A Randomized Trial of Low-Dose Aspirin in the Prevention of Clinical Type 2 Diabetes in Women
|
|
|
- George Potter
- 5 years ago
- Views:
Transcription
1 A Randomized Trial of Low-Dose Aspirin in the Prevention of Clinical Type 2 Diabetes in Women The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published Version Accessed Citable Link Terms of Use Pradhan, Aruna D., Nancy R. Cook, JoAnn E. Manson, Paul M. Ridker, and Julie E. Buring A Randomized Trial of Low- Dose Aspirin in the Prevention of Clinical Type 2 Diabetes in Women. Diabetes Care 32(1): 3-8. doi: /dc August 25, :03:53 PM EDT This article was downloaded from Harvard University's DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at (Article begins on next page)
2 Clinical Care/Education/Nutrition/Psychosocial Research O R I G I N A L A R T I C L E A Randomized Trial of Low-Dose Aspirin in the Prevention of Clinical Type 2 Diabetes in Women ARUNA D. PRADHAN, MD 1,2,3,4 NANCY R. COOK, SCD 1,2,3 JOANN E. MANSON, MD 3 1,2,3,5,6 PAUL M. RIDKER, MD JULIE E. BURING, SCD 1,3 OBJECTIVE Subclinical inflammation is linked with the development of type 2 diabetes, and epidemiologic data suggest that this association may be stronger in women. Although small clinical studies have shown a prominent hypoglycemic effect of short-term high-dose aspirin, no randomized trials have directly evaluated the efficacy of aspirin in diabetes prevention at doses acceptable for use in routine clinical practice. We evaluated whether chronic low-dose aspirin prevents the development of clinical diabetes among initially healthy American women. RESEARCH DESIGN AND METHODS Subjects were enrolled in the Women s Health Study, a 10-year randomized double-blind, placebo-controlled trial of aspirin and vitamin E for primary prevention of cardiovascular disease and cancer. Between 1992 and 1995, 38,716 women aged 45 years and free of clinical diabetes were randomly assigned to either low-dose aspirin or placebo (median follow-up 10.2 years). Documented clinical type 2 diabetes was prospectively evaluated throughout the trial. RESULTS Among women randomly assigned to receive aspirin (n 19,326) or placebo (n 19,390), there was no statistically significant difference in the incidence of type 2 diabetes. There were 849 cases of diabetes in the aspirin group and 847 in the placebo group (rate ratio 1.01 [95% CI ]). Stratification by diabetes risk factors including age, BMI, family history of diabetes, physical activity, A1C, and high-sensitivity C-reactive protein did not support a modulating effect of these variables. Analyses accounting for treatment duration and adherence similarly found no beneficial effects. CONCLUSIONS These data suggest that long-term low-dose aspirin does not prevent the development of clinical type 2 diabetes in initially healthy women. The ability of salicylates, such as aspirin, to reduce glucose levels was described 125 years ago (1). This effect was largely forgotten until the emergence of recent data linking inflammation with the development of type 2 diabetes. A wealth of experimental and epidemiological evidence now indicates that insulin resistance and type 2 diabetes are, in Diabetes Care 32:3 8, 2009 part, obesity-linked inflammatory disorders (2), and the presence of subclinical inflammation is now known to be a potent indicator of risk for this disease. This relationship may be of particular importance in the pathogenesis of diabetes in women among whom obesity-triggered inflammation may be heightened compared with that in men (3). These findings From the 1 Center for Cardiovascular Disease Prevention, Brigham and Women s Hospital and Harvard Medical School, Boston, Massachusetts; the 2 Donald W. Reynolds Center for Cardiovascular Research, Brigham and Women s Hospital and Harvard Medical School, Boston, Massachusetts; the 3 Division of Preventive Medicine, Brigham and Women s Hospital and Harvard Medical School, Boston, Massachusetts; the 4 Division of Cardiology, VA Boston Medical Center, Boston, Massachusetts; the 5 Leducq Center for Molecular and Genetic Epidemiology of Cardiovascular Disorders, Brigham and Women s Hospital and Harvard Medical School, Boston, Massachusetts; and the 6 Division of Cardiovascular Medicine, Brigham and Women s Hospital and Harvard Medical School, Boston, Massachusetts. Corresponding author: Aruna D. Pradhan, apradhan@partners.org. Received 1 July 2008 and accepted 18 September Published ahead of print at on 3 October DOI: /dc Clinical trial reg. no. NCT , clinicaltrials.gov by the American Diabetes Association. Readers may use this article as long as the work is properly cited, the use is educational and not for profit, and the work is not altered. See org/licenses/by-nc-nd/3.0/ for details. The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. have spurred interest in the use of antiinflammatory drugs in diabetes prevention and treatment (4). However, data pertaining to this novel approach are sparse with no large-scale randomized studies available to date. Aspirin is an anti-inflammatory agent with pleiotropic actions, many of which remain poorly understood. The cellular and molecular mechanisms of the hypoglycemic response to aspirin are an area of active investigation but likely involve anti-inflammatory pathways distinct from effects on prostaglandin synthesis (5,6). Several small clinical studies (7 10) have reported that short-term high-dose aspirin (3 10 g/day for 3 days 3 weeks) improves glucose handling and may ameliorate insulin resistance in diabetic patients, albeit with a high rate of side effects. Although data are not available on the hypoglycemic action of low-dose aspirin, several short-term clinical trials demonstrated that aspirin triggers the production of anti-inflammatory mediators (11) and lowers systemic levels of inflammatory biomarkers at doses as low as 30 mg/day (12). Whether chronic lowdose aspirin therapy has favorable clinical effects is unknown. We assessed whether long-term lowdose aspirin therapy reduces the incidence of clinical type 2 diabetes in the randomized treatment arms of the Women s Health Study (WHS). The WHS tested the efficacy of low-dose aspirin in the primary prevention of cardiovascular disease and cancer over a 10-year period in a large group of initially healthy women. The occurrence of clinical diabetes was ascertained prospectively throughout the trial. RESEARCH DESIGN AND METHODS The WHS was a 2 2 factorial trial evaluating the balance of risks and benefits of low-dose aspirin (100 mg every other day; Bayer Healthcare) and vitamin E (600 IU -tocopherol every other day; Natural Source Vitamin E Association) in the primary prevention of cardiovascular disease and cancer (13 15). The dose of 100 mg every other day was chosen to be the lowest dose that DIABETES CARE, VOLUME 32, NUMBER 1, JANUARY
3 Aspirin does not lower diabetes incidence would have a cardioprotective effect while minimizing gastrointestinal side effects. Although not a prespecified primary end point of the WHS, clinical diabetes was a key outcome of interest and prospectively ascertained throughout the duration of the trial. Written informed consent was obtained from all women. The study was approved by the institutional review board of the Brigham and Women s Hospital and monitored by an external data and safety monitoring board. Setting and participants Details of the study design have previously been described (13,16). Women were eligible if they were at least 45 years of age without a previous history of cancer (except nonmelanoma skin cancer), cardiovascular disease, or other major chronic illness; had no history of adverse effects from aspirin; were not taking aspirin or nonsteroidal anti-inflammatory drugs (or were willing to forgo their use during the trial); and were not taking anticoagulants or individual supplements of vitamin A, E, or -carotene more than once a week. A total of 39,876 women were willing, eligible, and compliant during a 3-month placebo run-in period and underwent random assignment: 19,934 were assigned to receive aspirin and 19,942 to receive placebo. In the present analyses, we excluded women with reported physician-diagnosed diabetes at baseline (n 1,160), leaving a total of 38,716 women free of clinical diabetes at entry into the trial; 19,326 were assigned to receive aspirin and 19,390 to receive placebo. Additional details including a Consolidated Standards of Reporting Trials (CONSORT) flow diagram are provided in supplemental data and supplemental Fig. 1 (available in an online appendix at /dc ). Randomization and interventions With use of a computer-generated table of random numbers, treatment assignments were made within seven age-groups (45 49, 50 54, 55 59, 60 64, 65 69, 70 74, and 75 years) using block sizes of 16. Random assignment took place from 30 April 1993 through 24 January Each year, women received calendar packs that contained amber capsules (vitamin E or placebo) and white pills (aspirin or placebo) on alternate days. Every 6 months for the first year and annually thereafter, they also received follow-up questionnaires inquiring about compliance with pill-taking, potential adverse effects, occurrence of end points, and risk factors. Study medications were continued in blinded fashion through the scheduled end of the trial (31 March 2004). Compliance On the basis of self-reported adherence, compliance, defined as taking at least two-thirds of their aspirin or matching placebo tablets, was 76.1% at 5 years and 67.0% at 10 years. Averaged throughout the trial, compliance was slightly higher in the placebo (73.7%) group than in the active (72.5%) group (P 0.004). Nontrial use of aspirin or aspirin-containing products on 4 days/month ( drop-ins ) was 11.6% at 5 years and 19.2% at 10 years. Averaged throughout the trial, it was somewhat higher in the placebo (13.0%) group than in the active (12.7%) group (P 0.10). Outcomes and follow-up Details regarding the ascertainment of incident diabetes in the WHS have been reported previously (17). Information on newly diagnosed diabetes was collected on every follow-up questionnaire from baseline through the end of the trial. All participants were asked annually In the past year, were you newly diagnosed with diabetes mellitus? Subjects also provided the month and year of diagnosis. Confirmation of diabetes was conducted in a blinded fashion using American Diabetes Association diagnostic criteria (18). Selfreported cases were investigated by either telephone interview conducted by a physician or a previously validated selfadministered supplemental questionnaire that inquired about symptoms, diagnostic testing, and use of diabetes medications (19). The response rate was high with 90% of women who reported incident diabetes responding to either the telephone interview or supplemental questionnaire. On the basis of responses to the supplemental questionnaire, 77.2% of those with confirmed cases reported use of antidiabetic agents. Only women with confirmed cases were analyzed in this report. Because the vast majority of diabetes diagnosed at age 45 years is of the type 2 variant, incident diabetes in the WHS is considered to be type 2 diabetes. Glucose screening rates were assessed during follow-up. When asked about screening for diabetes on the 9-year questionnaire, 71.8 and 68.2% of nondiabetic women reported having a fasting glucose test performed within the preceding 5 and 3 years, respectively. Screening rates were equivalent between the two treatment arms: 68.3 versus 68.1% in the preceding 3 years in the aspirin and placebo groups, respectively (P 0.78). These values are similar to contemporaneous diabetes screening rates; among patients in a U.S. managed care population, the occurrence of any glucose testing (random or fasting) over a 3-year period was 71.5% for women 45 years (20). Statistical analysis All primary analyses were performed on an intention-to-treat basis. We used Cox proportional hazards models to estimate the rate ratio (RR) and 95% CIs, comparing event rates in the aspirin and placebo groups after adjustment for age and other randomized treatment assignments (vitamin E and -carotene, which was a component of the trial for a median of 2.1 years) (21). The proportionality assumption of constant hazards over time was tested using an interaction term of aspirin with the logarithm of time. The divergence of diabetes incidence over time between groups was estimated using Kaplan-Meier survival curves and the logrank test was computed to compare curves. Exploratory subgroup analyses were conducted to examine the effect of aspirin according to the baseline presence of major risk factors for type 2 diabetes including age-group, BMI group, family history of diabetes in a first-degree relative, physical activity, menopausal status, and hormone therapy. Categories are specified in Table 1. Among women providing baseline blood specimens (n 27,167), subgroup analyses were performed after stratification by levels of total cholesterol, LDL cholesterol, HDL cholesterol, the total cholesterol to HDL cholesterol ratio, non-hdl cholesterol, A1C, and highsensitivity C-reactive protein (hscrp). To estimate the effect of treatment duration, we fit two separate proportional hazards models to the experience of the first 5 years and after 5 years of follow-up. To assess the impact of potentially undiagnosed diabetes at baseline, we conducted sensitivity analyses in which women with diabetes diagnosed during the first 2 and 5 years of follow-up were excluded. To examine the effect of actual as opposed to assigned aspirin use, we performed additional analyses in which subjects were censored if and when they stopped tak- 4 DIABETES CARE, VOLUME 32, NUMBER 1, JANUARY 2009
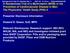
4 Pradhan and Associates Table 1 Baseline characteristics of the study population ing at least two-thirds of their study pills. To assess the effect of nontrial aspirin use, we also performed analyses in which censoring occurred when women either stopped taking at least two-thirds of their study pills or reported outside use of aspirin or aspirin-containing products for 4 days/month. All analyses were performed with SAS (version 9.1; SAS Institute, Cary, NC). A two-sided significance level of 0.05 was used. RESULTS The aspirin and placebo groups were similar with respect to baseline characteristics (Table 1). Among women providing blood specimens, there were also no significant differences in lipid levels, A1C, or hscrp. The median duration of follow-up was 10.2 years Aspirin Placebo Total population n 19,326 19,390 Age category (%) years years years BMI category (%) 25.0 kg/m kg/m kg/m Ethnicity (%) White not of Hispanic origin African American Hispanic American Indian or Alaskan Native Asian or Pacific Islander Unknown (none of the above) Family history of diabetes (%)* Current smoker (%) Exercise once weekly (%) Alcohol consumption once weekly (%) History of hypertension (%) History of hyperlipidemia (%) Postmenopausal (%) Baseline use of hormone therapy (%) Available baseline blood specimen n 13,595 13,572 Total cholesterol (mmol/l) 5.41 ( ) 5.38 ( ) LDL cholesterol (mmol/l) 3.15 ( ) 3.13 ( ) HDL cholesterol (mmol/l) 1.35 ( ) 1.35 ( ) Total cholesterol to HDL cholesterol ratio 4.0 ( ) 3.9 ( ) Non-HDL cholesterol(mmol/l) 3.99 ( ) 3.98 ( ) A1C (%) 5.0 ( ) 5.0 ( ) hscrp (mg/l) 2.0 ( ) 2.0 ( ) Data are % or median (interquartile range). *Family history of diabetes in a first-degree relative (mother, father, sister, or brother). Physical activity defined by number of episodes of vigorous physical activity per week. (mean 9.8 years). At completion of the trial, 1,696 cases of confirmed incident clinical type 2 diabetes had occurred. There were 849 cases in the aspirin group and 847 cases in the placebo group, with no significant risk reduction (RR for aspirin versus placebo 1.01 [95% CI ]). The cumulative incidence curves according to treatment assignment were similar throughout follow-up (Fig. 1) (log-rank P 0.92). A test of the proportionality hazards assumption showed no deviation from proportionality (P 0.27). In separate analyses that considered newly diagnosed diabetes in the first 5 years of follow-up versus thereafter, aspirin therapy was associated with an RR of 0.99 (95% CI ) during the first 5 years and 1.01 ( ) after 5 years. We found no difference when patients with potentially undiagnosed diabetes documented during the first 2 or 5 years of follow-up were excluded. In these analyses the RRs were 0.99 ( ) and 1.01 ( ), respectively. There was no evidence that any diabetes risk factors considered modified the effect of aspirin on diabetes incidence (Table 2) (P interaction 0.2 for clinical risk factors). Although we did find statistically significant risk reductions among women who had total cholesterol levels 6.21 mmol/l (RR 0.77 [95% CI ], P 0.028) and LDL cholesterol 4.13 mmol/l (0.76 [ ], P 0.049), with borderline nonsignificant findings for women with total cholesterol to HDL cholesterol ratio 6.0 (0.81 [ ], P 0.053), these findings must be interpreted with caution given both the lack of a consistent risk increase across biomarker categories and the large number of subgroups examined. There was no statistically significant interaction for any of the biomarkers assessed (P interaction 0.39 for total cholesterol, 0.26 for LDL cholesterol, 0.13 for the total cholesterol to HDL cholesterol ratio, and 0.2 for all others). Because compliance diminished over time, sensitivity analyses were performed that censored noncompliant women at the time they stopped taking at least twothirds of their study pills during the preceding year. In this analysis, there was also no significant benefit of aspirin (RR 1.02 [95% CI ]). When women were censored at the time they either stopped taking at least two-thirds of their study pills or started outside aspirin or aspirin-containing medications on 4 days/month, findings were similarly nonsignificant. In addition, there was no evidence of effect modification by other randomized treatments. With regard to safety, as expected, there were slightly higher rates of clinically significant bleeding episodes and other side effects in the aspirin arm compared with placebo: 4.5 vs. 3.7% (RR 1.22, P 0.001) for any bleeding event, 0.6 vs. 0.4% (RR 1.37, P 0.03) for transfusion-requiring bleeding, 2.7 vs. 2.1% (RR 1.30, P 0.001) for peptic ulcer, 15.2 vs. 14.4% (RR 1.06, P 0.02) for hematuria, and 19.0 vs. 16.5% (RR 1.17, P 0.001) for epistaxis. CONCLUSIONS In this largescale, long-term trial of initially healthy women, there was no association of 100 DIABETES CARE, VOLUME 32, NUMBER 1, JANUARY
5 Aspirin does not lower diabetes incidence Figure 1 Cumulative incidence of type 2 diabetes in the aspirin and placebo groups. mg aspirin on alternate days with the overall incidence of clinical type 2 diabetes. Treatment duration, 5 years or 5 years, did not have an impact on our results, and the treatment effect did not vary significantly across subgroups of women at high risk for diabetes due to the presence of clinical risk factors, dyslipidemia, elevated A1C, or hscrp. There was no difference when women with early cases, presumably undiagnosed at baseline, were excluded or in analyses accounting for adherence to randomized assignment. Aspirin therapy was associated with a higher incidence of clinically important bleeding events. The increasing incidence of diabetes, high treatment costs, and disproportionate impact on cardiovascular disease in women highlight the need to identify prevention strategies applicable on a broad population basis. Preventive measures with potential dual effects on cardiovascular and diabetes risk reduction are particularly appealing because cardiometabolic risk factors often coincide in the same individual. The main findings of the Women s Health Study were published previously and demonstrated that longterm treatment with low-dose aspirin, although resulting in no significant benefit or harm on the end point of any first major cardiovascular event, did significantly reduce the risk of stroke overall (RR 0.83 [95% CI ]) and myocardial infarction among women aged 65 years (0.66 [ ]) (13). The current analysis addresses the important question of whether low-dose aspirin has added benefits for diabetes prevention in this large population of otherwise healthy women. This report provides the only randomized data available in this regard. One of the least appreciated pharmacological actions of aspirin is its ability to lower glucose levels. Attempts to delineate this effect have been made in several small clinical studies. Among both nondiabetic and diabetic patients, short-term high-dose aspirin (3 10 g/day for up to 3 weeks) is consistently associated with higher basal and stimulated insulin concentrations and reduced glucose excursion during glucose tolerance testing (8 10,22). However, whether aspirin has a net beneficial contribution to glucose homeostasis has been controversial, with several studies suggesting that any favorable hypoglycemic action is offset by deterioration in insulin sensitivity (23,24). Higher doses of aspirin given over a longer period were more recently found to have broad therapeutic benefits in a detailed study of nine patients with overt type 2 diabetes (10). At the end of 2 weeks, aspirin at a dose of 7 g/day lowered fasting glucose ( 25%) and C-reactive protein ( 15%) while improving glucose tolerance ( 20%), reducing basal hepatic glucose production (20%), and increasing insulin-stimulated peripheral glucose uptake (20%). Importantly, however, the high dose of aspirin used in all of these studies is known to cause serious side effects. The current analysis directly evaluated whether a low dose of aspirin acceptable for long-term use in routine clinical practice is effective in reducing clinical type 2 diabetes. Although we found no evidence to support this approach, there are three main possibilities that could account for our null results. First, the dose of aspirin used in this study may be insufficient to impart a clinical benefit. As noted, prior studies evaluating the glucose metabolic effects of aspirin have used far higher doses. Data pertaining to potential hypoglycemic actions of low-dose aspirin have not been available. However, short-term treatment with doses as low as mg/day have been shown to improve several systemic inflammatory parameters, including lowering soluble CD40 ligand and promoting counterregulatory anti-inflammatory mediators such as 15-epi-lipoxin A 4 (11,12). Despite these latter findings, our data demonstrate that chronic treatment with 100 mg aspirin on alternate days does not prevent clinical diabetes. However, intermediate doses as high as 1.3 g/day have been used in long-term clinical trials of cardiovascular disease prevention, and these settings may offer additional opportunities to evaluate this issue if diabetes incidence was also ascertained. Second, women enrolled in the WHS were generally at low risk for diabetes as evidenced by the low prevalence of obesity, predominantly non-hispanic white ethnicity, and lower rates of clinical diabetes compared with similarly aged women in the overall U.S. population (25). It is possible that the use of aspirin in a higher risk group may have led to detection of a beneficial effect. However, in subgroup analyses of women with highrisk features we did not find strong support for differential effects. Furthermore, there was no benefit among women having evidence of inflammation as reflected by elevated hscrp. Third, underdiagnosis of diabetes may have influenced our results. Women did not undergo systematic screening for diabetes or glucose intolerance as a part of the study; thus, we detected clinical diabetes as ascertained during routine clinical practice rather than all women with biochemical evidence of disease. However, any misclassification due to unrecognized diabetes would be nondifferential between treatment arms and unlikely to lead to important alterations in the estimation of relative effects. In addition, we are reassured that reported diabetes screening rates among participants in our study were similar to contemporaneous screening rates in the general population and equivalent in the two treatment groups. Major strengths of this analysis include the randomized setting, large study 6 DIABETES CARE, VOLUME 32, NUMBER 1, JANUARY 2009
6 Pradhan and Associates Table 2 Incidence rates and rate ratio of type 2 diabetes in clinically important subgroups Aspirin group Placebo group Characteristic Sample size (n) No. events Events per 1,000 patient-years No. events Events per 1,000 patient-years RR (95% CI)* P interaction Age years 23, ( ) years 11, ( ) 65 years 3, ( ) BMI kg/m 2 19, ( ) kg/m 2 11, ( ) 30 kg/m 2 6, ( ) Family history of diabetes 0.68 No 29, ( ) Yes 9, ( ) Exercise once weekly 0.35 No 22, ( ) Yes 16, ( ) Hypertension 0.52 No 29, ( ) Yes 9, ( ) Hyperlipidemia 0.75 No 27, ( ) Yes 11, ( ) Menopause and HT 0.63 Premenopausal 10, ( ) Uncertain 6, ( ) Postmenopausal, HT 11, ( ) Postmenopausal, no HT 9, ( ) Total cholesterol mmol/l 11, ( ) mmol/l 10, ( ) 6.21 mmol/l 5, ( ) LDL cholesterol mmol/l 6, ( ) mmol/l 9, ( ) mmol/l 6, ( ) 4.13 mmol/l 3, ( ) HDL cholesterol mmol/l 11, ( ) 1.30 mmol/l 15, ( ) Total cholesterol to HDL cholesterol ratio , ( ) , ( ) 6.0 2, ( ) Non-HDL cholesterol mmol/l 7, ( ) mmol/l 8, ( ) 4.13 mmol/l 11, ( ) A1C % 13, ( ) 5.0% 13, ( ) hscrp mg/l 8, ( ) mg/l 9, ( ) 3.0 mg/l 9, ( ) Data are % unless otherwise indicated. Analyses involving biomarkers are restricted to women providing baseline blood specimens (n 27,167). *RR of clinical diabetes in the aspirin versus placebo group adjusted for age and randomized treatment assignment to vitamin E and -carotene. Family history of diabetes in a first-degree relative (mother, father, sister, or brother). Physical activity defined by number of episodes of vigorous physical activity per week. HT, hormone therapy. DIABETES CARE, VOLUME 32, NUMBER 1, JANUARY
7 Aspirin does not lower diabetes incidence population, long treatment duration, large number of events, and ability to examine several large high-risk subgroups of women. In addition, baseline blood specimens were provided by roughly 70% of women in whom hscrp and A1C levels were available. Importantly, systematic data were also collected on the occurrence of significant bleeding events. Limitations of this study have been alluded to previously. An important limitation is the lack of systematic screening for more sensitive measures of glucose intolerance and insulin resistance during follow-up. While the costs associated with such diagnostic testing are prohibitive in this large-scale setting, we cannot with the current data determine the potential impact of low-dose aspirin on these subclinical markers of incipient disease. In summary, aspirin at a dose of 100 mg on alternate days is not effective for the prevention of clinical type 2 diabetes among otherwise healthy women at generally low risk for this disease. Our data do not pertain to other salicylate agents currently being evaluated for diabetes treatment or to intermediate or high doses of long-term aspirin in primary prevention. However, even at the low dose evaluated in this trial, the use of aspirin was associated with a significant increase in clinically important bleeding events and any potential benefit at higher doses, if found, must take into account this potential for excess risk. Acknowledgments This work was supported by grants (HL and CA-47988) from the National Heart, Lung, and Blood Institute and the National Cancer Institute (Bethesda, MD). Aspirin and aspirin placebo were provided by Bayer HealthCare. Vitamin E and vitamin E placebo were provided by the Natural Source Vitamin E Association. No other potential conflicts of interest relevant to this article were reported. References 1. Ebstein W: Zur therapie des diabetes mellitus, insbesondere uber die anwendeng der salicylauren natron bei demselben. Berlin Klin Wochenschr 13: , Shoelson SE, Lee J, Goldfine AB: Inflammation and insulin resistance. J Clin Invest 116: , Thorand B, Baumert J, Kolb H, Meisinger C, Chambless L, Koenig W, Herder C: Sex differences in the prediction of type 2 diabetes by inflammatory markers: results from the MONICA/KORA Augsburg casecohort study, Diabetes Care 30: , Fleischman A, Shoelson SE, Bernier R, Goldfine AB: Salsalate improves glycemia and inflammatory parameters in obese young adults. Diabetes Care 31: , Yuan M, Konstantopoulos N, Lee J, Hansen L, Li ZW, Karin M, Shoelson SE: Reversal of obesity- and diet-induced insulin resistance with salicylates or targeted disruption of Ikk. Science 293: , Gao Z, Zuberi A, Quon MJ, Dong Z, Ye J: Aspirin inhibits serine phosphorylation of insulin receptor substrate 1 in tumor necrosis factor-treated cells through targeting multiple serine kinases. J Biol Chem 278: , Reid J, Lightbody TD: The insulin equivalence of salicylate. Br Med J 1: , Micossi P, Pontiroli AE, Baron SH, Tamayo RC, Lengel F, Bevilacqua M, Raggi U, Norbiato G, Foa PP: Aspirin stimulates insulin and glucagon secretion and increases glucose tolerance in normal and diabetic subjects. Diabetes 27: , Prince RL, Larkins RG, Alford FP: The effect of acetylsalicylic acid on plasma glucose and the response of glucose regulatory hormones to intravenous glucose and arginine in insulin treated diabetics and normal subjects. Metabolism 30: , Hundal RS, Petersen KF, Mayerson AB, Randhawa PS, Inzucchi S, Shoelson SE, Shulman GI: Mechanism by which highdose aspirin improves glucose metabolism in type 2 diabetes. J Clin Invest 109: , Chiang N, Bermudez EA, Ridker PM, Hurwitz S, Serhan CN: Aspirin triggers antiinflammatory 15-epi-lipoxin A4 and inhibits thromboxane in a randomized human trial. Proc Natl Acad Sci U S A 101: , Santilli F, Davi G, Consoli A, Cipollone F, Mezzetti A, Falco A, Taraborelli T, Devangelio E, Ciabattoni G, Basili S, Patrono C: Thromboxane-dependent CD40 ligand release in type 2 diabetes mellitus. JAm Coll Cardiol 47: , Ridker PM, Cook NR, Lee IM, Gordon D, Gaziano JM, Manson JE, Hennekens CH, Buring JE: A randomized trial of low-dose aspirin in the primary prevention of cardiovascular disease in women. N Engl J Med 352: , Cook NR, Lee IM, Gaziano JM, Gordon D, Ridker PM, Manson JE, Hennekens CH, Buring JE: Low-dose aspirin in the primary prevention of cancer: the Women s Health Study: a randomized controlled trial. JAMA 294:47 55, Lee IM, Cook NR, Gaziano JM, Gordon D, Ridker PM, Manson JE, Hennekens CH, Buring JE: Vitamin E in the primary prevention of cardiovascular disease and cancer: the Women s Health Study: a randomized controlled trial. JAMA 294: 56 65, Rexrode KM, Lee IM, Cook NR, Hennekens CH, Buring JE: Baseline characteristics of participants in the Women s Health Study. J Womens Health Gend Based Med 9:19 27, Liu S, Lee IM, Song Y, Van Denburgh M, Cook NR, Manson JE, Buring JE: Vitamin E and risk of type 2 diabetes in the Women s Health Study randomized controlled trial. Diabetes 55: , Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 20: , Ding EL, Song Y, Manson JE, Pradhan AD, Buring JE, Liu S: Accuracy of administrative coding for type 2 diabetes in children, adolescents, and young adults. Diabetes Care 30:e98, 2007; author reply e Ealovega MW, Tabaei BP, Brandle M, Burke R, Herman WH: Opportunistic screening for diabetes in routine clinical practice. Diabetes Care 27:9 12, Lee IM, Cook NR, Manson JE, Buring JE, Hennekens CH: -Carotene supplementation and incidence of cancer and cardiovascular disease: the Women s Health Study. J Natl Cancer Inst 91: , Giugliano D, Torella R, Siniscalchi N, Improta L, D Onofrio F: The effect of acetylsalicylic acid on insulin response to glucose and arginine in normal man. Diabetologia 14: , Newman WP, Brodows RG: Aspirin causes tissue insensitivity to insulin in normal man. J Clin Endocrinol Metab 57: , Bratusch-Marrain PR, Vierhapper H, Komjati M, Waldhausl WK: Acetyl-salicylic acid impairs insulin-mediated glucose utilization and reduces insulin clearance in healthy and non-insulin-dependent diabetic man. Diabetologia 28: , Centers for Disease Control and Prevention: National Diabetes Surveillance System: incidence of diabetes [article online], Available from diabetes/statistics/incidence/table5.htm. Accessed 21 May DIABETES CARE, VOLUME 32, NUMBER 1, JANUARY 2009
A Randomized Trial of Low-Dose Aspirin in the Prevention of Clinical Type 2 Diabetes in Women
 Diabetes Care Publish Ahead of Print, published online October 3, 2008 Low-Dose Aspirin Does Not Lower Diabetes Incidence in Women A Randomized Trial of Low-Dose Aspirin in the Prevention of Clinical Type
Diabetes Care Publish Ahead of Print, published online October 3, 2008 Low-Dose Aspirin Does Not Lower Diabetes Incidence in Women A Randomized Trial of Low-Dose Aspirin in the Prevention of Clinical Type
Dietary Fatty Acids and the Risk of Hypertension in Middle-Aged and Older Women
 07/14/2010 Dietary Fatty Acids and the Risk of Hypertension in Middle-Aged and Older Women First Author: Wang Short Title: Dietary Fatty Acids and Hypertension Risk in Women Lu Wang, MD, PhD, 1 JoAnn E.
07/14/2010 Dietary Fatty Acids and the Risk of Hypertension in Middle-Aged and Older Women First Author: Wang Short Title: Dietary Fatty Acids and Hypertension Risk in Women Lu Wang, MD, PhD, 1 JoAnn E.
The Framingham Coronary Heart Disease Risk Score
 Plasma Concentration of C-Reactive Protein and the Calculated Framingham Coronary Heart Disease Risk Score Michelle A. Albert, MD, MPH; Robert J. Glynn, PhD; Paul M Ridker, MD, MPH Background Although
Plasma Concentration of C-Reactive Protein and the Calculated Framingham Coronary Heart Disease Risk Score Michelle A. Albert, MD, MPH; Robert J. Glynn, PhD; Paul M Ridker, MD, MPH Background Although
A: Epidemiology update. Evidence that LDL-C and CRP identify different high-risk groups
 A: Epidemiology update Evidence that LDL-C and CRP identify different high-risk groups Women (n = 27,939; mean age 54.7 years) who were free of symptomatic cardiovascular (CV) disease at baseline were
A: Epidemiology update Evidence that LDL-C and CRP identify different high-risk groups Women (n = 27,939; mean age 54.7 years) who were free of symptomatic cardiovascular (CV) disease at baseline were
The Impact of Diabetes Mellitus and Prior Myocardial Infarction on Mortality From All Causes and From Coronary Heart Disease in Men
 Journal of the American College of Cardiology Vol. 40, No. 5, 2002 2002 by the American College of Cardiology Foundation ISSN 0735-1097/02/$22.00 Published by Elsevier Science Inc. PII S0735-1097(02)02044-2
Journal of the American College of Cardiology Vol. 40, No. 5, 2002 2002 by the American College of Cardiology Foundation ISSN 0735-1097/02/$22.00 Published by Elsevier Science Inc. PII S0735-1097(02)02044-2
Val-MARC: Valsartan-Managing Blood Pressure Aggressively and Evaluating Reductions in hs-crp
 Página 1 de 5 Return to Medscape coverage of: American Society of Hypertension 21st Annual Scientific Meeting and Exposition Val-MARC: Valsartan-Managing Blood Pressure Aggressively and Evaluating Reductions
Página 1 de 5 Return to Medscape coverage of: American Society of Hypertension 21st Annual Scientific Meeting and Exposition Val-MARC: Valsartan-Managing Blood Pressure Aggressively and Evaluating Reductions
journal of medicine The new england A Randomized Trial of Low-Dose Aspirin in the Primary Prevention of Cardiovascular Disease in Women abstract
 The new england journal of medicine established in 1812 march 31, 2005 vol. 352 no. 13 A Randomized Trial of Low-Dose in the Primary Prevention of Cardiovascular Disease in Women Paul M Ridker, M.D., Nancy
The new england journal of medicine established in 1812 march 31, 2005 vol. 352 no. 13 A Randomized Trial of Low-Dose in the Primary Prevention of Cardiovascular Disease in Women Paul M Ridker, M.D., Nancy
JUPITER NEJM Poll. Panel Discussion: Literature that Should Have an Impact on our Practice: The JUPITER Study
 Panel Discussion: Literature that Should Have an Impact on our Practice: The Study Kaiser COAST 11 th Annual Conference Maui, August 2009 Robert Blumberg, MD, FACC Ralph Brindis, MD, MPH, FACC Primary
Panel Discussion: Literature that Should Have an Impact on our Practice: The Study Kaiser COAST 11 th Annual Conference Maui, August 2009 Robert Blumberg, MD, FACC Ralph Brindis, MD, MPH, FACC Primary
Misperceptions still exist that cardiovascular disease is not a real problem for women.
 Management of Cardiovascular Risk Factors in the Cynthia A., MD University of California, San Diego ARHP 9/19/08 Disclosures Research support Wyeth, Lilly, Organon, Novo Nordisk, Pfizer Consultant fees
Management of Cardiovascular Risk Factors in the Cynthia A., MD University of California, San Diego ARHP 9/19/08 Disclosures Research support Wyeth, Lilly, Organon, Novo Nordisk, Pfizer Consultant fees
Egg Consumption and Risk of Type 2 Diabetes in Men and Women
 Egg Consumption and Risk of Type 2 Diabetes in Men and The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published Version
Egg Consumption and Risk of Type 2 Diabetes in Men and The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published Version
Headache, migraine and risk of brain tumors in women: prospective cohort study
 Kurth et al. The Journal of Headache and Pain (2015) 16:17 DOI 10.1186/s10194-015-0501-0 RESEARCH ARTICLE Open Access Headache, migraine and risk of brain tumors in women: prospective cohort study Tobias
Kurth et al. The Journal of Headache and Pain (2015) 16:17 DOI 10.1186/s10194-015-0501-0 RESEARCH ARTICLE Open Access Headache, migraine and risk of brain tumors in women: prospective cohort study Tobias
Normal Fasting Plasma Glucose and Risk of Type 2 Diabetes Diagnosis
 CLINICAL RESEARCH STUDY Normal Fasting Plasma Glucose and Risk of Type 2 Diabetes Diagnosis Gregory A. Nichols, PhD, Teresa A. Hillier, MD, MS, Jonathan B. Brown, PhD, MPP Center for Health Research, Kaiser
CLINICAL RESEARCH STUDY Normal Fasting Plasma Glucose and Risk of Type 2 Diabetes Diagnosis Gregory A. Nichols, PhD, Teresa A. Hillier, MD, MS, Jonathan B. Brown, PhD, MPP Center for Health Research, Kaiser
journal of medicine The new england Rosuvastatin to Prevent Vascular Events in Men and Women with Elevated C-Reactive Protein Abstract
 The new england journal of medicine established in 1812 november 20, 2008 vol. 359 no. 21 to Prevent Vascular Events in Men and Women with Elevated C-Reactive Protein Paul M Ridker, M.D., Eleanor Danielson,
The new england journal of medicine established in 1812 november 20, 2008 vol. 359 no. 21 to Prevent Vascular Events in Men and Women with Elevated C-Reactive Protein Paul M Ridker, M.D., Eleanor Danielson,
EPIDEMIOLOGY AND BIOSTATISTICS. Aspirin Use and Risk of Cataract in Posttrial Follow-up of Physicians Health Study I
 EPIDEMIOLOGY AND BIOSTATISTICS Aspirin Use and Risk of Cataract in Posttrial Follow-up of Physicians Health Study I William G. Christen, ScD; Umed A. Ajani, MBBS; Debra A. Schaumberg, ScD; Robert J. Glynn,
EPIDEMIOLOGY AND BIOSTATISTICS Aspirin Use and Risk of Cataract in Posttrial Follow-up of Physicians Health Study I William G. Christen, ScD; Umed A. Ajani, MBBS; Debra A. Schaumberg, ScD; Robert J. Glynn,
John J.P. Kastelein MD PhD Professor of Medicine Dept. of Vascular Medicine Academic Medial Center / University of Amsterdam
 Latest Insights from the JUPITER Study John J.P. Kastelein MD PhD Professor of Medicine Dept. of Vascular Medicine Academic Medial Center / University of Amsterdam Inflammation, hscrp, and Vascular Prevention
Latest Insights from the JUPITER Study John J.P. Kastelein MD PhD Professor of Medicine Dept. of Vascular Medicine Academic Medial Center / University of Amsterdam Inflammation, hscrp, and Vascular Prevention
A Randomized Trial of a Multivitamin (MVM) in the Prevention of Cardiovascular Disease in Men: The Physicians Health Study (PHS) II
 A Randomized Trial of a Multivitamin (MVM) in the Prevention of Cardiovascular Disease in Men: The Physicians Health Study (PHS) II Presenter Disclosure Information Howard D. Sesso, ScD, MPH Relevant Disclosures:
A Randomized Trial of a Multivitamin (MVM) in the Prevention of Cardiovascular Disease in Men: The Physicians Health Study (PHS) II Presenter Disclosure Information Howard D. Sesso, ScD, MPH Relevant Disclosures:
Elevated Risk of Cardiovascular Disease Prior to Clinical Diagnosis of Type 2 Diabetes
 Epidemiology/Health Services/Psychosocial Research O R I G I N A L A R T I C L E Elevated Risk of Cardiovascular Disease Prior to Clinical Diagnosis of Type 2 Diabetes FRANK B. HU, MD 1,2,3 MEIR J. STAMPFER,
Epidemiology/Health Services/Psychosocial Research O R I G I N A L A R T I C L E Elevated Risk of Cardiovascular Disease Prior to Clinical Diagnosis of Type 2 Diabetes FRANK B. HU, MD 1,2,3 MEIR J. STAMPFER,
SESSION 3 11 AM 12:30 PM
 SESSION 3 11 AM 12:30 PM for the Primary Prevention of Cardiovascular Disease: A Personalized Approach SPEAKER Samia Mora MD, MHS Presenter Disclosure Information The following relationships exist related
SESSION 3 11 AM 12:30 PM for the Primary Prevention of Cardiovascular Disease: A Personalized Approach SPEAKER Samia Mora MD, MHS Presenter Disclosure Information The following relationships exist related
Antihypertensive Trial Design ALLHAT
 1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
Rotating night shift work and risk of psoriasis in US women
 Rotating night shift work and risk of psoriasis in US women The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published
Rotating night shift work and risk of psoriasis in US women The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published
ORIGINAL INVESTIGATION. Self-Selected Posttrial Aspirin Use and Subsequent Cardiovascular Disease and Mortality in the Physicians Health Study
 ORIGINAL INVESTIGATION Self-Selected Posttrial Aspirin Use and Subsequent Cardiovascular Disease and Mortality in the Physicians Health Study Nancy R. Cook, ScD; Patricia R. Hebert, PhD; JoAnn E. Manson,
ORIGINAL INVESTIGATION Self-Selected Posttrial Aspirin Use and Subsequent Cardiovascular Disease and Mortality in the Physicians Health Study Nancy R. Cook, ScD; Patricia R. Hebert, PhD; JoAnn E. Manson,
Lipoprotein Particle Size and Concentration by Nuclear Magnetic Resonance and Incident Type 2 Diabetes in Women
 Diabetes Publish Ahead of Print, published online February 25, 2010 Lipoprotein Particle Size and Concentration by Nuclear Magnetic Resonance and Incident Type 2 Diabetes in Women Samia Mora, MD, MHS,
Diabetes Publish Ahead of Print, published online February 25, 2010 Lipoprotein Particle Size and Concentration by Nuclear Magnetic Resonance and Incident Type 2 Diabetes in Women Samia Mora, MD, MHS,
Patients with the metabolic syndrome are at increased risk
 Clinical Investigation and Reports C-Reactive Protein, the Metabolic Syndrome, and Risk of Incident Cardiovascular Events An 8-Year Follow-Up of 14 719 Initially Healthy American Women Paul M Ridker, MD;
Clinical Investigation and Reports C-Reactive Protein, the Metabolic Syndrome, and Risk of Incident Cardiovascular Events An 8-Year Follow-Up of 14 719 Initially Healthy American Women Paul M Ridker, MD;
Aspirin for the Prevention of Cardiovascular Disease
 Detail-Document #250601 This Detail-Document accompanies the related article published in PHARMACIST S LETTER / PRESCRIBER S LETTER June 2009 ~ Volume 25 ~ Number 250601 Aspirin for the Prevention of Cardiovascular
Detail-Document #250601 This Detail-Document accompanies the related article published in PHARMACIST S LETTER / PRESCRIBER S LETTER June 2009 ~ Volume 25 ~ Number 250601 Aspirin for the Prevention of Cardiovascular
C-REACTIVE PROTEIN AND LDL CHOLESTEROL FOR PREDICTING CARDIOVASCULAR EVENTS
 COMPARISON OF C-REACTIVE PROTEIN AND LOW-DENSITY LIPOPROTEIN CHOLESTEROL LEVELS IN THE PREDICTION OF FIRST CARDIOVASCULAR EVENTS PAUL M. RIDKER, M.D., NADER RIFAI, PH.D., LYNDA ROSE, M.S., JULIE E. BURING,
COMPARISON OF C-REACTIVE PROTEIN AND LOW-DENSITY LIPOPROTEIN CHOLESTEROL LEVELS IN THE PREDICTION OF FIRST CARDIOVASCULAR EVENTS PAUL M. RIDKER, M.D., NADER RIFAI, PH.D., LYNDA ROSE, M.S., JULIE E. BURING,
Disclosures. Diabetes and Cardiovascular Risk Management. Learning Objectives. Atherosclerotic Cardiovascular Disease
 Disclosures Diabetes and Cardiovascular Risk Management Tony Hampton, MD, MBA Medical Director Advocate Aurora Operating System Advocate Aurora Healthcare Downers Grove, IL No conflicts or disclosures
Disclosures Diabetes and Cardiovascular Risk Management Tony Hampton, MD, MBA Medical Director Advocate Aurora Operating System Advocate Aurora Healthcare Downers Grove, IL No conflicts or disclosures
Carlo Patrono, MD, FESC. New York, 8 th December Catholic University School of Medicine, Rome, Italy. New York Cardiovascular Symposium
 Aspirin in Primary and Secondary Cardiovascular Disease Prevention. Still Four Questions: About Enteric-Coated, Indicated Doses, Use in Diabetes, Use in PVD Carlo Patrono, MD, FESC Catholic University
Aspirin in Primary and Secondary Cardiovascular Disease Prevention. Still Four Questions: About Enteric-Coated, Indicated Doses, Use in Diabetes, Use in PVD Carlo Patrono, MD, FESC Catholic University
Epidemiological studies indicate that a parental or family
 Maternal and Paternal History of Myocardial Infarction and Risk of Cardiovascular Disease in Men and Women Howard D. Sesso, ScD, MPH; I-Min Lee, MBBS, ScD; J. Michael Gaziano, MD, MPH; Kathryn M. Rexrode,
Maternal and Paternal History of Myocardial Infarction and Risk of Cardiovascular Disease in Men and Women Howard D. Sesso, ScD, MPH; I-Min Lee, MBBS, ScD; J. Michael Gaziano, MD, MPH; Kathryn M. Rexrode,
9/29/2015. Primary Prevention of Heart Disease: Objectives. Objectives. What works? What doesn t?
 Primary Prevention of Heart Disease: What works? What doesn t? Samia Mora, MD, MHS Associate Professor, Harvard Medical School Associate Physician, Brigham and Women s Hospital October 2, 2015 Financial
Primary Prevention of Heart Disease: What works? What doesn t? Samia Mora, MD, MHS Associate Professor, Harvard Medical School Associate Physician, Brigham and Women s Hospital October 2, 2015 Financial
A Proposed Randomized Trial of Cocoa Flavanols and Multivitamins in the Prevention of Cardiovascular Disease and Cancer
 A Proposed Randomized Trial of Cocoa Flavanols and Multivitamins in the Prevention of Cardiovascular Disease and Cancer JoAnn E. Manson, MD, DrPH Howard D. Sesso, ScD, MPH Brigham and Women's Hospital
A Proposed Randomized Trial of Cocoa Flavanols and Multivitamins in the Prevention of Cardiovascular Disease and Cancer JoAnn E. Manson, MD, DrPH Howard D. Sesso, ScD, MPH Brigham and Women's Hospital
Aspirin prevents stroke but not MI in women; Vitamin E has no effect on CV disease or cancer
 MEDICAL GRAND ROUNDS CME CREDIT JULIE E. BURING, ScD* Professor of Medicine, Harvard Medical School; Division of Preventive Medicine, Brigham and Women s Hospital, Boston; principal investigator, Women
MEDICAL GRAND ROUNDS CME CREDIT JULIE E. BURING, ScD* Professor of Medicine, Harvard Medical School; Division of Preventive Medicine, Brigham and Women s Hospital, Boston; principal investigator, Women
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Online Supplementary Material
 Section 1. Adapted Newcastle-Ottawa Scale The adaptation consisted of allowing case-control studies to earn a star when the case definition is based on record linkage, to liken the evaluation of case-control
Section 1. Adapted Newcastle-Ottawa Scale The adaptation consisted of allowing case-control studies to earn a star when the case definition is based on record linkage, to liken the evaluation of case-control
Experimental Design. Terminology. Chusak Okascharoen, MD, PhD September 19 th, Experimental study Clinical trial Randomized controlled trial
 Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
The Metabolic Syndrome: Is It A Valid Concept? YES
 The Metabolic Syndrome: Is It A Valid Concept? YES Congress on Diabetes and Cardiometabolic Health Boston, MA April 23, 2013 Edward S Horton, MD Joslin Diabetes Center Harvard Medical School Boston, MA
The Metabolic Syndrome: Is It A Valid Concept? YES Congress on Diabetes and Cardiometabolic Health Boston, MA April 23, 2013 Edward S Horton, MD Joslin Diabetes Center Harvard Medical School Boston, MA
Experiences with interim trial monitoring, particularly with early stopped trials
 Experiences with interim trial monitoring, particularly with early stopped trials 1 Robert J Glynn, ScD Divisions of Preventive Medicine and Pharmacoepidemiology & Pharmacoeconomics, Brigham & Women s
Experiences with interim trial monitoring, particularly with early stopped trials 1 Robert J Glynn, ScD Divisions of Preventive Medicine and Pharmacoepidemiology & Pharmacoeconomics, Brigham & Women s
Contents. Version 1.0: 01/02/2010 Protocol# ISRCTN Page 1 of 7
 Contents 1. INTRODUCTION... 2 2. STUDY SYNOPSIS... 2 3. STUDY OBJECTIVES... 2 3.1. Primary Objective... 2 3.2. Secondary Objectives... 2 3.3. Assessment of Objectives... 3 3.4. Change the Primary Objective
Contents 1. INTRODUCTION... 2 2. STUDY SYNOPSIS... 2 3. STUDY OBJECTIVES... 2 3.1. Primary Objective... 2 3.2. Secondary Objectives... 2 3.3. Assessment of Objectives... 3 3.4. Change the Primary Objective
SESSION 5 2:20 3:35 PM
 SESSION 5 2:20 3:35 PM for the Primary Prevention of Cardiovascular Disease: A Personalized Approach SPEAKER Samia Mora MD, MHS Presenter Disclosure Information The following relationships exist related
SESSION 5 2:20 3:35 PM for the Primary Prevention of Cardiovascular Disease: A Personalized Approach SPEAKER Samia Mora MD, MHS Presenter Disclosure Information The following relationships exist related
The COSMOS Trial. (COcoa Supplement and Multivitamins Outcomes Study) JoAnn E. Manson, MD, DrPH Howard D. Sesso, ScD, MPH
 COSMOS Trial The COSMOS Trial (COcoa Supplement and Multivitamins Outcomes Study) JoAnn E. Manson, MD, DrPH Howard D. Sesso, ScD, MPH Brigham and Women's Hospital Harvard Medical School Garnet L. Anderson,
COSMOS Trial The COSMOS Trial (COcoa Supplement and Multivitamins Outcomes Study) JoAnn E. Manson, MD, DrPH Howard D. Sesso, ScD, MPH Brigham and Women's Hospital Harvard Medical School Garnet L. Anderson,
ORIGINAL INVESTIGATION. Alcohol Consumption and Mortality in Men With Preexisting Cerebrovascular Disease
 ORIGINAL INVESTIGATION Alcohol Consumption and Mortality in Men With Preexisting Cerebrovascular Disease Vicki A. Jackson, MD; Howard D. Sesso, ScD; Julie E. Buring, ScD; J. Michael Gaziano, MD Background:
ORIGINAL INVESTIGATION Alcohol Consumption and Mortality in Men With Preexisting Cerebrovascular Disease Vicki A. Jackson, MD; Howard D. Sesso, ScD; Julie E. Buring, ScD; J. Michael Gaziano, MD Background:
The JUPITER trial: What does it tell us? Alice Y.Y. Cheng, MD, FRCPC January 24, 2009
 The JUPITER trial: What does it tell us? Alice Y.Y. Cheng, MD, FRCPC January 24, 2009 Learning Objectives 1. Understand the role of statin therapy in the primary and secondary prevention of stroke 2. Explain
The JUPITER trial: What does it tell us? Alice Y.Y. Cheng, MD, FRCPC January 24, 2009 Learning Objectives 1. Understand the role of statin therapy in the primary and secondary prevention of stroke 2. Explain
Of the 1.5 million heart attacks
 CARDIOLOGY PATIENT PAGE CARDIOLOGY PATIENT PAGE C-Reactive Protein A Simple Test to Help Predict Risk of Heart Attack and Stroke Paul M Ridker, MD, MPH Of the 1.5 million heart attacks and 600 000 strokes
CARDIOLOGY PATIENT PAGE CARDIOLOGY PATIENT PAGE C-Reactive Protein A Simple Test to Help Predict Risk of Heart Attack and Stroke Paul M Ridker, MD, MPH Of the 1.5 million heart attacks and 600 000 strokes
AGROWING BODY OF LITERAture
 ORIGINAL CONTRIBUTION JAMA-EXPRESS Low-Dose Aspirin in the Primary Prevention of Cancer The Women s Health Study: A Randomized Controlled Trial Nancy R. Cook, ScD I-Min Lee, MBBS, ScD J. Michael Gaziano,
ORIGINAL CONTRIBUTION JAMA-EXPRESS Low-Dose Aspirin in the Primary Prevention of Cancer The Women s Health Study: A Randomized Controlled Trial Nancy R. Cook, ScD I-Min Lee, MBBS, ScD J. Michael Gaziano,
ORIGINAL INVESTIGATION. C-Reactive Protein Concentration and Incident Hypertension in Young Adults
 ORIGINAL INVESTIGATION C-Reactive Protein Concentration and Incident Hypertension in Young Adults The CARDIA Study Susan G. Lakoski, MD, MS; David M. Herrington, MD, MHS; David M. Siscovick, MD, MPH; Stephen
ORIGINAL INVESTIGATION C-Reactive Protein Concentration and Incident Hypertension in Young Adults The CARDIA Study Susan G. Lakoski, MD, MS; David M. Herrington, MD, MHS; David M. Siscovick, MD, MPH; Stephen
Hypertension in 2015: SPRINT-ing ahead of JNC-8. MAJ Charles Magee, MD MPH FACP Director, WRNMMC Hypertension Clinic
 Hypertension in 2015: SPRINT-ing ahead of JNC-8 MAJ Charles Magee, MD MPH FACP Director, WRNMMC Hypertension Clinic Conflits of interest? None Disclaimer The opinions contained herein are not to be considered
Hypertension in 2015: SPRINT-ing ahead of JNC-8 MAJ Charles Magee, MD MPH FACP Director, WRNMMC Hypertension Clinic Conflits of interest? None Disclaimer The opinions contained herein are not to be considered
(n=6279). Continuous variables are reported as mean with 95% confidence interval and T1 T2 T3. Number of subjects
 Table 1. Distribution of baseline characteristics across tertiles of OPG adjusted for age and sex (n=6279). Continuous variables are reported as mean with 95% confidence interval and categorical values
Table 1. Distribution of baseline characteristics across tertiles of OPG adjusted for age and sex (n=6279). Continuous variables are reported as mean with 95% confidence interval and categorical values
Ten Year Risk for CVD Event by Systolic HTN and CVD Risk Factors (Where s Age?)
 Prevention and Treatment of CVD in Older Patients with Diabetes and Pre Diabetes: Hypertension and Dyslipidemia ASP Workshop on Diabetes Mellitus and Cardiovascular Disease in Older Adults Pentagon City
Prevention and Treatment of CVD in Older Patients with Diabetes and Pre Diabetes: Hypertension and Dyslipidemia ASP Workshop on Diabetes Mellitus and Cardiovascular Disease in Older Adults Pentagon City
THE C-REACTIVE PROTEIN IS A
 ORIGINAL CONTRIBUTION C-Reactive Protein and the Risk of Developing Hypertension Howard D. Sesso, ScD, MPH Julie E. Buring, ScD Nader Rifai, PhD Gavin J. Blake, MD, MPH J. Michael Gaziano, MD, MPH Paul
ORIGINAL CONTRIBUTION C-Reactive Protein and the Risk of Developing Hypertension Howard D. Sesso, ScD, MPH Julie E. Buring, ScD Nader Rifai, PhD Gavin J. Blake, MD, MPH J. Michael Gaziano, MD, MPH Paul
New evidences in heart failure: the GISSI-HF trial. Aldo P Maggioni, MD ANMCO Research Center Firenze, Italy
 New evidences in heart failure: the GISSI-HF trial Aldo P Maggioni, MD ANMCO Research Center Firenze, Italy % Improving survival in chronic HF and LV systolic dysfunction: 1 year all-cause mortality 20
New evidences in heart failure: the GISSI-HF trial Aldo P Maggioni, MD ANMCO Research Center Firenze, Italy % Improving survival in chronic HF and LV systolic dysfunction: 1 year all-cause mortality 20
ORIGINAL INVESTIGATION. C-Reactive Protein in the Prediction of Rheumatoid Arthritis in Women
 ORIGINAL INVESTIGATION C-Reactive Protein in the Prediction of Rheumatoid Arthritis in Women Nancy A. Shadick, MD, MPH; Nancy R. Cook, ScD; Elizabeth W. Karlson, MD; Paul M Ridker, MD, MPH; Nancy E. Maher,
ORIGINAL INVESTIGATION C-Reactive Protein in the Prediction of Rheumatoid Arthritis in Women Nancy A. Shadick, MD, MPH; Nancy R. Cook, ScD; Elizabeth W. Karlson, MD; Paul M Ridker, MD, MPH; Nancy E. Maher,
Andrew Cohen, MD and Neil S. Skolnik, MD INTRODUCTION
 2 Hyperlipidemia Andrew Cohen, MD and Neil S. Skolnik, MD CONTENTS INTRODUCTION RISK CATEGORIES AND TARGET LDL-CHOLESTEROL TREATMENT OF LDL-CHOLESTEROL SPECIAL CONSIDERATIONS OLDER AND YOUNGER ADULTS ADDITIONAL
2 Hyperlipidemia Andrew Cohen, MD and Neil S. Skolnik, MD CONTENTS INTRODUCTION RISK CATEGORIES AND TARGET LDL-CHOLESTEROL TREATMENT OF LDL-CHOLESTEROL SPECIAL CONSIDERATIONS OLDER AND YOUNGER ADULTS ADDITIONAL
Factorial Study Design 07/18/12
 Disclaimer: The following information is fictional and is only intended for the purposes of illustrating key concepts for results data entry in the Protocol Registration System (PRS). Example Factorial
Disclaimer: The following information is fictional and is only intended for the purposes of illustrating key concepts for results data entry in the Protocol Registration System (PRS). Example Factorial
Randomised aspirin assignment and risk of adultonset asthma in the Women s Health Study
 Thorax Online First, published on March 13, 2008 as 10.1136/thx.2007.091447 Asthma 1 Division of Preventive Medicine, Department of Women s Hospital, Harvard Medical School, Boston, MA, USA; 2 Division
Thorax Online First, published on March 13, 2008 as 10.1136/thx.2007.091447 Asthma 1 Division of Preventive Medicine, Department of Women s Hospital, Harvard Medical School, Boston, MA, USA; 2 Division
The American Diabetes Association estimates
 DYSLIPIDEMIA, PREDIABETES, AND TYPE 2 DIABETES: CLINICAL IMPLICATIONS OF THE VA-HIT SUBANALYSIS Frank M. Sacks, MD* ABSTRACT The most serious and common complication in adults with diabetes is cardiovascular
DYSLIPIDEMIA, PREDIABETES, AND TYPE 2 DIABETES: CLINICAL IMPLICATIONS OF THE VA-HIT SUBANALYSIS Frank M. Sacks, MD* ABSTRACT The most serious and common complication in adults with diabetes is cardiovascular
Blood pressure and risk of developing type 2 diabetes mellitus: The Women s Health Study
 European Heart Journal (2007) 28, 2937 2943 doi:10.1093/eurheartj/ehm400 Clinical research Hypertension Blood pressure and risk of developing type 2 diabetes mellitus: The Women s Health Study David Conen
European Heart Journal (2007) 28, 2937 2943 doi:10.1093/eurheartj/ehm400 Clinical research Hypertension Blood pressure and risk of developing type 2 diabetes mellitus: The Women s Health Study David Conen
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and
Diabetes and the Heart
 Diabetes and the Heart Jeffrey Boord, MD, MPH Advances in Cardiovascular Medicine Kingston, Jamaica December 6, 2012 Outline Screening for diabetes in patients with CAD Screening for CAD in patients with
Diabetes and the Heart Jeffrey Boord, MD, MPH Advances in Cardiovascular Medicine Kingston, Jamaica December 6, 2012 Outline Screening for diabetes in patients with CAD Screening for CAD in patients with
Eugene Barrett M.D., Ph.D. University of Virginia 6/18/2007. Diagnosis and what is it Glucose Tolerance Categories FPG
 Diabetes Mellitus: Update 7 What is the unifying basis of this vascular disease? Eugene J. Barrett, MD, PhD Professor of Internal Medicine and Pediatrics Director, Diabetes Center and GCRC Health System
Diabetes Mellitus: Update 7 What is the unifying basis of this vascular disease? Eugene J. Barrett, MD, PhD Professor of Internal Medicine and Pediatrics Director, Diabetes Center and GCRC Health System
Aspirin Use and Risk of Atrial Fibrillation in the Physicians' Health Study
 Aspirin Use and Risk of Atrial Fibrillation in the Physicians' Health Study The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation
Aspirin Use and Risk of Atrial Fibrillation in the Physicians' Health Study The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation
Low HDL and Diabetic Dyslipidemia
 The Lowdown: Low HDL and Diabetic Dyslipidemia Patients with diabetes commonly have a low-density lipoprotein cholesterol (LDL-C) no higher than that of the general population. What treatment is warranted
The Lowdown: Low HDL and Diabetic Dyslipidemia Patients with diabetes commonly have a low-density lipoprotein cholesterol (LDL-C) no higher than that of the general population. What treatment is warranted
Folate, vitamin B 6, and vitamin B 12 are cofactors in
 Research Letters Dietary Folate and Vitamin B 6 and B 12 Intake in Relation to Mortality From Cardiovascular Diseases Japan Collaborative Cohort Study Renzhe Cui, MD; Hiroyasu Iso, MD; Chigusa Date, MD;
Research Letters Dietary Folate and Vitamin B 6 and B 12 Intake in Relation to Mortality From Cardiovascular Diseases Japan Collaborative Cohort Study Renzhe Cui, MD; Hiroyasu Iso, MD; Chigusa Date, MD;
Inflammation plays a major role in atherosclerosis, 1 and
 Soluble P-Selectin and the Risk of Future Cardiovascular Events Paul M. Ridker, MD; Julie E. Buring, ScD; Nader Rifai, PhD Background P-selectin, a cell-surface adhesion molecule involved in leukocyte
Soluble P-Selectin and the Risk of Future Cardiovascular Events Paul M. Ridker, MD; Julie E. Buring, ScD; Nader Rifai, PhD Background P-selectin, a cell-surface adhesion molecule involved in leukocyte
New therapeutic targets for T2DM
 New therapeutic targets for T2DM Targeting inflammation: NF- B, salsalate Gwanpyo Koh Department of Internal Medicine Jeju National University School of Medicine Introduction Obesity is occurring at epidemic
New therapeutic targets for T2DM Targeting inflammation: NF- B, salsalate Gwanpyo Koh Department of Internal Medicine Jeju National University School of Medicine Introduction Obesity is occurring at epidemic
Diabetes: Staying Two Steps Ahead. The prevalence of diabetes is increasing. What causes Type 2 diabetes?
 Focus on CME at the University of University Manitoba of Manitoba : Staying Two Steps Ahead By Shagufta Khan, MD; and Liam J. Murphy, MD The prevalence of diabetes is increasing worldwide and will double
Focus on CME at the University of University Manitoba of Manitoba : Staying Two Steps Ahead By Shagufta Khan, MD; and Liam J. Murphy, MD The prevalence of diabetes is increasing worldwide and will double
 PATCH Analysis Plan v1.2.doc Prophylactic Antibiotics for the Treatment of Cellulitis at Home: PATCH Analysis Plan for PATCH I and PATCH II Authors: Angela Crook, Andrew Nunn, James Mason and Kim Thomas,
PATCH Analysis Plan v1.2.doc Prophylactic Antibiotics for the Treatment of Cellulitis at Home: PATCH Analysis Plan for PATCH I and PATCH II Authors: Angela Crook, Andrew Nunn, James Mason and Kim Thomas,
MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebocontrolled
 Articles MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebocontrolled trial Heart Protection Study Collaborative Group* Summary Background
Articles MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebocontrolled trial Heart Protection Study Collaborative Group* Summary Background
Effect of the PCSK9 Inhibitor Evolocumab on Cardiovascular Outcomes
 Effect of the PCSK9 Inhibitor Evolocumab on Cardiovascular Outcomes MS Sabatine, RP Giugliano, SD Wiviott, FJ Raal, CM Ballantyne, R Somaratne, J Legg, SM Wasserman, R Scott, MJ Koren, and EA Stein for
Effect of the PCSK9 Inhibitor Evolocumab on Cardiovascular Outcomes MS Sabatine, RP Giugliano, SD Wiviott, FJ Raal, CM Ballantyne, R Somaratne, J Legg, SM Wasserman, R Scott, MJ Koren, and EA Stein for
Atherosclerotic Disease Risk Score
 Atherosclerotic Disease Risk Score Kavita Sharma, MD, FACC Diplomate, American Board of Clinical Lipidology Director of Prevention, Cardiac Rehabilitation and the Lipid Management Clinics September 16,
Atherosclerotic Disease Risk Score Kavita Sharma, MD, FACC Diplomate, American Board of Clinical Lipidology Director of Prevention, Cardiac Rehabilitation and the Lipid Management Clinics September 16,
Effects of whole grain intake on weight changes, diabetes, and cardiovascular Disease
 Effects of whole grain intake on weight changes, diabetes, and cardiovascular Disease Simin Liu, MD, ScD Professor of Epidemiology and Medicine Director, Center for Global Cardiometabolic Health Brown
Effects of whole grain intake on weight changes, diabetes, and cardiovascular Disease Simin Liu, MD, ScD Professor of Epidemiology and Medicine Director, Center for Global Cardiometabolic Health Brown
rosuvastatin, 5mg, 10mg, 20mg, film-coated tablets (Crestor ) SMC No. (725/11) AstraZeneca UK Ltd.
 rosuvastatin, 5mg, 10mg, 20mg, film-coated tablets (Crestor ) SMC No. (725/11) AstraZeneca UK Ltd. 09 September 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
rosuvastatin, 5mg, 10mg, 20mg, film-coated tablets (Crestor ) SMC No. (725/11) AstraZeneca UK Ltd. 09 September 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Diabetes. Ref HSCW 024
 Diabetes Ref HSCW 024 Why is it important? Diabetes is an increasingly common, life-long, progressive but largely preventable health condition affecting children and adults, causing a heavy burden on health
Diabetes Ref HSCW 024 Why is it important? Diabetes is an increasingly common, life-long, progressive but largely preventable health condition affecting children and adults, causing a heavy burden on health
Diabetes and Lipid Screening Among Patients in Primary Care: A Cohort Study
 Diabetes and Lipid Screening Among Patients in Primary Care: A Cohort Study The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters.
Diabetes and Lipid Screening Among Patients in Primary Care: A Cohort Study The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters.
Treatment of Cardiovascular Risk Factors. Kevin M Hayes D.O. F.A.C.C. First Coast Heart and Vascular Center
 Treatment of Cardiovascular Risk Factors Kevin M Hayes D.O. F.A.C.C. First Coast Heart and Vascular Center Disclosures: None Objectives What do risk factors tell us What to check and when Does treatment
Treatment of Cardiovascular Risk Factors Kevin M Hayes D.O. F.A.C.C. First Coast Heart and Vascular Center Disclosures: None Objectives What do risk factors tell us What to check and when Does treatment
Type 2 Diabetes Mellitus in Adolescents PHIL ZEITLER MD, PHD SECTION OF ENDOCRINOLOGY DEPARTMENT OF PEDIATRICS UNIVERSITY OF COLORADO DENVER
 Type 2 Diabetes Mellitus in Adolescents PHIL ZEITLER MD, PHD SECTION OF ENDOCRINOLOGY DEPARTMENT OF PEDIATRICS UNIVERSITY OF COLORADO DENVER Yes! Is Type 2 diabetes the same in kids as in adults? And No!
Type 2 Diabetes Mellitus in Adolescents PHIL ZEITLER MD, PHD SECTION OF ENDOCRINOLOGY DEPARTMENT OF PEDIATRICS UNIVERSITY OF COLORADO DENVER Yes! Is Type 2 diabetes the same in kids as in adults? And No!
Early-Adulthood Cardiovascular Disease Risk Factor Profiles Among Individuals With and Without Diabetes in the Framingham Heart Study
 Early-Adulthood Cardiovascular Disease Risk Factor Profiles Among Individuals With and Without Diabetes in the Framingham Heart Study The Harvard community has made this article openly available. Please
Early-Adulthood Cardiovascular Disease Risk Factor Profiles Among Individuals With and Without Diabetes in the Framingham Heart Study The Harvard community has made this article openly available. Please
Moderate alcohol consumption is associated with decreased
 Alcohol Consumption and Plasma Concentration of C-Reactive Protein Michelle A. Albert, MD, MPH; Robert J. Glynn, PhD; Paul M Ridker, MD, MPH Background Moderate alcohol intake has been associated with
Alcohol Consumption and Plasma Concentration of C-Reactive Protein Michelle A. Albert, MD, MPH; Robert J. Glynn, PhD; Paul M Ridker, MD, MPH Background Moderate alcohol intake has been associated with
Research Article Comparison of Different Anthropometric Measurements and Inflammatory Biomarkers
 International Inflammation Volume 2012, Article ID 124693, 5 pages doi:10.1155/2012/124693 Research Article Comparison of Different Anthropometric Measurements and Inflammatory Biomarkers Yaron Arbel,
International Inflammation Volume 2012, Article ID 124693, 5 pages doi:10.1155/2012/124693 Research Article Comparison of Different Anthropometric Measurements and Inflammatory Biomarkers Yaron Arbel,
Kathryn M. Rexrode, MD, MPH. Assistant Professor. Division of Preventive Medicine Brigham and Women s s Hospital Harvard Medical School
 Update: Hormones and Cardiovascular Disease in Women Kathryn M. Rexrode, MD, MPH Assistant Professor Division of Preventive Medicine Brigham and Women s s Hospital Harvard Medical School Overview Review
Update: Hormones and Cardiovascular Disease in Women Kathryn M. Rexrode, MD, MPH Assistant Professor Division of Preventive Medicine Brigham and Women s s Hospital Harvard Medical School Overview Review
Fasting or non fasting?
 Vascular harmony Robert Chilton Professor of Medicine University of Texas Health Science Center Director of Cardiac Catheterization labs Director of clinical proteomics Which is best to measure Lower continues
Vascular harmony Robert Chilton Professor of Medicine University of Texas Health Science Center Director of Cardiac Catheterization labs Director of clinical proteomics Which is best to measure Lower continues
Clinical Trial Synopsis TL-OPI-518, NCT#
 Clinical Trial Synopsis, NCT# 00225264 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl vs Glimepiride
Clinical Trial Synopsis, NCT# 00225264 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl vs Glimepiride
PHYSICAL INACTIVITY AND BODY
 ORIGINAL CONTRIBUTION Relationship of Physical Activity vs Body Mass Index With Type 2 Diabetes in Women Amy R. Weinstein, MD, MPH Howard D. Sesso, ScD, MPH I. Min Lee, MBBS, ScD Nancy R. Cook, ScD JoAnn
ORIGINAL CONTRIBUTION Relationship of Physical Activity vs Body Mass Index With Type 2 Diabetes in Women Amy R. Weinstein, MD, MPH Howard D. Sesso, ScD, MPH I. Min Lee, MBBS, ScD Nancy R. Cook, ScD JoAnn
Biostatistics and Epidemiology Step 1 Sample Questions Set 1
 Biostatistics and Epidemiology Step 1 Sample Questions Set 1 1. A study wishes to assess birth characteristics in a population. Which of the following variables describes the appropriate measurement scale
Biostatistics and Epidemiology Step 1 Sample Questions Set 1 1. A study wishes to assess birth characteristics in a population. Which of the following variables describes the appropriate measurement scale
1. Which one of the following patients does not need to be screened for hyperlipidemia:
 Questions: 1. Which one of the following patients does not need to be screened for hyperlipidemia: a) Diabetes mellitus b) Hypertension c) Family history of premature coronary disease (first degree relatives:
Questions: 1. Which one of the following patients does not need to be screened for hyperlipidemia: a) Diabetes mellitus b) Hypertension c) Family history of premature coronary disease (first degree relatives:
2.0 Synopsis. Choline fenofibrate capsules (ABT-335) M Clinical Study Report R&D/06/772. (For National Authority Use Only) Name of Study Drug:
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Choline Fenofibrate (335) Name of Active Ingredient:
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Choline Fenofibrate (335) Name of Active Ingredient:
Biostatistics and Epidemiology Step 1 Sample Questions Set 2. Diagnostic and Screening Tests
 Biostatistics and Epidemiology Step 1 Sample Questions Set 2 Diagnostic and Screening Tests 1. A rare disorder of amino acid metabolism causes severe mental retardation if left untreated. If the disease
Biostatistics and Epidemiology Step 1 Sample Questions Set 2 Diagnostic and Screening Tests 1. A rare disorder of amino acid metabolism causes severe mental retardation if left untreated. If the disease
Non-insulin treatment in Type 1 DM Sang Yong Kim
 Non-insulin treatment in Type 1 DM Sang Yong Kim Chosun University Hospital Conflict of interest disclosure None Committee of Scientific Affairs Committee of Scientific Affairs Insulin therapy is the mainstay
Non-insulin treatment in Type 1 DM Sang Yong Kim Chosun University Hospital Conflict of interest disclosure None Committee of Scientific Affairs Committee of Scientific Affairs Insulin therapy is the mainstay
Bariatric Surgery versus Intensive Medical Therapy for Diabetes 3-Year Outcomes
 The new england journal of medicine original article Bariatric Surgery versus Intensive Medical for Diabetes 3-Year Outcomes Philip R. Schauer, M.D., Deepak L. Bhatt, M.D., M.P.H., John P. Kirwan, Ph.D.,
The new england journal of medicine original article Bariatric Surgery versus Intensive Medical for Diabetes 3-Year Outcomes Philip R. Schauer, M.D., Deepak L. Bhatt, M.D., M.P.H., John P. Kirwan, Ph.D.,
Measures of Obesity and Cardiovascular Risk Among Men and Women
 Journal of the American College of Cardiology Vol. 52, No. 8, 2008 2008 by the American College of Cardiology Foundation ISSN 0735-1097/08/$34.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2008.03.066
Journal of the American College of Cardiology Vol. 52, No. 8, 2008 2008 by the American College of Cardiology Foundation ISSN 0735-1097/08/$34.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2008.03.066
A Prospective Study of Cigarette Smoking and Risk of Incident Hypertension in Women
 Journal of the American College of Cardiology Vol. 50, No. 21, 2007 2007 by the American College of Cardiology Foundation ISSN 0735-1097/07/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2007.08.017
Journal of the American College of Cardiology Vol. 50, No. 21, 2007 2007 by the American College of Cardiology Foundation ISSN 0735-1097/07/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2007.08.017
Assessing Cardiovascular Risk to Optimally Stratify Low- and Moderate- Risk Patients. Copyright. Not for Sale or Commercial Distribution
 CLINICAL Viewpoint Assessing Cardiovascular Risk to Optimally Stratify Low- and Moderate- Risk Patients Copyright Not for Sale or Commercial Distribution By Ruth McPherson, MD, PhD, FRCPC Unauthorised
CLINICAL Viewpoint Assessing Cardiovascular Risk to Optimally Stratify Low- and Moderate- Risk Patients Copyright Not for Sale or Commercial Distribution By Ruth McPherson, MD, PhD, FRCPC Unauthorised
DIABETES. A growing problem
 DIABETES A growing problem Countries still grappling with infectious diseases such as tuberculosis, HIV/AIDS and malaria now face a double burden of disease Major social and economic change has brought
DIABETES A growing problem Countries still grappling with infectious diseases such as tuberculosis, HIV/AIDS and malaria now face a double burden of disease Major social and economic change has brought
Inflammation as A Target for Therapy. Focus on Residual Inflammatory Risk
 ESC Rome Monday August 29, 2016 Inflammation as A Target for Therapy Focus on Residual Inflammatory Risk Paul M Ridker, MD Eugene Braunwald Professor of Medicine Harvard Medical School Director, Center
ESC Rome Monday August 29, 2016 Inflammation as A Target for Therapy Focus on Residual Inflammatory Risk Paul M Ridker, MD Eugene Braunwald Professor of Medicine Harvard Medical School Director, Center
Controversies in Preventative Cardiology
 Controversies in Preventative Cardiology Francisco Lopez-Jimenez, M.D., M.Sc, FACC, FAHA Professor of Medicine, Mayo Medical School Chair, Division of Preventive Cardiology Co-Director, Artificial Intelligence
Controversies in Preventative Cardiology Francisco Lopez-Jimenez, M.D., M.Sc, FACC, FAHA Professor of Medicine, Mayo Medical School Chair, Division of Preventive Cardiology Co-Director, Artificial Intelligence
Disclosures. Learning Objectives. Effects of Hormone Therapy on the Metabolic Syndrome and Cardiovascular Disease. None
 Effects of Hormone Therapy on the Metabolic Syndrome and Cardiovascular Disease Micol S. Rothman, MD Associate Professor of Medicine Endocrinology, Diabetes and Metabolism Clinical Director Metabolic Bone
Effects of Hormone Therapy on the Metabolic Syndrome and Cardiovascular Disease Micol S. Rothman, MD Associate Professor of Medicine Endocrinology, Diabetes and Metabolism Clinical Director Metabolic Bone
C-Reactive Protein Levels and Outcomes after Statin Therapy
 The new england journal of medicine original article C-Reactive Protein Levels and Outcomes after Statin Therapy Paul M Ridker, M.D., Christopher P. Cannon, M.D., David Morrow, M.D., Nader Rifai, Ph.D.,
The new england journal of medicine original article C-Reactive Protein Levels and Outcomes after Statin Therapy Paul M Ridker, M.D., Christopher P. Cannon, M.D., David Morrow, M.D., Nader Rifai, Ph.D.,
Oral Hypoglycemics and Risk of Adverse Cardiac Events: A Summary of the Controversy
 Oral Hypoglycemics and Risk of Adverse Cardiac Events: A Summary of the Controversy Jeffrey Boord, MD, MPH Advances in Cardiovascular Medicine Kingston, Jamaica December 7, 2012 VanderbiltHeart.com Outline
Oral Hypoglycemics and Risk of Adverse Cardiac Events: A Summary of the Controversy Jeffrey Boord, MD, MPH Advances in Cardiovascular Medicine Kingston, Jamaica December 7, 2012 VanderbiltHeart.com Outline
Weigh the benefit of statin treatment: LDL & Beyond
 Weigh the benefit of statin treatment: LDL & Beyond Duk-Woo Park, MD, PhD Heart Institute, University of Ulsan College of Medicine, Asan Medical, Seoul, Korea FOURIER Further cardiovascular OUtcomes Research
Weigh the benefit of statin treatment: LDL & Beyond Duk-Woo Park, MD, PhD Heart Institute, University of Ulsan College of Medicine, Asan Medical, Seoul, Korea FOURIER Further cardiovascular OUtcomes Research
Supplementary Online Content
 Supplementary Online Content Larsen JR, Vedtofte L, Jakobsen MSL, et al. Effect of liraglutide treatment on prediabetes and overweight or obesity in clozapine- or olanzapine-treated patients with schizophrenia
Supplementary Online Content Larsen JR, Vedtofte L, Jakobsen MSL, et al. Effect of liraglutide treatment on prediabetes and overweight or obesity in clozapine- or olanzapine-treated patients with schizophrenia
