Preliminary Assessment of Celecoxib and Microdiode Pulse Laser Treatment of Diabetic Macular Edema
|
|
|
- Willa Newton
- 5 years ago
- Views:
Transcription
1 Preliminary Assessment of Celecoxib and Microdiode Pulse Laser Treatment of Diabetic Macular Edema The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation CHEW, EMILY Y., JONGHYEON KIM, HANNA R. COLEMAN, LLOYD PAUL AIELLO, GARY FISH, MICHAEL IP, JULIA A. HALLER, et al PRELIMINARY ASSESSMENT OF CELECOXIB AND MICRODIODE PULSE LASER TREATMENT OF DIABETIC MACULAR EDEMA. Retina 30, no. 3: doi: / iae.0b013e3181bcf1a0. Published Version doi: /iae.0b013e3181bcf1a0 Citable link Terms of Use This article was downloaded from Harvard University s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at nrs.harvard.edu/urn-3:hul.instrepos:dash.current.terms-ofuse#laa
2 NIH Public Access Author Manuscript Published in final edited form as: Retina March ; 30(3): doi: /iae.0b013e3181bcf1a0. Preliminary Assessment of Celecoxib and Microdiode Pulse Laser Treatment of Diabetic Macular Edema Emily Y. Chew, MD *, Jonghyeon Kim, PhD, Hanna R. Coleman, MD *, Lloyd Paul Aiello, MD, PhD, Gary Fish, MD, Michael Ip, MD, Julia A. Haller, MD **, Maria Figueroa, MSc, Daniel Martin, MD, David Callanan, MD, Robert Avery, MD, Keri Hammel, MSc, Darby J. S. Thompson, PhD, and Frederick L. Ferris III, MD * * Clinical Trials Branch, Division of Epidemiology and Clinical Applications, National Eye Institute, National Institutes of Health, Bethesda, Maryland The EMMES Corporation, Rockville, Maryland Beetham Eye Institute, Joslin Diabetes Center and Department of Ophthalmology, Harvard Medical School, Boston, Massachusetts Texas Retina Associates, Dallas, Texas Department of Ophthalmology, University of Wisconsin, Madison, Wisconsin ** Wilmer Eye Institute, Baltimore, Maryland Emory Eye Center, Atlanta, Georgia California Retina Consultants, Santa Barbara, California Abstract Purpose Inflammation may play an important role in the pathogenesis of diabetic macular edema, a major cause of vision loss in persons with diabetes. The purpose of this study was to evaluate combined antiinflammatory therapy and laser approaches for treating patients with diabetic macular edema. Methods In this prospective, factorial, randomized, multicenter trial, we compared cyclooxygenase-2 inhibitor (celecoxib) with placebo and diode grid laser with standard Early Treatment Diabetic Retinopathy Study focal laser treatment in 86 participants with diabetic macular edema. The primary outcome is change in visual acuity of 15 letters from baseline, and the secondary outcomes include a 50% reduction in the retinal thickening of diabetic macular edema measured by optical coherence tomography and a 50% reduction in leakage severity on fluorescein angiography. Results Visual acuity and retinal thickening data from >2 years of follow-up did not show evidence of differences between the medical and laser treatments. However, participants assigned to the celecoxib group were more likely to have a reduction in fluorescein leakage when compared with the placebo group (odds ratio = 3.6; P < 0.01). Conclusion This short-term study did not find large visual function benefits of treatment with celecoxib or diode laser compared with those of standard laser treatment. A suggestive effect of celecoxib in reducing fluorescein leakage was observed. Keywords diabetic macular edema; celecoxib; diode grid laser photocoagulation; ETDRS type focal photocoagulation; optical coherence tomography Reprint requests: Emily Y. Chew, MD, National Institutes of Health, Building 10, CRC, Room , 10 Center Drive, MSC 1204, Bethesda, MD ; echew@nei.nih.gov. Julia A. Haller now at Will's Eye Philadelphia, Pennsylvania Robert Avery now at Department of Ophthalmology, Cleveland Eye Clinic, Cleveland, Ohio
3 Chew et al. Page 2 Study Design Macular edema as a result of diabetic retinopathy can lead to central vision loss. It is one of the major causes of visual loss in the United States, 1 with 4.1 million Americans (age 40 years) estimated to have diabetic retinopathy. 2 Vision-threatening diabetic retinopathy, which includes both proliferative diabetic retinopathy and diabetic macular edema (DME), is estimated to occur in 899,000 persons with the majority having type 2 diabetes. 2 The estimated proportions of persons with diabetes affected by macular edema are type 1 diabetes, 20%; type 2 diabetes, using insulin, 25%; and type 2 diabetes, not using insulin, 14%. 3 The prevalence of diabetes is expected to substantially increase by 2020 with the increasing prevalence of diabetes mellitus in the aging U.S. population. When macular edema is established, focal laser photocoagulation reduces the risk of moderate visual loss (loss of 15 letters from baseline on the Early Treatment Diabetic Retinopathy Study [ETDRS] visual acuity charts) by 50%. 4 However, treatment is often not associated with improvement in visual acuity. Furthermore, the focal laser burns may result in an increase in atrophy of the retinal pigment epithelium and progressive enlargement of the initial focal scars of laser photocoagulation and eventually visual loss with central scotomas and decreased color vision. 5 Reduction of tissue destruction from laser burns while maintaining the beneficial treatment effect would potentially reduce this long-term risk of laser treatment. These adverse side effects of the continuous wavelength laser used in the ETDRS technique may theoretically be diminished by using a micropulse diode laser grid, which would result in subthreshold burns with the accompanying reduction of damage from photocoagulation to the retinal pigment epithelium. 6,7 This technique has been evaluated in case series without concurrent controls This study was designed to compare the micropulse diode grid laser with the standard ETDRS focal laser technique using the continuous wavelength laser. Animal models of diabetic retinopathy demonstrate that inflammation plays an important role in diabetic retinopathy. 11 In early onset of diabetic retinopathy, up-regulation of cyclooxygenase-2 (COX-2) occurs in retinal cells. This results in elevated prostaglandin production, leading to an increased expression of vascular endothelial growth factor and an increased risk of vascular leakage and retinal neovascularization. 12 High doses of aspirin and intermediate doses of COX-2 inhibitors (celecoxib) were apparently beneficial in early experimental diabetic retinopathy. 13 Other investigations have shown that periocular celecoxib-poly (lactide-co-glycolide) microparticles inhibited elevations of retinal vascular endothelial growth factor and other factors for as long as 60 days in a diabetic animal model. 14 Cyclo-oxygenase-2 inhibitors have been demonstrated to have antiangiogenic effects in a rat model of ischemic proliferative retinopathy, 15 and COX-2 expression is evident in human retina of individuals with diabetes. 8 These data suggest that treatment with a COX-2 inhibitor may be beneficial in the treatment of DME. The purpose of this study was to gather pilot data to evaluate the feasibility of mounting a larger multicenter study to evaluate the efficacy of celecoxib (a COX-2 inhibitor and a nonsteroid anti-inflammatory drug) and of a modified laser photocoagulation technique for the treatment of DME. This multicenter study was designed to investigate the feasibility and the proof-of-principle study for 2 new treatment approaches for patients with diabetic retinopathy and clinically significant macular edema (n = 100), potentially leading to initiation of a larger Phase 3 clinical trial.
4 Chew et al. Page 3 Design A total of 86 participants were enrolled from December 2002 through November 2004 at 8 centers in the United States after reviewing and signing an informed consent form approved by the respective sites' Institutional Review Boards. This study was prematurely terminated because of safety concerns of celecoxib treatment reported by other large trials. 16 Eligibility criteria included age 18 years, evidence of type 1 or 2 diabetes, evidence of clinically significant macular edema in at least one eye that requires laser treatment, and bestcorrected visual acuity of 20/400 or better using the ETDRS refraction and visual acuity measurement protocol. 17 In addition, exclusion criteria included the following: retinopathy that may require scatter photocoagulation immediately, other retinal and systemic diseases that might confound the outcomes (age-related macular degeneration, uveitis, drug toxicity, sarcoidosis, inflammatory bowel disease, and so on), and a history of myocardial infarction within the previous 2 years. Participants were randomly assigned to receive an oral dose of 400 mg (200 mg 2 times a day) of celecoxib or a matching placebo only daily for 3 months without laser, and the medical therapy of celecoxib continued until the study was stopped. All participants and the research team at each clinical site were masked to the study medication assignments. Eligible eyes of participants were randomly assigned to receive either ETDRS focal laser 18 or diode grid laser photocoagulation at their 3-month visit if macular edema persisted. If both eyes were eligible, the right eye was designated as the study eye and was randomly assigned to receive one of two laser therapies with the fellow eye assigned to receive the alternate laser treatment. The first laser treatment was given when the study ophthalmologist judged that the laser treatment was medically indicated, starting 3 months after randomization to celecoxib or placebo. All laser treatments given during the first year were performed according to the randomized laser treatment assignment. After 1 year of followup, participants, who had their eyes initially treated with diode laser treatment, were allowed to receive ETDRS laser treatment if additional treatment was required. The micropulse diode technique was conducted with the 810 diode laser (Iris Medical OcuLight SLx, IRIDEX Corporation, Mountain View, CA) by using the MicroPulse operating mode. The treatment consisted of 125-μm size burns using a 10% duty cycle with 0.2-second pulse envelope duration with a maximum power of 1.8 W. These were subthreshold burns that covered the entire area of macular edema with minimal clinical detection or fluorescein angiographic changes associated with these laser burns. The ETDRS focal treatment using yellow or green laser was a technique of direct laser photocoagulation ( μm spot size) at microaneurysms with light burns, not necessary to whiten or darken the lesions. When no focal leakage was identified, a grid laser with light burns was administered to sufficiently cover the area of edema. The burns were spaced greater than one burn width apart with the sparing of the 500 μm from the center of the fovea. Study visits were scheduled 3 and 6 months after enrollment and every 6 months thereafter for a total of 3 years of follow-up. At each study visit, a comprehensive ophthalmic examination was conducted including best-corrected visual acuity, slit-lamp biomicroscopy, dilated fundus examination, retinal thickness with the optical coherence tomography (Stratus OCT, Carl Zeiss Meditec, Dublin, CA), and fluorescein angiography (FA). Laser photocoagulation therapy was considered for all participants with clinically significant macular edema at each study visit. The study design and study data were reviewed and monitored by the National Eye Institute's Data and Safety Monitoring Committee.
5 Chew et al. Page 4 Outcomes The designated primary outcome was a 15-letter score improvement in best-corrected visual acuity from baseline to the third year. The secondary outcome was a significant reduction of macular edema at the end of the third year. A significant reduction was defined as a 50% reduction in retinal thickening as measured by optical coherence tomography and a 50% reduction in leakage area as measured by FA. The optical coherence tomographies and FAs were graded by the Fundus Photograph Reading Center at the University of Wisconsin, whose graders were also masked to the assignment of the medical therapy. 19,20 An additional secondary outcome was the number of laser treatments required during the course of the study. Safety measurements, including laboratory tests and adverse event assessments, were also collected. Because of the premature study termination, outcomes through the second year were analyzed. This early termination also reduced the power of the study because the projected sample size of 100 was not achieved. Change in Recruitment and Follow-Up Statistical Methods Results On December 17, 2004, the National Institutes of Health suspended the use of celecoxib for all participants in a large colorectal cancer prevention clinical trial, the Adenoma Prevention with Celecoxib Trial, because analyses by the trial's Data Safety and Monitoring Committee found a two- to threefold increased risk of major fatal and nonfatal cardiovascular events for participants taking celecoxib compared with those in the placebo group. 21 In accordance with the Data and Safety Monitoring Committee's action on other National Institutes of Health-supported studies using a COX-2 inhibitor, the National Eye Institute Data and Safety Monitoring Committee thoroughly reviewed all the reported adverse events as well as the main study results on December 20, 2004, and recommended that the National Eye Institute close the enrollment of participants and also suspend the use of celecoxib or placebo for all enrolled participants. The participants were followed for 1 year after discontinuing the study drug. This resulted in curtailed follow-up, especially for participants who were enrolled late in the study, and lowered the power of the study evaluating the longterm effects of study treatments. The cross-sectional analyses of the primary and secondary outcomes for each study visit were examined using two-sample mean and proportion comparison tests. The generalized estimating equation method was used to adjust the difference between the two groups for the correlation between bilateral outcomes. 22 When a statistically significant difference existed between the two groups, the extended generalized estimating equation method was applied to investigate the trends and variations in the outcome measures during the course of the study. 23 To allow for the unequal length of follow-up of each participant, the proportional hazard model for the first occurrence of event of interest was also considered if the result from the generalized estimating equation model was statistically significant. Parsimonious and well-fitted models for each outcome were derived by backward variable selection of the following variables: age and baseline level of each outcome. All the data analyses were conducted using SAS version 8.2 (SAS Institute, Inc., Cary, NC). Baseline Demographics and Characteristics Eighty-six participants with diabetic retinopathy and clinically significant macular edema were enrolled (Table 1). Forty-seven participants were randomly assigned to the placebo group (34% women and 74% white) and 39 participants to the celecoxib group (43% women
6 Chew et al. Page 5 and 59% white) (Figure 1). The median ages for the placebo and celecoxib participants were 61 and 58 years, respectively. All the participants have received diabetic treatments, including insulin, oral medication, or diet (94% and 92% of placebo and celecoxib participants, respectively, on insulin or oral medication for at least 1 year). Participants with both eyes eligible for the study numbered 14 in the placebo group and 9 in the celecoxib group. The median visual acuity score and retinal thickening (thickness 175 μm) at the center subfield at baseline was 75 letters (20 of 32) and 135 μm and 82 letters (20 of 25) and 85 μm for the placebo and celecoxib groups, respectively. There was an imbalance of the retinal thickness between the placebo and celecoxib groups. Follow-Up and Duration of Drug Treatment Visual Acuity Laser Treatments Among the 86 enrolled participants, 78 (91%) participants (43 in the placebo group and 31 in the celecoxib group) completed 1 year of follow-up and 54 (63%) participants (29 in the placebo group and 25 in the celecoxib group) finished the 2-year follow-up visit (Table 2). Before cessation of the study therapy on December 31, 2004, 47 participants (26 in the placebo group and 21 in the celecoxib group) had received study medication for at least 1 year. Until study closeout, 9 placebo participants (19%) and 3 celecoxib participants (8%) experienced a serious, cardiovascular adverse event (exact P = 0.225). The data from this trial collected through January 31, 2005, have been analyzed with the data from 5 other randomized, controlled trials of celecoxib supported by the National Cancer Institute in a project called the Cross-Trials Safety Analysis for the risk of serious cardiovascular events caused by the use of celecoxib. 15 This small study has too few participants to yield clinically meaningful data (Table 2). Table 3 (Figure 2) compares mean changes in visual acuity by treatment group (placebo or celecoxib) at each study visit. No significant differences were detected in mean visual acuity. Additionally, the rates of vision loss of 3 lines from baseline to 1 and 2 years were also similar (P = 0.82 and P = 0.66 for comparison of mean changes, respectively). During the first year, 7 placebo participants (12%) and 3 celecoxib participants (8%) lost 3 lines of vision. Three placebo participants (5%) and 2 celecoxib participants (5%) gained 3 lines of vision. Overall, the drug-treated group and placebo group do not differ significantly in visual acuity results (either improvement or worsening) at any of the time points. Fifty-eight participants needed at least one laser treatment for their macular edema (nine participants needed laser treatment in both eyes and received different laser treatments in each). More specifically, 41 eyes of 34 participants in the placebo group (34 of 47 [72%]) and 26 eyes of 24 participants in the celecoxib group (24 of 39 [62%]) required at least 1 laser treatment throughout the follow-up period. The participants in the placebo group had slightly higher rates of requiring laser treatments than those participants in the celecoxib group, but this was not statistically significant (odds ratio [OR] = 1.63; P = 0.36 for participants and OR = 1.73; P = 0.15 for eyes). After the 1-year follow-up visit, the investigator was permitted to treat the participants with either lasers (11 participants switched from diode laser to ETDRS laser). Table 4 compares the proportion of laser retreatment among the laser-treated eyes. The proportion was estimated from a binomial probability distribution with the denominator being the number of opportunities to receive laser retreatment before the last visit. These analyses do not provide evidence that celecoxib reduces the need for laser retreatment (P 0.54).
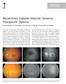
7 Chew et al. Page 6 Fluorescein Angiographic Leakage Severity The number of participants eligible for the analysis of fluorescein leakage was decreased because the FAs of 4 participants were not gradable, and 6 participants had stage 1 or less in severity of macular edema at baseline, making them ineligible for analysis of the reduction of FA leakage by 2 steps. At the first-year follow-up visit, participants in the celecoxib group had a higher chance of having reduced FA leakage severity than did the participants in the placebo group (43% vs 26% complete case analysis; 33% vs 22% intention-to-treat analysis), but this difference was not statistically significant (exact P > 0.19). At the secondyear follow-up visit, 3 of 24 (12%) participants in the placebo group and 9 of 20 (45%) participants in the celecoxib group experienced a 2-step reduction in leakage severity (exact P = 0.02). These statistically significant effects of celecoxib on the 2-step leakage severity reduction also appear in the intention-to-treat analysis that treats all missing outcomes as failures. Although the statistical significance levels are diminished, the effects are still suggestive in the most conservative analysis (OR = 4.11; exact P = at year 2). Table 5 presents repeated measures analyses of a 2-step reduction in leakage severity. Celecoxib participants had reduced leakage severity with a higher likelihood than did placebo participants (unadjusted OR = 2.45; P = and unadjusted OR = 3.31; P = for 1- and 2-year results, respectively). Adjusting for baseline leakage severity, follow-up year, and laser treatment, the reduction in leakage associated with celecoxib remains significant at 2 years (OR = 3.52; P = 0.002). Repeated measure analyses based on the entire follow-up data set have similar results. Figure 3 displays Kaplan Meier estimates of the incidence of the first event of a 2-step reduction in leakage severity. Cox proportional hazards model, for the time to the first occurrence of a 2-step reduction in leakage severity, shows that there is a 1.8 times higher chance of reduction in leakage severity (P = 0.067) in participants assigned to the celecoxib group than that in participants assigned to the placebo group. Figure 4 shows the typical increased fluorescein leakage in the study eye of a participant at baseline (Figure 4A). There was a reduction in the fluorescein leakage of 2 steps along the severity scale at 24 months (Figure 4B). Retinal Thickening Measured on Optical Coherence Tomography Discussion Retinal thickening measurements at the center subfield, the excessive thickness compared with that of a normal eye (defined as 175 μm), are right skewed and subject to high variation and therefore transformation may be required (e.g., log transformation); however, the results based on this transformation (not shown) do not differ from those based on raw scales. Participants in both treatment groups presented with reduced retinal thickening by 26 μm to 31 μm on average during the first year (P = 0.66 in Table 6) and by >64 μm during the second year (P = 0.94 in Table 6). In the first year, 4 of 40 eyes in the placebo group (10%) and 5 of 27 eyes in the celecoxib group (18%) had a 50% reduction in retinal thickening. Eleven eyes (27%) of the placebo participants and 8 eyes (30%) of the celecoxib participants had at least a 25% reduction in the first year. These reduction rates do not differ between the two treatment groups (P 0.26 for either size reduction). Within the specific laser type, both treatment groups showed a similar degree of reduction ( 30 μm) in retinal thickening. Overall, there are no statistically significant differences in changes in retinal thickening by treatment group or laser treatment. The results of this pilot study suggest that it is feasible to mount a multicenter study to evaluate the role of medical and laser therapies for the treatment of DME. However, the COX-2 inhibitor chosen for this study, celecoxib, was associated with an increased risk of cardiovascular disease in other clinical trials. The increased risk observed in several large
8 Chew et al. Page 7 trials makes it difficult to launch a large clinical trial in persons with diabetes who are already at an increased risk for cardiovascular disease. Acknowledgments This pilot study has insufficient power to evaluate the potential beneficial effect of both the medical and laser treatment approaches. Although the early discontinuation of celecoxib in this study may have reduced any measurable beneficial effects, these data did not provide evidence of the treatment effects from these therapeutic strategies. The inflammatory pathways found in the animal data are compelling, and it is intriguing that the medical therapy with the COX-2 inhibitor resulted in an apparently greater reduction in fluorescein leakage compared with the placebo group. These leakage results were verified by regrading the leakage at the centralized reading center with similar results. This apparent reduction in fluorescein leakage suggests that COX-2 inhibitors or other anti-inflammatory treatments may have a role in therapy for DME. Focal laser photocoagulation treatment given in the focal ETDRS fashion is the only therapy currently demonstrated to be beneficial for the treatment of DME. The ETDRS showed that this treatment reduced the 3-year rate of moderate vision loss ( 3 lines of acuity loss) by 50%. 6 However, only 17% of the treated eyes that had baseline acuity worse than 20/40 experienced moderate visual gain ( 3 lines of acuity improvement). 6 Our results from the laser arm showed that there were no large differences detected between the two laser techniques evaluated in this study. It should also be noted that during the first year, 11 of those assigned to diode laser were switched to the ETDRS type of treatment. This pilot study was too small and its follow-up too short to evaluate whether some of the possible side effects of laser photocoagulation such as enlargement of laser burns, increases in scotomas, and decreased vision can be attenuated with diode laser treatment. Studies with longer duration and larger numbers of participants would be required to answer this question. The current investigations of possible therapies for DME have included intravitreal triamcinolone, 24 oral protein kinase C inhibitors, 25 intravitreal aptamers, 26,27 antibodies directed against vascular endothelial growth factor, 28,29 and vitrectomy. 30 Intravitreal triamcinolone injection does address the issue of inflammation, but intravitreal injections are accompanied by the potential adverse effects of raised intraocular pressure, the development of cataract, and the rare possibility of endophthalmitis. A recently completed trial of intravitreal triamcinolone injections versus DME confirmed the importance of laser photocoagulation after 2 years of follow-up. 31 Intravitreal steroids may have some shortterm beneficial effect, and the effects of combined steroids and laser are yet to be fully evaluated. Studies are also underway to evaluate the use of intravitreal antivascular endothelial growth factor therapy for DME. Laboratory data and observational studies suggest that inflammation may play a potential role in the pathogenesis of diabetic macular retinopathy. Although this study failed to provide evidence that COX-2 inhibitors may be effective in the treatment of DME, there was some evidence of a reduction of fluorescein leakage, and future investigations of antiinflammatory agents for the treatment of DME seem warranted. Supported by the National Eye Institute or National Institutes of Health intramural program. The study medication and funds for the examinations of the participants enrolled in the extramural clinics were provided by Pharmacia.
9 Chew et al. Page 8 References 1. The Eye Disease Prevalence Research Group. Causes and prevalence of visual impairment among adults in the United States. Arch Ophthalmol 2004;122: [PubMed: ] 2. The Eye Disease Prevalence Research Group. The prevalence of diabetic retinopathy among adults in the United States. Arch Ophthalmol 2004;122: [PubMed: ] 3. Klein R, Klein B, Moss SE, Davis MD, DeMets DL. The Wisconsin epidemiologic study of diabetic retinopathy. IV. Diabetic macular edema. Ophthalmology 1984;91: [PubMed: ] 4. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study Research Group. Arch Ophthalmol 1985;103: [PubMed: ] 5. Schatz H, Maderia D, McDonald HR, Johnson RN. Progressive enlargement of laser scars following grid laser photocoagulation for diffuse diabetic macular edema. Arch Ophthalmol 1991;109: [PubMed: ] 6. Mainster MA. Decreasing retinal photocoagulation damage: principles and techniques. Semin Ophthalmol 1999;14: [PubMed: ] 7. Roider J. Laser treatment of retinal diseases by subthreshold laser effects. Semin Ophthalmol 1999;14: [PubMed: ] 8. Roider J, Brinkmann R, Wirbelauder C, Laqua H, Birngruber R. Subthreshold (retinal pigment epithelium) photocoagulation in macular diseases: a pilot study. Br J Ophthalmol 2000;84: [PubMed: ] 9. Luttrull JK, Musch DC, Mainster MA. Subthreshold diode micropulse photocoagulation for the treatment of clinically significant diabetic macular edema. Br J Ophthalmol 2005;89: [PubMed: ] 10. Sivaprasad S, Sandhu R, Tandon A, Sayed-Ahmed K, McHugh DA. Subthreshold micropulse diode laser photocoagulation for clinically significant diabetic macular oedema: a three-year follow-up. Clin Experiment Ophthalmol 2007;7: [PubMed: ] 11. Johnson EI, Dunlop ME, Larkins RG. Increased vasodilatory prostaglandin production in the diabetic rat retinal vasculature. Curr Eye Res 1999;18: [PubMed: ] 12. Aiello LP, Avery RL, Arrigg PG, et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med 1994;331: [PubMed: ] 13. Joussen AM, Poulaki V, Mitsiades N, et al. Nonsteroidal anti-inflammatory drugs prevent early diabetic retinopathy via TNF-alpha suppression. FASEB J 2002;16: [PubMed: ] 14. Amrite AC, Ayalasomayajula SP, Cheruvu NPS, Kompella UB. Single periocular injection of celecoxib-plga microparticles inhibits diabetes induced elevations of retinal PGE 2, VEGF, and vascular leakage. Invest Ophthalmol Vis Sci 2006;47: [PubMed: ] 15. Sennlaub F, Valamanesh F, Vazquez-Tello A, et al. Cyclooxygenase-2 in human and experimental ischemic proliferative retinopathy. Circulation 2003;108: [PubMed: ] 16. Solomon SD, Wittes J, Finn PV, et al. Cardiovascular risk of celecoxib in 6 randomized placebocontrolled trials: the cross trial safety analysis. Circulation 2008;117: [PubMed: ] 17. Ferris FL, Kassoff A, Bresnick GH, Bailey I. New visual acuity charts for clinical research. Am J Ophthalmol 1982;94: [PubMed: ] 18. Treatment techniques and clinical guidelines for photocoagulation of diabetic macular edema. Early Treatment Diabetic Retinopathy Study Report Number 2. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 1987;94: [PubMed: ] 19. Davis MD, Bressler SB, Aiello LP, et al. Comparison of time-domain OCT and fundus photographic assessments of retinal thickening in eyes with diabetic macular edema. Invest Ophthalmol Vis Sci 2008;49: [PubMed: ] 20. Classification of diabetic retinopathy from fluorescein angiograms. ETDRS report number 11. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 1991;98: [PubMed: ]
10 Chew et al. Page Solomon SD, McMurray JJV, Pfeffet MA, et al. Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N Engl J Med 2005;352: [PubMed: ] 22. Liang KY, Zeger SL. Longitudinal data analysis using generalized linear model. Biometrika 1986;73: Zhao LP, Prentice RL. Correlated binary regression using a quadratic exponential model. Biometrika 1990;77: Martidis A, Duker JS, Greenberg PB, et al. Intravitreal triamcinolone for refractory diabetic macular edema. Ophthalmology 2002;109: [PubMed: ] 25. The PKC-DRS Study Group. The effect of ruboxistaurin on visual loss in patients with moderately severe to very severe nonproliferative diabetic retinopathy: initial results of the Protein Kinase C beta Inhibitor Diabetic Retinopathy Study (PKC-DRS) multicenter randomized clinical trial. Diabetes 2005;54: [PubMed: ] 26. Cunningham ET, Adamis AP, Altaweel M. A phase II randomized double-masked trial of pegaptanib, an anti-vascular endothelial growth factor aptamer, for diabetic macular edema. Ophthalmology 2005;112: [PubMed: ] 27. Cunningham ET Jr, Adamis AP, Altaweel M, Macugen Diabetic Retinopathy Study Group. A phase II randomized double-masked trial of an anti-vascular endothelial growth factor aptamer, for diabetic macular edema. Ophthalmology 2005;112: [PubMed: ] 28. Haritoglou C, Kook D, Neubauer A, et al. Intravitreal bevacizumab (Avastin) therapy for persistent diffuse diabetic macular edema. Retina 2006;26: [PubMed: ] 29. Nguyen QD, Tatlipinar S, Shah SM, et al. Vascular endothelial growth factor is a critical stimulus for diabetic macular edema. Am J Ophthalmol 2006;142: [PubMed: ] 30. Pendergast SD, Hassan TS, Williams GA, et al. Vitrectomy for diffuse diabetic macular edema associated with a taut premacular posterior hyaloid. Am J Ophthalmol 2000;130: [PubMed: ] 31. Diabetic Retinopathy Clinical Research Network. A randomized trial comparing intravitreal triamcinolone acetonide and focal/grid photocoagulation for diabetic macular edema. Ophthalmology 2008;115: [PubMed: ]
11 Chew et al. Page 10 Fig. 1. The enrollment and randomization of the study participants.
12 Chew et al. Page 11 Fig. 2. The mean change in visual acuity in participants assigned to celecoxib or placebo.
13 Chew et al. Page 12 Fig. 3. Life table estimates of the time to progression in fluorescein leakage in participants assigned to celecoxib or placebo.
14 Chew et al. Page 13 Fig. 4. At baseline (A), the clinically significant macular edema of this left study eye of a 31-yearold man with a 16-year history of diabetes had fluorescein leakage that was reduced significantly at 24 months. He did not receive laser photocoagulation during the course of the study (B).
15 Chew et al. Page 14 Table 1 Participant Demographic and Baseline Characteristics by Treatment Group Placebo Celecoxib Randomized participants 47 (100%) 39 (100%) Sex, female 16 (34%) 17 (43%) Race, white 35 (74%) 23 (59%) Age (median years) year diabetic treatment of insulin or oral medication 44 (94%) 36 (92%) Number of eyes * VA (median letter score) Retinal thickening at the center subfield (median) Laser type assigned for eligible eyes Diode ETDRS Laser type given for eligible eyes by randomization Diode ETDRS * Both eyes are eligible in 23 participants (14 in the placebo group and 9 in the celecoxib group). Retinal thickening at the center subfield is excessive thickness >175 (the difference of retinal thickness between the study participants and a normal subject). VA, visual acuity.
16 Chew et al. Page 15 Table 2 Participants' Follow-Up and Duration on Study Therapy Before Mandated Discontinuation by Treatment Group Placebo Celecoxib P Randomized participants 47 (100%) 39 (100%) Follow-up (months) * <12 2 (4%) 6 (15%) 12 <24 16 (34%) 8 (21%) (62%) 25 (64%) Duration (months) on drug treatment <12 21 (45%) 18 (46%) 12 <24 24 (51%) 19 (49%) 24 2 (4%) 2 (5%) Serious cardiovascular events Entire follow-up 9 (19%) 3 (8%) CTSA 6 (13%) 2 (5%) * Follow-up is based on the most recent clinical/ophthalmic examination visit. Follow-up before mandatory discontinuation of masked and randomized study medication (either placebo or celecoxib). Follow-up until study closeout. Cross-Trials Safety Analysis (CTSA, this includes this study of diabetic retinopathy and other studies of randomized, controlled trials of celecoxib for adenoma of the gastrointestinal system, breast cancer, and for Alzheimer disease) has adjudicated cardiovascular adverse events from randomization to 1 year after study therapy discontinuation (January 31, 2005); 1 death because of stroke of a placebo participant is not included.
17 Chew et al. Page 16 Visual Acuity by Treatment Group Table 3 Month Treatment Group Number of Eyes (Participants) Mean/Median Change in Letter Score * 95% CI for Mean P Gain of 15-Letter Score Loss of 15-Letter Score 6 Placebo 57 (44) 1.42/ /57 (1.8%) 2/57 (3.5%) Celecoxib 40 (34) 3.15/ /40 (5.0%) 3/40 (7.5%) 12 Placebo 56 (43) 2.25/ /56 (5.4%) 7/56 (12.5%) Celecoxib 37 (31) 2.89/ /37 (5.4%) 3/37 (8.1%) 18 Placebo 51 (39) 1.86/ /51 (5.9%) 6/51 (11.8%) Celecoxib 35 (29) 3.77/ /35 (0%) 3/35 (8.6%) 24 Placebo 35 (28) 1.42/ /35 (5.7%) 5/35 (14.3%) Celecoxib 29 (25) 3.03/ /29 (10.3%) 4/29 (13.8%) * Change is the difference from baseline. Positive (negative) values indicate the increase (decrease) in visual acuity letter score. The significance level of the treatment group effect in an analysis of variance for repeated measures; data to incorporate the correlation between bilateral outcomes. CI, confidence interval.
18 Chew et al. Page 17 Table 4 Laser Retreatments Among Laser-Treated Eyes Number of Eyes With Laser Retreatment Placebo (%) * Celecoxib (%) * OR P Diode (19 placebo; 10 celecoxib) ETDRS (22 placebo; 16 celecoxib) Total (41 placebo; 26 celecoxib) * Rates were estimated using a binomial probability model with the number of opportunities to receive laser retreatments before last visit. Reference group is placebo.
19 Chew et al. Page 18 Table 5 The Reduction of Fluorescein Leakage by 2 or More Steps in Eyes With at Least Stage 2 Severity of Macular Edema at Baseline in Those Subjects Assigned to Celecoxib Treatment * Month Covariate Adjustment Factors OR 95% CI for OR P 12 None Baseline severity and year Baseline severity, year, and laser treated None Baseline severity and year Baseline severity, year, and laser treated None Baseline severity and year Baseline severity, year, and laser treated * Analysis of variance model for repeated measures. Reference group is placebo. CI, confidence interval.
20 Chew et al. Page 19 Retinal Thickening of Eligible Study Eyes by Treatment Group * Table 6 Change in Retinal Thickening Relative Change in Retinal Thickening Month Treatment Group Number of Eyes Median/Mean 95% CI for Mean P 50% Reduction 25% Reduction 12 Placebo / /40 (10%) 11/40 (27.5%) Celecoxib / /27 (18.5%) 8/27 (29.6%) 18 Placebo / /37 (18.9%) 11/37 (29.7%) Celecoxib / /21 (23.8%) 6/21 (28.6%) 24 Placebo / /24 (29%) 11/24 (45.8%) Celecoxib / /16 (23.8%) 7/21 (33.3%) * Retinal thickening at the center subfield is excessive thickness >175 (the difference of retinal thickness between the study participants and a normal subject). Negative (positive) sign indicates the decrease (increase) of retinal thickening. Significance level of the treatment group effect in an analysis of variance for repeated measures data to incorporate the correlation between bilateral outcomes. CI, confidence interval.
Diabetic retinopathy (DR) progressively
 FOCUS ON DIABETIC MACULAR EDEMA * Frederick L. Ferris III, MD ABSTRACT The current state of treatment for diabetic macular edema (DME) is focused on slowing the rate of vision loss through assessment of
FOCUS ON DIABETIC MACULAR EDEMA * Frederick L. Ferris III, MD ABSTRACT The current state of treatment for diabetic macular edema (DME) is focused on slowing the rate of vision loss through assessment of
In its initial report, the Early Treatment Diabetic Retinopathy. A Severity Scale for Diabetic Macular Edema Developed from ETDRS Data
 A Severity Scale for Diabetic Macular Edema Developed from ETDRS Data Ronald E. Gangnon, 1,2 Matthew D. Davis, 3 Larry D. Hubbard, 3 Lloyd M. Aiello, 4 Emily Y. Chew, 5 Frederick L. Ferris III, 5 Marian
A Severity Scale for Diabetic Macular Edema Developed from ETDRS Data Ronald E. Gangnon, 1,2 Matthew D. Davis, 3 Larry D. Hubbard, 3 Lloyd M. Aiello, 4 Emily Y. Chew, 5 Frederick L. Ferris III, 5 Marian
Andrew J. Barkmeier, MD; Benjamin P. Nicholson, MA; Levent Akduman, MD
 c l i n i c a l s c i e n c e Effectiveness of Laser Photocoagulation in Clinically Significant Macular Edema With Focal Versus Diffuse Parafoveal Thickening on Optical Coherence Tomography Andrew J. Barkmeier,
c l i n i c a l s c i e n c e Effectiveness of Laser Photocoagulation in Clinically Significant Macular Edema With Focal Versus Diffuse Parafoveal Thickening on Optical Coherence Tomography Andrew J. Barkmeier,
Diabetic retinopathy (DR) is the leading PROCEEDINGS EVIDENCE-BASED DATA IN THE TREATMENT OF DIABETIC RETINOPATHY*
 EVIDENCE-BASED DATA IN THE TREATMENT OF DIABETIC RETINOPATHY* Lloyd Paul Aiello, MD, PhD, and Jennifer K. Sun, MD, MPH ABSTRACT There is a wide array of research into the pathophysiology and treatment
EVIDENCE-BASED DATA IN THE TREATMENT OF DIABETIC RETINOPATHY* Lloyd Paul Aiello, MD, PhD, and Jennifer K. Sun, MD, MPH ABSTRACT There is a wide array of research into the pathophysiology and treatment
Clinical Trials in Diabetic Retinopathy. Harry W. Flynn Jr., M.D. Nidhi Relhan Batra, M.D.
 1 Clinical Trials in Diabetic Retinopathy 2018 Harry W. Flynn Jr., M.D. Nidhi Relhan Batra, M.D. Bascom Palmer Eye Institute 900 N.W. 17th Street Miami, FL 33136 Phone: (305) 326-6118 Fax: (305) 326-6417
1 Clinical Trials in Diabetic Retinopathy 2018 Harry W. Flynn Jr., M.D. Nidhi Relhan Batra, M.D. Bascom Palmer Eye Institute 900 N.W. 17th Street Miami, FL 33136 Phone: (305) 326-6118 Fax: (305) 326-6417
CLINICAL SCIENCES. Results After Lens Extraction in Patients With Diabetic Retinopathy. Early Treatment Diabetic Retinopathy Study Report Number 25
 CLINICAL SCIENCES Results After Lens Extraction in Patients With Diabetic Retinopathy Early Treatment Diabetic Retinopathy Study Report Number 25 Emily Y. Chew, MD; William E. Benson, MD; Nancy A. Remaley,
CLINICAL SCIENCES Results After Lens Extraction in Patients With Diabetic Retinopathy Early Treatment Diabetic Retinopathy Study Report Number 25 Emily Y. Chew, MD; William E. Benson, MD; Nancy A. Remaley,
Effect of Ruboxistaurin on Blood Retinal Barrier Permeability in Relation to Severity of Leakage in Diabetic Macular Edema
 Effect of Ruboxistaurin on Blood Retinal Barrier in Relation to Severity of Leakage in Diabetic Macular Edema The Harvard community has made this article openly available. Please share how this access
Effect of Ruboxistaurin on Blood Retinal Barrier in Relation to Severity of Leakage in Diabetic Macular Edema The Harvard community has made this article openly available. Please share how this access
Diabetic retinopathy (DR) represents a
 THE MOLECULAR BIOLOGY OF DIABETIC RETINOPATHY: OPPORTUNITIES FOR THERAPEUTIC INTERVENTION Lloyd P. Aiello, MD, PhD ABSTRACT Hyperglycemia is the principal underlying cause of diabetic microvascular complications,
THE MOLECULAR BIOLOGY OF DIABETIC RETINOPATHY: OPPORTUNITIES FOR THERAPEUTIC INTERVENTION Lloyd P. Aiello, MD, PhD ABSTRACT Hyperglycemia is the principal underlying cause of diabetic microvascular complications,
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Treatment for Central-Involved Diabetic Macular Edema in Eyes with Very Good Visual Acuity
 Diabetic Retinopathy Clinical Research Network Treatment for Central-Involved Diabetic Macular Edema in Eyes with Very Good Visual Acuity Version 3.0 April 18, 2014 Treatment of CIDME in Eyes with Good
Diabetic Retinopathy Clinical Research Network Treatment for Central-Involved Diabetic Macular Edema in Eyes with Very Good Visual Acuity Version 3.0 April 18, 2014 Treatment of CIDME in Eyes with Good
Randomized Trial Evaluating Short- Term Effects of Intravitreal Ranibizumab or Triamcinolone Acetonide on Macular
 Randomized Trial Evaluating Short- Term Effects of Intravitreal Ranibizumab or Triamcinolone Acetonide on Macular Edema After Focal/grid Laser for Diabetic Macular Edema in Eyes Also Receiving Panretinal
Randomized Trial Evaluating Short- Term Effects of Intravitreal Ranibizumab or Triamcinolone Acetonide on Macular Edema After Focal/grid Laser for Diabetic Macular Edema in Eyes Also Receiving Panretinal
Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment
 9:30 AM Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment Lee Jampol, MD OBJECTIVE To assess subsequent visual and anatomic outcomes of eyes with
9:30 AM Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment Lee Jampol, MD OBJECTIVE To assess subsequent visual and anatomic outcomes of eyes with
COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY
 Original Article COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY Aggarwal Somesh V 1, Shah Sonali N 2, Bharwada Rekha
Original Article COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY Aggarwal Somesh V 1, Shah Sonali N 2, Bharwada Rekha
A retrospective nonrandomized study was conducted at 3
 Department of Ophthalmology, Kangbuk Samsung Hospital, Sungkyunkwan University College of Medicine 1, Seoul, Korea Hangil Eye Hospital 2, Incheon, Korea Seoul National University Bundang Hospital 3, Seongnam,
Department of Ophthalmology, Kangbuk Samsung Hospital, Sungkyunkwan University College of Medicine 1, Seoul, Korea Hangil Eye Hospital 2, Incheon, Korea Seoul National University Bundang Hospital 3, Seongnam,
Molecular Biology of Diabetic Retinopathy
 Opportunities for Therapeutic Intervention: Molecular Biology of Diabetic Retinopathy Jointly sponsored by The Dulaney Foundation and DIABETIC MICROVASCULAR COMPLICATIONS TODAY. Release Date: January 2006.
Opportunities for Therapeutic Intervention: Molecular Biology of Diabetic Retinopathy Jointly sponsored by The Dulaney Foundation and DIABETIC MICROVASCULAR COMPLICATIONS TODAY. Release Date: January 2006.
Case Report Inherent Challenges in Managing Long Standing Refractory Diabetic Macular Edema
 Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report Inherent Challenges in Managing Long Standing Refractory Diabetic Macular Edema V Swetha E Jeganathan 1,2 * and Karen Madill 3 1 Department of Ophthalmology,
Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report Inherent Challenges in Managing Long Standing Refractory Diabetic Macular Edema V Swetha E Jeganathan 1,2 * and Karen Madill 3 1 Department of Ophthalmology,
EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA
 Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
VISUAL OUTCOME IN DIABETIC MACULAR EDEMA AFTER GRID LASER TREATMENT
 The Professional Medical Journal DOI: 10.17957/TPMJ/16.2856 ORIGINAL PROF-2856 VISUAL OUTCOME IN DIABETIC MACULAR EDEMA AFTER GRID LASER TREATMENT 1. MBBS.FCPS Assistant Professor Ophthalmology Independent
The Professional Medical Journal DOI: 10.17957/TPMJ/16.2856 ORIGINAL PROF-2856 VISUAL OUTCOME IN DIABETIC MACULAR EDEMA AFTER GRID LASER TREATMENT 1. MBBS.FCPS Assistant Professor Ophthalmology Independent
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Subclinical Diabetic Macular Edema Study
 Diabetic Retinopathy Clinical Research Network Subclinical Diabetic Macular Edema Study Version 2.0 March 21, 2006 Subclinical DME Protocol v2 0 3-21-06.doc Table of Contents Chapter 1. Background Information
Diabetic Retinopathy Clinical Research Network Subclinical Diabetic Macular Edema Study Version 2.0 March 21, 2006 Subclinical DME Protocol v2 0 3-21-06.doc Table of Contents Chapter 1. Background Information
INTRODUCTION. Trans Am Ophthalmol Soc 2010;108:62-76
 INTERPRETING THICKNESS CHANGES IN THE DIABETIC MACULA: THE PROBLEM OF SHORT- TERM VARIATION IN OPTICAL COHERENCE TOMOGRAPHY MEASURED MACULAR THICKENING (AN AMERICAN OPHTHALMOLOGICAL SOCIETY THESIS) BY
INTERPRETING THICKNESS CHANGES IN THE DIABETIC MACULA: THE PROBLEM OF SHORT- TERM VARIATION IN OPTICAL COHERENCE TOMOGRAPHY MEASURED MACULAR THICKENING (AN AMERICAN OPHTHALMOLOGICAL SOCIETY THESIS) BY
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
Diabetic Retinopathy: Managing the Extremes. J. Michael Jumper, MD West Coast Retina
 Diabetic Retinopathy: Managing the Extremes J. Michael Jumper, MD West Coast Retina Case 1: EC 65 y.o. HM No vision complaints Meds: Glyburide Metformin Pioglitazone Va: 20/20 OU 20/20 Case 2: HS 68 y.o.
Diabetic Retinopathy: Managing the Extremes J. Michael Jumper, MD West Coast Retina Case 1: EC 65 y.o. HM No vision complaints Meds: Glyburide Metformin Pioglitazone Va: 20/20 OU 20/20 Case 2: HS 68 y.o.
Study of clinical significance of optical coherence tomography in diagnosis & management of diabetic macular edema
 Original Research Article Study of clinical significance of optical coherence tomography in diagnosis & management of diabetic macular edema Neha Kantilal Desai 1,*, Somesh Vedprakash Aggarwal 2, Sonali
Original Research Article Study of clinical significance of optical coherence tomography in diagnosis & management of diabetic macular edema Neha Kantilal Desai 1,*, Somesh Vedprakash Aggarwal 2, Sonali
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network Intravitreous Anti-VEGF Treatment for Prevention of Vision Threatening Diabetic Retinopathy in Eyes at High Risk Version 4.0 September 19, 2017 Anti-VEGF
Diabetic Retinopathy Clinical Research Network Intravitreous Anti-VEGF Treatment for Prevention of Vision Threatening Diabetic Retinopathy in Eyes at High Risk Version 4.0 September 19, 2017 Anti-VEGF
ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS
 ORIGINAL ARTICLE ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS Aggarwal Somesh VP 1, Shah Sonali N 2, Bharwada Rekha M 3,
ORIGINAL ARTICLE ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS Aggarwal Somesh VP 1, Shah Sonali N 2, Bharwada Rekha M 3,
Diagnosis and treatment of diabetic retinopathy. Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City
 Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
The Diabetic Retinopathy Clinical Research Network. Management of DME in Eyes with PDR
 The Diabetic Retinopathy Clinical Research Network Management of DME in Eyes with PDR 1 What Has Been Learned? Diabetic Retinopathy Treatment Protocol F: Results suggest that clinically meaningful differences
The Diabetic Retinopathy Clinical Research Network Management of DME in Eyes with PDR 1 What Has Been Learned? Diabetic Retinopathy Treatment Protocol F: Results suggest that clinically meaningful differences
Paradigm Shift in the treatment of Diabetic Retinopathy. Haytham I. S. Salti, MD Associate Professor
 Paradigm Shift in the treatment of Diabetic Retinopathy Haytham I. S. Salti, MD Associate Professor Disclosure No financial interests related to the subject matter of this talk This presentation includes
Paradigm Shift in the treatment of Diabetic Retinopathy Haytham I. S. Salti, MD Associate Professor Disclosure No financial interests related to the subject matter of this talk This presentation includes
Intravitreal Ranibizumab or Triamcinolone Acetonide in Combination with Laser Photocoagulation for Diabetic Macular Edema
 Diabetic Retinopathy Clinical Research Network Intravitreal Ranibizumab or Triamcinolone Acetonide in Combination with Laser Photocoagulation for Diabetic Macular Edema Version 6.0 May 21, 2010 Note: See
Diabetic Retinopathy Clinical Research Network Intravitreal Ranibizumab or Triamcinolone Acetonide in Combination with Laser Photocoagulation for Diabetic Macular Edema Version 6.0 May 21, 2010 Note: See
International Journal of Health Sciences and Research ISSN:
 International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
Recalcitrant Diabetic Macular Oedema: Therapeutic Options
 December 2007 A. Giridhar et al. - Recalcitrant DME 451 CONSULTATION S E C T I O N Recalcitrant Diabetic Macular Oedema: Therapeutic Options Dr. Cyrus M Shroff 1, Dr. N S Muralidhar 2, Dr. R Narayanan
December 2007 A. Giridhar et al. - Recalcitrant DME 451 CONSULTATION S E C T I O N Recalcitrant Diabetic Macular Oedema: Therapeutic Options Dr. Cyrus M Shroff 1, Dr. N S Muralidhar 2, Dr. R Narayanan
New Developments in the treatment of Diabetic Retinopathy
 New Developments in the treatment of Diabetic Retinopathy B. Jeroen Klevering University Medical Centre Nijmegen - The Netherlands Topics Management of diabetic retinopathy Interventions a. primary (prevention)
New Developments in the treatment of Diabetic Retinopathy B. Jeroen Klevering University Medical Centre Nijmegen - The Netherlands Topics Management of diabetic retinopathy Interventions a. primary (prevention)
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network Prompt Panretinal Photocoagulation Versus Intravitreal Ranibizumab with Deferred Panretinal Photocoagulation for Proliferative Diabetic Retinopathy (Protocol
Diabetic Retinopathy Clinical Research Network Prompt Panretinal Photocoagulation Versus Intravitreal Ranibizumab with Deferred Panretinal Photocoagulation for Proliferative Diabetic Retinopathy (Protocol
 measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
Diabetic Retinopathy A Presentation for the Public
 Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network Prompt Panretinal Photocoagulation Versus Intravitreal Ranibizumab with Deferred Panretinal Photocoagulation for Proliferative Diabetic Retinopathy Version
Diabetic Retinopathy Clinical Research Network Prompt Panretinal Photocoagulation Versus Intravitreal Ranibizumab with Deferred Panretinal Photocoagulation for Proliferative Diabetic Retinopathy Version
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network An Observational Study of the Development of Diabetic Macular Edema Following Scatter Laser Photocoagulation Version 1.4 June 24, 2005 PRP Obs Study Protocol
Diabetic Retinopathy Clinical Research Network An Observational Study of the Development of Diabetic Macular Edema Following Scatter Laser Photocoagulation Version 1.4 June 24, 2005 PRP Obs Study Protocol
Anti Vascular Endothelial Growth Factor Pharmacotherapy for Diabetic Macular Edema
 Ophthalmic Technology Assessment Anti Vascular Endothelial Growth Factor Pharmacotherapy for Diabetic Macular Edema A Report by the American Academy of Ophthalmology Allen C. Ho, MD, Ingrid U. Scott, MD,
Ophthalmic Technology Assessment Anti Vascular Endothelial Growth Factor Pharmacotherapy for Diabetic Macular Edema A Report by the American Academy of Ophthalmology Allen C. Ho, MD, Ingrid U. Scott, MD,
Evaluation of Changes of Macular Thickness in Diabetic Retinopathy after Cataract Surgery
 pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2011;25(4):238-242 DOI: 10.3341/kjo.2011.25.4.238 Evaluation of Changes of Macular Thickness in Diabetic Retinopathy after Cataract Surgery Original
pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2011;25(4):238-242 DOI: 10.3341/kjo.2011.25.4.238 Evaluation of Changes of Macular Thickness in Diabetic Retinopathy after Cataract Surgery Original
Diabetic Macular Edema Treatment in the 21st Century
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Scott M. Pfahler D.O. Dayton Vitreo-Retinal Associates AOCOO-HNS Palm Springs, CA 2012
 Scott M. Pfahler D.O. Dayton Vitreo-Retinal Associates AOCOO-HNS Palm Springs, CA 2012 Proliferative Diabetic Retinopathy Laser Treatments Medical Treatment Surgical Treatment Diabetic Macular Edema Laser
Scott M. Pfahler D.O. Dayton Vitreo-Retinal Associates AOCOO-HNS Palm Springs, CA 2012 Proliferative Diabetic Retinopathy Laser Treatments Medical Treatment Surgical Treatment Diabetic Macular Edema Laser
Abbreviated Drug Evaluation: Fluocinolone acetonide intravitreal implant (Retisert )
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Protocol. This trial protocol has been provided by the authors to give readers additional information about their work.
 Protocol This trial protocol has been provided by the authors to give readers additional information about their work. Protocol for: The Diabetic Retinopathy Clinical Research Network. Aflibercept, bevacizumab,
Protocol This trial protocol has been provided by the authors to give readers additional information about their work. Protocol for: The Diabetic Retinopathy Clinical Research Network. Aflibercept, bevacizumab,
Therapy Quick Guide. Leading MicroPulse surgeons share tissue-sparing laser techniques for the treatment of retinal disorders and glaucoma
 Therapy Quick Guide Leading MicroPulse surgeons share tissue-sparing laser techniques for the treatment of retinal disorders and glaucoma Become a member of the IRIDEX MicroPulse Forum. Learn more at iridex.com/micropulse
Therapy Quick Guide Leading MicroPulse surgeons share tissue-sparing laser techniques for the treatment of retinal disorders and glaucoma Become a member of the IRIDEX MicroPulse Forum. Learn more at iridex.com/micropulse
DIABETIC RETINOPATHY IS
 CLINICAL SCIENCES Agreement of the Heidelberg Retina Tomograph II Macula Edema Module With Fundus Biomicroscopy in Diabetic Maculopathy Mila Kisilevsky, MD; Christopher Hudson, PhD; John G. Flanagan, PhD;
CLINICAL SCIENCES Agreement of the Heidelberg Retina Tomograph II Macula Edema Module With Fundus Biomicroscopy in Diabetic Maculopathy Mila Kisilevsky, MD; Christopher Hudson, PhD; John G. Flanagan, PhD;
Macular edema (ME) is the most common
 MANAGEMENT OF RETINAL VEIN OCCLUSIONS * Peter A. Campochiaro, MD ABSTRACT Macular edema (ME) is the most common cause of reduced vision in patients with retinal vein occlusions (RVOs). The primary cause
MANAGEMENT OF RETINAL VEIN OCCLUSIONS * Peter A. Campochiaro, MD ABSTRACT Macular edema (ME) is the most common cause of reduced vision in patients with retinal vein occlusions (RVOs). The primary cause
Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema
 111 ORIGINAL Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema Toshihiko Nagasawa, Takeshi Naito, Shingo Matsushita, Hiroyuki Sato, Takashi Katome, and
111 ORIGINAL Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema Toshihiko Nagasawa, Takeshi Naito, Shingo Matsushita, Hiroyuki Sato, Takashi Katome, and
Original Policy Date
 MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
Review Article Combination of Anti-VEGF and Laser Photocoagulation for Diabetic Macular Edema: A Review
 Hindawi Ophthalmology Volume 2017, Article ID 2407037, 7 pages https://doi.org/10.1155/2017/2407037 Review Article Combination of Anti-VEGF and Laser Photocoagulation for Diabetic Macular Edema: A Review
Hindawi Ophthalmology Volume 2017, Article ID 2407037, 7 pages https://doi.org/10.1155/2017/2407037 Review Article Combination of Anti-VEGF and Laser Photocoagulation for Diabetic Macular Edema: A Review
Facts About Diabetic Eye Disease
 Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
VERTEPORFIN IN PHOTODYNAMIC THERAPY STUDY GROUP
 Verteporfin Therapy of Subfoveal Choroidal Neovascularization in Age-related Macular Degeneration: Two-year Results of a Randomized Clinical Trial Including Lesions With Occult With No Classic Choroidal
Verteporfin Therapy of Subfoveal Choroidal Neovascularization in Age-related Macular Degeneration: Two-year Results of a Randomized Clinical Trial Including Lesions With Occult With No Classic Choroidal
Subgroup Analysis of the MARINA Study of Ranibizumab in Neovascular Age-Related Macular Degeneration
 Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
 Research Article http://www.alliedacademies.org/clinical-ophthalmology-and-vision-science/ The trial: A pilot study to assess the efficacy, durability, and safety of combination ranibizumab + peripheral
Research Article http://www.alliedacademies.org/clinical-ophthalmology-and-vision-science/ The trial: A pilot study to assess the efficacy, durability, and safety of combination ranibizumab + peripheral
Diabetic Retinopatathy
 Diabetic Retinopatathy Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Arctic DX Macula Risk Advanced
Diabetic Retinopatathy Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Arctic DX Macula Risk Advanced
Optical Coherence Tomography Fast versus Regular Macular Thickness Mapping in Diabetic Retinopathy
 Original Paper DOI: 10.1159/000127830 Received: March 18, 2007 Accepted after revision: July 9, 2007 Published online: April 25, 2008 Optical Coherence Tomography Fast versus Regular Macular Thickness
Original Paper DOI: 10.1159/000127830 Received: March 18, 2007 Accepted after revision: July 9, 2007 Published online: April 25, 2008 Optical Coherence Tomography Fast versus Regular Macular Thickness
Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema
 Original Article Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema Karen Joyce G. Castro, MD, Marie Joan V. Loy, MD International Eye Institute St. Luke s Medical Center
Original Article Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema Karen Joyce G. Castro, MD, Marie Joan V. Loy, MD International Eye Institute St. Luke s Medical Center
Past ocular history. DME Case 1. Patient presents blurred VA. Hemoglobin A1c 11.5% -- patient states sugars have not been in good control
 Past ocular history Patient presents blurred VA DME Case 1 Hemoglobin A1c 11.5% -- patient states sugars have not been in good control PDR with macular edema OU Rishi Singh MD Cleveland Clinic OD OS 1
Past ocular history Patient presents blurred VA DME Case 1 Hemoglobin A1c 11.5% -- patient states sugars have not been in good control PDR with macular edema OU Rishi Singh MD Cleveland Clinic OD OS 1
OCT Assessment of the Vitreoretinal Relationship in CSME
 December 2007 Sonia Rani John et al. - IFIS 375 ORIGINAL ARTICLE OCT Assessment of the Vitreoretinal Relationship in CSME Dr. Manoj S. DNB FRCS, Dr. Unnikrishnan Nair MS DO FRCS, Dr. Gargi Sathish MS Introduction
December 2007 Sonia Rani John et al. - IFIS 375 ORIGINAL ARTICLE OCT Assessment of the Vitreoretinal Relationship in CSME Dr. Manoj S. DNB FRCS, Dr. Unnikrishnan Nair MS DO FRCS, Dr. Gargi Sathish MS Introduction
Diabetic Management beyond traditional risk factors and LDL-C control: Can we improve macro and microvascular risks?
 Retinopathy Diabetes has a negative effect on eyes in many ways, increasing the risk of cataracts for example, but the most common and serious ocular complication of diabetes is retinopathy. Diabetic retinopathy
Retinopathy Diabetes has a negative effect on eyes in many ways, increasing the risk of cataracts for example, but the most common and serious ocular complication of diabetes is retinopathy. Diabetic retinopathy
IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy
 Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy Fawwaz Al Mamoori* Medical Retina Department, Eye Specialty Hospital,
Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy Fawwaz Al Mamoori* Medical Retina Department, Eye Specialty Hospital,
Citation. As Published Publisher. Version
 Effect of Intravitreous Anti Vascular Endothelial Growth Factor Therapy on Choroidal Thickness in Neovascular Age-Related Macular Degeneration Using Spectral-Domain The MIT Faculty has made this article
Effect of Intravitreous Anti Vascular Endothelial Growth Factor Therapy on Choroidal Thickness in Neovascular Age-Related Macular Degeneration Using Spectral-Domain The MIT Faculty has made this article
Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient
 Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient Rova Virgana FK Unpad Pusat Mata Nasional RS Mata Cicendo Bandung Eye Center (Hospital and Clinic) PIT IDI Jabar 2018 Keys Facts from WHO
Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient Rova Virgana FK Unpad Pusat Mata Nasional RS Mata Cicendo Bandung Eye Center (Hospital and Clinic) PIT IDI Jabar 2018 Keys Facts from WHO
Diabetic Retinopathy: Recent Advances in Treatment and Treatment Approaches
 Diabetic Retinopathy: Recent Advances in Treatment and Treatment Approaches Dr. David Wong Associate Professor Retina Specialist, Department of Ophthalmology & Vision Sciences, University of Toronto, Canada
Diabetic Retinopathy: Recent Advances in Treatment and Treatment Approaches Dr. David Wong Associate Professor Retina Specialist, Department of Ophthalmology & Vision Sciences, University of Toronto, Canada
Jay M. Haynie, O.D.; F.A.A.O. Olympia Tacoma Renton Kennewick Washington
 Jay M. Haynie, O.D.; F.A.A.O. Olympia Tacoma Renton Kennewick Washington I Jay M. Haynie, OD, FAAO have received honoraria from the following companies: Reichert Technologies Notal Vision Carl Zeiss Meditec
Jay M. Haynie, O.D.; F.A.A.O. Olympia Tacoma Renton Kennewick Washington I Jay M. Haynie, OD, FAAO have received honoraria from the following companies: Reichert Technologies Notal Vision Carl Zeiss Meditec
Despite our growing knowledge of the
 OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
Is OCT-A Needed As An Investigative Tool During The Management Of Diabetic Macular Edema
 Is OCT-A Needed As An Investigative Tool During The Management Of Diabetic Macular Edema Ayman M Khattab MD, FRCS Professor of Ophthalmology Cairo University Diabetic Macular Edema (DME) Diabetic macular
Is OCT-A Needed As An Investigative Tool During The Management Of Diabetic Macular Edema Ayman M Khattab MD, FRCS Professor of Ophthalmology Cairo University Diabetic Macular Edema (DME) Diabetic macular
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration
 Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration
 A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
The Human Eye. Cornea Iris. Pupil. Lens. Retina
 The Retina Thin layer of light-sensitive tissue at the back of the eye (the film of the camera). Light rays are focused on the retina then transmitted to the brain. The macula is the very small area in
The Retina Thin layer of light-sensitive tissue at the back of the eye (the film of the camera). Light rays are focused on the retina then transmitted to the brain. The macula is the very small area in
CLINICAL SCIENCES. Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration
 CLINICAL SCIENCES Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration Three-Year Results of an Open-Label Extension of 2 Randomized Clinical Trials TAP Report
CLINICAL SCIENCES Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration Three-Year Results of an Open-Label Extension of 2 Randomized Clinical Trials TAP Report
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Navilas Clinical Results: Diabetic Macular Edema Introducing a Better Treatment Paradigm for Patients and Physicians
 Navilas Clinical Results: Diabetic Macular Edema Introducing a Better Treatment Paradigm for Patients and Physicians 16 % Injection-free after loading phase 65 % Injection-free after loading phase Navilas
Navilas Clinical Results: Diabetic Macular Edema Introducing a Better Treatment Paradigm for Patients and Physicians 16 % Injection-free after loading phase 65 % Injection-free after loading phase Navilas
SAFETY OF TRANSFOVEAL SUBTHRESHOLD DIODE MICROPULSE LASER FOR FOVEA-INVOLVING DIABETIC MACULAR EDEMA IN EYES WITH GOOD VISUAL ACUITY
 SAFETY OF TRANSFOVEAL SUBTHRESHOLD DIODE MICROPULSE LASER FOR FOVEA-INVOLVING DIABETIC MACULAR EDEMA IN EYES WITH GOOD VISUAL ACUITY JEFFREY K. LUTTRULL, MD,* STEPHEN H. SINCLAIR, MD Purpose: To determine
SAFETY OF TRANSFOVEAL SUBTHRESHOLD DIODE MICROPULSE LASER FOR FOVEA-INVOLVING DIABETIC MACULAR EDEMA IN EYES WITH GOOD VISUAL ACUITY JEFFREY K. LUTTRULL, MD,* STEPHEN H. SINCLAIR, MD Purpose: To determine
Diabetic maculopathy 11/ An update on. Miss Vasuki Sivagnanavel
 Miss Vasuki Sivagnanavel Consultant Ophthalmologist An update on Diabetic maculopathy Despite advances in the management of diabetes, diabetic retinopathy is already the commonest cause of blindness among
Miss Vasuki Sivagnanavel Consultant Ophthalmologist An update on Diabetic maculopathy Despite advances in the management of diabetes, diabetic retinopathy is already the commonest cause of blindness among
Effects of Dilation on Electronic-ETDRS Visual Acuity in Diabetic Patients
 Effects of Dilation on Electronic-ETDRS Visual Acuity in Diabetic Patients The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation
Effects of Dilation on Electronic-ETDRS Visual Acuity in Diabetic Patients The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation
Original Article. Authors
 Original Article APMC-303 Efficacy of Intra-Vitreal Bevacizumab Combined with Phaco- Emulsification in the Prophylaxis of Macular Edema in Patients with Non-Proliferative Diabetic Retinopathy Muhammad
Original Article APMC-303 Efficacy of Intra-Vitreal Bevacizumab Combined with Phaco- Emulsification in the Prophylaxis of Macular Edema in Patients with Non-Proliferative Diabetic Retinopathy Muhammad
Instudies of vascular endothelial growth factor
 MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
Ranibizumab and Bevacizumab for Treatment of Neovascular Age-related Macular Degeneration
 and for Treatment of Neovascular Age-related Macular Degeneration Two-Year Results Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group* Writing Committee: Daniel F. Martin,
and for Treatment of Neovascular Age-related Macular Degeneration Two-Year Results Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group* Writing Committee: Daniel F. Martin,
R&M Solutions
 Mohamed Hosny El-Bradey, MD., Assistant Professor of Ophthalmology, Tanta University. Wael El Haig, MD., Professor of Ophthalmology. Zagazeeg University. 1 Myopic CNV is considered the most common vision
Mohamed Hosny El-Bradey, MD., Assistant Professor of Ophthalmology, Tanta University. Wael El Haig, MD., Professor of Ophthalmology. Zagazeeg University. 1 Myopic CNV is considered the most common vision
Factors Associated with Visual Outcome after Photocoagulation for Diabetic Retinopathy
 Investigative Ophthalmology & Visual Science, Vol. 30, No. 1, January 1989 Copyright Association for Research in Vision and Ophthalmology Factors Associated with Visual Outcome after Photocoagulation for
Investigative Ophthalmology & Visual Science, Vol. 30, No. 1, January 1989 Copyright Association for Research in Vision and Ophthalmology Factors Associated with Visual Outcome after Photocoagulation for
NEOVASCULAR AGE-RELATED MACULAR DEGENERation
 Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Retinal vein occlusion (RVO) is a vascular disease
 INTRAVITREAL RANIBIZUMAB FOR RETINAL VEIN OCCLUSION THROUGH 1 YEAR IN CLINICAL PRACTICE TROELS BRYNSKOV, MD,* HENRIK KEMP, MD,* TORBEN L. SØRENSEN, MD, DMSC* Purpose: To evaluate the efficacy and safety
INTRAVITREAL RANIBIZUMAB FOR RETINAL VEIN OCCLUSION THROUGH 1 YEAR IN CLINICAL PRACTICE TROELS BRYNSKOV, MD,* HENRIK KEMP, MD,* TORBEN L. SØRENSEN, MD, DMSC* Purpose: To evaluate the efficacy and safety
Diabetes mellitus: A risk factor affecting visual outcome in branch retinal vein occlusion
 European Journal of Ophthalmology / Vol. 13 no. 7, 2003 / pp. 648-652 Diabetes mellitus: A risk factor affecting visual outcome in branch retinal vein occlusion J. SWART 1,2, J.W. REICHERT-THOEN 1, M.S.
European Journal of Ophthalmology / Vol. 13 no. 7, 2003 / pp. 648-652 Diabetes mellitus: A risk factor affecting visual outcome in branch retinal vein occlusion J. SWART 1,2, J.W. REICHERT-THOEN 1, M.S.
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network Intravitreal Ranibizumab or Triamcinolone Acetonide as Adjunctive Treatment to Panretinal Photocoagulation for Proliferative Diabetic Retinopathy Version
Diabetic Retinopathy Clinical Research Network Intravitreal Ranibizumab or Triamcinolone Acetonide as Adjunctive Treatment to Panretinal Photocoagulation for Proliferative Diabetic Retinopathy Version
The limited number of currently approved
 CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
A Phase 2 Evaluation of Anti-VEGF Therapy for Diabetic Macular Edema: Bevacizumab (Avastin)
 Diabetic Retinopathy Clinical Research Network A Phase 2 Evaluation of Anti-VEGF Therapy for Diabetic Macular Edema: Bevacizumab (Avastin) Version1.2 April 27, 2006 Avastin Protocol 4-27-06 V1.2.doc Contact
Diabetic Retinopathy Clinical Research Network A Phase 2 Evaluation of Anti-VEGF Therapy for Diabetic Macular Edema: Bevacizumab (Avastin) Version1.2 April 27, 2006 Avastin Protocol 4-27-06 V1.2.doc Contact
Keiji Inagaki, 1,2 Kishiko Ohkoshi, 1 Sachiko Ohde, 3 Gautam A. Deshpande, 3 Nobuyuki Ebihara, 2 and Akira Murakami 2. 1.
 Journal of Ophthalmology, Article ID 251257, 10 pages http://dx.doi.org/10.1155/2014/251257 Clinical Study Subthreshold Micropulse Photocoagulation for Persistent Macular Edema Secondary to Branch Retinal
Journal of Ophthalmology, Article ID 251257, 10 pages http://dx.doi.org/10.1155/2014/251257 Clinical Study Subthreshold Micropulse Photocoagulation for Persistent Macular Edema Secondary to Branch Retinal
PART 1: GENERAL RETINAL ANATOMY
 PART 1: GENERAL RETINAL ANATOMY General Anatomy At Ora Serrata At Optic Nerve Head Fundoscopic View Of Normal Retina What Is So Special About Diabetic Retinopathy? The WHO definition of blindness is
PART 1: GENERAL RETINAL ANATOMY General Anatomy At Ora Serrata At Optic Nerve Head Fundoscopic View Of Normal Retina What Is So Special About Diabetic Retinopathy? The WHO definition of blindness is
ILUVIEN IN DIABETIC MACULAR ODEMA
 1 ILUVIEN IN DIABETIC MACULAR ODEMA Marie Tsaloumas Consultant Ophthalmic Surgeon Queen Elizabeth Hospital, Birmingham bars conference 2104 1 2 Declaration of interest I have sat on Advisory boards for
1 ILUVIEN IN DIABETIC MACULAR ODEMA Marie Tsaloumas Consultant Ophthalmic Surgeon Queen Elizabeth Hospital, Birmingham bars conference 2104 1 2 Declaration of interest I have sat on Advisory boards for
Retinopathy in a diabetic population
 Kathmandu University Medical Journal (2007), Vol. 5, No. 2, Issue 18, 204-209 Retinopathy in a diabetic population Shrestha S 1, Malla OK 2, Karki DB 3, Byanju RN 4 2 Fellow of NAMS, 3 Professor, NAMS,
Kathmandu University Medical Journal (2007), Vol. 5, No. 2, Issue 18, 204-209 Retinopathy in a diabetic population Shrestha S 1, Malla OK 2, Karki DB 3, Byanju RN 4 2 Fellow of NAMS, 3 Professor, NAMS,
Neuropathy (NAION) and Avastin. Clinical Assembly of the AOCOO-HNS Foundation May 9, 2013
 Non Arteritic Ischemic Optic Neuropathy (NAION) and Avastin Shalom Kelman, MD Clinical Assembly of the AOCOO-HNS Foundation May 9, 2013 Anterior Ischemic Optic Neuropathy Acute, painless, visual loss,
Non Arteritic Ischemic Optic Neuropathy (NAION) and Avastin Shalom Kelman, MD Clinical Assembly of the AOCOO-HNS Foundation May 9, 2013 Anterior Ischemic Optic Neuropathy Acute, painless, visual loss,
Disease-Specific Fluorescein Angiography
 Ruth E. Picchiottino, CRA Disease-Specific Fluorescein Angiography 15 Disease-Specific Fluorescein Angiography Recommendations for tailoring retinal fluorescein angiography to diabetic retinopathy, macular
Ruth E. Picchiottino, CRA Disease-Specific Fluorescein Angiography 15 Disease-Specific Fluorescein Angiography Recommendations for tailoring retinal fluorescein angiography to diabetic retinopathy, macular
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network Peripheral Diabetic Retinopathy (DR) Lesions on Ultrawide-field Fundus Images and Risk of DR Worsening Over Time Version 4.0 July 21, 2017 peripheral dr lesions
Diabetic Retinopathy Clinical Research Network Peripheral Diabetic Retinopathy (DR) Lesions on Ultrawide-field Fundus Images and Risk of DR Worsening Over Time Version 4.0 July 21, 2017 peripheral dr lesions
Intravitreal bevacizumab for pediatric exudative retinal diseases
 Saudi Journal of Ophthalmology (2011) 25, 193 197 King Saud University Saudi Journal of Ophthalmology www.saudiophthaljournal.com www.ksu.edu.sa www.sciencedirect.com ORIGINAL ARTICLE Intravitreal bevacizumab
Saudi Journal of Ophthalmology (2011) 25, 193 197 King Saud University Saudi Journal of Ophthalmology www.saudiophthaljournal.com www.ksu.edu.sa www.sciencedirect.com ORIGINAL ARTICLE Intravitreal bevacizumab
Ophthalmic VEGF Inhibitors. Eylea (aflibercept), Macugen (pegaptanib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
