Beads-on-a- The 30nm Fibre Active Chromosome The Metaphase Chromosome. Less active genes During interphase During cell division
|
|
|
- Archibald Allison
- 5 years ago
- Views:
Transcription
1 Overview of Epigenetics Figure 2.2. The Increasing Structural Complexity of Genetic Information from the Double-Helical Structure of DNA, through Nucleosome and Chromatin Structures, to the Chromosome DNA The Beads-on-a- Nucleosome String The 30nm Fibre Active Chromosome The Metaphase Chromosome Isolated Patches Genes under active transcription Less active genes During interphase During cell division Add core histones Add histone H1 Add further scaffold proteins Add further scaffold proteins Source: Histones are small, alkaline proteins that are associated with the DNA. The major types of histones are referred to as H1, H2A, H2B, H3, and H4, although additional types of histones exist. Portions of DNA wrap around the histones to form nucleosomes, which resemble a string of beads, as shown in Figure 2.2. Nucleosomes are the basic structural unit of chromatin, which is the complex between DNA and proteins in eukaryotic cells. As demonstrated in Figure 2.2, the extent of condensation of chromatin varies with different stages of the cell lifecycle. In nondividing (interphase) cells, most of the chromatin is euchromatin, which is relaxed chromatin that is not tightly condensed. However, some of the chromatin is heterochromatin, which is highly condensed. Genes in the heterochromatin cannot be transcribed. The N-terminal domain of histones can be modified by various enzymes. These modifications can include acetylation, methylation, ubiquitylation, or phosphorylation, and modifications of the histones may either activate or repress transcription of the genes. Figure 2.3 shows the nucleosome, with the N-terminal region of the histone, and the many different types of post-translational modifications that can occur. Figure 2.3. Schematic of the Nucleosome Showing Types of Post-Translational Modifications That Can Occur and the Enzymes Responsible for Them Acetyltransferases Deacetylases Ac Kinases P Ligases Ub Histone Me Me PKMTs Demethylases PRMTs Ac, acetylation; P, phosphorylation; Ub, ubiquitylation; Me, methylation; PKMT, protein lysine methyltransferase; PRMT, protein arginine methyltransferase Source: Insight Pharma Reports Nucleosome 8 Reproduction prohibited
2 Diagnostic Companies in Epigenetics Table 3.1. Companies Developing and/or Offering Epigenetic-Based Diagnostic Tests Company Product/Technology Status Comments Real-time PCR assay that detects the presence of methylated Septin9 in plasma For colorectal cancer screening Abbott Molecular Abbott RealTime ms9 Colorectal Cancer assay Available in Europe and Asia/Pacific 9/07: Epigenomics announced a nonexclusive strategic collaboration and license agreement with Abbott for development of an in vitro diagnostic test for early detection of colorectal cancer based on the Septin9 biomarker. 10/05: Rubicon Genomics announced a multiyear Research and Option Agreement with Abbott Molecular to develop and commercialize Rubicon s MethylPlex methylation detection technology for diagnosis and prognosis of cancer. Abbott Molecular Cancer tests based on Rubicon s MethylPlex methylation detection technology In development 1/09: Rubicon Genomics announced an agreement granting Abbott a nonexclusive license to Rubicon s MethylPlex technology for in vitro molecular diagnostics to detect the presence or risk of prostate, bladder, and colon cancer for screening and non-screening indications. Abbott also obtained a non-exclusive license for non-screening applications with other diseases and options to colon cancer markers developed in collaboration with Rubicon. Continued 20 Reproduction prohibited
3 Epigenetic Drugs On The Market And In Development Both FDA-approved DNA methyltransferase inhibitors are indicated for treatment of myelodysplastic syndrome (MDS). The numbers of people with MDS are not large (see Chapter 4). Thus, the clinical trials required for their approval were not large in terms of numbers of patients. Also, before approval of these agents, there were no approved treatment options for MDS and so the clinical trials were designed to compare the DNA methylation inhibitor plus best supportive care versus best supportive care alone. While the response rates with Vidaza or Dacogen were not high, there were no responses to best supportive care. Thus, treatment with a DNA methylation inhibitor was shown to be clearly better than no treatment (at least for those patients who responded to the therapy). However, these agents can cause a number of different adverse reactions. Selected data (response rates and most common adverse effects) from the clinical studies are discussed below. Additional data from these clinical studies can be found in the prescribing information for each drug. While these indirect DNA methylation inhibitors represent an important improvement in the care of patients with MDS, there is a clear need for better therapies. Researchers have been interested in the development of novel DNA methyltransferase inhibitors that can directly inhibit the enzyme (without prior incorporation into the cell s DNA), but identifying compounds that could be successfully commercialized as therapies has proven to be challenging. At this time, no direct inhibitors of DNA methyltransferase appear to be in clinical development. While one company (SuperGen) is developing a novel DNA methyltransferase inhibitor, that new agent (SGT-110) is a prodrug of decitabine, one of the two DNA methyltransferase inhibitors that is already FDA-approved. Table 5.1. Companies Marketing and Developing DNA Methylation Inhibitors Company Product/Technology Status Comments Celgene Vidaza (azacitidine) On the market DNA hypomethylation agent. It is incorporated into DNA and then inhibits DNA methylation. FDA-approved for treatment of myelodysplastic syndrome. Also approved in Europe. Eisai Dacogen (decitabine) On the market (United States) DNA hypomethylation agent. It is incorporated into DNA and then inhibits DNA methylation. FDA-approved for treatment of myelodysplastic syndrome. Continued 50 Reproduction prohibited
4 Epigenetic Drugs On The Market And In Development sales milestones, could exceed $350 million. In addition, Spectrum Pharmaceuticals will pay TopoTarget royalties if belinostat is successfully commercialized and sold by Spectrum Pharmaceuticals in the regions covered by the agreement. Spectrum Pharmaceuticals was granted the right to market belinostat in North America and India, and was granted a right of first offer for China. Belinostat is currently being evaluated in a registration clinical trial in patients with peripheral T-cell lymphoma (PTCL). This trial is being conducted under a Special Protocol Assessment (SPA), and belinostat has been granted both Orphan Drug and Fast Track designation by the FDA. In addition, belinostat is in a randomized Phase II study in combination with carboplatin and paclitaxel for treatment of patients with cancer of unknown origin (CUP). As part of the agreement between TopoTarget and Spectrum Pharmaceuticals, Spectrum Pharmaceuticals agreed to fund 100% of the costs for the PTCL study, and TopoTarget is funding 100% of the costs of the ongoing CUP clinical trial. In the future, clinical trial costs will be shared 70/30, with Spectrum Pharmaceuticals paying 70%. Belinostat is currently being evaluated in additional earlier-stage trials in other cancers, which are being sponsored by Spectrum Pharmaceuticals. Spectrum Pharmaceuticals also has an ongoing collaboration with the National Cancer Institute (NCI), which is conducting a number of clinical studies to evaluate belinostat. The February 2010 announcement of the agreement between TopoTarget and Spectrum Pharmaceuticals also reported that belinostat was being evaluated in 20 clinical trials in hematological and solid tumors, both in monotherapy and combination therapy regimens. Additional information about TopoTarget and the HDAC inhibitor belinostat can be found in the interview with Henri Lichenstein, PhD, TopoTarget s chief development officer (see Chapter 7). In this interview, he reports that one of belinostat s differentiating characteristics is its safety profile; they have not seen the hematological and cardiac toxicity that have been seen with other HDAC inhibitors. In addition, in May 2010, TopoTarget and Spectrum Pharmaceuticals both announced that abstracts on belinostat would be presented at the upcoming American Society of Clinical Oncology (ASCO) meeting. This includes an abstract on the ongoing trial of belinostat in patients with PTCL. As of January 4, 2010, 19 patients had been included in this clinical trial Emerging Therapies Directed Against Other Epigenetic Targets As discussed in the previous section, much of the current drug discovery and development activity in the field of epigenetics is focused on the class of drugs known as HDAC inhibitors. While companies in this field are working to differentiate their HDAC inhibitors from other emerging HDAC inhibitors, this is already a crowded area. Given this high level of competition, combined with the fact that many different enzymes are involved in epigenetics and thus are potential targets, some companies are already starting to develop potential therapies that have other (non-hdac) mechanisms of action. Examples are discussed below and in Table Reproduction prohibited
5 Epigenetic Drug & Diagnostic Pipelines: DNA Methylation, HDAC Inhibitors, and Emerging New Targets Table 6.1. Reported 2009 Sales of Approved Epigenetic Drugs Company Drug Sales/Comments DNA Methylation Inhibitors Celgene Vidaza (azacitidine) $387 million $150 million in the United States Eisai Dacogen (decitabine) 15 billion yen (total Eisai Dacogen sales) Johnson & Johnson Dacogen (decitabine) Dacogen s sales not reported HDAC Inhibitors Celgene Istodax (romidepsin) None (FDA-approved in late 2009; market launch in 2010) Merck Zolinza (vorinostat) Zolinza s sales not reported Source: Publicly reported figures Interest in the epigenetic market is being fueled by the large potential markets for these therapies. Potential patient populations for emerging new epigenetic therapies are discussed in Chapter 4. While these indications and the emerging therapies targeting these indications are promising, most of the epigenetic product candidates are in relatively early development. Of the agents included in Chapter 5 that are not yet on the market, only two have reached Phase III or registration clinical trials. Novartis panobinostat (LBH589) is in Phase III in patients with relapsed multiple myeloma, and a Phase III trial in Hodgkin s lymphoma is planned. In addition, TopoTarget s belinostat is in a registration clinical trial in patients with peripheral T-cell lymphoma (PTCL), which is another indication with a very small market. Belinostat is also being evaluated in a randomized Phase II study in combination with carboplatin and paclitaxel for treatment of patients with cancer of unknown origin (CUP). Thus, it is likely be several years before the combined market for epigenetic-based therapies is large Major Challenges/Hurdles and Strategic Issues for Diagnostic Companies in Epigenetics Research activity has exploded in the field of epigenetics, as demonstrated by the number of publications (see Figure 1.1 in Chapter 1). These ongoing research activities have resulted in significant advances in our knowledge of epigenetics. The first diagnostic tests have been CE-marked and are available in Europe, and initial tests are being offered as laboratory-developed tests by reference laboratories in the United States. Four epigenetic drugs have been approved by the FDA, including two agents that inhibit DNA methylation and two HDAC inhibitors. Reproduction prohibited 81
6 Business Considerations in Epigenetics Agreements in the Epigenetic Therapy Market; Companies Entering and Exiting the Market The epigenetic-based therapy market has evolved in recent years. Over 40 companies appear to be actively developing and/or commercializing epigenetic-based therapies. (See Chapter 5 and Tables ) Therapeutic companies in this field range from relatively new, venture capital-funded startups to large, multinational pharmaceutical companies, and include companies throughout this range (in terms of size and age of the companies). This has been a dynamic market with many corporate agreements (see Table 6.2). Some major pharmaceutical companies that were developing an epigenetic drug appear to have exited this market (Table 6.3). However, it is possible that one or more of these companies may decide to reenter the epigenetic therapy field. For example, in May 2009, AstraZeneca licensed its HDAC inhibitor AZD9468 to Celleron Therapeutics, which now calls this compound CDX101. As part of this agreement, AstraZeneca was granted the option to discuss reacquisition of commercialization rights in the future. Table 6.2. Selected Epigenetic Therapeutic Agreements Epigenetic Company Partner Company, or Company Out-Licensing Technology and Exiting Date (Or Date Announced) Subject of Agreement 4SC Nycomed July 2008 Announcement that 4SC had completed the transaction to acquire eight oncology programs from Nycomed. These programs included the HDAC inhibitors resminostat (4SC-201) and 4SC-202, plus two other HDAC inhibitor programs. [Nycomed is also included in Table 6.3.] Aphios VivaCell Biotechnology España March 2009 Announcement of collaborative research and development agreement between Aphios and VivaCell Biotechnology España to develop a combination therapy for treatment of HIV latency. This combination therapy consists of bryostatins (PKC modulators) and HDAC inhibitors. CellCentric Takeda Pharmaceutical February 2010 CellCentric announced that it had out-licensed one of its cancer programs to Takeda Pharmaceutical. This is an exclusive agreement that could potentially be worth more than $200 million to CellCentric over its course. Celleron Therapeutics AstraZeneca May 2009 Announcement that Celleron licensed the HDAC inhibitor AZD9468 from AstraZeneca. Celleron now calls it CDX101. AstraZeneca was granted the option to discuss reacquisition of commercialization rights in the future. [Astra- Zeneca is also included in Table 6.3.] Continued 92 Reproduction prohibited
7 Chapter 7 Expert Interviews 7.1. Walter C. Ogier President and Chief Executive Officer Acetylon Pharmaceuticals Insight Pharma Reports (IPR): Please provide a brief history of Acetylon Pharmaceuticals. Walter C. Ogier: While Acetylon is a relatively new company, founded in late 2008, we are the outgrowth of work that goes back six or more years within the confines of Harvard University and the Dana-Farber Cancer Institute. Harvard was the first to discover the existence of histone deacetylases (HDACs) and also identified the first half dozen or so of the human HDACs. There are now a total of 18 known HDACs, seven of which comprise the sirtuin family. Sirtris, which was taking these forward as drug opportunities, was acquired by GlaxoSmithKline in Sirtuins are NAD-dependent enzymes. They have a biological function that is distinct from the other 11 human HDACs that I will be talking about, which employ zinc atoms in their catalytic active site. But, strictly speaking, the sirtuins are part of the overall HDAC family. There have been some recent publications that cast doubt on the potential clinical utility of these enzymes and enzyme activators, but for several years Sirtris was a high-flyer (GlaxoSmithKline paid $720 million for the company). The research that has, at least in part, cast doubt on the potential utility of this subset of HDAC enzymes as drug targets was published by Pfizer. The sirtuins have generally been billed as potential drugs for treatment of diseases of aging and metabolic dysregulation, including type 2 diabetes and other disease indications which do not overlap substantially with the potential clinical utility of the 11 zinc-containing HDACs as drug targets. The portion of the HDAC family that Acetylon is interested in comprises the other 11 human HDACs. The first selective HDAC inhibitor was created by Stuart Schreiber and colleagues at Harvard University and was made widely available in academic circles. It was a compound called tubacin which selectively inhibits Reproduction prohibited 97
Epigenetics q&more 01.11
 Laurie. Knight, istockphoto.com Epigenetics 6 Bookmarks About the reading of genes in the Book of Life Prof. Dr. Manfred Jung, Julia M. Wagner, Institute for Pharmaceutical Sciences, Albert-Ludwig-University
Laurie. Knight, istockphoto.com Epigenetics 6 Bookmarks About the reading of genes in the Book of Life Prof. Dr. Manfred Jung, Julia M. Wagner, Institute for Pharmaceutical Sciences, Albert-Ludwig-University
Two-stage study designed to evaluate tolerability and efficacy of pracinostat combined with azacitidine in patients with high and very high risk MDS
 Helsinn Group and MEI Pharma Announce First Patient Dosed in Phase 2 Dose-Optimization Study of Pracinostat and Azacitidine in Myelodysplastic Syndrome Two-stage study designed to evaluate tolerability
Helsinn Group and MEI Pharma Announce First Patient Dosed in Phase 2 Dose-Optimization Study of Pracinostat and Azacitidine in Myelodysplastic Syndrome Two-stage study designed to evaluate tolerability
Epigenetics. Lyle Armstrong. UJ Taylor & Francis Group. f'ci Garland Science NEW YORK AND LONDON
 ... Epigenetics Lyle Armstrong f'ci Garland Science UJ Taylor & Francis Group NEW YORK AND LONDON Contents CHAPTER 1 INTRODUCTION TO 3.2 CHROMATIN ARCHITECTURE 21 THE STUDY OF EPIGENETICS 1.1 THE CORE
... Epigenetics Lyle Armstrong f'ci Garland Science UJ Taylor & Francis Group NEW YORK AND LONDON Contents CHAPTER 1 INTRODUCTION TO 3.2 CHROMATIN ARCHITECTURE 21 THE STUDY OF EPIGENETICS 1.1 THE CORE
Expert Intelligence for Better Decisions Epigenetics:
 Expert Intelligence for Better Decisions Epigenetics: Emerging Targets, Available Technologies, Expert Interviews, and an Epigenetic Community Perspective Using This Document Insight Pharma Reports are
Expert Intelligence for Better Decisions Epigenetics: Emerging Targets, Available Technologies, Expert Interviews, and an Epigenetic Community Perspective Using This Document Insight Pharma Reports are
Eukaryotic transcription (III)
 Eukaryotic transcription (III) 1. Chromosome and chromatin structure Chromatin, chromatid, and chromosome chromatin Genomes exist as chromatins before or after cell division (interphase) but as chromatids
Eukaryotic transcription (III) 1. Chromosome and chromatin structure Chromatin, chromatid, and chromosome chromatin Genomes exist as chromatins before or after cell division (interphase) but as chromatids
Sumitomo Dainippon Pharma to Acquire Tolero Pharmaceuticals, Inc. (US Biotechnology Company)
 December 21, 2016 Sumitomo Dainippon Pharma Co., Ltd. Sumitomo Dainippon Pharma to Acquire Tolero Pharmaceuticals, Inc. (US Biotechnology Company) Sumitomo Dainippon Pharma Co., Ltd. (Head office: Osaka,
December 21, 2016 Sumitomo Dainippon Pharma Co., Ltd. Sumitomo Dainippon Pharma to Acquire Tolero Pharmaceuticals, Inc. (US Biotechnology Company) Sumitomo Dainippon Pharma Co., Ltd. (Head office: Osaka,
Topotarget A/S. Corporate Presentation November 2011
 Topotarget A/S Corporate Presentation November 2011 Safe harbour statement This presentation may contain forward-looking statements, including statements about our expectations of the progression of our
Topotarget A/S Corporate Presentation November 2011 Safe harbour statement This presentation may contain forward-looking statements, including statements about our expectations of the progression of our
Oncology Pipeline Analytics
 Oncology Pipeline Analytics Trials, Drug Classes, Indications, Correlations and Strategies Report Brochure O n c o l o g y P i p e l i n e A n a l y t i c s Greystone Research Associates is pleased to
Oncology Pipeline Analytics Trials, Drug Classes, Indications, Correlations and Strategies Report Brochure O n c o l o g y P i p e l i n e A n a l y t i c s Greystone Research Associates is pleased to
Gene Expression DNA RNA. Protein. Metabolites, stress, environment
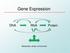 Gene Expression DNA RNA Protein Metabolites, stress, environment 1 EPIGENETICS The study of alterations in gene function that cannot be explained by changes in DNA sequence. Epigenetic gene regulatory
Gene Expression DNA RNA Protein Metabolites, stress, environment 1 EPIGENETICS The study of alterations in gene function that cannot be explained by changes in DNA sequence. Epigenetic gene regulatory
Investor Presentation
 Investor Presentation June 2017 Joe Turgeon President and Chief Operating Officer Safe Harbor Statement This presentation contains forward-looking statements regarding future events and the future performance
Investor Presentation June 2017 Joe Turgeon President and Chief Operating Officer Safe Harbor Statement This presentation contains forward-looking statements regarding future events and the future performance
Transcriptional control in Eukaryotes: (chapter 13 pp276) Chromatin structure affects gene expression. Chromatin Array of nuc
 Transcriptional control in Eukaryotes: (chapter 13 pp276) Chromatin structure affects gene expression Chromatin Array of nuc 1 Transcriptional control in Eukaryotes: Chromatin undergoes structural changes
Transcriptional control in Eukaryotes: (chapter 13 pp276) Chromatin structure affects gene expression Chromatin Array of nuc 1 Transcriptional control in Eukaryotes: Chromatin undergoes structural changes
Epigenetics Armstrong_Prelims.indd 1 04/11/2013 3:28 pm
 Epigenetics Epigenetics Lyle Armstrong vi Online resources Accessible from www.garlandscience.com, the Student and Instructor Resource Websites provide learning and teaching tools created for Epigenetics.
Epigenetics Epigenetics Lyle Armstrong vi Online resources Accessible from www.garlandscience.com, the Student and Instructor Resource Websites provide learning and teaching tools created for Epigenetics.
Eukaryotic Gene Regulation
 Eukaryotic Gene Regulation Chapter 19: Control of Eukaryotic Genome The BIG Questions How are genes turned on & off in eukaryotes? How do cells with the same genes differentiate to perform completely different,
Eukaryotic Gene Regulation Chapter 19: Control of Eukaryotic Genome The BIG Questions How are genes turned on & off in eukaryotes? How do cells with the same genes differentiate to perform completely different,
HALOZYME REPORTS SECOND QUARTER 2018 RESULTS
 Contacts: Robert H. Uhl Managing Director Westwicke Partners, LLC 858-356-5932 robert.uhl@westwicke.com Laurie Stelzer 858-704-8222 ir@halozyme.com FOR IMMEDIATE RELEASE HALOZYME REPORTS SECOND QUARTER
Contacts: Robert H. Uhl Managing Director Westwicke Partners, LLC 858-356-5932 robert.uhl@westwicke.com Laurie Stelzer 858-704-8222 ir@halozyme.com FOR IMMEDIATE RELEASE HALOZYME REPORTS SECOND QUARTER
EPIGENOMICS PROFILING SERVICES
 EPIGENOMICS PROFILING SERVICES Chromatin analysis DNA methylation analysis RNA-seq analysis Diagenode helps you uncover the mysteries of epigenetics PAGE 3 Integrative epigenomics analysis DNA methylation
EPIGENOMICS PROFILING SERVICES Chromatin analysis DNA methylation analysis RNA-seq analysis Diagenode helps you uncover the mysteries of epigenetics PAGE 3 Integrative epigenomics analysis DNA methylation
Infinity Highlights Promising Preclinical Hedgehog Data and Announces First Quarter Financial Results
 Infinity Highlights Promising Preclinical Hedgehog Data and Announces First Quarter Financial Results Heat Shock Protein 90 Program Continues to Advance Toward Potential Registration Trial CAMBRIDGE, Mass.,
Infinity Highlights Promising Preclinical Hedgehog Data and Announces First Quarter Financial Results Heat Shock Protein 90 Program Continues to Advance Toward Potential Registration Trial CAMBRIDGE, Mass.,
DISCOVERING AND DEVELOPING NEW IMMUNE ACTIVATING MECHANISMS AND ANTIBODIES TO IMPROVE CANCER SURVIVAL
 DISCOVERING AND DEVELOPING NEW IMMUNE ACTIVATING MECHANISMS AND ANTIBODIES TO IMPROVE CANCER SURVIVAL Annual General Meeting 24 April 2018 Björn Frendéus, Acting CEO & CSO 1 IMMUNOTHERAPY HAS TRANSFORMED
DISCOVERING AND DEVELOPING NEW IMMUNE ACTIVATING MECHANISMS AND ANTIBODIES TO IMPROVE CANCER SURVIVAL Annual General Meeting 24 April 2018 Björn Frendéus, Acting CEO & CSO 1 IMMUNOTHERAPY HAS TRANSFORMED
Transcription Regulation And Gene Expression in Eukaryotes Cycle G2 (lecture 13709) FS 2014 P Matthias & RG Clerc
 Transcription Regulation And Gene Expression in Eukaryotes Cycle G2 (lecture 13709) FS 2014 P Matthias & RG Clerc P. Matthias, 16 April 2014 DNA methylation basics Acetylation Acetyltransferases/Deacetylases
Transcription Regulation And Gene Expression in Eukaryotes Cycle G2 (lecture 13709) FS 2014 P Matthias & RG Clerc P. Matthias, 16 April 2014 DNA methylation basics Acetylation Acetyltransferases/Deacetylases
AVEO Oncology Announces Strategic Restructuring. AVEO to Host Conference Call Wednesday, June 5 at 8:30 a.m. ET
 NEWS RELEASE FOR IMMEDIATE RELEASE AVEO Oncology Announces Strategic Restructuring AVEO to Host Conference Call Wednesday, June 5 at 8:30 a.m. ET CAMBRIDGE, Mass., June 4, 2013 AVEO Oncology (NASDAQ: AVEO)
NEWS RELEASE FOR IMMEDIATE RELEASE AVEO Oncology Announces Strategic Restructuring AVEO to Host Conference Call Wednesday, June 5 at 8:30 a.m. ET CAMBRIDGE, Mass., June 4, 2013 AVEO Oncology (NASDAQ: AVEO)
Opportunities and Challenges in the Development of Companion Diagnostics
 Opportunities and Challenges in the Development of Companion Diagnostics E. Patrick Groody, Ph.D. Divisional Vice President Research and Development Abbott Molecular Agenda Value of Personalized Medicine
Opportunities and Challenges in the Development of Companion Diagnostics E. Patrick Groody, Ph.D. Divisional Vice President Research and Development Abbott Molecular Agenda Value of Personalized Medicine
Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies selectively targeting genetic drivers of cancer
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies
Case 2:09-cv FSH-PS Document 1-3 Filed 08/24/2009 Page 1 of 5. Exhibit B
 Case 2:09-cv-04329-FSH-PS Document 1-3 Filed 08/24/2009 Page 1 of 5 Exhibit B Case 2:09-cv-04329-FSH-PS Document 1-3 Filed 08/24/2009 Page 2 of 5 TO OUR FELLOW SHAREHOLDERS: In 2001 we made significant
Case 2:09-cv-04329-FSH-PS Document 1-3 Filed 08/24/2009 Page 1 of 5 Exhibit B Case 2:09-cv-04329-FSH-PS Document 1-3 Filed 08/24/2009 Page 2 of 5 TO OUR FELLOW SHAREHOLDERS: In 2001 we made significant
Developing & Commercializing Targeted Small Molecule Drugs in Cancer
 Developing & Commercializing Targeted Small Molecule Drugs in Cancer SAFE HARBOR STATEMENT 2 Forward-looking statements made in the course of this presentation are made pursuant to the safe harbor provisions
Developing & Commercializing Targeted Small Molecule Drugs in Cancer SAFE HARBOR STATEMENT 2 Forward-looking statements made in the course of this presentation are made pursuant to the safe harbor provisions
Dr Marta Boffito 15/10/ th Annual Resistance and Antiviral Therapy Meeting. Chelsea and Westminster Hospital, London
 18 th Annual Resistance and Antiviral Therapy Meeting Dr Marta Boffito Chelsea and Westminster Hospital, London Thursday 18 September 2014, Royal College of Physicians, London 1 virological response (SVR)
18 th Annual Resistance and Antiviral Therapy Meeting Dr Marta Boffito Chelsea and Westminster Hospital, London Thursday 18 September 2014, Royal College of Physicians, London 1 virological response (SVR)
Safe Harbor Statement
 September 2007 Safe Harbor Statement During the course of this presentation we will make forward-looking statements that involve risks and uncertainties associated with a developing pharmaceutical company.
September 2007 Safe Harbor Statement During the course of this presentation we will make forward-looking statements that involve risks and uncertainties associated with a developing pharmaceutical company.
37 th ANNUAL JP MORGAN HEALTHCARE CONFERENCE
 37 th ANNUAL JP MORGAN HEALTHCARE CONFERENCE January 2019 Safe Harbor Statement This presentation contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as
37 th ANNUAL JP MORGAN HEALTHCARE CONFERENCE January 2019 Safe Harbor Statement This presentation contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as
[ NASDAQ: MEIP ] August 2014
![[ NASDAQ: MEIP ] August 2014 [ NASDAQ: MEIP ] August 2014](/thumbs/86/94600707.jpg) [ NASDAQ: MEIP ] August 2014 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events or results may differ materially from those
[ NASDAQ: MEIP ] August 2014 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events or results may differ materially from those
Dynavax Corporate Presentation
 Dynavax Corporate Presentation Forward-Looking Statements This presentation contains forward-looking statements, including statements regarding our HEPLISAV-B TM regulatory submissions, product profile,
Dynavax Corporate Presentation Forward-Looking Statements This presentation contains forward-looking statements, including statements regarding our HEPLISAV-B TM regulatory submissions, product profile,
Oncology Market Forecast To 2013
 Oncology Market Forecast To 2013 How do you uncover the threats and opportunities that your oncology drug will face this year, next year and each year after that? Our new report answers your questions
Oncology Market Forecast To 2013 How do you uncover the threats and opportunities that your oncology drug will face this year, next year and each year after that? Our new report answers your questions
Third Quarter 2018 Financial Results. November 1, 2018
 Third Quarter 2018 Financial Results November 1, 2018 Agios Conference Call Participants Prepared Remarks Introduction RENEE LECK, Associate Director, Investor Relations Business Highlights & 2018 Key
Third Quarter 2018 Financial Results November 1, 2018 Agios Conference Call Participants Prepared Remarks Introduction RENEE LECK, Associate Director, Investor Relations Business Highlights & 2018 Key
Idenix Pharmaceuticals Building a Leading Antiviral Franchise. Cowen & Company 27 th Annual Healthcare Conference March 13, 2007 Boston
 Idenix Pharmaceuticals Building a Leading Antiviral Franchise Cowen & Company 27 th Annual Healthcare Conference March 13, 2007 Boston Safe Harbor This presentation includes forward-looking statements
Idenix Pharmaceuticals Building a Leading Antiviral Franchise Cowen & Company 27 th Annual Healthcare Conference March 13, 2007 Boston Safe Harbor This presentation includes forward-looking statements
Disrupting the Cell Cycle to Treat AML and MDS Rodman & Renshaw Conference
 CYC 682 Disrupting the Cell Cycle to Treat AML and MDS Rodman & Renshaw Conference September 2014 Disclaimer This presentation contains forward-looking statements within the meaning of the safe harbor
CYC 682 Disrupting the Cell Cycle to Treat AML and MDS Rodman & Renshaw Conference September 2014 Disclaimer This presentation contains forward-looking statements within the meaning of the safe harbor
Epigene.cs: What is it and how it effects our health? Overview. Dr. Bill Stanford, PhD OFawa Hospital Research Ins.tute University of OFawa
 Epigene.cs: What is it and how it effects our health? Dr. Bill Stanford, PhD OFawa Hospital Research Ins.tute University of OFawa Overview Basic Background Epigene.cs in general Epigene.cs in cancer Epigene.cs
Epigene.cs: What is it and how it effects our health? Dr. Bill Stanford, PhD OFawa Hospital Research Ins.tute University of OFawa Overview Basic Background Epigene.cs in general Epigene.cs in cancer Epigene.cs
Datamonitor Healthcare Pharma intelligence. GlaxoSmithKline: Company Analysis
 Datamonitor Healthcare Pharma intelligence GlaxoSmithKline: Company Analysis OVERVIEW Latest Earnings Review Q2 2018 [27 July 2018] Click to view analysis Latest Forecast Update Q2 2018 [2 August 2018]
Datamonitor Healthcare Pharma intelligence GlaxoSmithKline: Company Analysis OVERVIEW Latest Earnings Review Q2 2018 [27 July 2018] Click to view analysis Latest Forecast Update Q2 2018 [2 August 2018]
Cytomegalovirus (CMV) Infections - Pipeline Assessment and Market Forecasts to 2019
 Reference Code: GDHC504PRT Publication Date: January 2013 Cytomegalovirus Infections: Key Metrics in Seven Major Pharmaceutical Markets 2012 Epidemiology* Prevalent population (CMV prevalent cases in general
Reference Code: GDHC504PRT Publication Date: January 2013 Cytomegalovirus Infections: Key Metrics in Seven Major Pharmaceutical Markets 2012 Epidemiology* Prevalent population (CMV prevalent cases in general
Lecture 8. Eukaryotic gene regulation: post translational modifications of histones
 Lecture 8 Eukaryotic gene regulation: post translational modifications of histones Recap.. Eukaryotic RNA polymerases Core promoter elements General transcription factors Enhancers and upstream activation
Lecture 8 Eukaryotic gene regulation: post translational modifications of histones Recap.. Eukaryotic RNA polymerases Core promoter elements General transcription factors Enhancers and upstream activation
[ NASDAQ: MEIP ] Cowen and Company Health Care Conference March 2015
![[ NASDAQ: MEIP ] Cowen and Company Health Care Conference March 2015 [ NASDAQ: MEIP ] Cowen and Company Health Care Conference March 2015](/thumbs/90/104469787.jpg) [ NASDAQ: MEIP ] Cowen and Company Health Care Conference March 2015 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events or
[ NASDAQ: MEIP ] Cowen and Company Health Care Conference March 2015 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events or
Revolutionizing the Treatment of Cancer
 Revolutionizing the Treatment of Cancer September 2013 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results
Revolutionizing the Treatment of Cancer September 2013 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results
PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM
 HEMATOLOGY ONCOLOGY PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM COMMITTED TO ADVANCING DRUG DEVELOPMENT IN ONCOLOGY $ MARKETPLACE COMPLEXITIES increasingly competitive marketplace and rising cost pressures
HEMATOLOGY ONCOLOGY PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM COMMITTED TO ADVANCING DRUG DEVELOPMENT IN ONCOLOGY $ MARKETPLACE COMPLEXITIES increasingly competitive marketplace and rising cost pressures
DS-8201 Strategic Collaboration
 DS-8201 Strategic Collaboration DAIICHI SANKYO CO., LTD George Nakayama Chairman and CEO March 29, 2019 Forward-Looking Statements Management strategies and plans, financial forecasts, future projections
DS-8201 Strategic Collaboration DAIICHI SANKYO CO., LTD George Nakayama Chairman and CEO March 29, 2019 Forward-Looking Statements Management strategies and plans, financial forecasts, future projections
R. Piazza (MD, PhD), Dept. of Medicine and Surgery, University of Milano-Bicocca EPIGENETICS
 R. Piazza (MD, PhD), Dept. of Medicine and Surgery, University of Milano-Bicocca EPIGENETICS EPIGENETICS THE STUDY OF CHANGES IN GENE EXPRESSION THAT ARE POTENTIALLY HERITABLE AND THAT DO NOT ENTAIL A
R. Piazza (MD, PhD), Dept. of Medicine and Surgery, University of Milano-Bicocca EPIGENETICS EPIGENETICS THE STUDY OF CHANGES IN GENE EXPRESSION THAT ARE POTENTIALLY HERITABLE AND THAT DO NOT ENTAIL A
Are you the way you are because of the
 EPIGENETICS Are you the way you are because of the It s my fault!! Nurture Genes you inherited from your parents? Nature Experiences during your life? Similar DNA Asthma, Autism, TWINS Bipolar Disorders
EPIGENETICS Are you the way you are because of the It s my fault!! Nurture Genes you inherited from your parents? Nature Experiences during your life? Similar DNA Asthma, Autism, TWINS Bipolar Disorders
Media Contact: Ron Rogers Investor Contact: Scott Gleason (801) (801)
 News Release Media Contact: Ron Rogers Investor Contact: Scott Gleason (801) 584-3065 (801) 584-1143 rrogers@myriad.com sgleason@myriad.com Myriad Receives FDA Approval of BRACAnalysis CDx as Companion
News Release Media Contact: Ron Rogers Investor Contact: Scott Gleason (801) 584-3065 (801) 584-1143 rrogers@myriad.com sgleason@myriad.com Myriad Receives FDA Approval of BRACAnalysis CDx as Companion
[ NASDAQ: MEIP ] Wedbush PacGrow Healthcare Conference August 16-17, 2016
![[ NASDAQ: MEIP ] Wedbush PacGrow Healthcare Conference August 16-17, 2016 [ NASDAQ: MEIP ] Wedbush PacGrow Healthcare Conference August 16-17, 2016](/thumbs/79/78947392.jpg) [ NASDAQ: MEIP ] Wedbush PacGrow Healthcare Conference August 16-17, 2016 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events
[ NASDAQ: MEIP ] Wedbush PacGrow Healthcare Conference August 16-17, 2016 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events
Inovio Pharmaceuticals, Inc. (Exact name of registrant as specified in its charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of The Securities Exchange Act of 1934 Date of Report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of The Securities Exchange Act of 1934 Date of Report (Date of earliest event
[ NASDAQ: MEIP ] Stifel Healthcare Conference November 15-16, 2016
![[ NASDAQ: MEIP ] Stifel Healthcare Conference November 15-16, 2016 [ NASDAQ: MEIP ] Stifel Healthcare Conference November 15-16, 2016](/thumbs/90/104469815.jpg) [ NASDAQ: MEIP ] Stifel Healthcare Conference November 15-16, 2016 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events or results
[ NASDAQ: MEIP ] Stifel Healthcare Conference November 15-16, 2016 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events or results
Comparative effect of Various HDAC-inhibitors in-vitro on T- Cell Lymphoma cell lines alone and in combination with conventional anti-cancer drugs
 Comparative effect of Various HDAC-inhibitors in-vitro on T- Cell Lymphoma cell lines alone and in combination with conventional anti-cancer drugs Arshad H. Banday Mentor:Dr. Francisco Hernandez-Illizaliturri
Comparative effect of Various HDAC-inhibitors in-vitro on T- Cell Lymphoma cell lines alone and in combination with conventional anti-cancer drugs Arshad H. Banday Mentor:Dr. Francisco Hernandez-Illizaliturri
VeriStrat Poor Patients Show Encouraging Overall Survival and Progression Free Survival Signal; Confirmatory Phase 2 Study Planned by Year-End
 AVEO and Biodesix Announce Exploratory Analysis of VeriStrat-Selected Patients with Non-Small Cell Lung Cancer in Phase 2 Study of Ficlatuzumab Presented at ESMO 2014 Congress VeriStrat Poor Patients Show
AVEO and Biodesix Announce Exploratory Analysis of VeriStrat-Selected Patients with Non-Small Cell Lung Cancer in Phase 2 Study of Ficlatuzumab Presented at ESMO 2014 Congress VeriStrat Poor Patients Show
Global Strategic Partners Merck and Pfizer Finalize Agreement to Co-Promote XALKORI (crizotinib)
 Merck Media: Markus Talanow +49 6151 72 7144 Investor Relations: +49 6151 72 3321 Pfizer Media: Sally Beatty +1 212 733 6566 Investor Relations: Ryan Crowe +1 212 733 8160 News Release April 7, 2015 Not
Merck Media: Markus Talanow +49 6151 72 7144 Investor Relations: +49 6151 72 3321 Pfizer Media: Sally Beatty +1 212 733 6566 Investor Relations: Ryan Crowe +1 212 733 8160 News Release April 7, 2015 Not
Business Update & Financial Results for Q1 2018
 Business Update & Financial Results for Q1 2018 May 15, 2018 Disclaimer The statements made in this presentation may include forward-looking statements regarding the future operations of ERYTECH Pharma
Business Update & Financial Results for Q1 2018 May 15, 2018 Disclaimer The statements made in this presentation may include forward-looking statements regarding the future operations of ERYTECH Pharma
FOURTH QUARTER AND FULL YEAR 2018 FINANCIAL RESULTS AND BUSINESS UPDATE. Thursday, February 7, 2019
 FOURTH QUARTER AND FULL YEAR 2018 FINANCIAL RESULTS AND BUSINESS UPDATE Thursday, February 7, 2019 Today s Speakers Overview and Key Highlights Clay Siegall, President & CEO Financial Results and Guidance
FOURTH QUARTER AND FULL YEAR 2018 FINANCIAL RESULTS AND BUSINESS UPDATE Thursday, February 7, 2019 Today s Speakers Overview and Key Highlights Clay Siegall, President & CEO Financial Results and Guidance
Your partner for Medical Research and Development
 Your partner for Medical Research and Development Overview Integrated place for clinical research SERVICES AND RESOURCES DNA Sequencing Proteomics Imaging Histology Flow Cytometry Sample Processing Cryogenics
Your partner for Medical Research and Development Overview Integrated place for clinical research SERVICES AND RESOURCES DNA Sequencing Proteomics Imaging Histology Flow Cytometry Sample Processing Cryogenics
Spectrum Pharmaceuticals
 Spectrum Pharmaceuticals Joe Turgeon President and CEO June 2018 Investor Presentation 1 Safe Harbor Statement This presentation contains forward looking statements regarding future events and the future
Spectrum Pharmaceuticals Joe Turgeon President and CEO June 2018 Investor Presentation 1 Safe Harbor Statement This presentation contains forward looking statements regarding future events and the future
Personalized Therapeutics The Power of Epigenetics. Company Overview. June 2014
 Personalized Therapeutics The Power of Epigenetics Company Overview June 2014 2013 Accomplishments Forward Looking Statements This presentation contains forward-looking statements that involve substantial
Personalized Therapeutics The Power of Epigenetics Company Overview June 2014 2013 Accomplishments Forward Looking Statements This presentation contains forward-looking statements that involve substantial
Oncology Therapeutics without Compromise APRIL 2011
 Oncology Therapeutics without Compromise APRIL 2011 Forward Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties, including among other
Oncology Therapeutics without Compromise APRIL 2011 Forward Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties, including among other
INTERIM RESULTS AS OF JUNE 30, 2014 CSE/OMX:BAVA, OTC:BVNRY
 INTERIM RESULTS AS OF JUNE 30, 2014 Q2 AUGUST 28, 2014 CSE/OMX:BAVA, OTC:BVNRY FINANCIAL HIGHLIGHTS 2.6 million doses IMVAMUNE delivered to the SNS during H1 (0.9 million in Q2) Increased contribution
INTERIM RESULTS AS OF JUNE 30, 2014 Q2 AUGUST 28, 2014 CSE/OMX:BAVA, OTC:BVNRY FINANCIAL HIGHLIGHTS 2.6 million doses IMVAMUNE delivered to the SNS during H1 (0.9 million in Q2) Increased contribution
HDAC1 Inhibitor Screening Assay Kit
 HDAC1 Inhibitor Screening Assay Kit Catalog Number KA1320 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
HDAC1 Inhibitor Screening Assay Kit Catalog Number KA1320 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
HDAC Inhibitors and PARP inhibitors. Suresh Ramalingam, MD Associate Professor Chief of Thoracic Oncology Emory University School of Medicine
 HDAC Inhibitors and PARP inhibitors Suresh Ramalingam, MD Associate Professor Chief of Thoracic Oncology Emory University School of Medicine Histone Acetylation HAT Ac Ac Ac Ac HDAC Ac Ac Ac Ac mrna DACs
HDAC Inhibitors and PARP inhibitors Suresh Ramalingam, MD Associate Professor Chief of Thoracic Oncology Emory University School of Medicine Histone Acetylation HAT Ac Ac Ac Ac HDAC Ac Ac Ac Ac mrna DACs
Myeloid Differentiation Observed, Including Induction of CD38 in 85% of Evaluable Patients
 December 10, 2017 Syros Announces Initial Clinical Data from Ongoing Phase 2 Trial of SY-1425 Showing Biological and Clinical Activity as Single Agent in Genomically Defined AML and MDS Patients Clinical
December 10, 2017 Syros Announces Initial Clinical Data from Ongoing Phase 2 Trial of SY-1425 Showing Biological and Clinical Activity as Single Agent in Genomically Defined AML and MDS Patients Clinical
[ NASDAQ: MEIP ] Cantor Fitzgerald Healthcare Conference July 8, 2015
![[ NASDAQ: MEIP ] Cantor Fitzgerald Healthcare Conference July 8, 2015 [ NASDAQ: MEIP ] Cantor Fitzgerald Healthcare Conference July 8, 2015](/thumbs/86/94600704.jpg) [ NASDAQ: MEIP ] Cantor Fitzgerald Healthcare Conference July 8, 2015 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events or
[ NASDAQ: MEIP ] Cantor Fitzgerald Healthcare Conference July 8, 2015 Forward-Looking Statements These slides and the accompanying oral presentation contain forward-looking statements. Actual events or
Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma
 PRESS RELEASE IMMUNOCORE LIMITED Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma (Oxford, UK, 16 April 2015) Immunocore Limited, a world-leading
PRESS RELEASE IMMUNOCORE LIMITED Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma (Oxford, UK, 16 April 2015) Immunocore Limited, a world-leading
First Quarter 2018 Results
 First Quarter 2018 Results 1 April 27, 2017 Investor call Q1 2017 April 27, 2018 First quarter and after period close highlights and significant events Strong progress in the MIV-818 project Successful
First Quarter 2018 Results 1 April 27, 2017 Investor call Q1 2017 April 27, 2018 First quarter and after period close highlights and significant events Strong progress in the MIV-818 project Successful
SG Cowen 23rd Annual Health Care Conference March 18, Shire Pharmaceuticals Group plc
 SG Cowen 23rd Annual Health Care Conference March 18, 2003 Shire Pharmaceuticals Group plc THE SAFE HARBOR STATEMENT UNDER THE PRIVATE SECURITIES LITIGATION REFORM ACT OF 1995 Statements included herein
SG Cowen 23rd Annual Health Care Conference March 18, 2003 Shire Pharmaceuticals Group plc THE SAFE HARBOR STATEMENT UNDER THE PRIVATE SECURITIES LITIGATION REFORM ACT OF 1995 Statements included herein
AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer
 FOR IMMEDIATE RELEASE AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer Study designed to build upon safety profile demonstrated
FOR IMMEDIATE RELEASE AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer Study designed to build upon safety profile demonstrated
Acquisition of Tolero Pharmaceuticals, Inc. December 21, 2016 Sumitomo Dainippon Pharma Co., Ltd.
 Acquisition of Tolero Pharmaceuticals, Inc. December 21, 2016 Sumitomo Dainippon Pharma Co., Ltd. Acquisition of Tolero Pharmaceuticals, Inc. Significance of the Acquisition Acquire attractive compounds
Acquisition of Tolero Pharmaceuticals, Inc. December 21, 2016 Sumitomo Dainippon Pharma Co., Ltd. Acquisition of Tolero Pharmaceuticals, Inc. Significance of the Acquisition Acquire attractive compounds
NEWSLETTER June Summer greetings from Follicum
 NEWSLETTER June 2018 Follicum hopes that this newsletter reaches as many of the company's owners and stakeholders as possible. If you wish to follow our continued development, please register your e-mail
NEWSLETTER June 2018 Follicum hopes that this newsletter reaches as many of the company's owners and stakeholders as possible. If you wish to follow our continued development, please register your e-mail
VAL-083: Validated DNA-targeting Agent for Underserved Cancer Patients. September 2018
 VAL-083: Validated DNA-targeting Agent for Underserved Cancer Patients September 2018 Forward-Looking Statements Any statements contained in this presentation that do not describe historical facts may
VAL-083: Validated DNA-targeting Agent for Underserved Cancer Patients September 2018 Forward-Looking Statements Any statements contained in this presentation that do not describe historical facts may
Jefferies 2015 Global Healthcare Conference June 1, 2015
 Jefferies 2015 Global Healthcare Conference June 1, 2015 Page 0 Acceleron Forward-Looking Statements This presentation contains forward-looking statements. Forward-looking statements are neither historical
Jefferies 2015 Global Healthcare Conference June 1, 2015 Page 0 Acceleron Forward-Looking Statements This presentation contains forward-looking statements. Forward-looking statements are neither historical
Genta Incorporated. A Multiproduct Late-Stage Oncology Company
 Genta Incorporated A Multiproduct Late-Stage Oncology Company This presentation may contain forward-looking statements with respect to business conducted by Genta Incorporated. By their nature, forward-looking
Genta Incorporated A Multiproduct Late-Stage Oncology Company This presentation may contain forward-looking statements with respect to business conducted by Genta Incorporated. By their nature, forward-looking
Bio-Path Announces Clinical Update to Interim Analysis of Phase 2 Prexigebersen Trial in Acute Myeloid Leukemia
 Bio-Path Announces Clinical Update to Interim Analysis of Phase 2 Prexigebersen Trial in Acute Myeloid Leukemia Interim Data Update from Phase 2 Study Demonstrates Meaningful Clinical Improvement with
Bio-Path Announces Clinical Update to Interim Analysis of Phase 2 Prexigebersen Trial in Acute Myeloid Leukemia Interim Data Update from Phase 2 Study Demonstrates Meaningful Clinical Improvement with
PIERIS PHARMACEUTICALS, INC. (Exact Name of Registrant as Specified in its Charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event
Custom Intelligence. Alzheimer s Disease Landscape Summary
 Custom Intelligence Alzheimer s Disease Landscape Summary Prepared August 21 Alzheimer s Disease Landscape: Executive Summary Current standard of care in Alzheimer s Disease (AD) consists of symptomatic
Custom Intelligence Alzheimer s Disease Landscape Summary Prepared August 21 Alzheimer s Disease Landscape: Executive Summary Current standard of care in Alzheimer s Disease (AD) consists of symptomatic
Corporate Presentation May Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers
 Corporate Presentation May 2016 Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers Forward-looking statements / safe harbor This presentation and the accompanying oral commentary contain
Corporate Presentation May 2016 Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers Forward-looking statements / safe harbor This presentation and the accompanying oral commentary contain
Sunesis Pharmaceuticals Reports Second Quarter 2011 Financial Results
 Investor and Media Inquiries: David Pitts Argot Partners 212-600-1902 Eric Bjerkholt Sunesis Pharmaceuticals Inc. 650-266-3717 Sunesis Pharmaceuticals Reports Second Quarter 2011 Financial Results SOUTH
Investor and Media Inquiries: David Pitts Argot Partners 212-600-1902 Eric Bjerkholt Sunesis Pharmaceuticals Inc. 650-266-3717 Sunesis Pharmaceuticals Reports Second Quarter 2011 Financial Results SOUTH
Under the terms of the agreement, AVEO will receive an initial cash payment of $125 million, composed of a $75 million license fee and $50 million in
 Astellas Pharma Inc. AVEO Pharmaceuticals Corporate Communications Investor Contact: Astellas Pharma Inc. Monique Allaire AVEO Pharmaceuticals +81(3)-3244-3201 (617) 299-5810 Media Contact: Dan Budwick
Astellas Pharma Inc. AVEO Pharmaceuticals Corporate Communications Investor Contact: Astellas Pharma Inc. Monique Allaire AVEO Pharmaceuticals +81(3)-3244-3201 (617) 299-5810 Media Contact: Dan Budwick
AstraZeneca enhances long-term growth through Oncology investment in Acerta Pharma
 AstraZeneca enhances long-term growth through Oncology investment in Acerta Pharma PUBLISHED17 December 2015Transaction includes late-stage, potential best-in-class irreversible small molecule BTK inhibitor,
AstraZeneca enhances long-term growth through Oncology investment in Acerta Pharma PUBLISHED17 December 2015Transaction includes late-stage, potential best-in-class irreversible small molecule BTK inhibitor,
Myriad Genetics mychoice HRD Update 06/30/2016
 Myriad Genetics mychoice HRD Update 06/30/2016 1 Forward Looking Statements Some of the information presented here today may contain projections or other forward-looking statements regarding future events
Myriad Genetics mychoice HRD Update 06/30/2016 1 Forward Looking Statements Some of the information presented here today may contain projections or other forward-looking statements regarding future events
Bank of America Merrill Lynch 2016 Health Care Conference
 Bank of America Merrill Lynch 2016 Health Care Conference Dr. Steven Stein Chief Medical Officer David Gryska Chief Financial Officer May 11, 2016 Forward Looking Statements Except for the historical information
Bank of America Merrill Lynch 2016 Health Care Conference Dr. Steven Stein Chief Medical Officer David Gryska Chief Financial Officer May 11, 2016 Forward Looking Statements Except for the historical information
SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS
 SPHERIX Investor Relations Phone: (301) 897-2564 Email: info@spherix.com SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS BETHESDA, MD, May 21, 2010 - Spherix Incorporated (NASDAQ CM: SPEX), an innovator
SPHERIX Investor Relations Phone: (301) 897-2564 Email: info@spherix.com SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS BETHESDA, MD, May 21, 2010 - Spherix Incorporated (NASDAQ CM: SPEX), an innovator
Melanoma Research Alliance-Pfizer Academic-Industry Partnership Awards
 Melanoma Research Alliance-Pfizer Academic-Industry Partnership Awards Wendy K.D. Selig, President & CEO, MRA Julia Perkins, M.D. Medical Director, Oncology, Pfizer Melanoma Research Alliance Mission:
Melanoma Research Alliance-Pfizer Academic-Industry Partnership Awards Wendy K.D. Selig, President & CEO, MRA Julia Perkins, M.D. Medical Director, Oncology, Pfizer Melanoma Research Alliance Mission:
Positioned for Growth
 Positioned for Growth Annual Shareholders Meeting July 21, 2016 Paris Panayiotopoulos President and Chief Executive Officer David Sachs Non small cell lung cancer ARIAD clinical trial patient This presentation
Positioned for Growth Annual Shareholders Meeting July 21, 2016 Paris Panayiotopoulos President and Chief Executive Officer David Sachs Non small cell lung cancer ARIAD clinical trial patient This presentation
Nektar Investor & Analyst Call. Nektar & z Bristol-Myers Squibb Collaboration. February 14, 2018
 Nektar Investor & Analyst Call Nektar & z Bristol-Myers Squibb Collaboration February 14, 2018 This presentation includes forward-looking statements regarding Nektar s proprietary drug candidates, the
Nektar Investor & Analyst Call Nektar & z Bristol-Myers Squibb Collaboration February 14, 2018 This presentation includes forward-looking statements regarding Nektar s proprietary drug candidates, the
TELECONFERENCE FY February 2015
 TELECONFERENCE FY 2014 5 February 2015 Company disclaimer This presentation contains forward-looking statements that provide our expectations or forecasts of future events such as new product introductions,
TELECONFERENCE FY 2014 5 February 2015 Company disclaimer This presentation contains forward-looking statements that provide our expectations or forecasts of future events such as new product introductions,
NewLink Genetics Corporation Provides Operational Update and Reports Second Quarter 2015 Financial Results
 July 31, 2015 NewLink Genetics Corporation Provides Operational Update and Reports Second Quarter 2015 Financial Results -Management to Host Conference Call Today at 8:30 a.m. ET- AMES, Iowa, July 31,
July 31, 2015 NewLink Genetics Corporation Provides Operational Update and Reports Second Quarter 2015 Financial Results -Management to Host Conference Call Today at 8:30 a.m. ET- AMES, Iowa, July 31,
33 rd Annual J.P. Morgan Healthcare Conference. January 2015
 33 rd Annual J.P. Morgan Healthcare Conference January 2015 Forward-looking Statements This presentation contains forward-looking statements, which express the current beliefs and expectations of management.
33 rd Annual J.P. Morgan Healthcare Conference January 2015 Forward-looking Statements This presentation contains forward-looking statements, which express the current beliefs and expectations of management.
Jefferies 2016 Healthcare Conference. Reid Huber, PhD Chief Scientific Officer
 Jefferies 2016 Healthcare Conference Reid Huber, PhD Chief Scientific Officer June 8, 2016 Forward-looking Statements Except for the historical information set forth herein, the matters set forth in this
Jefferies 2016 Healthcare Conference Reid Huber, PhD Chief Scientific Officer June 8, 2016 Forward-looking Statements Except for the historical information set forth herein, the matters set forth in this
Corporate Overview May 8, 2014
 0 Corporate Overview May 8, 2014 NASDAQ: CRIS Forward Looking Statements This presentation contains statements about Curis future expectations, plans and prospects that constitute forward-looking statements
0 Corporate Overview May 8, 2014 NASDAQ: CRIS Forward Looking Statements This presentation contains statements about Curis future expectations, plans and prospects that constitute forward-looking statements
Photocure ASA Executing the Strategy
 Photocure ASA Executing the Strategy NOVEMBER 2012 ERIK DAHL, CFO KATHLEEN DEARDORFF, COO Disclaimer The information included in this Presentation contains certain forward-looking statements that address
Photocure ASA Executing the Strategy NOVEMBER 2012 ERIK DAHL, CFO KATHLEEN DEARDORFF, COO Disclaimer The information included in this Presentation contains certain forward-looking statements that address
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics. Jefferies 2014 Global Healthcare Conference Cynthia L. Sullivan, President and CEO
 IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases Jefferies 2014 Global Healthcare Conference Cynthia L. Sullivan, President and CEO Forward-Looking Statements This presentation,
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases Jefferies 2014 Global Healthcare Conference Cynthia L. Sullivan, President and CEO Forward-Looking Statements This presentation,
Myriad Genetics Corporate Presentation 6/4/13
 Myriad Genetics Corporate Presentation 6/4/13 Forward Looking Statement Some of the information presented here today may contain projections or other forward-looking statements regarding future events
Myriad Genetics Corporate Presentation 6/4/13 Forward Looking Statement Some of the information presented here today may contain projections or other forward-looking statements regarding future events
National Horizon Scanning Centre. Azacitidine (Vidaza) for myelodysplastic syndrome. September 2007
 Azacitidine (Vidaza) for myelodysplastic syndrome September 2007 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Azacitidine (Vidaza) for myelodysplastic syndrome September 2007 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Company presentation. Claudio Bordignon, Chairman and CEO. Jefferies Global Healthcare Conference New York, 7 June 2012
 Company presentation Claudio Bordignon, Chairman and CEO Jefferies Global Healthcare Conference New York, 7 June 2012 Forward-looking statements The presentation contains certain forward-looking statements.
Company presentation Claudio Bordignon, Chairman and CEO Jefferies Global Healthcare Conference New York, 7 June 2012 Forward-looking statements The presentation contains certain forward-looking statements.
Investor Presentation
 Investor Presentation February 2018 2 FORWARD-LOOKING INFORMATION The following presentation contains statements that are considered forward-looking information ( FLI ) within the meaning of securities
Investor Presentation February 2018 2 FORWARD-LOOKING INFORMATION The following presentation contains statements that are considered forward-looking information ( FLI ) within the meaning of securities
How Cells Divide. Chapter 10
 How Cells Divide Chapter 10 Bacterial Cell Division Bacteria divide by binary fission. -the single, circular bacterial chromosome is replicated -replication begins at the origin of replication and proceeds
How Cells Divide Chapter 10 Bacterial Cell Division Bacteria divide by binary fission. -the single, circular bacterial chromosome is replicated -replication begins at the origin of replication and proceeds
9/3/2009 DNA i DNA n euk euk yotes Organizatio Organ izatio n of o f gen ge e n tic Locati t on: In n ucleu e s material mater in e ial
 DNA in eukaryotes Organization of genetic material in eukaryotes Location: In nucleus In mitochondria DNA in eukaryotes Nuclear DNA: Long, linear molecules; Chromatin chromosomes; 10% of DNA in genes,
DNA in eukaryotes Organization of genetic material in eukaryotes Location: In nucleus In mitochondria DNA in eukaryotes Nuclear DNA: Long, linear molecules; Chromatin chromosomes; 10% of DNA in genes,
September 20, Submitted electronically to: Cc: To Whom It May Concern:
 History Study (NOT-HL-12-147), p. 1 September 20, 2012 Re: Request for Information (RFI): Building a National Resource to Study Myelodysplastic Syndromes (MDS) The MDS Cohort Natural History Study (NOT-HL-12-147).
History Study (NOT-HL-12-147), p. 1 September 20, 2012 Re: Request for Information (RFI): Building a National Resource to Study Myelodysplastic Syndromes (MDS) The MDS Cohort Natural History Study (NOT-HL-12-147).
Biochemical Determinants Governing Redox Regulated Changes in Gene Expression and Chromatin Structure
 Biochemical Determinants Governing Redox Regulated Changes in Gene Expression and Chromatin Structure Frederick E. Domann, Ph.D. Associate Professor of Radiation Oncology The University of Iowa Iowa City,
Biochemical Determinants Governing Redox Regulated Changes in Gene Expression and Chromatin Structure Frederick E. Domann, Ph.D. Associate Professor of Radiation Oncology The University of Iowa Iowa City,
FDA APPROVES HERCEPTIN FOR THE ADJUVANT TREATMENT OF HER2-POSITIVE NODE-POSITIVE BREAST CANCER
 NEWS RELEASE Media Contact: Kimberly Ocampo (650) 467-0679 Investor Contact: Sue Morris (650) 225-6523 Advocacy Contact: Ajanta Horan (650) 467-1741 FDA APPROVES HERCEPTIN FOR THE ADJUVANT TREATMENT OF
NEWS RELEASE Media Contact: Kimberly Ocampo (650) 467-0679 Investor Contact: Sue Morris (650) 225-6523 Advocacy Contact: Ajanta Horan (650) 467-1741 FDA APPROVES HERCEPTIN FOR THE ADJUVANT TREATMENT OF
H. LUNDBECK A/S. Teleconference. 10 August PM CET. Financial results Second quarter 2011
 H. LUNDBECK A/S Teleconference 10 August 2011-2PM CET Financial results Second quarter 2011 Company disclaimer This presentation contains forward-looking statements that provide our expectations or forecasts
H. LUNDBECK A/S Teleconference 10 August 2011-2PM CET Financial results Second quarter 2011 Company disclaimer This presentation contains forward-looking statements that provide our expectations or forecasts
MARCH 2017 RESEARCH REPORT
 1 MARCH 2017 RESEARCH REPORT LIQUID BIOPSY MARKETS by Lung, Breast, Colorectal, Prostate and Other. 2017-2020 Including Screening Market Potential Size with Executive and Consultant Guides Liquid Biopsy
1 MARCH 2017 RESEARCH REPORT LIQUID BIOPSY MARKETS by Lung, Breast, Colorectal, Prostate and Other. 2017-2020 Including Screening Market Potential Size with Executive and Consultant Guides Liquid Biopsy
