Paul McGovern, MD Vice President, Clinical Affairs Paratek Pharmaceuticals, Inc. Presented at ECCMID, 22 April 2018, Madrid, Spain.
|
|
|
- Oswald Bradford
- 6 years ago
- Views:
Transcription
1 A phase-3 randomized, double-blind, multicentre study to compare the safety and efficacy of oral omadacycline to oral linezolid for treating adult subjects with ABSSSI (OASIS-2 study) Paul McGovern, MD Vice President, Clinical Affairs Paratek Pharmaceuticals, Inc 4/22/ Presented at ECCMID, 22 April 2018, Madrid, Spain.
2 Omadacycline Investigational Product Overview Restoring Tetracycline Activity by Overcoming Resistance Aminomethylcycline Key Product Attributes 7-Position Modification: Overcomes Efflux Pump Safety and efficacy demonstrated in ABSSSI and CABP R1 R2 N R3 R1 R2H N R3 N H H H OH NH 2 N In-vitro activity against important community pathogens Two once-daily formulations intravenous and oral OH IV and oral doses with equivalent exposure (AUC 1 ) NH 2 OH O HO O O OH O OOH O H H O O 9-Position Modification: Overcomes Ribosomal Protection 4/22/ AUC = Area Under the Curve
3 Design of the Phase 3 OASIS-2 Study Once-Daily Oral Omadacycline vs Twice-Daily Oral Linezolid (1:1) ABSSSI N = 735 Oral Omadacycline 450mg once-daily Day 1 & 2 then 300mg once-daily Oral Linezolid 600mg twice-daily Day 1 FDA Day 7 to Day 14 End of Treatment (EOT) EMA Day 2-3 Early Clinical Response (ECR) 7-14 day after last treatment day Post-Treatment Evaluation (PTE) 4/22/2018 3
4 Demographic Characteristics Balanced Demographics Between Treatment Arms Gender a n (%) Characteristics Omadacycline Linezolid Female 126 (34.2) 147 (40.1) Male 242 (65.8) 220 (59.9) Age a (years) Mean (SD) 42.8 (12.72) 44.5 (13.11) BMI a,b (kg/m 2 ) Mean (SD) (6.472) (6.556) Lesion area c (cm 2 ) Mean (range) 422 ( ) 396 ( ) Type of Primary Infection c n (%) Wound Infection 210 (58.3) 214 (59.4) Cellulitis/erysipelas 86 (23.9) 84 (23.3) Major Abscess 64 (17.8) 62 (17.2) a Safety Population (OMC 368; LZD 367); b BMI = body mass index; c mitt Population (OMC 360; LZD 360) 4/22/2018 4
5 Oral Omadacycline Effective in ABSSSI Met Non-Inferiority for Both FDA and EMA Efficacy Endpoints Omadacycline Linezolid Clinical Success (%) mitt Early Clinical Response mitt PTE - Clinical Success CE-PTE - Clinical Success Delta (95% CI) +5.0 (-0.2, 10.3) Delta (95% CI) +3.3 (-2.2, 9.0) Delta (95% CI) +2.3 (-0.5, 5.8) FDA Primary Endpoint EMA Co-Primary Endpoints 4/22/2018 5
6 Clinical Response by Pathogen at PTE - micro-mitt Population Microbiological Responses Consistent with Clinical Responses Clinical Success, % S. aureus (All) MRSA MSSA S. anginosus Group S. pyogenes E. faecalis (VSE) OMC, n LZD, n /22/2018 6
7 Overview of Adverse Events Safety Population Omadacycline Was Safe and Generally Well Tolerated Subjects with at Least One Parameter Omadacycline N= 368 n(%) Linezolid N= 367 n(%) Treatment-Emergent Adverse Event (TEAE) a 197 (53.5) 137 (37.3) Severe TEAE 6 (1.6) 7 (1.9) Serious TEAE 5 (1.4) 5 (1.4) TEAE leading to premature discontinuation of test article b 6 (1.6) 3 (0.8) Most Frequent TEAEs a (>2% omadacycline arm) Nausea 111 (30.2) c 28 (7.6) Vomiting 62 (16.8) c 11 (3.0) Wound Infection 22 (6.0) 17 (4.6) ALT Increased 19 (5.2) 11 (3.0) AST Increased 17 (4.6) 12 (3.3) Diarrhea 15 (4.1) 10 (2.7) Headache 13 (3.5) 8 (2.2) Cellulitis 12 (3.3) 9 (2.5) Abdominal Pain Upper 10 (2.7) 4 (1.1) 4/22/ a TEAE is defined as an AE occurring after first dose of active test article; b 1 omadacycline subject discontinued due to nausea / vomiting C Day 1 & 2 nausea (25.3%) and vomiting (12.5%), Day 3 through EOT nausea (4.1%) and vomiting (4.1%)
8 OASIS-2 Study Conclusions In the phase 3 OASIS-2 trial, omadacycline was clinically effective and non-inferior to linezolid for treatment of ABSSSI Met both FDA and EMA primary endpoints High clinical response rates for the most common ABSSSI pathogens, including MRSA Omadacycline was safe and generally well-tolerated Nausea and vomiting TEAEs were more frequent during the first two days of the OASIS- 2 oral study, when the subjects received a loading dose of OMC (450 mg) Oral Omadacycline Is Clinically Effective as a Once-daily Oral Antibiotic With Activity Against the Most Common ABSSSI Pathogens Including MRSA 4/22/2018 8
9 Acknowledgments and Disclosures William O Riordan, MD 1 Carrie Cardenas, MD 1 Alissa Sirbu, BSN 2 Lynne Garrity-Ryan, PhD 2 Anita Das, PhD 2 Paul Eckburg, MD 2 Amy Manley, BS 2 Judith Steenburgen, PhD 2 Evan Tzanis, BA 2 Paul McGovern, MD 2 Evan Loh, MD 2 Special thanks to the patients and investigators who participated in this study This study was sponsored by Paratek Pharmaceuticals, Inc. 1 estudysite, San Diego, CA, USA. 2 Paratek Pharmaceuticals, Inc., King of Prussia, PA, USA. 4/22/2018 9
Comparison of Omadacycline and Tigecycline Pharmacodynamics in the Plasma, Epithelial Lining Fluid, and Alveolar Macrophages in Healthy Subjects
 Comparison of Omadacycline and Tigecycline Pharmacodynamics in the Plasma, Epithelial Lining Fluid, and Alveolar Macrophages in Healthy Subjects Karolyn S. Horn, Mark H. Gotfried, Judith N. Steenbergen,
Comparison of Omadacycline and Tigecycline Pharmacodynamics in the Plasma, Epithelial Lining Fluid, and Alveolar Macrophages in Healthy Subjects Karolyn S. Horn, Mark H. Gotfried, Judith N. Steenbergen,
ESTABLISH 2 Top Line Data Release
 The Future of Anti-Infectives ESTABLISH 2 Top Line Data Release March 25, 2013 1 Forward Looking Statements Statements contained in this data release regarding matters that are not historical facts are
The Future of Anti-Infectives ESTABLISH 2 Top Line Data Release March 25, 2013 1 Forward Looking Statements Statements contained in this data release regarding matters that are not historical facts are
Cellceutix Corporation Beverly, MA USA Abstract 2969; Presentation 0195; Hall J, 4:00pm. April 27, 2015
 ECCMID 2015 Copenhagen, Denmark 25 28 April 2015 Cellceutix Corporation Beverly, MA USA www.cellceutix.com A Randomized, Double-Blind Study Comparing Single-Dose and Short-Course Brilacidin to Daptomycin
ECCMID 2015 Copenhagen, Denmark 25 28 April 2015 Cellceutix Corporation Beverly, MA USA www.cellceutix.com A Randomized, Double-Blind Study Comparing Single-Dose and Short-Course Brilacidin to Daptomycin
1 University of Mississippi School of Pharmacy, Department of Pharmacy Practice, Jackson, MS
 DR KATIE ELIZABETH BARBER (Orcid ID : 0000-0002-8411-3111) DR JAMIE L. WAGNER (Orcid ID : 0000-0002-6006-7894) DR KAYLA R STOVER (Orcid ID : 0000-0002-8635-0137) Article type : Review of Therapeutics Omadacycline
DR KATIE ELIZABETH BARBER (Orcid ID : 0000-0002-8411-3111) DR JAMIE L. WAGNER (Orcid ID : 0000-0002-6006-7894) DR KAYLA R STOVER (Orcid ID : 0000-0002-8635-0137) Article type : Review of Therapeutics Omadacycline
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 18 October 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 18 October 2006 CUBICIN 350 mg (daptomycin), powder for perfusion solution Box of 1 bottle (CIP code: 567 219-3) CUBICIN
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 18 October 2006 CUBICIN 350 mg (daptomycin), powder for perfusion solution Box of 1 bottle (CIP code: 567 219-3) CUBICIN
Effect of Food on the Bioavailability of Omadacycline in Healthy Participants
 Pharmacokinetics Effect of Food on the Bioavailability of Omadacycline in Healthy Participants The Journal of Clinical Pharmacology 2017, 57(3) 321 327 C 2016 The Authors.The Journal of Clinical Pharmacology
Pharmacokinetics Effect of Food on the Bioavailability of Omadacycline in Healthy Participants The Journal of Clinical Pharmacology 2017, 57(3) 321 327 C 2016 The Authors.The Journal of Clinical Pharmacology
ESCMID Online Lecture Library. by author
 Hospital Universitario Virgen Macarena, Seville New drugs against MRSA and VRE L. Eduardo López Cortés Seville, 8th July Tedizolid Oxazolidinone Ceftaroline // Ceftobiprole 5 th gen cephalosporin Overview
Hospital Universitario Virgen Macarena, Seville New drugs against MRSA and VRE L. Eduardo López Cortés Seville, 8th July Tedizolid Oxazolidinone Ceftaroline // Ceftobiprole 5 th gen cephalosporin Overview
Open Forum Infectious Diseases MAJOR ARTICLE
 Open Forum Infectious Diseases MAJOR ARTICLE Efficacy and Safety of Oritavancin Relative to Vancomycin for Patients with Acute Bacterial Skin and Skin Structure Infections (ABSSSI) in the Outpatient Setting:
Open Forum Infectious Diseases MAJOR ARTICLE Efficacy and Safety of Oritavancin Relative to Vancomycin for Patients with Acute Bacterial Skin and Skin Structure Infections (ABSSSI) in the Outpatient Setting:
Options for Complicated Skin and Skin Structure Infections. Andrew F. Shorr, MD, MPH Washington Hospital Center Georgetown Univ.
 Options for Complicated Skin and Skin Structure Infections Andrew F. Shorr, MD, MPH Washington Hospital Center Georgetown Univ. Disclosures I have served as a consultant to, researcher/investigator for,
Options for Complicated Skin and Skin Structure Infections Andrew F. Shorr, MD, MPH Washington Hospital Center Georgetown Univ. Disclosures I have served as a consultant to, researcher/investigator for,
XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED.
 XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED. SAFE HARBOR DISCLAIMER These slides and the accompanying oral
XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED. SAFE HARBOR DISCLAIMER These slides and the accompanying oral
Rezafungin: A Novel Echinocandin. Taylor Sandison, MD MPH Chief Medical Officer ISHAM- Amsterdam July 2, 2018
 : A Novel Echinocandin Taylor Sandison, MD MPH Chief Medical Officer ISHAM- Amsterdam July 2, 2018 Disclosures Dr. Sandison is an employee of and stockholder in Cidara Therapeutics. Cidara Pipeline Program
: A Novel Echinocandin Taylor Sandison, MD MPH Chief Medical Officer ISHAM- Amsterdam July 2, 2018 Disclosures Dr. Sandison is an employee of and stockholder in Cidara Therapeutics. Cidara Pipeline Program
ANTIMICROBIALS AVAILABLE FOR
 ORIGINAL CONTRIBUTION Tedizolid Phosphate vs Linezolid for Treatment of Acute Bacterial Skin and Skin Structure Infections The ESTABLISH-1 Randomized Trial Philippe Prokocimer, MD Carisa De Anda, PharmD
ORIGINAL CONTRIBUTION Tedizolid Phosphate vs Linezolid for Treatment of Acute Bacterial Skin and Skin Structure Infections The ESTABLISH-1 Randomized Trial Philippe Prokocimer, MD Carisa De Anda, PharmD
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response
 Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response Blanca Serrano, 1 Silvia Delgado-Aros, 2 Fermín Mearin, 3 Constanza
Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response Blanca Serrano, 1 Silvia Delgado-Aros, 2 Fermín Mearin, 3 Constanza
I. Kalfus MD, D. Riff MD, R. Fathi PhD, D. Graham MD
 A Randomized Double Blind Placebo Controlled Phase III Study to Assess the Safety and Efficacy of Rifabutin Triple Therapy (RHB-105) for Helicobacter pylori (H. pylori) Infection in Dyspepsia Patients
A Randomized Double Blind Placebo Controlled Phase III Study to Assess the Safety and Efficacy of Rifabutin Triple Therapy (RHB-105) for Helicobacter pylori (H. pylori) Infection in Dyspepsia Patients
HTX-011, a Proprietary, Unique, Long-Acting Local Anesthetic, Reduces Acute Postoperative Pain Intensity and Opioid Consumption Following Bunionectomy
 HTX-011, a Proprietary, Unique, Long-Acting Local Anesthetic, Reduces Acute Postoperative Pain Intensity and Opioid Consumption Following Bunionectomy Eugene Viscusi, 1 Oscar DeLeon-Casasola, 2 TJ Gan,
HTX-011, a Proprietary, Unique, Long-Acting Local Anesthetic, Reduces Acute Postoperative Pain Intensity and Opioid Consumption Following Bunionectomy Eugene Viscusi, 1 Oscar DeLeon-Casasola, 2 TJ Gan,
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
Xerava (eravacycline) NEW PRODUCT SLIDESHOW
 Xerava (eravacycline) NEW PRODUCT SLIDESHOW Introduction Brand name: Xerava Generic name: Eravacycline Pharmacological class: Tetracycline antibiotic Strength and Formulation: 50mg; per vial; lyophilized
Xerava (eravacycline) NEW PRODUCT SLIDESHOW Introduction Brand name: Xerava Generic name: Eravacycline Pharmacological class: Tetracycline antibiotic Strength and Formulation: 50mg; per vial; lyophilized
LVHN Scholarly Works. Lehigh Valley Health Network. Nelson Kopyt DO, FASN, FACP Lehigh Valley Health Network,
 Lehigh Valley Health Network LVHN Scholarly Works Department of Medicine Efficacy and Safety of Oral Ferric Maltol (FM) in Treating Iron-Deficiency Anemia (IDA) in Patients with Chronic Kidney Disease
Lehigh Valley Health Network LVHN Scholarly Works Department of Medicine Efficacy and Safety of Oral Ferric Maltol (FM) in Treating Iron-Deficiency Anemia (IDA) in Patients with Chronic Kidney Disease
A randomised, double-blind, parallel group, multicentre study to compare the tolerability, safety, and efficacy of oxycodone with morphine in
 A randomised, double-blind, parallel group, multicentre study to compare the tolerability, safety, and efficacy of oxycodone with morphine in patients using i.v. patient-controlled analgesia (PCA) for
A randomised, double-blind, parallel group, multicentre study to compare the tolerability, safety, and efficacy of oxycodone with morphine in patients using i.v. patient-controlled analgesia (PCA) for
ABSSSI Case Study days (clinical evaluation)
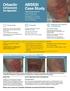 ABSSSI Case Study Cellulitis patient background 44-year-old single father Chronic renal insufficiency Hyperlipidemia, on rosuvastatin Has atrial fibrillation, on apixaban Fell and injured right leg At
ABSSSI Case Study Cellulitis patient background 44-year-old single father Chronic renal insufficiency Hyperlipidemia, on rosuvastatin Has atrial fibrillation, on apixaban Fell and injured right leg At
Cubicin (Daptomycin) Priv.Doz. Dr. med. Markus Rothenburger
 Cubicin (Daptomycin) Priv.Doz. Dr. med. Markus Rothenburger Cubicin (Daptomycin): A new generation of antibiotics First-in-class cyclic natural lipopeptide Activity against major gram positive pathogens
Cubicin (Daptomycin) Priv.Doz. Dr. med. Markus Rothenburger Cubicin (Daptomycin): A new generation of antibiotics First-in-class cyclic natural lipopeptide Activity against major gram positive pathogens
Study No: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Results. Subject Disposition and Demographics Of 37 enrolled subjects, 23 (62.2%) completed the study
 Poster 5-20 Effect of Gastric ph on the Bioavailability of in Healthy Subjects James Longstreth, PhD, Marijke H. Adams, PharmD, PhD, 2 Vasi Sperry, PhD, 3 Dan Kajdasz, PhD, 3 Carol R. Reed, MD 3 Longstreth
Poster 5-20 Effect of Gastric ph on the Bioavailability of in Healthy Subjects James Longstreth, PhD, Marijke H. Adams, PharmD, PhD, 2 Vasi Sperry, PhD, 3 Dan Kajdasz, PhD, 3 Carol R. Reed, MD 3 Longstreth
Antibiotics to treat multi-drug-resistant bacterial infections
 Antibiotics to treat multi-drug-resistant bacterial infections July 2013 JUNE 2013 Copyright 2013 Tetraphase Pharmaceuticals, Inc. 1 Forward Looking Statements and Other Important Cautions Any statements
Antibiotics to treat multi-drug-resistant bacterial infections July 2013 JUNE 2013 Copyright 2013 Tetraphase Pharmaceuticals, Inc. 1 Forward Looking Statements and Other Important Cautions Any statements
Sponsor / Company: Sanofi Drug substance(s): insulin glargine (HOE901) According to template: QSD VERSION N 4.0 (07-JUN-2012) Page 1
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Cabozantinib (Cometriq )
 Cabozantinib (Cometriq ) Workshop dose escalation EMA 4/5 Dec 2014 Frans Opdam, internist-clinical pharmacologist Clinical assessor, Dutch Medicines Agency Phase 3 Study XL184-301 (EXAM): Design Cabozantinib
Cabozantinib (Cometriq ) Workshop dose escalation EMA 4/5 Dec 2014 Frans Opdam, internist-clinical pharmacologist Clinical assessor, Dutch Medicines Agency Phase 3 Study XL184-301 (EXAM): Design Cabozantinib
Safety of Dalbavancin in the Treatment of Skin and Skin Structure Infections: A Pooled Analysis of Randomized, Comparative Studies
 Drug Saf (2016) 39:147 157 DOI 10.1007/s40264-015-0374-9 ORIGINAL RESEARCH ARTICLE Safety of Dalbavancin in the Treatment of Skin and Skin Structure Infections: A Pooled Analysis of Randomized, Comparative
Drug Saf (2016) 39:147 157 DOI 10.1007/s40264-015-0374-9 ORIGINAL RESEARCH ARTICLE Safety of Dalbavancin in the Treatment of Skin and Skin Structure Infections: A Pooled Analysis of Randomized, Comparative
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Lasmiditan (200 mg and 100 mg) Compared to Placebo for Acute Treatment of Migraine
 (200 mg and 100 mg) Compared to for Acute Treatment of Migraine Bernice Kuca, M.S. 1 ; Linda A. Wietecha, B.S.N., M.S. 2 ; Paul H. Berg, M.S. 2 ; Sheena K. Aurora, M.D. 2 1 CoLucid Pharmaceuticals, Inc.,
(200 mg and 100 mg) Compared to for Acute Treatment of Migraine Bernice Kuca, M.S. 1 ; Linda A. Wietecha, B.S.N., M.S. 2 ; Paul H. Berg, M.S. 2 ; Sheena K. Aurora, M.D. 2 1 CoLucid Pharmaceuticals, Inc.,
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
This presentation contains forward looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 relating to business,
 American Society of Nephrology (ASN) Migalastat Monotherapy Phase 3 Data N November b 15 15, 2014 Safe Harbor This presentation contains forward looking statements within the meaning of the Private Securities
American Society of Nephrology (ASN) Migalastat Monotherapy Phase 3 Data N November b 15 15, 2014 Safe Harbor This presentation contains forward looking statements within the meaning of the Private Securities
Sponsor / Company: Sanofi Drug substance(s): AMARYL M (1/250 mg) / HOE490
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
Development of a protease inhibitor-based single-tablet complete HIV-1 regimen of darunavir/cobicistat/emtricitabine/tenofovir
 Development of a protease inhibitor-based single-tablet complete HIV-1 regimen of darunavir/cobicistat/emtricitabine/tenofovir alafenamide () Bryan Baugh 1, Erika Van Landuyt 2, Simon Vanveggel 2, Herta
Development of a protease inhibitor-based single-tablet complete HIV-1 regimen of darunavir/cobicistat/emtricitabine/tenofovir alafenamide () Bryan Baugh 1, Erika Van Landuyt 2, Simon Vanveggel 2, Herta
Enhanced EARS-Net Surveillance 2017 First Half
 1 Enhanced EARS-Net Surveillance 2017 First Half In this report Main results for 2017, first half Breakdown of factors by organism and resistance subtype Device-association Data quality assessment Key
1 Enhanced EARS-Net Surveillance 2017 First Half In this report Main results for 2017, first half Breakdown of factors by organism and resistance subtype Device-association Data quality assessment Key
ICD-10 diagnostic codes consistent with ORBACTIV (oritavancin) indication
 ICD-10 diagnostic codes consistent with ORBACTIV (oritavancin) indication ICD-10-CM Diagnosis Codes Diagnosis INDICATION ORBACTIV (oritavancin) for injection is indicated for the treatment of adult patients
ICD-10 diagnostic codes consistent with ORBACTIV (oritavancin) indication ICD-10-CM Diagnosis Codes Diagnosis INDICATION ORBACTIV (oritavancin) for injection is indicated for the treatment of adult patients
Triptans: Nonresponse, Recurrence, and Serious AEs for Many Patients
 Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Efficacy, Safety and Tolerability of 150 mg Q2W Dose of the PCSK9 mab REGN727/SAR236553: Data from Three Phase 2 Studies
 Efficacy, Safety and Tolerability of 150 mg Q2W Dose of the PCSK9 mab REGN727/SAR236553: Data from Three Phase 2 Studies Michael J. Koren, 1 Evan A. Stein, 2 Eli M. Roth, 3 James M. McKenney, 4 Dan Gipe,
Efficacy, Safety and Tolerability of 150 mg Q2W Dose of the PCSK9 mab REGN727/SAR236553: Data from Three Phase 2 Studies Michael J. Koren, 1 Evan A. Stein, 2 Eli M. Roth, 3 James M. McKenney, 4 Dan Gipe,
Disclosures. REPLENISH Trial: Objective and Design. Background Use of compounded bioidentical hormone therapy (CBHT) has become highly prevalent
 17β Estradiol/Progesterone in a Single Oral Softgel Capsule (TX 001HR) Significantly Reduced Moderate to Severe Vasomotor Symptoms without Endometrial Hyperplasia Disclosures Research support: Actavis,
17β Estradiol/Progesterone in a Single Oral Softgel Capsule (TX 001HR) Significantly Reduced Moderate to Severe Vasomotor Symptoms without Endometrial Hyperplasia Disclosures Research support: Actavis,
Disclosure. This study was sponsored by Pfizer, Inc. All authors are employees of Pfizer, Inc. with ownership of stock in Pfizer, Inc.
 Disclosure This study was sponsored by Pfizer, Inc. All authors are employees of Pfizer, Inc. with ownership of stock in Pfizer, Inc. Effects of 12 Weeks of Treatment with RN316 (PF-04950615), a Humanized
Disclosure This study was sponsored by Pfizer, Inc. All authors are employees of Pfizer, Inc. with ownership of stock in Pfizer, Inc. Effects of 12 Weeks of Treatment with RN316 (PF-04950615), a Humanized
TP Larry Tsai, MD Chief Medical Officer Tetraphase Pharmaceuticals
 TP-676 Larry Tsai, MD Chief Medical Officer Tetraphase Pharmaceuticals Tetraphase Pharmaceuticals Overview Tetraphase is developing novel antibiotics for serious and life-threatening Gram-negative MDR
TP-676 Larry Tsai, MD Chief Medical Officer Tetraphase Pharmaceuticals Tetraphase Pharmaceuticals Overview Tetraphase is developing novel antibiotics for serious and life-threatening Gram-negative MDR
Columbia University Medical Center, New York, NY 2. Clinical Research Center, Eastern Virginia Medical School, Norfolk, VA 3
 17β-Estradiol/Progesterone in a Single Oral Softgel Capsule (TX-001HR) Significantly Reduced Moderate-to-Severe Vasomotor Symptoms without Endometrial Hyperplasia Rogerio A Lobo, MD 1 ; David F Archer,
17β-Estradiol/Progesterone in a Single Oral Softgel Capsule (TX-001HR) Significantly Reduced Moderate-to-Severe Vasomotor Symptoms without Endometrial Hyperplasia Rogerio A Lobo, MD 1 ; David F Archer,
Daniel Canafax, PharmD VP, Clinical Research Theravance, Inc.
 Demonstrates Improvement in Bowel Movement Frequency and Bristol Stool Scores in a Phase 2b Study of Patients with Opioid-Induced Constipation (OIC) Ross Vickery, PhD, 1 Yu-Ping Li, PhD, 1 Ullrich Schwertschlag,
Demonstrates Improvement in Bowel Movement Frequency and Bristol Stool Scores in a Phase 2b Study of Patients with Opioid-Induced Constipation (OIC) Ross Vickery, PhD, 1 Yu-Ping Li, PhD, 1 Ullrich Schwertschlag,
This was a randomized, double-blind, placebo-controlled, fixed-dose, parallel-group study.
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Oral Methylnaltrexone for the. Constipation in Patients with Chronic Non-cancer Pain
 Oral Methylnaltrexone for the Treatment of Opioid-induced Constipation in Patients with Chronic Non-cancer Pain Richard L. Rauck, 1 John F. Peppin, 2 Robert J.Israel, 3 Jennifer Carpenito, 3 Jeffrey Cohn,
Oral Methylnaltrexone for the Treatment of Opioid-induced Constipation in Patients with Chronic Non-cancer Pain Richard L. Rauck, 1 John F. Peppin, 2 Robert J.Israel, 3 Jennifer Carpenito, 3 Jeffrey Cohn,
Micafungin, a new Echinocandin: Pediatric Development
 Micafungin, a new Echinocandin: Pediatric Development Andreas H. Groll, M.D. Infectious Disease Research Program Center for Bone Marrow Transplantation and Department of Pediatric Hematology/Oncology University
Micafungin, a new Echinocandin: Pediatric Development Andreas H. Groll, M.D. Infectious Disease Research Program Center for Bone Marrow Transplantation and Department of Pediatric Hematology/Oncology University
Sponsor / Company: Sanofi Drug substance(s): Insulin Glargine (HOE901) Insulin Glulisine (HMR1964)
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Challenges of HABP/VABP Trials
 Challenges of HABP/VABP Trials CTTI - 22 April, 2013 H. David Friedland, MD Cerexa Inc., a wholly owned subsidiary of Forest Laboratories Disclosures Employee of Cerexa, a wholly owned subsidiary of Forest
Challenges of HABP/VABP Trials CTTI - 22 April, 2013 H. David Friedland, MD Cerexa Inc., a wholly owned subsidiary of Forest Laboratories Disclosures Employee of Cerexa, a wholly owned subsidiary of Forest
(b) (4) B. Recommendation on Phase 4 Studies and/or Risk Management Steps
 1 REVIEW AND EVALUATION OF CLINCIAL DATA NDA 20-822 (b) (4) 016 Sponsor: Forest Labs Drug: Celexa. (citalopram hydrocloride) Material Submitted: Pediatric Supplement (b) (4) 016 Date Submitted: 4/18/02
1 REVIEW AND EVALUATION OF CLINCIAL DATA NDA 20-822 (b) (4) 016 Sponsor: Forest Labs Drug: Celexa. (citalopram hydrocloride) Material Submitted: Pediatric Supplement (b) (4) 016 Date Submitted: 4/18/02
The results of the ARTEN study. Vicente Soriano Hospital Carlos III, Madrid, Spain
 The results of the ARTEN study Vicente Soriano Hospital Carlos III, Madrid, Spain Nevirapine: a well-defined efficacy and tolerability profile High efficacy levels 1 3 Well-defined safety profile 4 Favourable
The results of the ARTEN study Vicente Soriano Hospital Carlos III, Madrid, Spain Nevirapine: a well-defined efficacy and tolerability profile High efficacy levels 1 3 Well-defined safety profile 4 Favourable
ICVH 2016 Oral Presentation: 28
 Ledipasvir/Sofosbuvir Is Safe and Effective for the Treatment of Patients with Genotype 1 Chronic HCV Infection in Both HCV Mono- and HIV/HCV Coinfected Patients A Luetkemeyer 1, C Cooper 2, P Kwo 3, K
Ledipasvir/Sofosbuvir Is Safe and Effective for the Treatment of Patients with Genotype 1 Chronic HCV Infection in Both HCV Mono- and HIV/HCV Coinfected Patients A Luetkemeyer 1, C Cooper 2, P Kwo 3, K
ALTARGO TM Retapamulin. QUALITATIVE AND QUANTITATIVE COMPOSITION Retapamulin 10 mg per gram (1% w/w)
 ALTARGO TM Retapamulin QUALITATIVE AND QUANTITATIVE COMPOSITION Retapamulin 10 mg per gram (1% w/w) PHARMACEUTICAL FORM Ointment. CLINICAL PARTICULARS Indications ALTARGO (retapamulin) 1% ointment is indicated
ALTARGO TM Retapamulin QUALITATIVE AND QUANTITATIVE COMPOSITION Retapamulin 10 mg per gram (1% w/w) PHARMACEUTICAL FORM Ointment. CLINICAL PARTICULARS Indications ALTARGO (retapamulin) 1% ointment is indicated
Study Centers: This study was conducted in 2 centers in Italy.
 Title of Trial: A randomised, double-blind, placebo-controlled, two-period, two-sequence-crossover interaction study to assess the effect of safinamide on levodopa pharmacokinetics in subjects with Parkinson
Title of Trial: A randomised, double-blind, placebo-controlled, two-period, two-sequence-crossover interaction study to assess the effect of safinamide on levodopa pharmacokinetics in subjects with Parkinson
XERAVATM (eravacycline): A Novel Fluorocycline Antibacterial
 XERAVATM (eravacycline): A Novel Fluorocycline Antibacterial Matteo Bassetti, MD, PhD Infectious Diseases Division University of Udine and Santa Maria Misericordia University Hospital Udine, Italy Disclosures
XERAVATM (eravacycline): A Novel Fluorocycline Antibacterial Matteo Bassetti, MD, PhD Infectious Diseases Division University of Udine and Santa Maria Misericordia University Hospital Udine, Italy Disclosures
Development of Drugs for Bacteremia
 Development of Drugs for Bacteremia Charles Knirsch, MD, MPH VP, Clinical Research Pfizer Inc 1 Bacteremia Guidance Issues EMA guidance suggests that bacteremia is not a primary diagnosis but represents
Development of Drugs for Bacteremia Charles Knirsch, MD, MPH VP, Clinical Research Pfizer Inc 1 Bacteremia Guidance Issues EMA guidance suggests that bacteremia is not a primary diagnosis but represents
Linezolid - Tigecycline
 Linezolid - Tigecycline Paul M. Tulkens, MD, PhD Cellular and Molecular Pharmacology Louvain Drug Research Institute Catholic University of Louvain, Brussels, Belgium With the support of Wallonie-Bruxelles-International
Linezolid - Tigecycline Paul M. Tulkens, MD, PhD Cellular and Molecular Pharmacology Louvain Drug Research Institute Catholic University of Louvain, Brussels, Belgium With the support of Wallonie-Bruxelles-International
Pitavastatin 4 mg vs. Pravastatin 40 mg in HIV Dyslipidemia: Post- Hoc Analysis of the INTREPID Trial Based on the Independent CHD Risk Factor for Age
 Pitavastatin 4 mg vs. Pravastatin 40 mg in HIV Dyslipidemia: Post- Hoc Analysis of the INTREPID Trial Based on the Independent CHD Risk Factor for Age Craig A. Sponseller, Masaya Tanahashi, Hideki Suganami,
Pitavastatin 4 mg vs. Pravastatin 40 mg in HIV Dyslipidemia: Post- Hoc Analysis of the INTREPID Trial Based on the Independent CHD Risk Factor for Age Craig A. Sponseller, Masaya Tanahashi, Hideki Suganami,
David Y. Graham, MD Baylor College of Medicine, Houston TX, USA
 Phase III Randomized, Double Blind, Placebo- Controlled, Multicenter, Parallel Group Study to Assess the Efficacy and Safety of Add-On Fixed-Dose Anti- Mycobacterial Therapy (RHB-104) in Moderately to
Phase III Randomized, Double Blind, Placebo- Controlled, Multicenter, Parallel Group Study to Assess the Efficacy and Safety of Add-On Fixed-Dose Anti- Mycobacterial Therapy (RHB-104) in Moderately to
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See United States Package Insert (USPI)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
ABC/3TC/ZDV ABC PBO/3TC/ZDV
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Sponsor / Company: Sanofi Drug substance(s): SAR302503
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 Study No.: 29060/717 Title: A Double-Blind, Placebo-Controlled, 3-Arm, Fixed-Dose Study of CR Intermittent Dosing (12.5 mg and 25 mg) for Premenstrual Dysphoric Disorder Rationale: In most trials investigating
Study No.: 29060/717 Title: A Double-Blind, Placebo-Controlled, 3-Arm, Fixed-Dose Study of CR Intermittent Dosing (12.5 mg and 25 mg) for Premenstrual Dysphoric Disorder Rationale: In most trials investigating
PFIZER INC. Study Initiation Date and Completion Dates: 09 March 2000 to 09 August 2001.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Analysis of the Phase 3 ESTABLISH Trials: Tedizolid Versus Linezolid in Acute Bacterial Skin
 AAC Accepts, published online ahead of print on 24 November 2014 Antimicrob. Agents Chemother. doi:10.1128/aac.03688-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 Analysis
AAC Accepts, published online ahead of print on 24 November 2014 Antimicrob. Agents Chemother. doi:10.1128/aac.03688-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 Analysis
PFIZER INC. Study Initiation Date and Completion Dates: Information not available (Date of Statistical Report: 16 May 2004)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
IRBES_R_04320 Generic drug name: Irbesartan + amlodipine Date: Study Code: 31 active centers: 20 in Korea, 7 in India and 4 in Philippines.
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
Emricasan (IDN-6556) administered orally for 28 days lowers portal pressure in patients with compensated cirrhosis and severe portal hypertension
 Emricasan (IDN-6556) administered orally for 28 days lowers portal pressure in patients with compensated cirrhosis and severe portal hypertension Guadalupe Garcia-Tsao, Michael Fuchs, Mitchell Shiffman,
Emricasan (IDN-6556) administered orally for 28 days lowers portal pressure in patients with compensated cirrhosis and severe portal hypertension Guadalupe Garcia-Tsao, Michael Fuchs, Mitchell Shiffman,
Comparison of Omadacycline and Tigecycline Pharmacokinetics in the. Plasma, Epithelial Lining Fluid, and Alveolar Cells in Healthy Adult Subjects
 AAC Accepted Manuscript Posted Online 10 July 2017 Antimicrob. Agents Chemother. doi:10.1128/aac.01135-17 Copyright 2017 Gotfried et al. This is an open-access article distributed under the terms of the
AAC Accepted Manuscript Posted Online 10 July 2017 Antimicrob. Agents Chemother. doi:10.1128/aac.01135-17 Copyright 2017 Gotfried et al. This is an open-access article distributed under the terms of the
HIGHLIGHTS ESMO 2017 SUPPORTIVE AND PALLIATIVE CARE
 HIGHLIGHTS ESMO 2017 SUPPORTIVE AND PALLIATIVE CARE Florian Scotté MD-PhD Hôpital Foch, Suresnes, France esmo.org DISCLOSURE SLIDE Consultant / Advisory Boards / Speaker: Tesaro, Sanofi, Roche, MSD, TEVA,
HIGHLIGHTS ESMO 2017 SUPPORTIVE AND PALLIATIVE CARE Florian Scotté MD-PhD Hôpital Foch, Suresnes, France esmo.org DISCLOSURE SLIDE Consultant / Advisory Boards / Speaker: Tesaro, Sanofi, Roche, MSD, TEVA,
CAM2038 A new liquid-lipid crystal depot buprenorphine: A dose-ranging suite of weekly and monthly subcutaneous depot injections
 CAM2038 A new liquid-lipid crystal depot buprenorphine: A dose-ranging suite of weekly and monthly subcutaneous depot injections Dr. Fredrik Tiberg Assoc. Prof. President & CEO, Head R&D, Camurus Lund,
CAM2038 A new liquid-lipid crystal depot buprenorphine: A dose-ranging suite of weekly and monthly subcutaneous depot injections Dr. Fredrik Tiberg Assoc. Prof. President & CEO, Head R&D, Camurus Lund,
Sponsor: Sanofi Drug substance(s): GZ316455
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor: Sanofi Drug substance(s):
Tedizolid: a novel treatment for Gram + infections and its potential role in clinical practice
 Tedizolid: a novel treatment for Gram + infections and its potential role in clinical practice Paul M. Tulkens, MD, PhD Cellular and Molecular Pharmacology & Centre for Clinical Pharmacy Louvain Drug Research
Tedizolid: a novel treatment for Gram + infections and its potential role in clinical practice Paul M. Tulkens, MD, PhD Cellular and Molecular Pharmacology & Centre for Clinical Pharmacy Louvain Drug Research
TOUGH ON IMPETIGO GENTLE ON THE1 PATIENT. Demonstrated efficacy and safety1. New treatment option now available!
 New treatment option now available! TOUGH ON IMPETIGO GENTLE ON THE1 PATIENT Demonstrated efficacy and safety1 Home Efficacy Safety Convenience (ozenoxacin) is indicated for the topical treatment of impetigo
New treatment option now available! TOUGH ON IMPETIGO GENTLE ON THE1 PATIENT Demonstrated efficacy and safety1 Home Efficacy Safety Convenience (ozenoxacin) is indicated for the topical treatment of impetigo
Glecaprevir-Pibrentasvir in Cirrhotic Genotype 1, 2, 4, 5, and 6 EXPEDITION-1
 Phase 3 Treatment-Naïve and Treatment-Experienced Glecaprevir-Pibrentasvir in Cirrhotic Genotype 1, 2, 4, 5, and 6 EXPEDITION-1 EXPEDITION-1: Study Features EXPEDITION-1 Trial Design: Open-label, single-arm,
Phase 3 Treatment-Naïve and Treatment-Experienced Glecaprevir-Pibrentasvir in Cirrhotic Genotype 1, 2, 4, 5, and 6 EXPEDITION-1 EXPEDITION-1: Study Features EXPEDITION-1 Trial Design: Open-label, single-arm,
Study No.: ADF Title: Phase III study of adefovir dipivoxil (ADV) tablets in patients with compensated chronic hepatitis B -comparative study
 Study No.: ADF105220 Title: Phase III study of adefovir dipivoxil () tablets in patients with compensated chronic hepatitis B -comparative study against lamivudine ()- Rationale: This study wass a confirmatory
Study No.: ADF105220 Title: Phase III study of adefovir dipivoxil () tablets in patients with compensated chronic hepatitis B -comparative study against lamivudine ()- Rationale: This study wass a confirmatory
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
Perspective on methodological challenges: How Phase 2 studies influence design & conduct of Phase 3
 Perspective on methodological challenges: How Phase 2 studies influence design & conduct of Phase 3 Ilise Lombardo, M.D. Vice President, Clinical Research FORUM Pharmaceuticals 1 Objective: Overview Describe
Perspective on methodological challenges: How Phase 2 studies influence design & conduct of Phase 3 Ilise Lombardo, M.D. Vice President, Clinical Research FORUM Pharmaceuticals 1 Objective: Overview Describe
Tedizolid Phosphate (Sivextro) A Second-Generation Oxazolidinone to Treat Acute Bacterial Skin and Skin Structure Infections
 DrUG FOrECAST Tedizolid Phosphate (Sivextro) A Second-Generation Oxazolidinone to Treat Acute Bacterial Skin and Skin Structure Infections Elaine Wong, PharmD, BCPS; and Saba rab, PharmD, BCPS Dr. Wong
DrUG FOrECAST Tedizolid Phosphate (Sivextro) A Second-Generation Oxazolidinone to Treat Acute Bacterial Skin and Skin Structure Infections Elaine Wong, PharmD, BCPS; and Saba rab, PharmD, BCPS Dr. Wong
Efficacy/pharmacodynamics: 85 Safety: 89
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor/Company: Sanofi Drug substance:
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor/Company: Sanofi Drug substance:
Clinical Endpoint Bioequivalence Study Review in ANDA Submissions. Ying Fan, Ph.D.
 Clinical Endpoint Bioequivalence Study Review in ANDA Submissions Ying Fan, Ph.D. 1 Disclaimer This presentation constitutes an informal communication that represents the best judgment of the speaker at
Clinical Endpoint Bioequivalence Study Review in ANDA Submissions Ying Fan, Ph.D. 1 Disclaimer This presentation constitutes an informal communication that represents the best judgment of the speaker at
Plazomicin Versus Meropenem for the Treatment of Complicated Urinary Tract Infection and Acute Pyelonephritis: Results of the EPIC Study
 Plazomicin Versus Meropenem for the Treatment of Complicated Urinary Tract Infection and Acute Pyelonephritis: Results of the EPIC Study Daniel J. Cloutier 1, Loren G. Miller 2, Allison S. Komirenko 1,
Plazomicin Versus Meropenem for the Treatment of Complicated Urinary Tract Infection and Acute Pyelonephritis: Results of the EPIC Study Daniel J. Cloutier 1, Loren G. Miller 2, Allison S. Komirenko 1,
Summary ID#7029. Clinical Study Summary: Study F1D-MC-HGKQ
 CT Registry ID# 7029 Page 1 Summary ID#7029 Clinical Study Summary: Study F1D-MC-HGKQ Clinical Study Report: Versus Divalproex and Placebo in the Treatment of Mild to Moderate Mania Associated with Bipolar
CT Registry ID# 7029 Page 1 Summary ID#7029 Clinical Study Summary: Study F1D-MC-HGKQ Clinical Study Report: Versus Divalproex and Placebo in the Treatment of Mild to Moderate Mania Associated with Bipolar
Study Code: Date: 27 July 2007
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Generic drug name:
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Generic drug name:
APDW 2016 Poster No. a90312
 APDW 2016 Poster No. a90312 SYN-010, a Proprietary Modified-Release Formulation of Lovastatin Lactone, Lowered Breath Methane and Improved Stool Frequency in Patients with IBS-C Results of a multi-center,
APDW 2016 Poster No. a90312 SYN-010, a Proprietary Modified-Release Formulation of Lovastatin Lactone, Lowered Breath Methane and Improved Stool Frequency in Patients with IBS-C Results of a multi-center,
Treatment of acute bacterial skin and skin structure infection with single-dose dalbavancin in persons who inject drugs
 ORIGINAL RESEARCH A continuous publication, open access, peer-reviewed journal Treatment of acute bacterial skin and skin structure infection with single-dose dalbavancin in persons who inject drugs ACCESS
ORIGINAL RESEARCH A continuous publication, open access, peer-reviewed journal Treatment of acute bacterial skin and skin structure infection with single-dose dalbavancin in persons who inject drugs ACCESS
profiles Decision: Dalvance
 Patient candidate profiles Decision: Dalvance 2 A series of patient candidate profiles The following patient candidate profiles are hypothetical examples of treatment decisions that could be seen in the
Patient candidate profiles Decision: Dalvance 2 A series of patient candidate profiles The following patient candidate profiles are hypothetical examples of treatment decisions that could be seen in the
GSK Medicine: Study Number: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objective:
 GSK Medicine: abacavir (ABC)/dolutegravir (DTG)/lamivudine (3TC) Study Number: 201147 Title: A IIIb, randomized, open-label study of the safety, efficacy, and tolerability of switching to a fixed-dose
GSK Medicine: abacavir (ABC)/dolutegravir (DTG)/lamivudine (3TC) Study Number: 201147 Title: A IIIb, randomized, open-label study of the safety, efficacy, and tolerability of switching to a fixed-dose
Lilly Diabetes: Pipeline Update
 Lilly Diabetes: Pipeline Update June 16, 2014 Safe Harbor Provision This presentation contains forward-looking statements that are based on management's current expectations, but actual results may differ
Lilly Diabetes: Pipeline Update June 16, 2014 Safe Harbor Provision This presentation contains forward-looking statements that are based on management's current expectations, but actual results may differ
Impavido. (miltefosine) New Product Slideshow
 Impavido (miltefosine) New Product Slideshow Introduction Brand name: Impavido Generic name: Miltefosine Pharmacological class: Antileishmanial agent Strength and Formulation: 50mg; hard gel capsules Manufacturer:
Impavido (miltefosine) New Product Slideshow Introduction Brand name: Impavido Generic name: Miltefosine Pharmacological class: Antileishmanial agent Strength and Formulation: 50mg; hard gel capsules Manufacturer:
JAK Inhibitor CTP-543 Achieves Primary Endpoint in Phase 2 Trial in Alopecia Areata An interim analysis of 4 and 8 mg BID
 American Academy of Dermatology Annual Meeting March 2, 2019 JAK Inhibitor CTP-543 Achieves Primary Endpoint in Phase 2 Trial in Alopecia Areata An interim analysis of 4 and 8 mg BID James Cassella, PhD;
American Academy of Dermatology Annual Meeting March 2, 2019 JAK Inhibitor CTP-543 Achieves Primary Endpoint in Phase 2 Trial in Alopecia Areata An interim analysis of 4 and 8 mg BID James Cassella, PhD;
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
Bristol-Myers Squibb
 A Study of the Safety and Efficacy of plus Tenofovir in Adults with Chronic Hepatitis B Virus Infection with Previous Nucleoside/Nucleotide Treatment Failure () FINAL CLINICAL STUDY REPORT EUDRACT Number:
A Study of the Safety and Efficacy of plus Tenofovir in Adults with Chronic Hepatitis B Virus Infection with Previous Nucleoside/Nucleotide Treatment Failure () FINAL CLINICAL STUDY REPORT EUDRACT Number:
Synopsis Style Clinical Study Report SR EFC10139 Version number: 1 (electronic 2.0)
 SYNOPSIS Title of the study: A randomized, double-blind, parallel-group, multicenter, multinational study to assess the long-term effect, over 1 year, of rimonabant 10 mg in comparison with rimonabant
SYNOPSIS Title of the study: A randomized, double-blind, parallel-group, multicenter, multinational study to assess the long-term effect, over 1 year, of rimonabant 10 mg in comparison with rimonabant
Fusidic acid and erythromycin in the treatment of skin and soft tissue infection: a double blind study Wall A R, Menday A P
 Fusidic acid and erythromycin in the treatment of skin and soft tissue infection: a double blind study Wall A R, Menday A P Record Status This is a critical abstract of an economic evaluation that meets
Fusidic acid and erythromycin in the treatment of skin and soft tissue infection: a double blind study Wall A R, Menday A P Record Status This is a critical abstract of an economic evaluation that meets
Skin and soft tissue (SSTI) sepsis (surgery, antimicrobial therapy and more)
 Skin and soft tissue (SSTI) sepsis (surgery, antimicrobial therapy and more) Christian Eckmann Antibiotic Stewardship Expert ECDC Chief of Staff Department of General, Visceral and Thoracic Surgery Klinikum
Skin and soft tissue (SSTI) sepsis (surgery, antimicrobial therapy and more) Christian Eckmann Antibiotic Stewardship Expert ECDC Chief of Staff Department of General, Visceral and Thoracic Surgery Klinikum
