July 2017 Section H of the Pharmaceutical Schedule Update
|
|
|
- Ashlee Strickland
- 5 years ago
- Views:
Transcription
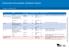
1 16 June 2017 July 2017 Section H of the Pharmaceutical Schedule Update The Section H Update summary is provided to DHB hospital pharmacists as a preview of the changes that will be announced in the next Update to Section H of the Pharmaceutical Schedule, and take effect from the beginning of next month. For any questions about these listing changes, or the Pharmaceutical Schedule in general, please contact us at enquiry@pharmac.govt.nz. Pharmaceutical Acetazolamide (Diamox) tab 250 mg Acitretin (Novatretin) cap 10 mg and 25 mg Adult diphtheria and tetanus vaccine (ADT Booster) inj 2 IU diphtheria toxoid with 20 IU tetanus toxoid in 0.5 ml syringe Alfentanil (Hameln) inj 0.5 mg per ml, 2 ml ampoule Amlodipine (Apo-Amlodipine) tab 2.5 mg, 5 mg and 10 mg Amorolfine (MycoNail) nail soln 5%, 5 ml Amoxicillin (Ibiamox) inj 250 mg, 500 mg and 1 g vials Atropine sulphate (Atropt) eye drops 1%, 15 ml Azithromycin tab 250 mg and 500 mg (Apo-Azithromycin), and grans for oral liq 200 mg per 5 ml (40 mg per ml) (Zithromax) Bendamustine hydrochloride (Ribomustin) inj 25 mg vial and 100 mg vial Benzylpenicillin sodium [Penicillin G] (Sandoz) inj 600 mg (1 million units) vial Betahistine dihydrochloride (Vergo 16) new listing inj 25 mg vial (p code ) inj 100 mg vial (p code )
2 tab 16 mg Bupivacaine hydrochloride (Marcain Isobaric) inj 5 mg per ml, 4 ml ampoule Bupivacaine hydrochloride (Marcain) inj 2.5 mg per ml, 100 ml bag Cefazolin (AFT) inj 500 mg vial and 1 g vial Cefotaxime (DBL Cefotaxime) inj 1 g vial Ciprofloxacin (Cipflox) tab 250 mg, 500 mg and 750 mg Clarithromycin (Apo-Clarithromycin) tab 250 mg and 500 mg Clarithromycin (Martindale) inj 500 mg vial Clonidine patch 2.5 mg, 100 mcg per day patch 5 mg, 200 mcg per day patch 7.5 mg, 300 mcg per day Cyproterone acetate with ethinyloestradiol (Ginet) tab 2 mg with ethinyloestradiol 35 mcg and 7 inert tablets Dexmedetomidine (Precedex) inj 100 mcg per ml, 2 ml vial Diphtheria, tetanus and pertussis vaccine (Boostrix) inj 2 IU diphtheria toxoid with 20 IU tetanus toxoid, 8 mcg pertussis toxoid, 8 mcg pertussis filamentous haemagluttinin and 2.5 mcg pertactin in 0.5 ml syringe Diphtheria, tetanus, pertussis and polio vaccine (Infanrix IPV) inj 30 IU diphtheria Brand change Mylan new listing and patch 2.5 mg, 100 mcg per day (p code ) patch 5 mg, 200 mcg per day (p code ) patch 7.5 mg, 300 mcg per day (p code ) Catapress-TTS-1, Catapress-TTS-2 and Catapress-TTS-3 patches to be delisted 1 September 2017
3 toxoid with 30IU tetanus toxoid, 25 mcg pertussis toxoid, 25 mcg pertussis filamentous haemagluttinin, 8 mcg pertactin and 80 D-antigen units poliomyelitis virus in 0.5 ml syringe Diphtheria, tetanus, pertussis, polio, hepatitis b and haemophilus influenzae type b vaccine (Infanrix-hexa) inj 30 IU diphtheria toxoid with 40 IU tetanus toxoid, 25 mcg pertussis toxoid, 25 mcg pertussis filamentous haemagluttinin, 8 mcg pertactin, 80 D-antigen units poliomyelitis virus, 10 mcg hepatitis B surface antigen in 0.5 ml syringe (1) and inj 10 mcg haemophilus influenzae type B vaccine vial Docetaxel (DBL Docetaxel) inj 10 mg per ml, 2 ml vial and 8 ml vial Docusate sodium (Coloxyl) tab 50 mg and 120 mg Donepezil hydrochloride (Donepezil-Rex) tab 5 mg and 10 mg Doxazosin (Apo-Doxazosin) tab 2 mg Doxazosin (Apo-Doxazosin) tab 4 mg Ephedrine (Max Health) inj 30 mg per ml, 1 ml ampoule Erlotinib (Tarceva) tab 100 mg and 150 mg Exemestane (Pfizer Exemestane) tab 25 mg Flucloxacillin (Flucloxin) inj 250 mg and 500 mg vials Flucloxacillin inj 1 g vial Gefitinib (Iressa) tab 250 mg price increase and Brand change Flucil new listing and (p code ) Flucloxin to be delisted 1 September 2017
4 Gliclazide (Glizide) tab 80 mg Glycerol liq Haemophilus influenzae type b vaccine (Hiberix) Haemophilus Influenzae type B polysaccharide 10 mcg conjugated to tetanus toxoid as carrier protein mcg; prefilled syringe plus vial 0.5 ml Haemophilus influenzae type b vaccine (Act-HIB) inj 10 mcg vial with diluent syringe Hepatitis A vaccine inj 720 ELISA units in 0.5 ml syringe (Havrix Junior) and inj 1440 ELISA units in 1 ml syringe (Havrix) Hepatitis B recombinant vaccine (HBvaxPRO) inj 5 mcg in 0.5 ml vial and inj 10 mcg in 1 ml vial Hepatitis B recombinant vaccine (HBvaxPRO) inj 40 mcg in 1 ml vial Hydrocortisone (ABM) powder Hydrocortisone and paraffin liquid and lanolin (DP Lotn HC) lotn 1% with paraffin liquid 15.9% and lanolin 0.6% Infliximab (Remicade) inj 100 mg Influenza vaccine (Influvac) inj 45 mcg in 0.5 ml syringe Iodised oil (Lipiodol Ultra Fluid) inj 38% w/w (480 mg per ml), 10 ml ampoule Isosorbide mononitrate (Duride) tab longacting 60 mg Ivabradine tab 5 mg Ketoconazole (Sebizole) shampoo 2%, Brand change healthe Glycerol BP Liquid, 500 ml new listing and (p code ) ABM, 2,000 ml to be delisted 1 September 2017 new listing and (p code ) to be delisted 1 October 2017 and price increase new presentation listing
5 100 ml Lamivudine (Zeffix) tab 100 mg and oral liq 5 mg per ml Lenalidomide (Revlimid) cap 15 mg new listing (p code ) Lidocaine [lignocaine] (LMX4) crm 4%, 5 g tubes Lidocaine [lignocaine] hydrochloride with adrenaline and tetracaine hydrochloride (Topicaine) soln 4% with adrenaline 0.1% and tetracaine hydrochloride 0.5%, 5 ml syringe Lopinavir with ritonavir (Kaletra) tab 200 mg with ritonavir 50 mg Magnesium sulphate (DBL) inj 2 mmol per ml, 5 ml ampoule Measles, mumps and rubella vaccine (Priorix) injection, measles virus 1,000 CCID50, mumps virus 5,012 CCID50, Rubella virus 1,000 CCID50; prefilled syringe/ampoule of diluent 0.5 ml Measles, mumps and rubella vaccine (M- M-R-II) inj 1000 TCID50 measles, TCID50 mumps and 1000 TCID50 rubella vial with diluent Pack size change 5 g tube new listing (p code ) 5 x 5 g tubes to be delisted 1 December 2017 new listing and (p code ) to be delisted 1 October 2017 Melatonin Circadin tab modified-release 2 mg new listing (p code ) tab 3 mg note added tab 1 mg and 2 mg to be delisted 1 January 2018 cap 2 mg and 3 mg to be delisted 1 January 2018 Meningococcal (A, C, Y and W-135) conjugate vaccine (Menactra) inj 4 mcg or each meningococcal polysaccharide conjugated to a total of approximately 48 mcg of diphtheria toxoid carrier per 0.5 ml vial
6 Meningococcal C conjugate vaccine (Neisvac-C) inj 10 mcg in 0.5 ml syringe, Methotrexate (Methotrexate Ebewe) inj 100 mg per ml, 50 ml vial Miconazole nitrate (Micreme) vaginal crm 2% with applicator, 40 g Morphine sulphate (Sevredol) tab immediate-release 10 mg and 20 mg Morphine sulphate (DBL Morphine Sulphate) inj 5 mg per ml, 1 ml ampoule; 10 mg per ml, 1 ml ampoule; 15 mg per ml, 1 ml ampoule; and 30 mg per ml, 1 ml ampoule Naltrexone hydrochloride (Naltraccord) tab 50 mg Nivolumab (Opdivo) inj 10 mg per ml, 4 ml vial and 10 ml vial Non-nucleoside reverse transcriptase inhibitors Noradrenaline (Noradrenaline BNM) inj 1 mg per ml, 4 ml ampoule Nucleoside reverse transcriptase inhibitors Olanzapine tab 2.5 mg, 5 mg and 10 mg (Zypine) and tab orodispersible 5 mg and 10 mg (Zypine ODT) Olopatadine (Patanol) eye drops 0.1%, 5 ml Oxazepam (Ox-Pam) tab 10 mg and 15 mg Paracetamol (Perfalgan) inj 10 mg per ml, 50 ml vial Paracetamol inj 10 mg per ml, 100 ml vial Pack size change 1 injection pack 10 injection pack to be delisted 1 July 2017 new listing and (p code ) price decrease to be delisted 1 September 2017 Brand change Paracetamol Kabi new listing and addition
7 of HSS (p code ) Perfalgan to be delisted 1 September 2017 Paracetamol with codeine (Paracetamol + Codeine (Relieve)) tab paracetamol 500 mg with codeine phosphate 8 mg, 1,000 tab pack Pamidronate disodium (Pamisol) inj 3 mg per ml, 10 ml vial and 9 mg per ml, 10 ml vial Pamidronate disodium (Pamisol) inj 6 mg per ml, 10 ml vial Pembrolizumab (Keytruda) inj 50 mg vial Perindopril (Apo-Perindopril) tab 2 mg and 4 mg Pethidine hydrochloride (DBL Pethidine Hydrochloride) inj 50 mg per ml, 1 ml and 2 ml ampoules Pneumococcal (PCV10) conjugated vaccine (Synflorix) inj 1 mcg of pneumococcal polysaccharide serotypes 1, 5, 6B, 7F, 9V, 14 and 23F; 3 mcg of pneumococcal polysaccharide serotypes 4, 18C and 19F in 0.5 ml prefilled syringe Pneumococcal (PCV13) conjugate vaccine (Prevenar 13) inj 30.8 mcg of pneumococcal polysaccharide serotypes 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F and 23F in 0.5 ml syringe Pneumococcal (PPV23) polysaccharide vaccine (Pneumovax 23) inj 575 mcg in 0.5 ml prefilled syringe (25 mcg of each 23 pneumococcal serotype) Poliomyelitis vaccine (IPOL) inj 80 D- antigen units in 0.5 ml syringe Poloxamer (Coloxyl) oral drops 10%, 30 ml Pack size change 1,000 tab pack and (p code ) 100 tab pack to be delisted 1 September 2017 price increase and new listing and (p code ) amended presentation description and restriction and
8 Procaine penicillin (Cilicaine) inj 1.5 g in 3.4 ml syringe Protease inhibitors Quetiapine (Quetapel) tab 25 mg, 100 mg, 200 mg and 300 mg Rifampicin (Rifadin) cap 150 mg and 300 mg, oral liq 100 mg per 5 ml, and inj 600 mg vial Rifaximin (Xifaxan) tab 550 mg Rituximab (Mabthera) inj 10 mg per ml, 10 ml vial and 50 ml vial Rizatriptan (Rizamelt) tab orodispersible 10 mg Ropivacaine hydrochloride inj 2 mg per ml, 100 ml and 200 ml bags Ropivacaine hydrochloride (Ropivacaine Kabi) inj 2 mg per ml, 10 ml and 20 ml ampoules; inj 7.5 mg per ml, 10 ml and 20 ml ampoules; and inj 10 mg per ml, 10 ml and 20 ml ampoules Rotavirus oral vaccine (Rotarix) oral susp live attenuated human rotavirus 1,000,000 CCID50 per dose, prefilled oral applicator Rotavirus live reassortant oral vaccine (RotaTeq) oral susp G1, G2, G3, G4, P1(8) 11.5 million CCID50 units per 2 ml, tube Roxithromycin (Rulide D) tab dispersible 50 mg Sildenafil inj 0.8 mg per ml, 12.5 ml vial Sodium citro-tartrate (Ural) grans eff 4 g Brand change Ropivacaine Kabi new listing and addition of HSS 100 ml bag (p code ) 200 ml bag (p code ) Naropin 100 ml and 200 ml bags to be delisted 1 September 2017 new listing and (p code ) to be delisted 1 October 2017 new listing (p code ) new listing and
9 sachets Strand transfer inhibitors Temazepam (Normison) tab 10 mg Testosterone cypionate (Depo- Testosterone) inj 100 mg per ml, 10 ml vial Timolol (Arrow-Timolol) eye drops 0.25% and 0.5%, 5 ml Tramadol hydrochloride tab sustained-release 100 mg (Tramal SR 100) tab sustained-release 150 mg (Tramal SR 150) tab sustained-release 200 mg (Tramal SR 200) Tramadol hydrochloride (Arrow-Tramadol) cap 50 mg Tramadol hydrochloride inj 50 mg per ml, 1 ml ampoule (Tramal 50) and 2 ml ampoule (Tramal 100) Triamcinolone acetonide (Aristocort) crm 0.02%, 100 g Triamcinolone acetonide (Aristocort) oint 0.02%, 100 g Triamcinolone acetonide (Kenacort-A 10) inj 10 mg per ml, 1 ml ampoule Triamcinolone acetonide (Kenacort-A 40) inj 40 mg per ml, 1 ml ampoule Triamcinolone acetonide (Kenalog in Orabase) paste 0.1%, 5 g Tuberculin PPD [mantoux] test (Tubersol) inj 5 TU per 0.1 ml, 1 ml vial Tuberculin, purified protein derivative inj 5 TU per 0.1 ml, 1 ml vial new listing and (p code ) to be delisted 1 July 2017
10 Ursodeoxycholic acid (Ursosan) cap 250 mg Vancomycin (Mylan) inj 500 mg vial Varicella vaccine [chickenpox vaccine] (Varilrix) inj 2000 PFU prefilled syringe plus vial Voriconazole (Vfend) inj 200 mg vial Zidovudine [AZT] with lamivudine (Alphapharm) tab 300 mg with lamivudine 150 mg new listing of 10 vial pack (p code ) amended chemical name and presentation description price increase
Notification of Product Changes (NOPC) forms, Containered Trade Product Pack (CTPP) codes and Pharmacodes
 28 February 2017 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2015/16 Invitation to Tender, d 6 November 2015 and
28 February 2017 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2015/16 Invitation to Tender, d 6 November 2015 and
Notification of Product Changes (NOPC) forms, Containered Trade Product Pack (CTPP) codes and Pharmacodes
 28 February 2017 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2015/16 Invitation to Tender, d 6 November 2015 and
28 February 2017 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2015/16 Invitation to Tender, d 6 November 2015 and
TENDER RESULTS. Notification of Product Changes (NOPC) forms and Pharmacodes
 30 June 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Hospital Supply Status for some products included in the 2012/13 Invitation to Tender, dated 31 October
30 June 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Hospital Supply Status for some products included in the 2012/13 Invitation to Tender, dated 31 October
Changes to the National Immunisation Schedule
 17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
Section H. Pharmaceuticals. New Zealand Pharmaceutical Schedule. Effective 1 July for Hospital. The Hospital Medicines List (HML)
 The Hospital Medicines List (HML) Section H for Hospital Management Agency Pharmaceuticals Update Update effective 1 August 2014 New Zealand Pharmaceutical Schedule Effective 1 July 2013 Cumulative for
The Hospital Medicines List (HML) Section H for Hospital Management Agency Pharmaceuticals Update Update effective 1 August 2014 New Zealand Pharmaceutical Schedule Effective 1 July 2013 Cumulative for
TENDER RESULTS. Notification of Product Changes (NOPC) forms and Pharmacodes. 30 May 2014
 30 May 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Hospital Supply Status for some products included in the 2012/13 Invitation to Tender, dated 31 October
30 May 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Hospital Supply Status for some products included in the 2012/13 Invitation to Tender, dated 31 October
TENDER RESULTS. Notification of Product Changes (NOPC) forms and Pharmacodes. 30 April 2014
 30 April 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2012/13 Invitation to Tender, dated 31 October 2012 and
30 April 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2012/13 Invitation to Tender, dated 31 October 2012 and
Release of the 2013/14 Invitation to Tender
 07 November 2013 Release of the 2013/14 Invitation to Tender The 2013/14 Invitation to Tender (2013/14 ITT) has been distributed today via the electronic tender (etender) system. If you do not receive
07 November 2013 Release of the 2013/14 Invitation to Tender The 2013/14 Invitation to Tender (2013/14 ITT) has been distributed today via the electronic tender (etender) system. If you do not receive
PHARMAC invites proposals for the supply of various vaccines in New Zealand.
 15 February 2016 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
15 February 2016 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
TENDER RESULTS. Notification of Product Changes (NOPC) forms and Pharmacodes. 28 February 2014
 28 February 2014 TENDER RESULTS PHARMAC has resolved to award tenders for for Sole Subsidised Supply Status and Status for some products included in the Invitation to Tender, dated 07 November 2013. Some
28 February 2014 TENDER RESULTS PHARMAC has resolved to award tenders for for Sole Subsidised Supply Status and Status for some products included in the Invitation to Tender, dated 07 November 2013. Some
Notification of Product Changes (NOPC) forms, Containered Trade Product Pack (CTPP) codes and Pharmacodes
 28 April 2017 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Hospital Supply Status for some products included in the 2016/17 Invitation to Tender, d 3 November
28 April 2017 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Hospital Supply Status for some products included in the 2016/17 Invitation to Tender, d 3 November
Update New Zealand Pharmaceutical Schedule Effective 1 September 2017
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 September 2017 Contents Summary of PHARMAC decisions effective 1 September 2017... 3 New listings... 5 Temozolomide
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 September 2017 Contents Summary of PHARMAC decisions effective 1 September 2017... 3 New listings... 5 Temozolomide
BabyJabs Vaccines. All vaccines are mercury- free We use aluminium- free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury- free We use aluminium- free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury- free We use aluminium- free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines. All vaccines are mercury-free We use aluminium-free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
Approval / Acknowledgement Letter Ref No. / Diary no F.No.12-48/GSK/PAC- Synflorix/15-BD 25-Mar-15 2 GlaxoSmithKline Pharmaceuticals Limited.
 1 GlaxoSmithKline Deletion of a n-significant in-process test limit during the purification process of the tetanus / diphtheria toxoids used in the manufacture of GSK's PCV vaccine [brand name: Synflorix]
1 GlaxoSmithKline Deletion of a n-significant in-process test limit during the purification process of the tetanus / diphtheria toxoids used in the manufacture of GSK's PCV vaccine [brand name: Synflorix]
BabyJabs Vaccines. All vaccines are mercury-free We use aluminium-free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
National Immunisation News
 July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
Vaccine ingredients. Fact sheet August 2017 page 1. Continued...
 Vaccine manufacture and composition is complex and tightly regulated to maximise safety. The safety of the individual components, and of the vaccine itself, must be demonstrated before a vaccine can be
Vaccine manufacture and composition is complex and tightly regulated to maximise safety. The safety of the individual components, and of the vaccine itself, must be demonstrated before a vaccine can be
TENDER RESULTS. Notification of Product Changes (NOPC) forms and Pharmacodes. 31 October 2014
 31 October 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2012/13 Invitation to Tender, dated 31 October 2012 and
31 October 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2012/13 Invitation to Tender, dated 31 October 2012 and
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 July 2015 Cumulative for May, June and July 2015 Contents Summary of PHARMAC decisions effective 1 July 2015... 3
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 July 2015 Cumulative for May, June and July 2015 Contents Summary of PHARMAC decisions effective 1 July 2015... 3
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines
 New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
Decision to amend access criteria for some vaccines
 3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow
3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
TRICARE Retail Vaccination Program Vaccine List - September 2018*
 Diphtheria, tetanus and pertussis Diphtheria and tetanus toxoids adsorbed acellular pertussis adsorbed DT DTaP Diphtheria and tetanus toxoids adsorbed Daptacel, Infanrix Tetanus and diphtheria toxoids
Diphtheria, tetanus and pertussis Diphtheria and tetanus toxoids adsorbed acellular pertussis adsorbed DT DTaP Diphtheria and tetanus toxoids adsorbed Daptacel, Infanrix Tetanus and diphtheria toxoids
Section H. Pharmaceuticals. New Zealand. Pharmaceutical Schedule. Effective 1 July for Hospital. The Hospital Medicines List (HML)
 The Hospital Medicines List (HML) Section H for Hospital Pharmaceuticals Pharmaceutical Management Agency Update Update effective 1 May 2014 New Zealand Cumulative for April and May 2014 Pharmaceutical
The Hospital Medicines List (HML) Section H for Hospital Pharmaceuticals Pharmaceutical Management Agency Update Update effective 1 May 2014 New Zealand Cumulative for April and May 2014 Pharmaceutical
Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age)
 Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age) Glossary: IPV= Injectable Polio vaccine OPV= Oral polio vaccine (these are oral drops, which is also given in "Pulse Polio
Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age) Glossary: IPV= Injectable Polio vaccine OPV= Oral polio vaccine (these are oral drops, which is also given in "Pulse Polio
Section H. Pharmaceuticals. New Zealand. Pharmaceutical Schedule. Effective 1 July for Hospital. The Hospital Medicines List (HML)
 The Hospital Medicines List (HML) Section H for Hospital Pharmaceuticals Pharmaceutical Management Agency Update Update effective 1 June 2014 New Zealand Cumulative for April, May and June 2014 Pharmaceutical
The Hospital Medicines List (HML) Section H for Hospital Pharmaceuticals Pharmaceutical Management Agency Update Update effective 1 June 2014 New Zealand Cumulative for April, May and June 2014 Pharmaceutical
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table of standardized
Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table of standardized
Pharmaceutical Management Agency. Section H Update. for Hospital. Pharmaceuticals. Effective 1 August 2018
 Pharmaceutical Management Agency Section H Update for Hospital Pharmaceuticals Effective 1 August 2018 Contents Summary of decisions effective 1 August 2018... 3 Section H changes to Part II... 8 Index...
Pharmaceutical Management Agency Section H Update for Hospital Pharmaceuticals Effective 1 August 2018 Contents Summary of decisions effective 1 August 2018... 3 Section H changes to Part II... 8 Index...
(price) Underlined indicates tender winning product now available at new subsidy $5.62 $4.89. $2.35 (Manuf price $5.26) $2.59
 Sole Supply - Pending Changes Quick Reference 1 *** Sorted by Sole *** Chemical Type, Strength, Pack Size Pharmacode Current New Tender of New of Sole Supply and Reference Start Thiamine hydrochloride
Sole Supply - Pending Changes Quick Reference 1 *** Sorted by Sole *** Chemical Type, Strength, Pack Size Pharmacode Current New Tender of New of Sole Supply and Reference Start Thiamine hydrochloride
National Immunisation News
 National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
Enhanced immunisation schedule Victoria
 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Immunise your child on time. It s their best protection
 Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Price Changes advised by Pharmac As at 12 June 2015
 s advised by Pharmac New to be available ex Manufacturer from 12th of preceding month. From Wholesale from date of supplier support which could vary. Product Supplier Schedule % + or - 01 Jun 15 31 Oct
s advised by Pharmac New to be available ex Manufacturer from 12th of preceding month. From Wholesale from date of supplier support which could vary. Product Supplier Schedule % + or - 01 Jun 15 31 Oct
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
Vaccine Label Examples
 Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Modern Drug House.
 +91-8048941768 Modern Drug House https://www.moderndrughouse.in/ We are one of the leading suppliers and traders of a wide range of Vaccine and Pharmaceutical Products. Owing to its effectiveness, purity
+91-8048941768 Modern Drug House https://www.moderndrughouse.in/ We are one of the leading suppliers and traders of a wide range of Vaccine and Pharmaceutical Products. Owing to its effectiveness, purity
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 September 2014 Contents Summary of PHARMAC decisions effective 1 September 2014... 3 Erythropoietin changes to subsidy...
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 September 2014 Contents Summary of PHARMAC decisions effective 1 September 2014... 3 Erythropoietin changes to subsidy...
Notification of Product Changes (NOPC) forms, Containered Trade Product Pack (CTPP) codes and Pharmacodes
 29 February 2016 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2014/15 Invitation to Tender, d 6 November 2014, and
29 February 2016 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2014/15 Invitation to Tender, d 6 November 2014, and
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
Aspirin. Iron Supplements
 Interim Final Rules for Non-Grandfathered Group Health Plans and Health Insurance Issuers Coverage of Preventive Services Under the Patient Protection and Affordable Care Act Aspirin Aspirin to Prevent
Interim Final Rules for Non-Grandfathered Group Health Plans and Health Insurance Issuers Coverage of Preventive Services Under the Patient Protection and Affordable Care Act Aspirin Aspirin to Prevent
PHARMAC invites proposals for the supply of various vaccines in New Zealand.
 28 June 2013 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
28 June 2013 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
TENDER RESULTS. Notification of Product Changes (NOPC) forms and Pharmacodes. 19 December 2014
 19 December 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2012/13 Invitation to Tender, dated 31 October 2012
19 December 2014 TENDER RESULTS PHARMAC has resolved to award tenders for Sole Subsidised Supply Status and Status for some products included in the 2012/13 Invitation to Tender, dated 31 October 2012
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation
 Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Vaccine Label Examples
 Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 May Contents Summary of PHARMAC decisions effective 1 May... 3 Abiraterone acetate (Zytiga) new listing... 5 Actavis
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 May Contents Summary of PHARMAC decisions effective 1 May... 3 Abiraterone acetate (Zytiga) new listing... 5 Actavis
Your DISPENSARY Tips as at 11 March 2016
 This information has been prepared to provided some essential buying tips for the upcoming months. It is designed to be used in conjunction with the full text documents contained within the Pharmac Updates
This information has been prepared to provided some essential buying tips for the upcoming months. It is designed to be used in conjunction with the full text documents contained within the Pharmac Updates
Update New Zealand Pharmaceutical Schedule Effective 1 February 2018 Cumulative for January and February 2018
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 February Cumulative for January and February Contents Summary of PHARMAC decisions effective 1 February... 3 New
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 February Cumulative for January and February Contents Summary of PHARMAC decisions effective 1 February... 3 New
14: Immunological products and vaccines
 File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 February Cumulative for December 2014 and January Contents Summary of PHARMAC decisions effective 1 February... 3
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 February Cumulative for December 2014 and January Contents Summary of PHARMAC decisions effective 1 February... 3
Healthy People 2020 objectives were released in 2010, with a 10-year horizon to achieve the goals by 2020.
 Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Vaccination Decision Making: What Providers Need to Know
 Objectives Vaccination Decision Making: What Providers Need to Know Kelli Smith, RN, BSN Iowa Department of Public Health Immunization Program Catch-up Schedule: how to most efficiently bring persons up-to-date
Objectives Vaccination Decision Making: What Providers Need to Know Kelli Smith, RN, BSN Iowa Department of Public Health Immunization Program Catch-up Schedule: how to most efficiently bring persons up-to-date
Release of the 2017/18 Invitation to Tender
 2 November 2017 Release of the 2017/18 Invitation to Tender The 2017/18 Invitation to Tender (2017/18 ITT) has been distributed today. If you have already registered your e-mail address with PHARMAC s
2 November 2017 Release of the 2017/18 Invitation to Tender The 2017/18 Invitation to Tender (2017/18 ITT) has been distributed today. If you have already registered your e-mail address with PHARMAC s
Immunisation schedule Victoria
 Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Coverage of Vaccines Medicaid and Child Health Plus Members
 Coverage of Vaccines Medicaid and Child Health Plus Members For children between the ages 0-18, routine recommended vaccinations are covered through Vaccines for Children program [VFC]. Fidelis Care will
Coverage of Vaccines Medicaid and Child Health Plus Members For children between the ages 0-18, routine recommended vaccinations are covered through Vaccines for Children program [VFC]. Fidelis Care will
Update New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 December 2017 Cumulative for September, October, November and December 2017 Contents Summary of PHARMAC decisions
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 December 2017 Cumulative for September, October, November and December 2017 Contents Summary of PHARMAC decisions
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 January Contents Summary of PHARMAC decisions effective 1 January... 4 What s changing?... 6 Metoprolol succinate
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 January Contents Summary of PHARMAC decisions effective 1 January... 4 What s changing?... 6 Metoprolol succinate
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES
 GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL
 STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL 114 F2 Students Please complete ALL information: Name of Pupil Grade Address of Pupil City
STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL 114 F2 Students Please complete ALL information: Name of Pupil Grade Address of Pupil City
Vaccines and other immunological antimicrobial therapy 1
 Vaccines and other immunological antimicrobial therapy 1 Vaccines Vaccine: a biological preparation that provides active acquired immunity to a particular disease. Vaccine typically contains an agent that
Vaccines and other immunological antimicrobial therapy 1 Vaccines Vaccine: a biological preparation that provides active acquired immunity to a particular disease. Vaccine typically contains an agent that
2007/08 Tender. Current Subsidy and price. Bendrofluazide Tab 5 mg; 500 tablets, bottle $21.50 $11.75 Arrow-Bendrofluazide (Arrow)
 31 March 2010 TENDER RESULTS The PHARMAC Board (or Chief Executive under delegated authority) has resolved to accept tenders for Sole Subsidised Supply and Status for some of the products included in the
31 March 2010 TENDER RESULTS The PHARMAC Board (or Chief Executive under delegated authority) has resolved to accept tenders for Sole Subsidised Supply and Status for some of the products included in the
What DO the childhood immunization footnotes reveal? Questions and answers
 What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
Release of the 2014/15 Invitation to Tender
 6 November 2014 Release of the 2014/15 Invitation to Tender The 2014/15 Invitation to Tender (2014/15 ITT) has been distributed today. Please note that the Electronic Portal will not be available until
6 November 2014 Release of the 2014/15 Invitation to Tender The 2014/15 Invitation to Tender (2014/15 ITT) has been distributed today. Please note that the Electronic Portal will not be available until
Price Changes advised by Pharmac As at 02 July 2015
 s advised by Pharmac New to be available ex Manufacturer from 12th of preceding month. From Wholesale from date of supplier support which could vary. Product Supplier Schedule 01 Jul 15 29 May 15 Irinotecan
s advised by Pharmac New to be available ex Manufacturer from 12th of preceding month. From Wholesale from date of supplier support which could vary. Product Supplier Schedule 01 Jul 15 29 May 15 Irinotecan
1. Executive Summary 2. Worldwide Pediatric Vaccines Market and Forecast ( ) 3. Worldwide Pediatric Vaccines Market Share & Forecast (Sector
 1. Executive Summary 2. Worldwide Pediatric Vaccines Market and Forecast (2006 ) 3. Worldwide Pediatric Vaccines Market Share & Forecast (Sector wise) (2005 ) 4. Worldwide Pediatric Vaccines Market Share
1. Executive Summary 2. Worldwide Pediatric Vaccines Market and Forecast (2006 ) 3. Worldwide Pediatric Vaccines Market Share & Forecast (Sector wise) (2005 ) 4. Worldwide Pediatric Vaccines Market Share
Objectives. Immunity. Childhood Immunization Risk of Non-Vaccinated Children 12/22/2015
 Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
M I C R O B I O L O G Y
 ninth edition TORTORA FUNKE CASE M I C R O B I O L O G Y a n i n t r o d u c t i o n 18 Practical Applications of Immunology PowerPoint Lecture Slide Presentation prepared by Christine L. Case Vaccine
ninth edition TORTORA FUNKE CASE M I C R O B I O L O G Y a n i n t r o d u c t i o n 18 Practical Applications of Immunology PowerPoint Lecture Slide Presentation prepared by Christine L. Case Vaccine
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 August 2014 Cumulative for May, June, July, and August 2014 Contents Summary of PHARMAC decisions effective 1 August
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 August 2014 Cumulative for May, June, July, and August 2014 Contents Summary of PHARMAC decisions effective 1 August
CHILDHOOD VACCINATION
 EPI (3) Age of Child How and Where is it given? CHILDHOOD VACCINATION Nicolette du Plessis Block 10 28/02/2012 10 weeks DTaP-IPV/Hib (2) Diphtheria, Tetanus, Acellular pertussis, Inactivated polio vaccine,
EPI (3) Age of Child How and Where is it given? CHILDHOOD VACCINATION Nicolette du Plessis Block 10 28/02/2012 10 weeks DTaP-IPV/Hib (2) Diphtheria, Tetanus, Acellular pertussis, Inactivated polio vaccine,
Update New Zealand Pharmaceutical Schedule Effective 1 April 2016 Cumulative for January, February, March and April 2016
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 April 2016 Cumulative for January, February, March and April 2016 Contents Summary of PHARMAC decisions effective
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 April 2016 Cumulative for January, February, March and April 2016 Contents Summary of PHARMAC decisions effective
Communicable Disease & Immunization
 Communicable Disease & Immunization Ingham County Health Surveillance Book 2016 Communicable Disease & Immunization - 1 Communicable Disease & Immunization T he control of communicable disease and immunization,
Communicable Disease & Immunization Ingham County Health Surveillance Book 2016 Communicable Disease & Immunization - 1 Communicable Disease & Immunization T he control of communicable disease and immunization,
IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY
 NATIONAL GUIDELINES PAEDIATRIC ONCOLOGY AND HAEMATOLOGY For the Care of Childhood Cancer in Specialist Child Cancer and Shared Care Centres. IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY Date
NATIONAL GUIDELINES PAEDIATRIC ONCOLOGY AND HAEMATOLOGY For the Care of Childhood Cancer in Specialist Child Cancer and Shared Care Centres. IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY Date
Update New Zealand Pharmaceutical Schedule Effective 1 July 2018 Cumulative for May, June and July 2018
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 July 2018 Cumulative for May, June and July 2018 Contents Summary of PHARMAC decisions effective 1 July 2018... 4
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 July 2018 Cumulative for May, June and July 2018 Contents Summary of PHARMAC decisions effective 1 July 2018... 4
Post-Transplant Vaccination and Re-Immunisation Procedure
 Post-Transplant Vaccination and Re-Immunisation Procedure Table of Contents Purpose... 1 Scope/Audience... 1 Associated documents and forms... 1 Definitions... 2 Background... 2 Vaccination Recommendations...
Post-Transplant Vaccination and Re-Immunisation Procedure Table of Contents Purpose... 1 Scope/Audience... 1 Associated documents and forms... 1 Definitions... 2 Background... 2 Vaccination Recommendations...
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 November 2015 Cumulative for September, October and November 2015 Contents Summary of PHARMAC decisions effective
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 November 2015 Cumulative for September, October and November 2015 Contents Summary of PHARMAC decisions effective
Update New Zealand Pharmaceutical Schedule Effective 1 September 2016
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 September 2016 Contents Summary of PHARMAC decisions effective 1 September 2016... 3 What s changing?... 5 Pembrolizumab
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 September 2016 Contents Summary of PHARMAC decisions effective 1 September 2016... 3 What s changing?... 5 Pembrolizumab
All Kindergarteners and 4-6 year old transfer students. 4 doses DTP or DTaP 1 dose must be at or after 4 years of age. None
 Parents Vaccines Required for School Entry in Michigan Whenever children are brought into group settings, there is a chance for diseases to spread. Children must follow state vaccine laws in order to attend
Parents Vaccines Required for School Entry in Michigan Whenever children are brought into group settings, there is a chance for diseases to spread. Children must follow state vaccine laws in order to attend
Update New Zealand Pharmaceutical Schedule Effective 1 May 2018
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 May Contents Summary of PHARMAC decisions effective 1 May... 3 New tender listings for 1 May... 4 New listings...
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 May Contents Summary of PHARMAC decisions effective 1 May... 3 New tender listings for 1 May... 4 New listings...
Update New Zealand Pharmaceutical Schedule Effective 1 March 2017 Cumulative for January, February and March 2017
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 March Cumulative for January, February and March Contents Summary of PHARMAC decisions effective 1 March... 3 What
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 March Cumulative for January, February and March Contents Summary of PHARMAC decisions effective 1 March... 3 What
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 October 2015 Cumulative for September and October 2015 Contents Summary of PHARMAC decisions effective 1 October
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 October 2015 Cumulative for September and October 2015 Contents Summary of PHARMAC decisions effective 1 October
State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010
 State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010 ALASKA IMMUNIZATION PROGRAM (907) 269-8000 OR 1-888-430-4321 http://www.epi.hss.state.ak.us/id/immune.stm (This page
State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010 ALASKA IMMUNIZATION PROGRAM (907) 269-8000 OR 1-888-430-4321 http://www.epi.hss.state.ak.us/id/immune.stm (This page
Update New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 April Cumulative for January, February, March and April Contents Summary of PHARMAC decisions effective 1 April...
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 April Cumulative for January, February, March and April Contents Summary of PHARMAC decisions effective 1 April...
2016 Vaccine Preventable Disease Summary
 2016 Vaccine Preventable Disease Summary 12251 James Street Holland, MI 49424 www.miottawa.org/healthdata Prepared October 2017 2016 Summary of Vaccine Preventable Diseases (VPDs) Reported to Ottawa County
2016 Vaccine Preventable Disease Summary 12251 James Street Holland, MI 49424 www.miottawa.org/healthdata Prepared October 2017 2016 Summary of Vaccine Preventable Diseases (VPDs) Reported to Ottawa County
Immunization Guidelines For the Use of State Supplied Vaccine July 1, 2011
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
VACCINATION. DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M.
 VACCINATION DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M. IMMUNIZATION Immunization is defined as the procedure by which the body is prepared to fight against a specific disease. It is used to induce the
VACCINATION DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M. IMMUNIZATION Immunization is defined as the procedure by which the body is prepared to fight against a specific disease. It is used to induce the
Update. New Zealand Pharmaceutical Schedule. Effective 1 November Cumulative for September, October and November 2018
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 November 2018 Cumulative for September, October and November 2018 Contents Summary of PHARMAC decisions effective
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 November 2018 Cumulative for September, October and November 2018 Contents Summary of PHARMAC decisions effective
Vaccinology 101 for Fellows
 Vaccinology 101 for Fellows Meg Fisher, MD Medical Director, The Children s Hospital Monmouth Medical Center An affiliate of the Saint Barnabas Health Care System Long Branch, NJ Disclosures I have no
Vaccinology 101 for Fellows Meg Fisher, MD Medical Director, The Children s Hospital Monmouth Medical Center An affiliate of the Saint Barnabas Health Care System Long Branch, NJ Disclosures I have no
2018/19 Immunisation programmes list of additional and enhanced services
 2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines
 Viral Hepatitis Prevention Board Hanoi, 25 26 July 2018 Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines Prof. Timo Vesikari Vaccine Research Center University
Viral Hepatitis Prevention Board Hanoi, 25 26 July 2018 Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines Prof. Timo Vesikari Vaccine Research Center University
Update New Zealand Pharmaceutical Schedule Effective 1 May 2016
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 May Contents Summary of PHARMAC decisions effective 1 May... 3 Sumatriptan update on Sumatriptan injections... 4
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 May Contents Summary of PHARMAC decisions effective 1 May... 3 Sumatriptan update on Sumatriptan injections... 4
Approval of proposals for various pharmaceuticals
 9 August 2010 Approval of proposals for various pharmaceuticals PHARMAC is pleased to announce the approval of proposals for pharmaceuticals in several therapeutic groups which aim to address some issues
9 August 2010 Approval of proposals for various pharmaceuticals PHARMAC is pleased to announce the approval of proposals for pharmaceuticals in several therapeutic groups which aim to address some issues
National Immunisation News The Newsletter of the HSE National Immunisation Office
 August 2017 1 National Immunisation News The Newsletter of the HSE National Immunisation Office CONTENTS Increase in MenC disease Measles outbreak in Europe Increase HPV Vaccine uptake to protect girls
August 2017 1 National Immunisation News The Newsletter of the HSE National Immunisation Office CONTENTS Increase in MenC disease Measles outbreak in Europe Increase HPV Vaccine uptake to protect girls
New Zealand Pharmaceutical Schedule
 Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 October 2014 Cumulative for September and October 2014 Contents Summary of PHARMAC decisions effective 1 October
Pharmaceutical Management Agency Update New Zealand Pharmaceutical Schedule Effective 1 October 2014 Cumulative for September and October 2014 Contents Summary of PHARMAC decisions effective 1 October
Immunisation Subcommittee of PTAC Meeting held 23 May (minutes for web publishing)
 Immunisation Subcommittee of PTAC Meeting held 23 May 2016 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology and
Immunisation Subcommittee of PTAC Meeting held 23 May 2016 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology and
A Review of the Pediatric Immunization Schedule
 A Review of the Pediatric Immunization Schedule Michaela Christian PGY-1 Pharmacy Practice Resident Mercy Hospital, A Campus of Plantation General Hospital Michaela.Christian@Hcahealthcare.com Objectives
A Review of the Pediatric Immunization Schedule Michaela Christian PGY-1 Pharmacy Practice Resident Mercy Hospital, A Campus of Plantation General Hospital Michaela.Christian@Hcahealthcare.com Objectives
National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health
 Page 1 of 8 National Immunisation News February 2008 National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health 2008: Issue 5 HSE National Cold Chain
Page 1 of 8 National Immunisation News February 2008 National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health 2008: Issue 5 HSE National Cold Chain
National Immunisation News
 DN DN W DN C DW DSC DSW DSE DS August 2018 National Immunisation News The Newsletter of the HSE National Immunisation Office Contents Vaccine uptake Changes to the Immunisation Guidelines for Ireland Vaccines
DN DN W DN C DW DSC DSW DSE DS August 2018 National Immunisation News The Newsletter of the HSE National Immunisation Office Contents Vaccine uptake Changes to the Immunisation Guidelines for Ireland Vaccines
