UC Irvine UC Irvine Previously Published Works
|
|
|
- Maud Hampton
- 5 years ago
- Views:
Transcription
1 UC Irvine UC Irvine Previously Published Works Title Pharmacological profile of the selective FAAH inhibitor KDS-413 (URB597) Permalink Journal CNS Drug Reviews, 12(1) ISSN X Authors Piomelli, D Tarzia, G Duranti, A et al. Publication Date DI /j x License CC BY 4. Peer reviewed escholarship.org Powered by the California Digital Library University of California
2 CNS Drug Reviews Vol. 12, No. 1, pp The Authors Journal compilation 26 Blackwell Publishing Inc. Pharmacological Profile of the Selective FAAH Inhibitor KDS-413 (URB597) Daniele Piomelli 1,2, Giorgio Tarzia 3, Andrea Duranti 3, Andrea Tontini 3, Marco Mor 4, Timothy R. Compton 1, livier Dasse 1, Edward P. Monaghan 1, Jeff A. Parrott 1, and David Putman 1 1 Kadmus Pharmaceuticals, Inc. Irvine, California, USA; 2 Departments of Pharmacology and Biological Chemistry, Center for Drug Discovery, University of California, Irvine, California, USA; 3 Institute of Medicinal Chemistry, University of Urbino Carlo Bo, Urbino, Italy; 4 Pharmaceutical Department, University of Parma, Parma, Italy Keywords: Anandamide Anxiety Cannabinoids Depression Fatty-acid amide hydrolase Pain. ABSTRACT In the present article, we review the pharmacological properties of KDS-413 (URB597), a highly potent and selective inhibitor of the enzyme fatty-acid amide hydrolase (FAAH), which catalyzes the intracellular hydrolysis of the endocannabinoid anandamide. In vitro, KDS-413 inhibits FAAH activity with median inhibitory concentrations (IC 5 ) of 5 nm in rat brain membranes and 3 nm in human liver microsomes. In vivo, KDS-413 inhibits rat brain FAAH activity after intraperitoneal (i.p.) administration with a median inhibitory dose (ID 5 ) of.15 mg kg. The compound does not significantly interact with other cannabinoid-related targets, including cannabinoid receptors and anandamide transport, or with a broad panel of receptors, ion channels, transporters and enzymes. By i.p. administration to rats and mice KDS-413 elicits significant, anxiolytic-like, antidepressant-like and analgesic effects, which are prevented by treatment with CB1 receptor antagonists. By contrast, at doses that significantly inhibit FAAH activity and substantially raise brain anandamide levels, KDS-413 does not evoke classical cannabinoid-like effects (e.g., catalepsy, hypothermia, hyperphagia), does not cause place preference, and does not produce generalization to the discriminative effects of the active ingredient of cannabis, Ä 9 -tetrahydrocannabinol (Ä 9 -THC). These findings suggest that KDS-413 acts by enhancing the tonic actions of anandamide on a subset of CB 1 receptors, which may Address correspondence and reprint requests to: Daniele Piomelli, PhD, Department of Pharmacology, 36 MSR II, University of California, Irvine, CA , USA; Tel.: +1 (949) , Fax: +1 (949) , piomelli@uci.edu 21
3 22 D. PIMELLI ET AL. normally be engaged in controlling emotions and pain. KDS-413 is orally available in rats and cynomolgus monkeys. Sub-chronic repeated dose studies (15 mg kg, per os)in these two species have not demonstrated systemic toxicity. Likewise, no toxicity was noted in bacterial cytotoxicity tests in vitro and in the Ames test. Furthermore, no deficits were observed in rats on the rotarod test after acute i.p. treatment with KDS-413 at doses up to 5 mg kg or in a functional observation battery after oral doses up to 15 mg kg. The results suggest that KDS-413 will offer a novel approach with a favorable therapeutic window for the treatment of anxiety, depression and pain. INTRDUCTIN The Endocannabinoid System The brain endocannabinoid signaling system is composed of three main elements. The first is represented by the G protein-coupled receptors that bind endogenous and exogenous cannabinoid ligands. Two such receptors have been molecularly cloned the CB 1, which is found almost everywhere in the body, but is most abundant in the central nervous system (CNS) (15); and the CB 2, which is primarily expressed in immune cells, but is also present at low levels in the brain (38,51). The second element is represented by the endocannabinoids, naturally occurring lipid molecules that bind to and activate cannabinoid receptors (11,35,46), are generated on demand by neurons and other cells (12,17,45), and are rapidly eliminated (3,45). The third element is represented by the proteins involved in the formation and elimination of the various endocannabinoid ligands identified thus far (42). Anandamide (arachidonoylethanolamide) was the first endocannabinoid substance to be discovered (11,42). Current evidence indicates that this lipid-derived mediator is released upon demand by stimulated neurons (12,17); activates cannabinoid receptors with high potency (11), and is rapidly eliminated through a two-step process consisting of carrier-mediated internalization followed by intracellular hydrolysis (4,12, 22). Fatty-Acid Amide Hydrolase (FAAH) Intracellular anandamide hydrolysis is primarily catalyzed by a membrane-bound serine hydrolase enzyme, whose existence was first suggested by biochemical experiments (1,21,44,49) and then demonstrated by purification, molecular cloning and heterologous expression (7). This enzyme, called FAAH in recognition of the large number of endogenous fatty-acid amides that are accepted as substrates, displays significant homology with the amidase signature family of enzymes. In vitro, FAAH can act as a hydrolytic enzyme not only for fatty-acid ethanolamides such as anandamide, but also for primary amides such as oleamide and even for esters such as 2-arachidonoylglycerol (2-AG), a major endocannabinoid ligand in the brain (35,45,46). Site-directed mutagenesis experiments have shown that this broad substrate preference may be due to a previously undescribed catalytic mechanism involving the amino acid residues lysine 142, serine 241, and serine 217 (34), whose energetic aspects have been investigated by computational methods (29).
4 KDS FAAH is abundantly expressed throughout the CNS, with particularly high levels in neocortex, hippocampus, and basal ganglia (15). In situ hybridization studies in the rat have shown that FAAH mrna levels are highest in the neocortex and hippocampus; intermediate in the cerebellum, thalamus, olfactory bulb and striatum; and lowest in hypothalamus, brain stem and pituitary gland. Immunohistochemical experiments confirmed these findings, showing that large principal neurons in the cerebral cortex, hippocampus, cerebellum and olfactory bulb have the highest levels of FAAH immunoreactivity. Moderate immunostaining has been also observed in amygdala, basal ganglia, ventral and posterior thalamus, deep cerebellar nuclei, superior colliculus, red nucleus, and motor neurons of the spinal cord (15). Many FAAH-positive neurons in the brain are found in proximity of nerve terminals that contain CB 1 cannabinoid receptors, supporting a role of FAAH in anandamide deactivation. There are, however, several regions of the brain where no such correlation can be demonstrated. This discrepancy is likely to reflect the participation of FAAH in the catabolism of non-cannabinoid fatty-acid ethanolamides, such as palmitoylethanolamide (PEA) and oleoylethanolamide (EA) (6,33,43), which are endogenous ligands for the nuclear receptor PPAR á (peroxisome proliferator-activated receptor type-á) (16,19,3). Consistent with this idea, pharmacological inhibition of FAAH activity or genetic disruption of the faah gene results in marked increases in the levels of these fatty acid ethanolamides along with anandamide (8,27). FAAH inhibitors A number of FAAH inhibitors have been described, including fatty-acid trifluoromethylketones, fluorophosphonates, á-keto esters and á-keto amides, bromoenol lactones, and non-steroidal antiinflammatory drugs. These compounds are exemplified by the bromoenol lactone BTNP (US patent 5,925,672 issued ), which inhibits FAAH activity with a half-maximal inhibitory concentration (IC 5 ) of 8 nm (3) and the fatty acid sulfonyl fluoride palmitylsulfonylfluoride (AM374), which inhibits FAAH activity with an IC 5 of 1 nm (US patent 5,688,825 issued ). These compounds lack, however, the target selectivity and or biological availability needed in a therapeutic drug. An emerging second generation of FAAH inhibitors comprises several structurally diverse groups of compounds (Fig. 1). These include substituted á-ketoheterocycles, developed at the Scripps Research Institute (US patent 6,462,54 issued ) (5); carbamate oxime (US patent 6,949,574 issued ) and bisarylimidazolyl derivatives (US patent 6,562,846 issued ; patent application W , ), developed at Bristol-Myers Squibb; dioxane-2-alkyl carbamates, developed at Sanofi- Synthélabo (patent application W 4 243, ), aryl alkyl carbamates (patent applications W published and W published ) and piperidinyl- and piperazinyl-alkyl carbamates (patent application W published ), developed at Sanofi-Synthélabo (Sanofi Aventis); tetrazole-based inhibitors, developed at Eli-Lilly (36); and alkylcarbamic acid aryl esters, developed at the Universities of California-Irvine, Urbino and Parma (patent application W , published ) (1,27,37,47,48). The index member of the latter
5 24 D. PIMELLI ET AL. NH 2 N H NH 2 H N KDS-413 (URB-597) Universities of California, Parma and Urbino Kadmus (Italy, USA) Sanofi-Synthélabo (France) L-135 The Scripps Research Institute (USA) N N N N HN Fig. 1. Chemical structures of various FAAH inhibitors. Bristol-Myers-Squibb (USA) F group of compounds, the compound URB597 (KDS-413) (Fig. 1) has been characterized both in vitro and in vivo and its properties are the subject of the present review. CHEMISTRY KDS-413 (cyclohexylcarbamic acid 3 -carbamoylbiphenyl-3-yl ester) is a crystalline white solid with a molecular weight of The compound has two hydrogen bond donors and five hydrogen bond acceptors. Its solubility in phosphate-buffered saline (PBS, ph 7.4) is 523 ng ml and it has a log D of 3.93 (n-octanol PBS, ph 7.4). KDS-413 has a high degree of membrane permeability, as determined in CaCo II TC7 cells, with an apparent permeability coefficient (A-B Permeability) of cm sec. It is stable under various conditions (heat, acid, peroxide, light, and simulated gastric fluid) (Kadmus Pharmaceuticals, unpublished results). The overall yield of a 3-kg bulk synthesis of the product is >8% with a purity of 98.5%. PHARMACLGY FAAH Inhibition in Vitro When FAAH activity was assayed in membrane fractions using [ 3 H]anandamide (anandamide[ethanolamine- 3 H]) as a substrate, KDS-413 was found to inhibit rat brain activity with an IC 5 of ~5 nm (27) and human liver activity with an IC 5 of 2.5 nm (Kadmus Pharmaceuticals, unpublished results). KDS-413 was also found to prevent the
6 [ H]AEA uptake (% control) A Veh AM44 [ H]AEA release (cpm) 3 KDS Vehicle KDS min 15min 3min AM44 + Fig. 2. Effects of the FAAH inhibitors, KDS-413, 1 nm, and KDS-411, 3 ìm, as well as anandamide transport inhibitor AM44 on (A) anandamide uptake into and (B) anandamide release from rat brain cortical neurons in primary cultures. Modified from ref. 27. B FAAH-catalyzed hydrolysis of [ 3 H]anandamide by primary cultures of rat cortical neurons with an IC 5 value of ~.5 nm (27). Notably, KDS-413 selectively blocks the breakdown of [ 3 H]anandamide without reducing its carrier-mediated uptake, causing non-metabolized [ 3 H]anandamide to accumulate in, and eventually exit from, the neurons. Thus, after a 4-min incubation with [ 3 H]anandamide, the intracellular content of non-metabolized [ 3 H]anandamide is markedly higher in inhibitor-treated than in control neurons (Fig. 2A). Similar results were obtained with KDS-411, a carbamate-based FAAH inhibitor that is weaker than KDS-413 at inhibiting FAAH activity (Fig. 2A). Importantly, the anandamide transport blocker N- (4-hydroxyphenyl) eicosa-5,8,11,15-tetraenamide (AM44) has an opposite effect, significantly reducing [ 3 H]anandamide internalization (Fig. 2A). When neurons treated with KDS-413 are exposed for 4 min to [ 3 H]anandamide and then incubated for 15 min in an [ 3 H]anandamide-free solution, ~43% of the accumulated [ 3 H]anandamide is released back into the medium (Fig. 2B). This process is linear with time (Fig. 2B) and is not inhibited by AM44 (Fig. 2A), suggesting that it is due to passive diffusion rather than reverse transport. No such time-dependent release is observed in control untreated neurons, the medium of which only contains residual levels of [ 3 H]anandamide carried over from the preincubation period (27). Together, these studies demonstrate that KDS-413 is potent at blocking anandamide hydrolysis in rat brain neurons without having any inhibitory effect on anandamide transport. As a result of its ability to block FAAH activity, KDS-413 causes extracellular levels of anandamide to increase. FAAH Inhibition in Vivo By intraperitoneal (i.p.) injections KDS-413 produced a profound dose-dependent inhibition of brain FAAH activity in rats, which is half-maximal at ~.15 mg kg (27). After injection of KDS-413 (.3 mg kg, i.p.), FAAH inhibition is rapid in onset (<15 min), persistent (>12 h) (Fig. 3A), and accompanied by significant elevations in the brain
7 26 D. PIMELLI ET AL. [3H]AEA hydrolysis (% control) A AEA (pmol/g) 6 3 B Veh 15 min 3 min 1h 2 h 4 h 6 h 16 h 24 h Veh 3 min 1h 2h 6h 16 h 24 h Fig. 3. Effects of parenteral administration of KDS-413 (.3 mg kg, i.p.) on (A) rat brain FAAH activity, assessed ex vivo by enzyme assay; and (B) brain anandamide levels, assessed ex vivo by liquid chromatography mass spectrometry. Modified from ref. 14. A 4 4 B Rectal temperature ( C) Rectal temperature ( C) Time (min) Time (min) Vehicle KDS-413 AEA AEA/KDS-413 Fig. 4. Effects of parenteral administration of anandamide (AEA, 5 mg kg, i.p.), KDS-413 (.3 mg kg, i.p.) or anandamide plus KDS-413 on body temperature in wild-type mice (+ +, left panel) or FAAH-null mice (, right panel). Modified from ref. 14. content of anandamide (Fig. 3B) and other fatty-acid ethanolamides that are substrates for FAAH (14). Similar changes in FAAH activity and fatty-acid ethanolamide levels are also observed in peripheral tissues (14). Significantly elevated anandamide levels are also observed in the brains of FAAH mice, and KDS-413 (.3 mg kg, i.p.) does not further alter these levels (14). In agreement with its inability to affect monoglyceride lipase (MGL) activity (Table 1), KDS-413 does not change the brain content of the endocannabinoid 2-arachidonoylglycerol (2-AG), a primary substrate for this enzyme (13). As previously observed in mutant FAAH mice, FAAH inhibition is associated with increased sensitivity to the administration of exogenous anandamide. Thus, KDS-413 (.3 mg kg, i.p.) markedly enhances the hypothermic response produced by a sub-threshold dose of anandamide (5 mg kg, i.p.), although it has no effect on body temperature when injected alone (Fig. 4A) (14). The effect of anandamide plus KDS-413 is mediated by CB 1 receptors, since it is prevented by the CB 1 antagonist rimonabant (27). Notably,
8 KDS TABLE 1. In vitro pharmacology of KDS-413. Effects of KDS-413 in various in vitro receptor binding and enzyme assays Assay Result Receptors Adenosine: A 1 (h), A 2A (h), A 3 (h) Adrenergic: á 1 (non-selective), á 2 (non-selective), â 1 (h), â 2 (h) Angiotensin-II: AT 1 (h), AT 2 (h) Benzodiazepine: BZD (central), BZD (peripheral) Bombesin: BB (non-selective) Bradykinin: B 2 (h) Calcitonin gene-related peptide: CGRP (h) Cannabinoid: CB 1 (h), CB 2 (h) Cholecystokinin: CCK A (h) (CCK 1 ), CCK B (h) (CCK 2 ) Dopamine: D 1 (h), D 2S (h), D 3 (h), D 4 (h), D 5 (h) Endothelin: ET A (h), ET B (h) GABA (non-selective) Galanin: GAL 1 (h), GAL 2 (h) Growth factors, cytokines, chemokines: CXCR 2 (h) (IL-8B), TNF-á (h), CCR 1 (h) Histamine: H 1 (h), H 2 (h) Melanocortin: MC 4 (h) Melatonin: MT 1 Muscarinic: M 1 (h), M 2 (h), M 23 (h), M 4 (h), M 5 (h) Neurokinin: NK 1 (h), NK 2 (h), NK 3 (h) Neuropeptide Y: Y 1 (h), Y 2 (h) Neurotensin: NT 1 (h) (NTS 1 ) piate: ä (h) (DP), ê (KP), ì (h) (MP) (agonist site) rphanin: RL 1 (h) (NP) PACAP: (PAC 1 ) (h) Phencyclidine: PCP Prostanoid: TXA 2 PGH 2 (h) (TP) Purinergic: P 2X,P 2Y Serotonin: 5-HT 1A (h),5-ht 1B, 5-HT 2A (h), 5-HT 2C (h), 5-HT 3 (h), 5-HT 5A (h) (5-ht5A), 5-HT 6 (h), 5-HT 7 (h) ó (non-selective) Somatostatin: Sst (non-selective) Vasopressin: V 1a (h) Vasoactive intestinal Peptide: VIP 1 (h) (VPAC 1 ) Ion Channels Ca 2+ channel (L, verapamil site) (phenylalkylamines) K V channel SK Ca channel Na + channel (site 2) Cl channel
9 28 D. PIMELLI ET AL. Assay TABLE 1 (continued) Result Transporters DA transporter (h) NE transporter (h) 5-HT transporter (h) Anandamide transport (human astrocytoma cells)* Enzymes CX-1 (h), CX-2 (h) Acetylcholinesterase (human and electric eel*) Butyryl cholinesterase (horse plasma)* Monoglyceride lipase (rat brain)* Unless marked with an asterisk (*), assays were performed at CEREP (Paris, France). FR BINDING ASSAYS: Specific ligand binding (radiolabeled reference ligand) to the receptors was determined as the difference between total binding and nonspecific binding in the presence of an excess of unlabelled ligand. Percent (%) inhibition of control specific binding of each reference compound was determined in the presence of KDS-413 (at 1 ìm unless marked with an asterisk. *). For enzymatic assays: CX-1, CX-2 and human acetylcholinesterase assays, enzymatic activity was determined under standard conditions and results were expressed as percent inhibition of control values obtained in the presence of KDS-413 (1 ìm). For binding and enzymatic assays, results showing an inhibition higher than 5% were considered significant. In each experiment, a reference compound was tested concurrently with KDS-413 at several concentrations (for IC 5 determinations) in order to assess assay suitability. Methods and results for anandamide transport, electric eel acetylcholinesterase, butyryl cholinesterase, and MGL (all marked with an asterisk, *) were previously reported in Kathuria et al., 23. h, human;, no significant inhibition. KDS-413 does not further increase the sensitivity to anandamide in FAAH mice (Fig. 4B) (14), supporting the notion that FAAH inhibition plays an exclusive role in mediating the actions of KDS-413. Target Selectivity in Vitro In vitro pharmacology screening showed that KDS-413 does not affect the activities of various serine hydrolases including human and electric-eel acetylcholinesterase, horse plasma butyryl cholinesterase, and rat brain MGL (Table 1). As noted above, the lack of MGL inhibition is particularly important because of the involvement of this enzyme in the biological inactivation of the endocannabinoid 2-AG (13,23,31). In vitro screening for interactions with other members of the endocannabinoid system demonstrated that KDS-413 has no effect on anandamide transport or CB 1 and CB 2 receptor binding (see above) (27). Furthermore, at a test concentration of 1 ìm, KDS-413 was found not to significantly interact with a broad panel of receptors, ion channels, neurotransmitter transporters and enzymes (Table 1), cytochrome P45 isoforms (Table 2), or HERG channel activity (not shown). In a proteomic-based selectivity screen based on the displacement of fluorophosphonate-rhodamine (FPR) from mouse brain protein extracts, KDS-413 was found to prevent FPR binding to triacylglycerol hydrolase (TGH) with an IC 5 of 192 nm
10 KDS TABLE 2. Effects of KDS-413 on cytochrome P45 enzymes activities Assay Result CYP1A2 inhibition (CEC substrate) No significant inhibition CYP2C9 inhibition (MFC substrate) No significant inhibition CYP2C19 inhibition (CEC substrate) No significant inhibition CYP2D6 inhibition (MFC substrate) No significant inhibition CYP3A4 inhibition (BFC substrate) No significant inhibition Activities of human cytochrome P45 enzymes (CYP) were assayed under standard conditions by CEREP (Paris, France) in the absence or presence of KDS-413 (1 ìm). Conversion of each substrate to each corresponding product (see below) was determined by fluorimetry. In each experiment, a reference compound was tested concurrently with KDS-413 at several concentrations (for IC 5 determinations) in order to assess assay suitability. Substrates and detected products were: for CYP1A2, CEC substrate (3-cyano-7-ethoxycoumarin) and CHC product (3-cyano-7-hydroxycoumarin); for CYP2C9, MFC substrate (7-methoxy-4-trifluoromethylcoumarin) and HFC product (7-hydroxy-4- trifluoromethylcoumarin); for CYP2C19, CEC substrate and CHC product; for CYP2D6, MFC substrate and HFC product; and for CYP3A4, BFC substrate [7-benzyloxy-4-trifluoromethylcoumarin] and HFC product. (28). However, direct in vitro measurements of enzyme activity showed that KDS-413 has no effect on either TGH (rat liver) or triacylglycerol lipase (rat white adipose tissue) activity at concentrations as high as 1 ìm. Moreover, in vivo administration of KDS-413 (3 mg kg, i.p.) failed to alter triacylglycerol levels in rat liver and white adipose tissue, although it significantly inhibited FAAH activity in the same tissues (Clapper and Piomelli, unpublished results). Collectively, these results indicate that KDS-413 is remarkably selective for FAAH. Anxiolytic-Like Activity Three lines of evidence suggest that the endocannabinoid system may be involved in the regulation of emotional reactivity. Firstly, CB 1 receptors are expressed at high levels in brain structures, such as the basolateral nucleus of the amygdala, which are implicated in the control of mood and anxiety (15). Secondly, administration of cannabinoid drugs produces marked emotional responses in both experimental animals and humans (25). Finally, pharmacological or genetic disruption of CB 1 receptor activity elicits anxiety-like behaviors in rodents, suggesting the existence of an intrinsic anxiolytic tone mediated by the endocannabinoid system (2,39,41,5). To determine whether KDS-413 affects anxiety-like behaviors, two pharmacologically validated animal models of anxiety were used: the elevated zero maze test and the isolation-induced ultrasonic emission test (which is also utilized to model antidepressantlike drug activity). The elevated zero maze consists of an elevated annular platform with two open and two closed quadrants and is based on the conflict between an animal s instinct to explore its environment and its fear of open spaces, where it may be attacked by predators. Clinically used anxiolytic drugs, such as the benzodiazepines, increase the proportion of time spent in, and the number of entries made into, the open compartments. Similarly, KDS-413 (.5.1 mg kg, i.p.) evoked anxiolytic-like responses at doses that correspond to those required to inhibit FAAH activity in vivo (Fig. 5). Consistent with an
11 3 D. PIMELLI ET AL. A 6 B 8 %Timeopen 4 2 %Timeopen 4 KDS KDS C 6 D 6 %Timeopen 4 2 %Timeopen 4 2 KDS-413 Ri KDS-411 Ri Fig. 5. (A, B) Effects of various doses (mg kg, i.p.) of KDS-413 on anxiety-like behavior (percent time in open quadrant of an elevated zero maze) in rats. (C, D) The CB 1 receptor antagonist rimonabant (Ri, 2 mg kg, i.p.) prevents the anxiolytic-like actions of KDS-413. The carbamate-based FAAH inhibitor, KDS-411, which is weaker than KDS-413 at inhibiting FAAH activity in vitro, also elicited anxiety-like behavior. Modified from ref. 27. involvement of anandamide, the anxiolytic-like effects of the FAAH inhibitor were attenuated by rimonabant (2 mg kg, i.p.) (Fig. 5B). These anxiolytic-like effects were confirmed by testing KDS-413 in the ultrasonic vocalization emission model, which measures the number of stress-induced vocalizations emitted by rat pups removed from their nest. As seen with anxiolytic drugs, KDS-413 (.1 mg kg, i.p.) strongly reduced ultrasonic calls in rat pups and this effect was antagonized by rimonabant (27). Antidepressant-Like Activity The positive responses elicited by KDS-413 in the isolation-induced ultrasonic vocalization test suggested the possibility that FAAH blockade might also result in antidepressant-like effects. To test this idea, the actions of KDS-413 were examined in two validated animal models of antidepressant drug activity the tail suspension test in C57Bl6 mice and the Porsolt s forced swimming test in Wistar rats (18). In the tail suspension test, acute administration of KDS-413 (.3.3 mg kg, i.p.) produced a dose-dependent increase in struggling time, which is characteristic of clinically used antidepressant agents such as desipramine and paroxetine (Fig. 6A). This effect was blocked by rimonabant
12 A Immobility time (sec) URB DES KDS B URB RIM + + Immobility time (sec) C Immobility time (sec) URB URB + + DES 2 RIM + + Fig. 6. Antidepressant-like effects of KDS-413 in the mouse tail-suspension test (TST). (A, C) Effects of KDS-413 (mg kg, i.p.) and desipramine (DES,2 mg kg, i.p.) in the TST after (A) single or (C) repeated administration (once daily for 4 days). (B, D) Single injection of rimonabant (RIM) (1 mg kg, i.p.) prevents the effects of (B) single or (D) repeated URB597 administration. Modified from ref. 18. D Immobility time (sec) (Fig. 6B). Importantly, a 4-day treatment with KDS-413 (.3.3 mg kg, i.p., once daily) caused similar effects, indicating lack of tolerance to the effects of the drug (Fig. 6C, D). In the Porsolt test, acute administration of KDS-413 (.1 mg kg, i.p.) elicited a significant decrease in floating time and a parallel increase in swimming time. An even more marked effect was obtained following a 4-day treatment with KDS-413 (.1 mg kg, i.p., once daily) (18). It is worth noting that the effects of KDS-413 in the Porsolt test resemble those produced by serotonergic antidepressants such as paroxetine, which increase swimming time, rather than noradrenergic antidepressants such as desipramine, which increase struggling time (4). Accordingly, we found that KDS-413 markedly stimulates firing activity in serotonergic neurons of the dorsal raphe nucleus (DRN) of anesthetized rats (18). After acute administration, KDS-413 (.3.3 mg kg, i.v.) produced a dose-dependent increase in mean firing activity of DRN neurons, an effect that was prevented by rimonabant (1 mg kg). Similar results were noted after subchronic administration (.1 mg kg, i.p., once daily for 4 or 21 days). The subchronic effects of KDS-413 were accompanied by augmented serotonin outflow in the hippocampus and the basolateral amygdala, as assessed by in vivo microdialysis in awake rats. Unlike most antidepressants, however, KDS-413, by acute or subchronic administration, did not reduce firing activity in noradrenergic neurons of the locus ceruleus, but rather increased such an activity (18). Collec-
13 32 D. PIMELLI ET AL. tively, the behavioral and electrophysiological experiments outlined above indicate that KDS-413 exerts profound antidepressant-like effects in rats and mice. Analgesic Activity KDS-413 was shown to exert moderate antinociceptive actions in the mouse hot-plate test, which measures the response of the animal to a noxious thermal stimulus. In this model, the compound modestly but significantly lengthened response latencies at a dose of.5 mg kg, i.p. (27). These effects were prevented by the CB 1 antagonist rimonabant (.2 mg kg, i.v.) (27). More marked analgesic effects of acute doses of KDS-413 (.1.3 mg kg, i.p.) were recently observed in the complete Freund s adjuvant model of arthritis pain in the rat (26). These effects were prevented by the CB 1 antagonist AM251, indicating that blockade of FAAH activity may result in CB 1 -mediated antiallodynic effects in an inflammatory pain model (26). Finally, administration of KDS-413 enhanced a form of foot shock-induced analgesia in rats, which is mediated by release of anandamide and 2-AG in the brainstem periaqueductal gray (23). Absence of Cannabinoid-Like Activity and Abuse Liability Though KDS-413 increases brain anandamide levels, this compound does not reproduce the spectrum of pharmacological responses produced by exogenous anandamide or other cannabinoid agonists. Systemic doses of KDS-413 (.3 mg kg, i.p.) that maximally block FAAH activity were found to produce no catalepsy (rigid immobility), hypothermia or hyperphagia, three typical signs of CB 1 receptor activation (27). Even more importantly, KDS-413 was shown to have no effect on two rat models of abuse liability, the conditioned place preference (CPP) test and the drug discrimination (DD) test (18). In the CPP test, acute administration of KDS-413 (.3.3 mg kg, i.p.) produced no preference compared to vehicle or to the direct cannabinoid agonist Win Likewise, in the DD test KDS-413 (.1 3 mg kg, i.p.) did not substitute for the plant-derived cannabinoid Ä 9 -THC or the synthetic cannabinoid agonist Win (Fig. 7). Additional Pharmacological Properties The availability of KDS-413 has prompted a broad range of efforts to investigate the pharmacological properties of FAAH inhibitors. For example, experiments in spontaneously hypertensive rats have shown that by parenteral administration KDS-413 (.1 1 mg kg, i.p.) reduces blood pressure, cardiac contractility, and vascular resistance to levels similar to those found in normotensive animals. These effects were prevented by administration of CB 1 antagonists. Similar responses were observed in two mechanistically distinct models of hypertension Dahl salt-sensitive rats and chronic angiotensin II infusion in rats but not in normotensive control rats. These results suggest that KDS-413 may normalize blood pressure in hypertensive rats by amplifying the cardiodepressor and vasodilator effects of endogenous anandamide and, as such, may offer a novel therapeutic strategy for the treatment of hypertension (2). In another study, KDS-413 (.1 3 mg kg, i.p.) was found to reduce dose-dependently carrageenan-induced edema formation in mice, an effect that was blocked by the CB 2 antagonist SR144528, but not by the CB 1 antagonist AM251 (24). A possible proinflammatory role
14 KDS Ä Time (sec) A URB % THC-Lever Selection B V Dose (mg/kg) C Fig. 7. Motivational profile of KDS-413. (A) Effects of KDS-413 (.3.3 mg kg, i.p., 2 h treatment) in the rat conditioned place preference test. Ä Time: difference in time spent in the non-preferred compartment between post- and pre-conditioning sessions. (B, C) Effects of WIN55,212-2 ( ), Ä 9 -THC ( ), KDS h before test session ( ), and KDS min before test session ( ) in rats trained to discriminate 3 mg kg Ä 9 -THC from vehicle. (B) Percent of responses on the Ä 9 -THC associated lever and (C) rate of lever pressing over the entire 3 min session. pen symbols represent respective vehicles. Modified from ref. 18. Response/sec V Dose (mg/kg) of FAAH is further suggested by the reduced response to inflammatory stimuli observed in FAAH-null mice (9). Cellular Mechanism of Action In vitro experiments show that KDS-413 causes non-metabolized anandamide to accumulate and, eventually, leak out of brain neurons (Fig. 2) (27). In vivo, this action is expected to result in the accumulation of anandamide outside cells and, consequently, in the increased local activation of CB 1 receptors. If anandamide was primarily generated in brain areas engaged in the processing of emotional information, this might help explain the restricted spectrum of actions, and more specifically the lack of cannabinoid-like side effects of KDS-413. This possibility is confirmed by studies, which show that ananda-
15 34 D. PIMELLI ET AL C max (ng/ml) Dose (mg/kg) Fig. 8. Exposure to KDS-413 following oral administration in rats. Animals received KDS-413 as a suspension (.5% carboxymethyl cellulose,.5% simethicone, and.4% polysorbate-8 in water). Exposure is plotted as C max vs. dose. (Kadmus Pharmaceuticals, unpublished results) mide levels in the amygdala and periacqueductal gray matter rise when animals are exposed to stressful stimuli (23,32). PHARMACKINETICS The pharmacokinetic properties of KDS-413 were assessed in rats following oral administration as a suspension. KDS-413 was absorbed at a moderate rate with peak plasma concentrations (C max ) achieved at 1.2 h after treatment. The oral elimination half-life of KDS-413 was approximately 2 h. Linear exposure (Area Under the Curve, AUC, and C max ) of KDS-413 was observed at doses of 1 to 1 mg kg (Fig. 8). In the brain, peak concentrations and AUC values for KDS-413 were similar to those observed in plasma. Maximum brain levels of KDS-413 were reached approximately 1 hour after administration. Accordingly, FAAH inhibition in the brain was rapid ( 1 h), sustained at >9% through 12 h and >6% through 24 h after an oral dose of 1 mg kg (Fig. 9). PRECLINICAL SAFETY EVALUATIN Formal preclinical safety studies to support an Investigational New Drug Application (IND) for KDS-413 have been initiated. To date, single-dose and 7- and 28-day repeated dose studies have been completed in rats; single-dose and 7-day repeated dose studies have been completed in cynomolgus monkeys and a 28-day repeated dose study is ongoing. Monkeys were chosen as the non-rodent species due to limited oral bioavailability in dogs. No signs of systemic toxicity have been noted at single oral doses up to 2 mg kg in rats or 15 mg kg in monkeys. In rats, this dose is at least 4-fold greater than that required to inhibit brain FAAH activity to less than 1% of baseline levels. Considering that plasma exposure at 1 mg kg p.o. is 2-fold greater than the effective
16 KDS Fig. 9. Effects of orally administrated KDS-413 (1 mg kg, p.o.) on rat brain FAAH activity, assessed ex vivo by enzyme assay. (Kadmus Pharmaceuticals, unpublished results) dose, these results suggest a therapeutic index of at least 2. No treatment-related clinical observations were noted during repeated daily dosing at 15 mg kg in rats (28 days) and monkeys (through 21 days to date). Terminal evaluations (following 7 days in monkeys and 28 days in rats) including blood chemistry, hematology, and gross necropsy showed no signs of toxicity. CNS safety pharmacology assessment involving a complete functional observation battery in rats revealed no overt effects following oral doses up to 15 mg kg. No deficits were observed on the rotarod test in rats after treatment with KDS-413 at doses up to 5 mg kg, i.p. (33-fold over the ID 5 for FAAH activity in the brain). Furthermore, in vitro bacterial cytotoxicity and Ames testing yielded negative results. Cardiovascular and respiratory safety pharmacology studies evaluating oral doses of 5, 275, and 15 mg kg will be completed in the first half of 26. CNCLUSINS KDS-413 and its analogs represent a novel class of agents that prevent anandamide deactivation by targeting the intracellular enzymatic activity of FAAH. KDS-413 inhibits FAAH activity with an IC 5 value of approximately 5 nm in rat brain membranes in vitro,.5 nm in intact rat neurons in vitro, 3 nm in human liver microsomes in vitro, and an ID 5 value of.15 mg kg following i.p. administration in the rat. KDS-413 has a remarkable selectivity for FAAH with no activity on other cannabinoid-related targets, including cannabinoid receptors, anandamide transport and monoglycerol lipase (the enzyme involved in the deactivation of the endocannabinoid ester 2-AG). Such target discrimination is matched by a lack of overt cannabimimetic effects in vivo. Thus, at doses that almost abolish FAAH activity and substantially raise brain anandamide levels, KDS-413 does not evoke catalepsy, reduce body temperature or stimulate feeding, three key signs of cannabinoid receptor activation. Moreover, the compound does not display abuse liability in two animal models. KDS-413 does elicit anxiolytic-like, antidepressant-like and analgesic responses, which parallel its ability to inactivate FAAH and are prevented by CB 1 receptor blockade. These findings indicate that KDS-413 acts
17 36 D. PIMELLI ET AL. by enhancing the tonic actions of anandamide on a subset of CB 1 receptors, which may normally be engaged in controlling emotions and pain. In addition, KDS-413 reduces carageenan-induced inflammation in mice and normalizes blood pressure in rat models of hypertension. To date, the ease of large-scale manufacturing, oral bioavailability, and safety profile indicate that KDS-413 is a promising new therapeutic agent for a variety of significant medical conditions, including anxiety, depression and pain. Acknowledgments. We are grateful to all members of the URB597 KDS-413 team in Irvine, Urbino and Parma, as well as to all our collaborators worldwide, for their invaluable contributions to the studies described in this article. Work in the Piomelli lab was funded by grants from the National Institutes of Health, University of California Discovery Program, National Alliance for Research in Schizophrenia and Depression and Kadmus Pharmaceuticals, Inc. Work in Urbino and Parma was funded by grants from the National Institutes of Health, Ministero dell Istruzione, dell Università e della Ricerca, and the University of Urbino Carlo Bo. Conflict of interest statement. Timothy R. Compton, livier Dasse, Edward P. Monaghan, Jeff A. Parrott and David Putman are employees of, and Daniele Piomelli is a consultant for Kadmus Pharmaceuticals Inc., the developer of KDS-413. Addendum. Chemical names. AM251, N-(Piperidin-1-yl)-5-(4-iodophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1Hpyrazole-3-carboxamide; SR144528, N-[1S]-endo-1,3,3-[trimethylbicyclo[2.2.1]heptan-2-yl]-5-(4-chloro-3-methylphenyl)-1-(4-methylbenzyl)-pyrazol 3-carboxamide; Win , (R)-(+)-[2.3-Dihydro-5-methyl-3-(4-morpholino-metyl)pyrrolo[1,2,3- de]-1,4-benzoxazin-6-yl]-1-naphthalenylmethanone mesylate. REFERENCES 1. Basso E, Duranti, A., Mor, M., et al. Tandem mass spectrometric data-faah inhibitory activity relationships of some carbamic acid -aryl esters. J Mass Spectrom 24;39: Batkai S, Pacher P, sei-hyiaman D, et al. Endocannabinoids acting at cannabinoid-1 receptors regulate cardiovascular function in hypertension. Circulation 24;11: Beltramo M, di Tomaso E, and Piomelli D. Inhibition of anandamide hydrolysis in rat brain tissue by (E)-6- (bromomethylene) tetrahydro-3-(1-naphthalenyl)-2h-pyran-2-one. FEBS Lett 1997;43: Beltramo M, Stella N, Calignano A, Lin SY, Makriyannis A, Piomelli D. Functional role of high-affinity anandamide transport, as revealed by selective inhibition. Science 1997;277: Boger DL, Sato H, Lerner AE, et al. Exceptionally potent inhibitors of fatty acid amide hydrolase: The enzyme responsible for degradation of endogenous oleamide and anandamide. Proc Natl Acad Sci USA 2; 97: Calignano A, La Rana G, Giuffrida A, Piomelli D. Control of pain initiation by endogenous cannabinoids. Nature 1998;394: Cravatt BF, Giang DK, Mayfield SP, Boger DL, Lerner RA, Gilula NB. Molecular characterization of an enzyme that degrades neuromodulatory fatty-acid amides. Nature 1996;384: Cravatt BF, Demarest K, Patricelli MP, et al. Supersensitivity to anandamide and enhanced endogenous cannabinoid signaling in mice lacking fatty acid amide hydrolase. Proc Natl Acad Sci USA 21;98:
18 KDS Cravatt BF, Saghatelian A, Hawkins EG, Clement AB, Bracey MH, Lichtman AH. Functional disassociation of the central and peripheral fatty acid amide signaling systems. Proc Natl Acad Sci USA 24;11: Désarnaud F, Cadas H, Piomelli D. Anandamide amidohydrolase activity in rat brain microsomes. Identification and partial characterization. J Biol Chem 1995;27: Devane WA, Hanus L, Breuer A, et al. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 1992;258: Di Marzo V, Fontana A, Cadas H, et al. Formation and inactivation of endogenous cannabinoid anandamide in central neurons. Nature 1994;372: Dinh TP, Freund TF, Piomelli D. A role for monoglyceride lipase in 2-arachidonoylglycerol inactivation. Chem Phys Lipids 22;121: Fegley D, Gaetani S, Duranti A, et al. Characterization of the fatty acid amide hydrolase inhibitor cyclohexyl carbamic acid 3 -carbamoyl-biphenyl-3-yl ester (URB597): Effects on anandamide and oleoylethanolamide deactivation. J Pharmacol Exp Ther 25;313: Freund TF, Katona I, Piomelli D. Role of endogenous cannabinoids in synaptic signaling. Physiol Rev 23;83: Fu J, Gaetani S, veisi F, Lo Verme J, et al. leylethanolamide regulates feeding and body weight through activation of the nuclear receptor PPAR-alpha. Nature 23;425: Giuffrida A, Parsons LH, Kerr TM, Rodríguez de Fonseca F, Navarro M, Piomelli D. Dopamine activation of endogenous cannabinoid signaling in dorsal striatum. Nat Neurosci 1999;2: Gobbi G, Bambico FR, Mangieri R, et al. Antidepressant-like activity and modulation of brain monoaminergic transmission by blockade of anandamide hydrolysis. Proc Natl Acad Sci USA 25;12: Guzman M, Lo Verme J, veisi F, Blazquez C, Piomelli D. leoylethanolamide stimulates lipolysis by activating the nuclear receptor PPAR-alpha. J Biol Chem 24;E-publication April Haller J, Varga B, Ledent C, Barna I, Freund TF. Context-dependent effects of CB 1 cannabinoid gene disruption on anxiety-like and social behaviour in mice. Eur J Neurosci 24;19: Hillard CJ, Wilkison DM, Edgemond WS, Campbell WB. Characterization of the kinetics and distribution of N-arachidonylethanolamine (anandamide) hydrolysis by rat brain. Biochim Biophys Acta 1995;1257: Hillard CJ, Campbell WB. Biochemistry and pharmacology of arachidonylethanolamide, a putative endogenous cannabinoid. J Lipid Res 1997;38: Hohmann AG, Suplita RL, Bolton NM, et al. An endocannabinoid mechanism for stress-induced analgesia. Nature 25;435: Holt S, Comelli F, Costa B, Fowler CJ. Inhibitors of fatty acid amide hydrolase reduce carrageenan-induced hind paw inflammation in pentobarbital-treated mice: Comparison with indomethacin and possible involvement of cannabinoid receptors. Br J Pharmacol 25;146: Iversen LL. The science of marijuana. xford: University Press, Jayamanne A, Greenwood R, Mitchell VA, Aslan S, Piomelli D, Vaughan CW. Actions of the FAAH inhibitor URB597 in neuropathic and inflammatory chronic pain models. Br J Pharmacol 25;147: Kathuria S, Gaetani S, Fegley D, et al. Modulation of anxiety through blockade of anandamide hydrolysis. Nat Med 23;9: Lichtman AH, Leung D, Shelton C, et al. Reversible inhibitors of fatty acid amide hydrolase that promote analgesia: Evidence for an unprecedented combination of potency and selectivity. J Pharmacol Exp Ther 24;311: Lodola A, Mor M, Hermann JC, Tarzia G, Piomelli D, Mulholland AJ. QM MM modelling of oleamide hydrolysis in fatty acid amide hydrolase (FAAH) reveals a new mechanism of nucleophile activation. Chem Commun (Camb) 25: LoVerme J, Fu J, Astarita G, et al. The nuclear receptor peroxisome proliferator-activated receptor-alpha mediates the anti-inflammatory actions of palmitoylethanolamide. Mol Pharmacol 25;67: Makara JK, Mor M, Fegley D, et al. Selective inhibition of 2-AG hydrolysis enhances endocannabinoid signaling in hippocampus. Nat Neurosci 25;8: Marsicano G, Wotjak CT, Azad SC, et al. The endogenous cannabinoid system controls extinction of aversive memories. Nature 22;418: Mazzari S, Canella R, Petrelli L, Marcolongo G, Leon A. N-(2-hydroxyethyl)hexadecanamide is orally active in reducing edema formation and inflammatory hyperalgesia by down-modulating mast cell activation. Eur J Pharmacol 1996;3: McKinney MK, Cravatt BF. Structure and function of fatty acid amide hydrolase. Annu Rev Biochem 24;74:
19 38 D. PIMELLI ET AL. 35. Mechoulam R, Ben-Shabat S, Hanus L, et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem Pharmacol 1995;5: Moore SA, Nomikos GG, Dickason-Chesterfield AK, et al. Identification of a high-affinity binding site involved in the transport of endocannabinoids. Proc Natl Acad Sci USA 25;12: Mor M, Rivara S, Lodola A, et al. Cyclohexylcarbamic acid 3 -or4 -substituted biphenyl-3-yl esters as fatty acid amide hydrolase inhibitors: Synthesis, quantitative structure-activity relationships, and molecular modeling studies. J Med Chem 24;47: Munro S, Thomas KL, Abu-Shaar M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993;365: Navarro M, Hernández E, Muñoz RM, et al. Acute administration of the CB 1 cannabinoid receptor antagonist SR A induces anxiety-like responses in the rat. Neuroreport 1997;8: Page ME, Detke MJ, Dalvi A, Kirby LG, Lucki I. Serotonergic mediation of the effects of fluoxetine, but not desipramine, in the rat forced swimming test. Psychopharmacology (Berl) 1999;147: Patel S, Roelke CT, Rademacher DJ, Hillard CJ. Inhibition of restraint stress-induced neural and behavioural activation by endogenous cannabinoid signalling. Eur J Neurosci 25;21: Piomelli D. The molecular logic of endocannabinoid signalling. Nat Rev Neurosci 23;4: Rodríguez de Fonseca F, Navarro M, Gómez R, et al. An anorexic lipid mediator regulated by feeding. Nature 21;414: Schmid PC, Zuzarte-Augustin ML, Schmid HH. Properties of rat liver N-acylethanolamine amidohydrolase. J Biol Chem 1985;26: Stella N, Schweitzer P, Piomelli D. A second endogenous cannabinoid that modulates long-term potentiation. Nature 1997;388: Sugiura T, Kondo S, Sukagawa A, et al. 2-Arachidonoylglycerol: A possible endogenous cannabinoid receptor ligand in brain. Biochem Biophys Res Commun 1995;215: Tarzia G, Duranti A, Tontini A, et al. Synthesis and structure-activity relationships of a series of pyrrole cannabinoid receptor agonists. Bioorg Med Chem 23;11: Tarzia G, Duranti A., Tontini A., et al. Synthesis and structure-activity relationships of FAAH inhibitors: Cyclohexylcarbamic acid biphenyl esters with chemical modulation at the proximal phenyl ring. Chem Med Chem 25;6: Ueda N, Yamamoto K, Yamamoto S, et al. Lipoxygenase-catalyzed oxygenation of arachidonylethanolamide, a cannabinoid receptor agonist. Biochim Biophys Acta 1995;1254: Uriguen L, Perez-Rial S, Ledent C, Palomo T, Manzanares J. Impaired action of anxiolytic drugs in mice deficient in cannabinoid CB 1 receptors. Neuropharmacology 24;46: Van Sickle MD, Duncan M, Kingsley PJ, et al. Identification and functional characterization of brainstem cannabinoid CB 2 receptors. Science 25;31:
Cannabinoid Therapeutics: Marijuana and Beyond Chair: Igor Grant, M.D.
 Cannabinoid Therapeutics: Marijuana and Beyond Chair: Igor Grant, M.D. Advances in understanding the molecular mechanisms underlying cannabinoid effects, coupled with increased public acceptance that cannabinoids
Cannabinoid Therapeutics: Marijuana and Beyond Chair: Igor Grant, M.D. Advances in understanding the molecular mechanisms underlying cannabinoid effects, coupled with increased public acceptance that cannabinoids
Antidepressant-like activity and modulation of brain monoaminergic transmission by blockade of anandamide hydrolysis
 Antidepressant-like activity and modulation of brain monoaminergic transmission by blockade of anandamide hydrolysis G. Gobbi*, F. R. Bambico*, R. Mangieri, M. Bortolato, P. Campolongo, M. Solinas, T.
Antidepressant-like activity and modulation of brain monoaminergic transmission by blockade of anandamide hydrolysis G. Gobbi*, F. R. Bambico*, R. Mangieri, M. Bortolato, P. Campolongo, M. Solinas, T.
JNJ : selective and slowly reversible FAAH inhibitor. Central and Peripheral PK/PD
 JNJ-42165279: selective and slowly reversible FAAH inhibitor Central and Peripheral PK/PD The Endocannabinoid System Research initiated by efforts to elucidate the active substance of Cannabis (THC in
JNJ-42165279: selective and slowly reversible FAAH inhibitor Central and Peripheral PK/PD The Endocannabinoid System Research initiated by efforts to elucidate the active substance of Cannabis (THC in
marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD
 marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD in this talk what is marijuana? the brain on marijuana is the teen brain special? current research what is marijuana?
marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD in this talk what is marijuana? the brain on marijuana is the teen brain special? current research what is marijuana?
UC Irvine UC Irvine Previously Published Works
 UC Irvine UC Irvine Previously Published Works Title Carrier-mediated transport and enzymatic hydrolysis of the endogenous cannabinoid 2- arachidonylglycerol Permalink https://escholarship.org/uc/item/92w2j8v2
UC Irvine UC Irvine Previously Published Works Title Carrier-mediated transport and enzymatic hydrolysis of the endogenous cannabinoid 2- arachidonylglycerol Permalink https://escholarship.org/uc/item/92w2j8v2
Characterization of the fatty-acid amide hydrolase inhibitor URB597: Effects on
 JPET Fast This Forward. article has not Published been copyedited on and December formatted. The 3, final 2004 version as DOI:10.1124/jpet.104.078980 may differ from this version. JPET # 78980 Characterization
JPET Fast This Forward. article has not Published been copyedited on and December formatted. The 3, final 2004 version as DOI:10.1124/jpet.104.078980 may differ from this version. JPET # 78980 Characterization
Cannabinoids 101. Matthew Hill, Ph.D. Hotchkiss Brain Institute University of Calgary
 Cannabinoids 101 Matthew Hill, Ph.D. Hotchkiss Brain Institute University of Calgary Disclosures Have received honoraria for Scientific Consultation: Pfizer International GW Pharmaceuticals Receive operating
Cannabinoids 101 Matthew Hill, Ph.D. Hotchkiss Brain Institute University of Calgary Disclosures Have received honoraria for Scientific Consultation: Pfizer International GW Pharmaceuticals Receive operating
Review of Neurochemistry What are neurotransmitters?
 Review of Neurochemistry What are neurotransmitters? In molecular terms, neurotransmitters are molecules that ( ) and of neurons by, for example, increasing or decreasing enzymatic activity or altering
Review of Neurochemistry What are neurotransmitters? In molecular terms, neurotransmitters are molecules that ( ) and of neurons by, for example, increasing or decreasing enzymatic activity or altering
Classes of Neurotransmitters. Neurotransmitters
 1 Drugs Outline 2 Neurotransmitters Agonists and Antagonists Cocaine & other dopamine agonists Alcohol & its effects / Marijuana & its effects Synthetic & Designer Drugs: Ecstasy 1 Classes of Neurotransmitters
1 Drugs Outline 2 Neurotransmitters Agonists and Antagonists Cocaine & other dopamine agonists Alcohol & its effects / Marijuana & its effects Synthetic & Designer Drugs: Ecstasy 1 Classes of Neurotransmitters
marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD
 marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD in this talk what is marijuana? the brain on marijuana is the teen brain special? current research what is marijuana?
marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD in this talk what is marijuana? the brain on marijuana is the teen brain special? current research what is marijuana?
The Endocannabinoid System
 The Endocannabinoid System Think of this system as the wizard behind the curtain. It underlies all the other systems, including the Autonomic Nervous System. Overall it influences well-being and the perception
The Endocannabinoid System Think of this system as the wizard behind the curtain. It underlies all the other systems, including the Autonomic Nervous System. Overall it influences well-being and the perception
Pathophysiology of Pain
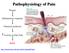 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Zoektocht naar innovatieve geneesmiddelen voor de toekomst - Verslaving en ander gedrag in moleculair perspectief
 Zoektocht naar innovatieve geneesmiddelen voor de toekomst - Verslaving en ander gedrag in moleculair perspectief Prof. dr. Chris Kruse Swammerdam Institute, UvA Solvay Pharmaceuticals Drug Discovery Assets
Zoektocht naar innovatieve geneesmiddelen voor de toekomst - Verslaving en ander gedrag in moleculair perspectief Prof. dr. Chris Kruse Swammerdam Institute, UvA Solvay Pharmaceuticals Drug Discovery Assets
Cannabinoids and Mental Health
 Cannabinoids and Mental Health https://upload.wikimedia.org/wikipedia/commons Karen M. Lounsbury, PhD Professor of Pharmacology 802-656-3231, Karen.lounsbury@uvm.edu Objectives Describe the underlying
Cannabinoids and Mental Health https://upload.wikimedia.org/wikipedia/commons Karen M. Lounsbury, PhD Professor of Pharmacology 802-656-3231, Karen.lounsbury@uvm.edu Objectives Describe the underlying
marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD
 marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD in this talk what is marijuana? the brain on marijuana is the teen brain special? current research what is marijuana?
marijuana and the teen brain MARY ET BOYLE, PH. D. DEPARTMENT OF COGNITIVE SCIENCE UCSD in this talk what is marijuana? the brain on marijuana is the teen brain special? current research what is marijuana?
Getting into the weed: the endocannabinoid system of the gut-brain axis in energy homeostasis. Keith Sharkey
 Getting into the weed: the endocannabinoid system of the gut-brain axis in energy homeostasis Keith Sharkey Department of Physiology & Pharmacology University of Calgary Dr. Keith Sharkey Financial Interest
Getting into the weed: the endocannabinoid system of the gut-brain axis in energy homeostasis Keith Sharkey Department of Physiology & Pharmacology University of Calgary Dr. Keith Sharkey Financial Interest
Na + /Cl - -dependent Neurotransmitter Transporters. Outline. Synaptic Transmission. How did the neurotransmitter cross the membrane?
 Na /Cl - -dependent Neurotransmitter Transporters Stine Meinild Lundby The Neurobiology Group Molecular, Cellular, and Integrative Physiology Section November 26 th, 2007 slide 1 Outline Synaptic neurotransmission
Na /Cl - -dependent Neurotransmitter Transporters Stine Meinild Lundby The Neurobiology Group Molecular, Cellular, and Integrative Physiology Section November 26 th, 2007 slide 1 Outline Synaptic neurotransmission
Insights into the mechanism and inhibition of fatty acid amide hydrolase from quantum mechanics/molecular mechanics (QM/MM) modelling
 Enzyme Mechanisms: Fast Reaction and Computational Approaches 363 Insights into the mechanism and inhibition of fatty acid amide hydrolase from quantum mechanics/molecular mechanics (QM/MM) modelling Alessio
Enzyme Mechanisms: Fast Reaction and Computational Approaches 363 Insights into the mechanism and inhibition of fatty acid amide hydrolase from quantum mechanics/molecular mechanics (QM/MM) modelling Alessio
Fundamentals of Pharmacology
 Fundamentals of Pharmacology Topic Page Receptors 2 Ion channels / GABA 4 GPCR s 6 TK receptors 8 Basics of PK 11 ADR s / Clinical study design 13 Introduction to the ANS 16 Cholinergic Pharmacology 20
Fundamentals of Pharmacology Topic Page Receptors 2 Ion channels / GABA 4 GPCR s 6 TK receptors 8 Basics of PK 11 ADR s / Clinical study design 13 Introduction to the ANS 16 Cholinergic Pharmacology 20
The Scientific Side of Medical Marijuana
 The Scientific Side of Medical Marijuana Ken Mackie, MD Indiana University Bloomington, IN December 3, 2009 kmackie@indiana.edu Financial disclosures NIH (NIDA) - research grants Alzheimer s Association
The Scientific Side of Medical Marijuana Ken Mackie, MD Indiana University Bloomington, IN December 3, 2009 kmackie@indiana.edu Financial disclosures NIH (NIDA) - research grants Alzheimer s Association
Basics of Pharmacology
 Basics of Pharmacology Pekka Rauhala Transmed 2013 What is pharmacology? Pharmacology may be defined as the study of the effects of drugs on the function of living systems Pharmacodynamics The mechanism(s)
Basics of Pharmacology Pekka Rauhala Transmed 2013 What is pharmacology? Pharmacology may be defined as the study of the effects of drugs on the function of living systems Pharmacodynamics The mechanism(s)
Regulation of Inflammatory Pain by Inhibition of Fatty Acid Amide Hydrolase (FAAH)
 JPET This Fast article Forward. has not been Published copyedited and on formatted. April 7, The 2010 final as version DOI:10.1124/jpet.109.164806 may differ from this version. Title page Regulation of
JPET This Fast article Forward. has not been Published copyedited and on formatted. April 7, The 2010 final as version DOI:10.1124/jpet.109.164806 may differ from this version. Title page Regulation of
Supplementary Material for Pacholec, M. et al. manuscript SRT1720, SRT2183, SRT1460, and
 Supplementary Material for Pacholec, M. et al. manuscript SRT1720, SRT2183, SRT1460, and resveratrol are not direct activators of SIRT1 Supplementary Figure Legends Figure S1. Determination of SIRT1 K
Supplementary Material for Pacholec, M. et al. manuscript SRT1720, SRT2183, SRT1460, and resveratrol are not direct activators of SIRT1 Supplementary Figure Legends Figure S1. Determination of SIRT1 K
Fatty Acid Amide Hydrolase Inhibitor Screening Assay Kit
 Fatty cid mide Hydrolase Inhibitor Screening ssay Kit Item No. 10005196 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd nn rbor, MI US TBLE OF CONTENTS
Fatty cid mide Hydrolase Inhibitor Screening ssay Kit Item No. 10005196 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd nn rbor, MI US TBLE OF CONTENTS
Reversible Inhibitors of Fatty Acid Amide Hydrolase That Promote Analgesia: Evidence for an Unprecedented Combination of Potency and Selectivity
 0022-3565/04/3112-441 448$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 311, No. 2 Copyright 2004 by The American Society for Pharmacology and Experimental Therapeutics 69401/1174177
0022-3565/04/3112-441 448$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 311, No. 2 Copyright 2004 by The American Society for Pharmacology and Experimental Therapeutics 69401/1174177
Neurotransmitter Systems III Neurochemistry. Reading: BCP Chapter 6
 Neurotransmitter Systems III Neurochemistry Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important
Neurotransmitter Systems III Neurochemistry Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important
PHRM20001: Pharmacology - How Drugs Work!
 PHRM20001: Pharmacology - How Drugs Work Drug: a chemical that affects physiological function in a specific way. Endogenous substances: hormones, neurotransmitters, antibodies, genes. Exogenous substances:
PHRM20001: Pharmacology - How Drugs Work Drug: a chemical that affects physiological function in a specific way. Endogenous substances: hormones, neurotransmitters, antibodies, genes. Exogenous substances:
Synergy between enzyme inhibitors of fatty acid amide hydrolase and cyclooxygenase in. visceral nociception*
 JPET Fast Forward. Published on January 9, 2009 as DOI:10.1124/jpet.108.143487 JPET 143487 Synergy between enzyme inhibitors of fatty acid amide hydrolase and cyclooxygenase in visceral nociception* Pattipati
JPET Fast Forward. Published on January 9, 2009 as DOI:10.1124/jpet.108.143487 JPET 143487 Synergy between enzyme inhibitors of fatty acid amide hydrolase and cyclooxygenase in visceral nociception* Pattipati
Bioorganic & Medicinal Chemistry Letters 15 (2005)
 Bioorganic & Medicinal Chemistry Letters 15 (2005) 1423 1428 Discovery of an exceptionally potent and selective class of fatty acid amide hydrolase inhibitors enlisting proteome-wide selectivity screening:
Bioorganic & Medicinal Chemistry Letters 15 (2005) 1423 1428 Discovery of an exceptionally potent and selective class of fatty acid amide hydrolase inhibitors enlisting proteome-wide selectivity screening:
Inhibition of Fatty-Acid Amide Hydrolase Accelerates Acquisition and Extinction Rates in a Spatial Memory Task
 (2007) 32, 1032 1041 & 2007 Nature Publishing Group All rights reserved 0893-133X/07 $30.00 www.neuropsychopharmacology.org Inhibition of Fatty-Acid Amide Hydrolase Accelerates Acquisition and Extinction
(2007) 32, 1032 1041 & 2007 Nature Publishing Group All rights reserved 0893-133X/07 $30.00 www.neuropsychopharmacology.org Inhibition of Fatty-Acid Amide Hydrolase Accelerates Acquisition and Extinction
History Of Medical Cannabis
 History Of Medical Cannabis Historical and archaeological evidence of widespread use in ancient times as medicine, food, textiles & for sacraments, rituals Possibly first domesticated crop Introduction
History Of Medical Cannabis Historical and archaeological evidence of widespread use in ancient times as medicine, food, textiles & for sacraments, rituals Possibly first domesticated crop Introduction
Cannabis. Member of the Cannabaceae family of flowering plants (along with hops) Cannabis sativa (v. sativa, indica, afghanica, ruderalis)
 Member of the Cannabaceae family of flowering plants (along with hops) sativa (v. sativa, indica, afghanica, ruderalis) Only females flowers contain high concentrations of psychoactive oils (cannabinoids)
Member of the Cannabaceae family of flowering plants (along with hops) sativa (v. sativa, indica, afghanica, ruderalis) Only females flowers contain high concentrations of psychoactive oils (cannabinoids)
Neurotransmitter Systems I Identification and Distribution. Reading: BCP Chapter 6
 Neurotransmitter Systems I Identification and Distribution Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the
Neurotransmitter Systems I Identification and Distribution Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the
 PSY 302 Lecture 6: The Neurotransmitters (continued) September 12, 2017 Notes by: Desiree Acetylcholine (ACh) CoA + Acetate Acetyl-CoA (mitochondria) (food, vinegar) + Choline ChAT CoA + ACh (lipids, foods)
PSY 302 Lecture 6: The Neurotransmitters (continued) September 12, 2017 Notes by: Desiree Acetylcholine (ACh) CoA + Acetate Acetyl-CoA (mitochondria) (food, vinegar) + Choline ChAT CoA + ACh (lipids, foods)
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Neural Communication. Central Nervous System Peripheral Nervous System. Communication in the Nervous System. 4 Common Components of a Neuron
 Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
JPET The endogenous cannabinoid anandamide produces THC-like discriminative and
 JPET Fast This article Forward. has not been Published copyedited on and January formatted. The 8, 2007 final version as DOI:10.1124/jpet.106.114124 may differ from this version. The endogenous cannabinoid
JPET Fast This article Forward. has not been Published copyedited on and January formatted. The 8, 2007 final version as DOI:10.1124/jpet.106.114124 may differ from this version. The endogenous cannabinoid
Pharmacology for CHEMISTS
 Pharmacology for CHEMISTS JOSEPH G. CANNON ACS Professional Reference Book AMERICAN CHEMICAL SOCIETY OXFORD UNIVERSITY PRESS Washington D.C. New York Oxford 1999 CONTENTS I Chemical and Biological Bases
Pharmacology for CHEMISTS JOSEPH G. CANNON ACS Professional Reference Book AMERICAN CHEMICAL SOCIETY OXFORD UNIVERSITY PRESS Washington D.C. New York Oxford 1999 CONTENTS I Chemical and Biological Bases
Inactivation of N-Acyl Phosphatidylethanolamine Phospholipase D Reveals Multiple Mechanisms for the Biosynthesis of Endocannabinoids
 4720 Biochemistry 2006, 45, 4720-4726 Articles Inactivation of N-Acyl Phosphatidylethanolamine Phospholipase D Reveals Multiple Mechanisms for the Biosynthesis of Endocannabinoids Donmienne Leung, Alan
4720 Biochemistry 2006, 45, 4720-4726 Articles Inactivation of N-Acyl Phosphatidylethanolamine Phospholipase D Reveals Multiple Mechanisms for the Biosynthesis of Endocannabinoids Donmienne Leung, Alan
The fatty acid amide hydrolase inhibitor URB 597: interactions with anandamide in rhesus monkeys
 655..666 BJP British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01388.x www.brjpharmacol.org RESEARCH PAPERbph_1388 The fatty acid amide hydrolase inhibitor URB 597: interactions with anandamide
655..666 BJP British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01388.x www.brjpharmacol.org RESEARCH PAPERbph_1388 The fatty acid amide hydrolase inhibitor URB 597: interactions with anandamide
The fatty acid amide hydrolase (FAAH) inhibitor PF-3845 acts in the nervous system to reverse LPS-induced tactile allodynia in mice
 2485..2496 BJP British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01445.x www.brjpharmacol.org Themed Section: Cannabinoids in Biology and Medicine, Part II RESEARCH PAPERbph_1445 The fatty acid
2485..2496 BJP British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01445.x www.brjpharmacol.org Themed Section: Cannabinoids in Biology and Medicine, Part II RESEARCH PAPERbph_1445 The fatty acid
(12) United States Patent Cravatt et a].
![(12) United States Patent Cravatt et a]. (12) United States Patent Cravatt et a].](/thumbs/92/109287340.jpg) US008772318B2 (12) United States Patent Cravatt et a]. (10) Patent N0.: (45) Date of Patent: US 8,772,318 B2 Jul. 8, 2014 (54) (75) (73) (21) (22) (86) (87) (65) (60) (51) (52) (58) METHODS AND COMPOSITIONS
US008772318B2 (12) United States Patent Cravatt et a]. (10) Patent N0.: (45) Date of Patent: US 8,772,318 B2 Jul. 8, 2014 (54) (75) (73) (21) (22) (86) (87) (65) (60) (51) (52) (58) METHODS AND COMPOSITIONS
Chemical Control of Behavior and Brain 1 of 9
 Chemical Control of Behavior and Brain 1 of 9 I) INTRO A) Nervous system discussed so far 1) Specific 2) Fast B) Other systems extended in space and time 1) Nonspecific 2) Slow C) Three components that
Chemical Control of Behavior and Brain 1 of 9 I) INTRO A) Nervous system discussed so far 1) Specific 2) Fast B) Other systems extended in space and time 1) Nonspecific 2) Slow C) Three components that
NIH Public Access Author Manuscript J Psychopharmacol. Author manuscript; available in PMC 2014 June 16.
 NIH Public Access Author Manuscript Published in final edited form as: J Psychopharmacol. 2013 June ; 27(6): 564 571. doi:10.1177/0269881113477710. AM404 attenuates reinstatement of nicotine seeking induced
NIH Public Access Author Manuscript Published in final edited form as: J Psychopharmacol. 2013 June ; 27(6): 564 571. doi:10.1177/0269881113477710. AM404 attenuates reinstatement of nicotine seeking induced
NIH Public Access Author Manuscript CNS Neurol Disord Drug Targets. Author manuscript; available in PMC 2010 June 1.
 NIH Public Access Author Manuscript Published in final edited form as: CNS Neurol Disord Drug Targets. 2009 December ; 8(6): 403 421. The Endocannabinoid System and Pain Josée Guindon and Andrea G. Hohmann
NIH Public Access Author Manuscript Published in final edited form as: CNS Neurol Disord Drug Targets. 2009 December ; 8(6): 403 421. The Endocannabinoid System and Pain Josée Guindon and Andrea G. Hohmann
NEW STRATEGIES FOR THE TREATMENT OF MOOD DISORDERS: THE TRIPLE REUPTAKE INHIBITORS
 Send Orders for Reprints to reprints@benthamscience.net 234 Neurobiology of Mood Disorders, 2013, 234-253 NEW STRATEGIES FOR THE TREATMENT OF MOOD DISORDERS: THE TRIPLE REUPTAKE INHIBITORS GAËL QUESSEVEUR,
Send Orders for Reprints to reprints@benthamscience.net 234 Neurobiology of Mood Disorders, 2013, 234-253 NEW STRATEGIES FOR THE TREATMENT OF MOOD DISORDERS: THE TRIPLE REUPTAKE INHIBITORS GAËL QUESSEVEUR,
Drug Receptor Interactions and Pharmacodynamics
 Drug Receptor Interactions and Pharmacodynamics Dr. Raz Mohammed MSc Pharmacology School of Pharmacy 22.10.2017 Lec 6 Pharmacodynamics definition Pharmacodynamics describes the actions of a drug on the
Drug Receptor Interactions and Pharmacodynamics Dr. Raz Mohammed MSc Pharmacology School of Pharmacy 22.10.2017 Lec 6 Pharmacodynamics definition Pharmacodynamics describes the actions of a drug on the
Neurophysiology and Neurochemistry in PsychoGeriatrics
 Tel Aviv University Sackler Faculty of Medicine CME in Psychiatry Neurophysiology and Neurochemistry in PsychoGeriatrics Nicola Maggio, MD, PhD Sackler Faculty of Medicine Tel Aviv University Department
Tel Aviv University Sackler Faculty of Medicine CME in Psychiatry Neurophysiology and Neurochemistry in PsychoGeriatrics Nicola Maggio, MD, PhD Sackler Faculty of Medicine Tel Aviv University Department
A New Era of Migraine Management: The Challenging Landscape in Prevention
 Provided by MediCom Worldwide, Inc. Supported by an educational grant from Teva Pharmaceuticals What is a Neuropeptide? Small chains of amino acids released by neural cells (neurons or glial cells) to
Provided by MediCom Worldwide, Inc. Supported by an educational grant from Teva Pharmaceuticals What is a Neuropeptide? Small chains of amino acids released by neural cells (neurons or glial cells) to
Effects of Alterations in Cannabinoid Signaling, Alone and in Combination with Morphine, on Pain-Elicited and Pain-Suppressed Behavior in Mice
 1521-0103/12/3421-177 187$25.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 342, No. 1 Copyright 2012 by The American Society for Pharmacology and Experimental Therapeutics 191478/3778300
1521-0103/12/3421-177 187$25.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 342, No. 1 Copyright 2012 by The American Society for Pharmacology and Experimental Therapeutics 191478/3778300
processes in the central nervous system (CNS), affecting many of the during the course of ethanol treatment. Ethanol stimulates the release of
 INTRODUCTION INTRODUCTION Neuroscience research is essential for understanding the biological basis of ethanol-related brain alterations and for identifying the molecular targets for therapeutic compounds
INTRODUCTION INTRODUCTION Neuroscience research is essential for understanding the biological basis of ethanol-related brain alterations and for identifying the molecular targets for therapeutic compounds
Endocannabinoid System Cannabinoid Drugs
 Cannabinoid Drugs STCM Conference Bern 22.1.2013 Rudolf Brenneisen University of Bern, DKF 1 www.phytopharm.dkf.unibe.ch rudolf.brenneisen@dkf.unibe.ch Milestones 1988: First cannabinoid receptor (rat
Cannabinoid Drugs STCM Conference Bern 22.1.2013 Rudolf Brenneisen University of Bern, DKF 1 www.phytopharm.dkf.unibe.ch rudolf.brenneisen@dkf.unibe.ch Milestones 1988: First cannabinoid receptor (rat
Mice lacking fatty acid amide hydrolase exhibit a cannabinoid receptor-mediated phenotypic hypoalgesia
 Pain 109 (2004) 319 327 www.elsevier.com/locate/pain Mice lacking fatty acid amide hydrolase exhibit a cannabinoid receptor-mediated phenotypic hypoalgesia Aron H. Lichtman a, *, Christopher C. Shelton
Pain 109 (2004) 319 327 www.elsevier.com/locate/pain Mice lacking fatty acid amide hydrolase exhibit a cannabinoid receptor-mediated phenotypic hypoalgesia Aron H. Lichtman a, *, Christopher C. Shelton
BACKGROUND AND PURPOSE
 2539..2548 BJP British Journal of Pharmacology DOI:1.1111/j.1476-5381.211.1467.x www.brjpharmacol.org Themed Section: Cannabinoids in Biology and Medicine, Part II RESEARCH PAPERbph_1467 The anandamide
2539..2548 BJP British Journal of Pharmacology DOI:1.1111/j.1476-5381.211.1467.x www.brjpharmacol.org Themed Section: Cannabinoids in Biology and Medicine, Part II RESEARCH PAPERbph_1467 The anandamide
NEURAL MECHANISMS OF SLEEP (p.1) (Rev. 3/21/07)
 NEURAL MECHANISMS OF SLEEP (p.1) (Rev. 3/21/07) 1. Revisitation of Bremer s 1936 Isolated Brain Studies Transected the brain: a. Cut between the medulla and the spinal cord ( encephale isole ) Note: recall
NEURAL MECHANISMS OF SLEEP (p.1) (Rev. 3/21/07) 1. Revisitation of Bremer s 1936 Isolated Brain Studies Transected the brain: a. Cut between the medulla and the spinal cord ( encephale isole ) Note: recall
Synaptic transmission
 Outline Synaptic transmission Sompol Tapechum M.D., Ph.D. Department of Physiology Faculty of Medicine Siriraj Hospital, Bangkok, Thailand. sisth@mahidol.ac.th 2 Structure of synapse Modes of synaptic
Outline Synaptic transmission Sompol Tapechum M.D., Ph.D. Department of Physiology Faculty of Medicine Siriraj Hospital, Bangkok, Thailand. sisth@mahidol.ac.th 2 Structure of synapse Modes of synaptic
Cannabinoid CB 1 Discrimination: Effects of Endocannabinoids and Catabolic Enzyme Inhibitors
 1521-0103/363/3/314 323$25.00 https://doi.org/10.1124/jpet.117.244392 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS J Pharmacol Exp Ther 363:314 323, December 2017 Copyright ª 2017 by The American
1521-0103/363/3/314 323$25.00 https://doi.org/10.1124/jpet.117.244392 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS J Pharmacol Exp Ther 363:314 323, December 2017 Copyright ª 2017 by The American
NEUROMUSCULAR BLOCKING AGENTS
 NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
Rapid elevations in limbic endocannabinoid content by glucocorticoid hormones in vivo
 Psychoneuroendocrinology (2010) 35, 1333 1338 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/psyneuen Rapid elevations in limbic endocannabinoid content by glucocorticoid
Psychoneuroendocrinology (2010) 35, 1333 1338 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/psyneuen Rapid elevations in limbic endocannabinoid content by glucocorticoid
The Nervous System Mark Stanford, Ph.D.
 The Nervous System Functional Neuroanatomy and How Neurons Communicate Mark Stanford, Ph.D. Santa Clara Valley Health & Hospital System Addiction Medicine and Therapy Services The Nervous System In response
The Nervous System Functional Neuroanatomy and How Neurons Communicate Mark Stanford, Ph.D. Santa Clara Valley Health & Hospital System Addiction Medicine and Therapy Services The Nervous System In response
NIH Public Access Author Manuscript J Mol Neurosci. Author manuscript; available in PMC 2008 February 20.
 NIH Public Access Author Manuscript Published in final edited form as: J Mol Neurosci. 2007 September ; 33(1): 18 24. STUDIES OF ANANDAMIDE ACCUMULATION INHIBITORS IN CEREBELLAR GRANULE NEURONS: COMPARISON
NIH Public Access Author Manuscript Published in final edited form as: J Mol Neurosci. 2007 September ; 33(1): 18 24. STUDIES OF ANANDAMIDE ACCUMULATION INHIBITORS IN CEREBELLAR GRANULE NEURONS: COMPARISON
Research Article. Inhibitors of Endocannabinoid-Metabolizing Enzymes Reduce Precipitated Withdrawal Responses in THC-Dependent Mice
 The AAPS Journal ( # 2009) DOI: 10.1208/s12248-009-9110-7 Research Article Theme: NIDA Symposium: Drugs of Abuse: Drug Development and Cannabinoids Guest Editor: Rao Rapaka Inhibitors of Endocannabinoid-Metabolizing
The AAPS Journal ( # 2009) DOI: 10.1208/s12248-009-9110-7 Research Article Theme: NIDA Symposium: Drugs of Abuse: Drug Development and Cannabinoids Guest Editor: Rao Rapaka Inhibitors of Endocannabinoid-Metabolizing
Psychoactive drugs Drugs which affect mental processes. Legal but restricted (by prescription only)
 Psychoactive drugs Drugs which affect mental processes mood, perception, memory, state of consciousness May be illegal: Heroin Cocaine Legal: Alcohol nicotine Legal but restricted (by prescription only)
Psychoactive drugs Drugs which affect mental processes mood, perception, memory, state of consciousness May be illegal: Heroin Cocaine Legal: Alcohol nicotine Legal but restricted (by prescription only)
Lecture: Medical Marijuana Long term effects and interac ons with common medicines Neuropharamacology of the brain with cannabis
 Lecture: Medical Marijuana Long term effects and interac ons with common medicines Neuropharamacology of the brain with cannabis William Morrone, DO Robert Piccinini, DO Endocannabinoids in Disease and
Lecture: Medical Marijuana Long term effects and interac ons with common medicines Neuropharamacology of the brain with cannabis William Morrone, DO Robert Piccinini, DO Endocannabinoids in Disease and
Chapter 2: Cellular Mechanisms and Cognition
 Chapter 2: Cellular Mechanisms and Cognition MULTIPLE CHOICE 1. Two principles about neurons were defined by Ramón y Cajal. The principle of connectional specificity states that, whereas the principle
Chapter 2: Cellular Mechanisms and Cognition MULTIPLE CHOICE 1. Two principles about neurons were defined by Ramón y Cajal. The principle of connectional specificity states that, whereas the principle
Synthetic cannabinoids: Flying high under the RADAR Brett Ginsburg, Ph.D.
 Synthetic cannabinoids: Flying high under the RADAR Brett Ginsburg, Ph.D. Introduction Brett Ginsburg, Ph.D. Associate Professor Department of Psychiatry Introduction What are synthetic cannabinoids? Manufacture
Synthetic cannabinoids: Flying high under the RADAR Brett Ginsburg, Ph.D. Introduction Brett Ginsburg, Ph.D. Associate Professor Department of Psychiatry Introduction What are synthetic cannabinoids? Manufacture
Brain Mechanisms of Emotion 1 of 6
 Brain Mechanisms of Emotion 1 of 6 I. WHAT IS AN EMOTION? A. Three components (Oately & Jenkins, 1996) 1. caused by conscious or unconscious evaluation of an event as relevant to a goal that is important
Brain Mechanisms of Emotion 1 of 6 I. WHAT IS AN EMOTION? A. Three components (Oately & Jenkins, 1996) 1. caused by conscious or unconscious evaluation of an event as relevant to a goal that is important
At a Glance. Background Information. Lesson 3 Drugs Change the Way Neurons Communicate
 Lesson 3 Drugs Change the Way Neurons Communicate Overview Students build upon their understanding of neurotransmission by learning how different drugs of abuse disrupt communication between neurons. Students
Lesson 3 Drugs Change the Way Neurons Communicate Overview Students build upon their understanding of neurotransmission by learning how different drugs of abuse disrupt communication between neurons. Students
THC and cannabidiol affect CB 1 receptor function
 THC and cannabidiol affect CB 1 receptor function Dr. Eileen Denovan-Wright, PhD Department of Pharmacology, Dalhousie University, Halifax, Nova Scotia, Canada XXIX W.O. MCCORMICK ACADEMIC DAY CONFERENCE
THC and cannabidiol affect CB 1 receptor function Dr. Eileen Denovan-Wright, PhD Department of Pharmacology, Dalhousie University, Halifax, Nova Scotia, Canada XXIX W.O. MCCORMICK ACADEMIC DAY CONFERENCE
EFFECTS OF ACUTELY APPLIED CANNABINOID CB1 LIGANDS ON NOCICEPTION IN RATS WITH A MODEL OF DEPRESSION
 ý Comptes rendus de l Académie bulgare des Sciences ÌÓÑ ÆÓ ¾¼½ MEDECINE Pathophysiologie EFFECTS OF ACUTELY APPLIED CANNABINOID CB1 LIGANDS ON NOCICEPTION IN RATS WITH A MODEL OF DEPRESSION Miroslav Marinov,
ý Comptes rendus de l Académie bulgare des Sciences ÌÓÑ ÆÓ ¾¼½ MEDECINE Pathophysiologie EFFECTS OF ACUTELY APPLIED CANNABINOID CB1 LIGANDS ON NOCICEPTION IN RATS WITH A MODEL OF DEPRESSION Miroslav Marinov,
Cogs 107b Systems Neuroscience lec9_ neuromodulators and drugs of abuse principle of the week: functional anatomy
 Cogs 107b Systems Neuroscience www.dnitz.com lec9_02042010 neuromodulators and drugs of abuse principle of the week: functional anatomy Professor Nitz circa 1986 neurotransmitters: mediating information
Cogs 107b Systems Neuroscience www.dnitz.com lec9_02042010 neuromodulators and drugs of abuse principle of the week: functional anatomy Professor Nitz circa 1986 neurotransmitters: mediating information
BIPN140 Lecture 8: Synaptic Transmission II
 BIPN140 Lecture 8: Synaptic Transmission II 1. Postsynaptic Receptors: Metabotropic & Ionotropic 2. Postsynaptic Responses (Postsynaptic Potentials, PSPs) 3. Neurotransmitters Su (FA16) Chemical Synapse:
BIPN140 Lecture 8: Synaptic Transmission II 1. Postsynaptic Receptors: Metabotropic & Ionotropic 2. Postsynaptic Responses (Postsynaptic Potentials, PSPs) 3. Neurotransmitters Su (FA16) Chemical Synapse:
The Nervous System. Anatomy of a Neuron
 The Nervous System Chapter 38.1-38.5 Anatomy of a Neuron I. Dendrites II. Cell Body III. Axon Synaptic terminal 1 Neuron Connections dendrites cell body terminal cell body cell body terminals dendrites
The Nervous System Chapter 38.1-38.5 Anatomy of a Neuron I. Dendrites II. Cell Body III. Axon Synaptic terminal 1 Neuron Connections dendrites cell body terminal cell body cell body terminals dendrites
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Abbott s TRPV1 Antagonist Program
 Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
Bin Pan, Wei Wang, Jonathan Z. Long, Dalong Sun, Cecilia J. Hillard, Benjamin F. Cravatt, and Qing-song Liu
 0022-3565/09/3312-591 597$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 331, No. 2 Copyright 2009 by The American Society for Pharmacology and Experimental Therapeutics 158162/3523384
0022-3565/09/3312-591 597$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 331, No. 2 Copyright 2009 by The American Society for Pharmacology and Experimental Therapeutics 158162/3523384
CURRENT REVIEW. Endocannabinoids and Their Implications for Epilepsy. Neurophysiological Properties of the Endocannabinoids Systems
 CURRENT REVIEW Endocannabinoids and Their Implications for Epilepsy Bradley E. Alger, Ph.D. University of Maryland School of Medicine, Baltimore, Maryland This review covers the main features of a newly
CURRENT REVIEW Endocannabinoids and Their Implications for Epilepsy Bradley E. Alger, Ph.D. University of Maryland School of Medicine, Baltimore, Maryland This review covers the main features of a newly
Blockade of 2-AG hydrolysis by selective monoacylglycerol lipase inhibitor JZL184. enhances retrograde endocannabinoid signaling
 JPET Fast Forward. Published on August 7, 2009 as DOI:10.1124/jpet.109.158162 JPET #158162 Blockade of 2-AG hydrolysis by selective monoacylglycerol lipase inhibitor JZL184 enhances retrograde endocannabinoid
JPET Fast Forward. Published on August 7, 2009 as DOI:10.1124/jpet.109.158162 JPET #158162 Blockade of 2-AG hydrolysis by selective monoacylglycerol lipase inhibitor JZL184 enhances retrograde endocannabinoid
Organization of the nervous system. [See Fig. 48.1]
![Organization of the nervous system. [See Fig. 48.1] Organization of the nervous system. [See Fig. 48.1]](/thumbs/90/103926552.jpg) Nervous System [Note: This is the text version of this lecture file. To make the lecture notes downloadable over a slow connection (e.g. modem) the figures have been replaced with figure numbers as found
Nervous System [Note: This is the text version of this lecture file. To make the lecture notes downloadable over a slow connection (e.g. modem) the figures have been replaced with figure numbers as found
Class 5 the neurotransmitters (drugs)
 Victoria September 7th 2017 Class 5 the neurotransmitters (drugs) psychopharmacology: study of the effects of a drug on behavior pharmacokinetics: study of the fate / movement of substances administered
Victoria September 7th 2017 Class 5 the neurotransmitters (drugs) psychopharmacology: study of the effects of a drug on behavior pharmacokinetics: study of the fate / movement of substances administered
SAMPLE EXAMINATION QUESTIONS
 SAMPLE EXAMINATION QUESTIONS PLEASE NOTE, THE QUESTIONS BELOW SAMPLE THE ENTIRE LECTURE COURSE AND THEREORE INCLUDE QUESTIONS ABOUT TOPICS THAT WE HAVE NOT YET COVERED IN CLASS. 1. Which of the following
SAMPLE EXAMINATION QUESTIONS PLEASE NOTE, THE QUESTIONS BELOW SAMPLE THE ENTIRE LECTURE COURSE AND THEREORE INCLUDE QUESTIONS ABOUT TOPICS THAT WE HAVE NOT YET COVERED IN CLASS. 1. Which of the following
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Neurotransmitter: dopamine. Physiology of additive drugs. Dopamine and reward. Neurotransmitter: dopamine
 Physiology of additive drugs Cocaine, methamphetamine, marijuana, and opiates influence the neurotransmitter dopamine. Neurotransmitter: dopamine Dopamine - a neurotransmitter associated with several functions,
Physiology of additive drugs Cocaine, methamphetamine, marijuana, and opiates influence the neurotransmitter dopamine. Neurotransmitter: dopamine Dopamine - a neurotransmitter associated with several functions,
Glycine-gated ion channels Converging mechanism and therapeutic potentials
 Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Chapter 6 Communication, Integration, and Homeostasis
 Chapter 6 Communication, Integration, and Homeostasis About This Chapter Cell-to-cell communication Signal pathways Novel signal molecules Modulation of signal pathways Homeostatic reflex pathways Cell-to-Cell
Chapter 6 Communication, Integration, and Homeostasis About This Chapter Cell-to-cell communication Signal pathways Novel signal molecules Modulation of signal pathways Homeostatic reflex pathways Cell-to-Cell
The postmortal accumulation of brain N-arachidonylethanolamine (anandamide) is dependent upon fatty acid amide hydrolase activity
 The postmortal accumulation of brain N-arachidonylethanolamine (anandamide) is dependent upon fatty acid amide hydrolase activity Sachin Patel,* Erica J. Carrier,* W-S. Vanessa Ho,* David J. Rademacher,*
The postmortal accumulation of brain N-arachidonylethanolamine (anandamide) is dependent upon fatty acid amide hydrolase activity Sachin Patel,* Erica J. Carrier,* W-S. Vanessa Ho,* David J. Rademacher,*
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Neurophysiology of the Regulation of Food Intake and the Common Reward Pathways of Obesity and Addiction. Laura Gunter
 Neurophysiology of the Regulation of Food Intake and the Common Reward Pathways of Obesity and Addiction Laura Gunter The Brain as the Regulatory Center for Appetite The brain is the integration center
Neurophysiology of the Regulation of Food Intake and the Common Reward Pathways of Obesity and Addiction Laura Gunter The Brain as the Regulatory Center for Appetite The brain is the integration center
NERVOUS SYSTEM NERVOUS SYSTEM. Somatic nervous system. Brain Spinal Cord Autonomic nervous system. Sympathetic nervous system
 SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
Chapter 4. Psychopharmacology. Copyright Allyn & Bacon 2004
 Chapter 4 Psychopharmacology This multimedia product and its contents are protected under copyright law. The following are prohibited by law: any public performance or display, including transmission of
Chapter 4 Psychopharmacology This multimedia product and its contents are protected under copyright law. The following are prohibited by law: any public performance or display, including transmission of
Drugs, The Brain, and Behavior
 Drugs, The Brain, and Behavior John Nyby Department of Biological Sciences Lehigh University What is a drug? Difficult to define Know it when you see it Neuroactive vs Non-Neuroactive drugs Two major types
Drugs, The Brain, and Behavior John Nyby Department of Biological Sciences Lehigh University What is a drug? Difficult to define Know it when you see it Neuroactive vs Non-Neuroactive drugs Two major types
Cannabidiol as a potential treatment for anxiety disorders. Esther Blessing, MD PhD
 Cannabidiol as a potential treatment for anxiety disorders Esther Blessing, MD PhD Overview History and of cannabidiol (CBD) and cannabis Safety and tolerability of CBD CBD pharmacology Clinical need for
Cannabidiol as a potential treatment for anxiety disorders Esther Blessing, MD PhD Overview History and of cannabidiol (CBD) and cannabis Safety and tolerability of CBD CBD pharmacology Clinical need for
Endocannabinoid Modulation of Scratching Response in an Acute Allergenic Model: A New Prospective Neural Therapeutic Target for Pruritus
 0022-3565/09/3291-314 323$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 329, No. 1 Copyright 2009 by The American Society for Pharmacology and Experimental Therapeutics 150136/3454849
0022-3565/09/3291-314 323$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 329, No. 1 Copyright 2009 by The American Society for Pharmacology and Experimental Therapeutics 150136/3454849
Chapter 2 Transport Systems
 Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Communication within a Neuron
 Neuronal Communication, Ph.D. Communication within a Neuron Measuring Electrical Potentials of Axons The Membrane Potential The Action Potential Conduction of the Action Potential 1 The withdrawal reflex
Neuronal Communication, Ph.D. Communication within a Neuron Measuring Electrical Potentials of Axons The Membrane Potential The Action Potential Conduction of the Action Potential 1 The withdrawal reflex
The Putative Endocannabinoid Transport Blocker LY Is a Potent Inhibitor of FAAH and Several Other Brain Serine Hydrolases
 Published on Web 07/06/2006 The Putative Endocannabinoid Transport Blocker LY2183240 Is a Potent Inhibitor of FAAH and Several Other Brain Serine Hydrolases Jessica P. Alexander and Benjamin F. Cravatt*
Published on Web 07/06/2006 The Putative Endocannabinoid Transport Blocker LY2183240 Is a Potent Inhibitor of FAAH and Several Other Brain Serine Hydrolases Jessica P. Alexander and Benjamin F. Cravatt*
Action Potentials and Synaptic Transmission. BIO 219 Napa Valley College Dr. Adam Ross
 Action Potentials and Synaptic Transmission BIO 219 Napa Valley College Dr. Adam Ross Review of action potentials Nodes of Ranvier Nucleus Dendrites Cell body In saltatory conduction, the nerve impulses
Action Potentials and Synaptic Transmission BIO 219 Napa Valley College Dr. Adam Ross Review of action potentials Nodes of Ranvier Nucleus Dendrites Cell body In saltatory conduction, the nerve impulses
Close to site of release (at synapse); binds to receptors in
 Chapter 18: The Endocrine System Chemical Messengers 1. Neural 2. Endocrine 3. Neuroendocrine 4. Paracrine 5. Autocrine Endocrine System --Endocrine and nervous systems work together --Endocrine vs. Nervous
Chapter 18: The Endocrine System Chemical Messengers 1. Neural 2. Endocrine 3. Neuroendocrine 4. Paracrine 5. Autocrine Endocrine System --Endocrine and nervous systems work together --Endocrine vs. Nervous
CNS Control of Food Intake. Adena Zadourian & Andrea Shelton
 CNS Control of Food Intake Adena Zadourian & Andrea Shelton Controlling Food Intake Energy Homeostasis (Change in body adiposity + compensatory changes in food intake) Background Information/Review Insulin
CNS Control of Food Intake Adena Zadourian & Andrea Shelton Controlling Food Intake Energy Homeostasis (Change in body adiposity + compensatory changes in food intake) Background Information/Review Insulin
