Intraocular Radiation Therapy for Age-Related Macular Degeneration
|
|
|
- Avice Atkins
- 5 years ago
- Views:
Transcription
1 Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER Medical Policies are developed to provide guidance for members and providers regarding coverage in accordance with contract terms. Benefit determinations are based in all cases on the applicable contract language. To the extent there may be any conflict between the Medical Policy and contract language, the contract language takes precedence. PLEASE NOTE: Contracts exclude from coverage, among other things, services or procedures that are considered investigational or cosmetic. Providers may bill members for services or procedures that are considered investigational or cosmetic. Providers are encouraged to inform members before rendering such services that the members are likely to be financially responsible for the cost of these services. DESCRIPTION Epiretinal radiation describes the intraocular administration of radiation to the choroidal vascular bed of the retina to treat age-related macular degeneration (AMD). MEDICAL POLICY CRITERIA Epiretinal radiation therapy for the treatment of subfoveal choroidal neovascularization in patients with neovascular age-related macular degeneration is considered investigational. NOTE: A summary of the supporting rationale for the policy criteria is at the end of the policy. CROSS REFERENCES 1. Charged-Particle (Proton) Radiotherapy, Medicine, Policy No Stereotactic Radiosurgery and Stereotactic Body Radiation Therapy, Surgery Policy No. 16 BACKGROUND Age-related macular degeneration (AMD) is characterized in its earliest stages by minimal MED134 1
2 visual impairment and the presence of large drusen and other pigmentary abnormalities on ophthalmoscopic examination. Two distinctively different forms of degeneration may be observed. The first, called the atrophic or areolar or dry form, evolves slowly. Atrophic AMD is the most common form of degeneration and may be a precursor of the more visually impairing exudative neovascular form, also referred to as disciform or wet AMD. The wet form is distinguished from the atrophic form by the development of choroidal neovascularization (CNV) and serous or hemorrhagic detachment of the retinal pigment epithelium. Risk of developing severe irreversible loss of vision is greatly increased by the presence of CNV. REGULATORY STATUS There are no devices specifically approved by the U.S. Food and Drug Administration (FDA) for this procedure. An investigational device exemption has been granted by FDA for a phase III multicenter trial to provide data for application to the FDA of the NeoVista Epi-Rad90 Ophthalmic System. This system has been developed to treat CNV by focal delivery of radiation to a subfoveal choroidal neovascular lesion. Using a standard vitrectomy procedure, the cannula tip of a handheld (pipette-like) surgical device is inserted into the vitreous cavity and positioned under visual guidance over the target lesion. The radiation source (strontium- 90) is advanced down the cannula until it reaches the tip, which is then held in place over the lesion for a prescribed time to deliver focused radiation. The system is designed to deliver a one-time peak dose of beta particle energy (24 Gy) for a target area 3 mm in depth and up to 5.4 mm in diameter. This is believed to be below the dose that is toxic to the retina and optic nerve, and radiation exposure outside of the target area is expected to be minimal. NOTE: Proton beam therapy and stereotactic radiation therapy for choroidal neovascularization (CNV) are considered in separate medical policies. See cross-reference section above. EVIDENCE SUMMARY Evidence from randomized controlled trials (RCTs) comparing patients treated with epiretinal radiation therapy, a form of brachytherapy, with those receiving standard treatment (such as treatment with anti vascular endothelial growth factors) is necessary in order to establish the safety and efficacy of epiretinal radiation in the treatment of wet age-related macular degeneration (AMD). EPI-RAD90 BRACHYTHERAPY Randomized Controlled Trials (RCTs) In 2016 Jackson reported the initial results from a phase 3 Macular Epiretinal Brachytherapy versus Ranibizumab (Lucentis) Only Treatment (MERLOT) trial, which was designed to investigate the safety and efficacy of epimacular brachytherapy (EMBT) as a second-line treatment for chronic, active neovascular AMD. [1] The primary objective was to assess if EMBT reduced the ongoing need for anti-vegf therapy in those who had already commenced intravitreal injections. This study was a multi-center randomized controlled trial that enrolled 363 participants with AMD who were receiving ranibizumab therapy at the time of screening, with 224 patients being randomized to the ranibizumab plus EBMT arm, and the remaining 119 patients remaining in the ranibizumab alone group. At twelve months follow-up, the investigators reported that the proportion of participants losing fewer than 15 letters was 84% in the EMBT arm and 92% in the ranibizumab arm (p = 0.007). All of measurable outcomes MED134 2
3 were not significant between the treatment groups. The investigators concluded that although still needing longer term follow-up to determine safety, that the initial results did not support the use of EMBT for chronic, active, neovascular AMD. In a multicenter, randomized, active-controlled, phase III clinical trial, authors evaluated the safety and efficacy of epimacular brachytherapy (EMBT) for the treatment of neovascular AMD [2] using results from the CABERNET study, which included 494 patients with treatmentnaïve neovascular AMD. Authors concluded the 2-year efficacy study did not support the routine use of EMBT for treatment-naïve wet AMD, despite an acceptable safety profile. Authors suggested that further safety review is required. Using the same patient data set from the CABERNET study, authors reported the fluorescein angiography (FA) and optical coherence tomography (OCT) results of a clinical trial of EMBT used for the treatment of neovascular AMD. [3] Authors concluded that both FA and OCT suggested that EMBT with pro re nata (PRN) ranibizumab results in an inferior structural outcome than quarterly plus PRN ranibizumab. Authors suggested that a non-vision-threatening radiation retinopathy occurs in 2.9% of eyes over 24 months, but longer follow-up is needed. Nonrandomized Studies In 2016, Ranjbar reported results from an retrospective study of 32 patients (32 eyes) with neovascular AMD who met criteria for the INTREPID trial for best responders and were treated with SRT (16-Gy) along with aflibercept or ranibizumab. [4] For the study s primary outcome, the number of anti-vegf treatments in the 12 months post-srt, significantly fewer intravitreal injections were given compared with the year preceding SRT (3.47 vs 6.81, P< ). No ocular or systemic adverse events occurred. Results at 12- and 24-month results from the multicenter MERITAGE study (NCT ) were reported in 2012 and [5-7] MERITAGE was a Phase I/II study of the EPI-RAD90 for the treatment of subfoveal CNV associated with wet AMD in patients requiring continued anti-vegf therapy to maintain an adequate response. Following a single 24-Gy dose, the 53 patients in the study received retreatment with ranibizumab administered monthly (as needed). At 12-month follow-up, 81% of patients maintained stable vision (loss of fewer than 15 letters) with a mean of 3.49 anti-vegf injections (0.29 per month). This was compared with 0.45 injections per participant per month in the 12 months before the study. Over 24 months, 68% of patients maintained stable vision with a mean of 8.7 anti-vegf injections (0.72 per month), which was not less than the number of injections required in the 12 months before treatment. A total of three publications from two studies have been reported by Avila on epiretinal radiation using the EPI-RAD90 system. [8-10] One report described 12-month safety and visual acuity results of a feasibility study in 34 treatment-naïve patients recruited between February 2005 and February [10] The second report described 12-month safety and visual acuity results from 24-Gy epiretinal radiation combined with bevacizumab in 34 treatment-naïve patients enrolled between June 2006 and April [9] Adverse events related to the device or procedure included subretinal hemorrhage (n=1), retinal tear (n=1), subretinal fibrosis (n=2), epiretinal membrane (n=1), and cataract (6 of 24; 24 patients were phakic at baseline). All occurrences of cataracts were deemed to be related to the vitrectomy procedure. Two- and 3- year results from this trial were published in [8] All 34 subjects were followed up for 24 months; one site that enrolled 19 patients agreed to reconsenting and following patients for three years. On average, the cohort of subjects followed for 36 months received 3.0 bevacizumab injections. MED134 3
4 A total of 12 of the 24 phakic patients (50%) developed cataracts, and 4 had phacoemulsification with intraocular lens implantation. The mean change in visual acuity at 36 months was +3.9 letters. Seven of 13 phakic patients (54%) developed cataracts, and 4 had phacoemulsification with intraocular lens implantation. One case of nonproliferative radiation retinopathy was observed at 36 months of follow-up. Section Summary While these studies contribute to the body of knowledge on epiretinal radiation by providing direction for future research, the evidence from these studies do not permit conclusions due to methodological limitations, including non-random allocation of treatment and a lack of adequate comparison group. Lack of adequate control groups limits the ability to control for many types of bias that may influence treatment outcomes. PRACTICE GUIDELINE SUMMARY AMERICAN ACADEMY OF OPHTHALMOLOGY (AAO) In 2015, the AAO updated their Preferred Practice Parameter regarding AMD. [11] For extrafoveal CNV lesions, guideline authors stated that the evidence is insufficient to recommend radiation therapy. There was no discussion or recommendation for the use of radiation treatment for subfoveal CNV. SUMMARY There is not enough research to show that epiretinal radiation therapy improves health outcomes for people with wet age-related macular degeneration (AMD). No clinical guidelines based on research recommend epiretinal radiation therapy for people with wet AMD. Therefore, epiretinal radiation is considered investigational for all indications, including but not limited to wet AMD. REFERENCES 1. Jackson, TL, Desai, R, Simpson, A, et al. Epimacular Brachytherapy for Previously Treated Neovascular Age-Related Macular Degeneration (MERLOT): A Phase 3 Randomized Controlled Trial. Ophthalmology Jun;123(6): PMID: Dugel, PU, Bebchuk, JD, Nau, J, et al. Epimacular brachytherapy for neovascular agerelated macular degeneration: a randomized, controlled trial (CABERNET). Ophthalmology Feb;120(2): PMID: Jackson, TL, Dugel, PU, Bebchuk, JD, et al. Epimacular Brachytherapy for Neovascular Age-Related Macular Degeneration (CABERNET): Fluorescein Angiography and Optical Coherence Tomography. Ophthalmology Mar 12. PMID: Ranjbar, M, Kurz, M, Holzhey, A, Melchert, C, Rades, D, Grisanti, S. Stereotactic radiotherapy in neovascular age-related macular degeneration: Real-life efficacy and morphological evaluation of the outer retina-choroid complex. Medicine Dec;95(52):e5729. PMID: MED134 4
5 5. Dugel, PU, Petrarca, R, Bennett, M, et al. Macular epiretinal brachytherapy in treated age-related macular degeneration: MERITAGE study: twelve-month safety and efficacy results. Ophthalmology Jul;119(7): PMID: Petrarca, R, Dugel, PU, Nau, J, Slakter, JS, Jaffe, GJ, Jackson, TL. Macular epiretinal brachytherapy in treated age-related macular degeneration (MERITAGE): month 12 optical coherence tomography and fluorescein angiography. Ophthalmology Feb;120(2): PMID: Petrarca, R, Dugel, PU, Bennett, M, et al. Macular epiretinal brachytherapy in treated age-related macular degeneration (MERITAGE): month 24 safety and efficacy results. Retina May;34(5): PMID: Avila, MP, Farah, ME, Santos, A, et al. Three-year safety and visual acuity results of epimacular 90 strontium/90 yttrium brachytherapy with bevacizumab for the treatment of subfoveal choroidal neovascularization secondary to age-related macular degeneration. Retina Jan;32(1):10-8. PMID: Avila, MP, Farah, ME, Santos, A, Duprat, JP, Woodward, BW, Nau, J. Twelve-month short-term safety and visual-acuity results from a multicentre prospective study of epiretinal strontium-90 brachytherapy with bevacizumab for the treatment of subfoveal choroidal neovascularisation secondary to age-related macular degeneration. Br J Ophthalmol Mar;93(3): PMID: Avila, MP, Farah, ME, Santos, A, et al. Twelve-month safety and visual acuity results from a feasibility study of intraocular, epiretinal radiation therapy for the treatment of subfoveal CNV secondary to AMD. Retina Feb;29(2): PMID: American Academy of Ophthalmology (AAO). January Prefered Practice Pattern. Age-related macular degeneration.. [cited 06/25/2018]; Available from: BlueCross BlueShield Association Medical Policy Reference Manual "Intraocular Radiotherapy for Age-Related Macular Degeneration." Policy No. 134 [12] CODES Codes Number Description CPT 0190T Placement of intraocular radiation source applicator (List separately in addition to primary procedure) Note: 0190T differs from code (destruction of localized lesion of the retina (e.g., macular edema, tumors), one or more sessions; radiation by implantation of source) because the radiation source is not implanted Vitrectomy, mechanical, pars plana approach HCPCS None Note: 0190T is to be used in conjunction with Date of Origin: August 2008 MED134 5
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
Intraocular Radiotherapy for Age- Related Macular Degeneration
 Intraocular Radiotherapy for Age- Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2017 Origination: 9/2008 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Intraocular Radiotherapy for Age- Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2017 Origination: 9/2008 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
A Little Physics. How Does It Work? Radiation Therapy for Choroidal Neovascularisation in AMD A Review. => cell death
 Radiation Therapy for Choroidal Neovascularisation in AMD A Review Jen Anikina ST4 Hillingdon Hospital A Little Physics One gray is the absorption of one joule of ionizing radiation energy per kilogram
Radiation Therapy for Choroidal Neovascularisation in AMD A Review Jen Anikina ST4 Hillingdon Hospital A Little Physics One gray is the absorption of one joule of ionizing radiation energy per kilogram
Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283
 Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Implantable Miniature Telescope for Age Related Macular Degeneration Reference Number: CP.MP.517
 Clinical Policy: Implantable Miniature Telescope for Age Related Macular Reference Number: CP.MP.517 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Implantable Miniature Telescope for Age Related Macular Reference Number: CP.MP.517 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Ophthalmic VEGF Inhibitors. Eylea (aflibercept), Macugen (pegaptanib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Diagnosis and treatment of diabetic retinopathy. Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City
 Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
Fundamentals of Retina Coding
 Fundamentals of Retina Coding Presented by: Joy Woodke, COE, OCS Sunday, April 2, 2017 ASRS Business of Retina Meeting Dallas, TX American Academy of Ophthalmic Executives Financial Disclosure Joy Woodke,
Fundamentals of Retina Coding Presented by: Joy Woodke, COE, OCS Sunday, April 2, 2017 ASRS Business of Retina Meeting Dallas, TX American Academy of Ophthalmic Executives Financial Disclosure Joy Woodke,
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187
 Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Ophthalmology Macular Pathways
 Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR EFFECTIVE DATE: 07/20/00 SUBFOVEAL CHOROIDAL NEOVASCULARIZATION
 MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR PAGE: 1 OF: 8 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR PAGE: 1 OF: 8 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Instudies of vascular endothelial growth factor
 MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
Photodynamic Therapy for Choroidal Neovascularization
 Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Management of Neovascular AMD
 Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
The College of Optometrists - Learning outcomes for the Professional Certificate in Medical Retina
 Learning outcomes for the Professional Certificate in Medical Retina, incorporating diabetic retinopathy screening and age related macular degeneration The professional certificate is a prerequisite to
Learning outcomes for the Professional Certificate in Medical Retina, incorporating diabetic retinopathy screening and age related macular degeneration The professional certificate is a prerequisite to
Clinical Trials Related to Age Related Macular Degeneration
 Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration
 A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
Original Policy Date
 MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Health Technology Appraisal. Aflibercept for treating diabetic macular oedema.
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials
 Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials Glenn J. Jaffe, MD, 1 Daniel F. Martin, MD, 2 Cynthia A. Toth, MD, 1 Ebenezer Daniel, MPH, PhD,
Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials Glenn J. Jaffe, MD, 1 Daniel F. Martin, MD, 2 Cynthia A. Toth, MD, 1 Ebenezer Daniel, MPH, PhD,
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D.
 Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
A population based study of macular choroidal neovascularization using optical coherence tomography in Eastern China
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 8: 371-376, 2014 A population based study of macular choroidal neovascularization using optical coherence tomography in Eastern China JIE ZHAO, JUN HU, HAO LU and
EXPERIMENTAL AND THERAPEUTIC MEDICINE 8: 371-376, 2014 A population based study of macular choroidal neovascularization using optical coherence tomography in Eastern China JIE ZHAO, JUN HU, HAO LU and
London Medicines Evaluation Network Review
 London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
International Journal of Health Sciences and Research ISSN:
 International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: December 7, 2011 Subject: Page: 1 of 23
 Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
Darren J. Bell, M.D. Texas A&M University College of Medicine College Station, Texas Doctor of Medicine
 Darren J. Bell, M.D. Education Texas A&M University College of Medicine College Station, Texas Doctor of Medicine 1997-2001 Texas A&M University College Station, Texas Bachelor of Science, Biomedical Engineering
Darren J. Bell, M.D. Education Texas A&M University College of Medicine College Station, Texas Doctor of Medicine 1997-2001 Texas A&M University College Station, Texas Bachelor of Science, Biomedical Engineering
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes
 Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes Daniel E. Charles, MD, Martin Charles, MD, Gisela Jelusich,
Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes Daniel E. Charles, MD, Martin Charles, MD, Gisela Jelusich,
The Foundation WHAT IS THE RETINA?
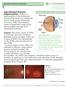 Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
Royal Berkshire Hospital Dunedin Hospital. Prince Charles Eye Unit Pi Princess Margaret Hospital
 Vitreoretinal Surgery Mr Vaughan Tanner www.tanner-eyes.co.uk eyes Reading Royal Berkshire Hospital Dunedin Hospital Windsor Prince Charles Eye Unit Pi Princess Margaret Hospital Success rates VR surgery
Vitreoretinal Surgery Mr Vaughan Tanner www.tanner-eyes.co.uk eyes Reading Royal Berkshire Hospital Dunedin Hospital Windsor Prince Charles Eye Unit Pi Princess Margaret Hospital Success rates VR surgery
Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular Degeneration
 Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis
 Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis G Pron August 2014 Ontario Health Technology Assessment Series; Vol. 14:
Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis G Pron August 2014 Ontario Health Technology Assessment Series; Vol. 14:
Facts About Diabetic Eye Disease
 Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: September 13, 2011 Subject: Page: 1 of 18
 Last Review Status/Date: June 2014 Page: 1 of 18 Description Angiogenesis inhibitors (e.g., ranibizumab, bevacizumab, pegaptanib, aflibercept) are being evaluated for the treatment of disorders of choroidal
Last Review Status/Date: June 2014 Page: 1 of 18 Description Angiogenesis inhibitors (e.g., ranibizumab, bevacizumab, pegaptanib, aflibercept) are being evaluated for the treatment of disorders of choroidal
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.08 Photodynamic Therapy for Choroidal Neovascularization 9.03.20 Intraocular Radiation Therapy for
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.08 Photodynamic Therapy for Choroidal Neovascularization 9.03.20 Intraocular Radiation Therapy for
 measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment
 Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014
 From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit)
 Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit) Line of Business: Medi-Cal Effective Date: May 17, 2017 Revision Date: May 17, 2017 This policy has
Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit) Line of Business: Medi-Cal Effective Date: May 17, 2017 Revision Date: May 17, 2017 This policy has
Long-term Management of AMD. Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan
 Long-term Management of AMD Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan DEFINITION 1 Age-related macular degeneration (AMD) is a disorder of the macula characterized
Long-term Management of AMD Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan DEFINITION 1 Age-related macular degeneration (AMD) is a disorder of the macula characterized
MONTHLY ADMINISTERED INTRAVITREOUS PHARmacotherapy
 Polypoidal Choroidal Vasculopathy Masquerading as Neovascular Age-Related Macular Degeneration Refractory to Ranibizumab ALEXANDROS N. STANGOS, JAGDEEP SINGH GANDHI, JAYASHREE NAIR-SAHNI, HEINRICH HEIMANN,
Polypoidal Choroidal Vasculopathy Masquerading as Neovascular Age-Related Macular Degeneration Refractory to Ranibizumab ALEXANDROS N. STANGOS, JAGDEEP SINGH GANDHI, JAYASHREE NAIR-SAHNI, HEINRICH HEIMANN,
HIGH DOSE TISSUE PLASMINOGEN ACTIVATOR (TPA) (100 MCG/0.1 ML) AND C3F8 GAS IN PNEUMATIC DISPLACEMENT OF SUBMACULAR
 HIGH DOSE TISSUE PLASMINOGEN ACTIVATOR (TPA) (100 MCG/0.1 ML) AND C3F8 GAS IN PNEUMATIC DISPLACEMENT OF SUBMACULAR HAEMORRHAGE. Stephen A.M. De Souza MD, FRCSC Matthew J Welch MD, Raza M Shah MD, Alan
HIGH DOSE TISSUE PLASMINOGEN ACTIVATOR (TPA) (100 MCG/0.1 ML) AND C3F8 GAS IN PNEUMATIC DISPLACEMENT OF SUBMACULAR HAEMORRHAGE. Stephen A.M. De Souza MD, FRCSC Matthew J Welch MD, Raza M Shah MD, Alan
Clinical Policy: Aflibercept (Eylea) Reference Number: CP.PHAR.184
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment
 Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
The Foundation WHAT IS THE RETINA? continued next page. RETINA HEALTH SERIES Facts from the ASRS
 The Foundation American Society of Retina Specialists Committed to improving the quality of life of all people with retinal disease. Intravitreal Injections An intravitreal (pronounced in tra VIT re al)
The Foundation American Society of Retina Specialists Committed to improving the quality of life of all people with retinal disease. Intravitreal Injections An intravitreal (pronounced in tra VIT re al)
Corporate Medical Policy
 Corporate Medical Policy Intravitreal Implant File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravitreal_implant 11/2010 6/2017 6/2018 6/2017 Description of Procedure or Service
Corporate Medical Policy Intravitreal Implant File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravitreal_implant 11/2010 6/2017 6/2018 6/2017 Description of Procedure or Service
Intravitreal Corticosteroid Implants
 Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015
 Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015 So What Parts of the Eye Retina are Affected by VHL Neural tissue
Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015 So What Parts of the Eye Retina are Affected by VHL Neural tissue
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration
 Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements
 Perspective DOI: 10.1167/tvst.4.3.6 Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements Irmela Mantel 1,2 1 Department
Perspective DOI: 10.1167/tvst.4.3.6 Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements Irmela Mantel 1,2 1 Department
Drug Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
A Patient s Guide to Diabetic Retinopathy
 Diabetic Retinopathy A Patient s Guide to Diabetic Retinopathy 840 Walnut Street, Philadelphia PA 19107 www.willseye.org Diabetic Retinopathy 1. Definition Diabetic retinopathy is a complication of diabetes
Diabetic Retinopathy A Patient s Guide to Diabetic Retinopathy 840 Walnut Street, Philadelphia PA 19107 www.willseye.org Diabetic Retinopathy 1. Definition Diabetic retinopathy is a complication of diabetes
Photodynamic Therapy for Treatment of Subfoveal Choroidal Neovascularization in Exudative Age-Related Macular Degeneration
 Clinical Medicine Journal Vol. 4, No. 4, 2018, pp. 60-66 http://www.aiscience.org/journal/cmj ISSN: 2381-7631 (Print); ISSN: 2381-764X (Online) Photodynamic Therapy for Treatment of Subfoveal Choroidal
Clinical Medicine Journal Vol. 4, No. 4, 2018, pp. 60-66 http://www.aiscience.org/journal/cmj ISSN: 2381-7631 (Print); ISSN: 2381-764X (Online) Photodynamic Therapy for Treatment of Subfoveal Choroidal
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY
 EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
A NOVEL COMPOUND FOR TREATMENT OF WET AMD
 A NOVEL COMPOUND FOR TREATMENT OF WET AMD Clinical studies are evaluating a new anti-vegf agent s potential to join current treatment options. BY SARADHA CHEXAL, MD; IVANA GUNDERSON, BS; BRIAN B. BERGER,
A NOVEL COMPOUND FOR TREATMENT OF WET AMD Clinical studies are evaluating a new anti-vegf agent s potential to join current treatment options. BY SARADHA CHEXAL, MD; IVANA GUNDERSON, BS; BRIAN B. BERGER,
Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration
 REVIEW ARTICLE Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration Karen M. Wai, Rishi P. Singh Department of Ophthalmology,
REVIEW ARTICLE Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration Karen M. Wai, Rishi P. Singh Department of Ophthalmology,
Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema
 111 ORIGINAL Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema Toshihiko Nagasawa, Takeshi Naito, Shingo Matsushita, Hiroyuki Sato, Takashi Katome, and
111 ORIGINAL Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema Toshihiko Nagasawa, Takeshi Naito, Shingo Matsushita, Hiroyuki Sato, Takashi Katome, and
These issues are covered in more detail below.
 26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
Corinne GONZALEZ Ophthalmology (MD, Ph.D ) Retinal Medical Specialist
 Corinne GONZALEZ Ophthalmology (MD, Ph.D ) Retinal Medical Specialist PRESENT PROFESSIONAL POSITION OPHTALMOLOGIST DOCTOR, Private practice Retinal medical Specialist, DMLA in particular Installation year
Corinne GONZALEZ Ophthalmology (MD, Ph.D ) Retinal Medical Specialist PRESENT PROFESSIONAL POSITION OPHTALMOLOGIST DOCTOR, Private practice Retinal medical Specialist, DMLA in particular Installation year
Prevention and treatment of agerelated macular degeneration
 THERAPY REVIEW Prevention and treatment of agerelated macular degeneration SARAH HORTON AND CATHERINE GULY Age-related macular degeneration (AMD) is a common cause of visual loss in older people and GPs
THERAPY REVIEW Prevention and treatment of agerelated macular degeneration SARAH HORTON AND CATHERINE GULY Age-related macular degeneration (AMD) is a common cause of visual loss in older people and GPs
EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO
 EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO Yehia Hashad, M.D. Vice President and Global Therapeutic Area Head
EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO Yehia Hashad, M.D. Vice President and Global Therapeutic Area Head
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
NIH Public Access Author Manuscript JAMA Ophthalmol. Author manuscript; available in PMC 2013 September 10.
 NIH Public Access Author Manuscript Published in final edited form as: JAMA Ophthalmol. 2013 May ; 131(5): 693 694. doi:10.1001/jamaophthalmol.2013.692. Effect of Intravitreous Anti Vascular Endothelial
NIH Public Access Author Manuscript Published in final edited form as: JAMA Ophthalmol. 2013 May ; 131(5): 693 694. doi:10.1001/jamaophthalmol.2013.692. Effect of Intravitreous Anti Vascular Endothelial
ANTIVASCULAR ENDOTHELIAL GROWTH FACTOR
 THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY
MEDICAL POLICY SUBJECT: TRANSPUPILLARY THERMOTHERAPY. POLICY NUMBER: CATEGORY: Technology Assessment
 MEDICAL POLICY SUBJECT: TRANSPUPILLARY EDITED DATE: 08/20/15, 08/18/16, 08/17/17 PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply.
MEDICAL POLICY SUBJECT: TRANSPUPILLARY EDITED DATE: 08/20/15, 08/18/16, 08/17/17 PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply.
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
EPIRETINAL MEMBRANES IN NEOVASCULAR AGE-RELATED MACULAR DEGENERATION. Effect on Outcomes of Anti-vascular Endothelial Growth Factor Therapy
 EPIRETINAL MEMBRANES IN NEOVASCULAR AGE-RELATED MACULAR DEGENERATION Effect on Outcomes of Anti-vascular Endothelial Growth Factor Therapy EMINE ESRA KARACA, MD,* BURÇIN KEPEZ YILDIZ, MD, MEHMET ÖZGÜR
EPIRETINAL MEMBRANES IN NEOVASCULAR AGE-RELATED MACULAR DEGENERATION Effect on Outcomes of Anti-vascular Endothelial Growth Factor Therapy EMINE ESRA KARACA, MD,* BURÇIN KEPEZ YILDIZ, MD, MEHMET ÖZGÜR
Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related macular degeneration.
 Thomas Jefferson University Jefferson Digital Commons Wills Eye Institute Papers Wills Eye Institute 4-1-2008 Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related
Thomas Jefferson University Jefferson Digital Commons Wills Eye Institute Papers Wills Eye Institute 4-1-2008 Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related
Can intravitreal tissue plasminogen activator and SF6 gas facilitate management of macular degeneration with photodynamic therapy?
 European Journal of Ophthalmology / Vol. 18 no. 4, 2008 / pp. 591-594 Can intravitreal tissue plasminogen activator and SF6 gas facilitate management of macular degeneration with photodynamic therapy?
European Journal of Ophthalmology / Vol. 18 no. 4, 2008 / pp. 591-594 Can intravitreal tissue plasminogen activator and SF6 gas facilitate management of macular degeneration with photodynamic therapy?
Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical setting
 Korb et al. BMC Ophthalmology 2013, 13:84 RESEARCH ARTICLE Open Access Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical
Korb et al. BMC Ophthalmology 2013, 13:84 RESEARCH ARTICLE Open Access Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Retina Conference. Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014
 Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
Acknowledgements. Outline. Who were von Hippel and Lindau? Eugen von Hippel German Ophthalmologist
 Ophthalmic Therapies & Standard of Care Acknowledgements Eric Jonasch, MD & Surena Matin, MD Collaborators Franco DeMonte, MD Marcy Johnson Ian McCutcheon, MD Chaan Ng, MD Nancy Perrier, MD Dawid Schellingerhout,
Ophthalmic Therapies & Standard of Care Acknowledgements Eric Jonasch, MD & Surena Matin, MD Collaborators Franco DeMonte, MD Marcy Johnson Ian McCutcheon, MD Chaan Ng, MD Nancy Perrier, MD Dawid Schellingerhout,
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE)
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA68; The clinical and cost effectiveness of photodynamic therapy for age-related macular degeneration TA68 guidance
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA68; The clinical and cost effectiveness of photodynamic therapy for age-related macular degeneration TA68 guidance
Navilas Clinical Results: Diabetic Macular Edema Introducing a Better Treatment Paradigm for Patients and Physicians
 Navilas Clinical Results: Diabetic Macular Edema Introducing a Better Treatment Paradigm for Patients and Physicians 16 % Injection-free after loading phase 65 % Injection-free after loading phase Navilas
Navilas Clinical Results: Diabetic Macular Edema Introducing a Better Treatment Paradigm for Patients and Physicians 16 % Injection-free after loading phase 65 % Injection-free after loading phase Navilas
Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant
 Ophthalmol Ther (2015) 4:51 58 DOI 10.1007/s40123-015-0028-0 CASE REPORT Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant Thomas Bertelmann
Ophthalmol Ther (2015) 4:51 58 DOI 10.1007/s40123-015-0028-0 CASE REPORT Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant Thomas Bertelmann
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Dan Roberts. International Low Vision Support Group NEWSLETTER. This Month. In This Issue. 1: This Month 2-6: News
 In This Issue 1: This Month 2-6: News Eye & Body Complete from Biosyntrx Extra Strength Protection Call for information 800-688-6815 The ILVSG is sponsored by MD Foundation www.eyesight.org 888-633-3937
In This Issue 1: This Month 2-6: News Eye & Body Complete from Biosyntrx Extra Strength Protection Call for information 800-688-6815 The ILVSG is sponsored by MD Foundation www.eyesight.org 888-633-3937
The limited number of currently approved
 CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
Despite our growing knowledge of the
 OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
Digital Journal of Ophthalmology Digital Journal of Ophthalmology. Abstract
 Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
Diabetic Retinopathy A Presentation for the Public
 Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Introduction How the eye works
 1 Introduction Diabetic retinopathy is a condition that can cause permanent loss of eyesight and even blindness. It is a major cause of loss of vision. But if a person with diabetes receives proper eye
1 Introduction Diabetic retinopathy is a condition that can cause permanent loss of eyesight and even blindness. It is a major cause of loss of vision. But if a person with diabetes receives proper eye
Brampton Hurontario Street Brampton, ON L6Y 0P6
 Diabetic Retinopathy What is Diabetic Retinopathy Diabetic retinopathy is one of the leading causes of blindness world-wide. Diabetes damages blood vessels in many organs of the body including the eyes.
Diabetic Retinopathy What is Diabetic Retinopathy Diabetic retinopathy is one of the leading causes of blindness world-wide. Diabetes damages blood vessels in many organs of the body including the eyes.
Update 1. Red Eye. Adenovirus Numbers 3/4/14. For Your Eyes Only 2014 Ophthalmology Update. TKorn, MD, FACS San Diego, California
 For Your Eyes Only 2014 Ophthalmology Update TKorn, MD, FACS San Diego, California Update 1 Red Eye Adenovirus Numbers 15.7 - Adults touch their face in an hour 1-5 weeks - Adenovirus infectivity period
For Your Eyes Only 2014 Ophthalmology Update TKorn, MD, FACS San Diego, California Update 1 Red Eye Adenovirus Numbers 15.7 - Adults touch their face in an hour 1-5 weeks - Adenovirus infectivity period
Update 1. Red Eye. Adenovirus Numbers 7/29/14. Adenovirus Conjunctivitis Outbreaks. Adenovirus Conjunctivitis Management
 For Your Eyes Only 2014 Ophthalmology Update Update 1 Red Eye TKorn, MD, FACS San Diego, California Adenovirus Numbers Adenovirus Conjunctivitis Outbreaks 15.7 - Adults touch their face in an hour 1-5
For Your Eyes Only 2014 Ophthalmology Update Update 1 Red Eye TKorn, MD, FACS San Diego, California Adenovirus Numbers Adenovirus Conjunctivitis Outbreaks 15.7 - Adults touch their face in an hour 1-5
Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment
 9:30 AM Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment Lee Jampol, MD OBJECTIVE To assess subsequent visual and anatomic outcomes of eyes with
9:30 AM Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment Lee Jampol, MD OBJECTIVE To assess subsequent visual and anatomic outcomes of eyes with
