ASPECTRUM of precisely defined eating disorders, including
|
|
|
- Loreen Gallagher
- 6 years ago
- Views:
Transcription
1 X/99/$03.00/0 Vol. 84, No. 2 Journal of Clinical Endocrinology and Metabolism Printed in U.S.A. Copyright 1999 by The Endocrine Society Impact of Binge Eating on Metabolic and Leptin Dynamics in Normal Young Women* ANN E. TAYLOR, JANE HUBBARD, AND ELLEN J. ANDERSON Reproductive Endocrine Unit and National Center for Infertility Research (A.E.T.), and the Mallinkrodt General Clinical Research Center (J.H., E.J.A.), Massachusetts General Hospital, Boston, Massachusetts ASPECTRUM of precisely defined eating disorders, including anorexia nervosa, bulimia nervosa, and binge eating disorder, are remarkably common in college age American women (1). Up to 30% of obese Americans seeking weight loss therapy (2 4) and up to 50% of obese adolescent girls (5) meet newly described strict criteria for binge eating disorder (DSM-IV). In addition, an even larger number of Americans exhibit some binge eating behavior that does not meet these strict criteria. In one series, 29.6% of college women presenting to a primary care clinic admitted to binge eating episodes (6). Both binge eating disorder and bingeing behavior are associated with a poorer response to weight loss therapy (3, 7) and poorer glycemic control in adolescent women with diabetes mellitus (8, 9). In addition, some reports suggest that binge eating may be associated with polycystic ovary syndrome (10). Other hormonal and metabolic defects, including hypogonadotropic amenorrhea, hypercortisolism, and insulin resistance, have been clearly described in women with the more severe classic eating disorders, such Received September 14, Revision received November 4, Accepted November 10, Address all correspondence and requests for reprints to: Ann E. Taylor, M.D., Reproductive Endocrine Unit, Massachusetts General Hospital, BHX-5, 55 Fruit Street, Boston, Massachusetts aetaylor@partners.org. * This work was supported by Grants R29-HD , P30-HD 28138, and M01-RR ABSTRACT Well defined eating disorders such as anorexia nervosa and bulimia are associated with significant known health risks. Although binge eating behavior is increased in unsuccessfully dieting obese women, other health implications of this common eating pattern are unknown. We hypothesized that ingestion of an entire day s calories at one time in the evening, a common eating practice among Americans, would lead to disruptions in glucose, insulin, and leptin metabolism and in menstrual cyclicity, even in healthy young women. Seven lean women without a history of eating disorders were studied on two occasions separated by one or two menstrual cycles. During one admission, they ate three regular meals plus a snack on each of 3 days. On the other admission, they ate the same number of calories, macronutrient matched to the normal diet, in a single evening meal. Glucose, insulin, and leptin were measured frequently for h beginning at 0800 h on the third day of each diet, and an insulin tolerance test was performed while the subjects were fasting on the fourth day. Daily blood samples were obtained until ovulation was documented to assess any impact on menstrual function. Ingestion of an entire day s calories at dinner resulted in a significant increase in fasting glucose levels and a dramatic increase in insulin responses to the evening meal. The diurnal pattern of leptin secretion was altered, such that the gradual rise in leptin from 0800 h observed during the normal diet was abolished, and leptin did not begin to rise during the binge diet until at least 2 h after the evening meal. No changes were demonstrated in insulin sensitivity, follicular growth, or ovulation between the two diets. We conclude that 1) ingestion of a large number of calories at one time (binge eating) impacts metabolic parameters even when total calories and macronutrients are appropriate for weight; 2) the timing of energy intake is an independent determinant of the diurnal rhythm of leptin secretion, indicating a relatively acute affect of energy balance on leptin dynamics; 3) the mechanism of exaggerated insulin secretion after a binge meal remains to be determined, but may be related to the altered diurnal pattern of leptin secretion; and 4) as most binge eating episodes in the population are associated with the ingestion of excess calories, it is hypothesized that binge eating behavior is associated with even greater metabolic dysfunction than that described herein. (J Clin Endocrinol Metab 84: , 1999) as anorexia nervosa and bulimia nervosa, but few studies have evaluated the consequences of binge eating alone. There are several potential mechanisms by which binge eating may cause or contribute to obesity. First, frequent binge meals would result in excess caloric ingestion, but a compensatory change in basal energy expenditure is not observed (11). Second, even if total calories were normal, the ingestion of fewer large meals may be metabolically worse than the ingestion of frequent small meals. Although the ingestion of six or nine small meals in a day has previously been demonstrated to reduce insulin secretion compared to the ingestion of three calorie- and nutrient-matched meals (12 14), the effects of a single daily meal, as is frequently observed in bingers and in many allegedly healthy Americans, has not previously been determined. If insulin levels are further increased by bingeing, they may contribute to increased hunger (15). Finally, recent studies suggest that the adipocyte hormone leptin could modify the response to binge eating. Serum leptin levels demonstrate a diurnal rhythm with a peak in the middle of the night (16, 17) when most humans are not eating and a nadir first thing in the morning. Fasting results in a gradual decline in leptin levels, first observed after 6 8 h (18). Thus, modulation of the diurnal pattern of leptin secretion by the timing of meals might have important implications for modulation of hunger. In addition to potential metabolic effects, binge eating is 428
2 METABOLIC IMPACT OF BINGE EATING 429 hypothesized to have effects on the hypothalamic-pituitarygonadal axis. For example, an increase in serum insulin levels may increase ovarian androgen secretion (19, 20) and/or modify pituitary gonadotropin secretion (21), thereby disrupting menstrual cyclicity. In addition, abolition of the diurnal pattern of leptin secretion is associated with amenorrhea in women athletes (22), suggesting that the leptin pattern is either a marker for or a response to a normal reproductive hormone axis. This study was designed to determine whether binge eating has adverse hormonal and metabolic effects. To study the effects of large meals per se independent of other potentially negative behaviors associated with bingeing, we studied lean normal women with regular ovulatory menstrual cycles and no history of eating disorders, who received meals that were matched for calorie and fat content. Subjects Subjects and Methods Seven lean normal Caucasian women with regular menstrual cycles participated in this study. Subjects were healthy with no medical illnesses and were taking no medications. Each had a history of regular 26- to 32-day menstrual cycles. Ovulation was documented in the luteal phase before the study by a serum progesterone level greater than 6 ng/dl. Subjects had no history of eating disorders and no evidence of a current eating disorder based on the BITE Edinburgh Bulimic Investigation Test questionnaire (23, 24). All subjects had normal TSH and PRL levels. The protocol was approved by the Massachusetts General Hospital Subcommittee on Human Studies, and all subjects gave their informed consent. Protocol Each subject was admitted to the Massachusetts General Hospital twice between h in the early to midfollicular phase (cycle days 1 7) during two different menstrual cycles separated by less than 3 months. Each admission lasted for 3 days, during which all caloric ingestion was monitored and controlled. During one admission, subjects received three meals a day and an afternoon snack, with 20% of calories at 0800 h for breakfast, 30% of calories at noon for lunch, 10% of calories at 1500 h for a snack, and the remaining 40% of calories at 1800 h for dinner. During the other admission, in random order, subjects received 100% of calories at 1800 h, with nothing but water and noncaloric, noncaffeinated beverages the rest of the day. For both diets, total calories were individually adjusted to maintain weight and were matched between studies of the same individual, and macronutrients were distributed as 50 55% carbohydrate, 35 40% fat, and 10 15% protein. Twenty-four-hour collections of urine were obtained each day for measurements of creatinine, cortisol, and catecholamines (epinephrine, norepinephrine, and dopamine) as monitors of stress during the admissions. On the third day (after 48 h of the test diet), each subject had blood sampled from 0800 h every 15 min for insulin and leptin and every 30 min for glucose. The first three women underwent 12 h of frequent sampling until 2000 h, but the sampling window was extended to 2200 h (14 h) for the remainder of the subjects, as glucose levels had not fallen to baseline by 2000 h after the 1800 h meal. At 0800 h on the fourth day, fasting blood was obtained for serum insulin, glucose, lipid, and androgen determinations before an insulin tolerance test. Regular human insulin (0.1 U/kg) was administered iv, and blood samples were obtained at 5, 0, 3, 5, 7, 10, and 15 min for measurement of glucose (25, 26). The insulin tolerance test was terminated by administration of iv glucose and/or the immediate ingestion of breakfast before discharge. After each discharge, each subject obtained a daily blood sample until ovulation was documented by urinary LH kits (Sure Step Ovulation Predictor, Applied Biotech, Inc., San Diego, CA). The ovulation date of the first cycle was used to predict menses and schedule the second admission on a similar cycle day as the first. Subjects were requested to take oral iron replacement (ferrous gluconate, 300 mg daily or twice daily) from the time of acceptance into the study until completion of the menstrual cycle of the second study. Assays Glucose was measured by the glucose hexokinase method. Insulin was measured by an automated immunometric assay with a sensitivity of 1.0 IU/L and intra- and interassay coefficients of variation of 3% and 6 12%, respectively. Leptin was measured by a RIA (Linco Research, Inc. St. Charles, MO) with a sensitivity of 0.5 ng/ml and intra- and interassay coefficients of variation of 4.5 6% and %, respectively. Urinary free cortisol was measured in a coated tube RIA (Incstar Corp., Stillwater, MN) after methylene chloride extraction. Urinary catecholamines were measured by Nichols Institute Diagnostics (San Juan Capistrano, CA). LH, FSH, estradiol, progesterone, testosterone, androstenedione, 17-hydroxyprogesterone, and dehydroepiandrosterone sulfate were measured by in-house RIAs as previously described (27). Cholesterol was measured by peroxidase-phenol-4-amino phenazone modified Trinder method, triglycerides by peroxidase enzymatic reaction, and high density lipoprotein and low density lipoprotein were measured by an enzymatic, colorimetric, nonprecipitate method. All lipids were measured using Boehringer Mannheim reagents (Indianapolis, IN) on a Hitachi Scientific Instruments 917 machine (Hialeah, FL). Statistical analysis Fasting glucose, insulin, and leptin on day 3 and fasting glucose, insulin, androgens, and lipids on day 4 were compared between diets by paired t testing. Mean levels of glucose, insulin, and leptin for the frequent sampling sessions were determined by averaging the individual samples and were compared by paired t testing. Similarly, mean levels of glucose and insulin were determined and compared for the window after the evening meal (1815 to 2000 or 2000 h). Lastly, glucose, insulin, and leptin levels across the day were compared by ANOVA for repeated measures. Insulin sensitivity was estimated from the glucose disappearance rate (K itt ), which was calculated from the slope of the fall in glucose after insulin administration (26). The day of the LH surge was determined by using three of the following four criteria: the day of the peak LH, the day of the peak FSH, the day of or the day after the estradiol peak, and the day that progesterone doubled or exceeded 0.6 ng/ml (28). The day of the LH surge and the day of onset of menses were used to determine follicular and luteal phase lengths for each subject. LH, FSH, and estradiol levels were centered to the day of ovulation for each subject and meaned between subjects. In addition, classic statistical methods for cross-over study designs were employed to demonstrate that there was no effect of diet order on any of the variables tested (29). Results Patient characteristics The mean subject age was yr (range, yr) and the mean body mass index was kg/m 2 (range, ). The mean height was cm (range, ). During the normal diet, the mean body weight was kg on day 1 and kg on day 4 (P 0.01, by paired t test). On the binge diet, the mean weight was kg on day 1 and kg on day 4 (P 0.18). However, there was no significant difference in weights on the last day of the two diets. Metabolic characteristics Mean glucose and insulin levels over the 12 h of frequent sampling on the third day of the diets are shown for the normal and binge diets in Fig. 1. On the normal diet, glucose and insulin rose as expected after each meal and gradually declined after dinner. On the binge diet, glucose and insulin
3 430 TAYLOR, HUBBARD, AND ANDERSON JCE&M 1999 Vol 84 No 2 FIG. 1. Mean glucose and insulin levels ( SEM) from h during the normal (open circles) and binge (filled circles) diets. Inverted triangles identify each time point at which levels are significantly different between the two diets, based on paired t testing. TABLE 1. Lipids and androgens Normal Binge Mean SEM Mean SEM Paired, two-sided t test (P value) Total cholesterol (mg/dl) Triglycerides (mg/dl) HDL cholesterol (mg/dl) LDL cholesterol (mg/dl) Testosterone (ng/dl) Hydroxy progesterone (ng/ml) Androstenedione (ng/ml) DHEA-S ( g/ml) Follicular phase length (days) levels fell gradually across the day, rising after the evening meal. Fasting glucose was significantly higher on the binge diet on both days 3 and 4 (day 3: normal, 86 2 mg/dl; binge, 94 2 mg/dl; P 0.003). The mean glucose level across the duration of the study was significantly lower in the binge patients because they did not have postprandial increases after breakfast and lunch (normal mean, 107 2; binge, 97 2 mg/dl; P 0.003). However, the glucose response to the evening meal (sum glucose from h) was remarkably similar between the two diets (normal, ; binge, mg/dl; P 0.37) considering that more than twice as many calories were ingested for the binge dinner. Fasting insulin levels were not significantly different on either the third or fourth day of either diet (normal, ; binge, U/mL; P 0.89). Similarly, the mean insulin levels from the entire frequent sampling study did not differ between the diets (normal, ; binge, U/mL; P 0.94). However, the insulin response to the evening meal (sum of insulin measurements from h) was strikingly increased after the binge dinner (normal, ; binge, U/mL; P 0.01) despite virtually identical glucose responses. The glucose responses to the insulin tolerance test were similar on the fourth day of the two diets, suggesting that there was no difference in insulin sensitivity. The K itt was U after the normal diet and after the binge diet (P 0.145). The binge diet did not result in any significant difference in fasting lipid levels by the fourth day of the diet (Table 1). Leptin The two diets resulted in strikingly different patterns of leptin secretion across the third day (Fig. 2). In each individual, serum leptin levels were consistently higher than baseline by 8 h after the initiation of breakfast on the normal diet and continued to rise at the end of the frequent sampling (Fig. 3). Conversely, during the binge diet, leptin levels continued to fall throughout the day and had not begun to rise by 2200 h. Thus, although there was no significant difference in fasting levels (normal, ; binge, ng/ml; P 0.29) between the two diets, the mean leptin levels across the third day were significantly lower on the binge diet (normal, ; binge, ng/ml; P 0.04). From
4 METABOLIC IMPACT OF BINGE EATING 431 FIG. 2. Leptin levels as a percentage of fasting levels during the normal diet ( SEM) from h during the normal (open circles) and binge (filled circles) diets. Inverted triangles identify each time point at which levels are significantly different between the two diets, based on paired t testing h, leptin averaged 45% lower on the binge diet than on the normal diet (P 0.005). Individual leptin secretion profiles from each individual (Fig. 3) demonstrate a pulsatile pattern of leptin secretion in all subjects. Menstrual cyclicity There was no significant difference in follicular or luteal phase length after 3 days of either diet (Table 1). After centering daily hormone levels to the day of ovulation, the patterns of gonadotropin and sex steroid secretion were indistinguishable after 3 days of either diet (data not shown). Finally, there was no difference in day 4 testosterone, 17- hydroxyprogesterone, androstenedione, or dehydroepiandrosterone sulfate (Table 1) between the two diets. Urine collections The data for urinary catecholamines, cortisol, creatinine, and volume are shown in Table 2. Although there was no difference in urine volume, creatinine, or catecholamine excretion between the first and second admissions, there was a small effect of admission order on total urinary free cortisol, such that cortisol excretion was higher during the first admission (52 9 vs g/24 h for admission 1 vs. 2, respectively; P 0.03). Although urine volume and creatinine and cortisol excretion were similar during the two diets, dopamine excretion was significantly greater during the binge diet (normal, ; binge, g/24 h; P 0.001). Discussion The present studies demonstrate that the ingestion of a single large daily meal results in metabolic changes in glucose, insulin, and leptin secretion. These findings are present in the healthiest subset of individuals, lean normal women without a history of eating disorders or menstrual dysfunction, and occurred without the ingestion of excess calories or excess fat. Fasting glucose was increased % on the binge compared to the normal diet. In addition, the insulin response to the binge evening meal was dramatically increased (108 29%) compared to the insulin response to the normal dinner. Previous studies have demonstrated that the ingestion of three meals a day results in higher total insulin levels than does the ingestion of six or nine small meals across the day (12 14). The current study, even given the limitation of sampling only for h, demonstrates that this trend extends to even a single daily meal. The mechanism of hyperinsulinemia after the binge meal in these normal subjects remains to be determined. Although the doubling of insulin levels in response to similar glucose levels raises the possibility that there is insulin resistance, the insulin tolerance test the next morning does not confirm this. However, we cannot exclude the possibility that there is acute insulin resistance after the evening meal that is resolved by the overnight fast. In addition, the insulin tolerance test may not be a sufficiently sensitive test to identify significant changes in insulin sensitivity in this normal population. In these studies, the increased insulin response to the binge dinner occurs without an increased glucose response compared to the normal dinner, raising the possibility that the observed hyperinsulinemia does not represent insulin resistance but, rather, an increase in other insulin secretagogues or a decrease in insulin suppressers. Glucagon-like peptide-1 (GLP-1) is a physiologically important regulator of insulin secretion that is itself secreted from the pancreas in response to oral, but not iv, glucose in a dose-dependent manner (30). Administration of increasing doses of exogenous GLP-1 stimulates increased insulin secretion up to 40% over the baseline (31, 32). Thus, increased GLP-1 secretion may explain part of the increased insulin response to the binge meal.
5 432 TAYLOR, HUBBARD, AND ANDERSON JCE&M 1999 Vol 84 No 2 FIG. 3. Individual leptin profiles during the normal (left) and binge (right) diets. The two diets for each individual are shown in the same row. During the normal diet, leptin levels rise gradually across the day in all subjects, whereas they tend to fall during the binge diet. The vertical dotted lines represent meal times during the two calorie-matched diets. Pulsatile leptin secretion can be observed in all subjects, but is more pronounced in those with higher basal leptin levels. TABLE h urine collections Admission order Diet Admit 1 Admit 2 P Normal Binge P Epinephrine ( g/24 h) 3.62 (0.51) 2.95 (0.79) (0.87) 3.47 (0.87) 0.64 Norepinephrine ( g/24 h) (2.46) (2.46) (2.87) (3.15) 0.22 Total catecholamines ( g/24 h) (2.78) (2.82) (3.13) (3.76) 0.17 Dopamine ( g/24 h) (32.1) (46.3) (32.8) (62.9) Creatinine (mg/24 h) (60.3) (121.4) (145.8) (53.7) 0.96 Urinary free cortisol ( g/24 h) (8.51) (6.25) (8.96) (8.03) 0.11 Vol (ml) (123.8) (185.6) (262.62) (244.29) 0.14 Values are means (SEM). Leptin may also influence insulin secretion. In ambient glucose conditions, physiological leptin administration suppresses insulin secretion from rat islet cells or a human islet cell line (33). During the binge diet, leptin levels were 45% lower at the time of the evening meal compared to those during the normal diet. Thus, decreased leptin may also contribute to the increased insulin response to the binge meal. A diurnal rhythm of leptin has previously been described (16) and has been postulated by some to be related to the cortisol diurnal rhythm (34 36). Our studies demonstrate that the pattern of food ingestion itself is an important factor
6 METABOLIC IMPACT OF BINGE EATING 433 controlling the leptin secretion profile, even when total 24-h cortisol excretion is constant. The current study design could not detect any potential change in the cortisol diurnal rhythm, however. In this study in which meal timing has been carefully controlled for 3 days, leptin secretion begins to increase approximately 6 h after food ingestion, a more acute response than may have been appreciated in previous studies. Careful circadian studies in which sleep, position, and caloric intake are constant for at least 24 h will be required to determine whether the entire diurnal rhythm of leptin is merely secondary to the pattern of food ingestion or whether there is also an underlying circadian rhythm that might reflect other circadian neuroendocrine rhythms. In addition, it will be critical to determine whether the leptin diurnal rhythm can be modulated by the timing of food intake, as opposed to the amount, as recent studies of women athletes demonstrate that a flattened leptin diurnal rhythm is one of the few parameters that distinguish amenorrheic from eumenorrheic subjects (22). Such studies suggest that the pattern of leptin secretion across the day may have profound reproductive implications. Preliminary data from other studies of similar subjects in whom blood sampling continues for a full 24 h (Taylor, A. E., unpublished) suggest that a leptin diurnal rhythm is still maintained, although phase shifted, during binge eating. This is consistent with recent studies demonstrating a change in the leptin diurnal rhythm in response to simulated jet lag, even before the cortisol rhythm changed significantly (37). Thus, it may not be surprising that 3 days of binge eating were insufficient to induce a significant change in follicular or luteal phase length in the lean healthy young women. Whether modification of the pattern of caloric intake, with or without caloric restriction, will induce a physiologically significant flattening of the leptin diurnal rhythm remains to be determined. Our studies confirm the pulsatile nature of leptin secretion (34, 38), which is visible even when serum is sampled as infrequently as every 15 min. However, recent reports that leptin pulses typically occur approximately every 30 min (38) suggests that the sampling frequency for detecting the true frequency of pulsatile leptin secretion should be at least once every 5 min. This estimate is based on previous evidence that sampling frequency should be 6 times the interpulse interval (39). Because the sampling protocol employed in this study was suboptimal, formal pulse analysis was not performed. However, by visual inspection of Fig. 3, it appears that leptin pulsatile secretion is modulated by dietary intake, with an increase in pulse amplitude as overall leptin levels are also rising. The use of a more frequent sampling regimen will be required to determine whether pulse frequency changes with meals. The urine measurements of volume, creatinine, cortisol, and catecholamines were intended as a control for stress responses and metabolic balance during the 3 days of hospitalization and dietary control. As the development of obesity has been correlated with decreased sympathetic outflow in many experimental models (40), it was critical to demonstrate that bingeing did not exert its effects through modulation of epinephrine and norepinephrine excretion. Although there was a statistically significant increase in cortisol excretion during the first admission, the absolute magnitude was small and well within the normal range, making it unlikely to be physiologically relevant. Importantly, there was no difference in cortisol excretion between the two diets. However, there was a striking increase in urinary dopamine excretion during the binge diet. Increased dopamine excretion has been associated with insulin resistance (41) and hypertension (42) in different populations, suggesting that it may be a true marker of an adverse outcome of binge eating. As the urine collections were for the full 24 h, whereas blood sampling only lasted for h, the increased urinary dopamine secretion with the binge diet suggests that the exaggerated insulin secretion persisted throughout the night. In summary, the ingestion of a single large daily meal has metabolic consequences, including increased fasting glucose levels, increased insulin responses to the binge meal, and modulation of the leptin diurnal rhythm. These factors, demonstrated here in young healthy lean women, are likely to be exacerbated in individuals who are already obese or insulin resistant. Because hyperinsulinemia has been associated with many adverse health outcomes, including an increased risk of cardiovascular disease, hypertension, and dyslipidemia, further studies are required to determine the long term consequences of the common practice of eating large evening meals. Acknowledgments We are grateful to Brain McCourt and Brooke Wexler for recruiting and managing the subjects and data for this complex protocol, and to the nurses on the Mallinkrodt General Clinical Research Center for their fastidious care of our subjects. References 1. Health and Public Policy Committee, American College of Physicians Eating disorders: anorexia nervosa and bulimia. Ann Intern Med. 105: Adami GF, Gandolfo P, Scopinaro N Binge eating in obesity. Int J Obesity. 20: Ferguson, KJ; Spitzer, RL Binge eating disorder in a community-based sample of successful and unsuccessful dieters. Int J Eating Disord. 18: Bruce, B; Wilfley, D Binge eating among the overweight population: a serious and prevalent problem. J Am Diet Assoc. 96: Berkowitz R, Stunkard AJ, Stallings VA Binge-eating disorder in obese adolescent girls. Ann NY Acad Sci. 699: Kurtzman FD, Yager J, Landsverk J, Wiesmeier E, Bodurka DC Eating disorders among selected female student populations at UCLA. J Am Diet Assoc. 89: Smith MC, Sundhaus E, Porzelius LK Effect of binge eating on the prediction of weight loss in obese women. J Behav Med. 18: Stancin T, Link DL, Reuter JM Binge eating and purging in young women with IDDM. Diabetes Care. 12: Wing RR, Nowalk MP, Marcus MD, Koeske R, Finegold D Subclinical eating disorders and glycemic control in adolescents with type I diabetes. Diabetes Care. 9: McCluskey SE, Lacey JH, Pearce JM Binge-eating and polycystic ovaries [Letter]. Lancet. 340: Alger S, Seagle H, Ravussin E Food intake and energy expenditure in obese female bingers and non-bingers. Int J Obesity. 19: Jones PJH, Leitch CA, Pederson RA Meal-freqeuncy effects on plasma hormone concentrations and cholesterol synthesis in humans. Am J Clin Nutr. 57: Arnold LM, Ball MJ, Duncan AW, Mann J Effect of isoenergetic intake of three or nine meals on plasma lipoproteins and glucose metabolism. Am J Clin Nutr. 57: Jenkins DJA, Wolever TMS, Vuksan V, et al Nibbling vs. gorging: metabolic advantages of increased meal frequency. N Engl J Med. 321: Stricker EM Biological bases of hunger and satiety: therapeutic implications. Nutr Rev. 42: Sinha MK, Ohannesian JP, Heiman ML, et al Nocturnal rise of leptin in lean, obese, and non-insulin-dependent diabetes mellitus subjects. J Clin Invest. 97: Pombo M, Herrera-Justiniano E, Considine RV, et al Nocturnal rise of
7 434 TAYLOR, HUBBARD, AND ANDERSON JCE&M 1999 Vol 84 No 2 leptin in normal prepubertal and pubertal children and in patients with perinatal stalk-transection syndrome. J Clin Endocrinol Metab. 82: Boden G, Chen X, Mozzoli M, Ryan I Effect of fasting on serum leptin in normal human subjects. J Clin Endocrinol Metab. 81: Barbieri RL, Makris A, Ryan KJ Insulin stimulates androgen accumulation in incubations of human ovarian stroma and theca. Obstet Gynecol. 64: Hernandez ER, Resnick CE, Svoboda ME, Van Wyk JJ, Payne DW, Adashi EY Somatomedin-C/insulin-like growth factor I as an enhancer of androgen biosynthesis by cultured rat ovarian cells. Endocrinology. 122: Adashi EY, Hsueh AJW, Yen SSC Insulin enhancement of luteinizing hormone and follicle-stimulating hormone release by cultured pituitary cells. Endocrinology. 108: Laughlin GA, Yen SSC Nutritional and endocrine-metabolic aberrations in amenorrheic athletes. J Clin Endocrinol Metab. 81: Henderson M, Freeman CPL A self-rating scale for bulimia: the BITE. Br J Psychol. 150: Freeman CPL, Henderson M The BITE: indices of agreement. Br J Psychol. 152: Bonora E, Moghetti P, Zancanaro C, et al Estimates of in vivo insulin action in man: comparison of insulin tolerance tests with euglycemic and hyperglycemic glucose clamp studies. J Clin Endocrinol Metab. 68: Grulet H, Durlach V, Hecart AC, Gross A, Leutenegger M Study of the rate of early glucose disappearance following insulin injection: insulin sensitivity index. Diabetes Res Clin Pract. 20: Taylor AE, McCourt B, Martin KA, et al Determinants of abnormal gonadotropin secretion in clinically defined women with polycystic ovary syndrome. J Clin Endocrinol Metab. 82: Filicori M, Santoro N, Merriam GR, Crowley Jr WF Characterization of the physiological pattern of episodic gonadotropin secretion throughout the human menstrual cycle. J Clin Endocrinol Metab. 62: Armitage P, Hills M The two-period crossover trial. Statistician. 31: Habener JF The Incretin Notion and its relevance to diabetes. Endocrinol Metab Clin North Am. 22: Nathan DM, Schreiber E, Fogel H, Mojsov S, Habener JF Insulinotropic action of glucagonlike peptide-1-(7 37) in diabetic and nondiabetic subjects. Diabetes Care. 15: Elahi D, McAloon-Dyke M, Fukagawa NK, et al The insulinotropic actions of glucose-dependent insulinotropic polypeptide (GIP) and glucagonlike peptide-1 (7 37) in normal and diabetic subjects. Regul Pept. 51: Kieffer TJ, Heller RS, Leech CA, Holz GG, Habener JF Leptin suppression of insulin secretion by the activation of ATP-sensitive K channels in pancreatic -cells. Diabetes. 46: Licinio J, Mantzoros C, Negrao AB, et al Human leptin levels are pulsatile and inversely related to pituitary-adrenal function. Nat Med. 3: Korbonits M, Trainer PJ, Little JA, et al Leptin levels do not change acutely with food administration in normal or obese subjects, but are negatively correlated with pituitary-adrenal activity. Clin Endocrinol (Oxf). 46: Dagogo-Jack S, Selke G, Melson AK, Newcomer JW Robust leptin secretory response to dexamethasone in obese subjects. J Clin Endocrinol Metab. 82: Schoeller DA, Cella LK, Sinha MK, Caro JF Entrainment of the diurnal rhythm of plasma leptin to meal timing. J Clin Invest. 100: Sinha MK, Sturis J, Ohannesian J, et al Ultradian oscillations of leptin secretion in humans. Biochem Biophys Res Commun. 228: Filicori M, Flamigni C, Crowley Jr WF The critical role of blood sampling frequency in the estimation of episodic luteinizing hormone secretion in normal women. In: Crowley Jr WF, Hofler JG, eds. The episodic secretion of hormones. New York: Wiley and Sons; Bray GA, York DA Hypothalamic and genetic obesity in experimental animals: an autonomic and endocrine hypothesis. Physiol Rev. 59: Young JB, Troisi RJ, Weiss ST, Parker DR, Sparrow D, Landsberg L Relationship of catecholamine excretion to body size, obesity, and nutrient intake in middle-aged and elderly men. Am J Clin Nutr. 56: Saito I, Itsuji S, Takeshita E, et al Increased urinary dopamine excretion in young patients with essential hypertension. Clin Exp Hypertension. 16:29 39.
Jessicah S. Collins, Jennifer P. Beller, Christine Burt Solorzano, James T. Patrie, R. Jeffrey Chang, John C. Marshall, Christopher R.
 Supplemental Materials for manuscript entitled Blunted Day-Night Changes in Luteinizing Hormone Pulse Frequency in Girls with Obesity: the Potential Role of Hyperandrogenemia Jessicah S. Collins, Jennifer
Supplemental Materials for manuscript entitled Blunted Day-Night Changes in Luteinizing Hormone Pulse Frequency in Girls with Obesity: the Potential Role of Hyperandrogenemia Jessicah S. Collins, Jennifer
ROLE OF HORMONAL ASSAY IN DIAGNOSING PCOD DR GAANA SREENIVAS (JSS,MYSURU)
 ROLE OF HORMONAL ASSAY IN DIAGNOSING PCOD DR GAANA SREENIVAS (JSS,MYSURU) In 1935, Stein and Leventhal described 7 women with bilateral enlarged PCO, amenorrhea or irregular menses, infertility and masculinizing
ROLE OF HORMONAL ASSAY IN DIAGNOSING PCOD DR GAANA SREENIVAS (JSS,MYSURU) In 1935, Stein and Leventhal described 7 women with bilateral enlarged PCO, amenorrhea or irregular menses, infertility and masculinizing
Reproductive FSH. Analyte Information
 Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
INSULIN RESISTANCE, POLYCYSTIC OVARIAN SYNDROME An Overview
 INSULIN RESISTANCE, POLYCYSTIC OVARIAN SYNDROME An Overview University of PNG School of Medicine & Health Sciences Division of Basic Medical Sciences PBL MBBS III VJ Temple 1 Insulin Resistance: What is
INSULIN RESISTANCE, POLYCYSTIC OVARIAN SYNDROME An Overview University of PNG School of Medicine & Health Sciences Division of Basic Medical Sciences PBL MBBS III VJ Temple 1 Insulin Resistance: What is
WEIGHT GAIN DURING MENOPAUSE EMERGING RESEARCH
 MENOPAUSE WHEN DOES IT OCCUR? The cessation of the menstrual cycle for one year. WEIGHT GAIN DURING MENOPAUSE EMERGING RESEARCH Jan Schroeder, Ph.D. Chair of The Department of Kinesiology California State
MENOPAUSE WHEN DOES IT OCCUR? The cessation of the menstrual cycle for one year. WEIGHT GAIN DURING MENOPAUSE EMERGING RESEARCH Jan Schroeder, Ph.D. Chair of The Department of Kinesiology California State
Effect of troglitazone on endocrine and ovulatory performance in women with insulin resistance related polycystic ovary syndrome
 FERTILITY AND STERILITY VOL. 71, NO. 2, FEBRUARY 1999 Copyright 1999 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Effect of troglitazone
FERTILITY AND STERILITY VOL. 71, NO. 2, FEBRUARY 1999 Copyright 1999 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Effect of troglitazone
Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018
 Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018 Learning Objectives At the conclusion of this lecture, learners should: 1) Know the various diagnostic
Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018 Learning Objectives At the conclusion of this lecture, learners should: 1) Know the various diagnostic
Polycystic Ovary Syndrome
 What is the polycystic ovary syndrome? Polycystic Ovary Syndrome The polycystic ovary syndrome (PCOS) is a clinical diagnosis characterized by the presence of two or more of the following features: irregular
What is the polycystic ovary syndrome? Polycystic Ovary Syndrome The polycystic ovary syndrome (PCOS) is a clinical diagnosis characterized by the presence of two or more of the following features: irregular
Diagnos-Techs, Inc. Clinical & Research Laboratory PO BOX , Tukwila, WA Tel: (425) CLIA License # 50D
 Clinical & Research Laboratory PO BOX 389662, Tukwila, WA 98138-0662 Tel: (425) 251-0596 CLIA License # 50D0630141 ASI TAP Adrenal Stress Index Free Cortisol Rhythm 07:00-08:00 AM 11 Depressed 13-24 nm
Clinical & Research Laboratory PO BOX 389662, Tukwila, WA 98138-0662 Tel: (425) 251-0596 CLIA License # 50D0630141 ASI TAP Adrenal Stress Index Free Cortisol Rhythm 07:00-08:00 AM 11 Depressed 13-24 nm
BIOLOGY - CLUTCH CH.45 - ENDOCRINE SYSTEM.
 !! www.clutchprep.com Chemical signals allow cells to communicate with each other Pheromones chemical signals released to the environment to communicate with other organisms Autocrine signaling self-signaling,
!! www.clutchprep.com Chemical signals allow cells to communicate with each other Pheromones chemical signals released to the environment to communicate with other organisms Autocrine signaling self-signaling,
Endocrinology of the Female Reproductive Axis
 Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
EFFECT OF WEIGHT LOSS ON OVARIAN AND ADRENAL ANDROGEN SENSITIVITY. Department OB/GYN. EVMA Norfolk, Va Telephone: (804)
 This study predates but predicts the use of glucophage, exercise and diet for the treatment of PCO. EFFECT OF WEIGHT LOSS ON OVARIAN AND ADRENAL ANDROGEN SENSITIVITY Principal Investigator: Hofheimer Hall
This study predates but predicts the use of glucophage, exercise and diet for the treatment of PCO. EFFECT OF WEIGHT LOSS ON OVARIAN AND ADRENAL ANDROGEN SENSITIVITY Principal Investigator: Hofheimer Hall
The Intricate Web Of Hormone Relationships Dr. Ritamarie Loscalzo
 The Intricate Web Of Hormone Relationships Dr. Ritamarie Loscalzo Medical Disclaimer: The information in this presentation is not intended to replace a one-onone relationship with a qualified health care
The Intricate Web Of Hormone Relationships Dr. Ritamarie Loscalzo Medical Disclaimer: The information in this presentation is not intended to replace a one-onone relationship with a qualified health care
Endocrine part one. Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy
 Endocrine part one Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy HORMONES Hormones are chemicals released by a cell or a gland
Endocrine part one Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy HORMONES Hormones are chemicals released by a cell or a gland
Effect of macronutrients and mixed meals on incretin hormone secretion and islet cell function
 Effect of macronutrients and mixed meals on incretin hormone secretion and islet cell function Background. Following meal ingestion, several hormones are released from the gastrointestinal tract. Some
Effect of macronutrients and mixed meals on incretin hormone secretion and islet cell function Background. Following meal ingestion, several hormones are released from the gastrointestinal tract. Some
Study of the correlation between growth hormone deficiency and serum leptin, adiponectin, and visfatin levels in adults
 Study of the correlation between growth hormone deficiency and serum leptin, adiponectin, and visfatin levels in adults Z.-P. Li 1, M. Zhang 2, J. Gao 3, G.-Y. Zhou 3, S.-Q. Li 1 and Z.-M. An 3 1 Golden
Study of the correlation between growth hormone deficiency and serum leptin, adiponectin, and visfatin levels in adults Z.-P. Li 1, M. Zhang 2, J. Gao 3, G.-Y. Zhou 3, S.-Q. Li 1 and Z.-M. An 3 1 Golden
Digestion: Endocrinology of Appetite
 Digestion: Endocrinology of Dr. Ritamarie Loscalzo Medical Disclaimer: The information in this presentation is not intended to replace a one on one relationship with a qualified health care professional
Digestion: Endocrinology of Dr. Ritamarie Loscalzo Medical Disclaimer: The information in this presentation is not intended to replace a one on one relationship with a qualified health care professional
Relation of luteinizing hormone levels to body mass index in premenopausal women
 FERTILITY AND STERILITY VOL. 69, NO. 3, MARCH 1998 Copyright 1998 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Relation of luteinizing
FERTILITY AND STERILITY VOL. 69, NO. 3, MARCH 1998 Copyright 1998 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Relation of luteinizing
ENDOCRINOLOGY. Dr.AZZA SAJID ALKINANY 2 nd STAGE
 ENDOCRINOLOGY Dr.AZZA SAJID ALKINANY 2 nd STAGE THE RELATIONSHIP AMONG THE HYPOTHALMUS,POSTERIOR PITUITARY AND TARGET TISSUES. The posterior pituitary does not produce its own hormones, but stores and
ENDOCRINOLOGY Dr.AZZA SAJID ALKINANY 2 nd STAGE THE RELATIONSHIP AMONG THE HYPOTHALMUS,POSTERIOR PITUITARY AND TARGET TISSUES. The posterior pituitary does not produce its own hormones, but stores and
Case Questions. Polycystic Ovarian Syndrome: Treatment Goals and Options. Differential Diagnosis of Hyperandrogenic Anovulation
 Polycystic Ovarian Syndrome: Treatment Goals and Options Marc Cornier, MD Division of Endocrinology, Metabolism and Diabetes Colorado Center for Health and Wellness University of Colorado School of Medicine
Polycystic Ovarian Syndrome: Treatment Goals and Options Marc Cornier, MD Division of Endocrinology, Metabolism and Diabetes Colorado Center for Health and Wellness University of Colorado School of Medicine
SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY
 1 SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL SEMINAR: SEX HORMONES PART 1 An Overview What are steroid hormones? Steroid
1 SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL SEMINAR: SEX HORMONES PART 1 An Overview What are steroid hormones? Steroid
PCOS & Diet Therapy. Dr. Ladan Giahi Immunonutritionist Avicenna Research Institute October 2015
 PCOS & Diet Therapy Dr. Ladan Giahi Immunonutritionist Avicenna Research Institute October 2015 Questions to be discussed: 1) Why dietary modification is considered as first line of treatment? 2) What
PCOS & Diet Therapy Dr. Ladan Giahi Immunonutritionist Avicenna Research Institute October 2015 Questions to be discussed: 1) Why dietary modification is considered as first line of treatment? 2) What
Polycystic Ovary Syndrome
 Polycystic Ovary Syndrome Kathleen Colleran, MD Professor of Medicine University of New Mexico HSC Presented for COMM-TC May 4, 2012 Objectives Understand the pathophysiology of PCOS Understand how to
Polycystic Ovary Syndrome Kathleen Colleran, MD Professor of Medicine University of New Mexico HSC Presented for COMM-TC May 4, 2012 Objectives Understand the pathophysiology of PCOS Understand how to
Hormonal regulation of. Physiology Department Medical School, University of Sumatera Utara
 Hormonal regulation of nutrient metabolism Physiology Department Medical School, University of Sumatera Utara Homeostasis & Controls Successful compensation Homeostasis reestablished Failure to compensate
Hormonal regulation of nutrient metabolism Physiology Department Medical School, University of Sumatera Utara Homeostasis & Controls Successful compensation Homeostasis reestablished Failure to compensate
The enteroinsular axis in the pathogenesis of prediabetes and diabetes in humans
 The enteroinsular axis in the pathogenesis of prediabetes and diabetes in humans Young Min Cho, MD, PhD Division of Endocrinology and Metabolism Seoul National University College of Medicine Plasma glucose
The enteroinsular axis in the pathogenesis of prediabetes and diabetes in humans Young Min Cho, MD, PhD Division of Endocrinology and Metabolism Seoul National University College of Medicine Plasma glucose
PCOS and Your Fertility. Jim Toner, MD, PhD Atlanta Center for Reproductive Medicine
 PCOS and Your Fertility Jim Toner, MD, PhD Atlanta Center for Reproductive Medicine PCOS Consequences Androgen excess Acne, oily skin Unwanted hair, male pattern baldness Insulin excess Diabetes, heart
PCOS and Your Fertility Jim Toner, MD, PhD Atlanta Center for Reproductive Medicine PCOS Consequences Androgen excess Acne, oily skin Unwanted hair, male pattern baldness Insulin excess Diabetes, heart
POLYCYSTIC OVARIAN SYNDROME Laura Tatpati, MD Reproductive Endocrinology and Infertility. Based on: ACOG No. 108 Oct 2009; reaffirmed 2015
 POLYCYSTIC OVARIAN SYNDROME Laura Tatpati, MD Reproductive Endocrinology and Infertility Based on: ACOG No. 108 Oct 2009; reaffirmed 2015 NO DISCLOSURES PATIENT 26 years old presents with complaint of
POLYCYSTIC OVARIAN SYNDROME Laura Tatpati, MD Reproductive Endocrinology and Infertility Based on: ACOG No. 108 Oct 2009; reaffirmed 2015 NO DISCLOSURES PATIENT 26 years old presents with complaint of
Study No.: Title: Rationale: Phase Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Reproductive physiology
 Reproductive physiology Sex hormones: Androgens Estrogens Gestagens Learning objectives 86 (also 90) Sex Genetic sex Gonadal sex Phenotypic sex XY - XX chromosomes testes - ovaries external features Tha
Reproductive physiology Sex hormones: Androgens Estrogens Gestagens Learning objectives 86 (also 90) Sex Genetic sex Gonadal sex Phenotypic sex XY - XX chromosomes testes - ovaries external features Tha
Hormone Balance - Female Report SAMPLE. result graph based on Luteal Phase. result graph based on Luteal Phase
 Patient Name: Patient DOB: Gender: Physician: Test Hormone Balance - Female Report SAMPLE Grote, Mary Jane Batch Number: B6437 2/16/1954 Accession Number: N52281 F Date Received: 2/3/2015 Any Lab Test
Patient Name: Patient DOB: Gender: Physician: Test Hormone Balance - Female Report SAMPLE Grote, Mary Jane Batch Number: B6437 2/16/1954 Accession Number: N52281 F Date Received: 2/3/2015 Any Lab Test
Reproductive outcome in women with body weight disturbances
 Reproductive outcome in women with body weight disturbances Zeev Shoham M.D. Dep. Of OB/GYN Kaplan Hospital, Rehovot, Israel Weight Status BMI (kg/m 2 ) Underweight
Reproductive outcome in women with body weight disturbances Zeev Shoham M.D. Dep. Of OB/GYN Kaplan Hospital, Rehovot, Israel Weight Status BMI (kg/m 2 ) Underweight
Overview of Reproductive Endocrinology
 Overview of Reproductive Endocrinology I have no conflicts of interest to report. Maria Yialamas, MD Female Hypothalamic--Gonadal Axis 15 4 Hormone Secretion in the Normal Menstrual Cycle LH FSH E2, Progesterone,
Overview of Reproductive Endocrinology I have no conflicts of interest to report. Maria Yialamas, MD Female Hypothalamic--Gonadal Axis 15 4 Hormone Secretion in the Normal Menstrual Cycle LH FSH E2, Progesterone,
The Mediterranean Diet: HOW and WHY It Works So Well for T2DM
 The Mediterranean Diet: HOW and WHY It Works So Well for T2DM Susan L. Barlow, RD, CDE. Objectives 1. Discuss the effects of meal size on GLP-1 concentrations. 2. Compare and contrast the specific effects
The Mediterranean Diet: HOW and WHY It Works So Well for T2DM Susan L. Barlow, RD, CDE. Objectives 1. Discuss the effects of meal size on GLP-1 concentrations. 2. Compare and contrast the specific effects
ENDOCRINOLOGY COORDINATION OF PHYSIOLOGICAL PROCESSES:
 ENDOCRINOLOGY COORDINATION OF PHYSIOLOGICAL PROCESSES: -In a living organism there must be coordination of number of physiological activities taking place simultaneously such as: movement, respiration,
ENDOCRINOLOGY COORDINATION OF PHYSIOLOGICAL PROCESSES: -In a living organism there must be coordination of number of physiological activities taking place simultaneously such as: movement, respiration,
Endocrine System. Modified by M. Myers
 Endocrine System Modified by M. Myers 1 The Endocrine System 2 Endocrine Glands The endocrine system is made of glands & tissues that secrete hormones. Hormones are chemicals messengers influencing a.
Endocrine System Modified by M. Myers 1 The Endocrine System 2 Endocrine Glands The endocrine system is made of glands & tissues that secrete hormones. Hormones are chemicals messengers influencing a.
Monday, 7 th of July 2008 ( ) University of Buea MED30. (GENERAL ENDOCRINOLOGY) Exam ( )
 .. Monday, 7 th of July 2008 (8 30-11. 30 ) Faculty of Health Sciences University of Buea MED30 304 Programme in Medicine (GENERAL ENDOCRINOLOGY) Exam (2007-2008).. Multiple Choice Identify the letter
.. Monday, 7 th of July 2008 (8 30-11. 30 ) Faculty of Health Sciences University of Buea MED30 304 Programme in Medicine (GENERAL ENDOCRINOLOGY) Exam (2007-2008).. Multiple Choice Identify the letter
Leptin concentrations in the follicular phase of spontaneous cycles and cycles superovulated with follicle stimulating hormone
 Human Reproduction vol.13 no.5 pp.1152 1156, 1998 Leptin concentrations in the follicular phase of spontaneous cycles and cycles superovulated with follicle stimulating hormone I.E.Messinis 1,4, S.Milingos
Human Reproduction vol.13 no.5 pp.1152 1156, 1998 Leptin concentrations in the follicular phase of spontaneous cycles and cycles superovulated with follicle stimulating hormone I.E.Messinis 1,4, S.Milingos
The Players. Liver Thyroid Adrenals Pancreas Reproductive System Pituitary Gut Bacteria
 The Players Part I Quick Review Understanding some of the key systems and their relationship to hormones is the best place to start It will help with some of the hormone interconnections Key to understanding
The Players Part I Quick Review Understanding some of the key systems and their relationship to hormones is the best place to start It will help with some of the hormone interconnections Key to understanding
The Endocrine System. I. Overview of the Endocrine System. II. Three Families of Hormones. III. Hormone Receptors. IV. Classes of Hormone Receptor
 The Endocrine System I. Overview of the Endocrine System A. Regulates long term metabolic processes B. Releases hormones from endocrine cells 1. Hormones are chemicals 2. Alter metabolism of cells 3. Release
The Endocrine System I. Overview of the Endocrine System A. Regulates long term metabolic processes B. Releases hormones from endocrine cells 1. Hormones are chemicals 2. Alter metabolism of cells 3. Release
ULTIMATE BEAUTY OF BIOCHEMISTRY. Dr. Veena Bhaskar S Gowda Dept of Biochemistry 30 th Nov 2017
 ULTIMATE BEAUTY OF BIOCHEMISTRY Dr. Veena Bhaskar S Gowda Dept of Biochemistry 30 th Nov 2017 SUSPECTED CASE OF CUSHING S SYNDROME Clinical features Moon face Obesity Hypertension Hunch back Abdominal
ULTIMATE BEAUTY OF BIOCHEMISTRY Dr. Veena Bhaskar S Gowda Dept of Biochemistry 30 th Nov 2017 SUSPECTED CASE OF CUSHING S SYNDROME Clinical features Moon face Obesity Hypertension Hunch back Abdominal
Hormones and the Endocrine System Chapter 45. Intercellular communication. Paracrine and Autocrine Signaling. Signaling by local regulators 11/26/2017
 Hormones and the Endocrine System Chapter 45 Intercellular communication Endocrine signaling Local regulators Paracrine and autocrine signaling Neuron signaling Synaptic and neuroendocrine signaling Paracrine
Hormones and the Endocrine System Chapter 45 Intercellular communication Endocrine signaling Local regulators Paracrine and autocrine signaling Neuron signaling Synaptic and neuroendocrine signaling Paracrine
BIOL 2458 A&P II CHAPTER 18 SI Both the system and the endocrine system affect all body cells.
 BIOL 2458 A&P II CHAPTER 18 SI 1 1. Both the system and the endocrine system affect all body cells. 2. Affect on target cells by the system is slow. Affect on target cells by the system is fast. INTERCELLULAR
BIOL 2458 A&P II CHAPTER 18 SI 1 1. Both the system and the endocrine system affect all body cells. 2. Affect on target cells by the system is slow. Affect on target cells by the system is fast. INTERCELLULAR
Endocrine System. Chemical Control
 Endocrine System Chemical Control Endocrine System - the system that secretes hormones in the body - hormones can last for minutes or for hours - a major gland, once called the master gland, is the pituitary
Endocrine System Chemical Control Endocrine System - the system that secretes hormones in the body - hormones can last for minutes or for hours - a major gland, once called the master gland, is the pituitary
Daily blood hormone levels related to the luteinizing hormone surge in anovulatory cycles
 FRTILITY AND STRILITY Copyright 1983 The American Fertility Society Printed in U.8A. Daily blood hormone levels related to the luteinizing hormone surge in anovulatory cycles Chung H. Wu, M.D. * F. Susan
FRTILITY AND STRILITY Copyright 1983 The American Fertility Society Printed in U.8A. Daily blood hormone levels related to the luteinizing hormone surge in anovulatory cycles Chung H. Wu, M.D. * F. Susan
Lab Activity 21. Endocrine System Glucometer. Portland Community College BI 232
 Lab Activity 21 Endocrine System Glucometer Portland Community College BI 232 2 Hormone Functions ACTH (adrenocorticotropic hormone) Regulates the activity of the cortex of the adrenal gland TSH (thyroid
Lab Activity 21 Endocrine System Glucometer Portland Community College BI 232 2 Hormone Functions ACTH (adrenocorticotropic hormone) Regulates the activity of the cortex of the adrenal gland TSH (thyroid
Polycystic ovarian syndrome Can lifestyle modifications help?? Dr Saloni Assistant Professor, Deptt of OBG BPSGMC for Women, Khanpur Kalan
 Polycystic ovarian syndrome Can lifestyle modifications help?? Dr Saloni Assistant Professor, Deptt of OBG BPSGMC for Women, Khanpur Kalan Life-long condition Hirsutism Menstrual irregularities? Pronounced
Polycystic ovarian syndrome Can lifestyle modifications help?? Dr Saloni Assistant Professor, Deptt of OBG BPSGMC for Women, Khanpur Kalan Life-long condition Hirsutism Menstrual irregularities? Pronounced
IMPACT OF NUTRITION ON GYNECOLOGICAL HEALTH OF FEMALE ADOLESCENTS
 IMPACT OF NUTRITION ON GYNECOLOGICAL HEALTH OF FEMALE ADOLESCENTS Kedikova S. MD, PhD Medical University Sofia, Bulgaria University Hospital Maichin dom Sofia NUTRITION WHO - Nutrition is the intake of
IMPACT OF NUTRITION ON GYNECOLOGICAL HEALTH OF FEMALE ADOLESCENTS Kedikova S. MD, PhD Medical University Sofia, Bulgaria University Hospital Maichin dom Sofia NUTRITION WHO - Nutrition is the intake of
Leptin: Amenorrhea, Reproduction, Anorexia. Hazel Leung, Ahrad Nathan, Seja Saddy, Judy Tang
 Leptin: Amenorrhea, Reproduction, Anorexia Hazel Leung, Ahrad Nathan, Seja Saddy, Judy Tang Introduction: What is leptin? Adipocyte-derived hormone (WAT) Receptor roles Class I cytokine receptor superfamily
Leptin: Amenorrhea, Reproduction, Anorexia Hazel Leung, Ahrad Nathan, Seja Saddy, Judy Tang Introduction: What is leptin? Adipocyte-derived hormone (WAT) Receptor roles Class I cytokine receptor superfamily
Achieving Pregnancy: Obesity and Infertility. Jordan Vaughan, MSN, APN, WHNP-BC Women s Health Nurse Practitioner Nashville Fertility Center
 Achieving Pregnancy: Obesity and Infertility Jordan Vaughan, MSN, APN, WHNP-BC Women s Health Nurse Practitioner Nashville Fertility Center Disclosures Speakers Bureau EMD Serono Board of Directors Nurse
Achieving Pregnancy: Obesity and Infertility Jordan Vaughan, MSN, APN, WHNP-BC Women s Health Nurse Practitioner Nashville Fertility Center Disclosures Speakers Bureau EMD Serono Board of Directors Nurse
Food Intake Regulation & the Clock. Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD
 Food Intake Regulation & the Clock Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD Circadian disruption affect multiple organ systems: The diagram provides examples of how circadian disruption
Food Intake Regulation & the Clock Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD Circadian disruption affect multiple organ systems: The diagram provides examples of how circadian disruption
Reproductive Health and Pituitary Disease
 Reproductive Health and Pituitary Disease Janet F. McLaren, MD Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics and Gynecology jmclaren@uabmc.edu Objectives
Reproductive Health and Pituitary Disease Janet F. McLaren, MD Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics and Gynecology jmclaren@uabmc.edu Objectives
Bone Metabolism in Postmenopausal Women Influenced by the Metabolic Syndrome
 Bone Metabolism in Postmenopausal Women Influenced by the Metabolic Syndrome Thomas et al. Nutrition Journal (2015) 14:99 DOI 10.1186/s12937-015-0092-2 RESEARCH Open Access Acute effect of a supplemented
Bone Metabolism in Postmenopausal Women Influenced by the Metabolic Syndrome Thomas et al. Nutrition Journal (2015) 14:99 DOI 10.1186/s12937-015-0092-2 RESEARCH Open Access Acute effect of a supplemented
Laura Stewart, MD, FRCPC Clinical Associate Professor Division of Pediatric Endocrinology University of British Columbia
 Precocious Puberty Laura Stewart, MD, FRCPC Clinical Associate Professor Division of Pediatric Endocrinology University of British Columbia Faculty Disclosure Faculty: Laura Stewart No relationships with
Precocious Puberty Laura Stewart, MD, FRCPC Clinical Associate Professor Division of Pediatric Endocrinology University of British Columbia Faculty Disclosure Faculty: Laura Stewart No relationships with
Comprehensive assessments of neuroendocrine status. Salivary Sex Steroid Hormones and Adrenal/HPA Axis Function
 ENDOCRINOLOGY Comprehensive assessments of neuroendocrine status Salivary Sex Steroid Hormones and Adrenal/HPA Axis Function Neuro-Biogenic Amine and Metabolite Profiles (Neurotransmitters) Metabolomic
ENDOCRINOLOGY Comprehensive assessments of neuroendocrine status Salivary Sex Steroid Hormones and Adrenal/HPA Axis Function Neuro-Biogenic Amine and Metabolite Profiles (Neurotransmitters) Metabolomic
Rhythm Plus- Comprehensive Female Hormone Profile
 Rhythm Plus- Comprehensive Female Hormone Profile Patient: SAMPLE REPORT DOB: Sex: F Order Number: K00000 Completed: Received: Collected: SAMPLE REPORT Sample # Progesterone (pg/ml) Hormone Results Oestradiol
Rhythm Plus- Comprehensive Female Hormone Profile Patient: SAMPLE REPORT DOB: Sex: F Order Number: K00000 Completed: Received: Collected: SAMPLE REPORT Sample # Progesterone (pg/ml) Hormone Results Oestradiol
Growth Hormone, Somatostatin, and Prolactin 1 & 2 Mohammed Y. Kalimi, Ph.D.
 Growth Hormone, Somatostatin, and Prolactin 1 & 2 Mohammed Y. Kalimi, Ph.D. I. Growth Hormone (somatotropin): Growth hormone (GH) is a 191 amino acid single chain polypeptide (MW 22,000 daltons). Growth
Growth Hormone, Somatostatin, and Prolactin 1 & 2 Mohammed Y. Kalimi, Ph.D. I. Growth Hormone (somatotropin): Growth hormone (GH) is a 191 amino acid single chain polypeptide (MW 22,000 daltons). Growth
NIH Public Access Author Manuscript Eat Weight Disord. Author manuscript; available in PMC 2012 January 23.
 NIH Public Access Author Manuscript Published in final edited form as: Eat Weight Disord. 2008 December ; 13(4): e96 e99. Night eating status and influence on body weight, body image, hunger, and cortisol
NIH Public Access Author Manuscript Published in final edited form as: Eat Weight Disord. 2008 December ; 13(4): e96 e99. Night eating status and influence on body weight, body image, hunger, and cortisol
Chapter 11. Endocrine System
 Chapter 11 Endocrine System 1 Introduction A. The endocrine system is made up of the cells, tissues, and organs that secrete hormones into body fluids. B. Hormones diffuse into the bloodstream to act target
Chapter 11 Endocrine System 1 Introduction A. The endocrine system is made up of the cells, tissues, and organs that secrete hormones into body fluids. B. Hormones diffuse into the bloodstream to act target
To test your basal rates it will be important that you keep the same schedule on the days you are testing. Do not do basal rate testing if you:
 Basal Rate Testing: Your provider has recommended that you complete one or more basal rate tests. These worksheets will help you in testing your pump settings and making small changes to make sure your
Basal Rate Testing: Your provider has recommended that you complete one or more basal rate tests. These worksheets will help you in testing your pump settings and making small changes to make sure your
Targeted Nutrition Therapy Nutrition Masters Course
 Targeted Nutrition Therapy Nutrition Masters Course Nilima Desai, MPH, RD Learning Objectives Review clinical studies on innovative, targeted nutrition therapies for: o Blood glucose management o Dyslipidemia
Targeted Nutrition Therapy Nutrition Masters Course Nilima Desai, MPH, RD Learning Objectives Review clinical studies on innovative, targeted nutrition therapies for: o Blood glucose management o Dyslipidemia
Test Results SB Samples Arrived: 01/15/2014 Samples Collected: Saliva: 01/11/14 07:14 Date Closed: 01/18/2014
 Test Results 8605 SW Creekside Place Beaverton, OR 97008 Phone: 503-466-2445 Fax: 503-466-1636 info@zrtlab.com www.zrtlab.com 2014 01 15 001 SB Samples Arrived: 01/15/2014 Samples Collected: Saliva: 01/11/14
Test Results 8605 SW Creekside Place Beaverton, OR 97008 Phone: 503-466-2445 Fax: 503-466-1636 info@zrtlab.com www.zrtlab.com 2014 01 15 001 SB Samples Arrived: 01/15/2014 Samples Collected: Saliva: 01/11/14
Diabetes: Definition Pathophysiology Treatment Goals. By Scott Magee, MD, FACE
 Diabetes: Definition Pathophysiology Treatment Goals By Scott Magee, MD, FACE Disclosures No disclosures to report Definition of Diabetes Mellitus Diabetes Mellitus comprises a group of disorders characterized
Diabetes: Definition Pathophysiology Treatment Goals By Scott Magee, MD, FACE Disclosures No disclosures to report Definition of Diabetes Mellitus Diabetes Mellitus comprises a group of disorders characterized
Polycystic Ovarian Syndrome (PCOS) LOGO
 Polycystic Ovarian Syndrome (PCOS) Ma qianhong Ob/Gyn Department LOGO Contents Epidemiology and Definition Pathophysiology, Endocrinological Features Diagnostic Criteria Treatment Prognosis Introduction
Polycystic Ovarian Syndrome (PCOS) Ma qianhong Ob/Gyn Department LOGO Contents Epidemiology and Definition Pathophysiology, Endocrinological Features Diagnostic Criteria Treatment Prognosis Introduction
Underlying causes of obesity include a host of various genetic, environmental, and psychological factors.
 Chapter 15 Weight Management Chapter 15 Lesson 15.1 Key Concepts Underlying causes of obesity include a host of various genetic, environmental, and psychological factors. Realistic weight management focuses
Chapter 15 Weight Management Chapter 15 Lesson 15.1 Key Concepts Underlying causes of obesity include a host of various genetic, environmental, and psychological factors. Realistic weight management focuses
Diabetes: Across the Lifespan Friday, October 17, Obesity, Insulin Resistance and Type 2 Diabetes Cardiovascular Risks in Children.
 Diabetes: Across the Lifespan Friday, October 17, 2014 Obesity, Insulin Resistance and Type 2 Diabetes Cardiovascular Risks in Children. Don P. Wilson, M.D., FNLA Diplomate, Am Brd of Clinical Lipidology
Diabetes: Across the Lifespan Friday, October 17, 2014 Obesity, Insulin Resistance and Type 2 Diabetes Cardiovascular Risks in Children. Don P. Wilson, M.D., FNLA Diplomate, Am Brd of Clinical Lipidology
Your Reported Urinary Free Cortisone Pattern
 P: 1300 688 522 E: info@nutripath.com.au A: PO Box 442 Ashburton VIC 3142 TEST PATIENT Sex : D Collected : 00-00-0000 111 ROAD TEST SUBURB AB : 00000000 UR#:0000000 TEST PHYSICIAN DR JOHN DOE 111 CLINIC
P: 1300 688 522 E: info@nutripath.com.au A: PO Box 442 Ashburton VIC 3142 TEST PATIENT Sex : D Collected : 00-00-0000 111 ROAD TEST SUBURB AB : 00000000 UR#:0000000 TEST PHYSICIAN DR JOHN DOE 111 CLINIC
PCOS Awareness Symposium Atlanta September 24 th, Preventing Diabetes & Cardiovascular Disease in PCOS
 PCOS Awareness Symposium Atlanta September 24 th, 2016 Preventing Diabetes & Cardiovascular Disease in PCOS Katherine Sherif, MD Professor & Vice Chair, Department of Medicine Director, Jefferson Women
PCOS Awareness Symposium Atlanta September 24 th, 2016 Preventing Diabetes & Cardiovascular Disease in PCOS Katherine Sherif, MD Professor & Vice Chair, Department of Medicine Director, Jefferson Women
WEIGHT CHANGE AND ANDROGEN LEVELS DURING CONTRACEPTIVE TREATMENT OF WOMEN AFFECTED BY POLYCYSTIC OVARY
 ENDOCRINE REGULATIONS, VOL. 40, 119-123, 2006 119 WEIGHT CHANGE AND ANDROGEN LEVELS DURING CONTRACEPTIVE TREATMENT OF WOMEN AFFECTED BY POLYCYSTIC OVARY J. VRBIKOVA, K. DVORAKOVA, M. HILL, L. STARKA Institute
ENDOCRINE REGULATIONS, VOL. 40, 119-123, 2006 119 WEIGHT CHANGE AND ANDROGEN LEVELS DURING CONTRACEPTIVE TREATMENT OF WOMEN AFFECTED BY POLYCYSTIC OVARY J. VRBIKOVA, K. DVORAKOVA, M. HILL, L. STARKA Institute
Anatomy and Physiology. The Endocrine System
 Anatomy and Physiology The Endocrine System The endocrine system includes anything that secretes hormones directly into body fluids. Endocrine glands include: the thyroid, parathyroid, adrenal, kidney,
Anatomy and Physiology The Endocrine System The endocrine system includes anything that secretes hormones directly into body fluids. Endocrine glands include: the thyroid, parathyroid, adrenal, kidney,
Chapter 11 - Endocrine System
 Chapter 11 - Endocrine System 11.1 Introduction A. The endocrine system is made up of the cells, tissues, and organs that secrete hormones into body fluids. B. The body has two kinds of glands, exocrine
Chapter 11 - Endocrine System 11.1 Introduction A. The endocrine system is made up of the cells, tissues, and organs that secrete hormones into body fluids. B. The body has two kinds of glands, exocrine
Chapter 26. Hormones and the Endocrine System. Lecture by Edward J. Zalisko
 Chapter 26 Hormones and the Endocrine System PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Copyright 2009 Pearson Education, Inc. Lecture
Chapter 26 Hormones and the Endocrine System PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Copyright 2009 Pearson Education, Inc. Lecture
The Impact of Insulin Resistance on Long-Term Health in PCOS
 Saturday, April 16 th, 2016 PCOS Challenge & Thomas Jefferson University PCOS Awareness Symposium Philadelphia The Impact of Insulin Resistance on Long-Term Health in PCOS Katherine Sherif, MD Professor
Saturday, April 16 th, 2016 PCOS Challenge & Thomas Jefferson University PCOS Awareness Symposium Philadelphia The Impact of Insulin Resistance on Long-Term Health in PCOS Katherine Sherif, MD Professor
Chapter 20. Endocrine System Chemical signals coordinate body functions Chemical signals coordinate body functions. !
 26.1 Chemical signals coordinate body functions Chapter 20 Endocrine System! Hormones Chemical signals Secreted by endocrine glands Usually carried in the blood Cause specific changes in target cells Secretory
26.1 Chemical signals coordinate body functions Chapter 20 Endocrine System! Hormones Chemical signals Secreted by endocrine glands Usually carried in the blood Cause specific changes in target cells Secretory
12/13/2017. Important references for PCOS. Polycystic Ovarian Syndrome (PCOS) for the Family Physician. 35 year old obese woman
 Polycystic Ovarian Syndrome (PCOS) for the Family Physician Barbara S. Apgar MD, MS Professor or Family Medicine University of Michigan Ann Arbor, Michigan Important references for PCOS Endocrine Society
Polycystic Ovarian Syndrome (PCOS) for the Family Physician Barbara S. Apgar MD, MS Professor or Family Medicine University of Michigan Ann Arbor, Michigan Important references for PCOS Endocrine Society
Corporate Medical Policy Testosterone Pellet Implantation for Androgen Deficiency
 Corporate Medical Policy Testosterone Pellet Implantation for Androgen Deficiency File Name: Origination: Last CAP Review: Next CAP Review: Last Review: testosterone_pellet_implantation_for_androgen_deficiency
Corporate Medical Policy Testosterone Pellet Implantation for Androgen Deficiency File Name: Origination: Last CAP Review: Next CAP Review: Last Review: testosterone_pellet_implantation_for_androgen_deficiency
Sleep and Weight Control
 A Wake-up Call Charles H. Samuels, MD, CCFP, DABSM As presented at the Sleep and Respiration Rounds held at the Lung Association Sleep Centre at Foothills Medical Centre, Calgary, Alberta (May 2005) Jill
A Wake-up Call Charles H. Samuels, MD, CCFP, DABSM As presented at the Sleep and Respiration Rounds held at the Lung Association Sleep Centre at Foothills Medical Centre, Calgary, Alberta (May 2005) Jill
CREATING A PCOS TREATMENT PLAN. Ricardo Azziz, M.D., M.P.H., M.B.A. Georgia Regents University
 CREATING A PCOS TREATMENT PLAN Ricardo Azziz, M.D., M.P.H., M.B.A. Georgia Regents University PCOS: CREATING A TREATMENT PLAN Good treatment plans are based on sound and complete evaluations History of
CREATING A PCOS TREATMENT PLAN Ricardo Azziz, M.D., M.P.H., M.B.A. Georgia Regents University PCOS: CREATING A TREATMENT PLAN Good treatment plans are based on sound and complete evaluations History of
Salivary Hormone Testing
 Salivary Hormone Testing Oral Fluids Mirror Of The Body Fluid levels of molecules can be use for therapeutic, drug dependency or hormones etc. purposes Emotional status elements connected to depression
Salivary Hormone Testing Oral Fluids Mirror Of The Body Fluid levels of molecules can be use for therapeutic, drug dependency or hormones etc. purposes Emotional status elements connected to depression
I. Endocrine System & Hormones Figure 1: Human Endocrine System
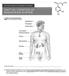 I. Endocrine System & Hormones Figure 1: Human Endocrine System Endocrine System: a) Endocrine glands are ductless since they lack specific vessels for the transport of hormones throughout the body. Instead,
I. Endocrine System & Hormones Figure 1: Human Endocrine System Endocrine System: a) Endocrine glands are ductless since they lack specific vessels for the transport of hormones throughout the body. Instead,
Reproductive cyclicity 19. Introduction. Page 1. repro and its story lines. Male repro: a simpler way of control
 Reproductive cyclicity 19 Male repro: a simpler way of control Menstrual cycles: ovary / uterine anatomy and cell types, follicular phase, ovulation, luteal phase, cyclicity Race events: removal of P4
Reproductive cyclicity 19 Male repro: a simpler way of control Menstrual cycles: ovary / uterine anatomy and cell types, follicular phase, ovulation, luteal phase, cyclicity Race events: removal of P4
Therapeutic Cohort Results
 Patient: PAGE LOVE DOB: January 11, 1983 Sex: F MRN: 1232704193 Order Number: J9020008 Completed: July 08, 2016 Received: July 02, 2016 Collected: July 01, 2016 Aum Healing Center Sarika Arora MD 332 Newbury
Patient: PAGE LOVE DOB: January 11, 1983 Sex: F MRN: 1232704193 Order Number: J9020008 Completed: July 08, 2016 Received: July 02, 2016 Collected: July 01, 2016 Aum Healing Center Sarika Arora MD 332 Newbury
The role of proteins and amino acids in food intake and satiety
 The role of proteins and amino acids in food intake and satiety Daniel TOME AGROPARISTECH Departement of Life Sciences and Health INRA, UMR914 Nutrition physiology and ingestive behavior, Paris, France
The role of proteins and amino acids in food intake and satiety Daniel TOME AGROPARISTECH Departement of Life Sciences and Health INRA, UMR914 Nutrition physiology and ingestive behavior, Paris, France
TEST REPORT # U. Patient Name: Sleep Balance Patient Phone Number: TEST NAME RESULTS 07/30/18 RANGE
 TEST REPORT Ordering Provider: John Doe, ND 8605 SW Creekside Place Beaverton, OR 97008 Phone: 503-466-2445 Fax: 503-466-1636 Samples Received 08/06/18 Report Date 08/10/18 Samples Collected Urine - 07/30/18
TEST REPORT Ordering Provider: John Doe, ND 8605 SW Creekside Place Beaverton, OR 97008 Phone: 503-466-2445 Fax: 503-466-1636 Samples Received 08/06/18 Report Date 08/10/18 Samples Collected Urine - 07/30/18
Diagnos-Techs, Inc. Clinical & Research Laboratory th Ave S. Bldg. G, Kent WA Tel: (425) CLIA License # 50D
 Accession # Diagnos-Techs, Inc. Clinical & Research Laboratory 191 66th Ave S. Bldg. G, Kent WA 9823 Tel: (4) 1-96 CLIA License # D6141 Received : 11//213 Completed: 11/8/213 Reported : 11/8/213 BOULDER
Accession # Diagnos-Techs, Inc. Clinical & Research Laboratory 191 66th Ave S. Bldg. G, Kent WA 9823 Tel: (4) 1-96 CLIA License # D6141 Received : 11//213 Completed: 11/8/213 Reported : 11/8/213 BOULDER
Therapeutic Cohort Results
 Patient: SAMPLE PATIENT DOB: Sex: MRN: Menopause Plus - Salivary Profile Therapeutic Cohort Results Hormone Average Result QUINTILE DISTRIBUTION 1st 2nd 3rd 4th 5th Therapeutic Range* Estradiol (E2) 8.7
Patient: SAMPLE PATIENT DOB: Sex: MRN: Menopause Plus - Salivary Profile Therapeutic Cohort Results Hormone Average Result QUINTILE DISTRIBUTION 1st 2nd 3rd 4th 5th Therapeutic Range* Estradiol (E2) 8.7
Human Biochemistry. Hormones
 Human Biochemistry Hormones THE ENDOCRINE SYSTEM THE ENDOCRINE SYSTEM THE ENDOCRINE SYSTEM The ENDOCRINE SYSTEM = the organ system that regulates internal environment conditions by secreting hormones into
Human Biochemistry Hormones THE ENDOCRINE SYSTEM THE ENDOCRINE SYSTEM THE ENDOCRINE SYSTEM The ENDOCRINE SYSTEM = the organ system that regulates internal environment conditions by secreting hormones into
Investigation of adrenal functions in patients with idiopathic hyperandrogenemia
 European Journal of Endocrinology (26) 155 37 311 ISSN 84-4643 CLINICAL STUDY Investigation of adrenal functions in patients with idiopathic hyperandrogenemia Hulusi Atmaca, Fatih Tanriverdi 1, Kursad
European Journal of Endocrinology (26) 155 37 311 ISSN 84-4643 CLINICAL STUDY Investigation of adrenal functions in patients with idiopathic hyperandrogenemia Hulusi Atmaca, Fatih Tanriverdi 1, Kursad
Adrenal Glands. Adrenal Glands. Adrenal Glands. Adrenal Glands. Adrenal Glands 4/12/2016. Controlled by both nerves and hormones.
 Glands http://www.hawaiilife.com/articles/2012/03/good-news-vacation-rental-owners/ 70 Figure 10.14a gland Glands cortex Mineralocorticoids Gonadocorticoids Glucocorticoids medulla Epinephrine Norepinephrine
Glands http://www.hawaiilife.com/articles/2012/03/good-news-vacation-rental-owners/ 70 Figure 10.14a gland Glands cortex Mineralocorticoids Gonadocorticoids Glucocorticoids medulla Epinephrine Norepinephrine
Adrenal Stress Profile (Saliva)
 Adrenal Stress Profile (Saliva) Ireland Cortisol Levels Sample 1 Post Awakening Sample 2 (+ 4-5 Hours) Sample 3 (+ 4-5 Hours) Sample 4 (Prior to Sleep) 11.42 2.10 1.60 0.47 Sum of Cortisol 15.590 DHEA
Adrenal Stress Profile (Saliva) Ireland Cortisol Levels Sample 1 Post Awakening Sample 2 (+ 4-5 Hours) Sample 3 (+ 4-5 Hours) Sample 4 (Prior to Sleep) 11.42 2.10 1.60 0.47 Sum of Cortisol 15.590 DHEA
4/23/2013. Environmental Agents. Foods Ingested. Stress. Genetics. Bodily Functions. Diet and Supplement Regimen
 1 Exercise, and Preventative Medical Practices Diet and Supplement Regimen Sleep, Relaxation, Social and Emotional Health "Systems biology...is about putting together rather than taking apart, integration
1 Exercise, and Preventative Medical Practices Diet and Supplement Regimen Sleep, Relaxation, Social and Emotional Health "Systems biology...is about putting together rather than taking apart, integration
Objectives 10/11/2013. Diabetes- The Real Cost of Sugar. Diabetes 101: What is Diabetes. By Ruth Nekonchuk RD CDE LMNT
 Diabetes- The Real Cost of Sugar By Ruth Nekonchuk RD CDE LMNT Objectives To explain diabetes To explain the risks of diabetes To enumerate the cost of diabetes to our country To enumerate the cost of
Diabetes- The Real Cost of Sugar By Ruth Nekonchuk RD CDE LMNT Objectives To explain diabetes To explain the risks of diabetes To enumerate the cost of diabetes to our country To enumerate the cost of
The endocrine system is complex and sometimes poorly understood.
 1 CE Credit Testing the Endocrine System for Adrenal Disorders and Diabetes Mellitus: It Is All About Signaling Hormones! David Liss, BA, RVT, VTS (ECC) Platt College Alhambra, California For more information,
1 CE Credit Testing the Endocrine System for Adrenal Disorders and Diabetes Mellitus: It Is All About Signaling Hormones! David Liss, BA, RVT, VTS (ECC) Platt College Alhambra, California For more information,
Hormones. Introduction to Endocrine Disorders. Hormone actions. Modulation of hormone levels. Modulation of hormone levels
 Introduction to Endocrine Disorders Hormones Self-regulating system (homeostasis) Affect: Growth Metabolism Reproduction Fluid and electrolyte balance Hormone actions Endocrine gland Hormone synthesis
Introduction to Endocrine Disorders Hormones Self-regulating system (homeostasis) Affect: Growth Metabolism Reproduction Fluid and electrolyte balance Hormone actions Endocrine gland Hormone synthesis
Short-term Satiety of High Protein Formula on Obese Subjects: A Pilot Study
 2012 International Conference on Nutrition and Food Sciences IPCBEE vol. 39 (2012) (2012) IACSIT Press, Singapore Short-term Satiety of High Protein Formula on Obese Subjects: A Pilot Study Kamalita Pertiwi
2012 International Conference on Nutrition and Food Sciences IPCBEE vol. 39 (2012) (2012) IACSIT Press, Singapore Short-term Satiety of High Protein Formula on Obese Subjects: A Pilot Study Kamalita Pertiwi
2.0 Synopsis. Lupron Depot M Clinical Study Report R&D/09/093. (For National Authority Use Only) to Part of Dossier: Name of Study Drug:
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: Name of Study Drug: Volume: Abbott-43818 (ABT-818) leuprolide acetate for depot suspension (Lupron Depot ) Name of
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: Name of Study Drug: Volume: Abbott-43818 (ABT-818) leuprolide acetate for depot suspension (Lupron Depot ) Name of
C h a p t e r 3 8 Cushing s Syndrome : Current Concepts in Diagnosis and Management
 C h a p t e r 3 8 Cushing s Syndrome : Current Concepts in Diagnosis and Management Padma S Menon Professor of Endocrinology, Seth G S Medical College & KEM Hospital, Mumbai A clinical syndrome resulting
C h a p t e r 3 8 Cushing s Syndrome : Current Concepts in Diagnosis and Management Padma S Menon Professor of Endocrinology, Seth G S Medical College & KEM Hospital, Mumbai A clinical syndrome resulting
N. Shirazian, MD. Endocrinologist
 N. Shirazian, MD Internist, Endocrinologist Inside the ovary Day 15-28: empty pyfollicle turns into corpus luteum (yellow body) Immature eggs Day 1-13: 13: egg developing inside the growing follicle Day
N. Shirazian, MD Internist, Endocrinologist Inside the ovary Day 15-28: empty pyfollicle turns into corpus luteum (yellow body) Immature eggs Day 1-13: 13: egg developing inside the growing follicle Day
METABOLIC SYNDROME IN REPRODUCTIVE FEMALES
 METABOLIC SYNDROME IN REPRODUCTIVE FEMALES John J. Orris, D.O., M.B.A Division Head, Reproductive Endocrinology & Infertility, Main Line Health System Associate Professor, Drexel University College of
METABOLIC SYNDROME IN REPRODUCTIVE FEMALES John J. Orris, D.O., M.B.A Division Head, Reproductive Endocrinology & Infertility, Main Line Health System Associate Professor, Drexel University College of
Evaluation and Management of Pituitary Failure. Dr S. Ali Imran MBBS, FRCP (Edin), FRCPC Professor of Medicine Dalhousie University, Halifax, NS
 Evaluation and Management of Pituitary Failure Dr S. Ali Imran MBBS, FRCP (Edin), FRCPC Professor of Medicine Dalhousie University, Halifax, NS Conflict of Interest None Objectives Diagnostic approach
Evaluation and Management of Pituitary Failure Dr S. Ali Imran MBBS, FRCP (Edin), FRCPC Professor of Medicine Dalhousie University, Halifax, NS Conflict of Interest None Objectives Diagnostic approach
