Effect of the temperature of polyurethane dissolution on the mechanism of wet-casting membrane formation
|
|
|
- Shonda Maxwell
- 5 years ago
- Views:
Transcription
1 European Polymer Journal 39 (2003) Effect of the temperature of polyurethane dissolution on the mechanism of wet-casting membrane formation Liao-Ping Cheng a, Yen-Shih Huang b, Tai-Horng Young b, * a Department of Chemical Engineering, Tamkang University, Taipei 25137, Taiwan, ROC b Institute of Biomedical Engineering, College of Medicine and College of Engineering, National Taiwan University, Taipei 10016, Taiwan, ROC Received 22 February 2002; received in revised form 5 August 2002; accepted 4 September 2002 Abstract An analysis of data from scanning electron microscopy (SEM), gel permeation chromatography (GPC) and differential scanning calorimetry (DSC) for two series of polyurethane membranes is presented. Membranes were prepared by direct immersion of the casting solution of polyurethane, dissolved in dimethyl formamide at different temperatures, into either water or 1-octanol bath at 25 C. Depending on the temperature of polyurethane dissolution, a gradual variety in the membrane structure was observed by SEM. As the temperature of polymer dissolution was increased, the membrane structure changed from dense to cellular or particulate morphologies. On the basis of the GPC and DSC results, polyurethane molecular weight decreased but the degree of microphase separation caused by clustering of some of the soft and hard segments into separate domains in the membrane increased with increasing the temperature of polymer dissolution. It is thus proposed that the change in membrane structure is due to the variation of molecular weight of polyurethane, which in turn can change polymer chain mobility during membrane formation. In addition, the extent of microphase separation was described and related to the particulate morphology when 1-octanol was used as the nonsolvent. Ó 2002 Elsevier Science Ltd. All rights reserved. Keywords: Polyurethane; Dissolution temperature; Particulate membranes 1. Introduction Recently polymeric membranes have achieved commercial importance in a variety of separation applications [1]. Nonsolvent-induced phase inversion process is one of the most widely used means of preparing polymeric membranes [2]. In this process, a polymer solution is brought into contact with nonsolvent to induce phase separation, which forms the membrane. Polymer dissolution is a slow process so dissolving polymer in solvent is usually speeded by agitation. However, polymer * Corresponding author. Tel.: x1455; fax: address: thyoung@ha.mc.ntu.edu.tw (T.-H. Young). dissolution process may be still quite slow for polymers with high molecular weight and at high concentration. Alternative approach is to increase the dissolution temperature to enhance rate of polymer dissolution. In this work, the relationship between the temperature of polyurethane dissolution and the membrane structure was studied. Polyurethane used in this study was the commercial Pellethane AE (Dow Chemical Company), based on 4,4 0 -methylene bis (phenylene isocyanate) (MDI), polytetramethylene oxide (PTMO) and 1,4 butanediol. Membranes were prepared by direct immersion of polyurethane solution into either water or 1-octanol bath. The prepared membranes were analyzed by scanning electron microscopy (SEM), gel permeation chromatography (GPC) and differential scanning calorimetry (DSC) to elucidate the membrane formation mechanism /02/$ - see front matter Ó 2002 Elsevier Science Ltd. All rights reserved. PII: S (02)
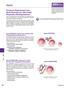
2 602 L.-P. Cheng et al. / European Polymer Journal 39 (2003) Polyurethanes are a large family of synthetic polymers that are composed of alternating soft polyether segments, most commonly long linear polyethers, and hard urethane segments consisting of urethane linkages and aromatic groups. The existence of microphase separation, caused by clustering of some of the hard and soft segments into separate domains, has been well established [3]. The excellent mechanical properties of polyurethanes can be ascribed to the coexistence of these domains. While polyurethanes are susceptible to thermal, chemical and hydrolytic degradation [4], they may undergo different degrees of degradation in a given solvent at high temperature. Therefore, when the temperature of polymer dissolution is increased, polyurethane can dissolve more readily but this effect may decrease its molecular weight to some degree. In addition, Matsumoto et al. [5] have proposed that the polymer molecular weight can influence the poly (ethylene-co-vinyl alcohol) (EVAL) membrane structure. Membranes prepared from high molecular weight EVAL had large closed pores and a relatively lower number of pores, while those prepared from low molecular weight EVAL yielded small open pores and a large number of pores. Hence, the purpose of this paper was to examine whether polyurethane molecular weight degraded at higher dissolution temperature to result in the change of the membrane structure. We found the membrane structure changed from dense to cellular or particulate morphologies when the temperature of polymer dissolution was increased. Recently, particulate membranes have been explored as an alternative to various application of replacing traditional dense, porous or asymmetrical membranes in our laboratory [6 10]. Different particulate membranes have proved useful in microfiltration [6,7], plasma protein separation [8,9], and cell culture [10]. Therefore, this paper provides an easy method to prepare polyurethane membranes with the particulate morphology. Furthermore, the extent of microphase separation caused by clustering of hard and soft segments was described and related to the particulate morphology. at 60, 90 and 120 C, respectively, for 3 days and then kept at 25 C for 1 day. These dopes were commonly observed to precipitate upon standing, thus, they were heated to 60 C to ensure complete dissolution before immersing into the precipitation bath at 25 C. The precipitation time required for water and 1-octanol were 1 min and 1 h, respectively. The residual solvent was removed from the membrane by a series of washing steps. Finally, the formed membrane was dried in open air Membrane characterization The morphology of the membrane was examined in different views by using SEM. The samples were sputtered with gold and palladium in a vacuum and using a Hitachi S-800 microscope at 20 kv. To evaluate the effect of polymer dissolution temperature on thermal degradation of polyurethane, the membranes were analyzed by GPC using the PLgel 5 lm mixed-d column from Polymer Lab., UK. The prepared membranes were dissolved in tetrahydrofuran (THF) in the concentration of 0.5% by weight. Ultra pure-grade THF (Alps Chem., Taiwan) was used as the mobile phase. The effluent flow rate was maintained at 1.0 ml/ min using a SPECTROFLOW 400 pump (Applied Biosystem Corporation, USA). The refractive index detector in this GPC system was Shodex RI SE-61 (Showa Denko Corporation, Japan). Monodisperse polystyrene standards dissolved in THF were used to calibrate the system. The thermal transition behavior of each membrane was studied using a sensitive DSC 2010 (TA instruments Ltd., USA) to understand the degree of microphase separation in the polyurethanes. DSC was calibrated by using indium as standards. The measurements were performed under a nitrogen atmosphere at a heating rate of 10 C/min to observe the glassy transition temperature and characteristic endothermic transition. The characteristic endothermic transition is taken as the temperature at the endothermic peak of calorimetric transition. 2. Experimental 2.1. Membrane preparation Polyurethane (Pellethane AE) membranes were prepared using direct immersion precipitation method. A homogeneous dope composed of 3 g of Pellethane elastomers and 7 g of dimethyl formamide (DMF) was spread uniformly on a glass plate to form a laminate (250 lm) which was then immersed immediately into water or 1-octanol to effect polymer precipitation. In this work, dopes were prepared by dissolving Pellethane 3. Results 3.1. SEM analysis Fig. 1 shows the top and cross-sectional views of polyurethane membranes, which was prepared by immersing the polyurethane solution dissolved in DMF at different temperatures into water. In the cross-sectional photographs, upper side corresponds to the top surface. When polyurethane was dissolved at 60 C, there were
3 L.-P. Cheng et al. / European Polymer Journal 39 (2003) Fig. 1. SEM photomicrographs of polyurethane membranes prepared by immersing the polyurethane solution dissolved at different temperatures into water: (a) 60 C, top surface; (b) 60 C, cross section; (c) 90 C, top surface; (d) 90 C, cross section; (e) 120 C, top surface; (f) 120 C, cross section. few pores like the artifact of a rough and sunken surface on the membrane surface (Fig. 1(a)) but no porous structure was observed in the cross-sectional photograph (Fig. 1(b)). Therefore, the whole structure in this case is regarded as a dense morphology. On the other hand, when the polyurethane dissolution temperature was increased to 120 C, the porous structure was formed in the membrane surface (Fig. 1(e)) and its cross section consisted of cellular pores enclosed in the continuous polymer matrix phase (Fig. 1(f)). These pores were separated and had the nearly same size (ca. several lm). Such a porous configuration is similar to the so-called sponge structure that are produced by the liquid liquid demixing process. The structure of the membrane prepared by dissolving polyurethane at 90 C is shown in Fig. 1(c) and (d). A rough surface and an irregular cross section with some pores were observed. Such a structure represents the intermediate case between membranes prepared by dissolving polyurethane at 60 and 120 C. Fig. 2 presents the top and cross-sectional views of polyurethane membranes prepared by immersing the polyurethane solution dissolved in DMF at different temperatures into 1-octanol. When polyurethane was dissolved at 60 C, the membrane surface obviously had many spherical particles (Fig. 2(a)), which was supported by a thick dense sublayer (Fig. 2(b)). Besides few pores existing near the membrane surface, there was almost not any pores or particles existing in the cross section. When the temperature of polymer dissolution
4 604 L.-P. Cheng et al. / European Polymer Journal 39 (2003) Fig. 2. SEM photomicrographs of polyurethane membranes prepared by immersing the polyurethane solution dissolved at different temperatures into 1-octanol: (a) 60 C, top surface; (b) 60 C, cross section; (c) 90 C, top surface; (d) 90 C, cross section; (e) 120 C, top surface; (f) 120 C, cross section. was increased to 90 C, the membrane surface and cross section (Fig. 2(c) and (d)) were still similar to those of membranes prepared by dissolving polyurethane at 60 C. However, when the polyurethane dissolution temperature was increased to 120 C, spherical particles existed not only on the membrane surface (Fig. 2(e)) but also throughout the cross section (Fig. 2(f)). Although the particles were difficult to distinguish each other due to incomplete coalescence of particles, the size of the particles approximately fell in the range of a few micrometers. The mechanical strength of particulate membranes depends heavily upon the links between polymer particles. Since the linkage points are relatively limited, only on the interface between spherical particles, their strength is not that satisfactory as membranes with cellular pores enclosed in the continuous polymer matrix phase. However, the mechanical strength of the particulate membrane is still strong enough to various applications [6 10]. In addition, such porous and open structure can fulfill the requirement of scaffolds to maximize cell adhesion, growth and differentiation in tissue engineering applications. Therefore, in our laboratory, particulate membranes are used as a supporting material to act as a scaffold for cell growth. We have previously demonstrated that osteoblast cells cultured on particulate surfaces were well spread and flattened, and had a great number of adhesion [11].
5 L.-P. Cheng et al. / European Polymer Journal 39 (2003) GPC analysis Table 1 shows the weight average molecular weight and molecular weight distribution of prepared polyurethane membranes. The data reveals the weight average molecular weight of polyurethane decreased as the dissolution temperature was increased. Grassie and Zulfiqar claimed that the thermal degradation of polyurethanes is primarily a depolycondensation process, namely the reverse of the reaction in which they are formed [12]. Based on the present result, polyurethane degradation was greatly influenced by the dissolution temperature, which might further influence the membrane formation mechanism (discussed below). In addition, the polydispersity of polyurethane also shows a decreasing trend. This can be ascribed to the fact that the low molecular weight fragments degraded at higher dissolution temperature were removed during membrane formation. Therefore, the loss of low molecular weight fragments should have more impact on the weight average molecular weight and decreased molecular weight distribution DSC analysis Fig. 3 shows the DSC thermograms collected from polyurethane membranes prepared by immersing the polyurethane solution dissolved in DMF at 60 C into water and 1-octanol. Briefly, the typical glass transition temperature (T g ) of the soft segment around )50 C and a broad peak with a maximum around 150 C were found. This broad peak present in the temperature range C can be considered as a merged endotherm from two peaks at approximately 70 and 150 C, which are due to the disruption of short and long range order of hard segments, respectively, as proposed by Seymour and Cooper [3]. Similar DSC curves were obtained for all polyurethane membranes studied (not shown here). The T g of the soft segment and the peak temperature of merged endotherm for all prepared polyurethane membranes are summarized in Table 2. The position of Table 1 Weight average molecular weight and molecular weight distribution of prepared polyurethane membranes Polyurethane dissolution M w (g) b MWD c temperature ( C) a , , , a The change of molecular weight of polyurethane membranes was independent of the nonsolvent so dependent on the polyurethane dissolution temperature only. b M w : weight average molecular weight. c MWD: molecular weight distribution. Fig. 3. DSC thermograms from polyurethane membranes prepared by immersing the polyurethane solution dissolved at 60 C into water and 1-octanol. Table 2 DSC first heat data for polyurethane membranes Precipitation medium Polyurethane dissolution temperature ( C) Glass transition temperature of the soft segment ( C) Water 60 ) Water 90 ) Water 120 ) Octanol 60 ) Octanol 90 ) Octanol 120 ) Peak temperature of the merged endotherm ( C) peak temperature of the merged endotherm presents no discernible quantitative trend but the T g of the soft segment shows the polymer dissolution temperature could modify the state of microphase separation. When water was used as the nonsolvent, T g decreased from )45.4 to )56.7 C with increasing the polyurethane dissolution temperature from 60 to 120 C, which is substantially higher than that of pure soft-segment polymer. (The T g of poly (tetramethylene oxide) is about )82 C [13].) This indicates a certain degree of hard and soft segment mixing. Conversely, the polyurethane membranes with using 1-octanol as the nonsolvent exhibited slight lower T g of )53.1 to )60.7 C, which appears that hard segment solubilization in the soft segment matrix is smaller. This indicates an improved microphase separation when 1-octanol was used as the nonsolvent to prepare polyurethane membranes. Furthermore, polyurethane membranes exhibited a decrease of T g by 7 to 11 C when the polymer dissolution temperature was increased, irrespective of immersing polyurethane solution into water or 1-octanol. This
6 606 L.-P. Cheng et al. / European Polymer Journal 39 (2003) suggests polyurethane membranes consist of higher purity of the soft segment phase when the polymer dissolution temperature was increased. This is probably caused by the fact that the soft segments and the hard segments could organize the membrane microstructure in the course of preparation of the membrane. However, this process will be impeded in the case of polymer with higher molecular weight due to the serious molecular entanglements. Therefore, polyurethane molecules with low molecular weight in the casting solution can reorganize the membrane microstructure during membrane formation. 4. Discussion The formation of porous structure in a membrane is dictated by the sequence of the phase transition events in the precipitation process [14,15]. Many parameters, such as the compositions of the casting solution and the precipitation bath, and the temperature of the system, are known to exert certain influences on the morphology of the formed membranes [1,2]. In this study, depending on the polyurethane dissolution temperature, a gradual variety in membrane structures could be observed via SEM. In addition, two different types of membranes were synthesized in two nonsolvent-dmf polyurethane systems. At low dissolution temperatures (e.g., 60 C), phase separation of polyurethane solution by exchange of solvent and nonsolvent was clearly limited, which forms the dense structure. Whereas at elevated dissolution temperatures (e.g., 120 C) liquid liquid demixing process dominated the precipitation process by using water as the nonsolvent and the membrane became cellular in its morphology. As the nonsolvent using 1- octanol, the membrane showed a homogeneous particulate morphology while the cellular morphology was suppressed. It is desirable to characterize the differences in morphology of the two series of polyurethane membranes. Since membrane preparation is a nonequilibrium process, this clearly implies that the change in membrane structure is attributed to the arrangement of polymer chains during membrane formation. When casting solutions containing high molecular weight polymer are immersed in a coagulation bath, the molecular entanglements of polymer should restrict chain mobility. Therefore, polymer molecules are brought rapidly into a state wherein vitrification takes place and the structure is frozen. Indeed, GPC studies suggested that the polyurethane dissolution temperature had pronounced effect on the polyurethane degradation, as indicated in Table 1. Therefore, at low dissolution temperatures (e.g., 60 C), polyurethane molecules tended to aggregate to form a dense structure by virtue of a great number of molecular entanglements. This membrane was very hard and stiff during membrane formation so as to decrease the possibility of microporous regions besides the membrane surface. Conversely, as the molecular weight was decreased at elevated dissolution temperatures (e.g., 120 C), polyurethane molecules with sufficient mobility in the membrane solution can arrange themselves faster than the nonsolvent-induced polymer vitrification. Hence, polymer molecules underwent liquid liquid demixing or solid liquid demixing according to the thermodynamic equilibrium. Consequently, the structure of the polyurethane membranes did not only depend on the dissolution temperature but also varied with the nonsolvents used. Another interesting feature of polyurethane membranes is that they can form a symmetric structure by constituent particles bonded to each other when 1-octanol was used as the nonsolvent. It is known that many investigators have prepared different polymer membranes with similar particulate morphology by using 1- octanol as the nonsolvent though these polymers have rather different chemical properties [8,16 18]. With different circumstances, there can be two possible mechanisms for the formation of particles in membranes. The first possibility is that particles are generated by the spinodal demixing [19]. The second possible origin of particles is crystallization of polymer during the membrane formation process [14,20,21]. Since the building particles in our polyurethane membranes are greater than the size of nodules caused by the spinodal demixing, it is suggested that particle structure cannot be ascribed to the spinodal demixing. However, it is also not certain at this point whether the particle structure in our polyurethane membranes is generated by crystallization of polyurethane. Polyurethane membranes may be considered as soft segments dispersed within the hard domains and as hard segments dispersed in the soft matrix [3]. Thus, it is reasonable to suggest the particle structure can be attributed to an improvement in the extent of microphase separation in polyurethane membranes with 1- octanol as the nonsolvent. Indeed, DSC studies show that polyurethane membranes prepared by using 1-octanol as the nonsolvent had a well-phase-separated morphology, a lower value in T g of the soft segment. The decrease of T g could be explained by assuming that the rearrangement of polymer chains during the precipitation process produces a decrease of the percent of soft segments dissolved in the hard segments phase. For these reasons, we propose that locus of particle nucleus resulted from microphase separated polymers, which is beneficial to the chains to pack into a regular lattice. It seems logical that the small initially formed particles needed better chain mobility and longer induction time. This is consistent with the observed longer precipitation time for low molecular weight polyurethane membranes to present a symmetric structure by constituent particles bonded to each other.
7 L.-P. Cheng et al. / European Polymer Journal 39 (2003) In summary, the exact nature of particles has not yet been satisfactorily resolved but it should be noted that polyurethane membranes prepared from using 1-octanol as the nonsolvent contained a well-phase-separated morphology, suggesting that nonsolvent-induced ordering played an important role during membrane formation. Consequently, the complex membrane structures can be ascribed to various levels of packing order in the soft segment and hard segment domains. Acknowledgements This work is supported by the National Science Council of the Republic of China. References [1] Mulder M. Basic principles of membrane technology. Dordrecht, The Netherlands: Kluwer; [2] Kesting RE. Synthetic polymeric membranes. New York: John Wiley and Sons; [3] Seymour RW, Cooper SL. Thermal analysis of polyurethane block polymers. Macromolecules 1973;6: [4] Lelah MD, Cooper SL. Polyurethanes in medicine. Boca Raton: CRC Press; [5] Matsumoto T, Nakamae K, Ochiumi T, Horie S. Effect of molecular weight of ethylene vinyl alcohol copolymer on membrane structures. J Membr Sci 1981;9: [6] Cheng LP, Lin HY, Chen LW, Young TH. Solute rejection of dextran by EVAL membranes with asymmetric and particulate morphologies. Polymer 1998;39: [7] Young TH, Cheng LP, Lin HY. Interesting behavior for filtration of macromolecules through EVAL membranes. Polymer 2000;41: [8] Lin DT, Cheng LP, Kang YJ, Chen LW, Young TH. Effects of precipitation conditions on the membrane morphology and permeation characteristics. J Membr Sci 1998;140: [9] Young TH, Lin DT, Chen LY, Huang YH, Chiu WY. Membranes with a particulate morphology prepared by a dry wet casting process. Polymer 1999;40: [10] Young TH, Yao CH, Sun JS, Lai CP, Chen LW. The effect of morphology variety of EVAL membranes on the behavior of myoblasts in vitro. Biomaterials 1998;19: [11] Wang JH, Yao CH, Chuang WY, Young TH. Development of biodegradable polyesterurethane membranes with different surface morphologies for the culture of osteoblasts. J Biomed Mater Res 2000;51: [12] Grassie N, Zulfiqar M. Thermal degradation of the polyurethane from 1,4-butanediol and methylene bis(4- phenyl isocyanate). J Polym Sci, Polym Chem Ed 1978;16: [13] Wang CB, Cooper SL. Morphology and properties of segmented polyether polyurethaneureas. Macromolecules 1983;16: [14] Young TH, Lai JY, Yu WM, Cheng LP. Equilibrium phase behavior of the membrane forming water DMSO EVAL copolymer system. J Membr Sci 1997;128: [15] Young TH, Hsieh CC, Chen LY, Huang YS. The formation mechanism of membranes prepared from the crystalline EVAL polymer water (nonsolvent) 2-propanol (nonsolvent) system. J Membr Sci 1999;159:21 7. [16] Broens L, Koenhen DM, Smolders CA. On the mechanism of formation of asymmetric ultra- and hyper-filtration membranes. Desalination 1977;22: [17] Wijmans JG, Rutten HJJ, Smolders CA. Phase separation phenomena in solutions of poly(2,6-dimethyl-1,4-phenyleneoxide) in mixtures of trichloroethylene, 1-octanol, and methanol: relationship to membrane formation. J Polym Sci, Polym Phys 1985;23: [18] Burghardt WR, Yilmaz L, McHugh AJ. Glass transition, crystallization and thermoreversible gelation in ternary PPO solution; relationship to asymmetric formation. Polymer 1987;28: [19] Nunes SP, Inoue T. Evidence for spinodal decomposition and nucleation and growth mechanisms during membrane formation. J Membr Sci 1996;111: [20] Bulte AMW, Folkers B, Mulder MHV, Smolders CA. Membranes of semicrystalline aliphatic polyamide nylon 4,6: formation by diffusion-induced phase separation. J Appl Polym Sci 1993;50: [21] Cheng LP, Dwan AW, Gryte CC. Membrane formation by isothermal precipitation in polyamide formic acid water systems. II. Precipitation dynamics. J Polym Sci, Polym Phys 1995;33:
E ect of evaporation temperature on the formation of particulate membranes from crystalline polymers by dry-cast process
 European Polymer Journal 38 2002) 63±72 www.elsevier.com/locate/europolj E ect of evaporation temperature on the formation of particulate membranes from crystalline polymers by dry-cast process Tai-Horng
European Polymer Journal 38 2002) 63±72 www.elsevier.com/locate/europolj E ect of evaporation temperature on the formation of particulate membranes from crystalline polymers by dry-cast process Tai-Horng
Development of biodegradable polyesterurethane membranes with different surface morphologies for the culture of osteoblasts
 Development of biodegradable polyesterurethane membranes with different surface morphologies for the culture of osteoblasts Jyh-Horng Wang, 1 Chun-Hsu Yao, 2 Wen-Yuan Chuang, 3 Tai-Horng Young 3 1 Department
Development of biodegradable polyesterurethane membranes with different surface morphologies for the culture of osteoblasts Jyh-Horng Wang, 1 Chun-Hsu Yao, 2 Wen-Yuan Chuang, 3 Tai-Horng Young 3 1 Department
Study on effective parameters on phase separation in Segmented polyurethanes
 Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, 16-20 September 2007 Study on effective parameters on phase separation in Segmented polyurethanes M. Amrollahi, G. Mir Mohamad
Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, 16-20 September 2007 Study on effective parameters on phase separation in Segmented polyurethanes M. Amrollahi, G. Mir Mohamad
Supplementary data. High-Performance Ultrafiltration Membranes Based on Polyethersulfone/Graphene Oxide Composites
 Supplementary data High-Performance Ultrafiltration Membranes Based on Polyethersulfone/Graphene Oxide Composites Fengmin Jin a, Wei Lv b,a, Chen Zhang a, Zhengjie Li a, Rongxin Su a, Wei Qi a, Quan-Hong
Supplementary data High-Performance Ultrafiltration Membranes Based on Polyethersulfone/Graphene Oxide Composites Fengmin Jin a, Wei Lv b,a, Chen Zhang a, Zhengjie Li a, Rongxin Su a, Wei Qi a, Quan-Hong
Influence of External Coagulant Water Types on the Performances of PES Ultrafiltration Membranes
 30 Journal of Membrane and Separation Technology, 2012, 1, 30-34 Influence of External Coagulant Water Types on the Performances of PES Ultrafiltration Membranes Jing He, Lingyun Ji and Baoli Shi * Polymer
30 Journal of Membrane and Separation Technology, 2012, 1, 30-34 Influence of External Coagulant Water Types on the Performances of PES Ultrafiltration Membranes Jing He, Lingyun Ji and Baoli Shi * Polymer
Synthesis of organophilic ZIF-71 membranes for pervaporation. solvent separation
 Supporting Information Synthesis of organophilic ZIF-71 membranes for pervaporation solvent separation Xueliang Dong, Y. S. Lin* School for Engineering of Matter, Transport and Energy, Arizona State University,
Supporting Information Synthesis of organophilic ZIF-71 membranes for pervaporation solvent separation Xueliang Dong, Y. S. Lin* School for Engineering of Matter, Transport and Energy, Arizona State University,
TABLE OF CONTENT CHAPTER CONTENT PAGE
 vii TABLE OF CONTENT CHAPTER CONTENT PAGE TITLE PAGE DECLARATION DEDICATION ACKNOWLEDGEMENT ABSTRACT ABSTRAK TABLE OF CONTENT LIST OF TABLES LIST OF FIGURES NOMENCLATURE LIST OF APPENDICES i ii iii iv
vii TABLE OF CONTENT CHAPTER CONTENT PAGE TITLE PAGE DECLARATION DEDICATION ACKNOWLEDGEMENT ABSTRACT ABSTRAK TABLE OF CONTENT LIST OF TABLES LIST OF FIGURES NOMENCLATURE LIST OF APPENDICES i ii iii iv
Characterization of Polyamide 66 Membranes Prepared by Phase Inversion Using Formic Acid and Hydrochloric Acid Such as Solvents
 Materials Research. 2011; 14(4): 547-551 2011 DDOI: 10.1590/S1516-14392011005000087 Characterization of Polyamide 66 Membranes Prepared by Phase Inversion Using Formic Acid and Hydrochloric Acid Such as
Materials Research. 2011; 14(4): 547-551 2011 DDOI: 10.1590/S1516-14392011005000087 Characterization of Polyamide 66 Membranes Prepared by Phase Inversion Using Formic Acid and Hydrochloric Acid Such as
Supplementary figures
 Supplementary figures Supplementary Figure 1. Overlaid GPC traces (RI) of the PFDMS 61 aliquot, pure PFDMS 61-b- PMVS 574 copolymer and BCP P after sulfurisation of pendant phosphines. 1 Supplementary
Supplementary figures Supplementary Figure 1. Overlaid GPC traces (RI) of the PFDMS 61 aliquot, pure PFDMS 61-b- PMVS 574 copolymer and BCP P after sulfurisation of pendant phosphines. 1 Supplementary
Accessory Publication
 10.1071/CH09088_AC CSIRO 2009 Accessory Publication: Australian Journal of Chemistry, 2009, 62(8), 790 793 Thermally Responsive Elastomeric Supramolecular Polymers Featuring Flexible Aliphatic Hydrogen
10.1071/CH09088_AC CSIRO 2009 Accessory Publication: Australian Journal of Chemistry, 2009, 62(8), 790 793 Thermally Responsive Elastomeric Supramolecular Polymers Featuring Flexible Aliphatic Hydrogen
Homopolymers as Structure-Driving Agents in Semicrystalline Block Copolymer Micelles
 Supporting information for: Homopolymers as Structure-Driving Agents in Semicrystalline Block Copolymer Micelles Georgios Rizis, Theo G. M. van de Ven*, Adi Eisenberg* Department of Chemistry, McGill University,
Supporting information for: Homopolymers as Structure-Driving Agents in Semicrystalline Block Copolymer Micelles Georgios Rizis, Theo G. M. van de Ven*, Adi Eisenberg* Department of Chemistry, McGill University,
Techniques RESULTS AND DISCUSSION. Materials Description Source Code
 NOTES Study of Thermal Properties and Morphology of Interpenetrating Polymer Networks from Natural Rubber and Polyacrylamide The historical synthesis of the interpenetrating polymer network (IPN) by Millar'
NOTES Study of Thermal Properties and Morphology of Interpenetrating Polymer Networks from Natural Rubber and Polyacrylamide The historical synthesis of the interpenetrating polymer network (IPN) by Millar'
Study of Hydrogen-Bonding Strength in Poly( -caprolactone) Blends by DSC and FTIR
 Study of Hydrogen-Bonding Strength in Poly( -caprolactone) Blends by DSC and FTIR SHIAO WEI KUO, CHIH FENG HUANG, FENG CHIH CHANG Institute of Applied Chemistry, National Chiao Tung University, Hsinchu,
Study of Hydrogen-Bonding Strength in Poly( -caprolactone) Blends by DSC and FTIR SHIAO WEI KUO, CHIH FENG HUANG, FENG CHIH CHANG Institute of Applied Chemistry, National Chiao Tung University, Hsinchu,
Comparison of Melting Behaviors of Edible Oils Using Conventional and Hyper Differential Scanning Calorimetric Scan Rates
 ASEAN Food Journal 14 (1): 25-35 (2007) Comparison of Melting Behaviors of Edible Oils Using Conventional and Hyper Differential Scanning Calorimetric Scan Rates 25 Comparison of Melting Behaviors of Edible
ASEAN Food Journal 14 (1): 25-35 (2007) Comparison of Melting Behaviors of Edible Oils Using Conventional and Hyper Differential Scanning Calorimetric Scan Rates 25 Comparison of Melting Behaviors of Edible
The preparation and performance characteristics of polyvinyl chloride-co-vinyl acetate modified membranes
 Available online at www.sciencedirect.com Energy Procedia 5 (211) 8 1162 IAEED21 The preparation and performance characteristics of polyvinyl chloride-co-vinyl acetate modified membranes Li Wenjuan a,
Available online at www.sciencedirect.com Energy Procedia 5 (211) 8 1162 IAEED21 The preparation and performance characteristics of polyvinyl chloride-co-vinyl acetate modified membranes Li Wenjuan a,
ASIAN JOURNAL OF CHEMISTRY
 Asian Journal of Chemistry; Vol. 27, No. 8 (2015), 2801-2805 ASIAN JOURNAL OF CHEMISTRY http://dx.doi.org/10.14233/ajchem.2015.18058 Study on Enhancing Zinc Dusts with Direct Sulphuric Acid Leaching H.Y.
Asian Journal of Chemistry; Vol. 27, No. 8 (2015), 2801-2805 ASIAN JOURNAL OF CHEMISTRY http://dx.doi.org/10.14233/ajchem.2015.18058 Study on Enhancing Zinc Dusts with Direct Sulphuric Acid Leaching H.Y.
Differential scanning calorimetry of hydrolysed mangrove tannin
 Polymer International Polym Int 49:574-578 (2000) Differential scanning calorimetry of hydrolysed mangrove tannin S Sowunmi, 1 * RO Ebewele, 2 O Peters 3 and AH Conner 4 1 Department of Chemical Engineering,
Polymer International Polym Int 49:574-578 (2000) Differential scanning calorimetry of hydrolysed mangrove tannin S Sowunmi, 1 * RO Ebewele, 2 O Peters 3 and AH Conner 4 1 Department of Chemical Engineering,
International Journal of Mechanical Engineering and Technology (IJMET) IAEME Scopus
 International Journal of Mechanical Engineering and Technology (IJMET) Volume 5, Issue 2, February 2014, pp. 194 206, Article ID: IJMET_05_02_022 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=5&itype=2
International Journal of Mechanical Engineering and Technology (IJMET) Volume 5, Issue 2, February 2014, pp. 194 206, Article ID: IJMET_05_02_022 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=5&itype=2
Introduction to Membrane
 Heinrich Strathmann Introduction to Membrane Science and Technology WILEY- VCH WILEY-VCH Verlag GmbH & Co. KG aa V Preface Symbols XIII XV 1 Introduction 2 1.1 Overview of Membrane Science and Technology
Heinrich Strathmann Introduction to Membrane Science and Technology WILEY- VCH WILEY-VCH Verlag GmbH & Co. KG aa V Preface Symbols XIII XV 1 Introduction 2 1.1 Overview of Membrane Science and Technology
Supporting Information. Self-assembly in a drying nanofluid droplet: Spontaneous formation of 3D fibre network structures
 Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 2014 Supporting Information Self-assembly in a drying nanofluid droplet: Spontaneous formation of
Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 2014 Supporting Information Self-assembly in a drying nanofluid droplet: Spontaneous formation of
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information (ESI) One-Step Encapsulation and Triggered Release Based on
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information (ESI) One-Step Encapsulation and Triggered Release Based on
International Journal of Chemistry
 Studies on Thermal, Mechanical and Morphological Behaviour of Caprolactam Blocked Methylenediphenyl Diisocyanate and Bismaleimide Modified Epoxy Matrices Daofang Shi Tianjin Polytechnic University NO.63
Studies on Thermal, Mechanical and Morphological Behaviour of Caprolactam Blocked Methylenediphenyl Diisocyanate and Bismaleimide Modified Epoxy Matrices Daofang Shi Tianjin Polytechnic University NO.63
EVALUATION OF EFFERVESCENT FLOATING TABLETS. 6.7 Mathematical model fitting of obtained drug release data
 EVALUATION OF EFFERVESCENT FLOATING TABLETS 6.1 Technological characteristics of floating tablets 6.2 Fourier transform infrared spectroscopy (FT-IR) 6.3 Differential scanning calorimetry (DSC) 6.4 In
EVALUATION OF EFFERVESCENT FLOATING TABLETS 6.1 Technological characteristics of floating tablets 6.2 Fourier transform infrared spectroscopy (FT-IR) 6.3 Differential scanning calorimetry (DSC) 6.4 In
Relating Performance of Thin-Film Composite Forward Osmosis Membranes to Support Layer Formation and Structure
 Relating Performance of Thin-Film Composite Forward Osmosis Membranes to Support Layer Formation and Structure Submitted to Journal of Membrane Science October 1, 2010 Alberto Tiraferri, Ngai Yin Yip,
Relating Performance of Thin-Film Composite Forward Osmosis Membranes to Support Layer Formation and Structure Submitted to Journal of Membrane Science October 1, 2010 Alberto Tiraferri, Ngai Yin Yip,
Supporting information
 S1 Supporting information Biodegradable Injectable Polymer Systems Exhibiting Temperature-Responsive Irreversible Sol-to-Gel Transition by Covalent Bond Formation Yasuyuki YOSHIDA 1,2, Keisuke KAWAHARA
S1 Supporting information Biodegradable Injectable Polymer Systems Exhibiting Temperature-Responsive Irreversible Sol-to-Gel Transition by Covalent Bond Formation Yasuyuki YOSHIDA 1,2, Keisuke KAWAHARA
Fabrication of Nanofiltration Membrane from Polysulfone Ultrafiltration Membrane Via Photo Polymerization
 Fabrication of Nanofiltration Membrane from Polysulfone Ultrafiltration Membrane Via Photo Polymerization A. Akbari 1*, M. Homayoonfal 1 Nanoscience and Nanotechnology Institute, University of Kashan,
Fabrication of Nanofiltration Membrane from Polysulfone Ultrafiltration Membrane Via Photo Polymerization A. Akbari 1*, M. Homayoonfal 1 Nanoscience and Nanotechnology Institute, University of Kashan,
Chapter CHAPTER 7. ELECTRICAL PROPERTIES OF ZnO DOPED MAGESIUM ALUMIUM SILICATE GLASS-CERAMICS
 Chapter 7 102 CHAPTER 7 ELECTRICAL PROPERTIES OF ZnO DOPED MAGESIUM ALUMIUM SILICATE GLASS-CERAMICS Chapter 7 103 CHAPTER 7 ELECTRICAL PROPERTIES OF ZnO DOPED MAGNESIUM ALUMINUM SILICATE GLASS-CERAMICS
Chapter 7 102 CHAPTER 7 ELECTRICAL PROPERTIES OF ZnO DOPED MAGESIUM ALUMIUM SILICATE GLASS-CERAMICS Chapter 7 103 CHAPTER 7 ELECTRICAL PROPERTIES OF ZnO DOPED MAGNESIUM ALUMINUM SILICATE GLASS-CERAMICS
Supporting Information. Tuning the Morphology and Activity of Electrospun. Enhance Chemical Warfare Agent Removal
 Supporting Information Tuning the Morphology and Activity of Electrospun Polystyrene/UiO-66-NH 2 Metal-Organic Framework Composites to Enhance Chemical Warfare Agent Removal Gregory W. Peterson,*,, Annie
Supporting Information Tuning the Morphology and Activity of Electrospun Polystyrene/UiO-66-NH 2 Metal-Organic Framework Composites to Enhance Chemical Warfare Agent Removal Gregory W. Peterson,*,, Annie
Degradation and formation of bisphenol A in polycarbonate used in dentistry
 J Med Dent Sci 2004; 51: 1 6 Original Article Degradation and formation of bisphenol A in polycarbonate used in dentistry Mariko Watanabe Department of Applied Functional Molecules, Division of Biofunctional
J Med Dent Sci 2004; 51: 1 6 Original Article Degradation and formation of bisphenol A in polycarbonate used in dentistry Mariko Watanabe Department of Applied Functional Molecules, Division of Biofunctional
Sulfate Radical-Mediated Degradation of Sulfadiazine by CuFeO 2 Rhombohedral Crystal-Catalyzed Peroxymonosulfate: Synergistic Effects and Mechanisms
 Supporting Information for Sulfate Radical-Mediated Degradation of Sulfadiazine by CuFeO 2 Rhombohedral Crystal-Catalyzed Peroxymonosulfate: Synergistic Effects and Mechanisms Submitted by Yong Feng, Deli
Supporting Information for Sulfate Radical-Mediated Degradation of Sulfadiazine by CuFeO 2 Rhombohedral Crystal-Catalyzed Peroxymonosulfate: Synergistic Effects and Mechanisms Submitted by Yong Feng, Deli
7. SUMMARY, CONCLUSION AND RECOMMENDATIONS
 211 7. SUMMARY, CONCLUSION AND RECOMMENDATIONS Drug absorption from the gastro intestinal tract can be limited by various factors with the most common one being poor aqueous solubility and poor permeability
211 7. SUMMARY, CONCLUSION AND RECOMMENDATIONS Drug absorption from the gastro intestinal tract can be limited by various factors with the most common one being poor aqueous solubility and poor permeability
8 Influence of permeation modulators on the behaviour of a SC lipid model mixture
 8 Influence of permeation modulators on the behaviour of a SC lipid model mixture 8.1 Introduction In the foregoing parts of this thesis, a model membrane system of SC lipids has been developed and characterized.
8 Influence of permeation modulators on the behaviour of a SC lipid model mixture 8.1 Introduction In the foregoing parts of this thesis, a model membrane system of SC lipids has been developed and characterized.
POLYMER REINFORCEMENT
 POLYMER REINFORCEMENT CG2 NanoCoatings Inc. 2007 CG 2 NanoCoatings Inc. 21 Pine Needles Court, Suite 200, Ottawa, ON K2S 1G5 CANADA T 613.435.7747 F 413.638.3933 W www.cg2nanocoatings.com POLYMER REINFORCEMENT
POLYMER REINFORCEMENT CG2 NanoCoatings Inc. 2007 CG 2 NanoCoatings Inc. 21 Pine Needles Court, Suite 200, Ottawa, ON K2S 1G5 CANADA T 613.435.7747 F 413.638.3933 W www.cg2nanocoatings.com POLYMER REINFORCEMENT
Bio-based Building Blocks - Priplast. Taipei, 24 th May 2017
 Bio-based Building Blocks - Priplast Taipei, 24 th May 2017 Bio-based Building Blocks Company introduction Raw materials Technology Features & benefits Conclusion Our History Where We Operate Our Technologies
Bio-based Building Blocks - Priplast Taipei, 24 th May 2017 Bio-based Building Blocks Company introduction Raw materials Technology Features & benefits Conclusion Our History Where We Operate Our Technologies
Supporting Information. Transformation of Framework Solids into Processible Metallo-polymers
 Supporting Information Transformation of Framework Solids into Processible Metallo-polymers Eun-Young Choi, Chunji Gao, Hong-Jun Lee, O-Pil Kwon*, Suck-Hyun Lee* Department of Molecular Science and Technology,
Supporting Information Transformation of Framework Solids into Processible Metallo-polymers Eun-Young Choi, Chunji Gao, Hong-Jun Lee, O-Pil Kwon*, Suck-Hyun Lee* Department of Molecular Science and Technology,
Thermal Properties and Morphology of Biodegradable PLA/Starch Compatibilized Blends
 J. Ind. Eng. Chem., Vol. 13, No. 3, (2007) 457-464 Thermal Properties and Morphology of Biodegradable PLA/Starch Compatibilized Blends Woo Yeul Jang, Boo Young Shin, Tae Jin Lee, and Ramani Narayan* School
J. Ind. Eng. Chem., Vol. 13, No. 3, (2007) 457-464 Thermal Properties and Morphology of Biodegradable PLA/Starch Compatibilized Blends Woo Yeul Jang, Boo Young Shin, Tae Jin Lee, and Ramani Narayan* School
Supporting Information. Scalable Chitosan-Graphene Oxide Membranes: The Effect of GO Size on. Properties and Cross-Flow Filtration Performance
 Supporting Information Scalable Chitosan-Graphene Oxide Membranes: The Effect of GO Size on Properties and Cross-Flow Filtration Performance Mojtaba Abolhassani, a Chris S. Griggs, b Luke A. Gurtowski,
Supporting Information Scalable Chitosan-Graphene Oxide Membranes: The Effect of GO Size on Properties and Cross-Flow Filtration Performance Mojtaba Abolhassani, a Chris S. Griggs, b Luke A. Gurtowski,
Advances in Environmental Biology
 AENSI Journals Advances in Environmental Biology ISSN-1995-0756 EISSN-1998-1066 Journal home page: http://www.aensiweb.com/aeb/ Investigation of Optimum Drying Conditions for Pure PES Membranes for Gas
AENSI Journals Advances in Environmental Biology ISSN-1995-0756 EISSN-1998-1066 Journal home page: http://www.aensiweb.com/aeb/ Investigation of Optimum Drying Conditions for Pure PES Membranes for Gas
P-STARCH-15 Gelatinization and retrogradation properties of hypochlorite-oxidized cassava starch
 P-STARCH-15 Gelatinization and retrogradation properties of hypochlorite-oxidized cassava starch Kunruedee Sangseethong a, Niti Termvejsayanon a and Klanarong Sriroth b,c a Cassava and Starch Technology
P-STARCH-15 Gelatinization and retrogradation properties of hypochlorite-oxidized cassava starch Kunruedee Sangseethong a, Niti Termvejsayanon a and Klanarong Sriroth b,c a Cassava and Starch Technology
PREPARATION AND PROPERTIES OF MODIFIED CARBOXYLMETHYL CELLULOSE WITH CASSAVA STARCH
 E_E0011 1 PREPARATIN AND PRPERTIES F MDIFIED CARBXYLMETHYL CELLULSE WITH CASSAVA STARCH Janthanipa Nuim, Sa-Ad Riyajan * Department of Materials Science and Technology and Natural Products Research Center,
E_E0011 1 PREPARATIN AND PRPERTIES F MDIFIED CARBXYLMETHYL CELLULSE WITH CASSAVA STARCH Janthanipa Nuim, Sa-Ad Riyajan * Department of Materials Science and Technology and Natural Products Research Center,
Preparation and Application of SPPEES-TiO 2 Composite Micro-porous UF Membrane for Refinery Effluent Treatment
 International Journal of Environmental Research and Development. ISSN 2249-3131 Volume 4, Number 2 (2014), pp. 147-152 Research India Publications http://www.ripublication.com/ijerd.htm Preparation and
International Journal of Environmental Research and Development. ISSN 2249-3131 Volume 4, Number 2 (2014), pp. 147-152 Research India Publications http://www.ripublication.com/ijerd.htm Preparation and
Measuring Lipid Composition LC-MS/MS
 Project: Measuring Lipid Composition LC-MS/MS Verification of expected lipid composition in nanomedical controlled release systems by liquid chromatography tandem mass spectrometry AUTHORED BY: DATE: Sven
Project: Measuring Lipid Composition LC-MS/MS Verification of expected lipid composition in nanomedical controlled release systems by liquid chromatography tandem mass spectrometry AUTHORED BY: DATE: Sven
CHAPTER 4 EFFECT OF OXALIC ACID ON THE OPTICAL, THERMAL, DIELECTRIC AND MECHANICAL BEHAVIOUR OF ADP CRYSTALS
 67 CHAPTER 4 EFFECT OF OXALIC ACID ON THE OPTICAL, THERMAL, DIELECTRIC AND MECHANICAL BEHAVIOUR OF ADP CRYSTALS 4.1 INTRODUCTION Oxalic acid is a hydrogen-bonded material. It is the only possible compound
67 CHAPTER 4 EFFECT OF OXALIC ACID ON THE OPTICAL, THERMAL, DIELECTRIC AND MECHANICAL BEHAVIOUR OF ADP CRYSTALS 4.1 INTRODUCTION Oxalic acid is a hydrogen-bonded material. It is the only possible compound
A quantitative study of chemical kinetics for the synthesis of. doped oxide nanocrystals using FTIR spectroscopy
 Supporting information A quantitative study of chemical kinetics for the synthesis of doped oxide nanocrystals using FTIR spectroscopy Na Zhang, 1,2 Xin Wang, 1 Zhizhen Ye 1 and Yizheng Jin, 1,3* 1 State
Supporting information A quantitative study of chemical kinetics for the synthesis of doped oxide nanocrystals using FTIR spectroscopy Na Zhang, 1,2 Xin Wang, 1 Zhizhen Ye 1 and Yizheng Jin, 1,3* 1 State
Engineering the Growth of TiO 2 Nanotube Arrays on Flexible Carbon Fibre Sheets
 Engineering the Growth of TiO 2 Nanotube Arrays on Flexible Carbon Fibre Sheets Peng Chen, a Li Gu, b Xiudong Xue, a Mingjuan Li a and Xuebo Cao* a a Key Lab of Organic Synthesis of Jiangsu Province and
Engineering the Growth of TiO 2 Nanotube Arrays on Flexible Carbon Fibre Sheets Peng Chen, a Li Gu, b Xiudong Xue, a Mingjuan Li a and Xuebo Cao* a a Key Lab of Organic Synthesis of Jiangsu Province and
Supporting Information
 Supporting Information Polymer Micelles Stabilization-n-Demand through Reversible Photocrosslinking 1. Synthesis and Characterization of Diblock Copolymers Materials. Cu(I)Br, 2-Bromo-2-methylpropionyl
Supporting Information Polymer Micelles Stabilization-n-Demand through Reversible Photocrosslinking 1. Synthesis and Characterization of Diblock Copolymers Materials. Cu(I)Br, 2-Bromo-2-methylpropionyl
Introduction of innovate membranes in water-treatment
 Introduction of innovate membranes in water-treatment 2013. 11. 12 Young June Won Water Environment-Membrane Technology Lab. School of Chemical and Biological Engineering, Seoul National University, Korea
Introduction of innovate membranes in water-treatment 2013. 11. 12 Young June Won Water Environment-Membrane Technology Lab. School of Chemical and Biological Engineering, Seoul National University, Korea
Original Research Biodegradation and Characterization Studies of Different Kinds of Polyurethanes with Several Enzyme Solutions
 Pol. J. Environ. Stud. Vol. 21, No. 6 (2012, 1777-1782 Original Research Biodegradation and Characterization Studies of Different Kinds of Polyurethanes with Several Enzyme Solutions Erhan Ozsagıroglu*,
Pol. J. Environ. Stud. Vol. 21, No. 6 (2012, 1777-1782 Original Research Biodegradation and Characterization Studies of Different Kinds of Polyurethanes with Several Enzyme Solutions Erhan Ozsagıroglu*,
Hybrid polymer/lipid vesicles: fine control of the lipid and polymer distribution in the binary membrane
 SUPPORTING INFORMATIONS FOR Hybrid polymer/lipid vesicles: fine control of the lipid and polymer distribution in the binary membrane Maud Chemin, a,b Pierre-Marie Brun, a,b Sébastien Lecommandoux, a,b
SUPPORTING INFORMATIONS FOR Hybrid polymer/lipid vesicles: fine control of the lipid and polymer distribution in the binary membrane Maud Chemin, a,b Pierre-Marie Brun, a,b Sébastien Lecommandoux, a,b
Supporting Information
 Supporting Information Advanced ultrafiltration membranes based on functionalized poly(arylene ether sulfone) block copolymers Mahendra Kumar and Mathias Ulbricht* Lehrstuhl für Technische Chemie II, Universität
Supporting Information Advanced ultrafiltration membranes based on functionalized poly(arylene ether sulfone) block copolymers Mahendra Kumar and Mathias Ulbricht* Lehrstuhl für Technische Chemie II, Universität
EFFECT OF COMPRESSED CO 2 ON THE PROPERTIES OF AOT IN ISOOCTANE REVERSE MICELLAR SOLUTION AND ITS APPLICATION TO RECOVER NANOPARTICLES
 EFFECT OF COMPRESSED CO 2 ON THE PROPERTIES OF AOT IN ISOOCTANE REVERSE MICELLAR SOLUTION AND ITS APPLICATION TO RECOVER NANOPARTICLES Dongxia Liu, Jianling Zhang, Tiancheng Mu, Jing Chen, Weize Wu, Jun
EFFECT OF COMPRESSED CO 2 ON THE PROPERTIES OF AOT IN ISOOCTANE REVERSE MICELLAR SOLUTION AND ITS APPLICATION TO RECOVER NANOPARTICLES Dongxia Liu, Jianling Zhang, Tiancheng Mu, Jing Chen, Weize Wu, Jun
THE USE OF ARABIC GUM AS AN ADDITIVE TO POLYSULFONE MEMBRANES. Abstract
 THE USE OF ARABIC GUM AS AN ADDITIVE TO POLYSULFONE MEMBRANES Yehia Manawi, Viktor Kochkodan, Muataz Atieh* Qatar Environment and Energy Research Institute (QEERI), Hamad bin Khalifa University (HBKU),
THE USE OF ARABIC GUM AS AN ADDITIVE TO POLYSULFONE MEMBRANES Yehia Manawi, Viktor Kochkodan, Muataz Atieh* Qatar Environment and Energy Research Institute (QEERI), Hamad bin Khalifa University (HBKU),
Basic Principles of Membrane Technology
 Basic Principles of Membrane Technology Basic Principles of Membrane Technology by Marcel Mulder Center for Membrane Science and Technology, University oftwente, Enschede, The Netherlands KLUWER ACADEMIC
Basic Principles of Membrane Technology Basic Principles of Membrane Technology by Marcel Mulder Center for Membrane Science and Technology, University oftwente, Enschede, The Netherlands KLUWER ACADEMIC
Comprehensive Study of SLE as a Sample. Preparation Tool for Bioanalysis
 Comprehensive Study of SLE as a Sample Preparation Tool for Bioanalysis Wan Wang, Warren Chen, Jerry Wang Bonna-Agela Technologies 179 Southern Street, West TEDA, Tianjin, China Abstract A simple, fast,
Comprehensive Study of SLE as a Sample Preparation Tool for Bioanalysis Wan Wang, Warren Chen, Jerry Wang Bonna-Agela Technologies 179 Southern Street, West TEDA, Tianjin, China Abstract A simple, fast,
Polybenzimidazole block sulfonated poly(arylene ether sulfone) ionomers
 Supplementary information Polybenzimidazole block sulfonated poly(arylene ether sulfone) ionomers Feifei Ng, a Byungchan Bae, a Kenji Miyatake* a,b and Masahiro Watanabe* a a Fuel Cell Nanomaterials Center
Supplementary information Polybenzimidazole block sulfonated poly(arylene ether sulfone) ionomers Feifei Ng, a Byungchan Bae, a Kenji Miyatake* a,b and Masahiro Watanabe* a a Fuel Cell Nanomaterials Center
Properties of Oxidized Cassava Starch as Influenced by Oxidant Concentration and Reaction Time
 Properties of Oxidized Cassava Starch as Influenced by Oxidant Concentration and Reaction Time P-STARCH-26 Kunruedee Sangseethong 1 and Klanarong Sriroth 2,3 1 Cassava and Starch Technology Research Unit,
Properties of Oxidized Cassava Starch as Influenced by Oxidant Concentration and Reaction Time P-STARCH-26 Kunruedee Sangseethong 1 and Klanarong Sriroth 2,3 1 Cassava and Starch Technology Research Unit,
Synthesis of Polyesters from Azelaic Acid and Glycerol by Polycondensation Reaction
 Synthesis of Polyesters from Azelaic Acid and Glycerol by Polycondensation Reaction Thitipat Chongcharoenchaikul 1, Patchanita Thamyongkit 2 and Sirilux Poompradub 1* 1 Department of Chemical Technology,
Synthesis of Polyesters from Azelaic Acid and Glycerol by Polycondensation Reaction Thitipat Chongcharoenchaikul 1, Patchanita Thamyongkit 2 and Sirilux Poompradub 1* 1 Department of Chemical Technology,
Fe-doped ZnO synthesized by parallel flow precipitation process for improving photocatalytic activity
 IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Fe-doped ZnO synthesized by parallel flow precipitation process for improving photocatalytic activity To cite this article: Q
IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Fe-doped ZnO synthesized by parallel flow precipitation process for improving photocatalytic activity To cite this article: Q
Sokalan PA types. Sokalan PA 15 Sokalan PA 40. Sokalan PA 20 Sokalan PA 50. Sokalan PA 25 PN Granules Sokalan PA 80 S. Technical Information
 Technical Information Sokalan PA types TI/ES 1151 e July 1992 (DFC) Supersedes TI/P 3037 e dated May 1988 Sokalan PA 13 PN Sokalan PA 30 CL Sokalan PA 15 Sokalan PA 40 Sokalan PA 20 PN Sokalan PA 40 Powder
Technical Information Sokalan PA types TI/ES 1151 e July 1992 (DFC) Supersedes TI/P 3037 e dated May 1988 Sokalan PA 13 PN Sokalan PA 30 CL Sokalan PA 15 Sokalan PA 40 Sokalan PA 20 PN Sokalan PA 40 Powder
# Supplementary Material (ESI) for Molecular BioSystems # This journal is The Royal Society of Chemistry 2005
 Supporting Information Multifunctional Polymeric Micelles with Folate-Mediated Cancer Cell Targeting and ph-triggered Drug Releasing Properties for Active Intracellular Drug Delivery Younsoo Bae, Woo-Dong
Supporting Information Multifunctional Polymeric Micelles with Folate-Mediated Cancer Cell Targeting and ph-triggered Drug Releasing Properties for Active Intracellular Drug Delivery Younsoo Bae, Woo-Dong
MINIMIZING MORPHOLOGICAL DEFECTS OF PEI HOLLOW FIBRE MEMBRANE BY OPTIMIZING THE DOPE VISCOSITY
 MINIMIZING MORPHOLOGICAL DEFECTS OF PEI HOLLOW FIBRE MEMBRANE BY OPTIMIZING THE DOPE VISCOSITY Asif Jamil, Oh Pei Ching and Azmi M. Shariff Department of Chemical Engineering, Universiti Teknologi PETRONAS,
MINIMIZING MORPHOLOGICAL DEFECTS OF PEI HOLLOW FIBRE MEMBRANE BY OPTIMIZING THE DOPE VISCOSITY Asif Jamil, Oh Pei Ching and Azmi M. Shariff Department of Chemical Engineering, Universiti Teknologi PETRONAS,
The Effect of Molecular Weight of Polycaprolactone on the Ester Interchange Reactions during Melt Blending with Poly(ethylene terephthalate)
 Polymer Journal, Vol. 34, No. 5, pp 313 319 (2002) The Effect of Molecular Weight of Polycaprolactone on the Ester Interchange Reactions during Melt Blending with Poly(ethylene terephthalate) Kyung Yul
Polymer Journal, Vol. 34, No. 5, pp 313 319 (2002) The Effect of Molecular Weight of Polycaprolactone on the Ester Interchange Reactions during Melt Blending with Poly(ethylene terephthalate) Kyung Yul
CHAPTER VI FACTORIAL STUDIES ON THE EFFECTS OF CYCLODEXTRINS AND SOLUTOL HS15 ON THE SOLUBILITY AND DISSOLUTION RATE OF EFAVIRENZ AND RITONAVIR
 CHAPTER VI FACTORIAL STUDIES ON THE EFFECTS OF CYCLODEXTRINS AND SOLUTOL HS15 ON THE SOLUBILITY AND DISSOLUTION RATE OF EFAVIRENZ AND RITONAVIR Efavirenz and ritonavir, two widely prescribed anti retroviral
CHAPTER VI FACTORIAL STUDIES ON THE EFFECTS OF CYCLODEXTRINS AND SOLUTOL HS15 ON THE SOLUBILITY AND DISSOLUTION RATE OF EFAVIRENZ AND RITONAVIR Efavirenz and ritonavir, two widely prescribed anti retroviral
Starch-PVA composite films: towards a new generation of biodegradable packaging material
 Starch-PVA composite films: towards a new generation of biodegradable packaging material Amalia Cano, Maite Chafér, Amparo Chiralt and Chelo González Institute of Food Engineering for the Development (IIAD)
Starch-PVA composite films: towards a new generation of biodegradable packaging material Amalia Cano, Maite Chafér, Amparo Chiralt and Chelo González Institute of Food Engineering for the Development (IIAD)
Xianren Zhang ( 张现仁 )
 The interaction between nanoparticles and membranes: from cytotoxicity to drug delivery Xianren Zhang ( 张现仁 ) zhangxr@mail.buct.edu.cn State Key Laboratory of Organic-Inorganic Composites, Beijing University
The interaction between nanoparticles and membranes: from cytotoxicity to drug delivery Xianren Zhang ( 张现仁 ) zhangxr@mail.buct.edu.cn State Key Laboratory of Organic-Inorganic Composites, Beijing University
Chemical Surface Transformation 1
 Chemical Surface Transformation 1 Chemical reactions at Si H surfaces (inorganic and organic) can generate very thin films (sub nm thickness up to µm): inorganic layer formation by: thermal conversion:
Chemical Surface Transformation 1 Chemical reactions at Si H surfaces (inorganic and organic) can generate very thin films (sub nm thickness up to µm): inorganic layer formation by: thermal conversion:
Supporting Information. Zinc hydroxide nanostrands: unique precursor for ZIF-8. thin membranes toward highly size-sieving gas separation
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Zinc hydroxide nanostrands: unique precursor for ZIF-8 thin membranes
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Zinc hydroxide nanostrands: unique precursor for ZIF-8 thin membranes
XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES.
 XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES. II. THE PREPARATION OF GLUCOSE UREIDE. BY ALEXANDER HYND. From the Department of Physiology, University of St Andrews. (Received
XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES. II. THE PREPARATION OF GLUCOSE UREIDE. BY ALEXANDER HYND. From the Department of Physiology, University of St Andrews. (Received
Cellulose Fibers and Microcellular Foam Starch Composites
 Cellulose Fibers and Microcellular Foam Starch Composites Richard A. Venditti*, Joel J. Pawlak, Andrew R. Rutledge, Janderson L. Cibils Forest Biomaterials Science and Engineering NC State University,
Cellulose Fibers and Microcellular Foam Starch Composites Richard A. Venditti*, Joel J. Pawlak, Andrew R. Rutledge, Janderson L. Cibils Forest Biomaterials Science and Engineering NC State University,
DRAFT PROPOSAL FOR THE INTERNATIONAL PHARMACOPOEIA: CARBAMAZEPINI COMPRESSI - CARBAMAZEPINE TABLETS
 December 2015 Draft document for comment 1 2 3 4 5 6 DRAFT PROPOSAL FOR THE INTERNATIONAL PHARMACOPOEIA: CARBAMAZEPINI COMPRESSI - CARBAMAZEPINE TABLETS (December 2015) REVISED DRAFT FOR COMMENT Should
December 2015 Draft document for comment 1 2 3 4 5 6 DRAFT PROPOSAL FOR THE INTERNATIONAL PHARMACOPOEIA: CARBAMAZEPINI COMPRESSI - CARBAMAZEPINE TABLETS (December 2015) REVISED DRAFT FOR COMMENT Should
CHEMICALLY MODIFIED LIGNIN A POTENTIAL RESOURCE MATERIAL FOR COMPOSITES WITH BETTER STABILITY
 International Journal of Science, Environment and Technology, Vol. 4, No 1, 2015, 183 189 ISSN 2278-3687 (O) CHEMICALLY MODIFIED LIGNIN A POTENTIAL RESOURCE MATERIAL FOR COMPOSITES WITH BETTER STABILITY
International Journal of Science, Environment and Technology, Vol. 4, No 1, 2015, 183 189 ISSN 2278-3687 (O) CHEMICALLY MODIFIED LIGNIN A POTENTIAL RESOURCE MATERIAL FOR COMPOSITES WITH BETTER STABILITY
The Influence of PEG400 and Acetone on Polysulfone Membrane Morphology and Fouling Behaviour
 J. Eng. Technol. Sci., Vol. 48, No. 2, 2016, 135-149 135 The Influence of PEG400 and Acetone on Polysulfone Membrane Morphology and Fouling Behaviour P.T.P. Aryanti, Shelli R. Joscarita, Anita K. Wardani,
J. Eng. Technol. Sci., Vol. 48, No. 2, 2016, 135-149 135 The Influence of PEG400 and Acetone on Polysulfone Membrane Morphology and Fouling Behaviour P.T.P. Aryanti, Shelli R. Joscarita, Anita K. Wardani,
4. Monitoring of dissolution induced changes in film coat
 4. Monitoring of dissolution induced changes in film coat composition 4.1 Introduction As membrane controlled drug delivery coatings are subjected to changes in coating composition, it is necessary to
4. Monitoring of dissolution induced changes in film coat composition 4.1 Introduction As membrane controlled drug delivery coatings are subjected to changes in coating composition, it is necessary to
Analysis of the Non-Ionic Surfactant Triton-X Using UltraPerformance Convergence Chromatography (UPC 2 ) with MS and UV Detection
 Analysis of the Non-Ionic Surfactant Triton-X Using UltraPerformance Convergence Chromatography (UPC 2 ) with MS and UV Detection Jane Cooper, 1 Baiba Cabovska 2 1 Waters Corporation, Wilmslow, UK 2 Waters
Analysis of the Non-Ionic Surfactant Triton-X Using UltraPerformance Convergence Chromatography (UPC 2 ) with MS and UV Detection Jane Cooper, 1 Baiba Cabovska 2 1 Waters Corporation, Wilmslow, UK 2 Waters
Aeris. Precision Engineered Core- Shell Particles for Ultra-High Resolution BioSeparations. Aeris PEPTIDE. Aeris WIDEPORE
 Aeris Precision Engineered Core- Shell Particles for Ultra-High Resolution BioSeparations Aeris is a specialized line of reversed phase core-shell UHPLC columns, built exclusively for the ultra-high performance
Aeris Precision Engineered Core- Shell Particles for Ultra-High Resolution BioSeparations Aeris is a specialized line of reversed phase core-shell UHPLC columns, built exclusively for the ultra-high performance
Colloid Chemistry. Lecture #2 Association colloid
 Colloid Chemistry Lecture #2 Association colloid 1 https://ilustracionmedica.wordpress.com/2014/08/27/fisicos-haciendo-medicina-john-tyndall/ Solution Classical vs. Colloid solution Tyndall effect Increased
Colloid Chemistry Lecture #2 Association colloid 1 https://ilustracionmedica.wordpress.com/2014/08/27/fisicos-haciendo-medicina-john-tyndall/ Solution Classical vs. Colloid solution Tyndall effect Increased
Oil Soluble Silicones. Southeast Chapter March 19, 2015
 Oil Soluble Silicones Southeast Chapter March 19, 2015 1 Background! Silicone compounds have been known since 1860, but were of little commercial interest until the 1940's. 2 Background! Silicone compounds
Oil Soluble Silicones Southeast Chapter March 19, 2015 1 Background! Silicone compounds have been known since 1860, but were of little commercial interest until the 1940's. 2 Background! Silicone compounds
Journal of Chemical and Pharmaceutical Research, 2015, 7(8): Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 215, 7(8):257-261 Research Article ISSN : 975-7384 CODEN(USA) : JCPRC5 Pulping process for rice straw in basic ionic liquid
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 215, 7(8):257-261 Research Article ISSN : 975-7384 CODEN(USA) : JCPRC5 Pulping process for rice straw in basic ionic liquid
Bio-based Solutions for High-Performance Polyurethane Adhesives. Angela Smits Bio-based Adhesives Conference 2013
 Bio-based Solutions for High-Performance Polyurethane Adhesives Angela Smits Bio-based Adhesives Conference 213 Bio-based building blocks sustainable solutions CRODA: Naturally derived speciality chemicals
Bio-based Solutions for High-Performance Polyurethane Adhesives Angela Smits Bio-based Adhesives Conference 213 Bio-based building blocks sustainable solutions CRODA: Naturally derived speciality chemicals
H 2 O. Liquid, solid, and vapor coexist in the same environment
 Water H 2 O Liquid, solid, and vapor coexist in the same environment WATER MOLECULES FORM HYDROGEN BONDS Water is a fundamental requirement for life, so it is important to understand the structural and
Water H 2 O Liquid, solid, and vapor coexist in the same environment WATER MOLECULES FORM HYDROGEN BONDS Water is a fundamental requirement for life, so it is important to understand the structural and
Efficient Metal-Free Pathway to Vinyl Thioesters with Calcium Carbide as the Acetylene Source
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information Efficient Metal-Free Pathway to Vinyl Thioesters with Calcium Carbide
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information Efficient Metal-Free Pathway to Vinyl Thioesters with Calcium Carbide
Supporting information for the manuscript
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Supporting information for the manuscript Toward enhanced photoactivity
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Supporting information for the manuscript Toward enhanced photoactivity
NANOSTRUCTURAL ZnO FABRICATION BY VAPOR-PHASE TRANSPORT IN AIR
 International Journal of Modern Physics B Vol. 18, No. 0 (2004) 1 8 c World Scientific Publishing Company NANOSTRUCTURAL ZnO FABRICATION BY VAPOR-PHASE TRANSPORT IN AIR C. X. XU, X. W. SUN, B. J. CHEN,
International Journal of Modern Physics B Vol. 18, No. 0 (2004) 1 8 c World Scientific Publishing Company NANOSTRUCTURAL ZnO FABRICATION BY VAPOR-PHASE TRANSPORT IN AIR C. X. XU, X. W. SUN, B. J. CHEN,
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Artepillin C, is it a good marker for quality control of Brazilian Green Propolis? Cui-ping Zhang 1, Xiao-ge Shen 1, Jia-wei Chen 1, Xia-sen Jiang 1, Kai Wang 2, Fu-liang Hu 1 *
SUPPLEMENTARY MATERIAL Artepillin C, is it a good marker for quality control of Brazilian Green Propolis? Cui-ping Zhang 1, Xiao-ge Shen 1, Jia-wei Chen 1, Xia-sen Jiang 1, Kai Wang 2, Fu-liang Hu 1 *
Effect of Rejection Performance on Hollow Fiber Composite Reverse Osmosis Membrane by Alcohols Additives
 Effect of Rejection Performance on Hollow Fiber Composite Reverse Osmosis Membrane by Alcohols Additives Xinghua Wang & Wei Wang Key Laboratory of Hollow Fiber Membrane Material and Membrane Process Tianjin
Effect of Rejection Performance on Hollow Fiber Composite Reverse Osmosis Membrane by Alcohols Additives Xinghua Wang & Wei Wang Key Laboratory of Hollow Fiber Membrane Material and Membrane Process Tianjin
ANALYSIS OF STARCH USING THE ALLIANCE GPCV2000 SYSTEM
 ANALYSIS OF STARCH USING THE ALLIANCE GPCV2000 SYSTEM Jinchuan Yang and Alice J. Di Gioia Waters Corporation, 34 Maple Street, Milford, MA 01757 INTRODUCTION Starch forms a major part of the plant food
ANALYSIS OF STARCH USING THE ALLIANCE GPCV2000 SYSTEM Jinchuan Yang and Alice J. Di Gioia Waters Corporation, 34 Maple Street, Milford, MA 01757 INTRODUCTION Starch forms a major part of the plant food
TABLE I 24-hr Annealing: Effect of Annealing Temperature (0 C) on Gelatinization Characteristics of Normal and Waxy Maize Starch Primary Endotherm
 for measurement of starch gelatinization, using differential scanning calorimetry (DSC). DSC endotherms for cereal starches were also reported by Stevens and Elton (1971). DSC data were interpreted in
for measurement of starch gelatinization, using differential scanning calorimetry (DSC). DSC endotherms for cereal starches were also reported by Stevens and Elton (1971). DSC data were interpreted in
Formation of optimal surface of dental implants by CST SLA Process
 Formation of optimal surface of dental implants by CST SLA Process Research Report of Finish Line Materials & Processes Ltd www.finishlinemp.com with SEM-EDS analysis at Technion Research and Development
Formation of optimal surface of dental implants by CST SLA Process Research Report of Finish Line Materials & Processes Ltd www.finishlinemp.com with SEM-EDS analysis at Technion Research and Development
INFLUENCE OF ADDITIVES IN SPINNING DOPE AND SPIN BATH TEMPERATURES ON STRUCTURE AND PROPERTIES OF ACRYLONITRILE TERPOLYMER FIBERS
 INFLUENCE OF ADDITIVES IN SPINNING DOPE AND SPIN BATH TEMPERATURES ON STRUCTURE AND PROPERTIES OF ACRYLONITRILE TERPOLYMER FIBERS by M. SURYA KUMARI DEPARTMENT OF TEXTILE TECHNOLOGY Submitted in fulfilment
INFLUENCE OF ADDITIVES IN SPINNING DOPE AND SPIN BATH TEMPERATURES ON STRUCTURE AND PROPERTIES OF ACRYLONITRILE TERPOLYMER FIBERS by M. SURYA KUMARI DEPARTMENT OF TEXTILE TECHNOLOGY Submitted in fulfilment
Supporting Information
 S1 Supporting Information Efficient productions of 5-hydroxymethylfurfural and alkyl levulinate from biomass carbohydrate using ionic liquid-based polyoxometalate salts Jinzhu Chen,* a Guoying Zhao a and
S1 Supporting Information Efficient productions of 5-hydroxymethylfurfural and alkyl levulinate from biomass carbohydrate using ionic liquid-based polyoxometalate salts Jinzhu Chen,* a Guoying Zhao a and
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information ovel pseudo[2]rotaxanes constructed by selfassembly of dibenzyl
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information ovel pseudo[2]rotaxanes constructed by selfassembly of dibenzyl
Bio Ionic Liquids: Room Temperature Ionic Liquids Composed Wholly of Biomaterials
 SUPPORTING INFORMATION Bio Ionic Liquids: Room Temperature Ionic Liquids Composed Wholly of Biomaterials Yukinobu Fukaya a, Yoshiki Iizuka a, Kenta Sekikawa a, and Hiroyuki Ohno a * a Department of Biotechnology,
SUPPORTING INFORMATION Bio Ionic Liquids: Room Temperature Ionic Liquids Composed Wholly of Biomaterials Yukinobu Fukaya a, Yoshiki Iizuka a, Kenta Sekikawa a, and Hiroyuki Ohno a * a Department of Biotechnology,
Understanding the Variables that Define Tg for Kraft Lignin
 Understanding the Variables that Define Tg for Kraft Lignin Hasan Sadeghifar, Dimitris S. Argyropoulos Departments of Chemistry and Forest Biomaterials North Carolina State University Raleigh, NC USA Objective
Understanding the Variables that Define Tg for Kraft Lignin Hasan Sadeghifar, Dimitris S. Argyropoulos Departments of Chemistry and Forest Biomaterials North Carolina State University Raleigh, NC USA Objective
Thermal Properties and Moisture Absorption of LDPE/Banana Starch Biocomposite Films
 Journal of Metals, Materials and Minerals. Vol. 12 No. 1 pp. 1-1, 22 Thermal Properties and Moisture Absorption of LDPE/Banana Starch Biocomposite Films Duangdao AHT-ONG* and Kanjana CHAROENKONGTHUM Department
Journal of Metals, Materials and Minerals. Vol. 12 No. 1 pp. 1-1, 22 Thermal Properties and Moisture Absorption of LDPE/Banana Starch Biocomposite Films Duangdao AHT-ONG* and Kanjana CHAROENKONGTHUM Department
The Role of Physical Properties Data in Product Development
 The Role of Physical Properties Data in Product Development From Molecules to Market Gent June 18 th and 19 th 2008 Eckhard Flöter Unilever R&D Vlaardingen Physical Properties Data Melting point, heat
The Role of Physical Properties Data in Product Development From Molecules to Market Gent June 18 th and 19 th 2008 Eckhard Flöter Unilever R&D Vlaardingen Physical Properties Data Melting point, heat
Keywords: Additives, asymmetric membrane, cellulose acetate, PEG
 Research Journal of Applied Sciences, Engineering and Technology 7(18): 3852-3859, 2014 DOI:10.19026/rjaset.7.742 ISSN: 2040-7459; e-issn: 2040-7467 2014 Maxwell Scientific Publication Corp. Submitted:
Research Journal of Applied Sciences, Engineering and Technology 7(18): 3852-3859, 2014 DOI:10.19026/rjaset.7.742 ISSN: 2040-7459; e-issn: 2040-7467 2014 Maxwell Scientific Publication Corp. Submitted:
Supporting Information for. Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of. 3,5-Disubstituted Pyridines: Mechanistic Studies
 Supporting Information for Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of 3,5-Disubstituted Pyridines: Mechanistic Studies Ta-Hsien Chuang* a, Yu-Chi Chen b and Someshwar Pola
Supporting Information for Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of 3,5-Disubstituted Pyridines: Mechanistic Studies Ta-Hsien Chuang* a, Yu-Chi Chen b and Someshwar Pola
Albert J.J. Woortman, Peter A.M. Steeneken Thermoreversible particle gels based on physically modified starch
 Albert J.J. Woortman, Peter A.M. Steeneken Thermoreversible particle gels based on physically modified starch TNO Nutrition and Food Research Stärke-Tagung, Detmold, 21-23 April 2004 1 TOPICS FOR TODAY
Albert J.J. Woortman, Peter A.M. Steeneken Thermoreversible particle gels based on physically modified starch TNO Nutrition and Food Research Stärke-Tagung, Detmold, 21-23 April 2004 1 TOPICS FOR TODAY

 &+$37(5,, ;IJ;H?
&+$37(5,, ;IJ;H?