Clinical Trial Designs for Topical Antifungal Treatments of Onychomycosis and Implications on Clinical Practice
|
|
|
- Griselda Morris
- 5 years ago
- Views:
Transcription
1 Clinical Trial Designs for Topical Antifungal Treatments of Onychomycosis and Implications on Clinical Practice Phoebe Rich, MD; Tracey C. Vlahovic, DPM; Warren S. Joseph, DPM; Lee T. Zane, MD; Steve B. Hall, PharmD; Nicole Gellings Lowe, PhD; Chris G. Adigun, MD PRACTICE POINTS Despite similar overall designs, notable differences in the study designs of phase 3 clinical trials investigating tavaborole, efinaconazole, and ciclopirox for the treatment of onychomycosis are likely to have had an effect on the reported results, making the efficacy of these agents difficult to compare. The primary difference between studies for tavaborole, efinaconazole, and ciclopirox include the age range of participants, the range of mycotic nail involvement, the presence/absence of tinea pedis, and the nail trimming/debridement protocols used. Without head-to-head investigations, there is room for prescribing clinicians to interpret study results for these agents differently. There currently are 3 topical agents approved by the US Food and Drug Administration (FDA) to treat onychomycosis: tavaborole, efinaconazole, and ciclopirox. The phase 3 clinical trial designs for these treatments and their notable differences make it difficult for clinicians to interpret the data into clinical practice. For example, the primary end point predominantly used to assess efficacy in all the trials is complete cure, defined as no involvement of the nail plus mycologic cure; also, a notable number of patients fail to achieve a complete cure despite clear improvement in the nail. Despite close similarities in the end points and overall design of the clinical trials used for these agents, differences in design are notable, including the age range of participants, the range of mycotic nail involvement, the presence/ absence of tinea pedis, and the nail trimming/debridement protocols used. The differences in clinical trial designs for the 3 FDA-approved topical agents and the lack of head-to-head studies makes efficacy interpretation and comparison inappropriate. This article reviews the phase 3 clinical trials that led to FDA approval of these agents, focusing on their similarities and differences. Cutis. 2017;100: RELATED CONTENT ONLINE Cosmetic Corner: Dermatologists Weigh in on Athlete s Foot Products >> Efinaconazole Solution 10% for Treatment of Toenail Onychomycosis in Latino Patients >> Over-the-counter and Natural Remedies for Onychomycosis: Do They Really Work? >> Dr. Rich is from Oregon Dermatology and Research, Portland. Dr. Vlahovic is from Temple University School of Podiatric Medicine, Philadelphia, Pennsylvania. Dr. Joseph is from Roxborough Memorial Hospital, Philadelphia. Dr. Zane was from Anacor Pharmaceuticals, Inc, Palo Alto, California. Drs. Hall and Gellings Lowe are from Medical Affairs, Sandoz, a Novartis division, Princeton, New Jersey. Dr. Adigun is from Pinehurst Skin Center, North Carolina. Dr. Rich has received research grants as a principal investigator from Anacor Pharmaceuticals, Inc; Moberg Pharma North America LLC; Sandoz, a Novartis division; Valeant Pharmaceuticals International, Inc; and Viamet Pharmaceuticals, Inc. Dr. Vlahovic is a consultant and speaker for PharmaDerm. Dr. Joseph is a speaker for PharmaDerm and Valeant Pharmaceuticals International, Inc. Dr. Zane was an employee and shareholder for Anacor Pharmaceuticals, Inc. Drs. Hall and Gellings Lowe are employees of Sandoz, a Novartis division. Dr. Adigun is an advisory board member for Sandoz, a Novartis division. Correspondence: Steve B. Hall, PharmD, 100 College Rd West, Princeton, NJ (steve.hall@sandoz.com). VOL. 100 NO. 4 I OCTOBER
2 Onychomycosis is a fungal nail infection primarily caused by dermatophytes. 1 If left untreated, the infection can cause nail destruction and deformities, 1 resulting in pain and discomfort, 2 impaired foot mobility, 3 and an overall reduced quality of life. 1 Onychomycosis is a chronic condition that requires long treatment periods due to the slow growth rates of toenails. 1 To successfully cure the condition, fungal eradication must be achieved. Prior to the US Food and Drug Administration (FDA) approval of tavaborole and efinaconazole, ciclopirox was the only approved topical treatment for onychomycosis. 4 The recent approval of tavaborole and efinaconazole has increased treatment options available to patients and has started to pave the way for future topical treatments. This article discusses the 3 approved topical treatments for onychomycosis and focuses on the design of the phase 3 clinical trials that led to their approval. Topical Agents Used to Treat Onychomycosis Tavaborole, efinaconazole, and ciclopirox have undergone extensive clinical investigation to receive FDA approval. Results from pivotal phase 3 studies establishing the efficacy and safety of each agent formed the basis for regulatory submission. Although it may seem intuitive to compare the relative performance of these agents based on their respective phase 3 clinical trial data, there are important differences in study methodology, conduct, and populations that prevent direct comparisons. The FDA provides limited guidance to the pharmaceutical industry on how to conduct clinical trials for potential onychomycosis treatments. Comparative efficacy and safety claims are limited based on cross-study comparisons. The details of the phase 3 trial designs are summarized in the Table. Tavaborole Tavaborole is a boron-based treatment with a novel mechanism of action. 5 Tavaborole binds to the editing domain of leucyl transfer ribonucleic acid synthetase via an integrated boron atom and inhibits fungal protein synthesis. 6 Two identical randomized, double-blind, vehicle-controlled, parallel-group, phase 3 clinical trials evaluating tavaborole were performed. 5 The first study (registered at with the identifier NCT ) included 594 participants from 27 sites in the United States and Mexico and was conducted between December 2010 and November The second study (NCT ) included 604 participants from 32 sites in the United States and Canada and was conducted between February 2011 and January Eligible participants 18 years and older had distal subungual onychomycosis (DSO) of the toenails affecting 20% to 60% of 1 or more target great toenails (TGTs), tested positive for fungus using potassium hydroxide (KOH) wet mounts and positive for Trichophyton rubrum and Trichophyton mentagrophytes on fungal culture diagnostic tests, had distal TGT thickness of 3 mm or less, and had 3 mm or more of clear nail between the proximal nail fold and the most proximal visible mycotic border. 5 Those with active tinea pedis requiring treatment or with a history of chronic moccasin-type tinea pedis were excluded. Participants were randomized to receive either tavaborole or vehicle (2:1). Treatments were applied once daily to all infected toenails for a total of 48 weeks, and nail debridement (defined as partial or complete removal of the toenail) was not permitted. Notably, controlled trimming of the nail was allowed to 1 mm of the leading nail edge. Regular assessments of each toenail for disease involvement, onycholysis, and subungual hyperkeratosis were made at screening, baseline, week 2, week 6, and every 6 weeks thereafter until week 52. Subungual TGT samples were taken at screening and every 12 weeks during the study for examination at a mycology laboratory, which performed KOH and fungal culture tests. A follow-up assessment was made at week The primary end point was complete cure of the TGT at week 52, with secondary end points of completely A B FIGURE 1. Illustrative case of toenail onychomycosis before (A) and at week 52 (B) in tavaborole phase 3 clinical trials. Almost clear nail with negative mycology was achieved at week 52 after 48 weeks of treatment with tavaborole. 260 I CUTIS
3 THERAPEUTICS FOR THE CLINICIAN co py and was conducted at a total of 74 sites in Japan (33 sites), Canada (7 sites), and the United States (34 sites) between December 2009 and September The second study (NCT ) had 785 participants and was conducted at 44 sites in Canada (8 sites) and the United States (36 sites) between December 2009 and October Participants aged 18 to 70 years with a clinical diagnosis of DSO affecting 1 or more TGT were eligible to participate.7 Other eligibility criteria included an uninfected toenail length 3 mm or more from the proximal nail fold, a maximum toenail thickness of 3 mm, positive KOH wet mounts, and positive dermatophyte or mixed dermatophyte/candida cultures. Dermatophytes included T rubrum and T mentagrophytes. Those with severe moccasin-type tinea pedis were excluded. Participants were randomized to receive efinaconazole or vehicle (3:1). Once-daily treatments were self-applied to nails for T IS D o no t or almost clear TGT nail ( 10% dystrophic nail), completely or almost clear TGT nail ( 10% dystrophic nail) plus negative mycology, and negative mycology of TGT.5 Examples of TGTs in participants who achieved complete cure and almost clear nails with negative mycology before and after treatment with tavaborole are shown in Figure 1. An example of a patient considered to have treatment failure is shown in Figure 2. This patient showed marked improvement in nail appearance and had a negative culture result but had a positive KOH test, which demonstrates the stringency in which topical agents are judged in onychomycosis trials.5 Efinaconazole Efinaconazole is a topical triazole antifungal specifically indicated to treat onychomycosis. Two identical randomized, vehicle-controlled, doubleblind, multicenter trials were performed to assess the safety and efficacy of efinaconazole solution 10%.7 The first study (NCT ) involved 870 participants A B C U FIGURE 2. Clinical example of a treatment failure from the tavaborole phase 3 clinical trials. A patient before treatment (A) and at week 52 (B) who achieved an almost completely clear nail plus negative culture but positive potassium hydroxide preparation results after 48 weeks of treatment with tavaborole. A B FIGURE 3. Representative clinical example of an onychomycosis patient before (A) and after treatment with efinaconazole for 48 weeks (B) with a trimmed nail, achieving complete cure. Reprinted from the Journal of the American Academy of Dermatology, Copyright 2013, with permission from Elsevier.7 VOL. 100 NO. 4 I OCTOBER
4 Summary of Phase 3 Clinical Trials for US Food and Drug Administration Approved Topical Therapies for Onychomycosis Clinicaltrials.gov Identifier Study Population Mean Age (Range), y Participant Race Randomization Mycotic Area Involvement Presence of Tinea Pedis Nail Trimming Debridement Treatment Application Tavaborole solution 5% 5 NCT ; NCT ; 604 Efinaconazole solution 10% 7 NCT ; NCT ; (18 88); 55 (20 81) 52 (18 71); 51 (18 71) Ciclopirox nail lacquer topical solution 8% 8 N/A 223; (18 70); 50 (19 70) Study 1 (tavaborole vs vehicle): white, 79.2% vs 78.4%; black, 4.8% vs 6.2%; Asian, 0.5% vs 0%; other, 15.5% vs 15.5%; study 2 (tavaborole vs vehicle): white, 89.6% vs 89.3%; black, 5.3% vs 6.8%; Asian, 2.8% vs 1.0%; other, 2.3% vs 2.9% Study 1 (efinaconazole vs vehicle): white, 64.8% vs 65.4%; black, 5.5% vs 3.3%; Asian, 28.8% vs 29.4%; other, 0.9% vs 1.9%; study 2 (efinaconazole vs vehicle): white, 90.9% vs 81.6%; black, 5.9% vs 10.4%; Asian, 1.9% vs 3.0%; other, 2.2% vs 5.0% Study 1 (ciclopirox vs vehicle): white, 94.6% vs 91.9%; black, 0.9% vs 1.8%; Hispanic, 3.6% vs 6.3%; other, 0.9% vs 0%; study 2 (ciclopirox vs vehicle): white, 86.6% vs 88.1%; black, 3.4% vs 5.1%; Asian, 3.4% vs 0%; Hispanic, 5.0% vs 5.1%; other, 1.7% vs 1.7% 2:1 20% 60% Active tinea pedis requiring treatment excluded, history of chronic moccasin-type tinea pedis excluded 3:1 20% 50% Severe moccasintype tinea pedis excluded 1:1 20% 65% Tinea pedis not excluded Trimming limited by investigator to no closer than 1 mm from the hyponychium Trimming not specifically controlled by the protocol Trimming allowed Not permitted Treatment applied once daily on, around, and under the infected TGT and other infected non-tgts in a thin even layer using a glass dropper Not permitted Treatment applied once daily via brush applicator to the clean dry nail plate surface, the lateral and proximal nail folds, the hyponychium, and the undersurface of the nail plate Performed every month Treatment applied once daily to the entire nail plate including 5 mm of the proximal nail fold, hyponychium, and ventral surface using a brush applicator; repeated daily for 7 d before removing lacquer Abbreviations: TGT, target great toenail; N/A, not available. a Participants who were clinically cured only. Treatment Period/Final Assessment 48 wk/52 wk 48 wk/52 wk 48 wk/ wk after treatment a 262 I CUTIS
5 48 weeks. Clinical assessments were made at baseline and every 12 weeks until week 48, with a follow-up assessment at week 52. No nail trimming protocol was provided. 7 The primary end point of the efinaconazole phase 3 trials was complete cure at week 52, with secondary end points including mycologic cure, treatment success ( 5% mycotic nail), and complete or almost complete cure (negative culture and KOH, 5% mycotic nail). An example of a complete cure from baseline to week 52 is shown in Figure 3. 7 Ciclopirox Ciclopirox was the first topical therapy to be approved for the treatment of onychomycosis. Ciclopirox is a broad-spectrum antifungal agent that inhibits metal-dependent enzymes, which are responsible for the degradation of toxic peroxides in fungal cells. The safety and efficacy of ciclopirox nail lacquer topical solution 8% also was investigated in 2 identical phase 3 clinical trials. 8 The first study was conducted at 9 sites in the United States between June 1994 and June 1996 and included 223 participants. The second study was conducted at 9 sites in the United States between July 1994 and April 1996 and included 237 participants. Eligible participants were required to have DSO in at least one TGT, positive KOH wet mount with positive dermatophyte culture, and 20% to 65% nail involvement. 8 Those with tinea pedis were not excluded. Participants were randomized to receive once-daily treatment with ciclopirox or vehicle (1:1)(applied to all toenails and affected fingernails) for 48 weeks. The product was to be removed by the patient with alcohol on a weekly basis. Trimming was allowed as necessary, and mechanical debridement by the physician could be performed monthly. Assessments were made every 4 weeks, and mycologic examinations were performed every 12 weeks. Participants who were clinically cured were assessed further in a 12- to 24-week posttreatment follow-up period. 8 The primary end point of complete cure and secondary end points of treatment success (negative culture and KOH, 10% mycotic nail), mycologic cure, and negative mycologic culture were assessed at week Phase 3 Clinical Trial Similarities and Differences The phase 3 clinical trials used to investigate the safety and efficacy of tavaborole, 5 efinaconazole, 7 and ciclopirox 8 were similar in their overall design. All trials were randomized, double-blind, vehicle-controlled studies in patients with DSO. Each agent was assessed using a once-daily application for a treatment period of 48 weeks. Primary differences among study designs included the age range of participants, the range of mycotic nail involvement, the presence/absence of tinea pedis, and the nail trimming/debridement protocols used. Differences were observed in the patient eligibility criteria of these trials. Both mycotic area and participant age range were inconsistent for each agent (etable). Participants with larger mycotic areas usually have a poorer prognosis, as they tend to have a greater fungal load. 9 A baseline mycotic area of 20% to 60%, 5 20% to 50%, 7 and 20% to 65% 8 at baseline was required for the tavaborole, efinaconazole, and ciclopirox trials, respectively. Variations in mycotic area between trials can affect treatment efficacy, as clinical cures can be reached quicker by patients with smaller areas of infection. Of note, the average mycotic area of involvement was not reported in the tavaborole studies but was 36% and 40% for the efinaconazole and ciclopirox studies, respectively. 5,8 It also is more difficult to achieve complete cure in older patients, as they have poor circulation and reduced nail growth rates. 1,10 The participant age range was 18 to 88 years in the tavaborole trials, with 8% of the participants older than 70 years, 5 compared to 18 to 71 years in both the efinaconazole and ciclopirox trials. 7,8 The average age of participants in each study was approximately 54, 51, and 50 years for tavaborole, efinaconazole, and ciclopirox, respectively. Because factors impacting treatment failure can increase with age, efficacy results can be confounded by differing age distributions across different studies. Another important feature that differed between the clinical trials was the approach to nail trimming defined as shortening of the free edge of the nail distal to the hyponychium which varies from debridement in that the nail plate is removed or reduced in thickness proximal to the hyponychium. In the tavaborole trials, trimming was controlled to within 1 mm of the free edge of the nail, 5 whereas the protocol used for the ciclopirox trials allowed nail trimming as necessary as well as moderate debridement before treatment application and on a monthly basis. 8 Debridement is an important component in all ciclopirox trials, as it is used to reduce fungal load. 11 No trimming control was provided during the efinaconazole trials; however, debridement was prohibited. 7 These differences can dramatically affect the study results, as residual fungal elements and portions of infected nails are removed during the trimming process in an uncontrolled manner, which can affect mycologic testing results as well as the clinical efficacy results determined through investigator evaluation. Discrepancies regarding nail trimming approach inevitably makes the trial results difficult to compare, as mycologic cure is not translatable between studies. Furthermore, somewhat unusually, complete cure rate variations were observed between different study centers in the efinaconazole trials. Japanese centers in the first efinaconazole study (NCT ) had higher complete cure rates in both the efinaconazole and vehicle treatment arms, which is notable because approximately 29% of participants in this study were Asian, mostly hailing from 33 Japanese centers. The reason for these confounding results is unknown and requires further analysis. Lastly, the presence or absence of tinea pedis can affect the response to onychomycosis treatment. In VOL. 100 NO. 4 I OCTOBER
6 the tavaborole trials, patients with active interdigital tinea pedis or exclusively plantar tinea pedis or chronic moccasin-type tinea pedis requiring treatment were excluded from the studies. 5 In contrast, only patients with severe moccasin-type tinea pedis were excluded in efinaconazole trials. 7 The ciclopirox studies had no exclusions based on presence of tinea pedis. 8 These differences are noteworthy, as tinea pedis can serve as a reservoir for fungal infection if not treated and can lead to recurrence of onychomycosis. 12 Conclusion In recent years, disappointing efficacy has resulted in the failure of several topical agents for onychomycosis during their development; however, there are several aspects to consider when examining efficacy data in onychomycosis studies. Obtaining a complete cure in onychomycosis is difficult. Because patients applying treatments at home are unlikely to undergo mycologic testing to confirm complete cure, visual inspections are helpful to determine treatment efficacy. Despite similar overall designs, notable differences in the study designs of the phase 3 clinical trials investigating tavaborole, efinaconazole, and ciclopirox are likely to have had an effect on the reported results, making the efficacy of the agents difficult to compare. It is particularly tempting to compare the primary end point results of each trial, especially considering tavaborole and efinaconazole had primary end points with the same parameters; however, there are several other factors (eg, age range of study population, extent of infection, nail trimming, patient demographics) that may have affected the outcomes of the studies and precluded a direct comparison of any end points. Without head-to-head investigations, there is room for prescribing clinicians to interpret results differently. Acknowledgment Writing and editorial assistance was provided by ApotheCom Associates, LLC, Yardley, Pennsylvania, and was supported by Sandoz, a Novartis division. REFERENCES 1. Elewski BE. Onychomycosis: pathogenesis, diagnosis, and management. Clin Microbiol Rev. 1998;11: Thomas J, Jacobson GA, Narkowicz CK, et al. Toenail onychomycosis: an important global disease burden. J Clin Pharm Ther. 2010;35: Scher RK. Onychomycosis: a significant medical disorder. J Am Acad Dermatol. 1996;35(3, pt 2):S2-S5. 4. Del Rosso JQ. The role of topical antifungal therapy for onychomycosis and the emergence of newer agents. J Clin Aesthet Dermatol. 2014;7: Elewski BE, Aly R, Baldwin SL, et al. Efficacy and safety of tavaborole topical solution, 5%, a novel boron-based antifungal agent, for the treatment of toenail onychomycosis: results from 2 randomized phase-iii studies. J Am Acad Dermatol. 2015;73: Rock FL, Mao W, Yaremchuk A, et al. An antifungal agent inhibits an aminoacyl-trna synthetase by trapping trna in the editing site. Science. 2007;316: Elewski BE, Rich P, Pollak R, et al. Efinaconazole 10% solution in the treatment of toenail onychomycosis: two phase III multicenter, randomized, double-blind studies. J Am Acad Dermatol. 2013;68: Gupta AK, Joseph WS. Ciclopirox 8% nail lacquer in the treatment of onychomycosis of the toenails in the United States. J Am Pod Med Assoc. 2000;90: Carney C, Tosti A, Daniel R, et al. A new classification system for grading the severity of onychomycosis: Onychomycosis Severity Index. Arch Dermatol. 2011;147: Gupta AK. Onychomycosis in the elderly. Drugs Aging. 2000;16: Gupta AK, Malkin KF. Ciclopirox nail lacquer and podiatric practice. J Am Podiatr Med Assoc. 2000;90: Scher RK, Baran R. Onychomycosis in clinical practice: factors contributing to recurrence. Br J Dermatol. 2003;149(suppl 65): I CUTIS
DISTAL LATERAL SUBUNGUAL ONYCHOMYCOSIS
 Boni E. Elewski, MD James Elder Professor of Dermatology University of Alabama RESEARCH GRANTS -TO UNIVERSITY Dusa, Meiji, Valeant, Viamet Amgen, Abbvie, Boehringer Ingelheim, Celgene, Lilly, Merck, Novartis,
Boni E. Elewski, MD James Elder Professor of Dermatology University of Alabama RESEARCH GRANTS -TO UNIVERSITY Dusa, Meiji, Valeant, Viamet Amgen, Abbvie, Boehringer Ingelheim, Celgene, Lilly, Merck, Novartis,
Fungal Resistance, Biofilm, and Its Impact In the Management of Nail Infection
 Fungal Resistance, Biofilm, and Its Impact In the Management of Nail Infection Faculty Raza Aly, PhD, MPH Professor Emeritus University of California Medical Center (MSSF) Professor, Dermatology Faculty
Fungal Resistance, Biofilm, and Its Impact In the Management of Nail Infection Faculty Raza Aly, PhD, MPH Professor Emeritus University of California Medical Center (MSSF) Professor, Dermatology Faculty
 Page 1 of 5 Management strategies for onychomycosis in special patient populations Phoebe Rich, Nathaniel J. Jellinek and David M. Pariser Dermatology News. 46.8 (Aug. 2015): ps13. Copyright: COPYRIGHT
Page 1 of 5 Management strategies for onychomycosis in special patient populations Phoebe Rich, Nathaniel J. Jellinek and David M. Pariser Dermatology News. 46.8 (Aug. 2015): ps13. Copyright: COPYRIGHT
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other EFINACONAZOLE JUBLIA 41184 TAVABOROLE KERYDIN 41353 GUIDELINES FOR USE 1. Does the patient have a diagnosis of onychomycosis of the fingernail or toenail? If yes,
Generic Brand HICL GCN Exception/Other EFINACONAZOLE JUBLIA 41184 TAVABOROLE KERYDIN 41353 GUIDELINES FOR USE 1. Does the patient have a diagnosis of onychomycosis of the fingernail or toenail? If yes,
Classification. Distal & Lateral Subungual OM. White Superficial OM. Proximal Subungual OM. Candidal OM. Total dystrophic OM
 Onychomycosis Commonest dermatological condition Definition: Infection of the nail caused by fungi that include dermatophytes, non-dermatophyte moulds and yeasts (mainly Candida). 80% of all OM affects
Onychomycosis Commonest dermatological condition Definition: Infection of the nail caused by fungi that include dermatophytes, non-dermatophyte moulds and yeasts (mainly Candida). 80% of all OM affects
UPDATE ON ONYCHOMYCOSIS TREATMENTS
 UPDATE ON ONYCHOMYCOSIS TREATMENTS Tracey C. Vlahovic, DPM FFPM RCPS (Glasg) Clinical Professor, Dept. of Podiatric Medicine Temple University School of Podiatric Medicine, Philadelphia, PA CONFLICTS OF
UPDATE ON ONYCHOMYCOSIS TREATMENTS Tracey C. Vlahovic, DPM FFPM RCPS (Glasg) Clinical Professor, Dept. of Podiatric Medicine Temple University School of Podiatric Medicine, Philadelphia, PA CONFLICTS OF
Therapeutics for the Clinician. Efinaconazole Solution 10%: Topical Antifungal Therapy for Toenail Onychomycosis CUTIS
 Solution 10%: Topical Antifungal Therapy for Toenail Onychomycosis Antonella Tosti, MD Practice Points Toenail onychomycosis is a common disease with limited treatment options, as treatment failures and
Solution 10%: Topical Antifungal Therapy for Toenail Onychomycosis Antonella Tosti, MD Practice Points Toenail onychomycosis is a common disease with limited treatment options, as treatment failures and
 Page 1 of 9 Current and emerging options in the treatment of onychomycosis Boni Elewshi, David Pariser, Phoebe Rich and Richard K. Scher Family Practice News. 43.14 (Sept. 1, 2013): ps11. Copyright: COPYRIGHT
Page 1 of 9 Current and emerging options in the treatment of onychomycosis Boni Elewshi, David Pariser, Phoebe Rich and Richard K. Scher Family Practice News. 43.14 (Sept. 1, 2013): ps11. Copyright: COPYRIGHT
Investor Presentation December 2012
 Investor Presentation December 2012 1 Safe Harbor Statement Certain items in this presentation and other matters discussed today or answers that may be given to questions asked could constitute forwardlooking
Investor Presentation December 2012 1 Safe Harbor Statement Certain items in this presentation and other matters discussed today or answers that may be given to questions asked could constitute forwardlooking
J. Hibler, D.O. OhioHealth - O Bleness Memorial Hospital, Athens, Ohio. AOCD Annual Conference Orlando, Florida
 J. Hibler, D.O. OhioHealth - O Bleness Memorial Hospital, Athens, Ohio AOCD Annual Conference Orlando, Florida 10.18.15 A) Onychodystrophy B) Onychogryphosis Question dogma Michael Conroy, MD C) Onychomycosis
J. Hibler, D.O. OhioHealth - O Bleness Memorial Hospital, Athens, Ohio AOCD Annual Conference Orlando, Florida 10.18.15 A) Onychodystrophy B) Onychogryphosis Question dogma Michael Conroy, MD C) Onychomycosis
OTE. Efinaconazole (Jublia ): A New Topical Therapy for Toenail Onychomycosis. Vol. 30, Issue 10 July Established 1985
 HAR Vol. 30, Issue 10 July 2015 M A N OTE Established 1985 Efinaconazole (Jublia ): A New Topical Therapy for Toenail Onychomycosis Austin Hewlett, harmd/mba Candidate O nychomycosis is a chronic fungal
HAR Vol. 30, Issue 10 July 2015 M A N OTE Established 1985 Efinaconazole (Jublia ): A New Topical Therapy for Toenail Onychomycosis Austin Hewlett, harmd/mba Candidate O nychomycosis is a chronic fungal
Clinical Evaluation of Safety and Efficacy of a New Topical Treatment for Onychomycosis
 October 2011 1186 Volume 10 Issue 10 Copyright 2011 ORIGINAL ARTICLES Clinical Evaluation of Safety and Efficacy of a New Topical Treatment for Onychomycosis Adnan Nasir MD,a Beth Goldstein MD,b Martin
October 2011 1186 Volume 10 Issue 10 Copyright 2011 ORIGINAL ARTICLES Clinical Evaluation of Safety and Efficacy of a New Topical Treatment for Onychomycosis Adnan Nasir MD,a Beth Goldstein MD,b Martin
Clinical clearing of moderate and severe onychomycosis with the Nd:YAG 1064nm laser and post treatment prevention with tolnaftate
 Clinical clearing of moderate and severe onychomycosis with the Nd:YAG 1064nm laser and post treatment prevention with tolnaftate by Myron A. Bodman DPM 1*, Marie Mantini Blazer DPM 1, Bryan D. Caldwell
Clinical clearing of moderate and severe onychomycosis with the Nd:YAG 1064nm laser and post treatment prevention with tolnaftate by Myron A. Bodman DPM 1*, Marie Mantini Blazer DPM 1, Bryan D. Caldwell
Therapeutics for the Clinician
 Luliconazole for the Treatment of Interdigital Tinea Pedis: A Double-blind, Vehicle-Controlled Study Michael Jarratt, MD; Terry Jones, MD; Steven Kempers, MD; Phoebe Rich, MD; Katy Morton; Norifumi Nakamura,
Luliconazole for the Treatment of Interdigital Tinea Pedis: A Double-blind, Vehicle-Controlled Study Michael Jarratt, MD; Terry Jones, MD; Steven Kempers, MD; Phoebe Rich, MD; Katy Morton; Norifumi Nakamura,
TRANSPARENCY COMMITTEE OPINION. 27 January 2010
 The legally binding text is the original French version TRAPARENCY COMMITTEE OPINION 27 January 2010 80 mg/g, medicated nail lacquer B/1 glass vial of 3.3 ml (CIP: 395 010-4) B/1 glass vial of 6.6 ml (CIP:
The legally binding text is the original French version TRAPARENCY COMMITTEE OPINION 27 January 2010 80 mg/g, medicated nail lacquer B/1 glass vial of 3.3 ml (CIP: 395 010-4) B/1 glass vial of 6.6 ml (CIP:
Medical Directive. Medical Director: Date Revised: January 23, Executive Director: Date Revised: January 23, 2019
 Medical Directive Assessment and Treatment of Onychomycosis and Tinea Pedis Assigned Number: 026 Activation Date: January 1, 2019 Review due by: December 1, 2020 Approval Signature & Date Medical Director:
Medical Directive Assessment and Treatment of Onychomycosis and Tinea Pedis Assigned Number: 026 Activation Date: January 1, 2019 Review due by: December 1, 2020 Approval Signature & Date Medical Director:
STUDY. A New Classification System for Grading the Severity of Onychomycosis
 STUDY A New Classification System for Grading the Severity of Onychomycosis Onychomycosis Severity Index Caitlin Carney, MD; Antonella Tosti, MD; Ralph Daniel, MD; Richard Scher, MD; Phoebe Rich, MD; Jamie
STUDY A New Classification System for Grading the Severity of Onychomycosis Onychomycosis Severity Index Caitlin Carney, MD; Antonella Tosti, MD; Ralph Daniel, MD; Richard Scher, MD; Phoebe Rich, MD; Jamie
Onychomycosis and tinea pedis in patients with diabetes
 PUBLICATION OF THIS ARTICLE WAS SUPPORTED BY AN EDUCATIONAL GRANT FROM NOVARTIS PHARMACEUTICALS CORPORATION Onychomycosis and tinea pedis in patients with diabetes Phoebe Rich, MD Portland, Oregon ONYCHOMYCOSIS
PUBLICATION OF THIS ARTICLE WAS SUPPORTED BY AN EDUCATIONAL GRANT FROM NOVARTIS PHARMACEUTICALS CORPORATION Onychomycosis and tinea pedis in patients with diabetes Phoebe Rich, MD Portland, Oregon ONYCHOMYCOSIS
Laser Treatment of Onychomycosis. Description
 Subject: Laser Treatment of Onychomycosis Page: 1 of 8 Last Review Status/Date: September 2014 Laser Treatment of Onychomycosis Description Onychomycosis is a common fungal infection of the nail. Currently
Subject: Laser Treatment of Onychomycosis Page: 1 of 8 Last Review Status/Date: September 2014 Laser Treatment of Onychomycosis Description Onychomycosis is a common fungal infection of the nail. Currently
RESULTS. TABLE 1 FOOT, TOE, AND TOENAIL EXAMINATION CONDITION (Number of patients with)
 Over the several decades that I have been in practice I have been struggling to provide my patients with onychomycosis an effective treatment for this pervasive and embarrassing disease. I have had several
Over the several decades that I have been in practice I have been struggling to provide my patients with onychomycosis an effective treatment for this pervasive and embarrassing disease. I have had several
Onychomycosis: Diagnosis, Treatment, and Prevention Strategies
 A CME/CE CERTIFIED SUPPLEMENT TO ORIGINAL RELEASE DATE: MARCH 2016 MOST RECENT REVIEW DATE: MARCH 2016 EXPIRATION DATE: FEBRUARY 28, 2018 ESTIMATED TIME TO COMPLETE ACTIVITY: 2.5 HOURS Onychomycosis: Diagnosis,
A CME/CE CERTIFIED SUPPLEMENT TO ORIGINAL RELEASE DATE: MARCH 2016 MOST RECENT REVIEW DATE: MARCH 2016 EXPIRATION DATE: FEBRUARY 28, 2018 ESTIMATED TIME TO COMPLETE ACTIVITY: 2.5 HOURS Onychomycosis: Diagnosis,
ORIGINAL INVESTIGATION. Boni E. Elewski, MD; James Leyden, MD; Michael G. Rinaldi, PhD; Ercem Atillasoy, MD
 Office Practice Based Confirmation of Onychomycosis A US Nationwide Prospective Survey ORIGINAL INVESTIGATION Boni E. Elewski, MD; James Leyden, MD; Michael G. Rinaldi, PhD; Ercem Atillasoy, MD Background:
Office Practice Based Confirmation of Onychomycosis A US Nationwide Prospective Survey ORIGINAL INVESTIGATION Boni E. Elewski, MD; James Leyden, MD; Michael G. Rinaldi, PhD; Ercem Atillasoy, MD Background:
ONYCHOMYCOSIS LET S HELP OUR PATIENTS ELIMINATE IT
 ONYCHOMYCOSIS LET S HELP OUR PATIENTS ELIMINATE IT WHY AM I STANDING IN FRONT OF YOU TODAY? TO PRESENT MY PROTOCOL ON HOW I HELP MY PATIENTS CURE THEIR ONYCHOMYCOSIS Erin King Registered Practical Nurse/Podortho
ONYCHOMYCOSIS LET S HELP OUR PATIENTS ELIMINATE IT WHY AM I STANDING IN FRONT OF YOU TODAY? TO PRESENT MY PROTOCOL ON HOW I HELP MY PATIENTS CURE THEIR ONYCHOMYCOSIS Erin King Registered Practical Nurse/Podortho
TIME TO CLEAN IT UP. TOENAIL FUNGUS Still your dirty secret? WITH JUBLIA
 TOENAIL FUNGUS Still your dirty secret? TIME TO CLEAN IT UP WITH JUBLIA PRESCRIBED BRAND FOR TOENAIL FUNGUS Visit JubliaRx.com for a $25 Instant Savings Offer* at Walgreens and other participating independent
TOENAIL FUNGUS Still your dirty secret? TIME TO CLEAN IT UP WITH JUBLIA PRESCRIBED BRAND FOR TOENAIL FUNGUS Visit JubliaRx.com for a $25 Instant Savings Offer* at Walgreens and other participating independent
Corporate Medical Policy
 Corporate Medical Policy Laser Treatment of Onychomycosis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: laser_treatment_of_onychomycosis 5/2013 11/2017 11/2018 11/2017 Description
Corporate Medical Policy Laser Treatment of Onychomycosis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: laser_treatment_of_onychomycosis 5/2013 11/2017 11/2018 11/2017 Description
The Nail Common terms & Anatomy Onychomycosis Causes and Background Onychomycosis Classification Treatment Basics
 FOX Podiatry Applications The Nail Common terms & Anatomy Onychomycosis Causes and Background Onychomycosis Classification Treatment Basics The Nail - Areas definitions Proximal nail fold Nail plate area
FOX Podiatry Applications The Nail Common terms & Anatomy Onychomycosis Causes and Background Onychomycosis Classification Treatment Basics The Nail - Areas definitions Proximal nail fold Nail plate area
ONYCHOMYCOSIS LASER THERAPY
 FOX ONYCHOMYCOSIS LASER THERAPY 1064 nm FOX 1064 nm, 10W IDEAL FOR: ONYCHOMYCOSIS FUNGAL NAIL INFECTION LASER INNOVATION MADE IN GERMANY www.arclaser.com 2 FOX Laser offers an effective, pain free method
FOX ONYCHOMYCOSIS LASER THERAPY 1064 nm FOX 1064 nm, 10W IDEAL FOR: ONYCHOMYCOSIS FUNGAL NAIL INFECTION LASER INNOVATION MADE IN GERMANY www.arclaser.com 2 FOX Laser offers an effective, pain free method
Onychomycosis is a relatively common dermatologic presentation
 Practical Advice for the Dermatology dermphysician Assistant perspectives December 2012 Vol. 8, No. 4 Management of Onychomycosis: A Review By Coyle S. Connolly, DO Onychomycosis is a relatively common
Practical Advice for the Dermatology dermphysician Assistant perspectives December 2012 Vol. 8, No. 4 Management of Onychomycosis: A Review By Coyle S. Connolly, DO Onychomycosis is a relatively common
CASE STUDY ADULT CASES OF ONYCHOMYCOSIS. Warren S. Joseph, DPM, FIDSA
 ADULT CASES OF ONYCHOMYCOSIS Warren S. Joseph, DPM, FIDSA BACKGROUND (CASE 1) A 41-year-old woman presented with no complaints of pain, but she was concerned about discoloration of her toenails. She has
ADULT CASES OF ONYCHOMYCOSIS Warren S. Joseph, DPM, FIDSA BACKGROUND (CASE 1) A 41-year-old woman presented with no complaints of pain, but she was concerned about discoloration of her toenails. She has
A class IIa medical device intended for mild-to-moderate fungal nail infection PRODUCT MONOGRAPH
 A class IIa medical device intended for mild-to-moderate fungal nail infection PRODUCT MONOGRAPH AWB-2052628721 Date of Preparation March 2017 Introduction to Bayer Bayer is a Life Science company with
A class IIa medical device intended for mild-to-moderate fungal nail infection PRODUCT MONOGRAPH AWB-2052628721 Date of Preparation March 2017 Introduction to Bayer Bayer is a Life Science company with
Diagnosis and Treatment of Infectious, Inflammatory and Neoplastic Nail Conditions
 OFFICE DERMATOLOGY, PART I 0025-7125/98 $8.00 +.OO NAIL DISORDERS Diagnosis and Treatment of Infectious, Inflammatory and Neoplastic Nail Conditions Phoebe Rich, MD Nail problems (Table 1) are common complaints
OFFICE DERMATOLOGY, PART I 0025-7125/98 $8.00 +.OO NAIL DISORDERS Diagnosis and Treatment of Infectious, Inflammatory and Neoplastic Nail Conditions Phoebe Rich, MD Nail problems (Table 1) are common complaints
Clinical and Microbiological study of Tinea unguium in a tertiary care centre
 ISSN: 2319-7706 Volume 4 Number 4 (2015) pp. 899-905 http://www.ijcmas.com Original Research Article Clinical and Microbiological study of Tinea unguium in a tertiary care centre Arti Agrawal 1 *, Uma
ISSN: 2319-7706 Volume 4 Number 4 (2015) pp. 899-905 http://www.ijcmas.com Original Research Article Clinical and Microbiological study of Tinea unguium in a tertiary care centre Arti Agrawal 1 *, Uma
BJD British Journal of Dermatology. Summary. What s already known about this topic? CLINICAL TRIALS
 CLINICAL TRIALS BJD British Journal of Dermatology Efficacy of topical resin lacquer, amorolfine and oral terbinafine for treating toenail onychomycosis: a prospective, randomized, controlled, investigator-blinded,
CLINICAL TRIALS BJD British Journal of Dermatology Efficacy of topical resin lacquer, amorolfine and oral terbinafine for treating toenail onychomycosis: a prospective, randomized, controlled, investigator-blinded,
Prevalence and Risk Factors of Tinea Unguium and Tinea Pedis in the General Population in Spain
 JOURNAL OF CLINICAL MICROBIOLOGY, Sept. 2000, p. 3226 3230 Vol. 38, No. 9 0095-1137/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Prevalence and Risk Factors of Tinea
JOURNAL OF CLINICAL MICROBIOLOGY, Sept. 2000, p. 3226 3230 Vol. 38, No. 9 0095-1137/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Prevalence and Risk Factors of Tinea
Conflicts of Interest. Accure Medical, LLC: Tri-Founder, Tri-Owner, Member Board of Directors
 Nail Therapeutics Molly Hinshaw, MD Associate Professor of Dermatology Director, Nail Clinic University of Wisconsin School of Medicine and Public Health Conflicts of Interest Accure Medical, LLC: Tri-Founder,
Nail Therapeutics Molly Hinshaw, MD Associate Professor of Dermatology Director, Nail Clinic University of Wisconsin School of Medicine and Public Health Conflicts of Interest Accure Medical, LLC: Tri-Founder,
LUZU (luliconazole) external cream
 LUZU (luliconazole) external cream Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
LUZU (luliconazole) external cream Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
Luliconazole Demonstrates Potent In vitro Activity against the Dermatophytes. Recovered from Patients with Onychomycosis
 AAC Accepts, published online ahead of print on 7 April 2014 Antimicrob. Agents Chemother. doi:10.1128/aac.02706-13 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 3 4 5 6 7
AAC Accepts, published online ahead of print on 7 April 2014 Antimicrob. Agents Chemother. doi:10.1128/aac.02706-13 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 3 4 5 6 7
Rapid Visible Improvements in Onychomycosis: An Open-Label Clinical Investigation of Kerasal Nail TM
 Journal of Cosmetics, Dermatological Sciences and Applications, 2017, 7, 57-66 http://www.scirp.org/journal/jcdsa ISSN Online: 2161-4512 ISSN Print: 2161-4105 Rapid Visible Improvements in Onychomycosis:
Journal of Cosmetics, Dermatological Sciences and Applications, 2017, 7, 57-66 http://www.scirp.org/journal/jcdsa ISSN Online: 2161-4512 ISSN Print: 2161-4105 Rapid Visible Improvements in Onychomycosis:
Laser Treatment of Onychomycosis
 Laser Treatment of Onychomycosis Policy Number: 2.01.89 Last Review: 5/2018 Origination: 11/2015 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Laser Treatment of Onychomycosis Policy Number: 2.01.89 Last Review: 5/2018 Origination: 11/2015 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Diagnosis,Therapy and Prophylaxis of Fungal Diseases
 mycoses Diagnosis,Therapy and Prophylaxis of Fungal Diseases Original article Treatment of distal subungual onychomycosis with a topical preparation of urea, propylene glycol and lactic acid: results of
mycoses Diagnosis,Therapy and Prophylaxis of Fungal Diseases Original article Treatment of distal subungual onychomycosis with a topical preparation of urea, propylene glycol and lactic acid: results of
INTRODUCTION. Rapid clearance of sub-ungual onychomycosis by controlled micro penetration and topical terbinafine solution using the Clearanail device
 Rapid clearance of sub-ungual onychomycosis by controlled micro penetration and topical terbinafine solution using the Clearanail device Dr Ivan Bristow, Faculty of Health Sciences, University of Southampton
Rapid clearance of sub-ungual onychomycosis by controlled micro penetration and topical terbinafine solution using the Clearanail device Dr Ivan Bristow, Faculty of Health Sciences, University of Southampton
Source of effectiveness data The effectiveness evidence was derived from a systematic review of published studies.
 Pharmacoeconomic analysis of ciclopirox nail lacquer solution 8% and the new oral antifungal agents used to treat dermatophyte toe onychomycosis in the United States Gupta A K Record Status This is a critical
Pharmacoeconomic analysis of ciclopirox nail lacquer solution 8% and the new oral antifungal agents used to treat dermatophyte toe onychomycosis in the United States Gupta A K Record Status This is a critical
Catamaran Prior Authorization Department. Phone: Fax: Prescriber Information. Member Information.
 Prescriber Information Last Name First Name DEA / NPI Specialty Phone Member Information Last Name Fax - - - - First Name Member ID Number DOB Medication Information: Drug Name and Strength: Quantity:
Prescriber Information Last Name First Name DEA / NPI Specialty Phone Member Information Last Name Fax - - - - First Name Member ID Number DOB Medication Information: Drug Name and Strength: Quantity:
TIME TO CLEAN IT UP. TOENAIL FUNGUS Still your dirty secret? WITH JUBLIA FIGHTING TOENAIL FUNGUS CAN BE FUN
 TOENAIL FUNGUS Still your dirty secret? TIME TO CLEAN IT UP WITH JUBLIA JubliApp FIGHTING TOENAIL FUNGUS CAN BE FUN Download JubliApp for tools, tips, and support Learn more at JubliApp.com Visit JubliaRx.com
TOENAIL FUNGUS Still your dirty secret? TIME TO CLEAN IT UP WITH JUBLIA JubliApp FIGHTING TOENAIL FUNGUS CAN BE FUN Download JubliApp for tools, tips, and support Learn more at JubliApp.com Visit JubliaRx.com
Activity of TDT 067 (Terbinafine in Transfersome) against Agents of Onychomycosis, as Determined by Minimum Inhibitory and Fungicidal Concentrations
 JOURNAL OF CLINICAL MICROBIOLOGY, May 2011, p. 1716 1720 Vol. 49, No. 5 0095-1137/11/$12.00 doi:10.1128/jcm.00083-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Activity of
JOURNAL OF CLINICAL MICROBIOLOGY, May 2011, p. 1716 1720 Vol. 49, No. 5 0095-1137/11/$12.00 doi:10.1128/jcm.00083-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Activity of
LUZU (luliconazole) Cream, 1% for topical use Initial U.S. Approval: 2013
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUZU safely and effectively. See full prescribing information for LUZU. LUZU (luliconazole) Cream,
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUZU safely and effectively. See full prescribing information for LUZU. LUZU (luliconazole) Cream,
The US Onychomycosis Market (2017 Edition) July 2017
 The US Onychomycosis Market (2017 Edition) July 2017 The US Onychomycosis Market Report Scope of the Report The report entitled The US Onychomycosis Market (2017 Edition), provides analysis of the US onychomycosis
The US Onychomycosis Market (2017 Edition) July 2017 The US Onychomycosis Market Report Scope of the Report The report entitled The US Onychomycosis Market (2017 Edition), provides analysis of the US onychomycosis
16.9 hours. Not reported 19.9 hours. Metabolism: Not known Elimination: Not known. Efficacy:
 Brand Name: Luzu Generic Name: luliconazole Manufacturer 1 : Medicis Pharmaceutical Corporation Drug Class 1,2,3,4 : Imidazole antifungal, topical Uses Labeled Uses 1,2,3,4 : Topical treatment of interdigital
Brand Name: Luzu Generic Name: luliconazole Manufacturer 1 : Medicis Pharmaceutical Corporation Drug Class 1,2,3,4 : Imidazole antifungal, topical Uses Labeled Uses 1,2,3,4 : Topical treatment of interdigital
Ciclopirox Topical Solution, 8%, (Nail Lacquer), should be used only under medical supervision as described above. The effectiveness and safety of Cic
 117 Ciclopirox Topical Solution, 8% (Nail Lacquer) For use on fingernails and toenails and immediately adjacent skin only Not for use in eyes DESCRIPTION Ciclopirox Topical Solution, 8%, (Nail Lacquer)
117 Ciclopirox Topical Solution, 8% (Nail Lacquer) For use on fingernails and toenails and immediately adjacent skin only Not for use in eyes DESCRIPTION Ciclopirox Topical Solution, 8%, (Nail Lacquer)
Achilles foot screening project. Achilles foot screening project 1 3. , 20, Jpn. J. Med. Mycol. Vol. 44, , 2003 ISSN
 Jpn. J. Med. Mycol. Vol. 44, 253260, 2003 ISSN 09164804, 20,,.,,,,,,,,.,.,,,,. KOH,,,,.,,,,.,,,. Key words: tinea pedis, onychomycosis, prevalence, risk factor, epidemiology.,,,., 13,,, 3.,,. 1960,., 3
Jpn. J. Med. Mycol. Vol. 44, 253260, 2003 ISSN 09164804, 20,,.,,,,,,,,.,.,,,,. KOH,,,,.,,,,.,,,. Key words: tinea pedis, onychomycosis, prevalence, risk factor, epidemiology.,,,., 13,,, 3.,,. 1960,., 3
NAIL SURGERY TECHNIQUES
 NAIL SURGERY TECHNIQUES TRACEY C. VLAHOVIC, DPM FFPM RCPS (GLASG) ASSOCIATE PROFESSOR, J STANLEY AND PEARL LANDAU FACULTY FELLOW TEMPLE UNIVERSITY SCHOOL OF PODIATRIC MEDICINE, PHILA, PA THE AGENDA Pincer
NAIL SURGERY TECHNIQUES TRACEY C. VLAHOVIC, DPM FFPM RCPS (GLASG) ASSOCIATE PROFESSOR, J STANLEY AND PEARL LANDAU FACULTY FELLOW TEMPLE UNIVERSITY SCHOOL OF PODIATRIC MEDICINE, PHILA, PA THE AGENDA Pincer
A COMPARATIVE STUDY OF EFFICACY OF TERBINAFINE AND FLUCONAZOLE IN PATIENTS OF TINEA CORPORIS
 Int. J. Pharm. Med. & Bio. Sc. 2013 Kumar Amit et al., 2013 Research Paper ISSN 2278 5221 www.ijpmbs.com Vol. 2, No. 4, October 2013 2013 IJPMBS. All Rights Reserved A COMPARATIVE STUDY OF EFFICACY OF
Int. J. Pharm. Med. & Bio. Sc. 2013 Kumar Amit et al., 2013 Research Paper ISSN 2278 5221 www.ijpmbs.com Vol. 2, No. 4, October 2013 2013 IJPMBS. All Rights Reserved A COMPARATIVE STUDY OF EFFICACY OF
Biomedicine and Nursing 2017;3(1)
 The innovative management of Onychomycosis by the use of Diode Laser 808-nm: A Pilot Study Al-Sadat Mosbeh.(MD) 1, Ahmed Aladl(MD) 2, Hamed Mohamed Abdo.(MD) 3 1 Department of Dermatology, Faculty of Medicine,
The innovative management of Onychomycosis by the use of Diode Laser 808-nm: A Pilot Study Al-Sadat Mosbeh.(MD) 1, Ahmed Aladl(MD) 2, Hamed Mohamed Abdo.(MD) 3 1 Department of Dermatology, Faculty of Medicine,
Ketoconazole pills for toenail fungus
 Ketoconazole pills for toenail fungus The Borg System is 100 % Ketoconazole pills for toenail fungus Uses. See also Warning section. Ketoconazole is used to treat certain serious fungal infections in the
Ketoconazole pills for toenail fungus The Borg System is 100 % Ketoconazole pills for toenail fungus Uses. See also Warning section. Ketoconazole is used to treat certain serious fungal infections in the
JEADV REVIEW ARTICLE. Abstract. Conflicts of interest. Funding sources
 DOI: 10.1111/jdv.12458 JEADV REVIEW ARTICLE Opportunistic toenail onychomycosis. The fungal colonization of an available nail unit space by non-dermatophytes is produced by the trauma of the closed shoe
DOI: 10.1111/jdv.12458 JEADV REVIEW ARTICLE Opportunistic toenail onychomycosis. The fungal colonization of an available nail unit space by non-dermatophytes is produced by the trauma of the closed shoe
fosravuconazole, onychomycosis, oral antifungal agents, randomized controlled trial, ravuconazole.
 doi: 10.1111/1346-8138.14607 Journal of Dermatology 2018; 45: 1151 1159 ORIGINAL ARTICLE Efficacy and safety of fosravuconazole L-lysine ethanolate, a novel oral triazole antifungal agent, for the treatment
doi: 10.1111/1346-8138.14607 Journal of Dermatology 2018; 45: 1151 1159 ORIGINAL ARTICLE Efficacy and safety of fosravuconazole L-lysine ethanolate, a novel oral triazole antifungal agent, for the treatment
Extended Abstract 1. Methods:
 Extended Abstract 1 Article: Chretien J.H. Efficacy of Undecylenic Acid- Zinc Undecylenate Powder in Culture Positive Tinea Pedis. International Journal of Dermatology. 1980; 19: 51-54. http://onlinelibrary.wiley.com.proxy.lib.uwaterloo.ca/doi/10.1111/j.1365-4362.1980.tb01997.x/pdf
Extended Abstract 1 Article: Chretien J.H. Efficacy of Undecylenic Acid- Zinc Undecylenate Powder in Culture Positive Tinea Pedis. International Journal of Dermatology. 1980; 19: 51-54. http://onlinelibrary.wiley.com.proxy.lib.uwaterloo.ca/doi/10.1111/j.1365-4362.1980.tb01997.x/pdf
Medical Policy. MP Laser Treatment of Onychomycosis
 Medical Policy MP 2.01.89 BCBSA Ref. Policy: 2.01.89 Last Review: 12/27/2017 Effective Date: 12/27/2017 Section: Medicine Related Policies 2.01.71 Nonpharmacologic Treatment of Rosacea 9.01.502 Experimental
Medical Policy MP 2.01.89 BCBSA Ref. Policy: 2.01.89 Last Review: 12/27/2017 Effective Date: 12/27/2017 Section: Medicine Related Policies 2.01.71 Nonpharmacologic Treatment of Rosacea 9.01.502 Experimental
Outline Dermatomycoses Definition: diseases or fungal infections of the skin Transmission of Dermatomycoses Case Report 1 Presentation of Disease
 Outline Dermatomycoses Tinea corporis,tinea capitis,tinea pedis, Tinea cruris, Definition: diseases or fungal infections of the skin Dermatophyte infections are caused by Trichophyton, Microsporum, and
Outline Dermatomycoses Tinea corporis,tinea capitis,tinea pedis, Tinea cruris, Definition: diseases or fungal infections of the skin Dermatophyte infections are caused by Trichophyton, Microsporum, and
overview of current strategies for monotherapy and combination therapy
 ORIGINAL ARTICLE JEADV (2005) 19, 21 29 DOI: 10.1111/j.1468-3083.2004.00988.x Topical antifungal drugs for the treatment of onychomycosis: an Blackwell Publishing, Ltd. overview of current strategies for
ORIGINAL ARTICLE JEADV (2005) 19, 21 29 DOI: 10.1111/j.1468-3083.2004.00988.x Topical antifungal drugs for the treatment of onychomycosis: an Blackwell Publishing, Ltd. overview of current strategies for
Types of Skin Infections
 Anatomy of Skin Types of Skin Infections Bacterial Impetigo Folliculitis Acne Fungal /Parasitic Tinea Pedis Tinea Cruris Tinea Versicolor Tinea Corporis Toenail fungus Allergic/Irritation conditions Dermatitis
Anatomy of Skin Types of Skin Infections Bacterial Impetigo Folliculitis Acne Fungal /Parasitic Tinea Pedis Tinea Cruris Tinea Versicolor Tinea Corporis Toenail fungus Allergic/Irritation conditions Dermatitis
Podiatric Perspectives on Nail Surgery
 Podiatric Perspectives on Nail Surgery Tracey C. Vlahovic, DPM Clinical Associate Professor, J. Stanley and Pearl Landau Faculty Fellow Temple University School of Podiatric Medicine Philadelphia, PA Common
Podiatric Perspectives on Nail Surgery Tracey C. Vlahovic, DPM Clinical Associate Professor, J. Stanley and Pearl Landau Faculty Fellow Temple University School of Podiatric Medicine Philadelphia, PA Common
Clinical and Mycologic Characteristics of Onychomycosis in Diabetic Patients
 Acta Dermatovenerologica Croatica 2010;18(2):84-91 CLINICAL ARTICLE Clinical and Mycologic Characteristics of Onychomycosis in Diabetic Patients Nawaf Al-Mutairi, Bayoumy Ibrahim Eassa, Dhuha Abdullah
Acta Dermatovenerologica Croatica 2010;18(2):84-91 CLINICAL ARTICLE Clinical and Mycologic Characteristics of Onychomycosis in Diabetic Patients Nawaf Al-Mutairi, Bayoumy Ibrahim Eassa, Dhuha Abdullah
mycoses Onychomycosis in Tehran: mycological study of 504 patients Summary Introduction
 mycoses Diagnosis,Therapy and Prophylaxis of Fungal Diseases Original article Onychomycosis in Tehran: mycological study of 504 patients S. J. Hashemi, 1 M. Gerami, 1 E. Zibafar, 1 M. Daei, 1 M. Moazeni
mycoses Diagnosis,Therapy and Prophylaxis of Fungal Diseases Original article Onychomycosis in Tehran: mycological study of 504 patients S. J. Hashemi, 1 M. Gerami, 1 E. Zibafar, 1 M. Daei, 1 M. Moazeni
Papers. Double blind, randomised study of continuous terbinafine compared with intermittent itraconazole in treatment of toenail onychomycosis
 Double blind, randomised study of continuous terbinafine compared with intermittent itraconazole in treatment of toenail onychomycosis E Glyn V Evans, Bárdur Sigurgeirsson for the LION study group Abstract
Double blind, randomised study of continuous terbinafine compared with intermittent itraconazole in treatment of toenail onychomycosis E Glyn V Evans, Bárdur Sigurgeirsson for the LION study group Abstract
A Prospective Study on the Epidemiology of Onychomycosis in Tertiary Care Hospital
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 7 Number 08 (2018) Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2018.708.383
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 7 Number 08 (2018) Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2018.708.383
Below is the indication and summary of the most serious and most common risks associated with the use of Naftin Cream, 2%. 2
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Silver Spring, MD 20993 TRANSMITTED BY FACSIMILE Misty M. D Ottavio, Senior Manager Regulatory Affairs 4215 Tudor
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Silver Spring, MD 20993 TRANSMITTED BY FACSIMILE Misty M. D Ottavio, Senior Manager Regulatory Affairs 4215 Tudor
Fungal nail infection: diagnosis and management
 Follow the link from the online version of this article to obtain certified continuing medical education credits Fungal nail infection: diagnosis and management Samantha Eisman, Rodney Sinclair Sinclair
Follow the link from the online version of this article to obtain certified continuing medical education credits Fungal nail infection: diagnosis and management Samantha Eisman, Rodney Sinclair Sinclair
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: itraconazole (Sporanox) Reference Number: HIM.PA.36 Effective Date: 05/16 Last Review Date: Line of Business: Health Insurance Marketplace Coding Implications Revision Log See Important
Clinical Policy: itraconazole (Sporanox) Reference Number: HIM.PA.36 Effective Date: 05/16 Last Review Date: Line of Business: Health Insurance Marketplace Coding Implications Revision Log See Important
International Journal of Scientific & Engineering Research Volume 9, Issue 4, April ISSN Laser Treatment of Onychomycosis
 International Journal of Scientific & Engineering Research Volume 9, Issue 4, April-2018 712 Laser Treatment of Onychomycosis Olga Olisova, MD, PhD 1, Igor Pinson, MD, PhD 2, Ekaterina Anpilogova, MD 1
International Journal of Scientific & Engineering Research Volume 9, Issue 4, April-2018 712 Laser Treatment of Onychomycosis Olga Olisova, MD, PhD 1, Igor Pinson, MD, PhD 2, Ekaterina Anpilogova, MD 1
Pharmacokinetics of three doses of once-weekly fluconazole (150, 300, and 450 mg) in distal subungual onychomycosis of the toenail
 SUPPORTED BY AN EDUCATIONAL GRANT FROM U.S. PHARMACEUTICALS, PFIZER INC. Pharmacokinetics of three doses of once-weekly fluconazole (150, 300, and 450 mg) in distal subungual onychomycosis of the toenail
SUPPORTED BY AN EDUCATIONAL GRANT FROM U.S. PHARMACEUTICALS, PFIZER INC. Pharmacokinetics of three doses of once-weekly fluconazole (150, 300, and 450 mg) in distal subungual onychomycosis of the toenail
In 2005, the estimated number of
 Treatment of Onychomycosis in Diabetic Patients Jason A. Winston and Jami L. Miller, MD In 2005, the estimated number of Americans with diabetes was 20.8 million people, with an additional 1.5 million
Treatment of Onychomycosis in Diabetic Patients Jason A. Winston and Jami L. Miller, MD In 2005, the estimated number of Americans with diabetes was 20.8 million people, with an additional 1.5 million
Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence
 Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence Presentation at Remium Capital Market Day, December 2, 2014 Peter Wolpert, CEO & Founder 0
Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence Presentation at Remium Capital Market Day, December 2, 2014 Peter Wolpert, CEO & Founder 0
IJBCP International Journal of Basic & Clinical Pharmacology
 Print ISSN 2319-2003 Online ISSN 2279-0780 IJBCP International Journal of Basic & Clinical Pharmacology doi: 10.18203/2319-2003.ijbcp20150020 Research Article Efficacy safety of 1% terbinafine hydrochloride
Print ISSN 2319-2003 Online ISSN 2279-0780 IJBCP International Journal of Basic & Clinical Pharmacology doi: 10.18203/2319-2003.ijbcp20150020 Research Article Efficacy safety of 1% terbinafine hydrochloride
Phoebe Rich MD Adjunct Professor OHSU Portland, Oregon
 Nail Tips for Diagnosis and Management of Nail Disorders Winter Clinical Dermatology Conference 2017 Hawaii Phoebe Rich MD Adjunct Professor OHSU Portland, Oregon Objectives diagnostic clues for benign
Nail Tips for Diagnosis and Management of Nail Disorders Winter Clinical Dermatology Conference 2017 Hawaii Phoebe Rich MD Adjunct Professor OHSU Portland, Oregon Objectives diagnostic clues for benign
Background 1/29/2018. Lasers and Lights for Onychomycosis KEYVAN NOURI, MD. Keyvan Nouri, M.D. White superficial. onychomycosis
 Lasers and Lights for Onychomycosis KEYVAN NOURI, MD Keyvan Nouri, M.D. Professor of Dermatology, Ophthalmology, Otolaryngology & Surgery Louis C. Skinner, Jr., M.D. Endowed Chair in Dermatology Richard
Lasers and Lights for Onychomycosis KEYVAN NOURI, MD Keyvan Nouri, M.D. Professor of Dermatology, Ophthalmology, Otolaryngology & Surgery Louis C. Skinner, Jr., M.D. Endowed Chair in Dermatology Richard
NAIL DRUG DELIVERY SYSTEM-A PROMISING ROUTE TO TREAT NAIL DISORDERS
 NAIL DRUG DELIVERY SYSTEM-A PROMISING ROUTE TO TREAT NAIL DISORDERS KAVYANJANA R NAIR, JASMIN ELSA MATHEW, PRIYA THOMAS, JIJU V, ELESSY ABRAHAM NAZARETH COLLEGE OF PHARMACY OTHERA P.O.,THIRUVALLA, KERALA
NAIL DRUG DELIVERY SYSTEM-A PROMISING ROUTE TO TREAT NAIL DISORDERS KAVYANJANA R NAIR, JASMIN ELSA MATHEW, PRIYA THOMAS, JIJU V, ELESSY ABRAHAM NAZARETH COLLEGE OF PHARMACY OTHERA P.O.,THIRUVALLA, KERALA
Rx only FOR TOPICAL DERMATOLOGIC USE ONLY, NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE
 STZC:R1 ERTACZO TM (SERTACONAZOLE NITRATE) CREAM, 2% Rx only FOR TOPICAL DERMATOLOGIC USE ONLY, NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE DESCRIPTION: ERTACZO TM (sertaconazole nitrate) Cream, 2%,
STZC:R1 ERTACZO TM (SERTACONAZOLE NITRATE) CREAM, 2% Rx only FOR TOPICAL DERMATOLOGIC USE ONLY, NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE DESCRIPTION: ERTACZO TM (sertaconazole nitrate) Cream, 2%,
This PDF is available for free download from a site hosted by Medknow Publications. ( Original Article ABSTRACT
 Original Article A clinical and mycological study of onychomycosis in HIV infection Amar Surjushe, Ratnakar Kamath, Chetan Oberai, Dattatray Saple, Minal Thakre*, Sujata Dharmshale**, Aruna Gohil** Department
Original Article A clinical and mycological study of onychomycosis in HIV infection Amar Surjushe, Ratnakar Kamath, Chetan Oberai, Dattatray Saple, Minal Thakre*, Sujata Dharmshale**, Aruna Gohil** Department
MERCY RETREAT Dermatology
 MERCY RETREAT 2016 Dermatology INFECTIONS IN DERMATOLOGY Why we do talk about infections today? These are some of the most commonly seen dermatologic diseases that present to primary care physician office
MERCY RETREAT 2016 Dermatology INFECTIONS IN DERMATOLOGY Why we do talk about infections today? These are some of the most commonly seen dermatologic diseases that present to primary care physician office
Comparison of in vitro antifungal activities of efinaconazole and. currently available antifungal agents against a variety of pathogenic
 AAC Accepts, published online ahead of print on 14 January 2013 Antimicrob. Agents Chemother. doi:10.1128/aac.02056-12 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 Comparison
AAC Accepts, published online ahead of print on 14 January 2013 Antimicrob. Agents Chemother. doi:10.1128/aac.02056-12 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 Comparison
TINEA (FUNGAL) INFECTION
 1 Medical Topics - Tinea TINEA (FUNGAL) INFECTION Tinea infection There are 3 main groups of fungal organisms that can cause skin infections. They include dermatophytes, yeast and moulds. Dermatophytes
1 Medical Topics - Tinea TINEA (FUNGAL) INFECTION Tinea infection There are 3 main groups of fungal organisms that can cause skin infections. They include dermatophytes, yeast and moulds. Dermatophytes
Abbreviated Class Update: Topical Antifungal Agents. Month/Year of Review: September 2014 Date of Last Review: March 2014
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Comparison of diagnostic methods in the diagnosis of dermatomycoses and onychomycoses
 Original article Comparison of diagnostic methods in the diagnosis of dermatomycoses and onychomycoses V. Panasiti, 1 R. G. Borroni, 1 V. Devirgiliis, 1 M. Rossi, 1 L. Fabbrizio, 1 R. Masciangelo, 2 U.
Original article Comparison of diagnostic methods in the diagnosis of dermatomycoses and onychomycoses V. Panasiti, 1 R. G. Borroni, 1 V. Devirgiliis, 1 M. Rossi, 1 L. Fabbrizio, 1 R. Masciangelo, 2 U.
Clinico-mycological correlation in onychomycosis in a tertiary level hospital
 Original Article Clinico-mycological correlation in onychomycosis in a tertiary level hospital Abu Noman Md Iftekhar Rafiq*, A S M Zakaria**, Lubna Khondker**, Md Shirajul Islam Khan, Sharmin Doulah, Samaresh
Original Article Clinico-mycological correlation in onychomycosis in a tertiary level hospital Abu Noman Md Iftekhar Rafiq*, A S M Zakaria**, Lubna Khondker**, Md Shirajul Islam Khan, Sharmin Doulah, Samaresh
75th AAD Annual Meeting
 75th AAD Annual Meeting Poster nº 4873 A phase 3 randomized, double-blind, trial comparing the efficacy and safety of the fixed combination calcipotriene 0.005% (Cal) and betamethasone dipropionate 0.064%
75th AAD Annual Meeting Poster nº 4873 A phase 3 randomized, double-blind, trial comparing the efficacy and safety of the fixed combination calcipotriene 0.005% (Cal) and betamethasone dipropionate 0.064%
Title. Establishment of a novel model of onychomycosis in the rabbit. for evaluation of antifungal agents. Running title
 AAC Accepts, published online ahead of print on May 0 Antimicrob. Agents Chemother. doi:./aac.00- Copyright 0, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
AAC Accepts, published online ahead of print on May 0 Antimicrob. Agents Chemother. doi:./aac.00- Copyright 0, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
To order reprints or e-prints of JDD articles please contact JDD
 June 2017 534 VOLUME 16 ISSUE 6 Copyright 2017 ORIGINAL ARTICLE Journal of Drugs in Dermatology The Efficacy and Safety of Azelaic Acid 15% Foam in the Treatment of Truncal Acne Vulgaris Lauren K. Hoffman
June 2017 534 VOLUME 16 ISSUE 6 Copyright 2017 ORIGINAL ARTICLE Journal of Drugs in Dermatology The Efficacy and Safety of Azelaic Acid 15% Foam in the Treatment of Truncal Acne Vulgaris Lauren K. Hoffman
Oral treatments for fungal infections of the skin of the foot (Review)
 Cochrane Database of Systematic Reviews Oral treatments for fungal infections of the skin of the foot (Review) Bell-Syer SEM, Khan SM, Torgerson DJ Bell-Syer SEM, Khan SM, Torgerson DJ. Oral treatments
Cochrane Database of Systematic Reviews Oral treatments for fungal infections of the skin of the foot (Review) Bell-Syer SEM, Khan SM, Torgerson DJ Bell-Syer SEM, Khan SM, Torgerson DJ. Oral treatments
U008 A Practical Approach to the Management of Onychomycosis: Diagnosis
 U008 A Practical Approach to the Management of Onychomycosis: Diagnosis Molly Hinshaw, MD Assoc. Prof. Dermatology & Dermatopathology UWHealth Madison, WI Key Points 1. Confirm dx before initiating treatment
U008 A Practical Approach to the Management of Onychomycosis: Diagnosis Molly Hinshaw, MD Assoc. Prof. Dermatology & Dermatopathology UWHealth Madison, WI Key Points 1. Confirm dx before initiating treatment
DERMATOPHYTE AND NON-DERMATOPHYTE ONYCHOMYCOSIS IN SINGAPORE
 Australas J. Dermatol 992; : 59-6 DERMATOPHYTE AND NON-DERMATOPHYTE ONYCHOMYCOSIS IN SINGAPORE JOYCE TENG-EE LIM, HOCK CHENG CHUA AND CHEE LEOK GOH Singapore SUMMARY Onychomycosis is caused by dermatophytes,
Australas J. Dermatol 992; : 59-6 DERMATOPHYTE AND NON-DERMATOPHYTE ONYCHOMYCOSIS IN SINGAPORE JOYCE TENG-EE LIM, HOCK CHENG CHUA AND CHEE LEOK GOH Singapore SUMMARY Onychomycosis is caused by dermatophytes,
Chapter 10 Nail Disorders & Diseases
 Chapter 10 Nail Disorders & Diseases MULTIPLE CHOICE 1. The surface of a healthy natural nail should be. a. smooth and spotted b. flexible and spotted c. smooth and unspotted d. pitted and rough ANS: C
Chapter 10 Nail Disorders & Diseases MULTIPLE CHOICE 1. The surface of a healthy natural nail should be. a. smooth and spotted b. flexible and spotted c. smooth and unspotted d. pitted and rough ANS: C
Pennsylvania Academy of Family Physicians Foundation & UPMC 43rd Refresher Course in Family Medicine CME Conference March 10 13, 2016
 Pennsylvania Academy of Family Physicians Foundation & UPMC 43rd Refresher Course in Family Medicine CME Conference March 10 13, 2016 Disclosures: Dermatology Potpourri Authur Huen, MD Speaker has no disclosures
Pennsylvania Academy of Family Physicians Foundation & UPMC 43rd Refresher Course in Family Medicine CME Conference March 10 13, 2016 Disclosures: Dermatology Potpourri Authur Huen, MD Speaker has no disclosures
Daily Oxymetazoline Cream Demonstrates High and Sustained Efficacy in Patients With Persistent Erythema of Rosacea Through 52 Weeks of Treatment
 Accepted Manuscript Daily Oxymetazoline Cream Demonstrates High and Sustained Efficacy in Patients With Persistent Erythema of Rosacea Through 52 Weeks of Treatment Michael H. Gold, MD, FAAD, Mark Lebwohl,
Accepted Manuscript Daily Oxymetazoline Cream Demonstrates High and Sustained Efficacy in Patients With Persistent Erythema of Rosacea Through 52 Weeks of Treatment Michael H. Gold, MD, FAAD, Mark Lebwohl,
Modelling A decision tree was used to estimate benefits and costs of alternative agents.
 Economic evaluation of antifungal agents in the treatment of toenail onychomycosis in Germany Van Doorslaer E K, Tormans G, Gupta A K, Van Rossem K, Eggleston A, Dubois D J, De Doncker P, Haneke E Record
Economic evaluation of antifungal agents in the treatment of toenail onychomycosis in Germany Van Doorslaer E K, Tormans G, Gupta A K, Van Rossem K, Eggleston A, Dubois D J, De Doncker P, Haneke E Record
Nail Matrix Disorders Nail Fold Disorders Nail Bed Disorders Nail Plate Disorders
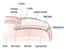 The most commonly reported nail conditions, seen by podiatrists, are: Onychocryptosis, 26% Onychauxis & onychogryphosis 22.8% Onychophosis 18.8% Onychomycosis 8.3% Nail Matrix Disorders Nail Fold Disorders
The most commonly reported nail conditions, seen by podiatrists, are: Onychocryptosis, 26% Onychauxis & onychogryphosis 22.8% Onychophosis 18.8% Onychomycosis 8.3% Nail Matrix Disorders Nail Fold Disorders
At Risk Foot Care Protocol Menu
 At Risk Foot Care Protocol Menu New Patient /Existing Patient Update Evaluation Questions: Are you Diabetic? If yes, how long have you been Diabetic (when was it diagnosed?) Is your diabetes under control
At Risk Foot Care Protocol Menu New Patient /Existing Patient Update Evaluation Questions: Are you Diabetic? If yes, how long have you been Diabetic (when was it diagnosed?) Is your diabetes under control
How to cure toenail fungus
 P ford residence southampton, ny How to cure toenail fungus What Makes Zeta Clear The Best Fungal Nail Treatment. Zeta Clear is more effective than any other toenail fungus cream or home treatment out
P ford residence southampton, ny How to cure toenail fungus What Makes Zeta Clear The Best Fungal Nail Treatment. Zeta Clear is more effective than any other toenail fungus cream or home treatment out
P4081 Secukinumab skin clearance is associated with greater improvements in patient-reported pain, itching, and scaling
 P4081 Secukinumab skin clearance is associated with greater improvements in patient-reported pain, itching, and scaling Mark Lebwohl, 1 Andrew Blauvelt, 2 Matthias Augustin, 3 Yang Zhao, 4 Isabelle Gilloteau,
P4081 Secukinumab skin clearance is associated with greater improvements in patient-reported pain, itching, and scaling Mark Lebwohl, 1 Andrew Blauvelt, 2 Matthias Augustin, 3 Yang Zhao, 4 Isabelle Gilloteau,
