Patient Safety: A Quality System Approach To POCT QC/QA
|
|
|
- Natalie May
- 6 years ago
- Views:
Transcription
1 Patient Safety: A Quality System Approach To POCT QC/QA Ellis Jacobs, Ph.D., DABCC New York University School of Medicine Coler-Goldwater Specialty Hospital & Nursing Facility New York, New York
2 Point-of-Care Testing Characteristics A broad based process. Unrestricted to location, personnel or test menu. A collective, multidisciplinary effort. Simple to use technology Potentially low volume testing
3 POCT versus Central Lab Testing Central Lab POCT Testing personnel Pathologists,, PhDs, Med. Lab Technologists Nurses, other care givers Primary duties Laboratory testing Patient care Knows laboratory testing Extensive Minimal Understands instrument s quality checks Extensive Minimal Can interpret QC data Yes Probably not Skills to resolve problems, troubleshooting Yes Recognizes quality testing Yes Not necessarily No
4 Potential Analytes for POCT Bilirubin Blood Gases BUN Cardiac Markers CBC Chloesterol/Trigs Drugs Fecal Occult Blood Gastric Occult Blood Glucose Gram Stains HgB/Hct HgB A1C Infectious Diseases Lactate Na, K, Ca ++, Cl, Mg ++ O 2 Sat Platelet Function Pregnancy PT/PTT/ACT Urinary microalbumin/creatinine Urinalysis/Specific Gravity
5 Point-of-Care Tests (POCT) NOT considered laboratory testing Breath alcohol Continuous glucose monitors Pulse oximeters Transcutaneous bilirubinometers Ex vivo ABG Biosensor Technologies (monitors)
6 2001 Predicted Growth in POCT 12-16% annual growth Currently 1 in 4 test done by POC In 10 years ~40% by POC Currently $450 million industry In 2025, $950 million industry
7 Actual Growth in POCT 2008 Worldwide IVD Market - $42.1 Billion (46B in 2010) 2008 Worldwide POCT Market - $13.1 Billion (31%) 2010 Worldwide Professional POCT Market - $4 Billion ~10-12% annual growth
8 POC Testing Environments All testing performed at the patient s side Hospital Settings Out-of- Hospital Care Alternate, Home Settings
9 Trends in Healthcare Provision POCT Laboratory Home Primary Care Centre Community Treatment Centre Local Hospital Referral/ Specialist Hospital
10 Moderators of POCT Growth Quality Assurance Quality Control - Matrix/Electronic Regulatory Requirements Record Keeping/Data Management Finances
11 POC Testing Knowledge Flow Health Care Provider Determines Need for Data Data entry into LIS Sample Obtained Sample Processed At POC Sample Received & Processed in Lab Sample Transported To Satellite Lab
12 POCT Quality Assurance Dilemma Due to the rapid availability of results with POCT, data can be seen and acted upon prior to any QC checks or other external mechanism of assuring test results can be applied to these systems.
13 QA Issues With POC Testing Who performs testing and their training Pre-analytical variables and the ability to recognize them Reagent Testing Instrument verification Maintenance requirements Result reporting & charting
14 Quality System Hierarchy TQM Quality Management Quality Systems Quality Assurance Quality Control
15 POCT as a TQM Project Multidisciplinary team approach Looking at entire system, rather than individual performance On-going evaluation & refinement (CQI) Cost savings Improvement in delivery of critical laboratory services
16 Quality Management System Model Laboratory s Path of Worklow Preanalytical Analytical Postanalytical QSEs encompass the entire path
17 What is a Quality System? The quality management system approach applies a core set of quality system essentials (QSEs), basic to any organization, to all operations in any health care service s path of workflow (ie, operational aspects that define how a particular product or service is provided).
18 Quality Service Essentials (QSEs) Facilities & Safety Documents & Records Equipment Customer Service Information Management Process Control Quality System Process Improvement Assessments External & Internal Organization Personnel Occurrence Management Purchasing & Inventory
19 Quality Service Essentials (QSEs) Equipment Process Improvement Facilities & Safety The Lab Organization Customer Service Measurment Occurrence Management Personnel Purchasing & Inventory Documents & Records Assessments External & Internal The Work Process Control Information Management
20 Quality of Health Care in U.S. Institute of Medicine Medical errors cause 44,000 to 98,000 deaths each year» Equivalent to 200 deaths each day in airline crashes» Fifth leading cause of death in U.S. Ahead of diabetes, breast cancer, HIV» Lab testing certainly contributes to deaths Lab is looking for built-in safeguards to prevent errors To Err is Human: Building a Safer Health System. Washington, DC, National Academy Press; 2000
21 Sources of Testing Error Preanalytical 68% 62% Analytical 13% 15% Postanalytical 19% 23% Plebani M, Carraro P, Clin Chem 1997;43: Carraro P, Plebani M, Clin Chem 2007;53;
22 Laboratory Testing Potential Sources of Errors patient Transmit result Record result Prepare request form Phlebotomy doctor Report result Transport sample Validate result Quality control Analyse sample Prepare sample Register sample
23 Potential Impact of POCT on Laboratory Errors Pre-Analytical Patient Identification Specimen Identification Improper result validation (QC) Post-Analytical Routing Excessive turn-around time Analytical Method Calibration Interferences Results out of measurement range Quality Assessment (EQA/PT)
24 POCT & Patient Safety: Quality Testing Criteria Correct test ordered Correct patient Correct time for collection Correct specimen and processing Correct (accurate) test result Correct patient record Correct clinical interpretation of POCT result(s) Correct and timely clinical response
25 Best Practices for Glucose POCT Positive Patient ID- two identifiers Operator Certification Regular Calibration & QC Use Fresh Reagents Prevent Reagent Contamination Prevent Substance Interference Prevent Blood Sampling Errors
26 Evolution of POCT Manual Automation A process or system operating automatically Autonomation Intelligent automation detects single defective operation and automatically stops Ehrmeyer S, Lassig R. Clin Chem Lab Med 2007;45(6):
27 Managing Sources of POCT Errors Designed out of the product Tested for Warned about
28 Evolution of Glucose POCT Technology Manual Testing Incorrect sample amount Incorrect reagent amount Incorrect mixing Wrong position of testing device Wrong wait time Color blindness
29 Evolution of Glucose POCT Technology Manual Methods 1 st /2 nd Generation Instruments Wipe/Wipeless technology Operator ID / Patient ID Reduced operator intervention Operator prompts Check on reagent viability QC lock-outs Rudimentary Data Management
30 Evolution of Glucose POCT Technology Manual Tests 1 st /2 nd Generation Instruments Current Technology Electrochemical Technology Ability to use universal specimen types Extended linearity Minimally Invasive Technology ( <3 ul Sample Size) Consolidated Testing Platforms Real Time Data Management and Connectivity
31 Precision PcX Reduces Interference Risk Glucose-specific strip technology Minimizes interference from many nonglucose substances in the blood. Patient safe for patients undergoing peritoneal dialysis using Extraneal (icodextrin). Individually foil wrapped and bar-coded strips - reduces risk of contamination and helps assure fresh reagents for each test. Reduces Risk of Sampling Errors Test begins when adequate sample is detected, reducing risk of short-sampling and over-sampling errors
32 safepico Blood Gas Syringe Pre-barcoded arterial syringe for positive patient identification Establishes and Maintains Sample ID throughout testing process
33 Unit use and POCT devices It is often suggested that QC has no role in a unit use device because QC of a single unit (good or bad result) does not inform about other units [same argument would apply to non POCT analyzers in main lab that use discrete (unit use) reagent packs] IMS fulfills QC role in unit use devices Unit use and continuous flow systems are not that different
34 Characteristics of Unit-Use Test The container where the test is performed is always discarded after each test. Reagents, calibrators, and wash solutions are typically segregated as one test. There is no interaction of reagents, calibrators, and wash solutions from test to test.
35 Nature of QC Procedures Use of electronic checks, including any instrument software features that serve as error detection or prevention mechanisms Use and number of surrogate samples, where appropriate, to be included as part of the QC procedure Testing of controls that are engineered into the test system
36 Abaxis Piccolo
37 Triage Cardiac Markers
38 Surrogate QC doesn t detect all errors
39 Non-Surrogate Sample QC Includes all forms of quality control other than the measurement of a surrogate sample, usually integrated into the device electronic QC (which simulates signals electronically), ex. i-stat automated procedural controls (which ensure that certain steps of the procedure occur appropriately), ex. Immunochromatography test kits automated internal quality controls (which may, for example, ensure the quality of a raw signal), ex. diagnostic pattern recognition systems, ex. GEM iqm
40 Immunochromatography Urine Dipstick
41 Gem Premier 4000 Continuously monitors all critical components of blood gas testing in real time to assure accurate results Automatically assures that each test meets demanding quality specifications Immediately detects, corrects and documents errors Eliminates labor and material costs associated with traditional QC Assures that optimal quality control protocols are followed at all times, regardless of operator training
42 Internal monitoring systems (IMS) IMS are a collection of hardware and software that detect errors and prevent the effect of the error from occurring Example: Noise in the signal of a patient sample is detected, the result is flagged and not reported IMS are not new although always improved, they have been in systems for over 30 years
43 Internal monitoring systems Internal monitoring systems don t detect all errors, because Complexity of instrument systems prevents perfect failure mode models There is management pressure to release new products quickly There is insufficient knowledge to design things right the first time
44 Non-Surrogate QC and QC Surrogate and Non-Surrogate QC are not completely redundant do not detect all errors Surrogate QC Non Surrogate QC
45 Critical Factors in QC Decisions
46 Critical Factors in QC Decisions QC must be able to detect mistakes to enable immediate correction
47 Critical Factors in QC Decisions QC must be able to detect mistakes to enable immediate correction Risks and costs must be weighed
48 Critical Factors in QC Decisions QC must be able to detect mistakes to enable immediate correction Risks and costs must be weighed QC is only one part of the quality management system
49 Critical Factors in QC Decisions QC must be able to detect mistakes to enable immediate correction Risks and costs must be weighed QC is only one part of the quality management system Not all laboratories have the same competencies and organization
50 Critical Factors in QC Decisions QC must be able to detect mistakes to enable immediate correction Risks and costs must be weighed QC is only one part of the quality management system Not all laboratories have the same competencies and organization Science and common sense must converge
51 Thinking in the POCT Box Incorrect Identification Sample Handling/ Transport Equipment Malfunction Improper Data Entry Insufficient Sample Pre Analytical 62% Analytical 15% Post Analytical 23% Reporting or Analysis Incorrect Sample Sample Condition Sample Mix-Ups/ Interferences Delayed Turn-around Time As autononmation reduces errors in the box, further reductions must occur outside the box.
52 Thinking Outside the POCT Box Pre-pre: Phsician must consider» What POCT is available?» What POCT will best serve the patient?» Will an immediate answer improve the patient s outcome? Post-post: Is the Physician?» Receptive to using an immediate POCT result» Able to interpret result in the patient s context» Amenable to initiating an immediate response
53 The Problem with Pedestals
54 QUESTIONS
Patient Safety: A Quality System Approach To POCT QC/QA
 Patient Safety: A Quality System Approach To POCT QC/QA Ellis Jacobs, Ph.D., DABCC New York University School of Medicine Coler-Goldwater Specialty Hospital & Nursing Facility New York, New York Point-of-Care
Patient Safety: A Quality System Approach To POCT QC/QA Ellis Jacobs, Ph.D., DABCC New York University School of Medicine Coler-Goldwater Specialty Hospital & Nursing Facility New York, New York Point-of-Care
Ellis Jacobs, Ph.D., DABCC. New York University School of Medicine Bellevue Hospital Center New York, New York
 Ellis Jacobs, Ph.D., DABCC New York University School of Medicine Bellevue Hospital Center New York, New York ADA Recommendation In terms of glucose in venous plasma WHO Recommendation In terms of glucose
Ellis Jacobs, Ph.D., DABCC New York University School of Medicine Bellevue Hospital Center New York, New York ADA Recommendation In terms of glucose in venous plasma WHO Recommendation In terms of glucose
Quality Control for Point-of-Care Testing
 Quality Control for Point-of-Care Testing James H. Nichols, PhD, DABCC, FACB Professor of Pathology, Microbiology and Immunology Medical Director of Clinical Chemistry and Point-of-Care Testing Vanderbilt
Quality Control for Point-of-Care Testing James H. Nichols, PhD, DABCC, FACB Professor of Pathology, Microbiology and Immunology Medical Director of Clinical Chemistry and Point-of-Care Testing Vanderbilt
C30-A2 ISBN ISSN Point-of-Care Blood Glucose Testing in Acute and Chronic Care Facilities; Approved Guideline Second Edition
 ISBN 1-56238-471-6 ISSN 0273-3099 Point-of-Care Blood Glucose Testing in Acute and Chronic Care Facilities; Approved Guideline Second Edition Volume 22 Number 17 David B. Sacks, M.D., Chairholder Patricia
ISBN 1-56238-471-6 ISSN 0273-3099 Point-of-Care Blood Glucose Testing in Acute and Chronic Care Facilities; Approved Guideline Second Edition Volume 22 Number 17 David B. Sacks, M.D., Chairholder Patricia
Point of Care testing refers to all laboratory testing that is done outside of the walls of the clinical laboratory in the proximity of the patient.
 1 2 Point of Care testing refers to all laboratory testing that is done outside of the walls of the clinical laboratory in the proximity of the patient. All such tests are considered lab tests and are
1 2 Point of Care testing refers to all laboratory testing that is done outside of the walls of the clinical laboratory in the proximity of the patient. All such tests are considered lab tests and are
Individual Quality Control Plans (IQCP) Risk Assessment
 Individual Quality Control Plans (IQCP) Risk Assessment James H. Nichols, PhD, DABCC, FACB Professor of Clinical Pathology, Microbiology and Immunology Medical Director of Clinical Chemistry and Point-of-Care
Individual Quality Control Plans (IQCP) Risk Assessment James H. Nichols, PhD, DABCC, FACB Professor of Clinical Pathology, Microbiology and Immunology Medical Director of Clinical Chemistry and Point-of-Care
Basic knowledge of POCT instuments. The Tide Resort Bangsan Beach 18 June 2009
 Basic knowledge of POCT instuments The Tide Resort Bangsan Beach 18 June 2009 1 POCT instruments concepts Point of care testing Diagnostic medical devices used at or near the site of patient care Bed site,
Basic knowledge of POCT instuments The Tide Resort Bangsan Beach 18 June 2009 1 POCT instruments concepts Point of care testing Diagnostic medical devices used at or near the site of patient care Bed site,
Point of Care Testing for INR using CoaguChek XS Plus
 Point of Care Testing, Pathology Page 1 of 18 Point of Care Testing for INR using CoaguChek XS Plus EDITION No 1.4 DATE OF ISSUE Feb 2014 REVIEW INTERVAL AUTHOR LOCATION OF COPIES 3 YEARS D O Neill 1.
Point of Care Testing, Pathology Page 1 of 18 Point of Care Testing for INR using CoaguChek XS Plus EDITION No 1.4 DATE OF ISSUE Feb 2014 REVIEW INTERVAL AUTHOR LOCATION OF COPIES 3 YEARS D O Neill 1.
The analytical phase
 The analytical phase Result interpretation Test request Result Sampling Black box: the lab ANALYTICAL PHASE The CASE Uncle Pete, 67 years old Marked abdominal pain 8 pm, ED Acute abdomen? Assessment (+
The analytical phase Result interpretation Test request Result Sampling Black box: the lab ANALYTICAL PHASE The CASE Uncle Pete, 67 years old Marked abdominal pain 8 pm, ED Acute abdomen? Assessment (+
Point of Care Testing (POCT)
 Accounting for Pre-analytical Variables in POCT L.V. Rao, PhD Senior Clinical Laboratory Director Associate Professor, Pathology UMass Memorial Medical Center (Worcester, MA) Senior Scientific Director
Accounting for Pre-analytical Variables in POCT L.V. Rao, PhD Senior Clinical Laboratory Director Associate Professor, Pathology UMass Memorial Medical Center (Worcester, MA) Senior Scientific Director
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY ASSAY AND INSTRUMENT COMBINATION TEMPLATE
 510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY ASSAY AND INSTRUMENT COMBINATION TEMPLATE A. 510(k) Number: k063276 B. Purpose for Submission: New device C. Measurand: Urinary Glucose, Blood,
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY ASSAY AND INSTRUMENT COMBINATION TEMPLATE A. 510(k) Number: k063276 B. Purpose for Submission: New device C. Measurand: Urinary Glucose, Blood,
Quality Specifications for POCT
 Quality Specifications for POCT the patient journey perspective Christopher P Price Department of Clinical Biochemistry, University of Oxford, Oxford, UK Quality Specifications fit-for-purpose QUALITY
Quality Specifications for POCT the patient journey perspective Christopher P Price Department of Clinical Biochemistry, University of Oxford, Oxford, UK Quality Specifications fit-for-purpose QUALITY
The challenges of quality assessment for point-of-care testing. Anne Stavelin. EQALM Symposium, Bergen 8 October General challenges
 The challenges of quality assessment for point-of-care testing Anne Stavelin EQALM Symposium, Bergen 8 October 2015 www.noklus.no Norwegian quality improvement of primary care laboratories General challenges
The challenges of quality assessment for point-of-care testing Anne Stavelin EQALM Symposium, Bergen 8 October 2015 www.noklus.no Norwegian quality improvement of primary care laboratories General challenges
The good, the bad, the ugly A month in the life of a Point Of Care Coordinator
 The good, the bad, the ugly A month in the life of a Point Of Care Coordinator Stefanie Van Heule Ghent University Hospital 23/10/2014 Ghent University Hospital ± 3000 patients / day ± 62 000 total admissions
The good, the bad, the ugly A month in the life of a Point Of Care Coordinator Stefanie Van Heule Ghent University Hospital 23/10/2014 Ghent University Hospital ± 3000 patients / day ± 62 000 total admissions
Interference and Point-of-Care Testing Devices
 Interference and Point-of-Care Testing Devices Nam K. Tran, PhD, HCLD, (ABB), FACB Associate Clinical Professor Director of Clinical Chemistry, Special Chemistry/Toxicology and POCT 1 Learning Objectives
Interference and Point-of-Care Testing Devices Nam K. Tran, PhD, HCLD, (ABB), FACB Associate Clinical Professor Director of Clinical Chemistry, Special Chemistry/Toxicology and POCT 1 Learning Objectives
SRT-100 Skin cancer TReaTmenT Simplified
 SRT-100 Skin cancer treatment simplified More Patients, Few Choices Cases of skin cancer have been on the rise for decades, and are dramatically escalating as the population ages. According to the Centers
SRT-100 Skin cancer treatment simplified More Patients, Few Choices Cases of skin cancer have been on the rise for decades, and are dramatically escalating as the population ages. According to the Centers
CLINITEK Novus Automated Urine Chemistry Analyzer
 CLINITEK, Atlas, Novus, Status+, and all associated marks are trademarks of Siemens Healthcare Diagnostics Inc. or its affiliates. All other trademarks are properties of their respective owners. Product
CLINITEK, Atlas, Novus, Status+, and all associated marks are trademarks of Siemens Healthcare Diagnostics Inc. or its affiliates. All other trademarks are properties of their respective owners. Product
PROCEDURE. TITLE: Bedside Glucose Monitoring PC Laboratory. Issuing Department: Clinical Director Signature: Departments Involved:
 PROCEDURE TITLE: Bedside Glucose Monitoring Issuing Department: Clinical Director Signature: Departments Involved: Laboratory Nursing Effective Date: 10/97 Review Dates: 09/01, 07/02, 05/13 Revision Dates:
PROCEDURE TITLE: Bedside Glucose Monitoring Issuing Department: Clinical Director Signature: Departments Involved: Laboratory Nursing Effective Date: 10/97 Review Dates: 09/01, 07/02, 05/13 Revision Dates:
Title/Description: Point of Care Glucose Testing-Waived Department: Organization Wide Personnel: All Effective Date: November 29, 2016 Revised:
 Title/Description: Point of Care Glucose Testing-Waived Department: Organization Wide Personnel: All Effective Date: November 29, 2016 Revised: PURPOSE The Abbott FreeStyle Precision Pro (FSPP) Blood Glucose
Title/Description: Point of Care Glucose Testing-Waived Department: Organization Wide Personnel: All Effective Date: November 29, 2016 Revised: PURPOSE The Abbott FreeStyle Precision Pro (FSPP) Blood Glucose
The Virtues and Pitfalls of Implementing a New Test
 The Virtues and Pitfalls of Implementing a New Test James H. Nichols, Ph.D., DABCC, FACB Professor of Clinical Pathology, Microbiology and Immunology Associate Medical Director for Clinical Operations
The Virtues and Pitfalls of Implementing a New Test James H. Nichols, Ph.D., DABCC, FACB Professor of Clinical Pathology, Microbiology and Immunology Associate Medical Director for Clinical Operations
Errors in POCT: When Improbable Situations Become Possible
 Errors in POCT: When Improbable Situations Become Possible James H. Nichols, Ph.D., DABCC, FACB Chairholder CLSI EP23 Document Development Committee Director, Section of Clinical Chemistry Department of
Errors in POCT: When Improbable Situations Become Possible James H. Nichols, Ph.D., DABCC, FACB Chairholder CLSI EP23 Document Development Committee Director, Section of Clinical Chemistry Department of
Siemens Healthcare Diagnostics
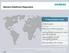 Siemens Healthcare Diagnostics A Global Industry Leader 3.7bn in sales More than 14,500 employees Serving 30,000 customers in more than 120 countries Approximately 244,000 instruments installed R&D spending
Siemens Healthcare Diagnostics A Global Industry Leader 3.7bn in sales More than 14,500 employees Serving 30,000 customers in more than 120 countries Approximately 244,000 instruments installed R&D spending
cobas u 601 urine analyzer Providing precise and secure test results
 Providing precise and secure test results 1 of 6 Providing precise and secure test results cobas u 601 urine analyzer is a modular, analytical system platform for urine strip testing and features a fully
Providing precise and secure test results 1 of 6 Providing precise and secure test results cobas u 601 urine analyzer is a modular, analytical system platform for urine strip testing and features a fully
Fundamentals of Pulse Oximetry
 Donald Gessling presents: Fundamentals of Pulse Oximetry Sponsored by: Fluke Biomedical Wednesday, September 14, 2016, 2:00pm ET PRESENTER: DONALD GESSLING, FLUKE BIOMEDICAL Donald Gessling, Senior Engineer
Donald Gessling presents: Fundamentals of Pulse Oximetry Sponsored by: Fluke Biomedical Wednesday, September 14, 2016, 2:00pm ET PRESENTER: DONALD GESSLING, FLUKE BIOMEDICAL Donald Gessling, Senior Engineer
MARSHALLTOWN MEDICAL & SURGICAL CENTER Marshalltown, Iowa
 MARSHALLTOWN MEDICAL & SURGICAL CENTER Marshalltown, Iowa CARE OF PATIENT POLICY & PROCEDURE Policy Number: 4:10 Subject: Policy: Glucose Monitoring (Accuchek) Nursing department staff and laboratory staff
MARSHALLTOWN MEDICAL & SURGICAL CENTER Marshalltown, Iowa CARE OF PATIENT POLICY & PROCEDURE Policy Number: 4:10 Subject: Policy: Glucose Monitoring (Accuchek) Nursing department staff and laboratory staff
Rapid Diagnostics CHAI Experience. 6 th Moving Forward in Diagnostics Forum Les Pensieres November 7, 2012
 Rapid Diagnostics CHAI Experience 6 th Moving Forward in Diagnostics Forum Les Pensieres November 7, 2012 CHAI is working in 12 countries on HIV POC test implementation Kenya Tanzania Ethiopia Malawi Mozambique
Rapid Diagnostics CHAI Experience 6 th Moving Forward in Diagnostics Forum Les Pensieres November 7, 2012 CHAI is working in 12 countries on HIV POC test implementation Kenya Tanzania Ethiopia Malawi Mozambique
Implementing of Good Laboratory Practice (GLP) in Food Analysis Laboratories of Baghdad University
 ISSN (Online): 2319-764 Index Copernicus Value (215): 78.96 Impact Factor (215): 6.391 Implementing of Good Laboratory Practice (GLP) in Food Analysis Laboratories of Baghdad University Mahmud Abdullah
ISSN (Online): 2319-764 Index Copernicus Value (215): 78.96 Impact Factor (215): 6.391 Implementing of Good Laboratory Practice (GLP) in Food Analysis Laboratories of Baghdad University Mahmud Abdullah
Summary of Significant Changes at this Revision
 COPY Summary of Significant Changes at this Revision CR5890 Removal of Ketone guidelines - refer to Specialist Diabetes nurse (adult wards); Children's ward has their own protocol Remove all references
COPY Summary of Significant Changes at this Revision CR5890 Removal of Ketone guidelines - refer to Specialist Diabetes nurse (adult wards); Children's ward has their own protocol Remove all references
The Laboratories Role in Global Health
 The Laboratories Role in Global Health Larry Westerman, Ph.D. International Laboratory Branch Division of Global HIV/AIDS Centers for Disease Control and Prevention Center for Global Health International
The Laboratories Role in Global Health Larry Westerman, Ph.D. International Laboratory Branch Division of Global HIV/AIDS Centers for Disease Control and Prevention Center for Global Health International
Abbott PXP Glucose Meters Resource: Lin Gustafson, MT(ASCP) Point of Care Coordinator, Laboratory
 Abbott PXP Glucose Meters 2015 Resource: Lin Gustafson, MT(ASCP) Point of Care Coordinator, Laboratory Objectives After you complete this Computer-Based Learning (CBL) module, you should be able to: Explain
Abbott PXP Glucose Meters 2015 Resource: Lin Gustafson, MT(ASCP) Point of Care Coordinator, Laboratory Objectives After you complete this Computer-Based Learning (CBL) module, you should be able to: Explain
IAS Presentation at Kuwait Labs nd Kuwait Conference for Laboratory Quality and Accreditation Empowering Laboratory Personnel
 IAS Presentation at Kuwait Labs 2017 2nd Kuwait Conference for Laboratory Quality and Accreditation Empowering Laboratory Personnel September 26, 2017 Kuwait City Lawrence J. O Connor, P.E., L.S., F. NSPE
IAS Presentation at Kuwait Labs 2017 2nd Kuwait Conference for Laboratory Quality and Accreditation Empowering Laboratory Personnel September 26, 2017 Kuwait City Lawrence J. O Connor, P.E., L.S., F. NSPE
Topic: Q&A from CAP Webinar: Stay in Compliance With the CMS Directive Regarding PT Testing on Multiple Instruments Date: November 12, 2015
 Topic: Q&A from CAP Webinar: Stay in Compliance With the CMS Directive Regarding PT Testing on Multiple Instruments Date: November 12, 2015 The questions below were received from attendees of the CAP webinar
Topic: Q&A from CAP Webinar: Stay in Compliance With the CMS Directive Regarding PT Testing on Multiple Instruments Date: November 12, 2015 The questions below were received from attendees of the CAP webinar
SAMPLE. Glucose Monitoring in Settings Without Laboratory Support
 POCT13c Glucose Monitoring in Settings Without Laboratory Support This guideline focuses on performance of point-of-care glucose monitoring systems, with an emphasis on safety practices, quality control,
POCT13c Glucose Monitoring in Settings Without Laboratory Support This guideline focuses on performance of point-of-care glucose monitoring systems, with an emphasis on safety practices, quality control,
Solutions at GE Inspection Technologies
 Solutions at GE Inspection Technologies Business Overview Paul A. Meyer Our Company A global leader in technology-driven inspection solutions that deliver productivity, quality, and safety to our customers
Solutions at GE Inspection Technologies Business Overview Paul A. Meyer Our Company A global leader in technology-driven inspection solutions that deliver productivity, quality, and safety to our customers
Auto Validation and Management Reports Using Sysmex WAM. Alfonso Ziccardi Laboratory Operation Manager for AP/CP/Safety Officer
 Auto Validation and Management Reports Using Sysmex WAM Alfonso Ziccardi Laboratory Operation Manager for AP/CP/Safety Officer SUCCESS BEYOND FINANCES Hematology Testing Breakdown NYHQ Descrete testing
Auto Validation and Management Reports Using Sysmex WAM Alfonso Ziccardi Laboratory Operation Manager for AP/CP/Safety Officer SUCCESS BEYOND FINANCES Hematology Testing Breakdown NYHQ Descrete testing
Balancing investment in point of care diagnostics versus laboratory testing in low resource settings. June 28, 2011
 Balancing investment in point of care diagnostics versus laboratory testing in low resource settings Workshop on TB and HIV Diagnostics Workshop on TB and HIV Diagnostics June 28, 2011 Complexity Delivery
Balancing investment in point of care diagnostics versus laboratory testing in low resource settings Workshop on TB and HIV Diagnostics Workshop on TB and HIV Diagnostics June 28, 2011 Complexity Delivery
It s a Gas! Issues in the Blood Gas Laboratory. D. Robert Dufour, MD, FCAP Consultant Pathologist VA Medical Center, Washington DC
 It s a Gas! Issues in the Blood Gas Laboratory D. Robert Dufour, MD, FCAP Consultant Pathologist VA Medical Center, Washington DC Learning Objectives After participating in this session, you will be able
It s a Gas! Issues in the Blood Gas Laboratory D. Robert Dufour, MD, FCAP Consultant Pathologist VA Medical Center, Washington DC Learning Objectives After participating in this session, you will be able
BÜHLMANN fcal turbo. Calprotectin turbidimetric assay for professional use. Reagent Kit B-KCAL-RSET. Revision date:
 BÜHLMANN fcal turbo Calprotectin turbidimetric assay for professional use Reagent Kit B-KCAL-RSET Revision date: 2017-02-24 BÜHLMANN LABORATORIES AG Baselstrasse 55 4124 Schönenbuch, Switzerland Tel.:
BÜHLMANN fcal turbo Calprotectin turbidimetric assay for professional use Reagent Kit B-KCAL-RSET Revision date: 2017-02-24 BÜHLMANN LABORATORIES AG Baselstrasse 55 4124 Schönenbuch, Switzerland Tel.:
TITLE: Whole Blood Glucose Monitoring Using the Nova StatStrip Meter
 Page 4 of 14 IV. Performance of Quality Control A. Purpose: To detect any errors due to system failures or operator performance B. Frequency: Levels 1 and 3 controls are run every 24 hours of use. C. Quality
Page 4 of 14 IV. Performance of Quality Control A. Purpose: To detect any errors due to system failures or operator performance B. Frequency: Levels 1 and 3 controls are run every 24 hours of use. C. Quality
Mass Spectrometry Made Simple for Clinical Diagnostic Labs
 Mass Spectrometry Made Simple for Clinical Diagnostic Labs The SCIEX Topaz System with FDA-Cleared Topaz Vitamin D 200M Assay Kit Mass Spectrometry Made Simple SCIEX Topaz System for Clinical Diagnostics
Mass Spectrometry Made Simple for Clinical Diagnostic Labs The SCIEX Topaz System with FDA-Cleared Topaz Vitamin D 200M Assay Kit Mass Spectrometry Made Simple SCIEX Topaz System for Clinical Diagnostics
NexScreen Cup. Urine Drug Screen * READ POLICY PRIOR TO STARTING TUTORIAL
 NexScreen Cup Urine Drug Screen * READ POLICY PRIOR TO STARTING TUTORIAL Before Testing a Patient Orient yourself to your working area: Locate the NexScreen Cup. Locate testing supplies: Positive and Negative
NexScreen Cup Urine Drug Screen * READ POLICY PRIOR TO STARTING TUTORIAL Before Testing a Patient Orient yourself to your working area: Locate the NexScreen Cup. Locate testing supplies: Positive and Negative
URYXXON 300 Instrumental evaluation of urine test sticks
 URYXXON 300 Instrumental evaluation of urine test sticks MACHEREY-NAGEL URYXXON 300 Instrumental evaluation Boost your lab performance immediate startup one measurement every 8 seconds (400 sticks/h) bar
URYXXON 300 Instrumental evaluation of urine test sticks MACHEREY-NAGEL URYXXON 300 Instrumental evaluation Boost your lab performance immediate startup one measurement every 8 seconds (400 sticks/h) bar
POINT OF CARE DIAGNOSTICS
 ENSURING ACCESS TO POINT OF CARE DIAGNOSTICS IN DECENTRALISED HIV AIDS PROGRAMMES REQUEST FOR EXPRESSION OF INTEREST Technology : Point of Care HIV diagnostic and treatment monitoring devices for Early
ENSURING ACCESS TO POINT OF CARE DIAGNOSTICS IN DECENTRALISED HIV AIDS PROGRAMMES REQUEST FOR EXPRESSION OF INTEREST Technology : Point of Care HIV diagnostic and treatment monitoring devices for Early
M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017
 M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017 If laboratory results are required on a STAT basis, the designated commercial medical laboratory
M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017 If laboratory results are required on a STAT basis, the designated commercial medical laboratory
HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2017Apr19
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2017Apr19
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2017Apr19
Management of Central Venous Access Devices. Blood Glucose Monitoring
 Management of Central Venous Access Devices Blood Glucose Monitoring Purpose To provide education on the standard of care regarding the use and monitoring of the Accu- Chek Blood glucose machine, including
Management of Central Venous Access Devices Blood Glucose Monitoring Purpose To provide education on the standard of care regarding the use and monitoring of the Accu- Chek Blood glucose machine, including
Delta Check Calculation Guide
 Delta Check Calculation Guide National Technology 2017, All Rights Reserved By Senior Scientific Researcher, Asmaa Taher Table of Contents Definition... 2 Purpose... 2 Delta Check Research Studies... 2
Delta Check Calculation Guide National Technology 2017, All Rights Reserved By Senior Scientific Researcher, Asmaa Taher Table of Contents Definition... 2 Purpose... 2 Delta Check Research Studies... 2
SAMPLE. Clinical and Laboratory Standards Institute Advancing Quality in Health Care Testing
 Vol. 32 No. 12 Replaces EP27-P Vol. 29 No. 16 How to Construct and Interpret an Error Grid for Quantitative Diagnostic Assays; Approved Guideline This guideline describes what an error grid is, why it
Vol. 32 No. 12 Replaces EP27-P Vol. 29 No. 16 How to Construct and Interpret an Error Grid for Quantitative Diagnostic Assays; Approved Guideline This guideline describes what an error grid is, why it
Polymer Technology Systems, Inc. CardioChek PA Comparison Study
 Polymer Technology Systems, Inc. CardioChek PA Comparison Study Evaluation Protocol: Accuracy Precision Clinical Correlation PTS Panels Lipid Panel Test Strips For Use in Comparisons to a Reference Laboratory
Polymer Technology Systems, Inc. CardioChek PA Comparison Study Evaluation Protocol: Accuracy Precision Clinical Correlation PTS Panels Lipid Panel Test Strips For Use in Comparisons to a Reference Laboratory
Alere Triage System. Knowing now matters when rapid treatment decisions can be made at the point-of-care.
 System Knowing now matters when rapid treatment decisions can be made at the point-of-care. The Alere Triage System The MeterPro is utilized in thousands of healthcare facilities worldwide. A comprehensive
System Knowing now matters when rapid treatment decisions can be made at the point-of-care. The Alere Triage System The MeterPro is utilized in thousands of healthcare facilities worldwide. A comprehensive
future technology healthcare Introducing the KUDUwave TM
 future technology healthcare Introducing the KUDUwave TM By harnessing artificial intelligence and automation we are disrupting the industry by making primary health care both affordable and accessible
future technology healthcare Introducing the KUDUwave TM By harnessing artificial intelligence and automation we are disrupting the industry by making primary health care both affordable and accessible
HIV Viral Load Quality Assessment Program Summary for Panel HIVVL 2017Oct27
 1 The National Laboratory for HIV Reference Services is Accredited to ISO 15189 and ISO 17043 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology
1 The National Laboratory for HIV Reference Services is Accredited to ISO 15189 and ISO 17043 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology
HIV Viral Load Quality Assessment Program Summary for Panel HIVVL 2018Oct26
 1 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Viral Load Quality Assessment Program Summary
1 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Viral Load Quality Assessment Program Summary
Routine, Every 2 hours, Starting today, If temperature greater than 38.5 C initiate Evaluation for Possible Sepsis Physician Order #829
 Height Weight Allergies General Vital Signs [X] Frequent vital signs Indication: Q15 minutes x (# of occurrences): 4 Q30 minutes x (# of occurrences): 4 Q1 hour x (# of occurrences): Q2 hours x (# of occurrences):
Height Weight Allergies General Vital Signs [X] Frequent vital signs Indication: Q15 minutes x (# of occurrences): 4 Q30 minutes x (# of occurrences): 4 Q1 hour x (# of occurrences): Q2 hours x (# of occurrences):
Chapter 14: Arterial Puncture Procedures
 Objectives Chapter 14: Arterial Puncture Procedures 1. Define the key terms and abbreviations listed at the beginning of this chapter. 2. State the primary reason for performing arterial punctures and
Objectives Chapter 14: Arterial Puncture Procedures 1. Define the key terms and abbreviations listed at the beginning of this chapter. 2. State the primary reason for performing arterial punctures and
Quality control in the hematology laboratory for the sake of the patient. Dr Marion Münster Manager Medical & Scientific Affairs Sysmex South Africa
 Quality control in the hematology laboratory for the sake of the patient Dr Marion Münster Manager Medical & Scientific Affairs Sysmex South Africa The FBC. Most frequently requested lab test Usually delegated
Quality control in the hematology laboratory for the sake of the patient Dr Marion Münster Manager Medical & Scientific Affairs Sysmex South Africa The FBC. Most frequently requested lab test Usually delegated
Point of Care (POC) Glucose Testing Revised on: December, 2018 Reference: Policy LAB 23-A; Point of Care Nova Stat Strip Glucose Meter
 Point of Care (POC) Glucose Testing Revised on: December, 2018 Reference: Policy LAB 23-A; Point of Care Nova Stat Strip Glucose Meter The Nova Stat Strip Blood Glucose meter is UHB s approved Glucometer
Point of Care (POC) Glucose Testing Revised on: December, 2018 Reference: Policy LAB 23-A; Point of Care Nova Stat Strip Glucose Meter The Nova Stat Strip Blood Glucose meter is UHB s approved Glucometer
WHO Prequalification of In Vitro Diagnostics Programme
 WHO Prequalification of In Vitro Diagnostics Programme International HIV/Viral Hepatitis Co-Infection Satellite Meeting 19 July 2014, Melbourne Anita Sands Prequalification Diagnostics Team Department
WHO Prequalification of In Vitro Diagnostics Programme International HIV/Viral Hepatitis Co-Infection Satellite Meeting 19 July 2014, Melbourne Anita Sands Prequalification Diagnostics Team Department
Analytical and Clinical Performance of the epoc Blood Analysis System. Experience at a Large Tertiary Academic Medical Center
 AJCP / Original Article Analytical and Clinical Performance of the epoc Blood Analysis System Experience at a Large Tertiary Academic Medical Center Brie A. Stotler, MD, MPH, 1,2 and Alexander Kratz, MD,
AJCP / Original Article Analytical and Clinical Performance of the epoc Blood Analysis System Experience at a Large Tertiary Academic Medical Center Brie A. Stotler, MD, MPH, 1,2 and Alexander Kratz, MD,
Community-Based Point-of-Care Testing: From Innovative Care Model to Common Practice
 Community-Based Point-of-Care Testing: From Innovative Care Model to Common Practice Donald G. Klepser, PhD, MBA Associate Professor University of Nebraska Medical Center Brian Bobby Rite Aid Vice President,
Community-Based Point-of-Care Testing: From Innovative Care Model to Common Practice Donald G. Klepser, PhD, MBA Associate Professor University of Nebraska Medical Center Brian Bobby Rite Aid Vice President,
Med Chem 535P ~ Diagnostic Medicinal Chemistry. General Comments
 Med Chem 535P ~ Diagnostic Medicinal Chemistry General Comments Most blood chemistry and serology assays are performed automatically. Larger clinical laboratories often use sophisticated analyzers that
Med Chem 535P ~ Diagnostic Medicinal Chemistry General Comments Most blood chemistry and serology assays are performed automatically. Larger clinical laboratories often use sophisticated analyzers that
The Quidel Triage System
 The Quidel Triage System Automated. Calibrated. Connected. Professional diagnostics at the point-of-care BR1026500EN00.qxp_Layout 1 8/28/18 2:28 PM Page 4 The Quidel Triage System Fast quantitative results
The Quidel Triage System Automated. Calibrated. Connected. Professional diagnostics at the point-of-care BR1026500EN00.qxp_Layout 1 8/28/18 2:28 PM Page 4 The Quidel Triage System Fast quantitative results
Aviva Combo BLOOD GLUCOSE METER. Advanced Owner s Booklet for Self-Testing Only
 Aviva Combo BLOOD GLUCOSE METER Advanced Owner s Booklet for Self-Testing Only Before You Start Testing About the meter and test strips Carefully read and follow the instructions in the Getting Started
Aviva Combo BLOOD GLUCOSE METER Advanced Owner s Booklet for Self-Testing Only Before You Start Testing About the meter and test strips Carefully read and follow the instructions in the Getting Started
Quality Assurance Policy. for the. Procurement of HIV Point-of-Care Technology. under the UNITAID Grant
 Quality Assurance Policy for the Procurement of HIV Point-of-Care Technology under the UNITAID Grant UNICEF Supply Division Quality Assurance Centre and Health Technology Centre Revisions Version Date
Quality Assurance Policy for the Procurement of HIV Point-of-Care Technology under the UNITAID Grant UNICEF Supply Division Quality Assurance Centre and Health Technology Centre Revisions Version Date
Precision ceedpro. Online Training for. Glucometer & Capillary Collection
 Online Training for Precision ceedpro Glucometer & Capillary Collection How long will it take? This module takes approximately 30 minutes to complete. What if I have to leave before I finish? Progress
Online Training for Precision ceedpro Glucometer & Capillary Collection How long will it take? This module takes approximately 30 minutes to complete. What if I have to leave before I finish? Progress
T R A I N I N G G U I D E
 TRAINING GUIDE InstaCube Oral Fluid Drug Test For Forensic Use Only Powered By: Contents 3 The information in this presentation is a general overview on performing and interpreting the InstaCube Oral Fluid
TRAINING GUIDE InstaCube Oral Fluid Drug Test For Forensic Use Only Powered By: Contents 3 The information in this presentation is a general overview on performing and interpreting the InstaCube Oral Fluid
Role of Core Lab in Clinical Trial M. Therese Tupas-Habib, RDCS Manager, Cardiovascular Core Lab MedStar Health Research Institute
 Role of Core Lab in Clinical Trial M. Therese Tupas-Habib, RDCS Manager, Cardiovascular Core Lab MedStar Health Research Institute ASE Echo in Clinical Trials JASE Oct 2004 Why Echo? - non-invasive diagnostic
Role of Core Lab in Clinical Trial M. Therese Tupas-Habib, RDCS Manager, Cardiovascular Core Lab MedStar Health Research Institute ASE Echo in Clinical Trials JASE Oct 2004 Why Echo? - non-invasive diagnostic
Alere Technologies GmbH
 Alere Technologies GmbH Mobile Smart Instruments for Automated Image Analysis and Point of Care Diagnosis April 2011 Alere Technologies GmbH Alere Technologies GmbH Based in Jena, Thuringia, Germany ~300
Alere Technologies GmbH Mobile Smart Instruments for Automated Image Analysis and Point of Care Diagnosis April 2011 Alere Technologies GmbH Alere Technologies GmbH Based in Jena, Thuringia, Germany ~300
040716_ANDI_VENTIS WEBSITE(new) Printout 1. HOME PAGE. Welcome to our world of Dry Powder Inhalers!
 1. HOME PAGE Welcome to our world of Dry Powder Inhalers! Andi-Ventis Ltd, established in 2001 is a joint venture between Medochemie Ltd, Cyprus and Miat SpA Italy, founded to carry out the pharmaceutical
1. HOME PAGE Welcome to our world of Dry Powder Inhalers! Andi-Ventis Ltd, established in 2001 is a joint venture between Medochemie Ltd, Cyprus and Miat SpA Italy, founded to carry out the pharmaceutical
Operator s Manual. Blood Glucose and β-ketone Monitoring System. Abbott Diabetes Care Inc South Loop Road Alameda, CA USA
 Blood Glucose and β-ketone Monitoring System Operator s Manual Precision Xceed Pro, TrueID, TrueMeasure, TrueAccess, PrecisionWeb, Precision, MediSense, Optium and Precision PCx are trademarks of the Abbott
Blood Glucose and β-ketone Monitoring System Operator s Manual Precision Xceed Pro, TrueID, TrueMeasure, TrueAccess, PrecisionWeb, Precision, MediSense, Optium and Precision PCx are trademarks of the Abbott
SEXUAL HEALTH LAB PRODUCTS
 SEXUAL HEALTH LAB PRODUCTS Quick, reliable results for in-office testing Start treatments sooner. Enhance patient outcomes. Long waits for results. Delayed diagnosis and treatment. Cancelled follow-up
SEXUAL HEALTH LAB PRODUCTS Quick, reliable results for in-office testing Start treatments sooner. Enhance patient outcomes. Long waits for results. Delayed diagnosis and treatment. Cancelled follow-up
THE MINIMED 670G SYSTEM SCHOOL NURSE GUIDE
 THE MINIMED 670G SYSTEM SCHOOL NURSE GUIDE Indicated for type 1 patients 14 and over. Prescription required. WARNING: Medtronic performed an evaluation of the MiniMed 670G system and determined that it
THE MINIMED 670G SYSTEM SCHOOL NURSE GUIDE Indicated for type 1 patients 14 and over. Prescription required. WARNING: Medtronic performed an evaluation of the MiniMed 670G system and determined that it
WELCOME! HemoCue Presentation CPOCT San Diego, September 20 th Annika Eriksson Global Application Manager HemoCue AB
 WELCOME! HemoCue Presentation CPOCT San Diego, September 20 th 2014 Annika Eriksson Global Application Manager HemoCue AB HemoCue Part of the Danaher Family 2 Pioneers in Point of care Testing When HemoCue
WELCOME! HemoCue Presentation CPOCT San Diego, September 20 th 2014 Annika Eriksson Global Application Manager HemoCue AB HemoCue Part of the Danaher Family 2 Pioneers in Point of care Testing When HemoCue
PROCEDURE NO. POC LBH. Printed copies are for reference only. Please refer to the electronic copy for the latest version.
 Department Of Pathology POC.514.06- Blood Glucose Monitoring Accu-Chek Inform II Procedure-LBH Version# 6 Department PROCEDURE NO. PAGE NO. Point-of-Care Testing POC.514.05 LBH 1 OF 7 Printed copies are
Department Of Pathology POC.514.06- Blood Glucose Monitoring Accu-Chek Inform II Procedure-LBH Version# 6 Department PROCEDURE NO. PAGE NO. Point-of-Care Testing POC.514.05 LBH 1 OF 7 Printed copies are
HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
Bloodborne Pathogens. Montclair Kimberley Academy 1
 Bloodborne Pathogens Montclair Kimberley Academy 1 Introduction! Approximately 5.6 million workers in health care and other facilities are at risk of exposure to bloodborne pathogens such as human immunodeficiency
Bloodborne Pathogens Montclair Kimberley Academy 1 Introduction! Approximately 5.6 million workers in health care and other facilities are at risk of exposure to bloodborne pathogens such as human immunodeficiency
H5N1 real- time Reverse Transcription Polymerase Chain Reaction (RT-PCR) assay
 Case Study 1: H5N1 real- time Reverse Transcription Polymerase Chain Reaction (RT-PCR) assay Detection kit that can accurately and rapidly detect all known strains of the H5N1 Avian Influenza A virus in
Case Study 1: H5N1 real- time Reverse Transcription Polymerase Chain Reaction (RT-PCR) assay Detection kit that can accurately and rapidly detect all known strains of the H5N1 Avian Influenza A virus in
Regulations: Minimal Sedation. Jason H. Goodchild, DMD
 Regulations: Minimal Sedation Jason H. Goodchild, DMD August 2016 Caveats 1. The regulations about to be presented are accurate and current as of today. 2. This could change tomorrow. 3. It is up to every
Regulations: Minimal Sedation Jason H. Goodchild, DMD August 2016 Caveats 1. The regulations about to be presented are accurate and current as of today. 2. This could change tomorrow. 3. It is up to every
Fraser Health Authority Accu-chek Inform II: Training Script
 1. Introduction Ensure that each participant Signs the attendance Sheet (please PRINT first & last name, and middle initial legibly) Refer to; For Reference Only: o Quick Reference Guide o What s new document
1. Introduction Ensure that each participant Signs the attendance Sheet (please PRINT first & last name, and middle initial legibly) Refer to; For Reference Only: o Quick Reference Guide o What s new document
ADVANCED MANUFACTURING CAREER CLUSTER
 ADVANCED MANUFACTURING CAREER CLUSTER QUALITY ASSURANCE/ CONTROL PRODUCTION Industrial Production Managers MAINTENANCE AND REPAIR Research & Development/ Materials Industrial and Manufacturing Mechanical
ADVANCED MANUFACTURING CAREER CLUSTER QUALITY ASSURANCE/ CONTROL PRODUCTION Industrial Production Managers MAINTENANCE AND REPAIR Research & Development/ Materials Industrial and Manufacturing Mechanical
AMDS Partners and Stakeholders Meeting CHAI HIV Diagnostics Forecasting Overview th September, 2014
 AMDS Partners and Stakeholders Meeting CHAI HIV Diagnostics Forecasting Overview 29-30 th September, 2014 Agenda Introduction Overview of Global Diagnostics Forecasting Overview of Country Forecasting
AMDS Partners and Stakeholders Meeting CHAI HIV Diagnostics Forecasting Overview 29-30 th September, 2014 Agenda Introduction Overview of Global Diagnostics Forecasting Overview of Country Forecasting
Measuring Lipid Composition LC-MS/MS
 Project: Measuring Lipid Composition LC-MS/MS Verification of expected lipid composition in nanomedical controlled release systems by liquid chromatography tandem mass spectrometry AUTHORED BY: DATE: Sven
Project: Measuring Lipid Composition LC-MS/MS Verification of expected lipid composition in nanomedical controlled release systems by liquid chromatography tandem mass spectrometry AUTHORED BY: DATE: Sven
BLOOD IS COMPLEX ANALYZING IT SHOULDN T BE DxH 500 * *Not available for sale in the U.S.
 BLOOD IS COMPLEX ANALYZING IT SHOULDN T BE DxH 500 * *Not available for sale in the U.S. Introducing the DxH 500, an Open-vial Hematology System *. Quality 5-part Differential in a Compact Design The first
BLOOD IS COMPLEX ANALYZING IT SHOULDN T BE DxH 500 * *Not available for sale in the U.S. Introducing the DxH 500, an Open-vial Hematology System *. Quality 5-part Differential in a Compact Design The first
True Status A B. HTLV I Positive. Indeterminate. HTLV I/II Negative HV15 HV16 HV18 HV21 HV22 HV44 HV55. Selected Legend: Flagged: Unresolved
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 206Apr2 Panel
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 206Apr2 Panel
Hematology & Coagulation Practicum Objectives CLS - 647
 Hematology & Coagulation Practicum Objectives CLS - 647 The following objectives are to be completed by the student for successful completion of this clinical rotation. The objectives within the psychomotor
Hematology & Coagulation Practicum Objectives CLS - 647 The following objectives are to be completed by the student for successful completion of this clinical rotation. The objectives within the psychomotor
Vision and strategies to Increase Access to Innovative HIV Diagnostic Technologies. Willy Urassa. AMD STAKE HOLDERS MEETING 7-8 May 2013
 Vision and strategies to Increase Access to Innovative HIV Diagnostic Technologies Willy Urassa 1 Outline Definition Introduction Vision Broad strategies Progress so far Recent experience 2 Expansion of
Vision and strategies to Increase Access to Innovative HIV Diagnostic Technologies Willy Urassa 1 Outline Definition Introduction Vision Broad strategies Progress so far Recent experience 2 Expansion of
Contents. Preface v Reviewers vii
 Contents Preface v Reviewers vii 1 Administrative Procedures 1 1-1 Incoming Telephone Calls 1 Patient Appointments 1-2 New Patient Appointments 2 1-3 Established Patient Appointments 3 1-4 Referrals to
Contents Preface v Reviewers vii 1 Administrative Procedures 1 1-1 Incoming Telephone Calls 1 Patient Appointments 1-2 New Patient Appointments 2 1-3 Established Patient Appointments 3 1-4 Referrals to
Implementation and outcomes of point-ofcare testing in the emergency department of a large urban academic medical center
 Implementation and outcomes of point-ofcare testing in the emergency department of a large urban academic medical center Kent Lewandrowski, MD Associate Chief Of Pathology, Massachusetts General Hospital
Implementation and outcomes of point-ofcare testing in the emergency department of a large urban academic medical center Kent Lewandrowski, MD Associate Chief Of Pathology, Massachusetts General Hospital
Conversion Guide. UNIT I THE HEALTHCARE SETTING 1 2 UNIT I THE HEALTHCARE SETTING Past And Present And The
 UNIT I THE HEALTHCARE SETTING 1 2 UNIT I THE HEALTHCARE SETTING 1 2 1 Past And Present And The 3 38 1 Phlebotomy: Past And Present And 3 34 Healthcare Setting The Healthcare Setting Phlebotomy: A Historical
UNIT I THE HEALTHCARE SETTING 1 2 UNIT I THE HEALTHCARE SETTING 1 2 1 Past And Present And The 3 38 1 Phlebotomy: Past And Present And 3 34 Healthcare Setting The Healthcare Setting Phlebotomy: A Historical
EasyPlus Titration. Food & Beverage Analysis Made Easy Swiss Precision for Your Analysis
 EasyPlus Titration Food & Beverage Analysis Made Easy Swiss Precision for Your Analysis Food & Beverage Titrators Titrators for Your Samples Full Package for Your F&B Analysis The food and beverage industries
EasyPlus Titration Food & Beverage Analysis Made Easy Swiss Precision for Your Analysis Food & Beverage Titrators Titrators for Your Samples Full Package for Your F&B Analysis The food and beverage industries
For In Vitro Diagnostic Use
 SYNCHRON System(s) Chemistry Information Sheet Lactate REF A95550 For In Vitro Diagnostic Use ANNUAL REVIEW Refer to Review and Revision Coversheet at the front of each method. PRINCIPLE INTENDED USE reagent,
SYNCHRON System(s) Chemistry Information Sheet Lactate REF A95550 For In Vitro Diagnostic Use ANNUAL REVIEW Refer to Review and Revision Coversheet at the front of each method. PRINCIPLE INTENDED USE reagent,
Pathology the missing link in global health care delivery Quentin Eichbaum
 Pathology the missing link in global health care delivery Quentin Eichbaum MD, PhD, MPH, MFA, MMHC, JD, BSc(Med)(Hons), BA(Hons), FCAP, FASCP Professor or Pathology, Microbiology and Immunology Professor
Pathology the missing link in global health care delivery Quentin Eichbaum MD, PhD, MPH, MFA, MMHC, JD, BSc(Med)(Hons), BA(Hons), FCAP, FASCP Professor or Pathology, Microbiology and Immunology Professor
Rapid-VIDITEST FOB Blister
 Rapid-VIDITEST FOB Blister One Step Fecal Occult Blood Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST FOB Blister One Step Fecal Occult Blood Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565,
HI Total Acidity Mini Titrator for Water Analysis
 HI 84530 Total Acidity Mini Titrator for Water Analysis Piston Driven Pump with Dynamic Dosing The HI 84530 incorporates dynamic dosing to provide precison titrant delivery. Dynamic dosing adjusts the
HI 84530 Total Acidity Mini Titrator for Water Analysis Piston Driven Pump with Dynamic Dosing The HI 84530 incorporates dynamic dosing to provide precison titrant delivery. Dynamic dosing adjusts the
Procine sphingomyelin ELISA Kit
 Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Goal of site data management 12/2/2009. Ultimate goal: reliable and valid clinical trial to improve health
 RC-4 Overview of Clinical Site Operations and Management Unit 4: Overview of Data Quality and Monitoring Principal Investigators Lynda Wilson, PhD, RN Marti Rice, PhD, RN Program Manager TBA Course Instructors:
RC-4 Overview of Clinical Site Operations and Management Unit 4: Overview of Data Quality and Monitoring Principal Investigators Lynda Wilson, PhD, RN Marti Rice, PhD, RN Program Manager TBA Course Instructors:
i-stat CHEM8 + Cartridge Quick Reference Guide
 i-stat CHEM8 + Cartridge Quick Reference Guide For use with a Certificate of Waiver The CHEM8+ cartridge has tests for sodium, potassium, chloride, total carbon dioxide, ionized calcium, glucose, urea
i-stat CHEM8 + Cartridge Quick Reference Guide For use with a Certificate of Waiver The CHEM8+ cartridge has tests for sodium, potassium, chloride, total carbon dioxide, ionized calcium, glucose, urea
Bruce Thomadsen. University of Wisconsin - Madison
 Root Cause Analysis Bruce Thomadsen University of Wisconsin - Madison Analysis Assume you have an event. What to do? The analysis would like to get to the root cause. RCA Team First, assemble an root-cause
Root Cause Analysis Bruce Thomadsen University of Wisconsin - Madison Analysis Assume you have an event. What to do? The analysis would like to get to the root cause. RCA Team First, assemble an root-cause
