Galvanic Corrosion Behavior of Orthodontic Archwire Alloys Coupled to Bracket Alloys
|
|
|
- Avis Wells
- 5 years ago
- Views:
Transcription
1 Original Article Galvanic Corrosion Behavior of Orthodontic Archwire Alloys Coupled to Bracket Alloys Masahiro Iijima a ; Kazuhiko Endo b ; Toshihiro Yuasa c ; Hiroki Ohno d ; Kazuo Hayashi e ; Mitsugi Kakizaki f ; Itaru Mizoguchi g ABSTRACT The purpose of this study was to provide a quantitative assessment of galvanic corrosion behavior of orthodontic archwire alloys coupled to orthodontic bracket alloys in 0.9% NaCl solution and to study the effect of surface area ratios. Two common bracket alloys, stainless steels and titanium, and four common wire alloys, nickel-titanium (NiTi) alloy, -titanium ( -Ti) alloy, stainless steel, and cobalt-chromium-nickel alloy, were used. Three different area ratios, 1:1, 1:2.35, and 1:3.64, were used; two of them assumed that the multibracket appliances consists of 14 brackets and inch of round archwire or inch of rectangular archwire. The galvanic current was measured for 3 successive days using zero-impedance ammeter. When the NiTi alloy was coupled with Ti (1:1, 1:2.35, and 1:3.64 of the surface area ratio) or -Ti alloy was coupled with Ti (1:2.35 and 1:3.64 of the surface area ratio), Ti initially was the anode and corroded. However, the polarity reversed in 1 hour, resulting in corrosion of the NiTi or -Ti. The NiTi alloy coupled with SUS 304 or Ti exhibited a relatively large galvanic current density even after 72 hours. It is suggested that coupling SUS 304-NiTi and Ti-NiTi may remarkably accelerate the corrosion of NiTi alloy, which serves as the anode. The different anode-cathode area ratios used in this study had little effect on galvanic corrosion behavior. KEY WORDS: Galvanic corrosion; Archwire; Bracket INTRODUCTION Fixed orthodontic metallic appliances such as brackets, archwires, and molar bands are manufactured from base metal alloys such as stainless steel, cobalta Assistant Professor, Department of Orthodontics, Health Sciences University of Hokkaido, Hokkaido, Japan. b Associate Professor, Department of Dental Materials Science, Health Sciences University of Hokkaido, Hokkaido, Japan. c Postgraduate Student, Department of Orthodontics, Health Sciences University of Hokkaido, Hokkaido, Japan. d Professor, Department of Dental Materials Science, Health Sciences University of Hokkaido, Hokkaido, Japan. e Instructor, Department of Orthodontics, Health Sciences University of Hokkaido, Hokkaido, Japan. f Dental Technician, School of Dentistry, Health Sciences University of Hokkaido, Hokkaido, Japan. g Professor, Department of Orthodontics, Health Sciences University of Hokkaido, Hokkaido, Japan. Corresponding author: Masahiro Iijima, DDS, PhD, Department of Orthodontics, School of Dentistry, Health Sciences University of Hokkaido, Kanazawa 1757, Ishikari-Tobetsu, Hokkaido , Japan ( iijima@hoku-iro-u.ac.jp) Accepted: May Submitted: December by The EH Angle Education and Research Foundation, Inc. chromium-nickel (CoCrNi) alloy, nickel-titanium (NiTi) alloy, -titanium ( -Ti) alloy, and pure titanium. 1 These orthodontic alloys, except for -Ti alloy and pure titanium, contain nickel, and nickel produces more allergic reactions during orthodontic treatment than any other metal. 2 5 Because the release of nickel ions from orthodontic alloys is a clinical concern, general corrosion resistance of orthodontic metal has been widely investigated by many researchers In a clinical situation, two dissimilar alloys having different corrosion potentials are often placed in contact such as in orthodontic brackets and archwires. This can cause galvanic corrosion that leads to preferential release of metal ions from the anodic metal or alloy Furthermore, the surface area ratio of the two dissimilar alloys is a very important factor because it affects the galvanic corrosion behavior An unfavorable area ratio, which consists of a large cathode and a small anode, might lead to a greater corrosion rate from the anodic alloy. 11 It is also difficult to determine the real surface area ratio between brackets and archwire in clinical use, which has not been performed in previous studies. The purpose of this study was to provide a quanti- 705
2 706 IIJIMA, ENDO, YUASA, OHNO, HAYASHI, KAKIZAKI, MIZOGUCHI TABLE 1. Compositions (Mass %) of Each Alloy a Ni Ti Fe Cr Mo Mn Co Zr Sn other Stainless steel (SUS 304) 8.05 balance Titanium (ASTM grade2) balance 0.63 NiTi alloy Co Cr Ni alloy titanium alloy balance a NiTi indicates nickel-titanium; Co Cr Ni, cobalt-chromium-nickel. tative assessment of the galvanic corrosion behavior of orthodontic archwire alloys coupled to orthodontic bracket alloys in 0.9% NaCl solution and to study the effect of surface area ratios. The hypotheses in this study were that (1) different kinds of coupled alloys have different galvanic corrosion behavior variants and (2) the surface area ratio affects the galvanic corrosion behavior. MATERIALS AND METHODS In this study, two common bracket alloys, stainless steels (Daido Steel, Nagoya, Japan; SUS 304) and titanium (KOBELCO, Hyogo, Japan; ASTM Grade 2), were used. The stainless steel (SUS 304) and titanium (Ti) were rolled sheets and were cut into disk-shaped samples. Four common archwire alloys, NiTi alloy (Ormco, Calif), SUS 304, cobalt-chromium-nickel alloy (Elgiloy blue, Rocky Mountain Orthodontics, Colo), and -titanium alloy (TMA wire, Ormco), were used. The NiTi was a wiredrawing sample with an approximately 8 mm diameter that was cut into disks of 2 mm thickness. The CoCrNi alloy and -Ti were the archwire products, and the disk-shaped samples were obtained by casting with an arc-melting gas pressure casting machine under an argon atmosphere (Vulcan- T, Shofu Inc, Kyoto, Japan). The nominal compositions for these alloys are shown in Table 1. Estimation of the ratio of the brackets and archwire area Three-dimensional computed models of the brackets (Metal Bracket, Dentsply Sankin, Tokyo, Japan) were constructed on the basis of each exact design drawing, as provided by the manufacture. The surface areas of the brackets were calculated using a computer-aided data analysis system, which consisted of a graphical workstation (Zx1, Intergraph, Huntsville, Ala), and data-processing and data-analyzing software (I-DEAS, SDRC, Milford, Conn) (Table 2). To minimize the computation error, the base plane of the bracket was deleted from the calculated results. Table 3 shows the total surface areas of the brackets and archwires. The lengths of the archwires were 115 mm for the mandibular arch and 123 mm for the maxillary arch, TABLE 2. (mm 2 ) The Surface Area of Each Bracket and Molar Tube Upper central incisor Upper lateral incisor Lower incisor Canine Premolar First molar Second molar TABLE 3. Total Surface Area of the Brackets, Molar Tube, and Archwires (mm 2 ) a Bracket Upper Lower Molar tube Upper (0.016 inch) Lower (0.016 inch) Archwire Upper ( inch) Lower ( inch) a Total surface area of each side were consisted of 10 brackets and four molar tubes. To estimate total area of archwires, 115mm for mandibular arch and 123mm for maxillary upper were used in this study. and the archwire surface areas were calculated using the value of the cross section claimed by the manufacturer. In this study, three kinds of area ratio, 1:1, 1: 2.35, and 1:3.64, were used, and two of them (1:2.35 and 1:3.64) assumed that the multibracket appliances consist of 14 brackets and inch (0.41 mm) of round archwire or inch ( mm) of rectangular archwire. Specimen preparation Specimens of alloys were disk shaped. Three different diameters, 7.5, 11.5, and 14.3 mm, for bracket alloys and one diameter, 7.5 mm, for archwire alloy were used in this study to obtain three area ratios. After specimens were encapsulated in epoxy resin, one side of the surface was polished mechanically to a mirrorlike finish using silicone carbide paper followed by m alumina paste.
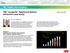
3 GALVANIC CORROSION OF ORTHODONTIC ALLOYS 707 FIGURE 1. Representative corrosion potential of the NiTi, -Ti, SUS 304, and Ti during the first 24 hours of immersion in 0.9% NaCl solution. NiTi indicates nickel-titanium; -Ti, -titanim. Evaluation of corrosion potential for uncoupled alloys and galvanic corrosion for coupled alloys The measurement of the potential difference between a working electrode (sample) and a reference electrode is performed with a voltmeter, which should be allowed as little current as possible. This is achieved by having an extremely high internal resistance within the voltmeter. 15 In this study, the corrosion potentials (E corr ) of the uncoupled alloys were measured for three successive days using an electrometer with an input impedance of An Ag/AgCl electrode (saturated KCl) was used as the reference electrode. The electrolyte, 0.9% NaCl solution, was exposed to air and the temperature was kept at 37 C during the experiments. The use of ammeters in galvanic corrosion current measurements should always be treated with caution because their finite resistance adds to the resistance within the corrosion cell and can affect the reactions, which occur. Instruments known as zero-resistance ammeters are used for accurate measurements. 15 In this study, the galvanic current between seven different coupled alloys (SUS 304-NiTi, SUS Ti, SUS 304-CoCrNi, Ti-NiTi, Ti- -Ti, Ti-SUS 304, and Ti-CoCrNi) was measured for 3 successive days using zero-impedance ammeter (2090, Toho Technical Research, Tokyo, Japan). The distance between the two specimens was approximately 10 mm. A total of three replicate samples were investigated both in the corrosion potential measurement for uncoupled alloys and in the galvanic current measurement for coupled samples. FIGURE 2. Representative galvanic current density of archwire alloys (NiTi, -Ti, SUS 304, CoCrNi) coupled with bracket alloys (SUS 304, Ti) with area ratio of 1:1 in 0.9% NaCl solution. NiTi indicates nickel-titanium; -Ti, -titanim; and CoCr, cobalt-chromium. RESULTS Figure 1 shows the variations in the corrosion potentials (E corr ) of representative uncoupled alloys during the first 24 hours of immersion in 0.9% NaCl solution. The corrosion potentials (E corr ) of the uncoupled -Ti, CoCrNi, SUS 304, and Ti moved in the noble direction a few hours after starting immersion, whereas the corrosion potential of the NiTi moved in the less noble direction a few hours after starting immersion. Comparing the steady-state values of E corr, SUS 304 and CoCrNi indicated more noble corrosion potential (approximately 50 mv for both samples) than any other samples. In contrast, the corrosion potential for the NiTi (approximately 400 mv) was much lower than that for any other samples. The corrosion potentials obtained from Ti and -Ti (approximately 200 mv) were intermediate. The galvanic current density was calculated from the measured galvanic current and surface area of the alloy, which served as an anode during most of the measurement time. Figures 2 through 4 show the variation in the galvanic current density during the first 24 hours of immersion; similar results were found for second and third samples. Table 4 shows the mean values of the galvanic current density obtained from all coupled alloys at 24, 48, and 72 hours. In all coupled samples, the galvanic current density decreased with time and reached a nearly constant value after 24 hours. In NiTi alloy coupled with Ti (1:1, 1:2.35, and 1:3.64 of the surface ratio) or -Ti alloy coupled with Ti (1:2.35 and 1:3.64 of the surface ratio), Ti was initially the anode and corroded. However, the polarity reversed in 1 hour, resulting in corrosion of the NiTi or -Ti. The galvanic current density for NiTi alloy coupled with SUS 304 or Ti at 72 hours of immersion was to A/cm 2, which was more than one order of magnitude higher than that observed for the other couples.
4 708 IIJIMA, ENDO, YUASA, OHNO, HAYASHI, KAKIZAKI, MIZOGUCHI FIGURE 3. Representative galvanic current density of archwire alloys (NiTi, -Ti, SUS 304, CoCrNi) coupled with bracket alloys (SUS 304, Ti) with area ratio of 1:2.35 during the first 24 hours of immersion in 0.9% NaCl solution. NiTi indicates nickel-titanium; -Ti, titanim; and CoCr, cobalt-chromium. FIGURE 4. Representative galvanic current density of archwire alloys (NiTi, -Ti, SUS 304, CoCrNi) coupled with bracket alloys (SUS 304, Ti) with area ratio of 1:3.64 during the first 24 hours of immersion in 0.9% NaCl solution. NiTi indicates nickel-titanium; -Ti, titanim; and CoCr, cobalt-chromium. DISCUSSION This study focused on galvanic corrosion behavior for orthodontic archwire alloys coupled to orthodontic bracket alloys with different compositions using diskshaped specimens with relatively similar surface conditions. All specimens were polished mechanically to a mirrorlike finish to minimize the difference of specimen surface conditions such as surface roughness and the structure of the surface oxide film. Also, the adjustment of the surface area was easier by using disk-shaped specimens. The starting point for the manufacturing of orthodontic wires is the casting of an ingot having the appropriate alloy composition. 1 This ingot is subjected to a series of mechanical reduction operations until the cross section is sufficiently small for wiredrawing, and then, the wiredrawing is performed. 1 Heat treatments are necessary during wire manufacturing to eliminate the extensive work hardening, which occurs during the various stages of mechanical reduction. 1 Previous study has demonstrated that the oxide film on the commercial NiTi orthodontic wire, TABLE 4. Mean Galvanic Current Density at 24, 48, and 72 h ( A/cm 2 ) a Area Ratio Coupled Alloys Mean (Range) at 24 h Mean (Range) at 48 h Mean (Range) at 72 h 1:1 SUS-NiTi ( ) ( ) ( ) SUS-CoCr ( ) ( ) ( ) SUS- -Ti ( ) ( ) ( ) Ti-NiTi ( ) ( ) ( ) Ti-CoCr ( ) ( ) ( ) Ti-SUS ( ) ( ) ( ) Ti- -Ti ( ) 0 ( ) ( ) 1:2:35 SUS-NiTi ( ) ( ) ( ) SUS-CoCr ( ) 0 (0 0) 0 ( ) SUS- -Ti ( ) ( ) ( ) Ti-NiTi ( ) ( ) ( ) Ti-CoCr ( ) ( ) ( ) Ti-SUS ( ) ( ) ( ) Ti- -Ti ( ) 0 (0 0) 0 (0 0) 1:3:64 SUS-NiTi ( ) ( ) ( ) SUS-CoCr ( ) ( ) ( ) SUS- -Ti ( ) ( ) ( ) Ti-NiTi ( ) ( ) ( ) Ti-CoCr ( ) ( ) ( ) Ti-SUS ( ) ( ) ( ) Ti- -Ti ( ) 0 (0 0) 0 (0 0) a NiTi indicates nickel-titanium alloy; CoCr, cobalt-chromium alloy; and -Ti, -titanium alloy.
5 GALVANIC CORROSION OF ORTHODONTIC ALLOYS formed by wire manufacturing processes such as heat treatment and pickling, causes an increase in the corrosion resistance. When this occurs, the amount of released metal ions from the commercial NiTi orthodontic wire was lower than that from the disk-shaped specimen with a polished surface in 0.9% NaCl solution. 7 In another previous study that measured the free corrosion potential in the 0.9% NaCl solution for commercial orthodontic brackets, orthodontic archwire, and coil springs demonstrated that the two piece type brackets made by soldering or welding had a remarkably lower corrosion potential value in comparison with one piece type brackets because the contact point between the wing and the base might be a susceptible site for localized corrosion. 16 Consequently, corrosion behavior of wrought orthodontic brackets and orthodontic archwire with various surface conditions in the real oral environment should be extremely complex and may differ slightly from the results obtained in this study, which used disk-shaped specimens with polished surfaces. However, the tendency of both galvanic corrosion behaviors obtained from disk-shaped specimens with polished surface and that obtained from wrought orthodontic brackets and orthodontic archwires should be similar. In electrochemical corrosion, a galvanic cell is created when two different metals, or different areas on the same metal, are coupled. In galvanic corrosion, some current flows between the anodic and the cathodic areas situated at different parts of a metallic surface or between different metals of the same or different materials. The driving force for corrosion is a potential difference between the different materials. Clinically, mixed alloys having different corrosion potentials are often placed in contact in the oral environment, as with orthodontic brackets and archwires. This can cause galvanic corrosion that leads to preferential release of metal ions from the anodic alloy Another clinically important example of this phenomenon is the concentration cell corrosion that takes place under the oral debris covering a pit on a metallic appliance. Because of the lower concentration of oxygen compared with the bulk oral environment, a rapid attack occurs at the bottom of the pit. 17 The multiplied effect of concentration cell corrosion and galvanic corrosion may accelerate corrosion behavior remarkably. In this study, the galvanic current decreased with time and reached a nearly constant value after 24 hours for all coupled samples (Figures 2 through 4). In NiTi alloy coupled with Ti (1:1, 1:2.35, and 1:3.64 of the surface ratio), Ti was initially anode and corroded; however, the polarity reversed in 1 hour, resulting in corrosion of the NiTi. This phenomenon can be explained by the time change of corrosion potential (E corr ) because the corrosion potential of the Ti was lower 709 than that of NiTi immediately after starting immersion and moved in the noble direction in a few hours. On the other hand, the corrosion potential of the NiTi moved in the less noble direction after the starting immersion (Figure 1). A previous study using X-ray photoelectron spectroscopy (XPS) demonstrated that the thickness of the surface films formed on titanium specimens immersed in electrolyte solutions (ph 4.5, 5.2, 7.4) at 37 C increased during immersion. 18 Similarly, the surface oxide film of Ti, which mainly is composed of TiO 2, was probably aged along with increasing the thickness of the surface oxide film in this study. The result of this was that the corrosion potential of the Ti moved in the noble direction. Another previous study also demonstrated the surface oxide film of the commercial orthodontic NiTi wire aged in the 0.9% NaCl solution and the thickness of the oxide film increased during immersion in the 0.9% NaCl solution. 7 However, the corrosion potential of the NiTi moved in the less noble direction after starting immersion in this study. The XPS demonstrated that the NiTi alloy surface under the passive film was rich in Ni because of a preferential oxidation of Ti. 7 The enrichment of Ni at the alloy/oxide film interface may be related to the decrease in corrosion potential. Further research is necessary to study more details of these contradictory phenomena. The galvanic current density for NiTi alloy coupled with SUS 304 or Ti at 72 hours of immersion was to A/cm 2, which was more than one order of magnitude higher than that observed for the other couples. The reason for this is that there were relatively large differences in the corrosion potentials (E corr ) between the NiTi alloy and the other two alloys in the uncoupled sample. The corrosion rate of uncoupled NiTi in the 0.9% NaCl solution could be approximated from the amount of nickel ions released into the solution using Faraday s law. The average corrosion rate during 28 days thus estimated was 0.54 na/cm However, the galvanic current density obtained in this study at 72 hours of immersion was found to be increased from approximately 80 to 210 times when the NiTi was galvanically coupled with SUS 304 or Ti. It is suggested that coupling SUS 304-NiTi and Ti-NiTi may remarkably accelerate the corrosion of NiTi alloy, which serves as the anode. The surface area ratio of two dissimilar alloys is an extremely important factor because it affects the galvanic corrosion behavior. 11,13,14 An unfavorable area ratio, which consists of a large cathode and a small anode, might lead to the greater corrosion rate of the anodic alloy, 11 possibly causing problems if the anodic alloy contained a high-risk element such as nickel. This study used a computer-aided data analysis system to estimate the surface area of the brackets
6 710 IIJIMA, ENDO, YUASA, OHNO, HAYASHI, KAKIZAKI, MIZOGUCHI accurately. The results obtained show that differing surface area ratios of two dissimilar alloys had little effect on galvanic corrosion behavior. This is in agreement with another recent study that compared corrosion rates of three different ratios of surface areas between 2205 stainless steel, which is a dual phase stainless steel, and SUS 316L. 11 Figure 5 illustrates the effect of anode-cathode area ratio on the anodic and cathodic polarization curves for (1) an actively corroding alloy and (2) a passive alloy when each alloy is galvanically coupled to an alloy that has more positive potential (cathodic). When equal areas of the anodic and cathodic alloys are coupled, the corrosion rate of the active and the passive alloys are I corr,a 1 and I corr,p 1, respectively. The corrosion rate of the active alloy is increased from I corr,a 1 to I corr,a 2 as the area of cathodic alloy is increased from S to 3.64S. In contrast to this, the corrosion current of the passive alloy is not markedly influenced by the surface area of the cathodic alloy because the anodic current of the passive alloy is almost constant, independent of potential in the passive region. Because the alloys used in this study were all passive, the surface area ratio of two dissimilar alloys had little effect on the corrosion rate, as shown in Figure 5b. Although polished disk-shaped specimens were used for brackets and archwire alloys in this study, commercial archwires have a thick oxide film of approximately two nm thickness on the surface as a result of the production processes, such as the heat treatment and pickling processes. This causes an increase in both the general and the localized corrosion resistance. 7,10 Furthermore, the surface of the brackets and archwires could be scratched by bending and ligating clinically. Thus, further investigations are required to determine the galvanic corrosion behavior in clinical applications. CONCLUSIONS In NiTi alloy coupled with Ti for all the surface ratio (1:1, 1:2.35, and 1:3.64), Ti was initially anode and corroded; however, the polarity reversed within one hour, resulting in corrosion of NiTi. It is suggested that coupling SUS 304-NiTi and Ti- NiTi may remarkably accelerate the corrosion of NiTi alloy, which served as the anode, because NiTi alloy coupled with SUS 304 or Ti exhibited more than one order of magnitude higher than that observed for the other couples at 72 hours of immersion. The different anodic-cathodic area ratios used in this study (1:1, 1:2.35, and 1:3.64) had little effect on galvanic corrosion behavior. FIGURE 5. Effect of anode-cathode area ratio on the polarization diagram of two galvanically coupled alloys. (A) Anodic polarization curve of an active alloy with surface area of S cm 2. (B) Anodic polarization curve of a passive alloy with surface area of S cm 2. (C) Cathodic polarization curve when each alloy is coupled to an alloy (S cm 2 ) with relatively noble potential. (D) Cathodic polarization curve when each alloy is coupled to an alloy (3.64 cm 2 ) with relatively noble potential. ACKNOWLEDGMENTS We wish to thank Ormco for providing the nickel-titanium specimen for this investigation. This research was partly supported by a grant-in-aid for scientific research from the Ministry of Education, Culture, Sports, Science and Technology, Japan ( ).
7 GALVANIC CORROSION OF ORTHODONTIC ALLOYS REFERENCES 1. Brantley WA. Orthodontic wires. In: Brantley WA, Eliades T, eds. Orthodontic Materials: Scientific and Clinical Aspects. Stuttgart: Thieme; 2001: Bass JK, Fine HF, Cisneros GJ. Nickel hypersensitivity in the orthodontic patient. Am J Orthod Dentofacial Orthop. 1993;103: Kerosuo H, Kullaa A, Kerosuo E, Kanerva L, Hensten-Pettersen A. Nickel allergy in adolescents in relation to orthodontic treatment and piercing of ears. Am J Orthod Dentofacial Orthop. 1996;109: Dunlap CL, Vincent SK, Barker BF. Allergic reaction to orthodontic wire: report of case. J Am Dent Assoc. 1989;118: Greppi AL, Smith DC, Woodside DG. Nickel hypersensitivity reactions in orthodontic patients. Univ Tor Dent J. 1989;J3: Kerosuo H, Moe G, Kleven E. In vitro release of nickel and chromium from different types of simulated orthodontic appliances. Angle Orthod. 1995;65(2): Iijima M, Endo K, Ohno H, Yonekura Y, Mizoguchi I. Corrosion behavior and surface structure of orthodontic Ni-Ti alloy wires. Dent Mater J. 2001;20(1): Jia W, Beatty MW, Reinhardt RA, Petro TM, Cohen DM, Maze CR, Strom EA, Hoffman M. Nickel release from orthodontic arch wires and cellular immune response to various nickel concentrations. J Biomed Mater Res. 1999;48: Hunt NP, Cunningham SJ, Golden CG, Sheriff M. An investigation into the effect of polishing on surface hardness and 711 corrosion of orthodontic archwires. Angle Orthod. 1999; 69(5): Yonekura Y, Endo K, Iijima M, Ohno H, Mizoguchi I. In vitro corrosion characteristics of commercially available orthodontic wires. Dent Mater J. 2004;23(2): Platt JA, Guzman A, Zuccari A, Thornburg DW, Rhodes BF, Oshida Y, Moore BK. Corrosion behavior of 2205 duplex stainless steel. Am J Orthod Dentofacial Orthop. 1997;112: Venugopalan R, Lucas LC. Evaluation of restorative and implant alloys galvanically coupled to titanium. Dent Mater. 1998;14: Karov J, Hinberg I. Galvanic corrosion of selected dental alloys. J Oral Rehabil. 2001;28: Lim S, Takada Y, Kim K, Okuno O. Ions released from dental amalgams in contact with titanium. Dent Mater J. 2003; 22(1): Trethewey KR, Chamberlain J. Enabling theory for aqueous corrosion. In: Trethewey KR, Chamberlain J, eds. Corrosion for Students of Science and Engineering. New York, NY: John Wiley & Sons Inc; 1988: Yuasa T, Endo K, Iijima M, Yonekura Y, Ohno H, Mizoguchi I. Study of dissimilar metal corrosion of orthodontic metallic appliances corrosion potentials measured in saline solution. Higashi Nippon Dent J. 2004;23: Trethewey KR, Chamberlain J. Crevice and pitting corrosion. In: Trethewey KR, Chamberlain J, eds. Corrosion for Students of Science and Engineering. New York, NY: John Wiley & Sons Inc; 1988: Hanawa T, Ota M. Calcium phosphate naturally formed on titanium in electrolyte solution. Biomaterials. 1991;12(8):
INFLUENCE OF ARTIFICIAL SALIVA ON NiTi ORTHODONTIC WIRES: A STUDY ON THE SURFACE CHARACTERIZATION
 Materials Science, Vol. 47, No. 6, May, 2012 (Ukrainian Original Vol. 47, No. 6, November December, 2011) INFLUENCE OF ARTIFICIAL SALIVA ON NiTi ORTHODONTIC WIRES: A STUDY ON THE SURFACE CHARACTERIZATION
Materials Science, Vol. 47, No. 6, May, 2012 (Ukrainian Original Vol. 47, No. 6, November December, 2011) INFLUENCE OF ARTIFICIAL SALIVA ON NiTi ORTHODONTIC WIRES: A STUDY ON THE SURFACE CHARACTERIZATION
Assessment of nickel titanium and beta titanium corrosion resistance behavior in fluoride and chloride environments
 Original Article Assessment of nickel titanium and beta titanium corrosion resistance behavior in fluoride and chloride environments Elisa J. Kassab a ; José Ponciano Gomes b ABSTRACT Objective: To assess
Original Article Assessment of nickel titanium and beta titanium corrosion resistance behavior in fluoride and chloride environments Elisa J. Kassab a ; José Ponciano Gomes b ABSTRACT Objective: To assess
Galvanic Corrosion among Different Combination of Orthodontic Archwires and Stainless Steel Brackets
 Original Research Galvanic Corrosion among Different Combination of Orthodontic Archwires and Stainless Steel Brackets Farzin Heravi 1, Nima Mokhber 2, Elnaz Shayan 3 1 Dental Materials Research Center,
Original Research Galvanic Corrosion among Different Combination of Orthodontic Archwires and Stainless Steel Brackets Farzin Heravi 1, Nima Mokhber 2, Elnaz Shayan 3 1 Dental Materials Research Center,
An Accurate Methodology to detect Leaching of Nickel and Chromium Ions in the Initial Phase of Orthodontic Treatment: An in vivo Study
 JCDP ORIGINAL RESEARCH Leaching 10.5005/jp-journals-10024-1828 of Nickel and Chromium Ions An Accurate Methodology to detect Leaching of Nickel and Chromium Ions in the Initial Phase of Orthodontic Treatment:
JCDP ORIGINAL RESEARCH Leaching 10.5005/jp-journals-10024-1828 of Nickel and Chromium Ions An Accurate Methodology to detect Leaching of Nickel and Chromium Ions in the Initial Phase of Orthodontic Treatment:
GAC Archwire Collection
 GAC Archwire Collection All the Metal That Matters Copper to Nickel and All Points In-Between Your choice in archwires is a critical decision in your quest for excellence. GAC has been recognized as a
GAC Archwire Collection All the Metal That Matters Copper to Nickel and All Points In-Between Your choice in archwires is a critical decision in your quest for excellence. GAC has been recognized as a
Assessing the corrosion behaviour of Nitinol for minimally-invasive device design
 Min Invas Ther & Allied Technol 2000: 9(2) 67 74 Assessing the corrosion behaviour of Nitinol for minimally-invasive device design R. Venugopalan 1 and C. Trépanier 2 1 Department of Biomedical Engineering,
Min Invas Ther & Allied Technol 2000: 9(2) 67 74 Assessing the corrosion behaviour of Nitinol for minimally-invasive device design R. Venugopalan 1 and C. Trépanier 2 1 Department of Biomedical Engineering,
wiirre e a a n n d d w wiirre e ffo orrms ms
 wire and wire forms wire and wire forms wire and wire forms Leone orthodontic wires are available in a range of alloys and different grades of elasticity and hardness to meet any therapeutic requirements.
wire and wire forms wire and wire forms wire and wire forms Leone orthodontic wires are available in a range of alloys and different grades of elasticity and hardness to meet any therapeutic requirements.
Corrosion Inhibition of Titanium in Artificial Saliva Containing Fluoride
 Leonardo Journal of Sciences ISSN 1583-0233 Issue 11, July-December 2007 p. 33-40 Corrosion Inhibition of Titanium in Artificial Saliva Containing Fluoride Latifa KINANI and Abdelilah CHTAINI* Equipe d
Leonardo Journal of Sciences ISSN 1583-0233 Issue 11, July-December 2007 p. 33-40 Corrosion Inhibition of Titanium in Artificial Saliva Containing Fluoride Latifa KINANI and Abdelilah CHTAINI* Equipe d
Effects of a diamond-like carbon coating on the frictional properties of orthodontic wires
 Original Article Effects of a diamond-like carbon coating on the frictional properties of orthodontic wires Takeshi Muguruma a ; Masahiro Iijima b ; William A. Brantley c ; Itaru Mizoguchi d ABSTRACT Objective:
Original Article Effects of a diamond-like carbon coating on the frictional properties of orthodontic wires Takeshi Muguruma a ; Masahiro Iijima b ; William A. Brantley c ; Itaru Mizoguchi d ABSTRACT Objective:
The management of impacted
 Using a rigid hook and spring auxiliary slid onto the archwire to direct eruption of impacted teeth BY S. JAY BOWMAN, DMD, MSD, AND ALDO CARANO, DR ODONT, MS, SPEC ORTHOD Figure 1: A 12-year-old female
Using a rigid hook and spring auxiliary slid onto the archwire to direct eruption of impacted teeth BY S. JAY BOWMAN, DMD, MSD, AND ALDO CARANO, DR ODONT, MS, SPEC ORTHOD Figure 1: A 12-year-old female
Comparative analysis of load/deflection ratios of conventional and heat-activated rectangular NiTi wires
 Comparative analysis of load/deflection ratios of conventional and heat-activated rectangular NiTi wires Fabio Schemann-Miguel 1, Flávio Cotrim-Ferreira 2, Alessandra Motta Streva 3, Alexander Viégas de
Comparative analysis of load/deflection ratios of conventional and heat-activated rectangular NiTi wires Fabio Schemann-Miguel 1, Flávio Cotrim-Ferreira 2, Alessandra Motta Streva 3, Alexander Viégas de
An Effectiv Rapid Molar Derotation: Keles K
 An Effectiv ective e and Precise Method forf Rapid Molar Derotation: Keles K TPA Ahmet Keles, DDS, DMSc 1 /Sedef Impar, DDS 2 Most of the time, Class II molar relationships occur due to the mesiopalatal
An Effectiv ective e and Precise Method forf Rapid Molar Derotation: Keles K TPA Ahmet Keles, DDS, DMSc 1 /Sedef Impar, DDS 2 Most of the time, Class II molar relationships occur due to the mesiopalatal
Effect of Fluoride on Nickel-Titanium and Stainless Steel. Orthodontic Archwires: An In-Vitro Study
 Original Article Effect of Fluoride on Nickel-Titanium and Stainless Steel Orthodontic Archwires: An In-Vitro Study Farzin Heravi 1, Mohamad Hadi Moayed 2, Nima Mokhber 3 1 Department of Orthodontics,School
Original Article Effect of Fluoride on Nickel-Titanium and Stainless Steel Orthodontic Archwires: An In-Vitro Study Farzin Heravi 1, Mohamad Hadi Moayed 2, Nima Mokhber 3 1 Department of Orthodontics,School
Effects of CO 2 laser debonding of a ceramic bracket on the mechanical properties of enamel
 Original Article Effects of CO 2 laser debonding of a ceramic bracket on the mechanical properties of enamel Masahiro Iijima a ; Yoshitaka Yasuda b ; Takeshi Muguruma c ; Itaru Mizoguchi d ABSTRACT Objective:
Original Article Effects of CO 2 laser debonding of a ceramic bracket on the mechanical properties of enamel Masahiro Iijima a ; Yoshitaka Yasuda b ; Takeshi Muguruma c ; Itaru Mizoguchi d ABSTRACT Objective:
History of NiTinol. Ni = Nickel Ti = Titanium N = Naval O = Ordnance L = Laboratory
 History of NiTinol Ni = Nickel Ti = Titanium N = Naval O = Ordnance L = Laboratory NiTinol was discovered in a laboratory of the U.S. Marines in 1962 (US-Naval Ordnance Laboratory). The U.S. Marines wanted
History of NiTinol Ni = Nickel Ti = Titanium N = Naval O = Ordnance L = Laboratory NiTinol was discovered in a laboratory of the U.S. Marines in 1962 (US-Naval Ordnance Laboratory). The U.S. Marines wanted
Measurements of the torque moment in various archwire bracket ligation combinations
 European Journal of Orthodontics 34 (2012) 374 380 doi:10.1093/ejo/cjr022 Advance Access Publication 13 May 2011 The Author 2011. Published by Oxford University Press on behalf of the European Orthodontic
European Journal of Orthodontics 34 (2012) 374 380 doi:10.1093/ejo/cjr022 Advance Access Publication 13 May 2011 The Author 2011. Published by Oxford University Press on behalf of the European Orthodontic
Faculty of Dentistry, National Yang-Ming University, Taipei, Taiwan, ROC.
 Original Articles Corrosion behavior of titanium-containing orthodontic archwires in artificial saliva: effects of fluoride ions and plasma immersion ion implantation treatment HER-HSIUNG HUANG 1 CHIA-CHING
Original Articles Corrosion behavior of titanium-containing orthodontic archwires in artificial saliva: effects of fluoride ions and plasma immersion ion implantation treatment HER-HSIUNG HUANG 1 CHIA-CHING
With judicious treatment planning, the clinical
 CLINICIAN S CORNER Selecting custom torque prescriptions for the straight-wire appliance Earl Johnson San Francisco, Calif Selecting custom torque prescriptions based on the treatment needs of each patient
CLINICIAN S CORNER Selecting custom torque prescriptions for the straight-wire appliance Earl Johnson San Francisco, Calif Selecting custom torque prescriptions based on the treatment needs of each patient
Dental Materials Journal 2015; 34(3): Keisuke TOCHIGI, Souichiro ODA and Kazuhito ARAI INTRODUCTION
 Dental Materials Journal 2015; 34(3): 388 393 Influences of archwire size and ligation method on the force magnitude delivered by nickel-titanium alloy archwires in a simulation of mandibular right lateral
Dental Materials Journal 2015; 34(3): 388 393 Influences of archwire size and ligation method on the force magnitude delivered by nickel-titanium alloy archwires in a simulation of mandibular right lateral
Electrochemical Properties of Suprastructures Galvanically Coupled to a Titanium Implant
 Electrochemical Properties of Suprastructures Galvanically Coupled to a Titanium Implant Keun-Taek Oh, Kyoung-Nam Kim Department of Dental Biomaterials and Bioengineering and Research Center for Orofacial
Electrochemical Properties of Suprastructures Galvanically Coupled to a Titanium Implant Keun-Taek Oh, Kyoung-Nam Kim Department of Dental Biomaterials and Bioengineering and Research Center for Orofacial
Nickel and Chromium Ions Release from Stainless Steel Bracket Immersed in Fluoridated Mouthwash
 from Stainless Steel Bracket Immersed in Fluoridated Mouthwash Ida Bagus Narmada 1 *, Ria Anbar Baya 2, Thalca Hamid 3 1. Departement of Orthodontics, Faculty of Dental Medicine, Universitas Airlangga,
from Stainless Steel Bracket Immersed in Fluoridated Mouthwash Ida Bagus Narmada 1 *, Ria Anbar Baya 2, Thalca Hamid 3 1. Departement of Orthodontics, Faculty of Dental Medicine, Universitas Airlangga,
Valeri Petrov 1, Ivana Ilievska 2, Laura Andreeva 1, Valdek Mikli 3, Andrzej Zaleski 4, Angelina Stoyanova-Ivanova 2
 DOI: 10.18044/Medinform.201741.559 Studying the Effects of Autoclaving Process on Orthodontic Archwires Valeri Petrov 1, Ivana Ilievska 2, Laura Andreeva 1, Valdek Mikli 3, Andrzej Zaleski 4, Angelina
DOI: 10.18044/Medinform.201741.559 Studying the Effects of Autoclaving Process on Orthodontic Archwires Valeri Petrov 1, Ivana Ilievska 2, Laura Andreeva 1, Valdek Mikli 3, Andrzej Zaleski 4, Angelina
Three-Dimensional Analysis of Dental Casts Based on a Newly Defined Palatal Reference Plane
 Original Article Three-Dimensional Analysis of Dental Casts Based on a Newly Defined Palatal Reference Plane Kazuo Hayashi, DDS, PhD a ; Jun Uechi, DDS a ; Itaru Mizoguchi, DDS, PhD a Abstract: The purpose
Original Article Three-Dimensional Analysis of Dental Casts Based on a Newly Defined Palatal Reference Plane Kazuo Hayashi, DDS, PhD a ; Jun Uechi, DDS a ; Itaru Mizoguchi, DDS, PhD a Abstract: The purpose
R J M E Romanian Journal of Morphology & Embryology
 Rom J Morphol Embryol 2015, 56(3):1119 1125 ORIGINAL PAPER R J M E Romanian Journal of Morphology & Embryology http://www.rjme.ro/ Assessment of orthodontic biomaterials cytotoxicity: an in vitro study
Rom J Morphol Embryol 2015, 56(3):1119 1125 ORIGINAL PAPER R J M E Romanian Journal of Morphology & Embryology http://www.rjme.ro/ Assessment of orthodontic biomaterials cytotoxicity: an in vitro study
SE NiTi Heat Activated NiTi Ultra T Heat Activated NiTi Stainless Steel Beta Titanium. Ultra T Archwires
 S1:1 170421 7390 SOLITAIRE Single Pack s OrthoQuest offers the highest quality wires available in the orthodontic industry. Our wires are designed to exact specifications by trained engineers and manufactured
S1:1 170421 7390 SOLITAIRE Single Pack s OrthoQuest offers the highest quality wires available in the orthodontic industry. Our wires are designed to exact specifications by trained engineers and manufactured
Experience with Contemporary Tip-Edge plus Technique A Case Report.
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861. Volume 13, Issue 3 Ver. I. (Mar. 2014), PP 12-17 Experience with Contemporary Tip-Edge plus Technique A Case
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861. Volume 13, Issue 3 Ver. I. (Mar. 2014), PP 12-17 Experience with Contemporary Tip-Edge plus Technique A Case
Effect of surface treatment with commercial primers on tensile bond strength of auto-polymerizing resin to magnetic stainless steel
 Effect of surface treatment with commercial primers on tensile bond strength of auto-polymerizing resin to magnetic stainless steel Jian-rong Chen, DDS, a,b Kenji Oka, DDS, PhD, c Wei Hua, DDS, a and Tetsuo
Effect of surface treatment with commercial primers on tensile bond strength of auto-polymerizing resin to magnetic stainless steel Jian-rong Chen, DDS, a,b Kenji Oka, DDS, PhD, c Wei Hua, DDS, a and Tetsuo
Anchorage system. tomas / Sets Page 270 tomas / Pins Page 271 Instruments and accessories Page 273 Patient consultation material Page 282
 tomas / Sets Page 270 tomas / Pins Page 271 Instruments and accessories Page 273 Patient consultation material Page 282 266 . innovative comprehensive efficient. Dentaurum Online Shop shop.dentaurum.com
tomas / Sets Page 270 tomas / Pins Page 271 Instruments and accessories Page 273 Patient consultation material Page 282 266 . innovative comprehensive efficient. Dentaurum Online Shop shop.dentaurum.com
of the strain and of the temperature. Superelasticity in Orthodontics was first described for Japanese NiTi, which
 ISSN: 0975-766X CODEN: IJPTFI Available Online through Research Article www.ijptonline.com SUPERELASTICITY OF NiTi Niha Naveed*, Dr. Sri Rengalakshmi Final Year BDS, Department of Orthodontics, Saveetha
ISSN: 0975-766X CODEN: IJPTFI Available Online through Research Article www.ijptonline.com SUPERELASTICITY OF NiTi Niha Naveed*, Dr. Sri Rengalakshmi Final Year BDS, Department of Orthodontics, Saveetha
Acknowledgments Introduction p. 1 Objectives p. 1 Goals p. 2 History of Dental Materials p. 3 The Oral Environment p. 4 Characteristics of the Ideal
 Preface p. v Acknowledgments p. vii Introduction p. 1 Objectives p. 1 Goals p. 2 History of Dental Materials p. 3 The Oral Environment p. 4 Characteristics of the Ideal Dental Material p. 5 Quality Assurance
Preface p. v Acknowledgments p. vii Introduction p. 1 Objectives p. 1 Goals p. 2 History of Dental Materials p. 3 The Oral Environment p. 4 Characteristics of the Ideal Dental Material p. 5 Quality Assurance
3M Incognito Appliance System extraction case study.
 SM 3M Health Care Academy 3M Incognito Appliance System extraction case study. Toru Inami, DDS, Ph.D. Dr. Toru Inami graduated from the Aichigakuin University School of Dentistry in 1976. From 1977 to
SM 3M Health Care Academy 3M Incognito Appliance System extraction case study. Toru Inami, DDS, Ph.D. Dr. Toru Inami graduated from the Aichigakuin University School of Dentistry in 1976. From 1977 to
A Severe Reaction to Ni-Containing Orthodontic Appliances
 Case Report A Severe Reaction to Ni-Containing Orthodontic Appliances Olga Elpis Kolokitha a ; Evangelia Chatzistavrou b ABSTRACT Exposure to nickel-containing orthodontic appliances may cause intra- or
Case Report A Severe Reaction to Ni-Containing Orthodontic Appliances Olga Elpis Kolokitha a ; Evangelia Chatzistavrou b ABSTRACT Exposure to nickel-containing orthodontic appliances may cause intra- or
Treatment of Class II, Division 2 Malocclusion with Miniscrew Supported En-Masse Retraction: Is Deepbite Really an Obstacle for Extraction Treatment?
 TURKISH JOURNAL of DOI: 10.5152/TurkJOrthod.2017.17034 CASE REPORT Treatment of Class II, Division 2 Malocclusion with Miniscrew Supported En-Masse Retraction: Is Deepbite Really an Obstacle for Extraction
TURKISH JOURNAL of DOI: 10.5152/TurkJOrthod.2017.17034 CASE REPORT Treatment of Class II, Division 2 Malocclusion with Miniscrew Supported En-Masse Retraction: Is Deepbite Really an Obstacle for Extraction
Archwires & Accessories
 Archwires & Accessories Orthodontic products-catalog 01 55 Archwires and Accessories Summary Archwire Shapes 58 Initialloy 59 BioEdge 60 BioActive 61 Reverse Curve 62 Beta Titanium 62 Stainless Steel Archwires
Archwires & Accessories Orthodontic products-catalog 01 55 Archwires and Accessories Summary Archwire Shapes 58 Initialloy 59 BioEdge 60 BioActive 61 Reverse Curve 62 Beta Titanium 62 Stainless Steel Archwires
Controlled Space Closure with a Statically Determinate Retraction System
 Original Article Controlled Space Closure with a Statically Determinate Retraction System Kwangchul Choy, DDS, MS, PhD a ; Eung-Kwon Pae, DDS, MSc, PhD b ; Kyung-Ho Kim, DDS, MS, PhD c ; Young Chel Park,
Original Article Controlled Space Closure with a Statically Determinate Retraction System Kwangchul Choy, DDS, MS, PhD a ; Eung-Kwon Pae, DDS, MSc, PhD b ; Kyung-Ho Kim, DDS, MS, PhD c ; Young Chel Park,
A New Fixed Interarch Device for Class II Correction
 A New Fixed Interarch Device for Class II Correction WILLIAM VOGT, DDS Fixed devices are increasingly being used for molar distalization in Class II treatment because they eliminate the need for special
A New Fixed Interarch Device for Class II Correction WILLIAM VOGT, DDS Fixed devices are increasingly being used for molar distalization in Class II treatment because they eliminate the need for special
Canine Extrusion Technique with SmartClip Self-Ligating Brackets
 Canine Extrusion Technique with SmartClip Self-Ligating Brackets Dr. Luis Huanca Ghislanzoni Dr. Luis Huanca received his DDS in 2006 and the MS and Specialist in Orthodontics in 2009 from the University
Canine Extrusion Technique with SmartClip Self-Ligating Brackets Dr. Luis Huanca Ghislanzoni Dr. Luis Huanca received his DDS in 2006 and the MS and Specialist in Orthodontics in 2009 from the University
Mandibular Protraction Appliance IV
 CARLOS M. COELHO FILHO, DDS, MSD Articles published by Pancherz 1,2 on the Herbst* appliance in the last two decades seem to have stimulated the development of several other fixed appliances that work
CARLOS M. COELHO FILHO, DDS, MSD Articles published by Pancherz 1,2 on the Herbst* appliance in the last two decades seem to have stimulated the development of several other fixed appliances that work
INDICATIONS. Fixed Appliances are indicated when precise tooth movements are required
 DEFINITION Fixed Appliances are devices or equipments that are attached to the teeth, cannot be removed by the patient and are capable of causing tooth movement. INDICATIONS Fixed Appliances are indicated
DEFINITION Fixed Appliances are devices or equipments that are attached to the teeth, cannot be removed by the patient and are capable of causing tooth movement. INDICATIONS Fixed Appliances are indicated
ORTHOdontics SLIDING MECHANICS
 ORTHOdontics PGI/II SLIDING MECHANICS FOCUS ON TARGETED SPACE GAINING AND ITS APPLICATIONS, INCLUDING WITH RAPID PALATAL EXPANDIONS. ALSO INCLUDES RETENTION AND CLINICAL PEARLS FACULTY: Joseph Ghafari,
ORTHOdontics PGI/II SLIDING MECHANICS FOCUS ON TARGETED SPACE GAINING AND ITS APPLICATIONS, INCLUDING WITH RAPID PALATAL EXPANDIONS. ALSO INCLUDES RETENTION AND CLINICAL PEARLS FACULTY: Joseph Ghafari,
Use of a Tip-Edge Stage-1 Wire to Enhance Vertical Control During Straight Wire Treatment: Two Case Reports
 Case Report Use of a Tip-Edge Stage-1 Wire to Enhance Vertical Control During Straight Wire Treatment: Two Case Reports Helen Taylor, BDS, MScD, DOrth, MOrth, FDSRCS(Eng) a Abstract: Vertical control is
Case Report Use of a Tip-Edge Stage-1 Wire to Enhance Vertical Control During Straight Wire Treatment: Two Case Reports Helen Taylor, BDS, MScD, DOrth, MOrth, FDSRCS(Eng) a Abstract: Vertical control is
Treatment of an open bite case with 3M Clarity ADVANCED Ceramic Brackets and miniscrews.
 SM 3M Health Care Academy Treatment of an open bite case with 3M Clarity ADVANCED Ceramic Brackets and miniscrews. Dr. J.C. Pérez-Varela MD, DDS, MS, Ph.D. Specialist in Orthodontics. Doctor of Medicine
SM 3M Health Care Academy Treatment of an open bite case with 3M Clarity ADVANCED Ceramic Brackets and miniscrews. Dr. J.C. Pérez-Varela MD, DDS, MS, Ph.D. Specialist in Orthodontics. Doctor of Medicine
ORTHODONTIC WIRES. 1. Commercial name: Contour NiTi Memoflex NiTi Cuper NiTi Stainless Steel TMA Archwire Sequence Springs
 PAGE 1 of 6 1. Commercial name: Contour NiTi Memoflex NiTi Cuper NiTi Stainless Steel TMA Archwire Sequence Springs 2. Manufacturer: Aditek do Brasil Ltda. Address: Cesário Mota, 14 ZIP: 14140-000 - Cravinhos
PAGE 1 of 6 1. Commercial name: Contour NiTi Memoflex NiTi Cuper NiTi Stainless Steel TMA Archwire Sequence Springs 2. Manufacturer: Aditek do Brasil Ltda. Address: Cesário Mota, 14 ZIP: 14140-000 - Cravinhos
Surface Protection of Niti- Shape Memory Alloy by Colgate Visible White Toothpaste
 International Journal of ChemTech Research CODEN (USA): IJCRGG, ISSN: 0974-4290, ISSN(Online):2455-9555 Vol.10 No.5, pp 761-769, 2017 Surface Protection of Niti- Shape Memory Alloy by Colgate Visible White
International Journal of ChemTech Research CODEN (USA): IJCRGG, ISSN: 0974-4290, ISSN(Online):2455-9555 Vol.10 No.5, pp 761-769, 2017 Surface Protection of Niti- Shape Memory Alloy by Colgate Visible White
In vitro Assessment of Clasps of Cobalt-Chromium and Nickel-titanium Alloys in Removable Prosthesis
 ORIGINAL RESEARCH Cobalt-Chromium 10.5005/jp-journals-10024-1836 and Nickel-titanium Clasps In vitro Assessment of Clasps of Cobalt-Chromium and Nickel-titanium Alloys in Removable Prosthesis 1 Mohammed
ORIGINAL RESEARCH Cobalt-Chromium 10.5005/jp-journals-10024-1836 and Nickel-titanium Clasps In vitro Assessment of Clasps of Cobalt-Chromium and Nickel-titanium Alloys in Removable Prosthesis 1 Mohammed
Load-Deflection Characteristics of Superelastic Nickel-Titanium Wires
 Original Article Load-Deflection Characteristics of Superelastic Nickel-Titanium Wires Theodosia N. Bartzela a ; Christiane Senn b ; Andrea Wichelhaus c ABSTRACT Objective: To determine the mechanical
Original Article Load-Deflection Characteristics of Superelastic Nickel-Titanium Wires Theodosia N. Bartzela a ; Christiane Senn b ; Andrea Wichelhaus c ABSTRACT Objective: To determine the mechanical
Integrative Orthodontics with the Ribbon Arch By Larry W. White, D.D.S., M.S.D.
 Integrative Orthodontics with the Ribbon Arch By Larry W. White, D.D.S., M.S.D. Abstract The ribbon arch previously had great popularity and utility early in the 20 th century, but lost its appeal as edgewise
Integrative Orthodontics with the Ribbon Arch By Larry W. White, D.D.S., M.S.D. Abstract The ribbon arch previously had great popularity and utility early in the 20 th century, but lost its appeal as edgewise
Department of Orthodontics, Faculty of Dentistry, Mahidol University.
 Dental Journal Tensile properties of general purpose stainless steel wire formed for orthodontic use Pornkiat Churnjitapirom 1, Surachai Dechkunakorn 2, Niwat Anuwongnukroh 2 1 Research Office, Faculty
Dental Journal Tensile properties of general purpose stainless steel wire formed for orthodontic use Pornkiat Churnjitapirom 1, Surachai Dechkunakorn 2, Niwat Anuwongnukroh 2 1 Research Office, Faculty
6. Timing for orthodontic force
 6. Timing for orthodontic force Orthodontic force is generally less than 300gm, so early mechanical stability is enough for immediate orthodontic force. There is no actually difference in success rate
6. Timing for orthodontic force Orthodontic force is generally less than 300gm, so early mechanical stability is enough for immediate orthodontic force. There is no actually difference in success rate
(12) Patent Application Publication (10) Pub. No.: US 2005/ A1
 (19) United States US 2005O130094A1 (12) Patent Application Publication (10) Pub. No.: US 2005/0130094A1 Graham (43) Pub. Date: Jun. 16, 2005 (54) ORTHODONTIC ACCESSORY ARCH BAR (52) U.S. Cl.... 433/20
(19) United States US 2005O130094A1 (12) Patent Application Publication (10) Pub. No.: US 2005/0130094A1 Graham (43) Pub. Date: Jun. 16, 2005 (54) ORTHODONTIC ACCESSORY ARCH BAR (52) U.S. Cl.... 433/20
Delta Force. Bracket System. Putting you in the driver s seat for ultimate control
 Delta Force Bracket System Putting you in the driver s seat for ultimate control Variable Force Orthodontics The Delta Force Bracket System incorporates an advanced design that allows you to control the
Delta Force Bracket System Putting you in the driver s seat for ultimate control Variable Force Orthodontics The Delta Force Bracket System incorporates an advanced design that allows you to control the
Review of Orthodontic Archwires
 Chapter 2 Review of Orthodontic Archwires 2.1 Introduction The archwire, through mechanical interaction with the bracket slots, are designed to move teeth from malocclusion to a preferred dental occlusion.
Chapter 2 Review of Orthodontic Archwires 2.1 Introduction The archwire, through mechanical interaction with the bracket slots, are designed to move teeth from malocclusion to a preferred dental occlusion.
The Rondeau System. Brackets, Bands, Buccal Tubes and Archwires. Orthodontic supplies recommended by Dr. Brock Rondeau
 The Rondeau System Brackets, Bands, Buccal Tubes and Archwires Orthodontic supplies recommended by Dr. Brock Rondeau Achieve Consistently Accurate Placement The Di-MIM Mini-Twin Bracket s rhomboid shape
The Rondeau System Brackets, Bands, Buccal Tubes and Archwires Orthodontic supplies recommended by Dr. Brock Rondeau Achieve Consistently Accurate Placement The Di-MIM Mini-Twin Bracket s rhomboid shape
Department of Industrial Chemistry, Pukyong National University, Busan 48547, Republic of Korea
 Met. Mater. Int., Vol. 22, No. 5 (2016), pp. 781~788 doi: 10.1007/s12540-016-6124-4 Galvanic Corrosion of Cu Coupled to Au on a Print Circuit Board; Effects of Pretreatment Solution and Etchant Concentration
Met. Mater. Int., Vol. 22, No. 5 (2016), pp. 781~788 doi: 10.1007/s12540-016-6124-4 Galvanic Corrosion of Cu Coupled to Au on a Print Circuit Board; Effects of Pretreatment Solution and Etchant Concentration
LINGUAL STRAIGHTWIRE BRACKET SYSTEM Designed with Drs. Kyoto Takemoto and Giuseppe Scuzzo
 LINGUAL STRAIGHTWIRE BRACKET SYSTEM Designed with Drs. Kyoto Takemoto and Giuseppe Scuzzo THE WORLD S FIRST SELF-LIGATING SQUARE SLOT BRACKET Simplicity I Efficiency I Comfort ALIAS LINGUAL BRACKET SYSTEM
LINGUAL STRAIGHTWIRE BRACKET SYSTEM Designed with Drs. Kyoto Takemoto and Giuseppe Scuzzo THE WORLD S FIRST SELF-LIGATING SQUARE SLOT BRACKET Simplicity I Efficiency I Comfort ALIAS LINGUAL BRACKET SYSTEM
The Tip-Edge appliance and
 Figure 1: Internal surfaces of the edgewise archwire slot are modified to create the Tip-Edge archwire slot. Tipping surfaces (T) limit crown tipping during retraction. Uprighting surfaces (U) control
Figure 1: Internal surfaces of the edgewise archwire slot are modified to create the Tip-Edge archwire slot. Tipping surfaces (T) limit crown tipping during retraction. Uprighting surfaces (U) control
Effect of Sodium Fluoride Mouthwash on the Frictional Resistance of Orthodontic Wires
 Original Article Effect of Sodium Fluoride Mouthwash on the Frictional Resistance of Orthodontic Wires Allahyar Geramy 1, Tabassom Hooshmand 2, Tahura Etezadi 3 1 Professor, Dental Research Center, Dentistry
Original Article Effect of Sodium Fluoride Mouthwash on the Frictional Resistance of Orthodontic Wires Allahyar Geramy 1, Tabassom Hooshmand 2, Tahura Etezadi 3 1 Professor, Dental Research Center, Dentistry
ISW for the treatment of moderate crowding dentition with unilateral second molar impaction
 International Research Journal of Public and Environmental Health Vol.5 (6),pp. 90-103, September 2018 Available online at https://www.journalissues.org/irjpeh/ https://doi.org/10.15739/irjpeh.18.013 Copyright
International Research Journal of Public and Environmental Health Vol.5 (6),pp. 90-103, September 2018 Available online at https://www.journalissues.org/irjpeh/ https://doi.org/10.15739/irjpeh.18.013 Copyright
eral Maxillary y Molar Distalization with Sliding Mechanics: Keles Slider
 Bilater eral Maxillary y Molar Distalization with Sliding Mechanics: Keles Slider Ahmet Keles, DDS, DMSc 1 /Binnur Pamukcu, DDS 2 /Ebru Cetinkaya Tokmak, DDS 2 Aim: To introduce a new intraoral appliance
Bilater eral Maxillary y Molar Distalization with Sliding Mechanics: Keles Slider Ahmet Keles, DDS, DMSc 1 /Binnur Pamukcu, DDS 2 /Ebru Cetinkaya Tokmak, DDS 2 Aim: To introduce a new intraoral appliance
rocky mountain orthodontics functionaleducation
 rocky mountain orthodontics 3D 1ST PHASE FIXED/REMOVABLE (WILSON ) Without changing your chosen technique, RMO 3D 1st Phase FIXED/REMOVABLE Modular Orthodontics TM (Wilson ) can help your practice target
rocky mountain orthodontics 3D 1ST PHASE FIXED/REMOVABLE (WILSON ) Without changing your chosen technique, RMO 3D 1st Phase FIXED/REMOVABLE Modular Orthodontics TM (Wilson ) can help your practice target
Frictional Forces of a Fixed, Esthetic Orthodontic Appliance System
 UNLV Theses, Dissertations, Professional Papers, and Capstones 12-1-2012 Frictional Forces of a Fixed, Esthetic Orthodontic Appliance System Brady Michael Nance University of Nevada, Las Vegas, brady.nance@sdm.unlv.edu
UNLV Theses, Dissertations, Professional Papers, and Capstones 12-1-2012 Frictional Forces of a Fixed, Esthetic Orthodontic Appliance System Brady Michael Nance University of Nevada, Las Vegas, brady.nance@sdm.unlv.edu
Orthodontic bracket slot dimensions as measured from entire bracket series
 Original Article Orthodontic bracket slot dimensions as measured from entire bracket series Paul Brown a ; Warren Wagner b ; Hyden Choi c ABSTRACT Objective: To measure the slot dimensions of an entire
Original Article Orthodontic bracket slot dimensions as measured from entire bracket series Paul Brown a ; Warren Wagner b ; Hyden Choi c ABSTRACT Objective: To measure the slot dimensions of an entire
Keeping all these knowledge in mind I will show you 3 cases treated with the Forsus appliance.
 Due to technical difficulties there were some audio problems with the webinar recording. Starting at 27:54, please use this guide to follow along with Dr. Kercelli s presentation. Keeping all these knowledge
Due to technical difficulties there were some audio problems with the webinar recording. Starting at 27:54, please use this guide to follow along with Dr. Kercelli s presentation. Keeping all these knowledge
TREE ESSENTIAL ELEMENT. ZINC (Zn)
 Pub. No. 17 April 2016 TREE ESSENTIAL ELEMENT ZINC (Zn) By Dr. Kim D. Coder, Professor of Tree Biology & Health Care Warnell School of Forestry & Natural Resources, University of Georgia Zinc (Zn) is a
Pub. No. 17 April 2016 TREE ESSENTIAL ELEMENT ZINC (Zn) By Dr. Kim D. Coder, Professor of Tree Biology & Health Care Warnell School of Forestry & Natural Resources, University of Georgia Zinc (Zn) is a
essentialstm cutting your consumable costs brackets tubes wires springs instruments T:
 2017 essentialstm cutting your consumable costs brackets tubes wires springs instruments T: W: www.tocdental.com www.tocdental.com 1 Value Stainless Steel Brackets Manufactured from advanced 17-4 stainless
2017 essentialstm cutting your consumable costs brackets tubes wires springs instruments T: W: www.tocdental.com www.tocdental.com 1 Value Stainless Steel Brackets Manufactured from advanced 17-4 stainless
A finite element analysis of the effects of archwire size on orthodontic tooth movement in extraction space closure with miniscrew sliding mechanics
 Kawamura and Tamaya Progress in Orthodontics (2019) 20:3 https://doi.org/10.1186/s40510-018-0255-8 RESEARCH Open Access A finite element analysis of the effects of archwire size on orthodontic tooth movement
Kawamura and Tamaya Progress in Orthodontics (2019) 20:3 https://doi.org/10.1186/s40510-018-0255-8 RESEARCH Open Access A finite element analysis of the effects of archwire size on orthodontic tooth movement
Tahereh Hosseinzadeh Nik 1, Tabassom Hooshmand 2*, Habibeh Farazdaghi 3, Arash Mehrabi 1 and Elham S Emadian Razavi 1
 Hosseinzadeh Nik et al. Progress in Orthodontics 2013, 14:48 RESEARCH Open Access Effect of chlorhexidine-containing prophylactic agent on the surface characterization and frictional resistance between
Hosseinzadeh Nik et al. Progress in Orthodontics 2013, 14:48 RESEARCH Open Access Effect of chlorhexidine-containing prophylactic agent on the surface characterization and frictional resistance between
The Loading And Unloading Properties Of Various Arch Wires As A Function Of Cross Sectional Dimension And Inter Bracket Span Width System
 The Loading And Unloading roperties Of Various Arch Wires As A Function Of Cross Sectional Dimension And Inter Bracket Span Width System Authors: Thomas Mathew MDS Orthodontics, Lecturer, Faculty of Dentistry,
The Loading And Unloading roperties Of Various Arch Wires As A Function Of Cross Sectional Dimension And Inter Bracket Span Width System Authors: Thomas Mathew MDS Orthodontics, Lecturer, Faculty of Dentistry,
A randomized clinical trial to compare three methods of orthodontic space closure
 Journal of Orthodontics, Vol. 29, 2002, 31 36 SCIENTIFIC SECTION A randomized clinical trial to compare three methods of orthodontic space closure V. Dixon, M. J. F. Read, K. D. O Brien, H. V. Worthington
Journal of Orthodontics, Vol. 29, 2002, 31 36 SCIENTIFIC SECTION A randomized clinical trial to compare three methods of orthodontic space closure V. Dixon, M. J. F. Read, K. D. O Brien, H. V. Worthington
Case Report Unilateral Molar Distalization: A Nonextraction Therapy
 Case Reports in Dentistry Volume 2012, Article ID 846319, 4 pages doi:10.1155/2012/846319 Case Report Unilateral Molar Distalization: A Nonextraction Therapy M. Bhanu Prasad and S. Sreevalli Department
Case Reports in Dentistry Volume 2012, Article ID 846319, 4 pages doi:10.1155/2012/846319 Case Report Unilateral Molar Distalization: A Nonextraction Therapy M. Bhanu Prasad and S. Sreevalli Department
The effective parameters on the corrosion behavior of dental amalgam
 The effective parameters on the corrosion behavior of dental amalgam Mortazavi V. DDS, MSc 1, Fathi M.H. PhD 2 1 Associate Prof., Dept. of Operative of Dentistry, Dental School, Isfahan University of Medical
The effective parameters on the corrosion behavior of dental amalgam Mortazavi V. DDS, MSc 1, Fathi M.H. PhD 2 1 Associate Prof., Dept. of Operative of Dentistry, Dental School, Isfahan University of Medical
Indian Journal of Basic and Applied Medical Research; June 2014: Vol.-3, Issue- 3, P
 Case Report: Custom made modified distal jet appliance an effective and economic appliance. Dr.Falguni Mehta 1, Dr.Renuka Patel 2,Dr.Harshik Parekh 3,Dr.Manop Agrawal 4 1Head Of Department, Department
Case Report: Custom made modified distal jet appliance an effective and economic appliance. Dr.Falguni Mehta 1, Dr.Renuka Patel 2,Dr.Harshik Parekh 3,Dr.Manop Agrawal 4 1Head Of Department, Department
An estimated 25-30% of all orthodontic patients can benefit from maxillary
 2017 JCO, Inc. May not be distributed without permission. www.jco-online.com A New Appliance for Efficient Molar Distalization VAIBHAV GANDHI, BDS, MDS FALGUNI MEHTA, BDS, MDS HARSHIK PAREKH, BDS, MDS
2017 JCO, Inc. May not be distributed without permission. www.jco-online.com A New Appliance for Efficient Molar Distalization VAIBHAV GANDHI, BDS, MDS FALGUNI MEHTA, BDS, MDS HARSHIK PAREKH, BDS, MDS
Mechanical properties of orthodontic wires covered with a polyether ether ketone tube
 Original Article Mechanical properties of orthodontic wires covered with a polyether ether ketone tube Nobukazu Shirakawa a ; Toshio Iwata b ; Shinjiro Miyake b ; Takero Otuka b ; So Koizumi b ; Toshitugu
Original Article Mechanical properties of orthodontic wires covered with a polyether ether ketone tube Nobukazu Shirakawa a ; Toshio Iwata b ; Shinjiro Miyake b ; Takero Otuka b ; So Koizumi b ; Toshitugu
Aesthetic Brackets and Accessories
 Aesthetic Brackets and Accessories Our Strength is our Service Ortho-Care s version of the MBT prescription shown in this chapter is not claimed to be a duplication, and does not in any way imply endorsement
Aesthetic Brackets and Accessories Our Strength is our Service Ortho-Care s version of the MBT prescription shown in this chapter is not claimed to be a duplication, and does not in any way imply endorsement
Low-Force Mechanics Nonextraction. Estimated treatment time months (Actual 15 mos 1 week). Low-force mechanics.
 T.S. Age: 43 Years 1 Month Diagnosis: Class I Nonextraction Adult (severe crowding, very flat profile with tissue-grafting indications) Background: This case is very similar to the previous case of a 14-year-old.
T.S. Age: 43 Years 1 Month Diagnosis: Class I Nonextraction Adult (severe crowding, very flat profile with tissue-grafting indications) Background: This case is very similar to the previous case of a 14-year-old.
Orthodontics (Ort) #910 Orthodontic Technique Lab Course Director: Dr. Brian Preston Instructor: Brian D. Willison CDT
 Orthodontics (Ort) #910 Orthodontic Technique Lab Course Director: Dr. Brian Preston Instructor: Brian D. Willison CDT Purpose: The Orthodontic Technique Lab Course is the introductory level for the first
Orthodontics (Ort) #910 Orthodontic Technique Lab Course Director: Dr. Brian Preston Instructor: Brian D. Willison CDT Purpose: The Orthodontic Technique Lab Course is the introductory level for the first
Third-Order Torque and Self-Ligating Orthodontic Bracket Type Effects on Sliding Friction
 Original Article Third-Order Torque and Self-Ligating Orthodontic Bracket Type Effects on Sliding Friction Michael Chung a ; Robert J. Nikolai b ; Ki Beom Kim c ; Donald R. Oliver c ABSTRACT Objective:
Original Article Third-Order Torque and Self-Ligating Orthodontic Bracket Type Effects on Sliding Friction Michael Chung a ; Robert J. Nikolai b ; Ki Beom Kim c ; Donald R. Oliver c ABSTRACT Objective:
Treatment of a malocclusion characterized
 CONTINUING EDUCATION ARTICLE Cephalometric evaluation of open bite treatment with NiTi arch wires and anterior elastics Nazan Küçükkeleș, DDS, PhD, a Ahu Acar, DDS, PhD, b Arzu A. Demirkaya, DDS, c Berna
CONTINUING EDUCATION ARTICLE Cephalometric evaluation of open bite treatment with NiTi arch wires and anterior elastics Nazan Küçükkeleș, DDS, PhD, a Ahu Acar, DDS, PhD, b Arzu A. Demirkaya, DDS, c Berna
IJPCDR ORIGINAL RESEARCH ABSTRACT INTRODUCTION
 ORIGINAL RESEARCH Comparison of the Conventional Method using Intraoral Periapical with the Contemporary Imaging Technology (Spiral Computed Tomography) for the Amount of Apical Root Resorption K. V. Sujan
ORIGINAL RESEARCH Comparison of the Conventional Method using Intraoral Periapical with the Contemporary Imaging Technology (Spiral Computed Tomography) for the Amount of Apical Root Resorption K. V. Sujan
Advantages. Fantastic. systems. Together with Unitek Lateral Development Archwires, this system puts you in control to
 3M Self-Ligating Appliances Lateral Development System Treatment Advantages Fantastic Finishes For orthodontic professionals who choose lateral development as a desired treatment outcome, 3M now offers
3M Self-Ligating Appliances Lateral Development System Treatment Advantages Fantastic Finishes For orthodontic professionals who choose lateral development as a desired treatment outcome, 3M now offers
Wires and clasps. ONLINE SHOP shop.dentaurum.com. Large selection excellent quality. DENTAURUM
 Wires and clasps. Large selection excellent quality. DENTAURUM ONLINE SHOP shop.dentaurum.com 285 A wide range of products. The Dentaurum product range supports your daily lab work and makes life easier.
Wires and clasps. Large selection excellent quality. DENTAURUM ONLINE SHOP shop.dentaurum.com 285 A wide range of products. The Dentaurum product range supports your daily lab work and makes life easier.
Study Of Release Of Nickel And Chromium Ions From Different Brands Of Stainless Steel Brackets -An Invitro Study.
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-83, p-issn: 2279-861.Volume 13, Issue 7 Ver. I (July. 21), PP 63-67 Study Of Release Of ckel And Chromium Ions From Different Brands
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-83, p-issn: 2279-861.Volume 13, Issue 7 Ver. I (July. 21), PP 63-67 Study Of Release Of ckel And Chromium Ions From Different Brands
Use of Cantilever Mechanics for Impacted Teeth: Case Series
 Send Orders of Reprints at reprints@benthamscience.net 186 The Open Dentistry Journal, 2013, 7, 186-197 Use of Cantilever Mechanics for Impacted Teeth: Case Series Open Access Sergio Paduano 1,*, Gianrico
Send Orders of Reprints at reprints@benthamscience.net 186 The Open Dentistry Journal, 2013, 7, 186-197 Use of Cantilever Mechanics for Impacted Teeth: Case Series Open Access Sergio Paduano 1,*, Gianrico
Correction of a maxillary canine-first premolar transposition using mini-implant anchorage
 CASE REPORT Correction of a maxillary canine-first premolar transposition using mini-implant anchorage Mehmet Oguz Oztoprak, DDS, MSc, a Cigdem Demircan, DDS, b Tulin Arun, PhD, DDS, MSc c Transposition
CASE REPORT Correction of a maxillary canine-first premolar transposition using mini-implant anchorage Mehmet Oguz Oztoprak, DDS, MSc, a Cigdem Demircan, DDS, b Tulin Arun, PhD, DDS, MSc c Transposition
Immediate versus Delayed Force Application after Orthodontic Bonding; An In Vitro Study
 Journal of Dentistry, Tehran University of Medical Sciences Original Article Immediate versus Delayed Force Application after Orthodontic Bonding; An In Vitro Study M. Basafa 1, F. Farzanegan 2 1 Professor,
Journal of Dentistry, Tehran University of Medical Sciences Original Article Immediate versus Delayed Force Application after Orthodontic Bonding; An In Vitro Study M. Basafa 1, F. Farzanegan 2 1 Professor,
Unilateral Horizontally Impacted Maxillary Canine and First Premolar Treated with a Double Archwire Technique
 Case Report Unilateral Horizontally Impacted Maxillary Canine and First Premolar Treated with a Double Archwire Technique Chien-Lun Peng a ; Yu-Yu Su b ; Sheng-Yang Lee c Abstract: A patient with a unilateral
Case Report Unilateral Horizontally Impacted Maxillary Canine and First Premolar Treated with a Double Archwire Technique Chien-Lun Peng a ; Yu-Yu Su b ; Sheng-Yang Lee c Abstract: A patient with a unilateral
Simply Great Wire! Great prices.
 2017 CATALOG Simply Great Wire! Great prices. CONTENTS 2-4 NiTi Wires 5 Stainless Steel Wires 6-7 Archforms 8 Beta Titanium Molybdenum Wires 9 Wire Auxiliaries WWW.HIGHLANDMETALS.COM NiTi Wires SuperElastic
2017 CATALOG Simply Great Wire! Great prices. CONTENTS 2-4 NiTi Wires 5 Stainless Steel Wires 6-7 Archforms 8 Beta Titanium Molybdenum Wires 9 Wire Auxiliaries WWW.HIGHLANDMETALS.COM NiTi Wires SuperElastic
Identify the role of the skeletal system particularly in relation to maintaining an upright stance and protecting vital organs.
 9.3.3 The wide range of movements, continual absorption of shocks and diseases make the skeletal system vulnerable to damage but new technologies are allowing the replacement of some damaged structures.
9.3.3 The wide range of movements, continual absorption of shocks and diseases make the skeletal system vulnerable to damage but new technologies are allowing the replacement of some damaged structures.
Premium Heat Activated Arch Wire
 Premium Heat Activated Arch Wire TM Premium Heat Activated Arch Wire American Orthodontics is dedicated to continuous improvement, and delivers on this commitment by manufacturing our Tanzo Premium Heat
Premium Heat Activated Arch Wire TM Premium Heat Activated Arch Wire American Orthodontics is dedicated to continuous improvement, and delivers on this commitment by manufacturing our Tanzo Premium Heat
REDUCING TEST BIAS IN EVALUATING REACH COMPLIANT PAINT SYSTEMS SAFRAN TECH (SAFRAN RESEARCH CENTER)
 REDUCING TEST BIAS IN EVALUATING REACH COMPLIANT PAINT SYSTEMS NIHAD BEN SALAH, R&T MANAGER ADVANCED PROCESSES BAPTISTE FEDI, R&T ENGINEER ELECTROCHEMICAL PROCESSES VIRGINIE MATHIVET, STUDENT IN ENGINEERING
REDUCING TEST BIAS IN EVALUATING REACH COMPLIANT PAINT SYSTEMS NIHAD BEN SALAH, R&T MANAGER ADVANCED PROCESSES BAPTISTE FEDI, R&T ENGINEER ELECTROCHEMICAL PROCESSES VIRGINIE MATHIVET, STUDENT IN ENGINEERING
THE USE OF VACCUM FORM RETAINERS FOR RELAPSE CORRECTION
 THE USE OF VACCUM FORM RETAINERS FOR RELAPSE CORRECTION Azrul Hafiz Abdul Aziz 1 and Haslinda Ramli 2 1,2 Islamic Science University of Malaysia, Faculty of Dentistry, Level 15, Tower B, Persiaran MPAJ,
THE USE OF VACCUM FORM RETAINERS FOR RELAPSE CORRECTION Azrul Hafiz Abdul Aziz 1 and Haslinda Ramli 2 1,2 Islamic Science University of Malaysia, Faculty of Dentistry, Level 15, Tower B, Persiaran MPAJ,
Dual Force Cuspid Retractor
 CLINICAL INNOVATION 1 Matrishva B Vyas, 2 Neeraj Alladwar ABSTRACT The most time consuming stage of bicuspid extraction-based treatment is cuspid retraction. Cuspid retraction with both types of conventional
CLINICAL INNOVATION 1 Matrishva B Vyas, 2 Neeraj Alladwar ABSTRACT The most time consuming stage of bicuspid extraction-based treatment is cuspid retraction. Cuspid retraction with both types of conventional
Quality. Elegance. Innovation.
 Quality. Elegance. Innovation. Triumph Orthodontic Instruments Quality and engineering come together for superior performance The Triumph Orthodontic Instrument line provides the orthodontic professional
Quality. Elegance. Innovation. Triumph Orthodontic Instruments Quality and engineering come together for superior performance The Triumph Orthodontic Instrument line provides the orthodontic professional
Orthodontic treatment for jaw defor. Sakamoto, T; Sakamoto, S; Harazaki, Author(s) Yamaguchi, H. Journal Bulletin of Tokyo Dental College, 4
 Orthodontic treatment for jaw defor Titlelip and palate patients with the co external-expansion arch and a facia Sakamoto, T; Sakamoto, S; Harazaki, uthor(s) Yamaguchi, H Journal ulletin of Tokyo Dental
Orthodontic treatment for jaw defor Titlelip and palate patients with the co external-expansion arch and a facia Sakamoto, T; Sakamoto, S; Harazaki, uthor(s) Yamaguchi, H Journal ulletin of Tokyo Dental
Angiogenesis of Metal Ions by Components of Dental Gold-silver-palladium Alloys used by Dental Prosthetics in Vitro
 J Oral Tissue Engin 2013;11(1):79-84 ORIGINAL ARTICLE Angiogenesis of Metal Ions by Components of Dental Gold-silver-palladium Alloys used by Dental Prosthetics in Vitro Koichi IMAI 1, Kazuhiko SUESE 2,
J Oral Tissue Engin 2013;11(1):79-84 ORIGINAL ARTICLE Angiogenesis of Metal Ions by Components of Dental Gold-silver-palladium Alloys used by Dental Prosthetics in Vitro Koichi IMAI 1, Kazuhiko SUESE 2,
Effect of Recycling and Autoclave Sterilization on the Unloading Forces of NiTi Closed-Coil Springs: An In Vitro Study
 Momeni Danaei Sh., et al. J Dent Shiraz Univ Med Sci., Dec. 0; (): 8-90. Original Article Effect of Recycling and Autoclave Sterilization on the Unloading Forces of NiTi Closed-Coil Springs: An In Vitro
Momeni Danaei Sh., et al. J Dent Shiraz Univ Med Sci., Dec. 0; (): 8-90. Original Article Effect of Recycling and Autoclave Sterilization on the Unloading Forces of NiTi Closed-Coil Springs: An In Vitro
Enhancing stability and increasing range of motion. Metasul LDH Large Diameter Head
 Enhancing stability and increasing range of motion Metasul LDH Large Diameter Head The Metasul large diameter head technology is the result of in-depth research, development and clinical experience that
Enhancing stability and increasing range of motion Metasul LDH Large Diameter Head The Metasul large diameter head technology is the result of in-depth research, development and clinical experience that
MATERIAL CHARACTERISTICS OF THE ORTHODONTIC ARCHWIRES
 DAAAM INTERNATIONAL SCIENTIFIC BOOK 20 pp. 30-308 CHAPTER 2 MATERIAL CHARACTERISTICS OF THE ORTHODONTIC ARCHWIRES SZUHANEK, C. Abstract Our aim was to investigate the mechanical properties of coated superelastic
DAAAM INTERNATIONAL SCIENTIFIC BOOK 20 pp. 30-308 CHAPTER 2 MATERIAL CHARACTERISTICS OF THE ORTHODONTIC ARCHWIRES SZUHANEK, C. Abstract Our aim was to investigate the mechanical properties of coated superelastic
Is nickel titanium superior to multistranded stainless steel wire in aligning crowded lower anteriors? A comparative in-vivo study.
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861. Volume 9, Issue 3 (Jul.- Aug. 2013), PP 47-51 Is nickel titanium superior to multistranded stainless steel
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861. Volume 9, Issue 3 (Jul.- Aug. 2013), PP 47-51 Is nickel titanium superior to multistranded stainless steel
