Systematic review of epidemiological evidence
|
|
|
- Shanna Lindsey
- 6 years ago
- Views:
Transcription
1 Non-specific effects of BCG, DTP and measles vaccines Systematic review of epidemiological evidence Julian Higgins on behalf of University of Bristol: Julian Higgins (Co-PI), José López-López, Katherine Chaplin, Hannah Christensen, Natasha Martin, Jelena Savović, Jonathan Sterne Enhance Reviews: Karla Soares-Weiser (Co-PI), Artemisia Kakourou, Maria Christou. University of California, Berkeley: Arthur Reingold (Co-PI), Michelle Beam, Emi Han, Emma Smith, Paul Zhang 1
2 2 Systematic review objectives to review published and grey literature on epidemiological studies addressing non-specific effects of BCG, DTP and measles-containing vaccines on: i. mortality from causes other than those conditions that the vaccine is designed to prevent and, ii. on all-cause mortality in children under five years of age. to appraise the evidence critically
3 3 Research questions Is administration of each vaccine given in infancy associated with an effect on each mortality outcome in children up to five years of age? Is there a difference of the effect: between boys and girls? by age dose is received and number of doses? by prior, or co-administration of vitamin A and/ or other vaccines? by sequence/order in which vaccines are given?
4 4 Outline What we sought Articles we found Comments on review process overlapping samples risk of bias Results for main comparisons and comparisons of vaccine strategies Critical appraisal (risk of bias) Summary
5 Criteria for inclusion Participants: children up to 5 years Intervention: vaccination (BCG, DTP or measles) Comparators: no vaccination (BCG, DTP or measles respectively) or simultaneous administration of another vaccine Comparisons of different sequences of vaccine also included Excluding high-titre and medium-title measles vaccine Outcomes: mortality (as previous slide) Study designs: randomized (or quasi-randomized) controlled trials; cohort studies; case-control studies Data sources: primary research papers; or re-analyses of primary studies with full articles describing methodology (published or unpublished, any language) 5
6 Included Eligibility Screening Identification Records identified through database searching N = 5,550 Additional records identified by contacting experts in the field N = 809 Records identified through WHO International Clinical Trials Registry N = 670 Records after duplicates removed N = 5,600 Records screened N = 5,600 Records excluded: N = 4,723 (Databases) N = 660 (WHO registry) Full-text articles scanned for eligibility N = 852 Full-text articles excluded, with reasons N = 639 Articles identified through reference lists: N = 13 Articles identified through the Working Group: N = 12 Full-text articles assessed for eligibility N = 238 Full-text articles included (N = 73) Full-text articles with additional information relevant to the included papers (N = 35) Full-text articles excluded, after checking eligibility N = 130. Reasons: Study design: N = 91 No mortality: N = 51 No data on 5 years: N = 14 No data on vaccines: N = 37 PDF not obtained: N = data points extracted Records of potentially relevant ongoing studies (N = 10)
7 7 Locations of studies (73 articles) USA (2) Amr-A Canada (1) India (5) Sear-D Bangladesh (6) Amr-D Haiti (1) Philippines (2) Papua New Guinea (1) Wpr-B Algeria (1) Senegal (5) Afr-D Guinea-Bissau (37) Burkina Faso (1) Ghana (4) Benin (1) Nigeria (1) South Africa (1) Dem. Rep. Congo (1) Sudan (1) Burundi (1) Malawi (1) Afr-E Emr-D
8 37 articles from Guinea-Bissau Cacheu Quinhamel Oio Gabu 6 articles from rural areas: Oio, Cacheu, Gabu, Quinhamel, Biombo Guinea-Bissau (37) 3 articles reporting data on hospitalized children (Simao Mendes National Hospital) 28 articles from Bandim area 8
9 37 articles from Guinea-Bissau Cacheu Oio Gabu Quinhamel Mindara Bissau Belem Bandim 9
10 10 Overlap of children (1/4) From the protocol: For each study, we will use the best available comparison-level data or group-level data to derive the rate ratio (RR) for vaccinated compared with unvaccinated individuals, with 95% confidence interval (CI)
11 11 Overlap of children (2/4) Grouped articles by similarity, e.g. e.g.1 Guinea-Bissau group A papers relating to a randomized trial of BCG (early vs delayed) and vitamin A supplementation in low-birth weight infants in the Bandim area of Bissau also data on measles vaccine and mortality e.g.2 Guinea-Bissau group C papers relating to a randomized trial of two vs. one dose of measles vaccine in the same area
12 GOAL VACCINES SAMPLE SIZE FOLLOW UP (MO) #9436 #9434 Re-analysis of 3 RCTs of VAS Re-analysis of 2 RCTs of 2 vs 1 MV doses BCG, MV 1, MV 1, #25 RCT of BCG and VAS in LBW BCG, DTP 2,343 6 #61 RCT of BCG in LBW BCG #166 RCT of BCG and VAS in LBW BCG, DTP1 2, #339 RCT of BCG and VAS in LBW BCG 1, #324 RCT of BCG revaccination BCG, DTP 2, #1731 RCT of 2 vs 1 MV doses MV 6, #1986 Observational study of children 6-17 months received VAS BCG, DTP, MV 1, Guinea Bissau A Guinea Bissau B Guinea Bissau C 12
13 13 Overlap of children (4/4) BUT... likely that part of the population receiving BCG at birth (Guinea-Bissau A) later participated in the measles vaccine trial (Guinea-Bissau C) SO... created birth cohorts (which may divide up differently for different vaccines) We developed a detailed algorithm to select one result for each comparison (where available) to represent the children in each birth cohort avoids double counting of children
14 14 Risk of bias assessment For RCTs: Cochrane tool for risk of bias in randomized trials Observational studies: In-development Cochrane tool for risk of bias in non-randomized studies project led from University of Bristol with international methodologists from (among others) universities of Harvard, Leiden, Liverpool, London School of Hygiene and Tropical Medicine, McGill, McMaster, Ottawa, Oxford, Paris Descartes, Toronto; and from RTI International, UK Medical Research Council, Nordic Cochrane Centre
15 15 Organization of results Birth This schedule is our reference point BCG All results here are for all-cause mortality DTP MV
16 16 Organization of results BCG Birth No BCG DTP MV Comparison 1 BCG vs No BCG Impact of age, gender, vitamin A status General remark: Follow-up is often quite short, so as not to be affected by subsequent vaccines
17 17 Organization of results Birth BCG Comparison 2 DTP vs No DTP Impact of age, gender, vitamin A status DTP No DTP MV
18 18 Organization of results Birth BCG Comparison 5 Measles vaccine vs No measles vaccine Impact of age, gender, vitamin A status DTP MV No MV
19 19 Organization of results BCG DTP Birth BCG with DTP Comparison 3 Reference schedule for BCG and DTP vs Simultaneous administration MV
20 20 Organization of results BCG Birth DTP Comparison 4 Reference schedule for BCG and DTP vs DTP before BCG DTP BCG MV
21 21 Organization of results DTP MV Birth BCG DTP with MV Comparison 6 Reference schedule for DTP and measles vs Simultaneous administration
22 22 Organization of results DTP Birth BCG MV Comparison 7 Reference schedule for DTP and measles vs DTP after measles vaccine MV DTP
23 23 Birth BCG No BCG DTP MV Results 1. BCG VS NO BCG
24 24 BCG and all-cause mortality 18 independent birth cohorts 5 trials, 13 observational studies 4 excluded from analysis due to very high risk of bias Total sample size approx. 36,000 children range 105 to Follow up ranges from 1 to 60 months of age
25 Studies BCG and all-cause mortality Results Birth cohort Age at first dose Observation period 1. Randomized and quasi-randomized trials Canada Guinea-Bissau (early) Guinea-Bissau (main) USA c.1935 USA c days 2 days 2 days 0-4 years 7-10 days age 60 months age 1 month age 1 month age 48 months age 60 months 2. Case-control studies Benin Cohort studies Guinea-Bissau Guinea-Bissau Guinea-Bissau India India Malawi Papua New Guinea Senegal Excluded (Very high risk of bias) Bangladesh Burkina Faso Ghana India NR NR (0-8 months) 1-7 days Median 1 month Median 1.6 months Median 19 days Median 16 days Median 1 month NR (by 12 months in 44%) 0-2 months Mean 4.8 months NR (by 12 months in 57%) Mean 17 days age 4-36 months age 8 months age 6 months 6 months follow-up age 12 months age 6 months age 8 months age 1-6 months age 24 months age 0-60 months 6 months follow-up age 60 months age 1.2 months Reduce mortality Relative mortality rate (with 95% confidence interval) Increase mortality Further details Vaccine beneficial Vaccine harmful No effect 25
26 BCG and all-cause mortality 26 Birth cohort Age at first dose Observation period 1. Randomized and quasi-randomized trials Canada Guinea-Bissau (early) Guinea-Bissau (main) USA c.1935 USA c days 2 days 2 days 0-4 years 7-10 days age 60 months age 1 month age 1 month age 48 months age 60 months 2. Case-control studies Benin NR age 4-36 months 3. Cohort studies Guinea-Bissau Guinea-Bissau Guinea-Bissau India India Malawi Papua New Guinea Senegal NR (0-8 months) 1-7 days Median 1 month Median 1.6 months Median 19 days Median 16 days Median 1 month NR (by 12 months in 44%) age 8 months age 6 months 6 months follow-up age 12 months age 6 months age 8 months age 1-6 months age 24 months Excluded (Very high risk of bias) Bangladesh Burkina Faso Ghana India months Mean 4.8 months NR (by 12 months in 57%) Mean 17 days age 0-60 months 6 months follow-up age 60 months age 1.2 months Vaccine beneficial Vaccine harmful
27 Is there a difference in the effect of Birth cohort Age of vaccination BCG by age? Follow-up Different ages at vaccination Bangladesh Guinea-Bissau vaccination at 0-2 months vaccination at 2-6 months vaccination at 6-12 months vaccination at months vaccination at first week vaccination after first week age 0-60 months up to 6 months Supported by evidence from trials of early vs delayed BCG Vaccine beneficial Vaccine harmful 27
28 Is there a difference in the effect of BCG by gender? Birth cohort Analysis of boy/girl differences in effect ( statistical interaction ) Burkina Faso Guinea-Bissau Guinea-Bissau Guinea-Bissau [RCT] India India Malawi Papua New Guinea Senegal Boys benefit more Girls benefit more 28
29 29 Birth BCG DTP No DTP MV Results 2. DTP VS NO DTP
30 30 DTP and all-cause mortality 16 independent birth cohorts of children, all observational 6 excluded from analysis due to very high risk of bias Always given with OPV where information available (8 out of 10 studies) Total sample size approx. 28,000 children range 132 to 9085 Follow up ranges from 6 to 36 months of age
31 DTP and all-cause mortality 1. Case-control studies Benin Age at first dose NR Observation period age 4-36 months 2. Cohort studies Bangladesh Burkina Faso Guinea-Bissau Guinea-Bissau Guinea-Bissau India Malawi Papua New Guinea Senegal Median 2.8 months Mean 6.3 months NR (3-8 months) Median 3 months NR (1.5-6 months) Median 2 months Median 2.2 months Before 3 months for most NR (before 9 months) mo 6 months follow-up age 8 months 6 months follow-up age 6 months age 6 months age 8 months age 1-5 months age 24 months Excluded (Very high risk of bias) Ghana Ghana Guinea-Bissau India India Philippines NR NR (by 12 months in 47%) NR (from 1.2 months) Median 3.8 months Mean 2 months NR (before 7 months) age months age 60 months age months age 12 months age 8 months age 30 months Vaccine beneficial Vaccine harmful 31
32 32 Is there a difference in the effect of DTP by age? No within-cohort comparisons of effects at different ages at vaccination
33 33 Is there a difference in the effect of DTP by gender? Analysis of boy/girl differences in effect ( statistical interaction ) Bangladesh Burkina Faso Guinea-Bissau Guinea-Bissau Guinea-Bissau Guinea-Bissau India India Malawi Papua New Guinea Philippines Senegal Boys benefit more Girls benefit more
34 Is there a difference in the effect of Boys Bangladesh Burkina Faso Guinea-Bissau Guinea-Bissau Guinea-Bissau India Malawi Papua New Guinea Senegal Boys (Very high risk of bias) Guinea-Bissau India Philippines Girls Bangladesh Burkina Faso Guinea-Bissau Guinea-Bissau Guinea-Bissau India Malawi Papua New Guinea Senegal Girls (Very high risk of bias) Guinea-Bissau India Philippines DTP by gender? Vaccine beneficial Vaccine harmful 34
35 3. BCG AND DTP GIVEN Birth SIMULTANEOUSLY BCG DTP BCG with DTP MV Observation period Bangladesh India Senegal mo age 12 months age 24 months Favours simultaneous Favours reference 35
36 Birth 4. DTP BEFORE BCG BCG DTP Observation period DTP MV BCG Bangladesh mo India age 12 months Senegal age 24 months Comparison of DTP before or with BCG Papua New Guinea Papua New Guinea age 1-5 months age 6-11 months Favours reverse order Favours reference 36
37 37 Birth BCG DTP MV No MV Results 5. MEASLES VACCINE VS NO MEASLES VACCINE
38 38 Measles vaccine and all-cause mortality 28 independent birth cohorts 4 trials, 24 observational studies 6 excluded from analysis due to very high risk of bias Total sample size approx. 116,000 children range 99 to 36,650 Follow up ranges from 9 to 60 months of age
39 Measles vaccine and all-cause mortality 1. Randomized trials Guinea-Bissau Guinea-Bissau Guinea-Bissau Nigeria c Case-control studies Benin India Cohort studies Bangladesh Bangladesh DR Congo Guinea-Bissau Guinea-Bissau Guinea-Bissau Guinea-Bissau Guinea-Bissau Haiti India India Malawi Median 10.8 months Papua New Guinea NR (by 12 months in 74%) Senegal Mean 15.8 months Senegal Mean 11.6 months Senegal NR (by 12 [24] mo in 9% [20%]) Excluded (Very high risk of bias) Burundi Ghana Ghana Ghana India Senegal Age at first dose 6 months 6 months 4.5 months 6-24 months NR NR (before 12 months) NR (from 9 months) NR (from 9 months) Mean 8.8 months NR (6-35 months) NR (6-35 months) Median 11.1; 15 months Median 10.6 months NR (by 12 mo in 55%) Median 9 months NR (at 6-8 [8-11] mo in 85% [15%]) Median 9.4 months NR (at 9-11 months in 59%) NR (6-35 months) Median 9.1 months NR (by 12 [24] mo in 5% [64%]) Mean 9.4 months Median 9.7 months Observation period age 6-9 months age 6-9 months age months 6-20 months follow-up age 4-36 months age months age 9-60 months age 9-60 months age 7-21 months 13 months follow-up 12 months follow-up age 17.5 months or more age 7-19 months age 9-24 months age 9-39 months age months age months age 9-18 months age 6-11 months age 9-24 months age 9-24 months age 24 months 6 months follow-up 4 months follow-up age 9-11 months age 60 months age 9-15 months age up to 24 months Vaccine beneficial Vaccine harmful 39
40 Is there a difference in the effect of measles vaccine by age? Different ages at vaccination Benin [CC] Age of vaccination up to 12 months after 12 months Follow-up age up to 35 months Vaccine beneficial Vaccine harmful
41 Is there a difference in the effect of measles vaccine by gender? Analysis of boy/girl differences in effect ( statistical interaction ) Ghana Guinea-Bissau Guinea-Bissau [RCT] Guinea-Bissau Guinea-Bissau [RCT] India Malawi Senegal Senegal Senegal Senegal Boys benefit more Girls benefit more 41
42 Is there a difference in the effect of measles vaccine by gender? Boys Guinea-Bissau [RCT] Guinea-Bissau Guinea-Bissau [RCT] Malawi Senegal Senegal Senegal Senegal Boys (Very high risk of bias) Ghana Guinea-Bissau India Girls Guinea-Bissau [RCT] Guinea-Bissau Guinea-Bissau [RCT] Malawi Senegal Senegal Senegal Senegal Girls (Very high risk of bias) Ghana Guinea-Bissau India Vaccine beneficial Vaccine harmful 42
43 43 6. DTP AND MEASLES VACCINE GIVEN SIMULTANEOUSLY Birth BCG Observation period DTP MV DTP with MV Guinea-Bissau & Guinea-Bissau India Malawi Senegal age 6-17 months age 9-24 months age months age 9-18 months age 24 months Favours simultaneous Favours reference
44 44 7. DTP AFTER MEASLES VACCINE Birth BCG DTP MV MV DTP Observation period Guinea-Bissau & India Senegal age 6-17 months age months age 24 months Favours reversed Favours reference
45 45 Comment RISK OF BIAS
46 Risk of bias is not the same as Bias Imprecision characteristics that raise risk of bias reflected in the confidence interval Quality Reporting bias can occur in well-conducted studies good methods may have been used but not well reported 46
47 47 Main concerns about risk of bias Confounding Misclassification Selection bias Co-interventions
48 48 Confounding (inherent differences between children vaccinated and children not vaccinated) Example: DR Congo (MV) (included in analysis) GROUP 1: Offered vaccination GROUP 1v: Received vaccination GROUP 2: Unvaccinated (in a different area)
49 Confounding (inherent differences between children vaccinated and children not vaccinated) Example: DR Congo (MV) (included in analysis) No allowance for possible differences between vaccinated and unvaccinated children GROUP 1: Offered vaccination GROUP 1v: Received vaccination GROUP 2: Unvaccinated (in a different area) there were no gross social differences between the two areas 49
50 Confounding: frail children Frail children believed less likely to be vaccinated Disappointing amount of evidence about this So those vaccinated inherently less likely to die even if vaccine has no effect So naive comparison of vaccinated vs not vaccinated likely to be biased in favour of vaccine Lack of comprehensive adjustment for frailty e.g. India (included in analysis) 50
51 Confounding: age e.g. India (excluded from analysis) Can compute unadjusted comparison of DTP vs no DTP (i.e. BCG only), but children are at very different ages Bias depends on mortality patterns over time 51
52 52 Misclassification bias (determining non-vaccination) e.g. Burkina-Faso (included in analysis) Researchers visited families every 6-12 months Collected information from vaccination cards Vaccinated: Vaccination recorded on vaccination card Unvaccinated: When the card was not seen, we assumed that the child had not been vaccinated It s possible these children would have been vaccinated: if so the result is biased towards no effect (towards the null)
53 53 Misclassification bias (survival bias) Major problem can occur if vaccination status is updated retrospectively particularly if vaccination cards are destroyed when a child dies particularly if there is a long period between visits to the children Excluded any study with potentially serious biases in this respect
54 54 Bias arising from selection of participants long after vaccines were given e.g. Philippines (excluded from analysis) Children aged up to 30 months at recruitment DTP should be given at 6, 10 and 14 weeks A randomized trial would start follow-up at intervention
55 55 Co-interventions e.g. Ghana (excluded from analysis) Vaccines are highly correlated so effect for BCG includes effects of DTP and measles vaccine (Other critical problems with this study due to annual visits to children)
56 CONCLUDING REMARKS 56
57 57 Summary There was some evidence from randomized trials, but with short follow up All observational studies were considered to be at high risk of bias some excluded because judged to be at very high risk Biases due to selective reporting may also be present, but these are very difficult to detect
58 58 Summary: BCG Is administration of BCG vaccine given in infancy associated with an effect on all-cause mortality in children up to five years of age? May reduce risk of all-cause mortality GRADE: Very little confidence Is there a difference of the effect: between boys and girls? No difference apparent by age dose is received? Suggestion of greater benefit when given earlier by prior, or co-administration of vitamin A and/ or other vaccines? Insufficient evidence
59 Summary: DTP (1/2) Is administration of DTP vaccine (with OPV) given in infancy associated with an effect on all-cause mortality in children up to five years of age? Inconsistent evidence* GRADE: Very little confidence Is there a difference of the effect: between boys and girls? Effects may be more deleterious or variable in girls by age dose is received and number of doses received? No data available by prior, or co-administration of vitamin A and/ or other vaccines? Insufficient evidence *No studies reported on DTP without OPV 59
60 Summary: DTP (2/2) Is there a difference of the effect: by sequence/order in which vaccines are given? When administered simultaneously with BCG May reduce risk of all-cause mortality GRADE: Very little confidence compared with BCG before DTP When administered before BCG No difference apparent GRADE: Very little confidence When administered simultaneously with measles vaccine May increase risk of all-cause mortality compared with DTP before measles GRADE: Very little confidence When administered after measles vaccine May increase risk of all-cause mortality GRADE: Very little confidence compared with DTP before measles 60
61 61 Summary: Measles vaccine Is administration of measles vaccine given in infancy associated with an effect on all-cause mortality in children up to five years of age? May reduce risk of all-cause mortality GRADE: Limited confidence Is there a difference of the effect: between boys and girls? Suggestion of (greater) benefit in girls by age dose is received? Suggestion of greater benefit when given earlier by prior, or co-administration of vitamin A and/ or other vaccines? Insufficient evidence
62 62 Concluding remarks There is not a single approach to design and analysis of studies in this research area Leaves open the possibility that investigators may have tried multiple ways to select and analyse the data Thereby putting the accessible literature as a whole at risk of bias
Systematic review of the non- specific effects of BCG, DTP and measles containing vaccines
 Systematic review of the non- specific effects of BCG, DTP and measles containing vaccines Higgins JPT, Soares- Weiser K, Reingold A 13 March 2014 Contents 1 Executive Summary... 3 2 Background... 4 3
Systematic review of the non- specific effects of BCG, DTP and measles containing vaccines Higgins JPT, Soares- Weiser K, Reingold A 13 March 2014 Contents 1 Executive Summary... 3 2 Background... 4 3
Measles vaccination in presence of maternal measles antibodies confers Nonspecific beneficial effects on child survival
 Measles vaccination in presence of maternal measles antibodies confers Nonspecific beneficial effects on child survival Christine Stabell Benn, Cesario Martins & Peter Aaby Bandim Health Project, Guinea-Bissau
Measles vaccination in presence of maternal measles antibodies confers Nonspecific beneficial effects on child survival Christine Stabell Benn, Cesario Martins & Peter Aaby Bandim Health Project, Guinea-Bissau
WG: Vaccinations and child survival Focus:
 WG: Vaccinations and child survival Focus: Monitoring childhood interventions including routine services and campaigns => To find possible changes in policy Why is that necessary? Paradox: All interventions
WG: Vaccinations and child survival Focus: Monitoring childhood interventions including routine services and campaigns => To find possible changes in policy Why is that necessary? Paradox: All interventions
ANNEX C: RISK- OF- BIAS ASSESSMENTS
 Systematic review of the non- specific effects of BCG, DTP and measles containing vaccines ANNEX C: RISK- OF- BIAS ASSESSMENTS 1 Risk of bias assessments for randomized trials of BCG... 2 2 Risk of bias
Systematic review of the non- specific effects of BCG, DTP and measles containing vaccines ANNEX C: RISK- OF- BIAS ASSESSMENTS 1 Risk of bias assessments for randomized trials of BCG... 2 2 Risk of bias
Authors response. Editorial. Responses to referees: Impact of BCG, DTP and measles-containing vaccines on childhood mortality: a systematic review
 Responses to referees: Impact of BCG, DTP and measles-containing vaccines on childhood mortality: a systematic review We are very grateful for all of the comments, and we respond to each below. In a version
Responses to referees: Impact of BCG, DTP and measles-containing vaccines on childhood mortality: a systematic review We are very grateful for all of the comments, and we respond to each below. In a version
Determinants of Vaccination Status in Rural Guinea-Bissau. Linda Hornshøj
 Determinants of Vaccination Status in Rural Guinea-Bissau Linda Hornshøj Background In1974 World Health organization (WHO) launched the Expanded Program on Immunization (EPI) to provide routine vaccinations
Determinants of Vaccination Status in Rural Guinea-Bissau Linda Hornshøj Background In1974 World Health organization (WHO) launched the Expanded Program on Immunization (EPI) to provide routine vaccinations
How to optimise immunisation: a live vaccine last policy. Frank Shann University of Melbourne
 How to optimise immunisation: a live vaccine last policy Frank Shann University of Melbourne We can no longer assume that a vaccine: 1. Acts independently of other vaccines 2. Influences only infections
How to optimise immunisation: a live vaccine last policy Frank Shann University of Melbourne We can no longer assume that a vaccine: 1. Acts independently of other vaccines 2. Influences only infections
BMJ Open. For peer review only -
 Testing the hypothesis the diphtheria-tetanus-pertussis vaccine has negative non-specific and sex-differential effects on child survival in low-income countries. A review Journal: Manuscript ID: bmjopen-0-0000
Testing the hypothesis the diphtheria-tetanus-pertussis vaccine has negative non-specific and sex-differential effects on child survival in low-income countries. A review Journal: Manuscript ID: bmjopen-0-0000
Update from GAVI Aurelia Nguyen
 Update from GAVI Aurelia Nguyen (Copenhagen, Denmark, 27 June 2012) GAVI vaccine support Currently supported vaccines: pentavalent, pneumococcal, rotavirus, meningitis A, human papillomavirus (HPV), rubella,
Update from GAVI Aurelia Nguyen (Copenhagen, Denmark, 27 June 2012) GAVI vaccine support Currently supported vaccines: pentavalent, pneumococcal, rotavirus, meningitis A, human papillomavirus (HPV), rubella,
Analysis of Coverage of Fully Immunized Child (FIC), Associated Factors, Outcomes, and Impact Using Routinely Collected Population Cohort Data
 Analysis of Coverage of Fully Immunized Child (FIC), Associated Factors, Outcomes, and Impact Using Routinely Collected Population Cohort Data 21-214 April 215 Analysis of Coverage of Fully Immunized Child
Analysis of Coverage of Fully Immunized Child (FIC), Associated Factors, Outcomes, and Impact Using Routinely Collected Population Cohort Data 21-214 April 215 Analysis of Coverage of Fully Immunized Child
Rotavirus vaccines: Issues not fully addressed in efficacy trials
 Rotavirus vaccines: Issues not fully addressed in efficacy trials TM Umesh D. Parashar Lead, Viral Gastroenteritis Team CDC, Atlanta, USA uparashar@cdc.gov 1 Two New Rotavirus Vaccines Licensed in 2006
Rotavirus vaccines: Issues not fully addressed in efficacy trials TM Umesh D. Parashar Lead, Viral Gastroenteritis Team CDC, Atlanta, USA uparashar@cdc.gov 1 Two New Rotavirus Vaccines Licensed in 2006
The effect of vitamin A provided in campaign may depend on vaccination. Observational study from Guinea-Bissau
 The effect of vitamin A provided in campaign may depend on vaccination status: Observational study from Guinea-Bissau Ane Bærent Fisker, PhD student Bandim Health Project, Guinea-Bissau & Denmark Dept.
The effect of vitamin A provided in campaign may depend on vaccination status: Observational study from Guinea-Bissau Ane Bærent Fisker, PhD student Bandim Health Project, Guinea-Bissau & Denmark Dept.
Is early measles vaccination associated with stronger survival benefits than later measles vaccination?
 Hansen et al. BMC Public Health (2018) 18:984 https://doi.org/10.1186/s12889-018-5866-y RESEARCH ARTICLE Open Access Is early measles vaccination associated with stronger survival benefits than later measles
Hansen et al. BMC Public Health (2018) 18:984 https://doi.org/10.1186/s12889-018-5866-y RESEARCH ARTICLE Open Access Is early measles vaccination associated with stronger survival benefits than later measles
Update on Progress and Challenges in Achieving Measles and Rubella Targets
 Update on Progress and Challenges in Achieving Measles and Rubella Targets SAGE Meeting 6 November 2013 P. Strebel, WHO/IVB/EPI Overview Global progress Regional updates Summary 2 Measles and Rubella Targets
Update on Progress and Challenges in Achieving Measles and Rubella Targets SAGE Meeting 6 November 2013 P. Strebel, WHO/IVB/EPI Overview Global progress Regional updates Summary 2 Measles and Rubella Targets
Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook. UNICEF Supply Division
 Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook UNICEF Supply Division Update: October 2013 0 Pneumococcal Conjugate Vaccine (PCV) Supply & Demand Outlook October 2013 Update Key updates
Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook UNICEF Supply Division Update: October 2013 0 Pneumococcal Conjugate Vaccine (PCV) Supply & Demand Outlook October 2013 Update Key updates
GOAL 2: ACHIEVE RUBELLA AND CRS ELIMINATION. (indicator G2.2) Highlights
 GOAL 2: ACHIEVE RUBELLA AND CRS ELIMINATION (indicator G2.2) Highlights As of December 2014, 140 Member States had introduced rubella vaccines; coverage, however, varies from 12% to 94% depending on region.
GOAL 2: ACHIEVE RUBELLA AND CRS ELIMINATION (indicator G2.2) Highlights As of December 2014, 140 Member States had introduced rubella vaccines; coverage, however, varies from 12% to 94% depending on region.
Implications of beneficial off-target effects for upcoming eradication campaigns (measles and polio)
 Implications of beneficial off-target effects for upcoming eradication campaigns (measles and polio) Ane Bærent Fisker, MD, PhD Research Center for Vitamins and Vaccines, Bandim Health Project, Statens
Implications of beneficial off-target effects for upcoming eradication campaigns (measles and polio) Ane Bærent Fisker, MD, PhD Research Center for Vitamins and Vaccines, Bandim Health Project, Statens
RESEARCH. Non-specific effects of standard measles vaccine at 4.5 and 9 months of age on childhood mortality: randomised controlled trial
 Non-specific effects of standard measles vaccine at 4.5 and 9 months of age on childhood mortality: randomised controlled trial Peter Aaby, professor, 1,2 Cesário L Martins, clinician, 1 May-Lill Garly,
Non-specific effects of standard measles vaccine at 4.5 and 9 months of age on childhood mortality: randomised controlled trial Peter Aaby, professor, 1,2 Cesário L Martins, clinician, 1 May-Lill Garly,
The Promise of Introducing Rubella Containing Vaccines on the Impact of Rubella and Measles Control
 The Promise of Introducing Rubella Containing Vaccines on the Impact of Rubella and Measles Control GAVI Partners Forum 4 6 December 2012, Dar es Salaam, Tanzania Maya van den Ent, MPH, UNICEF Peter Strebel,
The Promise of Introducing Rubella Containing Vaccines on the Impact of Rubella and Measles Control GAVI Partners Forum 4 6 December 2012, Dar es Salaam, Tanzania Maya van den Ent, MPH, UNICEF Peter Strebel,
Epidemiology of measles in infants younger than 6 months: analysis of surveillance data
 Epidemiology of measles in infants younger than 6 months: analysis of surveillance data 2011-2016 An analysis of the epidemiology of measles in infants younger than 6 months was conducted by the U.S. CDC
Epidemiology of measles in infants younger than 6 months: analysis of surveillance data 2011-2016 An analysis of the epidemiology of measles in infants younger than 6 months was conducted by the U.S. CDC
Comments on this bulletin are welcome. Please to Dr. Mary Agócs
 Global Invasive Bacterial Vaccine Preventable Diseases (IB-VPD) Information and Surveillance Bulletin Reporting Period: January through December 2010 Volume 4: October 2011 The World Health Organization
Global Invasive Bacterial Vaccine Preventable Diseases (IB-VPD) Information and Surveillance Bulletin Reporting Period: January through December 2010 Volume 4: October 2011 The World Health Organization
Lessons learned from the IeDEA West Africa Collaboration
 Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
Evidence based recommendations on nonspecific. BCG, DTP-containing and measlescontaining. on mortality in children under 5 years of age
 Evidence based recommendations on nonspecific effects of BCG, DTP-containing and measlescontaining vaccines on mortality in children under 5 years of age T. Nolan Chair of SAGE Working Group on NSEV Today's(questions(
Evidence based recommendations on nonspecific effects of BCG, DTP-containing and measlescontaining vaccines on mortality in children under 5 years of age T. Nolan Chair of SAGE Working Group on NSEV Today's(questions(
Overview of WHO/UNICEF Immunization Coverage Estimates
 Overview of WHO/UNICEF Immunization Coverage Estimates Marta Gacic-Dobo, Anthony Burton, David Durrheim Meeting of the Strategic Advisory Group of Experts on Immunization (SAGE) 8-10 November 2011 CCV/CICG,
Overview of WHO/UNICEF Immunization Coverage Estimates Marta Gacic-Dobo, Anthony Burton, David Durrheim Meeting of the Strategic Advisory Group of Experts on Immunization (SAGE) 8-10 November 2011 CCV/CICG,
Systematic Reviews Protocols. A Reingold --- PI, Epidemiology review A Pollard -- PI, Immunological review
 Systematic Reviews Protocols A Reingold --- PI, Epidemiology review A Pollard -- PI, Immunological review Two systematic reviews EPIDEMIOLOGICAL STUDIES Evidence available to document magnitude of any
Systematic Reviews Protocols A Reingold --- PI, Epidemiology review A Pollard -- PI, Immunological review Two systematic reviews EPIDEMIOLOGICAL STUDIES Evidence available to document magnitude of any
IMMUNIZATION & VACCINE PREVENTABLE DISEASES
 IMMUNIZATION & VACCINE PREVENTABLE DISEASES MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2015 (Vol 3, issue N 6 ) Special issue on WHO/UNICEF Estimates of National Immunization Coverage
IMMUNIZATION & VACCINE PREVENTABLE DISEASES MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2015 (Vol 3, issue N 6 ) Special issue on WHO/UNICEF Estimates of National Immunization Coverage
What is this document and who is it for?
 Measles and Rubella Initiative s Standard Operating Procedures for Accessing Support for Measles and Rubella Supplementary Immunization Activities During 2016 In the context of measles and rubella elimination
Measles and Rubella Initiative s Standard Operating Procedures for Accessing Support for Measles and Rubella Supplementary Immunization Activities During 2016 In the context of measles and rubella elimination
Annex 2 A. Regional profile: West Africa
 Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
EXPLANATION OF INDICATORS CHOSEN FOR THE 2017 ANNUAL SUN MOVEMENT PROGRESS REPORT
 UNICEF / Zar Mon Annexes EXPLANATION OF INDICATORS CHOSEN FOR THE 2017 ANNUAL SUN MOVEMENT PROGRESS REPORT This report includes nine nutrition statistics, as per the 2017 Global Nutrition Report. These
UNICEF / Zar Mon Annexes EXPLANATION OF INDICATORS CHOSEN FOR THE 2017 ANNUAL SUN MOVEMENT PROGRESS REPORT This report includes nine nutrition statistics, as per the 2017 Global Nutrition Report. These
Methodological issues in the use of anthropometry for evaluation of nutritional status
 Methodological issues in the use of anthropometry for evaluation of nutritional status Mercedes de Onis WHO Department of Nutrition Time schedule child anthropometry Measurement Time frame Frequency
Methodological issues in the use of anthropometry for evaluation of nutritional status Mercedes de Onis WHO Department of Nutrition Time schedule child anthropometry Measurement Time frame Frequency
Global landscape analysis and literature review of 2 nd Year of Life immunization platform
 Global landscape analysis and literature review of 2 nd Year of Life immunization platform Global Vaccine and Immunization Research Forum 15-17 March 2016 Johannesburg, South Africa Imran Mirza; Celina
Global landscape analysis and literature review of 2 nd Year of Life immunization platform Global Vaccine and Immunization Research Forum 15-17 March 2016 Johannesburg, South Africa Imran Mirza; Celina
Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes
 Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes Dr Nora Dellepiane/Dr Anil Kumar Chawla WHO/HQ-Geneva, Switzerland 1 Expedited review procedure
Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes Dr Nora Dellepiane/Dr Anil Kumar Chawla WHO/HQ-Geneva, Switzerland 1 Expedited review procedure
TT Procured by UNICEF
 TT TT Procured by UNICEF 2001-08 250,000,000 200,000,000 150,000,000 100,000,000 50,000,000 0 2001 2002 2003 2004 2005 2006 2007 2008 Routine SIA TT historical demand and forecast overview Upcoming Tender
TT TT Procured by UNICEF 2001-08 250,000,000 200,000,000 150,000,000 100,000,000 50,000,000 0 2001 2002 2003 2004 2005 2006 2007 2008 Routine SIA TT historical demand and forecast overview Upcoming Tender
1) SO1: We would like to suggest that the indicator used to measure vaccine hesitancy be DTP 1 to measles first dose dropout.
 To SAGE Secretariat, WHO Dear Professor Helen Rees, Dear Dr. Jean Marie Okwo-Bele, On behalf of the Civil Society Constituency of the GAVI Alliance, we would like to thank SAGE and its members for the
To SAGE Secretariat, WHO Dear Professor Helen Rees, Dear Dr. Jean Marie Okwo-Bele, On behalf of the Civil Society Constituency of the GAVI Alliance, we would like to thank SAGE and its members for the
Current State of Global HIV Care Continua. Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2
 Current State of Global HIV Care Continua Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2 1) International Association of Providers of AIDS Care
Current State of Global HIV Care Continua Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2 1) International Association of Providers of AIDS Care
Global reductions in measles mortality and the risk of measles resurgence
 Global reductions in measles mortality 2000 2008 and the risk of measles resurgence Measles is one of the most contagious human diseases. In 1980 before the use of measles vaccine was widespread, there
Global reductions in measles mortality 2000 2008 and the risk of measles resurgence Measles is one of the most contagious human diseases. In 1980 before the use of measles vaccine was widespread, there
IMMUNIZATION VACCINES & EMERGENCIES
 No report No data No data IMMUNIZATION VACCINES & EMERGENCIES ROUTINE IMMUNIZATION PERFORMANCE IN THE AFRICAN REGION October 2013 issue Number of children vaccinated with the 3 rd dose of DTPcontaining
No report No data No data IMMUNIZATION VACCINES & EMERGENCIES ROUTINE IMMUNIZATION PERFORMANCE IN THE AFRICAN REGION October 2013 issue Number of children vaccinated with the 3 rd dose of DTPcontaining
Systematic Review of Non-Specific Immunological Effects of Vaccination
 Systematic Review of Non-Specific Immunological Effects of Vaccination Professor Andrew J Pollard Dr Rama Kandasamy Merryn Voysey University of Oxford Immunology Non-specific effects of the immune system
Systematic Review of Non-Specific Immunological Effects of Vaccination Professor Andrew J Pollard Dr Rama Kandasamy Merryn Voysey University of Oxford Immunology Non-specific effects of the immune system
Value of post-licensure data to assess public health value Example of rotavirus vaccines
 Value of post-licensure data to assess public health value Example of rotavirus vaccines TM Umesh D. Parashar Lead, Viral Gastroenteritis Team CDC, Atlanta, USA uparashar@cdc.gov 1 Two New Rotavirus Vaccines
Value of post-licensure data to assess public health value Example of rotavirus vaccines TM Umesh D. Parashar Lead, Viral Gastroenteritis Team CDC, Atlanta, USA uparashar@cdc.gov 1 Two New Rotavirus Vaccines
Pharmacovigilance BMGF Perspective. Raj Long Bill & Melinda Gates Foundation
 Pharmacovigilance BMGF Perspective Raj Long Bill & Melinda Gates Foundation Overview 1. Key challenges -Recap Safety Surveillance Working Group (2012/2013) 2. Overall BMGF strategy and approach 3. Key
Pharmacovigilance BMGF Perspective Raj Long Bill & Melinda Gates Foundation Overview 1. Key challenges -Recap Safety Surveillance Working Group (2012/2013) 2. Overall BMGF strategy and approach 3. Key
Vaccines against Rotavirus & Norovirus. Umesh D. Parashar CDC, Atlanta, GA
 TM Vaccines against Rotavirus & Norovirus Umesh D. Parashar CDC, Atlanta, GA 1 Rotavirus is the Leading Cause Of Severe Diarrhea in Children
TM Vaccines against Rotavirus & Norovirus Umesh D. Parashar CDC, Atlanta, GA 1 Rotavirus is the Leading Cause Of Severe Diarrhea in Children
Comparative Analyses of Adolescent Nutrition Indicators
 Comparative Analyses of Adolescent Nutrition Indicators Rukundo K. Benedict, PhD The DHS Program Stakeholders Consultation on Adolescent Girls Nutrition: Evidence, Guidance, and Gaps October 30 31, 2017
Comparative Analyses of Adolescent Nutrition Indicators Rukundo K. Benedict, PhD The DHS Program Stakeholders Consultation on Adolescent Girls Nutrition: Evidence, Guidance, and Gaps October 30 31, 2017
Statistical considerations in indirect comparisons and network meta-analysis
 Statistical considerations in indirect comparisons and network meta-analysis Said Business School, Oxford, UK March 18-19, 2013 Cochrane Comparing Multiple Interventions Methods Group Oxford Training event,
Statistical considerations in indirect comparisons and network meta-analysis Said Business School, Oxford, UK March 18-19, 2013 Cochrane Comparing Multiple Interventions Methods Group Oxford Training event,
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2016 (Vol 4, issue N 5) Special issue on 2015 WHO/UNICEF Estimates of National Immunization Coverage (WUENIC)
IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2016 (Vol 4, issue N 5) Special issue on 2015 WHO/UNICEF Estimates of National Immunization Coverage (WUENIC)
Impact and cost-effectiveness of rotavirus vaccination in 73 Gavi countries
 August 29, 2018 Impact and cost-effectiveness of rotavirus vaccination in 73 Gavi countries Thirteenth International Rotavirus Symposium 29 31 August 2018 Minsk, Belarus Frédéric Debellut Health Economist,
August 29, 2018 Impact and cost-effectiveness of rotavirus vaccination in 73 Gavi countries Thirteenth International Rotavirus Symposium 29 31 August 2018 Minsk, Belarus Frédéric Debellut Health Economist,
WHO Rotavirus Vaccine Update Dr. Fatima Serhan WHO/FWC/IVB/EPI
 WHO Rotavirus Vaccine Update 2012 Dr. Fatima Serhan WHO/FWC/IVB/EPI WHO estimates that 453 000 child deaths occurred during 2008 due to rotavirus infection 41057 32653 39144 98621 82% of these deaths occurred
WHO Rotavirus Vaccine Update 2012 Dr. Fatima Serhan WHO/FWC/IVB/EPI WHO estimates that 453 000 child deaths occurred during 2008 due to rotavirus infection 41057 32653 39144 98621 82% of these deaths occurred
NO CHILD SHOULD DIE OF DIABETES
 NO CHILD SHOULD DIE OF DIABETES THE PROBLEM There is an estimated 1 million youth under the age of 25 with diabetes in the world. In high income countries, children with diabetes have full access to the
NO CHILD SHOULD DIE OF DIABETES THE PROBLEM There is an estimated 1 million youth under the age of 25 with diabetes in the world. In high income countries, children with diabetes have full access to the
Global Measles and Rubella Update. April 2018
 Global Measles and Rubella Update April 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep Oct
Global Measles and Rubella Update April 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep Oct
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION July August 2017 (Vol 5 issue N 4) Special issue featuring WHO/UNICEF Estimates of National Immunization Coverage (WUENIC
IMMUNIZATION VACCINE DEVELOPMENT IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION July August 2017 (Vol 5 issue N 4) Special issue featuring WHO/UNICEF Estimates of National Immunization Coverage (WUENIC
SAGE recommendations on non-specific effects of vaccines and their implementation
 SAGE recommendations on non-specific effects of vaccines and their implementation Andrea Vicari Initiative for Vaccine Research Immunization, Vaccines and Biologicals Topics Process of developing immunization
SAGE recommendations on non-specific effects of vaccines and their implementation Andrea Vicari Initiative for Vaccine Research Immunization, Vaccines and Biologicals Topics Process of developing immunization
Is the decline in neonatal mortality in northern Ghana, , associated with the decline in the age of BCG vaccination? An ecological study
 Syddansk Universitet Is the decline in neonatal mortality in northern Ghana, 1996-2012, associated with the decline in the age of vaccination? An ecological study Welaga, Paul; Debpuur, Cornelius; Aaby,
Syddansk Universitet Is the decline in neonatal mortality in northern Ghana, 1996-2012, associated with the decline in the age of vaccination? An ecological study Welaga, Paul; Debpuur, Cornelius; Aaby,
PROGRESS REPORT ON CHILD SURVIVAL: A STRATEGY FOR THE AFRICAN REGION. Information Document CONTENTS
 29 June 2009 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, 31 August 4 September 2009 Provisional agenda item 9.2 PROGRESS REPORT ON CHILD SURVIVAL: A
29 June 2009 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, 31 August 4 September 2009 Provisional agenda item 9.2 PROGRESS REPORT ON CHILD SURVIVAL: A
GRADE tables to assist guideline development and recommendations. Plain Language Summary of Results
 Seasonal Malaria Chemoprevention (formally known as Intermittent Preventive Treatment in children) for preventing malaria morbidity in children aged less than 5 years living in areas of marked seasonal
Seasonal Malaria Chemoprevention (formally known as Intermittent Preventive Treatment in children) for preventing malaria morbidity in children aged less than 5 years living in areas of marked seasonal
Evolving Realities of HIV Treatment in Resource-limited Settings
 Evolving Realities of HIV Treatment in Resource-limited Settings Papa Salif Sow MD, MSc Department of Infectious Diseases University of Dakar, Senegal Introduction: ARV access in RLS Scale-up of ART has
Evolving Realities of HIV Treatment in Resource-limited Settings Papa Salif Sow MD, MSc Department of Infectious Diseases University of Dakar, Senegal Introduction: ARV access in RLS Scale-up of ART has
WHO FCTC Global Knowledge Hub on Smokeless Tobacco
 ICMR- National Institute of Cancer Prevention & Research WHO FCTC Global Knowledge Hub on Smokeless Tobacco INTER-COUNTRY MEETING ON SMOKELESS TOBACCO POLICY 16 th -18 th AUG 2017 www.untobaccocontrol.org/kh/smokeless-tobacco/
ICMR- National Institute of Cancer Prevention & Research WHO FCTC Global Knowledge Hub on Smokeless Tobacco INTER-COUNTRY MEETING ON SMOKELESS TOBACCO POLICY 16 th -18 th AUG 2017 www.untobaccocontrol.org/kh/smokeless-tobacco/
Disparities in access: renewed focus on the underserved. Rick Johnston, WHO UNC Water and Health, Chapel Hill 13 October, 2014
 Disparities in access: renewed focus on the underserved Rick Johnston, WHO UNC Water and Health, Chapel Hill 13 October, 2014 Sector proposal for post-2015 targets By 2030: to eliminate open defecation;
Disparities in access: renewed focus on the underserved Rick Johnston, WHO UNC Water and Health, Chapel Hill 13 October, 2014 Sector proposal for post-2015 targets By 2030: to eliminate open defecation;
Update on Meningococcal A Vaccine Development and Introduction
 Update on Meningococcal A Vaccine Development and Introduction WHO Product Development for Vaccines Advisory Committee Meeting (PD-VAC) @ Geneva, 7-9 September 2015 Dr Marie-Pierre Preziosi, WHO Initiative
Update on Meningococcal A Vaccine Development and Introduction WHO Product Development for Vaccines Advisory Committee Meeting (PD-VAC) @ Geneva, 7-9 September 2015 Dr Marie-Pierre Preziosi, WHO Initiative
New and updated indicators for assessing infant and young child feeding practices. Presentation outline
 New and updated indicators for assessing infant and young child feeding practices Bernadette Daelmans (WHO) on behalf of the Working Group on IYCF indicators Presentation outline The rationale for developing
New and updated indicators for assessing infant and young child feeding practices Bernadette Daelmans (WHO) on behalf of the Working Group on IYCF indicators Presentation outline The rationale for developing
Financing malaria control
 Chapter 6. Financing malaria control The three major sources of funds for malaria control pro - grammes are national government spending, external assistance from donors and household or private out-of-pocket
Chapter 6. Financing malaria control The three major sources of funds for malaria control pro - grammes are national government spending, external assistance from donors and household or private out-of-pocket
Original article. What is already known on this topic. What this study adds
 An additional appendix is published online only. To view this fi le please visit the journal online (http://adc.bmj.com/ content/early/recent) 1 Bandim Health Project, Bissau, Guinea-Bissau 2 Bandim Health
An additional appendix is published online only. To view this fi le please visit the journal online (http://adc.bmj.com/ content/early/recent) 1 Bandim Health Project, Bissau, Guinea-Bissau 2 Bandim Health
Scaling Up Nutrition Action for Africa
 Scaling Up Nutrition Action for Africa Where are we and what challenges are need to be addressed to accelerate malnutrition? Lawrence Haddad Global Alliance for Improved Nutrition Why should African political
Scaling Up Nutrition Action for Africa Where are we and what challenges are need to be addressed to accelerate malnutrition? Lawrence Haddad Global Alliance for Improved Nutrition Why should African political
APPENDICES BACKGROUND PAPER ON THE RTS,S/AS01 MALARIA VACCINE SEPTEMBER 2015
 APPENDICES BACKGROUND PAPER ON THE RTS,S/AS01 MALARIA VACCINE SEPTEMBER 2015 PREPARED BY THE JOINT TECHNICAL EXPERT GROUP ON MALARIA VACCINES (JTEG) AND WHO SECRETARIAT 1 Appendix 1. Clinical studies conduc
APPENDICES BACKGROUND PAPER ON THE RTS,S/AS01 MALARIA VACCINE SEPTEMBER 2015 PREPARED BY THE JOINT TECHNICAL EXPERT GROUP ON MALARIA VACCINES (JTEG) AND WHO SECRETARIAT 1 Appendix 1. Clinical studies conduc
Aboubacar Kampo Chief of Health UNICEF Nigeria
 Aboubacar Kampo Chief of Health UNICEF Nigeria Many thanks to UNICEF colleagues in Supply Division-Copenhagen and NY for contributing to this presentation Thirty-five countries are responsible for 98%
Aboubacar Kampo Chief of Health UNICEF Nigeria Many thanks to UNICEF colleagues in Supply Division-Copenhagen and NY for contributing to this presentation Thirty-five countries are responsible for 98%
Prioritizing Emergency Polio Eradication Activities
 Prioritizing Emergency Polio Eradication Activities Managing the Financing Gap the other half of the Emergency Hamid Jafari GPEI Financing 2012-13: Budget = $2.23 b - Confirmed contributions = $1.14 b
Prioritizing Emergency Polio Eradication Activities Managing the Financing Gap the other half of the Emergency Hamid Jafari GPEI Financing 2012-13: Budget = $2.23 b - Confirmed contributions = $1.14 b
UNAIDS 2013 AIDS by the numbers
 UNAIDS 2013 AIDS by the numbers 33 % decrease in new HIV infections since 2001 29 % decrease in AIDS-related deaths (adults and children) since 2005 52 % decrease in new HIV infections in children since
UNAIDS 2013 AIDS by the numbers 33 % decrease in new HIV infections since 2001 29 % decrease in AIDS-related deaths (adults and children) since 2005 52 % decrease in new HIV infections in children since
SGCEP SCIE 1121 Environmental Science Spring 2012 Section Steve Thompson:
 SGCEP SCIE 1121 Environmental Science Spring 2012 Section 20531 Steve Thompson: steventhompson@sgc.edu http://www.bioinfo4u.net/ 1 First, a brief diversion... Into... how to do better on the next exam,
SGCEP SCIE 1121 Environmental Science Spring 2012 Section 20531 Steve Thompson: steventhompson@sgc.edu http://www.bioinfo4u.net/ 1 First, a brief diversion... Into... how to do better on the next exam,
Africa s slow fertility transition
 Africa s slow fertility transition John Bongaarts Population Council, New York Süssmilch Lecture Max Planck Institute, Rostock 3 Sep 215 Billions 4 3 Population projections for sub-saharan Africa 215 projection
Africa s slow fertility transition John Bongaarts Population Council, New York Süssmilch Lecture Max Planck Institute, Rostock 3 Sep 215 Billions 4 3 Population projections for sub-saharan Africa 215 projection
Global Advisory Committee on Vaccine Safety (GACVS) Report from the June 2017 meeting
 Global Advisory Committee on Vaccine Safety (GACVS) Report from the June 2017 meeting Topics discussed in June Safety monitoring of the RTS,S vaccine pilot implementation programme Safety update of BCG
Global Advisory Committee on Vaccine Safety (GACVS) Report from the June 2017 meeting Topics discussed in June Safety monitoring of the RTS,S vaccine pilot implementation programme Safety update of BCG
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005
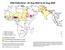 Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
POLIOMYELITIS ERADICATION: PROGRESS REPORT. Information Document CONTENTS BACKGROUND PROGRESS MADE NEXT STEPS... 12
 5 August 9 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, August 4 September 9 POLIOMYELITIS ERADICATION: PROGRESS REPORT Information Document CONTENTS
5 August 9 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, August 4 September 9 POLIOMYELITIS ERADICATION: PROGRESS REPORT Information Document CONTENTS
Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis
 EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
NO MORE MISSED MDG4 OPPORTUNITIES: OPTIMIZING EXISTING HEALTH PLATFORMS FOR CHILD SURVIVAL. Measles & Rubella Campaigns
 NO MORE MISSED MDG4 OPPORTUNITIES: OPTIMIZING EXISTING HEALTH PLATFORMS FOR CHILD SURVIVAL Measles & Rubella Campaigns With fewer than 600 days remaining to the Millennium Development Goal (MDG) deadline,
NO MORE MISSED MDG4 OPPORTUNITIES: OPTIMIZING EXISTING HEALTH PLATFORMS FOR CHILD SURVIVAL Measles & Rubella Campaigns With fewer than 600 days remaining to the Millennium Development Goal (MDG) deadline,
PREVENTION OF MENINGOCOCCAL MENINGITIS BY VACCINATION IN THE AFRICAN MENINGITIS BELT
 PREVENTION OF MENINGOCOCCAL MENINGITIS BY VACCINATION IN THE AFRICAN MENINGITIS BELT Brian Greenwood London School of Hygiene & Tropical Medicine ADVAC, Annecy May 19 th 2014 Cytoplasmic proteins bacterium
PREVENTION OF MENINGOCOCCAL MENINGITIS BY VACCINATION IN THE AFRICAN MENINGITIS BELT Brian Greenwood London School of Hygiene & Tropical Medicine ADVAC, Annecy May 19 th 2014 Cytoplasmic proteins bacterium
Requirements for new trials to examine offtarget. vaccination. Workshop on: Off-target (heterologous/non-specific) effects of vaccination.
 Requirements for new trials to examine offtarget effects of vaccination Workshop on: Off-target (heterologous/non-specific) effects of vaccination Les Pensieres Jun 8-10, 2015 PEM Fine London School of
Requirements for new trials to examine offtarget effects of vaccination Workshop on: Off-target (heterologous/non-specific) effects of vaccination Les Pensieres Jun 8-10, 2015 PEM Fine London School of
World Health organization/ International Society of Hypertension (WH0/ISH) risk prediction charts
 World Health organization/ International Society of Hypertension (WH0/ISH) risk prediction charts (charts in colour) (These charts will be updated in 2014) 2 1. Introduction 2. Instructions on how to use
World Health organization/ International Society of Hypertension (WH0/ISH) risk prediction charts (charts in colour) (These charts will be updated in 2014) 2 1. Introduction 2. Instructions on how to use
Measles in infants less than 6 months of age and effectiveness and safety of vaccination.
 Measles in infants less than 6 months of age and effectiveness and safety of vaccination. Dr. N.S.Crowcroft on behalf of the SAGE Measles and Rubella working group Member of SAGE Measles and Rubella Working
Measles in infants less than 6 months of age and effectiveness and safety of vaccination. Dr. N.S.Crowcroft on behalf of the SAGE Measles and Rubella working group Member of SAGE Measles and Rubella Working
O c t o b e r 1 0,
 STUDENT VOICES FROM THE FIELD: WORLD HEALTH ORGANIZATION GENEVA, SWITZERLAND O c t o b e r 1 0, 2 0 1 6 M o l l y P e z z u l o University at Albany School of Public Health MPH Epidemiology Candidate 17
STUDENT VOICES FROM THE FIELD: WORLD HEALTH ORGANIZATION GENEVA, SWITZERLAND O c t o b e r 1 0, 2 0 1 6 M o l l y P e z z u l o University at Albany School of Public Health MPH Epidemiology Candidate 17
Protocol Synopsis. Administrative information
 Protocol Synopsis Item (SPIRIT item no.) Administrative information Title (1) Introduction Description of research question (6a) Description An optimal schedule for the post-polio eradication era: multicentre
Protocol Synopsis Item (SPIRIT item no.) Administrative information Title (1) Introduction Description of research question (6a) Description An optimal schedule for the post-polio eradication era: multicentre
Analysis of Immunization Financing Indicators from the WHO-UNICEF Joint Reporting Form (JRF),
 Analysis of Immunization Financing Indicators from the WHO-UNICEF Joint Reporting Form (JRF), 2008-2010 Claudio Politi and Oumar Sagna Department of Immunization Vaccines and Biologicals, World Health
Analysis of Immunization Financing Indicators from the WHO-UNICEF Joint Reporting Form (JRF), 2008-2010 Claudio Politi and Oumar Sagna Department of Immunization Vaccines and Biologicals, World Health
Downloaded from:
 Theiss-Nyland, K; Lynch, M; Lines, J (2016) Assessing the availability of LLINs for continuous distribution through routine antenatal care and the Expanded Programme on Immunizations in sub- Saharan Africa.
Theiss-Nyland, K; Lynch, M; Lines, J (2016) Assessing the availability of LLINs for continuous distribution through routine antenatal care and the Expanded Programme on Immunizations in sub- Saharan Africa.
Closing the loop: translating evidence into enhanced strategies to reduce maternal mortality
 Closing the loop: translating evidence into enhanced strategies to reduce maternal mortality Washington DC March 12th 2008 Professor Wendy J Graham Opinion-based decisionmaking Evidence-based decision-making
Closing the loop: translating evidence into enhanced strategies to reduce maternal mortality Washington DC March 12th 2008 Professor Wendy J Graham Opinion-based decisionmaking Evidence-based decision-making
VIEW-hub Report: Global Vaccine Introduction and Implementation
 A report on current global access to new childhood vaccines VIEW-hub Report: Global Vaccine Introduction and Implementation December 2016 INTERNAL VERSION Developed from data in VIEW-hub www.view-hub.org
A report on current global access to new childhood vaccines VIEW-hub Report: Global Vaccine Introduction and Implementation December 2016 INTERNAL VERSION Developed from data in VIEW-hub www.view-hub.org
The building block framework: health systems and policies to save the lives of women, newborns and children
 The building block framework: health systems and policies to save the lives of women, newborns and children Coverage of effective interventions is to a large extent the result of the quality and effectiveness
The building block framework: health systems and policies to save the lives of women, newborns and children Coverage of effective interventions is to a large extent the result of the quality and effectiveness
Lessons Learned from Adverse Events and Assessment of Causal Relationships. Neal A. Halsey Johns Hopkins University
 Lessons Learned from Adverse Events and Assessment of Causal Relationships Neal A. Halsey Johns Hopkins University Lessons Learned 1000s of lessons learned Many vaccines not successful Some caused harm
Lessons Learned from Adverse Events and Assessment of Causal Relationships Neal A. Halsey Johns Hopkins University Lessons Learned 1000s of lessons learned Many vaccines not successful Some caused harm
COLD CHAIN EQUIPMENT OPTIMISATION PLATFORM (CCEOP)
 COLD CHAIN EQUIPMENT OPTIMISATION PLATFORM (CCEOP) Sushila Maharjan Senior Manager, Innovative Finance International Conference on Sustainable Cooling World Bank Washington DC - 29 November 2018 Reach
COLD CHAIN EQUIPMENT OPTIMISATION PLATFORM (CCEOP) Sushila Maharjan Senior Manager, Innovative Finance International Conference on Sustainable Cooling World Bank Washington DC - 29 November 2018 Reach
1. Africa Centre for Health and Population Studies 2. London School of Hygiene and Tropical Medicine 3. University College London
 A systematic review of the effects of interrupted antiretroviral interventions for prevention of mother-to-child transmission of HIV on maternal disease progression and survival Naidu KK 1, Mori R 2, Newell
A systematic review of the effects of interrupted antiretroviral interventions for prevention of mother-to-child transmission of HIV on maternal disease progression and survival Naidu KK 1, Mori R 2, Newell
Conclusions of the SAGE Working Group on Measles and Rubella June 2017, Geneva
 Conclusions of the SAGE Working Group on Measles and Rubella 21-22 June 2017, Geneva WHO Policy Recommendation on administration of MCV to infants
Conclusions of the SAGE Working Group on Measles and Rubella 21-22 June 2017, Geneva WHO Policy Recommendation on administration of MCV to infants
Pakistan: WHO and UNICEF estimates of immunization coverage: 2017 revision
 Pakistan: WHO and UNICEF estimates of immunization coverage: 2017 revision July 7, 2018; page 1 WHO and UNICEF estimates of national immunization coverage - next revision available July 15, 2019 data received
Pakistan: WHO and UNICEF estimates of immunization coverage: 2017 revision July 7, 2018; page 1 WHO and UNICEF estimates of national immunization coverage - next revision available July 15, 2019 data received
Health systems and HIV: advocacy. Interagency Coalition on AIDS and Development
 Health systems and HIV: Priorities for civil society advocacy Michelle Munro Interagency Coalition on AIDS and Development 1 Overview GTAG, civil society and health systems advocacy Health systems and
Health systems and HIV: Priorities for civil society advocacy Michelle Munro Interagency Coalition on AIDS and Development 1 Overview GTAG, civil society and health systems advocacy Health systems and
Selected vaccine introduction status into routine immunization
 Selected introduction status into routine infant immunization worldwide, 2003 This report summarizes the current status of national immunization schedules in 2003, as reported by Member States in the /UNICEF
Selected introduction status into routine infant immunization worldwide, 2003 This report summarizes the current status of national immunization schedules in 2003, as reported by Member States in the /UNICEF
Controlling Vaccine Preventable Diseases in the US and Global Immunization Efforts
 Controlling Vaccine Preventable Diseases in the US and Global Immunization Efforts Anne Schuchat, MD RADM, US Public Health Service Assistant Surgeon General Director, National Center for Immunization
Controlling Vaccine Preventable Diseases in the US and Global Immunization Efforts Anne Schuchat, MD RADM, US Public Health Service Assistant Surgeon General Director, National Center for Immunization
AIDS in Africa. An Update. Basil Reekie
 AIDS in Africa An Update Basil Reekie Contents General Statistics The trend of HIV in Africa Ugandan experience UNAIDS 2006 Latest African Statistics by Country HIV Intervention Light at the end of the
AIDS in Africa An Update Basil Reekie Contents General Statistics The trend of HIV in Africa Ugandan experience UNAIDS 2006 Latest African Statistics by Country HIV Intervention Light at the end of the
Age-Sex Structure for Selected African countries in the early 2000s
 Age-Sex Structure for Selected African countries in the early 2000s Abstract This paper uses data from the recent United Nations population projections to examine the changing age-sex structure in selected
Age-Sex Structure for Selected African countries in the early 2000s Abstract This paper uses data from the recent United Nations population projections to examine the changing age-sex structure in selected
#4 (Tuberculosis OR TB OR Pulmonary Tuberculosis OR Multi-Drug Resistant Tuberculosis OR MDR OR Extremely-Drug Resistant Tuberculosis or XDR)
 DATABASE Cochrane Database of Systematic Reviews DATE 15 DECEMBER 2015 #1 Safety of Crucell Ad35/ AERAS-402 OR Effectiveness of Crucell Ad35/ Aeras-402 OR Safety of MVA85A/ Aeras-485 OR Effectiveness of
DATABASE Cochrane Database of Systematic Reviews DATE 15 DECEMBER 2015 #1 Safety of Crucell Ad35/ AERAS-402 OR Effectiveness of Crucell Ad35/ Aeras-402 OR Safety of MVA85A/ Aeras-485 OR Effectiveness of
Eligibility List 2018
 The Global Fund s 2017-2022 strategy and allocation-based approach enables strategic investment to accelerate the end of HIV/AIDS, tuberculosis and malaria and build resilient and sustainable systems for
The Global Fund s 2017-2022 strategy and allocation-based approach enables strategic investment to accelerate the end of HIV/AIDS, tuberculosis and malaria and build resilient and sustainable systems for
Certificate of Immunization
 Certificate of Immunization Required of all students (Page 1 of 5) QUESTIONS? 440-775-8180 or student.health@oberlin.edu (e-mail preferred) RETURN TO: Oberlin College Student Health Services 247 W. Lorain
Certificate of Immunization Required of all students (Page 1 of 5) QUESTIONS? 440-775-8180 or student.health@oberlin.edu (e-mail preferred) RETURN TO: Oberlin College Student Health Services 247 W. Lorain
TB, BCG and other things. Chris Conlon Infectious Diseases Oxford
 TB, BCG and other things Chris Conlon Infectious Diseases Oxford Epidemiology Latent TB IGRA BCG >50/100000
TB, BCG and other things Chris Conlon Infectious Diseases Oxford Epidemiology Latent TB IGRA BCG >50/100000
HPV Vaccine Lessons Learned & New Ways Forward
 HPV Vaccine Lessons Learned & New Ways Forward The Gavi Alliance June 2016, Geneva www.gavi.org Overview 1 Background 2 Lessons learned 3 New Way Forward 2 1 Background 3 HPV Background HPV is responsible
HPV Vaccine Lessons Learned & New Ways Forward The Gavi Alliance June 2016, Geneva www.gavi.org Overview 1 Background 2 Lessons learned 3 New Way Forward 2 1 Background 3 HPV Background HPV is responsible
