Clinical Policy Title: Breath Testing for H. Pylori
|
|
|
- Cameron Evans
- 5 years ago
- Views:
Transcription
1 Clinical Policy Title: Breath Testing for H. Pylori Clinical Policy Number: Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 19, 2015 Next Review Date: August, 2016 Related policies: Policy contains: H. pylori or Helicobacter pylori infection. Urea Breath Testing (UBT). Stool Antigen Testing (SAT) or Fecal Anitgen Testing (FAT). None. ABOUT THIS POLICY: AmeriHealth Caritas Pennsylvania has developed clinical policies to assist with making coverage determinations. AmeriHealth Caritas Pennsylvania s clinical policies are based on guidelines from established industry sources, such as the Centers for Medicare & Medicaid Services (CMS), state regulatory agencies, the American Medical Association (AMA), medical specialty professional societies, and peer-reviewed professional literature. These clinical policies along with other sources, such as plan benefits and state and federal laws and regulatory requirements, including any state- or plan-specific definition of medically necessary, and the specific facts of the particular situation are considered by AmeriHealth Caritas Pennsylvania when making coverage determinations. In the event of conflict between this clinical policy and plan benefits and/or state or federal laws and/or regulatory requirements, the plan benefits and/or state and federal laws and/or regulatory requirements shall control. AmeriHealth Caritas clinical policies are for informational purposes only and not intended as medical advice or to direct treatment. Physicians and other health care providers are solely responsible for the treatment decisions for their patients. AmeriHealth Caritas Pennsylvania s clinical policies are reflective of evidence- based medicine at the time of review. As medical science evolves, AmeriHealth Caritas Pennsylvania will update its clinical policies as necessary. AmeriHealth Caritas Pennsylvania s clinical policies are not guarantees of payment. Coverage policy AmeriHealth Caritas Pennsylvania considers the use of noninvasive diagnostic testing for Helicobacter pylori (H. pylori) including serology, urease breath testing (UBT) or stool antigene testing (SAT) to be clinically proven and, therefore, medically necessary when any of the following criteria are met: Criteria for medical necessity (at least one must be met) Evaluation of new onset dyspepsia in the following groups: o Those less than 55 years of age without alarm features, including bleeding, anemia, early satiety, unexplained weight loss, progressive dysphagia, odynophagia, persistent vomiting or family history of gastrointestinal cancers. o Active peptic ulcer disease (PUD). o Past history of peptic ulcers. o Gastric mucosa-associate tissue lymphoma (MALT). Recurrent dyspeptic symptoms despite two weeks of antibiotic treatment. Recurrent dyspeptic symptoms suggestive of reinfection with H. pylori. 1
2 Post-treatment eradication evaluation (no sooner than four weeks after completion of treatment and using either UBT or SAT) is only required in patients with: o Peptic ulcer disease (PUD). o Persistent dyspeptic symptoms. o H. pylori associated MALT lymphoma. o Previously resected early gastric cancers. Limitations: All other uses of non-invasive diagnostic testing for H. pylori are not medically necessary, including any of the following: Screening individuals without documented upper gastrointestinal symptoms. Screening individuals with non-invasive techniques who have documented alarm features or are greater than 55, with new onset dyspepsia (upper gastrointestinal endoscopy is indicated). Screening individuals with non-invasive techniques who have had upper gastrointestinal endoscopy, with testing for H. pylori within preceding six weeks. Screening individuals without dyspeptic symptoms post-treatment (other than groups listed above). Screening using UBT or SAT for those without a two week washout period of proton pump inhibitors (PPIs). Using UBT with carbon-14 in pregnant women or children. Using serologic testing as an eradication evaluation measure post-antibiotic treatment. Using both UBT and SAT concurrently, as simultaneous testing is not medically necessary. NOTE: The following CPT codes are not included in the Pennsylvania Medicaid fee schedule: Helicobacter pylori; breath test analysis for urease activity, non-radioactive isotope Helicobacter pylori, stool Alternative covered services: Clinical evaluation by physicians and appropriate standard diagnostic procedures. Background H. pylori is a gram-negative bacteria that is found in the luminal surface of the gastric epithelium. H. pylori is responsible for the inflammation of the underlying mucosa of the gastric epithelium. This bacteria is unique in its ability to survive in the acidic environment of the stomach because of its high urease, which is an enzyme that converts the urea of gastric juice to basic ammonia and carbon dioxide activity. H. pylori infection can be a cause of chronic gastritis, peptic ulcer disease and gastric malignancy. It is considered a class I carcinogen by the World Health Organization (WHO). 2
3 Estimates of the prevalence of H. pylori infection indicate that at least 50 percent of the world s population is affected. The likelihood of infection increases with age and lower socioeconomic status, meaning that prevalence varies across groups. In addition, men are more likely than women to be infected. Within the U.S., estimates of prevalence range from percent of the general public. Acquisition of the infection occurs mostly during childhood, due to poor living conditions. The improved living conditions of children within the U.S. indicate continued falling prevalence rates. The majority of individuals infected with H. pylori do not display clinically significant complications and will not require treatment. Signs, symptoms and conditions associated with H. pylori: H. pylori presents in both adults and children and can be diagnosed at any age, although it is more commonly found in older adults. Recognition, testing, and treatment for H. pylori can be difficult because of the variety of non-specific gastrointestinal signs and symptoms at presentation. H. pylori causes many symptoms of indigestion or dyspepsia defined as long-term pain in the upper abdomen. However, H. pylori is not the sole cause of these symptoms, which can also be linked to non-ulcer dyspepsia or gastroesophageal reflux disease (GERD). The most frequent features reported in patients with H. pylori are heartburn or epigastric pain; burping; bloating or post-prandial fullness; nausea; vomiting; slow digestion or delayed gastric emptying and acid hyper secretion. H. pylori has been linked to multiple upper gastrointestinal diseases. The bacteria inflame the mucosa lining stimulating increased gastrin, a hormone that stimulates stomach acid release. Elevated levels of gastrin cause excess acid secretion from all areas of the stomach, (even those areas not infected). In turn, the high acidity that further damages the mucosa lining of the stomach, causes ulceration and metaplasia. These areas are then at increased risk for additional infection by the H. pylori bacteria. There is a strong association between the H. pylori infection, gastric cancer and gastric MALT lymphomas. Eradication of the infection provides a cure to 80 percent of duodenal ulcers unrelated to non-steroidal anti-inflammatory drugs (NSAIDs), reduces progression of atrophic gastritis (inflammation of the mucosa lining) and reduces localized gastric MALT lymphoma. Diagnosis of H. pylori: Diagnosis of the infection is important to its management. Proper eradication treatment can resolve symptoms of dyspepsia, if it is related to an underlying ulcer disease caused by H. pylori. However, screening is not recommended in all patients with symptoms of dyspepsia, as empiric treatment with acid suppression is effective among some populations. Further, many conditions including GERD and non-ulcer dyspepsia are not associated with H. pylori. There are numerous different diagnosis tests for H. pylori including endoscopy, serology, UBT and SAT. These tests are used for diagnosis and evaluation of eradication after treatment. The most invasive diagnostic tool is endoscopy, which entails a biopsy of cells to be taken from the prepyloric region or fundic region. Endoscopy is not required for diagnosis and is not often the test of choice to confirm diagnosis. 3
4 Serologic tests identify the IgG (immunoglobulin) antibodies response to H. pylori bacteria within an individual s blood. This type of testing has lower sensitivity at 85 percent and specificity at 79 percent. The results can be reported as equivocal. It is not useful in testing for eradication of the bacteria, as antibodies may persist for months beyond treatment despite eradication of the bacteria. A common noninvasive test is urea breath testing or UBT; this test relies on the bacteria s ability to convert carbon dioxide urea. UBT centers on labeling isotopes of carbon dioxide urea that are ingested by the patient. The test compares a baseline sample with the post administration level of carbon dioxide using a mass spectrometer or infrared spectrophotometer. This test is commonly used to measure the success of treatment and is widely used, as the samples are easy to collect and can be sent to central laboratories. This reduces the need for expensive equipment at individual providers. Confirmation of diagnosis and eradication is also possible with stool antigen testing, also referred to as fecal antigen testing. H. pylori presence is detected by identifying H. pylori specific antigens, using either monoclonal or polyclonal antibodies. Monoclonal testing has been shown to be more sensitive and specific. This type of testing is both less expensive than UBT, requires less equipment and is just as effective. Additionally, this test s accuracy is less affected by PPI use than UBT. Searches AmeriHealth Caritas Pennsylvania searched PubMed and the databases of: UK National Health Services Centre for Reviews and Dissemination. Agency for Healthcare Research and Quality s National Guideline Clearinghouse and other evidencebased practice centers. The Centers for Medicare & Medicaid Services (CMS). We conducted searches on June 20, Search terms were Helicobacter pylori, [MeSH] or "diagnostic test," or stool antigen or urea breath test. We included: Systematic reviews, which pool results from multiple studies to achieve larger sample sizes and greater precision of effect estimation than in smaller primary studies. Systematic reviews use predetermined transparent methods to minimize bias, effectively treating the review as a scientific endeavor, and are thus rated highest in evidence-grading hierarchies. Guidelines based on systematic reviews. Economic analyses, such as cost-effectiveness, and benefit or utility studies (but not simple cost studies), reporting both costs and outcomes sometimes referred to as efficiency studies which also rank near the top of evidence hierarchies. 4
5 Findings Table 1 summarizes the findings from four systematic reviews, of the clinical validity of multiple types of non-invasive H. pylori diagnostic tests. Table 2 lists four systematic reviews of non-invasive testing, one randomized control trial comparing diagnostic tests and four professional guidelines related to diagnosis and treatment of H. pylori. Serologic testing for H. pylori: The results from available systematic reviews listed in Tables 1 and 2 indicate that serologic testing for antibodies linked to H. pylori infections demonstrate high sensitivity and specificity for initial diagnosis. These tests are especially useful in areas with high prevalence of the infection, but antibodies specifying this infection can differ locally, and therefore the accuracy of this test can range (Childs 2000). Because of the cost effectiveness and ease of blood tests, this is a first line diagnostic measure in some places, but confirmation requires additional testing if the area has a low prevalence (Ling 2013, Chey 2007). Further serologic testing is unreliable for post-treatment eradication verification, as antibodies for H. pylori may remain in the blood for six to 12 months after eradication (Childs 2000). UBT testing: The evidence for UBT testing for screening for H. Pylori in a test and treat strategy is based on three systematic reviews (Ling 2013, Ferwana 2015, Childs 2006) and professional guidelines. As shown in Table 1, UBT has high sensitivity and specificity, both >90 percent and can also be used to evaluate if eradication treatments were successful. UBT is considered the gold standard for post-treatment testing (Childs 2000). This test can be prohibitively expensive and some facilities may not have the materials on- site to perform. UBT testing in women and children should only use carbon-13 isotopes, as they provide lower doses of radiation. Table 1. Pooled estimates of sensitivity and specificity of non-invasive tests for H. Pylori diagnosis. Type of Test Systematic Se (%) Sp (%) review Serological Childs OHTAS (95% CI, ) 71.7 (95% CI, ) ICSI UBT Childs Monoclonal Stool Calvet (95% CI, 83 95) 89.5 (95% CI, 81 95) OHTAS (95% CI, ) 91.6 (95% CI, ) Ferwana (95% CI, 95 97) 93 (95% CI, 91 94) ICSI NICE ACG
6 Gisbert (95% CI, 93 96) 97 (95% CI, 94 98) MAS (95% CI, ) depending on test Polyclonal stool Calvet (95% CI, 83 95) 93 (95% CI, 84 97) Stool testing (with no differentiation to type) Se = sensitivity; Sp = specificity Summary of clinical evidence: ACG Gisbert (95% CI, 80 85) 96 (95% CI, 94 97) ICSI Citation Ferwana (2015) Content, methods, recommendations Key points: Systematic Review of 3999 patients for the accuracy of UBT (Level B) Twenty-three studies pooled found UBT testing of Se=96% and Sp=93%. UBT showed significant discrimination ability between infected and non-infected states. May provide better comprehensive results than endoscopy because eliminate error with endoscopic biopsy when H. pylori has inconsistent distribution and does not rely on pathologist. Oral flora s urease activity may have some impact on accuracy of results. NICE (2014) National Institute for Health and Care Excellence Dyspepsia and GERD Recommendations (Level C) OHTAS (2013) Serological testing is not a viable option for post-treatment/eradication evaluation. Dyspepsia and acute gastrointestinal bleeding require same day referral to endoscopic procedure. Prior to UBT or SAT testing, patient must undergo two week washout out of PPIs, as they can affect results. Systematic Review of 21 studies with 4536 patient testing accuracy of carbon-13 UBT in uninvestigated Dyspepsia (Level A) Calvet (2009) Serology and UBT are comparably sensitive, but UBT is more sensitive. Clinically, serological is a first-line diagnostic test in the United States; UBT or SAT are also considered first-line diagnostic methods. Lack of standards in process of UBT creates variability in results Stool Testing is most accurate non-invasive test (Level B) Randomized controlled trial (RCT) (n=199) found among patients with dyspepsia, demonstrated the most accurate alternative to endoscopy is Stool Antigen testing, as UBT 6
7 ACG (updated 2007) had lower accuracy. American College of Gastroenterology Guidelines for Management of Helicobacter Infection (Level C) Gisbert (2006) ACG (2005) Testing for proof of eradication post-treatment is required for those with Peptic Ulcer Disease with persistent dyspeptic symptoms, H. pylori MALT lymphoma and resected early gastric cancers. Test and treat in those with active PUD, past history of peptic ulcers, and gastric MALT or those greater than 55 years of age without alarm symptoms. UBT is the most reliable method to test for post-treatment eradication. Post-treatment eradication testing should be at least four weeks after antibiotic regimen completion. 13c is preferred method of UBT in children and pregnant women, as lower doses of radiation. Cost of UBT testing is driven by expensive and rare equipment and costly labeled isotopes. SAT and UBT are interchangeable diagnostic methods. Use of serological testing for specific antibodies is acceptable in areas of high prevalence (including urban or immigrant areas) as the positive predictive value is good. In low prevalence areas (~20%), avoid relying on antibody or confirm with SAT or UBT before treatment. Systematic review of 2,499 patients evaluated with fecal antigen testing (Level A) Pre-treatment pooled Se=94%, Sp=97%, monoclonal fecal antigen testing (FAT) had higher sensitivity (Se=95%, Sp=96%) compared to the polyclonal test (Se=83%, Sp=96%). Post-treatment evaluation shows distinct difference between monoclonal (Se=91%, Sp=95%) and polyclonal (Se=76%, SP=95%), as monoclonal is more sensitive. Monoclonal testing shows better distinction between infection and non-infection, with no equivocal response. American College of Gastroenterology Guidelines on Management of Dyspepsia (Level C) ICSI (2004) Those greater than 55 years of age, with alarm signs (bleeding, anemia, early satiety, unexplained weight loss, progressive dysphagia, odynophagia, persistent vomiting and family history of gastrointestinal cancers) should be examined with endoscopy. When prevalence is greater than 10%, use test and treat strategy using non-invasive diagnostic methods; when prevalence is less than 10%, initiate empiric PPI treatment and revert to test and treat for H. pylori, if PPI therapy is unsuccessful. Serological tests require local validation; sensitivity and specificity can range depending on geography. Institute for Clinical Systems Improvement Algorithm Annotations for Dyspepsia and GERD (Level C) Symptoms persisting after four weeks of treatment require additional screening with endoscopy. Prior PUD is a predictor of recurrent ulcers, estimates range from 30-95% of PUD are due to H. pylori, so testing is required for those with past medical history. Most cost effective first line test is serological. 7
8 Childs (2000) Systematic Review of noninvasive and post treatment testing Serology is not accurate for all patient populations and requires local validation; can have six to 12 month lag time in detecting changes in infection status, although may be the cheapest option. UBT is non-invasive test of choice. Post-treatment testing is required in those with severe or complicated DU or GU, MALT lymphoma and early gastric cancer. H. pylori treatment guided by testing is preferred choice, although cost savings are slow to see. Can lower cost of treatment further by using empiric treatment where there is high prevalence of infection and DU. Se, sensitivity; Sp, specificity Glossary Dyspepsia General discomfort of the upper gastrointestinal tract, including upper abdominal pain, heartburn, acid reflux, nausea or vomiting. Endoscopy A non-surgical procedure to enable providers to examine interior structures, such as organs or joints using an endoscope, a flexible tube with a light and camera. Samples can be taken during this procedure. Gastric MALT lymphoma A cancer involving the mucosa-associated lymphoid tissue of the stomach that originates in B cells and is related to inflammation. Gastritis Inflammation of the lining of the stomach. May cause symptoms including upper abdominal pain, vomiting, nausea, bloating, loss of appetite or heart burn. Gastroesophageal reflux disease (GERD) A disease caused by chronic back-flow of acid from the stomach into the esophagus, causing heartburn and leading to irritation and possible damage to the lining of the esophagus. Histology The study of the anatomy of cells and tissues. In medicine, this microscopic study is performed by pathologists to correctly diagnose certain cancers and other diseases. Non-steroidal anti-inflammatory drugs (NSAIDs) A class of drugs with both analgesic and antiinflammatory effects. Serology Testing of blood serum for the presence of specific products that can serve as indicators of disease. Related policies 8
9 AmeriHealth Caritas Pennsylvania Utilization Management program description. References Professional society guidelines/other: Chey WD, Wong BC. American College of Gastroenterology guideline on the management of Helicobacter pylori infection. Am J Gastroenterol. 2007; 102(8): Dyspepsia and gastro-oesophageal reflux disease: investigation and management of dyspepsia, symptoms suggestive of gasto-oesophageal reflux disease, or both. Clinical guideline (update). National Institute of Clinical Excellence (NICE). Published Sep Updated Nov Accessed Jul. 7, Ling D. Carbon-13 urea breath test for Helicobacter pylori infection in patients with uninvestigated ulcerlike dyspepsia: an evidence-based analysis. Ont Health Technol Assess Ser. 2013; 13(19): Healthcare Guideline: Dyspepsia and GERD. Institute for Clinical Systems Improvement Elective/Literature/Dyspepsia guidelines ICSI 2004 Accessed Jul. 6 th, Talley NJ, Vakil N. Guidelines for the management of dyspepsia. Am J Gastroenterol. 2005; 100(10): Peer-reviewed references: Calvet X, Sánchez-delgado J, Montserrat A, et al. Accuracy of diagnostic tests for Helicobacter pylori: a reappraisal. Clin Infect Dis. 2009; 48(10): Fashner J, Gitu AC. Diagnosis and Treatment of Peptic Ulcer Disease and H. pylori Infection. Am Fam Physician. 2015; 91(4): Ferwana M, Abdulmajeed I, Alhajiahmed A, et al. Accuracy of urea breath test in Helicobacter pylori infection: meta-analysis. World J Gastroenterol. 2015; 21(4): Gisbert JP, De la morena F, Abraira V. Accuracy of monoclonal stool antigen test for the diagnosis of H. pylori infection: a systematic review and meta-analysis. Am J Gastroenterol. 2006; 101(8): Mccoll KE. Clinical practice. Helicobacter pylori infection. N Engl J Med. 2010; 362(17): Childs S, Roberts A, Meinecke-Schmidt V, dewit N, Rubin G. The management of Helicobacter pylori infection in primary care: a systematic review of the literature. Family Practice. 2000; 17(suppl 2): S6. Tonkic A, Tonkic M, Lehours P, Mégraud F. Epidemiology and diagnosis of Helicobacter pylori infection. Helicobacter. 2012; 17 Suppl 1:
10 Clinical trials: Searched clinicaltrials.gov on July 1, 2015 using term Helicobacter pylori diagnosis. 56 Open Studies. Seven studies found, one relevant. National University Hospital, Singapore. Gamma-Glutamyl Transpeptidase (GGT): A Potential Diagnostic Marker for Hellicobacter Pylori Infections. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US) [cited 2015 July 6. ]. Available from: NLM Identifier: NCT There are many ongoing open enrollment clinical trials for diagnosis of H. pylori but the majority are related to endoscopic procedures not outlined in this policy. CMS National Coverage Determinations (NCDs): No NCDs identified as of the writing of this policy. Local Coverage Determinations (LCDs): No LCDs identified as of the writing of this policy. Commonly submitted codes Below are the most commonly submitted codes for the service(s)/item(s) subject to this policy. This is not an exhaustive list of codes. Providers are expected to consult the appropriate coding manuals and bill accordingly. CPT codes Description Helicobacter pylori; breath test analysis for urease activity, non-radioactive isotope Helicobacter pylori, stool ICD-9 Code Description Comment Helicobacter pylori (H. pylori) Marginal zone lymphoma involving intra-abdominal lymph nodes Peptic ulcer, site unspecified, without mention of obstruction Peptic ulcer, site unspecified, with obstruction Dyspepsia ICD-10 Code Description Comment B96.81 H. pylori as the cause of diseases classified elsewhere 10
11 C88.4 MALT lymphoma K27.9 Peptic ulcer, site unspecified R10.13 Dyspepsia HCPCS Level II NA Description Comment 11
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2017 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2017 Next
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: July 3, 2018 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: July 3, 2018 Next
Helicobacter Pylori Testing HELICOBACTER PYLORI TESTING HS-131. Policy Number: HS-131. Original Effective Date: 9/17/2009
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Clinical Policy: Helicobacter Pylori Serology Testing Reference Number: CP.MP.153
 Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Urea Breath Test for Diagnosis of Helicobactor pylori. Original Policy Date 12:2013
 MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MEDICAL POLICY EFFECTIVE DATE: 05/19/11 REVISED DATE: 05/24/12, 05/23/13 ARCHIVED DATE: 05/22/14 EDITED DATE: 05/28/15, 05/25/16, 05/18/17, 05/17/18
 MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Clinical Policy Title: Strep testing
 Clinical Policy Title: Strep testing Clinical Policy Number: 07.01.09 Effective Date: December 1, 2017 Initial Review Date: October 19, 2017 Most Recent Review Date: November 16, 2017 Next Review Date:
Clinical Policy Title: Strep testing Clinical Policy Number: 07.01.09 Effective Date: December 1, 2017 Initial Review Date: October 19, 2017 Most Recent Review Date: November 16, 2017 Next Review Date:
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD)
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD) Routine endoscopic investigation of patients of any age, presenting with dyspepsia
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD) Routine endoscopic investigation of patients of any age, presenting with dyspepsia
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA o Patients of any age with ALARM signs should be referred through the 2-week referral system o Routine endoscopic investigation
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA o Patients of any age with ALARM signs should be referred through the 2-week referral system o Routine endoscopic investigation
Treatment of Helicobacter pylori Infection
 Treatment of Helicobacter pylori Infection Epidemiology of H. pylori infection (North America) Which are the high risk groups? Epidemiology of H. pylori infection (North America) Which are the high risk
Treatment of Helicobacter pylori Infection Epidemiology of H. pylori infection (North America) Which are the high risk groups? Epidemiology of H. pylori infection (North America) Which are the high risk
June By: Reza Gholami
 ACG/CAG guideline on Management of Dyspepsia June 2017 By: Reza Gholami DEFINITION OF DYSPEPSIA AND SCOPE OF THE GUIDELINE Dyspepsia was originally defined as any symptoms referable to the upper gastrointestinal
ACG/CAG guideline on Management of Dyspepsia June 2017 By: Reza Gholami DEFINITION OF DYSPEPSIA AND SCOPE OF THE GUIDELINE Dyspepsia was originally defined as any symptoms referable to the upper gastrointestinal
Clinical Policy Title: Abdominal aortic aneurysm screening
 Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: July 20, 2017 Next
Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: July 20, 2017 Next
Management of dyspepsia and of Helicobacter pylori infection
 Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Non-Ulcer Dyspepsia: what is it? What can we do with these patients? Overview. Dyspepsia Definition. Functional Dyspepsia. Dyspepsia the Basics
 Non-Ulcer : what is it? What can we do with these patients? Temporal Changes and Geographic Variations in Developing Peptic Ulcer Disease Gastric Cancer 1900 Eamonn M M Quigley MD FACG Alimentary Pharmabiotic
Non-Ulcer : what is it? What can we do with these patients? Temporal Changes and Geographic Variations in Developing Peptic Ulcer Disease Gastric Cancer 1900 Eamonn M M Quigley MD FACG Alimentary Pharmabiotic
Management of Dyspepsia
 MPharm Programme Management of Dyspepsia Slide 1 of 28 Learning Objectives Understand the principles and wider implications underpinning evidence based therapeutics in the key clinical specialities Objectively
MPharm Programme Management of Dyspepsia Slide 1 of 28 Learning Objectives Understand the principles and wider implications underpinning evidence based therapeutics in the key clinical specialities Objectively
Clinical Policy Title: Cardiac rehabilitation
 Clinical Policy Title: Cardiac rehabilitation Clinical Policy Number: 04.02.02 Effective Date: September 1, 2013 Initial Review Date: February 19, 2013 Most Recent Review Date: February 6, 2018 Next Review
Clinical Policy Title: Cardiac rehabilitation Clinical Policy Number: 04.02.02 Effective Date: September 1, 2013 Initial Review Date: February 19, 2013 Most Recent Review Date: February 6, 2018 Next Review
ACG Clinical Guideline: Treatment of Helicobacter pylori Infection
 ACG Clinical Guideline: Treatment of Helicobacter pylori Infection William D. Chey, MD, FACG 1, Grigorios I. Leontiadis, MD, PhD 2, Colin W. Howden, MD, FACG 3 and Steven F. Moss, MD, FACG 4 1 Division
ACG Clinical Guideline: Treatment of Helicobacter pylori Infection William D. Chey, MD, FACG 1, Grigorios I. Leontiadis, MD, PhD 2, Colin W. Howden, MD, FACG 3 and Steven F. Moss, MD, FACG 4 1 Division
Clinical Policy Title: Measurement of serum antibodies to infliximab and adalimumab
 Clinical Policy Title: Measurement of serum antibodies to infliximab and adalimumab Clinical Policy Number: 01.01.03 Effective Date: January 1, 2016 Initial Review Date: September 16, 2015 Most Recent
Clinical Policy Title: Measurement of serum antibodies to infliximab and adalimumab Clinical Policy Number: 01.01.03 Effective Date: January 1, 2016 Initial Review Date: September 16, 2015 Most Recent
American College of Gastroenterology Guideline on the Management of Helicobacter pylori Infection
 American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Clinical Policy Title: Breast cancer index genetic testing
 Clinical Policy Title: Breast cancer index genetic testing Clinical Policy Number: 02.01.22 Effective Date: January 1, 2017 Initial Review Date: October 19, 2016 Most Recent Review Date: October 19, 2016
Clinical Policy Title: Breast cancer index genetic testing Clinical Policy Number: 02.01.22 Effective Date: January 1, 2017 Initial Review Date: October 19, 2016 Most Recent Review Date: October 19, 2016
Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden
 Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden Clinical Policy Number: 05.01.03 Effective Date: January 1, 2016 Initial Review Date: July 15, 2015 Most Recent Review Date:
Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden Clinical Policy Number: 05.01.03 Effective Date: January 1, 2016 Initial Review Date: July 15, 2015 Most Recent Review Date:
Peptic Ulcer Disease Update
 Peptic Ulcer Disease Update Col Pat Storms RAM 2005 Disclosure Information 84th Annual AsMA Scientific Meeting Col Patrick Storms I have no financial relationships to disclose. I will discuss the following
Peptic Ulcer Disease Update Col Pat Storms RAM 2005 Disclosure Information 84th Annual AsMA Scientific Meeting Col Patrick Storms I have no financial relationships to disclose. I will discuss the following
6/25/ % 20% 50% 19% Functional Dyspepsia Peptic Ulcer GERD Cancer Other
 Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
Clinical Policy Title: Genicular nerve block
 Clinical Policy Title: Genicular nerve block Clinical Policy Number: 14.01.10 Effective Date: October 1, 2017 Initial Review Date: September 21, 2017 Most Recent Review Date: October 19, 2017 Next Review
Clinical Policy Title: Genicular nerve block Clinical Policy Number: 14.01.10 Effective Date: October 1, 2017 Initial Review Date: September 21, 2017 Most Recent Review Date: October 19, 2017 Next Review
The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus
 University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden
 Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden Clinical Policy Number: 05.01.03 Effective Date: January 1, 2016 Initial Review Date: July 15, 2015 Most Recent Review Date:
Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden Clinical Policy Number: 05.01.03 Effective Date: January 1, 2016 Initial Review Date: July 15, 2015 Most Recent Review Date:
Proton Pump Inhibitors Drug Class Prior Authorization Protocol
 Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Disclosures. Co-founder and Chief Science Officer, TechLab
 H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
Clinical Policy Title: Abdominal aortic aneurysm screening
 Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: June 5, 2018 Next
Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: June 5, 2018 Next
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD. Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School
 PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening
 Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening Clinical Policy Number: 01.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21, 2015 Most Recent
Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening Clinical Policy Number: 01.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21, 2015 Most Recent
Clinical Policy Title: Abdominal aortic aneurysm screening
 Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: June 5, 2018 Next
Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: June 5, 2018 Next
Clinical Policy Title: Zoster (shingles) vaccine
 Clinical Policy Title: Zoster (shingles) vaccine Clinical Policy Number: 18.02.10 Effective Date: June 1, 2018 Initial Review Date: April 10, 2018 Most Recent Review Date: May 1, 2018 Next Review Date:
Clinical Policy Title: Zoster (shingles) vaccine Clinical Policy Number: 18.02.10 Effective Date: June 1, 2018 Initial Review Date: April 10, 2018 Most Recent Review Date: May 1, 2018 Next Review Date:
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
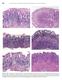 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs Prof. Sheila Crowe
 Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori?
 What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
Outline. Definition (s) Epidemiology Pathophysiology Management With an emphasis on recent developments
 Chronic Dyspepsia Eamonn M M Quigley MD FRCP FACP MACG FRCPI Lynda K and David M Underwood Center for Digestive Disorders Houston Methodist Hospital Houston, Texas Outline Definition (s) Epidemiology Pathophysiology
Chronic Dyspepsia Eamonn M M Quigley MD FRCP FACP MACG FRCPI Lynda K and David M Underwood Center for Digestive Disorders Houston Methodist Hospital Houston, Texas Outline Definition (s) Epidemiology Pathophysiology
Clinical Policy Title: Ketamine for treatment-resistant depression
 Clinical Policy Title: Ketamine for treatment-resistant depression Clinical Policy Number: 00.02.13 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: January
Clinical Policy Title: Ketamine for treatment-resistant depression Clinical Policy Number: 00.02.13 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: January
Module 2 Heartburn Glossary
 Absorption Antacids Antibiotic Module 2 Heartburn Glossary Barrett s oesophagus Bloating Body mass index Burping Chief cells Colon Digestion Endoscopy Enteroendocrine cells Epiglottis Epithelium Absorption
Absorption Antacids Antibiotic Module 2 Heartburn Glossary Barrett s oesophagus Bloating Body mass index Burping Chief cells Colon Digestion Endoscopy Enteroendocrine cells Epiglottis Epithelium Absorption
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection
 Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Guidelines for the Management of Dyspepsia and GORD. Gastroenterology/ Acute Adult Governance. Drugs and Therapeutics Committee
 Guidelines for the Management of Dyspepsia and GORD Document type: Version: 3.0 Author (name): Author (designation): Validated by Prescribing Dr. G. Lipscomb Date validated October 2015 Ratified by: Date
Guidelines for the Management of Dyspepsia and GORD Document type: Version: 3.0 Author (name): Author (designation): Validated by Prescribing Dr. G. Lipscomb Date validated October 2015 Ratified by: Date
Peptic ulcer disease. Nomin-Erdene. D SOM-531
 Peptic ulcer disease Nomin-Erdene. D SOM-531 Learning objectives Stomach gross anatomy PUD Epidemiology Pathogenesis Clinical manifestation Diagnosing Treatment Complicated ulcer disease Surgical procedures
Peptic ulcer disease Nomin-Erdene. D SOM-531 Learning objectives Stomach gross anatomy PUD Epidemiology Pathogenesis Clinical manifestation Diagnosing Treatment Complicated ulcer disease Surgical procedures
Peptic ulcer disease Disorders of the esophagus
 Peptic ulcer disease Disorders of the esophagus Peptic ulcer disease Burning epigastric pain Exacerbated by fasting Improved with meals Ulcer: disruption of mucosal integrity >5 mm in size, with depth
Peptic ulcer disease Disorders of the esophagus Peptic ulcer disease Burning epigastric pain Exacerbated by fasting Improved with meals Ulcer: disruption of mucosal integrity >5 mm in size, with depth
EDUCATION PRACTICE. Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? Clinical Scenario.
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2008;6:1086 1090 EDUCATION PRACTICE Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? RICHARD J. SAAD* and WILLIAM D.
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2008;6:1086 1090 EDUCATION PRACTICE Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? RICHARD J. SAAD* and WILLIAM D.
Functional Dyspepsia. Norbert Welkovics Heine van der Walt
 Norbert Welkovics Heine van der Walt Characteristics: Central abdomen Pain or discomfort Not associated with bowel movements No structural or biochemical abnormalty Definition Part of Gastroduodenal disorders
Norbert Welkovics Heine van der Walt Characteristics: Central abdomen Pain or discomfort Not associated with bowel movements No structural or biochemical abnormalty Definition Part of Gastroduodenal disorders
TRANSPARENCY COMMITTEE OPINION. 13 December 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
Clinical Policy Title: Pharmacogenomic tests for psychiatric medications
 Clinical Policy Title: Pharmacogenomic tests for psychiatric medications Clinical Policy Number: 02.02.01 Effective Date: October 1, 2015 Initial Review Date: April 15, 2015 Most Recent Review Date: May
Clinical Policy Title: Pharmacogenomic tests for psychiatric medications Clinical Policy Number: 02.02.01 Effective Date: October 1, 2015 Initial Review Date: April 15, 2015 Most Recent Review Date: May
Management of dyspepsia in adults in primary care
 Dyspepsia Management of dyspepsia in adults in primary care June 2005. The recommendations on referral for endoscopy in this NICE guideline have been amended in line with the recommendation in the NICE
Dyspepsia Management of dyspepsia in adults in primary care June 2005. The recommendations on referral for endoscopy in this NICE guideline have been amended in line with the recommendation in the NICE
QUICK QUERIES. Topical Questions, Sound Answers
 QUICK QUERIES Topical Questions, Sound Answers Dyspepsia: An Evidence-Based Approach Alan B. R. Thomson, MD, PhD, FRCPC, FACP, FACG Presented at the University of Alberta s Medical Grand Rounds, University
QUICK QUERIES Topical Questions, Sound Answers Dyspepsia: An Evidence-Based Approach Alan B. R. Thomson, MD, PhD, FRCPC, FACP, FACG Presented at the University of Alberta s Medical Grand Rounds, University
GI update. Common conditions and concerns my patients frequently asked about
 GI update Common conditions and concerns my patients frequently asked about Specific conditions I ll try to cover today 1. Colon polyps, colorectal cancer and colonoscopy 2. Crohn s disease 3. Peptic ulcer
GI update Common conditions and concerns my patients frequently asked about Specific conditions I ll try to cover today 1. Colon polyps, colorectal cancer and colonoscopy 2. Crohn s disease 3. Peptic ulcer
Updates in Evaluation and Management of Dyspepsia and H. Pylori Infection
 Updates in Evaluation and Management of Dyspepsia and H. Pylori Infection Isabel Lee, MD Associate Professor of Health Sciences UCSF Department of Family and Community Medicine Disclosures None 2 Session
Updates in Evaluation and Management of Dyspepsia and H. Pylori Infection Isabel Lee, MD Associate Professor of Health Sciences UCSF Department of Family and Community Medicine Disclosures None 2 Session
Clinical Policy Title: Room humidifiers
 Clinical Policy Title: Room humidifiers Clinical Policy Number: 17.02.05 Effective Date: February 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date: November 16, 2016 Next Review Date:
Clinical Policy Title: Room humidifiers Clinical Policy Number: 17.02.05 Effective Date: February 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date: November 16, 2016 Next Review Date:
Clinical Policy Title: Vacuum assisted closure in surgical wounds
 Clinical Policy Title: Vacuum assisted closure in surgical wounds Clinical Policy Number: 17.03.00 Effective Date: September 1, 2015 Initial Review Date: June 16, 2013 Most Recent Review Date: August 17,
Clinical Policy Title: Vacuum assisted closure in surgical wounds Clinical Policy Number: 17.03.00 Effective Date: September 1, 2015 Initial Review Date: June 16, 2013 Most Recent Review Date: August 17,
CHAPTER 18. PEPTIC ULCER DISEASE, SELF-ASSESSMENT QUESTIONS. 1. Which of the following is not a common cause of peptic ulcer disease (PUD)?
 CHAPTER 18. PEPTIC ULCER DISEASE, SELF-ASSESSMENT QUESTIONS 1. Which of the following is not a common cause of peptic ulcer disease (PUD)? A. Chronic alcohol ingestion B. Nonsteroidal antiinflammatory
CHAPTER 18. PEPTIC ULCER DISEASE, SELF-ASSESSMENT QUESTIONS 1. Which of the following is not a common cause of peptic ulcer disease (PUD)? A. Chronic alcohol ingestion B. Nonsteroidal antiinflammatory
One-third of adults experience pain or discomfort in
 GASTROENTEROLOGY 2002;122:1270 1285 Dyspepsia Management in Primary Care: A Decision Analysis of Competing Strategies BRENNAN M. R. SPIEGEL,* NIMISH B. VAKIL, and JOSHUA J. OFMAN*, *Department of Medicine
GASTROENTEROLOGY 2002;122:1270 1285 Dyspepsia Management in Primary Care: A Decision Analysis of Competing Strategies BRENNAN M. R. SPIEGEL,* NIMISH B. VAKIL, and JOSHUA J. OFMAN*, *Department of Medicine
Clinical Policy: Fecal Calprotectin Assay Reference Number: CP.MP.135
 Clinical Policy: Reference Number: CP.MP.135 Effective Date: 11/16 Last Review Date: 11/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.135 Effective Date: 11/16 Last Review Date: 11/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening
 Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening Clinical Policy Number: 01.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21, 2015 Most Recent
Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening Clinical Policy Number: 01.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21, 2015 Most Recent
Gilles Jequier. Commercial Director Organobalance, a Novozymes Company
 "Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
"Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
Setting The setting was primary and secondary care. The economic study was carried out in the UK.
 Helicobacter pylori "test and treat" or endoscopy for managing dyspepsia: an individual patient data meta-analysis Ford A C, Qume M, Moayyedi P, Arents N L, Lassen A T, Logan R F, McColl K E, Myres P,
Helicobacter pylori "test and treat" or endoscopy for managing dyspepsia: an individual patient data meta-analysis Ford A C, Qume M, Moayyedi P, Arents N L, Lassen A T, Logan R F, McColl K E, Myres P,
Setting The setting was secondary care. The economic study was carried out in Hong Kong, China.
 Cost-effectiveness of Helicobacter pylori "test and treat" for patients with typical reflux symptoms in a population with a high prevalence of H. pylori infection: a Markov model analysis You J H, Wong
Cost-effectiveness of Helicobacter pylori "test and treat" for patients with typical reflux symptoms in a population with a high prevalence of H. pylori infection: a Markov model analysis You J H, Wong
Assessment of symptomatic response as predictor of Helicobacter pylori status following eradication therapy in patients with ulcer
 618 University Department of Medicine and Therapeutics, Western Infirmary, Glasgow G11 6NT, UK K E L McColl A El-Nujumi L S Murray E M El-Omar A Dickson A W Kelman T E Hilditch Correspondence to: Professor
618 University Department of Medicine and Therapeutics, Western Infirmary, Glasgow G11 6NT, UK K E L McColl A El-Nujumi L S Murray E M El-Omar A Dickson A W Kelman T E Hilditch Correspondence to: Professor
University Medical Center at Brackenridge. Gastroenterology Clinic Worksheet
 Gastroenterology Clinic Worksheet 1. GI Bleeding (occult or symptomatic) a. CBC b. Iron, Ferritin b. Medication history 2. Iron Deficiency Anemia and no evident source (if no iron deficiency consider hematological
Gastroenterology Clinic Worksheet 1. GI Bleeding (occult or symptomatic) a. CBC b. Iron, Ferritin b. Medication history 2. Iron Deficiency Anemia and no evident source (if no iron deficiency consider hematological
Type of intervention Diagnosis. Economic study type Cost-effectiveness analysis.
 Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Clinical Policy Title: Altered auditory feedback devices for speech dysfluency (stuttering)
 Clinical Policy Title: Altered auditory feedback devices for speech dysfluency (stuttering) Clinical Policy Number: 17.02.02 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent
Clinical Policy Title: Altered auditory feedback devices for speech dysfluency (stuttering) Clinical Policy Number: 17.02.02 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent
Clinical Policy Title: Tactile breast imaging
 Clinical Policy Title: Tactile breast imaging Clinical Policy Number: 05.01.07 Effective Date: February 1, 2018 Initial Review Date: November 16, 2017 Most Recent Review Date: January 11, 2018 Next Review
Clinical Policy Title: Tactile breast imaging Clinical Policy Number: 05.01.07 Effective Date: February 1, 2018 Initial Review Date: November 16, 2017 Most Recent Review Date: January 11, 2018 Next Review
Comparison of stool antigen and urea breath test (UBT) methods for detection of Helicobacter pylori infection and the risk factors
 International Journal of Current Research in Medical Sciences ISSN: 2454-5716 P-ISJN: A4372-3064, E -ISJN: A4372-3061 www.ijcrims.com Original Research Article Volume 3, Issue 12-2017 Comparison of stool
International Journal of Current Research in Medical Sciences ISSN: 2454-5716 P-ISJN: A4372-3064, E -ISJN: A4372-3061 www.ijcrims.com Original Research Article Volume 3, Issue 12-2017 Comparison of stool
instrument. When 13C-UBT positive value is greater than or equal to / - 0.4, the the subject can be 1. Data and methods details are as follows:
 Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Clinical Policy: Gastric Electrical Stimulation Reference Number: CP.MP.40
 Clinical Policy: Reference Number: CP.MP.40 Effective Date: 09/09 Last Review Date: 10/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.40 Effective Date: 09/09 Last Review Date: 10/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
A Trip Through the GI Tract: Common GI Diseases and Complaints. Jennifer Curtis, MD
 A Trip Through the GI Tract: Common GI Diseases and Complaints Jennifer Curtis, MD Colon Cancer How does it develop? Most cancers arise from polyps Over time these can turn into cancer Combination of genetic
A Trip Through the GI Tract: Common GI Diseases and Complaints Jennifer Curtis, MD Colon Cancer How does it develop? Most cancers arise from polyps Over time these can turn into cancer Combination of genetic
Clinical Policy Title: Ear tubes (tympanostomy)
 Clinical Policy Title: Ear tubes (tympanostomy) Clinical Policy Number: 11.03.05 Effective Date: January 1, 2015 Initial Review Date: September 17, 2014 Most Recent Review Date: September 21, 2017 Next
Clinical Policy Title: Ear tubes (tympanostomy) Clinical Policy Number: 11.03.05 Effective Date: January 1, 2015 Initial Review Date: September 17, 2014 Most Recent Review Date: September 21, 2017 Next
The Nobel Prize in Physiology or Medicine for 2005
 The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
Investigating dyspepsia Rocco Maurizio Zagari, Lorenzo Fuccio, Franco Bazzoli
 DATE: 11/5/2008-10:27:27 ID:(BMJ)zagr584193 /(Jouve)bmj-001985 DOI: 10.1136/bmj.a1400 Topic(s): Type: InSection:review-article For the full versions of these articles see bmj.com CLINICAL REVIEW Investigating
DATE: 11/5/2008-10:27:27 ID:(BMJ)zagr584193 /(Jouve)bmj-001985 DOI: 10.1136/bmj.a1400 Topic(s): Type: InSection:review-article For the full versions of these articles see bmj.com CLINICAL REVIEW Investigating
Update on the pathological classification of gastritis. Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada
 Update on the pathological classification of gastritis Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada CLASSIFICATION GASTRITIS GASTROPATHY 1. Acute 2. Chronic 3. Uncommon
Update on the pathological classification of gastritis Hala El-Zimaity, M.D. M.S. Epidemiology McMaster University Hamilton, Ontario Canada CLASSIFICATION GASTRITIS GASTROPATHY 1. Acute 2. Chronic 3. Uncommon
KK College of Nursing Peptic Ulcer Badil D ass Dass, Lecturer 25th July, 2011
 KK College of Nursing Peptic Ulcer Badil Dass, Lecturer 25 th July, 2011 Objectives: By the end of this lecture, the students t will be able to: Define peptic pp ulcer Describe the etiology and pathology
KK College of Nursing Peptic Ulcer Badil Dass, Lecturer 25 th July, 2011 Objectives: By the end of this lecture, the students t will be able to: Define peptic pp ulcer Describe the etiology and pathology
Clinical Policy Title: Pharmocogenetic testing for warfarin (Coumadin ) sensitivity
 Clinical Policy Title: Pharmocogenetic testing for warfarin (Coumadin ) sensitivity Clinical Policy Number: 02.01.13 Effective Date: September 1, 2013 Initial Review Date: May 15, 2013 Most Recent Review
Clinical Policy Title: Pharmocogenetic testing for warfarin (Coumadin ) sensitivity Clinical Policy Number: 02.01.13 Effective Date: September 1, 2013 Initial Review Date: May 15, 2013 Most Recent Review
Developing Evidence-Based Best Practices for the Prescribing and Use of Proton Pump Inhibitors in Canada
 Developing Evidence-Based Best Practices for the Prescribing and Use of Proton Pump Inhibitors in Canada Presented by: Sumeet R. Singh, COMPUS April 4, 2006 Background COMPUS Objective: To identify and
Developing Evidence-Based Best Practices for the Prescribing and Use of Proton Pump Inhibitors in Canada Presented by: Sumeet R. Singh, COMPUS April 4, 2006 Background COMPUS Objective: To identify and
Treating H. pylori in 2016
 Treating H. pylori in 2016 William D. Chey, MD, FACG Professor of Medicine University of Michigan The Case: A 38 yo Russian man presents with recurrent epigastric pain which occurs after meals and sometimes
Treating H. pylori in 2016 William D. Chey, MD, FACG Professor of Medicine University of Michigan The Case: A 38 yo Russian man presents with recurrent epigastric pain which occurs after meals and sometimes
Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection
 Gastroenterology Research and Practice Volume 211, Article ID 946, 6 pages doi:1.1155/211/946 Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection S. Redéen, 1, 2 F. Petersson,
Gastroenterology Research and Practice Volume 211, Article ID 946, 6 pages doi:1.1155/211/946 Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection S. Redéen, 1, 2 F. Petersson,
CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University
 CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University 1 Definition of FGID Chronic and recurrent symptoms of the gastrointestinal
CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University 1 Definition of FGID Chronic and recurrent symptoms of the gastrointestinal
COMPUS OPTIMAL THERAPY REPORT. Supporting Informed Decisions. À l appui des décisions éclairées
 OPTIMAL THERAPY REPORT COMPUS Volume 1, Issue 5 March 2007 Current Practice Analysis Report for the Prescribing and Use of Proton Pump Inhibitors (PPIs) Supporting Informed Decisions À l appui des décisions
OPTIMAL THERAPY REPORT COMPUS Volume 1, Issue 5 March 2007 Current Practice Analysis Report for the Prescribing and Use of Proton Pump Inhibitors (PPIs) Supporting Informed Decisions À l appui des décisions
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師
 Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY. Robin Warren
 - Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
Treatment and Screening of H. pylori Infection in Alaskan Populations
 Treatment and Screening of H. pylori Infection in Alaskan Populations Matthew F. Deraedt, Pharm.D. Lieutenant United States Public Health Service Alaska Native Medical Center Pharmacy Practice PGY-1 Resident
Treatment and Screening of H. pylori Infection in Alaskan Populations Matthew F. Deraedt, Pharm.D. Lieutenant United States Public Health Service Alaska Native Medical Center Pharmacy Practice PGY-1 Resident
Setting The setting was primary care. The economic analysis was conducted in Glasgow, UK.
 Helicobacter pylori eradication for peptic ulceration: an observational study in a Scottish primary care setting Forrest E H, MacKenzie J F, Stuart R C, Morris A J Record Status This is a critical abstract
Helicobacter pylori eradication for peptic ulceration: an observational study in a Scottish primary care setting Forrest E H, MacKenzie J F, Stuart R C, Morris A J Record Status This is a critical abstract
Helicobacter Pylori Test-and-Treat Strategy for Management of Dyspepsia: A Comprehensive Review
 Citation: (2013) 4, e32; doi:10.1038/ctg.2013.3 & 2013 the American College of Gastroenterology All rights reserved 2155-384X/13 www.nature.com/ctg CLINICAL/NARRATIVE REVIEW Helicobacter Pylori Test-and-Treat
Citation: (2013) 4, e32; doi:10.1038/ctg.2013.3 & 2013 the American College of Gastroenterology All rights reserved 2155-384X/13 www.nature.com/ctg CLINICAL/NARRATIVE REVIEW Helicobacter Pylori Test-and-Treat
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse What is gastritis? Gastritis is a condition in which the stomach lining known as the mucosa is inflamed, or swollen. The stomach lining contains
Gastritis National Digestive Diseases Information Clearinghouse What is gastritis? Gastritis is a condition in which the stomach lining known as the mucosa is inflamed, or swollen. The stomach lining contains
Bleeding in the Digestive Tract
 Bleeding in the Digestive Tract National Digestive Diseases Information Clearinghouse National Institute of Diabetes and Digestive and Kidney Diseases NATIONAL INSTITUTES OF HEALTH U.S. Department of Health
Bleeding in the Digestive Tract National Digestive Diseases Information Clearinghouse National Institute of Diabetes and Digestive and Kidney Diseases NATIONAL INSTITUTES OF HEALTH U.S. Department of Health
Setting The setting was primary care. The economic study was conducted in Canada.
 Treating Helicobacter pylori infection in primary care patients with uninvestigated dyspepsia: the Canadian adult dyspepsia empiric treatment - Helicobacter pylori positive (CADET-Hp) randomised controlled
Treating Helicobacter pylori infection in primary care patients with uninvestigated dyspepsia: the Canadian adult dyspepsia empiric treatment - Helicobacter pylori positive (CADET-Hp) randomised controlled
Clinical Policy Title: Computerized gait analysis
 Clinical Policy Title: Computerized gait analysis Clinical Policy Number: 15.01.01 Effective Date: October 1, 2014 Initial Review Date: May 21, 2014 Most Recent Review Date: June 22, 2017 Next Review Date:
Clinical Policy Title: Computerized gait analysis Clinical Policy Number: 15.01.01 Effective Date: October 1, 2014 Initial Review Date: May 21, 2014 Most Recent Review Date: June 22, 2017 Next Review Date:
Functional Dyspepsia
 Functional Dyspepsia American College of Gastroenterology Boston Massachusetts, June 2015 Brian E. Lacy, PhD, MD, FACG Professor of Medicine Geisel School of Medicine at Dartmouth Chief, Section of Gastroenterology
Functional Dyspepsia American College of Gastroenterology Boston Massachusetts, June 2015 Brian E. Lacy, PhD, MD, FACG Professor of Medicine Geisel School of Medicine at Dartmouth Chief, Section of Gastroenterology
