Non-invasive techniques for the diagnosis of Helicobacter pylori infection
|
|
|
- Myron Mitchell
- 6 years ago
- Views:
Transcription
1 REVIEW Non-invasive techniques for the diagnosis of Helicobacter pylori infection L. Gatta, C. Ricci, A. Tampieri and D. Vaira Department of Internal Medicine and Gastroenterology, Nuove Patologie, Bologna, Italy Helicobacter pylori infection can be diagnosed by invasive techniques requiring endoscopy and biopsy (histologic examination, culture, polymerase chain reaction), and non-invasive techniques (serology, urea breath test, urine or blood, detection of H. pylori antigen in stool specimen). However, recent studies have demonstrated that a strategy of `testing and treating' for H. pylori in uninvestigated, young (<50 years), dyspeptic patients in primary care is safe and reduces the need for endoscopy. Indeed, a number of clinical guidelines recommend non-invasive testing in dyspeptic patients followed by treatment of H. pylori in primary care based on clinical and economic analyses. Several non-invasive tests are currently available on the market. The choice depends on the clinical circumstances, the likelihood ratio of positive and negative tests, the cost-effectiveness of the testing strategy, and, nally, the availability of the tests. Nevertheless, two non-invasive tests are commonly used: the urea breath test, and the stool antigen test. Keywords Helicobacter pylori, dyspepsie, diagnosis, non invasive tests Accepted 9 December 2002 Clin Microbiol Infect 2003; 9: 489±496 INTRODUCTION In 1983, a report was published of unidenti ed gastric bacteria [1]. Since the discovery of Helicobacter pylori, we have learned much about this pathogen. It is one of the most common causes of chronic infection in humans [2], causes peptic ulcer disease and chronic gastritis, and is also strongly associated with gastric malignancies: indeed, it has been classi ed as a Class I carcinogen [3]. H. pylori infection can be diagnosed by invasive techniques (i.e. requiring endoscopy and biopsy), and non-invasive techniques. Historically, the diagnosis of H. pylori infection has been based on histologic examination of biopsy samples taken during endoscopy, culture, and the rapid urease test. However, endoscopy is expensive, is unpleasant for patients, and carries a small but de nite risk of complications [4]. Thus, the use of Corresponding author and reprint requests: D. Vaira, Department of Internal Medicine and Gastroenterology, Nuove Patologie, Via Massarenti 9, 40138, Bologna, Italy Tel: Fax: vairadin@med.unibo.it non-invasive tests to diagnose H. pylori infection is becoming more frequent. Furthermore, several guidelines recommend the use of non-invasive tests as rst-line treatment in the management of dyspepsia [5±7]. There are several non-invasive tests for the diagnosis of H. pylori infection. Choosing the `right' non-invasive test is not easy, and several aspects need to be considered: sensitivity, speci city, and the post-test probability of having or not having the infection following a positive or negative result [8]. We also have to look carefully at the clinical situation, i.e. pre-test probability of the infection in our population, the age of the patients, their present complaints, and past medical history. Finally, we have to consider economic evaluations that assess the costs and the consequences of the alternative strategies for the detection and/or management of H. pylori infection. There are two different types of non-invasive test: direct and indirect (Figure 1). The direct tests look for direct evidence of the presence of H. pylori. The stool test assesses the presence of bacterial antigens in stool. Indirect tests assess the presence of the infection by evaluating indirect evidence such as the presence of antibodies to H. pylori, or the amount of labeled CO 2 in the breath, an expression of the urease activity of the bacterium. ß 2003 Copyright by the European Society of Clinical Microbiology and Infectious Diseases
2 490 Clinical Microbiology and Infection, Volume 9 Number 6, June 2003 Figure 1 Non-invasive tests. NON-INVASIVE INDIRECT TECHNIQUES Immunologic tests Serologic tests The serologic tests are based on the detection of speci c anti-h. pylori IgG antibodies in the patient's serum. Serology was the rst non-invasive technique, even though, in principle, it has limitations. The most important is that we are not able to distinguish between active infection and a previous `contact'. Antibody levels persist in the blood for long periods of time, and as more and more patients with H. pylori infection are treated, persistent antibodies will lead to false-positive results. A meta-analysis of 21 studies with commercially available ELISA serology kits reported overall sensitivity and speci city of 85% and 79%, respectively [9]. Furthermore, the Medical Devices Agency of Great Britain evaluated a large number of ELISA tests [10]. Five hundred and eighty-eight samples of sera were assessed with 16 different tests. The overall accuracy of the assays averaged 78% (range 68±82%)for all sera. Thus, the accuracy of these tests is no longer adequate to justify their use on clinical or economic grounds. However, there are other elds in which serology can be useful, such as: large epidemiologic surveys, studies on age at acquisition of the infection, and assessment of detection of antibodies against the two important proteins CagA and VacA using different techniques (ELISA, recombinant immunoblot assay, Western blot)for research purpose. CagA and VacA are two important pathologic markers. The former is a 120-kDa protein with high immunogenic power, and its gene (caga)is located in the pathogenicity island (PAI)of the chromosome of H. pylori. Individuals infected with CagA-positive strains of H. pylori have a more severe in ammatory reaction, a greater degree of gastric atrophy and intestinal metaplasia, and a higher incidence of duodenal ulcer and intestinal-type gastric cancer [11]. The vacuolating toxin (VacA)is an 87-kDa protein, and its gene is situated a signi cant distance from the PAI. However, it is secreted by Helicobacter strains, that contain the PAI. It is activated at low ph, is resistant to acid and pepsin, causes vacuolation in cell culture in vitro, and damages mouse gastric epithelium in vivo [12]. A comparison of two anti-caga ELISA tests (HeloriCTX, Eurospital It)showed sensitivity and speci city of 100% and 76%, and 90% and 94%, respectively [13]. Western blotting has been developed into commercial test kits; in recent studies, Helico-blot 2.0 and Recombinant Immunoblot Assay Strip (RIBA)showed good sensitivity and speci city [14]. Near patient tests Near patient tests were developed to provide rapid diagnosis of H. pylori infection in clinics or physicians' of ces. They are technically simple to perform, and most of them currently use one-step tools on a drop of whole blood, while others require separation of serum, which diminishes their usefulness as near patient kits. Nevertheless, general remarks can be made about these tests. The most accurate `blood test' detects serum antibodies from clotted blood taken at venesection. Fingerprick test results can vary when there is dif culty in obtaining a drop of blood. Squeezing the nger can change the hematocrit by mixing tissue uid with the blood sample, changing the concentration of antibody in the serum. Whole blood tests are
3 Gatta et al Non-invasive techniques for diagnosis of H. pylori infection 491 in uenced by chylomicrons, which may affect the permeability of the blood plasma through the various membranes used in diagnostic tests. Recent evidence accumulated from 3805 patients in eight studies performed in 1999±2000 suggests that these tests have considerably lower sensitivity and speci city than originally assumed. The mean sensitivity (weighted for number of patients studied)was 71.1%, and the speci city was 87.6% [15]. Tests on saliva and urine Some studies have looked at saliva and urine as possible non-invasive samples in which to detect antibodies to H. pylori. However, results with the salivary assay have been disappointing (sensitivity 81%, speci city 73%)[16]. Regarding urine, there has been a study reporting sensitivity and speci- city of 86% and 91% in 132 patients [17]. However, these data have not been con rmed by a multicenter European trial using the same urine assay in a large population, where sensitivity and speci city of 89% and 69%, respectively, were reported [18]. Urease activity-based tests Urea breath test H. pylori is a strong urease producer. This enzyme splits urea into ammonia and CO 2. The test is based on this. Patients ingest urea labeled with either 13 Cor 14 C, and the labeled urea comes into contact with the mucosa and diffuses through the mucus towards the H. pylori and the mucosal blood supply. Urea hydrolysis occurring within the mucous layer produces ammonia and labeled CO 2. This is close to the epithelial blood supply, and, because of the concentration gradient, within a few minutes the labeled CO 2 will appear in the breath. Several doses of 14 C-labeled urea have been used in clinical studies, ranging from 37 kbq (1 mci)to 185 kbq (5 mci). Labeled urea is usually given to the patient with a test meal, to delay gastric emptying. After ingestion of the urea, breath samples are collected for up to 20 min by the patient exhaling into a CO 2 -trapping agent (hyamine). The radioactivity of each sample is measured by a scintillation counter, and the results are expressed as a percentage of the administered dose, adjusted for endogenous CO 2 production, or directly as counts/min. The sensitivity and speci city of the test are about 97% and 95%, respectively. However, the 14 C-urea breath test is reported to be unreliable in assessing patients who have had gastric surgery, and in patients who have been receiving proton pump inhibitors or ranitidine [19]. Recently, a new device for 14 C-urea breath testing has been described [20]. It uses a at breath card, which is able to adsorb exhaled CO 2 via chemical bonding. This card is then read using a small analyzer with a slot into which the card is put. This type of device might became a genuine `near patient test', where 14 C-urea breath testing is allowed, since it is easy to perform and has the advantage of being cheaper than 13 C-urea breath testing. However, concern with 14 C usually arises because of its long half-life, and the amount of radiation. Munster et al. reported that approximately 90% of the 14 C from a urea breath test is eliminated as CO 2 in the breath or urine after 3 days; the amount of isotope retained in the body is negligible [21]. Furthermore, the cumulative lifetime radiation exposure from this test has been calculated to be not more than 0.3 mrem/mci, which is considered to be equal to the background radiation that a person is exposed to in 1 day [22]. For this reason, the 1-mCi 14 C dose has been permitted, without restriction, for general use in the USA (Nuclear Radioactive Drug Committee, USA, us10fcr Radioactive drug), while in Europe the 14 C-urea breath test cannot yet be used with children and women of childbearing age. The 13 C-urea breath test is identical to the 14 C-urea breath test, except that 13 C is a non-radioactive isotope of 12 C, and its detection requires a mass spectrometer rather than a scintillation counter. Because 13 C is a naturally occurring isotope, it is measured as a ratio of 12 C, which is the most abundant isotope [23,24]. The 13 C-urea breath test is based on a simple principle: in the presence of H. pylori, isotopically labeled urea is rapidly hydrolyzed by the bacteria's urease, and the released CO 2 exhaled in the expired breath. Since its rst description by Graham et al. [25], who used 350 mg of 13 C-urea, this test has been extensively modi ed, including variations in the dose of labeled urea, sampling time, test meal, and cutoff values. These modi cations have been designed to reduce the duration of the test, improve its accuracy, or reduce the amount of the expensive substrate ( 13 C)used in the test. Tests employing a dose of 75 mg of 13 C-urea have proved to be as accurate as those using higher amounts, and are less expensive. This dose has been increasingly adopted in research studies, as
4 492 Clinical Microbiology and Infection, Volume 9 Number 6, June 2003 well as in most of the commercially available diagnostic kits [26]. Conventional urea breath test protocols use a test meal to delay gastric emptying and to allow time for an even distribution of the 13 C throughout the stomach. However, there is evidence that absence of a test meal does not affect accuracy [27]. Another area in which improvements in the conventional breath test have been possible is the duration of the test. Many breath test protocols require a baseline sample and another sample obtained 30 min after ingestion of the 13 C. Recently, a new type of urea breath test has been described that uses a tablet formulation of 13 C-urea, which allows breath sampling to be performed as early as 10 min after ingestion of the tablet with excellent accuracy [28,29]. Furthermore, the advantage of supplying the urea solution in a tablet is that interference from ureaseproducing bacteria in the oropharynx, which may cause false-positive results in earlier breath test protocols, is avoided [30,31]. Analysis of the results from studies in which the tests were evaluated against an accepted standard con rms their accuracy. In 3643 patients studied in 1999±2000, the weighted means for the sensitivity and speci- city of the urea breath test were 94.7% and 95.7%, respectively [15]. Serum bicarbonate The assessment of 13 C-bicarbonate from serum samples after oral ingestion of 13 C urea is not practicable for a urea breath test [32]. However, it was recently reported to have good accuracy in the post-treatment setting. Ammonia vapor Recently, a new device for assessing the presence of H. pylori infection has been described. It uses the urease activity of the bacterium, but in a different way (Figure 2). Urea is split into ammonia and CO 2. If the gastric ph is suf ciently high (ph > 9.25), ammonia gas can be detected in the breath [33]. The theoretical advantages of this test are important. It does not require any type of urea, because the test uses the physiologic amount of the urea present in the stomach. Furthermore, to raise the ph in the stomach, all that is needed is a glass of sparkling water and a little magnesium. At present, we only have preliminary results from 10 patients, but more studies are required on a method that might dramatically change the cost of the diagnosis. Figure 2 Detection of ammonia vapor in the mouth. NON-INVASIVE DIRECT TECHNIQUES Stool antigen test Over the last few years, an enzyme immunoassay (EIA)that detects the presence of H. pylori antigen in stool specimens has become available, and has undergone testing for the initial diagnosis of H. pylori infection and for the con rmation of eradication after treatment (Figure 3). A polyclonal anti- H. pylori capture antibody absorbed to microwells is the most widely used [34] test, but a monoclonal antibody test has been recently described. The polyclonal antibody test has been extensively evaluated in the diagnosis of H. pylori infection before therapy. In studies performed during 1999 and the rst 6 months of 2001, 3419 patients were evaluated, and the weighted means for sensitivity and speci city were 93.2% and 93.2%, respectively [15]. These studies suggest that the test is comparable to the urea breath test for the initial detection of H. pylori infection. Consequently, the European Helicobacter Pylori study group has recommended the use of the urea breath test or stool testing in the initial diagnosis of H. pylori infection. There has been some variability in the results reported by different investigators in the post-therapy setting. Some of these differences may be due to the standard used for comparisons. In the studies performed during 1999 and the rst 6 months of 2001, which used standards as recommended by the working party of the European H. pylori study group, 332 patients were evaluated. The weighted mean sensitivity and speci city of the polyclonal test were 92.1% and 87.6%, respectively. However, in the studies performed during the same period, which used only the urea breath test as a comparator, 790 patients were evaluated,
5 Gatta et al Non-invasive techniques for diagnosis of H. pylori infection 493 Figure 3 Stool antigen test: how does it work? with weighted mean sensitivity and speci city of 88.8% and 87.3% being reported [15]. Although more studies in the post-therapy setting are necessary, the European Helicobacter study group (Maastricht 2000)has suggested that the polyclonal stool test may be an alternative to breath testing after treatment [5]. In addition, a recent study showed the reliability of the polyclonal test in identifying patients still infected as early as 14 days after the end of the treatment (Table 1) [35]. For monoclonal antibodies, only two full articles have been published. The rst, in a pretreatment setting, found a sensitivity and speci city of 96.3% and 88.5%, respectively, in 53 Table 1 Early follow-up with the polyclonal stool test Day a Probability that the patients still have H. pylori infection at day 35 Probability that the patients will be free of H. pylori infection at day 35 patients, while the second, in a post-treatment setting, assessed sensitivity and speci city as 88.6% and 94.7% in 178 patients [36,37]. However, despite the encouraging results, more studies need to be performed on the monoclonal test. CONCLUSIONS Non-invasive tests are becoming more and more important in the clinical management of the dyspeptic patient. Indeed, a number of clinical guidelines recommend non-invasive testing in dyspeptic patients, followed by treatment of H. pylori in primary care: the Maastricht II European Consensus, the European Society for Primary Care Gastroenterology, and the American Gastroenterological Association [5±7]. All recommend a `test and treat' strategy in patients under 45 or 55 years and with no alarm symptoms (i.e. anemia, weight loss, dysphagia, palpable mass, malabsorption, etc.)(figure 4). There are several sources of clinical evidence on which this strategy is founded. McColl et al. recently performed a randomized, controlled trial on 708 dyspeptic patients, and (95% CI: 0.19±0.99)(95% CI: 0.74±0.91) (95% CI: 0.69±1)(95% CI: 0.82±0.97) reported that, in patients less than 55 years of (95% CI: 0.68±1)(95% CI: 0.87±0.99) age with uncomplicated dyspepsia, `test and treat' (95% CI: 0.77±0.96)(95% CI: 0.89±1) was as effective and safe as endoscopy. Furthermore, they found that the strategy was as reassur- (95% CI: 0.71±1)(95% CI: 0.94±1) ing to the patients as endoscopy [38]. Similarly, Arents et al. showed that the `test and treat' strategy reduced the total number of endoscopies a Days after start of treatment. by 62% as compared with a prompt Positive or negative for H. pylori by biopsies 35 days after start of treatment. endoscopy
6 494 Clinical Microbiology and Infection, Volume 9 Number 6, June 2003 Figure 4 Management of uninvestigated dyspepsia. strategy, with similar outcomes in dyspeptic symptoms, quality of life, and patient satisfaction [39]. It has also been shown that the cost of management per patient per year for dyspeptic patients of the `test and treat' strategy was , while that of endoscopy was [40]. This strategy seems also to reduce the morbidity of dyspeptic patients. In a preliminary study performed on 267 dyspeptic patients recruited from a large car company, Madisch et al. found that the H. pylori `test and treat' strategy was effective in reducing both disease-related direct costs (i.e. antacid consumption, house doctor consultations)and indirect costs (i.e. absenteeism from work)[41]. The cost-effectiveness of diagnostic testing was evaluated recently in an economic model. The costs of different testing strategies were evaluated when the pretest probability was low, intermediate, or high. In general, the cost of serology is low in most countries, while the urea breath test and the stool antigen test are more expensive. Despite this, the improved accuracy of the stool and breath tests makes either of these tests cost-effective compared to serology or near patient tests. The lower the prevalence (and the pretest probability of infection), the less useful are serology and whole blood tests [42]. However, some other considerations need to be taken into account. If we consider what test patients would prefer, there is little doubt that this is urea breath test. However, if we consider the issue from the point of view of the healthcare providers, things are different. The cost of a mass spectrophotometer is high, its service requirements for one year are extremely expensive, not including spare parts, and, nally, dedicated personnel are needed. On the other hand, the stool test only requires an optical spectrophotometer, which is usually present in any laboratory, with negligible costs for maintenance or spare parts. Also, it does not require dedicated personnel. Hence, the breath test may be the best in private practice, but not in the national healthcare system, where, probably, the stool test is more cost-effective. REFERENCES 1. Warren JR, Marshall BJ. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet 1983; I: 1273±5. 2. Graham DY. Therapy of Helicobacter pylori: current status and issues. Gastroenterology 2000; 118: S2±8. 3. International Agency for Research on Cancer, World Health Organization. Infection with Helicobacter pylori. In: Schistosomes, liver flukes and Helicobacter pylori. Lyon: IARC, 1994: 177± complication_upergi.asp 5. Malfertheiner P, Megraud F, O'Morain C et al. Current concepts in the management of H. pylori infection- The Maastricht Consensus Report. Aliment Pharmacol Ther 2002; 16: 167± Rubin G, Meineche-Schmidt V, Roberts A, Childs S, de Wit N. The management of H. pylori infection in primary care. Guidelines from the ESPCG. Eur J Clin Pract 1999; 5: 98± American Gastroenterological Association. Medical position statement. Evaluation of dyspepsia. Gastroenterology 1998; 114: 579± Sackett DL, Haynes BR, Guyatt GH, Tugwell P. Clinical epidemiology: a basic science for clinical medicine. In: Sackett DL, Hayness RB, Guyatt GH, Tugwell P, eds. Interpretation of diagnostic data, 2nd edn. London: Little, Brown, 1985: 69± Loy CT, Irwig LM, Katelaris PH et al. Do commercial serological kits for Helicobacter pylori infection
7 Gatta et al Non-invasive techniques for diagnosis of H. pylori infection 495 differ in accuracy? A meta-analysis. Am J Gastroenterol 1996; 91: 1138± Stevens M, Livsey S, Swann R, Rathbone B. Evaluation of sixteen EIAs for the detection of antibodies to Helicobacter pylori. London: Department of Health, 1997: 1± Axon ATR. Are all helicobacters equal? Mechanisms of gastroduodenal pathology and their clinical implications. Gut 1999; 45(suppl I): I1±I Telford JL, Ghiara Dellorco M. Gene structure of the Helicobacter pylori cytotoxin and evidence of its key role in gastric disease. J Exp Med 1994; 179: 1653± Basso D, Stefani A, Brigato L et al. Serum antibodies anti H. pylori and anti CagA: a comparison between four different assays. J Clin Lab Anal 1999; 13: 194± Fusconi M, Vaira D, Menegatti M et al. Anti-cagA reactivity in Helicobacter pylori negative subjects: a comparison of three different methods. Dig DisSci 1999; 44: 1691± Vaira D, Vakil N. Blood, urine, stool, breath, money and Helicobacter pylori. Gut 2001; 48: 287± Luzza F, Study Group SIGE H. pylori Italy. Evaluation of a commercial serological kit for detection of salivary immunoglobulin G to Helicobacter pylori: a multicenter study. Eur J Gastroenterol 2000; 12: 1117± Miwa H, Hirose M, Kikuchi S et al. How useful is the detection kit for antibody to Helicobacter pylori in urine (URINELISA)in clinical practice? Am J Gastroenterol 1999; 94: 3460± Leodolter A, Vaira D, Bazzoli F, Hirschl A, Megraud F, Malfertheiner P. European Multicentre validation trial of two non-invasive tests for detection of antibody to H. pylori: urine based Elisa and rapid urine test. Gut 2001; 49: A Chey WD, Woods M, Scheiman JM et al. Lansoprazole and ranitidine affect the accuracy of the 14 C- urea breath test by a ph-dependent mechanism. Am J Gastroenterol 1997; 92: 446± Hegedus O, Ryden J, Rehnberg AS, Nilsson S, Hellstrom PM. Validated accuracy of a novel urea breath test for rapid Helicobacter pylori detection and in-office analysis. Eur J Gastroenterol Hepatol 2002; 14: 513± Munster DJ, Chapman BA, Burt MJ et al. The fate of ingested 14C-urea in the urea breath test for Helicobacter pylori infection. Scand J Gastroenterol 1993; 28: 661± Goddard AF, Logan RP. Urea breath tests for detecting Helicobacter pylori. Aliment Pharmacol Ther 1997; 11: 641± Raju GS, Smith MJ, Morton D et al. Mini dose (1mCi) 14 C urea breath test for the detection of Helicobacter pylori. Am J Gatroenterol 1994; 89: 1027± Peura DA, Pambianco DJ, Dye KR et al. Microdose of 14 C urea breath test offers diagnosis of Helicobacter pylori in 10 minutes. Am J Gastroenterol 1996; 91: 233± Graham DY, Klein PD, Evans DJ Jr et al. Campylobacter pylori detected non-invasively by the 13 C-urea breath test. Lancet 1987; 1: 1174± Parente F, Bianchi-Porro G. The 13 C-urea breath test for non-invasive diagnosis of Helicobacter pylori infection: which procedure and which measuring equipment? Eur J Gastroenterol Hepatol 2001; 13: 803± Hamlet A, Stage L, Lonroth H et al. A novel tablet based 13 C urea breath test for Helicobacter pylori with enhanced performance during acid suppression therapy. Scand J Gastroenterol 1999; 34: 367± Hamlet A, Stage L, LoÈnroth H et al. A novel tabletbased 13 C urea breath test for Helicobacter pylori with enhanced performance during acid suppression therapy. Scand J Gastroenterol 1999; 34: 367± Vaira D, Gatta L, Ricci C et al. A novel 50 mg urea tablet based 13 C-UBT for diagnosing and monitoring H. pylori infection. Gastroenterology 2002; 122: A Hine MK, O'Donnell JF. Incidence of urease producing bacteria in saliva. J Dent Res 1943; 22: 103± Klein TD, Graham DY. Minimum analysis requirements for the detection of Helicobacter pylori infection by the 13 C urea breath test. Am J Gastroenterol 1993; 88: 1865± Moulton-Barret RG, Triadafilopoulos G, Michener R, Gologorsky D. Serum 13 C bicarbonate in the assessment of gastric Helicobacter pylori urease activity. Am J Gastroenterol 1993; 88: 369± Dun CDR, Blac M, Cowell DC et al. Ammonia vapour in the mouth as a diagnostic marker for Helicobacter pylori infection: preliminary proof of principal pharmacological investigations. Br J Biomed Sci 2000; 58: 66± Vaira D, Malfertheiner P, Megraud F et al. Diagnosis of Helicobacter pylori infection with a new noninvasive antigen-based assay. Lancet 1999; 354: 30± Vaira D, Vakil N, Menegatti M et al. The stool antigen test for detection of Helicobacter pylori after eradication therapy. Ann Intern Med 2002; 136: 280± Agha-Amiri K, Peitz U, Mainz D, Kahl S, Leodolter A, Malfertheiner P. A novel immunoassay based on monoclonal antibodies for the detection of Helicobacter pylori antigens in human stool. Z Gastroenterol 2001; 39: 555± Leodolter A, Peitz U, Ebert MP, Agha-Amiri K, Malfertheiner P. Comparison of two enzyme immunoassays for the assessment of Helicobacter pylori status in stool specimens after eradication therapy. Am J Gastroenterol 2002; 97: 1682± McColl K, Murray LS, Grillen D et al. Randomised trial of endoscopy with testing for Helicobacter pylori
8 496 Clinical Microbiology and Infection, Volume 9 Number 6, June 2003 compared with non-invasive H. pylori testing alone in the management of dyspepsia. BMJ 2002; 324: 999± Arents NL, Thijs JC, Van Zwet AA, Kleibeur JH. Screening and treating for Helicobacter pylori (`test and treat strategy')in dyspepsia reduces number of endoscopies with similar clinical outcome as compared to prompt endoscopy. Gastroenterology 2001; 120: Jones R, Tait C, Sladen G, Weston-Baker J. A trial of a test and treat strategy for Helicobacter pylori positive dyspeptic patients in general practice. Int J Clin Pract 1999; 53: 413± Madisch A, Grabowski G, Guth A, von Guericke O. Hp test and treat strategy reduces both diseaserelated absenteeism from work, house doctor consultations and antacid consumption in uninvestigated dyspeptic staff members of a large factory. Gastroenterology 2001; 120: Vakil N, Rhew D, Soll A, Ofman J. The costeffectiveness of diagnostic testing strategies for H. pylori. Am J Gastroenterol 2000; 95: 1691±8.
Urea Breath Test for Diagnosis of Helicobactor pylori. Original Policy Date 12:2013
 MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
Helicobacter Pylori Testing HELICOBACTER PYLORI TESTING HS-131. Policy Number: HS-131. Original Effective Date: 9/17/2009
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
TRANSPARENCY COMMITTEE OPINION. 13 December 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans
 ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
MEDICAL POLICY EFFECTIVE DATE: 05/19/11 REVISED DATE: 05/24/12, 05/23/13 ARCHIVED DATE: 05/22/14 EDITED DATE: 05/28/15, 05/25/16, 05/18/17, 05/17/18
 MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
New immunoassay for the detection of Helicobacter pylori infection compared with urease test,
 Chapter V New immunoassay for the detection of Helicobacter pylori infection compared with urease test, 13C breath test and histology: validation in the primary care setting Catherine F Weijnen 1, Henriëtte
Chapter V New immunoassay for the detection of Helicobacter pylori infection compared with urease test, 13C breath test and histology: validation in the primary care setting Catherine F Weijnen 1, Henriëtte
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection
 Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study
 Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus
 University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2017 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2017 Next
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
An evaluation of whole blood testing for Helicobacter pylori in general practice
 Aliment Pharmacol Ther 1998; 12: 641±645. An evaluation of whole blood testing for Helicobacter pylori in general practice N. J. TALLEY, J. R. LAMBERT*, S. HOWELL, H. H.-X. XIA, S. K. LIN* & L. AGREUS
Aliment Pharmacol Ther 1998; 12: 641±645. An evaluation of whole blood testing for Helicobacter pylori in general practice N. J. TALLEY, J. R. LAMBERT*, S. HOWELL, H. H.-X. XIA, S. K. LIN* & L. AGREUS
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
ORIGINAL ARTICLE /j x
 ORIGINAL ARTICLE.1111/j.1469-691.6.1514.x Comparison of the performance of serological kits for Helicobacter pylori infection with European and Asian study populations T. T. H. Hoang 1,4, A.-S. Rehnberg
ORIGINAL ARTICLE.1111/j.1469-691.6.1514.x Comparison of the performance of serological kits for Helicobacter pylori infection with European and Asian study populations T. T. H. Hoang 1,4, A.-S. Rehnberg
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
H. pylori IgM CLIA kit
 H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
H. pylori Stool Rapid Test (Cassette)
 H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori?
 What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
Intragastric Acidification Reduces the Occurrence of False-Negative Urea Breath Test Results in Patients Taking a Proton Pump Inhibitor
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
Helicobacter pylori:an Emerging Pathogen
 Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in Detecting Helicobacter pylori Infection ABSTRACT
 Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Helicobacter pylori Infection in Children: A New Focus
 Helicobacter pylori Infection in Children: A New Focus John KC Yee Research Lab of Oral H. pylori USA Background Helicobacter pylori (H. pylori) infection places a heavy burden on medical and economic
Helicobacter pylori Infection in Children: A New Focus John KC Yee Research Lab of Oral H. pylori USA Background Helicobacter pylori (H. pylori) infection places a heavy burden on medical and economic
Clinical Policy Title: Breath Testing for H. Pylori
 Clinical Policy Title: Breath Testing for H. Pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 19, 2015 Next Review
Clinical Policy Title: Breath Testing for H. Pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 19, 2015 Next Review
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
Disclosures. Co-founder and Chief Science Officer, TechLab
 H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
KEYWORDS Dyspepsia, Acid Peptic Disease, Helicobacter Pylori, Urease, Giemsa, Peptic Ulcer, Non-Ulcer Dyspepsia.
 INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
Clinical Policy: Helicobacter Pylori Serology Testing Reference Number: CP.MP.153
 Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: July 3, 2018 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: July 3, 2018 Next
Framing Helicobacter pylori: The Etiology of Peptic Ulcers and Gastritis
 Framing Helicobacter pylori: The Etiology of Peptic Ulcers and Gastritis By Aja Dunn Gastritis (inflammation of the stomach); Etiologic agent - Helicobacter pylori (1). Transmission H. pylori infection
Framing Helicobacter pylori: The Etiology of Peptic Ulcers and Gastritis By Aja Dunn Gastritis (inflammation of the stomach); Etiologic agent - Helicobacter pylori (1). Transmission H. pylori infection
Clinical practice - breath tests
 Clinical practice - breath tests G D Bell Gastroenterology Department, Sunderland District General Hospital, Sunderland, UK The underlying principle of the two non-invasive radio-labelled urea breath tests
Clinical practice - breath tests G D Bell Gastroenterology Department, Sunderland District General Hospital, Sunderland, UK The underlying principle of the two non-invasive radio-labelled urea breath tests
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師
 Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Limited impact on endoscopy demand from a primary care based test and treat dyspepsia management strategy:
 Original Papers Limited impact on endoscopy demand from a primary care based test and treat dyspepsia management strategy: the results of a randomised controlled trial Ian S Shaw, Roland M Valori, André
Original Papers Limited impact on endoscopy demand from a primary care based test and treat dyspepsia management strategy: the results of a randomised controlled trial Ian S Shaw, Roland M Valori, André
Management of dyspepsia and of Helicobacter pylori infection
 Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Type of intervention Diagnosis. Economic study type Cost-effectiveness analysis.
 Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
H elicobacter pylori is a human pathogen that causes
 1543 STOMACH Effect of Helicobacter pylori eradication on development of dyspeptic and reflux disease in healthy asymptomatic subjects D Vaira, N Vakil, M Rugge, L Gatta, C Ricci, M Menegatti, G Leandro,
1543 STOMACH Effect of Helicobacter pylori eradication on development of dyspeptic and reflux disease in healthy asymptomatic subjects D Vaira, N Vakil, M Rugge, L Gatta, C Ricci, M Menegatti, G Leandro,
Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients with Dyspepsia
 Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar
 International Journal of Gastroenterology, Hepatology, Transplant & Nutrition Original Article Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar Hnin
International Journal of Gastroenterology, Hepatology, Transplant & Nutrition Original Article Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar Hnin
instrument. When 13C-UBT positive value is greater than or equal to / - 0.4, the the subject can be 1. Data and methods details are as follows:
 Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
EDUCATION PRACTICE. Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? Clinical Scenario.
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2008;6:1086 1090 EDUCATION PRACTICE Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? RICHARD J. SAAD* and WILLIAM D.
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2008;6:1086 1090 EDUCATION PRACTICE Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? RICHARD J. SAAD* and WILLIAM D.
Maastricht Ⅴ /Florence
 2016 21 10 577 Maastricht Ⅴ /Florence 200001 2015 10 8 9 Maastricht V 1 / 2 3 4 / 5 Maastricht Ⅴ Interpretation of Management of Helicobacter pylori Infection the Maastricht Ⅴ / Florence Consensus Report
2016 21 10 577 Maastricht Ⅴ /Florence 200001 2015 10 8 9 Maastricht V 1 / 2 3 4 / 5 Maastricht Ⅴ Interpretation of Management of Helicobacter pylori Infection the Maastricht Ⅴ / Florence Consensus Report
Accuracy of a Monoclonal Antibody-based Stool Antigen Test in the Diagnosis of Helicobacter pylori Infection
 ORIGINAL ARTICLE Accuracy of a Monoclonal Antibody-based Stool Antigen Test in the Diagnosis of Helicobacter pylori Infection A. M. Asfeldt, M.-L. Løchen, B. Straume, S. E. Steigen, J. Florholmen, R. Goll,
ORIGINAL ARTICLE Accuracy of a Monoclonal Antibody-based Stool Antigen Test in the Diagnosis of Helicobacter pylori Infection A. M. Asfeldt, M.-L. Løchen, B. Straume, S. E. Steigen, J. Florholmen, R. Goll,
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs Prof. Sheila Crowe
 Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Journal of Microbiological Methods
 Ž. Journal of Microbiological Methods 46 2001 235 240.elsevier.comrlocaterjmicmeth Ne immunoassay for the detection of Helicobacter pylori infection compared ith urease test, 13 C breath test and histology:
Ž. Journal of Microbiological Methods 46 2001 235 240.elsevier.comrlocaterjmicmeth Ne immunoassay for the detection of Helicobacter pylori infection compared ith urease test, 13 C breath test and histology:
Review article: 13 C-urea breath test in the diagnosis of Helicobacter pylori infection a critical review
 Aliment Pharmacol Ther 2004; 20: 1001 1017. doi: 10.1111/j.1365-2036.2004.02203.x Review article: 13 C-urea breath test in the diagnosis of Helicobacter pylori infection a critical review J. P. GISBERT
Aliment Pharmacol Ther 2004; 20: 1001 1017. doi: 10.1111/j.1365-2036.2004.02203.x Review article: 13 C-urea breath test in the diagnosis of Helicobacter pylori infection a critical review J. P. GISBERT
Rapid-VIDITEST. Helicobacter pylori. One step Helicobacter pylori Blister test. Instruction manual
 Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
American College of Gastroenterology Guideline on the Management of Helicobacter pylori Infection
 American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
Index. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Comparing furazolidone and tetracycline in quadruple therapy for eradication helicobacter pylori in dyspepsia patients
 Gastroenterology and Hepatology from bed to bench. 2008;1(1):39-43 2008 RCGLD, Research Center for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Comparing furazolidone and tetracycline in quadruple
Gastroenterology and Hepatology from bed to bench. 2008;1(1):39-43 2008 RCGLD, Research Center for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Comparing furazolidone and tetracycline in quadruple
Helicobacter Pylori Test-and-Treat Strategy for Management of Dyspepsia: A Comprehensive Review
 Citation: (2013) 4, e32; doi:10.1038/ctg.2013.3 & 2013 the American College of Gastroenterology All rights reserved 2155-384X/13 www.nature.com/ctg CLINICAL/NARRATIVE REVIEW Helicobacter Pylori Test-and-Treat
Citation: (2013) 4, e32; doi:10.1038/ctg.2013.3 & 2013 the American College of Gastroenterology All rights reserved 2155-384X/13 www.nature.com/ctg CLINICAL/NARRATIVE REVIEW Helicobacter Pylori Test-and-Treat
SCIENTIFIC DISCUSSION
 SCIENTIFIC DISCUSSION This module reflects the initial scientific discussion and scientific discussion on procedures, which have been finalised before 1 October 2001. For scientific information on procedures
SCIENTIFIC DISCUSSION This module reflects the initial scientific discussion and scientific discussion on procedures, which have been finalised before 1 October 2001. For scientific information on procedures
Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori
 ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
Assessment of symptomatic response as predictor of Helicobacter pylori status following eradication therapy in patients with ulcer
 618 University Department of Medicine and Therapeutics, Western Infirmary, Glasgow G11 6NT, UK K E L McColl A El-Nujumi L S Murray E M El-Omar A Dickson A W Kelman T E Hilditch Correspondence to: Professor
618 University Department of Medicine and Therapeutics, Western Infirmary, Glasgow G11 6NT, UK K E L McColl A El-Nujumi L S Murray E M El-Omar A Dickson A W Kelman T E Hilditch Correspondence to: Professor
6/25/ % 20% 50% 19% Functional Dyspepsia Peptic Ulcer GERD Cancer Other
 Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
Summary HTA. Efficacy and cost-effectiveness of the
 Summary HTA HTA-Report Summary Efficacy and cost-effectiveness of the 13 C-urea breath test as the primary diagnostic investigation for the detection of Helicobacter pylori infection compared to invasive
Summary HTA HTA-Report Summary Efficacy and cost-effectiveness of the 13 C-urea breath test as the primary diagnostic investigation for the detection of Helicobacter pylori infection compared to invasive
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY. Robin Warren
 - Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
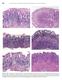 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
Helicobacter pylori Eradication Therapy Success Regarding Different Treatment Period Based on Clarithromycin or Metronidazole Triple-Therapy Regimens
 Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Gilles Jequier. Commercial Director Organobalance, a Novozymes Company
 "Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
"Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
Treating H. pylori in 2016
 Treating H. pylori in 2016 William D. Chey, MD, FACG Professor of Medicine University of Michigan The Case: A 38 yo Russian man presents with recurrent epigastric pain which occurs after meals and sometimes
Treating H. pylori in 2016 William D. Chey, MD, FACG Professor of Medicine University of Michigan The Case: A 38 yo Russian man presents with recurrent epigastric pain which occurs after meals and sometimes
NCCLS. Accutest H. pylori WB Test. (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only
 NCCLS Accutest H. pylori WB Test (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only A Rapid Visual Test for the Qualitative Detection of IgG Antibodies Specific to Helicobacter
NCCLS Accutest H. pylori WB Test (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only A Rapid Visual Test for the Qualitative Detection of IgG Antibodies Specific to Helicobacter
Investigating dyspepsia Rocco Maurizio Zagari, Lorenzo Fuccio, Franco Bazzoli
 DATE: 11/5/2008-10:27:27 ID:(BMJ)zagr584193 /(Jouve)bmj-001985 DOI: 10.1136/bmj.a1400 Topic(s): Type: InSection:review-article For the full versions of these articles see bmj.com CLINICAL REVIEW Investigating
DATE: 11/5/2008-10:27:27 ID:(BMJ)zagr584193 /(Jouve)bmj-001985 DOI: 10.1136/bmj.a1400 Topic(s): Type: InSection:review-article For the full versions of these articles see bmj.com CLINICAL REVIEW Investigating
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line)
 For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
Non-Invasive Assessment of Intestinal Function
 Overview Non-Invasive Assessment of Intestinal Function Introduction This paper will demonstrate that the 13 C-sucrose breath test ( 13 C-SBT) determines the health and function of the small intestine.
Overview Non-Invasive Assessment of Intestinal Function Introduction This paper will demonstrate that the 13 C-sucrose breath test ( 13 C-SBT) determines the health and function of the small intestine.
Development of a Lateral-flow Immunochromatographic Strip Using Gold Nanoparticles for Helicobacter pylori Detection
 Development of a Lateral-flow Immunochromatographic Strip Using Gold Nanoparticles for Helicobacter pylori Detection Duangporn Werawatganon MD*, Mongkol Pongsuchart MSc**, Amornpun Sereemaspun MD***, Orrawadee
Development of a Lateral-flow Immunochromatographic Strip Using Gold Nanoparticles for Helicobacter pylori Detection Duangporn Werawatganon MD*, Mongkol Pongsuchart MSc**, Amornpun Sereemaspun MD***, Orrawadee
Health-related anxiety and the effect of open-access endoscopy in US patients with dyspepsia
 Aliment Pharmacol Ther 23; 17: 835 84. doi: 1.146/j.269-2813.23.1497.x Health-related anxiety and the effect of open-access endoscopy in US patients with dyspepsia A. QUADRI & N. VAKIL University of Wisconsin
Aliment Pharmacol Ther 23; 17: 835 84. doi: 1.146/j.269-2813.23.1497.x Health-related anxiety and the effect of open-access endoscopy in US patients with dyspepsia A. QUADRI & N. VAKIL University of Wisconsin
Indigestion (dyspepsia)
 Commissioning pathways Indigestion (dyspepsia) Supplementary information to be read in conjunction with the pathway Reference Supplementary Information 1.1 Symptom Description Annual incidence 40%. Prevalence
Commissioning pathways Indigestion (dyspepsia) Supplementary information to be read in conjunction with the pathway Reference Supplementary Information 1.1 Symptom Description Annual incidence 40%. Prevalence
The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori
 The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori Barry Marshall The Gold-Standard Non-invasive Diagnostic Test for The Detection of H.pylori As a Healthcare Provider... Do you
The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori Barry Marshall The Gold-Standard Non-invasive Diagnostic Test for The Detection of H.pylori As a Healthcare Provider... Do you
Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings
 Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
imedpub Journals
 Research Article imedpub Journals www.imedpub.com Journal of Molecular Microbiology Comparative Evaluation of Stool Antigen Immunoassay and Blood Antibody Test Methods for the Screening of Helicobacter
Research Article imedpub Journals www.imedpub.com Journal of Molecular Microbiology Comparative Evaluation of Stool Antigen Immunoassay and Blood Antibody Test Methods for the Screening of Helicobacter
Failure of first-line eradication treatment significantly increases prevalence of antimicrobial-resistant Helicobacter pylori clinical isolates
 cp60392 Module 1 Journal of Clinical Pathology 28/8/08 12:38:20 Topics: ; Failure of first-line eradication treatment significantly increases prevalence of antimicrobial-resistant Helicobacter pylori clinical
cp60392 Module 1 Journal of Clinical Pathology 28/8/08 12:38:20 Topics: ; Failure of first-line eradication treatment significantly increases prevalence of antimicrobial-resistant Helicobacter pylori clinical
Comparison of stool antigen and urea breath test (UBT) methods for detection of Helicobacter pylori infection and the risk factors
 International Journal of Current Research in Medical Sciences ISSN: 2454-5716 P-ISJN: A4372-3064, E -ISJN: A4372-3061 www.ijcrims.com Original Research Article Volume 3, Issue 12-2017 Comparison of stool
International Journal of Current Research in Medical Sciences ISSN: 2454-5716 P-ISJN: A4372-3064, E -ISJN: A4372-3061 www.ijcrims.com Original Research Article Volume 3, Issue 12-2017 Comparison of stool
CLIA Complexity for Whole Blood: WAIVED. Intended Use. Summary and Explanation
 CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
The diagnosis and management of H. pylori infection in Singapore
 Singapore Med J 2017; 58(5): 234-240 doi: 10.11622/smedj.2017037 CMEArticle The diagnosis and management of H. pylori infection in Singapore Claire Alexandra Zhen Chew 1, MBChB, Tong Fong Lye 2, MBBS,
Singapore Med J 2017; 58(5): 234-240 doi: 10.11622/smedj.2017037 CMEArticle The diagnosis and management of H. pylori infection in Singapore Claire Alexandra Zhen Chew 1, MBChB, Tong Fong Lye 2, MBBS,
Evaluation of Western Blot CagA Seropositivity in Helicobacter pylori-seropositive and -Seronegative Subjects
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Feb. 2005, p. 304 309 Vol. 12, No. 2 1071-412X/05/$08.00 0 doi:10.1128/cdli.12.2.304 309.2005 Copyright 2005, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Feb. 2005, p. 304 309 Vol. 12, No. 2 1071-412X/05/$08.00 0 doi:10.1128/cdli.12.2.304 309.2005 Copyright 2005, American Society for Microbiology. All Rights
Management of Dyspepsia
 MPharm Programme Management of Dyspepsia Slide 1 of 28 Learning Objectives Understand the principles and wider implications underpinning evidence based therapeutics in the key clinical specialities Objectively
MPharm Programme Management of Dyspepsia Slide 1 of 28 Learning Objectives Understand the principles and wider implications underpinning evidence based therapeutics in the key clinical specialities Objectively
Treatment of H. pylori Infection: The Reality
 YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 119-124. Copyright 1999. All rights reserved. Treatment of H. pylori Infection: The Reality Nimish Vakil University of Wisconsin Medical School, Milwaukee
YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 119-124. Copyright 1999. All rights reserved. Treatment of H. pylori Infection: The Reality Nimish Vakil University of Wisconsin Medical School, Milwaukee
COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION
 Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
ReQuest H. Pylori IgG
 Intended use 01-550H 96 Test Set Enzyme Immunoassay for the Detection of Anti- Helicobacter pylori IgG Antibodies in Human Serum The SeraQuest H. Pylori IgG is a qualitative enzyme immunoassay (EIA) kit
Intended use 01-550H 96 Test Set Enzyme Immunoassay for the Detection of Anti- Helicobacter pylori IgG Antibodies in Human Serum The SeraQuest H. Pylori IgG is a qualitative enzyme immunoassay (EIA) kit
Survey on repeat prescribing for acid suppression drugs in primary care in Cornwall and the Isles of Scilly
 Aliment Pharmacol Ther 1999; 13: 813±817. Survey on repeat prescribing for acid suppression drugs in primary care in Cornwall and the Isles of Scilly R. BOUTET, M. WILCOCK & I. MACKENZIE 1 Department of
Aliment Pharmacol Ther 1999; 13: 813±817. Survey on repeat prescribing for acid suppression drugs in primary care in Cornwall and the Isles of Scilly R. BOUTET, M. WILCOCK & I. MACKENZIE 1 Department of
A Heaney, J S A Collins, RGPWatson, R J McFarland, K B Bamford, T C K Tham
 186 Royal Victoria Hospital, Belfast, A Heaney J S A Collins Department of Medicine, Queen s University, Belfast, RGPWatson Ulster Hospital, Dundonald, R J McFarland T C K Tham Department of Microbiology
186 Royal Victoria Hospital, Belfast, A Heaney J S A Collins Department of Medicine, Queen s University, Belfast, RGPWatson Ulster Hospital, Dundonald, R J McFarland T C K Tham Department of Microbiology
Validation of western Helicobacter pylori IgG antibody assays in Korean adults
 Journal of Medical Microbiology (2015), 64, 513 518 DOI 10.1099/jmm.0.000050 Validation of western Helicobacter pylori IgG antibody assays in Korean adults Sun-Young Lee, 1 Hee-Won Moon, 2 Mina Hur 2 and
Journal of Medical Microbiology (2015), 64, 513 518 DOI 10.1099/jmm.0.000050 Validation of western Helicobacter pylori IgG antibody assays in Korean adults Sun-Young Lee, 1 Hee-Won Moon, 2 Mina Hur 2 and
Validation of serological tests for Helicobacter pylori infection in an Irish population
 original paper Validation of serological tests for Helicobacter pylori infection in an Irish population NP Breslin, JM Lee, MJ Buckley, E Balbirnie 1, D Rice, 1 CA O'Morain Tallaght Regional Hospital,
original paper Validation of serological tests for Helicobacter pylori infection in an Irish population NP Breslin, JM Lee, MJ Buckley, E Balbirnie 1, D Rice, 1 CA O'Morain Tallaght Regional Hospital,
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line) Reagents
 For in vitro diagnostic use. CLIA Complexity: MODERATE INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to
For in vitro diagnostic use. CLIA Complexity: MODERATE INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to
The Nobel Prize in Physiology or Medicine for 2005
 The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
For the convenience of storage, Cassette and Sample Tubes can be stored separately. OneStep. H.Pylori Antigen. RapiCard InstaTest.
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
Peptic Ulcer Bleeding Risk. The Role of Helicobacter Pylori Infection in NSAID/Low-Dose Aspirin Users
 684 ORIGINAL CONTRIBUTIONS nature publishing group see related editorial on page x Peptic Ulcer Bleeding Risk. The Role of Helicobacter Pylori Infection in NSAID/Low-Dose Aspirin Users C. Sostres, MD 1,
684 ORIGINAL CONTRIBUTIONS nature publishing group see related editorial on page x Peptic Ulcer Bleeding Risk. The Role of Helicobacter Pylori Infection in NSAID/Low-Dose Aspirin Users C. Sostres, MD 1,
Magaji et al., Afr. J. Infect. Dis. 2(2): THE ROLE OF AN AGGRESSIVE FACTOR IN PEPTIC ULCER DISEASE (PUD)
 Magaji et al., Afr. J. Infect. Dis. 2(2): 80-84 80 Review Paper ISSN: 2006-0165 2008 THE ROLE OF AN AGGRESSIVE FACTOR IN PEPTIC ULCER DISEASE (PUD) Magaji, R.A. 1,b *, Tanko, Y. 2 and Magaji, G.M. 3 Afr.
Magaji et al., Afr. J. Infect. Dis. 2(2): 80-84 80 Review Paper ISSN: 2006-0165 2008 THE ROLE OF AN AGGRESSIVE FACTOR IN PEPTIC ULCER DISEASE (PUD) Magaji, R.A. 1,b *, Tanko, Y. 2 and Magaji, G.M. 3 Afr.
Helicobacter pylori IgA ELISA Kit
 Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD. Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School
 PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
