Research Article. Katherine M. Bell-McGuinn, 1 Alfred L. Garfall, 1 Matthew Bogyo, 2 Douglas Hanahan, 3 and Johanna A. Joyce 1. Abstract.
|
|
|
- Camron Craig
- 5 years ago
- Views:
Transcription
1 Research Article Inhibition of Cysteine Cathepsin Protease Activity Enhances Chemotherapy Regimens by Decreasing Tumor Growth and Invasiveness in a Mouse Model of Multistage Cancer Katherine M. Bell-McGuinn, 1 Alfred L. Garfall, 1 Matthew Bogyo, 2 Douglas Hanahan, 3 and Johanna A. Joyce 1 1 Cancer Biology and Genetics Program, Memorial Sloan-Kettering Cancer Center, New York, New York; 2 Departments of Pathology, Microbiology and Immunology, Stanford School of Medicine, Stanford, California; and 3 Department of Biochemistry and Biophysics, Diabetes Center, and Comprehensive Cancer Center, University of California at San Francisco, San Francisco, California Abstract Increases in protease expression and activity are associated with malignant progression and poor patient prognosis in a number of human cancers. Members of the papain family of cysteine cathepsins are among the protease classes that have been functionally implicated in cancer. Inhibition of the cysteine cathepsin family using a pan-cathepsin inhibitor, JPM-OEt, led to tumor regression in the RIP1-Tag2 (RT2) mouse model of pancreatic islet cell tumorigenesis. The present study was designed to determine whether this cathepsin inhibitor, when used in combination with chemotherapy, would increase antitumor efficacy. RT2 mice were treated in a late-stage regression trial with three different chemotherapy regimens, alone or in combination with the cathepsin inhibitor, JPM-OEt. Cyclophosphamide was administered in either a maximum tolerated dose (MTD) regimen, a metronomic continuous low-dose regimen, or a chemoswitch regimen consisting of MTD followed by metronomic dosing. Mice were sacrificed at a defined end point and tumor burden was assessed followed by a detailed analysis of cell proliferation, apoptosis, vascularization, and invasiveness in the treated and control lesions. An additional cohort of mice was followed for survival analysis. The cathepsin inhibitor plus the chemo-switch regimen of cyclophosphamide led to the most pronounced reduction in tumor burden and greatest increase in overall survival. Cysteine cathepsin inhibition resulted in a significant decrease in tumor invasiveness, which was further augmented in combination with each of the chemotherapy dosing regimens. These results encourage the development and continuing evaluation of cysteine cathepsin inhibitors as cancer therapeutics. [Cancer Res 2007;67(15): ] Introduction Among the critical hallmark capabilities that cancers must acquire in their progression to malignancy are the ability to switch on angiogenesis, activate invasion, and promote metastasis (1). Proteolysis is central to all of these processes. Controlled degradation of the extracellular matrix (ECM) and vascular or epithelial basement membranes (BM) is necessary for angiogenesis Requests for reprints: Johanna A. Joyce, Cancer Biology and Genetics Program, Memorial Sloan-Kettering Cancer Center, Box 372, 1275 York Avenue, New York, NY Phone: ; Fax: ; joycej@mskcc.org. I2007 American Association for Cancer Research. doi: / can and invasion of tumor cells, both into the surrounding normal tissue and the blood and lymphatic systems. BM/ECM remodeling is mediated in an orchestrated manner by several families of matrix-degrading enzymes, including proteases of the cysteine, serine, and matrix metalloproteinase (MMP) classes, as well as endoglycosidases, such as heparanase. However, tumor cell growth, invasion, and metastasis rely not only on capabilities intrinsic to the cancerous cells but also on the ability of the tumor to effectively interact with and modify the tissue microenvironment, both locally and at distant metastatic sites. The tumor microenvironment consists of a multitude of cells including innate and adaptive immune cells, stromal fibroblasts, and blood and lymphatic vascular networks. Increasing awareness of the complex and reciprocal interactions between cancerous and stromal cells has broadened our understanding of how tumors develop and progress (2, 3) and, in the case of the tumor-associated vasculature, has already led to effective treatments (reviewed in ref. 4). In many cases, it is the host stromal cells, rather than the cancer cells, that produce proteolytic enzymes in the tumor microenvironment, and several studies in preclinical cancer models have shown the critical importance of host-supplied proteases to tumor progression (5 7). Furthermore, numerous clinical reports have documented a significant association between the increased expression and/or activity of several members of these proteolytic enzyme families and poor prognosis (reviewed in refs. 8 10). Thus, proteolytic enzymes produced by tumor cells and/or surrounding tumor-associated stromal cells represent attractive candidates for anticancer therapies. However, clinical trials of MMP inhibitors were unsuccessful due to poor efficacy and toxicity (11), and preclinical trials involving xenograft tumor models were equivocal. More recent studies involving genetically engineered mouse models of cancer suggest that MMP inhibitors could have utility, if administered in the correct regimens and likely at early stages of tumor development (12, 13), providing that toxicities associated with MMP inhibition in normal tissues can be limited. Alternatively, the papain family of cysteine cathepsin proteases has been reported recently as a new class with potentially broader mechanistic efficacy and limited toxicity (14). The human family of cysteine cathepsins includes 11 members (cathepsins B, C, F, H, K, L, O, S, V, W, and X), which share a conserved active site (15, 16). They primarily function as intracellular proteases mediating terminal nonspecific bulk proteolysis in the acidic environment of lysosomes (15). However, in a variety of malignant tumors, cysteine cathepsins are overexpressed and have altered localization to the invasive tumor margin (reviewed in ref. 16). Furthermore, their increased expression correlates with more aggressive tumors and poorer prognoses for Cancer Res 2007; 67: (15). August 1,
2 Cysteine Cathepsin Inhibition Decreases Tumor Invasion patients (reviewed in ref. 8). In vitro studies have shown that inhibition of cysteine cathepsin activity can reduce tumor cell invasiveness and growth (17, 18). In addition, recent experiments in which cathepsins B, L, or S were deleted in transgenic mouse models of pancreatic islet or mammary cancer resulted in significant reductions in tumor burden (7, 19 21) and reduced invasiveness in the RIP1-Tag2 (RT2) model (19), thus validating these proteases as critical targets of the broad-spectrum cysteine cathepsin inhibitor, JPM-OEt. We showed previously that treatment with JPM-OEt impaired tumor growth, vascularity, and invasiveness; there was, however, no significant effect on tumor cell apoptosis (14). We hypothesized, therefore, that the addition of cytotoxic chemotherapy to cysteine cathepsin inhibition might improve antitumor efficacy by targeting these distinctive hallmark capabilities. Cytotoxic chemotherapy has classically been given in a maximum tolerated dose (MTD) regimen. Large doses of chemotherapy are given in episodic dosing schedules to allow for recovery from cytotoxic effects on normal tissues, most notably in the rapidly dividing hematopoietic and mucosal lining cells (22). Recently, an alternative chemotherapeutic regimen, involving metronomic dosing, has been receiving increasing attention (23 25). Metronomic dosing provides a frequent, low dose of chemotherapy, typically administered daily or continuously. This dosing regimen was initially developed for its apoptotic effects on the tumor vasculature (25), but it has also been shown to have tumoricidal effects (26, 27). Prior studies showed that metronomic dosing of cyclophosphamide is a safe and efficacious treatment option in the mouse model of multistage cancer used in the present studies (28). Additional experiments have shown that a combination chemo-switch regimen using MTD cyclophosphamide followed by metronomic dose cyclophosphamide is more effective than either individual dosing schedule at reducing tumor growth and increasing survival in RT2 mice (29). The RT2 transgenic mouse model of pancreatic islet cell carcinogenesis provides a highly reproducible model of tumorigenesis that progresses through defined sequential stages, including hyperplastic/dysplastic islets, angiogenic islets, and progressive grades of carcinoma (30). This model has been used extensively in preclinical trials to test the efficacy of various chemotherapeutic and antiangiogenic regimens when given at defined windows during the course of tumor progression (29, 31, 32). In the present study, we have chosen to use the regression trial design with treatment between 12 and 16 weeks of age to focus our investigation on well-established carcinomas. This situation is most comparable with the clinical setting in which a patient presents for treatment with a significant tumor burden. The chemotherapy regimen of cyclophosphamide, although not typically used for pancreatic neuroendocrine tumors, was chosen based on prior studies showing efficacy of this agent in this model system (28, 29). The cysteine cathepsin inhibitor JPM-OEt, which binds covalently in the active site and irreversibly inhibits the cysteine cathepsin family (14, 33), was used alone or in combination with the cyclophosphamide treatment regimens. In the present study, cathepsin inhibition was investigated in combination with cyclophosphamide when given in MTD, metronomic, or chemo-switch dosing regimens. Materials and Methods Experimental animals. The generation of RT2 mice as a model of pancreatic islet cell carcinogenesis has been reported previously (30). Mice were maintained in accordance with institutional guidelines at Memorial Sloan-Kettering Cancer Center (MSKCC) and University of California at San Francisco governing the care of laboratory animals. From 12 weeks of age, all RT2 mice received 50% sugar food (Harlan Teklad) and 5% sugar water to relieve hypoglycemia induced by the insulin-secreting tumors. Drugs and treatments. The cysteine cathepsin inhibitor JPM-OEt was synthesized (34) and administered as described previously (14). JPM-OEt was administered in 20% DMSO/80% H 2 O twice daily by i.p. injection for a total dose of 50 mg/kg/d. The MTD regimen of cyclophosphamide (Sigma) was delivered as a previously reported 21-day cycle of 105 mg/kg administered i.p. every other day for a total of three doses, followed by 2 weeks of rest (24). An additional three doses were given to complete the 4-week treatment period in the regression study. For the survival studies, the 3-week cycle (1 week MTD, 2 weeks rest) was repeated as necessary. The metronomic dosing of cyclophosphamide was administered via the drinking water as reported previously (29), resulting in a dose of 10 mg/kg/d for each mouse. For the survival studies, metronomic dosing was continued without interruption. The chemo-switch regimen incorporated an initial three doses of MTD treatment followed by metronomic dosing of cyclophosphamide for the remainder of the 4-week treatment interval in the regression trial. For the survival studies in the chemo-switch treated mice, metronomic dosing was continued without interruption after the initial MTD dosing in week 12. Experimental trials and tumor burden analysis. Drug treatments were started when mice reached the age of 12 weeks and continued until mice were 16 weeks for the regression trials. For the survival trials, mice were continuously monitored for signs of hypoglycemic shock or drug side effects and were sacrificed if found moribund and cachexic or if body weight loss exceeded 15%. Mice were anesthetized and underwent heart perfusion using 10 ml PBS followed by 10 ml of 10% zinc-buffered formalin (Medical Chemical Corp.). The pancreas and spleen of the mice were dissected and macroscopic tumors (z1 1 mm) were excised and measured. Tumor volume was calculated using the formula V = a b 2 p / 6 where a and b equal the longer and shorter diameter of the tumor, respectively. The volumes of all tumors from each mouse were added to give the total tumor burden per animal. Tissue preparation and immunohistochemistry. Tissues were prepared as described previously for frozen and paraffin embedding (35). FITC-lectin (36), bromodeoxyuridine (BrdUrd) staining, and H&E grading (35) were done as described previously. Microvasculature in mice not perfused with FITC-lectin was visualized using a purified rat anti-mouse pan-endothelial cell antigen monoclonal antibody (mab; MECA-32, BD PharMingen) and a donkey anti-rat Alexa Fluor 488 secondary antibody (Molecular Probes). Apoptotic cells were visualized using a rabbit anticleaved caspase-3 antibody (Cell Signaling Technology) and a goat antirabbit Alexa Fluor 568 secondary antibody (Molecular Probes). Tissues were counterstained with 4,6-diamidino-2-phenylindole containing mounting medium (Vector Laboratories). Analysis of cell proliferation, apoptosis, tumor vasculature, and tumor invasion. In all histologic analyses, 16-week-old treated tumors were compared with untreated controls at the 12-week-old starting point of the regression trial because control RT2 mice do not survive to 16 weeks in the absence of treatment. In mice bearing more than five tumors, five tumors were selected at random for analysis. For mice with five tumors or less, all tumors were analyzed. Cell proliferation was quantified as the percentage of BrdUrd-positive cells out of the total number of tumor cells per high-power (400) field. Tumors were analyzed from three to six mice per treatment group with the exception of the metronomic treatment group for which there was only one mouse available for BrdUrd analysis (total, 434 fields). Tumor cell apoptosis was quantified by counting the number of cleaved caspase-3 positive cells per high-power (400) field. Tumors were analyzed from three to five mice per treatment group (total, 472 fields). Microvessel density (MVD) was quantified by counting the total number of FITC-lectin positive or MECA-32 positive structures observed in each low-power (200) field. Vessel branching was quantified by dividing the number of vessel segments (defined as segments between successive branchpoints) over the total vessel length, essentially as described previously (37). For MVD and branching analyses, two comparisons were done: first, lesions of Cancer Res 2007; 67: (15). August 1, 2007
3 Cancer Research all sizes were analyzed and second, a subgroup of larger tumors (defined as area z microscope fields, corresponding to f45% of all lesions analyzed; tumor volume, z3 mm 3 ) were separately analyzed. Tumors were analyzed from two to six mice per treatment group, for a total of 344 microscope fields (200), using Volocity 4.0 imaging software (Improvision). For analysis of tumor invasion, tissue sections were stained by H&E and graded as described previously (35). All tumors in each of the treatment and control groups were analyzed (total, 360 tumors). For all analyses, tumors were scored and graded blindly by K.M.B.M, A.L.G., or J.A.J. and subsequently decoded. Statistical analysis. For analysis of tumor burden, cell proliferation, apoptosis, MVD, and vessel branching, means and SEs were calculated for all tumors from each treatment group using the Kruskal-Wallis test followed by the Dunn s multiple comparisons test (InStat, GraphPad Software, Inc.). Statistical comparison of tumor grades was done using a cumulative logit model (38) with generalized estimating equations to correct for correlations within individual mice. Results Cathepsin inhibition in combination with a chemo-switch regimen results in significant tumor regression and increased overall survival. Treatment of RT2 mice was started at 12 weeks of age, at a point when the mice had a substantial tumor burden, and at best only a few weeks to live. Cohorts of control RT2 mice were sacrificed at 12 weeks of age to determine the initial baseline tumor burden at the starting point of the trial, and also at 13.5 weeks, which is end stage in the absence of treatment (Fig. 1A). As shown in Fig. 1A, tumors progress rapidly in untreated RT2 mice with the mean tumor burden increasing from to mm 3 in 1.5 weeks. We could not observe control RT2 mice beyond the week time point because they become ill from their tumor burden and die shortly thereafter in the absence of treatment. Mice treated with MTD chemotherapy showed a widely variable tumor burden (Fig. 1A), as some tumors seemed to become resistant to this treatment with time, whereas others continued to respond, as has been reported previously in this model (29). This phenomenon is also seen in the clinic as patients tumors become resistant to MTD cytotoxic regimens and often recur aggressively over time. Consistent with previously reported results, daily treatment with the cysteine cathepsin inhibitor JPM-OEt reduced tumor burden when compared with the 13.5-week controls (Fig. 1A). The combination of JPM-OEt with the MTD regimen resulted in a more consistent response from all tumors, compared with MTD treatment alone, and a significant decline in tumor burden compared with the 13.5-week controls (P < 0.05; Fig. 1A). Figure 1. Effects of cathepsin inhibition combined with distinct chemotherapy dosing regimens on tumor burden and overall survival. A, tumor volume of untreated RT2 mice at 12 wks (control; n = 15) and 13.5 wks (control; n = 8) and mice at 16 wks after 4 wks of treatment with the pan-cysteine cathepsin inhibitor JPM-OEt (JPM; n = 12), MTD cyclophosphamide (MTD; n = 17), JPM-OEt plus MTD cyclophosphamide (JPM+MTD; n = 12), metronomic dose cyclophosphamide (Met; n = 11), JPM-OEt plus metronomic dose cyclophosphamide (JPM+Met; n = 10), the chemo-switch regimen of MTD cyclophosphamide followed by metronomic dose cyclophosphamide (Switch; n = 12), or JPM-OEt plus the chemo-switch regimen (JPM+Switch; n = 13). *, P < 0.05; ***, P < B, survival of RT2 mice after no treatment (control; n = 9) or after treatment with the pan-cysteine cathepsin inhibitor JPM-OEt (n = 7), MTD cyclophosphamide (n = 6), JPM-OEt plus MTD cyclophosphamide (n = 7), the chemo-switch regimen of MTD cyclophosphamide followed by metronomic dose cyclophosphamide (n = 7), or JPM-OEt plus the chemo-switch regimen (n = 8). Cancer Res 2007; 67: (15). August 1,
4 Cysteine Cathepsin Inhibition Decreases Tumor Invasion Figure 2. Effects of cathepsin inhibition combined with distinct chemotherapy dosing regimens on apoptosis. A, apoptotic cells were detected using an antibody against cleaved caspase-3 and representative images are shown for control 12-week-old RT2 mice (a) and 16-week-old RT2 mice treated with JPM-OEt (b), MTD cyclophosphamide (c), JPM-OEt plus MTD cyclophosphamide (d), metronomic cyclophosphamide (e), JPM-OEt plus metronomic cyclophosphamide (f), chemo-switch cyclophosphamide (g), and JPM-OEt plus chemo-switch cyclophosphamide (h). Bar, 50 Am. B, the relative numbers of apoptotic cells (cleaved caspase-3 positive) per high-power field (400) in 16-week-old treated mice versus 12-week-old control mice. Columns, mean; bars, SE. **, P < Metronomically dosed cyclophosphamide administered either alone or in combination with JPM-OEt did not have a significant effect on total tumor volume (Fig. 1A). The most pronounced decline in tumor burden was observed in the chemo-switch groups (P < 0.001; Fig. 1A). These results indicate the initial MTD chemotherapy is able to debulk the tumor, whereas the metronomic therapy evidently reduces regrowth/relapse and tumor progression. Notably, the addition of JPM-OEt to the chemo-switch regimen led to an even further regression in tumor burden (P < 0.001; Fig. 1A). Given the intriguing effects on reducing tumor volume following the MTD and chemo-switch regimens in combination with cysteine cathepsin inhibition, further studies were done to analyze the potential outcome of these treatments on overall survival. Whereas the median overall survival was not significantly increased in mice treated with JPM-OEt compared with controls, we observed a trend toward increased survival when JPM-OEt was given in combination with MTD cyclophosphamide, and the greatest improvement in overall survival was seen in the chemo-switch plus JPM-OEt treated animals (Fig. 1B). In addition to significant reductions in tumor burden and increased overall survival in some of these treatment groups, it is important to note that toxicity was minimal (as assessed by weight loss, lethargy, cachexia, etc.) in the various treatment arms involving metronomic or chemo-switch cyclophosphamide alone or in combination with JPM-OEt. As expected, there was some toxicity associated with the repeated MTD regimen when administered for more than one cycle, either alone or in combination with JPM-OEt. The combination of cyclophosphamide and the cathepsin inhibitor, JPM-OEt, increased apoptosis compared with JPM- OEt treatment alone. Previous studies using JPM-OEt in regression trials in the RT2 model showed that, while reducing cell proliferation, angiogenesis, and invasion, it did not increase the frequency of tumor apoptosis (14). We hypothesized that the addition of chemotherapy would lead to an increase in cell death and thus further reduce tumor growth. In this histopathologic analysis, as well as in those described below, we analyzed tumors from all treatment arms at the defined end point of +4 weeks after initiating treatment at 12 weeks of age. Consistent with prior results (14), JPM-OEt treatment alone did not significantly affect apoptosis in 16-week RT2 tumors when compared with 12-weekold control tumors (Fig. 2). A trend toward increased levels of apoptosis was seen in the cyclophosphamide-treated groups (MTD, metronomic, and chemo-switch regimens) when analyzed at the defined 16-week end point, although the only statistically significant difference was in the metronomic cyclophosphamidetreated animals when compared with the 12-week controls (2-fold increase in apoptosis; P < 0.01; Fig. 2B). We anticipate that an overall time course of the pathologic response and future analyses of early time points in particular will reveal increased apoptosis for those regimens involving high-dose chemotherapy. We found that the addition of the cathepsin inhibitor to the MTD regimen or the metronomic dose regimen actually resulted in a trend toward fewer apoptotic cells when compared with the chemotherapy treatment alone. Based on recent data showing that RT2 mice lacking cathepsin B, S, or L had increased levels of tumor cell death (19), this result may seem surprising. However, there are several reports of cathepsins acting as effectors of apoptotic pathways (39, 40); thus, it is possible that inhibiting certain members of the cysteine cathepsin family may block apoptosis in some settings. Cathepsin inhibition in combination with either MTD or metronomic dosing of cyclophosphamide further reduces tumor cell proliferation. The cysteine cathepsin inhibitor JPM- OEt used as monotherapy has been shown previously to decrease the proliferative capacity of tumor cells as measured by BrdUrd incorporation (14). Therefore, we analyzed tumors from all groups at the +4-week treatment end point for proliferation rates. The current results with JPM-OEt alone (Fig. 3A, b) are consistent with our previous study. Cyclophosphamide, when given in MTD fashion, also reduced proliferation (P < 0.001; Fig. 3A, c), more so than JPM-OEt alone. The combination was even more pronounced (P < 0.001; Fig. 3A, d). Whereas metronomic cyclophosphamide alone did not seem to affect proliferation (Fig. 3A, e), the Cancer Res 2007; 67: (15). August 1, 2007
5 Cancer Research combination of metronomic cyclophosphamide with JPM-OEt reduced proliferation further than JPM-OEt alone (P < 0.001; Fig. 3A, f ). The chemo-switch (Fig. 3A, g) and JPM-OEt plus chemo-switch cyclophosphamide (Fig. 3A, h) regimens did not significantly alter the proliferative capacity of the tumors evident at the +4-week defined treatment end point when compared with control tumors at the 12-week starting point (Fig. 3B). Several possible explanations may account for these apparently counterintuitive observations, which will be addressed further in the discussion. Figure 3. Cell proliferation is reduced following JPM or MTD cyclophosphamide administered alone or in combination. A, proliferating cells were detected by BrdUrd incorporation and representative images are shown for control 12-week-old RT2 mice (a) and 16-week-old RT2 mice treated with JPM-OEt (b), MTD cyclophosphamide (c), JPM-OEt plus MTD cyclophosphamide (d), metronomic cyclophosphamide (e), JPM-OEt plus metronomic cyclophosphamide (f), chemo-switch cyclophosphamide (g), and JPM-OEt plus chemo-switch cyclophosphamide (h). Bar, 50 Am. B, the percentages of proliferating (BrdUrd positive) cells per high-power field (400) in 16-week-old treated mice versus 12-week-old control mice. Columns, mean; bars, SE. *, P < 0.05; ***, P < Reduced tumor vascularity following cathepsin inhibition. Next, the tumor vasculature was assessed among the various treatment groups at the defined end point of the trial by quantifying the MVD, as a measure of angiogenesis inhibition (Fig. 4). Whereas the metronomic and chemo-switch dosing of cyclophosphamide both alone and in combination with JPM-OEt showed a trend toward declining numbers of vessels, there was no change in the RT2 mice treated with combined MTD cyclophosphamide plus JPM-OEt. In fact, only the JPM-OEt treatment alone resulted in a statistically significant decline in tumor vascularity (56% reduction; P < 0.01; Fig. 4B) when compared with the 12-week control tumors, in an analysis that included lesions of all sizes. However, as there is not necessarily a linear correlation between tumor size and tumor vessel density (41), and as the dependence on angiogenesis may occur only when a tumor reaches a critical size, it is perhaps not surprising that MVD is not substantially affected in several of these treatment groups at the 16-week end point, as their tumor burden is significantly reduced compared with controls. Indeed, when we did the comparative analyses on larger tumors only (tumor volume, z3 mm 3 ) in which there may be a greater dependency on angiogenesis, the reduction in MVD was more pronounced in the JPM-OEt plus metronomic and JPM-OEt plus switch chemotherapy treatment groups when compared with those chemotherapy regimens in the absence of JPM-OEt (Fig. 4B). In certain combinations, the addition of cyclophosphamide seemed to abrogate the effect of cathepsin inhibition alone on decreasing tumor microvasculature. In fact, JPM-OEt plus MTD cyclophosphamide or MTD cyclophosphamide alone had the greatest number of microvessels seen among the treatment groups (Fig. 4B). One possible explanation for this result is that MTD cyclophosphamide treatment has been shown previously to enhance endothelial progenitor cell mobilization (42), which could effectively interfere with the ability of JPM-OEt to inhibit tumor vascularization. Alternatively, in other situations, the blood vessels dictate tissue size by maintaining a fixed vascular density, whereby reduced capability for angiogenesis restrains growth and size without evidencing reduced vascularity (43). We next asked whether, in addition to decreased MVD, the tumor vessels in the different treatment groups showed any evidence of vessel normalization. Tumor vessel normalization has been observed following several antiangiogenic therapies and proposed as a mechanism by which the efficacy of standard chemotherapy or radiotherapy is increased when combined with antiangiogenic agents, in part due to improved drug delivery (44). Tumor vessels are typically leaky, chaotic, tortuous, and disorganized, and increased vessel branching is used as a hallmark of abnormal vasculature (45). Thus, we analyzed vessel branching in the different treatment groups compared with the control group and found a decrease ranging from 22% to 31% (Fig. 4C). Again, when vessel branching was compared in larger tumors only, this reduction was most pronounced in the JPM plus chemo-switch treatment group (48% reduction; P < 0.001; Fig. 4C), the treatment group in which tumor burden was also most significantly reduced (Fig. 1A). It should be noted that whereas the treatment groups that would be expected to be antiangiogenic (JPM-OEt, metronomic, and chemo-switch) all had decreased vessel branching, the MTD-alone group did too, albeit with a smaller reduction than in each of the other groups (Fig. 4C). This may reflect, in part, changes in the vasculature that are secondary to effects on tumor cell metabolism, as discussed by Hlatky et al. (41). Cancer Res 2007; 67: (15). August 1,
6 Cysteine Cathepsin Inhibition Decreases Tumor Invasion Figure 4. Tumor vascularity and vessel branching are reduced following cathepsin inhibition. A, the tumor vasculature was visualized by perfusion of mice with FITC-lectin or by staining with a pan-endothelial cell antigen mab (MECA-32). Representative images are shown for 12-week-old control mice (a) and 16-week-old mice treated with JPM-OEt (b), MTD cyclophosphamide (c), JPM-OEt plus MTD cyclophosphamide (d), metronomic cyclophosphamide (e), JPM-OEt plus metronomic cyclophosphamide (f), chemo-switch cyclophosphamide (g), and JPM-OEt plus chemo-switch cyclophosphamide (h). Bar, 50 Am. B, MVD quantified per low-power field (200) in 16-week-old treated mice versus 12-week-old control mice. Top, MVD in all lesions analyzed; bottom, the comparison for the subgroup of larger tumors alone (defined as area z microscope fields, corresponding to f45% of all lesions analyzed; tumor volume, z3 mm 3 ). C, vessel branching quantified per low-power field (200) by dividing the number of vessel segments (as defined between successive branchpoints) over the total vessel length in millimeters. Top, vessel branching in all lesions analyzed; bottom, the comparison for the subgroup of larger tumors alone. Columns, mean; bars, SE. *, P < 0.05; **, P < 0.01; ***, P < Cathepsin inhibition is necessary and sufficient to reduce tumor invasion. Tumor invasion was assessed by grading lesions as encapsulated (Tum), microinvasive (IC1), or invasive (IC2), and representative examples of each type of tumor are shown in Fig. 5A (a c). As graphed in Fig. 5B, the majority of tumors in the untreated control animals were either microinvasive or invasive. Cyclophosphamide had no effect on the prevalence of invasive carcinomas, irrespective of the dosing regimen (MTD, metronomic, chemo-switch). In contrast, and consistent with previous studies involving JPM-OEt (14) or cathepsin gene knockouts (19), cathepsin inhibition significantly shifted the distribution of tumors to less invasive types (P < 0.05; Fig. 5B). This effect was seen in all treatment regimens containing JPM-OEt regardless of the dosing schedule of cyclophosphamide that was given in combination with the cathepsin inhibitor. Notably, the shift toward encapsulated tumors was most pronounced in the JPM-OEt plus MTD cyclophosphamide and the JPM-OEt plus chemo-switch regimen groups (P < 0.001), the two treatment groups that also showed the trend toward greatest overall survival (Fig. 1B). Discussion In this report, we show for the first time that a novel biological targeting agent, an inhibitor of cysteine cathepsin protease activity, has combinatorial efficacy with both traditional and experimental chemotherapy regimens. We compared three dosing schedules for the chemotherapeutic drug, cyclophosphamide, and found that the chemo-switch regimen (29) produced the best responses in terms of reducing tumor size and malignancy, and increasing survival, which may provide important insights into the design of future clinical trials with these agents. Whereas the combination regimen of chemo-switch cyclophosphamide and cathepsin inhibition was most effective in reducing tumor burden and extending survival in this mouse model of tumorigenesis, some of the effects of the regimen on apoptosis, proliferation, and tumor vasculature at the 16-week end point were not as pronounced compared with the other treatment arms of the study that were less efficacious in reducing overall tumor burden. This counterintuitive observation may reflect the defined time point of the analyses after 4 weeks of treatment, which may not reveal important but transitory effects occurring earlier in the therapeutic regimen. Future studies are planned to assess these parameters at earlier time points following the initiation of treatment at 12 weeks to determine if the biological effects of cathepsin inhibition in combination with chemo-switch chemotherapy are more pronounced initially. For example, it is possible that there is an initial spike of increased apoptosis and decreased cell proliferation, which debulks the tumor mass, perhaps inducing those tumors to then enter a dormant state by the time they are assessed histologically at the 16-week end point. The ability of the cysteine cathepsin inhibitor JPM-OEt to significantly reduce tumor invasiveness may ultimately be the most important benefit of this targeted agent. Prior in vitro studies showed inhibition of tumor cell invasiveness in the Cancer Res 2007; 67: (15). August 1, 2007
7 Cancer Research presence of antibody and small-molecule inhibitors targeting cathepsins (17, 18) but did not assess potential targets. Conceivable mechanisms of action of the inhibitor may include impairment of cathepsin-dependent remodeling and hence structural maintenance of the ECM and BM components, preservation of E-cadherin on cancer cells at the tumor border, perturbed cleavage of additional proteins involved in the metastatic process, or inhibition of tumor-associated macrophage function (20). Cathepsins have been shown to degrade components of the ECM and BM (46, 47), and it is likely that cathepsin inhibitors allow the local tissue environment to retain structural integrity, thus impairing tumor cell invasion. Indeed, we have shown recently that certain cathepsins cleave the cell adhesion protein, E-cadherin, in vitro and in vivo (19, 20). Levels of E-cadherin were maintained in RT2 mice lacking cathepsins B, L, or S, all of which had significant reductions in tumor invasion (19). Further investigation is currently under way to delineate the exact mechanism whereby cysteine cathepsin inhibitors decrease the invasive potential of tumors in combination with chemotherapy, a unique and important mode of action of these agents. One explanation may be that combining chemotherapy with JPM-OEt simply further reduces the overall pool of tumor cells that could ultimately develop invasive behavior when compared with JPM-OEt alone. However, cathepsin inhibition is clearly required for the reduction in invasiveness in the combined treatment groups, as none of the three chemotherapy alone regimens had any effect on tumor invasion. Translational considerations. Recent pharmacokinetic studies of the JPM-OEt inhibitor have revealed that this ostensibly cellpermeable ethyl ester is converted to the carboxylic acid form in vivo by esterases in the serum (48) and therefore is not expected to be cell permeable. Indeed, we infer that the relatively low toxicity of JPM-OEt in treated mice can be attributed to its decreased ability to enter cells and inhibit normal lysosomal cathepsin activity. Moreover, we suspect that JPM-OEt is preferentially targeting cell surface or secreted cathepsins in the tumor microenvironment, effectively producing a therapeutic window that maximizes inhibition of aberrant (tumor promoting) extracellular cathepsin activity while minimizing toxic side effects by limiting inhibition of the normal lysosomal pool of cathepsin activity. In addition to the development of novel targeted inhibitors, such as JPM-OEt, changes in the dosing schedule of traditional anticancer therapies, such as chemotherapy or radiation, have been investigated as a means to improve their therapeutic efficacy. For example, in recent years, metronomic dosing of chemotherapeutic agents has gained increasing attention (25, 49). Although the idea of a chronic low dose of chemotherapy that would be relatively nontoxic to patients yet still effective against tumor cells and tumor endothelium sounds promising, comparatively few patient responses have been seen with this type of dosing schedule when given alone. Clinical trials using metronomic dose schedules have also rarely directly compared the metronomic regimen with an alternative MTD regimen or with a regimen combining the two, comparable with the chemo-switch dosing in this study. In one ongoing trial, the Southwest Oncology Group (SWOG) recently reported promising results using a chemo-switch regimen design in the neoadjuvant treatment of breast cancer patients (50). In this trial, patients were randomized to receive the standard MTD Adriamycin and cyclophosphamide followed by paclitaxel versus a chemo-switch regimen of weekly Adriamycin and daily low-dose cyclophosphamide followed by paclitaxel. Patients receiving the chemo-switch regimen had significantly more pronounced pathologic responses in their primary tumors and were more likely to have no lymph node involvement at the time of surgery (50). A similar study in the adjuvant setting is ongoing in the SWOG 0221 trial. Based on our Figure 5. Cathepsin inhibition significantly reduces tumor invasion. A, representative images showing an encapsulated tumor (a), a microinvasive carcinoma (b), and an invasive carcinoma (c). Bar, 50 Am. B, relative proportions of encapsulated (Tum), microinvasive (IC1), and highly invasive (IC2) carcinomas in 12-week RT2 controls compared with 16-week-old RT2 mice after treatment with JPM-OEt (JPM), MTD cyclophosphamide (MTD), JPM-OEt plus MTD cyclophosphamide (JPM+MTD), metronomic cyclophosphamide (Met), JPM-OEt plus metronomic cyclophosphamide (JPM+Met), chemoswitch cyclophosphamide (Switch), and JPM-OEt plus chemo-switch cyclophosphamide (JPM+Switch). Fewer invasive tumors were seen in all treatment groups receiving JPM-OEt. No decrease in invasiveness was seen in any of the cyclophosphamide treatment groups alone, regardless of the dosing schedule. *, P < 0.05; ***, P < Cancer Res 2007; 67: (15). August 1,
8 Cysteine Cathepsin Inhibition Decreases Tumor Invasion results with a preclinical model in the present study, we would encourage wider use of such chemo-switch regimen designs in future clinical trials of anticancer agents to maximize therapeutic efficacy. In conclusion, we have shown that cysteine cathepsin inhibition in combination with two distinct regimens of chemotherapy administration (MTD or chemo-switch) results in tumor regression, decreased tumor invasiveness, and increased survival. Ongoing experiments in preclinical models of other tumor types should reveal the general therapeutic feasibility of cathepsin inhibition, alone and in combination with chemotherapy. We propose that the addition of JPM-OEt to currently used cytotoxic chemotherapy regimens in the clinic has the potential to add significant benefits both in terms of a reduction in tumor burden and an increase in overall survival and that this may largely be mediated by the ability of JPM-OEt to reduce the invasive capability of tumors. Acknowledgments Received 2/13/2007; revised 4/30/2007; accepted 5/17/2007. Grant support: Sidney Kimmel Foundation for Cancer Research, V Foundation for Cancer Research, and Society of MSKCC (J.A. Joyce); NIH T32 training grant (K.M. Bell- McGuinn); the Howard Hughes Medical Institute Medical Research Training Program (A.L. Garfall); NIH U54 RR and NIH P01 CA (M. Bogyo); and the U.S. National Cancer Institute, the William F. Bowes Charitable Foundation, and an American Cancer Society Professorship (D. Hanahan). The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. We thank Jhoanna Berrocal, Danxia Ke, and Ehud Drori for excellent technical assistance and Elyn Reidel (MSKCC Epidemiology and Biostatistics Department) for excellent statistical analysis and advice. References 1. Hanahan D, Weinberg RA. The hallmarks of cancer. Cell 2000;100: Bissell MJ, Radisky D. Putting tumours in context. Nat Rev Cancer 2001;1: Joyce JA. Therapeutic targeting of the tumor microenvironment. Cancer Cell 2005;7: Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. Nature 2005;438: Coussens LM, Tinkle CL, Hanahan D, Werb Z. MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell 2000;103: Acuff HB, Carter KJ, Fingleton B, Gorden DL, Matrisian LM. Matrix metalloproteinase-9 from bone marrowderived cells contributes to survival but not growth of tumor cells in the lung microenvironment. Cancer Res 2006;66: Vasiljeva O, Papazoglou A, Kruger A, et al. Tumor cellderived and macrophage-derived cathepsin B promotes progression and lung metastasis of mammary cancer. Cancer Res 2006;66: Berdowska I. Cysteine proteases as disease markers. Clin Chim Acta 2004;342: Laufs S, Schumacher J, Allgayer H. Urokinase-receptor (u-par): an essential player in multiple games of cancer: a review on its role in tumor progression, invasion, metastasis, proliferation/dormancy, clinical outcome, and minimal residual disease. Cell Cycle 2006;5: Overall CM, Kleifeld O. Tumour microenvironment opinion: validating matrix metalloproteinases as drug targets and anti-targets for cancer therapy. Nat Rev Cancer 2006;6: Overall CM, Kleifeld O. Towards third generation matrix metalloproteinase inhibitors for cancer therapy. Br J Cancer 2006;94: Bergers G, Hanahan D. Combining antiangiogenic agents with metronomic chemotherapy enhances efficacy against late-stage pancreatic islet carcinomas in mice. Cold Spring Harb Symp Quant Biol 2002;67: Giraudo E, Inoue M, Hanahan D. An aminobisphosphonate targets MMP-9-expressing macrophages and angiogenesis to impair cervical carcinogenesis. J Clin Invest 2004;114: Joyce JA, Baruch A, Chehade K, et al. Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis. Cancer Cell 2004;5: Turk V, Turk B, Turk D. Lysosomal cysteine proteases: facts and opportunities. EMBO J 2001;20: Mohamed MM, Sloane BF. Cysteine cathepsins: multifunctional enzymes in cancer. Nat Rev Cancer 2006;6: Premzl A, Zavasnik-Bergant V, Turk V, Kos J. Intracellular and extracellular cathepsin B facilitate invasion of MCF-10A neot cells through reconstituted extracellular matrix in vitro. Exp Cell Res 2003;283: Sever N, Filipic M, Brzin J, Lah TT. Effect of cysteine proteinase inhibitors on murine B16 melanoma cell invasion in vitro. Biol Chem 2002;383: Gocheva V, Zeng W, Ke D, et al. Distinct roles for cysteine cathepsin genes in multistage tumorigenesis. Genes Dev 2006;20: Gocheva V, Joyce JA. Cysteine cathepsins and the cutting edge of cancer invasion. Cell Cycle 2007;6: Wang B, Sun J, Kitamoto S, et al. Cathepsin S controls angiogenesis and tumor growth via matrix-derived angiogenic factors. J Biol Chem 2006;281: Kaufman D, Chabner BA. Clinical strategies for cancer treatment: the role of drugs. Chapter 1. 2nd ed. In: Chabner BA, Longo D, editors. Cancer chemotherapy and biotherapy principles and practice. Philadelphia: Lippincott-Raven Publishers; Browder T, Butterfield CE, Kraling BM, et al. Antiangiogenic scheduling of chemotherapy improves efficacy against experimental drug-resistant cancer. Cancer Res 2000;60: Klement G, Baruchel S, Rak J, et al. Continuous lowdose therapy with vinblastine and VEGF receptor-2 antibody induces sustained tumor regression without overt toxicity. J Clin Invest 2000;105:R Kerbel RS, Kamen BA. The anti-angiogenic basis of metronomic chemotherapy. Nat Rev Cancer 2004;4: Kim JT, Kim JS, Ko KW, et al. Metronomic treatment of temozolomide inhibits tumor cell growth through reduction of angiogenesis and augmentation of apoptosis in orthotopic models of gliomas. Oncol Rep 2006;16: Munoz R, Man S, Shaked Y, et al. Highly efficacious nontoxic preclinical treatment for advanced metastatic breast cancer using combination oral UFT-cyclophosphamide metronomic chemotherapy. Cancer Res 2006; 66: Man S, Bocci G, Francia G, et al. Antitumor effects in mice of low-dose (metronomic) cyclophosphamide administered continuously through the drinking water. Cancer Res 2002;62: Pietras K, Hanahan D. A multitargeted, metronomic, and maximum-tolerated dose chemo-switch regimen is antiangiogenic, producing objective responses and survival benefit in a mouse model of cancer. J Clin Oncol 2005;23: Hanahan D. Heritable formation of pancreatic h-cell tumours in transgenic mice expressing recombinant insulin/simian virus 40 oncogenes. Nature 1985;315: Bergers G, Javaherian K, Lo KM, Folkman J, Hanahan D. Effects of angiogenesis inhibitors on multistage carcinogenesis in mice. Science 1999;284: Joyce JA, Freeman C, Meyer-Morse N, Parish CR, Hanahan D. A functional heparan sulfate mimetic implicates both heparanase and heparan sulfate in tumor angiogenesis and invasion in a mouse model of multistage cancer. Oncogene 2005;24: Meara JP, Rich DH. Mechanistic studies on the inactivation of papain by epoxysuccinyl inhibitors. J Med Chem 1996;39: Chehade KAH, Baruch A, Verhelst SHL, Bogyo M. An improved preparation of the activity-based probe JPM- OEt and in situ applications. Synthesis 2005;2: Lopez T, Hanahan D. Elevated levels of IGF-1 receptor convey invasive and metastatic capability in a mouse model of pancreatic islet tumorigenesis. Cancer Cell 2002;1: Inoue M, Hager JH, Ferrara N, Gerber HP, Hanahan D. VEGF-A has a critical, nonredundant role in angiogenic switching and pancreatic h cell carcinogenesis. Cancer Cell 2002;1: Kashiwagi S, Izumi Y, Gohongi T, et al. NO mediates mural cell recruitment and vessel morphogenesis in murine melanomas and tissue-engineered blood vessels. J Clin Invest 2005;115: McCullagh P. Regression models for ordinal data. J R Stat Soc 1980;42: Isahara K, Ohsawa Y, Kanamori S, et al. Regulation of a novel pathway for cell death by lysosomal aspartic and cysteine proteinases. Neuroscience 1999;91: Guicciardi ME, Miyoshi H, Bronk SF, Gores GJ. Cathepsin B knockout mice are resistant to tumor necrosis factor-a-mediated hepatocyte apoptosis and liver injury: implications for therapeutic applications. Am J Pathol 2001;159: Hlatky L, Hahnfeldt P, Folkman J. Clinical application of antiangiogenic therapy: microvessel density, what it does and doesn t tell us. J Natl Cancer Inst 2002;94: Bertolini F, Paul S, Mancuso P, et al. Maximum tolerable dose and low-dose metronomic chemotherapy have opposite effects on the mobilization and viability of circulating endothelial progenitor cells. Cancer Res 2003;63: Rupnick MA, Panigrahy D, Zhang CY, et al. Adipose tissue mass can be regulated through the vasculature. Proc Natl Acad Sci U S A 2002;99: Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science 2005;307: Jain RK. Molecular regulation of vessel maturation. Nat Med 2003;9: Buck MR, Karustis DG, Day NA, Honn KV, Sloane BF. Degradation of extracellular-matrix proteins by human cathepsin B from normal and tumour tissues. Biochem J 1992;282: Mai J, Sameni M, Mikkelsen T, Sloane BF. Degradation of extracellular matrix protein tenascin-c by cathepsin B: an interaction involved in the progression of gliomas. Biol Chem 2002;383: Sadaghiani AM, Verhelst SHL, Gocheva V, et al. Design, synthesis and evaluation of in vivo potency and selectivity of epoxysuccinyl-based inhibitors of papain family cysteine proteases. Chem Biol 2007;14: Gasparini G. Metronomic scheduling: the future of chemotherapy? Lancet Oncol 2001;2: Ellis GK, Green SJ, Russell CA, Royce ME, Perez EA, Livingston RB. SWOG 0012, a randomized phase III comparison of standard doxorubicin (A) and cyclophosphamide (C) followed by weekly paclitaxel (T) versus weekly doxorubicin and daily oral cyclophosphamide plus G-CSF (G) followed by weekly paclitaxel as neoadjuvant therapy for inflammatory and locally advanced breast cancer [abstract LBA537]. J Clin Oncol 2006;24:LBA Cancer Res 2007; 67: (15). August 1, 2007
Cancer Metronomic Therapy Milan, February 26, 2016
 Cancer Metronomic Therapy Milan, February 26, 2016 Metronomic Chemotherapy: Evolution and Development of the Concept Robert S. Kerbel, PhD Senior Scientist Sunnybrook Research Institute Professor, Dept.
Cancer Metronomic Therapy Milan, February 26, 2016 Metronomic Chemotherapy: Evolution and Development of the Concept Robert S. Kerbel, PhD Senior Scientist Sunnybrook Research Institute Professor, Dept.
Inflammatory Cells and Metastasis
 Inflammatory Cells and Metastasis Experimentelle Krebsforschung SS 07 Gerhard Christofori Institute of Biochemistry and Genetics Department of Clinical-Biological Sciences Center of Biomedicine University
Inflammatory Cells and Metastasis Experimentelle Krebsforschung SS 07 Gerhard Christofori Institute of Biochemistry and Genetics Department of Clinical-Biological Sciences Center of Biomedicine University
STATE OF THE ART 4: Combination Immune Therapy-Chemotherapy. Elizabeth M. Jaffee (JHU) James Yang (NCI) Jared Gollob (Duke) John Kirkwood (UPMI)
 STATE OF THE ART 4: Combination Immune Therapy-Chemotherapy Elizabeth M. Jaffee (JHU) James Yang (NCI) Jared Gollob (Duke) John Kirkwood (UPMI) Topics for Consideration What are the rules for integrating
STATE OF THE ART 4: Combination Immune Therapy-Chemotherapy Elizabeth M. Jaffee (JHU) James Yang (NCI) Jared Gollob (Duke) John Kirkwood (UPMI) Topics for Consideration What are the rules for integrating
RIP-Tag2 mouse model as a Paradigm for Target. Search in NETs
 RIP-Tag2 mouse model as a Paradigm for Target Search in NETs Oriol Casanovas, Ph.D. Tumor Angiogenesis Group INSTITUT CATALÀ d ONCOLOGIA IDIBELL Barcelona (SPAIN) Therapeutic Targeting of the Tumor Stroma
RIP-Tag2 mouse model as a Paradigm for Target Search in NETs Oriol Casanovas, Ph.D. Tumor Angiogenesis Group INSTITUT CATALÀ d ONCOLOGIA IDIBELL Barcelona (SPAIN) Therapeutic Targeting of the Tumor Stroma
JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T. preclinical development for cancer indications 6 and one, the vascular endothelial
 NUMBER FEBRUARY VOLUME 23 d 5 d 10 05 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T A Multitargeted, Metronomic, and Maximum-Tolerated Dose Chemo-Switch Regimen is Antiangiogenic, Producing
NUMBER FEBRUARY VOLUME 23 d 5 d 10 05 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T A Multitargeted, Metronomic, and Maximum-Tolerated Dose Chemo-Switch Regimen is Antiangiogenic, Producing
METRONOMIC CHEMOTHERAPY
 Vet Times The website for the veterinary profession https://www.vettimes.co.uk METRONOMIC CHEMOTHERAPY Author : Shasta Lynch Categories : Vets Date : October 3, 2011 Shasta Lynch looks at the promising
Vet Times The website for the veterinary profession https://www.vettimes.co.uk METRONOMIC CHEMOTHERAPY Author : Shasta Lynch Categories : Vets Date : October 3, 2011 Shasta Lynch looks at the promising
Neoplasia 18 lecture 8. Dr Heyam Awad MD, FRCPath
 Neoplasia 18 lecture 8 Dr Heyam Awad MD, FRCPath ILOS 1. understand the angiogenic switch in tumors and factors that stimulate and inhibit angiogenesis. 2. list the steps important for tumor metastasis
Neoplasia 18 lecture 8 Dr Heyam Awad MD, FRCPath ILOS 1. understand the angiogenic switch in tumors and factors that stimulate and inhibit angiogenesis. 2. list the steps important for tumor metastasis
The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis
 The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis Nam Deuk Kim, Ph.D. Pusan National University Contents Part 1. The process of angiogenesis Part 2. The role of angiopoietins
The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis Nam Deuk Kim, Ph.D. Pusan National University Contents Part 1. The process of angiogenesis Part 2. The role of angiopoietins
The Angiopoietin Axis in Cancer
 Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
Angiogenesis as a therapeutic target
 Angiogenesis as a therapeutic target Lecture Experimentelle Krebsforschung SS 07 Prof. Gerhard Christofori Institute of Biochemistry and Genetics Department of Clinical-Biological Sciences University of
Angiogenesis as a therapeutic target Lecture Experimentelle Krebsforschung SS 07 Prof. Gerhard Christofori Institute of Biochemistry and Genetics Department of Clinical-Biological Sciences University of
PATHOBIOLOGY OF NEOPLASIA
 PATHOBIOLOGY OF NEOPLASIA Department of Pathology Gadjah Mada University School of Medicine dr. Harijadi Blok Biomedis, 6 Maret 2009 [12] 3/17/2009 1 The pathobiology of neoplasia Normal cells Malignant
PATHOBIOLOGY OF NEOPLASIA Department of Pathology Gadjah Mada University School of Medicine dr. Harijadi Blok Biomedis, 6 Maret 2009 [12] 3/17/2009 1 The pathobiology of neoplasia Normal cells Malignant
Signaling Vascular Morphogenesis and Maintenance
 Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Cancer Biology Course. Invasion and Metastasis
 Cancer Biology Course Invasion and Metastasis 2016 Lu-Hai Wang NHRI Cancer metastasis Major problem: main reason for killing cancer patients, without it cancer can be cured or controlled. Challenging questions:
Cancer Biology Course Invasion and Metastasis 2016 Lu-Hai Wang NHRI Cancer metastasis Major problem: main reason for killing cancer patients, without it cancer can be cured or controlled. Challenging questions:
Sequential Dose-Dense Adjuvant Therapy With Doxorubicin, Paclitaxel, and Cyclophosphamide
 Sequential Dose-Dense Adjuvant Therapy With Doxorubicin, Paclitaxel, and Cyclophosphamide Review Article [1] April 01, 1997 By Clifford A. Hudis, MD [2] The recognition of paclitaxel's (Taxol's) activity
Sequential Dose-Dense Adjuvant Therapy With Doxorubicin, Paclitaxel, and Cyclophosphamide Review Article [1] April 01, 1997 By Clifford A. Hudis, MD [2] The recognition of paclitaxel's (Taxol's) activity
Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer
 Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer X.L. Liu 1, L.D. Liu 2, S.G. Zhang 1, S.D. Dai 3, W.Y. Li 1 and L. Zhang 1 1 Thoracic Surgery,
Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer X.L. Liu 1, L.D. Liu 2, S.G. Zhang 1, S.D. Dai 3, W.Y. Li 1 and L. Zhang 1 1 Thoracic Surgery,
Tumor Associated Macrophages as a Novel Target for Cancer Therapy
 Tumor mass Tumor Associated Macrophage Tumor Associated Macrophages as a Novel Target for Cancer Therapy This booklet contains forward-looking statements that are based on Amgen s current expectations
Tumor mass Tumor Associated Macrophage Tumor Associated Macrophages as a Novel Target for Cancer Therapy This booklet contains forward-looking statements that are based on Amgen s current expectations
Rationale for VEGFR-targeted Therapy in RCC
 Rationale for VEGFR-targeted Therapy in RCC EIKCS, Budapest, May 2013 Tim Eisen Tim Eisen - Disclosures Company Research Support Advisory Board Trial Management Group Honoraria Astra Zeneca + + + Astellas
Rationale for VEGFR-targeted Therapy in RCC EIKCS, Budapest, May 2013 Tim Eisen Tim Eisen - Disclosures Company Research Support Advisory Board Trial Management Group Honoraria Astra Zeneca + + + Astellas
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
SHREE ET AL, SUPPLEMENTAL MATERIALS. (A) Workflow for tumor cell line derivation and orthotopic implantation.
 SHREE ET AL, SUPPLEMENTAL MATERIALS SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure 1. Derivation and characterization of TS1-TGL and TS2-TGL PyMT cell lines and development of an orthotopic
SHREE ET AL, SUPPLEMENTAL MATERIALS SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure 1. Derivation and characterization of TS1-TGL and TS2-TGL PyMT cell lines and development of an orthotopic
Cancer Tumor Therapy Drug Vicrostatin Shows. Promising Inhibition of Glioma Growth and. Angiogenesis in Vivo. Rupan Bose
 Bose p. 1 of 23. Cancer Tumor Therapy Drug Vicrostatin Shows Promising Inhibition of Glioma Growth and Angiogenesis in Vivo Rupan Bose B.A. Neuroscience 2011 Candidate Senior Thesis Dana and David Dornsife
Bose p. 1 of 23. Cancer Tumor Therapy Drug Vicrostatin Shows Promising Inhibition of Glioma Growth and Angiogenesis in Vivo Rupan Bose B.A. Neuroscience 2011 Candidate Senior Thesis Dana and David Dornsife
Adverse side effects associated to metronomic chemotherapy
 Adverse side effects associated to metronomic chemotherapy Elisabetta Munzone, MD Division of Medical Senology Istituto Europeo di Oncologia Milano, Italy LDM: the optimal biological dose Although there
Adverse side effects associated to metronomic chemotherapy Elisabetta Munzone, MD Division of Medical Senology Istituto Europeo di Oncologia Milano, Italy LDM: the optimal biological dose Although there
Author's response to reviews
 Author's response to reviews Title: Rapid Induction of Orthotopic Hepatocellular Carcinoma in Immune-competent Rats by Ultrasound-guided Method and the Subsequent Low-dose Chemotherapy Authors: Hoi-Hung
Author's response to reviews Title: Rapid Induction of Orthotopic Hepatocellular Carcinoma in Immune-competent Rats by Ultrasound-guided Method and the Subsequent Low-dose Chemotherapy Authors: Hoi-Hung
Anti-angiogenesis in cancer; met and unmet goals - an interview with Robert Kerbel
 Int. J. Dev. Biol. 55: 395-398 doi: 10.1387/ijdb.103217fb www.intjdevbiol.com Anti-angiogenesis in cancer; met and unmet goals - an interview with Robert Kerbel FRANCESCO BERTOLINI* Laboratory of Hematology-Oncology,
Int. J. Dev. Biol. 55: 395-398 doi: 10.1387/ijdb.103217fb www.intjdevbiol.com Anti-angiogenesis in cancer; met and unmet goals - an interview with Robert Kerbel FRANCESCO BERTOLINI* Laboratory of Hematology-Oncology,
BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY
 BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY Cancer is a group of more than 100 different diseases that are characterized by uncontrolled cellular growth,
BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY Cancer is a group of more than 100 different diseases that are characterized by uncontrolled cellular growth,
INTRODUCTION Ovarian cancer is the leading cause of mortality from gynecologic malignancies in the industrialized countries and is responsible for
 INTRODUCTION Ovarian cancer is the leading cause of mortality from gynecologic malignancies in the industrialized countries and is responsible for more deaths than both cervical and endometrial tumours.
INTRODUCTION Ovarian cancer is the leading cause of mortality from gynecologic malignancies in the industrialized countries and is responsible for more deaths than both cervical and endometrial tumours.
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates
 Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL. PhD THESIS
 UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL PhD THESIS THE IMPORTANCE OF TUMOR ANGIOGENESIS IN CEREBRAL TUMOR DIAGNOSIS AND THERAPY ABSTRACT PhD COORDINATOR: Prof. univ. dr. DRICU Anica PhD
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL PhD THESIS THE IMPORTANCE OF TUMOR ANGIOGENESIS IN CEREBRAL TUMOR DIAGNOSIS AND THERAPY ABSTRACT PhD COORDINATOR: Prof. univ. dr. DRICU Anica PhD
Tissue renewal and Repair. Nisamanee Charoenchon, PhD Department of Pathobiology, Faculty of Science
 Tissue renewal and Repair Nisamanee Charoenchon, PhD Email: nisamanee.cha@mahidol.ac.th Department of Pathobiology, Faculty of Science Topic Objectives 1. Describe processes of tissue repair, regeneration
Tissue renewal and Repair Nisamanee Charoenchon, PhD Email: nisamanee.cha@mahidol.ac.th Department of Pathobiology, Faculty of Science Topic Objectives 1. Describe processes of tissue repair, regeneration
Supplementary Figure 1. Deletion of Smad3 prevents B16F10 melanoma invasion and metastasis in a mouse s.c. tumor model.
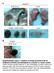 A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
I TESSUTI: Dott.ssa Liliana Belgioia Università degli Studi di Genova
 I TESSUTI: 1. Repair, Radiosensitivity, Recruitment, Repopulation, Reoxygenation 2. Acute and chronic hypoxia 3. Tissue microenvironment and tissue organization Dott.ssa Liliana Belgioia Università degli
I TESSUTI: 1. Repair, Radiosensitivity, Recruitment, Repopulation, Reoxygenation 2. Acute and chronic hypoxia 3. Tissue microenvironment and tissue organization Dott.ssa Liliana Belgioia Università degli
Backgrounder. 1. What are targeted therapies? 2. How do targeted therapies work?
 Backgrounder TARGETED THERAPIES FOR CANCER 1. What are targeted therapies? 2. How do targeted therapies work? 3. What are some of the different types of targeted therapy? 4. What are the potential benefits
Backgrounder TARGETED THERAPIES FOR CANCER 1. What are targeted therapies? 2. How do targeted therapies work? 3. What are some of the different types of targeted therapy? 4. What are the potential benefits
Heterotypy and Angiogenesis
 Heterotypy and Angiogenesis Tumors are perpetual wounds 1. Normally stroma and epithelia converse at a distance. 2. Juxtaposition of stroma and epithelia is indicative of tissue damage. 4. Activate strategies
Heterotypy and Angiogenesis Tumors are perpetual wounds 1. Normally stroma and epithelia converse at a distance. 2. Juxtaposition of stroma and epithelia is indicative of tissue damage. 4. Activate strategies
The 2010 Gastrointestinal Cancers Symposium Oral Abstract Session: Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract
 The 2010 Gastrointestinal Cancers Symposium : Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract Abstract #131: Phase I study of MK 0646 (dalotuzumab), a humanized monoclonal antibody against
The 2010 Gastrointestinal Cancers Symposium : Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract Abstract #131: Phase I study of MK 0646 (dalotuzumab), a humanized monoclonal antibody against
DAWNING OF THE AGE OF ANGIOGENESIS
 DAWNING OF THE AGE OF ANGIOGENESIS Bob Leibowitz, M.D. DIPLOMATE AMERICAN BOARDS OF INTERNAL MEDICINE AND SUBSPECIALTIES OF MEDICAL ONCOLOGY AND HEMATOLOGY December 1997 April 2004 (Revised) Angiogenesis
DAWNING OF THE AGE OF ANGIOGENESIS Bob Leibowitz, M.D. DIPLOMATE AMERICAN BOARDS OF INTERNAL MEDICINE AND SUBSPECIALTIES OF MEDICAL ONCOLOGY AND HEMATOLOGY December 1997 April 2004 (Revised) Angiogenesis
Additional file 6. Summary of experiments characterizing development of adaptive immune response
 Additional file 6. Summary of experiments characterizing development of adaptive immune response Tests performed to evaluate the development of adaptive immunity in patients are summarized in Table 1.
Additional file 6. Summary of experiments characterizing development of adaptive immune response Tests performed to evaluate the development of adaptive immunity in patients are summarized in Table 1.
Biochemistry of Carcinogenesis. Lecture # 35 Alexander N. Koval
 Biochemistry of Carcinogenesis Lecture # 35 Alexander N. Koval What is Cancer? The term "cancer" refers to a group of diseases in which cells grow and spread unrestrained throughout the body. It is difficult
Biochemistry of Carcinogenesis Lecture # 35 Alexander N. Koval What is Cancer? The term "cancer" refers to a group of diseases in which cells grow and spread unrestrained throughout the body. It is difficult
A Comparison of Endothelial Cell-Selective Adhesion Molecule and von Willebrand Factor Expression in Breast Cancer Growth and Metastasis
 Original Article Middle East Journal of Cancer; April 2018; 9(2): 85-90 A Comparison of Endothelial Cell-Selective Adhesion Molecule and von Willebrand Factor Expression in Breast Cancer Growth and Metastasis
Original Article Middle East Journal of Cancer; April 2018; 9(2): 85-90 A Comparison of Endothelial Cell-Selective Adhesion Molecule and von Willebrand Factor Expression in Breast Cancer Growth and Metastasis
BL-8040: BEST-IN-CLASS CXCR4 ANTAGONIST FOR TREATMENT OF ONCOLOGICAL MALIGNANCIES. Overview and Mechanism of Action Dr.
 BL-8040: BEST-IN-CLASS CXCR4 ANTAGONIST FOR TREATMENT OF ONCOLOGICAL MALIGNANCIES Overview and Mechanism of Action Dr. Leah Klapper, CSO 88 BL-8040: Novel CXCR4 Antagonist For Hematological Cancers Indications:
BL-8040: BEST-IN-CLASS CXCR4 ANTAGONIST FOR TREATMENT OF ONCOLOGICAL MALIGNANCIES Overview and Mechanism of Action Dr. Leah Klapper, CSO 88 BL-8040: Novel CXCR4 Antagonist For Hematological Cancers Indications:
Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach
 Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach Oncolytic immunotherapy Oncolytic immunotherapy the use of a genetically modified virus to attack tumors and induce a systemic immune response
Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach Oncolytic immunotherapy Oncolytic immunotherapy the use of a genetically modified virus to attack tumors and induce a systemic immune response
Breast Cancer: the interplay of biology, drugs, radiation. Prof. L. Livi Università degli Studi di Firenze. Brescia, October 3rd 4th, 2013
 Breast Cancer: the interplay of biology, drugs, radiation Prof. L. Livi Università degli Studi di Firenze Brescia, October 3rd 4th, 2013 BACKGROUND (1) The complex interactions between tumor-specific signaling
Breast Cancer: the interplay of biology, drugs, radiation Prof. L. Livi Università degli Studi di Firenze Brescia, October 3rd 4th, 2013 BACKGROUND (1) The complex interactions between tumor-specific signaling
Adaptive immune responses: T cell-mediated immunity
 MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
Introduction. Cancer Biology. Tumor-suppressor genes. Proto-oncogenes. DNA stability genes. Mechanisms of carcinogenesis.
 Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
Nintedanib in Oncology Backgrounder
 For media outside the US, UK and Canada only Nintedanib in Oncology Backgrounder 1. What is nintedanib? 2. How does nintedanib work? 3. Data overview 4. Additional clinical data 5. Nintedanib approval
For media outside the US, UK and Canada only Nintedanib in Oncology Backgrounder 1. What is nintedanib? 2. How does nintedanib work? 3. Data overview 4. Additional clinical data 5. Nintedanib approval
Quantitative analysis of mast cells in invasive ductal carcinoma
 ISSN: 2347-3215 Volume 2 Number 12 (December-2014) pp. 91-96 www.ijcrar.com Quantitative analysis of mast cells in invasive ductal carcinoma M.N.Divyarani*, M.L.Harendra Kumar, B.V.Swaroop raj, and U.D.Manoranjan
ISSN: 2347-3215 Volume 2 Number 12 (December-2014) pp. 91-96 www.ijcrar.com Quantitative analysis of mast cells in invasive ductal carcinoma M.N.Divyarani*, M.L.Harendra Kumar, B.V.Swaroop raj, and U.D.Manoranjan
Can we prevent metastasis?
 Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
My name is Dr. David Ilson, Professor of Medicine at Memorial Sloan Kettering Cancer Center and Weill Cornell Medical Center in New York, New York.
 Welcome to this CME/CE-certified activity entitled, Integrating the Latest Advances Into Clinical Experience: Data and Expert Insights From the 2016 Meeting on Gastrointestinal Cancers in San Francisco.
Welcome to this CME/CE-certified activity entitled, Integrating the Latest Advances Into Clinical Experience: Data and Expert Insights From the 2016 Meeting on Gastrointestinal Cancers in San Francisco.
The Need for a PARP in vivo Pharmacodynamic Assay
 The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D. Chief Scientific Officer Trevigen, Inc. Gaithersburg, MD Poly(ADP-ribose) polymerases are promising therapeutic targets. In response
The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D. Chief Scientific Officer Trevigen, Inc. Gaithersburg, MD Poly(ADP-ribose) polymerases are promising therapeutic targets. In response
1.The metastatic cascade. 2.Pathologic features of metastasis. 3.Therapeutic ramifications
 Metastasis 1.The metastatic cascade 2.Pathologic features of metastasis 3.Therapeutic ramifications Sir James Paget (1814-1899) British Surgeon/ Pathologist Paget s disease of bone Paget s disease of the
Metastasis 1.The metastatic cascade 2.Pathologic features of metastasis 3.Therapeutic ramifications Sir James Paget (1814-1899) British Surgeon/ Pathologist Paget s disease of bone Paget s disease of the
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 Page 1 09/10/2013 AD Award Number: W81XWH-12-1-0453 TITLE: The cytoplasm translocation of the androgen receptor cofactor p44 as a target for prostate cancer treatment PRINCIPAL INVESTIGATOR: Zhengxin Wang
Page 1 09/10/2013 AD Award Number: W81XWH-12-1-0453 TITLE: The cytoplasm translocation of the androgen receptor cofactor p44 as a target for prostate cancer treatment PRINCIPAL INVESTIGATOR: Zhengxin Wang
Mechanisms of Resistance to Antiangiogenic. Martin J. Edelman, MD University of Maryland Greenebaum Cancer Center Dresden, 2012
 Mechanisms of Resistance to Antiangiogenic Agents Martin J. Edelman, MD University of Maryland Greenebaum Cancer Center Dresden, 2012 Angiogenesis: A fundamental attribute of cancer Premise of Anti-angiogenic
Mechanisms of Resistance to Antiangiogenic Agents Martin J. Edelman, MD University of Maryland Greenebaum Cancer Center Dresden, 2012 Angiogenesis: A fundamental attribute of cancer Premise of Anti-angiogenic
Reprogramming Tumor Associated Dendritic Cells for Immunotherapy
 Reprogramming Tumor Associated Dendritic Cells for Immunotherapy Edgar Engleman, M.D. Professor of Pathology and Medicine Stanford University Disclosures: Founder of Dendreon, a biotechnology company that
Reprogramming Tumor Associated Dendritic Cells for Immunotherapy Edgar Engleman, M.D. Professor of Pathology and Medicine Stanford University Disclosures: Founder of Dendreon, a biotechnology company that
OMICS Journals are welcoming Submissions
 OMICS Journals are welcoming Submissions OMICS International welcomes submissions that are original and technically so as to serve both the developing world and developed countries in the best possible
OMICS Journals are welcoming Submissions OMICS International welcomes submissions that are original and technically so as to serve both the developing world and developed countries in the best possible
Supplementary Information. Detection and delineation of oral cancer with a PARP1 targeted optical imaging agent
 Supplementary Information Detection and delineation of oral cancer with a PARP1 targeted optical imaging agent Authors: Susanne Kossatz a, Christian Brand a, Stanley Gutiontov b, Jonathan T.C. Liu c, Nancy
Supplementary Information Detection and delineation of oral cancer with a PARP1 targeted optical imaging agent Authors: Susanne Kossatz a, Christian Brand a, Stanley Gutiontov b, Jonathan T.C. Liu c, Nancy
DOCTORAL THESIS SUMMARY
 UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA DOCTORAL THESIS HISTOPATHOLOGICAL AND IMMUNOHISTOCHEMICAL STUDY OF GASTRIC CARCINOMAS SUMMARY Scientific Coordinator: Univ. Prof. Dr. SIMIONESCU CRISTIANA EUGENIA
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA DOCTORAL THESIS HISTOPATHOLOGICAL AND IMMUNOHISTOCHEMICAL STUDY OF GASTRIC CARCINOMAS SUMMARY Scientific Coordinator: Univ. Prof. Dr. SIMIONESCU CRISTIANA EUGENIA
Chapter 2 Staging of Breast Cancer
 Chapter 2 Staging of Breast Cancer Zeynep Ozsaran and Senem Demirci Alanyalı 2.1 Introduction Five decades ago, Denoix et al. proposed classification system (tumor node metastasis [TNM]) based on the dissemination
Chapter 2 Staging of Breast Cancer Zeynep Ozsaran and Senem Demirci Alanyalı 2.1 Introduction Five decades ago, Denoix et al. proposed classification system (tumor node metastasis [TNM]) based on the dissemination
Comparison of CD10 expression in stroma of epithelial and mesenchymal tumors of the breast
 Global Advanced Research Journal of Medicine and Medical Science (ISSN: 2315-5159) Vol. 4(1) pp. 051-056, January, 2015 Available online http://garj.org/garjmms/index.htm Copyright 2015 Global Advanced
Global Advanced Research Journal of Medicine and Medical Science (ISSN: 2315-5159) Vol. 4(1) pp. 051-056, January, 2015 Available online http://garj.org/garjmms/index.htm Copyright 2015 Global Advanced
A holistic approach to targeting breast cancer part II: Micronutrient synergy. Presented by: Dr. Neha Shanker DRRI
 A holistic approach to targeting breast cancer part II: Micronutrient synergy Presented by: Dr. Neha Shanker DRRI Overview of the previous webinar In the last presentation we talked about: Increase in
A holistic approach to targeting breast cancer part II: Micronutrient synergy Presented by: Dr. Neha Shanker DRRI Overview of the previous webinar In the last presentation we talked about: Increase in
Therapeutic targeting neuraminidase-1 in multi-stage of tumorigenesis
 Therapeutic targeting neuraminidase-1 in multi-stage of tumorigenesis Myron R. Szewczuk Dept. Biomedical and Molecular Sciences, Queen's University, Kingston, K7L 3N6 Ontario, Canada HIGHLIGHTS. An innovative
Therapeutic targeting neuraminidase-1 in multi-stage of tumorigenesis Myron R. Szewczuk Dept. Biomedical and Molecular Sciences, Queen's University, Kingston, K7L 3N6 Ontario, Canada HIGHLIGHTS. An innovative
Pharmacokinetics, pharmacodynamics and pharmacogenetics of metronomic chemotherapy
 Divisione di Farmacologia Dipartimento di Medicina Clinica and Sperimentale Università di Pisa U.O. Farmacologia Clinica Azienda Ospedaliero Universitaria Pisana Pharmacokinetics, pharmacodynamics and
Divisione di Farmacologia Dipartimento di Medicina Clinica and Sperimentale Università di Pisa U.O. Farmacologia Clinica Azienda Ospedaliero Universitaria Pisana Pharmacokinetics, pharmacodynamics and
Clinical significance of CD44 expression in children with hepatoblastoma
 Clinical significance of CD44 expression in children with hepatoblastoma H.-Y. Cai 1 *, B. Yu 1 *, Z.-C. Feng 2, X. Qi 1 and X.-J. Wei 1 1 Department of General Surgery, General Hospital of Beijing Military
Clinical significance of CD44 expression in children with hepatoblastoma H.-Y. Cai 1 *, B. Yu 1 *, Z.-C. Feng 2, X. Qi 1 and X.-J. Wei 1 1 Department of General Surgery, General Hospital of Beijing Military
Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1
 Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1 CHRISTOPH RADER, 2 MIKHAIL POPKOV, JOHN A. NEVES, AND CARLOS F. BARBAS III 2 Department of Molecular Biology and The
Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1 CHRISTOPH RADER, 2 MIKHAIL POPKOV, JOHN A. NEVES, AND CARLOS F. BARBAS III 2 Department of Molecular Biology and The
Research Article Stromal Expression of CD10 in Invasive Breast Carcinoma and Its Correlation with ER, PR, HER2-neu, and Ki67
 SAGE-Hindawi Access to Research International Breast Cancer Volume 20, Article ID 47957, 4 pages doi:0.406/20/47957 Research Article Stromal Expression of CD0 in Invasive Breast Carcinoma and Its Correlation
SAGE-Hindawi Access to Research International Breast Cancer Volume 20, Article ID 47957, 4 pages doi:0.406/20/47957 Research Article Stromal Expression of CD0 in Invasive Breast Carcinoma and Its Correlation
Heather Wakelee, M.D.
 Heather Wakelee, M.D. Assistant Professor of Medicine, Oncology Stanford University Sponsored by Educational Grant Support from Adjuvant (Post-Operative) Lung Cancer Chemotherapy Heather Wakelee, M.D.
Heather Wakelee, M.D. Assistant Professor of Medicine, Oncology Stanford University Sponsored by Educational Grant Support from Adjuvant (Post-Operative) Lung Cancer Chemotherapy Heather Wakelee, M.D.
Corporate Medical Policy
 Corporate Medical Policy Ado-Trastuzumab Emtansine (Trastuzumab-DM1) for Treatment of File Name: Origination: Last CAP Review: Next CAP Review: Last Review: ado_trastuzumab_emtansine_(trastuzumab-dm1)_for_treatment_of_her-2_positivemalignancies
Corporate Medical Policy Ado-Trastuzumab Emtansine (Trastuzumab-DM1) for Treatment of File Name: Origination: Last CAP Review: Next CAP Review: Last Review: ado_trastuzumab_emtansine_(trastuzumab-dm1)_for_treatment_of_her-2_positivemalignancies
Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin
 Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin Hodgkin s s Lymphoma Targeted Cancer Therapies William Westlin, Ph.D. Vice President, Preclinical Research Discovery of Fumagillin
Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin Hodgkin s s Lymphoma Targeted Cancer Therapies William Westlin, Ph.D. Vice President, Preclinical Research Discovery of Fumagillin
Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San
 Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San Francisco Lung Cancer Classification Pathological Classification
Osamu Tetsu, MD, PhD Associate Professor Department of Otolaryngology-Head and Neck Surgery School of Medicine, University of California, San Francisco Lung Cancer Classification Pathological Classification
Table S2. Expression of PRMT7 in clinical breast carcinoma samples
 Table S2. Expression of PRMT7 in clinical breast carcinoma samples (All data were obtained from cancer microarray database Oncomine.) Analysis type* Analysis Class(number sampels) 1 2 3 4 Correlation (up/down)#
Table S2. Expression of PRMT7 in clinical breast carcinoma samples (All data were obtained from cancer microarray database Oncomine.) Analysis type* Analysis Class(number sampels) 1 2 3 4 Correlation (up/down)#
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/55957 holds various files of this Leiden University dissertation Author: Dekker T.J.A. Title: Optimizing breast cancer survival models based on conventional
Cover Page The handle http://hdl.handle.net/1887/55957 holds various files of this Leiden University dissertation Author: Dekker T.J.A. Title: Optimizing breast cancer survival models based on conventional
Post Neoadjuvant therapy: issues in interpretation
 Post Neoadjuvant therapy: issues in interpretation Disclosure: Overview D Prognostic features in assessment of post treatment specimens: Tumor size Cellularity Grade Receptors LN Neoadjuvant chemotherapy:
Post Neoadjuvant therapy: issues in interpretation Disclosure: Overview D Prognostic features in assessment of post treatment specimens: Tumor size Cellularity Grade Receptors LN Neoadjuvant chemotherapy:
New Biological and Immunological Therapies for Cancer
 New Biological and Immunological Therapies for Cancer Sant P. Chawla, M.D., FRACP The Sarcoma Oncology Center, Santa Monica CA 90403 7 th International Conference on Drug Discovery &Therapy 1 Promising
New Biological and Immunological Therapies for Cancer Sant P. Chawla, M.D., FRACP The Sarcoma Oncology Center, Santa Monica CA 90403 7 th International Conference on Drug Discovery &Therapy 1 Promising
number Done by Corrected by Doctor Maha Shomaf
 number 21 Done by Ahmad Rawajbeh Corrected by Omar Sami Doctor Maha Shomaf Ability to Invade and Metastasize The metastatic cascade can be subdivided into two phases: 1-invasion of ECM and vascular dissemination:
number 21 Done by Ahmad Rawajbeh Corrected by Omar Sami Doctor Maha Shomaf Ability to Invade and Metastasize The metastatic cascade can be subdivided into two phases: 1-invasion of ECM and vascular dissemination:
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Vascular Endothelial Growth Factor in Human Colon Cancer: Biology and Therapeutic Implications
 Vascular Endothelial Growth Factor in Human Colon Cancer: iology and Therapeutic Implications LEE M. ELLIS, YUTK TKHSHI, WENIO LIU, RYMOND M. SHHEEN University of Texas M.D. nderson Cancer Center, Departments
Vascular Endothelial Growth Factor in Human Colon Cancer: iology and Therapeutic Implications LEE M. ELLIS, YUTK TKHSHI, WENIO LIU, RYMOND M. SHHEEN University of Texas M.D. nderson Cancer Center, Departments
METRIC Study Key Eligibility Criteria
 The METRIC Study METRIC Study Key Eligibility Criteria The pivotal METRIC Study is evaluating glembatumumab vedotin in patients with gpnmb overexpressing metastatic triple-negative breast cancer (TNBC).
The METRIC Study METRIC Study Key Eligibility Criteria The pivotal METRIC Study is evaluating glembatumumab vedotin in patients with gpnmb overexpressing metastatic triple-negative breast cancer (TNBC).
CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow
 White Paper September 2016 CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow Lily C. Trajman, PhD Introduction: Hematopoietic Stem Cells (HSCs)
White Paper September 2016 CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow Lily C. Trajman, PhD Introduction: Hematopoietic Stem Cells (HSCs)
Oncotype DX testing in node-positive disease
 Should gene array assays be routinely used in node positive disease? Yes Christy A. Russell, MD University of Southern California Oncotype DX testing in node-positive disease 1 Validity of the Oncotype
Should gene array assays be routinely used in node positive disease? Yes Christy A. Russell, MD University of Southern California Oncotype DX testing in node-positive disease 1 Validity of the Oncotype
Chapter 7 Conclusions
 VII-1 Chapter 7 Conclusions VII-2 The development of cell-based therapies ranging from well-established practices such as bone marrow transplant to next-generation strategies such as adoptive T-cell therapy
VII-1 Chapter 7 Conclusions VII-2 The development of cell-based therapies ranging from well-established practices such as bone marrow transplant to next-generation strategies such as adoptive T-cell therapy
Melatonin and its Role in the Inhibition of Breast Cancer Ciara Nicol Ross Copyright 2014 by Ciara Ross and Koni Stone
 1 Melatonin and its Role in the Inhibition of Breast Cancer Ciara Nicol Ross Copyright 2014 by Ciara Ross and Koni Stone Cancer is a disease caused by out of control division of abnormal cells within a
1 Melatonin and its Role in the Inhibition of Breast Cancer Ciara Nicol Ross Copyright 2014 by Ciara Ross and Koni Stone Cancer is a disease caused by out of control division of abnormal cells within a
CRYOSENSITIZERS: Ablation at the Edge
 CRYOSENSITIZERS: Ablation at the Edge John G. Baust Institute of Biomedical Technology State University of New York Binghamton, NY USA JGBaust@binghamton.edu Precision targeting Challenge Uncertain margin
CRYOSENSITIZERS: Ablation at the Edge John G. Baust Institute of Biomedical Technology State University of New York Binghamton, NY USA JGBaust@binghamton.edu Precision targeting Challenge Uncertain margin
CANCER THERAPEUTICS: A NOVEL APPROACH
 CANCER THERAPEUTICS: A NOVEL APPROACH Mary Dwyer, Ph.D. HBRI and ChemRegen, Inc. SCDMDG Meeting October 23, 212 Outline Introduction Hit, HBRI1: identification & characterization Leads, HBRI2 & HBRI3:
CANCER THERAPEUTICS: A NOVEL APPROACH Mary Dwyer, Ph.D. HBRI and ChemRegen, Inc. SCDMDG Meeting October 23, 212 Outline Introduction Hit, HBRI1: identification & characterization Leads, HBRI2 & HBRI3:
Design, Synthesis, and Evaluation of In Vivo Potency and Selectivity of Epoxysuccinyl-Based Inhibitors of Papain-Family Cysteine Proteases
 Article Design, Synthesis, and Evaluation of In Vivo Potency and Selectivity of Epoxysuccinyl-Based Inhibitors of Papain-Family Cysteine Proteases Amir Masoud Sadaghiani, 1 Steven H.L. Verhelst, 1 Vasilena
Article Design, Synthesis, and Evaluation of In Vivo Potency and Selectivity of Epoxysuccinyl-Based Inhibitors of Papain-Family Cysteine Proteases Amir Masoud Sadaghiani, 1 Steven H.L. Verhelst, 1 Vasilena
Immunotherapy Concept Turned Reality
 Authored by: Jennifer Dolan Fox, PhD VirtualScopics Inc. jennifer_fox@virtualscopics.com +1 585 249 6231 Immunotherapy Concept Turned Reality Introduction While using the body s own immune system as a
Authored by: Jennifer Dolan Fox, PhD VirtualScopics Inc. jennifer_fox@virtualscopics.com +1 585 249 6231 Immunotherapy Concept Turned Reality Introduction While using the body s own immune system as a
Marshall T Bell Research Resident University of Colorado Grand Rounds Nov. 21, 2011
 Marshall T Bell Research Resident University of Colorado Grand Rounds Nov. 21, 2011 Most common form of cancer in adults ages 25-29 3-5% of skin cancers but 65-75% of deaths Most common metastasis to small
Marshall T Bell Research Resident University of Colorado Grand Rounds Nov. 21, 2011 Most common form of cancer in adults ages 25-29 3-5% of skin cancers but 65-75% of deaths Most common metastasis to small
1. The metastatic cascade. 3. Pathologic features of metastasis. 4. Therapeutic ramifications. Which malignant cells will metastasize?
 1. The metastatic cascade 3. Pathologic features of metastasis 4. Therapeutic ramifications Sir James Paget (1814-1899) British Surgeon/ Pathologist Paget s disease of Paget s disease of the nipple (intraductal
1. The metastatic cascade 3. Pathologic features of metastasis 4. Therapeutic ramifications Sir James Paget (1814-1899) British Surgeon/ Pathologist Paget s disease of Paget s disease of the nipple (intraductal
Environmental and genetic changes are associated with carcinogenesis. Since many cancers exhibit a 'field effect', many
 Name: Score: / Quiz 10 on Cancer Biology Part 1 Environmental and genetic changes are associated with carcinogenesis. Since many cancers exhibit a 'field effect', many normal-appearing cells in a target
Name: Score: / Quiz 10 on Cancer Biology Part 1 Environmental and genetic changes are associated with carcinogenesis. Since many cancers exhibit a 'field effect', many normal-appearing cells in a target
Cathepsin S Inhibitors for the Treatment of Inflammatory Bowel Disease. Therapeutic Area Partnerships Presentation.
 Inhibitors for the Treatment of Inflammatory Bowel Disease Therapeutic Area Partnerships Presentation November 29, 2012 Cathepsin Cysteine Proteinases Fibrosis Cathepsin O Cathepsin B Cathepsin C Cathepsin
Inhibitors for the Treatment of Inflammatory Bowel Disease Therapeutic Area Partnerships Presentation November 29, 2012 Cathepsin Cysteine Proteinases Fibrosis Cathepsin O Cathepsin B Cathepsin C Cathepsin
IMMUNOHISTOCHEMICAL EXPRESSION OF TISSUE INHIBITOR OF METALLOPROTEINASE-1 (TIMP-1) IN INVASIVE BREAST CARCINOMA
 & IMMUNOHISTOCHEMICAL EXPRESSION OF TISSUE INHIBITOR OF METALLOPROTEINASE-1 (TIMP-1) IN INVASIVE BREAST CARCINOMA Suada Kuskunović*, Svjetlana Radović, Mirsad Dorić, Ajna Hukić, Mirsad Babić, Ivana Tomić,
& IMMUNOHISTOCHEMICAL EXPRESSION OF TISSUE INHIBITOR OF METALLOPROTEINASE-1 (TIMP-1) IN INVASIVE BREAST CARCINOMA Suada Kuskunović*, Svjetlana Radović, Mirsad Dorić, Ajna Hukić, Mirsad Babić, Ivana Tomić,
Cabozantinib for medullary thyroid cancer. February 2012
 Cabozantinib for medullary thyroid cancer February 2012 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive
Cabozantinib for medullary thyroid cancer February 2012 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive
Supplemental Material
 Supplemental Material Supplementary Fig. 1. EETs stimulate primary tumor growth. a) Schematic presentation of genetic and pharmacological tools used to manipulate endogenous EET levels. b) Endothelial
Supplemental Material Supplementary Fig. 1. EETs stimulate primary tumor growth. a) Schematic presentation of genetic and pharmacological tools used to manipulate endogenous EET levels. b) Endothelial
Convergent and Divergent Mechanisms in Aging and Cancer
 Convergent and Divergent Mechanisms in Aging and Cancer Mariana S. De Lorenzo, PhD Department of Cell Biology & Molecular Medicine delorems@umdnj.edu LEARNING OBJECTIVES 1. To identify convergent and divergent
Convergent and Divergent Mechanisms in Aging and Cancer Mariana S. De Lorenzo, PhD Department of Cell Biology & Molecular Medicine delorems@umdnj.edu LEARNING OBJECTIVES 1. To identify convergent and divergent
Breast cancer as a systemic disease: a view of metastasis
 Review Click here for more articles from the symposium doi: 10.1111/joim.12084 Breast cancer as a systemic disease: a view of metastasis A. J. Redig 1 & S. S. McAllister 1,2,3 From the 1 Division of Hematology,
Review Click here for more articles from the symposium doi: 10.1111/joim.12084 Breast cancer as a systemic disease: a view of metastasis A. J. Redig 1 & S. S. McAllister 1,2,3 From the 1 Division of Hematology,
Corporate Medical Policy
 Corporate Medical Policy Pertuzumab for Treatment of Malignancies File Name: Origination: Last CAP Review: Next CAP Review: Last Review: pertuzumab_for_treatment_of_malignancies 2/2013 4/2017 4/2018 6/2017
Corporate Medical Policy Pertuzumab for Treatment of Malignancies File Name: Origination: Last CAP Review: Next CAP Review: Last Review: pertuzumab_for_treatment_of_malignancies 2/2013 4/2017 4/2018 6/2017
Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors
 Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors See the related Commentary beginning on page 1277. Gabriele Bergers, 1,2 Steven Song, 1,2 Nicole
Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors See the related Commentary beginning on page 1277. Gabriele Bergers, 1,2 Steven Song, 1,2 Nicole
DEVELOPMENT OF CELLULAR IMMUNOLOGY
 DEVELOPMENT OF CELLULAR IMMUNOLOGY 1880 s: Antibodies described (dominated studies of immunology until 1960 s) 1958: Journal of Immunology (137 papers) lymphocyte not listed in index Two papers on transfer
DEVELOPMENT OF CELLULAR IMMUNOLOGY 1880 s: Antibodies described (dominated studies of immunology until 1960 s) 1958: Journal of Immunology (137 papers) lymphocyte not listed in index Two papers on transfer
Author s response to reviews
 Author s response to reviews Title: Case Report of an uncommon response to metronomic therapy in a heavily pre treated patient with Metastatic Carcinosarcoma Authors: Debora Gagliato (dgagliato@gmail.com)
Author s response to reviews Title: Case Report of an uncommon response to metronomic therapy in a heavily pre treated patient with Metastatic Carcinosarcoma Authors: Debora Gagliato (dgagliato@gmail.com)
CANCER IMMUNOPATHOLOGY. Eryati Darwin Faculty of Medicine Andalas University
 CANCER IMMUNOPATHOLOGY Eryati Darwin Faculty of Medicine Andalas University Padang 18 Mei 2013 INTRODUCTION Tumor: cells that continue to replicate, fail to differentiate into specialized cells, and become
CANCER IMMUNOPATHOLOGY Eryati Darwin Faculty of Medicine Andalas University Padang 18 Mei 2013 INTRODUCTION Tumor: cells that continue to replicate, fail to differentiate into specialized cells, and become
complemented with SipA ( SipA/pSipA) or SL1344 WT for 48 hours, after which the
 P-gp expression (% of control) 12 1 8 6 4 2 * * Untreated SipA SipA/pSipA WT Supplementary Figure 1. SipA modulates the expression of P-gp in healthy murine intestinal epithelium in vivo. Salmonella Typhimurium
P-gp expression (% of control) 12 1 8 6 4 2 * * Untreated SipA SipA/pSipA WT Supplementary Figure 1. SipA modulates the expression of P-gp in healthy murine intestinal epithelium in vivo. Salmonella Typhimurium
Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis
 Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis Johanna A. Joyce, 1,2 Amos Baruch, 1,4 Kareem Chehade, 1,4 Nicole Meyer-Morse, 1,2 Enrico
Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis Johanna A. Joyce, 1,2 Amos Baruch, 1,4 Kareem Chehade, 1,4 Nicole Meyer-Morse, 1,2 Enrico
Deregulation of signal transduction and cell cycle in Cancer
 Deregulation of signal transduction and cell cycle in Cancer Tuangporn Suthiphongchai, Ph.D. Department of Biochemistry Faculty of Science, Mahidol University Email: tuangporn.sut@mahidol.ac.th Room Pr324
Deregulation of signal transduction and cell cycle in Cancer Tuangporn Suthiphongchai, Ph.D. Department of Biochemistry Faculty of Science, Mahidol University Email: tuangporn.sut@mahidol.ac.th Room Pr324
Oncolytic Viruses: Reovirus
 T S X : O N C N A S D A Q : O N C Y International Society for Biological Therapy of Cancer 2008 Oncology Biologics Development Primer Oncolytic Viruses: Reovirus REOLYSIN - mode of action REOLYSIN contains
T S X : O N C N A S D A Q : O N C Y International Society for Biological Therapy of Cancer 2008 Oncology Biologics Development Primer Oncolytic Viruses: Reovirus REOLYSIN - mode of action REOLYSIN contains
