IVI Cholera Activities
|
|
|
- Milo Hubbard
- 5 years ago
- Views:
Transcription
1 IVI Cholera Activities 5th Initiative Against Diarrheal and Enteric Diseases in Asia (IDEA) Hanoi, March 6-9, Dr. Julia Lynch Deputy Director General Development and Delivery
2 Outline Introduce IVI Review OCV Development morcvax Shanchol Euvichol Cholvax Single Dose Study Campaigns and Demonstration projects 2
3 IVI is a Vaccine R&D center with a Global Health mission Developing countries free of suffering from infectious diseases Discover, develop and deliver safe, effective and affordable vaccines for global public health International Organization UNDP initiative First international organization in Korea (1997) 35 countries and WHO as state parties Global Vaccine Research Institute HQ and laboratory in Seoul Field programs in 29 countries: Asia, Africa, Latin America 3
4 IVI Full Spectrum: Bench to Delivery Pathogen genotyping Novel antigens Novel adjuvants New delivery mechanisms New routes of administration Laboratory process development Assay development Technology transfer for large-scale production Clinical development Regulatory expertise Epidemiological and Socioeconomic studies Vaccine feasibility/acceptance Field effectiveness studies Cost-effectiveness and impact analyses Dissemination to stakeholders DCVM
5 Development of Killed Oral WC Vaccine Technology transfer from University of Gothenburg, Professor Jan Holmgren to VABIOTECH in 1980s Inactivated Whole Cell-only vaccine, without CTB component Vaccine reformulated by VABIOTECH, proven safe and effective in Vietnamese people and licensed as ORC-Vax in 1997 IVI engaged VABIOTECH to modify and reformulate vaccine to meet WHO standards (2004) Modification of Strains, Production, Standardization & QC A Safety and Immunogenicity Study of a 2-Dose Regimen of the Reformulated Bivalent Killed Oral Cholera Vaccine in Vietnamese Subjects (NIHE/IVI) Licensed in Vietnam (morcvax )
6 Cholera Vaccine (Inactivated Whole Cell Oral Cholera vaccine) IVI Technology Transfers Company Partnership Stage of development Vabiotech (Vie tnam) IVI re-formulated, redeveloped process to meet WHO standards. Licensed in Vietnam (morcvax ) Shantha (India) Technology transfer May 2008 Licensed in India (Feb 2009). WHO prequalified Sep Because NRA of Vietnam not recognized by WHO at that time, morcvax could not be WHO approved and made available on global PH market IVI Tech transferred the vaccine to Shantha in 2008 for production in India Shanchol
7 Shanchol Trial: Phase III Study of the Reformulated Bivalent Killed Oral Cholera Vaccine in Kolkata (NICED/IVI) To assess the protection of a two-dose regimen of the reformulated oral killed WC cholera vaccine against episodes of cholera severe enough to require medical treatment A cluster-randomised, double-blind, placebo-controlled trial Cholera endemic urban slums of Kolkata, India 65,000 subjects (3,478 clusters) Age > 1 year (excluding pregnant women) Two doses, 14 days apart 7
8 OCV Safety: Shanchol Common Adverse Events
9 Protective Efficacy during Three Years of Follow-up in Per Protocol Analyses, by Year of Follow-up Year of Follow-up First Year Second Year Third year Vaccine Placebo Vaccine Placebo Vaccine Placebo n=31,93 2 n=34,96 8 N=30,53 2 N=33,46 6 n=28,976 n=31,677 Cholera Episodes Incidence (per 100,000 persondays) Protective Efficacy (95% CI lower boundary) Adjusted analysis 41% (-13%) 76%** (52%) 65%** (44%) *p<.05; **p<.01
10 5 Year Protective Efficacy (per protocol) Overall occurrence of cholera episodes, and cumulative protective efficacy of the OCV during the 5 years of followup Vaccine Placebo Crude protective efficacy(pe) N Cases N Cases (95% CI; p-value) Adjusted protective efficac y (95%CI; p-value) 31, , % (53-75%; < 001) 65% (52-74%; < 001)* Age stratified point estimates of efficacy were not statistically different between age groups, however, suggest reduced efficacy for under 5yo More cases were prevented in children under 5 than other age groups
11 Herd Effect: Re-analysis of the Bangladesh Field Trial of Killed, Oral Cholera Vaccines (Ali, Lancet, 2005) A re-analysis of the 1985 trial of killed whole cellbased oral cholera vaccines found: -- Non-vaccinees were protected against cholera if they lived in neighborhoods with high levels of vaccine coverage -- Vaccinated persons had higher levels of protection if they lived in highly vaccinated neighborhoods
12
13 Cholera Vaccine (Inactivated Whole Cell Oral Cholera vaccine) IVI Technology Transfers Company Partnership Stage of development Vabiotech (Vietnam) IVI re-formulated, redeveloped proc ess to meet WHO standards. Licensed in Vietnam (morcvax ) Shantha (India) Technology transfer May 2008 Licensed in India (Feb 2009). WHO prequalified Sep OCV should be used in endemic areas and should be considered f or use in areas at risk for outbreaks in conjunction with other prevention and control strategies
14 IVI engaged Eubiologics in a tech transfer Euvichol Development Strategy -Tech Transfer of identical process and materials -Conduct comparative biophysical and quality analysis -Conduct clinical trials to evaluate safety and immunogenicity (non- inferiority) compared to Shanchol
15 Euvichol Each oral dose of 1.5mL contains Vaccine Strain V. cholera 01 Inaba E1 Tor strain Phil 6973 formaldehyde killed V. cholerae O1 Ogawa classical strain Cair o 50 heat killed V. cholerae O1 Ogawa classical strain strai n Cairo 60 formaldehyde killed V. cholera O1 Inaba classical strain Cairo 4 8 heat killed V. cholerae O139 strain 4260B formaldehy de killed Thiomersal B.P. Buffer VVM 13 Quantity 600 Elisa Units( EU) of lipopolys accharide(lps) 300 EU of LPS 300 EU of LPS 300 EU of LPS 600 EU of LPS Not more than 0.02%(w/v) q.s to 1.5mL Each oral dose of 1.5mL contains Description Composition i n 1.5ml V. Cholerae O1 and O139 bivalent inactivated v accine Composition VVM VVM 30 V. Cholerae O1 inaba Cairo 48, Heat inacti vated V. Cholerae O1 Phil 6973 El Tor, Formalin inactivated V. Cholerae O1 Ogawa Cairo 50, Formalin inactivated Qua ntity 300 L.E.U* 600 L.E.U 300 L.E.U V. Cholerae O1 Cairo 50, Heat inactivated 300 L.E.U V. Cholerae O B, Formalin inactivated Phosphate buffered saline(ph7.3) 600 L.E.U 20m M Thimerosal 0.15 mg
16 Cholera Vaccine (Inactivated Whole Cell Oral Cholera vaccine) IVI Technology Transfers Company Partnership Stage of development Vabiotech (Vietnam) morcvax IVI re-formulated, redevelope d process to meet WHO stand ards. Licensed in Vietnam (morcvax ) Shantha (India) Shanchol Technology transfer May 2008 Licensed in India (Feb 2009). WHO prequalified Sep Eubiologics (Korea) Euvichol Technology transfer Korean export license 2014 WHO prequalified Dec 2015 All OCV Technology Transfer activities funded by the Bill and Melinda Gates Foundation
17 Euvichol (Eubiologics, Korea) 100L Formulation Market authorization from Korean FDA Jan 2015 WHO PQ obtained Dec 2015 Euvichol 600L/no-thimerosal variation capacity up to 25M doses/year Bridging trial (100L to 600L) in the Philippines May 5, participants : Age 1-40y Variation Approval from WHO received Sept 2016 Eubiologics expected to be the major supplier of OCV to the WHO stockpile (Funder: BMGF)
18 Euvichol-P Thermostability: currently VVM 30 (30 days at 37C) Easier administration Lower production cost Lower cost of delivery 18
19 Cholera Vaccine (Inactivated Whole Cell Oral Cholera vaccine) IVI Technology Transfers Company Partnership Stage of development Vabiotech (Vietnam) morcvax IVI re-formulated, redevelope d process to meet WHO stand ards. Licensed in Vietnam (morcvax ) Shantha (India) Shanchol Technology transfer May 2008 Licensed in India (Feb 2009). WHO prequalified Sep Eubiologics (Korea) Euvichol Technology transfer Korean export license 2014 WHO prequalified Dec 2015 Incepta (Bangladesh ) Cholvax Technology transfer May 2014 IVI conducting clinical trials in Bangla desh, license in Bangladesh expected 2017/18. All OCV Technology Transfer activities funded by the Bill and Melinda Gates Foundation
20 Cholvax (Incepta, Bangladesh) : Supply OCV Tech Transfer to Incepta 2014 GMP Support (ongoing) Clinical Trial: Non-inferiority to Shanchol (icddr,b) Safety, Immunogenicity, lot-to-lot consistency 2,052 subjects (3 age cohorts) Oct Nov Dec Jan Feb Mar Apr May Jun Jul Aug Sep Oct Nov Dec Jan Feb Mar Apr Project start Regulatory Approval Adult Cohort 6-17yo Cohort 1-5yo Cohort Study Report Challenges to achieve WHO PQ Recognition of NRA (~2020) (Funder: BMGF)
21 Single Dose Study (icddr,b): Flexibility An individually randomized, placebo controlled trial of a single dose of Shanchol in an endemic setting 205,000 subjects enrolled Primary Objective: Protective efficacy of a single dose of Shanchol during initial 6 months following dosing Secondary Objective: Protective efficacy of a single dose of Shanchol at 12, 18, and 24 months following dosing 6 Month vaccine protective efficacy (PE) 40% (95% CI lower-bound (LB)=16%; P= 006) against all cholera cases 63% (95% CI=24%-82%; P=.007) against severely dehydrating cholera cases Efficacy only in >5 years of age 2 Year follow-up being analyzed now (Funder: BMGF)
22 Vaccination campaigns 2015/16 Demonstrate feasibility, cost effectiveness in different settings Year Location/Type Target # Coverage Outcomes 2016 /17 Nepal Pre-emptive 25,000 90% M&E In progress Expect Cost of Delivery and assessment of Choltool 2015 Nsanje, Malawi Reactive 2015 Shashemene Ethiopia 160,000 1 st 98% 2 nd 68% ~62,000 1 st 76% 2 nd 65% Acceptability, feasibility, Effectiveness (on going) Delivery and Cost of Illness (COI) Cost-Effectiveness Analysis Acceptability Feasibility 2015 Newakot and D hading, Nepal Pre-emptive 10,000 1 st 105% 2 nd 96% Feasibility of delivering OCV in earthquake aff ected districts (during monsoon season) using government infrastructure
23 Expanding Use of OCV: Malawi Campaign February 2015 : 1 st cholera case lab confirmed in Nsanje District 4 March 2015 : 72 cases, 2 deaths 50,000 person pre-emptive campaign became a 160,000 person reactive effort (collaboration with WHO, MoH) Camps hosting the internally displaced populations and surrounding villages 320,000 doses 110,000 redirected from planned pre-emptive campaign 210,000 dispatched via the ICG Stockpile Coverage : 1 st dose 98%, 2 nd dose 68%
24 Expanding Use of OCV: Malawi Campaign 2015 Cholera Surveillance in Malawi (CSIMA) Goal: To determine the 2-year protective effectiveness of OCV delivered through a reactive campaign in Nsanje District and increase the capacity for diarrheal surveillance in Nsanje and Chikwawa Districts -Vaccine Effectiveness (VE): case-control study design -Vaccination campaign delivery costs (Choltool) -Cost of illness: public and private expenditures for treatment and productivity losses associated with cholera -Cost-effectiveness -Capacity building and strengthening surveillance system: support of Nsanje/Chikwawa laboratories (Funder: BMGF)
25 Rotary Nepal Project: Cholera Prevention and Control (CCPC) in Nepal Preemptive Campaign in rural hot spot in Nepal Banke District, (Terai plain ): 25,000 target population 2 doses of Euvichol Funders: Rotary International, IVI Korean Support Committee Partners: Outcomes: Rotary Korea and Rotary Nagarjun Nepal JHUSPH-M&E GoN, MOH (EDCD) District Public Health Office Feasibility Assessment of Choltool comparison of the prediction of campaign costs with actual costs Vaccination: Dec 2016 Jan 2017 M&E: ongoing (~90% coverage)
26 Thank You IVI website Like us Follow us
27 Signatories to IVI s Establishment Agreement
28 Global Footprint 28
29 IVI Cholera Vaccine Program Strategy Development and Delivery Strategic Goals OCV Supply Program Objectives Increasing the availability of OCV Key Activities Partnership with manufacturers for TT, GMP, GCP, Clinical Development Two OCVs licensed and WHO-PQed One more OCV (Cholvax) in development Easier OCV Delivery Flexibility of Use Alternative dosing schedule(14 vs 28) Efficacy of a Single Dose of OCV OCV Use & Introduction Expand the use of OCV and gather evidence for introduction Vaccination Campaigns Feasibility, Safety, Effectiveness of OCV Delivery Costs and Cost of Illness Cost-Effectiveness Analysis
30 Comparison of Reformulated vs Vietnamese Oral Killed WC Vaccines Strain Vietnamese Vaccine Reformulated Vaccine Formalin-Killed El Tor Inaba (Phil 6973) Heat-Killed Classical Ogawa (Cairo 50) Formalin-Killed Classical Inaba (569B) Formalin-Killed Classical Ogawa (Cairo 50) Heat-Killed Classical Inaba (Cairo 48) 0139 (4260B) 5 x cells 600 EU LPS 2.5 x cells 300 EU LPS 2.5 x cells EU LPS EU LPS 5 x cells 600 EU LPS
31 WHO pre-qualified and available OCVs Dukoral Killed whole cell vaccine + B (binding) subunit of cholera toxin Requires buffer ( ml) 2 doses for age>5 yrs. and 3 doses for age 2-5 yrs. Vaccine efficacy of 60% sustained over 2 years High vaccine price, mainly for travelers Monovalent (O1) Shanchol Killed whole cell vaccine (no cholera toxin subunit) Buffer is not required 2 doses for all age groups (1+ years) Efficacy of 66% for 3 years Low-cost Bivalent (both O1 and O139)
32 WHO pre-qualified and available OCVs Dukoral Killed whole cell vaccine + B (binding) subunit of cholera toxin Requires buffer ( ml) 2 doses for age>5 yrs. and 3 doses for age 2-5 yrs. Vaccine efficacy of 60% sustained over 2 years High vaccine price, mainly for travelers Monovalent (O1) Shanchol Killed whole cell vaccine (no cholera toxin subunit) Buffer is not required 2 doses for all age groups (1+ years) Efficacy of 66% for 5 years Low-cost Bivalent (both O1 and O139)
33 IVI s Cholera Program Activities Overview (Supply, Flexibility of Use, Introduction) VABIOTECH SHANTHA Tech Transfer (PH 4) study Bridging (PH 4) study IVI Shanchol PH 3 Kolkata Bridging (PH 4) India Dosing interval study India Single dose Study Bangladesh Bridging (PH 4) Ethiopia OCV Pilot Introduction Orissa Vaccination Malawi Ethiopia Reactive campaign Malawi, Nepal EUBIOLOGICS Tech Transfer Euvichol Toxicity & PH 1 by EuB Euvichol Bridging trial Vaccination Nepal INCEPTA Tech Transfer 2014 Toxicity study Cholvax Cholvax licensure trial CHOVI investment case DOVE
Cholera vaccines: an update. Dipika Sur MD Consultant Translational Health Science and Technology Institute, INDIA Former Scientist 'G' ICMR
 Cholera vaccines: an update Dipika Sur MD Consultant Translational Health Science and Technology Institute, INDIA Former Scientist 'G' ICMR Cholera is caused by poverty, lack of sanitation and clean water
Cholera vaccines: an update Dipika Sur MD Consultant Translational Health Science and Technology Institute, INDIA Former Scientist 'G' ICMR Cholera is caused by poverty, lack of sanitation and clean water
5th Meeting of the Initiative against Diarrheal & Enteric diseases in Asia IDEA
 5th Meeting of the Initiative against Diarrheal & Enteric diseases in Asia IDEA Firdausi Qadri icddr,b Country Situation Update: Bangladesh Hanoi, Vietnam 7th March 2017 1 [Insert presentation title] 2
5th Meeting of the Initiative against Diarrheal & Enteric diseases in Asia IDEA Firdausi Qadri icddr,b Country Situation Update: Bangladesh Hanoi, Vietnam 7th March 2017 1 [Insert presentation title] 2
Recent WHO Data (2002)
 Recent WHO Data (2002) 142,311 cases and 4,564 deaths, from 52 countries 97% of cases reported from Africa These represent gross underestimates: -- More realistic estimates : 5.5 million cases; 100,000
Recent WHO Data (2002) 142,311 cases and 4,564 deaths, from 52 countries 97% of cases reported from Africa These represent gross underestimates: -- More realistic estimates : 5.5 million cases; 100,000
Update on the Use of Oral Cholera Vaccines (OCV) and Integration with WaSH: Challenges and Opportunities
 Update on the Use of Oral Cholera Vaccines (OCV) and Integration with WaSH: Challenges and Opportunities Kashmira Date U.S. Centers for Disease Control and Prevention (CDC) Global Taskforce on Cholera
Update on the Use of Oral Cholera Vaccines (OCV) and Integration with WaSH: Challenges and Opportunities Kashmira Date U.S. Centers for Disease Control and Prevention (CDC) Global Taskforce on Cholera
RIGHT (Research Investment for Global Health Technology, Korean GHIT) November 2017
 RIGHT (Research Investment for Global Health Technology, Korean GHIT) November 2017 ROK intends to create a new public-private partnership modeled after successful Japanese GHIT fund Research Investment
RIGHT (Research Investment for Global Health Technology, Korean GHIT) November 2017 ROK intends to create a new public-private partnership modeled after successful Japanese GHIT fund Research Investment
Technical Note. November 2016
 Technical Note Evidence of the risks and benefits of vaccinating pregnant women with WHO pre-qualified cholera vaccines during mass campaigns November 2016 Background Three oral cholera vaccines (OCVs)
Technical Note Evidence of the risks and benefits of vaccinating pregnant women with WHO pre-qualified cholera vaccines during mass campaigns November 2016 Background Three oral cholera vaccines (OCVs)
Oral Killed Whole Cell Vaccines: review of available evidence of vaccine effectiveness and safety across different settings and age groups
 Oral Killed Whole Cell Vaccines: review of available evidence of vaccine effectiveness and safety across different settings and age groups Francisco J. Luquero On behalf of the OCV- SAGE working group:
Oral Killed Whole Cell Vaccines: review of available evidence of vaccine effectiveness and safety across different settings and age groups Francisco J. Luquero On behalf of the OCV- SAGE working group:
IVI STRATEGY ARTICULATION. October 12, 2015
 IVI STRATEGY ARTICULATION October 12, 2015 Overall messages for today Our Vision and Mission are as relevant today as they have ever been We need to focus on the diseases and activities where we can have
IVI STRATEGY ARTICULATION October 12, 2015 Overall messages for today Our Vision and Mission are as relevant today as they have ever been We need to focus on the diseases and activities where we can have
Technical Note. 13 January 2016
 Technical Note Evidence of the risks and benefits of vaccinating pregnant women with WHO pre-qualified cholera vaccines during mass campaigns 13 January 2016 Background Two oral cholera vaccines (OCVs)
Technical Note Evidence of the risks and benefits of vaccinating pregnant women with WHO pre-qualified cholera vaccines during mass campaigns 13 January 2016 Background Two oral cholera vaccines (OCVs)
Modeling the effectiveness of mass cholera vaccination in Bangladesh
 Modeling the effectiveness of mass cholera vaccination in Bangladesh Dennis Chao April 20, 2016 1 / 20 Vibrio cholerae in Bangladesh Ali et al. Updated Global Burden of Cholera in Endemic Countries. PLoS
Modeling the effectiveness of mass cholera vaccination in Bangladesh Dennis Chao April 20, 2016 1 / 20 Vibrio cholerae in Bangladesh Ali et al. Updated Global Burden of Cholera in Endemic Countries. PLoS
Updates on Typhoid Conjugate vaccine Development: IVI
 Updates on Typhoid Conjugate vaccine Development: IVI Sushant Sahastrabuddhe 8 th International Conference on Typhoid Fever and Other Invasive Salmonelloses 2 nd March 2013 Outline of the presentation
Updates on Typhoid Conjugate vaccine Development: IVI Sushant Sahastrabuddhe 8 th International Conference on Typhoid Fever and Other Invasive Salmonelloses 2 nd March 2013 Outline of the presentation
Developing a novel Group B Streptococcus (GBS) Vaccine. Patrick Tippoo
 Developing a novel Group B Streptococcus (GBS) Vaccine Patrick Tippoo Presentation 1. Overview of Biovac 2. GBS Project Overview 3. African Significance Biovac Overview A Centre of Excellence rooted in
Developing a novel Group B Streptococcus (GBS) Vaccine Patrick Tippoo Presentation 1. Overview of Biovac 2. GBS Project Overview 3. African Significance Biovac Overview A Centre of Excellence rooted in
Global deployment of Oral Cholera Vaccine (OCV) Dipika Sur MD Consultant THSTI, India
 Global deployment of Oral Cholera Vaccine (OCV) Dipika Sur MD Consultant THSTI, India Stockpiling for OCV Global stockpile of oral cholera vaccine (OCV) created since 2013 as additional tool to help control
Global deployment of Oral Cholera Vaccine (OCV) Dipika Sur MD Consultant THSTI, India Stockpiling for OCV Global stockpile of oral cholera vaccine (OCV) created since 2013 as additional tool to help control
Towards a Sustainable Global Infrastructure for Medical Countermeasures
 Towards a Sustainable Global Infrastructure for Medical Countermeasures Institute of Medicine The Public Health Emergency Medical Countermeasures Enterprise: Innovative Strategies to Enhance Products from
Towards a Sustainable Global Infrastructure for Medical Countermeasures Institute of Medicine The Public Health Emergency Medical Countermeasures Enterprise: Innovative Strategies to Enhance Products from
Background Paper on Whole-Cell, Killed, Oral Cholera Vaccines
 Background Paper on Whole-Cell, Killed, Oral Cholera Vaccines Prepared by the SAGE Working Group on Oral Cholera Vaccines, the World Health Organization (WHO) Secretariat, and the Centers for Disease Control
Background Paper on Whole-Cell, Killed, Oral Cholera Vaccines Prepared by the SAGE Working Group on Oral Cholera Vaccines, the World Health Organization (WHO) Secretariat, and the Centers for Disease Control
COUNTRY NAME: BANGLADESH. GTFCC 5-6 Dec 2018
 COUNTRY NAME: BANGLADESH GTFCC 5-6 Dec 2018 Ending Cholera for Bangladesh- one of the 20 countries that hopefully will eliminate the disease The overall objective of the renewed strategy is to reduce the
COUNTRY NAME: BANGLADESH GTFCC 5-6 Dec 2018 Ending Cholera for Bangladesh- one of the 20 countries that hopefully will eliminate the disease The overall objective of the renewed strategy is to reduce the
No
 No.13 / - http://www.who.int/wer... : :...... /.. : http://www.who.int/immunization/documents/positionpapers/en/index.html.. 1 O1). (O139......... /, /, /,. /, /, ).O1.(...... /... - O1 - -. Deen JL et
No.13 / - http://www.who.int/wer... : :...... /.. : http://www.who.int/immunization/documents/positionpapers/en/index.html.. 1 O1). (O139......... /, /, /,. /, /, ).O1.(...... /... - O1 - -. Deen JL et
Prospective Models of Vaccine Security Collaborations in Research and Development
 Prospective Models of Vaccine Security Collaborations in Research and Development Georges Thiry, PhD IVI (International Vaccine Institute) Phuket, Oct 01, 2014 Content R&D Examples of collaborations from
Prospective Models of Vaccine Security Collaborations in Research and Development Georges Thiry, PhD IVI (International Vaccine Institute) Phuket, Oct 01, 2014 Content R&D Examples of collaborations from
WHO Consultation on oral cholera vaccine (OCV) stockpile strategic framework: potential objectives and possible policy options
 WHO/IVB/12.05 ORIGINAL: ENGLISH WHO Consultation on oral cholera vaccine (OCV) stockpile strategic framework: potential objectives and possible policy options 18-20 September 2011 Geneva, Switzerland Immunization,
WHO/IVB/12.05 ORIGINAL: ENGLISH WHO Consultation on oral cholera vaccine (OCV) stockpile strategic framework: potential objectives and possible policy options 18-20 September 2011 Geneva, Switzerland Immunization,
History, implementation and impact of MenA conjugate on disease burden in Africa
 History, implementation and impact of MenA conjugate on disease burden in Africa First Regional Meningococcal Symposium, 19-20 March 2012 Buenos Aires, Argentina Co-hosted by the Sabin Vaccine Institute
History, implementation and impact of MenA conjugate on disease burden in Africa First Regional Meningococcal Symposium, 19-20 March 2012 Buenos Aires, Argentina Co-hosted by the Sabin Vaccine Institute
GSK s Candidate Influenza A (H5N1) Virus Monovalent Vaccine
 GSK s Candidate Influenza A (H5N1) Virus Monovalent Vaccine Regulatory Pathway for Licensure VRBPAC, February 29, 2012 Katalin Abraham, Director, US Regulatory Affairs GSK Biologicals GSK s Influenza Vaccines
GSK s Candidate Influenza A (H5N1) Virus Monovalent Vaccine Regulatory Pathway for Licensure VRBPAC, February 29, 2012 Katalin Abraham, Director, US Regulatory Affairs GSK Biologicals GSK s Influenza Vaccines
GAVI s Financing for Pneumococcal Vaccines, including the Advance Market Commitment
 GAVI s Financing for Pneumococcal Vaccines, including the Advance Market Commitment Tania Cernuschi Third Regional Pneumococcal Symposium, Istanbul, 13th - 14th February 2008 1 The GAVI Alliance Public-private
GAVI s Financing for Pneumococcal Vaccines, including the Advance Market Commitment Tania Cernuschi Third Regional Pneumococcal Symposium, Istanbul, 13th - 14th February 2008 1 The GAVI Alliance Public-private
Determining the Feasibility of an Oral Cholera Vaccination (OCV) Campaign
 Determining the Feasibility of an Oral Cholera Vaccination (OCV) Campaign Updated November, 2016 Examining the feasibility of a vaccination campaign is critical for making decisions about whether or not
Determining the Feasibility of an Oral Cholera Vaccination (OCV) Campaign Updated November, 2016 Examining the feasibility of a vaccination campaign is critical for making decisions about whether or not
UNICEF Procurement Advancements DCVMN Annual Meeting Hanoi, Vietnam October 2013
 UNICEF Procurement Advancements DCVMN Annual Meeting Hanoi, Vietnam October 2013 Presentation overview Scope of UNICEF procurement Overview of UNICEF procurement Vaccine market updates UNICEF procures
UNICEF Procurement Advancements DCVMN Annual Meeting Hanoi, Vietnam October 2013 Presentation overview Scope of UNICEF procurement Overview of UNICEF procurement Vaccine market updates UNICEF procures
Measles Containing Vaccines. UNICEF Supply Division Industry Consultation Meeting January 2012
 Measles Containing Vaccines UNICEF Supply Division Industry Consultation Meeting 25-26 January 2012 Content Measles monovalent vaccine: Programme Updates and Procurement Overview Measles and Rubella vaccine:
Measles Containing Vaccines UNICEF Supply Division Industry Consultation Meeting 25-26 January 2012 Content Measles monovalent vaccine: Programme Updates and Procurement Overview Measles and Rubella vaccine:
Experiences with pre-emptive cholera vaccination in emergency situations
 Experiences with pre-emptive cholera vaccination in emergency situations Meeting on integrating vaccines into global cholera control efforts Les Pensières, Veyrier du Lac, France 14-17.04.2009 Dr Claire-Lise
Experiences with pre-emptive cholera vaccination in emergency situations Meeting on integrating vaccines into global cholera control efforts Les Pensières, Veyrier du Lac, France 14-17.04.2009 Dr Claire-Lise
BIOST/STAT 578 A Statistical Methods in Infectious Diseases Lecture 16 February 26, Cholera: ecological determinants and vaccination
 BIOST/STAT 578 A Statistical Methods in Infectious Diseases Lecture 16 February 26, 2009 Cholera: ecological determinants and vaccination Latest big epidemic in Zimbabwe Support International Vaccine
BIOST/STAT 578 A Statistical Methods in Infectious Diseases Lecture 16 February 26, 2009 Cholera: ecological determinants and vaccination Latest big epidemic in Zimbabwe Support International Vaccine
THE SITUATION OF YELLOW FEVER IN THE AFRICAN REGION: THE PLAN TO END YF EPIDEMICS IN 2026
 THE SITUATION OF YELLOW FEVER IN THE AFRICAN REGION: THE PLAN TO END YF EPIDEMICS IN 2026 Dr Zabulon Yoti WHO AFRO Technical Coordinator for Health Emergencies 1 About 100 acute public health events annually
THE SITUATION OF YELLOW FEVER IN THE AFRICAN REGION: THE PLAN TO END YF EPIDEMICS IN 2026 Dr Zabulon Yoti WHO AFRO Technical Coordinator for Health Emergencies 1 About 100 acute public health events annually
Progress and Challenges of Enteric Vaccines. Dr. Tajul Islam A Bari icddr,b Bangladesh
 Progress and Challenges of Enteric Vaccines Dr. Tajul Islam A Bari icddr,b Bangladesh New enteric vaccines in use Cholera Rotavirus Typhoid Cholera- History During the 19th century, cholera spread across
Progress and Challenges of Enteric Vaccines Dr. Tajul Islam A Bari icddr,b Bangladesh New enteric vaccines in use Cholera Rotavirus Typhoid Cholera- History During the 19th century, cholera spread across
Herd Protective Effects of Vaccines. John Clemens icddr,b, Dhaka, Bangladesh
 Herd Protective Effects of Vaccines John Clemens icddr,b, Dhaka, Bangladesh Promising Vaccine Candidate Phase I. Safe and Immunogenic in Healthy Adults? Yes No Phase II. Safe and Immunogenic in the Target
Herd Protective Effects of Vaccines John Clemens icddr,b, Dhaka, Bangladesh Promising Vaccine Candidate Phase I. Safe and Immunogenic in Healthy Adults? Yes No Phase II. Safe and Immunogenic in the Target
Challenges of use of cholera vaccines in Haiti and the Americas
 http://www.paho.org Challenges of use of cholera vaccines in Haiti and the Americas Dr. Cuauhtémoc Ruiz Matus Comprehensive Family Immunization Program Background Topics Consultations at PAHO Challenges
http://www.paho.org Challenges of use of cholera vaccines in Haiti and the Americas Dr. Cuauhtémoc Ruiz Matus Comprehensive Family Immunization Program Background Topics Consultations at PAHO Challenges
Overview of Measlescontaining. through UNICEF. Overview of [VACCINE] through UNICEF
![Overview of Measlescontaining. through UNICEF. Overview of [VACCINE] through UNICEF Overview of Measlescontaining. through UNICEF. Overview of [VACCINE] through UNICEF](/thumbs/80/80697958.jpg) Overview of Measlescontaining vaccines through UNICEF Overview of [VACCINE] through UNICEF Children wait to be immunized against measles and rubella at the Losikito primary school in Tanzania. Source:
Overview of Measlescontaining vaccines through UNICEF Overview of [VACCINE] through UNICEF Children wait to be immunized against measles and rubella at the Losikito primary school in Tanzania. Source:
Epidemiological Situation of Cholera and its Response in Nepal
 Epidemiological Situation of Cholera and its Response in Nepal Dr Basu Dev Pandey Deputy Director General Department of Health Services Ministry of Health, Nepal Email: drbasupandey@gmail.com Population:
Epidemiological Situation of Cholera and its Response in Nepal Dr Basu Dev Pandey Deputy Director General Department of Health Services Ministry of Health, Nepal Email: drbasupandey@gmail.com Population:
Summaries of Complementary Presentations on Agenda Item:
 Summaries of Complementary Presentations on Agenda Item: WHO s Role in Supporting Emerging Vaccine Manufacturers To inform the SAGE discussion on this agenda item, four presentations have been solicited
Summaries of Complementary Presentations on Agenda Item: WHO s Role in Supporting Emerging Vaccine Manufacturers To inform the SAGE discussion on this agenda item, four presentations have been solicited
VACCINE MARKETS OVERVIEW SESSION
 VACCINE MARKETS OVERVIEW SESSION Robyn Iqbal Program Officer, Vaccine Delivery Market Dynamics UNICEF SD Manufacturers Meeting Copenhagen October 8, 2014 DRAFT Bill & Melinda Gates Foundation DISCUSSION
VACCINE MARKETS OVERVIEW SESSION Robyn Iqbal Program Officer, Vaccine Delivery Market Dynamics UNICEF SD Manufacturers Meeting Copenhagen October 8, 2014 DRAFT Bill & Melinda Gates Foundation DISCUSSION
Hilleman Laboratories
 Hilleman Laboratories If I had to name a person who has done more for the benefit of human health, with less recognition than anyone else, it would be Maurice Hilleman. Maurice should be recognized as
Hilleman Laboratories If I had to name a person who has done more for the benefit of human health, with less recognition than anyone else, it would be Maurice Hilleman. Maurice should be recognized as
NVI Experience and Concept of Central Technology Hub
 NVI Experience and Concept of Central Technology Hub Jan Hendriks Netherlands Vaccine Institute (NVI) MSF/Oxfam Consultation improving access and stimulating vaccine development for use in resource pour
NVI Experience and Concept of Central Technology Hub Jan Hendriks Netherlands Vaccine Institute (NVI) MSF/Oxfam Consultation improving access and stimulating vaccine development for use in resource pour
FMD Control Initiatives in Bangladesh
 FMD Control Initiatives in Bangladesh Dr. Md. Mohsin Ali Dr. Md. Ainul Haque Department of Livestock Services, Bangladesh Country Profile In Short Bangladesh is a Republic of South Asia It is bordered
FMD Control Initiatives in Bangladesh Dr. Md. Mohsin Ali Dr. Md. Ainul Haque Department of Livestock Services, Bangladesh Country Profile In Short Bangladesh is a Republic of South Asia It is bordered
Protecting people against known and emerging infectious diseases globally
 Protecting people against known and emerging infectious diseases globally 18 October 2017 SAGE Geneva By Mahima Datla President, DCVMN 1 Agenda Introduction to DCVMN DCVMN s contributions Suggested areas
Protecting people against known and emerging infectious diseases globally 18 October 2017 SAGE Geneva By Mahima Datla President, DCVMN 1 Agenda Introduction to DCVMN DCVMN s contributions Suggested areas
WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES
 WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES Immunization and vaccine development in the last decade Since the Millennium Summit in 2000, immunization substantial international
WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES Immunization and vaccine development in the last decade Since the Millennium Summit in 2000, immunization substantial international
Herd Protection- Efficacy vs effectiveness The Importance of Cluster-Randomized Trials to assess Vaccine Herd Protection
 Herd Protection- Efficacy vs effectiveness The Importance of Cluster-Randomized Trials to assess Vaccine Herd Protection Anna Lena Lopez, MD, MPH University of the Philippines Manila- National Institutes
Herd Protection- Efficacy vs effectiveness The Importance of Cluster-Randomized Trials to assess Vaccine Herd Protection Anna Lena Lopez, MD, MPH University of the Philippines Manila- National Institutes
Vibriocidal Antibody Responses to a Bivalent Killed Whole-Cell Oral Cholera Vaccine in a Phase III Trial in Kolkata, India
 Vibriocidal Antibody Responses to a Bivalent Killed Whole-Cell Oral Cholera Vaccine in a Phase III Trial in Kolkata, India Suman Kanungo 1 *, Anna Lena Lopez 2, Mohammad Ali 3, Byomkesh Manna 1, Deok Ryon
Vibriocidal Antibody Responses to a Bivalent Killed Whole-Cell Oral Cholera Vaccine in a Phase III Trial in Kolkata, India Suman Kanungo 1 *, Anna Lena Lopez 2, Mohammad Ali 3, Byomkesh Manna 1, Deok Ryon
Establishing a cholera stockpile: What do we need? Alejandro Costa Epidemic Readiness and Intervention
 Establishing a cholera stockpile: What do we need? Alejandro Costa Epidemic Readiness and Intervention Outline 1. Experience Meningococcal vaccine stockpile Yellow Fever vaccine stockpile 2. Criteria for
Establishing a cholera stockpile: What do we need? Alejandro Costa Epidemic Readiness and Intervention Outline 1. Experience Meningococcal vaccine stockpile Yellow Fever vaccine stockpile 2. Criteria for
The Influenza Season Stephen L. Cochi, M.D., M.P.H. Acting Director National Immunization Program, CDC
 The Influenza Season 2003-04 Stephen L. Cochi, M.D., M.P.H. Acting Director National Immunization Program, CDC 2003-2004 Influenza Season Influenza activity onset earlier than usual Children appeared disproportionately
The Influenza Season 2003-04 Stephen L. Cochi, M.D., M.P.H. Acting Director National Immunization Program, CDC 2003-2004 Influenza Season Influenza activity onset earlier than usual Children appeared disproportionately
Thailand s avian influenza control and pandemic influenza preparedness. Supamit Chunsuttiwat Ministry of Public Health 12 July 2006
 Thailand s avian influenza control and pandemic influenza preparedness Supamit Chunsuttiwat Ministry of Public Health 12 July 2006 Avian influenza situation 2004-05 Human cases Poultry outbreaks 250 200
Thailand s avian influenza control and pandemic influenza preparedness Supamit Chunsuttiwat Ministry of Public Health 12 July 2006 Avian influenza situation 2004-05 Human cases Poultry outbreaks 250 200
GSK Medicine: Study No.: Title: Rationale: Study Period Objectives: Indication: Study Investigators/Centers: Research Methods: Data Source:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Total population 24,759,000. Live births (LB) 342,458. Children <1 year 337,950. Children <5 years 1,698,664. Children <15 years 5,233,093
 DPR Korea 4 Immunization system highlights There is a comprehensive multiyear plan (cmyp) for immunization covering -5. A standing national technical advisory group on immunization (NTAGI) with formal
DPR Korea 4 Immunization system highlights There is a comprehensive multiyear plan (cmyp) for immunization covering -5. A standing national technical advisory group on immunization (NTAGI) with formal
Vaccines against Rotavirus & Norovirus. Umesh D. Parashar CDC, Atlanta, GA
 TM Vaccines against Rotavirus & Norovirus Umesh D. Parashar CDC, Atlanta, GA 1 Rotavirus is the Leading Cause Of Severe Diarrhea in Children
TM Vaccines against Rotavirus & Norovirus Umesh D. Parashar CDC, Atlanta, GA 1 Rotavirus is the Leading Cause Of Severe Diarrhea in Children
Cholera. Report by the Secretariat
 EXECUTIVE BOARD EB128/13 128th Session 9 December 2010 Provisional agenda item 4.10 Cholera Report by the Secretariat 1. In May 2010, the Executive Board at its 127th session considered a report on cholera
EXECUTIVE BOARD EB128/13 128th Session 9 December 2010 Provisional agenda item 4.10 Cholera Report by the Secretariat 1. In May 2010, the Executive Board at its 127th session considered a report on cholera
Evidence review on the immunogenicity, efficacy/effectiveness and safety of typhoid conjugate vaccines
 Evidence review on the immunogenicity, efficacy/effectiveness and safety of typhoid conjugate vaccines SAGE meeting, Geneva, 17-19 October, 2017 Myron M. (Mike) Levine, MD, DTPH Grollman Distinguished
Evidence review on the immunogenicity, efficacy/effectiveness and safety of typhoid conjugate vaccines SAGE meeting, Geneva, 17-19 October, 2017 Myron M. (Mike) Levine, MD, DTPH Grollman Distinguished
Strengthening Veterinary Services in Asia
 Strengthening Veterinary Services in Asia 4 th Steering Committee Highly Pathogenic Emerging and Re-Emerging Diseases (HPED) Programme Session 3 Progress on the OIE component Tokyo (Japan) 16 July 2013
Strengthening Veterinary Services in Asia 4 th Steering Committee Highly Pathogenic Emerging and Re-Emerging Diseases (HPED) Programme Session 3 Progress on the OIE component Tokyo (Japan) 16 July 2013
COUNTRY NAME: BANGLADESH. 5 th Annual Meeting of the GTFCC June 2018
 COUNTRY NAME: BANGLADESH 5 th Annual Meeting of the GTFCC 13-14 June 2018 OBJECTIVES AND TIMELINES Objectives Alignment of National Program on Diarrheal Diseases Prevention, Management & Control with the
COUNTRY NAME: BANGLADESH 5 th Annual Meeting of the GTFCC 13-14 June 2018 OBJECTIVES AND TIMELINES Objectives Alignment of National Program on Diarrheal Diseases Prevention, Management & Control with the
MARKET NEWS for pig meat
 MARKET NEWS for pig meat Market analysis 17 December 2018 Week 51 MARKET SITUATION Europe: Trading in legs and other cuts is at unchanged or slightly falling prices. UK: Prices are reported as stable.
MARKET NEWS for pig meat Market analysis 17 December 2018 Week 51 MARKET SITUATION Europe: Trading in legs and other cuts is at unchanged or slightly falling prices. UK: Prices are reported as stable.
The MenAfrivac Experience A Successful Approach. Dr Bernard FRITZELL BFL conseils France
 The MenAfrivac Experience A Successful Approach Dr Bernard FRITZELL BFL conseils France 1 2 1996 Ministers of Health and Interior from 16 African countries recognized epidemic meningitis as a high priority
The MenAfrivac Experience A Successful Approach Dr Bernard FRITZELL BFL conseils France 1 2 1996 Ministers of Health and Interior from 16 African countries recognized epidemic meningitis as a high priority
Controlled Human Infection Models and Enteric Vaccine Development
 Controlled Human Infection Models and Enteric Vaccine Development 2018 Global Vaccine and Immunization Research Forum March 20, 2018 Beth Kirkpatrick, MD Objectives: What are Controlled Human Infection/Challenge
Controlled Human Infection Models and Enteric Vaccine Development 2018 Global Vaccine and Immunization Research Forum March 20, 2018 Beth Kirkpatrick, MD Objectives: What are Controlled Human Infection/Challenge
Inaugural Keynote Lecture: Contributions of Gavi and DCVMN to the global vaccine markets
 Inaugural Keynote Lecture: Contributions of Gavi and DCVMN to the global vaccine markets Seth Berkley M.D, CEO, Seoul www.gavi.org 1 Gavi s mission, model and achievements to date Gavi s mission Saving
Inaugural Keynote Lecture: Contributions of Gavi and DCVMN to the global vaccine markets Seth Berkley M.D, CEO, Seoul www.gavi.org 1 Gavi s mission, model and achievements to date Gavi s mission Saving
Report to the. GAVI Alliance Board November 2013
 Report to the GAVI Alliance Board 21-22 November 2013 Subject: Report of: Authored by: Agenda item: Category: Consent Agenda: Opening of Funding Window for Japanese Encephalitis Hind Khatib-Othman, Managing
Report to the GAVI Alliance Board 21-22 November 2013 Subject: Report of: Authored by: Agenda item: Category: Consent Agenda: Opening of Funding Window for Japanese Encephalitis Hind Khatib-Othman, Managing
Oral Vaccines Against Cholera
 Clinical Infectious Diseases Advance Access published April 20, 2011 INVITED ARTICLE VACCINES Stanley Plotkin, Section Editor Oral Vaccines Against Cholera Sunheang Shin, Sachin N. Desai, Binod K. Sah,
Clinical Infectious Diseases Advance Access published April 20, 2011 INVITED ARTICLE VACCINES Stanley Plotkin, Section Editor Oral Vaccines Against Cholera Sunheang Shin, Sachin N. Desai, Binod K. Sah,
WHE Situation Report. June, 2018 Situation Report No M KEY FIGURES HIGHLIGHTS. There has been significant decrease in the
 WHE Situation Report 5.4 M In Need of Health Services KEY FIGURES 5.4M 2.6 M Displaced 4.3 M People Targeted 341 EWARN Sentinel Sites HIGHLIGHTS 5,582 Cholera Cases 49 WHO STAFF IN THE COUNTRY There has
WHE Situation Report 5.4 M In Need of Health Services KEY FIGURES 5.4M 2.6 M Displaced 4.3 M People Targeted 341 EWARN Sentinel Sites HIGHLIGHTS 5,582 Cholera Cases 49 WHO STAFF IN THE COUNTRY There has
Total population 1,212,110. Live births (LB) 43,924. Children <1 year 40,351. Children <5 years 192,340. Children <15 years 510,594
 Draft version for the SEAR-ITAG 5-9 June 5 All data provisional as of May 5 Timor Leste 4 Immunization system highlights There is a comprehensive multi-year plan (cmyp) for immunization covering 3-5. 8%
Draft version for the SEAR-ITAG 5-9 June 5 All data provisional as of May 5 Timor Leste 4 Immunization system highlights There is a comprehensive multi-year plan (cmyp) for immunization covering 3-5. 8%
Vaccine Introduction & Uptake Timing Benchmark Project
 Vaccine Introduction & Uptake Timing Benchmark Project Lois Privor-Dumm Managing Director Policy, Advocacy & Communications Team Lead International Vaccine Access Center Johns Hopkins Bloomberg School
Vaccine Introduction & Uptake Timing Benchmark Project Lois Privor-Dumm Managing Director Policy, Advocacy & Communications Team Lead International Vaccine Access Center Johns Hopkins Bloomberg School
CHOLERA UPDATES in Indonesia
 CHOLERA UPDATES in Indonesia Musal Kadim MD Gastrohepatology Division, Child Health Department, University of Indonesia Indonesian Pediatric Society Cholera epidemiology update History 7 cholera pandemics
CHOLERA UPDATES in Indonesia Musal Kadim MD Gastrohepatology Division, Child Health Department, University of Indonesia Indonesian Pediatric Society Cholera epidemiology update History 7 cholera pandemics
Meningitis Vaccine Procurement
 Meningitis Vaccine Procurement Meningitis belt countries in Africa Meningitis AC - Procurement overview Historically limited funding and supply. Only 1 manufacturer for many years. UNICEF has only procured
Meningitis Vaccine Procurement Meningitis belt countries in Africa Meningitis AC - Procurement overview Historically limited funding and supply. Only 1 manufacturer for many years. UNICEF has only procured
Vaccine prevention for existing and emerging viral threats
 Vaccine prevention for existing and emerging viral threats Viruses in May Blue Mountains 18 May 2018 www.ncirs.edu.au https://www.apprise.org.au/ Prof Kristine Macartney Director, NCIRS www.ncirs.usyd.edu.au
Vaccine prevention for existing and emerging viral threats Viruses in May Blue Mountains 18 May 2018 www.ncirs.edu.au https://www.apprise.org.au/ Prof Kristine Macartney Director, NCIRS www.ncirs.usyd.edu.au
Total population 1,265,308,000. Live births (LB) 27,016,000. Children <1 year 25,928,200. Children <5 years 23,818,000. Children <15 years 25,639,000
 India 4 Immunization system highlights There is a comprehensive multiyear plan (cmyp) for immunization covering 3-7. National technical advisory group on immunization (NTAGI) exists and formal written
India 4 Immunization system highlights There is a comprehensive multiyear plan (cmyp) for immunization covering 3-7. National technical advisory group on immunization (NTAGI) exists and formal written
Lopez, AL; Deen, J; Azman, AS; Luquero, FJ; Kanungo, S; Dutta, S; von Seidlein, L; Sack, DA
 MSF Field Research Immunogenicity and Protection from a Single Dose of Internationally available killed oral Cholera Vaccine: a systematic review and meta-analysis Authors Citation DOI Publisher Journal
MSF Field Research Immunogenicity and Protection from a Single Dose of Internationally available killed oral Cholera Vaccine: a systematic review and meta-analysis Authors Citation DOI Publisher Journal
OIE Situation Report for Highly Pathogenic Avian Influenza
 OIE Situation Report for Highly Pathogenic Avian Influenza Latest update: 28/02/2018 The epidemiology of avian influenza is complex. The virus constantly evolves and the behavior of each new subtype (and
OIE Situation Report for Highly Pathogenic Avian Influenza Latest update: 28/02/2018 The epidemiology of avian influenza is complex. The virus constantly evolves and the behavior of each new subtype (and
Overview of the Malaria Vaccine Implementation Programme (MVIP) Prof. Fred Were SAGE meeting 17 April, 2018
 Overview of the Malaria Vaccine Implementation Programme (MVIP) Prof. Fred Were SAGE meeting 17 April, 2018 1 Objectives Brief review Background EMA positive opinion and WHO recommendations Funding Description
Overview of the Malaria Vaccine Implementation Programme (MVIP) Prof. Fred Were SAGE meeting 17 April, 2018 1 Objectives Brief review Background EMA positive opinion and WHO recommendations Funding Description
PATH Influenza Vaccine Projects
 PATH Influenza Vaccine Projects Overview John W. Boslego, MD John Boslego Director, Vaccine Development Global Program March 25 th, 2014 Influenza Vaccine Project (IVP) at PATH IVP Goal: Advance the development
PATH Influenza Vaccine Projects Overview John W. Boslego, MD John Boslego Director, Vaccine Development Global Program March 25 th, 2014 Influenza Vaccine Project (IVP) at PATH IVP Goal: Advance the development
Bangladesh: How to move from data to implementation. K. Zaman, MBBS, PhD Senior Scientist and Epidemiologist icddr,b
 Bangladesh: How to move from data to implementation K. Zaman, MBBS, PhD Senior Scientist and Epidemiologist icddr,b Rotavirus in Bangladesh Estimated 2.4 million cases of rotavirus diarrhoea annually in
Bangladesh: How to move from data to implementation K. Zaman, MBBS, PhD Senior Scientist and Epidemiologist icddr,b Rotavirus in Bangladesh Estimated 2.4 million cases of rotavirus diarrhoea annually in
Eradicating Polio: Why and How? Dr Roland Sutter, World Health Organization 20 September 2011
 Eradicating Polio: Why and How? Dr Roland Sutter, World Health Organization 20 Why? Polio Crippled for life Primarily affects children We have a historic opportunity. Preventable with vaccine 1955: polio
Eradicating Polio: Why and How? Dr Roland Sutter, World Health Organization 20 Why? Polio Crippled for life Primarily affects children We have a historic opportunity. Preventable with vaccine 1955: polio
Cholera Clinical Guidelines. OAHPP Rounds January 11, 2011 Vanessa G. Allen MD FRCPC Medical Microbiologist
 Cholera Clinical Guidelines OAHPP Rounds January 11, 2011 Vanessa G. Allen MD FRCPC Medical Microbiologist Overview Cholera clinical guidelines Development and review process Content Reference for diagnosis,
Cholera Clinical Guidelines OAHPP Rounds January 11, 2011 Vanessa G. Allen MD FRCPC Medical Microbiologist Overview Cholera clinical guidelines Development and review process Content Reference for diagnosis,
Update on Implementation of NUV. Carsten Mantel WHO/FCH/IVB/EPI
 Update on Implementation of NUV Carsten Mantel WHO/FCH/IVB/EPI Introduction of New Vaccines The continuum of activity REGULATORY NORMS & STANDARDS VACCINE SUPPLY PROCUREMENT PRICING FINANCING SURVEILLANCE
Update on Implementation of NUV Carsten Mantel WHO/FCH/IVB/EPI Introduction of New Vaccines The continuum of activity REGULATORY NORMS & STANDARDS VACCINE SUPPLY PROCUREMENT PRICING FINANCING SURVEILLANCE
Mathematical modeling of cholera
 Mathematical modeling of cholera Dennis Chao Center for Statistics and Quantitative Infectious Diseases (CSQUID) Vaccine and Infectious Disease Division Fred Hutchinson Cancer Research Center 22 April,
Mathematical modeling of cholera Dennis Chao Center for Statistics and Quantitative Infectious Diseases (CSQUID) Vaccine and Infectious Disease Division Fred Hutchinson Cancer Research Center 22 April,
GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA
 GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA Dr Gounalan Pavade OIE Regional Expert Group Meeting for the Control of Avian Influenza in Asia Sapporo, Japan 3-5 October 2017 1 Avian influenza Outbreaks
GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA Dr Gounalan Pavade OIE Regional Expert Group Meeting for the Control of Avian Influenza in Asia Sapporo, Japan 3-5 October 2017 1 Avian influenza Outbreaks
Expedited procedure for evaluating pandemic influenza A (H1N1) 2009 vaccines
 Expedited procedure for evaluating pandemic influenza A (H1N1) 2009 vaccines Preamble On 11 June 2009, WHO declared an influenza pandemic caused by influenza A (H1N1) 2009 virus 1. On 13 July 2009, WHO
Expedited procedure for evaluating pandemic influenza A (H1N1) 2009 vaccines Preamble On 11 June 2009, WHO declared an influenza pandemic caused by influenza A (H1N1) 2009 virus 1. On 13 July 2009, WHO
POLIO ERADICATION AND POST-CERTIFICATION STRATEGY
 POLIO ERADICATION AND POST-CERTIFICATION STRATEGY PRE-BOARD MEETING 5 June 2018, Geneva Reach every child www.gavi.org Agenda Topic Presenter 1. Polio eradication update WHO 10 minutes 2. IPV supply, demand
POLIO ERADICATION AND POST-CERTIFICATION STRATEGY PRE-BOARD MEETING 5 June 2018, Geneva Reach every child www.gavi.org Agenda Topic Presenter 1. Polio eradication update WHO 10 minutes 2. IPV supply, demand
Total population 20,675,000. Live births (LB) 349,715. Children <1 year 346,253. Children <5 years 1,778,050. Children <15 years 5,210,100
 Sri Lanka 4 Immunization system highlights There is a comprehensive multi-year plan (cmyp) for immunization system strengthening covering -6. A national policy on immunization has been developed. A standing
Sri Lanka 4 Immunization system highlights There is a comprehensive multi-year plan (cmyp) for immunization system strengthening covering -6. A national policy on immunization has been developed. A standing
Adjuvant technology transfer hub
 Adjuvant technology transfer hub An example of transferring «enabling technologies» Nicolas Collin, DVM, PhD Vaccine Formulation Laboratory, University of Lausanne WH Collaborating Centre Workshop on Technology
Adjuvant technology transfer hub An example of transferring «enabling technologies» Nicolas Collin, DVM, PhD Vaccine Formulation Laboratory, University of Lausanne WH Collaborating Centre Workshop on Technology
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005
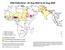 Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
Development of a Group A meningococcal conjugate vaccine for sub-saharan Africa: clinical trial results
 Development of a Group A meningococcal conjugate vaccine for sub-saharan Africa: clinical trial results Global Immunization Meeting 2009, United Nations HQ New York City, February 2009 MenA conjugate vaccine
Development of a Group A meningococcal conjugate vaccine for sub-saharan Africa: clinical trial results Global Immunization Meeting 2009, United Nations HQ New York City, February 2009 MenA conjugate vaccine
Maternal Immunization: Unique considerations of public health value of vaccines given to pregnant women
 Maternal Immunization: Unique considerations of public health value of vaccines given to pregnant women Estimating the full public health value of vaccines Kathleen M. Neuzil, MD, MPH Professor of Medicine
Maternal Immunization: Unique considerations of public health value of vaccines given to pregnant women Estimating the full public health value of vaccines Kathleen M. Neuzil, MD, MPH Professor of Medicine
OIE Situation Report for Avian Influenza
 OIE Situation Report for Avian Influenza Latest update: 25/01/2018 The epidemiology of avian influenza is complex. The virus constantly evolves and the behavior of each new subtype (and strains within
OIE Situation Report for Avian Influenza Latest update: 25/01/2018 The epidemiology of avian influenza is complex. The virus constantly evolves and the behavior of each new subtype (and strains within
Update on Progress and Challenges in Achieving Measles and Rubella Targets
 Update on Progress and Challenges in Achieving Measles and Rubella Targets SAGE Meeting 6 November 2013 P. Strebel, WHO/IVB/EPI Overview Global progress Regional updates Summary 2 Measles and Rubella Targets
Update on Progress and Challenges in Achieving Measles and Rubella Targets SAGE Meeting 6 November 2013 P. Strebel, WHO/IVB/EPI Overview Global progress Regional updates Summary 2 Measles and Rubella Targets
Global Overview Polio Partners Group, December Note: Gavi requirements of $122.2 million are not included in this slide
 Global Overview Polio Partners Group, December 2015 1 Note: Gavi requirements of $122.2 million are not included in this slide The Polio Endgame Plan 1. Poliovirus detection & interruption 2. OPV2 withdrawal,
Global Overview Polio Partners Group, December 2015 1 Note: Gavi requirements of $122.2 million are not included in this slide The Polio Endgame Plan 1. Poliovirus detection & interruption 2. OPV2 withdrawal,
Influenza immunization in pregnancy: observations in mother, fetus, infant
 Influenza immunization in pregnancy: observations in mother, fetus, infant Mark C. Steinhoff, MD Director, Global Health Center Professor of Pediatrics Cincinnati Children s Hospital Medical Center Cincinnati
Influenza immunization in pregnancy: observations in mother, fetus, infant Mark C. Steinhoff, MD Director, Global Health Center Professor of Pediatrics Cincinnati Children s Hospital Medical Center Cincinnati
Maternal Influenza Immunization at WHO
 Maternal Influenza Immunization at WHO Dr. Justin R. Ortiz Medical Officer Initiative for Vaccine Research GAP Partners Meeting Dubai, UAE 26 March 2014 WHO Position Paper Mother s Gift Trial GAVI Review
Maternal Influenza Immunization at WHO Dr. Justin R. Ortiz Medical Officer Initiative for Vaccine Research GAP Partners Meeting Dubai, UAE 26 March 2014 WHO Position Paper Mother s Gift Trial GAVI Review
Influenza Surveillance Animal and Public Health Partnership. Jennifer Koeman Director, Producer and Public Health National Pork Board
 Influenza Surveillance Animal and Public Health Partnership Jennifer Koeman Director, Producer and Public Health National Pork Board Outline Background on influenza surveillance in swine Case example animal
Influenza Surveillance Animal and Public Health Partnership Jennifer Koeman Director, Producer and Public Health National Pork Board Outline Background on influenza surveillance in swine Case example animal
The Case for Reactive Mass Oral Cholera Vaccinations
 The Case for Reactive Mass Oral Cholera Vaccinations Rita Reyburn 1 *, Jacqueline L. Deen 1, Rebecca F. Grais 2, Sujit K. Bhattacharya 3, Dipika Sur 3, Anna L. Lopez 1, Mohamed S. Jiddawi 4, John D. Clemens
The Case for Reactive Mass Oral Cholera Vaccinations Rita Reyburn 1 *, Jacqueline L. Deen 1, Rebecca F. Grais 2, Sujit K. Bhattacharya 3, Dipika Sur 3, Anna L. Lopez 1, Mohamed S. Jiddawi 4, John D. Clemens
Expanded Programme on Immunization (EPI)
 Timor-Leste 217 Expanded Programme on Immunization (EPI) FACT SHEET Acronyms AD Auto disable MCV1 First dose measles containing vaccine AEFI Adverse events following immunization MCV2 Second dose measles
Timor-Leste 217 Expanded Programme on Immunization (EPI) FACT SHEET Acronyms AD Auto disable MCV1 First dose measles containing vaccine AEFI Adverse events following immunization MCV2 Second dose measles
Global Health Security: Preparedness and Response: can we do better and stay safe?
 Global Health Security: Preparedness and Response: can we do better and stay safe? John Watson Health Protection Directorate, Public Health England Formerly Deputy Chief Medical Officer, Department of
Global Health Security: Preparedness and Response: can we do better and stay safe? John Watson Health Protection Directorate, Public Health England Formerly Deputy Chief Medical Officer, Department of
Vaccine Innovation and Adult Immunization Landscape
 Vaccine Innovation and Adult Immunization Landscape National Adult and Influenza Immunization Summit, May 12-14, 2015 Phyllis Arthur Senior Director Vaccines, Immunotherapeutics & Diagnostics Policy parthur@bio.org
Vaccine Innovation and Adult Immunization Landscape National Adult and Influenza Immunization Summit, May 12-14, 2015 Phyllis Arthur Senior Director Vaccines, Immunotherapeutics & Diagnostics Policy parthur@bio.org
Efficacy of a single-dose regimen of inactivated whole-cell oral cholera vaccine: results from 2 years of follow-up of a randomised trial
 Efficacy of a single-dose regimen of inactivated whole-cell oral cholera vaccine: results from 2 years of follow-up of a randomised trial Firdausi Qadri, Mohammad Ali, Julia Lynch, Fahima Chowdhury, Ashraful
Efficacy of a single-dose regimen of inactivated whole-cell oral cholera vaccine: results from 2 years of follow-up of a randomised trial Firdausi Qadri, Mohammad Ali, Julia Lynch, Fahima Chowdhury, Ashraful
Date rights url accessed: 19 March Copyright and License Policies
 PUBLISHED VERSION Sur, Dipika; Kanungo, Suman; Sah, Binod; Manna, Byomkesh; Ali, Mohammad; Paisley, Allison M.; Niyogi, Swapan K.; Park, Jin Kyung; Sarkar, Banawarilal; Puri, Mahesh K.; Kim, Deok Ryun;
PUBLISHED VERSION Sur, Dipika; Kanungo, Suman; Sah, Binod; Manna, Byomkesh; Ali, Mohammad; Paisley, Allison M.; Niyogi, Swapan K.; Park, Jin Kyung; Sarkar, Banawarilal; Puri, Mahesh K.; Kim, Deok Ryun;
CASE STUDY Improving the quality of VMMC services at Mangochi, Mzimba North, and Nkhotakota District Hospitals in Malawi
 CASE STUDY Improving the quality of VMMC services at Mangochi, Mzimba North, and Nkhotakota District Hospitals in Malawi With support from United States Agency for International Development (USAID), multi-sectoral
CASE STUDY Improving the quality of VMMC services at Mangochi, Mzimba North, and Nkhotakota District Hospitals in Malawi With support from United States Agency for International Development (USAID), multi-sectoral
New vaccine technologies: Promising advances may save more lives
 New vaccine technologies: Promising advances may save more lives Vaccine Technology III June 10, 2010 PATH s vision A world where innovation ensures that health is within reach for everyone. 2 PATH s mission
New vaccine technologies: Promising advances may save more lives Vaccine Technology III June 10, 2010 PATH s vision A world where innovation ensures that health is within reach for everyone. 2 PATH s mission
Update on Status of Wild Poliovirus Outbreak in Kenya. 9 th Meeting of the IMB 1-3 October 2013 London, UK
 Update on Status of Wild Poliovirus Outbreak in Kenya 9 th Meeting of the IMB 1-3 October 2013 London, UK Contents Background Confirmation and Immediate Response High Risk Areas Coordination of Response
Update on Status of Wild Poliovirus Outbreak in Kenya 9 th Meeting of the IMB 1-3 October 2013 London, UK Contents Background Confirmation and Immediate Response High Risk Areas Coordination of Response
TECHNICAL UPDATE: POLIO ERADICATION AND ROUTINE IMMUNIZATION STRENGTHENING. Gavi Board pre-meeting December 9, 2014, Geneva.
 TECHNICAL UPDATE: POLIO ERADICATION AND ROUTINE IMMUNIZATION STRENGTHENING Gavi Board pre-meeting December 9, 2014, Geneva www.gavi.org SESSION OUTLINE Global polio eradication update Overview of Polio
TECHNICAL UPDATE: POLIO ERADICATION AND ROUTINE IMMUNIZATION STRENGTHENING Gavi Board pre-meeting December 9, 2014, Geneva www.gavi.org SESSION OUTLINE Global polio eradication update Overview of Polio
Global and Regional update on Polio Eradication Activities. Kenya Paediatric Association Pride Inn, Mombasa April 26, 2018
 Global and Regional update on Polio Eradication Activities Kenya Paediatric Association Pride Inn, Mombasa April 26, 2018 Presentation Outline 1. Background information 2. Poliovirus detection & interruption
Global and Regional update on Polio Eradication Activities Kenya Paediatric Association Pride Inn, Mombasa April 26, 2018 Presentation Outline 1. Background information 2. Poliovirus detection & interruption
Malaria Vaccine Implementation Programme (MVIP) update and framework for policy decision. Mary J Hamel, WHO MPAC, 11 April 2018
 Malaria Vaccine Implementation Programme (MVIP) update and framework for policy decision Mary J Hamel, WHO MPAC, 11 April 2018 MVIP update Background to MVIP MVIP Updates Regulatory Vaccine introduction
Malaria Vaccine Implementation Programme (MVIP) update and framework for policy decision Mary J Hamel, WHO MPAC, 11 April 2018 MVIP update Background to MVIP MVIP Updates Regulatory Vaccine introduction
