health care professional (genetic counselor) to provide appropriate genetic risk assessment to determine the individual s risk for developing cancer. IV. Genetic testing in the absence of a personal or family history of breast cancer is not medically appropriate due to the low probability of detecting a mutation and the considerable psychological impact that may be involved. Widespread screening of specific sub-populations is not endorsed. V. If the genetic test is being done for knowledge only and that knowledge will not alter management or treatment of the patient or family member then the testing is not medically appropriate. VI. The Health Plan and its employees adhere to all state and federal laws concerning the confidentiality of genetic testing and the results of genetic testing. All records, findings and results of any genetic test performed on any person shall be deemed confidential and shall not be disclosed without the written informed consent of the person to whom such genetic test relates. This information shall not be released to any person or organization not specifically authorized by the individual subject of the test. VII. Laboratories performing clinical tests must be certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA). DESCRIPTION: Cowden Syndrome Cowden syndrome is an autosomal dominant disorder associated with germline mutations in the PTEN (phosphatase and tensin homolog) tumor suppressor gene. It is considered to be part of the spectrum of PTEN hamartoma tumor syndromes (PHTS) which also includes Bannayan-Riley-Ruvalcaba syndrome (BRRS), PTEN-related Proteus syndromes (PS), and Proteus-like syndrome (PLS). Additional clinical syndromes related to germline mutations in PTEN include Lermitte-Duclos disease and autism spectrum disorders with macrocephaly, both of which have been associated with Cowden syndrome. Cowden syndrome has been conservatively estimated to occur in 1 in 200,000, with an estimated penetrance of 80%. CS is associated with multiple hamartomas and/or cancerous lesions in various organs and tissues, including the skin, mucous membranes, breast, thyroid, endometrium and brain. Women diagnosed with CS have a high risk of benign fibrocystic breast disease and their lifetime risk of breast cancer has been estimated at 25-50% with an average age of diagnosis of years. LiFraumeni Syndrome Li Fraumeni syndrome (LFS) is a rare hereditary cancer syndrome associated with germline TP53 gene mutations. It has been estimated to be involved in only 1% hereditary breast cancer cases, but results from recent studies show that it may be more common than previously believed. Germline mutations in the TP53 gene have been observed in over 50% of families meeting the classical definition of LFS. LFS is a highly penetrant cancer syndrome and is characterized by a wide spectrum of neoplasms occurring at a young age. It is associated with soft tissue sarcomas, osteosarcomas
4 PAGE: 4 OF: 7 (Ewing s sarcoma less likely in cases of LFS), premenopausal breast cancer, acute leukemia, colon cancer, adrenal cortex and brain tumors. The core LFS tumors are noted to be sarcoma, breast cancer, adrenocortical tumors, and certain brain tumors because they are the more predominant cancers in this syndrome. Individuals with LFS often present with certain cancers in early childhood and have an increased risk of developing multiple cancers in their lifetime. Other Gene Mutations Mutations in genes other than the BRCA1 and BRCA2 have been reported to be associated with an increased risk of breast cancer. It is now possible to test for multiple breast and/or ovarian cancer susceptibility genes in parallel using multigene or multiplex panels. High-penetrance gene mutations such as TP53 and PTEN are thought to account for a large proportion of familial breast cancers while gene mutations in CDH1 and STK11have been associated with hereditary diffuse gastric cancer, which includes an increased risk for lobular breast cancer and Peutz-Jeghers syndrome, respectively (refer to Corporate Medical Policy # regarding Genetic Testing for Inherited Susceptibility to Colorectal Cancer). Mutations in genes such as CHEK2, BRIP1, RAD51C, and PALB2, among other mutations have been confirmed to be associated with increased risk and at this time are considered to be genes with low- or moderate-penetrance. Those high penetrance and low-and moderate penetrance genes listed above are examples of the mutations included in the multiplex panels. These panels may be resource efficient in terms of time and costs, but currently there is no consensus on recommendations for optimal management or surveillance approaches for carrier of these lower or moderate penetrance genes and there is no data available to address cancer risk assessments in individuals who are found to carry multiple gene mutations with moderate penetrance. Additional genetic counseling approaches must be vetted and developed in order to adequately address the limitations and implications associated with interpretation of multiplex testing results. Consequently these multiplex panels are not yet considered part of standard clinical practice. Examples of panels include the BreastNext, CancerNext, and OvaNext panels offered by Ambry Genetics while Myriad Genetics offers the myrisk Hereditary Cancer panel as well as, Prolaris, Melaris, and Panexia panels. RATIONALE: A systematic review by Pilarski et al., was conducted related to the clinical features reported in individuals with a PTEN mutation, and revised diagnostic criteria were proposed. The authors concluded that there was insufficient evidence to support inclusion of benign breast disease, uterine fibroids, or genitourinary malformations as diagnostic criteria. There was sufficient evidence to include autism spectrum disorders, colon cancer, esophageal glycogenic acanthosis, penile macules, renal cell carcinoma, testicular lipomatosis and vascular anomalies, and these clinical features are included in Cowden syndrome testing minor criteria in National Comprehensive Cancer Network (NCCN) guidelines. The International Cowden Consortium criteria for Cowden syndrome have been updated several times since 1996 and is the basis for the National Comprehensive Cancer Network (NCCN) (2014) criteria for PTEN mutation testing. The NCCN criteria have recently revised the list of criteria associated with the syndrome as well as the combinations of criteria that establish which individuals are candidates for PTEN gene mutation testing. The criteria have been divided into 3 categories depending on diagnostic features, major and minor features associated with Cowden s syndrome. In addition, a first-degree relative of an affected individual with one or more major, or two or more minor criteria, along with a relative diagnosed with Cowden syndrome or Bannayan-Riley-Ruvalcaba syndrome (who has not been tested) would also meet the threshold for PTEN testing. The National Comprehensive Cancer Network (NCCN) (2014) for Genetic/familial high-risk assessment: breast and ovarian utilizes both the classic Li Fraumeni clinical criteria and the Chompret criteria when recommending genetic testing for a mutation in the TP53 gene. The NCCN panel also included lung bronchoalveolar cancer and leukemia as one of the core tumor types. Testing individuals with choroid plexus carcinoma diagnosed at any age and regardless of family history based upon high incidence of TP53 mutations found in patients with this rare form of brain tumor has also been recommended. Women with early-onset breast cancer (age of diagnosis 35 years or younger) with or without core history of tumor types are another group for whom TP53 mutation testing may be considered. A member of a family with a known TP53 mutation is considered to be at sufficient risk to warrant gene mutation testing, even in the absence of any other risk factors.
5 PAGE: 5 OF: 7 The National Comprehensive Cancer Network (NCCN) (2014) include new genetic testing panels using next generation sequencing for hereditary breast, ovarian and other cancers in the Genetic/familial High-Risk Assessment: Breast and Ovarian (2014) guidelines. The guidelines state that these panels are intended for individuals who have tested negative for high penetrance genes (BRCA1/2) and for those whose family history is suggestive of more than one syndrome. Limitations of these panels include unknown percentage of variants of unknown significance, uncertainty of the level of risk associated with most of these genes, and lack of clear guidelines on risk management of carriers of some of these mutations. NCCN recommends that these multigene hereditary cancer panels should only be ordered in consultation with a cancer genetics professional. CODES: Number Description Eligibility for reimbursement is based upon the benefits set forth in the member s subscriber contract. CODES MAY NOT BE COVERED UNDER ALL CIRCUMSTANCES. PLEASE READ THE POLICY AND GUIDELINES STATEMENTS CAREFULLY. Codes may not be all inclusive as the AMA and CMS code updates may occur more frequently than policy updates. CPT: PTEN (phosphatase and tensin homolog) (eg, Cowden syndrome, PTEN hamartoma tumor syndrome) gene analysis; full sequence analysis HCPCS: PTEN (phosphatase and tensin homolog) (eg, Cowden syndrome, PTEN hamartoma tumor syndrome) gene analysis; known familial variant PTEN (phosphatase and tensin homolog) (eg, Cowden syndrome, PTEN hamartoma tumor syndrome) gene analysis; duplication/deletion variant No specific code(s) Copyright 2014 American Medical Association, Chicago, IL ICD9: Malignant neoplasm female breast (code range) Malignant neoplasm male breast (code range) Malignant neoplasm of ovary Malignant neoplasm of ovary Secondary malignant neoplasm of ovary Secondary malignant neoplasm of breast Carcinoma insitu of breast Carcinoma insitu of other and unspecified female genital organs (includes ovary) V10.3 Personal history malignant neoplasm of breast (classifiable to 174 & 175) V10.43 Personal history malignant neoplasm of ovary V16.3 Family history of malignant neoplasm breast (classifiable to 174) V16.41 Family history of malignant neoplasm ovary V84.01 Genetic susceptibility to malignant neoplasm of breast ICD10: C C Malignant neoplasm of breast (code range) C56.1-C56.9 C79.60-C79.62 Malignant neoplasm of ovary (code range) Secondary malignant neoplasm of ovary (code range) C79.81 Secondary malignant neoplasm of breast
6 PAGE: 6 OF: 7 REFERENCES: D05.00-D05.02 D05.10-D05.12 D05.80-D05.92 D07.30-D07.39 Lobular carcinoma in situ of breast (code range) Intraductal carcinoma in situ of breast (code range) Carcinoma in situ of breast, specified, unspecified (code range) Carcinoma in situ of other and unspecified female genital organs (code range) Z15.01 Genetic susceptibility to malignant neoplasm of breast Z80.3 Family history of malignant neoplasm of breast Z80.41 Family history of malignant neoplasm of ovary Z85.3 Personal history of malignant neoplasm of breast Z85.43 Personal history of malignant neoplasm of ovary Bird TD. Hereditary Ataxia Overview. Gene Reviews. Initial Posting 1998 Oct 28; Last update: 2014 Feb 27. Funded by the NIH.[ accessed 3/10/14. Coulet F, et al. Germline RAD51C mutations in ovarian cancer susceptibility. Clin Genet 2013 Apr;83(4): Cybulski C, et al. Risk of breast cancer in women with a CHEK2 mutation with and without a family history of breast cancer. J Clin Oncol 2011 Oct 1;29(28): Eng C. PTEN Hamartoma Tumor Syndrome (PHTS). Gene Reviews. Initial Posting 2001 Nov 29; Last update: 2014 Jan 23. Funded by the NIH. [ accessed 3/10/14. *Eng C. Will the real Cowden syndrome please stand up: revised diagnostic criteria. J Med Genet 2000;37(11): Hayes, Inc. Hayes Medical Technology Report. Genetic Testing for Susceptibility To Breast Cancer. Lansdale, PA: Hayes, Inc.; December 20, Hayes, Inc. Hayes Medical Technology Report. Genetic Testing for Susceptibility To Ovarian Cancer. Lansdale, PA: Hayes, Inc.; February 28, 2005.Hollestelle A, et al. Discovering moderate-risk breast cancer susceptibility genes. Curr Opin Genet Dev 2010 Jun;20(3): Kobayashi H, et al. Hereditary breast and ovarian cancer susceptibility genes (review). Oncol Rep 2013 Sep;30(3): Limacher JM et al. Two metachronous tumors in the radiotherapy fields of a patient with Li-Fraumeni syndrome. Int J Cancer 2001;96(4): Mahdi KM, et al. Hereditary genes and SNPs associated with breast cancer. Asian Pac J Prev 2013;14(6): Pennington KP, et al. BRCA1, TP53, and CHEK2 germline mutations in uterine serous carcinoma. Cancer 2013 Jan 15;119(2): Petrucelli N, et al. BRCA1 and BRCA2 Hereditary Breast and Ovarian Cancer. Gene Reviews. Initial Posting 1998 Sep 04; Last update: 2013 Sep 26. Funded by the NIH.[ accessed 3/10/14. Pierce LJ, et al. Radiotherapy in the treatment of hereditary breast cancer. Semin Radiat Oncol 2011 Jan;21(1): Pilarski R, et al. Predicting PTEN mutations: an evaluation of Cowden syndrome and Bannayan-Rile-Ruvalcaba syndrome clinical features. J Med Genet 2011;48(8): Rath MG, et al. Prevalence of TP53 germline mutations in HER2+ breast cancer patients. Breast Cancer Res Treat 2013;139(1): Schneider K, et al. Li-Fraumeni Syndrome. Gene Reviews. Initial Posting 1999 Jan 19; Last update: 2013 Apr 11. Funded by the NIH. [ accessed 3/10/14.
7 PAGE: 7 OF: 7 Weischer M, et al. CHEK2*1100delC genotyping for clinical assessment of breast cancer risk: meta-analyses of 26,000 patient cases and 27,000 controls. J Clin Oncol 2008; 26(4): Zhang B, et al. Genetic variants associated with breast-cancer risk: comprehensive research synopsis, meta-analysis, and epidemiological evidence. Lancet Oncol May 2011; 12(5): * key article KEY WORDS: PTEN, Cowden Syndrome, TP53, Li Fraumeni Syndrome, BreastNext, CancerNext, OvaNext, MyRisk Hereditary Cancer, Melaris, Prolaris, Panexia. CMS COVERAGE FOR MEDICARE PRODUCT MEMBERS There is currently no National Coverage Determination (NCD) or Local Coverage Determination (LCD) for genetic testing for
MEDICAL POLICY SUBJECT: GENETIC TESTING FOR SUSCEPTIBILITY TO HEREDITARY CANCERS EFFECTIVE DATE: 06/19/14 REVISED DATE: 10/15/15, 10/20/16, 01/18/18
 MEDICAL POLICY SUBJECT: GENETIC TESTING FOR SUSCEPTIBILITY REVISED DATE: 10/15/15, 10/20/16, 01/18/18 PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy
MEDICAL POLICY SUBJECT: GENETIC TESTING FOR SUSCEPTIBILITY REVISED DATE: 10/15/15, 10/20/16, 01/18/18 PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy
Prior Authorization. Additional Information:
 Genetic Testing for Cowden Syndrome - PTEN Gene MP9488 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic
Genetic Testing for Cowden Syndrome - PTEN Gene MP9488 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic
Cowden Syndrome PTEN Hamartoma Tumor Syndrome. ACCME/Disclosure. 1. Background. Outline
 MASSACHUSETTS GENERAL HOSPITAL HARVARD MEDICAL SCHOOL PATHOLOGY Cowden Syndrome PTEN Hamartoma Tumor Syndrome ACCME/Disclosure Vania Nosé, MD, PhD Professor of Pathology Director of Anatomic Pathology
MASSACHUSETTS GENERAL HOSPITAL HARVARD MEDICAL SCHOOL PATHOLOGY Cowden Syndrome PTEN Hamartoma Tumor Syndrome ACCME/Disclosure Vania Nosé, MD, PhD Professor of Pathology Director of Anatomic Pathology
Corporate Medical Policy
 Corporate Medical Policy Genetic Testing for PTEN Hamartoma Tumor Syndrome File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_pten_hamartoma_tumor_syndrome 4/2013
Corporate Medical Policy Genetic Testing for PTEN Hamartoma Tumor Syndrome File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_pten_hamartoma_tumor_syndrome 4/2013
BRCAplus. genetic testing for hereditary breast cancer
 BRCAplus genetic testing for hereditary breast cancer Developed in collaboration with Fox Chase Cancer Center and the Arcadia University Genetic Counseling Program. Causes of Hereditary Breast Cancer familial
BRCAplus genetic testing for hereditary breast cancer Developed in collaboration with Fox Chase Cancer Center and the Arcadia University Genetic Counseling Program. Causes of Hereditary Breast Cancer familial
Populations Interventions Comparators Outcomes Individuals: With clinical signs of a PTEN hamartoma tumor syndrome. are:
 Protocol Genetic Testing for PTEN Hamartoma Tumor Syndrome (20488) Medical Benefit Effective Date: 07/01/14 Next Review Date: 05/18 Preauthorization Yes Review Dates: 05/13, 05/14, 05/15, 05/16, 05/17
Protocol Genetic Testing for PTEN Hamartoma Tumor Syndrome (20488) Medical Benefit Effective Date: 07/01/14 Next Review Date: 05/18 Preauthorization Yes Review Dates: 05/13, 05/14, 05/15, 05/16, 05/17
MEDICAL POLICY SUBJECT: GENETIC TESTING FOR HEREDITARY BRCA MUTATIONS. POLICY NUMBER: CATEGORY: Laboratory Test
 MEDICAL POLICY SUBJECT: GENETIC TESTING FOR HEREDITARY BRCA MUTATIONS POLICY NUMBER: 2.02.06 CATEGORY: Laboratory Test EFFECTIVE DATE: 01/17/02 REVISED DATE: 08/21/03, 09/16/04, 08/18/05, 06/15/06, 04/17/14,
MEDICAL POLICY SUBJECT: GENETIC TESTING FOR HEREDITARY BRCA MUTATIONS POLICY NUMBER: 2.02.06 CATEGORY: Laboratory Test EFFECTIVE DATE: 01/17/02 REVISED DATE: 08/21/03, 09/16/04, 08/18/05, 06/15/06, 04/17/14,
GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer
 GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer Causes of Hereditary Ovarian and Uterine Cancer uterine cancer ovarian cancer Sporadic 75-90% Sporadic 70-80% Hereditary, 5% Lynch syndrome
GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer Causes of Hereditary Ovarian and Uterine Cancer uterine cancer ovarian cancer Sporadic 75-90% Sporadic 70-80% Hereditary, 5% Lynch syndrome
Result Navigator. Positive Test Result: PTEN. After a positive test result, there can be many questions about what to do next. Navigate Your Results
 Result Navigator Positive Test Result: PTEN Positive test results identify a change, or misspelling, of DNA that is known or predicted to cause an increased risk for cancer. DNA is the blueprint of life
Result Navigator Positive Test Result: PTEN Positive test results identify a change, or misspelling, of DNA that is known or predicted to cause an increased risk for cancer. DNA is the blueprint of life
GYNplus. genetic testing for hereditary ovarian and/or uterine cancer
 GYNplus genetic testing for hereditary ovarian and/or uterine cancer What Are the Causes of Hereditary Ovarian and Uterine Cancer? uterine cancer ovarian cancer sporadic 70-80% hereditary 5% Lynch syndrome
GYNplus genetic testing for hereditary ovarian and/or uterine cancer What Are the Causes of Hereditary Ovarian and Uterine Cancer? uterine cancer ovarian cancer sporadic 70-80% hereditary 5% Lynch syndrome
MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers
 POLICY: PG0067 ORIGINAL EFFECTIVE: 07/30/02 LAST REVIEW: 01/25/18 MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers GUIDELINES This policy does not certify benefits or authorization of benefits,
POLICY: PG0067 ORIGINAL EFFECTIVE: 07/30/02 LAST REVIEW: 01/25/18 MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers GUIDELINES This policy does not certify benefits or authorization of benefits,
Multigene Panel Testing for Hereditary Cancer Risk
 Multigene Panel Testing for Hereditary Cancer Risk Dana Zakalik, M.D. Director, Nancy and James Grosfeld Cancer Genetics Center Professor, OUWB Medical School MCC Annual Meeting November 4, 2015 Outline
Multigene Panel Testing for Hereditary Cancer Risk Dana Zakalik, M.D. Director, Nancy and James Grosfeld Cancer Genetics Center Professor, OUWB Medical School MCC Annual Meeting November 4, 2015 Outline
Related Policies None
 Medical Policy MP 2.04.88 BCBSA Ref. Policy: 2.04.88 Last Review: 02/26/2018 Effective Date: 02/26/2018 Section: Medicine Related Policies None DISCLAIMER Our medical policies are designed for informational
Medical Policy MP 2.04.88 BCBSA Ref. Policy: 2.04.88 Last Review: 02/26/2018 Effective Date: 02/26/2018 Section: Medicine Related Policies None DISCLAIMER Our medical policies are designed for informational
Carol Christianson, MS, CGC Genetic Counselor West Michigan Cancer Center
 Carol Christianson, MS, CGC Genetic Counselor West Michigan Cancer Center Following this presentation you will be able to: Identify cancer survivors in your practice who might benefit from genetic counseling
Carol Christianson, MS, CGC Genetic Counselor West Michigan Cancer Center Following this presentation you will be able to: Identify cancer survivors in your practice who might benefit from genetic counseling
Hereditary Breast and Ovarian Cancer Rebecca Sutphen, MD, FACMG
 Hereditary Breast and Ovarian Cancer 2015 Rebecca Sutphen, MD, FACMG Among a consecutive series of 11,159 women requesting BRCA testing over one year, 3874 responded to a mailed survey. Most respondents
Hereditary Breast and Ovarian Cancer 2015 Rebecca Sutphen, MD, FACMG Among a consecutive series of 11,159 women requesting BRCA testing over one year, 3874 responded to a mailed survey. Most respondents
6/8/17. Genetics 101. Professor, College of Medicine. President & Chief Medical Officer. Hereditary Breast and Ovarian Cancer 2017
 Genetics 101 Hereditary Breast and Ovarian Cancer 2017 Rebecca Sutphen, MD, FACMG Professor, College of Medicine President & Chief Medical Officer INVASIVE CANCER GENETICALLY ALTERED CELL HYPERPLASIA DYSPLASIA
Genetics 101 Hereditary Breast and Ovarian Cancer 2017 Rebecca Sutphen, MD, FACMG Professor, College of Medicine President & Chief Medical Officer INVASIVE CANCER GENETICALLY ALTERED CELL HYPERPLASIA DYSPLASIA
Genetic Risk Assessment for Cancer
 Genetic Risk Assessment for Cancer Jennifer Siettmann, MS CGC Certified Genetic Counselor/Cancer Risk Counselor Banner Good Samaritan Cancer Screening & Prevention Program Objectives Describe the role
Genetic Risk Assessment for Cancer Jennifer Siettmann, MS CGC Certified Genetic Counselor/Cancer Risk Counselor Banner Good Samaritan Cancer Screening & Prevention Program Objectives Describe the role
 BUY ONE GENE, GET ONE 30 FREE! G E N E P A N E L T E S T I N G F O R H E R E D I T A R Y C A N C E R : G E T T I N G I T R I G H T THIS COULD BE YOU ONE DAY SOON: Starting out in your new OB/Gyn practice
BUY ONE GENE, GET ONE 30 FREE! G E N E P A N E L T E S T I N G F O R H E R E D I T A R Y C A N C E R : G E T T I N G I T R I G H T THIS COULD BE YOU ONE DAY SOON: Starting out in your new OB/Gyn practice
Hereditary Cancer Risk Assessment for Gynecological Cancers. FarrNezhatMD.com
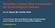 Hereditary Cancer Risk Assessment for Gynecological Cancers FarrNezhatMD.com Image credit: PLOS blogs 5-10% hereditary 10-20% 70-80% sporadic Genetic Changes and Cancer Cancer begins with a genetic
Hereditary Cancer Risk Assessment for Gynecological Cancers FarrNezhatMD.com Image credit: PLOS blogs 5-10% hereditary 10-20% 70-80% sporadic Genetic Changes and Cancer Cancer begins with a genetic
Genetic Testing for BRCA1 and BRCA2 Genes
 Genetic Testing for BRCA1 and BRCA2 Genes MP9478 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic counseling
Genetic Testing for BRCA1 and BRCA2 Genes MP9478 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic counseling
Identification of patients suggestive of hereditary breast and ovarian cancer syndrome that warrants further professional evaluation.
 Allina Breast Program Committee Consensus Guidelines These guidelines apply to clinical interventions that have well-documented outcomes, but whose outcomes are not clearly desirable to all patients Identification
Allina Breast Program Committee Consensus Guidelines These guidelines apply to clinical interventions that have well-documented outcomes, but whose outcomes are not clearly desirable to all patients Identification
Clinical Cancer Genetics
 Clinical Cancer Genetics Lisen Axell, MS, CGC University of Colorado Cancer Center Individuals with cancer Making surgical decisions (lump vs. mast) Making treatment decisions (XRT) Concerns for additional
Clinical Cancer Genetics Lisen Axell, MS, CGC University of Colorado Cancer Center Individuals with cancer Making surgical decisions (lump vs. mast) Making treatment decisions (XRT) Concerns for additional
GENETIC MANAGEMENT OF A FAMILY HISTORY OF BREAST AND / OR OVARIAN CANCER. Dr Abhijit Dixit. Family Health Clinical Genetics
 GENETIC MANAGEMENT OF A FAMILY HISTORY OF BREAST AND / OR OVARIAN CANCER Full Title of Guideline: Author (include email and role): Division & Speciality: Scope (Target audience, state if Trust wide): Review
GENETIC MANAGEMENT OF A FAMILY HISTORY OF BREAST AND / OR OVARIAN CANCER Full Title of Guideline: Author (include email and role): Division & Speciality: Scope (Target audience, state if Trust wide): Review
Assessing Your Patient s Breast Cancer Risk: Is Genetic Testing Necessary?
 May 16, 2016 Assessing Your Patient s Breast Cancer Risk: Is Genetic Testing Necessary? Presenter: Emily Kuchinsky, MS, CGC 1 Experiences with Genetic Testing Adverse Events in Cancer Genetic Testing:
May 16, 2016 Assessing Your Patient s Breast Cancer Risk: Is Genetic Testing Necessary? Presenter: Emily Kuchinsky, MS, CGC 1 Experiences with Genetic Testing Adverse Events in Cancer Genetic Testing:
10/21/ New Release, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
Genetic Testing: who, what, why?
 Genetic Testing: who, what, why? Gina Westhoff MD LMG Gynecologic Oncology March 16, 2019 Disclosures Speaker for Merck (unrelated to today s topic) Objectives Determine who should undergo genetic risk
Genetic Testing: who, what, why? Gina Westhoff MD LMG Gynecologic Oncology March 16, 2019 Disclosures Speaker for Merck (unrelated to today s topic) Objectives Determine who should undergo genetic risk
Genetic Testing for Li-Fraumeni Syndrome
 Protocol Genetic Testing for Li-Fraumeni Syndrome (204101) Medical Benefit Effective Date: 10/01/14 Next Review Date: 07/18 Preauthorization Yes Review Dates: 07/14, 07/15, 07/16, 07/17 Preauthorization
Protocol Genetic Testing for Li-Fraumeni Syndrome (204101) Medical Benefit Effective Date: 10/01/14 Next Review Date: 07/18 Preauthorization Yes Review Dates: 07/14, 07/15, 07/16, 07/17 Preauthorization
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: July 15, 2018 Related Policies: None Genetic Testing for PTEN Hamartoma Tumor Syndrome Description The PTEN hamartoma tumor syndrome (PHTS) includes several syndromes
FEP Medical Policy Manual Effective Date: July 15, 2018 Related Policies: None Genetic Testing for PTEN Hamartoma Tumor Syndrome Description The PTEN hamartoma tumor syndrome (PHTS) includes several syndromes
Genetic Testing for Familial Gastrointestinal Cancer Syndromes. C. Richard Boland, MD La Jolla, CA January 21, 2017
 Genetic Testing for Familial Gastrointestinal Cancer Syndromes C. Richard Boland, MD La Jolla, CA January 21, 2017 Disclosure Information C. Richard Boland, MD I have no financial relationships to disclose.
Genetic Testing for Familial Gastrointestinal Cancer Syndromes C. Richard Boland, MD La Jolla, CA January 21, 2017 Disclosure Information C. Richard Boland, MD I have no financial relationships to disclose.
Predictive and Diagnostic Testing for Cancer in Women. Aparna Rajadhyaksha MD
 Predictive and Diagnostic Testing for Cancer in Women Aparna Rajadhyaksha MD Hereditary Cancer s in Women BRCA1 &2 Other Breast Cancer Genes Li Fraumeni PTEN CHEK2 BRCA1&2 t BRCA1 is part of a complex
Predictive and Diagnostic Testing for Cancer in Women Aparna Rajadhyaksha MD Hereditary Cancer s in Women BRCA1 &2 Other Breast Cancer Genes Li Fraumeni PTEN CHEK2 BRCA1&2 t BRCA1 is part of a complex
Policy Specific Section: Medical Necessity and Investigational / Experimental. October 15, 1997 October 9, 2013
 Medical Policy Genetic Testing for Hereditary Breast and/or Ovarian Cancer Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date:
Medical Policy Genetic Testing for Hereditary Breast and/or Ovarian Cancer Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date:
WHAT IS A GENE? CHROMOSOME DNA PROTEIN. A gene is made up of DNA. It carries instructions to make proteins.
 WHAT IS A GENE? CHROMOSOME E GEN DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
WHAT IS A GENE? CHROMOSOME E GEN DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
Reference #: SYS-PC-VPCI-CG-002. Origination Date: June 2012 Next Review Date: April 2019 Effective Date: April 2016
 Oncology Service Line-Allina Health System-wide Consensus Guidelines: Identification of Breast Cancer Patients at Risk for Inherited Cancer Risks These guidelines apply to clinical interventions that have
Oncology Service Line-Allina Health System-wide Consensus Guidelines: Identification of Breast Cancer Patients at Risk for Inherited Cancer Risks These guidelines apply to clinical interventions that have
Germline Testing for Hereditary Cancer with Multigene Panel
 Germline Testing for Hereditary Cancer with Multigene Panel Po-Han Lin, MD Department of Medical Genetics National Taiwan University Hospital 2017-04-20 Disclosure No relevant financial relationships with
Germline Testing for Hereditary Cancer with Multigene Panel Po-Han Lin, MD Department of Medical Genetics National Taiwan University Hospital 2017-04-20 Disclosure No relevant financial relationships with
Genetic Testing for BRCA1 and BRCA2 Genes
 Genetic Testing f BRCA1 and BRCA2 Genes MP9478 Covered Service: Pri Authization Required: Additional Infmation: Yes when meets criteria below Yes--as shown below Pre and post-test genetic counseling is
Genetic Testing f BRCA1 and BRCA2 Genes MP9478 Covered Service: Pri Authization Required: Additional Infmation: Yes when meets criteria below Yes--as shown below Pre and post-test genetic counseling is
Genetic Risk Assessment for Cancer
 Genetic Risk Assessment for Cancer Jennifer Siettmann, MS CGC Certified Genetic Counselor Banner MD Anderson Cancer Center Objectives Describe the role of genetic counseling and genetic testing in patient
Genetic Risk Assessment for Cancer Jennifer Siettmann, MS CGC Certified Genetic Counselor Banner MD Anderson Cancer Center Objectives Describe the role of genetic counseling and genetic testing in patient
WHAT IS A GENE? CHROMOSOME DNA PROTEIN. A gene is made up of DNA. It carries instructions to make proteins.
 WHAT IS A GENE? CHROMOSOME GENE DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
WHAT IS A GENE? CHROMOSOME GENE DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
Corporate Medical Policy
 Corporate Medical Policy Genetic Cancer Susceptibility Panels Using Next Generation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_cancer_susceptibility_panels_using_next_generation_sequencing
Corporate Medical Policy Genetic Cancer Susceptibility Panels Using Next Generation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_cancer_susceptibility_panels_using_next_generation_sequencing
ASSESSING THE PRACTICES OF GENETIC COUNSELORS REGARDING HEAD CIRCUMFERENCE MEASUREMENT IN HEREDITARY CANCER ASSESSMENT. Amanda Sue Matchette
 ASSESSING THE PRACTICES OF GENETIC COUNSELORS REGARDING HEAD CIRCUMFERENCE MEASUREMENT IN HEREDITARY CANCER ASSESSMENT by Amanda Sue Matchette Bachelor of Science, Susquehanna University, 2009 Master of
ASSESSING THE PRACTICES OF GENETIC COUNSELORS REGARDING HEAD CIRCUMFERENCE MEASUREMENT IN HEREDITARY CANCER ASSESSMENT by Amanda Sue Matchette Bachelor of Science, Susquehanna University, 2009 Master of
GENETIC TESTING FOR HEREDITARY CANCER
 UnitedHealthcare Oxford Clinical Policy GENETIC TESTING FOR HEREDITARY CANCER Policy Number: DIAGNOSTIC 004.27 T2 Effective Date: November 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy GENETIC TESTING FOR HEREDITARY CANCER Policy Number: DIAGNOSTIC 004.27 T2 Effective Date: November 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/108891
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/108891
Medical Policy Manual. Topic: Genetic Testing for Hereditary Breast and/or Ovarian Cancer. Date of Origin: January 27, 2011
 Medical Policy Manual Topic: Genetic Testing for Hereditary Breast and/or Ovarian Cancer Date of Origin: January 27, 2011 Section: Genetic Testing Last Reviewed Date: July 2014 Policy No: 02 Effective
Medical Policy Manual Topic: Genetic Testing for Hereditary Breast and/or Ovarian Cancer Date of Origin: January 27, 2011 Section: Genetic Testing Last Reviewed Date: July 2014 Policy No: 02 Effective
Hereditary Breast and Ovarian Cancers: Risk Management Recommendations. Objectives
 Hereditary Breast and Ovarian Cancers: Risk Management Recommendations Tuya Pal, MD Moffitt Cancer Center Objectives 1 Identify genes associated with Hereditary Breast/Ovarian Cancer, including new and
Hereditary Breast and Ovarian Cancers: Risk Management Recommendations Tuya Pal, MD Moffitt Cancer Center Objectives 1 Identify genes associated with Hereditary Breast/Ovarian Cancer, including new and
CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION
 CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION CentoCancer our most comprehensive oncogenetics panel for hereditary mutations Hereditary pathogenic variants confer an increased risk of developing
CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION CentoCancer our most comprehensive oncogenetics panel for hereditary mutations Hereditary pathogenic variants confer an increased risk of developing
Genetic Testing for Lynch Syndrome
 Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post-test genetic counseling is
Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post-test genetic counseling is
Myriad Financial Assistance Program (MFAP)
 Myriad Financial Assistance Program (MFAP) MEDICAL CRITERIA Hereditary Cancer Products The Myriad Financial Assistance Program offers aid to patients who meet specific financial and medical requirements.
Myriad Financial Assistance Program (MFAP) MEDICAL CRITERIA Hereditary Cancer Products The Myriad Financial Assistance Program offers aid to patients who meet specific financial and medical requirements.
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing
 Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Policy Number: 2.04.93 Last Review: 7/2017 Origination: 7/2013 Next Review: 7/2018 Policy Blue Cross and Blue Shield of Kansas City
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Policy Number: 2.04.93 Last Review: 7/2017 Origination: 7/2013 Next Review: 7/2018 Policy Blue Cross and Blue Shield of Kansas City
Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian
 Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian ANNOUNCER OPEN: Welcome to CME on ReachMD. This segment, entitled Inherited Susceptibility to Cancer: What Do Primary Care Providers
Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian ANNOUNCER OPEN: Welcome to CME on ReachMD. This segment, entitled Inherited Susceptibility to Cancer: What Do Primary Care Providers
Prior Authorization. Additional Information:
 Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Corporate Medical Policy Genetic Testing for Breast and Ovarian Cancer
 Corporate Medical Policy Genetic Testing for Breast and Ovarian Cancer File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_breast_and_ovarian_cancer 8/1997 8/2017
Corporate Medical Policy Genetic Testing for Breast and Ovarian Cancer File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_breast_and_ovarian_cancer 8/1997 8/2017
Genetic Panel Testing and Implications for Cancer Care
 Genetic Panel Testing and Implications for Cancer Care Dana Zakalik, M.D. Nancy and James Grosfeld Cancer Genetics Center Professor, OUWB Medical School MCC Board of Directors Meeting September 28, 2016
Genetic Panel Testing and Implications for Cancer Care Dana Zakalik, M.D. Nancy and James Grosfeld Cancer Genetics Center Professor, OUWB Medical School MCC Board of Directors Meeting September 28, 2016
GEN ETICS AN D GEN OM ICS IN CANCER PREVENTION AN D TREATM EN T. Robert Nathan Slotnick MD PhD Director, Medical Genetics and Genomics
 GEN ETICS AN D GEN OM ICS IN CANCER PREVENTION AN D TREATM EN T Robert Nathan Slotnick MD PhD Director, Medical Genetics and Genomics The Medical/Surgical/Radiation Oncologist s View of Genetics Cancer
GEN ETICS AN D GEN OM ICS IN CANCER PREVENTION AN D TREATM EN T Robert Nathan Slotnick MD PhD Director, Medical Genetics and Genomics The Medical/Surgical/Radiation Oncologist s View of Genetics Cancer
Genetic Testing for Li-Fraumeni Syndrome. Description
 Subject: Genetic Testing for Li-Fraumeni Syndrome Page: 1 of 10 Last Review Status/Date: March 2017 Genetic Testing for Li-Fraumeni Syndrome Description Li-Fraumeni syndrome (LFS) is a cancer predisposition
Subject: Genetic Testing for Li-Fraumeni Syndrome Page: 1 of 10 Last Review Status/Date: March 2017 Genetic Testing for Li-Fraumeni Syndrome Description Li-Fraumeni syndrome (LFS) is a cancer predisposition
Genetic/Familial High-Risk Assessment: Breast and Ovarian
 Clinical Genetic/Familial High-Risk Assessment: Breast and Ovarian Version 1.2005 Continue Assessment: Panel Members * Mary B. Daly, MD, PhD/Chair Fox Chase Cancer Center Jennifer E. Axilbund, MS, CGS
Clinical Genetic/Familial High-Risk Assessment: Breast and Ovarian Version 1.2005 Continue Assessment: Panel Members * Mary B. Daly, MD, PhD/Chair Fox Chase Cancer Center Jennifer E. Axilbund, MS, CGS
Genetic Determinants, Risk Assessment and Management
 Genetic Determinants, Risk Assessment and Management Rachel Rando, MS, CGC Genetic Counselor Hunterdon Regional Cancer Center Flemington, NJ I have no disclosures. Acknowledgements: Staff of Hunterdon
Genetic Determinants, Risk Assessment and Management Rachel Rando, MS, CGC Genetic Counselor Hunterdon Regional Cancer Center Flemington, NJ I have no disclosures. Acknowledgements: Staff of Hunterdon
Hereditary Breast and Ovarian Cancer Screening & Genetic Counseling
 Hereditary Breast and Ovarian Cancer Screening & Genetic Counseling Cecelia Bellcross, PhD, MS, CGC Emory University School of Medicine Department of Human Genetics The First Line of Defense February 21,
Hereditary Breast and Ovarian Cancer Screening & Genetic Counseling Cecelia Bellcross, PhD, MS, CGC Emory University School of Medicine Department of Human Genetics The First Line of Defense February 21,
MEDICAL POLICY SUBJECT: GENETIC TESTING FOR INHERITED SUSCEPTIBILITY TO COLORECTAL CANCER. POLICY NUMBER: CATEGORY: Laboratory Test
 MEDICAL POLICY SUBJECT: GENETIC TESTING FOR INHERITED SUSCEPTIBILITY TO COLORECTAL CANCER POLICY NUMBER: 2.02.11 CATEGORY: Laboratory Test EFFECTIVE DATE: 09/16/99 REVISED DATE: 04/19/01, 05/16/02, 06/19/03,
MEDICAL POLICY SUBJECT: GENETIC TESTING FOR INHERITED SUSCEPTIBILITY TO COLORECTAL CANCER POLICY NUMBER: 2.02.11 CATEGORY: Laboratory Test EFFECTIVE DATE: 09/16/99 REVISED DATE: 04/19/01, 05/16/02, 06/19/03,
The benefit of. knowing. Genetic testing for hereditary cancer. A patient support guide
 The benefit of knowing Genetic testing for hereditary cancer A patient support guide Does cancer run in your family? Cancer is more common in some families. Sometimes cancer is caused by a change in a
The benefit of knowing Genetic testing for hereditary cancer A patient support guide Does cancer run in your family? Cancer is more common in some families. Sometimes cancer is caused by a change in a
Genetic Testing of Inherited Cancer Predisposition Genetic Testing - Oncology
 Genetic Testing of Inherited Cancer Predisposition Genetic Testing - Oncology Policy Number: Original Effective Date: MM.02.010 05/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration
Genetic Testing of Inherited Cancer Predisposition Genetic Testing - Oncology Policy Number: Original Effective Date: MM.02.010 05/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration
Risk Assessment and Risk Management
 Risk Assessment and Risk Management Epworth Benign Breast Disease Symposium Dr Laura Chin-Lenn 12 November 2016 Why identify those at increased risk of breast cancer? Should I be worried? 1 Why identify
Risk Assessment and Risk Management Epworth Benign Breast Disease Symposium Dr Laura Chin-Lenn 12 November 2016 Why identify those at increased risk of breast cancer? Should I be worried? 1 Why identify
A Patient s Guide to Hereditary Cancer. Is Hereditary Cancer Testing Right for You?
 A Patient s Guide to Hereditary Cancer Is Hereditary Cancer Testing Right for You? What is Hereditary Cancer? Most cancers occur in people who do not have a strong family history of that cancer. This is
A Patient s Guide to Hereditary Cancer Is Hereditary Cancer Testing Right for You? What is Hereditary Cancer? Most cancers occur in people who do not have a strong family history of that cancer. This is
Primary Care Approach to Genetic Cancer Syndromes
 Primary Care Approach to Genetic Cancer Syndromes Jason M. Goldman, MD, FACP FAU School of Medicine Syndromes Hereditary Breast and Ovarian Cancer (HBOC) Hereditary Nonpolyposis Colorectal Cancer (HNPCC)
Primary Care Approach to Genetic Cancer Syndromes Jason M. Goldman, MD, FACP FAU School of Medicine Syndromes Hereditary Breast and Ovarian Cancer (HBOC) Hereditary Nonpolyposis Colorectal Cancer (HNPCC)
Prophylactic Mastectomy
 Prophylactic Mastectomy Policy Number: Original Effective Date: MM.06.010 01/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO 08/24/2012 Section: Surgery Place(s) of Service: Inpatient I.
Prophylactic Mastectomy Policy Number: Original Effective Date: MM.06.010 01/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO 08/24/2012 Section: Surgery Place(s) of Service: Inpatient I.
Germline Multigene Panel Testing in Oncology: Genetic Counseling Perspective
 Germline Multigene Panel Testing in Oncology: Genetic Counseling Perspective Sarah L. Campian, MS CGC Certified Genetic Counselor Nancy & James Grosfeld Cancer Genetics Center Objectives Identify patients/families
Germline Multigene Panel Testing in Oncology: Genetic Counseling Perspective Sarah L. Campian, MS CGC Certified Genetic Counselor Nancy & James Grosfeld Cancer Genetics Center Objectives Identify patients/families
Yes when meets criteria below. Dean Health Plan covers when Medicare also covers the benefit.
 Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Why Test for Hereditary Cancer in Preventive Care?
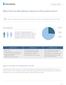 Why Test for Hereditary Cancer in Preventive Care? Millions of people are sidelined by cancer. Wouldn't it be worth it for your patients to know their risk? background HEREDITARY (5-10%) More than 1 in
Why Test for Hereditary Cancer in Preventive Care? Millions of people are sidelined by cancer. Wouldn't it be worth it for your patients to know their risk? background HEREDITARY (5-10%) More than 1 in
Hereditary Cancer Products
 Hereditary Products Integrated BRACAnalysis (BRCA1 and BRCA2 sequencing and large rearrangement testing (BART)), covered when: PERSONAL HISTORY of Breast (must meet at least 1) Diagnosed
Hereditary Products Integrated BRACAnalysis (BRCA1 and BRCA2 sequencing and large rearrangement testing (BART)), covered when: PERSONAL HISTORY of Breast (must meet at least 1) Diagnosed
Test Information Sheet
 OncoGeneDx: Breast Cancer Management Panel Panel Gene List: ATM, BRCA1, BRCA2, CDH1, CHEK2, NBN, PALB2, PTEN, TP53 Clinical Features: In the general population, approximately 1 in 8 women (12%) will develop
OncoGeneDx: Breast Cancer Management Panel Panel Gene List: ATM, BRCA1, BRCA2, CDH1, CHEK2, NBN, PALB2, PTEN, TP53 Clinical Features: In the general population, approximately 1 in 8 women (12%) will develop
HEREDITARY CANCER SYNDROMES: IDENTIFYING THOSE AT RISK
 HEREDITARY CANCER SYNDROMES: IDENTIFYING THOSE AT RISK OCTOBER 20, 2017 JESSICA CARY, MS, RN, CGC cary@newecs.org GENETIC COUNSELOR OBJECTIVES 1) Taking a Cancer Family History 2) Identify patients and/or
HEREDITARY CANCER SYNDROMES: IDENTIFYING THOSE AT RISK OCTOBER 20, 2017 JESSICA CARY, MS, RN, CGC cary@newecs.org GENETIC COUNSELOR OBJECTIVES 1) Taking a Cancer Family History 2) Identify patients and/or
Policy and Procedure. Department: Utilization Management. SNP, CHP, MetroPlus Gold, Goldcare I&II, Market Plus, Essential, HARP
 Retired Date: Page 1 of 9 1. POLICY DESCRIPTION: BRCA 1&2 Genetic Testing 2. RESPONSIBLE PARTIES: Medical Management Administration, Utilization Management, Integrated Care Management, Pharmacy, Claim
Retired Date: Page 1 of 9 1. POLICY DESCRIPTION: BRCA 1&2 Genetic Testing 2. RESPONSIBLE PARTIES: Medical Management Administration, Utilization Management, Integrated Care Management, Pharmacy, Claim
Management of BRCA Positive Breast Cancer. Archana Ganaraj, MD February 17, 2018 UPDATE ON WOMEN S HEALTH
 Management of BRCA Positive Breast Cancer Archana Ganaraj, MD February 17, 2018 UPDATE ON WOMEN S HEALTH The number of American women who have lost their lives to breast cancer outstrips the total number
Management of BRCA Positive Breast Cancer Archana Ganaraj, MD February 17, 2018 UPDATE ON WOMEN S HEALTH The number of American women who have lost their lives to breast cancer outstrips the total number
Breast Cancer Risk Assessment: Genetics, Risk Models, and Screening. Amie Hass, MSN, ARNP, FNP-BC Hall-Perrine Cancer Center
 Breast Cancer Risk Assessment: Genetics, Risk Models, and Screening Amie Hass, MSN, ARNP, FNP-BC Hall-Perrine Cancer Center Disclosure- I DO NOT HAVE any relevant financial interest with any entity producing,
Breast Cancer Risk Assessment: Genetics, Risk Models, and Screening Amie Hass, MSN, ARNP, FNP-BC Hall-Perrine Cancer Center Disclosure- I DO NOT HAVE any relevant financial interest with any entity producing,
Genetic Testing for Li-Fraumeni Syndrome
 Protocol Genetic Testing for Li-Fraumeni Syndrome (204101) Medical Benefit Effective Date: 10/01/18 Next Review Date: 07/19 Preauthorization Yes Review Dates: 07/14, 07/15, 07/16, 07/17, 07/18 Preauthorization
Protocol Genetic Testing for Li-Fraumeni Syndrome (204101) Medical Benefit Effective Date: 10/01/18 Next Review Date: 07/19 Preauthorization Yes Review Dates: 07/14, 07/15, 07/16, 07/17, 07/18 Preauthorization
Hereditary Cancer Update Strengthening Linkages Workshop April 22, 2017
 Hereditary Cancer Update Strengthening Linkages Workshop April 22, 2017 Renée Perrier, MD MSc FRCPC Clinical Assistant Professor University of Calgary, Department of Medical Genetics Medical Director,
Hereditary Cancer Update Strengthening Linkages Workshop April 22, 2017 Renée Perrier, MD MSc FRCPC Clinical Assistant Professor University of Calgary, Department of Medical Genetics Medical Director,
Genetic Testing for Li-Fraumeni Syndrome
 Medical Policy Manual Genetic Testing, Policy No. 82 Genetic Testing for Li-Fraumeni Syndrome Next Review: August 2018 Last Review: August 2017 Effective: January 1, 2018 IMPORTANT REMINDER Medical Policies
Medical Policy Manual Genetic Testing, Policy No. 82 Genetic Testing for Li-Fraumeni Syndrome Next Review: August 2018 Last Review: August 2017 Effective: January 1, 2018 IMPORTANT REMINDER Medical Policies
Germline Genetic Testing for Breast Cancer Risk
 Kathmandu, Bir Hospital visit, August 2018 Germline Genetic Testing for Breast Cancer Risk Evidence-based Genetic Screening Rodney J. Scott Demography in New South Wales (total population ~ 7,000,000)
Kathmandu, Bir Hospital visit, August 2018 Germline Genetic Testing for Breast Cancer Risk Evidence-based Genetic Screening Rodney J. Scott Demography in New South Wales (total population ~ 7,000,000)
Cancer Genomics 101. BCCCP 2015 Annual Meeting
 Cancer Genomics 101 BCCCP 2015 Annual Meeting Objectives Identify red flags in a person s personal and family medical history that indicate a potential inherited susceptibility to cancer Develop a systematic
Cancer Genomics 101 BCCCP 2015 Annual Meeting Objectives Identify red flags in a person s personal and family medical history that indicate a potential inherited susceptibility to cancer Develop a systematic
Information for You and Your Family
 Information for You and Your Family What is Prevention? Cancer prevention is action taken to lower the chance of getting cancer. In 2017, more than 1.6 million people will be diagnosed with cancer in the
Information for You and Your Family What is Prevention? Cancer prevention is action taken to lower the chance of getting cancer. In 2017, more than 1.6 million people will be diagnosed with cancer in the
Corporate Medical Policy
 Corporate Medical Policy Moderate Penetrance Variants Associated with Breast Cancer in File Name: Origination: Last CAP Review: Next CAP Review: Last Review: moderate_penetrance_variants_associated_with_breast_cancer_
Corporate Medical Policy Moderate Penetrance Variants Associated with Breast Cancer in File Name: Origination: Last CAP Review: Next CAP Review: Last Review: moderate_penetrance_variants_associated_with_breast_cancer_
Re: NC Medicaid and NC Health Choice Program coverage of genetic testing for susceptibility to breast and ovarian cancer
 March 23, 2018 North Carolina (NC) Division of Medical Assistance (DMA), Clinical Policy Section C/O Pam Greeson, BSN, RN-C, pam.greeson@dhhs.nc.gov Nurse Consultant, Division of Medical Assistance Practitioners,
March 23, 2018 North Carolina (NC) Division of Medical Assistance (DMA), Clinical Policy Section C/O Pam Greeson, BSN, RN-C, pam.greeson@dhhs.nc.gov Nurse Consultant, Division of Medical Assistance Practitioners,
GeneHealth BreastGene_New qxp_Layout 1 21/02/ :42 Page 3 BreastGene GeneHealth UK
 GeneHealth BreastGene_New 8.2.17.qxp_Layout 1 21/02/2017 16:42 Page 3 BreastGene GeneHealth UK BreastGene What is hereditary breast cancer? Breast cancer is the most common cancer in the UK. Unfortunately
GeneHealth BreastGene_New 8.2.17.qxp_Layout 1 21/02/2017 16:42 Page 3 BreastGene GeneHealth UK BreastGene What is hereditary breast cancer? Breast cancer is the most common cancer in the UK. Unfortunately
Genetic Cancer Susceptibility Panels Using Next Generation Sequencing. Policy Specific Section: September 27, 2013 January 30, 2015
 Medical Policy Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Type: Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date: Effective Date:
Medical Policy Genetic Cancer Susceptibility Panels Using Next Generation Sequencing Type: Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date: Effective Date:
Populations Interventions Comparators Outcomes Individuals: With a personal and/or family history suggesting an inherited cancer syndrome
 Genetic Cancer Susceptibility Panels Using Next- Generation (20493) Medical Benefit Effective Date: 01/01/18 Next Review Date: 09/18 Preauthorization No Review Dates: 09/13, 09/14, 09/15, 09/16, 09/17
Genetic Cancer Susceptibility Panels Using Next- Generation (20493) Medical Benefit Effective Date: 01/01/18 Next Review Date: 09/18 Preauthorization No Review Dates: 09/13, 09/14, 09/15, 09/16, 09/17
The Value of Panel Testing in Inherited Breast Cancer Risk Assessment. Rodney J. Scott Division of Molecular Medicine
 The Value of Panel Testing in Inherited Breast Cancer Risk Assessment Rodney J. Scott Division of Molecular Medicine Mutation Detection Next Generation DNA sequencing has revolutionised mutation detection
The Value of Panel Testing in Inherited Breast Cancer Risk Assessment Rodney J. Scott Division of Molecular Medicine Mutation Detection Next Generation DNA sequencing has revolutionised mutation detection
Therapy-related acute myeloid leukemia with germline TP53 mutation (Li-Fraumeni syndrome) SH Chelsey Deel MD Teresa Scordino MD
 Therapy-related acute myeloid leukemia with germline TP53 mutation (Li-Fraumeni syndrome) SH2017-167 Chelsey Deel MD Teresa Scordino MD Clinical History HPI: 44 year old Caucasian female referred for evaluation
Therapy-related acute myeloid leukemia with germline TP53 mutation (Li-Fraumeni syndrome) SH2017-167 Chelsey Deel MD Teresa Scordino MD Clinical History HPI: 44 year old Caucasian female referred for evaluation
PROVIDER POLICIES & PROCEDURES
 PROVIDER POLICIES & PROCEDURES GENETIC CANCER SUSCEPTIBILITY PANELS USING NEXT GENERATION SEQUENCING The purpose of this document is to assist providers enrolled in the Connecticut Medical Assistance Program
PROVIDER POLICIES & PROCEDURES GENETIC CANCER SUSCEPTIBILITY PANELS USING NEXT GENERATION SEQUENCING The purpose of this document is to assist providers enrolled in the Connecticut Medical Assistance Program
BRCA 1/2. Breast cancer testing THINK ABOUT TOMORROW, TODAY
 BRCA 1/2 Breast cancer testing THINK ABOUT TOMORROW, TODAY 5 10% of patients with breast and/or ovarian cancer have a hereditary form1. For any individual carrying a mutation in BRCA1 or BRCA2, the lifetime
BRCA 1/2 Breast cancer testing THINK ABOUT TOMORROW, TODAY 5 10% of patients with breast and/or ovarian cancer have a hereditary form1. For any individual carrying a mutation in BRCA1 or BRCA2, the lifetime
OBJECTIVES 8/25/2017. An attempt to organize the chaos
 High Risk for Breast Cancer and Genetics: Who? What? Where? When? An attempt to organize the chaos Presented at Winds of Change Conference November 3, 2017 by Carol Hager, MSN, CRNP and Allison Haener,
High Risk for Breast Cancer and Genetics: Who? What? Where? When? An attempt to organize the chaos Presented at Winds of Change Conference November 3, 2017 by Carol Hager, MSN, CRNP and Allison Haener,
Policy Specific Section: Medical Necessity and Investigational / Experimental. October 14, 1998 March 28, 2014
 Medical Policy Genetic Testing for Colorectal Cancer Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date: Effective Date: October
Medical Policy Genetic Testing for Colorectal Cancer Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date: Effective Date: October
Are you at risk of Hereditary Cancer? Your Guide to the Answers
 Are you at risk of Hereditary Cancer? Your Guide to the Answers What is Hereditary Cancer? The genes we are born with may contribute to our risk of developing certain types of cancer, including breast,
Are you at risk of Hereditary Cancer? Your Guide to the Answers What is Hereditary Cancer? The genes we are born with may contribute to our risk of developing certain types of cancer, including breast,
Utilization of BRCA Testing. Breast and Ovarian Cancer in Texas
 Utilization of BRCA Testing in Older Ode Women with Breast and Ovarian Cancer in Texas Ana M. Rodriguez, MD Assistant Professor Department of Obstetrics and Gynecology University of Texas Medical Branch
Utilization of BRCA Testing in Older Ode Women with Breast and Ovarian Cancer in Texas Ana M. Rodriguez, MD Assistant Professor Department of Obstetrics and Gynecology University of Texas Medical Branch
Genetic Testing Today: What Genes Can Tell Us. Living Beyond Breast Cancer Conference Kara N. Maxwell, MD, PhD University of Pennsylvania
 Genetic Testing Today: What Genes Can Tell Us Living Beyond Breast Cancer Conference Kara N. Maxwell, MD, PhD University of Pennsylvania Overview of talk PART 1: Overview of Genetics PART 2: BRCA1/2 PART
Genetic Testing Today: What Genes Can Tell Us Living Beyond Breast Cancer Conference Kara N. Maxwell, MD, PhD University of Pennsylvania Overview of talk PART 1: Overview of Genetics PART 2: BRCA1/2 PART
MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND (TRUS)
 MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND 06/16/05, 05/18/06, 03/15/07, 02/21/08 PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under
MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND 06/16/05, 05/18/06, 03/15/07, 02/21/08 PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under
Genetic/Familial High-Risk Assessment: Breast and Ovarian
 Clinical in Oncology Genetic/Familial High-Risk Assessment: Breast and Ovarian V.1.2009 Continue www.nccn.org Assessment: Panel Members * Mary B. Daly, MD, PhD/Chair Fox Chase Cancer Center Jennifer E.
Clinical in Oncology Genetic/Familial High-Risk Assessment: Breast and Ovarian V.1.2009 Continue www.nccn.org Assessment: Panel Members * Mary B. Daly, MD, PhD/Chair Fox Chase Cancer Center Jennifer E.
Genetic testing and pancreatic disease
 Genetic testing and pancreatic disease February 2 d, 2018 Yale Pancreas Symposium 2018: Multidisciplinary Management of Pancreatic Cancer Xavier Llor, M.D., PhD. Associate Professor of Medicine Co-Director,
Genetic testing and pancreatic disease February 2 d, 2018 Yale Pancreas Symposium 2018: Multidisciplinary Management of Pancreatic Cancer Xavier Llor, M.D., PhD. Associate Professor of Medicine Co-Director,
Clinical Policy Title: Genetic testing for breast and ovarian cancer
 Clinical Policy Title: Genetic testing for breast and ovarian cancer Clinical Policy Number: 02.01.02 Effective Date: September 1, 2013 Initial Review Date: March 21, 2013 Most Recent Review Date: May
Clinical Policy Title: Genetic testing for breast and ovarian cancer Clinical Policy Number: 02.01.02 Effective Date: September 1, 2013 Initial Review Date: March 21, 2013 Most Recent Review Date: May
COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014
 COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014 WENDY MCKINNON, MS, CGC CERTIFIED GENETIC COUNSELOR FAMILIAL CANCER PROGRAM UNIVERSIT Y OF VERMONT MEDICAL CENTER 1 CHARACTERISTICS
COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014 WENDY MCKINNON, MS, CGC CERTIFIED GENETIC COUNSELOR FAMILIAL CANCER PROGRAM UNIVERSIT Y OF VERMONT MEDICAL CENTER 1 CHARACTERISTICS
Understanding Your Positive Result. A guide to understanding your risk and taking action
 Understanding Your Positive Result A guide to understanding your risk and taking action 2017 Myriad Genetics, Inc. 320 Wakara Way, Salt Lake City, Utah 84108 PH: 1-800-469-7423 FX: 801-584-3615 1 PART
Understanding Your Positive Result A guide to understanding your risk and taking action 2017 Myriad Genetics, Inc. 320 Wakara Way, Salt Lake City, Utah 84108 PH: 1-800-469-7423 FX: 801-584-3615 1 PART
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 BRCA1 and BRCA2 Testing Page 1 of 26 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: BRCA1 and BRCA2 Testing Pre-Determination of Services IS REQUIRED by the Member
BRCA1 and BRCA2 Testing Page 1 of 26 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: BRCA1 and BRCA2 Testing Pre-Determination of Services IS REQUIRED by the Member
