Sugammadex: A New Wheel Has Been Invented. Ashlee A. Wheeler, BSN, RN, SRNA
|
|
|
- Osborne Miles
- 5 years ago
- Views:
Transcription
1 Sugammadex: A New Wheel Has Been Invented Ashlee A. Wheeler, BSN, RN, SRNA
2 Disclaimer Statement Conflict of Interest: None I have not received any compensation for this presentation.
3 Objectives The participant will be able to Describe the mechanism of action for sugammadex. List the indications and contraindications for the use of sugammadex. Explain when to utilize doses of 2 mg/kg, 4 mg/kg, and 16 mg/kg of sugammadex.
4 What is Sugammadex? It is the first drug in a new class of medications called selective relaxant binding agents Brand name is Bridion Received U.S. Food and Drug (FDA) approval on December 15 th, Is indicated for the reversal of neuromuscular blockade caused by the aminosteroid non-depolarizing muscle relaxants rocuronium bromide or vecuronium bromide
5 What's in a name Su-in Sugammadex stands for sugar -gammadex stands for the structural molecule gammacyclodextrin There are 3 naturally occurring cyclodextrins Alpha Beta Gamma Sugammadex is a modified γ-cyclodextrin
6 Encapsulating The exterior of the structure is hydrophilic and the interior is lipophilic It is able to bind with rocuronium or vecuronium in a tight 1:1 ratio Sugammadex has a high association rate and very low dissociation rate Estimated that for every 25 million rocuronium-sugammadex complexes that form, only one dissociates Intermolecular (van der Waals ) forces Thermodynamic (hydrogen) bonds Hydrophobic interactions
7 Sugammadex-How does it work? When administered it binds with the free floating drug in the blood plasma This causes a concentration gradient to form, moving rocuronium from the neuromuscular junction back into the blood plasma where it can continue to be encapsulated by sugammadex
8 Metabolism and Elimination Neither sugammadex or the sugammadex-rocuronium complex undergoes any metabolism and does not form any metabolites within the body Both sugammadex and the sugammadex-rocuronium complex are cleared by the kidneys nearly 100% Normal glomerular filtration rate: 70% excreted in 6 hours over 90% within 24 hours
9 Indications Intravenous administration Reversal of neuromuscular blockade induced by: Rocuronium bromide Vecuronium bromide Currently only approved for use in adults In Europe it is also been approved for the reversal of rocuronium in children and adolescents
10 Contraindications Absolute contraindication is: known hypersensitivity to sugammadex or any of its components.
11 Considerations: Renal Impairment Patients with renal impairment: currently only recommended in patients who have a creatinine clearance greater than 30 ml/min In patients with a creatinine clearance less than 30 ml/min it can take 48 hours or more for the rocuronium-sugammadex molecule to be eliminated It is not known how long the complex will stay bound within the body In a study by Cammu et al. (2012) it was found that the rocuroniumsugammadex complex can be filtered by high-flux dialysis filters but not low-flux High-flux filters allow molecules that are ,000 Da to pass Sugammadex exceeds 2,000 Da
12 Considerations: Chemotherapy Patients who are currently on prescribed chemotherapy drug Toremifene There could be a delay in reversal if Toremifene has been given the same day as the surgery.
13 Considerations: Oral Contraceptives Women taking oral birth control with estrogen or progestogen complexes: Sugammadex will bind to the progestogen and block it, therefore it is the equivalent of missing a dose If given sugammadex, an alternative means of birth control should be used for seven days post administration
14 Considerations: Pharmacology Compatible delivery fluids: Normal Saline 5% Dextrose Ringer s Lactate 5% Dextrose in Normal Saline 2.5% Dextrose in 0.45% Normal Saline Physically incompatible: Verapamil Ondansetron (Zofran) Ranitidine (Zantac)
15 Safety Concerns Application submitted to the FDA and the European Union authorities in 2008 European Union authorities-approved FDA-denied due to concerns about hypersensitivity reactions in healthy volunteers who received high does The incidence of hypersensitivity in all studies was less than 1% Since its European approval in 2008 there have been over 9 million patient exposures-15 probable cases of sugammadex anaphylaxis worldwide
16 Study by Godai et al, (2012) show 3 suspected cases of sugammadex-induced hypersensitivity reactions over a 1 year period out of 1864 patient exposures % Sugammadex appears to be a safe and well-tolerated agent No deaths have been related to the administration of sugammadex
17 Side Effects Most Frequent Adverse Effects: Hypotension Coughing Nausea Vomiting Sensation of a change in temperature Abnormal level of N- Acetylglucosaminidase in the urine Reactions are typically seen within 5 minutes of administration Appear to be more frequent at higher clinical doses Dry Mouth
18 Dosing Regimen Dose range: 2 mg/kg if 2/4 twitches are present in response to train-of-four; 4 mg/kg if no TOF twitches are present but 1-2 post-tetanic counts are seen 16 mg/kg within 3 minutes of 1.2 mg/kg rocuronium, in a cannot intubate cannot ventilate situation Delivered as a bolus within 10 seconds. Onset: Less than 3 minutes
19 Dosing: Re-administration Options for neuromuscular block after sugammadex 2-4 mg/kg administration: Succinylcholine Benzylisoquinolinium agents Cisatracurium or Atracurium Rocuronium* or Vecuronium*
20 Dosing: Re-administration Rocuronium* or Vecuronium* Up to 4mg/kg of sugammadex 5 minutes 1.2 mg/kg rocuronium ** 4 hours 0.6 mg/kg rocuronium 0.1 mg/kg vecuronium 16 mg/kg of sugammadex 24 hours Rocuronium or Vecuronium ** Onset may be delayed up to 4 minutes ** Duration shortened up to 15 minutes
21 Dose
22 Train of Four
23 Reversal of 1.2 mg/kg Rocuronium
24 Compared to Neostigmine This mechanism of action, as you can see, is completely different from neostigmine No parasympathetic effects. No anticholinergic drugs required. Speed of reversal has been found to be 3-8 times faster than neostigmine
25 Figure 1: 2: Time (Minutes) from Administration of BRIDION of BRIDION or Neostigmine or Neostigmine at the at Reappearance the Reappearance of T 2 after of T2 Vecuronium after Rocuronium to Recovery to of the the T4/T1 T 4 /T 1 Ration to to
26 Cost Prices from York Hospital Reversal for 100kg patient Drug Dose Price per vial $ to reverse a 100kg patient Neostigmine 0.05 mg/kg (max 5mg) $13.21/10 ml vial $39.39 Glycopyrrolate 200 mcg per 1 mg of neostigmine $13.09/2 ml vial (2 vials, 800mcg) $26.18 Sugammadex 2 mg/kg $88.28/2 ml vial $ mg/kg $161.69/5 ml vial $ mg/kg 3 x 5 ml vial = $ x 2 ml vial = $88.28 $573.35
27 Cost Prices from New Hanover Medical Group-Wilmington NC Drug Dose Price per vial $ to reverse a 100kg patient Neostigmine Glycopyrrolate 0.05 mg/kg (max 5mg) 200 mcg per 1 mg of neostigmine $58.51/10 ml vial $89.65 $31.14/5 ml vial (1mg) Sugammadex 2 mg/kg $88.59/2 ml vial $88.59 J. Wheeler, personal communication, September 1, 2016
28 Can Sugammadex be costeffective? In a study by Carron et al. (2016) they looked at the clinical outcomes and the cost-effectiveness of sugammadex Cost savings were shown by decreasing the recovery time from neuromuscular blockage in the OR Additional savings: Reducing rate of postoperative residual curarization Time spent in the recovery room Decreased the rate of unexpected ICU admissions
29 Patient Populations Children: Not currently FDA approved for the use in patients 17 years of age Elderly: >65 years old. No difference in dose recommendations as long as kidney function is normal, recovery time may be prolonged from < 2 minutes to < 4 minutes Obesity: Dose based on actual body weight Pregnancy and breast feeding: Animal studies showed no signs of direct or indirect negative side effects Unknown if it is excreted in breast milk; animal studies do show a degree of excretion Oral absorption of sugammadex is low and no effects on infants are expected
30 Myasthenia Gravis Autoimmune disease resulting in a decrease in the number of postsynaptic acetylcholine receptors Patients are treated with anticholinesterase medications such as pyridostigmine Typically neuromuscular blocking agents are avoided in these patients These patients are resistant to succinylcholine requiring higher doses to achieve paralysis They can show sensitivity to nondepolarizing neuromuscular blockers requiring ½ of the ED 95 dose to achieve paralysis Reversing nondepolarizing neuromuscular blockers is avoided to prevent cholinergic crisis that can occur since patients are currently on these medications for their illness
31 Myasthenia Gravis-Continued Sugammadex may change the anesthetic treatment for these patients. Study by Ulke et al. (2013) enrolled 10 patients with myasthenia gravis who were scheduled for a video-assisted thoracoscopic extended thymectomy (VATET) All patients received 0.3 mg/kg of rocuronium for intubation and supplemental doses if required; total dose of rocuronium was 48 mg (±16 mg) At the end of the case all patients received 2 mg/kg of sugammadex All patient s achieved a train of four ratio of >0.9. All were extubated in the operating room and none required mechanical ventilation due to respiratory failure or myasthenic crisis
32 Case Study- Rocuronium Anaphylaxis 33-year-old, 77 kg, female laparoscopic procedure for the investigation of infertility 30 seconds after receiving 30 mg of rocuronium Tachycardia 122 beats per minute Cough Difficult mask ventilation After intubation-visual confirmation of tube placement No carbon dioxide detected on capnography Airway pressures were elevated Sp O2 decreased to 80% No carotid pulse detected CPR started IV fluids administered Increasing doses of epinephrine, initial 200 mcg, 800 mcg, 1 mg
33 Case Study-Continued 19 minutes into resuscitation 4 mg of epinephrine 2000 ml of Lactated Ringer s 1500 ml of colloid 500 mg of sugammadex (6.5 mg/kg) administered during chest compressions 45 seconds after administration during chest compressions, patient opened her eyes and reached for the endotracheal tube Blood pressure 111/56 Heart rate 126 beats minute Sp O2 97% Patient was transferred to an off site intensive care unit Within 30 minutes of arrival to the ICU patient was extubated and no vasopressor support was required Patient was discharged 48 hours after the event with no additional complications and no recollection of the event
34 Questions??
35 References Abad Gurumeta, A., Ripollés Melchor, J., Casans Francés, R., Espinosa, A., Martínez Hurtado, E., Fernández Pérez, C.,... & Calvo Vecino, J. M. (2015). A systematic review of sugammadex vs neostigmine for reversal of neuromuscular blockade. Anaesthesia, 70(12), Barash, P. G., Cullen, B. F., Stoelting, R. K., Cahalan, M. K., Stock, M. C., & Ortega, R. (2013). Clinical anesthesia (7 th ed.). Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins. Cammu, G., Van Vlem, B., van den Heuvel, M., Stet, L., el Galta, R., Eloot, S., & Demeyer, I. (2012). Dialysability of sugammadex and its complex with rocuronium in intensive care patients with severe renal impairment. British journal of anaesthesia, 109(3), Carron, M., Baratto, F., Zarantonello, F., & Ori, C. (2016). Sugammadex for reversal of neuromuscular blockade: a retrospective analysis of clinical outcomes and cost-effectiveness in a single center. ClinicoEconomics and outcomes research: CEOR, 8, 43. Chambers, L. (2016). Pharmacy and therapeutics committee formulary review sugammadex (Bridion). [Handout]. Wellspan. Godai, K., Hasegawa-Moriyama, M., Kuniyoshi, T., Kakoi, T., Ikoma, K., Isowaki, S.,... & Kanmura, Y. (2012). Three cases of suspected sugammadex-induced hypersensitivity reactions. British journal of anaesthesia, 109(2), Ledowski, T. (2015). Sugammadex: What do we know and what do we still need to know? A review of the recent (2013 to 2014) literature. Anaesthesia & Intensive Care, 43(1). McDonnell, N. J., Pavy, T. J. G., Green, L. K., & Platt, P. R. (2011). Sugammadex in the management of rocuronium- induced anaphylaxis. British journal of anaesthesia, 106(2), Merck Sharp & Dohme Corp. (2015). Sugammadex [package insert]. Retrieved April 14, 2016 from Nag, K., Singh, D. R., Shetti, A. N., Kumar, H., Sivashanmugam, T., & Parthasarathy, S. (2013). Sugammadex: A revolutionary drug in neuromuscular pharmacology. Anesthesia, Essays and Researches, 7(3), 302.
36 References Naguib, M. (2007). Sugammadex: Another milestone in clinical neuromuscular pharmacology. Anesthesia & Analgesia, 104(3), Peeters, P. A., van den Heuvel, M. W., van Heumen, E., Passier, P. C., Smeets, J. M., van Iersel, T., & Zwiers, A. (2010). Safety, tolerability and pharmacokinetics of sugammadex using single high doses (up to 96 mg/kg) in healthy adult subjects. Clinical drug investigation, 30(12), Schaller, S. J., & Fink, H. (2013). Sugammadex as a reversal agent for neuromuscular block: an evidence-based review. Core Evid, 8(8), Sungur Ulke, Z., Yavru, A., Camci, E., Ozkan, B., Toker, A., & Senturk, M. (2013). Rocuronium and sugammadex in patients with myasthenia gravis undergoing thymectomy. Acta Anaesthesiologica Scandinavica, 57(6), UpToDate. (2016). Sugammadex: Drug Information. In: UpToDate online. Retrieved from: source=search_result&search=sugammadex&selectedtitle=1%7e25 Welliver, M. (2006). New drug sugammadex: A selective relaxant binding agent. AANA journal, 74(5). Welliver, M., & Cheek, D. (2009). An update on sugammadex sodium. AANA journal, 77(3). Welliver, M., Cheek, D., Osterbrink, J., & McDonough, J. (2015). Worldwide experience with sugammadex sodium: Implications for the United States. AANA journal, 83(2), 107. Williamson, R. M., Mallaiah, S., & Barclay, P. (2011). Rocuronium and sugammadex for rapid sequence induction of obstetric general anaesthesia. Acta Anaesthesiologica Scandinavica, 55(6),
37
Running head: SUGAMMADEX AND MYASTHENIA GRAVIS 1
 Running head: SUGAMMADEX AND MYASTHENIA GRAVIS 1 Sugammadex in Patients with Myasthenia Gravis Jennifer A. Madsen University of Kansas SUGAMMADEX AND MYASTHENIA GRAVIS 2 Title of Proposed Research Project
Running head: SUGAMMADEX AND MYASTHENIA GRAVIS 1 Sugammadex in Patients with Myasthenia Gravis Jennifer A. Madsen University of Kansas SUGAMMADEX AND MYASTHENIA GRAVIS 2 Title of Proposed Research Project
An Overview of Sugammadex
 B A S I C S C I E N C E Tutorial 332 An Overview of Sugammadex Dr Ciara Mitchell Anaesthetic Registrar, Royal Victoria Hospital, Belfast, Northern Ireland Dr Steve Lobaz Consultant in Anaesthetics and
B A S I C S C I E N C E Tutorial 332 An Overview of Sugammadex Dr Ciara Mitchell Anaesthetic Registrar, Royal Victoria Hospital, Belfast, Northern Ireland Dr Steve Lobaz Consultant in Anaesthetics and
TRANSPARENCY COMMITTEE OPINION. 21 January 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 21 January 2009 BRIDION 100 mg/ml, solution for injection Box containing 10 x 2 ml bottles, CIP: 573 553-9 Box containing
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 21 January 2009 BRIDION 100 mg/ml, solution for injection Box containing 10 x 2 ml bottles, CIP: 573 553-9 Box containing
The Latest Approaches to Reversal of Neuromuscular Blocking Agents
 The Latest Approaches to Reversal of Neuromuscular Blocking Agents Janay Bailey, Pharm.D. Anesthesiology 2017; 126:173-90 Objectives Pharmacists Determine optimal paralytic choices in knowing if reversal
The Latest Approaches to Reversal of Neuromuscular Blocking Agents Janay Bailey, Pharm.D. Anesthesiology 2017; 126:173-90 Objectives Pharmacists Determine optimal paralytic choices in knowing if reversal
Bridion 100 mg/ml Solution for injection Sugammadex
 Bridion 100 mg/ml Solution for injection Sugammadex 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium
Bridion 100 mg/ml Solution for injection Sugammadex 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium
Sugammadex: A Comprehensive Review of the Published Human Science, Including Renal Studies
 Sugammadex: A Comprehensive Review of the Published Human Science, Including Renal Studies Kelsey Martin, MD. CA3 Resident, Indiana University School of Medicine Department of Anesthesia Article Abstract
Sugammadex: A Comprehensive Review of the Published Human Science, Including Renal Studies Kelsey Martin, MD. CA3 Resident, Indiana University School of Medicine Department of Anesthesia Article Abstract
Anesthesia and Neuromuscular Blockade: A Guide for Hospital Pharmacists. Upon completion of this activity, participants will be better able to:
 Anesthesia and Neuromuscular Blockade: A Guide for Hospital Pharmacists EDUCATIONAL OBJECTIVES Upon completion of this activity, participants will be better able to: 1. Understand the use of neuromuscular
Anesthesia and Neuromuscular Blockade: A Guide for Hospital Pharmacists EDUCATIONAL OBJECTIVES Upon completion of this activity, participants will be better able to: 1. Understand the use of neuromuscular
1. NAME OF THE MEDICINAL PRODUCT
 08-15 BRIDIO Solution for Injection ORGAO Brand of sugammadex FOR ITRAVEOUS IJECTIO 1. AME OF THE MEDICIAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AD QUATITATIVE COMPOSITIO 1 ml
08-15 BRIDIO Solution for Injection ORGAO Brand of sugammadex FOR ITRAVEOUS IJECTIO 1. AME OF THE MEDICIAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AD QUATITATIVE COMPOSITIO 1 ml
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium equivalent
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium equivalent
BRIDION sugammadex (as sodium salt)
 BRIDION sugammadex (as sodium salt) (i) NAME OF THE MEDICINE Bridion 100 mg/ml Solution for Injection Molecular Formula: C 72 H 104 O 48 S 8 Na 8 Molecular mass: 2178.01 Cas. Registry No: 343306-79-6 Chemical
BRIDION sugammadex (as sodium salt) (i) NAME OF THE MEDICINE Bridion 100 mg/ml Solution for Injection Molecular Formula: C 72 H 104 O 48 S 8 Na 8 Molecular mass: 2178.01 Cas. Registry No: 343306-79-6 Chemical
BRIDION sugammadex (as sodium salt)
 NEW ZEALAND DATA SHEET BRIDION sugammadex (as sodium salt) (i) NAME OF THE MEDICINE Bridion 100 mg/ml Solution for Injection Molecular Formula: C 72 H 104 O 48 S 8 Na 8 Molecular mass: 2178.01 Cas. Registry
NEW ZEALAND DATA SHEET BRIDION sugammadex (as sodium salt) (i) NAME OF THE MEDICINE Bridion 100 mg/ml Solution for Injection Molecular Formula: C 72 H 104 O 48 S 8 Na 8 Molecular mass: 2178.01 Cas. Registry
Core Safety Profile. Pharmaceutical form(s)/strength: Solution for Injection CZ/H/PSUR/0005/002 Date of FAR:
 Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise
 Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise 1. Read chapter 2. Review objectives (p.305) 3. Review key terms and definitions (p.305) Add: Cholinesterase inhibitor Vagal
Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise 1. Read chapter 2. Review objectives (p.305) 3. Review key terms and definitions (p.305) Add: Cholinesterase inhibitor Vagal
Effects of dexamethasone on sugammadex reversal times of rocuronium: a systematic review protocol
 Effects of dexamethasone on sugammadex reversal times of rocuronium: a systematic review protocol Cassie R. Held Mackenzie D. Sullivan The Center for Translational Research: a Joanna Briggs Institute Center
Effects of dexamethasone on sugammadex reversal times of rocuronium: a systematic review protocol Cassie R. Held Mackenzie D. Sullivan The Center for Translational Research: a Joanna Briggs Institute Center
Sugammadex in patients with chronic renal failure: two case reports
 ISSN: 2203-1413 Vol.04 No.02 Sugammadex in patients with chronic renal failure: two case reports Gwi Eun Yeo 1, Byung Gun Lim 1*, So Hyun Lee 1, Won Joon Lee 1, Il Ok Lee 1 1. Department of Anesthesiology
ISSN: 2203-1413 Vol.04 No.02 Sugammadex in patients with chronic renal failure: two case reports Gwi Eun Yeo 1, Byung Gun Lim 1*, So Hyun Lee 1, Won Joon Lee 1, Il Ok Lee 1 1. Department of Anesthesiology
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium equivalent
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains sugammadex sodium equivalent
PRODUCT MONOGRAPH BRIDION. solution for injection, 100 mg/ml sugammadex (as sugammadex sodium) Selective Relaxant Binding Agent
 PRDUCT MNGRAPH BRIDIN solution for injection, 100 mg/ml sugammadex (as sugammadex sodium) Selective Relaxant Binding Agent Merck Canada Inc. 16750 route Transcanadienne Kirkland, QC Canada H9H 4M7 www.merck.ca
PRDUCT MNGRAPH BRIDIN solution for injection, 100 mg/ml sugammadex (as sugammadex sodium) Selective Relaxant Binding Agent Merck Canada Inc. 16750 route Transcanadienne Kirkland, QC Canada H9H 4M7 www.merck.ca
Is Sugammadex the right choice for reversal? YES NO
 Is Sugammadex the right choice for reversal? YES NO Residual Neuromuscular Blockade (rnmb) rnmb after GA using nondepolarizing NMBA s has pathophysiological, clinical, and economic consequences. Significant
Is Sugammadex the right choice for reversal? YES NO Residual Neuromuscular Blockade (rnmb) rnmb after GA using nondepolarizing NMBA s has pathophysiological, clinical, and economic consequences. Significant
Package leaflet: Information for the user. Bridion 100 mg/ml solution for injection sugammadex
 Package leaflet: Information for the user Bridion 100 mg/ml solution for injection sugammadex Read all of this leaflet carefully before you are given this medicine because it contains important information
Package leaflet: Information for the user Bridion 100 mg/ml solution for injection sugammadex Read all of this leaflet carefully before you are given this medicine because it contains important information
Pharmacology of the Neuromuscular Junction (NMJ)
 Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
University of Mississippi Medical Center University of Mississippi Health Care. Pharmacy and Therapeutics Committee Medication Use Evaluation
 University of Mississippi Medical Center University of Mississippi Health Care Pharmacy and Therapeutics Committee Medication Use Evaluation Cisatracurium (Nimbex ) and Vecuronium (Norcuron ) PURPOSE The
University of Mississippi Medical Center University of Mississippi Health Care Pharmacy and Therapeutics Committee Medication Use Evaluation Cisatracurium (Nimbex ) and Vecuronium (Norcuron ) PURPOSE The
Disclosure Statement:
 Quite possibly the most interesting drug NOT approved by the FDA Yet. Sal J Morana RPh, PhD Chief Operating Officer (formerly the Pharmacy Director) Porter Medical Center Middlebury, VT Adjunct Faculty/Preceptor
Quite possibly the most interesting drug NOT approved by the FDA Yet. Sal J Morana RPh, PhD Chief Operating Officer (formerly the Pharmacy Director) Porter Medical Center Middlebury, VT Adjunct Faculty/Preceptor
New York Science Journal 2017;10(6) Efficacy And Safety Of Sugammadex In Reversing Nmb (Rocuronium) In Adults
 Efficacy And Safety Of Sugammadex In Reversing Nmb (Rocuronium) In Adults Mahmoud Abd Elrahman El Sherbeny; Ehab Ahmed Abd Elrahman; Reda Khalil Kamal; Mohamed Ahmed Mohamed Abozena Anesthesia and ICU
Efficacy And Safety Of Sugammadex In Reversing Nmb (Rocuronium) In Adults Mahmoud Abd Elrahman El Sherbeny; Ehab Ahmed Abd Elrahman; Reda Khalil Kamal; Mohamed Ahmed Mohamed Abozena Anesthesia and ICU
Pharmacology of the Neuromuscular Junction (NMJ)
 Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Chapter 18. Skeletal Muscle Relaxants (Neuromuscular Blocking Agents)
 Chapter 18 Skeletal Muscle Relaxants (Neuromuscular Blocking Agents) Uses of Neuromuscular Blocking Facilitate intubation Surgery Agents Enhance ventilator synchrony Reduce intracranial pressure (ICP)
Chapter 18 Skeletal Muscle Relaxants (Neuromuscular Blocking Agents) Uses of Neuromuscular Blocking Facilitate intubation Surgery Agents Enhance ventilator synchrony Reduce intracranial pressure (ICP)
A Nondepolarizing Neuromuscular Blocking (NMB) Agent
 DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
The cholinesterase inhibitors, neostigmine and edrophonium,
 Reversal of Rocuronium-Induced Neuromuscular Blockade: A Comparison with Glycopyrrolate and Atropine Ozlem Sacan, MD Paul F. White, MD, PhD Burcu Tufanogullari, MD Kevin Klein, MD BACKGROUND: is a modified
Reversal of Rocuronium-Induced Neuromuscular Blockade: A Comparison with Glycopyrrolate and Atropine Ozlem Sacan, MD Paul F. White, MD, PhD Burcu Tufanogullari, MD Kevin Klein, MD BACKGROUND: is a modified
Anesthetic concerns when paralyzing is not an option. By: Ashley Evick, BSN, SRNA
 Anesthetic concerns when paralyzing is not an option By: Ashley Evick, BSN, SRNA Introduction Neuromuscular blockade is utilized in many of the surgeries performed today. There are two types of neuromuscular
Anesthetic concerns when paralyzing is not an option By: Ashley Evick, BSN, SRNA Introduction Neuromuscular blockade is utilized in many of the surgeries performed today. There are two types of neuromuscular
200 mg/20 ml (10 mg/ml) in single-dose vials (3)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use NIMBEX safely and effectively. See full prescribing information for NIMBEX. NIMBEX (cisatracurium
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use NIMBEX safely and effectively. See full prescribing information for NIMBEX. NIMBEX (cisatracurium
Sugammadex: An Update
 The Journal of Critical Care Medicine 2016;2(1):16-21 Sugammadex: An Update BRIEF REPORT DOI: 10.1515/jccm-2016-0005 Tiberiu Ezri 1,2*, Mona Boaz 3, Alexander Sherman 1, Marwan Armaly 4, Yitzhak Berlovitz
The Journal of Critical Care Medicine 2016;2(1):16-21 Sugammadex: An Update BRIEF REPORT DOI: 10.1515/jccm-2016-0005 Tiberiu Ezri 1,2*, Mona Boaz 3, Alexander Sherman 1, Marwan Armaly 4, Yitzhak Berlovitz
Joseph F. Answine, MD
 Joseph F. Answine, MD Joseph F. Answine, MD Staff Anesthesiologist Pinnacle Health Hospitals Harrisburg, PA Clinical Associate Professor of Anesthesiology Pennsylvania State University Hospital Reversal
Joseph F. Answine, MD Joseph F. Answine, MD Staff Anesthesiologist Pinnacle Health Hospitals Harrisburg, PA Clinical Associate Professor of Anesthesiology Pennsylvania State University Hospital Reversal
FOR REPRESENTATIVE EDUCATION
 Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
Anesthesia: Essays and Researches
 A E R Editor-in-Chief : Mohamad Said Maani Takrouri (KSA) Open Access HTML Format For entire Editorial Board visit : http://www.aeronline.org/editorialboard.asp Anesthesia: Essays and Researches Review
A E R Editor-in-Chief : Mohamad Said Maani Takrouri (KSA) Open Access HTML Format For entire Editorial Board visit : http://www.aeronline.org/editorialboard.asp Anesthesia: Essays and Researches Review
Sugammadex: Pharmacology, Safety, and Associated Anesthetic Implications
 Sugammadex: Pharmacology, Safety, and Associated Anesthetic Implications LESSON 24 Volume 39* 4/06/2017 L. Harold Barnwell, DNAP, CRNA Staff Anesthetist and Affiliate Faculty Department of Anesthesiology
Sugammadex: Pharmacology, Safety, and Associated Anesthetic Implications LESSON 24 Volume 39* 4/06/2017 L. Harold Barnwell, DNAP, CRNA Staff Anesthetist and Affiliate Faculty Department of Anesthesiology
Anesthetic management of a patient with Myasthenia Gravis for abdominal surgery.
 The Greek E-Journal of Perioperative Medicine 2017; 16(c): 47-54 (ISSN 1109-6888) www.e-journal.gr/ Ελληνικό Περιοδικό Περιεγχειρητικής Ιατρικής 2017; 16(c): 47-54 (ISSN 1109-6888) www.e-journal.gr/ 47
The Greek E-Journal of Perioperative Medicine 2017; 16(c): 47-54 (ISSN 1109-6888) www.e-journal.gr/ Ελληνικό Περιοδικό Περιεγχειρητικής Ιατρικής 2017; 16(c): 47-54 (ISSN 1109-6888) www.e-journal.gr/ 47
NEUROMUSCULAR BLOCKING AGENTS
 NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
08-15 NORCURON ORGANON
 08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
The influence of unrestricted use of sugammadex on clinical anaesthetic practice in a tertiary teaching hospital
 Anaesth Intensive Care 2012; 40: 333-339 The influence of unrestricted use of sugammadex on clinical anaesthetic practice in a tertiary teaching hospital R. W. WATTS*, J. A. LONDON, R. M. A. W. van WIJK,
Anaesth Intensive Care 2012; 40: 333-339 The influence of unrestricted use of sugammadex on clinical anaesthetic practice in a tertiary teaching hospital R. W. WATTS*, J. A. LONDON, R. M. A. W. van WIJK,
NEOSTIGMINE VERSUS SUGAMMADEX FOR REVERSAL OF NEUROMUSCULAR BLOCK
 EDITORIAL NEOSTIGMINE VERSUS SUGAMMADEX FOR REVERSAL OF NEUROMUSCULAR BLOCK Neostigmine is the classic acetylcholinesterase antagonist, which is widely used for reversal of neuromuscular block of all nondepolarising
EDITORIAL NEOSTIGMINE VERSUS SUGAMMADEX FOR REVERSAL OF NEUROMUSCULAR BLOCK Neostigmine is the classic acetylcholinesterase antagonist, which is widely used for reversal of neuromuscular block of all nondepolarising
New Drugs in Pediatric Anesthesia
 New Drugs in Pediatric Anesthesia Anne M. Lynn MD Seattle Children s Hospital University of Washington School of Medicine There s nothing like a nice, focused topic to start this meeting. So many drugs,
New Drugs in Pediatric Anesthesia Anne M. Lynn MD Seattle Children s Hospital University of Washington School of Medicine There s nothing like a nice, focused topic to start this meeting. So many drugs,
TITLE: Sugammadex for the Reversal of Neuromuscular Blockade in Adult Patients: A Review of Clinical Effectiveness and Cost-Effectiveness
 TITLE: Sugammadex for the Reversal of Neuromuscular Blockade in Adult Patients: A Review of Clinical Effectiveness and Cost-Effectiveness DATE: 26 May 2016 CONTEXT AND POLICY ISSUES Neuromuscular blockade,
TITLE: Sugammadex for the Reversal of Neuromuscular Blockade in Adult Patients: A Review of Clinical Effectiveness and Cost-Effectiveness DATE: 26 May 2016 CONTEXT AND POLICY ISSUES Neuromuscular blockade,
POLICY and PROCEDURE
 Misericordia Community Hospital Administration of Intravenous FentaNYL During Labour POLICY and PROCEDURE Labour and Delivery Manual Original Date Revised Date Approved by: Director, Women s Health, Covenant
Misericordia Community Hospital Administration of Intravenous FentaNYL During Labour POLICY and PROCEDURE Labour and Delivery Manual Original Date Revised Date Approved by: Director, Women s Health, Covenant
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection
 New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
New Zealand Datasheet
 New Zealand Datasheet Name of Medicine ONREX Tablets Ondansetron hydrochloride dihydrate tablets 4mg and 8mg. Presentation ONREX tablets 4 mg: White, circular, biconvex, film coated tablet debossed with
New Zealand Datasheet Name of Medicine ONREX Tablets Ondansetron hydrochloride dihydrate tablets 4mg and 8mg. Presentation ONREX tablets 4 mg: White, circular, biconvex, film coated tablet debossed with
Benztropine and trihexyphenidyl: Centrally acting antimuscarinic agents used for treatment of Parkinson disease & extrapyramidal symptoms.
 Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
Effect of Vecuronium in different age group
 Original Research Article Effect of Vecuronium in different age group Bharti Rajani 1, Hitesh Brahmbhatt 2, Hemlata Chaudhry 2, Hiren Parmar 3* 1 Associate Professor, Department of Anesthesiology, GMERS
Original Research Article Effect of Vecuronium in different age group Bharti Rajani 1, Hitesh Brahmbhatt 2, Hemlata Chaudhry 2, Hiren Parmar 3* 1 Associate Professor, Department of Anesthesiology, GMERS
Are We Safely Dosing Medications in Obese Children? Geisinger Health System Bloomsburg University School of Nurse Anesthesia Kasie Worley, SRNA
 Are We Safely Dosing Medications in Obese Children? Geisinger Health System Bloomsburg University School of Nurse Anesthesia Kasie Worley, SRNA Target Objectives Review the basics of pharmacokinetic
Are We Safely Dosing Medications in Obese Children? Geisinger Health System Bloomsburg University School of Nurse Anesthesia Kasie Worley, SRNA Target Objectives Review the basics of pharmacokinetic
ADVERSE REACTIONS Most common adverse reactions during treatment: nausea, vomiting, and tachycardia. (6)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AKOVAZ safely and effectively. See full prescribing information for AKOVAZ. AKOVAZ (ephedrine sulfate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AKOVAZ safely and effectively. See full prescribing information for AKOVAZ. AKOVAZ (ephedrine sulfate
MUSCLE RELAXANTS. Mr. D.Raju, M.pharm, Lecturer
 MUSCLE RELAXANTS Mr. D.Raju, M.pharm, Lecturer Muscle Relaxants are classified as: I)Peripherally acting A.Neuromuscular blocking agents:- 1) Depolarizing muscle relaxants. 2) Non-depolarizing muscle relaxants
MUSCLE RELAXANTS Mr. D.Raju, M.pharm, Lecturer Muscle Relaxants are classified as: I)Peripherally acting A.Neuromuscular blocking agents:- 1) Depolarizing muscle relaxants. 2) Non-depolarizing muscle relaxants
PRODUCT MONOGRAPH. Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium. Non-depolarizing Skeletal Neuromuscular Blocking Agent
 PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
Surgical Care at the District Hospital. EMERGENCY & ESSENTIAL SURGICAL CARE
 Surgical Care at the District Hospital 1 14 Practical Anesthesia Key Points 2 14.1 General Anesthesia Have a clear plan before starting anesthesia Never use an unfamiliar anesthetic technique in an emergency
Surgical Care at the District Hospital 1 14 Practical Anesthesia Key Points 2 14.1 General Anesthesia Have a clear plan before starting anesthesia Never use an unfamiliar anesthetic technique in an emergency
Problem Based Learning. Problem. Based Learning
 Problem 2013 Based Learning Problem Based Learning Your teacher presents you with a problem in anesthesia, our learning becomes active in the sense that you discover and work with content that you determine
Problem 2013 Based Learning Problem Based Learning Your teacher presents you with a problem in anesthesia, our learning becomes active in the sense that you discover and work with content that you determine
Neuromuscular Junction
 Muscle Relaxants Neuromuscular Junction Cholinergic antagonists Neuromuscular-blocking agents (mostly nicotinic antagonists): interfere with transmission of efferent impulses to skeletal muscles. These
Muscle Relaxants Neuromuscular Junction Cholinergic antagonists Neuromuscular-blocking agents (mostly nicotinic antagonists): interfere with transmission of efferent impulses to skeletal muscles. These
NERVOUS SYSTEM NERVOUS SYSTEM. Somatic nervous system. Brain Spinal Cord Autonomic nervous system. Sympathetic nervous system
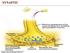 SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
Neuromuscular Blockers
 Neuromuscular Blockers Joanne Leung joanneleung22@hotmail.com Oct 14, 2014 Objectives After this lecture, you should be able to: Describe the physiology of the neuromuscular junction Differentiate the
Neuromuscular Blockers Joanne Leung joanneleung22@hotmail.com Oct 14, 2014 Objectives After this lecture, you should be able to: Describe the physiology of the neuromuscular junction Differentiate the
PROSTIGMIN (neostigmine bromide)
 PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
HST-151 Clinical Pharmacology in the Operating Room
 Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructors: Dr. Carl Rosow, Dr. David Standaert and Prof. Gary Strichartz 1 HST-151 Clinical Pharmacology in
Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructors: Dr. Carl Rosow, Dr. David Standaert and Prof. Gary Strichartz 1 HST-151 Clinical Pharmacology in
ZOFRAN TABLETS GlaxoSmithKline
 ZOFRAN TABLETS GlaxoSmithKline Ondansetron QUALITATIVE AND QUANTITATIVE COMPOSITION ZOFRAN tablets 4 mg: Each tablet contains ondansetron 4 mg as hydrochloride dihydrate. ZOFRAN tablets 8 mg: Each tablet
ZOFRAN TABLETS GlaxoSmithKline Ondansetron QUALITATIVE AND QUANTITATIVE COMPOSITION ZOFRAN tablets 4 mg: Each tablet contains ondansetron 4 mg as hydrochloride dihydrate. ZOFRAN tablets 8 mg: Each tablet
Comparative study of intubating conditions after Rocuronium and Suxamethonium (study of 80 cases)
 ISPUB.COM The Internet Journal of Anesthesiology Volume 20 Number 1 Comparative study of intubating conditions after Rocuronium and Suxamethonium (study of 80 cases) K Bhati, V Parmar Citation K Bhati,
ISPUB.COM The Internet Journal of Anesthesiology Volume 20 Number 1 Comparative study of intubating conditions after Rocuronium and Suxamethonium (study of 80 cases) K Bhati, V Parmar Citation K Bhati,
Succinycholine: Succinylcholine has no place in pediatric anesthesia. Wads Ames MBBS FRCA
 Succinycholine: Succinylcholine has no place in pediatric anesthesia Wads Ames MBBS FRCA Food And Drug Administration Created in 1906 Responsible for protecting and promoting public health through the
Succinycholine: Succinylcholine has no place in pediatric anesthesia Wads Ames MBBS FRCA Food And Drug Administration Created in 1906 Responsible for protecting and promoting public health through the
50% Concentrated Injection
 NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
Neuromuscular Monitoring and Patient Safety:
 Neuromuscular Monitoring and Patient Safety: Pulmonary Complications of Residual Block CEEA Course Tuesday, Sept 4, 2012 Târgu Mureş, Romania Sorin J. Brull, MD, FCARCSI (Hon) Editor, Patient Safety Section
Neuromuscular Monitoring and Patient Safety: Pulmonary Complications of Residual Block CEEA Course Tuesday, Sept 4, 2012 Târgu Mureş, Romania Sorin J. Brull, MD, FCARCSI (Hon) Editor, Patient Safety Section
Ganglion blocking agents
 Ganglion blocking agents -out of date -Specifically act on the nicotinic receptors of both parasymphatetic and sympathetic ganglia - no selectivity toward PG or SG -These drugs are non-depolarizing, competitive
Ganglion blocking agents -out of date -Specifically act on the nicotinic receptors of both parasymphatetic and sympathetic ganglia - no selectivity toward PG or SG -These drugs are non-depolarizing, competitive
Rocuronium allergy. David Spoerl HUG (University Hospital Geneva)
 Rocuronium allergy David Spoerl HUG (University Hospital Geneva) Rocuronium use Sugammadex (Bridion ) Modified γ-cyclodextrin with 8 sugar molecules Designed to encapsulate rocuronium and antagonize its
Rocuronium allergy David Spoerl HUG (University Hospital Geneva) Rocuronium use Sugammadex (Bridion ) Modified γ-cyclodextrin with 8 sugar molecules Designed to encapsulate rocuronium and antagonize its
Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml
 New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
Guide to Neuromuscular Blocking Agents
 Guide to Neuromuscular Blocking Agents EMILY STERLING, PHARMD Associate Clinical Instructor University of Washington School of Pharmacy Seattle, Washington P. SHANE WINSTEAD, PHARMD ICU Clinical Pharmacy
Guide to Neuromuscular Blocking Agents EMILY STERLING, PHARMD Associate Clinical Instructor University of Washington School of Pharmacy Seattle, Washington P. SHANE WINSTEAD, PHARMD ICU Clinical Pharmacy
HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use North American Coral Snake Antivenin (Equine) safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use North American Coral Snake Antivenin (Equine) safely and effectively. See full prescribing information
SUMMARY OF PRODUCT CHARACTERISTICS. 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
Atrovent Administration
 Atrovent Administration ICEMA Training 2007 Sherri Shimshy RN OBJECTIVES Describe the pharmacology of Atrovent Identify the indications for use of Atrovent in the Adult Population Identify the indications
Atrovent Administration ICEMA Training 2007 Sherri Shimshy RN OBJECTIVES Describe the pharmacology of Atrovent Identify the indications for use of Atrovent in the Adult Population Identify the indications
A systematic review of sugammadex vs neostigmine for reversal of neuromuscular blockade
 Anaesthesia 5, 7, 5 Review Article doi:./anae.77 A systematic review of sugammadex vs neostigmine for reversal of neuromuscular blockade A. Abad-Gurumeta, J. Ripolles-Melchor, R. Casans-Frances, A. Espinosa,
Anaesthesia 5, 7, 5 Review Article doi:./anae.77 A systematic review of sugammadex vs neostigmine for reversal of neuromuscular blockade A. Abad-Gurumeta, J. Ripolles-Melchor, R. Casans-Frances, A. Espinosa,
(PP XI) Dr. Samir Matloob
 DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
Title: Management of Allergic Reactions after IV Contrast in Magnetic Resonance Imaging
 ABSTRACT FOR SPS POSTER CASE PRESENTATION K Singer Title: Management of Allergic Reactions after IV Contrast in Magnetic Resonance Imaging Introduction: Children undergoing radiologic imaging frequently
ABSTRACT FOR SPS POSTER CASE PRESENTATION K Singer Title: Management of Allergic Reactions after IV Contrast in Magnetic Resonance Imaging Introduction: Children undergoing radiologic imaging frequently
Help Prevent Medication Errors with PDC Healthcare s Anesthesia Labels & Tapes
 Help Prevent Medication Errors with PDC Healthcare s Anesthesia Labels & Tapes Where positive identification meets safe, efficient patient care. Improve Patient Safety with Accurate Medication Labeling
Help Prevent Medication Errors with PDC Healthcare s Anesthesia Labels & Tapes Where positive identification meets safe, efficient patient care. Improve Patient Safety with Accurate Medication Labeling
Shaded areas=not MARKETED 24/2/09
 PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
The Neuromuscular Junction: Pharmacologic Applications During Anesthesia
 Page 1 The Neuromuscular Junction: Pharmacologic Applications During Anesthesia Cynthia A. Lien, M.D. New York, NewYork Introduction The neuromuscular junction and neuromuscular transmission are well-studied
Page 1 The Neuromuscular Junction: Pharmacologic Applications During Anesthesia Cynthia A. Lien, M.D. New York, NewYork Introduction The neuromuscular junction and neuromuscular transmission are well-studied
receptors determines clinical symptoms of the patients and modulates the sensitivity to nondepolarizing neuromuscular
 346 Anesthesiology 2000; 93:346 50 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Preanesthetic Train-of-four Fade Predicts the Atracurium Requirement of Myasthenia
346 Anesthesiology 2000; 93:346 50 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Preanesthetic Train-of-four Fade Predicts the Atracurium Requirement of Myasthenia
Case Report Laparoscopic Sleeve Gastrectomy in a Morbidly Obese Patient with Myasthenia Gravis: A Review of the Management
 Case Reports in Medicine Volume 2015, Article ID 593586, 4 pages http://dx.doi.org/10.1155/2015/593586 Case Report Laparoscopic Sleeve Gastrectomy in a Morbidly Obese Patient with Myasthenia Gravis: A
Case Reports in Medicine Volume 2015, Article ID 593586, 4 pages http://dx.doi.org/10.1155/2015/593586 Case Report Laparoscopic Sleeve Gastrectomy in a Morbidly Obese Patient with Myasthenia Gravis: A
ROSOBAC-1GM / ROSOBAC-FORT
 ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
Population pharmacokinetic pharmacodynamic analysis for sugammadex-mediated. reversal of rocuronium-induced. neuromuscular blockade
 British Journal of Clinical Pharmacology DOI:./j.365-225.20.04000.x Population pharmacokinetic pharmacodynamic analysis for sugammadex-mediated reversal of rocuronium-induced neuromuscular blockade Huub
British Journal of Clinical Pharmacology DOI:./j.365-225.20.04000.x Population pharmacokinetic pharmacodynamic analysis for sugammadex-mediated reversal of rocuronium-induced neuromuscular blockade Huub
DO WE NEED TO USE SUGAMMADEX AT THE END OF A GENERAL ANESTHESIA TO REVERSE THE ACTION OF NEUROMUSCULAR BLOKING AGENTS?
 1 Experts opinion and Point of view are papers reviewing the literature, usually invited by the Editor in Chief and do not exceed 3000 and 2000 words with 50 and 40 references respectively. parole: 2666
1 Experts opinion and Point of view are papers reviewing the literature, usually invited by the Editor in Chief and do not exceed 3000 and 2000 words with 50 and 40 references respectively. parole: 2666
Printed copies of this document may not be up to date, obtain the most recent version from
 Children s Acute Transport Service Clinical Guidelines Acute Severe Asthma Document Control Information Author E Randle Author Position CATS Consultant Document Owner E Polke Document Owner Position Co-ordinator
Children s Acute Transport Service Clinical Guidelines Acute Severe Asthma Document Control Information Author E Randle Author Position CATS Consultant Document Owner E Polke Document Owner Position Co-ordinator
Introduction Corresponding author:
 Anesthetic Management Of Aortic Valve Replacement In A Myasthenia Gravis Patient, The Era Of A New Reversal Alia S. Dabbous 1*, Patricia W Nehme 1 and Ahmad M Abou Leila 2 A 66 year old man diagnosed with
Anesthetic Management Of Aortic Valve Replacement In A Myasthenia Gravis Patient, The Era Of A New Reversal Alia S. Dabbous 1*, Patricia W Nehme 1 and Ahmad M Abou Leila 2 A 66 year old man diagnosed with
Zekeriyya Alanoglu. Definition of Residual Neuromuscular Block
 Postoperative Residual Neuromuscular Block Zekeriyya Alanoglu Almost 60 years ago the use neuromuscular blocking agents (NMBA) was associated with significantly increased risk of perioperative (approx.
Postoperative Residual Neuromuscular Block Zekeriyya Alanoglu Almost 60 years ago the use neuromuscular blocking agents (NMBA) was associated with significantly increased risk of perioperative (approx.
Farmadol. Paracetamol 10 mg/ml INFUSION SOLUTION
 Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
NIMBEX Injection. This information is intended for U.S. residents only. only. (cisatracurium besylate)
 This information is intended for U.S. residents only. NIMBEX Injection (cisatracurium besylate) only DESCRIPTION CLINICAL PHARMACOLOGY INDICATIONS AND USAGE CONTRAINDICATIONS WARNINGS PRECAUTIONS ADVERSE
This information is intended for U.S. residents only. NIMBEX Injection (cisatracurium besylate) only DESCRIPTION CLINICAL PHARMACOLOGY INDICATIONS AND USAGE CONTRAINDICATIONS WARNINGS PRECAUTIONS ADVERSE
PHA Second Exam. Fall On my honor, I have neither given nor received unauthorized aid in doing this assignment.
 PHA 5127 Second Exam Fall 2012 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Put all answers on the bubble sheet TOTAL /150 pts 1 Question Set I (True or
PHA 5127 Second Exam Fall 2012 On my honor, I have neither given nor received unauthorized aid in doing this assignment. Name Put all answers on the bubble sheet TOTAL /150 pts 1 Question Set I (True or
Goal-Directed Fluid Therapy: A New Way of Thinking. Ji Su Jenny Kim & Logan D. MacLean SRNA, BSN, CCRN DNP Candidates
 Goal-Directed Fluid Therapy: A New Way of Thinking Ji Su Jenny Kim & Logan D. MacLean SRNA, BSN, CCRN DNP Candidates Goal-Directed Fluid Therapy Map History of Fluid Management Significance of Fluid Management
Goal-Directed Fluid Therapy: A New Way of Thinking Ji Su Jenny Kim & Logan D. MacLean SRNA, BSN, CCRN DNP Candidates Goal-Directed Fluid Therapy Map History of Fluid Management Significance of Fluid Management
NEW ZEALAND DATA SHEET. Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution containing 2.5 mg/ml Neostigmine methylsulphate.
 NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
TRACRIUM GlaxoSmithKline
 TRACRIUM GlaxoSmithKline Atracurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: A sterile solution containing 10 mg atracurium besylate per ml, without an antimicrobial preservative, supplied in
TRACRIUM GlaxoSmithKline Atracurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: A sterile solution containing 10 mg atracurium besylate per ml, without an antimicrobial preservative, supplied in
PARACOD Tablets (Paracetamol + Codeine phosphate)
 Published on: 22 Sep 2014 PARACOD Tablets (Paracetamol + Codeine phosphate) Composition PARACOD Tablets Each effervescent tablet contains: Paracetamol IP...650 mg Codeine Phosphate IP... 30 mg Dosage Form/s
Published on: 22 Sep 2014 PARACOD Tablets (Paracetamol + Codeine phosphate) Composition PARACOD Tablets Each effervescent tablet contains: Paracetamol IP...650 mg Codeine Phosphate IP... 30 mg Dosage Form/s
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION
 AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
Awake regional versus general anesthesia in preterms and ex-preterm infants for herniotomy
 Awake regional versus general anesthesia in preterms and ex-preterm infants for herniotomy Department of Anaesthesia University Children s Hospital Zurich Switzerland Epidemiology Herniotomy needed in
Awake regional versus general anesthesia in preterms and ex-preterm infants for herniotomy Department of Anaesthesia University Children s Hospital Zurich Switzerland Epidemiology Herniotomy needed in
References. This guideline booklet is made possible by an unrestricted educational grant from
 References Revised Edition 3 Pühringer FK, Kristen P, Rex C. Sugammadex Reversal of Rocuronium- Induced Neuromuscular Block in Caesarean Section Patients: A Series of Seven Cases; Br J Anaesth. 2010 Nov;105(5):657-60.
References Revised Edition 3 Pühringer FK, Kristen P, Rex C. Sugammadex Reversal of Rocuronium- Induced Neuromuscular Block in Caesarean Section Patients: A Series of Seven Cases; Br J Anaesth. 2010 Nov;105(5):657-60.
A Comparative Study between Propofol and Thiopentone as Induction Agents in Myasthenia Gravis Patients for Thymectomy
 Original Article DOI: 1.17354/ijss/15/278 A Comparative Study between Propofol and Thiopentone as Induction Agents in Myasthenia Gravis Patients for Thymectomy Venkata Sesha Sai Krishna Manne 1, Madhavi
Original Article DOI: 1.17354/ijss/15/278 A Comparative Study between Propofol and Thiopentone as Induction Agents in Myasthenia Gravis Patients for Thymectomy Venkata Sesha Sai Krishna Manne 1, Madhavi
ZOFRAN INJECTION GlaxoSmithKline
 ZOFRAN INJECTION GlaxoSmithKline Ondansetron QUALITATIVE AND QUANTITATIVE COMPOSITION Each 1 ml of aqueous solution contains 2 mg ondansetron as hydrochloride dihydrate. PHARMACEUTICAL FORM A clear, colourless,
ZOFRAN INJECTION GlaxoSmithKline Ondansetron QUALITATIVE AND QUANTITATIVE COMPOSITION Each 1 ml of aqueous solution contains 2 mg ondansetron as hydrochloride dihydrate. PHARMACEUTICAL FORM A clear, colourless,
Cholinergic antagonists
 Cholinergic antagonists Cholinergic antagonists: They are also called anticholinergic drugs or cholinergic blockers, this group include: 1.Antimuscarinic agents ( atropine, ipratropium, scopolamine) 2.
Cholinergic antagonists Cholinergic antagonists: They are also called anticholinergic drugs or cholinergic blockers, this group include: 1.Antimuscarinic agents ( atropine, ipratropium, scopolamine) 2.
