Deep Supervision for Pancreatic Cyst Segmentation in Abdominal CT Scans
|
|
|
- Ronald Floyd
- 6 years ago
- Views:
Transcription
1 Deep Supervision for Pancreatic Cyst Segmentation in Abdominal CT Scans Yuyin Zhou 1, Lingxi Xie 2( ), Elliot K. Fishman 3, Alan L. Yuille 4 1,2,4 The Johns Hopkins University, Baltimore, MD 21218, USA 3 The Johns Hopkins University School of Medicine, Baltimore, MD 21287, USA 1 zhouyuyiner@gmail.com xc@gmail.com 3 efishman@jhmi.edu 4 alan.l.yuille@gmail.com Abstract. Automatic segmentation of an organ and its cystic region is a prerequisite of computer-aided diagnosis. In this paper, we focus on pancreatic cyst segmentation in abdominal CT scan. This task is important and very useful in clinical practice yet challenging due to the low contrast in boundary, the variability in location, shape and the different stages of the pancreatic cancer. Inspired by the high relevance between the location of a pancreas and its cystic region, we introduce extra deep supervision into the segmentation network, so that cyst segmentation can be improved with the help of relatively easier pancreas segmentation. Under a reasonable transformation function, our approach can be factorized into two stages, and each stage can be efficiently optimized via gradient back-propagation throughout the deep networks. We collect a new dataset with 131 pathological samples, which, to the best of our knowledge, is the largest set for pancreatic cyst segmentation. Without human assistance, our approach reports a 63.44% average accuracy, measured by the Dice-Sørensen coefficient (DSC), which is higher than the number (60.46%) without deep supervision. 1 Introduction In 2012, pancreatic cancers of all types were the 7th most common cause of cancer deaths, resulting in 330,000 deaths globally [15]. By the time of diagnosis, pancreatic cancer has often spread to other parts of the body. Therefore, it is very important to use medical imaging analysis to assist identifying malignant cysts in the early stages of pancreatic cancer to increase the survival chance of a patient [3]. The emerge of deep learning has largely advanced the field of computer-aided diagnosis (CAD). With the help of the state-of-the-art deep convolutional neural networks [7][14], such as the fully-convolutional networks (FCN) [10] for semantic segmentation, researchers have achieved accurate segmentation on many abdominal organs. There are often different frameworks for segmenting different organs [1][13]. Meanwhile, it is of great interest to find the lesion area in an organ [16][2][5], which, frequently, is even more challenging due to the tiny volume and variable properties of these parts.
2 2 Y. Zhou et al. This paper focuses on segmenting pancreatic cyst from abdominal CT scan. Pancreas is one of the abdominal organs that are very difficult to be segmented even in the healthy cases [13][12][17], mainly due to the low contrast in the boundary and the high variability in its geometric properties. In the pathological cases, the difference in the pancreatic cancer stage also impacts both the morphology of the pancreas and the cyst [4][8]. Despite the importance of pancreatic cyst segmentation, this topic is less studied: some of the existing methods are based on old-fashioned models [6], and a state-of-the-art approach [3] requires a bounding box of the cyst to be annotated beforehand, as well as a lot of interactive operations throughout the segmentation process to annotate some voxels on or off the target. These requirements are often unpractical when the user is not well knowledgable in medicine (e.g., a common patient). This paper presents the first system which produces reasonable pancreatic cyst segmentation without human assistance on the testing stage. Intuitively, the pancreatic cyst is often closely related to the pancreas, and thus segmenting the pancreas (relatively easier) may assist the localization and segmentation of the cyst. To this end, we introduce deep supervision [9] into the original segmentation network, leading to a joint objective function taking both the pancreas and the cyst into consideration. Using a reasonable transformation function, the optimization process can be factorized into two stages, in which we first find the pancreas, and then localize and segment the cyst based on the predicted pancreas mask. Our approach works efficiently based on a recent published coarse-to-fine segmentation approach [17]. We perform experiments on a newly collected dataset with 131 pathological samples from CT scan. Without human assistance on the testing stage, our approach achieves an average Dice- Sørensen coefficient (DSC) of 63.44%, which is practical for clinical applications. 2 Approach 2.1 Formulation Let a CT-scanned image be a 3D volume X. Each volume is annotated with ground-truth pancreas segmentation P and cyst segmentation C, and both of them are of the same dimensionality as X. P i = 1 and C i = 1 indicate a foreground voxel of pancreas and cyst, respectively. Denote a cyst segmentation model as M : C = f(x; Θ), where Θ denotes the model parameters. The loss function can be written as L(C, C ). In a regular deep neural network such as our baseline, the fully-convolutional network (FCN) [10], we optimize L with respect to the network weights Θ via gradient back-propagation. To deal with small targets, we follow [11] to compute the DSC loss function: L(C, C ) = 2 i CiC i i Ci+ i C i can be easily computed. Pancreas is a small organ in human body, which typically occupies less than 1% voxels in a CT volume. In comparison, the pancreatic cyst is even smaller. In our newly collected dataset, the fraction of the cyst, relative to the entire volume, is often much smaller than 0.1%. In a very challenging case, the cyst. The gradient L(C,C ) C
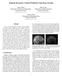
3 Deep Supervision for Pancreatic Cyst Segmentation in Abdominal CT Scans 3 Input Image Global Segmentation Local Segmentation Case #123 DSC = 0.00% DSC = 85.21% Fig. 1. A relatively difficult case in pancreatic cyst segmentation and the results produced by different input regions, namely using the entire image and the region around the ground-truth pancreas mask (best viewed in color). The cystic, predicted and overlapping regions are marked by red, green and yellow, respectively. For better visualization, the right two figures are zoomed in w.r.t. the red frame. only occupies % of the volume, or around 1.5% of the pancreas. This largely increases the difficulty of segmentation or even localization. Figure 1 shows a representative example where cyst segmentation fails completely when we take the entire 2D slice as the input. To deal with this problem, we note that the location of the pancreatic cyst is highly relevant to the pancreas. Denote the set of voxels of the pancreas as P = {i P i = 1}, and similarly, the set of cyst voxels as C = {i C i = 1}. Frequently, a large fraction of C falls within P (e.g., P C / C > 95% in 121 out of 131 cases in our dataset). Starting from the pancreas mask increases the chance of accurately segmenting the cyst. Figure 1 shows an example of using the ground-truth pancreas mask to recover the failure case of cyst segmentation. This inspires us to perform cyst segmentation based on the pancreas region, which is relatively easy to detect. To this end, we introduce the pancreas mask P as an explicit variable of our approach, and append another term to the loss function to jointly optimize both pancreas and cyst segmentation networks. Mathematically, let the pancreas segmentation model be M P : P = f P (X; Θ P ), and the corresponding loss term be L P (P, P ). Based on P, we create a smaller input region by applying a transformation X = σ[x, P], and feed X to the next stage. Thus, the cyst segmentation model can be written as M C : C = f C (X ; Θ C ), and we have the corresponding loss them L C (C, C ). To optimize both Θ P and Θ C, we consider the following loss function: L(P, P, C, C ) = λl P (P, P ) + (1 λ) L C (C, C ), (1) where λ is the balancing parameter defining the weight between either terms. 2.2 Optimization We use gradient descent for optimization, which involves computing the gradients over Θ P and Θ C. Among these, = LC Θ C, and thus we can compute it via L Θ C
4 4 Y. Zhou et al. training only training / testing Pancreas Ground-Truth Pancreas Loss Function L P P, P Cyst Ground-Truth Cyst Loss Function L C C, C Deep Net for Pancreas Segmentation X = σ X, P Deep Net for Cyst Segmentation Input Volume for Pancreas Segmentation Output Volume for Pancreas Segmentation Input Volume for Cyst Segmentation Output Volume for Cyst Segmentation Fig. 2. The framework of our approach (best viewed in color). Two deep segmentation networks are stacked, and two loss functions are computed. The predicted pancreas mask is used in transforming the input image for cyst segmentation. standard back-propagation in a deep neural network. On the other hand, Θ P is involved in both loss terms, and applying the chain rule yields: L = L P + L C Θ P Θ P X X P P. (2) Θ P The second term on the right-hand side depends on the definition of X = σ[x, P]. In practice, we define a simple transformation to simplify the computation. The intensity value (directly related to the Hounsfield units in CT scan) of each voxel is either preserved or set as 0, and the criterion is whether there exists a nearby voxel which is likely to fall within the pancreas region: X i = X i I{ j P j > 0.5 i j < t}, (3) where t is the threshold which is the farthest distance from a cyst voxel to the pancreas volume. We set t = 15 in practice, and our approach is not sensitive to this parameter. With this formulation, i.e., X i P j = 0 almost everywhere. Thus, we have X L P = 0 and Θ P = LP Θ P. This allows us to factorize the optimization into two stages in both training and testing. Since L Θ P and L Θ C are individually optimized, the balancing parameter λ in Eqn (1) can be ignored. The overall framework is illustrated in Figure 2. In training, we directly set X = σ[x, P ], so that the cyst segmentation model M C receives more reliable supervision. In testing, starting from X, we compute P, X and C orderly. Dealing with two stages individually reduces the computational overheads. It is also possible to formulate the second stage as multi-label segmentation. The implementation details follow our recent work [17], which achieves the state-of-the-art performance in the NIH pancreas segmentation dataset [13]. Due to the limited amount of training data, instead of applying 3D networks directly, we cut each 3D volume into a series of 2D pieces, and feed them into a fully-convolutional network (FCN) [10]. This operation is performed along three directions, namely the coronal, sagittal and axial views. At the testing stage, the
5 Deep Supervision for Pancreatic Cyst Segmentation in Abdominal CT Scans 5 cues from three views are fused, and each voxel is considered to fall on foreground if at least two views predict so. In the pathological dataset, we observe a large variance in the morphology of both pancreas and cyst. which increases the difficulty for the deep network to converge in the training process. Consequently, using one single model may result in less stable segmentation results. In practice, we use the FCN-8s model [10] from the pre-trained weights on the PascalVOC dataset. This model is based on a 16-layer VGGNet [14], and we believe a deeper network may lead to better results. We fine-tune it through 60K iterations with a learning rate of Nine models, namely the snapshots after {20K, 25K,..., 60K} iterations, are used to test each volume, and the final result is obtained by computing the union of nine predicted foreground voxel sets. Regarding other technical details, we simply follow [17], including using the DSC loss layer instead of the voxel-wise loss layer to prevent the bias towards background [11], and applying a flexible convergence condition for fine-tuning at the testing stage. 3 Experiments 3.1 Dataset and Evaluation We evaluate our approach on a dataset collected by the radiologists in our team. This dataset contains 131 contrast-enhanced abdominal CT volumes, and each of them is manually labeled with both pancreas and pancreatic cyst masks. The resolution of each CT scan is L, where L [358, 1121] is the number of sampling slices along the long axis of the body. The slice thickness varies from 0.5mm 1.0mm. We split the dataset into 4 fixed folds, and each of them contains approximately the same number of samples. We apply cross validation, i.e., training our approach on 3 out of 4 folds and testing it on the remaining one. We measure the segmentation accuracy by computing the Dice-Sørensen Coefficient (DSC) for each 3D volume. This is a similarity metric between the prediction voxel set A and the ground-truth set G, its mathematical form is DSC(A, G) = 2 A G A + G. We report the average DSC score together with other statistics over all 131 testing cases from 4 testing folds. 3.2 Results Cystic Pancreas Segmentation. We first investigate pathological pancreas segmentation which serves as the first stage of our approach. With the baseline approach described in [17], we obtain an average DSC of ± 9.72%. Please note that this number is lower than ± 5.68%, which was reported by the same approach in the NIH pancreas segmentation dataset with 82 healthy samples. Meanwhile, we report 34.65% DSC in the worst pathological case, while this number is 62.43% in the NIH dataset [17]. Therefore, we can conclude that a cystic pancreas is more difficult to segment than a normal case.
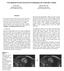
6 6 Y. Zhou et al. Method Mean DSC Max/Min DSC Pancreas Segmentation, w/ GT Pancreas B-Box ± /69.54 Pancreas Segmentation ± /34.65 Cyst Segmentation, w/ GT Cyst B-Box ± /24.69 Cyst Segmentation, w/o Deep Supervision ± /0.00 Cyst Segmentation, w/ Deep Supervision ± /0.00 Table 1. Pancreas and cyst segmentation accuracy, measured by DSC (%), produced by different approaches. Bold fonts indicate the results that oracle information (groundtruth bounding box) is not used. With deep supervision, the average accuracy cyst segmentation is improved, and the standard deviation is decreased. Pancreatic Cyst Segmentation. Based on the predicted pancreas mask, we now study the pancreatic cyst segmentation which is the second stage in our approach. Over 131 testing cases, our approach reports an average DSC of 63.60±27.71%, obtaining 2.98% absolute or 4.92% relative accuracy gain over the baseline. The high standard deviation (27.71%) indicates the significant variance in the difficulty of cyst segmentation. On the one hand, our approach can report rather high accuracy (e.g., > 95% DSC) in some easy cases. On the other hand, in some challenging cases, if the oracle cyst bounding box is unavailable, both approaches (with or without deep supervision) can come into a complete failure (i.e., DSC is 0%). In comparison, our approach with deep supervision misses 8 cyst cases, while the version without deep supervision misses 16. To the best of our knowledge, pancreatic cyst segmentation is very few studied previously. A competitor is [3] published in 2016, which combines random walk and region growth for segmentation. However, it requires the user to annotate the region-of-interest (ROI) beforehand, and provide interactive annotations on foreground/background voxels throughout the segmentation process. In comparison, when the bounding box is provided or not, our approach achieves 77.92% and 63.44% average accuracies, respectively. Being cheap or free in extra annotation, our approach can be widely applied to automatic diagnosis, especially for the common users without professional knowledge in medicine. Visualization. Three representative cases are shown in Figure 3. In the first case, both the pancreas and the cyst can be segmented accurately from the original CT scan. In the second case, however, the cyst is small in volume and less discriminative in contrast, and thus an accurate segmentation is only possible when we roughly localize the pancreas and shrink the input image size accordingly. The accuracy gain of our approach mainly owes to the accuracy gain of this type of cases. The third case shows a failure example of our approach, in which an inaccurate pancreas segmentation leads to a complete missing in cyst detection. Note that the baseline approach reports a 59.93% DSC in this case, and, if the oracle pancreas bounding box is provided, we can still achieve a DSC of 77.56%. This inspires us that cyst segmentation can sometimes help pancreas segmentation, and this topic is left for future research.
7 Deep Supervision for Pancreatic Cyst Segmentation in Abdominal CT Scans 7 Input Image Pancreas Segmentation Fine-scaled Input Cyst Segmentation (+) Cyst Segmentation (-) Case #111 DSC = 68.21% DSC = 84.06% DSC = 74.86% Case #123 DSC = 73.59% DSC = 84.70% DSC = 0.00% This slice is ignored in testing Case #130 DSC = 34.65% DSC = 0.00% DSC = 59.93% Fig. 3. Sample pancreas and pancreatic cyst segmentation results (best viewed in color). From left to right: input image (in which pancreas and cyst are marked in red and green, respectively), pancreas segmentation result, and cyst segmentation results when we apply deep supervision (denoted by +) or not (-). The figures in the right three columns are zoomed in w.r.t. the red frames. In the last example, pancreas segmentation fails in this slice, resulting in a complete failure in cyst segmentation. 4 Conclusions This paper presents the first system for pancreatic cyst segmentation which can work without human assistance on the testing stage. Motivated by the high relevance of a cystic pancreas and a pancreatic cyst, we formulate pancreas segmentation as an explicit variable in the formulation, and introduce deep supervision to assist the network training process. The joint optimization can be factorized into two stages, making our approach very easy to implement. We collect a dataset with 131 pathological cases. Based on a coarse-to-fine segmentation algorithm, our approach produces reasonable cyst segmentation results. It is worth emphasizing that our approach does not require any extra human annotations on the testing stage, which is especially practical in assisting common patients in cheap and periodic clinical applications. This work teaches us that a lesion can be detected more effectively by considering its highly related organ(s). This knowledge, being simple and straightforward, is useful in the future work in pathological organ or lesion segmentation. Acknowledgements. This work was supported by the Lustgarten Foundation for Pancreatic Cancer Research. We thank Dr. Seyoun Park for enormous help.
8 8 Y. Zhou et al. References 1. Al-Ayyoub, M., Alawad, D., Al-Darabsah, K., Aljarrah, I.: Automatic Detection and Classification of Brain Hemorrhages. WSEAS Transactions on Computers (2013) 2. Christ, P., Ettlinger, F., Grün, F., Elshaera, M., Lipkova, J., Schlecht, S., Ahmaddy, F., Tatavarty, S., Bickel, M., Bilic, P., et al.: Automatic Liver and Tumor Segmentation of CT and MRI Volumes using Cascaded Fully Convolutional Neural Networks. arxiv preprint arxiv: (2017) 3. Dmitriev, K., Gutenko, I., Nadeem, S., Kaufman, A.: Pancreas and Cyst Segmentation. SPIE Medical Imaging pp C 97842C (2016) 4. Gintowt, A., Hac, S., Dobrowolski, S., Śledziński, Z.: An Unusual Presentation of Pancreatic Pseudocyst Mimicking Cystic Neoplasm of the Pancreas: a Case Report. Cases Journal 2(1), 9138 (2009) 5. Havaei, M., Davy, A., Warde-Farley, D., Biard, A., Courville, A., Bengio, Y., Pal, C., Jodoin, P., Larochelle, H.: Brain Tumor Segmentation with Deep Neural Networks. Medical Image Analysis (2017) 6. Klauß, M., Schöbinger, M., Wolf, I., Werner, J., Meinzer, H., Kauczor, H., Grenacher, L.: Value of Three-dimensional Reconstructions in Pancreatic Carcinoma Using Multidetector CT: Initial Results. World Journal of Gastroenterology 15(46), (2009) 7. Krizhevsky, A., Sutskever, I., Hinton, G.: ImageNet Classification with Deep Convolutional Neural Networks. Advances in Neural Information Processing Systems (2012) 8. Lasboo, A., Rezai, P., Yaghmai, V.: Morphological Analysis of Pancreatic Cystic Masses. Academic Radiology 17(3), (2010) 9. Lee, C., Xie, S., Gallagher, P., Zhang, Z., Tu, Z.: Deeply-Supervised Nets. International Conference on Artificial Intelligence and Statistics (2015) 10. Long, J., Shelhamer, E., Darrell, T.: Fully Convolutional Networks for Semantic Segmentation. Computer Vision and Pattern Recognition (2015) 11. Milletari, F., Navab, N., Ahmadi, S.: V-Net: Fully Convolutional Neural Networks for Volumetric Medical Image Segmentation. arxiv preprint arxiv: (2016) 12. Roth, H., Lu, L., Farag, A., Sohn, A., Summers, R.: Spatial Aggregation of Holistically-Nested Networks for Automated Pancreas Segmentation. International Conference on Medical Image Computing and Computer-Assisted Intervention (2016) 13. Roth, H., Lu, L., Farag, A., Shin, H., Liu, J., Turkbey, E., Summers, R.: DeepOrgan: Multi-level Deep Convolutional Networks for Automated Pancreas Segmentation. International Conference on Medical Image Computing and Computer- Assisted Intervention (2015) 14. Simonyan, K., Zisserman, A.: Very Deep Convolutional Networks for Large-Scale Image Recognition. International Conference on Learning Representations (2015) 15. Stewart, B., Wild, C., et al.: World Cancer Report 2014 (2014) 16. Wang, D., Khosla, A., Gargeya, R., Irshad, H., Beck, A.: Deep Learning for Identifying Metastatic Breast Cancer. arxiv preprint arxiv: (2016) 17. Zhou, Y., Xie, L., Shen, W., Fishman, E., Yuille, A.: Pancreas Segmentation in Abdominal CT Scan: A Coarse-to-Fine Approach. arxiv preprint arxiv: (2016)
HHS Public Access Author manuscript Med Image Comput Comput Assist Interv. Author manuscript; available in PMC 2018 January 04.
 Discriminative Localization in CNNs for Weakly-Supervised Segmentation of Pulmonary Nodules Xinyang Feng 1, Jie Yang 1, Andrew F. Laine 1, and Elsa D. Angelini 1,2 1 Department of Biomedical Engineering,
Discriminative Localization in CNNs for Weakly-Supervised Segmentation of Pulmonary Nodules Xinyang Feng 1, Jie Yang 1, Andrew F. Laine 1, and Elsa D. Angelini 1,2 1 Department of Biomedical Engineering,
arxiv: v1 [cs.cv] 13 Jul 2018
![arxiv: v1 [cs.cv] 13 Jul 2018 arxiv: v1 [cs.cv] 13 Jul 2018](/thumbs/84/89699669.jpg) Multi-Scale Convolutional-Stack Aggregation for Robust White Matter Hyperintensities Segmentation Hongwei Li 1, Jianguo Zhang 3, Mark Muehlau 2, Jan Kirschke 2, and Bjoern Menze 1 arxiv:1807.05153v1 [cs.cv]
Multi-Scale Convolutional-Stack Aggregation for Robust White Matter Hyperintensities Segmentation Hongwei Li 1, Jianguo Zhang 3, Mark Muehlau 2, Jan Kirschke 2, and Bjoern Menze 1 arxiv:1807.05153v1 [cs.cv]
arxiv: v1 [cs.cv] 17 Aug 2017
![arxiv: v1 [cs.cv] 17 Aug 2017 arxiv: v1 [cs.cv] 17 Aug 2017](/thumbs/78/78278805.jpg) Deep Learning for Medical Image Analysis Mina Rezaei, Haojin Yang, Christoph Meinel Hasso Plattner Institute, Prof.Dr.Helmert-Strae 2-3, 14482 Potsdam, Germany {mina.rezaei,haojin.yang,christoph.meinel}@hpi.de
Deep Learning for Medical Image Analysis Mina Rezaei, Haojin Yang, Christoph Meinel Hasso Plattner Institute, Prof.Dr.Helmert-Strae 2-3, 14482 Potsdam, Germany {mina.rezaei,haojin.yang,christoph.meinel}@hpi.de
Object Detectors Emerge in Deep Scene CNNs
 Object Detectors Emerge in Deep Scene CNNs Bolei Zhou, Aditya Khosla, Agata Lapedriza, Aude Oliva, Antonio Torralba Presented By: Collin McCarthy Goal: Understand how objects are represented in CNNs Are
Object Detectors Emerge in Deep Scene CNNs Bolei Zhou, Aditya Khosla, Agata Lapedriza, Aude Oliva, Antonio Torralba Presented By: Collin McCarthy Goal: Understand how objects are represented in CNNs Are
Multi-attention Guided Activation Propagation in CNNs
 Multi-attention Guided Activation Propagation in CNNs Xiangteng He and Yuxin Peng (B) Institute of Computer Science and Technology, Peking University, Beijing, China pengyuxin@pku.edu.cn Abstract. CNNs
Multi-attention Guided Activation Propagation in CNNs Xiangteng He and Yuxin Peng (B) Institute of Computer Science and Technology, Peking University, Beijing, China pengyuxin@pku.edu.cn Abstract. CNNs
POC Brain Tumor Segmentation. vlife Use Case
 Brain Tumor Segmentation vlife Use Case 1 Automatic Brain Tumor Segmentation using CNN Background Brain tumor segmentation seeks to separate healthy tissue from tumorous regions such as the advancing tumor,
Brain Tumor Segmentation vlife Use Case 1 Automatic Brain Tumor Segmentation using CNN Background Brain tumor segmentation seeks to separate healthy tissue from tumorous regions such as the advancing tumor,
Comparison of Two Approaches for Direct Food Calorie Estimation
 Comparison of Two Approaches for Direct Food Calorie Estimation Takumi Ege and Keiji Yanai Department of Informatics, The University of Electro-Communications, Tokyo 1-5-1 Chofugaoka, Chofu-shi, Tokyo
Comparison of Two Approaches for Direct Food Calorie Estimation Takumi Ege and Keiji Yanai Department of Informatics, The University of Electro-Communications, Tokyo 1-5-1 Chofugaoka, Chofu-shi, Tokyo
arxiv: v1 [cs.cv] 30 May 2018
![arxiv: v1 [cs.cv] 30 May 2018 arxiv: v1 [cs.cv] 30 May 2018](/thumbs/85/92577534.jpg) A Robust and Effective Approach Towards Accurate Metastasis Detection and pn-stage Classification in Breast Cancer Byungjae Lee and Kyunghyun Paeng Lunit inc., Seoul, South Korea {jaylee,khpaeng}@lunit.io
A Robust and Effective Approach Towards Accurate Metastasis Detection and pn-stage Classification in Breast Cancer Byungjae Lee and Kyunghyun Paeng Lunit inc., Seoul, South Korea {jaylee,khpaeng}@lunit.io
B657: Final Project Report Holistically-Nested Edge Detection
 B657: Final roject Report Holistically-Nested Edge Detection Mingze Xu & Hanfei Mei May 4, 2016 Abstract Holistically-Nested Edge Detection (HED), which is a novel edge detection method based on fully
B657: Final roject Report Holistically-Nested Edge Detection Mingze Xu & Hanfei Mei May 4, 2016 Abstract Holistically-Nested Edge Detection (HED), which is a novel edge detection method based on fully
arxiv: v2 [cs.cv] 19 Dec 2017
![arxiv: v2 [cs.cv] 19 Dec 2017 arxiv: v2 [cs.cv] 19 Dec 2017](/thumbs/89/97557782.jpg) An Ensemble of Deep Convolutional Neural Networks for Alzheimer s Disease Detection and Classification arxiv:1712.01675v2 [cs.cv] 19 Dec 2017 Jyoti Islam Department of Computer Science Georgia State University
An Ensemble of Deep Convolutional Neural Networks for Alzheimer s Disease Detection and Classification arxiv:1712.01675v2 [cs.cv] 19 Dec 2017 Jyoti Islam Department of Computer Science Georgia State University
A HMM-based Pre-training Approach for Sequential Data
 A HMM-based Pre-training Approach for Sequential Data Luca Pasa 1, Alberto Testolin 2, Alessandro Sperduti 1 1- Department of Mathematics 2- Department of Developmental Psychology and Socialisation University
A HMM-based Pre-training Approach for Sequential Data Luca Pasa 1, Alberto Testolin 2, Alessandro Sperduti 1 1- Department of Mathematics 2- Department of Developmental Psychology and Socialisation University
Highly Accurate Brain Stroke Diagnostic System and Generative Lesion Model. Junghwan Cho, Ph.D. CAIDE Systems, Inc. Deep Learning R&D Team
 Highly Accurate Brain Stroke Diagnostic System and Generative Lesion Model Junghwan Cho, Ph.D. CAIDE Systems, Inc. Deep Learning R&D Team Established in September, 2016 at 110 Canal st. Lowell, MA 01852,
Highly Accurate Brain Stroke Diagnostic System and Generative Lesion Model Junghwan Cho, Ph.D. CAIDE Systems, Inc. Deep Learning R&D Team Established in September, 2016 at 110 Canal st. Lowell, MA 01852,
CSE Introduction to High-Perfomance Deep Learning ImageNet & VGG. Jihyung Kil
 CSE 5194.01 - Introduction to High-Perfomance Deep Learning ImageNet & VGG Jihyung Kil ImageNet Classification with Deep Convolutional Neural Networks Alex Krizhevsky, Ilya Sutskever, Geoffrey E. Hinton,
CSE 5194.01 - Introduction to High-Perfomance Deep Learning ImageNet & VGG Jihyung Kil ImageNet Classification with Deep Convolutional Neural Networks Alex Krizhevsky, Ilya Sutskever, Geoffrey E. Hinton,
arxiv: v2 [cs.cv] 7 Jun 2018
![arxiv: v2 [cs.cv] 7 Jun 2018 arxiv: v2 [cs.cv] 7 Jun 2018](/thumbs/79/79761281.jpg) Deep supervision with additional labels for retinal vessel segmentation task Yishuo Zhang and Albert C.S. Chung Lo Kwee-Seong Medical Image Analysis Laboratory, Department of Computer Science and Engineering,
Deep supervision with additional labels for retinal vessel segmentation task Yishuo Zhang and Albert C.S. Chung Lo Kwee-Seong Medical Image Analysis Laboratory, Department of Computer Science and Engineering,
Computer based delineation and follow-up multisite abdominal tumors in longitudinal CT studies
 Research plan submitted for approval as a PhD thesis Submitted by: Refael Vivanti Supervisor: Professor Leo Joskowicz School of Engineering and Computer Science, The Hebrew University of Jerusalem Computer
Research plan submitted for approval as a PhD thesis Submitted by: Refael Vivanti Supervisor: Professor Leo Joskowicz School of Engineering and Computer Science, The Hebrew University of Jerusalem Computer
Chair for Computer Aided Medical Procedures (CAMP) Seminar on Deep Learning for Medical Applications. Shadi Albarqouni Christoph Baur
 Chair for (CAMP) Seminar on Deep Learning for Medical Applications Shadi Albarqouni Christoph Baur Results of matching system obtained via matching.in.tum.de 108 Applicants 9 % 10 % 9 % 14 % 30 % Rank
Chair for (CAMP) Seminar on Deep Learning for Medical Applications Shadi Albarqouni Christoph Baur Results of matching system obtained via matching.in.tum.de 108 Applicants 9 % 10 % 9 % 14 % 30 % Rank
arxiv: v1 [cs.cv] 9 Oct 2018
![arxiv: v1 [cs.cv] 9 Oct 2018 arxiv: v1 [cs.cv] 9 Oct 2018](/thumbs/95/126101195.jpg) Automatic Segmentation of Thoracic Aorta Segments in Low-Dose Chest CT Julia M. H. Noothout a, Bob D. de Vos a, Jelmer M. Wolterink a, Ivana Išgum a a Image Sciences Institute, University Medical Center
Automatic Segmentation of Thoracic Aorta Segments in Low-Dose Chest CT Julia M. H. Noothout a, Bob D. de Vos a, Jelmer M. Wolterink a, Ivana Išgum a a Image Sciences Institute, University Medical Center
A deep learning model integrating FCNNs and CRFs for brain tumor segmentation
 A deep learning model integrating FCNNs and CRFs for brain tumor segmentation Xiaomei Zhao 1,2, Yihong Wu 1, Guidong Song 3, Zhenye Li 4, Yazhuo Zhang,3,4,5,6, and Yong Fan 7 1 National Laboratory of Pattern
A deep learning model integrating FCNNs and CRFs for brain tumor segmentation Xiaomei Zhao 1,2, Yihong Wu 1, Guidong Song 3, Zhenye Li 4, Yazhuo Zhang,3,4,5,6, and Yong Fan 7 1 National Laboratory of Pattern
Differentiating Tumor and Edema in Brain Magnetic Resonance Images Using a Convolutional Neural Network
 Original Article Differentiating Tumor and Edema in Brain Magnetic Resonance Images Using a Convolutional Neural Network Aida Allahverdi 1, Siavash Akbarzadeh 1, Alireza Khorrami Moghaddam 2, Armin Allahverdy
Original Article Differentiating Tumor and Edema in Brain Magnetic Resonance Images Using a Convolutional Neural Network Aida Allahverdi 1, Siavash Akbarzadeh 1, Alireza Khorrami Moghaddam 2, Armin Allahverdy
Early Detection of Lung Cancer
 Early Detection of Lung Cancer Aswathy N Iyer Dept Of Electronics And Communication Engineering Lymie Jose Dept Of Electronics And Communication Engineering Anumol Thomas Dept Of Electronics And Communication
Early Detection of Lung Cancer Aswathy N Iyer Dept Of Electronics And Communication Engineering Lymie Jose Dept Of Electronics And Communication Engineering Anumol Thomas Dept Of Electronics And Communication
Research Article Multiscale CNNs for Brain Tumor Segmentation and Diagnosis
 Computational and Mathematical Methods in Medicine Volume 2016, Article ID 8356294, 7 pages http://dx.doi.org/10.1155/2016/8356294 Research Article Multiscale CNNs for Brain Tumor Segmentation and Diagnosis
Computational and Mathematical Methods in Medicine Volume 2016, Article ID 8356294, 7 pages http://dx.doi.org/10.1155/2016/8356294 Research Article Multiscale CNNs for Brain Tumor Segmentation and Diagnosis
DeepMiner: Discovering Interpretable Representations for Mammogram Classification and Explanation
 DeepMiner: Discovering Interpretable Representations for Mammogram Classification and Explanation Jimmy Wu 1, Bolei Zhou 1, Diondra Peck 2, Scott Hsieh 3, Vandana Dialani, MD 4 Lester Mackey 5, and Genevieve
DeepMiner: Discovering Interpretable Representations for Mammogram Classification and Explanation Jimmy Wu 1, Bolei Zhou 1, Diondra Peck 2, Scott Hsieh 3, Vandana Dialani, MD 4 Lester Mackey 5, and Genevieve
Holistically-Nested Edge Detection (HED)
 Holistically-Nested Edge Detection (HED) Saining Xie, Zhuowen Tu Presented by Yuxin Wu February 10, 20 What is an Edge? Local intensity change? Used in traditional methods: Canny, Sobel, etc Learn it!
Holistically-Nested Edge Detection (HED) Saining Xie, Zhuowen Tu Presented by Yuxin Wu February 10, 20 What is an Edge? Local intensity change? Used in traditional methods: Canny, Sobel, etc Learn it!
Segmentation of Tumor Region from Brain Mri Images Using Fuzzy C-Means Clustering And Seeded Region Growing
 IOSR Journal of Computer Engineering (IOSR-JCE) e-issn: 2278-0661,p-ISSN: 2278-8727, Volume 18, Issue 5, Ver. I (Sept - Oct. 2016), PP 20-24 www.iosrjournals.org Segmentation of Tumor Region from Brain
IOSR Journal of Computer Engineering (IOSR-JCE) e-issn: 2278-0661,p-ISSN: 2278-8727, Volume 18, Issue 5, Ver. I (Sept - Oct. 2016), PP 20-24 www.iosrjournals.org Segmentation of Tumor Region from Brain
PET in Radiation Therapy. Outline. Tumor Segmentation in PET and in Multimodality Images for Radiation Therapy. 1. Tumor segmentation in PET
 Tumor Segmentation in PET and in Multimodality Images for Radiation Therapy Wei Lu, Ph.D. Department of Radiation Oncology Mallinckrodt Institute of Radiology Washington University in St. Louis Outline
Tumor Segmentation in PET and in Multimodality Images for Radiation Therapy Wei Lu, Ph.D. Department of Radiation Oncology Mallinckrodt Institute of Radiology Washington University in St. Louis Outline
Motivation: Attention: Focusing on specific parts of the input. Inspired by neuroscience.
 Outline: Motivation. What s the attention mechanism? Soft attention vs. Hard attention. Attention in Machine translation. Attention in Image captioning. State-of-the-art. 1 Motivation: Attention: Focusing
Outline: Motivation. What s the attention mechanism? Soft attention vs. Hard attention. Attention in Machine translation. Attention in Image captioning. State-of-the-art. 1 Motivation: Attention: Focusing
Comparative Study of K-means, Gaussian Mixture Model, Fuzzy C-means algorithms for Brain Tumor Segmentation
 Comparative Study of K-means, Gaussian Mixture Model, Fuzzy C-means algorithms for Brain Tumor Segmentation U. Baid 1, S. Talbar 2 and S. Talbar 1 1 Department of E&TC Engineering, Shri Guru Gobind Singhji
Comparative Study of K-means, Gaussian Mixture Model, Fuzzy C-means algorithms for Brain Tumor Segmentation U. Baid 1, S. Talbar 2 and S. Talbar 1 1 Department of E&TC Engineering, Shri Guru Gobind Singhji
Copyright 2008 Society of Photo Optical Instrumentation Engineers. This paper was published in Proceedings of SPIE, vol. 6915, Medical Imaging 2008:
 Copyright 2008 Society of Photo Optical Instrumentation Engineers. This paper was published in Proceedings of SPIE, vol. 6915, Medical Imaging 2008: Computer Aided Diagnosis and is made available as an
Copyright 2008 Society of Photo Optical Instrumentation Engineers. This paper was published in Proceedings of SPIE, vol. 6915, Medical Imaging 2008: Computer Aided Diagnosis and is made available as an
arxiv: v1 [stat.ml] 23 Jan 2017
![arxiv: v1 [stat.ml] 23 Jan 2017 arxiv: v1 [stat.ml] 23 Jan 2017](/thumbs/81/83414183.jpg) Learning what to look in chest X-rays with a recurrent visual attention model arxiv:1701.06452v1 [stat.ml] 23 Jan 2017 Petros-Pavlos Ypsilantis Department of Biomedical Engineering King s College London
Learning what to look in chest X-rays with a recurrent visual attention model arxiv:1701.06452v1 [stat.ml] 23 Jan 2017 Petros-Pavlos Ypsilantis Department of Biomedical Engineering King s College London
Confidence-based Ensemble for GBM brain tumor segmentation
 Confidence-based Ensemble for GBM brain tumor segmentation Jing Huo 1, Eva M. van Rikxoort 1, Kazunori Okada 2, Hyun J. Kim 1, Whitney Pope 1, Jonathan Goldin 1, Matthew Brown 1 1 Center for Computer vision
Confidence-based Ensemble for GBM brain tumor segmentation Jing Huo 1, Eva M. van Rikxoort 1, Kazunori Okada 2, Hyun J. Kim 1, Whitney Pope 1, Jonathan Goldin 1, Matthew Brown 1 1 Center for Computer vision
EARLY STAGE DIAGNOSIS OF LUNG CANCER USING CT-SCAN IMAGES BASED ON CELLULAR LEARNING AUTOMATE
 EARLY STAGE DIAGNOSIS OF LUNG CANCER USING CT-SCAN IMAGES BASED ON CELLULAR LEARNING AUTOMATE SAKTHI NEELA.P.K Department of M.E (Medical electronics) Sengunthar College of engineering Namakkal, Tamilnadu,
EARLY STAGE DIAGNOSIS OF LUNG CANCER USING CT-SCAN IMAGES BASED ON CELLULAR LEARNING AUTOMATE SAKTHI NEELA.P.K Department of M.E (Medical electronics) Sengunthar College of engineering Namakkal, Tamilnadu,
Image-Based Estimation of Real Food Size for Accurate Food Calorie Estimation
 Image-Based Estimation of Real Food Size for Accurate Food Calorie Estimation Takumi Ege, Yoshikazu Ando, Ryosuke Tanno, Wataru Shimoda and Keiji Yanai Department of Informatics, The University of Electro-Communications,
Image-Based Estimation of Real Food Size for Accurate Food Calorie Estimation Takumi Ege, Yoshikazu Ando, Ryosuke Tanno, Wataru Shimoda and Keiji Yanai Department of Informatics, The University of Electro-Communications,
When Saliency Meets Sentiment: Understanding How Image Content Invokes Emotion and Sentiment
 When Saliency Meets Sentiment: Understanding How Image Content Invokes Emotion and Sentiment Honglin Zheng1, Tianlang Chen2, Jiebo Luo3 Department of Computer Science University of Rochester, Rochester,
When Saliency Meets Sentiment: Understanding How Image Content Invokes Emotion and Sentiment Honglin Zheng1, Tianlang Chen2, Jiebo Luo3 Department of Computer Science University of Rochester, Rochester,
Skin cancer reorganization and classification with deep neural network
 Skin cancer reorganization and classification with deep neural network Hao Chang 1 1. Department of Genetics, Yale University School of Medicine 2. Email: changhao86@gmail.com Abstract As one kind of skin
Skin cancer reorganization and classification with deep neural network Hao Chang 1 1. Department of Genetics, Yale University School of Medicine 2. Email: changhao86@gmail.com Abstract As one kind of skin
arxiv: v2 [cs.cv] 22 Mar 2018
![arxiv: v2 [cs.cv] 22 Mar 2018 arxiv: v2 [cs.cv] 22 Mar 2018](/thumbs/94/122062874.jpg) Deep saliency: What is learnt by a deep network about saliency? Sen He 1 Nicolas Pugeault 1 arxiv:1801.04261v2 [cs.cv] 22 Mar 2018 Abstract Deep convolutional neural networks have achieved impressive performance
Deep saliency: What is learnt by a deep network about saliency? Sen He 1 Nicolas Pugeault 1 arxiv:1801.04261v2 [cs.cv] 22 Mar 2018 Abstract Deep convolutional neural networks have achieved impressive performance
Classification of breast cancer histology images using transfer learning
 Classification of breast cancer histology images using transfer learning Sulaiman Vesal 1 ( ), Nishant Ravikumar 1, AmirAbbas Davari 1, Stephan Ellmann 2, Andreas Maier 1 1 Pattern Recognition Lab, Friedrich-Alexander-Universität
Classification of breast cancer histology images using transfer learning Sulaiman Vesal 1 ( ), Nishant Ravikumar 1, AmirAbbas Davari 1, Stephan Ellmann 2, Andreas Maier 1 1 Pattern Recognition Lab, Friedrich-Alexander-Universität
3D Deep Learning for Multi-modal Imaging-Guided Survival Time Prediction of Brain Tumor Patients
 3D Deep Learning for Multi-modal Imaging-Guided Survival Time Prediction of Brain Tumor Patients Dong Nie 1,2, Han Zhang 1, Ehsan Adeli 1, Luyan Liu 1, and Dinggang Shen 1(B) 1 Department of Radiology
3D Deep Learning for Multi-modal Imaging-Guided Survival Time Prediction of Brain Tumor Patients Dong Nie 1,2, Han Zhang 1, Ehsan Adeli 1, Luyan Liu 1, and Dinggang Shen 1(B) 1 Department of Radiology
LUNG NODULE SEGMENTATION IN COMPUTED TOMOGRAPHY IMAGE. Hemahashiny, Ketheesan Department of Physical Science, Vavuniya Campus
 LUNG NODULE SEGMENTATION IN COMPUTED TOMOGRAPHY IMAGE Hemahashiny, Ketheesan Department of Physical Science, Vavuniya Campus tketheesan@vau.jfn.ac.lk ABSTRACT: The key process to detect the Lung cancer
LUNG NODULE SEGMENTATION IN COMPUTED TOMOGRAPHY IMAGE Hemahashiny, Ketheesan Department of Physical Science, Vavuniya Campus tketheesan@vau.jfn.ac.lk ABSTRACT: The key process to detect the Lung cancer
arxiv: v3 [cs.cv] 26 May 2018
![arxiv: v3 [cs.cv] 26 May 2018 arxiv: v3 [cs.cv] 26 May 2018](/thumbs/86/94630916.jpg) DeepEM: Deep 3D ConvNets With EM For Weakly Supervised Pulmonary Nodule Detection Wentao Zhu, Yeeleng S. Vang, Yufang Huang, and Xiaohui Xie University of California, Irvine Lenovo AI Lab {wentaoz1,ysvang,xhx}@uci.edu,
DeepEM: Deep 3D ConvNets With EM For Weakly Supervised Pulmonary Nodule Detection Wentao Zhu, Yeeleng S. Vang, Yufang Huang, and Xiaohui Xie University of California, Irvine Lenovo AI Lab {wentaoz1,ysvang,xhx}@uci.edu,
Compute-aided Differentiation of Focal Liver Disease in MR Imaging
 1063 Compute-aided Differentiation of Focal Liver Disease in MR Imaging Xuejun Zhang a, Masayuki Kanematsu b, Hiroshi Fujita c, Takeshi Hara c, Hiroshi Kondo b, Xiangrong Zhou c, Wenguang Li a and Hiroaki
1063 Compute-aided Differentiation of Focal Liver Disease in MR Imaging Xuejun Zhang a, Masayuki Kanematsu b, Hiroshi Fujita c, Takeshi Hara c, Hiroshi Kondo b, Xiangrong Zhou c, Wenguang Li a and Hiroaki
Segmentation of Cell Membrane and Nucleus by Improving Pix2pix
 Segmentation of Membrane and Nucleus by Improving Pix2pix Masaya Sato 1, Kazuhiro Hotta 1, Ayako Imanishi 2, Michiyuki Matsuda 2 and Kenta Terai 2 1 Meijo University, Siogamaguchi, Nagoya, Aichi, Japan
Segmentation of Membrane and Nucleus by Improving Pix2pix Masaya Sato 1, Kazuhiro Hotta 1, Ayako Imanishi 2, Michiyuki Matsuda 2 and Kenta Terai 2 1 Meijo University, Siogamaguchi, Nagoya, Aichi, Japan
Mammogram Analysis: Tumor Classification
 Mammogram Analysis: Tumor Classification Term Project Report Geethapriya Raghavan geeragh@mail.utexas.edu EE 381K - Multidimensional Digital Signal Processing Spring 2005 Abstract Breast cancer is the
Mammogram Analysis: Tumor Classification Term Project Report Geethapriya Raghavan geeragh@mail.utexas.edu EE 381K - Multidimensional Digital Signal Processing Spring 2005 Abstract Breast cancer is the
Final Report: Automated Semantic Segmentation of Volumetric Cardiovascular Features and Disease Assessment
 Final Report: Automated Semantic Segmentation of Volumetric Cardiovascular Features and Disease Assessment Tony Lindsey 1,3, Xiao Lu 1 and Mojtaba Tefagh 2 1 Department of Biomedical Informatics, Stanford
Final Report: Automated Semantic Segmentation of Volumetric Cardiovascular Features and Disease Assessment Tony Lindsey 1,3, Xiao Lu 1 and Mojtaba Tefagh 2 1 Department of Biomedical Informatics, Stanford
Automatic Definition of Planning Target Volume in Computer-Assisted Radiotherapy
 Automatic Definition of Planning Target Volume in Computer-Assisted Radiotherapy Angelo Zizzari Department of Cybernetics, School of Systems Engineering The University of Reading, Whiteknights, PO Box
Automatic Definition of Planning Target Volume in Computer-Assisted Radiotherapy Angelo Zizzari Department of Cybernetics, School of Systems Engineering The University of Reading, Whiteknights, PO Box
Deep CNNs for Diabetic Retinopathy Detection
 Deep CNNs for Diabetic Retinopathy Detection Alex Tamkin Stanford University atamkin@stanford.edu Iain Usiri Stanford University iusiri@stanford.edu Chala Fufa Stanford University cfufa@stanford.edu 1
Deep CNNs for Diabetic Retinopathy Detection Alex Tamkin Stanford University atamkin@stanford.edu Iain Usiri Stanford University iusiri@stanford.edu Chala Fufa Stanford University cfufa@stanford.edu 1
Magnetic Resonance Contrast Prediction Using Deep Learning
 Magnetic Resonance Contrast Prediction Using Deep Learning Cagan Alkan Department of Electrical Engineering Stanford University calkan@stanford.edu Andrew Weitz Department of Bioengineering Stanford University
Magnetic Resonance Contrast Prediction Using Deep Learning Cagan Alkan Department of Electrical Engineering Stanford University calkan@stanford.edu Andrew Weitz Department of Bioengineering Stanford University
Fully Convolutional Network-based Multi-Task Learning for Rectum and Rectal Cancer Segmentation
 Fully Convolutional Network-based Multi-Task Learning for Rectum and Rectal Cancer Segmentation Joohyung Lee 1, Ji Eun Oh 1, Min Ju Kim 2, Bo Yun Hur 2, Sun Ah Cho 1 1, 2*, and Dae Kyung Sohn 1 Innovative
Fully Convolutional Network-based Multi-Task Learning for Rectum and Rectal Cancer Segmentation Joohyung Lee 1, Ji Eun Oh 1, Min Ju Kim 2, Bo Yun Hur 2, Sun Ah Cho 1 1, 2*, and Dae Kyung Sohn 1 Innovative
Convolutional Neural Networks for Estimating Left Ventricular Volume
 Convolutional Neural Networks for Estimating Left Ventricular Volume Ryan Silva Stanford University rdsilva@stanford.edu Maksim Korolev Stanford University mkorolev@stanford.edu Abstract End-systolic and
Convolutional Neural Networks for Estimating Left Ventricular Volume Ryan Silva Stanford University rdsilva@stanford.edu Maksim Korolev Stanford University mkorolev@stanford.edu Abstract End-systolic and
arxiv: v2 [cs.cv] 8 Mar 2018
![arxiv: v2 [cs.cv] 8 Mar 2018 arxiv: v2 [cs.cv] 8 Mar 2018](/thumbs/87/97094636.jpg) Automated soft tissue lesion detection and segmentation in digital mammography using a u-net deep learning network Timothy de Moor a, Alejandro Rodriguez-Ruiz a, Albert Gubern Mérida a, Ritse Mann a, and
Automated soft tissue lesion detection and segmentation in digital mammography using a u-net deep learning network Timothy de Moor a, Alejandro Rodriguez-Ruiz a, Albert Gubern Mérida a, Ritse Mann a, and
Convolutional Neural Networks (CNN)
 Convolutional Neural Networks (CNN) Algorithm and Some Applications in Computer Vision Luo Hengliang Institute of Automation June 10, 2014 Luo Hengliang (Institute of Automation) Convolutional Neural Networks
Convolutional Neural Networks (CNN) Algorithm and Some Applications in Computer Vision Luo Hengliang Institute of Automation June 10, 2014 Luo Hengliang (Institute of Automation) Convolutional Neural Networks
International Journal of Advances in Engineering Research. (IJAER) 2018, Vol. No. 15, Issue No. IV, April e-issn: , p-issn:
 SUPERVISED MACHINE LEARNING ALGORITHMS: DEVELOPING AN EFFECTIVE USABILITY OF COMPUTERIZED TOMOGRAPHY DATA IN THE EARLY DETECTION OF LUNG CANCER IN SMALL CELL Pushkar Garg Delhi Public School, R.K. Puram,
SUPERVISED MACHINE LEARNING ALGORITHMS: DEVELOPING AN EFFECTIVE USABILITY OF COMPUTERIZED TOMOGRAPHY DATA IN THE EARLY DETECTION OF LUNG CANCER IN SMALL CELL Pushkar Garg Delhi Public School, R.K. Puram,
Understanding Convolutional Neural
 Understanding Convolutional Neural Networks Understanding Convolutional Neural Networks David Stutz July 24th, 2014 David Stutz July 24th, 2014 0/53 1/53 Table of Contents - Table of Contents 1 Motivation
Understanding Convolutional Neural Networks Understanding Convolutional Neural Networks David Stutz July 24th, 2014 David Stutz July 24th, 2014 0/53 1/53 Table of Contents - Table of Contents 1 Motivation
A deep learning model integrating FCNNs and CRFs for brain tumor segmentation
 A deep learning model integrating FCNNs and CRFs for brain tumor segmentation Xiaomei Zhao 1, Yihong Wu 1, Guidong Song 2, Zhenye Li 3, Yazhuo Zhang 2,3,4,5, and Yong Fan 6 1 National Laboratory of Pattern
A deep learning model integrating FCNNs and CRFs for brain tumor segmentation Xiaomei Zhao 1, Yihong Wu 1, Guidong Song 2, Zhenye Li 3, Yazhuo Zhang 2,3,4,5, and Yong Fan 6 1 National Laboratory of Pattern
A Novel Method using Convolutional Neural Network for Segmenting Brain Tumor in MRI Images
 A Novel Method using Convolutional Neural Network for Segmenting Brain Tumor in MRI Images K. Meena*, S. Pavitra**, N. Nishanthi*** & M. Nivetha**** *UG Student, Department of Electronics and Communication
A Novel Method using Convolutional Neural Network for Segmenting Brain Tumor in MRI Images K. Meena*, S. Pavitra**, N. Nishanthi*** & M. Nivetha**** *UG Student, Department of Electronics and Communication
Shu Kong. Department of Computer Science, UC Irvine
 Ubiquitous Fine-Grained Computer Vision Shu Kong Department of Computer Science, UC Irvine Outline 1. Problem definition 2. Instantiation 3. Challenge 4. Fine-grained classification with holistic representation
Ubiquitous Fine-Grained Computer Vision Shu Kong Department of Computer Science, UC Irvine Outline 1. Problem definition 2. Instantiation 3. Challenge 4. Fine-grained classification with holistic representation
On Training of Deep Neural Network. Lornechen
 On Training of Deep Neural Network Lornechen 2016.04.20 1 Outline Introduction Layer-wise Pre-training & Fine-tuning Activation Function Initialization Method Advanced Layers and Nets 2 Neural Network
On Training of Deep Neural Network Lornechen 2016.04.20 1 Outline Introduction Layer-wise Pre-training & Fine-tuning Activation Function Initialization Method Advanced Layers and Nets 2 Neural Network
Rich feature hierarchies for accurate object detection and semantic segmentation
 Rich feature hierarchies for accurate object detection and semantic segmentation Ross Girshick, Jeff Donahue, Trevor Darrell, Jitendra Malik UC Berkeley Tech Report @ http://arxiv.org/abs/1311.2524! Detection
Rich feature hierarchies for accurate object detection and semantic segmentation Ross Girshick, Jeff Donahue, Trevor Darrell, Jitendra Malik UC Berkeley Tech Report @ http://arxiv.org/abs/1311.2524! Detection
Expert identification of visual primitives used by CNNs during mammogram classification
 Expert identification of visual primitives used by CNNs during mammogram classification Jimmy Wu a, Diondra Peck b, Scott Hsieh c, Vandana Dialani, MD d, Constance D. Lehman, MD e, Bolei Zhou a, Vasilis
Expert identification of visual primitives used by CNNs during mammogram classification Jimmy Wu a, Diondra Peck b, Scott Hsieh c, Vandana Dialani, MD d, Constance D. Lehman, MD e, Bolei Zhou a, Vasilis
Lung Region Segmentation using Artificial Neural Network Hopfield Model for Cancer Diagnosis in Thorax CT Images
 Automation, Control and Intelligent Systems 2015; 3(2): 19-25 Published online March 20, 2015 (http://www.sciencepublishinggroup.com/j/acis) doi: 10.11648/j.acis.20150302.12 ISSN: 2328-5583 (Print); ISSN:
Automation, Control and Intelligent Systems 2015; 3(2): 19-25 Published online March 20, 2015 (http://www.sciencepublishinggroup.com/j/acis) doi: 10.11648/j.acis.20150302.12 ISSN: 2328-5583 (Print); ISSN:
arxiv: v2 [cs.lg] 30 Oct 2013
![arxiv: v2 [cs.lg] 30 Oct 2013 arxiv: v2 [cs.lg] 30 Oct 2013](/thumbs/76/73582431.jpg) Prediction of breast cancer recurrence using Classification Restricted Boltzmann Machine with Dropping arxiv:1308.6324v2 [cs.lg] 30 Oct 2013 Jakub M. Tomczak Wrocław University of Technology Wrocław, Poland
Prediction of breast cancer recurrence using Classification Restricted Boltzmann Machine with Dropping arxiv:1308.6324v2 [cs.lg] 30 Oct 2013 Jakub M. Tomczak Wrocław University of Technology Wrocław, Poland
Lung Cancer Diagnosis from CT Images Using Fuzzy Inference System
 Lung Cancer Diagnosis from CT Images Using Fuzzy Inference System T.Manikandan 1, Dr. N. Bharathi 2 1 Associate Professor, Rajalakshmi Engineering College, Chennai-602 105 2 Professor, Velammal Engineering
Lung Cancer Diagnosis from CT Images Using Fuzzy Inference System T.Manikandan 1, Dr. N. Bharathi 2 1 Associate Professor, Rajalakshmi Engineering College, Chennai-602 105 2 Professor, Velammal Engineering
Quantifying Radiographic Knee Osteoarthritis Severity using Deep Convolutional Neural Networks
 Quantifying Radiographic Knee Osteoarthritis Severity using Deep Convolutional Neural Networks Joseph Antony, Kevin McGuinness, Noel E O Connor, Kieran Moran Insight Centre for Data Analytics, Dublin City
Quantifying Radiographic Knee Osteoarthritis Severity using Deep Convolutional Neural Networks Joseph Antony, Kevin McGuinness, Noel E O Connor, Kieran Moran Insight Centre for Data Analytics, Dublin City
An Overview and Comparative Analysis on Major Generative Models
 An Overview and Comparative Analysis on Major Generative Models Zijing Gu zig021@ucsd.edu Abstract The amount of researches on generative models has been grown rapidly after a period of silence due to
An Overview and Comparative Analysis on Major Generative Models Zijing Gu zig021@ucsd.edu Abstract The amount of researches on generative models has been grown rapidly after a period of silence due to
An Artificial Neural Network Architecture Based on Context Transformations in Cortical Minicolumns
 An Artificial Neural Network Architecture Based on Context Transformations in Cortical Minicolumns 1. Introduction Vasily Morzhakov, Alexey Redozubov morzhakovva@gmail.com, galdrd@gmail.com Abstract Cortical
An Artificial Neural Network Architecture Based on Context Transformations in Cortical Minicolumns 1. Introduction Vasily Morzhakov, Alexey Redozubov morzhakovva@gmail.com, galdrd@gmail.com Abstract Cortical
Shu Kong. Department of Computer Science, UC Irvine
 Ubiquitous Fine-Grained Computer Vision Shu Kong Department of Computer Science, UC Irvine Outline 1. Problem definition 2. Instantiation 3. Challenge and philosophy 4. Fine-grained classification with
Ubiquitous Fine-Grained Computer Vision Shu Kong Department of Computer Science, UC Irvine Outline 1. Problem definition 2. Instantiation 3. Challenge and philosophy 4. Fine-grained classification with
Automated detection of masses on whole breast volume ultrasound scanner: false positive reduction using deep convolutional neural network
 Automated detection of masses on whole breast volume ultrasound scanner: false positive reduction using deep convolutional neural network Yuya Hiramatsu a, Chisako Muramatsu* a, Hironobu Kobayashi b, Takeshi
Automated detection of masses on whole breast volume ultrasound scanner: false positive reduction using deep convolutional neural network Yuya Hiramatsu a, Chisako Muramatsu* a, Hironobu Kobayashi b, Takeshi
BLADDERSCAN PRIME PLUS TM DEEP LEARNING
 BLADDERSCAN PRIME PLUS TM DEEP LEARNING BladderScan Prime Plus from Verathon Takes Accuracy and Ease of Use to a New Level Powered by ImageSense TM deep learning technology, an advanced implementation
BLADDERSCAN PRIME PLUS TM DEEP LEARNING BladderScan Prime Plus from Verathon Takes Accuracy and Ease of Use to a New Level Powered by ImageSense TM deep learning technology, an advanced implementation
Detection and Classification of Lung Cancer Using Artificial Neural Network
 Detection and Classification of Lung Cancer Using Artificial Neural Network Almas Pathan 1, Bairu.K.saptalkar 2 1,2 Department of Electronics and Communication Engineering, SDMCET, Dharwad, India 1 almaseng@yahoo.co.in,
Detection and Classification of Lung Cancer Using Artificial Neural Network Almas Pathan 1, Bairu.K.saptalkar 2 1,2 Department of Electronics and Communication Engineering, SDMCET, Dharwad, India 1 almaseng@yahoo.co.in,
arxiv: v2 [cs.cv] 10 Apr 2017 Abstract
![arxiv: v2 [cs.cv] 10 Apr 2017 Abstract arxiv: v2 [cs.cv] 10 Apr 2017 Abstract](/thumbs/85/92918924.jpg) SCAN: Structure Correcting Adversarial Network for Organ Segmentation in Chest X-rays Wei Dai, Joseph Doyle, Xiaodan Liang, Hao Zhang, Nanqing Dong, Yuan Li, Eric P. Xing Petuum Inc. {wei.dai,joe.doyle,xiaodan.,hao.zhang,nanqing.dong,christy.li,eric.xing}@petuum.com
SCAN: Structure Correcting Adversarial Network for Organ Segmentation in Chest X-rays Wei Dai, Joseph Doyle, Xiaodan Liang, Hao Zhang, Nanqing Dong, Yuan Li, Eric P. Xing Petuum Inc. {wei.dai,joe.doyle,xiaodan.,hao.zhang,nanqing.dong,christy.li,eric.xing}@petuum.com
Lung Nodule Segmentation Using 3D Convolutional Neural Networks
 Lung Nodule Segmentation Using 3D Convolutional Neural Networks Research paper Business Analytics Bernard Bronmans Master Business Analytics VU University, Amsterdam Evert Haasdijk Supervisor VU University,
Lung Nodule Segmentation Using 3D Convolutional Neural Networks Research paper Business Analytics Bernard Bronmans Master Business Analytics VU University, Amsterdam Evert Haasdijk Supervisor VU University,
Automatic Atlas-based Three-label Cartilage Segmentation from MR Knee Images
 Automatic Atlas-based Three-label Cartilage Segmentation from MR Knee Images Liang Shan Department of Computer Science UNC Chapel Hill shan@cs.unc.edu Marc Niethammer Department of Computer Science UNC
Automatic Atlas-based Three-label Cartilage Segmentation from MR Knee Images Liang Shan Department of Computer Science UNC Chapel Hill shan@cs.unc.edu Marc Niethammer Department of Computer Science UNC
arxiv: v1 [cs.cv] 3 Apr 2018
![arxiv: v1 [cs.cv] 3 Apr 2018 arxiv: v1 [cs.cv] 3 Apr 2018](/thumbs/90/103707026.jpg) Towards whole-body CT Bone Segmentation André Klein 1,2, Jan Warszawski 2, Jens Hillengaß 3, Klaus H. Maier-Hein 1 arxiv:1804.00908v1 [cs.cv] 3 Apr 2018 1 Division of Medical Image Computing, Deutsches
Towards whole-body CT Bone Segmentation André Klein 1,2, Jan Warszawski 2, Jens Hillengaß 3, Klaus H. Maier-Hein 1 arxiv:1804.00908v1 [cs.cv] 3 Apr 2018 1 Division of Medical Image Computing, Deutsches
arxiv: v3 [stat.ml] 27 Mar 2018
![arxiv: v3 [stat.ml] 27 Mar 2018 arxiv: v3 [stat.ml] 27 Mar 2018](/thumbs/82/87026346.jpg) ATTACKING THE MADRY DEFENSE MODEL WITH L 1 -BASED ADVERSARIAL EXAMPLES Yash Sharma 1 and Pin-Yu Chen 2 1 The Cooper Union, New York, NY 10003, USA 2 IBM Research, Yorktown Heights, NY 10598, USA sharma2@cooper.edu,
ATTACKING THE MADRY DEFENSE MODEL WITH L 1 -BASED ADVERSARIAL EXAMPLES Yash Sharma 1 and Pin-Yu Chen 2 1 The Cooper Union, New York, NY 10003, USA 2 IBM Research, Yorktown Heights, NY 10598, USA sharma2@cooper.edu,
arxiv: v2 [cs.cv] 3 Jun 2018
![arxiv: v2 [cs.cv] 3 Jun 2018 arxiv: v2 [cs.cv] 3 Jun 2018](/thumbs/89/97775247.jpg) S4ND: Single-Shot Single-Scale Lung Nodule Detection Naji Khosravan and Ulas Bagci Center for Research in Computer Vision (CRCV), School of Computer Science, University of Central Florida, Orlando, FL.
S4ND: Single-Shot Single-Scale Lung Nodule Detection Naji Khosravan and Ulas Bagci Center for Research in Computer Vision (CRCV), School of Computer Science, University of Central Florida, Orlando, FL.
Tumor cut segmentation for Blemish Cells Detection in Human Brain Based on Cellular Automata
 Tumor cut segmentation for Blemish Cells Detection in Human Brain Based on Cellular Automata D.Mohanapriya 1 Department of Electronics and Communication Engineering, EBET Group of Institutions, Kangayam,
Tumor cut segmentation for Blemish Cells Detection in Human Brain Based on Cellular Automata D.Mohanapriya 1 Department of Electronics and Communication Engineering, EBET Group of Institutions, Kangayam,
Brain Tumour Diagnostic Support Based on Medical Image Segmentation
 Brain Tumour Diagnostic Support Based on Medical Image Segmentation Z. Měřínský, E. Hošťálková, A. Procházka Institute of Chemical Technology, Prague Department of Computing and Control Engineering Abstract
Brain Tumour Diagnostic Support Based on Medical Image Segmentation Z. Měřínský, E. Hošťálková, A. Procházka Institute of Chemical Technology, Prague Department of Computing and Control Engineering Abstract
Deep Networks and Beyond. Alan Yuille Bloomberg Distinguished Professor Depts. Cognitive Science and Computer Science Johns Hopkins University
 Deep Networks and Beyond Alan Yuille Bloomberg Distinguished Professor Depts. Cognitive Science and Computer Science Johns Hopkins University Artificial Intelligence versus Human Intelligence Understanding
Deep Networks and Beyond Alan Yuille Bloomberg Distinguished Professor Depts. Cognitive Science and Computer Science Johns Hopkins University Artificial Intelligence versus Human Intelligence Understanding
NIH Public Access Author Manuscript Proc SPIE. Author manuscript; available in PMC 2014 February 07.
 NIH Public Access Author Manuscript Published in final edited form as: Proc SPIE. 2007 March 5; 6512: 651236. doi:10.1117/12.708950. Semi-Automatic Parcellation of the Corpus Striatum Ramsey Al-Hakim a,
NIH Public Access Author Manuscript Published in final edited form as: Proc SPIE. 2007 March 5; 6512: 651236. doi:10.1117/12.708950. Semi-Automatic Parcellation of the Corpus Striatum Ramsey Al-Hakim a,
Copyright 2007 IEEE. Reprinted from 4th IEEE International Symposium on Biomedical Imaging: From Nano to Macro, April 2007.
 Copyright 27 IEEE. Reprinted from 4th IEEE International Symposium on Biomedical Imaging: From Nano to Macro, April 27. This material is posted here with permission of the IEEE. Such permission of the
Copyright 27 IEEE. Reprinted from 4th IEEE International Symposium on Biomedical Imaging: From Nano to Macro, April 27. This material is posted here with permission of the IEEE. Such permission of the
Deep learning approaches to medical applications
 Deep learning approaches to medical applications Joseph Paul Cohen Postdoctoral Fellow at Montreal Institute for Learning Algorithms Friend of the Farlow Fellow at Harvard University U.S. National Science
Deep learning approaches to medical applications Joseph Paul Cohen Postdoctoral Fellow at Montreal Institute for Learning Algorithms Friend of the Farlow Fellow at Harvard University U.S. National Science
Y-Net: Joint Segmentation and Classification for Diagnosis of Breast Biopsy Images
 Y-Net: Joint Segmentation and Classification for Diagnosis of Breast Biopsy Images Sachin Mehta 1, Ezgi Mercan 1, Jamen Bartlett 2, Donald Weaver 2, Joann G. Elmore 1, and Linda Shapiro 1 1 University
Y-Net: Joint Segmentation and Classification for Diagnosis of Breast Biopsy Images Sachin Mehta 1, Ezgi Mercan 1, Jamen Bartlett 2, Donald Weaver 2, Joann G. Elmore 1, and Linda Shapiro 1 1 University
Automatic Detection of Knee Joints and Quantification of Knee Osteoarthritis Severity using Convolutional Neural Networks
 Automatic Detection of Knee Joints and Quantification of Knee Osteoarthritis Severity using Convolutional Neural Networks Joseph Antony 1, Kevin McGuinness 1, Kieran Moran 1,2 and Noel E O Connor 1 Insight
Automatic Detection of Knee Joints and Quantification of Knee Osteoarthritis Severity using Convolutional Neural Networks Joseph Antony 1, Kevin McGuinness 1, Kieran Moran 1,2 and Noel E O Connor 1 Insight
Neural Network Diagnosis of Avascular Necrosis from Magnetic Resonance Images
 Neural Network Diagnosis of Avascular Necrosis from Magnetic Resonance Images Armando Manduca Dept. of Physiology and Biophysics Mayo Clinic Rochester, MN 55905 Paul Christy Dept. of Diagnostic Radiology
Neural Network Diagnosis of Avascular Necrosis from Magnetic Resonance Images Armando Manduca Dept. of Physiology and Biophysics Mayo Clinic Rochester, MN 55905 Paul Christy Dept. of Diagnostic Radiology
arxiv: v1 [cs.cv] 2 Jun 2017
![arxiv: v1 [cs.cv] 2 Jun 2017 arxiv: v1 [cs.cv] 2 Jun 2017](/thumbs/74/71415505.jpg) INTEGRATED DEEP AND SHALLOW NETWORKS FOR SALIENT OBJECT DETECTION Jing Zhang 1,2, Bo Li 1, Yuchao Dai 2, Fatih Porikli 2 and Mingyi He 1 1 School of Electronics and Information, Northwestern Polytechnical
INTEGRATED DEEP AND SHALLOW NETWORKS FOR SALIENT OBJECT DETECTION Jing Zhang 1,2, Bo Li 1, Yuchao Dai 2, Fatih Porikli 2 and Mingyi He 1 1 School of Electronics and Information, Northwestern Polytechnical
Early Diagnosis of Autism Disease by Multi-channel CNNs
 Early Diagnosis of Autism Disease by Multi-channel CNNs Guannan Li 1,2, Mingxia Liu 2, Quansen Sun 1(&), Dinggang Shen 2(&), and Li Wang 2(&) 1 School of Computer Science and Engineering, Nanjing University
Early Diagnosis of Autism Disease by Multi-channel CNNs Guannan Li 1,2, Mingxia Liu 2, Quansen Sun 1(&), Dinggang Shen 2(&), and Li Wang 2(&) 1 School of Computer Science and Engineering, Nanjing University
Proceedings of the UGC Sponsored National Conference on Advanced Networking and Applications, 27 th March 2015
 Brain Tumor Detection and Identification Using K-Means Clustering Technique Malathi R Department of Computer Science, SAAS College, Ramanathapuram, Email: malapraba@gmail.com Dr. Nadirabanu Kamal A R Department
Brain Tumor Detection and Identification Using K-Means Clustering Technique Malathi R Department of Computer Science, SAAS College, Ramanathapuram, Email: malapraba@gmail.com Dr. Nadirabanu Kamal A R Department
Multi-Scale Salient Object Detection with Pyramid Spatial Pooling
 Multi-Scale Salient Object Detection with Pyramid Spatial Pooling Jing Zhang, Yuchao Dai, Fatih Porikli and Mingyi He School of Electronics and Information, Northwestern Polytechnical University, China.
Multi-Scale Salient Object Detection with Pyramid Spatial Pooling Jing Zhang, Yuchao Dai, Fatih Porikli and Mingyi He School of Electronics and Information, Northwestern Polytechnical University, China.
Healthcare Research You
 DL for Healthcare Goals Healthcare Research You What are high impact problems in healthcare that deep learning can solve? What does research in AI applications to medical imaging look like? How can you
DL for Healthcare Goals Healthcare Research You What are high impact problems in healthcare that deep learning can solve? What does research in AI applications to medical imaging look like? How can you
Deep learning and non-negative matrix factorization in recognition of mammograms
 Deep learning and non-negative matrix factorization in recognition of mammograms Bartosz Swiderski Faculty of Applied Informatics and Mathematics Warsaw University of Life Sciences, Warsaw, Poland bartosz_swiderski@sggw.pl
Deep learning and non-negative matrix factorization in recognition of mammograms Bartosz Swiderski Faculty of Applied Informatics and Mathematics Warsaw University of Life Sciences, Warsaw, Poland bartosz_swiderski@sggw.pl
PathGAN: Visual Scanpath Prediction with Generative Adversarial Networks
 PathGAN: Visual Scanpath Prediction with Generative Adversarial Networks Marc Assens 1, Kevin McGuinness 1, Xavier Giro-i-Nieto 2, and Noel E. O Connor 1 1 Insight Centre for Data Analytic, Dublin City
PathGAN: Visual Scanpath Prediction with Generative Adversarial Networks Marc Assens 1, Kevin McGuinness 1, Xavier Giro-i-Nieto 2, and Noel E. O Connor 1 1 Insight Centre for Data Analytic, Dublin City
Computational modeling of visual attention and saliency in the Smart Playroom
 Computational modeling of visual attention and saliency in the Smart Playroom Andrew Jones Department of Computer Science, Brown University Abstract The two canonical modes of human visual attention bottomup
Computational modeling of visual attention and saliency in the Smart Playroom Andrew Jones Department of Computer Science, Brown University Abstract The two canonical modes of human visual attention bottomup
Effective Diagnosis of Alzheimer s Disease by means of Association Rules
 Effective Diagnosis of Alzheimer s Disease by means of Association Rules Rosa Chaves (rosach@ugr.es) J. Ramírez, J.M. Górriz, M. López, D. Salas-Gonzalez, I. Illán, F. Segovia, P. Padilla Dpt. Theory of
Effective Diagnosis of Alzheimer s Disease by means of Association Rules Rosa Chaves (rosach@ugr.es) J. Ramírez, J.M. Górriz, M. López, D. Salas-Gonzalez, I. Illán, F. Segovia, P. Padilla Dpt. Theory of
Automated Mass Detection from Mammograms using Deep Learning and Random Forest
 Automated Mass Detection from Mammograms using Deep Learning and Random Forest Neeraj Dhungel 1 Gustavo Carneiro 1 Andrew P. Bradley 2 1 ACVT, University of Adelaide, Australia 2 University of Queensland,
Automated Mass Detection from Mammograms using Deep Learning and Random Forest Neeraj Dhungel 1 Gustavo Carneiro 1 Andrew P. Bradley 2 1 ACVT, University of Adelaide, Australia 2 University of Queensland,
Cancer Cells Detection using OTSU Threshold Algorithm
 Cancer Cells Detection using OTSU Threshold Algorithm Nalluri Sunny 1 Velagapudi Ramakrishna Siddhartha Engineering College Mithinti Srikanth 2 Velagapudi Ramakrishna Siddhartha Engineering College Kodali
Cancer Cells Detection using OTSU Threshold Algorithm Nalluri Sunny 1 Velagapudi Ramakrishna Siddhartha Engineering College Mithinti Srikanth 2 Velagapudi Ramakrishna Siddhartha Engineering College Kodali
arxiv: v1 [cs.cv] 24 Jul 2018
![arxiv: v1 [cs.cv] 24 Jul 2018 arxiv: v1 [cs.cv] 24 Jul 2018](/thumbs/82/86822918.jpg) Multi-Class Lesion Diagnosis with Pixel-wise Classification Network Manu Goyal 1, Jiahua Ng 2, and Moi Hoon Yap 1 1 Visual Computing Lab, Manchester Metropolitan University, M1 5GD, UK 2 University of
Multi-Class Lesion Diagnosis with Pixel-wise Classification Network Manu Goyal 1, Jiahua Ng 2, and Moi Hoon Yap 1 1 Visual Computing Lab, Manchester Metropolitan University, M1 5GD, UK 2 University of
Medical Image Analysis
 Medical Image Analysis 1 Co-trained convolutional neural networks for automated detection of prostate cancer in multiparametric MRI, 2017, Medical Image Analysis 2 Graph-based prostate extraction in t2-weighted
Medical Image Analysis 1 Co-trained convolutional neural networks for automated detection of prostate cancer in multiparametric MRI, 2017, Medical Image Analysis 2 Graph-based prostate extraction in t2-weighted
COMPUTERIZED SYSTEM DESIGN FOR THE DETECTION AND DIAGNOSIS OF LUNG NODULES IN CT IMAGES 1
 ISSN 258-8739 3 st August 28, Volume 3, Issue 2, JSEIS, CAOMEI Copyright 26-28 COMPUTERIZED SYSTEM DESIGN FOR THE DETECTION AND DIAGNOSIS OF LUNG NODULES IN CT IMAGES ALI ABDRHMAN UKASHA, 2 EMHMED SAAID
ISSN 258-8739 3 st August 28, Volume 3, Issue 2, JSEIS, CAOMEI Copyright 26-28 COMPUTERIZED SYSTEM DESIGN FOR THE DETECTION AND DIAGNOSIS OF LUNG NODULES IN CT IMAGES ALI ABDRHMAN UKASHA, 2 EMHMED SAAID
Deep-Learning Based Semantic Labeling for 2D Mammography & Comparison of Complexity for Machine Learning Tasks
 Deep-Learning Based Semantic Labeling for 2D Mammography & Comparison of Complexity for Machine Learning Tasks Paul H. Yi, MD, Abigail Lin, BSE, Jinchi Wei, BSE, Haris I. Sair, MD, Ferdinand K. Hui, MD,
Deep-Learning Based Semantic Labeling for 2D Mammography & Comparison of Complexity for Machine Learning Tasks Paul H. Yi, MD, Abigail Lin, BSE, Jinchi Wei, BSE, Haris I. Sair, MD, Ferdinand K. Hui, MD,
Implementation of Automatic Retina Exudates Segmentation Algorithm for Early Detection with Low Computational Time
 www.ijecs.in International Journal Of Engineering And Computer Science ISSN: 2319-7242 Volume 5 Issue 10 Oct. 2016, Page No. 18584-18588 Implementation of Automatic Retina Exudates Segmentation Algorithm
www.ijecs.in International Journal Of Engineering And Computer Science ISSN: 2319-7242 Volume 5 Issue 10 Oct. 2016, Page No. 18584-18588 Implementation of Automatic Retina Exudates Segmentation Algorithm
Network Dissection: Quantifying Interpretability of Deep Visual Representation
 Name: Pingchuan Ma Student number: 3526400 Date: August 19, 2018 Seminar: Explainable Machine Learning Lecturer: PD Dr. Ullrich Köthe SS 2018 Quantifying Interpretability of Deep Visual Representation
Name: Pingchuan Ma Student number: 3526400 Date: August 19, 2018 Seminar: Explainable Machine Learning Lecturer: PD Dr. Ullrich Köthe SS 2018 Quantifying Interpretability of Deep Visual Representation
