Section: Medicine Last Reviewed Date: October Policy No: 77 Effective Date: January 1, 2014
|
|
|
- Alexandrina Boyd
- 5 years ago
- Views:
Transcription
1 Medical Policy Manual Topic: Autologous Blood-Derived Growth Factors as a Treatment for Wound Healing and Other Miscellaneous Conditions Date of Origin: November 1999 Section: Medicine Last Reviewed Date: October 2013 Policy No: 77 Effective Date: January 1, 2014 IMPORTANT REMINDER Medical Policies are developed to provide guidance for members and providers regarding coverage in accordance with contract terms. Benefit determinations are based in all cases on the applicable contract language. To the extent there may be any conflict between the Medical Policy and contract language, the contract language takes precedence. PLEASE NOTE: Contracts exclude from coverage, among other things, services or procedures that are considered investigational or cosmetic. Providers may bill members for services or procedures that are considered investigational or cosmetic. Providers are encouraged to inform members before rendering such services that the members are likely to be financially responsible for the cost of these services. DESCRIPTION A variety of growth factors have been found to play a role in wound healing, including platelet-derived growth factor (PDGF), epidermal growth factor, fibroblast growth factors, transforming growth factors, and insulin-like growth factor. Topically applied autologous platelet-derived growth factors have been most extensively investigated for clinical use in wound healing. For example, platelets are a rich source of PDGFs, transforming growth factors (which function as a mitogen for fibroblasts, smooth muscle cells, and osteoblasts) and vascular endothelial growth factors. Autologous platelet concentrate suspended in plasma, also known as platelet-rich plasma (PRP) or buffy coat, can be prepared from samples of centrifuged autologous blood. Exposure to a solution of thrombin and calcium chloride results in the polymerization of fibrin from fibrinogen, creating a platelet gel. The platelet gel can then be applied to wounds or may be used as an adjunct to surgery to promote hemostasis and accelerate healing. Activated platelets then degranulate, releasing the various growth factors. Regulatory Status There are a number of commercially available centrifugation devices used for the preparation of PRP. For example, AutoloGel (Cytomedix) and SafeBlood (SafeBlood Technologies) are two related but 1 MED77
2 distinct autologous blood-derived preparations that can be prepared at the bedside for immediate application. Both AutoloGel and SafeBlood have been specifically marketed for wound healing. Other devices may be used in the operating room setting, such as Medtronic Electromedic, Elmd-500 Autotransfusion system, the Plasma Saver device, or the Smart PreP device. In the operating room setting, PRP has been investigated as an adjunct to a variety of periodontal, reconstructive, and orthopedic procedures. In addition, platelet-rich plasma has also been proposed as a primary treatment of miscellaneous conditions such as epicondylitis, plantar fasciitis and Dupuytren s contracture. The Magellan Autologous Platelet Separator System (Medtronic) includes a disposables kit designed for use with the Magellan Autologous Platelet Separator portable tabletop centrifuge. BioMet Biologics received marketing clearance through the FDA s 510(k) process for a gravitational platelet separation system (GPS II), which uses a disposable separation tube for centrifugation and a dual cannula tip to mix the platelets and thrombin at the surgical site. Filtration or plasmapheresis may also be used to produce platelet-rich concentrates. The use of different devices and procedures can lead to variable concentrations of active platelets and associated proteins, increasing variability between studies of clinical efficacy. Platelet-rich plasma must be distinguished from fibrin glues or sealants, which have been used for many years as a surgical adjunct to promote local hemostasis at incision sites. Autologous fibrin glue or sealants can be created from platelet-poor plasma, and consists primarily of fibrinogen. Commercial fibrin glues are created from pooled homologous human donors; Tisseel (Baxter) and Hemaseal are examples of commercially available fibrin sealants. This policy does not address the use of fibrin sealants. Note: This policy is not intended to address Regranex (becaplermin gel), which is not an autologous platelet-derived growth factor. MEDICAL POLICY CRITERIA Autologous blood-derived growth factors (i.e. platelet rich plasma) are considered investigational for all indications including but not limited to: A. Wounds, including but not limited to: 1. Acute traumatic or surgical wounds 2. Chronic non-healing wounds B. Disorder of joint structures, including but not limited to the following: 1. Achilles tendinopathy 2. Degenerative disorders of the joint, including but not limited to cartilage lesions 3. Dupuytren s contracture 4. Lateral epicondylitis (e.g., tennis elbow, elbow epicondylar tendinosis) 5. Osteoarthritis 6. Patellar tendinosis (jumper s knee) 2 MED77
3 7. Tendinopathy 8. Traumatic joint injury C. Plantar fasciitis D. As an adjunct to surgical procedures, including but not limited to: 1. Spinal fusion 2. Sinus surgery 3. Maxillofacial and periodontal surgery 4. Arthroplasty (e.g., rotator cuff repair, repair of structures of the knee) E. Injection of ligament tears with any type of blood-derived growth factor, whether from the patient or another source F. Ophthalmologic conditions or procedures SCIENTIFIC EVIDENCE In evaluating the efficacy for use of autologous blood-derived formulas, randomized controlled trials (RCTs) are needed which compare the effects of active and placebo formulas on the healing rates of patients with comparable conditions. Ideally, patients and investigators would be blinded as to group assignment. The focus of the literature appraisal below is on evidence from randomized controlled trials. Formula Preparation Several articles described different methods of preparation of autologous platelet-rich plasma, and noted variability in platelet concentration and viability depending on the preparation. [1-6] The clinical significance of these differences is unclear. Wound Healing Acute Wounds Use of autologous platelet-derived growth factor (PDGF) as a primary treatment of soft tissue injuries is in an early stage, and RCTs are lacking. Evidence is insufficient to permit conclusions concerning the effect of this technology on health outcomes. Kazakos and colleagues reported a prospective controlled study of the treatment of acute traumatic wounds with platelet gel in 59 consecutive patients (27 PRP and 32 controls). [7] Conventional treatment consisted of topical washing and cleaning of the wounds, removal of the necrotic tissue, and dressing with Vaseline gauze every 2 days. In all patients with open tibial fractures, an external fixation system was applied. PRP gel, prepared with specialized tubes and a bench-top centrifuge, was applied to the 3 MED77
4 wounds after surgical debridement and placement of the external fixation system. The time needed for preparation and application of the PRP gel was 52 minutes. PRP gel was then applied to the wounds once weekly in the outpatient clinic until there was adequate tissue regeneration (mean of 21 days) to undergo reconstructive plastic surgery. Control patients receiving conventional treatment required a mean of 41 days for adequate tissue regeneration. Pain scores were significantly lower in the PRPtreated patients at 2 and 3 weeks (VAS score of 58 PRP vs. 80 controls). Although these results are encouraging, additional study with a larger number of subjects is needed. Chronic Wounds Systematic Reviews A 2012 Cochrane review included 9 RCTs (325 participants) on PRP for treating chronic wounds. [8] This review was restricted to studies where PRP was compared with no additional treatment or a placebo. Four RCTs included patients with mixed chronic wounds, 3 included patients with venous leg ulcers, and 2 RCTs included patients with diabetic foot ulcers. Only 1 study was considered to be at low risk of bias. After a median treatment time of 12 weeks, there was no significant difference between the PRP and control groups in complete healing of diabetic foot ulcers, venous leg ulcers, or mixed chronic wounds. There was no significant difference in the area epithelialized in 3 RCTs of mixed chronic wounds. In 2 RCTs of mixed chronic wounds, there was a significant difference favoring PRP in the wound area that was healed. The review concluded that there is no current evidence to suggest that autologous PRP is of value for treating chronic wounds. A 2009 systematic review identified 42 controlled trials on PRP; 20 of these were RCTs and included in the systematic review. [9] The 20 RCTs comprised 11 studies on oral and maxillofacial surgery, seven on chronic skin ulcers, and two on surgery wounds. The authors concluded that PRP improved the gingival recession but not the clinical attachment level in chronic periodontitis. Results were inconclusive for the healing of skin ulcers, and there were little safety data. Non-randomized controlled studies were identified but not reviewed for chronic elbow tendinosis, muscle strains, lumbar spinal fusions, and other orthopedic procedures. An industry-funded systematic review included 21 studies on PRP gel for cutaneous wound healing, 12 of which were RCTs. [10] There were three main types of wounds, including open chronic wounds, acute surgical wounds with primary closure, and acute surgical wound with secondary closure. Study quality was found to vary considerably, with three studies rated as high quality and six rated as poor quality. Two additional studies could not be rated because they were published only as an abstract and letter. The primary outcome measure for this meta-analysis was complete wound healing. Overall, results from the RCTs were mixed, i.e. some trials reported a benefit but others did not. Of four RCTs that evaluated complete healing of chronic wounds, two reported a statistically significant benefit for PRP, and meta-analysis of the four RCTs showed a significant combined effect of PRP for complete healing of chronic wounds. However, two of the four studies were rated as low quality and the other two could not be rated because they were presented only in abstract or letter form. Meta-analysis could not be conducted for complete healing of acute primary or secondary closure wounds. The meta-analysis of the effect of PRP on complete wound healing of chronic wounds was limited by the inclusion of poor quality studies. There were no high-quality RCTs that showed an improvement in complete healing with PRP. A 2012 systematic review included 23 randomized trials and ten prospective cohort studies that compared PRP to placebo, corticosteroid, or a standard procedure. [11] Of 22 RCTs that evaluated functional outcomes, six showed a functional benefit of PRP, 15 showed no difference between PRP and the control, and one showed a significant functional advantage for the control group. For most of the studies the outcome measures differed, but six RCTs (n=358) and three prospective cohort 4 MED77
5 studies (n=88) reported results of PRP using a visual analog score (VAS) and were combined for analysis. These studies assessed injuries to the acromion, rotator cuff, lateral humeral epicondyle, anterior cruciate ligament (ACL), patella, tibia, and spine. Follow-up ranged from 6 weeks to 24 months. No significant benefit of PRP was found for the six RCTs or the three prospective cohort studies. Interpretation of this systematic review is limited by the combination of a wide variety of conditions, as well as the lack of standardization of platelet-separation techniques and outcome measures in the primary literature. Randomized Controlled Trials No new RCTs have been published since the systematic reviews summarized above. Nonsurgical Treatment for Musculoskeletal Disorders Miscellaneous Tendinopathy Systematic Reviews Three recently published systematic reviews for various tendinopathies found few randomized trials, and no studies of high-quality design. [4,12,13] While uncontrolled trials showed promising results, those studies with a control group reported no significant benefit from use of PRP compared with patients who did not receive PRP. These systematic reviews concluded that well-designed, large, long-term, randomized trials with appropriate control groups are needed to determine the impact of PRP for chronic tendinopathies. Lateral Epicondylitis Randomized Controlled Trials In 2013, Krogh et al. reported a double-blind placebo-controlled RCT in 60 patients with chronic lateral epicondylitis. [14] Patients were randomized to receive either a blinded injection of PRP, saline, or corticosteroid injection. In order to maintain blinding, a blood sample was taken from all patients, and patients were blindfolded during the blood sampling and injections. At 1 month, corticosteroid injections reduced pain to a greater extent than either PRP or saline. At 3-month follow-up, there was no significant difference between the groups in the primary endpoint of pain reduction. Corticosteroid injection was more effective than saline or PRP in reducing color Doppler activity and tendon thickness. A double-blind randomized trial of PRP for lateral epicondylitis was reported by Peerbooms and colleagues in [15] Two-year follow-up of this RCT was reported in 2011 by Gosens et al. [16] One hundred patients with chronic (longer than 6 months) epicondylitis were randomized to receive corticosteroid injection (n=49) and to receive PRP injection (n=51). Stretching and exercise protocols were followed by each group, and normal sport or recreational activities were allowed as tolerated 4 weeks after injection. Eight patients were lost to follow-up, and their last scores were carried forward. Success was defined as 25% reduction in pain on VAS or Disabilities of the Arm, Shoulder, and hand (DASH) outcomes measure score after 1 year without a re-intervention. Initially, mean VAS was 70.1 in the PRP-treated patients and 65.8 in the corticosteroid group. DASH scores were and 131.2, respectively. 5 MED77
6 At 4 and 8 weeks after injection, outcomes on VAS and DASH scores were significantly better in the corticosteroid group. At 12 weeks, between-group differences were not significant. After 1 year, 73% of PRP and 49% of steroid-treated patients met criteria for success on pain VAS; 73% of the PRP group and 51% the steroid group were successful using DASH outcome measures (P=0.005). At two years, both VAS and DASH scores were significantly better in the PRP group (21.3 and 17.6, respectively) compared with the corticosteroid group (42.4 and 36.5, respectively). Success on the DASH was achieved by 73% of the PRP group and 39% of the corticosteroid group, while more patients in the corticosteroid group (47% vs. 14%) had deteriorated at two years. Additional studies are needed with PRP in this condition. Achilles Tendinopathy Randomized Controlled Trials A single center, randomized, double-blind, placebo-controlled trial of PRP injection in patients with chronic midportion Achilles tendinopathy was reported by de Vos et al. in [17] Fifty-four patients were randomized to receive PRP or saline injection, and all patients performed eccentric exercises. The Victorian Institute of Sports Assessment-Achilles (VISA-A) questionnaire evaluating pain score and activity level was completed at baseline and at 6, 12, and 24 weeks. The mean VISA-A score improved significantly after 24 weeks in both groups, and the between-group difference was not statistically significant. There were no significant differences on secondary measures of patient satisfaction and number of patients returning to their desired sport. This lack of significant difference in clinical and ultrasonographic outcomes continued through one-year follow-up, reported in a separate article. [18] Plantar Fasciitis The 2012 systematic review by Sheth et al. identified three nonrandomized uncontrolled studies that evaluated the effect of autologous blood injections. [11] No controlled trials have been identified that evaluated the effect of PRP for plantar fasciitis. Osteochondral Lesions In 2012, Mei-Dan et al. reported a quasi-randomized trial of 29 patients with 30 osteochondral lesions of the talus assigned to three intra-articular injections of hyaluronate or PRP. (21) At 28-week follow-up, scores on the American Orthopaedic Foot and Ankle Society (AOFAS) Ankle-Hindfoot Scale (AHFS) improved to a greater extent in the PRP group (from 68 to 92) than the hyaluronate group (from 66 to 78). Subjective global function also improved to a greater extent in the PRP group (from 58 to 91) than the hyaluronate group (from 56 to 73). Interpretation of the composite measures of VAS pain and VAS function is limited by differences in the groups at baseline. Neither the patients nor the evaluators were blinded to treatment in this small study. Osteoarthritis Randomized Controlled Trials A double-blind study from 2012 randomized 109 patients with knee chondropathy or osteoarthritis (Kellgen-Lawrence grades I, II, or III) to 3 weekly injections of PRP (previously frozen aliquot) or HA. [19] (26) At 12 months of follow-up, there was significant improvement on 6 MED77
7 IKDC [International Knee Documentation Committee], VAS, TEGNER, and KOOS scores in both groups, but no significant difference between the groups. In an additional 2012 RCT, 4 intra-articular injections of PRP or hyaluronic acid (HA) were compared in 120 patients with gonarthrosis (early stage knee arthritis, Kellgen-Lawrence grades I, II, or III). [20] At 4 weeks, both groups showed improvement in Western Ontario and McMaster (WOMAC) scores compared to baseline. In this non-blinded study, WOMAC scores for the PRP group continued to improve over 24 weeks (79.6 at baseline, 49.6 at 4 weeks and 36.5 at 24 weeks), whereas WOMAC scores in the HA group declined in the post-treatment period (75.4 at baseline, 49.6 at 4 weeks, and 65.1 at 24 weeks). In 2013, Patel et al. reported a double-blind placebo-controlled RCT with 78 patients (156 knees). [21] This study compared a single injection of PRP, 2 injections of PRP, or a single injection of saline. Adverse effects at the time of injection were observed in 22% of patients in the 1 injection PRP group and 44% in the 2 injection PRP group, compared to none in the saline group. At 6 month follow-up WOMAC scores had improved in the 2 PRP groups but not in the control group. VAS pain scores improved from 4.54 to 2.16 after a single injection of PRP and from 4.64 to 2.54 after 2 injections of PRP. VAS scores did not change in the placebo-control group (from 4.57 at baseline to 4.61 at 6-month follow-up). A 2009 report from Europe described a prospective study of intra-articular injection of PRP in 100 consecutive patients affected by chronic degenerative cartilage lesions. [22] Patients had a history of pain or swelling of the knee for at least 4 months and imaging findings on radiograph or magnetic resonance imaging (MR) of degenerative changes in the joint; 58 knees presented with a degenerative chondral lesion, 33 with early osteoarthritis, and 24 had advanced osteoarthritis. Exclusion criteria included systemic disorders, axial malalignment, severe cardiovascular diseases, infections, or immunodepression. Three injections were administered at 21-day intervals. Evaluation was conducted in 91 patients (91% follow-up) before and at the end of the 3 treatments and at 6 and 12 months after treatment. The International Knee Documentation Committee (IKDC) objective score improved from 46% (of normal and nearly normal knees) to 78% at the end of therapy, declining to 67% at 12-month follow-up. The IKDC subjective score improved from 41 to 63 after treatment, with a score of 61 at 12-month followup. Treatment was less effective in older, heavier, and more advanced osteoarthritis patients than in younger patients with less severe chondral damage. Adjunct to Surgical Procedures Spinal Fusion Randomized Controlled Trials Only one randomized controlled trial was found for use of autologous growth factor concentrate (AGF), including PRP, as an adjunct to lumbar fusion. [23] In this small trial, outcomes for 40 patients who underwent spinal fusion with (n=20) versus w/o (n=20) AGF. One patient per group was lost to followup. No significant between-group differences were found with CT scan at 1 year, which showed osseous healing in all but one patient. The pain and function outcomes at 2 years follow-up also showed no significant between-group differences. The authors concluded that use of PRP as an adjunct to spinal fusion was not justified. Shoulder Surgery Randomized Controlled Trials 7 MED77
8 Rotator Cuff Repair In 2012, Rodeo et al. reported a RCT arthroscopic rotator cuff tendon repair with or without PRP (platelet rich fibrin matrix) in 79 patients. [24] Follow-up at 6 weeks, 3 months, and 12 months postoperatively found no significant differences between the PRP and control groups for tendon healing, tendon vascularity, manual muscle strength, or clinical rating scales. Logistic regression analysis suggested that PRP might have a negative effect on healing. Castricini et al randomized 88 patients with a rotator cuff tear to arthroscopic repair without (n=45) or with (n=43) augmentation with platelet-rich fibrin matrix. [25] At average follow-up of 20.2 months (range, months), both groups demonstrated statistically significant improvement in the primary endpoint (Constant Scores evaluating pain, activities of daily living, range of movement, and power), but the between-group difference was not significant. A double-blind quasi-randomized trial from 2013 assigned 60 patients in alternating order to receive rotator cuff surgery with or without PRP (platelet rich fibrin matrix). [26] Mean surgery time was increased by about 10 minutes in the PRP group. At 1-year follow-up, there was no significant difference between the groups in VAS pain scores, narcotic use, recovery of motion, simple shoulder test (SST), and American Shoulder and Elbow Surgeons (ASES) shoulder scores. Mean University of California-Los Angeles (UCLA) scores were slightly lower in the PRP group (27.94 vs ). There were no significant differences between the groups on MRI scans at 3 to 5 month follow-up. In 2012, Gumina et al. reported a RCT of platelet-leukocyte membrane in 80 patients with full thickness rotator cuff tear randomized to arthroscopic repair with or without PRP. [27] Both age and Constant score were significantly different at baseline. At a mean 13-month follow-up, the SST and the change in Constant score did not differ significantly between the 2 groups. Independent evaluation with MRI found that rotator cuff retears occurred only in the control group, and that the use of the PRP membrane resulted in significantly better repair integrity. Randelli et al. randomized 53 patients in a double-blind study to activated PRP or to no treatment after arthroscopic rotator cuff repair. [28] VAS pain scores in the PRP group were lower than controls at baseline (4.8 vs. 6.4) through 30 days after surgery (1.1 vs. 2.4). At 3 months after surgery, the PRP group had higher scores on Constant scores (65.0 vs. 57.8) and the Simple Shoulder Test (8.9 vs. 7.1), University of California (UCLA, 26.9 vs. 24.2) and strength in external rotation (3.0 vs. 2.1). There was no difference in functional outcomes between the groups at 6, 12, and 24 months after surgery and no difference in the healing rate measured by magnetic resonance imaging (MRI) at one year or more after surgery. This study was limited by the difference in VAS between the groups at baseline. A small double-blind RCT of PRP for rotator cuff healing without surgical repair was reported by Rha et al. in [29] In this study, 39 patients with tendinosis or a partial tear were randomized to 2 sessions of dry needling or 2 PRP injections. For dry needling, a needle was passed through the lesion of the tendon approximately times. PRP was injected into or around the lesion under ultrasound guidance. Both groups showed an improvement in the Shoulder Pain and Disability Index (SPDI) over the 6 months of the study. At 2 weeks, 3 months and 6 months after the treatment, the mean SPDI score was significantly better in the PRP group (17.7 vs. 29.5). Range of motion was generally not different between the groups. 8 MED77
9 In a small 2013 RCT study, Kesikburun et al. investigated the effect of PRP injections on pain and shoulder function in 40 patients with chronic rotator cuff tendinopathy following a 6-week standard exercise program. Patients were randomized into a PRP group (n=20) or placebo group (n=20). At one-year follow-up, the authors reported the PRP injections were no more effective in improving quality of life, pain, disability, and shoulder range of motion than patients that received placebo. [30] Subacromial Decompression Surgery Everts and colleagues reported a rigorously conducted, small (n=40) double-blinded RCT of platelet and leukocyte-rich plasma (PLRP) gel following open subacromial decompression surgery in a carefully selected patient population. [31] Blood was drawn from all patients after induction of anesthesia to maintain blinding. PLRP with autologous thrombin was injected into both the subacromial intracapsular space and the subcutaneous layer covering the incision during wound closure. Postoperative examinations at 1, 2, 4, and 6 weeks were performed by independent evaluators; unique patient identifier codes were used to maintain patient and investigator blinding. Neither self-assessed nor physician-assessed instability were improved. Both subjective pain and use of pain medication were significantly lower in the PRP group across the 6 weeks of measurements. For example, at 2 weeks after surgery VAS scores for pain were lower by about 50% in the PLRP group (close to 4 in the control group and close to 2 in the PLRP group) and only 1 patient (5%) was taking pain medication compared with 10 (50%) control patients. Objective measures of range of motion showed clinically significant improvement in the PLRP group across the 6-week assessment period. Significantly more patients in the PLRP group reported improvements in activities of daily living such as ability to sleep on the operated shoulder at 4 weeks after surgery and earlier return to work. Knee Surgeries Randomized controlled trials In 2012, de Almeida et al. reported a small (n=27) randomized trial of the effect of PRP gel on the harvest site of the patellar tendon during anterior cruciate ligament (ACL) reconstruction. [32] VAS for pain in the postoperative period was significantly lower in the PRP group compared to the control group (3.8 vs. 5.1). At 6 months, assessment by MRI showed a smaller gap in the patellar tendon in the PRP group (4.9 mm vs. 9.4 mm), but there was no significant difference between groups for the Tegner questionnaire or isokinetic testing. Nin and colleagues randomized 100 patients undergoing arthroscopic patellar tendon allograft anterior cruciate ligament reconstruction; platelet-enriched gel was used in one group (n=50), and a non-gel group (n=50) served as control. [33] The use of platelet-derived growth factors (PDGF), on the graft and inside the tibial tunnel in patients treated with bone-patellar tendonbone allografts has no discernible clinical or biomechanical effect at 2-year follow-up. Cervellin et al randomized 40 young athletes undergoing anterior cruciate ligament (ACL) reconstruction to either a control group (n=20) or a PRP group (n=20). [34] The PRP group received PRP gel to both the patellar and tendon bone plug harvest site. At 1-year follow-up, the PRP group reported significantly less pain during activities involving the knee. There was no significant difference between the two groups for Visual Analog Scale or for bone defect filling per MRI. Long Bone Nonunion 9 MED77
10 Systematic Review A 2012 Cochrane review found only one small (n=21) RCT of PRP for long bone healing. [35] However, only studies where PRP was compared with no additional treatment or a placebo were included in the review; therefore the authors did not include the larger RCT by Calori et al. described below. Randomized Controlled Trial Calori et al. compared application of PRP or recombinant human bone morphogenetic protein-7 (rhbmp-7) for the treatment of long bone nonunions in an RCT with 120 patients and ten surgeons. [36] Inclusion criteria were post-traumatic atrophic nonunion for at least nine months, with no signs of healing over the last three months, and considered as treatable only by means of fixation revision. Autologous bone graft had been used in a prior surgery in 23 cases in the rhbmp-7 group and in 21 cases in the PRP group. Computer-generated randomization was developed to create two homogeneous groups; there were generally similar numbers of tibial, femoral, humeral, ulnar, and radial nonunions in the two groups. Following randomization, the patients underwent surgery for nonunion, including bone grafts according to the surgeon s choice (66.6% of rhbmp-7 and 80% of PRP patients). Clinical and radiologic evaluations by one radiologist and two surgeons trained in the study protocol revealed fewer unions in the PRP group (68%) compared with the rhbmp-7 group (87%). Clinical and radiographic healing times were also found to be slower by 13 14% with PRP. Other Surgical Procedures No difference was found between the treatment and control groups in studies of PRP use in the following surgical procedures: Sinus surgery [37] Periodontal surgery [38] Incision site wound closure in vascular surgeries [39-41] Blepharoplasty [42] Tonsillectomy in children [43] Ophthalmologic Conditions and Procedures Use of PRP has been studied as a treatment of persistent corneal defects [44], symptomatic dry eye [45], chemical burns [46], post-lasik ocular surface syndrome [47]. Studies are limited to small pilot studies with no control group for comparison. No randomized trials were identified. Clinical Practice Guidelines A 2009 practice guideline from the National Pressure Ulcer Advisory Panel and the European Pressure Ulcer Advisory Panel concluded, The combined clinical evidence on platelet-derived growth factor (PDGF) suggests that PDGF-BB may improve healing of pressure ulcers. However, the evidence is not sufficient to recommend this treatment for routine use. [48] No other clinical practice guidelines from U.S. professional associations were found that address the use of platelet-derived growth factor for any condition. Summary 10 MED77
11 The current evidence is insufficient to permit conclusions about the benefits of autologous plateletderived growth factors (PDGF) in the treatment of any condition. In addition, there are no clinical practice guidelines from U.S. professional associations that recommend the use of PDGF. Therefore, the use of PDGF for any condition is considered investigational. [49] REFERENCES 1. Eppley, BL, Woodell, JE, Higgins, J. Platelet quantification and growth factor analysis from platelet-rich plasma: implications for wound healing. Plast Reconstr Surg Nov;114(6): PMID: Crovetti, G, Martinelli, G, Issi, M, et al. Platelet gel for healing cutaneous chronic wounds. Transfus Apher Sci Apr;30(2): PMID: Kevy, SV, Jacobson, MS. Comparison of methods for point of care preparation of autologous platelet gel. J Extra Corpor Technol Mar;36(1): PMID: de Vos, RJ, van Veldhoven, PL, Moen, MH, Weir, A, Tol, JL, Maffulli, N. Autologous growth factor injections in chronic tendinopathy: a systematic review. Br Med Bull. 2010;95: PMID: Castillo, TN, Pouliot, MA, Kim, HJ, Dragoo, JL. Comparison of growth factor and platelet concentration from commercial platelet-rich plasma separation systems. Am J Sports Med Feb;39(2): PMID: Mazzucco, L, Balbo, V, Cattana, E, Guaschino, R, Borzini, P. Not every PRP-gel is born equal. Evaluation of growth factor availability for tissues through four PRP-gel preparations: Fibrinet, RegenPRP-Kit, Plateltex and one manual procedure. Vox Sang Aug;97(2): PMID: Kazakos, K, Lyras, DN, Verettas, D, Tilkeridis, K, Tryfonidis, M. The use of autologous PRP gel as an aid in the management of acute trauma wounds. Injury Aug;40(8): PMID: Martinez-Zapata, MJ, Marti-Carvajal, AJ, Sola, I, et al. Autologous platelet-rich plasma for treating chronic wounds. Cochrane Database Syst Rev. 2012;10:CD PMID: Martinez-Zapata, MJ, Marti-Carvajal, A, Sola, I, et al. Efficacy and safety of the use of autologous plasma rich in platelets for tissue regeneration: a systematic review. Transfusion Jan;49(1): PMID: Carter, MJ, Fylling, CP, Parnell, LK. Use of platelet rich plasma gel on wound healing: a systematic review and meta-analysis. Eplasty. 2011;11:e38. PMID: Sheth, U, Simunovic, N, Klein, G, et al. Efficacy of autologous platelet-rich plasma use for orthopaedic indications: a meta-analysis. J Bone Joint Surg Am Feb 15;94(4): PMID: Taylor, DW, Petrera, M, Hendry, M, Theodoropoulos, JS. A systematic review of the use of platelet-rich plasma in sports medicine as a new treatment for tendon and ligament injuries. Clin J Sport Med Jul;21(4): PMID: Hoksrud, AF, Bahr, R. Injectable agents derived from or targeting vascularity: has clinical acceptance in managing tendon disorders superseded scientific evidence? J Musculoskelet Neuronal Interact Jun;11(2): PMID: Krogh, TP, Fredberg, U, Stengaard-Pedersen, K, Christensen, R, Jensen, P, Ellingsen, T. Treatment of lateral epicondylitis with platelet-rich plasma, glucocorticoid, or saline: a randomized, double-blind, placebo-controlled trial. Am J Sports Med Mar;41(3): PMID: MED77
12 15. Peerbooms, JC, Sluimer, J, Bruijn, DJ, Gosens, T. Positive effect of an autologous platelet concentrate in lateral epicondylitis in a double-blind randomized controlled trial: platelet-rich plasma versus corticosteroid injection with a 1-year follow-up. Am J Sports Med Feb;38(2): PMID: Gosens, T, Peerbooms, JC, van Laar, W, den Oudsten, BL. Ongoing positive effect of plateletrich plasma versus corticosteroid injection in lateral epicondylitis: a double-blind randomized controlled trial with 2-year follow-up. Am J Sports Med Jun;39(6): PMID: de Vos, RJ, Weir, A, van Schie, HT, et al. Platelet-rich plasma injection for chronic Achilles tendinopathy: a randomized controlled trial. JAMA Jan 13;303(2): PMID: de Jonge, S, de Vos, RJ, Weir, A, et al. One-Year Follow-up of Platelet-Rich Plasma Treatment in Chronic Achilles Tendinopathy: A Double-Blind Randomized Placebo-Controlled Trial. Am J Sports Med Aug;39(8): PMID: Filardo, G, Kon, E, Di Martino, A, et al. Platelet-rich plasma vs hyaluronic acid to treat knee degenerative pathology: study design and preliminary results of a randomized controlled trial. BMC Musculoskelet Disord. 2012;13:229. PMID: Cerza, F, Carni, S, Carcangiu, A, et al. Comparison between hyaluronic acid and platelet-rich plasma, intra-articular infiltration in the treatment of gonarthrosis. Am J Sports Med Dec;40(12): PMID: Patel, S, Dhillon, MS, Aggarwal, S, Marwaha, N, Jain, A. Treatment with platelet-rich plasma is more effective than placebo for knee osteoarthritis: a prospective, double-blind, randomized trial. Am J Sports Med Feb;41(2): PMID: Kon, E, Buda, R, Filardo, G, et al. Platelet-rich plasma: intra-articular knee injections produced favorable results on degenerative cartilage lesions. Knee Surg Sports Traumatol Arthrosc Apr;18(4): PMID: Sys, J, Weyler, J, Van Der Zijden, T, Parizel, P, Michielsen, J. Platelet-rich plasma in monosegmental posterior lumbar interbody fusion. Eur Spine J Jul 10. PMID: Rodeo, SA, Delos, D, Williams, RJ, Adler, RS, Pearle, A, Warren, RF. The effect of platelet-rich fibrin matrix on rotator cuff tendon healing: a prospective, randomized clinical study. Am J Sports Med Jun;40(6): PMID: Castricini, R, Longo, UG, De Benedetto, M, et al. Platelet-rich plasma augmentation for arthroscopic rotator cuff repair: a randomized controlled trial. Am J Sports Med Feb;39(2): PMID: Weber, SC, Kauffman, JI, Parise, C, Weber, SJ, Katz, SD. Platelet-rich fibrin matrix in the management of arthroscopic repair of the rotator cuff: a prospective, randomized, double-blinded study. Am J Sports Med Feb;41(2): PMID: Gumina, S, Campagna, V, Ferrazza, G, et al. Use of platelet-leukocyte membrane in arthroscopic repair of large rotator cuff tears: a prospective randomized study. J Bone Joint Surg Am Aug 1;94(15): PMID: Randelli, P, Arrigoni, P, Ragone, V, Aliprandi, A, Cabitza, P. Platelet rich plasma in arthroscopic rotator cuff repair: a prospective RCT study, 2-year follow-up. J Shoulder Elbow Surg Jun;20(4): PMID: Rha, DW, Park, GY, Kim, YK, Kim, MT, Lee, SC. Comparison of the therapeutic effects of ultrasound-guided platelet-rich plasma injection and dry needling in rotator cuff disease: a randomized controlled trial. Clin Rehabil Feb;27(2): PMID: Kesikburun, S, Tan, AK, Yilmaz, B, Yasar, E, Yazicioglu, K. Platelet-Rich Plasma Injections in the Treatment of Chronic Rotator Cuff Tendinopathy: A Randomized Controlled Trial With 1- Year Follow-up. Am J Sports Med Jul 26. PMID: MED77
13 31. Everts, PA, Devilee, RJ, Brown Mahoney, C, et al. Exogenous application of platelet-leukocyte gel during open subacromial decompression contributes to improved patient outcome. A prospective randomized double-blind study. Eur Surg Res. 2008;40(2): PMID: de Almeida, AM, Demange, MK, Sobrado, MF, Rodrigues, MB, Pedrinelli, A, Hernandez, AJ. Patellar tendon healing with platelet-rich plasma: a prospective randomized controlled trial. Am J Sports Med Jun;40(6): PMID: Nin, JR, Gasque, GM, Azcarate, AV, Beola, JD, Gonzalez, MH. Has platelet-rich plasma any role in anterior cruciate ligament allograft healing? Arthroscopy Nov;25(11): PMID: Cervellin, M, de Girolamo, L, Bait, C, Denti, M, Volpi, P. Autologous platelet-rich plasma gel to reduce donor-site morbidity after patellar tendon graft harvesting for anterior cruciate ligament reconstruction: a randomized, controlled clinical study. Knee Surg Sports Traumatol Arthrosc Jun 16. PMID: Griffin, XL, Wallace, D, Parsons, N, Costa, ML. Platelet rich therapies for long bone healing in adults. Cochrane Database Syst Rev. 2012;7:CD PMID: Calori, GM, Tagliabue, L, Gala, L, d'imporzano, M, Peretti, G, Albisetti, W. Application of rhbmp-7 and platelet-rich plasma in the treatment of long bone non-unions: a prospective randomised clinical study on 120 patients. Injury Dec;39(12): PMID: Rice, DH. Platelet-rich plasma in endoscopic sinus surgery. Ear Nose Throat J Aug;85(8):516, 8. PMID: Yassibag-Berkman, Z, Tuncer, O, Subasioglu, T, Kantarci, A. Combined use of platelet-rich plasma and bone grafting with or without guided tissue regeneration in the treatment of anterior interproximal defects. J Periodontol May;78(5): PMID: Buchwald, D, Kaltschmidt, C, Haardt, H, Laczkovics, A, Reber, D. Autologous platelet gel fails to show beneficial effects on wound healing after saphenectomy in CABG patients. J Extra Corpor Technol Sep;40(3): PMID: Almdahl, SM, Veel, T, Halvorsen, P, Vold, MB, Molstad, P. Randomized prospective trial of saphenous vein harvest site infection after wound closure with and without topical application of autologous platelet-rich plasma. Eur J Cardiothorac Surg Jan;39(1):44-8. PMID: Lawlor, DK, Derose, G, Harris, KA, Lovell, MB, Novick, TV, Forbes, TL. The role of plateletrich plasma in inguinal wound healing in vascular surgery patients. Vasc Endovascular Surg Apr;45(3): PMID: Vick, VL, Holds, JB, Hartstein, ME, Rich, RM, Davidson, BR. Use of autologous platelet concentrate in blepharoplasty surgery. Ophthal Plast Reconstr Surg Mar-Apr;22(2): PMID: Sidman, JD, Lander, TA, Finkelstein, M. Platelet-rich plasma for pediatric tonsillectomy patients. Laryngoscope Oct;118(10): PMID: Lopez-Plandolit, S, Morales, MC, Freire, V, Etxebarria, J, Duran, JA. Plasma rich in growth factors as a therapeutic agent for persistent corneal epithelial defects. Cornea Aug;29(8): PMID: Alio, JL, Colecha, JR, Pastor, S, Rodriguez, A, Artola, A. Symptomatic dry eye treatment with autologous platelet-rich plasma. Ophthalmic Res. 2007;39(3): PMID: Marquez De Aracena Del Cid, R, Montero De Espinosa Escoriaza, I. Subconjunctival application of regenerative factor-rich plasma for the treatment of ocular alkali burns. Eur J Ophthalmol Nov-Dec;19(6): PMID: MED77
14 47. Alio, JL, Pastor, S, Ruiz-Colecha, J, Rodriguez, A, Artola, A. Treatment of ocular surface syndrome after LASIK with autologous platelet-rich plasma. J Refract Surg Jun;23(6): PMID: National Pressure Ulcer Advisory Panel, European Pressure Ulcer Advisory Panel. Pressure ulcer treatment recommendations. In: Prevention and treatment of pressure ulcers: clinical practice guideline. Washington (DC): National Pressure Ulcer Advisory Panel; p [cited 10/09/2012]; Available from: BlueCross BlueShield Association Medical Policy Reference Manual "Recombinant and Autologous Platelet-Derived Growth Factors as a Treatment of Wound Healing and Other Conditions." Policy No CROSS REFERENCES Stem-cell Therapy for Peripheral Arterial Disease, Medicine, Policy No. 141 Orthopedic Applications of Stem-Cell Therapy, Medicine, Policy No. 142 CODES NUMBER DESCRIPTION CPT 0232T Injection(s) platelet rich plasma, any tissue including image guidance, harvesting and preparation when performed. HCPCS G0460 Autologous platelet rich plasma for chronic wounds/ulcers, including phlebotomy, centrifugation, and all other preparatory procedures and administration, per treatment P9020 S9055 Platelet rich plasma, each unit Procuren or other growth factor preparation to promote wound healing 14 MED77
10/8/14. Revision Date(s): Policy Number: MCP-207. Review Date: 12/16/15, 9/15/16
 Subject: Platelet-rich Plasma (PRP) Policy Number: MCP-207 Review Date: 12/16/15, 9/15/16 Revision Date(s): Original Effective Date: 10/8/14 DISCLAIMER This Molina Clinical Policy (MCP) is intended to
Subject: Platelet-rich Plasma (PRP) Policy Number: MCP-207 Review Date: 12/16/15, 9/15/16 Revision Date(s): Original Effective Date: 10/8/14 DISCLAIMER This Molina Clinical Policy (MCP) is intended to
Orthopedic Applications of Platelet- Rich Plasma
 Orthopedic Applications of Platelet- Rich Plasma Policy Number: 2.01.98 Last Review: 5/2018 Origination: 5/2016 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Orthopedic Applications of Platelet- Rich Plasma Policy Number: 2.01.98 Last Review: 5/2018 Origination: 5/2016 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Corporate Medical Policy Growth Factors in Wound Healing
 Corporate Medical Policy Growth Factors in Wound Healing File Name: Origination: Last CAP Review: Next CAP Review: Last Review: growth_factors_in_wound_healing 4/1993 11/2017 11/2018 11/2017 Description
Corporate Medical Policy Growth Factors in Wound Healing File Name: Origination: Last CAP Review: Next CAP Review: Last Review: growth_factors_in_wound_healing 4/1993 11/2017 11/2018 11/2017 Description
Orthopedic Applications of Platelet-Rich Plasma
 Section: Medicine Effective Date: October 15, Subject: Orthopedic Applications of Platelet-Rich Plasma Page: 1 of 11 Last Review Status/Date: September Orthopedic Applications of Platelet-Rich Plasma Summary
Section: Medicine Effective Date: October 15, Subject: Orthopedic Applications of Platelet-Rich Plasma Page: 1 of 11 Last Review Status/Date: September Orthopedic Applications of Platelet-Rich Plasma Summary
Page: 1 of 16. Recombinant and Autologous Platelet-Derived Growth Factors as a Treatment of Wound Healing and Other Conditions
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 2.01.16 Last Review Status/Date: June 2013 Page: 1 of 16 Derived Growth Factors as a Treatment of Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 2.01.16 Last Review Status/Date: June 2013 Page: 1 of 16 Derived Growth Factors as a Treatment of Description
Recombinant and Autologous Platelet-Derived Growth Factors as a Treatment of Wound Healing and Other Conditions. Original Policy Date 12:2013
 MP 2.01.14 Recombinant and Autologous Platelet-Derived Growth Factors as a Treatment of Wound Healing and Other Conditions Medical Policy Section Medicine Issue 12:2013 Subsection Original Policy Date
MP 2.01.14 Recombinant and Autologous Platelet-Derived Growth Factors as a Treatment of Wound Healing and Other Conditions Medical Policy Section Medicine Issue 12:2013 Subsection Original Policy Date
DESCRIPTION OF PROCEDURE/SERVICE/PHARMACEUTICAL
 Subject: Platelet-rich Plasma (PRP) Policy Number: MCP-207 Review Date: 12/16/15, 9/15/16, 9/19/17 Revision Date(s): Original Effective Date: 10/8/14 DISCLAIMER This Molina Clinical Policy (MCP) is intended
Subject: Platelet-rich Plasma (PRP) Policy Number: MCP-207 Review Date: 12/16/15, 9/15/16, 9/19/17 Revision Date(s): Original Effective Date: 10/8/14 DISCLAIMER This Molina Clinical Policy (MCP) is intended
Neuropathic diabetic ulcers extending into the subcutaneous tissue Pressure ulcers extending into the subcutaneous tissue
 Original Issue Date (Created): July 1, 2002 Most Recent Review Date (Revised): September 24, 2013 Effective Date: November 1, 2013 I. POLICY Recombinant platelet-derived growth factor (i.e., becaplermin)
Original Issue Date (Created): July 1, 2002 Most Recent Review Date (Revised): September 24, 2013 Effective Date: November 1, 2013 I. POLICY Recombinant platelet-derived growth factor (i.e., becaplermin)
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Orthopedic Applications of Platelet-Rich Plasma Page 1 of 23 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Orthopedic Applications of Platelet-Rich Plasma Professional
Orthopedic Applications of Platelet-Rich Plasma Page 1 of 23 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Orthopedic Applications of Platelet-Rich Plasma Professional
Clinical Review Criteria
 Clinical Review Criteria Autologous Platelet Derived Wound Healing Factors for Treatment of: Non Healing Cutaneous Wounds (Procuren) Non-Healing Fractures and the Associated GEM 21STM Device Platelet Rich
Clinical Review Criteria Autologous Platelet Derived Wound Healing Factors for Treatment of: Non Healing Cutaneous Wounds (Procuren) Non-Healing Fractures and the Associated GEM 21STM Device Platelet Rich
Orthopedic Applications of Platelet-Rich Plasma
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Clinical Policy Title: Platelet rich plasma
 Clinical Policy Title: Platelet rich plasma Clinical Policy Number: 05.02.10 Effective Date: February 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date: November 16, 2017 Next Review
Clinical Policy Title: Platelet rich plasma Clinical Policy Number: 05.02.10 Effective Date: February 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date: November 16, 2017 Next Review
ORTHOPEDICS BONE Recalcitrant nonunions In total hip replacement total knee surgery increased callus volume
 ORTHOPEDICS Orthopedics has to do with a variety of tissue: bone, cartilage, tendon, ligament, muscle. In this regard orthopedic and sports medicine share the same tissue targets. Orthopedics is mostly
ORTHOPEDICS Orthopedics has to do with a variety of tissue: bone, cartilage, tendon, ligament, muscle. In this regard orthopedic and sports medicine share the same tissue targets. Orthopedics is mostly
Clinical Policy Title: Platelet rich plasma
 Clinical Policy Title: Platelet rich plasma Clinical Policy Number: 05.02.10 Effective Date: February 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date: November 16, 2016 Next Review
Clinical Policy Title: Platelet rich plasma Clinical Policy Number: 05.02.10 Effective Date: February 1, 2017 Initial Review Date: November 16, 2016 Most Recent Review Date: November 16, 2016 Next Review
Preliminary Report Choosing Wisely Identifying Musculoskeletal Interventions with Limited Levels of Efficacy in the Shoulder & Elbow.
 Preliminary Report Choosing Wisely Identifying Musculoskeletal Interventions with Limited Levels of Efficacy in the Shoulder & Elbow. Prepared for The Canadian Orthopaedic Association Contents Executive
Preliminary Report Choosing Wisely Identifying Musculoskeletal Interventions with Limited Levels of Efficacy in the Shoulder & Elbow. Prepared for The Canadian Orthopaedic Association Contents Executive
Recombinant and Autologous Platelet-Derived Growth Factors for Wound Healing and Other Non Orthopedic Conditions
 Recombinant and Autologous Platelet-Derived Growth Factors for Wound Healing and Other Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary,
Recombinant and Autologous Platelet-Derived Growth Factors for Wound Healing and Other Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary,
PLATELET DERIVED GROWTH FACTORS FOR TREATMENT OF WOUNDS
 MEDICAL POLICY PLATELET DERIVED GROWTH FACTORS FOR TREATMENT OF WOUNDS Policy Number: 2011T0523E Effective Date: May 1, 2011 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S.
MEDICAL POLICY PLATELET DERIVED GROWTH FACTORS FOR TREATMENT OF WOUNDS Policy Number: 2011T0523E Effective Date: May 1, 2011 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S.
STUDY OF PLATELET RICH PLASMA INJECTIONS IN PATIENTS OF TENDINOPATHY IN SOUTH GUJARAT POPULATION
 ORIGINAL ARTICLE STUDY OF PLATELET RICH PLASMA INJECTIONS IN PATIENTS OF TENDINOPATHY IN SOUTH GUJARAT POPULATION Manish Patel 1, Chintan Sheth 2, Jignesh Patel 1, PrabhavTijoriwala 1 Author s Affiliations:
ORIGINAL ARTICLE STUDY OF PLATELET RICH PLASMA INJECTIONS IN PATIENTS OF TENDINOPATHY IN SOUTH GUJARAT POPULATION Manish Patel 1, Chintan Sheth 2, Jignesh Patel 1, PrabhavTijoriwala 1 Author s Affiliations:
Intraarticular platelet-rich plasma injection in the treatment of knee osteoarthritis: review and recommendations.
 Am J Phys Med Rehabil. 2014 Nov;93(11 Suppl 3):S108-21. doi: 10.1097/PHM.0000000000000115. Intraarticular platelet-rich plasma injection in the treatment of knee osteoarthritis: review and recommendations.
Am J Phys Med Rehabil. 2014 Nov;93(11 Suppl 3):S108-21. doi: 10.1097/PHM.0000000000000115. Intraarticular platelet-rich plasma injection in the treatment of knee osteoarthritis: review and recommendations.
An Owner's Guide to Natural Healing. Autologous Conditioned Plasma (ACP)
 An Owner's Guide to Natural Healing Autologous Conditioned Plasma (ACP) Healing after an injury involves a well-orchestrated and complex series of events where proteins in the blood have primary roles,
An Owner's Guide to Natural Healing Autologous Conditioned Plasma (ACP) Healing after an injury involves a well-orchestrated and complex series of events where proteins in the blood have primary roles,
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Recombinant and Autologous Platelet-Derived Growth Factors Page 1 of 20 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Recombinant and Autologous Platelet-Derived
Recombinant and Autologous Platelet-Derived Growth Factors Page 1 of 20 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Recombinant and Autologous Platelet-Derived
Considered Judgement Form
 Considered Judgement Form This form is a checklist of issues that may be considered by the Purchasing Guidance Advisory Group when making purchasing recommendations. Meeting date: 14 October 2014 Topic:
Considered Judgement Form This form is a checklist of issues that may be considered by the Purchasing Guidance Advisory Group when making purchasing recommendations. Meeting date: 14 October 2014 Topic:
Evidence Based Report Autologous Blood Injections for Musculoskeletal Disorders
 Evidence Based Report Autologous Blood Injections for Musculoskeletal Disorders Reviewer Mark Ayson Date Report Completed September 2014 Important Note: The purpose of this brief report is to summarise
Evidence Based Report Autologous Blood Injections for Musculoskeletal Disorders Reviewer Mark Ayson Date Report Completed September 2014 Important Note: The purpose of this brief report is to summarise
Platelet Rich Plasma (PRP) injections. by Dr George Pitsis
 Platelet Rich Plasma (PRP) injections by Dr George Pitsis Platelet Rich Plasma (PRP) injections have in the more recent years attracted significant attention as a clinical tools to assist with treatment
Platelet Rich Plasma (PRP) injections by Dr George Pitsis Platelet Rich Plasma (PRP) injections have in the more recent years attracted significant attention as a clinical tools to assist with treatment
IMPORTANT REMINDER DESCRIPTION
 Medical Policy Manual Medicine, Policy No. 40 Prolotherapy Next Review: January 2019 Last Review: February 2018 Effective: March 1, 2018 IMPORTANT REMINDER Medical Policies are developed to provide guidance
Medical Policy Manual Medicine, Policy No. 40 Prolotherapy Next Review: January 2019 Last Review: February 2018 Effective: March 1, 2018 IMPORTANT REMINDER Medical Policies are developed to provide guidance
Conflicts of Interest. I have no conflicts of interest regarding this presentation
 Conflicts of Interest I have no conflicts of interest regarding this presentation Ramon Ylanan MD CAQSM Team Physician University of Arkansas Advanced Orthopeadic Specialists Goals Background Healing Response
Conflicts of Interest I have no conflicts of interest regarding this presentation Ramon Ylanan MD CAQSM Team Physician University of Arkansas Advanced Orthopeadic Specialists Goals Background Healing Response
In the Treatment of Patients With Knee Joint Osteoarthritis, Are Platelet Rich Plasma Injections More Effective Than Hyaluronic Acid Injections?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2015 In the Treatment of Patients With Knee
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2015 In the Treatment of Patients With Knee
Platelet Rich Plasma: Hoax or Hope
 Platelet Rich Plasma: Hoax or Hope Peter Pryzbylkowski, MD Professional Background Double Boarded in both Anesthesiology and Pain Medicine Residency and Fellowship at the University of Pennsylvania Assistant
Platelet Rich Plasma: Hoax or Hope Peter Pryzbylkowski, MD Professional Background Double Boarded in both Anesthesiology and Pain Medicine Residency and Fellowship at the University of Pennsylvania Assistant
A Patient s Guide to Platelet-Rich Plasma Treatment of Musculoskeletal Compliments of: The Central Orthopedic Group
 A Patient s Guide to Platelet-Rich Plasma Treatment of Musculoskeletal Problems The Central Orthopedic Group 651 Old Country Road Plainview, NY 11803 Phone: 5166818822 Fax: 5166813332 p.lettieri@aol.com
A Patient s Guide to Platelet-Rich Plasma Treatment of Musculoskeletal Problems The Central Orthopedic Group 651 Old Country Road Plainview, NY 11803 Phone: 5166818822 Fax: 5166813332 p.lettieri@aol.com
Platelet-rich plasma application in the management of chronic tendinopathies
 Acta Orthop. Belg., 2013, 79, 10-15 REVIEW ARTICLE Platelet-rich plasma application in the management of chronic tendinopathies Jean-François Kaux, Jean-Michel Crielaard From the University and University
Acta Orthop. Belg., 2013, 79, 10-15 REVIEW ARTICLE Platelet-rich plasma application in the management of chronic tendinopathies Jean-François Kaux, Jean-Michel Crielaard From the University and University
Cigna Medical Coverage Policy
 Cigna Medical Coverage Policy Subject Autologous Platelet-Derived Growth Factors (Platelet-Rich Plasma [PRP]) Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information...
Cigna Medical Coverage Policy Subject Autologous Platelet-Derived Growth Factors (Platelet-Rich Plasma [PRP]) Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information...
Platelet-Rich Plasma Compared With Other Common Injection Therapies in the Treatment of Chronic Lateral Epicondylitis
 Journal of Sport Rehabilitation, 2016, 25, 77-82 http://dx.doi.org/10.1123/jsr.2014-0198 2016 Human Kinetics, Inc. CRITICALLY APPRAISED TOPIC Platelet-Rich Plasma Compared With Other Common Injection Therapies
Journal of Sport Rehabilitation, 2016, 25, 77-82 http://dx.doi.org/10.1123/jsr.2014-0198 2016 Human Kinetics, Inc. CRITICALLY APPRAISED TOPIC Platelet-Rich Plasma Compared With Other Common Injection Therapies
A Patient s Guide to Platelet-Rich Plasma Treatment of Musculoskeletal Problems
 A Patient s Guide to Platelet-Rich Plasma Treatment of Musculoskeletal Problems Iain is a specialist in musculoskeletal imaging and the diagnosis of musculoskeletal pain. This information is provided with
A Patient s Guide to Platelet-Rich Plasma Treatment of Musculoskeletal Problems Iain is a specialist in musculoskeletal imaging and the diagnosis of musculoskeletal pain. This information is provided with
Biologics in ACL: What s the Data?
 Biologics in ACL: What s the Data? Jo A. Hannafin, M.D., Ph.D. Professor of Orthopaedic Surgery, Weill Cornell Medical College Attending Orthopaedic Surgeon and Senior Scientist Sports Medicine and Shoulder
Biologics in ACL: What s the Data? Jo A. Hannafin, M.D., Ph.D. Professor of Orthopaedic Surgery, Weill Cornell Medical College Attending Orthopaedic Surgeon and Senior Scientist Sports Medicine and Shoulder
Corporate Medical Policy
 Corporate Medical Policy Continuous Passive Motion in the Home Setting File Name: Origination: Last CAP Review: Next CAP Review: Last Review: continuous_passive_motion_in_the_home_setting 9/1993 6/2018
Corporate Medical Policy Continuous Passive Motion in the Home Setting File Name: Origination: Last CAP Review: Next CAP Review: Last Review: continuous_passive_motion_in_the_home_setting 9/1993 6/2018
BIOLOGICS STEM CELL AND PLATELET- RICH PLASMA FOR JOINT MANAGEMENT 1/10/ AAOS ANNUAL MEETING 2018 AAOS ANNUAL MEETING
 STEM CELL AND PLATELET- RICH PLASMA FOR JOINT MANAGEMENT BIOLOGICS o Injectable therapies that may suppress inflammation and promote regenerative pathways o Natural products that are harvested and are
STEM CELL AND PLATELET- RICH PLASMA FOR JOINT MANAGEMENT BIOLOGICS o Injectable therapies that may suppress inflammation and promote regenerative pathways o Natural products that are harvested and are
KNEE INJURIES IN SPORTS MEDICINE
 KNEE INJURIES IN SPORTS MEDICINE Irving Raphael, MD June 13, 2014 RSM Medical Associates Head Team Physician Syracuse University Outline Meniscal Injuries anatomy Exam Treatment ACL Injuries Etiology Physical
KNEE INJURIES IN SPORTS MEDICINE Irving Raphael, MD June 13, 2014 RSM Medical Associates Head Team Physician Syracuse University Outline Meniscal Injuries anatomy Exam Treatment ACL Injuries Etiology Physical
Treatment of Lateral Elbow Tendinopathy: Medical and Surgical Interventions
 APPENDIX G Treatment of Lateral Elbow Tendinopathy: Medical and Surgical Interventions The purpose of this document is to provide information for physiotherapists of common medical and surgical interventions
APPENDIX G Treatment of Lateral Elbow Tendinopathy: Medical and Surgical Interventions The purpose of this document is to provide information for physiotherapists of common medical and surgical interventions
Therapy with PRP. Flachau S.I.T.E.M.S.H Dr. Jürgen BARTHOFER. Trauma Hospital Linz - Austria Team Doctor ÖSV (Ski Jumping and Nordic Combined)
 Dr. Jürgen BARTHOFER Trauma Hospital Linz - Austria Team Doctor ÖSV (Ski Jumping and Nordic Combined) Flachau S.I.T.E.M.S.H 2014 Where are we? Methods : PRP (Platelet-rich Plasma) Buffy-Coat / Plasma-based
Dr. Jürgen BARTHOFER Trauma Hospital Linz - Austria Team Doctor ÖSV (Ski Jumping and Nordic Combined) Flachau S.I.T.E.M.S.H 2014 Where are we? Methods : PRP (Platelet-rich Plasma) Buffy-Coat / Plasma-based
Anterior Cruciate Ligament (ACL) Injuries
 Anterior Cruciate Ligament (ACL) Injuries Mark L. Wood, MD The anterior cruciate ligament (ACL) is one of the most commonly injured ligaments of the knee. The incidence of ACL injuries is currently estimated
Anterior Cruciate Ligament (ACL) Injuries Mark L. Wood, MD The anterior cruciate ligament (ACL) is one of the most commonly injured ligaments of the knee. The incidence of ACL injuries is currently estimated
Priorities Forum Statement
 Priorities Forum Statement Number 75 Subject Autologous Blood and Platelet-Rich Plasma Injection in Tendinopathy Date of decision February 2017 Date refreshed February 2017 Date of review February 2020
Priorities Forum Statement Number 75 Subject Autologous Blood and Platelet-Rich Plasma Injection in Tendinopathy Date of decision February 2017 Date refreshed February 2017 Date of review February 2020
Foot, Ankle, Knee & Hip Surgery Update. What s s New. Platelet Rich Plasma (PRP) Platelet Rich Plasma Total Ankle Replacement.
 Foot, Ankle, Knee & Hip Surgery Update Geoffrey S. Landis D.O. April 29, 2010 Southwestern Conference on Medicine What s s New Platelet Rich Plasma Total Platelet Rich Plasma (PRP) Why- Need or Desire
Foot, Ankle, Knee & Hip Surgery Update Geoffrey S. Landis D.O. April 29, 2010 Southwestern Conference on Medicine What s s New Platelet Rich Plasma Total Platelet Rich Plasma (PRP) Why- Need or Desire
Arthroscopic Rotator Cuff Repair Techniques What should we really be doing?
 COA 2014 Arthroscopic Rotator Cuff Repair Techniques What should we really be doing? C. Benjamin Ma, MD Chief, Sports Medicine and Shoulder Surgery University of California, San Francisco Department of
COA 2014 Arthroscopic Rotator Cuff Repair Techniques What should we really be doing? C. Benjamin Ma, MD Chief, Sports Medicine and Shoulder Surgery University of California, San Francisco Department of
Clinical Policy Title: Growth factor treatment for wound healing/musculoskeletal uses
 Clinical Policy Title: Growth factor treatment for wound healing/musculoskeletal uses Clinical Policy Number: 16.02.02 Effective Date: January 1, 2015 Initial Review Date: August 20, 2014 Most Recent Review
Clinical Policy Title: Growth factor treatment for wound healing/musculoskeletal uses Clinical Policy Number: 16.02.02 Effective Date: January 1, 2015 Initial Review Date: August 20, 2014 Most Recent Review
Platelet Rich Plasma (PRP) Dania Segreti, SPT Vanguard In-service - July 31, 2013
 Platelet Rich Plasma (PRP) Dania Segreti, SPT Vanguard In-service - July 31, 2013 What is PRP? In medicine since 1970s First uses in bone healing began in late 1990s Gained popularity for tissue healing
Platelet Rich Plasma (PRP) Dania Segreti, SPT Vanguard In-service - July 31, 2013 What is PRP? In medicine since 1970s First uses in bone healing began in late 1990s Gained popularity for tissue healing
John J Christoforetti, MD Pittsburgh, Pennsylvania
 ARTHROSCOPIC ASSISTED PROXIMAL HAMSTRINGS REPAIR WITH HUMAN ACELLULAR DERMAL ALLOGRAFT PATCH AUGMENTATION FOR REVISION OF FAILED PROXIMAL HAMSTRINGS REPAIR: SHORT TERM CLINICAL AND MRI RESULT John J Christoforetti,
ARTHROSCOPIC ASSISTED PROXIMAL HAMSTRINGS REPAIR WITH HUMAN ACELLULAR DERMAL ALLOGRAFT PATCH AUGMENTATION FOR REVISION OF FAILED PROXIMAL HAMSTRINGS REPAIR: SHORT TERM CLINICAL AND MRI RESULT John J Christoforetti,
ACL Athletic Career. ACL Rupture - Warning Features Intensive pain Immediate swelling Locking Feel a Pop Dead leg Cannot continue to play
 FIMS Ambassador Tour to Eastern Europe, 2004 Belgrade, Serbia Montenegro Acute Knee Injuries - Controversies and Challenges Professor KM Chan OBE, JP President of FIMS Belgrade ACL Athletic Career ACL
FIMS Ambassador Tour to Eastern Europe, 2004 Belgrade, Serbia Montenegro Acute Knee Injuries - Controversies and Challenges Professor KM Chan OBE, JP President of FIMS Belgrade ACL Athletic Career ACL
Welcome to White Rock Orthopaedic Surgery Centre
 White Rock Orthopaedic Surgery Excellence in Orthopaedic Surgery since 2008 Over 700 surgical cases (spring of 2013) Fully Accredited Class-1 Facility Expedient initial assessment Secure, online registration
White Rock Orthopaedic Surgery Excellence in Orthopaedic Surgery since 2008 Over 700 surgical cases (spring of 2013) Fully Accredited Class-1 Facility Expedient initial assessment Secure, online registration
Clinical Policy Title: Growth factor treatment for wound healing/musculoskeletal uses
 Clinical Policy Title: Growth factor treatment for wound healing/musculoskeletal uses Clinical Policy Number: 16.02.02 Effective Date: January 1, 2015 Initial Review Date: August 20, 2014 Most Recent Review
Clinical Policy Title: Growth factor treatment for wound healing/musculoskeletal uses Clinical Policy Number: 16.02.02 Effective Date: January 1, 2015 Initial Review Date: August 20, 2014 Most Recent Review
REFERENCES for PLATELET RICH PLASMA (PRP)
 REFERENCES for PLATELET RICH PLASMA (PRP) Daif ET. Autologous blood injection as a new treatment modality for chronic recurrent temporomandibular joint dislocation. Oral Surg Oral Med Oral Pathol Oral
REFERENCES for PLATELET RICH PLASMA (PRP) Daif ET. Autologous blood injection as a new treatment modality for chronic recurrent temporomandibular joint dislocation. Oral Surg Oral Med Oral Pathol Oral
Platelet Rich Plasma CLINICAL BRIEF
 Platelet Rich Plasma CLINICAL BRIEF Introduction As physicians continue to seek biologic solutions to improve healing of soft tissue pathologies, worldwide interest in the use of platelet rich plasma (PRP)
Platelet Rich Plasma CLINICAL BRIEF Introduction As physicians continue to seek biologic solutions to improve healing of soft tissue pathologies, worldwide interest in the use of platelet rich plasma (PRP)
Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June Section: Surgery Last Reviewed Date: June 2013
 Medical Policy Manual Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June 2007 Section: Surgery Last Reviewed Date: June 2013 Policy No: 157 Effective Date: August 1, 2013 IMPORTANT REMINDER
Medical Policy Manual Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June 2007 Section: Surgery Last Reviewed Date: June 2013 Policy No: 157 Effective Date: August 1, 2013 IMPORTANT REMINDER
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: July 15, 2017 Related Policies: 1.01.05 Ultrasound Accelerated Fracture Healing Device 7.01.07 Electrical Bone Growth Stimulation of the Appendicular Skeleton
FEP Medical Policy Manual Effective Date: July 15, 2017 Related Policies: 1.01.05 Ultrasound Accelerated Fracture Healing Device 7.01.07 Electrical Bone Growth Stimulation of the Appendicular Skeleton
LARS (Ligament Augmentation & Reconstruction System) Literature
 LARS-Related Studies and Papers ACL: 1. Level of Evidence: IV Li, H. et al (2011). Enhancement of the osseointegration of a polyethylene Terephthalate artificial ligament graft in a bone tunnel using 58S
LARS-Related Studies and Papers ACL: 1. Level of Evidence: IV Li, H. et al (2011). Enhancement of the osseointegration of a polyethylene Terephthalate artificial ligament graft in a bone tunnel using 58S
Tendon Fenestration. Disclosures. Outline: questions. Introduction: Peritendon Steroid Injections. Jon A. Jacobson, MD. Patellar Tendon: tendinosis
 Tendon Fenestration Jon A. Jacobson, MD Professor of Radiology Director, Division of Musculoskeletal Radiology University of Michigan Disclosures Consultant: Bioclinica Advisory Board: GE, Philips Book
Tendon Fenestration Jon A. Jacobson, MD Professor of Radiology Director, Division of Musculoskeletal Radiology University of Michigan Disclosures Consultant: Bioclinica Advisory Board: GE, Philips Book
Biologics for Sports Injuries
 Biologics for Sports Injuries Jason L. Dragoo, MD Associate Professor Department of Orthopaedic Surgery Introduction Survey of common conditions Cover only injuries with evidence NOT RTC RTC Pectoralis
Biologics for Sports Injuries Jason L. Dragoo, MD Associate Professor Department of Orthopaedic Surgery Introduction Survey of common conditions Cover only injuries with evidence NOT RTC RTC Pectoralis
Adult Reconstruction Hip Education Tracks
 Adult Reconstruction Hip Education Tracks Adult Reconstruction Hip Track for the Specialist - HIP1 ICL 281 A Case-based Approach to High Risk Total Hip - When Do I Do Something Differently? ICL 241 The
Adult Reconstruction Hip Education Tracks Adult Reconstruction Hip Track for the Specialist - HIP1 ICL 281 A Case-based Approach to High Risk Total Hip - When Do I Do Something Differently? ICL 241 The
STATE OF THE ART OF ACL SURGERY (Advancements that have had an impact)
 STATE OF THE ART OF ACL SURGERY (Advancements that have had an impact) David Drez, Jr., M.D. Clinical Professor of Orthopaedics LSU School of Medicine Financial Disclosure Dr. David Drez has no relevant
STATE OF THE ART OF ACL SURGERY (Advancements that have had an impact) David Drez, Jr., M.D. Clinical Professor of Orthopaedics LSU School of Medicine Financial Disclosure Dr. David Drez has no relevant
Sonographically occult intrasubstance tendon tears revealed by platelet rich plasma injection: evidence of a frequently overlooked pathology?
 Sonographically occult intrasubstance tendon tears revealed by platelet rich plasma injection: evidence of a frequently overlooked pathology? Poster No.: C-1603 Congress: ECR 2013 Type: Scientific Exhibit
Sonographically occult intrasubstance tendon tears revealed by platelet rich plasma injection: evidence of a frequently overlooked pathology? Poster No.: C-1603 Congress: ECR 2013 Type: Scientific Exhibit
Section: Medicine Last Reviewed Date: June Policy No: 130 Effective Date: September 1, 2014
 Medical Policy Manual Topic: Manipulation Under Anesthesia for the Treatment of Pain Date of Origin: April 2009 Section: Medicine Last Reviewed Date: June 2014 Policy No: 130 Effective Date: September
Medical Policy Manual Topic: Manipulation Under Anesthesia for the Treatment of Pain Date of Origin: April 2009 Section: Medicine Last Reviewed Date: June 2014 Policy No: 130 Effective Date: September
MEDICAL POLICY SUBJECT: COMPUTER ASSISTED NAVIGATION FOR KNEE AND HIP ARTHROPLASTY
 MEDICAL POLICY SUBJECT: COMPUTER ASSISTED PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: COMPUTER ASSISTED PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: COMPUTER ASSISTED NAVIGATION FOR KNEE AND HIP ARTHROPLASTY
 MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
Goals and Objectives for the Orthopaedic Surgery Resident McGill Orthopaedic Sports Medicine and Minimally Invasive (MGH & Shriners) Junior Residents
 Goals and Objectives for the Orthopaedic Surgery Resident McGill Orthopaedic Sports Medicine and Minimally Invasive (MGH & Shriners) Junior Residents The following document is intended to guide you in
Goals and Objectives for the Orthopaedic Surgery Resident McGill Orthopaedic Sports Medicine and Minimally Invasive (MGH & Shriners) Junior Residents The following document is intended to guide you in
Shockwave Therapies for Musculoskeletal Problems Useful Literature
 Shockwave Therapies for Musculoskeletal Problems Useful Literature I have some 1600 references related to shockwave therapy. Those listed below are the main musculoskeletal related papers from 2005-07
Shockwave Therapies for Musculoskeletal Problems Useful Literature I have some 1600 references related to shockwave therapy. Those listed below are the main musculoskeletal related papers from 2005-07
Medical Policy Manual. Topic: Peripheral Subcutaneous Field Stimulation Date of Origin: April Section: Surgery Last Reviewed Date: April 2014
 Medical Policy Manual Topic: Peripheral Subcutaneous Field Stimulation Date of Origin: April 2013 Section: Surgery Last Reviewed Date: April 2014 Policy No: 188 Effective Date: July 1, 2014 IMPORTANT REMINDER
Medical Policy Manual Topic: Peripheral Subcutaneous Field Stimulation Date of Origin: April 2013 Section: Surgery Last Reviewed Date: April 2014 Policy No: 188 Effective Date: July 1, 2014 IMPORTANT REMINDER
Torn ACL - Anatomic Footprint ACL Reconstruction
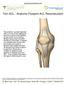 Torn ACL - Anatomic Footprint ACL Reconstruction The anterior cruciate ligament (ACL) is one of four ligaments that are crucial to the stability of your knee. It is a strong fibrous tissue that connects
Torn ACL - Anatomic Footprint ACL Reconstruction The anterior cruciate ligament (ACL) is one of four ligaments that are crucial to the stability of your knee. It is a strong fibrous tissue that connects
Goals and Objectives for the Orthopaedic Surgery Resident McGill Orthopaedic Sports Medicine (SMH) Knee & Shoulder Surgery
 Goals and Objectives for the Orthopaedic Surgery Resident McGill Orthopaedic Sports Medicine (SMH) Knee & Shoulder Surgery PGY 1 & 2 1. Medical Expert A. Basic Science a) General Knowledge of functional
Goals and Objectives for the Orthopaedic Surgery Resident McGill Orthopaedic Sports Medicine (SMH) Knee & Shoulder Surgery PGY 1 & 2 1. Medical Expert A. Basic Science a) General Knowledge of functional
Page 1 of 6. Appendix 1
 Page 1 Appendix 1 Rotation Objectives and Schedule 1. Introductory Month 4 weeks 2. Total Joints 4 weeks a. Diagnosis and management of hip and knee arthritis b. Indications for surgery c. Implant selection;
Page 1 Appendix 1 Rotation Objectives and Schedule 1. Introductory Month 4 weeks 2. Total Joints 4 weeks a. Diagnosis and management of hip and knee arthritis b. Indications for surgery c. Implant selection;
Autologous Platelets for Treatment of Tendonopathy of the Foot and Ankle
 Autologous Platelets for Treatment of Tendonopathy of the Foot and Ankle BY ADAM M BUDNY, DPM This article is written exclusively for Podiatry Management by the American College of Foot and Ankle Surgeons
Autologous Platelets for Treatment of Tendonopathy of the Foot and Ankle BY ADAM M BUDNY, DPM This article is written exclusively for Podiatry Management by the American College of Foot and Ankle Surgeons
Humber. Arthroscopy Knee
 Humber Arthroscopy Knee Intervention Diagnostic & Therapeutic Arthroscopy Knee OPCS Codes W85 Therapeutic endoscopic operations on cavity of knee joint W851 Endoscopic removal of loose body from knee joint
Humber Arthroscopy Knee Intervention Diagnostic & Therapeutic Arthroscopy Knee OPCS Codes W85 Therapeutic endoscopic operations on cavity of knee joint W851 Endoscopic removal of loose body from knee joint
Platelet-Rich Plasma Can Be Used to Successfully Treat Elbow Ulnar Collateral Ligament Insufficiency in High- Level Throwers
 Platelet-Rich Plasma Can Be Used to Successfully Treat Elbow Ulnar Collateral Ligament Insufficiency in High- Level Throwers Am J Orthop. 2016 May;45(4):296-300 Authors: Dines JS Williams PN Elattrache
Platelet-Rich Plasma Can Be Used to Successfully Treat Elbow Ulnar Collateral Ligament Insufficiency in High- Level Throwers Am J Orthop. 2016 May;45(4):296-300 Authors: Dines JS Williams PN Elattrache
ORTHOPEDIC SPECIALISTS STEM CELLS FOR THE TREATMENT OF PAIN DISCOVERING A NEW PATH TO WELLNESS
 ORTHOPEDIC SPECIALISTS STEM CELLS FOR THE TREATMENT OF PAIN DISCOVERING A NEW PATH TO WELLNESS A LETTER TO OUR PATIENTS Dear Patient, As your healthcare provider, it is our medical obligation to provide
ORTHOPEDIC SPECIALISTS STEM CELLS FOR THE TREATMENT OF PAIN DISCOVERING A NEW PATH TO WELLNESS A LETTER TO OUR PATIENTS Dear Patient, As your healthcare provider, it is our medical obligation to provide
Tendon & Ligament Application of PRP
 Tendon & Ligament Application of PRP Sang Chul Lee, M.D, PhD. Department of Physical Medicine & Rehabilitation, Myongji hospital, Kwandong University College of Medicine Tendon and Ligament Low metabolic
Tendon & Ligament Application of PRP Sang Chul Lee, M.D, PhD. Department of Physical Medicine & Rehabilitation, Myongji hospital, Kwandong University College of Medicine Tendon and Ligament Low metabolic
SPORTS MEDICINE OVERUSE MANAGEMENT PRINCIPLES FOR
 SPORTS MEDICINE OVERUSE INJURIES: DIAGNOSIS AND MANAGEMENT PRINCIPLES FOR THE FAMILY PHYSICIAN DR NG CHUNG SIEN MBBS (SPORE) MSPMED (AUSTRALIA) DFD CAW SENIOR REGISTRAR CHANGI SPORTS MEDICINE CENTRE CHANGI
SPORTS MEDICINE OVERUSE INJURIES: DIAGNOSIS AND MANAGEMENT PRINCIPLES FOR THE FAMILY PHYSICIAN DR NG CHUNG SIEN MBBS (SPORE) MSPMED (AUSTRALIA) DFD CAW SENIOR REGISTRAR CHANGI SPORTS MEDICINE CENTRE CHANGI
Rotator Cuff Repair Outcomes. Patrick Birmingham, MD
 Rotator Cuff Repair Outcomes Patrick Birmingham, MD Outline Arthroscopic Vs. Mini-open Subjective Outcomes Objective Outcomes Timing Arthroscopic Vs. Mini-open Sauerbrey Arthroscopy 2005 Twenty-six patients
Rotator Cuff Repair Outcomes Patrick Birmingham, MD Outline Arthroscopic Vs. Mini-open Subjective Outcomes Objective Outcomes Timing Arthroscopic Vs. Mini-open Sauerbrey Arthroscopy 2005 Twenty-six patients
Platelet-Rich Plasma in the Lower Extremity
 Platelet-Rich Plasma in the Lower Extremity Angelo J. Colosimo, MD Head Orthopaedic Surgeon University of Cincinnati Athletics Director of Sports Medicine University of Cincinnati Medical Center Associate
Platelet-Rich Plasma in the Lower Extremity Angelo J. Colosimo, MD Head Orthopaedic Surgeon University of Cincinnati Athletics Director of Sports Medicine University of Cincinnati Medical Center Associate
Platelet Rich Plasma In Musculoskeletal Pathologies
 Western University Scholarship@Western Electronic Thesis and Dissertation Repository May 2016 Platelet Rich Plasma In Musculoskeletal Pathologies Nicole L. Kaniki The University of Western Ontario Supervisor
Western University Scholarship@Western Electronic Thesis and Dissertation Repository May 2016 Platelet Rich Plasma In Musculoskeletal Pathologies Nicole L. Kaniki The University of Western Ontario Supervisor
Osteochondral allografting for all other joints is not covered as the evidence is insufficient to determine the
 Medical Coverage Policy Osteochondral Autologous Chrondrocyte Implantation for Focal Articular Cartilage Lesions EFFECTIVE DATE: 05 20 2008 POLICY LAST UPDATED: 10 16 2018 OVERVIEW A variety of procedures
Medical Coverage Policy Osteochondral Autologous Chrondrocyte Implantation for Focal Articular Cartilage Lesions EFFECTIVE DATE: 05 20 2008 POLICY LAST UPDATED: 10 16 2018 OVERVIEW A variety of procedures
Clinical Policy: EpiFix Wound Treatment
 Clinical Policy: Reference Number: PA.CP.MP.140 Effective Date: 03/18 Last Review Date: 04/18 Coding Implications Revision Log Description EpiFix (MiMedx Group) is dehydrated human amniotic tissue that
Clinical Policy: Reference Number: PA.CP.MP.140 Effective Date: 03/18 Last Review Date: 04/18 Coding Implications Revision Log Description EpiFix (MiMedx Group) is dehydrated human amniotic tissue that
Chronic musculoskeletal pain accounts for the most commonly reported medical
 The Emerging Use of Platelet-Rich Plasma in Musculoskeletal Medicine Jamil Bashir, MD Alberto J. Panero, DO Andrew L. Sherman, MD From the Department of Physical Medicine and Rehabilitation at the University
The Emerging Use of Platelet-Rich Plasma in Musculoskeletal Medicine Jamil Bashir, MD Alberto J. Panero, DO Andrew L. Sherman, MD From the Department of Physical Medicine and Rehabilitation at the University
COMPARATIVE EFFECTIVENESS OF ULTRASOUND GUIDED INTRATENDINOUS PROLOTHERAPY INJECTION WITH CONVENTIONAL TREATMENT TO TREAT
 SCIENTIFIC ARTICLE COMPARATIVE EFFECTIVENESS OF ULTRASOUND GUIDED INTRATENDINOUS PROLOTHERAPY INJECTION WITH CONVENTIONAL TREATMENT TO TREAT FOCAL SUPRASPINATUS TENDINOSIS : RANDOMISED CONTROL STUDY 1,2
SCIENTIFIC ARTICLE COMPARATIVE EFFECTIVENESS OF ULTRASOUND GUIDED INTRATENDINOUS PROLOTHERAPY INJECTION WITH CONVENTIONAL TREATMENT TO TREAT FOCAL SUPRASPINATUS TENDINOSIS : RANDOMISED CONTROL STUDY 1,2
Inflammation is Not the Enemy
 6/22/2017 Inflammation is Not the Enemy Sean Mulvaney, MD 1 6/22/2017 2 6/22/2017 Lascaux 7.4 Billion 3 This image cannot currently be displayed. 6/22/2017 Goals 4 ANTI INFLAMMATORY THERAPIES NSAIDS 5
6/22/2017 Inflammation is Not the Enemy Sean Mulvaney, MD 1 6/22/2017 2 6/22/2017 Lascaux 7.4 Billion 3 This image cannot currently be displayed. 6/22/2017 Goals 4 ANTI INFLAMMATORY THERAPIES NSAIDS 5
Massive Rotator Cuff Tears. Rafael M. Williams, MD
 Massive Rotator Cuff Tears Rafael M. Williams, MD Rotator Cuff MRI MRI Small / Partial Thickness Medium Tear Arthroscopic View Massive Tear Fatty Atrophy Arthroscopic View MassiveTears Tear is > 5cm
Massive Rotator Cuff Tears Rafael M. Williams, MD Rotator Cuff MRI MRI Small / Partial Thickness Medium Tear Arthroscopic View Massive Tear Fatty Atrophy Arthroscopic View MassiveTears Tear is > 5cm
Prolotherapy. Description
 Subject: Prolotherapy Page: 1 of 9 Last Review Status/Date: December 2014 Prolotherapy Description Prolotherapy describes a procedure intended for healing and strengthening ligaments and tendons by injecting
Subject: Prolotherapy Page: 1 of 9 Last Review Status/Date: December 2014 Prolotherapy Description Prolotherapy describes a procedure intended for healing and strengthening ligaments and tendons by injecting
AN INTRODUCTION TO REGENERATIVE MEDICINE
 AN INTRODUCTION TO REGENERATIVE MEDICINE You ve undoubtedly come across some discussion of stem cells, likely with regard to stem cell research. But stem cells have a wide variety of uses in the medical
AN INTRODUCTION TO REGENERATIVE MEDICINE You ve undoubtedly come across some discussion of stem cells, likely with regard to stem cell research. But stem cells have a wide variety of uses in the medical
REVISITING SOME CONTROVERSIAL TOPICS
 Volume 22 Dr. Jacob Lazarovic SVP/Chief Medical Officer TAKING OUR PULSE REVISITING SOME CONTROVERSIAL TOPICS Practicing medicine, or making decisions about the appropriateness of treatment, and the presence
Volume 22 Dr. Jacob Lazarovic SVP/Chief Medical Officer TAKING OUR PULSE REVISITING SOME CONTROVERSIAL TOPICS Practicing medicine, or making decisions about the appropriateness of treatment, and the presence
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: July15, 2017 Related Policies: 2.01.16 Recombinant and Autologous Platelet-Derived Growth Factors for Healing and Other Non Orthopedic Conditions 7.01.113 Bioengineered
FEP Medical Policy Manual Effective Date: July15, 2017 Related Policies: 2.01.16 Recombinant and Autologous Platelet-Derived Growth Factors for Healing and Other Non Orthopedic Conditions 7.01.113 Bioengineered
Forms list... 2 Examples of a few form layouts Scannable Forms... 13
 Forms Forms list... 2 Examples of a few form layouts... 10 Scannable Forms... 13 There are forms available to match all the screens in the Socrates database. They are available in either a word.doc format
Forms Forms list... 2 Examples of a few form layouts... 10 Scannable Forms... 13 There are forms available to match all the screens in the Socrates database. They are available in either a word.doc format
The Society for Patient Centered Orthopedics. Choosing Wisely List. James Rickert, MD 1
 The Society for Patient Centered Orthopedics Choosing Wisely List James Rickert, MD 1 Extremities and Trauma Vertebroplasty Rotator Cuff Repair: For atraumatic (degenerative) tears in patients greater
The Society for Patient Centered Orthopedics Choosing Wisely List James Rickert, MD 1 Extremities and Trauma Vertebroplasty Rotator Cuff Repair: For atraumatic (degenerative) tears in patients greater
Anterior Cruciate Ligament (ACL) Tears
 WASHINGTON UNIVERSITY ORTHOPEDICS Anterior Cruciate Ligament (ACL) Tears Knowing what to expect for ACL surgery is key for a healthy surgery and recovery. Our sports medicine specialists are committed
WASHINGTON UNIVERSITY ORTHOPEDICS Anterior Cruciate Ligament (ACL) Tears Knowing what to expect for ACL surgery is key for a healthy surgery and recovery. Our sports medicine specialists are committed
Corporate Medical Policy
 Corporate Medical Policy Electrical Bone Growth Stimulation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: electrical_bone_growth_stimulation 4/1981 6/2017 6/2018 6/2017 Description
Corporate Medical Policy Electrical Bone Growth Stimulation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: electrical_bone_growth_stimulation 4/1981 6/2017 6/2018 6/2017 Description
Survivorship After Meniscal Allograft Transplantation According To Articular Cartilage Status
 # 154134 Survivorship After Meniscal Allograft Transplantation According To Articular Cartilage Status Jun-Gu Park, Seong-Il Bin, Jong-Min Kim, Bum Sik Lee Department of Orthopaedic Surgery, Asan Medical
# 154134 Survivorship After Meniscal Allograft Transplantation According To Articular Cartilage Status Jun-Gu Park, Seong-Il Bin, Jong-Min Kim, Bum Sik Lee Department of Orthopaedic Surgery, Asan Medical
3/21/2011 PCL INJURY WITH OPERATIVE TREATMENT A CASE STUDY PCL PCL MECHANISM OF INJURY PCL PREVALENCE
 PCL PCL INJURY WITH OPERATIVE TREATMENT A CASE STUDY K. Anderson, S. Hjortedal, Y. Jingi, E. Sutcliffe & S. Witschen Washington State University Origin Posterior aspect of tibia Insertion Medial femoral
PCL PCL INJURY WITH OPERATIVE TREATMENT A CASE STUDY K. Anderson, S. Hjortedal, Y. Jingi, E. Sutcliffe & S. Witschen Washington State University Origin Posterior aspect of tibia Insertion Medial femoral
Extracorporeal Shockwave Therapy. Outcomes in Shoulder Tendinopathy and Plantar Fasciitis. American University of Beirut Medical Center
 Extracorporeal Shockwave Therapy. Outcomes in Shoulder Tendinopathy and Plantar Fasciitis Nagham HADDAD, PT Nagham HADDAD, PT American University of Beirut Medical Center Introduction: Tendinosis is the
Extracorporeal Shockwave Therapy. Outcomes in Shoulder Tendinopathy and Plantar Fasciitis Nagham HADDAD, PT Nagham HADDAD, PT American University of Beirut Medical Center Introduction: Tendinosis is the
The healthcare environment is
 Review Article Platelet-rich Plasma in Orthopaedic Applications: Evidence-based Recommendations for Treatment Downloaded from http://journals.lww.com/jaaos by k38zdthxkv6xg2uya2wsbisxlamxpb/xenqi6wdshkkgg3a+mcn2rqarcprdp+lutjpipmob6bnm1owem6xa6l/kfgyxmyzaq6rkxqdhadam14buwtbhhljupt3iwmhetb6kybrxtrg=
Review Article Platelet-rich Plasma in Orthopaedic Applications: Evidence-based Recommendations for Treatment Downloaded from http://journals.lww.com/jaaos by k38zdthxkv6xg2uya2wsbisxlamxpb/xenqi6wdshkkgg3a+mcn2rqarcprdp+lutjpipmob6bnm1owem6xa6l/kfgyxmyzaq6rkxqdhadam14buwtbhhljupt3iwmhetb6kybrxtrg=
Ankle Arthritis PATIENT INFORMATION. The ankle joint. What is ankle arthritis?
 PATIENT INFORMATION Ankle Arthritis The ankle joint The ankle is a very complex joint. It is actually made up of two joints: the true ankle joint and the subtalar ankle joint. The ankle joint consists
PATIENT INFORMATION Ankle Arthritis The ankle joint The ankle is a very complex joint. It is actually made up of two joints: the true ankle joint and the subtalar ankle joint. The ankle joint consists
Protocol. Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion Procedures
 Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion (70185) Medical Benefit Effective Date: 07/01/12 Next Review Date: 01/19 Preauthorization Yes Review Dates: 09/07, 09/08, 09/09, 05/10,
Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion (70185) Medical Benefit Effective Date: 07/01/12 Next Review Date: 01/19 Preauthorization Yes Review Dates: 09/07, 09/08, 09/09, 05/10,
SUPPLEMENTAL DIGITAL CONTENT (SDC)
 Orozco_Osteoarthritis_MSC_v4.3.doc Mar_1_2013 1 SUPPLEMENTAL DIGITAL CONTENT (SDC) Contents: SUPPLEMENTARY TABLES Supplementary Table S1. Antecedent history of the patients included in this trial. Supplementary
Orozco_Osteoarthritis_MSC_v4.3.doc Mar_1_2013 1 SUPPLEMENTAL DIGITAL CONTENT (SDC) Contents: SUPPLEMENTARY TABLES Supplementary Table S1. Antecedent history of the patients included in this trial. Supplementary
Prof. FABIO CERZA A.Carcangiu, S.Carnì, A. Pecora, I. Di Vavo, V. Schiavilla
 Department of Orthopaedics and Traumatology P.Colombo Hospital Velletri(ROME) Prof. FABIO CERZA A.Carcangiu, S.Carnì, A. Pecora, I. Di Vavo, V. Schiavilla 4 million Italians suffer from arthrosis Most
Department of Orthopaedics and Traumatology P.Colombo Hospital Velletri(ROME) Prof. FABIO CERZA A.Carcangiu, S.Carnì, A. Pecora, I. Di Vavo, V. Schiavilla 4 million Italians suffer from arthrosis Most
Comparison of outcomes in patients with generalized ligamentous laxity and without generalized laxity in the arthroscopic Modified Broströ m
 Comparison of outcomes in patients with generalized ligamentous laxity and without generalized laxity in the arthroscopic Modified Broströ m Operation for Chronic Lateral Ankle Instability NO CONFLICT
Comparison of outcomes in patients with generalized ligamentous laxity and without generalized laxity in the arthroscopic Modified Broströ m Operation for Chronic Lateral Ankle Instability NO CONFLICT
