Patients with type 2 diabetes are commonly
|
|
|
- Roger Caldwell
- 6 years ago
- Views:
Transcription
1 ORIGINAL ARTICLE Interaction of Oral Antidiabetic Drugs With Hepatic Uptake Transporters Focus on Organic Anion Transporting Polypeptides and Organic Cation Transporter Iouri Bachmakov, Hartmut Glaeser, Martin F. Fromm, and Jörg König OBJECTIVE The uptake of drugs into hepatocytes is a key determinant for hepatic metabolism, intrahepatic action, their subsequent systemic plasma concentrations, and extrahepatic actions. In vitro and in vivo studies indicate that many drugs used for treatment of cardiovascular diseases (e.g., oral antidiabetic drugs, statins) are taken up into hepatocytes by distinct organic anion transporters (organic anion transporting polypeptides [OATPs]; gene symbol SLCO/SLC2) or organic cation transporters (OCTs; gene symbol SLC22). Because most patients with type 2 diabetes receive more than one drug and inhibition of drug transporters has been recognized as a new mechanism underlying drug-drug interactions, we tested the hypothesis of whether oral antidiabetic drugs can inhibit the transport mediated by hepatic uptake transporters. RESEARCH DESIGN AND METHODS Using stably transfected cell systems recombinantly expressing the uptake transporters OATPB, OATPB3, OATP2B, or OCT, we analyzed whether the antidiabetic drugs repaglinide, rosiglitazone, or metformin influence the transport of substrates and drugs (for OATPs, sulfobromophthalein [BSP] and pravastatin; for OCT, -methyl-4-phenylpyridinium [MPP ] and metformin). RESULTS Metformin did not inhibit the uptake of OATP and OCT substrates. However, OATP-mediated BSP and pravastatin uptake and OCT-mediated MPP and metformin uptake were significantly inhibited by repaglinide (half-maximal inhibitory concentration [IC 5 ] mol/l) and rosiglitazone (IC mol/l). CONCLUSIONS These in vitro results demonstrate that alterations of uptake transporter function by oral antidiabetic drugs have to be considered as potential mechanisms underlying drug-drug interactions. Diabetes 57: , 28 From the Institute of Experimental and Clinical Pharmacology and Toxicology, Friedrich-Alexander-University Erlangen-Nuremberg, Erlangen, Germany. Corresponding author: Dr. Jörg König, Institute of Experimental and Clinical Pharmacology and Toxicology, Friedrich-Alexander-University Erlangen-Nuremberg, Fahrstrasse 7, 954 Erlangen, Germany. joerg. koenig@pharmakologie.med.uni-erlangen.de. Received for publication October 27 and accepted in revised form 2 February 28. Published ahead of print at on 26 February 28. DOI:.2337/db7-55. BSP, sulfobromophthalein; HEK293 cell, human embryonic kidney cell; IC 5, half-maximal inhibitory concentration; MDCKII cell, Madin-Darby canine kidney cell; MPP, -methyl-4-phenylpyridinium; OATP, organic anion transporting polypeptide; OCT, organic cation transporter. 28 by the American Diabetes Association. The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked advertisement in accordance with 8 U.S.C. Section 734 solely to indicate this fact. Patients with type 2 diabetes are commonly treated with more than one drug. It is therefore essential to understand mechanisms underlying drug-drug interactions, which might cause changes in the pharmacokinetics and effects of these drugs. It is now well established that drug transporters are major determinants of drug disposition and effects (,2). Moreover, inhibition or induction of drug transporters are newly recognized mechanisms of drug-drug interactions (3 5). Oral antidiabetic drugs need to be taken up from the portal venous blood via the basolateral membrane into hepatocytes before they are metabolized in the cell, cause drug effects via intrahepatic mechanisms, or are further transported back into the systemic circulation for extrahepatic effects (6 9). Recent in vitro and in vivo studies highlighted that the transporter-mediated uptake of oral antidiabetic drugs is an important determinant for their disposition and effects (9 ). These uptake mechanisms are also important for disposition and action of a broad variety of cardiovascular drugs frequently used concomitantly in patients with type 2 diabetes (e.g., statins, ACE inhibitors, and angiotensin type receptor blockers) (2). This study focuses on uptake transporters belonging to the organic anion transporting polypeptide (OATP; gene symbol SLCO/SLC2) and organic cation transporter (OCT; gene symbol SLC22) families. Members of the OATP family transport a variety of anionic endogenous substances and drugs, including hydroxymethylglutaryl-coa reductase inhibitors, such as fluvastatin, pitavastatin, pravastatin, and rosuvastatin (2,3). Furthermore, it has been shown that glitazones and repaglinide may be substrates for the hepatocyte-specific OATP family member OATPB (9,4). OATPB, OATPB3, and OATP2B are localized in the basolateral membrane of human hepatocytes mediating the uptake of endogenous substances and drugs from the portal venous blood (5 7). Members of the OCT family play essential roles in the handling of cationic drugs and endogenously synthesized organic cations. Human OCT is expressed primarily in the liver, localized in the basolateral membrane of hepatocytes mediating the hepatic uptake of several cationic drugs (e.g., metformin). In addition, various drugs (e.g., cimetidine, desipramine, midazolam, citalopram, and clonidine) have been identified to inhibit OCT-mediated uptake (8). Despite the increasingly recognized role of hepatic uptake transporters for drug disposition, it has not been DIABETES, VOL. 57, JUNE
2 ORAL ANTIDIABETIC DRUGS AND UPTAKE TRANSPORTERS TABLE Comparison of pharmacokinetic data of oral antidiabetic drugs in humans with IC 5 values of OATPB-, OATPB3-, and OATP2B-mediated BSP uptake and OCT-mediated MPP uptake obtained in the present study A B C MPP + uptake [pmol x mg - x min - ] MPP + net uptake [pmol x mg - x min - ] Metformin uptake [pmol x mg- x min - ] Dosage (mg) 5 MPP Cmax (mg/l) MPP + 5 µm K m = 2. µm Control 2 3 OCT 4 * 5 MPP + concentration [µm] 4 metformin * Control Cmax Cport. vn. OCT IC 5 OATPB IC 5 OATPB3 IC 5 OATP2B IC 5 OCT References Metformin (BSP) (BSP) (BSP), (MPP ) 4 Repaglinide (BSP) 5.6 (BSP) 5.2 (BSP).8 (MPP ) 42 (pravastatin)* (pravastatin)*.6 (metformin) Rosiglitazone (BSP).9 (BSP) 5.2 (BSP) 3.4 (MPP ) 43 (pravastatin) (pravastatin) 6.9 (metformin) IC 5 data are derived from the measurements shown in Figures 3 8; respective tested substrates are indicated in parentheses. *IC 5 value was not determined, but there is an inhibition at all inhibitor concentrations investigated. IC 5 value was not determined, but there is an inhibition at the highest inhibitor concentration tested. Cmax, maximal plasma concentration in the systemic circulation; Cport.vn., predicted concentration in portal venous blood (according to Ito et al. 35 ). studied systematically whether the oral antidiabetic drugs repaglinide, rosiglitazone, and metformin, which have previously been shown to interact with OATPs or OCTs (9,4,8), are inhibitors of hepatic uptake transport proteins. To gain more insights into the potential role of uptake transporters for antidiabetic drug mediated drugdrug interactions, we used cell systems stably expressing the human uptake transporters OATPB, OATPB3, OATP2B, or OCT and tested the influence of these oral antidiabetic drugs on the uptake of OATP substrates and OCT substrates. RESEARCH DESIGN AND METHODS Chemicals and antibodies. [ 3 H]sulfobromophthalein ([ 3 H]BSP; 7,585 GBq/ mmol) was obtained from Hartmann Analytic (Braunschweig, Germany). [ 3 H]-methyl-4-phenylpyridinium ([ 3 H]MPP ; 85 Ci/mmol), [ 3 H]rosiglitazone (5 Ci/mmol), and unlabeled rosiglitazone were obtained from BIOTREND Chemikalien (Cologne, Germany). Unlabeled metformin and poly-d-lysine hydrobromide were purchased from Sigma-Aldrich Chemie (Taufkirchen, Germany). Unlabeled repaglinide was obtained from Chemos (Regenstauf, Germany). [ 4 C]metformin hydrochloride (. Ci/mmol) was provided by Merck (Darmstadt, Germany). Methanol (hypergrade), n-hexane (pro-analysis), acetonitrile (hypergrade), and acetic acid (supra-pure) were purchased from Merck. Diethyl ether (99.8% purity), ammonium acetate (pro-analysis), and ibuprofen were obtained from Sigma-Aldrich Chemie. Cell culture. Stably transfected human embryonic kidney cells (HEK293 cells) and Madin-Darby canine kidney cells (MDCKII cells) were cultured as described previously (5,9). For uptake and inhibition experiments, stably transfected HEK293 cells recombinantly expressing human OATPB (HEK- OATPB), human OATPB3 (HEK-OATPB3) (5), or human OATP2B (HEK- OATP2B) and the respective HEK control cells (HEK-Co/48, transfected with the empty expression vector pcdna3.; or HEK-Co/Hy, transfected with the empty expression vector pcdna3./hygro) were seeded in. mg/ml poly-d-lysine coated 2-well plates (Cell Culture Multiwell Plate Cellstar; Greiner Bio-One, Frickenhausen, Germany) at an initial density of 7, cells/well. The cells were grown to confluence for day and then induced with mmol/l sodium butyrate (Merck) for 24 h before the uptake experiments to obtain higher levels of the recombinant protein (2). MDCKII cells were transfected with the respective plasmid pcdna3./ Hygro( )-OCT containing the full-length cdna encoding the human OCT- protein (NM_357) using Effectene transfection reagent (Qiagen, Hilden, Germany). After hygromycin (8 g/ml) selection, single colonies were characterized for SLC22A (encoding human OCT) mrna expression by real-time PCR analysis. Vector transfected MDCKII control cells were estab- FIG.. Characterization of stably transfected MDCK cells. A: Intracellular MPP accumulation in MDCKII-OCT and MDCKII-control (Control) cells after -min incubation with 5 mol/l MPP. B: Net intracellular MPP accumulation in MDCKII-OCT cells after -min incubation with increasing MPP concentrations. The uptake was obtained by subtracting the uptake in vector-transfected cells (control) from that in OCT-expressing cells. K m and V max values were calculated by fitting the data to a one-site binding curve. C: Intracellular metformin accumulation in MDCKII-OCT and MDCKII-control (Control) cells after -min incubation with mol/l metformin. Each value is the mean SE (n 3). *P <. vs. control. 464 DIABETES, VOL. 57, JUNE 28
3 I. BACHMAKOV AND ASSOCIATES Repaglinide Rosiglitazone Metformin [ 3 H]BSP transport (%) [ 3 H]BSP transport (%) [ 3 H]BSP transport (%) IC 5 = 2.2 µm IC 5 = 6. µm 5 5 IC 5 = 5.6 µm IC 5 =.9 µm 5 5 IC 5 = 5.2 µm IC 5 = 5.2 µm OATPB OATPB3 OATP2B Repaglinide Concentration [µm] Rosiglitazone Concentration [µm] Metformin Concentration [µm] FIG. 2. Inhibition of OATP-mediated BSP uptake by oral antidiabetic drugs. Inhibitory effect of repaglinide, rosiglitazone, and metformin on OATPB-, OATPB3-, and OATP2B-mediated BSP uptake after -min incubation. IC 5 values were calculated by fitting the data to a sigmoidal dose-response regression curve. Data are shown as the percentage of the mediated BSP uptake in the absence of oral antidiabetic drugs. Each value is the mean SE (n 3). lished by the same method using the respective expression plasmid without insert for transfection. For MPP uptake experiments, MDCKII cells were seeded in. mg/ml poly-d-lysine coated 2-well plates (Cell Culture Multiwell Plate Cellstar; Greiner Bio-One) at an initial density of 7, cells per well. The cells were grown to confluence for 2 days and induced with mmol/l sodium butyrate (Merck) for 24 h before the uptake experiments (2). Uptake assays. Before starting the uptake experiments with the HEK cells, cells were washed with prewarmed (37 C) uptake buffer (42 mmol/l NaCl, 5 mmol/l KCl, mmol/l K 2 HPO 4,.2 mmol/l MgSO 4,.5 mmol/l CaCl 2, 5 mmol/l glucose, and 2.5 mmol/l HEPES, ph 7.3). [ 3 H]BSP was dissolved in uptake buffer, and unlabeled BSP was added to the final concentration of.5 mol/l BSP for studies with HEK-OATPB and mol/l BSP for studies with HEK-OATPB3 and HEK-OATP2B, respectively. To characterize repaglinide and rosiglitazone as inhibitors of OATP-mediated uptake, both drugs were added in increasing concentrations (. mol/l). The cells were incubated with the solution at 37 C for min as described previously (5). Afterward, the cells were washed three times with ice-cold uptake buffer before lysing the cells with.2% SDS. The intracellular accumulation of radioactivity was detected by liquid scintillation counting (Perkin Elmer Life Sciences). For the experiments using pravastatin as substrate for OATPB and OATPB3, a concentration of 5 mol/l pravastatin was used. To test the inhibitory effect of repaglinide and rosiglitazone, each drug was added in concentrations of and mol/l. The uptake assay was performed as described above. The intracellular pravastatin concentration was determined by liquid chromatography with tandem mass spectrometry detection analysis as previously described (5). The uptake experiments with MDCKII-OCT cells were carried out in an analogous manner. [ 3 H]MPP was dissolved in uptake buffer, and unlabeled MPP was added to the final concentration of 3 mol/l. To characterize repaglinide, rosiglitazone, and metformin as inhibitors of OCT-mediated uptake, the drugs were added in increasing concentrations of., mol/l for metformin and. mol/l for repaglinide and rosiglitazone, respectively. For experiments using metformin as substrate of OCT, [ 4 C]metformin was dissolved in uptake buffer, and unlabeled metformin was added to a final concentration of mol/l. To test the inhibitory effect of repaglinide and rosiglitazone, each drug was added in a concentration of. mol/l. All experiments were repeated at least three times. Data analysis. The OATPB-, OATPB3-, OATP2B-, and OCT-mediated net uptake was obtained by subtracting the uptake in vector-transfected cells DIABETES, VOL. 57, JUNE
4 ORAL ANTIDIABETIC DRUGS AND UPTAKE TRANSPORTERS from that in OATPB-, OATPB3-, OATP2B-, and OCT-expressing cells. The percentage of uptake inhibition was calculated from control experiments in the absence of antidiabetic drugs (% uptake). The corresponding half-maximal inhibitory concentration (IC 5 ) values for uptake inhibition were calculated by fitting the data to a sigmoidal dose-response regression curve (Prism 4. 24; GraphPad Software, San Diego, CA). Effects of repaglinide and rosiglitazone on pravastatin uptake in OATPB and OATPB3 cells were analyzed with unpaired two-tailed t test (Prism 4. 24; GraphPad Software). A value of P.5 was required for statistical significance. RESULTS Characterization of MDCKII-OCT cells. To examine the inhibitory potency of antidiabetic drugs on OCT- mediated uptake, MDCKII cells were stably transfected with the SLC22A cdna and selected for a high expression of the uptake transporter OCT. The SLC22A mrna expression of the selected cell clones was analyzed using quantitative real-time PCR. This analysis demonstrated a high SLC22A mrna expression in several MDCKII-OCT clones compared with vector-transfected cells. The cell clone with the highest mrna expression was used for further transport experiments using the prototypic tritiumlabeled substrate [ 3 H]MPP.MPP was shown to be a high-affinity substrate for OCT with a K m value of 2 mol/l, which is in accordance with previously published data (8). The uptake experiments (Fig. A and B) demonstrated that MDCKII-OCT cells were able to mediate uptake of MPP (5 mol/l) into cells with an uptake ratio of 3.7-fold compared with control cells. Furthermore, we confirmed that metformin, previously shown to be a substrate for OCT with a K m value of,47 mol/l (8), is also transported by the newly established MDCKII-OCT cells (Fig. C). Inhibition of OATPs and OCT by antidiabetic drugs. The results of the inhibition experiments are summarized in Table and Figs Metformin did not inhibit OATPB-, OATPB3-, or OATP2B-mediated BSP uptake (Fig. 2) or uptake of the OCT substrate MPP (Fig. 4). However, OATP-mediated BSP and pravastatin uptake and OCT-mediated MPP and metformin uptake were significantly inhibited by repaglinide (Figs. 2 4; Table ) and rosiglitazone (Figs. 2 4; Table ). DISCUSSION Patients with type 2 diabetes frequently have to be treated with more than one drug. Effects of oral antidiabetic drugs depend on the extent of drug absorption from the gut lumen, on metabolism of the drug in the liver, and on the extent of its excretion into bile and urine (2). In general, modification of all these processes by a second, concomitantly administered drug can alter the effects of oral antidiabetic drugs (2). Recently, it was recognized that a broad variety of drugs, including many cardiovascular drugs such as statins and angiotensin II-receptor antagonists, is transported through biological membranes via specific transport proteins (5,22 24). For example, the efflux transporter P- glycoprotein, which translocates its substrates from the inside of the cell to the outside (e.g., from the hepatocyte into bile) is a major determinant of drug effects (). If P-glycoprotein mediated drug excretion is inhibited by a second, concomitantly administered compound, drug plasma concentrations increase significantly and may result in drug toxicity (3). In addition to efflux transporters (e.g., P-glycoprotein), uptake transporters such as the OATPs or the OCTs are A Pravastatin uptake [%] B Pravastatin uptake [%] OATPB OATPB3 Control µm µm + Repaglinide µm µm + Rosiglitazone FIG. 3. Inhibition of the OATPB- and OATPB3-mediated pravastatin uptake by repaglinide and rosiglitazone. Inhibitory effect using and mol/l concentrations of the oral antidiabetic drugs repaglinide and rosiglitazone on OATPB-mediated (A) and OATPB3- mediated (B) 5 mol/l pravastatin uptake after -min incubation. Data are shown as the percentage of the transporter-mediated pravastatin uptake in the absence of oral antidiabetic drugs (Control). Each value is the mean SE (n 3). *P <.5; P <. vs. control. important for drug disposition. For example, they mediate the uptake of multiple cardiovascular drugs from the portal venous blood into the hepatocytes (2,8). This transport process is an essential prerequisite for subsequent drug metabolism in the hepatocytes. The goal of our in vitro study was to investigate whether the oral antidiabetic drugs repaglinide, rosiglitazone, and metformin are inhibitors of the hepatic uptake transporters of the OATP family (OATPB, OATPB3, and OATP2B) and of the OCT family member OCT. Whereas metformin did not affect the function of hepatic uptake transporters, both repaglinide and rosiglitazone were significant inhibitors of organic anion and organic cation transport. The following clinical consequences can result from the observed interaction with repaglinide and rosiglitazone. First, if hepatic uptake of a drug (e.g., pravastatin) is inhibited by repaglinide and rosiglitazone and possibly by further oral antidiabetic drugs, the first step (i.e., uptake into hepatocytes) of biliary drug elimination is blocked. This causes increased plasma concentrations and an increased risk of adverse drug reactions. The importance of an impaired uptake transporter function is convincingly shown for OATPB. One polymorphism (c.52t C) in the gene encoding for OATPB results in a significantly reduced OATPB function (), a situation that is comparable to the OATPB function in the pres- * 466 DIABETES, VOL. 57, JUNE 28
5 I. BACHMAKOV AND ASSOCIATES A repaglinide rosiglitazone metformin [ 3 H]MPP + transport [%] 5 IC 5 =.8 µm 5 IC 5 = 3.4 µm 5.. Repaglinide concentration [µm].. Rosiglitazone concentration [µm].. Metformin concentration [µm] B [ 4 C]Metformin transport (%) 5 repaglinide IC 5 =.6 µm. Repaglinide concentration [µm] 5. IC 5 = 6.9 µm rosiglitazone Rosiglitazone concentration [µm] FIG. 4. Inhibition of MPP and metformin uptake by oral antidiabetic drugs in MCDKII- OCT cells. A: Inhibitory effect of repaglinide, rosiglitazone, and metformin on OCT- mediated MPP (3 mol/l) uptake after - min incubation. Data are shown as the percentage of the OCT-mediated MPP uptake in the absence of repaglinide, rosiglitazone, or metformin. B: Inhibitory effect of repaglinide and rosiglitazone on OCT-mediated metformin ( mol/l) uptake after - min incubation. Data are shown as the percentage of the OCT-mediated metformin uptake in the absence of repaglinide or rosiglitazone. IC 5 values were calculated by fitting the data to a sigmoidal dose-response regression curve. Each value is the mean SE (n 4). ence of drugs, which inhibit this uptake transporter. Several studies showed that subjects carrying this polymorphism with the reduced uptake transporter function have significantly higher plasma concentrations of pravastatin (26 29). Interestingly, for other drugs, no influence of this genetic variation could be observed (3). This could be due to an overlapping substrate spectrum with OATPB3, possibly compensating for a reduced uptake mediated by a mutated OATPB protein. A second clinical consequence of decreased hepatic drug uptake is a reduced effect of the affected drug (e.g., pravastatin). This option applies if the drug effect (e.g., for pravastatin, cholesterol-lowering effect) is mediated via mechanisms within the hepatocyte. A genetically determined reduced hepatic uptake of pravastatin was recently shown to cause a reduced effect of pravastatin (3 33). The third possible clinical consequence of inhibition of uptake transporter function is increased extrahepatic effects (e.g., for statins, an increased risk of myotoxicity). This reduced benefit (cholesterol-lowering effect)-to-risk (myotoxicity) ratio for OATPB-dependent statins by inhibition of hepatic uptake was recently highlighted in an excellent review by Neuvonen et al. (34). It should be noted that our study focused on in vitro studies on mechanisms of drug interactions with oral antidiabetic drugs. As outlined below, the clinical consequences (e.g., pharmacodynamic consequences) are difficult to predict and require further clinical studies. Another interesting observation of our study was the unexpected stimulation of OATPB3-mediated pravastatin uptake by rosiglitazone (Fig. 3). Similar observations have been made for other substrates (5); however, the molecular mechanisms underlying this effect are unclear and require further studies. As drugs reach the portal vein directly after intestinal absorption, the drug concentration in portal venous blood is higher than in the systemic circulation. For estimation of the drug concentrations in portal venous blood, we used the method of Ito et al. (35) (Table ). The predicted portal venous blood concentrations for repaglinide and rosiglitazone are in the same low micromolar range as the determined IC 5 values for inhibition of hepatic uptake, suggesting that inhibition of hepatic drug transporters might cause clinically relevant drug-drug interactions in humans. To the best of our knowledge, no clinical studies have, so far, been conducted to investigate an impact of repaglinide or rosiglitazone on disposition of pravastatin or other OATP substrates in humans. Pharmacodynamic interactions between metformin and rosiglitazone (36,37) or repaglinide (38) in patients have intensively been investigated. For both rosiglitazone and repaglinide, additional effects on glucose metabolism in patients with type 2 diabetes receiving simultaneously metformin have been DIABETES, VOL. 57, JUNE
6 ORAL ANTIDIABETIC DRUGS AND UPTAKE TRANSPORTERS shown. Whether these effects can in part be explained by an interaction with hepatic metformin uptake is presently unclear. One study examined the pharmacokinetic interaction between rosiglitazone and metformin in humans (39), however, without a significant pharmacokinetic interaction between these drugs. The question arises of whether our findings on inhibition of uptake transporter function by oral antidiabetic drugs can be used to draw general conclusions regarding drug-drug interactions. Similar to the present data, macrolid antibiotics were previously shown to cause a relevant inhibition of hepatic uptake transporters (5). Moreover, there is evidence that other oral antidiabetic drugs (glibenclamide) are inhibitors of hepatic uptake transporters (4). Thus, our in vitro data generated in this study indicate that inhibition of uptake transporters by oral antidiabetic drugs is a newly recognized mechanism of potential drug-drug interactions. Our data provide the basis for controlled clinical studies to clarify the importance of drug-drug interactions with oral antidiabetic drugs in studies with healthy volunteers and patients. ACKNOWLEDGMENTS This work was supported by Deutsche Forschungsgemeinschaft Grant DFG Ko 22/-3 and Grant DFG FR 298/5-, by Deutsche Krebshilfe Grant 7854, and by the Johannes and Frieda Marohn-Foundation. We thank B. Endress, C. Hoffmann, and K. Singer for excellent technical assistance. We thank Dr. D. Keppler (German Cancer Research Center, Heidelberg, Germany) for providing the plasmids pcdna3.( )-OATPB and pcdna3./hygro( )-OATPB3. REFERENCES. Fromm MF: Importance of P-glycoprotein at blood-tissue barriers. Trends Pharmacol Sci : , Ho RH, Kim RB: Transporters and drug therapy: implications for drug disposition and disease. Clin Pharmacol Ther 78:26 277, 3. Fromm MF, Kim RB, Stein CM, Wilkinson GR, Roden DM: Inhibition of P-glycoprotein-mediated drug transport: a unifying mechanism to explain the interaction between digoxin and quinidine. Circulation 99: , Greiner B, Eichelbaum M, Fritz P, Kreichgauer HP, von Richter O, Zundler J, Kroemer HK: The role of intestinal P-glycoprotein in the interaction of digoxin and rifampin. J Clin Invest 4:47 53, Seithel A, Eberl S, Singer K, Auge D, Heinkele G, Wolf NB, Dörje F, Fromm MF, König J: The influence of macrolide antibiotics on the uptake of organic anions and drugs mediated by OATPB and OATPB3. Drug Metab Dispos 35: , Bailey CJ, Turner RC: Metformin. N Engl J Med 334: , Balfour JA, Plosker GL: Rosiglitazone. Drugs 57:92 93, 999; discussion Kajosaari LI, Laitila J, Neuvonen PJ, Backman JT: Metabolism of repaglinide by CYP2C8 and CYP3A4 in vitro: effect of fibrates and rifampicin. Basic Clin Pharmacol Toxicol 97:249 6, 9. Niemi M, Backman JT, Kajosaari LI, Leathart JB, Neuvonen M, Daly AK, Eichelbaum M, Kivisto KT, Neuvonen PJ: Polymorphic organic anion transporting polypeptide B is a major determinant of repaglinide pharmacokinetics. Clin Pharmacol Ther 77: ,. Shu Y, Brown C, Castro RA, Shi RJ, Lin ET, Owen RP, Sheardown SA, Yue L, Burchard EG, Brett CM, Giacomini KM: Effect of genetic variation in the organic cation transporter, OCT, on metformin pharmacokinetics. Clin Pharmacol Ther 83:273 28, 28. Shu Y, Sheardown SA, Brown C, Owen RP, Zhang S, Castro RA, Ianculescu AG, Yue L, Lo JC, Burchard EG, Brett CM, Giacomini KM: Effect of genetic variation in the organic cation transporter (OCT) on metformin action. J Clin Invest 7:422 43, König J, Seithel A, Gradhand U, Fromm MF: Pharmacogenomics of human OATP transporters. Naunyn Schmiedebergs Arch Pharmacol 372: , Hagenbuch B, Meier PJ: The superfamily of organic anion transporting polypeptides. Biochim Biophys Acta 69: 8, Nozawa T, Sugiura S, Nakajima M, Goto A, Yokoi T, Nezu J, Tsuji A, Tamai I: Involvement of organic anion transporting polypeptides in the transport of troglitazone sulfate: implications for understanding troglitazone hepatotoxicity. Drug Metab Dispos 32:29 294, Hsiang B, Zhu Y, Wang Z, Wu Y, Sasseville V, Yang WP, Kirchgessner TG: A novel human hepatic organic anion transporting polypeptide (OATP2): identification of a liver-specific human organic anion transporting polypeptide and identification of rat and human hydroxymethylglutaryl-coa reductase inhibitor transporters. J Biol Chem 274: , König J, Cui Y, Nies AT, Keppler D: Localization and genomic organization of a new hepatocellular organic anion transporting polypeptide. J Biol Chem 2: , 2 7. König J, Cui Y, Nies AT, Keppler D: A novel human organic anion transporting polypeptide localized to the basolateral hepatocyte membrane. Am J Physiol Gastrointest Liver Physiol 278:G56 G64, 2 8. Koepsell H, Lips K, Volk C: Polyspecific organic cation transporters: structure, function, physiological roles, and biopharmaceutical implications. Pharm Res 24:227, Letschert K, Keppler D, König J: Mutations in the SLCOB3 gene affecting the substrate specificity of the hepatocellular uptake transporter OATPB3 (OATP8). Pharmacogenetics 4:44 452, Cui Y, König J, Buchholz JK, Spring H, Leier I, Keppler D: Drug resistance and ATP-dependent conjugate transport mediated by the apical multidrug resistance protein, MRP2, permanently expressed in human and canine cells. Mol Pharmacol 55: , Levy RH, Thummel KE, Trager WF, Hansten PD, Eichelbaum M: Metabolic Drug Interactions. London, Lippincott Williams & Wilkins, Hochman JH, Pudvah N, Qiu J, Yamazaki M, Tang C, Lin JH, Prueksaritanont T: Interactions of human P-glycoprotein with simvastatin, simvastatin acid, and atorvastatin. Pharm Res 2:686 69, Ishiguro N, Maeda K, Kishimoto W, Saito A, Harada A, Ebner T, Roth W, Igarashi T, Sugiyama Y: Predominant contribution of OATPB3 to the hepatic uptake of telmisartan, an angiotensin II receptor antagonist, in humans. Drug Metab Dispos 34:9 5, Yamashiro W, Maeda K, Hirouchi M, Adachi Y, Hu Z, Sugiyama Y: Involvement of transporters in the hepatic uptake and biliary excretion of valsartan, a selective antagonist of the angiotensin II AT-receptor, in humans. Drug Metab Dispos 34:247 4, 26. Tirona RG, Leake BF, Merino G, Kim RB: Polymorphisms in OATP-C: identification of multiple allelic variants associated with altered transport activity among European- and African-Americans. J Biol Chem 276: , Igel M, Arnold KA, Niemi M, Hofmann U, Schwab M, Lütjohann D, von Bergmann K, Eichelbaum M, Kivistö KT: Impact of the SLCOB polymorphism on the pharmacokinetics and lipid-lowering efficacy of multipledose pravastatin. Clin Pharmacol Ther 79:49 426, Mwinyi J, Johne A, Bauer S, Roots I, Gerloff T: Evidence for inverse effects of OATP-C (SLC2A6) 5 and b haplotypes on pravastatin kinetics. Clin Pharmacol Ther :45 42, Niemi M, Schaeffeler E, Lang T, Fromm MF, Neuvonen M, Kyrklund C, Backman JT, Kerb R, Schwab M, Neuvonen PJ, Eichelbaum M, Kivistö KT: High plasma pravastatin concentrations are associated with single nucleotide polymorphisms and haplotypes of organic anion transporting polypeptide-c (OATP-C, SLCOB). Pharmacogenetics 4:429 44, Nishizato Y, Ieiri I, Suzuki H, Kimura M, Kawabata K, Hirota T, Takane H, Irie S, Kusuhara H, Urasaki Y, Urae A, Higuchi S, Otsubo K, Sugiyama Y: Polymorphisms of OATP-C (SLC2A6) and OAT3 (SLC22A8) genes: consequences for pravastatin pharmacokinetics. Clin Pharmacol Ther 73: , Kalliokoski A, Neuvonen M, Neuvonen PJ, Niemi M: No significant effect of SLCOB polymorphism on the pharmacokinetics of rosiglitazone and pioglitazone. Br J Clin Pharmacol 65:78 86, Kivistö KT, Niemi M: Influence of drug transporter polymorphisms on pravastatin pharmacokinetics in humans. Pharm Res 24: , Niemi M, Neuvonen PJ, Hofmann U, Backman JT, Schwab M, Lütjohann D, von Bergmann K, Eichelbaum M, Kivistö KT: Acute effects of pravastatin on cholesterol synthesis are associated with SLCOB (encoding OATPB) haplotype *7. Pharmacogenet Genomics 5:33 39, 33. Tachibana-Iimori R, Tabara Y, Kusuhara H, Kohara K, Kawamoto R, Nakura J, Tokunaga K, Kondo I, Sugiyama Y, Miki T: Effect of genetic polymorphism of OATP-C (SLCOB) on lipid-lowering response to HMG- CoA reductase inhibitors. Drug Metab Pharmacokinet 9:3 38, DIABETES, VOL. 57, JUNE 28
7 I. BACHMAKOV AND ASSOCIATES 34. Neuvonen PJ, Niemi M, Backman JT: Drug interactions with lipid-lowering drugs: mechanisms and clinical relevance. Clin Pharmacol Ther 8:565 58, Ito K, Iwatsubo T, Kanamitsu S, Ueda K, Suzuki H, Sugiyama Y: Prediction of pharmacokinetic alterations caused by drug-drug interactions: metabolic interaction in the liver. Pharmacol Rev 5:387 42, Fonseca V, Rosenstock J, Patwardhan R, Salzman A: Effect of metformin and rosiglitazone combination therapy in patients with type 2 diabetes mellitus: a randomized controlled trial. JAMA 283:695 72, Gomez-Perez FJ, Fanghanel-Salmon G, Antonio Barbosa J, Montes-Villarreal J, Berry RA, Warsi G, Gould EM: Efficacy and safety of rosiglitazone plus metformin in Mexicans with type 2 diabetes. Diabete Metab Res Rev 8:27 34, Moses R, Slobodniuk R, Boyages S, Colagiuri S, Kidson W, Carter J, Donnelly T, Moffitt P, Hopkins H: Effect of repaglinide addition to metformin monotherapy on glycemic control in patients with type 2 diabetes. Diabetes Care 22:9 24, Di Cicco RA, Allen A, Carr A, Fowles S, Jorkasky DK, Freed MI: Rosiglitazone does not alter the pharmacokinetics of metformin. J Clin Pharmacol 4:28 285, 2 4. Grube M, Kock K, Oswald S, Draber K, Meissner K, Eckel L, Bohm M, Felix SB, Vogelgesang S, Jedlitschky G, Siegmund W, Warzok R, Kroemer HK: Organic anion transporting polypeptide 2B is a high-affinity transporter for atorvastatin and is expressed in the human heart. Clin Pharmacol Ther 8:67 62, GLUCOPHAGE GX: Metformin hydrochloride extended-release tablets [article online]. Princeton, NJ, Bristol-Myers Squibb Company. Available from Accessed 2 June Hatorp V, Walther KH, Christensen MS, Haug-Pihale G: Single-dose pharmacokinetics of repaglinide in subjects with chronic liver disease. J Clin Pharmacol 4:42 52, Kirchheiner J, Thomas S, Bauer S, Tomalik-Scharte D, Hering U, Doroshyenko O, Jetter A, Stehle S, Tsahuridu M, Meineke I, Brockmoller J, Fuhr U: Pharmacokinetics and pharmacodynamics of rosiglitazone in relation to CYP2C8 genotype. Clin Pharmacol Ther 8: , 26 DIABETES, VOL. 57, JUNE
polymorphism on repaglinide pharmacokinetics persists over a wide dose range
 British Journal of Clinical Pharmacology DOI:./j.365-5.8.387.x The effect of SLCOB polymorphism on repaglinide pharmacokinetics persists over a wide dose range Annikka Kalliokoski, Mikko Neuvonen, Pertti
British Journal of Clinical Pharmacology DOI:./j.365-5.8.387.x The effect of SLCOB polymorphism on repaglinide pharmacokinetics persists over a wide dose range Annikka Kalliokoski, Mikko Neuvonen, Pertti
The Influence of Macrolide Antibiotics on the Uptake of Organic Anions and Drugs Mediated by OATP1B1 and OATP1B3
 0090-9556/07/3505-779 786$20.00 DRUG METABOLISM AND DISPOSITION Vol. 35, No. 5 Copyright 2007 by The American Society for Pharmacology and Experimental Therapeutics 14407/3199708 DMD 35:779 786, 2007 Printed
0090-9556/07/3505-779 786$20.00 DRUG METABOLISM AND DISPOSITION Vol. 35, No. 5 Copyright 2007 by The American Society for Pharmacology and Experimental Therapeutics 14407/3199708 DMD 35:779 786, 2007 Printed
Influence of Non-Steroidal Anti-Inflammatory Drugs on Organic Anion Transporting Polypeptide (OATP) 1B1- and OATP1B3- Mediated Drug Transport
 0090-9556/11/3906-1047 1053$25.00 DRUG METABOLISM AND DISPOSITION Vol. 39, No. 6 Copyright 2011 by The American Society for Pharmacology and Experimental Therapeutics 37622/3688980 DMD 39:1047 1053, 2011
0090-9556/11/3906-1047 1053$25.00 DRUG METABOLISM AND DISPOSITION Vol. 39, No. 6 Copyright 2011 by The American Society for Pharmacology and Experimental Therapeutics 37622/3688980 DMD 39:1047 1053, 2011
Influence of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) on. OATP1B1- and OATP1B3-mediated Drug Transport
 DMD Fast This article Forward. has not been Published copyedited on and March formatted. 9, The 2011 final version as doi:10.1124/dmd.110.037622 may differ from this version. DMD # 37622 Influence of Non-Steroidal
DMD Fast This article Forward. has not been Published copyedited on and March formatted. 9, The 2011 final version as doi:10.1124/dmd.110.037622 may differ from this version. DMD # 37622 Influence of Non-Steroidal
Pharmacokinetic Determinants of Statin-Induced Myopathy
 Pharmacokinetic Determinants of Statin-Induced Myopathy Rommel G. Tirona, B.Sc.Phm., Ph.D. Departments of Physiology & Pharmacology and Medicine The University of Western Ontario, London, Ontario, Canada
Pharmacokinetic Determinants of Statin-Induced Myopathy Rommel G. Tirona, B.Sc.Phm., Ph.D. Departments of Physiology & Pharmacology and Medicine The University of Western Ontario, London, Ontario, Canada
In vitro substrate-dependent inhibition of OATP1B1 and its impact on DDI prediction
 SSX 3 rd Annual Conference (Oct 11, 2018) In vitro substrate-dependent inhibition of OATP1B1 and its impact on DDI prediction Yoshitane Nozaki, PhD DMPK Tsukuba Organic Anion Transporting Polypeptide (OATP)
SSX 3 rd Annual Conference (Oct 11, 2018) In vitro substrate-dependent inhibition of OATP1B1 and its impact on DDI prediction Yoshitane Nozaki, PhD DMPK Tsukuba Organic Anion Transporting Polypeptide (OATP)
T Eley, Y-H Han, S-P Huang, B He, W Li, W Bedford, M Stonier, D Gardiner, K Sims, P Balimane, D Rodrigues, RJ Bertz
 IN VIVO AND IN VITRO ASSESSMENT OF ASUNAPREVIR (ASV; BMS-650032) AS AN INHIBITOR AND SUBSTRATE OF ORGANIC ANION TRANSPORT POLYPEPTIDE (OATP) TRANSPORTERS IN HEALTHY VOLUNTEERS T Eley, Y-H Han, S-P Huang,
IN VIVO AND IN VITRO ASSESSMENT OF ASUNAPREVIR (ASV; BMS-650032) AS AN INHIBITOR AND SUBSTRATE OF ORGANIC ANION TRANSPORT POLYPEPTIDE (OATP) TRANSPORTERS IN HEALTHY VOLUNTEERS T Eley, Y-H Han, S-P Huang,
Use of in vitro cell assays and noninvasive imaging techniques to reduce animal experiments in drug development
 Use of in vitro cell assays and noninvasive imaging techniques to reduce animal experiments in drug development J. Jia, M. Keiser, S. Oswald, W. Siegmund Department of Clinical Pharmacology, Ernst-Moritz-Arndt
Use of in vitro cell assays and noninvasive imaging techniques to reduce animal experiments in drug development J. Jia, M. Keiser, S. Oswald, W. Siegmund Department of Clinical Pharmacology, Ernst-Moritz-Arndt
Different Effects of SLCO1B1 Polymorphism on the Pharmacokinetics of Atorvastatin and Rosuvastatin
 nature publishing group Different Effects of SLCO1B1 Polymorphism on the Pharmacokinetics of Atorvastatin and Rosuvastatin MK Pasanen 1, H Fredrikson 1, PJ Neuvonen 1 and M Niemi 1 Thirty-two healthy volunteers
nature publishing group Different Effects of SLCO1B1 Polymorphism on the Pharmacokinetics of Atorvastatin and Rosuvastatin MK Pasanen 1, H Fredrikson 1, PJ Neuvonen 1 and M Niemi 1 Thirty-two healthy volunteers
Stable transfected HEK293-OATP cells for transporter analysis A model system for the assay of drug uptake.
 Stable transfected HEK293-OATP cells for transporter analysis A model system for the assay of drug uptake. PRIMACYT Cell Culture Technology GmbH, Hagenower Str. 73, D-19061 Schwerin, Germany E-mail: info@primacyt.com,
Stable transfected HEK293-OATP cells for transporter analysis A model system for the assay of drug uptake. PRIMACYT Cell Culture Technology GmbH, Hagenower Str. 73, D-19061 Schwerin, Germany E-mail: info@primacyt.com,
Yvonne Y. Lau, Hideaki Okochi, Yong Huang, and Leslie Z. Benet
 0090-9556/06/3407-1175 1181$20.00 DRUG METABOLISM AND DISPOSITION Vol. 34, No. 7 Copyright 2006 by The American Society for Pharmacology and Experimental Therapeutics 9076/3121398 DMD 34:1175 1181, 2006
0090-9556/06/3407-1175 1181$20.00 DRUG METABOLISM AND DISPOSITION Vol. 34, No. 7 Copyright 2006 by The American Society for Pharmacology and Experimental Therapeutics 9076/3121398 DMD 34:1175 1181, 2006
Cryo Characterization Report (CCR)
 Human Cryopreserved Hepatocytes Lot number: HUM4061B Date: October 19, 2014 Cryo Characterization Report (CCR) Lot Overview Qualification Catalog Number Quantity Cryopreserved human hepatocytes, Qualyst
Human Cryopreserved Hepatocytes Lot number: HUM4061B Date: October 19, 2014 Cryo Characterization Report (CCR) Lot Overview Qualification Catalog Number Quantity Cryopreserved human hepatocytes, Qualyst
HEK293 cells transfected with human MATE1, MATE2-K, or vector control were established by
 SUPPLEMENTAL DIGITAL CONTENT METHODS In Vitro Metformin Transport Studies Effect of Dolutegravir on Metformin Transport by MATE1 and MATE2-K HEK293 cells transfected with human MATE1, MATE2-K, or vector
SUPPLEMENTAL DIGITAL CONTENT METHODS In Vitro Metformin Transport Studies Effect of Dolutegravir on Metformin Transport by MATE1 and MATE2-K HEK293 cells transfected with human MATE1, MATE2-K, or vector
Transporters DDI-2018
 Transporters DDI-2018 Mark S. Warren, Ph.D. June 16, 2018 Senior Director of Assay Services DDI-2018: 21 st Conference on DDIs FDA guidance documents: A 21 year history 1997 2006 2012 2017 Each year, large
Transporters DDI-2018 Mark S. Warren, Ph.D. June 16, 2018 Senior Director of Assay Services DDI-2018: 21 st Conference on DDIs FDA guidance documents: A 21 year history 1997 2006 2012 2017 Each year, large
SLCO1B1 Pharmacogenetic Competency
 SLCO1B1 Pharmacogenetic Competency Updated on 6/2015 Pre-test Question # 1 Which of the following is not currently a recognized SLCO1B1 phenotype? a) Low function b) Normal function c) Intermediate function
SLCO1B1 Pharmacogenetic Competency Updated on 6/2015 Pre-test Question # 1 Which of the following is not currently a recognized SLCO1B1 phenotype? a) Low function b) Normal function c) Intermediate function
Determinants of Drug Disposition
 Drug Transporters: In Vitro and Knockout Model Systems, Pharmacogenomics, and Clinical Relevance Richard B. Kim MD, FRCP(C) Professor & Chair, Division of Clinical Pharmacology Director, Centre for Clinical
Drug Transporters: In Vitro and Knockout Model Systems, Pharmacogenomics, and Clinical Relevance Richard B. Kim MD, FRCP(C) Professor & Chair, Division of Clinical Pharmacology Director, Centre for Clinical
Assessment of inhibitory effect of many therapeutically important drugs on bile acid transport by NTCP, BSEP and other transporters
 ct 6, 26 Falk Symposium 155; XIX International Bile Acid Meeting Assessment of inhibitory effect of many therapeutically important drugs on bile acid transport by NTCP, BSEP and other transporters Kazuya
ct 6, 26 Falk Symposium 155; XIX International Bile Acid Meeting Assessment of inhibitory effect of many therapeutically important drugs on bile acid transport by NTCP, BSEP and other transporters Kazuya
Evaluation of Proposed In Vivo Probe Substrates and Inhibitors for Phenotyping Transporter Activity in Humans
 Supplement Article Evaluation of Proposed In Vivo Probe Substrates and Inhibitors for Phenotyping Transporter Activity in Humans The Journal of Clinical Pharmacology (2016), 56(S7) S82 S98 C 2016, The
Supplement Article Evaluation of Proposed In Vivo Probe Substrates and Inhibitors for Phenotyping Transporter Activity in Humans The Journal of Clinical Pharmacology (2016), 56(S7) S82 S98 C 2016, The
Withdrawal of Cerivastatin Revealed a Flaw of Post-marketing Surveillance System in the United States
 Bull. Natl. Inst. Health Sci., 123 Notes # Withdrawal of Cerivastatin Revealed a Flaw of Post-marketing Surveillance System in the United States Journal of American Medical Association (JAMA) Statin HMG
Bull. Natl. Inst. Health Sci., 123 Notes # Withdrawal of Cerivastatin Revealed a Flaw of Post-marketing Surveillance System in the United States Journal of American Medical Association (JAMA) Statin HMG
Current Approaches and Applications of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters
 Current Approaches and Applications of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters Uwe Fuhr, University Hospital Cologne 1 Definition Phenotyping is quantifying the in vivo activity
Current Approaches and Applications of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters Uwe Fuhr, University Hospital Cologne 1 Definition Phenotyping is quantifying the in vivo activity
Welcome to the webinar... We will begin shortly
 Welcome to the webinar... We will begin shortly Evaluation of Ketoconazole and Its Alternative Clinical CYP3A4/5 Inhibitors as Inhibitors of Drug Transporters: The In Vitro Effects of Ketoconazole, Ritonavir,
Welcome to the webinar... We will begin shortly Evaluation of Ketoconazole and Its Alternative Clinical CYP3A4/5 Inhibitors as Inhibitors of Drug Transporters: The In Vitro Effects of Ketoconazole, Ritonavir,
Itraconazole and Clarithromycin as Ketoconazole Alternatives for Clinical CYP3A Inhibition Studies to Quantify Victim DDI Potential
 Itraconazole and Clarithromycin as Ketoconazole Alternatives for Clinical CYP3A Inhibition Studies to Quantify Victim DDI Potential Alice Ban Ke, Ph.D. Consultant & Scientific Advisor Simcyp Limited Alice.Ke@certara.com
Itraconazole and Clarithromycin as Ketoconazole Alternatives for Clinical CYP3A Inhibition Studies to Quantify Victim DDI Potential Alice Ban Ke, Ph.D. Consultant & Scientific Advisor Simcyp Limited Alice.Ke@certara.com
Transporters and Drug-Drug Interactions: Important Determinants of Drug Disposition and Effects s
 Supplemental Material can be found at: /content/suppl/2013/05/23/65.3.944.dc1.html 1521-0081/65/3/944 966$25.00 http://dx.doi.org/10.1124/pr.113.007518 PHARMACOLOGICAL REVIEWS Pharmacol Rev 65:944 966,
Supplemental Material can be found at: /content/suppl/2013/05/23/65.3.944.dc1.html 1521-0081/65/3/944 966$25.00 http://dx.doi.org/10.1124/pr.113.007518 PHARMACOLOGICAL REVIEWS Pharmacol Rev 65:944 966,
BACKGROUND AND PURPOSE
 1836..1847 British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01672.x www.brjpharmacol.org RESEARCH PAPERbph_1672 Identification of drugs and drug metabolites as substrates of multidrug resistance
1836..1847 British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01672.x www.brjpharmacol.org RESEARCH PAPERbph_1672 Identification of drugs and drug metabolites as substrates of multidrug resistance
Diabetes Care Publish Ahead of Print, published online December 10, 2009
 Diabetes Care Publish Ahead of Print, published online December 10, 2009 TNF-α-C-857T polymorphism, LDL-cho & statins Association of the TNF-α-C-857T Polymorphism with Resistance to the Cholesterol-Lowering
Diabetes Care Publish Ahead of Print, published online December 10, 2009 TNF-α-C-857T polymorphism, LDL-cho & statins Association of the TNF-α-C-857T Polymorphism with Resistance to the Cholesterol-Lowering
Exploiting BDDCS and the Role of Transporters
 Exploiting BDDCS and the Role of Transporters (Therapeutic benefit of scientific knowledge of biological transporters, understanding the clinical relevant effects of active transport on oral drug absorption)
Exploiting BDDCS and the Role of Transporters (Therapeutic benefit of scientific knowledge of biological transporters, understanding the clinical relevant effects of active transport on oral drug absorption)
Fundamentals of Membrane Transporters and their Role in In Vivo PK/PD of Drugs
 Fundamentals of Membrane Transporters and their Role in In Vivo PK/PD of Drugs Jash Unadkat, Ph.D. Department of Pharmaceutics University of Washington Seattle, WA 98195 http://depts.washington.edu/pceut/faculty_research/faculty_members/unadkat_jashvant.html
Fundamentals of Membrane Transporters and their Role in In Vivo PK/PD of Drugs Jash Unadkat, Ph.D. Department of Pharmaceutics University of Washington Seattle, WA 98195 http://depts.washington.edu/pceut/faculty_research/faculty_members/unadkat_jashvant.html
Application of a Physiologically Based Pharmacokinetic Model to Predict OATP1B1-Related Variability in Pharmacodynamics of Rosuvastatin
 Original Article Citation: CPT Pharmacometrics Syst. Pharmacol. (), e; doi:./psp.. ASCPT All rights reserved -/ Application of a Physiologically Based Pharmacokinetic Model to Predict OATPB-Related Variability
Original Article Citation: CPT Pharmacometrics Syst. Pharmacol. (), e; doi:./psp.. ASCPT All rights reserved -/ Application of a Physiologically Based Pharmacokinetic Model to Predict OATPB-Related Variability
Strategy on Drug Transporter Investigation Why, How, Which & When. Jasminder Sahi
 Strategy on Drug Transporter Investigation Why, How, Which & When Jasminder Sahi Intestine Drug Absorption PEPT1 OATPs MCTs AE2 Epithelial Cell MCTs MRP3 Liver Excretion via Liver Kidney MRPs OATPs N PT1
Strategy on Drug Transporter Investigation Why, How, Which & When Jasminder Sahi Intestine Drug Absorption PEPT1 OATPs MCTs AE2 Epithelial Cell MCTs MRP3 Liver Excretion via Liver Kidney MRPs OATPs N PT1
Influence of SLCO1B1 and CYP2C8 gene polymorphisms on rosiglitazone pharmacokinetics in healthy volunteers
 Influence of SLCO1B1 and CYP2C8 gene polymorphisms on rosiglitazone pharmacokinetics in healthy volunteers Christina L. Aquilante, 1* Lane R. Bushman, 1 Shannon D. Knutsen, 1 Lauren E. Burt, 1 Lucille
Influence of SLCO1B1 and CYP2C8 gene polymorphisms on rosiglitazone pharmacokinetics in healthy volunteers Christina L. Aquilante, 1* Lane R. Bushman, 1 Shannon D. Knutsen, 1 Lauren E. Burt, 1 Lucille
Investigating Transporter-Mediated Drug-Drug Interactions Using a Physiologically Based Pharmacokinetic Model of Rosuvastatin
 Citation: CPT Pharmacometrics Syst. Pharmacol. (2017) 6, 228 238; VC 2017 ASCPT All rights reserved doi:10.1002/psp4.12168 ORIGINAL ARTICLE Investigating Transporter-Mediated Drug-Drug Interactions Using
Citation: CPT Pharmacometrics Syst. Pharmacol. (2017) 6, 228 238; VC 2017 ASCPT All rights reserved doi:10.1002/psp4.12168 ORIGINAL ARTICLE Investigating Transporter-Mediated Drug-Drug Interactions Using
EVALUATION OF DRUG-DRUG INTERACTION POTENTIAL BETWEEN SACUBITRIL/VALSARTAN (LCZ696) AND STATINS USING A PHYSIOLOGICALLY- BASED PHARMACOKINETIC MODEL
 Drug metabolism and Pharmacokinetics/PK Sciences EVALUATIN F DRUG-DRUG INTERACTIN PTENTIAL BETWEEN SACUBITRIL/VALSARTAN (LCZ696) AND STATINS USING A PHYSILGICALLY- BASED PHARMACKINETIC MDEL Imad Hanna,
Drug metabolism and Pharmacokinetics/PK Sciences EVALUATIN F DRUG-DRUG INTERACTIN PTENTIAL BETWEEN SACUBITRIL/VALSARTAN (LCZ696) AND STATINS USING A PHYSILGICALLY- BASED PHARMACKINETIC MDEL Imad Hanna,
Falk Symposium 156: Genetics in Liver Disease. Pharmacogenetics. Gerd Kullak-Ublick
 Falk Symposium 156: Genetics in Liver Disease Pharmacogenetics Gerd Kullak-Ublick Division of Clinical Pharmacology and Toxicology Department of Internal Medicine University Hospital Zurich Freiburg, 8.
Falk Symposium 156: Genetics in Liver Disease Pharmacogenetics Gerd Kullak-Ublick Division of Clinical Pharmacology and Toxicology Department of Internal Medicine University Hospital Zurich Freiburg, 8.
Suitability of Digoxin as a P Glycoprotein Probe: Implications of Other Transporters on Sensitivity and Specificity
 Review Suitability of Digoxin as a P Glycoprotein Probe: Implications of Other Transporters on Sensitivity and Specificity The Journal of Clinical Pharmacology XX(XX) 1 11 2013, The American College of
Review Suitability of Digoxin as a P Glycoprotein Probe: Implications of Other Transporters on Sensitivity and Specificity The Journal of Clinical Pharmacology XX(XX) 1 11 2013, The American College of
NIH Public Access Author Manuscript Clin Pharmacol Ther. Author manuscript; available in PMC 2010 November 9.
 NIH Public Access Author Manuscript Published in final edited form as: Clin Pharmacol Ther. 2008 February ; 83(2): 273 280. doi:10.1038/sj.clpt.6100275. Effect of Genetic Variation in the Organic Cation
NIH Public Access Author Manuscript Published in final edited form as: Clin Pharmacol Ther. 2008 February ; 83(2): 273 280. doi:10.1038/sj.clpt.6100275. Effect of Genetic Variation in the Organic Cation
Yoshihisa Shitara, Masaru Hirano, Hitoshi Sato, and Yuichi Sugiyama
 0022-3565/04/3111-228 236$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 311, No. 1 Copyright 2004 by The American Society for Pharmacology and Experimental Therapeutics 68536/1171497
0022-3565/04/3111-228 236$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 311, No. 1 Copyright 2004 by The American Society for Pharmacology and Experimental Therapeutics 68536/1171497
DRUG METABOLISM AND PHARMACOKINETICS (DMPK) Lena Gustavsson, H. Lundbeck A/S, November 2015
 DRUG METABOLISM AND PHARMACOKINETICS (DMPK), H. Lundbeck A/S, LEGU@lundbeck.com November 2015 DMPK in Drug Discovery and Development Agenda Introduction Optimizing pharmacokinetic properties Absorption
DRUG METABOLISM AND PHARMACOKINETICS (DMPK), H. Lundbeck A/S, LEGU@lundbeck.com November 2015 DMPK in Drug Discovery and Development Agenda Introduction Optimizing pharmacokinetic properties Absorption
Kinetic characterization of rat hepatic uptake of 16 actively transported drugs. Yoshiyuki Yabe, Aleksandra Galetin and J Brian Houston
 DMD Fast Forward. Published on July 5, 2011 as doi:10.1124/dmd.111.040477 Kinetic characterization of rat hepatic uptake of 16 actively transported drugs Yoshiyuki Yabe, Aleksandra Galetin and J Brian
DMD Fast Forward. Published on July 5, 2011 as doi:10.1124/dmd.111.040477 Kinetic characterization of rat hepatic uptake of 16 actively transported drugs Yoshiyuki Yabe, Aleksandra Galetin and J Brian
Prediction of in vivo hepatic clearance and DDI of OATP substrates: Comparison of different in vitro approaches. Yuichi Sugiyama
 Prediction of in vivo hepatic clearance and DDI of OATP substrates: Comparison of different in vitro approaches. Yuichi Sugiyama Sugiyama Laboratory, RIKEN Innovation Center, RIKEN, Research Cluster for
Prediction of in vivo hepatic clearance and DDI of OATP substrates: Comparison of different in vitro approaches. Yuichi Sugiyama Sugiyama Laboratory, RIKEN Innovation Center, RIKEN, Research Cluster for
DIFFERENTIAL INTERACTION OF 3-HYDROXY-3-METHYLGLUTARYL-COA REDUCTASE INHIBITORS WITH ABCB1, ABCC2, AND OATP1B1
 0090-9556/05/3304-537 546$20.00 DRUG METABOLISM AND DISPOSITION Vol. 33, No. 4 Copyright 2005 by The American Society for Pharmacology and Experimental Therapeutics 2477/1196870 DMD 33:537 546, 2005 Printed
0090-9556/05/3304-537 546$20.00 DRUG METABOLISM AND DISPOSITION Vol. 33, No. 4 Copyright 2005 by The American Society for Pharmacology and Experimental Therapeutics 2477/1196870 DMD 33:537 546, 2005 Printed
MODULE PHARMACOKINETICS WRITTEN SUMMARY
 MODULE 2.6.4. PHARMACOKINETICS WRITTEN SUMMARY m2.6.4. Pharmacokinetics Written Summary 2013N179518_00 TABLE OF CONTENTS PAGE 1. BRIEF SUMMARY...4 2. METHODS OF ANALYSIS...5 3. ABSORPTION...6 4. DISTRIBUTION...7
MODULE 2.6.4. PHARMACOKINETICS WRITTEN SUMMARY m2.6.4. Pharmacokinetics Written Summary 2013N179518_00 TABLE OF CONTENTS PAGE 1. BRIEF SUMMARY...4 2. METHODS OF ANALYSIS...5 3. ABSORPTION...6 4. DISTRIBUTION...7
Involvement of transporters in the hepatic uptake and biliary excretion of
 DMD This Fast article Forward. has not been Published copyedited on and formatted. April 19, The 2006 final as version doi:10.1124/dmd.105.008938 may differ from this version. Involvement of transporters
DMD This Fast article Forward. has not been Published copyedited on and formatted. April 19, The 2006 final as version doi:10.1124/dmd.105.008938 may differ from this version. Involvement of transporters
Univ.-Doz. Prof. Dr. W. Renner
 Pharmacogenetics of statin-inducedinduced myopathies Wilfried Renner Medical University Graz Clinical Institute of Medical and Chemical Laboratory Diagnostics It started with a fungus 1973: Isolation of
Pharmacogenetics of statin-inducedinduced myopathies Wilfried Renner Medical University Graz Clinical Institute of Medical and Chemical Laboratory Diagnostics It started with a fungus 1973: Isolation of
RISK FACTORS AND DRUG TO STATIN-INDUCED MYOPATHY
 RISK FACTORS AND DRUG INTERACTION PREDISPOSING TO STATIN-INDUCED MYOPATHY Assist. Prof. Dr. Verawan Uchaipichat Clinical Pharmacy Department Khon Kaen University Advanced Pharmacotherapy 2012 Updated d
RISK FACTORS AND DRUG INTERACTION PREDISPOSING TO STATIN-INDUCED MYOPATHY Assist. Prof. Dr. Verawan Uchaipichat Clinical Pharmacy Department Khon Kaen University Advanced Pharmacotherapy 2012 Updated d
DRUG-DRUG INTERACTIONS OF GLECAPREVIR AND PIBRENTASVIR WITH PRAVASTATIN, ROSUVASTATIN, OR DABIGATRAN ETEXILATE
 DRUG-DRUG INTERACTIONS OF GLECAPREVIR AND PIBRENTASVIR WITH PRAVASTATIN, ROSUVASTATIN, OR DABIGATRAN ETEXILATE Matthew P. Kosloski, Weihan Zhao, Hong Li, Stanley Subhead Wang, Calibri Joaquin 14pt, Valdes,
DRUG-DRUG INTERACTIONS OF GLECAPREVIR AND PIBRENTASVIR WITH PRAVASTATIN, ROSUVASTATIN, OR DABIGATRAN ETEXILATE Matthew P. Kosloski, Weihan Zhao, Hong Li, Stanley Subhead Wang, Calibri Joaquin 14pt, Valdes,
Wakaba Yamashiro, Kazuya Maeda, Masakazu Hirouchi, Yasuhisa Adachi, Zhuohan Hu, and Yuichi Sugiyama
 0090-9556/06/3407-1247 1254$20.00 DRUG METABOLISM AND DISPOSITION Vol. 34, No. 7 Copyright 2006 by The American Society for Pharmacology and Experimental Therapeutics 8938/3122648 DMD 34:1247 1254, 2006
0090-9556/06/3407-1247 1254$20.00 DRUG METABOLISM AND DISPOSITION Vol. 34, No. 7 Copyright 2006 by The American Society for Pharmacology and Experimental Therapeutics 8938/3122648 DMD 34:1247 1254, 2006
Inhibition of Human Hepatic Bile Acid Transporters as Contributing Factors to Drug-Induced Liver Injury
 Inhibition of Human Hepatic Bile Acid Transporters as Contributing Factors to Drug-Induced Liver Injury Kenneth R. Brouwer, Ph.D., RPh Chief Scientific Officer DDI Meeting June 2017 Seattle, Washington
Inhibition of Human Hepatic Bile Acid Transporters as Contributing Factors to Drug-Induced Liver Injury Kenneth R. Brouwer, Ph.D., RPh Chief Scientific Officer DDI Meeting June 2017 Seattle, Washington
Interaction of baicalin with transporters
 Interaction of baicalin with transporters PhD thesis Kalaposné Kovács Bernadett Semmelweis University Doctoral School of Pharmaceutical Sciences Supervisor: External Consultant: Official reviewers: Dr.
Interaction of baicalin with transporters PhD thesis Kalaposné Kovács Bernadett Semmelweis University Doctoral School of Pharmaceutical Sciences Supervisor: External Consultant: Official reviewers: Dr.
Drug Metabolism and Disposition
 Investigation of the Impact of Substrate Selection on In Vitro Organic Anion Transporting Polypeptide 1B1 Inhibition Profiles for the Prediction of Drug-drug Interactions Saki Izumi, Yoshitane Nozaki,
Investigation of the Impact of Substrate Selection on In Vitro Organic Anion Transporting Polypeptide 1B1 Inhibition Profiles for the Prediction of Drug-drug Interactions Saki Izumi, Yoshitane Nozaki,
Clinical Implications of Pharmacogenetic Variation on the Effects of Statins
 REVIEW ARTICLE Drug Saf 2011; 34 (1): 1-19 0114-5916/11/0001-0001/$49.95/0 ª 2011 Adis Data Information BV. All rights reserved. Clinical Implications of Pharmacogenetic Variation on the Effects of Statins
REVIEW ARTICLE Drug Saf 2011; 34 (1): 1-19 0114-5916/11/0001-0001/$49.95/0 ª 2011 Adis Data Information BV. All rights reserved. Clinical Implications of Pharmacogenetic Variation on the Effects of Statins
A mong the 20 leading prescription
 Safety and Statins: Pharmacologic and Clinical Perspectives Michael B. Bottorff, PharmD A mong the 20 leading prescription drugs in the United States, 3 agents atorvastatin (Lipitor), simvastatin (Zocor),
Safety and Statins: Pharmacologic and Clinical Perspectives Michael B. Bottorff, PharmD A mong the 20 leading prescription drugs in the United States, 3 agents atorvastatin (Lipitor), simvastatin (Zocor),
Antihyperlipidemic Drugs
 Antihyperlipidemic Drugs Hyperlipidemias. Hyperlipoproteinemias. Hyperlipemia. Hypercholestrolemia. Direct relationship with acute pancreatitis and atherosclerosis Structure Lipoprotein Particles Types
Antihyperlipidemic Drugs Hyperlipidemias. Hyperlipoproteinemias. Hyperlipemia. Hypercholestrolemia. Direct relationship with acute pancreatitis and atherosclerosis Structure Lipoprotein Particles Types
Organic anion transporter protein (OATP1B1) encoded by SLCO1B1 gene polymorphism (388A>G) & susceptibility in gallstone disease
 Indian J Med Res 29, February 2009, pp 70-75 Organic anion transporter protein (OATPB) encoded by SLCOB gene polymorphism (388A>G) & susceptibility in gallstone disease Charulata Jindal, Sandeep Kumar
Indian J Med Res 29, February 2009, pp 70-75 Organic anion transporter protein (OATPB) encoded by SLCOB gene polymorphism (388A>G) & susceptibility in gallstone disease Charulata Jindal, Sandeep Kumar
Drug Interactions Year 2 Clinical Pharmacology
 1 Drug Interactions Year 2 Clinical Pharmacology Prof Mark McKeage Department of Pharmacology & Clinical Pharmacology 2 Objectives Explain the potential for interacting drugs to cause beneficial and harmful
1 Drug Interactions Year 2 Clinical Pharmacology Prof Mark McKeage Department of Pharmacology & Clinical Pharmacology 2 Objectives Explain the potential for interacting drugs to cause beneficial and harmful
Effects of Uptake and Efflux Transporter Inhibition on Erythromycin Breath Test Results
 nature publishing group Effects of Uptake and Efflux Transporter Inhibition on Erythromycin Breath Test Results LA Frassetto 1, S Poon 2, C Tsourounis 2, C Valera 2 and LZ Benet 3 The erythromycin breath
nature publishing group Effects of Uptake and Efflux Transporter Inhibition on Erythromycin Breath Test Results LA Frassetto 1, S Poon 2, C Tsourounis 2, C Valera 2 and LZ Benet 3 The erythromycin breath
Introduction. Drug Design, Development and Therapy. Choon Ok Kim 1 Eun Sil Oh 2 Hohyun Kim 3 Min Soo Park 1,4. Original Research
 Drug Design, Development and Therapy open access to scientific and medical research Open Access Full Text Article Original Research Pharmacokinetic interactions between glimepiride and rosuvastatin in
Drug Design, Development and Therapy open access to scientific and medical research Open Access Full Text Article Original Research Pharmacokinetic interactions between glimepiride and rosuvastatin in
Biopharmaceutics Drug Disposition Classification System (BDDCS) --- Its Impact and Application
 Biopharmaceutics Drug Disposition Classification System (BDDCS) --- Its Impact and Application Leslie Z. Benet, Ph.D. Professor of Bioengineering and Therapeutic Sciences Schools of Pharmacy and Medicine
Biopharmaceutics Drug Disposition Classification System (BDDCS) --- Its Impact and Application Leslie Z. Benet, Ph.D. Professor of Bioengineering and Therapeutic Sciences Schools of Pharmacy and Medicine
Multiple Transporters Affect the Disposition of Atorvastatin and Its Two Active Hydroxy Metabolites: Application of in Vitro and ex Situ Systems
 0022-3565/06/3162-762 771$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 316, No. 2 Copyright 2006 by The American Society for Pharmacology and Experimental Therapeutics 93088/3076593
0022-3565/06/3162-762 771$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 316, No. 2 Copyright 2006 by The American Society for Pharmacology and Experimental Therapeutics 93088/3076593
Trimethoprim and the CYP2C8*3 allele have opposite effects. on the pharmacokinetics of pioglitazone
 DMD Fast This article Forward. has not Published been copyedited on and October formatted. The 3, 2007 final version as doi:10.1124/dmd.107.018010 may differ from this version. Trimethoprim and the CYP2C8*3
DMD Fast This article Forward. has not Published been copyedited on and October formatted. The 3, 2007 final version as doi:10.1124/dmd.107.018010 may differ from this version. Trimethoprim and the CYP2C8*3
Genetics and Genomics: Influence on Individualization of Medication Regimes
 Genetics and Genomics: Influence on Individualization of Medication Regimes Joseph S Bertino Jr., Pharm.D., FCCP Schenectady, NY USA Goals and Objectives To discuss pharmacogenetics and pharmacogenomics
Genetics and Genomics: Influence on Individualization of Medication Regimes Joseph S Bertino Jr., Pharm.D., FCCP Schenectady, NY USA Goals and Objectives To discuss pharmacogenetics and pharmacogenomics
Commentary. New mechanisms by which statins lower plasma cholesterol. Henri Brunengraber
 Commentary New mechanisms by which statins lower plasma cholesterol Henri Brunengraber Department of Nutrition Case Western Reserve University Cleveland, OH 44109 Phone: 216 368 6429 Fax: 216 368 6560
Commentary New mechanisms by which statins lower plasma cholesterol Henri Brunengraber Department of Nutrition Case Western Reserve University Cleveland, OH 44109 Phone: 216 368 6429 Fax: 216 368 6560
Investigation of the Rate-Determining Process in the Hepatic Elimination of HMG-CoA Reductase Inhibitors in Rats and Humans
 9-9556//382-25 222$2. DRUG METABOLISM AND DISPOSITION Vol. 38, No. 2 Copyright 2 by The American Society for Pharmacology and Experimental Therapeutics 3254/3548264 DMD 38:25 222, 2 Printed in U.S.A. Investigation
9-9556//382-25 222$2. DRUG METABOLISM AND DISPOSITION Vol. 38, No. 2 Copyright 2 by The American Society for Pharmacology and Experimental Therapeutics 3254/3548264 DMD 38:25 222, 2 Printed in U.S.A. Investigation
Vectorial Transport by Double-Transfected Cells Expressing the Human Uptake Transporter SLC21A8 and the Apical Export Pump ABCC2
 0026-895X/01/6005-934 943$3.00 MOLECULAR PHARMACOLOGY Vol. 60, No. 5 Copyright 2001 The American Society for Pharmacology and Experimental Therapeutics 1103/938989 Mol Pharmacol 60:934 943, 2001 Printed
0026-895X/01/6005-934 943$3.00 MOLECULAR PHARMACOLOGY Vol. 60, No. 5 Copyright 2001 The American Society for Pharmacology and Experimental Therapeutics 1103/938989 Mol Pharmacol 60:934 943, 2001 Printed
COMPARISON OF THREE IN VITRO MODELS EXPRESSING THE MEMBRANE DRUG TRANSPORTER OATP1B1
 Master thesis submitted for the degree Candidata pharmaciae COMPARISON OF THREE IN VITRO MODELS EXPRESSING THE MEMBRANE DRUG TRANSPORTER OATP1B1 Maria Ulvestad Department of Pharmaceutical Biosciences,
Master thesis submitted for the degree Candidata pharmaciae COMPARISON OF THREE IN VITRO MODELS EXPRESSING THE MEMBRANE DRUG TRANSPORTER OATP1B1 Maria Ulvestad Department of Pharmaceutical Biosciences,
1.* Dosage. A. Adults
 3-Hydroxy-3-Methylglutaryl Coenzyme A (HMG-CoA) Reductase Inhibitors [Developed, November 1994; Revised, October 1996; September 1997; September 1998; October 1999; November 1999; August 2000; September
3-Hydroxy-3-Methylglutaryl Coenzyme A (HMG-CoA) Reductase Inhibitors [Developed, November 1994; Revised, October 1996; September 1997; September 1998; October 1999; November 1999; August 2000; September
Building innovative drug discovery alliances. Hepatic uptake and drug disposition o in vitro and in silico approaches
 Building innovative drug discovery alliances Hepatic uptake and drug disposition o in vitro and in silico approaches Dr Beth Williamson Evotec AG, 2017 Outline Importance of predicting clearance In vitro
Building innovative drug discovery alliances Hepatic uptake and drug disposition o in vitro and in silico approaches Dr Beth Williamson Evotec AG, 2017 Outline Importance of predicting clearance In vitro
Supporting information
 Supporting information Intracellular drug bioavailability: a new predictor of system dependent drug disposition Mateus, André 1* ; Treyer, Andrea 1* ; Wegler, Christine 1,2 ; Karlgren, Maria 1 ; Matsson,
Supporting information Intracellular drug bioavailability: a new predictor of system dependent drug disposition Mateus, André 1* ; Treyer, Andrea 1* ; Wegler, Christine 1,2 ; Karlgren, Maria 1 ; Matsson,
The importance of pharmacogenetics in the treatment of epilepsy
 The importance of pharmacogenetics in the treatment of epilepsy Öner Süzer and Esat Eşkazan İstanbul University, Cerrahpaşa Faculty of Medicine, Department of Pharmacology and Clinical Pharmacology Introduction
The importance of pharmacogenetics in the treatment of epilepsy Öner Süzer and Esat Eşkazan İstanbul University, Cerrahpaşa Faculty of Medicine, Department of Pharmacology and Clinical Pharmacology Introduction
Human TRPC6 Ion Channel Cell Line
 TECHNICAL DATA SHEET ValiScreen Ion Channel Cell Line Caution: For Laboratory Use. A research product for research purposes only Human TRPC6 Ion Channel Cell Line Product No.: AX-012-C Lot No.: 512-548-A
TECHNICAL DATA SHEET ValiScreen Ion Channel Cell Line Caution: For Laboratory Use. A research product for research purposes only Human TRPC6 Ion Channel Cell Line Product No.: AX-012-C Lot No.: 512-548-A
CYP2C8 Activity Recovers within 96 Hours after Gemfibrozil Dosing: Estimation of CYP2C8 Half-Life Using Repaglinide as an in Vivo Probe
 0090-9556/09/3712-2359 2366$20.00 DRUG METABOLISM AND DISPOSITION Vol. 37, No. 12 Copyright 2009 by The American Society for Pharmacology and Experimental Therapeutics 29728/3537718 DMD 37:2359 2366, 2009
0090-9556/09/3712-2359 2366$20.00 DRUG METABOLISM AND DISPOSITION Vol. 37, No. 12 Copyright 2009 by The American Society for Pharmacology and Experimental Therapeutics 29728/3537718 DMD 37:2359 2366, 2009
Biopharmaceutics Drug Disposition Classification System (BDDCS) and Its Application in Drug Discovery and Development
 Biopharmaceutics Drug Disposition Classification System (BDDCS) and Its Application in Drug Discovery and Development Leslie Z. Benet, Ph.D. Professor of Bioengineering and Therapeutic Sciences University
Biopharmaceutics Drug Disposition Classification System (BDDCS) and Its Application in Drug Discovery and Development Leslie Z. Benet, Ph.D. Professor of Bioengineering and Therapeutic Sciences University
Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved
 1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
The Future of In Vitro Systems for the Assessment of Induction and Suppression of Enzymes and Transporters
 The Future of In Vitro Systems for the Assessment of Induction and Suppression of Enzymes and Transporters AAPS, San Diego November 6 th, 2014 Andrew Parkinson, XPD Consulting Lisa Almond, Simcyp-Certara
The Future of In Vitro Systems for the Assessment of Induction and Suppression of Enzymes and Transporters AAPS, San Diego November 6 th, 2014 Andrew Parkinson, XPD Consulting Lisa Almond, Simcyp-Certara
DRUG INTERACTION BETWEEN SIMVASTATIN AND ITRACONAZOLE IN MALE AND FEMALE RATS
 0090-9556/01/2907-1068 1072$3.00 DRUG METABOLISM AND DISPOSITION Vol. 29, No. 7 Copyright 2001 by The American Society for Pharmacology and Experimental Therapeutics 331/915884 DMD 29:1068 1072, 2001 Printed
0090-9556/01/2907-1068 1072$3.00 DRUG METABOLISM AND DISPOSITION Vol. 29, No. 7 Copyright 2001 by The American Society for Pharmacology and Experimental Therapeutics 331/915884 DMD 29:1068 1072, 2001 Printed
Prediction of the Effects of Renal Impairment on the Clearance for Organic Cation Drugs that. undergo Renal Secretion: A Simulation-Based Study
 DMD Fast Forward. Published on February 28, 2018 as DOI: 10.1124/dmd.117.079558 This article has not been copyedited and formatted. The final version may differ from this version. Prediction of the Effects
DMD Fast Forward. Published on February 28, 2018 as DOI: 10.1124/dmd.117.079558 This article has not been copyedited and formatted. The final version may differ from this version. Prediction of the Effects
Involvement of Intestinal Uptake Transporters in the Absorption of Azithromycin and Clarithromycin in the Rat
 0090-9556/08/3612-2492 2498$20.00 DRUG METABOLISM AND DISPOSITION Vol. 36, No. 12 Copyright 2008 by The American Society for Pharmacology and Experimental Therapeutics 22285/3402776 DMD 36:2492 2498, 2008
0090-9556/08/3612-2492 2498$20.00 DRUG METABOLISM AND DISPOSITION Vol. 36, No. 12 Copyright 2008 by The American Society for Pharmacology and Experimental Therapeutics 22285/3402776 DMD 36:2492 2498, 2008
Pelagia Research Library
 Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2015, 6(1):6-10 ISSN: 0976-8688 CODEN (USA): PSHIBD Validated RP-HPLC method for simultaneous estimation of metformin hydrochloride
Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2015, 6(1):6-10 ISSN: 0976-8688 CODEN (USA): PSHIBD Validated RP-HPLC method for simultaneous estimation of metformin hydrochloride
Comparison Between the US FDA, Japan PMDA and EMA In Vitro DDI Guidance: Are we Close to Harmonization?
 Comparison Between the US FDA, Japan PMDA and EMA In Vitro DDI Guidance: Are we Close to Harmonization? Brian Ogilvie, Ph.D. VP Scientific Consulting XenoTech, LLC bogilvie@xenotechllc.com 14 Jun, 2018
Comparison Between the US FDA, Japan PMDA and EMA In Vitro DDI Guidance: Are we Close to Harmonization? Brian Ogilvie, Ph.D. VP Scientific Consulting XenoTech, LLC bogilvie@xenotechllc.com 14 Jun, 2018
Pharmacogenomics and Pharmacokinetics ^
 Pharmacogenomics and Pharmacokinetics ^ avid F. Kisor, B.S., Pharm.. Profeor of Pharmacokinetics epartment of Pharmaceutical and Biomedical Sciences Raabe College of Pharmacy Ohio Northern University Learning
Pharmacogenomics and Pharmacokinetics ^ avid F. Kisor, B.S., Pharm.. Profeor of Pharmacokinetics epartment of Pharmaceutical and Biomedical Sciences Raabe College of Pharmacy Ohio Northern University Learning
In Vitro and In Vivo Interaction Studies Between Lesinurad, a Selective Urate Reabsorption Inhibitor, and Major Liver or Kidney Transporters
 Clin Drug Investig (2016) 36:443 452 DOI 10.1007/s40261-016-0386-y ORIGINAL RESEARCH ARTICLE In Vitro and In Vivo Interaction Studies Between Lesinurad, a Selective Urate Reabsorption Inhibitor, and Major
Clin Drug Investig (2016) 36:443 452 DOI 10.1007/s40261-016-0386-y ORIGINAL RESEARCH ARTICLE In Vitro and In Vivo Interaction Studies Between Lesinurad, a Selective Urate Reabsorption Inhibitor, and Major
CO-ADMINISTRATION WITH GRAZOPREVIR AND ELBASVIR HAS NO EFFECT ON PRAVASTATIN EXPOSURE BUT INCREASES ROSUVASTATIN EXPOSURE IN HEALTHY SUBJECTS
 CO-ADMINISTRATION WITH GRAZOPREVIR AND ELBASVIR HAS NO EFFECT ON PRAVASTATIN EXPOSURE BUT INCREASES ROSUVASTATIN EXPOSURE IN HEALTHY SUBJECTS Luzelena Caro 1, William L. Marshall 1, Hwa-Ping Feng 1, Zifang
CO-ADMINISTRATION WITH GRAZOPREVIR AND ELBASVIR HAS NO EFFECT ON PRAVASTATIN EXPOSURE BUT INCREASES ROSUVASTATIN EXPOSURE IN HEALTHY SUBJECTS Luzelena Caro 1, William L. Marshall 1, Hwa-Ping Feng 1, Zifang
Henriette E Meyer zu Schwabedissen 1, Werner Siegmund 2, Heyo K Kroemer 3 and Jens D Rollnik 4*
 Meyer zu Schwabedissen et al. BMC Research Notes 2014, 7:688 CASE REPORT Open Access Creatine kinase elevation caused by a combination of fluvastatin and telmisartan in a patient heterozygous for the CYP2C9*3
Meyer zu Schwabedissen et al. BMC Research Notes 2014, 7:688 CASE REPORT Open Access Creatine kinase elevation caused by a combination of fluvastatin and telmisartan in a patient heterozygous for the CYP2C9*3
Hepatic Efflux Transporters: Relevance to Drug-Drug Interactions and Drug Toxicity
 Hepatic Efflux Transporters: Relevance to Drug-Drug Interactions and Drug Toxicity Kim L. R. Brouwer, PharmD, PhD W.R. Kenan, Jr., Distinguished Professor Associate Dean for Research and Graduate Education
Hepatic Efflux Transporters: Relevance to Drug-Drug Interactions and Drug Toxicity Kim L. R. Brouwer, PharmD, PhD W.R. Kenan, Jr., Distinguished Professor Associate Dean for Research and Graduate Education
Pharmacogenetics and Pharmacokinetics
 Chapter 2 Pharmacogenetics and Pharmacokinetics Mauro Saivezzo/ShutterStock, Inc. L earning O bjectives Upon completion of this chapter, the student will be able to: 1. Recognize the influence of genetic
Chapter 2 Pharmacogenetics and Pharmacokinetics Mauro Saivezzo/ShutterStock, Inc. L earning O bjectives Upon completion of this chapter, the student will be able to: 1. Recognize the influence of genetic
Complexities of Hepatic Drug Transport: How Do We Sort It All Out?
 Complexities of Hepatic Drug Transport: How Do We Sort It All Out? Keith A. Hoffmaster Pfizer Research Technology Center Cambridge, MA NEDMDG 2005 Summer Symposium 06.08.2005 The Challenge Intestinal uptake
Complexities of Hepatic Drug Transport: How Do We Sort It All Out? Keith A. Hoffmaster Pfizer Research Technology Center Cambridge, MA NEDMDG 2005 Summer Symposium 06.08.2005 The Challenge Intestinal uptake
Effects of grapefruit juice on the absorption of levothyroxine
 et al. DOI:10.1111/j.1365-2125.2005.02433.x British Journal of Clinical Pharmacology Effects of grapefruit juice on the absorption of levothyroxine Jari J. Lilja, 1 Kalevi Laitinen 2 & Pertti J. Neuvonen
et al. DOI:10.1111/j.1365-2125.2005.02433.x British Journal of Clinical Pharmacology Effects of grapefruit juice on the absorption of levothyroxine Jari J. Lilja, 1 Kalevi Laitinen 2 & Pertti J. Neuvonen
Different effects of itraconazole on the pharmacokinetics of fluvastatin and lovastatin
 Different effects of itraconazole on the pharmacokinetics of fluvastatin and lovastatin K. T. Kivistö, T. Kantola & P. J. Neuvonen Department of Clinical Pharmacology, University of Helsinki and Helsinki
Different effects of itraconazole on the pharmacokinetics of fluvastatin and lovastatin K. T. Kivistö, T. Kantola & P. J. Neuvonen Department of Clinical Pharmacology, University of Helsinki and Helsinki
DRUG INTERACTIONS WITH GRAZOPREVIR AND ELBASVIR
 DRUG INTERACTIONS WITH GRAZOPREVIR AND ELBASVIR Pharmacology NS3/4A protease inhibitor NS5A inhibitor Adult Dose Investigational: 100 mg once daily Investigational: 50 mg once daily Being developed as
DRUG INTERACTIONS WITH GRAZOPREVIR AND ELBASVIR Pharmacology NS3/4A protease inhibitor NS5A inhibitor Adult Dose Investigational: 100 mg once daily Investigational: 50 mg once daily Being developed as
Effect of Multiple-Dose Ketoconazole and the Effect of Multiple-Dose Rifampin on Pharmacokinetics (PK) of the HCV NS3 Protease Inhibitor Asunaprevir
 Effect of Multiple-Dose Ketoconazole and the Effect of Multiple-Dose Rifampin on Pharmacokinetics (PK) of the HCV NS3 Protease Inhibitor Asunaprevir Eley T, 1 He B, 1 Huang S-P, 2 Stonier M, 1 Bedford
Effect of Multiple-Dose Ketoconazole and the Effect of Multiple-Dose Rifampin on Pharmacokinetics (PK) of the HCV NS3 Protease Inhibitor Asunaprevir Eley T, 1 He B, 1 Huang S-P, 2 Stonier M, 1 Bedford
Development of a Cell-Based High-Throughput Assay to Screen for Inhibitors of Organic Anion Transporting Polypeptides 1B1 and 1B3
 Current Chemical Genomics, 2010, 4, 1-8 1 Open Access Development of a Cell-Based High-Throughput Assay to Screen for Inhibitors of Organic Anion Transporting Polypeptides 1B1 and 1B3 Chunshan Gui 1, Amanda
Current Chemical Genomics, 2010, 4, 1-8 1 Open Access Development of a Cell-Based High-Throughput Assay to Screen for Inhibitors of Organic Anion Transporting Polypeptides 1B1 and 1B3 Chunshan Gui 1, Amanda
Pursuing the holy grail of predicting and verifying tissue drug concentrations: A proteomics and PET imaging approach
 Pursuing the holy grail of predicting and verifying tissue drug concentrations: A proteomics and PET imaging approach Jashvant (Jash) Unadkat Milo Gibaldi Endowed Professor Dept. of Pharmaceutics School
Pursuing the holy grail of predicting and verifying tissue drug concentrations: A proteomics and PET imaging approach Jashvant (Jash) Unadkat Milo Gibaldi Endowed Professor Dept. of Pharmaceutics School
Fenofibrate interaction
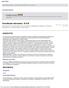 Search Query (DRUGGENERICNAME = Avandia ) AND (RECORDTYPE = Case Report ) 1 Documents Exported 1. Fenofibrate interaction First report of an interaction with concomitant rosiglitazone leading to muscular
Search Query (DRUGGENERICNAME = Avandia ) AND (RECORDTYPE = Case Report ) 1 Documents Exported 1. Fenofibrate interaction First report of an interaction with concomitant rosiglitazone leading to muscular
Regulation of the cell surface expression and transport capacity of BSEP by small chemical molecules
 Regulation of the cell surface expression and transport capacity of by small chemical molecules Hisamitsu Hayashi and Yuichi Sugiyama Dept. of Molecular Pharmacokinetics, Graduate School of Pharmaceutical
Regulation of the cell surface expression and transport capacity of by small chemical molecules Hisamitsu Hayashi and Yuichi Sugiyama Dept. of Molecular Pharmacokinetics, Graduate School of Pharmaceutical
CHOLESTAGEL 625 mg Genzyme
 CHOLESTAGEL 625 mg Genzyme 1. NAME OF THE MEDICINAL PRODUCT Cholestagel 625 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 625 mg colesevelam hydrochloride (hereafter
CHOLESTAGEL 625 mg Genzyme 1. NAME OF THE MEDICINAL PRODUCT Cholestagel 625 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 625 mg colesevelam hydrochloride (hereafter
Caveat: Validation and Limitations of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters
 Caveat: Validation and Limitations of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters Uwe Fuhr, University Hospital Cologne 1 How to Safeguard that Metrics Reflect E/T Activity? in healthy
Caveat: Validation and Limitations of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters Uwe Fuhr, University Hospital Cologne 1 How to Safeguard that Metrics Reflect E/T Activity? in healthy
cationic molecule, paracellular diffusion would be thought of as its primary mode of transport across epithelial cells.
 ABSTRACT Metformin is the most widely prescribed anti-hyperglycemic agent for Type 2 Diabetes Mellitus (T2DM). Despite its frequent use, the intestinal absorption mechanism of this orally administered
ABSTRACT Metformin is the most widely prescribed anti-hyperglycemic agent for Type 2 Diabetes Mellitus (T2DM). Despite its frequent use, the intestinal absorption mechanism of this orally administered
Evaluation of Drug-Drug Interactions FDA Perspective
 Evaluation of Drug-Drug Interactions FDA Perspective Kellie Schoolar Reynolds, Pharm.D. Deputy Director Division of Clinical Pharmacology IV Office of Clinical Pharmacology Office of Translational Sciences
Evaluation of Drug-Drug Interactions FDA Perspective Kellie Schoolar Reynolds, Pharm.D. Deputy Director Division of Clinical Pharmacology IV Office of Clinical Pharmacology Office of Translational Sciences
The CYP2C8 inhibitor gemfibrozil does not affect the pharmacokinetics of zafirlukast
 Author manuscript, published in "European Journal of Clinical Pharmacology 67, 2 (2010) 151-155" DOI : 10.1007/s00228-010-0908-0 The CYP2C8 inhibitor gemfibrozil does not affect the pharmacokinetics of
Author manuscript, published in "European Journal of Clinical Pharmacology 67, 2 (2010) 151-155" DOI : 10.1007/s00228-010-0908-0 The CYP2C8 inhibitor gemfibrozil does not affect the pharmacokinetics of
Efficient Liver Targeting and Uptake by Novel Tenofovir Prodrug, CMX157, For the Treatment of Hepatitis B
 Efficient Liver Targeting and Uptake by Novel Tenofovir Prodrug, CMX157, For the Treatment of Hepatitis B R Rush 1, J Greytok 2, T Matkovits 2, R Driz 2, JZ Sullivan-Bólyai 2, and D Standring 3 1 Allon
Efficient Liver Targeting and Uptake by Novel Tenofovir Prodrug, CMX157, For the Treatment of Hepatitis B R Rush 1, J Greytok 2, T Matkovits 2, R Driz 2, JZ Sullivan-Bólyai 2, and D Standring 3 1 Allon
Predict GENES. Select right drug. Select right dose. Develop new drugs. Non-Response Response Adverse Reaction
 Predict Select right drug Select right dose Develop new drugs GENES Non-Response Response Adverse Reaction Phase I: Each Chromosome Has Many Genes Chromosome 12 ~ 1,300 Genes Examples of Personalized Medicines
Predict Select right drug Select right dose Develop new drugs GENES Non-Response Response Adverse Reaction Phase I: Each Chromosome Has Many Genes Chromosome 12 ~ 1,300 Genes Examples of Personalized Medicines
