Pharmacodynamic Effects of Ghrelin Agonist Relamorelin (RM-131) in Patients with Type 1 and Type 2 Diabetes Mellitus and Delayed Gastric Emptying
|
|
|
- Christiana Curtis
- 5 years ago
- Views:
Transcription
1 Pharmacodynamic Effects of Ghrelin Agonist Relamorelin (RM-131) in Patients with Type 1 and Type 2 Diabetes Mellitus and Delayed Gastric Emptying Andrea Shin Motility Conference 2/4/15
2 Disclosures No conflicts of interest Supported by the NIH CTSA grant UL1 TR000135, and a research grant from Rhythm Pharmaceuticals, Boston, MA
3 Outline Background Clinical Symptoms Diagnostic Assessment Pharmacologic Therapies Aims Findings and Results Summary and Future Directions
4 Diabetic Gastroparesis Upper GI symptoms and delayed gastric emptying (GE) Nausea, vomiting, early satiety (fullness), bloating, pain Asymptomatic (delayed GE) Symptoms in 5-12% patients with diabetes 1,2 Poorer glycemic control Anxiety, depression, and neuroticism 3 More likely to have cardiovascular disease, nephropathy, hypertension, retinopathy 4 1. Bytzer P et al. Arch Intern Med Maleki D et al. Arch Intern Med Talley NJ et al. Am J Gastroenterol Hyett B et al. Gastroenterology 2009
5 Diabetic Gastroparesis is a common cause of gastroparesis among tertiary referral patients Parkinson s Disease 10% Other 3% Postsurgical 14% Diabetic 29% Intestinal Pseudoobstruction 4% Scleroderma 4% Idiopathic 36% Bityutskiy et al. Am J Gastroenterol 1997
6 Comparison of techniques for GE assessment Indication / function measured Device, assembly or special requirements Scintigraphy Stable isotope breath test Gastric emptying External gamma camera; isotope-labeled meal Wireless pressure and ph capsule Gastric emptying Emptying and pressure amplitude Breath collection vials; stable isotope-labeled meal Intraluminal capsule with miniaturized strain gauge and ph measurement Ultrasonography Gastric emptying 2D or 3D ultrasound equipment Placement of device - - Capsule swallowed On abdomen repeatedly Performance / versatility / interpretation Duration of study (hours, h) Excellent; standardized meals, data acquisition and interpretation Typically 4h, could be added to small bowel and colon transit Becoming standardized; performance related to mathematics analysis Standard acquisition; delayed emptying fairly valid; pressures of unclear significance 3-4h 6h, could be added to small bowel and colon transit Availability / potential use Cost Becoming standardized; performance related to technical expertise; best for liquid emptying Typically 2h Szarka LA, Camilleri M. Am J Physiol 2009
7 Gastric Emptying Scintigraphy (GES) Gold standard for GE assessment Society of Nuclear Medicine & The American Neurogastroenterology and Motility Society Performed with standard low-fat meal Solid-phase GE to document delayed GE Simultaneous assessment of liquid GE May sensitivity? Relationship between solid and liquid GE unclear
8 Indications for GES Diabetic patients with upper GI symptoms Poor glycemic control Considering or are taking hypoglycemic medications that may slow GE Severe reflux symptoms
9 GES Preparation Stop all motility-altering medications for 2-3 days (prokinetics, opiates, anticholinergics) No smoking/alcohol consumption on test day Fasting blood glucose < 275 mg/dl on test day** What level of hyperglycemia is important? Bytzer et al. Am J Gastroenterol 2002 Bharucha et al. Clin Endocrinol (Oxf) Hasler WL et al. Gastro 1995 Bharucha et al. Clin Gastroenterol Hepatol 2014
10 GES Procedure Procedure: Overnight fast Standardized test meal within 10 minutes (255kcal) Imaging at baseline, 1, 2, 4 hours after meal ingestion Minimum of 4 hours for reliable estimate of T 1/2 0.5mCi 99mTc
11 Normal and delayed GE in patient with type 1 DM 0hr 2hr 4hr Normal GE GE=67% Delayed GE GE=29% Quantification of GE using computerized software Results are expressed as % radioactivity retained in the stomach at each time point Delayed GE if: > 60% retention at 2h or > 10% retention at 4 hours Females on average 15% slower than males GE=100% GE=57%
12 Merits & Limitations of GES Non-invasive Direct measure of GE Quantitative assessment Radiation exposure Limited access to gamma-camera Lack of adherence to standardized protocol Assess GE both solids and liquids Significant intra-individual CV (24%)? Characterize intragastric distribution of contents Limitations of low-fat, low-fiber meal
13 Treatment for Gastroparesis First line therapy: Nutrition, hydration, glycemic control Metoclopramide Risk of neurological side effects (tardive dyskinesia) Limited to no more than 3 consecutive months Domperidone Erythromycin tachyphylaxis Symptomatic treatment anti-emetics, pain management Surgery and/or Botox
14 Ghrelin Camilleri M et al. Nat Rev Gastroenterol Hepatol 2009
15 Promotes gastric motility in animal models The role of Ghrelin Ghrelin is a potential treatment for delayed gastric emptying (DGE) Short half-life, plasma instability Camilleri M et al. Nat Rev Gastroenterol Hepatol 2009
16 Synthetic Ghrelin Agonists TZP-101 (ulimorelin) Macrocyclic peptidomimetic Potent binding affinity for the ghrelin receptor Accelerated GE Ejskaer N et al. Aliment Pharmacol Ther 2009
17 Change in mean Nausea/Vomiting subscale scores (a) and Vomiting scores (b) over time. Wo et al. Aliment Pharmacol Ther 2010
18 A phase 2a, DB, RCT 28-day study of TZP-102, a ghrelin receptor agonist for diabetic gastroparesis Background: TZP-102 (macrocyclic, selective, oral ghrelin-r agonist) Methods DB, RCT of 92 outpatients with diabetic gastroparesis; oncedaily 10-mg (n = 22), 20-mg (n = 21), 40-mg (n = 23) TZP-102 or placebo (n = 26). The primary endpoint was the change in GE T ½ utilizing 13 C- Octanoate breath test (350 kcal, 7g fat meal) Conclusion: TZP-102 for 28 days, at doses of 10-40mg once daily, does not accelerate gastric emptying but it is was well-tolerated and resulted in a reduction in symptoms of gastroparesis Ejskaer N et al. Neurogastroenterol Motil 2013
19 Oral TZP-102 in Diabetic Gastroparesis Aim: Two phase 2b RCTs (TZP-102-CL-G003 and TZP-102-CL-G004) to evaluate 12 weeks of oral TZP-102 in patients with diabetic gastroparesis Primary outcome: Average change from baseline through end-of treatment in Daily Diary of Gastroparesis Symptoms Questionnaire (GSDD) Results: Improvement in the GSDD observed in all treatment arms GSDD Composite score 10 mg TZP mg TZP mg TZP mg TZP mg TZP mg TZP-102 Placebo Placebo Placebo BL Week 12 Δ from BL BL Week 12 Δ from BL Baseline Week 12 Δ from BL 3.5± ± ±1.2 P= ± ± ±1.3 P= ± ± ±1.2 McCallum RW et al. Neurogastroenterol Motil 2013
20 Novel ghrelin agonist, RM-131 RM-131 (Relamorelin) Pentapeptide synthetic ghrelin agonist Longer plasma T 1/2 >100-fold potency for prokinetic effects than native ghrelin in animal models PK and PD data from healthy volunteer studies Single-ascending dose study of 36 healthy males Mean T 1/2 for elimination 5-19 hours Acceleration of GE at doses 10 μg Maximal effect at 100 μg dose level
21 Randomized Controlled Phase Ib Study of Ghrelin Agonist, RM-131, in Type 2 Diabetic Women with Delayed Gastric Emptying: Pharmacokinetics and Pharmacodynamics Shin A, Camilleri M, Busciglio I, Burton D, Stoner E, Noonan P, Gottesdiener K, Smith SA, Vella A, Zinsmeister AR
22 Objectives Primary objective: To investigate the PD profile of a single dose of RM-131 in type 2 diabetes mellitus (T2DM) patients with gastrointestinal cardinal symptoms (GCSI) and prior documentation of DGE Secondary objective: To evaluate symptoms and safety of a single dose of RM-131 in T2DM patients with GCSI and prior documentation of DGE
23 Methods Study Design: Randomized, double-blind, placebocontrolled, single-dose, two-period, crossover study Main eligibility criteria: T2DM with (a) documented DGE by scintigraphy or gastric emptying breath test and (b) >3 months history of symptoms of gastroparesis Ages 18 to 60 years Controlled T2DM (HbA1c <8.5%) Stable concomitant medications Prior exclusion of upper GI mechanical obstruction BMI kg/m 2 PD profile, safety, and symptoms were assessed in both periods
24 Methods 100 µg s.c. injection (RM or placebo) 100 µg s.c. injection (RM or placebo) D1 D2 7 day washout D1 D2 Period 1 Period 2 GE (gastric emptying solids and liquids) CF6 (colonic filling % at 6h) Hormonal levels, safety, pharmacokinetic (PK) samples Symptoms Validated scintigraphy was used to assess GE and CF6 after a standardized meal 255 kcal meal (72% carbohydrate, 24% protein, 2% fat, and 2% fiber) given 30 min post-dosing
25 Patient Characteristics All 10 patients in the study were female Mean values (+SEM) at study entry: Age (years): 51.8 (+2.5) BMI (kg/m 2 ): 31.1 (+1.8) HbA1c (%): 7.2 (+0.4) Total GCSI-DD score: 1.32 (+0.2)
26 A single dose of RM-131 Decreases GE T 1/2 by 66% GE, min GE Sol GE Liq t 1/2 Lag time t 1/2 t 1/2 p=0.01 p<0.10 placebo RM p=0.14 p=0.11 CF6 Effect of RM-131 on gastric emptying (solids and liquids) and colonic filling at 6 hours in all 10 patients CF6, %
27 Accelerated GE T 1/2 in 9 of 10 Patients 250 Patient Patient 2 Patient 3 GE T 1/ Patient 4 Patient 5 Patient 6 Patient 7 Patient 8 Patient 9 Patient 10 Mean value 0 Placebo Treatment RM-131
28 Order Effect Effect of RM-131 on solid GE T 1/2 and CF6 larger when participant received RM-131 first Supportive analysis of Period 1 alone Period 1 showed significant drug effects for GE T 1/2 and CF6. Estimated in solid GE T 1/2 was 43 min (95%CI 10-75) or 61%.
29 RM-131 Modulates All Gastric and Small Bowel Transit Parameters in Period p=0.02 p=0.016 Period 1 data placebo, n=5 RM -131,- n=5 p= p= GE, min p=0.024 p= CF6 % 20 p= t 1/2 GE Sol Lag time GE Liq t 1/2 CF6 0
30 Glycemic and Hormonal Effects Glucose & Insulin: Higher 120 minute blood glucose (p=0.07) with RM-131 No significant effects on insulin Hormonal Effects: Baseline hormone levels were not different on the two treatment days Expected acute post-dose increases in min AUC in GH, cortisol and prolactin levels with RM-131 were observed (all p<0.02)
31 Symptoms and Safety Symptoms: Single dose study, not designed nor powered to assess symptoms No significant effects (p>0.5) on total GCSI-DD or composite score of nausea, bloating, postprandial fullness, and pain Safety: RM-131 was generally well tolerated Total number of adverse events (AEs) (p=0.016) higher with RM-131, but none were serious Light-headedness reported more often on RM-131 All AEs resolved spontaneously
32 Conclusions RM-131 greatly accelerates gastric emptying in patients with T2DM and delayed gastric emptying Overall, a 66% decrease in gastric emptying half time was observed Greatest improvement was observed in those with most abnormal gastric emptying Further clinical investigation of this promising and novel pharmacologic agent in the treatment of diabetic gastroparesis is needed
33 Ghrelin Agonist RM-131 Accelerates Gastric Emptying of Solids and Reduces Symptoms in Type 1 Diabetics: A Randomized Trial Shin A, Camilleri M, Busciglio I, Burton D, Smith SA, Vella A, Ryks M, Rhoten D, Zinsmeister AR
34 Objective To investigate the PD profile and effects on upper GI symptoms, safety and tolerability of a single dose of RM-131 in patients with type 1 diabetes mellitus (T1DM) and prior documentation of DGE
35 Methods Study Design: Randomized, double-blind, placebocontrolled, single-dose, two-period, crossover study Eligibility criteria: Males & females ages years T1DM with (a) documented DGE by scintigraphy or gastric emptying breath test and (b) >3 months history of symptoms of gastroparesis HbA1c <10.1 % Prior exclusion of upper GI mechanical obstruction BMI kg/m 2 Medical records reviewed, baseline ECG obtained Enrolled and randomized by a computer-generated allocation schedule
36 Study Procedures 100 µg s.c. injection (RM or placebo) 100 µg s.c. injection (RM or placebo) D1 D2 7 day washout D1 D2 Period 1 Period 2 GE (gastric emptying solids and liquids) CF6 (colonic filling % at 6h) Symptoms
37 Patient Characteristics All 10 patients completed the study (2M, 8F) Mean values (+SEM) at screening: HbA1c: % Age: y BMI: kg/m 2 Total GCSI-DD score: (median 1.71) Total NVFP score: Absence of sinus arrhythmia observed in 6/10 patients, indicating the presence of cardiovagal dysfunction
38 Summary of the effects of RM-131 on GE for solids and liquids = -54.7% placebo RM-131 Time, minutes All p=ns = -36.4% = -12.6% 0 GE T 1/2 solid GE lag solid GE T 1/2 liquid CF6
39 Effect of RM-131 on %GE at 1, 2, 4 hours and CF6 120 p<0.05 p=ns p=ns 100 p<0.01 GE, % emptied = 72.3% CF6, % 20 = 0% 0 = 60% = 19.4% GE 1h GE 2h GE 4h CF6
40 Symptoms and Blood Glucose Data show median (IQR) Placebo RM-131 P value % Difference Total GCSI-DD average score # (0.75,2.08) (0.00, 0.67) Average score of combined nausea, vomiting, postprandial fullness, upper abdominal pain 1.00 (0.50, 2.00) 0.25 (0.00,0.50) 0.041# Blood glucose at 120 min, mg/dl 248 (182,273) 231 (152,290) ns Median % difference among all participants for RM-131 minus placebo (within patient) relative to overall means (within patient); 100X [(within subject delta) / (within subject mean)] Data compared using Wilcoxon signed rank test or *paired t-test and #paired t test with Hochberg step-up correction; ns=not significant IQR=interquartile range
41 Placebo (N) Safety RM-131 (N) Severity as Described by Participant Relation to Study Medication Any adverse event possible Hyperhidrosis 0 2 moderate possible Fatigue 1 1 mild to moderate possible Abdominal pain 1 0 severe unlikely Irritation at injection site 0 1 mild likely Hunger* 0 5 mild to moderate possible Shakiness 0 1 moderate possible Euphoria 1 0 moderate unlikely Hyperglycemia 0 2 moderate to severe possible Hypoglycemia 1 0 mild possible Burning in feet 1 0 moderate unlikely Flank pain 1 0 mild possible Abdominal pressure 0 1 mild possible Flatulence 1 0 mild possible Borborygmi 1 0 mild possible *p=0.0625; all other p=ns (comparisons performed using McNemar s Test)
42 Conclusions Improvement in GE T 1/2 solid, GE 1h, and GE 2h in T1DM with RM-131 Comparable to the 66.1% in T2DM Significant improvement in total GCSI-DD and NVFP scores Appears effective in patients with cardiovascular neuropathy Further study of medium/long-term efficacy
43 Lembo et al. DDW 2014 Phase 2 RCT to investigate safety and efficacy of RM-131 in patients with diabetic gastroparesis Design: 1 week single-blind pcbo run-in followed by randomization to pcbo vs. RM-131 (10 ug SC BID or 10 ug SC QD). GE breath test at baseline and at 28 days Daily symptom diary (nausea, pain, bloating, earlying satiety) Results: 204 patients randomized (32.3%M, mean age 55.1 y, mean BMI 32.6 kg/m2, 11.9% Type 1 DM) Relamorelin (10 μg BID), resulted in significant acceleration of gastric emptying (p < 0.03) Significant improvements in vomiting endpoints on relamorelin treatment compared to placebo
44 Future Directions Larger sample sizes Evaluate medium to long-term effects and safety Efficacy among patients with moderate to severe gastroparesis Other conditions such as idiopathic gastroparesis, post-surgical or post-vagotomy gastroparesis, post-operative ileus, or chronic constipation
45 Acknowledgements Dr. Michael Camilleri Division of Endocrinology, Metabolism and Diabetes: Drs. Adrian Vella and Steven Smith Biomedical Statistics and Informatics: Dr. Alan Zinsmeister Study coordinators & technicians: Duane Burton, Irene Busciglio, Michael Ryks, Deb Rhoten Rhythm Pharmaceuticals
Gastroparesis: Diagnosis and Management
 Gastroparesis: Diagnosis and Management Rodica Pop-Busui MD, PhD Professor of Internal Medicine, Metabolism, Endocrinology and Diabetes, University of Michigan, Ann Arbor, MI Disclosures Astra Zeneca Research
Gastroparesis: Diagnosis and Management Rodica Pop-Busui MD, PhD Professor of Internal Medicine, Metabolism, Endocrinology and Diabetes, University of Michigan, Ann Arbor, MI Disclosures Astra Zeneca Research
Management of Gastroparesis
 Management of Gastroparesis Bible Class Jan Hendrik Niess As published in Am J Gastroenterol. 2013 Jan;108(1):18-37 What is the definition of gastroparesis? What are cardinal symptoms of gastroparesis?
Management of Gastroparesis Bible Class Jan Hendrik Niess As published in Am J Gastroenterol. 2013 Jan;108(1):18-37 What is the definition of gastroparesis? What are cardinal symptoms of gastroparesis?
DIABETIC GASTROPARESIS OBESE VERSUS NON OBESE PATIENTS
 Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 4 (53) No. 2-2011 DIABETIC GASTROPARESIS OBESE VERSUS NON OBESE PATIENTS L. POANTA 1 D. L. DUMITRAŞCU 1 Abstract: Symptoms
Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 4 (53) No. 2-2011 DIABETIC GASTROPARESIS OBESE VERSUS NON OBESE PATIENTS L. POANTA 1 D. L. DUMITRAŞCU 1 Abstract: Symptoms
Gastroparesis. - Recent advances in the pathophysiology and treatment -
 ICDM 2015 Gastroparesis - Recent advances in the pathophysiology and treatment - Department of Internal Medicine, College of Medicine, St. Paul s hospital, The Catholic University of Korea, Seoul, Korea
ICDM 2015 Gastroparesis - Recent advances in the pathophysiology and treatment - Department of Internal Medicine, College of Medicine, St. Paul s hospital, The Catholic University of Korea, Seoul, Korea
GASTROPARESIS. C. Prakash Gyawali, MD Professor of Medicine Washington University in St. Louis
 GASTROPARESIS C. Prakash Gyawali, MD Professor of Medicine Washington University in St. Louis Symptom Definitions Nausea: a subjective feeling of wanting to vomit Vomiting: forceful expulsion of gastroduodenal
GASTROPARESIS C. Prakash Gyawali, MD Professor of Medicine Washington University in St. Louis Symptom Definitions Nausea: a subjective feeling of wanting to vomit Vomiting: forceful expulsion of gastroduodenal
Outline. Definition (s) Epidemiology Pathophysiology Management With an emphasis on recent developments
 Chronic Dyspepsia Eamonn M M Quigley MD FRCP FACP MACG FRCPI Lynda K and David M Underwood Center for Digestive Disorders Houston Methodist Hospital Houston, Texas Outline Definition (s) Epidemiology Pathophysiology
Chronic Dyspepsia Eamonn M M Quigley MD FRCP FACP MACG FRCPI Lynda K and David M Underwood Center for Digestive Disorders Houston Methodist Hospital Houston, Texas Outline Definition (s) Epidemiology Pathophysiology
Is a field of radiology that uses unsealed radiation for diagnostic or therapeutic reasons Is a method of physiologic imaging, very different than
 Jeremy Flowers DNP Is a field of radiology that uses unsealed radiation for diagnostic or therapeutic reasons Is a method of physiologic imaging, very different than x-ray or CT that is anatomic imaging
Jeremy Flowers DNP Is a field of radiology that uses unsealed radiation for diagnostic or therapeutic reasons Is a method of physiologic imaging, very different than x-ray or CT that is anatomic imaging
Dr. Patsy Smyth, FNP-BC
 Dr. Patsy Smyth, FNP-BC Gastroparesis literally translated means stomach paralysis. Gastroparesis is a syndrome characterized by delayed gastric emptying in absence of mechanical obstruction of the stomach.
Dr. Patsy Smyth, FNP-BC Gastroparesis literally translated means stomach paralysis. Gastroparesis is a syndrome characterized by delayed gastric emptying in absence of mechanical obstruction of the stomach.
Our evidence. Your expertise. SmartPill : The data you need to evaluate motility disorders.
 Our evidence. Your expertise. SmartPill : The data you need to evaluate motility disorders. SmartPill benefits your practice: Convenient performed right in your office Test standardization Provides direct
Our evidence. Your expertise. SmartPill : The data you need to evaluate motility disorders. SmartPill benefits your practice: Convenient performed right in your office Test standardization Provides direct
Motility Conference Ghrelin
 Motility Conference Ghrelin Emori Bizer, M.D. Division of Gastroenterology/Hepatology November 21, 2007 Ghrelin: Basics Hormone produced by the A-like A endocrine cells in the oxyntic mucosa (stomach body
Motility Conference Ghrelin Emori Bizer, M.D. Division of Gastroenterology/Hepatology November 21, 2007 Ghrelin: Basics Hormone produced by the A-like A endocrine cells in the oxyntic mucosa (stomach body
Gut involvement in PoTS an overview
 Gut involvement in PoTS an overview Qasim Aziz, PhD, FRCP Centre for Neuroscience and Trauma Wingate Institute of Neurogastroenterology Case Hx * 28 year old lady presents with a long hx of constipation
Gut involvement in PoTS an overview Qasim Aziz, PhD, FRCP Centre for Neuroscience and Trauma Wingate Institute of Neurogastroenterology Case Hx * 28 year old lady presents with a long hx of constipation
Number of studies. Endoscopic finding. Number of subjects. Pooled prevalence 95% CI
 Clinical Approach to the Patient t with Dyspepsia William D. Chey, MD, FACG Professor of Medicine University of Michigan Prevalence of Endoscopic Findings in Individuals with Dyspepsia Systematic Review
Clinical Approach to the Patient t with Dyspepsia William D. Chey, MD, FACG Professor of Medicine University of Michigan Prevalence of Endoscopic Findings in Individuals with Dyspepsia Systematic Review
Functional Dyspepsia
 Functional Dyspepsia American College of Gastroenterology Boston Massachusetts, June 2015 Brian E. Lacy, PhD, MD, FACG Professor of Medicine Geisel School of Medicine at Dartmouth Chief, Section of Gastroenterology
Functional Dyspepsia American College of Gastroenterology Boston Massachusetts, June 2015 Brian E. Lacy, PhD, MD, FACG Professor of Medicine Geisel School of Medicine at Dartmouth Chief, Section of Gastroenterology
Does the Injection of Botulinum Toxin Improve Symptoms in Patients With Gastroparesis?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Does the Injection of Botulinum Toxin
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Does the Injection of Botulinum Toxin
ACG Clinical Guideline: Management of Gastroparesis
 ACG Clinical Guideline: Management of Gastroparesis Michael Camilleri, MD 1, Henry P. Parkman, MD 2, Mehnaz A. Shafi, MD 3, Thomas L. Abell, MD 4 and Lauren Gerson, MD, MSc 5 1 Department of Gastroenterology,
ACG Clinical Guideline: Management of Gastroparesis Michael Camilleri, MD 1, Henry P. Parkman, MD 2, Mehnaz A. Shafi, MD 3, Thomas L. Abell, MD 4 and Lauren Gerson, MD, MSc 5 1 Department of Gastroenterology,
Gut complications in autonomic dysfunction Qasim Aziz, PhD, FRCP
 Gut complications in autonomic dysfunction Qasim Aziz, PhD, FRCP Centre for Neuroscience and Trauma Wingate Institute of Neurogastroenterology GI involvement in autonomic dysfunction Conditions Diabetes
Gut complications in autonomic dysfunction Qasim Aziz, PhD, FRCP Centre for Neuroscience and Trauma Wingate Institute of Neurogastroenterology GI involvement in autonomic dysfunction Conditions Diabetes
Ingestible ph and Pressure Capsule
 Medical Policy Manual Medicine, Policy No. 117 Ingestible ph and Pressure Capsule Next Review: October 2018 Last Review: October 2017 Effective: December 1, 2017 IMPORTANT REMINDER Medical Policies are
Medical Policy Manual Medicine, Policy No. 117 Ingestible ph and Pressure Capsule Next Review: October 2018 Last Review: October 2017 Effective: December 1, 2017 IMPORTANT REMINDER Medical Policies are
Public Statement: Medical Policy
 ARBenefits Approval: 10/12/11 Medical Policy Title: Gastric Neurostimulation Effective Date: 01/01/2012 Document: ARB0166 Revision Date: Code(s): 0155T Laparoscopy, surgical; implantation or replacement
ARBenefits Approval: 10/12/11 Medical Policy Title: Gastric Neurostimulation Effective Date: 01/01/2012 Document: ARB0166 Revision Date: Code(s): 0155T Laparoscopy, surgical; implantation or replacement
Medical Policy. MP Ingestible ph and Pressure Capsule
 Medical Policy BCBSA Ref. Policy: 2.01.81 Last Review: 11/15/2018 Effective Date: 11/15/2018 Section: Medicine Related Policies 2.01.20 Esophageal ph Monitoring 6.01.33 Wireless Capsule Endoscopy as a
Medical Policy BCBSA Ref. Policy: 2.01.81 Last Review: 11/15/2018 Effective Date: 11/15/2018 Section: Medicine Related Policies 2.01.20 Esophageal ph Monitoring 6.01.33 Wireless Capsule Endoscopy as a
Ingestible ph and Pressure Capsule
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Gender aspects suggestive of gastroparesis in patients with diabetes mellitus: a cross-sectional survey
 Dickman et al. BMC Gastroenterology 2014, 14:34 RESEARCH ARTICLE Open Access Gender aspects suggestive of gastroparesis in patients with diabetes mellitus: a cross-sectional survey Ram Dickman 1,5*, Julio
Dickman et al. BMC Gastroenterology 2014, 14:34 RESEARCH ARTICLE Open Access Gender aspects suggestive of gastroparesis in patients with diabetes mellitus: a cross-sectional survey Ram Dickman 1,5*, Julio
The Opportunity: c-ibs and pain relief with confidence YKP10811
 The Opportunity: c-ibs and pain relief with confidence YKP10811 1 TABLE OF CONTENTS Profile Summary Clinical Data Mode of Action Pharmacologic Profile Safety and Toxicity Profile ADME Overview vs. Competitors
The Opportunity: c-ibs and pain relief with confidence YKP10811 1 TABLE OF CONTENTS Profile Summary Clinical Data Mode of Action Pharmacologic Profile Safety and Toxicity Profile ADME Overview vs. Competitors
Gastroparesis: A chronic disorder
 Diagnosis and Management of Gastroparesis: The Stomach that Refuses to Empty Improving Symptoms or Gastric Emptying Henry P. Parkman, MD Professor of Medicine Director of GI Motility Laboratory Gastroenterology
Diagnosis and Management of Gastroparesis: The Stomach that Refuses to Empty Improving Symptoms or Gastric Emptying Henry P. Parkman, MD Professor of Medicine Director of GI Motility Laboratory Gastroenterology
Disclosures. Objectives. Gastroparesis: Are We Moving On? Gastroparesis: Clinical Overview
 Gastroparesis: Are We Moving On? Anthony Lembo, M.D. Associate Professor of Medicine Harvard Medical School Beth Israel Deaconess Medical Center Boston, MA Disclosures Consultant Alkermes, Allergan, Forest,
Gastroparesis: Are We Moving On? Anthony Lembo, M.D. Associate Professor of Medicine Harvard Medical School Beth Israel Deaconess Medical Center Boston, MA Disclosures Consultant Alkermes, Allergan, Forest,
Gastroparesis and other upper GI problems DR ANDREW DAVIES
 Gastroparesis and other upper GI problems DR ANDREW DAVIES Outline Gastroparesis Hiccoughs Gastroparesis Gastroparesis Definition: A syndrome of objectively delayed gastric emptying in the absence of mechanical
Gastroparesis and other upper GI problems DR ANDREW DAVIES Outline Gastroparesis Hiccoughs Gastroparesis Gastroparesis Definition: A syndrome of objectively delayed gastric emptying in the absence of mechanical
Diabetic Gastroparesis. Evan M. Klass, MD, FACP February 16, 2017
 Diabetic Gastroparesis Evan M. Klass, MD, FACP February 16, 2017 Scope of the problem The disorder can affect any part of the GI tract Although 10-20% of the general population suffer from functional GI
Diabetic Gastroparesis Evan M. Klass, MD, FACP February 16, 2017 Scope of the problem The disorder can affect any part of the GI tract Although 10-20% of the general population suffer from functional GI
GASTRIC EMPTYING STUDY (SOLID)
 GASTRIC EMPTYING STUDY (SOLID) Aim To evaluate patients with symptoms of altered of gastric emptying and/or motility, and quantitatively measure the rate of gastric emptying. This study provides a physiologic,
GASTRIC EMPTYING STUDY (SOLID) Aim To evaluate patients with symptoms of altered of gastric emptying and/or motility, and quantitatively measure the rate of gastric emptying. This study provides a physiologic,
Kitti Chuenyong 1, Attaporn Pornananrat 1, Veerasak Sarinnapakorn 2, Pichit Kuanrakcharoen 3
 RESEARCH ARTICLE Early Detection of Diabetic Gastroparesis in Type 2 Diabetes Using Diabetic Peripheral Neuropathy as a Surrogate Marker: A Pilot Study in Rajavithi Hospital Kitti Chuenyong 1, Attaporn
RESEARCH ARTICLE Early Detection of Diabetic Gastroparesis in Type 2 Diabetes Using Diabetic Peripheral Neuropathy as a Surrogate Marker: A Pilot Study in Rajavithi Hospital Kitti Chuenyong 1, Attaporn
Introduction. The oxytocin/ / vasopressin receptor antagonist, atosiban, delays the gastric emptying of a semisolid meal compared to saline in human
 The oxytocin/ / vasopressin receptor antagonist, atosiban, delays the gastric emptying of a semisolid meal compared to saline in human Bodil Ohlsson, et. al., Sweden BMC Gastroenterology, 2006 Presented
The oxytocin/ / vasopressin receptor antagonist, atosiban, delays the gastric emptying of a semisolid meal compared to saline in human Bodil Ohlsson, et. al., Sweden BMC Gastroenterology, 2006 Presented
Chief of Endocrinology East Orange General Hospital
 Targeting the Incretins System: Can it Improve Our Ability to Treat Type 2 Diabetes? Darshi Sunderam, MD Darshi Sunderam, MD Chief of Endocrinology East Orange General Hospital Age-adjusted Percentage
Targeting the Incretins System: Can it Improve Our Ability to Treat Type 2 Diabetes? Darshi Sunderam, MD Darshi Sunderam, MD Chief of Endocrinology East Orange General Hospital Age-adjusted Percentage
June By: Reza Gholami
 ACG/CAG guideline on Management of Dyspepsia June 2017 By: Reza Gholami DEFINITION OF DYSPEPSIA AND SCOPE OF THE GUIDELINE Dyspepsia was originally defined as any symptoms referable to the upper gastrointestinal
ACG/CAG guideline on Management of Dyspepsia June 2017 By: Reza Gholami DEFINITION OF DYSPEPSIA AND SCOPE OF THE GUIDELINE Dyspepsia was originally defined as any symptoms referable to the upper gastrointestinal
New Tests and Treatments for Dyspepsia and Irritable Bowel Syndrome
 New Tests and Treatments for Dyspepsia and Irritable Bowel Syndrome Soojong Hong Chae, MD Clinical Assistant Professor Digestive Diseases and Nutrition University of South Florida ROME III Functional dyspepsia
New Tests and Treatments for Dyspepsia and Irritable Bowel Syndrome Soojong Hong Chae, MD Clinical Assistant Professor Digestive Diseases and Nutrition University of South Florida ROME III Functional dyspepsia
Alimentary Pharmacology and Therapeutics SUMMARY
 Alimentary Pharmacology and Therapeutics Randomised clinical trial: ghrelin agonist TZP-101 relieves gastroparesis associated with severe nausea and vomiting randomised clinical study subset data J. M.
Alimentary Pharmacology and Therapeutics Randomised clinical trial: ghrelin agonist TZP-101 relieves gastroparesis associated with severe nausea and vomiting randomised clinical study subset data J. M.
Austin Radiological Association Nuclear Medicine Procedure GASTRIC EMPTYING STUDY (Tc-99m-Sulfur Colloid in Egg)
 Austin Radiological Association Nuclear Medicine Procedure GASTRIC EMPTYING STUDY (Tc-99m-Sulfur Colloid in Egg) Overview Indications The Gastric Emptying Study demonstrates the movement of an ingested
Austin Radiological Association Nuclear Medicine Procedure GASTRIC EMPTYING STUDY (Tc-99m-Sulfur Colloid in Egg) Overview Indications The Gastric Emptying Study demonstrates the movement of an ingested
Practical Strategies for the Clinical Use of Incretin Mimetics CME/CE. CME/CE Released: 09/15/2009; Valid for credit through 09/15/2010
 Practical Strategies for the Clinical Use of Incretin Mimetics CME/CE Robert R. Henry, MD Authors and Disclosures CME/CE Released: 09/15/2009; Valid for credit through 09/15/2010 Introduction Type 2 diabetes
Practical Strategies for the Clinical Use of Incretin Mimetics CME/CE Robert R. Henry, MD Authors and Disclosures CME/CE Released: 09/15/2009; Valid for credit through 09/15/2010 Introduction Type 2 diabetes
Small-Bowel and colon Transit. Mahsa Sh.Nezami October 2016
 Small-Bowel and colon Transit Mahsa Sh.Nezami October 2016 Dyspeptic symptoms related to dysmotility originating from the small bowel or colon usually include : Abdominal pain Diarrhea Constipation However,
Small-Bowel and colon Transit Mahsa Sh.Nezami October 2016 Dyspeptic symptoms related to dysmotility originating from the small bowel or colon usually include : Abdominal pain Diarrhea Constipation However,
GSK Medicine: Study Number: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Omeprazole delays gastric emptying in healthy volunteers: an effect prevented by tegaserod
 Aliment Pharmacol Ther 2005; 22: 59 65. doi: 10.1111/j.1365-2036.2005.02528.x Omeprazole delays gastric emptying in healthy volunteers: an effect prevented by tegaserod G. TOUGAS*,,D.L.EARNESTà, Y. CHEN*,
Aliment Pharmacol Ther 2005; 22: 59 65. doi: 10.1111/j.1365-2036.2005.02528.x Omeprazole delays gastric emptying in healthy volunteers: an effect prevented by tegaserod G. TOUGAS*,,D.L.EARNESTà, Y. CHEN*,
Non-insulin treatment in Type 1 DM Sang Yong Kim
 Non-insulin treatment in Type 1 DM Sang Yong Kim Chosun University Hospital Conflict of interest disclosure None Committee of Scientific Affairs Committee of Scientific Affairs Insulin therapy is the mainstay
Non-insulin treatment in Type 1 DM Sang Yong Kim Chosun University Hospital Conflict of interest disclosure None Committee of Scientific Affairs Committee of Scientific Affairs Insulin therapy is the mainstay
Safety, Tolerability, Pharmacokinetics,and Pharmacodynamics of Multiple Rising Doses of Empagliflozin in Patients with Type 2 Diabetes Mellitus
 Safety, Tolerability, Pharmacokinetics,and Pharmacodynamics of Multiple Rising Doses of Empagliflozin in Patients with Type 2 Diabetes Mellitus Journal Club (May 2013) Sara Al-Sharhan, Pharm D intern-
Safety, Tolerability, Pharmacokinetics,and Pharmacodynamics of Multiple Rising Doses of Empagliflozin in Patients with Type 2 Diabetes Mellitus Journal Club (May 2013) Sara Al-Sharhan, Pharm D intern-
Sponsor / Company: Sanofi Drug substance(s): insulin glargine (HOE901) According to template: QSD VERSION N 4.0 (07-JUN-2012) Page 1
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
Subject: Gastric Electric Simulation in Gastroparesis Patient. Review: HJ Hiew Dec Recommendation
 Subject: Gastric Electric Simulation in Gastroparesis Patient Review: HJ Hiew Dec 2014 Recommendation GES may be effective in reducing nausea and vomiting symptoms and need of enteral/parenteral nutrition
Subject: Gastric Electric Simulation in Gastroparesis Patient Review: HJ Hiew Dec 2014 Recommendation GES may be effective in reducing nausea and vomiting symptoms and need of enteral/parenteral nutrition
Clinical Policy: Gastric Electrical Stimulation
 Clinical Policy: Reference Number: CP.MP.40 Last Review Date: 08/18 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Description
Clinical Policy: Reference Number: CP.MP.40 Last Review Date: 08/18 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Description
Emerging Treatments for IBS-C and Clinical Trial Endpoints
 Emerging Treatments for IBS-C and Clinical Trial Endpoints Lin Chang, M.D. Oppenheimer Family Center for Neurobiology of Stress David Geffen School of Medicine at UCLA Learning Objectives Describe current
Emerging Treatments for IBS-C and Clinical Trial Endpoints Lin Chang, M.D. Oppenheimer Family Center for Neurobiology of Stress David Geffen School of Medicine at UCLA Learning Objectives Describe current
Clinical Policy: Gastric Electrical Stimulation Reference Number: CP.MP.40
 Clinical Policy: Reference Number: CP.MP.40 Effective Date: 09/09 Last Review Date: 10/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.40 Effective Date: 09/09 Last Review Date: 10/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Mechanisms and Clinical Efficacy of Lixisenatide for the Management of Type 2 Diabetes
 Adv Ther (2013) 30(2):81 101. DOI 10.1007/s12325-013-0009-4 REVIEW Mechanisms and Clinical Efficacy of Lixisenatide for the Management of Type 2 Diabetes Michael Horowitz Christopher K. Rayner Karen L.
Adv Ther (2013) 30(2):81 101. DOI 10.1007/s12325-013-0009-4 REVIEW Mechanisms and Clinical Efficacy of Lixisenatide for the Management of Type 2 Diabetes Michael Horowitz Christopher K. Rayner Karen L.
APDW 2016 Poster No. a90312
 APDW 2016 Poster No. a90312 SYN-010, a Proprietary Modified-Release Formulation of Lovastatin Lactone, Lowered Breath Methane and Improved Stool Frequency in Patients with IBS-C Results of a multi-center,
APDW 2016 Poster No. a90312 SYN-010, a Proprietary Modified-Release Formulation of Lovastatin Lactone, Lowered Breath Methane and Improved Stool Frequency in Patients with IBS-C Results of a multi-center,
Novel Formulations to Modify Mealtime Insulin Kinetics
 Novel Formulations to Modify Mealtime Insulin Kinetics Alan Krasner, Roderike Pohl, Patrick Simms, Philip Pichotta, Robert Hauser, Errol De Souza Biodel, Inc. Danbury, CT Disclosure All authors are employees
Novel Formulations to Modify Mealtime Insulin Kinetics Alan Krasner, Roderike Pohl, Patrick Simms, Philip Pichotta, Robert Hauser, Errol De Souza Biodel, Inc. Danbury, CT Disclosure All authors are employees
Carminatives IBS C 11/12/10. Partial List of Carminatives and Secondary Benefits. Partial List of Carminatives and Secondary Benefits
 IBS C Individuals experience a range of symptoms in addition to constipation (straining and hard stools) but also report abdominal pain and bloating as particularly troubling. Carminatives Estimated average
IBS C Individuals experience a range of symptoms in addition to constipation (straining and hard stools) but also report abdominal pain and bloating as particularly troubling. Carminatives Estimated average
EDUCATION PRACTICE. Management of Delayed Gastric Emptying. Clinical Scenario. The Problem. Management Strategies and Supporting Evidence
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2005;3:642 646 EDUCATION PRACTICE Management of Delayed Gastric Emptying FRANK K. FRIEDENBERG and HENRY P. PARKMAN Temple University Hospital, Philadelphia, Pennsylvania
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2005;3:642 646 EDUCATION PRACTICE Management of Delayed Gastric Emptying FRANK K. FRIEDENBERG and HENRY P. PARKMAN Temple University Hospital, Philadelphia, Pennsylvania
SYNOPSIS OF RESEARCH REPORT (PROTOCOL BC20779)
 TITLE OF THE STUDY / REPORT No. / DATE OF REPORT INVESTIGATORS / CENTERS AND COUNTRIES Clinical Study Report Protocol BC20779: Multicenter, double-blind, randomized, placebo-controlled, dose ranging phase
TITLE OF THE STUDY / REPORT No. / DATE OF REPORT INVESTIGATORS / CENTERS AND COUNTRIES Clinical Study Report Protocol BC20779: Multicenter, double-blind, randomized, placebo-controlled, dose ranging phase
GI Complications in heds and HSD
 GI Complications in heds and HSD Qasim Aziz PhD, FRCP Professor in Neurogastroenterology Neurogastroenterology Group GUT = Gastrointestinal (GI) tract Oesophagus / gullet Gastro-oesophageal junction Stomach
GI Complications in heds and HSD Qasim Aziz PhD, FRCP Professor in Neurogastroenterology Neurogastroenterology Group GUT = Gastrointestinal (GI) tract Oesophagus / gullet Gastro-oesophageal junction Stomach
William D Chey, 1 Anthony J Lembo, 2 James A Phillips, 3 David P Rosenbaum 4
 Efficacy and safety of tenapanor in patients with constipationpredominant irritable bowel syndrome: a 12-week, double-blind, placebocontrolled, randomized phase 2b trial William D Chey, 1 Anthony J Lembo,
Efficacy and safety of tenapanor in patients with constipationpredominant irritable bowel syndrome: a 12-week, double-blind, placebocontrolled, randomized phase 2b trial William D Chey, 1 Anthony J Lembo,
Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response
 Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response Blanca Serrano, 1 Silvia Delgado-Aros, 2 Fermín Mearin, 3 Constanza
Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response Blanca Serrano, 1 Silvia Delgado-Aros, 2 Fermín Mearin, 3 Constanza
What you really need to know about Gastroparesis?
 What you really need to know about Gastroparesis? John M. Wo, MD Division of Gastroenterology/Hepatology Director of GI Motility and Neurogastroenterology 8/3/2016 1/4/2017 1 What you really need to know
What you really need to know about Gastroparesis? John M. Wo, MD Division of Gastroenterology/Hepatology Director of GI Motility and Neurogastroenterology 8/3/2016 1/4/2017 1 What you really need to know
SYNOPSIS. Administration: subcutaneous injection Batch number(s):
 SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, 2-arm parallel-group, multicenter 24-week study followed by an extension assessing the efficacy and safety of AVE0010 on top
SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, 2-arm parallel-group, multicenter 24-week study followed by an extension assessing the efficacy and safety of AVE0010 on top
Opioid-Induced Constipation
 Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
DOMPERIDONE BNF 4.6. Domperidone is a dopamine type 2-receptor antagonist. It is structurally related to the
 DOMPERIDONE BNF 4.6 Class: Prokinetic anti-emetic. Indications: Nausea and vomiting, dysmotility dyspepsia, gastro-oesophageal reflux. Pharmacology Domperidone is a dopamine type 2-receptor antagonist.
DOMPERIDONE BNF 4.6 Class: Prokinetic anti-emetic. Indications: Nausea and vomiting, dysmotility dyspepsia, gastro-oesophageal reflux. Pharmacology Domperidone is a dopamine type 2-receptor antagonist.
Early satiety and postprandial fullness in gastroparesis correlate with gastroparesis severity, gastric emptying, and water load testing
 Received: 16 July 2016 Accepted: 23 September 2016 DOI: 10.1111/nmo.12981 ORIGINAL ARTICLE Early satiety and postprandial fullness in gastroparesis correlate with gastroparesis severity, gastric emptying,
Received: 16 July 2016 Accepted: 23 September 2016 DOI: 10.1111/nmo.12981 ORIGINAL ARTICLE Early satiety and postprandial fullness in gastroparesis correlate with gastroparesis severity, gastric emptying,
1. BACKGROUND. Lovastatin β-hydroxyacid No effect on methane production Inhibits cholesterol synthesis
 1. BACKGRUND Methane production by the archeon Methanobrevibacter smithii (M. smithii) is a ubiquitous process in the intestine, disposing of hydrogen and other by-products formed during carbohydrate digestion
1. BACKGRUND Methane production by the archeon Methanobrevibacter smithii (M. smithii) is a ubiquitous process in the intestine, disposing of hydrogen and other by-products formed during carbohydrate digestion
Functional Heartburn and Dyspepsia
 Functional Heartburn and Dyspepsia Nicholas Shaheen, MD, MPH Center for Esophageal Diseases and Swallowing University of North Carolina Objectives Understand the means of diagnosing functional heartburn
Functional Heartburn and Dyspepsia Nicholas Shaheen, MD, MPH Center for Esophageal Diseases and Swallowing University of North Carolina Objectives Understand the means of diagnosing functional heartburn
GERD: 2014 Dilemmas and Solutions. Ronnie Fass MD, FACP Professor of Medicine Case Western Reserve University
 GERD: 2014 Dilemmas and Solutions Ronnie Fass MD, FACP Professor of Medicine Case Western Reserve University How to Maximize Your PPI Treatment? Improve compliance and adherance Fass R. Am J Gastroenterol.
GERD: 2014 Dilemmas and Solutions Ronnie Fass MD, FACP Professor of Medicine Case Western Reserve University How to Maximize Your PPI Treatment? Improve compliance and adherance Fass R. Am J Gastroenterol.
MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES
 Enrique Rey Professor of Medicine Head. Department of Digestive Diseases Hospital Clínico San Carlos Complutense University Madrid, Spain MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES CONSTIPATION:
Enrique Rey Professor of Medicine Head. Department of Digestive Diseases Hospital Clínico San Carlos Complutense University Madrid, Spain MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES CONSTIPATION:
Lawrence R. Schiller, MD, FACG. Digestive Health Associates of Texas Baylor University Medical Center Dallas, Texas.
 Chronic Nausea & Vomiting Lawrence R. Schiller, MD, FACG Digestive Health Associates of Texas Baylor University Medical Center Dallas, Texas Nausea & Vomiting Common symptoms Quite troubling to patients
Chronic Nausea & Vomiting Lawrence R. Schiller, MD, FACG Digestive Health Associates of Texas Baylor University Medical Center Dallas, Texas Nausea & Vomiting Common symptoms Quite troubling to patients
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of gastroelectrical stimulation for gastroparesis Gastroparesis is a long-term condition
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of gastroelectrical stimulation for gastroparesis Gastroparesis is a long-term condition
Adlyxin. (lixisenatide) New Product Slideshow
 Adlyxin (lixisenatide) New Product Slideshow Introduction Brand name: Adlyxin Generic name: Lixisenatide Pharmacological class: Glucagon-like peptide-1 (GLP-1) receptor agonist Strength and Formulation:
Adlyxin (lixisenatide) New Product Slideshow Introduction Brand name: Adlyxin Generic name: Lixisenatide Pharmacological class: Glucagon-like peptide-1 (GLP-1) receptor agonist Strength and Formulation:
Evaluation of gastrointestinal transit in clinical practice: position paper of the American and European Neurogastroenterology and Motility Societies
 Neurogastroenterol Motil (2011) 23, 8 23 doi: 10.1111/j.1365-2982.2010.01612.x REVIEW ARTICLE Evaluation of gastrointestinal transit in clinical practice: position paper of the American and European Neurogastroenterology
Neurogastroenterol Motil (2011) 23, 8 23 doi: 10.1111/j.1365-2982.2010.01612.x REVIEW ARTICLE Evaluation of gastrointestinal transit in clinical practice: position paper of the American and European Neurogastroenterology
1.* Dosage/Administration
 Pramlintide (Symlin ) [Developed, February 2006; Revised, May 2006; January 2010; December 2011; October 2013] MEDICAID DRUG USE REVIEW CRITERIA FOR OUTPATIENT USE Information on indications for use or
Pramlintide (Symlin ) [Developed, February 2006; Revised, May 2006; January 2010; December 2011; October 2013] MEDICAID DRUG USE REVIEW CRITERIA FOR OUTPATIENT USE Information on indications for use or
I. ALL CLAIMS: HEALTH CARE PROFESSIONALS
 HCP Prescribing Information Date/Version January 2015 Version 2 Page: 1 of 5 I. ALL CLAIMS: HEALTH CARE PROFESSIONALS Indications and Usage Saxenda (liraglutide [rdna origin] injection) is indicated as
HCP Prescribing Information Date/Version January 2015 Version 2 Page: 1 of 5 I. ALL CLAIMS: HEALTH CARE PROFESSIONALS Indications and Usage Saxenda (liraglutide [rdna origin] injection) is indicated as
23-Aug-2011 Lixisenatide (AVE0010) - EFC6014 Version number: 1 (electronic 1.0)
 SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, multicenter 24-week study followed by an extension assessing the efficacy and safety of AVE0010 on top of metformin
SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, multicenter 24-week study followed by an extension assessing the efficacy and safety of AVE0010 on top of metformin
Oral Methylnaltrexone for the. Constipation in Patients with Chronic Non-cancer Pain
 Oral Methylnaltrexone for the Treatment of Opioid-induced Constipation in Patients with Chronic Non-cancer Pain Richard L. Rauck, 1 John F. Peppin, 2 Robert J.Israel, 3 Jennifer Carpenito, 3 Jeffrey Cohn,
Oral Methylnaltrexone for the Treatment of Opioid-induced Constipation in Patients with Chronic Non-cancer Pain Richard L. Rauck, 1 John F. Peppin, 2 Robert J.Israel, 3 Jennifer Carpenito, 3 Jeffrey Cohn,
IBS The Physiologist s Perspective
 IBS The Physiologist s Perspective Dr Anthony R. Hobson PhD Consultant Clinical Scientist, London The Functional Gut Clinic Reclaiming the F word The f-word functional has become a by-word for failure
IBS The Physiologist s Perspective Dr Anthony R. Hobson PhD Consultant Clinical Scientist, London The Functional Gut Clinic Reclaiming the F word The f-word functional has become a by-word for failure
Clinical Snapshot: Making sense of Autonomic Neuropathy. M S Kamaruddin Consultant Physician South Tees NHS Foundation Trust
 Clinical Snapshot: Making sense of Autonomic Neuropathy M S Kamaruddin Consultant Physician South Tees NHS Foundation Trust Diabetic autonomic neuropathy Classified as clinical or subclinical based on
Clinical Snapshot: Making sense of Autonomic Neuropathy M S Kamaruddin Consultant Physician South Tees NHS Foundation Trust Diabetic autonomic neuropathy Classified as clinical or subclinical based on
Effects of rehabilitation management on gastric emptying function in older adults with diabetes
 Effects of rehabilitation management on gastric emptying function in older adults with diabetes Z.M. Shao 1 *, J.F. Yao 2 *, J. Chen 2 *, Z.W. Yu 3, X.F. Yu 2, J.J. Zheng 3, X. Tang 4 1 Office of the Director
Effects of rehabilitation management on gastric emptying function in older adults with diabetes Z.M. Shao 1 *, J.F. Yao 2 *, J. Chen 2 *, Z.W. Yu 3, X.F. Yu 2, J.J. Zheng 3, X. Tang 4 1 Office of the Director
Safety profile of Liraglutide: Recent Updates. Mohammadreza Rostamzadeh,M.D.
 Safety profile of Liraglutide: Recent Updates Mohammadreza Rostamzadeh,M.D. Pancreatitis: Victoza post-marketing experience: spontaneous reports of pancreatitis For the majority of the cases, there is
Safety profile of Liraglutide: Recent Updates Mohammadreza Rostamzadeh,M.D. Pancreatitis: Victoza post-marketing experience: spontaneous reports of pancreatitis For the majority of the cases, there is
OVERVIEW 1. RADIOPHARMACEUTICAL UTILIZED. a) The most commonly used radiopharmaceutical is Tc-99m labeled egg whites (albumin).
 OVERVIEW a) A Gastric Emptying Study is a radionuclide diagnostic imaging study. The most common type of gastric emptying study is a procedure that is done by nuclear medicine physicians using a small
OVERVIEW a) A Gastric Emptying Study is a radionuclide diagnostic imaging study. The most common type of gastric emptying study is a procedure that is done by nuclear medicine physicians using a small
Irritable Bowel Syndrome: Last year FODMAPs, this year bile acids
 Irritable Bowel Syndrome: Last year FODMAPs, this year bile acids Lana Bistritz, MD FRCPC Division of Gastroenterology Royal Alexandra Hospital Disclosures I have no financial conflicts of interest relevant
Irritable Bowel Syndrome: Last year FODMAPs, this year bile acids Lana Bistritz, MD FRCPC Division of Gastroenterology Royal Alexandra Hospital Disclosures I have no financial conflicts of interest relevant
IBS: overview and assessment of pain outcomes and implications for inclusion criteria
 IBS: overview and assessment of pain outcomes and implications for inclusion criteria William D. Chey, MD Professor of Medicine University of Michigan What is the Irritable Bowel Syndrome Symptom based
IBS: overview and assessment of pain outcomes and implications for inclusion criteria William D. Chey, MD Professor of Medicine University of Michigan What is the Irritable Bowel Syndrome Symptom based
GASTROPARESIS 101: BEGINNERS GUIDE TO UNDERSTANDING GASTROPARESIS By: Dr. Josh Ferguson 2017
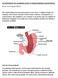 GASTROPARESIS 101: BEGINNERS GUIDE TO UNDERSTANDING GASTROPARESIS By: Dr. Josh Ferguson 2017 This tough looking word, pronounced gas-tro-par-ees-is, is simple enough in its meaning. Gastro means stomach.
GASTROPARESIS 101: BEGINNERS GUIDE TO UNDERSTANDING GASTROPARESIS By: Dr. Josh Ferguson 2017 This tough looking word, pronounced gas-tro-par-ees-is, is simple enough in its meaning. Gastro means stomach.
Gastric Electrical Stimulation
 Gastric Electrical Stimulation Policy Number: 7.01.73 Last Review: 6/2017 Origination: 7/2002 Next Review: 6/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for
Gastric Electrical Stimulation Policy Number: 7.01.73 Last Review: 6/2017 Origination: 7/2002 Next Review: 6/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for
Bariatric Surgery vs. Intensive Medical Therapy in Obese Diabetic Patients: 3-Year Outcomes
 Bariatric Surgery vs. Intensive Medical Therapy in Obese Diabetic Patients: 3-Year Outcomes Results of the STAMPEDE Trial Philip R Schauer, Deepak L Bhatt, John P Kirwan, Kathy Wolski, Stacy A Brethauer,
Bariatric Surgery vs. Intensive Medical Therapy in Obese Diabetic Patients: 3-Year Outcomes Results of the STAMPEDE Trial Philip R Schauer, Deepak L Bhatt, John P Kirwan, Kathy Wolski, Stacy A Brethauer,
Gastric Electrical Stimulation
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Subclinical hypothyroidism (SCH) is defined as an elevated
 JCEM ONLINE Brief Report Endocrine Care Effects of L-Thyroxine on Gastric Motility and Ghrelin in Subclinical Hypothyroidism: A Prospective Study Asena Gökçay Canpolat, Taylan Kav, Bülent Sı vrı, and Bülent
JCEM ONLINE Brief Report Endocrine Care Effects of L-Thyroxine on Gastric Motility and Ghrelin in Subclinical Hypothyroidism: A Prospective Study Asena Gökçay Canpolat, Taylan Kav, Bülent Sı vrı, and Bülent
Is there a role for medical marijuana in the management of IBD? Jami A. Kinnucan, MD Division of Gastroenterology University of Michigan Hospital
 Is there a role for medical marijuana in the management of IBD? Jami A. Kinnucan, MD Division of Gastroenterology University of Michigan Hospital Disclosures No relevant financial or relationship disclosures
Is there a role for medical marijuana in the management of IBD? Jami A. Kinnucan, MD Division of Gastroenterology University of Michigan Hospital Disclosures No relevant financial or relationship disclosures
Sponsor. Generic Drug Name. Trial Indication(s) Protocol Number. Protocol Title. Clinical Trial Phases. Study Start/End Dates
 Sponsor Novartis Generic Drug Name Lumiracoxib Trial Indication(s) Safety study effects on small bowel Protocol Number CCOX189A2425 Protocol Title A 16-day, randomized, double-blind, double-dummy, placebo-controlled,
Sponsor Novartis Generic Drug Name Lumiracoxib Trial Indication(s) Safety study effects on small bowel Protocol Number CCOX189A2425 Protocol Title A 16-day, randomized, double-blind, double-dummy, placebo-controlled,
Original Policy Date
 MP 7.01.57 Gastric Electrical Stimulation Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy
MP 7.01.57 Gastric Electrical Stimulation Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: gastric_electrical_stimulation 9/2003 5/2017 5/2018 5/2017 Description of Procedure or Service Gastric electrical
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: gastric_electrical_stimulation 9/2003 5/2017 5/2018 5/2017 Description of Procedure or Service Gastric electrical
Chapter 1. General introduction and outline
 Chapter 1 General introduction and outline General introduction The function of the stomach comprises storage of ingested food, production of gastric secretion and mixing food with gastric secretion,
Chapter 1 General introduction and outline General introduction The function of the stomach comprises storage of ingested food, production of gastric secretion and mixing food with gastric secretion,
Christine Pelkman, PhD
 Christine Pelkman, PhD Dr. Pelkman is a graduate faculty member in Nutrition, and Director of the Nutrition and Health Research Laboratory at the University of Buffalo. She completed her doctoral and postdoctoral
Christine Pelkman, PhD Dr. Pelkman is a graduate faculty member in Nutrition, and Director of the Nutrition and Health Research Laboratory at the University of Buffalo. She completed her doctoral and postdoctoral
SLENDESTA POTATO EXTRACT PROMOTES SATIETY IN HEALTHY HUMAN SUBJECTS: IOWA STATE UNIVERSITY STUDY Sheila Dana, Michael Louie, Ph.D. and Jiang Hu, Ph.D.
 SLENDESTA POTATO EXTRACT PROMOTES SATIETY IN HEALTHY HUMAN SUBJECTS: IOWA STATE UNIVERSITY STUDY Sheila Dana, Michael Louie, Ph.D. and Jiang Hu, Ph.D. INTRODUCTION KEY CONCLUSIONS Excessive calorie intake
SLENDESTA POTATO EXTRACT PROMOTES SATIETY IN HEALTHY HUMAN SUBJECTS: IOWA STATE UNIVERSITY STUDY Sheila Dana, Michael Louie, Ph.D. and Jiang Hu, Ph.D. INTRODUCTION KEY CONCLUSIONS Excessive calorie intake
Subcutaneous exendin (9-39) effectively treats post-bariatric hypoglycemia
 Subcutaneous exendin (9-39) effectively treats post-bariatric hypoglycemia C OLLEEN M. CRAIG, M.D. T RACEY L. MC L AUGHLIN, M.D., M.S. Division of Endocrinology, Metabolism & Gerontology Stanford University
Subcutaneous exendin (9-39) effectively treats post-bariatric hypoglycemia C OLLEEN M. CRAIG, M.D. T RACEY L. MC L AUGHLIN, M.D., M.S. Division of Endocrinology, Metabolism & Gerontology Stanford University
Efficacy/pharmacodynamics: 85 Safety: 89
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor/Company: Sanofi Drug substance:
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor/Company: Sanofi Drug substance:
Investigators, study sites Multicenter, 35 US sites. Coordinating Investigator: Richard Bergenstal, MD
 STUDY SYNOPSIS Study number Title HMR1964A/3502 Apidra (insulin glulisine) administered in a fixed-bolus regimen vs. variable-bolus regimen based on carbohydrate counting in adult subjects with type 2
STUDY SYNOPSIS Study number Title HMR1964A/3502 Apidra (insulin glulisine) administered in a fixed-bolus regimen vs. variable-bolus regimen based on carbohydrate counting in adult subjects with type 2
OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES
 OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES Introduction Introduction Mean faecal weight 128 g / cap / day Mean range 51-796 g Absolute range 15-1505 g Main factors affecting mass are caloric intake,
OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES Introduction Introduction Mean faecal weight 128 g / cap / day Mean range 51-796 g Absolute range 15-1505 g Main factors affecting mass are caloric intake,
Treatment of gastrointestinal autonomic neuropathy
 Diabetologia (2016) 59:409 413 DOI 10.1007/s00125-015-3828-9 MINI-REVIEW Treatment of gastrointestinal autonomic neuropathy Hans Törnblom 1 Received: 31 October 2015 /Accepted: 5 November 2015 /Published
Diabetologia (2016) 59:409 413 DOI 10.1007/s00125-015-3828-9 MINI-REVIEW Treatment of gastrointestinal autonomic neuropathy Hans Törnblom 1 Received: 31 October 2015 /Accepted: 5 November 2015 /Published
Beyond A1C. Non-glycemic Effects of GLP-1 Receptor Agonists. Olga Astapova MD, PhD Luis Chavez MD URMC Endocrinology Fellows
 Beyond A1C Non-glycemic Effects of GLP-1 Receptor Agonists Olga Astapova MD, PhD Luis Chavez MD URMC Endocrinology Fellows Disclosures No conflicts of interest. Learning Objectives 1. Understand the physiological
Beyond A1C Non-glycemic Effects of GLP-1 Receptor Agonists Olga Astapova MD, PhD Luis Chavez MD URMC Endocrinology Fellows Disclosures No conflicts of interest. Learning Objectives 1. Understand the physiological
Gastric Emptying Time after Laparoscopic Sleeve Gastrectomy
 International Journal of Current Research in Medical Sciences ISSN: 2454-5716 P-ISJN: A4372-3064, E -ISJN: A4372-3061 www.ijcrims.com Original Research Article Volume 4, Issue 7-2018 Gastric Emptying Time
International Journal of Current Research in Medical Sciences ISSN: 2454-5716 P-ISJN: A4372-3064, E -ISJN: A4372-3061 www.ijcrims.com Original Research Article Volume 4, Issue 7-2018 Gastric Emptying Time
SYNOPSIS. Number of subjects: Planned: 22 Randomized: 23 Treated: 23. Evaluated: Pharmacodynamic: 22 Safety: 23 Pharmacokinetics: 22
 SYNOPSIS Title of the study: A randomized, cross-over, open, euglycemic clamp study on the relative bioavailability and activity of 0.6 U/kg insulin glargine and 20 µg lixisenatide, given as on-site mix
SYNOPSIS Title of the study: A randomized, cross-over, open, euglycemic clamp study on the relative bioavailability and activity of 0.6 U/kg insulin glargine and 20 µg lixisenatide, given as on-site mix
pissn: eissn: Journal of Neurogastroenterology and Motility
 JNM J Neurogastroenterol Motil, Vol. 20 No. 3 July, 2014 pissn: 2093-0879 eissn: 2093-0887 http://dx.doi.org/10.5056/jnm14022 Original Article Effects of Chili Treatment on Gastrointestinal and Rectal
JNM J Neurogastroenterol Motil, Vol. 20 No. 3 July, 2014 pissn: 2093-0879 eissn: 2093-0887 http://dx.doi.org/10.5056/jnm14022 Original Article Effects of Chili Treatment on Gastrointestinal and Rectal
