HHS Public Access Author manuscript Ophthalmology. Author manuscript; available in PMC 2017 February 01.
|
|
|
- Lucy Lang
- 5 years ago
- Views:
Transcription
1 Effect of prior anti-vegf injections on the risk of retained lens fragments and endophthalmitis post cataract surgery in the elderly Paul Hahn, MD, PhD 1, Arseniy P. Yashkin, PhD 2, and Frank A. Sloan, PhD 2 1 Department of Ophthalmology, Duke University Medical Center, Durham, NC 2 Department of Economics, Duke University, Durham, NC Abstract HHS Public Access Author manuscript Published in final edited form as: Ophthalmology February ; 123(2): doi: /j.ophtha Objective To investigate the effect of prior intravitreal anti-vascular endothelial growth factor (anti-vegf) injections on surgical and postoperative complication rates associated with cataract surgery in a nationally representative longitudinal sample of elderly persons. Design Retrospective, longitudinal cohort analysis Participants 203,643 Medicare beneficiaries who underwent cataract surgery from January 1, 2009 to December 31, Methods Using the 5% sample of Medicare claims data, the study assessed risks of 3 adverse outcomes following receipt of cataract surgery for beneficiaries with a history of prior intravitreal injections. Risks of these outcomes in beneficiaries with a history of intravitreal injections relative to those without were calculated using the Cox proportional hazard model. Main Outcome Measures The primary outcome was the risk of subsequent removal of retained lens fragments within 28 days following cataract surgery. Secondary outcomes were a new diagnosis of acute (<40 days) or delayed onset (40+ days) endophthalmitis as well as risk of a new primary open angle glaucoma diagnosis within 365 days following cataract surgery. Results Prior intravitreal anti-vegf injections were associated with a significantly increased risk of subsequent retained lens fragment removal within 28 days following cataract surgery (Hazard Ratio (HR): 2.26; 95% Confidence Interval (CI) ). Prior injections were also associated with increased risk of HR both acute (HR:2.29; 95% CI ) and delayed onset endophthalmitis (HR 3.65; 95% CI ). Prior injections were not a significant indicator of increased risk of a new primary open angle glaucoma diagnosis. Corresponding Author and Address for Reprints: Frank A. Sloan, PhD, 213 Social Sciences Building, Box 90097, Duke University, Durham, NC 27708, Tel: (919) , Fax: (919) , fsloan@duke.edu. Conflict of Interest: No conflicting relationship exists for any author. Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
2 Hahn et al. Page 2 Conclusions A history of intravitreal injections may be a risk factor for cataract surgeryrelated intraoperative complications and endophthalmitis. Given the frequency of intravitreal injections and cataract surgery, increased preoperative assessment, additional intraoperative caution, and postoperative vigilance are recommended in patients with a prior history of intravitreal injections undergoing cataract extraction. INTRODUCTION Intravitreal injections of anti-vascular endothelial growth factor (anti-vegf) agents have revolutionized the treatment of retinal diseases, and along with cataract surgery, are the most common ophthalmologic procedures performed in the United States (U.S.). 1 Analyses of anti-vegf injection-associated adverse events have generally focused on systemic complications, particularly thromboembolic events, and intraocular complications, including endophthalmitis, inflammation, retinal detachment, and elevated intraocular pressure. 2, 3 There are a few published individual reports of needle penetration into the lens resulting in subclinical as well rapid cataract-inducing lenticular trauma following intravitreal injection. 4 7 A single case of lens trauma was reported in the combined Phase 3 data of the pivotal ANCHOR (Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in AMD) and MARINA (Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular AMD) trials following a total of 754 ranibizumab (Lucentis; Genentech, Inc; San Francisco, CA) injections for treatment of neovascular AMD. 8, 9 However, the impact of intravitreal injections on surgical complications associated with cataract surgery has not been rigorously investigated in large studies. In addition to lenticular trauma, there is evidence that repeated intravitreal injections may be associated with sustained elevation of the intraocular pressure (IOP), which may secondarily increase rates of open angle glaucoma Cataract extraction can result in a decrease in IOP 13 and offset this elevation. Additionally, post-injection antibiotics, which were routinely administered until a recent paradigm shift away from their use after demonstration of a secondary increase in antibiotic-resistant ocular flora, 14, 15 may result in an increase in the rate of post-cataract endophthalmitis in eyes with a history of intravitreal injections. These relationships between intravitreal injections and surgical complications, post-cataract surgery endophthalmitis, and post-cataract surgery incidence of primary open angle glaucoma (POAG) have not been examined in large numbers, which are needed to elicit statistical significance in these rare events. We used the 5% sample of Medicare claims data to determine the incidence of retained lens fragments requiring subsequent removal (as a surrogate for intraoperative complications), POAG, and both acute (<40 days) and delayed onset (40+ days) 16 endophthalmitis associated with cataract surgery in patients with and without a history of prior intravitreal anti-vegf injections. We provide evidence suggesting that intravitreal injections may be associated with an increased risk of intraoperative complications and endophthalmitis.
3 Hahn et al. Page 3 METHODS Under a Duke University Institutional Review Board-approved protocol that adhered to the tenets of the Declaration of Helsinki, this study used data from the Medicare 5% claims and enrollment files provided by the U.S. Centers for Medicare and Medicaid Services (CMS) on a restricted-access public use basis. This database contains longitudinal information on beneficiary demographic characteristics, diagnoses (International Classification of Diseases, 9th Revision Clinical Modification (ICD-9)), and procedures (Current Procedure Terminology - 4 (CPT-4) from a nationally representative sample of 5% of the total population of Medicare beneficiaries. The claims files were used to identify Medicare beneficiaries with cataract surgery performed between January 1, 2009 and December 31, 2013 (Table 1). January 1, 2009 was chosen as the earliest date for inclusion in this study to allow a 3-year look-back period, i.e., to 2006, the year when the use of anti-vegf agents had reached widespread use. 17 For this same reason, beneficiaries had to be at least 68 years of age at baseline. Beneficiaries who enrolled in Medicare Advantage (MA), a private alternative to traditional Medicare, were excluded from the analysis sample since claims data are not available for such Medicare beneficiaries. We also excluded beneficiaries who moved outside the U.S. at any time during the study period. Finally, beneficiaries with diagnoses of traumatic cataract, cataract secondary to ocular disorders, cataract associated with other disorders, infantile/congenital cataract, endophthalmitis, or those with an intravitreal injection of corticosteroids, pars plana vitrectomy, or gas pneumatic retinopexy performed during the look-back period were excluded from the sample. When more than one cataract surgery procedure was performed during the study period, the date of the earliest procedure was chosen as the baseline date while the date of the second procedure was treated as a censoring event as it would not be possible to determine which operation, or which eye was associated with an observed outcome. To be included in the POAG sub-sample an additional restriction of not having a prior diagnosis of glaucoma was imposed. After imposing these restrictions the final sample sizes were: 203,646 for RLF removal/endophthalmitis and 139,656 for POAG (Table 2). The Cox proportional hazard model as implemented by Stata 11 (StataCorp Stata Statistical Software: Release 11. College Station, TX: StataCorp LP) was used for the analysis with censoring methods devised for ophthalmologic studies according to the method of Javitt, et. al.. 18 Individuals were treated as censored at the earliest of: the date of the second cataract surgery, death, entrance into a MA plan, and the end of the follow-up period (28 days RLF, 365 days POAG, <40 days acute endophthalmitis, 365 days delayed onset endophthalmitis. Common covariates were: prior history of intravitreal anti- VEGF injections, beneficiary age, male gender, race black and other (reference group: white race), indicators for the calendar year 2010, 2011, 2012, and 2013 (reference group: 2009), and a measure of severe co-morbidities represented by the Charlson index. 19 Anti- VEGF injections were specified by the CPT-4 code for intravitreal injection (67028) with a simultaneous claim for Healthcare Common Procedure Coding System (HCPCS) codes for pegaptanib sodium (Macugen; Valeant Pharmaceuticals, Inc., Montreal, CA), bevaciziumab (Avastin, Genentech, Inc.), ranibizumab (Lucentis, Genentech, Inc.), aflibercept (Eylea, Regeneron, Inc., Tarrytown, NY), and unclassified biologics (previously reported to be used
4 Hahn et al. Page 4 RESULTS almost exclusively for coding of bevacizumab prior to establishment of its HCPCS code 20 ). Binary variables for diabetic retinopathy glaucoma,, and pseudoexfoliation at baseline were included as additional covariates in the equation for RLF-removal. Diabetes mellitus was the additional covariate in the POAG analysis. The proportion of beneficiaries who received anti-vegf injections during the look-back period was 1.51% of the RLF-removal/endophthalmitis sub-sample, and 1.38% of the POAG sub-sample (Table 3). The age, gender and racial composition of the 2 sub-samples were similar, and each study year contributed a comparable number of beneficiaries. Within 28 days of cataract surgery, 0.20% of all eligible beneficiaries (0.43% with prior injections and 0.20% without prior injections) underwent subsequent RLF removal. Endophthalmitis was diagnosed within <40 days for 0.08% of all eligible beneficiaries (0.29% with prior injections and 0.08% without prior injections), while within days post surgery, 0.04% received a diagnosis of endophthalmitis (0.23% with prior injections and 0.04% without prior injections). Within 1 year of cataract surgery, 0.35% of eligible beneficiaries (0.77% with prior injections and 0.34% without prior injections) were diagnosed with POAG, and 0.20% of eligible beneficiaries (0.75% with prior injections and 0.18% without prior injections) were diagnosed with endophthalmitis.. Beneficiaries who had a history of prior anti-vegf injections had a higher risk of failure (as shown by the cumulative hazard function) in all parts of the analysis (Figure 1). A prior history of anti-vegf treatment was associated with a 126% increase [Hazard Ratio (HR): 2.26; 95% Confidence Interval (CI): ] in the risk of requiring subsequent RLF-removal within 28 days of cataract surgery (Table 4). Men were at increased risk [HR: 1.38; 95% CI: ]. The risk of RLF removal increased slightly with age [HR: 1.03; 95% CI: ]. Prior anti-vegf treatment was not a significant predictor of increased risk of being diagnosed with POAG within a year of cataract surgery. Male gender [HR: 1.37; 95% CI: ], black race [HR: 3.18; 95% CI: ], other race/ethnicity [HR: 2.02; 95% CI: ], and older age [HR: 1.02; 95% CI: ] were associated with increased risk. A history of prior anti-vegf injections was the primary factor associated with increased risk of endophthlamitis up to40 days [HR:2.29; 95% CI: ] and days [HR:3.65; 95% CI: ] from the cataract surgery date. The Charlson index was associated with a minor increase in risk [HR: 1.06; 95% CI: ] in the probability of being diagnosed with endophthalmitis within <40 days only. Of the 171 total endophthalmitis claims within < 40 days (ICD x), the mean (standard deviation) time to diagnosis was (11.67) days, and 6.43% (n=11) were coded as chronic endophthalmitis (ICD ). Similarly, of the 79 total endophthalmitis claims within days of surgery, the mean (standard deviation) time to diagnosis was (31.20) days, and 6.3% (n=5) were coded as chronic endophthalmitis.
5 Hahn et al. Page 5 DISCUSSION The relationship between cataract surgery-associated complications and prior intravitreal injections of anti-vegf agents has not been fully investigated to date. In a retrospective analysis of the pivotal ANCHOR and MARINA Phase 3 trials, Rosenfeld and co-authors concluded that cataract surgery is safe in patients with neovascular age-related macular degeneration (AMD) treated with ranibizumab. 21 Prompting their post hoc analysis was longstanding debated concern for progression of AMD following cataract surgery. This study focused on visual outcomes and did not specifically mention analysis of injectionrelated surgical complications in the 23 injection-treated eyes that underwent cataract extraction. Similarly, in other small retrospective case series, a history of prior intravitreal injections was not found to confer significant risk around cataract extraction of either AMD progression or intraoperative complications Our study utilized a large, nationally representative, longitudinal sample of U.S. elderly individuals to investigate these relationships. A key strength of our study is the large, nationally representative sample size, critical for yielding ample statistical power required to detect differences in uncommon outcomes that are likely not detectable with small clinical studies. Given these large number of observations available from the 5% Medicare claims data, we could limit our analysis to injection of anti-vegf agents, excluding beneficiaries with prior injection of steroids due to potential confounding effects with their well-known association with posterior subcapsular cataract formation, increased intraocular pressure, and glaucoma. 25, 26 A nationwide case-control study in which primary medical records data was obtained to confirm findings generated by the above analytic method confirmed the accuracy of estimates of outcome and proportional hazard despite the second eye ambiguity 27. We identified an increased risk of subsequent RLF-removal within 28 days following cataract surgery in patients with a prior history of intravitreal anti-vegf injections. Of note, in 2001, the anterior vitrectomy codes (CPT and 67010) were bundled with the cataract surgery codes (CPT and 66984). As a result of this coding change, identification of patients with intraoperative complications during cataract surgery, such as posterior capsular rupture requiring anterior vitrectomy, was not possible from analysis of claims data. Our finding of 126% increased risk is almost certainly an underestimate of the true risk for posterior capsular rupture, which can often be managed without the need for subsequent RLF-removal. In a large retrospective analysis of cataract surgery performed in the United States Veterans Health Administration system, RLF occurred in 0.20% of veterans, consistent with the overall rate of 0.20% identified in this study, while posterior capsular rupture and/or anterior vitrectomy occurred in 3.5% of veterans. 28 Multivariate analysis confirmed increased risk of RLF in beneficiaries who were male and elderly, consistent with known associations for increased risk of posterior capsular rupture. 29 Our analysis did not detect an increased risk of POAG following cataract surgery in beneficiaries with a prior history of anti-vegf intravitreal injections, contrary to recent studies identifying sustained elevation of intraocular pressure with intravitreal injections Further studies, with pathophysiologic and clinic correlations, are warranted to determine if significant IOP elevations are truly associated with intravitreal injections and
6 Hahn et al. Page 6 if cataract extraction affects this potential risk of POAG, through synergistic or antagonistic effects, in individuals with prior anti-vegf treatment. Multivariate analysis confirmed increased risk for POAG in beneficiaries who were older, male, black, and of other race, consistent with known associations. 30 Our analysis revealed an increased risk of endophthalmitis following cataract surgery in beneficiaries with a history of anti-vegf injections. In the group of endophthalmitis diagnoses within <40 days, the mean time to endophthalmitis was 11 days following cataract surgery, consistent with an association with cataract surgery in the setting of prior injections. In contrast, in both groups of endophthalmitis within <40 days and days, only approximately 6% of endophthalmitis claims were coded as chronic endophthalmitis. Furthermore, there was no difference in rate of chronic endophthalmitis between groups, suggesting that the increased risk of endophthalmitis days following cataract surgery may not represent chronic endophthalmitis from cataract surgery but rather acute endophthalmitis due to ongoing injections or possibly other procedures. Further analysis of the Medicare claims data is needed to determine if this increased risk of both early-onset and delayed endophthalmitis in beneficiaries with prior anti-vegf is affected by a history of cataract surgery or simply represents the increased risk inherent in intravitreal injections. This increased diagnosis of endophthalmitis is unlikely to result from differences in follow up, as the aggressive course of endophthalmitis will invariably prompt immediate evaluation and treatment. Although we excluded those with a prior history of an endophthalmitis Medicare claim, we did not exclude beneficiaries having received prior intravitreal injections of antibiotics. Inclusion of unclassified injections, previously shown to consist almost 100% of bevacizumab, 20 may have inadvertently included beneficiaries incorrectly coded for an injection of antibiotic agents, resulting in selection bias. Further studies are needed to confirm and better understand this relationship. Finally increased risk for the development of endophthalmitis within <40 days of surgery was observed in Medicare beneficiaries with a higher Charlson index 31, 32 We acknowledge several important study limitations. The Medicare database consists of information collected for billing purposes and not for clinical investigations. Conditions may sometimes have been incorrectly coded. In addition, the database does not include all potentially clinically relevant codes. The Medicare database is limited in its inability to identify which eye has the disease, and it is possible that the associations observed in this study apply to the same individual but in different eyes. By taking only the earliest case of cataract surgery, and censoring the patient should a subsequent cataract surgery occur, any errors in our study due to this limitation are likely to be on the conservative side. Beneficiaries with a history of prior intravitreal injections likely have a chronic eye disease requiring ongoing treatment, and increased diagnoses may have been secondary to differences in follow-up; this possibility is unlikely to affect subsequent removal of lens fragments, which is typically determined at the time of cataract surgery, or diagnosis of endophthalmitis given its aggressive course. Despite these limitations, Medicare claims data have been shown to be able to properly identify clinical phenomena in numerous studies Use of such data in ophthalmology dates back to , 37. Such data have
7 Hahn et al. Page 7 Acknowledgments been used to assess increased risk associated with ancillary procedures that occur at a date following receipt of cataract surgery 38. In a separate study (Paul Hahn, Kim Jiramongkolchai, Sandra Stinnett, Terry Kim. Increased rate of intraoperative complications during cataract surgery following intravitreal injections. Manuscript in preparation), Hahn and colleagues investigated clinical data from a review of 10,103 cataract surgeries at the Duke Eye Center from , in which we compared rates of intraoperative complications (posterior capsular rupture and/or anterior vitrectomy and/or unplanned 3-piece intraocular lens placement) during cataract surgery as well as rates of post-cataract surgery glaucoma-related outcomes and endophthalmitis in 197 eyes with prior intravitreal injections compared to a matched control group. In this clinical study, specific cataract surgery-related complications were directly assessed based on review of clinical records, identifying a significantly increased rate in eyes with a prior history of intravitreal injection compared to eyes without a history of injections (3% vs 0%, p=0.03). No difference in intraocular pressure or change in glaucoma medication or surgery requirements was noted after excluding steroid-injected patients, and no cases of endophthalmitis were identified in either group. While this clinical study lacked sufficiently large numbers to appropriately power such an analysis of rare events or subtle changes, such clinical records-based data provides complementary evidence to this claims-based analysis, which together suggest that a history of prior intravitreal injections may be a risk factor for cataract surgery-related complications. As the indications and need for intravitreal injections continue to rapidly grow, the burden of treatment has begun to extend beyond the retina specialist. Inadvertent interaction between the injection needle and the crystalline lens may result in increased rate of cataract surgeryrelated complications, and we recommend an intimate understanding of intraocular anatomic relationships, particularly of the posterior segment and lens, for practitioners performing intravitreal injections. Practitioners who perform such procedures should be aware of this potential for violation of the posterior capsule and be particularly mindful of these anatomic intraocular relationships, which are often displaced as the eye is intentionally torqued during the injection procedure. Similarly, cataract surgeons should be aware that a history of injections, which is becoming increasingly prevalent, may be an important risk factor for intraoperative complications. A vigilant preoperative assessment of the lens and posterior chamber should be performed with an understanding of surgical procedure modifications that can be performed to minimize intraoperative complications in the setting of a violated posterior capsule. 6 Cataract surgeons should also be aware that a history of prior injections may result in increased risk of postoperative endophthalmitis in addition to risk of intraoperative complications. Further studies are warranted to better explore these interactions. Financial Support: Paul Hahn reports a consulting agreement unrelated to the submitted work with Second Sight Medical Products. Arseniy Yashkin and Frank Sloan report that work on this article was supported in part by the National Institute on Aging (grant R01-AG017473)
8 Hahn et al. Page 8 The sponsor or funding organization had no role in the design or conduct of this research References 1. [Accessed January 29, 2014] Centers for Medicare and Medicaid Services Part B National Summary Data File Available at: Order/NonIdentifiableDataFiles/PartBNationalSummaryDataFile.html 2. Falavarjani KG, Nguyen QD. Adverse events and complications associated with intravitreal injection of anti-vegf agents: a review of literature. Eye (Lond). 2013; 27(7): [PubMed: ] 3. Day S, Acquah K, Mruthyunjaya P, et al. Ocular complications after anti-vascular endothelial growth factor therapy in Medicare patients with age-related macular degeneration. Am J Ophthalmol. 2011; 152(2): [PubMed: ] 4. Jonas JB, Spandau UH, Schlichtenbrede F. Short-term complications of intravitreal injections of triamcinolone and bevacizumab. Eye (Lond). 2008; 22(4): [PubMed: ] 5. Khalifa YM, Pantanelli SM. Quiescent posterior capsule trauma after intravitreal injection: implications for the cataract surgeon. J Cataract Refract Surg. 2011; 37(7):1364. [PubMed: ] 6. Saeed MU, Prasad S. Management of cataract caused by inadvertent capsule penetration during intravitreal injection of ranibizumab. J Cataract Refract Surg. 2009; 35(11): [PubMed: ] 7. Coca-Robinot J, Casco-Silva B, Armada-Maresca F, Garcia-Martinez J. Accidental injections of dexamethasone intravitreal implant (Ozurdex) into the crystalline lens. Eur J Ophthalmol. 2014; 24(4): [PubMed: ] 8. Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006; 355(14): [PubMed: ] 9. Brown DM, Michels M, Kaiser PK, et al. Ranibizumab versus verteporfin photodynamic therapy for neovascular age-related macular degeneration: Two-year results of the ANCHOR study. Ophthalmology. 2009; 116(1): e5. [PubMed: ] 10. Hoang QV, Mendonca LS, Della Torre KE, et al. Effect on intraocular pressure in patients receiving unilateral intravitreal anti-vascular endothelial growth factor injections. Ophthalmology. 2012; 119(2): [PubMed: ] 11. Bakri SJ, Moshfeghi DM, Francom S, et al. Intraocular pressure in eyes receiving monthly ranibizumab in 2 pivotal age-related macular degeneration clinical trials. Ophthalmology. 2014; 121(5): [PubMed: ] 12. SooHoo JR, Seibold LK, Kahook MY. The link between intravitreal antivascular endothelial growth factor injections and glaucoma. Curr Opin Ophthalmol. 2014; 25(2): [PubMed: ] 13. Slabaugh MA, Chen PP. The effect of cataract extraction on intraocular pressure. Curr Opin Ophthalmol. 2014; 25(2): [PubMed: ] 14. Kim SJ, Toma HS. Ophthalmic antibiotics and antimicrobial resistance a randomized, controlled study of patients undergoing intravitreal injections. Ophthalmology. 2011; 118(7): [PubMed: ] 15. Storey P, Dollin M, Pitcher J, et al. The role of topical antibiotic prophylaxis to prevent endophthalmitis after intravitreal injection. Ophthalmology. 2014; 121(1): [PubMed: ] 16. Lalwani GA, Flynn HW, Scott IU, et al. Acute-onset endophthalmitis after clear corneal cataract surgery ( ): clinical features, causative organisms, and visual acuity outcomes. Ophthalmology. 2008; 115(3): [PubMed: ] 17. Stein JD, Hanrahan BW, Comer GM, Sloan FA. Diffusion of technologies for the care of older adults with exudative age-related macular degeneration. American journal of ophthalmology. 2013; 155(4): e2. [PubMed: ] 18. Javitt JC, Vitale S, Canner JK, et al. National outcomes of cataract extraction I: retinal detachment after inpatient surgery. Ophthalmology. 1991; 98(6): [PubMed: ]
9 Hahn et al. Page Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. Journal of chronic diseases. 1987; 40(5): [PubMed: ] 20. Brechner RJ, Rosenfeld PJ, Babish JD, Caplan S. Pharmacotherapy for neovascular age-related macular degeneration: an analysis of the 100% 2008 medicare fee-for-service part B claims file. Am J Ophthalmol. 2011; 151(5): e1. [PubMed: ] 21. Rosenfeld PJ, Shapiro H, Ehrlich JS, et al. Cataract surgery in ranibizumab-treated patients with neovascular age-related macular degeneration from the phase 3 ANCHOR and MARINA trials. Am J Ophthalmol. 2011; 152(5): [PubMed: ] 22. Jonas JB, Kreissig I, Degenring RF. Cataract surgery after intravitreal injection of triamcinolone acetonide. Eye (Lond). 2004; 18(4): [PubMed: ] 23. Muzyka-Wozniak M. Phacoemulsification in eyes with neovascular AMD treated with anti-vegf injections. Eur J Ophthalmol. 2011; 21(6): [PubMed: ] 24. Tabandeh H, Chaudhry NA, Boyer DS, et al. Outcomes of cataract surgery in patients with neovascular age-related macular degeneration in the era of anti-vascular endothelial growth factor therapy. J Cataract Refract Surg. 2012; 38(4): [PubMed: ] 25. Gillies MC, Kuzniarz M, Craig J, et al. Intravitreal triamcinolone-induced elevated intraocular pressure is associated with the development of posterior subcapsular cataract. Ophthalmology. 2005; 112(1): [PubMed: ] 26. Kersey JP, Broadway DC. Corticosteroid-induced glaucoma: a review of the literature. Eye (Lond). 2006; 20(4): [PubMed: ] 27. Tielsch JM, Legro MW, Cassard SD, et al. Risk factors for retinal detachment after cataract surgery: a population-based case-control study. Ophthalmology. 1996; 103(10): [PubMed: ] 28. Greenberg PB, Tseng VL, Wu WC, et al. Prevalence and predictors of ocular complications associated with cataract surgery in United States veterans. Ophthalmology. 2011; 118(3): [PubMed: ] 29. American Academy of Ophthalmology Cataract and Anterior Segment Panel. Cataract in the Adult Eye. San Francisco, CA: American Academy of Ophthalmology; Preferred Practice Pattern Guidelines. Available at: American Academy of Ophthalmology Glaucoma Panel. Primary Open-Angle Glaucoma. San Francisco, CA: American Academy of Ophthalmology; Preferred Practice Pattern Guidelines. Available at Hatch WV, Cernat G, Wong D, et al. Risk factors for acute endophthalmitis after cataract surgery: a population-based study. Ophthalmology. 2009; 116(3): [PubMed: ] 32. Miller JJ, Scott IU, Flynn HW Jr, et al. Acute-onset endophthalmitis after cataract surgery ( ): incidence, clinical settings, and visual acuity outcomes after treatment. Am J Ophthalmol. 2005; 139(6): [PubMed: ] 33. Winkelmayer WC, Schneeweiss S, Mogun H, et al. Identification of individuals with CKD from Medicare claims data: a validation study. American Journal of Kidney Diseases. 2005; 46(2): [PubMed: ] 34. Hennessy S, Leonard CE, Freeman CP, et al. Validation of diagnostic codes for outpatient originating sudden cardiac death and ventricular arrhythmia in Medicaid and Medicare claims data. Pharmacoepidemiology and drug safety. 2010; 19(6): [PubMed: ] 35. Javitt JC, McBean AM, Sastry SS, DiPaolo F. Accuracy of coding in Medicare Part B claims: cataract as a case study. Archives of ophthalmology. 1993; 111(5): [PubMed: ] 36. Javitt JC, Vitale S, Canner JK, et al. National outcomes of cataract extraction: endophthalmitis following inpatient surgery. Archives of ophthalmology. 1991; 109(8): [PubMed: ] 37. Sloan FA, Brown DS, Carlisle ES, et al. Estimates of incidence rates with longitudinal claims data. Archives of Ophthalmology. 2003; 121(10): [PubMed: ] 38. Stein JD, Zacks DN, Grossman D, et al. Adverse events after pars plana vitrectomy among Medicare beneficiaries. Archives of ophthalmology. 2009; 127(12): [PubMed: ]
10 Hahn et al. Page 10 Figure 1. Selected Outcomes Following Cataract Surgery solid line No Anti-VEGF Injections; dash-dotted line Anti-VEGF Injections
11 Hahn et al. Page 11 Table 1 List of Study Codes Condition Administrative Code Cataract surgery CPT-4: with C9399 J3490 J3590 J2503 J9035 Q2024 C9257 C9233 J2778 Intravitreal injection of anti-vegf agent CPT-4: Q2046 C9291 on the same claim Outcomes Removal of RLF CPT-4: Primary open angle glaucoma ICD-9: 365.1x Endophthalmitis ICD-9: 360.0x Exclusion restrictions Intravitreal injection of corticosteroid CPT-4: with J3300 J3301 J3302 J7312 J1100 C9256 J1094 on the same claim Pars plana vitrectomy CPT-4: Gas pneumatic retinopexy CPT-4: Traumatic cataract ICD-9: 366.2x Cataract 2/2 ocular disorders ICD-9: 366.3x Cataract associated with other disorders ICD-9: 366.4x Infantile/congenital cataract ICD-9: 366.0x Coexisting conditions Pseudoexfoliation ICD-9: Diabetes mellitus ICD-9: 250.xx Diabetic retinopathy ICD-9: 362.0x Other glaucoma ICD-9: x 365.2x 365.3x 365.4x 365.5x 365.6x 365.7x 365.8x 365.9x Codes are drawn from International Classification of Disease, 9th Revision, Clinical Modification (ICD-9), and Current Procedural Terminology (CPT-4). Excludes Primary open angle glaucoma (ICD-9: 365.1x)
12 Hahn et al. Page 12 Table 2 Sample Selection Summary Sample Size Initial Sample 278,199 Loss to age restriction (68+) 33,040 Loss to Medicare Advantage membership restriction 12,358 Loss to location restriction 1,188 Loss to medical condition or procedure restriction 27,163 Loss due to prior steroid injection 563 Loss due to endophthalmitis prior to surgery 244 Retained Lens Fragment (RLF) Removal/Endophthalmitis Sample 203,643 Loss due to prior diagnosis of glaucoma 63,987 Primary Open Angle Glaucoma Sample 139,656
13 Hahn et al. Page 13 Table 3 Summary Statistics Sample Variable RLF Removal / Endophthalmitis Primary Open Angle Glaucoma Removal of RLF within 28 days Endophthalmitis within 39 days Endophthalmitis 40+ days Primary open angle glaucoma within 365 days Intravitreal injection of anti-vegf agent Diabetes mellitus Diabetic retinopathy Any glaucoma diagnosis Primary open angle glaucoma Other glaucoma in the absence of primary open angle glaucoma Pseudoexfoliation Charlson Index 4.51 (3.68) 4.39 (3.62) Male White Black Other Age (6.01) (5.97) Year Year Year Year Year N 203, ,656 Note: Numbers provided are proportion of sample for categorical variables and sample means with standard deviations in parentheses for continuous variables (Charlson Index, age).
14 Hahn et al. Page 14 Table 4 Cox Proportional Hazard Model Results Intravitreal injection of anti- VEGF agent Diabetes mellitus Diabetic retinopathy Glaucoma Pseudoexfoliation Charlson index Male Black Other Age Anterior Vitrectomy HR [95% CI] Primary Open Angle Glaucoma HR [95% CI] Outcome Endophthalmitis <40 days HR [95% CI] Endophthalmitis 40 days HR [95% CI] 2.26 * * 3.65 ** [ ] [ ] [ ] [ ] 1.25 [ ] 1.12 [ ] 1.77 [ ] Hazard Ratio (HR) and 95% Confidence Interval (CI) * p 0.05; ** p 0.01; Hazard ratios significant at these levels are in bold [ ] ** 1.02 [ ] [ ] [ ] [ ] 1.38 * 1.37 ** [ ] [ ] [ ] [ ] ** [ ] [ ] [ ] [ ] ** [ ] [ ] [ ] [ ] 1.03 ** 1.02 * [ ] [ ] [ ] [ ] Note: Analysis also includes covariates for the year of cataract surgery (not shown). None of the associated hazard ratios are significant at conventional levels.
Frequency of sustained IOP elevation in patients receiving intravitreal injections of anti-vegf agents for exudative age-related macular degeneration
 Frequency of sustained IOP elevation in patients receiving intravitreal injections of anti-vegf agents for exudative age-related Aim Frequency of sustained IOP elevation in patients receiving intravitreal
Frequency of sustained IOP elevation in patients receiving intravitreal injections of anti-vegf agents for exudative age-related Aim Frequency of sustained IOP elevation in patients receiving intravitreal
Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit)
 Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit) Line of Business: Medi-Cal Effective Date: May 17, 2017 Revision Date: May 17, 2017 This policy has
Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit) Line of Business: Medi-Cal Effective Date: May 17, 2017 Revision Date: May 17, 2017 This policy has
Subgroup Analysis of the MARINA Study of Ranibizumab in Neovascular Age-Related Macular Degeneration
 Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Ophthalmic VEGF Inhibitors. Eylea (aflibercept), Macugen (pegaptanib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Optometric Postoperative Cataract Surgery Management
 Financial Disclosures Optometric Postoperative Cataract Surgery Management David Dinh, OD Oak Cliff Eye Clinic Dallas Eye Consultants March 10, 2015 Comanagement Joint cooperation between two or more specialists
Financial Disclosures Optometric Postoperative Cataract Surgery Management David Dinh, OD Oak Cliff Eye Clinic Dallas Eye Consultants March 10, 2015 Comanagement Joint cooperation between two or more specialists
Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice
 Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice Andrew A. Moshfeghi, MD, MBA 1 ; John D. Pitcher, MD 2,3 ; Genevieve Lucas, B. Comm. 4 ; Nick
Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice Andrew A. Moshfeghi, MD, MBA 1 ; John D. Pitcher, MD 2,3 ; Genevieve Lucas, B. Comm. 4 ; Nick
Review Article Ophthalmic Vigilance in Antiangiogenic Pharmacotherapy
 Cronicon OPEN ACCESS OPHTHALMOLOGY Review Article Ophthalmic Vigilance in Antiangiogenic Pharmacotherapy Marianne Shahsuvaryan* Department of Ophthalmology, Yerevan State Medical University, Armenia *Corresponding
Cronicon OPEN ACCESS OPHTHALMOLOGY Review Article Ophthalmic Vigilance in Antiangiogenic Pharmacotherapy Marianne Shahsuvaryan* Department of Ophthalmology, Yerevan State Medical University, Armenia *Corresponding
The Economic$ and Epidemiology of Intravitreal Injections
 The Economic$ and Epidemiology of Intravitreal Injections 2016 Proctor Lecture I have the following financial interests or relationships to disclose: Alcon Laboratories, Inc.: Consultant/Advisor Allergan,
The Economic$ and Epidemiology of Intravitreal Injections 2016 Proctor Lecture I have the following financial interests or relationships to disclose: Alcon Laboratories, Inc.: Consultant/Advisor Allergan,
Date approved: 04/18/18. Approved by: Pharmacy and Therapeutics Quality Management Subcommittee Effective Date: Department of Origin: Pharmacy
 Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY
 EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration
 A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Measure #192: Cataracts: Complications within 30 Days Following Cataract Surgery Requiring Additional Surgical Procedures
 Measure #192: Cataracts: Complications within 30 Days Following Cataract Surgery Requiring Additional Surgical Procedures 2012 PHYSICIAN QUALITY REPORTING OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Measure #192: Cataracts: Complications within 30 Days Following Cataract Surgery Requiring Additional Surgical Procedures 2012 PHYSICIAN QUALITY REPORTING OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
Anti VEGF Agents in Retinal Disorders Current Scenario
 Retina Anti VEGF Agents in Retinal Disorders Current Scenario Charu Gupta MS Charu Gupta MS, Cyrus M. Shroff MD Shroff Eye Centre, New Delhi T is a group of proteins involved in the regulation of angiogenesis,
Retina Anti VEGF Agents in Retinal Disorders Current Scenario Charu Gupta MS Charu Gupta MS, Cyrus M. Shroff MD Shroff Eye Centre, New Delhi T is a group of proteins involved in the regulation of angiogenesis,
The Era of anti- - - VEGF Kirk L. Halvorson, OD
 The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
2017 MIPS Quality Category Measures and Benchmarks for Ophthalmology
 2017 MIPS Quality Category Measures and Benchmarks for Ophthalmology Physicians must report on 50% of all patients, if reporting via registry or EHR, and 50% of all Medicare Part B patients if reporting
2017 MIPS Quality Category Measures and Benchmarks for Ophthalmology Physicians must report on 50% of all patients, if reporting via registry or EHR, and 50% of all Medicare Part B patients if reporting
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Health Technology Appraisal. Aflibercept for treating diabetic macular oedema.
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
 Measure #192 (NQF 0564): Cataracts: Complications within 30 Days Following Cataract Surgery Requiring Additional Surgical Procedures National Quality Strategy Domain: Patient Safety 2016 PQRS OPTIONS FOR
Measure #192 (NQF 0564): Cataracts: Complications within 30 Days Following Cataract Surgery Requiring Additional Surgical Procedures National Quality Strategy Domain: Patient Safety 2016 PQRS OPTIONS FOR
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis)
 Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
2016 PQRS Recommended Measures for: Ophthalmology
 Measures Groups Choose 1 Measures Group Report on a minimum of 20 eligible patients (at least 11 must be Medicare Part B FFS patients) #130: Documentation of Current Medications in the Medical Record #226:
Measures Groups Choose 1 Measures Group Report on a minimum of 20 eligible patients (at least 11 must be Medicare Part B FFS patients) #130: Documentation of Current Medications in the Medical Record #226:
84 Year Old with Rosacea
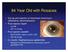 84 Year Old with Rosacea S/p tap and injection of intravitreal vancomycin, ceftazidime, dexamethasone Post-injection day#1 Va HM IOP 14 mmhg Post-injection week#3 BCVA 20/20-3 (plano +0.50 x 180) IOP 23
84 Year Old with Rosacea S/p tap and injection of intravitreal vancomycin, ceftazidime, dexamethasone Post-injection day#1 Va HM IOP 14 mmhg Post-injection week#3 BCVA 20/20-3 (plano +0.50 x 180) IOP 23
Diabetic Retinopathy: Managing the Extremes. J. Michael Jumper, MD West Coast Retina
 Diabetic Retinopathy: Managing the Extremes J. Michael Jumper, MD West Coast Retina Case 1: EC 65 y.o. HM No vision complaints Meds: Glyburide Metformin Pioglitazone Va: 20/20 OU 20/20 Case 2: HS 68 y.o.
Diabetic Retinopathy: Managing the Extremes J. Michael Jumper, MD West Coast Retina Case 1: EC 65 y.o. HM No vision complaints Meds: Glyburide Metformin Pioglitazone Va: 20/20 OU 20/20 Case 2: HS 68 y.o.
Note: This is an outcome measure and will be calculated solely using registry data.
 Quality ID #388: Cataract Surgery with Intra-Operative Complications (Unplanned Rupture of Posterior Capsule Requiring Unplanned Vitrectomy) National Quality Strategy Domain: Patient Safety 2018 OPTIONS
Quality ID #388: Cataract Surgery with Intra-Operative Complications (Unplanned Rupture of Posterior Capsule Requiring Unplanned Vitrectomy) National Quality Strategy Domain: Patient Safety 2018 OPTIONS
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Ocular Complications after Intravitreal Bevacizumab Injection in Eyes with Choroidal and Retinal Neovascularization
 Original Article Ocular Complications after Intravitreal Bevacizumab Injection in Eyes with Choroidal and Retinal Neovascularization Aimal Khan, P.S Mahar, Azfar Nafees Hanfi, Umair Qidwai Pak J Ophthalmol
Original Article Ocular Complications after Intravitreal Bevacizumab Injection in Eyes with Choroidal and Retinal Neovascularization Aimal Khan, P.S Mahar, Azfar Nafees Hanfi, Umair Qidwai Pak J Ophthalmol
Anti Vascular Endothelial Growth Factor Pharmacotherapy for Diabetic Macular Edema
 Ophthalmic Technology Assessment Anti Vascular Endothelial Growth Factor Pharmacotherapy for Diabetic Macular Edema A Report by the American Academy of Ophthalmology Allen C. Ho, MD, Ingrid U. Scott, MD,
Ophthalmic Technology Assessment Anti Vascular Endothelial Growth Factor Pharmacotherapy for Diabetic Macular Edema A Report by the American Academy of Ophthalmology Allen C. Ho, MD, Ingrid U. Scott, MD,
NEOVASCULAR AGE-RELATED MACULAR DEGENERation
 Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
MEDICAL POLICY SUBJECT: CORNEAL ULTRASOUND PACHYMETRY. POLICY NUMBER: CATEGORY: Technology Assessment
 MEDICAL POLICY SUBJECT: CORNEAL ULTRASOUND,, PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: CORNEAL ULTRASOUND,, PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
London Medicines Evaluation Network Review
 London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related macular degeneration.
 Thomas Jefferson University Jefferson Digital Commons Wills Eye Institute Papers Wills Eye Institute 4-1-2008 Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related
Thomas Jefferson University Jefferson Digital Commons Wills Eye Institute Papers Wills Eye Institute 4-1-2008 Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related
Clinical Trials Related to Age Related Macular Degeneration
 Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
ANTIVASCULAR ENDOTHELIAL GROWTH FACTOR
 THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
04/11/2014. Retina Coding and Reimbursement 101. Financial Disclosure. Chief Complaint
 Retina Coding and Reimbursement 101 William T. Koch, COA, COE, CPC Administrative Director Director of Billing Operations The Retina Institute St. Louis, Missouri Advisory Boards Allergan Genentech Regeneron
Retina Coding and Reimbursement 101 William T. Koch, COA, COE, CPC Administrative Director Director of Billing Operations The Retina Institute St. Louis, Missouri Advisory Boards Allergan Genentech Regeneron
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Ophthalmologic Policy. Vascular Endothelial Growth Factor (VEGF) Inhibitors
 Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
Note: This is an outcome measure and will be calculated solely using registry data.
 Quality ID #385: Adult Primary Rhegmatogenous Retinal Detachment Surgery: Visual Acuity Improvement Within 90 Days of Surgery National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR
Quality ID #385: Adult Primary Rhegmatogenous Retinal Detachment Surgery: Visual Acuity Improvement Within 90 Days of Surgery National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR
Surgery Outcomes Study
 British 3tournal of Ophthalmology 1996;80:689-693 689 Department of Social Medicine, University of Copenhagen, Denmark J C Norregaard T F Andersen Danish Institute of Clinical Epidemiology, Copenhagen,
British 3tournal of Ophthalmology 1996;80:689-693 689 Department of Social Medicine, University of Copenhagen, Denmark J C Norregaard T F Andersen Danish Institute of Clinical Epidemiology, Copenhagen,
Vanderbilt Eye Institute Clinical Trials
 April, 2010 Vanderbilt Eye Institute Clinical Trials Ophthalmology Actively Recruiting Studies For information on our clinical trials and other studies, please contact: Sandy Owings, COA, CCRP Clinic Director
April, 2010 Vanderbilt Eye Institute Clinical Trials Ophthalmology Actively Recruiting Studies For information on our clinical trials and other studies, please contact: Sandy Owings, COA, CCRP Clinic Director
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
TREATMENT STRATEGIES FOR CHORIORETINAL VASCULAR DISEASES: ADVANTAGES AND DISADVANTAGES OF INDIVIDUALISED THERAPY
 TREATMENT STRATEGIES FOR CHORIORETINAL VASCULAR DISEASES: ADVANTAGES AND DISADVANTAGES OF INDIVIDUALISED THERAPY *Michael W. Stewart Professor and Chairman, Mayo School of Medicine, Department of Ophthalmology,
TREATMENT STRATEGIES FOR CHORIORETINAL VASCULAR DISEASES: ADVANTAGES AND DISADVANTAGES OF INDIVIDUALISED THERAPY *Michael W. Stewart Professor and Chairman, Mayo School of Medicine, Department of Ophthalmology,
Diabetic maculopathy 11/ An update on. Miss Vasuki Sivagnanavel
 Miss Vasuki Sivagnanavel Consultant Ophthalmologist An update on Diabetic maculopathy Despite advances in the management of diabetes, diabetic retinopathy is already the commonest cause of blindness among
Miss Vasuki Sivagnanavel Consultant Ophthalmologist An update on Diabetic maculopathy Despite advances in the management of diabetes, diabetic retinopathy is already the commonest cause of blindness among
Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular Degeneration
 Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
04/06/2015. Documentation Do s and Don ts In The Retina Practice. Financial Disclosure. Documentation Dos and Don ts
 Documentation Do s and Don ts In The Retina Practice William T. Koch, COA, COE, CPC Administrative Director Director of Billing Operations The Retina Institute St. Louis, Missouri Advisory Boards Allergan
Documentation Do s and Don ts In The Retina Practice William T. Koch, COA, COE, CPC Administrative Director Director of Billing Operations The Retina Institute St. Louis, Missouri Advisory Boards Allergan
Cataract surgery is the leading cause of malpractice claims (OMIC) Complicated CE/IOL: Choices the anterior segment surgeon can make
 Posterior Segment Complications and Management of Retained Lens Material Jay M. Stewart, MD Cataract surgery is the leading cause of malpractice claims (OMIC) Complicated CE/IOL: Choices the anterior segment
Posterior Segment Complications and Management of Retained Lens Material Jay M. Stewart, MD Cataract surgery is the leading cause of malpractice claims (OMIC) Complicated CE/IOL: Choices the anterior segment
Landmark Tube Trials
 SECTION EDITOR: BARBARA SMIT, MD, PhD Landmark Tube Trials A review of key findings from recent multicenter randomized clinical trials involving tube shunts. BY AMBIKA HOGUET, MD, AND STEVEN J. GEDDE,
SECTION EDITOR: BARBARA SMIT, MD, PhD Landmark Tube Trials A review of key findings from recent multicenter randomized clinical trials involving tube shunts. BY AMBIKA HOGUET, MD, AND STEVEN J. GEDDE,
Management of Neovascular AMD
 Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
2018 MIPS Quality Category Measures and Benchmarks for Ophthalmology
 2018 MIPS Quality Category Measures and Benchmarks for Ophthalmology Physicians must report on 60% of all patients, if reporting via registry or EHR, and 60% of all Medicare Part B patients if reporting
2018 MIPS Quality Category Measures and Benchmarks for Ophthalmology Physicians must report on 60% of all patients, if reporting via registry or EHR, and 60% of all Medicare Part B patients if reporting
Cataract and Diabetic macular edema: What should I do?
 Cataract and Diabetic macular edema: What should I do? Irini Chatziralli Ophthalmic Surgeon, University Scholar 2 nd Department of Ophthalmology, University of Athens (Director: Prof P. Theodossiadis)
Cataract and Diabetic macular edema: What should I do? Irini Chatziralli Ophthalmic Surgeon, University Scholar 2 nd Department of Ophthalmology, University of Athens (Director: Prof P. Theodossiadis)
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Diabetic retinopathy (DR) progressively
 FOCUS ON DIABETIC MACULAR EDEMA * Frederick L. Ferris III, MD ABSTRACT The current state of treatment for diabetic macular edema (DME) is focused on slowing the rate of vision loss through assessment of
FOCUS ON DIABETIC MACULAR EDEMA * Frederick L. Ferris III, MD ABSTRACT The current state of treatment for diabetic macular edema (DME) is focused on slowing the rate of vision loss through assessment of
Note: This is an outcome measure and will be calculated solely using registry data.
 Measure #384: Adult Primary Rhegmatogenous Retinal Detachment Surgery: No Return to the Operating Room Within 90 Days of Surgery National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR
Measure #384: Adult Primary Rhegmatogenous Retinal Detachment Surgery: No Return to the Operating Room Within 90 Days of Surgery National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR
Ranibizumab and Bevacizumab for Treatment of Neovascular Age-related Macular Degeneration
 and for Treatment of Neovascular Age-related Macular Degeneration Two-Year Results Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group* Writing Committee: Daniel F. Martin,
and for Treatment of Neovascular Age-related Macular Degeneration Two-Year Results Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group* Writing Committee: Daniel F. Martin,
Risk of Fractures Following Cataract Surgery in Medicare Beneficiaries
 Risk of Fractures Following Cataract Surgery in Medicare Beneficiaries Victoria L. Tseng, MD, Fei Yu, PhD, Flora Lum, MD, Anne L. Coleman, MD, PhD JAMA. 2012;308(5):493-501 Background Visual impairment
Risk of Fractures Following Cataract Surgery in Medicare Beneficiaries Victoria L. Tseng, MD, Fei Yu, PhD, Flora Lum, MD, Anne L. Coleman, MD, PhD JAMA. 2012;308(5):493-501 Background Visual impairment
Clinical Policy: Implantable Miniature Telescope for Age Related Macular Degeneration Reference Number: CP.MP.517
 Clinical Policy: Implantable Miniature Telescope for Age Related Macular Reference Number: CP.MP.517 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Implantable Miniature Telescope for Age Related Macular Reference Number: CP.MP.517 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Diabetic Retinopatathy
 Diabetic Retinopatathy Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Arctic DX Macula Risk Advanced
Diabetic Retinopatathy Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Arctic DX Macula Risk Advanced
Curriculum Vitae Ryan M. Rich, M.D.
 Retina Consultants of Southern Colorado, P.C. 3030 North Circle Drive, Suite 301 Colorado Springs, CO 80909 (719) 473-9595 DOB: March 20, 1972 Citizenship: USA CURRENT POSITION 2008-present Retina Consultants
Retina Consultants of Southern Colorado, P.C. 3030 North Circle Drive, Suite 301 Colorado Springs, CO 80909 (719) 473-9595 DOB: March 20, 1972 Citizenship: USA CURRENT POSITION 2008-present Retina Consultants
Original Policy Date
 MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
Instudies of vascular endothelial growth factor
 MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA
 Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema
 Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema Young Jae Choi, MD, In Kyung Oh, MD, Jae Ryung Oh, MD, PhD, Kuhl Huh, MD, PhD Department of Ophthalmology,
Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema Young Jae Choi, MD, In Kyung Oh, MD, Jae Ryung Oh, MD, PhD, Kuhl Huh, MD, PhD Department of Ophthalmology,
Outcomes of Intravitreal Anti-VEGF Therapy for Diabetic Macular Edema in Routine Clinical Practice
 Outcomes of Intravitreal Anti-VEGF Therapy for Diabetic Macular Edema in Routine Clinical Practice John D. Pitcher, MD 1,2 ; Andrew A. Moshfeghi, MD 3 ; Genevieve Lucas, B. Comm. 4 ; Nick Boucher, BS 4
Outcomes of Intravitreal Anti-VEGF Therapy for Diabetic Macular Edema in Routine Clinical Practice John D. Pitcher, MD 1,2 ; Andrew A. Moshfeghi, MD 3 ; Genevieve Lucas, B. Comm. 4 ; Nick Boucher, BS 4
Issue 15 The following key clinical peer reviewed journals will be reviewed: MONTHLY RESEARCH UPDATE 151(3) American Journal of Ophthalmology 129(5)
 Welcome to Bausch and Lomb s monthly research update. With our background in clinical ophthalmic research, mainly of the anterior eye, Bausch and Lomb have asked us to produce an independent report of
Welcome to Bausch and Lomb s monthly research update. With our background in clinical ophthalmic research, mainly of the anterior eye, Bausch and Lomb have asked us to produce an independent report of
Applications of Sustained Release Delivery Systems in Ocular Disease
 Applications of Sustained Release Delivery Systems in Ocular Disease Formulation and Delivery Systems For Peptide and Protein 2012 December 5, 2012 Christopher A. Rhodes, Ph.D. Principal, Christopher A
Applications of Sustained Release Delivery Systems in Ocular Disease Formulation and Delivery Systems For Peptide and Protein 2012 December 5, 2012 Christopher A. Rhodes, Ph.D. Principal, Christopher A
Practical Care of the Cataract Patient with Retinal Disease
 Practical Care of the Cataract Patient with Retinal Disease Brooks R. Alldredge, OD, FAAO Kelly L. Cyr, OD, FAAO The Retina Center Eye Associates of New Mexico 4411 The 25 Way NE, Suite 325 Albuquerque,
Practical Care of the Cataract Patient with Retinal Disease Brooks R. Alldredge, OD, FAAO Kelly L. Cyr, OD, FAAO The Retina Center Eye Associates of New Mexico 4411 The 25 Way NE, Suite 325 Albuquerque,
Fundamentals of Retina Coding
 Fundamentals of Retina Coding Presented by: Joy Woodke, COE, OCS Sunday, April 2, 2017 ASRS Business of Retina Meeting Dallas, TX American Academy of Ophthalmic Executives Financial Disclosure Joy Woodke,
Fundamentals of Retina Coding Presented by: Joy Woodke, COE, OCS Sunday, April 2, 2017 ASRS Business of Retina Meeting Dallas, TX American Academy of Ophthalmic Executives Financial Disclosure Joy Woodke,
Clinical Study Intravitreal Dexamethasone in the Management of Delayed-Onset Bleb-Associated Endophthalmitis
 International Inflammation Volume 2012, Article ID 503912, 5 pages doi:10.1155/2012/503912 Clinical Study Intravitreal Dexamethasone in the Management of Delayed-Onset Bleb-Associated Endophthalmitis David
International Inflammation Volume 2012, Article ID 503912, 5 pages doi:10.1155/2012/503912 Clinical Study Intravitreal Dexamethasone in the Management of Delayed-Onset Bleb-Associated Endophthalmitis David
 www.brisbaneeyeclinic.com.au Brisbane Eye Clinic is a modern ophthalmology practice focused on the provision of excellent medical eye care. The Clinic has two convenient consulting locations, our Wickham
www.brisbaneeyeclinic.com.au Brisbane Eye Clinic is a modern ophthalmology practice focused on the provision of excellent medical eye care. The Clinic has two convenient consulting locations, our Wickham
Facts About Diabetic Eye Disease
 Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Combination Treatment of Diabetic Macular Edema with Anti-Vascular Endothelial Growth Factor and Steroids: Analysis of DRCR.
 REVIEW ARTICLE Combination Treatment of Diabetic Macular Edema with Anti-Vascular Endothelial Growth Factor and Steroids: Analysis of DRCR.net Protocol U Cindy Ung 1, Kareem Moussa 1, Yoshihiro Yonekawa
REVIEW ARTICLE Combination Treatment of Diabetic Macular Edema with Anti-Vascular Endothelial Growth Factor and Steroids: Analysis of DRCR.net Protocol U Cindy Ung 1, Kareem Moussa 1, Yoshihiro Yonekawa
PRECISION PROGRAM. Injection Technique Quick-Reference Guide. Companion booklet for the Video Guide to Injection Technique
 Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Intravitreal Injection
 for patients Eye Clinic Ipswich Hospital Tel: 01473 703230 Intravitreal Injection What is an intravitreal injection? An intravitreal injection is the injection of a drug into the vitreous body (the jelly
for patients Eye Clinic Ipswich Hospital Tel: 01473 703230 Intravitreal Injection What is an intravitreal injection? An intravitreal injection is the injection of a drug into the vitreous body (the jelly
THE ECONOMIC BURDEN OF DIABETIC MACULAR EDEMA IN A WORKING AGE AND COMMERCIALLY INSURED POPULATION. Christopher John Wallick
 THE ECONOMIC BURDEN OF DIABETIC MACULAR EDEMA IN A WORKING AGE AND COMMERCIALLY INSURED POPULATION Christopher John Wallick A thesis submitted in partial fulfillment of the requirements for the degree
THE ECONOMIC BURDEN OF DIABETIC MACULAR EDEMA IN A WORKING AGE AND COMMERCIALLY INSURED POPULATION Christopher John Wallick A thesis submitted in partial fulfillment of the requirements for the degree
Retinal Diseases. Age-Related Macular Degeneration. What Is AMD? Risk Factors for AMD
 Santosh C. Patel, M.D. Retina Specialists The Latest And Greatest in the Management of Retinal Diseases February 13, 2007 Retinal Diseases Majority of Blindness in Civilized World AMD Diabetic Retinopathy
Santosh C. Patel, M.D. Retina Specialists The Latest And Greatest in the Management of Retinal Diseases February 13, 2007 Retinal Diseases Majority of Blindness in Civilized World AMD Diabetic Retinopathy
The Journal of Thoracic and Cardiovascular Surgery
 Accepted Manuscript Chronic type A dissection: when to operate? Francois Dagenais, MD PII: S0022-5223(18)33131-3 DOI: https://doi.org/10.1016/j.jtcvs.2018.11.032 Reference: YMTC 13781 To appear in: The
Accepted Manuscript Chronic type A dissection: when to operate? Francois Dagenais, MD PII: S0022-5223(18)33131-3 DOI: https://doi.org/10.1016/j.jtcvs.2018.11.032 Reference: YMTC 13781 To appear in: The
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Digital Journal of Ophthalmology, Vol. 21 Digital Journal of Ophthalmology, Vol. 21
 Original Article Feasibility and efficacy of a mass switch from ranibizumab (Lucentis) to bevacizumab (Avastin) for treatment of neovascular age-related macular degeneration Michael T. Andreoli, MD, a,b
Original Article Feasibility and efficacy of a mass switch from ranibizumab (Lucentis) to bevacizumab (Avastin) for treatment of neovascular age-related macular degeneration Michael T. Andreoli, MD, a,b
Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema
 111 ORIGINAL Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema Toshihiko Nagasawa, Takeshi Naito, Shingo Matsushita, Hiroyuki Sato, Takashi Katome, and
111 ORIGINAL Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema Toshihiko Nagasawa, Takeshi Naito, Shingo Matsushita, Hiroyuki Sato, Takashi Katome, and
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Note: This is an outcome measure and will be calculated solely using MIPS eligible clinician, group, or third party intermediary submitted data.
 Quality ID #384: Adult Primary Rhegmatogenous Retinal Detachment Surgery: No Return to the Operating Room Within 90 Days of Surgery National Quality Strategy Domain: Effective Clinical Care Meaningful
Quality ID #384: Adult Primary Rhegmatogenous Retinal Detachment Surgery: No Return to the Operating Room Within 90 Days of Surgery National Quality Strategy Domain: Effective Clinical Care Meaningful
MEASURING THE SIZE OF A TREATMENT EFFECT: RELATIVE RISK REDUCTION, ABSOLUTE RISK REDUCTION, AND NUMBER NEEDED TO TREAT
 MEASURING THE SIZE OF A TREATMENT EFFECT: RELATIVE RISK REDUCTION, ABSOLUTE RISK REDUCTION, AND NUMBER NEEDED TO TREAT Hussein Hollands, MD, MS (epid) Peter J. Kertes, MD, CM, FRCS(C) 72 The purpose of
MEASURING THE SIZE OF A TREATMENT EFFECT: RELATIVE RISK REDUCTION, ABSOLUTE RISK REDUCTION, AND NUMBER NEEDED TO TREAT Hussein Hollands, MD, MS (epid) Peter J. Kertes, MD, CM, FRCS(C) 72 The purpose of
Clinical Policy: Aflibercept (Eylea) Reference Number: CP.PHAR.184
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Ophthalmology: Questions and Answers. Current Topics in Ophthalmology. Disclosures: Common Questions:
 Current Topics in Ophthalmology Ophthalmology: Questions and Answers Jacque Duncan, MD Professor, Clinical Ophthalmology UCSF Primary Care Medicine: Principles and Practice October 30, 2013 With help from:
Current Topics in Ophthalmology Ophthalmology: Questions and Answers Jacque Duncan, MD Professor, Clinical Ophthalmology UCSF Primary Care Medicine: Principles and Practice October 30, 2013 With help from:
Presumed Sterile Endophthalmitis After Intravitreal Triamcinolone (Kenalog) More Common and Less Benign Than We Thought?
 original clinical study Presumed Sterile Endophthalmitis After Intravitreal Triamcinolone (Kenalog) More ommon and ess Benign Than We Thought? Angie H.. Fong, FOphthHK, FHKAM,* and armen K. M. han, FSEd(Ophth),
original clinical study Presumed Sterile Endophthalmitis After Intravitreal Triamcinolone (Kenalog) More ommon and ess Benign Than We Thought? Angie H.. Fong, FOphthHK, FHKAM,* and armen K. M. han, FSEd(Ophth),
Darren J. Bell, M.D. Texas A&M University College of Medicine College Station, Texas Doctor of Medicine
 Darren J. Bell, M.D. Education Texas A&M University College of Medicine College Station, Texas Doctor of Medicine 1997-2001 Texas A&M University College Station, Texas Bachelor of Science, Biomedical Engineering
Darren J. Bell, M.D. Education Texas A&M University College of Medicine College Station, Texas Doctor of Medicine 1997-2001 Texas A&M University College Station, Texas Bachelor of Science, Biomedical Engineering
Aging & Ophthalmology
 Aging & Ophthalmology Pr Jean-Marie Rakic Dr Denis Malaise January 2018 Major ocular diseases 1. Cataract 2. Age-related macular degeneration 3. Ischemic optic neuropathy 4. Horton arteritis 5. Glaucoma
Aging & Ophthalmology Pr Jean-Marie Rakic Dr Denis Malaise January 2018 Major ocular diseases 1. Cataract 2. Age-related macular degeneration 3. Ischemic optic neuropathy 4. Horton arteritis 5. Glaucoma
International Journal of Health Sciences and Research ISSN:
 International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
 www.brisbaneeyeclinic.com.au Brisbane Eye Clinic is a modern ophthalmology practice focused on the provision of excellent medical eye care. The Clinic has two convenient consulting locations, our Wickham
www.brisbaneeyeclinic.com.au Brisbane Eye Clinic is a modern ophthalmology practice focused on the provision of excellent medical eye care. The Clinic has two convenient consulting locations, our Wickham
OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS
 UnitedHealthcare Commercial Medical Benefit Drug Policy OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS Policy Number: PHA020 Effective Date: September 1, 2018 Table of Contents
UnitedHealthcare Commercial Medical Benefit Drug Policy OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS Policy Number: PHA020 Effective Date: September 1, 2018 Table of Contents
Updates and Controversies
 Updates and Controversies Philippine Journal of OPHTHALMOLOGY Vascular endothelial growth factor (VEGF) and inflammation. VEGF-A circulates normally in the body and is essential in endothelial cell growth.
Updates and Controversies Philippine Journal of OPHTHALMOLOGY Vascular endothelial growth factor (VEGF) and inflammation. VEGF-A circulates normally in the body and is essential in endothelial cell growth.
Intravitreal Corticosteroid Implants
 Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
The Wilmer Eye Institute s 34 th Annual Current Concepts in Ophthalmology March 13-17, 2017 Vail Marriott * Vail, Colorado
 The Wilmer Eye Institute s 34 th Annual Current Concepts in Ophthalmology March 13-17, 2017 Vail Marriott * Vail, Colorado Tentative 1/23/17 PROGRAM MONDAY, MARCH 13, 2017 Morning Session 6:00-7:00 Registration
The Wilmer Eye Institute s 34 th Annual Current Concepts in Ophthalmology March 13-17, 2017 Vail Marriott * Vail, Colorado Tentative 1/23/17 PROGRAM MONDAY, MARCH 13, 2017 Morning Session 6:00-7:00 Registration
Diagnosis and treatment of diabetic retinopathy. Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City
 Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Dr. Harvey Richman, OD, FAAO, FCOVD Diplomate American Board of Optometry Executive Committee AOA Third Party Center Founder Ask the AOA Coding
 Dr. Harvey Richman, OD, FAAO, FCOVD Diplomate American Board of Optometry Executive Committee AOA Third Party Center Founder Ask the AOA Coding Experts 92000 Codes Special Ophthalmological Services Describe
Dr. Harvey Richman, OD, FAAO, FCOVD Diplomate American Board of Optometry Executive Committee AOA Third Party Center Founder Ask the AOA Coding Experts 92000 Codes Special Ophthalmological Services Describe
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Small Capsulorrhexis Leads to Loss of Visual Function at Night
 Small Capsulorrhexis Leads to Loss of Visual Function at Night Contributions of the Capsulorrhexis to Straylight. van der Meulen IJ, Engelbrecht LA, et al:: Arch Ophthalmol 2009; 127 (October): 1290-1295
Small Capsulorrhexis Leads to Loss of Visual Function at Night Contributions of the Capsulorrhexis to Straylight. van der Meulen IJ, Engelbrecht LA, et al:: Arch Ophthalmol 2009; 127 (October): 1290-1295
