BD BBL CHROMagar Staph aureus
|
|
|
- Margery Johnson
- 5 years ago
- Views:
Transcription
1 INSTRUCTIONS FOR USE READY-TO-USE PLATED MEDIA PA Rev.: Sep 2011 BD BBL CHROMagar Staph aureus INTENDED USE BBL CHROMagar Staph aureus is a selective medium for the isolation, enumeration and identification of Staphylococcus aureus from clinical and food sources. Confirmatory testing of typical isolates from clinical sources is not required. BBL CHROMagar Staph aureus (prepared plated medium) has been validated by the AOAC Research Institute under the Performance Tested Methods SM Program for the analysis of shell eggs, smoked salmon and cooked roast beef when using AOAC and ISO methods. 1,2 Confirmatory testing of mauve-colored colonies obtained from the food matrices mentioned above is required. PRINCIPLES AND EXPLANATION OF THE PROCEDURE Microbiological method. S. aureus is a well documented pathogen. It is responsible for infections ranging from superficial to systemic. 3,4 Due to the prevalence of this organism and its clinical implications, detection is of utmost importance. Staphylococcal food poisoning caused by S. aureus is one of the most common types of foodborne illness worldwide. Its detection and enumeration help provide information about the potential health hazard of food, as well as being an indicator of poor hygiene. 5 It is also recommended that this organism be used as an indicator of water quality. 6 BBL CHROMagar Staph aureus is intended for the isolation, enumeration and identification of S. aureus based on the formation of mauve-colored colonies. The addition of chromogenic substrates to the medium facilitates the differentiation of S. aureus from other organisms. An advantage BBL CHROMagar Staph aureus has over some traditional media, such as Baird- Parker Agar, is the ability to identify S. aureus in 24 h as opposed to 48 h. BBL CHROMagar Staph aureus was originally developed by A. Rambach, CHROMagar, Paris, France. BD, under a licensing agreement, has optimized this formulation utilizing proprietary intellectual property used in the manufacturing of the BBL CHROMagar Staph aureus prepared plated medium. Specially selected Difco peptones supply nutrients. The addition of selective agents inhibits the growth of gram-negative organisms, yeast and some grampositive cocci. The chromogen mix consists of artificial substrates (chromogens), which release an insoluble colored compound when hydrolyzed by specific enzymes. This facilitates the detection and differentiation of S. aureus from other organisms. S. aureus utilizes one of the chromogenic substrates, producing mauve-colored colonies. The growth of mauve-colored colonies at 24 h is considered positive for S. aureus on BBL CHROMagar Staph aureus. Bacteria other than S. aureus may utilize other chromogenic substrates resulting in blue, blue-green, or if no chromogenic substrates are utilized, natural colored colonies. *PRODUCER-SUPPLIED SAMPLES OF THIS TEST KIT MODEL WERE INDEPENDENTLY EVALUATED BY THE AOAC RESEARCH INSTITUTE AND WERE FOUND TO PERFORM TO THE PRODUCER'S SPECIFICATIONS AS STATED IN THE TEST KIT'S DESCRIPTIVE INSERT. THE PRODUCER CERTIFIES THIS KIT CONFORMS IN ALL RESPECTS TO THE SPECIFICATIONS ORIGINALLY EVALUATED BY THE AOAC RESEARCH INSTITUTE AS DETAILED IN Performance Tested Methods sm CERTIFICATE NUMBER PA Page 1 of 7
2 REAGENTS BBL CHROMagar Staph aureus Approximate Formula* Per Liter of Purified Water Chromopeptone 40.0 g Sodium Chloride 25.0 g Chromogenic Mix 0.5 g Inhibitory Agents 0.07 g Agar 14.0 g ph: 6,8 +/- 0,2 *Adjusted and/or supplemented as required to meet performance criteria. PRECAUTIONS. For professional use only. If excessive moisture is observed, invert the bottom over an off-set lid and allow to air dry in order to prevent formation of a seal between the top and bottom of the plate during incubation. Protect from light during drying. See STORAGE AND SHELF LIFE. Do not use plates if they show evidence of microbial contamination, discoloration, drying, cracking or other signs of deterioration. Pathogenic microorganisms, including hepatitis viruses and Human Immunodeficiency Virus, may be present in clinical specimens. Standard precautions 7-10 and institutional guidelines should be followed in handling all items contaminated with blood and other body fluids. Observe aseptic techniques and established precautions against microbiological hazards throughout all procedures. After use, prepared plates, specimen containers and other contaminated materials must be sterilized by autoclaving before discarding. Consult GENERAL INSTRUCTIONS FOR USE document for aseptic handling procedures, biohazards, and disposal of used product. STORAGE AND SHELF LIFE On receipt, store plates in the dark at 2 to 8 C, in their original sleeve wrapping until just prior to use. Avoid freezing and overheating. The plates may be inoculated up to the expiration date (see package label) and incubated for the recommended incubation times. Plates from opened stacks of 10 plates can be used for one week when stored in a clean area at 2 to 8 C. USER QUALITY CONTROL Check performance by inoculating a representative sample of plates with pure cultures of stable control organisms that produce known, desired reactions (for details, see GENERAL INSTRUCTIONS FOR USE document). The test strains mentioned in the Table below are recommended. Incubate aerobically for 18 to 24 hours at 35 2 C in the dark. Strains Growth Results Staphylococcus aureus ATCC Growth; mauve colonies Staphylococcus saprophyticus ATCC Growth; green to blue-green colonies Proteus mirabilis ATCC Inhibition (partial to complete) Uninoculated Colorless to light amber, clear to trace hazy Quality control requirements must be performed in accordance with applicable local, state and/or federal regulations or accreditation requirements and your laboratory s standard Quality Control procedures. It is recommended that the clinical user refer to pertinent Clinical and Laboratory Standards Institute (formerly NCCLS) guidelines for appropriate Quality Control practices. PROCEDURE Materials Provided BBL CHROMagar Staph aureus, provided in 90 mm Stacker dishes Materials Required But Not Provided: Ancillary culture media, reagents, quality control organisms and other laboratory equipment as required. PA Page 2 of 7
3 Specimen Types Refer to appropriate texts or standards for details in specimen/sample collection and handling procedures. This medium is used for the isolation of Staohylococcus aureus from all types of clinical specimens. See also PERFORMANCE CHARACTERISTICS AND LIMITATIONS OF THE PROCEDURE. For agrifood testing, follow appropriate standard methods for details on sample preparation and processing according to sample type and geographic location. Test Procedure Observe aseptic techniques. The agar surface should be smooth and moist, but without excessive moisture. For clinical specimens, as soon as possible after receipt in the laboratory, inoculate onto a BBL CHROMagar Staph aureus plate and streak for isolation. If the specimen is cultured from a swab, roll the swab gently over a small area of the surface at the edge, then streak from this area with a loop. Incubate plates aerobically at 35 ± 2 C for h in an inverted position (agar-side up). For food samples, consult appropriate references and follow applicable standard methods. Inoculate the homogenized food samples onto BBL CHROMagar Staph aureus using the spread plate technique. Incubate plates aerobically at C for h in an inverted position (agar-side up). Results After proper incubation, read plates against a white background. S. aureus will produce mauve to orange/mauve colored colonies on the BBL CHROMagar medium. Most gram-positive organisms, if not inhibited, will produce blue, blue-green or natural color (colorless, white or cream) colonies. Gram-negative organisms and yeasts are partially to completely inhibited. PERFORMANCE CHARACTERISTICS AND LIMITATIONS OF THE PROCEDURE Performance Results 12 Clinical Testing 1. In a field trial conducted at a large US metropolitan hospital, 201 throat and sputum specimens from cystic fibrosis patients and 459 nasal specimens from other hospital patients were evaluated on BBL CHROMagar Staph aureus. BBL CHROMagar Staph aureus was compared to blood agar or Mannitol Salt Agar, with isolate confirmation by slide coagulase. S. aureus was recovered from 190 combined specimens. BBL CHROMagar Staph aureus detected 9 additional S. aureus positive cultures which were not recovered on conventional media. Four potential false positives were also observed on the BBL CHROMagar Staph aureus medium following 24 h incubation: two corynebacteria and two coagulase-negative staphylococci. BBL CHROMagar Staph aureus produced an overall sensitivity of 99.5% and a specificity of 99.2% In a European study, one hundred sixty five (165) specimens from a routine lab, consisting of 100 specimens shown to contain S. aureus by standard methods (= known positive specimens) and 65 known negative specimens, were streaked on CHROMagar Staph aureus, Mannitol Salt Agar and Columbia Agar with 5% Sheep Blood. The specimen types are shown in Table 1. Plates were incubated for 20 to 24 hours at 35 to 37 C and were read for colonies suspicious of S. aureus. Tube coagulase tests were set up from all suspicious colonies on all three media. Also, the amount of S.aureus growth (scored semi-quantitatively), was determined. Table 1: Specimen Types Specimen Types S. aureus known positive specimens Abscesses 14 6 Ascites fluid 1 0 Bone 1 0 Bursa 1 0 PA Page 3 of 7 S. aureus known negative specimens
4 Catheter 4 0 Drainage 1 0 Fistula 0 1 Surgery specimens Miscellaneous swabs Phlegmona 1 0 Tracheal secretions 1 0 Wounds Total Results: Of the 165 specimens, 100 yielded growth of S. aureus on any medium, as confirmed by coagulase testing from all media. On CHROMagar Staph aureus, 100 specimens yielded growth of S. aureus; on Mannitol Salt Agar, 91 yielded S. aureus; on Columbia Agar together with coagulase testing, 98 specimens were positive for S. aureus. There was one false positive on CHROMagar Staph aureus that turned out to be Streptococcus agalactiae. Upon restreaking the strain on CHROMagar Staph aureus, the colonies were violet rather than rose to mauve. Among the known negative specimens, there were 5 cultures with violet or lilac colonies which were similar to S. aureus in color. However, they could be easily differentiated from S. aureus colonies (=rose to mauve) by the reader who was not too familiar with the colony colors of the different species growing on the medium (only a summary of colors and organisms occurring on the medium had been given to her before the study was begun). The sensitivities of CHROMagar Staph aureus (based on rose to mauve colony color), Mannitol Salt Agar (based on colonies surrounded by yellow medium), and Columbia Agar (growth of typical S. aureus colonies together with coagulase testing) were 100%, 91%, and 98%. The specificity of CHROMagar Staph aureus was 98.5%. On CHROMagar Staph aureus, the growth intensity was ly more frequently higher than on MSA (p= 0.05); see Table 2. CHROMagar Staph aureus can be used for a wide variety of specimens. Table 2: Growth intensity of S. aureus isolates on CSA* as compared to MSA* Growth intensity CSA = Col CSA> Col Col>CSA CSA=MSA CSA>MSA MSA>CSA Number of specimens ** 8 * CSA, CHROMagar Staph aureus; MSA, Mannitol Salt Agar; Col, Columbia Agar with 5% Sheep Blood ** difference statistically (p= 0.05) Agrifood Testing BBL CHROMagar Staph aureus was validated by the AOAC Research Institute under the Performance Tested Methods Program. The medium was evaluated by an external reference laboratory, as well as internally at BD, for the recovery and enumeration of S. aureus in cooked roast beef, smoked salmon and shell eggs. The recovery and enumeration of S. aureus on BBL CHROMagar Staph aureus was compared to the AOAC and ISO reference plated medium, Baird-Parker Agar, using the recommended diluents at low, medium and high inoculum levels of S. aureus. After 24 h incubation, enumeration was performed on BBL CHROMagar Staph aureus and after 48 h on Baird-Parker Agar. Based on statistical analysis, no difference was found between the reference methods and the BBL CHROMagar Staph aureus method for any food type or contamination level, with the exception of a low-level smoked salmon sample. The low contamination level of smoked salmon demonstrated a statistical difference in internal testing using the ISO method; i.e., the BBL CHROMagar Staph aureus method at 24 h recovered more colonies (log 10 = 2.04) than the ISO reference at 48 h (log 10 = 1.64). The repeatability precision estimates of the BBL CHROMagar Staph aureus method were satisfactory. The correlation coefficients ranged from 92.6% to 99.4%, demonstrating good correlation for all contamination levels in all food types. Data PA Page 4 of 7
5 is summarized in Tables 1 and 2. No false-positive colonies were recovered from the food matrices using BBL CHROMagar Staph aureus. All mauve colonies were confirmed as S. aureus with no discrepancies. Additionally, 30 strains of S. aureus, including known enterotoxin-producing strains, and 37 non-s. aureus isolates were evaluated producing both a sensitivity and specificity of 100% on BBL CHROMagar Staph aureus. 11 Table 1. Summary of AOAC and ISO External Testing of Cooked Roast Beef and Shell Eggs 1-3 AOAC Inoculum Level Paired t-test or One-way ANOVA a Repeatability (standard deviation) b Low Not Cooked Medium Not 0.04 Roast Beef High Not Low Not Shell Eggs Medium Not High Not ISO Inoculum Level Paired t-test or Repeatability One-way ANOVA a (standard deviation) b Low Not Cooked Medium Not Roast Beef High Not Low Not Shell Eggs Medium Not High Not Footnotes see Table 2 Square of linear correlation coefficient 96.0% 95.5% Square of linear correlation coefficient c 94.6% 92.6% Table 2. Summary of AOAC and ISO External and Internal Testing of Smoked Salmon Inoculum Level Paired t-test or One-way ANOVA a Repeatability (standard deviation) b Square of linear correlation coefficient c Low Not Not AOAC Medium Not Not % 93.2% Smoked Salmon High Not Not Low Not Significant d Medium Not Not ISO Smoked salmon High Not Not % 97.4% a Paired t-test and one-way ANOVA analysis used to evaluate comparable performance of BBL CHROMagar Staph aureus versus the reference medium by comparing the mean of the log 10 of the colony counts. b Repeatability demonstrates BBL CHROMagar Staph aureus produces comparable results between the tests run on the same material and method. c Square of linear correlation coefficient is used to evaluate precision of quantitative methods over different S. aureus counts. d BBL CHROMagar Staph aureus recovered more colonies than the ISO reference method. Limitations of the Procedure Occasionally some strains of staphylococci, other than S. aureus, such as: S. cohnii, S. intermedius, and S. schleiferi, as well as corynebacteria and yeasts, may produce mauvecolored colonies at 24 h. 11 Differentiation of S. aureus from non-s. aureus can be accomplished by coagulase, other biochemicals or Gram stain. Resistant gram-negative bacilli, which typically appear as small blue colonies, may also break through. PA Page 5 of 7
6 Incubation beyond 24 h (clinical) and 28 h (food) is not recommended due to an increase in potential false positives. If incubation time is exceeded, mauve-colored colonies should be confirmed prior to reporting as S. aureus. Incubation less than the recommended 20 h may result in a lower percentage of correct results being obtained. Due to the natural golden pigment of some S. aureus strains, colony color may appear orangemauve. REFERENCES 1. AOAC Official Method Staphylococcus aureus in foods. Surface plating method for isolation and enumeration International Organization for Standards (ISO). Microbiology of food and animal feeding stuffs - Horizontal method for the enumeration of coagulase-positive Staphylococci (Staphylococcus aureus and other species) - Part 1: Technique using Baird Parker agar medium, 1st ed., ISO : Jablonski, L.M. and G.A. Bohach Staphylococcus aureus. In M. Doyle, L. Beuchat and T. Montville (eds.), Food microbiology fundamentals and frontiers. ASM, Washington, DC. 4. Bannerman, T.L Staphylococcus, Micrococcus, and other catalase-positive cocci that grow aerobically. In P.R. Murray, E.J. Baron, J.H. Jorgensen, M.A. Pfaller, and R.H. Yolken (eds.), Manual of clinical microbiology, 8 th edition. ASM, Washington DC. 5. Bennett, R.W. and G.A. Lancette Staphylococcus aureus. In FDA, Bacteriological analytical manual, 8th ed. AOAC International, Gaithersburg, MD. 6. Toranzos, G.A., G.A. McFeters and J.J. Borrego Detection of microorganisms in environmental freshwaters and drinking waters. In C. Hurst, R. Crawford, G. Knudsen, M. McInerney and L. Stetzenbach (eds.), Manual of environmental microbiology, 2nd ed. ASM Press, Washington, DC. 7. Clinical and Laboratory Standards Institute (CLSI, formerly NCCLS). Approved Guideline M29. Protection of laboratory workers from occupationally acquired infections.search for latest version at 8. Garner, J.S Hospital Infection Control Practices Advisory Committee, U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Guideline for isolation precautions in hospitals. Infect. Control Hospital Epidemiol. 17: U.S. Department of Health and Human Services Biosafety in microbiological and biomedical laboratories, HHS Publication (CDC), 4th ed. U.S. Government Printing Office, Washington, DC. 10. Directive 2000/54/EC of the European Parliament and of the Council of 18 September 2000 on the protection of workers from risks related to exposure to biological agents at work (seventh individual directive within the meaning of Article 16(1) of Directive 89/391/EEC). Official Journal L262, 17/10/2000, p Data on file, BD Diagnostic Systems. PACKAGING/AVAILABILITY REF Ready-to-use Plated Media, cpu 20 REF Ready-to-use Plated Media, cpu 120 FURTHER INFORMATION For further information please contact your local BD representative. Becton Dickinson GmbH Tullastrasse 8 12 D Heidelberg/Germany Phone: Fax: Reception_Germany@europe.bd.com PA Page 6 of 7
7 AOAC is a trademark and Performance Tested Methods is a service mark of AOAC International. CHROMagar is a trademark of Dr. A. Rambach. Difco is a trademark of Difco Laboratories, Inc., subsidiary of Becton, Dickinson and Company. ATCC is a trademark of the American Type Culture Collection BD, BD Logo and all other trademarks are property of Becton, Dickinson and Company BD PA Page 7 of 7
Petrifilm. Staph Express Count Plate. Interpretation Guide
 3 Interpretation Guide Staph Express Count Plate This guide familiarizes you with results on 3M Staph Express Count Plates. For more information, contact the 3M Microbiology representative nearest you.
3 Interpretation Guide Staph Express Count Plate This guide familiarizes you with results on 3M Staph Express Count Plates. For more information, contact the 3M Microbiology representative nearest you.
Count. Enterobacteriaceae. Interpretation Guide. 3M Food Safety 3M Petrifilm Enterobacteriaceae Count Plate
 3M Food Safety 3M Petrifilm Enterobacteriaceae Count Plate Enterobacteriaceae Count Interpretation Guide This guide familiarizes you with results on 3M Petrifilm Enterobacteriaceae Count Plates. For more
3M Food Safety 3M Petrifilm Enterobacteriaceae Count Plate Enterobacteriaceae Count Interpretation Guide This guide familiarizes you with results on 3M Petrifilm Enterobacteriaceae Count Plates. For more
β CARBA Test Rapid detection of carbapenemase-producing Enterobacteriaceae strains Contents 1. INTENDED USE
 β CARBA Test 25 68260 Rapid detection of carbapenemase-producing Enterobacteriaceae strains 881159 2015/05 Contents 1. INTENDED USE 2. SUMMARY AND EXPLANATION OF THE TEST 3. PRINCIPLE OF THE PROCEDURE
β CARBA Test 25 68260 Rapid detection of carbapenemase-producing Enterobacteriaceae strains 881159 2015/05 Contents 1. INTENDED USE 2. SUMMARY AND EXPLANATION OF THE TEST 3. PRINCIPLE OF THE PROCEDURE
Cotinine (Mouse/Rat) ELISA Kit
 Cotinine (Mouse/Rat) ELISA Kit Catalog Number KA2264 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Cotinine (Mouse/Rat) ELISA Kit Catalog Number KA2264 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
FOOD SAFETY. VIDAS Staph enterotoxin II (SET2) Ultra Performance Summary CONFIDENCE DECIDE WITH
 FOOD DECIDE WITH SAFETY CONFIDENCE VIDAS Staph enterotoxin II (SET2) Ultra Performance Summary Food safety professionals have a variety of test kits to choose from when looking to fill a need in their
FOOD DECIDE WITH SAFETY CONFIDENCE VIDAS Staph enterotoxin II (SET2) Ultra Performance Summary Food safety professionals have a variety of test kits to choose from when looking to fill a need in their
Interpretation Guide. Enterobacteriaceae Count Plate
 Interpretation Guide The 3M Petrifilm Enterobacteriaceae Count Plate is a sample-ready-culture medium system that contains modified Violet Red Bile Glucose (VRBG) nutrients, a cold-watersoluble gelling
Interpretation Guide The 3M Petrifilm Enterobacteriaceae Count Plate is a sample-ready-culture medium system that contains modified Violet Red Bile Glucose (VRBG) nutrients, a cold-watersoluble gelling
HyServe Compact Dry X-SA Method for the Enumeration of Staphylococcus aureus in Foods NordVal No: 042. HyServe Compact Dry X-SA
 c/o Danish Technical University Kemitorvet B201 DK-2800 Kgs Lyngby, Denmark www.nmkl.org Issued for: HyServe Compact Dry X-SA Method for the Enumeration of Staphylococcus aureus in Foods NordVal No: 042
c/o Danish Technical University Kemitorvet B201 DK-2800 Kgs Lyngby, Denmark www.nmkl.org Issued for: HyServe Compact Dry X-SA Method for the Enumeration of Staphylococcus aureus in Foods NordVal No: 042
Strep-a-Test Twister Test
 Strep-a-Test Twister Test Code: 24524 A rapid test for the qualitative detection of Strep A antigen in throat swab specimens. For professional in vitro diagnostic use only. INTENDED USE The Strep A Twist
Strep-a-Test Twister Test Code: 24524 A rapid test for the qualitative detection of Strep A antigen in throat swab specimens. For professional in vitro diagnostic use only. INTENDED USE The Strep A Twist
USE: Isolation and differentiation of Gram (-) enteric bacilli (MAC) / Coliform Testing / Recovery of Stressed Coliforms (EMB)
 MAC/EMB Code 5544 MacConkey Agar (MAC) Eosin Methylene Blue Agar (EMB) USE: Isolation and differentiation of Gram (-) enteric bacilli (MAC) / Coliform Testing / Recovery of Stressed Coliforms (EMB) Side
MAC/EMB Code 5544 MacConkey Agar (MAC) Eosin Methylene Blue Agar (EMB) USE: Isolation and differentiation of Gram (-) enteric bacilli (MAC) / Coliform Testing / Recovery of Stressed Coliforms (EMB) Side
ENG MYCO WELL D- ONE REV. 1.UN 29/09/2016 REF. MS01283 REF. MS01321 (COMPLETE KIT)
 ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
See external label 96 tests HSV 2 IgA. Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Impact of Sodium Reduction on Survival of Listeria monocytogenes in Sliced Process Cheese
 Impact of Sodium Reduction on Survival of Listeria monocytogenes in Sliced Process Cheese July 2013 By: Dr. Francisco Diez Gonzalez University of Minnesota Dr. Mastura Akhtar Partners: Midwest Dairy Association
Impact of Sodium Reduction on Survival of Listeria monocytogenes in Sliced Process Cheese July 2013 By: Dr. Francisco Diez Gonzalez University of Minnesota Dr. Mastura Akhtar Partners: Midwest Dairy Association
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
320 MBIO Microbial Diagnosis. Aljawharah F. Alabbad Noorah A. Alkubaisi 2017
 320 MBIO Microbial Diagnosis Aljawharah F. Alabbad Noorah A. Alkubaisi 2017 Pathogens of the Urinary tract The urinary system is composed of organs that regulate the chemical composition and volume of
320 MBIO Microbial Diagnosis Aljawharah F. Alabbad Noorah A. Alkubaisi 2017 Pathogens of the Urinary tract The urinary system is composed of organs that regulate the chemical composition and volume of
Microbiology products. Liofilchem Chromatic
 ET Microbiology products M IC R O PL A N www.liofilchem.net Liofilchem Chromatic c h ro m o g e n i c c u l t u re m e d i a LIOFILCHEM s.r.l. Via Scozia, Zona Industriale 64026 Roseto degli Abruzzi (Te)
ET Microbiology products M IC R O PL A N www.liofilchem.net Liofilchem Chromatic c h ro m o g e n i c c u l t u re m e d i a LIOFILCHEM s.r.l. Via Scozia, Zona Industriale 64026 Roseto degli Abruzzi (Te)
NOVASTREAK. Microbial Contamination Monitoring Device TYPICAL CULTURAL MORPHOLOGY Baird Parker Agar. S. aureus growth on Baird Parker Agar
 NOVASTREAK Microbial Contamination Monitoring Device TYPICAL CULTURAL MORPHOLOGY Baird Parker Agar S. aureus growth on Baird Parker Agar Baird Parker Agar is used for the selective isolation and enumeration
NOVASTREAK Microbial Contamination Monitoring Device TYPICAL CULTURAL MORPHOLOGY Baird Parker Agar S. aureus growth on Baird Parker Agar Baird Parker Agar is used for the selective isolation and enumeration
Human Influenza A (Swine Flu) Rapid test
 Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Rapid-VIDITEST. Strep A Card. One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
DIAGNOSTIC AUTOMATION, INC.
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Petrifilm. Enterobacteriaceae Count Plate. Interpretation Guide
 3 Interpretation Guide Petrifilm Enterobacteriaceae Count Plate This guide familiarizes you with results on 3M Petrifilm Enterobacteriaceae Count plates. For more information contact the official 3M Microbiology
3 Interpretation Guide Petrifilm Enterobacteriaceae Count Plate This guide familiarizes you with results on 3M Petrifilm Enterobacteriaceae Count plates. For more information contact the official 3M Microbiology
Herpes Simplex Virus 2 IgM HSV 2 IgM
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Herpes Simplex Virus 2 IgG HSV 2 IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Medical Microbiology
 Lecture 5!!!!!!ƒš!!Œ!!! š!!œ!! Œ!!!! Dr. Ismail I. Daood Medical Microbiology!! Systematic Bacteriology Gram-Positive Cocci : GENUS : Staphylococcus : The general properties of Staphylococcus are Gram-
Lecture 5!!!!!!ƒš!!Œ!!! š!!œ!! Œ!!!! Dr. Ismail I. Daood Medical Microbiology!! Systematic Bacteriology Gram-Positive Cocci : GENUS : Staphylococcus : The general properties of Staphylococcus are Gram-
A.F. GENITAL SYSTEM. ITEMS NECESSARY BUT NOT INCLUDED IN THE KIT A.F. GENITAL SYSTEM Reagent (ref ) Mycoplasma Transport Broth (ref.
 A.F. GENITAL SYSTEM ENGLISH System for detection, count and susceptibility test of pathogenic urogenital microorganisms DESCRIPTION A.F. GENITAL SYSTEM is a 24-well system containing desiccated biochemical
A.F. GENITAL SYSTEM ENGLISH System for detection, count and susceptibility test of pathogenic urogenital microorganisms DESCRIPTION A.F. GENITAL SYSTEM is a 24-well system containing desiccated biochemical
HBeAg and HBeAg Ab ELISA Kit
 HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
Rapid-VIDITEST. Strep A Blister. One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Rapid-VIDITEST Swine Flu
 Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
EXPERIMENT. Food Safety
 EXPERIMENT Food Safety Hands-On Labs, Inc. Version 42-0308 -00-01 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to organize
EXPERIMENT Food Safety Hands-On Labs, Inc. Version 42-0308 -00-01 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to organize
Toxoplasma gondii IgM (Toxo IgM)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test
 Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test Complexity: WAIVED for fingerstick whole blood and venipuncture whole blood. A Certificate of CLIA Waiver is required to perform the test
Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test Complexity: WAIVED for fingerstick whole blood and venipuncture whole blood. A Certificate of CLIA Waiver is required to perform the test
MALDI Sepsityper Kit
 Instructions for Use MALDI Sepsityper Kit Kit for identification of microorganisms from positive blood cultures using the MALDI Biotyper system CARE products are designed to support our worldwide customers
Instructions for Use MALDI Sepsityper Kit Kit for identification of microorganisms from positive blood cultures using the MALDI Biotyper system CARE products are designed to support our worldwide customers
See external label 2 C-8 C 96 tests Chemiluminescence. CMV IgM. Cat # Diluted samples, controls & calibrator 100 µl 30 minutes
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Petrifilm Enterobacteriaceae Count Plate
 Petrifilm Enterobacteriaceae Count Plate Interpretation Guide 1 2 1 2 Enterobacteriaceae count = 1 It is easy to count Enterobacteriaceae colonies on a Petrifilm Enterobacteriaceae count plate. A red indicator
Petrifilm Enterobacteriaceae Count Plate Interpretation Guide 1 2 1 2 Enterobacteriaceae count = 1 It is easy to count Enterobacteriaceae colonies on a Petrifilm Enterobacteriaceae count plate. A red indicator
Microbiological Methods V-A- 1 SALMONELLA SPECIES PRESUMPTIVE AND CONFIRMATION TESTS
 Microbiological Methods V-A- 1 PRESUMPTIVE AND CONFIRMATION TESTS PRINCIPLE SCOPE Enrichment and selective procedures are used to provide a reasonably sensitive, definitive and versatile means of qualitatively
Microbiological Methods V-A- 1 PRESUMPTIVE AND CONFIRMATION TESTS PRINCIPLE SCOPE Enrichment and selective procedures are used to provide a reasonably sensitive, definitive and versatile means of qualitatively
Puritan Fecal Opti- Swab Collection and Transport System
 Puritan Fecal Opti- Swab Collection and Transport System Puritan Medical Products Co. LLC P.O. Box 149, 31 School Street Guilford, Maine, USA 04443-0149 Tel: 800-321- 2313 (US and Canada) 207-876- 3311
Puritan Fecal Opti- Swab Collection and Transport System Puritan Medical Products Co. LLC P.O. Box 149, 31 School Street Guilford, Maine, USA 04443-0149 Tel: 800-321- 2313 (US and Canada) 207-876- 3311
Human Cytomegalovirus IgM ELISA Kit
 Human Cytomegalovirus IgM Catalog No: IRAPKT2012 ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgM ELISA is intended for use in the detection of IgM antibodies to Cytomegalovirus (CMV) infection in human
Human Cytomegalovirus IgM Catalog No: IRAPKT2012 ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgM ELISA is intended for use in the detection of IgM antibodies to Cytomegalovirus (CMV) infection in human
CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol
 CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol (Cat. No.:EK-310-91) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE The
CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol (Cat. No.:EK-310-91) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE The
REGIONAL STANDARD 1: DRAFT CASSAVA FLOUR - Specifications. Latest edition October 1, Reference number CARSPS 1: 2017
 REGIONAL STANDARD 1: 2017 Latest edition October 1, 2017 DRAFT CASSAVA FLOUR - Specifications Reference number CARSPS 1: 2017 CAHFSA 2017 Section Contents Page 0.0 Foreword 3 1.0 Scope 3 2.0 Normative
REGIONAL STANDARD 1: 2017 Latest edition October 1, 2017 DRAFT CASSAVA FLOUR - Specifications Reference number CARSPS 1: 2017 CAHFSA 2017 Section Contents Page 0.0 Foreword 3 1.0 Scope 3 2.0 Normative
SAMPLE PROCEDURE , 07/11
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
NOTE: Poor growth and a weak esculin reaction may be seen after 40 hours of incubation for some enterococci.
 LIS/EMB Code 5542 COMING SOON! BioPaddles Colony Identification App Listeria Agar (LIS) Eosin Methylene Blue Agar (EMB) USE: Enumeration and selective isolation of Listeria spp.(lis) Isolation and differentiation
LIS/EMB Code 5542 COMING SOON! BioPaddles Colony Identification App Listeria Agar (LIS) Eosin Methylene Blue Agar (EMB) USE: Enumeration and selective isolation of Listeria spp.(lis) Isolation and differentiation
See external label 2 C-8 C 96 tests CHEMILUMINESCENCE. CMV IgG. Cat # Step (20-25 C Room temp.) Volume
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
APPLICATION Detection and isolation of pathogenic intestinal bacteria including Shigella and Salmonella from surfaces, food, or liquid samples.
 HEK/SS Code 5543 COMING SOON! BioPaddles Colony Identification App Hektoen Enteric Agar (HEK) Salmonella Shigella Agar (SS) USE: Detection and isolation of pathogenic intestinal bacteria including Shigella
HEK/SS Code 5543 COMING SOON! BioPaddles Colony Identification App Hektoen Enteric Agar (HEK) Salmonella Shigella Agar (SS) USE: Detection and isolation of pathogenic intestinal bacteria including Shigella
TANZANIA BUREAU OF STANDARDS
 TBS/AFDC 14 (5259) P3 Ghee Specification DRAFT TANZANIA STANDARD TANZANIA BUREAU OF STANDARDS Ghee Specification 0 FOREWORD Ghee is a milk product obtained from butter or cream. In Tanzania manufacture
TBS/AFDC 14 (5259) P3 Ghee Specification DRAFT TANZANIA STANDARD TANZANIA BUREAU OF STANDARDS Ghee Specification 0 FOREWORD Ghee is a milk product obtained from butter or cream. In Tanzania manufacture
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Rubella virus IgG ELISA Kit
 Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Labquality External Quality Assesment Programmes General Bacteriology 1 1/2010
 Labquality External Quality Assesment Programmes General Bacteriology 1 1/2010 Photos and text: Markku Koskela, M.D., Ph.D. Clinical microbiology specialist Oulu, Finland Sample 1/2010 Pus from an infected
Labquality External Quality Assesment Programmes General Bacteriology 1 1/2010 Photos and text: Markku Koskela, M.D., Ph.D. Clinical microbiology specialist Oulu, Finland Sample 1/2010 Pus from an infected
Helicobacter pylori IgA ELISA Kit
 Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
BACTERIAL EXAMINATION OF WATER
 BACTERIAL EXAMINATION OF WATER The bacteriological examination of water is performed routinely by water utilities and many governmental agencies to ensure a safe supply of water for drinking, bathing,
BACTERIAL EXAMINATION OF WATER The bacteriological examination of water is performed routinely by water utilities and many governmental agencies to ensure a safe supply of water for drinking, bathing,
Procine sphingomyelin ELISA Kit
 Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Mycoplasma pneumoniae IgG ELISA Kit
 Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
HOSPITAL INFECTION CONTROL
 HOSPITAL INFECTION CONTROL Objectives To be able to define hospital acquired infections discuss the sources and routes of transmission of infections in a hospital describe methods of prevention and control
HOSPITAL INFECTION CONTROL Objectives To be able to define hospital acquired infections discuss the sources and routes of transmission of infections in a hospital describe methods of prevention and control
GB Translated English of Chinese Standard: GB NATIONAL STANDARD OF THE
 Translated English of Chinese Standard: GB4789.10-2016 www.chinesestandard.net Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 4789.10-2016 National food safety standard
Translated English of Chinese Standard: GB4789.10-2016 www.chinesestandard.net Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 4789.10-2016 National food safety standard
BChek Strep A Test (Dipstick) For detection of group A streptococcal antigen directly from throat swabs / /04 INTENDED USE The BD Chek St
 Revisions Rev from Rev to ECO # NEW 01 2742-04 SO 0191-5 Notes: 1. BD Cat. Number 256018 2. Blank (Sheet) Size : Length: 7.5 Width: 18 Number of Pages: 8 Number of Sheets: 1 Page Size: Length 7.5 Width
Revisions Rev from Rev to ECO # NEW 01 2742-04 SO 0191-5 Notes: 1. BD Cat. Number 256018 2. Blank (Sheet) Size : Length: 7.5 Width: 18 Number of Pages: 8 Number of Sheets: 1 Page Size: Length 7.5 Width
The composition can be adjusted / supplemented in order to achieve optimal performance.
 TECHNICAL DATA SHEET OXFORD AGAR DETECTION OF LISTERIA 1 INTENDED USE Oxford Agar is a selective medium used for the differentiation, the isolation and the enumeration of Listeria monocytogenes from milk
TECHNICAL DATA SHEET OXFORD AGAR DETECTION OF LISTERIA 1 INTENDED USE Oxford Agar is a selective medium used for the differentiation, the isolation and the enumeration of Listeria monocytogenes from milk
1430 West McCoy Lane Santa Maria, CA p:
 091217TR HardyCHROM BluEcoli 1 HardyCHROM Candida 2 HardyCHROM ECC 3 HardyCHROM ESBL 4 HardyCHROM Listeria 5 HardyCHROM MRSA 6 HardyCHROM O157 7 HardyCHROM Salmonella 8 HardyCHROM SS NoPRO 9 HardyCHROM
091217TR HardyCHROM BluEcoli 1 HardyCHROM Candida 2 HardyCHROM ECC 3 HardyCHROM ESBL 4 HardyCHROM Listeria 5 HardyCHROM MRSA 6 HardyCHROM O157 7 HardyCHROM Salmonella 8 HardyCHROM SS NoPRO 9 HardyCHROM
PRESENTER: DENNIS NYACHAE MOSE KENYATTA UNIVERSITY
 18/8/2016 SOURCES OF MICROBIAL CONTAMINANTS IN BIOSAFETY LABORATORIES IN KENYA PRESENTER: DENNIS NYACHAE MOSE KENYATTA UNIVERSITY 1 INTRODUCTION Contamination occurs through avoidable procedural errors
18/8/2016 SOURCES OF MICROBIAL CONTAMINANTS IN BIOSAFETY LABORATORIES IN KENYA PRESENTER: DENNIS NYACHAE MOSE KENYATTA UNIVERSITY 1 INTRODUCTION Contamination occurs through avoidable procedural errors
Measles IgM ELISA Kit
 Measles IgM ELISA Kit Catalog Number KA2257 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Measles IgM ELISA Kit Catalog Number KA2257 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
EXPERIMENT. Fomite Transmission. Hands-On Labs, Inc. Version
 EXPERIMENT Fomite Transmission Hands-On Labs, Inc. Version 42-0243-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to
EXPERIMENT Fomite Transmission Hands-On Labs, Inc. Version 42-0243-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to
Rapid-VIDITEST. Influenza A+B
 Rapid-VIDITEST Influenza A+B (One step Influenza A+B blister Test for the detection of Influenza type A and type B from nasal swabs, nasal wash or nasal aspirate specimens). Instruction manual Producer:
Rapid-VIDITEST Influenza A+B (One step Influenza A+B blister Test for the detection of Influenza type A and type B from nasal swabs, nasal wash or nasal aspirate specimens). Instruction manual Producer:
Human Cytomegalovirus Virus (CMV) IgG ELISA Kit
 Human Cytomegalovirus Virus Catalog No: IRAPKT1410 (CMV) IgG ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgG ELISA is intended for use in evaluating a patient s serologic status to cytomegalovirus (CMV)
Human Cytomegalovirus Virus Catalog No: IRAPKT1410 (CMV) IgG ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgG ELISA is intended for use in evaluating a patient s serologic status to cytomegalovirus (CMV)
Triiodothyronine (T3) ELISA
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Triiodothyronine (T3) ELISA Kit is intended for the detection of total T3 in human serum or plasma. For research
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Triiodothyronine (T3) ELISA Kit is intended for the detection of total T3 in human serum or plasma. For research
Rapid-VIDITEST. Influenza A
 Rapid-VIDITEST Influenza A (One step Influenza A Card test for the detection of Influenza type A antigen from human nasopharyngeal specimens (swab, nasopharyngeal wash and aspirate). Instruction manual
Rapid-VIDITEST Influenza A (One step Influenza A Card test for the detection of Influenza type A antigen from human nasopharyngeal specimens (swab, nasopharyngeal wash and aspirate). Instruction manual
LESSON 2.6 WORKBOOK Diagnosing infections, and, what s up your nose?
 Staphylococcus aureus Morphology: The physical form or structure of a microbe.. LESSON 2.6 WORKBOOK Diagnosing infections, and, what s up your nose? Now we have discussed the different requirements that
Staphylococcus aureus Morphology: The physical form or structure of a microbe.. LESSON 2.6 WORKBOOK Diagnosing infections, and, what s up your nose? Now we have discussed the different requirements that
AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS
 CLARITY Strep A Dipsticks FOR LABORATORY AND PROFESSIONAL USE AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS CLIA COMPLEXITY: Waived CLARITY Strep A Dipsticks:
CLARITY Strep A Dipsticks FOR LABORATORY AND PROFESSIONAL USE AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS CLIA COMPLEXITY: Waived CLARITY Strep A Dipsticks:
25(OH) Vitamin D ELISA (BD-220BA), 192 Tests
 INTENDED USE The 25-hydroxy (25-OH) Vitamin D ELISA is intended for the quantitative determination of total 25-OH Vitamin D in human serum and Plasma. SUMMARY AND EXPLANATION Vitamin D is a steroid hormone
INTENDED USE The 25-hydroxy (25-OH) Vitamin D ELISA is intended for the quantitative determination of total 25-OH Vitamin D in human serum and Plasma. SUMMARY AND EXPLANATION Vitamin D is a steroid hormone
CLIA Complexity: WAIVED
 CLIA Complexity: WAIVED INTENDED USE The is intended for the rapid, qualitative detection of Group A Streptococcal antigen from throat swabs or confirmation of presumptive Group A Streptococcal colonies
CLIA Complexity: WAIVED INTENDED USE The is intended for the rapid, qualitative detection of Group A Streptococcal antigen from throat swabs or confirmation of presumptive Group A Streptococcal colonies
Mouse/Rat THYROXINE (T4) ELISA Catalog No (96 Tests)
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Mouse/Rat Thryroxine Kit is intended for the detection of total T4 in mouse/rat serum or plasma. SUMMARY AND EXPLANATION
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Mouse/Rat Thryroxine Kit is intended for the detection of total T4 in mouse/rat serum or plasma. SUMMARY AND EXPLANATION
Anthrax protective antigen IgG ELISA Kit
 Anthrax protective antigen IgG ELISA Kit Catalog Number KA0953 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3
Anthrax protective antigen IgG ELISA Kit Catalog Number KA0953 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3
Draft for comments only Not to be cited as East African Standard
 EAST AFRICAN STANDARD Albumen flakes of non-edible quality Specification EAST AFRICAN COMMUNITY CD/K/608:2010 ICS 67.120.20 EAC 2010 First Edition 2010 Foreword Development of the East African Standards
EAST AFRICAN STANDARD Albumen flakes of non-edible quality Specification EAST AFRICAN COMMUNITY CD/K/608:2010 ICS 67.120.20 EAC 2010 First Edition 2010 Foreword Development of the East African Standards
Rubella Latex Agglutination Test
 Rubella Latex Agglutination Test Cat. No.:DLAT1088 Pkg.Size:30T Intended use The Rubella Latex Agglutination Test is a rapid latex particle agglutination test for the qualitative and semi-quantitative
Rubella Latex Agglutination Test Cat. No.:DLAT1088 Pkg.Size:30T Intended use The Rubella Latex Agglutination Test is a rapid latex particle agglutination test for the qualitative and semi-quantitative
Oral Specimen Collection Device
 Oral Specimen Collection Device For In Vitro Diagnostic Use NAME AND INTENDED USE The Intercept Oral Specimen Collection Device is intended for use in the collection, preservation and transport of oral
Oral Specimen Collection Device For In Vitro Diagnostic Use NAME AND INTENDED USE The Intercept Oral Specimen Collection Device is intended for use in the collection, preservation and transport of oral
Insulin (Porcine/Canine) ELISA
 Insulin (Porcine/Canine) ELISA For the quantitative measurement of insulin in Porcine/Canine serum and plasma (EDTA) For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 80-INSPO-E01
Insulin (Porcine/Canine) ELISA For the quantitative measurement of insulin in Porcine/Canine serum and plasma (EDTA) For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 80-INSPO-E01
TANZANIA BUREAU OF STANDARDS
 TBS/AFDC 22 (5279) P3 Dried meat Specification DRAFT TANZANIA STANDARD TANZANIA BUREAU OF STANDARDS Dried meat Specification 0 FOREWORD Dried meat is a meat product obtained through appropriate techniques
TBS/AFDC 22 (5279) P3 Dried meat Specification DRAFT TANZANIA STANDARD TANZANIA BUREAU OF STANDARDS Dried meat Specification 0 FOREWORD Dried meat is a meat product obtained through appropriate techniques
II- Streptococci. Practical 3. Objective: Required materials: Classification of Streptococci: Streptococci can be classified according to:
 Practical 3 II- Streptococci Objective: 1. Use of blood agar to differentiate between,, and hemolytic streptococci. 2. To know Gram reaction, shape and arrangement of streptococci. 3. To differentiate
Practical 3 II- Streptococci Objective: 1. Use of blood agar to differentiate between,, and hemolytic streptococci. 2. To know Gram reaction, shape and arrangement of streptococci. 3. To differentiate
DRAFT EAST AFRICAN STANDARD
 DEAS 915: 2018 ICS 67.100.10 DRAFT EAST AFRICAN STANDARD Ghee Specification EAST AFRICAN COMMUNITY EAC 2018 First Edition 2018 DEAS 915: 2018 Copyright notice This EAC document is copyright-protected
DEAS 915: 2018 ICS 67.100.10 DRAFT EAST AFRICAN STANDARD Ghee Specification EAST AFRICAN COMMUNITY EAC 2018 First Edition 2018 DEAS 915: 2018 Copyright notice This EAC document is copyright-protected
Staphylococci. Gram stain: gram positive cocci arranged in clusters.
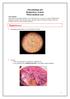 Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
APOB (Human) ELISA Kit
 APOB (Human) ELISA Kit Catalog Number KA4330 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
APOB (Human) ELISA Kit Catalog Number KA4330 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Proficiency Testing. Food Microbiology. October 2015
 Proficiency Testing Food Microbiology October 215 Edition Version 1 (215-11-26) Editor in chief Hans Lindmark, head of Biology department, National Food Agency Responsible for the scheme Laurence Nachin,
Proficiency Testing Food Microbiology October 215 Edition Version 1 (215-11-26) Editor in chief Hans Lindmark, head of Biology department, National Food Agency Responsible for the scheme Laurence Nachin,
IV2-113E Use by. Invitron Glargine ELISA Kit REF LOT IVD. Definitions. English. For in-vitro diagnostic use. Instructions for use.
 Definitions Instructions for use REF Catalogue number IV2-113E Use by English Invitron Glargine ELISA Kit For in-vitro diagnostic use Σ 96 LOT IVD Lot/Batch Code Storage temperature limitations In vitro
Definitions Instructions for use REF Catalogue number IV2-113E Use by English Invitron Glargine ELISA Kit For in-vitro diagnostic use Σ 96 LOT IVD Lot/Batch Code Storage temperature limitations In vitro
Human HBcAb IgM ELISA kit
 Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Food Safety Training
 Food Safety Training Good Personal Hygiene Wash hands frequently, especially after eating, drinking, smoking, touching your face, nose, ears, hair, handling waste, using the restroom, coughing or sneezing,
Food Safety Training Good Personal Hygiene Wash hands frequently, especially after eating, drinking, smoking, touching your face, nose, ears, hair, handling waste, using the restroom, coughing or sneezing,
Chlamydia Trachomatis IgA
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Survival of Aerobic and Anaerobic Bacteria in
 APPLIED MICROBIOLOGY, Mar. 1968, p. 445-449 Copyright 1968 American Society for Microbiology Vol. 16, No. 3 Printed in U.S.A. Survival of Aerobic and Anaerobic Bacteria in Chicken Meat During Freeze-Dehydration,
APPLIED MICROBIOLOGY, Mar. 1968, p. 445-449 Copyright 1968 American Society for Microbiology Vol. 16, No. 3 Printed in U.S.A. Survival of Aerobic and Anaerobic Bacteria in Chicken Meat During Freeze-Dehydration,
SIV p27 Antigen ELISA Catalog Number:
 INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
Micro lab notes. Dana alsulaibi
 Micro lab notes Dana alsulaibi Respiratory system Microbiology laboratory section Gram Positive Coccus These are the most common microorganisms to appear on a throat swab Staphylococcus Spp. Streptococcus
Micro lab notes Dana alsulaibi Respiratory system Microbiology laboratory section Gram Positive Coccus These are the most common microorganisms to appear on a throat swab Staphylococcus Spp. Streptococcus
T3 (Total) (Mouse/Rat) ELISA Kit
 T3 (Total) (Mouse/Rat) ELISA Kit Catalog Number KA0925 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
T3 (Total) (Mouse/Rat) ELISA Kit Catalog Number KA0925 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Most pathogenic Staph cause infection after a breach in the skin. PrepTalk Clicker Questions. First Case - Lulu. Diagnostic information
 Most pathogenic Staph cause infection after a breach in the skin. PrepTalk Clicker Questions A. True B. False Did you Prep? Which factor is most strongly correlated with the virulence of pathogenic staphylococci?
Most pathogenic Staph cause infection after a breach in the skin. PrepTalk Clicker Questions A. True B. False Did you Prep? Which factor is most strongly correlated with the virulence of pathogenic staphylococci?
PLATELIA CANDIDA Ab PLUS Negative and Positive Controls. Quality Control Human Sera
 PLATELIA CANDIDA Ab PLUS Negative and Positive Controls 48 62787 Quality Control Human Sera CONTENTS 1. INTENDED USE...3 2. INDICATIONS FOR USE...3 3. REAGENTS...3 4. WARNING FOR USERS...4 5. PRECAUTIONS
PLATELIA CANDIDA Ab PLUS Negative and Positive Controls 48 62787 Quality Control Human Sera CONTENTS 1. INTENDED USE...3 2. INDICATIONS FOR USE...3 3. REAGENTS...3 4. WARNING FOR USERS...4 5. PRECAUTIONS
DRAFT UGANDA STANDARD
 DRAFT UGANDA STANDARD DUS 1801 First Edition 2017-mm-dd Dried Fish Maws Specification Reference number DUS 1801:2017 UNBS 2017 DUS 1801: 2017 Compliance with this standard does not, of itself confer immunity
DRAFT UGANDA STANDARD DUS 1801 First Edition 2017-mm-dd Dried Fish Maws Specification Reference number DUS 1801:2017 UNBS 2017 DUS 1801: 2017 Compliance with this standard does not, of itself confer immunity
HSV-1 IgM ELISA. Catalog No (96 Tests) For Research Use Only. Not for use in Diagnostic Procedures.
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. HSV-1 IgM ELISA Kit is intended for the detection of IgM antibody to HSV-1 in human serum or plasma. SUMMARY AND
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. HSV-1 IgM ELISA Kit is intended for the detection of IgM antibody to HSV-1 in human serum or plasma. SUMMARY AND
TSH Receptor Autoantibody ELISA
 Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it TSH Receptor Autoantibody ELISA Product Data Sheet Cat.
Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it TSH Receptor Autoantibody ELISA Product Data Sheet Cat.
Thyroid Stimulating Hormone (TSH) ELISA Catalog No. GWB , legacy id (96 Tests)
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. TSH ELISA Kit is intended for the quantitative measurement of TSH in human serum or plasma. For research use only.
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. TSH ELISA Kit is intended for the quantitative measurement of TSH in human serum or plasma. For research use only.
Human Alpha 1 microglobulin ELISA Kit
 Human Alpha 1 microglobulin ELISA Kit Catalogue No.: EH4144 Size: 48T/96T Reactivity: Human Range:0.625-40ng/ml Sensitivity:
Human Alpha 1 microglobulin ELISA Kit Catalogue No.: EH4144 Size: 48T/96T Reactivity: Human Range:0.625-40ng/ml Sensitivity:
