Clinical Study Effects of Intravitreal Ranibizumab Injection on Chinese Patients with Wet Age-Related Macular Degeneration: 5-Year Follow-Up Results
|
|
|
- Hector Mills
- 5 years ago
- Views:
Transcription
1 Ophthalmology Volume 2016, Article ID , 5 pages Clinical Study Effects of Intravitreal Ranibizumab Injection on Chinese Patients with Wet Age-Related Macular Degeneration: 5-Year Follow-Up Results Yingyi Lu, Jianfeng Huang, Jing Zhao, Xiaobing Yu, Li Long, and Hong Dai Department of Ophthalmology, Beijing Hospital, National Center of Gerontology, No. 1 Dahua Road, Dongdan, Beijing , China Correspondence should be addressed to Hong Dai; dai-hong@x263.net Received 23 March 2016; Revised 23 August 2016; Accepted 30 August 2016 Academic Editor: Theodore Leng Copyright 2016 Yingyi Lu et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Purpose. To observe the effect of intravitreal ranibizumab injection on wet age-related macular degeneration (wamd) over 5 years in Chinese patients. Methods. Thirty-seven patients who were diagnosed with wamd in our hospital from June 2007 to June 2014 were retrospectively reviewed. The PRN regimen and the treatment and extend regimen were applied. Best corrected visual acuity (BCVA), number of ranibizumab injections, and changes in the choroidal neovascularization (CNV) lesion over 5 years were analyzed. Results. The mean BCVA measured by the ETDRS chart at baseline was 47.4 and 5 years after the treatment it was letters, which was significantly different (p = 0.013). Fourteen eyes (37.8%) had improved visual acuity after 5 years. The number of injections in 5 years was 11.53, and most of the injections were in the first two years. Seventeen (45.9%) cases developed fibrous lesions, and 2 (5.4%) cases had atrophic lesions after 5 years. The fibrosis/atrophy was significantly correlated with the injection numbers (Pearson, r = 0.663,andp = 0.000). Conclusion. Most of the patients can maintain visual acuity treated by ranibizumab in the first 3 years. After 5 years, some patients can still improve or maintain visual acuity. Fibrous scarring of the lesion is the main reason for a decrease in vision of wamd patients. 1. Introduction Age-related macular degeneration (AMD) is the most important cause of blindness in aged populations in developed countries. As the aging population and average life expectancy increase, the incidence of AMD is growing in developing countries[1].theefficacyandsafetyoftheanti-vegfmedicine, ranibizumab (Lucentis), to treat wet age-related macular degeneration (wamd) have been demonstrated in many large samples, multicenter randomized clinical trials, and substantial clinical practice [2, 3]. It is now one of the most widely used treatments for wamd and choroidal neovascularization (CNV) secondary to many other reasons [4 6]. We arethefirsttoreporttheuseofranibizumabtotreatwamd in Chinese patients, and the aim of this study is to summarize the long-term effect of ranibizumab on wamd over a period of 5 years in Chinese patients. 2. Materials and Methods 2.1. Subjects. Thirty-seven eyes from 37 patients, who were treated with intravitreal ranibizumab injection in Ophthalmology Department of Beijing Hospital from June 2007 to June 2014, were retrospectively analyzed. The inclusion criteria included (1) corresponding to the diagnostic standard of wamd, (2) being able to be followed up promptly according to the treatment protocol, (3) having a follow-up time of more than 60 months, and (4) no other anti-vegf drugs orothertreatmentsduringfollow-up.atbaseline,alleyes had an activating lesion, which presented with retinal edema, thickening, or patched retinal bleeding. FFA/ICGA showed CNV leakage. All lesions were smaller than 4 DA, 14 (37.8%) of which were accompanied by PED Examination. All patients were examined by the ETDRS chart for visual acuity, ophthalmoscope, FFA, ICGA, and
2 2 Ophthalmology OCT on their first visits. The ETDRS visual acuity, ophthalmoscope, and OCT were examined on every follow-up. FFA/ICGA was selectively examined. 40 The BCVA at baseline and in 5th year after treatment 2.3. Treatment Method. Intravitreal ranibizumab injection was performed after diagnosis and after informed consent was signed. Ranibizumab (0.5 mg) was injected according to the standard protocol. The anterior segment, IOP, and routine fundus test were examined within the first week after the injection to survey the operation-related complications Treatment Protocol and Follow-Up. The treatment was injected monthly 3 times at the loading dose and then every month thereafter. Retreatment was given based on the examination results. In the recent 2 years, the follow-up was instructedbythet&eprotocol,inwhichthefollow-uptime was gradually extended for 2 weeks up to more than 3 months if the lesion was stabilized. If the lesion relapsed, the followup went back to once a month again. In the first two years, according to the standard mentioned in the PrONTO study, wechosetoretreatthepatientswhen2ofthefollowing conditions were met: (1) the BCVA decreased more than 5 letters or the VA decreased subjectively; (2) subretinal or intraretinal fluid accumulation on the macula area was measured by OCT; (3) there was a new hemorrhage lesion on the macula; (4) CNV leakage increased or a new lesion developedaccordingtoffaand/oricga.inthelatestage, which was 2 years later, we choose to retreat the patients according to the activation of the lesion as follows: relapse or accumulation of intraretinal or subretinal fluid and new onset hemorrhagelesionrelatedtothevadecrease.weperformed follow-up in the patients with stable PED or a prolonged intraretinal cyst. The follow-up time was months (average 67.4). During follow-up, 5 eyes were injected with a higher dose (1.0 mg, 0.1 ml) for an increasingly shortened relapse interval. The retreatment criteria are patients with persistent recurrent macular intraretinal or subretinal fluid within 30 days Statistical Analysis. AlldatawereanalyzedbySPSS16. One-way analysis of variance was used to analyze the BCVA at baseline and at every year after treatment. The relationship between the fibrosis/atrophy of the lesion in the 5th year after treatment and the injection times was analyzed by Pearson correlation analysis. A p value < 0.05 was considered statistically significant. 3. Results 3.1. Patients. There were 22 male patients and 15 female patients, and there were 17 right eyes and 20 left eyes. The age ofthepatientsrangedfrom59to82years,andtheaverage age was 74.5 ± 6.0 years. Nine of the 37 eyes (24.3%) had a PDT, avastin, or TA treatment history. Twenty-eight eyes were treatment-naïve. The VA of 37 patients before treatment ranged from 0.01 to 0.1. Among them, the VA of 8 eyes (21.6%) was 0.5, the VA of 21 eyes (56.8%) was between 0.1 and 0.4, and the VA of 8 eyes (21.6%) was 0.1. The mean BCVA Number of cases BCVA Baseline Treatment 5th year Figure 1: The distribution of the mean BCVAs before treatment and 5 years after treatment. Table 1: Visual acuity gain or loss at the 5th year after treatment. Visual acuity ETDRS VA at the 5th year after treatment (n =37), n% Gain 15 letters 5 (13.5%) Gain within 15 letters 9 (24.3%) Loss within 15 letters 10 (27.0%) Loss 15 letters 13 (35.1%) examined using the ETDRS chart was 47.4 letters before treatment. Five years after the treatment, the VA of 6 eyes (16.2%) was 0.5, the VA of 16 eyes (43.2%) was between 0.1 and0.4,andthevaof15eyes(40.5%)was 0.1 (Figure 1). The mean BCVA examined by the ETDRS chart was letters (Table 1). There was no statistically significant difference between the BCVA at baseline and that in the first 3 years after treatment (p = 1.314,0.595,and0.148).TheBCVAatbaseline and in the 5th year after the treatment had statistically significant difference (p = 0.013). The BCVA in the 5th year after the treatment was significantly different (p = 0.024) from that in the first 2 years after the treatment (p = 0.03 and 0.025) (Figure 2) Outcome of the Lesions. The lesions were estimated by fundus image and OCT 5 years after the treatment to check the formation of geographic atrophy and/or fibrotic scar. The lesion was stable at the first year after the treatment in 5 of the 36 patients; the VA increased and there was no recurrence. Twenty-six eyes (70.3%) had no treatment after
3 Ophthalmology 3 ETDRS The mean ETDRS letter score over 5 years (year) Error bars: 95% CI Figure 2: The mean BCVAs examined by the ETDRS chart at baseline and at every year after treatment. Follow-up visit Table 2: The mean treatment times in 5 years. Total number receiving injection (n =37) 1st year 5.93 ± nd year 3.41 ± rd year 0.91 ± th year 0.74 ± th year 0.54 ± years because the lesion was stable or became a fibrous scar. Four eyes (10.8%) still needed uninterruptible treatment becauseofactivationofthelesionafter5years.fiveyearsafter the treatment, 17 eyes (47.22%) had fibrosis, and 2 eyes (5.4%) hadanatrophicchange.thefibrosis/atrophyofthecnv lesion 5 years after the treatment was significantly correlated with the intravitreal injection times (Pearson, r = 0.663,and p = 0.000) Treatment Times. The number of injections ranged from 3 to 31 in 5 years, and the average number was The injection was concentrated in the first 2 years, and, from the third year on, the injection number was gradually reduced (Table 2). Five patients (13.5%) underwent more than 20 injections, 18 (48.6%) patients underwent injections, and 14 (37.8%) patients underwent fewer than 10 injections. 4. Discussion Our previous reports showed that the use of ranibizumab to treat wamd in Chinese people has clear short-term efficacy and safety [7 9]. The long-term follow-up results of this study demonstrate that although the average BCVA decreased by 13 letters compared with the BCVA at baseline after 5- year treatment with ranibizumab, 37.8% of the patients had improved vision and 13.5% among whom gain 15 letters or more, indicating that the long-term efficacy of ranibizumab to treat wamd in Chinese people is clear and definite. Our results are similar to the other researches [10, 11]. We found that the average BCVA in the first, second, and third years had no significant difference compared with that at baseline, indicating that most of the patients can maintain their visual function in the first three years, results which are similar to other reports [12]. In the late stage, the average BCVA significantly decreased, which coincided with the change in the CNV lesion. The proportion and extent of fibrosis of the CNV lesion increased with prolonged follow-up. The CNVlesionin46%ofthepatientsinthisstudyshowed fibrosis, while only 5.4% had atrophy. The proportion and extent of fibrosis in Chinese people are much higher than those in other reports [13, 14], whether because some patients had previously undergone PDT treatment or because the different pathophysiological changes in Chinese people are not yet clear. Eyes that received monthly ranibizumab had a higher incidence of geographic atrophy when compared with PRN treatment [15]. This was similar to the results of our PRN treatment, in which the geographic atrophy was less than the fibrosis. This decrease in vision was accompanied by expansion of the size of the total neovascular complex comprising neovascularization, scarring, and atrophy and by persistence of fluid on OCT [15]. We also found that the number of injections is correlated with fibrosis. Fibrosis may beduetomultiplefactors,andtheexactcausehasyetto be studied. Fibrous scar is the major reason for a decline of vision in wamd patients, and reducing or avoiding fibrosis is an effective approach to improving vision, suggesting that combined therapy with antifibrosis might be the new trend for CNV treatment. Our results demonstrate that the average injection time is nearly 12, and 13.5% of patients were injected more than 12 times, while 37.2% were injected fewer than 10 times. Our average injection time was less than that of CATT study, in which the number was 15.4 [15]. Patients who undergo fewer injections can be classified into 2 subtypes according to the CNV features. First, when the lesion is small, treatment is effective, and patients rarely relapse. Second, when the lesion becomes a fibrous scar, the treatment is stopped. From a statistical point of view, most injections occur during the first year and second year, which is related to the increase in CNV fibrosis in some of the patients. Our results show that 10% of patients need treatment after 5 years due to lesion activation, indicating that some wamd patients require long-term, continuous treatment [9]. In some patients, the repetitive injection interval is shorter with increases in the injection times, and we increased ranibizumab dose to 1.0 mg, which can extend the efficacy period, suggesting that some patients may be resistant to anti-vegf medicine after longterm application. Another study from our group indicates that the probable anti-vegf antibody can affect the efficacy [16 18]. During treatment, an individualized regime that is based on the patient s reaction is needed [19, 20]. For anti- VEGF drugs that have poor outcome, increasing the dosage, changing the drug, and combined treatment can be applied [21].
4 4 Ophthalmology In the WAMD treatment guidelines, the follow-up time for patients is recommended as once per month. In clinical practice, especially during long-term follow-up, once per month is a high burden for both patients and doctors [22]. In the early stage, the patients are followed up once a month, for those whose CNV lesion is relatively steady and who can use the Amsler grid by themselves, so the T&E follow-up program is adopted. In the cases where there are stable visual acuity, an absence of macular hemorrhage, and a dry OCT, thefollow-upintervalsincreaseto6weekswithouttreatment. If there are no changes, the next visit is scheduled for 8 weeks later. If there is a change, the patient comes for injection and examination after 4 weeks. Practice shows that this program not only reduces the number of follow-up visits but also can promptly detect the change in the CNV, suggesting that it can be used in clinical practice. wamd patients commonly have age-related cataracts [23]. With the increase in the number of injections or prolonged follow-up time, cataracts will develop and impair the patients visual function. Patients and doctors should be concerned about whether cataract surgery can improve the vision of wamd patients, whether the surgery can influence the CNV lesion, and when to perform the surgery. In our study, 7 cases underwent cataract surgery combined with IOL implantation during the 5 years of follow-up, and most of the patients had improved visual function or visual clarity. Another study of ours demonstrated that half of the wamd patients have improved visual acuity after cataract surgery, and70%ofthepatientshaveimprovedvisualclarityorvisual field vision. The immediate postoperative OCT examination did not reveal any CNV lesion changes. For some patients, the anti-vegf drug injection and cataract surgery were performed simultaneously, and there was no interaction between them. In wamd patients, if a significant cataract develops, surgery can be performed even if the CNV lesion isactive.however,becauseoftheuncertaintyofimproved visual acuity after cataract surgery, a preoperative visual prognosis and sufficient explanation are needed. Although the impact on visual acuity is multifactorial during long-term follow-up and the implementation of the treatment program is not uniform because different doctors and patients can affect the results of this study, our data reflects clinical treatment and can provide a reference for clinical use. Competing Interests The authors declare that they have no competing interests. Acknowledgments This work was supported by the National Natural Science Foundation of China (NSFC Grant ), the Beijing Natural Science Foundation (BNSF Grant ), and the Beijing Municipal Science & Technology Commission (the Capital Characteristic Clinic Project Z ). References [1] J.P.Real,G.E.Granero,M.O.DeSantisetal., Rateofvision loss in neovascular age-related macular degeneration explored, Graefe s Archive for Clinical and Experimental Ophthalmology, vol. 253, no. 11, pp , [2]S.B.Bloch,M.LaCour,B.Sanderetal., Predictorsof1- year visual outcome in neovascular age-related macular degeneration following intravitreal ranibizumab treatment, Acta Ophthalmologica,vol.91,no.1,pp.42 47,2013. [3] K. M. Droege, A. Caramoy, A. Kersten et al., Patient preference of ranibizumab treatment regimen for neovascular age-related macular degeneration monthly injections versus pro re nata, Graefe s Archive for Clinical and Experimental Ophthalmology, vol. 252, no. 1, pp , [4] J.F.Huang,T.Chen,Y.Y.Lu,L.Long,andH.Dai, Retinoschisis and intravitreal ranibizumab treatment for myopic choroidal neovascularization, Chinese Medical Journal,vol.127,no.11,pp , [5] X. Yang and H. Dai, Intravitreal ranibizumab for the treatment of pathological myopia associated with choroidal neovascularization in Chinese patients, Chinese Medical Journal, vol. 127, no.16,pp ,2014. [6]S.Rofagha,R.B.Bhisitkul,D.S.Boyer,S.R.Sadda,andK. Zhang, Seven-year outcomes in ranibizumab-treated patients in ANCHOR, MARINA, and HORIZON: a multicenter cohort study (SEVEN-UP), Ophthalmology,vol.120,no.11,pp , [7] H. Dai, Issues raised in clinical researches using anti-vascular endothelial growth factor drugs to treat choroidal neovascularization, Chinese Ocular Fundus Diseases,vol.27,no. 6, pp , [8] H. Dai, M. Yue, X. Yang et al., Clinical observations of intravitreal injection of Ranibizumab combined with photodynamic therapy for exudative age related macular degeneration, Chinese Optometry Ophthalmology and Visual Science, vol. 13, no. 2, pp , [9] H. Dai, X. B. Yu, and L. Long, Intravitreal Ranibizumab regimens for exudative aged-related macular degeneration, Chinese Ocular Fundus Diseases,vol.26,no.1,2010. [10]C.I.FrennessonandS.E.G.Nilsson, Athree-yearfollowup of ranibizumab treatment of exudative AMD: impact on the outcome of carrying forward the last acuity observation in drop-outs, Acta Ophthalmologica, vol.92,no.3,pp , [11] F. Ziemssen, N. Eter, S. Fauser et al., Retrospective investigation of anti-vegf treatment reality and effectiveness in patients with neovascular age-related macular degeneration (AMD) in Germany: treatment reality of ranibizumab for neovascular AMD in Germany, Ophthalmologe,vol.112,no.3,pp , [12] N. Rayess, S. K. Houston III, O. P. Gupta, A. C. Ho, and C. D. Regillo, Treatment outcomes after 3 years in neovascular age-related macular degeneration using a treat-and-extend regimen, American Ophthalmology,vol.159,no.1,pp. 3 8.e1, [13] E.Daniel,C.A.Toth,J.E.Grunwaldetal., Riskofscarinthe comparison of Age-related Macular Degeneration Treatments Trials, Ophthalmology, vol. 121, no. 3, pp , [14] P. J. Rosenfeld, H. Shapiro, L. Tuomi, M. Webster, J. Elledge, and B. Blodi, Characteristics of patients losing vision after 2 years
5 Ophthalmology 5 of monthly dosing in the phase III ranibizumab clinical trials, Ophthalmology,vol.118,no.3,pp ,2011. [15] G. M. Maureen, F. M. Daniel, and Y. Gui-shuang, Fiveyear outcomes with anti-vascular endothelial growth factor treatment of neovascular age-related macular degeneration: the comparison of age-related macular degeneration treatments trials, Ophthalmology,vol.123,no.8,pp ,2016. [16] H. Dai and Y. Y. Lu, Influence factors and strategies of anti- VEGF therapy for wet AMD, Chinese Experimental Ophthalmology,vol.34,no.1,pp.1 4,2016. [17] H. Dai and Y. Y. Lu, Overall assessment of the factors influencing the effect of anti-vascular endothelial growth factor for neovascular age-related macular degeneration to improve the comprehensive benefit of treatment, Chinese Ocluar Fundus Diseases,vol.32,no.1,pp.8 11,2016. [18] X. Gu, X. Yu, and H. Dai, Intravitreal injection of ranibizumab for treatment of age-related macular degeneration: effects on serum VEGF concentration, Current Eye Research,vol.39,no. 5, pp , [19] P. Lanzetta, P. Mitchell, S. Wolf, and D. Veritti, Different antivascular endothelial growth factor treatments and regimens and their outcomes in neovascular age-related macular degeneration: a literature review, British Ophthalmology, vol. 97, no. 12, pp , [20] S. Pagliarini, S. Beatty, B. Lipkova et al., A 2-year, phase IV, multicentre, observational study of ranibizumab 0.5 mg in patients with neovascular age-related macular degeneration in routine clinical practice: the EPICOHORT study, Ophthalmology, vol. 2014, Article ID , 9 pages, [21] C. K. Chan, P. Abraham, D. Sarraf, A. S. D. Nuthi, S. G. Lin, and C. A. McCannel, Earlier therapeutic effects associated with high dose (2.0 mg) ranibizumab for treatment of vascularized pigment epithelial detachments in age-related macular degeneration, Eye,vol.29, no.1,pp , [22] O. P. Gupta, G. Shienbaum, A. H. Patel, C. Fecarotta, R. S. Kaiser, and C. D. Regillo, A treat and extend regimen using ranibizumab for neovascular age-related macular degeneration: clinical and economic impact, Ophthalmology, vol. 117, no. 11, pp ,2010. [23] R. Klein and B. E. Klein, The prevalence of age-related eye diseases and visual impairment in aging: current estimates, InvestigativeOphthalmology&VisualScience,vol.54,no.14,p. 13, 2013.
6 MEDIATORS of INFLAMMATION The Scientific World Journal Gastroenterology Research and Practice Diabetes Research International Endocrinology Immunology Research Disease Markers Submit your manuscripts at BioMed Research International PPAR Research Obesity Ophthalmology Evidence-Based Complementary and Alternative Medicine Stem Cells International Oncology Parkinson s Disease Computational and Mathematical Methods in Medicine AIDS Behavioural Neurology Research and Treatment Oxidative Medicine and Cellular Longevity
Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular Degeneration
 Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration
 A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements
 Perspective DOI: 10.1167/tvst.4.3.6 Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements Irmela Mantel 1,2 1 Department
Perspective DOI: 10.1167/tvst.4.3.6 Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements Irmela Mantel 1,2 1 Department
Clinical Trials Related to Age Related Macular Degeneration
 Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical setting
 Korb et al. BMC Ophthalmology 2013, 13:84 RESEARCH ARTICLE Open Access Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical
Korb et al. BMC Ophthalmology 2013, 13:84 RESEARCH ARTICLE Open Access Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical
Management of Neovascular AMD
 Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Anti-VEGF drugs & CATT Results. in AMD. Apollo Hospitals, Hyderabad. Mallika Goyal, MD
 Anti-VEGF drugs & CATT Results in AMD Mallika Goyal, MD Apollo Hospitals, Hyderabad Vascular Endothelial Growth Factor Napoleone Ferrara at Genentech 1989 identified & cloned the gene Protein that causes
Anti-VEGF drugs & CATT Results in AMD Mallika Goyal, MD Apollo Hospitals, Hyderabad Vascular Endothelial Growth Factor Napoleone Ferrara at Genentech 1989 identified & cloned the gene Protein that causes
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Retinal Pigment Epithelial Tears (Rips) in the ERA of Anti Vegf - When and Why?
 Original Article Retinal Pigment Epithelial Tears (Rips) in the ERA of Anti Vegf - When and Why? Dr Sreelekha S. MS, DNB, DO, FRCS, Dr Manoj Soman DNB, FICO, FRCS, Dr. Unni Nair MS, FRCS, Dr. Ruminder
Original Article Retinal Pigment Epithelial Tears (Rips) in the ERA of Anti Vegf - When and Why? Dr Sreelekha S. MS, DNB, DO, FRCS, Dr Manoj Soman DNB, FICO, FRCS, Dr. Unni Nair MS, FRCS, Dr. Ruminder
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
London Medicines Evaluation Network Review
 London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
Instudies of vascular endothelial growth factor
 MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014
 From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
JasonC.S.Yam, 1 Gabriela S. L. Chong, 2 Patrick K. W. Wu, 2 Ursula S. F. Wong, 2 Clement W. N. Chan, 2 and Simon T. C. Ko 2. 1.
 BioMed Research International, Article ID 482093, 4 pages http://dx.doi.org/10.1155/2014/482093 Research Article Predictive Factors Affecting the Short Term and Long Term Exodrift in Patients with Intermittent
BioMed Research International, Article ID 482093, 4 pages http://dx.doi.org/10.1155/2014/482093 Research Article Predictive Factors Affecting the Short Term and Long Term Exodrift in Patients with Intermittent
Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials
 Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials Glenn J. Jaffe, MD, 1 Daniel F. Martin, MD, 2 Cynthia A. Toth, MD, 1 Ebenezer Daniel, MPH, PhD,
Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials Glenn J. Jaffe, MD, 1 Daniel F. Martin, MD, 2 Cynthia A. Toth, MD, 1 Ebenezer Daniel, MPH, PhD,
The Era of anti- - - VEGF Kirk L. Halvorson, OD
 The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY
 EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION
 THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION MOESTIDJAB DEPARTMENT OF OPHTHALMOLOGY SCHOOL OF MEDICINE AIRLANGGA UNIVERSITY DR SOETOMO HOSPITAL SURABAYA INTRODUCTION
THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION MOESTIDJAB DEPARTMENT OF OPHTHALMOLOGY SCHOOL OF MEDICINE AIRLANGGA UNIVERSITY DR SOETOMO HOSPITAL SURABAYA INTRODUCTION
Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice
 Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice Andrew A. Moshfeghi, MD, MBA 1 ; John D. Pitcher, MD 2,3 ; Genevieve Lucas, B. Comm. 4 ; Nick
Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice Andrew A. Moshfeghi, MD, MBA 1 ; John D. Pitcher, MD 2,3 ; Genevieve Lucas, B. Comm. 4 ; Nick
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Case Report Optic Disk Pit with Sudden Central Visual Field Scotoma
 Case Reports in Ophthalmological Medicine Volume 2016, Article ID 1423481, 4 pages http://dx.doi.org/10.1155/2016/1423481 Case Report Optic Disk Pit with Sudden Central Visual Field Scotoma Nikol Panou
Case Reports in Ophthalmological Medicine Volume 2016, Article ID 1423481, 4 pages http://dx.doi.org/10.1155/2016/1423481 Case Report Optic Disk Pit with Sudden Central Visual Field Scotoma Nikol Panou
Effectiveness of Ranibizumab for Neovascular Age-related Macular Degeneration using Clinician Determined Retreatment Strategy
 Effectiveness of Ranibizumab for Neovascular Age-related Macular Degeneration using Clinician Determined Retreatment Strategy Anupma Kumar, Jayashree N Sahni, Alexandros N Stangos, Claudio Campa, Simon
Effectiveness of Ranibizumab for Neovascular Age-related Macular Degeneration using Clinician Determined Retreatment Strategy Anupma Kumar, Jayashree N Sahni, Alexandros N Stangos, Claudio Campa, Simon
Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis
 Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis G Pron August 2014 Ontario Health Technology Assessment Series; Vol. 14:
Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis G Pron August 2014 Ontario Health Technology Assessment Series; Vol. 14:
Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis)
 Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes
 Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes Daniel E. Charles, MD, Martin Charles, MD, Gisela Jelusich,
Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes Daniel E. Charles, MD, Martin Charles, MD, Gisela Jelusich,
The Foundation WHAT IS THE RETINA?
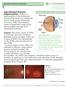 Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
New STAIRWAY study data shows potential for extended durability with Roche s faricimab in neovascular age-related macular degeneration (namd)
 Media Release New STAIRWAY study data shows potential for extended durability with Roche s faricimab in neovascular age-related macular degeneration (namd) Faricimab the first bispecific antibody designed
Media Release New STAIRWAY study data shows potential for extended durability with Roche s faricimab in neovascular age-related macular degeneration (namd) Faricimab the first bispecific antibody designed
Photodynamic Therapy for Choroidal Neovascularization
 Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
These issues are covered in more detail below.
 26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
Original Article Ranibizumab as needed therapy for wet age-related macular degeneration combined with serous pigment epithelial detachment
 Int J Clin Exp Med 2016;9(7):13650-13656 www.ijcem.com /ISSN:1940-5901/IJCEM0026582 Original Article Ranibizumab as needed therapy for wet age-related macular degeneration combined with serous pigment
Int J Clin Exp Med 2016;9(7):13650-13656 www.ijcem.com /ISSN:1940-5901/IJCEM0026582 Original Article Ranibizumab as needed therapy for wet age-related macular degeneration combined with serous pigment
Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration
 REVIEW ARTICLE Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration Karen M. Wai, Rishi P. Singh Department of Ophthalmology,
REVIEW ARTICLE Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration Karen M. Wai, Rishi P. Singh Department of Ophthalmology,
Digital Journal of Ophthalmology Digital Journal of Ophthalmology. Abstract
 Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
1. Introduction. Correspondence should be addressed to Abdullah Ozkaya; Received 4 June 2013; Accepted 3 October 2013
 Ophthalmology Volume 2013, Article ID 786107, 5 pages http://dx.doi.org/10.1155/2013/786107 Clinical Study Is Spectral-Domain Optical Coherence Tomography Essential for Flexible Treatment Regimens with
Ophthalmology Volume 2013, Article ID 786107, 5 pages http://dx.doi.org/10.1155/2013/786107 Clinical Study Is Spectral-Domain Optical Coherence Tomography Essential for Flexible Treatment Regimens with
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: December 7, 2011 Subject: Page: 1 of 23
 Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
VEGF Trap-Eye: New Data Confirm Successes in the Treatment of Age-related Macular Degeneration
 Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Final Phase 2 Results Presented at Retina Society Meeting VEGF Trap-Eye: New Data Confirm Successes in the Treatment
Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Final Phase 2 Results Presented at Retina Society Meeting VEGF Trap-Eye: New Data Confirm Successes in the Treatment
Clinical Study Incidence of Retinopathy of Prematurity in Extremely Premature Infants
 ISRN Pediatrics, Article ID 134347, 4 pages http://dx.doi.org/10.1155/2014/134347 Clinical Study Incidence of Retinopathy of Prematurity in Extremely Premature Infants Alparslan Fahin, Muhammed Fahin,
ISRN Pediatrics, Article ID 134347, 4 pages http://dx.doi.org/10.1155/2014/134347 Clinical Study Incidence of Retinopathy of Prematurity in Extremely Premature Infants Alparslan Fahin, Muhammed Fahin,
Anti VEGF Agents in Retinal Disorders Current Scenario
 Retina Anti VEGF Agents in Retinal Disorders Current Scenario Charu Gupta MS Charu Gupta MS, Cyrus M. Shroff MD Shroff Eye Centre, New Delhi T is a group of proteins involved in the regulation of angiogenesis,
Retina Anti VEGF Agents in Retinal Disorders Current Scenario Charu Gupta MS Charu Gupta MS, Cyrus M. Shroff MD Shroff Eye Centre, New Delhi T is a group of proteins involved in the regulation of angiogenesis,
International Journal of Health Sciences and Research ISSN:
 International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
Research Article The Impact of the Menstrual Cycle on Perioperative Bleeding in Vitreoretinal Surgery
 Hindawi Ophthalmology Volume 2017, Article ID 9549284, 4 pages https://doi.org/10.1155/2017/9549284 Research Article The Impact of the Menstrual Cycle on Perioperative Bleeding in Vitreoretinal Surgery
Hindawi Ophthalmology Volume 2017, Article ID 9549284, 4 pages https://doi.org/10.1155/2017/9549284 Research Article The Impact of the Menstrual Cycle on Perioperative Bleeding in Vitreoretinal Surgery
Subgroup Analysis of the MARINA Study of Ranibizumab in Neovascular Age-Related Macular Degeneration
 Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Diabetic Macular Edema Treatment in the 21st Century
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,700 108,500 1.7 M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,700 108,500 1.7 M Open access books available International authors and editors Downloads Our
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
NEOVASCULAR AGE-RELATED MACULAR DEGENERation
 Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Wet AMD. dept headline headline. headline headline. From Start to Finish
 Wet AMD dept headline headline From Start to Finish headline headline Deck Anti-VEGF therapy for patients with wet AMD is a standard of care that saves sight for thousands of people each year. dept byline
Wet AMD dept headline headline From Start to Finish headline headline Deck Anti-VEGF therapy for patients with wet AMD is a standard of care that saves sight for thousands of people each year. dept byline
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England. Retinal Vein Occlusion (RVO) with Macular oedema (MO)
 Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Drug Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Research Article Differentiation between Good and Low-Responders to Intravitreal Ranibizumab for Macular Edema Secondary to Retinal Vein Occlusion
 Hindawi Publishing Corporation Journal of Ophthalmology Volume 2016, Article ID 9875741, 6 pages http://dx.doi.org/10.1155/2016/9875741 Research Article Differentiation between Good and Low-Responders
Hindawi Publishing Corporation Journal of Ophthalmology Volume 2016, Article ID 9875741, 6 pages http://dx.doi.org/10.1155/2016/9875741 Research Article Differentiation between Good and Low-Responders
Stabilization of visual acuity with photodynamic therapy in eyes with chorioretinal anastomoses
 Graefe s Arch Clin Exp Ophthalmol (2004) 242:368 376 CLINICAL INVESTIGATION DOI 10.1007/s00417-003-0844-0 Rufino M. Silva José R. Faria de Abreu António Travassos José G. Cunha-Vaz Stabilization of visual
Graefe s Arch Clin Exp Ophthalmol (2004) 242:368 376 CLINICAL INVESTIGATION DOI 10.1007/s00417-003-0844-0 Rufino M. Silva José R. Faria de Abreu António Travassos José G. Cunha-Vaz Stabilization of visual
Treatment options for the wet form of age-related macular degeneration a perspective
 Perspective Treatment options for the wet form of age-related macular degeneration a perspective Marion R. Munk 1,2, Rene Rückert 3 1 Department of Ophthalmology, Inselspital, University Hospital Bern,
Perspective Treatment options for the wet form of age-related macular degeneration a perspective Marion R. Munk 1,2, Rene Rückert 3 1 Department of Ophthalmology, Inselspital, University Hospital Bern,
Retina Conference. Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014
 Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
A prospective nonrandomized clinical study on exudative age related macular degeneration
 Original Article A prospective nonrandomized clinical study on exudative age related macular degeneration Ayakutty Muni Raja, Siddharam Janti, Charanya Chendilnathan, Adnan Matheen Department of Ophthalmology,
Original Article A prospective nonrandomized clinical study on exudative age related macular degeneration Ayakutty Muni Raja, Siddharam Janti, Charanya Chendilnathan, Adnan Matheen Department of Ophthalmology,
Secondary Analysis of Clinical Trials Data A Biostatistician s Experience
 Secondary Analysis of Clinical Trials Data A Biostatistician s Experience Gui-shuang Ying, PhD Center for Preventive Ophthalmology and Biostatistics Perelman School of Medicine University of Pennsylvania
Secondary Analysis of Clinical Trials Data A Biostatistician s Experience Gui-shuang Ying, PhD Center for Preventive Ophthalmology and Biostatistics Perelman School of Medicine University of Pennsylvania
Opinion 4 December 2013
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 4 December 2013 LUCENTIS 10 mg/ml, solution for injection Vial of 0.23 ml (CIP: 34009 378 101 5 9) Applicant: Novartis
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 4 December 2013 LUCENTIS 10 mg/ml, solution for injection Vial of 0.23 ml (CIP: 34009 378 101 5 9) Applicant: Novartis
Short-term outcomes of switching anti-vegf agents in eyes with treatment-resistant wet AMD
 Batioglu et al. BMC Ophthalmology (2015) 15:40 DOI 10.1186/s12886-015-0025-z RESEARCH ARTICLE Open Access Short-term outcomes of switching anti-vegf agents in eyes with treatment-resistant wet AMD Figen
Batioglu et al. BMC Ophthalmology (2015) 15:40 DOI 10.1186/s12886-015-0025-z RESEARCH ARTICLE Open Access Short-term outcomes of switching anti-vegf agents in eyes with treatment-resistant wet AMD Figen
Growth Conference. October 19, May 2013
 Dawson Corporate James Securities Presentation 3rd Annual Small Cap Growth Conference October 19, 2017 May 2013 Safe Harbor This document contains forward-looking statements within the meaning of the "safe
Dawson Corporate James Securities Presentation 3rd Annual Small Cap Growth Conference October 19, 2017 May 2013 Safe Harbor This document contains forward-looking statements within the meaning of the "safe
Prevention and treatment of agerelated macular degeneration
 THERAPY REVIEW Prevention and treatment of agerelated macular degeneration SARAH HORTON AND CATHERINE GULY Age-related macular degeneration (AMD) is a common cause of visual loss in older people and GPs
THERAPY REVIEW Prevention and treatment of agerelated macular degeneration SARAH HORTON AND CATHERINE GULY Age-related macular degeneration (AMD) is a common cause of visual loss in older people and GPs
Ranibizumab and pegaptanib for the treatment of agerelated macular degeneration
 Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
Addressing Sub-optimal Response During the Treatment of Wet AMD
 JULY/AUGUST 2014 Highlights from a roundtable discussion Part 2 of 2 Addressing Sub-optimal Response During the Treatment of Wet AMD Roundtable Faculty Peter K. Kaiser, MD (Moderator) David M. Brown, MD
JULY/AUGUST 2014 Highlights from a roundtable discussion Part 2 of 2 Addressing Sub-optimal Response During the Treatment of Wet AMD Roundtable Faculty Peter K. Kaiser, MD (Moderator) David M. Brown, MD
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D.
 Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
Research Article Predictive Factors for Medical Consultation for Sore Throat in Adults with Recurrent Pharyngotonsillitis
 International Otolaryngology Volume 2016, Article ID 6095689, 5 pages http://dx.doi.org/10.1155/2016/6095689 Research Article Predictive Factors for Medical Consultation for Sore Throat in Adults with
International Otolaryngology Volume 2016, Article ID 6095689, 5 pages http://dx.doi.org/10.1155/2016/6095689 Research Article Predictive Factors for Medical Consultation for Sore Throat in Adults with
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
ANTIVASCULAR ENDOTHELIAL GROWTH FACTOR
 THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
Earlier therapeutic effects associated with high dose (2.0 mg) Ranibizumab for treatment of vascularized pigment epithelial detachments in agerelated
 (2015) 29, 80 87 & 2015 Macmillan Publishers Limited All rights reserved 0950-222X/15 www.nature.com/eye CLINICAL STUDY 1 Southern California Desert Retina Consultants, Palm Desert, CA, USA 2 Loma Linda
(2015) 29, 80 87 & 2015 Macmillan Publishers Limited All rights reserved 0950-222X/15 www.nature.com/eye CLINICAL STUDY 1 Southern California Desert Retina Consultants, Palm Desert, CA, USA 2 Loma Linda
The Use of Intravitreal Aflibercept in the Treatment of Wet Type of Age Related Macular Degeneration
 BMH Medical Journal 2015;2(2):31-36 Review Article The Use of Intravitreal Aflibercept in the Treatment of Wet Type of Age Related Macular Degeneration Rejith Rag, MBBS, FRCS, DO, FICO Department of Ophthalmology,
BMH Medical Journal 2015;2(2):31-36 Review Article The Use of Intravitreal Aflibercept in the Treatment of Wet Type of Age Related Macular Degeneration Rejith Rag, MBBS, FRCS, DO, FICO Department of Ophthalmology,
FROM OUTDATED TO UPDATED Eminence-Based Medicine
 FROM OUTDATED TO UPDATED Eminence-Based Medicine Evidence-Based Medicine A REVIEW OF KEY CLINICAL TRIALS Anthony DeWilde, OD FAAO 1 EMINENCE BASED MEDICINE 2 EVIDENCE BASED MEDICINE 3 4 CLINICAL TRIALS
FROM OUTDATED TO UPDATED Eminence-Based Medicine Evidence-Based Medicine A REVIEW OF KEY CLINICAL TRIALS Anthony DeWilde, OD FAAO 1 EMINENCE BASED MEDICINE 2 EVIDENCE BASED MEDICINE 3 4 CLINICAL TRIALS
Photodynamic Therapy for Treatment of Subfoveal Choroidal Neovascularization in Exudative Age-Related Macular Degeneration
 Clinical Medicine Journal Vol. 4, No. 4, 2018, pp. 60-66 http://www.aiscience.org/journal/cmj ISSN: 2381-7631 (Print); ISSN: 2381-764X (Online) Photodynamic Therapy for Treatment of Subfoveal Choroidal
Clinical Medicine Journal Vol. 4, No. 4, 2018, pp. 60-66 http://www.aiscience.org/journal/cmj ISSN: 2381-7631 (Print); ISSN: 2381-764X (Online) Photodynamic Therapy for Treatment of Subfoveal Choroidal
Technology appraisal guidance Published: 27 August 2008 nice.org.uk/guidance/ta155
 Ranibizumab and pegaptanib for the treatment of age-related macular degeneration Technology appraisal guidance Published: 27 August 2008 nice.org.uk/guidance/ta155 NICE 2018. All rights reserved. Subject
Ranibizumab and pegaptanib for the treatment of age-related macular degeneration Technology appraisal guidance Published: 27 August 2008 nice.org.uk/guidance/ta155 NICE 2018. All rights reserved. Subject
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration
 Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Vascular Disease Ocular Manifestations of Systemic Hypertension
 Vascular Disease Ocular Manifestations of Systemic Hypertension Maynard L. Pohl, OD, FAAO Pacific Cataract & Laser Institute 10500 NE 8 th Street, Suite 1650 Bellevue, WA 98004 USA 425-462-7664 Cerebrovascular
Vascular Disease Ocular Manifestations of Systemic Hypertension Maynard L. Pohl, OD, FAAO Pacific Cataract & Laser Institute 10500 NE 8 th Street, Suite 1650 Bellevue, WA 98004 USA 425-462-7664 Cerebrovascular
Case Report Increase in Central Retinal Edema after Subthreshold Diode Micropulse Laser Treatment of Chronic Central Serous Chorioretinopathy
 Case Reports in Ophthalmological Medicine Volume 2015, Article ID 813414, 4 pages http://dx.doi.org/10.1155/2015/813414 Case Report Increase in Central Retinal Edema after Subthreshold Diode Micropulse
Case Reports in Ophthalmological Medicine Volume 2015, Article ID 813414, 4 pages http://dx.doi.org/10.1155/2015/813414 Case Report Increase in Central Retinal Edema after Subthreshold Diode Micropulse
Despite our growing knowledge of the
 OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy
 Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy Fawwaz Al Mamoori* Medical Retina Department, Eye Specialty Hospital,
Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy Fawwaz Al Mamoori* Medical Retina Department, Eye Specialty Hospital,
Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2015; 159:XX.
 The efficacy of ranibizumab treatment in clinical practice in patients with the wet form of age-related macular degeneration. The results of the Czech National Registry Oldrich Chrapek a, Jiri Jarkovsky
The efficacy of ranibizumab treatment in clinical practice in patients with the wet form of age-related macular degeneration. The results of the Czech National Registry Oldrich Chrapek a, Jiri Jarkovsky
Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema
 Original Article Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema Karen Joyce G. Castro, MD, Marie Joan V. Loy, MD International Eye Institute St. Luke s Medical Center
Original Article Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema Karen Joyce G. Castro, MD, Marie Joan V. Loy, MD International Eye Institute St. Luke s Medical Center
Section 21, Ophthalmological Preparations. Ranibizumab (Lucentis ) Addition
 Application for inclusion in the WHO Essential Medicines List Section 21, Ophthalmological Preparations Ranibizumab (Lucentis ) Addition Document type: Document status: Application for inclusion in WHO
Application for inclusion in the WHO Essential Medicines List Section 21, Ophthalmological Preparations Ranibizumab (Lucentis ) Addition Document type: Document status: Application for inclusion in WHO
R. J. L. F. Loffeld, 1 P. E. P. Dekkers, 2 and M. Flens Introduction
 ISRN Gastroenterology Volume 213, Article ID 87138, 5 pages http://dx.doi.org/1.1155/213/87138 Research Article The Incidence of Colorectal Cancer Is Decreasing in the Older Age Cohorts in the Zaanstreek
ISRN Gastroenterology Volume 213, Article ID 87138, 5 pages http://dx.doi.org/1.1155/213/87138 Research Article The Incidence of Colorectal Cancer Is Decreasing in the Older Age Cohorts in the Zaanstreek
Long-term Management of AMD. Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan
 Long-term Management of AMD Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan DEFINITION 1 Age-related macular degeneration (AMD) is a disorder of the macula characterized
Long-term Management of AMD Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan DEFINITION 1 Age-related macular degeneration (AMD) is a disorder of the macula characterized
Abicipar pegol for wet age related macular degeneration
 EVIDENCE BRIEFING AUGUST 2018 Abicipar pegol for wet age related macular degeneration NIHRIO ID 7084 NICE ID 9724 Developer/Company Allergan Ltd and Molecular Partners UKPS ID 646978 Licensing and market
EVIDENCE BRIEFING AUGUST 2018 Abicipar pegol for wet age related macular degeneration NIHRIO ID 7084 NICE ID 9724 Developer/Company Allergan Ltd and Molecular Partners UKPS ID 646978 Licensing and market
Outcomes of Persistently Active Neovascular Age-Related Macular Degeneration. Treated with VEGF Inhibitors: Observational Study data.
 Outcomes of Persistently Active Neovascular Age-Related Macular Degeneration Treated with VEGF Inhibitors: Observational Study data. Daniel Barthelmes 1,9, Richard Walton 1, Anna E Campain 1, Judy M Simpson
Outcomes of Persistently Active Neovascular Age-Related Macular Degeneration Treated with VEGF Inhibitors: Observational Study data. Daniel Barthelmes 1,9, Richard Walton 1, Anna E Campain 1, Judy M Simpson
Treatment practice in the
 Strategies for managing neovascular AMD and in routine clinical care BY ROD MCNEIL Treatment practice in the management of neovascular age-related macular degeneration (AMD) and diabetic macular oedema
Strategies for managing neovascular AMD and in routine clinical care BY ROD MCNEIL Treatment practice in the management of neovascular age-related macular degeneration (AMD) and diabetic macular oedema
Supplement to March Ranibizumab for Visual Impairment in DME: An Overview of The Evidence SPONSORED BY NOVARTIS PHARMA AG
 Supplement to March 2018 Ranibizumab for Visual Impairment in DME: An Overview of The Evidence SPONSORED BY NOVARTIS PHARMA AG Ranibizumab for Visual Impairment in DME: An Overview of The Evidence BY PROF.
Supplement to March 2018 Ranibizumab for Visual Impairment in DME: An Overview of The Evidence SPONSORED BY NOVARTIS PHARMA AG Ranibizumab for Visual Impairment in DME: An Overview of The Evidence BY PROF.
Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre
 Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre 11:00-11:55 WS #115: The Revolution in Macular Degeneration Management 12:05-13:00 WS #127: The Revolution in Macular
Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre 11:00-11:55 WS #115: The Revolution in Macular Degeneration Management 12:05-13:00 WS #127: The Revolution in Macular
Correspondence should be addressed to Alicia McMaster;
 Cancer Research Volume 2013, Article ID 308236, 5 pages http://dx.doi.org/10.1155/2013/308236 Research Article Taxpas: Epidemiological and Survival Data in Breast Cancer Patients Treated with a Docetaxel-Based
Cancer Research Volume 2013, Article ID 308236, 5 pages http://dx.doi.org/10.1155/2013/308236 Research Article Taxpas: Epidemiological and Survival Data in Breast Cancer Patients Treated with a Docetaxel-Based
Angiogenesis and Role of Anti-VEGF Therapy
 Review Article Angiogenesis and Role of Anti-VEGF Therapy P.S. Mahar, Azfar N. Hanfi, Aimal Khan Pak J Ophthalmol 2009, Vol. 25 No. 3.....................................................................................................
Review Article Angiogenesis and Role of Anti-VEGF Therapy P.S. Mahar, Azfar N. Hanfi, Aimal Khan Pak J Ophthalmol 2009, Vol. 25 No. 3.....................................................................................................
Case Report Diagnosis and Monitoring of Choroidal Osteoma through Multimodal Imaging
 Case Reports in Medicine, Article ID 393804, 4 pages http://dx.doi.org/10.1155/2014/393804 Case Report Diagnosis and Monitoring of Choroidal Osteoma through Multimodal Imaging Theodoros Empeslidis, Usman
Case Reports in Medicine, Article ID 393804, 4 pages http://dx.doi.org/10.1155/2014/393804 Case Report Diagnosis and Monitoring of Choroidal Osteoma through Multimodal Imaging Theodoros Empeslidis, Usman
Good News for The Macular Generation. Dr Andrew Thompson FRANZCO
 Good News for The Macular Generation Dr Andrew Thompson FRANZCO Anti-VEGFs for wet MD are one of the greatest advances in medicine in past decade Prof. Gary Brown Director Wills Eye Institue Retina Service
Good News for The Macular Generation Dr Andrew Thompson FRANZCO Anti-VEGFs for wet MD are one of the greatest advances in medicine in past decade Prof. Gary Brown Director Wills Eye Institue Retina Service
Clinical Study Metastasectomy of Pulmonary Metastases from Osteosarcoma: Prognostic Factors and Indication for Repeat Metastasectomy
 Respiratory Medicine Volume 2015, Article ID 570314, 5 pages http://dx.doi.org/10.1155/2015/570314 Clinical Study Metastasectomy of Pulmonary Metastases from Osteosarcoma: Prognostic Factors and Indication
Respiratory Medicine Volume 2015, Article ID 570314, 5 pages http://dx.doi.org/10.1155/2015/570314 Clinical Study Metastasectomy of Pulmonary Metastases from Osteosarcoma: Prognostic Factors and Indication
Research Article Predictions of the Length of Lumbar Puncture Needles
 Computational and Mathematical Methods in Medicine, Article ID 732694, 5 pages http://dx.doi.org/10.1155/2014/732694 Research Article Predictions of the Length of Lumbar Puncture Needles Hon-Ping Ma, 1,2
Computational and Mathematical Methods in Medicine, Article ID 732694, 5 pages http://dx.doi.org/10.1155/2014/732694 Research Article Predictions of the Length of Lumbar Puncture Needles Hon-Ping Ma, 1,2
